Contents
What is thyroglobulin
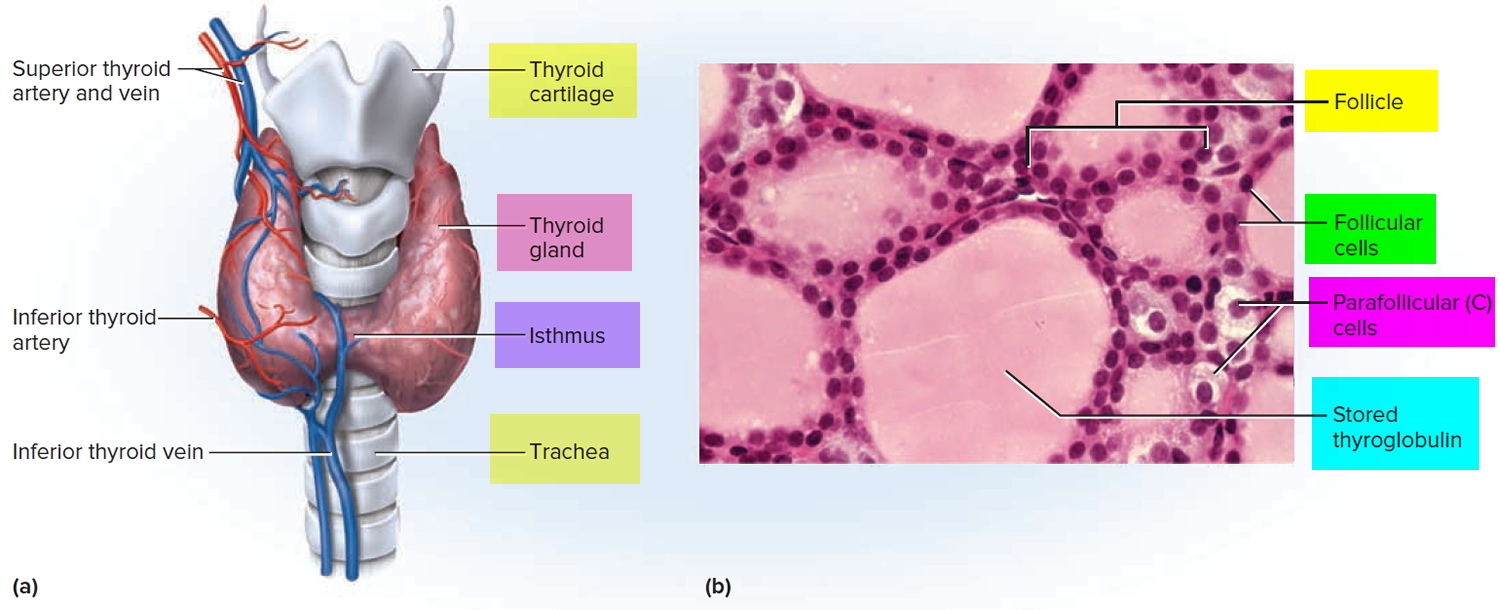
Thyroglobulin is thyroid-specific glycoprotein (approximately 660 KDa) and the precursor of thyroid hormones, is produced by your thyroid gland. The thyroid gland produces hormones thyroxine (T4) and triiodothyronine (T3) that help to regulate the rate at which your body uses energy. The thyroid gland is a small, butterfly-shaped organ that lies flat against the windpipe in the throat and is composed primarily of very small, ball-shaped structures called follicles that produce and store thyroglobulin.
Thyroglobulin serves as the source of thyroxine (T4) and triiodothyronine (T3) production within the lumen of thyroid follicles. For thyroxine (T4) and triiodothyronine (T3) release, thyroglobulin is reabsorbed into thyrocytes and proteolytically degraded, liberating T4 and T3 for secretion. The production of thyroxine (T4) and triiodothyronine (T3) hormones and their release into the bloodstream are stimulated by the pituitary hormone thyroid stimulating hormone (TSH).
Thyroglobulin is produced by all healthy individuals, and normally its concentration in blood is very low. Small amounts of intact thyroglobulin are secreted alongside thyroxine (T4) and triiodothyronine (T3) hormones and are detectable in the serum of healthy individuals, with levels roughly paralleling thyroid size (0.5-1.0 ng/mL thyroglobulin per gram thyroid tissue, depending on thyroid-stimulating hormone [TSH] level). In situations of disordered thyroid growth (e.g., goiter), increased thyroid activity (e.g., Grave disease), or thyroid glandular destruction (e.g., thyroiditis) larger amounts of thyroglobulin may be released into the circulation.
Thyroglobulin concentration increases in both benign (Graves disease, subacute thyroiditis, Hashimoto thyroiditis) and malignant conditions (thyroid adenoma, thyroid papillary and follicular cancer). Therefore, it is on one hand a non-specific indicator of a thyroid dysfunction and on the other hand, a tumor marker that can be used to monitor patients with thyroid papillary and follicular cancer after diagnosis.
The thyroglobulin test is primarily used as a tumor marker to evaluate the effectiveness of treatment for thyroid cancer (e.g., differentiated follicular cell-derived thyroid carcinoma) and to monitor for recurrence. Not every thyroid cancer will produce thyroglobulin, but the most common types, the well-differentiated papillary and follicular thyroid cancers, frequently do, resulting in increased levels of thyroglobulin in the blood. Because thyroglobulin is thyroid-specific, serum thyroglobulin concentrations should be undetectable, or very low, after the thyroid gland is removed during treatment for thyroid cancer.
