Contents
What is mucus
Mucus is a thick, slimy secretion that is essential for many biological functions including lubrication, hydration, and protection of the underlying epithelia 1. Mucus is a complex biological material that lubricates and protects the human lungs, gastrointestinal (GI) tract, vagina, eyes, and other moist mucosal surfaces. Mucus is composed of water (∼95%), salts, lipids, and proteins, but its viscous and gel-like properties are largely governed by O-glycoproteins named gel-forming/gelling mucins 2. Mucus serves as a physical barrier against foreign particles, including toxins, pathogens, and environmental ultrafine particles, while allowing rapid passage of selected gases, ions, nutrients, and many proteins 1.
The function of mucus varies between different organs. At exposed surfaces, such as those of the gastrointestinal tract, airways, female reproductive tract, and eyes, mucus acts as the outermost line of protection against foreign pathogens 3, toxins 4, and environmental ultrafine particles 5. In the gastrointestinal (GI) tract, mucus also aids the transport of chyme (partially digested food) from the gut to the colon by serving as a lubricant during the peristaltic process while allowing rapid entry and exit of nutrients and waste 6. At the surfaces of internal organs, mucus serves as a lubricant to minimize friction between organs. In performing its numerous functions, mucus is continuously secreted, shed, and finally digested, recycled, or discarded.
The intestinal mucosa contains billions of commensal bacteria, which represent a permanent challenge to the integrity of the epithelial surface 7. However, commensal bacteria compete for nutrients and sites of epithelial adherence with unwanted bacteria, protecting the underlying epithelium from penetration by pathogenic bacteria 8. Modifications to the mucus properties can greatly affect mucus layer functioning. For example, an intestinal mucus layer that is too thin, as found in inflammatory bowel diseases (IBDs), will facilitate bacteria reaching the epithelial cells, which may trigger inflammation because of the dysregulated immune response to host intestinal microbiota. This has been demonstrated in several mouse models with a defective mucus layer, which leads to direct contact between bacteria and the epithelium associated with a severe intestinal inflammation 9. Conversely, thick mucus in the lungs makes it difficult to expel, leading to lung obstruction as found in cystic fibrosis (CF) and chronic obstructive pulmonary disease (COPD). Thickened secretions are also features in the gastrointestinal system in cystic fibrosis (CF) 10. Meconium ileus and distal intestinal obstruction syndrome develop when thick mucous secretions occlude the hollow gastrointestinal lumen. In the pancreas and bile ducts of cystic fibrosis patients, thickened secretions may cause obstruction and acute and chronic inflammation. Strategies aimed at modulating mucus properties in vivo are limited, partly owing to the complexity of mucin macromolecules.
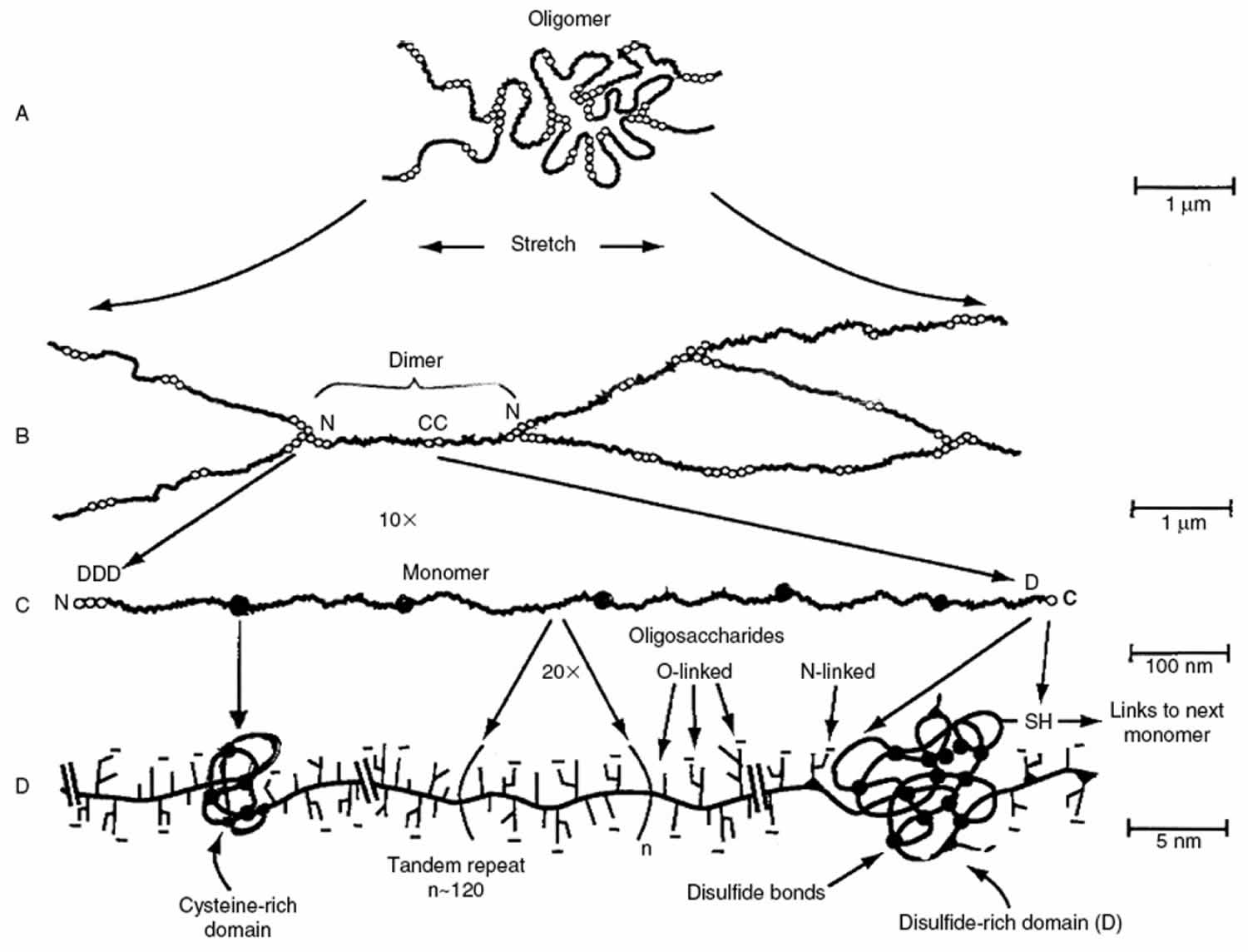
Figure 1. Mucus (biochemical features of gel-forming mucins)
Footnote: Major biochemical features of gel-forming mucins. (A) Several mucin monomers are shown linked together in an oligomeric gel. (B) Mucin monomers are crosslinked end-to-end via disulfide bonds between disulfide-rich domains (labeled “D”) near the amino- and carboxyl-termini 11. (C) Interspersed along each fiber are “naked” globular protein regions, with small exposed hydrophobic patches [198]. These regions are stabilized by multiple disulfide bonds. (D) Individual mucin fibers are densely glycosylated with O- and N-linked glycans, most of which are negatively charged with sialic acids or sulfate groups 11.
[Source 6 ]Mucus is composed primarily of crosslinked, bundled, and entangled mucin fibers secreted by both goblet cells and the seromucinous glands of the lamina propria at the apical epithelium (Figure 1) 12. Mucin fibers, typically 10–40 million Da in size and 3–10 nm in diameter 13, are proteins glycosylated via proline, threonine, and/or serine residues by O-linked N-acetyl galactosamine as well as N-linked sulfate-bearing glycans 14. Glycan coverage of mucins is dense, with 25–30 carbohydrate chains per 100 amino acid residues 15, and contributes up to 80% of the dry weight of mucus 16. Most mucin glycoproteins have a high sialic acid and sulfate content, which leads to a strongly negative surface that increases the rigidity of the polymer via charge repulsion 13. Sialomucin content is suggested to be highly correlated to mucus viscosity and elasticity 17.
