Contents
What causes drooling
Drooling is also known as sialorrhea or ptyalis, is a debilitating symptom which occurs when there is excess saliva in the mouth beyond the lip margin 1. Drooling is common in normally developed babies but subsides between the ages 15 to 36 months with establishment of salivary continence 1. Drooling is considered abnormal after age 4 2. Excessive drooling is a major issue in children with cerebral palsy and adults with neurodegenerative disorders 3.
A mild degree of drooling is normal in in infants and toddlers. Normal children usually stop drooling by two years of age. As such, no treatment is necessary for these children. No active management is necessary for patients who have little functional or psychological impairment apart from their objectively mild or intermittent drooling 4. Likewise, no active treatment is necessary if the child’s neurological status has not plateaued for at least six months. Improvement in the child’s drooling may occur as the child recovers from a neurological insult 5. Drooling in infants and young children may get worse with colds and allergies. Some drooling may occur with teething.
Drooling may happen if your body makes too much saliva. Infections can cause this, including:
- Mononucleosis
- Peritonsillar abscess
- Strep throat
- Sinus infections
- Tonsillitis
Other conditions that can cause too much saliva are:
- Allergies
- Heartburn or GERD (gastroesophageal reflux disease)
- Poisoning (especially by pesticides)
- Pregnancy (may be due to pregnancy side effects, such as nausea or reflux)
- Reaction to snake or insect venom
- Swollen adenoids
- Use of certain medicines
Drooling may also be caused by nervous system disorders that make it hard to swallow. Examples are:
- Amyotrophic lateral sclerosis, or ALS
- Autism
- Cerebral palsy (CP)
- Down syndrome
- Multiple sclerosis
- Parkinson disease
- Stroke
Pathologic drooling can be an isolated phenomenon due to hypersalivation or occur in conjunction with several neurologic disorders such as amyotrophic lateral sclerosis (ALS), cerebral palsy, Parkinson’s disease or as a side effect of medications 3. In children, the most common cause of drooling is cerebral palsy, which persists in 10%–38% of these individuals 6. In adults, Parkinson’s disease is the most common cause 7 with 70%–80% of Parkinson’s disease patients demonstrating drooling 8. In 30%–80% of schizophrenic patients, hypersalivation when taking clozapine is manifested 9. Regardless of the cause, drooling is problematic, leading to clinical and functional complications such as impairment in social functioning (embarrassment and isolation), aspiration, skin breakdown, bad odor, and infection 10.
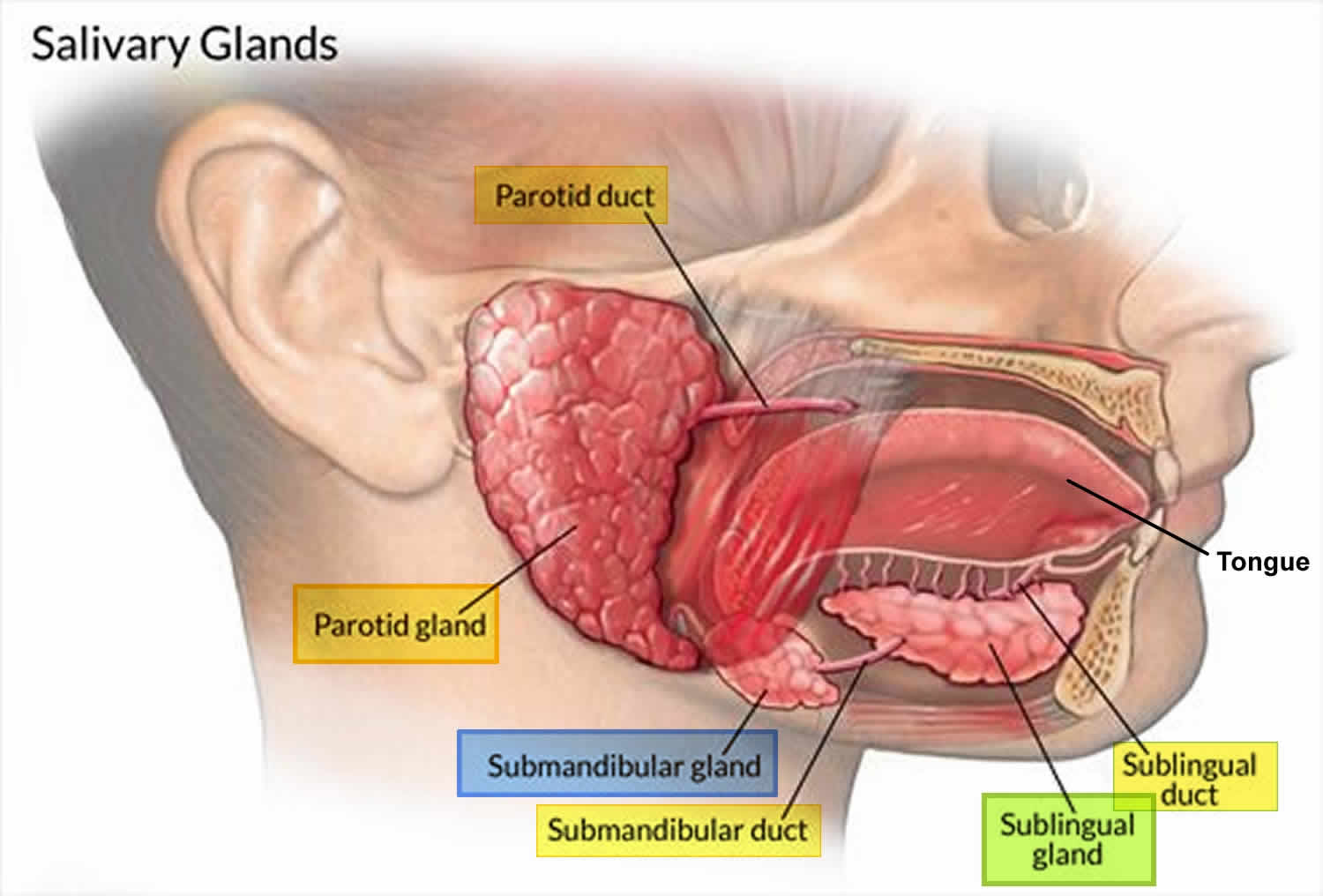
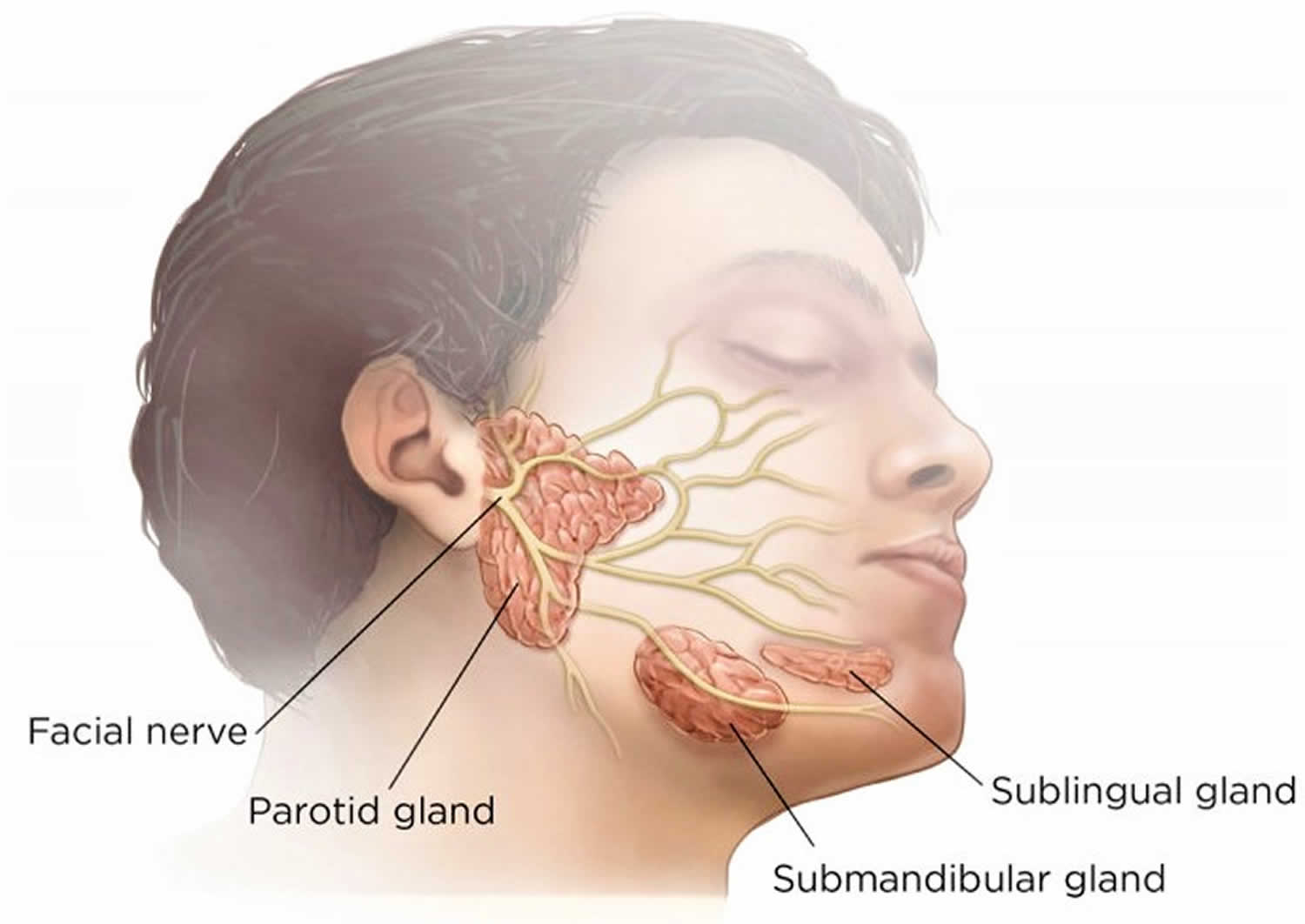
The major salivary glands include parotid, submandibular, and sublingual glands; the largest being the parotid gland. These glands secrete saliva which has a major role in lubrication, digestion, immunity and maintenance of homeostasis in the human body 11. The parotid gland is located in the preauricular region, along the posterior surface of the mandible and is divided by the facial nerve into a superficial lobe and a deep lobe. The submandibular gland is the second largest major salivary gland, is located in the submandibular triangle, and lies posterolateral to the mylohyoid muscle. The sublingual gland is the smallest of the three and lies in the anterior floor of the mouth 12. Saliva is produced in high volumes relative to the mass of each gland (parotid produces the most because it is the largest) and is almost completely controlled extrinsically by the autonomic nervous system, both sympathetic and parasympathetic divisions 11.
