Contents
What is mast cell activation syndrome
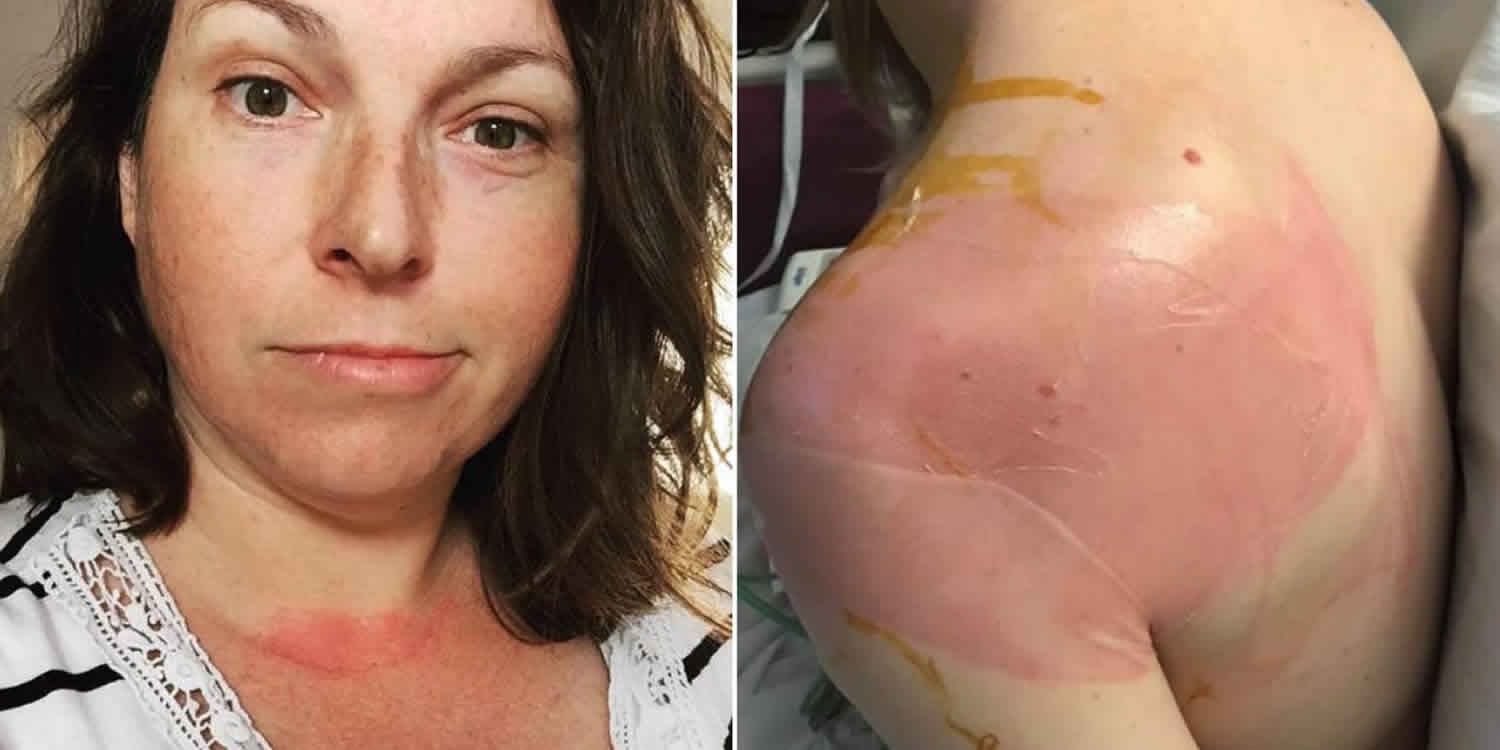
Mast cell activation syndrome is an immunological condition in which mast cells mistakenly release too many chemical mediators, resulting in the patient experiencing repeated episodes of the symptoms of anaphylaxis – allergic symptoms such as hives, swelling, itching, low blood pressure (hypotension), lightheadedness, flushing, difficulty breathing, wheezing, coughing, abdominal pain, cramping, severe diarrhea 1 and potential problems with “brain fog” or other difficulties with memory 2. The mast cell activation syndrome episodes are called “idiopathic” which means that the mechanism is unknown – that is, not caused by allergic antibody or secondary to other known conditions that activate normal mast cells. The onset of mast cell activation syndrome is often sudden, affecting both children and adults, sometimes in family groups, mimicking many other conditions and presenting a wide-range of different symptoms that can be baffling for both the patient and their physician.