At the chemical level, mucus is an integrated structure of biopolymers. Its physical behavior is complex (non-Newtonian), with highly variable properties that are between those of a viscous liquid and an elastic solid. Rheological measurements, including viscosity (resistance to flow) and elasticity (stiffness), are often used together to describe the consistency of mucus. The rheological properties of mucus vary as a function of shear stress, time scale (rate) of shearing, and length scale. Changes in the rheological properties of mucus may greatly affect its ability to function as a lubricant, selective barrier, and the body’s first line of defense against infection 18. If mucus becomes too thick, for example in severe bronchitis 19 or cystic fibrosis 20 where the sputum viscosity can be more than 100,000 times that of water, patients experience great difficulty in mucus clearance, resulting in bacterial overgrowth. On the other hand, in women with bacterial vaginosis, the viscosity of vaginal fluids is significantly lower than in those with normal flora, which may be responsible for the increased risk of infection by HIV and Neisseria gonorrhoeae, as well as other adverse gynecological conditions 21.
Human respiratory mucus
The air your breathe is full of dust, dirt, and air-borne microorganisms. To keep your lungs clear and healthy, this debris must be constantly swept out. To perform this task, the larger airways are lined by a relatively thick respiratory epithelium (Figure 2), which is the principal barrier protecting the lungs from the external environment. The human airway epithelium is comprised of three major cell types, basal cells, ciliated cells with cilia that beat and secretory cells (Goblet cells and Club cells) and a small number of endocrine cells, secreting serotonin and peptides that act as local mediators (Figure 3). These signal molecules affect nerve endings and other neighboring cells in the respiratory tract, so as to help regulate the rate of mucus secretion and ciliary beating, the contraction of surrounding smooth muscle cells that can constrict the airways, and other functions. Basal cells are also present, and serve as stem cells for renewal of the epithelium.
Airway mucus is a heterogeneous mixture of secreted polypeptides, cells, and cellular debris that are present in the airway surface lining fluid subphase or are tethered together at the fluid surface by oligomeric mucin complexes 22. Mucins are glycoproteins that due to their heavy glycosylation (∼ 75–90% carbohydrate by mass) can exhibit very high molecular weights (in the million Da range).
Where does mucus come from
Goblet cells (so named because of their shape), together with submucosal glands are the principal secretors of MUC5AC and MUC5B mucin glycoproteins respectively 23. These gel-forming mucins form the glycoprotein component of airway mucus. MUC5AC and MUC5B form homotypic polymers (i.e., MUC5AC monomers bond only with MUC5AC, and MUC5B monomers bond only with MUC5B), structured as long single chains rather than branches . They form the mucus gel both by entanglement in a mesh and by noncovalent calcium-dependent cross-linking of adjacent polymers 24. The glycan side chains bind large amounts of liquid (hundreds of times their weight), which allows mucus to act as a lubricant and the gel layer to serve as a liquid reservoir for the periciliary layer 25. The hydration of mucus dramatically affects its viscous and elastic properties, which in turn determine how effectively it is cleared by ciliary action and cough 26. Healthy mucus contains 3% solids, with the consistency of egg white. However, mucin hypersecretion or dysregulation of surface liquid volume may increase the concentration of solids up to 15%, resulting in viscous and elastic mucus that is not easily cleared 27. In addition, dehydrated mucus adheres more readily to the airway wall 28. Pulmonary disease conditions, such as cystic fibrosis (CF), chronic obstructive pulmonary disorder (COPD), and asthma, generally result in an increase in the viscoelasticity of mucus, owing in part to reduced water content and an increased fraction of glycoproteins 29, 30.
The mucus secreted by the goblet cells forms a viscoelastic blanket about 5 μm thick over the tops of the cilia. The cilia, all beating in the same direction, at a rate of about 12 beats per second, sweep the mucus out of the lungs, carrying with it the debris that has become stuck to it. This conveyor belt for the removal of rubbish from the lungs is called the mucociliary escalator. Of course, some inhaled particles may reach the alveoli themselves, where there is no escalator. Here, the unwanted matter is removed by yet another class of specialized cells, macrophages, which roam the lungs and engulf foreign matter and kill and digest bacteria. Many millions of macrophages, loaded with debris, are swept out of the lungs every hour on the mucociliary escalator.
At the upper end of the respiratory tract, the wet mucus-covered respiratory epithelium gives way abruptly to stratified squamous epithelium. This cell sheet is structured for mechanical strength and protection, and, like epidermis, it consists of many layers of flattened cells densely packed with keratin. It differs from epidermis in that it is kept moist and its cells retain their nucleus even in the outermost layers. Abrupt boundaries of epithelial cell specialization, such as that between the mucous and the stratified squamous epithelium of the respiratory tract, are also found in other parts of the body, but very little is known about how they are created and maintained.
Goblet cell differentiation is believed to have evolved to combat parasitic infection and is an essential step for the clearance of inhaled pathogens from the airways 31. In healthy individuals, transient goblet cell differentiation results in an increase in goblet cell number at the affected areas of the epithelium, and temporarily upregulates mucus production. In contrast, chronic airways diseases, such as asthma, chronic obstructive pulmonary disease (COPD) and cystic fibrosis (CF), are characterized by a persistent goblet cell phenotype and continually high levels of secreted mucins 32. Over time this persistent mucin production is linked to intraluminal mucus accumulation, increased rates of infection and airway obstruction 33.
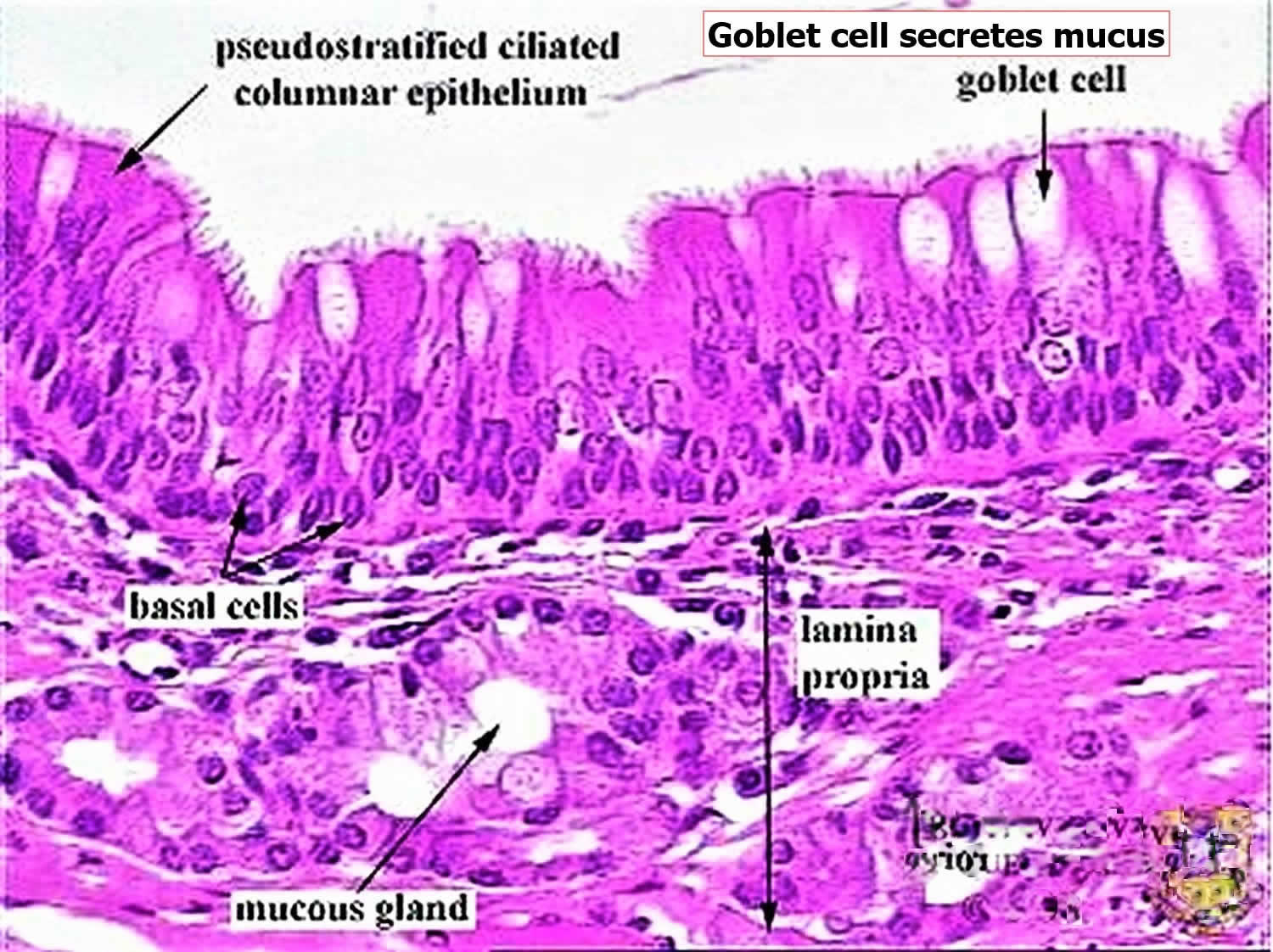
Figure 2. Respiratory epithelium
Footnote: The goblet cells secrete mucus, which forms a blanket over the tops of the ciliated cells. The regular, coordinated beating of the cilia sweeps the mucus up and out of the airways, carrying any debris that is stuck to it. The mechanism that coordinates the ciliary beating is a mystery, but it seems to reflect an intrinsic polarity in the epithelium. If a segment of rabbit trachea is surgically reversed, it carries on sweeping mucus, but in the wrong direction, back down toward the lung, in opposition to adjacent unreversed portions of trachea.
