Contents
What is insulinoma
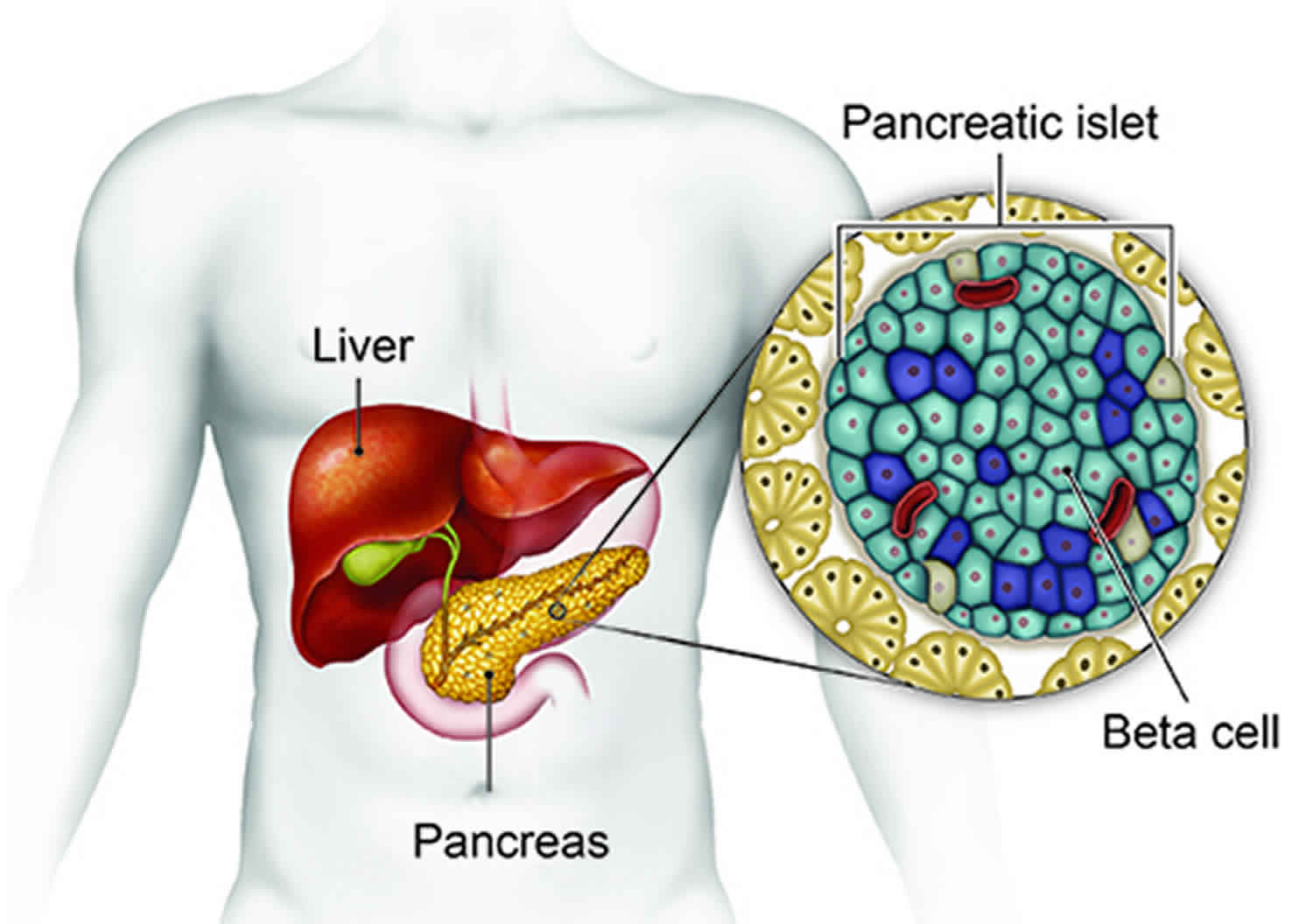
Insulinoma is a very rare pancreatic beta cells tumor that produce too much insulin. Some insulinomas also secrete other hormones, such as gastrin, 5-hydroxyindolic acid, adrenocorticotropic hormone (ACTH), glucagon, human chorionic gonadotropin, and somatostatin 1. Insulinoma has an incidence of 0.4 per 100,000 person years or 4 cases per million per year. Insulin is a hormone that reduces the level of sugar in your blood by helping it move into cells. As a result, people with insulinomas generally have very low blood sugar levels which can be associated with anxiety, confusion, hunger, a fast heart rate, and sweating. In severe cases, very low blood sugar level can lead to seizures, coma or even death 1. Approximately 90 percent of insulinomas are benign (noncancerous) and solitary 1. In most cases, the underlying cause of insulinoma is unknown. However, people with specific genetic syndromes such as multiple endocrine neoplasia type 1 (MEN-1), Von Hippel-Lindau syndrome, Neurofibromatosis type 1, and tuberous sclerosis are at higher risk of insulinomas and other endocrine tumors 2. Multiple insulinoma tumors occur in multiple endocrine neoplasia type 1 (MEN1). Cancer is more frequent in multiple endocrine neoplasia type 1 (MEN-1) than sporadic insulinomas. The diagnosis of insulinoma requires demonstration of inappropriately high insulin for the prevailing blood glucose in a 72 hour fast. A high proinsulin: insulin ratio indicates cancer.
