Contents
- 4 Types of Exercise
- What are the Health Benefits of Regular Exercise
- Benefits of Regular Exercise
- Exercise and Calories Burned
- Getting lots of exercise
- Exercise and Obesity
- How Much Activity Do People Need to Prevent Weight Gain?
- How Much Activity Do People Need to Lose Weight?
- Exercise and Hyperlipidemia
- Exercise and Metabolic syndrome
- Exercise and Type 2 diabetes
- Exercise and Type 1 diabetes
- Exercise and Hypertension
- Exercise and Stroke
- Exercise and Coronary heart disease
- Exercise and Heart failure
- Exercise and Intermittent claudication
- Exercise and Chronic obstructive pulmonary disease
- Exercise and Bronchial asthma
- Exercise and Cystic fibrosis
- Exercise and Depression
- Exercise and Anxiety
- Exercise and Stress
- Exercise and Schizophrenia
- Exercise and Dementia
- Exercise and Parkinson’s disease
- Exercise and Multiple sclerosis
- What is known about the relationship between exercise and cancer risk ?
- How might physical activity be linked to reduced risks of cancer ?
- Is physical activity beneficial for cancer survivors ?
- Exercise and Polycystic ovarian syndrome
- Exercise while Pregnant and After having your Baby
- Is it safe to exercise during pregnancy ?
- Are there certain conditions that make exercise during pregnancy unsafe ?
- What are the benefits of exercise during pregnancy ?
- How much exercise should you do during pregnancy ?
- What changes occur in the body during pregnancy that can affect your exercise routine ?
- What precautions should you take when exercising during pregnancy ?
- What are some safe exercises you can do during pregnancy ?
- What exercises you should avoid during pregnancy ?
- What are warning signs that you should stop exercising ?
- Why is it important to keep exercising after your baby is born ?
- How much exercise you should do after you have a baby ?
- What are muscle-strengthening workouts and how often should you do them ?
- When can you start exercising after pregnancy ?
- What are some guidelines you can follow when you begin exercising after pregnancy ?
- What are some ways to start exercising ?
- Where you can find out about exercise classes ?
- What can you do if you want to exercise but don’t want to join a gym ?
- How you can stay motivated once you start exercising ?
- How should you prepare for your workout ?
- How you should warm up before your workout ?
- How you should cool down after your workout ?
- Exercise for Older Adults and is Essential for Healthy Aging
- Exercise for Children
- How much Exercise or Physical Activity do you need ?
- Target Heart Rate and Estimated Maximum Heart Rate 309
- Maximum Heart Rate = 220 – Age
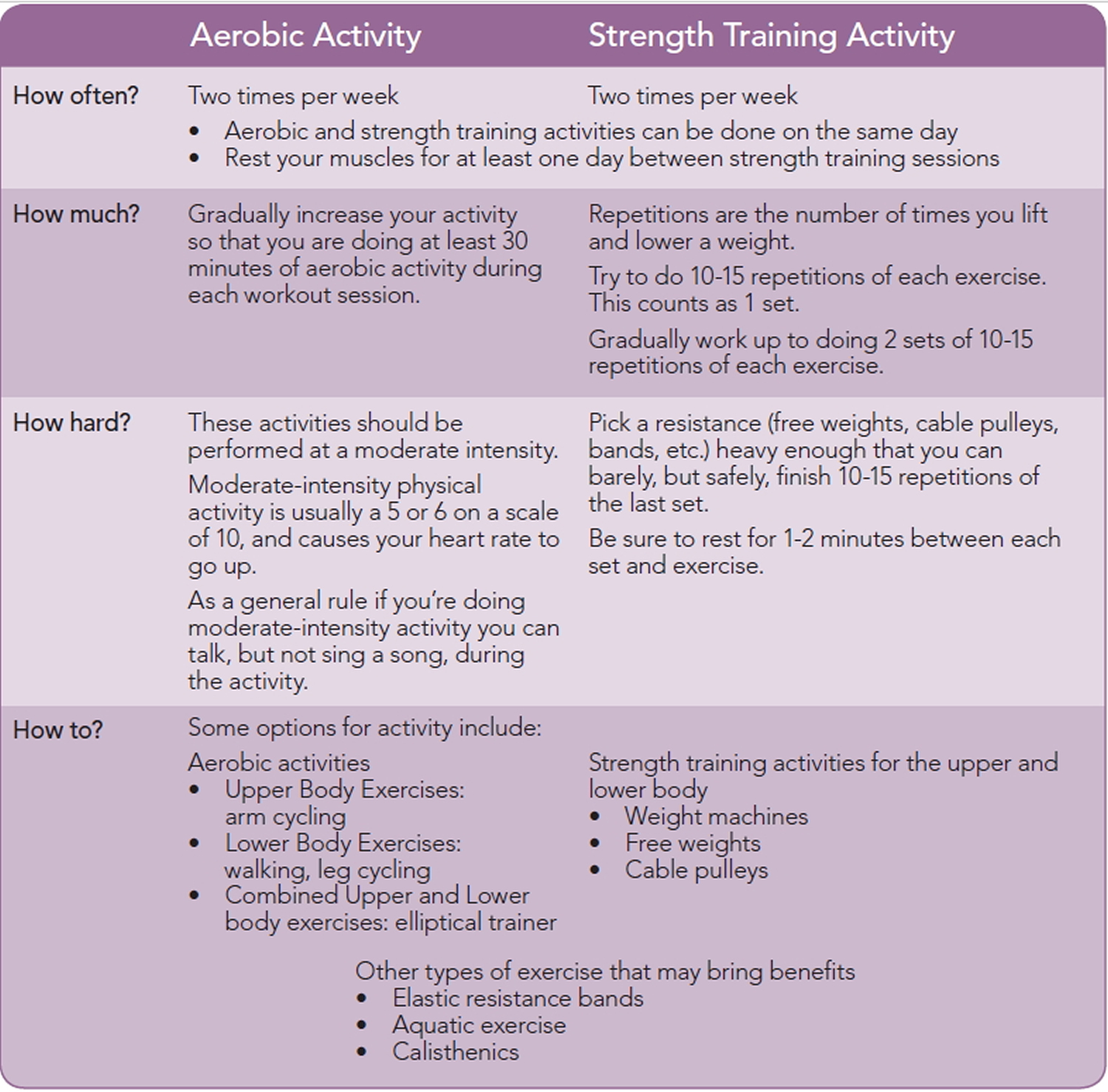
4 Types of Exercise
Exercise and physical activity fall into four basic categories—endurance, strength, balance, and flexibility. Most people tend to focus on one activity or type of exercise and think they’re doing enough. Each type is different, though. Doing them all will give you more benefits. Mixing it up also helps to reduce boredom and cut your risk of injury.
Though we’ve described each type separately, some activities fit into more than one category. For example, many endurance activities also build strength. Strength exercises also help improve balance.
1) Endurance
Endurance, or aerobic, activities increase your breathing and heart rate. These activities help keep you healthy, improve your fitness, and help you do the tasks you need to do every day. Endurance exercises improve the health of your heart, lungs, and circulatory system. They also delay or prevent many diseases that are common in older adults such as diabetes, heart disease, and osteoporosis. Building your endurance makes it easier to carry out many of your everyday activities.
- Brisk walking or jogging
- Yard work (mowing, raking, digging)
- Cycling
- Dancing
2) Strength
Strength exercises make your muscles stronger. Even small increases in strength can make a big difference in your ability to stay independent and carry out everyday activities, such as climbing stairs and carrying groceries. These exercises also are called “strength training” or “resistance training.”
- Lifting weights
- Using a resistance band
- Using your own body weight
- Carry groceries in from the car
- Pick up bags of mulch.
3) Balance
Balance exercises help prevent falls, a common problem in older adults. Many lower-body strength exercises also will improve your balance.
- Standing on one foot
- Heel-to-toe walk
- Tai Chi
4) Flexibility
Flexibility exercises stretch your muscles and can help your body stay limber. Being flexible gives you more freedom of movement for other exercises as well as for your everyday activities.
- Shoulder and upper arm stretch
- Calf stretch
- Yoga
- Bend down to tie your shoes
- Stretch to clean hard to reach areas of the house.
How Exercise Can Help You ?
Exercise and physical activity are good for just about everyone, including older adults. No matter your health and physical abilities, you can gain a lot by staying active. In fact, in most cases you have more to lose by not being active.
Here are just a few of the benefits. Exercise and physical activity:
- Can help maintain and improve your physical strength and fitness.
- Can help maintain and increases your flexibility
- Gives you more energy
- Helps control your weight
- Helps build and maintain strong bones
- Helps prevent or reduce the risk of major diseases, such as cardiovascular disease, diabetes mellitus, and certain types of cancer
- Can help improve your ability to do the everyday things you want to do.
- Can help improve your balance.
- Can help manage and improve diseases like diabetes, heart disease, and osteoporosis.
- Can help reduce feelings of depression and may improve mood and overall well-being. Relieves stress, improves sleep quality, and can help ease depression and anxiety
- May improve your ability to shift quickly between tasks, plan an activity, and ignore irrelevant information.
The key word in all these benefits is YOU — how fit and active you are now and how much effort you put into being active. To gain the most benefits, enjoy all 4 types of exercise, stay safe while you exercise, and be sure to eat a healthy diet, too!
Exercise as Appetite Suppressant
Most weight loss occurs because of decreased caloric intake. However, evidence shows the only way to maintain weight loss is to be engaged in regular physical activity.
Another benefit of regular physical activity of any sort is that it temporarily curbs your appetite. Of course, many people joke that after a workout they feel extremely hungry and promptly indulge in a snack. But because exercise raises resting energy expenditure, people continue to burn calories at a relatively high rate. So a moderate snack after exercising does not erase the benefits of exercise in helping people control their weight.
Biologists now believe that most cells in your body are designed to fall apart after relatively short life spans. The net result is that you are actively destroying large parts of your body all the time. On purpose. To make room for new growth. For example, the muscle cells in your thigh are replaced every four months; you have armies of special cells whose job is to dissolve your bones so other cells can build them up again. The trick is to grow more than you throw out. This is where exercise comes in. It turns out that your muscles control the chemistry of growth throughout your body.
What are the Health Benefits of Regular Exercise
Most importantly, physical activity reduces risks of cardiovascular disease, cancer and diabetes beyond that produced by weight reduction alone. Regular physical activity helps maintain a healthy body. Exercise helps increase endurance, promotes good balance, and keeps you flexible — all of which help get you through the day at your best.
Is being sedentary linked to health risks ?
Yes. Sedentary behavior, such as prolonged periods of television viewing, sitting, and lying down, is a risk factor for developing chronic conditions, including cancer, cardiovascular disease, and type 2 diabetes, and for premature death 1, 2. In some studies, the association of sedentary behavior with these outcomes is independent of physical activity—that is, the increased risks of disease associated with being sedentary are not lessened by physical activity. However, in one large meta-analysis 3 an increase in cancer mortality was seen only in those sedentary people with the least physical activity.
Although exercise has long been associated with good health, it was only 20 years ago that lack of sufficient physical activity/exercise was considered a major risk factor of equal importance as high cholesterol, high blood pressure, and smoking 4.
Physical activity 5 helps to :
- Maintain weight.
- Reduce high blood pressure.
- Reduce risk for type 2 diabetes, heart attack, stroke, and several forms of cancer.
- Reduce arthritis pain and associated disability.
- Reduce risk for osteoporosis and falls.
- Reduce symptoms of depression and anxiety.
As the evidence base for the health benefits continued to expand, updated recommendations were issued by the American College of Sports Medicine and AHA in 2007 and the first ever federal guidelines on physical activity in 2008 6, 7, 8.
Likewise, there are clear recommendations of the importance of exercise for secondary prevention of coronary and cardiovascular diseases 9. These reports document that sufficient amounts of physical activity or exercise are beneficial in the prevention and/or treatment of many chronic diseases. Despite the well accepted benefits, surveillance data from 2005 showed a majority of U.S. adults (53.3% of women and 50.3% of men) do not obtain sufficient physical activity for health-enhancement (defined as > 30 minutes at a moderate-intensity on most days of the week) 10.
Benefits of Regular Exercise
These studies 11, 12, 13, 14, 15, 16 revealed skeletal muscle as an endocrine organ releasing cytokines and other peptides. Since the initial reports 17, numerous research teams have demonstrated that physical exercise induces elevation of the plasma content of several cytokines, including tumor necrosis factor TNF-α, interleukins IL-1β, IL-6, IL-8, and IL-15, and leukemia inhibitory factor (LIF) 18. Indeed, in skeletal muscles, Interleukin 6 (IL-6) acts in autocrine or paracrine manner signaling through IL-6Ra receptors to increase glucose uptake and fat breakdown via phosphorylation of protein kinase B and AMP-activated protein kinase (AMPK), respectively, whereas acting in endocrine manner it provides energy supply via increase in glucose production in the liver and lipolysis (fat breakdown) in the adipose tissue 19. Interleukin 15 (IL-15) decreases lipid deposition in preadipocytes and the mass of white adipose tissue 20. Leukemia inhibitory factor (LIF) induces cell proliferation, which is considered to be essential for proper muscle hypertrophy and regeneration 21. Considering this, the role of interleukins and other myokines in the beneficial action of exercise in the treatment of metabolic, cardiovascular, lung, and musculoskeletal disorders has been widely reviewed and discussed 22.
Regular exercise also increases the amount of energy you burn while you are exercising. But it also boosts your resting energy expenditure — the rate at which you burn calories when the workout is over and you are resting. Resting energy expenditure remains elevated as long as you exercise at least three days a week on a regular basis.
Because resting energy expenditure accounts for 60% to 75% of the calories you burn each day, any increase in resting energy expenditure is extremely important to your weight-loss effort. The kinds of vigorous activity that can stimulate your metabolism include walking briskly for two miles or riding a bike uphill. Even small, incremental amounts of energy expenditure, like standing up instead of sitting down, can add up.
Exercise and Calories Burned
Regular physical activity is important for good health and it’s especially important if you’re trying to lose weight or to maintain a healthy weight. The burning of calories through physical activity, combined with reducing the number of calories you eat, creates a “calorie deficit” that results in weight loss. There are numerous ways you can exercise every day. Help determine what exercise is right for you using the following information on calories burned by type of workout.
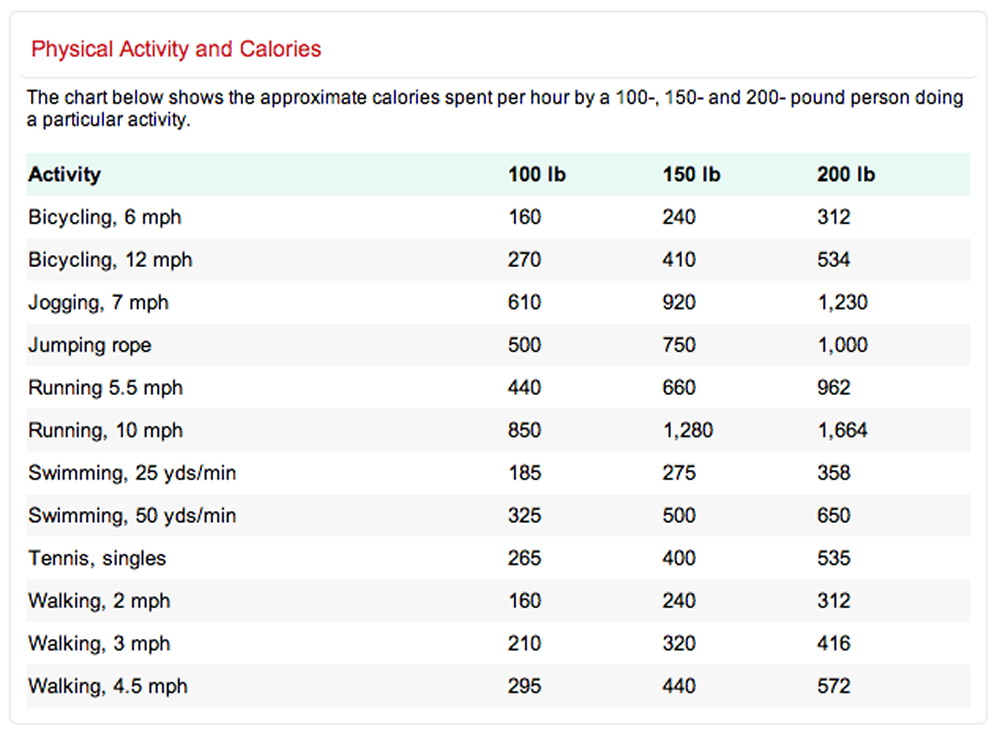 [Source 23]
[Source 23]The following table shows calories used in common physical activities at both moderate and vigorous levels 5.
Calories Used per Hour in Common Physical Activities | ||
|---|---|---|
| Moderate Physical Activity | Approximate Calories/30 Minutes for a 154 lb Person1 | Approximate Calories/Hr for a 154 lb Person1 |
| Hiking | 185 | 370 |
| Light gardening/yard work | 165 | 330 |
| Dancing | 165 | 330 |
| Golf (walking and carrying clubs) | 165 | 330 |
| Bicycling (<10 mph) | 145 | 290 |
| Walking (3.5 mph) | 140 | 280 |
| Weight lifting (general light workout) | 110 | 220 |
| Stretching | 90 | 180 |
| Vigorous Physical Activity | Approximate Calories/30 Minutes for a 154 lb Person1 | Approximate Calories/Hr for a 154 lb Person1 |
| Running/jogging (5 mph) | 295 | 590 |
| Bicycling (>10 mph) | 295 | 590 |
| Swimming (slow freestyle laps) | 255 | 510 |
| Aerobics | 240 | 480 |
| Walking (4.5 mph) | 230 | 460 |
| Heavy yard work (chopping wood) | 220 | 440 |
| Weight lifting (vigorous effort) | 220 | 440 |
| Basketball (vigorous) | 220 | 440 |
| Calories burned per hour will be higher for persons who weigh more than 154 lbs (70 kg) and lower for persons who weigh less. [Source: Adapted from Dietary Guidelines for Americans 2005, page 16, Table 4 24] | ||
Getting lots of exercise
The table below lists the calories burned by doing dozens of activities listed by category (such as gym activities, training and sports activities, home repair etc.) for 30 minutes. Activities and exercises include walking (casual, race, and everything in between), swimming, jogging, yoga, and even watching TV and sleeping. In each category, activities are listed from least to most calories burned.
| Gym Activities | 125-pound person | 155-pound person | 185-pound person |
| Weight Lifting: general | 90 | 112 | 133 |
| Aerobics: water | 120 | 149 | 178 |
| Stretching, Hatha Yoga | 120 | 149 | 178 |
| Calisthenics: moderate | 135 | 167 | 200 |
| Riders: general | 150 | 186 | 222 |
| Aerobics: low impact | 165 | 205 | 244 |
| Stair Step Machine: general | 180 | 223 | 266 |
| Teaching aerobics | 180 | 223 | 266 |
| Weight Lifting: vigorous | 180 | 223 | 266 |
| Aerobics, Step: low impact | 210 | 260 | 311 |
| Aerobics: high impact | 210 | 260 | 311 |
| Bicycling, Stationary: moderate | 210 | 260 | 311 |
| Rowing, Stationary: moderate | 210 | 260 | 311 |
| Calisthenics: vigorous | 240 | 298 | 355 |
| Circuit Training: general | 240 | 298 | 355 |
| Rowing, Stationary: vigorous | 255 | 316 | 377 |
| Elliptical Trainer: general | 270 | 335 | 400 |
| Ski Machine: general | 285 | 353 | 422 |
| Aerobics, Step: high impact | 300 | 372 | 444 |
| Bicycling, Stationary: vigorous | 315 | 391 | 466 |
| Training and Sport Activities | |||
| Billiards | 75 | 93 | 111 |
| Bowling | 90 | 112 | 133 |
| Dancing: slow, waltz, foxtrot | 90 | 112 | 133 |
| Frisbee | 90 | 112 | 133 |
| Volleyball: non-competitive, general play | 90 | 112 | 133 |
| Water Volleyball | 90 | 112 | 133 |
| Archery: non-hunting | 105 | 130 | 155 |
| Golf: using cart | 105 | 130 | 155 |
| Hang Gliding | 105 | 130 | 155 |
| Curling | 120 | 149 | 178 |
| Gymnastics: general | 120 | 149 | 178 |
| Horseback Riding: general | 120 | 149 | 178 |
| Tai Chi | 120 | 149 | 178 |
| Volleyball: competitive, gymnasium play | 120 | 149 | 178 |
| Walking: 3.5 mph (17 min/mi) | 120 | 149 | 178 |
| Badminton: general | 135 | 167 | 200 |
| Walking: 4 mph (15 min/mi) | 135 | 167 | 200 |
| Kayaking | 150 | 186 | 222 |
| Skateboarding | 150 | 186 | 222 |
| Snorkeling | 150 | 186 | 222 |
| Softball: general play | 150 | 186 | 222 |
| Walking: 4.5 mph (13 min/mi) | 150 | 186 | 222 |
| Whitewater: rafting, kayaking | 150 | 186 | 222 |
| Dancing: disco, ballroom, square | 165 | 205 | 244 |
| Golf: carrying clubs | 165 | 205 | 244 |
| Dancing: Fast, ballet, twist | 180 | 223 | 266 |
| Fencing | 180 | 223 | 266 |
| Hiking: cross-country | 180 | 223 | 266 |
| Skiing: downhill | 180 | 223 | 266 |
| Swimming: general | 180 | 223 | 266 |
| Walk/Jog: jog <10 min. | 180 | 223 | 266 |
| Water Skiing | 180 | 223 | 266 |
| Wrestling | 180 | 223 | 266 |
| Basketball: wheelchair | 195 | 242 | 289 |
| Race Walking | 195 | 242 | 289 |
| Ice Skating: general | 210 | 260 | 311 |
| Racquetball: casual, general | 210 | 260 | 311 |
| Rollerblade Skating | 210 | 260 | 311 |
| Scuba or skin diving | 210 | 260 | 311 |
| Sledding, luge, toboggan | 210 | 260 | 311 |
| Soccer: general | 210 | 260 | 311 |
| Tennis: general | 210 | 260 | 311 |
| Basketball: playing a game | 240 | 298 | 355 |
| Bicycling: 12-13.9 mph | 240 | 298 | 355 |
| Football: touch, flag, general | 240 | 298 | 355 |
| Hockey: field & ice | 240 | 298 | 355 |
| Rock Climbing: rappelling | 240 | 298 | 355 |
| Running: 5 mph (12 min/mile) | 240 | 298 | 355 |
| Running: pushing wheelchair, marathon wheeling | 240 | 298 | 355 |
| Skiing: cross-country | 240 | 298 | 355 |
| Snow Shoeing | 240 | 298 | 355 |
| Swimming: backstroke | 240 | 298 | 355 |
| Volleyball: beach | 240 | 298 | 355 |
| Bicycling: BMX or mountain | 255 | 316 | 377 |
| Boxing: sparring | 270 | 335 | 400 |
| Football: competitive | 270 | 335 | 400 |
| Orienteering | 270 | 335 | 400 |
| Running: 5.2 mph (11.5 min/mile) | 270 | 335 | 400 |
| Running: cross-country | 270 | 335 | 400 |
| Bicycling: 14-15.9 mph | 300 | 372 | 444 |
| Martial Arts: judo, karate, kickbox | 300 | 372 | 444 |
| Racquetball: competitive | 300 | 372 | 444 |
| Rope Jumping | 300 | 372 | 444 |
| Running: 6 mph (10 min/mile) | 300 | 372 | 444 |
| Swimming: breaststroke | 300 | 372 | 444 |
| Swimming: laps, vigorous | 300 | 372 | 444 |
| Swimming: treading, vigorous | 300 | 372 | 444 |
| Water Polo | 300 | 372 | 444 |
| Rock Climbing: ascending | 330 | 409 | 488 |
| Running: 6.7 mph (9 min/mile) | 330 | 409 | 488 |
| Swimming: butterfly | 330 | 409 | 488 |
| Swimming: crawl | 330 | 409 | 488 |
| Bicycling: 16-19 mph | 360 | 446 | 533 |
| Handball: general | 360 | 446 | 533 |
| Running: 7.5 mph (8 min/mile) | 375 | 465 | 555 |
| Running: 8.6 mph (7 min/mile) | 435 | 539 | 644 |
| Bicycling: > 20 mph | 495 | 614 | 733 |
| Running: 10 mph (6 min/mile) | 495 | 614 | 733 |
| Outdoor Activities | |||
| Planting seedlings, shrubs | 120 | 149 | 178 |
| Raking Lawn | 120 | 149 | 178 |
| Sacking grass or leaves | 120 | 149 | 178 |
| Gardening: general | 135 | 167 | 200 |
| Mowing Lawn: push, power | 135 | 167 | 200 |
| Operate Snow Blower: walking | 135 | 167 | 200 |
| Plant trees | 135 | 167 | 200 |
| Gardening: weeding | 139 | 172 | 205 |
| Carrying & stacking wood | 150 | 186 | 222 |
| Digging, spading dirt | 150 | 186 | 222 |
| Laying sod / crushed rock | 150 | 186 | 222 |
| Mowing Lawn: push, hand | 165 | 205 | 244 |
| Chopping & splitting wood | 180 | 223 | 266 |
| Shoveling Snow: by hand | 180 | 223 | 266 |
| Home & Daily Life Activities | |||
| Sleeping | 19 | 23 | 28 |
| Watching TV | 23 | 28 | 33 |
| Reading: sitting | 34 | 42 | 50 |
| Standing in line | 38 | 47 | 56 |
| Cooking | 75 | 93 | 111 |
| Child-care: bathing, feeding, etc. | 105 | 130 | 155 |
| Food Shopping: with cart | 105 | 130 | 155 |
| Moving: unpacking | 105 | 130 | 155 |
| Playing w/kids: moderate effort | 120 | 149 | 178 |
| Heavy Cleaning: wash car, windows | 135 | 167 | 200 |
| Child games: hop-scotch, jacks, etc. | 150 | 186 | 222 |
| Playing w/kids: vigorous effort | 150 | 186 | 222 |
| Moving: household furniture | 180 | 223 | 266 |
| Moving: carrying boxes | 210 | 260 | 311 |
| Home Repair | |||
| Auto Repair | 90 | 112 | 133 |
| Wiring and Plumbing | 90 | 112 | 133 |
| Carpentry: refinish furniture | 135 | 167 | 200 |
| Lay or remove carpet/tile | 135 | 167 | 200 |
| Paint, paper, remodel: inside | 135 | 167 | 200 |
| Cleaning rain gutters | 150 | 186 | 222 |
| Hanging storm windows | 150 | 186 | 222 |
| Paint house: outside | 150 | 186 | 222 |
| Carpentry: outside | 180 | 223 | 266 |
| Roofing | 180 | 223 | 266 |
| Occupational Activities | |||
| Computer Work | 41 | 51 | 61 |
| Light Office Work | 45 | 56 | 67 |
| Sitting in Meetings | 49 | 60 | 72 |
| Desk Work | 53 | 65 | 78 |
| Sitting in Class | 53 | 65 | 78 |
| Truck Driving: sitting | 60 | 74 | 89 |
| Bartending/Server | 75 | 93 | 111 |
| Heavy Equip. Operator | 75 | 93 | 111 |
| Police Officer | 75 | 93 | 111 |
| Theater Work | 90 | 112 | 133 |
| Welding | 90 | 112 | 133 |
| Carpentry Work | 105 | 130 | 155 |
| Coaching Sports | 120 | 149 | 178 |
| Masseur, standing | 120 | 149 | 178 |
| Construction, general | 165 | 205 | 244 |
| Coal Mining | 180 | 223 | 266 |
| Horse Grooming | 180 | 223 | 266 |
| Masonry | 210 | 260 | 311 |
| Forestry, general | 240 | 298 | 355 |
| Heavy Tools, not power | 240 | 298 | 355 |
| Steel Mill: general | 240 | 298 | 355 |
| Firefighting | 360 | 446 | 533 |
Exercise and Obesity
The importance of physical activity for weight loss assessed by body weight or BMI is controversial, but physical training leads to a reduction in fat mass and abdominal obesity, in addition to counteracting loss of muscle mass during dieting. Strong evidence exists that physical activity is important for preventing weight gain in general, as well as for maintaining body weight after weight loss.
Weight loss through physical training
A Cochrane Review from 2006 26 comprising 3476 overweight or obese individuals studied 41 randomized controlled trials and concluded that physical activity alone induced significant weight loss, while physical activity combined with a restricted diet and dietary counseling was more effective. High-intensity physical activity was more effective than moderate activity. The authors defined physical training as “any form of physical exercise that is repeated regularly for a certain period of time”. A prerequisite was that the physical training had to be quantifiable. The physical training intervention mainly consisted of walking, using an exercise bike, jogging, and weight training. In most of the studies, the intensity of the training was greater than 60% of the maximum oxygen uptake/heart rate. The participants exercised most frequently for 40–50 min per session, 3–5 times a week. All of the studies showed that physical exercise induced a slight reduction in body weight and BMI. The combination of exercise and diet resulted in an average greater weight loss and a greater decrease in BMI than diet alone. Without diet, high-intensity physical training (~60% of the maximum oxygen uptake/pulse) led to greater weight loss than low-intensity physical training.
