Contents
- What is pemphigus foliaceus
- Pemphigus foliaceus causes
- Pemphigus foliaceus pathology
- Pemphigus foliaceus signs and symptoms
- Pemphigus foliaceus diagnosis
- Pemphigus foliaceus differential diagnosis
- Pemphigus foliaceus treatment
- Pemphigus foliaceus prognosis
What is pemphigus foliaceus
Pemphigus foliaceus is a rare potentially life-threatening autoimmune blistering skin disease that only affects the skin in which immunoglobulin G4 (IgG4) autoantibodies are directed against desmoglein-1 (Dsg1) which is found in desmosomes in the keratinocytes near the superficial layers of the epidermis, causing the separation of keratinocytes (acantholysis) and cleavage and the formation of flaccid vesicles or blisters 1, 2, 3, 4. Pemphigus foliaceus is characterized by blistering lesions on otherwise healthy-looking skin. The blisters that form in pemphigus foliaceus have a delicate outer surface which can be easily damaged leaving extensive raw areas (erosions) which can be painful. Blisters tend to form when the skin is rubbed (Nikolsky sign). Nikolsky sign is a skin finding in which the top layers of the skin slip away from the lower layers when rubbed, forming a blister within minutes 5.
Normally your immune system makes antibodies to protect you against bacteria, viruses, fungi, parasites and cancer cells and these antibodies do not attack your own body. However, in pemphigus foliaceus, your immune system makes antibodies (autoantibodies) that attack the “glue” holding the outer layer of the skin (epidermis) together, making it fragile and prone to blistering. It is not clear why some people develop these autoantibodies. The autoantibodies in pemphigus foliaceus attack proteins called desmogleins. These proteins form the “glue” that holds the cells of the skin together. When the autoantibodies formed in pemphigus foliaceus attack the desmoglein proteins, the cells in the skin no longer hold together and separate. This causes blisters that are typical of pemphigus foliaceus.
Pemphigus foliaceus should not be confused with pemphigus vulgaris which is a more severe related condition. Pemphigus foliaceous commonly affects the scalp, face, trunk, armpits and genital areas. The skin lesions are typically small, scattered blisters that rapidly evolve into scaly, crusted erosions. Skin erosions (sores) are painful and can affect quality of life including disturbing sleep. Pemphigus foliaceus does not usually affect other parts of the body such as the mucous membranes, it only affects the skin and antibodies are exclusively directed against desmoglein-1 (Dsg1) 6. This is in contrast to pemphigus vulgaris where there may be extensive mucous membrane involvement (mouth, eyelids etc.). Pemphigus vulgaris is characterized by blisters and erosions of the skin and/or mucous membranes, and circulating antibodies directed against desmoglein-1 (Dsg1) and desmoglein-3 (Dsg3) 7, 8.
Pemphigus foliaceus affects less than 5 people per 1 million people worldwide each year. Pemphigus foliaceous affects men and women equally. The average age of onset is usually 50 to 60 years. Pemphigus foliaceus rarely occur in children. More commonly, it occurs in people originating from North Africa, Turkey, and South America.
Variants of pemphigus foliaceus have been described and they include 1, 9, 10, 11, 12, 13, 2:
- Cazenave also commonly known as classical pemphigus foliaceus or sporadic pemphigus foliaceus. Sporadic pemphigus foliaceus or classical pemphigus foliaceus is known to be more prevalent in middle-aged and elderly people and to be extremely rare in children.
- Endemic pemphigus foliaceus also commonly known as Fogo selvagem (FS). Endemic pemphigus foliaceus is common in Tunisia and Brazil. Endemic pemphigus foliaceus appears to be set off by a virus transmitted by an insect bite (black fly – Simulium nigrimanum) 1. Endemic pemphigus foliaceus frequently occurs in genetically related family members.
- Pemphigus erythematosus also commonly known as Senear-Usher syndrome. Pemphigus erythematosus (Senear-Usher syndrome) is a variant, in which patients have immunological features of both cutaneous lupus erythematosus and pemphigus: IgG and C3 deposition at the basement membrane zone and on the cell surfaces of keratinocytes, in addition to circulating antinuclear antibodies 14, 15, 16, 2. Progression to lupus erythematosus is rare 2. The clinical hallmarks of pemphigus erythematosus (Senear-Usher syndrome) are scaly, erythematous lesions over the nose, nasolabial folds, and malar areas of the face, that resemble the “butterfly” distribution of lupus. Lesions may also affect the preauricular region. Hyperkeratotic scars with erythema and superficial blisters can be present on the chest. Sunlight may exacerbate the disease. Oral mucosa is rarely involved 2. The immunopathology of pemphigus erythematosus was described by Chorzelski et al. 17, demonstrating the presence of immunoglobulin (Ig) and complement at the dermo-epidermal junction resembling the lupus band test. Pemphigus erythematosus patients also exhibited antinuclear antibodies (ANA) and the report proposed the coexistence of pemphigus and lupus erythematosus. The histology and anti-Dsg1 serological marker of pemphigus foliaceus and pemphigus erythematosus are the same 18.
- Pemphigus herpetiformis (pemphigus resembling dermatitis herpetiformis). Widespread clusters of itchy papules and vesicles develop on an erythematous background. Pemphigus herpetiformis usually evolves into classical pemphigus foliaceus but has also been described as preceding pemphigus vulgaris. In general, the clinical course is benign 2.
- IgA pemphigus foliaceus. Immunoglobulin A pemphigus or IgA pemphigus is a rare autoimmune blistering skin disorder, manifests as painful, itchy blisters that fill with neutrophils and evolve into pustules 19. IgA pemphigus has 2 major subtypes: subcorneal pustular dermatosis (SPD) and intraepidermal neutrophilic (IEN) dermatosis. Both subtypes exhibit distinct immunologic mechanisms targeting keratinocyte cell surface components 20, 19. Subcorneal pustular dermatosis type has deposition of intracellular IgA against the glycoprotein desmocollin-1 seen predominantly in the upper epidermis. Intraepidermal neutrophilic type has deposition of autoantibodies against desmoglein members of the cadherin superfamily seen predominantly in lower epidermis. IgA pemphigus has been associated with cancers (multiple myeloma) and several chronic conditions (monoclonal IgA gammopathy, human immunodeficiency virus infection, Sjögren’s syndrome, rheumatoid arthritis, and Crohn’s disease) 20.
Myasthenia gravis, thymoma, or both have been associated with pemphigus vulgaris and pemphigus foliaceus 21.
Pemphigus foliaceus is not an infection and therefore cannot be caught or passed on to anyone else. Although it is not contagious, and to date, spread by blood products or body fluids has not been documented 22, 23.
Pemphigus foliaceus diagnosis generally requires a skin biopsy, which shows typical features of rounded-up separated keratinocytes called acantholytic cells within the blisters in the upper layers of the epidermis. Pemphigus foliaceus is confirmed by direct immunofluorescence (DIF) staining of the skin sections to reveal antibodies. In some cases, circulating antibodies can be detected by a blood test using indirect immunofluorescence test (IIF) or enzyme-linked immunosorbent assay (ELISA).
The hallmark of pemphigus is the finding of immunoglobulin G (IgG) autoantibodies against the cell surface of keratinocytes with direct immunofluorescence (DIF). The diagnosis of pemphigus should be seriously questioned if the test result of direct immunofluorescence (DIF) is negative. It is important that the biopsy for direct immunofluorescence (DIF) be performed on the normal-appearing perilesional skin, as immune reactants can be difficult to detect in the blistered epidermis
Direct immunofluorescence (DIF) and indirect immunofluorescent (IIF) findings are usually indistinguishable from pemphigus vulgaris, with intercellular immunoglobulin G (IgG) and C3 throughout the epidermis. Occasionally intercellular staining is restricted to the upper layers of the epidermis, both on direct and indirect immunofluorescence.
Indirect immunofluorescence (IIF) is positive in over 85% of pemphigus foliaceus, enzyme-linked immunosorbent assay (ELISA) detects anti-desmoglein-1 antibodies in up to 71% of patients, but immunoblotting is less sensitive 24.
Recently, trichoscopy has been evaluated as a useful tool in the differential diagnosis of scalp pemphigus. Extravasations and yellow hemorrhagic crusts were the most frequent findings. The “fried egg sign” (yellow dots with a whitish halo) was identified as a new trichoscopic feature of pemphigus 25.
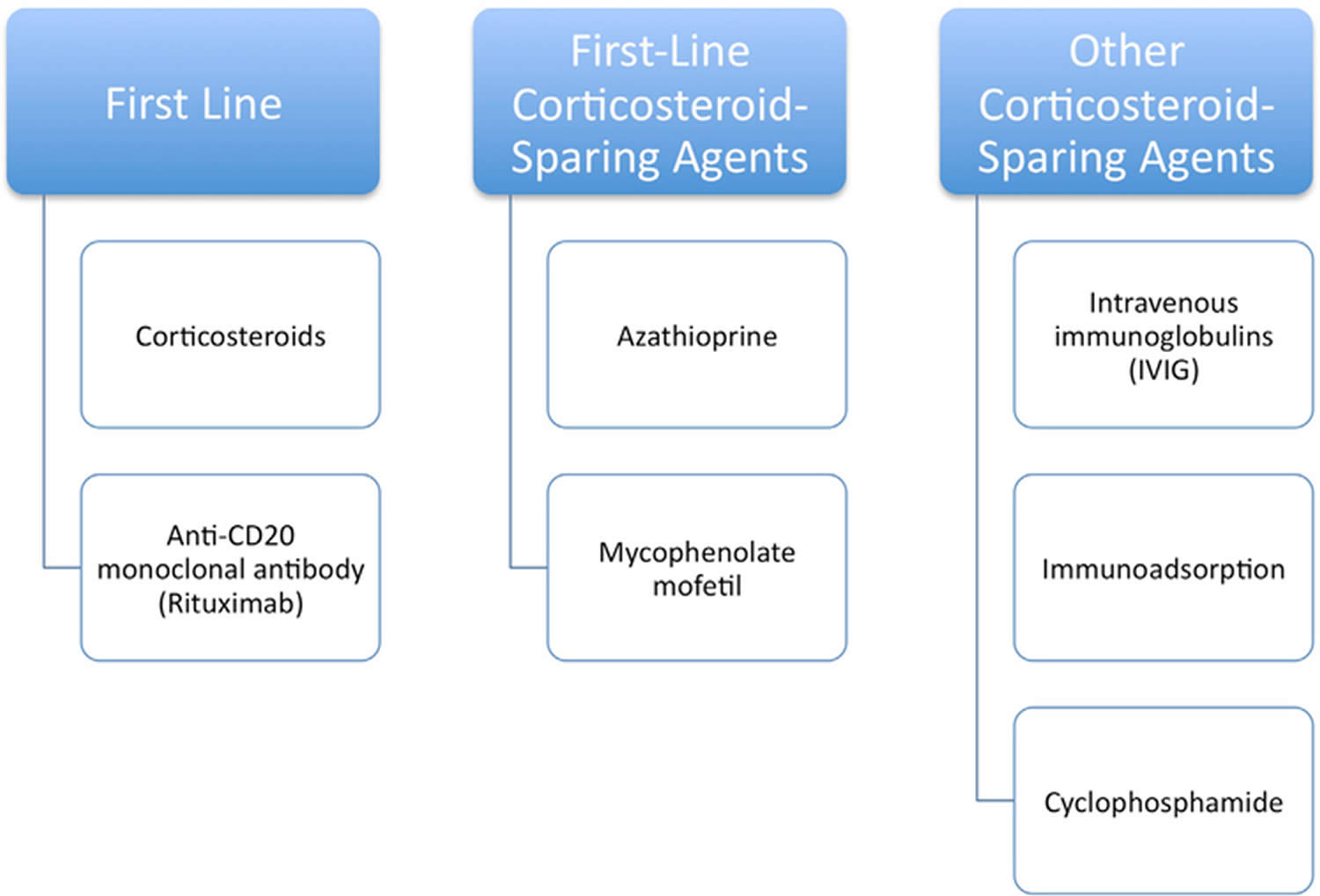
Pemphigus foliaceus treatment is to prevent new areas from developing bacterial infections and promote healing of affected areas. Topical treatment with corticosteroids is usually all that is necessary for mild pemphigus foliaceus. For more severe cases treatment is similar to that for pemphigus vulgaris. Corticosteroids represent the first-line treatment in all forms of pemphigus 2. Prednisone at 0.5 to 1.5 mg/kg per day or prednisolone 20 to 40 mg per day may be used 2. If initial control is not reached within 2 weeks, a higher prednisone dose is advised 2. Considering the serious side effects of high and prolonged doses of systemic corticosteroid therapy, systemic immunomodulatory medication is recommended, especially with azathioprine, mycophenolate mofetil, or methotrexate, the most important of which is azathioprine. For severe and refractory cases, rituximab, intravenous immunoglobulin (IVIG), and eventually cyclophosphamide can be indicated 1. Oral antibiotics may be prescribed to treat secondary bacterial infection.