Approximately 10% of insulinomas are malignant (metastases are present). Approximately 10% of patients have multiple insulinomas; of patients with multiple insulinomas, 50% have multiple endocrine neoplasia type 1 (MEN 1). Insulinomas are associated with multiple endocrine neoplasia type 1 (MEN-1) in 5% of patients. On the other hand, 21% of patients with multiple endocrine neoplasia type 1 (MEN-1) develop insulinomas. Because of the association of insulinomas with MEN 1, consideration should be given to screening family members of insulinoma patients for multiple endocrine neoplasia type 1 (MEN-1).
The pancreas is an organ in your abdomen. The pancreas makes several enzymes and hormones, including the hormone insulin. Insulin’s job is to reduce the level of sugar (glucose) in the blood by helping sugar move into cells.
Most of the time when your blood sugar level decreases, the pancreas stops making insulin to make sure that your blood sugar stays in the normal range. Tumors of the pancreas that produce too much insulin are called insulinomas. Insulinomas keep making insulin, and can make your blood sugar drops too low (hypoglycemia).
A high blood insulin level causes a low blood sugar level (hypoglycemia). Hypoglycemia may be mild, leading to symptoms such as anxiety and hunger. Or it can be severe, leading to seizures, coma, and even death.
Localization of the tumor by transabdominal ultrasound and computed tomography is the preferred initial option followed by endoscopic ultrasonography or arterial stimulation with hepatic venous sampling. For single solitary tumors, surgery to remove the tumor is the treatment of choice 1. With multiple tumors experts recommended that venous sampling regionalizes the origin of the insulin and directs the region for resection. With suggestion of malignancy e.g. elevated pancreastatin, NKA, and proinsulin/insulin ratio of >0.8, experts recommend debulking of the pancreatic tumors including surrounding lymph nodes 3. If hyperinsulinemia and hypoglycemia persist, diazoxide with a thiazide diuretic relieves hypoglycemia. Liver metastases can be resected, or treated by bland or chemo-embolization, radiofrequency and cryoablation depending on availability at the institution. In patients with unresectable metastatic tumors we prefer Lanreotide which is approved for control of tumor growth and if necessary peptide receptor radiotherapy (PRRT) with an option to utilize temodar and xeloda. For intractable hypoglycemia with gastric bypass programmed pumps containing glucagon have shown promise.
Insulinoma symptoms
Insulinomas are characterized clinically by the Whipple triad, as follows:
- Presence of symptoms of hypoglycemia (about 85% of patients)
- Documented low blood sugar at the time of symptoms
- Reversal of symptoms by glucose administration
Insulinoma symptoms are most common when you skip or delay a meal. Symptoms may include:
- Anxiety, behavior changes, or confusion
- Clouded vision
- Loss of consciousness or coma
- Convulsions or tremor
- Dizziness or headache
- Hunger or weight gain
- Fast heart rate or palpitations
- Sweating
About 85% of patients with insulinoma present with one of the following symptoms of hypoglycemia:
- Diplopia
- Blurred vision
- Palpitations
- Weakness
Hypoglycemia can also result in the following:
- Confusion
- Abnormal behavior
- Unconsciousness
- Amnesia
- Adrenergic symptoms (from hypoglycemia-related adrenalin release): Weakness, sweating, tachycardia, palpitations, and hunger
- Seizures
Insulinoma possible complications
Insulinoma complications may include:
- Severe hypoglycemic reaction
- Spread of a cancerous tumor (metastasis)
- Diabetes if the entire pancreas is removed (rare), or food not being absorbed if too much of the pancreas is removed
- Inflammation and swelling of the pancreas
Insulinoma diagnosis
Insulinoma tests
After fasting, your blood may be tested for:
- Blood C-peptide level
- Blood glucose level
- Blood insulin level
- Drugs that cause the pancreas to release insulin
- The response of your body to a shot of glucagon
Lab studies
Failure of endogenous insulin secretion to be suppressed by hypoglycemia is the hallmark of an insulinoma. Thus, the finding of inappropriately elevated levels of insulin in the face of hypoglycemia is the key to diagnosis.
