Contents
- What is double outlet right ventricle
- Double outlet right ventricle types
- Double outlet right ventricle survival rate
- Double outlet right ventricle life expectancy
- Double outlet right ventricle causes
- Double outlet right ventricle symptoms
- Double outlet right ventricle diagnosis
- Double outlet right ventricle treatment
- Double outlet right ventricle surgery
What is double outlet right ventricle
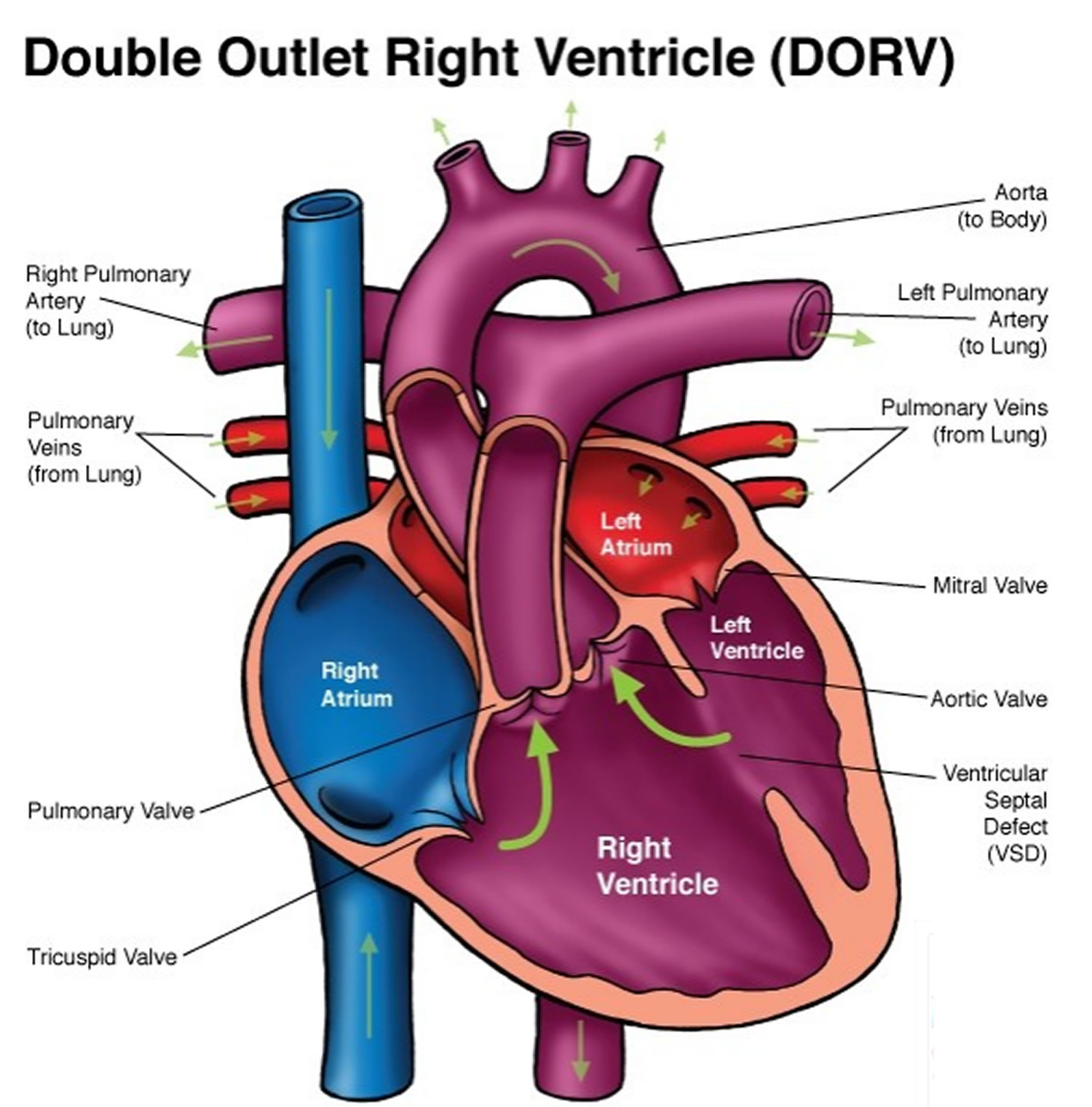
Double outlet right ventricle is a group of complex congenital (present at birth) heart abnormalities. In double-outlet right ventricle, the aorta and pulmonary artery connect partially or completely to the right ventricle 1). The position and size of the great arteries vary and other cardiac malformations are commonly present and include pulmonary outflow or left ventricular outflow obstruction, and major atrioventricular valve abnormalities. The aorta may be on the right or left and may be anterior, posterior, or lateral. In addition double outlet right ventricle is commonly associated with syndromes and chromosomal abnormalities. A hole also exists between the two ventricles (ventricular septal defect). In a normal heart, as shown on the left, the pulmonary artery connects to the right ventricle and the aorta connects to the left ventricle (Figure 1). Double outlet right ventricle accounts for less than 1% of all congenital heart defects 2). The mean incidence is 4 to 1,000 to 50 to 1,000 live births 3).