Thyroglobulin testing may be used, along with a thyroid stimulating hormone (TSH) test, prior to thyroid cancer treatment to determine whether the cancer is producing thyroglobulin. If it is, then the test can be ordered at intervals after treatment to monitor for cancer recurrence. Several thyroglobulin levels may be ordered over a period of time (serial samples) to look at the change in concentration. The change often provides more information than a single value.
Thyroglobulin testing is also occasionally ordered to help determine the cause of hyperthyroidism and to monitor the effectiveness of treatment for conditions such as Graves disease.
Rarely, thyroglobulin test may be ordered to help differentiate between subacute thyroiditis and thyrotoxicosis factitia and to determine the cause of congenital hypothyroidism in newborns.
Current clinical guidelines consider a serum thyroglobulin of >1 ng/mL in an athyrotic individual (person whose thyroid gland has been surgically removed) as suspicious of possible residual or recurrent disease. To improve diagnostic accuracy, it is recommended that at least initially this measurement is obtained after thyroid stimulating hormone (TSH) stimulation, either following thyroid hormone withdrawal, or after injection of recombinant human TSH. Most patients will have a relatively low risk of recurrence, and will thereafter only require unstimulated thyroglobulin measurement. If unstimulated (on thyroxine) serum thyroglobulin measurements are <0.1 to 0.2 ng/mL, the risk of disease is <1%. Patients with higher thyroglobulin levels, who have no demonstrable remnant of thyroid tissue, might require additional testing, such as further stimulated thyroglobulin measurements, neck ultrasound, or isotope imaging. A stimulated thyroglobulin >2 ng/mL is considered suspicious.
The presence of anti-thyroglobulin autoantibodies (thyroglobulin antibody), which occur in 15% to 30% of thyroid cancer patients, could lead to misleading thyroglobulin results. In immunometric assays, the presence of thyroglobulin antibody can lead to false-low results; whereas it might lead to false-high results in competitive assays.