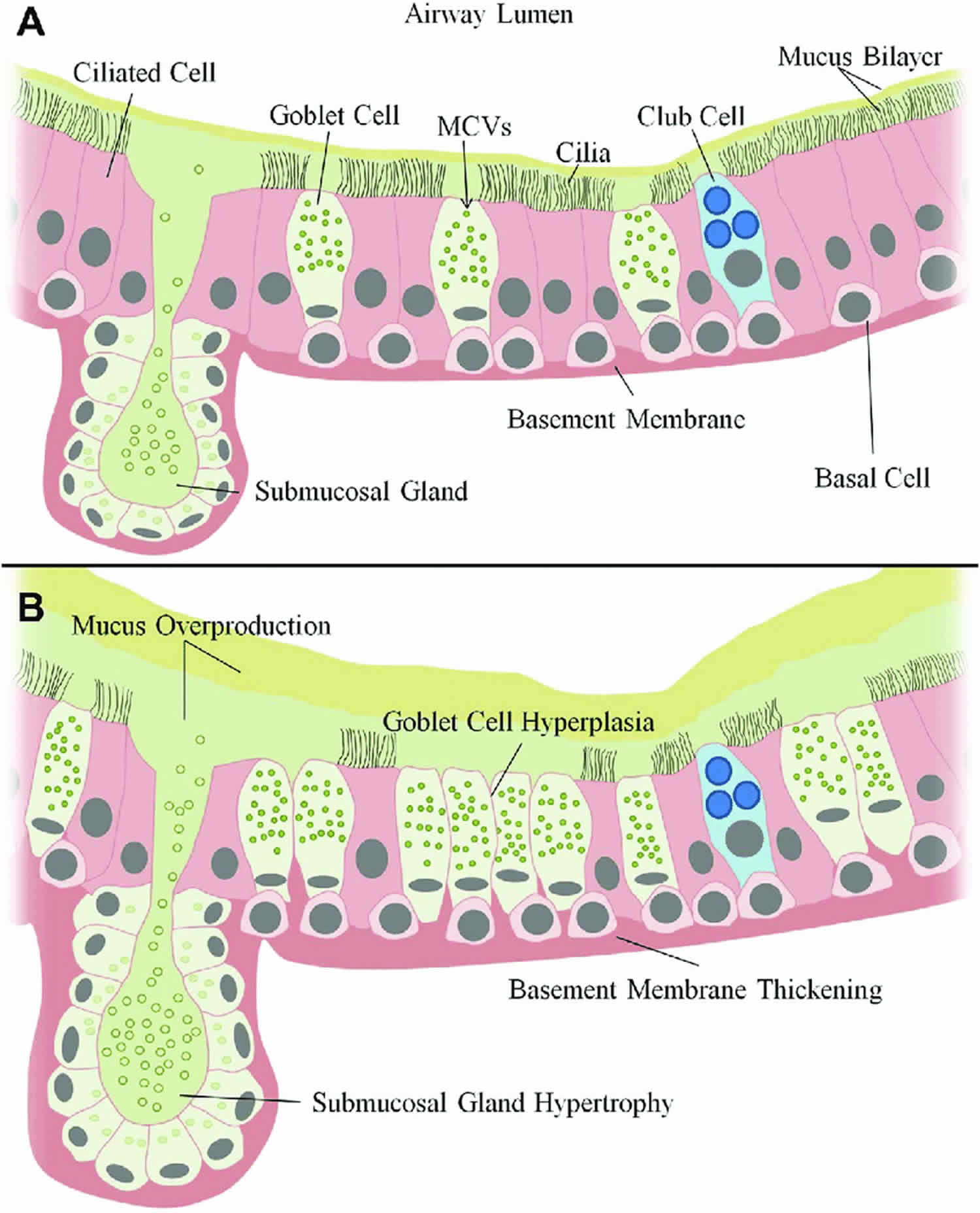
[Source 34 ]Figure 3. Human airway epithelium
Footnote: Normal vs diseased airway epithelium. In the normal airway epithelium (A) basal cells undergo regulated differentiation to form two major cell types, ciliated cells or secretory (Club and Goblet) cells. Goblet cells and submucosal glands are responsible for regulated mucus secretion into the airway which is normally present as a mucous bilayer. Club cells act to protect the airways through detoxification of harmful compounds and secretion of glycoproteins and lipids which physically protect surfactant and small airways. Mucocilliary clearance which is dependent on ciliary function actively moves mucus to remove foreign particles. In chronic airways disease (B) persistent goblet cell differentiation results in goblet cell hyperplasia. In addition to goblet cell differentiation, is the enlargement or hypertrophy of submucosal glands. These two events dramatically upregulate the production and secretion of mucin glycoproteins and result in a highly viscous and thickened mucus layer that is difficult to clear. This persistent goblet cell differentiation is directly linked to increased mucus accumulation in airways.
Abbreviation: MCVs = mucin containing vesicles.