Work-up before corticosteroid or immunosuppressive therapy 3:
- Complete blood count (CBC)
- Creatinine, blood electrolyte levels
- Transaminases, g-glutamyltransferase (GGT), alkaline phosphatase (ALP) levels
- Total serum protein, albumin level
- Fasting serum glucose level
- Hepatitis B, hepatitis C, and HIV
- Quantiferon gold or Purified Protein Derivative (PPD) test also known as a tuberculin skin test (TST) or Mantoux test is recommended
- Recommended, on indication or optional:
- Serum IgA deficiency should be ruled out prior to IVIG treatment
- Analysis of thiopurine methyltransferase activity is recommended when azathioprine is considered in countries where genetic polymorphisms for decreased thiopurine methyltransferase activity levels are more common
- Chest radiograph if Quantiferon gold or tuberculin skin test (TST) is abnormal
- Beta-hCG test is recommended to exclude pregnancy in women of childbearing potential
- Osteodensitometry is recommended prior to corticosteroid treatment
- Ocular examination (glaucoma, cataract) is recommended
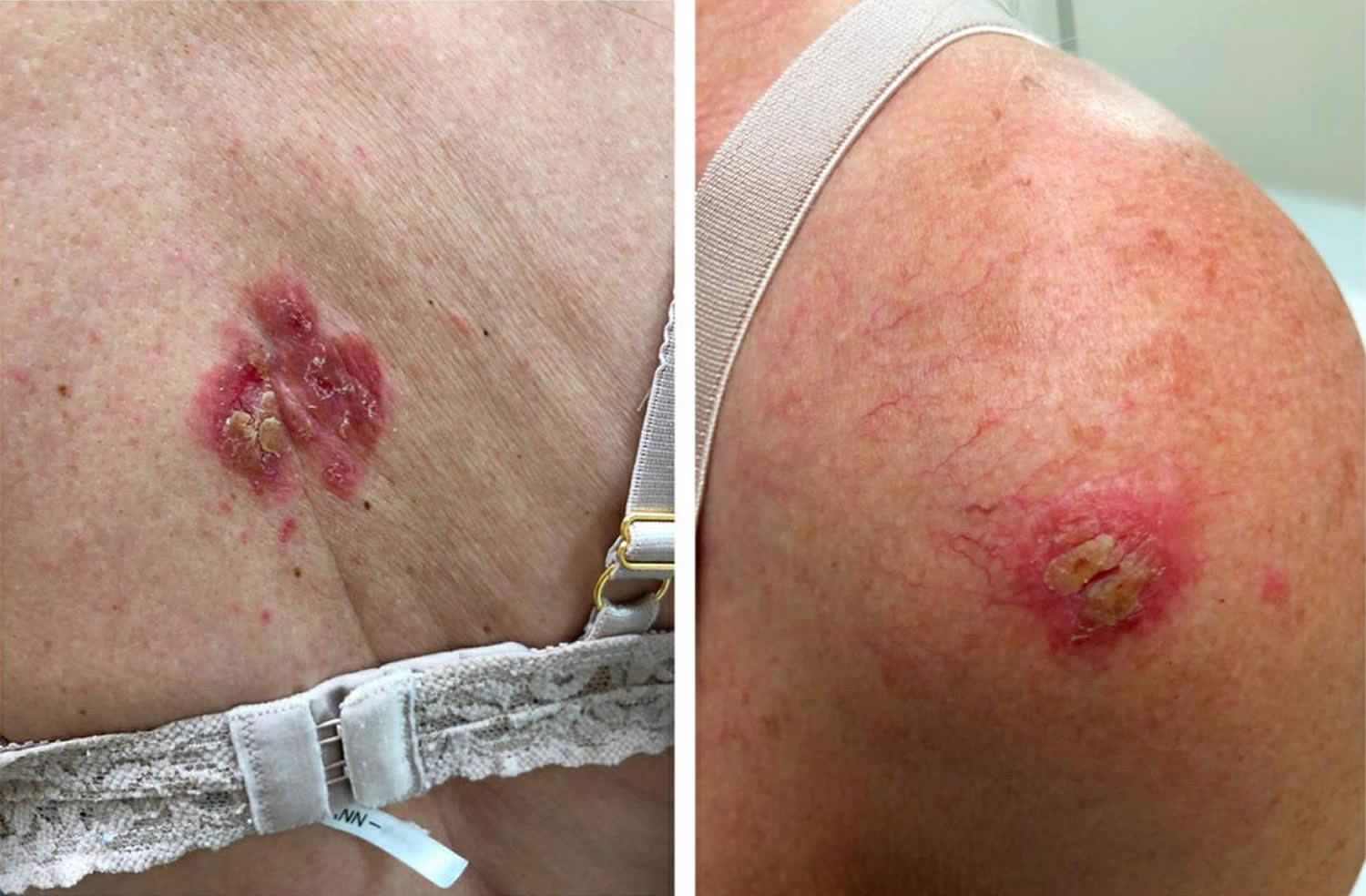
Figure 1. Pemphigus foliaceus
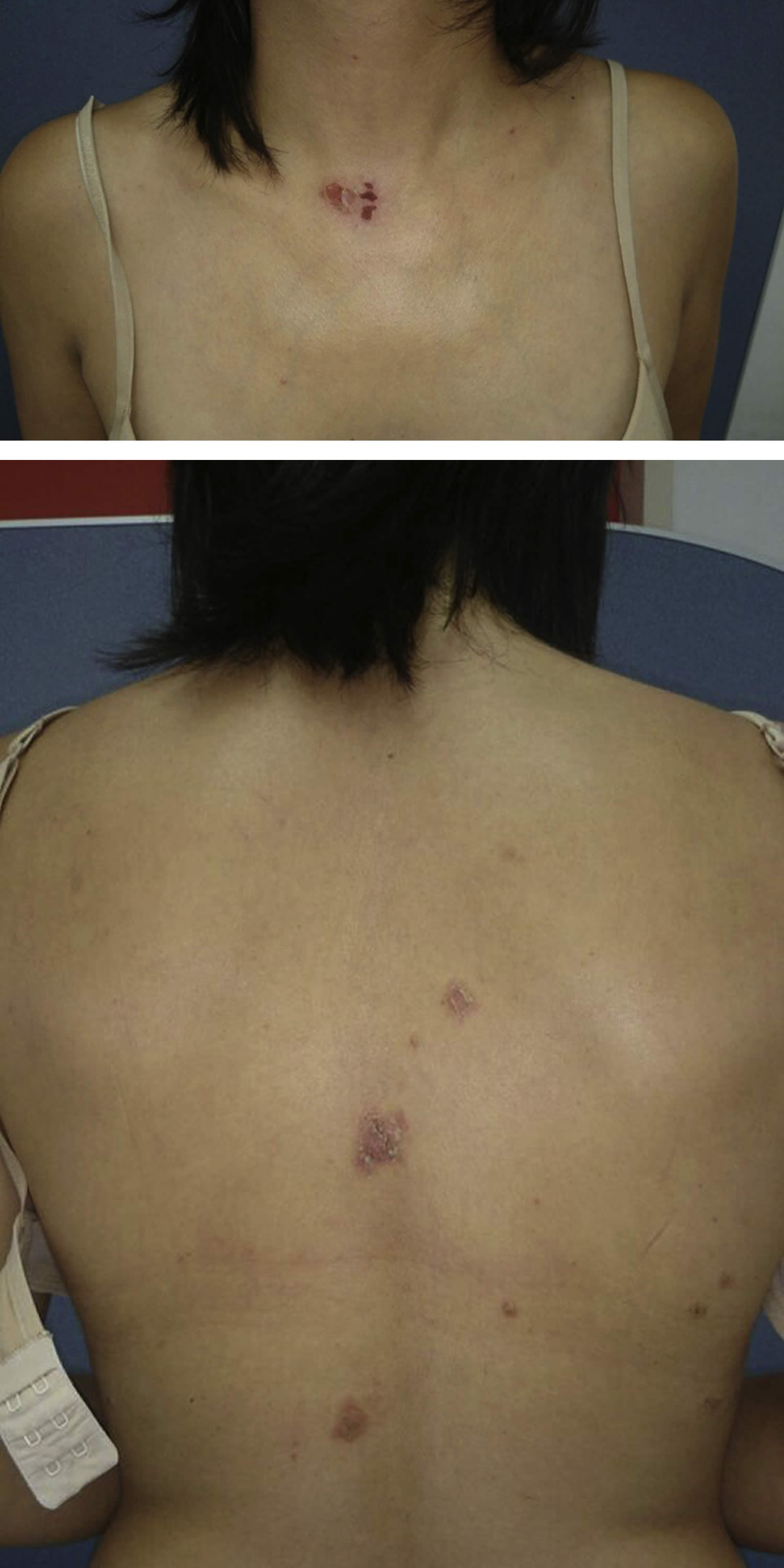
[Source 26 ]Figure 2. Drug induced pemphigus foliaceus
Footnotes: Pemphigus foliaceus due to taking oral St John’s wort (Hypericum perforatum). A 37-year-old woman with no personal or family history of disease, who suffered from a itchy rash on her trunk for 2 months. Initial lesions were small vesicles that rapidly developed into crusted erosions. One week before the appearance of skin lesions, she started taking St. John’s wort orally (900 mg/day) as a remedy for fatigue associated with insomnia. Skin examination found several erosive crusted and well-defined patches with reddish edges on her lower neck and upper back. No lesions appeared on the limbs, face, cheeks, mucous membranes, nails, and scalp. Nikolsky’s sign was negative. A skin biopsy found an intraepidermal blister and inflammatory dermal infiltrate composed of lymphocytes and eosinophils. Direct immunofluorescence (DIF) on perilesional skin found deposits of IgG and C3 on keratinocytes’ surface and along the dermo-epidermal junction. Enzyme-linked immunosorbent assay screening found a high titer of antidesmoglein 1 antibodies (108 U/mL) and low titer of antidesmoglein 3 antibodies (0.85 U/mL). According to the clinical and diagnostic features, pemphigus foliaceus was diagnosed. Oral prednisone, 0.5 mg/kg/d and clobetasol propionate, 0.05% cream daily were prescribed; however, within 1 month there was no clinical improvement. Consequently, the patient was treated with azathioprine, 100 mg/d, and St. John’s wort use was discontinued. After 2 months, the disease completely resolved with no residual skin damage. Azathioprine was discontinued after 10 months of remission. The patient had no new active skin lesions for 12 months, during which she didn’t receive any treatment.
