Contents
Inner ear anatomy
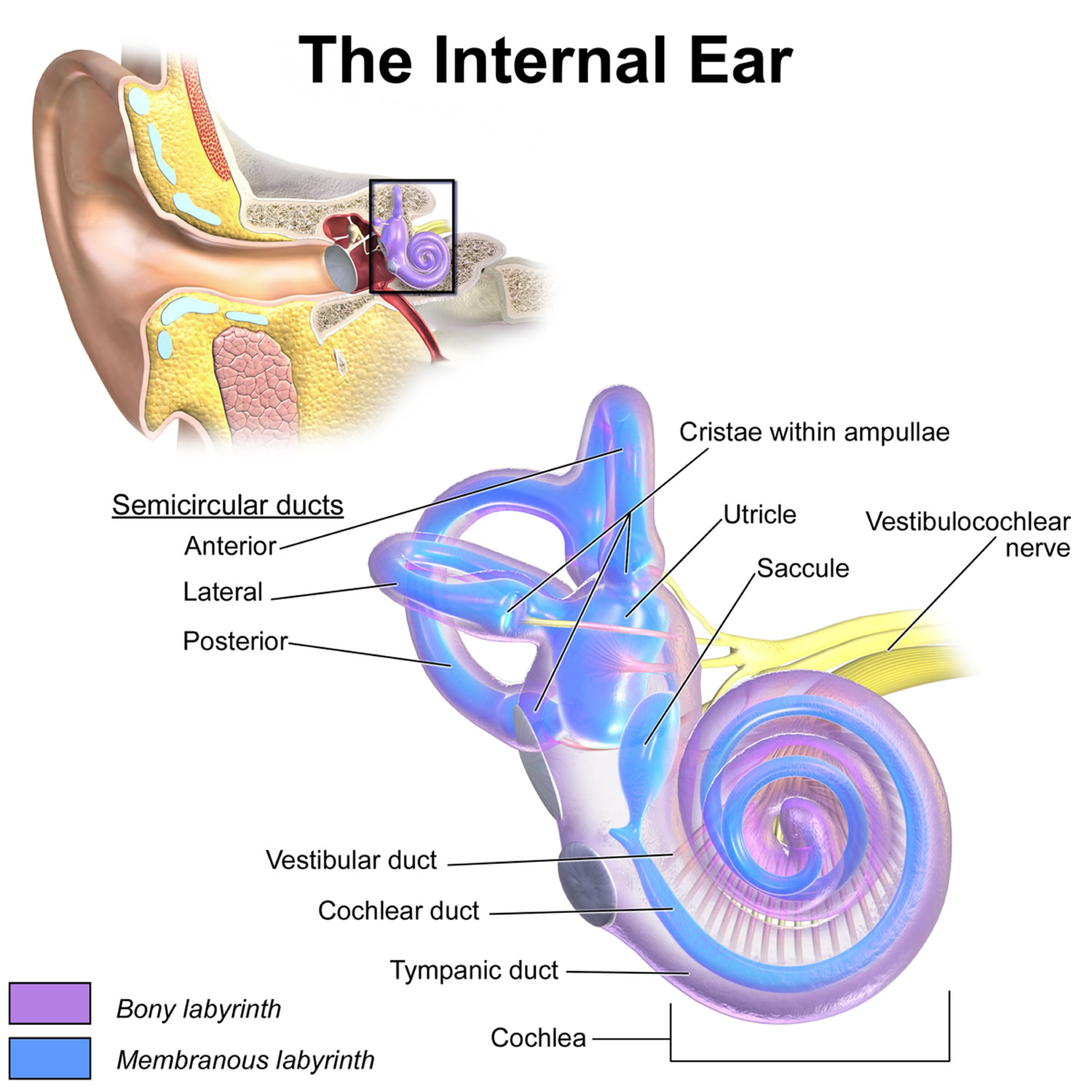
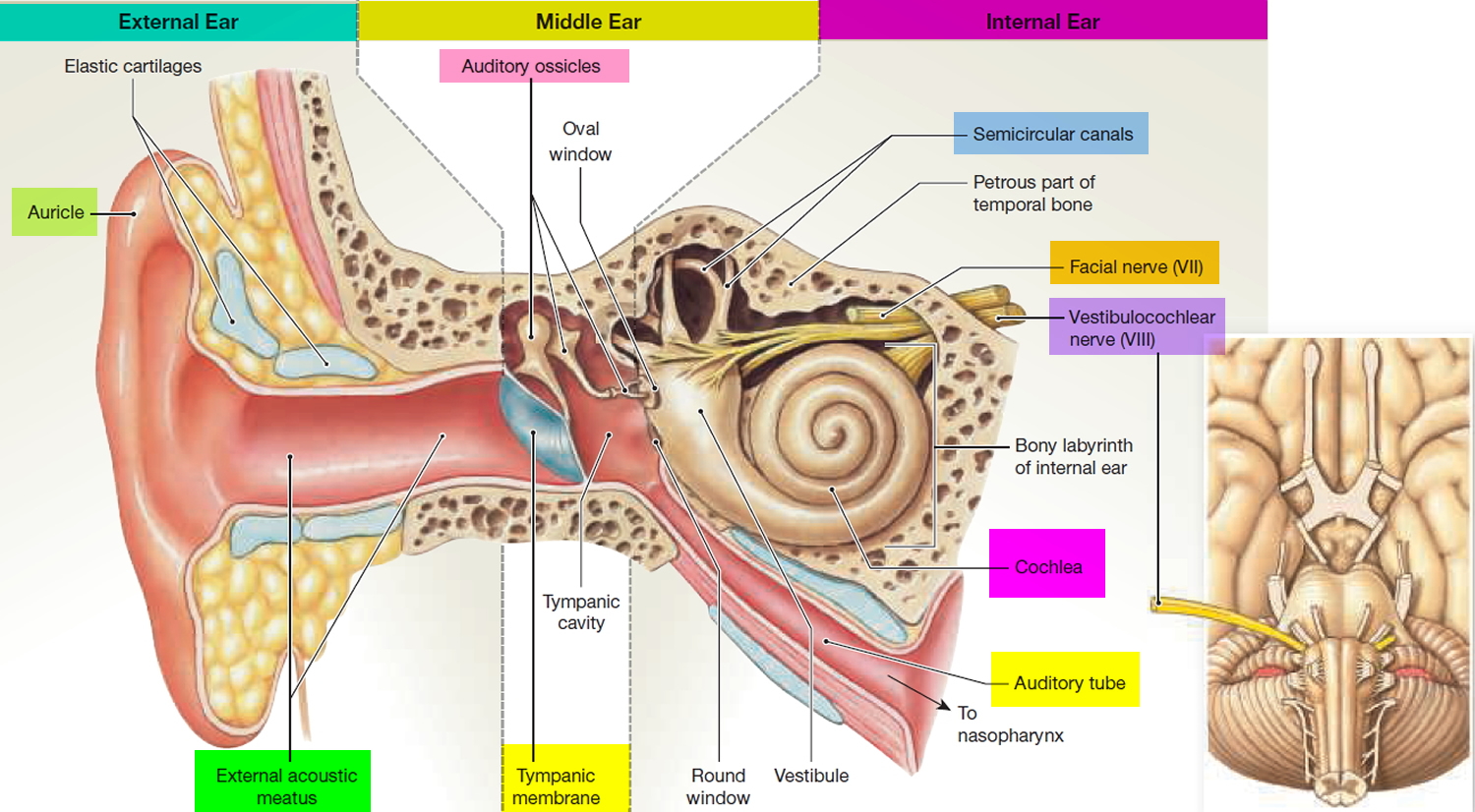
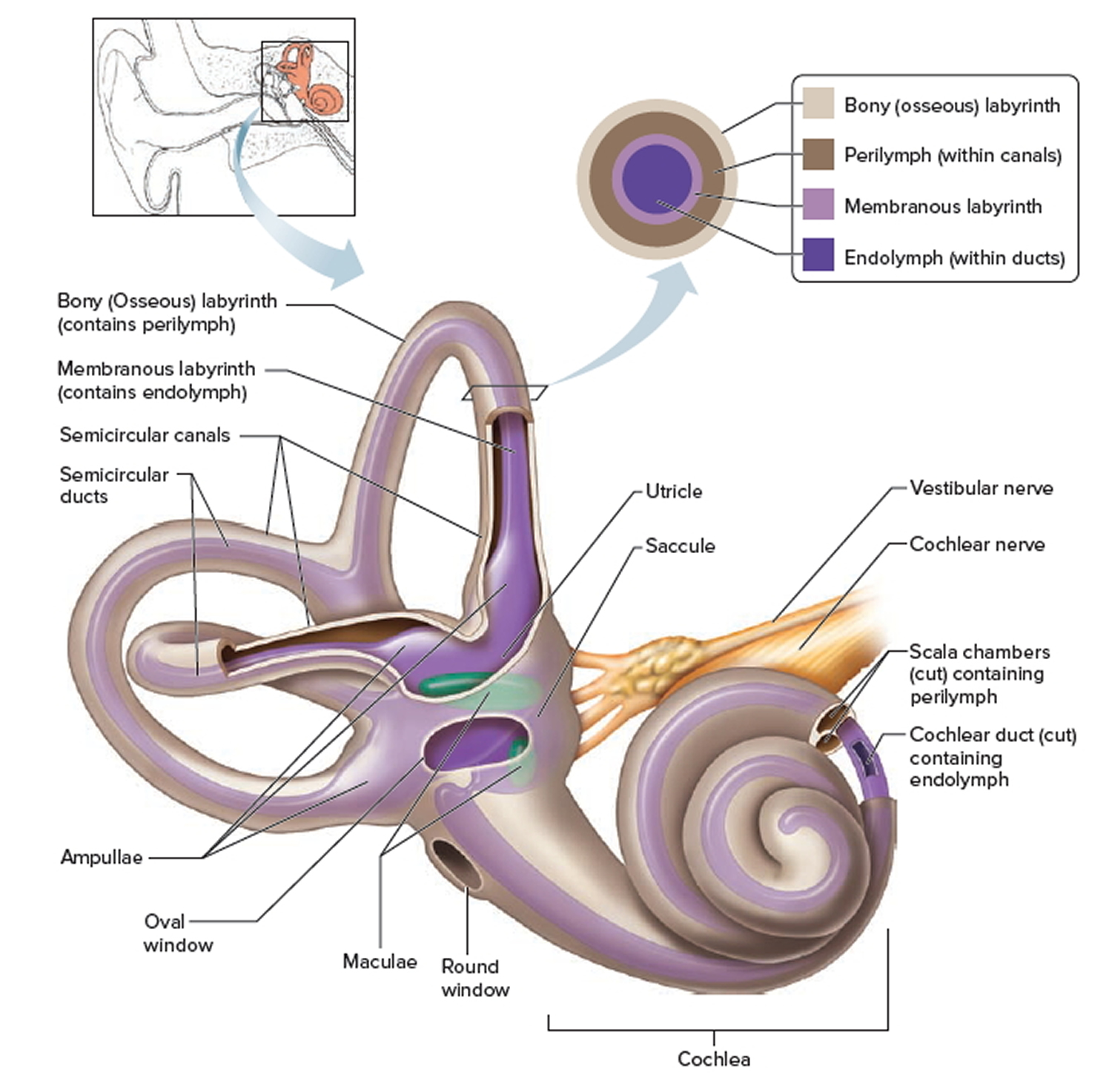
You rely on the inner ear, an intricate sensory organ, to hear and to maintain balance. The inner ear is a complex system of communicating chambers and tubes called a labyrinth. Each ear has two parts to the labyrinth—the bony (osseus) labyrinth and the membranous labyrinth (Figure 1). The bony labyrinth is a cavity within the temporal bone. The membranous labyrinth is a tube of similar shape that lies within the bony labyrinth. Between the bony and membranous labyrinths is a fluid called perilymph, which is secreted by cells in the wall of the bony labyrinth. The membranous labyrinth contains another fluid, called endolymph.
The parts of the labyrinths include three membranous semicircular ducts within three bony semicircular canals, and a cochlea. The semicircular canals and associated structures provide a sense of equilibrium (balance). The cochlea functions in hearing.
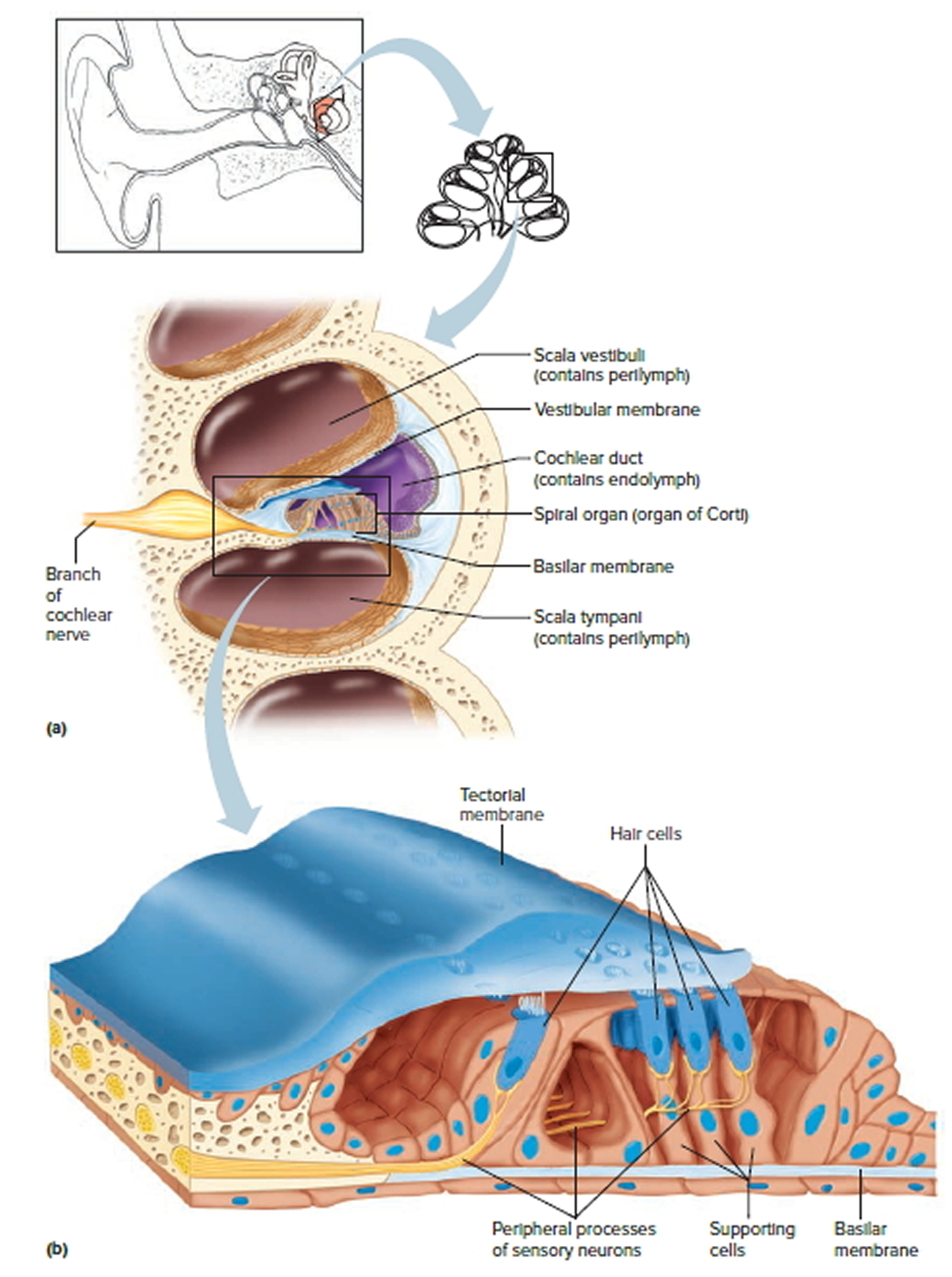
The cochlea has a bony core and a thin, bony shelf that extends out from the core and coils around it. The shelf divides the bony labyrinth of the cochlea into upper and lower compartments. The upper compartment, called the scala vestibuli, leads from the oval window to the tip of the cochlea. The lower compartment, the scala tympani, extends from the tip of the cochlea to a membrane-covered opening in the wall of the middle ear called the round window.