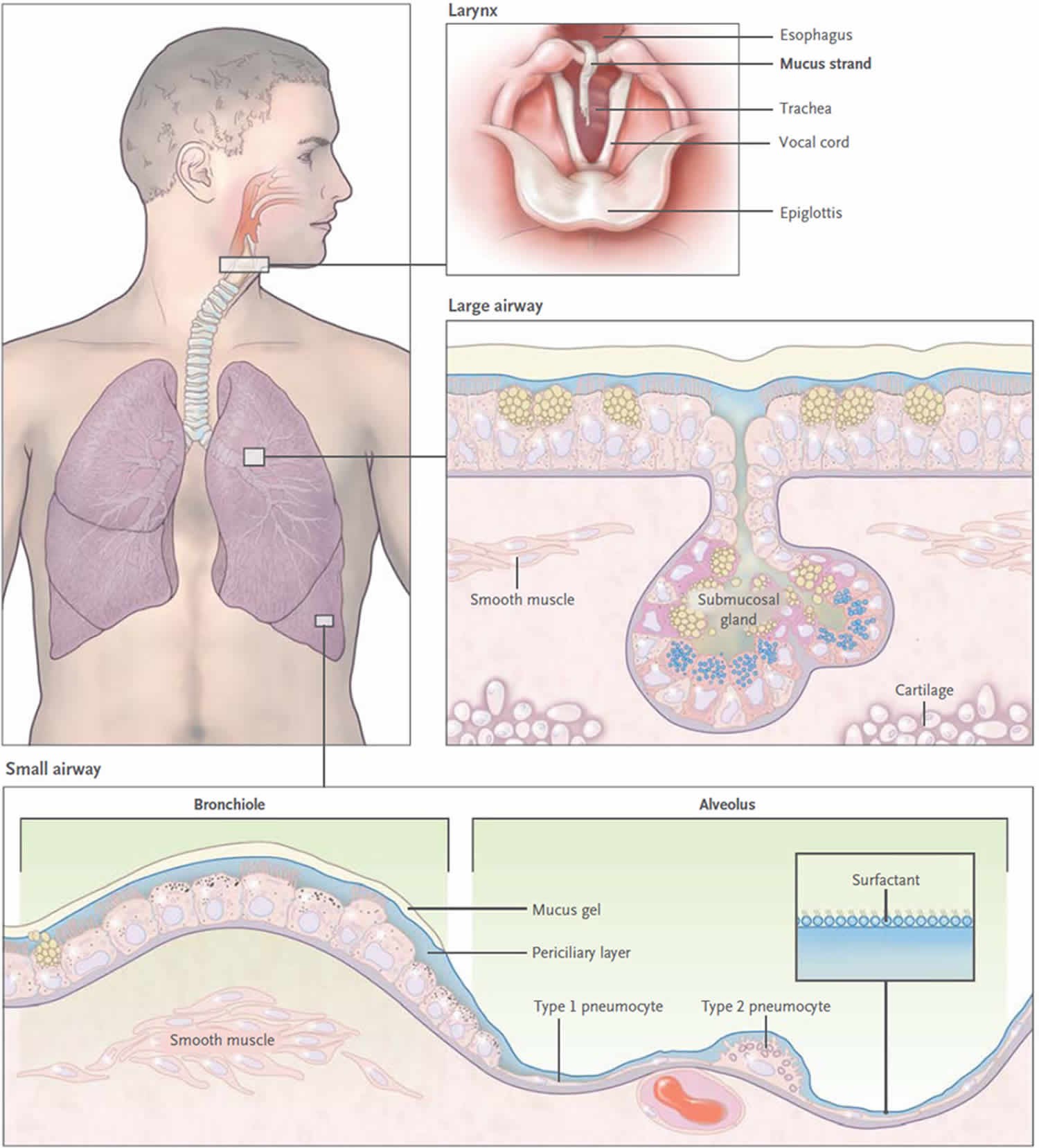
[Source 35 ]Figure 4. Mucus clearance in normal airways
Footnote: Mucus is continuously swept from distal to proximal airways. In the most distal bronchioles, epithelial cells are cuboidal and do not produce mucin (bottom box), and bronchiolar patency is stabilized by surfactant from adjacent alveoli.8 In the adjacent small airways, a thin mucus gel layer is produced by columnar secretory (Clara) cells that do not stain for intracellular mucins because they are produced in low amounts and steadily secreted. In the large airways lined by a pseudostratified epithelium, a thick mucus gel layer (up to 50 µm) accumulates from mucus transported from distal airways and additional mucins are produced by surface secretory cells and glands. After mucus ascends the trachea, it is propelled through the vocal cords by ciliary epithelium in the posterior commissure of the larynx. It then enters the pharynx and is swallowed, with approximately 30 ml of airway mucus eliminated by the gastrointestinal tract daily. The vocal cords are covered by squamous epithelium, so they do not participate in ciliary clearance, although they promote cough clearance by closing while expiratory pressure builds and then opening suddenly so airflow is forceful.
[Source 36 ]What causes excessive airway mucus
Effective mucus clearance is essential for lung health, and airway disease is a consistent consequence of poor clearance. Healthy mucus is a gel with low viscosity and elasticity that is easily transported by ciliary action, whereas pathologic mucus has higher viscosity and elasticity and is less easily cleared 26. The conversion from healthy to pathologic mucus occurs by multiple mechanisms that change its hydration and biochemical constituents; these include abnormal secretion of salt and water, increased production of mucins, infiltration of mucus with inflammatory cells, and heightened bronchovascular permeability (Figure 5). The accumulation of mucus results from some combination of overproduction and decreased clearance, and persistent accumulation can lead to infection and inflammation by providing an environment for microbial growth 36.
The principal symptoms of impaired mucus clearance are cough and shortness of breath (dyspnea). Cough is caused by the stimulation of vagal afferents in the intrapulmonary airways or the larynx and pharynx 37. Patients often infer that laryngopharyngeal stimulation, described as “a tickle in the throat,” results from “postnasal drip,” since they recognize that gravity causes mucus to descend from the nasopharynx but are generally unaware that it also ascends from the lungs by ciliary action. Dyspnea is caused when mucus obstructs airflow by occupying the lumen of numerous airways 38 . Physical signs of impaired mucus clearance include cough, bronchial breath sounds, rhonchi (continuous low pitched, rattling lung sounds that often resemble snoring), and wheezes. Retained mucus and inflammatory exudates may appear as localized atelectasis or linear or branched opacities on plain radiographs of the chest, and as luminal filling in proximal airways or tree-in-bud opacities in peripheral airways on computed tomographic examination 39. It is important to recognize the role of mucus in clinical presentation. It is necessary to clear mucus from the airway lumen in order to resolve symptoms and allow effective delivery of aerosol therapies. In addition, the presence of mucus may be a sign of underlying inflammation or infection that may warrant additional treatment.
Mucus dysfunction occurs in virtually all inflammatory airway diseases. Acute viral and bacterial infections and chronic diseases such as primary ciliary dyskinesia, non–cystic fibrosis bronchiectasis (which is often caused by atypical mycobacterial infection), panbronchiolitis, and immunodeficiency states (e.g., hypogammaglobulinemia, human immunodeficiency virus infection, organ transplantation, and hematologic malignant conditions) all have a component of mucus dysfunction. In addition, retained mucus is a problem in intubated patients and those in whom lung mechanics are disrupted as a result of paralysis, immobilization, or surgery; atelectasis and pneumonia are common complications in such patients. Genomic markers in chromosomal region 11p15.5 (which encompasses MUC5AC and MUC5B) have been reported to be associated with asthma severity 40 and panbronchiolitis 41, although mechanisms leading to disease susceptibility have not yet been defined.