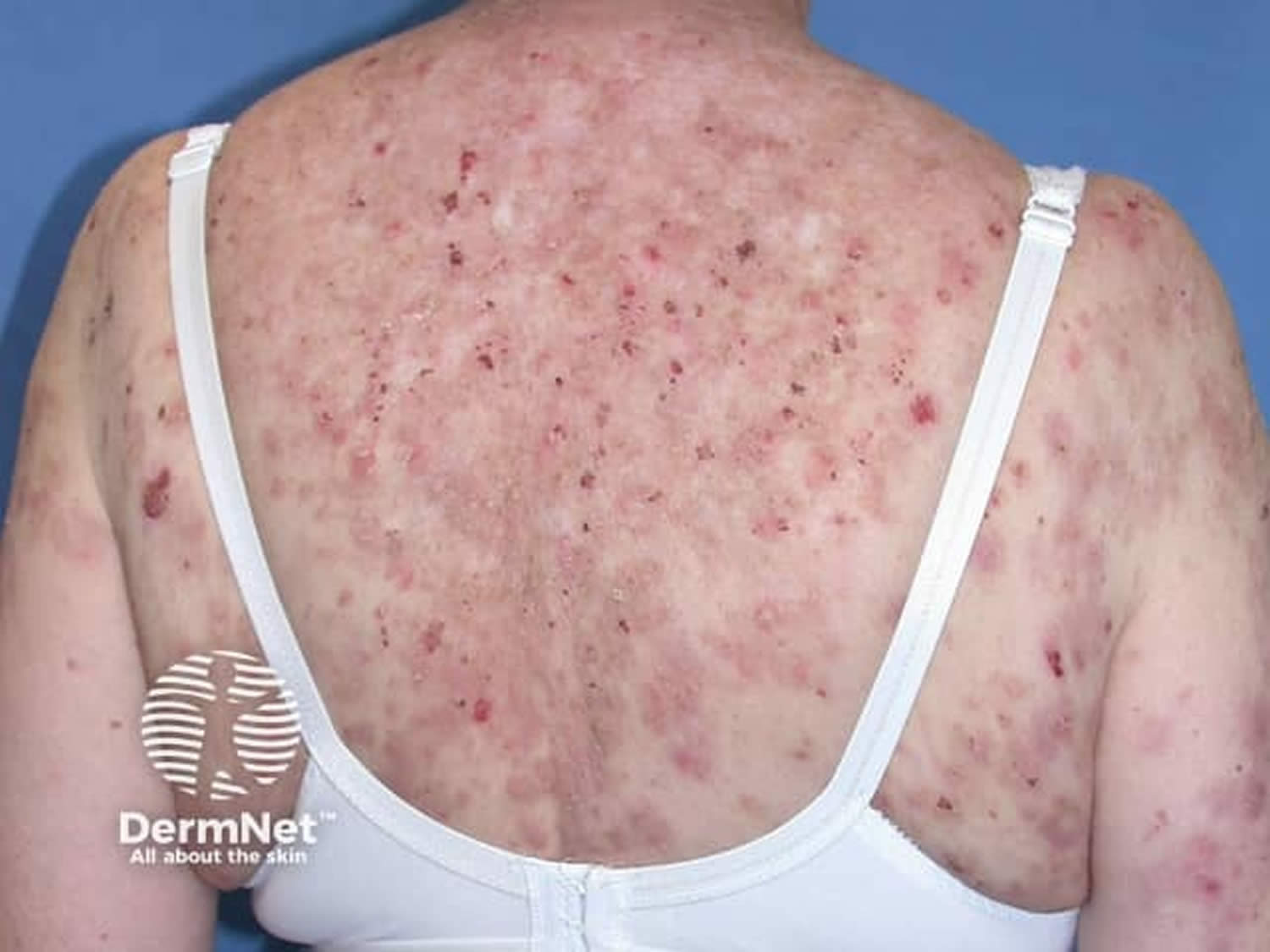
[Source 27 ]Figure 3. Pemphigus foliaceus
Footnote: Pemphigus foliaceus with hyperkeratotic plaques on the mid aspect of the back and left shoulder.
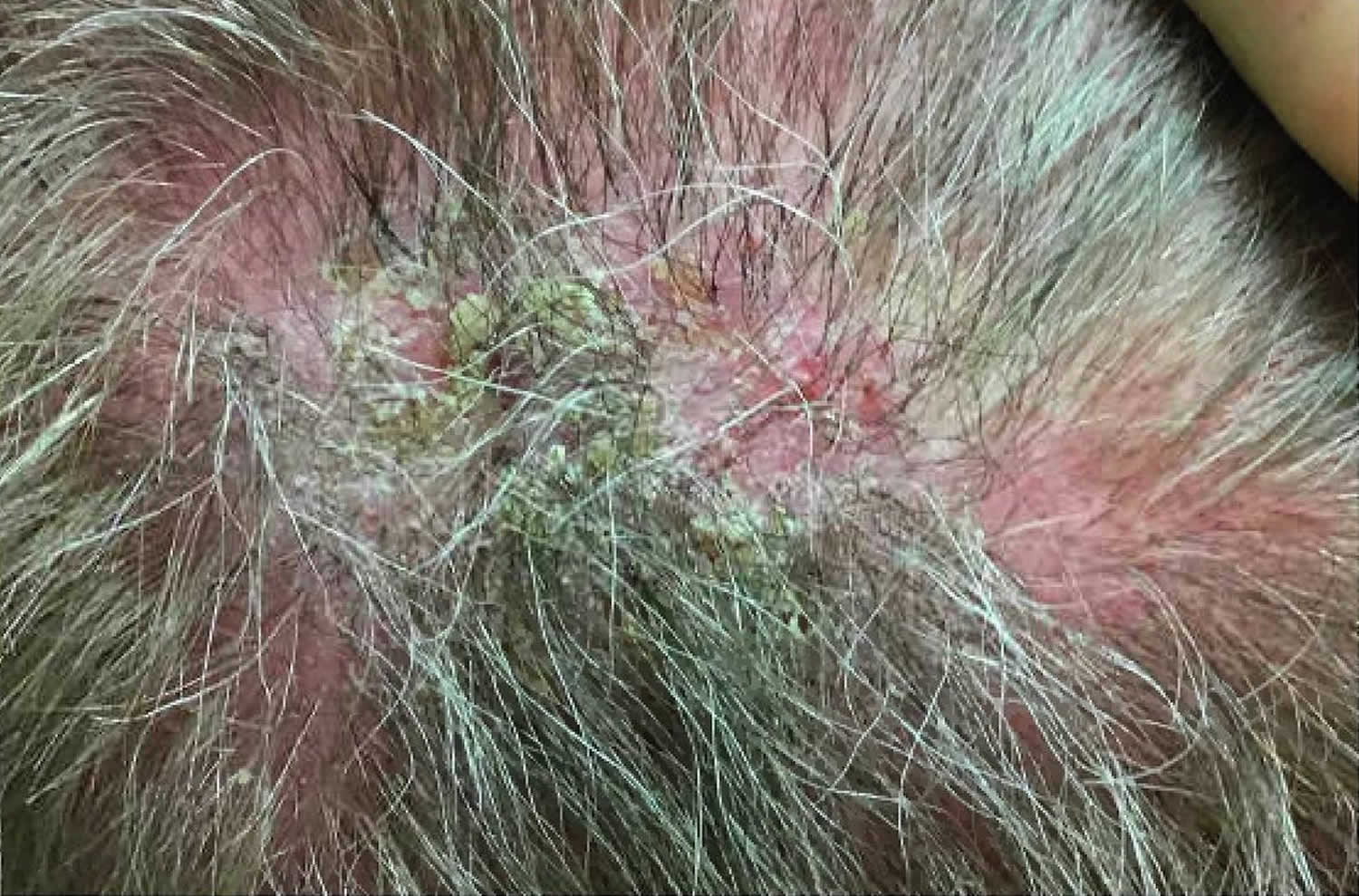
[Source 28 ]Figure 4. Pemphigus foliaceus scalp
Footnote: Pemphigus foliaceus scalp with thin plaques and cornflake scales and superficial erosions on the scalp.
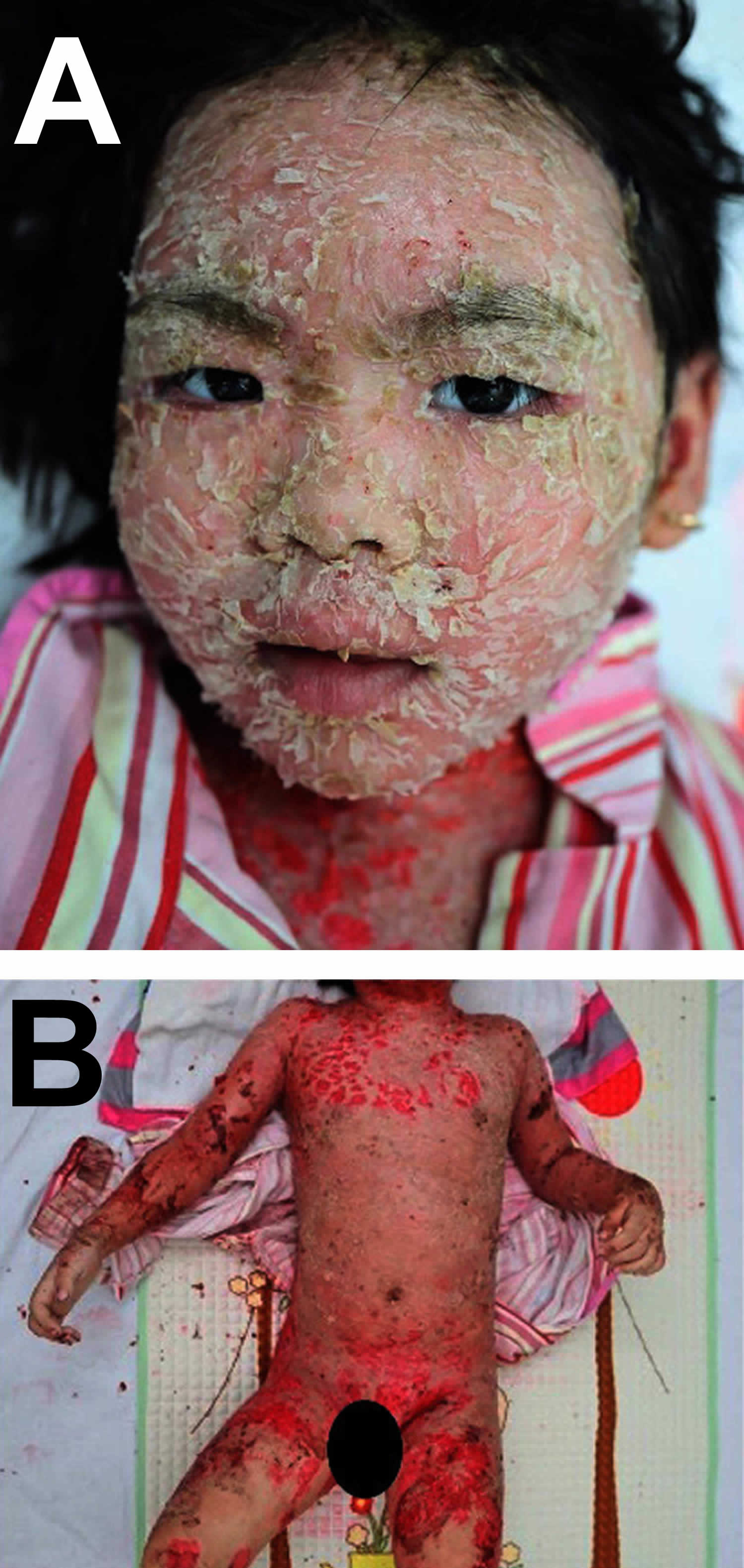
[Source 29 ]Figure 5. Pemphigus foliaceus (severe)
Footnotes: Severe pemphigus foliaceus. (A) Large flaky scales in the background of diffuse erythema on the face of a pemphigus foliaceus patient. (B) Diffuse scaly and crusted lesions on the trunk and extremities of a pemphigus foliaceus patient.