The part of the membranous labyrinth within the cochlea is called the cochlear duct. It lies between the two bony compartments and ends as a closed sac near the tip of the cochlea. The cochlear duct is separated from the scala vestibuli by a vestibular membrane (Reissner’s membrane) and from the scala tympani by a basilar membrane. The basilar membrane has many thousands of elastic fibers, allowing it to move in response to sound vibrations. Sound vibrations entering the perilymph at the oval window travel along the scala vestibuli and pass through the vestibular membrane and into the endolymph of the cochlear duct, where they move the basilar membrane.
After passing through the basilar membrane, the vibrations enter the perilymph of the scala tympani. Their forces are dissipated into the air in the tympanic cavity by movement of the membrane covering the round window.
The spiral organ (organ of Corti) contains the hearing receptors. It is located on the upper surface of the basilar membrane and stretches from the apex to the base of the cochlea. The receptor cells, called hair cells, are organized in rows and have many hairlike processes that project into the endolymph of the cochlear duct. Above these hair cells is a tectorial membrane attached to the bony shelf of the cochlea, passing over the receptor cells and contacting the tips of their hairs.
As sound vibrations move the basilar membrane, the hairs move back and forth against the tectorial membrane and the resulting mechanical deformation of the hairs stimulates the hair cells (Figure 4. The Cochlea). Hair cells at different locations along the length of the cochlear duct respond to different frequencies (pitch) of sound vibrations. This enables us to hear sounds of different pitch simultaneously.
Hair cells are epithelial but function somewhat like neurons. For example, when a hair cell is at rest, its membrane is polarized. When it is stimulated, selective ion channels open, depolarizing the membrane and making it more permeable to calcium ions. The hair cell has no axon or dendrites, but it has neurotransmitter-containing vesicles near its base. As calcium ions diffuse into the cell, some of these vesicles fuse with the cell membrane and release a neurotransmitter by exocytosis. The neurotransmitter stimulates the dendrites of nearby sensory neurons. In response these neurons send action potentials along the cochlear branch of the vestibulocochlear nerve to the auditory cortex of the temporal lobe of the brain.
The ear of a young person with normal hearing can detect sound waves with frequencies ranging from 20 to more than 20,000 vibrations per second. The range of greatest sensitivity is 2,000 to 3,000 vibrations per second. More-intense stimulation of the hair cells causes more action potentials per second to reach the auditory cortex, and we sense a louder sound.
Units called decibels (dB) measure sound intensity on a logarithmic scale. The decibel scale begins at 0 dB, which is the intensity of the sound that is least perceptible by a normal human ear. A sound of 10 dB is 10 times as intense as the least perceptible sound; a sound of 20 dB is 100 times as intense; and a sound of 30 dB is 1,000 times as intense. A whisper has an intensity of about 40 dB, normal conversation measures 60 to 70 dB, and heavy traffic produces about 80 dB. A sound of 120 dB, common at a rock concert, produces discomfort, and a sound of 140 dB, such as that emitted by a jet plane at takeoff, causes pain.
Frequent or prolonged exposure to sounds with intensities above 85 dB can damage hearing receptors and cause permanent hearing loss.
Auditory Pathways
The nerve fibers associated with hearing enter the auditory pathways, which pass into the auditory cortices of the temporal lobes of the cerebrum. Here they are interpreted. On the way, some of these fibers cross over, so that impulses arising from each ear are interpreted on both sides of the brain. Consequently, damage to a temporal lobe on one side of the brain does not necessarily cause complete hearing loss in the ear on that side.
Several factors cause partial or complete hearing loss. Interference with the transmission of vibrations to the inner ear is called conductive hearing loss. Conductive hearing loss may be due to plugging of the external acoustic meatus or to changes in the eardrum or auditory ossicles. For example, the eardrum may harden as a result of disease and become less responsive to sound waves, or disease or injury may tear or perforate the eardrum.
Damage to the cochlea, auditory nerve, or auditory pathways can cause sensorineural hearing loss. Loud sounds, tumors in the central nervous system, brain damage from vascular accidents, or use of certain drugs can also cause sensorineural hearing loss.
Steps in the Generation of Sensory Impulses from the Ear
- Sound waves enter external acoustic meatus.
- Sound waves cause eardrum to reproduce vibrations coming from sound source.
- Auditory ossicles amplify and transfer vibrations to end of stapes.
- Movement of stapes at oval window transfers vibrations to perilymph in scala vestibuli.
- Vibrations pass through vestibular membrane and enter endolymph of cochlear duct, where they move the basilar membrane.
- Different frequencies of vibration of basilar membrane stimulate different sets of receptor cells.
- As a receptor cell depolarizes, its membrane becomes more permeable to calcium ions.
- Inward diffusion of calcium ions causes vesicles at base of the receptor cell to release neurotransmitter.
- Neurotransmitter stimulates dendrites of nearby sensory neurons.
- Sensory impulses are triggered on fibers of the cochlear branch of vestibulocochlear nerve.
- Auditory cortices of temporal lobes interpret sensory impulses.
Figure 1. Inner ear anatomy
Figure 2. Parts of the inner ear
Footnotes: The inner ear (internal ear) is the innermost part of your ear consisting of the cochlea (the snail-shaped portion on the right), and the semicircular canals on the left represent the vestibular system. The inner ear is mainly responsible for sound detection and balance. The inner ear consists of the bony labyrinth, a hollow cavity in the temporal bone of the skull with a system of passages comprising two main functional parts: (1) The cochlea, dedicated to hearing; converting sound pressure patterns from the outer ear into electrochemical impulses which are passed on to the brain via the auditory nerve. (2) The vestibular system, dedicated to balance. The inner ear is innervated by cranial nerve eight (CN VIII) also known as vestibulocochlear nerve that consists of the vestibular and cochlear nerves 1. The vestibular nerve is primarily responsible for maintaining body balance and eye movements, while the cochlear nerve is responsible for hearing 2. Damage to the vestibular nerve results in vertigo, a balance disorder, and nystagmus 2. The endolymph (blue and purple fluid) is the fluid contained in the membranous labyrinth of the inner ear and has a higher level of potassium than sodium.
Figure 3. Inner ear bones
Note: A closer look at the inner ear. Perilymph separates the bony (osseous) labyrinth of the inner ear from the membranous labyrinth, which contains endolymph. Note that areas of bony labyrinth have been removed to reveal underlying structures.
Figure 4. The Cochlea (cross section view)
Note: a) Cross section of the cochlea. (b) The spiral organ and the tectorial membrane.
Vestibulo-ocular reflex
The Vestibulo-Ocular Reflex is an essential function of your vestibular system that helps you maintain a stable perception of the world around you by maintaining your postural balance and spatial orientation in response to changes in your environment such as while you engage in normal movements throughout your day like walking 3, 4. The vestibulo-ocular reflex keeps you steady and balanced even though your eyes and head are continuously moving when you perform most actions. When you make a head movement, your eye muscles are triggered instantly to create an eye movement opposite to that of your head movement at the exact same speed to readjust your visual world, which, in turn, stabilizes your retinal image (line of sight in space) by keeping your eyes still in space and focused on an object, despite you head moving 5.
The Vestibulo-Ocular Reflex (VOR) involves three parts 5, 6, 7:
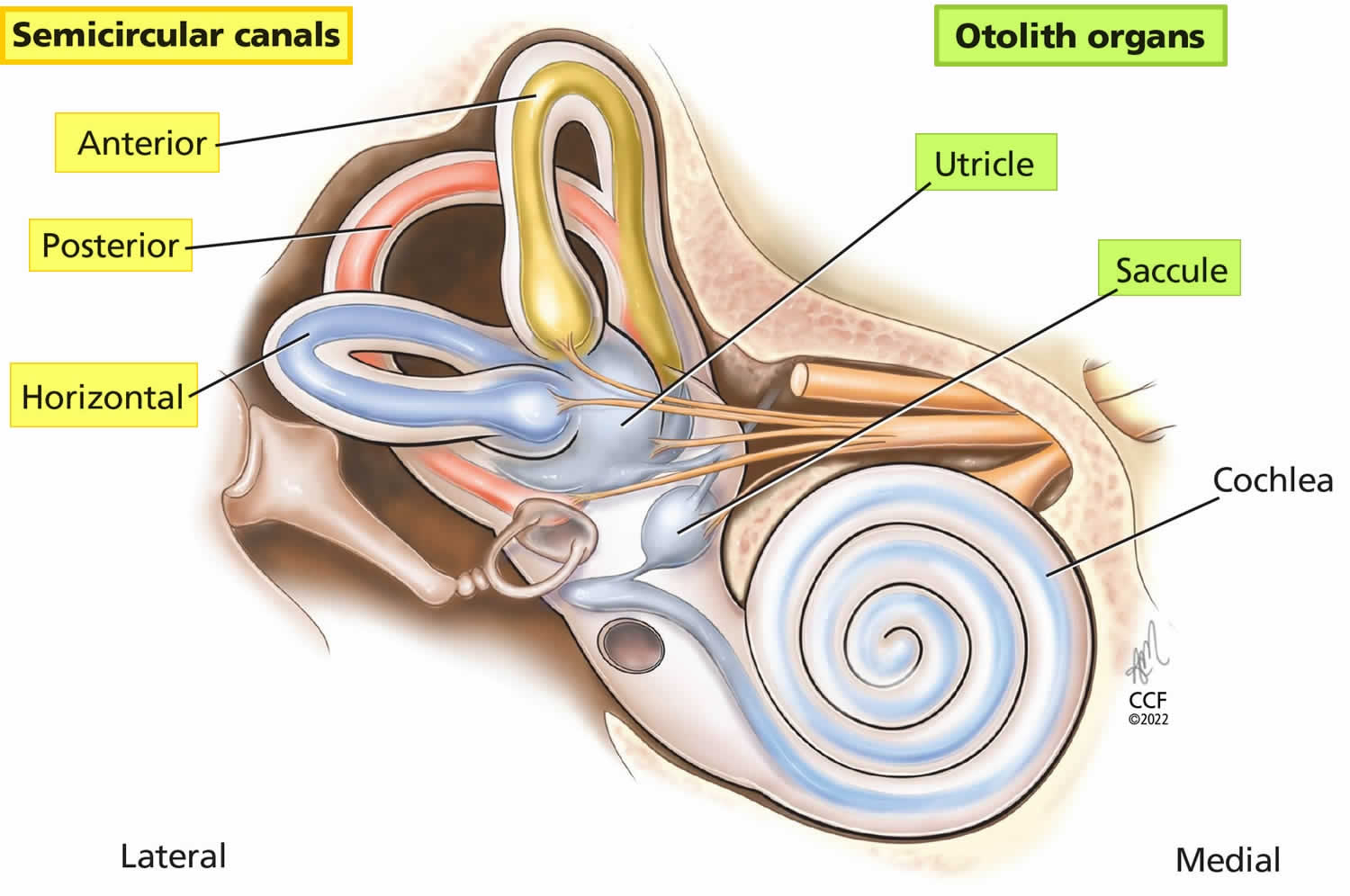
- Peripheral sensory apparatus which consists of a set of motion sensors: the semicircular canals (anterior, posterior, horizontal) and the otolith organs (saccule and utricle) (Figures 1 to 5).
- The semicircular canals are tubes coiled within your inner ear filled with liquid called the endolymph and lined with hair cells. Instead of sound waves, these tiny hairs react to sense rotational and angular movements of your head. The semicircular canals are mostly responsible for rotary motion or motion not in a straight line.
- Humans have a total of 6 semicircular canals arranged in three main planes (anterior, posterior, horizontal) on both sides; each plane has a pair of canals. The 3 in the right ear are functionally paired with the 3 in the left ear. The semicircular canals are referred to according to their orientation: lateral (horizontal), anterior (superior), and posterior. These structures are positioned at roughly right angles in relation to one another. The horizontal (lateral) semicircular canal, the shortest of the three, is set at an angle of about 30 degrees to the horizontal plane. The anterior (superior) and posterior loops are in diagonal vertical planes that intersect at a 90-degree angle. The 3 semicircular canals (anterior, posterior, horizontal) are positioned at 90-degree angles to one another and thus can sense rotation in all directions:
- The right horizontal with the left horizontal
- The right anterior with the left posterior
- The right posterior with the left anterior.
- A semicircular canal will get stimulated by head motion towards that semicircular canal 8. When a semicircular canal on one side is activated, there is inhibition of the other one during angular head movements. For example, when turning your head to look over your right shoulder, the right horizontal semicircular canal is excited and the left horizontal is inhibited. When there are head movements and rotation, the velocity of the movements dictates the difference in firing rate between the two semicircular canals 3. Hair cells are embedded in a fluid and gelatinous structure called the ampulla in each semicircular canal. When you rotate your head, the canal will move relative to the fluid inside of it, which will create a force against the ampulla, causing hair cells to bend. When your head is at rest, vestibular afferents have tonic discharge; this results in a balance between the semicircular canal pairs. Cases of nystagmus and ocular misalignment may be interpreted by understanding the organization and structure of the semicircular canals and otolith organs within the head 5. Information regarding linear head acceleration, angular velocity, and orientation of the head relative to gravity are all collected by these peripheral motion sensors. These sensors relay this information to the central nervous system (brain and spinal cord). Here, information from somatosensors (carrying other sensory information) is combined with motion sensory information to calculate head orientation.
- The semicircular canals (anterior, posterior, horizontal) sense angular head acceleration are responsible for detecting head rotation and the otolith organs (saccule and utricle) sense linear head acceleration and therefore detect the position of your head relative to gravity and head translation, providing your internal cues for orientation of position in space, movement, gaze stabilization, and postural control. Body movements cause the fluid in the semicircular canals to move and stimulate cilia on sensory hair cells. This triggers transmission of neural signals to the brain to initiate appropriate reflex responses for the eyes, head, and postural adjustments. These reflexive movements allow you to see things clearly when your head and body are in motion and keep you from falling.