In double-outlet right ventricle a heart condition present at birth (congenital) — the main artery that carries blood from the heart to the body (aorta) and the artery that directs blood from the heart to the lungs (pulmonary artery) are partially or completely connected to the lower right heart chamber (right ventricle). Sometimes these blood vessels are also reversed from their normal positions (transposed). In a normal heart, the pulmonary artery is connected to the right ventricle and the aorta is connected to the left ventricle.
In people with double-outlet right ventricle, there is also a hole between the lower heart chambers (ventricles), called a ventricular septal defect (VSD), which can be located in several places in the wall between the ventricles. This causes oxygen-rich blood to flow from the left ventricle to the right ventricle and mix with oxygen-poor blood. Due to this mixing, children born with double-outlet right ventricle can have oxygen saturations that are lower than normal.
Too much blood flow may then be delivered through the pulmonary artery to the lungs, causing heart failure and poor growth over time. In other cases, blood flow through the pulmonary artery may be reduced, which can cause your child’s skin to turn a bluish color (cyanosis).
Double outlet right ventricle is initially associated with excessive pulmonary blood flow and frequent recurrent pulmonary infections 4). Cyanosis may be present soon after birth. The onset of pulmonary hypertension often leads to a progressive reduction in exercise capacity and fatigue resulting from impaired respiratory and heart function, which cause stunted childhood development. The disorder is complex and is often associated with other forms of intracardiac or extracardiac malformation. Pulmonary hypotension usually arises early and eventually results in heart and lung dysfunction, with a missed opportunity to performing surgery. It is generally considered that these patients often develop significant congestive heart failure due to a large left-to-right shunt, and surgical repair should be carried out in the first several months of life 5).
Your child will need surgery to correct this defect and any associated defects. Some children will require surgery in the first few days after birth, and others may have surgery at a few months of age.
Some people may have other associated congenital heart defects, including other holes in the heart (such as an atrial septal defect), heart valve problems or blood vessel problems. The presence of other heart defects may change the medications used and type of surgery performed.
Figure 1. Double outlet right ventricle
Note: Both the pulmonary artery (PA) and aorta (AO) arise from the right ventricle in double-outlet right ventricle (DORV). Blood from the left ventricle must exit through a large ventricular septal defect (VSD). Most often the great arteries, PA and AO, are side by side and separated by the conus septum. In double-outlet right ventricle, the pulmonary and aortic valves are at the same level. Subtypes of double-outlet right ventricle are based upon the position of the VSD and presence or absence of pulmonary stenosis or right ventricular outflow tract obstruction (RVOTO).
Double outlet right ventricle with subpulmonary VSD: Fetal echocardiogram
Double outlet right ventricle types
These two great arteries – the aorta and pulmonary artery – may override the ventricular septum by more than 50%, 90%, or both arteries may arise fully from the right ventricle (200%) 6). Double outlet right ventricle is frequently associated with other intracardiac or extracardiac malformations.
The current double outlet right ventricle classification system is based on the international nomenclature databases adopted by the Society of Thoracic Surgeons and the European Association of Cardiothoracic Surgery 7). There are four types of double outlet right ventricle 8):
- Tetralogy of Fallot type,
- Transposition of the great arteries type,
- Ventricular septal defect [VSD] type, and
- Remote VSD type have been classified
These four types differ significantly in their surgical treatment, postoperative management, and prognosis. Except for Tetralogy of Fallot type, which usually has pulmonary hypertension, the other three types frequently induce pulmonary hypertension. Preoperative pulmonary hypertension was concerned as an important risk factor for early deaths in the surgical treatment of double outlet right ventricle 9).