The Cochrane Review showed that physical training for overweight and obese adults had positive effects on both body weight and risk factors for cardiovascular disease. Physical training combined with a restricted diet/dietary counseling reduces body weight slightly but significantly more than a restricted diet/dietary counseling only. Studies with physical training without dietary change showed that high-intensity physical training reduced body weight more than low-intensity physical training. These results are consistent with other meta-analyses 27.
Maintaining body weight through physical exercise
A 2001 meta-analysis 28 comprised six non-randomized studies containing information about the importance of physical activity for maintaining body weight after weight loss. The group of physically active subjects initially lost 21 kg, while the group of physically inactive subjects lost 22 kg. After 2.7 years, the weight loss in the physically active group was 15 and 7 kg for the physically inactive group.
A Danish follow-up study 29 included 118 overweight post-menopausal women who had completed a randomized weight loss intervention in which they were allocated to 12 weeks of diet alone, diet plus physical training or to the control group. The 12 weeks of training had no long-term effect, but a significant effect on body weight and fat mass, if the women continued to exercise on their own.
Observational studies generally indicate that physical activity has a positive effect on maintenance of weight loss after a diet 30. Individuals who increase their level of physical activity after a diet maintain their weight better in some studies 31, while other studies cannot demonstrate an effect from physical activity 32. Non-randomized weight loss studies with a prospective follow-up find that individuals with a high level of physical activity gain less weight than individuals who do not exercise 33. One study found no such correlation 34.
Studies in which participants were randomized to physical training or to a control group 35 (n = 672) assessed the effect of physical activity on maintaining body weight. The patients who exercised had a weight gain of 4.8 kg, while the control group gained 6.0 kg. A number of studies 36 assessed patients (n = 475) who were randomized to a weight reduction program with or without physical training. After 1–2 years, the exercise group had gained 4.8 kg on average, while the control group had gained 6.6 kg. Similar results were confirmed in a 1997 meta-analysis 37, which showed that among 493 moderately overweight individuals, there was an average weight loss of 11 kg after 15 weeks of a restricted diet/counseling or restricted diet/counseling plus training. After 1 year, the restricted diet/counseling group had maintained a weight loss of 6.6 kg, while the restricted diet/dietary counseling plus exercise group had maintained its weight loss of 8.6 kg.
A literature review of 26 articles assessed the independent effects of normal weight vs obesity: fit vs unfit and physically active vs physically inactive. The risk of all-cause mortality and cardiovascular death was lower in individuals with high BMI who were physically fit compared to individuals with normal BMI and a lower level of physical fitness. The literature review, however, could not confirm results from other studies that showed that a high level of physical activity gave the same protection as being physically fit. Individuals with a high BMI and a high level of physical activity had a greater risk of developing type 2 diabetes and cardiovascular disease than those with a normal BMI and low level of physical activity.
There are many possible explanations as to why physical fitness and not a high level of physical activity protect against the serious health consequences of overweight and obesity. Information on physical activity in most studies is based on self-reported information, which is subject to considerable inaccuracy, while fitness is an objective measure. Another possible explanation is that primarily physical activity of high intensity leads to improved fitness and thereby protection against diseases associated with obesity 38.
Obesity is often associated with hypertension, hypercholesterolemia, hypertriglyceridemia, and insulin resistance. The effect of physical training on these risk markers is described separately on pages 14, 16 and 26. Obesity is also frequently associated with erectile dysfunction, which physical training can contribute to prevent 39.
How Much Activity Do People Need to Prevent Weight Gain?
Weight gain during adulthood can increase the risk of heart disease, diabetes, and other chronic conditions. Since it’s hard for people to lose weight and keep it off, it’s better to prevent weight gain in the first place. Encouragingly, there’s strong evidence that staying active can help people slow down or stave off “middle-age spread” 40. The more active people are, the more likely they are to keep their weight steady 41; the more sedentary, the more likely they are to gain weight over time 42. But it’s still a matter of debate exactly how much activity people need to avoid gaining weight. The latest evidence suggests that the recommended two and a half hours a week may not be enough.
The Women’s Health Study, for example, followed 34,000 middle-age women for 13 years to see how much physical activity they needed to stay within 5 pounds of their weight at the start of the study. Researchers found that women in the normal weight range at the start needed the equivalent of an hour a day of moderate-to-vigorous physical activity to maintain a steady weight 43.
Vigorous activities seem to be more effective for weight control than slow walking 44, 45. The Nurses’ Health Study II 46, for example, followed more than 18,000 women for 16 years to study the relationship between changes in physical activity and weight. Although women gained, on average, about 20 pounds over the course of the study, those who increased their physical activity by 30 minutes per day gained less weight than women whose activity levels stayed steady. And the type of activity made a difference: Bicycling and brisk walking helped women avoid weight gain, but slow walking did not.
How Much Activity Do People Need to Lose Weight?
Exercise can help promote weight loss, but it seems to work best when combined with a lower calorie eating plan 47. If people don’t curb their calories, however, they likely need to exercise for long periods of time-or at a high intensity-to lose weight 48, 49.
In one study 48, for example, researchers randomly assigned 175 overweight, inactive adults to either a control group that did not receive any exercise instruction or to one of three exercise regimens-low intensity (equivalent to walking 12 miles/week), medium intensity (equivalent to jogging 12 miles/week), or high intensity (equivalent to jogging 20 miles per week). All study volunteers were asked to stick to their usual diets. After six months, those assigned to the high-intensity regimen lost abdominal fat, whereas those assigned to the low- and medium-intensity exercise regimens had no change in abdominal fat 48.
More recently, researchers conducted a similar trial with 320 post-menopausal women 50, randomly assigning them to either 45 minutes of moderate-to-vigorous aerobic activity, five days a week, or to a control group. Most of the women were overweight or obese at the start of the study. After one year, the exercisers had significant decreases in body weight, body fat, and abdominal fat, compared to the non-exercisers 50.
The Bottom Line
- For Weight Control, Aim for an Hour of Activity a Day
Being moderately active for at least 30 minutes a day on most days of the week can help lower the risk of chronic disease. But to stay at a healthy weight, or to lose weight, most people will need more physical activity-at least an hour a day-to counteract the effects of increasingly sedentary lifestyles, as well as the strong societal influences that encourage overeating.
Keep in mind that staying active is not purely an individual choice: The so-called “built environment”-buildings, neighborhoods, transportation systems, and other human-made elements of the landscape-influences how active people are 51. People are more prone to be active, for example, if they live near parks or playgrounds, in neighborhoods with sidewalks or bike paths, or close enough to work, school, or shopping to safely travel by bike or on foot. People are less likely to be active if they live in sprawling suburbs designed for driving or in neighborhoods without recreation opportunities.
Local and state governments wield several policy tools for shaping people’s physical surroundings, such as planning, zoning, and other regulations, as well as setting budget priorities for transportation and infrastructure. Strategies to create safe, active environments include curbing traffic to make walking and cycling safer, building schools and shops within walking distance of neighborhoods, and improving public transportation, to name a few. Such changes are essential to make physical activity an integral and natural part of people’s everyday lives-and ultimately, to turn around the obesity epidemic.
How much exercise do you need for general good health?
For general good health, the 2008 Physical Activity Guidelines for Americans 52 recommends that adults get a minimum of 2-1/2 hours per week of moderate-intensity aerobic activity. Yet many people may need more than 2-1/2 hours of moderate intensity activity a week to stay at a stable weight 52.
The Women’s Health Study 53, for example, followed 34,000 middle-aged women for 13 years to see just how much physical activity they needed to stay within 5 pounds of their weight at the start of the study. Researchers found that women who were in the normal weight range at the start of the study needed the equivalent of an hour a day of physical activity to stay at a steady weight 53.
If you are exercising mainly to lose weight, 60 minutes or so a day may be effective in conjunction with a healthy diet 54.
If you currently don’t exercise and aren’t very active during the day, any increase in exercise or physical activity is good for you.
Aerobic physical activity—any activity that causes a noticeable increase in your heart rate—is especially beneficial for disease prevention.
Some studies show that walking briskly for even one to two hours a week (15 to 20 minutes a day) starts to decrease the chances of having a heart attack or stroke, developing diabetes, or dying prematurely.
You can combine moderate and vigorous exercise over the course of the week, and it’s fine to break up your activity into smaller bursts as long as you sustain the activity for at least 10 minutes.
Exercise Intensity:
Moderate-intensity aerobic activity is any activity that causes a slight but noticeable increase in breathing and heart rate. One way to gauge moderate activity is with the “talk test”—exercising hard enough to break a sweat but not so hard you can’t comfortably carry on a conversation.
Vigorous-intensity aerobic activity causes more rapid breathing and a greater increase in heart rate, but you should still be able to carry on a conversation—with shorter sentences.
Here is a summary of the 2008 Physical Activity Guidelines for Americans 52
Children and adolescents should get at least 1 hour or more a day of physical activity in age-appropriate activities, spending most of that engaged in moderate- or vigorous–intensity aerobic activities. They should partake in vigorous-intensity aerobic activity on at least three days of the week, and include muscle-strengthening and bone strengthening activities on at least three days of the week.
Healthy adults should get a minimum of 2-1/2 hours per week of moderate-intensity aerobic activity, or a minimum of 1-1/4 hours per week of vigorous-intensity aerobic activity, or a combination of the two. That could mean a brisk walk for 30 minutes a day, five days a week; a high-intensity spinning class one day for 45 minutes, plus a half hour jog another day; or some other combination of moderate and vigorous activity. Doubling the amount of activity (5 hours moderate- or 2-1/2 hours vigorous-intensity aerobic activity) provides even more health benefits. Adults should also aim to do muscle-strengthening activities at least two days a week.
Healthy older Adults should follow the guidelines for healthy adults. Older adults who cannot meet the guidelines for healthy adults because of chronic conditions should be as physically active as their abilities and conditions allow. People who have chronic conditions such as arthritis and type 2 diabetes should talk to a healthcare provider about the amount and type of activity that is best. Physical activity can help people manage chronic conditions, as long as the activities that individuals choose match their fitness level and abilities. Even just an hour a week of activity has health benefits. Older adults who are at risk of falling should include activities that promote balance.
Strength training for all ages
Studies have shown strength training to increase lean body mass, decrease fat mass, and increase resting metabolic rate (a measurement of the amount of calories burned per day) in adults 55. While strength training on its own typically does not lead to weight loss 52, its beneficial effects on body composition may make it easier to manage one’s weight and ultimately reduce the risk of disease, by slowing the gain of fat—especially abdominal fat 56.
- Muscle is metabolically active tissue; it utilizes calories to work, repair, and refuel itself. Fat, on the other hand, doesn’t use as much energy. We slowly lose muscle as part of the natural aging process, which means that the amount of calories we need each day starts to decrease, and it becomes easier to gain weight.
- Strength training regularly helps preserve lean muscle tissue and can even rebuild some that has been lost already.
- Weight training has also been shown to help fight osteoporosis. For example, a study in postmenopausal women examined whether regular strength training and high-impact aerobics sessions would help prevent osteoporosis. Researchers found that the women who participated in at least two sessions a week for three years were able to preserve bone mineral density at the spine and hip; over the same time period, a sedentary control group showed bone mineral density losses of 2 to 8 percent 57.
- In older populations, resistance training can help maintain the ability to perform functional tasks such as walking, rising from a chair, climbing stairs, and even carrying one’s own groceries. An emerging area of research suggests that muscular strength and fitness may also be important to reducing the risk of chronic disease and mortality, but more research is needed 58, 59.
- A systematic review of 8 studies 60 examining the effects of weight-bearing and resistance-based exercises on the bone mineral density in older men found resistance training to be an effective strategy for preventing osteoporosis in this population. Resistance training was found to have more positive effects on bone mineral density than walking, which has a lower impact 60.
The Physical Activity Guidelines for Americans recommends that muscle strengthening activities be done at least two days a week 52. Different types of strength training activities are best for different age groups.
- When talking about the benefits of exercise, keeping the heart and blood vessels healthy usually gets most of the attention. For many individuals, though, stretching and strength training exercises may be just as important.
- Strength training, also known as resistance training, weight training, or muscle-strengthening activity, is one of the most beneficial components of a fitness program.
Children and Adolescents: Choose unstructured activities rather than weight lifting exercises 52.
Examples:
- Playing on playground equipment
- Climbing trees
- Playing tug-of-war
Active Adults: Weight training is a familiar example, but there are other options 52:
- Calisthenics that use body weight for resistance (such as push-ups, pull-ups, and sit-ups)
- Carrying heavy loads
- Heavy gardening (such as digging or hoeing)
Older Adults: The guidelines for older adults are similar to those for adults; older adults who have chronic conditions should consult with a health care provider to set their activity goals. Muscle strengthening activities in this age group include the following 52:
- Digging, lifting, and carrying as part of gardening
- Carrying groceries
- Some yoga and tai chi exercises
- Strength exercises done as part of a rehab program or physical therapy
Flexibility training
Flexibility training or stretching exercise is another important part of overall fitness. It may help older adults preserve the range of motion they need to perform daily tasks and other physical activities 61.
- The American Heart Association 55 recommends that healthy adults engage in flexibility training two to three days per week, stretching major muscle and tendon groups.
- For older adults, the American Heart Association and American College of Sports Medicine recommend two days a week of flexibility training, in sessions at least 10 minutes long 61. Older adults who are at risk of falling should also do exercises to improve their balance.
Exercise and Hyperlipidemia
Hyperlipidemia is a group of disorders of lipoprotein metabolism entailing elevated blood levels of certain forms of cholesterol and triglyceride. Primary hyperlipidemia caused by environmental and genetic factors are by far the most frequent, accounting for 98% of all cases. Isolated hypercholesterolemia and combined dyslipidemia are the most frequent types of dyslipidemia, and are due to excessive intake of fat in most people. These types of dyslipidemia entail an elevated risk of atherosclerosis. There is consensus that physical activity protects against the development of cardiovascular diseases 62, 63 and it has been suggested that one of many mechanisms could be a positive effect of exercise on the lipid profile of the blood 64. Epidemiological studies indicate that physical activity prevents hyperlipidemia 65.
Today, evidence shows that a large volume of physical training, independent of weight loss, has a beneficial effect on the lipid profile of the blood. A number of review articles summarize this knowledge 66.
A 2007 meta-analysis studied the effect of training on high-density lipoprotein (HDL) cholesterol. The analysis included 25 randomized controlled trials. The training comprised walking, cycling or swimming 67. Training had a significant but moderate effect on HDL cholesterol. The minimum amount of physical activity needed to cause an effect was 120 min of physical activity weekly or an energy expenditure equivalent to 3780 kJ. The duration of the physical activity was more important than its intensity. Each time the duration of the physical activity was increased by 10 min, the HDL cholesterol level increased on average by 1.4 mg/dL (0.036 mmol/L).
The average effect of physical activity on HDL is clinically relevant, albeit somewhat smaller than the effect achieved when using drugs that lower lipid levels 68. It is estimated that each time HDL increases 0.025 mmol/L, the cardiovascular risk goes down by 2% for men and by at least 3% for women 69. Training induced a mean increase of 0.036 mmol/L in the level of HDL. For the subgroup of individuals with a BMI of less than 28 and a total cholesterol level over 5.7 mmol/L, it was found that exercise induced an increase of 0.054 mmol/L in the level of HDL 70. For the latter group, physical training was thus able to reduce the cardiovascular risk by about 4% for men and by 6% for women.
A review article from 2014 66 includes 13 published investigations and two review articles and conclude that both aerobic, resistance exercise and the combination of aerobic and resistance training have impact on cholesterol levels and blood lipids.
A randomized clinical controlled trial evaluated the effect of training volume and intensity in a study comprising 111 physically inactive overweight men with mild to moderate hyperlipidemia 70. The subjects were randomized to a control group or 8 months of high-volume/high-intensity physical training [32 km/week at 65–80% maximum oxygen uptake (VO2max)]; low volume/high intensity (19 km/week at 65–80% of VO2max) or low volume/low intensity (19 km/week at 40–55% of VO2max). This study distinguishes itself by evaluating an extensive lipid profile in which the size of the lipid–protein particles is also included. Subjects were asked to maintain their weight and individuals with excessive weight loss were excluded. Despite this, there was a small but significant amount of weight loss in the training groups. All of the training groups achieved a positive effect on their lipid profile compared to the control group, but there was no significant difference in the effect of training in the two groups with a low volume of exercise, although the high-intensity group achieved a greater improvement in fitness. There is a significantly better effect from a high volume of physical training on virtually all lipid parameters, although the two groups with high-intensity training achieved the same improvement in fitness level. There was no effect on the total cholesterol level. High-volume/high-intensity training reduced the level of low-density lipoprotein (LDL), intermediate-density lipoprotein (IDL), and small LDL particles and increased the size of the LDL particles and the level of HDL. All of the groups had a positive effect on the level of triglyceride, VLD triglyceride, and the size of VLDL. Thus, the volume of training had clear effects, but the intensity of the training had less impact.
A 2010 meta-analysis compared 13 randomized controlled trials that examined the effect of resistance training on parameters related to metabolic syndrome. Resistance training showed a significant effect on obesity, HbA1c, and systolic blood pressure, but no effect on total cholesterol, HDL cholesterol, or LDL cholesterol 71.
A 2012 systematic review 72 assessed the effect of supervised exercise interventions on lipid profiles and blood pressure control in patients with type 2 diabetes. Forty-two RCTs (2808 subjects) met inclusion criteria and were included in the meta-analysis. It was concluded that supervised exercise is effective in improving blood pressure control, lowering LDL-C, and elevating HDL-C levels in people with diabetes.
Exercise and Metabolic syndrome
Metabolic syndrome is also known as insulin resistance syndrome, as one of the traits of the disorder is reduced insulin activity. There are several definitions for metabolic syndrome but it encompasses abdominal obesity, insulin resistance, hypertension, and hyperlipidemia.
The International Diabetes Federation 73 defines metabolic syndrome as follows:
Abdominal obesity, i.e., waist circumference ≥94 cm for men and ≥80 cm for women, plus at least two of the following four risk factors:
| Plasma concentration of triglycerides | ≥1.7 mmol/L |
| Plasma concentration of HDL cholesterol | <1.0 mmol/L for men and <1.2 mmol/L for women |
| Blood pressure | Systolic blood pressure ≥130 mmHg or diastolic blood pressure ≥85 mmHg or receiving antihypersensitive therapy |
| Plasma concentration of glucose (fasting) | ≥5.6 mmol/L or type 2 diabetes |
Metabolic syndrome rarely occurs in people with normal weight but it can occur and there is a higher incidence among members of the Pakistani and Turkish ethnic minorities than members of the general population with the same BMI. Metabolic syndrome is a precursor of type 2 diabetes and large-scale epidemiological studies show that physical activity can prevent the onset of metabolic syndrome 74.
Physical exercise and insulin resistance/prevention of type 2 diabetes
A 2008 Cochrane Review 75 assessed the effect of a combination of diet and physical exercise as prophylaxis against type 2 diabetes. Physical exercise varied from a recommended increase in daily physical activity to supervised physical training of varying intensity and up to several times a week. Most programs included walking, running, or cycling at different intensities. The diets were low calorie with reduced fat and high fiber.
The participants in the analysis had a pathological glucose tolerance and/or metabolic syndrome. The analysis included eight trials with 2241 participants in one group, who were prescribed physical activity and placed on a diet as described above, and 2509 control persons. The studies ran over a period of 1 and 6 years. Exercise and diet significantly lowered the risk of type 2 diabetes. A significant impact on body weight, BMI, waist-to-hip ratio, and waist circumference was also identified, as was a moderate impact on blood lipids. The intervention had a marked effect on both systolic and diastolic blood pressure 75.
The isolated effect of exercise alone as prevention against diabetes in patients with pathological glucose tolerance is sparsely documented but there is solid evidence pointing to the effect of combined physical exercise and diet. A Chinese study divided 577 people with pathological glucose tolerance into four groups: diet, exercise, diet + exercise and control, and monitored them over 6 years 76. The risk of diabetes fell by 31% in the diet group, by 46% in the exercise group, and by 42% in the diet + exercise group.
In a Swedish study, 6956 men aged 48 were given a health check-up. Those with pathological glucose tolerance were divided into two groups: (a) exercise + diet (n = 288) and (b) no intervention (n = 135) 77 and were monitored over 12 years. The mortality rate was the same in the intervention group as among the healthy control group (6.5% vs 6.2%) and lower than in the group with pathological glucose tolerance, which did not exercise (6.5% vs 14%). Thus, among all the participants with pathological glucose tolerance there was a predictive effect of intervention but not a predictive effect of BMI, blood pressure, smoking, cholesterol, or glucose level.
Two randomized controlled trials included people with pathological glucose tolerance and found that changes in lifestyle protected against development of type 2 diabetes. A Finnish study randomized 522 overweight middle-aged men and women with pathological glucose tolerance to physical exercise and diet or control 78 and monitored them over 3.2 years. The lifestyle intervention consisted of individual counseling on reduction of calorie intake, reduction of fat intake, and an increase in fiber-rich foods and daily physical activity. The risk of type 2 diabetes fell by 58% in the intervention group. The greatest effect was recorded with the patients who underwent the most extensive lifestyle changes 79.
An American study randomized 3234 people with pathological glucose tolerance to either treatment with metformin or a lifestyle program involving moderate physical activity in the form of at least 150 min of brisk walking a week and a reduced-calorie diet or no intervention. The subjects were monitored over 2.8 years 80. The lifestyle intervention group had a 58% lower risk of contracting type 2 diabetes. Thus, the reduction matched the findings in the Finnish study 78, while the metformin treatment only reduced the risk of diabetes by 31%. As can be seen, it is not formally possible to assess the isolated effect of exercise with respect to diet in three of the studies mentioned 80, but the intervention group experienced only a moderate weight loss.
In the Finnish study, weight loss after 2 years was 3.5 kg in the intervention group vs 0.8 kg in the control group 78. The intervention group thus experienced a drop in BMI from around 31 to around 30 in the Finnish study 78 and from 34 to 33 in the American study 80.
Physical exercise and abdominal obesity
Visceral fat constitutes an independent risk factor for developing heart disease. A cross-sectional study showed that overweight men with a high level of fitness have a significantly lower visceral fat than overweight men with a poor level of fitness 81.
A group of young, healthy, normal weight men who normally walked 10 000 paces every day reduced their paces to 1500 per day over a period of 2 weeks. They experienced a significant rise in volume of visceral fat (7%) despite a total average weight loss of 1.2 kg 82.
Irrespective of other fat deposits, abdominal obesity is a major risk factor for hyperlipidemia 83, lower glucose tolerance 84, insulin resistance 85, systemic inflammation 86, hypertension 87, type 2 diabetes 88, and all-cause mortality 89. There is a link between regular physical activity, with or without weight loss, and reduction in visceral fat volume 90.
Increasing physical activity to 60 min/day over 3 months has been found to reduce visceral fat volume by about 30%. It should be emphasized, however, that changes in visceral fat volume as a response to physical exercise vary considerably and that it is not possible to identify a clear correlation between amount of physical exercise and reduction in visceral fat.
In relation to reduction of visceral fat tissue deposits, however, no specific method exists (surgery, diet, physical activity, etc.) for achieving this. Intervention-induced reduction of visceral fat tissue deposits relates to reduction of the total volume of fat tissue and the initial ratio of volume of visceral fat tissue to volume of total fat, regardless of how the reduction in fat tissue is achieved.
Studies have shown that an increase in daily physical activity leads to a significant reduction in quantity of visceral fat and/or abdominal circumference, despite no or minimal alteration in total body weight. Thus, studies on people with type 2 diabetes show that 2–3 months of regular moderate-intensity aerobic training leads to a significant reduction in quantity of visceral fat. There is a corresponding finding for healthy, normal weight pre-menopausal women, healthy, middle-aged men and HIV-positive men with lipodystrophy.
Middle-aged, normal weight or overweight men, and overweight women can expect to see a reduction in visceral fat volume (−10 to −19%) after 3 months of regular physical activity. These results also apply to older, overweight individuals (60–80 years). As a result of exercise, either strength or stamina training for 80 min a week, test subjects did not accumulate visceral fat after dieting and losing weight, while the control group that did not exercise increased their volume of visceral fact by 38%.
Exercise and Type 2 diabetes
The positive gains from physical exercise for patients with type 2 diabetes are very well documented and there is an international consensus that physical exercise is one of the three cornerstones in the treatment of diabetes, along with diet and medication 91.
Several reviews 92 report that increased physical exercise produce a significant improvement in glucose control in people with type 2 diabetes, yielding an average improvement in hemoglobin A1c (HbA1c) of between −0.4% and −0.6%.
A 2006 Cochrane Review, which includes 14 randomized controlled trials with a total of 377 patients with type 2 diabetes, compares the independent effect of training with no training 93. The training interventions were 8–10 months in length and consisted of progressive aerobic training, strength training or a combination of the two, with typically three training sessions per week. Compared to the control group, the training interventions showed a significant improvement in glycemic control in the form of a reduction in HbA1c (glycated hemoglobin) of 0.6%. By comparison, intensive glycemic control using metformin showed a reduction in HbAc1 of 0.6%, and a risk reduction of 32% for diabetes-related complications and of 42% for diabetes-related mortality 94.
Despite the clear effect of exercise training on metabolic control, there was no significant effect on body weight. The reason for this is presumably that the exercise group reduced fat mass but increased muscle mass. One of the studies in the meta-analysis reported an increase in fat-free mass of 6.3 kg, measured by dual energy X-ray absorptiometry (DXA) scanning, and a reduction in visceral fat volume, measured in by magnetic resonance imaging (MRI) scanning, of −45.5 cm2. No adverse effects of physical exercise were reported.
Physical exercise significantly reduced insulin response as an expression of increased insulin sensitivity and triglyceride levels. This Cochrane Review found no significant difference with regard to quality of life, plasma cholesterol, or blood pressure 93. The findings from the Cochrane Review 93 agree with the conclusions from a 2001 meta-analysis, which also evaluated the impact of a minimum 8-week training program on glycemic control 95. Training was found to have no effect on body weight 95. There are several possible explanations for this: the training period was relatively short, the patients over-compensated for their loss of energy by eating more, or patients lost fat but their volume of fat-free mass increased. There is reason to assume that the final explanation is the most significant one. It is well-known that physically inactive people who start to exercise increase their fat-free mass 96. Only one of the studies included in the meta-analysis assessed abdominal obesity using MRI scanning 97. The aerobic training program (55 min three times a week over 10 weeks) resulted in a reduction of abdominal subcutaneous fat, measured using MRI scanning and visceral fat. The same study did not identify any effect from exercise on body weight.
A 2007 meta-analysis assessed the effect of self-management interventions with a view to increasing physical activity levels in patients with type 2 diabetes. The analysis involved 103 trials with 10 455 subjects. Self-management training was found to have a significant effect of 0.45% on HbA1c.
Interventions that included several different lifestyle recommendations such as diet and physical activity had less effect than interventions that included physical exercise only. Basic levels of HbA1c and BMI were not related to metabolic effect. The overall conclusion is that self-management interventions that include physical activity increase metabolic control 98.
A 2009 systematic review included nine studies with 372 patients with type 2 diabetes. Progressive resistance training vs no training induced a statistically significant reduction in HbA1c of 0.3%. There was no difference between resistance training and aerobic training as far as the effect on changes in HbA1c was concerned. Progressive resistance training resulted in large improvements in strength compared to aerobic training or no training. No significant effect on body composition was found 99.
A meta-analysis from 2013 found that exercise lowers postprandial glucose but not fasting glucose in type 2 diabetes 100. A 2007 meta-analysis evaluated the effect of aerobic physical training for a minimum of 8 weeks on lipids and lipoproteins in patients with type 2 diabetes. The analysis included seven trials with 220 men and women, of which 112 were in a training group and 108 in a control group. A statistically significant reduction of approximately 5% in LDL cholesterol was found but there was no significant effect with regard to triglycerides, HDL cholesterol, or total cholesterol 101.
A 2011 meta-analysis concluded that structured exercise training that consists of aerobic exercise, resistance training, or both combined is associated with HbA1c reduction in patients with type 2 diabetes. Structured exercise training of more than 150 min/week is associated with greater HbA1c declines than that of 150 min or less per week. Physical activity advice is associated with lower HbA1c, but only when combined with dietary advice 92.
A systematic review and meta-analysis from 2014 compared resistance exercise and aerobic exercise and concluded that there was no evidence that resistance exercise differs from aerobic exercise in impact on glucose control, cardiovascular risk markers or safety. Using one or the other type of exercise for type 2 diabetes may be less important than doing some form of physical activity 102.
Measures of fasting glucose and HbA1c do not accurately represent glycemic control because they do not reflect what occurs after meals and throughout the day in the free-living condition 103. An accumulating body of evidence now suggests that postprandial glucose fluctuations are more tightly correlated with microvascular and macrovascular morbidities and cardiovascular mortality than HbA1c or fasting glucose, stagnant measure of glycemia. It is therefore important that unlike medications, which generally have a poor effect at improving postprandial glucose, exercise has been proven effective in reducing postprandial glycemic excursions in as little as a few days 103.
Effect on fitness and muscle strength
Poor fitness is an independent prognostic marker for death in patients with type 2 diabetes 104. A meta-analysis 105 assesses the effect of a minimum of 8 weeks of physical training on maximum oxygen uptake (VO2max). The analysis involved 266 patients with type 2 diabetes. Average training quantity was 3.4 sessions/week, each lasting 49 min; intensity was 50–75% of maximum pulse; the length of the interventions was in average 20 weeks. Altogether, there was an 11.8% increase in VO2max in the training group vs a drop of 1% in the control group.