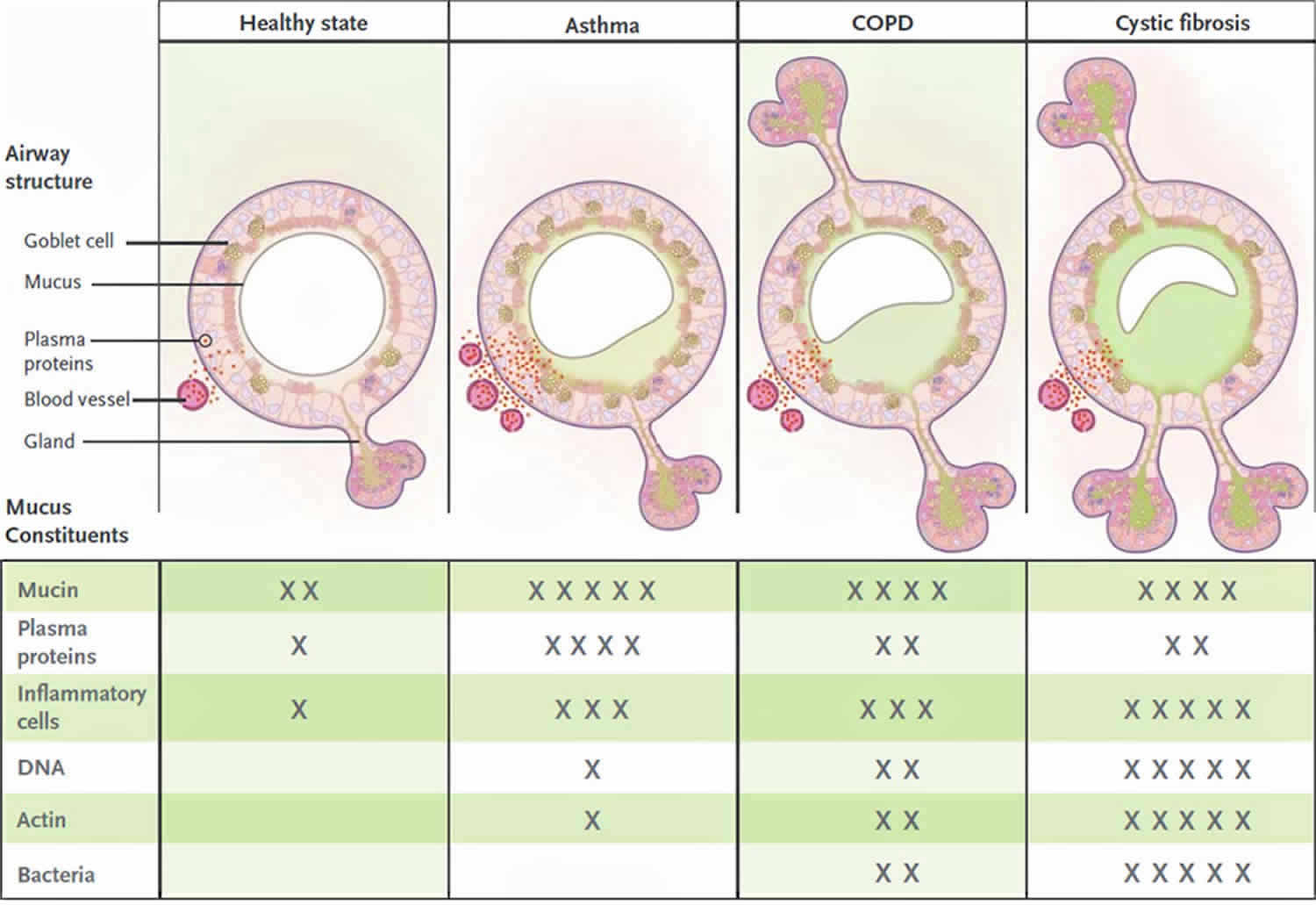
Figure 5. Airway Mucosal Disease and Mucus Characteristics
Footnotes: The top panels show the contribution of mucosal disease to abnormal mucus. In asthma, airway remodeling is characterized by increases in epithelial mucin stores because of surface epithelial mucous metaplasia with modest hyperplasia and increased numbers of subepithelial bronchial microvessels that become leaky during inflammation. Changes in submucosal glands are not prominent except in severe disease. In chronic obstructive pulmonary disease (COPD), increased mucin stores occur because of surface epithelial mucous metaplasia and some hyperplasia, together with increases in the volume and number of the submucosal glands. Bronchial microvessel remodeling is not as prominent as in asthma. In cystic fibrosis, epithelial mucin stores are similar to normal levels (possibly because of increased secretion), but submucosal glands are very prominent. Bronchial microvessel remodeling is not as prominent as in asthma. Not shown in the top panels are the increased numbers of inflammatory cells in the airway wall and lumen, which occur in all airway diseases. Products of inflammatory cell death include DNA and actin polymers, which are important constituents of pathologic mucus. The bottom panels list some of the constituents of mucus in health and in airway disease. The degree of cellular inflammation and biochemical constituents of mucus differ among airway diseases. The data for asthma reflect changes that occur in acute severe asthma. The number of Xs indicates the relative abundance of the constituents in each disease state.
[Source 36 ]Effects of smoking on the respiratory system
Smoking may cause a person to become easily “winded” during even moderate exercise because several factors decrease respiratory efficiency in smokers: (1) Nicotine constricts terminal bronchioles, which decreases airflow into and out of the lungs. (2) Carbon monoxide in smoke binds to hemoglobin and reduces its oxygen-carrying capability. (3) Irritants in smoke cause increased mucus secretion by the mucosa of the bronchial tree and swelling of the mucosal lining, both of which impede airflow into and out of the lungs. (4) Irritants in smoke also inhibit the movement of cilia and destroy cilia in the lining of the respiratory system. Thus, excess mucus and foreign debris are not easily removed, which further adds to the difficulty in breathing. The irritants can also convert the normal respiratory epithelium into stratified squamous epithelium, which lacks cilia and goblet cells. (5) With time, smoking leads to destruction of elastic fibers in the lungs and is the prime cause of emphysema (COPD). These changes cause collapse of small bronchioles and trapping of air in alveoli at the end of exhalation. The result is less efficient gas exchange.
How to get rid of excess mucus
The development of rationally designed treatments for pathologic mucus has been hindered by a lack of understanding of the mechanisms of mucus dysfunction. Over-the-counter medications for airway mucus dysfunction, including guaifenesin, have not been rigorously evaluated in clinical trials, and they are not recommended in treatment guidelines for cystic fibrosis, asthma, or chronic obstructive pulmonary disease (COPD) 42. Multiple additional agents with uncertain mechanisms are used worldwide without clear evidence of a benefit 43, 44. Asthma, chronic obstructive pulmonary disease (COPD), and cystic fibrosis (CF) have important differences in pathologic mucus, and mucus treatment should be tailored accordingly. Current therapies do this to an extent, but they may be facilitated by therapies that are currently under development. Therapies can be subdivided into those that decrease mucin production, those that decrease mucin secretion, those that promote mucus clearance, and those that treat airway infection (Table 1).
Recent insights into the formation of pathologic mucus in disease have led to the introduction of tailored therapies such as hydration by means of aerosolized hypertonic saline solutions or the reduction of mucus viscosity and elasticity by aerosolized dornase alfa 36. Targeted treatment of pathologic airway mucus not only improves symptoms of cough and dyspnea but also decreases the frequency of disease-related exacerbations and slows disease progression. Elucidation of how mucin production is controlled is still needed, since that might allow the development of additional therapies to prevent overproduction 36.