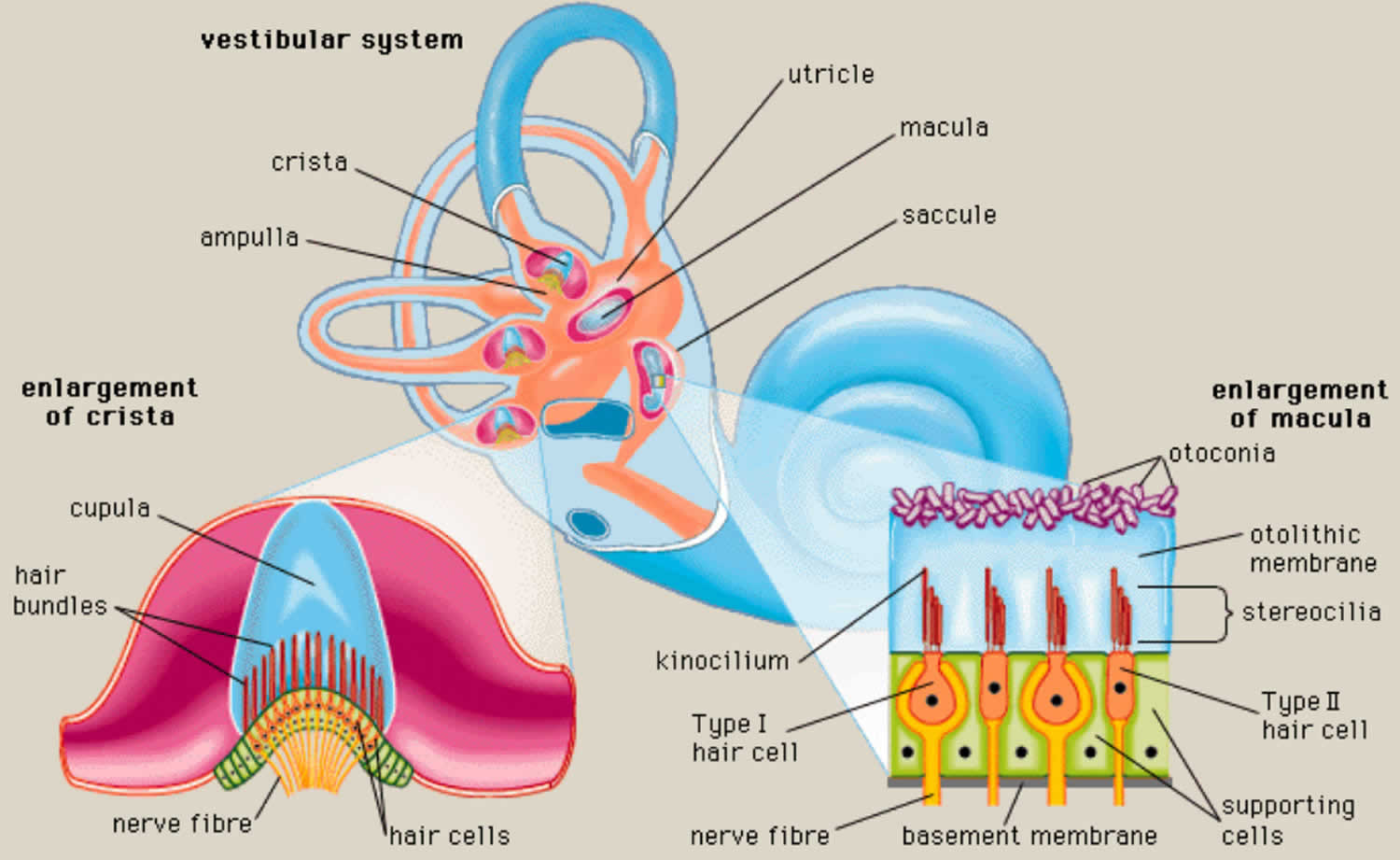
- Each semicircular canal has an ampulla, an expanded bulb-shaped structure that contains the hair cells (crista ampullaris), as well as a fan-like structure known as the cupula, which separates the semicircular canal from the vestibule (see Figures 1 and 3). The ampulla contains a fluid known as endolymph, which is also present in the cochlea, and the crista, a cone-shaped structure lined with hair cells and supporting cells. The hair cells form stereocilia, protrusions that extend into the crista. The longest of the stereocilia are the kinocilia, which point in a single direction and are sensitive to movement. The kinocilia extend from the crista into the cupula, a jellylike mass that surrounds the hair cells completely, separating them from the endolymph. The crista is flexible. When your head moves, endolymph pushes the cupula one way or the other, which in turn displaces the hairs and alters the electrical potential of the hair cells.
- The cupula has the same density as the endolymph and therefore semicircular canal does not respond to changes in the orientation of the head with respect to gravity. With angular head movements, however, the inertia of the endolymph results in a pressure on the cupula, which stimulates the hair cells through facilitation (increased firing rate) or inhibition (decreased firing rate). If the fluid is moving toward the ampulla, the horizontal canal is facilitated, while the semicircular canals in the vertical plane on the same side are inhibited. Hence, neural activities of the semicircular canal pairs are opposite, which results in a push–pull mechanism of facilitation on one side and inhibition on the other 9. The resting firing rate in the absence of motion would therefore be equal for the right and left labyrinths.
- Humans have a total of 6 semicircular canals arranged in three main planes (anterior, posterior, horizontal) on both sides; each plane has a pair of canals. The 3 in the right ear are functionally paired with the 3 in the left ear. The semicircular canals are referred to according to their orientation: lateral (horizontal), anterior (superior), and posterior. These structures are positioned at roughly right angles in relation to one another. The horizontal (lateral) semicircular canal, the shortest of the three, is set at an angle of about 30 degrees to the horizontal plane. The anterior (superior) and posterior loops are in diagonal vertical planes that intersect at a 90-degree angle. The 3 semicircular canals (anterior, posterior, horizontal) are positioned at 90-degree angles to one another and thus can sense rotation in all directions:
- The otolith organs (saccule and utricle) contain dense crystals called otoconia resting on top of the sensory organs, are gravity-sensitive and provide sensory neural input related to linear head acceleration.
- The utricle senses linear movements of the head in the horizontal plane.
- The saccule senses linear movements of the head in the vertical plane.
- Additionally, the utricle and saccule convey information about spinal and leg muscles for balance strategies (ankle, hip, and stepping) via the Vestibulo-Spinal Reflex (VSR), which is primarily to maintain upright posture during movement. The otoliths (otoconia) further convey information regarding the direction of gravity through head tilt, which is incorporated into locomotion. The function of the otolith organs is accomplished by otoconia (otoliths) – calcium carbonate crystals that are denser than the surrounding endolymph – remaining partially stable while the head moves around them (Figure 4). It is important to note that the utricle and saccule also have connections with eyes muscles as noted with the semicircular canal. However, the direct pathways of rotational compensatory eye movements are much greater than those of linear translation, resulting in minimal contributions from the otoliths to the Vestibulo-Ocular Reflex (VOR) as compared to the semicircular canal. With linear head movements (e.g., leaning to your side), the otoconia move, signaling reflexive responses similar to those of the semicircular canals to maintain eye, head, and body equilibrium.
- If otoconia (otoliths) become displaced from the utricle into the semicircular canals, then brief spells of vertigo are experienced related to position changes, a condition called benign paroxysmal positional vertigo (BPPV). Traumatic brain injury is a common cause of benign paroxysmal positional vertigo (BPPV) in the adult population, although it rarely occurs in adolescents 10, 11, 12.
- The first-order information processing from the inner ear involves a number of associated reflexes: the Vestibulo-Collic Reflex (VCR), the Cervico-Ocular Reflex (COR), the Cervico-Spinal Reflex (CSR), and the Cervico-Collic Reflex (CCR) 7. The Vestibulo-Collic Reflex (VCR) acts on the neck muscles to stabilize the head in space during body movements and is considered a righting reflex 7. The Vestibulo-Collic Reflex (VCR) lessens head oscillations that occur in more rapid, dynamic movements 13. Although the neural pathways and actions are not fully understood, it is suspected that Vestibulo-Collic Reflex (VCR) is mediated through the otolith organs and medial Vestibulo-Spinal Reflex (VSR) 14. The Cervico-Ocular Reflex (COR) acts on eye movements similar to the Vestibulo-Ocular Reflex (VOR), receiving input from the neck proprioceptors, and is of particular importance as a compensatory strategy if bilateral vestibular nerve loss occurs. The Cervico-Ocular Reflex (COR) is not thought to make any notable contributions to gaze stability in normal subjects because the gain is notably less than the Vestibulo-Ocular Reflex (VOR) 15. The Cervico-Spinal Reflex (CSR) acts on the extremities and is synonymous with the tonic neck reflex. The Cervico-Spinal Reflex (CSR) interacts with the Vestibulo-Spinal Reflex (VSR) 16. Finally, the Cervico-Collic Reflex (CCR) acts on deep neck muscles to assist in head stabilization during head and body movements. Body rotation with the head stabilized elicits the Cervico-Collic Reflex (CCR), whereas head rotation with the body stabilized elicits the Vestibulo-Ocular Reflex (VOR) 17.
- The semicircular canals are tubes coiled within your inner ear filled with liquid called the endolymph and lined with hair cells. Instead of sound waves, these tiny hairs react to sense rotational and angular movements of your head. The semicircular canals are mostly responsible for rotary motion or motion not in a straight line.
- A central processing mechanism
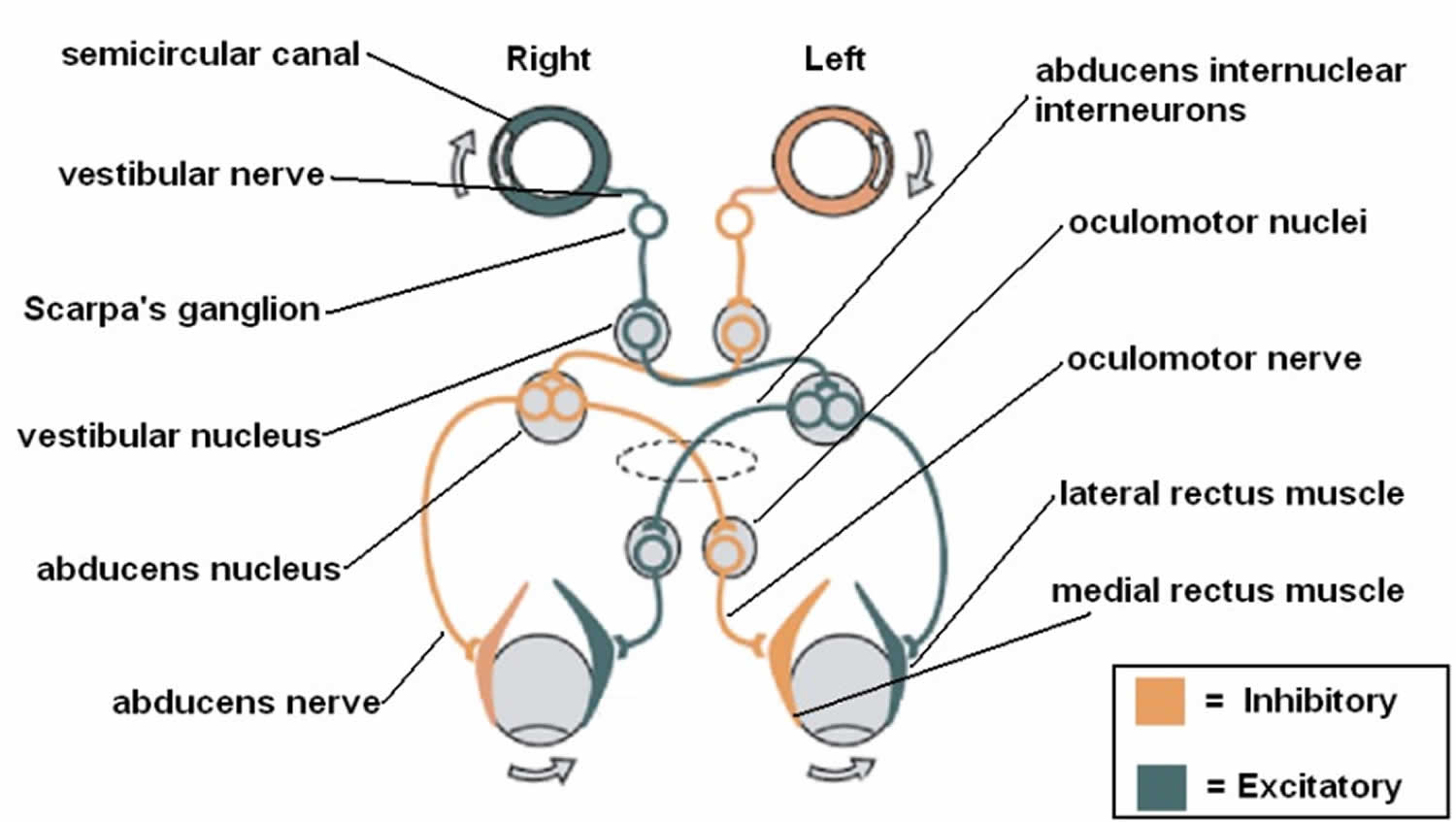
- Vestibular information is primarily processed in the brainstem via the cranial nerve eight (CN VIII) also known as vestibulocochlear nerve that consists of the vestibular and cochlear nerves 3, 1. The signals enter the vestibular nuclear complex, located in the medulla and pons 1. Four main nuclei comprise the vestibular nuclear complex (superior, inferior, medial, and lateral) 9, 18, 19. Most of the semicircular canal inputs synapse with the superior and medial vestibular nuclei via first-order sensory neurons of cranial nerve eight (CN VIII), while second-order sensory neurons send signals ipsilaterally and contralaterally via the medial longitudinal fasciculus to the oculomotor cranial nerves, oculomotor nerve (the third cranial nerve; CN III), trochlear nerve (fourth cranial nerve; CN IV), and abducens nerve (sixth cranial nerve; (CN VI). Lastly, a third motor neuron excites the extraocular muscles, causing conjugate eye movements that are equal to and opposite of head movements 20, 7. This results in gaze stabilization during head movements.
- The vestibular system is unique in that it also sends information via the inferior cerebellar peduncle to the cerebellum to modulate or fine-tunine the Vestibulo-Ocular Reflex (VOR). The flocculonodular lobe and fastigial nuclei are responsible for this fine-tuning. Vestibular data is also processed by the cerebellum, which then aids in the regulation of vestibular reflexes, maintaining posture, and coordination 21, 18. Other connections to the cerebral cortex, thalamus, and reticular formation assist in coordinating efforts between other central networks and the vestibular system, allowing for enhanced spatial awareness 7.
- There are additional vestibular connections to the thalamus, cerebral (vestibular) cortex, and reticular formation. These extensive connections result in a coordinated effort between the vestibular system and other central networks to contribute to arousal and conscious awareness of the head and body in space 20.
- A motor output which are the eye muscles.