Double outlet right ventricle survival rate
In a series 10) of 61 patients (36 males and 25 females) aged 2 weeks to 26 years with double outlet right ventricle (two great arteries overriding at least 50%) complicated by pulmonary hypertension who underwent surgical treatment, 31 of 36 patients aged <1 year recovered uneventfully. However, 5 of 36 infants died (13.9%), 3 of the 36 infant patients underwent secondary surgery to correct postoperative complications (two patients underwent successful closure of a residual shunt in the intracardiac channel, and one patient was revised for a residual shunt in the intracardiac channel combined with enlarging an right ventricle outflow tract stenosis) 11). In contrast to the infant group above, all operations were successfully performed in the 22 patients aged between 1 and 14 years, including in 3 patients who had undergone a previous banding procedure. No patients died or required further surgery. Senning procedure and Mustard procedure were performed in three adult patients to reverse the features of hypoxia; one died from low cardiac output syndrome 12). Although the average survival time after diagnosis of pulmonary hypertension was estimated to be 5–7 years nowadays, the mortality was raised significantly after 3 years old 13), 14), 15), 16). Therefore, according to that study observation 17), it was safe for patients with double outlet right ventricle and pulmonary hypertension to receive surgical treatment between 1 and 3 years old. Younger infants and adult patients may have higher mortality.
Double outlet right ventricle life expectancy
How well the baby does depends on:
- The size and location of the VSD
- The size of the pumping chambers
- The location of the aorta and pulmonary artery
- The presence of other complications (such as coarctation of the aorta and mitral valve problems)
- The baby’s overall health at the time of diagnosis
- Whether lung damage has occurred from too much blood flowing to the lungs for a long period of time
Double outlet right ventricle possible complications
Complications from double outlet right ventricle may include:
- Heart failure
- High blood pressure in the lungs (pulmonary hypertension), which untreated can lead to permanent lung damage
- Death
Double outlet right ventricle causes
In a normal heart structure, the aorta connects to the left ventricle. The pulmonary artery normally is connected to the right ventricle. In double outlet right ventricle, both arteries flow out of the right ventricle. This is a problem because the right ventricle carries oxygen-poor blood. This blood is then circulated throughout the body.
Another defect called a ventricular septal defect (VSD) always occurs with double outlet right ventricle. Other conditions that may be part of the defect are pulmonary valve stenosis and transposition of the great arteries.
Oxygen-rich blood from the lungs flows from the left side of the heart, through the VSD opening and into the right ventricle. This helps the infant with double outlet right ventricle by allowing oxygen-rich blood to mix with oxygen-poor blood. Even with this mixture, the body may not get enough oxygen. This makes the heart work harder to meet the body’s needs. There are several types of double outlet right ventricle.
The difference between these types is the location of the VSD as it relates to the location of the pulmonary artery and aorta. The symptoms and severity of the problem will depend on the type of double outlet right ventricle. The presence of pulmonary valve stenosis also affects the condition.
People with double outlet right ventricle often have other heart defects, such as:
- Endocardial cushion defects (the walls separating all 4 chambers of the heart are poorly formed or absent)
- Coarctation of the aorta (narrowing of the aorta)
- Mitral valve problems
- Pulmonary atresia (pulmonary valve does not form properly)
- Pulmonary valve stenosis (narrowing of the pulmonary valve)
- Right-sided aortic arch (aortic arch is on right instead of the left)
- Transposition of the great arteries (the aorta and pulmonary artery are switched)
Double outlet right ventricle symptoms
Common symptoms of double outlet right ventricle in babies include:
- rapid breathing
- rapid heartbeat
- sweating
- disinterest in feeding or tiring while feeding
- poor weight gain
- blue color of the skin, lips and nail beds (cyanosis)
- heart murmur (detected by doctor)
In older children, symptoms may include:
- fatigue
- shortness of breath
Double outlet right ventricle diagnosis
To diagnose double-outlet right ventricle, doctors will generally perform an echocardiogram. Echocardiograms use sound waves to produce an image of the heart. Sound waves bounce off the heart and produce moving images that can be viewed on a video screen.
This test can help doctors to look closely at the heart, including the ventricles, aorta and pulmonary arteries as well as the heart valves. Doctors often use this test to diagnose this condition and any associated defects, and determine the appropriate treatment.