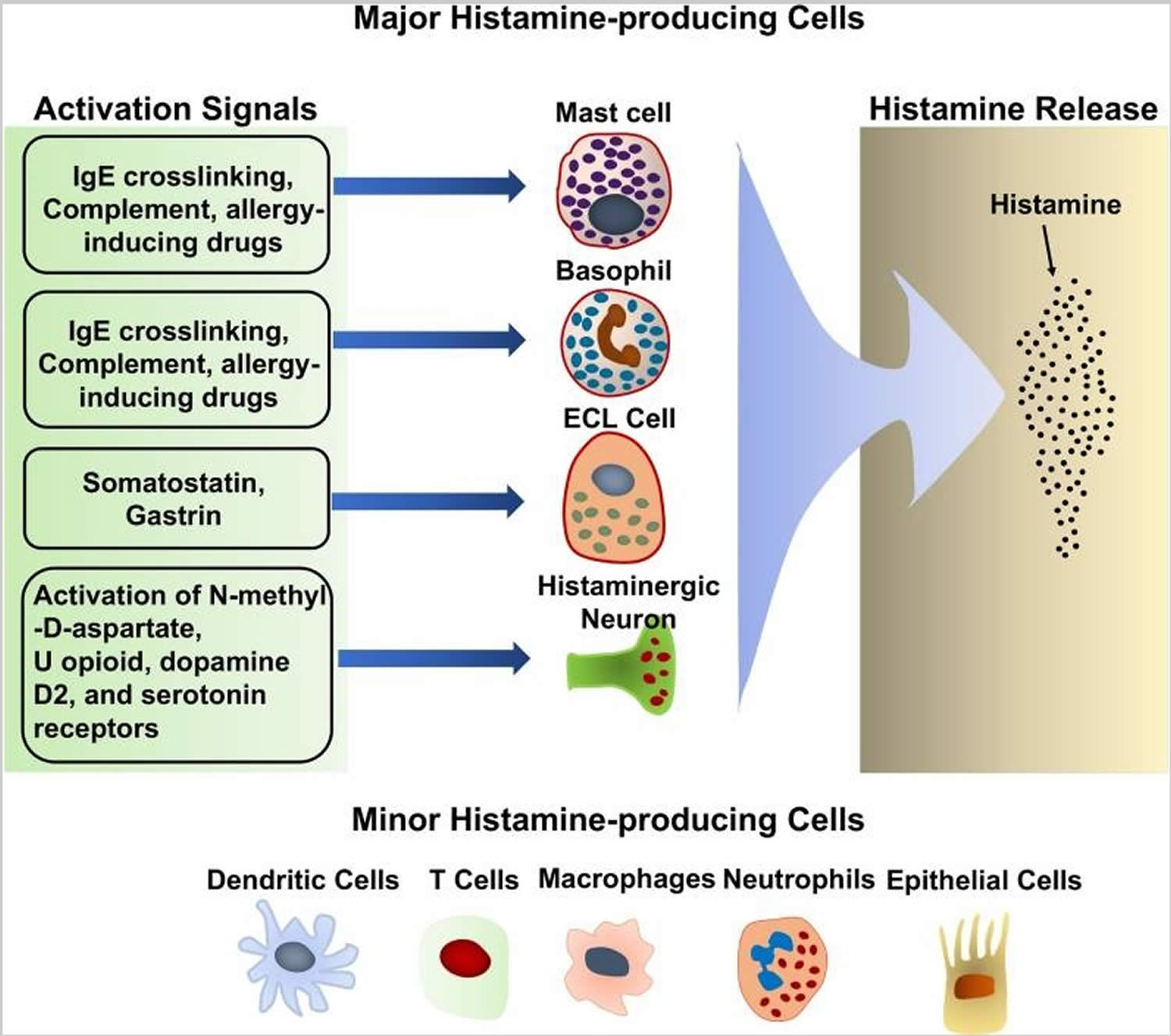
Mast cells are immune cells, a type of blood cell that play an important role in your body’s immune system. Mast cells reside in all your body tissues and form part of your body’s innate immune system. The classic and most well-known function of mast cells involves the inflammatory cascade. The inflammatory effect of the mast cell is used by the innate immune system as a first line of defense. Mast cells react to foreign bodies and injury by releasing a variety of potent chemical mediators, such as histamine, when activated. Histamine induces white blood cell chemotaxis, airway smooth muscle constriction, and increased vascular permeability 3. Other mediators released from the mast cell granules include prostaglandins, leukotrienes, tryptase, chymase, and TNF-alpha. In a healthy person these chemicals will act beneficially to protect and heal the body, but in a person with mast cell activation syndrome these same chemicals are inappropriately triggered and released and have a negative effect on the body. Amongst the triggers are a variety of different foods, exercise, chemicals, fragrances and stress. Many mast cell activation syndrome sufferers struggle to identify their triggers and continue to discover new triggers for many years after diagnosis.
