Contents
What is histamine
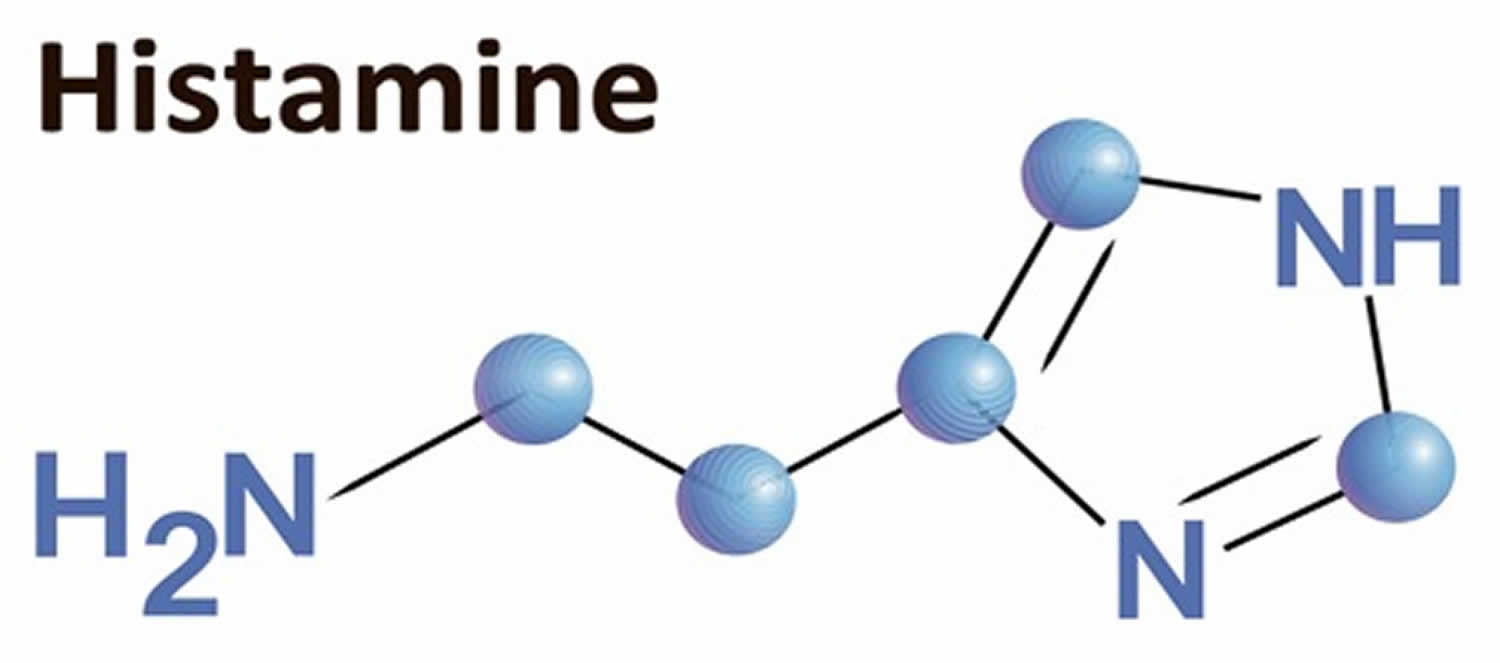
Histamine is a biogenic amine (2-[4-imidazolyl]ethylamine) and is synthesized by the pyridoxal phosphate (vitamin B-6)–containing L-histidine decarboxylase from the amino acid histidine 1. Histamine is synthesized by mast cells, basophils, platelets, histaminergic neurons, and enterochromaffine cells, where it is stored intracellularly in vesicles and released on stimulation 1. Histamine is a potent mediator of numerous biologic reactions. Besides the well-known triggering of degranulation of mast cells by crosslinking of the FcεRI receptor by specific allergens, several other nonimmunologic stimuli, such as neuropeptides, complement factors (i.e, C3a and C5a), cytokines, hyperosmolarity, lipoproteins, adenosine, superoxidases 2, hypoxia, chemical and physical factors (e.g, extreme temperatures, traumas) 3, or alcohol and certain food and drugs, may activate mast cells.
Due to microbial contamination, food and beverages sometimes contain varying amounts of histamine in relevant amounts. Therefore, spoiled or fermented foods can contain high levels of histamine. In particular, food that undergoes microbial ripening, such as cheese, salami, sauerkraut, or red wine, can contain high levels of histamine (see Tables 3 and 4 below) 1. Histamine intolerance belongs to the group of non-IgE-mediated hypersensitivity-like reactions, and is known as a pharmacological food intolerance. Currently, no valid in vitro tests can prove histamine intolerance; thus, a double-blind, placebo-controlled food challenge test remains the gold standard for diagnostic workup of non-IgE-mediated food intolerance 4.
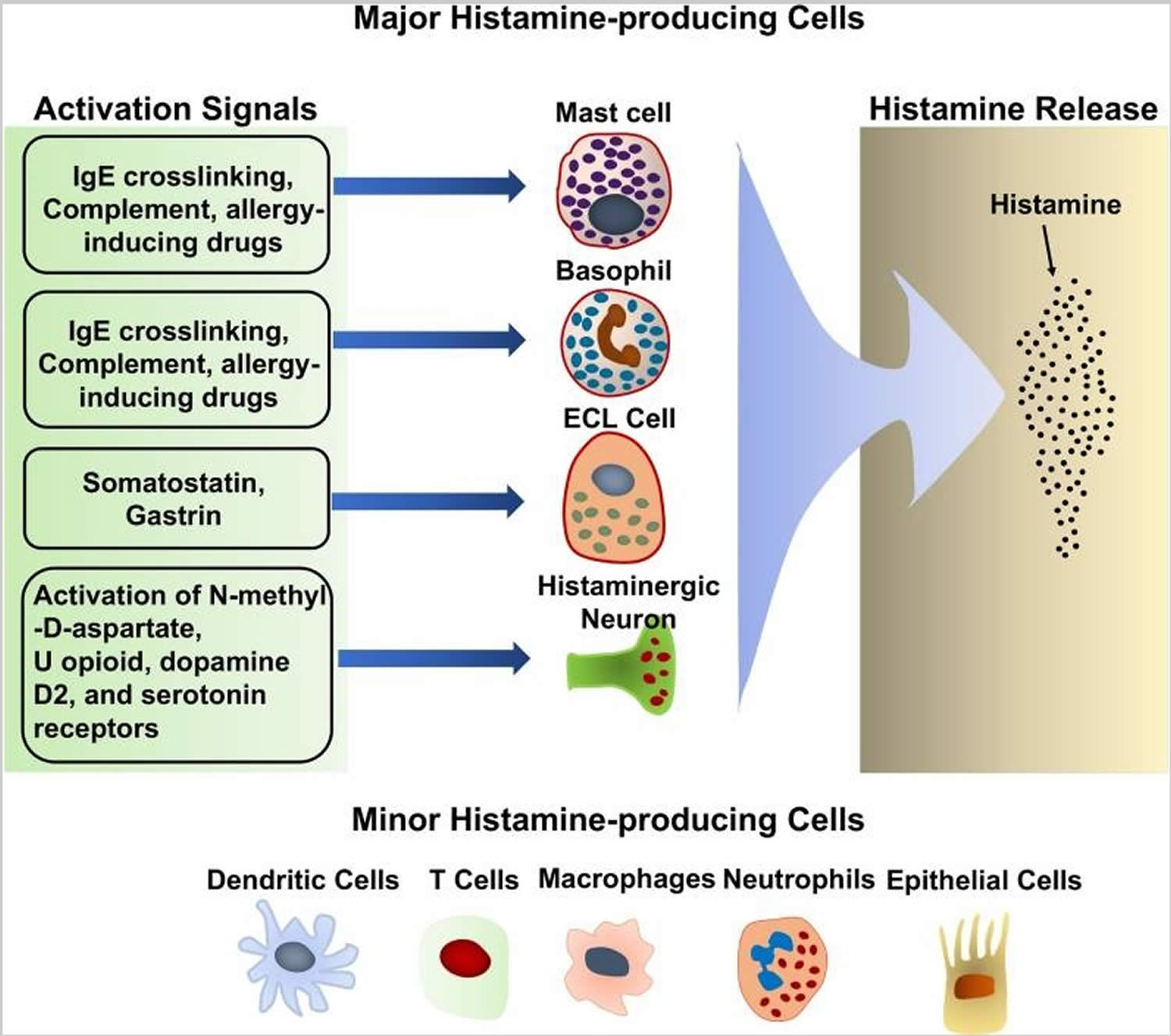
Histamine producing cells and stimuli that trigger histamine release
Histamine is synthesized primarily by mast cells, basophils, histaminergic neurons in the basal ganglia of the brain and enterochromaffin-like cells in the stomach 5. These cells produce large amounts of histamine and are thought to be the major histamine-producing cells (Figure 1). They continuously synthesize histamine, which is then linked to the carboxyl group of heparin and stored in intracellular granules until the cells receive the appropriate activating stimulus. Upon external stimulation, these cells degranulate, releasing the stored histamine. Stimuli that trigger histamine release by these major histamine-producing cells have been reviewed extensively 6. Antigen crosslinking of antigen-specific IgE bound to the high-affinity IgE receptor, FcεRI, on the mast cell and basophil surface is the most