Contents
What is hydromorphone
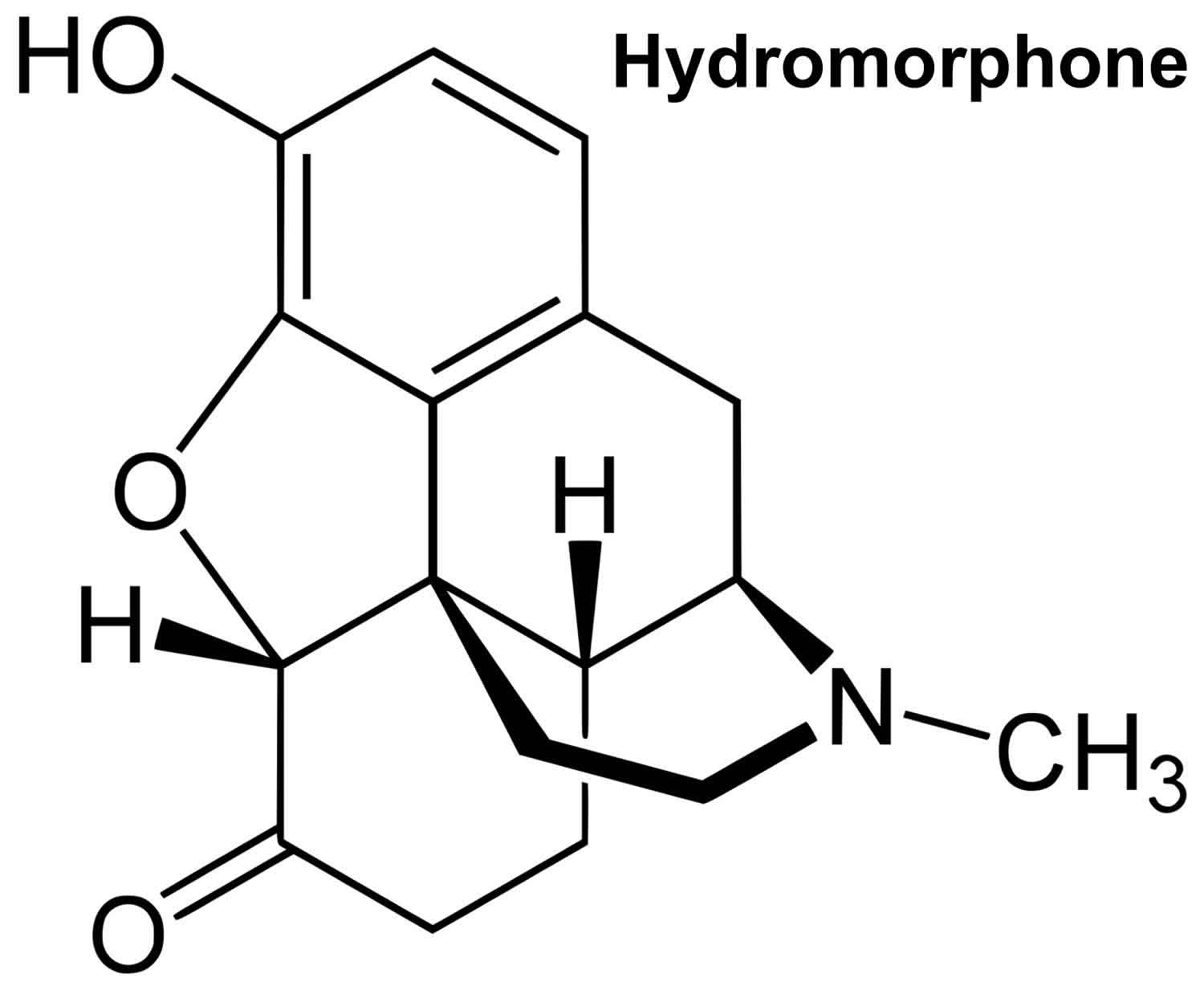
Hydromorphone (Dilaudid) is used to relieve moderate to severe acute pain, and severe chronic pain 1. Hydromorphone is in a class of medications called opiate (narcotic) analgesics. Hydromorphone is 2-8x more potent than morphine but shorter duration and greater sedation. Hydromorphone is an opioid agonist that binds to several opioid receptors. Hydromorphone analgesic effect is through its effect on the mu-opioid receptors, changing the way the brain and nervous system respond to pain. Hydromorphone also acts centrally at the level medulla, depressing the respiratory drive and suppressing cough.