Older patients with type 2 diabetes (n = 31) were randomized to a 24-month resistance training program. Average increase in muscle strength was 31% in the exercise group, while no effect on muscle strength was identified in the control group 106. Patients with type 2 diabetes can thus adapt to training with regard to both fitness and muscle strength.
There was a more striking impact on fitness when the physical exercise was supervised, was done in groups and took place over a long period. There was no correlation between level of fitness improvement and HbA1c, age, BMI, or sex 107.
Effect on mortality
The Look AHEAD study included 16 centers in the United States, and randomly assigned 5145 overweight or obese patients with type 2 diabetes to participate in an intensive lifestyle intervention that promoted weight loss through decreased caloric intake and increased physical activity (intervention group) or to receive diabetes support and education (control group). The trial was stopped early on the basis of a futility analysis when the median follow-up was 9.6 years as the intervention did not reduce the rate of cardiovascular events in overweight or obese adults with type 2 diabetes. It is noteworthy that although weight loss was greater in the intervention group than in the control group, there was only an initial improvement in fitness and only when related to weight loss. The exercise training was not supervised and it appears that the intervention actually had very little effect on physical fitness 108.
Exercise and Type 1 diabetes
Type 1 diabetes is an autoimmune disease that occurs in children or adults. The disease is caused by the destruction of beta cells in the pancreas, which stops production of insulin. The etiology is still unknown, but environmental factors (e.g., viruses and chemicals), genetic disposition and autoimmune reactions all play a part.
Patients with type 1 diabetes have a high risk of developing cardiovascular disease 109, and physical activity offers good prevention 110. It is therefore important for patients with type 1 diabetes to be physically active on a regular basis. Insulin requirement decreases during physical activity, which is why patients must reduce their insulin dose if they plan to do physical training 111 and/or ingest carbohydrates in connection with training 112. Patients with type 1 diabetes thus need guidance on how to avoid hypoglycemia so that they, like others, can benefit from the positive effects of physical activity against other diseases.
A systematic review from 2014 analyzed physical activity interventions in children and young people with Type 1 diabetes mellitus. A total of 26 articles (10 randomized and 16 non-randomized studies), published in the period 1964–2012, were reviewed. Meta-analyses showed potential benefits of physical activity on HbA1c, BMI, triglycerides, and cholesterol 113.
There are relatively few studies that shed light on the specific impact of training in patients with type 1 diabetes, but in general little or no difference in glycemic control can be identified in patients with type 1 diabetes who are physically active compared to those who are inactive 114. Some studies find no improvement in HbA1c with physical training 115, whereas other find that the most physically active patients have the lowest HbA1c 116.
A large study included 4655 patients and found an inverse dose–response association was found between physical activity level and HbA1c 116. Another study showed that intense physical activity was associated with better metabolic control in patients with type 1 diabetes.
An observational, cross-sectional study included 130 adult patients with type 1 diabetes. The study found no differences in HbA1c levels in relation to time dedicated to moderate physical activities. However, patients who dedicated more than 150 min/week to intense physical activity had lower levels of HbA1c (HbA1c: 7.2 ± 1.0% vs 7.8 ± 1.1% vs 8.0 ± 1.0% in more than 149 min, between 0 and 149 min or 0 min of intense physical activity per week, respectively) 117.
On the other hand patients with type 1 diabetes – like non-diabetics – improve insulin sensitivity 118, which is associated with a lower (ca. 5%) reduction in the exogenous insulin requirements 119. Endothelial dysfunction is a trait of some 120, though not all 121, patients with type 1 diabetes, and the effect of physical training on this parameter is only sparsely illuminated. Endothelial function has been found to be both improved 122 and unchanged 114 after physical training.
Physical training possibly has a positive impact on the lipid profile, also in patients with type 1 diabetes. Controlled studies show that training reduces the level of LDL cholesterol and triglycerides in the blood 123 and increases the level of HDL cholesterol 123 and HDL cholesterol/total cholesterol ratio 118. The ratio, however, has not been thoroughly investigated and there might also be a difference between the sexes 124. In uncontrolled or cross-sectional trials a link has been found between training and an increase in HDL2 cholesterol and a decrease in serum triglyceride and LDL cholesterol 125.
A randomized controlled trial examined the effect of 30–60 min of running at a moderate intensity 3–5 times a week over 12–16 weeks. The study included young men with type 1 diabetes (n = 28 and the control group n = 28). Aerobic training increased fitness, exercise capacity, and improved lipid profile 123. A controlled study showed that 4 months of aerobic training increased fitness by 27%, reduced insulin requirement 126, and improved endothelial function 127 in patients with type 1 diabetes.
Exercise and Hypertension
Hypertension is a significant risk factor for stroke, acute myocardial infarction, heart failure, and sudden death. The borderline between low and normal blood pressure is fuzzy, as the incidence of these cardiovascular diseases already rises from a relatively low blood pressure level. A meta-analysis involving 61 prospective studies (1 million people) showed a linear relationship between decrease in the risk of cardiovascular mortality and decrease in blood pressure to a systolic blood pressure of below 115 mmHg and a diastolic blood pressure of below 75 mmHg 128. A decrease of 20 mmHg in systolic blood pressure or 10 mmHg in diastolic blood pressure halves the risk of cardiovascular mortality. Thus, for example, a person with systolic blood pressure of 120 mmHg has half the risk of cardiovascular mortality as a person with systolic blood pressure of 140 mmHg 128. Hypertension is defined as systolic blood pressure >140 and diastolic blood pressure >90 mmHg. According to this definition, about 20% of the population have high blood pressure or require blood pressure-lowering medication 129. However, the borderlines between optimal and normal blood pressure and between mild, moderate, and severe hypertension are arbitrary 129. Large-scale epidemiological studies indicate that regular physical exercise and/or fitness prevents hypertension or lowers blood pressure 130.
Effect on resting blood pressure (normotensive and hypertensive)
Several meta-analyses have concluded that physical exercise has a positive effect on blood pressure in both normotensive and hypertensive cases 131.
A meta-analysis included randomized controlled trials lasting ≥4 weeks investigating the effects of exercise on blood pressure in healthy adults (age ≥18 years) 132.
The study included 93 trials, involving 105 endurance, 29 dynamic resistance, 14 combined, and 5 isometric resistance groups, totaling 5223 participants (3401 exercise and 1822 control). Systolic BP (SBP) was reduced after endurance (−3.5 mmHg), dynamic resistance (−1.8 mmHg), and isometric resistance (−10.9 mmHg) but not after combined training. Reductions in diastolic BP (DBP) were observed after endurance (−2.5 mmHg), dynamic resistance (−3.2 mmHg), isometric resistance (−6.2 mmHg), and combined (−2.2 mmHg) training. BP reductions after endurance training were greater in 26 study groups of hypertensive subjects (−8.3 mmHg) than in 50 groups of pre-hypertensive subjects (−2.1) and 29 groups of subjects with normal BP levels (−0.75). BP reductions after dynamic resistance training were largest for pre-hypertensive participants (−4.0 mmHg) compared with patients with hypertension or normal BP. It was concluded that endurance, dynamic resistance, and isometric resistance training lower SBP and DBP, whereas combined training lowers only DBP. Data from a small number of isometric resistance training studies suggest this form of training has the potential for the largest reductions in SBP.
A meta-analysis from 2010 focused specifically on the effect of isometric exercise, which has not traditionally been recommended as an alternative to dynamic exercise 133. Five trials were identified including a total of 122 subjects. Isometric exercise for <1 h/week reduced systolic blood pressure by 10.4 mmHg and diastolic blood pressure by 6.7 mmHg. Also this study found that isometric exercise induces changes in blood pressure that are similar to that of endurance or dynamic resistance training and similar to those achieved with a single pharmacological agent. Interestingly, a smaller study suggested that even handgrip exercise had effect on blood pressure-lowering effects 134.
Another meta-analysis from 2013 included aerobic exercise training studies among previously sedentary older adults 135. Twenty-three studies, representing a total of 1226 older subjects, were included in the final analysis. Robust statistically significant effects were found when older exercisers were compared with the control group, representing a 3.9% reduction in SBP and a 4.5% reduction in DBP.
A meta-analysis was carried out in 2007 136 involving randomized controlled trials in which the training consisted of either endurance or resistance training. The meta-analysis was based on 72 trials and 105 study groups. Physical training was found to induce a significant reduction in resting blood pressure and systolic/diastolic blood pressure, measured during outpatient visits, of 3.0/2.4 mmHg and 3.3/3.5 mmHg, respectively. The reduction in blood pressure was more pronounced for the 30 hypersensitive trial groups, in which an effect of −6.9/−4.9 was achieved, while the normotensive group achieved an effect of −1.9/−1.6. Training had a positive effect on a number of clinical and paraclinical variables, namely systemic vascular resistance, plasma noradrenalin, plasma renin activity, body weight, abdominal girth, fat percentage, HOMA, and HDL cholesterol.
An expert panel of the American College of Sports Medicine (ACSM) 137 extrapolated data from a total of 16 studies involving patients with hypertension (systolic blood pressure >140 mmHg; diastolic blood pressure >90 mmHg) and found the effect of physical training in people with hypertension to be a decrease in blood pressure of 7.4 mmHg (systolic) and 5.8 mmHg (diastolic). A general finding was that the blood pressure-lowering effect of physical training was most pronounced in the patients with the highest blood pressure.
A meta-analysis from 2011 identified studies that had examined the effect of strength training on blood pressure and other cardiovascular risk factors in adults 138.
The study included 28 randomized, controlled trials, involving 33 study groups and 1012 participants. Overall, resistance training induced a significant blood pressure reduction in 28 normotensive or pre-hypertensive study groups [−3.9/−3.9 mmHg], whereas the reduction [−4.1/−1.5 mmHg] was not significant for the five hypertensive study groups. When study groups were divided according to the mode of training, isometric handgrip training in three groups resulted in a larger decrease in blood pressure [−13.5 /−6.1 mmHg] than dynamic resistance training in 30 groups [−2.8/−2.7 mmHg]. This meta-analysis supports the blood pressure-lowering potential of dynamic resistance training and point at an interesting effect of isometric handgrip training. The latter study adds to previous meta-analysis 139.
Blood pressure was measured daily (24 h) in 11 studies 137 and showed the same effect from training as the studies mentioned above.
Acute effect of physical activity
Physical activity induced a decrease in blood pressure after it was carried out. This decrease in blood pressure typically lasted for 4–10 h, but was measured up to 22 h later. The average decrease was 15 mmHg and 4 mmHg for systolic and diastolic blood pressure, respectively137. This means that people with hypertension can achieve normotensive values many hours of the day, which should be seen as having considerable clinical significance 137.
Overall, it is well documented that training for hypersensitive people induces a clinically relevant lowering of blood pressure. Conventional treatment using blood pressure-lowering medication typically brings about a decrease in diastolic blood pressure of the same level 140, which in the long run lowers the risk of strokes by an estimated 30% and the risk of ischemic cardiac death by 30%. A meta-analysis involving one million people calculates that a reduction in systolic blood pressure of just 2 mmHg reduces stroke mortality by 10% and ischemic cardiac death mortality by 7% among middle-aged people 128. These calculations coincide with findings from earlier analyses 141.
Exercise and Stroke
Stroke (cerebrovascular accident, brain attack) is defined by WHO as a rapid onset disorder of brain function with symptoms lasting more than 24 h or leading to death, and where the probable cause is vascular. The reasons are infarction due to cardiac embolism, intracerebral hemorrhage, or subarachnoid hemorrhage after ruptured aneurysm. The average age is 75 years, but 20% of the patients are less than 65 years of age. Depending on localization of the brain damage, different parts of the brain functions are affected, but the majority of stroke patients have unilateral paresis of the upper and lower extremities, while about one-third also have aphasia. Moreover, most of the patients need hospitalization and will require rehabilitation 142.
Most stroke patients are affected cognitively and emotionally after their attack. Approximately one-third of them experience post-stroke depression 143. These effects coupled with low physical function make it difficult to comply with recommendations for physical activity. Patients with stroke generally have low levels of physical activity 144.
Physical inactivity is a risk factor for atherosclerosis and hypertension, which explains why physical inactivity in epidemiological studies is a prognostic factor for apoplexy 145. Stroke patients who have a relatively high level of physical activity have been found to have comparatively fewer severe stroke episodes and better recovery of function after 2 years 146.
There is evidence that aerobic exercise in patients with stroke has a positive effect on walking speed and function. Furthermore, there is support for an effect on mortality.
A meta-analysis from 2014 included 38 randomized controlled trials. There was high evidence that in the subacute stage of stroke, specific walking training resulted in improved walking speed and distance compared with traditional walking training of the same intensity. In the chronic stage, walking training resulted in increased walking speed and walking distance compared with no/placebo treatment, and increased walking speed compared with overall physiotherapy. On average, 24 training sessions for 7 weeks were needed 147.
A meta-analysis from 2013 of randomized trials included nine studies of treadmill training comprising 977 participants and found evidence that, for people with stroke who can walk, treadmill training without body weight support results in faster walking speed and greater distance than no intervention/non-walking intervention and the benefit is maintained beyond the training period 148.
Another study from 2013 included meta-analyses of randomized controlled trials with mortality outcomes comparing the effectiveness of exercise and drug interventions with each other or with control (placebo or usual care). In total, they included 16 (four exercise and 12 drug) meta-analyses. Three trials concerned exercise interventions among patients with stroke (n = 227) compared with 10 trials of anticoagulants (n = 22 786), 14 of antiplatelets (n = 43 041), and three directly comparing anticoagulants with antiplatelets (n = 11 567). Physical activity interventions were more effective than drug treatment among patients with stroke 149.
As physical activity helps to prevent risk factors for stroke, i.e., hypertension, atherosclerosis, and type 2 diabetes, it is likely that physical training of stroke patients can prevent new episodes of stroke, but there is no evidence to support this.
A Cochrane Review from 2013 included 45 trials, involving 2188 participants, which comprised cardiorespiratory (22 trials, 995 participants), resistance (eight trials, 275 participants), and mixed training interventions (15 trials, 918 participants). It was concluded that there is sufficient evidence to incorporate cardiorespiratory and mixed training, involving walking, within post-stroke rehabilitation programs to improve the speed and tolerance of walking; improvement in balance may also occur. Presently, there is however insufficient evidence to support the use of resistance training 150.
However, individuals with hemiparesis are often older and have a low level of activity, and it must be assumed that this group can achieve the same advantages from strength training as individuals without neurological deficits. In other words, physically active people with hemiparesis will have lower mortality, more active years, reduced risk of metabolic syndrome, and increased bone density 151.
Exercise and Coronary heart disease
The term coronary heart disease (CHD) refers to a pathophysiological condition where a decrease in blood flow to the heart muscle causes ischemia, i.e., reduces oxygen supply. The most common cause is atherosclerotic constriction in the coronary arteries, but myocardial ischemia can also occur in patients with heart valve disease, hypertrophic cardiomyopathy, severe hypertension and abnormal tendency toward coronary spasm. Level of physical activity and cardio-respiratory fitness correlate with cardiovascular endpoints in healthy people and in patients with CHD 104.
There is solid evidence demonstrating the effect of physical training on patients with coronary heart disease. Physical training improves survival rates and is assumed to have a direct effect on the pathogenesis of the disease.
A Cochrane Review from 2011 aimed at determining the effectiveness of exercise-based cardiac rehabilitation (exercise training alone or in combination with psychosocial or educational interventions) on mortality, morbidity and health-related quality of life of patients with CHD 152. The study included men and women of all ages who have had myocardial infarction (MI), coronary artery bypass graft (CABG), or percutaneous transluminal coronary angioplasty (PTCA), or who have angina pectoris or coronary artery disease defined by angiography. The Cochrane systematic review included 47 studies randomizing 10 794 patients to exercise-based cardiac rehabilitation or usual care. In medium to longer term (i.e., 12 or more months follow-up) exercise-based cardiac rehabilitation reduced overall and cardiovascular mortality [RR: 0.87 and 0.74 respectively], and hospital admissions [RR: 0.69] in the shorter term (<12 months follow-up). Cardiac rehabilitation did not reduce the risk of total MI, CABG, or PTCA. In 7of 10 trials reporting health-related quality of life using validated measures was there evidence of a significantly higher level of quality of life with exercise-based cardiac rehabilitation than usual care. Thus, it was concluded that exercise-based cardiac rehabilitation is effective in reducing total and cardiovascular mortality (in medium- to long-term studies) and hospital admissions (in short-term studies) but not total MI or revascularization (CABG or PTCA). The latter review is in agreement with previous meta-analyses 153.
Physical training of patients with CHD was found to reduce total cholesterol and triglyceride levels and systolic blood pressure. Many of the subjects in the training groups had stopped smoking 153.
Exercise and Heart failure
Heart failure is a condition where the heart is unable to pump sufficiently to maintain blood flow to meet the metabolic needs of the peripheral tissue 154. Heart failure is a clinical syndrome with symptoms such as fluid retention, breathlessness, or excessive tiredness when resting or exercising, and with objective symptoms of reduced systolic function of the left ventricle at rest.
Asymptomatic left ventricular dysfunction is often the precursor of this syndrome. Symptoms vary from very light restriction of function to seriously disabling symptoms. Heart failure is often divided into left-sided (the most frequent and best researched type) and right-sided heart failure and into acute (pulmonary edema, cardiogenic shock) and chronic heart failure. Heart failure is often caused by ischemic disease, but can also be caused by, for example, hypertension or valvular heart disease 154.
Maximum oxygen uptake (VO2max) is lower in patients with heart failure 155. This is caused, among other things, by the reduced pumping function of the heart and by peripheral conditions in the muscles 156. A common symptom with heart failure patients is muscle atrophy, tiredness, and reduced muscle strength 157. Heart failure patients are characterized by defects in the renin–angiotensin system, increased levels of cytokines, also TNF, increased noradrenalin, and insulin resistance. These metabolic conditions can all be significant factors in the development of muscle atrophy with heart failure, although no direct link between VO2max and noradrenalin has been found. Thus, heart failure patients tend to have poor fitness, poor muscle strength and muscle atrophy. The characteristic symptom of tiredness is presumably related to diminishing physical ability. While there was a consensus in the 1970s that patients with all stages of heart failure should be advised to refrain from physical activity and be prescribed bed rest, the consensus now is the opposite 155.
Exercise and Intermittent claudication
Arterial insufficiency in the lower limbs (lower limb ischemia, leg ischemia) is a chronic obstructive disease in the aorta below the outlet of the renal arteries, iliac artery and the arteries in the lower limbs probably caused by atherosclerosis. It is estimated that at least 4% of all people above the age of 65 have peripheral arteriosclerosis, which in 50% of cases causes intermittent pain (intermittent claudication). A minority of patients experience the progression of peripheral arteriosclerosis, which results in pain while at rest and ulcerations. Owing to the realization that medical treatment of the disease has limited efficacy, the international consensus today is that physical training is a key factor in the treatment of patients with intermittent claudication 158. As the intermittent claudication becomes more severe, function level decreases and quality of life becomes affected. Increasing pain when walking and the consequent fear of moving gradually causes the patient to become static and socially isolated. In the long term, this leads to deterioration of fitness and the progression of arteriosclerosis, reduced muscle strength and muscle atrophy, trapping the patient in a vicious circle of poor fitness, pain, and social isolation. Physical activity can be used to interrupt this vicious circle and directly affect the pathogenesis of the disease by increasing fitness and muscle strength, changing pain thresholds and the perception of pain, allaying fear, and preventing disease progression.
There is solid evidence demonstrating the beneficial effect of physical training on patients with intermittent claudication. A Cochrane Reviews from 2014 159 included 30 trials, involving a total of 1816 participants with stable leg pain. The follow-up period ranged from 2 weeks to 2 years. The types of exercise varied from strength training to polestriding and upper or lower limb exercises; generally supervised sessions were at least twice a week. Most trials used a treadmill walking test for one of the outcome measures. Twenty trials compared exercise with usual care or placebo, the remainder of the trials compared exercise to medication or pneumatic calf compression. Overall, when taking the first time point reported in each of the studies, exercise significantly improved maximal walking time when compared with usual care or placebo: mean difference (MD) 4.51 min with an overall improvement in walking ability of approximately 50% to 200%. Walking distances were also significantly improved: pain-free walking distance MD 82.29 meters and maximum walking distance MD 108.99 meters. Improvements were seen for up to 2 years. The effect of exercise, when compared with placebo or usual care, was inconclusive on mortality, amputation, and peak exercise calf blood flow due to limited data. At 3 months, physical function, vitality, and role physical were reported to be improved with exercise in two trials. At 6 months, five trials reported outcomes of a significantly improved physical summary score and mental summary score secondary to exercise. The authors concluded that exercise programs are of significant benefit compared with placebo or usual care in improving walking time and distance in people with leg pain from intermittent claudication who were considered to be fit for exercise intervention. This was obtained without any demonstrable effect on the patients’ ankle blood pressure measurements. The results were not conclusive for mortality and amputation.
In a controlled study, physical training was compared with percutaneous transluminal angioplasty (PTA) and the finding was that there was no significant difference after 6 months 160. A review article by Chong et al. 161 assessed the results of physical training (9 studies, 294 patients) and PTA (12 studies, 2071 patients). The authors concluded that it was essentially impossible to compare the effect of two treatments in one non-controlled study design, but reported that the effect of PTA was minimally better than training, although PTA involved the risk of serious side effects.
A randomized study compared the effect of (a) physical exercise alone, (b) surgery, and (c) physical exercise + surgery. All groups achieved the same effect on walking distance, but there were side effects in 18% of the patients who underwent surgery 162.
Another randomized study compared the effect of physical training and antithrombotic therapy 163. A significantly larger improvement in walking distance was recorded for the group that exercised (86%) compared with the group that received medication (38%). A meta-analysis found that training programs were substantially cheaper and involved fewer risks than either surgery or PTA 164. Furthermore, a meta-analysis has shown that quality of life increases with walking distance 165.
A recent review analyzed the safety of supervised exercise training in patients with intermittent claudication 166. There has been concern regarding the safety of performing supervised exercise training because intermittent claudication patients are at risk for cardiovascular events. The review selected 121 articles, of which 74 met the inclusion criteria. Studies represent 82 725 h of training in 2876 patients with intermittent claudication. Eight adverse events were reported, six of cardiac, and two of non-cardiac origin, resulting in an only all-cause complication rate of one event per 10 340 patient-hours.
Exercise and Chronic obstructive pulmonary disease
Chronic obstructive pulmonary disease (COPD) is characterized by an irreversible decrease in lung function. Advanced-stage COPD is a long and painful process of gradually increasing and ultimately disabling breathlessness as the main symptom. Today the international consensus is that rehabilitation programs are an important part of COPD treatment, which follows from the realization that drug therapy for COPD is inadequate.
A vicious cycle of deterioration in physical capacity, shortness of breath, anxiety, and social isolation develops. Rehabilitation can break this cycle by introducing physical training, psychological support and networking with other COPD patients 167.
Reduction in muscle strength is a major cause of reduced exercise capacity and physical functional level 168. A minor study showed that muscle mass in the quadriceps was approximately 15% less and muscle strength about 50% lower in elderly men with COPD than in healthy, physically inactive peers 169.
The positive impact of physical exercise for patients with COPD is well documented. A 2015 Cochrane Review/meta-analysis 170 added to previous meta-analyses. The 2015 update includes 65 RCTs involving 3822 participants. A total of 41 of the pulmonary rehabilitation programs were hospital based, 23 were community based and one study had both a hospital component and a community component. Most programs were of 12-week or 8-week duration with an overall range of 4–52 weeks. The authors found statistically significant improvement for all included outcomes. In four important domains of quality of life (Chronic Respiratory Questionnaire (CRQ) scores for dyspnea, fatigue, emotional function, and mastery), the effect was larger than the minimal clinically important difference of 0.5 units. Statistically significant improvements were noted in all domains of the St. George’s Respiratory Questionnaire, and improvement in total score was better than 4 units. Both functional exercise and maximal exercise showed statistically significant improvement. Researchers reported an increase in maximal exercise capacity [mean Wmax (W)] in participants allocated to pulmonary rehabilitation compared with usual care. In relation to functional exercise capacity, the 6-min walk distance mean treatment effect was greater than the threshold of clinical significance. The subgroup analysis, which compared hospital-based programs vs community-based programs, provided evidence of a significant difference in treatment effect between subgroups for all domains of the CRQ, with higher mean values, on average, in the hospital-based pulmonary rehabilitation group than in the community-based group. Subgroup analysis performed to look at the complexity of the pulmonary rehabilitation program provided no evidence of a significant difference in treatment effect between subgroups that received exercise only and those that received exercise combined with more complex interventions.
Studies show that rehabilitation programs lead to fewer hospitalizations and thus save resources 171. Most studies use high-intensity walking exercise. One study compared the effect of walking or cycling at 80% of VO2max vs working out in the form of Callanetics exercises and found that high-intensity training increased fitness while the workout program increased arm muscle stamina. Both programs had a positive effect on the experience of dyspnea 172. Oxygen treatment in conjunction with intensive training for patients with COPD increased training intensity and thus improved fitness in one study 173, but not in another 174. It is recommended that oxygenation therapy should be provided at the end of training if the patients are hypoxic or become desaturated during the training 175. Training to music gave better results than without music, presumably because patients who run with music perceive the physical exertion to be less, even though they are doing the same amount of exercise. Specific training for inspiratory muscles increased the stamina of these muscles but did not give the patients a lower perception of dyspnea or improved fitness 176. Thus, strong evidence exists that endurance training as part of pulmonary rehabilitation in patients with COPD improves exercise capacity and health-related quality of life. However, dyspnea limits the exercise intensity. Therefore, resistance training, which may cause less dyspnea, could be an alternative. Moreover, low muscle mass is associated with increased risk of mortality 177.
A recent systematic review 178 compared the effect of resistance and endurance training. The authors included eight randomized controlled trials (328 participants) and found that resistance training appeared to induce the same beneficial effects as endurance training. It was therefore recommended that resistance training should be considered according to patient preferences when designing a pulmonary rehabilitation program for patients with COPD. The same authors performed another systematic review 179 in which they assessed the efficiency of combining resistance training with endurance training compared with endurance training alone. For this analysis, they included 11 randomized controlled trials (331 participants) and 2 previous systematic reviews. They found equal improvements in quality of life, walking distance, and exercise capacity. However, they also found moderate evidence of a significant increase in leg muscle strength favoring a combination of resistance and endurance training and recommend that resistance training should be incorporated in rehabilitation of COPD together with endurance training.
Exercise and Bronchial asthma
Bronchial asthma (asthma) is a chronic inflammatory disorder characterized by episodic reversible impairment of pulmonary function and airway hyper-responsiveness to a variety of stimuli 180. Allergies are a major cause of asthma symptoms, especially in children, while many adults have asthma without an allergic component. Environmental factors, including tobacco smoke and air pollution, contribute to the development of asthma.
Physical exercise poses a particular problem for asthmatics as physical activity can provoke bronchoconstriction in most asthmatics 181. Regular physical activity is important in the rehabilitation of patients with asthma 182. Asthmatics need to be taught how to prevent exercise-induced symptoms in order to benefit, just like other people, from the positive effects of physical activity against other diseases. With children in particular, it is important that they are taught how physical activity can be adapted to asthma due to its importance for their motor and social development.
Exercise-induced asthma can be prevented by warming up thoroughly and by using a number of anti-asthma drugs, e.g., short- or long-acting beta-agonists, leukotriene antagonists, or chromones. Another way to help eliminate some exercise-induced symptoms is to adjust the prophylactic treatment so that the asthma and thus airway responsiveness are under control. Regular intake of anti-asthma medicine, especially inhaled steroids, is crucial to enabling physical training. Moreover, it is important to be aware of triggers such as airway infections or triggers in the surroundings where physical activity is carried out, e.g., pollen, mold fungus, cold, air pollution, and tobacco smoke. Some studies 183, but not in others 184, have found physical fitness to be poor in asthmatics. Irrespective of how physically fit the patient is, guidance and medicine are important to enabling all asthmatics the opportunity to be physically active without worrying about the symptoms.
A Cochrane Review from 2013 185 included randomized trials of people over 8 years of age with asthma who were randomized to undertake physical training or not. Physical training had to be undertaken for at least 20 min, two times a week, over a minimum period of 4 weeks. Twenty-one studies including 772 participants were included. Physical training was well tolerated with no adverse effects reported. None of the studies mentioned worsening of asthma symptoms following physical training. Physical training showed marked improvement in cardiopulmonary fitness as measured by an increase in maximum oxygen uptake; however, no statistically significant effects were observed for forced expiratory volume in 1 second, forced vital capacity, minute ventilation at maximal exercise, or peak expiratory flow rate. It was concluded that physical training showed a significant improvement in fitness, though no effects were observed in other measures of pulmonary function.
A Cochrane study concluded that swimming training is beneficial for children and adolescents with asthma 186.
A non-controlled trial showed that it is possible for adult asthmatics to participate in high-intensity training 187. The patients trained in an indoor swimming pool at 80–90% of maximum oxygen uptake (VO2max) for 45 min, initially once a week and then twice a week for 10 weeks. Physical fitness improved and there were fewer cases of exercise-induced asthma attacks, less anxiety in connection with physical exertion and less of a feeling of dyspnea. At the 3-year follow-up examination, 68% of the patients were still physically active and trained one to two times a week 187. Physical training has a positive effect on the psycho-social morbidity and quality of life of asthma patients 188.
