Contents
Bacillary peliosis
Bacillary peliosis also called bacillary peliosis hepatis or Bartonella peliosis hepatis is a blood vessel condition characterized by multiple, randomly distributed, blood-filled and cyst-like cavities throughout the liver that is secondary to Gram-negative Bartonella henselae bacteria infection in immunocompromised patients, first described in human immunodeficiency virus (HIV) infected or AIDS individuals 1, 2, 3, 4. Most patients with bacillary peliosis develop symptoms such as anorexia, weight loss, enlarged tender lymph nodes (lymphadenopathy), chills, fever, headache and malaise in addition to gastrointestinal symptoms or abdominal pain 2, 5, 6, 7, 8, 9. Approximately 1% to 2% of patients with Bartonella infection develop severe systemic disease with involvement of the liver, spleen, bone, central nervous system (brain and spinal cord) or lung 7. Approximately 25% of patients have lymphadenopathy (enlarged lymph nodes), but often the classic skin papule of cat-scratch disease is absent 7, 8, 9.
Bacillary peliosis hepatis patients have enlarged liver (hepatomegaly), and some also have enlarged spleen (splenomegaly). One-quarter of the patients also had skin bacillary angiomatosis (cutaneous bacillary angiomatosis) lesions. Abdominal CT of the peliotic liver usually reveals numerous hypodense lesions, but this appearance is not specific for bacillary peliosis; therfore the diagnosis of Bartonella infection must be confirmed by histopathological evaluation. Additionally, some HIV-infected patients with hepatic Bartonella infection develop inflammatory nodules in the liver without the vascular proliferative characteristics of peliosis hepatis. Patients with splenic bacillary peliosis can have thrombocytopenia or pancytopenia and abdominal ascites.
HIV-associated bacillary peliosis hepatis is an unusual, treatable opportunistic infection that responds to antibiotics. Inadequately treated, HIV-associated bacillary peliosis can be progressive and may be fatal 10, 1.
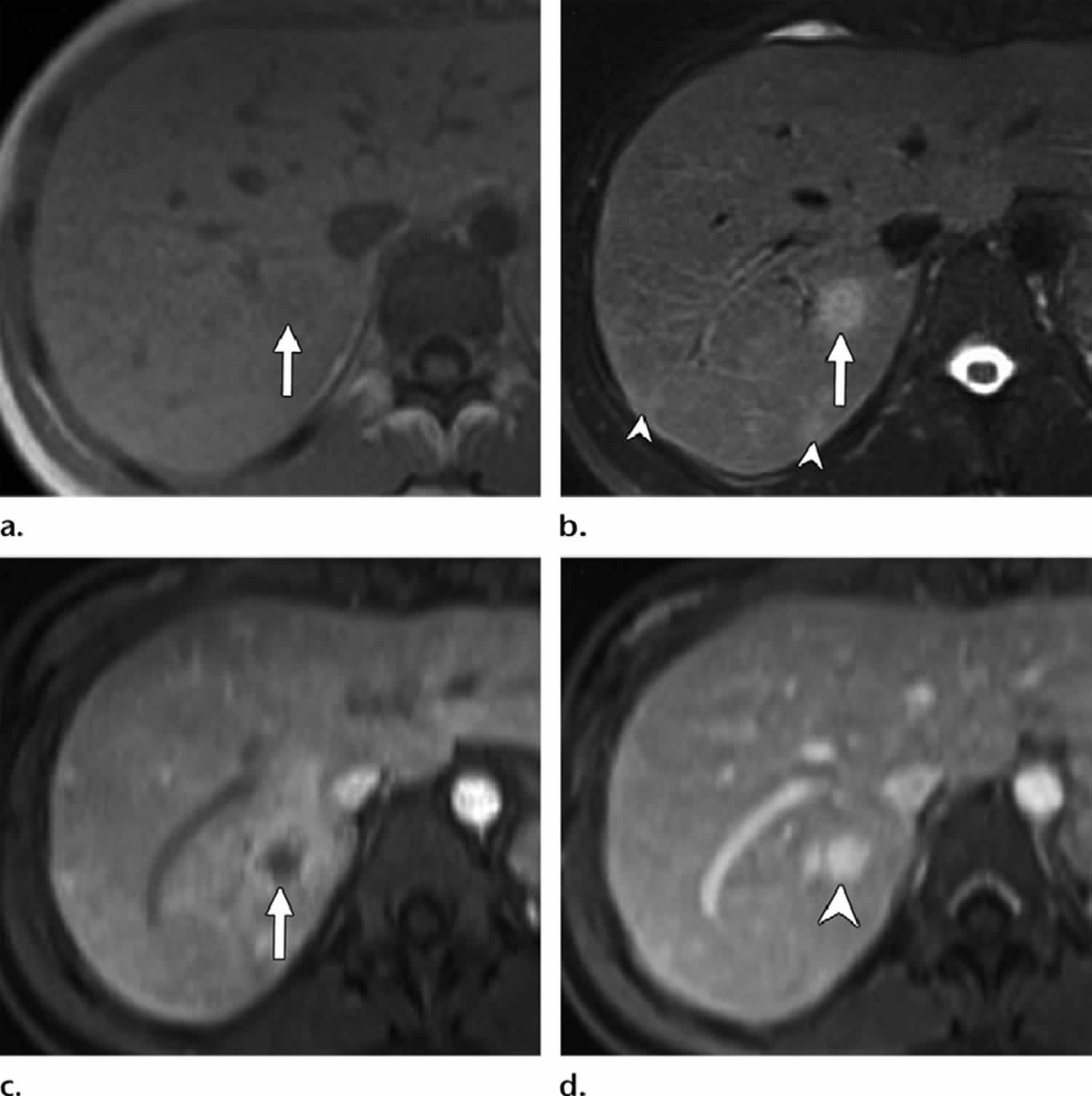
Figure 1. Bacillary peliosis
Footnotes: Bacillary peliosis in a 32-year-old man with HIV/AIDS who presented with fever. (a) Axial in-phase T1-weighted MR image shows an ill-defined iso-to mildly hypointense nodule in the posterior segment of the right hepatic lobe (arrow). (b) Axial fat-saturated T2-weighted MR image shows moderate to high signal intensity of the nodule (arrow). Smaller similar subcapsular nodules are seen posteriorly (arrowheads). (c) Axial contrast-enhanced early phase fatsuppressed T1-weighted MR image shows the hypointense right hepatic nodule with irregular continuous peripheral enhancement (arrow). (d) Axial contrast-enhanced delayed phase T1-weighted MR image shows delayed enhancement within the nodule (arrowhead), which reaches a signal intensity similar to that of the surrounding hepatic venous structures because of contrast agent retention within dilated sinusoids in the lesion.