Hydromorphone comes as oral solutions, tablets, an extended-release (long-acting) tablets to take by mouth, rectal suppositories and injectable formulations. Hydromorphone can be administered through intramuscular, intravenous, subcutaneous or oral routes. The liquid hydromorphone is usually taken every 3 to 6 hours and the tablets are usually taken every 4 to 6 hours. The extended-release tablets are taken once daily with or without food. The intramuscular, intravenous, or subcutaneous hydromorphone is injected at a concentration up to 10 mg/ml. Take hydromorphone at around the same time(s) every day. Follow the directions on your prescription label carefully, and ask your doctor or pharmacist to explain any part you do not understand. Take hydromorphone exactly as directed.
Hydromorphone formulations
As mentioned above, oral tablets can be in the immediate release form (Dilaudid) and the extended-release form (Exalgo).
- Dilaudid oral solutions have a dosage of 1 mg/1 ml, while oral tablets can be either 2 mg, 4 mg, or 8 mg.
- The dosage of Exalgo oral tablets is either 8 mg, 12 mg, 16 mg, or 32 mg. This latter does not have an oral solution form.
- Injection solutions can be found in the following concentrations: 1 mg/ml, 2 mg/ml, 4 mg/ml, and 10 mg/ml.
- Intravenous solutions are available in the following formulations: 2 mg/1 ml, 2500 mg/250 ml, 10 mg/1 ml, 500 mg/50 ml.
Do not allow the hydromorphone liquid to touch your skin or clothing. If such contact occurs, remove any clothes exposed to the oral liquid and wash your skin thoroughly with cool water.
Hydromorphone extended-release tablets are used to relieve severe pain in people who are expected to need pain medication around the clock for a long time and who cannot be treated with other medications. Hydromorphone extended-release tablets should only be used to treat people who are tolerant (used to the effects of the medication) to opioid medications because they have taken this type of medication for at least one week and should not be used to treat mild or moderate pain, short-term pain, pain after an operation or medical or dental procedure, or pain that can be controlled by medication that is taken as needed.
Your doctor may start you on a low dose of hydromorphone and gradually increase your dose, not more often than once every 3 to 4 days. Your doctor may decrease your dose if you experience side effects. Tell your doctor if you feel that your pain is not controlled or if you experience side effects during your treatment with hydromorphone. Do not change the dose of your medication without talking to your doctor.
Do not stop taking hydromorphone without talking to your doctor. Your doctor will probably decrease your dose gradually. If you suddenly stop taking hydromorphone, you may experience withdrawal symptoms including restlessness, teary eyes, runny nose, yawning, sweating, chills, hair standing on end, muscle or joint pain, widening of the pupils (black circles in the middle of the eyes), irritability, anxiety, backache, weakness, stomach cramps, difficulty falling asleep or staying asleep, nausea, loss of appetite, vomiting, diarrhea, fast breathing, or fast heartbeat. Your doctor will probably decrease your dose gradually. If you do not take hydromorphone extended-release tablets for longer than 3 days for any reason, talk to your doctor before you start taking the medication again.
Hydromorphone has potential adverse effects on several organ systems: the integumentary, gastrointestinal, neurologic, cardiovascular, endocrine and respiratory systems.
- Common side effects of hydromorphone include flushing, pruritus, sweating, dry mouth, nausea/vomiting, constipation, asthenia, dizziness, headache, and somnolence.
- Serious side effects of hydromorphone include hypotension, syncope, adrenal insufficiency, coma, raised intracranial pressure, seizure, suicidal thoughts, apnea, respiratory depression or respiratory arrest, drug dependence or drug withdrawal, and drug withdrawal syndrome in newborns.