Thyroglobulin antibodies are immune proteins produced that target thyroglobulin. Depending on the test method used by the laboratory, thyroglobulin antibodies can interfere with the results of the thyroglobulin test if they are present in the blood of the person being tested. If the thyroglobulin antibodies are present, either the thyroglobulin test cannot be used to monitor levels or a method that is not affected by thyroglobulin antibody must be used to monitor levels.
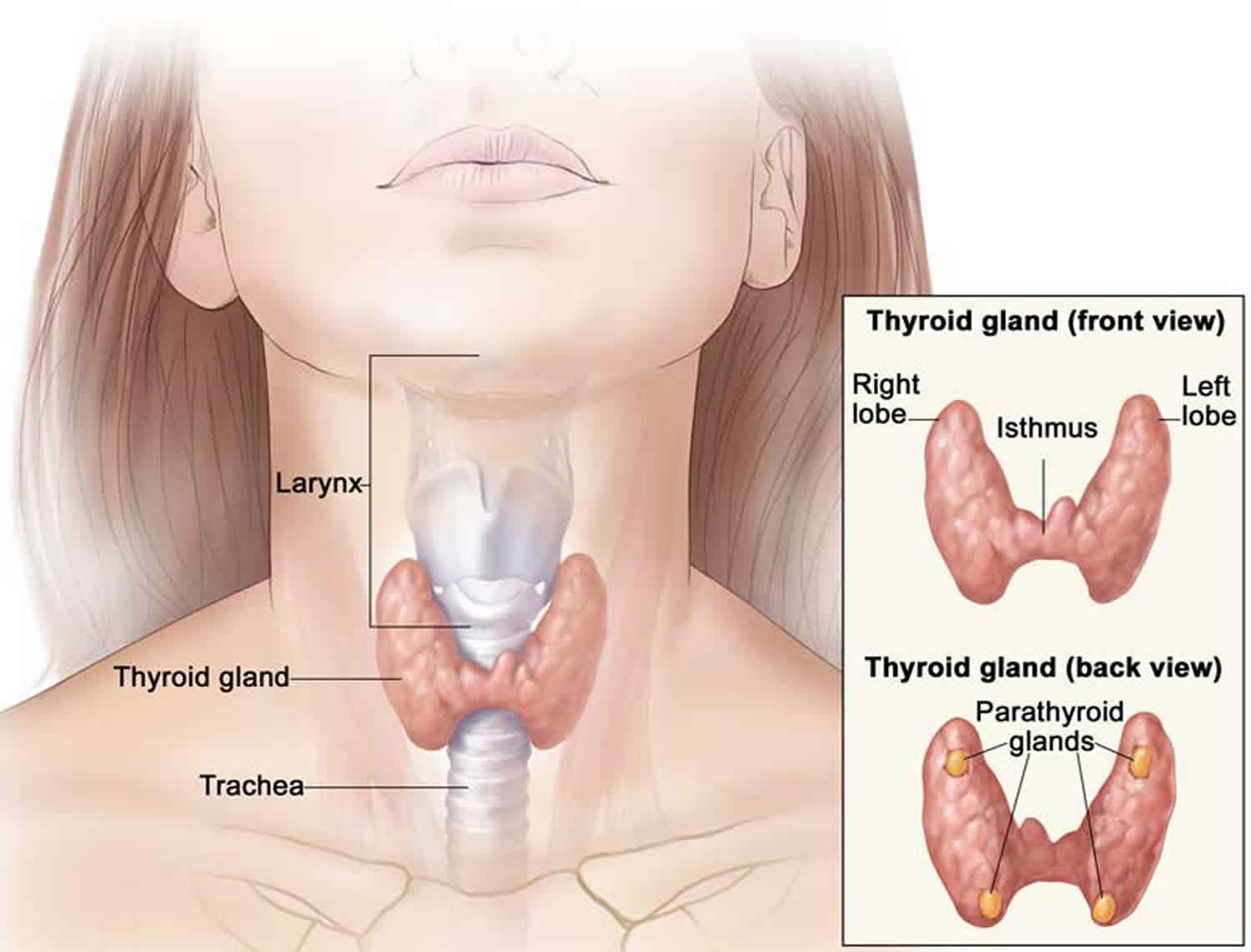
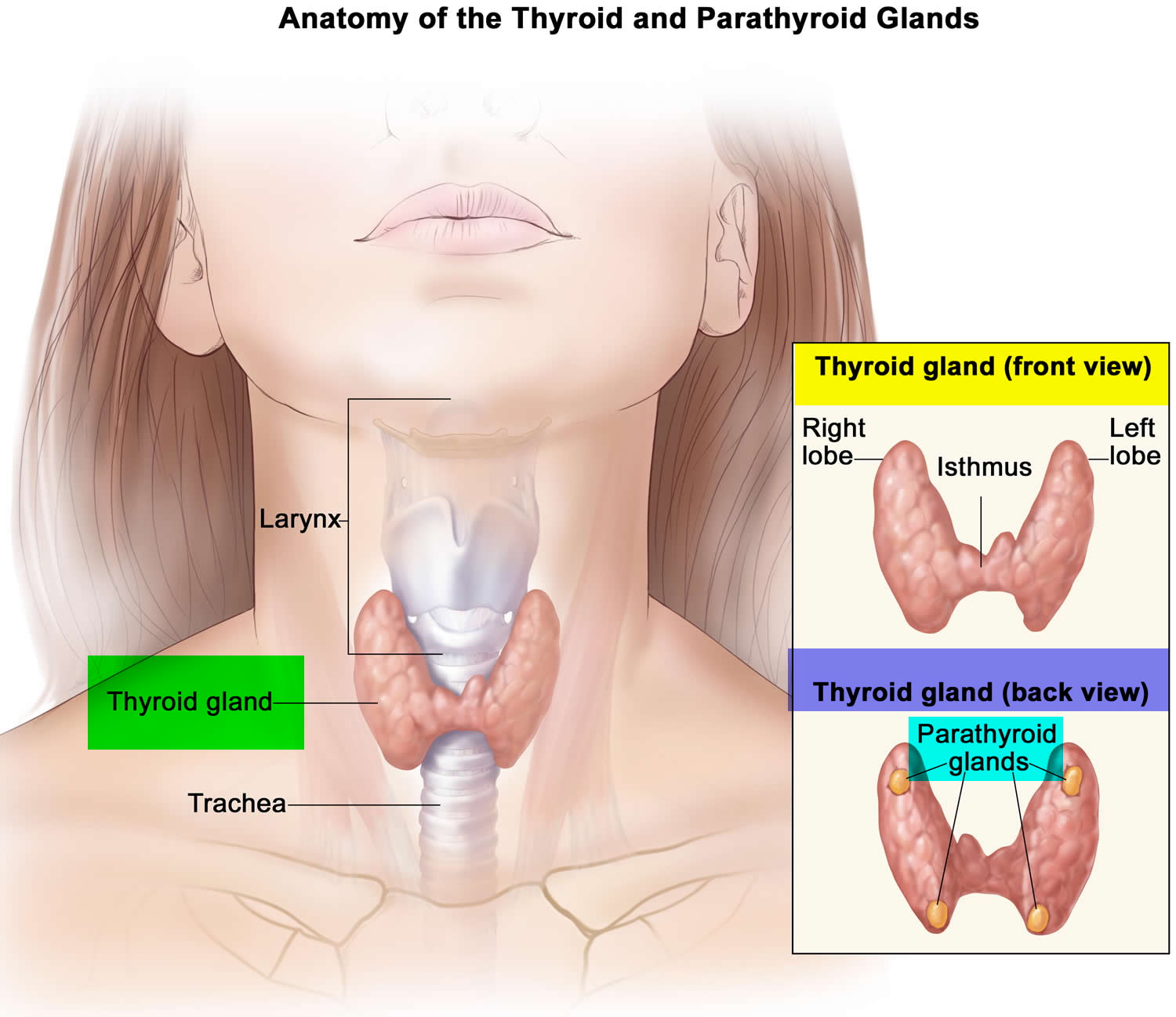
Figure 1. Thyroid gland location
Figure 2. Thyroid gland anatomy
Thyroglobulin antibody
Thyroglobulin autoantibodies are antibodies that develop when a person’s immune system mistakenly targets thyroglobulin, the storage form of thyroid hormones, leading to chronic inflammation of the thyroid (thyroiditis), tissue damage, and/or disruption of thyroid function. Laboratory tests detect the presence and measure the quantity of specific thyroid autoantibodies in the blood. Circulating autoantibodies to thyroglobulin (thyroglobulin antibody) and thyroperoxidase (thyroperoxidase antibody) are hallmarks of the two most common types of autoimmune thyroid diseases, i.e. chronic autoimmune thyroiditis [Hashimoto’s thyroiditis, with its classical goitrous form and the atrophic variant] 1 and Graves’ disease 2. Thyroglobulin antibody, usually at low concentrations, may also be detected in patients with non-autoimmune thyroid disease, including nontoxic multinodular goiter 3 and papillary thyroid carcinoma 4, and in individuals with no apparent thyroid disease (normal individuals) 3. Thyroglobulin antibody can interfere with thyroglobulin measurement, which represents an essential tool for the follow-up of patients with differentiated thyroid cancer (papillary thyroid carcinoma and follicular thyroid carcinoma) 5.
