Contents
- What is leiomyosarcoma
- Leiomyosarcoma Classification
- Uterine leiomyosarcoma
- Leiomyosarcoma causes
- Leiomyosarcoma prognosis
- Leiomyosarcoma Staging
- Leiomyosarcoma survival rate
- Leiomyosarcoma symptoms
- Leiomyosarcoma diagnosis
- Leiomyosarcoma treatment
What is leiomyosarcoma
A leiomyosarcoma is an aggressive soft tissue sarcoma derived from smooth muscle cells typically of uterine, gastrointestinal or soft tissue origin. Geographically, leiomyosarcoma is found worldwide. Leiomyosarcoma is rare, accounting for between 10 and 20% of soft tissue sarcomas. Soft tissue sarcomas are uncommon accounting for 1-2% of all cancers. Leiomyosarcoma occurs with highest incidence in adulthood, with sex incidence being more common in women typically occurring in the 5th and 6th decades of life. The majority of leiomyosarcomas originate in the muscle layers of the gastrointestinal tract or uterus 1).
Leiomyosarcoma is classified as a soft tissue sarcoma. Sarcomas are malignant tumors that arise from the connective tissue, which connects, supports and surrounds various structures and organs in the body. Soft tissue includes fat, muscle, nerves, tendons, and blood and lymph vessels. The exact cause of leiomyosarcoma, including uterine leiomyosarcoma, is unknown.
Leiomyosarcomas affect both males and females. According to the American Cancer Society, at least 15,000 new cases of soft tissue sarcoma occur each year in the U.S. Soft tissue sarcomas affect men and women equally and occur more often in adults than children or adolescents. Soft tissue sarcomas account for 1 percent of all adult cancers in the U.S. According to one estimate, leiomyosarcomas account for 7-11 percent of all cases of soft tissue sarcomas.
Muscle is the tissue responsible for body movements and the changes in the shape and size of internal organs. It consists of parallel arrangements of highly specialized cells containing contractile elements – chiefly actin and myosin filaments.
There are 2 main types of muscle (based on their appearance under the light microscope): smooth muscle and striated muscle. Smooth muscle is a component of many tissues and organs. Smooth muscle is the muscle that is found in the gastrointestinal, the uterus, respiratory and genitourinary tracts, the blood vessels, the iris and ciliary body of the eye, the scrotum, and in the hair follicles. Smooth muscle is specialized for a slow and prolonged contraction, and may contract in a wave-like manner (peristalsis) or may remain contracted for an extended period of time without becoming fatigued. Smooth muscle contraction is regulated by the autonomic nervous system. As a result, leiomyosarcoma can arise at almost any anatomic site in the human body. In women, approximately one third of leiomyosarcomas originate in the gastrointestinal tract, particularly the small bowel and colon and another one third are found in the uterus.
Leiomyosarcoma also can affect large blood vessels, particularly the vein that carries blood from the lower part of the body back to the heart (inferior vena cava) and the artery that carries blood from the heart to the lungs (pulmonary artery). Leiomyosarcoma tumor can also occur in other blood vessels such as the great saphenous vein, femoral vein, iliac vein, popliteal vein, and renal vein 2). In fact leiomyosarcoma is the most common primary malignancy of the inferior vena cava (IVC), and represents approximately 10% of primary retroperitoneal sarcomas 3).
Leiomyosarcoma cancer spreads by continuous extension into adjacent structures. Hematogenous (blood stream) dissemination with distant metastases to the lungs and liver can also occur.
Most forms of leiomyosarcoma are aggressive tumors that may spread (metastasize) to other areas of the body such as the lungs or liver, potentially causing life-threatening complications. Leiomyosarcoma has a high risk of recurring after treatment, if not diagnosed early.
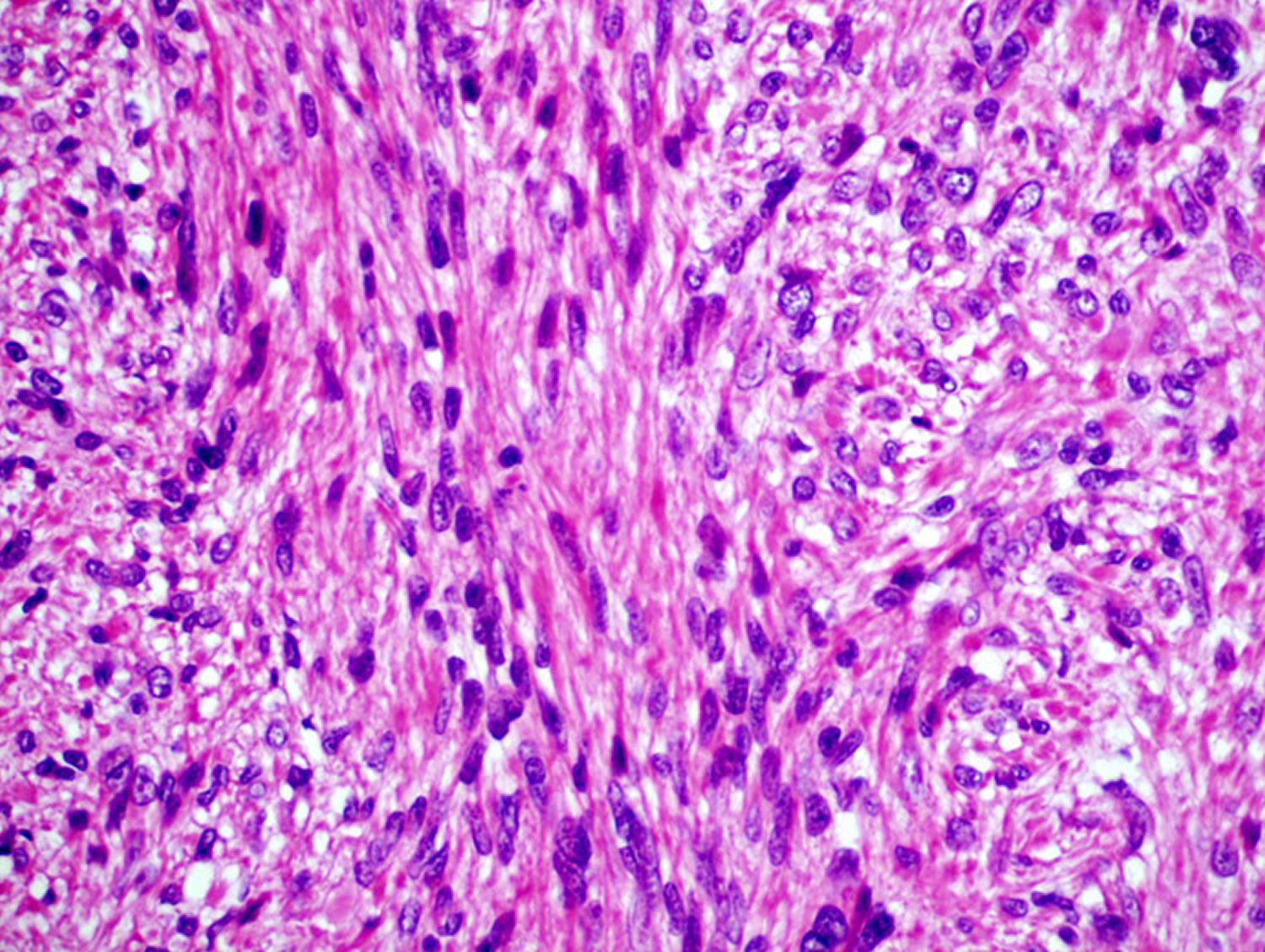
Leiomyosarcoma diagnosis should be performed in highly specialized centers and is established by the presence of morphologically typical spindle cells for smooth muscle differentiation and the positivity of a smooth muscle actin and other muscle markers on tumor cells, such as desmin and h-caldesmon 4). Furthermore, the specific characteristic of primary bone leiomyosarcoma is the absence of either osteoid or chondroid matrix 5). The molecular pathogenesis and biological heterogeneity of leiomyosarcoma have not yet been clarified.