Hydromorphone may be habit forming, especially with prolonged use. Take hydromorphone exactly as directed. Do not take more of it, take it more often, or take it in a different way than directed by your doctor. While you are taking hydromorphone, discuss with your health care provider your pain treatment goals, length of treatment, and other ways to manage your pain. Tell your doctor if you or anyone in your family drinks or has ever drunk large amounts of alcohol, uses or has ever used street drugs, or has overused prescription medications, or if you have or have ever had depression or another mental illness. There is a greater risk that you will overuse hydromorphone if you have or have ever had any of these conditions. Talk to your health care provider immediately and ask for guidance if you think that you have an opioid addiction or call the U.S. Substance Abuse and Mental Health Services Administration (SAMHSA) National Helpline at 1-800-662-HELP.
Hydromorphone may cause serious or life-threatening breathing problems, especially during the first 24 to 72 hours of your treatment and any time your dose is increased. Your doctor will monitor you carefully during your treatment. Tell your doctor if you have slowed breathing or have or have ever had asthma. Your doctor will probably tell you not to take hydromorphone. Also tell your doctor if you have or have ever had lung disease such as chronic obstructive pulmonary disease (a group of diseases that affect the lungs and airways), a head injury or any condition that increases the pressure in your brain. or kyphoscoliosis (curving of the spine that may cause breathing problems). The risk that you will develop breathing problems may be higher if you are an older adult, or are weakened or malnourished due to disease. If you experience any of the following symptoms, call your doctor immediately or get emergency medical treatment: slowed breathing, long pauses between breaths, or shortness of breath.
Taking certain medications during your treatment with hydromorphone may increase the risk that you will develop serious or life-threatening breathing problems, sedation, or coma. Tell your doctor if you are taking or plan to take any of the following medications: benzodiazepines such as alprazolam (Xanax), chlordiazepoxide (Librium), clonazepam (Klonopin), diazepam (Diastat, Valium), estazolam, flurazepam, lorazepam (Ativan), oxazepam, temazepam (Restoril), and triazolam (Halcion); medications for mental illness or nausea; muscle relaxants; other narcotic pain medications; sedatives; sleeping pills; or tranquilizers. Your doctor may need to change the doses of your medications and will monitor you carefully. If you use hydromorphone with any of these medications and you develop any of the following symptoms, call your doctor immediately or seek emergency medical care: unusual dizziness, lightheadedness, extreme sleepiness, slowed or difficult breathing, or unresponsiveness. Be sure that your caregiver or family members know which symptoms may be serious so they can call the doctor or emergency medical care if you are unable to seek treatment on your own.
Drinking alcohol, taking prescription or nonprescription medications that contain alcohol, or using street drugs during your treatment with hydromorphone increases the risk that you will experience serious, life-threatening side effects. Do not drink alcohol, take prescription or nonprescription medications that contain alcohol, or use street drugs during your treatment.
Swallow the extended-release tablets whole. Do not split, chew, dissolve, or crush them. If you swallow broken, chewed, crushed, or dissolved tablets you may receive too much hydromorphone at once instead of receiving the medication slowly over time. This may cause serious breathing problems or death.
Do not allow anyone else to take your medication. Hydromorphone may harm or cause death to other people who take your medication, especially children. Keep hydromorphone in a safe place so that no one else can take it accidentally or on purpose. Be especially careful to keep hydromorphone out of the reach of children. Keep track of how many tablets or how much liquid is left so you will know if any medication is missing. Dispose of unwanted or no longer needed tablets, extended-release tablets, and liquid by flushing the medication down the toilet.
Tell your doctor if you are pregnant or plan to become pregnant. If you take hydromorphone regularly during your pregnancy, your baby may experience life-threatening withdrawal symptoms after birth. Tell your baby’s doctor right away if your baby experiences any of the following symptoms: irritability, hyperactivity, abnormal sleep, high-pitched cry, uncontrollable shaking of a part of the body, vomiting, diarrhea, or failure to gain weight.
If you are taking hydromorphone extended-release tablets, your doctor or pharmacist will give you the manufacturer’s patient information sheet (Medication Guide) when you begin your treatment and each time you fill your prescription. Read the information carefully and ask your doctor or pharmacist if you have any questions. You can also visit the Food and Drug Administration (FDA) website (https://www.fda.gov/Drugs/DrugSafety/ucm085729.htm) or the manufacturer’s website to obtain the Medication Guide.
Talk to your doctor about the risks of taking hydromorphone.