Thyroglobulin antibody normal level < 4.0 IU/mL
What does it mean to have high thyroglobulin antibodies?
Circulating autoantibodies to thyroglobulin (thyroglobulin antibody) and thyroperoxidase (thyroperoxidase antibody) are hallmarks of the two most common types of autoimmune thyroid diseases, i.e. chronic autoimmune thyroiditis [Hashimoto’s thyroiditis, with its classical goitrous form and the atrophic variant] 1 and Graves’ disease 2.
What is Hashimoto thyroiditis?
Hashimoto thyroiditis also known as chronic lymphocytic thyroiditis, is a condition in which your immune system attacks your thyroid gland. The thyroid gland is part of your endocrine system, which produces hormones that coordinate many of your body’s functions. Hashimoto thyroiditis is the most common form of thyroid gland inflammation (thyroiditis) and the most frequent cause of decreased thyroid hormone production and underactive thyroid gland (hypothyroidism).
Hashimoto thyroiditis is the most common cause of hypothyroidism in the United States. Hashimoto thyroiditis can affect anyone at any age but occurs most commonly in women who are between 30 and 50 years of age. Hashimoto thyroiditis can also occur in men and women of any age and in children. About 1 in 1,000 people are diagnosed annually with Hashimoto thyroiditis, and the number has been increasing over time due to improvements in diagnostic techniques. The ratio of women to men diagnosed with the disease is 20 to 1. People with a family history of thyroid diseases or with other autoimmune diseases, especially type 1 diabetes or adrenal insufficiency are at increased risk.
The thyroid gland is a butterfly-shaped organ that lies flat against the windpipe in the throat. It produces the hormones thyroxine (T4) and triiodothyronine (T3) and plays an important role in controlling the body’s metabolism.
With Hashimoto thyroiditis, the thyroid becomes enlarged (called a goiter), firm, and rubbery but not usually tender. Thyroid gland tissue is slowly destroyed by white blood cells called lymphocytes that move into the thyroid gland and by one or more thyroid autoantibodies. This causes a progressive decrease in the production of thyroid hormones.
Doctors test your thyroid function to help detect Hashimoto thyroiditis. Treatment of Hashimoto thyroiditis with thyroid hormone replacement usually is simple and effective.
Hashimoto thyroiditis symptoms
You might not notice signs or symptoms of Hashimoto thyroiditis at first, or you may notice a swelling at the front of your throat (goiter). Hashimoto thyroiditis typically progresses slowly over years and causes chronic thyroid damage, leading to a drop in thyroid hormone levels in your blood. The signs and symptoms are mainly those of an underactive thyroid gland (hypothyroidism).
Signs and symptoms of hypothyroidism include:
- Fatigue and sluggishness
- Increased sensitivity to cold
- Constipation
- Pale, dry skin
- A puffy face
- Brittle nails
- Hair loss
- Enlargement of the tongue
- Unexplained weight gain
- Muscle aches, tenderness and stiffness
- Joint pain and stiffness
- Muscle weakness
- Excessive or prolonged menstrual bleeding (menorrhagia)
- Depression
- Memory lapses
Causes of Hashimoto thyroiditis
Hashimoto thyroiditis is an autoimmune disorder in which your immune system creates antibodies that damage your thyroid gland. Doctors don’t know what causes your immune system to attack your thyroid gland. Some scientists think a virus or bacterium might trigger the response, while others believe a genetic flaw may be involved.
A combination of factors — including heredity, sex and age — may determine your likelihood of developing the disorder.
Risk factors for Hashimoto thyroiditis
These factors may contribute to your risk of developing Hashimoto’s disease:
- Sex. Women are much more likely to get Hashimoto’s disease.
- Age. Hashimoto’s disease can occur at any age but more commonly occurs during middle age.
- Heredity. You’re at higher risk for Hashimoto’s disease if others in your family have thyroid or other autoimmune diseases.
- Other autoimmune disease. Having another autoimmune disease — such as rheumatoid arthritis, type 1 diabetes or lupus — increases your risk of developing Hashimoto’s disease.
- Radiation exposure. People exposed to excessive levels of environmental radiation are more prone to Hashimoto’s disease.
Complications of Hashimoto thyroiditis
Left untreated, an underactive thyroid gland (hypothyroidism) caused by Hashimoto’s disease can lead to a number of health problems:
- Goiter. Constant stimulation of your thyroid to release more hormones may cause the gland to become enlarged, a condition known as goiter. Hypothyroidism is one of the most common causes of goiters. Although generally not uncomfortable, a large goiter can affect your appearance and may interfere with swallowing or breathing.
- Heart problems. Hashimoto’s disease also may be associated with an increased risk of heart disease, primarily because high levels of low-density lipoprotein (LDL) cholesterol — the “bad” cholesterol — can occur in people with an underactive thyroid gland (hypothyroidism). If left untreated, hypothyroidism can lead to an enlarged heart and, possibly, heart failure.
