Contents
What is pseudomonas
Pseudomonas infection is caused by strains of bacteria found widely in the environment; the most common type causing infections in humans is called Pseudomonas aeruginosa. Serious Pseudomonas infections usually occur in people in the hospital and/or with weakened immune systems 1. Pseudomonas infections of the blood, pneumonia, and infections following surgery can lead to severe illness and death in these people. However, healthy people can also develop mild illnesses with Pseudomonas aeruginosa, especially after exposure to water. Ear infections, especially in children, and more generalized skin rashes (hot tub rash or Pseudomonas folliculitis) may occur after exposure to inadequately chlorinated hot tubs or swimming pools. Eye infections have occasionally been reported in persons using extended-wear contact lenses.
The genus Pseudomonas contains more than 140 species, most of which are saprophytic (organism which gets its energy from dead and decaying organic matter). More than 25 species are associated with humans 2. Most pseudomonads known to cause disease in humans are associated with opportunistic infections. These include Pseudomonas aeruginosa, Pseudomonas fluorescens, Pseudomonas putida, Pseudomonas cepacia, Pseudomonas stutzeri, Pseudomonas maltophilia, and Pseudomonas putrefaciens. Only two species, Pseudomonas mallei and Pseudomonas pseudomallei, produce specific human diseases: glanders and melioidosis 2. Pseudomonas aeruginosa and Pseudomonas maltophilia account for approximately 80 percent of pseudomonads recovered from clinical specimens. Because of the frequency with which it is involved in human disease, Pseudomonas aeruginosa has received the most attention.
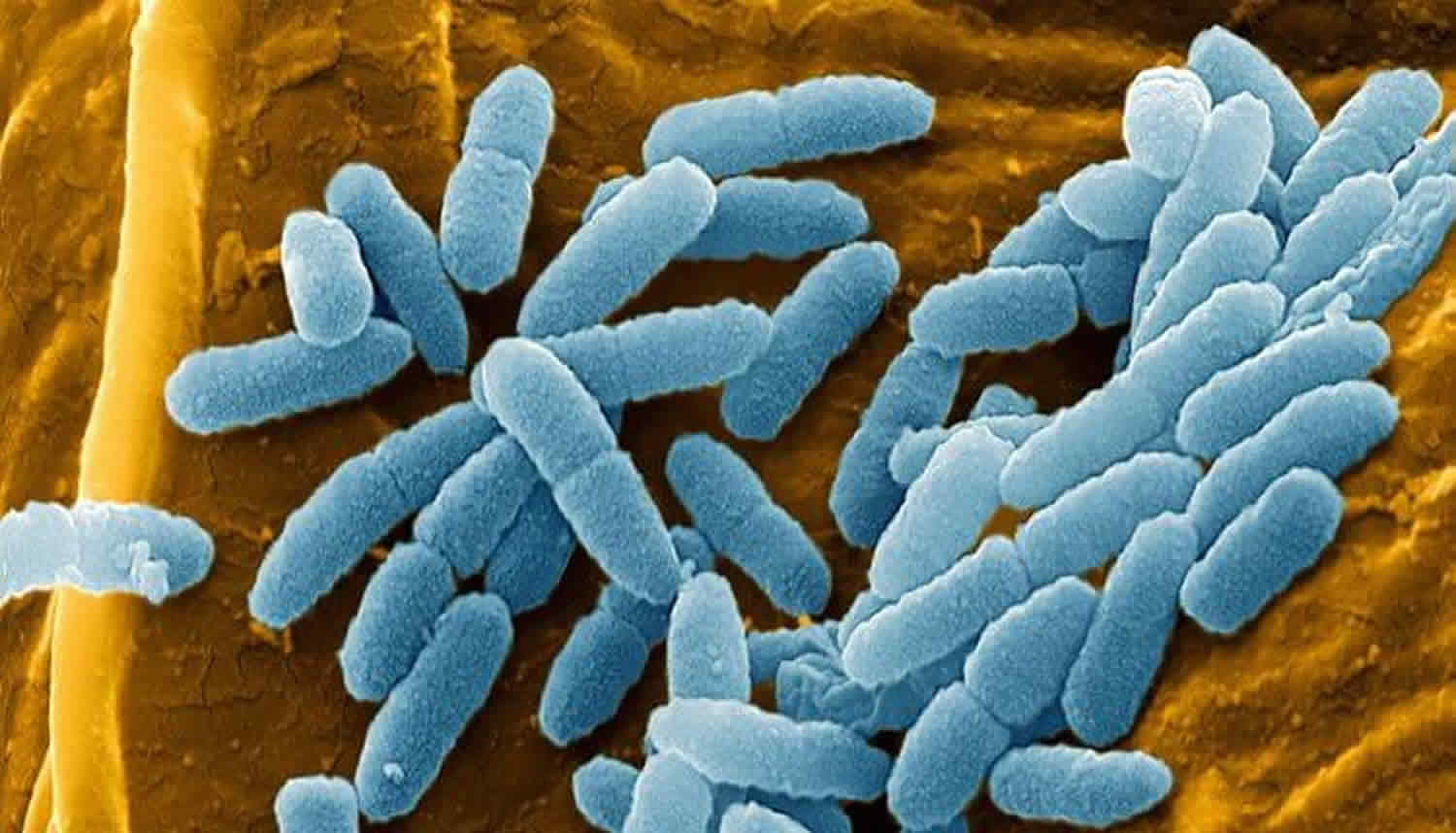
Pseudomonas aeruginosa is a ubiquitous free-living Gram-negative aerobic rod-shaped bacterium that inhabits a wide range of environments, such as water, soil, the rhizosphere (narrow region of soil that is directly influenced by root secretions and associated soil microorganisms), and animals 3. Pseudomonas aeruginosa is found in the skin of some healthy persons and has been isolated from the throat (5 percent) and stool (3 percent) of nonhospitalized patients 2. The gastrointestinal carriage rates increase in hospitalized patients to 20 percent within 72 hours of admission. Within the hospital, Pseudomonas aeruginosa finds numerous reservoirs: disinfectants, respiratory equipment, food, sinks, taps, and mops. Furthermore, it is constantly reintroduced into the hospital environment on fruits, plants, vegetables, and patients transferred from other facilities. Spread occurs from patient to patient on the hands of hospital personnel, by direct patient contact with contaminated reservoirs, and by the ingestion of contaminated foods and water.
Although Pseudomonas aeruginosa seldom causes disease in healthy individuals, it is a major threat to hospitalized patients, particularly those with serious underlying diseases such as cancer and burns. The high mortality associated with these infections is due to a combination of weakened host defenses, bacterial resistance to antibiotics, and the production of extracellular bacterial enzymes and toxins.
Pseudomonas aeruginosa is a versatile opportunistic pathogen causing a wide variety of hospital-acquired acute infections in immunocompromised patients as well as chronic respiratory infections in patients suffering from cystic fibrosis or other chronic respiratory diseases 4. Several traits contribute to its ability to colonize and persist in the lungs of chronically infected patients, including development of high resistance to antimicrobials and hypermutability, biofilm growth, and alginate hyperproduction, or a customized pathogenicity, which may include the loss of classical virulence factors and metabolic changes 4. Prior use of antibiotics and prior hospital or ICU stay was the most significant risk factors for acquisition of resistant Pseudomonas aeruginosa 5.
From the clinical point of view, infections caused by Pseudomonas aeruginosa can be classified as 6:
- Acute superficial, noninvasive, infections in immunocompetent patients.
- This group includes the following entities:
- external otitis (swimmer’s ear),
- hot tub rash (Pseudomonas folliculitis),
- perichondritis,
- keratitis associated with the use of contact lens,
- hydromassage-associated folliculitis,
- paronychia (green nail syndrome),
- palmoplantar hidradenitis,
- foot bones osteomyelitis (secondary to puncture wounds by objects penetrating sport shoes), and
- interdigital intertrigo.
- In all these cases, the infection that follows the exposure to a high Pseudomonas aeruginosa inocula could be self-limited or respond to topical or oral ciprofloxacin treatment, and only exceptionally could pose problems in relation to the presence or development of resistance.
- This group includes the following entities:
- Acute invasive infections in patients with significant comorbidities or immunodepression.
- This group includes:
- bacteremia,
- nosocomial pneumonia or ventilator associated pneumonia,
- endocarditis in parenteral drug users,
- pacemaker infections,
- necrotizing enterocolitis in the neutropenic patient,
- post-surgical meningitis,
- cerebrospinal fluid shunt infection,
- necrotizing fasciitis,
- gangrenous ecthyma,
- tertiary peritonitis or peritonitis associated with ambulatory peritoneal dialysis,
- malignant external otitis,
- central venous catheter infection,
- burn wound infection, and
- urinary tract infection (pyelonephritis or prostatitis) in patients with vesical catheters.