There are hundreds of minor salivary glands throughout the mouth and the aerodigestive tract. Unlike the major salivary glands, these glands are too small to be seen without a microscope. Most are found in the lining of the lips, the tongue, and the roof of the mouth, as well as inside the cheeks, nose, sinuses, and larynx (voice box).
Figure 1. Major salivary glands
Figure 2. Facial nerve (cranial nerve 7) and salivary glands
In the unstimulated state, 70% of saliva is secreted by submandibular and sublingual glands. Conversely, in the stimulated state the parotids glands provide most of the saliva. The flow of saliva is five times greater in the stimulated state than in the resting state 13. An example of an exogenous source causing stimulation is chewing.
Drooling can be either due to increased production of saliva (idiopathic or drug-induced) or related to failure of mechanisms that clear and remove saliva from the oral cavity. Disturbance in the coordination of orofacial and palate—lingual musculature is one mechanism that can lead to pooling of saliva in anterior portion of mouth. Ultimately, muscle incoordination inhibits the initiation of the swallow reflex, thereby further disrupting the path of saliva from the mouth to the oropharynx 14. Salivary secretion is regulated via a reflex arch which has various influences. The afferent branch consists of chemoreceptors in taste buds and mechanoreceptors in the periodontal ligament. Afferent innervations of cranial nerves 5, 7, 9 and 10 also play a role by carrying impulses to salivary nuclei in the medulla oblongata 15. Efferent influences are mainly parasympathetic via cranial nerve 7 which control the submandibular, sublingual, and other minor glands, and cranial nerve 9 which influences the parotid gland 15.
Drooling occurring with neurologic illnesses is usually due to impaired swallowing as a result of impaired neuromuscular function. Neuromucular activity of swallowing involves efficient coordination of several structures including the oral cavity, pharynx, larynx, and esophagus 13. These structures coordinate to form three phases; an oral phase which is under voluntary control, followed by the pharyngeal and esophageal phases which are under involuntary control 16. Spontaneous swallowing is necessary for drool control 13. In children with neurologic disorders, drooling appears to be an effect of inefficient tongue and/or bulbar control, rather than increased salivary secretion 17.
Causes of drooling
Causes of drooling include:
Central nervous system and muscular disorders
- Parkinson’s disease
- Pseudobulbar palsy
- Bulbar palsy
- Stroke
Hypersecretion
- Inflammation (teething, dental caries, oral-cavity infection, rabies)
- Medication side effects (tranquilizers, anticonvulsants)
- Gastroesophageal reflux
- Toxin exposure (mercury vapor)
Anatomic
- Macroglossia (enlarged tongue)
- Oral incompetence
- Dental malocclusion
- Orthodontic problems
- Oropharyngeal lesions
- Esophageal lesions
- Head and neck surgical defects (i.e., “Andy Gump” deformity)
Developmental
- Mental retardation
- Cerebral palsy
- Familial dysautonomia (Riley-Day syndrome)
- Wilson disease
- Rett syndrome
Physiological
- Teething
- Nausea
- Foods
- Emotional stimuli
Developmental causes
A mild degree of drooling is normal during infancy. The problem seems to be more prominent around five to six months of age when salivation increases to its full capacity. Drooling occurs because of the infant’s limited ability to swallow, the lack of front teeth to serve as a dam and the adaptation of the infant’s mouth in the opening position. Drooling normally disappears by two years of age as a consequence of physiological maturity of oral motor function.
Physiological causes
Drooling is a common sign of teething 18. The salivary reflex is stimulated by eruptions of teeth with resulting hypersecretion of saliva.
Hypersecretion of saliva occurs with nausea presumably due to reflexes originating in the stomach and intestines. The salivatory nuclei are excited by both taste and tactile stimuli from the tongue and other areas of the mouth. Ingestion of certain foods, particularly sour or spicy ones, may increase salivary flow. Salivation can also be stimulated by impulses arriving in the salivatory nuclei from higher centres of the brain. As such, marked salivation may occur when a person smells or eats his or her favourite foods.
Hypersecretion of saliva may also occur with pleasurable sensation or anticipated pain, presumably through activation of higher centres.
Central nervous system and muscular disorders
Drooling is a common occurrence in children with central nervous system and muscular disorders, such as cerebral palsy, facial nerve palsy, myasthenia gravis and polymyositis. It has been estimated that 25% to 35% of children with cerebral palsy drool to varying degrees and 10% of these children have embarrassing drooling 19. A significant number of these patients have dysfunction in the oral and pharyngeal phases of swallowing, insufficient sensory appreciation of external salivary loss, or a structural or functional inability to close the lips during the oral phase of swallowing 20. Dysfunction in the oral and pharyngeal phases of swallowing may be secondary to uncoordinated tongue movements, high tonus and spastic contraction of the pharyngoesophageal sphincter, dyscoordination between the pharynx and sphincter, and a lack of coordinated control of head and neck musculature 5.
Mental retardation
Drooling occurs in approximately 10% of children with mental retardation 21. Drooling may be secondary to a delay in the development of coordinated swallowing movement, inefficient and infrequent swallowing, lack of awareness of oral incompetence, and incomplete lip closure during swallowing 22. Many droolers have an infantile tongue thrust, which may cause problems with eating and swallowing 23.
Oropharyngeal lesions
Acute infections involving the mouth or throat such as gingivostomatitis from herpes simplex virus or coxsackievirus may cause hypersecretion of saliva. Other oropharyngeal lesions may cause drooling because of pain or difficulty in swallowing. These include severe tonsillitis, peritonsillar or retropharyngeal abscess, epiglottitis and damage to the oral or pharyngeal mucosa from caustic ingestion or direct trauma 24.
Esophageal lesions
Drooling may result from esophageal obstruction such as may occur with esophageal stricture or a foreign body in the esophagus 25. Drooling may also result from the ingestion of caustics or corrosive acids with injury to the esophagus 26.
Gastroesophageal reflux
Episodic hypersalivation and drooling may result from gastroesophageal reflux. It is believed that stimulation of the esophagus by gastric acids excites an esophagosalivary reflex 27.
Drugs and chemicals
Drugs that may cause hypersalivation include morphine, pilocarpine, methacholine, haloperidol and clozapine 28. Drooling secondary to the use of benzodiazepines such as nitrazepam can be explained by drug-induced cricopharyngeal incoordination with impaired swallowing 29.