Exercise and Cystic fibrosis
Cystic fibrosis is the most commonly occurring autosomal recessive, genetic, potentially life-threatening disease 189. Incidence among Caucasians is one in 2500. Cystic fibrosis is a system disease, but the predominant symptom is progressive obstructive pulmonary disease, which over time leads to respiratory failure and respiratory heart disease 190. Diminished pulmonary function restricts physical development, resulting in lower fitness and muscle function and the patients often develop osteoporosis and diabetes. Evidence exists that patients with cystic fibrosis have reduced physical fitness 191 and exercise has been identified as an independent predictor of mortality and morbidity in cystic fibrosis patients 192.
Moreover, exercise intolerance is associated with reduced pulmonary function as well as daily activity levels and infections. The goal of physical training for cystic fibrosis patients is to:
- Mobilize the mucus in the lungs and to stimulate an increase in mucociliary transport
- Achieve a satisfactory level of fitness and strength to be able to maintain a normal capacity for exercise
- Maintain normal mobility, especially of the chest, to ensure that mucus clearance therapy is effective
- Prevent osteoporosis and diseases related to physical inactivity
- Increase self-confidence.
In theory, physical training may lower the risk for diabetes and infectious episodes.
In general, the evidence is poor for the effect of physical training in patients with cystic fibrosis, but it has been found that physical activity improves exercise capacity, slows the decline in lung function, and improves quality of life in patients with cystic fibrosis 193.
In a Cochrane review from 2008 191, seven studies which included 231 participants met the inclusion criteria. The review provided some but limited evidence from both short- and long-term studies that aerobic or anaerobic physical training has a positive effect on primary outcomes (exercise capacity, strength, and lung function) but improvements are not consistent between studies.
A systematic 2010 meta-analysis assessed the effect of physical exercise on children with cystic fibrosis. The analysis covered only four randomized controlled trials 194 and included 221 children with cystic fibrosis. The selected studies are strikingly heterogeneous concerning the severity of the cystic fibrosis, duration of the training intervention, and the mix of aerobic exercise and resistance training. All in all physical training was found to have a positive effect on lung function, muscle strength and fitness 195.
Exercise and Depression
Depression is a common and important cause of morbidity and mortality worldwide. Depression is commonly treated with antidepressants and/or psychological therapy, but some people may prefer alternative approaches such as exercise.
A comprehensive study comprising 156 subjects over the age of 50 with severe depression randomized the patients to 4 months of aerobic exercise, 4 months of treatment with antidepressants (sertraline), and 4 months of treatment with both sertraline and physical training 196. The exercise groups received supervised training of a relatively high intensity three times a week. Each session began with 10 min of warm-up, 30 min of cycling or jogging, and 5 min of cool down. Participants were assigned individual training ranges equivalent to 70% to 85% of heart rate reserve calculated from the maximum heart rate achieved during the treadmill test. The intensity was checked three times during each training session. The medical treatment had a quicker initial effect, but after 4 months, there was no difference between the three groups for symptoms of depression 196. The patients were examined again after 10 months 197. This check-up showed that there were significantly lower levels of depression symptoms and fewer incidences of relapse in the exercise groups. When all of the patients were analyzed together using multivariate analysis, there was a reduced risk of depression symptoms if the patients were physically active, regardless of group. This last fact does not exclude that the least depressed individuals had the strongest desire to exercise.
The 2013 Cochrane Review 198 concluded that the tendency was that more sessions have a larger effect on mood than a smaller number of sessions, and that resistance and mixed training were more effective than aerobic training. It appears, however, that it is not possible to determine the optimum type, frequency, and duration of exercise, whether it should be performed supervised or unsupervised, indoors or outdoors, or in a group or alone.
A meta-analysis from 2015 found some evidence that exercise may be effective in treating depression during pregnancy but the conclusion is based on only six low-moderate quality trials 199. Another meta-analysis from 2014 found strong evidence for an effect of exercise on depression in patients with heart failure 200.
Exercise and Anxiety
There is some, but limited knowledge about the effects of physical activity as a treatment for anxiety 201, 202. In several randomized controlled trials involving subjects with a normal or increased level of anxiety who do not meet the criteria for psychiatric diagnosis, it has been shown that physical activity can reduce symptoms of anxiety and tension. It is uncertain, however, whether it has a long-lasting effect 203.
Classical studies from the 1970s examine individuals who experienced anxiety attacks when they had to ride the bus. When the subjects got on the bus, they were gripped by anxiety, had a high pulse, were sweating, and felt they were unable to breathe. The patients were asked to run for the bus so they had a high pulse and were breathless when they got on it. Consequently, their physical symptoms were at their maximum level and did not worsen when they got on the bus. The patients attributed their high heart rate, tendency to perspire, and breathlessness to running for the bus and their fear declined 204.
A meta-analysis from 2010 comprising 40 studies concludes that physical training reduces symptoms of anxiety in people with chronic illnesses, including cardiovascular disease, fibromyalgia, multiple sclerosis, mental disorders, cancer, and chronic obstructive pulmonary disease 205.
Exercise and Stress
There is some, though modest evidence that physical training can have a positive effect on psychological stress symptoms. Physically fit individuals exhibit fewer pronounced signs of the physiological activation associated with psychosocial stress 206.
An American study 207 examines the relationship between level of physical fitness and mental well-being. The 5451 adult subjects (aged 20–88) in the study completed a treadmill test to determine their fitness level and filled out a questionnaire asking them to indicate their participation in leisure and sports activities over the preceding 3 months. The subjects’ mental well-being was evaluated based on questionnaires asking about the presence of symptoms of depression. The subjects were divided into three groups, depending on their level of fitness as measured by the treadmill test. The subjects were then divided into an additional four groups based on self-reported participation in regular exercise activities (inactive, insufficiently active, sufficiently active, and very active). The study showed the subjects who were more physically fit and more physically active experienced fewer symptoms of depression. Furthermore, an association was found between physical fitness and overall mental well-being.
One intervention study 208 examines the effect of physical training on stress in adolescents (13- to 17-year olds). Sixty subjects were randomized into four groups. Over a period of 10 weeks, three of the groups completed training programs comprising, respectively, high-intensity aerobic exercise (70–75% of maximum pulse), moderately intense aerobic exercise (50–60% of maximum pulse), and stretching and flexibility training, while the last group did not exercise and thus served as the control group. Before and after the training program, the subjects completed questionnaires to determine self-reported stress levels (perceived stress scale), anxiety, and depression. They also did a step test to determine their level of fitness based on heart rate values. The group that did high-intensity cardio achieved a lower resting heart rate and improved diastolic blood pressure compared to the other groups. With regard to the self-reported stress level, the questionnaire results showed that the group that did high-intensity exercise had the greatest reduction in stress and anxiety symptoms. The findings from the study indicate that a relatively short period of training can have beneficial psychological effects in adolescents, including reducing stress if the exercise is of high intensity.
Another study showed, however, that moderately intense exercise can also reduce stress indicators 209. After 12 weeks of exercise, the subjects in the moderate-intensity exercise group [40–50% of maximum oxygen uptake (VO2max)] had a lower resting blood pressure and a lower blood pressure in response to a stress compared to the group that did high-intensity exercise (70–80% of VO2max).
The type of exercise also appears to determine whether it has a positive effect on stress 210. One study randomized healthy English police officers to either 10 weeks of aerobic training (n = 28) or 10 weeks of strength training (n = 24), while a group of 25 male police officers served as the control group. After the training period, the subjects in the aerobic exercise group had significantly less work-related stress than both the strength training and control group. There was, on the other hand, no difference in work-related stress between the strength training and control groups after the training period.
A systematic Review from 2014 211 suggests that qigong exercise immediately relieve anxiety among healthy adults, compared to lecture attendance and structured movements only.
In summary, there is thus some evidence to suggest that regular exercise and being physically fit can reduce stress levels. The degree of perceived stress is less apparent the higher one’s level of physical fitness is. There are divergent research findings in terms of whether to exercise at a high or moderate intensity to avoid stress, but aerobic exercise seems to have a better effect than strength training.
Exercise and Schizophrenia
Schizophrenia is the name for a group of mental disorders characterized by abnormal thoughts and emotions. Typical symptoms of schizophrenia are hallucinations, delusions, and thought disorder. Other symptoms include social withdrawal, lack of energy, impoverished language, lack of emotion and cognitive symptoms, such as problems with verbal learning, visual learning, social cognition, speed of information processing, and problems with forming and finding words. Life time risk is below 1% worldwide and schizophrenia affects 24 million people around the world 212.
A meta-analysis from 2015 213 identified 20 eligible studies. Exercise interventions have no significant effect on body mass index (BMI), but can improve physical fitness and other cardiometabolic risk factors. Psychiatric symptoms were significantly reduced by interventions using around 90 min of moderate-to-vigorous exercise per week. This amount of exercise was also reported to improve functioning, co-morbid disorders, and cognition.
A systematic review from 2012 included 10 randomized controlled trials; six of these studies addressed the use of aerobic and strength exercises. In two of these studies, yoga techniques also were investigated. Four studies addressed the use of progressive muscle relaxation. There is evidence that aerobic and strength exercises and yoga reduce psychiatric symptoms, state anxiety, and psychological distress and improve health-related quality of life, that aerobic exercise improves short-term memory, that progressive muscle relaxation reduces state anxiety and psychological distress, and that physical training therapy in general offers added value in the multidisciplinary care of people with schizophrenia 214. Routine physical activity/exercise appears to decrease the severity of negative symptoms 215, reduce stress and anxiety 216, improve concentration and attention 217, and reduce depression severity 218 in schizophrenia.
Older studies indicate that physical activity reduces auditory hallucinations 219. Other studies show that physically active patients experience auditory hallucinations as less distressing 220.
People with schizophrenia are often overweight because their medication stimulates their appetite and at the same time increases social withdrawal and a physically inactive lifestyle. A healthy diet and physical activity are found to affect patients’ weight and simple health parameters such as triglycerides, total cholesterol, plasma insulin, and plasma glucose in the same way as they are said to affect the weight of people who do not suffer from mental illness 221.
People with schizophrenia often have accompanying symptoms, such as anxiety and stress, and physical activity can help alleviate these symptoms. In some cases, physical activity can provide an infrastructure and an environment that support social interactions and thus help the patients to establish networks, counteracting the tendency toward social withdrawal. Patients with schizophrenia are found to have a lower hippocampus volume compared to healthy people, a finding that may be significant for the pathogenesis of the chronic psychotic symptoms 222. A 2010 study shows that aerobic exercise over a 3-month period led to an increase in the size of the hippocampus and an improvement in the short-term memory of people with schizophrenia 223.
Exercise and Dementia
Despite the strong evidence that physical exercise may lower the risk of dementia, there are relatively few studies allowing to conclude on the effects of exercise in patients with a diagnosis of dementia. According to a Cochrane review 2013, there is promising evidence that exercise programs can have a significant impact in improving ability to perform activity of daily living (ADL) and possibly in improving cognition in people with dementia, although some caution is advised in interpreting these findings 224.
Sixteen trials with 937 participants met the inclusion criteria. The included trials were highly heterogeneous in terms of subtype and severity of participants’ dementia, and type, duration, and frequency of exercise. Only two trials included participants living at home. The meta-analysis suggested that exercise programs might have a significant impact on improving cognitive functioning and found a significant effect of exercise programs on the ability of people with dementia to perform ADLs. It was further found that the burden experienced by informal caregivers providing care in the home may be reduced when they supervise the participation of the family member with dementia in an exercise program.
One study 225 included 31 elderly subjects with dementia randomized to a training group or a control group. The training program comprised 15 weeks of 1 h of physical activity three times a week. After 15 weeks, the training group had improved their cognitive function, while the control group experienced a decline in cognitive function. The training group improved their gate function, but the opposite was true in the control group. Similar to the previous study, other studies find that simple interventions in the form of a few hours of physical activity per week have a positive effect on physical function in elderly people with dementia 226.
Most older persons with dementia living in nursing homes spend their days without engaging in much physical activity. A systematic review therefore looked at the influence that the environment has on their level of physical activity. Three hundred and twenty-six studies were selected as potentially relevant; of these, 24 met all the inclusion criteria. Positive results on the residents’ levels of physical activity were found for music, a homelike environment and functional modifications 227.
Studies have also been undertaken examining whether physical activity affects the cognitive function of elderly people without dementia. Lautenschlager et al. 228 included 170 elderly subjects who experienced subjective memory impairment without dementia. The participants were randomized to either a control group or a training group, which did a 24-week home-based program with 3 × 50 min workouts a week. The training had a significant, albeit modest, positive effect on the participant’s cognitive function. The effect was still present 1 year after cessation of exercise 228.
Baker et al. 229 included 33 middle-aged and older people with mild cognitive impairment who were randomized to either a control group that did stretching and balance exercises or to a training group that did intensive aerobic training 45–60 min a day, 4 days a week for up to 6 months. There was a significant positive effect on the participants’ cognitive function measured with the help of a neurological test battery. The effect was twice as strong for women as for men 229.
Erickson et al. 230 included 120 elderly subjects who were randomized to either a control group that did stretching and muscle training or to a training group that walked three times a week for 40 min for a year at a brisk pace. The physical training had a significantly positive effect on the volume of the hippocampus and a non-significant effect on the participants’ spatial memory 230.
Overall, there is some evidence that physical activity prevents dementia but only modest evidence for an effect of physical activity on cognitive function in people who have already developed dementia. Physical training has a positive effect on physical function, for example, the gait function of people with dementia.
Exercise and Parkinson’s disease
Parkinson’s disease is the second most common neurodegenerative disease after Alzheimer’s disease and affects approximately seven million people globally. Parkinson’s disease is more common in the elderly and prevalence rises from 1% in those over 60 years of age to 4% of the population over 80. The mean age of onset is around 60 years, although 5–10% of cases, classified as young onset, begin between the ages of 20 and 50 231.
There is evidence pointing to the positive impact of physical training 232. A 2010 Cochrane Review 233 assessed the significance of treadmill training. The analysis included eight trials involving 203 participants. Treadmill training was found to increase walking speed, stride, and walking distance. The conclusions of the analysis echoed those of a systematic review from 2008 234. These analyses add to a 2001 meta-analysis 235 involving a wide range of therapy, all-round physical training, sensory training, and mobility training. The duration of the physical exercise was 3–21 weeks with a total of 9–157.5 h of training. Overall, it was established that the training regime had a significant impact on walking speed.
A prospective crossover study investigated the effects of 4 weeks of treadmill training with partial body weight support and general physiotherapy (n = 10). The study found that the aerobic exercise, unlike the unspecific physiotherapy, improved the patients’ ability to manage their daily lives (ADL) and their muscle function 236. In a later study, patients were randomized for the same form of training or physiotherapy 237 (n = 24) and were monitored for 6 months. The training had a sustainable effect, especially on gait function.
In one study, 33 patients were randomized to walking training over 4 weeks or to conventional physiotherapy. The walking training had a positive effect on gait function 238. Randomizing for 10 weeks’ training with or without supervision by the physiotherapist showed that the supervised training had a greater effect than the training program carried out by the patients independently at home 239.
Another study compared group boxing training to traditional group exercise on function and quality of life in persons with Parkinson’s disease. A convenience sample of adults with Parkinson’s disease (n = 31) were randomly assigned to boxing training or traditional exercise for 24–36 sessions, each lasting 90 min, over 12 weeks. Boxing training included: stretching, boxing (e.g., lateral foot work, punching bags), resistance exercises, and aerobic training. Traditional exercise included: stretching, resistance exercises, aerobic training, and balance activities. The traditional exercise group demonstrated greater gains in balance confidence than the boxing group. Only the boxing group demonstrated significant improvements in gait velocity and endurance over time. Both groups demonstrated significant improvements with the balance, mobility, and quality of life 240.
A 16-month randomized controlled exercise intervention investigated three exercise approaches: flexibility/balance/function exercise (FBF), supervised aerobic exercise (AE), and home-based exercise (control). The participants were 121 individuals with early- or mid-stage Parkinson’s disease. The FBF program (individualized spinal and extremity flexibility exercises followed by group balance/functional training) was supervised by a physical therapist. The AE program (using a treadmill, bike, or elliptical trainer) was supervised by an exercise trainer. Supervision was provided 3 days/week for 4 months, and then monthly (16 months total). The control group participants exercised at home using the National Parkinson Foundation Fitness Counts program, with one supervised, clinic-based group session/month. Of the 121 participants, 86.8%, 82.6%, and 79.3% completed 4, 10, and 16 months, respectively, of the intervention. Findings demonstrated overall functional benefits at 4 months in the FBF group and improved walking economy (up to 16 months) in the AE group. Thus, both FBF and AE programs may be important for people with early- and mid-stage Parkinson’s disease 241.
The latter study showed that supervised training may have long-term effects in Parkinson’s disease. A pilot study explored the feasibility, acceptability, and preliminary evidence of the effectiveness of a virtual exercise coach to promote daily walking in community-dwelling persons with Parkinson’s disease. Twenty patients participated in this phase 1, single-group, non-randomized clinical trial. The subjects were instructed to interact with the virtual exercise coach for 5 min, wear a pedometer, and walk daily for 1 month. At the study completion, there was 100% retention rate. Interaction history revealed that the participants logged in for a mean (SD) of 25.4 days of the recommended 30 days. The mean adherence to daily walking was 85%. Both gait speed and the 6-min walk test significantly improved 242.
Exercise Guidelines for Adults With Parkinson’s Disease
People with Parkinson’s who exercise fare better over time than those who are not active.
- Physical activity should be initiated early in the diagnosis and be a life-long commitment.
- Engaging in aerobic activity, along with other activities for strength, flexibility and balance, improves Parkinson’s symptoms and sense of well-being.
Why Aerobic Activities ?
- Aerobic activities make the body’s large muscles move in a rhythmic manner for a sustained period of time.
- Aerobic activities improve physical fitness, including strength and endurance.
- Aerobic activities have a positive effect on slowness and stiffness, as well as mood, and quality of life.
- Examples: brisk walking, swimming, cycling, dancing, water aerobics, skating, hiking, treadmill or elliptical, Wii.
Why Flexibility Activities ?
- Flexibility or stretching exercises improve mobility, increase range of motion, and reduce stiffness.
- Improving range of motion affects posture and walking ability making everyday activities easier.
- Examples: Tai Chi, stretching.
Why Strengthening Activities ?
- Strengthening activities improve muscle strength, walking speed, posture and overall physical fitness.
- Improving strength will help everyday activities, such as getting up from a chair, easier to manage.
- Examples: yard work or gardening, weights/resistance (free weights, elastic bands, body weight)
Why Balance Activities ?
- Balance activities improve posture and stability.
- Better balance reduces the fear of falling and helps in performing daily tasks.
- Examples: Yoga, hiking, Wii.
Exercise and Multiple sclerosis
Multiple sclerosis is a chronic disease normally resulting in gradual, progressive disability. The number of people with multiple sclerosis is 2–2.5 million (approximately 30 per 100 000) globally, with rates varying widely in different regions. The disease occurs more often in women than in men and it usually develops between the ages of 20–40. It is characterized by recurring neurological deficits (attacks) in different parts of the nervous system caused by local demyelination processes (plaques). Over time, the symptoms spread to different parts of the body. Individual attacks can manifest themselves in highly different ways, but common symptoms are paresis, disturbed sensation, ataxia, loss of autonomic functions, weakness, and fatigue. The symptomatology of each patient is different, depending on the location of the plaques, which makes evidence-based studies difficult to carry out.
A systematic review from 2013 included 54 studies and found strong evidence that exercise performed two times/week at a moderate intensity increases aerobic capacity and muscular strength. The authors concluded that among those with mild to moderate disability from multiple sclerosis, there is sufficient evidence that exercise training is effective for improving both aerobic capacity and muscular strength and that exercise may improve mobility, fatigue, and health-related quality of life 244.
It has been suggested that physical exercise might have the potential to have an impact on multiple sclerosis pathology and thereby slow down the disease process in patients with multiple sclerosis. However, it was recently concluded that although some evidence supports the possibility of a disease-modifying potential of exercise (or physical activity) in MS patients, future studies using better methodologies are needed to confirm this 245.
A systematic review from 2012 evaluated the effect of resistance training 246. Sixteen studies were included. The authors found strong evidence regarding the beneficial effect of progressive resistance training on muscle strength. Regarding functional capacity, balance, and self-reported measures (fatigue, quality of life, and mood) evidence is less strong, but the tendency is overall positive. Indications of an effect on underlying mechanisms such as muscle morphological changes, neural adaptations and cytokines also exist, but the studies investigating these aspects are few and inconclusive 246. A 2009 meta-analysis 247, which includes 22 studies involving nearly 600 people with multiple sclerosis, assesses the effect of physical training on walking ability. The physical training involved physiotherapy both with and without equipment and different forms of physical training on land and in water. Walking ability was found to improvement by 19% and this increased to 32% if the training was supervised. The meta-analysis found that the physical training described above has an overall positive effect on patients’ quality of life 248.
A 2007 Cochrane Review evaluates the impact of multi-disciplinary interventions 249. The main conclusion of the study, and that of a 2005 Cochrane Review, is that further research is needed on the effects of structured physical training for this patient group 250.
A randomized controlled study assessed the effects of progressive strength training and identified a clear impact on strength and function after 12 weeks 251, as well as an improvement in depression score, fatigue symptoms, and quality of life 252.
The importance of aerobic exercise was assessed in a randomized controlled trial involving 54 patients with multiple sclerosis 253, which was randomized to a control group or training program consisting of 3 × 40 min of combined arm and leg bicycle ergometry over 15 weeks. The training group increased maximum oxygen uptake (VO2max), improved leg and arm muscle strength, improved bladder function, and showed fewer depression and fatigue symptoms. There was also an improvement in lipid profile.
Another randomized control trial 254 involving 18 patients with multiple sclerosis found that 6 months of physical training increased mobility but had only a moderate effect on walking ability. Two randomized controlled trials showed that it was possible to increase inspiratory and expiratory muscle strength and thus cough force after 3 months of training for the respiratory muscles 255.
A 2001 meta-analysis involving 23 studies shows that ergotherapy/physiotherapy increases muscle strength, mobility, and physical well-being, and improves the ability to carry out daily functions such as getting dressed and taking care of personal hygiene 256.
A meta-analysis from 2014 concludes that the available evidence indicates that exercise training can yield a small, yet statistically significant and reliable reduction in depressive symptoms for people with multiple sclerosis 257.
parkbenefits, adults aged 18-64 years with multiple sclerosis who have mild to moderate disability need at least:
- 30 minutes of moderate intensity aerobic activity, 2 times per week, AND
- Strength training exercises for major muscle groups, 2 times per week.
Meeting these guidelines may also reduce fatigue, improve mobility and enhance elements of health-related quality of life.
What is known about the relationship between exercise and cancer risk ?
Physical activity (exercise) is essential for people to maintain a balance between the number of calories consumed and the number of calories used. Consistently expending fewer calories than are consumed leads to obesity, which scientists have convincingly linked to increased risks of 13 different cancers 259. Additionally, evidence indicates that physical activity may reduce the risks of several cancers through other mechanisms, independent of its effect on obesity.
There is substantial evidence that higher levels of physical activity are linked to lower risks of several cancers 260.
- Colon cancer: Colon cancer is one of the most extensively studied cancers in relation to physical activity 261. A 2009 meta-analysis of 52 epidemiologic studies that examined the association between physical activity and colon cancer risk found that the most physically active individuals had a 24% lower risk of colon cancer than those who were the least physically active 262. A pooled analysis of data on leisure-time physical activity (activities done at an individual’s discretion generally to improve or maintain fitness or health) from 12 prospective U.S. and European cohort studies reported a risk reduction of 16%, when comparing individuals who were most active to those where least active 263. Incidence of both distal colon and proximal colon cancers is lower in people who are more physically active than in those who are less physically active 264, 265. Physical activity is also associated with a decreased risk of colon adenomas (polyps), a type of colon polyp that may develop into colon cancer 266. However, it is less clear whether physical activity is associated with lower risks that polyps that have been removed will come back 267, 268, 269.
- Breast cancer: Many studies show that physically active women have a lower risk of breast cancer than inactive women; in a 2013 meta-analysis of 31 prospective studies, the average breast cancer risk reduction associated with physical activity was 12% (12). Physical activity has been associated with a reduced risk of breast cancer in both premenopausal and postmenopausal women; however, the evidence for an association is stronger for postmenopausal breast cancer 270, 271, 272, 273. Women who increase their physical activity after menopause may also have a lower risk of breast cancer than women who do not 274, 273.
- Endometrial cancer: Many studies have examined the relationship between physical activity and the risk of endometrial cancer (cancer of the lining of the uterus). In a meta-analysis of 33 studies, the average endometrial cancer risk reduction associated with high versus low physical activity was 20% 275. There is some evidence that the association between physical activity and endometrial cancer risk may reflect the effect of physical activity on obesity, a known risk factor for endometrial cancer 275, 276, 277.
For a number of other cancers, there is more limited evidence of a relationship with physical activity. In a study of over 1 million individuals, leisure-time physical activity was linked to reduced risks of esophageal adenocarcinoma, liver cancer, gastric cardia cancer (a type of stomach cancer), kidney cancer, myeloid leukemia, myeloma, and cancers of the head and neck, rectum, and bladder 263. These results are generally corroborated by large cohort studies 278 or meta-analyses 279, 280, 281.
How might physical activity be linked to reduced risks of cancer ?
Exercise has a number of biological effects on the body, some of which have been proposed to explain associations with specific cancers, including:
- Lowering the levels of hormones, such as insulin and estrogen, and of certain growth factors that have been associated with cancer development and progression 282 [breast, colon]
- Helping to prevent obesity and decreasing the harmful effects of obesity, particularly the development of insulin resistance (failure of the body’s cells to respond to insulin)
- Reducing inflammation
- Improving immune system function
- Altering the metabolism of bile acids, resulting in decreased exposure of the gastrointestinal tract to these suspected carcinogens 283, 284 [colon]
- Reducing the amount of time it takes for food to travel through the digestive system, which decreases gastrointestinal tract exposure to possible carcinogens [colon]
Is physical activity beneficial for cancer survivors ?
Research indicates that physical activity may have beneficial effects for several aspects of cancer survivorship–specifically, weight gain, quality of life, cancer recurrence or progression, and prognosis (likelihood of survival) 285, 286. Most of the evidence for the potential benefits of physical activity in cancer survivors comes from people diagnosed with breast, prostate, or colorectal cancer 1.
- Weight gain. Both reduced physical activity and the side effects of cancer treatment can contribute to weight gain after a cancer diagnosis. In a cohort study (a type of epidemiologic study), weight gain after breast cancer diagnosis was linked to worse survival 287. In a 2012 meta-analysis of randomized controlled clinical trials examining physical activity in cancer survivors, physical activity was found to reduce both body mass index and body weight 288.
- Quality of life. A 2012 Cochrane Collaboration systematic review of controlled clinical trials of exercise interventions in cancer survivors indicated that physical activity may have beneficial effects on overall health-related quality of life and on specific quality-of-life issues, including body image/self-esteem, emotional well-being, sexuality, sleep disturbance, social functioning, anxiety, fatigue, and pain 289. In a 2012 meta-analysis of randomized controlled trials examining physical activity in cancer survivors, physical activity was found to reduce fatigue and depression and to improve physical functioning, social functioning, and mental health 288.
- Recurrence, progression, and survival. Being physically active after a cancer diagnosis is linked to better cancer-specific outcomes for several cancer types 290.
- Breast cancer: Consistent evidence from epidemiologic studies links physical activity after diagnosis with better breast cancer outcomes 291, 292. For example, a large cohort study found that women who exercised moderately (the equivalent of walking 3 to 5 hours per week at an average pace) after a breast cancer diagnosis had approximately 40% to 50% lower risks of breast cancer recurrence, death from breast cancer, and death from any cause compared with more sedentary women 293. The potential physical activity benefit with regard to death from breast cancer was most apparent in women with hormone receptor–positive tumors 293.Another prospective cohort study found that women who had breast cancer and who engaged in recreational physical activity roughly equivalent to walking at an average pace of 2 to 2.9 mph for 1 hour per week had a 35% to 49% lower risk of death from breast cancer compared with women who engaged in less physical activity 294.Colorectal cancer: Evidence from multiple epidemiologic studies suggests that physical activity after a colorectal cancer diagnosis is associated with reduced risks of dying from colorectal cancer 295. In a large prospective cohort of patients with colorectal cancer, those who engaged in leisure-time physical activity had a 31% lower risk of death than those who did not, independent of their leisure-time physical activity before diagnosis 296.Prostate cancer: Limited evidence from a few epidemiologic studies has suggested a possible link between physical activity and better outcomes among men diagnosed with prostate cancer. In one study, men with nonmetastatic prostate cancer who engaged in vigorous activity for at least 3 hours per week had a 61% lower risk of death from prostate cancer compared with men who engaged in vigorous activity for less than 1 hour per week 297. Another study of men with localized prostate cancer found that higher levels of physical activity were associated with reduced overall and prostate cancer–specific mortality 298.