Table 1. Treatment of Mucus Hypersecretion
| Purpose and treatment | Comments |
|---|---|
| Decrease mucin production | |
| Available treatments | |
| Glucocorticoids | Allergen-induced increases in numbers of airway goblet cells are inhibited by glucocorticoids. In acute severe asthma, glucocorticoids may also reduce bronchovascular permeability and promote neutrophildriven mucus turnover. Glucocorticoids are much less effective in treating mucus in other airway diseases. |
| Agents in development | |
| ErbB-receptor inhibitors | A recent trial of inhaled BIBW 2948 BS, an epidermal growth factor receptor inhibitor (ClinicalTrials.gov number, NCT00423137), did not show a benefit in reducing airway mucin gene expression or epithelial mucin stores in patients with COPD, and the treatment was associated with adverse effects on lung and liver function. |
| Decrease mucin secretion | |
| Available treatments | |
| None | |
| Agents in development | |
| MARCKS inhibitors | MARCKS regulates the reconfiguration of actin filaments at the apical pole of goblet cells during mucin secretion. A peptide derived from the MARCKS N-terminal that is myristoylated so that it enters cells inhibits stimulated mucin secretion in mice. In a phase 2 trial (NCT00648245), this peptide is being administered by aerosol in patients with COPD who have mucus hypersecretion. |
| Botulinum neurotoxins | Botulinum neurotoxins are zinc proteases that cleave SNARE proteins to inhibit release of synaptic vesicles. The C and E neurotoxins have been engineered so that they are active against non-neuronal SNARE isoforms, inhibiting epithelial-cell mucin secretion, and the modified C toxin has been conjugated to epidermal growth factor to promote delivery to goblet cells, but no related clinical trial has been registered. |
| Promote mucus clearance | |
| Available treatments | |
| Physical measures | Chest percussion and postural drainage improve clearance of purulent airway mucus in cystic fibrosis. The value of alternative methods, including positive expiratory pressure, flutter valves, or high-frequency chest-compression vests, is difficult to assess objectively, although trials are in progress (e.g., NCT01057524). Mucus clearance is probably aided by any maneuver that promotes coughing and increased minute ventilation, including exercise. Since airflow generates shear stress on airway cell surfaces that stimulate release of nucleotides that interact with P2Y2 receptors to regulate mucus hydration, there are both mechanical and biochemical mechanisms of benefit from nonpharmacologic approaches to mucus clearance. |
| Bronchodilators | Bronchodilation with beta-adrenergic agonists or anticholinergic drugs may improve mucus clearance in the short term because of an enlarged luminal diameter. In addition, beta-adrenergic agonists increase the frequency of ciliary beats, and anticholinergic drugs may decrease surface mucin secretion and mucin secretion from the submucosal gland. However, beta-adrenergic agonists up-regulate mucin production in animal models, so they are not recommended for long-term treatment of mucus hypersecretion. The long-term effects of both classes of bronchodilators on mucus clearance warrant further study. |
| Inhaled dornase alfa | Inhaled dornase alfa hydrolyzes DNA, improves lung function, and decreases the frequency of exacerbation in patients with cystic fibrosis, in whom airway mucus concentrations of DNA are very high (5–10 mg/ml). The concentration of DNA in other airway diseases, including non−cystic fibrosis bronchiectasis, COPD, and asthma, is 1/5 to 1/10 as great; dornase alfa does not have beneficial effects in these diseases and may even be harmful. |
| Inhaled hypertonic saline | Treatment twice daily with aerosolized 7% hypertonic saline solution is associated with significant improvements in mucus clearance, modest improvements in airflow, and clinically meaningful reductions in rates of exacerbation among patients with cystic fibrosis. The mechanism of benefit is thought to be rehydration of the periciliary layer through the drawing of water from epithelial cells, but other mechanisms such as promotion of cough and direct effects on mucus elasticity and entanglement may also contribute. Trials are under way for the treatment of other airway diseases with 3 to 7% hypertonic saline; these diseases include infantile bronchiolitis (NCT01016249, NCT00677729, NCT00729274, NCT00619918, NCT00151905, and NCT00696540), COPD (NCT00639236), atelectasis (NCT00671723), non−cystic fibrosis bronchiectasis (NCT00484263), and asthma (NCT01073527). |
| N-acetylcysteine | N-acetylcysteine breaks the disulfide bonds that link mucin monomers to polymers, and it is very effective in vitro in solubilizing sputum. Case reports attest to its usefulness when applied through the bronchoscope to break up mucus plugs. Clinical studies of N-acetylcysteine and carbocysteine in COPD have shown some promise. Aerosolized N-acetylcysteine also can be irritating to the airway, so its routine use is not recommended. Oral N-acetylcysteine is under study as an antiinflammatory (glutathione-replenishing and antioxidant) treatment in cystic fibrosis and COPD (NCT00969904 and NCT00809094). |
| Agents in development | |
| Mannitol | Nonabsorbable osmotic agents have a theoretical advantage of drawing liquid into the periciliary layer for longer periods than sodium chloride. Inhaled mannitol is undergoing safety and efficacy testing in cystic fibrosis, COPD, and bronchiectasis (NCT00446680, NCT00117208, and NCT00669331). Its use in children with cystic fibrosis is associated with bronchoconstriction and cough, but a 3-month treatment protocol showed similar efficacy to that of dornase alfa. |
| P2Y2 agonists | P2Y2 agonists promote the activity of calcium-activated chloride channels and inhibit the activity of epithelial sodium channels, so they may normalize the height of the periciliary layer and improve mucus clearance, especially in cystic fibrosis. Phase 2 and 3 trials of denufusol in cystic fibrosis are ongoing (NCT00625612 and NCT00357279). |
| CFTR modulation | Several therapeutic agents are in development to augment the function of mutant CFTR, including ataluren to promote read-through of premature termination codons (NCT00803205), VX-809 to promote transport of misfolded CFTR protein to the cell surface (NCT00865904 and NCT00966602), and VX-770 to promote the opening of CFTR proteins expressed on the cell surface (NCT00909532 and NCT00966602). |
| Epithelial sodium-channel modulation | An aerosolized inhibitor of epithelial sodium-channel function, GS-9411, is being evaluated as a potential therapy to improve airway hydration and mucociliary clearance in cystic fibrosis (NCT00800579, NCT009999531, NCT01025713, and NCT00951522). |
| Actin filament depolymerizing agents, proteases, and antiproteases | Gelsolin and thymosin β4 depolymerize actin filaments, which could be helpful in promoting mucus clearance in cystic fibrosis; however, the results of clinical studies of recombinant human plasma gelsolin in cystic fibrosis in the 1990s were not promising, and no trials are currently registered. Proteases that digest gel-forming mucins could be helpful in treating mucus plugs in severe asthma, although no trials are yet under way. Inhaled alpha1-antitrypsin is being studied to prevent lung parenchymal damage from leukocyte proteases in cystic fibrosis (NCT00499837). |
| Treat airway infection | |
| Available treatments | |
| Inhaled antibiotics | Treatment with inhaled tobramycin is associated with improved lung function and reduced exacerbation frequency in patients with cystic fibrosis. |
| Oral antibiotics | Prolonged use of azithromycin improves lung function in cystic fibrosis, although it is associated with increased nausea and diarrhea. Long-term treatment with oral erythromycin reduces the frequency of exacerbation in COPD and several groups, including the COPD Clinical Research Network (NCT00119860), are studying azithromycin in patients with this condition. The value of oral antibiotics in the short-term treatment of COPD exacerbations is questionable. In patients with bronchiectasis who do not have cystic fibrosis, long-term treatment with low-dose azithromycin reduces the frequency of exacerbation and improves lung function. |
| Intravenous antibiotics | Intravenous antibiotics directed against pseudomonas species infections are effective in the treatment of pulmonary exacerbations of cystic fibrosis. |
| Agents in development | |
| Inhaled antibiotics | Inhaled aztreonam reduced the time to exacerbation in cystic fibrosis in two phase 3 studies (NCT00112359, NCT00104520), and a study comparing inhaled aztreonam with inhaled tobramycin in cystic fibrosis is ongoing (NCT00757237). Also under study are inhaled ciprofloxacin (NCT00645788), liposomal amikacin (NCT00558844), and tobramycin combined with fosfomycin (NCT00794586). |
Abbreviations: *CFTR = cystic fibrosis transmembrane conductance regulator; COPD = chronic obstructive pulmonary disease; MARCKS = myristoylated alanine-rich C-kinase substrate; SNARE = soluble N-ethylmaleimide–sensitive factor attachment protein receptor.
[Source 36 ]Home remedies
There are things you can try at home to relieve your symptoms, such as:
- avoiding things that trigger your symptoms, such as allergens or smoky places
- taking sips of cold water when you feel the need to clear your throat – constantly clearing your throat may make things worse
- using a saline nasal rinse several times a day – these can be bought from a pharmacy or made at home with half a teaspoon of salt in a pint of boiled water that’s been left to cool
- avoiding warm, dry atmospheres, such as places with air conditioning and car heating systems – placing plants or bowls of water in a room may help to keep the air humid
- staying well hydrated
- talking to a pharmacist about suitable over-the-counter medications – including decongestants, antihistamines or steroid nasal sprays
Techniques to bring up mucus
People with chronic obstructive pulmonary disease (COPD) and chronic bronchitis often produce a large amount of mucus. If the mucus is allowed to collect in the airways, breathing may become difficult, and infection may occur. Techniques to remove mucus are often done after using an inhaled bronchodilator medication. The medication helps loosen the mucus and open the airways to make the techniques more effective. Common techniques used to help remove mucus include these, which can be ordered and demonstrated by your doctor.
