Contents
What is Triploidy
Triploidy also called triploid syndrome, triploidy syndrome or chromosome triploidy syndrome is a medical term to describe is a rare chromosomal abnormality where there are 3 copies of single chromosome in a cell (69 chromosomes), instead of the usual 23 pairs of chromosomes (46 chromosomes) that may occur in 2 to 3% of all pregnancies (Figure 2) 1, 2, 3, 4, 5, 6, 7, 8, 9. As such, triploidy (69 chromosomes) is estimated to occur in 1 of 3,500 pregnancies at 12 weeks, 1 in 30,000 at 16 weeks and 1 in 250,000 at 20 weeks gestation 3. Triploidy is a common cause of early first-trimester miscarriages with triploidy accounting for approximately 20% of chromosomally abnormal first-trimester miscarriages. Only very rarely are babies with triploidy born alive as most are stillborn and even more rarely do they survive beyond the first few days of life 5.
Humans typically have 46 chromosomes (23 pairs of chromosomes) in each cell of their body, made up of 22 paired chromosomes (also called autosomes) and one pair of sex chromosomes (i.e., XX or XY sex chromosomes since they, the X and Y chromosome, help determine what sex or gender you are). These 46 chromosomes (23 pairs of chromosomes) contain between 20,000 and 25,000 genes.
People usually have two copies of each chromosome. One copy is inherited from their mother (via the egg) and the other from their father (via the sperm). A sperm and an egg each contain one set of 23 chromosomes. When the sperm fertilises the egg, two copies of each chromosome are present (and therefore two copies of each gene), and so an embryo forms.
The chromosomes that determine the sex of the baby (X and Y chromosomes) are called sex chromosomes. Typically, the mother’s egg contributes an X chromosome, and the father’s sperm provides either an X or a Y chromosome. A person with an XX pairing of sex chromosomes is biologically female, while a person with an XY pairing is biologically male.
As well as determining sex, the sex chromosomes carry genes that control other body functions. There are many genes located on the X chromosome, but only a few on the Y chromosome. Genes that are on the X chromosome are said to be X-linked. Genes that are on the Y chromosome are said to be Y-linked.
Fetuses with triploidy, the extra set of chromosomes can be from the mother (digynic triploidy) or from the father (diandric triploidy) in origin. On prenatal ultrasound scan, triploidy of maternal origin (digynic triploidy) is typically associated with severe intrauterine growth retardation (IUGR) and a small non‐cystic placenta and have a better chance to reach the second trimester of gestation or even to be born 1, 10. In contrast, triploidy of paternal origin (diandric triploidy) often demonstrates normal fetal growth with multiple placental cysts 11, 12, 10.
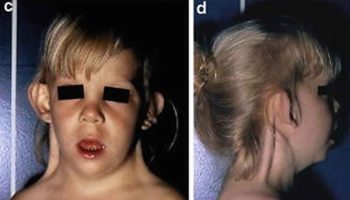
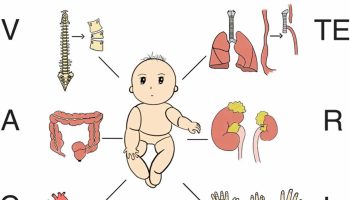
Infants affected with complete triploidy may have a variety of birth defects such as heart defects, abnormal brain development, adrenal and kidney defects (cystic kidneys), spinal cord malformations (neural tube defects), severe intrauterine growth retardation (IUGR) and abnormal facial features (widely spaced eyes, low nasal bridge, low-set malformed ears, small jaw, absent/small eye, and cleft lip and palate) 13. The third and fourth fingers of the hands and the second and third toes of the feet may be fused (also called syndactyly) and the hands may have unusual single palmar creases (simian creases) 13. There may also be liver and gallbladder defects, twisted intestines and deformities of the fingers and toes. The placenta in triploidy may be immature, large, and filled with cysts. Individuals who are mosaic will survive longer than those with complete triploidy but usually have intellectual disability, developmental delay, depression, seizures, short stature, obesity and other abnormalities. Mosaic triploidy means that some cells have the normal number of 46 chromosomes and other cells have 69 chromosomes per cell.
The pregnant mother carrying a triploidy fetus sometimes experiences increase in blood pressure (hypertension), swelling (edema), and excretion of albumin in the urine (albuminuria). This condition is called toxemia or preeclampsia.
Triploidy may be prenatally diagnosed through cytogenetic analysis of cells obtained through procedures such as amniocentesis. Fetal nuchal translucency, ventral wall defects, syndactyly, and other anatomic abnormalities on prenatal ultrasound scan in the first trimester is frequently increased in fetuses with triploidy 14, 15, 16. The incidence of triploidy at approximately 11–13 weeks gestation has been estimated to be approximately 1 in 4800 pregnancies 1, decreasing to less than 1 per 27,000 in the second trimester 17. Rare cases (possibly mosaic or chimeric) can survive to the third trimester, and these usually result in stillbirth or neonatal death 18. Although reports exist regarding multiple recurrences of maternal triploidy (digynic triploidy) pregnancies 19, the overall risk for recurrence has not been established. For paternal triploidy (diandric triploidy) pregnancies, recurrence risk is approximately twice the general population risk 20.
Triploidy is also associated with elevated maternal serum alpha fetoprotein (AFP) and total and beta-human chorionic gonadotropin (hCG) levels and low maternal serum pregnancy-assisted plasma protein-A (PAPP-A) levels in the first trimester 21, 22. Fetal ultrasound examination during first and second trimester of gestation is most important, and gives support in discovery of chromosomal aberration of the fetus and congenital anomalies 23.
For pregnancies with a diagnosis of triploidy, serial human chorionic gonadotropin (hCG) tests are recommended to ensure complete removal of residual trophoblastic tissue 24, 25.