- Mental health issues. Depression may occur early in Hashimoto’s disease and may become more severe over time. Hashimoto’s disease can also cause sexual desire (libido) to decrease in both men and women and can lead to slowed mental functioning.
- Myxedema. This rare, life-threatening condition can develop due to long-term hypothyroidism as a result of untreated Hashimoto’s disease. Its signs and symptoms include drowsiness followed by profound lethargy and unconsciousness. A myxedema coma may be triggered by exposure to cold, sedatives, infection or other stress on your body. Myxedema requires immediate emergency medical treatment.
- Birth defects. Babies born to women with untreated hypothyroidism due to Hashimoto’s disease may have a higher risk of birth defects than do babies born to healthy mothers. Doctors have long known that these children are more prone to intellectual and developmental problems. There may be a link between hypothyroid pregnancies and birth defects, such as a cleft palate. A connection also exists between hypothyroid pregnancies and heart, brain and kidney problems in infants. If you’re planning to get pregnant or if you’re in early pregnancy, be sure to have your thyroid level checked.
Hashimoto thyroiditis diagnosis
In general, your doctor may test for Hashimoto thyroiditis if you’re feeling increasingly tired or sluggish, have dry skin, constipation, and a hoarse voice, or have had previous thyroid problems or goiter.
Diagnosis of Hashimoto thyroiditis is based on your signs and symptoms and the results of blood tests that measure levels of thyroid hormone and thyroid-stimulating hormone (TSH) produced in the pituitary gland. These may include:
- A hormone test. Blood tests can determine the amount of hormones produced by your thyroid and pituitary glands. If your thyroid is underactive, the level of thyroid hormone is low. At the same time, the level of thyroid stimulating hormone (TSH) is elevated because your pituitary gland tries to stimulate your thyroid gland to produce more thyroid hormone.
- An antibody test. Because Hashimoto thyroiditis is an autoimmune disorder, the cause involves production of abnormal antibodies. A blood test may confirm the presence of antibodies against thyroid peroxidase (thyroid peroxidase antibodies), an enzyme normally found in the thyroid gland that plays an important role in the production of thyroid hormones.
In the past, doctors weren’t able to detect an underactive thyroid (hypothyroidism), the main indicator of Hashimoto thyroiditis, until symptoms were fairly advanced. But by using the sensitive TSH test, doctors can diagnose thyroid disorders much earlier, often before you experience symptoms.
Because the thyroid stimulating hormone (TSH) test is the best screening test, your doctor will likely check TSH first and follow with a thyroid hormone test if needed. Thyroid stimulating hormone (TSH) tests also play an important role in managing hypothyroidism. These tests also help your doctor determine the right dosage of medication, both initially and over time.
Hashimoto thyroiditis treatment
Treatment for Hashimoto thyroiditis may include observation and use of medications. If there’s no evidence of hormone deficiency, and your thyroid is functioning normally, your doctor may suggest a wait-and-see approach. If you need medication, chances are you’ll need it for the rest of your life.
Synthetic hormones
If Hashimoto thyroiditis causes thyroid hormone deficiency, you may need replacement therapy with thyroid hormone. This usually involves daily use of the synthetic thyroid hormone levothyroxine (Levoxyl, Synthroid, others).
Synthetic levothyroxine is identical to thyroxine, the natural version of this hormone made by your thyroid gland. The oral medication restores adequate hormone levels and reverses all the symptoms of hypothyroidism.
Treatment with levothyroxine is usually lifelong, but because the dosage you need may change, your doctor is likely to check your TSH level about every 12 months.
Monitoring the dosage
To determine the right dosage of levothyroxine initially, your doctor generally checks your level of TSH after a few weeks of treatment. Excessive amounts of thyroid hormone can accelerate bone loss, which may make osteoporosis worse or add to your risk of this disease. Overtreatment with levothyroxine can also cause heart rhythm disorders (arrhythmias).
If you have coronary artery disease or severe hypothyroidism, your doctor may start treatment with a smaller amount of medication and gradually increase the dosage. Progressive hormone replacement allows your heart to adjust to the increase in metabolism.
Levothyroxine causes virtually no side effects when used in the appropriate dose and is relatively inexpensive. If you change brands, let your doctor know to ensure you’re still receiving the right dosage.
Also, don’t skip doses or stop taking the drug. If you do, signs and symptoms will gradually return.
Effects of other substances
Certain medications, supplements and some foods may affect your ability to absorb levothyroxine. However, taking levothyroxine four hours before or after other medications could remedy the problem. Talk to your doctor if you eat large amounts of soy products or a high-fiber diet or if you take any of the following:
- Iron supplements, including multivitamins that contain iron
- Cholestyramine (Prevalite), a medication used to lower blood cholesterol levels
- Aluminum hydroxide, which is found in some antacids
- Sucralfate, an ulcer medication
- Calcium supplements
Is a combination of hormones needed?
Levothyroxine is the synthetic form of the natural thyroxine (T4). Thyroxine (T4) is converted into triiodothyronine (T3) in your body. While most people are treated successfully with levothyroxine alone, some people don’t feel completely normal on levothyroxine.
Researchers have investigated whether adjusting standard hypothyroidism treatment to replace some thyroxine (T4) with small amounts of triiodothyronine (T3) may offer benefit. But, the majority of studies have determined that the addition of triiodothyronine (T3) does not offer any advantage over treatment with thyroxine (T4) alone.