robust stimulus that triggers histamine release by these cells 6. Substance P and allergy-inducing drugs that bind to G-protein-coupled receptors can also trigger basophils and mast cells to release histamine via different signaling pathway 7. In addition, complement components, such as the C3a and C5a “anaphylatoxins,” have also been shown to induce histamine release by mast cells 8. Many cytokines, including IL-3, IL-18, IL-33, GM-CSF, and SCF, promote histamine synthesis 9. In general, cytokines alone do not induce histamine release although it remains controversial whether IL-33 can have this effect. Some reports describe that IL-33 stimulates histamine release 10, while other reports dispute this 11. It is suggested that IL-33 alone does not induce histamine release by basophils, but enhances histamine release in response to IgE/FcεRI crosslinking 12.
Additional histamine-producing cells have also been identified, including T cells 13, dendritic cells 14, macrophages 15, and epithelial cells 16 (Figure 1). In contrast to mast cells and basophils, these cells produce relative small quantities of histamine and do not store it in their cytoplasm 17. The small amounts of histamine that they produced are released without external stimulation 17. The biological significance of the small amounts of histamine produced by these minor histamine-producing cells remains unclear. Cell type-specific deletion of the Hdc gene, which encodes HDC (histidine decarboxylase), an enzyme essential for histamine synthesis, would shed light on the role of histamine synthesis and secretion by the minor histamine-producing cells.
Figure 1. Histamine-producing cells and stimuli that trigger histamine release
[Source 5]What does histamine do?
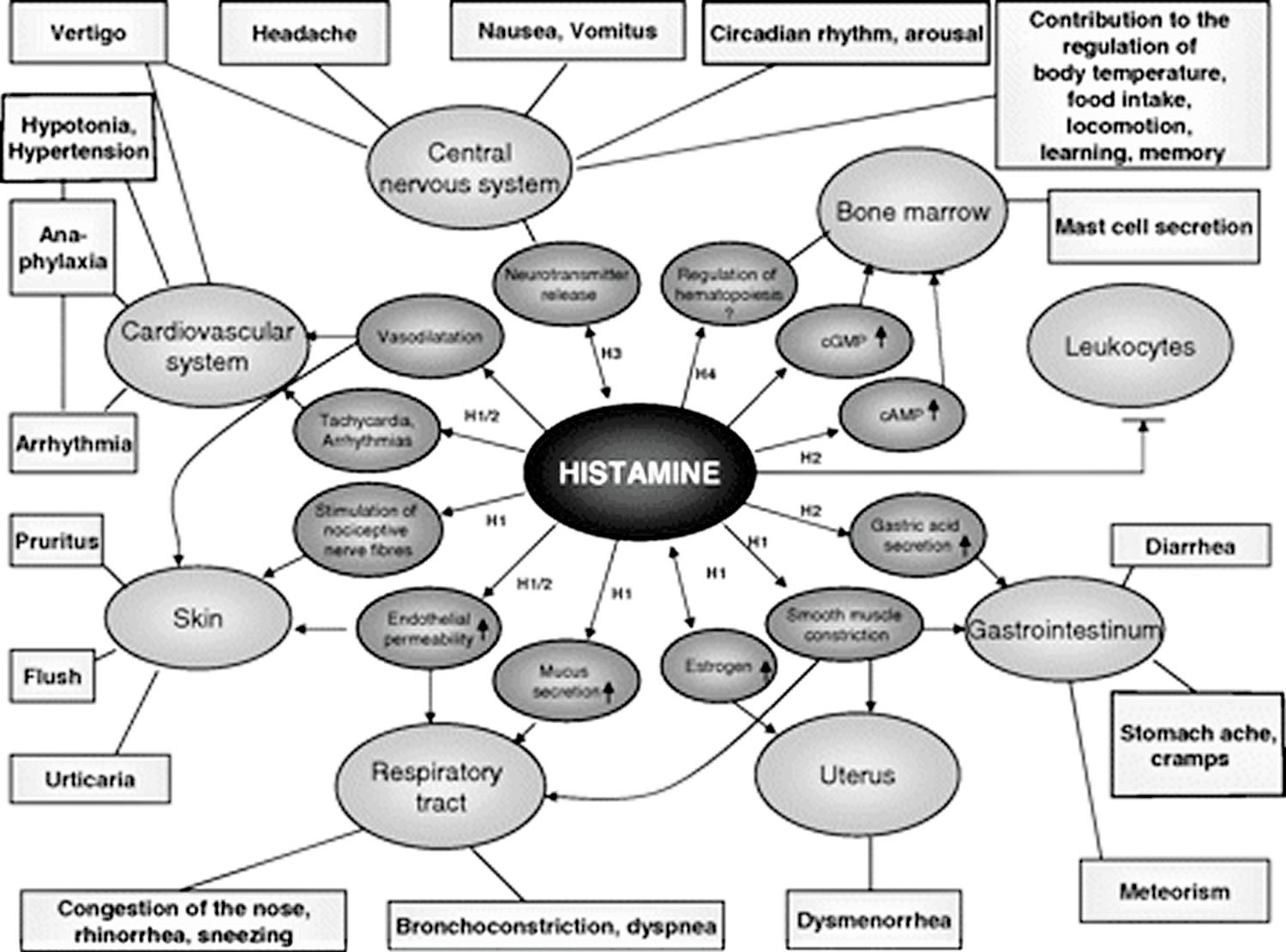
Histamine exerts its effects by binding to its 4 receptors [histamine 1 receptor (H1R), H2R, H3R, and and H4R] on target cells in various tissues (see Figure 2 and Table 1). Histamine causes smooth muscle cell contraction, vasodilatation (blood vessels to dilate), increased vascular permeability and mucus secretion, tachycardia (increase heart rate), alterations of blood pressure, and arrhythmias (abnormal heart rates or rhythms), and histamine stimulates gastric acid secretion and nociceptive nerve fibers. In addition, histamine has been known to play various roles in neurotransmission, immunomodulation, hematopoiesis (red blood cell formation), wound healing, day-night rhythm, and the regulation of histamine- and polyamine-induced cell proliferation and angiogenesis in tumor models 18 and intestinal ischemia 19.