Are there genetic tests available to assess a persons risk for leiomyosarcoma?
No. Doctors are unaware of genetic tests to help inform family members about leiomyosarcoma risks. While leiomyosarcomas are due to genetic changes in the tumor cells, these genetic changes are not inherited. They occur only in the cancer tissue. It is very rare for leiomyosarcoma to occur in multiple members of the same family.
That being said, there are a few genetic syndromes that can run in families, where leiomyosarcoma may occur as part of the syndrome. Having a single relative with a leiomyosarcoma alone does not typically raise suspicion for these syndromes, however a doctor may suspect these syndromes in families with a certain combination of cancers and other symptoms. These syndromes include, hereditary retinoblastoma, Li-Fraumeni syndrome, Neurofibromatosis type 1, tuberous sclerosis, nevoid basal cell carcinoma syndrome, Gardner syndrome, and Werner syndrome.
Leiomyosarcoma Classification
Histologically, soft tissue leiomyosarcomas that arise in different anatomic locations are similar. However, based on the location of the tumor, prognosis and possible treatments differ. For this reason leiomyosarcoma of soft tissues is divided into four groups. Furthermore there are sporadic case reports of primary leiomyosarcoma of bone, a clinically distinct entity.
- Leiomyosarcoma of Soft Tissue Retroperitoneal Somatic soft tissue
- Leiomyosarcoma of Cutaneous Origin
- Leiomyosarcoma of Vascular Origin (large vessel)
- Leiomyosarcoma in the Immunocompromised Host
- Leiomyosarcoma of Bone
Leiomyosarcoma of Soft Tissue
Immunohistochemical analysis suggests that the cell line of origin of leiomyosarcoma is the smooth muscle cell. The most common site of leiomyosarcoma of soft tissue is the retroperitoneum, accounting for 50% of all cases 6). Smooth muscle sarcomas arising from the abdominal viscera or uterus are considered to be distinct disease entities. Other sites of involvement include the deep soft tissues of the extremities and are referred to as leiomyosarcoma of somatic soft tissue 7). Soft tissue leiomyosarcoma was at one time believed to arise from leiomyomas, however, this is now thought to be an extremely rare occurrence. Most malignant leiomyosarcomas arise independently, and are not associated with benign tumors. Histologic studies of somatic soft tissue leiomyosarcomas have shown that many, if not all, of these tumors arise directly from the smooth muscle cells lining small blood vessels.
When the retroperitoneum is involved, presenting symptoms are usually vague abdominal discomfort, an abdominal mass and weight loss. Peripherally located masses present as an enlarging mass, often painless, with few constitutional signs. Due to the deep inaccessible location and large volume of the abdominal cavity, leiomyosarcomas of the retroperitoneum tend to be significantly larger than those of the extremities at presentation. Retroperitoneal leiomyosarcoma is an aggressive disease that is often not amenable to complete surgical resection.
Leiomyosarcoma of Cutaneous Origin
Leiomyosarcoma can arise within the dermis. When this occurs it is referred to as cutaneous leiomyosarcoma. Unlike other forms of leiomyosarcoma, men are affected more than women at a ratio of 2:1 8). These lesions are typically small when first diagnosed (1-2 cm), and prognosis is generally good 9). When leiomyosarcoma develops within the dermis itself it is thought to be derived from the pilar arrecti.20 Tumors that develop within subcutaneous tissue arise from small or microscopic vessels and should be considered leiomyosarcoma of somatic soft tissue. The behavior of these tumors is more consistent with that of deeper tumors than intradermal tumors. When the lesion is confined to the dermis, metastasis typically does not occur 10). Deeper lesions can metastasize in up to 30-40% of cases, usually hematogenously to the lungs 11). Treatment consists of wide resection, and is often curative when the lesion is initially confined to the dermis, regardless of histologic grade.
Leiomyosarcoma of Vascular Origin
Leiomyosarcoma rarely arises directly from major blood vessels, however, when it does, it is termed leiomyosarcoma of vascular origin. There have been only a few hundred published reports of leiomyosarcoma of vascular origin. In one review of 86 cases, leiomyosarcoma of vascular origin was shown to have a propensity for lower pressure systems. Most commonly affected were the larger veins (68 cases), specifically the inferior vena cava (in 33 cases), and less commonly the pulmonary artery (10 cases) and rarely peripheral arteries (8 cases) 12).
If the tumor develops in the inferior vena cava in the supra-hepatic segment, Budd-Chiari syndrome develops: hepatomegaly, jaundice, and ascites. These tumors are usually not surgically resectable. Tumors that arise in the inferior vena cava below the liver present with lower extremity edema and vague abdominal pain. Symptoms are defined by the anatomic location of the lesion, and the local vascular physiology and drainage patterns.
Arterial leiomyosarcoma usually affects the pulmonary artery. Patients will typically complain of dyspnea and chest discomfort, relating to the arterial obstruction. Symptoms are related to the vascular distribution of the affected artery and the presence or absence of collateral blood flow.
Leiomyosarcoma in the Immunocompromised Host
Since the 1970s there have been a number of cases of leiomyosarcoma reported in immunocompromised patients having undergone transplantation and treated with immunosuppressive regimens 13). More recently, there have been further case reports involving people infected with the HIV/AIDS virus 14). There appears to be a relationship between these immunocompromised patients and super-infection with Epstein-Barr virus (EBV). Case reports of synchronous multiple leiomyosarcoma have been published where clonal analysis have shown that the individual tumors arose independently from each other 15). It is not known what interaction exists between immuno-incompentence and Epstein-Barr virus (EBV) infection that predisposes to leiomyosarcoma.