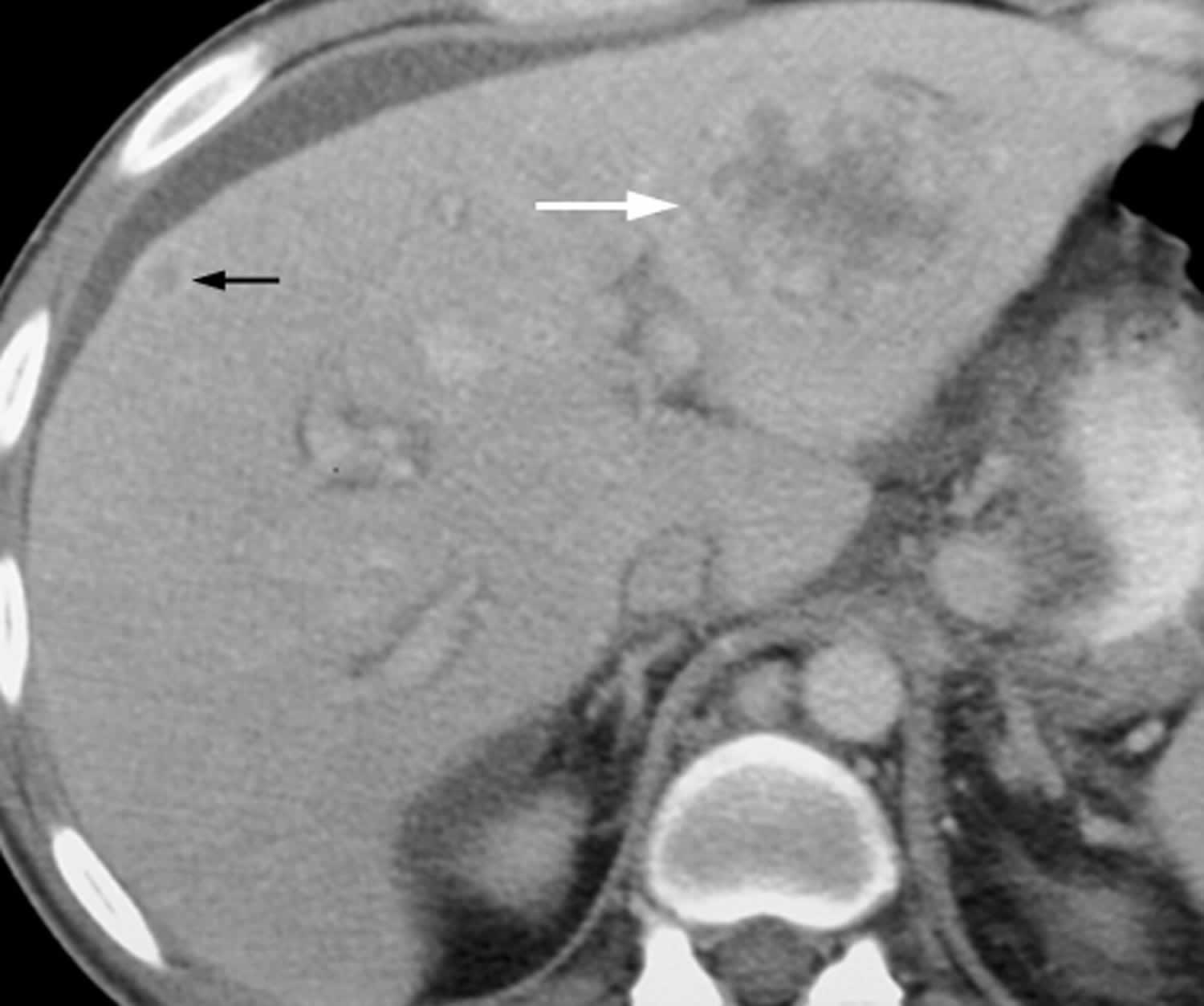
[Source 11 ]Figure 2. Bacillary peliosis
Footnotes: 44-year-old man with AIDS and bacillary peliosis. Transverse contrast-enhanced CT image shows large ill-defined, hypoattenuating lesion (white arrow) with heterogeneous peripheral enhancement within left liver lobe. Smaller subcapsular hypoattenuating lesion (black arrow) with ring enhancement can also be seen in right liver lobe.
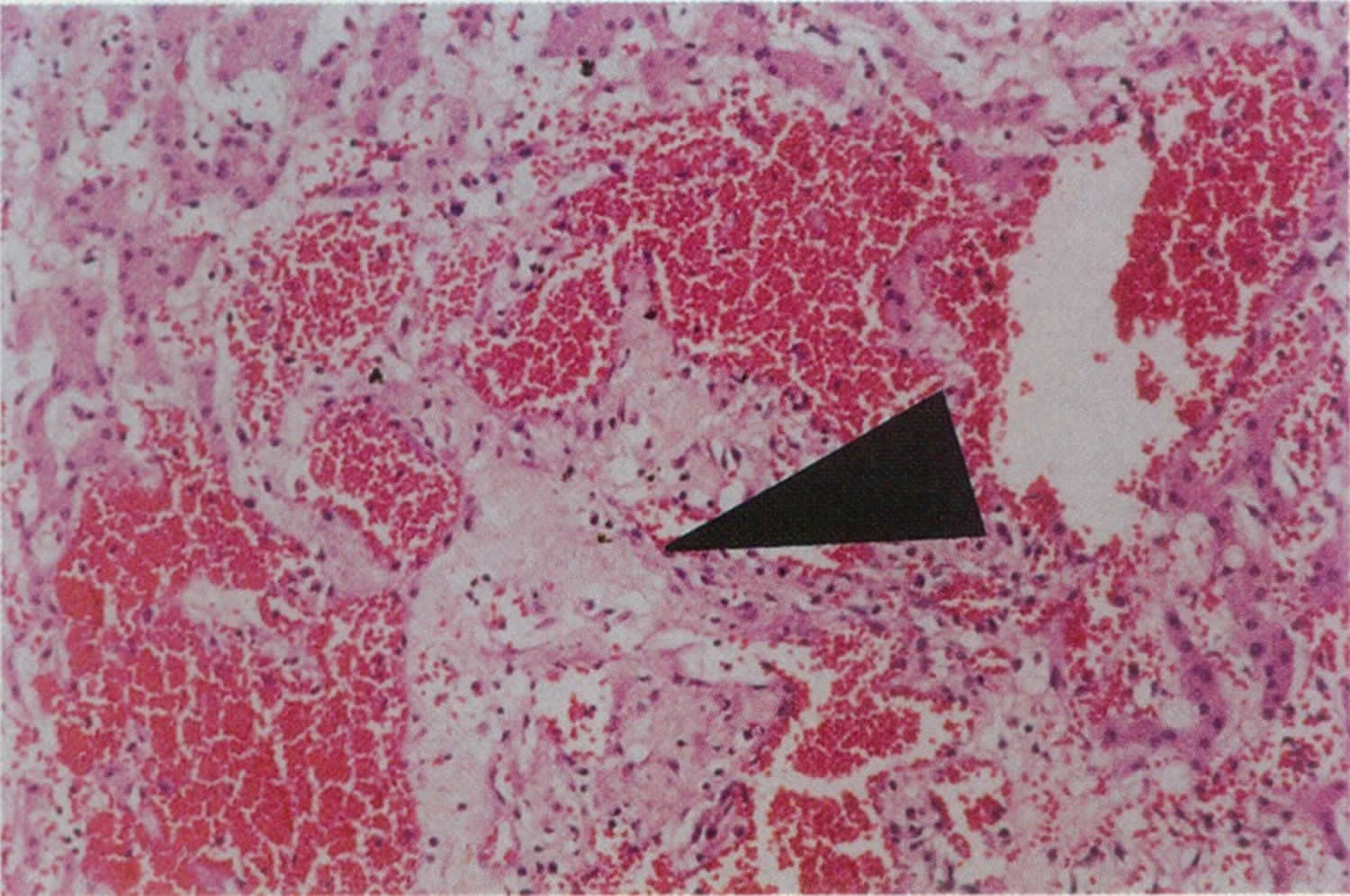
[Source 12 ]Figure 3. Peliosis hepatis liver biopsy specimen