The biochemical diagnosis of insulinoma is established in 95% of patients during prolonged fasting (up to 72 hour) 4 when the following results are found:
- Serum insulin levels of 10 µU/mL or more (normal < 6 µU/mL)
- Glucose levels of less than 40 mg/dL
- C-peptide levels exceeding 2.5 ng/mL (normal < 2 ng/mL)
- Proinsulin levels greater than 25% (or up to 90%) of immunoreactive insulin levels
- Screening for sulfonylurea negative
The rate of decline in glucose has not warranted the use of a glucometer. More than 95% of cases can be diagnosed based on responses to a 72-hour fast. Serial glucose and insulin levels are obtained over the 72 hours until the patient becomes symptomatic. Because the absolute insulin level is not elevated in all patients with insulinomas, a normal level does not rule out the disease. However, a fasting insulin level of greater than 24 μU/mL is found in approximately 50% of patients with insulinoma 3. Values of insulin greater than 7 μU/mL after a more prolonged fast in the presence of a blood glucose less than 40 mg/dL also are highly suggestive 3. A refinement in the interpretation of glucose and insulin levels has been established by determining the ratio of insulin levels (mU/mL) to the concomitant glucose level (mg/dL). An insulin/glucose ratio of greater than 0.3 has been found in virtually all patients proven to have an insulinoma or other islet cell disease causing organic hyperinsulinism. The accuracy of the test can be increased by calculating the amended insulin/glucose ratio as follows:
- Amended ratio = [insulin (mU/mL) x 100]/[glucose (mg/dL) – 30] Normal <50.
If the value is greater than 50, then organic hyperinsulinism is certain 5.
Imaging studies
CT, MRI, or PET scan of the abdomen may be done to look for a tumor in the pancreas. If a tumor is not seen in the scans, one of the following tests may be performed:
- Endoscopic ultrasound (EUS) (test that uses a flexible scope and sound waves to view digestive organs). Detects 77% of insulinomas in the pancreas 6
- Intraoperative transabdominal high-resolution ultrasonography with the transducer wrapped in a sterile rubber glove and passed over the exposed pancreatic surface: Detects more than 90% of insulinomas
- Computed tomography (CT) scanning: 82-94% sensitivity
- Magnetic resonance imaging (MRI) 7
- Pancreatic arteriography (test that uses special dye to view the arteries in the pancreas): Selective arteriography has 82% accuracy, with a 5% false-positive rate; arteriography with catheterization of small arterial branches of the celiac system combined with calcium injections and simultaneous measurements of hepatic vein insulin during each selective calcium injection localizes tumors in 47% of patients
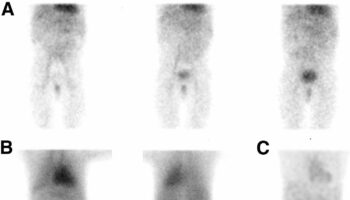
- PET/CT with gallium-68 DOTA-(Tyr3)-octreotate (Ga-DOTATATE): 90% sensitivoity; possible adjunct study when other imaging studies are negative and minimally invasive surgery is planned 8
- Octreotide scan (special test that checks for cancer cells in the body)
- Pancreatic venous sampling for insulin (test that helps locate the exact location of the tumor that is making too much insulin)
Localization of insulinoma
Once the diagnosis of insulinoma is confirmed, every effort should be made to localize the insulinoma tumor. Preoperative localization is important because approximately 30% of insulinomas are less than 1 cm in diameter and 10% are multiple. In addition, 10 to 15% are malignant and 10% will have either islet cell hyperplasia or nesidioblastosis and no tumor at all 9. Techniques most commonly used to demonstrate tumors in the upper abdomen include contrast enhanced CT and MRI scans of the abdomen, and endoscopic ultrasound. Each modality has variable reported abilities to identify insulinomas, likely reflecting institutional expertise. Reported sensitivities of identifying insulinomas are 69% -94.4% for CT, 63% for MRI and 89%-93.5% for endoscopic ultrasound 10. Endoscopic ultrasound is better able to detect lesions in the head of the pancreas (sensitivity 92.6%) as compared to the tail (40%).