Exercise and Polycystic ovarian syndrome
Polycystic ovary (PCO) is a term used when ovaries have numerous cysts, i.e., blisters, on their surface. PCO is diagnosed using ultrasound scanning. Most women are diagnosed with the condition in connection with examination for irregular menstruation or infertility 299. PCO occurs in approximately 20% of all women of child-bearing age. Another term is polycystic ovary syndrome (PCOS), which occurs in 15% of women. The term PCOS indicates that women diagnosed with PCO can also have several other signs of hormone disorders.
PCOS is diagnosed in women who fulfill at least two of the following criteria: (a) PCO established after ultrasound scan; (b) irregular or no ovulation; and/or (c) increased body hair or increased levels of testosterone in the blood. Other causes of these symptoms must be ruled out, as several other hormone disorders can produce the same symptoms as PCOS. One of the most common symptoms of PCOS is an irregular menstrual cycle with long intervals between menstruation and no menstruation. This can be an indication of an ovulation disorder. Women with PCOS often tend to be overweight with an increased waist-to-hip ratio, and a large number of them are severely overweight. Many also have excess body hair, for example facial hair. Others have acne and a tendency to have thin hair or hair loss. Increased levels of testosterone are another frequent symptom.
The various symptoms in connection with PCOS manifest themselves very differently in each individual. Some women with PCOS have very light symptoms while others are more affected. PCOS can be present for a number of years without clinical symptoms before it becomes apparent, often in connection with an increase in weight and physical inactivity. The symptoms can also change over the years. Initial symptoms can already occur at the age of 14.
For many years, research on PCOS has focused on fertility and on investigating how women with PCOS can have a normal pregnancy. Families of women with PCOS, however, often have a high incidence of type 2 diabetes. Women with PCOS often have insulin resistance, higher cholesterol in the blood, abdominal obesity, and early signs of atherosclerosis. As the definition of PCOS has changed throughout the years, it is difficult to clearly and conclusively interpret earlier and present-day research in the field, but the evidence points to women with PCOS having a higher risk of developing clinical cardiovascular disease and type 2 diabetes. Of note, young women with PCOS engaged in physical activities less than controls. Also, women with PCOS were less likely to be aware of the positive effects of exercise on their health.
There is some evidence that physical exercise can have an impact. There is a 2011 systematic review that consists of eight manuscripts, five randomized controlled trials and three cohort studies. All studies involved moderate physical exercise (aerobic or strength training) over a period of 12–24 weeks. Training was found to have a positive effect on ovulation, insulin resistance, and weight loss. It was, however, not possible to identify a specific form of training program that produced the optimum effect 300. This conclusion is supported by other systematic reviews 301. Training in connection with a hypocaloric diet leads to better body composition than diet alone 302 and exercise training enhances flow-mediated dilation, an adaptation associated with reduced cardiovascular disease risk. Of note, this effect occurred independent of changes in body composition 303.
Exercise while Pregnant and After having your Baby
It’s perfectly okay to to be physically active while you’re pregnant and after having your baby 304. The American College of Sports Medicine (ACSM) and American Congress of Obstetrics and Gynecology (ACOG) have similar guidelines that encourage pregnant women to engage in health-related physical activity during pregnancy 305.
Physical activity in pregnancy has minimal risks and has been shown to benefit most women, although some modification to exercise routines may be necessary because of normal anatomic and physiologic changes and fetal requirements. Women with uncomplicated pregnancies should be encouraged to engage in aerobic and strength-conditioning exercises before, during, and after pregnancy 305. Obstetrician–gynecologists and other obstetric care providers should carefully evaluate women with medical or obstetric complications before making recommendations on physical activity participation during pregnancy 305. Although frequently prescribed, bed rest is only rarely indicated and, in most cases, allowing ambulation should be considered. Regular physical activity during pregnancy improves or maintains physical fitness, helps with weight management, reduces the risk of gestational diabetes in obese women, and enhances psychologic well-being. An exercise program that leads to an eventual goal of moderate-intensity exercise for at least 20–30 minutes per day on most or all days of the week should be developed with the patient and adjusted as medically indicated 305.
Healthy women who already do vigorous-intensity aerobic activity, such as running, or large amounts of activity can continue doing so during and after their pregnancy provided they stay healthy and discuss with their health care provider how and when activity should be adjusted over time.
If you are a healthy pregnant or postpartum woman, physical activity is good for your overall health. For example, moderate-intensity physical activity, such as brisk walking, keeps your heart and lungs healthy during and after pregnancy. Physical activity also helps improve your mood throughout the postpartum period. After you have your baby, exercise helps maintain a healthy weight, and when combined with eating fewer calories helps with weight loss.
According to scientific evidence, the risks of moderate-intensity aerobic activity, such as brisk walking, are very low for healthy pregnant women. Physical activity does not increase your chances of low-birth weight, early delivery, or early pregnancy loss. It’s also not likely that the composition or amount of your breast milk or your baby’s growth will be affected by physical activity.
Unless you have a medical reason to avoid physical activity during or after your pregnancy, you can begin or continue moderate-intensity aerobic activity. If you begin physical activity during your pregnancy, start slowly and increase your amount gradually over time. While pregnant, you should avoid doing any activity that involves lying on your back or that puts you at risk of falling or abdominal injury, such as horseback riding, soccer, or basketball 304.
Is it safe to exercise during pregnancy ?
If you are healthy and your pregnancy is normal, it is safe to continue or start most types of exercise, but you may need to make a few changes. Physical activity does not increase your risk of miscarriage, low birth weight, or early delivery 306. However, it is important to discuss exercise with your obstetrician or other member of your health care team during your early prenatal visits. If your health care professional gives you the OK to exercise, you can decide together on an exercise routine that fits your needs and is safe during pregnancy.
Are there certain conditions that make exercise during pregnancy unsafe ?
Women with the following conditions or pregnancy complications should not exercise during pregnancy 306:
- Certain types of heart and lung diseases
- Cervical insufficiency (inability of the cervix to retain a pregnancy in the second trimester) or cerclage (a procedure in which the cervical opening is closed with stitches in order to prevent or delay preterm birth)
- Being pregnant with twins or triplets (or more) with risk factors for preterm labor
- Placenta previa (a condition in which the placenta lies very low in the uterus, so that the opening of the uterus is partially or completely covered) after 26 weeks of pregnancy.
- Preterm (born before 37 completed weeks of pregnancy) labor or ruptured membranes (your water has broken) during this pregnancy
- Preeclampsia or pregnancy-induced high blood pressure. Preeclampsia: A disorder that can occur during pregnancy or after childbirth in which there is high blood pressure and other signs of organ injury, such as an abnormal amount of protein in the urine, a low number of platelets, abnormal kidney or liver function, pain over the upper abdomen, fluid in the lungs, or a severe headache or changes in vision.
- Severe anemia. Abnormally low levels of blood or red blood cells in the bloodstream. Most cases are caused by iron deficiency or lack of iron.
What are the benefits of exercise during pregnancy ?
Regular exercise during pregnancy benefits you and your baby in these key ways:
- Reduces back pain
- Eases constipation
- May decrease your risk of gestational diabetes, preeclampsia, and cesarean delivery
- Promotes healthy weight gain during pregnancy
- Improves your overall general fitness and strengthens your heart and blood vessels
- Helps you to lose the baby weight after your baby is born
How much exercise should you do during pregnancy ?
The Centers for Disease Control and Prevention recommend that pregnant women get at least 150 minutes of moderate-intensity aerobic activity every week. An aerobic activity is one in which you move large muscles of the body (like those in the legs and arms) in a rhythmic way. Moderate intensity means you are moving enough to raise your heart rate and start sweating. You still can talk normally, but you cannot sing.
Examples of moderate-intensity aerobic activity include brisk walking and general gardening (raking, weeding, or digging). You can divide the 150 minutes into 30-minute workouts on 5 days of the week or into smaller 10-minute workouts throughout each day.
If you are new to exercise, start out slowly and gradually increase your activity. Begin with as little as 5 minutes a day. Add 5 minutes each week until you can stay active for 30 minutes a day.
If you were very active before pregnancy, you can keep doing the same workouts with your health care professional’s approval. However, if you start to lose weight, you may need to increase the number of calories that you eat.
What changes occur in the body during pregnancy that can affect your exercise routine ?
Your body goes through many changes during pregnancy. It is important to choose exercises that take these changes into account:
- Joints—The hormones made during pregnancy cause the ligaments that support your joints to become relaxed. This makes the joints more mobile and at risk of injury. Avoid jerky, bouncy, or high-impact motions that can increase your risk of being hurt.
- Balance—During pregnancy, the extra weight in the front of your body shifts your center of gravity. This places stress on joints and muscles, especially those in your pelvis and low back. Because you are less stable and more likely to lose your balance, you are at greater risk of falling.
- Breathing—When you exercise, oxygen and blood flow are directed to your muscles and away from other areas of your body. While you are pregnant, your need for oxygen increases. As your belly grows, you may become short of breath more easily because of increased pressure of the uterus on the diaphragm (a muscle that aids in breathing). These changes may affect your ability to do strenuous exercise, especially if you are overweight or obese.
What precautions should you take when exercising during pregnancy ?
There are a few precautions that pregnant women should keep in mind during exercise:
- Drink plenty of water before, during, and after your workout. Signs of dehydration include dizziness, a racing or pounding heart, and urinating only small amounts or having urine that is dark yellow.
- Wear a sports bra that gives lots of support to help protect your breasts. Later in pregnancy, a belly support belt may reduce discomfort while walking or running.
- Avoid becoming overheated, especially in the first trimester. Drink plenty of water, wear loose-fitting clothing, and exercise in a temperature-controlled room. Do not exercise outside when it is very hot or humid.
- Avoid standing still or lying flat on your back as much as possible. When you lie on your back, your uterus presses on a large vein that returns blood to the heart. Standing motionless can cause blood to pool in your legs and feet. Both of these positions can decrease the amount of blood returning to your heart and may cause your blood pressure to decrease for a short time.
What are some safe exercises you can do during pregnancy ?
Whether you are new to exercise or it already is part of your weekly routine, choose activities that experts agree are safest for pregnant women:
- Walking—Brisk walking gives a total body workout and is easy on the joints and muscles.
- Swimming and water workouts—Water workouts use many of the body’s muscles. The water supports your weight so you avoid injury and muscle strain. If you find brisk walking difficult because of low back pain, water exercise is a good way to stay active.
- Stationary bicycling—Because your growing belly can affect your balance and make you more prone to falls, riding a standard bicycle during pregnancy can be risky. Cycling on a stationary bike is a better choice.
- Modified yoga and modified Pilates—Yoga reduces stress, improves flexibility, and encourages stretching and focused breathing. There are even prenatal yoga and Pilates classes designed for pregnant women. These classes often teach modified poses that accommodate a pregnant woman’s shifting balance.
- You also should avoid poses that require you to be still or lie on your back for long periods.
If you are an experienced runner, jogger, or racquet-sports player, you may be able to keep doing these activities during pregnancy. Discuss these activities with your health care professional.
What exercises you should avoid during pregnancy ?
While pregnant, avoid activities that put you at increased risk of injury, such as the following:
- Contact sports and sports that put you at risk of getting hit in the abdomen, including ice hockey, boxing, soccer, and basketball
- Skydiving
- Activities that may result in a fall, such as downhill snow skiing, water skiing, surfing, off-road cycling, gymnastics, and horseback riding
- “Hot yoga” or “hot Pilates,” which may cause you to become overheated
- Scuba diving
- Activities performed above 6,000 feet (if you do not already live at a high altitude)
What are warning signs that you should stop exercising ?
Stop exercising and call your obstetrician or other member of your health care team if you have any of these signs or symptoms:
- Bleeding from the vagina
- Feeling dizzy or faint
- Shortness of breath before starting exercise
- Chest pain
- Headache
- Muscle weakness
- Calf pain or swelling
- Regular, painful contractions of the uterus
- Fluid leaking from the vagina
Why is it important to keep exercising after your baby is born ?
Exercising after your baby is born may help improve mood and decreases the risk of deep vein thrombosis, a condition that can occur more frequently in women in the weeks after childbirth. In addition to these health benefits, exercise after pregnancy can help you lose the extra pounds that you may have gained during pregnancy.
Exercise has the following benefits for postpartum women:
- It helps strengthen and tone abdominal muscles.
- It boosts energy.
- It may be useful in preventing postpartum depression.
- It promotes better sleep.
- It relieves stress.
How much exercise you should do after you have a baby ?
After having a baby, it is recommended that you get at least 150 minutes of moderate-intensity aerobic activity every week.
What is aerobic activity ?
An aerobic activity is one in which you move large muscles of the body (like those in the legs and arms) in a rhythmic way.
What is moderate-intensity activity ?
Moderate intensity means you are moving enough to raise your heart rate and start sweating. You can still talk normally, but you cannot sing. Examples of moderate-intensity aerobic activities include brisk walking and riding a bike on a level surface. You can choose to divide the 150 minutes into 30-minute workouts on 5 days of the week or into smaller 10-minute sessions throughout each day. For example, you could go for three 10-minute walks each day.
What is vigorous-intensity activity ?
A vigorous-intensity activity is one in which it is hard to talk without pausing for breath. If you followed a vigorous-intensity exercise program before pregnancy, it may be possible to return to your regular workouts soon after the baby is born. Be sure to get your health care provider’s approval. Examples include running, jumping rope, and swimming laps.
What are muscle-strengthening workouts and how often should you do them ?
This type of exercise works the body’s major muscle groups, such as the legs, arms, and hips. Examples include yoga, Pilates, lifting weights, sit-ups, and push-ups. There also are special Kegel exercises that help tone the muscles of the pelvic floor. Muscle-strengthening activities should be done in addition to your aerobic activity on at least 2 days a week.
When can you start exercising after pregnancy ?
If you had a healthy pregnancy and a normal vaginal delivery, you should be able to start exercising again soon after the baby is born. Usually, it is safe to begin exercising a few days after giving birth—or as soon as you feel ready. If you had a cesarean delivery or other complications, ask your health care provider when it is safe to begin exercising again.
What are some guidelines you can follow when you begin exercising after pregnancy ?
Aim to stay active for 20–30 minutes a day. When you first start exercising after childbirth, try simple postpartum exercises that help strengthen major muscle groups, including abdominal and back muscles. Gradually add moderate-intensity exercise. Remember, even 10 minutes of exercise benefits your body. If you exercised vigorously before pregnancy or you are a competitive athlete, you can work up to vigorous-intensity activity. Stop exercising if you feel pain.
What are some ways to start exercising ?
When you are ready to start exercising, walking is a great way to get back in shape. Walking outside has an added bonus because you can push your baby in a stroller. There are special strollers made for this kind of activity, but using a regular stroller is fine. Another good way to get daily exercise is by joining an exercise class. Working out with a group and socializing with group members can help keep you motivated.
Where you can find out about exercise classes ?
Check with your local fitness clubs or community centers for classes that interest you, such as yoga, Pilates, spinning, and dance. Some gyms even offer special postpartum exercise classes and classes you can take with your baby. You also might consider working out with a personal trainer for the first few weeks.
What can you do if you want to exercise but don’t want to join a gym ?
If you do not want to join a gym but want the benefits of having someone to exercise with, ask a friend to be your workout buddy. If you want to exercise on your own, check out fitness DVDs and online exercise programs. Many are designed for women who have just had a baby. Some even show you how to involve your baby in the exercise routines.
How you can stay motivated once you start exercising ?
You may already have a great exercise tool in your pocket. Smart phone apps for exercise and fitness can help you stay motivated, keep track of your progress, and connect you with others with the same exercise goals. Many apps are free or cost very little.
How should you prepare for your workout ?
As you get ready for your workout, follow these steps:
- Wear loose-fitting clothing that will help keep you cool.
- If you are breastfeeding, feed your baby or express your milk before your workout to avoid any discomfort that may come from engorged breasts.
- Wear a bra that fits well and gives plenty of support to protect your breasts.
- Have a bottle of water handy and take several sips during your workout.
How you should warm up before your workout ?
Spend 10 minutes warming up to get your muscles ready for exercise. Try stretches for the lower back, pelvis, and thighs. Hold stretches for several seconds and return to the starting position. Walking in place also is a good way to warm up.
How you should cool down after your workout ?
End your workout with a 5-minute cool-down period that brings your heart rate back to normal. Walk slowly in place and stretch again to help avoid soreness.
Exercise for Older Adults and is Essential for Healthy Aging
Physical Activity is essential to healthy aging. As an older adult, regular physical activity is one of the most important things you can do for your health. It can prevent many of the health problems that seem to come with age. It also helps your muscles grow stronger so you can keep doing your day-to-day activities without becoming dependent on others 304.
Not doing any physical activity can be bad for you, no matter your age or health condition. Keep in mind, some physical activity is better than none at all. Your health benefits will also increase with the more physical activity that you do.
If you’re 65 years of age or older, are generally fit, and have no limiting health conditions you can follow the guidelines listed below 304.
- Older adults need at least:
2 hours and 30 minutes (150 minutes) of moderate-intensity aerobic activity (i.e., brisk walking) every week and weight training muscle-strengthening activities on 2 or more days a week that work all major muscle groups (legs, hips, back, abdomen, chest, shoulders, and arms).
OR
1 hour and 15 minutes (75 minutes) of vigorous-intensity aerobic activity (i.e., jogging or running) every week and weight training muscle-strengthening activities on 2 or more days a week that work all major muscle groups (legs, hips, back, abdomen, chest, shoulders, and arms).
OR
An equivalent mix of moderate- and vigorous-intensity aerobic activity and weight training muscle-strengthening activities on 2 or more days a week that work all major muscle groups (legs, hips, back, abdomen, chest, shoulders, and arms).
- For Even Greater Health Benefits
5 hours (300 minutes) each week of moderate-intensity aerobic activity and weight training muscle-strengthening activities on 2 or more days a week that work all major muscle groups (legs, hips, back, abdomen, chest, shoulders, and arms).
OR
2 hours and 30 minutes (150 minutes) each week of vigorous-intensity aerobic activity and weight training muscle-strengthening activities on 2 or more days a week that work all major muscle groups (legs, hips, back, abdomen, chest, shoulders, and arms).
OR
An equivalent mix of moderate- and vigorous-intensity aerobic activity and weight training muscle-strengthening activities on 2 or more days a week that work all major muscle groups (legs, hips, back, abdomen, chest, shoulders, and arms).
- Muscle-strengthening activities
Besides aerobic activity, you need to do things to make your muscles stronger at least 2 days a week. These types of activities will help keep you from losing muscle as you get older.
To gain health benefits, muscle-strengthening activities need to be done to the point where it’s hard for you to do another repetition without help. A repetition is one complete movement of an activity, like lifting a weight or doing one sit-up. Try to do 8—12 repetitions per activity that count as 1 set. Try to do at least 1 set of muscle-strengthening activities, but to gain even more benefits, do 2 or 3 sets.
There are many ways you can strengthen your muscles, whether it’s at home or the gym. The activities you choose should work all the major muscle groups of your body (legs, hips, back, chest, abdomen, shoulders, and arms). You may want to try:
- Lifting weights
- Working with resistance bands
- Doing exercises that use your body weight for resistance (push ups, sit ups)
- Heavy gardening (digging, shoveling)
- Yoga
What’s the Signal To Maintain Health and overcome Aging ?
The master signal for health, optimism, energy, effectiveness, and youth — is serious exercise, six days a week, until the day you die. Seventy percent of aging is in our own hands! You can overcome 70 percent of normal aging, until almost the end of life. You can be roughly the same woman or man you were at forty-five or fifty until you are eighty and beyond.
Exercising for 45-60 minutes a day for 6 days a week, sounds like it’s quite a lot, but it is actually a terrific deal because the body’s decay signals are running, nonstop, 24 / 7, and they are very effective. After age forty you lose up to 10 percent of your muscle mass a decade and the same is true for bone mass, as well as for coordination and balance. The amazing news is that we are able to reverse all that—over 70 percent of it anyway—by moving, doing physical exercise a measly forty-five to sixty minutes a day, six days a week.
Most Americans get less than 10 minutes of moderate exercise every day.
The nerve impulse to contract a muscle also sends a tiny signal to build it up, creating a moment-to-moment chemical balance between growth and decay within the muscle. Billions of signals racing around to every cell in the body, all day long. The system is amazingly subtle and complex; think for a second about the millions of mini-signals to the muscles every instant, just to enable us to stand upright. But curiously, the signals are pretty simple. It’s always one of two things: Grow or atrophy. Grow or decay. That’s the message to the cells all day
long, throughout our lives. So here’s the connection: When you exercise fairly hard, you stress your muscles, and you actually injure them slightly. When cells sense the damage from exercise, they automatically release chemicals to start the inflammation. The chemicals that
control inflammation are called cytokines; hundreds of them are at work in your body, regulating growth and decay down to the microscopic level.
Exercise for Children
Children and adolescents should do 60 minutes (1 hour) or more of physical activity each day 307.
Just make sure your child or adolescent is doing three types of physical activity:
- Aerobic Activity
Aerobic activity should make up most of your child’s 60 or more minutes of physical activity each day. This can include either moderate-intensity aerobic activity, such as brisk walking, or vigorous-intensity activity, such as running. Be sure to include vigorous-intensity aerobic activity on at least 3 days per week.
- Muscle Strengthening
Include muscle strengthening activities, such as gymnastics or push-ups, at least 3 days per week as part of your child’s 60 or more minutes.
- Bone Strengthening
Include bone strengthening activities, such as jumping rope or running, at least 3 days per week as part of your child’s 60 or more minutes.
Some physical activity is better-suited for children than adolescents. For example, children do not usually need formal muscle-strengthening programs, such as lifting weights. Younger children usually strengthen their muscles when they do gymnastics, play on a jungle gym or climb trees. As children grow older and become adolescents, they may start structured weight programs. For example, they may do these types of programs along with their football or basketball team practice.

How much Exercise or Physical Activity do you need ?
When it comes to weight management, people vary greatly in how much physical activity they need. Here are some guidelines to follow:
- To maintain your weight
Work your way up to 150 minutes of moderate-intensity aerobic activity, 75 minutes of vigorous-intensity aerobic activity, or an equivalent mix of the two each week. Strong scientific evidence shows that physical activity can help you maintain your weight over time. However, the exact amount of physical activity needed to do this is not clear since it varies greatly from person to person. It’s possible that you may need to do more than the equivalent of 150 minutes of moderate-intensity activity a week to maintain your weight.
- To lose weight and keep it off
You will need a high amount of physical activity unless you also adjust your diet and reduce the amount of calories you’re eating and drinking. Getting to and staying at a healthy weight requires both regular physical activity and a healthy eating plan.
What do moderate and vigorous-intensity mean ?
To understand and measure the intensity (the level of effort required by a person to do an activity) of aerobic activity pay attention to how your physical activity affects your heart rate and breathing.
The talk test is a simple way to measure relative intensity. In general, if you’re doing moderate-intensity activity you can talk, but not sing, during the activity. If you’re doing vigorous-intensity activity, you will not be able to say more than a few words without pausing for a breath 308.
Absolute Intensity
The amount of energy used by the body per minute of activity. The table below lists examples of activities classified as moderate-intensity or vigorous-intensity based upon the amount of energy used by the body while doing the activity 308.
- Moderate: While performing the physical activity, if your breathing and heart rate is noticeably faster but you can still carry on a conversation — it’s probably moderately intense. Examples include—
+ Walking briskly (a 15-minute mile).
+ Light yard work (raking/bagging leaves or using a lawn mower).
+ Water aerobics
+ Bicycling slower than 10 miles per hour (biking at a casual pace)
+ Tennis (doubles)
+ Ballroom dancing
+ General gardening
+ Light snow shoveling.
+ Actively playing with children.
- Vigorous: Your heart rate is increased substantially and you are breathing too hard and fast to have a conversation, it’s probably vigorously intense. Examples include—
+ Jogging/running.
+ Hiking uphill or with a heavy backpack
+ Tennis (singles)
+ Aerobic dancing
+ Jumping rope
+ Swimming laps.
+ Rollerblading/inline skating at a brisk pace.
+ Cross-country skiing.
+ Most competitive sports (football, basketball, or soccer).
+ Jumping rope.
 Target Heart Rate and Estimated Maximum Heart Rate 309
Target Heart Rate and Estimated Maximum Heart Rate 309
One way of monitoring physical activity intensity is to determine whether a your pulse or heart rate is within the target zone during physical activity.
The basic way to calculate your maximum heart rate is to subtract your age from 220. For example, if you’re 45 years old, subtract 45 from 220 to get a maximum heart rate of 175. This is the maximum number of times your heart should beat per minute during exercise.
Maximum Heart Rate = 220 – Age
- For moderate-intensity physical activity, a person’s target heart rate should be 50 to 70% of his or her maximum heart rate. This maximum rate is based on the person’s age. An estimate of a person’s maximum age-related heart rate can be obtained by subtracting the person’s age from 220. For example, for a 50-year-old person, the estimated maximum age-related heart rate would be calculated as 220 – 50 years = 170 beats per minute (bpm). The 50% and 70% levels would be:
- 50% level: 170 x 0.50 = 85 bpm, and
70% level: 170 x 0.70 = 119 bpm
Thus, moderate-intensity physical activity for a 50-year-old person will require that the heart rate remains between 85 and 119 bpm during physical activity.
- For vigorous-intensity physical activity, a person’s target heart rate should be 70 to 85% of his or her maximum heart rate. To calculate this range, follow the same formula as used above, except change “50 and 70%” to “70 and 85%”. For example, for a 35-year-old person, the estimated maximum age-related heart rate would be calculated as 220 – 35 years = 185 beats per minute (bpm). The 70% and 85% levels would be:
- 70% level: 185 x 0.70 = 130 bpm, and
85% level: 185 x 0.85 = 157 bpm
Thus, vigorous-intensity physical activity for a 35-year-old person will require that the heart rate remains between 130 and 157 bpm during physical activity.
- Taking Your Heart Rate
Generally, to determine whether you are exercising within the heart rate target zone, you must stop exercising briefly to take your pulse. You can take the pulse at the neck, the wrist, or the chest. We recommend the wrist. You can feel the radial pulse on the artery of the wrist in line with the thumb. Place the tips of the index and middle fingers over the artery and press lightly. Do not use the thumb. Take a full 60-second count of the heartbeats, or take for 30 seconds and multiply by 2. Start the count on a beat, which is counted as “zero.” If this number falls between 85 and 119 bpm in the case of the 50-year-old person, he or she is active within the target range for moderate-intensity activity.
Exercise improves general well-being.
People should engage in adequate levels of physical activity throughout their lives. The range of psychological and emotional benefits you get from exercise may be just as rich as what it can offer your physical body. At least 30 minutes of regular, moderate-intensity physical activity on most days reduces the risk of cardiovascular disease, type 2 diabetes, colon cancer and breast cancer. Muscle strengthening and balance training can reduce falls and improve mobility among older adults. Physical activity may be required for weight control.
Here are just some of the mental boosts you can benefit from when you incorporate regular physical activity into your healthy lifestyle.
You’ll be less stressed out. If you can’t eliminate stress from your life, you can at least learn to cope with it better. According to the American Psychological Association, exercise may have a positive impact on neurochemicals involved in stress responses by giving the body the chance to practice dealing with stress. When you have the cardiovascular system working with the nervous system and the muscular system and all sorts of other systems in the body, it naturally becomes more efficient when faced with stressful situations.
You may not suffer from as much anxiety or depression.
While there are varying levels of severity in anxiety and depression, anyone suffering from either condition shouldn’t discount the power of physical activity. When you exercise, your brain releases feel-good chemicals that may help combat negative thoughts, reduce chemicals in the immune systems that can worsen depression and create a calming effect by increasing your body temperature.
You’ll slow down the aging process of your brain and improve memory.
As we age, our cognitive abilities begin to deteriorate, but health and lifestyle factors can play an important role in how fast that happens. In one study, researchers assessed cognition, resting cerebral blood flow and cardiovascular fitness in a group of adults ages 57-75 before, in the middle of and after their 12-week aerobic workout regimen. They found that the exercises helped the participants’ improve their memory, brain health and physical fitness.
Your thinking and decision-making capabilities may improve.
Anyone who really wants to maximize their brainpower and increase productivity knows how important it is to avoid being sedentary. One study that monitored the activity of mice found that increased levels of cardiovascular exercise can produce more brain cells. Another study found that strenuous exercises leads to increased levels of proteins in the brain, called brain-derived neurotrophic factor (or BDNF). All of this is great news for those of us who want to keep our brains healthy and performing optimally.
You’ll get a boost in confidence.
Exercise may be hard for some people. It’s both physically uncomfortable to do and mentally uncomfortable to keep it up regularly when you really just don’t feel like doing it. But with a good, solid habit comes increased strength and goal achievements, which can have a great effect on your confidence levels. And that’s not even counting the added bonus of seeing and feeling the physical changes in your body.
You’ll be more present.
We already covered how regular exercise can help with anxiety, but it’s worth noting here at the end that it can pretty much help with clearing out all the crazy thoughts that are flying through your head. For many people, exercise has a meditative effect on them. It forces you to focus on the moment and what you’re doing—feeling your muscles work and your heart pump. And if you can get outside in a park or on a trail while doing it, that’s even better!
There’s a lot to be said about how our bodies were made to move, and it’s important to remember this as we’re all sitting at our office desks, driving in our cars, lounging on our couches and staring at our smartphones. One could argue that it really lays the foundation for how we live our lives.