- This group includes:
- Chronic infections. Usually, isolates of Pseudomonas aeruginosa from patients with cystic fibrosis produce an extracellular polysaccharide, alginate, conferring mucoid-type colonies. The same pheno-type could be observed in bronchial infections in patients with bronchiectasis, advanced COPD or panbronchiolitis. These strains are usually less virulent and rarely produce bacteremia or extend beyond the lung. However, growth within biofilms makes difficult its eradication, and in advanced stages it is not possible with current treatments.
Pseudomonas aeruginosa also is known as a frequent opportunistic pathogen in both animals and humans 7. Due to Pseudomonas aeruginosa potential for developing resistance against multiple antibiotic and antiseptic compounds, Pseudomonas aeruginosa also has emerged as a major, difficult-to-treat hospital-acquired pathogen. Pseudomonas aeruginosa is an important cause of morbidity and mortality in high-risk patients, such as immunosuppressed, cystic fibrosis, burn, cancer, and ventilated intensive care patients 8. To date, countless Pseudomonas aeruginosa hospital outbreaks have been described that were caused by patient-to-patient transmission, environmental sources, or contaminated medical devices 9. Pseudomonas aeruginosa also has been reported as a cause of pseudo-outbreaks due to the contamination of media used for clinical specimen collection, transport, or analysis 10.
Pseudomonas infections are generally treated with antibiotics. Unfortunately, in hospitalized patients, Pseudomonas infections, like those caused by many other hospital bacteria, are becoming more difficult to treat because of increasing antibiotic resistance. Selecting the right antibiotic usually requires that a specimen from a patient be sent to a laboratory to test to see which antibiotics might still be effective for treating the infection.
Multidrug-resistant Pseudomonas can be deadly for patients in critical care and Pseudomonas aeruginosa is a major cause of death in patients with cystic fibrosis 4. An estimated 51,000 healthcare-associated Pseudomonas aeruginosa infections occur in the United States each year 1, 11. More than 6,700 (13%) of these are multidrug-resistant, with roughly 400 deaths per year attributed to these infections 12.
The World Health Organization places carbapenem-resistant Pseudomonas aeruginosa as a critical priority pathogen that desperately requires new treatment options 13. Increasing rates of multidrug-resistant Pseudomonas aeruginosa in healthcare-associated infections and among hospitalized patients is a major public health problem 14. Multidrug-resistant Pseudomonas aeruginosa infections in the hospital setting are associated with poor outcomes including increased resource utilization and costs, morbidity, and mortality 15.
In addition to having intrinsic drug resistance mechanisms, Pseudomonas aeruginosa bacterium is able to acquire exogenous genes, resulting in the emergence of multidrug-resistant strains of Pseudomonas aeruginosa, which are resistant to carbapenems, aminoglycosides, and fluoroquinolones 16.
Who is at risk for Pseudomonas infection?
Patients in hospitals, especially those on breathing machines, those with devices such as catheters, and patients with wounds from surgery or from burns are potentially at risk for serious, life-threatening infections.
How is Pseudomonas aeruginosa spread?
In hospitals, where the most serious infections occur, Pseudomonas can be spread on the hands of healthcare workers or by equipment that gets contaminated and is not properly cleaned.
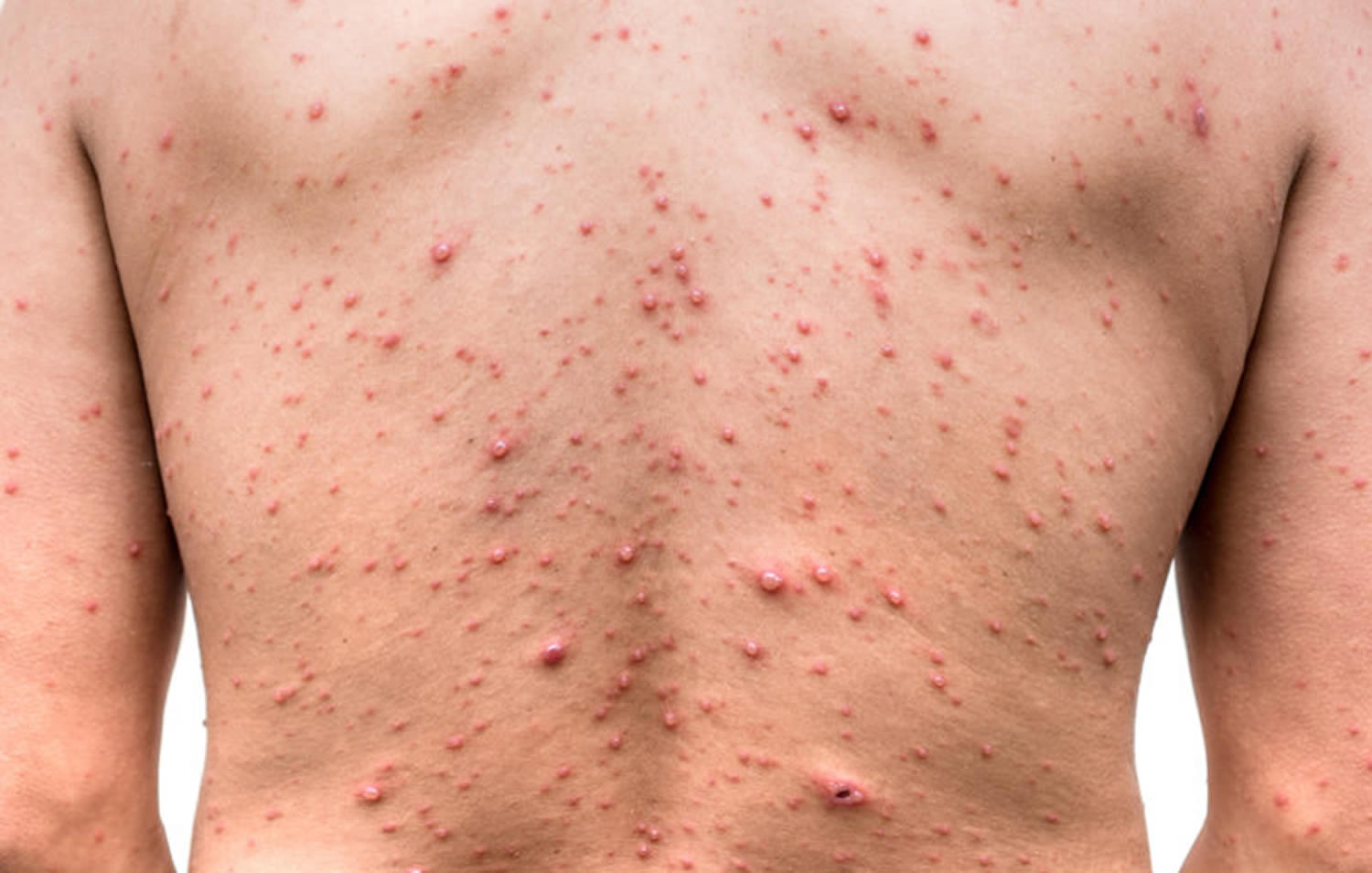
Pseudomonas folliculitis
If contaminated water comes in contact with a person’s skin for a long period of time, it can cause a rash called “hot tub rash” or Pseudomonas folliculitis. Hot tub rash is often caused by infection with the germ Pseudomonas aeruginosa. This germ is common in the environment (for example, in the water and soil) and is microscopic, so it can’t be seen with the naked eye. Hot tub rash can affect people of all ages.
Pseudomonas folliculitis is an infection of the skin. Symptoms of hot tub rash include:
- Itchy spots on the skin that become a bumpy red rash.
- The rash is worse in areas that were previously covered by a swimsuit.
- Pus-filled blisters around hair follicles.
Most hot tub rashes clear up without medical treatment; if your rash lasts longer than a few days, contact a health care provider for treatment.
Your doctor may wish to obtain a bacterial culture of one of the pus-filled bumps in order to establish the diagnosis.
No special therapy is required, typically. However, in severe cases, your doctor may recommend the following:
- Acetic acid compresses. Discomfort associated with the condition can be soothed with the application of cloths soaked in vinegar and applied directly to the affected area for 15 minutes at a time.
- An oral antibiotic, such as ciprofloxacin
- Topical antibiotic cream, such as gentamicin
Figure 1. Pseudomonas folliculitis (hot tub rash)
How is hot tub rash spread at recreational water venues?
Hot tub rash can occur if contaminated water comes in contact with skin for a long period of time. The rash usually appears within a few days of being in a poorly maintained hot tub (or spa), but it can also appear within a few days after swimming in a poorly maintained pool or contaminated lake. Most rashes clear up in a few days without medical treatment. However, if your rash lasts longer than a few days, consult your health care provider.
It is best to thoroughly clean any pools or hot tubs thought to be contaminated and keep chlorine levels in check to avoid a recurrence of this infection.