Footnote: Liver biopsy specimen with hematoxylin and eosin staining at 45x magnification showing dilated vascular spaces (peliotic spaces) filled with red blood cells are surrounded by cords of hepatocytes. Note central area of pale myxoid stroma (arrow)
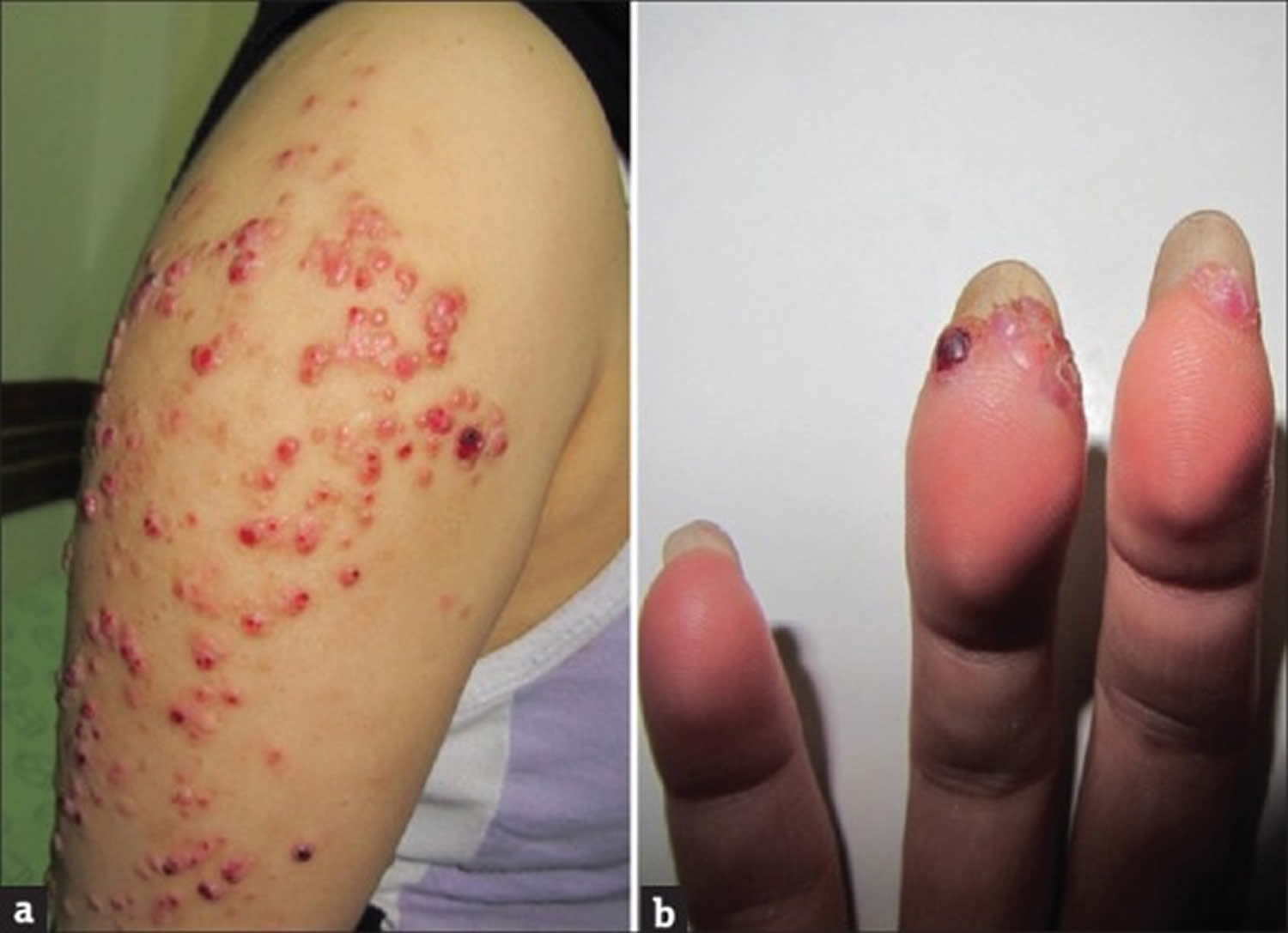
[Source 1 ]Figure 4. Bacillary angiomatosis
Footnotes: Bacillary angiomatosis clinical appearance. Angiomatous papule and nodules (a) arm and (b) fingers. A 26-year-old healthy woman had developed papules and nodules on the right arm 6 months ago. Despite treatment with short course systemic antibiotics the lesions had enlarged and gradually extended to the forearm and hand and also to her fingers.