Leiomyosarcoma of Bone
Primary leiomyosarcoma of bone is extremely rare. There have been approximately 90 cases reported since initially described in 1965 16). Many cases that are thought to represent primary disease of bone, after further investigation, actually represent metastatic disease from another site or bony invasion from a neighboring soft tissue lesion. Most cases of leiomyosarcoma of bone reported so far have been in the metaphysis of long bones. These lesions are thought to arise from the smooth muscle cells lining the intraosseous vessels or from pluripotent mesenchymal cells. The histology is the same as leiomyosarcoma of soft tissue. These tumors have an equal or slightly male-predominant gender distribution. The radiographic appearance of these tumors is typically a radiolucent lesion in the metaphysis of a long bone, although the tumor has been described in other locations as well. A permeative appearance is characteristic 17). There are no specific radiographic features that can diagnose leiomyosarcoma by radiography alone.
Uterine leiomyosarcoma
Uterine leiomyosarcoma is a rare cancer that starts in the muscular wall of the uterus (the myometrium). Uterine leiomyosarcoma is by far the most common uterine sarcoma type accounting for 25–36% of uterine sarcoma and 1% of all malignancies 18). Uterine leiomyosarcoma can grow and spread quickly and has a strong metastatic potential to distant sites, because of its aggressiveness and propensity for hematogenous spread. Uterine leiomyosarcoma has a poor prognosis due to a high metastatic recurrence rate 19).
Doctors don’t know exactly what causes most uterine leiomyosarcomas, but certain risk factors have been identified. Research is helping to learn more about this rare disease.
Uterine leiomyosarcoma is an extremely rare form of cancer, estimated to occur in 6 out of every 1,000,000 women in the United States each year. The average age at diagnosis is 51. Uterine leiomyosarcomas account for 1-2 percent of all malignant tumors of the uterus.
Uterine leiomysarcomas most commonly arise de novo; however, a minority (5%) may be associated with prior irradiation. The peak incidence occurs in the 30–40 age range and reaches a plateau in the middle age. Uterine leiomyosarcoma usually presents with features of vaginal bleeding (77–95%), pelvic pain (33%), uterine enlargement or a palpable pelvic mass (20–50%) 20).
The commonly reported sites of metastasis from leiomysarcoma are the lung, kidney and liver 21). Spread to the thyroid, brain, bone, skeletal muscle, heart, parotid gland and the oral cavity have also been reported 22). Uterine leiomyosarcoma should be distinguished from benign uterine metastasizing leiomyoma which is diagnosed several years after myomectomy or hysterectomy with most commonly radiographic appearance of slow-growing solitary or multiple lung nodules.
Currently, the treatment of uterine leiomyosarcoma is mostly by surgery with some adjuvant therapies, such as cytotoxic chemotherapy and radiotherapy 23).
Risk Factors for uterine leiomyosarcoma
A risk factor is anything that affects your chance of getting a disease such as cancer. Different cancers have different risk factors. For example, exposing skin to strong sunlight is a risk factor for skin cancer. Smoking is a risk factor for many cancers.
There are different kinds of risk factors. Some, such as your age or race, can’t be changed. Others may be related to personal choices such as smoking, drinking, or diet. Some factors influence risk more than others. But risk factors don’t tell us everything. Having a risk factor, or even several, does not mean that a person will get the disease. Also, not having any risk factors doesn’t mean that you won’t get the disease.
These factors are known to change a woman’s risk of developing a uterine sarcoma.
Pelvic radiation therapy
High-energy (ionizing) radiation used to treat some cancers can damage cells’ DNA, sometimes increasing the risk of developing a second type of cancer. If you’ve had pelvic radiation, your risk for developing uterine sarcoma is increased. These cancers usually are diagnosed 5 to 25 years after you’ve been exposed to the radiation.
Race
Uterine sarcomas are about twice as common in African-American women as they are in white or Asian women. The reason for this is unknown.
RB gene changes
Women who have had a type of eye cancer called retinoblastoma that was caused by being born with an abnormal copy of the RB gene have an increased risk of uterine leiomyosarcomas.
Remember, that these factors increase the risk for developing some uterine sarcomas, but they may not always cause the disease.
Uterine leiomyosarcoma prevention
Most cases of uterine leiomyosarcoma cannot be prevented. Although pelvic radiation increases the risk of developing a uterine sarcoma, the benefit of pelvic radiation in treating other cancers far outweighs the risk of developing a rare cancer such as uterine sarcoma many years later.
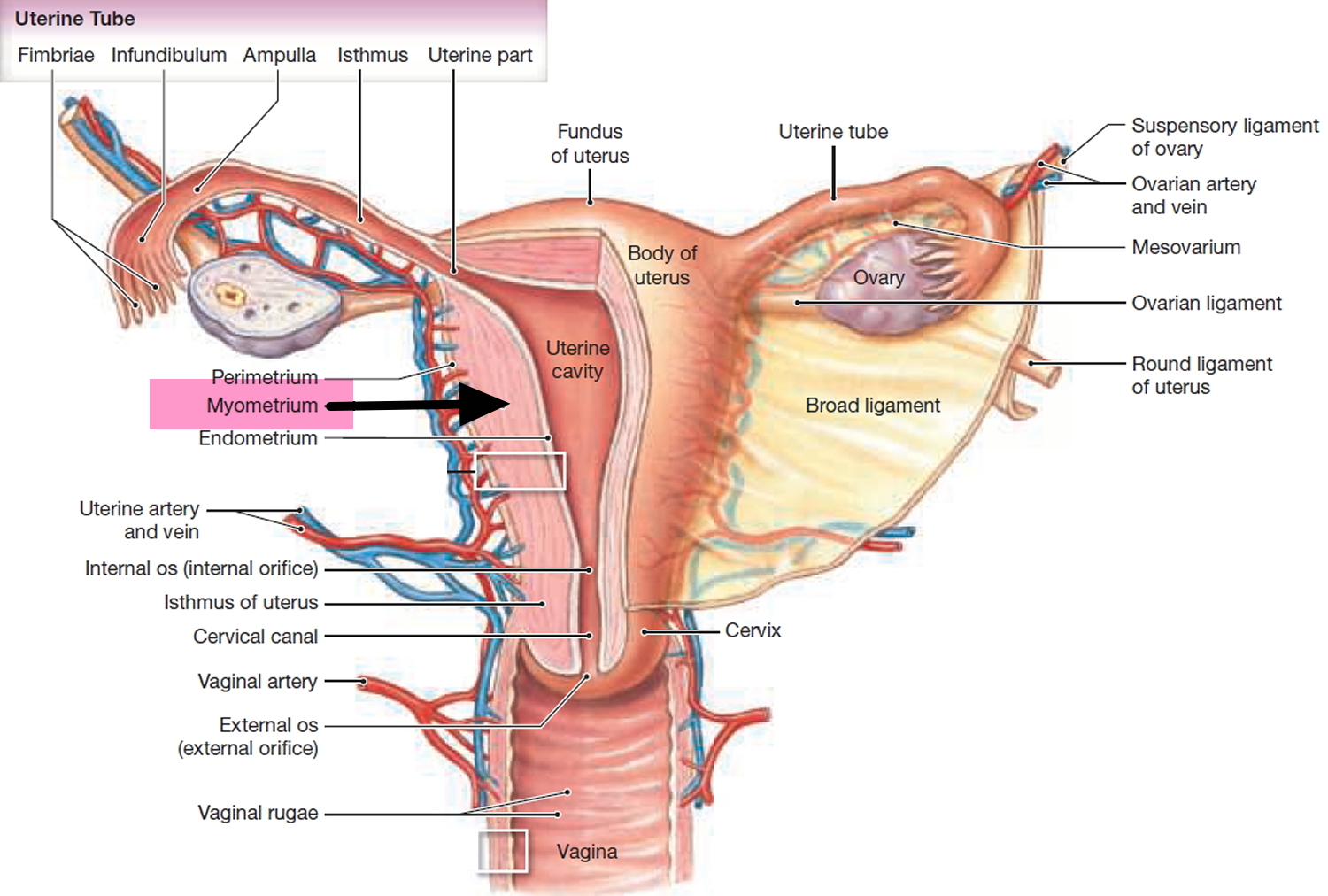
About the uterus
The uterus is a hollow organ, usually about the size and shape of a medium-sized pear:
- The lower end of the uterus, which extends into the vagina, is the cervix.