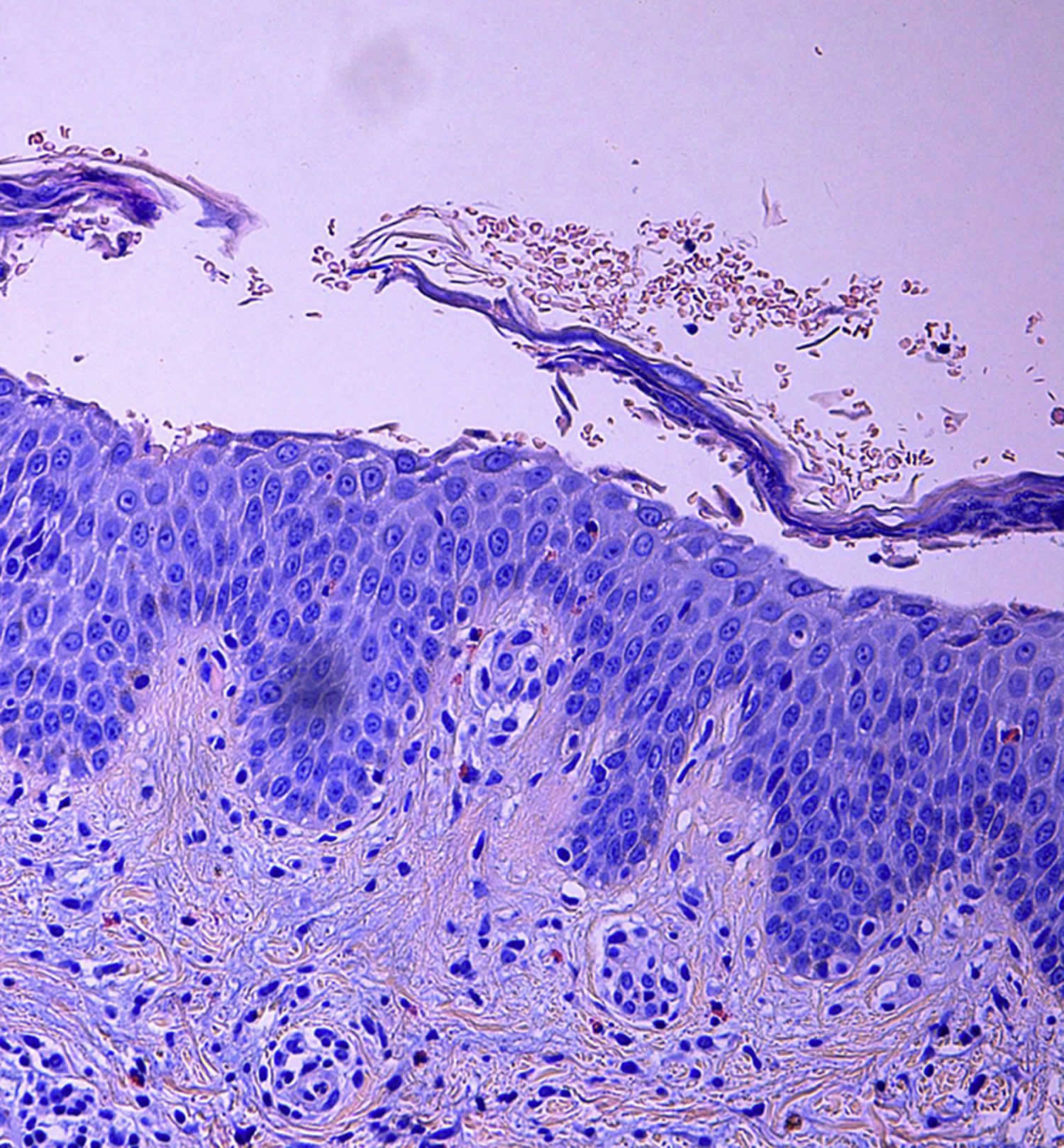
[Source 30 ]Figure 6. Skin biopsy of a pemphigus foliaceus patient with hematoxylin-eosin staining
Footnotes: Intraepidermal subcorneal blister with acantholytic keratinocytes and eosinophils. Inflammatory superficial dermal infiltrate composed of lymphocytes and eosinophils (Hematoxylin-eosin stain; original magnification: ×20).
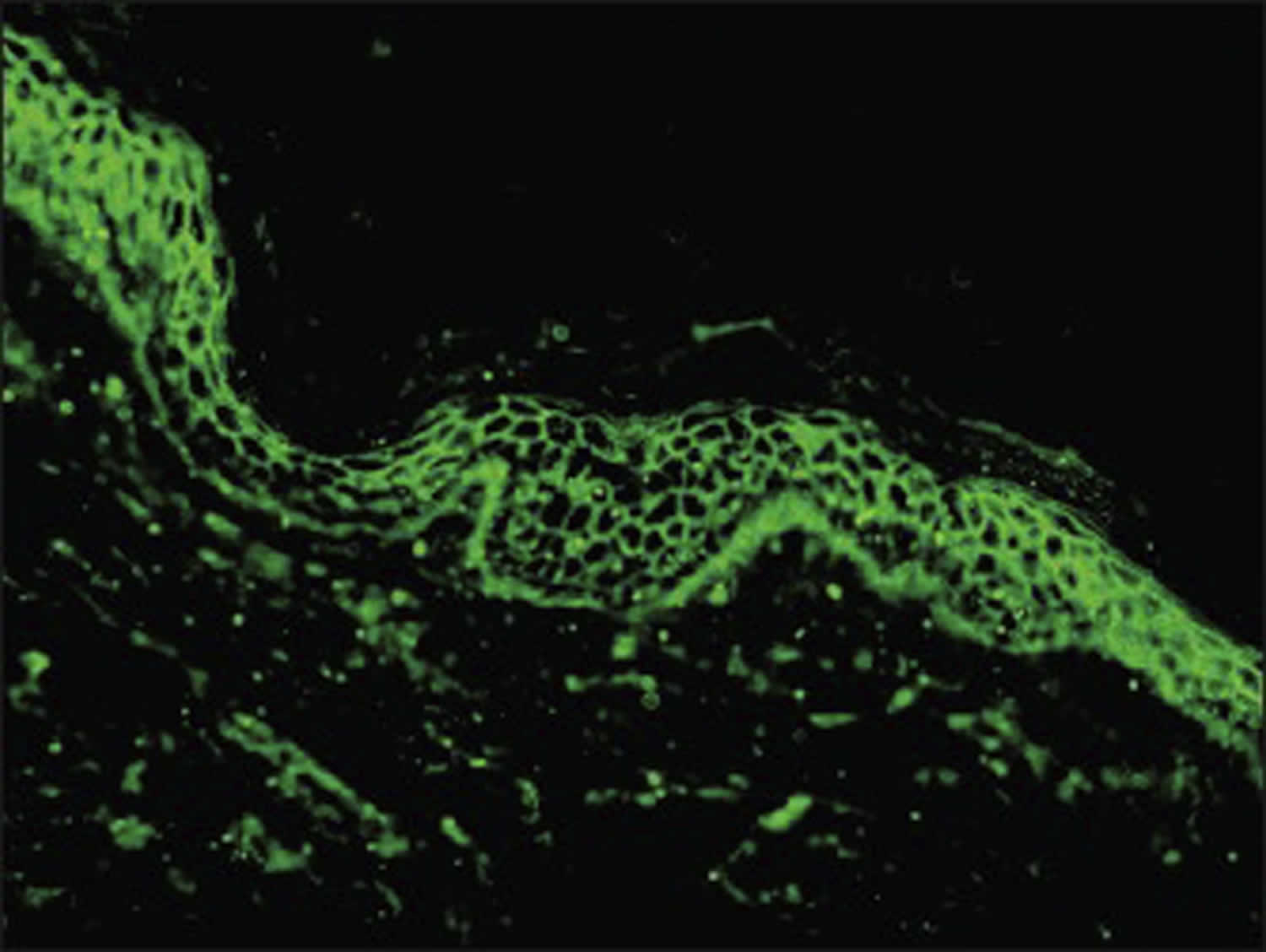
[Source 27 ]Figure 7. Pemphigus foliaceus direct immunofluorescence
Footnotes: Positive direct immunofluorescence (DIF) findings with deposits of IgG and C3 on keratinocytes’ surface and along the dermo-epidermal junction of a pemphigus foliaceus patient.
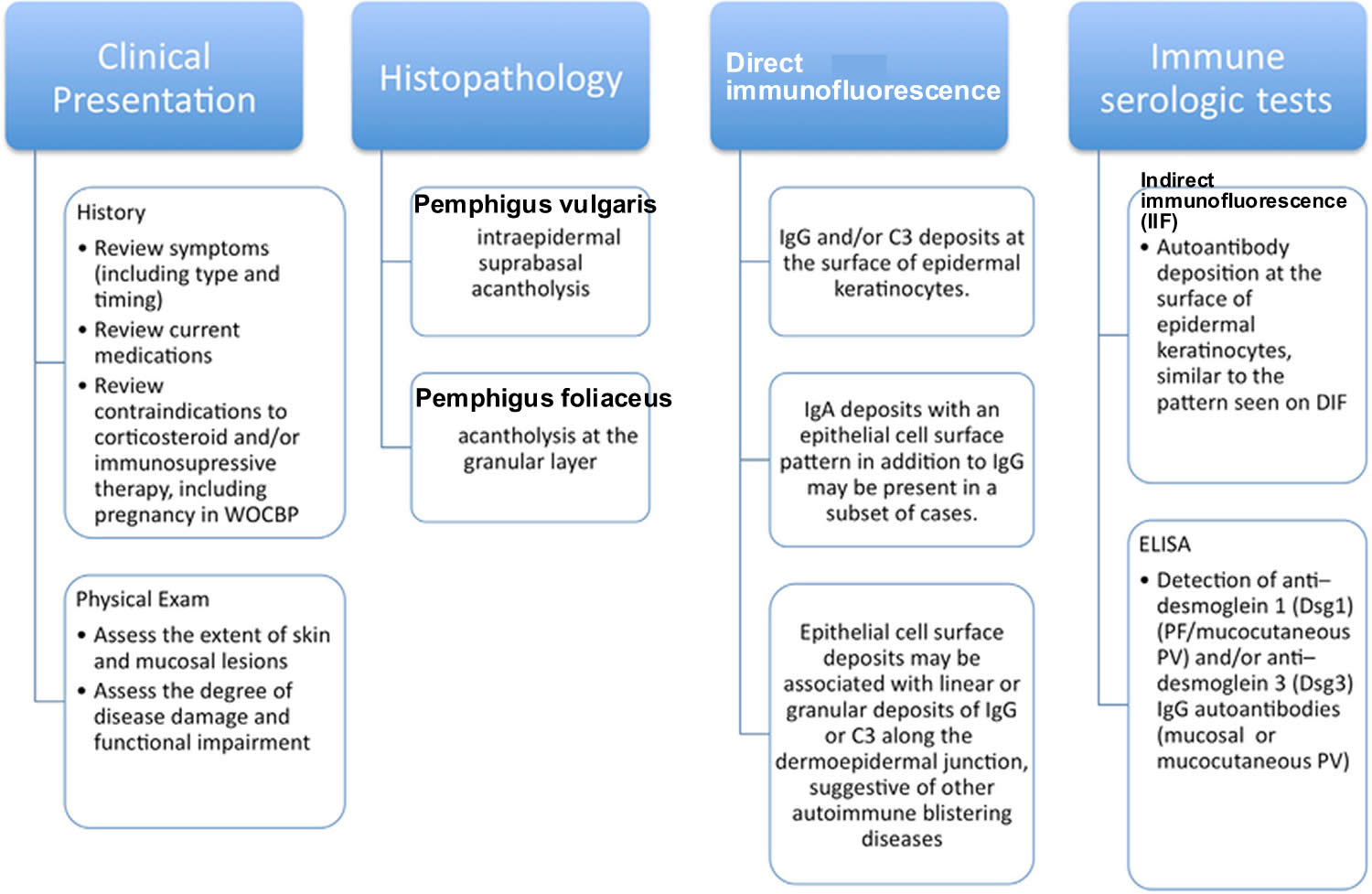
[Source 27 ]Figure 8. Diagnosis of pemphigus
Footnotes: Diagnosis of pemphigus requires clinical presentation and histopathology that are consistent with pemphigus and either a positive direct immunofluorescence (DIF) microscopy result or serologic detection of autoantibodies against epithelial cell surface antigens.
Abbreviations: ELISA = enzyme-linked immunosorbent assay; PF = pemphigus foliaceus; PV = pemphigus vulgaris; WOCBP = women of childbearing potential.
[Source 3 ]Figure 9. Pemphigus foliaceus treatment options
Footnotes: The principal objective is to promote the healing of blisters and erosions, prevent development of new lesions, and minimize serious side effects of treatment.
[Source 3 ]Figure 10. Pemphigus foliaceus treatment algorithm
Abbreviations: FS = Fogo selvagem (endemic pemphigus foliaceus); BSA = body surface area (1% means the sum of injured areas corresponding to the palm area); CS = corticosteroid; DDS = diamino-diphenil-sulfone or dapsone; AZA = azathioprine; MMF = mycophenolate mofetil; MTX = methotrexate; IVIg = intravenous immunoglobulin.