- The central nervous system (brain and spinal cord) is the central processing mechanism, which sends its outputs to the spinal cord and eye muscles to generate the Vestibulo-Ocular Reflex. Each semicircular canal has excitatory projections to a pair of extraocular muscles in each eye and inhibitory projections to an antagonistic pair of muscles 8. In addition to the Vestibulo-Ocular Reflex (VOR), you also have a Vestibulo-Spinal Reflex (VSR), which prevents falls by maintaining head and postural stability. It does this by creating compensatory body movements 5. To attain an even better orientation, this information also goes to cortical structures (posterior insular vestibular cortex) for further integration with tactile, auditory, and proprioceptive inputs. Actions of the Vestibulo-Ocular Reflex (VOR) and Vestibulo-Spinal Reflex (VSR) become controlled and continuously adjusted as needed by the central nervous system (brain and spinal cord) 5.
The inner ear is innervated by cranial nerve eight (CN VIII) also known as vestibulocochlear nerve that consists of the vestibular nerve and cochlear nerve 1. The vestibular nerve is primarily responsible for maintaining body balance and eye movements, while the cochlear nerve is responsible for hearing 2. Damage to the vestibular nerve results in vertigo, a balance disorder, and nystagmus 2.
Ewald’s three laws for vestibular diagnostic testing 7
- Stimulation of a semicircular canal generates eye movements in the plane of that canal.
- In the horizontal semicircular canals, movement of the endolymph toward the ampulla (ampullopetal) causes deflection of the cupula toward the utricle (utriculopetal) and results in excitation (greater neural activity) compared to movement away from the utricle (utriculofugal).
- In the vertical semicircular canals (anterior and posterior), movement of the endolymph away from the ampulla (ampullofugal) causes greater deflection of the cupula away from the utricle (utriculofugal) and results in a greater excitation compared to utriculopetal movement, which is opposite to that in the horizontal canals.
Figure 5. Semicircular canals and the otolith organs
Footnote: The 3 semicircular canals (anterior, posterior, horizontal) sense angular head accelerations and the otolith organs (saccule, uticle) sense linear head accelerations, providing internal cues for orientation of position in space, movement, gaze stabilization, and postural control.
[Source 6 ]Figure 6. Semicircular canal anatomy
Footnote: The semicircular canals are part of the vestibular system of the inner ear. The semicircular canals are curved, looped structures that are referred to according to their orientation: lateral (horizontal), anterior (superior), and posterior. These structures are positioned at roughly right angles in relation to one another. The lateral semicircular canal, the shortest of the three, is set at an angle of about 30 degrees to the horizontal plane. The anterior and posterior loops are in diagonal vertical planes that intersect at a 90-degree angle.
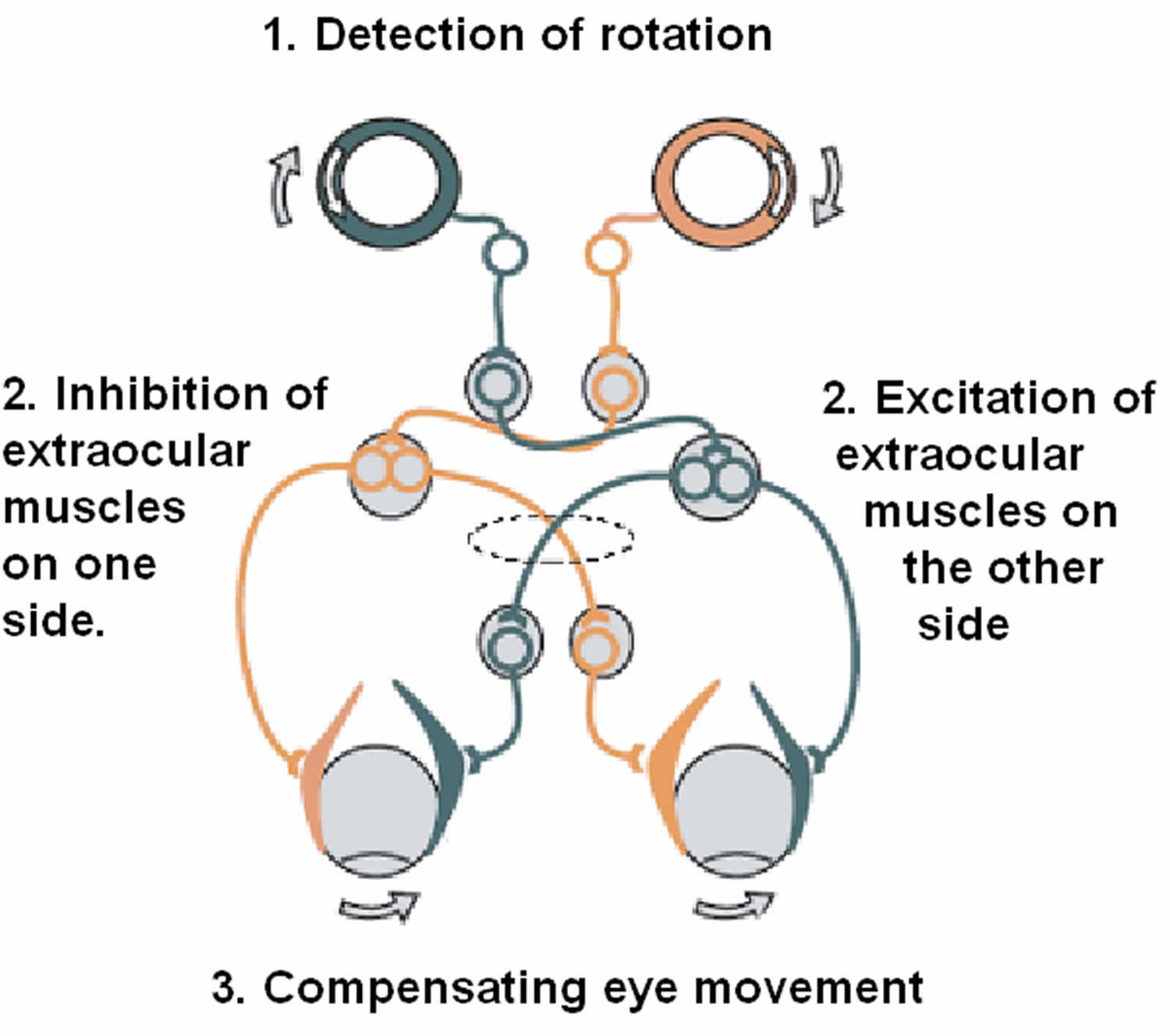
Figure 7. Vestibulo-ocular reflex (VOR)
Footnote: The vestibulo-ocular reflex (VOR). A rotation of the head is detected, which triggers an inhibitory signal to the extraocular muscles on one side and an excitatory signal to the muscles on the other side. The result is a compensatory movement of the eyes.
[Source 3 ]Sense of Equilibrium
The sense of equilibrium (balance) is really two senses:
- Static equilibrium and
- Dynamic equilibrium—that come from different sensory organs.
The organs of static equilibrium sense the position of the head, maintaining balance, stability and posture when the head and body are still. When the head and body suddenly move or rotate, the organs of dynamic equilibrium detect such motion and aid in maintaining balance.
Static Equilibrium
The organs of static equilibrium are in the vestibule, a bony chamber between the semicircular canals and the cochlea. The membranous labyrinth inside the vestibule consists of two expanded chambers—a utricle and a saccule (see Figures 2 and 3).
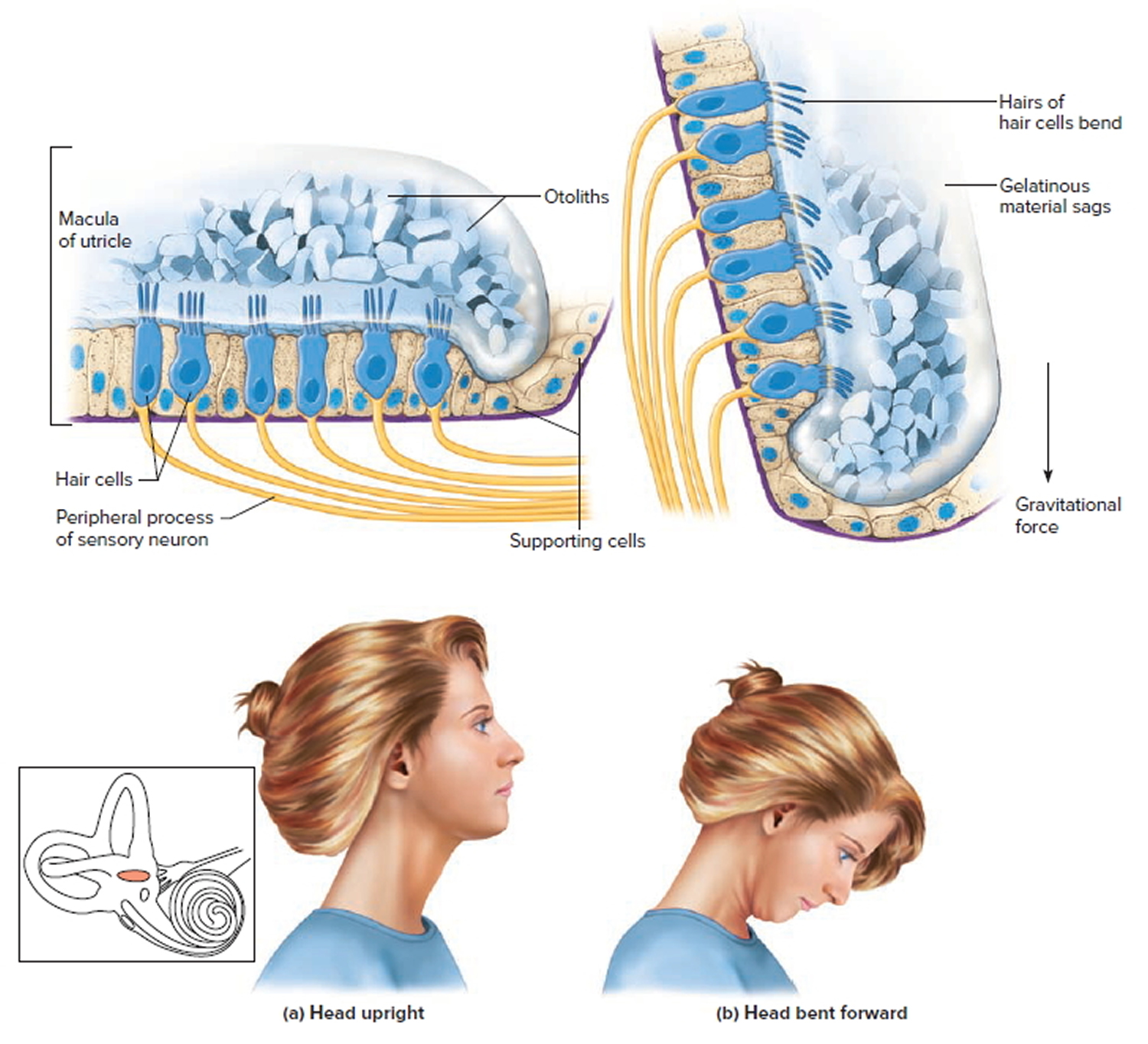
The saccule and utricle each have a tiny structure called a macula. Maculae have many hair cells, which serve as sensory receptors. The hairs of the hair cells project into a mass of gelatinous material, which has grains of calcium carbonate (otoliths) embedded in it. These particles add weight to the gelatinous structure.
Bending the head forward, backward, or to either side tilts the gelatinous masses of the maculae, and as they sag in response to gravity, the hairs projecting into them bend. This action causes the hair cells to signal the sensory neurons associated with them in a manner similar to that of hair cells associated with hearing. The resulting action potentials are conducted into the central nervous system on the vestibular branch of the vestibulocochlear nerve, informing the brain of the head’s new position. The brain responds by adjusting the pattern of motor impulses to skeletal muscles, which contract or relax to maintain balance.
Figure 8. Inner ear maculae respond to changes in head position
Footnote:The hair cells within the utricle are surrounded by a gelatinous substance and hold the microscopic otoliths (otoconia). (a) Orientation of hair cells within the macula of the utricle with the head in an upright position. (b) Macula of the utricle with the head bent forward, which results in a deflection of the hair cells due to a gravitational pull.
Dynamic Equilibrium
The organs of dynamic equilibrium are the three semicircular canals in the labyrinth. They detect motion of the head and aid in balancing the head and body during sudden movement. These canals lie at right angles to each other (see Figure 2).
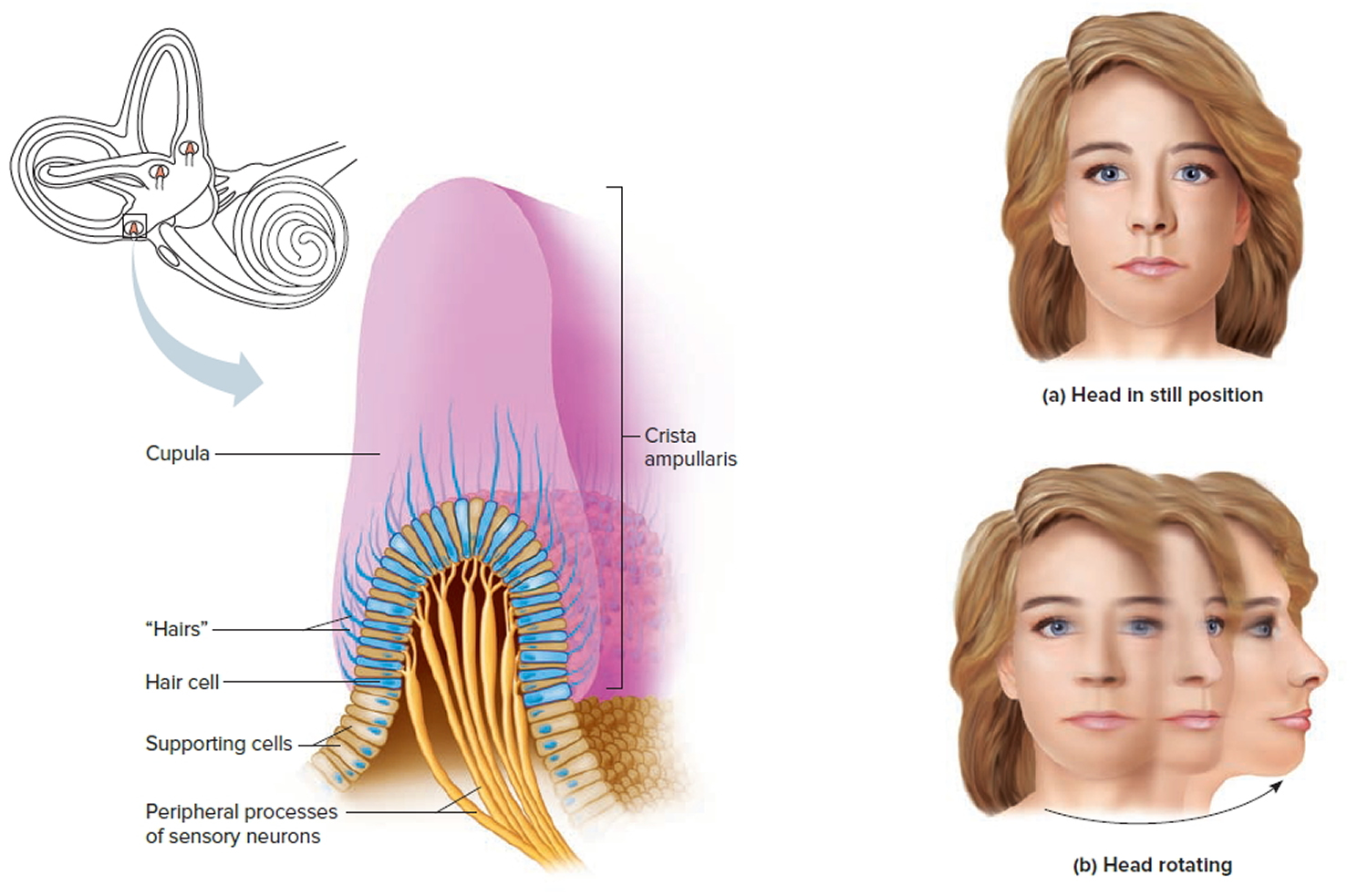
Suspended in the perilymph of the bony portion of each semicircular canal is a membranous semicircular duct that ends in a swelling called an ampulla, which
houses the sensory organs of the semicircular canals. Each of these sensory organs, called a crista ampullaris, contains a number of sensory hair cells and supporting cells. Like the hairs of the maculae, the hair cells of the crista ampullaris extend upward into a dome-shaped, gelatinous mass called the cupula (Figure 6). When the head is stationary, the cupula of the crista ampullaris remains upright. When the head is moving rapidly, the cupula bends opposite the motion of the head, stimulating sensory receptors.