Until the past decade, the only study considered to be of proven value in the localization of insulinomas was selective pancreatic angiography 4. During this procedure, highly selective injections of contrast, subtraction procedures, and magnification views increase the number of insulinomas identified. In one large series, 90% of insulinomas were reported to be localized by angiography alone 4; however, most groups report less satisfactory results 11. A summary of all reports in the literature found that approximately 60% of insulinomas have been detected by this method 12. Selective intra-arterial injection of calcium with sampling of hepatic vein insulin appears to improve the ability to detect insulinomas, 13 similar to the results seen with intra-arterial secretin in gastrinoma.
Percutaneous trans-hepatic venous sampling of insulin from pancreatic veins has been used successfully in localizing occult sources of hyperinsulinism 14. Experts now believe that the combination of a secretin test to determine the nature of the hyperinsulinism (e.g., distinction of hyperplasia from adenoma or multiple adenomatosis) with percutaneous trans-hepatic venous sampling to localize the source provides the best means of establishing the specific cause of organic hyperinsulinism with near certainty. A skilled angiographer and careful analysis of the hormonal data in relationship to the venous anatomy in the individual case are required.
A variety of other imaging methods have been investigated. A recent study suggests that the glucagon-like peptide 1 (GLP-1) receptor scan is useful to localize occult insulinomas that cannot be identified by other means 15. A prospective multicenter study from Europe showed that GLP-1 receptor imaging of patients with insulinoma had a positive predictive value of 83% and a higher sensitivity than CT or MRI 16. Multi-modality imaging with specifically designed protocols appears to increase the yield of conventional imaging studies 17. Recently, use of [18F] fluorodopa positron emission tomography has been shown useful for diagnosing and localizing congenital hyperinsulinism in infants 18 and insulinoma or beta cell hyperplasia in adults patients 19. However further study in a larger number of patients as well as comparisons to other non-invasive and invasive imaging studies will be required to more precisely define the utility of these localization methods in patients with hyperinsulinism.
If percutaneous trans-hepatic venous sampling is not available and preoperative localization by angiography or other techniques has been negative, the surgeon may use intraoperative ultrasound if a careful exploration fails to detect a tumor. Some who have used this technique routinely have reported excellent results. Ultrasound does not identify hyperplasia or nesidioblastosis, however, and its sensitivity appears to be operator dependent.
The role of extensive pre-operative localization tests has been questioned. One study showed that intraoperative inspection and palpation localized lesions in 91% and intraoperative ultrasound in 93% of cases. Interestingly, all 5 occult tumors were palpable at surgery. These investigators suggest that percutaneous trans-hepatic venous sampling is helpful in localizing lesions before re-operation, but question the need for extensive pre-operative localization prior to initial exploration in patients believed to have insulinoma 20. On the other hand, another experienced group has shown that since 1994, pre-operative imaging including the use of endoscopic ultrasound was able to identify the tumors 98% of the time. They also found that palpation and intraoperative ultrasound detected 92% of tumors 21.
Insulinoma treatment
Surgery
Because insulinoma resection achieves cure in 90% of patients, surgery is currently the therapy of choice. Enucleation is the preferred technique.
If there is a single tumor, it will be removed. If there are many tumors, part of the pancreas will need to be removed. At least 15% of the pancreas must be left to produce normal levels of enzymes for digestion.
In rare cases, the entire pancreas is removed if there are many insulinomas or they’ll continue to come back. Removing the entire pancreas leads to diabetes because there is no longer any insulin being produced. Insulin shots (injections) are then required.
If no tumor is found during surgery, or if you can’t have surgery, you may get the medicine diazoxide to lower insulin production and prevent hypoglycemia. A water pill (diuretic) is given with this medicine to prevent the body from retaining fluid. Octreotide is another medicine that is used to reduce insulin release in some people.