If more information is needed, doctors may conduct cardiac CT and MRI scans, or cardiac catheterization. In cardiac catheterization, your child’s doctor inserts a thin, flexible tube (catheter) into an artery or vein in the groin or neck and threads it into the heart. A dye is injected through the catheter to make the heart structures more visible on X-ray pictures. Cardiac catheterization also measures pressure and oxygen levels in the chambers of the heart and in the blood vessels.
Double outlet right ventricle treatment
Several types of surgery may be performed, depending on the specific heart defect and any associated defects.
Factors that determine the type and number of operations the baby needs include:
- The type of double outlet right ventricle
- The severity of the defect
- The presence of other problems in the heart
- The child’s overall condition
Depending on the type of defect, surgeons may:
- Create a tunnel (baffle) through the VSD to connect the left ventricle to the aorta
- Switch the aorta and pulmonary artery positions, if they are in reversed positions, in order to connect the pulmonary artery to the right ventricle and the aorta to the left ventricle
- Insert a patch between the ventricles to close the hole between the ventricles (VSD)
- Insert a blood vessel to connect the right ventricle to the pulmonary artery to allow more blood flow if the pulmonary artery is small or absent
- Widen the narrowed pulmonary artery to allow more blood flow
- Place an aorta to pulmonary artery shunt to provide more blood flow to the pulmonary arteries
- Conduct a series of other procedures to allow blood to move to the lungs and for the heart to function with one ventricle, if the defect is complex
- Repair any other congenital heart defects, such as atrial septal defects or patent ductus arteriosus
Another procedure, called an arterial switch operation, is necessary if the aorta and the pulmonary artery are reversed in relation to each other. For this procedure, a tunnel from the VSD to the pulmonary artery is created, connecting the left ventricle to the pulmonary artery. Then, the vessels are disconnected and reconnected so that the pulmonary artery becomes the aorta and the aortic valve is connected to the pulmonary artery, and the holes between the left and right ventricles of the heart are closed.
In some infants with inadequate blood flow to their lungs at birth, a temporary procedure may be done to insert a shunt between the aorta and the pulmonary artery. The shunt is then removed later in life during your child’s definitive cardiac surgery. If your child has other heart conditions associated with double outlet right ventricle, he or she may also need other types of surgery.
Adults who were born with double-outlet right ventricle will need lifelong care and regular follow-up evaluations from cardiologists trained in evaluating and treating congenital heart conditions (adult congenital cardiologists) to monitor for any changes in their condition. Surgery may need to be performed later in life for valve disease such as narrowing or leakage of the heart valves. Some adults need close monitoring of their aortas and pulmonary arteries, especially if they required surgery early in life. A small group of adults may require medications for treatment of decreased function involving the right or left ventricles.
Double outlet right ventricle surgery
Regardless of the double outlet right ventricle classification system used, the choice of surgical approach depends on 3 factors:
- the relative positions of the great arteries,
- the relationship between the arteries and the ventricular septal defect (VSD), and
- the presence or absence of right ventricular outflow tract obstruction.
The four types of double outlet right ventricle 18):
- Tetralogy of Fallot type,
- Transposition of the great arteries type,
- Ventricular septal defect [VSD] type, and
- Remote VSD type
The first 2 types of double outlet right ventricle—the VSD type and the Tetralogy of Fallot type—have clear surgical options. However, the choice of surgical treatment for double outlet right ventricle with transposition of the great arteries is more difficult because right ventricular outflow tract obstruction may also be present; the current classification system does not adequately account for this possibility. This is especially problematic for patients with a noncommitted VSD and abnormal great arteries because they might need arterial switch operations or double-root translocations 19).
Choice of surgical method and prevention of complications
Pathological changes in patients with double outlet right ventricle are varied, necessitating an individualized surgical strategy to optimize success. The suture technique used in intracardiac channel repair of VSD type double outlet right ventricle should be sufficient to prevent residual shunt formation. In cases of restricted VSD, enlargement of the outflow defect should be carried out, with attention paid to avoid outflow tract stenosis from both the left and right ventricular chambers. To avoid right ventricle outflow tract stenosis, the tract should simultaneously be enlarged with a patch. Eight of our patients in this group received right ventricle outflow tract patch widening. For patients with double outlet right ventricle of remote VSD type, a total intracardiac conduit may be needed. For transposition of the great arteries type double outlet right ventricle, an arterial switch operation remains the best choice. During the operation, coronary anastomotic patency should be confirmed, and care taken to prevent distortion and stenosis. Aortic and pulmonary valves should be protected. It is important to avoid stenosis of the pulmonary artery and its branches during pulmonary artery anastomosis. If the aorta and pulmonary artery are located in an anterior and posterior position and the VSD is below the pulmonary valve, arterial switch surgery can be performed after establishing an intracardiac channel.