Rapid movement of the head or body stimulates the hair cells of the crista ampullaris (Figure 9). At such times, the semicircular canals move with the head or body, but the fluid inside the membranous ducts remains stationary. Imagine turning rapidly while holding a full glass of water. This action bends the cupula in one or more of the canals in a direction opposite that of the head or body movement, and the hairs embedded in it also bend. The stimulated hair cells signal their associated neurons, which conduct impulses to the brain. The brain interprets these impulses as a movement in a particular direction.
Parts of the cerebellum are particularly important in interpreting impulses from the semicircular canals. Analysis of such information allows the brain to predict the consequences of rapid body movements. By modifying signals to appropriate skeletal muscles, the cerebellum can maintain balance.
Other sensory structures aid in maintaining equilibrium. For example, certain mechanoreceptors (proprioceptors), particularly those associated with the joints of the neck, inform the brain about the position of body parts. In addition, the eyes detect changes in position that result from body movements. Such visual information is so important that even if the organs of equilibrium are damaged, a person may be able to maintain normal balance by keeping the eyes open and moving slowly.
The nausea, vomiting, dizziness, and headache of motion sickness arise from sensations that don’t make sense. The eyes of a person reading in a moving car, for example, signal the brain that the person is stationary, because the print doesn’t move. However, receptors in the skin detect bouncing, swaying, starting, and stopping as the inner ear detects movement. The contradiction triggers the symptoms. Similarly, in a passenger of an airplane flying through heavy turbulence, receptors in the skin and inner ear register the chaos outside, but the eyes focus on the immobile seats and surroundings.
To prevent or lessen the misery of motion sickness, focus on the horizon or an object in the distance ahead. Medications are available by pill (diphenhydramine and dimenhydrinate) and, for longer excursions, in a skin patch (scopolamine).
Figure 9. Dynamic inner ear balance organs (crista ampullaris) within the Semicricular ducts
Inner ear vertigo
Vertigo, a type of dizziness, is an unpleasant disturbance of spatial orientation or to the erroneous perception of movement 22. Vertigo involves a perceived movement either of one’s own body, such as swaying or rotation, or of the environment, or both. Alongside headache, dizziness and vertigo are among the more common symptoms with which patients present to physicians in general, not just to neurologists. Their lifetime prevalence is approximately 20% to 30% 23. Dizziness, a common symptom that affects more than 90 million Americans, has been reported to be the most common complaint in patients 75 years of age or older 24.
The relative frequencies of various syndromes presenting with dizziness and vertigo are listed in table 1.
Table 1. The relative frequencies of different dizziness and vertigo syndromes
| Diagnosis | Number of patients | Percent |
| Benign paroxysmal positioning vertigo | 1336 | 18.6 |
| Phobic postural vertigo | 1127 | 15.6 |
| Central vestibular vertigo | 893 | 12.4 |
| Basilar/vestibular migraine | 738 | 10.2 |
| Menière’s disease | 677 | 9.4 |
| Vestibular neuritis | 531 | 7.4 |
| Bilateral vestibulopathy | 367 | 5.1 |
| Vestibular paroxysmia | 284 | 3.9 |
| Psychogenic dizziness | 228 | 3.2 |
| Perilymph fistula | 44 | 0.6 |
| Dizziness syndromes of unclear etiology | 239 | 3.3 |
| Other | 741 | 10.3 |
| Overall | 7205 |
The important criteria for distinguishing among them are as follows 25:
- The type of dizziness/vertigo: rotatory vertigo resembles the sensation of being on a merry-go-round (in vestibular neuritis and other disorders), while postural vertigo resembles the sensation of riding in a boat (e.g., in bilateral vestibulopathy). Many patients use the term “dizziness” for lightheadedness without any sensation of movement (e.g., in drug intoxication).
- The duration of dizziness/vertigo: attacks may last for seconds or minutes (as in vestibular paroxysm) or hours (as in Menière’s disease or vestibular migraine). Persistent vertigo lasting days or weeks is seen in vestibular neuritis, among other conditions. Attacks of postural vertigo lasting minutes to hours can be produced, for example, by brainstem transient ischemic attacks.
- Precipitating and exacerbating factors of dizziness and vertigo: the symptoms arise at rest in some conditions (e.g., vestibular neuritis); they can also arise when the patient walks (as in bilateral vestibulopathy) or be induced by turning the head to the right or left (as in vestibular paroxysm). Other possible precipitating factors include turning in bed (as in benign paroxysmal positioning vertigo [BPPV]), coughing, pressing, and loud tones of a particular frequency (Tullio’s phenomenon, seen in perilymph fistula), as well as certain social or environmental conditions (e.g., phobic postural vertigo).
- The accompanying symptoms, if present, may arise from the inner ear – e.g., attacks of intense tinnitus, hearing impairment, and a pressure sensation in the ear, which are typical of Menière’s disease. Diplopia, sensory disturbances, dysphagia, dysarthria, and paralysis of arms and legs are symptoms of central origin that usually arise in the brainstem. Headache or a history of migraine may point to the diagnosis of vestibular migraine but can also be caused by brainstem ischemia or posterior fossa hemorrhage.
Acute Severe Dizziness
The patient who presents with sudden onset severe dizziness – in the absence of prior similar episodes – has the “acute severe dizziness” presentation 26. Patients with acute severe dizziness appear ill due to the dizziness and accompanying nausea and vomiting. Impaired ability to walk is also common. Though rigorous epidemiological studies are lacking, the most common cause is an acute lesion – presumed viral in origin – of the vestibular nerve on one side, so-called vestibular neuritis 27. The mechanism underlying vestibular neuritis is the eighth cranial nerve is affected in vestibular neuritis. Patients with vestibular neuritis nearly always report true vertigo, which is characteristically described as visualized spinning of the environment. The symptoms are typically severe for 1-2 days with gradual resolution over weeks to months. It is exceedingly rare to have more than one bout of vestibular neuritis, so an alternative diagnosis should be considered whenever more than one episode is reported.
It is now clear that a small stroke within the posterior fossa can present as acute severe dizziness, closely mimicking vestibular neuritis 28, 29, 30. The first step to distinguishing vestibular neuritis from stroke is asking the patient about other neurological symptoms such as focal numbness, focal weakness, or slurred speech. Mild double vision can result from a peripheral vestibular lesion so this symptom is not a reliable discriminator. The next step is the physical examination. Patients with vestibular neuritis have highly characteristic exam features. Only in an extremely rare case can all of the vestibular neuritis exam features be mimicked by a stroke.
Nystagmus in Acute Severe Dizziness
Nystagmus is a term used to describe alternating slow and fast movements of the eyes. These alternating movements give the appearance that the eyes are beating toward one or more directions. Patients with vestibular neuritis have a peripheral vestibular pattern of nystagmus. In this setting, the peripheral vestibular pattern is a unidirectional, principally horizontal pattern of nystagmus. This description means that the nystagmus beats is in only one direction (i.e., a left-beating nystagmus never converts to right-beating, or a right-beating nystagmus never converts to left-beating). Patients with a peripheral disorder demonstrate nystagmus to the contralateral side which suppresses with visual fixation. Nystagmus improves with gaze towards the lesion and worsens with gaze opposite the lesion 31. Conversely, bi-directional gaze-evoked nystagmus (i.e., right beating nystagmus present with gaze toward the right, and left-beating nystagmus present with gaze toward the left side) is a central nervous system pattern of nystagmus. Other central nervous system patterns are pure torsional nystagmus or spontaneous vertical (typically downbeat) nystagmus. With an acute peripheral vestibular lesion, the only pattern of nystagmus that can result is unidirectional nystagmus. In acute severe dizziness presentations, any other pattern should be considered a central nervous system sign. Patients often prefer to keep their eyes closed early on, but the eyes should be opened and the pattern of nystagmus defined.
Nystagmus in vestibular neuritis is spontaneous (i.e., present in primary gaze) for at least the first several hours of symptoms. Following this initial time period, the nystagmus may only be identified during gaze testing (i.e., having the patient look to each side) or if visual fixation is blocked. Patients can suppress peripheral vestibular nystagmus by visual fixation on a target, so removing the patient’s ability to fixate can bring out the spontaneous nystagmus. The simplest way to block fixation is to place a blank sheet of paper a few inches in front of the patient and then observe for spontaneous nystagmus from the side.
The reason for the characteristic pattern of nystagmus in vestibular neuritis is an imbalance in the peripheral vestibular signals to the brain. Normally, the peripheral vestibular system on each side has a baseline firing rate of action potentials that functions to drive the eyes toward the other side. When the peripheral vestibular system on each side is intact, the input from each side is balance so the eyes remain stationary. When an acute lesion occurs on one side, the input from the opposite side is unopposed. As a result, the eyes will be “pushed” toward the lesioned side. This movement of the eyes is the slow phase of nystagmus. When the eyes reach a critical point off center, the brain responds by generating a corrective eye movement to move the eyes back. This is the fast phase of nystagmus. Since the direction of the fast phase gives the appearance that the eyes are beating in that direction, an acute left peripheral vestibular lesion leads to spontaneous right beating nystagmus. Over time, the asymmetry resolves or the brain compensates for the asymmetry.
Vascular Causes of Acute Severe Dizziness
Although vestibular neuritis is the most common cause of the acute dizziness presentation, no laboratory or imaging test exists to confirm a viral etiology. A peripheral vestibular lesion can be caused by a vascular occlusion of the blood supply to the peripheral vestibular components, though presumably this cause is much less common.
Stroke should be a serious consideration in the patient who presents with the acute dizziness presentation. Dizziness is a symptom of stroke in 50% of stroke presentations 32. Most stroke patients that report dizziness as a symptom have other prominent central nervous system features, but a small stroke of the cerebellum or brain stem can present with isolated dizziness (i.e., dizziness without other accompanying central nervous system signs or symptoms). In a population-based study, about 3% of patients with dizziness had a stroke etiology, but less than 1% of patients with isolated dizziness had stroke as the etiology 33. However, a prospective study of 24 patients with acute severe dizziness reported 6 patients (25%) with stroke etiology 28. Patients with stroke presenting as isolated dizziness may report imbalance, true vertigo, a more vague dizziness sensation, or a combination of these. Nausea and vomiting are also common, as they are with vestibular neuritis. Unfortunately, computerized tomography (CT) scans are an extremely insensitive test for acute stroke presentations in general 34, and particularly so for infarction within the posterior fossa 35. A stroke within the posterior fossa may not appear on a CT scan for days or weeks because of artifacts or poor resolution. Because of this, CT should never be considered as a means of excluding stroke. Magnetic resonance imaging (MRI) is a much more sensitive test, but is not a practical test to screen for stroke in emergency department dizziness presentations. Like CT, the sensitivity of the test is the lowest for stroke of the posterior fossa 36.
The key features discriminating stroke from vestibular neuritis are the pattern of nystagmus and the results of the head thrust test. Down-beating nystagmus or bidirectional gaze-evoked nystagmus are both immediate indications that the localization must be in the central nervous system. These patterns are not caused by lesions of the peripheral vestibular system. This is the reason that an examination of ocular movements is required before a diagnosis is even considered. Another highly suspicious pattern of nystagmus is a pure torsional pattern. There are now case reports of patients who have unidirectional horizontal nystagmus and a stroke etiology so the pattern of nystagmus should not be the sole criterion.3-5 A patient with unidirectional nystagmus, a positive head thrust in the direction opposite the fast phase of nystagmus, and no other neurological features can be diagnosed with vestibular neuritis with a high level of certainty. It would take a well placed and small stroke to cause the peripheral vestibular pattern of nystagmus and a corresponding positive head thrust test without any other central nervous system features. Though all patients with vestibular neuritis are unsteady walking, the inability to walk is another red flag.5 Finally, a person’s risk for stroke based on stroke risk factors should be considered. Though no validated scale exists to grade stroke risk based on stroke risk factors in this population, a stroke work-up is reasonable in patients with a high risk for stroke. One should not be over reliant on stroke risk factors as discriminators, however, since other stroke mechanisms – such as arterial dissection – occur in the absence of stroke risk factors.
Management of Acute Severe Dizziness
The management of the acute dizziness presentation begins with supportive care. If stroke is suspected then a neuro-imaging study should be considered. Though CT could serve as the initial study, a normal result on CT should provide little confidence that stroke can be excluded. In this situation, an MRI or hospital admission for close observation should be considered. If stroke is confirmed to be the cause and the patient presents within three hours of onset, thrombolytic treatment should be considered. A short course of corticosteroids should be considered for patients with vestibular neuritis. A randomized controlled trial showed that patients with vestibular neuritis treated with corticosteroids within three days of symptom onset had a higher likelihood of recovery of the peripheral vestibular caloric response at 12 months 37. However this study did not test whether the patient’s functional or symptomatic outcome improved, and corticosteroids are not without potential side effects. After the initial severe symptomatic time period, it is important that patients resume activities because this helps the brain to compensate for the asymmetry of vestibular signals. A formal vestibular therapy program has been shown in a randomized trial to improve outcomes in patients with vestibular neuritis 38.
Treatment for vertigo
The treatment of dizziness and vertigo 25 may include medication, physical therapy, and psychotherapy; a few limited cases may require surgical treatment. Before the treatment is begun, the patient should be told that the prognosis is generally good: many of these conditions have a favorable spontaneous course, both because peripheral vestibular dysfunction tends to improve and because there is central vestibular compensation for asymmetrical peripheral vestibular tone. Moreover, most of these conditions can be treated successfully.
Labyrinthitis
Labyrinthitis is an inflammatory disorder of the membranous labyrinth, affecting both the vestibular and cochlear end organs 31. It may present unilaterally or bilaterally, and similar to vestibular neuronitis, it is often preceded by an upper respiratory infection. This disorder occurs when infectious microorganisms or inflammatory mediators invade the membranous labyrinth, damaging the vestibular and auditory end organs. Potential etiologies include viral pathogens, bacterial invasion, bacterial toxins, and systemic disease 31.