[Source 1 ]Who gets pemphigus foliaceus?
Pemphigus foliaceus affects people of all races, age and sex. Pemphigus foliaceus appears most commonly between the ages of 50-60 years.
Can pemphigus foliaceus be cured?
Pemphigus foliaceus is a long-lasting (chronic) condition. However it usually can be well controlled with treatment. There will be occasions when it flares up and other occasions when it improves. Currently, there is no way of predicting when flares will occur or how severe they will be. Treatment is aimed at controlling the condition and preventing flares.
Is pemphigus foliaceus hereditary?
Pemphigus foliaceus does not pass from generation to generation. However, genetic factors (the genes we inherit from our parents) are thought to be important if someone develops pemphigus foliaceus in combination with environmental factors.
Pemphigus foliaceus causes
Pemphigus foliaceus is an autoimmune disorder, that means your immune systems starts reacting against your tissue.
The building block cells of the skin epidermis are called keratinocytes. These cells are cemented together at special sticky spots called desmosomes. In pemphigus foliaceus, autoantibodies
bind to a protein called desmoglein-1 (Dsg1), a 160-KDa desmosomal cadherin, which is found in desmosomes in the keratinocytes near the top of the epidermis. The result is the surface keratinocytes separate from each other, and are replaced by fluid or blister. Because the blister is very close to the surface of the skin, the blisters rupture easily. In most cases, the autoantibodies are immunoglobulin type G (IgG) but in IgA pemphigus, the autoantibodies are immunoglobulin A (IgA).
Pemphigus foliaceus is sometimes provoked by sun exposure.
Endemic pemphigus foliaceus occurs in South America, where it is commonly known as Fogo selvagem. It appears to be set off by a virus transmitted by an insect bite (black fly – Simulium nigrimanum) 1.
Pemphigus foliaceus can occur in genetically related relatives (~20%) and has been correlated with the presence of HLA-DR4, DR-14, and DR-1, but unlike pemphigus vulgaris, no single allele has been associated with the disease 31, 32, 33, 34.
Penicillamine, nifedipine, captopril, enalapril or nonsteroidal anti-inflammatory drugs (NSAIDs) most often provoke drug-induced pemphigus foliaceus. Other drugs include penicillins, cephalosporins and rifampicin 35, 36. If the drug is stopped, there is a 50% chance the pemphigus foliaceus will clear up.
Pemphigus foliaceus pathology
Experimental evidence suggests that autoantibodies (antibodies made against a person’s own body) in all forms of pemphigus foliaceus are pathogenic 2. Those autoantibodies recognize desmoglein-1 (Dsg1), a 160-KDa transmembrane glycoprotein of the cadherin family (calcium-dependent cell adhesion molecules) in desmosomes, which is expressed more strongly in the skin from the upper trunk. Desmoglein-1 (Dsg1) is present but only weakly in mucosae, accounting for the lack of mucosal involvement in pemphigus foliaceous 2. Some patients have also desmoglein-3 antibodies in the absence of clinical evolution 37. Based on the desmoglein compensation theory, desmoglein-1 (Dsg1) expression is more intense in the upper portions of the epidermis, whereas in pemphigus vulgaris, desmoglein-3 (Dsg-3) predominates in the lower layers of the epidermis and in mucous membranes 9, 38, 39.
Patients with both sporadic and endemic forms of pemphigus foliaceus have anti-desmoglein-1 (anti-Dsg1), the titer correlating both with the extent and activity of the disease 40. Autoantibodies that trigger pemphigus foliaceus belong to the immunoglobulin G4 (IgG4) subclass. Normal controls in endemic areas have low levels of IgG1 and IgG4, but in endemic pemphigus foliaceus (Fogo selvagem) patients, the IgG1 levels are the same but IgG4 is significantly elevated by up to 19-fold 1. Immunoglobulin G4 (IgG4) is pathogenic in the mouse model of endemic pemphigus foliaceus (Fogo selvagem). In the preclinical phase, IgG1 autoantibodies are directed against the EC5 ectodomains of desmoglein-1 (Dsg1) and in the disease phase, IgG4 autoantibodies react against EC1-2-a phenomenon that is known as epitope spreading 41, 42, 43, 44.
Some pemphigus foliaceus cases have been associated with the use of certain drugs. In patients receiving penicillamine, pemphigus foliaceus is seen more commonly than pemphigus vulgaris, with a ratio of 4:1 2. Penicillamine and captopril contain sulfhydryl groups that are speculated to interact with the sulfhydryl groups in desmoglein-1 (Dsg1) and desmoglein-3 (Dsg3). Most patients with drug-induced pemphigus go into remission after the offending drug is discontinued 45.
Pemphigus foliaceus signs and symptoms
Pemphigus foliaceus produces superficial thin-walled fragile blisters (collections of clear fluid within the skin) confined to the skin that burst easily, leaving raw areas known as erosions, without involvement of mucous membranes. Erosions are sores that can ooze fluid or become crusty and infected. This can lead to discoloration of the skin.
People with pemphigus foliaceus is usually otherwise in good health. Patients may complain of burning, itch, and pain. The onset is usually subtle with a few scattered, crusted lesions involving the seborrheic areas: scalp, face, chest, and upper back. Small fluid-filled blisters first form on the trunk. Being superficial within the upper epidermis, they rupture very easily, and only erosions may be seen. On the face, scalp and upper trunk, the lesions are often scaly and crusty on a red and inflamed base. A burning sensation or localized pain may be felt.
Blisters are typically not found or clinically evident because they are superficial and rupture easily, often only the resultant crust and scale are seen. The Nikolsky sign is present. Pemphigus foliaceus may stay localized for years, or it may rapidly progress to generalized involvement, resulting in exfoliative erythroderma. Mucous membrane involvement is uncommon, even with widespread disease.
Pemphigus foliaceus diagnosis
It is advisable that your family doctor refers you to a skin specialist (dermatologist). A dermatologist can make a provisional diagnosis of pemphigus foliaceus by examining the affected area of your skin with a complete review of the medications you’re taking to exclude the possibility of drug-induced pemphigus foliaceus.
Pemphigus foliaceus diagnosis generally requires a skin biopsy of a fresh lesion for histology to determine the site of blister formation, which shows typical features of rounded-up separated keratinocytes called acantholytic cells within the blisters in the upper layers of the epidermis. Pemphigus foliaceus is confirmed by direct immunofluorescence (DIF) staining of the skin sections to reveal antibodies. In some cases, circulating antibodies can be detected by a blood test using indirect immunofluorescence test (IIF) or enzyme-linked immunosorbent assay (ELISA).
A skin biopsy specimen of a recent (less than 24 hour) small vesicle consisting of the peripheral portion of a blister (1/3 of the sample) and perilesional skin (placed in 4% formalin solution) (2/3 of the sample) should be obtained for routine histopathologic analysis: acantholysis at the granular layer in pemphigus foliaceus or intraepidermal suprabasal acantholysis in pemphigus vulgaris.
The hallmark of pemphigus is the finding of immunoglobulin G (IgG) autoantibodies against the cell surface of keratinocytes with direct immunofluorescence (DIF). The diagnosis of pemphigus should be seriously questioned if the test result of direct immunofluorescence (DIF) of skin biopsy is negative. It is important that the skin biopsy for direct immunofluorescence (DIF) be performed on the normal-appearing perilesional skin, as immune reactants can be difficult to detect in the blistered epidermis. Skin biopsy of perilesional skin (≤1 cm from a recent lesion), put into a cryotube for transportation in saline (delivery <36 hour) in a cylinder of liquid nitrogen or Michel’s fixative for direct immunofluorescence (DIF) microscopy analysis 3.
Direct immunofluorescence (DIF) and indirect immunofluorescent (IIF) findings are usually indistinguishable from pemphigus vulgaris, with intercellular immunoglobulin G (IgG) and C3 throughout the epidermis 3. Occasionally intercellular staining is restricted to the upper layers of the epidermis, both on direct and indirect immunofluorescence.
- Direct immunofluorescence (DIF) microscopy: IgG and/or C3 deposits at the surface of epidermal keratinocytes. The smooth and reticular staining pattern is also referred to as chicken wire, honeycomb, or fishnet-like 3
- IgA deposits with an epithelial cell surface pattern in addition to IgG may be present in a subset of cases 3
- Epithelial cell surface deposits may be associated with linear or granular deposits of IgG or C3 along the dermoepidermal junction, suggestive of other autoimmune blistering diseases (including paraneoplastic pemphigus or pemphigus erythematosus) or the coexistence of pemphigus and pemphigoid 3
Indirect immunofluorescence (IIF) is positive in over 85% of pemphigus foliaceus and enzyme-linked immunosorbent assay (ELISA) detects anti-desmoglein-1 antibodies in up to 71% of patients, but immunoblotting is less sensitive 24. In general, the ELISA index correlates with the extent and/or activity of disease and prognostic value for relapse, helping to guide treatment. Enzyme-linked immunosorbent assay (ELISA) can aid in monitoring disease activity and the response to treatment 1.
Recently, trichoscopy has been evaluated as a useful tool in the differential diagnosis of scalp pemphigus. Extravasations and yellow hemorrhagic crusts were the most frequent findings. The “fried egg sign” (yellow dots with a whitish halo) was identified as a new trichoscopic feature of pemphigus 25.
Histopathology
The histopathology of early blisters in pemphigus foliaceus demonstrates separation of keratinocytes (acantholysis) just below stratum corneum and in the granular layer 2. The stratum corneum is often lost from the surface. The deeper epidermis remains intact 2. Histological features may not be diagnostic in the early stages, and eosinophilic spongiosis is an early manifestation 2.
In early lesions, vacuoles form in the intercellular spaces in the upper levels of the epidermis 2. These coalesce to form clefts and superficial bullae high in the granular layer or immediately below the stratum corneum. Bullae contain fibrin, neutrophils, and scattered acantholytic keratinocytes. These superficial blisters are histologically indistinguishable from those seen in staphylococcal scalded skin syndrome or bullous impetigo because desmoglein-1 (Dsg1) is targeted in both of these diseases.
Older lesions are acanthotic, papillomatous, and hyperkeratotic with focal parakeratosis 2. Dyskeratotic cells in the granular layer of older lesions distinguish pemphigus foliaceus from pemphigus vulgaris. The dermis may show an inflammatory infiltrate of eosinophils and neutrophils 3, 46.
Pemphigus foliaceus differential diagnosis
Pemphigus foliaceus differential diagnosis includes other forms of pemphigus, bullous impetigo, subcorneal pustular dermatosis, subacute cutaneous lupus erythematosus, and seborrheic dermatitis. The demonstration of immunoglobulin G (IgG) autoantibodies against epidermal surfaces is essential to determine if the disorder accounts for a form of pemphigus 2.
Pemphigus foliaceus treatment
The aims of treatment are to prevent new blisters forming and to heal broken areas of skin. Treatment involves long-term use of topical and oral medicines. In severe cases, intravenous medication may be needed as a hospital in-patient. Most treatments work by suppressing your immune system.