Mast cells also play a role in tissue repair and angiogenesis. Upon injury, mast cells release procoagulant cytokines, leukotrienes, and platelet-activating factor independent of IgE mechanisms. Later, heparin, tryptase, and t-PA from the mast cell modulate blood flow to increase nutrient and immune cell delivery. Inflammatory mediators promote the differentiation and growth of fibroblasts and endothelial cells. Mast cells also possess many angiogenic cytokines, such as VEGF and FGF 2. Finally, mast cells have been implicated in the wound contraction and the regeneration of nerve fibers.
No deficiency in mast cells has ever been noted, implying that the functions of the mast cell are vital for life 3.
The cause of mast cell activation syndrome is unknown. Mast cell activation syndrome forms part of a spectrum of mast cell disorders involving proliferation and/or excessive sensitivity of mast cells, it has been identified since 2007 4. Mast cell activation syndrome features inappropriate mast cell activation with little or no increase in the number of mast cells, unlike in mastocytosis. Mastocytosis involves inappropriate mast cell activation as well as an increased number of mast cells. Mastocytosis is a rare but relatively well known mast cell disorder and is currently easier to diagnose than mast cell activation syndrome.
There is at present no cure for mast cell activation syndrome. However, mast cell activation syndrome episodes respond to treatment with inhibitors or blockers of mast cell mediators. Treatment includes several combinations of medications like anti-histamines and mast cell stabilizers 5.
Figure 1. Histamine-producing cells and stimuli that trigger histamine release
[Source 6]Mast cell activation syndrome symptoms
Mast cell activation syndrome symptoms are:
- Heart related symptoms: rapid pulse (tachycardia), low blood pressure (hypotension) and passing out (syncope).
- Skin related symptoms: itching (pruritus), hives (urticaria), swelling (angioedema) and skin turning red (flushing).
- Lung related symptoms: wheezing, shortness of breath and harsh noise when breathing (stridor) that occurs with throat swelling.
- Gastrointestinal tract symptoms: diarrhea, nausea with vomiting and crampy abdominal pain.
Mast cell activation syndrome diagnosis
Mast cell activation syndrome is difficult to diagnose with great confidence. Patients often spend decades going in circles visiting many different specialists and achieving no proper diagnosis. There are now some doctors who understand the issues and there are tests that can support a diagnosis. Diagnostic criteria were not agreed until as recently as 2011 by an international group of doctors. The criteria rely on a number of factors: typical clinical symptoms, evidence of mediator release and response to medication, as well as a thorough discounting of any other potential diseases.
Current provisional criteria to define mast cell activation syndrome 7
Major criteria
- Constellation of clinical complaints attributable to pathologically increased mast cell activity (mast cell mediator release syndrome)
Minor criteria
- Focal or disseminated increased number of mast cells in marrow and/or extracutaneous organ(s) (e.g., gastrointestinal tract biopsies; CD117-, tryptase-, and CD25-stained)
- Abnormal spindle-shaped morphology in >25 % of mast cells in marrow or other extracutaneous organ(s)
- Abnormal mast cell expression of CD2 and/or CD25 (i.e., co-expression of CD117/CD25 or CD117/CD2)
- Detection of genetic changes in mast cells from the blood, bone marrow, or extracutaneous organs for which an impact on the state of activity of affected mast cells in terms of an increased activity has been proven
- Evidence (typically from body fluids such as whole blood, serum, plasma, or urine) of above-normal levels of mast cell mediators including:
mast cell activation syndrome- Tryptase in blood
- Histamine or its metabolites (e.g., N-methylhistamine) in the urine
- Heparin in the blood
- Chromogranin A in the blood (potential confounders of cardiac or renal failure, neuroendocrine tumors, or recent proton pump inhibitor use were excluded)
- Other relatively mast cell-specific mediators (e.g., eicosanoids including prostaglandin PGD2, its metabolite 11-β-PGF2α, or leukotriene E4)
- Symptomatic response to inhibitors of mast cell activation or mast cell mediator production or action (e.g., histamine H1 and/or H2 receptor antagonists, cromolyn)
Diagnosis of mast cell activation syndrome made by either (1) the major criterion plus any one of the minor criteria or (2) any three minor criteria
8.