Pharmacologic therapy
Patients with diffuse hyperinsulinism for whom an operation is planned first should have a trial of treatment with diazoxide and a natriuretic benzothiadiazine. Medical treatment is required for the great majority of malignant insulinomas because only occasionally are they cured by operation. Medical treatment for benign insulinomas includes a change in meals to include unrefined carbohydrate given as frequently as required to prevent hypoglycemia. Antihormonal therapy may be useful if diet is insufficient. The management of malignant insulinoma is antihormonal and antitumor therapy.
Pharmacologic treatment is designed to prevent hypoglycemia and, in patients with malignant tumors, to reduce the tumor burden. Agents used in this therapy include the following:
- Diazoxide: Reduces insulin secretion
- Hydrochlorothiazide: Counteracts edema and hyperkalemia secondary to diazoxide and potentiates its hyperglycemic effect
- Somatostatin analogs (octreotide, lanreotide): Prevent hypoglycemia
- Everolimus: For patients with metastatic insulinoma and refractory hypoglycemia
Diazoxide is related to the thiazide diuretics and reduces insulin secretion. Adverse effects include sodium retention, a tendency to congestive cardiac failure, and hirsutism. Prescribe hydrochlorothiazide to counteract the edema and hyperkalemia secondary to diazoxide and to potentiate its hyperglycemic effect.
Of patients with insulinoma, 50% may benefit from the somatostatin analog octreotide to prevent hypoglycemia 22. The effect of the therapy depends on the presence of somatostatin receptor subtype 2 on insulinoma tumor cells. Use of the somatostatin analogs lanreotide and pasireotide have also been reported 23.
As studies have shown, an OctreoScan is not a prerequisite before starting octreotide treatment. In patients with insulinoma and a negative scan finding, somatostatin decreased insulin levels significantly and lowered the incidence of hypoglycemic events 24.
Indications for chemotherapy include progressive disease with an increase of greater than 25% of the main tumor masses in a follow-up period of 12 months, or tumor symptoms not treatable with other methods. Combination regimens have achieved better results than single agents.
The current medical treatment choices are those used for any metastatic neuroendocrine gastroenteric pancreatic tumor. Options are as follows:
- Combination therapy with streptozotocin plus doxorubicin or 5-fluorouracil: Streptozotocin alone may achieve a partial response in 50% of patients and complete response in 20%. The median survival in one study was 16 months. With streptozotocin plus 5-fluorouracil, 33% of patients show complete response, with the median survival increasing to 26 months.
- Dacarbazine- and temozolomide-based regimens: There appears to be a correlation between expression of methylguanine DNA methyltransferase (MGMT) and temozolomide responsiveness in advanced neuroendocrine tumors. Methylguanine DNA methyltransferase is an enzyme that is responsible for DNA repair induced by alkylating agent chemotherapy.
- Molecularly targeted therapy: This includes vascular endothelial growth factor receptor (VEGFR) inhibitors such as sunitinib, sorafenib, pazopanib, or cabozantinib.
- Mammalian target of rapamycin (mTOr) inhibitors: Everolimus, with or without octreotide
- Peptide receptor radioligand therapy: In January 2018, the US Food & Drug Administration approved lutetium [177Lu] oxodotreotide (Lutathera) for the treatment of patients with somatostatin receptor–positive gastroenteropancreatic neuroendocrine tumors. Approval was based on results of the phase III NETTER-1 trial 25
There is a single case report of successful control of intractable hypoglycemia in an elderly man with metastatic insulinoma through the use of oral rapamycin (sirolimus), 2 mg/day 26.
The mTOR inhibitor everolimus is approved for treatment of locally advanced or metastatic neuroendocrine tumors of pancreatic origin. In a French study 27, everolimus therapy normalized blood glucose levels in 11 of 12 patients with metastatic insulinoma and refractory hypoglycemia, with the therapeutic effect maintained for a median duration of 6.5 months (range 1-35+ months). However, three patients discontinued everolimus because of cardiac and/or pulmonary adverse events, which led to two deaths 27.
Ablation
Ablation is a nonsurgical option that has had some long-term success.CT-guided radiofrequency ablation has been used successfully to treat insulinoma in an elderly patient whose hypoglycemia that was refractory to diazoxide, and who was not a candidate for surgery because of comorbidities and poor physical condition 28. In addition to radiofrequency ablation, successful results have been attained with embolization and with ethanol, among other techniques 29.