Figure 2. Histamine effects
[Source 1]Table 1. Histamine effects according to plasma histamine concentration (ng/mL or nanogram per mililiter)
| Histamine | Clinical effect |
|---|---|
| 0–1 | Reference |
| 1–2 | ↑ Gastric acid secretion ↑ Heart rate |
| 3–5 | Tachycardia, headache, flush, urticaria, pruritus |
| 6–8 | ↓ Arterial pressure |
| 7–12 | Bronchospasm |
| ≈100 | Cardiac arrest |
Histamine metabolism
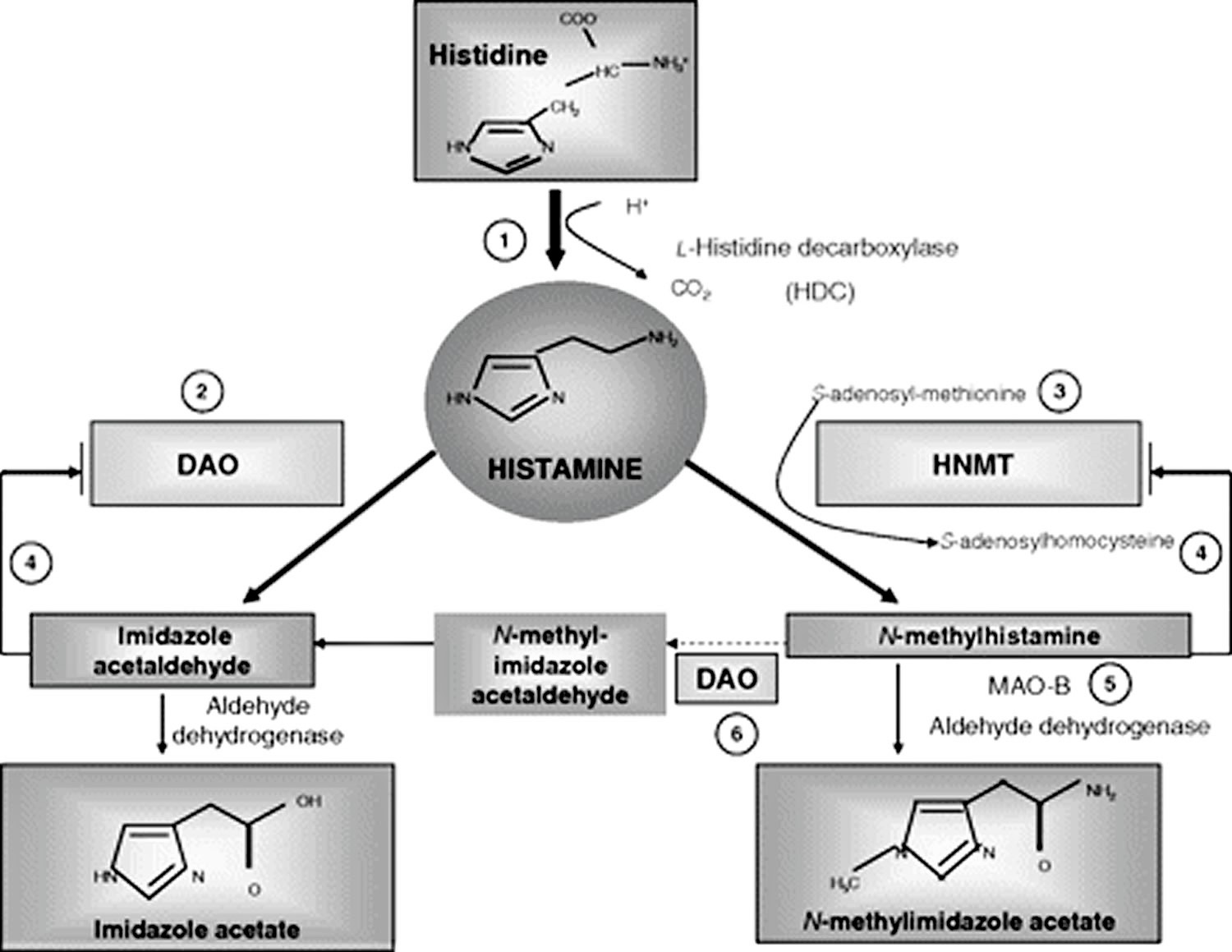
Histamine can be metabolized in 2 ways (Figure 2 and Table 2):
- By oxidative deamination by diamine oxidase (DAO) (former name: histaminase) or
- By ring methylation by histamine-N-methyltransferase 20.