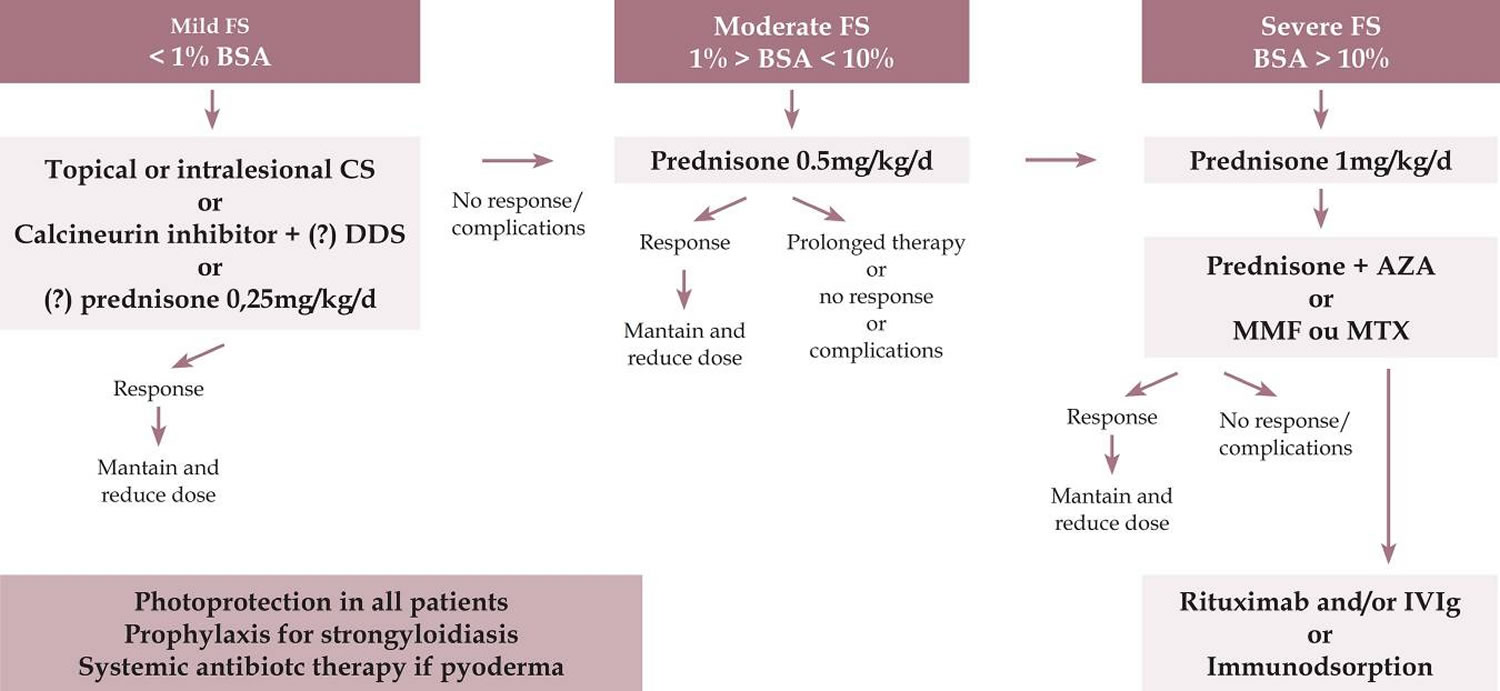
In deciding which treatment regimen to prescribe for a patient with pemphigus foliaceus, the extent of pemphigus should be determined. A practical method would be to consider up to 1% involvement as a mild clinical form, up to 10% as moderate, and over 10% of compromised body area (pemphigus foliaceus active lesions) as severe, with 1% representing the sum of the area of pemphigus foliaceus lesions, corresponding approximately to the hand palmar area 1. In some hospitals, a protocol is included to verify extent and severity of the lesions using the Pemphigus Area and Activity Score (PAAS) and Autoimmune Bullous Skin Disorder Intensity Score (ABSIS) scores 47, 48.
Prior to initiation of therapy, complete blood count (CBC), creatinine, sodium, potassium, transaminases, gamma-glutamyl transferase (GGT), alkaline phosphatase (ALP), total proteins and protein fractions, fasting blood glucose, serology for hepatitis B and C and human immunodeficiency virus (HIV), and chest x-ray should be examined 1. Optional recommendations include: ruling out IgA deficiency prior to intravenous immunoglobulin (IVIG); measuring thiopurine methyltransferase activity before azathioprine; abdominal ultrasonography (optional), Purified Protein Derivative (PPD) test also known as a tuberculin skin test (TST) or Mantoux test, or Quantiferon if the risk for tuberculosis is high; measuring activity of glucose-6-phosphate dehydrogenase (G6PD), bilirubins, and reticulocytes prior to dapsone; beta-HCG (human chorionic gonadotrophin) to exclude pregnancy; bone densitometry before corticotherapy; and an ophthalmological evaluation to rule out glaucoma and cataract. Regardless of the parasitological stool examination, preventive treatment of strongyloidiasis is recommended, as is systemic antibiotic therapy if pyoderma is present 1.
Corticosteroids represent the first-line treatment in all forms of pemphigus 2. In localized pemphigus foliaceus, with a limited number of lesions (up to 1% of body area), topical (moderate to high potency) or intralesional corticosteroid therapy (triamcinolone acetonide 2 to 3 mg/ml) is used 1. Associated with topical therapy, one can prescribe dapsone 50 to 100mg/day 1. In small series, dapsone monotherapy at higher doses (200-300mg/day) has been suggested to be effective in the initial treatment of certain patients with pemphigus foliaceus. In nonresponsive patients, low dosages of prednisone (up to 0.25 mg/kg/day) may be employed. Systemic corticosteroid therapy (prednisone/prednisolone) is prescribed when topical treatment does not control the disease or if the cutaneous condition worsens, as evidence by an increase in lesion number, at a dosage of 0.5 mg/kg/day.
In severe disseminated pemphigus foliaceus (above 10% of body area), prednisone at 0.5 to 1.5 mg/kg per day or prednisolone 20 to 40 mg per day may be used 2. Systemic corticosteroid therapy remains the most widely used, recognized, and established treatment option, due to its high efficacy and rapid control. If initial control is not reached within 2 weeks, a higher prednisone dose is advised 2. No statistically significant difference has been observed in the time to disease control between higher and lower doses of systemic corticosteroids 1. There are also no differences in the average side effects. Therefore, treatment is started at smaller doses, increasing the dose until clinical control of the disease is attained 1. Equivalent doses of triamcinolone may be administered in pemphigus foliaceus-resistant patients.
If no new lesions develop within 2 weeks, a reduction in corticosteroid from 0.25 mg/kg every 2 weeks to 30 mg/day is suggested, with subsequent reductions of 5 mg every 2 weeks until 10 mg/day is reached. Subsequently, the dosage is reduced by 2.5 mg/week until 0, and adjuvant (corticoid-sparing drug) therapy is maintained.
Considering that the use of prolonged and high-dose systemic corticosteroids can lead to severe or even fatal side effects, it is necessary in patients with disseminated pemphigus foliaceus to administer a combination of corticoid-sparing drugs (adjuvants) early, including methotrexate, azathioprine, and mycophenolate mofetil. According to recent publications, the ideal combination is deflazacort and azathioprine. Factors that should be considered in the choice of adjuvant are its availability, cost, and side effects. The evidence indicates that the major benefit of adjuvant use is the corticoid-sparing effect-not in modifying the course of the disease. In addition, adjuvants reduce the risk of relapse without altering remission rates.
Azathioprine 1 to 3 mg/kg per day (beginning at 50 mg/day, increasing progressively until the total daily dose is reached) and mycophenolate mofetil 2 g per day (starting dosage of 1 g/day, with a gradual increase of 500mg/day to improve gastric tolerance) or mycophenolic acid (1440 mg/day) are effective add-ons (adjuvants) to oral steroids but the best time for its introduction remains unclear (early versus treatment-resistant cases) 2. The activity of thiopurine-methyl transferase should be measured, if possible, prior to azathioprine initiation, because if it is low, medication may compromise the bone marrow. It should be noted that the onset of action with mycophenolate mofetil is slow and evidence of response occurs between 2 and 12 months of continued use 2. Another possible side effect of mycophenolate is pancytopenia.
Other treatment options for refractory pemphigus foliaceus or in case of contraindications to immunosuppressive agents include hydroxychloroquine 200 mg twice daily, dapsone 100 mg per day or up to 1.5 mg/kg per day, methotrexate 10 to 20 mg/week, intravenous immunoglobulin 2 g/kg per month or rituximab (monoclonal anti-CD20) given as either 4 weekly infusions of 375 mg/m² or 2 × 1000 mg (2 weeks apart).
Methotrexate is an interesting option, based on its low cost and wide availability. However, it is liver toxic. Methotrexate is used at 7.5 to 25 mg/week, administered over 1 or 2 consecutive days. After 24 hours, folic acid should be prescribed at a dose of 5 mg. Alcohol, sulfamidic derivatives, and allopurinol are all banned 1.
Dapsone 100 mg/day or up to 1.5 mg/kg/day, because it has also corticosteroid-sparing effects. However, glucose-6-phosphate dehydrogenase (G6PD) activity should be evaluated beforehand. Considering that pemphigus is an antibody-mediated disease, dapsone use is controversial 1.
Cyclophosphamide can also be used at 500 mg IV as a bolus or 2 mg/kg/day. Cyclophosphamide has corticosteroid-sparing effects, but the risk of sterility, hemorrhagic cystitis, and secondary cancer should be considered 1.
Rituximab (monoclonal anti-CD20) is indicated when the patient is refractory to conventional therapy or if prednisone is required at dosages of higher than 10 mg/day in combination with an immunosuppressant for more than 6 months 1. Rituximab is administered as pulse therapy, preferably at a dose of 1 g IV, repeated after 15 days (rheumatoid arthritis protocol) or at 375 mg/m²/week in 4 sessions (protocol for lymphoma). Lower doses are ineffective 1. If necessary, the protocol should be repeated in 6 months. Rituximab can be combined with prednisone in a regression scheme of up to 4 months or with an immunosuppressive agent (up to 12 months) 1. Hypersensitivity to murine proteins should be excluded. Adverse effects include infections (up to 10%), reactions during infusion, and rarely Stevens-Johnson syndrome and progressive multifocal leukoence-phalopathy (potentially life-threatening complications) 1.
Historically, Rituximab (monoclonal anti-CD20) has been used for treatment-refractory pemphigus and for patients with severe side effects to conventional immunosuppressive treatments. However, recent data indicate that the combination of rituximab and prednisone may be effective as an initial treatment for pemphigus, allowing the doses of prednisone to be reduced 1. Rituximab (monoclonal anti-CD20) is associated with lower rates of side effects compared with high and prolonged doses of corticoid monotherapy.
In certain centers that specialize in the treatment of pemphigus (pemphigus foliaceus and pemphigus vulgaris), for moderate and severe clinical forms, rituximab, in combination with systemic corticosteroid therapy, has been used as initial and maintenance treatment in all patients.
Intravenous immunoglobulin (IVIG) is indicated for very severe, refractory patients, those who present with significant adverse effects, and severe and disseminated forms of pemphigus that require a more rapid clinical response 1. Infused immunoglobulin (IVIG) appears to accelerate pathogenic catabolism of IgG autoantibodies. Doses of 2 to 3 g/kg/cycle are recommended (cycle of 4 to 5 consecutive days) every 30 days 1. Systemic corticosteroid and adjuvant drugs are maintained and have been used in combination with rituximab. Aseptic meningitis is a rare side effect. IgA deficiency should be excluded before starting this treatment 1. Patients with IgA deficiency should receive IgA-depleted IVIG treatment 3.
Further research continues to find better treatments or combinations of treatment for pemphigus foliaceus. Immunoadsorption also known as immunoapheresis is a selective extracorporeal clearance technique that removes immunoglobulins, especially IgG1, IgG2, and IgG4, from the plasma 49. With immunoapheresis, circulating levels of immunoglobulins can be decreased by over 80%. It is another option for refractory or very severe patients and is available in advanced centers for autoimmune diseases. Monthly cycles of 4 consecutive days are performed, with 2.5 times the plasma volume being perfused per day. Immunoapheresis contraindications include severe systemic infection, severe cardiovascular disease, extensive hemorrhagic diathesis, hypersensitivity against components of the immunoadsorption column, and the use of angiotensin-converting enzyme inhibitors (ACE inhibitors) 1.
Due to the potential side effects of therapy, patients should be monitored closely. After discontinuation of systemic corticosteroids in patients with complete remission, adjuvant immunosuppression can also be reduced over 6 to 12 months. The discontinuation of therapy is based on the clinical presentation with a complete absence of active cutaneous lesions over several months. Negative or low ELISA-Dsg1 values or negative immunofluorescence are useful to support the discontinuation of therapy 3, 50, 51.
Other practical tips:
- Keep all of your appointments with healthcare professionals.
- Adhere to the blood monitoring regimen your doctor gives you.
- Take medicines as advised by your specialist and never stop taking corticosteroids suddenly without talking to your doctor.
- You may be provided with a steroid card to alert other healthcare professionals that you are taking this medication, and provide guidance on what to do if you become unwell.