- Schmid D, Leitzmann MF. Association between physical activity and mortality among breast cancer and colorectal cancer survivors: a systematic review and meta-analysis. Annals of Oncology 2014; 25(7):1293-311. https://www.ncbi.nlm.nih.gov/pubmed/24644304[↩][↩]
- Biswas A, Oh PI, Faulkner GE, et al. Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults: a systematic review and meta-analysis. Annals of Internal Medicine 2015; 162(2):123-132. https://www.ncbi.nlm.nih.gov/pubmed/25599350[↩]
- Ekelund U, Steene-Johannessen J, Brown WJ, et al. Does physical activity attenuate, or even eliminate, the detrimental association of sitting time with mortality? A harmonised meta-analysis of data from more than 1 million men and women. Lancet 2016; 388(10051):1302-1310. https://www.ncbi.nlm.nih.gov/pubmed/27475271[↩]
- Fletcher GF, Blair SN, Blumenthal J, Caspersen C, Chaitman B, Epstein S, Falls H, Froelicher ES, Froelicher VF, Pina IL. Statement on exercise. Benefits and recommendations for physical activity programs for all Americans. A statement for health professionals by the committee on exercise and cardiac rehabilitation of the council on clinical cardiology, American Heart Association. Circulation. 1992;86:340-344.[↩]
- Centers for Disease Control and Prevention – Physical Activity for a Healthy Weight – https://www.cdc.gov/healthyweight/physical_activity/index.html[↩][↩]
- Haskell WL, Lee I-M, Pate RR, Powell KE, Blair SN, Franklin BA, Macera CA, Heath GW, Thompson PD, Bauman A. Physical activity and public health: Updated recommendation for adults. Med. Sci. Sports Exerc. 2007;39:1423-1434.[↩]
- Nelson ME, Rejeski WJ, Blair SN, Duncan PW, Judge JO, King AC, Macera CA, Castaneda-Sceppa C. Physical activity and public health in older adults. Med. Sci. Sports Exerc. 2007;39:1435-1445.[↩]
- US Department of Health and Human Services. Physical activity guidelines for Americans. 2008[↩]
- Smith SC, Jr., Benjamin EJ, Bonow RO, Braun LT, Creager MA, Franklin BA, Gibbons RJ, Grundy SM, Hiratzka LF, Jones DW, Lloyd-Jones DM, Minissian M, Mosca L, Peterson ED, Sacco RL, Spertus J, Stein JH, Taubert KA. AHA/ACCF secondary prevention and risk reduction therapy for patients with coronary and other atherosclerotic vascular disease: 2011 update: A guideline from the American Heart Association and American College of Cardiology Foundation endorsed by the World Heart Federation and the Preventive Cardiovascular Nurses Association. J Am Coll Cardiol. 2011;58:2432-2446.[↩]
- Prevalence of regular physical activity among adults–United States, 2001 and 2005. MMWR Morb Mortal Wkly Rep. 2007;56:1209-1212.[↩]
- Production of interleukin-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma interleukin-6. Steensberg A, van Hall G, Osada T, Sacchetti M, Saltin B, Klarlund Pedersen B. J Physiol. 2000 Nov 15; 529 Pt 1():237-42. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2270169/[↩]
- Transcriptional activation of the IL-6 gene in human contracting skeletal muscle: influence of muscle glycogen content. Keller C, Steensberg A, Pilegaard H, Osada T, Saltin B, Pedersen BK, Neufer PD. FASEB J. 2001 Dec; 15(14):2748-50. https://www.ncbi.nlm.nih.gov/pubmed/11687509/[↩]
- Contractile C2C12 myotube model for studying exercise-inducible responses in skeletal muscle. Nedachi T, Fujita H, Kanzaki M. Am J Physiol Endocrinol Metab. 2008 Nov; 295(5):E1191-204. https://www.ncbi.nlm.nih.gov/pubmed/18780777/[↩]
- Contractile activity of human skeletal muscle cells prevents insulin resistance by inhibiting pro-inflammatory signalling pathways. Lambernd S, Taube A, Schober A, Platzbecker B, Görgens SW, Schlich R, Jeruschke K, Weiss J, Eckardt K, Eckel J. Diabetologia. 2012 Apr; 55(4):1128-39. https://www.ncbi.nlm.nih.gov/pubmed/22282161/[↩]
- Electrical pulse stimulation of cultured human skeletal muscle cells as an in vitro model of exercise. Nikolić N, Bakke SS, Kase ET, Rudberg I, Flo Halle I, Rustan AC, Thoresen GH, Aas V. PLoS One. 2012; 7(3):e33203. https://www.ncbi.nlm.nih.gov/pubmed/22457744/[↩]
- Cytokine response of primary human myotubes in an in vitro exercise model. Scheler M, Irmler M, Lehr S, Hartwig S, Staiger H, Al-Hasani H, Beckers J, de Angelis MH, Häring HU, Weigert C. Am J Physiol Cell Physiol. 2013 Oct 15; 305(8):C877-86. https://www.ncbi.nlm.nih.gov/pubmed/23926130/[↩]
- Enhanced release of cytokines, interleukin-2 receptors, and neopterin after long-distance running. Sprenger H, Jacobs C, Nain M, Gressner AM, Prinz H, Wesemann W, Gemsa D. Clin Immunol Immunopathol. 1992 May; 63(2):188-95. https://www.ncbi.nlm.nih.gov/pubmed/1611721/[↩]
- Kapilevich LV, Zakharova AN, Kabachkova AV, Kironenko TA, Orlov SN. Dynamic and Static Exercises Differentially Affect Plasma Cytokine Content in Elite Endurance- and Strength-Trained Athletes and Untrained Volunteers. Frontiers in Physiology. 2017;8:35. doi:10.3389/fphys.2017.00035. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5276859/[↩]
- Beneficial health effects of exercise–the role of IL-6 as a myokine. Pedersen BK, Fischer CP. Trends Pharmacol Sci. 2007 Apr; 28(4):152-6. https://www.ncbi.nlm.nih.gov/pubmed/17331593/[↩]
- Interleukin-15 stimulates adiponectin secretion by 3T3-L1 adipocytes: evidence for a skeletal muscle-to-fat signaling pathway. Quinn LS, Strait-Bodey L, Anderson BG, Argilés JM, Havel PJ. Cell Biol Int. 2005 Jun; 29(6):449-57. https://www.ncbi.nlm.nih.gov/pubmed/15951205/[↩]
- Leukaemia inhibitory factor is expressed in rat gastrocnemius muscle after contusion and increases proliferation of rat L6 myoblasts via c-Myc signalling. Srikuea R, Esser KA, Pholpramool C. Clin Exp Pharmacol Physiol. 2011 Aug; 38(8):501-9. https://www.ncbi.nlm.nih.gov/pubmed/21585421/[↩]
- Exercise as medicine – evidence for prescribing exercise as therapy in 26 different chronic diseases. Pedersen BK, Saltin B. Scand J Med Sci Sports. 2015 Dec; 25 Suppl 3():1-72. http://onlinelibrary.wiley.com/doi/10.1111/sms.12581/full[↩]
- American Heart Association – What Exercise is Right for Me? – https://www.goredforwomen.org/live-healthy/heart-healthy-exercises/what-exercise-is-right-for-me/[↩]
- https://health.gov/dietaryguidelines/dga2005/document/html/chapter3.htm#table4[↩]
- Harvard University. Harvard Medical School. Calories burned in 30 minutes for people of three different weights. https://www.health.harvard.edu/diet-and-weight-loss/calories-burned-in-30-minutes-of-leisure-and-routine-activities[↩]
- Shaw K, Gennat H, O’Rourke P, Del Mar C. Exercise for overweight or obesity. Cochrane Database Syst Rev 2006: CD003817. https://www.ncbi.nlm.nih.gov/pubmed/17054187[↩]
- Johns DJ, Hartmann-Boyce J, Jebb SA, Aveyard P. Diet or exercise interventions vs combined behavioral weight management programs: a systematic review and meta-analysis of direct comparisons. J Acad Nutr Diet 2014: 114: 1557–1568. https://www.ncbi.nlm.nih.gov/pubmed/25257365[↩]
- Anderson JW, Konz EC, Frederich RC, Wood CL. Long-term weight-loss maintenance: a meta-analysis of US studies. Am J Clin Nutr 2001: 74: 579–584. https://www.ncbi.nlm.nih.gov/pubmed/11684524[↩]
- Svendsen OL, Hassager C, Christiansen C. Six months’ follow-up on exercise added to a short-term diet in overweight postmenopausal women – effects on body composition, resting metabolic rate, cardiovascular risk factors and bone. Int J Obes Relat Metab Disord 1994: 18: 692–698. https://www.ncbi.nlm.nih.gov/pubmed/7866465[↩]
- Donnelly JE, Smith B, Jacobsen DJ, Kirk E, Dubose K, Hyder M, Bailey B, Washburn R. The role of exercise for weight loss and maintenance. Best Pract Res Clin Gastroenterol 2004: 18: 1009–1029. https://www.ncbi.nlm.nih.gov/pubmed/15561636[↩]
- Fogelholm M, Kukkonen-Harjula K. Does physical activity prevent weight gain – a systematic review. Obes Rev 2000: 1: 95–111. https://www.ncbi.nlm.nih.gov/pubmed/12119991[↩]
- Crawford DA, Jeffery RW, French SA. Television viewing, physical inactivity and obesity. Int J Obes Relat Metab Disord 1999: 23: 437–440. https://www.ncbi.nlm.nih.gov/pubmed/10340824[↩]
- McGuire MT, Wing RR, Klem ML, Hill JO. Behavioral strategies of individuals who have maintained long-term weight losses. Obes Res 1999: 7: 334–341. https://www.ncbi.nlm.nih.gov/pubmed/10440589[↩]
- Sarlio-Lahteenkorva S, Rissanen A, Kaprio J. A descriptive study of weight loss maintenance: 6 and 15 year follow-up of initially overweight adults. Int J Obes Relat Metab Disord 2000: 24: 116–125. https://www.ncbi.nlm.nih.gov/pubmed/10702760[↩]
- Fogelholm M, Kukkonen-Harjula K, Nenonen A, Pasanen M. Effects of walking training on weight maintenance after a very-low-energy diet in premenopausal obese women: a randomized controlled trial. Arch Intern Med 2000: 160: 2177–2184. https://www.ncbi.nlm.nih.gov/pubmed/10904461[↩]
- Wadden TA, Vogt RA, Foster GD, Anderson DA. Exercise and the maintenance of weight loss: 1-year follow-up of a controlled clinical trial. J Consult Clin Psychol 1998: 66: 429–433. https://www.ncbi.nlm.nih.gov/pubmed/9583346[↩]
- Miller WC, Koceja DM, Hamilton EJ. A meta-analysis of the past 25 years of weight loss research using diet, exercise or diet plus exercise intervention. Int J Obes Relat Metab Disord 1997: 21: 941–947. https://www.ncbi.nlm.nih.gov/pubmed/9347414[↩]
- Fogelholm M. Physical activity, fitness and fatness: relations to mortality, morbidity and disease risk factors. A systematic review. Obes Rev 2010: 11: 202–221. https://www.ncbi.nlm.nih.gov/pubmed/19744231[↩]
- Esposito K, Giugliano F, Di Palo C, Giugliano G, Marfella R, D’Andrea F, D’Armiento M, Giugliano D. Effect of lifestyle changes on erectile dysfunction in obese men: a randomized controlled trial. JAMA 2004: 291: 2978–2984. https://www.ncbi.nlm.nih.gov/pubmed/15213209[↩]
- Wareham NJ, van Sluijs EM, Ekelund U. Physical activity and obesity prevention: a review of the current evidence. Proc Nutr Soc. 2005; 64:229-47. https://www.ncbi.nlm.nih.gov/pubmed/15960868[↩]
- Seo DC, Li K. Leisure-time physical activity dose-response effects on obesity among US adults: results from the 1999-2006 National Health and Nutrition Examination Survey. J Epidemiol Community Health. 2010; 64:426-31. http://jech.bmj.com/content/64/5/426.long[↩]
- Lewis CE, Smith DE, Wallace DD, Williams OD, Bild DE, Jacobs DR, Jr. Seven-year trends in body weight and associations with lifestyle and behavioral characteristics in black and white young adults: the CARDIA study. Am J Public Health. 1997; 87:635-42. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1380845/pdf/amjph00503-0109.pdf[↩]
- Lee IM, Djousse L, Sesso HD, Wang L, Buring JE. Physical activity and weight gain prevention. JAMA. 2010; 303:1173-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2846540/[↩]
- Lusk AC, Mekary RA, Feskanich D, Willett WC. Bicycle riding, walking, and weight gain in premenopausal women. Arch Intern Med. 2010; 170:1050-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3119355/[↩]
- Mekary RA, Feskanich D, Malspeis S, Hu FB, Willett WC, Field AE. Physical activity patterns and prevention of weight gain in premenopausal women. Int J Obes (Lond). 2009; 33:1039-47. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2746452/[↩]
- Mekary RA, Feskanich D, Hu FB, Willett WC, Field AE. Physical activity in relation to long-term maintenance after intentional weight loss in premenopausal women. Obesity (Silver Spring, Md). 2010;18(1):167-174. doi:10.1038/oby.2009.170. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2798010/[↩]
- U.S. Dept. of Health and Human Services. 2008 Physical Activity Guidelines for Americans; 2008. https://health.gov/paguidelines/[↩]
- Slentz CA, Aiken LB, Houmard JA, et al. Inactivity, exercise, and visceral fat. STRRIDE: a randomized, controlled study of exercise intensity and amount. J Appl Physiol. 2005; 99:1613-8. https://www.physiology.org/doi/pdf/10.1152/japplphysiol.00124.2005[↩][↩][↩]
- McTiernan A, Sorensen B, Irwin ML, et al. Exercise effect on weight and body fat in men and women. Obesity (Silver Spring). 2007; 15:1496-512. https://onlinelibrary.wiley.com/doi/pdf/10.1038/oby.2007.178[↩]
- Friedenreich CM, Woolcott CG, McTiernan A, et al. Adiposity changes after a 1-year aerobic exercise intervention among postmenopausal women: a randomized controlled trial. Int J Obes (Lond). 2010. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3061001/[↩][↩]
- Sallis JF, Glanz K. Physical activity and food environments: solutions to the obesity epidemic. Milbank Q. 2009; 87:123-54. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2879180/[↩]
- Physical Activity Guidelines for Americans. https://health.gov/paguidelines/[↩][↩][↩][↩][↩][↩][↩][↩]
- Lee, I.M., et al., Physical activity and weight gain prevention. JAMA, 2010. 303(12): p. 1173-9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2846540/[↩][↩]
- Jakicic, J.M., et al., Effect of exercise duration and intensity on weight loss in overweight, sedentary women: a randomized trial. JAMA, 2003. 290(10): p. 1323-30. https://www.ncbi.nlm.nih.gov/pubmed/12966123[↩]
- Williams, M.A., et al., Resistance exercise in individuals with and without cardiovascular disease: 2007 update: a scientific statement from the American Heart Association Council on Clinical Cardiology and Council on Nutrition, Physical Activity, and Metabolism. Circulation, 2007. 116(5): p. 572-84. http://circ.ahajournals.org/content/116/5/572.long[↩][↩]
- Schmitz, K.H., et al., Strength training and adiposity in premenopausal women: strong, healthy, and empowered study. Am J Clin Nutr, 2007. 86(3): p. 566-72. https://www.ncbi.nlm.nih.gov/pubmed/17823418[↩]
- Engelke, K., et al., Exercise maintains bone density at spine and hip EFOPS: a 3-year longitudinal study in early postmenopausal women. Osteoporos Int, 2006. 17(1): p. 133-42.[↩]
- Ling, C.H., et al., Handgrip strength and mortality in the oldest old population: the Leiden 85-plus study. CMAJ, 2010. 182(5): p. 429-35. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2842834[↩]
- Ruiz, J.R., et al., Association between muscular strength and mortality in men: prospective cohort study. BMJ, 2008. 337: p. a439. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2453303/[↩]
- Bolam, K.A., J.G. van Uffelen, and D.R. Taaffe, The effect of physical exercise on bone density in middle-aged and older men: A systematic review. Osteoporos Int, 2013. https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0055053/[↩][↩]
- Nelson, M.E., et al., Physical activity and public health in older adults: recommendation from the American College of Sports Medicine and the American Heart Association. Circulation, 2007. 116(9): p. 1094-105. http://circ.ahajournals.org/content/116/9/1094.long[↩][↩]
- National Heart, Lung and Blood Institute. Obesity education initiative expert panel: Clinical guidelines on the identification, evaluation and treatment of overweight and obesity in adults: The evidence report. 98-4083. Bethesda, MD: NIH, 1998: 1–228.[↩]
- Brown DR, Pate RR, Pratt M, Wheeler F, Buchner D, Ainsworth B, Macera C. Physical activity and public health: training courses for researchers and practitioners. Public Health Rep 2001: 116: 197–202. https://www.ncbi.nlm.nih.gov/pubmed/12034908[↩]
- National Institutes of Health Consensus Development Panel. Triglyceride, DLD, and CHD. JAMA 1993: 269: 505–520. https://www.ncbi.nlm.nih.gov/pubmed/8419671[↩]
- Forde OH, Thelle DS, Arnesen E, Mjos OD. Distribution of high density lipoprotein cholesterol according to relative body weight, cigarette smoking and leisure time physical activity. The Cardiovascular Disease Study in Finnmark 1977. Acta Med Scand 1986: 219: 167–171. https://www.ncbi.nlm.nih.gov/pubmed/3962733[↩]
- Mann S, Beedie C, Jimenez A. Differential effects of aerobic exercise, resistance training and combined exercise modalities on cholesterol and the lipid profile: review, synthesis and recommendations. Sports Med 2014: 44: 211–221. https://www.ncbi.nlm.nih.gov/pubmed/24174305[↩][↩]
- Kodama S, Tanaka S, Saito K, Shu M, Sone Y, Onitake F, Suzuki E, Shimano H, Yamamoto S, Kondo K, Ohashi Y, Yamada N, Sone H. Effect of aerobic exercise training on serum levels of high-density lipoprotein cholesterol: a meta-analysis. Arch Intern Med 2007: 167: 999–1008. https://www.ncbi.nlm.nih.gov/pubmed/17533202[↩]
- Knopp RH. Drug treatment of lipid disorders. N Engl J Med 1999: 341: 498–511. https://www.ncbi.nlm.nih.gov/pubmed/10441607[↩]
- Nicklas BJ, Katzel LI, Busby-Whitehead J, Goldberg AP. Increases in high-density lipoprotein cholesterol with endurance exercise training are blunted in obese compared with lean men. Metabolism 1997: 46: 556–561. https://www.ncbi.nlm.nih.gov/pubmed/9160824[↩]
- Kraus WE, Houmard JA, Duscha BD, Knetzger KJ, Wharton MB, McCartney JS, Bales CW, Henes S, Samsa GP, Otvos JD, Kulkarni KR, Slentz CA. Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med 2002: 347: 1483–1492. https://www.ncbi.nlm.nih.gov/pubmed/12421890[↩][↩]
- Strasser B, Siebert U, Schobersberger W. Resistance training in the treatment of the metabolic syndrome: a systematic review and meta-analysis of the effect of resistance training on metabolic clustering in patients with abnormal glucose metabolism. Sports Med 2010: 40: 397–415. https://www.ncbi.nlm.nih.gov/pubmed/20433212[↩]
- Hayashino Y, Jackson JL, Fukumori N, Nakamura F, Fukuhara S. Effects of supervised exercise on lipid profiles and blood pressure control in people with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Diabetes Res Clin Pract 2012: 98: 349–360. https://www.ncbi.nlm.nih.gov/pubmed/23116535[↩]
- Ford ES. Prevalence of the metabolic syndrome defined by the International Diabetes Federation among adults in the U.S. Diabetes Care 2005: 28: 2745–2749. https://www.ncbi.nlm.nih.gov/pubmed/16249550[↩]
- Ilanne-Parikka P, Laaksonen DE, Eriksson JG, Lakka TA, Lindstr J, Peltonen M, Aunola S, Keinanen-Kiukaanniemi S, Uusitupa M, Tuomilehto J. Leisure-time physical activity and the metabolic syndrome in the Finnish diabetes prevention study. Diabetes Care 2010: 33: 1610–1617. https://www.ncbi.nlm.nih.gov/pubmed/20413514[↩]
- Orozco LJ, Buchleitner AM, Gimenez-Perez G, Roque IF, Richter B, Mauricio D. Exercise or exercise and diet for preventing type 2 diabetes mellitus. Cochrane Database Syst Rev 2008: CD003054. https://www.ncbi.nlm.nih.gov/pubmed/18646086[↩][↩]
- Pan XR, Li GW, Hu YH, Wang JX, Yang WY, An ZX, Hu ZX, Lin J, Xiao JZ, Cao HB, Liu PA, Jiang XG, Jiang YY, Wang JP, Zheng H, Zhang H, Bennett PH, Howard BV. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes Study. Diabetes Care 1997: 20: 537–544. https://www.ncbi.nlm.nih.gov/pubmed/9096977[↩]
- Eriksson KF, Lindgarde F. No excess 12-year mortality in men with impaired glucose tolerance who participated in the Malmo Preventive Trial with diet and exercise. Diabetologia 1998: 41: 1010–1016. https://www.ncbi.nlm.nih.gov/pubmed/9754818[↩]
- Tuomilehto J, Lindstrom J, Eriksson JG, Valle TT, Hamalainen H, Ilanne-Parikka P, Keinanen-Kiukaanniemi S, Laakso M, Louheranta A, Rastas M, Salminen V, Uusitupa M. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001: 344: 1343–1350. https://www.ncbi.nlm.nih.gov/pubmed/11333990[↩][↩][↩][↩]
- Lindstrom J, Louheranta A, Mannelin M, Rastas M, Salminen V, Eriksson J, Uusitupa M, Tuomilehto J. The Finnish Diabetes Prevention Study (DPS): lifestyle intervention and 3-year results on diet and physical activity. Diabetes Care 2003b: 26: 3230–3236. https://www.ncbi.nlm.nih.gov/pubmed/14633807[↩]
- Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002: 346: 393–403. https://www.ncbi.nlm.nih.gov/pubmed/11832527[↩][↩][↩]
- O’Donovan G, Thomas EL, McCarthy JP, Fitzpatrick J, Durighel G, Mehta S, Morin SX, Goldstone AP, Bell JD. Fat distribution in men of different waist girth, fitness level and exercise habit. Int J Obes (Lond) 2009: 33: 1356–1362. https://www.ncbi.nlm.nih.gov/pubmed/19752873[↩]
- Olsen RH, Krogh-Madsen R, Thomsen C, Booth FW, Pedersen BK. Metabolic responses to reduced daily steps in healthy nonexercising men. JAMA 2008: 299: 1261–1263. https://www.ncbi.nlm.nih.gov/pubmed/18349087[↩]
- Janiszewski PM, Kuk JL, Ross R. Is the reduction of lower-body subcutaneous adipose tissue associated with elevations in risk factors for diabetes and cardiovascular disease? Diabetologia 2008: 51: 1475–1482. https://www.ncbi.nlm.nih.gov/pubmed/18535815[↩]
- Janssen I, Fortier A, Hudson R, Ross R. Effects of an energy-restrictive diet with or without exercise on abdominal fat, intermuscular fat, and metabolic risk factors in obese women. Diabetes Care 2002: 25: 431–438. www.ncbi.nlm.nih.gov/pubmed/11874926[↩]
- Ross R, Aru J, Freeman J, Hudson R, Janssen I. Abdominal adiposity and insulin resistance in obese men. Am J Physiol Endocrinol Metab 2002: 282: E657–E663. https://www.ncbi.nlm.nih.gov/pubmed/11832370[↩]
- Forouhi NG, Sattar N, McKeigue PM. Relation of C-reactive protein to body fat distribution and features of the metabolic syndrome in Europeans and South Asians. Int J Obes Relat Metab Disord 2001: 25: 1327–1331. https://www.ncbi.nlm.nih.gov/pubmed/11571595[↩]
- Hayashi T, Boyko EJ, Leonetti DL, McNeely MJ, Newell-Morris L, Kahn SE, Fujimoto WY. Visceral adiposity is an independent predictor of incident hypertension in Japanese Americans. Ann Intern Med 2004: 140: 992–1000. https://www.ncbi.nlm.nih.gov/pubmed/15197016[↩]
- Boyko EJ, Fujimoto WY, Leonetti DL, Newell-Morris L. Visceral adiposity and risk of type 2 diabetes: a prospective study among Japanese Americans. Diabetes Care 2000: 23: 465–471. https://www.ncbi.nlm.nih.gov/pubmed/10857936[↩]
- Kuk JL, Katzmarzyk PT, Nichaman MZ, Church TS, Blair SN, Ross R. Visceral fat is an independent predictor of all-cause mortality in men. Obesity (Silver Spring) 2006: 14: 336–341. https://www.ncbi.nlm.nih.gov/pubmed/16571861[↩]
- Janiszewski PM, Ross R. Physical activity in the treatment of obesity: beyond body weight reduction. Appl Physiol Nutr Metab 2007: 32: 512–522. https://www.ncbi.nlm.nih.gov/pubmed/17510691[↩]
- American Diabetes Association. Clinical practice recommendations. Diabetes Care 2002: Jan: S1–S147.[↩]
- Umpierre D, Ribeiro PA, Kramer CK, Leitao CB, Zucatti AT, Azevedo MJ, Gross JL, Ribeiro JP, Schaan BD. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. JAMA 2011: 305: 1790–1799. https://www.ncbi.nlm.nih.gov/pubmed/21540423[↩][↩]
- Thomas DE, Elliott EJ, Naughton GA. Exercise for type 2 diabetes mellitus. Cochrane Database Syst Rev 2006: 19: CD002968.[↩][↩][↩]
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34. Lancet 1998: 352: 854–865. https://www.ncbi.nlm.nih.gov/pubmed/9742977[↩]
- Boule NG, Haddad E, Kenny GP, Wells GA, Sigal RJ. Effects of exercise on glycemic control and body mass in type 2 diabetes mellitus: a meta-analysis of controlled clinical trials. JAMA 2001: 286: 1218–1227. https://www.ncbi.nlm.nih.gov/pubmed/11559268[↩][↩]
- Fox ML, Keteyian SJ. Fox’s physiological basis for exercise physiology. New York: McGraw-Hill Co., 1998.[↩]
- Mourier A, Gautier JF, De Kerviler E, Bigard AX, Villette JM, Garnier JP, Duvallet A, Guezennec CY, Cathelineau G. Mobilization of visceral adipose tissue related to the improvement in insulin sensitivity in response to physical training in NIDDM. Effects of branched-chain amino acid supplements. Diabetes Care 1997: 20: 385–391. https://www.ncbi.nlm.nih.gov/pubmed/9051392[↩]
- Conn VS, Hafdahl AR, Mehr DR, LeMaster JW, Brown SA, Nielsen PJ. Metabolic effects of interventions to increase exercise in adults with type 2 diabetes. Diabetologia 2007: 50: 913–921. https://www.ncbi.nlm.nih.gov/pubmed/17342472[↩]
- Irvine C, Taylor NF. Progressive resistance exercise improves glycaemic control in people with type 2 diabetes mellitus: a systematic review. Aust J Physiother 2009: 55: 237–246. https://www.ncbi.nlm.nih.gov/pubmed/19929766[↩]
- MacLeod SF, Terada T, Chahal BS, Boule NG. Exercise lowers postprandial glucose but not fasting glucose in type 2 diabetes: a meta-analysis of studies using continuous glucose monitoring. Diabetes Metab Res Rev 2013: 29: 593–603. https://www.ncbi.nlm.nih.gov/pubmed/24038928[↩]
- Kelley GA, Kelley KS. Effects of aerobic exercise on lipids and lipoproteins in adults with type 2 diabetes: a meta-analysis of randomized-controlled trials. Public Health 2007: 121: 643–655. https://www.ncbi.nlm.nih.gov/pubmed/17544042[↩]
- Yang Z, Scott CA, Mao C, Tang J, Farmer AJ. Resistance exercise versus aerobic exercise for type 2 diabetes: a systematic review and meta-analysis. Sports Med 2014: 44: 487–499. https://www.ncbi.nlm.nih.gov/pubmed/24297743[↩]
- Kearney ML, Thyfault JP. Exercise and postprandial glycemic control in type 2 diabetes. Curr Diabetes Rev 2015: doi: 10.2174/1573399811666150615112441. https://www.ncbi.nlm.nih.gov/pubmed/26073704[↩][↩]
