What is glycerol
Glycerol is also called glycerine or glycerin, is a trihydroxy sugar alcohol that is an intermediate in carbohydrate and lipid metabolism. Glycerol occurs naturally in several types of lipid and is an endogenous metabolite in mammals. Glycerol is rapidly and near completely absorbed from the gastrointestinal tract, it is then distributed into the total body water space and primarily metabolized in the liver. After phosphorylation and oxidation, glycerol is used as an energy substrate via glycolysis or participates in gluconeogenesis and lipogenesis. When the body uses stored fat as a source of energy, glycerol and fatty acids are released into the bloodstream. Circulating glycerol does not glycate proteins as do glucose or fructose, and does not lead to the formation of advanced glycation endproducts. Glycerol is extensively oxidized and exhaled as carbon dioxide, with only minor amounts excreted via urine or feces. Glycerol is a colorless, odorless, viscous liquid that is sweet-tasting and non-toxic. Glycerol (glycerin) is used as a solvent, emollient, humectant and vehicle in various pharmaceutical preparations, or sweetening agent in the food industry. As a sugar substitute, it has approximately 27 kilocalories per teaspoon (sugar has 20) and is 60% as sweet as sucrose. It does not feed the bacteria that form plaques and cause dental cavities. As a food additive, glycerol is labeled as E number E422. It is added to icing (frosting) to prevent it from setting too hard. Glycerol (glycerin) elevates the blood plasma osmolality thereby extracting water from tissues into interstitial fluid and plasma. Glycerol concentrations in blood higher than 92 mg/L would result in renal elimination of glycerol in rats and humans by preventing water reabsorption in the proximal tubule in the kidney leading to an increase in water and sodium excretion and a reduction in blood volume. Administered rectally, glycerol exerts a hyperosmotic laxative effect by attracting water into the rectum, thereby relieving constipation. In addition, glycerol (glycerin) is used as a solvent, humectant and vehicle in various pharmaceutical preparations.
In a subchronic toxicity study (in drinking water) in rats, the effects reported were observed with doses in the range of the oral median lethal dose (Lethal Dose 50% or LD50) values for glycerol. Lethal Dose 50% (LD50) or median lethal dose is the amount of the substance required (usually per body weight) to kill 50% of the test population The local irritating effects of glycerol in the gastrointestinal tract reported in some gavage studies in rat (100% glycerol at 2,800 mg/kg body weight (bw) per day, the lowest dose tested), and dogs (100% glycerol at 5,600 mg/kg body weight per day) was likely due to the hygroscopic and osmotic effects of large bolus doses of glycerol administered by gavage.
Glycerol did not show any genotoxic activity in a variety of in vitro assays. A lack of valid in vivo genotoxicity data was not of concern since clear negative findings were observed in in vitro assays. On this basis, the European Food Safety Authority (EFSA) Panel considered that glycerol as a food additive did not raise concern with respect to genotoxicity 1).
From the available chronic toxicity and carcinogenicity studies, glycerol was not carcinogenic in mice and rats and did not show evidence of adverse effects in a 2‐year chronic toxicity study. The European Food Safety Authority (EFSA) Panel noted that no adverse effects were reported in rats receiving doses up to 10,000 mg/kg body weight per day for 1 year, the highest dose tested. The European Food Safety Authority (EFSA) Panel also noted that there was no increase in tumor incidences in rats receiving doses up to 5,000 mg/kg body weight per day for 2 years, the highest dose tested 2).
The reports of the two and multigeneration reproductive toxicity studies have limitations but no adverse effects were reported. Prenatal developmental toxicity studies were also limited but showed no dose‐related developmental and maternal effects up to the highest dose tested (1,280, 1,600 or 1,180 mg glycerol/kg body weight per day for mice, rats and rabbits, respectively) 3).
The European Food Safety Authority (EFSA) Panel considered that none of the animal studies available identified an adverse effect for glycerol 4).