Diet
The cornerstone of medical management of insulinoma and other forms of hyperinsulinism is the diet.
Considerations include the following:
- Because insulin secretion by most insulinomas is not responsive to glucose levels, carbohydrate feedings every 2-3 hours can help maintain euglycemia, although obesity may develop.
- Glucagon should be available for emergency use.
- Exercise may aggravate hypoglycemia in patients with insulinoma.
Not uncommonly, patients may avoid symptoms of hypoglycemia for variable periods of time by shortening the number of hours between feedings. For some, the inclusion of a bedtime (11:00 pm) feeding is sufficient; for others, a midmorning, midafternoon, and/or a 3:00 snack are necessary. Although the tumor may be stimulated occasionally to secrete insulin by the ingestion of carbohydrates, it is inadvisable to restrict the intake of carbohydrate. More slowly absorbable forms of carbohydrates (e.g. starches, bread, potatoes, rice) generally are preferred. During hypoglycemic episodes, rapidly absorbable forms (e.g. fruit juices with added glucose or sucrose) are indicated. In patients with severe refractory hypoglycemia, use of a continuous intravenous infusion of glucose, coupled with increased dietary intake of carbohydrate, frequently alleviates hypoglycemia long enough to institute additional therapy.
Insulinoma prognosis
Approximately 90-95% of insulinomas are non-cancerous (benign). Long-term cure with complete resolution of preoperative symptoms is expected after complete resection. But a severe hypoglycemic reaction or the spread of a cancerous tumor to other organs can be life-threatening.
Recurrence of benign insulinomas was observed in 5.4% of patients in a series of 120 patients over a period of 4-17 years. The same diagnostic and therapeutic approach was recommended, including surgical exploration and tumor resection.
Patients may develop nonfunctioning metastatic disease to the liver up to 14 years after insulinoma resection 30. Note that some insulinomas are indolent (depending on the tumor biology), resulting in prolonged survival.
- Insulinoma. https://emedicine.medscape.com/article/283039-overview[↩][↩][↩][↩]
- Pancreatic Neuroendocrine Neoplasms (pNENs). https://rarediseases.org/rare-diseases/pancreatic-neuroendocrine-neoplasms-pnens/[↩]
- Vinik A, Feliberti E, Perry RR. Insulinomas. [Updated 2017 Jul 3]. In: Feingold KR, Anawalt B, Boyce A, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK278981[↩][↩][↩]
- Edis AJ, McIlrath DC, van Heerden JA et al. Insulinoma–current diagnosis and surgical management. Curr Probl Surg 1976; 13(10):1-45.[↩][↩][↩]
- Fajans SS, Vinik AI. Diagnosis and treatment of insulinoma. In: Fanten RJ, Manni A, editors. Diagnosis and Management of Endocrine Related Tumors. Boston: Martinus Nijhoff, 1984: 235.[↩]
- McLean A. Endoscopic ultrasound in the detection of pancreatic islet cell tumours. Cancer Imaging. 2004 Mar 29. 4(2):84-91.[↩]
- Zhu L, Xue H, Sun Z, Li P, Qian T, Xing X, et al. Prospective comparison of biphasic contrast-enhanced CT, volume perfusion CT, and 3 Tesla MRI with diffusion-weighted imaging for insulinoma detection. J Magn Reson Imaging. 2017 Apr 17[↩]
- Nockel P, Babic B, Millo C, Herscovitch P, Patel D, Nilubol N, et al. Localization of Insulinoma Using 68Ga-DOTATATE PET/CT Scan. J Clin Endocrinol Metab. 2017 Jan 1. 102 (1):195-199[↩]
- Vinik A. Endocrine tumors of the gastroenteropancreatic axis. Diagnosis and Management of Endocrine-Related Tumors. 1984.[↩]
- Joseph AJ, Kapoor N, Simon EG et al. Endoscopic ultrasonography–a sensitive tool in the preoperative localization of insulinoma. Endocr Pract 2013; 19(4):602-608.[↩]