Because of the risk for maternal malignant trophoblastic disease, early detection of triploidy is important 26. Conventional first and second trimester maternal serum biochemical tests can help screen for triploid pregnancies 27, 28, but in some countries serum testing is less widely utilized because of the increasing use of cell‐free DNA (cfDNA) based non‐invasive prenatal testing for autosomal trisomies, sex chromosome abnormalities and some other imbalances. Counting‐based cell‐free DNA (cfDNA) methods are unable to routinely detect triploidy because there is no proportional change in the number of DNA fragments across the different autosomes. As the American College of Obstetricians and Gynecologists noted in a 2020 Practice Bulletin 29, single‐nucleotide polymorphism‐based non‐invasive prenatal testing can identify triploidy by detecting the presence of the additional haploid chromosome set 30. However, the single‐nucleotide polymorphism pattern seen with triploidy can sometimes be difficult to distinguish from the pattern seen with dizygotic twins, particularly when the fetal fraction is low 31
Treatment of triploid syndrome is symptomatic and supportive. The most frequent cause of death is pneumonia and general respiratory problems.
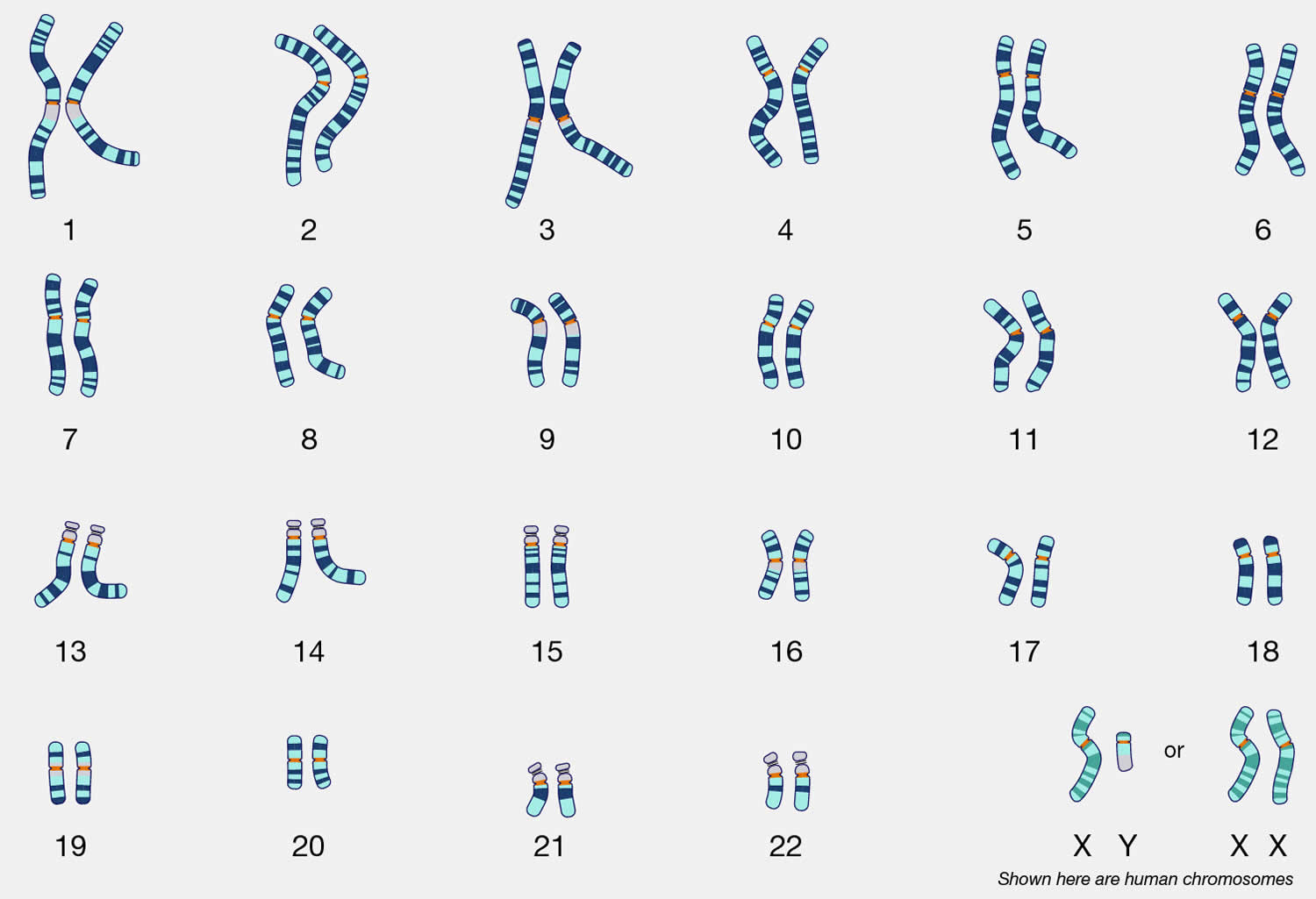
Figure 1. Normal human karyotype (46 chromosomes)
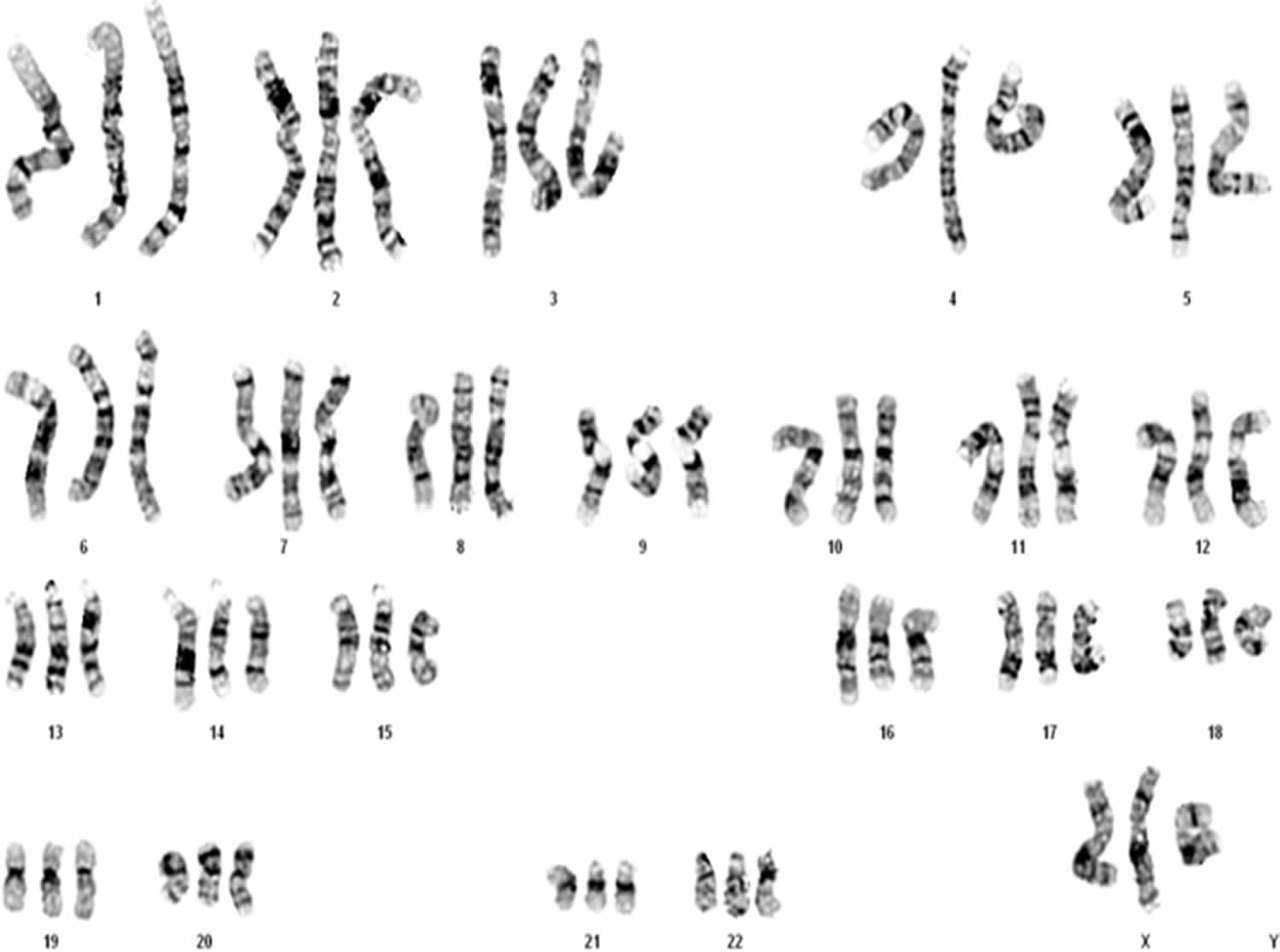
[Source 32 ]Figure 2. Triploidy (karyotype of 69XXX)
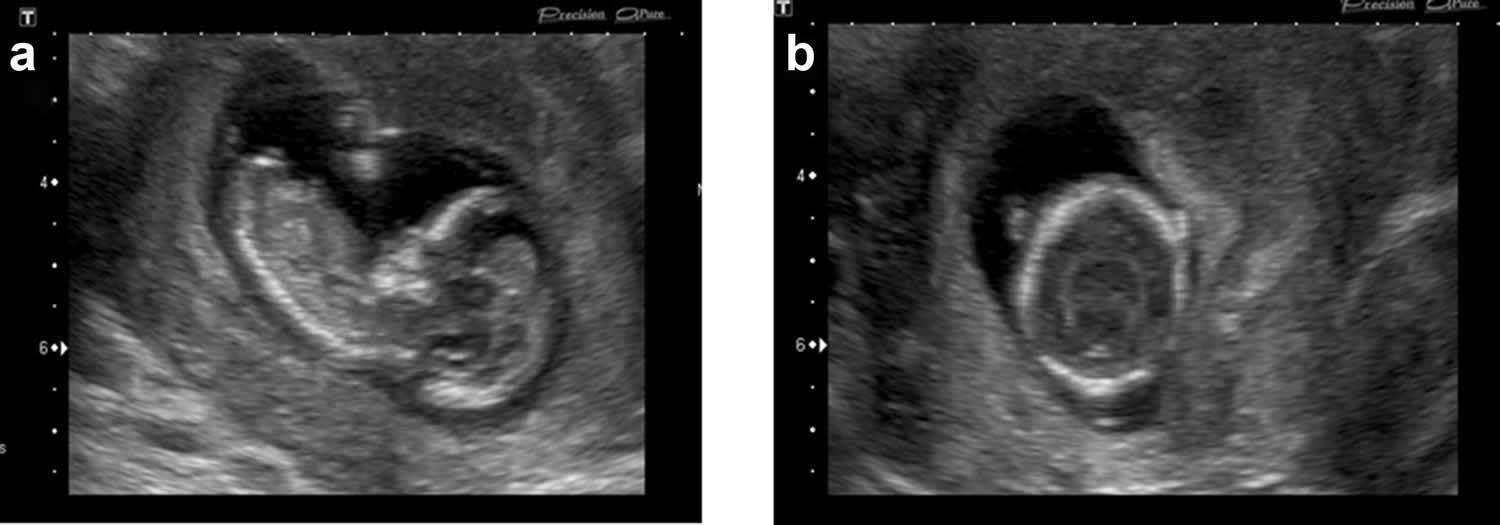
[Source 33 ]Figure 3. Triploidy ultrasound
Footnotes: Prenatal ultrasound of triploidy fetus at 12 weeks of gestation shows (a) a relative large head and an absent nasal bone, and (b) semilobar holoprosencephaly. The pregnancy was terminated subsequently, and a 50-g fetus was delivered with a relatively enlarged head and premaxillary agenesis with a median facial cleft. The placenta was small and non-cystic. Postnatal cytogenetic analysis of the umbilical cord revealed a karyotype of 69, XXX (Figure 2).