The European Food Safety Authority (EFSA) Panel considered that the therapeutic oral use of glycerol at 1,000–1,500 mg/kg body weight given as bolus in patients with glaucoma triggered an increase in plasma osmolality and dehydration and resulted in some patients with side effects such as headache, nausea and vomiting. Given the dose‐ and time period‐range reported in a study, the European Food Safety Authority (EFSA) Panel calculated that the minimum dose of glycerol required to induce a therapeutic reduction in intracranial pressures was within the range of 125–333 mg/kg body weight per hour. The European Food Safety Authority (EFSA) Panel considered that a conservative estimate of the lowest oral bolus dose of glycerol required for therapeutic effect was 125 mg/kg body weight per hour. The European Food Safety Authority (EFSA) Panel considered this dose would also be responsible for the side effects (nausea, headache and/or vomiting) observed in some patients.
The European Food Safety Authority (EFSA) Panel considered that the exposure estimates in all exposure scenarios resulted in overestimates of the exposure to glycerol from its use as a food additive according. Furthermore, the European Food Safety Authority (EFSA) Panel noted that the additional exposure to glycerol via natural sources, including wine and honey (the sources for which analytical data were available) did not significantly change the exposure to glycerol (E 422). The highest 95th percentile of exposure of glycerol and natural sources was estimated at 460 mg/kg body weight per day in children in the refined non‐brand‐loyal exposure scenario.
The European Food Safety Authority (EFSA) Panel considered that the most relevant situation where an acute bolus exposure to glycerol used as a food additive can be similar to the one occurring during therapeutic use is consumption of a beverage. Therefore, the European Food Safety Authority (EFSA) Panel calculated the volume of flavored drinks required to be consumed in order to exceed the acute bolus exposure (125 mg/kg body weight per hour) calculated by the European Food Safety Authority (EFSA) Panel to be the minimum dose required to have a therapeutic effect. When considering the available data, the European Food Safety Authority (EFSA) Panel noted that infants and toddlers can be exposed to more than 125 mg glycerol/kg body weight per hour by drinking less than the volume of one can (330 mL) of a flavored drink.
According to the conceptual framework for the risk assessment of certain food additives re‐evaluated under European Food Safety Authority (EFSA) Panel on Food Additives and Nutrient Sources added to Food 2014 and given that:
- the safety assessment carried out by the Panel is limited to the use and use levels received from industry and member states in 28 food categories out of 68 food categories in which glycerol is authorized;
- the highest 95th percentile of exposure of glycerol and natural sources was estimated at 460 mg/kg body weight per day in children in the refined non‐brand‐loyal exposure scenario;
- glycerol as a food additive is identical to a compound which is a normal constituent in the body (an endogenous compound) and is a regular component of the diet;
- sufficient toxicity data were available;
- the toxicological studies in animals did not provide any indication for adverse effects, including at the highest dose tested in a chronic toxicity study (10,000 mg/kg body weight per day);
The European Food Safety Authority (EFSA) Panel concluded that there is no need for a numerical Acceptable Daily Intake (ADI) for glycerol.
The European Food Safety Authority (EFSA) Panel concluded that there is no safety concern regarding the use of glycerol as a food additive for the general population at the refined exposure assessment for the reported uses of glycerol as a food additive 5).
However, the European Food Safety Authority (EFSA) Panel identified that there remain uncertainties over the lack of identification and quantification of residuals, especially those that are genotoxic and carcinogenic. The European Food Safety Authority (EFSA) Panel noted that these residuals are mostly present when chemical synthesis is used to produce glycerol. The European Food Safety Authority (EFSA) Panel concluded that the manufacturing process for glycerol should not allow the production of a food additive, which contains these residuals at a level, which would result in a margin of exposure (MOE) below 10,000.