Increases in serum mast cell tryptase and in urine levels of N-methylhistamine, 11B -Prostaglandin F2α (11B-PGF2α) and/or Leukotriene E4 (LTE4) are the only useful tests in diagnosis of mast cell activation syndrome 1. Total serum mast cell tryptase should be drawn between 30 minutes and two hours after the start of an episode, with baseline level obtained many days later. The urine tests are performed on a 24 hour collection of urine that is started immediately. Since these are not standard laboratory tests, patients should work with their local allergist who can communicate with emergency and lab personnel to assure they are ordered and completed in a timely fashion.
The first challenge of suspected mast cell activation syndrome sufferers is to find a doctor who has the time and experience to assimilate a complicated medical history. Note that there is no one definitive test for mast cell activation syndrome. Tryptase is reported by many doctors in the published literature to be an unreliable marker for mast cell activation syndrome. Recognizing this, tests for other mediators such as histamine and prostaglandin have recently become available. Other tests can also be used to support a diagnosis. Note that these tests are not definitive, capturing and measuring these chemicals is extremely challenging and if positive other potential causes have to be discounted. A doctor that will listen carefully to the wide range of symptoms and ask themselves what could be the unifying factor, is key. At present there are few doctors that have developed the clinical experience to feel confident in making a diagnosis and consequently mast cell activation syndrome patients are often invisible to the health system. In the USA and Germany, clinical tests for a range of mediators have been available for years and there is a great deal of clinical experience and indeed published literature on the subject. Though these tests are not in themselves complete proof of mast cell involvement, when taken with the criteria, they provide reasonable confidence in a diagnosis.
The patient’s blood should be tested for mutation of mast cell growth receptor KIT, called KIT D816V. If positive, it indicates a clonal mast cell disorder. A negative blood test for KIT D816V is helpful but not 100% accurate, so one of several scoring systems should be used, to follow symptoms and lab results to determine if the presentation is consistent with a clonal mast cell disorder. If so, a bone marrow biopsy and aspirate is indicated. The biopsy offers a high level of ability (sensitivity) to find KIT D816V mutation and allows examining bone marrow mast cells for their shape and abnormal cell surface markers. If the bone marrow biopsy is negative for abnormal and clonal mast cells, it establishes the diagnosis of idiopathic mast cell activation syndrome.
Mast cell activation syndrome treatment
The goals of treatment are both diagnostic and patient relief. Lack of response to these treatments suggests that mast cell activation syndrome is not present. The available treatments for mast cell activation syndrome stabilize the mast cells and mitigate the effects of the chemicals they release, e.g. anti-histamines and mast cell stabilizers. Avoiding triggers is also a key part in coping with mast cell activation syndrome.
There is a wide range in the variety of patients’ response to treatment. It can often take some time to work out what the best medication and dosage are. An added complication is that many patients suffer adverse reactions to the drugs themselves or to the fillers, coloring and preservatives. With a trial and error approach many patients are successful in moderating their symptoms although quality of life can still be affected.
The treatment of acute episodes should follow the recommendations for treatment of anaphylaxis, starting with epinephrine (adrenaline), if indicated by the severity of symptoms.
Antihistamines, such as the first generation histamine type 1 receptor blockers diphenhydramine and hydroxyzine, can be effective for itching, abdominal discomfort and flushing, but their use may be limited by side effects (sleepiness). Second generation antihistamines, including loratadine, cetirizine and fexofenadine, are preferable due to fewer side effects.
Treatment with histamine type 2 receptor blockers, such as ranitidine or famotidine, can be helpful for abdominal pain and nausea.
Aspirin blocks production of prostaglandin D2 and can reduce flushing.
Montelukast and zafirlukast block the effects of leukotriene C4 (LTC4) and zileuton blocks LTC4 production, so these reduce wheezing and abdominal cramping.
Corticosteroids are helpful for edema, hives and wheezing but should only be used as a last resort.
Omalizumab (which blocks binding of IgE antibody to its receptors) has been reported to reduce mast cell reactivity and sensitivity to activation which can reduce anaphylactic episodes. Prospective randomized studies should confirm these encouraging results and allow more patients to benefit from this effective and safe treatment option.