- The upper part of the uterus is the body, also known as the corpus.
The body of the uterus has 3 layers:
- The inner layer or lining is the endometrium.
- The serosa is the layer of tissue coating the outside of the uterus.
- In the middle is a thick layer of muscle known as the myometrium. This muscle layer is needed to push a baby out during childbirth.
Figure 1. Uterus
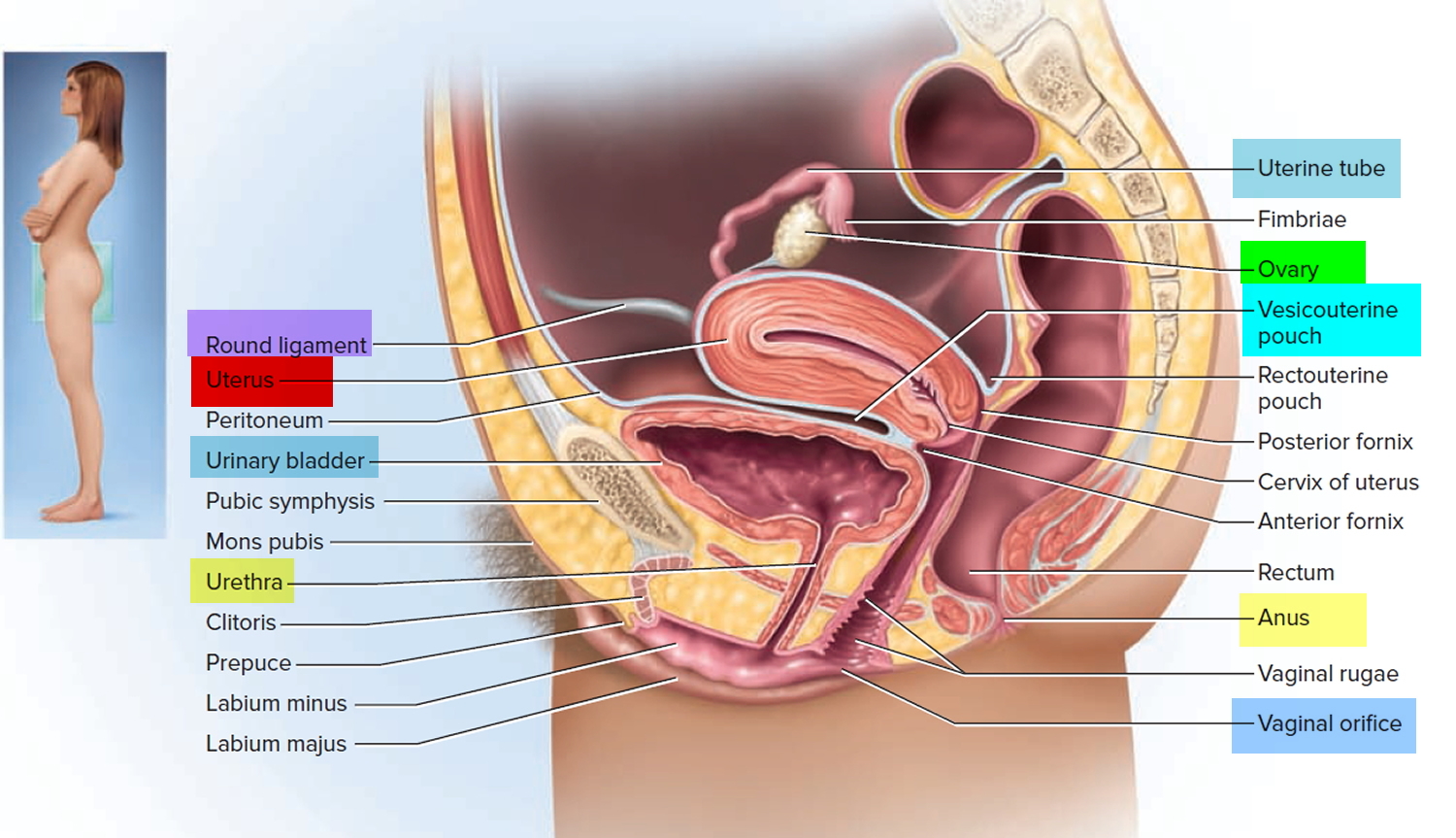
Figure 2. Uterus location
Uterine Leiomyosarcoma symptoms
In most cases, the possibility of uterine sarcoma is suggested by certain symptoms. These symptoms don’t always mean that a woman has uterine sarcoma. In fact, they are more often caused by something else, such as non-cancerous changes in the uterus (like fibroids), pre-cancerous overgrowth of the endometrium, or endometrial carcinoma. Still, if you’re having these problems, see a doctor to find the cause and get any needed treatment.
Abnormal bleeding or spotting
About 85% of patients diagnosed with uterine sarcomas have irregular vaginal bleeding (between periods) or bleeding after menopause. This symptom is more often caused by something other than cancer, but it’s important to have any irregular bleeding checked right away.
If you’ve gone through menopause, any vaginal bleeding or spotting is abnormal, and it should be reported to your health care professional right away.
Vaginal discharge
About 10% of women with uterine sarcomas have a vaginal discharge that does not have any visible blood. A discharge is most often a sign of infection or another non-cancer condition, but it also can be a sign of cancer. Any abnormal discharge should be checked by a health care professional.
Pelvic pain and/or a mass
When they’re first diagnosed, about 10% of women with uterine sarcomas have pelvic pain and/or a mass (tumor) that can be felt. You or your doctor may be able to feel the mass in your uterus, or you might have a feeling of fullness in your belly and/or pelvis.
Uterine Leiomyosarcoma diagnosis
Many uterine leiomyosarcomas are diagnosed during or after surgery for what’s thought to be benign fibroid tumors.