Hydromorphone precautions
Before taking hydromorphone
You should NOT take hydromorphone if you have ever had an allergic reaction to hydromorphone or other narcotic medicines, or if you have:
- severe asthma or breathing problems;
- a blockage in your stomach or intestines; or
- a bowel obstruction called paralytic ileus.
Do not use hydromorphone if you have used an MAO (monoamine oxidase) inhibitor in the past 14 days. A dangerous drug interaction could occur. MAO inhibitors include isocarboxazid, linezolid, methylene blue injection, phenelzine, rasagiline, selegiline, tranylcypromine, and others.
Tell your doctor if you have ever had:
- a head injury, brain tumor, or seizures;
- alcoholism, drug addiction, or mental illness;
- urination problems;
- liver or kidney disease;
- a sulfite allergy;
- problems with your gallbladder, pancreas, or thyroid
Tell your doctor and pharmacist what prescription and nonprescription medications, vitamins, nutritional supplements, and herbal products you are taking or plan to take. Be sure to mention the medications listed in the IMPORTANT WARNING section and any of the following: buprenorphine (Buprenex, Butrans, in Suboxone, in Zubsolv, others); butorphanol; cyclobenzaprine (Amrix); dextromethorphan (found in many cough medications; in Nuedexta); ipratropium (Atrovent); medications for glaucoma, irritable bowel disease, Parkinson’s disease, ulcers, and urinary problems; lithium (Lithobid); medications for migraine headaches such as almotriptan (Axert), eletriptan (Relpax), frovatriptan (Frova), naratriptan (Amerge), rizatriptan (Maxalt), sumatriptan (Alsuma, Imitrex, in Treximet), and zolmitriptan (Zomig); mirtazapine (Remeron); nalbuphine; pentazocine (Talwin); 5HT3 serotonin blockers such as alosetron (Lotronex), dolasetron (Anzemet), granisetron (Kytril), ondansetron (Zofran, Zuplenz), or palonosetron (Aloxi); selective serotonin-reuptake inhibitors such as citalopram (Celexa), escitalopram (Lexapro), fluoxetine (Prozac, Sarafem, in Symbyax), fluvoxamine (Luvox), paroxetine (Brisdelle, Prozac, Pexeva), and sertraline (Zoloft); serotonin and norepinephrine reuptake inhibitors such as desvenlafaxine (Khedezla, Pristiq), duloxetine (Cymbalta), milnacipran (Savella), and venlafaxine (Effexor); trazodone (Oleptro); and tricyclic antidepressants (‘mood elevators’) such as amitriptyline, clomipramine (Anafranil), desipramine (Norpramin), doxepin (Silenor), imipramine (Tofranil), nortriptyline (Pamelor), protriptyline (Vivactil), and trimipramine (Surmontil). Also tell your doctor or pharmacist if you are taking or receiving any of the following monoamine oxidase (MAO) inhibitors or have stopped taking them within the past 2 weeks: isocarboxazid (Marplan), linezolid (Zyvox), methylene blue, phenelzine (Nardil), selegiline (Eldepryl, Emsam, Zelapar), or tranylcypromine (Parnate). Many other medications may also interact with hydromorphone, so be sure to tell your doctor about all the medications you are taking, even those that do not appear on this list. Your doctor may need to change the doses of your medications or monitor you carefully for side effects.
If you use opioid medicine while you are pregnant, your baby could become dependent on the drug. This can cause life-threatening withdrawal symptoms in the baby after it is born. Babies born dependent on opioids may need medical treatment for several weeks.
Do not breast-feed. Hydromorphone can pass into breast milk and may cause drowsiness or breathing problems in a nursing baby. Neonatal withdrawal syndrome occurs in the neonates of women with chronic hydromorphone usage. For this purpose, prolonged treatment of hydromorphone is to be avoided during pregnancy. Whenever neonatal withdrawal syndrome is expected, adequate management should be available, which includes proper administration of Morphine to the neonate.