[Source 33 ]What is a karyotype?
A karyotype is an individual’s complete set of chromosomes (see Figure 1) 32. Karyotype describes the amount of chromosome count and morphology under the light microscope. Karyotype also refers to a laboratory-produced image of a person’s chromosomes isolated from an individual cell and arranged in numerical order 32. Karyotypes can be used for many purposes, such as to look for abnormalities in chromosome number or structure in prenatal diagnostics or tumor studies. Also, to understand cellular function, taxonomic relationships, and providing information about past evolutionary events.
To make a karyotype, scientists take a picture of the chromosomes from one cell, cut them out, and arrange them using size, banding pattern, and centromere positions as guides. The typical human karyotype contains 22 pairs of autosomal chromosomes and one pair of sex chromosomes (i.e., XX or XY sex chromosomes) (see Figure 1). The most common karyotypes for a female contain two X chromosomes and are denoted for the sex XX. Males usually have both an X and a Y chromosome, denoted for the sex XY.
What is a chromosome?
Chromosomes are thread-like structures located inside the nucleus of animal and plant cells. Each chromosome is made of protein and a single molecule of deoxyribonucleic acid (DNA). Passed from parents to offspring, DNA contains the specific instructions that make each type of living creature unique.
The term chromosome comes from the Greek words for color (chroma) and body (soma). Scientists gave this name to chromosomes because they are cell structures, or bodies, that are strongly stained by some colorful dyes used in research.
For an organism to grow and function properly, cells must constantly divide to produce new cells to replace old, worn-out cells. During cell division, it is essential that DNA remains intact and evenly distributed among cells. Chromosomes are a key part of the process that ensures DNA is accurately copied and distributed in the vast majority of cell divisions. Still, mistakes do occur on rare occasions.
Changes in the number or structure of chromosomes in new cells may lead to serious problems. For example, in humans, one type of leukemia and some other cancers are caused by defective chromosomes made up of joined pieces of broken chromosomes.
It is also crucial that reproductive cells also called the gametes, such as eggs and sperm, contain the right number of chromosomes and that those chromosomes have the correct structure. If not, the resulting offspring may fail to develop properly. For example, people with triploidy syndrome have 3 copies of each chromosome in a cell (68–80 chromosomes), instead of the usual 23 pairs of chromosomes (46 chromosomes).
What is meiosis?
Meiosis is a type of cell division in sexually reproducing organisms that reduces the number of chromosomes in the sex cells also called the gametes such as an egg and sperm 34. Human body cells also called somatic cells are diploid which means they have two sets of chromosomes, one from each parent 34. Humans have 46 chromosomes in almost every cell, 23 that came from one of your parents and 23 very similar chromosomes that came from the other one of your parents. To maintain this state, the egg and sperm that unite during fertilization must be haploid meaning each sex cells (an egg or a sperm) must have a single set of chromosomes. During meiosis, each diploid cell undergoes two rounds of division to yield four haploid daughter cells — the gametes.
It is really important to have the right number of chromosomes in a cell. If a cell has extra chromosomes or is missing a chromosome, that can have very substantial impacts on how it functions. You can think of meiosis as a way cells very carefully count and divide their chromosomes so that each gamete such as an egg and sperm has exactly 23 chromosomes. Then when an egg with its 23 chromosomes is fertilized by a sperm with its 23 chromosomes, the resulting fertilized egg has exactly 46 chromosomes (23 that came from your mother and another 23 that came from your father). And a new human that grows from that fertilized egg will have 46 chromosomes in all of its cells.
Triploidy causes
Triploidy is the presence of a complete additional set of chromosomes. Three different mechanisms have been described that are responsible for triploidy 5, 35, 36, 37, 38, 39, 40:
- Fertilization of an egg by a sperm that has an extra set of chromosomes due to non-disjunction event in meiosis 1 or 2 of spermatogenesis (the production of sperm from the primordial germ cells);
- Fertilization of an egg that has an extra set of chromosomes by a normal sperm due to non-disjunction in meiosis 1 or 2 of oogenesis (the process of formation of ovum);
- Fertilization of an egg by two sperms also known as dispermy. Dispermy is considered to be the most common cause of triploidy.