Whether histamine is catabolized by diamine oxidase (DAO) or histamine-N-methyltransferase is supposed to depend on the localization of histamine. The diamine oxidase (DAO) protein is stored in plasma membrane–associated vesicular structures in epithelial cells and is secreted into the circulation on stimulation 21. Therefore, it has been proposed that diamine oxidase (DAO) may be responsible for scavenging extracellular histamine (e.g, after ingestion of histamine-rich food) after mediator release. Conversely, histamine-N-methyltransferase, the second most important enzyme inactivating histamine, is a cytosolic protein 22, which can convert histamine only in the intracellular space of cells 23. Thus, the enzymes do not seem to compete for the substrate, although they have a similar affinity for histamine and they are expressed in some overlapping tissues. Histamine-N-methyltransferase has a slightly higher affinity for histamine [Michaelis-Menten constant (kM): 6–13 μmol/L] than does diamine oxidase (DAO) (kM: 20 μmol/L). In mammals, diamine oxidase (DAO) expression is restricted to specific tissues; the highest activities are shown for small bowel and colon 24 and for placenta and kidney 23. Lower diamine oxidase (DAO) activity has been discussed as a potential indicator of intestinal mucosa damage in inflammatory and neoplastic diseases 25 and in persons undergoing chemotherapy 26. Histamine-N-methyltransferase is widely expressed in human tissues; the greatest expression is in kidney and liver, followed by spleen, colon, prostate, ovary, spinal cord cells, bronchi, and trachea27. Histamine-N-methyltransferase is regarded as the key enzyme for histamine degradation in the bronchial epithelium 28.
Figure 2. Histamine metabolism
Footnotes: Summary of the histamine metabolism. The biogenic amine histamine is synthesized by decarboxylation of the amino acid histidine catalyzed by l-histidine decarboxylase (HDC) (1). Histamine can be metabolized by extracellular oxidative deamination of the primary amino group by diamine oxidase (DAO) (2) or intracellular methylation of the imidazole ring by histamine-N-methyltransferase (HNMT) (3). Therefore, insufficient enzyme activity caused by enzyme deficiency or inhibition may lead to accumulation of histamine. Both enzymes can be inhibited by their respective reaction products in a negative feedbackloop (4). N-Methylhistamine is oxidatively deaminated to N-methyl-imidazole acetaldehyde by monoamine oxidase B (MAO B) (5) or by DAO (6). Because the methylation pathway takes place in the cytosolic compartment of cells, MAO B (5) has been suggested to catalyze this reaction in vivo 26.
[Source 1]Table 2. Characteristics of the histamine-degrading enzymes diamine oxidase (DAO) and histamine N-methyl-transferase (HNMT)
| DAO | HNMT | |
|---|---|---|
| Gene | ||
| Gene map locus | Chromosome 7q35 | Chromosome 2q22 |
| Gene | 10 kbp, 5 exons, 4 introns | 35 kbp, 6 exons |
| Associated with SNPs | Inflammatory and neoplastic gastrointestinal diseases such as food allergy, gluten-sensitive enteropathy, Crohn disease, ulcerative colitis, and colon adenoma | Asthma |
| Protein | Soluble homodimeric glycoprotein of MR 200 000 with subunits of 70–125 kDa; 750 amino acid residues | Soluble, cytosolic protein of MR 33 000 with subunits of 29–34 kDa; 292 amino acid residues |
| Enzyme | ||
| Group | Copper-containing amine oxidases | Methyltransferases |
| Active form | Homodimer with the active-site cofactor 2,4,5-trihydroxyphenylalanine quinone (Topa quinone) | Monomer with a 2-domain structure |
| Enzyme kinetics (km) | Histamine, 20 μmol/L | Histamine, 6–13 μmol/L |
| Putrescine, 350 μmol/L | S-adenosyl-l-methionine, 6–10 μmmol/L | |
| Spermidine, 3 mmol/L | ||
| Optimum pH | 7.2 | 7.5–9.0 |
| Inhibititors | Copper-chelating agents, eg cyanide Carbonylgroup reagents, eg, aminoguanidine, semibarbacide | Reaction products: N-methylhistamine, S-adenosyl-l-homocysteine |
| Sulphydryl groups: p-chloromercuriobenzoate | ||
| Major expression | Intestine, kidney, placenta | Highest: kidney and liver; considerable: spleen, colon, prostate, ovary, spinal cord cells, trachea, and bronchi; to a smaller amount, nearly ubiquitous expression |
| Storage | Plasma membrane–associated vesicular structures in epithelial cells, secretion into the circulation upon stimulation | Cytosolic compartment of the cells |
| Function | Extracellular scavenger of histamine and other diamines by oxidative deamination of the primary amino group of histamine | Intracellular histamine inactivation by methylation of the imidazole ring |
Abbreviations:
SNPs = single-nucleotide polymorphisms; kbp = kilobase pair; MR = molecular weight; kDa = kiloDalton; kM = Michaelis-Menten constant.
[Source 1]Histamine and atopic eczema
Higher basal plasma histamine concentrations 29 and increased spontaneous histamine release toward different stimuli 30 and after food challenges 31 have been shown in patients with severe atopic eczema than in control subjects. In addition, reduced diamine oxidase (DAO) activities have been shown in a subgroup of atopic eczema patients 32. Thus, these patients have a significantly greater occurrence of chronic headache, dysmenorrhea, flushing, gastrointestinal symptoms, and intolerance to alcohol and food than do control subjects. Reduction of both the symptoms of histamine intolerance and the severity score of atopic dermatitis has been shown in a subgroup of patients with atopic eczema and low diamine oxidase (DAO) serum activity who were following a histamine-free diet for 2 week 32. Orally ingested histamine has been shown to aggravate eczema in atopic eczema patients in a double-blind, placebo-controlled provocation 33. A feedback inhibition of diamine oxidase (DAO) through its degradation product imidazole acetic acid 34 or substrate inhibition 35 caused by the elevated histamine concentrations in atopic eczema may be a pathomechanism of a reduced histamine degradation capacity in a subgroup of patients with atopic eczema.
Histamine and sexual steroids
In the female genital tract, histamine is mainly produced by mast cells, endothelial cells, and epithelial cells in the uterus and ovaries. Histamine-intolerant women often suffer from headache that is dependent on their menstrual cycle and from dysmenorrhea. Besides the conctractile action of histamine, these symptoms may be explained by the interplay of histamine and hormones. Histamine has been shown to stimulate, in a dose-dependent manner, the synthesis of estradiol via H1R (histamine 1 receptor); meanwhile, only a moderate effect on progesterone synthesis was observed 36. The painful uterine contractions of primary dysmenorrhea are mainly caused by an increased mucosal production of prostaglandin F2α stimulated by estradiol and attenuated by progesterone. Thus, histamine may augment dysmenorrhea by increasing estrogen concentrations. And, in reverse, estrogen can influence histamine action. A significant increase in weal and flare size in response to histamine has been observed to correspond to ovulation and peak estrogen concentrations 37. In pregnancy, diamine oxidase (DAO) is produced at very high concentrations by the placenta 38, and its concentration may become 500 times that when the woman is not pregnant 38. This increased diamine oxidase (DAO) production in pregnant women may be the reason why, in women with food intolerance, remissions frequently occur during pregnancy 39.