- Myers J, Prakash M, Froelicher V, Do D, Partington S, Atwood JE. Exercise capacity and mortality among men referred for exercise testing. N Engl J Med 2002: 346: 793–801. https://www.ncbi.nlm.nih.gov/pubmed/11893790[↩][↩]
- Boule NG, Kenny GP, Haddad E, Wells GA, Sigal RJ. Meta-analysis of the effect of structured exercise training on cardiorespiratory fitness in Type 2 diabetes mellitus. Diabetologia 2003: 46: 1071–1081. https://www.ncbi.nlm.nih.gov/pubmed/12856082[↩]
- Brandon LJ, Gaasch DA, Boyette LW, Lloyd AM. Effects of long-term resistive training on mobility and strength in older adults with diabetes. J Gerontol A Biol Sci Med Sci 2003: 58: 740–745. https://www.ncbi.nlm.nih.gov/pubmed/12902533[↩]
- Nielsen PJ, Hafdahl AR, Conn VS, LeMaster JW, Brown SA. Meta-analysis of the effect of exercise interventions on fitness outcomes among adults with type 1 and type 2 diabetes. Diabetes Res Clin Pract 2006: 74: 111–120. https://www.ncbi.nlm.nih.gov/pubmed/16735074[↩]
- Wing RR, Bolin P, Brancati FL, Bray GA, Clark JM, Coday M, Crow RS, Curtis JM, Egan CM, Espeland MA, Evans M, Foreyt JP, Ghazarian S, Gregg EW, Harrison B, Hazuda HP, Hill JO, Horton ES, Hubbard VS, Jakicic JM, Jeffery RW, Johnson KC, Kahn SE, Kitabchi AE, Knowler WC, Lewis CE, Maschak-Carey BJ, Montez MG, Murillo A, Nathan DM, Patricio J, Peters A, Pi-Sunyer X, Pownall H, Reboussin D, Regensteiner JG, Rickman AD, Ryan DH, Safford M, Wadden TA, Wagenknecht LE, West DS, Williamson DF, Yanovski SZ. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med 2013: 369: 145–154. https://www.ncbi.nlm.nih.gov/pubmed/23796131[↩]
- Krolevski AS. Magnitude and determinants of coronary artery disease in juvenile-onset, insulin-dependent diabetes mellitus. Am J Cardiol 1987: 59: 750–755. https://www.ncbi.nlm.nih.gov/pubmed/3825934[↩]
- Moy CS, Songer TJ, LaPorte RE, Dorman JS, Kriska AM, Orchard TJ, Becker DJ, Drash AL. Insulin-dependent diabetes mellitus, physical activity, and death. Am J Epidemiol 1993: 137: 74–81. https://www.ncbi.nlm.nih.gov/pubmed/8434575[↩]
- Rabasa-Lhoret R, Bourque J, Ducros F, Chiasson JL. Guidelines for premeal insulin dose reduction for postprandial exercise of different intensities and durations in type 1 diabetic subjects treated intensively with a basal-bolus insulin regimen (ultralente-lispro). Diabetes Care 2001: 24: 625–630. https://www.ncbi.nlm.nih.gov/pubmed/11315820[↩]
- Soo K, Furler SM, Samaras K, Jenkins AB, Campbell LV, Chisholm DJ. Glycemic responses to exercise in IDDM after simple and complex carbohydrate supplementation. Diabetes Care 1996: 19: 575–579. https://www.ncbi.nlm.nih.gov/pubmed/8725854[↩]
- Quirk H, Blake H, Tennyson R, Randell TL, Glazebrook C. Physical activity interventions in children and young people with Type 1 diabetes mellitus: a systematic review with meta-analysis. Diabet Med 2014: 31: 1163–1173. https://www.ncbi.nlm.nih.gov/pubmed/24965376[↩]
- Veves A, Saouaf R, Donaghue VM, Mullooly CA, Kistler JA, Giurini JM, Horton ES, Fielding RA. Aerobic exercise capacity remains normal despite impaired endothelial function in the micro- and macrocirculation of physically active IDDM patients. Diabetes 1997: 46: 1846–1852. https://www.ncbi.nlm.nih.gov/pubmed/9356035[↩][↩]
- Kennedy A, Nirantharakumar K, Chimen M, Pang TT, Hemming K, Andrews RC, Narendran P. Does exercise improve glycaemic control in type 1 diabetes? A systematic review and meta-analysis. PLoS ONE 2013: 8: e58861. https://www.ncbi.nlm.nih.gov/pubmed/23554942[↩]
- Beraki A, Magnuson A, Sarnblad S, Aman J, Samuelsson U. Increase in physical activity is associated with lower HbA1c levels in children and adolescents with type 1 diabetes: results from a cross-sectional study based on the Swedish pediatric diabetes quality registry (SWEDIABKIDS). Diabetes Res Clin Pract 2014: 105: 119–125. https://www.ncbi.nlm.nih.gov/pubmed/24846445[↩][↩]
- Carral F, Gutierrez JV, Ayala MC, Garcia G, Aguilar M. Intense physical activity is associated with better metabolic control in patients with type 1 diabetes. Diabetes Res Clin Pract 2013: 101: 45–49. https://www.ncbi.nlm.nih.gov/pubmed/23684761[↩]
- Yki-Jarvinen H, DeFronzo RA, Koivisto VA. Normalization of insulin sensitivity in type I diabetic subjects by physical training during insulin pump therapy. Diabetes Care 1984: 7: 520–527. https://www.ncbi.nlm.nih.gov/pubmed/6391876[↩][↩]
- Wallberg-Henriksson H, Gunnarsson R, Henriksson J, Ostman J, Wahren J. Influence of physical training on formation of muscle capillaries in type I diabetes. Diabetes 1984: 33: 851–857. https://www.ncbi.nlm.nih.gov/pubmed/6468766[↩]
- Skyrme-Jones RA, O’Brien RC, Luo M, Meredith IT. Endothelial vasodilator function is related to low-density lipoprotein particle size and low-density lipoprotein vitamin E content in type 1 diabetes. J Am Coll Cardiol 2000: 35: 292–299. https://www.ncbi.nlm.nih.gov/pubmed/10676672[↩]
- Pinkney JH, Downs L, Hopton M, Mackness MI, Bolton CH. Endothelial dysfunction in Type 1 diabetes mellitus: relationship with LDL oxidation and the effects of vitamin E. Diabet Med 1999: 16: 993–999. https://www.ncbi.nlm.nih.gov/pubmed/10656227[↩]
- Seeger JP, Thijssen DH, Noordam K, Cranen ME, Hopman MT, Nijhuis-van der Sanden MW. Exercise training improves physical fitness and vascular function in children with type 1 diabetes. Diabetes Obes Metab 2011: 13: 382–384. https://www.ncbi.nlm.nih.gov/pubmed/21226819[↩]
- Laaksonen DE, Atalay M, Niskanen LK, Mustonen J, Sen CK, Lakka TA, Uusitupa MI. Aerobic exercise and the lipid profile in type 1 diabetic men: a randomized controlled trial. Med Sci Sports Exerc 2000: 32: 1541–1548. https://www.ncbi.nlm.nih.gov/pubmed/10994902[↩][↩][↩]
- Wallberg-Henriksson H, Gunnarsson R, Rossner S, Wahren J. Long-term physical training in female type 1 (insulin-dependent) diabetic patients: absence of significant effect on glycaemic control and lipoprotein levels. Diabetologia 1986: 29: 53–57. https://www.ncbi.nlm.nih.gov/pubmed/3956895[↩]
- Lehmann R, Kaplan V, Bingisser R, Bloch KE, Spinas GA. Impact of physical activity on cardiovascular risk factors in IDDM. Diabetes Care 1997: 20: 1603–1611. https://www.ncbi.nlm.nih.gov/pubmed/9314643[↩]
- Wiesinger GF, Pleiner J, Quittan M, Fuchsjager-Mayrl G, Crevenna R, Nuhr MJ, Francesconi C, Seit HP, Francesconi M, Fialka-Moser V, Wolzt M. Health related quality of life in patients with long-standing insulin dependent (type 1) diabetes mellitus: benefits of regular physical training. Wien Klin Wochenschr 2001: 113: 670–675. https://www.ncbi.nlm.nih.gov/pubmed/11603101[↩]
- Fuchsjager-Mayrl G, Pleiner J, Wiesinger GF, Sieder AE, Quittan M, Nuhr MJ, Francesconi C, Seit HP, Francesconi M, Schmetterer L, Wolzt M. Exercise training improves vascular endothelial function in patients with type 1 diabetes. Diabetes Care 2002: 25: 1795–1801. https://www.ncbi.nlm.nih.gov/pubmed/12351480[↩]
- Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002: 360: 1903–1913. https://www.ncbi.nlm.nih.gov/pubmed/12493255[↩][↩][↩]
- Burt VL, Whelton P, Roccella EJ, Brown C, Cutler JA, Higgins M, Horan MJ, Labarthe D. Prevalence of hypertension in the US adult population. Results from the Third National Health and Nutrition Examination Survey, 1988-1991. Hypertension 1995: 25: 305–313. https://www.ncbi.nlm.nih.gov/pubmed/7875754[↩][↩]
- Fagard RH, Cornelissen V. Physical activity, exercise, fitness and blood pressure. In: Battagay EJ, Lip GYH, Bakris GL, eds. Hypertension, principles and practice. Boca Raton: Taylor and Francis Books, 2005: 195–206.[↩]
- Carlson DJ, Dieberg G, Hess NC, Millar PJ, Smart NA. Isometric exercise training for blood pressure management: a systematic review and meta-analysis. Mayo Clin Proc 2014: 89: 327–334. https://www.ncbi.nlm.nih.gov/pubmed/24582191[↩]
- Cornelissen VA, Smart NA. Exercise training for blood pressure: a systematic review and meta-analysis. J Am Heart Assoc 2013: 2: e004473. https://www.ncbi.nlm.nih.gov/pubmed/23525435[↩]
- Owen A, Wiles J, Swaine I. Effect of isometric exercise on resting blood pressure: a meta analysis. J Hum Hypertens 2010: 24: 796–800. https://www.ncbi.nlm.nih.gov/pubmed/20182455[↩]
- Kelley GA, Kelley KS. Isometric handgrip exercise and resting blood pressure: a meta-analysis of randomized controlled trials. J Hypertens 2010: 28: 411–418. https://www.ncbi.nlm.nih.gov/pubmed/20009767[↩]
- Huang G, Shi X, Gibson CA, Huang SC, Coudret NA, Ehlman MC. Controlled aerobic exercise training reduces resting blood pressure in sedentary older adults. Blood Press 2013: 22: 386–394. https://www.ncbi.nlm.nih.gov/pubmed/23550511[↩]
- Fagard RH, Cornelissen VA. Effect of exercise on blood pressure control in hypertensive patients. Eur J Cardiovasc Prev Rehabil 2007: 14: 12–17. https://www.ncbi.nlm.nih.gov/pubmed/17301622[↩]
- Pescatello LS, Franklin BA, Fagard R, Farquhar WB, Kelley GA, Ray CA. American College of Sports Medicine position stand. Exercise and hypertension. Med Sci Sports Exerc 2004: 36: 533–553. https://www.ncbi.nlm.nih.gov/pubmed/15076798[↩][↩][↩][↩]
- Cornelissen VA, Fagard RH, Coeckelberghs E, Vanhees L. Impact of resistance training on blood pressure and other cardiovascular risk factors: a meta-analysis of randomized, controlled trials. Hypertension 2011: 58: 950–958. https://www.ncbi.nlm.nih.gov/pubmed/21896934[↩]
- Cornelissen VA, Fagard RH. Effect of resistance training on resting blood pressure: a meta-analysis of randomized controlled trials. J Hypertens 2005: 23: 251–259. https://www.ncbi.nlm.nih.gov/pubmed/15662209[↩]
- Blood Pressure Lowering Treatment Trialists’ Collaboration. Effects of ACE inhibitors, calcium antagonists, and other blood-pressure-lowering drugs: results of prospectively designed overviews of randomised trials. Lancet 2000: 355: 1955–1964. https://www.ncbi.nlm.nih.gov/pubmed/10859041[↩]
- Cook NR, Cohen J, Hebert PR, Taylor JO, Hennekens CH. Implications of small reductions in diastolic blood pressure for primary prevention. Arch Intern Med 1995: 155: 701–709. https://www.ncbi.nlm.nih.gov/pubmed/7695458[↩]
- Hisham NF, Bayraktutan U. Epidemiology, pathophysiology, and treatment of hypertension in ischaemic stroke patients. J Stroke Cerebrovasc Dis 2013: 22: e4–e14. https://www.ncbi.nlm.nih.gov/pubmed/22682972[↩]
- Paolucci S, Gandolfo C, Provinciali L, Torta R, Toso V. The Italian multicenter observational study on post-stroke depression (DESTRO). J Neurol 2006: 253: 556–562. https://www.ncbi.nlm.nih.gov/pubmed/16767539[↩]
- Rand D, Eng JJ, Tang PF, Jeng JS, Hung C. How active are people with stroke?: use of accelerometers to assess physical activity. Stroke 2009: 40: 163–168. https://www.ncbi.nlm.nih.gov/pubmed/18948606[↩]
- Boysen G, Krarup LH. Benefits of physical activity for stroke survivors. Expert Rev Neurother 2009: 9: 147–149. https://www.ncbi.nlm.nih.gov/pubmed/19210189[↩]
- Krarup LH, Truelsen T, Gluud C, Andersen G, Zeng X, Korv J, Oskedra A, Boysen G. Prestroke physical activity is associated with severity and long-term outcome from first-ever stroke. Neurology 2008: 71: 1313–1318. https://www.ncbi.nlm.nih.gov/pubmed/18936423[↩]
- Peurala SH, Karttunen AH, Sjogren T, Paltamaa J, Heinonen A. Evidence for the effectiveness of walking training on walking and self-care after stroke: a systematic review and meta-analysis of randomized controlled trials. J Rehabil Med 2014: 46: 387–399. https://www.ncbi.nlm.nih.gov/pubmed/24733289[↩]
- Polese JC, Ada L, Dean CM, Nascimento LR, Teixeira-Salmela LF. Treadmill training is effective for ambulatory adults with stroke: a systematic review. J Physiother 2013: 59: 73–80. https://www.ncbi.nlm.nih.gov/pubmed/23663792[↩]
- Naci H, Ioannidis JP. Comparative effectiveness of exercise and drug interventions on mortality outcomes: metaepidemiological study. BMJ 2013: 347: f5577. https://www.ncbi.nlm.nih.gov/pubmed/24473061[↩]
- Saunders DH, Sanderson M, Brazzelli M, Greig CA, Mead GE. Physical fitness training for stroke patients. Cochrane Database Syst Rev 2013: 10: CD003316. https://www.ncbi.nlm.nih.gov/pubmed/24142492[↩]
- Ryan AS, Ivey FM, Prior S, Li G, Hafer-Macko C. Skeletal muscle hypertrophy and muscle myostatin reduction after resistive training in stroke survivors. Stroke 2011: 42: 416–420. https://www.ncbi.nlm.nih.gov/pubmed/21164115[↩]
- Heran BS, Chen JM, Ebrahim S, Moxham T, Oldridge N, Rees K, Thompson DR, Taylor RS. Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst Rev 2011: CD001800. https://www.ncbi.nlm.nih.gov/pubmed/21735386[↩]
- Taylor RS, Brown A, Ebrahim S, Jolliffe J, Noorani H, Rees K, Skidmore B, Stone JA, Thompson DR, Oldridge N. Exercise-based rehabilitation for patients with coronary heart disease: systematic review and meta-analysis of randomized controlled trials. Am J Med 2004: 116: 682–692. https://www.ncbi.nlm.nih.gov/pubmed/15121495[↩][↩]
- Braunwald E, Libby P. Braunwald’s heart disease: a textbook of cardiovascular medicine. Philadelpia: Saunders Elsevier, 2008.[↩][↩]
- Gianuzzi P, Tavazzi L. Working Group Report. Recommendations for exercise training in chronic heart failure patients. Eur Heart J. 2001: 22: 125–135.[↩][↩]
- Sullivan MJ, Green HJ, Cobb FR. Skeletal muscle biochemistry and histology in ambulatory patients with long-term heart failure. Circulation 1990: 81: 518–527. https://www.ncbi.nlm.nih.gov/pubmed/2297859[↩]
- Harrington D, Anker SD, Chua TP, Webb-Peploe KM, Ponikowski PP, Poole-Wilson PA, Coats AJ. Skeletal muscle function and its relation to exercise tolerance in chronic heart failure. J Am Coll Cardiol 1997: 30: 1758–1764. https://www.ncbi.nlm.nih.gov/pubmed/9385904[↩]
- TASC. Management of peripheral arterial disease. Eur J Vasc Endovasc Surg 2000: 19: S1–S250.[↩]
- Lane R, Ellis B, Watson L, Leng GC. Exercise for intermittent claudication. Cochrane Database Syst Rev 2014: 7: CD000990. https://www.ncbi.nlm.nih.gov/pubmed/25037027[↩]
- Creasy TS, McMillan PJ, Fletcher EW, Collin J, Morris PJ. Is percutaneous transluminal angioplasty better than exercise for claudication? Preliminary results from a prospective randomised trial. Eur J Vasc Surg 1990: 4: 135–140. https://www.ncbi.nlm.nih.gov/pubmed/2140987[↩]
- Chong PF, Golledge J, Greenhalgh RM, Davies AH. Exercise therapy or angioplasty? A summation analysis. Eur J Vasc Endovasc Surg 2000: 20: 4–12. https://www.ncbi.nlm.nih.gov/pubmed/10906290[↩]
- Lundgren F, Dahllof AG, Schersten T, Bylund-Fellenius AC. Muscle enzyme adaptation in patients with peripheral arterial insufficiency: spontaneous adaptation, effect of different treatments and consequences on walking performance. Clin Sci (Lond) 1989: 77: 485–493. https://www.ncbi.nlm.nih.gov/pubmed/2555105[↩]
- Mannarino E, Pasqualini L, Innocente S, Scricciolo V, Rignanese A, Ciuffetti G. Physical training and antiplatelet treatment in stage II peripheral arterial occlusive disease: alone or combined? Angiology 1991: 42: 513–521. https://www.ncbi.nlm.nih.gov/pubmed/1863010[↩]
- Vries JA, Wong JB, Donaldson MC, Hunink MG. Intermittent claudication: cost-effectiveness of revascularization versus exercise therapy. Radiology 2002: 222: 25–36. https://www.ncbi.nlm.nih.gov/pubmed/11756701[↩]
- Regensteiner JG, Ware JE Jr, McCarthy WJ, Zhang P, Forbes WP, Heckman J, Hiatt WR. Effect of cilostazol on treadmill walking, community-based walking ability, and health-related quality of life in patients with intermittent claudication due to peripheral arterial disease: meta-analysis of six randomized controlled trials. J Am Geriatr Soc 2002: 50: 1939–1946. https://www.ncbi.nlm.nih.gov/pubmed/12473004[↩]
- Gommans LN, Fokkenrood HJ, van Dalen HC, Scheltinga MR, Teijink JA, Peters RJ. Safety of supervised exercise therapy in patients with intermittent claudication. J Vasc Surg 2015: 61: 512–518. https://www.ncbi.nlm.nih.gov/pubmed/25441008[↩]
- Rugbjerg M, Iepsen UW, Jorgensen KJ, Lange P. Effectiveness of pulmonary rehabilitation in COPD with mild symptoms: a systematic review with meta-analyses. Int J Chron Obstruct Pulmon Dis 2015: 2015(10): 791–801: doi:10.2147/COPD.S78607.[↩]
- Hamilton AL, Killian KJ, Summers E, Jones NL. Muscle strength, symptom intensity, and exercise capacity in patients with cardiorespiratory disorders. Am J Respir Crit Care Med 1995: 152: 2021–2031. https://www.ncbi.nlm.nih.gov/pubmed/8520771[↩]
- Kongsgaard M, Backer V, Jorgensen K, Kjaer M, Beyer N. Heavy resistance training increases muscle size, strength and physical function in elderly male COPD-patients – a pilot study. Respir Med 2004: 98: 1000–1007. https://www.ncbi.nlm.nih.gov/pubmed/15481277[↩]
- McCarthy B, Casey D, Devane D, Murphy K, Murphy E, Lacasse Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2015: 2: CD003793. https://www.ncbi.nlm.nih.gov/pubmed/25705944[↩]
- Griffiths TL, Phillips CJ, Davies S, Burr ML, Campbell IA. Cost effectiveness of an outpatient multidisciplinary pulmonary rehabilitation programme. Thorax 2001: 56: 779–784. https://www.ncbi.nlm.nih.gov/pubmed/11562517[↩]
- Normandin EA, McCusker C, Connors M, Vale F, Gerardi D, ZuWallack RL. An evaluation of two approaches to exercise conditioning in pulmonary rehabilitation. Chest 2002: 121: 1085–1091. https://www.ncbi.nlm.nih.gov/pubmed/11948036[↩]
- Hawkins P, Johnson LC, Nikoletou D, Hamnegard CH, Sherwood R, Polkey MI, Moxham J. Proportional assist ventilation as an aid to exercise training in severe chronic obstructive pulmonary disease. Thorax 2002: 57: 853–859. https://www.ncbi.nlm.nih.gov/pubmed/12324670[↩]
- Wadell K, Henriksson-Larsen K, Lundgren R. Physical training with and without oxygen in patients with chronic obstructive pulmonary disease and exercise-induced hypoxaemia. J Rehabil Med 2001: 33: 200–205. https://www.ncbi.nlm.nih.gov/pubmed/11585150[↩]
- American Thoracic Society. Pulmonary rehabilitation – 1999. Official statement of the American Thoracic Society, November 1998. Am J Respir Crit Care Med 1999: 1999(159): 1666–1682.[↩]
- Scherer TA, Spengler CM, Owassapian D, Imhof E, Boutellier U. Respiratory muscle endurance training in chronic obstructive pulmonary disease: impact on exercise capacity, dyspnea, and quality of life. Am J Respir Crit Care Med 2000: 162: 1709–1714. https://www.ncbi.nlm.nih.gov/pubmed/11069801[↩]
- Marquis K, Debigare R, Lacasse Y, LeBlanc P, Jobin J, Carrier G, Maltais F. Midthigh muscle cross-sectional area is a better predictor of mortality than body mass index in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2002: 166: 809–813. https://www.ncbi.nlm.nih.gov/pubmed/12231489[↩]
- Iepsen UW, Jorgensen KJ, Ringbaek T, Hansen H, Skrubbeltrang C, Lange P. A systematic review of resistance training versus endurance training in COPD. J Cardiopulm Rehabil Prev 2015b: 35: 163–172.[↩]
- Iepsen UW, Jorgensen KJ, Ringbaek T, Hansen H, Skrubbeltrang C, Lange P. A combination of resistance and endurance training increases leg muscle strength in COPD: an evidence-based recommendation based on systematic review with meta-analyses. Chron Respir Dis 2015a: 12: 132–145.[↩]
- National Institutes of Health. Global initiative for asthma. National Heart, Lung, and Blood Institute Publication No. 95-3659. Bethesda, MD: NHLBI, 1995: 6.[↩]
- Carlsen KH, Carlsen KC. Exercise-induced asthma. Paediatr Respir Rev 2002: 3: 154. https://www.ncbi.nlm.nih.gov/pubmed/12297065[↩]
- Orenstein DM. Asthma and sports. In: Bar-Or O, ed. The child and the adolescent athlete. London: Blackwell, 1996: 433–454.[↩]
- Malkia E, Impivaara O. Intensity of physical activity and respiratory function in subjects with and without bronchial asthma. Scand J Med Sci Sports 1998: 8: 27–32. https://www.ncbi.nlm.nih.gov/pubmed/9502307[↩]
- Santuz P, Baraldi E, Filippone M, Zacchello F. Exercise performance in children with asthma: is it different from that of healthy controls? Eur Respir J 1997: 10: 1254–1260. https://www.ncbi.nlm.nih.gov/pubmed/9192925[↩]
- Carson KV, Chandratilleke MG, Picot J, Brinn MP, Esterman AJ, Smith BJ. Physical training for asthma. Cochrane Database Syst Rev 2013: 9: CD001116. https://www.ncbi.nlm.nih.gov/pubmed/24085631[↩]
- Beggs S, Foong YC, Le HC, Noor D, Wood-Baker R, Walters JA. Swimming training for asthma in children and adolescents aged 18 years and under. Cochrane Database Syst Rev 2013: 4: CD009607. https://www.ncbi.nlm.nih.gov/pubmed/23633375[↩]
- Emtner M, Herala M, Stalenheim G. High-intensity physical training in adults with asthma. A 10-week rehabilitation program. Chest 1996: 109: 323–330. https://www.ncbi.nlm.nih.gov/pubmed/8620700[↩][↩]
- Turner S, Eastwood P, Cook A, Jenkins S. Improvements in symptoms and quality of life following exercise training in older adults with moderate/severe persistent asthma. Respiration 2011: 81: 302–310. https://www.ncbi.nlm.nih.gov/pubmed/20501982[↩]
- Varlotta L. Management and care of the newly diagnosed patient with cystic fibrosis. Curr Opin Pulm Med 1998: 4: 311–318. https://www.ncbi.nlm.nih.gov/pubmed/10813208[↩]
- Davis PB, Drumm M, Konstan MW. Cystic fibrosis. Am J Respir Crit Care Med 1996: 154: 1229–1256. https://www.ncbi.nlm.nih.gov/pubmed/8912731[↩]
- Bradley J, Moran F. Physical training for cystic fibrosis. Cochrane Database Syst Rev 2002: 1–27.[↩][↩]
- Moorcroft AJ, Dodd ME, Webb AK. Exercise testing and prognosis in adult cystic fibrosis. Thorax 1997: 52: 291–293. https://www.ncbi.nlm.nih.gov/pubmed/9093351[↩]
- Hulzebos E, Dadema T, Takken T. Measurement of physical activity in patients with cystic fibrosis: a systematic review. Expert Rev Respir Med 2013: 7: 647–653. https://www.ncbi.nlm.nih.gov/pubmed/24224507[↩]
- Orenstein DM, Hovell MF, Mulvihill M, Keating KK, Hofstetter CR, Kelsey S, Morris K, Nixon PA. Strength vs aerobic training in children with cystic fibrosis: a randomized controlled trial. Chest 2004: 126: 1204–1214. https://www.ncbi.nlm.nih.gov/pubmed/15486384[↩]
- van Doorn N. Exercise programs for children with cystic fibrosis: a systematic review of randomized controlled trials. Disabil Rehabil 2010: 32: 41–49. https://www.ncbi.nlm.nih.gov/pubmed/19925275[↩]
- Blumenthal JA, Babyak MA, Moore KA, Craighead WE, Herman S, Khatri P, Waugh R, Napolitano MA, Forman LM, Appelbaum M, Doraiswamy PM, Krishnan KR. Effects of exercise training on older patients with major depression. Arch Intern Med 1999: 159: 2349–2356. https://www.ncbi.nlm.nih.gov/pubmed/10547175[↩][↩]
- Babyak M, Blumenthal JA, Herman S, Khatri P, Doraiswamy M, Moore K, Craighead WE, Baldewicz TT, Krishnan KR. Exercise treatment for major depression: maintenance of therapeutic benefit at 10 months. Psychosom Med 2000: 62: 633–638. https://www.ncbi.nlm.nih.gov/pubmed/11020092[↩]
- Cooney GM, Dwan K, Greig CA, Lawlor DA, Rimer J, Waugh FR, McMurdo M, Mead GE. Exercise for depression. Cochrane Database Syst Rev 2013: 9: CD004366. https://www.ncbi.nlm.nih.gov/pubmed/24026850[↩]
- Daley AJ, Foster L, Long G, Palmer C, Robinson O, Walmsley H, Ward R. The effectiveness of exercise for the prevention and treatment of antenatal depression: systematic review with meta-analysis. BJOG 2015: 122: 57–62. https://www.ncbi.nlm.nih.gov/pubmed/24935560[↩]
- Tu RH, Zeng ZY, Zhong GQ, Wu WF, Lu YJ, Bo ZD, He Y, Huang WQ, Yao LM. Effects of exercise training on depression in patients with heart failure: a systematic review and meta-analysis of randomized controlled trials. Eur J Heart Fail 2014: 16: 749–757. https://www.ncbi.nlm.nih.gov/pubmed/24797230[↩]
- Bartley CA, Hay M, Bloch MH. Meta-analysis: aerobic exercise for the treatment of anxiety disorders. Prog Neuropsychopharmacol Biol Psychiatry 2013: 45: 34–39. https://www.ncbi.nlm.nih.gov/pubmed/23643675[↩]
- Wegner M, Helmich I, Machado S, Nardi AE, Arias-Carrion O, Budde H. Effects of exercise on anxiety and depression disorders: review of meta- analyses and neurobiological mechanisms. CNS Neurol Disord Drug Targets 2014: 13: 1002–1014. https://www.ncbi.nlm.nih.gov/pubmed/24923346[↩]
- Conn VS. Anxiety outcomes after physical activity interventions: meta-analysis findings. Nurs Res 2010: 59: 224–231. https://www.ncbi.nlm.nih.gov/pubmed/20410849[↩]
- Orwin A. Treatment of a situational phobia – a case for running. Br J Psychiatry 1974: 125: 95–98. https://www.ncbi.nlm.nih.gov/pubmed/4855252[↩]
- Herring MP, O’Connor PJ, Dishman RK. The effect of exercise training on anxiety symptoms among patients: a systematic review. Arch Intern Med 2010: 170: 321–331. https://www.ncbi.nlm.nih.gov/pubmed/20177034[↩]
- Rimmele U, Zellweger BC, Marti B, Seiler R, Mohiyeddini C, Ehlert U, Heinrichs M. Trained men show lower cortisol, heart rate and psychological responses to psychosocial stress compared with untrained men. Psychoneuroendocrinology 2007: 32: 627–635. https://www.ncbi.nlm.nih.gov/pubmed/17560731[↩]