The European Food Safety Authority (EFSA) Panel concluded that if 3‐monochloropropane‐1,2‐diol (3‐MCPD) is present at its maximum authorised level of 0.1 mg 3‐MCPD/kg glycerol, the maximum exposure to 3‐MCPD was below the tolerable daily intake (TDI) of 0.8 μg/kg body weight per day, and therefore exposure via glycerol alone was of no concern.
The European Food Safety Authority (EFSA) Panel could not calculate exposures to other genotoxic impurities or contaminants that may be present in glycerol as a result of the manufacturing process, e.g. glycidol, due to the lack of data on their concentrations in the food additive.
The European Food Safety Authority (EFSA) Panel concluded that infants and toddlers could be exposed to more than 125 mg/kg body weight per hour by drinking less than the volume of one can (330 mL) of a flavored drink. The European Food Safety Authority (EFSA) Panel further concluded that the acute bolus exposure to glycerol by its use as a food additive should stay below doses at which pharmacological or side effects could occur.
The European Food Safety Authority (EFSA) Panel recommended that:
- given that during the manufacturing processes of glycerol, genotoxic impurities – e.g. glycidol, epichlorohydrin – could be formed, limits for such impurities should be included in the specifications for glycerol (E 422);
- given that during the manufacturing processes of glycerol, other potential impurities of toxicological concern (e.g. dichlorohydrin) could be formed, limits for such impurities should be included in the specifications for glycerol (E 422);
- more data should be generated to decrease uncertainty arising from the presence in the food additive (E 422) of compounds of toxicological concern (e.g. acrolein, 3‐MCPD or 3‐MCPD ester), which can be produced under some food processing conditions (e.g. use of glycerol in parallel with lactic acid bacteria; use of glycerol in food containing significant amounts of sodium chloride (more than 5%) and treated at temperatures above 160°C, etc.);