Viral labyrinthitis usually occurs in adults in their fourth to seventh decades of life. Bacterial labyrinthitis may result from both otogenic and meningitic infection, progressing to involve the labyrinth. Labyrinthitis of otogenic origin can be observed in any age group and may result from cholesteatoma or otitis media 31. Meningitic labyrinthitis is more common in children less than 2 years of age, who are more susceptible to developing meningitis 31. Otogenic infections typically cause unilateral symptoms while meningitic infections cause bilateral symptoms 31.
Unlike vestibular neuronitis, patients with labyrinthitis present with complaints indicative of both vestibular and cochlear damage. Vertigo presents suddenly and is accompanied by hearing loss 31. Electronystagmography may reveal nystagmus, and audiometry will reveal a sensorineural hearing loss or mixed hearing loss if middle ear effusion is present 31. Depending on the source of infection, patients may also present with findings consistent with otitis media, mastoiditis, or meningitis.
Treatment is aimed primarily at eradication of the underlying infection and supportive care 31. Middle ear effusions and mastoiditis should be drained and treated with antibiotics. Meningitis should be treated with culture-directed antibiotics with central nervous system penetration and appropriate consultation. Anti-emetics and anti-nausea medications are helpful during the acute phase.
Peripheral vestibular vertigo
A functional classification of peripheral vestibular disorders divides them into three main types, which can be distinguished on the basis of their typical symptoms and signs (table 2):
- Chronic, bilateral dysfunction of the vestibular nerve or the peripheral vestibular organs;
- Acute, unilateral vestibular dysfunction;
- Paroxysmal pathological excitation or inhibition of the vestibular nerve or vestibular organs.
Table 2. The presenting manifestations and causes of peripheral vestibular types of vertigo
| Type of disorder | Presenting manifestations | Examples and causes |
|---|---|---|
| Chronic, bilateral peripheral vestibular dysfunction |
| Bilateral vestibulopathy due to (e.g.):
|
| Acute/subacute unilateral vestibular dysfunction (labyrinth and/or vestibular nerve) with asymmetrical vestibular tone |
|
due to reactivation of a latent herpes simplex virus type 1 infection |
| Inappropriate unilateral paroxysmal excitation or loss of function of the peripheral vestibular system |
|
|
Benign paroxysmal positioning vertigo (BPPV)
This is the most common type of vertigo; it mainly affects older patients (Table 1) and has a lifetime prevalence of 2.4% 23. It is characterized by brief attacks of rotational vertigo, accompanied by vertical positioning nystagmus that rotates toward the lower of the two ears and beats toward the forehead. Nystagmus is a term used to describe alternating slow and fast movements of the eyes 26. These alternating movements give the appearance that the eyes are beating toward one or more directions. The attacks are precipitated by reclination of the head, or by lateral positioning of the head or body, with the affected ear downward 22. After a change in position of one of these types, rotational vertigo and nystagmus arise after a latency of a few seconds and then take a characteristic crescendo-decrescendo course, lasting a total of 30 to 60 seconds 22. The nystagmus corresponds to a so-called ampullofugal excitation of the affected posterior vertical semicircular canal of the affected (lower) ear.
More than 90% of cases are idiopathic 22; the remaining, symptomatic cases are most commonly due to head trauma, vestibular neuritis, or Menière’s disease 39. Benign paroxysmal positioning vertigo is called “benign” because it usually resolves spontaneously within a few weeks or months; in some cases, however, it can last for years. If left untreated, it persists in about 30% of patients. Benign paroxysmal positioning vertigo also arises with greater than usual frequency after prolonged bed rest necessitated by other diseases, or after surgery. Benign paroxysmal positioning vertigo of the horizontal semicircular canal is rare and is precipitated by rotation of the head in the recumbent position.
The canalolithiasis hypothesis explains all of the manifestations of positioning vertigo and nystagmus 40. According to this hypothesis, the condition is due to the presence of agglomerates of many otoconia (small crystals of calcium carbonate in the saccule and utricle of the inner ear) that nearly fill the lumen of the semicircular canal and are freely mobile within it, instead of the small pieces of particulate matter that adhere firmly to the cupula (so-called cupulolithiasis).
Benign paroxysmal positioning vertigo is treated with positioning maneuvers: rapid repositioning of the head can move the otoconial agglomerate out of the semicircular canal so that it can no longer cause positioning vertigo 22. The treatments of choice are the Semont 41 and Epley maneuvers. For the Semont maneuver, see Figure 7; the Epley maneuver involves rotation of the patient in the recumbent position with the head hanging down. Most patients can perform these maneuvers themselves after brief training. The two are equally effective, and the cure rate is more than 95% within a few days, as shown by multiple controlled studies and meta-analyses 42. The rate of recurrence of benign paroxysmal positioning vertigo is about 15% to 30% per year. The symptoms eventually recur at some time after effective treatment in about 50% of patients 43 but can then be treated effectively a second time in the same manner.
Epley Maneuver to treat BPPV Vertigo
Deep Head Hanging Maneuver to treat BPPV Vertigo
Lempert (BBQ) Maneuver to treat BPPV Vertigo
Half Somersault Maneuver to treat BPPV Vertigo
Figure 10. The treatment of benign paroxysmal positioning vertigo with the Semont maneuver
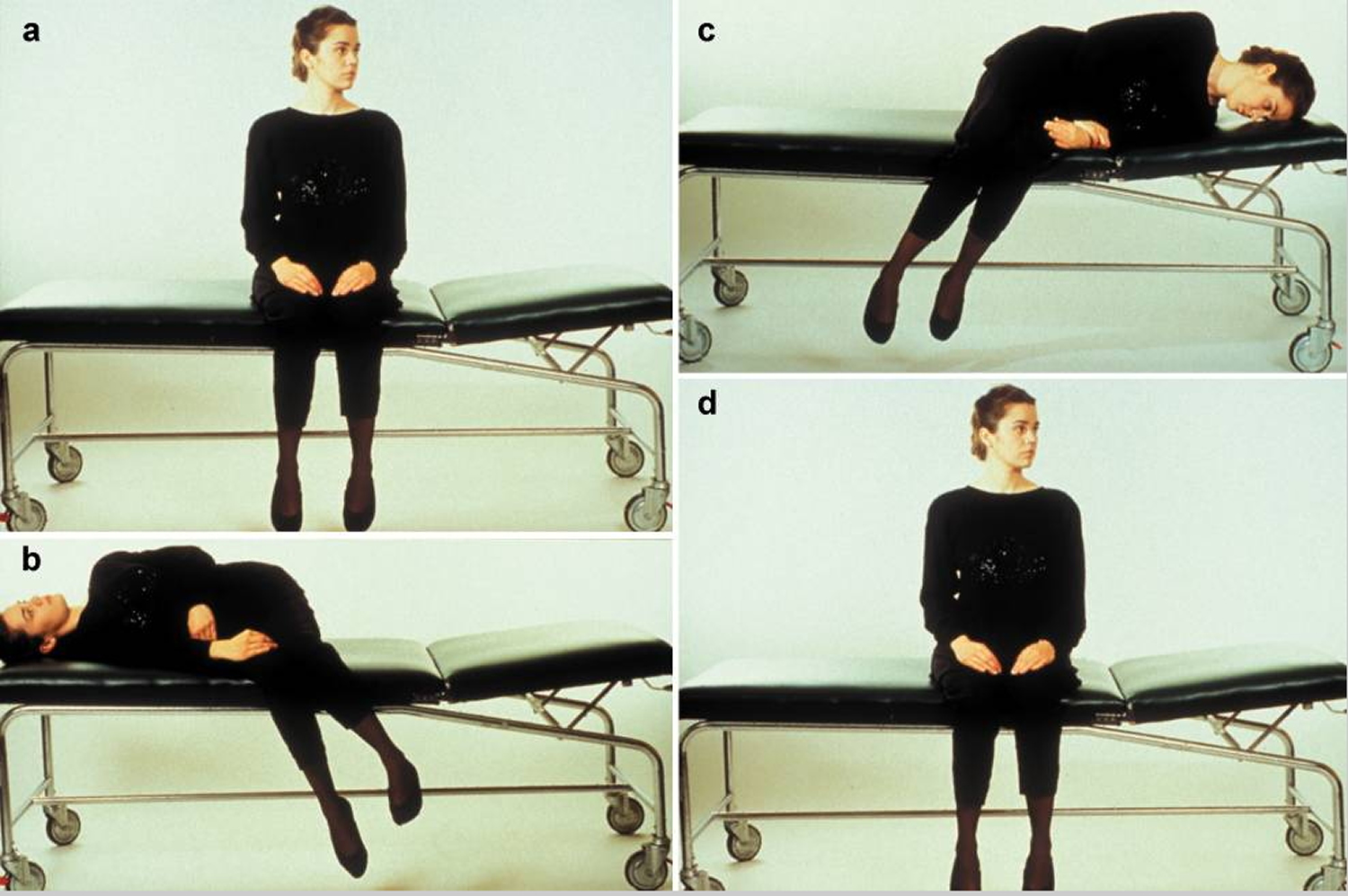
Footnote: The illustration shows the treatment of benign paroxysmal positioning vertigo (BPPV) due to canalolithiasis of the right posterior semicircular canal.
a) In the initial, sitting position, the head is turned 45° to the side of the unaffected (“healthy”) ear.
b) The patient is laid on the right side, i.e., on the side of the affected ear, while the head is kept in 45° of rotation to the other side. This induces movement of the particulate matter in the posterior semicircular canal by gravity, leading to rotatory nystagmus toward the lower ear that extinguishes after a brief interval. The patient should maintain this position for about one minute.
c) While the head is still kept in 45° of rotation toward the side of the healthy ear, the patient is rapidly swung over to the side of the unaffected ear, so that the nose now points downward. The particulate matter in the semicircular canal now moves toward the exit from the canal. This position, too, should be maintained for at least one minute.
d) The patient returns slowly to the initial, sitting position. The particulate matter settles in the utricular space, where it can no longer induce rotatory vertigo. This sequence (a-d) should be performed three times in a row three times per day, in the morning, at noon, and at night. Most patients are free of symptoms after doing this for three days.
[Source 22]Vestibular neuritis
The clinical syndrome of vestibular neuritis is characterized by the following:
- Persistent rotational vertigo with a pathological inclination of the visual vertical axis toward the side of the affected labyrinth
- Spontaneous, horizontally rotating nystagmus toward the unaffected side, producing apparent movement of the environment (“oscillopsia”)
- Gait deviation and falling tendency toward the affected side
- Nausea and vomiting
- Unilateral dysfunction of the horizontal semicircular canal, as revealed by the Halmagyi-Curthoys head impulse test 44 for the function of the vestibulo-ocular reflex, as well as by caloric testing.
A viral and/or autoimmune as cause for vestibular neuritis is probable but has not yet been proven. Autopsy studies have revealed inflammatory degeneration of the vestibular nerve, the presence of viral DNA from herpes simplex virus type I, and the so-called “latency-associated transcript” (LAT) in vestibular ganglion cells 45. The treatment is symptomatic, causal, and physiotherapeutic:
- Symptomatic treatment: antivertiginous medications, such as 100 to 300 mg of dimenhydrinate, should be given only in the first three days and only if necessary to treat severe nausea and vomiting, because they delay the development of central compensation mechanisms.
- “Causal” treatment: a four-armed, placebo-controlled trial was performed, based on the assumption that vestibular neuritis is caused by the reactivation of a latent herpes simplex virus type 1 infection. The trial revealed that monotherapy with a glucocorticoid-methylprednisolone at an initial dose of 100 mg daily, reduced in 20-mg steps every four days, significantly improved the recovery of peripheral vestibular function. The administration of valacyclovir alone had no effect, nor did its administration in combination with the glucocorticoid have any additional effect 46.
- Physical therapy: a further principle of treatment is the promotion of central compensation by physical therapy. Equilibrium training significantly lessens the time required for vestibulospinal compensation and postural regulation to develop 47. Voluntary eye movements and fixation are exercised in order to improve impaired visual fixation; furthermore, active head movements are exercised to realign the vestibular reflex, as well as balance tasks and goal-directed movements.
Menière’s disease
This condition is probably due to labyrinthine endolymphatic hydrops with periodic rupturing of the membrane that separates the endolymphatic and perilymphatic spaces. These ruptures precipitate the paroxysmal attacks that last a few minutes to hours 48. The ultimate etiology is impaired resorption in the endolymphatic sac due to perisaccular fibrosis or to obliteration of the endolymphatic duct. Attacks are produced when rupture of the endolymphatic tube causing calcium-induced depolarization of the vestibulocochlear nerve. A classic Menière’s attack consists of rotatory vertigo, tinnitus, hearing impairment, and pressure sensation in one ear. The lifetime prevalence of this condition is approximately 0.5% 23. It usually begins on one side, and the frequency of attacks is highly variable. Menière’s disease becomes bilateral in 50% of cases 49 and is the second most common cause of bilateral vestibulopathy.
Its treatment is based on two principles:
- Treatment of individual attacks: vertigo and nausea can be improved with antivertiginous medications just as in the treatment of other types of acute labyrinthine dysfunction. For example, 100 mg dimenhydrate suppositories can be used.
- Attack prophylaxis: this type of treatment is aimed at improving the underlying endolymphatic hydrops. Despite the high prevalence of Menière’s disease and the large number of clinical studies that have been performed, there is still no treatment of this type that has been conclusively shown to be effective. The spectrum of recommendations ranges from a sodium-free diet to diuretics, transtympanic gentamicin instillation (20 to 40 mg given repeatedly, at intervals of several weeks, until symptoms improve), betahistine, and surgical procedures 48. A beneficial effect on the frequency of attacks has been reported for transtympanic gentamicin 42 and for the prolonged high-dose administration of betahistine hydrochloride (48 mg tid for 12 months). The latter dose of betahistine hydrochloride is currently recommended on the basis of a recently reported observational treatment study in 112 patients who were treated for at least 12 months at doses of 16, 24, or 48 mg tid 50. The highest dose led to a statistically significantly greater reduction of attack frequency and was well tolerated.
Vestibular Schwannoma
Vestibular schwannoma is the most common intracranial neoplasm producing vestibular symptoms, affecting one in every 100,000 people per year 51. These are usually slow-growing, benign tumors that originate from the Schwann cells lining the vestibular portion of cranial nerve VIII 31. Occasionally these tumors arise from the cochlear branch of the eighth nerve, but this is reported in less than 5% of cases 52. Patients may present with either unilateral or bilateral vestibular schwannoma. Bilateral vestibular schwannoma is associated with neurofibromatosis II, which is additionally characterized by glioma, meningioma, subcapsular ventricular opacities, and less frequently, peripheral neurofibromata and café au lait spots.
Patients may present with episodic or positional vertigo, disequilibrium, tinnitus, and usually asymmetric hearing loss. Early in the disease, when the tumor is small, patients complain of dizziness, hearing loss, and tinnitus, due to compression of the vestibulocochlear nerve. The slow growth often allows for central compensation, alleviating vertigo. With continued growth, the tumor can press against the facial or trigeminal nerve causing facial weakness and numbness, respectively. Eventually, the tumor grows to a size where it compresses the brainstem and cerebellum causing truncal ataxia, dysmetria, disequilibrium, and possibly death.