If you will be taking the extended-release tablets, also tell your doctor if you have ever had surgery that caused a change in the way food moves through your stomach or intestines or if you have any condition that cause narrowing of the esophagus (tube that carries food from the mouth to the stomach), stomach, or intestines such as cystic fibrosis (a condition that causes the body to produce thick, sticky mucus that may clog the pancreas, lungs, and other parts of the body), peritonitis (inflammation of the lining of the abdomen (stomach area), Meckel’s diverticulum (a bulge in the lining of the small intestine that is present at birth), chronic intestinal pseudo-obstruction (condition in which the muscles in the intestine do not move food smoothly through the intestine), or inflammatory bowel disease (IBD) a group of conditions that cause inflammation of the lining of the intestine. Your doctor may tell you not to take hydromorphone extended-release tablets.
What other drugs will affect hydromorphone?
Opioid medication can interact with many other drugs and cause dangerous side effects or death. Be sure your doctor knows if you also use:
- other narcotic medications–opioid pain medicine or prescription cough medicine;
- a sedative like Valium–diazepam, alprazolam, lorazepam, Ativan, Klonopin, Restoril, Tranxene, Versed, Xanax, and others;
- drugs that make you sleepy or slow your breathing–a sleeping pill, muscle relaxer, tranquilizer, antidepressant, or antipsychotic medicine; or
- drugs that affect serotonin levels in your body–a stimulant, or medicine for depression, Parkinson’s disease, migraine headaches, serious infections, or prevention of nausea and vomiting.
This list is not complete. Other drugs may affect hydromorphone, including prescription and over-the-counter medicines, vitamins, and herbal products. Not all possible interactions are listed here.
What should I avoid while using hydromorphone?
Do not drink alcohol. Dangerous side effects or death could occur.
Avoid driving or hazardous activity until you know how hydromorphone will affect you. Dizziness or drowsiness can cause falls, accidents, or severe injuries.
Hydromorphone pharmacology
Immediate-release oral formulations have an onset of action at 15 to 30 minutes, peak at 30 to 60 minutes, and last 3 to 4 hours. Half-life is 2 to 3 hours.
Extended-release formulations have an onset of action at 6 hours, peak at 9 hours, and lasts 13 hours. Half-life is approximately 11 hours but varies between 8 to 15 hours.
The volume of distribution is 4 L/kg, where 8% to 19% of the drug is protein bound. Metabolism occurs in the liver through glucuronidation where most of it is converted to hydromorphone-3-glucuronide.
Excretion is mainly through urine in the glucuronidated form. The residual unchanged form is excreted in the urine (7%) and in the feces (1%).
The equivalence of hydromorphone to other opioid is presented below.
Hydromorphone dosage
Follow the directions on your prescription label and read all medication guides. Never use hydromorphone in larger amounts, or for longer than prescribed. Tell your doctor if you feel an increased urge to take more of this medicine.
Never share opioid medicine with another person, especially someone with a history of drug abuse or addiction. MISUSE CAN CAUSE ADDICTION, OVERDOSE, OR DEATH. Keep the medication in a place where others cannot get to it. Selling or giving away opioid medicine is against the law.
Stop taking all other around-the-clock narcotic pain medications when you start taking hydromorphone.
Swallow the capsule or tablet whole to avoid exposure to a potentially fatal overdose. Do not crush, chew, break, open, or dissolve.
Measure liquid medicine carefully. Use the dosing syringe provided, or use a medicine dose-measuring device (not a kitchen spoon).
Do not stop using hydromorphone suddenly, or you could have unpleasant withdrawal symptoms. Ask your doctor how to safely stop using hydromorphone.
Never crush or break a hydromorphone pill to inhale the powder or mix it into a liquid to inject the drug into your vein. This can cause in death.
Store at room temperature away from moisture, heat, and light. Throw away any unused liquid after 90 days.
Keep track of your medicine. You should be aware if anyone is using it improperly or without a prescription.
Do not keep leftover opioid medication. Just one dose can cause death in someone using this medicine accidentally or improperly. Ask your pharmacist where to locate a drug take-back disposal program. If there is no take-back program, flush the unused medicine down the toilet.
Adult Dose for Pain
The following dosing recommendations can only be considered suggested approaches to what is actually a series of clinical decisions over time; each patient should be managed individually.
ORAL:
- Immediate-release tablets: Initial dose: 2 to 4 mg orally every 4 to 6 hours
- Immediate-release oral liquid: Initial dose: 2.5 to 10 mg orally every 3 to 6 hours
- Maintenance dose: Gradually increase dose if analgesia is inadequate, as tolerance develops, or if pain severity increases.