How do I protect myself, my family, and others?
Because hot tubs have warmer water than pools, chlorine or other disinfectants used to kill germs (like Pseudomonas aeruginosa) break down faster. This can increase the risk of hot tub rash infection for swimmers.
Reduce the Risk of Hot Tub Rash:
- Remove your swimsuit and shower with soap after getting out of the water.
- Clean your swimsuit after getting out of the water.
- Ask your pool/hot tub operator if disinfectant (for example, chlorine) and pH levels are checked at least twice per day—hot tubs and pools with good disinfectant and pH control are less likely to spread germs.
- Use pool test strips to check the pool or hot tub yourself for adequate disinfectant (chlorine or bromine) levels. The Centers for Disease Control and Prevention (CDC) recommends for pools and hot tubs that:
- Pools: free chlorine (1–3 parts per million or ppm)
- Hot Tubs: free chlorine (2–4 ppm) or bromine (4–6 ppm).
- Both hot tubs and pools should have a pH level of 7.2–7.8.
- If you find improper chlorine, bromine, or pH levels, tell the hot tub/pool operator or owner immediately.
What can I ask the hot tub operator?
- What was the most recent health inspection score for the hot tub?
- Are disinfectant and pH levels checked at least twice per day?
- Are disinfectant and pH levels checked more often when the hot tub is being used by a lot of people?
- Are the following maintenance activities performed regularly:
- Removal of the slime or biofilm layer by scrubbing and cleaning?
- Replacement of the hot tub water filter according to manufacturer’s recommendations?
- Replacement of hot tub water?
How can Pseudomonas infections be prevented?
In the hospital, careful attention to routine infection control practices, especially hand hygiene and environmental cleaning, can substantially lower the risk of infection.
Outside the hospital, avoid hot tubs or pools that may be poorly maintained, and keep contact lenses, equipment, and solutions from becoming contaminated.
Residential Pool or Hot Tub Owners
Owning an at-home pool or hot tub can be fun for the whole family. However, it is important for people who own pools or hot tubs in their homes to know how to take care of them and keep themselves and their families healthy and safe.
Chlorine and pH
Chlorine and pH, are the first defense against germs that can make swimmers sick. As a residential pool or hot tub/spa owner, it is your responsibility to regularly check the chlorine concentration and pH of the pool or hot tub/spa water to help protect yourself and your family and friends from recreational water illnesses.
What does chlorine do?
Chlorine is added to the water to kill germs. But it does not work right away. If used properly, free chlorine (free available chlorine is the more active form of chlorine that kills germs) can kill most germs within a few minutes. The Centers for Disease Control and Prevention (CDC) recommends pH 7.2–7.8 and a free chlorine concentration of at least 1 ppm in pools and at least 3 ppm in hot tubs/spas.
If using cyanuric acid, a chlorine stabilizer, or chlorine products with cyanuric acid (for example, products commonly known as dichlor or trichlor [see product label]), CDC recommends pH 7.2–7.8 and a free available chlorine concentration of at least 2 ppm in pools. CDC recommends not using cyanuric acid or chlorine products with cyanuric acid in hot tubs/spas.
Why is pH important?
Three reasons. First, as pH goes up, the ability of free chlorine to kill germs decreases, especially if pH is >8.0. Second, as pH goes down, especially if pH is <7.0, the ability of free chlorine to kill germs increases but the pool or hot tub/spa pipes are more likely to corrode or break down. Third, keeping the pH in the 7.2–7.8 range helps keep swimmers comfortable in the water by helping to prevent eye and skin irritation. This means keeping the pH in the 7.2–7.8 range best balances killing germs to prevent recreational water illness, the lifespan of the pipes, and swimmers’ comfort.
Table 1. pH and its effects
| Water Quality | pH |
| – Poor Chlorine Disinfection – Eye Irritation – Skin Irritation | > 8.0 |
| – Most Ideal for Eye Comfort and Killing Germs | 7.8 7.6 7.2 |
| – Eye Irritation – Skin Irritation – Pipe Corrosion | < 7.0 |
How do I test chlorine and pH levels in my pool?
For most accurate testing results, pool and hot tub/spa owners should use a DPD (N,N Diethyl-1,4 Phenylenediamine Sulfate) test kit to measure free chlorine and pH. Of note, if the free chlorine concentration of the water sampled from the pool or hot tub/spa is more than 10 ppm, the test might partially or totally bleach out, resulting in a false low or 0 ppm free chlorine result. Pool and hot tub/spa owners alternatively can use test strips, with the understanding they are less accurate than the DPD (N,N Diethyl-1,4 Phenylenediamine Sulfate) test kit. Regardless of what test is used, owners should always follow manufacturer’s directions.
How often should I test chlorine and pH levels in my pool?
Test free chlorine concentration and pH at least twice per day and more often when the pool or hot tub/spa is being used often.
Why does chlorine need to be tested regularly?
All sorts of things can reduce chlorine concentration in pool or hot tub/spa water. Free available chlorine breaks down pee, poop, sweat, and dirt from swimmers’ bodies instead of killing germs and uses up chlorine, which means there is less to kill germs. The sunlight and hot tub/spa jets creating droplets or mists from the water also uses up free chlorine. That’s why the free chlorine concentration must be routinely tested. And remember, the time it takes for free chlorine to kill germs is also affected by the other member of the disinfection team, pH.
How quickly does chlorine kill common illness-causing germs?
Free chlorine kills most bacteria, such as E. coli 0157:H7, in less than a minute if its concentration and pH are maintained as CDC recommends. However, a few germs are moderately (Giardia, Hepatitis A) to very (Cryptosporidium) chlorine tolerant. The table below shows the approximate times it takes for free chlorine to kill these germs.
| Free Available Chlorine Germ-Killing Timetable | |
|---|---|
| E. coli 0157:H7 (Bacterium) | less than 1 minute |
| Hepatitis A (Virus) | approximately 16 minutes |
| Giardia (Parasite) | approximately 45 minutes |
| Cryptosporidium (Parasite) | approximately 15,300 minutes (10.6 days) |
Footnotes:
- Times based on 1 ppm free chlorine at pH 7.5 and 77°F (25°C)
- These disinfection times are only for pools and hot tubs/spas that do not use cyanuric acid. Disinfection times are longer in the presence of cyanuric acid.