Some are diagnosed because of symptoms. If you have symptoms of uterine cancer, the first step is to see your doctor.
Consultation, medical history, and physical exam
Your doctor will ask you about your personal and family medical history, examine you, and might order some tests. You also will be asked about any symptoms, risk factors, and other health problems. A general physical and a pelvic exam will be done. An ultrasound may be used to look at the inside of your uterus.
If your doctor suspects cancer, you may be referred to a gynecologist or a doctor specializing in cancers of the female reproductive system (called a gynecologic oncologist).
Sampling and testing endometrial tissue
To find the cause of abnormal uterine bleeding, a small piece of tissue (a sample) will be taken from the lining of the uterus and looked at with a microscope. The tissue can be removed by endometrial biopsy or by dilation and curettage (D&C). Often a hysteroscopy is done with the D&C..
These procedures let the doctor see if the bleeding is caused by a endometrial overgrowth that’s not cancer (hyperplasia), endometrial carcinoma, uterine leiomyosarcoma, or some other problem. The tests will find many endometrial stromal leiomyosarcomas and undifferentiated leiomyosarcomas, but less than half of leiomyosarcoma (abbreviated LMSs). These tests don’t find all leiomyosarcomas because these cancers start in the muscle layer of the wall of the uterus. To be found by an endometrial biopsy or D&C, they need to have spread from the middle (muscle) layer to the inner lining of the uterus. In most cases, the only way to diagnose a leiomyosarcoma by removing it with surgery.
Endometrial biopsy
In this procedure, a very thin, flexible tube is put into the uterus through the cervix. Then, using suction, a small amount of the uterine lining (endometrium) is taken out through the tube. Suctioning takes about a minute or less. The discomfort is a lot like severe menstrual cramps and can be helped by taking a nonsteroidal anti-inflammatory drug like ibuprofen an hour before the biopsy. This procedure is usually done in the doctor’s office.
Hysteroscopy
This procedure allows doctors to look inside the uterus. A tiny telescope is put into the uterus through the cervix. To get a better view, the uterus is then expanded by filling it with salt water (saline). This lets the doctor see and take out anything abnormal, such as a cancer or a polyp. This procedure is usually done with the patient awake, using local anesthesia (numbing medicine). But if a polyp or mass has to be removed, general or regional anesthesia is sometimes used. (General anesthesia means you are given drugs that put you into a deep sleep and keep you from feeling pain. Regional anesthesia is a nerve block that numbs a larger area of the body).
Dilation and curettage
If the results of the endometrial biopsy are not clear (meaning they can’t tell for sure if cancer is present), a procedure called dilation and curettage (D&C) must be done. A D&C is usually done in the outpatient surgery area of a clinic or hospital. It’s done while the woman is under general or regional anesthesia or conscious sedation (medicine is given into a vein to make her drowsy). It takes about an hour. In a D&C, the cervix is dilated (opened) and a special surgical tool is used to scrape the endometrial tissue from inside the uterus. A hysteroscopy may be done as well. Some women have mild to moderate cramping and discomfort after this procedure.
Testing endometrial tissue
Any tissue samples taken out are looked at under a microscope to see if cancer is present. If cancer is found, the lab report will say if it’s a carcinoma or leiomyosarcoma, what type it is, and its grade.
A tumor’s grade is based on how much it looks like normal tissue under the microscope. If the tumor looks a lot like normal tissue, it’s called low grade. If it doesn’t at all look like normal tissue, it’s high grade. The rate at which the cancer cells appear to be growing is another important factor in grading a uterine leiomyosarcoma. High-grade leiomyosarcomas tend to grow and spread faster than low-grade leiomyosarcomas.
The tissue may also be tested to see if the cancer cells have estrogen receptors and progesterone receptors. These hormone receptors are found on many endometrial stromal sarcomas. Cancers with estrogen receptors on the cells are more likely to grow in response to estrogen, while those with progesterone receptors often have their growth decreased by progesterone. These cancers may stop growing (or even shrink) when treated with certain hormone drugs. Checking for these receptors helps predict which patients will benefit from treatment with these drugs.
Cystoscopy and proctoscopy
If a woman has signs or symptoms that suggest uterine leiomyosarcoma has spread to the bladder or rectum, the inside of these organs can be looked at through a lighted tube. These exams are called cystoscopy and proctoscopy, respectively. They are rarely done in the diagnosis and work-up of patients with uterine leiomyosarcoma.
Imaging tests
Transvaginal ultrasound
Ultrasound tests use sound waves to take pictures of parts of the body. For a transvaginal ultrasound, a probe that gives off sound waves is put into the vagina. The sound waves are used to create images of the uterus and other pelvic organs. These images can often show if there’s a tumor and if it affects the myometrium (muscular layer of the uterus).
For an ultrahysterosonogram or saline infusion sonogram, salt water (saline) is put into the uterus through a small tube before the transvaginal sonogram. This allows the doctor to see changes in the uterine lining more clearly.
Computed tomography
The CT scan is an x-ray test that produces detailed cross-sectional images of your body. Instead of taking one picture, like a standard x-ray, a CT scanner takes many pictures as it rotates around you. A computer then combines these pictures into an image of a slice of your body.
CT scans are rarely used to diagnose uterine cancer, but they may be helpful in seeing if the cancer has spread to other organs.
CT-guided needle biopsy: CT scans can also be used to guide a biopsy needle precisely into a suspected tumor. For this procedure, the patient remains on the CT scanning table while the doctor moves a biopsy needle through the skin and toward the tumor. CT scans are repeated until the needle is within the tumor. A fine needle biopsy sample or a larger core needle biopsy sample is then removed to be looked at with a microscope. This isn’t done to biopsy tumors in the uterus, but might be used to biopsy areas that look like metastasis (cancer spread).
Magnetic resonance imaging
MRI scans use radio waves and strong magnets instead of x-rays. The energy from the radio waves is absorbed and then released in a pattern formed by the type of tissue and by certain diseases. A computer translates the pattern of radio waves given off by the tissues into a very detailed image of parts of the body. MRI scans can help tell if a uterine tumor looks like cancer, but a biopsy is still needed to tell for sure.
MRI scans are also very helpful in looking for cancer that has spread to the brain and spinal cord.
Positron emission tomography scan
In a PET scan, radioactive glucose (sugar) is injected into the patient’s vein. Because many cancers use glucose much faster than normal tissues, the radioactivity tends to collect in the cancer. A scanner can then spot the radioactive deposits. This test can be helpful for spotting small collections of cancer cells that have spread beyond the uterus (matastasized).
Chest x-ray
A regular (plain) x-ray of the chest may be done to see if a uterine leiomyosarcoma has spread to the lungs and as part of the testing before surgery.
Uterine Leiomyosarcoma Staging
The 2 systems used for staging uterine sarcoma, the FIGO (International Federation of Gynecology and Obstetrics) system and the American Joint Committee on Cancer (AJCC) TNM staging system are basically the same.