Histamine and drugs
The effect of drugs as specific diamine oxidase (DAO) inhibitors and their capacity to induce histamine intolerance have been shown in various studies with human placental diamine oxidase (DAO) and in animal experiments40. A clinically relevant activity via histamine release or inhibition of DAO has been observed for various drugs 41. Therefore, the intake of drugs, especially long-term medication, should be considered in interpretation of histamine intolerance symptoms and diamine oxidase (DAO) concentrations.
Drugs releasing histamine or inhibiting diamine oxidase (DAO)
| Substance class | Agent interfering with the histamine metabolism |
|---|---|
| Contrast media | |
| Muscle relaxants | Pancuronium, alcuronium, d-tubocurarine |
| Narcotics | Thiopental |
| Analgetics | Morphine, pethidine, nonsteroidal antiinflammatory drugs, acetylsalicylic acid, metamizole |
| Local anesthetics | Prilocaine |
| Antihypotonics | Dobutamine |
| Antihypertensive drugs | Verapamil, alprenolol, dihydralazine |
| Antiarrhythmics | Propafenone |
| Diuretics | Amiloride |
| Drugs influencing gut motility | Metoclopramide |
| Antibiotics | Cefuroxime, cefotiam, isoniazid, pentamidin, clavulanic acid, choroquine |
| Mucolytics | Acetylcysteine, ambroxol |
| Broncholytics | Aminophylline |
| H2-receptor antagonists | Cimetidine |
| Cytostatics | Cyclophosphamide |
| Antidepressants | Amitriptyline |
Histamine diet
Histamine and other biogenic amines are present to various degrees in many foods, and their presence increases with maturation 42. The formation of biogenic amines in food requires the availability of free amino acids, the presence of decarboxylase-positive microorganisms, and conditions allowing bacterial growth and decarboxylase activity. Free amino acids either occur as such in foods or may be liberated by proteolysis during processing or storage 43. Numerous bacteria and some yeasts display high L-histidine decarboxylase activity and thus have the capacity to form histamine. Histidine is generated from autolytic or bacterial processes 44. Therefore, high concentrations of histamine are found mainly in products of microbial fermentation, such as aged cheese 45, sauerkraut, wine 46, and processed meat 47 (see Tables 3 and 4 below) or in microbially spoiled food. Thus, histamine, tyramine, putrescine, and cadaverine serve as indicators of hygienic food quality 43. Tyramine and putrescine also may lead to intolerance reactions in combination with histamine. Possible explanations may be the inhibition of diamine oxidase (DAO) by other amines 48 or the promotion of histamine liberation from the mucosa by putrescine 49.
Intolerance of tyramine that has vasoconstrictive properties that lead to hypertensive crisis and headache has been known mostly in patients taking monoamine oxidase (MAO)–inhibiting drugs. Orally administered tyramine in doses of 200 to 800 mg has been shown to increase systolic blood pressure by 30 mm Hg in otherwise unmedicated subjects. Conversely, in patients taking MAO-inhibiting drugs, the pressor sensitivity was 7- to 56-fold that in patients not taking MAO-inhibiting drugs 50. Eight double-blind, placebo-controlled studies have investigated the effect of tyramine on migraine. Two studies showed positive results in migraine patients who were sensitive to foods that are high in tyramine (n = 45) (19) or who had wine-provoked migraine (n = 19) 51; 6 studies showed negative results with 97 52 patients. The 2 positive studies and 2 of the negative studies were regarded as inconclusive 53 because of a lack of randomization 50, questionable blinding 51, or inappropriate selection of migraine patients without a history of suspected tyramine intolerance 54. Conversely, in 2 conclusive studies of migraine patients with a positive or negative dietary history, 125 mg oral tyramine did not precipitate more headaches than did placebo.
Histamine foods
Alcohol, especially red wine, is rich in histamine and is a potent inhibitor of diamine oxidase (DAO) 55. The relation between the ingestion of wine, an increase in plasma histamine, and the occurrence of sneezing, flushing, headache, asthma attacks, and other anaphylactoid reactions and a reduction of symptoms by antihistamines has been shown in various studies 56. However, among the multitude of substances contained in wine, other biogenic amines such as tyramine 51 and sulfites 57 have been supposed to contribute to symptoms summarized as “wine intolerance” or “red wine asthma” 57. In double-blind, placebo-controlled wine tests with healthy persons 58 and in patients with chronic urticaria and wine intolerance 59, the histamine content did not influence wine tolerance. In the latter group, an increase in plasma histamine could be shown, paradoxically, after ingestion of the histamine-poor wine. In these patients, the ethanol metabolite acetaldehyde has been discussed as a histamine-releasing substance 59. However, the high percentage of responses to the placebo (87%) could be responsible for the absence of an effect in this study 53. Another randomized double-blind, placebo-controlled oral wine challenge in patients with a history of red wine–provoked asthma (n = 18) found no relation between wine tolerance and the wine’s content of histamine or other amines but did find a greater bronchoconstrictive response to wine with a high sulfite content 57. Sulfiting agents are widely used as antioxidants and preservatives in foods, beverages, and pharmaceuticals. Adverse reactions with a presumed relation to sulfites include anaphylactic shock, bronchospasm, urticaria, angioedema, nausea, abdominal pain, diarrhea, stroke, and death 60. Sulfite hypersensitivity has been reported mainly in patients with chronic asthma; the estimated prevalence is 5–10% in all patients 61. Asthmatic reactions have been attributed to reflex activation of the parasympathetic system by the irritating effect of sulfites, possibly enhanced by a deficiency of sulfite oxidase. Besides this pseudoallergic mechanism, in at least some cases of sulfite hypersensitity, an immunoglobulin E (IgE)–mediated immediate-type allergic reaction must be considered 62. Sulfites may be contained in wine, but they are also contained in foods that are poor in histamine, such as fruit juice, frozen vegetables, and lettuce. Thus, in patients reporting intolerance to wine, a careful history of reactions to other foods rich in histamine or sulfites should be taken. In patients who are suspected of having sulfite intolerance, skin testing and a double-blind, placebo-controlled challenge with capsules containing increasing doses of bisulfite or placebo should be performed.
In contrast to an IgE–mediated food allergy, in which the ingestion of even a small amount of the allergen elicits symptoms, in histamine intolerance, the cumulative amount of histamine is crucial. Besides variations in the amount of histamine in food according to storage and maturation, the quantity consumed, the presence of other biogenic amines, and the additional intake of alcohol or diamine oxidase (DAO)-blocking drugs are pivotal factors in the tolerance of the ingested food. Generally, an upper limit of 100 mg histamine/kg in foods and of 2 mg histamine/L in alcoholic beverages has been suggested 63. This threshold may be too high, considering the occurrence of histamine-mediated symptoms after oral ingestion of 75 mg histamine in 5 of 10 females without a history of histamine intolerance 64.
