What is Pseudomyxoma Peritonei
Pseudomyxoma peritonei also known as PMP is a very rare cancer that causes a buildup of jelly-like mucus or gelatinous deposits in your abdomen and pelvis (the peritoneal cavity), leading to symptoms like abdominal swelling, pain, and constipation 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14. Pseudomyxoma peritonei typically originates from a polyp (a small growth) in the appendix but can also start in other organs like your ovary, large intestine or from other abdominal tissues. Mucus-secreting cells may attach to the peritoneal lining and continue to secrete mucus. The majority of people with pseudomyxoma peritonei (PMP) develop a “jelly belly” and need surgery to remove the tumors, often combined with chemotherapy called cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherap (HIPEC). However, your treatment depends on the size of the cancer and your general health. Your doctor might decide to closely monitor your cancer if it’s small, slow growing and you don’t currently need treatment. Pseudomyxoma peritonei is considered a rare condition, affecting 1 to 4 people per million each year 15, 16, 6. Pseudomyxoma peritonei incidence is higher in females, approximately 2 to 3 times that of males 17, 18, 19.
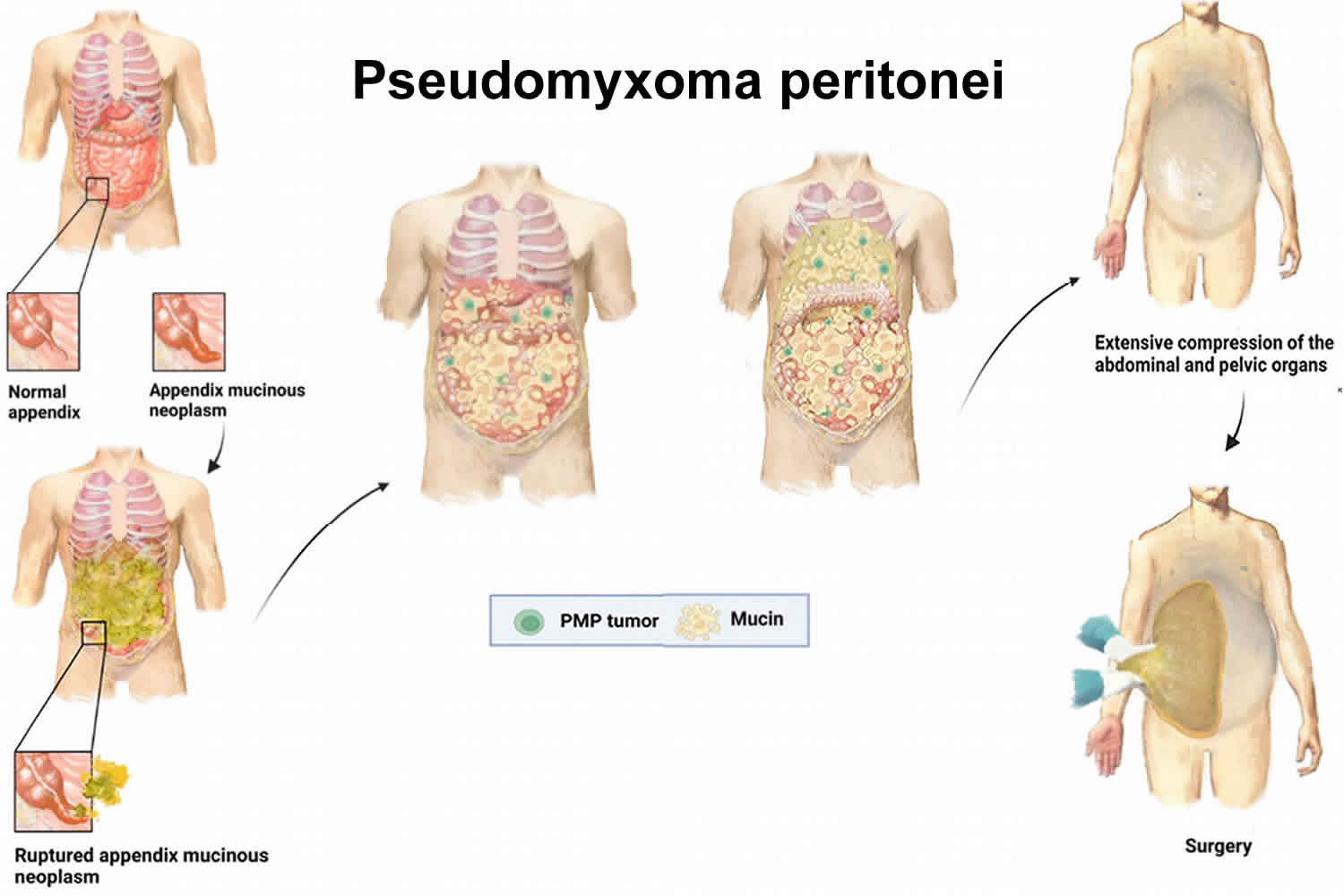
The name pseudomyxoma peritonei literally means “false mucinous tumor of the peritoneum”. Doctors call pseudomyxoma peritonei a false tumor because the cancer doesn’t form a tumor that can spread. Instead, pseudomyxoma peritonei usually starts as a cancerous polyp in your appendix. Pseudomyxoma peritonei commonly begins when a polyp in your appendix bursts, releasing mucin-producing cells into the peritoneum (the lining of your abdomen). When the polyp breaks through your appendix. It triggers a flood of mucin-producing cancer cells that, over time, affect your digestive system. That flood of jelly-like mucin is why some people call pseudomyxoma peritonei “jelly belly”.
Pseudomyxoma peritonei can also start in other organs like your ovary, large intestine or from other abdominal tissues.
Pseudomyxoma peritonei symptoms tend to be mild and may feel like common issues like constipation or indigestion. Sometimes,doctor detect pseudomyxoma peritonei early on while doing your annual exam or while checking for another medical condition. If you’re like most people, however, you’ll find out you have pseudomyxoma peritonei because your belly issues don’t go away or get worse.
In order to diagnose pseudomyxoma peritonei, your doctor will ask questions about your symptoms, like when you first noticed them or if they’re getting worse. Your doctor will do a physical examination and order blood tests and imaging tests to exclude other conditons.
Blood tests. Your doctor may order a complete blood count (CBC) and blood tests to look for tumor markers. Tumor markers are substances in your body that may indicate signs of cancer.
Imaging tests.
Your doctor may order the following imaging tests to look for issues like enlarged organs, fluid buildup in your abdomen (ascites) or mucin deposits in your belly (jelly belly): abdominal ultrasound, CT scan or MRI.
- Imaging tests. Imaging tests such as abdominal ultrasound, CT, MRI and PET scans. These scans help identify visible tumor spread, fluid buildup or organ involvement. However, imaging is often not sensitive enough to detect smaller cancers or early peritoneal disease.
- Biopsy of tissue from the peritoneum. A peritoneal biopsy is a procedure to remove a tissue sample from the peritoneum, the lining of your abdomen, for microscopic examination. It is used to diagnose conditions like peritoneal mesothelioma and can be performed using techniques such as a laparoscopy (keyhole surgery) or a CT-guided/ultrasound-guided core biopsy. The chosen method depends on your health, the location of the suspicious tissue, and the suitability for sampling.
- Peritoneal washing cytology. In this test, fluid from the abdominal cavity is surgically collected during a minor procedure. It’s then examined under a microscope. Doctors use peritoneal washing cytology to check for cancer cells floating in the peritoneal fluid. A medical pathologist will examine fluid or tissue cells under a microscope (cytology).
- Laparoscopy. Laparoscopy (keyhole surgery) is a safe, minimally invasive surgical procedure used to look directly inside your belly using a small camera. It allows your doctor to inspect the peritoneum, find hidden tumors, and take tissue or fluid samples for analysis.
- Tumor marker tests. Tumor marker tests use a sample of blood to look for chemicals made by cancer cells e.g. CEA, CA 199, CA 125.
Your treatment will depend on your situation, including your health. The main treatment for pseudomyxoma peritonei involves surgically removing as much of the tumor as possible, followed by chemotherapy delivered directly into your abdomen during the surgery called cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) 20, 21.
- Cytoreductive surgery (CRS) also known as debulking surgery: Cytoreductive surgery (CRS) is a surgical procedure to remove cancerous tumors from the abdomen and pelvic cavity that have spread to the peritoneum. Cytoreductive surgery (CRS) is often performed alongside heated intraperitoneal chemotherapy (HIPEC), where hot chemotherapy is circulated in the abdomen to kill any remaining microscopic cancer cells. The goal of cytoreductive surgery (CRS) is to completely remove all visible tumors, and the surgery may involve removing other affected organs as well.
- Hyperthermic intraperitoneal chemotherapy (HIPEC): Hyperthermic intraperitoneal chemotherapy (HIPEC) is a two-stage cancer treatment for advanced abdominal cancers that combines surgery to remove tumors with the administration of heated chemotherapy drugs directly into your abdominal cavity. After visible tumors are surgically removed, a heated chemotherapy solution is circulated throughout your abdomen for a short period, typically 90 minutes to two hours to kill any remaining microscopic cancer cells, reducing the risk of recurrence.
Your surgeon will only do cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) if the surgery removes all or almost all of the cancerous tissue or mucin. Your surgeon will make that decision during your surgery.
Other types of surgery treat pseudomyxoma peritonei may include:
- Bowel resection if you have mucin in your small or large intestine
- Cholecystectomy to remove your gallbladder.
- Hysterectomy with bilateral salpingo-oophorectomy to remove your fallopian tubes, ovaries and uterus.
- Liver capsulectomy to remove part of the surface of your liver.
- Omenectomy, which is removing the layer of fat that covers the front of your belly.
- Peritonectomy to remove your peritoneum. This is the membrane that lines the inside of your abdomen and pelvis.
- Splenectomy to remove your spleen.
Other treatments may include active surveillance, radiation therapy, systemic chemotherapy, or palliative care.
- Your doctor may recommend active surveillance if you have early or slow-growing pseudomyxoma peritonei and the risks of surgery outweigh the current risks of the disease. In active surveillance, your doctor will make regular checks on your overall health. Your doctor may do imaging tests and blood tests to see if pseudomyxoma peritonei is spreading.
- Palliative care. If you can’t have surgery, your doctor may recommend treatments like chemotherapy or radiation therapy that will slow down pseudomyxoma peritonei and ease your symptoms.
Doctor are investigating small bowel transplantation as treatment when you have pseudomyxoma peritonei in your small intestine.
Regular follow-up is necessary due to the risk of recurrence, even after successful treatment. In some cases, your doctor can treat and often cure pseudomyxoma peritonei. In some cases, surgery to remove all the cancer cells in your peritoneal cavity successfully controls pseudomyxoma peritonei for a long time 22, 23, 24. Other treatments may help you to live longer with it.
Figure 1. Pseudomyxoma peritonei
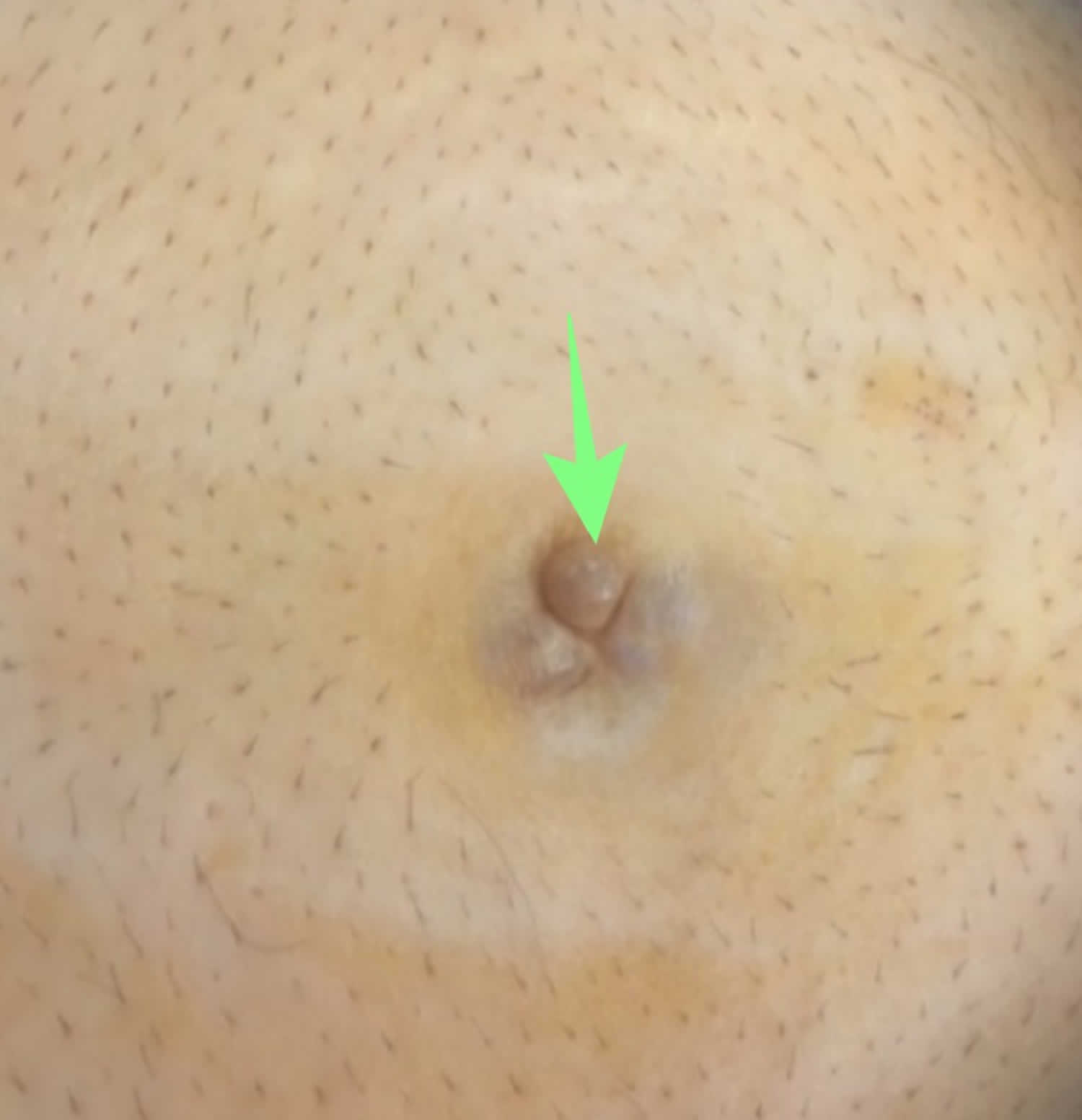
[Source 3 ]Figure 2. Pseudomyxoma peritonei umbilical nodule
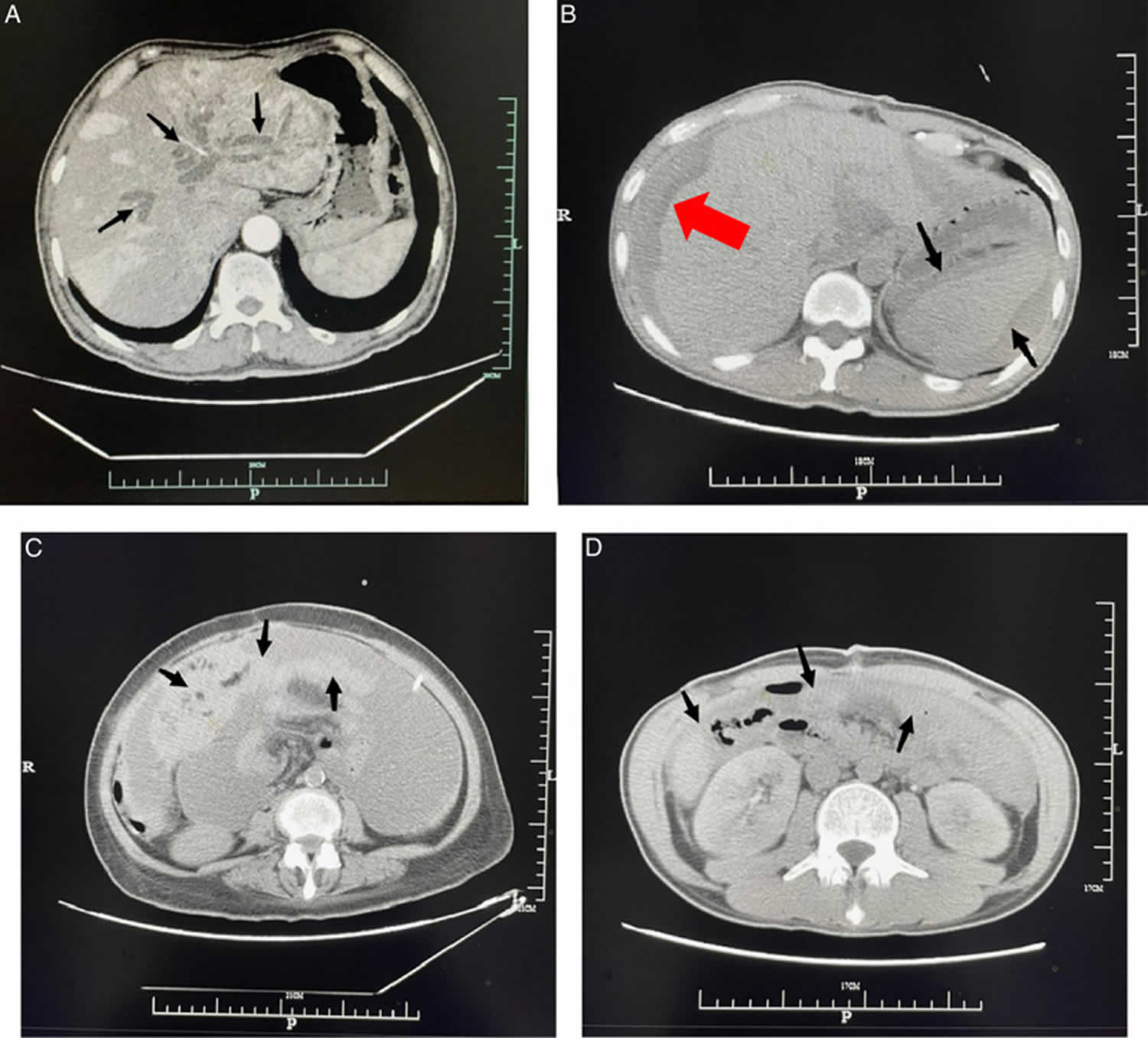
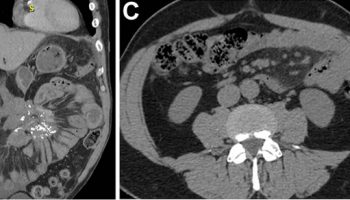
[Source 8 ]Figure 3. Pseudomyxoma Peritonei CT
Footnotes: (A) Scattered accumulations on liver (arrows in A). (B) Sign of liver scalloping (red arrow in B) and deformation of spleen (arrows in B). (C) Omental cake: floccus soft tissue density masses diffused inside the greater omentum and shaped it like biscuits (arrows in C). (D) Massive mucus implanted in the abdominal cavity (arrows in D).
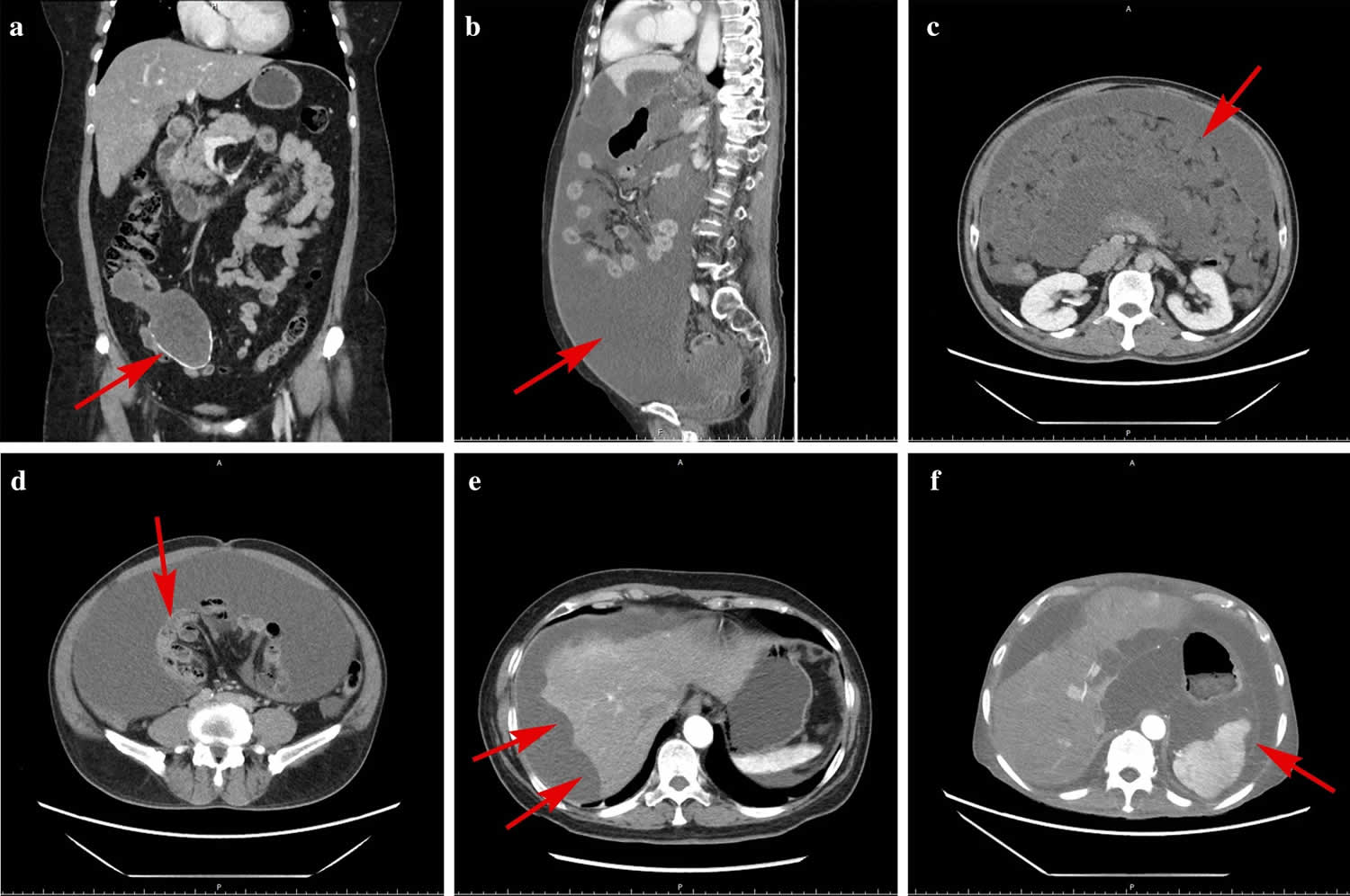
[Source 4 ]Figure 4. Pseudomyxoma Peritonei CT image
Footnotes: Typical computed tomography (CT) characteristics of pseudomyxoma peritonei. Computed tomography (CT) shows the following: (a) Enlargement of the appendiceal cavity and calcification of the appendiceal wall; (b) Abdominal girth enlargement caused by a large volume of intraperitoneal mucus deposits presenting as a “jelly belly”; (c) Thickened greater omentum presenting as an “omental cake”; (d) Small intestines compressed by mucus causing “central displacement”; (e) Scallop impression on the surface of the liver; (f) Contour deformation of the spleen
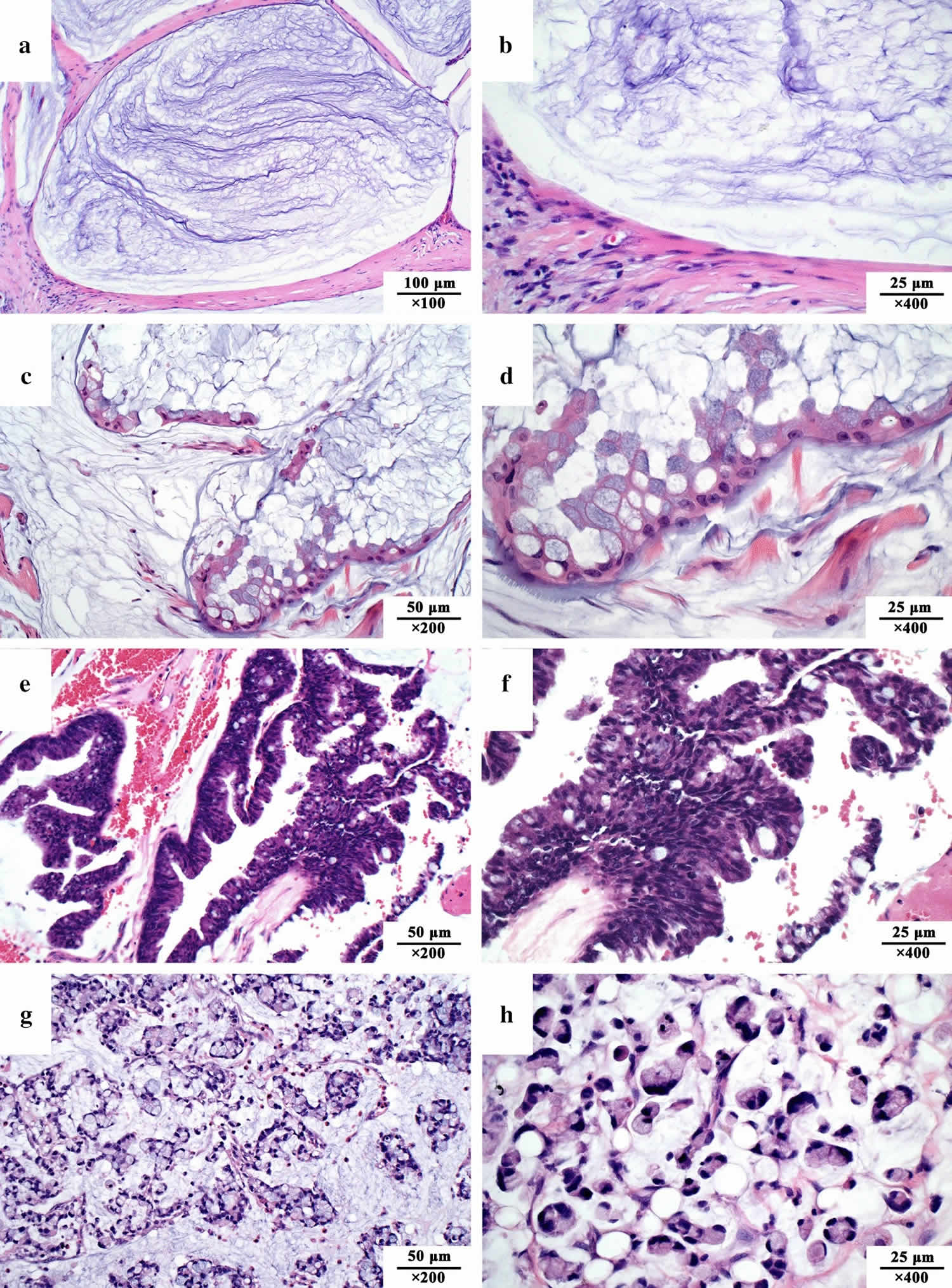
[Source 25 ]Figure 5. Pseudomyxoma peritonei pathological classification
Footnotes: Pathological classification of pseudomyxoma peritonei in the 2016 consensus of the Peritoneal Surface Oncology Group International (PSOGI). (a, b) Acellular mucin, without identifiable tumor cells in the disseminated peritoneal mucinous deposits; (c, d) Low-grade mucinous carcinoma peritonei, with tumor cells forming band-, island-, wave- or cluster-shaped tissue. Cancer cells present with a monolayer or pseudostratified arrangement, with slight nucleus atypia and rare mitotic figures; (e, f) High-grade mucinous carcinoma peritonei, with a complex structure presenting band-, island-, gland-, cribriform-shaped tissue, abundant cellularity, and at least local regional severe atypia; (g, h) High-grade mucinous carcinoma peritonei with signet ring cells, with abundant signet ring cells floating in the mucous pools. All sections were stained with H&E
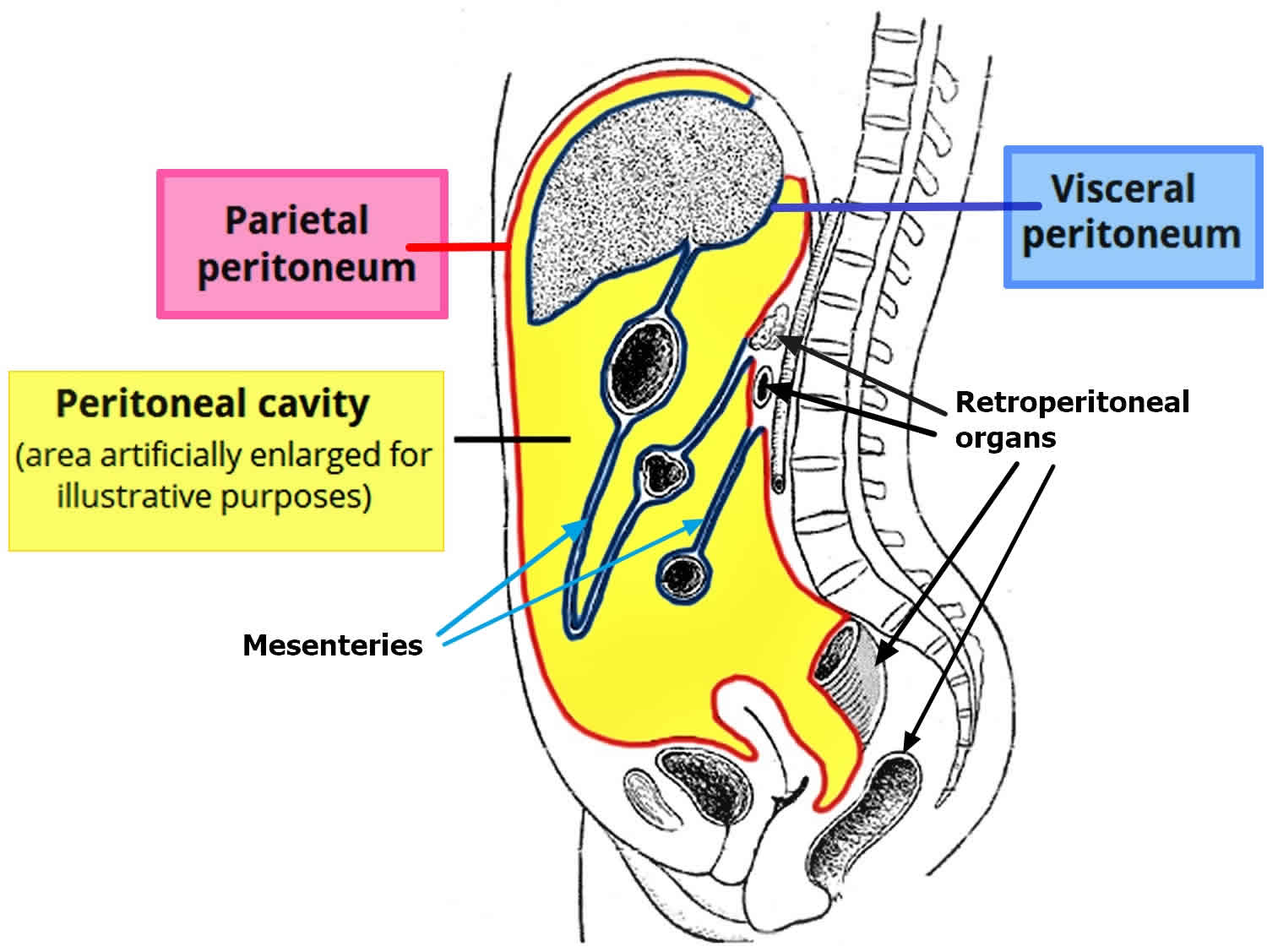
[Source 25 ]What is the peritoneum and peritoneal cavity?