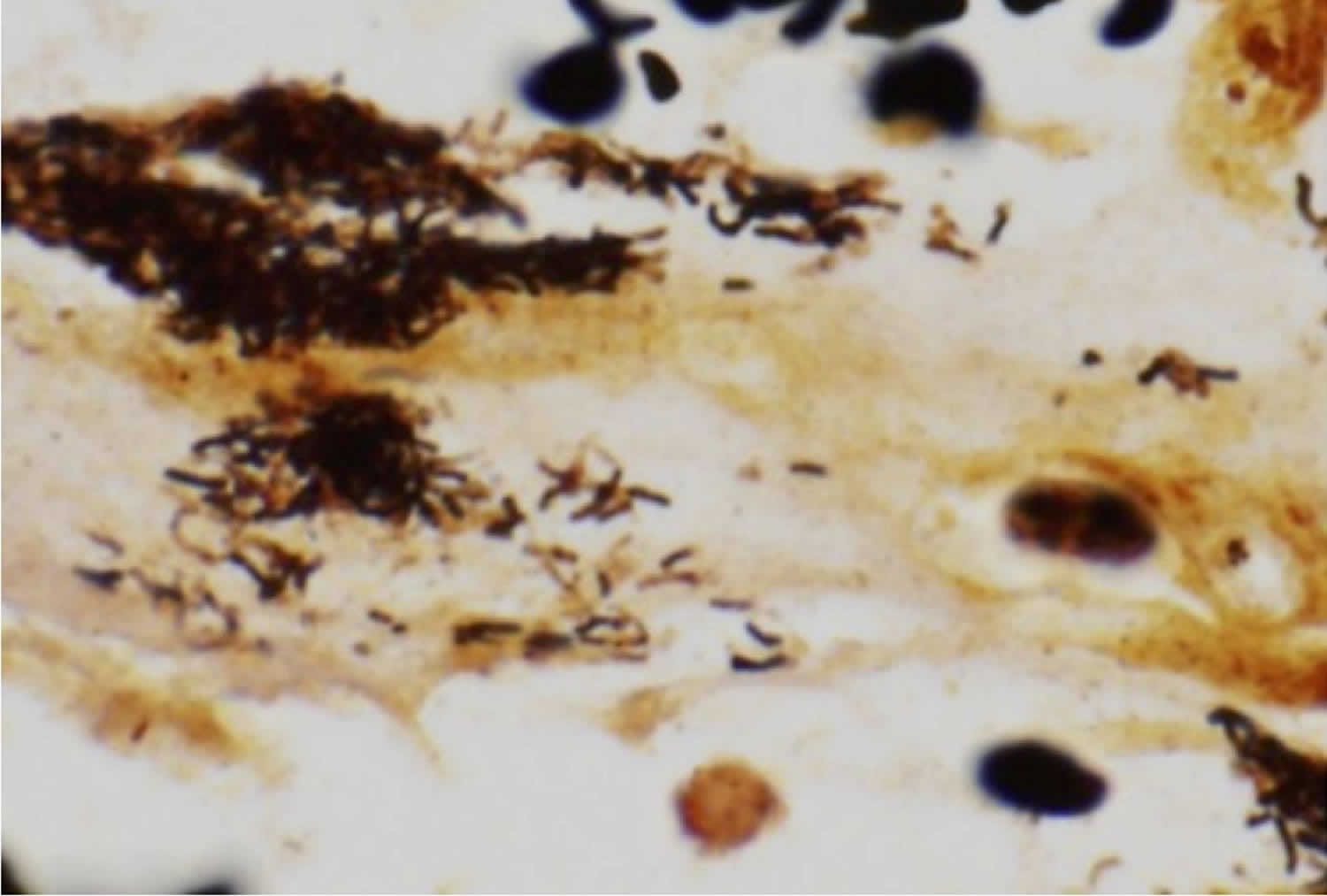
[Source 13 ]Figure 5. Bartonella bacteria
Footnote: Warthin-Starry stain demonstrating the characteristic pleomorphic bacilli of Bartonella bacteria in bone marrow core biopsy
[Source 2 ]Bacillary peliosis cause
Bartonella henselae the bacteria that cause bacillary peliosis commonly cause cat scratch disease or cat scratch fever 9, 8, 14, 15, 16, 17,18, 19. People become infected with Bartonella henselae from the scratch of domestic or feral cats, particularly kittens 16, 20, 21, 9. Cats can have fleas that carry Bartonella henselae bacteria. These bacteria can be transmitted from a cat to a person during a scratch that is contaminated with flea stool. Infected cats that lick a person’s open wound or bite can also spread Bartonella henselae bacteria. Some evidence suggests that these bacteria may spread directly to people by the bite of infected cat fleas, but this has not been proven.
Bartonella henselae infection or cat scratch disease occurs most often in children under the age of 15 (children 5 to 10 years of age) 9, 8, 14. Though more common in the southeast, cat scratch disease occurs throughout the United States. Stray cats are more likely than pets to be infected with B. henselae. In the United States, most cases of cat scratch disease occur in the fall and winter.
Bacillary peliosis symptoms
Most patients with bacillary peliosis develop symptoms such as anorexia, weight loss, enlarged tender lymph nodes, chills, headache, malaise and fever in addition to gastrointestinal symptoms or abdominal pain 2, 10, 5, 6, 7, 8, 9. Approximately 1% to 2% of patients with Bartonella infection develop severe systemic disease with involvement of the liver, spleen, bone, central nervous system (brain and spinal cord) or lung 7. Approximately 25% of patients have lymphadenopathy (enlarged lymph nodes), but often the classic skin papule of cat-scratch disease is absent 7, 8, 9.
. Bacillary peliosis can cause splenic peliosis and peliosis hepatis. Rupture with intraabdominal hemorrhage has previously been reported in both peliosis hepatis 22 and peliosis of the spleen 23, 24 and is another possible mode of presentation, underscoring the need for rapid diagnosis and therapy in these cases.
Bacillary peliosis diagnosis
Bacillary peliosis is very difficult to diagnose. Laboratory findings typically included a mild-to-moderate elevation of the serum aminotransferase level (alanine aminotransferase (ALT) and aspartate aminotransferase (AST)) and a moderate-to-severe elevation of the alkaline phosphatase (ALP) level in association with a normal or only mildly elevated bilirubin level 2. Progressive reduction in the number of blood cells (pancytopenia) occurred in some patients. Imaging studies showed enlarged abdominal lymph glands and a heterogeneous hepatic or splenic parenchyma in some cases 2.