Mustard or Senning surgical techniques are generally safe because of their relative simplicity. Both methods are useful for neglected cases with severe pulmonary hypertension, cyanosis, and hypoxia, in which the best opportunity for surgery has been missed. These palliative procedures will improve hypoxia and overall quality of life; however, the operative risk is high. During surgery, attention should be paid to prevent lung injury and maintain cardiac function; consequently, VSD closure can be avoided. One patient with mild pulmonary hypertension (<50 mmHg) was deemed suitable to undergo Rastelli surgery, with careful paid to prevent distortion and stenosis of intracardiac and extracardiac channels that would normally require reoperation. double outlet right ventricle is often associated with other intracardiac malformations such as atrial septal defect (ASD), patent ductus arteriosus (PDA), and atrioventricular valve regurgitation, which require concurrent correction.
Table 1. Surgical method selection with different types of double outlet right ventricle
| Operation selection | Double outlet right ventricle type, n | ||
|---|---|---|---|
| VSD | TGA | Remote VSD | |
| Operation type | |||
| Corrective surgery (intracardiac channel) | 30 | 1 | 6 |
| Switch | 0 | 19 | 0 |
| Senning | 0 | 3 | 0 |
| Mustard | 0 | 1 | 0 |
| Rastelli | 0 | 1 | 0 |
| Previous operation | |||
| Banding | 1 | 0 | 0 |
| Banding + shunt | 0 | 1 | 0 |
| Banding + correction of CoA | 0 | 1 | 0 |
| Secondary operation | |||
| RRS | 0 | 0 | 2 |
| RRS + ERVOTO | 1 | 0 | 0 |
Notes: DORV: Double outlet right ventricle; CoA: Coarctation of aorta; RRS: Repair of VSD residual shunt; ERVOTO: Enlargement of right ventricle outlet tract obstruction; VSD: Ventricular septal defect; TGA: Transposition of the great arteries.
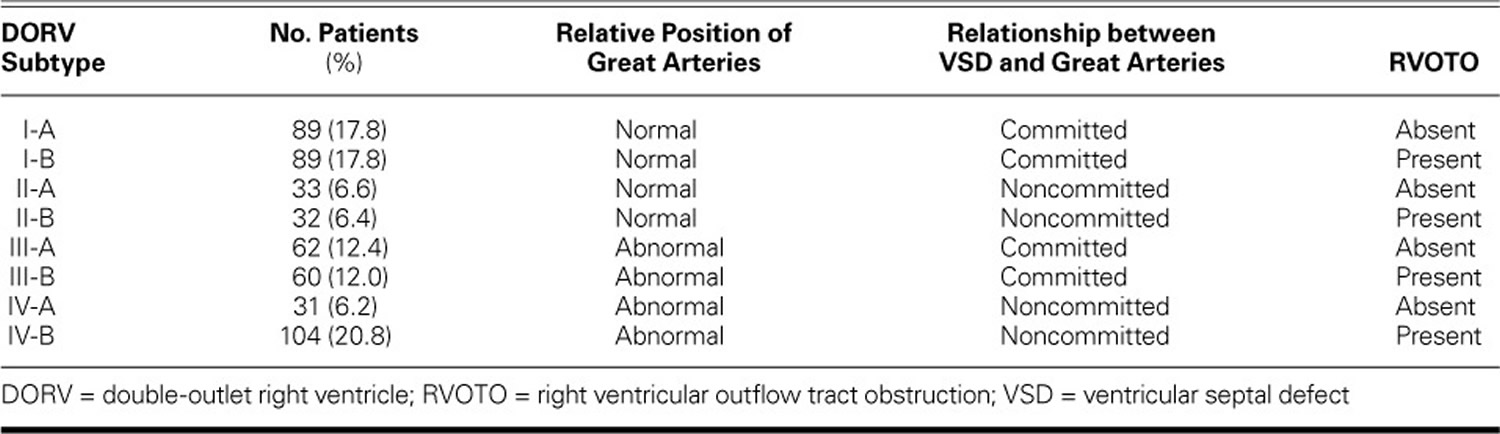
In another study 21) from February 2001 through July 2014 involving 500 patients diagnosed with double outlet right ventricle who underwent treatment at the Fuwai Hospital. Of the 500 patients, there were 299 males and 201 females. The mean age at diagnosis was 4.08 years (range, 6d–42 yr), and the mean weight was 13.3 ± 11.39 kg (range, 2.1–94 kg). The patients’ oxygen saturation levels ranged from 25% to 94% without supplemental oxygen 22). Diagnosis was based on 2-dimensional echocardiographic evidence. Cardiac catheterization and CT angiography were performed when necessary. To overcome the limitations of the Society of Thoracic Surgeons and the European Association of Cardiothoracic Surgery double outlet right ventricle classification for patients with transposition of the great arteries who may also have right ventricular outflow tract obstruction, the doctors at Fuwai Hospital 23) developed an echocardiographic classification system based on the spatial relationships of the great arteries, the relationship of the VSD to the arteries, and the morphology of the right ventricular outflow tract 24). They then compared their findings with those obtained through cardiac catheterization, computed tomographic (CT) angiography, and intraoperative inspection.