Hypersalivation is a prominent feature of poisoning with mercury, selenium and organophosphate compounds 30. Drooling may also result from cocaine or phencyclidine intoxication 31. In the neonatal period, drooling may be a sign of withdrawal from maternal substance abuse.
Familial dysautonomia (Riley-Day syndrome)
Drooling is common in children with familial dysautonomia (Riley-Day syndrome). Drooling in familial dysautonomia is often due to difficulty in swallowing. Mass et al 32 studied 13 children with familial dysautonomia and found an increase in basal secretion of major salivary glands in children with familial dysautonomia. The authors postulated that denervation supersensitivity of partially denervated salivary glands could play a role in the hyperfunctioning of salivary glands in familial dysautonomia 32.
Wilson disease
Wilson disease (hepatolenticular degeneration) can present with a variety of symptoms and signs. The most frequent ones are, in order of frequency, jaundice, dysarthria, clumsiness, tremor, drooling, gait disturbance, malaise and arthralgia 33. Drooling in Wilson disease can be ascribed to dysfunction in the oral and pharyngeal phases of swallowing.
Rett syndrome
Rett syndrome is a progressive neurological disorder estimated to affect 1:10,000 to 1:15,000 of live females. Drooling is common in children with Rett syndrome. Drooling can be accounted for by hypersalivation as well as a difficulty with swallowing saliva 34.
3 month old drooling
While a baby’s first tooth can begin as early as 3 months, most likely you’ll see the first tooth start pushing through your baby’s gum line when your little one is between 4 and 7 months old. Some babies get their first tooth a little earlier and others a little later. Teething is the process of teeth moving and breaking through the gums. This is a normal developmental stage for your baby.
Often, the first teeth to appear usually are the two middle bottom front teeth, also known as the central incisors. They’re usually followed 4 to 8 weeks later by the four front upper teeth (central and lateral incisors). About a month later, the lower lateral incisors (the two teeth flanking the bottom front teeth) will appear.
Next to break through are the first molars (the back teeth used for grinding food), then finally the eyeteeth (the pointy teeth in the upper jaw). Most kids have all 20 of their primary teeth by the time children are 30 months (two and one-half years) old. (If your child’s teeth come in much slower than this, speak to your dentist.)
In some rare cases, kids are born with one or two teeth or have a tooth emerge within the first few weeks of life. Unless the teeth interfere with feeding or are loose enough to pose a choking risk, this is usually not a cause for concern.
What are the signs and symptoms of teething?
The following are the most common signs and symptoms of teething. However, each baby may experience symptoms differently. For some babies, teething is painless. Others may have brief periods of irritability, while some may seem cranky for weeks, with crying spells and disrupted sleeping and eating patterns. Teething can be uncomfortable, but if your baby seems very fussy, talk to your doctor.
Signs and symptoms of teething may include:
- Drooling more than usual (drooling may start as early as three or four months of age, but is not always a sign of teething)
- Constantly putting fingers or fists in the mouth (babies like to chew on things whether or not they are teething)
- Swollen, or puffy area on gum
- Fussiness or crankiness
- Teething babies naturally seek to put pressure on the gum over the emerging tooth. That’s why a teething baby is eager to chew on fingers, cloth and toys.
Teething does NOT cause colds, rashes, diarrhea, or fever, but it can make a baby uncomfortable. If your baby does develop a fever during the teething phase, something else is probably causing the fever and you should see your baby’s doctor. If your baby becomes sick around the same time teeth are coming in, or seems to be cranky or fussy for longer than normal, it is important to evaluate the symptoms of that illness independently of the teething. See your child’s doctor for advice if your baby is sick.
How can you help your child with the discomforts of teething?
If your baby is cranky with teething, try giving him/her hard rubber toys, teething rings, or cold teething toys to chew on. Do not freeze teething toys or rings as these can hurt your baby’s gums. You can also rub your baby’s gum with your finger. Teething gels may not be helpful as they are quickly washed off if excessive drooling is present, which may cause the effect of the gels to be short-lived. Something cold on the gums usually soothes and numbs the gums better. Ask your baby’s doctor about pain-relieving medications for teething.
You can help your teething baby by:
- Gently wipe your baby’s face often with a cloth to remove the drool and prevent rashes from developing.
- Massaging your child’s gums with a clean finger or toothbrush.
- Providing a cold, wet washcloth for your child to chew.
- Giving your child a teething ring or toy made of soft silicone, rubber or wood. Make sure it’s big enough that it can’t be swallowed or choked on and that it can’t break into small pieces. A wet washcloth placed in the freezer for 30 minutes makes a handy teething aid. Be sure to take it out of the freezer before it becomes rock hard — you don’t want to bruise those already swollen gums — and be sure to wash it after each use.
- Rubber teething rings are also good, but avoid ones with liquid inside because they may break or leak. If you use a teething ring, chill it in the refrigerator, but NOT the freezer. Also, never boil to sterilize it — extreme changes in temperature could cause the plastic to get damaged and leak chemicals.
- Teething biscuits and frozen or cold food are only OK for kids who already eat solid foods. Don’t use them if your child has not yet started solids. And make sure to watch your baby to make sure that no pieces break off or pose a choking hazard.
- If your baby seems irritable, ask your doctor if it is OK to give a dose of acetaminophen or ibuprofen (for babies older than 6 months) to ease discomfort.
- Never place an aspirin against the tooth, and don’t rub alcohol on your baby’s gums.
- Never tie a teething ring around a baby’s neck or any other body part — it could get caught on something and strangle the baby.
- Don’t use teething necklaces made of amber. These can lead to strangulation or choking if pieces break off.
- Don’t use teething gels and tablets because they may not be safe for babies.
Chilling a wet washcloth or teething toy can provide additional relief, but don’t freeze it solid, as that will make it too hard for your baby’s teeth and gums. If the item was in the freezer, be sure to let it thaw partially before giving it to your baby.
Some popular — and safe — teething toys include:
- Sophie the Giraffe (rubber teething toy)
- Lifefactory silicone teething rings
- Wooden teething rings, such as the Grimm’s hedgehog teether
If your child is old enough to eat solid foods, offering semi-frozen, easily-mashed fruit is another option. For example, freeze a pineapple ring, then remove it from the freezer for a few minutes until it begins to soften. Teething wafers should only be given to babies who are eating solid foods and are old enough to handle the pieces that can break off. These wafers are a choking hazard to younger babies.
In most cases, all your teething child needs is a massage of their gums or something to chew on. But if that’s not enough, and they are clearly in pain, you might ask your doctor for guidance on an age-appropriate dose of pain reliever such as Tylenol.
AVOID these remedies
As you may have seen in the news, the FDA recently issued a warning against the use of homeopathic teething tablets and gels. In an investigation that is still ongoing, the FDA has associated the deaths of at least 10 babies and the serious illness of many others with the use of these remedies. CVS has removed all homeopathic teething remedies from its shelves, and parents are advised to throw away any homeopathic teething remedies they may have in their homes.
Over-the-counter gels and liquid benzocaine products, such as Anbesol® or Orajel®, are also unsafe for teething babies. The FDA issued a warning in 2011 against the use of these products with children under 2 years old due to the risk of methemoglobinemia — a sometimes-fatal condition that results in a severe drop in the amount of oxygen in the bloodstream.
Amber teething necklaces should also be avoided. These are sometimes sold with claims that the amber releases a pain-relieving chemical that is absorbed through the baby’s skin. However, there is no scientific evidence that they actually soothe pain, and they pose a potential risk for both strangulation and choking. The American Academy of Pediatrics advises parents not to allow babies to wear any jewelry because of these risks.
How should I care for my baby’s teeth?
The care and cleaning of your baby’s teeth is important for long-term dental health. Even though the first set of teeth will fall out, tooth decay makes them fall out more quickly, leaving gaps before the permanent teeth are ready to come in. The remaining primary teeth may then crowd together to attempt to fill in the gaps, which may cause the permanent teeth to come in crooked and out of place.
Daily dental care should begin even before your baby’s first tooth comes in. Wipe your baby’s gums daily with a clean, damp washcloth or gauze, or brush them gently with a soft, infant-sized toothbrush and water (no toothpaste!).
As soon as the first tooth appears, brush it with water and fluoridated toothpaste, using only a tiny amount.
It’s OK to use a little more toothpaste once a child is old enough to spit it out — usually around age 3. Choose one with fluoride and use only a pea-sized amount or less in younger kids. Don’t let your child swallow the toothpaste or eat it out of the tube because an overdose of fluoride can be harmful to kids.