Liver lesions are typically multiple and associated with abdominal lymphadenopathy and, in some cases, splenic lesions; many patients come to biopsy to exclude tumors 7, 14, 25. On laparotomy, the liver may be found to be studded with hard nodules of varying sizes 25.
Biopsy findings
Liver biopsy showed the presence of multiple blood-filled cystic spaces, foci of necrosis, fibromyxoid stroma, and clumps of granular purple material that correspond to organisms on Warthin-Starry stain and electron microscopy 26.
Bartonella infection may cause vascular proliferative lesions in the liver. Bacillary epithelioid angiomatosis is a vasoproliferative tissue reaction to Bartonella henselae or Bartonella quintana that usually occurs in immunocompromised hosts. Hepatic involvement shows sharply demarcated periportal areas in which the normal parenchyma is replaced by vascular tissue with extravasated erythrocytes, delicate spindle cells, neutrophils, and karyorrhexic debris, mimicking Kaposi sarcoma 27. Similar lesions have been designated as bacillary peliosis hepatis; these are characterized by the presence of multiple blood-filled cystic spaces, foci of necrosis, fibromyxoid stroma, and clumps of granular purple material that correspond to organisms on Warthin-Starry stain and electron microscopy 27. Bacillary peliosis hepatis can be mistaken for nonbacillary peliosis hepatis.
The hallmark lesion in hepatic cat-scratch disease is an irregular, stellate microabscess surrounded by a layer of palisading histiocytes, lymphocytes, and a rim of fibrous tissue 7, 25, 28. Younger lesions may show more necrosis with less organization of the inflammatory granulomatous response, whereas older lesions may show confluent granulomas with scarring and scant residual necrosis 7. These hepatic lesions are similar to the ones seen in lymph nodes in patients with cat-scratch disease, but they have also been noted in infections with Y. enterocolitica, F. tularensis, lymphogranuloma venereum, mycobacterial species, Candida, and Actinomyces 7. Other lesions may appear as small, rounded granulomas with giant cells and small foci of central necrosis, similar to caseating granulomas in mycobacterial or fungal infections. The background liver parenchyma shows sinusoidal dilatation, portal mixed but predominantly lymphocytic inflammatory infiltrates, portal fibrosis, periductal concentric fibrosis, and focal bile ductular proliferation 7. These changes are attributed to mass effect and are also seen in other infections that result in space-occupying lesions, such as pyogenic abscesses 7.
Warthin-Starry stains identify Bartonella bacteria in some cases (see Figure 5) and the organisms often cluster around vessels or along collagen fibers; the stain is not specific for the organism 8. Culture is difficult. The diagnosis can be confirmed by polymerase chain reaction (PCR) and Southern blot for Bartonella DNA on tissue, skin testing, or serology 9, 7, 28, 29.
Bacillary peliosis differential diagnosis
- Hepatic adenoma: Similar to peliosis, hepatic adenoma might also be associated with the long-term use of estrogens. In the case of diffuse peliosis hepatis, the differential diagnosis is relatively easy. In addition, the presence of fat in some adenomas is a useful sign to make a differential diagnosis. In certain instances, however, focal peliosis can be difficult to differentiate from adenomas. In these patients, biopsy is often required to reach a definitive diagnosis.
- Cavernous hemangioma: The typical enhancement pattern of hemangiomas (i.e., globular discontinuous contrast enhancement tends to be centripetal (periphery first) rather than centrifugal (center first)) is opposite of peliosis hepatis, and therefore differential diagnosis can be achieved in most patients. In addition, hemangiomas may be rather large lesions with a mass effect on the hepatic vessels, whereas peliotic lesions usually show no mass effect on hepatic vessels.
- Hepatocellular carcinoma (liver cancer): Hepatocellular carcinoma is usually hyperattenuating in the arterial phase with rapid washout in the portal venous phase and iso- or hypoattenuation in the delayed phase. Although rare, the possibility that peliosis hepatis may mimic the presence of hypervascular hepatocellular carcinoma has been reported in the literature. In these patients, biopsy is often necessary to reach a definitive diagnosis.
- Hepatic abscess (liver abscess): The differential diagnosis between peliosis hepatis and hepatic abscess is extremely important to avoid the percutaneous drainage of peliotic lesions, which can be dangerous and even fatal 30. With regard to imaging criteria, a hepatic abscess usually presents as a mass with a multiseptated or cluster-of-grapes appearance with nonenhancing contents.
- Focal nodular hyperplasia (benign tumor that forms in the liver): Focal nodular hyperplasias are typically homogeneously hyperattenuating masses on the arterial phase and isoattenuating on the portal venous and delayed phases. These lesions often have a central scar with low attenuation on the arterial and portal venous phases and enhancement on the delayed phase images. When such typical imaging characteristics of focal nodular hyperplasia are present, the differential diagnosis with peliosis hepatis can be achieved easily. Atypical forms of focal nodular hyperplasia may not show the characteristic enhancement patterns and the central scar just described, however, and thus pose some problems in the differential diagnosis with peliosis hepatis.
- Hepatic adenoma (an uncommon solid, benign liver lesion that develops in an otherwise normal-appearing liver) may contain fat
- Hypervascular metastases: Although some hypervascular metastases with fibrotic change can show mild hyperattenuation in the delayed phase, hypervascular metastases are usually totally hypoattenuating or isoattenuating in the delayed phase of contrast enhancement because of the rapid washout of contrast material. Thus, in general, peliotic lesions are rarely confused with hypervascular metastases.
- Hepatic sinusoidal dilation: usually the enhancement pattern is different on CT/MRI 31
Bacillary peliosis treatment
Bacillary peliosis is treated with antibiotic. Azithromyin, doxycycline, and gentamicin are all considered effective against Bartonella but the regimen of azithromycin and/or doxycycline is most commonly used in solid organ recipients 2. Macrolides (e.g., clarithromycin, azithromycin, erythromycin, clarithromycin, roxithromycin) also have immunomodulatory effects, which include reducing vascular endothelial growth factor (VEGF) activity, which may have additional benefits for Bartonella vascular lesions 32. The duration of therapy is not well-defined though given the high risk for morbidity in the immunosuppressed population and the propensity for relapse of Bartonella infections, a prolonged duration of antimicrobial therapy is typically favored 33.