Topical corticosteroids
Steroid creams and ointments are the most commonly prescribed treatment for pemphigus foliaceus. Topical corticosteroids are anti-inflammatory medications that work by suppressing the immune system. They relieve symptoms and calm inflammation and blistering. Higher strength steroids are usually needed and are typically applied once a day. When applied as directed by your healthcare professional, side effects are rare.
Oral corticosteroids
High doses of oral corticosteroids, usually prednisolone, are often given to bring pemphigus foliaceus under control (by suppressing the immune system). Once the condition is under control, the dose is then reduced slowly to minimise side effects. Some people need to continue taking a small daily dose to keep the disease in remission. Rarely, intravenous corticosteroids may have to be used in the form of IV pulse methylprednisolone.
- Systemic corticosteroid therapy prednisone at 0.5 to 1.5 mg/kg per day or prednisolone 20 to 40 mg per day.
- Systemic corticosteroids (oral or intravenous pulses) can be combined with an immunosuppressive adjuvant at the onset of therapy, especially in cases of increased risk of corticosteroid therapy, complications due to expected prolonged use (>4 months), or dose dependency above minimal therapy (>10 mg/day). However, there is limited evidence that the addition of adjuvants is superior to treatment with corticosteroids alone.
- Although limited, studies have not shown intravenous corticosteroid pulses to have an additional benefit on top of that of conventional first-line treatment with oral prednisone or prednisolone and immunosuppressive adjuvants. Although more evidence is needed, steroid pulse therapy in addition to conventional treatment should be reserved for refractory cases of pemphigus.
- Treat with the smallest dose for the shortest time possible to minimize risk of adverse events.
Immunosuppressants
People affected by pemphigus foliaceus often require an additional immunosuppressant agent. This is to allow the oral steroid dose to be weaned. Immunosuppressants work by dampening down the immune system and are given under the close supervision of a dermatology consultant. Options include 3:
- Azathioprine
- Mycophenolate mofetil
- Methotrexate
- Rituximab (anti-CD20 monoclonal antibodies)
These medications can have side effects, so people who take them must be monitored carefully including regular blood tests. More information can be found in the individual patient information leaflets for the corresponding drug.
Azathioprine
Azathioprine is currently the first-line corticosteroid-sparing agents 3. Start azathioprine at 50 mg/day the first week to detect idiosyncratic reactions such as sudden-onset fevers, oral ulcers, elevated liver function test results and/or drug reaction with eosinophilia and systemic symptoms (and in that case stop immediately), and then raise to desired dose 3. Although not predictive for idiosyncratic reactions, thiopurine methyltransferase activity should be evaluated in countries/ethnicities where there is a higher incidence of polymorphisms before commencing therapy because recommended azathioprine doses vary depending on thiopurine methyltransferase activity. In general, adults with pemphigus and high thiopurine methyltransferase activity are treated with normal doses of azathioprine (≥2.5 mg/kg/day) 3. Patients with intermediate or low thiopurine methyltransferase activity should receive a lower maintenance dose (≥0.5 to 1.5 mg/kg/day) depending on level of enzyme activity. Patients who lack thiopurine methyltransferase activity should avoid treatment with azathioprine 3.
Mycophenolate mofetil
Mycophenolate mofetil (30 mg/kg to 45 mg/kg/day) or mycophenolic acid (1440 mg/day) are effective add-ons (adjuvants) to oral steroids but the best time for its introduction remains unclear (early versus treatment-resistant cases) 2. It should be noted that the onset of action with mycophenolate mofetil is slow and evidence of response occurs between 2 and 12 months of continued use 2. Another possible side effect of mycophenolate is pancytopenia.
Methotrexate
Methotrexate is an interesting option, based on its low cost and wide availability. However, it is liver toxic. Methotrexate is used at 7.5 to 25 mg/week, administered over 1 or 2 consecutive days. After 24 hours, folic acid should be prescribed at a dose of 5 mg. Alcohol, sulfamidic derivatives, and allopurinol are all banned 1.
Rituximab (anti-CD20 monoclonal antibodies)
Currently there are 2 intravenous CD20 inhibitors available, rituximab and ofatumumab. All the published trials so far have used rituximab 3.
Rituximab is the first-line treatment in new-onset moderate-to-severe pemphigus and/or for patients who do not achieve clinical remission with systemic corticosteroids and/or immunosuppressive adjuvants 52. A course of intravenous rituximab consists of 2 × 1000 mg (2 weeks apart) or 4 × 375 mg/m² (1 week apart). Rituximab allows for more rapid tapering of corticosteroid doses and a major corticosteroid-sparing effect. The incidence of unforeseen fatal infections such as progressive multifocal leukoencephalopathy cannot be estimated due to the rarity of pemphigus.
Rituximab treatment can be repeated in cases of clinical relapse or as early as 6 months after treatment. Lower doses are sometimes used for retreatment. Combine with short-term (<4 months) systemic corticosteroids and long-term (>12 months) immunosuppressive treatment, although the need for immunosuppressive adjuvants in rituximab therapy remains unclear.
Other treatments
Cyclophosphamide, intravenous immunoglobulins (IVIG) and immunoadsorption (immunoapheresis) may be considered if other treatments are not effective.
Intravenous immunoglobulin (IVIG)
Intravenous immunoglobulin (IVIG) is indicated for very severe, refractory patients, those who present with significant adverse effects, and severe and disseminated forms of pemphigus that require a more rapid clinical response 1. Infused immunoglobulin (IVIG) appears to accelerate pathogenic catabolism of IgG autoantibodies. Doses of 2 to 3 g/kg/cycle are recommended (cycle of 4 to 5 consecutive days) every 30 days 1. Systemic corticosteroid and adjuvant drugs are maintained and have been used in combination with rituximab. Aseptic meningitis is a rare side effect. IgA deficiency should be excluded before starting this treatment 1. Patients with IgA deficiency should receive IgA-depleted IVIG treatment 3.
Cyclophosphamide
Cyclophosphamide is used in cases of limited resources or in severe cases that have not responded to other treatments. Cyclophosphamide is used as a drug of last resort on account of long-term side effects.
Cyclophosphamide can be used at 500 mg IV as a bolus or 2 mg/kg/day. Cyclophosphamide has corticosteroid-sparing effects, but the risk of sterility, hemorrhagic cystitis, and secondary cancer should be considered 1.
Immunoadsorption (immunoapheresis)
Immunoadsorption also known as immunoapheresis is a selective extracorporeal clearance technique that removes immunoglobulins, especially IgG1, IgG2, and IgG4, from the plasma 49. Immunoapheresis is a first-line treatment option in emergency situations or second-line corticosteroid-sparing agent where available. With immunoapheresis, circulating levels of immunoglobulins can be decreased by over 80%. It is another option for refractory or very severe patients and is available in advanced centers for autoimmune diseases. Monthly cycles of 4 consecutive days are performed, with 2.5 times the plasma volume being perfused per day. Immunoapheresis contraindications include severe systemic infection, severe cardiovascular disease, extensive hemorrhagic diathesis, hypersensitivity against components of the immunoadsorption column, and the use of angiotensin-converting enzyme inhibitors (ACE inhibitors) 1.
Tetracycline antibiotics
These are helpful for the inflammatory element of pemphigus foliaceus and used as a steroid sparing measure. These are used in the long term for their anti-inflammatory properties. Doxycycline is commonly used and is suitable for people with a penicillin allergy.
Maintenance after consolidation phase
Corticosteroid should be reduced progressively, after disease control or the end of the consolidation phase, reducing the corticosteroid dose by approximately 25% every 2 weeks until 20 mg/day and then slowly. If more than 3 lesions appear, return to the previous dose. If the disease recurs, return to the dose of the 2 previous phases. If it does not stabilize within 2 weeks, return to the starting dose. If the treatment is combined with an immunosuppressant, replace it or use rituximab, IVIG, or immunoadsorption. High levels of anti-Dsg-1 by ELISA indicate cutaneous relapses 1.
Clinical and laboratory monitoring
Clinical reassessments (skin and mucous membranes) should be performed every 2 weeks and after control, monthly. Significant side effects include diabetes mellitus, systemic arterial hypertension, and heart failure due to corticosteroid therapy; respiratory distress, anemia, and hepatitis (dapsone or methotrexate); respiratory infections and hepatitis (corticosteroid or immunosuppressants); mental disorders (corticosteroid); myopathy, osteoporosis, avascular bone necrosis, glaucoma, and cataract (corticosteroid); and hematological abnormalities (immunosuppressants).
Serological monitoring
Serology (Indirect immunofluorescence (IIF) and/or ELISA) should be performed at the outset of treatment, after 3 months, and then according to disease progression.
Treatment discontinuation
Discontinuation is based primarily on clinical signs and may be accompanied by anti-Dsg ELISA and indirect immunofluorescence (IIF). In some centers, negativity by indirect immunofluorescence (IIF) is recommended. Discontinue corticosteroid in patients with complete remission and minimal therapy, and discontinue adjuvants 6 to 12 months later.
Pemphigus foliaceus prognosis
Before the advent of steroid therapy (until the 1950s), pemphigus foliaceus was fatal in approximately 60% of patients and almost always fatal in elderly patients with concurrent medical problems 2. With steroid and immunosuppressive therapy, the mortality from pemphigus foliaceus has been dramatically reduced to 30% 1, 2. By combining steroid therapy with immunosuppressive adjuvants and recognizing and treating the adverse effects of systemic corticosteroids, the mortality rate was reduced to approximately 6% 1. Pemphigus foliaceus is now regarded as a benign disease that responds well to treatment and may remit 2. Approximately half of all patients experience remission but remain at risk of recurrence, even after years without treatment. The remaining patients keep their disease under control with low doses of therapy 1.