By the time all your baby’s teeth are in, try to brush them at least twice a day and especially after meals. It’s also important to get kids used to flossing early on. A good time to start flossing is when two teeth start to touch. Talk to your dentist for advice on flossing those tiny teeth. You also can get toddlers interested in the routine by letting them watch and imitate you as you brush and floss.
Another important tip for preventing tooth decay: Don’t let your baby fall asleep with a bottle. The milk or juice can pool in a baby’s mouth and cause tooth decay and plaque.
The American Dental Association (ADA) recommends that kids see a dentist by age 1, or within 6 months after the first tooth appears, to spot any potential problems and advise parents about preventive care.
Drooling diagnosis
A thorough history and physical examination are important in the evaluation of person with drooling.
History
Age of onset:
- Drooling in the neonatal period should alert the physician to the possibility of esophageal atresia or withdrawal from maternal substance abuse. A mild degree of drooling is normal during infancy.
Chronicity:
- An acute onset suggests an infection or drug intoxication. Drooling of long duration may be developmental or secondary to a structural lesion, neuromuscular disorder or mental retardation.
Severity:
- Severe drooling can lead to social embarrassment. The severity can be gauged by the frequency of bathing, wiping and need for bibs or clothing changes.
Precipitating factors:
- Any precipitating factors such as ingestion of food and teething should be noted.
Associated symptoms:
Fever, agitation, aphonia, dyspnea and stridor suggest epiglottitis. Fever, sore throat and dysphagia suggest peritonsillar abscess. A history of choking, gagging, coughing, vomiting and dyspnea suggests a foreign body in the esophagus. A history of regurgitations, especially since the neonatal period, is suggestive of gastroesophageal reflux 35. Lacrimation, sweating, headache, dizziness and cramps suggest intoxication with organophosphates. Feeding difficulties, excessive sweating, syncope, insensitivity to pain, slurred speech and seizures are features of familial dysautonomia. Developmental stagnation, altered communicative ability, loss of active play interaction, social withdrawal, stereotypic movements, periodic apnea, intermittent hyperventilation, constipation, weight loss, apparent insensitivity to pain, digit sucking or biting, and night-time laughing suggest Rett syndrome 34.
Developmental history:
- A thorough developmental history is of extreme importance. Generalized delay in all aspects of developmental milestones suggests mental retardation.
Drug use:
- A detailed drug history is important because the use of medication such as haloperidol, pilocarpine and diazepines may lead to drooling.
Psychosocial history:
- Any psychosocial or emotional stress should be noted as a potential cause of the drooling. In addition, the impact of drooling on the child and family should be noted.
Perinatal history:
- The perinatal history should include maternal illness during the pregnancy, gestational age at birth, birth weight, perinatal trauma, asphyxia and infections.
Past history:
- Significant illnesses such as cerebral palsy, facial nerve palsy, myasthenia gravis and gastroesophageal reflux should be noted.
Family history:
- A family history of Wilson disease, Rett syndrome or familial dysautonomia suggests the corresponding disorder.
Physical examination
General:
Weight, height and head circumference should be plotted on standard growth charts. Poor growth may indicate a chronic disorder such as Rett syndrome or intrauterine growth retardation. Vital signs such as temperature, respiratory rate, heart rate and blood pressure should be noted. If the patient has a fever, the fever may indicate an underlying infection. Intermittent hyperventilation suggests Rett syndrome. Postural hypotension is suggestive of familial dysautonomia. The patient’s clothes should be inspected for wetness or staining. Particular attention should be paid to tongue control, ulcers in the oral cavity and evidence of dental problems. A comprehensive developmental assessment should be done if mental retardation is suspected 36.
Associated signs:
Dysmorphic features may suggest certain syndromes associated with mental retardation 36. Fever, trismus, a swollen and inflamed tonsillar area, and deviation of the uvula to the opposite side suggest periton-sillar abscess. Toxicity, fever, respiratory distress with inspiratory stridor, flaring of the alae nasi and inspiratory retractions of the suprasternal notch suggest epiglottitis. Spasticity, hyperreflexia, ankle clonus, extensor plantar response, dysarthria, athetosis, ataxia and contractures suggest cerebral palsy. Inability to close the eye and drooling at the corner of the mouth point to facial nerve palsy. Jaundice, dysarthria, clumsiness, tremor, gait disturbances and the presence of Kayser-Fleischer rings point to Wilson disease 33.
Drooling tests
Laboratory tests are usually not necessary in the majority of children with drooling; tests should be ordered only when indicated by a history or physical examination. A complete blood count is useful if an infection is suspected. Anteroposterior radiographs of the neck using a soft tissue technique are very useful for localizing a radio-opaque foreign body, detecting increased thickness of the prevertebral soft tissue and confirming or ruling out a swollen epiglottis. An upper gastrointestinal series may be considered to rule out the possibility of esophageal stricture or gastroesophageal reflux 35.
The Denver Developmental Screening Test should be performed if mental retardation is suspected 36. Those children who are found to be abnormal by a screening test require more definitive testing with one of the standardized and validated tests of intelligence. The most widely used intelligence tests to assess the intellectual and adaptive functioning of a child are the Standford-Binet Intelligence Scale, the Bayley Scales of Infant Development, the Wechsler Intelligence Scale for Children-Revised, and the Wechsler Preschool and Primary Scale of Intelligence 36.
A method for quantitative measurement of drooling using radioisotopes has been described 37. The procedure consists of injecting a radioisotope into the subject, having the radioisotope excreted in the saliva and sampling the saliva periodically. From the measured radioactivity in the bibs and the salivary samples, the amount of drooled saliva can be calculated 37. The use of radioisotopes to quantify drooling is mainly of academic interest. Occasionally, it is used before surgery in some patients and may, in certain cases, provide a tool to measure the change in salivary flow following a surgical intervention.
How to stop drooling
Drooling is known to be difficult to treat. The underlying cause of drooling should be treated whenever possible. Epiglottitis should be treated with parenteral antibiotic therapy. Esophageal atresia or stricture should be corrected surgically. Drugs that may cause or aggravate drooling should be discontinued. The use of a barrier cream is recommended for children who have dermatitis resulting from drooling.
Management can be conservative or more invasive. Conservative treatments include changes in diet or habits, oral-motor exercises, intra-oral devices such as palatal training devices, and medical treatments such as medication or botulinum toxin injections 3. Behavior modification has been advocated for many decades but results are inconsistent and time consuming 38. More invasive treatments include surgery or radiation 39. While surgical cases seem to offer more permanent results, they are invasive and are not without side effects. Radiation is now rarely applied and is typically reserved for elderly patients who are not candidates for surgery and cannot tolerate medical therapies 40.
Table 1. Medications for treatment of excessive drooling
| Agent | How supplied | Dosage | Side effects | Cost* |
|---|---|---|---|---|
Glycopyrrolate | Scored tablets,† 1 or 2 mg | Adults: Start at 0.5 mg orally, one to three times daily; titrate to effectiveness and tolerability‡ | Constipation, excessive oral dryness, urinary retention, blurred vision, hyperactivity, irritability | $ 0.66 per 1-mg tablet |
Children: 0.04 mg per kg per dose orally, two to three times daily; titrate to effectiveness and tolerability | ||||
Scopolamine (Transderm Scop) | Patch, 1.5 mg | Apply patch every day | Pruritus at patch site, urinary retention, irritability, blurred vision, dizziness, glaucoma | 20.99 per four 1.5-mg patches |
Botulinum toxin A | Vial, 100 U per vial | Under ultrasound guidance, injections of 10 to 40 units into each submandibular and parotid gland | Pain at injection site, excessive oral dryness | 521.25 per vial |
Footnote:
*—Estimated cost to the pharmacist based on average wholesale prices in Red Book. Montvale, N.J.: Medical Economics Data, 2001. Cost to the patient will be higher, depending on prescription filling fee.
†—Tablets may be crushed, if patient prefers.
‡—The maximum daily dose for adults is 8 mg.
[Source 41 ]Behavioral or biofeedback modification
Children who have only a moderate drooling problem and have normal or close to normal intelligence and high motivation may benefit from behavioural or biofeedback modification 42. Behavioral or biofeedback modification programs have used various combinations of cuing, positive reinforcement and punishment. One method involves a number of sessions during which the patient is conditioned to swallow in response to an auditory cue emitted by an electronic device 42. Parental reminding to swallow is also effective.