However, most of the positive studies for intolerant reactions to sulfite, histamine, and other biogenic amines do not fulfill the current scientific criteria for providing substantiated evidence of the clinical effect of these foods. Nevertheless, patients who have a conclusive history of adverse reactions to food, alcohol, drugs containing histamine, other biogenic amines, and sulfite but without proof of IgE exist. In such patients, a double-blind, placebo-controlled provocation of the suspected causal agents under close supervision by experienced specialists should be performed after exclusion of other causal diseases and informed consent of the patients—if the provocation is not unreasonably hazardous, considering the grade of the anaphylactoid reaction. Because of the great effort, time, and costs or because of patients’ fear of a repeated reaction, double-blind, placebo-controlled provocations often are not performed in clinical practice, even when they are indicated.
Table 3. High histamine foods
| Food categories | Histamine | Recommended upper limit for histamine | Tyramine | |||
|---|---|---|---|---|---|---|
| mg/kg | mg/L | mg/kg | mg/L | mg/kg | mg/L | |
| Fish (frozen/smoked or salted/canned) | 200 | ND | ||||
| Mackerel | 1–20/1–1788/ND–210 | |||||
| Herring | 1–4/5–121/1–479 | |||||
| Sardine | ND/14–150/3–2000 | |||||
| Tuna | ND/ND/1–402 | |||||
| Cheese | No recommendation | |||||
| Gouda | 10–900 | 10–900 | ||||
| Camembert | 0–1000 | 0–4000 | ||||
| Cheddar | 0–2100 | 0–1500 | ||||
| Emmental | 5–2500 | 0–700 | ||||
| Swiss | 4–2500 | 0–700 | ||||
| Parmesan | 10–581 | 0–840 | ||||
| Meat | No recommendation | |||||
| Fermented sausage | ND–650 | ND–1237 | ||||
| Salami | 1–654 | – | ||||
| Fermented ham | 38–271 | 123–618 | ||||
| Vegetables | ||||||
| Sauerkraut | 0–229 | 10 | 2–951 | |||
| Spinach | 30–60 | |||||
| Eggplant | 26 | |||||
| Tomato ketchup | 22 | |||||
| Red wine vinegar | 4 | |||||
| Alcohol | ||||||
| White wine | ND–10 | 2 | 1–8 | |||
| Red wine | ND–30 | 2 | ND–25 | |||
| Top-fermented beer | ND–14 | 1.1–36.4 | ||||
| Bottom-fermented beer | ND–17 | 0.5–46.8 | ||||
| Champagne | 670 | |||||
Abbreviation: ND = not detected
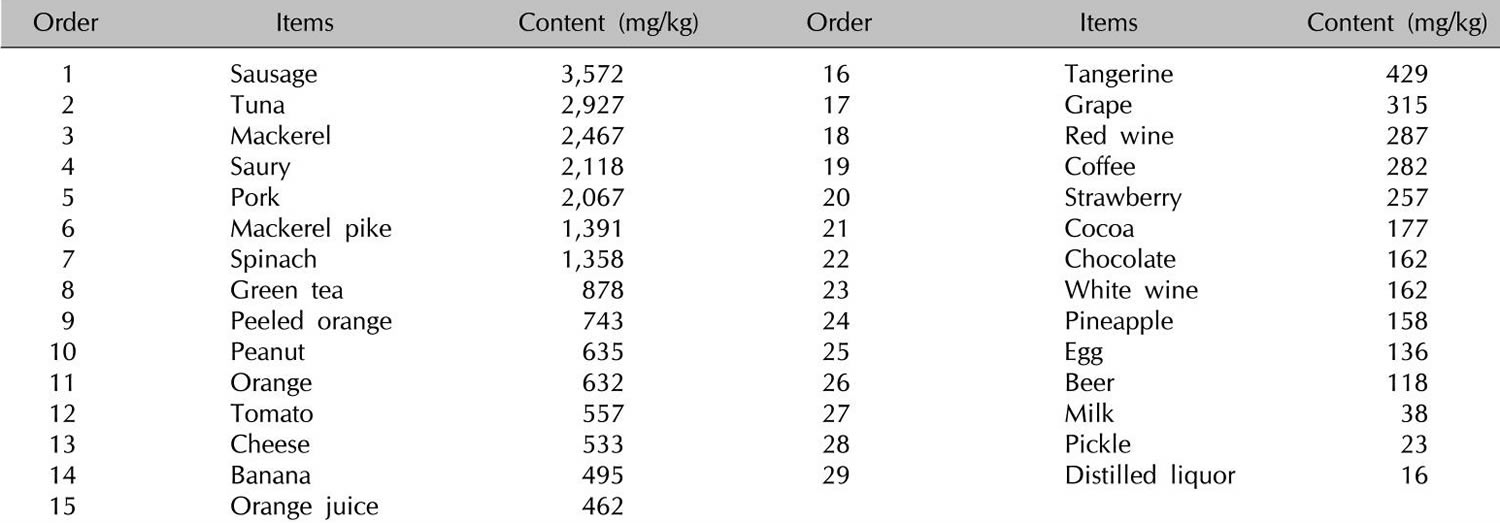
[Source 1]Table 4. Histamine food list
[Source 65]In addition to histamine-rich food, many foods such as citrus foods are considered to have the capacity to release histamine directly from tissue mast cells, even if they themselves contain only small amounts of histamine (Table 5). In vitro studies of persons with a history of pseudoallergic reactions to food have shown a fragility of duodenal mast cells with massive degranulation in the presence of histamine-releasing substances that is significantly greater than that shown by control subjects 66. However, clinical studies using oral challenge tests to support the hypothesis for the histamine-releasing capacity of foods are required 2.