Deep Coughing
Start by taking a deep breath. Hold the breath for 2-3 seconds. Use your stomach muscles to forcefully expel the air. Avoid a hacking cough or merely clearing the throat. A deep cough is less tiring and more effective in clearing mucus out of the lungs.
Huff Coughing
Huff coughing, or huffing, is an alternative to deep coughing if you have trouble clearing your mucus. Take a breath that is slightly deeper than normal. Use your stomach muscles to make a series of three rapid exhalations with the airway open, making a “ha, ha, ha” sound. Follow this by controlled diaphragmatic breathing and a deep cough if you feel mucus moving.
Other Techniques
If you produce a large amount of mucus, your health care provider may recommend other techniques to help clear the mucus.
- Devices such as the Acapella® or Aerobika® are small devices into which you exhale.
- Postural drainage is a technique that uses gravity to promote drainage of mucus from the lungs.
- Each technique can be ordered and demonstrated by your health care provider. Ask your health care provider if this may be helpful for you.
Exercise is also a good way to help bring up mucus in the lungs.
Mucus color chart
Discolored sputum (or respiratory discharge) is commonly interpreted by both patients and physicians as a clinical sign for the presence of bacterial infection 45. Thus in patients with acute respiratory infections yellowish and greenish sputum as well as certain non-specific symptoms such as fatigue are strong predictors for antibiotic prescribing in primary care 46. In reality, purulence primarily occurs when inflammatory cells or sloughed mucosal epithelial cells are present, and it can result from either viral or bacterial infection 47. Robertson discovered in 1955 that green sputum color is caused by crystallized peroxidase. Since this enzyme is released by leukocytes (white blood cells) that are part of the general immune response, discolored sputum color can be due to a variety of causes. Therefore, yellowish or green sputum may be a normal feature in patients with viral bronchitis. Also, sputum production in viral airway infections may be clear, white, or even tinged with blood 48. It has been shown that a yellowish or greenish sputum color is often related to the bacterial load of patients suffering from COPD (chronic obstructive pulmonary disease) exacerbation 49 or patients hospitalized due to respiratory conditions 50. In otherwise healthy patients only indirect evidence showed that yellowish or greenish sputum did not influence outcomes with or without antibiotic therapy 51. However, few studies have specifically examined the role of sputum color in primary care patients without chronic respiratory diseases, although these patients are the most prevalent in general practice. In everyday practice only sputum samples of problematic patients will be sent in for microbiological investigation. In a recent study, Johnson et al. 47 examined such 289 consecutive outpatient samples sent in by primary care physicians, finding that the bacterial yield from sputum colors green, yellow-green, yellow, and rust was higher than the yield from cream, white, or clear samples.
- Green or yellow mucus. This shade is usually a sign that you have an infection of some kind, like a cold or the flu. The green color comes from a protein released from your inflammatory cells. It’s a toxic substance that kills germs trying to get in.
- Red or pink mucus. If you’re sick and coughing a lot, you may notice blood-tinged mucus. This can come from broken blood vessels in your nose or throat. In some cases, it can also be a sign of cancer. Your doctor can do tests to find out the cause.
- Brown or black mucus. Dark-colored mucus can signal an infection. It’s also common in heavy smokers or people who are around smoke or coal dust at their job. This type of mucus also shows up in people with chronic lung disease. The color comes from a mix of blood and inflammation in the lungs.
So far there has been no evidence that discolored sputum is correlated with a bacterial cause of acute respiratory infections in otherwise healthy adults. This study 52 investigates the correlation of discolored sputum in patients with acute cough and a bacterial cause. The findings imply that the color of sputum or discharge cannot be used to differentiate between viral and bacterial infections in otherwise healthy adults 52. The color of sputum should not be used to make a decision on whether to prescribe an antibiotic within this group of patients.
In 28 samples (12%) a bacterial infection was proven. Yellowish or greenish color of the sputum sample and bacterial infection showed a significant correlation. The sensitivity of yellowish or greenish sputum used as a test for a bacterial infection was 0.79; the specificity was 0.46. The positive likelihood-ratio was 1.46.
Table 2. Sputum color and microbiological proof of bacterial infection
| Bacterial infection | No bacterial infection | Totals | |
| Yellowish or greenish sputum sample | 22 (16.2%) | 114 (83.8%) | 136 (100%) |
| Colorless sputum sample | 6 (5.7%) | 99 (94.3%) | 105 (100%) |
| Totals | 28 (11.6%) | 213 (88.4%) | 241 |
Conclusions
The sputum color or discharge of patients with acute cough and no underlying chronic lung disease cannot be used in the consultation with otherwise healthy adult patients suffering from acute cough to differentiate between viral and bacterial infections. Thus it should not be used to make a decision on the choice of therapy and especially not on whether to prescribe an antibiotic 52.
- Lai SK, Wang YY, Wirtz D, Hanes J. Micro- and macrorheology of mucus. Adv Drug Deliv Rev. 2009;61(2):86-100. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2736374/[↩][↩]
- Biological modeling of mucus to modulate mucus barriers. Jean-Luc Desseyn, Valérie Gouyer, and Frédéric Gottrand. American Journal of Physiology-Gastrointestinal and Liver Physiology 2016 310:4, G225-G227 https://www.physiology.org/doi/full/10.1152/ajpgi.00274.2015[↩]
- McAuley JL, Linden SK, Png CW, King RM, Pennington HL, Gendler SJ, Florin TH, Hill GR, Korolik V, McGuckin MA. MUC1 cell surface mucin is a critical element of the mucosal barrier to infection. J Clin Invest. 2007;117:2313–2324[↩]
- Strombeck DR, Harrold D. Binding of cholera toxin to mucins and inhibition by gastric mucin. Infect Immun. 1974;10:1266–1272.[↩]
- Kreyling WG, Semmler M, Moller W. Dosimetry and toxicology of ultrafine particles. J Aerosol Med. 2004;17:140–152.[↩]
- Cone R. Mucus. In: Michael WS, Lamm E, McGhee Jerry R, Mayer Lloyd, Mestecky Jiri, Bienenstock John, editors. Mucosal Immunlogy. San Diego: Academic Press; 1999. pp. 43–64.[↩][↩]
- Zhang K , Hornef MW , Dupont A .The intestinal epithelium as guardian of gut barrier integrity.Cell Microbiol 17: 1561-1569, 2015.[↩]
- Kaiko GE , Stappenbeck TS .Host-microbe interactions shaping the gastrointestinal environment.Trends Immunol 35: 538-548, 2014.[↩]
- Johansson MEV , Gustafsson JK , Holmen-Larsson J , Jabbar KS , Xia L , Xu H , Ghishan FK , Carvalho FA , Gewirtz AT , Sjovall H , Hansson GC .Bacteria penetrate the normally impenetrable inner colon mucus layer in both murine colitis models and patients with ulcerative colitis.Gut 63: 281-291, 2014.[↩]