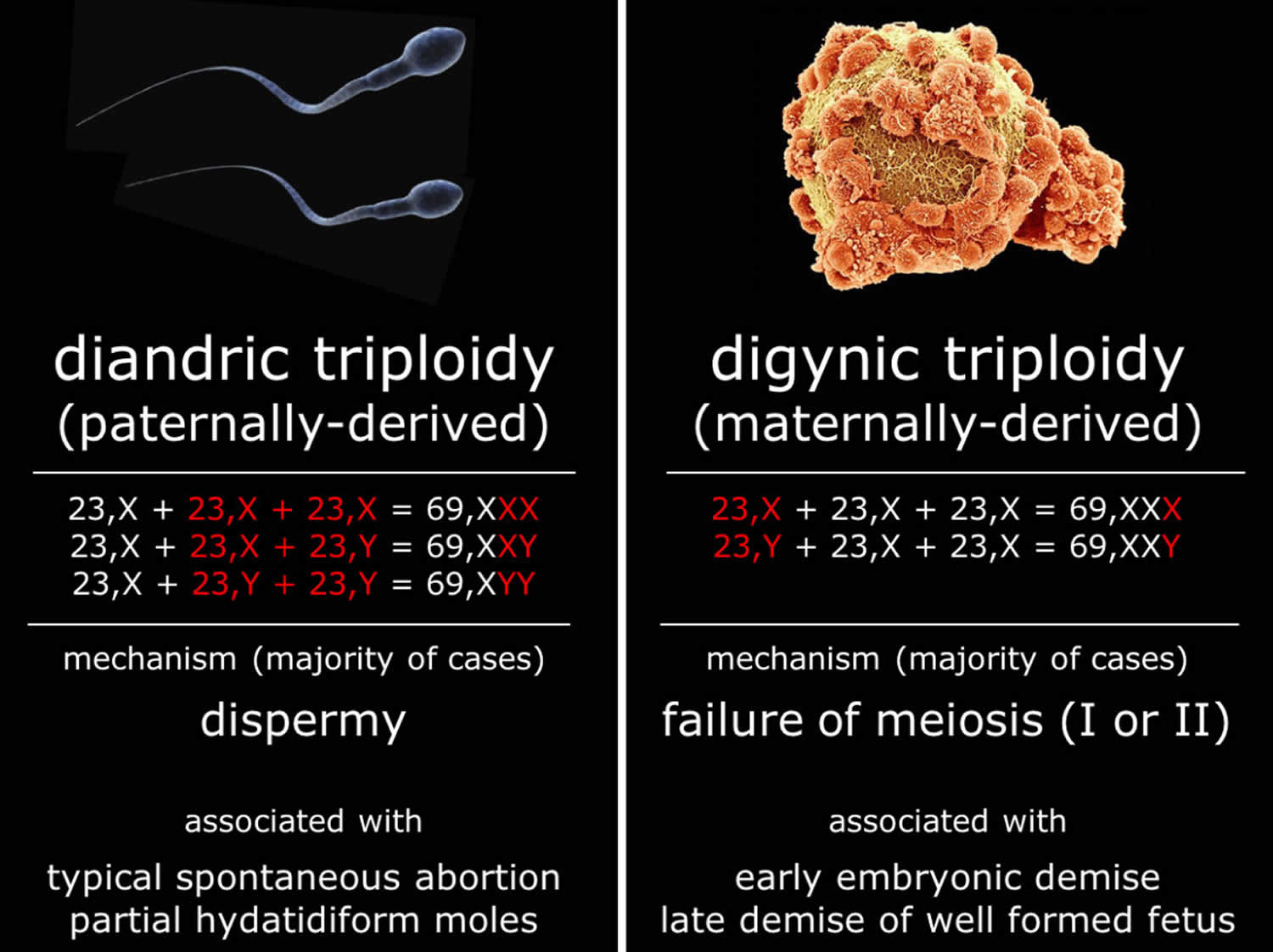
The extra single set of chromosomes may be from the mother (digynic triploidy) or from the father (diandric triploidy). Digynic triploidy may result from the fertilization of a normal oocyte (developing egg in an ovary) produced through an error at either the first or second meiotic division. Diandric triploidy may occur through fertilization of a normal oocyte by a diploid sperm as a result of an error at either the first or second meiotic division or through fertilization of an oocyte by two sperms (dispermy). According to previous reports, 66.4% of triploid infants are due to dispermy (fertilization of an oocyte by two sperms), 23.6% due to a diploid sperm and 10% due to a diploid egg.
Although reports exist regarding multiple recurrences of maternal triploidy (digynic triploidy) pregnancies 19, the overall risk for recurrence has not been established. For paternal triploidy (diandric triploidy) pregnancies, recurrence risk is approximately twice the general population risk 20.
The sex chromosomes in triploid individuals of maternal origin (digynic triploidy) should be in 50% of the cases XXX and 50% XXY, and in those of paternal origin (diandric triploidy), 25% XXX, 50% XXY and 25% XYY 3.
According to previous studies, the frequency of triploids is the following 3, 41, 40, 37:
- 31-49% 69XXX,
- 49-69% 69XXY
- 0,3% 69XYY.
The low frequency of the 69XYY chromosomal aberration suggests that this karyotype leads to low viability and early abortion of the zygote, otherwise the mechanism through which it occurs is very rare 41, 40, 37.
Triploidy does not run in families and is not associated with maternal or paternal age. Triploidy accounts for 2 to 3% of all pregnancies and males represent 51-69 % of all cases 42, 43.
Figure 4. Triploidy causes
[Source 4 ]Triploidy symptoms
Pregnancies with triploidy are usually miscarried early in the pregnancy 38. If the pregnancy continues to term, the infant usually dies within the first days of life. A few affected individuals have been reported to have survived to adulthood, but suffered from developmental delay, learning difficulties, seizures, hearing loss, heart defects, abnormal brain development, adrenal and kidney defects (cystic kidneys), spinal cord malformations (neural tube defects), severe intrauterine growth retardation (IUGR) and abnormal facial features (widely spaced eyes, low nasal bridge, low-set malformed ears, small jaw, absent/small eye, and cleft lip and palate) 44, 45, 46, 13. The third and fourth fingers of the hands and the second and third toes of the feet may be fused (also called syndactyly) and the hands may have unusual single palmar creases (simian creases) 13. There may also be liver and gallbladder defects, twisted intestines and deformities of the fingers and toes. The placenta in triploidy may be immature, large, and filled with cysts. Individuals who are mosaic will survive longer than those with complete triploidy but usually have intellectual disability, developmental delay, depression, seizures, short stature, obesity and other abnormalities. Mosaic triploidy means that some cells have the normal number of 46 chromosomes and other cells have 69 chromosomes per cell.
The pregnant mother carrying a triploidy fetus sometimes experiences increase in blood pressure (hypertension), swelling (edema), and excretion of albumin in the urine (albuminuria). This condition is called toxemia or preeclampsia.
Two different triploidy signs and symptoms are recognized based on whether the additional set of chromosomes is from the mother (digynic triploidy) or from the father (diandric triploidy) in nature 37:
- Type 1 is from the father triploidy (diandric triploidy) and the signs and symptoms are a large cystic placenta with a well-formed fetus and a normal or small (microcephalic) head.
- Type 2 is from the mother triploidy (digynic triploidy) and the signs and symptoms are small non-cystic placenta and a growth-retarded fetus with relative large head (macrocephaly).