- Galper DI, Trivedi MH, Barlow CE, Dunn AL, Kampert JB. Inverse association between physical inactivity and mental health in men and women. Med Sci Sports Exerc 2006: 38: 173–178. https://www.ncbi.nlm.nih.gov/pubmed/16394971[↩]
- Norris R, Carroll D, Cochrane R. The effects of physical activity and exercise training on psychological stress and well-being in an adolescent population. J Psychosom Res 1992: 36: 55–65. https://www.ncbi.nlm.nih.gov/pubmed/1538350[↩]
- Rogers MW, Probst MM, Gruber JJ, Berger R, Boone JB Jr. Differential effects of exercise training intensity on blood pressure and cardiovascular responses to stress in borderline hypertensive humans. J Hypertens 1996: 14: 1369–1375. https://www.ncbi.nlm.nih.gov/pubmed/8934367[↩]
- Norris R, Carroll D, Cochrane R. The effects of aerobic and anaerobic training on fitness, blood pressure, and psychological stress and well-being. J Psychosom Res 1990: 34: 367–375. https://www.ncbi.nlm.nih.gov/pubmed/2376838[↩]
- Wang CW, Chan CH, Ho RT, Chan JS, Ng SM, Chan CL. Managing stress and anxiety through qigong exercise in healthy adults: a systematic review and meta-analysis of randomized controlled trials. BMC Complement Altern Med 2014: 14: 8. https://www.ncbi.nlm.nih.gov/pubmed/24400778[↩]
- Rastad C, Martin C, Asenlof P. Barriers, benefits, and strategies for physical activity in patients with schizophrenia. Phys Ther 2014: 94: 1467–1479. https://www.ncbi.nlm.nih.gov/pubmed/24830718[↩]
- Firth J, Cotter J, Elliott R, French P, Yung AR. A systematic review and meta-analysis of exercise interventions in schizophrenia patients. Psychol Med 2015: 45: 1343–1361. https://www.ncbi.nlm.nih.gov/pubmed/25650668[↩]
- Vancampfort D, Probst M, Helvik SL, Catalan-Matamoros D, Lundvik-Gyllensten A, Gomez-Conesa A, Ijntema R, De HM. Systematic review of the benefits of physical therapy within a multidisciplinary care approach for people with schizophrenia. Phys Ther 2012b: 92: 11–23.[↩]
- Gorczynski P, Faulkner G. Exercise therapy for schizophrenia. Cochrane Database Syst Rev 2010: CD004412. https://www.ncbi.nlm.nih.gov/pubmed/20464730[↩]
- Fleshner M. Physical activity and stress resistance: sympathetic nervous system adaptations prevent stress-induced immunosuppression. Exerc Sport Sci Rev 2005: 33: 120–126. https://www.ncbi.nlm.nih.gov/pubmed/16006819[↩]
- Chaddock L, Erickson KI, Prakash RS, Kim JS, Voss MW, Vanpatter M, Pontifex MB, Raine LB, Konkel A, Hillman CH, Cohen NJ, Kramer AF. A neuroimaging investigation of the association between aerobic fitness, hippocampal volume, and memory performance in preadolescent children. Brain Res 2010: 1358: 172–183. https://www.ncbi.nlm.nih.gov/pubmed/20735996[↩]
- Laske C, Banschbach S, Stransky E, Bosch S, Straten G, Machann J, Fritsche A, Hipp A, Niess A, Eschweiler GW. Exercise-induced normalization of decreased BDNF serum concentration in elderly women with remitted major depression. Int J Neuropsychopharmacol 2010: 13: 595–602. https://www.ncbi.nlm.nih.gov/pubmed/20067661[↩]
- Lukoff D, Wallace CJ, Liberman RP, Burke K. A holistic program for chronic schizophrenic patients. Schizophr Bull 1986: 12: 274–282. https://www.ncbi.nlm.nih.gov/pubmed/3520804[↩]
- Shergill SS, Murray RM, McGuire PK. Auditory hallucinations: a review of psychological treatments. Schizophr Res 1998: 32: 137–150. https://www.ncbi.nlm.nih.gov/pubmed/9720119[↩]
- Wu RR, Zhao JP, Jin H, Shao P, Fang MS, Guo XF, He YQ, Liu YJ, Chen JD, Li LH. Lifestyle intervention and metformin for treatment of antipsychotic-induced weight gain: a randomized controlled trial. JAMA 2008: 299: 185–193. https://www.ncbi.nlm.nih.gov/pubmed/18182600[↩]
- Steen RG, Mull C, McClure R, Hamer RM, Lieberman JA. Brain volume in first-episode schizophrenia: systematic review and meta-analysis of magnetic resonance imaging studies. Br J Psychiatry 2006: 188: 510–518. https://www.ncbi.nlm.nih.gov/pubmed/16738340[↩]
- Pajonk FG, Wobrock T, Gruber O, Scherk H, Berner D, Kaizl I, Kierer A, Muller S, Oest M, Meyer T, Backens M, Schneider-Axmann T, Thornton AE, Honer WG, Falkai P. Hippocampal plasticity in response to exercise in schizophrenia. Arch Gen Psychiatry 2010: 67: 133–143. https://www.ncbi.nlm.nih.gov/pubmed/20124113[↩]
- Forbes D, Forbes SC, Blake CM, Thiessen EJ, Forbes S. Exercise programs for people with dementia. Cochrane Database Syst Rev 2015: 4: CD006489. https://www.ncbi.nlm.nih.gov/pubmed/25874613[↩]
- Kemoun G, Thibaud M, Roumagne N, Carette P, Albinet C, Toussaint L, Paccalin M, Dugue B. Effects of a physical training programme on cognitive function and walking efficiency in elderly persons with dementia. Dement Geriatr Cogn Disord 2010: 29: 109–114. https://www.ncbi.nlm.nih.gov/pubmed/20150731[↩]
- Steinberg M, Leoutsakos JM, Podewils LJ, Lyketsos CG. Evaluation of a home-based exercise program in the treatment of Alzheimer’s disease: the Maximizing Independence in Dementia (MIND) study. Int J Geriatr Psychiatry 2009: 24: 680–685. https://www.ncbi.nlm.nih.gov/pubmed/19089875[↩]
- Anderiesen H, Scherder EJ, Goossens RH, Sonneveld MH. A systematic review–physical activity in dementia: the influence of the nursing home environment. Appl Ergon 2014: 45: 1678–1686. https://www.ncbi.nlm.nih.gov/pubmed/24968971[↩]
- Lautenschlager NT, Cox KL, Flicker L, Foster JK, van Bockxmeer FM, Xiao J, Greenop KR, Almeida OP. Effect of physical activity on cognitive function in older adults at risk for Alzheimer disease: a randomized trial. JAMA 2008: 300: 1027–1037. https://www.ncbi.nlm.nih.gov/pubmed/18768414[↩][↩]
- Baker LD, Frank LL, Foster-Schubert K, Green PS, Wilkinson CW, McTiernan A, Plymate SR, Fishel MA, Watson GS, Cholerton BA, Duncan GE, Mehta PD, Craft S. Effects of aerobic exercise on mild cognitive impairment: a controlled trial. Arch Neurol 2010: 67: 71–79. https://www.ncbi.nlm.nih.gov/pubmed/20065132[↩][↩]
- Erickson KI, Voss MW, Prakash RS, Basak C, Szabo A, Chaddock L, Kim JS, Heo S, Alves H, White SM, Wojcicki TR, Mailey E, Vieira VJ, Martin SA, Pence BD, Woods JA, McAuley E, Kramer AF. Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci USA 2011: 108: 3017–3022. https://www.ncbi.nlm.nih.gov/pubmed/21282661[↩][↩]
- de Lau LM, Breteler MM. Epidemiology of Parkinson’s disease. Lancet Neurol 2006: 5: 525–535. https://www.ncbi.nlm.nih.gov/pubmed/16713924[↩]
- Konerth M, Childers J. Exercise: a possible adjunct therapy to alleviate early Parkinson disease. JAAPA 2013: 26: 30–33. https://www.ncbi.nlm.nih.gov/pubmed/23610834[↩]
- Mehrholz J, Friis R, Kugler J, Twork S, Storch A, Pohl M. Treadmill training for patients with Parkinson’s disease. Cochrane Database Syst Rev 2010: 20: CD007830.[↩]
- Goodwin VA, Richards SH, Taylor RS, Taylor AH, Campbell JL. The effectiveness of exercise interventions for people with Parkinson’s disease: a systematic review and meta-analysis. Mov Disord 2008: 23: 631–640. https://www.ncbi.nlm.nih.gov/pubmed/18181210[↩]
- de Goede CJ, Keus SH, Kwakkel G, Wagenaar RC. The effects of physical therapy in Parkinson’s disease: a research synthesis. Arch Phys Med Rehabil 2001: 82: 509–515. https://www.ncbi.nlm.nih.gov/pubmed/11295012[↩]
- Miyai I, Fujimoto Y, Ueda Y, Yamamoto H, Nozaki S, Saito T, Kang J. Treadmill training with body weight support: its effect on Parkinson’s disease. Arch Phys Med Rehabil 2000: 81: 849–852. https://www.ncbi.nlm.nih.gov/pubmed/10895994[↩]
- Miyai I, Fujimoto Y, Yamamoto H, Ueda Y, Saito T, Nozaki S, Kang J. Long-term effect of body weight-supported treadmill training in Parkinson’s disease: a randomized controlled trial. Arch Phys Med Rehabil 2002: 83: 1370–1373. https://www.ncbi.nlm.nih.gov/pubmed/12370870[↩]
- Yang YR, Lee YY, Cheng SJ, Wang RY. Downhill walking training in individuals with Parkinson’s disease: a randomized controlled trial. Am J Phys Med Rehabil 2010: 89: 706–714. https://www.ncbi.nlm.nih.gov/pubmed/20581650[↩]
- Dereli EE, Yaliman A. Comparison of the effects of a physiotherapist-supervised exercise programme and a self-supervised exercise programme on quality of life in patients with Parkinson’s disease. Clin Rehabil 2010: 24: 352–362. https://www.ncbi.nlm.nih.gov/pubmed/20360152[↩]
- Combs SA, Diehl MD, Chrzastowski C, Didrick N, McCoin B, Mox N, Staples WH, Wayman J. Community-based group exercise for persons with Parkinson disease: a randomized controlled trial. NeuroRehabilitation 2013: 32: 117–124. https://www.ncbi.nlm.nih.gov/pubmed/23422464[↩]
- Schenkman M, Hall DA, Baron AE, Schwartz RS, Mettler P, Kohrt WM. Exercise for people in early- or mid-stage Parkinson disease: a 16-month randomized controlled trial. Phys Ther 2012: 92: 1395–1410. https://www.ncbi.nlm.nih.gov/pubmed/22822237[↩]
- Ellis T, Latham NK, DeAngelis TR, Thomas CA, Saint-Hilaire M, Bickmore TW. Feasibility of a virtual exercise coach to promote walking in community-dwelling persons with Parkinson disease. Am J Phys Med Rehabil 2013: 92: 472–481. https://www.ncbi.nlm.nih.gov/pubmed/23552335[↩]
- Canadian Society for Exercise Physiology. Canadian Physical Activity Guidelines for Parkinson’s Disease. http://www.csep.ca/CMFiles/Guidelines/specialpops/PSC_Physical_Activity_resource_and_chart_final%20English%20march2012.pdf[↩]
- Latimer-Cheung AE, Pilutti LA, Hicks AL, Martin Ginis KA, Fenuta AM, MacKibbon KA, Motl RW. Effects of exercise training on fitness, mobility, fatigue, and health-related quality of life among adults with multiple sclerosis: a systematic review to inform guideline development. Arch Phys Med Rehabil 2013: 94: 1800–1828. https://www.ncbi.nlm.nih.gov/pubmed/23669008[↩]
- Amatya B, Khan F, La Mantia L, Demetrios M, Wade DT. Non pharmacological interventions for spasticity in multiple sclerosis. Cochrane Database Syst Rev 2013: 2: CD009974. https://www.ncbi.nlm.nih.gov/pubmed/23450612[↩]
- Kjolhede T, Vissing K, Dalgas U. Multiple sclerosis and progressive resistance training: a systematic review. Mult Scler 2012: 18: 1215–1228. https://www.ncbi.nlm.nih.gov/pubmed/22760230[↩][↩]
- Snook EM, Motl RW. Effect of exercise training on walking mobility in multiple sclerosis: a meta-analysis. Neurorehabil Neural Repair 2009: 23: 108–116. https://www.ncbi.nlm.nih.gov/pubmed/18948413[↩]
- Motl RW, Gosney JL. Effect of exercise training on quality of life in multiple sclerosis: a meta-analysis. Mult Scler 2008: 14: 129–135. https://www.ncbi.nlm.nih.gov/pubmed/17881388[↩]
- Khan F, Turner-Stokes L, Ng L, Kilpatrick T. Multidisciplinary rehabilitation for adults with multiple sclerosis. Cochrane Database Syst Rev 2007: CD006036. https://www.ncbi.nlm.nih.gov/pubmed/17443610[↩]
- Rietberg MB, Brooks D, Uitdehaag BM, Kwakkel G. Exercise therapy for multiple sclerosis. Cochrane Database Syst Rev 2005: CD003980. https://www.ncbi.nlm.nih.gov/pubmed/15674920[↩]
- Dalgas U, Stenager E, Jakobsen J, Petersen T, Hansen HJ, Knudsen C, Overgaard K, Ingemann-Hansen T. Resistance training improves muscle strength and functional capacity in multiple sclerosis. Neurology 2009: 73: 1478–1484. https://www.ncbi.nlm.nih.gov/pubmed/19884575[↩]
- Dalgas U, Stenager E, Jakobsen J, Petersen T, Hansen HJ, Knudsen C, Overgaard K, Ingemann-Hansen T. Fatigue, mood and quality of life improve in MS patients after progressive resistance training. Mult Scler 2010: 16: 480–490. https://www.ncbi.nlm.nih.gov/pubmed/20194584[↩]
- Petajan JH, Gappmaier E, White AT, Spencer MK, Mino L, Hicks RW. Impact of aerobic training on fitness and quality of life in multiple sclerosis. Ann Neurol 1996: 39: 432–441. https://www.ncbi.nlm.nih.gov/pubmed/8619521[↩]
- Rodgers MM, Mulcare JA, King DL, Mathews T, Gupta SC, Glaser RM. Gait characteristics of individuals with multiple sclerosis before and after a 6-month aerobic training program. J Rehabil Res Dev 1999: 36: 183–188. https://www.ncbi.nlm.nih.gov/pubmed/10659801[↩]
- Gosselink R, Kovacs L, Ketelaer P, Carton H, Decramer M. Respiratory muscle weakness and respiratory muscle training in severely disabled multiple sclerosis patients. Arch Phys Med Rehabil 2000: 81: 747–751. https://www.ncbi.nlm.nih.gov/pubmed/10857518[↩]
- Baker NA, Tickle-Degnen L. The effectiveness of physical, psychological, and functional interventions in treating clients with multiple sclerosis: a meta-analysis. Am J Occup Ther 2001: 55: 324–331. https://www.ncbi.nlm.nih.gov/pubmed/11723974[↩]
- Ensari I, Motl RW, Pilutti LA. Exercise training improves depressive symptoms in people with multiple sclerosis: results of a meta-analysis. J Psychosom Res 2014: 76: 465–471. https://www.ncbi.nlm.nih.gov/pubmed/24840141[↩]
- Canadian Society for Exercise Physiology. Canadian Physical Activity Guidelines FOR ADULTS WITH MULTIPLE SCLEROSIS. http://www.csep.ca/CMFiles/Guidelines/specialpops/CSEP_MS_PAGuidelines_adults_en.pdf[↩]
- Lauby-Secretan B, Scoccianti C, Loomis D, et al. Body Fatness and Cancer–Viewpoint of the IARC Working Group. New England Journal of Medicine 2016; 375(8):794-798. doi: 10.1056/NEJMsr1606602[↩]
- World Cancer Research Fund/American Institute for Cancer Research. Food, Nutrition, Physical Activity, and the Prevention of Cancer: a Global Perspective. Washington DC: AICR, 2007. http://www.aicr.org/assets/docs/pdf/reports/Second_Expert_Report.pdf[↩]
- Kruk J, Czerniak U. Physical activity and its relation to cancer risk: updating the evidence. Asian Pacific Journal of Cancer Prevention 2013; 14(7):3993-4003. https://www.ncbi.nlm.nih.gov/pubmed/23991944[↩]
- Wolin KY, Yan Y, Colditz GA, Lee IM. Physical activity and colon cancer prevention: a meta-analysis. British Journal of Cancer 2009; 100(4):611-616. https://www.ncbi.nlm.nih.gov/pubmed/19209175[↩]
- Moore SC, Lee IM, Weiderpass E, et al. Association of leisure-time physical activity with risk of 26 types of cancer in 1.44 million adults. JAMA Internal Medicine 2016; 176(6):816-825. https://www.ncbi.nlm.nih.gov/pubmed/27183032[↩][↩]
- Boyle T, Keegel T, Bull F, Heyworth J, Fritschi L. Physical activity and risks of proximal and distal colon cancers: a systematic review and meta-analysis. Journal of the National Cancer Institute 2012; 104(20):1548-1561. https://www.ncbi.nlm.nih.gov/pubmed/22914790[↩]
- Robsahm TE, Aagnes B, Hjartåker A, et al. Body mass index, physical activity, and colorectal cancer by anatomical subsites: a systematic review and meta-analysis of cohort studies. European Journal of Cancer Prevention 2013; 22(6):492-505. https://www.ncbi.nlm.nih.gov/pubmed/23591454[↩]
- Wolin KY, Yan Y, Colditz GA. Physical activity and risk of colon adenoma: a meta-analysis. British Journal of Cancer 2011; 104(5):882-885. https://www.ncbi.nlm.nih.gov/pubmed/21304525[↩]
- Slattery ML. Physical activity and colorectal cancer. Sports Medicine 2004; 34(4):239-252. www.ncbi.nlm.nih.gov/pubmed/15049716[↩]
- Wolin KY, Tuchman H. Physical activity and gastrointestinal cancer prevention. Recent Results in Cancer Research 2011; 186:73-100. https://www.ncbi.nlm.nih.gov/pubmed/21113761[↩]
- Colbert LH, Lanza E, Ballard-Barbash R, et al. Adenomatous polyp recurrence and physical activity in the Polyp Prevention Trial (United States). Cancer Causes and Control 2002; 13(5):445-453. www.ncbi.nlm.nih.gov/pubmed/12146849[↩]
- Wu Y, Zhang D, Kang S. Physical activity and risk of breast cancer: a meta-analysis of prospective studies. Breast Cancer Research and Treatment 2013; 137(3):869-882. https://www.ncbi.nlm.nih.gov/pubmed/23274845[↩]
- Eliassen AH, Hankinson SE, Rosner B, Holmes MD, Willett WC. Physical activity and risk of breast cancer among postmenopausal women. Archives of Internal Medicine 2010; 170(19):1758-1764. www.ncbi.nlm.nih.gov/pubmed/20975025[↩]
- Hildebrand JS, Gapstur SM, Campbell PT, Gaudet MM, Patel AV. Recreational physical activity and leisure-time sitting in relation to postmenopausal breast cancer risk. Cancer Epidemiology, Biomarkers & Prevention 2013; 22(10):1906-1912. https://www.ncbi.nlm.nih.gov/pubmed/24097200[↩]
- Fournier A, Dos Santos G, Guillas G, et al. Recent recreational physical activity and breast cancer risk in postmenopausal women in the E3N cohort. Cancer Epidemiology, Biomarkers & Prevention 2014; 23(9):1893-1902. https://www.ncbi.nlm.nih.gov/pubmed/25114017[↩][↩]
- Eliassen AH, Hankinson SE, Rosner B, Holmes MD, Willett WC. Physical activity and risk of breast cancer among postmenopausal women. Archives of Internal Medicine 2010; 170(19):1758-1764. https://www.ncbi.nlm.nih.gov/pubmed/20975025[↩]
- Schmid D, Behrens G, Keimling M, et al. A systematic review and meta-analysis of physical activity and endometrial cancer risk. European Journal of Epidemiology 2015; 30(5):397-412. https://www.ncbi.nlm.nih.gov/pubmed/25800123[↩][↩]
- Du M, Kraft P, Eliassen AH, et al. Physical activity and risk of endometrial adenocarcinoma in the Nurses’ Health Study. International Journal of Cancer 2014; 134(11):2707-2716. www.ncbi.nlm.nih.gov/pubmed/24213924[↩]
- Friedenreich C, Cust A, Lahmann PH, et al. Physical activity and risk of endometrial cancer: The European prospective investigation into cancer and nutrition. International Journal of Cancer 2007; 121(2):347-355. https://www.ncbi.nlm.nih.gov/pubmed/17357139[↩]
- Behrens G, Matthews CE, Moore SC, et al. The association between frequency of vigorous physical activity and hepatobiliary cancers in the NIH-AARP Diet and Health Study. European Journal of Epidemiology 2013; 28(1):55-66. www.ncbi.nlm.nih.gov/pubmed/23354983[↩]
- Behrens G, Jochem C, Keimling M, et al. The association between physical activity and gastroesophageal cancer: systematic review and meta-analysis. European Journal of Epidemiology 2014; 29(3):151-170. https://www.ncbi.nlm.nih.gov/pubmed/24705782[↩]
- Behrens G, Leitzmann MF. The association between physical activity and renal cancer: systematic review and meta-analysis. British Journal of Cancer 2013; 108(4):798-811. www.ncbi.nlm.nih.gov/pubmed/23412105[↩]
- Keimling M, Behrens G, Schmid D, Jochem C, Leitzmann MF. The association between physical activity and bladder cancer: systematic review and meta-analysis. British Journal of Cancer 2014; 110(7):1862-1870. Keimling M, Behrens G, Schmid D, Jochem C, Leitzmann MF. The association between physical activity and bladder cancer: systematic review and meta-analysis. British Journal of Cancer 2014; 110(7):1862-1870.[↩]
- Winzer BM, Whiteman DC, Reeves MM, Paratz JD. Physical activity and cancer prevention: a systematic review of clinical trials. Cancer Causes and Control 2011; 22(6):811-826. https://www.ncbi.nlm.nih.gov/pubmed/21461921[↩]
- Wertheim BC, Martinez ME, Ashbeck EL, et al. Physical activity as a determinant of fecal bile acid levels. Cancer Epidemiology, Biomarkers & Prevention 2009; 18(5):1591-1598. https://www.ncbi.nlm.nih.gov/pubmed/19383885[↩]
- Bernstein H, Bernstein C, Payne CM, Dvorakova K, Garewal H. Bile acids as carcinogens in human gastrointestinal cancers. Mutation Research 2005; 589(1):47-65. https://www.ncbi.nlm.nih.gov/pubmed/15652226[↩]
- Speck RM, Courneya KS, Masse LC, Duval S, Schmitz KH. An update of controlled physical activity trials in cancer survivors: a systematic review and meta-analysis. Journal of Cancer Survivorship 2010; 4(2):87-100. https://www.ncbi.nlm.nih.gov/pubmed/20052559[↩]
- Rock CL, Doyle C, Demark-Wahnefried W, et al. Nutrition and physical activity guidelines for cancer survivors. CA: A Cancer Journal for Clinicians 2012; 62(4):243-274. https://www.ncbi.nlm.nih.gov/pubmed/22539238[↩]
- Bradshaw PT, Ibrahim JG, Stevens J, et al. Postdiagnosis change in bodyweight and survival after breast cancer diagnosis. Epidemiology 2012; 23(2):320-327. https://www.ncbi.nlm.nih.gov/pubmed/22317813[↩]
- Fong DY, Ho JW, Hui BP, et al. Physical activity for cancer survivors: meta-analysis of randomised controlled trials. British Medical Journal 2012; 344:e70. https://www.ncbi.nlm.nih.gov/pubmed/22294757[↩][↩]
- Mishra SI, Scherer RW, Geigle PM, et al. Exercise interventions on health-related quality of life for cancer survivors. The Cochrane Database of Systematic Reviews 2012; 8:Cd007566. https://www.ncbi.nlm.nih.gov/pubmed/22895961[↩]
- Arem H, Moore SC, Park Y, et al. Physical activity and cancer-specific mortality in the NIH-AARP Diet and Health Study cohort. International Journal of Cancer 2014; 135(2):423-431. www.ncbi.nlm.nih.gov/pubmed/24311115[↩]
- Chlebowski RT. Nutrition and physical activity influence on breast cancer incidence and outcome. Breast 2013; 22 Suppl 2:S30-37. https://www.ncbi.nlm.nih.gov/pubmed/24074789[↩]
- Ibrahim EM, Al-Homaidh A. Physical activity and survival after breast cancer diagnosis: meta-analysis of published studies. Medical Oncology 2011; 28(3):753-765. https://www.ncbi.nlm.nih.gov/pubmed/20411366[↩]
- Holmes MD, Chen WY, Feskanich D, Kroenke CH, Colditz GA. Physical activity and survival after breast cancer diagnosis. JAMA 2005; 293(20):2479-2486. https://www.ncbi.nlm.nih.gov/pubmed/15914748[↩][↩]
- Holick CN, Newcomb PA, Trentham-Dietz A, et al. Physical activity and survival after diagnosis of invasive breast cancer. Cancer Epidemiology, Biomarkers & Prevention 2008; 17(2):379-386. https://www.ncbi.nlm.nih.gov/pubmed/18250341[↩]
- Ballard-Barbash R, Friedenreich CM, Courneya KS, et al. Physical activity, biomarkers, and disease outcomes in cancer survivors: a systematic review. Journal of the National Cancer Institute 2012; 104(11):815-840. https://www.ncbi.nlm.nih.gov/pubmed/22570317[↩]
- Arem H, Pfeiffer RM, Engels EA, et al. Pre- and postdiagnosis physical activity, television viewing, and mortality among patients with colorectal cancer in the National Institutes of Health-AARP Diet and Health Study. Journal of Clinical Oncology 2015; 33(2):180-188. https://www.ncbi.nlm.nih.gov/pubmed/25488967[↩]
- Kenfield SA, Stampfer MJ, Giovannucci E, Chan JM. Physical activity and survival after prostate cancer diagnosis in the health professionals follow-up study. Journal of Clinical Oncology 2011; 29(6):726-732. https://www.ncbi.nlm.nih.gov/pubmed/21205749[↩]
- Bonn SE, Sjölander A, Lagerros YT, et al. Physical activity and survival among men diagnosed with prostate cancer. Cancer Epidemiology, Biomarkers & Prevention 2015; 24(1):57-64. https://www.ncbi.nlm.nih.gov/pubmed/25527697[↩]
- Yii MF, Lim CE, Luo X, Wong WS, Cheng NC, Zhan X. Polycystic ovarian syndrome in adolescence. Gynecol Endocrinol 2009: 25: 634–639. https://www.ncbi.nlm.nih.gov/pubmed/19533479[↩]
- Harrison CL, Lombard CB, Moran LJ, Teede HJ. Exercise therapy in polycystic ovary syndrome: a systematic review. Hum Reprod Update 2011: 17: 171–183. https://www.ncbi.nlm.nih.gov/pubmed/20833639[↩]
- Thomson RL, Buckley JD, Brinkworth GD. Exercise for the treatment and management of overweight women with polycystic ovary syndrome: a review of the literature. Obes Rev 2011: 12: e202–e210. https://www.ncbi.nlm.nih.gov/pubmed/20546140[↩]
- Thomson RL, Buckley JD, Noakes M, Clifton PM, Norman RJ, Brinkworth GD. The effect of a hypocaloric diet with and without exercise training on body composition, cardiometabolic risk profile, and reproductive function in overweight and obese women with polycystic ovary syndrome. J Clin Endocrinol Metab 2008: 93: 3373–3380. https://www.ncbi.nlm.nih.gov/pubmed/18583464[↩]
- Sprung VS, Cuthbertson DJ, Pugh CJ, Aziz N, Kemp GJ, Daousi C, Green DJ, Cable NT, Jones H. Exercise training in polycystic ovarian syndrome enhances flow-mediated dilation in the absence of changes in fatness. Med Sci Sports Exerc 2013: 45: 2234–2242. https://www.ncbi.nlm.nih.gov/pubmed/24240117[↩]
- Centers for Disease Control and Prevention. – Healthy Pregnant or Postpartum Women – https://www.cdc.gov/physicalactivity/basics/pregnancy/index.htm[↩][↩][↩][↩]
- American Congress of Obstetricians and Gynecologists. Physical Activity and Exercise During Pregnancy and the Postpartum Period. https://www.acog.org/Resources-And-Publications/Committee-Opinions/Committee-on-Obstetric-Practice/Physical-Activity-and-Exercise-During-Pregnancy-and-the-Postpartum-Period[↩][↩][↩][↩]
- American Congress of Obstetricians and Gynecologists. Exercise During Pregnancy. https://www.acog.org/Patients/FAQs/Exercise-During-Pregnancy[↩][↩]
- Centers for Disease Control and Prevention. – How much physical activity do children need ? – https://www.cdc.gov/physicalactivity/basics/children/index.htm[↩]
- Centers for Disease Control and Prevention. – Measuring Physical Activity Intensity – https://www.cdc.gov/physicalactivity/basics/measuring/index.html[↩][↩]
- Centers for Disease Control and Prevention. – Target Heart Rate and Estimated Maximum Heart Rate – https://www.cdc.gov/physicalactivity/basics/measuring/heartrate.htm[↩]