The peritoneum covers all of the organs within your tummy (abdomen), such as the bowel and the liver. The peritoneum protects the organs and acts as a barrier to infection. The peritoneum has 2 layers. One layer lines the abdominal wall and is called the parietal layer. The other layer covers the organs and is called the visceral layer.
There is a small amount of fluid between the two layers, which separates them and allows them to slide over each other. This fluid allows us to move around without causing any friction on the layers. This fluid also contains antibodies to fight infection.
The peritoneal cavity is the space within your tummy (abdomen) that is lined by the peritoneum. The peritoneal cavity contains folds of the peritoneum like the omentum (which hangs down from the stomach) and the mesentery (which supports the intestines). The peritoneal cavity normally contains a small amount of lubricating fluid and is the space where many abdominal organs, such as the liver, stomach, and intestines, are located. Organs located entirely or mostly within the cavity are called intraperitoneal organs, including the stomach, liver, spleen, and parts of the intestines. The peritoneal cavity’s function is to support and protect these organs, allow for their free movement, and provide an entry point for the immune system. The peritoneal cavity contains specialized immune cells, such as macrophages, that are part of the body’s defense system and play a role in tissue homeostasis.
When excess fluid accumulates in the peritoneal cavity, it is a condition known as ascites. The peritoneal cavity can become inflamed or infected, a condition known as peritonitis, often caused by a rupture of an abdominal organ.
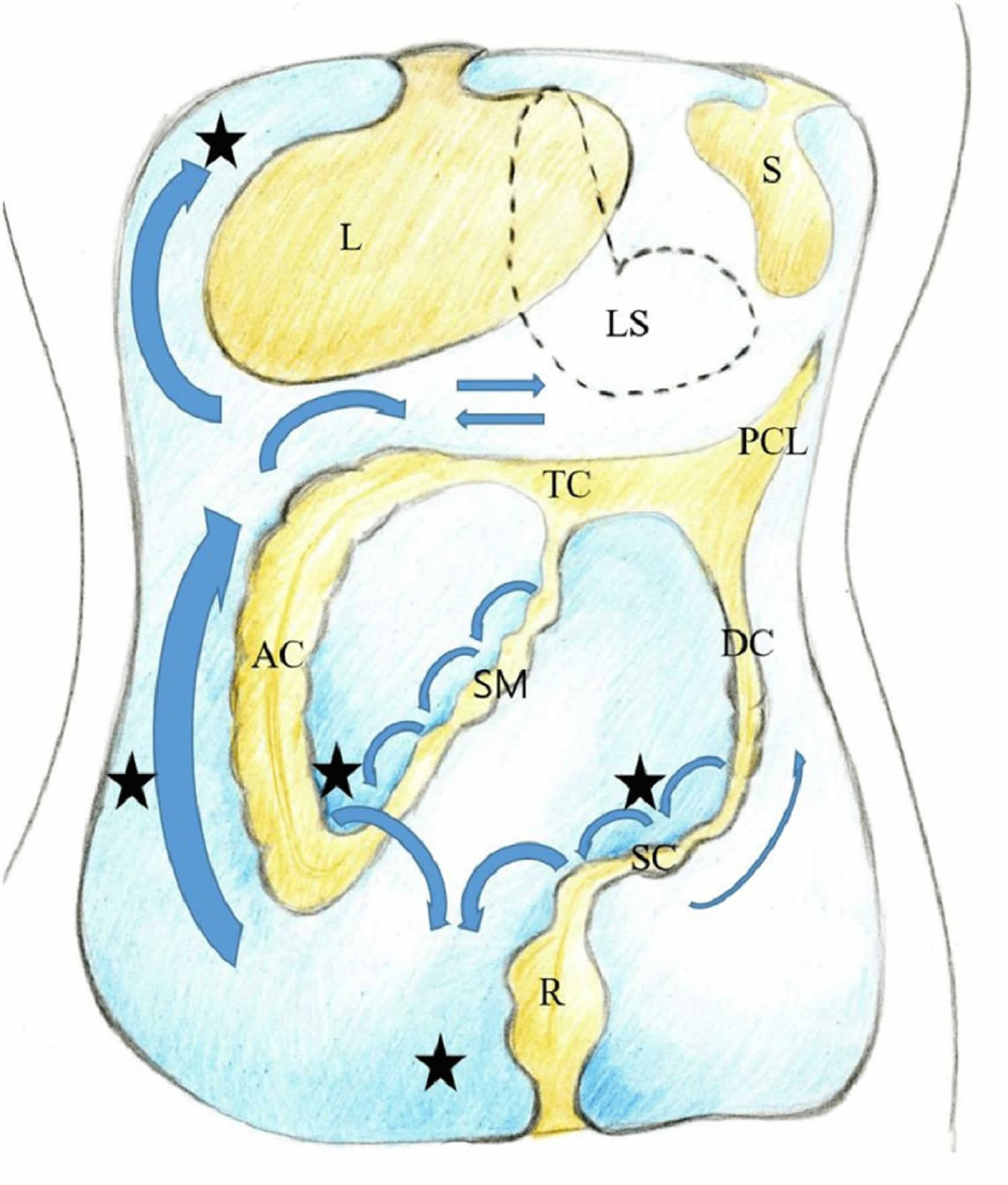
Footnotes: Drawing of the peritoneal cavity illustrating the flow of peritoneal fluid (arrows) and frequent locations for peritoneal seeding (closed stars).
Abbreviations: L = liver; LS = lesser sac; S = spleen; TC = transverse mesocolon; PCL = phrenicocolic ligament; AC = ascending colon; DC = descending colon; SM = small bowel mesentery; SC = sigmoid mesocolon; R = rectum.
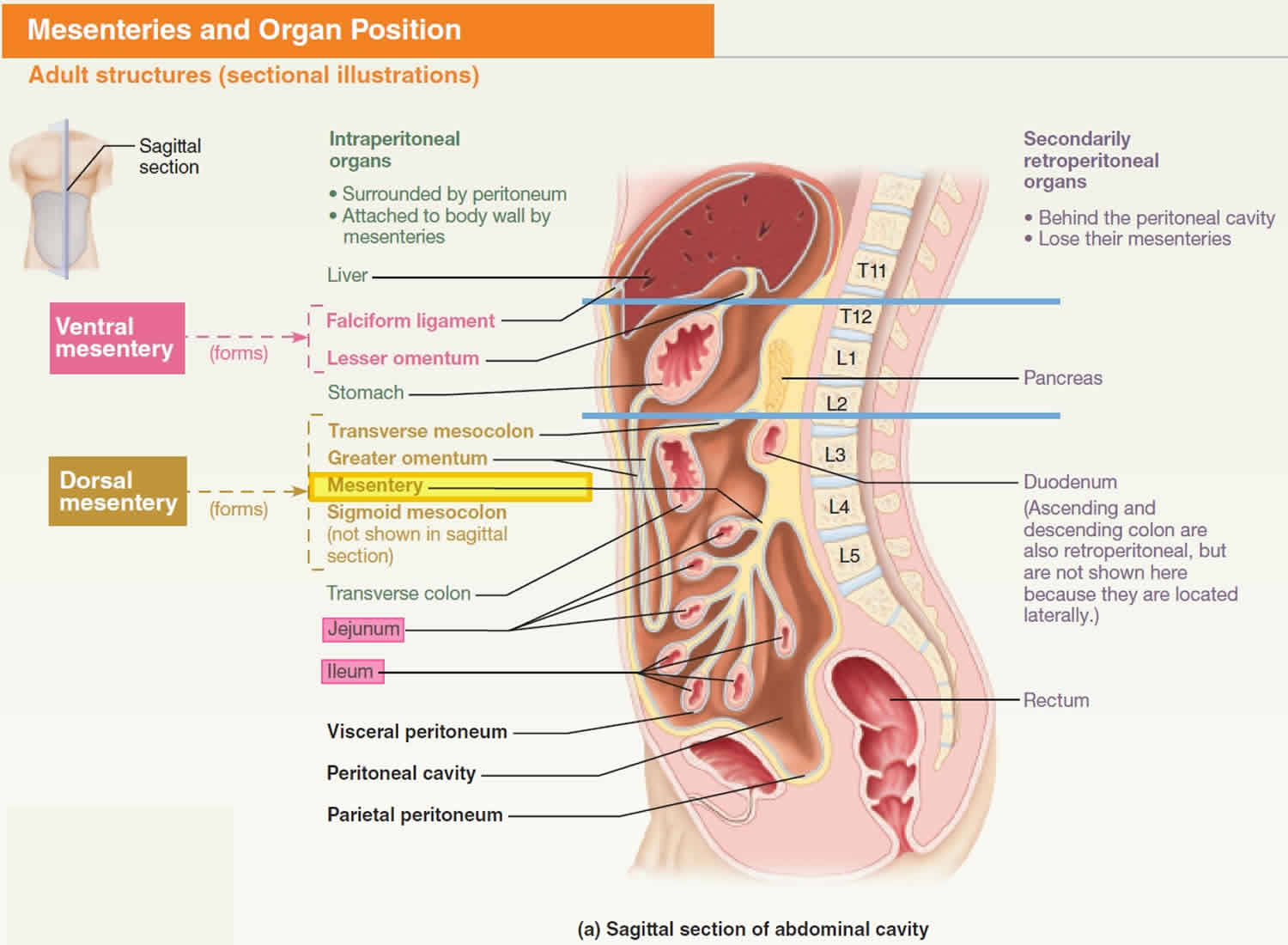
[Source 26 ]Figure 7. Mesentery anatomy
Footnotes: Anterior and sagittal sections of the abdominal cavity. The mesentery is a double fold of the peritoneum. There are true and specialized mesenteries. The true mesenteries connect to the posterior peritoneal wall. These are small bowel mesentery, transverse mesocolon and sigmoid mesentery (or mesosigmoid). The specialized mesenteries do not connect to the posterior peritoneal wall. These are greater omentum, lesser omentum and mesoappendix. The omentum is divided into the greater and lesser omentum. The greater omentum is subdivided into: gastrocolic ligament, gastrosplenic ligament and gastrophrenic ligament. The lesser omentum is subdivided into gastrohepatic ligament and hepatoduodenal ligament.
Pseudomyxoma peritonei causes
The cause of pseudomyxoma peritonei is unknown. Researchers know most pseudomyxoma peritonei starts with a mucinous tumor or polyp in your appendix that rupture, followed by massive colonization of tumor cells in the peritoneal cavity and continued production of mucus. Cases of pseudomyxoma peritonei caused by mucinous tumors originating from organs such as the ovaries, colon, pancreas, and urachus have also been reported 27, 28, 29, 30. Patients with familial adenomatous polyposis (FAP) demonstrate an increased risk of developing mucinous adenocarcinoma of the appendix. KRAS mutation has also been present in 70 % of appendiceal adenomas 31. Pseudomyxoma peritonei is more common in women than men. In women, this type of cancer can sometimes be confused with ovarian cancer. Ovarian cancer may also cause a swollen abdomen. Some types of ovarian cancer cells also produce mucin.
The main feature of pseudomyxoma peritonei is the extensive dissemination of copious mucus-containing tumor cells in the abdominal cavity. Mucus accumulation causes progressive abdominal distention, intestinal obstruction, malnutrition, cachexia, and ultimately death.
Pseudomyxoma peritonei pathological classification criteria and diagnostic terminology are confusing because there are multiple classification systems in the world (Table 1). This confusion is indicative of the diverse clinical manifestations, variable pathological characteristics, and elusive features of pseudomyxoma peritonei. The commonly used pathological classification methods in the literature include the Ronnett three-tier system 32, the Bradley two-tier system 33, and the WHO two-tier system 34. The simultaneous use of different classification systems may have the following disadvantages: (1) the research results of different centers are heterogeneous and are thus not conducive to the comparison of identical or similar studies; (2) as a rare disease, it is not conducive to the organization of relatively scarce research resources for collaborative studies; (3) Ronnett’s three-tier system includes non-appendiceal pseudomyxoma peritonei; and (4) both the Bradley and WHO systems leave out the classification of signet ring cells.
At the 12th International Conference on Peritoneal Carcinoma in Berlin in 2012, experts had heated discussions on pseudomyxoma peritonei pathological classification and diagnostic terminology. It was not until 2016 that a written consensus by the Peritoneal Surface Oncology Group International (PSOGI) on pseudomyxoma peritonei pathology classification and diagnostic terminology was published 35. Currently, the 2016 Peritoneal Surface Oncology Group International (PSOGI) classification system is widely recognized by peritoneal carcinomatosis experts around the world.
According to the Peritoneal Surface Oncology Group International (PSOGI) consensus, pseudomyxoma peritonei is divided into 4 categories:
- Acellular mucin;
- Low-grade mucinous carcinoma peritonei (LMCP) or disseminated peritoneal adenomucinosis (DPAM);
- High-grade mucinous carcinoma peritonei (HMCP) or peritoneal mucinous carcinomatosis (PMCA); and
- High-grade mucinous carcinoma peritonei with signet ring cells (HMCP-S) or peritoneal mucinous carcinomatosis with signet ring cells (PMCA⁃S).
It should be noted that DPAM and PMCA are synonyms for LMCP and HMCP, respectively, which are no longer recommended as standard pathological terminology 36.
Table 1. Histologic Classifications of Pseudomyxoma Peritonei
| Classification | Description | |
| Oscar Polano 1921 37 | The cystadenoma mucinosum peritonei simplex | Superficial implantation on the peritoneum |
| The cystadenoma malignum pseudomucinosum peritonei | Aggressive and destructive features with malignant performance of penetrating abdominal cavity in greater size, spreading to more sites and even perforating the intestines | |
| Ronnett et al 32 | Disseminated peritoneal adenomucinosis (DPAM) | DPAM comprised peritoneal lesions composed of numerous extracellular mucin-containing scant simple to focally proliferative mucinous epithelium with minimal-to-moderate cytologic atypia and inapparent mitotic activity, with or without an associated appendiceal mucinous adenoma |
| Peritoneal mucinous carcinomatosis (PMCA) | Peritoneal lesions that accord with morphologic and cytologic characteristics of carcinoma as more abundant epithelium proliferate in glands, nests, or individual cells, with or without an associated primary mucinous adenocarcinoma | |
| Hybrid tumors | Peritoneal mucinous adenocarcinoma with intermediate features | |
| Bradley et al 33 | Low-grade mucinous carcinoma peritonei (MCP-L) | Cases have a significant adenoma-like or well-differentiated component and should lack a poorly differentiated component including Signet ring cells |
| High-grade mucinous carcinoma peritonei (MCP-H) | Cases with moderately or any poorly differentiated component, that includes all cases with a well-developed signet-ring cell component | |
| AJCC and WHO 2010 38 | Low-grade pseudomyxoma peritonei | Mucin pools with low cellularity (<10%), bland cytology and nonstratified cuboidal epithelium |
| High-grade pseudomyxoma peritonei | Mucin pools with high cellularity, moderate/severe cytologic atypia and cribriform/signet ring morphology with desmoplastic stroma | |
| Peritoneal Surface Oncology Group International (PSOGI) 2016 35 | Acellular mucin (AC) | Mucin without epithelial cells |
| Low-grade mucinous carcinoma peritonei/disseminated peritoneal adenomucinosis (DPAM) | Pseudomyxoma peritonei with low-grade histologic features | |
| High-grade mucinous carcinoma peritonei/peritoneal mucinous carcinomatosis (PMCA) | Pseudomyxoma peritonei with high-grade histologic features | |
| High-grade mucinous carcinoma peritonei with signet ring cells/Peritoneal mucinous carcinomatosis with signet ring cells (PMCA-S) | Pseudomyxoma peritonei with signet ring cells |
Abbreviations: PMP = pseudomyxoma peritonei; PSOGI = Peritoneal Surface Oncology Group International; AJCC = American Joint Committee on Cancer; WHO = World Health Organization.
[Source 4 ]Table 2. Pathological classification and terminology of the pseudomyxoma peritonei
| 2016 Peritoneal Surface Oncology Group International (PSOGI) classification | Counterparts | |
|---|---|---|
| 2017 American Joint Committee on Cancer (AJCC) staging system, 8th edition (TNM) | 2019 World Health Organization (WHO) classification of tumors, 5th edition | |
| Acellular mucin: (1) Mucin without neoplastic epithelium; (2) Confined to or distant from organ surface | M1a | pM1a |
| Low-grade mucinous carcinoma peritonei (LMCP): (1) Low-grade cytology; (2) Rare mitosis; (3) Few tumoral mucinous epithelium (< 20% of tumor volume) | M1b. G1, well-differentiated | pM1b, Grade 1: (1) Hypocellular mucinous deposits; (2) Neoplastic epithelial elements have low-grade cytology; (3) No infiltrative-type invasion |
| High-grade mucinous carcinoma peritonei (HMCP): Features of one or more of the following (At least focally): (1) high-grade cytology; (2) Infiltration of adjacent tissues; (3) Invasion of vascular lymphatic vessels or surrounding nerves; (4) Cribriform growth; (5) Neoplastic mucinous epithelium (> 20% of tumor volume); Sub-classification based on differentiation (1) well-differentiated: Mainly composed of single- tubular glands; Tumor cell polarity exists; Obvious tumor cell atypia; Infiltrative components; (2) Moderately-differentiated: Solid sheet tumor cells mixed with adenoid structures; Minimal or no polarity; (3) Poorly-differentiated: Highly irregular to no adenoid differentiation Cell polarity disappears | M1b. G2 or G3, moderately- or poorly-differentiated | pM1b, Grade 2: (1) Hypercellular mucinous deposits as judged at 20 × magnification; (2) High-grade cytological features; (3) Infiltrative-type invasion characterized by jagged or angulated glands in a desmoplastic stroma, or a small mucin pool pattern with numerous mucin pools containing clusters of tumor cells |
| High-grade mucinous carcinoma peritonei with signet ring cells (HMCP-S): Tumor with signet ring cell component (signet ring cells ≥ 10%) | M1b. G3, poorly- differentiated; PMCA-S (peritoneal mucinous carcinomatosis with signet ring cells) | pM1b, Mucinous tumor deposits with signet-ring cells |
Abbreviations: PMP = pseudomyxoma peritonei; PSOGI = Peritoneal Surface Oncology Group International; AJCC = American Joint Committee on Cancer. AM = acellular mucin; LMCP = low-grade mucinous carcinoma peritonei; HMCP = high-grade mucinous carcinoma peritonei; HMCP-S = high-grade mucinous carcinoma peritonei with signet ring cells; DPAM, disseminated peritoneal adenomucinosis; PMCA-I, peritoneal mucinous carcinomatosis with intermediate feature; PMCA, peritoneal mucinous carcinomatosis; NA, not applicable
[Source 25 ]How does pseudomyxoma peritonei spread?
Pseudomyxoma peritonei doesn’t act like most cancers. It rarely spreads through the bloodstream or the lymphatic system to any other part of the body. Instead, it spreads inside your abdomen. The cancer cells generally spread by following the peritoneal fluid flow. They attach to the peritoneum at particular sites. Here they produce mucus which collects inside the abdomen and eventually causes symptoms. Without treatment, it will take over the peritoneal cavity. It can press on the bowel and other organs.
Pseudomyxoma peritonei develops very slowly. It might be years before you have any symptoms of this type of cancer. Because of this, it has usually spread beyond the appendix before diagnosis.
Pseudomyxoma peritonei signs and symptoms
Pseudomyxoma peritonei symptoms develop slowly and you may not notice them or assume your symptoms are from a common illness. Some people won’t have any symptoms of pseudomyxoma peritonei.
Pseudomyxoma peritonei symptoms may include:
- Swollen abdomen (abdominal distension). This is when your abdomen is noticeably swollen.
- Abdominal pain. This may feel like general discomfort or pain as mucin builds up in your belly.
- Constipation. Abdominal swelling may block your large intestine so you can’t pass poop.
- Difficulty getting pregnant. Pressure from mucin buildup or inflammation in your reproductive organs can make it difficult to conceive, especially for women.
- Hernias. Mucin buildup may push one of your organs through a gap in your muscle wall. Inguinal hernia is one of the most common pseudomyxoma peritonei signs in men.
- Loss of appetite.
- Feeling of fullness.
- Changes in bowel habits.
- Nausea.
Often, doctors can only diagnose pseudomyxoma peritonei properly after an operation to look into the abdomen. This operation is called a laparotomy.
Pseudomyxoma peritonei diagnosis
Pseudomyxoma peritonei symptoms tend to be mild and may feel like common issues like constipation or indigestion. Sometimes,doctor detect pseudomyxoma peritonei early on while doing your annual exam or while checking for another medical condition. If you’re like most people, however, you’ll find out you have pseudomyxoma peritonei because your belly issues don’t go away or get worse.
In order to diagnose pseudomyxoma peritonei, your doctor will ask questions about your symptoms, like when you first noticed them or if they’re getting worse. Your doctor will do a physical examination and order blood tests and imaging tests to exclude other conditons.
Blood tests. Your doctor may order a complete blood count (CBC) and blood tests to look for tumor markers. Tumor markers are substances in your body that may indicate signs of cancer.
Imaging tests.
Your doctor may order the following imaging tests to look for issues like enlarged organs, fluid buildup in your abdomen (ascites) or mucin deposits in your belly (jelly belly): abdominal ultrasound, CT scan or MRI.
Pseudomyxoma peritonei diagnosis may include:
- Imaging tests. Imaging tests such as CT, MRI and PET scans are typically the first step in looking for suspected peritoneal carcinomatosis. These scans help identify visible tumor spread, fluid buildup or organ involvement. However, imaging is often not sensitive enough to detect smaller cancers or early peritoneal disease. Therefore, a negative scan does not rule out peritoneal carcinomatosis.
- Biopsy of tissue from the peritoneum. A peritoneal biopsy is a procedure to remove a tissue sample from the peritoneum, the lining of your abdomen, for microscopic examination. It is used to diagnose conditions like peritoneal mesothelioma and can be performed using techniques such as a laparoscopy (keyhole surgery) or a CT-guided/ultrasound-guided core biopsy. The chosen method depends on your health, the location of the suspicious tissue, and the suitability for sampling.
- Peritoneal washing cytology. In this test, fluid from the abdominal cavity is surgically collected during a minor procedure. It’s then examined under a microscope. Doctors use peritoneal washing cytology to check for cancer cells floating in the peritoneal fluid. Even when no visible cancer is present, a positive cytology result is a strong sign that peritoneal spread has happened.
