Contents
Glanders
Glanders, also called quinia, malleus, droes or farcy, is a highly contagious and often fatal infectious disease that can pass from animals to humans (zoonotic disease) that is caused by Burkholderia mallei bacteria 1, 2, 3, 4, 5, 6, 7, 8, 9, 10. While humans can get glanders disease, glanders is primarily an infectious disease affecting horses restricted to parts of Africa, Asia, the Middle East, and Central and South America 11, 3. Glanders also affects donkeys and mules and can be naturally contracted by other mammals such as goats, dogs, and cats 12.
Glanders disease is closely monitored by the World Organization of Animal Health as it is considered a biosecurity and biosafety threat 13, 12, 14, 15, 16. The Burkholderia mallei bacteria is known to have been weaponized during both modern and ancient conflicts, most likely because of its potentially devastating effect on cavalry horses and pack animals 17, 18, 5. In fact, Burkholderia mallei (previously known as Pseudomonas mallei) was one of the first biological warfare agents used in the 20th century, specifically during World War 1 19, 20.
Glanders control programs have successfully eradicated the disease from North America, Western Europe, Japan and Australia 21.
Glanders continues to be extremely rare in humans. People who care for affected animals or handle infected specimens face an increased risk of getting glanders. Such individuals can include:
- Veterinarians
- Horse Caretakers e.g., hoof care workers (farriers), hide workers (flayers), transport workers, horse fanciers, and stable hands
- Laboratory workers
- Equine Butchers
- Abattoir Workers
In 2000, one case occurred in a research laboratory worker in the U.S. after accidental exposure 10.
In humans, glanders usually is acquired through direct skin or mucous membrane contact with infected animal tissues 10. The incubation period usually is 1 to 14 days 10.
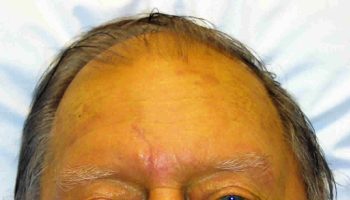
Glanders signs and symptms vary depending on the type of infection you have 22, 23: skin inoculation can result in localized Glanders disease with nodule formation and swollen inflamed lymph nodes (lymphandenitis) 22. Infections involving the mucous membranes (a membrane that lines the insides of organs and cavities throughout your body) in your eyes, nose, and respiratory tract will cause increased mucus production from the affected sites. Spreading to other locations in the body may occur 1-4 weeks after infection.
Glanders often manifests itself as lung infection. In lung infections, pneumonia (bronchopneumonia or lobar pneumonia), pulmonary abscesses, and pleural effusion can occur 22. Chest X-rays will show localized infection in the lobes of the lungs. Without treatment, glanders bloodstream infections (bacteremia) are usually fatal within 7 to 10 days.
The chronic form of glanders involves multiple abscesses (collection of pus) within the muscles and skin of the arms and legs or in the lungs, spleen, and/or liver 24.
Since human cases of glanders are extremely rare, there is limited information about antibiotic treatment in humans 25. Sulfadiazine has been found to be effective in experimental animals and in humans 25. In addition, the Burkholderia mallei bacteria that causes glanders is usually sensitive to 25:
- Tetracyclines
- Ciprofloxacin
- Streptomycin
- Novobiocin
- Gentamicin
- Imipenem
- Ceftazidime
- Sulfonamides
Sulfadiazine (25 mg/kg intravenous, four times a day) was efficacious in some cases 24. In mice, doxycycline and ciprofloxacin have been effective therapies 26.
The mortality of apparent infection was approximately 95% before the use of antimicrobial agents; however, except when glanders bloodstream infection (bacteremia) develops, better diagnosis and more appropriate therapy have lowered mortality 23.
At the present time there is no vaccine against glanders disease (Burkholderia mallei infection) is available.
Figure 1. Glanders disease in humans
Figure 2. Glanders disease time from exposure to symptoms
[Source 5 ]Glanders cause
Glanders is caused by a gram-negative aerobic nonmotile bacillus Burkholderia mallei (previously known as Pseudomonas mallei) that is transmitted to humans through contact with tissues or body fluids of infected animals 27, 28, 29, 30, 31, 32, 33. The Burkholderia mallei bacteria enter your body through cuts or abrasions in the skin and through mucosal surfaces such as the eyes and nose. The Burkholderia mallei bacteriamay also be inhaled via infected aerosols or dust contaminated by infected animals. Burkholderia mallei is an obligate mammalian pathogen and must cause the disease to be transmitted between hosts. Sporadic cases have been documented in veterinarians, horse caretakers, and laboratorians.