Pseudomonas infections
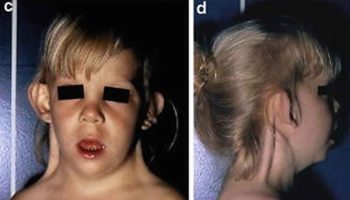
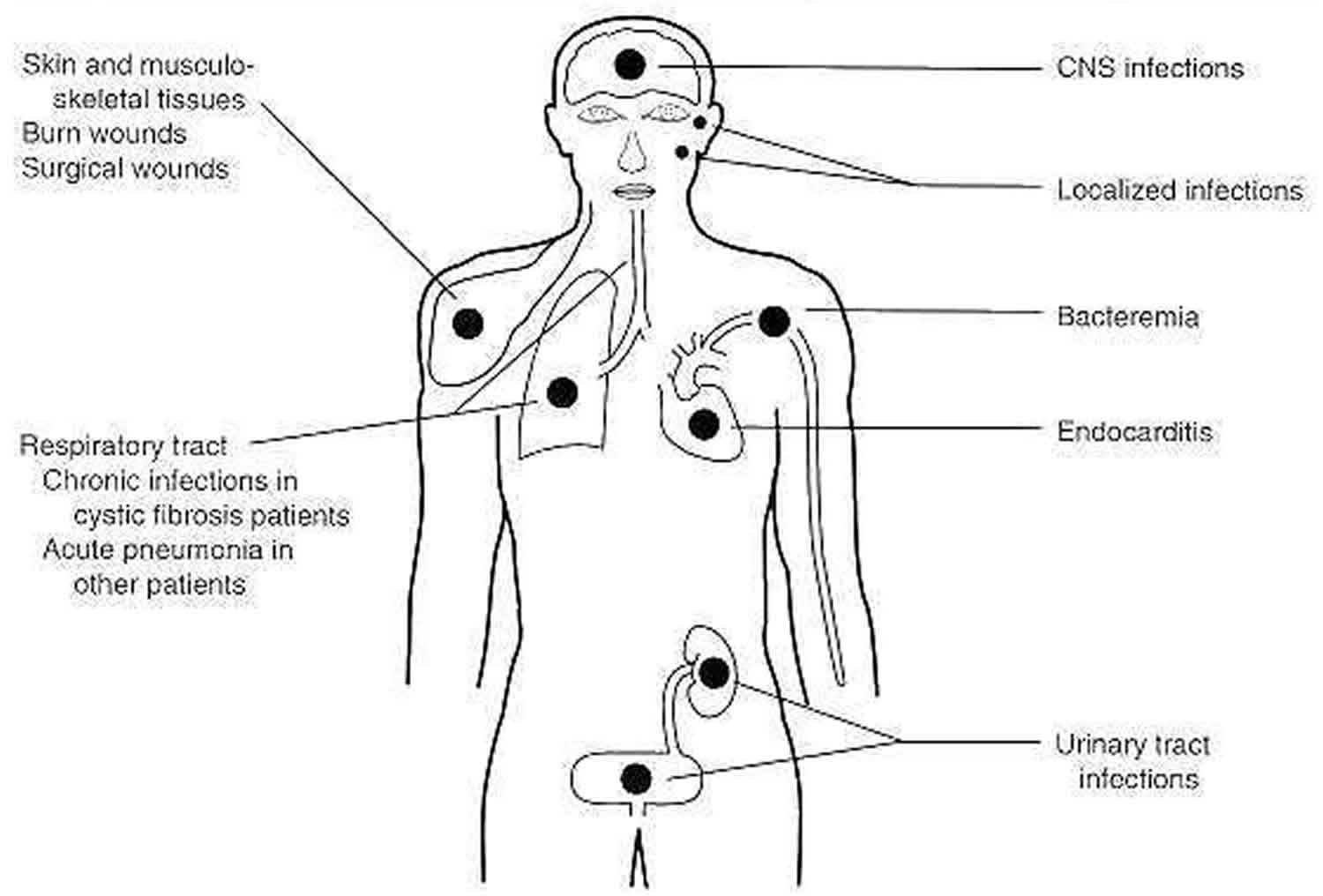
Pseudomonas aeruginosa an opportunistic pathogen may infect virtually any tissue and can cause various diseases (Figure 2). Pseudomonas aeruginosa infection is facilitated by the presence of underlying disease (e.g., cancer, cystic fibrosis) or by a breakdown in nonspecific host defenses (as in burns) 2. Localized infection following surgery or burns commonly results in a generalized and frequently fatal bacteremia. Urinary tract infections (UTIs) following introduction of Pseudomonas aeruginosa on catheters or in irrigating solutions are not uncommon. Furthermore, most cystic fibrosis patients are chronically colonized with Pseudomonas aeruginosa. Interestingly, cystic fibrosis patients rarely have Pseudomonas aeruginosa bacteremia, probably because of high levels of circulating Pseudomonas aeruginosa antibodies. However, most cystic fibrosis patients ultimately die of localized Pseudomonas aeruginosa infections. Necrotizing Pseudomonas aeruginosa pneumonia may occur in other patients following the use of contaminated respirators. Pseudomonas aeruginosa can cause severe corneal infections following eye surgery or injury. It is found in pure culture, especially in children with middle ear infections. It occasionally causes meningitis following lumbar puncture and endocarditis following cardiac surgery. It has been associated with some diarrheal disease episodes. Since the first reported case of Pseudomonas aeruginosa infection in 1890, the organism has been increasingly associated with bacteremia and currently accounts for 15 percent of cases of Gram-negative bacteremia. The overall mortality associated with Pseudomonas aeruginosa bacteremia is about 50 percent. Some infections (e.g., eye and ear infections) remain localized; others, such as wound and burn infections and infections in leukemia and lymphoma patients, result in sepsis. The difference is most probably due to altered host defenses.
Figure 2. Pseudomonas aeruginosa infections
 [Source 2]
[Source 2]Pseudomonas maltophilia is the second most frequently isolated pseudomonad species in clinical laboratories. In nature, Pseudomonas maltophilia is found in water and in both raw and pasteurized milk. It has been associated with a variety of opportunistic infections in humans, including pneumonia, endocarditis, urinary tract infections, wound infections, septicemia, and meningitis.
Pseudomonas cepacia, although primarily a plant pathogen (onion bulb rot), also is an opportunist. Most human infections caused by Pseudomonas cepacia are nosocomial and include endocarditis, necrotizing vasculitis, pneumonia, wound infections, and urinary tract infections. Pseudomonas cepacia causes chronic lung infections in cystic fibrosis patients. These infections differ from those caused by Pseudomonas aeruginosa in that Pseudomonas cepacia has become systemic in a number of cystic fibrosis patients, whereas Pseudomonas aeruginosa infections remain confined to the lungs. Pseudomonas cepacia is highly resistant to aminoglycosides and other antibiotics, making it very difficult to control.
Unlike most pseudomonads, Pseudomonas mallei and Pseudomonas pseudomallei can cause disease in otherwise healthy individuals. Pseudomonas mallei is the agent of glanders, a disease primarily of equines (horses). Humans generally become infected by inhalation or by direct contract through abraded skin. These infections are frequently fatal within 2 weeks of onset, although chronic infections also have been reported. Today, Pseudomonas mallei infections of equines are controlled and are rarely encountered in the western world. Similarly, melioidosis, an endemic glanderslike disease of animals and a human pulmonary infection caused by Pseudomonas pseudomallei, is rare in the western hemisphere. Melioidosis is still found in Southeast Asia, and travelers returning from that area are sometimes infected.
Pseudomonas aeruginosa bloodstream infection
Pseudomonas aeruginosa bloodstream infection is a serious disease that requires prompt attention and pertinent clinical decisions in order to achieve a satisfactory outcome. Currently, Pseudomonas spp. represent a leading cause of hospital-acquired bacteremia, accounting for 4% of all cases and being the third leading cause of gram-negative bloodstream infection 18. Several studies indicate an increased risk of death among patients with Pseudomonas aeruginosa bloodstream infection, as compared with the risk for similar patients with other gram-negative or Staphylococcus aureus bloodstream infection 19. Therefore, the adequate management of Pseudomonas aeruginosa should be considered as a significant challenge for clinicians.
Once Pseudomonas aeruginosa is isolated from blood, efforts should be made to establish the source of the infection and to choose an appropriate empirical antibiotic therapy as soon as possible. As for the source of the infection, most patients have an identifiable focus of infection at the time of initial evaluation, but the source remains unknown in up to 40% of the cases 19. The most common sources of Pseudomonas aeruginosa bloodstream infection are in respiratory (25%) and urinary tract (19%) followed by central venous catheter and skin and soft tissues 19. Identification of the source is essential because its adequate control represents a critical issue in the correct management of Pseudomonas aeruginosa infection (i.e. early CVC removal or surgical drainage of abscess). Accordingly, a careful patient history and a physical examination, as well as radiological and microbiological work-up are important.
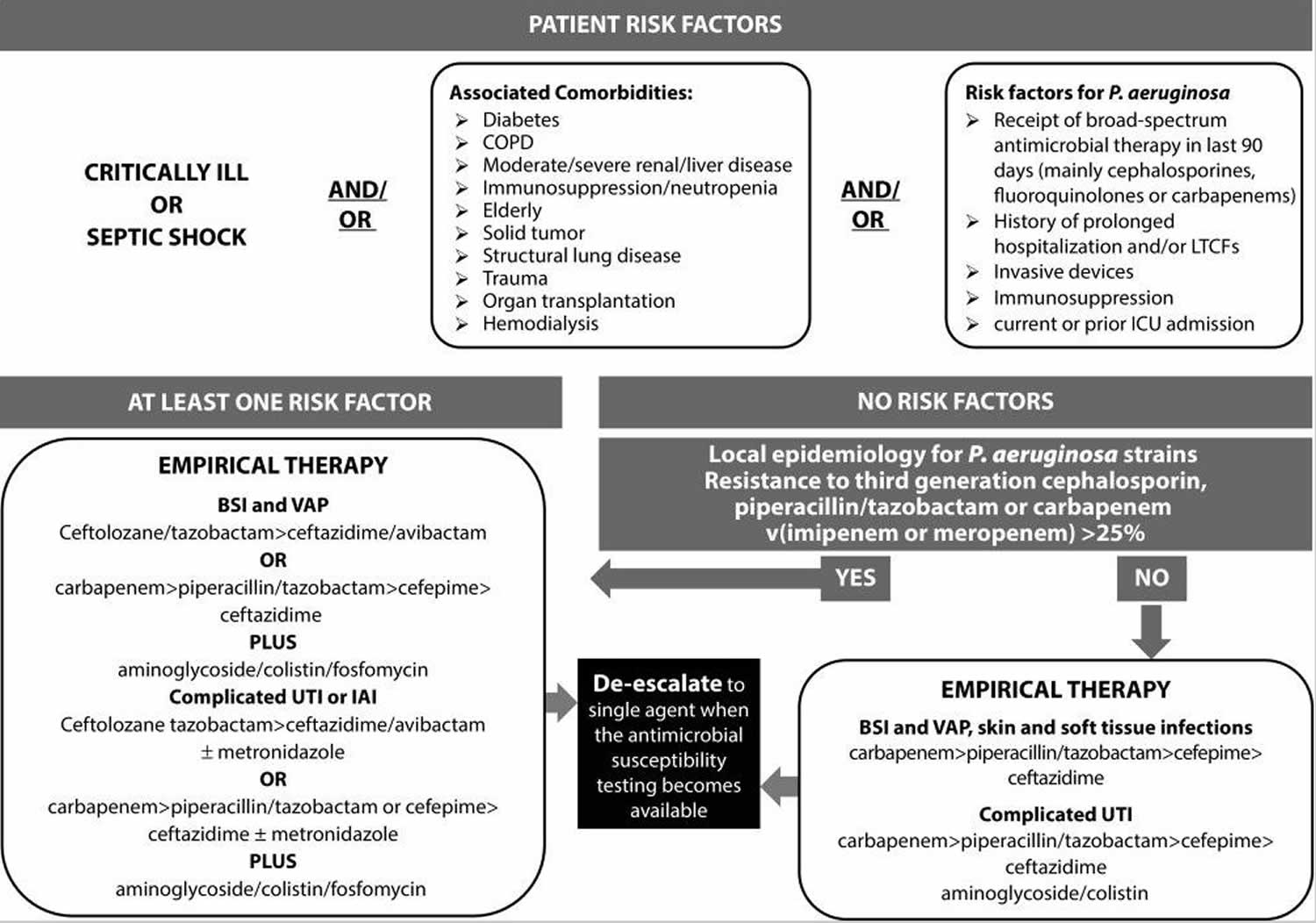
Empirical antibiotic therapy should include two agents from different classes with in vitro activity against Pseudomonas aeruginosa for all serious infections known or suspected to be caused by Pseudomonas aeruginosa. The rationale of the so-called ‘double coverage effect’ is to increase the likelihood that antibiotic treatment will be active against Pseudomonas aeruginosa, especially in the setting of a high-risk of antimicrobial resistance. Once results of susceptibility are available, definitive therapy should be tailored accordingly, using a single in vitro active agent with the highest antimicrobial activity and the lowest propensity to select resistance. Indeed, at the time of the present review, no convincing data exist demonstrating a mortality benefit to combination therapy (Figure 3) 20.