- Laparoscopy. Laparoscopic exploration (keyhole surgery) is a safe, minimally invasive surgical procedure used to look directly inside the abdominal cavity using a small camera. It allows your doctor to inspect the peritoneum, find hidden tumors, and take tissue or fluid samples. This test is especially valuable for finding peritoneal metastases that are too small to be seen with imaging.
- Tumor marker tests. Tumor marker tests use a sample of blood to look for chemicals made by cancer cells e.g. CEA, CA 199, CA 125.
- Circulating tumor DNA (ctDNA). This is a newer blood test that looks for small pieces of DNA from cancer cells in the blood. It can help find peritoneal cancer that doesn’t show up on scans. However, the role of ctDNA in diagnosis is still uncertain.
Sometimes metastatic cancer to the peritoneum is diagnosed during surgery for another issue or for another abdominal cancer.
A consensus on the preoperative evaluation for pseudomyxoma peritonei was reached in the 2008 PSOGI Consensus, which greatly facilitated patient diagnosis and selection, mainly including 4 aspects 39.
- Serum tumor markers, which mainly combined testing of carcinoembryonic antigen (CEA), carbohydrate antigen 125 (CA125), and carbohydrate antigen 199 (CA199). CEA, CA125, and CA199 are helpful indicators for evaluating the degree of tumor invasion, ascites production and tumor burden, and the proliferation of cancer cells, respectively.
- A computed tomography (CT) examination + 3D reconstruction is the optimal choice for routine preoperative examination. Typically, CT scan of pseudomyxoma peritonei revealed a right lower abdominal cystic or cystic-solid mass frequently with calcification; copious mucinous ascites in the abdominal cavity; extensive organ invasion or compression;
- Laparoscopic exploration and exfoliative cytology are both optional.
Imaging Studies
Several imaging studies may be utilized to evaluate patients with suspected or known peritoneal carcinomatosis 40. Although regular CT has been chosen as the preferred technique in the follow-up, omitting recurrence could happen when both peritoneum and omentum resections have been done and tumor infiltration comes along the small bowel 41. It is appropriate for low-grade pseudomyxoma peritonei patients to get annual CT scan of abdomen and pelvis in the first 6 years, chest examination and the frequency should be added if meets high-grade lesion 42. Magnetic resonance imaging (MRI) owns particular advantages in high sensitivity of the assessment of pseudomyxoma peritonei, as gelatinous implants that consist of plenty of water molecules show high signal intensity on T2-weighted images 43. It is deemed a normal phenomenon if peritoneal enhancement is equivalent to muscle enhancement 44. Magnetic resonance imaging (MRI) provides tough evidence of metastasis in the liver and perihepatic region for its favorable soft tissue contrast which is related to poor prognosis 41.
Computed tomography (CT)
A contrast-enhanced computed tomography (CT) scan of the abdomen and pelvis is the primary imaging modality for evaluating patients with suspected or known peritoneal carcinomatosis 45. A contrast-enhanced computed tomography (CT) scan of the abdomen and pelvis provides detailed anatomic information, suggesting tumor deposits along the peritoneal reflections, the greater and lesser omenta, mesentery, and serosal surfaces of abdominal organs. Features that raise suspicion for peritoneal carcinomatosis include soft-tissue nodules on the peritoneal surfaces, focal or diffuse peritoneal thickening, omental caking, and ascites. CT can also help identify mucinous components by revealing low-attenuation masses, characteristic of pseudomyxoma peritonei (see Image. Pseudomyxoma Peritonei) secondary to appendiceal or ovarian neoplasms. However, sensitivity may be limited for detecting smaller lesions (<5 mm), and the disease burden in the small bowel or mesentery is frequently underestimated due to overlapping loops of bowel and variable bowel distension 46.
Several radiologic signs can predict a high likelihood of nonresectability, eg, a large solitary mass in the upper abdomen (often >5 cm), diffuse small bowel wall thickening, or the “smudge” sign where small bowel loops appear matted together, findings sometimes referred to collectively as “Sugarbaker’s Criteria” or the Peritoneal Cancer Index (PCI). Although CT is routinely used to approximate the Peritoneal Cancer Index (PCI), studies suggest it can either underestimate or overestimate the true extent of disease by up to 20% to 30% 47.
Magnetic resonance imaging (MRI)
Magnetic resonance imaging (MRI) plays an increasingly vital role in evaluating peritoneal carcinomatosis, offering superior soft-tissue contrast and multi-planar capabilities without the risks associated with ionizing radiation. In particular, diffusion-weighted imaging enhances the detection of small peritoneal deposits and mucinous lesions, which are critical for assessing conditions, such as pseudomyxoma peritonei. When combined with contrast-enhanced sequences, MRI further refines staging by clearly delineating the extent of disease, a key factor in planning cytoreductive surgery and intraperitoneal chemotherapy 48.
Recent studies have demonstrated that MRI can achieve sensitivity values of approximately 90% to 95% and specificity values of around 85% to 92% for detecting peritoneal metastases, particularly for lesions larger than 5 mm 49.
Positron emission tomography (PET)
Fluorodeoxyglucose positron emission tomography (FDG-PET), typically combined with computed tomography (PET/CT), can reveal hypermetabolic foci within peritoneal surface cancers and detect extraperitoneal metastases that may alter therapeutic decision-making. Its sensitivity, however, is limited in low-cellularity mucinous neoplasms, including pseudomyxoma peritonei, where radiotracer uptake can be insufficient for accurate detection. Moreover, inflammatory changes or surgical manipulation can yield false-positive signals. Therefore, PET/CT is generally regarded as an adjunct to contrast-enhanced CT or MRI, most valuable for ruling out distant metastatic disease or clarifying ambiguous findings rather than serving as the principal staging modality 50, 51.
Diagnostic Laparoscopy
Diagnostic laparoscopy provides the most accurate assessment of peritoneal disease burden and distribution, while also allowing for the collection of biopsies 52. A diagnostic laparoscopy is a “keyhole” surgery that uses a thin, lighted camera to examine the abdominal and pelvic organs when other tests are inconclusive. A surgeon makes a small incision, often near the belly button, to insert the laparoscope and inflatable gas to get a clear view. Diagnostic laparoscopy should be performed in all patients for whom cytoreduction and intraperitoneal chemotherapy are considered, and is often also performed in cases where imaging is unclear 52. By permitting real-time assessment of tumor distribution and burden, particularly in areas prone to underestimation on CT or MRI, laparoscopy enables a more precise estimation of the Peritoneal Cancer Index (PCI) and aids in evaluating resectability (see Figure 8. Peritoneal Cancer Index below) 53, 54, 55.
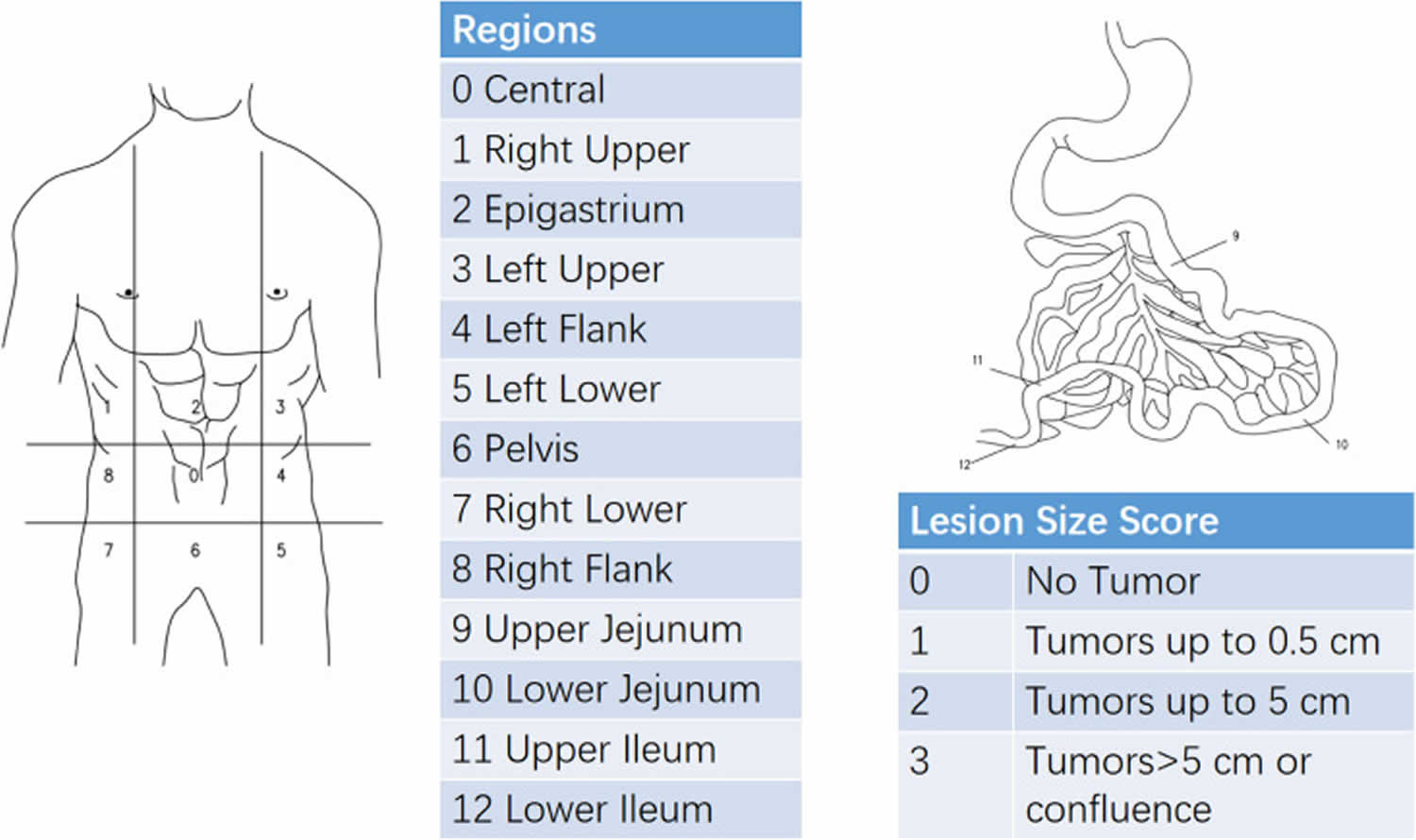
Peritoneal Cancer Index (PCI) is a standardized quantitative tool for estimating the extent and distribution of peritoneal carcinomatosis. It partitions the abdominal cavity into 13 discrete anatomical regions (0 through 12) and assigns each region a lesion size (LS) score ranging from 0 to 3. The Peritoneal Cancer Index (PCI) is then calculated by summing the lesion size scores across all areas, with a maximum possible value of 39 reflecting extensive disease. Notably, lesion size 3 (>5 cm or confluence of disease) is associated with a poor prognosis 56, 57.
In clinical practice, the Peritoneal Cancer Index (PCI) is a robust surrogate for tumor burden, correlates strongly with both resectability and survival outcomes, and often dictates eligibility for cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC). While radiologic estimates of the PCI using CT or MRI can guide patient selection for surgery, definitive scoring typically occurs intraoperatively via either laparotomy or, in selected centers, laparoscopic exploration. Accurate scoring is paramount, as patients with a PCI exceeding a certain threshold—commonly 20 for high-grade appendiceal or colorectal cancer, for example—are more likely to have incomplete cytoreduction and diminished survival benefits from aggressive surgical intervention. Consequently, the Peritoneal Cancer Index (PCI) is integral to risk stratification, surgical planning, and counseling regarding prognosis in the interprofessional management of peritoneal carcinomatosis 56.
Peritoneal Cancer Index (PCI)
The Peritoneal Cancer Index (PCI) is a numerical score used to quantify the extent of cancer spread throughout the abdomen and pelvis 56, 57. The Peritoneal Cancer Index (PCI) is determined during surgery by dividing the abdomen into 13 regions, scoring each from 0 to 3 based on tumor size, and summing the scores for a total Peritoneal Cancer Index (PCI) value 58, 59, 60. A lower Peritoneal Cancer Index (PCI) indicates less extensive disease and is associated with a better prognosis and a higher likelihood of successful complete cytoreductive surgery, while a higher Peritoneal Cancer Index (PCI) suggests more widespread disease.
How Peritoneal Cancer Index (PCI) is calculated 59, 60, 61:
- The abdomen and pelvis are divided into 13 regions:
- 0. Central.
- 1. Right upper.
- 2. Epigastrium.
- 3. Left upper.
- 4. Left flank.
- 5. Left lower.
- 6. Pelvis.
- 7. Right lower.
- 8. Right flank.
- 9. Upper jejunum.
- 10. Lower jejunum.
- 11. Upper ileum.
- 12. Lower ileum.
- Each region is assigned a score from 0 to 3, based on the size of the largest tumor in that region:
- 0: No tumor
- 1: Tumor implants up to 0.5 cm
- 2: Tumor implants between 0.5 cm and 5 cm
- 3: Tumor implants larger than 5 cm or clusters of implants
The scores from all 13 regions are added together to get a total Peritoneal Cancer Index (PCI) score. The sum of the scores of all regions gives the total Peritoneal Cancer Index (PCI) value, which can vary between 0 and 39.
What the Peritoneal Cancer Index (PCI) score means:
- Lower Peritoneal Cancer Index (PCI) ≥ 20: Indicates limited disease spread and is associated with a better prognosis and a higher chance of achieving complete cytoreductive surgery. These patients are optimal candidates for cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC).
- Higher Peritoneal Cancer Index (PCI) >20: Is associated with more widespread disease and a lower chance of achieving complete cytoreductive surgery.
- Peritoneal Cancer Index (PCI) of 20-30 still represents potentially resectable disease for some patients. Neoadjuvant chemotherapy may be used before CRS/HIPEC to help shrink the tumor volume. Outcomes begin to decrease more rapidly above a PCI of 25.
- Peritoneal Cancer Index (PCI) above 30-40 typically means the cancer has spread too extensively throughout the peritoneum to be fully resected surgically. Palliative management focusing on symptom relief usually provides the best option at this point rather than cure-directed treatment.
The Peritoneal Cancer Index (PCI) scoring system helps to evaluate tumor load in the abdominal cavity and has important significance for confirming regions in the peritoneum that need to be removed or stripped or whether an optimal cytoreductive surgery (CRS) can be performed. A high Peritoneal Cancer Index (PCI) score is an independent factor for poor Progression-Free Survival (PFS) which is the length of time a patient lives with their disease but without it getting worse, which is a key measure of how well a cancer treatment is working 62.
A higher Peritoneal Cancer Index (PCI) is a negative prognostic factor especially in cancers like ovarian cancer, colorectal cancer, and pseudomyxoma peritonei, meaning it is associated with poorer survival outcomes.
The Peritoneal Cancer Index (PCI) helps surgeons decide if a complete removal of the tumor is possible and if the patient is a good candidate for procedures like cytoreductive surgery combined with hyperthermic intraperitoneal chemotherapy (CRS-HIPEC).
Some studies use Peritoneal Cancer Index (PCI) categories of: 0-10 (very limited), 10-20 (limited), 20-30 (moderate), and over 30 (extensive). These provide a simplified way of stratifying patients into prognosis groups to help guide treatment decisions and set goals.
Changes in Peritoneal Cancer Index (PCI) over the course of treatment and follow-up indicate how well the cancer is being controlled. A declining Peritoneal Cancer Index (PCI) shows the therapy is working, while a rising PCI suggests progressive disease and treatment failure. Close monitoring Peritoneal Cancer Index (PCI) is used to detect recurrence early.
Diagnostic laparoscopy provides very accurate estimates for Peritoneal Cancer Index (PCI) along with probable completeness of the cytoreduction (CC) index and outcome assessment in terms of disease free survival [disease free survival (DFS) is the length of time after primary cancer treatment that a patient lives without any signs or symptoms of the cancer], overall survival (overall survival (OS) is the length of time from either the date of diagnosis or the start of treatment for a disease, such as cancer, that patients diagnosed with the disease are still alive) and quality of life.
The completeness of cytoreduction (CC) score is the main prognostic factor for peritoneal carcinomatosis patients (Figure 9). The completeness of cytoreduction (CC) score is suitable for pseudomyxoma peritonei, colon cancer peritoneal metastasis, peritoneal sarcomatosis, peritoneal malignant mesothelioma, and ovarian cancer peritoneal metastasis 63. The completeness of cytoreduction (CC) scoring standard has become not only an objective quantitative index and independent prognostic factor for evaluating the effect of tumor resection but also an important part of the standardized cytoreductive surgery (CRS). The specific evaluation is as follows:
- CC-0, no residual tumor nodule after cytoreduction;
- CC-1: residual tumor diameter < 2.5 mm;
- CC-2: residual tumor diameter 2.5 mm-2.5 cm; and
- CC-3: residual tumor diameter > 2.5 cm or the residual tumor cannot be removed or palliatively removed.
Involvement of the small bowel impacts the Peritoneal Cancer Index (PCI) score and can suggest a bad prognosis. The following are the usual surgical sites used for preoperative determination of the extent of the disease for exclusion from complete cytoreductive surgery 64:
- Massive mesenteric root infiltration not amenable to complete cytoreduction
- Significant pancreatic capsule infiltration or pancreatic involvement requiring major resection not feasibly or amenable to complete surgical cytoreduction
- More than one-third small bowel length involvement requiring resection
- Extensive hepatic metastasis
Some surgeons advocate the use of peritoneal surface disease severity score (PSDSS) for the early preoperative assessment of the prognosis based on the symptoms, Peritoneal Cancer Index (PCI), and Primary tumor histology. However, extensive study results are needed to implement it on a regular practice 64.
Figure 8. Peritoneal Cancer Index (PCI) scoring system (Sugarbaker score)
Footnotes: Sugarbaker’s Peritoneal Cancer Index (PCI) is a numerical score that is determined during surgery and it is used to quantify the extent of cancer spread throughout the abdomen and pelvis. The abdominal cavity is divided into 13 regions (from 0 to 12). A score of 0 to 3 is assigned to each of these regions depending on tumor size found there (0: no lesion; 1: lesion ≤ 0.5 cm; 2: lesions ≤ 5 cm; 3: lesions > 5 cm). The sum of these scores produces the PCI, ranging from 1 to 39.
[Source 58 ]Figure 9. Completeness of cytoreduction score (scoring criteria of the postoperative completeness of cytoreduction (CC) score)
[Source 25 ]Histopathology
Macroscopic examination often reveals abundant gelatinous pelvic or abdominal mucin or mucinous ascites accompanied by cystic epithelial implants on peritoneal surfaces. These lesions vary in size from a few mm to a few centimeters. A large ‘omental cake’ is also frequently found 65.
Multiple histological grading systems have been proposed for pseudomyxoma peritonei. Ronnett et al 32 first divided pseudomyxoma peritonei into two groups: disseminated peritoneal adenomucinosis (DPAM) and peritoneal mucinous carcinomatosis (PMCA). Disseminated peritoneal adenomucinosis (DPAM) is characterized by abundant mucus containing scanty mucinous epithelial cells with minimal cytological atypia and mitotic activity, while peritoneal mucinous carcinomatosis (PMCA) is featured by more abundant mucinous epithelial cells with high-grade cytological atypia and mitotic activity. In 2010, the World Health Organization (WHO) further refined the grading system 35, 7.
- Acellular mucin: Mucin within the peritoneal cavity without neoplastic epithelial cells.
- Low-grade mucinous carcinoma peritonei (synonymous with DPAM) presents as mucin pools with low cellularity (less than 10%), bland cytology and non-stratified cuboidal epithelium. Tumor cells are arranged in strips or gland-like structures. Infiltrative growth is not present.
- High-grade mucinous carcinoma peritonei (synonymous with PMCA): Mucin pools with high cellularity, moderate/severe cytological atypia, numerous mitoses, and cribriform growth pattern.
- Destructive infiltrative invasion of underlying organs is often present.
- High-grade mucinous carcinoma peritonei with signet ring cells: Any lesion with a component of signet ring cells, classified separately because of their worse prognosis 66, 67.
Immunohistochemistry
Researchers have suggested MUC 2 over-expression as a molecular marker for pseudomyxoma peritonei of intestinal origin 68. Appendiceal tumors also express CK20, CEA, and CDX2, and are usually negative for CK7 and CA 125 7. There are also reports of loss of protein expression of the repair genes MLH1 and PMS2 69.
Pseudomyxoma peritonei treatment
Your treatment depends on several factors. These include where the cancer is, and your general health. The main treatments for pseudomyxoma peritonei (PMP) involves surgically removing as much of the tumor as possible, followed by chemotherapy delivered directly into your abdomen during the surgery called cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) 25, 20, 21.
You might not start treatment straight away. Your doctor closely monitors your cancer in case you need treatment in the future. This is called watch and wait.
If you need treatment you might have:
- Cytoreductive surgery (CRS) also known as debulking surgery: Cytoreductive surgery (CRS) is a surgical procedure to remove cancerous tumors from the abdomen and pelvic cavity that have spread to the peritoneum. Cytoreductive surgery (CRS) is often performed alongside heated intraperitoneal chemotherapy (HIPEC), where hot chemotherapy is circulated in the abdomen to kill any remaining microscopic cancer cells. The goal of cytoreductive surgery (CRS) is to completely remove all visible tumors, and the surgery may involve removing other affected organs as well.
- Hyperthermic intraperitoneal chemotherapy (HIPEC): Hyperthermic intraperitoneal chemotherapy (HIPEC) is a two-stage cancer treatment for advanced abdominal cancers that combines surgery to remove tumors with the administration of heated chemotherapy drugs directly into your abdominal cavity. After visible tumors are surgically removed, a heated chemotherapy solution is circulated throughout your abdomen for a short period, typically 90 minutes to two hours to kill any remaining microscopic cancer cells, reducing the risk of recurrence.
Your surgeon will only do cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) if the surgery removes all or almost all of the cancerous tissue or mucin. Your surgeon will make that decision during your surgery.
Other types of surgery treat pseudomyxoma peritonei may include:
- Bowel resection if you have mucin in your small or large intestine
- Cholecystectomy to remove your gallbladder.
- Hysterectomy with bilateral salpingo-oophorectomy to remove your fallopian tubes, ovaries and uterus.
- Liver capsulectomy to remove part of the surface of your liver.
- Omenectomy, which is removing the layer of fat that covers the front of your belly.
- Peritonectomy to remove your peritoneum. This is the membrane that lines the inside of your abdomen and pelvis.
- Splenectomy to remove your spleen.
Other treatments may include active surveillance, radiation therapy, systemic chemotherapy, or palliative care.
- Your doctor may recommend active surveillance if you have early or slow-growing pseudomyxoma peritonei and the risks of surgery outweigh the current risks of the disease. In active surveillance, your doctor will make regular checks on your overall health. Your doctor may do imaging tests and blood tests to see if pseudomyxoma peritonei is spreading.
- Palliative care. If you can’t have surgery, your doctor may recommend treatments like chemotherapy or radiation therapy that will slow down pseudomyxoma peritonei and ease your symptoms.
Doctor are investigating small bowel transplantation as treatment when you have pseudomyxoma peritonei in your small intestine.
Regular follow-up is necessary due to the risk of recurrence, even after successful treatment. In some cases, your doctor can treat and often cure pseudomyxoma peritonei. In some cases, surgery to remove all the cancer cells in your peritoneal cavity successfully controls pseudomyxoma peritonei for a long time 22, 23, 24. Other treatments may help you to live longer with it.
Watch and wait
Watch and wait can also sometimes be called active surveillance. Your doctor might decide to closely monitor your pseudomyxoma peritonei cancer if it’s small and slow growing and you don’t currently need treatment. Your doctor will check up on you regularly. Your doctor will order regular blood tests and scans.
Cytoreductive surgery (CRS)
Cytoreductive surgery (CRS) also known as debulking surgery is a surgical procedure to remove all visible cancerous tumors from the abdomen and pelvic cavity that have spread to the peritoneum (the lining of the abdominal cavity), often involving peritonectomy and resection of involved visceral structures (eg, the omentum, spleen, segments of small or large bowel) and occasionally other organs (eg, liver wedge resections, hysterectomy, bilateral salpingo-oophorectomy in select cases) 52, 70. The cornerstone of pseudomyxoma peritonei treatment is carrying out cytoreductive surgery (CRS) to remove the tumor lesions visible to the naked eye as much as possible 3. With the development of surgical techniques and the accumulation of experience, the current cytoreductive surgery (CRS) strategy is characterized by a set of standardized procedures 71, 72. First, after opening the peritoneum, a comprehensive exploration of the abdominal and pelvic cavity is needed, and the peritoneal cancer index (PCI) score is determined 73. Then, the left upper peritoneum, right upper peritoneum, parietal anterior peritoneum, greater omentum, lesser omentum, spleen, and pelvic peritoneum are excised, and the stomach, small intestine, colon, and other widely implanted organs are excised as appropriate according to the individual situation of the patient 3.
Cytoreductive surgery (CRS) goal is to achieve a completeness of cytoreduction (CC) score of 0 or 1 (i.e, no visible disease or residual nodules <2.5 mm) because intraperitoneal chemotherapy penetrates only a few millimeters into tissue 52. Achieving a completeness of cytoreduction (CC) score of 0 or 1 resection is strongly associated with improved survival 74, 75, 76.
Finally, the completeness of the cytoreduction (CC) score is determined after surgery, according to the degree of cytoreductive surgery (CRS) 77. Cytoreductive surgery (CRS) reaching CC0 or CC1 is called complete cytoreduction surgery (CCRS). The reduction degree of tumor lesions is significantly related to the prognosis of pseudomyxoma peritonei patients, and even high-grade pseudomyxoma peritonei patients can obtain better overall survival [overall survival (OS) is the length of time from either the date of diagnosis or the start of treatment for a disease, such as cancer, that patients diagnosed with the disease are still alive] and disease free survival [disease free survival (DFS) is the length of time after primary cancer treatment that a patient lives without any signs or symptoms of the cancer] after reaching complete cytoreduction surgery (CCRS) 78, 79. Therefore, for the vast majority of pseudomyxoma peritonei patients, complete cytoreduction surgery (CCRS) should be achieved in surgery as much as possible, but some contraindications should be considered (see Table 3) 80.
Patient selection for cytoreductive surgery (CRS) depends on tumor and patient-related factors. Tumor burden is classically quantified using the Sugarbaker’s Peritoneal Cancer Index (PCI). Most centers discourage cytoreductive surgery (CRS) if the Peritoneal Cancer Index (PCI) substantially exceeds 20 for high-grade appendiceal or colorectal cancers, due to the lower likelihood of complete cytoreduction and diminished survival benefits. Favorable histopathologic subtypes (e.g, low-grade mucinous appendiceal neoplasms and epithelial mesothelioma) are typically more manageable to cytoreductive surgery (CRS) than high-grade or signet ring cell carcinomas 52. Additional considerations include performance status (ECOG/WHO) and comorbidities, as well as prior response to systemic therapies 52.
Cytoreductive surgery (CRS) is often combined with hyperthermic intraperitoneal chemotherapy (HIPEC), where a heated chemotherapy solution is circulated in the abdomen after the surgery to kill any remaining microscopic cancer cells 70. The goal of cytoreductive surgery (CRS) is to completely remove all visible tumors, and the surgery may involve removing other affected organs as well. Cytoreductive surgery (CRS) is a major operation used to manage selected patients with metastatic cancers from the peritoneum, such as those originating from colorectal, appendiceal, ovarian, or stomach cancers. The procedure can take several hours (4 to 10 hours). Patients often have a hospital stay of about 7 to 10 days. Recovery is a major process, and some patients may experience long-term gastrointestinal issues, fatigue, or depression.
Cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) are complex procedures that require extensive support and and should only be done in centers with experience. Careful patient selection is crucial to achieve good outcomes. It’s best for people who are healthy enough for surgery and whose cancer can be mostly or completely removed.
How cytoreductive surgery (CRS) works:
- Cytoreductive surgery (CRS): The surgeon removes all visible tumors and affected tissues, such as parts of the bowel, gallbladder, or spleen, to reduce the amount of cancer in your body.
- Hyperthermic intraperitoneal chemotherapy (HIPEC): After the visible tumors are removed, the abdominal cavity is filled with a heated chemotherapy solution for a period to kill any remaining microscopic cancer cells. The heat helps to increase the chemotherapy’s effectiveness and can deliver a higher local dose than systemic chemotherapy.
Cytoreductive surgery (CRS) benefits:
- Cytoreductive surgery (CRS) is a major operation that removes all visible cancer from the peritoneum and other organs in the abdomen.
- Can improve quality of life and increase survival rates for selected patients.
- Helps relieve symptoms associated with the cancer.
- May reduce tumor recurrence rates.
Cytoreductive surgery (CRS) risks:
Cytoreductive surgery (CRS) risks are similar to other major abdominal surgeries and can include bleeding, wound infection, blood clots, breathing difficulties, bowel obstruction, peritonitis and potential kidney failure. There is also a risk of allergic reaction to the chemotherapy drug.
Contraindications for cytoreductive surgery (CRS) include a Peritoneal Cancer Index (PCI) score >17 in colorectal-associated peritoneal cancer and Peritoneal Cancer Index (PCI) >12 in gastric cancer 81. Tumor involvement of critical anatomic sites in the abdomen and multiple extra-abdominal metastatic lesions also preclude cytoreductive surgery (CRS) 82.
Table 3. Cytoreductive surgery contraindications (Peritoneal Surface Oncology Group International expert consensus on contraindications to CCRS/HIPEC)
| Contraindication | Description | Peritoneal Surface Oncology Group International (PSOGI) Expert Consensus Rate |
|---|---|---|
| Absolute | Retraction due to mesenteric involvement | 64.30% |
| Extensive involvement of the small bowel serosa, unable to preserve 1.5–2 m of small bowel without tumor invasion | 58.90% | |
| Relative | Peritoneal cancer index ()PCI > 20 with aggressive histology (e.g., mucinous adenocarcinoma with signet ring cells, goblet cell carcinoid, and high-grade pseudomyxoma peritonei with signet ring cells) | 87.50% |
| Massive involvement of the liver hilum | 87.50% | |
| Age > 75 years old | 85.70% | |
| Extensive Infiltration of the pancreatic surface | 82.10% | |
| Requires complete gastrectomy | 80.40% | |
| Ureteral obstruction | 64.30% |
Abbreviations: PSOGI = Peritoneal Surface Oncology Group International; CCRS = complete cytoreduction surgery (cytoreductive to CC0 or CC1); HIPEC = hyperthermic intraperitoneal chemotherapy; PCI, peritoneal cancer index.
[Source 3 ]Hyperthermic intraperitoneal chemotherapy (HIPEC)
Hyperthermic intraperitoneal chemotherapy (HIPEC) is a surgical cancer treatment that involves removing tumors from the abdominal cavity and then flooding the area with heated chemotherapy drugs to kill any remaining cancer cells 70, 83. Hyperthermic intraperitoneal chemotherapy (HIPEC) is a two-stage procedure performed after the surgical removal of visible tumors, and it aims to reduce the risk of cancer recurrence by treating microscopic cancer cells within the peritoneal cavity 70. Hyperthermic intraperitoneal chemotherapy (HIPEC) is a major procedure with risks, and eligibility depends on factors like cancer type, spread, and overall health. Hyperthermic intraperitoneal chemotherapy (HIPEC) is used for certain advanced abdominal cancers, such as those of the appendix, colon, stomach, pancreas, ovaries, peritoneum and mesothelioma.
Hyperthermic intraperitoneal chemotherapy (HIPEC) is typically combined with cytoreductive surgery (CRS) 70. After the cancer is removed during cytoreductive surgery (CRS), the abdominal cavity is bathed with heated chemotherapy to target any remaining microscopic cancer cells. Heat (40 °C to 43 °C) enhances the cytotoxicity of chemotherapy and improves tissue penetration, it may increase tumor destruction in regions inaccessible to surgical resection. This combined approach, often referred to as CRS-HIPEC or HIPEC surgery, allows for higher medicine concentrations to reach cancer in the peritoneum. This can lessen the typical side effects people often have with systemic chemotherapy due to less absorption in the bloodstream 52.
Hyperthermic intraperitoneal chemotherapy (HIPEC) has unique advantages as an effective treatment for pseudomyxoma peritonei patients 3.
- Regarding pharmacokinetics, because the peritoneal–plasma barrier restricts the absorption of macromolecular chemotherapeutic drugs into the blood, intraperitoneal administration can often involve maintaining a high concentration of local drugs in the abdomen, while keeping the systemic drug level low. Furthermore, the concentration of intraperitoneal administration can be 1000 times higher than that of intravenous administration 84, 85.
- In terms of the thermal effect, a large number of studies have shown that, in the range of 41°C to 43 °C, the thermal effect has multiple inhibitory effects on tumor cells, while normal tissue cells are less affected 86, 87. This is related to an imbalance of the autostabilization mechanism caused by the increase in lysosome number and lysosomal enzyme activity in tumor cells, as well as the insufficient nutrient supply caused by reduced or even complete interruption of blood flow 88, 89.
- Regarding the synergistic effect, additive interaction exists between the thermal effect and the cytotoxicity of drugs, as has been confirmed in multiple studies 87, 90. This may be related to the increase in membrane permeability and the change in drug pharmacokinetics due to the thermal effect 91.
- However, HIPEC also has an obvious disadvantage: insufficient penetration depth (3mm to 5 mm) 92, 93. In pseudomyxoma peritonei patients, the tumor load in the abdominal cavity is large. If the residual tumor tissue is not controlled within the penetration range of HIPEC by CRS, it will be difficult to effectively kill tumor cells. This is also the reason why 2.5 mm is used as the threshold to distinguish complete cytoreduction surgery (CCRS). Only when complete cytoreduction surgery (CCRS) is achieved can HIPEC be used to obtain the best effect.
Currently, HIPEC protocols used to treat pseudomyxoma peritonei are based mainly on oxaliplatin or mitomycin C 80, 94, 95, 96. Due to the lack of sufficient prospective evidence, there has been controversy regarding the use of oxaliplatin or mitomycin C, and no international consensus has been formed. To compare the true effects and toxic side effects of oxaliplatin or mitomycin C, Levine et al. 97 conducted a multicenter randomized controlled trial in 2018 in patients with appendiceal-derived pseudomyxoma peritonei. The results showed that there was no significant difference in the incidence of platelets and leukopenia between the oxaliplatin group and mitomycin C group, and the 3-year overall survival (86.9% vs. 83.7%) [overall survival (OS) is the length of time from either the date of diagnosis or the start of treatment for a disease, such as cancer, that patients diagnosed with the disease are still alive] and disease free survival (64.8% vs. 66.8%) [disease free survival (DFS) is the length of time after primary cancer treatment that a patient lives without any signs or symptoms of the cancer] were similar. However, the oxaliplatin group reported better emotional and physical well-being. In 2020, Peritoneal Surface Oncology Group International (PSOGI) also launched expert voting for different HIPEC regimens and the results are shown in Table 4 80. However, none of these approaches reached the expert consensus threshold (>50%). It is expected that more clinical trials will be conducted in the future to reach a consensus on this issue.
How hyperthermic intraperitoneal chemotherapy (HIPEC) works:
- Surgical tumor removal: The first step is a surgery called cytoreductive surgery, where visible tumors are surgically removed from the abdomen.
- Heated chemotherapy: A heated chemotherapy solution is then pumped directly into the abdominal cavity through catheters.
- Circulation and temperature: The heated chemotherapy solution is circulated for about 90 minutes to 2 hours, often with the patient being physically rocked to ensure the drug reaches all areas of the cavity. The temperature is typically around 108 °F (42 °C).
- Draining: The solution is then drained from the abdomen, and the incisions are closed.
Hyperthermic intraperitoneal chemotherapy (HIPEC) has several advantages over standard chemotherapy:
- It is a single treatment done in the operating room, instead of multiple treatments over several weeks
- 90% of the drug stays within the abdominal cavity, decreasing toxic effects on the rest of the body
- It allows for a more intense dose of chemotherapy.
Hyperthermic intraperitoneal chemotherapy (HIPEC) provides a high, targeted dose of chemotherapy directly to cancer cells in your abdomen. Hyperthermic intraperitoneal chemotherapy (HIPEC) may offer a survival benefit for certain patients and reduces the risk of cancer recurrence.
Hyperthermic intraperitoneal chemotherapy (HIPEC) side effects:
Side effects of hyperthermic intraperitoneal chemotherapy (HIPEC) are similar to those experienced after major surgery and standard chemotherapy, and can include:
- Nausea and vomiting
- Electrolyte abnormalities
- Brief worsening of kidney function
Complication related to hyperthermic intraperitoneal chemotherapy (HIPEC):
- Oxaliplatin is used with dextrose solutions, so it could potentially contribute to postoperative acidosis and hyperglycemia
- Mitomycin C can cause neutropenia in about one-third of patients
- Other gastrointestinal side effects.
Table 4. Commonly used HIPEC regimens for pseudomyxoma peritonei
| HIPEC Regimens (Peritoneal Surface Oncology Group International Expert Consensus Rate) | Dose | Time | Intraperitoneal Component | Intravenous Component |
|---|---|---|---|---|
| Dutch High-Dose Mitomycin C Regimen: “Triple Dosing Regimen” (42.9%) 98 | 35 mg/m² | 90 minutes | Mitomycin C was added to 1.5% peritoneal dialysis solution at an initial dose of 17.5 mg/m², followed by 8.8 mg/m² after 30 minutes and 8.8 mg/m² after 60 minutes | NA |
| Glehen Medium-Dose Oxaliplatin Regimen (28.6%) 95 | 360 mg/m² | 30 minutes | Add oxaliplatin to 2 L/m² 5% dextrose solution and maintain intraperitoneal chemotherapy for 30 minutes | 1 hour before intraperitoneal chemotherapy, 5-fluorouracil 400 mg/m² and leucovorin 20 mg/m² were separately added to 250 mL of normal saline for rapid intravenous infusion |
| American Society of Peritoneal Surface Malignancy Low-Dose Mitomycin C Regimen: “Concentration-Based Regimen” (14.3%) 96 | 40 mg/3L | 90 minutes | Add mitomycin C to 1.5% peritoneal dialysis solution, the initial dose is 30 mg/3 L, and then add 10 mg after 60 minutes | Not applicable |
| PMI Basingstoke IP Chemotherapy Regimen: “Body Surface Area-Based” (10.7%) 80 | 10 mg/m² | 60 minutes | Add mitomycin C to 0.9% sodium chloride solution at 42 °C. Reduce the dose by 33% for obesity (BMI > 40), severe abdominal distension, and severe chemotherapy in the past 3 months | Not applicable |
| Elias High-Dose Oxaliplatin Regimen (8.9%) 94 | 460 mg/m² | 30 minutes | Add oxaliplatin to 2 L/m² 5% dextrose solution and maintain intraperitoneal chemotherapy for 30 minutes | 1 h before intraperitoneal chemotherapy, 5-fluorouracil 400 mg/m2 and leucovorin 20 mg/m2 were separately added to 250 mL of normal saline for rapid intravenous infusion |
| Wake Forest University Oxaliplatin Regimen (1.8%) 95 | 200 mg/m² | 120 minutes | Add oxaliplatin to 5% dextrose solution and maintain intraperitoneal chemotherapy for 120 minutes | Not applicable |
| Sugarbaker Regimen (1.8%) 99 | 15 mg/m² | 90 minutes | Add 15 mg/m² of mitomycin C and doxorubicin to 2 L 1.5% dextrose peritoneal dialysis solution and maintain intraperitoneal chemotherapy for 90 minutes | At the same time of intraperitoneal chemotherapy, 5-fluorouracil 400 mg/m² and leucovorin 20 mg/m² were separately added to 250 mL of normal saline for rapid intravenous infusion |
Abbreviations: PSOGI = Peritoneal Surface Oncology Group International; HIPEC = hyperthermic intraperitoneal chemotherapy; NA = not applicable.
[Source 3 ]How is HIPEC performed?
Generally, two methods for HIPEC are described: open abdomen technique and closed abdomen technique.
Open abdomen technique as described by Sugarbaker, at the end of the surgical cytoreduction (CRS), a Tenckhoff catheter and four closed suction drains are sutured to the skin and placed through the abdominal wall 70. The temperature probes are attached to the skin edge for intraperitoneal temperature monitoring. The skin edges at the level of the abdominal incisions are suspended until the Thompson self-retaining retractor by a monofilament to maintain open space in the abdominal cavity. For preventing the leakage of the chemotherapy solution, a plastic sheet is inserted into this suture. Continuous manipulation by the surgeon of the perfusion allows the uniform exposure of all anatomical structures to heat and chemotherapy during the procedure. A pumping system injects the chemotherapy infusion into the abdomen through the Tenckhoff catheter and pulls it through the drains with a constant flow. The heat exchanger keeps the intraperitoneal fluid temperature at 41 to 43 degrees, the drug is then administered in the circuit, and the timer for the perfusion is started. The duration varies from 30 min to 1 hour, depending on the type of cancer. A disadvantage of the open technique is the heat dissipation, making it more difficult to have a hyperthermic state 70.
Closed abdomen technique: Thermal catheters and probes are placed in the same way, but the skin edges of the laparotomy are tightly sutured to allow perfusion in a closed circuit 70. The abdominal wall is shaken manually by the surgeon during the infusion for uniform heat distribution. The volume of perfusate is greater in this technique to establish the circuit, and higher abdominal pressure is obtained during the perfusion, which facilitates tissue penetration of the drug. After infusion, the abdomen is reopened to remove the perfusate and the preparation of the anastomosis. Closed abdomen technique allows maintaining rapid attainment of hyperthermia because there is minimal heat loss 70.
HIPEC Indications
In addition to the surgical operability criteria and the comorbidity factors taking into account American Society of Anesthesiologists (ASA) score <3, liver and kidney function and the cumulative dose of chemotherapy treatments must be considered 70.
- In gastric adenocarcinoma, peritoneal carcinomatosis is very common; even after radical surgery, cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) seems to delay oncological progression without affecting long-term survival 70. The procedure remains in the experimental field and should be reserved for younger patients under the age of 60, who are active, with a peritoneal cancer index (PCI) <10 and small nodules, without ascites, extraperitoneal metastasis, or hepatic invasion that has responded well to neoadjuvant chemotherapy 100, 101.
- According to a review of the literature and meta-analysis, cytoreduction surgery with HIPEC prolongs overall survival and up to eight years after the gesture in ovarian neoplasia with peritoneal carcinosis 102.
- In colorectal cancer: the peritoneum is the second site most common metastatic after the liver, about 7 to 15% of colorectal cancer patients have synchronous peritoneal metastases, and 4 to 19% will develop one afterward.
An improvement in patient prognosis has been noticed since the introduction of cytoreduction surgery with HIPEC and modern chemotherapy regimens (capecitabine, oxaliplatin, irinotecan) combined with immunotherapy (bevacizumab, cetuximab, panitumumab) 103, 104.
- Peritoneal pseudomyxoma and mucinous tumors of the appendix: Cytoreduction surgery (CRS) with HIPEC has been well established as the only treatment for mucinous tumors with peritoneal dissemination, based on retrospective and comparative studies (Chua et al.) 62.
- Mesothelioma: Mesothelioma is a rare tumor of peritoneal origin. Cytoreduction surgery combined with HIPEC improves mean survival by up to 4 years but with repeated procedures 105.
HIPEC Contraindications
It has been shown by several studies that when the surgical cytoreduction (CRS) is incomplete, there is no longer any benefit from HIPEC on survival 106. Medically, in addition to comorbid factors such as severe cardiovascular disorders and serious lung disease, liver and kidney failure may complicate the procedure and should contraindicate the operation 70.
Allergy to cytotoxic agents is a formal contraindication to the HIPEC 70.
The threshold value of the peritoneal cancer index (PCI) score to perform a HIPEC in the peritoneal carcinomatosis of colorectal origin is 20 70. The survival overall at five years is virtually zero for patients with a peritoneal cancer index (PCI) greater than 20 107.
Surgically and in addition to a too high peritoneal cancer index (PCI) score, the invasion of some surgical sites also contraindicates the HIPEC 70. These are the root of the mesentery, the hepatic pedicle, retroperitoneum, and the bladder. Impairment of small intestinal function that potentially could induce a short bowel syndrome is also a contraindication 70.
Ultimately, surgical cytoreduction (CRS) will remove as many tumors as possible, while the associated HIPEC will make it possible to “sterilize” the tumor residues not visible to the naked eye 70. The purpose of cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) is to obtain a high local concentration of chemotherapy and a low systemic concentration 100.
HIPEC Complications
According to recent studies (meta-analysis), cytoreductive surgery (CRS) and HIPEC are associated with morbidity and mortality of approximately 33% and 2.8%, respectively 108. It should be remembered that heavy and complex surgical procedures likely lead to increased toxicity of HIPEC through changes in pharmacokinetics, protein losses, hepatic and renal metabolic restrictions, and stress-related bone marrow suppression 109. According to the literature, this increased toxicity is manifested by hematotoxicity and nephrotoxicity with respective frequencies of 5.6% and 1.7% 108.
Particular complications associated with the procedure are prolonged intestinal atony, increased fluid movement over the abdomen, delayed wound healing, and prolonged hospitalization.
Cases of intrathoracic chemotherapy have been described in cases of major surgery with diaphragm perforation. Have been identified as risk factors for this procedure’s serious complications: the general condition of the patient, age, the extent of peritoneal carcinoma, duration of the intervention, number of peritonectomy procedures, number of anastomoses, the quality of cytoreduction, and the dosage of intraperitoneal chemotherapy.
Other complications have been observed which are not related only to the HIPEC but are well associated with cytoreduction: Pancreatitis, fistula, Pulmonary embolism, and thrombosis.
Maximum Tumor Debulking-HIPEC
Maximum tumor debulking with HIPEC is a two-part surgical procedure where visible tumors are meticulously removed from your abdomen (complete cytoreduction surgery [CCRS]), followed by circulating heated chemotherapy directly inside the abdominal cavity (HIPEC) 82. The goal of the surgery is to leave no visible tumor or only very small residual tumors (completeness of cytoreduction (CC) score of 1), a size less than 2.5 mm, which is the limit for which HIPEC chemotherapy is effective. This combined approach is used for abdominal cancers that have spread to the peritoneum to improve outcomes, as systemic chemotherapy often cannot reach these tumors effectively.
Part 1: Maximum tumor debulking (cytoreduction surgery [CRS])
- Procedure: Surgeons meticulously remove all visible tumors and affected organs from the abdominal cavity. This is also known as cytoreductive surgery (CRS).
- Goal: To achieve a “complete cytoreduction,” leaving no visible tumors (completeness of cytoreduction (CC) score of 0) or only tiny tumor nodules less than 2.5 mm in size (completeness of cytoreduction (CC) score of 1).
- Rationale: Chemotherapy drugs in HIPEC can only penetrate a few millimeters into the tissue, so any larger residual tumors would not be adequately treated.
Part 2: HIPEC (hyperthermic intraperitoneal chemotherapy)
- Procedure: After the debulking surgery is complete, chemotherapy drugs are heated and pumped directly into the abdominal cavity for a specific period, typically 30 to 120 minutes.
- Goal: To kill any remaining microscopic cancer cells that were not visible during surgery.
- Rationale:
- Direct effect: The chemotherapy is applied directly to the surfaces where cancer cells may remain.
- Hyperthermia: Heating the chemotherapy drugs increases their effectiveness and allows them to penetrate the tissue better.
- Immune response: The heat can also help trigger the body’s immune response against cancer cells.
Although CCRS-HIPEC is an ideal treatment for pseudomyxoma peritonei, it is not always possible to achieve complete cytoreduction surgery (CCRS) based on the individual situation of each patient, especially for patients with relapse, extensive small intestine involvement, and poor underlying conditions 3. For them, achieving complete cytoreduction surgery (CCRS) at all costs may result in poorer quality of life and more serious surgical complications 110. However, tumor reduction without surgery relying only on relatively insensitive chemotherapy and other measures cannot achieve the effect of alleviating patients’ symptoms 111, 112, 113, 114. The idea of complete cytoreduction surgery (CCRS) provides a new choice for such patients. It aims to reduce the tumor load in the abdominal cavity as much as possible, while solving the main symptoms of patients, such as obstruction, without sacrificing the vast majority of abdominal organs and greatly increasing the probability of intestinal fistula and other serious complications in exchange for the complete reduction of tumor cells 80, 115. Delhorme et al. 115 conducted a retrospective study of 39 patients who underwent Maximum Tumor Debulking-HIPEC and found that the median overall survival [overall survival (OS) is the length of time from either the date of diagnosis or the start of treatment for a disease, such as cancer, that patients diagnosed with the disease are still alive] and disease free survival [disease free survival (DFS) is the length of time after primary cancer treatment that a patient lives without any signs or symptoms of the cancer] reached significance at 55.5 months and 20 months, respectively. Alves et al 116 reported that 20 patients who received Maximum Tumor Debulking-HIPEC showed significant improvements in appetite, mood, and health-related quality of life (HRQL) 1 year after surgery. In a vote of experts initiated by Peritoneal Surface Oncology Group International (PSOGI) in 2020, 98.2% of them recommended that, for patients unable to undergo complete cytoreduction surgery (CCRS), Maximum Tumor Debulking should be performed in experienced treatment centers, and 60.7% recommended routine HIPEC after Maximum Tumor Debulking 80. However, there is still a lack of prospective research evidence to prove that Maximum Tumor Debulking is superior to complete cytoreduction surgery (CCRS) for this type of pseudomyxoma peritonei patient, and the criteria for patient selection for Maximum Tumor Debulking need to be further clarified.
Early Postoperative Intraperitoneal Chemotherapy (EPIC)
Early Postoperative Intraperitoneal Chemotherapy (EPIC) is a cancer treatment that uses chemotherapy drugs delivered directly into the abdomen (intraperitoneal) in the days following surgery 117, 118, 119. Early Postoperative Intraperitoneal Chemotherapy (EPIC) offers a targeted approach to delivering chemotherapy directly to the peritoneal cavity, potentially enhancing therapeutic efficacy while minimizing systemic side effects. Early Postoperative Intraperitoneal Chemotherapy (EPIC) is often combined with cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC). Early Postoperative Intraperitoneal Chemotherapy (EPIC) provides a second opportunity to target and destroy cancer cells in the peritoneal cavity, especially those in areas that become inaccessible after the initial surgery. While some studies suggest potential survival benefits, especially for certain cancers, the use of EPIC is controversial due to increased risks of morbidity and longer hospital stays, leading many centers to use it less frequently today.
Early Postoperative Intraperitoneal Chemotherapy (EPIC) is initiated on the first postoperative day and then administered continuously for 5 to 7 days. During the procedure, a solution containing chemotherapy drugs is introduced into the peritoneal cavity, where it bathes the mesothelium for a duration between 4 and 24 hours. Subsequently, the solution is drained over an hour and then readministered 117, 118, 119. Before starting EPIC therapy, it is essential to ensure your postoperative status is stable, including a normal white blood cell count and tolerance to treatment. Depending on your condition, EPIC therapy may be delayed until the second postoperative day. A catheter is secured with sutures to facilitate drug delivery, and multiple closed suction drains are placed for drainage. The chemotherapy drugs employed in EPIC are typically cell-specific, in contrast to the cell cycle nonspecific drugs used in other intraperitoneal chemotherapy regimens such as HIPEC 120.
Generally, a cycle of 24 hour lasts for 5 days (1 to 5 days after surgery), and 5-fluorouracil is commonly used for chemotherapy 121. On the basis of low tumor load after cytoreductive surgery (CRS), long-term contact between chemotherapy drugs and abdominal residual lesions would theoretically have greater advantages 71. Chua et al 121 retrospectively analyzed the prognosis of 108 patients with low-grade malignant pseudomyxoma peritonei who received CRS-HIPEC/EPIC (n = 21) or CRS-HIPEC-EPIC (n = 87). The 5-year survival rate was higher in the CRS-HIPEC-EPIC group than in the CRS-HIPEC/EPIC group (86% vs. 64%) 121. Huang et al 122 found that EPIC combined with CRS-HIPEC was an independent influencing factor for improved prognosis in patients with both low- and high-grade pseudomyxoma peritonei. There was no significant difference with the CRS-HIPEC group in terms of total length of hospital stay, in-hospital mortality, and incidence of serious complications. However, other relevant studies have reached completely different conclusions. The combined use of EPIC has been shown to not only not significantly improve overall survival [overall survival (OS) is the length of time from either the date of diagnosis or the start of treatment for a disease, such as cancer, that patients diagnosed with the disease are still alive] and disease free survival [disease free survival (DFS) is the length of time after primary cancer treatment that a patient lives without any signs or symptoms of the cancer] but also increase the incidence of grade III/IV complications 123, 124. Since the above studies were retrospective, prospective clinical trials with large samples are needed to draw conclusions and resolve these contradictory findings. In 2020, Peritoneal Surface Oncology Group International (PSOGI) launched an expert vote on this issue, and 60.7% of the experts supported EPIC after CRS-HIPEC, while 39.3% were against it 80.
Hyperthermic Intrathoracic Chemotherapy (HITHOC)
Invasive changes in the pleura occur in approximately 5.4% of pseudomyxoma peritonei patients and are often associated with poor prognosis 125. The reason for this phenomenon may be related to the direct invasion of subphrenic lesions, lymphatic vessel metastasis of thoraco-abdominal communication, and implantation caused by accidental injury of the diaphragm during surgery 125, 126, 127.
Hyperthermic Intrathoracic Chemotherapy (HITHOC) is a procedure where heated chemotherapy is delivered directly into the chest cavity after a surgical removal of a tumor. Hyperthermic Intrathoracic Chemotherapy (HITHOC) is a component of a multimodal treatment strategy for certain pleural malignancies, such as malignant pleural mesothelioma, and is designed to target and kill remaining cancer cells, reduce recurrence, and improve survival rates compared to surgery alone.
How Hyperthermic Intrathoracic Chemotherapy (HITHOC) works:
- Cytoreductive surgery (CRS): A surgeon first removes as much of the tumor as possible from the chest cavity, which includes the lining of the lungs, chest wall, and diaphragm.
- Intraoperative chemotherapy: After surgery, heated chemotherapy drugs, often including cisplatin, are introduced directly into the thoracic cavity.
- Hyperthermia: The heat applied to the chemotherapy enhances its ability to penetrate and destroy cancer cells while potentially sparing healthy tissue from some side effects.