Antibiotic treatment of bacillary angiomatosis and bacillary peliosis hepatis has never been studied systematically. Two criteria must be met to achieve successful eradication of Bartonella infections in the immunocompromised patient: first, the specific strain of Bartonella henselae and Bartonella quintana infecting the patient must have excellent in vivo susceptibility to the prescribed antibiotic, and second, the treatment must be of sufficient duration to prevent relapse 33. The first patient with bacillary angiomatosis to be described was treated empirically with erythromycin, and the lesions resolved completely34. Subsequently, erythromycin has become the drug of first choice and has successfully been used to treat many patients with bacillary angiomatosis 35, 36. Treatment of bacillary angiomatosis and bacillary peliosis hepatis with oral doxycycline (100 mg twice daily) has also been consistently successful 35. Lesions resolved in several patients treated with ceftriaxone or fluoroquinolone compounds 37, 38, but the progression of bacillary angiomatosis lesions in patients has been observed during treatment with ciprofloxacin 39. Additionally, a Bartonella species has been isolated from the blood or bacillary angiomatosis lesions of patients being treated with narrow-spectrum cephalosporins 40, nafcillin, gentamicin, and trimethoprim-sulfamethoxazole (but never from patients being treated with a macrolide, rifamycin, or a tetracycline) 41. Experts therefore do not recommend fluoroquinolones, trimethoprim-sulfamethoxazole, or narrow-spectrum cephalosporins for the treatment of bacillary angiomatosis or bacillary peliosis hepatis 35. Treatment failures have been reported with many different antibiotics, and these are usually attributable to a lack of susceptibility of Bartonella in vivo and/or an insufficient duration of therapy 35, 42.
The drug of choice for the treatment of bacillary angiomatosis is erythromycin given p.o. (500 mg p.o. four times daily) for 3 months, but intravenous (IV) administration should be used in patients with severe disease 35. Patients intolerant of erythromycin can be treated with doxycycline (100 mg p.o. or i.v. twice daily) 35, 43, 44. The response to treatment appears to be equivalent whether erythromycin or doxycycline is prescribed 45. Combination therapy, with the addition of rifampin (300 mg p.o. twice daily) to either erythromycin or doxycycline, is recommended for immunocompromised patients with acute, life-threatening Bartonella infection. The intravenous route is especially important in cases of gastrointestinal intolerance or poor absorption. The combination of doxycycline and rifampin is preferred for the treatment of patients with CNS Bartonella infection because of the superior CNS penetration of doxycycline compared with those of the other first-line antibiotics.
The response to treatment can be dramatic in immunocompromised patients. In one patient who received a single 250-mg oral dose of erythromycin, blood cultures became sterile and a palpable subcutaneous lesion disappeared within hours (but recurred months later). More chronic lesions resolve more slowly, but after approximately 4 to 7 days of therapy, cutaneous bacillary angiomatosis lesions usually improve and resolve completely after 1 month of treatment 46. Bacillary bacillary peliosis hepatis responds more slowly than cutaneous bacillary angiomatosis, but hepatic lesions should improve after several months of treatment.
Relapses of bacillary peliosis hepatis and bacillary angiomatosis lesions in bone and skin have been reported frequently 47, 40, 48, 49. Relapses occur when antibiotics are given for a shorter duration (<3 months), especially in severely immunocompromised HIV-infected patients 35, 38. For this reason and from extensive experience treating patients with bacillary angiomatosis and bacillary peliosis hepatis, experts recommend that treatment be given for at least 3 months for bacillary angiomatosis and 4 months for bacillary peliosis hepatis 45, 50. All immunocompromised patients with a Bartonella infection should receive antibiotic therapy (erythromycin 500 mg p.o. four times daily or doxycycline 100 mg p.o. twice daily); patients who have relapses after the recommended treatment should then receive secondary prophylactic antibiotic treatment with erythromycin (500 mg p.o. four times daily) or doxycycline (100 mg p.o. twice daily) as long as they are immunocompromised 50. Of note, AIDS patients receiving prophylaxis with a macrolide or rifamycin antibiotic for Mycobacterium avium complex infection appear to be protected from developing infections with Bartonella species 41. Some immunocompromised patients develop a potentially life-threatening Jarisch-Herxheimer-like reaction within hours after institution of antibiotic therapy 40. Physicians should advise patients of this possible treatment complication, and patients with severe respiratory and/or cardiovascular compromise should be monitored carefully following institution of antimicrobial therapy 45.
- Perkocha LA, Geaghan SM, Yen TS, Nishimura SL, Chan SP, Garcia-Kennedy R, Honda G, Stoloff AC, Klein HZ, Goldman RL, et al. Clinical and pathological features of bacillary peliosis hepatis in association with human immunodeficiency virus infection. N Engl J Med. 1990 Dec 6;323(23):1581-6. doi: 10.1056/NEJM199012063232302[↩][↩][↩]