Figure 3. Pseudomonas aeruginosa infection patient management algorithm
Pseudomonas aeruginosa community-acquired pneumonia
Pseudomonas aeruginosa has been reported as a rare cause of community-acquired pneumonia (community-acquired pneumonia), affecting 1.1–8.3% of the patients requiring ICU admission 22. Despite this, Pseudomonas aeruginosa is actually considered the pathogen with the highest attributable mortality rate, ranging from 50 to 100% 22. The survival of these patients is related to early diagnosis and prompt initiation of adequate antibiotic therapy 23. Since antibiotic therapy for Pseudomonas aeruginosa is completely different from the standard therapy to treat common pathogens in community-acquired pneumonia, current guidelines stratify therapy recommendations on the basis of pseudomonal risk factors 24. Community-acquired pneumonia due to Pseudomonas aeruginosa should be considered in immunocompromised subjects (i.e. HIV patients, solid organ transplant) who received prior antibiotic use and with structural abnormalities such as cystic fibrosis, bronchiectasis and COPD (especially those requiring frequent corticosteroid therapy and/or antibiotic use) 24. Additional risk factors include male sex, low C-reactive protein and PSI risk class IV and V 23. Risk factors associated with the isolation of multi-drug resistance Pseudomonas aeruginosa in community-acquired pneumonia have been recently studied in a recent article including more than 2000 patients with community-acquired pneumonia, where the only risk factor was previous antibiotic therapy 23.
Therefore, from a clinical point of view, experts suggest use of antibiotics covering multi-drug resistance Pseudomonas aeruginosa in community-acquired pneumonia only when Pseudomonas aeruginosa is highly suspected.
Pseudomonas aeruginosa ventilator-associated pneumonia
Pseudomonas aeruginosa is one of the leading causes of ventilator-associated pneumonia (ventilator-associated pneumonia) in the US and Europe 25. Ventilator-associated pneumonia due to Pseudomonas aeruginosa is increasing in incidence and poses unique challenges for its clinical management. Risk factors for the development of Pseudomonas aeruginosa-related ventilator-associated pneumonia include prolonged mechanical ventilation 26, older age 26, prior Pseudomonas aeruginosa colonization 25, prior antibiotic therapy 25, admission to a ward with high incidence of Pseudomonas aeruginosa infections 26, solid cancer, and shock 25.
Recent data suggest that a diagnosis of Pseudomonas aeruginosa-related ventilator-associated pneumonia is frequently associated with the isolation of multi-drug resistance pathogens 25. multi-drug resistance Pseudomonas aeruginosa-related pneumonia appears to be an important determinant of excess length of stay in ICU, and prolonged mechanical ventilation, as well as a cause of increased in-hospital mortality compared to non-multi-drug resistance infection 27.
We recommend prescribing two anti-pseudomonal antibiotics from different classes as the initial therapy of Pseudomonas aeruginosa-related ventilator-associated pneumonia, especially when patients are hospitalized in units where >20% of Gram-negative isolates are resistant to a ‘backbone’ agent considered for monotherapy. Once the antibiotic susceptibility testing is known, monotherapy using an antibiotic to which the isolate is susceptible can be considered, except for patients who have septic shock or a high risk of death. An anti-pseudomonal cephalosporin, or a carbapenem, or an anti-pseudomonal β-lactam or β-lactamase inhibitors represent potential options for definitive therapy. Aminoglycosides should not be used as monotherapy because success rates for aminoglycosides are low 28. This may be due to the poor penetration of aminoglycosides into the lung, which require high peak serum concentrations to obtain adequate lung concentrations, thus increasing the risk of nephrotoxicity or ototoxicity 29. However, because in Europe fluoroquinolone resistance rate in Pseudomonas aeuruginosa exceeds 30% 30, experts suggest the use of combination therapy including aminoglycosides for empirical therapy of serious ventilator-associated pneumonia, if it is appropriately tailored on the basis of susceptibility data (Figure 3). As for aerosol therapy, experts do not routinely recommend the use of inhaled antibiotics for the treatment of Pseudomonas aeruginosa ventilator-associated pneumonia. However, they may be considered as an adjunctive to intravenous therapy in cases of infections due to multi-drug resistance strains.
Pseudomonas aeruginosa urinary tract infection
Patients with Pseudomonas aeruginosa urinary tract infection (UTI) are more likely to have chronic underlying disease (e.g. hypertension, cognitive impairment, diabetes mellitus) 31, present with healthcare-associated infection and recent urinary tract instrumentation and/or chronic indwelling urinary catheters. Mortality rates are higher and reported with rates up to 20% [94]. Advanced chronic disease and inadequate definitive antimicrobial treatment are associated with worse prognosis 31. Standard of care consists of elimination of the predisposing condition in combination with a single antibiotic therapy that is generally considered adequate for treatment 31 in absence of septic shock (Figure 3).
Pseudomonas aeruginosa skin and soft tissue infections
Pseudomonas aeruginosa causes a variety of skin and soft tissue infections ranging from the benign (e.g. cellulitis, post-surgical infections) to the immediately life threatening. Pseudomonas aeruginosa is one of the most common pathogen isolated from cellulitis in neutropenic patients, surgical site infections, infections following trauma or infections of chronic decubitus ulcers. Although combined antimicrobial and surgical debridement should be considered as standard of care, medical therapy alone maybe sufficient for some patients. For example, in acute cellulitis surgery is generally not necessary, whereas a surgical site infection or an infection of chronic decubitus ulcers requires surgical debridement to remove necrotic tissue. In all cases the importance of antimicrobial therapy is unquestioned. The optimal antibiotic regimen depends on in vitro susceptibility testing and includes an anti-pseudomonal beta-lactam, a carbapenem, or a fluoroquinolone. Although the usual duration of antibiotic therapy is 10 to 14 days, shorter courses could be considered in patients who received an adequate source control of infection and/or with prompt resolution of clinical signs and symptoms.
Among skin and soft tissue infection due to Pseudomonas aeruginosa, two clinical syndromes need special considerations: (1) ecthyma gangrenosum and (2) burn wound infections.
Ecthyma gangrenosum, classically reported in the setting of Pseudomonas aeruginosa blood stream infection in neutropenic patients, is a cutaneous vasculitis caused by bacterial invasion of the media and adventitia of the vessel wall with secondary ischemic necrosis 32. The lesion frequently begins as painless erythematous areas with papules and/or bullae that often rapidly progress becoming painful gangrenous ulcers 33. These lesions may be single or multiple and, although they can occur at any anatomic district, they are preferentially found in the gluteal and perineal areas. Once ecthyma gangrenosum is clinically suspected, prompt collection of blood cultures, culture of exudates from an aspirate or swab of lesion or skin biopsy should be collected to isolate Pseudomonas aeruginosa or other uncommon cause of viral, bacterial, mycobacterial or fungal pathogens potentially responsible 34.