- Drainage: The heated chemotherapy is then drained from the chest.
Hyperthermic Intrathoracic Chemotherapy (HITHOC) Benefits:
- Improved survival rates: Studies show Hyperthermic Intrathoracic Chemotherapy (HITOC) combined with surgery can lead to better survival rates for patients with pleural mesothelioma.
- Lower recurrence rates: The targeted delivery of chemotherapy helps to control primary tumors and reduce the likelihood of cancer recurrence.
- Higher concentration of chemotherapy: The direct, localized application results in higher concentrations of the drugs within the chest cavity and lower systemic toxicity compared to traditional chemotherapy.
The basic process of Hyperthermic Intrathoracic Chemotherapy (HITHOC) is similar to that of HIPEC, and keeping the diaphragm open during this process enables both HITHOC and HIPEC to be performed simultaneously 128. Several studies have shown that Hyperthermic Intrathoracic Chemotherapy (HITHOC) may be a potential treatment for patients with pleural involvement of pseudomyxoma peritonei 125, 127, 128, 129. However, Ashraf-Kashani et al 130 found that Hyperthermic Intrathoracic Chemotherapy (HITHOC) could lead to hemodynamic changes in patients. At present, most of these studies are presented in the form of case reports, the level of evidence is weak, and the safety of this approach needs to be further investigated.
Neoadjuvant Chemotherapy
Neoadjuvant chemotherapy is a cancer treatment given before surgery to shrink the tumor, making it easier to remove. Neoadjuvant chemotherapy can lead to less extensive surgery, better cosmetic results, and potentially improved long-term outcomes by destroying any microscopic cancer cells that have spread. Neoadjuvant chemotherapy may also provide a way to assess how effective the chemotherapy is for a specific patient’s cancer, which can guide future treatment.
Given the advantages of neoadjuvant chemotherapy for certain tumors, such as reducing tumor load, increasing the probability of radical resection, and improving prognosis 131, 132, some researchers have also discussed related aspects of pseudomyxoma peritonei. The results show that neoadjuvant chemotherapy does not improve the prognosis of patients with both low- and high-grade pseudomyxoma peritonei and can even lead to lower overall survival [overall survival (OS) is the length of time from either the date of diagnosis or the start of treatment for a disease, such as cancer, that patients diagnosed with the disease are still alive] and disease free survival [disease free survival (DFS) is the length of time after primary cancer treatment that a patient lives without any signs or symptoms of the cancer] 133, 134, 135, 136. However, Milovanov et al 136 found that neoadjuvant therapy improved overall survival (OS) at 1, 2, and 3 years in patients with peritoneal mucinous carcinomatosis with signet ring cells (PMCA-S) (94%, 67%, and 22% vs. 43%, 14%, and 14%). In addition, regarding the use of neoadjuvant chemotherapy, 87.3% of the Peritoneal Surface Oncology Group International (PSOGI) panel recommended fluoropyrimidine and an alkylating agent combination 80. In conclusion, current evidence suggests that patients pathologically classified as PMCA-S may benefit from neoadjuvant chemotherapy but that this therapeutic strategy is not suitable for the vast majority of pseudomyxoma peritonei patients, which may be related to the mucus barrier around tumor cells.
Systemic Chemotherapy
Systemic chemotherapy is a cancer treatment that uses drugs, delivered through the bloodstream, to reach and attack cancer cells throughout the entire body. Unlike local treatments, systemic chemo can treat cancer that has spread to distant organs or lymph nodes. These drugs can be administered via intravenous (IV) infusion, injection, or as oral pills. The need for systemic chemotherapy after CRS-HIPEC is a little-studied clinical topic in pseudomyxoma peritonei. Small-sample retrospective experiments from different centers have reported completely different results. Blackham et al (n = 22) found that, compared with CRS-HIPEC alone, additional systemic chemotherapy improved overall survival (OS) and progression-free survival (PFS) in patients with high-grade pseudomyxoma peritonei 134 . However, other studies have shown that postoperative systemic chemotherapy has no significant effect on overall survival (OS) or even has a negative effect 135, 137. In addition, the Peritoneal Surface Oncology Group International (PSOGI) expert group recommended that adjuvant (add-on) chemotherapy should be considered after completion of CRS-HIPEC for pseudomyxoma peritonei patients with a pathological classification of PMCA or PMCA-S (89.1%) and should not be completely abandoned for low-grade pseudomyxoma peritonei (90.9%) 80. However, it is undeniable that the effectiveness of systemic chemotherapy currently lacks direct medical evidence. In the future, it is necessary to conduct multicenter large-sample clinical studies to draw reliable conclusions to better regulate the treatment of pseudomyxoma peritonei.
Palliative Chemotherapy
Palliative chemotherapy is a potential treatment for pseudomyxoma peritonei patients who relapse or cannot be treated surgically due to their underlying conditions. At present, there is a lack of unified standards for chemotherapy regimens, and most centers use oxaliplatin- and 5-fluorouracil-based chemotherapy regimens 138, 112, 113, 114. Farquharson et al 138 conducted a phase II trial and found that 15 out of 40 patients with unresectable pseudomyxoma peritonei achieved significant clinical and radiographic responses after palliative chemotherapy (mitomycin C and capecitabine). Two patients were followed up with CRS-HIPEC due to good results. The overall 1- and 2-year overall survival rates were 84% and 61%, respectively 138. A series of subsequent experiments found that the disease control rate (DCR) after palliative chemotherapy was 65–88%, the median progression-free survival (PFS) was 8–13 months, and the median overall survival was 26.2–27.9 months 112, 113, 114. The chemotherapies used included FOLFOX4, mFOLFOX6 and capecitabine combined with cyclophosphamide 112, 113, 114. In addition, other studies found that the combination of bevacizumab, a monoclonal antibody targeting vascular endothelial growth factor (VEGF), can significantly improve progression-free survival (PFS) and overall survival in patients with pseudomyxoma peritonei, especially for patients with high-grade pseudomyxoma peritonei 139, 140. Therefore, more than 90.9% of the Peritoneal Surface Oncology Group International (PSOGI) expert group recommended that palliative chemotherapy be considered for patients with pseudomyxoma peritonei who were assessed as unfit for surgery or unresectable, regardless of the pathologic type of pseudomyxoma peritonei, and 78.2% recommended the combination of a single angiogenic inhibitor such as bevacizumab 80.
Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC)
Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is a newer method to treat peritoneal carcinomatosis. Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is a minimally invasive surgical technique that delivers low-dose chemotherapy as a pressurized aerosol directly into the abdominal cavity to treat peritoneal metastases 141. The pressure helps the chemotherapy drugs penetrate the tissue more deeply and evenly, potentially improving treatment effectiveness and reducing systemic side effects compared to traditional chemotherapy. This procedure, often performed via laparoscopy, is repeated every few weeks and can also help relieve symptoms like pain and swelling.
Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is typically considered for patients with peritoneal carcinomatosis that are not candidates for curative surgery (who may be deemed unresectable or unfit for surgery), such as those from ovarian, gastric, colorectal, or appendiceal cancers. Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is often recommended when other treatments have been ineffective or are not viable.
Relevant studies have shown that Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) applied to patients with peritoneal metastasis of gastrointestinal, appendix, ovary, and other malignant tumors can cause regression of peritoneal nodules or fibrosis, with good tumor response and safety 142, 143, 144, 145, 146. Only one clinical application of PIPAC in patients with pseudomyxoma peritonei has been reported. After three cycles of PIPAC (cisplatin 7.5 mg/m² combined with doxorubicin 1.5 mg/m², 12 mmHg, 37 °C, 30 min), the patient’s abdominal mucus and ascites basically disappeared, and 66% of intraoperative biopsy tissues indicated fibrosis and inflammation, with no tumor cells found 147. However, the actual efficacy and safety of PIPAC in pseudomyxoma peritonei patients remain to be evaluated. In addition, the comparison of efficacy between PIPAC and HIPEC is also a worthy clinical topic.
How pressurized intraperitoneal aerosol chemotherapy (PIPAC) works:
- Minimally invasive: It is performed using laparoscopy through a few small incisions.
- Pressurized aerosol: Chemotherapy drugs are delivered as a fine mist under high pressure, allowing for better distribution and deeper penetration into cancer tissue. During PIPAC, a special aerosolized mist of chemotherapy drug is put directly into the belly during a minimally invasive procedure. The chemotherapy drug is given as a fine aerosol under pressure, which helps it spread evenly throughout the abdomen.
- Procedure steps: The surgeon first takes tissue samples, then sprays the aerosolized chemotherapy for about 10 minutes, lets it sit for 30 minutes, and finally evacuates the gas before closing the incisions.
- Symptom relief: PIPAC can help ease symptoms such as abdominal pain and swelling, improving a patient’s quality of life.
- Repeated treatments: The procedure is typically repeated about every six weeks.
- Combined therapy: It is often used in conjunction with systemic chemotherapy.
Pressurized intraperitoneal aerosol chemotherapy (PIPAC) benefits and potential advantages:
- Improved drug delivery: The pressurized aerosol helps the chemotherapy spread more evenly throughout the abdominal cavity.
- Deeper tissue penetration: The pressure enhances drug absorption into the cancer tissue.
- Reduced systemic toxicity: Absorption into the bloodstream is limited, leading to potentially fewer systemic side effects like nausea, vomiting, and hair loss.
- Tissue analysis: Small tissue samples can be taken during the procedure, allowing doctors to study how the tumor responds to treatment.
Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is typically done when surgery isn’t possible or when surgery would be too risky or not helpful. PIPAC has also been found to have superior benefits of chemotherapy drug delivery to tumor tissue with a significant effect on tumor regression than conventional intraperitoneal chemotherapy or systemic chemotherapy 148.
Pressurized intraperitoneal aerosol chemotherapy (PIPAC) side effects:
- Some discomfort, minor abdominal pain, or mild nausea can occur after the procedure.
- More serious adverse events are uncommon, but can include abdominal pain, bleeding, or bowel obstruction.
Mucolytic Therapy
Mucin is the main component of intraperitoneal mucus in pseudomyxoma peritonei and is the core solution target of dissolving agents in mucolytic therapy. Mucolytic therapy uses medications to thin and break down thick mucus. Glycosidic bonds and disulfide bonds are the main chemical bonds for mucin polymerization and function 149. Therefore, any chemical that acts on either the glycosidic or disulfide bonds could theoretically degrade mucin. In the past, researchers have tried to use sodium bicarbonate and glucose to dissolve mucus in pseudomyxoma peritonei patients, but these reagents have mediocre effects and certain side effects, and they have not been further studied or promoted in clinical practice 150, 151, 152. In recent years, the use of a combination reagent of bromelain and N-acetylcysteine (NAC) developed by the Morris team has achieved significant mucolytic effects and high safety in preclinical studies and clinical trials. This represents a promising development for the clinical transformation and application of mucolytic therapy 153, 154, 155, 156.
Bromelain is a mixed enzyme extracted from the flesh, root, and skin of pineapple that can effectively hydrolyze glycosidic bonds 157. N-acetylcysteine (NAC) is a kind of respiratory suppurative mucolytic agent that has been widely used clinically and mainly relies on the destruction of disulfide bonds to complete the decomposition of mucin 158, 159. Morris’s team found, in the initial stage of the experiment, that 300 mg/mL bromelain +4% NAC could dissolve mucus at 3 hours in vitro and 72 hours in vivo without significant toxic side effects 155. In addition, they found that the combined reagents had similar mucolytic effects at 37 °C and 41 °C, suggesting that the combined reagents were effective even during HIPEC 155. On this basis, they also found that the combination reagent effectively reduced the expression and secretion of MUC2 and MUC5AC both in vitro (using LS174T colon cancer cells that expressed specific mucins instead of pseudomyxoma peritonei cells) and in vivo 153. They then verified its safety in a rat model, and the results showed that intraperitoneal injection of the combined agent was safe and had no significant impact on the healing of the colonic anastomosis. However, this study used a healthy rat model, and the impact on animal models under disease conditions is still unknown 160. Following these positive preclinical findings, the team conducted a multicenter prospective phase II trial to investigate the clinical efficacy of bromelain in combination with NAC in patients with recurrent peritoneal mucinous tumors and pseudomyxoma peritonei (NCT 03976973). The preliminary results showed that, in 20 patients with peritoneal disease (including 6 with pseudomyxoma peritonei), 73.2% of the treatment sites showed an objective response to extracting gelatinous or liquid tumor tissue directly from the drainage tube. In addition, complications were controllable 156. However, this study had a small sample size and lacked long-term prognostic results. In the future, more patients need to be included, and long-term prognostic results need to be tracked to support the conclusions of this study.
In addition, the team found that the combination of reagents significantly inhibited the proliferation of gastric and colon cancer cells (including the LS174T cell line) through synergistic and additive interactions 154. Its role may be related to interfering with the growth cycle of tumor cells and inducing apoptosis and autophagy 154. However, whether it can exert the same inhibitory effect in real pseudomyxoma peritonei cell lines remains to be further verified.
Antiangiogenic Therapy
Antiangiogenic therapy is a cancer treatment that stops tumors from growing and spreading by preventing the formation of new blood vessels that supply them with nutrients and oxygen. This is achieved by targeting factors like vascular endothelial growth factor (VEGF) which promotes angiogenesis. These therapies can be given as pills, infusions, or in combination with other treatments like chemotherapy or immunotherapy. Due to the small number of tumor cells and the large amount of mucus associated with pseudomyxoma peritonei, ordinary conventional chemotherapy drugs cannot play an effective role. Relevant studies have shown that antiangiogenic therapy may be an effective treatment 161, 162. Dohan et al 162 confirmed the existence of a large number of microvessels in pseudomyxoma peritonei tumor tissue through Doppler ultrasound and microangiography. Subsequent in vivo experiments showed that the survival time of bevacizumab-treated mice was significantly prolonged, blood vessels in tumor tissue gradually normalized, and the mean blood flow velocity slowed. In the same way, Andersson et al 161 found that the expression levels of vascular endothelial markers (CD31 and CD105) and angiogenic factors VEGFA, fibroblast growth factor 2 (FGF2), and soluble Fms-like tyrosine1 (sFLT1) in pseudomyxoma peritonei tumor tissue were significantly increased. In vivo studies have also shown that bevacizumab and aflibercept, an antiangiogenic drug targeting VEGFA, VEGFB, and placental growth factor (PlGF), both inhibit tumor growth to varying degrees, with the latter having a stronger effect. They suggest that PIGF and VEGFA are major targets for inhibiting pseudomyxoma peritonei angiogenesis 161. At present, preliminary results regarding bevacizumab treatment have been reported for small samples of patients with recurrent pseudomyxoma peritonei, but there is a lack of long-term prognostic evidence based on large samples 139, 140. In addition, angiogenesis inhibition is mainly applied to palliative treatment in pseudomyxoma peritonei, and its effect on preoperative neoadjuvant therapy or systemic treatment after CRS-HIPEC is unknown, which has certain research prospects.
Anti-Inflammatory Therapy
Anti-inflammatory therapy uses medications and targeted therapies to reduce inflammation. Mucin secretion is regulated by various mediators, including inflammatory cytokines 163, 164. Choudry et al 165 investigated the effect of anti-inflammatory therapy on pseudomyxoma peritonei through in vivo and in vitro experiments. The results showed that dexamethasone had dual inhibitory effects on the proliferation and mucin secretion of pseudomyxoma peritonei tumor cells 165. In addition, Celebrex (COX-2 inhibitor) reduced MUC2 expression levels only in an inflammatory environment, and its effect in vivo was less significant than that of dexamethasone 165. This study suggests a new strategy for the treatment of pseudomyxoma peritonei, but it is still necessary to further explore the mechanism of anti-inflammatory therapy in the future to provide sufficient preclinical research evidence for its widespread application.
Antibiotic Therapy
Past studies have described that bacteria such as Helicobacter pylori exist in pseudomyxoma peritonei tumor tissues and may participate in the progression of pseudomyxoma peritonei by influencing the secretion of MUC2 and inducing the EMT process 166, 167, 168. Based on this, Merrell et al 169 tracked the effect of perioperative anti-Helicobacter pylori therapy (lansoprazole, clarithromycin, and amoxicillin) on outcomes in patients with pseudomyxoma peritonei (n = 17). The results in patients with low-grade pseudomyxoma peritonei (n = 6) were as follows: five patients survived, and one patient was lost to follow-up. In those with high-grade pseudomyxoma peritonei (n = 11), five patients died from complications of pseudomyxoma peritonei, one died from other causes, and the remaining five patients survived. Although the prognosis is positive, especially for those with low-grade pseudomyxoma peritonei, these benefits cannot be determined to be due to anti-H. pylori therapy. In addition, one of the commonly used basic drugs of HIPEC is mitomycin C, which, as an antibiotic, also has a certain killing effect on bacteria in the abdominal cavity 168. Therefore, whether it is necessary to add antibiotic treatment during the perioperative period should be carefully considered. Moreover, more basic studies should be carried out to understand the role of bacteria in the progression of pseudomyxoma peritonei to guide the use of antibiotics in pseudomyxoma peritonei patients more scientifically.
Immunotoxin Therapy
Immunotoxin therapy uses a fusion protein of an antibody and a toxin to kill cancer cells. The antibody part targets specific antigens on cancer cells, directing the toxin to them. Once inside the cell, the toxin inhibits protein synthesis, leading to cell death. Immunotoxin therapy is effective for various cancers, particularly those that are resistant to traditional treatments like chemotherapy.
Immunotoxins are bifunctional molecules composed of monoclonal antibodies and toxins that rely on monoclonal antibodies to bind to target cells and exert cytotoxic effects through toxins 170. EpCAM is a type I transmembrane glycoprotein widely expressed in epithelial tumor cells such as the stomach, intestine, prostate, and lung 171. In recent years, a new immunotoxin drug, MOC31PE, which is covalent between MOC31, a monoclonal antibody specific to EpCAM, and Pseudomonas exotoxin (PE) A, which is secreted by Pseudomonas aeruginosa, has become a hot topic in many studies. MOC31PE specifically binds to EpCAM-expressing tumor cells, inhibits protein synthesis, and induces cell apoptosis and Phytophthora cell death by releasing Pseudomonas exotoxin (PE) 172, 173, 174. Currently, MOC31PE has been shown to have an encouraging prognosis in patients with peritoneal metastasis of colorectal cancer, with a 3-year overall survival of 72% and a median disease free surivival of 13 months, and has demonstrated good safety and tolerability 175, 176. Flatmark et al 174 found that EpCAM was also expressed in pseudomyxoma peritonei samples. MOC31PE alone or combined with mitomycin C showed significant tumor growth inhibition, and the combined effect was more obvious. This suggests that MOC31PE combined with mitomycin C-based HIPEC may be a more effective treatment. Therefore, it is worth carrying out targeted clinical trials in pseudomyxoma peritonei patients to further explore its actual efficacy and safety.
Targeted Hypoxia Therapy
Targeted hypoxia therapy is a cancer treatment strategy that focuses on the low-oxygen (hypoxic) environment within tumors, which is often resistant to conventional therapies. Key approaches include using hypoxia-activated prodrugs (HAPs) that become toxic only in hypoxic areas, inhibiting the hypoxia-inducible factor (HIF) pathway, which regulates genes related to survival and angiogenesis, and employing anti-angiogenic agents to normalize blood vessels and improve drug delivery. Other methods involve targeted radionuclide therapy and sensitizing hypoxic cells to radiation.
Due to the rapid proliferation of tumor cells and the imbalance of oxygen supply, an anoxic environment is very common in most tumor tissues 177. Hypoxia-inducible factor 1 (HIF-1) is a key transcription factor regulating oxygen homeostasis in the hypoxic microenvironment 178. Hypoxia-inducible factor 1 (HIF-1) is a heterodimer composed of two subunits, HIF-1α and HIF-1β. HIF-1β is structurally expressed, and HIF-1α is the main effector subunit. HIF-1α is degraded by ubiquitination in normal oxygen concentrations and is activated in low-oxygen environments to play an important regulatory role 179. At present, hypoxia-targeted therapy targeting HIF-1α has achieved significant efficacy in preclinical models of various tumors 180, 181. In addition, HIF-1α has been found to play a catalytic role in the expression and secretion of mucin 182, 183. Valenzuela-Molina et al 184 found that real-time oxygen microtension in soft and hard mucinous tumor tissues of pseudomyxoma peritonei patients was significantly reduced during surgery, and HIF-1α protein expression levels were increased. These results indicate that a hypoxia environment exists in pseudomyxoma peritonei. Dilly et al 185 also found that the HIF-1α expression level was significantly increased in pseudomyxoma peritonei tumor tissues, which could be associated with the MUC2 promoter (5′-ACGTGC-3′) interaction to regulate the expression level of MUC2. In addition, they found that HIF-1α inhibitors (YC-1 and BAY 87-2243) not only reduced the expression level of MUC2 but also effectively inhibited the progression of pseudomyxoma peritonei in mouse xenotransplantation models. This study suggests that hypoxia in pseudomyxoma peritonei regulates MUC2 expression by activating HIF-1α and that HIF-1α inhibitors may be an effective therapeutic strategy.
Immune Checkpoint Inhibitor Therapy
Immune checkpoint inhibitor therapy is a type of cancer immunotherapy (a cancer treatment that uses the body’s own immune system to fight cancer) that uses drugs to block “checkpoint” proteins, which prevents cancer cells from hiding from your immune system 186. These drugs release the “brakes” on the immune system, allowing immune cells, like T-cells (T lymphocytes), to recognize and attack cancer cells more effectively. Immune checkpoint inhibitor therapy works by preventing proteins like programmed cell death protein 1 (PD-1) and CTLA-4 (Cytotoxic T-lymphocyte-associated protein 4) from binding to their partners, which is a normal mechanism that stops the immune system from overreacting but can be exploited by cancer cells to avoid detection.
Immune checkpoints refer to a series of molecules that are expressed on immune cells and regulate the activation degree of the immune system, which is often compared to the “brake system” of immune cells to prevent the hyperactivation of the autoimmune system from causing damage to the body 187. Programmed cell death protein 1 (PD-1) is a widely studied immune checkpoint. Its combination with programmed cell death ligand 1 (PD-L1) expressed on tumor cells can maintain the immune system in the “brake state”, which leads to the reduction in tumor cell-specific immunity and promotes tumor progression. In recent years, PD-1/PD-L1 inhibitors have shown significant and long-lasting inhibitory effects in a variety of tumors 188, 189. Relevant studies have shown that PD-1 is found in approximately 36% of pseudomyxoma peritonei patients, and PD-L1 is found in approximately 16–18% 190, 191. Furthermore, the status of the MMR gene is related to the efficacy of anti-PD-1/anti-PD-L1 treatment, and mismatch repair deficiency (dMMR) patients treated with PD-1/PD-L1 inhibitors tend to have a better prognosis 192, 193, 194. Yan et al 195 conducted immunohistochemical tests on 155 pseudomyxoma peritonei surgical specimens, and the incidence of dMMR was approximately 6.3%. For this subset of pseudomyxoma peritonei patients, immune checkpoint inhibitor therapy may be a potential prognostic benefit. Immune checkpoint inhibitor therapy is one of the most promising new tumor immunotherapies 186.
How it works
- Normal immune response: Immune checkpoints are proteins on immune cells and other cells that act as “off-switches” to prevent the immune system from attacking healthy cells.
- Cancer’s evasion: Some cancer cells produce proteins (like PD-L1) that can bind to these checkpoint proteins on T-cells (like PD-1) and turn them off, preventing the immune system from attacking the tumor.
- Inhibitor’s action: Immune checkpoint inhibitors are drugs, often monoclonal antibodies, that block these checkpoint proteins from interacting with each other.
- Result: By blocking the “off-switch,” the T-cells are reactivated and can recognize and attack the cancer cells.
Target Mitogen-Activated Protein Kinase (MAPK) Signaling Pathway Therapy
The MAPK signaling pathway has been reported in many studies to play an important role in mucin secretion 196, 197. The high mutation rate of KRAS and the high expression of MUC2 are typical characteristics of pseudomyxoma peritonei. It is worth exploring whether the activation of the MAPK downstream signaling pathway mediated by KRAS mutation can play a role in regulating mucin secretion in pseudomyxoma peritonei. Kuracha et al 198 found that KRAS mutation activated the downstream PI3K/AKT and MEK/ERK signaling pathways, which were synergistically involved in maintaining MUC2 expression and tumor cell activity. A single application of a PI3K inhibitor (pictilisib) is prone to drug resistance, and in combination with an MEK inhibitor (cobimetinib), it can more effectively inhibit the expression of MUC2 and tumor growth. Dilly et al. 199 also confirmed that an MEK1/2 inhibitor (RDEA119) had a dual inhibitory effect on MUC2 expression and cell proliferation ability and that the use of RDEA119 in the peritoneal transplantation model of pseudomyxoma peritonei mice could significantly reduce tumor load and prolong survival time. It may inhibit the expression levels of downstream nuclear factor kappa B (NF-kB) and activating protein 1 (AP1) through the MEK-ERK pathway; thus, the activity of the MUC2 promoter is decreased. In addition, the KRAS mutation rate is high in pseudomyxoma peritonei patients, and KrasG12D is one of the most common mutation subtypes 190. Vázquez-Borrego et al 200 used a small molecule inhibitor MRTX1133 that can target the KrasG12D protein in a pseudomyxoma peritonei mouse model. The results showed that it can effectively inhibit tumor growth by reducing the MAPK and PI3K/AKT/mTOR signaling pathways. This opens up a new treatment direction for pseudomyxoma peritonei patients with KrasG12D mutations and provides a strong theoretical basis for subsequent clinical trials.
Target Immunosuppressive Factors
Targeting immunosuppressive factors involves therapies aimed at reversing the immune system’s suppression in conditions like cancer, which is often caused by immunosuppressive cells (like Tregs, MDSCs, and TAMs) and signaling pathways (like PD-1/PD-L1). These strategies include blocking the function of these immunosuppressive cells and their secreted factors, such as TGF-β, IL-10, and nitric oxide, or using targeted therapies to disrupt the signaling pathways they use to evade the immune system. The goal is to restore the immune system’s ability to fight cancer and other diseases.
Kusamura et al 191 found the presence of immunosuppressive factors in pseudomyxoma peritonei that are independent of KRAS and GNAS mutations, such as granulocyte macrophage colony-stimulating factor (GM-CSF) and A2A-adenosine receptor (A2AR). GM-CSF can further inhibit the anti-tumor ability of T lymphocytes by hindering the differentiation and maturation of dendritic cells 201. A2AR can inhibit the activity of NK cells and CD8+ T cells through the c-AMP/PKA signaling pathway 202. Relevant studies have shown that inhibiting GM-CSF and A2AR can effectively restore the activity of T cells, thereby alleviating immune suppression 203, 204. Therefore, targeting the inhibition of such immunosuppressive factors in pseudomyxoma peritonei may be a potentially effective treatment modality.