- Steed D, Collins J, Farris AB, Guarner J, Yarar D, Friedman-Moraco R, Doane T, Pouch S, Marshall Lyon G 3rd, Woodworth MH. Haemophagocytic lymphohistiocytosis associated with bartonella peliosis hepatis following kidney transplantation in a patient with HIV. Lancet Infect Dis. 2022 Oct;22(10):e303-e309. doi: 10.1016/S1473-3099(22)00276-6[↩][↩][↩][↩][↩][↩][↩]
- Maurin M, Birtles R, Raoult D. Current knowledge of Bartonella species. Eur J Clin Microbiol Infect Dis. 1997 Jul;16(7):487-506. doi: 10.1007/BF01708232[↩]
- Angelakis E, Raoult D. Pathogenicity and treatment of Bartonella infections. Int J Antimicrob Agents. 2014 Jul;44(1):16-25. doi: 10.1016/j.ijantimicag.2014.04.006[↩]
- Mohle-Boetani JC, Koehler JE, Berger TG, LeBoit PE, Kemper CA, Reingold AL, Plikaytis BD, Wenger JD, Tappero JW. Bacillary angiomatosis and bacillary peliosis in patients infected with human immunodeficiency virus: clinical characteristics in a case-control study. Clin Infect Dis. 1996 May;22(5):794-800. doi: 10.1093/clinids/22.5.794[↩][↩]
- Mosepele M, Mazo D, Cohn J. Bartonella infection in immunocompromised hosts: immunology of vascular infection and vasoproliferation. Clin Dev Immunol. 2012;2012:612809. doi: 10.1155/2012/612809[↩][↩]
- Lamps LW, Gray GF, Scott MA. The histologic spectrum of hepatic cat scratch disease. A series of six cases with confirmed Bartonella henselae infection. Am J Surg Pathol. 1996 Oct;20(10):1253-9. doi: 10.1097/00000478-199610000-00011[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Lenoir AA, Storch GA, DeSchryver-Kecskemeti K, Shackelford GD, Rothbaum RJ, Wear DJ, Rosenblum JL. Granulomatous hepatitis associated with cat scratch disease. Lancet. 1988 May 21;1(8595):1132-6. doi: 10.1016/s0140-6736(88)91952-6[↩][↩][↩][↩][↩][↩][↩]
- Arisoy ES, Correa AG, Wagner ML, Kaplan SL. Hepatosplenic cat-scratch disease in children: selected clinical features and treatment. Clin Infect Dis. 1999 Apr;28(4):778-84. doi: 10.1086/515197[↩][↩][↩][↩][↩][↩][↩][↩]
- Bass JW, Vincent JM, Person DA. The expanding spectrum of Bartonella infections: II. Cat-scratch disease. Pediatr Infect Dis J. 1997 Feb;16(2):163-79. doi: 10.1097/00006454-199702000-00002[↩][↩]
- Bächler P, Baladron MJ, Menias C, Beddings I, Loch R, Zalaquett E, Vargas M, Connolly S, Bhalla S, Huete Á. Multimodality Imaging of Liver Infections: Differential Diagnosis and Potential Pitfalls. Radiographics. 2016 Jul-Aug;36(4):1001-23. doi: 10.1148/rg.2016150196[↩]
- Iannaccone R, Federle MP, Brancatelli G, Matsui O, Fishman EK, Narra VR, Grazioli L, McCarthy SM, Piacentini F, Maruzzelli L, Passariello R, Vilgrain V. Peliosis hepatis: spectrum of imaging findings. AJR Am J Roentgenol. 2006 Jul;187(1):W43-52. https://doi.org/10.2214/AJR.05.0167[↩]
- Iraji F, Pourazizi M, Abtahi-Naeini B, Meidani M, Rajabi P. Bacillary Angiomatosis in Immunocompetent Patient with Atypical Manifestations. Indian J Dermatol. 2015 Sep-Oct;60(5):523. doi: 10.4103/0019-5154.164444[↩]
- Ventura A, Massei F, Not T, Massimetti M, Bussani R, Maggiore G. Systemic Bartonella henselae infection with hepatosplenic involvement. J Pediatr Gastroenterol Nutr. 1999 Jul;29(1):52-6. doi: 10.1097/00005176-199907000-00014[↩][↩][↩]
- Carithers HA. Cat-scratch disease. An overview based on a study of 1,200 patients. Am J Dis Child. 1985 Nov;139(11):1124-33. doi: 10.1001/archpedi.1985.02140130062031[↩]
- Margileth AM. Cat scratch disease. Adv Pediatr Infect Dis. 1993;8:1-21.[↩][↩]
- Chomel BB, Boulouis HJ, Breitschwerdt EB. Cat scratch disease and other zoonotic Bartonella infections. J Am Vet Med Assoc. 2004 Apr 15;224(8):1270-9. doi: 10.2460/javma.2004.224.1270[↩]
- Chomel BB, Kasten RW, Floyd-Hawkins K, Chi B, Yamamoto K, Roberts-Wilson J, Gurfield AN, Abbott RC, Pedersen NC, Koehler JE. Experimental transmission of Bartonella henselae by the cat flea. J Clin Microbiol. 1996 Aug;34(8):1952-6. doi: 10.1128/jcm.34.8.1952-1956.1996[↩]
- Chomel BB, Abbott RC, Kasten RW, Floyd-Hawkins KA, Kass PH, Glaser CA, Pedersen NC, Koehler JE. Bartonella henselae prevalence in domestic cats in California: risk factors and association between bacteremia and antibody titers. J Clin Microbiol. 1995 Sep;33(9):2445-50. doi: 10.1128/jcm.33.9.2445-2450.1995[↩]
- Scott MA, McCurley TL, Vnencak-Jones CL, Hager C, McCoy JA, Anderson B, Collins RD, Edwards KM. Cat scratch disease: detection of Bartonella henselae DNA in archival biopsies from patients with clinically, serologically, and histologically defined disease. Am J Pathol. 1996 Dec;149(6):2161-7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1865337/pdf/amjpathol00036-0364.pdf[↩]
- Hajjaji N, Hocqueloux L, Kerdraon R, Bret L. Bone infection in cat-scratch disease: a review of the literature. J Infect. 2007 May;54(5):417-21. doi: 10.1016/j.jinf.2006.10.045[↩]
- Bagheri SA, Boyer JL. Peliosis hepatis associated with androgenic-anabolic steroid therapy. A severe form of hepatic injury. Ann Intern Med. 1974 Nov;81(5):610-8. doi: 10.7326/0003-4819-81-5-610[↩]
- Taxy JB. Peliosis: a morphologic curiosity becomes an iatrogenic problem. Hum Pathol. 1978 May;9(3):331-40. doi: 10.1016/s0046-8177(78)80090-2[↩]
- Garcia RL, Khan MK, Berlin RB. Peliosis of the spleen with rupture. Hum Pathol. 1982 Feb;13(2):177-9. doi: 10.1016/s0046-8177(82)80123-8[↩]
- Malatack JJ, Jaffe R. Granulomatous hepatitis in three children due to cat-scratch disease without peripheral adenopathy. An unrecognized cause of fever of unknown origin. Am J Dis Child. 1993 Sep;147(9):949-53. doi: 10.1001/archpedi.1993.02160330039014[↩][↩][↩]