Oral therapy
Oral therapy for drooling encompasses the use of anticholinergic agents such as glycopyrrolate, benztropine, scopalamine and tropicamide 3. Biperiden has also been implicated for use in the literature but was found in one study to have an adverse effect on cognition, thus limiting its use 43. Glycopyrrolate oral solution is the first drug treatment which was approved in the United States for drooling in children with neurologic conditions 44. Anticholinergic agents work by downregulating acetylcholine and ultimately decreasing saliva secretion through the parasympathetic autonomic nervous system 39. Elderly patients have poor tolerance for anticholinergic agents. Glycopyrrolate specifically has a quaternary ammonium structure and thus cannot pass the blood-brain barrier in large amounts, ultimately decreasing the occurrence of central side effects 45. It is effective and safe at 1 mg, three times a day. Intraoral tropicamide films provide short-term relief of sialorrhea. One study provided evidence that 1 mg of tropicamide resulted in significant Visual Analog Scale (VAS) score decrease and reduction in saliva volume in non-demented Parkinson’s disease patients 46. Antireflux medication has also been suggested for use in drooling 47; however, there are no double-blind studies in the literature to offer evidence for this recommendation.
Topical therapy
One double-blind study of 30 patients handicapped by debilitating illnesses (cerebral palsy, epilepsy, autism, Down syndrome and rare disorders) is described in the literature using transdermal scopolamine 3. Moderate or severe mental retardation was present in 80% of patients. Severity of drooling was quantified using a modified Thomas-Stonell and Greenberg visual scale and frequency of drooling was monitored by number of bibs used each day. A significant drooling reduction was noted; however, four patients dropped out due to side effects and minor adverse events were noted in three others. It was concluded that though scopolamine can be useful to control drooling in severely disabled patients, it is not free from adverse effects, thus requiring careful patient selection 48.
Botulinum toxin injection
The effect of Botulinum toxin on drooling was first noted in Parkinson’s disease patients 49. Botulinum toxin, a potent neurotoxin blocks the release of acetylcholine and a number of other neurotransmitters from synaptic vesicles 49; in this case post-ganglionic parasympathetic fibers are cholinergic. Botulinum toxin injection has been reported to be effective in the treatment of drooling through various open label trials, retrospective studies, case studies and controlled clinical trials. Currently, three type A and one type B Botulinum toxin are approved for use in US. These include OnabotulinumtoxinA (A/Ona), AbobotulinumtoxinA (A/Abo), IncobotulinumtoxinA (A/Inco), and RimabotulinumtoxinB (B/Rima).
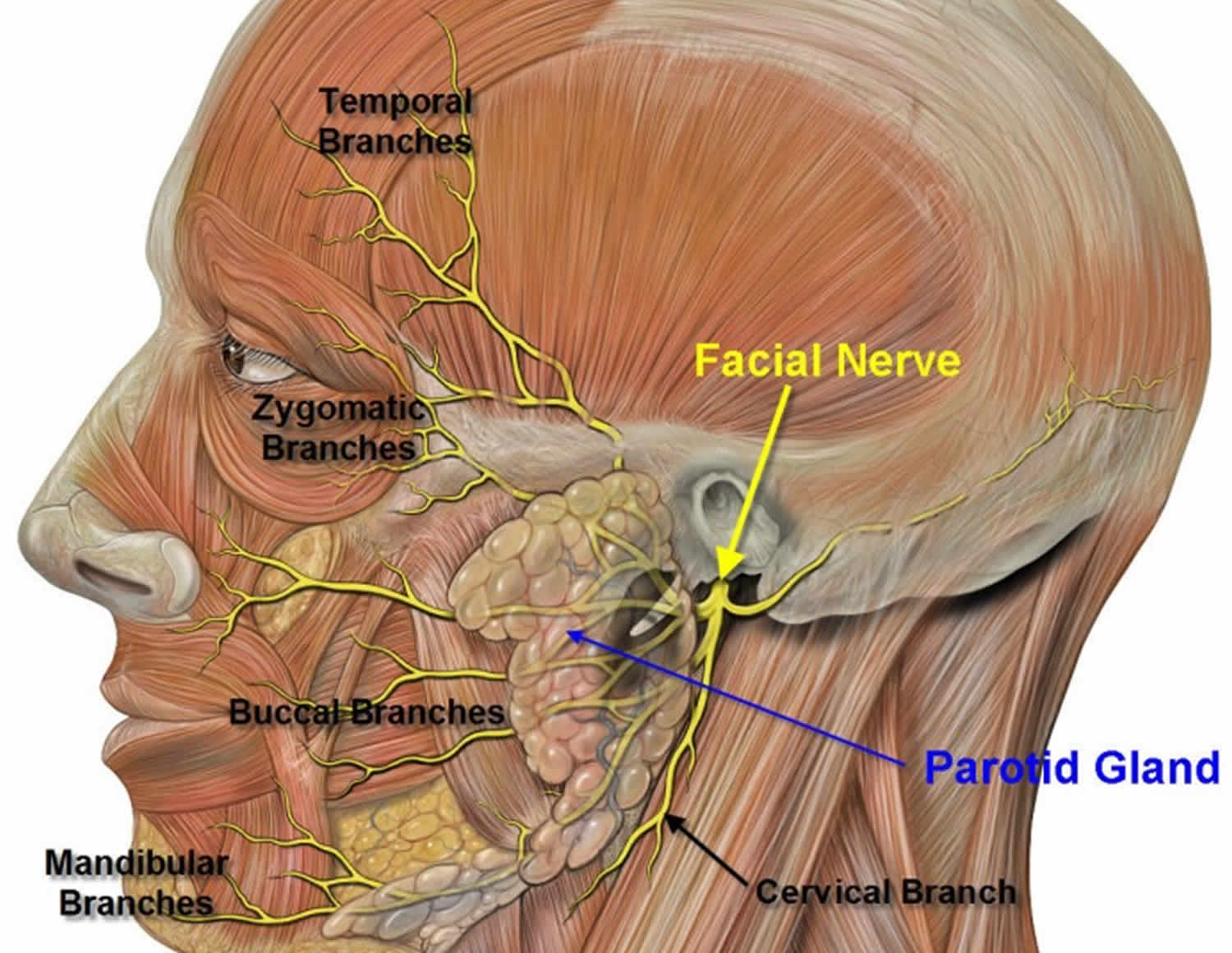
Below is the evidence for the use of Botulinum toxin in drooling divided into three categories: BotulinumtoxinA (BoNT-A) vs. placebo (Table 2), BotulinumtoxinB (BoNT-B) vs. placebo (Table 2) and comparator studies looking at BotulinumtoxinA (BoNT-A) vs. BotulinumtoxinB (BoNT-B) or BotulinumtoxinA (BoNT-A) vs. topical (transdermal) agents (Table 3). Botulinum toxin injections are given in the parotid and submandibular glands, as they are the greatest contributors to salivary production 50. The facial nerve, which is important in facial expression, is very close to the parotid gland; caution must be taken when injecting to avoid this nerve (Figure 3). Although local anesthesia may be used to reduce the pain of injection, out of all the studies reported in this review, only two used it. In one, authors used generalized anesthesia before injection in the pediatric population 51. In the other, local anesthesia with Emla cream was used in five of the patients 52.