- Porro AM, Hans Filho G, Santi CG. Consensus on the treatment of autoimmune bullous dermatoses: pemphigus vulgaris and pemphigus foliaceus – Brazilian Society of Dermatology. An Bras Dermatol. 2019 Apr;94(2 Suppl 1):20-32. doi: 10.1590/abd1806-4841.2019940206[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Lepe K, Yarrarapu SNS, Zito PM. Pemphigus Foliaceus. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499829[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Murrell DF, Peña S, Joly P, et al. Diagnosis and management of pemphigus: Recommendations of an international panel of experts. J Am Acad Dermatol. 2020 Mar;82(3):575-585.e1. doi: 10.1016/j.jaad.2018.02.021[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Ioannides D, Hytiroglou P, Phelps RG, Bystryn JC. Regional variation in the expression of pemphigus foliaceus, pemphigus erythematosus, and pemphigus vulgaris antigens in human skin. J Invest Dermatol. 1991 Feb;96(2):159-61. doi: 10.1111/1523-1747.ep12460927[↩]
- Ganapati S. Eponymous dermatological signs in bullous dermatoses. Indian J Dermatol. 2014;59(1):21–23. doi:10.4103/0019-5154.123483 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3884922[↩]
- Ruocco V, Ruocco E, Lo Schiavo A, Brunetti G, Guerrera LP, Wolf R. Pemphigus: etiology, pathogenesis, and inducing or triggering factors: facts and controversies. Clin Dermatol. 2013;31:374–381. doi: 10.1016/j.clindermatol.2013.01.004[↩]
- Cunha PR, Barraviera SR. Autoimmune bullous dermatoses. An Bras Dermatol. 2009;84:111–124. doi: 10.1590/s0365-05962009000200003[↩]
- Porro AM, Caetano Lde V, Maehara Lde S, Enokihara MM. Non-classical forms of pemphigus: pemphigus herpetiformis, IgA pemphigus, paraneoplastic pemphigus and IgG/IgA pemphigus. An Bras Dermatol. 2014;89:96–106. doi: 10.1590/abd1806-4841.20142459[↩]
- Bystryn JC, Rudolph JL. Pemphigus. Lancet. 2005;366:61–73. doi: 10.1016/S0140-6736(05)66829-8[↩][↩]
- Diaz LA, Sampaio SA, Rivitti EA, Martins CR, Cunha PR, Lombardi C, et al. Endemic pemphigus foliaceus (fogo selvagem). I. Clinical features and immunopathology. J Am Acad Dermatol. 1989;20:657–669. doi: 10.1016/s0190-9622(89)70079-7[↩]
- Amagai M. Pemphigus. In: Bolognia JL, Lorizzo JO, Schaffer JV, editors. Dermatology. 2nd ed. Vol. 1. China: Elsevier Saunders; 2012. pp. 461–474.[↩]
- Hans-Filho G, Cunha PR. Fogo Selvagem (Endemic Pemphigus Foliaceus) In: Tyring SK, Lupi O, Hengge UR, editors. Tropical Dermatology. 2nd ed. Edinburgh: Elsevier; 2017. pp. 427–432.[↩]
- Hans-Filho G, Aoki V, Rivitti E, Eaton DP, Lin MS, Diaz LA. Endemic pemphigus foliaceus (fogo selvagem) – 1998. The Cooperative Group on Fogo Selvagem Research. Clin Dermatol. 1999;17:225–235. doi: 10.1016/s0738-081x(99)00014-0[↩]
- Shimizu H, editor. Shimizu’s Textbook of Dermatology. Nakayama Shoten Publishers. Hokkaido University Press; Japan: Oct 1, 2007.[↩]
- Sánchez-Pérez J, García-Díez A. Pénfigo [Pemphigus]. Actas Dermosifiliogr. 2005 Jul-Aug;96(6):329-56. Spanish. doi: 10.1016/s0001-7310(05)73090-8[↩]
- Waschke J. The desmosome and pemphigus. Histochem Cell Biol. 2008 Jul;130(1):21-54. doi: 10.1007/s00418-008-0420-0[↩]
- Chorzelski T, Jablońska S, Blaszczyk M. Immunopathological investigations in the Senear-Usher syndrome (coexistence of pemphigus and lupus erythematosus). Br J Dermatol. 1968 Apr;80(4):211-7. doi: 10.1111/j.1365-2133.1968.tb11961.x[↩]
- Ávalos-Díaz E, Herrera Esparza R. Dermatological autoimmune diseases. In: Anaya JM, Shoenfeld Y, Rojas-Villarraga A, et al., editors. Autoimmunity: From Bench to Bedside [Internet]. Bogota (Colombia): El Rosario University Press; 2013 Jul 18. Chapter 34. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459479[↩]
- Aslanova M, Yarrarapu SNS, Syed HA, et al. IgA Pemphigus. [Updated 2024 May 1]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK519063[↩][↩]
- IgA pemphigus. https://dermnetnz.org/topics/iga-pemphigus[↩][↩]
- Sawamura S, Kajihara I, Makino K, Makino T, Fukushima S, Jinnin M, Oyama B, Hashimoto T, Ihn H. Systemic lupus erythematosus associated with myasthenia gravis, pemphigus foliaceus and chronic thyroiditis after thymectomy. Australas J Dermatol. 2017 Aug;58(3):e120-e122. doi: 10.1111/ajd.12510[↩]
- Pollmann R, Schmidt T, Eming R, Hertl M. Pemphigus: a Comprehensive Review on Pathogenesis, Clinical Presentation and Novel Therapeutic Approaches. Clin Rev Allergy Immunol. 2018 Feb;54(1):1-25. doi: 10.1007/s12016-017-8662-z[↩]
- Santi CG, Sotto MN. Immunopathologic characterization of the tissue response in endemic pemphigus foliaceus (fogo selvagem). J Am Acad Dermatol. 2001 Mar;44(3):446-50. doi: 10.1067/mjd.2001.112344[↩]
- Jiao D, Bystryn JC. Sensitivity of indirect immunofluorescence, substrate specificity, and immunoblotting in the diagnosis of pemphigus. J Am Acad Dermatol. 1997 Aug;37(2 Pt 1):211-6. doi: 10.1016/s0190-9622(97)80127-2[↩][↩]
- Sar-Pomian M, Kurzeja M, Rudnicka L, Olszewska M. The value of trichoscopy in the differential diagnosis of scalp lesions in pemphigus vulgaris and pemphigus foliaceus. An Bras Dermatol. 2014 Nov-Dec;89(6):1007-12. doi: 10.1590/abd1806-4841.20143830[↩][↩]
- Pemphigus foliaceus. https://dermnetnz.org/topics/pemphigus-foliaceus[↩]
- Imbernón-Moya A, Burgos F, Vargas-Laguna E, Fernández-Cogolludo E, Aguilar-Martínez A, Gallego-Valdés MÁ. Pemphigus foliaceus associated with Hypericum perforatum. JAAD Case Rep. 2016 Aug 17;2(4):326-8. doi: 10.1016/j.jdcr.2016.06.008[↩][↩][↩]
- Gui H, Young PA, So JY, Pol-Rodriguez M, Rieger KE, Lewis MA, Winge MCG, Bae GH. New-onset pemphigus vegetans and pemphigus foliaceus after SARS-CoV-2 vaccination: A report of 2 cases. JAAD Case Rep. 2022 Sep;27:94-98. doi: 10.1016/j.jdcr.2022.07.002[↩]
- Gilkey T, Hammond A, Gurnani P, Forouzandeh M, Hsu S, Motaparthi K. Subtype transition from pemphigus vulgaris to pemphigus foliaceus. JAAD Case Rep. 2022 Nov 9;31:31-34. doi: 10.1016/j.jdcr.2022.10.036[↩]
- Nguyen TTD, Chau TNT, Doan Vo PT, Nguyen HT. Sporadic Pemphigus Foliaceus in a 3-Year-Old Vietnamese Girl: A Case Report and Literature Review. Case Rep Dermatol Med. 2024 Feb 16;2024:6748340. doi: 10.1155/2024/6748340[↩]
- Warren SJ, Arteaga LA, Rivitti EA, Aoki V, Hans-Filho G, Qaqish BF, Lin MS, Giudice GJ, Diaz LA. The role of subclass switching in the pathogenesis of endemic pemphigus foliaceus. J Invest Dermatol. 2003 Jan;120(1):104-8. doi: 10.1046/j.1523-1747.2003.12017.x[↩]
- Arteaga LA, Prisayanh PS, Warren SJ, Liu Z, Diaz LA, Lin MS; Cooperative Group on Fogo Selvagem Research. A subset of pemphigus foliaceus patients exhibits pathogenic autoantibodies against both desmoglein-1 and desmoglein-3. J Invest Dermatol. 2002 May;118(5):806-11. doi: 10.1046/j.1523-1747.2002.01743.x[↩]
- Brenner S, Goldberg I. Drug-induced pemphigus. Clin Dermatol. 2011;29:455–457. doi: 10.1016/j.clindermatol.2011.01.016[↩]
- Moraes ME, Fernandez-Vina M, Lazaro A, Diaz LA, Filho GH, Friedman H, et al. An epitope in the third hypervariable region of the DRB1 gene is involved in the susceptibility to endemic pemphigus foliaceus (fogo selvagem) in three different Brazilian populations. Tissue Antigens. 1997;49:35–40. doi: 10.1111/j.1399-0039.1997.tb02707.x[↩]
- Qian Y, Jeong JS, Maldonado M, Valenzuela JG, Gomes R, Teixeira C, et al. Cutting Edge: Brazilian Pemphigus Foliaceus Anti-Desmoglein 1 Autoantibodies Cross- React with Sand Fly Salivary LJM11 Antigen. J Immunol. 2012;189:1535–1539. doi: 10.4049/jimmunol.1200842[↩]
- Marsden RA, Vanhegan RI, Walshe M, Hill H, Mowat AG. Pemphigus foliaceus induced by penicillamine. Br Med J. 1976;2:1423–1424. doi: 10.1136/bmj.2.6049.1423[↩]
- Sokol E, Kramer D, Diercks GFH, Kuipers J, Jonkman MF, Pas HH, Giepmans BNG. Large-Scale Electron Microscopy Maps of Patient Skin and Mucosa Provide Insight into Pathogenesis of Blistering Diseases. J Invest Dermatol. 2015 Jul;135(7):1763-1770. doi: 10.1038/jid.2015.109[↩]
- Piovezan BZ, Petzl-Erler ML. Both qualitative and quantitative genetic variation of MHC class II molecules may influence susceptibility to autoimmune diseases: The case of endemic pemphigus foliaceus. Hum Immunol. 2013;74:1134–1140. doi: 10.1016/j.humimm.2013.06.008[↩]
- Hans-Filho G, Aoki V, Bittner NRH, Bittner GC. Fogo selvagem: endemic pemphigus foliaceus. An Bras Dermatol. 2018;93:638–650. doi: 10.1590/abd1806-4841.20188235[↩]
- Gomi H, Kawada A, Amagai M, Matsuo I. Pemphigus erythematosus: detection of anti-desmoglein-1 antibodies by ELISA. Dermatology. 1999;199(2):188-9. doi: 10.1159/000018239[↩]
- Diaz LA, Prisayanh PS, Dasher DA, Li N, Evangelista F, Aoki V, et al. The IgM anti-desmoglein 1 response distinguishes Brazilian pemphigus foliaceus (fogo selvagem) from other forms of pemphigus. J Invest Dermatol. 2008;128:667–675. doi: 10.1038/sj.jid.5701121[↩]
- Evangelista F, Roth AJ, Prisayanh P, Temple BR, Li N, Qian Y, et al. Pathogenic IgG4 autoantibodies from endemic pemphigus foliaceus recognize a desmoglein-1 conformational epitope. J Autoimmun. 2018;89:171–185. doi: 10.1016/j.jaut.2017.12.017[↩]
- Yoshida K, Ishii K, Shimizu A, Yokouchi M, Amagai M, Shiraishi K, et al. Non-pathogenic pemphigus foliaceus (PF) IgG acts synergistically with a directly pathogenic PF IgG to incrase blistering by p38MAPK-dependent desmoglein 1 clustering. J Dermatol Sci. 2017;85:197–207. doi: 10.1016/j.jdermsci.2016.12.010[↩]
- Grando SA. Pemphigus autoimmunity: hypotheses and realities. Autoimmunity. 2012;45:7–35. doi: 10.3109/08916934.2011.606444[↩]
- Shirakata Y, Amagai M, Hanakawa Y, Nishikawa T, Hashimoto K. Lack of mucosal involvement in pemphigus foliaceus may be due to low expression of desmoglein 1. J Invest Dermatol. 1998 Jan;110(1):76-8. doi: 10.1046/j.1523-1747.1998.00085.x[↩]
- Jordan TJM, Affolter VK, Outerbridge CA, Goodale EC, White SD. Clinicopathological findings and clinical outcomes in 49 cases of feline pemphigus foliaceus examined in Northern California, USA (1987-2017). Vet Dermatol. 2019 Jun;30(3):209-e65. doi: 10.1111/vde.12731[↩]
- Daniel BS, Hertl M, Werth VP, Eming R, Murrell DF. Severity score indexes for blistering diseases. Clin Dermatol. 2012;30:108–113. doi: 10.1016/j.clindermatol.2011.03.017[↩]
- Sebaratnam DF, Murrell DF. Objective scoring systems for disease activity in autoimmune bullous disease. Dermatol Clin. 2011;29:515-20, xi. doi: 10.1016/j.det.2011.03.015[↩]
- Fuchs K, Rummler S, Ries W, Helmschrott M, Selbach J, Ernst F, Morath C, Gauly A, Atiye S, Stauss-Grabo M, Giefer M. Performance, clinical effectiveness, and safety of immunoadsorption in a wide range of indications. Ther Apher Dial. 2022 Feb;26(1):229-241. doi: 10.1111/1744-9987.13663[↩][↩]
- Sami N, Qureshi A, Ahmed AR. Steroid sparing effect of intravenous immunoglobulin therapy in patients with pemphigus foliaceus. Eur J Dermatol. 2002 Mar-Apr;12(2):174-8.[↩]
- Hymes SR, Jordon RE. Pemphigus foliaceus. Use of antimalarial agents as adjuvant therapy. Arch Dermatol. 1992 Nov;128(11):1462-4. doi: 10.1001/archderm.128.11.1462[↩]
- Joly P, Maho-Vaillant M, Prost-Squarcioni C, et al. French study group on autoimmune bullous skin diseases. First-line rituximab combined with short-term prednisone versus prednisone alone for the treatment of pemphigus (Ritux 3): a prospective, multicentre, parallel-group, open-label randomised trial. Lancet. 2017 May 20;389(10083):2031-2040. doi: 10.1016/S0140-6736(17)30070-3[↩]