- a numerical limit for acrolein should be included in the specifications for glycerol ;
- the maximum limits for the impurities of toxic elements (arsenic, lead, mercury and cadmium) in the specification for glycerol should be revised in order to ensure that glycerol as a food additive will not be a significant source of exposure to those toxic elements in food;
- more information on uses and use levels and analytical data should be made available to the European Food Safety Authority (EFSA) Panel in order to perform an adequate exposure assessment, in particular in the case of estimate acute exposure, more data on flavored drinks is needed.
Glycerol chemical formula
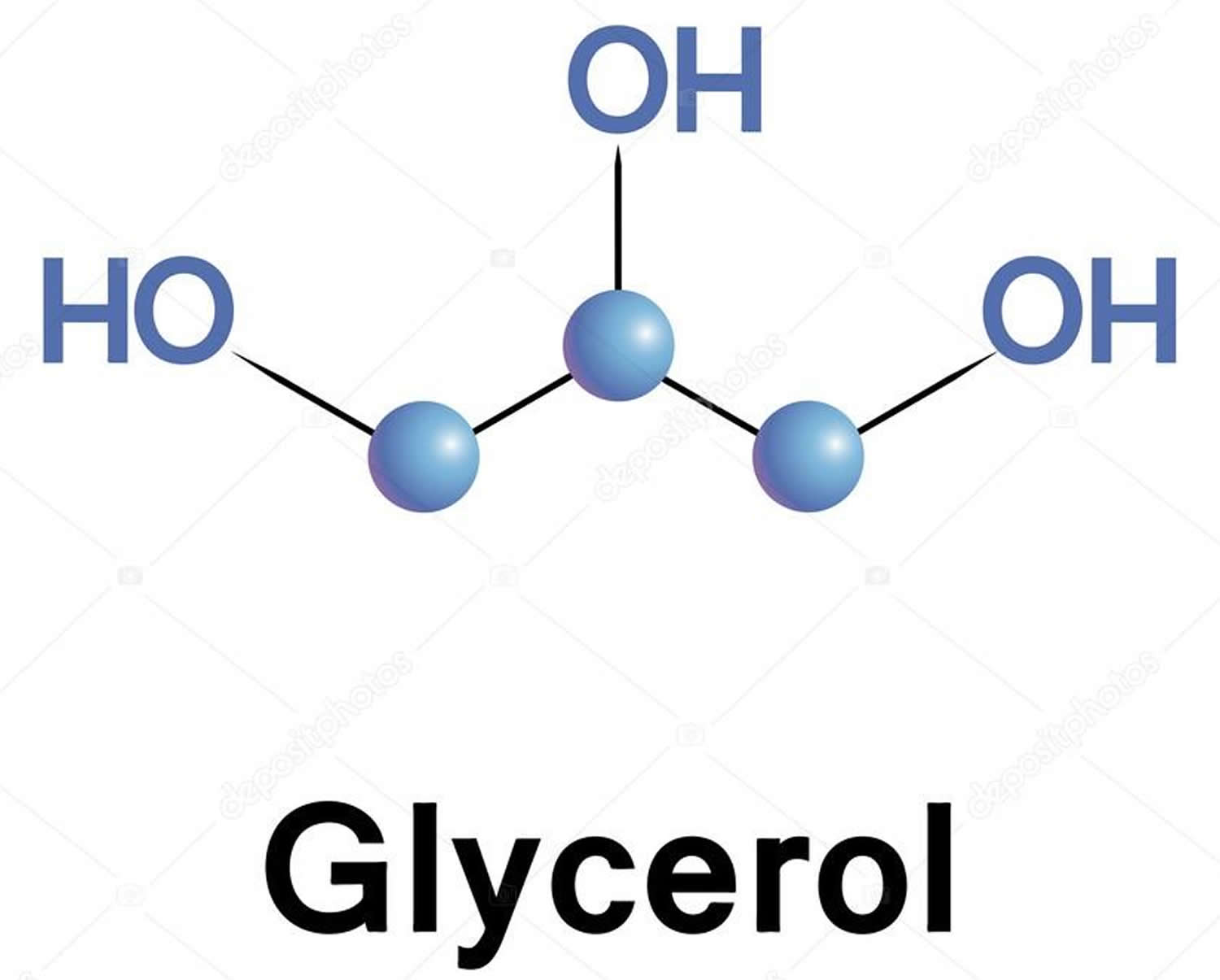
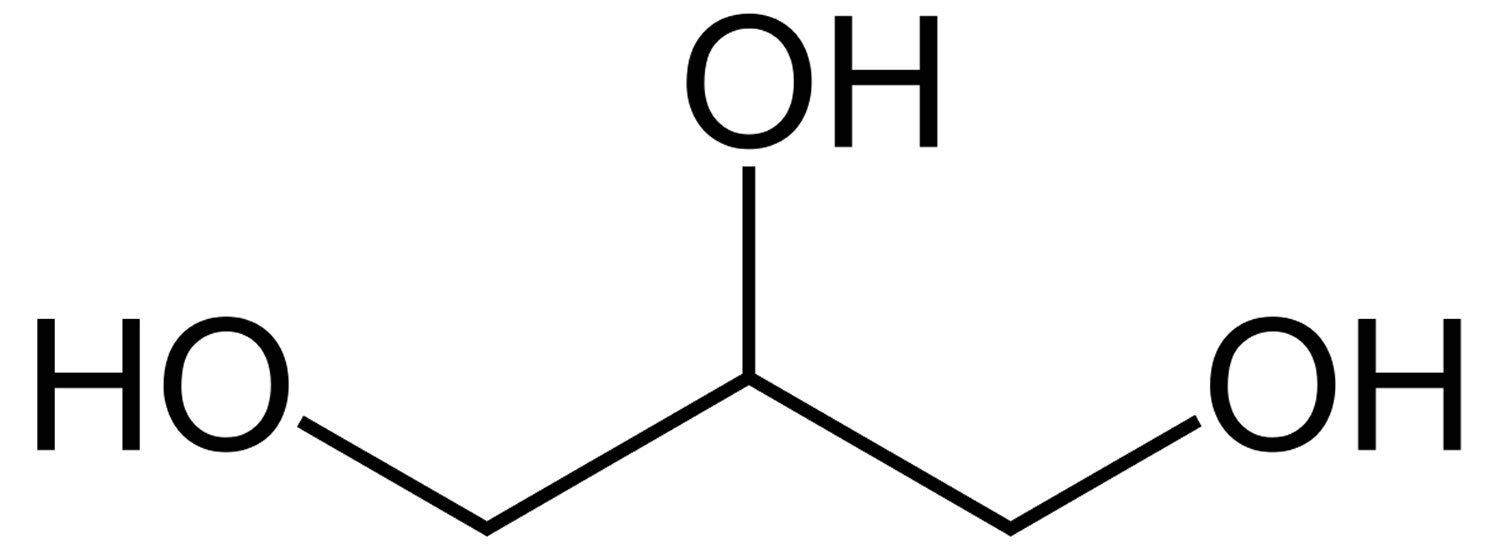
Glycerol is described as a clear, colourless, hygroscopic syrupy liquid, with not more than a slight characteristic odor, which is neither harsh nor disagreeable. Glycerol is miscible in water and ethanol and immiscible in ether 6). The glycerol backbone is found in all lipids known as triglycerides. Glycerol has three hydroxyl groups that are responsible for its solubility in water and its hygroscopic nature (Figure 1).
Glycerol is generally obtained from plant and animal sources where it occurs as triglycerides. Triglycerides are esters of glycerol with long-chain carboxylic acids. The hydrolysis, saponification, or transesterification of these triglycerides produces glycerol as well as the fatty acid derivative.
Typical plant sources include soybeans or palm. Animal-derived tallow is another source.
Glycerol from triglycerides is produced on a large scale, but the crude product is of variable quality, with a low selling price of as low as 2-5 U.S. cents per kilogram in 2011 7). Glycerol can be purified, but the process is expensive. Some glycerol is burned for energy, but its heat value is low.