According to the Centers for Disease Control and Prevention (CDC), cases of human-to-human transmission have not been reported in the U.S. 34.
Glanders pathophysiology
Burkholderia mallei bacteria can enter into cells and spread from cell to cell by polymerizing actin; this can lead to the fusion of cells and the formation of multinucleated giant cells 1. Both are resistant to a large number of antibiotics because of their ability to pump them out of the cell, and both possess type VI secretion systems 1. Burkholderia mallei cause damage to and death of endothelial cells lining blood vessels, which increases the likelihood of blood clots formation 1.
Glanders prevention
Presently, there is no vaccine available for glanders. In countries where glanders is endemic in animals, prevention of the disease in humans involves identification and elimination of the infection in the animal population.
Within the health care setting, transmission can be prevented by using standard and airborne precautions such as the use of disposable surgical masks, face shields, and gowns, when appropriate, to prevent splashing of mucous membranes and skin are sufficient to prevent transmission of this disease to those caring for patients, and biosafety level three is recommended for laboratory staff handling Burkholderia mallei 35.
Glanders symptoms
The incubation period of glanders is usually 1 to 21 days but can be months or years 36. In humans, the first symptom of glanders is usually fever, followed by pneumonia, pustules, and abscesses. The acute form of the disease usually is fatal within 7 to 10 days of onset. Chronic glanders does occur, which can cause death within months; survivors remain carriers of disease.
Glanders symptoms commonly include:
- Fever with chills and sweating
- Muscle aches
- Chest pain
- Muscle tightness
- Headache
- Nasal discharge
- Light sensitivity (sometimes with excessive tearing of the eyes)
The particular symptoms experienced, however, will vary depending on the type of infection. The four types of glanders disease, along with the symptoms associated with each, are listed below.
Localized Glanders disease
If there is a cut or scratch in the skin, a localized infection with ulceration may develop within 1 to 5 days at the site where the bacteria entered the body. Swollen inflamed lymph nodes (lymphandenitis) may also be apparent.
Infections involving the mucous membranes (a membrane that lines the insides of organs and cavities throughout your body) in your eyes, nose, and respiratory tract will cause increased mucus production from the affected sites. Spreading to other locations in the body may occur 1-4 weeks after infection.
Lung infection
Glanders often manifests itself as lung infection. In lung infections, pneumonia, pulmonary abscesses, and pleural effusion can occur. Chest X-rays will show localized infection in the lobes of the lungs. Patients with significant lung involvement may progress to respiratory failure, requiring mechanical ventilation. Sepsis and bleeding disorder also may occur.
The following first two criteria and at least one of last two criteria are proposed for diagnostic confirmation of acute glanders lung infection in humans:
- Development of constitutional symptoms such as fever, rigors, myalgias, fatigue, headache, severe malaise, and pleuritic chest pain 37.
- Chest X-ray exhibiting new onset of infiltrates either segmental or lobar which may appear as nodular opacities. May also exhibit cavitary lesions or abscesses not only in the lungs but virtually any remote sites such as spleen, liver, subcutaneous tissue and muscle.
- Positive isolation of Burkholderia mallei bacteria from sites such as cavitary lesions or abscesses not only in the lungs but virtually any remote site such as spleen, liver, subcutaneous tissue, and muscle as well as secretions, sputum, blood, and urine 38.
- Positive Burkholderia species non-specific indirect hemmagglutination assay titer and if positive a follow-up four-fold titer increase 38, 39.
Bloodstream infection
Without treatment, glanders bloodstream infections are usually fatal within 7 to 10 days.
Chronic Glanders disease
The chronic form of glanders involves multiple abscesses within the muscles and skin of the arms and legs or in the lungs, spleen, and/or liver.
Glanders complications
Glanders complications may include:
- Septic shock
- Abscesses of the liver, spleen, and prostate
- Death
Glanders diagnosis
Glanders is an internationally reportable disease 21. Glanders disease is diagnosed in the laboratory by isolating Burkholderia mallei from blood, sputum, urine, or skin lesions. Blood cultures are rarely positive in cases of Burkholderia mallei.