PARENTERAL: Multiple concentrations are available: Standard: 1 mg/mL, 2 mg/mL, 4 mg/mL
- High Potency (HP): for opioid tolerant patients only and should be used only if the amount of hydromorphone can be delivered accurately: 10 mg/mL
- IM or Subcutaneous: Initial dose: 1 mg to 2 mg IM or subcutaneously ever 2 to 3 hours as needed
- Adjust dose according to the severity of pain, the severity of adverse events, and the patients underlying disease and age.
- IV: Initial dose: 0.2 mg to 1 mg IV; administer slowly over at least 2 to 3 minutes.
- Dose should be titrated to achieve acceptable analgesia and tolerable adverse events.
CONVERSION FROM PRIOR OPIOIDS:
Initial daily dose should be 50% of the 24-hour calculated hydromorphone requirement divided by the number of doses permitted based on dosing interval.
- Dose should be titrated to achieve acceptable analgesia and tolerable adverse events.
- The following may be used as a guide to determine conversion to hydromorphone. A hydromorphone IM or subcutaneous dose of 1.3 to 2 mg is approximately equianalgesic to a hydromorphone oral dose of 6.5 to 7.5 mg.
- Hydromorphone IM or subcutaneous doses of 1.3 to 2 mg are approximately equianalgesic to the following parenteral doses:
- Morphine 10 mg; Oxymorphone 1 to 1.1 mg; Levorphanol 2 to 2.3 mg; Meperidine 75 to 100 mg; Methadone 10 mg; Nalbuphine 10 to 12 mg; Butorphanol 1.5 to 2.5 mg
- Hydromorphone oral doses of 6.5 to 7.5 mg are approximately equianalgesic to the following oral doses:
- Morphine 40 to 60 mg; Oxymorphone 6.6 mg; Levorphanol 4 mg; Meperidine 300 to 400 mg; Methadone 10 to 20 mg
Comments:
- Dose titration should be guided by need for analgesia rather than by dose.
- Due to risk of respiratory depression, Hydromorphone-HP injection is reserved for use in opioid tolerant patients only; opioid tolerant patients are those patients taking for one week or longer, at least: oral morphine 60 mg/day, fentanyl transdermal 25 mcg/hour, oral oxycodone 30 mg/day, oral hydromorphone 8 mg/day, oral oxymorphone 25 mg/day, or an equianalgesic dose of another opioid.
Use: Management of pain in patients where an opioid analgesic is appropriate.
SUPPOSITORY: Insert 3 mg suppository rectally every 6 to 8 hours
Comments: The labeling for this product has not been approved by FDA
Use: For the relief of moderate to severe pain such as that due to surgery, trauma, burns, cancer, biliary colic, myocardial infarction, or renal colic.
Adult Dose for Chronic Pain
The following dosing recommendations can only be considered suggested approaches to what is actually a series of clinical decisions over time; each patient should be managed individually. Discontinue all other around-the-clock opioid drugs when initiating therapy with extended-release hydromorphone.
- Due to risk of respiratory depression, EXTENDED-RELEASE tablets are reserved for use in opioid tolerant patients only; opioid tolerant patients are those patients taking for one week or longer, at least: oral morphine 60 mg/day, fentanyl transdermal 25 mcg/hour, oral oxycodone 30 mg/day, oral hydromorphone 8 mg/day, oral oxymorphone 25 mg/day, or an equianalgesic dose of another opioid.
Initial doses:
Conversion from Immediate-Release Oral Hydromorphone to Extended-Release tablets: Starting dose should be equivalent to the total daily oral immediate-release dose taken as extended-release tablet orally once a day.
Conversion from Other Oral Opioids to Extended-Release tablets: Starting dose should be equivalent to 50% of the calculated daily hydromorphone requirement taken as extended-release tablet orally once a day.
- Published potency tables can be used to estimate a patient’s 24-hour oral hydromorphone requirement, however, if used it is best to underestimate the 24-hour requirement due to substantial inter-patient variability; rescue medication can be provided as the dose is titrated
- Alternatively, the following conversion factors (CFs) may be used to convert selected oral opioids to hydromorphone extended-release tablets: Hydromorphone, CF=1; Codeine, CF=0.06; Hydrocodone, CF=0.4; Oxycodone, CF=0.4; Methadone, CF= 0.6, Morphine, CF=0.2, Oxymorphone, CF=0.6.