There is some evidence that triiodothyronine (T3) may offer benefit to certain subsets of people, such as people who have had their thyroid surgically removed (thyroidectomy). Research is ongoing.
Triiodothyronine (T3) can be given alone as liothyronine (Cytomel) or in combination with thyroxine (T4) as liotrix (Thyrolar). Taking a combination thyroxine (T4) and triiodothyronine (T3) ends up producing higher than normal levels of T3, especially soon after the medication is taken. This can cause a fast heart rate, anxiety and trouble sleeping.
But, for those who haven’t gotten enough relief from thyroxine (T4) alone, adding liothyronine (Cytomel) to standard levothyroxine treatment for a three- to six-month trial is a long enough period to see if the combination helps you.
Alternative medicine
Standard treatment for Hashimoto thyroiditis is levothyroxine, the synthetic form of thyroxine (T4). However, extracts are available that contain thyroid hormone derived from the thyroid glands of pigs. These products — Armour Thyroid, for example — contain both levothyroxine and triiodothyronine triiodothyronine (T3).
Doctors have a number of concerns about thyroid hormone extracts such as Armour Thyroid, including:
- The balance of thyroxine (T4) and triiodothyronine (T3) in animals isn’t the same as in humans.
- The exact amount of thyroxine (T4) and triiodothyronine (T3) in each batch of a natural extract product can vary, leading to unpredictable levels of these hormones in your blood.
What is Graves’ disease
Graves’ disease is an immune system disorder that results in the overproduction of thyroid hormones (hyperthyroidism). Although a number of disorders may result in hyperthyroidism, Graves’ disease is a common cause.
Because thyroid hormones affect a number of different body systems, signs and symptoms associated with Graves’ disease can be wide ranging and significantly influence your overall well-being. Although Graves’ disease may affect anyone, it’s more common among women and before the age of 40.
The primary treatment goals are to inhibit the overproduction of thyroid hormones and lessen the severity of symptoms.
Graves’ disease causes
Graves’ disease is caused by a malfunction in the body’s disease-fighting immune system, although the exact reason why this happens is still unknown.
One normal immune system response is the production of antibodies designed to target a specific virus, bacterium or other foreign substance. In Graves’ disease — for reasons that aren’t well understood — the body produces an antibody to one part of the cells in the thyroid gland, a hormone-producing gland in the neck.
Normally, thyroid function is regulated by a hormone released by a tiny gland at the base of the brain (pituitary gland). The antibody associated with Graves’ disease — thyrotropin receptor antibody (TRAb) — acts like the regulatory pituitary hormone. That means that TRAb overrides the normal regulation of the thyroid, causing an overproduction of thyroid hormones (hyperthyroidism).
Cause of Graves’ disease ophthalmopathy
This condition results from a buildup of certain carbohydrates in the muscles and tissues behind the eyes — the cause of which also isn’t known. It appears that the same antibody that can cause thyroid dysfunction may also have an “attraction” to tissues surrounding the eyes.
Graves’ ophthalmopathy often appears at the same time as hyperthyroidism or several months later. But signs and symptoms of ophthalmopathy may appear years before or after the onset of hyperthyroidism. Graves’ ophthalmopathy can also occur even if there’s no hyperthyroidism.
Risk factors for Graves’ disease
Although anyone can develop Graves’ disease, a number of factors can increase the risk of disease. These risk factors include the following:
- Family history. Because a family history of Graves’ disease is a known risk factor, there is likely a gene or genes that can make a person more susceptible to the disorder.
- Gender. Women are much more likely to develop Graves’ disease than are men.
- Age. Graves’ disease usually develops in people younger than 40.
- Other autoimmune disorders. People with other disorders of the immune system, such as type 1 diabetes or rheumatoid arthritis, have an increased risk.
- Emotional or physical stress. Stressful life events or illness may act as a trigger for the onset of Graves’ disease among people who are genetically susceptible.
- Pregnancy. Pregnancy or recent childbirth may increase the risk of the disorder, particularly among women who are genetically susceptible.
- Smoking. Cigarette smoking, which can affect the immune system, increases the risk of Graves’ disease. Smokers who have Graves’ disease are also at increased risk of developing Graves’ ophthalmopathy.
Graves’ disease signs and symptoms
Common signs and symptoms of Graves’ disease include:
- Anxiety and irritability
- A fine tremor of your hands or fingers
- Heat sensitivity and an increase in perspiration or warm, moist skin
- Weight loss, despite normal eating habits
- Enlargement of your thyroid gland (goiter)
- Change in menstrual cycles
- Erectile dysfunction or reduced libido
- Frequent bowel movements
- Bulging eyes (Graves’ ophthalmopathy)
- Fatigue
- Thick, red skin usually on the shins or tops of the feet (Graves’ dermopathy)
- Rapid or irregular heartbeat (palpitations)
Graves’ ophthalmopathy
About 30 percent of people with Graves’ disease show some signs and symptoms of a condition known as Graves’ ophthalmopathy. In Graves’ ophthalmopathy, inflammation and other immune system events affect muscles and other tissues around your eyes. The resulting signs and symptoms may include:
- Bulging eyes (exophthalmos)
- Gritty sensation in the eyes
- Pressure or pain in the eyes
- Puffy or retracted eyelids
- Reddened or inflamed eyes
- Light sensitivity
- Double vision
- Vision loss
Graves’ dermopathy
- An uncommon manifestation of Graves’ disease, called Graves’ dermopathy, is the reddening and thickening of the skin, most often on your shins or the tops of your feet.