Diagnosis begins with a thorough history and physical examination. An audiogram is important in documenting hearing loss and any asymmetries which may exist. If you suspect vestibular schwannoma, then imaging is necessary. Computed tomography of the head with contrast is helpful, but magnetic resonance imaging with and without enhancement is the preferred imaging modality.
Once vestibular schwannoma is confirmed radiographically, a decision should be made on how to proceed with treatment. Treatment options include surgical excision, radiation therapy, and observation with serial magnetic resonance imaging. In making this decision, one should consider the size of the lesion, age and health of the patient, and what symptoms are present. These patients should be referred to a neuro-otologist for management of their care.
Central vestibular syndromes
Central vestibular syndromes are mainly caused by lesions of the vestibular pathways, which arise in the vestibular nuclei in the caudal portion of the brainstem and proceed to the cerebellum, thalamus, and vestibular cortex, or by damage to the vestibulocerebellum 22. Pathological excitation is a rare cause, as occurs, for example, in the paroxysmal brainstem attacks with ataxia that can be produced by multiple sclerosis or vestibular epilepsy. The common causes of central vestibular vertigo include vestibular migraine and ischemic lesions in the brainstem 22. Furthermore, central vestibular disturbances arise in the setting of certain ocular motor disorders such as downbeat and upbeat nystagmus, as attacks in episodic ataxia type 2, and in vestibular migraine. These individual disorders, and the treatment of each, will be discussed in the following sections.
Motion sickness
Motion sickness is attributed to an incongruence in the sensory input from the vestibular, visual, and somato-sensory systems 53. Motion sickness occurs while riding in a car, boat, or airplane if the vestibular and somato-sensory systems sense movement, but the visual system does not.
On the first sensation of motion sickness, efforts should be made to bring vestibular, visual, and somato-sensory input back in congruence. For example, a person on a boat who starts to feel seasick should immediately watch the horizon. Seasickness can be prevented by applying a scopolamine patch (Transderm-Scop) behind one ear at least four hours before boating 54.
Vertebrobasilar Ischemic Stroke
The blood supply to the brainstem, cerebellum, and inner ear is derived from the vertebrobasilar system 31. Occlusion of any of the major branches of this system may result in vertigo. Symptoms of vertebrobasilar ischemic stroke are highly variable and depend on which of the three major circumferential branches are occluded; the posterior inferior cerebellar artery, anterior inferior cerebellar artery, or superior cerebellar artery. Numerous processes may occlude the vertebrobasilar system. The most common are atherosclerosis, emboli, and vertebral artery dissection. Vertebral artery dissection can result from trauma or neck manipulation, or can occur spontaneously. Less common causes include subclavian steal syndrome, hypercoagulation disorders, and inflammatory conditions.
As mentioned earlier, the symptoms associated with ischemic stroke in this area are highly variable and greatly dependent upon which branch of the system is occluded. Occlusion of the posterior inferior cerebellar artery will cause a lateral medullary infarction and result in lateral medullary syndrome, also known as Wallenberg’s syndrome. Expected manifestations include vertigo, nystagmus, gait disturbance, ipsilateral limb ataxia and facial pain or numbness, contralateral body hemianesthesia, Horner’s syndrome, dysphagia, hoarseness, and rarely, facial nerve paralysis.
Lateral pontomedullary infarction secondary to occlusion of the anterior inferior cerebellar artery will result in lateral inferior pontine syndrome. This syndrome is characterized by symptoms similar to Wallenberg’s syndrome with notable differences. Involvement of cranial nerves VII and VIII results in ipsilateral facial paralysis and tinnitus and hearing loss, respectively. Dysphagia and hoarseness, however, are not apparent as cranial nerves IX and X nuclei are uninvolved with occlusion of the anterior inferior cerebellar artery.
Lateral superior pontine syndrome occurs when the superior cerebellar artery is occluded. With this syndrome, one can expect vertigo, nystagmus, gait disturbance, ipsilateral limb ataxia and facial pain or numbness, contralateral body hemianesthesia, and Horner’s syndrome. Distinguishing this syndrome is the finding of contralateral impairment of vibration and temperature due to medial lemniscus involvement.
A high index of suspicion must be kept with any patient presenting with spontaneous vertigo to avoid missing the diagnosis of ischemic stroke. It is essential to consider stroke in any acutely vertiginous patient with concomitant neurological signs and symptoms. Once vertebrobasilar ischemic stroke is suspected, an expeditious work-up is necessary. This should include a thorough physical examination, imaging, and neurology consultation for both evaluation and treatment.
Vertebrobasilar Insufficiency
Vertebrobasilar insufficiency is synonymous with a transient ischemic attack (TIA) of the vertebrobasilar system. By definition, patients experience symptoms similar to those detailed above, but the symptoms resolve within 24 hours. If left untreated, the disease process will eventually progress to stroke with permanent or long-lasting seqeulae. Risk factors and causes are identical to those for VIS. Forty-eight percent of patients who suffer a vertebrobasilar ischemic stroke report a transient ischemic attack in the preceding days or weeks 55. In fact, 29% of patients suffer from at least one episode of vertigo, a symptom of vertebrobasilar insufficiency, prior to their vertebrobasilar insufficiency 56. Patients suffering a vertebrobasilar transient ischemic attack are likely to progress to stroke more quickly than those experiencing transient ischemic attacks in the anterior territory.
Vertebrobasilar insufficiency is a common cause of vertigo in the elderly. Symptoms may last from minutes to hours, but typically average 8 minutes in duration. In as many as one third of patients, vertigo is the only manifestation of their disease. Although this disease should always be in the differential, several months of recurrent vertigo unaccompanied by other neurological signs suggests another disorder. The likelihood of immediate stroke is less in patients presenting with only episodic vertigo. Patients who also present with paresis, blindness, or altered consciousness, however, should be evaluated urgently for fear of impending stroke. Evaluation is similar to that of an ischemic stroke. Treatment includes antiplatelets, anticoagulation, possible thrombolysis and percutaneous transluminal angioplasty, and neurological consultation.
The sudden onset of vertigo in a patient with additional neurologic symptoms (e.g., diplopia, dysarthria, dysphagia, ataxia, weakness) suggests the presence of vascular ischemia.
Treatment of transient ischemic attack and stroke includes preventing future events through blood pressure control, cholesterol-level lowering, smoking cessation, inhibition of platelet function (e.g., aspirin, clopidogrel [Plavix], aspirin-dipyridamole [Aggrenox]) and, possibly, anticoagulation (warfarin [Coumadin]).
Acute vertigo caused by a cerebellar or brainstem stroke is treated with vestibular suppressant medication and minimal head movement for the first day. As soon as tolerated, medication should be tapered, and vestibular rehabilitation exercises should be initiated 57, 58.
Placement of vertebrobasilar stents may be considered in a patient with symptomatic critical vertebral artery stenosis that is refractory to medical management 59. Rarely, infarction or hemorrhage in the cerebellum or brainstem may present with acute vertigo as the only neurologic symptom 60. Given the risk of brainstem compression with a large cerebellar stroke, neurosurgical decompression may be indicated.
Downbeat and upbeat nystagmus
Two types of vertically beating central nystagmus are of special importance: downbeat nystagmus and upbeat nystagmus, each named after the direction of the rapid, beating phase. Downbeat nystagmus is the most common type of acquired, persistent nystagmus 61. Both types manifest themselves above all with swaying nystagmus and unsteadiness of gait and only secondarily with oscillopsia, i.e., apparent movement of the environment due to oscillation of the retinal image. In distinction to spontaneous nystagmus such as in vestibular neurits, downbeat nystagmus and upbeat nystagmus are types of fixation nystagmus, i.e., their intensity increases with visual fixation. Both downbeat nystagmus and upbeat nystagmus always indicate the presence of a central disturbance and possess special localizing significance. Downbeat nystagmus is usually due to bilateral dysfunction of the flocculus 62; its three common causes are cerebellar atrophy, ischemia, and Arnold-Chiari malformation 61. Upbeat nystagmus – which, unlike downbeat nystagmus, generally persists for no more than a few weeks – can be caused by paramedian medullary or pontomesenchephalic lesions, e.g., brainstem infarct or hemorrhage.
A randomized, placebo-controlled study of downbeat nystagmus has shown that the potassium-channel blockers 3,4-diaminopyridine 63 and 4-aminopyridine can significantly improve this type of nystagmus 64. The dosage is 5 to 10 mg tid; follow-up ECG is necessary. The effectiveness of this treatment has since been confirmed by multiple studies. 4-Aminopyridine seems to be effective against upbeat nystagmus as well, but this has been documented to date only in a single case study 65.
Episodic ataxia type 2
The familial episodic ataxias are rare genetic diseases of autosomal dominant transmission. There are at least two well-defined varieties. Type 2 (EA 2) is characterized by recurrent attacks of dizziness and ataxia that are precipitated by physical activity, stress, or alcohol and usually last for hours. In between attacks, more than 90% of patients have marked central ocular motor disturbances, often downbeat nystagmus. EA 2 is caused by mutations in the CACNA1A gene (PQ calcium channel gene). Most patients can be treated successfully with acetazolamide. If this treatment is ineffective, or if adverse effects such as kidney stones develop, patients with EA 2 can also be treated with 4-aminopyridine (5 mg tid) 66.
Aminopyridines are thus an effective treatment for downbeat nystagmus, upbeat nystagmus, and episodic ataxias type 2 which is well tolerated at the low dose that is generally used. These studies have also led to the development of a new principle of treatment; activation of cerebellar Purkinje cells through potassium-channel blockade enhances the cerebellar inhibitory influence on the vestibular and cerebellar nuclei.
Vestibular migraine or migraine with vestibular aura
Vestibular migraine is characterized by recurrent attacks that last minutes to hours and usually consist of rotatory vertigo 67, 68. It is the most common cause of spontaneously occurring attacks of vertigo (Table 1). Its lifetime prevalence is 0.98% 23. In more than 60% of patients, these attacks are associated with headache and/or photophobia or phonophobia; the remaining patients have attacks of vertigo alone. Most patients also have migraine attacks with or without an aura; this fact makes the condition easier to diagnose. In some patients, the diagnosis can be made only on the basis of a positive response to the treatment of the individual attacks with medication and to pharmacological prophylaxis. The prophylactic treatment of vestibular migraine is analogous to that of migraine with aura and consists of the administration of beta-blockers, valproic acid, and topiramate. No randomized, controlled studies on the efficacy of medications for vestibular migraine have yet been published.
Phobic postural vertigo
Phobic postural vertigo is the second most common diagnosis in a specialized neurological ambulatory clinic for dizziness and vertigo. This disorder is not found in the diagnostic repertoire of most neurologists and ENT specialists. Patients with phobic postural vertigo usually complain of swaying vertigo, lightheadedness, and gait unsteadiness that are continually present but fluctuate in severity. These symptoms are often accompanied by anxiety and are situationally dependent. The precipitating factor may be the presence of a large crowd, or waiting in the check-out line at a store; often, avoidance behavior results 25. The symptoms typically improve when the patient participates in sports or has had a small amount of alcohol to drink. The affected patients often have an obsessive-compulsive personality, in the sense of “accentuated” personality traits, with a marked tendency toward introspection and a need to “have everything under control.” The central problem in phobic postural vertigo is the patient’s attempt to establish conscious control over body equilibrium, which leads to a “spiral of self-observation.” When this happens, the body’s own movements may be perceived as movements of the outside world. The main features of this disorder and its treatment are summarized in the box. The clinical neurological examination and ancillary tests reveal no relevant pathological findings.
Clinical features
- The patient has postural vertigo with unsteadiness of stance and gait; the neurological examination and ancillary tests are generally unremarkable
- Fluctuating unsteadiness of stance and gait with attacks of fear of falling, but without an actual fall
- Anxiety and autonomic disturbances sometimes occur during or just after the attacks
- The attacks are precipitated or exacerbated by typical situations, e.g., crowds, empty spaces, driving
- The symptoms often improve during sporting activity or after the consumption of a small amount of alcohol
- Increasingly severe avoidance behavior is common
The patient’s personality is usually of an obsessive-compulsive or reactive-depressive type. At the onset of the disorder, there is often a vestibular disturbance (25%) or a situation giving rise to particular stress (70%).
Treatment
- A thorough diagnostic assessment to allay the patient’s fear of having a serious organic disease
- Psycho-educative therapy to inform the patient about the pathological mechanism and the precipitating factors and situations
- Desensitization by self-exposure, i.e., the deliberate seeking out of situations that precipitate vertigo. Light sporting activities are also helpful.
- If the symptoms persist, pharmacotherapy, e.g., with selective serotonin reuptake inhibitors, and/or cognitive behavioral therapy are indicated
Treatment markedly improves symptoms in about 70% of patients 69.