Mast cell activation syndrome diet
Histamine diet
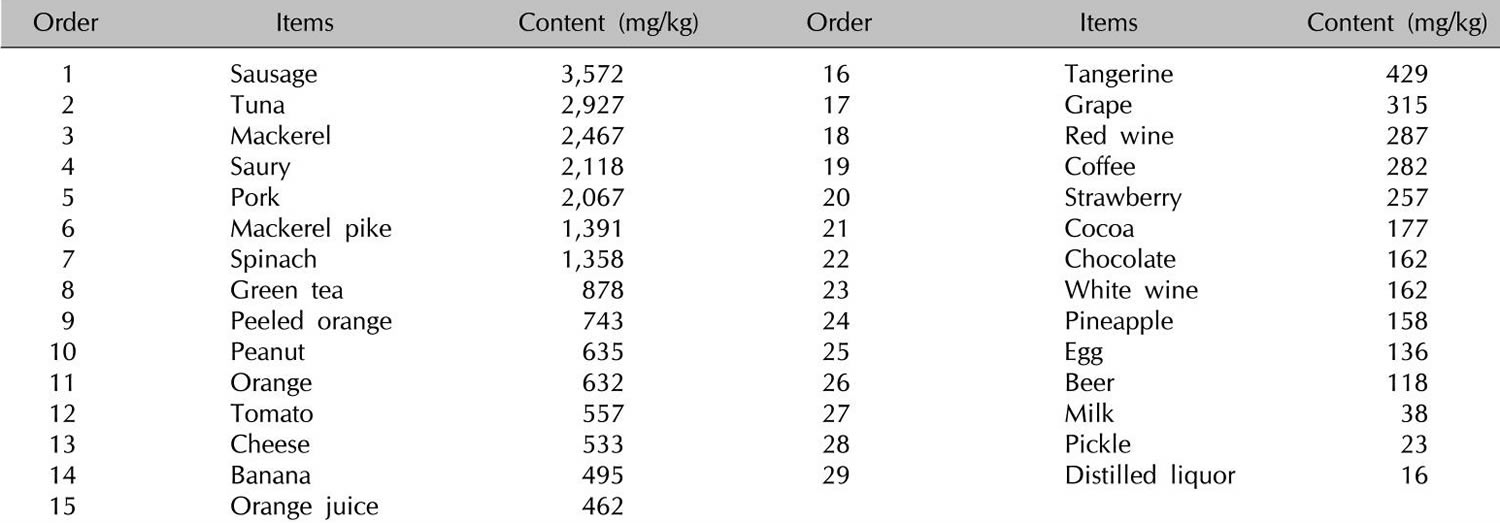
Histamine and other biogenic amines are present to various degrees in many foods, and their presence increases with maturation 9. The formation of biogenic amines in food requires the availability of free amino acids, the presence of decarboxylase-positive microorganisms, and conditions allowing bacterial growth and decarboxylase activity. Free amino acids either occur as such in foods or may be liberated by proteolysis during processing or storage 10. Numerous bacteria and some yeasts display high L-histidine decarboxylase activity and thus have the capacity to form histamine. Histidine is generated from autolytic or bacterial processes 11. Therefore, high concentrations of histamine are found mainly in products of microbial fermentation, such as aged cheese 12, sauerkraut, wine 13, and processed meat 14 (see Tables 3 and 4 below) or in microbially spoiled food. Thus, histamine, tyramine, putrescine, and cadaverine serve as indicators of hygienic food quality 10. Tyramine and putrescine also may lead to intolerance reactions in combination with histamine. Possible explanations may be the inhibition of diamine oxidase (DAO) by other amines 15 or the promotion of histamine liberation from the mucosa by putrescine 16.
Intolerance of tyramine that has vasoconstrictive properties that lead to hypertensive crisis and headache has been known mostly in patients taking monoamine oxidase (MAO)–inhibiting drugs. Orally administered tyramine in doses of 200 to 800 mg has been shown to increase systolic blood pressure by 30 mm Hg in otherwise unmedicated subjects. Conversely, in patients taking MAO-inhibiting drugs, the pressor sensitivity was 7- to 56-fold that in patients not taking MAO-inhibiting drugs 17. Eight double-blind, placebo-controlled studies have investigated the effect of tyramine on migraine. Two studies showed positive results in migraine patients who were sensitive to foods that are high in tyramine (n = 45) (19) or who had wine-provoked migraine (n = 19) 18; 6 studies showed negative results with 97 19 patients. The 2 positive studies and 2 of the negative studies were regarded as inconclusive 20 because of a lack of randomization 17, questionable blinding 18, or inappropriate selection of migraine patients without a history of suspected tyramine intolerance 21. Conversely, in 2 conclusive studies of migraine patients with a positive or negative dietary history, 125 mg oral tyramine did not precipitate more headaches than did placebo.