Pseudomyxoma peritonei prognosis
The prognosis of pseudomyxoma peritonei is highly correlated with its histopathological classification 1, 205. Table 5 shows three commonly used clinical pathological types. At present, the four-level classification system proposed by the Peritoneal Surface Oncology Group International (PSOGI) in 2016 has been widely recognized and accepted by front-line clinical workers around the world 206. The ten-year survival rate of patients with low-grade tumors or disseminated peritoneal adenomucinosis (DPAM) is 63% compared to 40.1% in patients with high-grade tumors or peritoneal mucinous carcinomatosis (PMCA) and 0% in patients with high-grade tumors with signet ring cells following the treatment of CRS/HIPEC in one recent study 207. However data varies between different studies 208, 209, 210, 211. Above all, patients who are undergoing CRS/HIPEC present with a better long-term outcome than those undergoing debulking surgery 212. Major complications after CRS/HIPEC include thromboembolism, anastomotic leak, bowel perforation, fistula formation, abscess, and wound dehiscence. There is also a high risk for neutropenia, sepsis, pleural effusion, and respiratory insufficiency 62, 213.
In 1980, Spratt et al 214 first applied cytoreductive surgery (CRS) combined with hyperthermic intraperitoneal chemotherapy (HIPEC) in the clinical treatment of pseudomyxoma peritonei patients, and since then, the prognosis of pseudomyxoma peritonei patients has significantly improved 78. According to the latest large sample retrospective analysis by Kusamura et al. 215, the 5-year survival rate of 1,548 pseudomyxoma peritonei patients who received CRS-HIPEC reached 57.8%. CRS-HIPEC has become the standard and preferred treatment protocol for pseudomyxoma peritonei and represents a historical milestone in pseudomyxoma peritonei treatment 216, 217. Although the overall survival [overall survival (OS) is the length of time from either the date of diagnosis or the start of treatment for a disease, such as cancer, that patients diagnosed with the disease are still alive] time of pseudomyxoma peritonei patients has been significantly prolonged, relevant studies have reported that approximately 24.2% of patients have tumor recurrence and progression after CRS-HIPEC 218. Although the rate of invasion of pseudomyxoma peritonei is significantly lower than that of other malignant tumors, and despite the fact that most patients can survive with tumors for a long time, when intestinal obstruction occurs again or other conditions caused by tumor recurrence require hospitalization for treatment, the therapeutic effect of CRS-HIPEC is often poor, and some patients lose the opportunity to repeat CRS-HIPEC 3. For these patients and for those who cannot receive standard treatment after assessment at the first visit, the current treatment methods are limited 111, 112, 113, 219, 114.
Table 5. Pseudomyxoma peritonei pathological grades
| Ronnett (1995) 220 | Peritoneal Surface Oncology Group International (PSOGI) (2016) 206 | 8th Edition American Joint Committee on Cancer (AJCC) Staging System (2017) 221 |
|---|---|---|
| NA | Acellular mucin (AM) Peritoneal lesions concentrated on or away from organ surfaces; composed of a large amount of mucin; without neoplastic epithelial cells | M1a |
| Disseminated peritoneal adenomucinosis (DPAM) Peritoneal lesions composed of abundant extracellular mucin and less focal mucinous epithelium; low cellular atypia; mitotic activity; with or without appendiceal mucinous adenoma | Low-grade mucinous carcinoma peritonei (LMCP)/Disseminated peritoneal adenomucinosis (DPAM) Peritoneal lesions show few low-grade epithelial cells (<20% of tumor volume); arranged in a single layer; mild cellular atypia; rare mitoses | M1b. G1 Well-differentiated |
| Peritoneal mucinous carcinomatosis (PMCA)/with intermediate feature (PMCA-I) Abundant epithelial cells in peritoneal lesions consistent with the architectural and cytological features of carcinoma; with or without primary mucinous adenocarcinoma/well-differentiated peritoneal mucinous carcinomatosis | High-grade mucinous carcinoma peritonei (HMCP)/peritoneal mucinous carcinomatosis (PMCA) Peritoneal lesions show abundant epithelial cells (>20% of tumor volume); high-grade histological features; infiltration of surrounding tissues; peripheral angiolymphatic and nerve invasion; cribriform growth Subclassification: – Well-differentiated (predominantly single-tubular glands; better cell polarity; obvious cellular atypia; invasive component) – Moderately differentiated (solid sheets of tumor cells with glandular structures; poor polarity) – Poorly differentiated (highly irregular to no adenoid structure; disappearance of cell polarity) | M1b. G2 or G3 Moderately or poorly differentiated |
| Not applicable | High-grade mucinous carcinoma peritonei with signet ring cells (HMCP-S)/Peritoneal mucinous carcinomatosis with signet ring cells (PMCA-S) High-grade histology of peritoneal lesions with signet-ring cell component (signet-ring cells ≥10%) | M1b. G3 Poorly differentiated; PMCA-S |
Abbreviations: PSOGI, Peritoneal Surface Oncology Group International; AJCC, American Joint Committee on Cancer; NA = not applicable.
[Source 3 ]Pseudomyxoma peritonei survival rate
Your survival rate is an estimate of how long you’ll live after receiving treatment for a specific disease. When you think about survival rates, it’s important to remember that pseudomyxoma peritonei is a rare disease that affects very few people. What researchers know about survival rates comes from what they learn from the experiences of small groups of people. If you have Pseudomyxoma Peritonei (PMP), ask your doctor to explain survival rate research and how it may apply in your situation.
Survival rates for pseudomyxoma peritonei (PMP) vary significantly based on treatment, with the cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS+HIPEC) combination showing the best outcomes. In one study, the 5-year survival rate was 62% and the median survival was 92 months, while other reports show 5-year survival rates from 49% to 92% and median survival up to 16.3 years. Factors like the tumor’s grade, completeness of cytoreduction, and a patient’s overall health influence the prognosis. Through a retrospective, multi-institutional study on 2298 patients treated at 16 specialized centers affiliated with the Peritoneal Surface Oncology Group International, Chua et al. reported a median survival rate of 196 months (16.3 years) and a median progression-free survival rate of 98 months (8.2 years) as well as 10- and 15-year survival rates of 63% and 59%, respectively 62.
There was a statistically significant difference in survival between cases classified as disseminated peritoneal adenomucinosis (DPAM), those classified as peritoneal mucinous carcinomatosis (PMCA) with intermediate or discordant features. and those classified as peritoneal mucinous carcinomatosis (PMCA) 32. The age-adjusted 5-year survival rates were 84% for patients with disseminated peritoneal adenomucinosis (DPAM). 37.6% for patients with peritoneal mucinous carcinomatosis (PMCA) with intermediate or discordant features, and 6.7% for patients with peritoneal mucinous carcinomatosis (PMCA) 32. The term disseminated peritoneal adenomucinosis (DPAM) should be used to diagnose the histologically benign peritoneal lesions associated with ruptared appendiceal mucinous adenomas and those that are pathologically identical but lack a demonstrable appendiceal adenoma. Cases with the pathologic features of adenocarcinoma should be designated peritoneal mucinous carcinomatosis (PMCA) because they have recognizably different pathologic features and a significantly worse prognosis 32.
Wheeler et al 222 reported that in patients with disseminated peritoneal adenomucinosis (DPAM), cytoreductive surgery (CRS) without intraperitoneal chemotherapy leads to a poorer prognosis, but does not affect patient survival. They contend that combining cytoreductive surgery with intraperitoneal chemotherapy in disseminated peritoneal adenomucinosis (DPAM) patients could add to the burden of treatment, and that cytoreductive surgery alone, without intraperitoneal chemotherapy, may be more advantageous for patients with pseudomyxoma peritonei with peritoneal mucinous carcinomatosis (PMCA) 222.
Patients who have all visible tumor removed (completeness of the cytoreduction (CC) score 0 or 1) have better outcomes than those where some tumor remains (completeness of the cytoreduction (CC) score 2 or 3) 22, 23, 24.
The 5-year disease-specific survival probability of pseudomyxoma peritonei patients after treatment by cytoreductive surgery (CRS) plus hyperthermic intraperitoneal chemotherapy (HIPEC) reaches 60% and median survival has been reported to range from 92 months up to 196 months (16.3 years). Patients with pseudomyxoma peritonei pathologically categorized as adenomucinosis benefit most from this treatment strategy with a 5-year disease-specific survival probability of 72%.
Even after successful treatment, recurrence is possible. Pseudomyxoma peritonei patients with high preoperative tumor marker levels tend to experience recurrence at some point in the future 223, 224, 225. Elevated levels of tumor markers such as CEA, CA19-9, and CA 125 during follow-up are directly correlated with pseudomyxoma peritonei recurrence. Conversely, patients with low tumor marker levels during follow-up tend to have better prognoses 223, 224, 226, 227, 225. While the 5-year and 10-year survival rates for pseudomyxoma peritonei patients treated with classical debulking surgery are 53% to 75% and 32% to 60%, respectively, an aggressive cytoreduction combined with intraoperative chemotherapy can increase the 10-year survival rate to 90% 226.
The majority of pseudomyxoma peritonei patients can achieve long-term survival and cure with CRS and HIPEC. In the largest multicenter study, including 2298 patients with pseudomyxoma peritonei, the reported median survival was 16.2 years and progression-free survival was 8.2 years. In a single-centre experience with 1000 patients similar results were reported. However, despite CRS CC0/1, up to 25% of patients may develop a recurrence, most commonly in the peritoneum: 20% in the first five years, 3% in the next 5–10 years and 2% a late recurrence >10 years after treatment. The management of recurrent pseudomyxoma peritonei can include active surveillance or repeat CRS and HIPEC. The five and ten-year overall survival of patients with recurrence is 74% and 54%, respectively.
Given the long-term survival of many patients with pseudomyxoma peritonei and, the invasive nature of CRS and HIPEC, it is important to consider the needs of pseudomyxoma peritonei survivors and to assess the long-term Health-Related Quality of Life (a measure of how a person’s health affects their overall well-being, encompassing physical, mental, and social domains) after CRS and HIPEC. While patients with pseudomyxoma peritonei and their caregivers reported negative effects on their Health-Related Quality of Life, results from previous publications show that Quality of Life improves significantly within one-year post-operatively. However, there is still limited information on the long-term Health-Related Quality of Life of patients with pseudomyxoma peritonei.
- Yu B, Raj MS. Pseudomyxoma Peritonei. [Updated 2024 Mar 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK541116[↩][↩]
- Askar A, Arpat A, Durgun V. Pseudomyxoma peritonei: The struggle of a lifetime and the hope of a cure – a rare diagnosis with review of the literature. North Clin Istanb. 2024 Jun 28;11(3):261-268. doi: 10.14744/nci.2023.50374[↩]
- Li X, Liu G, Wu W. Progress in Biological Research and Treatment of Pseudomyxoma Peritonei. Cancers (Basel). 2024 Apr 3;16(7):1406. doi: 10.3390/cancers16071406[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Ye S, Zheng S. Comprehensive Understanding and Evolutional Therapeutic Schemes for Pseudomyxoma Peritonei: A Literature Review. Am J Clin Oncol. 2022 May 1;45(5):223-231. doi: 10.1097/COC.0000000000000911[↩][↩][↩]
- Morera-Ocon FJ, Navarro-Campoy C. History of pseudomyxoma peritonei from its origin to the first decades of the twenty-first century. World J Gastrointest Surg. 2019 Sep 27;11(9):358-364. doi: 10.4240/wjgs.v11.i9.358[↩]
- Rizvi SA, Syed W, Shergill R. Approach to pseudomyxoma peritonei. World J Gastrointest Surg. 2018 Aug 27;10(5):49-56. doi: 10.4240/wjgs.v10.i5.49[↩][↩]
- Ramaswamy V. Pathology of Mucinous Appendiceal Tumors and Pseudomyxoma Peritonei. Indian J Surg Oncol. 2016 Jun;7(2):258-67. doi: 10.1007/s13193-016-0516-2[↩][↩][↩]
- Pantiora EV, Massaras D, Koutalas J, Melemeni A, Fragulidis GP. Pseudomyxoma Peritonei: Presentation of Two Cases and Challenging Issues in the Literature. Cureus. 2018 Dec 14;10(12):e3732. doi: 10.7759/cureus.3732[↩][↩]
- Guo AT, Song X, Wei LX, Zhao P. Histological origin of pseudomyxoma peritonei in Chinese women: clinicopathology and immunohistochemistry. World J Gastroenterol. 2011;17:3531–3537. doi: 10.3748/wjg.v17.i30.3531[↩]
- Kabbani W, et al. Mucinous and nonmucinous appendiceal adenocarcinomas: Different clinicopathological features but similar genetic alterations. Mod Pathol. 2002;15(6):599–605. doi: 10.1038/modpathol.3880572[↩]
- Pai RK, Longacre TA. Appendiceal mucinous tumors and pseudomyxoma peritonei: Histologic features diagnostic problems, and proposed classification. Adv Anat Pathol. 2005;12(6):291–311. doi: 10.1097/01.pap.0000194625.05137.51[↩]
- Sorungbe AO, Whiles E, Drye E, Oyibo SO. Pseudomyxoma Peritonei Secondary to a Primary Appendix Tumor: A Belly Full of Jelly. Cureus. 2019 Jul 24;11(7):e5221. doi: 10.7759/cureus.5221[↩]
- Guo AT, Song X, Wei LX, Zhao P. Histological origin of pseudomyxoma peritonei in Chinese women: clinicopathology and immunohistochemistry. World J Gastroenterol. 2011 Aug 14;17(30):3531-7. doi: 10.3748/wjg.v17.i30.3531[↩]
- Müller D. Das Appendix-Carcinom als Folge einer malignen Transformation eines villösen Adenoms [Appendiceal neoplasm resulting from a malignant transformation of a villous adenoma]. Chirurg. 1980 Sep;51(9):609-10. German.[↩]
- Carr N.J., Cecil T.D., Mohamed F., et al. A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am. J. Surg. Pathol. 2016;40:14–26. doi: 10.1097/PAS.0000000000000535[↩]
- Patrick-Brown T.D.J.H., Carr N.J., Swanson D.M., Larsen S., Mohamed F., Flatmark K. Estimating the Prevalence of Pseudomyxoma Peritonei in Europe Using a Novel Statistical Method. Ann. Surg. Oncol. 2020;28:252–257. doi: 10.1245/s10434-020-08655-8[↩]
- Mittal R., Chandramohan A., Moran B. Pseudomyxoma peritonei: Natural history and treatment. Int. J. Hyperth. 2017;33:511–519. doi: 10.1080/02656736.2017.1310938[↩]
- Smeenk R.M., van Velthuysen M.L., Verwaal V.J., Zoetmulder F.A. Appendiceal neoplasms and pseudomyxoma peritonei: A population based study. Eur. J. Surg. Oncol. 2008;34:196–201. doi: 10.1016/j.ejso.2007.04.002[↩]
- Zhong Y., Deng M., Xu R., Kokudo N., Tang W. Pseudomyxoma peritonei as an intractable disease and its preoperative assessment to help improve prognosis after surgery: A review of the literature. Intractable Rare Dis. Res. 2012;1:115–121. doi: 10.5582/irdr.2012.v1.3.115[↩]
- Stewart JHt, Shen P, Russell GB, et al. Appendiceal neoplasms with peritoneal dissemination: outcomes after cytoreductive surgery and intraperitoneal hyperthermic chemotherapy. Ann Surg Oncol. 2006;13(5):624–34. doi: 10.1007/s10434-006-9708-2[↩][↩]
- Sugarbaker P, Chang D. Results of treatment of 385 patients with peritoneal surface spread of appendiceal malignancy. Ann Surg Oncol. 1999;6(8):727–31. doi: 10.1007/s10434-999-0727-7[↩][↩]
- Smeenk RM, Verwaal VJ, Antonini N, Zoetmulder FA. Survival analysis of pseudomyxoma peritonei patients treated by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann Surg. 2007 Jan;245(1):104-9. doi: 10.1097/01.sla.0000231705.40081.1a[↩][↩][↩]
- Yang, R., Fu, YB., Li, XB. et al. Long-term survival in patients with PMP: a single-institutional retrospective study from China. World J Surg Onc 21, 347 (2023). https://doi.org/10.1186/s12957-023-03232-1[↩][↩][↩]
- Kung V, Delisle M, Alves S, Mohamed F, Cecil T, Moran B. Health related quality of life is excellent and sustained at two decades after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in survivors of pseudomyxoma peritonei of appendiceal origin. Eur J Surg Oncol. 2023 Nov;49(11):107045. doi: 10.1016/j.ejso.2023.107045[↩][↩][↩]
- Lin, YL., Xu, DZ., Li, XB. et al. Consensuses and controversies on pseudomyxoma peritonei: a review of the published consensus statements and guidelines. Orphanet J Rare Dis 16, 85 (2021). https://doi.org/10.1186/s13023-021-01723-6[↩][↩][↩][↩][↩]
- Cho JH, Kim SS. Peritoneal Carcinomatosis and Its Mimics: Review of CT Findings for Differential Diagnosis. J Belg Soc Radiol. 2020 Jan 30;104(1):8. doi: 10.5334/jbsr.1940[↩]
- de Bree E., Witkamp A., Van De Vijver M., Zoetmulde F. Unusual origins of Pseudomyxoma peritonei. J. Surg. Oncol. 2000;75:270–274. doi: 10.1002/1096-9098(200012)75:4<270::AID-JSO9>3.0.CO;2-V[↩]
- Gong Y., Wang X., Zhu Z. Pseudomyxoma Peritonei Originating from Transverse Colon Mucinous Adenocarcinoma: A Case Report and Literature Review. Gastroenterol. Res. Pr. 2020;2020:5826214. doi: 10.1155/2020/5826214[↩]
- Kataoka A., Ito K., Takemura N., Inagaki F., Mihara F., Gohda Y., Kiyomatsu T., Yamada K., Kojima N., Igari T., et al. Immunohistochemical staining as supportive diagnostic tool for pseudomyxoma peritonei arising from intraductal papillary mucinous neoplasm: A report of two cases and literature review. Pancreatology. 2020;20:1226–1233. doi: 10.1016/j.pan.2020.06.008[↩]
- Young R.H., Gilks C.B., Scully R.E. Mucinous tumors of the appendix associated with mucinous tumors of the ovary and pseudomyxoma peritonei. A clinicopathological analysis of 22 cases supporting an origin in the appendix. Am. J. Surg. Pathol. 1991;15:415–429. doi: 10.1097/00000478-199105000-00001[↩]
- Shih IM, Yan H, Speyrer D, Shmookler BM, Sugarbaker PH, Ronnett BM. Molecular genetic analysis of appendiceal mucinous adenomas in identical twins, including one with pseudomyxoma peritonei. Am J Surg Pathol. 2001 Aug;25(8):1095-9. doi: 10.1097/00000478-200108000-00017[↩]
- Ronnett BM, Zahn CM, Kurman RJ, Kass ME, Sugarbaker PH, Shmookler BM. Disseminated peritoneal adenomucinosis and peritoneal mucinous carcinomatosis. A clinicopathologic analysis of 109 cases with emphasis on distinguishing pathologic features, site of origin, prognosis, and relationship to “pseudomyxoma peritonei”. Am J Surg Pathol. 1995 Dec;19(12):1390-408. doi: 10.1097/00000478-199512000-00006[↩][↩][↩][↩][↩][↩]
- Bradley RF, Stewart JH 4th, Russell GB, Levine EA, Geisinger KR. Pseudomyxoma peritonei of appendiceal origin: a clinicopathologic analysis of 101 patients uniformly treated at a single institution, with literature review. Am J Surg Pathol. 2006 May;30(5):551-9. doi: 10.1097/01.pas.0000202039.74837.7d[↩][↩]
- Carr N, Sobin L. Adenocarcinoma of the appendix. In: Bosman F, Carneiro C, Hruban R, Theise N, editors. WHO classification of tumours of the digestive system. Lyon: IARC Press; 2010. p. 122–5.[↩]
- Carr NJ, Cecil TD, Mohamed F, Sobin LH, Sugarbaker PH, González-Moreno S, Taflampas P, Chapman S, Moran BJ; Peritoneal Surface Oncology Group International. A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am J Surg Pathol. 2016 Jan;40(1):14-26. doi: 10.1097/PAS.0000000000000535[↩][↩][↩]
- Valasek MA, Pai RK. An Update on the Diagnosis, Grading, and Staging of Appendiceal Mucinous Neoplasms. Adv Anat Pathol. 2018 Jan;25(1):38-60. doi: 10.1097/PAP.0000000000000178[↩]
- Krivsky LA. On the pseudomyxoma peritonei. BJOG Int J Obstet Gynaecol. 1921;28:204–227.[↩]
- Panarelli NC, Yantiss RK. Mucinous neoplasms of the appendix and peritoneum. Arch Pathol Lab Med. 2011 Oct;135(10):1261-8. doi: 10.5858/arpa.2011-0034-RA[↩]
- Li Y, Xu HB, Peng Z, Cui SZ, Wu W. Chinese expert consensus on cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for pseudomyxoma peritonei. Natl Med J China. 2019;99:1527–35.[↩]
- Miguez González J, Calaf Forn F, Pelegrí Martínez L, et al. Primary and secondary tumors of the peritoneum: key imaging features and differential diagnosis with surgical and pathological correlation. Insights Imaging. 2023 Jul 3;14(1):115. doi: 10.1186/s13244-023-01417-6[↩]
- Klumpp B, Aschoff P, Schwenzer N, Koenigsrainer I, Beckert S, Claussen CD, Miller S, Koenigsrainer A, Pfannenberg C. Correlation of preoperative magnetic resonance imaging of peritoneal carcinomatosis and clinical outcome after peritonectomy and HIPEC after 3 years of follow-up: preliminary results. Cancer Imaging. 2013 Dec 30;13(4):540-7. doi: 10.1102/1470-7330.2013.0044[↩][↩]
- Govaerts K, Chandrakumaran K, Carr NJ, Cecil TD, Dayal S, Mohamed F, Thrower A, Moran BJ. Single centre guidelines for radiological follow-up based on 775 patients treated by cytoreductive surgery and HIPEC for appendiceal pseudomyxoma peritonei. Eur J Surg Oncol. 2018 Sep;44(9):1371-1377. doi: 10.1016/j.ejso.2018.06.023[↩]
- Bouquot M, Dohan A, Gayat E, Barat M, Glehen O, Pocard M, Rousset P, Eveno C. Prediction of Resectability in Pseudomyxoma Peritonei with a New CT Score. Ann Surg Oncol. 2018 Mar;25(3):694-701. doi: 10.1245/s10434-017-6275-7[↩]
- Low RN, Barone RM, Lucero J. Comparison of MRI and CT for predicting the Peritoneal Cancer Index (PCI) preoperatively in patients being considered for cytoreductive surgical procedures. Ann Surg Oncol. 2015 May;22(5):1708-15. doi: 10.1245/s10434-014-4041-7[↩]
- Patel CM, Sahdev A, Reznek RH. CT, MRI and PET imaging in peritoneal malignancy. Cancer Imaging. 2011 Aug 24;11(1):123-39. doi: 10.1102/1470-7330.2011.0016[↩]
- de Bree E, Koops W, Kröger R, van Ruth S, Witkamp AJ, Zoetmulder FA. Peritoneal carcinomatosis from colorectal or appendiceal origin: correlation of preoperative CT with intraoperative findings and evaluation of interobserver agreement. J Surg Oncol. 2004 May 1;86(2):64-73. doi: 10.1002/jso.20049[↩]
- Rivard JD, Temple WJ, McConnell YJ, Sultan H, Mack LA. Preoperative computed tomography does not predict resectability in peritoneal carcinomatosis. Am J Surg. 2014 May;207(5):760-4; discussion 764-5. doi: 10.1016/j.amjsurg.2013.12.024[↩]
- Torkzad MR, Casta N, Bergman A, Ahlström H, Påhlman L, Mahteme H. Comparison between MRI and CT in prediction of peritoneal carcinomatosis index (PCI) in patients undergoing cytoreductive surgery in relation to the experience of the radiologist. J Surg Oncol. 2015 May;111(6):746-51. doi: 10.1002/jso.23878[↩]