- Kelly T , Buxbaum J .Gastrointestinal manifestations of cystic fibrosis.Dig Dis Sci 60: 1903-1913, 2015.[↩]
- Van Klinken BJ, Dekker J, Buller HA, Einerhand AW. Mucin gene structure and expression: protection vs. adhesion. Am J Physiol. 1995;269:G613–G627.[↩][↩]
- Thornton DJ, Sheehan JK. From mucins to mucus: toward a more coherent understanding of this essential barrier. Proc Am Thorac Soc. 2004;1:54–61.[↩]
- Shogren R, Gerken TA, Jentoft N. Role of glycosylation on the conformation and chain dimensions of O-linked glycoproteins: light-scattering studies of ovine submaxillary mucin. Biochemistry. 1989;28:5525–5536.[↩][↩]
- Carlstedt I, Sheehan JK. Macromolecular properties and polymeric structure of mucus glycoproteins. Ciba Foundation Symposium. 1984;109:157–172.[↩]
- Lamblin G, Lhermitte M, Klein A, Houdret N, Scharfman A, Ramphal R, Roussel P. The carbohydrate diversity of human respiratory mucins: a protection of the underlying mucosa? The American Review of Respiratory Disease. 1991;144:S19–S24.[↩]
- Masson PL, Heremans LF. Sputum proteins. In: Dulfano MF, editor. Fundamentals and Clinical Pathology. Springfield: Charles C. Thomas; 1973. pp. 412–474.[↩]
- Puchelle E, Zahm JM, Havez R. Biochemical and rheological data in sputum. 3. Relationship between the biochemical constituents and the rheological properties of sputum. Bulletin de physio-pathologie respiratoire. 1973;9:237–257.[↩]
- Randell SH, Boucher RC. Effective mucus clearance is essential for respiratory health. Am J Respir Cell Mol Biol. 2006;35:20–28.[↩]
- Dulfano MJ, Adler K, Philippoff W. Sputum viscoelasticity in chronic bronchitis. The American Review of Respiratory Disease. 1971;104:88–98.[↩]
- Dawson M, Wirtz D, Hanes J. Enhanced Viscoelasticity of Human Cystic Fibrotic Sputum Correlates with Increasing Microheterogeneity in Particle Transport. Journal of Biological Chemistry. 2003;278:50393–50401.[↩]
- Olmsted SS, Meyn LA, Rohan LC, Hillier SL. Glycosidase and proteinase activity of anaerobic gram-negative bacteria isolated from women with bacterial vaginosis. Sexually Transmitted Diseases. 2003;30:257–261.[↩]
- Williams OW, Sharafkhaneh A, Kim V, Dickey BF, Evans CM. Airway mucus: From production to secretion. Am J Respir Cell Mol Biol. 2006;34(5):527-36. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2644218/[↩]
- Hovenberg, H. W., Davies, J. R., & Carlstedt, I. (1996). Different mucins are produced by the surface epithelium and the submucosa in human trachea: Identification of MUC5AC as a major mucin from the goblet cells. Biochemical Journal, 318(Pt 1), 319–324.[↩]
- Thornton DJ, Rousseau K, McGuckin MA. Structure and function of the polymeric mucins in airways mucus. Annu Rev Physiol. 2008;70:459–486.[↩]
- Knowles MR, Boucher RC. Mucus clearance as a primary innate defense mechanism for mammalian airways. J Clin Invest. 2002;109:571–577[↩]
- Cone RA. Barrier properties of mucus. Adv Drug Deliv Rev. 2009;61:75–85.[↩][↩]
- Lai SK, Wang YY, Wirtz D, Hanes J. Micro- and macrorheology of mucus. Adv Drug Deliv Rev. 2009;61:86–100.[↩]
- Rubin BK. Mucus and mucins. Otolaryngol Clin North Am. 2010;43:27–34.[↩]
- Rubin BK. Mucus structure and properties in cystic fibrosis. Paediatr Respir Rev. 2007;8:4–7.[↩]
- Voynow JA, Gendler SJ, Rose MC. Regulation of mucin genes in chronic inflammatory airway diseases. Am J Respir Cell Mol Biol. 2006;34:661–665.[↩]
- Evans, C. M., Kim, K., Tuvim, M. J., & Dickey, B. F. (2009). Mucus hypersecretion in asthma: Causes and effects. Current Opinion in Pulmonary Medicine, 15, 4–11.[↩]
- McCullagh, C. M., Jamieson, A. M., Blackwell, J., & Gupta, R. (1995). Viscoelastic properties of human tracheobronchial mucin in aqueous solution. Biopolymers, 35, 149–159.[↩]
- Rose, M. C., & Voynow, J. A. (2006). Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiological Reviews, 86, 245–278.[↩]
- Alberts B, Johnson A, Lewis J, et al. Molecular Biology of the Cell. 4th edition. New York: Garland Science; 2002. The Airways and the Gut. Available from: https://www.ncbi.nlm.nih.gov/books/NBK26875/[↩]
- Persistent induction of goblet cell differentiation in the airways: Therapeutic approaches. Reid, A.T., Pharmacology and Therapeutics (2017), https://doi.org/10.1016/j.pharmthera.2017.12.009[↩]
- Fahy JV, Dickey BF. Airway mucus function and dysfunction. N Engl J Med. 2010;363(23):2233-47. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4048736/[↩][↩][↩][↩][↩][↩]
- Rubin BK. The role of mucus in cough research. Lung. 2010;188(Suppl):S69–S72.[↩]
- Bossé Y, Riesenfeld EP, Paré PD, Irvin CG. It’s not all smooth muscle: non-smooth-muscle elements in control of resistance to airflow. Annu Rev Physiol. 2010;72:437–462.[↩]
- Martinez S, Heyneman LE, McAdams HP, Rossi SE, Restrepo CS, Eraso A. Mucoid impactions: finger-in-glove sign and other CT and radiographic features. Radiographics. 2008;28:1369–1382.[↩]
- The Collaborative Study on the Genetics of Asthma (CSGA) A genome-wide search for asthma susceptibility loci in ethnically diverse populations. Nat Genet. 1997;15:389–392.[↩]
- Kamio K, Matsushita I, Hijikata M, et al. Promoter analysis and aberrant expression of the MUC5B gene in diffuse panbronchiolitis. Am J Respir Crit Care Med. 2005;171:949–957.[↩]
- Flume PA, Mogayzel PJ, Jr, Robinson KA, et al. Cystic fibrosis pulmonary guidelines: treatment of pulmonary exacerbations. Am J Respir Crit Care Med. 2009;180:802–808.[↩]
- Rogers DF. Mucoactive agents for airway mucus hypersecretory diseases. Respir Care. 2007;52:1176–1193.[↩]
- Boogaard R, de Jongste JC, Merkus PJ. Pharmacotherapy of impaired mucociliary clearance in non-CF pediatric lung disease: a review of the literature. Pediatr Pulmonol. 2007;42:989–1001.[↩]
- Wilm S, Knauf A, Kreilkamp R, Schlegel U, Altiner A. The doctor, his patient and the sputum. Z Allg Med. 2006;82:260–67.[↩]
- Cals JW, Boumans D, Lardinois RJ, Gonzales R, Hopstaken RM, Butler CC, et al. Public beliefs on antibiotics and respiratory tract infections: An internet-based questionnaire study. Br J Gen Pract. 2007;57:942–7.[↩]
- Johnson AL, Hampson DF, Hampson NB. Sputum color: Potential implications for clinical practice. 2008;53:450–4. http://rc.rcjournal.com/content/53/4/450/tab-pdf[↩][↩]
- Knutson D, Braun C. Diagnosis and management of acute bronchitis. Am Fam Physician. 2002;65:2039–44.[↩]
- Allegra L, Blasi F, Diano P, Cosentini R, Tarsia P, Confalonieri M, et al. Sputum color as a marker of acute bacterial exacerbations of chronic obstructive pulmonary disease. Respir Med. 2005;99:742–7.[↩]
- Flournoy DJ, Davidson LJ. Sputum quality: Can you tell by looking? Am J Infect Control. 1993;21:64–9.[↩]
- Stott NC, West RR. Randomised controlled trial of antibiotics in patients with cough and purulent sputum. BMJ. 1976;4:556–9[↩]
- Altiner A, Wilm S, Däubener W, et al. Sputum colour for diagnosis of a bacterial infection in patients with acute cough. Scand J Prim Health Care. 2009;27(2):70-3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3410464/[↩][↩][↩][↩]