Histamine foods
Alcohol, especially red wine, is rich in histamine and is a potent inhibitor of diamine oxidase (DAO) 22. The relation between the ingestion of wine, an increase in plasma histamine, and the occurrence of sneezing, flushing, headache, asthma attacks, and other anaphylactoid reactions and a reduction of symptoms by antihistamines has been shown in various studies 23. However, among the multitude of substances contained in wine, other biogenic amines such as tyramine 18 and sulfites 24 have been supposed to contribute to symptoms summarized as “wine intolerance” or “red wine asthma” 24. In double-blind, placebo-controlled wine tests with healthy persons 25 and in patients with chronic urticaria and wine intolerance 26, the histamine content did not influence wine tolerance. In the latter group, an increase in plasma histamine could be shown, paradoxically, after ingestion of the histamine-poor wine. In these patients, the ethanol metabolite acetaldehyde has been discussed as a histamine-releasing substance 26. However, the high percentage of responses to the placebo (87%) could be responsible for the absence of an effect in this study 20. Another randomized double-blind, placebo-controlled oral wine challenge in patients with a history of red wine–provoked asthma (n = 18) found no relation between wine tolerance and the wine’s content of histamine or other amines but did find a greater bronchoconstrictive response to wine with a high sulfite content 24. Sulfiting agents are widely used as antioxidants and preservatives in foods, beverages, and pharmaceuticals. Adverse reactions with a presumed relation to sulfites include anaphylactic shock, bronchospasm, urticaria, angioedema, nausea, abdominal pain, diarrhea, stroke, and death 27. Sulfite hypersensitivity has been reported mainly in patients with chronic asthma; the estimated prevalence is 5–10% in all patients 28. Asthmatic reactions have been attributed to reflex activation of the parasympathetic system by the irritating effect of sulfites, possibly enhanced by a deficiency of sulfite oxidase. Besides this pseudoallergic mechanism, in at least some cases of sulfite hypersensitity, an immunoglobulin E (IgE)–mediated immediate-type allergic reaction must be considered 29. Sulfites may be contained in wine, but they are also contained in foods that are poor in histamine, such as fruit juice, frozen vegetables, and lettuce. Thus, in patients reporting intolerance to wine, a careful history of reactions to other foods rich in histamine or sulfites should be taken. In patients who are suspected of having sulfite intolerance, skin testing and a double-blind, placebo-controlled challenge with capsules containing increasing doses of bisulfite or placebo should be performed.
In contrast to an IgE–mediated food allergy, in which the ingestion of even a small amount of the allergen elicits symptoms, in histamine intolerance, the cumulative amount of histamine is crucial. Besides variations in the amount of histamine in food according to storage and maturation, the quantity consumed, the presence of other biogenic amines, and the additional intake of alcohol or diamine oxidase (DAO)-blocking drugs are pivotal factors in the tolerance of the ingested food. Generally, an upper limit of 100 mg histamine/kg in foods and of 2 mg histamine/L in alcoholic beverages has been suggested 30. This threshold may be too high, considering the occurrence of histamine-mediated symptoms after oral ingestion of 75 mg histamine in 5 of 10 females without a history of histamine intolerance 31.
However, most of the positive studies for intolerant reactions to sulfite, histamine, and other biogenic amines do not fulfill the current scientific criteria for providing substantiated evidence of the clinical effect of these foods. Nevertheless, patients who have a conclusive history of adverse reactions to food, alcohol, drugs containing histamine, other biogenic amines, and sulfite but without proof of IgE exist. In such patients, a double-blind, placebo-controlled provocation of the suspected causal agents under close supervision by experienced specialists should be performed after exclusion of other causal diseases and informed consent of the patients—if the provocation is not unreasonably hazardous, considering the grade of the anaphylactoid reaction. Because of the great effort, time, and costs or because of patients’ fear of a repeated reaction, double-blind, placebo-controlled provocations often are not performed in clinical practice, even when they are indicated.