In more recent larger studies these triploidy signs and symptoms distinction between maternal and paternal triploidy has been challenged 47. Aside from the features defining type 1 and type 2 triploidy, the literature describes a multitude of other malformations. These include facial dysmorphism, central nervous system abnormalities in 50% of cases, cardiac defects in 31% of cases on second-trimester ultrasound, limb abnormalities and genito-urinary and gastrointestinal defects 48. Type 1 fetuses are reported to be more likely to miscarry early in pregnancy, whereas type 2 cases often survive until later in the pregnancy and are more likely to live beyond birth 49. Subsequent literature suggests that this needs further investigation with larger sample sizes, and that the parent of origin cannot be used to provide a prognosis for the pregnancy or postnatal survival 38.
Triploidy diagnosis
The presence of multiple major malformations, low amniotic fluid and/or growth restriction on fetal ultrasound during pregnancy raises the suspicion of triploidy 13, 50, 51, 52, 53. The diagnosis can be made during pregnancy by chromosome analysis (karyotyping) of cells obtained by amniocentesis or chorionic villus sampling (CVS) 13. The diagnosis can be confirmed after birth by chromosome analysis of tissue (skin) obtained from the affected infant 13.
Triploidy cannot be diagnosed by chromosome microarray testing. The accuracy of non-invasive prenatal testing using cell-free fetal DNA (cfDNA) in the diagnosis of triploidy is still being studied. Abnormal levels of specific maternal blood proteins such as alpha-fetoprotein (AFP), human chorionic gonadotropin (hCG), unconjugated estriol (uE3) and pregnancy-assisted plasma protein-A have been associated with an increased risk for triploidy 13, 54, 8.
Triploidy treatment
Treatment of triploid syndrome is symptomatic and supportive. The most frequent cause of death is pneumonia and general respiratory problems.
Triploidy survival rate
Live birth is exceedingly rare in cases of triploidy. Only 1 in 10,000 fetuses with triploidy will survive to fullterm, with only 1 in 1,200 of triploid fetuses surviving after birth 10. The frequency of triploidy in live births is 1 per 10,000 55 and males represent 51 to 69% of the cases 39.
Despite the fact that some infants with triploidy live longer, their outcome is still extremely poor 5. The literature reports on only a small number of long-term surviving infants 2, which is defined as those surviving more than 45 days 10. Since 1977 there have only been 11 published reports of babies with triploidy living beyond 45 days 2. The longest surviving child was only 10.5 months old and had severe neurological and developmental impairment 5. Because of the poor outcomes, extreme measures to prolong life are not usually considered 5. The management should however be discussed in a multidisciplinary setting and in conjunction with the parents in order to respect their views and decisions.
Infants affected with triploidy usually succumb to respiratory problems or pneumonia. It has been proposed that triploidy survival has improved owing to successful treatment of infections. The association between from the mother triploidy (digynic triploidy) and an increased chance of survival is perhaps now even less clear than previously thought 5.
- Kantor V, Jelsema R, Xu W, DiNonno W, Young K, Demko Z, Benn P. Non-invasive prenatal screening for fetal triploidy using single nucleotide polymorphism-based testing: Differential diagnosis and clinical management in cases showing an extra haplotype. Prenat Diagn. 2022 Jul;42(8):994-999. doi: 10.1002/pd.6169[↩][↩][↩]
- Takabachi, N., Nishimaki, S., Omae, M., Okuda, M., Fujita, S., Ishida, F., Horiguchi, H., Seki, K., Takahashi, T. and Yokota, S. (2008), Long-term survival in a 69,XXX triploid premature infant. Am. J. Med. Genet., 146A: 1618-1621. https://doi.org/10.1002/ajmg.a.32352[↩][↩][↩]
- Kolarski M, Ahmetovic B, Beres M, Topic R, Nikic V, Kavecan I, Sabic S. Genetic Counseling and Prenatal Diagnosis of Triploidy During the Second Trimester of Pregnancy. Med Arch. 2017 Apr;71(2):144-147. doi: 10.5455/medarh.2017.71.144-147[↩][↩][↩][↩]
- Liveborn infant with triploidy 70 , XXXY SWISS SOCIETY OF NEONATOLOGY. https://www.neonet.ch/application/files/9215/6622/2231/COTM_2016_07.pdf[↩][↩]
- Spencer, C E, Mofokeng, B, Turner, A, Nakwa, F, & Krause, A. (2017). Diandric triploidy in a liveborn infant with 3-4 syndactyly and a neural tube defect. South African Journal of Child Health, 11(4), 198-200. https://dx.doi.org/10.7196/sajch.2017.v11i4.1420[↩][↩][↩][↩][↩][↩][↩]
- Chen, H. (2016). Triploidy. In: Atlas of Genetic Diagnosis and Counseling. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-6430-3_232-2[↩]