Crude glycerol from the hydrolysis of triglycerides can be purified by treatment with activated carbon to remove organic impurities, alkali to remove unreacted glycerol esters, and ion exchange to remove salts. High purity glycerol (> 99.5%) is obtained by multi-step distillation; vacuum is helpful due to the high boiling point of glycerol (290 °C).
- Glycerol molecular weight is 92.094 g/mol
- Glycerol molar mass is 92.094 g/mol
- Glycerol melting point is 64.4 °F (18 °C)
- Glycerol boiling point is 554° F (290 °C) at 760 mm Hg
- Density of glycerol is 1.2613 g/cu cm at 68 °F (20 °C)
Figure 1. Glycerol molecule
Gycerol uses
Glycerol as an ingredient in food
The use of glycerol in beverages for athletes to enhance and maintain hydration status and to improve endurance exercise performance is described 8). Glycerol when used as a component of a hyperhydration strategy is prohibited by the World Anti‐Doping Agency (WADA) 9).
Pharmaceutical use
Glycerol is used in pharmaceutical products as an active ingredient as well as an excipient, e.g. solvent, plasticiser or lubricant 10). When glycerol is used as an excipient in medicinal products for oral administration and the glycerol intake is equal or above 10 g per single dose of the medicinal product, the information ‘May cause headache, stomach upset and diarrhea’ is required for the package leaflet 11). As an active ingredient, glycerol is used as an osmotic dehydrating agent. It is given orally, e.g. for the reduction in intra‐ocular pressure before and after ophthalmic surgery and as an adjunct in the management of acute glaucoma or to reduce intracranial pressure 12).
Glycerol has been used therapeutically by intravenously or oral administration to mobilize edema and to induce osmotic diuresis. Clinical experience and data from clinical studies are found in the literature indicating that doses up to 60,000 mg intravenously, infused over 1–4 hour, for a treatment duration of 1 week and in some studies, several weeks 13), and bolus oral doses (initial dose 1,500 mg/kg body weight, followed by 500–700 mg/kg body weight every 3 hour) 14) or 500–1,000 mg/kg body weight 15).