- Low RN, Sebrechts CP, Barone RM, Muller W. Diffusion-weighted MRI of peritoneal tumors: comparison with conventional MRI and surgical and histopathologic findings–a feasibility study. AJR Am J Roentgenol. 2009 Aug;193(2):461-70. doi: 10.2214/AJR.08.1753[↩]
- Li J, Yan R, Lei J, Jiang C. Comparison of PET with PET/CT in detecting peritoneal carcinomatosis: a meta-analysis. Abdom Imaging. 2015 Oct;40(7):2660-6. doi: 10.1007/s00261-015-0418-8[↩]
- Vietti Violi N, Gavane S, Argiriadi P, Law A, Heiba S, Bekhor EY, Babb JS, Ghesani M, Labow DM, Taouli B. FDG-PET/MRI for the preoperative diagnosis and staging of peritoneal carcinomatosis: a prospective multireader pilot study. Abdom Radiol (NY). 2023 Dec;48(12):3634-3642. doi: 10.1007/s00261-022-03703-1[↩]
- Menon G, Ramos Santillan V. Peritoneal Surface Malignancies. [Updated 2025 Aug 9]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK541114[↩][↩][↩][↩][↩][↩][↩]
- Oliveira AA, Morais J, Pires O, Marques IL. Peritoneal carcinomatosis: the importance of laparoscopy. BMJ Case Rep. 2021 Oct 1;14(10):e243972. doi: 10.1136/bcr-2021-243972[↩]
- Hanna DN, Ghani MO, Hermina A, Mina A, Bailey CE, Idrees K, Magge D. Diagnostic Laparoscopy in Patients With Peritoneal Carcinomatosis Is Safe and Does Not Delay Cytoreductive Surgery With Hyperthermic Intraperitoneal Chemotherapy. Am Surg. 2022 Apr;88(4):698-703. doi: 10.1177/00031348211048819[↩]
- Carboni F, Federici O, Giofrè M, Valle M. An 18-Year Experience in Diagnostic Laparoscopy of Peritoneal Carcinomatosis: Results from 744 Patients. J Gastrointest Surg. 2020 Sep;24(9):2096-2103. doi: 10.1007/s11605-019-04368-w[↩]
- Ihemelandu C. The Landmark Series: Scoring Systems for Primary Peritoneal Surface Malignancy. Ann Surg Oncol. 2023 Mar;30(3):1832-1837. doi: 10.1245/s10434-022-12941-y[↩][↩][↩]
- Dohan A, Hobeika C, Najah H, Pocard M, Rousset P, Eveno C. Preoperative assessment of peritoneal carcinomatosis of colorectal origin. J Visc Surg. 2018 Sep;155(4):293-303. https://doi.org/10.1016/j.jviscsurg.2018.01.002[↩][↩]
- Ye Z, Yu P, Cao Y, Chai T, Huang S, Cheng X, Du Y. Prediction of Peritoneal Cancer Index and Prognosis in Peritoneal Metastasis of Gastric Cancer Using NLR-PLR-DDI Score: A Retrospective Study. Cancer Manag Res. 2022 Jan 12;14:177-187. doi: 10.2147/CMAR.S343467[↩][↩]
- Elzarkaa AA, Shaalan W, Elemam D, Mansour H, Melis M, Malik E, Soliman AA. Peritoneal cancer index as a predictor of survival in advanced stage serous epithelial ovarian cancer: a prospective study. J Gynecol Oncol. 2018 Jul;29(4):e47. doi: 10.3802/jgo.2018.29.e47[↩][↩]
- What is the PCI score? https://www.pseudomyxomasurvivor.org/faq-what-is-the-pci-score[↩][↩]
- Sugarbaker PH. Peritoneal carcinomatosis: principles of management. Boston, MA: Kluwer Academic Publishers; 1996.[↩]
- Chua TC, Moran BJ, Sugarbaker PH, Levine EA, Glehen O, Gilly FN, Baratti D, Deraco M, Elias D, Sardi A, Liauw W, Yan TD, Barrios P, Gómez Portilla A, de Hingh IH, Ceelen WP, Pelz JO, Piso P, González-Moreno S, Van Der Speeten K, Morris DL. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J Clin Oncol. 2012 Jul 10;30(20):2449-56. doi: 10.1200/JCO.2011.39.7166[↩][↩][↩][↩]
- Fagotti A, Vizzielli G, Fanfani F, Costantini B, Ferrandina G, Gallotta V, Gueli Alletti S, Tortorella L, Scambia G. Introduction of staging laparoscopy in the management of advanced epithelial ovarian, tubal and peritoneal cancer: impact on prognosis in a single institution experience. Gynecol Oncol. 2013 Nov;131(2):341-6. doi: 10.1016/j.ygyno.2013.08.005[↩]
- Esquivel J, Lowy AM, Markman M, Chua T, Pelz J, Baratti D, Baumgartner JM, Berri R, Bretcha-Boix P, Deraco M, Flores-Ayala G, Glehen O, Gomez-Portilla A, González-Moreno S, Goodman M, Halkia E, Kusamura S, Moller M, Passot G, Pocard M, Salti G, Sardi A, Senthil M, Spilioitis J, Torres-Melero J, Turaga K, Trout R. The American Society of Peritoneal Surface Malignancies (ASPSM) Multiinstitution Evaluation of the Peritoneal Surface Disease Severity Score (PSDSS) in 1,013 Patients with Colorectal Cancer with Peritoneal Carcinomatosis. Ann. Surg. Oncol. 2014 Dec;21(13):4195-201.[↩][↩]
- Sugarbaker PH, Ronnett BM, Archer A, Averbach AM, Bland R, Chang D, Dalton RR, Ettinghausen SE, Jacquet P, Jelinek J, Koslowe P, Kurman RJ, Shmookler B, Stephens AD, Steves MA, Stuart OA, White S, Zahn CM, Zoetmulder FA. Pseudomyxoma peritonei syndrome. Adv Surg. 1996;30:233-80.[↩]
- Shetty S, Natarajan B, Thomas P, Govindarajan V, Sharma P, Loggie B. Proposed classification of pseudomyxoma peritonei: influence of signet ring cells on survival. Am Surg. 2013 Nov;79(11):1171-6.[↩]
- Carr NJ, Bibeau F, Bradley RF, Dartigues P, Feakins RM, Geisinger KR, Gui X, Isaac S, Milione M, Misdraji J, Pai RK, Rodriguez-Justo M, Sobin LH, van Velthuysen MF, Yantiss RK. The histopathological classification, diagnosis and differential diagnosis of mucinous appendiceal neoplasms, appendiceal adenocarcinomas and pseudomyxoma peritonei. Histopathology. 2017 Dec;71(6):847-858. doi: 10.1111/his.13324[↩]
- Ferreira CR, Carvalho JP, Soares FA, Siqueira SA, Carvalho FM. Mucinous ovarian tumors associated with pseudomyxoma peritonei of adenomucinosis type: immunohistochemical evidence that they are secondary tumors. Int J Gynecol Cancer. 2008 Jan-Feb;18(1):59-65. doi: 10.1111/j.1525-1438.2007.00988.x[↩]
- Fernandes ACO, Rocha GRMD, Oliveira AD, Guimarães MD, Carvalho SC, Chojniak R. Pseudomyxoma peritonei in a pediatric patient: A case report and literature review. Rev Assoc Med Bras (1992). 2018 Feb;64(2):195-199. doi: 10.1590/1806-9282.64.02.195[↩]
- Ben Aziz M, Di Napoli R. Cytoreduction (CRS) and Hyperthermic Intraperitoneal Chemotherapy (HIPEC) [Updated 2023 Jul 31]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK570563[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Lin Y.L., Xu D.Z., Li X.B., Yan F.C., Xu H.B., Peng Z., Li Y. Consensuses and controversies on pseudomyxoma peritonei: A review of the published consensus statements and guidelines. Orphanet. J. Rare Dis. 2021;16:85. doi: 10.1186/s13023-021-01723-6[↩][↩]
- Sugarbaker P.H. Peritonectomy procedures. Surg. Oncol. Clin. N. Am. 2003;12:703–727. doi: 10.1016/S1055-3207(03)00048-6[↩]
- Jacquet P., Sugarbaker P.H. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat. Res. 1996;82:359–374. doi: 10.1007/978-1-4613-1247-5_23[↩]
- Stewart JH 4th, Blazer DG 3rd, Calderon MJG, et al. The Evolving Management of Peritoneal Surface Malignancies. Curr Probl Surg. 2021 Apr;58(4):100860. doi: 10.1016/j.cpsurg.2020.100860[↩]
- Foster JM, Zhang C, Rehman S, Sharma P, Alexander HR. The contemporary management of peritoneal metastasis: A journey from the cold past of treatment futility to a warm present and a bright future. CA Cancer J Clin. 2023 Jan;73(1):49-71. doi: 10.3322/caac.21749[↩]
- Clair KH, Wolford J, Zell JA, Bristow RE. Surgical Management of Gynecologic Cancers. Surg Oncol Clin N Am. 2021 Jan;30(1):69-88. doi: 10.1016/j.soc.2020.09.004[↩]
- Sugarbaker P.H. Cytoreductive surgery and perioperative intraperitoneal chemotherapy as a curative approach to pseudomyxoma peritonei syndrome. Tumori. 2001;87:S3–S5. doi: 10.1177/030089160108700415[↩]
- Chua T.C., Moran B.J., Sugarbaker P.H., Levine E.A., Glehen O., Gilly F.N., Baratti D., Deraco M., Elias D., Sardi A., et al. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J. Clin. Oncol. 2012;30:2449–2456. doi: 10.1200/JCO.2011.39.7166[↩][↩]
- Ansari N., Chandrakumaran K., Dayal S., Mohamed F., Cecil T.D., Moran B.J. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in 1000 patients with perforated appendiceal epithelial tumours. Eur. J. Surg. Oncol. J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2016;42:1035–1041. doi: 10.1016/j.ejso.2016.03.017[↩]
- Govaerts K., Lurvink R.J., De Hingh I., Van der Speeten K., Villeneuve L., Kusamura S., Kepenekian V., Deraco M., Glehen O., Moran B.J., et al. Appendiceal tumours and pseudomyxoma peritonei: Literature review with PSOGI/EURACAN clinical practice guidelines for diagnosis and treatment. Eur. J. Surg. Oncol. 2021;47:11–35. doi: 10.1016/j.ejso.2020.02.012[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Anwar A, Kasi A. Peritoneal Cancer. [Updated 2024 Apr 30]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK562138[↩]
- Mehta SS, Bhatt A, Glehen O. Cytoreductive Surgery and Peritonectomy Procedures. Indian J Surg Oncol. 2016 Jun;7(2):139-51. doi: 10.1007/s13193-016-0505-5[↩][↩]
- Hyperthermic Intraperitoneal Chemotherapy (HIPEC). https://www.mdanderson.org/treatment-options/hyperthermic-intraperitoneal-chemotherapy.html[↩]
- Flessner M.F. The transport barrier in intraperitoneal therapy. Am. J. Physiol. Ren. Physiol. 2005;288:F433–F442. doi: 10.1152/ajprenal.00313.2004[↩]
- Jacquet P., Sugarbaker P.H. Peritoneal-plasma barrier. Cancer Treat. Res. 1996;82:53–63. doi: 10.1007/978-1-4613-1247-5_4[↩]
- Cavaliere R., Ciocatto E.C., Giovanella B.C., Heidelberger C., Johnson R.O., Margottini M., Mondovi B., Moricca G., Rossi-Fanelli A. Selective heat sensitivity of cancer cells. Biochemical and clinical studies. Cancer. 1967;20:1351–1381. doi: 10.1002/1097-0142(196709)20:9<1351::AID-CNCR2820200902>3.0.CO;2-#[↩]
- Sticca R.P., Dach B.W. Rationale for hyperthermia with intraoperative intraperitoneal chemotherapy agents. Surg. Oncol. Clin. N. Am. 2003;12:689–701. doi: 10.1016/S1055-3207(03)00029-2[↩][↩]
- Overgaard J. Effect of hyperthermia on malignant cells in vivo. A review and a hypothesis. Cancer. 1977;39:2637–2646. doi: 10.1002/1097-0142(197706)39:6<2637::AID-CNCR2820390650>3.0.CO;2-S[↩]
- Dudar TE, Jain RK. Differential response of normal and tumor microcirculation to hyperthermia. Cancer Res. 1984 Feb;44(2):605-12.[↩]
- Jacquet P., Averbach A., Stuart O.A., Chang D., Sugarbaker P.H. Hyperthermic intraperitoneal doxorubicin: Pharmacokinetics, metabolism, and tissue distribution in a rat model. Cancer Chemother. Pharmacol. 1998;41:147–154. doi: 10.1007/s00280005072[↩]
- de Bree E, Tsiftsis DD. Principles of perioperative intraperitoneal chemotherapy for peritoneal carcinomatosis. Recent Results Cancer Res. 2007;169:39-51. doi: 10.1007/978-3-540-30760-0_4[↩]
- El-Kareh A.W., Secomb T.W. A theoretical model for intraperitoneal delivery of cisplatin and the effect of hyperthermia on drug penetration distance. Neoplasia. 2004;6:117–127. doi: 10.1593/neo.03205[↩]
- Gonzalez-Moreno S., Gonzalez-Bayon L.A., Ortega-Perez G. Hyperthermic intraperitoneal chemotherapy: Rationale and technique. World J. Gastrointest. Oncol. 2010;2:68–75. doi: 10.4251/wjgo.v2.i2.68[↩]
- Elias D., Bonnay M., Puizillou J.M., Antoun S., Demirdjian S., El O.A., Pignon J.P., Drouard-Troalen L., Ouellet J.F., Ducreux M. Heated intra-operative intraperitoneal oxaliplatin after complete resection of peritoneal carcinomatosis: Pharmacokinetics and tissue distribution. Ann. Oncol. 2002;13:267–272. doi: 10.1093/annonc/mdf019[↩][↩]
- Stewart J.H., Shen P., Russell G., Fenstermaker J., McWilliams L., Coldrun F.M., Levine K.E., Jones B.T., Levine E.A. A phase I trial of oxaliplatin for intraperitoneal hyperthermic chemoperfusion for the treatment of peritoneal surface dissemination from colorectal and appendiceal cancers. Ann. Surg. Oncol. 2008;15:2137–2145. doi: 10.1245/s10434-008-9967-1[↩][↩][↩]
- Turaga K., Levine E., Barone R., Sticca R., Petrelli N., Lambert L., Nash G., Morse M., Adbel-Misih R., Alexander H.R., et al. Consensus guidelines from The American Society of Peritoneal Surface Malignancies on standardizing the delivery of hyperthermic intraperitoneal chemotherapy (HIPEC) in colorectal cancer patients in the United States. Ann. Surg. Oncol. 2014;21:1501–1505. doi: 10.1245/s10434-013-3061-z[↩][↩]
- Levine E.A., Votanopoulos K.I., Shen P., Russell G., Fenstermaker J., Mansfield P., Bartlett D., Stewart J.H. A Multicenter Randomized Trial to Evaluate Hematologic Toxicities after Hyperthermic Intraperitoneal Chemotherapy with Oxaliplatin or Mitomycin in Patients with Appendiceal Tumors. J. Am. Coll Surg. 2018;226:434–443. doi: 10.1016/j.jamcollsurg.2017.12.027[↩]
- de Bree E, Tsiftsis DD. Experimental and pharmacokinetic studies in intraperitoneal chemotherapy: from laboratory bench to bedside. Recent Results Cancer Res. 2007;169:53-73. doi: 10.1007/978-3-540-30760-0_5[↩]
- Van der Speeten K., Stuart O.A., Chang D., Mahteme H., Sugarbaker P.H. Changes induced by surgical and clinical factors in the pharmacology of intraperitoneal mitomycin C in 145 patients with peritoneal carcinomatosis. Cancer Chemother. Pharmacol. 2011;68:147–156. doi: 10.1007/s00280-010-1460-4[↩]
- Seshadri RA, Glehen O. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in gastric cancer. World J Gastroenterol. 2016 Jan 21;22(3):1114-30. doi: 10.3748/wjg.v22.i3.1114[↩][↩]
- Coccolini F, Cotte E, Glehen O, Lotti M, Poiasina E, Catena F, Yonemura Y, Ansaloni L. Intraperitoneal chemotherapy in advanced gastric cancer. Meta-analysis of randomized trials. Eur J Surg Oncol. 2014 Jan;40(1):12-26. doi: 10.1016/j.ejso.2013.10.019[↩]
- Huo YR, Richards A, Liauw W, Morris DL. Hyperthermic intraperitoneal chemotherapy (HIPEC) and cytoreductive surgery (CRS) in ovarian cancer: A systematic review and meta-analysis. Eur J Surg Oncol. 2015 Dec;41(12):1578-89. doi: 10.1016/j.ejso.2015.08.172[↩]
- Klos D, Riško J, Stašek M, Loveček M, Hanuliak J, Skalický P, Lemstrová R, Mohelníková BD, Študentová H, Neoral Č, Melichar B. Current status of cytoreductive surgery (CRS) and intraperitoneal hyperthermic chemotherapy (HIPEC) in the multimodal treatment of peritoneal surface malignancies. Cas Lek Cesk. 2018 Dec 17;157(8):419-428. English.[↩]
- Garland-Kledzik M, Uppal A, Naeini YB, et al. Utility of Immunoprofiling in the Selection of Patients with Colorectal Peritoneal Carcinomatosis for Cytoreductive Surgery (CRS) and Heated Intraperitoneal Chemotherapy (HIPEC). J Gastrointest Surg. 2021 Jan;25(1):233-240. doi: 10.1007/s11605-020-04886-y[↩]
- Dehal A, Smith JJ, Nash GM. Cytoreductive surgery and intraperitoneal chemotherapy: an evidence-based review-past, present and future. J Gastrointest Oncol. 2016 Feb;7(1):143-57. doi: 10.3978/j.issn.2078-6891.2015.112[↩]
- Lambert LA. Looking up: Recent advances in understanding and treating peritoneal carcinomatosis. CA Cancer J Clin. 2015 Jul-Aug;65(4):284-98. doi: 10.3322/caac.21277[↩]
- da Silva RG, Sugarbaker PH. Analysis of prognostic factors in seventy patients having a complete cytoreduction plus perioperative intraperitoneal chemotherapy for carcinomatosis from colorectal cancer. J Am Coll Surg. 2006 Dec;203(6):878-86. doi: 10.1016/j.jamcollsurg.2006.08.024[↩]
- Piso P, Nedelcut SD, Rau B, Königsrainer A, Glockzin G, Ströhlein MA, Hörbelt R, Pelz J. Morbidity and Mortality Following Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy: Data from the DGAV StuDoQ Registry with 2149 Consecutive Patients. Ann Surg Oncol. 2019 Jan;26(1):148-154. doi: 10.1245/s10434-018-6992-6[↩][↩]
- Elias D, Raynard B, Boige V, Laplanche A, Estphan G, Malka D, Pocard M. Impact of the extent and duration of cytoreductive surgery on postoperative hematological toxicity after intraperitoneal chemohyperthermia for peritoneal carcinomatosis. J Surg Oncol. 2005 Jun 15;90(4):220-5. doi: 10.1002/jso.20253[↩]
- Benhaim L., Faron M., Gelli M., Sourrouille I., Honoré C., Delhorme J.B., Elias D., Goere D. Survival after complete cytoreductive surgery and HIPEC for extensive pseudomyxoma peritonei. Surg. Oncol. 2019;29:78–83. doi: 10.1016/j.suronc.2019.03.004[↩]
- Farquharson A.L., Pranesh N., Witham G., Swindell R., Taylor M.B., Renehan A.G., Rout S., Wilson M.S., O’Dwyer S.T., Saunders M.P. A phase II study evaluating the use of concurrent mitomycin C and capecitabine in patients with advanced unresectable pseudomyxoma peritonei. Br. J. Cancer. 2008;99:591–596. doi: 10.1038/sj.bjc.6604522[↩][↩]
- Hiraide S., Komine K., Sato Y., Ouchi K., Imai H., Saijo K., Takahashi M., Takahashi S., Shirota H., Takahashi M., et al. Efficacy of modified FOLFOX6 chemotherapy for patients with unresectable pseudomyxoma peritonei. Int. J. Clin. Oncol. 2020;25:774–781. doi: 10.1007/s10147-019-01592-x[↩][↩][↩][↩][↩]
- Pietrantonio F., Maggi C., Fanetti G., Iacovelli R., Di Bartolomeo M., Ricchini F., Deraco M., Perrone F., Baratti D., Kusamura S., et al. FOLFOX-4 chemotherapy for patients with unresectable or relapsed peritoneal pseudomyxoma. Oncologist. 2014;19:845–850. doi: 10.1634/theoncologist.2014-0106[↩][↩][↩][↩][↩]
- Raimondi A., Corallo S., Niger M., Antista M., Randon G., Morano F., Milione M., Kusamura S., Baratti D., Guaglio M., et al. Metronomic Capecitabine with Cyclophosphamide Regimen in Unresectable or Relapsed Pseudomyxoma Peritonei. Clin. Color. Cancer. 2019;18:e179–e190. doi: 10.1016/j.clcc.2019.03.002[↩][↩][↩][↩][↩]
- Delhorme J.B., Elias D., Varatharajah S., Benhaim L., Dumont F., Honore C., Goere D. Can a Benefit be Expected from Surgical Debulking of Unresectable Pseudomyxoma Peritonei? Ann. Surg. Oncol. 2016;23:1618–1624. doi: 10.1245/s10434-015-5019-9[↩][↩]
- Alves S., Mohamed F., Yadegarfar G., Youssef H., Moran B.J. Prospective longitudinal study of quality of life following cytoreductive surgery and intraperitoneal chemotherapy for pseudomyxoma peritonei. Eur. J. Surg. Oncol. 2010;36:1156–1161. doi: 10.1016/j.ejso.2010.09.004[↩]
- Soucisse ML, Liauw W, Hicks G, Morris DL. Early postoperative intraperitoneal chemotherapy for lower gastrointestinal neoplasms with peritoneal metastasis: a systematic review and critical analysis. Pleura Peritoneum. 2019 Oct 4;4(3):20190007. doi: 10.1515/pp-2019-0007[↩][↩]
- Sugarbaker PH, Graves T, DeBruijn EA, Cunliffe WJ, Mullins RE, Hull WE, Oliff L, Schlag P. Early postoperative intraperitoneal chemotherapy as an adjuvant therapy to surgery for peritoneal carcinomatosis from gastrointestinal cancer: pharmacological studies. Cancer Res. 1990 Sep 15;50(18):5790-4.[↩][↩]
- Fung X, Li IC, Chandrakumaran K, Cecil T, Dayal S, Tzivanakis A, Moran B, Mohamed F. Early postoperative intraperitoneal chemotherapy (EPIC) following cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) in 632 patients with pseudomyxoma peritonei of appendiceal origin: A single institution experience. Eur J Surg Oncol. 2022 Jul;48(7):1614-1618. doi: 10.1016/j.ejso.2022.02.002[↩][↩]
- Goodman MD, McPartland S, Detelich D, Saif MW. Chemotherapy for intraperitoneal use: a review of hyperthermic intraperitoneal chemotherapy and early post-operative intraperitoneal chemotherapy. J Gastrointest Oncol. 2016 Feb;7(1):45-57. doi: 10.3978/j.issn.2078-6891.2015.111[↩]
- Chua T.C., Liauw W., Zhao J., Morris D.L. Comparative analysis of perioperative intraperitoneal chemotherapy regimen in appendiceal and colorectal peritoneal carcinomatosis. Int. J. Clin. Oncol. 2013;18:439–446. doi: 10.1007/s10147-012-0397-5[↩][↩][↩]
- Huang Y., Alzahrani N.A., Liauw W., Soudy H., Alzahrani A.M., Morris D.L. Early postoperative intraperitoneal chemotherapy is associated with survival benefit for appendiceal adenocarcinoma with peritoneal dissemination. Eur. J. Surg. Oncol. 2017;43:2292–2298. doi: 10.1016/j.ejso.2017.09.002[↩]
- Lam J.Y., McConnell Y.J., Rivard J.D., Temple W.J., Mack L.A. Hyperthermic intraperitoneal chemotherapy + early postoperative intraperitoneal chemotherapy versus hyperthermic intraperitoneal chemotherapy alone: Assessment of survival outcomes for colorectal and high-grade appendiceal peritoneal carcinomatosis. Am. J. Surg. 2015;210:424–430. doi: 10.1016/j.amjsurg.2015.03.008[↩]
- Tan G.H., Ong W.S., Chia C.S., Tham C.K., Soo K.C., Teo M.C. Does early post-operative intraperitoneal chemotherapy (EPIC) for patients treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (HIPEC) make a difference? Int. J. Hyperth. 2016;32:281–288. doi: 10.3109/02656736.2015.1135485[↩]
- Pestieau S.R., Esquivel J., Sugarbaker P.H. Pleural extension of mucinous tumor in patients with pseudomyxoma peritonei syndrome. Ann. Surg. Oncol. 2000;7:199–203. doi: 10.1007/BF02523654[↩][↩][↩]
- Ghosh R.K., Somasundaram M., Ravakhah K., Hassan C. Pseudomyxoma peritonei with intrathoracic extension: A rare disease with rarer presentation from low-grade mucinous adenocarcinoma of the appendix. BMJ Case Rep. 2016;2016:bcr2015211076. doi: 10.1136/bcr-2015-211076[↩]
- Zoetmulder F.A., Sugarbaker P.H. Patterns of failure following treatment of pseudomyxoma peritonei of appendiceal origin. Eur. J. Cancer. 1996;32A:1727–1733. doi: 10.1016/0959-8049(96)00178-5[↩][↩]
- Senthil M., Harrison L.E. Simultaneous bicavitary hyperthermic chemoperfusion in the management of pseudomyxoma peritonei with synchronous pleural extension. Arch. Surg. 2009;144:970–972. doi: 10.1001/archsurg.2009.166[↩][↩]
- Saladino E., Famulari C., La Monaca E., Fortugno A., Fleres F., Macrì A. Cytoreductive surgery plus bicavitary chemohyperthermia as treatment of pseudomixoma peritonei with pleural extension. A case report and review of the literature. Ann. Ital. Di Chir. 2014;85:372–376[↩]
- Ashraf-Kashani N., Bell J. Haemodynamic changes during hyperthermic intra-thoracic chemotherapy for pseudomyxoma peritonei. Int. J. Hyperth. 2017;33:675–678. doi: 10.1080/02656736.2017.1300944[↩]
- Joshi S.S., Badgwell B.D. Current treatment and recent progress in gastric cancer. CA Cancer J. Clin. 2021;71:264–279. doi: 10.3322/caac.21657[↩]
- Kerr A.J., Dodwell D., McGale P., Holt F., Duane F., Mannu G., Darby S.C., Taylor C.W. Adjuvant and neoadjuvant breast cancer treatments: A systematic review of their effects on mortality. Cancer Treat. Rev. 2022;105:102375. doi: 10.1016/j.ctrv.2022.102375[↩]
- Baratti D., Kusamura S., Nonaka D., Cabras A.D., Laterza B., Deraco M. Pseudomyxoma peritonei: Biological features are the dominant prognostic determinants after complete cytoreduction and hyperthermic intraperitoneal chemotherapy. Ann. Surg. 2009;249:243–249. doi: 10.1097/SLA.0b013e31818eec64[↩]
- Blackham A.U., Swett K., Eng C., Sirintrapun J., Bergman S., Geisinger K.R., Votanopoulos K., Stewart J.H., Shen P., Levine E.A. Perioperative systemic chemotherapy for appendiceal mucinous carcinoma peritonei treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J. Surg. Oncol. 2014;109:740–745. doi: 10.1002/jso.23547[↩][↩]
- Cummins K.A., Russell G.B., Votanopoulos K.I., Shen P., Stewart J.H., Levine E.A. Peritoneal dissemination from high-grade appendiceal cancer treated with cytoreductive surgery (CRS) and hyperthermic intraperitoneal chemotherapy (HIPEC) J. Gastrointest. Oncol. 2016;7:3–9. doi: 10.3978/j.issn.2078-6891.2015.101[↩][↩]
- Milovanov V., Sardi A., Ledakis P., Aydin N., Nieroda C., Sittig M., Nunez M., Gushchin V. Systemic chemotherapy (SC) before cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) in patients with peritoneal mucinous carcinomatosis of appendiceal origin (PMCA) Eur. J. Surg. Oncol. 2015;41:707–712. doi: 10.1016/j.ejso.2015.01.005[↩][↩]