They both stage (classify) this cancer based on 3 factors:
- The extent (size) of the tumor (T): How large is the cancer? Has the cancer grown out of the uterus into the pelvis or organs such as the bladder or rectum?
- The spread to nearby lymph nodes (N): Has the cancer spread to nearby lymph nodes?
- The spread (metastasis) to distant sites (M): Has the cancer spread to distant lymph nodes or organs?
Numbers or letters after T, N, and M provide more details about each of these factors. Higher numbers mean the cancer is more advanced. Once a person’s T, N, and M categories have been determined, this information is combined in a process called stage grouping to assign an overall stage.
The staging system in the table below uses the pathologic stage (also called the surgical stage). It is determined by examining tissue removed during an operation. Sometimes, if surgery is not possible right away, the cancer will be given a clinical stage instead. This is based on the results of a physical exam, biopsy, and imaging tests done before surgery. For more information see Cancer Staging.
The system described below is the most recent American Joint Committee on Cancer (AJCC) system. It went into effect January 2018. It is specific for staging two types of uterine sarcomas: leiomyosarcoma and endometrial stromal sarcoma.
Uterine sarcoma staging can be complex, so ask your doctor to explain it to you in a way you understand.
Table 1. Uterine sarcoma staging 24)
| Stage | Stage grouping | FIGO Stage | Stage description* |
| I | T1 N0 M0 | I | The cancer is growing in the uterus, but has not started growing outside the uterus. It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IA | T1a N0 M0 | IA | The cancer is only in the uterus and is no larger than 5 cm across (about 2 inches) (T1a). It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IB | T1b N0 M0 | IB | The cancer is only in the uterus and is larger than 5 cm across (about 2 inches). (T1b). It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| II
| T2 N0 M0 | II | The cancer is growing outside the uterus but is not growing outside of the pelvis (T2). It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IIIA | T3a N0 M0 | IIIA | The cancer is growing into tissues of the abdomen in one place only (T3a). It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IIIB | T3b N0 M0 | IIIB | The cancer is growing into tissues of the abdomen in 2 or more places (T3b). It has not spread to nearby lymph nodes (N0) or to distant sites (M0). |
| IIIC | T1-T3 N1 M0 | IIIC | The cancer is growing in the body of the uterus and it might have spread into tissues of the abdomen, but is not growing into the bladder or rectum (T1 to T3). The cancer has spread to nearby lymph nodes (N1), but not to distant sites (M0). |
| IVA | T4 Any N M0 | IVA | The cancer has spread to the rectum or urinary bladder (T4). It might or might not have spread to nearby lymph nodes (Any N) but has not spread to distant sites (M0). |
| IVB | Any T Any N M1 | IVB | The cancer has spread to distant sites such as the lungs, bones, or liver (M1). The cancer in the uterus can be any size and may or may not have grown into tissues in the pelvis and/or abdomen (including the bladder or rectum) (any T) and it might or might not have spread to nearby lymph nodes (Any N). |
* The following additional categories are not listed on the table above:
- TX: Main tumor cannot be assessed due to lack of information.
- T0: No evidence of a primary tumor.
- NX: Regional lymph nodes cannot be assessed due to lack of information.
Survival Rates for Uterine Leiomyosarcoma, by Stage
Survival rates are often used by doctors as a standard way of discussing a person’s prognosis (outlook). The 5-year survival rate refers to the percentage of patients who live at least 5 years after their cancer is diagnosed. Of course, many people live much longer than 5 years (and many are cured).
Five-year relative survival rates compare the survival of people with the cancer to the survival for people without the cancer. This is a way to take into account deaths from causes other than cancer. The 5-year relative survival rate is a better way to describe the impact of a particular type and stage of cancer on survival.
Survival rates are often based on previous outcomes of large numbers of people who had the disease, but they cannot predict what will happen in any particular person’s case. Many factors may affect a person’s outlook, such as:
- The stage of the cancer
- The type of sarcoma (leiomyosarcoma or endometrial stromal sarcoma)
- The grade of the sarcoma (low grade versus high grade)
- The woman’s general state of health
- The treatment received
Your doctor can tell you how the numbers below may apply to you, as he or she is familiar with your particular situation.
The survival statistics noted below come from the National Cancer Institute’s SEER program. They are based on women diagnosed with uterine sarcomas from 2004 to 2010. SEER doesn’t break down these statistics by AJCC or FIGO stage. Instead, SEER uses something called summary stages: localized, regional, and distant.
- Localized means the cancer is only in the uterus, and corresponds to stage I.
- Regional means the cancer has spread to nearby tissues or lymph nodes and includes stages II, and III.
- Distant means the cancer has spread further and includes stages IVA and IVB.
Uterine Leiomyosarcoma 5-Year Relative Survival 25)
- Localized ~ 63%
- Regional ~ 36%
- Distant ~ 14%
Uterine leiomyosarcoma treatment
Extrafascial hysterectomy with pelvic lymph node sampling with or without salpingo-oophorectomy is the surgical gold standard. Debate concerning removal of adnexa and the value of lymph node dissection is still ongoing 26). The survival of younger patients with uterine leiomyosarcoma without oophorectomy has been better in one study which is very controversial. The rate of lymph node metastasis has been between 0–47%, and in some studies survival has not been significantly affected as regards lymph node dissection 27). The role of adjuvant therapies is controversial. Radiotherapy seems to improve local control but not survival. Adjuvant chemotherapy does not decrease the risk of metastatic spread or improve survival. In recurrent uterine sarcomas the response rates in different chemotherapeutic regimens have been between 0–57%. However, the conclusion after a review of the literature was that it is reasonable to offer palliative adjuvant chemotherapy to patients with advanced uterine leiomyosarcoma. The effects of hormone therapy in cases of recurrent uterine leiomyosarcoma have been assessed in only a few studies 28).
Uterine leiomyosarcoma Stages I and II
Most women have surgery to remove the uterus (hysterectomy), as well as the fallopian tubes and ovaries (bilateral salpingo-oophorectomy). Pelvic and para-aortic lymph node dissection or laparoscopic lymph node sampling may be done if swollen nodes are seen on imaging tests. During surgery, organs near the uterus and the thin membrane that lines the pelvic and abdominal cavities (called the peritoneum) are closely checked to see if the cancer has spread beyond the uterus.
Very rarely, young women with low-grade leiomyosarcomas (LMS) that have not spread beyond the uterus may be able to have just the tumor removed, leaving the uterus, fallopian tubes, and ovaries in place. This is not standard treatment, little is known about long-term outcomes , and it’s not often offered. Still, it may be a choice for some women who want to be able to have children after cancer treatment. This option has risks, so women thinking about this surgery need to talk about the pros and cons with their treatment team before making a decision. It may also be possible to leave a young woman’s ovaries in place (but remove the uterus and fallopian tubes), since it isn’t clear that this will lead to worse outcomes. Again, this is not a standard treatment, and you should discuss the risks and benefits with your doctor. In either case, close follow-up is important, and morel surgery may be needed if the cancer comes back.