The European Food Safety Authority (EFSA) Panel considered that the therapeutic administration of glycerol by oral administration to patients reported to be suffering disease that could impact significantly on their physiological functionality was not relevant in the safety assessment of glycerol as a food additive. However, glycerol has been prescribed for reduction in intraocular pressure in patients with glaucoma and may be prescribed prior to intraocular surgery 16). For this indication, glycerol was reported to be administered orally as a bolus dose of 1,000–1,500 mg/kg body weight (as a 50% solution) with a daily total oral dose not higher than 120,000 mg (about 1,700 mg/kg body weight) 17). The European Food Safety Authority (EFSA) Panel considered that the therapeutic administration of glycerol by oral administration to patients reported to be suffering ocular disease – such as glaucoma and administration of glycerol to control participants in clinical studies – were relevant for the safety assessment of glycerol as a food additive.
The European Food Safety Authority (EFSA) Panel noted that for oral therapies to patients with ocular disease or healthy controls, glycerol was administered as a bolus at 1,350 mg/kg body weight 18); 1,500 mg/kg body weight 19); 1,500 mg/kg body weight 20); 1,000 mg/kg body weight 21); 1,000–1,270 mg/kg bw (McCurdy et al., 1966); 1,350 mg/kg body weight 22); 1,200 mg/kg body weight 23) and 1,260 mg/kg body weight 24). The only side effects observed in these oral therapies to patients with ocular disease or healthy controls were either none or one or more of nausea, headache and/or vomiting.
Guidelines for glycerol use in hyperhydration and rehydration associated with exercise report 28 studies in which oral doses between 500 mg/kg body weight and 1,500 mg/kg body weight were given in the total of 238 subjects 25). Three studies reported side effects after rapidly administering the glycerol as a concentrated bolus followed by fluid ingestion. Four subjects in two of the trials were nauseous after glycerol ingestion. In another study, two subjects developed diarrhea in the 24 hours after the trial. In a further three studies, a low incidence of gastrointestinal distress (bloatedness) or light‐headedness were reported.
Using the studies of Wald and McLaurin 26), the European Food Safety Authority (EFSA) Panel noted that patients were administered glycerol orally as bolus doses of 500–1,000 mg/kg body weight every 3–4 hour, dependent on the patients’ intracranial pressures (ICPs). Specific individual dosages ranged from 4,000 mg to 70,000 mg (average 54,000 mg) and it was administered via a nasogastric tube as a 50% solution (by mixing a 100% glycerol solution with an equal volume of either 5% dextrose in water or 5% dextrose in 0.4% normal saline, depending upon the systemic electrolyte status). If no change in intracranial pressure was achieved or a significant volume of solution was aspirated from the stomach, intravenous mannitol was administered and another trial of the drug was initiated 4–24 hour later. For the six patients in which data were reported in this study, maximum intracranial pressure reduction and maximum serum glycerol concentration occurred around 60–90 min after oral bolus ingestion. In most cases, the serum glycerol concentration returned to pre‐treatment levels around 3 hour after oral administration.
Given the dose‐ and time period‐range reported in this study, the European Food Safety Authority (EFSA) Panel calculated that the dose of glycerol required to induce a therapeutic reduction in intracranial pressure (ICP) was within the range of 125–333 mg/kg body weight per hour.
The European Food Safety Authority (EFSA) Panel therefore considered that a conservative estimate of the lowest oral bolus dose of glycerol required for therapeutic effect was 125 mg/kg body weight per hour. The European Food Safety Authority (EFSA) Panel considered this dose would also be responsible for the side effects (nausea, headache and/or vomiting) observed in some patients.