As for therapy, experts suggest administration of empirical combination therapy with beta lactams and aminoglycosides for high risk severe patients (i.e. neutropenic patients, severe immunocompromised patients or patients with septic shock) until the causative organism and its susceptibility are identified. Surgery is not generally warranted but extensive debridement could be required in patients with extensive necrosis (Figure 3) 35.
Regarding burn wound infection, Pseudomonas aeruginosa represents one of the most common pathogen-causing infection in burn injuries 36. Due to the severity of the patient’s condition and frequent antibiotic resistance, Pseudomonas aeruginosa is a dreaded cause of infection in such populations 37. Based on previous studies 38, the rate of positive swab or tissue culture results due to Pseudomonas aeruginosa in burn infection was as high as 57% whereas the proportion of bacteremia caused by Pseudomonas aeruginosa in burned populations was reported to be approximately 15% 39.
Superficial wound infections due to Pseudomonas aeruginosa have a characteristic yellow or green color with a malodorous smell. This infection may evolve to an invasive disease causing blue-purplish lesion of the skin 40.
Similar to ecthyma gangrenosum, experts also suggest, for burns wound infections, administration of empirical combination therapy until the causative organism and its susceptibility are identified. Surgical debridement can be indicated in some cases 40.
Pseudomonas aeruginosa treatment
Early administration of an adequate antibiotic regimen has been associated with favorable clinical outcome, especially among critically ill patients presenting with severe Pseudomonas aeruginosa infections 41; conversely, a delay in the prescription of an adequate antibiotic therapy has been related to a significant increase in mortality.
In recent years, the progressive increase in antibiotic resistance among Pseudomonas aeruginosa has been identified as the main reason for antibiotic inadequacy, with a negative impact on patient survival 42. The available evidence suggests that the greatest benefit of a combination therapy stems from the increased likelihood of choosing an effective agent during empirical therapy rather than to prevent the resistance during definitive therapy or to benefit of in vitro synergistic activity. Therefore, to balance between early antibiotic administration and risk of resistance selection, we suggest early administration of a combination regimen when Pseudomonas aeruginosa is suspected, followed by a prompt de-escalation when the antimicrobial susceptibility testing becomes available. Experts encourage an approach consisting of the prescription of an anti-pseudomonal β-lactam (piperacillin/tazobactam, ceftolozane/tazobactam, ceftazidime, cefepime, or a carbapenem) plus a second anti-pseudomonal agent (aminoglycoside or a fluoroquinolones).
Pseudomonas antibiotics
Standard antibiotic therapy may be inferior to some new comparator agents in the treatment of serious Pseudomonas aeruginosa infections, especially in the setting of increased antimicrobial resistance. Novel antibiotics with activity against Pseudomonas aeruginosa have become available in Europe in recent years and others are in advanced stages of clinical development (Table 2). In some cases, indirect evidence suggests their possible superiority over standard anti-pseudomonal regimens (Table 3) 43.
Ceftolozane-tazobactam
Ceftolozane-tazobactam is being developed to overcome Pseudomonas aeruginosa antimicrobial mechanisms of resistance, such as changes in porin permeability and upregulation of efflux pumps. This drug has an intrinsic potent anti-pseudomonal activity, because of a greater affinity for all essential penicillin binding proteins, including PBP1b, PBP1c and PBP3 44. Ceftolozane/Tazobactam has proven to have a potent in vitro activity against the majority of multi-drug resistance Pseudomonas aeruginosa strains (including ESBL but not carbapenemase producing strains). The US FDA has proposed clinical use of ceftolozane-tazobactam in complicated intra-abdominal and urinary tract infections 45. In addition, a study for treatment of hospital-acquired pneumonia including ventilator-associated pneumonia is currently ongoing. Data belonging to real-world studies using ceftolozane-tazobactam for the treatment of multi-drug resistance Pseudomonas aeruginosa infections showed positive outcomes in 71% of patients with multi-drug resistance Pseudomonas aeruginosa infections 46.
Ceftazidime-avibactam
Ceftazidime-avibactam is a novel β-lactam/β-lactamase inhibitors combination approved by FDA and European Medicines Agency for the treatment of complicated UTIs (urinary tract infections) and complicated intra-abdominal infection. In vitro studies showed that the combination of ceftazidime-avibactam is highly effective against Enterobacteriaceae producing KPCs, ESBLs OXA and AmpC enzymes. However, the drug has no activity against metallo-beta lactamases (MBL, VIM and NDM) and avibactam offers no enhanced activity against Pseudomonas aeruginosa 47. The effectiveness of ceftazidime-avibactam against ventilator-associated pneumonia has been analyzed in a phase III studies comparing this new drug with meropenem (NTC01808092) 48. The predominant Gram-negative pathogens isolated at baseline were Klebsiella pneumoniae and Pseudomonas aeruginosa, with 28% of patients having ≥1 ceftazidime non-susceptible isolate. In the clinically evaluable population, 356 patients received ceftazidime-avibactam and 370 received meropenem. The study met the criteria for non-inferiority of ceftazidime-avibactam since there was no difference between the groups regarding the outcome. Moreover, the efficacy of ceftazidime-avibactam against ceftazidime-non-susceptible strains was similar to that against ceftazidime-susceptible pathogens and was also comparable to meropenem.
Imipenem-cilastatin-relebactam
Relebactam is a diazabicyclooctanes β-lactamase inhibitors that inhibits the activity of class A and C β-lactamase, but does not have any activity against metallo- β-lactamase 49. The combination of imipenem-cilastatin with relebactam has shown to have synergistic activity against a wide spectrum of multi-drug resistance Gram-negative pathogens including Pseudomonas aeruginosa, KPC-producing K. pneumoniae and Enterobacter spp. 50. This drug has been mainly studied in patients with complicated intra-abdominal infection 51, complicated UTI and pyelonephritis 52 whereas a study on patients with hospital-acquired pneumonia or ventilator-associated pneumonia is currently ongoing 21.
Other new drugs such as plazomycin, meropenem-vaborbactam and aztreonam-avibactam have a limited effect on Pseudomonas aeruginosa 43.