Table 2. BotulinumtoxinA (BoNT-A) vs. Placebo studies
| Author/year | Method used to locate injection site | Method of injection | Number of sites injected | Use of anesthesia/type |
|---|---|---|---|---|
| Lipp et al. 2003 53 | Anatomic landmarks | 30-gauge, 25-mm needle were used to inject each parotid gland. (one in gland mass (0.3 mL), one above masseter 0.2 mL) | 2 per parotid | Not stated |
| Mancini 2003 54 | Ultrasound | Through a 26-gauge syringe, 0.65 mL of solution in each parotid gland and 0.35 mL of solution in each submandibular gland. | Not clear, but assumed 1 injection per gland on both sides | Not stated, however patients complained of painful injections |
| Lagalla et al. 2006 55 | Anatomic landmarks | 27-gauge needle penetrating to adepth of 1–1.5 cm into the preauricular portion of parotid gland, behind the angle of the ascending mandibular ramus, and then into the inferoposterior portion of the gland, lying just before the mastoid process. | 3 per parotid | Not states |
| Lin et al. 2008 56 | Ultrasound | Not stated in paper | Not stated in paper | Not stated |
| Alrefai et al. 2009 57 | Anatomic landmarks | Each side was injected with a 10 mm (30 G) needle into the paotid gland with 50 U. | 2 per parotid | Not used |
| Pei-Hsuan Wu et al. 2011 58 | Ultrasound | 30-gauge needle to the bilateral parotid and submandibular glands with concentration of 10 U/0.1 mL. | 4 (1 injection in both parotids and submandibular glands) | Not stated |
Table 3. Comparator studies
| Author/year | Assoc. illness | N | Class | Agent/dose | Glands injected | Primary outcome | Result | Side effects |
|---|---|---|---|---|---|---|---|---|
| Jongerius et al. 2004 59 | cerebral palsy | 45 | III | OnabotulinumtoxinA by weight vs. transdermal scopolamine | bilateral Submandibular gland | Drooling quotient, Teacher Drooling Scale (TDS) and Visual Analog Scale (VAS) | Drooling decreased with both scopolamine and OnabotulinumtoxinA injection; however greatest reductions were achieved 2 to 8 weeks after OnabotulinumtoxinA injection. 61.5% of patients responded to botulinum toxin injections. Statistical significance for Drooling quotient was stated at (p < 0.05). Drooling quotient had a response rate of 53% for scopolamine and 48.7% for OnabotulinumtoxinA | 71% had moderate to severe side effects for scopolamine. Only minimal and incidental side effects were reported for botulinum toxin. |
| Wilken et al. 2008 51 | cerebral palsy or neurodegenerative disorder | 30 | III | 100 /kg of RimabotulinumtoxinB or 80 MU of OnabotulinumtoxinA | bilateral Parotid gland and Submandibular gland | Parent questionnaire and Teacher Drooling Scale (TDS) | Four weeks after the first injection 29 patients responded with a reduction of Teacher Drooling Scale (TDS) score to 1 or 2 rated in the parent’ s questionnaire. | Intermittent problems with swallowing due to viscous saliva (5), unilateral parotitis (1). |
| Guibaldi et al. 2011 60 | Amyotrophic Lateral Sclerosis (ALS) (15) and Parkinson’s disease (12) | 27 | III | 250 U ABobotulinumtoxinA or 2,500 U RimabotulinumtoxinB | bilateral Parotid gland and Submandibular gland | Magnitude of change in saliva production determined by weighing five cotton rolls after retaining for 5 minutes in the mouth. | RimabotulinumtoxinB showed improvement in subjective and objective measured with a shorter latency for improvement onset when compared to ABobotulinumtoxinA (p = 0.002). Mean benefit of duration was 75 days for ABobotulinumtoxinA and 93 days for RimabotulinumtoxinB. | Change in saliva thickness |
As implied from this data, there is now strong evidence that both A and B types of botulinum toxins are effective in treatment of drooling and both have a low profile of side effects. Despite the strong level of evidence collectively, the cited studies are not without limitations. One such limitation is that of outcome measures. There is significant heterogeneity of outcome measures among the studies. In some studies outcome measures were poorly defined. Some studies measured only qualitative clinical scales (drooling and activity of daily living) 58, others measured quantitative data such as saliva flow through weight of dental rolls 61 and yet others measured both 56. Outcome measures were also evaluated at different time points (for most studies at four weeks).
Another limitation is variability of injection technique. A recent review paper stated that factors affecting diffusion and spread of botulinum toxin include dose, concentration and volume of injectate, number of injections, injection site, rate of injection, gauge of needle, and distance of needle tip from the neuromuscular junction 62. The optimal dose and dilution concentration (1:1, 2:1, 4:1) still deserve further investigation.
In regards to duration of effect, one study suggested a length of duration up to six months 60, whereas most others reported three months.
For location of injection, the studies presented focus on injections into the salivary glands (parotid and submandibular). Only one study discussed experience with direct injection into sublingual glands under the tongue with BotulinumtoxinB (BoNT-B) which lead to occurrence of dysphagia in two of four patients without any improvement in drooling, thus discouraging this approach 63.
A lack of correlation between reduction in salivary secretion and improvement in drooling has been observed in some studies and is probably related to the fact that factors influencing the severity of drooling and reduction of saliva secretion are variable 64.
Side effects in general were few and mild. Among them, transient dysphagia was the most worrisome, resolving in two weeks and affecting a small number of patients.
One comparator study suggested better efficacy for type B toxin. Another indicated more side effects with type B. These need to be further investigated with weighing the efficacy vs. side effects.
A recent meta-analysis study (2012) 65, which reviewed all randomized placebo-controlled trials encompassing 181 patients, concluded that botulinum toxin decreases the severity of drooling in patients with sialorrhea with statistical significance in both adult and pediatric populations. It was further concluded that both BotulinumtoxinA (BoNT-A) and BotulinumtoxinB (BoNT-B) produce similar effects 65. Furthermore, BotulinumtoxinA (BoNT-A) toxin doses greater than 50 U produced stronger effects. Increased saliva thickness (3.9%), dysphagia (3.3%), xerostomia (dry mouth) (3.3%), and pneumonia (2.2%) are noted as common side effects. Botulinum toxin treatment holds many advantages over older proposed methods of therapy: limited side effect profile, convenience, low risk of aspiration and its minimally invasive nature, are just a few 65. Limitations of using botulinum toxin injections include expense and need for repeated sedation, which is more problematic in pediatric population.
Surgery
Various surgical procedures have been suggested for treatment of drooling. These procedures all encompass an alteration in the anatomy of the salivary glands and include salivary gland excision, denervation of the salivary glands, and transposition or ligation of the salivary ducts 66. These procedures are reserved for severe cases and of course are not without their side effects.
The most definitive treatment of sialorrhea is surgery to excise the major salivary glands or to ligate or reroute the major salivary ducts. This procedure typically involves a combination parotid duct ligation or rerouting with either submandibular gland excision or duct rerouting. Sublingual gland excision is suggested if the submandibular ducts are rerouted to prevent formation of salivary retention cysts 67. Preservation of salivation with reduction of drooling has been demonstrated following rerouting of the parotid and submandibular ducts to the posterior oropharynx, and rerouting procedures spare patients external scars and the risk of facial nerve injury 68.
The most definitive surgical procedure, which includes bilateral parotid duct ligation and submandibular gland excision, is highly successful, with nearly total elimination of sialorrhea, a low incidence of facial weakness, and significant patient and caretaker satisfaction 69. Although this is the most invasive of treatment options, the severity of drooling may be sufficient to require such an aggressive therapy.
Table 4. Advantages and disadvantages of surgical therapies for excessive drooling
| Surgical therapy | Advantages | Disadvantages |
|---|---|---|
Submandibular duct relocation | No external scar | Duct relocation is an uncommon procedure |
Low incidence of ranula with sublingual gland excision | ||
Potential for anterior dental caries | ||
Without sublingual gland excision, patient may develop ranula | ||
Potential for aspiration | ||
Submandibular gland excision | Very good control of sialorrhea | External scar |
Commonly performed procedure | Potential for dental caries | |
Parotid duct relocation | Redirects flow in the stimulated state | Risk of sialocele |
Potential for aspiration | ||
Relocation is uncommon procedure | ||
Parotid duct ligation | Simple, fast procedure | Risk of sialocele |
Decreases flow in the stimulated state | ||
Transtympanic neurectomy | Technically easy, fast procedure | Predictable return of salivary function Requires multiple procedures |
Does not require general anesthesia | ||
Useful in elderly patients |
Glandular excision is definitely an option in the treatment of excessive drooling. Submandibular gland excision is a relatively simple procedure. Care has to be taken so as not to damage the mandibular branch of the facial nerve, the lingual nerve and the hypoglossal nerve. Excision of the submandibular gland leaves a visible scar, which is cosmetically unsightly. Parotidectomy is generally not recommended because of the risk of facial nerve injury 70.
The parasympathetic nerve supply to the submandibular gland and sublingual gland can be interrupted by sectioning of the chorda tympani nerve and that to the parotid gland by sectioning of the tympanic plexus. The procedure, which can be done via a tympanotomy approach to the middle ear, is easy to perform and has few complications. Denervation by tympanic neurectomy has a success rate of 50% to 90% 70. The long term failure rate has been estimated to be 25% to 50% 71. Regrowth of preganglionic fibres is believed to be the cause of late failures 72. The loss of taste to the anterior two-thirds of the tongue is insignificant when unilateral, but can be quite upsetting to the patient when bilateral. Because of the otological risks, the procedure is contraindicated in patients with unilateral sensorineural hearing loss 72. The procedure is best avoided in children with secretory otitis media 72.
Surgical ligation of the salivary ducts is easy to perform, and the results are immediate. However, postoperative facial swelling and pain may occur, particularly when the patient is eating. Other complications include recurrent infection in the affected glands, xerostomia and fistula formation 73.