Graves’ disease complications
Complications of Graves’ disease can include:
- Pregnancy issues. Possible complications of Graves’ disease during pregnancy include miscarriage, preterm birth, fetal thyroid dysfunction, poor fetal growth, maternal heart failure and preeclampsia. Preeclampsia is a maternal condition that results in high blood pressure and other serious signs and symptoms.
- Heart disorders. If left untreated, Graves’ disease can lead to heart rhythm disorders, changes in the structure and function of the heart muscles, and the inability of the heart to pump enough blood to the body (congestive heart failure).
- Thyroid storm. A rare, but life-threatening complication of Graves’ disease is thyroid storm, also known as accelerated hyperthyroidism or thyrotoxic crisis. It’s more likely when severe hyperthyroidism is untreated or treated inadequately. The sudden and drastic increase in thyroid hormones can produce a number of effects, including fever, profuse sweating, vomiting, diarrhea, delirium, severe weakness, seizures, markedly irregular heartbeat, yellow skin and eyes (jaundice), severe low blood pressure, and coma. Thyroid storm requires immediate emergency care.
- Brittle bones. Untreated hyperthyroidism also can lead to weak, brittle bones (osteoporosis). The strength of your bones depends, in part, on the amount of calcium and other minerals they contain. Too much thyroid hormone interferes with your body’s ability to incorporate calcium into your bones.
Graves’ disease diagnosis
The diagnosis of Graves’ disease may include:
- Physical exam. Your doctor examines your eyes to see if they’re irritated or protruding and looks to see if your thyroid gland is enlarged. Because Graves’ disease increases your metabolism, your doctor will check your pulse and blood pressure and look for signs of tremor.
- Blood sample. Your doctor will order blood tests to determine your levels of thyroid-stimulating hormone (TSH), the pituitary hormone that normally stimulates the thyroid gland, as well as levels of thyroid hormones. People with Graves’ disease usually have lower than normal levels of TSH and higher levels of thyroid hormones. Another laboratory test measures the levels of the antibody known to cause Graves’ disease. This test usually isn’t necessary to make a diagnosis, but a negative result might indicate another cause for hyperthyroidism.
- Radioactive iodine uptake. Your body needs iodine to make thyroid hormones. By giving you a small amount of radioactive iodine and later measuring the amount of it in your thyroid gland with a specialized scanning camera, your doctor can determine the rate at which your thyroid gland takes up iodine. The amount of radioactive iodine taken up by the thyroid gland helps determine if Graves’ disease or another condition is the cause of the hyperthyroidism. This test may be combined with a radioactive iodine scan to show a visual image of the uptake pattern.
- Ultrasound. Ultrasound uses high-frequency sound waves to produce images of structures inside the body. Ultrasound can show if the thyroid gland is enlarged, and is most useful in people who can’t undergo radioactive iodine uptake, such as pregnant women.
- Imaging tests. If the diagnosis of Graves’ ophthalmopathy isn’t clear from a clinical assessment, your doctor may order an imaging test, such as CT scan, a specialized X-ray technology that produces thin cross-sectional images. Magnetic resonance imaging (MRI), which uses magnetic fields and radio waves to create either cross-sectional or 3-D images, also may be used.
Graves’ disease treatment
The treatment goals for Graves’ disease are to inhibit the production of thyroid hormones and to block the effect of the hormones on the body. Some treatments include:
Radioactive iodine therapy
With this therapy, you take radioactive iodine, or radioiodine, by mouth. Because the thyroid needs iodine to produce hormones, the radioiodine goes into the thyroid cells and the radioactivity destroys the overactive thyroid cells over time. This causes your thyroid gland to shrink, and symptoms lessen gradually, usually over several weeks to several months.
Radioiodine therapy may increase your risk of new or worsened symptoms of Graves’ ophthalmopathy. This side effect is usually mild and temporary, but the therapy may not be recommended if you already have moderate to severe eye problems.
Other side effects may include tenderness in the neck and a temporary increase in thyroid hormones. Radioiodine therapy isn’t used for treating pregnant women or women who are breast-feeding.
Because this treatment causes thyroid activity to decline, you’ll likely need treatment later to supply your body with normal amounts of thyroid hormones.
Anti-thyroid medications
Anti-thyroid medications interfere with the thyroid’s use of iodine to produce hormones. These prescription medications include propylthiouracil and methimazole (Tapazole).
Because the risk of liver disease is more common with propylthiouracil, methimazole is considered the first choice when doctors prescribe medication.
When these two drugs are used alone, a relapse of hyperthyroidism may occur at a later time. Taking the drug for longer than a year, however, may result in better long-term results. Anti-thyroid drugs may also be used before or after radioiodine therapy as a supplemental treatment.
Side effects of both drugs include rash, joint pain, liver failure or a decrease in disease-fighting white blood cells. Methimazole isn’t used to treat pregnant women in the first trimester because of the slight risk of birth defects. Therefore, propylthiouracil is the preferred anti-thyroid drug during the first trimester for pregnant women. After the first trimester, methimazole use is generally resumed and propylthiouracil is no longer prescribed.
Beta blockers
These medications don’t inhibit the production of thyroid hormones, but they do block the effect of hormones on the body. They may provide fairly rapid relief of irregular heartbeats, tremors, anxiety or irritability, heat intolerance, sweating, diarrhea, and muscle weakness.