- Theresa A. Steeper, Hanan Rosenstein, John Weiser, Subbarao Inampudi, Dale C. Snover, Bacillary Epithelioid Angiomatosis Involving the Liver, Spleen, and Skin in an AIDS Patient with Concurrent Kaposi’s Sarcoma, American Journal of Clinical Pathology, Volume 97, Issue 5, 1 May 1992, Pages 713–718, https://doi.org/10.1093/ajcp/97.5.713[↩]
- Steeper TA, Rosenstein H, Weiser J, Inampudi S, Snover DC. Bacillary epithelioid angiomatosis involving the liver, spleen, and skin in an AIDS patient with concurrent Kaposi’s sarcoma. Am J Clin Pathol. 1992 May;97(5):713-8. doi: 10.1093/ajcp/97.5.713[↩][↩]
- VanderHeyden TR, Yong SL, Breitschwerdt EB, Maggi RG, Mihalik AR, Parada JP, Fimmel CJ. Granulomatous hepatitis due to Bartonella henselae infection in an immunocompetent patient. BMC Infect Dis. 2012 Jan 23;12:17. doi: 10.1186/1471-2334-12-17[↩][↩]
- Liston TE, Koehler JE. Granulomatous hepatitis and necrotizing splenitis due to Bartonella henselae in a patient with cancer: case report and review of hepatosplenic manifestations of bartonella infection. Clin Infect Dis. 1996 Jun;22(6):951-7. doi: 10.1093/clinids/22.6.951[↩]
- Cohen GS, Ball DS, Boyd-Kranis R, Gembala RB, Wurzel J. Peliosis hepatis mimicking hepatic abscess: fatal outcome following percutaneous drainage. J Vasc Interv Radiol. 1994 Jul-Aug;5(4):643-5. doi: 10.1016/s1051-0443(94)71572-4[↩]
- Yang DM, Jung DH, Park CH, Kim JE, Choi SJ. Imaging findings of hepatic sinusoidal dilatation. AJR Am J Roentgenol. 2004 Oct;183(4):1075-7. doi: 10.2214/ajr.183.4.1831075[↩]
- Zimmermann P, Ziesenitz VC, Curtis N, Ritz N. The Immunomodulatory Effects of Macrolides-A Systematic Review of the Underlying Mechanisms. Front Immunol. 2018 Mar 13;9:302. doi: 10.3389/fimmu.2018.00302[↩]
- Rolain JM, Brouqui P, Koehler JE, Maguina C, Dolan MJ, Raoult D. Recommendations for treatment of human infections caused by Bartonella species. Antimicrob Agents Chemother. 2004 Jun;48(6):1921-33. doi: 10.1128/AAC.48.6.1921-1933.2004[↩][↩]
- Stoler MH, Bonfiglio TA, Steigbigel RT, Pereira M. An atypical subcutaneous infection associated with acquired immune deficiency syndrome. Am J Clin Pathol. 1983 Nov;80(5):714-8. doi: 10.1093/ajcp/80.5.714[↩]
- Koehler JE, Tappero JW. Bacillary angiomatosis and bacillary peliosis in patients infected with human immunodeficiency virus. Clin Infect Dis. 1993 Oct;17(4):612-24. doi: 10.1093/clinids/17.4.612[↩][↩][↩][↩][↩][↩][↩]
- Tappero JW, Koehler JE, Berger TG, Cockerell CJ, Lee TH, Busch MP, Stites DP, Mohle-Boetani J, Reingold AL, LeBoit PE. Bacillary angiomatosis and bacillary splenitis in immunocompetent adults. Ann Intern Med. 1993 Mar 1;118(5):363-5. doi: 10.7326/0003-4819-118-5-199303010-00007[↩]
- Lucey D, Dolan MJ, Moss CW, Garcia M, Hollis DG, Wegner S, Morgan G, Almeida R, Leong D, Greisen KS, et al. Relapsing illness due to Rochalimaea henselae in immunocompetent hosts: implication for therapy and new epidemiological associations. Clin Infect Dis. 1992 Mar;14(3):683-8. doi: 10.1093/clinids/14.3.683[↩]
- Slater LN, Welch DF, Hensel D, Coody DW. A newly recognized fastidious gram-negative pathogen as a cause of fever and bacteremia. N Engl J Med. 1990 Dec 6;323(23):1587-93. doi: 10.1056/NEJM199012063232303[↩][↩]
- Tappero JW, Koehler JE. Cat-scratch Disease and Bacillary Angiomatosis. JAMA. 1991;266(14):1938–1939. doi:10.1001/jama.1991.03470140050020[↩]
- Koehler JE, Quinn FD, Berger TG, LeBoit PE, Tappero JW. Isolation of Rochalimaea species from cutaneous and osseous lesions of bacillary angiomatosis. N Engl J Med. 1992 Dec 3;327(23):1625-31. doi: 10.1056/NEJM199212033272303[↩][↩][↩]
- Koehler JE, Sanchez MA, Garrido CS, Whitfeld MJ, Chen FM, Berger TG, Rodriguez-Barradas MC, LeBoit PE, Tappero JW. Molecular epidemiology of bartonella infections in patients with bacillary angiomatosis-peliosis. N Engl J Med. 1997 Dec 25;337(26):1876-83. doi: 10.1056/NEJM199712253372603[↩][↩]
- Maurin M, Raoult D. Antimicrobial susceptibility of Rochalimaea quintana, Rochalimaea vinsonii, and the newly recognized Rochalimaea henselae. J Antimicrob Chemother. 1993 Oct;32(4):587-94. doi: 10.1093/jac/32.4.587[↩]
- Koehler JE. Bartonella-associated infections in HIV-infected patients. AIDS Clin Care. 1995 Dec;7(12):97-102.[↩]
- Ramírez Ramírez CR, Saavedra S, Ramírez Ronda CH. Bacillary angiomatosis: microbiology, histopathology, clinical presentation, diagnosis and management. Bol Asoc Med P R. 1996 Apr-Jun;88(4-6):46-51.[↩]
- Koehler, J. E., and D. A. Relman. 2002. Bartonella species, p. 925-931. In V. L. Yu, R. Weber, and D. Raoult (ed.), Antimicrobial therapy and vaccines. Apple Trees Production, LLC, New York, N.Y.[↩][↩][↩]
- Berger TG, Perkocha LA. Bacillary angiomatosis. AIDS Clin Rev. 1991:81-95.[↩]
- Gazineo JL, Trope BM, Maceira JP, May SB, Coelho JM, Lambert JS, Nogueira SA. Bacillary angiomatosis: description of 13 cases reported in five reference centers for AIDS treatment in Rio de Janeiro, Brazil. Rev Inst Med Trop Sao Paulo. 2001 Jan-Feb;43(1):1-6. doi: 10.1590/s0036-46652001000100001[↩]
- Krekorian TD, Radner AB, Alcorn JM, Haghighi P, Fang FC. Biliary obstruction caused by epithelioid angiomatosis in a patient with AIDS. Am J Med. 1990 Dec;89(6):820-2. doi: 10.1016/0002-9343(90)90231-2[↩]
- Szaniawski WK, Don PC, Bitterman SR, Schachner JR. Epithelioid angiomatosis in patients with AIDS. Report of seven cases and review of the literature. J Am Acad Dermatol. 1990 Jul;23(1):41-8. doi: 10.1016/0190-9622(90)70183-i[↩]
- Cotell SL, Noskin GA. Bacillary angiomatosis. Clinical and histologic features, diagnosis, and treatment. Arch Intern Med. 1994 Mar 14;154(5):524-8. doi: 10.1001/archinte.154.5.524[↩][↩]