Table 1. High histamine foods
| Food categories | Histamine | Recommended upper limit for histamine | Tyramine | |||
|---|---|---|---|---|---|---|
| mg/kg | mg/L | mg/kg | mg/L | mg/kg | mg/L | |
| Fish (frozen/smoked or salted/canned) | 200 | ND | ||||
| Mackerel | 1–20/1–1788/ND–210 | |||||
| Herring | 1–4/5–121/1–479 | |||||
| Sardine | ND/14–150/3–2000 | |||||
| Tuna | ND/ND/1–402 | |||||
| Cheese | No recommendation | |||||
| Gouda | 10–900 | 10–900 | ||||
| Camembert | 0–1000 | 0–4000 | ||||
| Cheddar | 0–2100 | 0–1500 | ||||
| Emmental | 5–2500 | 0–700 | ||||
| Swiss | 4–2500 | 0–700 | ||||
| Parmesan | 10–581 | 0–840 | ||||
| Meat | No recommendation | |||||
| Fermented sausage | ND–650 | ND–1237 | ||||
| Salami | 1–654 | – | ||||
| Fermented ham | 38–271 | 123–618 | ||||
| Vegetables | ||||||
| Sauerkraut | 0–229 | 10 | 2–951 | |||
| Spinach | 30–60 | |||||
| Eggplant | 26 | |||||
| Tomato ketchup | 22 | |||||
| Red wine vinegar | 4 | |||||
| Alcohol | ||||||
| White wine | ND–10 | 2 | 1–8 | |||
| Red wine | ND–30 | 2 | ND–25 | |||
| Top-fermented beer | ND–14 | 1.1–36.4 | ||||
| Bottom-fermented beer | ND–17 | 0.5–46.8 | ||||
| Champagne | 670 | |||||
Abbreviation: ND = not detected
[Source 32]Table 2. Histamine food list
[Source 33]In addition to histamine-rich food, many foods such as citrus foods are considered to have the capacity to release histamine directly from tissue mast cells, even if they themselves contain only small amounts of histamine (Table 5). In vitro studies of persons with a history of pseudoallergic reactions to food have shown a fragility of duodenal mast cells with massive degranulation in the presence of histamine-releasing substances that is significantly greater than that shown by control subjects 34. However, clinical studies using oral challenge tests to support the hypothesis for the histamine-releasing capacity of foods are required 35.
Table 3. Foods with suggested histamine-releasing capacities
| Plant-derived | Animal-derived | Other |
|---|---|---|
| Citrus fruit | Fish | Additives |
| Papaya | Crustaceans | Liquorice |
| Strawberries | Pork | Spices |
| Pineapple | Egg white | |
| Nuts | ||
| Peanuts | ||
| Tomatoes | ||
| Spinach | ||
| Chocolate |
- Mast Cell Activation Syndrome (MCAS). https://www.aaaai.org/conditions-and-treatments/related-conditions/mcas[↩][↩]
- Theoharides TC, Valent P, and Akin C. Mast Cells, Mastocytosis, and Related Disorders. N Engl J Med. July 9, 2015; 373(2):163-172. https://www.ncbi.nlm.nih.gov/pubmed/26154789[↩]
- Fong M, Crane JS. Histology, Mast Cells. [Updated 2018 Oct 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499904[↩][↩]
- What is Mast Cell Activation Syndrome? https://www.mastcellaction.org/about-mcas[↩]
- The Mast Cell Activation Syndrome: A Mini Review. MOJ Immunol. 2014; 2(1). https://medcraveonline.com/MOJI/MOJI-02-00032.pdf[↩]
- Huang H, Li Y, Liang J, Finkelman FD. Molecular Regulation of Histamine Synthesis. Frontiers in Immunology. 2018;9:1392. doi:10.3389/fimmu.2018.01392. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6019440/[↩]
- Afrin LB, Molderings GJ (2014) A concise, practical guide to diagnostic assessment for mast cell activation disease. World J Hematol 3:1–17[↩]
- Molderings, G.J., Haenisch, B., Brettner, S. et al. Naunyn-Schmiedeberg’s Arch Pharmacol (2016) 389: 671. https://doi.org/10.1007/s00210-016-1247-1[↩]
- Bodmer S, Imark C, Kneubuhl M. Biogenic amines in foods: histamine and food processing. Inflamm Res 1999;48:296–300.[↩]
- Sarkadi L. Histamine in food. In: Falus A, Grosman N, Darvas Z. eds. Histamine: biology and medical aspects. Budapest, Hungary: SpringMed Publishing, 2004:176–85.[↩][↩]
- Bover-Cid S, Holzapfel W. Biogenic amine production by bacteria. In: Morgan D, White A, Sánchez-Jiménez F, Bardócz S. eds. Biogenically active amines in food. Luxembourg City, Luxembourg: EC Publication, 2000:20–9.[↩]
- Beutling DM. Biogene Amine in der Ernährung. (Biogenic amines in nutrition.) Berlin, Germany: Springer, 1996[↩]