Table 2. Modified Fuwai Classification in 500 Patients with Double-Outlet Right Ventricle
[Source 25)]Of the 500 patients, 407 (81.4%) underwent surgery. Of those, 272 (66.8%) had biventricular repair, 99 (24.3%) had single-ventricle repair, and 36 (8.8%) had palliative procedures. At the time of surgery, the patients’ ages ranged from 4 months to 42 years, and 208 (51.1%) were younger than 2 years old. Cardiac catheterization and CT angiography were performed in 264 (52.8%) and 59 (11.8%) of the 500 patients, respectively. When we compared the findings of cardiac catheterization, CT angiography, and intraoperative inspection with the diagnosis made through preoperative echocardiography, we found agreement regarding the relative position of the great arteries in 414 of 414 patients (accuracy rate, 100%), and we found agreement regarding the relationship between the great arteries and the VSD in 347 of 378 patients (accuracy rate, 91.8%).
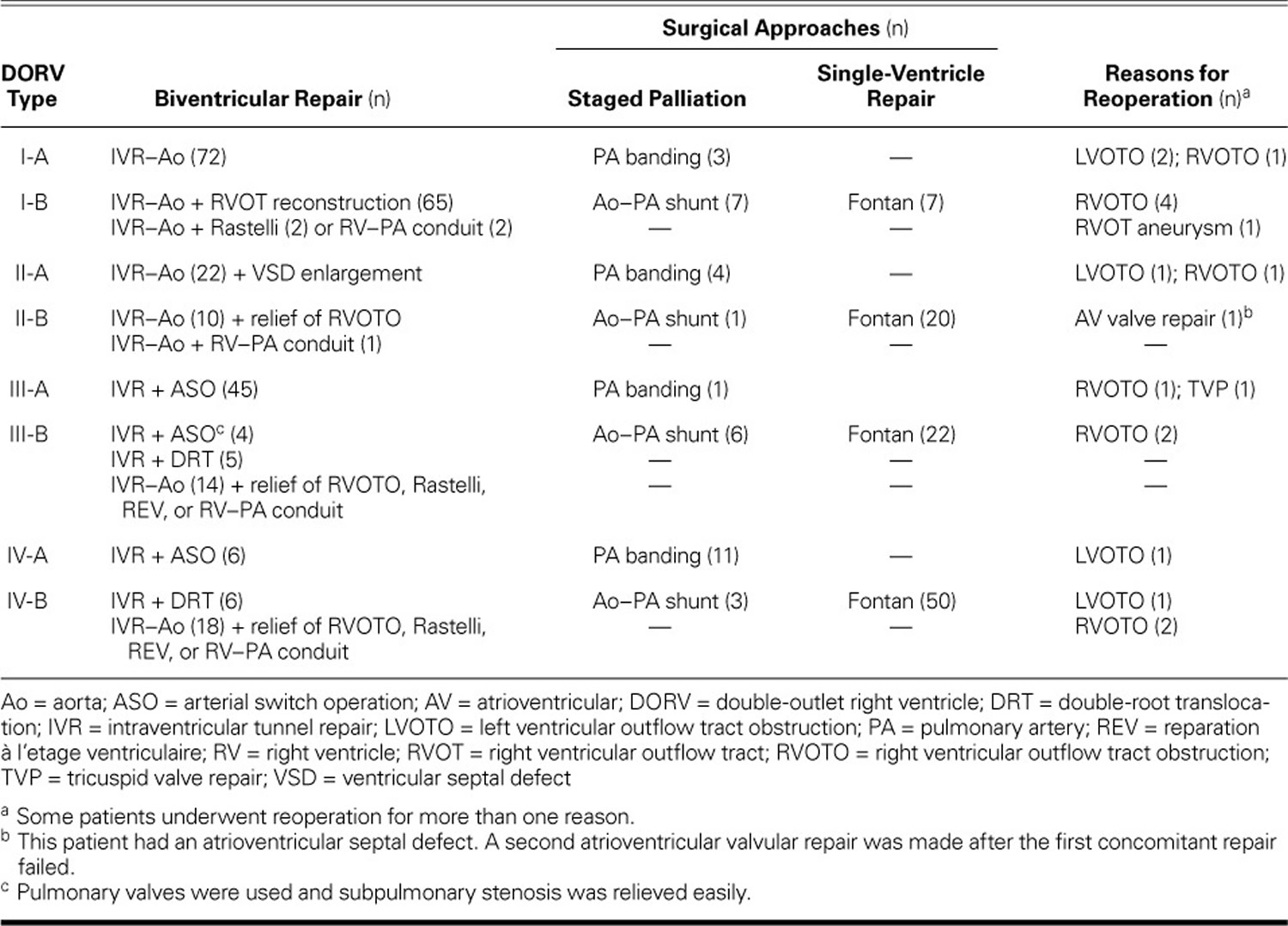
Table 3 shows the surgical management protocols 26) for each subtype. The surgical success rate was 92.1% for the double outlet right ventricle operations performed on patients categorized according to the modified Fuwai echocardiographic classification system. Of note, the surgical treatment of patients with double outlet right ventricle subtypes I-A, I-B, and III-A was similar.
In 22 patients with type II-A double outlet right ventricle, biventricular repairs were made; this involved intraventricular tunnel repair (IVR) from the left ventricle to the aorta and some additional techniques as needed, including enlargement of the VSD, use of multiple patches on the tunnel,8 resection of muscular bands, or reattachment of the tricuspid valve’s chordae tendineae. An intraventricular tunnel repair from the left ventricle to the aorta was performed in 11 patients with type II-B, in 14 with type III-B, and