Rerouting of the salivary ducts to the tonsillar fossa is easy to perform, and is the surgical treatment of choice 70. The procedure allows the affected individual to bypass the oral phase of swallowing and permits automatic pharyngeal swallowing (28,36). The sense of taste and the amount of saliva produced are not affected 72. Bilateral submandibular duct transposition has a high success rate with low morbidity 72. It leaves no external scars. Rerouting of the parotid ducts can be performed, if necessary, as a second stage procedure 74. Complications of rerouting of salivary ducts include ranula formation and stenosis of the transposed duct 70.
- Garnock-Jones K.P. Glycopyrrolate oral solution: For chronic, severe drooling in pediatric patients with neurologic conditions. Paediatric. Drugs. 2012;14:263–269. doi: 10.2165/11208120-000000000-00000. https://www.ncbi.nlm.nih.gov/pubmed/22646067[↩][↩]
- Hamdy S., Aziz Q., Rothwell J.C., Hobson A., Barlow J., Thompson D.G. Cranial nerve modulation of human cortical swallowing motor pathways. Am. J. Physiol. 1997;272:G802–G808.[↩]
- Lakraj AA, Moghimi N, Jabbari B. Sialorrhea: anatomy, pathophysiology and treatment with emphasis on the role of botulinum toxins. Toxins (Basel). 2013;5(5):1010-31. Published 2013 May 21. doi:10.3390/toxins5051010 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3709276/[↩][↩][↩][↩][↩][↩][↩]
- Finkelstein DM, Crysdale WS. Evaluation and management of the drooling patient. J Otolaryngol. 1992;21:414–8.[↩]
- Crysdale WS. Drooling. Experience with team assessment and management. Clin Pediatr. 1992;31:77–80.[↩][↩]
- Johnson H., Scott A. 6 Saliva Management. In: Cichero J.A.Y., Murdoch B.E., editors. Dysphagia: Foundation, Theory and Practice. John Wiley & Sons Ltd; West Sussex, UK: 2006. p. 126.[↩]
- Volonte M.A., Porta M., Comi G. Clinical assessment of dysphagia in early phases of parkinson’s disease. Neurol. Sci. 2002;23:S121–S122.[↩]
- Glickman S., Deaney C.N. Treatment of relative sialorrhoea with botulinum toxin type a: Description and rationale for an injection procedure with case report. Eur. J. Neurol. 2001;8:567–571. doi: 10.1046/j.1468-1331.2001.00328.x.[↩]
- Hung C.C., Fu P.K., Wang H.Y., Chan C.H., Lan T.H. Treatment effects of traditional chinese medicines suoquan pill and wuling powder on clozapine-induced hypersalivation in patients with schizophrenia: Study protocol of a randomized, placebo-controlled trial. J. Chin. Integr. Med. 2011;9:495–502. doi: 10.3736/jcim20110506.[↩]
- Scully C., Limeres J., Gleeson M., Tomas I., Diz P. Drooling. J. Oral Pathol. Med. 2009;38:321–327. doi: 10.1111/j.1600-0714.2008.00727.x[↩]
- Holsinger F.C., Bui D.T. Anatomy, function, and evaluation of the salivary glands. Salivary Glands Disorders. 2007;1:1–16. doi: 10.1007/978-3-540-47072-4_1.[↩][↩]
- Hollinshead W.H. Anatomy for Surgeons. Hoeber Medical Division, Harper and Row; New York, NY, USA: 1968. pp. 551–556.[↩]
- Scully C., Limeres J., Gleeson M., Tomas I., Diz P. Drooling. J. Oral Pathol. Med. 2009;38:321–327. doi: 10.1111/j.1600-0714.2008.00727.x.[↩][↩][↩]
- Myer C.M., 3rd. Sialorrhea. Pediatr. Clin. North Am. 1989;36:1495–1500.[↩]
- Garrett J.R., Proctor G.B. Control of Salivation. In: Linden D., Roger W.A., editors. The Scientific Basis of Eating: Taste and Smell, Salivation, Mastication and Swallowing and Their Dysfunctions; Frontiers of Oral Biology. Karger; Basel, Switzerland: 1998. pp. 135–155.[↩][↩]
- Dodds W.J. Physiology of swallowing. Dysphagia. 1989;3:171–178. doi: 10.1007/BF02407219.[↩]
- Senner J.E., Logemann J., Zecker S., Gaebler-Spira D. Drooling, saliva production, and swallowing in cerebral palsy. Dev. Med Child Neurol. 2004;46:801–806.[↩]
- Leung AK. Teething. Am Fam Physician. 1989;39:131–4.[↩]
- Becmeur F, Horta-Geraud P, Brunot B, Maniere MC, Prulhiere Y, Sauvage P. Diversion of salivary flow to treat patients with cerebral palsy. J Pediatr Surg. 1996;31:1629–33.[↩]
- Lespargot A, Langevin MF, Muller S, Guillemont S. Swallowing disturbances associated with drooling in cerebral-palsied children. Dev Med Child Neurol. 1993;35:298–304.[↩]
- Sellars SL. Surgery of sialorrhoea. J Laryngol Otol. 1985;99:1107–9.[↩]
- Blasco PA, Allaire JH. Drooling in the developmentally disabled: Management practices and recommendations. Consortium on Drooling. Dev Med Child Neurol. 1992;34:849–62.[↩]
- McCracken A. Drool control and tongue thrust therapy for the mentally retarded. Am J Occup Ther. 1978;32:79–85.[↩]
- Friedman NR, Mitchell RB, Pereira KD, Younis RT, Lazar RH. Peritonsillar abscess in early childhood. Presentation and management. Arch Otolaryngol Head Neck Surg. 1997;123:630–2.[↩]
- Swischuk LE. Cough and drooling for two days. Pediatr Emerg Care. 1995;11:397–8.[↩]
- Crain EF, Gershel JC, Mezey AP. Caustic ingestions. Symptoms as predictors of esophageal injury. Am J Dis Child. 1984;138:863–5.[↩]
- Mandel L, Tamari K. Sialorrhea and gastroesophageal reflux. J Am Dent Assoc. 1995;126:1537–41.[↩]
- The safety and efficacy of clozapine in severe treatment-resistant schizophrenic patients in the UK. Clozapine Study Group. Br J Psychiatry. 1993;163:150–4.[↩]
- Wyllie E, Wyllie R, Cruse RP, Rothner AD, Erenberg G. The mechanism of nitrazepam-induced drooling and aspiration. N Engl J Med. 1986;314:35–8.[↩]
- Nachlas NE, Johns NE. Regulation of secretion. In: Paparella MM, Shumrick DA, Gluckman JL, Meyerhoff WL, editors. Otolaryngology. Philadelphia: WB Saunders Co; 1991. pp. 396–400[↩]
- Mott SH, Packer RJ, Soldin SJ. Neurologic manifestations of cocaine exposure in childhood. Pediatrics. 1994;93:557–60[↩]
- Mass E, Wolff A, Gadoth N. Increased major salivary gland secretion in familial dysautonomia. Dev Med Child Neurol. 1996;38:133–8[↩][↩]
- Saito T. Presenting symptoms and natural history of Wilson disease. Eur J Pediatr. 1987;146:261–5.[↩][↩]
- Ribeiro RA, Romano AR, Birman EG, Mayer MP. Oral manifestations in Rett syndrome: A study of 17 cases. Pediatr Dent. 1997;19:349–52.[↩][↩]
- Leung AK. Gastroesophageal reflux in children. J Singapore Paediatr Soc. 1986;28:227–30.[↩][↩]
- Leung AK, Robson WL, Fagan J, Chopra S, Lim SH. Mental retardation. J R Soc Health. 1995;115:31–2. 37–9.[↩][↩][↩][↩]
- Ekedahl C, Hallen O. Quantitative measurement of drooling. Acta Otolaryngol. 1973;75:464–9.[↩][↩]
- Lin Y.C., Shieh J.Y., Cheng M.L., Yang P.Y. Botulinum toxin type a for control of drooling in asian patients with cerebral palsy. Neurology. 2008;70:316–318. doi: 10.1212/01.wnl.0000300421.38081.7d[↩]
- Fairhurst C.B., Cockerill H. Management of drooling in children. Arch. Dis. Child. Educ. Pract. Ed. 2011;96:25–30. doi: 10.1136/adc.2007.129478.[↩][↩]
- Borg M., Hirst F. The role of radiation therapy in the management of sialorrhea. Int. J. Radiat. Oncol. Biol. Phys. 1998;41:1113–1119. doi: 10.1016/S0360-3016(98)00153-9[↩]