Serologic assays are also available. Serologic, antigen-based tests include the complement fixation test, Western blot and enzyme-linked immunosorbent assay (ELISA). The complement fixation test is the official test for international trade of horses as outlined by the Office International des Epizooties (OIE) and is utilized by the United States Department of Agriculture’s Animal and Plant Health Inspection Service (APHIS). It is highly specific and can detect horses in early stages of infection, as well as chronic cases. However, in some cases false positive results have been reported. Supplementary testing of horses with positive results from the complement fixation test is performed with an ELISA test or Western blot to confirm the diagnosis.
Horses imported to the United States must test negative for glanders prior to release from import quarantine 21.
If a case of Glanders disease in horses is suspected in the United States, a state or federal animal health official must be notified immediately 21. To stop Glanders from becoming established and to avoid animal to human transfer, affected animals are returned to the country of origin or are euthanized 21. Animals that have been exposed to the infected individual are quarantined for testing 21.
Glanders treatment
Since human cases of glanders are extremely rare, there is limited information about antibiotic treatment in humans 25. Sulfadiazine has been found to be effective in experimental animals and in humans 25. In addition, the Burkholderia mallei bacteria that causes glanders is usually sensitive to 25, 40, 41, 42, 43:
- Tetracyclines
- Ciprofloxacin
- Streptomycin
- Novobiocin
- Gentamicin
- Imipenem
- Ceftazidime
- Sulfonamides
Given the lack of historical data, current treatment regimens for human glanders are not well understood at this point in time, and no FDA approved treatment or treatment regimen exists.
Of the human glanders cases recorded since the 1940s, several have been treated successfully with antibiotics 20, 44, 45, 46, 37. Sulfadiazine was used to successfully treat six of the previously discussed laboratory-acquired infections at Fort Detrick, as well as two additional cases of non-laboratory acquired glanders described in 1951 37, 46. The seventh case was successfully treated with a tetracycline derivative, aureomycin. In the most recent case of laboratory-acquired glanders, the patient initially received a 10-day course of a first-generation cephalosporin but symptoms persisted for a few weeks. The patient then received a 10-day course of clarithromycin, but symptoms returned 4 days after treatment was stopped. The patient was then successfully treated with imipenem and doxycycline intravenously for 1 month followed by oral azithromycin and doxycycline for 6 months 20, 44. A CT scan after this therapy showed improvement of spleen and liver abscesses.
An exhaustive review of current and experimental treatments for both glanders and melioidosis has been published 47. However, in order to manage laboratory exposures, treatment recommendations have been made 38. Symptomatic case treatment of glanders requires a concurrent two pronged approach consisting of both an intensive intravenous (IV) therapy and oral eradication therapy. IV medication options are imipenem, meropenem or ceftazidime with or without trimethoprim-sulfamethoxazole (TMP-SMX). IV dosing schedule should last a minimum of 10 days and may be longer depending on the severity of illness. Oral therapy timing is also dependent on the severity of the disease and may run 12 weeks to as long as 12 months in duration. The treatment of choice for oral antibiotic therapy options are trimethoprim-sulfamethoxazole as noted above with or without a secondary oral medication of doxycycline. Alternate therapies for trimethoprim-sulfamethoxazole resistant B. mallei or patient intolerance consist of doxycycline, macrolides, chloramphenicol, quinolones, or amoxicillin-clavulanate as noted in the Occupational Health Manual from the United States Army Medical Research Institute of Infectious Diseases (USAMRIID) 38. Since B. mallei and B. pseudomallei are similar, it is theorized that drug therapy experiences should be similar as well. Therefore, doxycycline, amoxicillin-clavulante and quinolones monotherapies are at a greater risk of relapse than trimethoprim-sulfamethoxazole and chloramphenicol 5.
Glanders prognosis
While there is no national or state surveillance for glanders disease in humans, the mortality rate for glanders lung infection has been reported to be 90-95% without treatment and up to 40% with treatment 5, 1, 48. The reported mortality rate for the septicemic form of glanders is greater than 95% without treatment with death occurs within 7 to 10 days of onset and as high as 50% with treatment 5, 1, 48. The mortality rate for the untreated skin form of glanders is 90-95% if it becomes systemic (bloodborne) and as high as 50% with treatment 5. In the case of chronic glanders, the mortality rate may be as high as 50% despite treatment 49, 1, 48. However, sensitivity data suggest newer antibiotics should be more efficacious. The latest estimates show the mortality rate for localized Glanders disease can be as low as 20% with appropriate treatment, and the overall mortality rate is 40% 48.