in 18 with type IV-B double outlet right ventricle; these patients also underwent concomitant reconstruction of the right ventricle outflow tract performed by means of a reparation à l’etage ventriculaire (REV) procedure, Rastelli procedure, or RV–PA conduit. The tricuspid papillary muscle was reimplanted on the tunnel patch in 2 patients with type II-A double outlet right ventricle, in one with type II-B, and in one with type IV-B.
Double-root translocation with baffling of the VSD to the neo-aorta was performed in 5 patients with type III-B and in 6 patients with type IV-B double outlet right ventricle. An arterial switch operation (ASO) was performed after successful relief of pulmonary valvular and subpulmonary stenosis in 4 patients with type III-B and in 6 patients with type IV-A double outlet right ventricle. Atrioventricular discord was present in 2 patients with type III-B and in one patient with type IV-B double outlet right ventricle; they underwent a Mustard or Senning atrial baffle procedure 27).
Follow-up information was available for 396 patients (mean, 1.1 yr; range, 0.5 mo–10.8 yr). During this time, 10 patients underwent surgery to relieve subaortic or subpulmonary stenosis and to resect an right ventricle outflow tract aneurysm 28).
Table 3. Surgical Protocols and Reasons for Reoperation by Type of double outlet right ventricle
[Source 29)]For patients with type I-A double outlet right ventricle who had aortic–mitral muscular separation or long fibrous continuity, the optimal surgical approach was an intraventricular tunnel repair from the left ventricle to the aorta; if a restrictive VSD was present, it was enlarged superiorly or anteriorly. Patients with type I-B double outlet right ventricle had a significant anterior malposition of the aorta that was repaired by using techniques similar to those described for the repair of tetralogy of Fallot.