- Sialorrhea: A Management Challenge. Am Fam Physician. 2004 Jun 1;69(11):2628-2635. https://www.aafp.org/afp/2004/0601/p2628.html[↩][↩]
- Drooling. Experience with team assessment and management. Crysdale WS. Clin Pediatr (Phila). 1992 Feb; 31(2):77-80.[↩][↩]
- Liang C.S., Ho P.S., Shen L.J., Lee W.K., Yang F.W., Chiang K.T. Comparison of the efficacy and impact on cognition of glycopyrrolate and biperiden for clozapine-induced sialorrhea in schizophrenic patients: A randomized, double-blind, crossover study. Schizophr. Res. 2010;119:138–144. doi: 10.1016/j.schres.2010.02.1060.[↩]
- Garnock-Jones K.P. Glycopyrrolate oral solution: For chronic, severe drooling in pediatric patients with neurologic conditions. Paediatric. Drugs. 2012;14:263–269. doi: 10.2165/11208120-000000000-00000.[↩]
- Arbouw M.E., Movig K.L., Koopmann M., Poels P.J., Guchelaar H.J., Egberts T.C., Neef C., van Vugt J.P. Glycopyrrolate for sialorrhea in parkinson disease: A randomized, double-blind, crossover trial. Neurology. 2010;74:1203–1207. doi: 10.1212/WNL.0b013e3181d8c1b7.[↩]
- Lloret S.P., Nano G., Carrosella A., Gamzu E., Merello M. A double-blind, placebo-controlled, randomized, crossover pilot study of the safety and efficacy of multiple doses of intra-oral tropicamide films for the short-term relief of sialorrhea symptoms in parkinson’s disease patients. J. Neurol. Sci. 2011;310:248–250. doi: 10.1016/j.jns.2011.05.021.[↩]
- Vashishta R., Nguyen S.A., White D.R., Gillespie M.B. Botulinum toxin for the treatment of sialorrhea: A meta-analysis. Otolaryngol. Head Neck Surg. 2013;148:191–196. doi: 10.1177/0194599812465059.[↩]
- Mato A., Limeres J., Tomas I., Munoz M., Abuin C., Feijoo J.F., Diz P. Management of drooling in disabled patients with scopolamine patches. Br. J. Clin. Pharmacol. 2010;69:684–688. doi: 10.1111/j.1365-2125.2010.03659.x[↩]
- Pal P.K., Calne D.B., Calne S., Tsui J.K. Botulinum toxin a as treatment for drooling saliva in pd. Neurology. 2000;54:244–247. doi: 10.1212/WNL.54.1.244.[↩][↩]
- Lagalla G., Millevolte M., Capecci M., Provinciali L., Ceravolo M.G. Long-lasting benefits of botulinum toxin type b in parkinson’s disease-related drooling. J. Neurol. 2009;256:563–567. doi: 10.1007/s00415-009-0085-1.[↩]
- Wilken B., Aslami B., Backes H. Successful treatment of drooling in children with neurological disorders with botulinum toxin a or b. Neuropediatrics. 2008;39:200–204. doi: 10.1055/s-0028-1112115.[↩][↩]
- Jongerius P.H., van den Hoogen F.J., van Limbeek J., Gabreels F.J., van Hulst K., Rotteveel J.J. Effect of botulinum toxin in the treatment of drooling: A controlled clinical trial. Pediatrics. 2004;114:620–627. doi: 10.1542/peds.2003-1104-L[↩]
- Pal P.K., Calne D.B., Calne S., Tsui J.K. Botulinum toxin a as treatment for drooling saliva in pd. Neurology. 2000;54:244–247. doi: 10.1212/WNL.54.1.244[↩]
- Lipp A., Trottenberg T., Schink T., Kupsch A., Arnold G. A randomized trial of botulinum toxin a for treatment of drooling. Neurology. 2003;61:1279–1281. doi: 10.1212/WNL.61.9.1279.[↩]
- Mancini F., Zangaglia R., Cristina S., Sommaruga M.G., Martignoni E., Nappi G., Pacchetti C. Double-blind, placebo-controlled study to evaluate the efficacy and safety of botulinum toxin type a in the treatment of drooling in parkinsonism. Mov. Disord. 2003;18:685–688. doi: 10.1002/mds.10420.[↩]
- Lin Y.C., Shieh J.Y., Cheng M.L., Yang P.Y. Botulinum toxin type a for control of drooling in asian patients with cerebral palsy. Neurology. 2008;70:316–318. doi: 10.1212/01.wnl.0000300421.38081.7d.[↩][↩]
- Lagalla G., Millevolte M., Capecci M., Provinciali L., Ceravolo M.G. Botulinum toxin type a for drooling in parkinson’s disease: A double-blind, randomized, placebo-controlled study. Mov. Disord. 2006;21:704–707. doi: 10.1002/mds.20793.[↩]
- Alrefai A.H., Aburahma S.K., Khader Y.S. Treatment of sialorrhea in children with cerebral palsy: A double-blind placebo controlled trial. Clin. Neurol. Neurosurg. 2009;111:79–82. doi: 10.1016/j.clineuro.2008.09.001.[↩][↩]
- Jongerius P.H., van den Hoogen F.J., van Limbeek J., Gabreels F.J., van Hulst K., Rotteveel J.J. Effect of botulinum toxin in the treatment of drooling: A controlled clinical trial. Pediatrics. 2004;114:620–627. doi: 10.1542/peds.2003-1104-L.[↩]
- Guidubaldi A., Fasano A., Ialongo T., Piano C., Pompili M., Masciana R., Siciliani L., Sabatelli M., Bentivoglio A.R. Botulinum toxin a versus b in sialorrhea: A prospective, randomized, double-blind, crossover pilot study in patients with amyotrophic lateral sclerosis or parkinson’s disease. Mov. Disord. 2011;26:313–319. doi: 10.1002/mds.23473.[↩][↩]
- Guidubaldi A., Fasano A., Ialongo T., Piano C., Pompili M., Masciana R., Siciliani L., Sabatelli M., Bentivoglio A.R. Botulinum toxin a versus b in sialorrhea: A prospective, randomized, double-blind, crossover pilot study in patients with amyotrophic lateral sclerosis or parkinson’s disease. Mov. Disord. 2011;26:313–319. doi: 10.1002/mds.23473[↩]
- Lim E.C., Seet R.C. Use of botulinum toxin in the neurology clinic. Nat. Rev. Neurol. 2010;6:624–636. doi: 10.1038/nrneurol.2010.149.[↩]
- Ondo W.G., Hunter C., Moore W. A double-blind placebo-controlled trial of botulinum toxin b for sialorrhea in parkinson’s disease. Neurology. 2004;62:37–40. doi: 10.1212/01.WNL.0000101713.81253.4C[↩]
- Wu K.P., Ke J.Y., Chen C.Y., Chen C.L., Chou M.Y., Pei Y.C. Botulinum toxin type a on oral health in treating sialorrhea in children with cerebral palsy: A randomized, double-blind, placebo-controlled study. J. Child Neurol. 2011;26:838–843. doi: 10.1177/0883073810395391[↩]
- Vashishta R., Nguyen S.A., White D.R., Gillespie M.B. Botulinum toxin for the treatment of sialorrhea: A meta-analysis. Otolaryngol. Head Neck Surg. 2013;148:191–196. doi: 10.1177/0194599812465059[↩][↩][↩]
- Crysdale W.S. Management options for the drooling patient. Ear Nose Throat J. 1989;68:820, 825–826, 829–830.[↩]
- Crysdale WS, Raveh E, McCann C, Roske L, Kotler A. Management of drooling in individuals with neurodisability: a surgical experience. Dev Med Child Neurol. 2001;43:379–83.[↩]
- Panarese A, Ghosh S, Hodgson D, McEwan J, Bull PD. Outcomes of submandibular duct re-implantation for sialorrhoea. Clin Otolaryngol. 2001;26:143–6.[↩]
- Shott SR, Myer CM 3d, Cotton RT. Surgical management of sialorrhea. Otolaryngol Head Neck Surg. 1989;101:47–50.[↩]
- Burton MJ. The surgical management of drooling. Dev Med Child Neurol. 1991;31:1110–6.[↩][↩][↩][↩]
- O’Dwyer TP, Conlon BJ. The surgical management of drooling –a 15 year follow-up. Clin Otolaryngol. 1997;22:284–7.[↩]
- Bailey CM. Management of the drooling child. Clin Otolaryngol. 1988;13:319–22.[↩][↩][↩][↩][↩]
- Harris SR, Purdy AH. Drooling and its management in cerebral palsy. Dev Med Child Neurol. 1987;29:807–11.[↩]
- Cotton RT, Richardson MA. The effect of submandibular duct rerouting in the treatment of sialorrhea in children. Otolaryngol Head Neck Surg. 1981;89:535–41.[↩]