Table 5. Foods with suggested histamine-releasing capacities
| Plant-derived | Animal-derived | Other |
|---|---|---|
| Citrus fruit | Fish | Additives |
| Papaya | Crustaceans | Liquorice |
| Strawberries | Pork | Spices |
| Pineapple | Egg white | |
| Nuts | ||
| Peanuts | ||
| Tomatoes | ||
| Spinach | ||
| Chocolate |
- Laura Maintz, Natalija Novak; Histamine and histamine intolerance, The American Journal of Clinical Nutrition, Volume 85, Issue 5, 1 May 2007, Pages 1185–1196, https://doi.org/10.1093/ajcn/85.5.1185[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Vlieg-Boerstra BJ, van der HS, Oude Elberink JN, Kluin-Nelemans JC, Dubois AE. Mastocytosis and adverse reactions to biogenic amines and histamine-releasing foods: what is the evidence? Neth J Med 2005;63:244–9.[↩][↩]
- Ring J. Angewandte Allergologie. (Implemented allergology.) Munich, Germany: Urban & Vogel, 2004[↩]
- Worm M, Fiedler EM, Dölle S, Schink T, Hemmer W, Jarisch R, et al. Exogenous histamine aggravates eczema in a subgroup of patients with atopic dermatitis. Acta Derm Venereol. 2009;89:52–56.[↩]
- Huang H, Li Y, Liang J, Finkelman FD. Molecular Regulation of Histamine Synthesis. Frontiers in Immunology. 2018;9:1392. doi:10.3389/fimmu.2018.01392. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6019440/[↩][↩]
- Gilfillan AM, Tkaczyk C. Integrated signalling pathways for mast-cell activation. Nat Rev Immunol (2006) 6(3):218–30.10.1038/nri1782[↩][↩]
- Cildir G, Pant H, Lopez AF, Tergaonkar V. The transcriptional program, functional heterogeneity, and clinical targeting of mast cells. J Exp Med (2017) 214(9):2491–506.10.1084/jem.20170910[↩]
- Woolhiser MR, Brockow K, Metcalfe DD. Activation of human mast cells by aggregated IgG through FcgammaRI: additive effects of C3a. Clin Immunol (2004) 110(2):172–80.10.1016/j.clim.2003.11.007[↩]
- Saluja R, Ketelaar ME, Hawro T, Church MK, Maurer M, Nawijn MC. The role of the IL-33/IL-1RL1 axis in mast cell and basophil activation in allergic disorders. Mol Immunol (2015) 63(1):80–5.10.1016/j.molimm.2014.06.018[↩]
- Silver MR, Margulis A, Wood N, Goldman SJ, Kasaian M, Chaudhary D. IL-33 synergizes with IgE-dependent and IgE-independent agents to promote mast cell and basophil activation. Inflamm Res (2010) 59(3):207–18.10.1007/s00011-009-0088-5[↩]
- Andrade MV, Iwaki S, Ropert C, Gazzinelli RT, Cunha-Melo JR, Beaven MA. Amplification of cytokine production through synergistic activation of NFAT and AP-1 following stimulation of mast cells with antigen and IL-33. Eur J Immunol (2011) 41(3):760–72.10.1002/eji.201040718[↩]
- Fux M, Pecaric-Petkovic T, Odermatt A, Hausmann OV, Lorentz A, Bischoff SC, et al. IL-33 is a mediator rather than a trigger of the acute allergic response in humans. Allergy (2014) 69(2):216–22.10.1111/all.12309[↩]
- Kubo Y, Nakano K. Regulation of histamine synthesis in mouse CD4+ and CD8+ T lymphocytes. Inflamm Res (1999) 48(3):149–53.10.1007/s000110050438[↩]
- Szeberenyi JB, Pallinger E, Zsinko M, Pos Z, Rothe G, Orso E, et al. Inhibition of effects of endogenously synthesized histamine disturbs in vitro human dendritic cell differentiation. Immunol Lett (2001) 76(3):175–82.10.1016/S0165-2478(01)00184-5[↩]
- Takamatsu S, Nakashima I, Nakano K. Modulation of endotoxin-induced histamine synthesis by cytokines in mouse bone marrow-derived macrophages. Inflamm Res (1997) 46(Suppl 1):S91–2.10.1007/s000110050106[↩]
- Stegaev V, Nies AT, Porola P, Mieliauskaite D, Sanchez-Jimenez F, Urdiales JL, et al. Histamine transport and metabolism are deranged in salivary glands in Sjogren’s syndrome. Rheumatology (Oxford) (2013) 52(9):1599–608.10.1093/rheumatology/ket188[↩]
- Konttinen YT, Husu H, Han X, Passani MB, Ballerini C, Stegaev V, et al. Non-professional histamine producing cells, immune responses and autoimmunity. In: Stark H, editor. , editor. H4 Receptor: A Novel Drug Target in Immunoregulation and Inflammation. (2013). p. 201–58.[↩][↩]
- Raithel M, Ulrich P, Hochberger J, Hahn EG. Measurement of gut diamine oxidase activity. Diamine oxidase as a new biologic marker of colorectal proliferation? Ann N Y Acad Sci 1998;859:262–6[↩]
- Kalchmair B, Klocker J, Perkmann R, et al. .Alterations in plasma amine oxidase activities in a compartment syndrome model. Inflamm Res 2003;52(suppl)1:S67–8.[↩]
- Schwelberger HG. Diamine oxidase (DAO) enzyme and gene. In: Falus A. ed. Histamine: biology and medical aspects. Budapest, Hungary: SpringMed Publishing, 2004:43–52.[↩]
- Schwelberger HG, Bodner E. Purification and characterization of diamine oxidase from porcine kidney and intestine. Biochim Biophys Acta 1997;1340:152–64.[↩]
- Brown DD, Tomchick R, Axelrod J. The distribution and properties of a histamine-methylating enzyme. J Biol Chem 1959;234:2948–50.[↩]
- Klocker J, Matzler SA, Huetz GN, Drasche A, Kolbitsch C, Schwelberger HG. Expression of histamine degrading enzymes in porcine tissues. Inflamm Res 2005;54(suppl):S54–7.[↩][↩]
- Raithel M, Kufner M, Ulrich P, Hahn EG. The involvement of the histamine degradation pathway by diamine oxidase in manifest gastrointestinal allergies. Inflamm Res 1999;48(suppl):S75–6.[↩]
- Schmidt WU, Sattler J, Hesterberg R, et al. .Human intestinal diamine oxidase (DAO) activity in Crohn’s disease: a new marker for disease assessment? Agents Actions 1990;30:267–70.[↩]
- Tsujikawa T, Uda K, Ihara T, Andoh A, Fujiyama Y, Bamba T. Changes in serum diamine oxidase activity during chemotherapy in patients with hematological malignancies. Cancer Lett 1999;147:195–8.[↩][↩]
- Schwelberger HG. Histamine N-methyltransferase (HNMT) enzyme and gene. In: Falus A. ed. Histamine: biology and medical aspects. Budapest, Hungary: SpringMed Publishing, 2004:53–9.[↩]
- Yamauchi K, Sekizawa K, Suzuki H, et al. .Structure and function of human histamine N-methyltransferase: critical enzyme in histamine metabolism in airway. Am J Physiol 1994;267:L342–9.[↩]
- Ionescu G, Kiehl R. Monoamine and diamine oxidase activities in atopic eczema. Allergy 1988;43:318–9.[↩]
- Ring J, Sedlmeier F, Dorsch W, Hermann K. In vitro IgE eluation and histamine releasability from peripheral leukocytes of atopics and normals. J Dermatol Sci 1991;2:413–21.[↩]