Glycerol therapy in diabetics
Hyperosmolar non‐ketotic coma occurred in diabetic patients after repeated use of oral and intravenous glycerol [e.g. Oakley and Ellis 27); Sear 28)]. According to Sear 29), the non‐ketotic hyperosmolar hyperglycaemic state usually occurs in cases of maturity onset diabetes or pre‐diabetes and not in non‐diabetic subjects.
Food industry
In food and beverages, glycerol serves as a humectant, solvent, and sweetener, and may help preserve foods. It is also used as filler in commercially prepared low-fat foods (e.g., cookies), and as a thickening agent in liqueurs. Glycerol and water are used to preserve certain types of plant leaves 30).
As used in foods, glycerol is categorized by the Academy of Nutrition and Dietetics as a carbohydrate. The U.S. Food and Drug Administration (FDA) carbohydrate designation includes all caloric macronutrients excluding protein and fat. Glycerol has a caloric density similar to table sugar, but a lower glycemic index and different metabolic pathway within the body.
It is also recommended as an additive when using polyol sweeteners such as erythritol and xylitol which have a cooling effect, due to its heating effect in the mouth, if the cooling effect is not wanted.
Personal care applications
Glycerol is used in medical, pharmaceutical and personal care preparations, mainly as a means of improving smoothness, providing lubrication, and as a humectant. It is found in allergen immunotherapies, cough syrups, elixirs and expectorants, toothpaste, mouthwashes, skin care products, shaving cream, hair care products, soaps, and water-based personal lubricants. In solid dosage forms like tablets, glycerol is used as a tablet holding agent. For human consumption, glycerol is classified by the U.S. FDA among the sugar alcohols as a caloric macronutrient.
Glycerol is a component of glycerin soap. Essential oils are added for fragrance. This kind of soap is used by people with sensitive, easily irritated skin because it prevents skin dryness with its moisturizing properties. It draws moisture up through skin layers and slows or prevents excessive drying and evaporation.
Glycerol can be used as a laxative when introduced into the rectum in suppository or small-volume (2–10 ml) enema form; it irritates the anal mucosa and induces a hyperosmotic effect 31).
Taken orally (often mixed with fruit juice to reduce its sweet taste), glycerol can cause a rapid, temporary decrease in the internal pressure of the eye. This can be useful for the initial emergency treatment of severely elevated eye pressure in glaucoma 32).
Antifreeze
Like ethylene glycol and propylene glycol, glycerol is a non-ionic kosmotrope that forms strong hydrogen bonds with water molecules, competing with water-water hydrogen bonds. This interaction disrupts the formation of ice . The minimum freezing point temperature is about −36 °F (−38 °C) corresponding to 70% glycerol in water.
Glycerol was historically used as an anti-freeze for automotive applications before being replaced by ethylene glycol, which has a lower freezing point 33). While the minimum freezing point of a glycerol-water mixture is higher than an ethylene glycol-water mixture, glycerol is not toxic and is being re-examined for use in automotive applications.
In the laboratory, glycerol is a common component of solvents for enzymatic reagents stored at temperatures below 0 °C due to the depression of the freezing temperature. It is also used as a cryoprotectant where the glycerol is dissolved in water to reduce damage by ice crystals to laboratory organisms that are stored in frozen solutions, such as bacteria, nematodes, and mammalian embryos.
Vibration dampening
Glycerol is used as fill for pressure gauges to dampen vibration 34). External vibrations, from compressors, engines, pumps, etc., produce harmonic vibrations within Bourdon gauges that can cause the needle to move excessively, giving inaccurate readings. The excessive swinging of the needle can also damage internal gears or other components, causing premature wear. Glycerol, when poured into a gauge to replace the air space, reduces the harmonic vibrations that are transmitted to the needle, increasing the lifetime and reliability of the gauge.
Film industry
Glycerol is used by the film industry when filming scenes involving water to stop areas from drying out too quickly.
Glycerine is used—combined with water (around in a 1:99 proportion)—to create a smooth smoky environment. The solution is vaporized and pushed into the room with a ventilator.
References [ + ]