- Robins JM, Bookstein JJ, Oberman HA, Fajans SS. Selective angiography in localizing islet-cell tumors of the pancreas. A further appraisal. Radiology 1973; 106(3):525-528.[↩]
- Vinik A, Strodel W, Cho K, Eckhauser F, Thompson N. Localization of hormonally active gastrointestinal tumors. Endocrine Surgery Update 1983.[↩]
- Gibril F, Reynolds JC, Doppman JL et al. Somatostatin receptor scintigraphy: its sensitivity compared with that of other imaging methods in detecting primary and metastatic gastrinomas. A prospective study. Ann Intern Med 1996; 125(1):26-34.[↩]
- Vinik AI, Thompson N. Controversies in the management of Zollinger-Ellison syndrome. Ann Intern Med 1986; 105:956-959.[↩]
- Wild D, Macke H, Christ E, Gloor B, Reubi JC. Glucagon-like peptide 1-receptor scans to localize occult insulinomas. N Engl J Med 2008; 359(7):766-768.[↩]
- Christ E, Wild D, Ederer S et al. Glucagon-like peptide-1 receptor imaging for the localisation of insulinomas: a prospective multicentre imaging study. Lancet Diabetes Endocrinol 2013; 1(2):115-122.[↩]
- McAuley G, Delaney H, Colville J et al. Multimodality preoperative imaging of pancreatic insulinomas. Clin Radiol 2005; 60(10):1039-1050.[↩]
- Hardy OT, Hernandez-Pampaloni M, Saffer JR et al. Accuracy of [18F]fluorodopa positron emission tomography for diagnosing and localizing focal congenital hyperinsulinism. J Clin Endocrinol Metab 2007; 92(12):4706-4711.[↩]
- Kauhanen S, Seppanen M, Minn H et al. Fluorine-18-L-dihydroxyphenylalanine (18F-DOPA) positron emission tomography as a tool to localize an insulinoma or beta-cell hyperplasia in adult patients. J Clin Endocrinol Metab 2007; 92(4):1237-1244.[↩]
- Ravi K, Britton BJ. Surgical approach to insulinomas: are pre-operative localisation tests necessary? Ann R Coll Surg Engl 2007; 89(3):212-217.[↩]
- Nikfarjam M, Warshaw AL, Axelrod L et al. Improved contemporary surgical management of insulinomas: a 25-year experience at the Massachusetts General Hospital. Ann Surg 2008; 247(1):165-172.[↩]
- Arnold R, Simon B, Wied M. Treatment of neuroendocrine GEP tumours with somatostatin analogues: a review. Digestion. 2000. 62 Suppl 1:84-91.[↩]
- Tirosh A, Stemmer SM, Solomonov E, Elnekave E, Saeger W, Ravkin Y, et al. Pasireotide for malignant insulinoma. Hormones (Athens). 2016 Apr. 15 (2):271-6.[↩]
- Keymeulen B, Bossuyt A, Peeters TL, Somers G. 111In-octreotide scintigraphy: a tool to select patients with endocrine pancreatic tumors for octreotide treatment?. Ann Nucl Med. 1995 Aug. 9(3):149-52.[↩]
- Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B, et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N Engl J Med. 2017 Jan 12. 376 (2):125-135.[↩]
- Bourcier ME, Sherrod A, DiGuardo M, Vinik AI. Successful control of intractable hypoglycemia using rapamycin in an 86-year-old man with a pancreatic insulin-secreting islet cell tumor and metastases. J Clin Endocrinol Metab. 2009 Sep. 94(9):3157-62.[↩]
- Bernard V, Lombard-Bohas C, Taquet MC, Caroli-Bosc FX, Ruszniewski P, Niccoli-Sire P, et al. Efficacy of Everolimus in Patients with Metastatic Insulinoma and Refractory Hypoglycemia. Eur J Endocrinol. 2013 Feb 7.[↩][↩]
- Limmer S, Huppert PE, Juette V, Lenhart A, Welte M, Wietholtz H. Radiofrequency ablation of solitary pancreatic insulinoma in a patient with episodes of severe hypoglycemia. Eur J Gastroenterol Hepatol. 2009 Sep. 21(9):1097-101[↩]
- Mele C, Brunani A, Damascelli B, Tichà V, Castello L, Aimaretti G, et al. Non-surgical ablative therapies for inoperable benign insulinoma. J Endocrinol Invest. 2017 Jul 28[↩]
- Gonzalez-Gonzalez A, Recio-Cordova JM. Liver metastases 9 years after removal of a malignant insulinoma which was initially considered benign. JOP. 2006. 7(2):226-9.[↩]