- Komnenić, M., Petrović, B., Tadić, J., Milićević, S., Stamenković, J., & Ljubić, A. (2016). A LIVEBORN INFANT WITH TRIPLOIDY 69,XXX: CASE REPORT.[↩]
- Jauniaux E, Brown R, Snijders RJ, Noble P, Nicolaides KH. Early prenatal diagnosis of triploidy. Am J Obstet Gynecol. 1997 Mar;176(3):550-4. doi: 10.1016/s0002-9378(97)70546-5[↩][↩]
- Galán F, Orts F, Aguilar MS, Clemente F, Loeda C, Aranda I, Jimenez Cobo B. 69,XXX karyotype in a malformed liveborn female. Maternal origin of triploidy. Ann Genet. 1991;34(1):37-9.[↩]
- Iliopoulos D, Vassiliou G, Sekerli E, Sidiropoulou V, Tsiga A, Dimopoulou D, Voyiatzis N. Long survival in a 69,XXX triploid infant in Greece. Genet Mol Res. 2005 Dec 30;4(4):755-9. https://www.funpecrp.com.br/gmr/year2005/vol4-4/pdf/gmr0174.pdf[↩][↩][↩][↩]
- Szulman AE, Philippe E, Boue JG, Boue A. Human triploidy: association with partial hydatidiform moles and nonmolar conceptuses. Hum Pathol. 1981;12(11):1016‐1021. 10.1016/s0046-8177(81)80259-6[↩]
- McFadden DE, Kalousek DK. Two different phenotypes of fetuses with chromosomal triploidy: correlation with parental origin of the extra haploid set. J Med Genet. 1991;38(4):535‐538. 10.1002/ajmg.1320380407[↩]
- Triploidy. https://rarediseases.org/rare-diseases/triploidy[↩][↩][↩][↩][↩][↩][↩][↩]
- Sehnert AJ, Rava RP, Bianchi DW. A new era in noninvasive prenatal testing. N Engl J Med. 2013 Nov 28;369(22):2164-5. doi: 10.1056/NEJMc1311604[↩]
- Norton ME, Rose NC, Benn P. Noninvasive prenatal testing for fetal aneuploidy: clinical assessment and a plea for restraint. Obstet Gynecol. 2013 Apr;121(4):847-850. doi: 10.1097/AOG.0b013e31828642c6[↩]
- Wertelecki W, Graham JM Jr, Sergovich FR. The clinical syndrome of triploidy. Obstet Gynecol. 1976 Jan;47(1):69-76.[↩]
- Huang T, Alberman E, Wald N, Summers AM. Triploidy identified through second‐trimester serum screening. Prenat Diagn. 2005;25(3):229‐233. 10.1002/pd.1115[↩]
- van de Laar I, Rabelink G, Hochstenbach R, Tuerlings J, Hoogeboom J, Giltay J. Diploid/triploid mosaicism in dysmorphic patients. Clin Genet. 2002;62(5):376‐382. 10.1034/j.1399-0004.2002.620504.x[↩]
- Filges I, Manokhina I, Penaherrera MS, et al. Recurrent triploidy due to a failure to complete maternal meiosis II: whole‐exome sequencing reveals candidate variants. Molec Hum Reprod. 2015;21(4):339‐346. 10.1093/molehr/gau112[↩][↩]
- Eagles N, Sebire NJ, Short D, Savage P, Seckl M, Fisher R. Risk of recurrent molar pregnancies following complete and partial hydatidiform moles. Hum Reprod. 2015;30(9):2055‐2063. 10.1093/humrep/dev169[↩][↩]
- American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 545: Noninvasive prenatal testing for fetal aneuploidy. Obstet Gynecol. 2012 Dec;120(6):1532-4. doi: 10.1097/01.AOG.0000423819.85283.f4[↩]
- Larion S, Warsof SL, Romary L, Mlynarczyk M, Peleg D, Abuhamad AZ. Association of combined first-trimester screen and noninvasive prenatal testing on diagnostic procedures. Obstet Gynecol. 2014 Jun;123(6):1303-1310. doi: 10.1097/AOG.0000000000000275[↩]
- Benacerraf BR, Gelman R, Frigoletto FD Jr. Sonographic identification of second-trimester fetuses with Down’s syndrome. N Engl J Med. 1987 Nov 26;317(22):1371-6. doi: 10.1056/NEJM198711263172203[↩]
- Ngan HYS, Seckl MJ, Berkowitz RS, et al. Update on the diagnosis and management of gestational trophoblastic disease. Int J Gynaecol Obstet. 2018;143(Suppl 2):79‐85. 10.1002/ijgo.12615[↩]
- Horowitz H, Eskander RN, Adelman MR, Burke W. Epidemiology, diagnosis, and treatment of gestational trophoblastic disease: a Society of Gynecologic Oncology evidenced‐based review and recommendation. Gynecol Oncol. 2021;163(3):605‐613. 10.1016/j.ygyno.2021.10.003[↩]
- Zalel Y, Shapiro I, Weissmann‐Brenner A, Berkenstadt M, Leibovitz Z, Bronshtein M. Prenatal sonographic features of triploidy at 12‐16 weeks. Prenat Diagn. 2016;36(7):650‐655. 10.1002/pd.4834[↩]
- Spencer K, Liao AW, Skentou H, et al. Screening for triploidy by fetal nuchal translucency and maternal serum free beta‐hCG and PAPP‐A at 10‐14 weeks of gestation. Prenat Diagn. 2000;20(6):495‐499. 10.1002/1097-0223(200006)20:6<495::aid-pd846>3.0.co;2-u[↩]
- Benn PA, Gainey A, Ingardia CJ, et al. Second trimester maternal serum analytes in triploid pregnancies: correlation with phenotype and sex chromosome complement. Prenat Diagn. 2001;21(8):680‐686. 10.1002/pd.139[↩]
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Obstetrics; Committee on Genetics; Society for Maternal‐Fetal Medicine . Screening for fetal chromosomal abnormalities: ACOG Practice Bulletin, number 226. Obstet Gynecol. 2020;136(4):e48‐e69. 10.1097/aog.0000000000004084[↩]
- Curnow KJ, Wilkins‐Haug L, Ryan A, et al. Detection of triploid, molar, and vanishing twin pregnancies by a single‐nucleotide polymorphism‐based noninvasive prenatal test. A J Obstet Gynecol. 2015;212(1):79.e71‐79. 10.1016/j.ajog.2014.10.012[↩]