Beta blockers include:
- Propranolol (Inderal)
- Atenolol (Tenormin)
- Metoprolol (Lopressor, Toprol-XL)
- Nadolol (Corgard)
Beta blockers aren’t often prescribed for people with asthma, because the drugs may trigger an asthma attack. These drugs may also complicate management of diabetes.
Surgery
Surgery to remove all or part of your thyroid (thyroidectomy or subtotal thyroidectomy) also is an option for the treatment of Graves’ disease. After the surgery, you’ll likely need treatment to supply your body with normal amounts of thyroid hormones.
Risks of this surgery include potential damage to the nerve that controls your vocal cords and the tiny glands located adjacent to your thyroid gland (parathyroid glands). Your parathyroid glands produce a hormone that controls the level of calcium in your blood. Complications are rare under the care of a surgeon experienced in thyroid surgery.
Treating Graves’ ophthalmopathy
Mild symptoms of Graves’ ophthalmopathy may be managed by using over-the-counter artificial tears during the day and lubricating gels at night. If your symptoms are more severe, your doctor may recommend:
- Corticosteroids. Treatment with prescription corticosteroids, such as prednisone, may diminish swelling behind your eyeballs. Side effects may include fluid retention, weight gain, elevated blood sugar levels, increased blood pressure and mood swings.
- Prisms. You may have double vision either because of Graves’ disease or as a side effect of surgery for Graves’ disease. Though they don’t work for everyone, prisms in your glasses may correct your double vision.
- Orbital decompression surgery. In this surgery, your doctor removes the bone between your eye socket (orbit) and your sinuses — the air spaces next to the orbit. This gives your eyes room to move back to their original position. This treatment is usually used if pressure on the optic nerve threatens the loss of vision. Possible complications include double vision.
- Orbital radiotherapy. Orbital radiotherapy was once a common treatment for Graves’ ophthalmopathy, but the benefits of the procedure aren’t clear. Orbital radiotherapy uses targeted X-rays over the course of several days to destroy some of the tissue behind your eyes. Your doctor may recommend orbital radiotherapy if your eye problems are worsening and prescription corticosteroids alone aren’t effective or well-tolerated.
Graves’ ophthalmopathy doesn’t always improve with treatment for Graves’ disease. Symptoms of Graves’ ophthalmopathy may even get worse for three to six months. After that, the signs and symptoms of Graves’ ophthalmopathy usually stabilize for a year or so and then begin to get better, often on their own.
Lifestyle and home remedies
If you have Graves’ disease, make your mental and physical well-being a priority:
Eating well and exercising can enhance the improvement in some symptoms while being treated and help you feel better in general. For example, because your thyroid controls your metabolism, you may have a tendency to gain weight when the hyperthyroidism is corrected. Brittle bones also can occur with Graves’ disease, and weight-bearing exercises can help maintain bone density.
Easing stress as much as you can may be helpful, as stress may trigger or worsen Graves’ disease. Listening to music, taking a warm bath or walking can help relax you and put you in a better frame of mind. Partner with your doctor to construct a plan that incorporates good nutrition, exercise and relaxation into your daily routine.
For Graves’ ophthalmopathy
These steps may make your eyes feel better if you have Graves’ ophthalmopathy:
- Apply cool compresses to your eyes. The added moisture may soothe your eyes.
- Wear sunglasses. When your eyes protrude, they’re more vulnerable to ultraviolet rays and more sensitive to bright light. Wearing sunglasses that wrap around the sides of your head will also lessen the irritation of your eyes from the wind.
- Use lubricating eyedrops. Eyedrops may relieve the dry, scratchy sensation on the surface of your eyes. A paraffin-based gel, such as Lacri-Lube, can be applied at night.
- Elevate the head of your bed. Keeping your head higher than the rest of your body lessens fluid accumulation in the head and may relieve the pressure on your eyes.
- Don’t smoke. Smoking worsens Graves’ ophthalmopathy.
For Graves’ dermopathy
If the disease affects your skin (Graves’ dermopathy), use over-the-counter creams or ointments containing hydrocortisone to relieve swelling and reddening. In addition, using compression wraps on your legs may help.
- Latrofa F, Pinchera A 2007 Autoimmune hypothyroidism. In: Weetman AP, ed. Autoimmune diseases in endocrinology. Totowa, NJ: Humana Press; 136–174[↩][↩]
- Marcocci C, Marino M 2005 Thyroid-directed antibodies. In: Braverman LE, Utiger RD, eds. Werner & Ingbar’s the thyroid: a fundamental and clinical text. Philadelphia: Lippincott, Williams & Wilkins; 360–372[↩][↩]
- Mariotti S, Pisani S, Russova A, Pinchera A 1982 A new solid-phase immunoradiometric assay for anti-thyroglobulin autoantibody. J Endocrinol Invest 5:227–233[↩][↩]
- Pacini F, Mariotti S, Formica N, Elisei R, Anelli S, Capotorti E, Pinchera A 1988 Thyroid autoantibodies in thyroid cancer: incidence and relationship with tumour outcome. Acta Endocrinol (Copenh) 119:373–380[↩]
- Mazzaferri EL, Robbins RJ, Spencer CA, Braverman LE, Pacini F, Wartofsky L, Haugen BR, Sherman SI, Cooper DS, Braunstein GD, Lee S, Davies TF, Arafah BM, Ladenson PW, Pinchera A 2003 A consensus report of the role of serum thyroglobulin as a monitoring method for low-risk patients with papillary thyroid carcinoma. J Clin Endocrinol Metab 88:1433–1441[↩]