- Schomas D.A., Miller R.C., Donohue J.H., Gill S., Thurmes P.J., Haddock M.G., Quevedo J.F., Gunderson L.L. Intraperitoneal treatment for peritoneal mucinous carcinomatosis of appendiceal origin after operative management: Long-term follow-up of the Mayo Clinic experience. Ann. Surg. 2009;249:588–595. doi: 10.1097/SLA.0b013e31819ec7e3[↩]
- Farquharson A.L., Pranesh N., Witham G., Swindell R., Taylor M.B., Renehan A.G., Rout S., Wilson M.S., O’Dwyer S.T., Saunders M.P. A phase II study evaluating the use of concurrent mitomycin C and capecitabine in patients with advanced unresectable pseudomyxoma peritonei. Br. J. Cancer. 2008;99:591–596. doi: 10.1038/sj.bjc.660452[↩][↩][↩]
- Pietrantonio F., Berenato R., Maggi C., Caporale M., Milione M., Perrone F., Tamborini E., Baratti D., Kusamura S., Mariani L., et al. GNAS mutations as prognostic biomarker in patients with relapsed peritoneal pseudomyxoma receiving metronomic capecitabine and bevacizumab: A clinical and translational study. J. Transl. Med. 2016;14:125. doi: 10.1186/s12967-016-0877-x[↩][↩]
- Choe J.H., Overman M.J., Fournier K.F., Royal R.E., Ohinata A., Rafeeq S., Beaty K., Phillips J.K., Wolff R.A., Mansfield P.F., et al. Improved Survival with Anti-VEGF Therapy in the Treatment of Unresectable Appendiceal Epithelial Neoplasms. Ann. Surg. Oncol. 2015;22:2578–2584. doi: 10.1245/s10434-014-4335-9[↩][↩]
- Grass F, Vuagniaux A, Teixeira-Farinha H, Lehmann K, Demartines N, Hübner M. Systematic review of pressurized intraperitoneal aerosol chemotherapy for the treatment of advanced peritoneal carcinomatosis. Br J Surg. 2017 May;104(6):669-678.[↩]
- Alyami M., Hübner M., Grass F., Bakrin N., Villeneuve L., Laplace N., Passot G., Glehen O., Kepenekian V. Pressurised intraperitoneal aerosol chemotherapy: Rationale, evidence, and potential indications. Lancet Oncol. 2019;20:e368–e377. doi: 10.1016/S1470-2045(19)30318-3[↩]
- Badgwell B. Is PIPAC a New Summit for Peritoneal Disease Treatment or are we Lost in the Snowstorm? Ann. Surg. Oncol. 2022;29:13–14. doi: 10.1245/s10434-021-10899-x[↩]
- Lurvink R.J., Rovers K.P., Nienhuijs S.W., Creemers G.J., Burger J.W.A., de Hingh I.H.J. Pressurized intraperitoneal aerosol chemotherapy with oxaliplatin (PIPAC-OX) in patients with colorectal peritoneal metastases-a systematic review. J. Gastrointest. Oncol. 2021;12:S242–S258. doi: 10.21037/jgo-20-257[↩]
- Solass W., Kerb R., Mürdter T., Giger-Pabst U., Strumberg D., Tempfer C., Zieren J., Schwab M., Reymond M.A. Intraperitoneal chemotherapy of peritoneal carcinomatosis using pressurized aerosol as an alternative to liquid solution: First evidence for efficacy. Ann. Surg. Oncol. 2014;21:553–559. doi: 10.1245/s10434-013-3213-1[↩]
- Solass W., Herbette A., Schwarz T., Hetzel A., Sun J.S., Dutreix M., Reymond M.A. Therapeutic approach of human peritoneal carcinomatosis with Dbait in combination with capnoperitoneum: Proof of concept. Surg. Endosc. 2012;26:847–852. doi: 10.1007/s00464-011-1964-y[↩]
- Tempfer C.B., Solass W., Buerkle B., Reymond M.A. Pressurized intraperitoneal aerosol chemotherapy (PIPAC) with cisplatin and doxorubicin in a woman with pseudomyxoma peritonei: A case report. Gynecol. Oncol. Rep. 2014;10:32–35. doi: 10.1016/j.gore.2014.10.001[↩]
- Hübner M, Teixeira H, Boussaha T, Cachemaille M, Lehmann K, Demartines N. [PIPAC–Pressurized intraperitoneal aerosol chemotherapy. A novel treatment for peritoneal carcinomatosis]. Rev Med Suisse. 2015 Jun 17;11(479):1325-30.[↩]
- Hansson G.C. Mucins and the Microbiome. Annu. Rev. Biochem. 2020;89:769–793. doi: 10.1146/annurev-biochem-011520-105053[↩]
- Mieda R, Aso C, Nishikawa K, Saito S, Goto F. [Transient hyperglycemia following intra-peritoneal irrigation with 5% glucose in a patient with pseudomyxoma peritonei]. Masui. 2007 Aug;56(8):959-61. Japanese.[↩]
- Roy W.J., Jr Thomas B.L., Horowitz I.R. Acute hyperglycemia following intraperitoneal irrigation with 10% dextrose in a patient with pseudomyxoma peritonei. Gynecol. Oncol. 1997;65:360–362. doi: 10.1006/gyno.1997.4657[↩]
- Shirasawa Y., Orita H., Ishida K., Morimoto Y., Matsumoto M., Sakabe T. Critical alkalosis following intraperitoneal irrigation with sodium bicarbonate in a patient with pseudomyxoma peritonei. J. Anesth. 2008;22:278–281. doi: 10.1007/s00540-008-0612-8[↩]
- Amini A., Masoumi-Moghaddam S., Ehteda A., Liauw W., Morris D.L. Depletion of mucin in mucin-producing human gastrointestinal carcinoma: Results from in vitro and in vivo studies with bromelain and N-acetylcysteine. Oncotarget. 2015;6:33329–33344. doi: 10.18632/oncotarget.5259[↩][↩]
- Amini A., Masoumi-Moghaddam S., Ehteda A., Morris D.L. Bromelain and N-acetylcysteine inhibit proliferation and survival of gastrointestinal cancer cells in vitro: Significance of combination therapy. J. Exp. Clin. Cancer Res. 2014;33:92. doi: 10.1186/s13046-014-0092-7[↩][↩][↩]
- Pillai K., Akhter J., Chua T.C., Morris D.L. A formulation for in situ lysis of mucin secreted in pseudomyxoma peritonei. Int. J. Cancer. 2014;134:478–486. doi: 10.1002/ijc.28380[↩][↩][↩]
- Valle S.J., Akhter J., Mekkawy A.H., Lodh S., Pillai K., Badar S., Glenn D., Power M., Liauw W., Morris D.L. A novel treatment of bromelain and acetylcysteine (BromAc) in patients with peritoneal mucinous tumours: A phase I first in man study. Eur. J. Surg. Oncol. 2021;47:115–122. doi: 10.1016/j.ejso.2019.10.033[↩][↩]
- Rowan AD, Buttle DJ, Barrett AJ. The cysteine proteinases of the pineapple plant. Biochem J. 1990 Mar 15;266(3):869-75. https://pmc.ncbi.nlm.nih.gov/articles/instance/1131219/pdf/biochemj00187-0239.pdf[↩]
- Turner J., Jones C.E. Regulation of mucin expression in respiratory diseases. Biochem. Soc. Trans. 2009;37:877–881. doi: 10.1042/BST0370877[↩]
- Rubin BK. Mucolytics, expectorants, and mucokinetic medications. Respir Care. 2007 Jul;52(7):859-65.[↩]
- Mekkawy AH, Breakeit M, Pillai K, Badar S, Akhter J, Valle SJ, Morris DL. Intraperitoneal BromAc® Does Not Interfere with the Healing of Colon Anastomosis. Cancers (Basel). 2023 Jun 24;15(13):3321. doi: 10.3390/cancers15133321[↩]
- Andersson Y., Fleten K.G., Abrahamsen T.W., Reed W., Davidson B., Flatmark K. Anti-Angiogenic Treatment in Pseudomyxoma Peritonei-Still a Strong Preclinical Rationale. Cancers. 2021;13:2819. doi: 10.3390/cancers13112819[↩][↩][↩]
- Dohan A., Lousquy R., Eveno C., Goere D., Broqueres-You D., Kaci R., Lehmann-Che J., Launay J.M., Soyer P., Bonnin P., et al. Orthotopic animal model of pseudomyxoma peritonei: An in vivo model to test anti-angiogenic drug effects. Am. J. Pathol. 2014;184:1920–1929. doi: 10.1016/j.ajpath.2014.03.004[↩][↩]
- Kufe D.W. Mucins in cancer: Function, prognosis and therapy. Nat. Rev. Cancer. 2009;9:874–885. doi: 10.1038/nrc2761[↩]
- Thai P., Loukoianov A., Wachi S., Wu R. Regulation of airway mucin gene expression. Annu. Rev. Physiol. 2008;70:405–429. doi: 10.1146/annurev.physiol.70.113006.100441[↩]
- Choudry H.A., Mavanur A., O’Malley M.E., Zeh H.J., Guo Z., Bartlett D.L. Chronic anti-inflammatory drug therapy inhibits gel-forming mucin production in a murine xenograft model of human pseudomyxoma peritonei. Ann. Surg. Oncol. 2012;19:1402–1409. doi: 10.1245/s10434-012-2242-5[↩][↩][↩]
- Gilbreath J.J., Semino-Mora C., Friedline C.J., Liu H., Bodi K.L., McAvoy T.J., Francis J., Nieroda C., Sardi A., Dubois A., et al. A core microbiome associated with the peritoneal tumors of pseudomyxoma peritonei. Orphanet. J. Rare Dis. 2013;8:105. doi: 10.1186/1750-1172-8-105[↩]
- Semino-Mora C., Testerman T.L., Liu H., Whitmire J.M., Studeman K., Jia Y., McAvoy T.J., Francis J., Nieroda C., Sardi A., et al. Antibiotic treatment decreases microbial burden associated with pseudomyxoma peritonei and affects beta-catenin distribution. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2013;19:3966–3976. doi: 10.1158/1078-0432.CCR-13-0616[↩]
- Khamzina Y., King M.C., Nieroda C., Merrell D.S., Sardi A., Gushchin V. The Role of Microorganisms in Appendiceal Pseudomyxoma Peritonei: A Review. Curr. Oncol. 2022;29:3576–3584. doi: 10.3390/curroncol29050289[↩][↩]
- Merrell D.S., McAvoy T.J., King M.C., Sittig M., Millar E.V., Nieroda C., Metcalf J.L., Blum F.C., Testerman T.L., Sardi A. Pre- and post-operative antibiotics in conjunction with cytoreductive surgery and heated intraperitoneal chemotherapy (HIPEC) should be considered for pseudomyxoma peritonei (PMP) treatment. Eur. J. Surg. Oncol. 2019;45:1723–1726. doi: 10.1016/j.ejso.2019.01.223[↩]
- Blythman H.E., Casellas P., Gros O., Gros P., Jansen F.K., Paolucci F., Pau B., Vidal H. Immunotoxins: Hybrid molecules of monoclonal antibodies and a toxin subunit specifically kill tumour cells. Nature. 1981;290:145–146. doi: 10.1038/290145a0[↩]
- Went P., Vasei M., Bubendorf L., Terracciano L., Tornillo L., Riede U., Kononen J., Simon R., Sauter G., Baeuerle P.A. Frequent high-level expression of the immunotherapeutic target Ep-CAM in colon, stomach, prostate and lung cancers. Br. J. Cancer. 2006;94:128–135. doi: 10.1038/sj.bjc.6602924[↩]
- Andersson Y., Juell S., Fodstad O. Downregulation of the antiapoptotic MCL-1 protein and apoptosis in MA-11 breast cancer cells induced by an anti-epidermal growth factor receptor-Pseudomonas exotoxin a immunotoxin. Int. J. Cancer. 2004;112:475–483. doi: 10.1002/ijc.20371[↩]
- Antignani A., FitzGerald D. Immunotoxins: The Role of the Toxin. Toxins. 2013;5:1486–1502. doi: 10.3390/toxins5081486[↩]
- Flatmark K., Guldvik I.J., Svensson H., Fleten K.G., Florenes V.A., Reed W., Giercksky K.E., Fodstad O., Andersson Y. Immunotoxin targeting EpCAM effectively inhibits peritoneal tumor growth in experimental models of mucinous peritoneal surface malignancies. Int. J. Cancer. 2013;133:1497–1506. doi: 10.1002/ijc.28158[↩][↩]
- Froysnes I.S., Andersson Y., Larsen S.G., Davidson B., Oien J.T., Julsrud L., Fodstad O., Dueland S., Flatmark K. ImmunoPeCa trial: Long-term outcome following intraperitoneal MOC31PE immunotoxin treatment in colorectal peritoneal metastasis. Eur. J. Surg. Oncol. 2021;47:134–138. doi: 10.1016/j.ejso.2019.04.014[↩]
- Froysnes I.S., Andersson Y., Larsen S.G., Davidson B., Oien J.T., Olsen K.H., Giercksky K.E., Julsrud L., Fodstad O., Dueland S., et al. Novel Treatment with Intraperitoneal MOC31PE Immunotoxin in Colorectal Peritoneal Metastasis: Results From the ImmunoPeCa Phase 1 Trial. Ann. Surg. Oncol. 2017;24:1916–1922. doi: 10.1245/s10434-017-5814-6[↩]
- Jing X., Yang F., Shao C., Wei K., Xie M., Shen H., Shu Y. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol. Cancer. 2019;18:157. doi: 10.1186/s12943-019-1089-9[↩]
- Masoud G.N., Li W. HIF-1alpha pathway: Role, regulation and intervention for cancer therapy. Acta Pharm. Sin B. 2015;5:378–389. doi: 10.1016/j.apsb.2015.05.007[↩]
- Zimna A., Kurpisz M. Hypoxia-Inducible Factor-1 in Physiological and Pathophysiological Angiogenesis: Applications and Therapies. Biomed. Res. Int. 2015;2015:549412. doi: 10.1155/2015/549412[↩]
- Ma Z., Xiang X., Li S., Xie P., Gong Q., Goh B.C., Wang L. Targeting hypoxia-inducible factor-1, for cancer treatment: Recent advances in developing small-molecule inhibitors from natural compounds. Semin Cancer Biol. 2022;80:379–390. doi: 10.1016/j.semcancer.2020.09.011[↩]
- Xia Y., Choi H.K., Lee K. Recent advances in hypoxia-inducible factor (HIF)-1 inhibitors. Eur. J. Med. Chem. 2012;49:24–40. doi: 10.1016/j.ejmech.2012.01.033[↩]
- Polosukhin V.V., Cates J.M., Lawson W.E., Milstone A.P., Matafonov A.G., Massion P.P., Lee J.W., Randell S.H., Blackwell T.S. Hypoxia-inducible factor-1 signalling promotes goblet cell hyperplasia in airway epithelium. J. Pathol. 2011;224:203–211. doi: 10.1002/path.2863[↩]
- Zhou X., Tu J., Li Q., Kolosov V.P., Perelman J.M. Hypoxia induces mucin expression and secretion in human bronchial epithelial cells. Transl. Res. J. Lab. Clin. Med. 2012;160:419–427. doi: 10.1016/j.trsl.2012.08.001[↩]
- Valenzuela-Molina F., Bura F.I., Vázquez-Borrego M.C., Granados-Rodríguez M., Rufián-Andujar B., Rufián-Peña S., Casado-Adam Á., Sánchez-Hidalgo J.M., Rodríguez-Ortiz L., Ortega-Salas R., et al. Intraoperative oxygen tension and redox homeostasis in Pseudomyxoma peritonei: A short case series. Front. Oncol. 2023;13:1076500. doi: 10.3389/fonc.2023.1076500[↩]
- Dilly A.K., Lee Y.J., Zeh H.J., Guo Z.S., Bartlett D.L., Choudry H.A. Targeting hypoxia-mediated mucin 2 production as a therapeutic strategy for mucinous tumors. Transl. Res. 2016;169:19–30.e1. doi: 10.1016/j.trsl.2015.10.006[↩]
- Liu Z., Yu X., Xu L., Li Y., Zeng C. Current insight into the regulation of PD-L1 in cancer. Exp. Hematol. Oncol. 2022;11:44. doi: 10.1186/s40164-022-00297-8[↩][↩]
- LaFleur M.W., Muroyama Y., Drake C.G., Sharpe A.H. Inhibitors of the PD-1 Pathway in Tumor Therapy. J. Immunol. 2018;200:375–383. doi: 10.4049/jimmunol.1701044[↩]
- Liu B., Song Y., Liu D. Recent development in clinical applications of PD-1 and PD-L1 antibodies for cancer immunotherapy. J. Hematol. Oncol. 2017;10:174. doi: 10.1186/s13045-017-0541-9[↩]
- Yi M., Zheng X., Niu M., Zhu S., Ge H., Wu K. Combination strategies with PD-1/PD-L1 blockade: Current advances and future directions. Mol. Cancer. 2022;21:28. doi: 10.1186/s12943-021-01489-2[↩]
- Gleeson E.M., Feldman R., Mapow B.L., Mackovick L.T., Ward K.M., Morano W.F., Rubin R.R., Bowne W.B. Appendix-derived Pseudomyxoma Peritonei (PMP): Molecular Profiling Toward Treatment of a Rare Malignancy. Am. J. Clin. Oncol. 2018;41:777–783. doi: 10.1097/COC.0000000000000376[↩][↩]
- Kusamura S., Busico A., Conca E., Capone I., Agnelli L., Lorenzini D., Brich S., Angelini M., Volpi C.C., Trupia D.V., et al. A2AR Expression and Immunosuppressive Environment Independent of KRAS and GNAS Mutations in Pseudomyxoma Peritonei. Biomedicines. 2023;11:2049. doi: 10.3390/biomedicines11072049[↩][↩]
- Yi M., Jiao D., Xu H., Liu Q., Zhao W., Han X., Wu K. Biomarkers for predicting efficacy of PD-1/PD-L1 inhibitors. Mol. Cancer. 2018;17:129. doi: 10.1186/s12943-018-0864-3[↩]
- Zhao P., Li L., Jiang X., Li Q. Mismatch repair deficiency/microsatellite instability-high as a predictor for anti-PD-1/PD-L1 immunotherapy efficacy. J. Hematol. Oncol. 2019;12:54. doi: 10.1186/s13045-019-0738-1[↩]
- Li X., Liu G., Wu W. Recent advances in Lynch syndrome. Exp. Hematol. Oncol. 2021;10:37. doi: 10.1186/s40164-021-00231-4[↩]
- Yan F., Lin Y., Zhou Q., Chang H., Li Y. Pathological prognostic factors of pseudomyxoma peritonei: Comprehensive clinicopathological analysis of 155 cases. Hum. Pathol. 2020;97:9–18. doi: 10.1016/j.humpath.2019.12.008[↩]
- Li J.D., Feng W., Gallup M., Kim J.H., Gum J., Kim Y., Basbaum C. Activation of NF-kappaB via a Src-dependent Ras-MAPK-pp90rsk pathway is required for Pseudomonas aeruginosa-induced mucin overproduction in epithelial cells. Proc. Natl. Acad. Sci. USA. 1998;95:5718–5723. doi: 10.1073/pnas.95.10.5718[↩]
- Perrais M., Pigny P., Copin M.-C., Aubert J.-P., Van Seuningen I. Induction of MUC2 and MUC5AC Mucins by Factors of the Epidermal Growth Factor (EGF) Family Is Mediated by EGF Receptor/Ras/Raf/Extracellular Signal-regulated Kinase Cascade and Sp1*. J. Biol. Chem. 2002;277:32258–32267. doi: 10.1074/jbc.M204862200[↩]
- Kuracha M.R., Thomas P., Loggie B.W., Govindarajan V. Bilateral blockade of MEK- and PI3K-mediated pathways downstream of mutant KRAS as a treatment approach for peritoneal mucinous malignancies. PLoS ONE. 2017;12:e0179510. doi: 10.1371/journal.pone.0179510[↩]
- Dilly A.K., Song X., Zeh H.J., Guo Z.S., Lee Y.J., Bartlett D.L., Choudry H.A. Mitogen-activated protein kinase inhibition reduces mucin 2 production and mucinous tumor growth. Transl. Res. 2015;166:344–354. doi: 10.1016/j.trsl.2015.03.004[↩]
- Vázquez-Borrego M.C., Granados-Rodríguez M., Bura F.I., Martínez-López A., Rufián-Andújar B., Valenzuela-Molina F., Rodríguez-Ortiz L., Haro-Yuste S., Moreno-Serrano A., Ortega-Salas R., et al. Antitumor effect of a small-molecule inhibitor of KRAS(G12D) in xenograft models of mucinous appendicular neoplasms. Exp. Hematol. Oncol. 2023;12:102. doi: 10.1186/s40164-023-00465-4[↩]
- Yan W.L., Shen K.Y., Tien C.Y., Chen Y.A., Liu S.J. Recent progress in GM-CSF-based cancer immunotherapy. Immunotherapy. 2017;9:347–360. doi: 10.2217/imt-2016-0141[↩]
- Vijayan D., Young A., Teng M.W.L., Smyth M.J. Targeting immunosuppressive adenosine in cancer. Nat. Rev. Cancer. 2017;17:709–724. doi: 10.1038/nrc.2017.86[↩]
- Khanna S., Graef S., Mussai F., Thomas A., Wali N., Yenidunya B.G., Yuan C., Morrow B., Zhang J., Korangy F., et al. Tumor-Derived GM-CSF Promotes Granulocyte Immunosuppression in Mesothelioma Patients. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018;24:2859–2872. doi: 10.1158/1078-0432.CCR-17-3757[↩]
- Hatfield S.M., Sitkovsky M. A2A adenosine receptor antagonists to weaken the hypoxia-HIF-1α driven immunosuppression and improve immunotherapies of cancer. Curr. Opin. Pharmacol. 2016;29:90–96. doi: 10.1016/j.coph.2016.06.009[↩]
- Overman MJ, Fournier K, Hu CY, Eng C, Taggart M, Royal R, Mansfield P, Chang GJ. Improving the AJCC/TNM staging for adenocarcinomas of the appendix: the prognostic impact of histological grade. Ann Surg. 2013 Jun;257(6):1072-8. doi: 10.1097/SLA.0b013e318269d680[↩]
- Carr N.J., Cecil T.D., Mohamed F., Sobin L.H., Sugarbaker P.H., González-Moreno S., Taflampas P., Chapman S., Moran B.J. A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am. J. Surg. Pathol. 2016;40:14–26. doi: 10.1097/PAS.0000000000000535[↩][↩]
- Baratti D, Kusamura S, Milione M, Bruno F, Guaglio M, Deraco M. Validation of the Recent PSOGI Pathological Classification of Pseudomyxoma Peritonei in a Single-Center Series of 265 Patients Treated by Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy. Ann Surg Oncol. 2018 Feb;25(2):404-413. doi: 10.1245/s10434-017-6252-1[↩]
- Pallas N, Karamveri C, Kyziridis D, Hristakis C, Kyriakopoulos V, Kalakonas A, Vaikos D, Tentes AK. Cytoreductive surgery and hyperthermic intraperitenoal chemotherapy (HIPEC) for colorectal and appendiceal carcinomas with peritoneal carcinomatosis. J BUON. 2017 Nov-Dec;22(6):1547-1553. https://www.jbuon.com/archive/22-6-1547.pdf[↩]
- McQuellon RP, Russell GB, Shen P, Stewart JH 4th, Saunders W, Levine EA. Survival and health outcomes after cytoreductive surgery with intraperitoneal hyperthermic chemotherapy for disseminated peritoneal cancer of appendiceal origin. Ann Surg Oncol. 2008 Jan;15(1):125-33. doi: 10.1245/s10434-007-9678-z[↩]
- Moran B, Cecil T, Chandrakumaran K, Arnold S, Mohamed F, Venkatasubramaniam A. The results of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in 1200 patients with peritoneal malignancy. Colorectal Dis. 2015 Sep;17(9):772-8. doi: 10.1111/codi.12975[↩]
- Chua TC, Yan TD, Saxena A, Morris DL. Should the treatment of peritoneal carcinomatosis by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy still be regarded as a highly morbid procedure?: a systematic review of morbidity and mortality. Ann Surg. 2009 Jun;249(6):900-7. doi: 10.1097/SLA.0b013e3181a45d86[↩]
- Järvinen P, Ristimäki A, Kantonen J, Aronen M, Huuhtanen R, Järvinen H, Lepistö A. Comparison of serial debulking and cytoreductive surgery with hyperthermic intraperitoneal chemotherapy in pseudomyxoma peritonei of appendiceal origin. Int J Colorectal Dis. 2014 Aug;29(8):999-1007. doi: 10.1007/s00384-014-1933-8[↩]
- Sugarbaker PH, Ryan DP. Cytoreductive surgery plus hyperthermic perioperative chemotherapy to treat peritoneal metastases from colorectal cancer: standard of care or an experimental approach? Lancet Oncol. 2012 Aug;13(8):e362-9. doi: 10.1016/S1470-2045(12)70210-3[↩]
- Spratt JS, Adcock RA, Muskovin M, Sherrill W, McKeown J. Clinical delivery system for intraperitoneal hyperthermic chemotherapy. Cancer Res. 1980 Feb;40(2):256-60.[↩]
- Kusamura S., Barretta F., Yonemura Y., Sugarbaker P.H., Moran B.J., Levine E.A., Goere D., Baratti D., Nizri E., Morris D.L., et al. The Role of Hyperthermic Intraperitoneal Chemotherapy in Pseudomyxoma Peritonei After Cytoreductive Surgery. JAMA Surg. 2021;156:e206363. doi: 10.1001/jamasurg.2020.6363[↩]
- Li Y., Yu Y., Liu Y. Report on the 9(th) International Congress on Peritoneal Surface Malignancies. Cancer Biol. Med. 2014;11:281–284. doi: 10.7497/j.issn.2095-3941.2014.04.008[↩]
- Moran B., Baratti D., Yan T.D., Kusamura S., Deraco M. Consensus statement on the loco-regional treatment of appendiceal mucinous neoplasms with peritoneal dissemination (pseudomyxoma peritonei) J. Surg. Oncol. 2008;98:277–282. doi: 10.1002/jso.21054[↩]
- Mercier F., Dagbert F., Pocard M., Goere D., Quenet F., Wernert R., Dumont F., Brigand C., Passot G., Glehen O., et al. Recurrence of pseudomyxoma peritonei after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. BJS Open. 2019;3:195–202. doi: 10.1002/bjs5.97[↩]
- Prabhu A., Brandl A., Wakama S., Sako S., Ishibashi H., Mizumoto A., Takao N., Noguchi K., Motoi S., Ichinose M., et al. Neoadjuvant Intraperitoneal Chemotherapy in Patients with Pseudomyxoma Peritonei-A Novel Treatment Approach. Cancers. 2020;12:2212. doi: 10.3390/cancers12082212[↩]
- Ronnett B.M., Zahn C.M., Kurman R.J., et al. Disseminated peritoneal adenomucinosis and peritoneal mucinous carcinomatosis. A clinicopathologic analysis of 109 cases with emphasis on distinguishing pathologic features, site of origin, prognosis, and relationship to “pseudomyxoma peritonei”. Am. J. Surg. Pathol. 1995;19:1390–1408. doi: 10.1097/00000478-199512000-00006[↩]
- Amin M.B., Edge S.B., Greene F.L., Byrd D.R., Brookland R.K., Washington M.K., Gershenwald J.E., Compton C.C., Hess K.R., Sullivan D.C., et al. AJCC Cancer Staging Manual. 8th ed. Springer; New York, NY, USA: 2017.[↩]
- Wheeler BR, Reddy SK, Kenwright D, Keating JP. Treating pseudomyxoma peritonei without heated intraperitoneal chemotherapy–a first look in New Zealand. N Z Med J. 2014 Feb 14;127(1389):31-9.[↩][↩]
- Alexander-Sefre F, Chandrakumaran K, Banerjee S, Sexton R, Thomas JM, Moran B. Elevated tumor markers prior to complete tumor removal in patients with pseudomyxoma peritonei predict early recurrence. Colorectal Dis. 2005;7:382–6. doi: 10.1111/j.1463-1318.2005.00773.x[↩][↩]
- Li C, Kanthan R, Kanthan SC. Pseudomyxoma peritonei–a revisit: report of 2 cases and literature review. World J Surg Oncol. 2006;4:60. doi: 10.1186/1477-7819-4-60[↩][↩]
- Kusamura S, Hutanu I, Baratti D, Deraco M. Circulating tumor markers: predictors of incomplete cytoreduction and powerful determinants of outcome in pseudomyxoma peritonei. J Surg Oncol. 2013;108:1–8. doi: 10.1002/jso.23329[↩][↩]
- Smeenk RM, Bruin SC, van Velthuysen ML, Verwaal VJ. Pseudomyxoma peritonei. Curr Probl Surg. 2008;45:527–75. doi: 10.1067/j.cpsurg.2008.04.003[↩][↩]
- de Oliveira AM, Rodrigues C, Borges A, Martins A, dos Santos S, Pires F, et al. Pseudomyxoma peritonei: a clinical case of this poorly understood condition. Int J Gen Med. 2014;7:137–41. doi: 10.2147/IJGM.S51504[↩]