In non-endemic areas, like the U.S., horses that test positive for glanders are usually euthanized or must be removed from the country 21. Acute and chronic forms of glanders are usually ultimately fatal 21. Acute cases typically succumb within a short period, whereas chronically infected animals can survive for years in some cases 21.
- Nguyen HVN, Smith ME, Hayoun MA. Glanders and Melioidosis. [Updated 2023 Aug 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK448110[↩][↩][↩][↩][↩][↩][↩]
- Kinoshita, Y., Cloutier, A.K., Rozak, D.A. et al. A novel selective medium for the isolation of Burkholderia mallei from equine specimens. BMC Vet Res 15, 133 (2019). https://doi.org/10.1186/s12917-019-1874-0[↩]
- Khan I, Wieler LH, Melzer F, Elschner MC, Muhammad G, Ali S, Sprague LD, Neubauer H, Saqib M. Glanders in animals: a review on epidemiology, clinical presentation, diagnosis and countermeasures. Transbound Emerg Dis. 2013;60(3):204–21.[↩][↩]
- Whitlock GC, Estes DM, Torres AG. Glanders: off to the races with Burkholderia mallei. FEMS Microbiol Lett. 2007 Dec;277(2):115-22. doi: 10.1111/j.1574-6968.2007.00949.x[↩]
- Van Zandt KE, Greer MT, Gelhaus HC. Glanders: an overview of infection in humans. Orphanet J Rare Dis. 2013 Sep 3;8:131. doi: 10.1186/1750-1172-8-131[↩][↩][↩][↩][↩][↩][↩]
- Howe, C. 1950. Glanders, p. 185-202. In H. A. Christian (ed.), The Oxford textbook of medicine. Oxford University Press, New York, N.Y.[↩]
- Jennings, W. E. 1963. Glanders, p. 264-292. In T. G. Hull (ed.), Diseases transmitted from animals to man. Charles C Thomas, Publisher, Springfield, Ill.[↩]
- Alibek K, Handelman S. Biohazard: the chilling true story of the largest covert biological weapons program in the world. New York, N.Y: Random House; 1999.[↩]
- Wheelis, M. First shots fired in biological warfare. Nature 395, 213 (1998). https://doi.org/10.1038/26089[↩]
- Centers for Disease Control and Prevention (CDC). Laboratory-acquired human glanders–Maryland, May 2000. MMWR Morb Mortal Wkly Rep. 2000 Jun 23;49(24):532-5. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm4924a3.htm[↩][↩][↩][↩]
- Elschner MC, Neubauer H, Sprague LD. The resurrection of Glanders in a new epidemiological scenario: a beneficiary of “global change”. Curr Clin Microbiol Rep. 2017;4(1):54–60.[↩]
- Glanders. https://www.cdc.gov/glanders/exposure/index.html[↩][↩]
- Godoy D, Randle G, Simpson AJ, Aanensen DM, Pitt TL, Kinoshita R, Spratt BG. Multilocus sequence typing and evolutionary relationships among the causative agents of melioidosis and glanders, Burkholderia pseudomallei and Burkholderia mallei. J Clin Microbiol. 2003 May;41(5):2068-79. doi: 10.1128/JCM.41.5.2068-2079.2003. Erratum in: J Clin Microbiol. 2003 Oct;41(10):4913.[↩]
- Carr-Gregory B, Waag DM. Glanders. In: Dembek ZF, editor. Medical aspects of biological warfare. Borden Institute, Office of the Surgeon General, AMEDD Center and School, Texas, US; 2007. p. 121–146.[↩]
- Khan I, Wieler LH, Melzer F, Elschner MC, Muhammad G, Ali S, Sprague LD, Neubauer H, Saqib M. Glanders in animals: a review on epidemiology, clinical presentation, diagnosis and countermeasures. Transbound Emerg Dis. 2013 Jun;60(3):204-21. doi: 10.1111/j.1865-1682.2012.01342.x[↩]
- Kettle AN, Wernery U. Glanders and the risk for its introduction through the international movement of horses. Equine Vet J. 2016 Sep;48(5):654-8. doi: 10.1111/evj.12599[↩]