- Bordoni B, Mankowski NL, Daly DT. Neuroanatomy, Cranial Nerve 8 (Vestibulocochlear) [Updated 2023 May 22]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537359[↩][↩][↩][↩]
- Benoudiba F, Toulgoat F, Sarrazin JL. The vestibulocochlear nerve (VIII). Diagn Interv Imaging. 2013 Oct;94(10):1043-50. https://doi.org/10.1016/j.diii.2013.08.015[↩][↩][↩][↩]
- Somisetty S, M Das J. Neuroanatomy, Vestibulo-ocular Reflex. [Updated 2023 Jul 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK545297[↩][↩][↩][↩]
- Yoo H, Mihaila DM. Neuroanatomy, Vestibular Pathways. [Updated 2022 Nov 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557380[↩]
- Fetter M. Vestibulo-ocular reflex. Dev Ophthalmol. 2007;40:35-51. https://doi.org/10.1159/000100348[↩][↩][↩][↩][↩]
- Benign paroxysmal positional vertigo: Effective diagnosis and treatment. Shayna R. Cole, Julie A. Honaker. Cleveland Clinic Journal of Medicine Nov 2022, 89 (11) 653-662; https://doi.org/10.3949/ccjm.89a.21057[↩][↩]
- Wallace B, Lifshitz J. Traumatic brain injury and vestibulo-ocular function: current challenges and future prospects. Eye Brain. 2016 Sep 6;8:153-164. doi: 10.2147/EB.S82670[↩][↩][↩][↩][↩][↩]
- Sherman KR, Keller EL. Vestibulo-ocular reflexes of adventitiously and congenitally blind adults. Invest Ophthalmol Vis Sci. 1986 Jul;27(7):1154-9. https://iovs.arvojournals.org/article.aspx?articleid=2177479[↩][↩]
- Jacobson GP, Shepard NT. Balance Function Assessment and Management. 2nd ed. California: Plural Publishing, Inc; 2016.[↩][↩]
- Ernst A, Basta D, Seidl RO, Todt I, Scherer H, Clarke A. Management of posttraumatic vertigo. Otolaryngol Head Neck Surg. 2005 Apr;132(4):554-8. doi: 10.1016/j.otohns.2004.09.034[↩]
- Dix MR, Hallpike CS. The pathology, symptomatology and diagnosis of certain common disorders of the vestibular system. Ann Otol Rhinol Laryngol. 1952;61:987–1016. doi: 10.1177/000348945206100403[↩]
- Dorigueto RS, Mazzetti KR, Gabilan YP, Ganança FF. Benign paroxysmal positional vertigo recurrence and persistence. Braz J Otorhinolaryngol. 2009 Jul-Aug;75(4):565-72. doi: 10.1016/s1808-8694(15)30497-3[↩]
- Goldberg JM, Cullen KE. Vestibular control of the head: possible functions of the vestibulocollic reflex. Exp Brain Res. 2011 May;210(3-4):331-45. doi: 10.1007/s00221-011-2611-5[↩]
- Pozzo T, Berthoz A, Papvov C. In: Taguchi K, Igarashi M, Mori S, editors. The effect of gravity on the coordination between posture and movement; Vestibular and Neural Front, Proceedings of the 12th International Symposium on Posture and Gait; Amsterdam: Elsevier; 1994. pp. 589–602.[↩]
- Bronstein AM, Hood JD. The cervico-ocular reflex in normal subjects and patients with absent vestibular function. Brain Res. 1986 May 14;373(1-2):399-408. doi: 10.1016/0006-8993(86)90355-0[↩]
- Pompeiano O. The tonic neck reflex: supraspinal control. In: Peterson BW, Richmond FJ, editors. Control of Head Movement. New York: Oxford University Press; 1988. pp. 108–119.[↩]
- Peterson BW, Goldberg J, Bilotto G, Fuller JH. Cervicocollic reflex: its dynamic properties and interaction with vestibular reflexes. J Neurophysiol. 1985 Jul;54(1):90-109. doi: 10.1152/jn.1985.54.1.90[↩]
- Carleton SC, Carpenter MB. Afferent and efferent connections of the medial, inferior and lateral vestibular nuclei in the cat and monkey. Brain Res. 1983 Nov 14;278(1-2):29-51. doi: 10.1016/0006-8993(83)90223-8[↩][↩]
- Brandt, T. and Dieterich, M. (1994), Vestibular syndromes in the roll plane: Topographic diagnosis from brainstem to cortex. Ann Neurol., 36: 337-347. https://doi.org/10.1002/ana.410360304[↩]
- Brandt T, Dieterich M. Vestibular syndromes in the roll plane: topographic diagnosis from brainstem to cortex. Ann Neurol. 1994 Sep;36(3):337-47. doi: 10.1002/ana.410360304[↩][↩]
- Purves D, Augustine GJ, Fitzpatrick D, et al., editors. Central vestibular pathways: eye, head, and body reflexes Neuroscience. 2nd ed. Sunderland, MA: Sinauer Associates; 2001.[↩]
- Strupp M, Brandt T. Diagnosis and Treatment of Vertigo and Dizziness. Deutsches Ärzteblatt International. 2008;105(10):173-180. doi:10.3238/arztebl.2008.0173. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2696792/[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Epidemiology of vertigo. Neuhauser HK. Curr Opin Neurol. 2007 Feb; 20(1):40-6. https://www.ncbi.nlm.nih.gov/pubmed/17215687/[↩][↩][↩][↩]
- Dizziness in primary care. Results from the National Ambulatory Medical Care Survey. Sloane PD. J Fam Pract. 1989 Jul; 29(1):33-8. https://www.ncbi.nlm.nih.gov/pubmed/2738548/[↩]
- Brandt T, Dieterich M, Strupp M. Vertigo – Leitsymptom Schwindel. Darmstadt: Steinkopff; 2003.[↩][↩][↩]
- Kerber KA. Vertigo and Dizziness in the Emergency Department. Emergency medicine clinics of North America. 2009;27(1):39-viii. doi:10.1016/j.emc.2008.09.002. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2676794/[↩][↩]
- Clinical practice. Vestibular neuritis. Baloh RW. N Engl J Med. 2003 Mar 13; 348(11):1027-32. https://www.ncbi.nlm.nih.gov/pubmed/12637613/[↩]
- Norrving B, Magnusson M, Holtas S. Isolated acute vertigo in the elderly; vestibular or vascular disease? Acta Neurol Scand. 1995 Jan;91(1):43–48. https://www.ncbi.nlm.nih.gov/pubmed/7732773[↩][↩]
- Lee H, Cho YW. A case of isolated nodulus infarction presenting as a vestibular neuritis. J Neurol Sci. 2004 Jun 15;221(12):117–119. https://www.ncbi.nlm.nih.gov/pubmed/15178226[↩]
- Lee H, Sohn SI, Cho YW, et al. Cerebellar infarction presenting isolated vertigo: frequency and vascular topographical patterns. Neurology. 2006 Oct 10;67(7):1178–1183. https://www.ncbi.nlm.nih.gov/pubmed/17030749[↩]
- Thompson TL, Amedee R. Vertigo: A Review of Common Peripheral and Central Vestibular Disorders. The Ochsner Journal. 2009;9(1):20-26. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3096243/[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Kleindorfer DO, Miller R, Moomaw CJ, et al. Designing a message for public education regarding stroke: does FAST capture enough stroke? Stroke. 2007 Oct;38(10):2864–2868. http://stroke.ahajournals.org/content/38/10/2864.long[↩]
- Kerber KA, Brown DL, Lisabeth LD, Smith MA, Morgenstern LB. Stroke Among Patients With Dizziness, Vertigo, and Imbalance in the Emergency Department: A Population-Based Study. Stroke; a journal of cerebral circulation. 2006;37(10):2484-2487. doi:10.1161/01.STR.0000240329.48263.0d. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1779945/[↩]
- Chalela JA, Kidwell CS, Nentwich LM, et al. Magnetic resonance imaging and computed tomography in emergency assessment of patients with suspected acute stroke: a prospective comparison. Lancet. 2007;369(9558):293-298. doi:10.1016/S0140-6736(07)60151-2. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1859855/[↩]
- Wasay M, Dubey N, Bakshi R. Dizziness and yield of emergency head CT scan: Is it cost effective? Emergency Medicine Journal : EMJ. 2005;22(4):312. doi:10.1136/emj.2003.012765. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1726733/pdf/v022p00312.pdf[↩]
- Oppenheim C, Stanescu R, Dormont D, et al. False-negative diffusion-weighted MR findings in acute ischemic stroke. AJNR Am J Neuroradiol. 2000 Sep;21(8):1434–1440. http://www.ajnr.org/content/21/8/1434.long[↩]
- Methylprednisolone, valacyclovir, or the combination for vestibular neuritis. Strupp M, Zingler VC, Arbusow V, Niklas D, Maag KP, Dieterich M, Bense S, Theil D, Jahn K, Brandt T. N Engl J Med. 2004 Jul 22; 351(4):354-61. http://www.nejm.org/doi/full/10.1056/NEJMoa033280[↩]
- Vestibular exercises improve central vestibulospinal compensation after vestibular neuritis. Strupp M, Arbusow V, Maag KP, Gall C, Brandt T. Neurology. 1998 Sep; 51(3):838-44. https://www.ncbi.nlm.nih.gov/pubmed/9748036/[↩]
- What inner ear diseases cause benign paroxysmal positional vertigo ? Karlberg M, Hall K, Quickert N, Hinson J, Halmagyi GM. Acta Otolaryngol. 2000 Mar; 120(3):380-5. https://www.ncbi.nlm.nih.gov/pubmed/10894413/[↩]
- Current view of the mechanism of benign paroxysmal positioning vertigo: cupulolithiasis or canalolithiasis ? Brandt T, Steddin S. J Vestib Res. 1993 Winter; 3(4):373-82. https://www.ncbi.nlm.nih.gov/pubmed/8275271/[↩]
- Curing the BPPV with a liberatory maneuver. Semont A, Freyss G, Vitte E. Adv Otorhinolaryngol. 1988; 42():290-3. https://www.ncbi.nlm.nih.gov/pubmed/3213745/[↩]
- Strupp M, Cnyrim C, Brandt T. Vertigo and dizziness: Treatment of benign paroxysmal positioning vertigo, vestibular neuritis and Menère’s disease. In: Candelise L, editor. Evidence-based Neurology – management of neurological disorders. Oxford: Blackwell Publishing; 2007. pp. 59–69.[↩][↩]
- Benign paroxysmal positioning vertigo: a long-term follow-up (6-17 years) of 125 patients. Brandt T, Huppert D, Hecht J, Karch C, Strupp M. Acta Otolaryngol. 2006 Feb; 126(2):160-3. https://www.ncbi.nlm.nih.gov/pubmed/16428193/[↩]
- Halmagyi GM, Curthoys IS. A clinical sign of canal paresis. Arch Neurol. 1988;45:737–739. https://www.ncbi.nlm.nih.gov/pubmed/3390028[↩]
- Prevalence of HSV-1 LAT in human trigeminal, geniculate, and vestibular ganglia and its implication for cranial nerve syndromes. Theil D, Arbusow V, Derfuss T, Strupp M, Pfeiffer M, Mascolo A, Brandt T. Brain Pathol. 2001 Oct; 11(4):408-13. https://www.ncbi.nlm.nih.gov/pubmed/11556685/[↩]
- Strupp M, Zingler VC, Arbusow V, et al. Methylprednisolone, valacyclovir, or the combination for vestibular neuritis. N Engl J Med. 2004;351:354–361. http://www.nejm.org/doi/full/10.1056/NEJMoa033280[↩]
- Strupp M, Arbusow V, Maag KP, Gall C, Brandt T. Vestibular exercises improve central vestibulospinal compensation after vestibular neuritis. Neurology. 1998;51:838–844. https://www.ncbi.nlm.nih.gov/pubmed/9748036[↩]
- Ménière’s disease. Minor LB, Schessel DA, Carey JP. Curr Opin Neurol. 2004 Feb; 17(1):9-16. https://www.ncbi.nlm.nih.gov/pubmed/15090872/[↩][↩]
- Ménière’s disease: a long-term follow-up study of bilateral hearing levels. Takumida M, Kakigi A, Takeda T, Anniko M. Acta Otolaryngol. 2006 Sep; 126(9):921-5. https://www.ncbi.nlm.nih.gov/pubmed/16864488/[↩]
- Strupp M, Huppert D, Frenzel C, Wagner J, Zingler VC, Mansmann U, Brandt T. Long-term prophylactic treatment of attacks of vertigo in Menière’s disease-comparison of a high with a low dosage of betahistine in an open trial. Acta Otolaryngol. 2008 May;128(5):520-4. doi: 10.1080/00016480701724912. https://www.ncbi.nlm.nih.gov/pubmed/18421605[↩]
- Epidemiology of acoustic neuromas. Tos M, Thomsen J. J Laryngol Otol. 1984 Jul; 98(7):685-92. https://www.ncbi.nlm.nih.gov/pubmed/6747450/[↩]
- Khrais T., Romano G., Sanna M. Nerve origin of vestibular schwannoma: a prospective study. J Laryngol Otol. 2008;122:128–131. https://www.ncbi.nlm.nih.gov/pubmed/18039415[↩]
- Fife TD. Episodic vertigo. In: Rakel RE, ed. Conn’s Current therapy, 1999: latest approved methods of treatment for the practicing physician. 51st ed. Philadelphia: Saunders, 1999:923–30.[↩]
- Flake ZA, Scalley RD, Bailey AG. Practical selection of antiemetics. Am Fam Physician. 2004;69:1169–74.[↩]
- Kim H. Y., Chung C. S., Moon S. Y., et al. Complete nonvisualization of basilar artery on MR angiography in patients with vertebrobasilar ischemic stroke: favorable outcome factors. Cerebrovasc Dis. 2004;18:269–276. https://www.ncbi.nlm.nih.gov/pubmed/15331872[↩]
- Grad A., Baloh R. W. Vertigo of vascular origin. Clinical and electronystagmographic features in 84 cases. Arch Neurol. 1989;46:281–284. https://www.ncbi.nlm.nih.gov/pubmed/2919982[↩]
- Quigley EM, Hasler WL, Parkman HP. AGA technical review on nausea and vomiting. Gastroenterology. 2001;120:263–86.[↩]
- Baloh RW. Vertigo in older people. Curr Treat Options Neurol. 2000;2:81–9.[↩]
- Levy EI, Hanel RA, Bendok BR, Boulos AS, Hartney ML, Guterman LR, et al. Staged stent-assisted angioplasty for symptomatic intracranial vertebrobasilar artery stenosis. J Neurosurg. 2002;97:1294–301.[↩]
- Norrving B, Magnusson M, Holtas S. Isolated acute vertigo in the elderly; vestibular or vascular disease?. Acta Neurol Scand. 1995;91:43–8.[↩]
- Downbeat nystagmus: aetiology and comorbidity in 117 patients. Wagner JN, Glaser M, Brandt T, Strupp M. J Neurol Neurosurg Psychiatry. 2008 Jun; 79(6):672-7. https://www.ncbi.nlm.nih.gov/pubmed/17872983/[↩][↩]
- Detection of floccular hypometabolism in downbeat nystagmus by fMRI. Kalla R, Deutschlander A, Hufner K, Stephan T, Jahn K, Glasauer S, Brandt T, Strupp M. Neurology. 2006 Jan 24; 66(2):281-3. https://www.ncbi.nlm.nih.gov/pubmed/16434677/[↩]
- Treatment of downbeat nystagmus with 3,4-diaminopyridine: a placebo-controlled study. Strupp M, Schüler O, Krafczyk S, Jahn K, Schautzer F, Büttner U, Brandt T. Neurology. 2003 Jul 22; 61(2):165-70. https://www.ncbi.nlm.nih.gov/pubmed/12874393/[↩]
- 4-aminopyridine restores vertical and horizontal neural integrator function in downbeat nystagmus. Kalla R, Glasauer S, Büttner U, Brandt T, Strupp M. Brain. 2007 Sep; 130(Pt 9):2441-51. https://www.ncbi.nlm.nih.gov/pubmed/17664175/[↩]
- 4-aminopyridine restores visual ocular motor function in upbeat nystagmus. Glasauer S, Kalla R, Büttner U, Strupp M, Brandt T. J Neurol Neurosurg Psychiatry. 2005 Mar; 76(3):451-3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1739571/pdf/v076p00451.pdf[↩]
- Treatment of episodic ataxia type 2 with the potassium channel blocker 4-aminopyridine. Strupp M, Kalla R, Dichgans M, Freilinger T, Glasauer S, Brandt T. Neurology. 2004 May 11; 62(9):1623-5. https://www.ncbi.nlm.nih.gov/pubmed/15136697/[↩]
- The interrelations of migraine, vertigo, and migrainous vertigo. Neuhauser H, Leopold M, von Brevern M, Arnold G, Lempert T. Neurology. 2001 Feb 27; 56(4):436-41. https://www.ncbi.nlm.nih.gov/pubmed/11222783/[↩]
- Episodic vertigo related to migraine (90 cases): vestibular migraine ? Dieterich M, Brandt T. J Neurol. 1999 Oct; 246(10):883-92. https://www.ncbi.nlm.nih.gov/pubmed/10552234/[↩]
- Phobic postural vertigo–a long-term follow-up (5 to 15 years) of 106 patients. Huppert D, Strupp M, Rettinger N, Hecht J, Brandt T. J Neurol. 2005 May; 252(5):564-9. https://www.ncbi.nlm.nih.gov/pubmed/15742115/[↩]