- Example: Sum the total daily dose of prior oral opioid; multiply that sum by the CF to obtain 24-hour oral requirement; the starting dose should be 50% of this 24-hour oral requirement, round down if necessary.
- These conversion factors cannot be used to convert from hydromorphone extended-release tablets to oral opioids as doing so will result in overestimation of the opioid dose and may result in fatal respiratory depression.
Conversion from Transdermal Fentanyl to Oral Hydromorphone Extended-Release: Initiate therapy 18 hours following removal of the transdermal fentanyl patch, starting dose should be determined using the conversion: 25 mcg fentanyl patch=12 mg oral extended-release hydromorphone; this 24-hour requirement should then be reduced by 50%, round down if necessary; calculated initial dose taken orally once a day.
TITRATION AND MAINTENANCE:
- Maintenance dose: Individually titrate to a dose that provides adequate analgesia and minimizes adverse reactions; dose adjustments may be made in 4 to 8 mg increments every 24 hours, every 3 to 4 days.
- Breakthrough Pain: If the level of pain increases after dose stabilization, attempt to identify the source before increasing dose; rescue medication with appropriate immediate-release analgesia may be helpful.
Comments:
- When converting from methadone, close monitoring is of particular importance due to methadone’s long half-life.
- Dose conversion should be done carefully and with close monitoring due to large patient variability in regards to opioid analgesic response.
- Extended-release tablets are not indicated as an as-needed analgesic.
- Upon cessation of therapy, gradually taper dose in physically dependent patient.
Use: For the management of pain severe enough to require daily, around-the-clock, long-term opioid treatment and for which alternative treatment options are inadequate.
Renal Dose Adjustments
Closely monitor for respiratory and CNS depression during initiation and dose escalation
Oral:
Immediate-release: Consider use of oral liquid to allow for dose adjustments
- Moderate renal impairment; Start on a lower dose
- Severe renal impairment: Starting dose should be even lower
Extended-release tablets:
- Moderate renal impairment: Initial dose: give 50% of the normal initial dose
- Severe renal impairment: Initial dose: give 25% of the normal initial dose; consider alternative analgesic that may permit more flexibility in dosing.
Parenteral:
- Depending on the extent of impairment: Initial dose should be one-fourth to one-half the usual dose
Liver Dose Adjustments
Closely monitor for respiratory and CNS depression during initiation and dose escalation
Oral:
Immediate-release: Consider use of oral liquid to allow for dose adjustments
- Moderate renal impairment; Start on a lower dose; no specific doses suggested
- Severe renal impairment: Starting dose should be even lower; no specific doses suggested
Extended-release tablets:
- Moderate hepatic impairment: Initial dose: give 25% of the normal initial dose; closely monitor for respiratory and CNS depression during initiation and dose escalation
- Severe hepatic impairment: Alternative analgesics are recommended.
Parenteral:
- Depending on the extent of impairment: Initial dose should be one-fourth to one-half the usual dose
Dose Adjustments
Elderly patients: Closely monitor for respiratory and CNS depression during initiation and dose escalation
In general, opioids used regularly should not be abruptly discontinued.
Cessation of therapy in the opioid-tolerant patient:
- Taper extended-release gradually by 25% to 50% every 2 or 3 days down to a dose of 8 mg per day before discontinuation to prevent signs and symptoms of withdrawal.
What happens if I miss a dose?
If you are taking the tablets or solution, take the missed dose as soon as you remember it. However, if it is almost time for the next dose, skip the missed dose and continue your regular dosing schedule. Do not take a double dose to make up for a missed one.
If you are taking the extended-release tablets, skip the missed dose and continue your regular dosing schedule. Do not take more than one dose of the extended-release tablets in 24 hours.