- Dance DAB. Melioidosis and glanders as possible biological weapons. In: Fong W, Alibek K, editors. Bioterrorism and infectious agents A new dilemma for the 21st century. Springer Science and Business Media; New York: 2005. pp. 99–145.[↩]
- Cheng AC, Currie BJ. Melioidosis: epidemiology, pathophysiology, and management. Clin Microbiol Rev. 2005 Apr;18(2):383-416. doi: 10.1128/CMR.18.2.383-416.2005. Erratum in: Clin Microbiol Rev. 2007 Jul;20(3):533. Dosage error in article text.[↩]
- Wheelis M. First shots fired in biological warfare. Nature. 1998 Sep 17;395(6699):213. doi: 10.1038/26089[↩]
- Gregory BC, Waag DM. In: Medical aspects of biological warfare. Dembek ZF, editor. Washington, DC: Borden Institute; 2007. Glanders; pp. 121–146.[↩][↩][↩]
- Glanders. https://ceh.vetmed.ucdavis.edu/health-topics/glanders[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Bartlett JG. Glanders. In: Gorbach SL, Bartlett JG, Blacklow NR, eds. Infectious diseases. 2nd ed. Philadelphia, Pennsylvania: WB Saunders Company, 1998:1578–80.[↩][↩][↩]
- Mandell GL, Bennett JE, Dolin R. Pseudomonas species (including melioidosis and glanders). In: Mandell GL, Bennett JE, Dolin R, eds. Principles and practices of infectious diseases. 4th ed. New York: Churchill Livingstone, 1995:2006–7.[↩][↩]
- Howe C, Miller WR. Human glanders: report of six cases. Ann Intern Med 1947;26:93–115. doi: 10.7326/0003-4819-26-1-93[↩][↩]
- Glanders. https://www.cdc.gov/glanders/treatment/index.html[↩][↩][↩][↩][↩][↩]
- Russell P, Eley SM, Ellis J, Green M, Bell DL, Kenny DJ, Titball RW. Comparison of efficacy of ciprofloxacin and doxycycline against experimental melioidosis and glanders. J Antimicrob Chemother. 2000 Jun;45(6):813-8. doi: 10.1093/jac/45.6.813[↩]
- Singha H, Elschner MC, Malik P, Saini S, Tripathi BN, Mertens-Scholz K, Brangsch H, Melzer F, Singh RK, Neubauer H. Molecular Typing of Burkholderia mallei Isolates from Equids with Glanders, India. Emerg Infect Dis. 2021 Jun;27(6):1745-1748. doi: 10.3201/eid2706.203232[↩]
- Jennings WE. In: Diseases transmitted from animals to man. Hull TG, editor. Springfield, Ill: Charles C. Thomas Publisher; 1963. Glanders; pp. 264–292.[↩]
- Bozue JA, Chaudhury S, Amemiya K, Chua J, Cote CK, Toothman RG, Dankmeyer JL, Klimko CP, Wilhelmsen CL, Raymond JW, Zavaljevski N, Reifman J, Wallqvist A. Phenotypic Characterization of a Novel Virulence-Factor Deletion Strain of Burkholderia mallei That Provides Partial Protection against Inhalational Glanders in Mice. Front Cell Infect Microbiol. 2016 Feb 26;6:21. doi: 10.3389/fcimb.2016.00021[↩]
- Wernery U, Chan E, Raghavan R, Teng JLL, Syriac G, Siu SY, Joseph M, Yeung ML, Jia L, Cai JP, Chiu TH, Lau SKP, Woo PCY. Development of a sensitive competitive enzyme-linked immunosorbent assay for serodiagnosis of Burkholderia mallei, a Tier 1 select agent. PLoS Negl Trop Dis. 2021 Dec 21;15(12):e0010007. doi: 10.1371/journal.pntd.0010007[↩]
- Grund ME, Choi SJ, McNitt DH, Barbier M, Hu G, LaSala PR, Cote CK, Berisio R, Lukomski S. Burkholderia collagen-like protein 8, Bucl8, is a unique outer membrane component of a putative tetrapartite efflux pump in Burkholderia pseudomallei and Burkholderia mallei. PLoS One. 2020 Nov 23;15(11):e0242593. doi: 10.1371/journal.pone.0242593[↩]