Table 2. New drugs and usual clinical dosage for new anti-Pseudomonas agents
| Drug | Current clinical indications | Usual clinical dosage for serious infections | Other comment |
|---|---|---|---|
| Cephalosporins | |||
| Cefiderocol | Complicated UTI | 2 g intravenous every 8 hours | – |
| Cephalosporin + β-lactamase inhibitor | |||
| Ceftolozane-tazobactam | Complicated UTI and IAI | Loading dose 1.5 g or 3 g intravenous in 1 hour, followed by 1.5 g or 3 g intravenous every 8 hours | Extended infusion (over 3 h) 1.5 g or 3 g every 8 hours is recommended |
| Ceftazidime-avibactam | Complicated UTI and IAI, HAP and VAP and Gram-negative infections when other treatments might not work | Loading dose 2.5 g intravenous in 1 hour, followed by 2.5 g intravenous every 8 hours | Extended infusion (over 3 h) 2.5 g every 8 hours is recommended |
| Carbapenem + β-lactamase inhibitor | |||
| Meropenem-vaborbactam | Complicated UTI | 2 g/2 g intravenous every 8 hours | Not active against multi-drug resistance strains |
| Imipenem-relebactam | Not yet approved by any regulatory authority | 500 mg/250 mg intravenous every 6 hours | Not active against multi-drug resistance strains |
| Aminoglycoside | |||
| Plazomicin | Not yet approved by any regulatory authority | 15 mg/kg every 24 hours | – |
Table 3. Advantages and disadvantages of new drugs for Pseudomonas aeruginosa infections
| Advantages |
| High activity against P. aeruginosa including multi-drug resistance strains |
| Predictable PK |
| Good safety profile and tolerability |
| Carbapenem sparing |
| Rapid tissue distribution |
| Disadvantages |
| Increased costs |
| No oral formulations to allow step-down therapy |
| Superinfection with even more resistant bacteria or fungi |
- Pseudomonas aeruginosa in Healthcare Settings. https://www.cdc.gov/hai/organisms/pseudomonas.html[↩][↩]
- Iglewski BH. Pseudomonas. In: Baron S, editor. Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch at Galveston; 1996. Chapter 27. Available from: https://www.ncbi.nlm.nih.gov/books/NBK8326[↩][↩][↩][↩][↩]
- Pseudomonas aeruginosa population structure revisited. Pirnay JP, Bilocq F, Pot B, Cornelis P, Zizi M, Van Eldere J, Deschaght P, Vaneechoutte M, Jennes S, Pitt T, De Vos D. PLoS One. 2009 Nov 13; 4(11):e7740. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2777410/[↩]
- Alexandro Rodríguez-Rojas, Antonio Oliver, Jesús Blázquez; Intrinsic and Environmental Mutagenesis Drive Diversification and Persistence of Pseudomonas aeruginosa in Chronic Lung Infections, The Journal of Infectious Diseases, Volume 205, Issue 1, 1 January 2012, Pages 121–127, https://doi.org/10.1093/infdis/jir690[↩][↩][↩]
- Raman G, Avendano EE, Chan J, Merchant S, Puzniak L. Risk factors for hospitalized patients with resistant or multidrug-resistant Pseudomonas aeruginosa infections: a systematic review and meta-analysis. Antimicrobial Resistance and Infection Control. 2018;7:79. doi:10.1186/s13756-018-0370-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6032536/[↩]
- Mensa J, Barberán J, Soriano A, et al. Antibiotic selection in the treatment of acute invasive infections by Pseudomonas aeruginosa: Guidelines by the Spanish Society of Chemotherapy. Revista Española de Quimioterapia. 2018;31(1):78-100. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6159363/[↩]
- Hallin M, Deplano A, Roisin S, et al. Pseudo-Outbreak of Extremely Drug-Resistant Pseudomonas aeruginosa Urinary Tract Infections Due to Contamination of an Automated Urine Analyzer. Journal of Clinical Microbiology. 2012;50(3):580-582. doi:10.1128/JCM.06268-11. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3295159/[↩]
- Pirnay J-P, Bilocq F, Pot B, et al. Pseudomonas aeruginosa Population Structure Revisited. Ahmed N, ed. PLoS ONE. 2009;4(11):e7740. doi:10.1371/journal.pone.0007740. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2777410/[↩]
- Diaz Granados CA, et al. 2009. Outbreak of Pseudomonas aeruginosa infection associated with contamination of a flexible bronchoscope. Infect. Control Hosp. Epidemiol. 30:550–555[↩]
- Silva CV, et al. 2003. Pseudo-outbreak of Pseudomonas aeruginosa and Serratia marcescens related to bronchoscopes. Infect. Control Hosp. Epidemiol. 24:195–197[↩]
- Community-acquired, hospital-acquired, and healthcare-associated pneumonia caused by Pseudomonas aeruginosa. Fujii A, Seki M, Higashiguchi M, Tachibana I, Kumanogoh A, Tomono K. Respir Med Case Rep. 2014; 12():30-3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4061442/[↩]
- Multidrug-resistant Pseudomonas aeruginosa. https://www.cdc.gov/drugresistance/biggest_threats.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fdrugresistance%2Fthreat-report-2013%2Findex.html#pse[↩]
- Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics. http://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en[↩]
- National surveillance of antimicrobial resistance in Pseudomonas aeruginosa isolates obtained from intensive care unit patients from 1993 to 2002. Obritsch MD, Fish DN, MacLaren R, Jung R. Antimicrob Agents Chemother. 2004 Dec; 48(12):4606-10.[↩]
- Nathwani D, Raman G, Sulham K, Gavaghan M, Menon V. Clinical and economic consequences of hospital-acquired resistant and multidrug-resistant Pseudomonas aeruginosa infections: a systematic review and meta-analysis. Antimicrob Resist Infect Control. 2014;3(1):32. doi: 10.1186/2047-2994-3-32[↩]
- Miyoshi-Akiyama T, Tada T, Ohmagari N, et al. Emergence and Spread of Epidemic Multidrug-Resistant Pseudomonas aeruginosa. Genome Biology and Evolution. 2017;9(12):3238-3245. doi:10.1093/gbe/evx243. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5726472/[↩]
- Residential Pool or Hot Tub Owners. Disinfection & Testing. https://www.cdc.gov/healthywater/swimming/residential/disinfection-testing.html[↩]
- Magill SS, Edwards JR, Bamberg W, Beldavs ZG, Dumyati G, Kainer MA, Lynfield R, Maloney M, McAllister-Hollod L, Nadle J, Ray SM, Thompson DL, Wilson LE, Fridkin SK. Emerging infections program healthcare-associated, T. antimicrobial use prevalence survey. Multistate point-prevalence survey of health care-associated infections. N Engl J Med. 2014;370(13):1198–208. http://dx.doi.org/10.1056/NEJMoa1306801[↩]
- Thaden JT, Park LP, Maskarinec SA, Ruffin F, Fowler VG, Jr, van Duin D. Results from a 13-year prospective cohort study show increased mortality associated with bloodstream infections caused by Pseudomonas aeruginosa compared to other bacteria. Antimicrob Agents Chemother. 2017;61(6) pii:e02671-16. http://dx.doi.org/10.1128/AAC.02671-16[↩][↩][↩]
- Pena C, Suarez C, Ocampo-Sosa A, Murillas J, Almirante B, Pomar V, Aguilar M, Granados A, Calbo E, Rodriguez-Bano J, Rodriguez F, Tubau F, Oliver A, Martinez-Martinez L D. Spanish Network for Research in Infectious. Effect of adequate single-drug vs combination antimicrobial therapy on mortality in Pseudomonas aeruginosa bloodstream infections: a post Hoc analysis of a prospective cohort. Clin Infect Dis. 2013;57(2):208–16. http://dx.doi.org/10.1093/cid/cit223[↩]
- Bassetti M, Vena A, Croxatto A, Righi E, Guery B. How to manage Pseudomonas aeruginosa infections. Drugs in Context. 2018;7:212527. doi:10.7573/dic.212527. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5978525/[↩][↩][↩][↩]
- Cilloniz C, Polverino E, Ewig S, Aliberti S, Gabarrus A, Menendez R, Mensa J, Blasi F, Torres A. Impact of age and comorbidity on cause and outcome in community-acquired pneumonia. Chest. 2013;144(3):999–1007. http://dx.doi.org/10.1378/chest.13-0062[↩][↩]
- Cilloniz C, Gabarrus A, Ferrer M, Puig de la Bellacasa J, Rinaudo M, Mensa J, Niederman MS, Torres A. Community-acquired pneumonia due to multidrug- and non-multidrug-resistant Pseudomonas aeruginosa. Chest. 2016;150(2):415–25. http://dx.doi.org/10.1016/j.chest.2016.03.042[↩][↩][↩]
- Mandell LA, Wunderink RG, Anzueto A, Bartlett JG, Campbell GD, Dean NC, Dowell SF, File TM, Jr, Musher DM, Niederman MS, Torres A, Whitney CG A. Infectious Diseases Society of, S. American Thoracic. American Thoracic, S. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44(Suppl 2):S27–72. http://dx.doi.org/10.1086/511159[↩][↩]
- Fernandez-Barat L, Ferrer M, De Rosa F, Gabarrus A, Esperatti M, Terraneo S, Rinaudo M, Li Bassi G, Torres A. Intensive care unit-acquired pneumonia due to Pseudomonas aeruginosa with and without multidrug resistance. J Infect. 2017;74(2):142–52. http://dx.doi.org/10.1016/j.jinf.2016.11.008[↩][↩][↩][↩][↩]