Type II double outlet right ventricle was characterized by normal spatial relationships of the great arteries and a remote VSD, and was repaired with a long tunnel connecting the left ventricle to the aorta. In patients with type II double outlet right ventricle, the width of the tunnel should be larger than that of the aortic annulus, and the tunnel should avoid the tricuspid inflow chamber 30), as determined echocardiographically. In some cases, a tunnel necessitated reimplantation of the tricuspid papillary muscle on the tunnel patch, as well as VSD enlargement. Evaluating the requirements for tunnel creation was the most important aspect of using echocardiograms for surgical planning. In our patients with type II-A double outlet right ventricle, the very remote VSDs were always in the inlet septum without extension to the perimembranous region; thus, anatomic repair was ill-advised. In these circumstances, pulmonary artery banding should be attempted first in a young child or infant, because this enables single-ventricle repair in the future. In both type II-A and type II-B patients, intraventricular tunnel repair-to-aorta techniques can be performed, with additional relief of right ventricular outflow tract obstruction. In type II double outlet right ventricle—as in type I double outlet right ventricle—the relationship of the great arteries is normal, so no arterial switch operation procedures are involved.
Because type III and type IV double outlet right ventricle are characterized by abnormal positions of the great arteries, bi-ventricular repair might necessitate reestablishing the normal spatial relations of the great arteries. Most patients with type III-A double outlet right ventricle have Taussig-Bing malformations, with 50% to 100% of the pulmonary artery overriding the VSD and no right ventricular outflow tract obstruction. These patients are always treated with an intraventricular tunnel repair and an arterial switch operation 31). Type III-B double outlet right ventricle is characterized by the presence of right ventricular outflow tract obstruction, as well as a pulmonary artery overriding the VSD, which could be easily confused with a transposition of the great arteries in association with pulmonary stenosis. In our opinion, the diagnosis of type III-B double outlet right ventricle should be made only in patients with a muscular or long pulmonary–mitral fibrous connection and a ≥90% overriding of the pulmonary artery. Because subaortic coni are always present in these patients, a double-root translocation is the optimal treatment.15 If preoperative echocardiograms show the 2 semilunar valvular annuli without well-developed, subaortic muscular coni, an intraventricular tunnel repair to the aorta—in conjunction with other procedures, including VSD enlargement, muscular band resection and reparation à l’etage ventriculaire, Rastelli procedures, or an RV–pulmonary artery conduit—might be indicated instead.
Type IV-A double outlet right ventricle is similar to type III-A in that it is characterized by transposition of the great arteries; however, it also has a remote VSD in which both great arteries arise from the RV with bilateral coni. In our type IV-A patients, we performed a biventricular repair to connect the left ventricle with the pulmonary artery, and we then performed an arterial switch operation. Of note, the palliative repairs and the therapeutic regimens that we used for patients with type II-A and IV-A double outlet right ventricle were similar.
Type IV-B double outlet right ventricle—the most complex subset of double outlet right ventricle in our cohort—was similar to type IV-A with right ventricular outflow tract obstruction and needed 1) the use of multiple patches, resection of muscular bands, and reattachment of the tricuspid valve’s chordae tendinae; or 2) the reestablishment of RV-to-pulmonary artery continuity by means of reparation à l’etage ventriculaire or Rastelli techniques. As is the case in patients with type II-A and IV-A double outlet right ventricle, echocardiograms can help to establish the size of the tunnel. If patients with type IV-B double outlet right ventricle have well-developed subaortic coni, a double-root translocation with tunneling of the VSD to the pulmonary artery is preferable.
In our cohort, patients with associated atrioventricular septal defects had mainly Rastelli type-C atrioventricular septal defects, which can occur in every type of double outlet right ventricle except type III-A. The VSD in these patients is usually in the inlet septum, far from both arteries, and is further separated from the great arteries by the tricuspid inflow chamber 32). These very remote VSDs cannot be connected to the great arteries unless a large superior component extends close to the aortic valve annulus. In that study, 10 of 51 patients with an atrioventricular septal defects underwent complete biventricular repairs despite the complex techniques necessary during the surgery.
Postoperative management
The postoperative management of these patients is very important. Routine use of inhaled nitric oxide and prostaglandins is helpful. Diuretic drugs and other supportive agents are essential. Sedation and adequate respiratory support, including extended ventilation, should be emphasized.
In conclusion, owing to the wide variety of pathologies encountered in double outlet right ventricle, the operative outcome is dependent on several factors including patient age, type of lesion, associated malformations, pulmonary hypertension, and surgical technique. Most patients can achieve a satisfactory surgical outcome with an individualized surgical strategy and technique and proper postoperative management.
References [ + ]