- Dyke JS, Huertas-Diaz MC, Michel F, Holladay NE, Hogan RJ, He B, Lafontaine ER. The Peptidoglycan-associated lipoprotein Pal contributes to the virulence of Burkholderia mallei and provides protection against lethal aerosol challenge. Virulence. 2020 Dec;11(1):1024-1040. doi: 10.1080/21505594.2020.1804275[↩]
- Wilkinson L. Glanders: medicine and veterinary medicine in common pursuit of a contagious disease. Med Hist. 1981 Oct;25(4):363-84. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1139069/pdf/medhist00089-0026.pdf[↩]
- Glanders. https://www.cdc.gov/glanders/transmission/index.html[↩]
- Biosafety in Microbiological and Biomedical Laboratories (BMBL) 6th Edition. https://www.cdc.gov/labs/pdf/SF__19_308133-A_BMBL6_00-BOOK-WEB-final-3.pdf[↩]
- Darling RG, Woods JB. US Army Medical Research Institute of Infectious Diseases Medical Management of Biological Casualties Handbook. 5. Fort Detrick, MD: USAMRIID; 2004. Glanders and Melioidosis; pp. 32–39.[↩]
- CALDERON HOWE, WINSTON R. MILLER. HUMAN GLANDERS: REPORT OF SIX CASES. Ann Intern Med.1947;26:93-115. doi:10.7326/0003-4819-26-1-93. https://doi.org/10.7326/0003-4819-26-1-93[↩][↩][↩]
- Rusnak J. Occupational health manual for laboratory exposures to select (BSL-3 & BSL-4) and other biological agents. 2011. pp. 3–51.[↩][↩][↩][↩]
- Peacock SJ. et al. Management of accidental laboratory exposure to Burkholderia pseudomallei and B. mallei. Emerg Infect Dis. 2008;14(7):e2. doi: 10.3201/eid1407.071501[↩]
- Lipsitz R, Garges S, Aurigemma R, Baccam P, Blaney DD, Cheng AC, Currie BJ, Dance D, Gee JE, Larsen J, Limmathurotsakul D, Morrow MG, Norton R, O’Mara E, Peacock SJ, Pesik N, Rogers LP, Schweizer HP, Steinmetz I, Tan G, Tan P, Wiersinga WJ, Wuthiekanun V, Smith TL. Workshop on treatment of and postexposure prophylaxis for Burkholderia pseudomallei and B. mallei Infection, 2010. Emerg Infect Dis. 2012 Dec;18(12):e2. doi: 10.3201/eid1812.120638[↩]
- Rhodes KA, Schweizer HP. Antibiotic resistance in Burkholderia species. Drug Resist Updat. 2016 Sep;28:82-90. doi: 10.1016/j.drup.2016.07.003[↩]
- Heine HS. et al. In vitro antibiotic susceptibilities of Burkholderia mallei (causative agent of glanders) determined by broth microdilution and E-test. Antimicrob Agents Chemother. 2001;45(7):2119–2121. doi: 10.1128/AAC.45.7.2119-2121.2001[↩]
- Thibault FM. et al. Antibiotic susceptibility of 65 isolates of Burkholderia pseudomallei and Burkholderia mallei to 35 antimicrobial agents. J Antimicrob Chemother. 2004;54(6):1134–1138. doi: 10.1093/jac/dkh471[↩]
- Srinivasan A. et al. Glanders in a military research microbiologist. N Engl J Med. 2001;345(4):256–258. doi: 10.1056/NEJM200107263450404[↩][↩]
- Womack CR, Wells EB. Co-existent chronic glanders and multiple cystic osseous tuberculosis treated with streptomycin. Am J Med. 1949;6(2):267–71. doi: 10.1016/0002-9343(49)90022-4[↩]
- ANSARI M, MINOU M. Deux cas de morve chronique humaine traités par les sulfamides [Two cases of chronic human glanders treated with sulfonamides]. Ann Inst Pasteur (Paris). 1951 Jul;81(1):98-102. Undetermined Language.[↩][↩]
- Estes DM. et al. Present and future therapeutic strategies for melioidosis and glanders. Expert Rev Anti Infect Ther. 2010;8(3):325–338. doi: 10.1586/eri.10.4[↩]
- Glanders. https://www.cdc.gov/glanders/health-care-worker/index.html[↩][↩][↩][↩]
- Rega PP, Darling RG. CBRNE – glanders and melioidosis. Emedicine J. 2011;1:1–5.[↩]