- Sampson HA, Jolie P. L.. Increased plasma histamine concentrations after food challenges in children with atopic dermatitis. N Engl J Med 1984;311:372–6.[↩]
- Maintz L, Benfadal S, Allam JP, Hagemann T, Fimmers R, Novak N. Evidence for a reduced histamine degradation capacity in a subgroup of patients with atopic eczema. J Allergy Clin Immunol 2006;117:1106–12.[↩][↩]
- Fiedler EM, Pelchrzim R, Focke M, Zuberbier T, Worm M. Bedeutung von exogen zugeführtem Histamin bei Patienten mit atopischer Dermatitis. (Histamine intolerance. Effect of ingested histamine on the skin status of atopic patients.) Allergo J 2004;13:S49–50[↩]
- Bieganski T, Osinska Z, Maslinski C. Inhibition of plant and mammalian diamine oxidase by substrate analogues. Agents Actions 1982;12:41–6.[↩]
- Ignesti G. Equations of substrate-inhibition kinetics applied to pig kidney diamine oxidase (DAO, E.C. 1.4.3.6). J Enzyme Inhib Med Chem 2003;18:463–73.[↩]
- Bodis J, Tinneberg HR, Schwarz H, Papenfuss F, Torok A, Hanf V. The effect of histamine on progesterone and estradiol secretion of human granulosa cells in serum-free culture. Gynecol Endocrinol 1993;7:235–9.[↩]
- Kalogeromitros D, Katsarou A, Armenaka M, Rigopoulos D, Zapanti M, Stratigos I. Influence of the menstrual cycle on skin-prick test reactions to histamine, morphine and allergen. Clin Exp Allergy 1995;25:461–6.[↩]
- Morel F, Surla A, Vignais PV. Purification of human placenta diamine oxidase. Biochem Biophys Res Commun 1992;187:178–86.[↩][↩]
- Jarisch R, Wantke F. Wine and headache. Int Arch Allergy Immunol 1996;110:7–12.[↩]
- Sattler J, Lorenz W. Intestinal diamine oxidases and enteral-induced histaminosis: studies on three prognostic variables in an epidemiological model. J Neural Transm Suppl 1990;32:291–314.[↩]
- Wantke F, Proud D, Siekierski E, Kagey-Sobotka A. Daily variations of serum diamine oxidase and the influence of H1 and H2 blockers: a critical approach to routine diamine oxidase assessment. Inflamm Res 1998;47:396–400.[↩]
- Bodmer S, Imark C, Kneubuhl M. Biogenic amines in foods: histamine and food processing. Inflamm Res 1999;48:296–300.[↩]
- Sarkadi L. Histamine in food. In: Falus A, Grosman N, Darvas Z. eds. Histamine: biology and medical aspects. Budapest, Hungary: SpringMed Publishing, 2004:176–85.[↩][↩]
- Bover-Cid S, Holzapfel W. Biogenic amine production by bacteria. In: Morgan D, White A, Sánchez-Jiménez F, Bardócz S. eds. Biogenically active amines in food. Luxembourg City, Luxembourg: EC Publication, 2000:20–9.[↩]
- Beutling DM. Biogene Amine in der Ernährung. (Biogenic amines in nutrition.) Berlin, Germany: Springer, 1996[↩]
- Pechanek U, Pfannhauser W, Woidich H. [Content of biogenic amines in four food groups of the Austrian marketplace]. Z Lebensm Unters Forsch 1983;176:335–40[↩]
- Nordic Council of Ministers Present status of biogenic amines in foods in Nordic countries. Tema Nord 2002: 524 (ISBN: 92-893-0773-0). Cited by: Sarkadi L. Histamine in food. In: Falus A, Grosman N, Darvas Z. eds. Histamine: biology and medical aspects. Budapest, Hungary: SpringMed Publishing, 2004:176–85.[↩]
- Sattler J, Lorenz W, Kubo K, Schmal A, Sauer S, Luben L. Food-induced histaminosis under diamine oxidase (DAO) blockade in pigs: further evidence of the key role of elevated plasma histamine levels as demonstrated by successful prophylaxis with antihistamines. Agents Actions 1989;27:212–4.[↩]
- Backhaus B, Raithel M, Hahn EG. Nicht-immunologisch induzierte Histaminfreisetzung an vitalen menschlichen Darmschleimhautbiopsien durch Stimulation mit Polyaminen. (Nonimmunologically induced histamine release of biopsies of vital human intestinal mucosa after stimulation with polyamines.) Allergo J 2005;14:41[↩]
- Bieck PR, Antonin KH. Oral tyramine pressor test and the safety of monoamine oxidase inhibitor drugs: comparison of brofaromine and tranylcypromine in healthy subjects. J Clin Psychopharmacol 1988;8:237–45.[↩][↩]
- Littlewood JT, Gibb C, Glover V, Sandler M, Davies PT, Rose FC. Red wine as a cause of migraine. Lancet 1988;1:558–9.[↩][↩][↩]
- Forsythe WI, Redmond A. Two controlled trials of tyramine in children with migraine. Dev Med Child Neurol 1974;16:794–9.[↩]
- Jansen SC, van DM, Bottema KC, Dubois AE. Intolerance to dietary biogenic amines: a review. Ann Allergy Asthma Immunol 2003;91:233–40.[↩][↩]
- Ziegler DK, Stewart R. Failure of tyramine to induce migraine. Neurology 1977;27:725–6.[↩]
- Izquierdo-Pulido, M. Biogenic amines in European beers. J Agric Food Chem 1996;44:33159–63.[↩]
- Wantke F, Hemmer W, Gotz M, Jarisch R. Adverse reactions to alcoholic beverages: a diagnostic guideline. Clin Exp Allergy 1997;27:343[↩]
- Dahl R, Henriksen JM, Harving H. Red wine asthma: a controlled challenge study. J Allergy Clin Immunol 1986;78:1126–9.[↩][↩][↩]
- Kanny G, Bauza T, Fremont S, et al. .Histamine content does not influence the tolerance of wine in normal subjects. Allerg Immunol (Paris) 1999;31:45–8.[↩]
- Kanny G, Gerbaux V, Olszewski A, et al. .No correlation between wine intolerance and histamine content of wine. J Allergy Clin Immunol 2001;107:375–8.[↩][↩]
- Yang WH, Purchase EC. Adverse reactions to sulfites. CMAJ 1985;133:865–7, 880.[↩]
- Gunnison AF, Jacobsen DW. Sulfite hypersensitivity. A critical review. CRC Crit Rev Toxicol 1987;17:185–214.[↩]
- Przybilla B, Ring J. [Sulfite hypersensitivity]. Hautarzt 1987;38:445–8[↩]
- Brink B, Damink C., Joosten HM, Huisin’t Veld JH. Occurrence and formation of biologically active amines in foods Int J Food Microbiol 1990;11:73–84.[↩]
- Wohrl S, Hemmer W, Focke M, Rappersberger K, Jarisch R. Histamine intolerance-like symptoms in healthy volunteers after oral provocation with liquid histamine. Allergy Asthma Proc 2004;25:305–11.[↩]
- Chung BY, Cho SI, Ahn IS, et al. Treatment of Atopic Dermatitis with a Low-histamine Diet. Annals of Dermatology. 2011;23(Suppl 1):S91-S95. doi:10.5021/ad.2011.23.S1.S91. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3199434/[↩]
- Moneret-Vautrin DA, de Korwin JD, Tisserant J, Grignon M, Claudot N. Ultrastructural study of the mast cells of the human duodenal mucosa. Clin Allergy 1984;14:471–81.[↩]