Women with stage I cancers may not need more treatment and are watched closely after surgery. In other cases, treatment with radiation, with or without chemo, may be needed after surgery if there’s a high chance of the cancer coming back in the pelvis. This is called adjuvant treatment. The goal of surgery is to take out all of the cancer, but the surgeon can only remove what can be seen. Tiny clumps of cancer cells that are too small to be seen can be left behind. Treatments given after surgery are meant to kill those cancer cells so that they don’t get the chance to grow into larger tumors. For leiomyosarcomas of the uterus, adjuvant radiation may lower the chance of the cancer growing back in the pelvis (called local recurrence), but it doesn’t seem to help women live longer.
Since the cancer can still come back in the lungs or other distant organs, some experts recommend giving chemo after surgery (adjuvant chemotherapy) for stage II cancers. Chemo is sometimes recommended for stage I uterine leiomyosarcoma as well, but it’s less clear that it’s really helpful. So far, results from studies of adjuvant chemo have been promising in early stage uterine leiomyosarcoma, but long-term follow-up is still needed to see if this treatment really helps women live longer. Studies of adjuvant therapy are in progress.
Uterine leiomyosarcoma Stage III
Surgery is done to remove all of the cancer. This includes removing the uterus (a hysterectomy), removing both fallopian tubes and ovaries (bilateral salpingo-oophorectomy), and lymph node dissection or sampling. If the tumor has spread to the vagina, part (or even all) of the vagina will need to be removed as well.
After surgery, treatment with radiation (with or without chemo) may be offered to lower the chance that the cancer will come back.
Women who are too sick (from other medical problems) to have surgery may be treated with radiation and/or chemo.
Uterine leiomyosarcoma Stage IV
This is divided into stage IVA and stage IVb.
- Stage IVA cancers have spread to nearby organs and tissues, such as the bladder or rectum, and maybe to nearby lymph nodes. These cancers might be able to be completely removed with surgery, and this is usually done if possible. If the cancer cannot be removed completely, radiation may be given, either alone or with chemo.
- Stage IVB cancers have spread outside the pelvis, most often to the lungs, liver, or bone. There’s no standard treatment for these cancers. Chemo may be able to shrink the tumors for a time, but is not thought to be able to cure the cancer. Radiation therapy, given along with chemo, may also be an option.
These cancers might also be treated with targeted therapy when other treatments don’t work. They’re often given along with chemo.
Leiomyosarcoma causes
The etiology (cause) of leiomyosarcoma is unknown. In individuals with cancer, including leiomyosarcoma, malignancies may develop due to abnormal changes in the structure and orientation of certain cells known as oncogenes or tumor suppressor genes. Oncogenes control cell growth; tumor suppressor genes control cell division and ensure that cells die at the proper time. The specific cause of changes to these genes is unknown. However, current research suggests that abnormalities of DNA (deoxyribonucleic acid), which is the carrier of the body’s genetic code, are the underlying basis of cellular malignant transformation. These abnormal genetic changes may occur spontaneously for unknown reasons or, more rarely, may be inherited.
The risk factors in the development of soft tissue sarcoma and leiomyosarcoma include certain familial syndromes such as neurofibromatosis (benign tumours that form on nerve endings or in the skin); Gardner’s syndrome and Li-Fraumeni syndrome; prior radiation; chronic lymphoedema; and exposure to some rare chemical agents (such as herbicides, arsenic, vinyl chloride, and dioxin). Damage or removal of lymph nodes during a previous cancer treatment is also a risk factor.
Leiomyosarcoma prognosis
The outlook for people with leiomyosarcoma depends on various factors, including tumor location, size, histological grading of the tumor and type, as well as the extent of spread. Some patients with low grade tumors or with tumors that have not spread beyond Stage I have had excellent prognoses. There are numerous long term survivors from this group. In general, high-grade tumors that have spread widely throughout the body have less favorable survival rates.
Leiomyosarcoma Staging
Staging of leiomyosarcoma is important both in guiding treatment and in providing prognostic information. While many staging systems exist for soft tissue sarcoma, the most commonly used system is the American Joint Committee on Cancer (AJCC) system 29). This system classifies the tumor based upon histologic grade, the tumor size, location as superficial or deep, and the presence or absence of metastatic disease (see Table 2).
The Surgical Staging System of the Musculoskeletal Tumor Society (MSTS) is also used. It is utilized for staging bone and soft tissue sarcomas, including leiomyosarcoma 30). This staging system classifies tumors as Ia, Ib, IIa, IIb, or III based upon the histologic grade of the tumor, its local extent and the presence or absences of macroscopic distant metastatic disease. If the tumor is localized to a single anatomic compartment, it is said to be confined. If it has spread locally beyond its initial compartment, then it is said to be unconfined (see Table 3).
Table 2: American Joint Committee on Cancer (AJCC) staging system
| Stage | Histological Grade | Size | Location (Relative to fascia) | Systemic / Metastatic Disease Present |
|---|---|---|---|---|
| IA | Low | < 5cm | Superficial or Deep | No |
| IB | Low | ≥ 5cm | Superficial | No |
| IIA | Low | ≥ 5cm | Deep | No |
| IIB | High | < 5cm | Superficial or Deep | No |
| IIC | High | ≥ 5cm | Superficial | No |
| III | High | ≥ 5cm | Deep | No |
| IV | Any | Any | Any | Yes |
Table 3: Musculoskeletal Tumor Society (MSTS) Staging system
| Stage | Histological Grade | Local Extent of Disease | Systemic / Metastatic Disease Present |
|---|---|---|---|
| Ia | Low | Confined | No |
| Ib | Low | Unconfined | No |
| Ia | High | Confined | No |
| Ib | High | Unconfined | No |
| III | Any | Any | Yes |
Leiomyosarcoma survival rate
Superficial leiomyosarcoma tumors discovered early in their course are associated with a good prognosis, and there is a 5 year survival rate of approximately 90%. Larger leiomyosarcoma tumors diagnosed later in their development can cause death through local extension and metastatic deposits. For example, the 5 year survival rates from uterine leiomyosarcoma are only 63% for stage I disease and fall to less than 20% when there are metastases present outside the uterus.
Since there is no actual data that grouped all leiomyosarcoma together. There is however survival data for soft tissue sarcoma. And since leiomyosarcoma made up about 20% of all sarcomas and the fact that leiomyosarcoma have many types, we can only guesstimate. The rates below are based on the stage of the cancer at the time of diagnosis. When looking at survival rates, it’s important to understand that the stage of a cancer does not change over time, even if the cancer progresses. A cancer that comes back or spreads is still referred to by the stage it was given when it was first found and diagnosed, but more information is added to explain the current extent of the cancer.(And the treatment plan is adjusted based on the change in cancer status.