- Pechanek U, Pfannhauser W, Woidich H. [Content of biogenic amines in four food groups of the Austrian marketplace]. Z Lebensm Unters Forsch 1983;176:335–40[↩]
- Nordic Council of Ministers Present status of biogenic amines in foods in Nordic countries. Tema Nord 2002: 524 (ISBN: 92-893-0773-0). Cited by: Sarkadi L. Histamine in food. In: Falus A, Grosman N, Darvas Z. eds. Histamine: biology and medical aspects. Budapest, Hungary: SpringMed Publishing, 2004:176–85.[↩]
- Sattler J, Lorenz W, Kubo K, Schmal A, Sauer S, Luben L. Food-induced histaminosis under diamine oxidase (DAO) blockade in pigs: further evidence of the key role of elevated plasma histamine levels as demonstrated by successful prophylaxis with antihistamines. Agents Actions 1989;27:212–4.[↩]
- Backhaus B, Raithel M, Hahn EG. Nicht-immunologisch induzierte Histaminfreisetzung an vitalen menschlichen Darmschleimhautbiopsien durch Stimulation mit Polyaminen. (Nonimmunologically induced histamine release of biopsies of vital human intestinal mucosa after stimulation with polyamines.) Allergo J 2005;14:41[↩]
- Bieck PR, Antonin KH. Oral tyramine pressor test and the safety of monoamine oxidase inhibitor drugs: comparison of brofaromine and tranylcypromine in healthy subjects. J Clin Psychopharmacol 1988;8:237–45.[↩][↩]
- Littlewood JT, Gibb C, Glover V, Sandler M, Davies PT, Rose FC. Red wine as a cause of migraine. Lancet 1988;1:558–9.[↩][↩][↩]
- Forsythe WI, Redmond A. Two controlled trials of tyramine in children with migraine. Dev Med Child Neurol 1974;16:794–9.[↩]
- Jansen SC, van DM, Bottema KC, Dubois AE. Intolerance to dietary biogenic amines: a review. Ann Allergy Asthma Immunol 2003;91:233–40.[↩][↩]
- Ziegler DK, Stewart R. Failure of tyramine to induce migraine. Neurology 1977;27:725–6.[↩]
- Izquierdo-Pulido, M. Biogenic amines in European beers. J Agric Food Chem 1996;44:33159–63.[↩]
- Wantke F, Hemmer W, Gotz M, Jarisch R. Adverse reactions to alcoholic beverages: a diagnostic guideline. Clin Exp Allergy 1997;27:343[↩]
- Dahl R, Henriksen JM, Harving H. Red wine asthma: a controlled challenge study. J Allergy Clin Immunol 1986;78:1126–9.[↩][↩][↩]
- Kanny G, Bauza T, Fremont S, et al. .Histamine content does not influence the tolerance of wine in normal subjects. Allerg Immunol (Paris) 1999;31:45–8.[↩]
- Kanny G, Gerbaux V, Olszewski A, et al. .No correlation between wine intolerance and histamine content of wine. J Allergy Clin Immunol 2001;107:375–8.[↩][↩]
- Yang WH, Purchase EC. Adverse reactions to sulfites. CMAJ 1985;133:865–7, 880.[↩]
- Gunnison AF, Jacobsen DW. Sulfite hypersensitivity. A critical review. CRC Crit Rev Toxicol 1987;17:185–214.[↩]
- Przybilla B, Ring J. [Sulfite hypersensitivity]. Hautarzt 1987;38:445–8[↩]
- Brink B, Damink C., Joosten HM, Huisin’t Veld JH. Occurrence and formation of biologically active amines in foods Int J Food Microbiol 1990;11:73–84.[↩]
- Wohrl S, Hemmer W, Focke M, Rappersberger K, Jarisch R. Histamine intolerance-like symptoms in healthy volunteers after oral provocation with liquid histamine. Allergy Asthma Proc 2004;25:305–11.[↩]
- Laura Maintz, Natalija Novak; Histamine and histamine intolerance, The American Journal of Clinical Nutrition, Volume 85, Issue 5, 1 May 2007, Pages 1185–1196, https://doi.org/10.1093/ajcn/85.5.1185[↩][↩]
- Chung BY, Cho SI, Ahn IS, et al. Treatment of Atopic Dermatitis with a Low-histamine Diet. Annals of Dermatology. 2011;23(Suppl 1):S91-S95. doi:10.5021/ad.2011.23.S1.S91. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3199434/[↩]
- Moneret-Vautrin DA, de Korwin JD, Tisserant J, Grignon M, Claudot N. Ultrastructural study of the mast cells of the human duodenal mucosa. Clin Allergy 1984;14:471–81.[↩]
- Vlieg-Boerstra BJ, van der HS, Oude Elberink JN, Kluin-Nelemans JC, Dubois AE. Mastocytosis and adverse reactions to biogenic amines and histamine-releasing foods: what is the evidence? Neth J Med 2005;63:244–9.[↩]