Hydromorphone side effects
Hydromorphone may cause side effects. Tell your doctor if any of these symptoms are severe or do not go away:
- headache
- difficulty falling asleep or staying asleep
- dry mouth
- lightheadedness
- drowsiness
- heavy sweating
- muscle, back or joint pain
- stomach pain
- anxiety
- flushing
- itching
- depression
Some side effects can be serious. If you experience any of these symptoms or those listed in the IMPORTANT WARNING section, call your doctor immediately or get emergency medical treatment:
- rash
- hives
- swelling of the eyes, face, lips, tongue, mouth, throat, arms, hands, feet, ankles, or lower legs
- difficulty breathing or swallowing
- hoarseness
- agitation, hallucinations (seeing things or hearing voices that do not exist), fever, sweating, confusion, fast heartbeat, shivering, severe muscle stiffness or twitching, loss of coordination, nausea, vomiting, or diarrhea
- nausea, vomiting, loss of appetite, weakness, or dizziness
- inability to get or keep an erection
- irregular menstruation
- decreased sexual desire
- seizures
- chest pain
- extreme drowsiness
- fainting
- lightheadedness when changing positions
Hydromorphone may cause other side effects. Call your doctor if you have any unusual problems while you are taking this medication.
While you are taking hydromorphone, you may be told to always have a rescue medication called naloxone available (e.g., home, office). Naloxone is used to reverse the life-threatening effects of an overdose. It works by blocking the effects of opiates to relieve dangerous symptoms caused by high levels of opiates in the blood. You will probably be unable to treat yourself if you experience an opiate overdose. You should make sure that your family members, caregivers, or the people who spend time with you know how to tell if you are experiencing an overdose, how to use naloxone, and what to do until emergency medical help arrives.
Naloxone (Narcan) is to given immediately in the intravenous, intramuscular, or subcutaneous form. The required dose is 0.4 mg to 2 mg every 2 to 3 minutes when needed, and not to exceed 0.001 mg/kg or 10 mg 1.
Your doctor or pharmacist will show you and your family members how to use the medication. Ask your pharmacist for the instructions or visit the manufacturer’s website to get the instructions. If someone sees that you are experiencing symptoms of an overdose, he or she should give you your first dose of naloxone, call your local emergency services number immediately, and stay with you and watch you closely until emergency medical help arrives. Your symptoms may return within a few minutes after you receive naloxone. If your symptoms return, the person should give you another dose of naloxone. Additional doses may be given every 2 to 3 minutes, if symptoms return before medical help arrives.
Hydromorphone overdose
People who take hydromorphone for pain should not drink alcohol. Combining alcohol with this drug increases the chance for dangerous side effects and overdose symptoms.
In case of overdose, call the poison control helpline at 1-800-222-1222. Information is also available online at (https://www.poisonhelp.org/help). If the victim has collapsed, had a seizure, has trouble breathing, or can’t be awakened, immediately call your local emergency services number.
Symptoms of a hydromorphone overdose include:
- Bluish-colored fingernails and lips
- Breathing problems, including slow and labored breathing, shallow breathing, or no breathing
- Cold, clammy skin
- Coma
- Confusion
- Constipation
- Dizziness
- Drowsiness
- Fatigue
- Flushing of the skin
- Itching
- Lightheadedness
- Loss of consciousness
- Low blood pressure
- Muscle twitches
- Nausea and vomiting
- Pinpoint pupils
- Spasms of the stomach and intestines
- Weakness
- Weak pulse
Warning: A severe overdose of hydromorphone can cause death.
What to expect at the emergency room
Take the container to the hospital with you, if possible.
The health care provider will measure and monitor the person’s vital signs, including temperature, pulse, breathing rate, and blood pressure.
Tests that may be done include:
- Blood and urine tests
- Chest x-ray
- CT scan (computerized tomography or advanced imaging)
- ECG (electrocardiogram, or heart tracing)
Treatment may include:
- Fluids through a vein (by IV)
- Medicine to reverse the effect of the hydromorphone and treat symptoms
- Activated charcoal
- Laxative
- Breathing support, including a tube through the mouth into the lungs and connected to a breathing machine (ventilator)
Hydromorphone overdose prognosis
People who quickly receive medicine (called an antidote) to reverse the effect of hydromorphone can recover within 1 to 4 hours. They may need to stay in the hospital for more doses of the antidote.
Complications such as pneumonia, muscle damage from lying on a hard surface for a long period of time, or brain damage from lack of oxygen may result in permanent disability. However, unless there are complications, long-term effects and death are rare.