The overall relative 5-year survival rate of people with soft tissue sarcomas is around 50% according to statistics from the National Cancer Institute (NCI). These statistics include people with Kaposi sarcoma, which has a poorer outlook than many sarcomas. The NCI doesn’t use the AJCC staging system. Instead, they group sarcomas only by whether they are still confined to the primary site (called localized) have spread to nearby lymph nodes or tissues (called regional); or have spread (metastasized) to sites away from the main tumor (called distant). The 5-year survival rates for soft tissue sarcomas have not changed much for many years. The corresponding 5-year relative survival rates were:
- 83% for localized sarcomas (56% of soft tissue sarcomas were localized when they were diagnosed)
- 54% for regional stage sarcomas; (19% were in this stage)
- 16% for sarcomas with distant spread (16% were in this stage)
The 10-year relative survival rate is only slightly worse for these stages, meaning that most people who survive 5 years are probably cured.
Leiomyosarcoma symptoms
The symptoms of a leiomyosarcoma vary depending upon the exact location, size, and spread of the tumor. A leiomyosarcoma, especially in the early stages, may not be associated with any obvious symptoms (asymptomatic). General symptoms associated with cancer may occur including fatigue, fever, weight loss, a general feeling of ill health (malaise), and nausea and vomiting.
Pain may occur in the affected area but is uncommon. Swelling usually occurs and a mass is commonly detected. Additional symptoms are specific to the exact location of the tumor. The tumors may cause bleeding in the gastrointestinal tract and cause black, tarry, foul-smelling stools (melena), or vomiting of blood (hematemesis) or abdominal discomforts). A leiomyosarcoma of the uterus may cause abnormal bleeding from the uterus into and the vagina, with abnormal vaginal discharge, and a change in bladder or bowel habits.
Most forms of leiomyosarcoma are aggressive tumors that may spread (metastasize) to other areas of the body such as the lungs or liver, potentially causing life-threatening complications. Leiomyosarcoma has a high risk of recurring after treatment, if not diagnosed early.
Leiomyosarcoma diagnosis
A diagnosis of a leiomyosarcoma may be made based upon a detailed patient history, a thorough clinical evaluation and a variety of tests including blood tests, surgical removal and microscopic examination of tissue (biopsies) and various imaging techniques.
To confirm a diagnosis of leiomyosarcoma a fine-needle aspiration may be performed. Fine-need aspiration (FNA) is a diagnostic technique in which a thin, hollow needle is passed though the skin and inserted into the nodule or mass to withdraw small samples of tissue. The collected tissue is then studied under a microscope. In some cases, FNA may prove inconclusive and physicians may perform a core (Trucut) biopsy or an incisional biopsy. During Trucut or incisional biopsy, a small sample tissue is surgically removed and sent to a pathology laboratory where it is processed and studied to determine its microscopic structure and makeup (histopathology).
Leiomyosarcoma can be mistaken for many other types of tumors and benign growths, including uterine fibroids (leiomyomas). In fact, most of the leiomyosarcomas that occur in the uterus are presumed to be fibroids because that’s what they look like on imaging tests. The truth is revealed only after a pathologist examines the tissue after a hysterectomy.
Specialized imaging techniques may be used to help evaluate the size, placement, and extension of the tumor and to serve as an aid for future surgical procedures, among individuals with leiomyosarcomas. Such imaging techniques may include computerized tomography (CT) scanning, magnetic resonance imaging (MRI) and ultrasound. During CT scanning, a computer and x-rays are used to create a film showing cross-sectional images of certain tissue structures. An MRI uses a magnetic field and radio waves to produce cross-sectional images of particular organs and bodily tissues. During an ultrasound reflected sound waves create an image of internal organs and other structures within the body.
Laboratory tests and specialized imaging tests may also be conducted to determine possible infiltration of regional lymph nodes and the presence of distant metastases.
Leiomyosarcoma treatment
Treatment of leiomyosarcoma revolves around surgical excision, regardless of the location of the tumor in the body. Radiotherapy may be a useful additive to improve local control or where a cancer is inoperable. In some cases of high-grade leiomyosarcoma, chemotherapy may have a role to reduce the risk of recurrence or to improve operability.
In addition, based upon primary tumor site, size, and other factors, recommended standard therapy may often include postoperative radiation to help treat known or possible residual disease. If initial surgery is not an option due to the specific location and/or progression of the malignancy, therapy may include radiation alone. Radiation therapy preferentially destroys or injures rapidly dividing cells, primarily cancerous cells. However, some healthy cells (e.g., hair follicles, bone marrow, etc.) may also be damaged, leading to certain side effects. Thus, during such therapy, the radiation is passed through diseased tissue in carefully calculated dosages to destroy cancer cells while minimizing exposure and damage to normal cells. Radiation therapy works to destroy cancer cells by depositing energy that damages their genetic material, preventing or slowing their growth and replication.
For some affected individuals, particularly those who have locally advanced, metastatic, or recurrent disease, therapy with certain anticancer drugs (chemotherapy) may also be recommended, possibly in combination with surgical procedures and/or radiation; physicians may recommend combination therapy with multiple chemotherapeutic drugs that have different modes of action in destroying tumor cells and/or preventing them from multiplying.
In most cases, however, chemotherapy and radiation therapy have had only limited success in slowing or stopping progression of leiomyosarcomas. Because of the rarity of leiomyosarcomas no standard or overall effective type of chemotherapy or radiation therapy has been identified. The use of chemotherapy and radiation therapy for the treatment of leiomyosarcomas remains under investigation.
In 2015, Trabectedin (Yondelis®) was approved as a chemotherapy for liposarcoma and leiomyosarcoma that cannot be removed by surgery or patients that previously underwent treatment that contained anthracycline. Trabectedin exerts its anti-cancer affect by a process called alkylation. Alkylation damages the DNA of cells, which prevents them from dividing, and causes them to die. Since cancer cells, in general, divide faster, and with less error-correcting than healthy cells, they are more sensitive to this damage.
It is important to realize that the treatment of leiomyosarcoma is a complicated process. Associated with the treatment of leiomyosarcoma cancer are the very real long-term sequelae of these treatments in those in whom they establish a cure. The surgical sequelae can be serious and immediately evident following wide excision of primary tumors. Consequences of the very high doses of radiotherapy required to establish control of the disease are less evident initially but develop following a lag period of some ten to fifteen years. These depend on the age of the patient and site of leiomyosarcoma, but radiation of large fields, as is often required, can have serious adverse effects on internal organs. In addition, the development of post-radiation tumors such as osteosarcoma is a very real problem. As patients with leiomyosarcoma tend to be older than those with rhabdomyosarcoma these complications of treatment tend to pose less of a problem.
Improvement in symptoms is an important measurement. Specific monitoring may take place through careful clinical follow-up and regular investigations including chest x-ray to exclude pulmonary metastases which could be resected.
The symptoms that may require attention are somatic pain from bone metastases, visceral pain from lung metastases and neurogenic pain if nerve tissue is compressed. Coughing and breathlessness from lung involvement may require specific treatment.
Additional symptoms due to the specific sites involved may require specialist treatment approaches. These can include problems such as lymphoedema where tumors compress the lymphatic drainage of a limb.
References [ + ]