- Venier AG, Gruson D, Lavigne T, Jarno P, L’Heriteau F, Coignard B, Savey A, Rogues AM R.-R. group. Identifying new risk factors for Pseudomonas aeruginosa pneumonia in intensive care units: experience of the French national surveillance, REA-RAISIN. J Hosp Infect. 2011;79(1):44–8. http://dx.doi.org/10.1016/j.jhin.2011.05.007[↩][↩][↩]
- Micek ST, Wunderink RG, Kollef MH, Chen C, Rello J, Chastre J, Antonelli M, Welte T, Clair B, Ostermann H, Calbo E, Torres A, Menichetti F, Schramm GE, Menon V. An international multicenter retrospective study of Pseudomonas aeruginosa nosocomial pneumonia: impact of multidrug resistance. Crit Care. 2015;19:219. http://dx.doi.org/10.1186/s13054-015-0926-5[↩]
- Kalil AC, Metersky ML, Klompas M, Muscedere J, Sweeney DA, Palmer LB, Napolitano LM, O’Grady NP, Bartlett JG, Carratala J, El Solh AA, Ewig S, Fey PD, File TM, Jr, Restrepo MI, Roberts JA, Waterer GW, Cruse P, Knight SL, Brozek JL. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63(5):e61–e111. http://dx.doi.org/10.1093/cid/ciw353[↩]
- Carcas AJ, Garcia-Satue JL, Zapater P, Frias-Iniesta J. Tobramycin penetration into epithelial lining fluid of patients with pneumonia. Clin Pharmacol Ther. 1999;65(3):245–50. http://dx.doi.org/10.1016/S0009-9236(99)70103-7[↩]
- Slekovec C, Robert J, Trystram D, Delarbre JM, Merens A, van der Mee-Marquet N, de Gialluly C, Costa Y, Caillon J, Hocquet D, Bertrand X ONERBA. Pseudomonas aeruginosa in French hospitals between 2001 and 2011: back to susceptibility. Eur J Clin Microbiol Infect Dis. 2014;33(10):1713–7. http://dx.doi.org/10.1007/s10096-014-2125-8[↩]
- Lamas Ferreiro JL, Alvarez Otero J, Gonzalez Gonzalez L, Novoa Lamazares L, Arca Blanco A, Bermudez Sanjurjo JR, Rodriguez Conde I, Fernandez Soneira M, de la Fuente Aguado J. Pseudomonas aeruginosa urinary tract infections in hospitalized patients: mortality and prognostic factors. PLoS One. 2017;12(5):e0178178. http://dx.doi.org/10.1371/journal.pone.0178178[↩][↩][↩]
- Sarkar S, Patra AK, Mondal M. Ecthyma gangrenosum in the periorbital region in a previously healthy immunocompetent woman without bacteremia. Indian Dermatol Online J. 2016;7(1):36–9. http://dx.doi.org/10.4103/2229-5178.174326[↩]
- Stevens DL, Bisno AL, Chambers HF, Dellinger EP, Goldstein EJ, Gorbach SL, Hirschmann JV, Kaplan SL, Montoya JG, Wade JC Infectious Diseases Society of America. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis. 2014;59(2):e10–52. http://dx.doi.org/10.1093/cid/ciu444[↩]
- Beasley K, Panach K, Dominguez AR. Disseminated Candida tropicalis presenting with Ecthyma-Gangrenosum-like Lesions. Dermatol Online J. 2016;22(1) pii:13030/qt7vg4n68j[↩]
- Khalil BA, Baillie CT, Kenny SE, Lamont GL, Turnock RR, Pizer BL, van Saene HF, Losty PD. Surgical strategies in the management of ecthyma gangrenosum in paediatric oncology patients. Pediatr Surg Int. 2008;24(7):793–7. http://dx.doi.org/10.1007/s00383-008-2159-z[↩]
- Azzopardi EA, Azzopardi E, Camilleri L, Villapalos J, Boyce DE, Dziewulski P, Dickson WA, Whitaker IS. Gram negative wound infection in hospitalised adult burn patients – systematic review and metanalysis. PLoS One. 2014;9(4):e95042. http://dx.doi.org/10.1371/journal.pone.0095042[↩]
- Williams FN, Herndon DN, Hawkins HK, Lee JO, Cox RA, Kulp GA, Finnerty CC, Chinkes DL, Jeschke MG. The leading causes of death after burn injury in a single pediatric burn center. Crit Care. 2009;13(6):R183. http://dx.doi.org/10.1186/cc8170[↩]
- Estahbanati HK, Kashani PP, Ghanaatpisheh F. Frequency of Pseudomonas aeruginosa serotypes in burn wound infections and their resistance to antibiotics. Burns. 2002;28(4):340–8. http://dx.doi.org/10.1016/S0305-4179(02)00024-4[↩]
- Gang RK, Bang RL, Sanyal SC, Mokaddas E, Lari AR. Pseudomonas aeruginosa septicaemia in burns. Burns. 1999;25(7):611–6. http://dx.doi.org/10.1016/S0305-4179(99)00042-X[↩]
- Norbury W, Herndon DN, Tanksley J, Jeschke MG, Finnerty CC. Infection in burns. Surg Infect (Larchmt) 2016;17(2):250–5. http://dx.doi.org/10.1089/sur.2013.134[↩][↩]
- Pena C, Suarez C, Tubau F, Dominguez A, Sora M, Pujol M, Gudiol F, Ariza J. Carbapenem-resistant Pseudomonas aeruginosa: factors influencing multidrug-resistant acquisition in non-critically ill patients. Eur J Clin Microbiol Infect Dis. 2009;28(5):519–22. http://dx.doi.org/10.1007/s10096-008-0645-9[↩]
- Garnacho-Montero J, Sa-Borges M, Sole-Violan J, Barcenilla F, Escoresca-Ortega A, Ochoa M, Cayuela A, Rello J. Optimal management therapy for Pseudomonas aeruginosa ventilator-associated pneumonia: an observational, multicenter study comparing monotherapy with combination antibiotic therapy. Crit Care Med. 2007;35(8):1888–95. http://dx.doi.org/10.1097/01.CCM.0000275389.31974.22[↩]
- Wright H, Bonomo RA, Paterson DL. New agents for the treatment of infections with Gram-negative bacteria: restoring the miracle or false dawn? Clin Microbiol Infect. 2017;23(10):704–12. http://dx.doi.org/10.1016/j.cmi.2017.09.001[↩][↩]
- Zhanel GG, Chung P, Adam H, Zelenitsky S, Denisuik A, Schweizer F, Lagace-Wiens PR, Rubinstein E, Gin AS, Walkty A, Hoban DJ, Lynch JP, III, Karlowsky JA. Ceftolozane/tazobactam: a novel cephalosporin/beta-lactamase inhibitor combination with activity against multidrug-resistant gram-negative bacilli. Drugs. 2014;74(1):31–51. http://dx.doi.org/10.1007/s40265-013-0168-2[↩]
- Solomkin J, Hershberger E, Miller B, Popejoy M, Friedland I, Steenbergen J, Yoon M, Collins S, Yuan G, Barie PS, Eckmann C. Ceftolozane/tazobactam plus metronidazole for complicated intra-abdominal infections in an era of multidrug resistance: results from a randomized, double-blind, phase 3 trial (ASPECT-cIAI) Clin Infect Dis. 2015;60(10):1462–71. http://dx.doi.org/10.1093/cid/civ097[↩]
- Haidar G, Philips NJ, Shields RK, Snyder D, Cheng S, Potoski BA, Doi Y, Hao B, Press EG, Cooper VS, Clancy CJ, Nguyen MH. Ceftolozane-Tazobactam for the treatment of multidrug-resistant Pseudomonas aeruginosa infections: clinical effectiveness and evolution of resistance. Clin Infect Dis. 2017;65(1):110–20. http://dx.doi.org/10.1093/cid/cix182[↩]
- Sader HS, Castanheira M, Farrell DJ, Flamm RK, Jones RN. Ceftazidime-avibactam activity when tested against ceftazidime-nonsusceptible Citrobacter spp., Enterobacter spp., Serratia marcescens, and Pseudomonas aeruginosa from Unites States medical centers (2011–2014) Diagn Microbiol Infect Dis. 2015;83(49):389–94. http://dx.doi.org/10.1016/j.diagmicrobio.2015.06.008[↩]
- Torres A, Zhong N, Pachl J, Timsit JF, Kollef M, Chen Z, Song J, Taylor D, Laud PJ, Stone GG, Chow JW. Ceftazidime-avibactam versus meropenem in nosocomial pneumonia, including ventilator-associated pneumonia (REPROVE): a randomised, double-blind, phase 3 non-inferiority trial. Lancet Infect Dis. 2018;18(3):285–95. http://dx.doi.org/10.1016/S1473-3099(17)30747-8[↩]
- Blizzard TA, Chen H, Kim S, Wu J, Bodner R, Gude C, Imbriglio J, Young K, Park YW, Ogawa A, Raghoobar S, Hairston N, Painter RE, Wisniewski D, Scapin G, Fitzgerald P, Sharma N, Lu J, Ha S, Hermes J, Hammond ML. Discovery of MK-7655, a beta-lactamase inhibitor for combination with Primaxin(R) Bioorg Med Chem Lett. 2014;24(3):780–5. http://dx.doi.org/10.1016/j.bmcl.2013.12.101[↩]
- Thaden JT, Pogue JM, Kaye KS. Role of newer and re-emerging older agents in the treatment of infections caused by carbapenem-resistant Enterobacteriaceae. Virulence. 2017;8(4):403–16. http://dx.doi.org/10.1080/21505594.2016.1207834[↩]
- Lucasti C, Vasile L, Sandesc D, Venskutonis D, McLeroth P, Lala M, Rizk ML, Brown ML, Losada MC, Pedley A, Kartsonis NA, Paschke A. Phase 2, Dose-Ranging study of relebactam with imipenem-cilastatin in subjects with complicated intra-abdominal infection. Antimicrob Agents Chemother. 2016;60(10):6234–43. http://dx.doi.org/10.1128/AAC.00633-16[↩]
- Sims M, Mariyanovski V, McLeroth P, Akers W, Lee YC, Brown ML, Du J, Pedley A, Kartsonis NA, Paschke A. Prospective, randomized, double-blind, Phase 2 dose-ranging study comparing efficacy and safety of imipenem/cilastatin plus relebactam with imipenem/cilastatin alone in patients with complicated urinary tract infections. J Antimicrob Chemother. 2017;72(9):2616–26. http://dx.doi.org/10.1093/jac/dkx139[↩]