- Benn P, Rebarber A. Non‐invasive prenatal testing in the management of twin pregnancies. Prenat Diagn. 2021;41(10):1233‐1240. 10.1002/pd.5989[↩]
- Karyotype. https://www.genome.gov/genetics-glossary/Karyotype[↩][↩][↩]
- Chuang TY, Chang SY, Chen CP, Lin MH, Chen CY, Chen SW, Chern SR, Lee CC, Town DD, Wang W. Digynic triploidy in a fetus presenting with semilobar holoprosencephaly. Taiwan J Obstet Gynecol. 2018 Dec;57(6):881-884. https://doi.org/10.1016/j.tjog.2018.11.001[↩][↩]
- Meiosis. https://www.genome.gov/genetics-glossary/Meiosis[↩][↩]
- Brancati F, Mingarelli R, Dallapiccola B. Recurrent triploidy of maternal origin. Eur J Hum Genet. 2003 Dec;11(12):972-4. doi: 10.1038/sj.ejhg.5201076[↩]
- O’Neill GT, Kaufman MH. Ovulation and fertilization of primary and secondary oocytes in LT/Sv strain mice. Gamete Res. 1987 Sep;18(1):27-36. doi: 10.1002/mrd.1120180105[↩]
- McFadden DE, Kalousek DK. Two different phenotypes of fetuses with chromosomal triploidy: correlation with parental origin of the extra haploid set. Am J Med Genet. 1991 Mar 15;38(4):535-8. doi: 10.1002/ajmg.1320380407[↩][↩][↩][↩]
- Baumer A, Balmer D, Binkert F, Schinzel A. Parental origin and mechanisms of formation of triploidy: a study of 25 cases. Eur J Hum Genet. 2000 Dec;8(12):911-7. doi: 10.1038/sj.ejhg.5200572[↩][↩][↩]
- McFadden DE, Langlois S. Parental and meiotic origin of triploidy in the embryonic and fetal periods. Clin Genet. 2000 Sep;58(3):192-200. doi: 10.1034/j.1399-0004.2000.580306.x[↩][↩]
- Zaragoza MV, Surti U, Redline RW, Millie E, Chakravarti A, Hassold TJ. Parental origin and phenotype of triploidy in spontaneous abortions: predominance of diandry and association with the partial hydatidiform mole. Am J Hum Genet. 2000 Jun;66(6):1807-20. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1378061/pdf/10801385.pdf[↩][↩][↩]
- McFadden DE, Robinson WP. Phenotype of triploid embryos. J Med Genet. 2006 Jul;43(7):609-12. doi: 10.1136/jmg.2005.037747[↩][↩]
- Gardner RJM, Sutherland GR, Schaffer LG. Chromosome Abnormalities and Genetic Counseling. 4th ed. >New York, NY: Oxford University Press; 2012.[↩]
- Wick JB, Johnson KJ, O’Brien J, Wick MJ. Second-trimester diagnosis of triploidy: a series of four cases. AJP Rep. 2013 May;3(1):37-40. doi: 10.1055/s-0032-1331378[↩]
- Khong TY, George K. Chromosomal abnormalities associated with a single umbilical artery. Prenat Diagn. 1992 Nov;12(11):965-8. doi: 10.1002/pd.1970121118[↩]
- Hassold T, Chen N, Funkhouser J, Jooss T, Manuel B, Matsuura J, Matsuyama A, Wilson C, Yamane JA, Jacobs PA. A cytogenetic study of 1000 spontaneous abortions. Ann Hum Genet. 1980 Oct;44(2):151-78. doi: 10.1111/j.1469-1809.1980.tb00955.x[↩]
- Doshi N, Surti U, Szulman AE. Morphologic anomalies in triploid liveborn fetuses. Hum Pathol. 1983 Aug;14(8):716-23. doi: 10.1016/s0046-8177(83)80145-2[↩]
- Joergensen MW, Niemann I, Rasmussen AA, Hindkjaer J, Agerholm I, Bolund L, Sunde L. Triploid pregnancies: genetic and clinical features of 158 cases. Am J Obstet Gynecol. 2014 Oct;211(4):370.e1-19. doi: 10.1016/j.ajog.2014.03.039[↩]
- Chen CP, Chern SR, Tsai FJ, Hsu CY, Ko K, Wang W. Prenatal diagnosis and molecular analysis of triploidy in a fetus with intrauterine growth restriction, relative macrocephaly and holoprosencephaly. Taiwan J Obstet Gynecol. 2009 Sep;48(3):323-6. https://doi.org/10.1016/S1028-4559(09)60318-1[↩]
- Hasegawa T, Harada N, Ikeda K, Ishii T, Hokuto I, Kasai K, Tanaka M, Fukuzawa R, Niikawa N, Matsuo N. Digynic triploid infant surviving for 46 days. Am J Med Genet. 1999 Dec 3;87(4):306-10. doi: 10.1002/(sici)1096-8628(19991203)87:4<306::aid-ajmg5>3.0.co;2-6[↩]
- Chen CP, Chen YY, Chern SR, Kuo YL, Lee CC, Wang W. First-trimester sonographic demonstration of digynic triploidy. Taiwan J Obstet Gynecol. 2013 Dec;52(4):613-5. https://doi.org/10.1016/j.tjog.2013.10.033[↩]
- Agathokleous M, Chaveeva P, Poon LC, Kosinski P, Nicolaides KH. Meta-analysis of second-trimester markers for trisomy 21. Ultrasound Obstet Gynecol. 2013 Mar;41(3):247-61. doi: 10.1002/uog.12364[↩]
- Nicolaides KH. Screening for chromosomal defects. Ultrasound Obstet Gynecol. 2003 Apr;21(4):313-21. doi: 10.1002/uog.128[↩]
- Philipp T, Grillenberger K, Separovic ER, Philipp K, Kalousek DK. Effects of triploidy on early human development. Prenat Diagn. 2004 Apr;24(4):276-81. doi: 10.1002/pd.789[↩]
- Nicolaides KH, Wright D, Poon LC, Syngelaki A, Gil MM. First-trimester contingent screening for trisomy 21 by biomarkers and maternal blood cell-free DNA testing. Ultrasound Obstet Gynecol. 2013 Jul;42(1):41-50. doi: 10.1002/uog.12511[↩]
- Jacobs PA, Melville M, Ratcliffe S, Keay AJ, Syme J. A cytogenetic survey of 11,680 newborn infants. Ann Hum Genet. 1974 May;37(4):359-76. doi: 10.1111/j.1469-1809.1974.tb01843.x[↩]