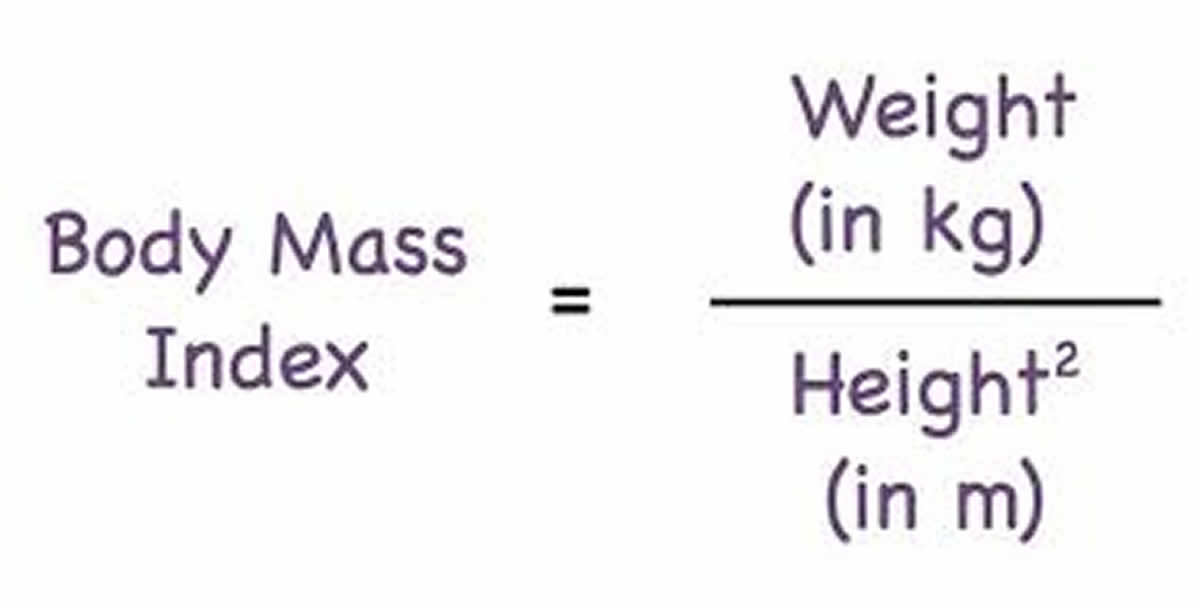Contents
Weight Loss and Diet Pills Review
More than two-thirds of adults and one-third of children in the United States are overweight or obese. Obesity or being overweight may increase the risk of many health problems, including type 2 diabetes, heart disease, and certain cancers. If you’re pregnant, excess weight may lead to short- and long-term health problems for you and your child. Achieving a healthy weight, eating a healthy diet, and being physically active can help prevent these weight-related diseases.
Some people, in their efforts to lose weight, turn to unproven dietary supplements (sometimes marketed as “fat burners” or appetite suppressants), which can have harmful side effects. If you’re thinking about starting a new weight-loss program, talk with your health care provider, who can assess your weight and health risks, determine whether you need to lose weight, and provide information that will help you make informed decisions about an effective weight-loss program.
The use of weight loss supplements for weight loss has become increasingly popular, as reflected by the $55.4 billion spent in the U.S. in 2006 for weight loss and diet control and on more than 85,000 different combinations of vitamins, minerals, botanicals, amino acids, probiotics, and other supplement ingredients 1, 2. Based on a study by the National Center for Complementary and Alternative Medicine, 36% of adults 3 and 1 in 10 teens 4 are using some form of weight loss supplements at some point in their lives, which rises to 62% when including megavitamins or prayer. Dietary supplements can easily be purchased by people of all ages at pharmacies, grocery stores, health food stores, gyms, and other retailers. And unlike prescription medications, supplements do not require premarketing approval before they reach store shelves. Under the Dietary Supplement Health and Education Act of 1994, anything labeled as a dietary supplement is assumed to be safe until proven otherwise. The FDA is charged with the unenviable task of identifying and removing dangerous supplements only after they have caused harm. Although dietary and herbal supplements are governed under the Dietary Supplement Health and Education Act of 1994, weight loss supplements are not presently regulated by the U.S. Federal Drug Administration and the safety profiles of many are unknown 5, 6. And potentially dangerous supplements are widely available 7.
More than 500 supplements have already been found to be adulterated with pharmaceuticals or pharmaceutical analogues, including new stimulants, novel anabolic steroids, unapproved antidepressants, banned weight-loss medications, and untested sildenafil analogues 8. In 2013 alone, researchers discovered two new stimulants in widely marketed supplements. Researchers have identified a new analogue of methamphetamine, N,α-diethyl-phenylethylamine (N,α-DEPEA), in a popular sports supplement 9. FDA scientists discovered another stimulant, β-methylphenethylamine (β-MePEA) — a novel analogue of amphetamine — in nine supplements 10. N,α-DEPEA and β-MePEA have never been studied in humans, and their adverse effects are entirely unknown; yet they are sold as “natural” products without having undergone any premarketing testing for safety. Although supplements containing N,α-DEPEA were voluntarily withdrawn from the market, supplements containing β-MePEA remain widely available. 7.
Epidemiologists at the Centers for Disease Control and Prevention recently confirmed what an astute liver-transplant surgeon in Honolulu already suspected: OxyElite Pro, a popular over-the-counter supplement, was responsible for a cluster of cases of severe hepatitis and liver failure 11. Although patients began to develop severe hepatitis in May 2013, the Food and Drug Administration (FDA), whose job it is to remove dangerous supplements from store shelves, did not learn of the cases until mid-September, 4 months later. By February 2014, the CDC had linked 97 cases, resulting in 47 hospitalizations, three liver transplantations, and one death, to OxyElite Pro. This dietary supplement was recalled, but nothing has been done to prevent another supplement from causing organ failure or death. Nor have any changes been made to improve the FDA’s ability to detect dangerous supplements 7.
The Dangers of Over-the-Counter Diet Pills and Muscle-Building Dietary Supplements
The American Academy of Pediatrics is strongly opposed to the use of supplements for weight loss and muscle building in adolescents 12, 13. Yet a national survey found that 11% of teens had ever used a weight loss supplement. In the same survey, 5% of teens had used creatine, one of many dietary supplements sold for muscle building.
Research shows that dietary supplements sold for weight loss and muscle building often contain drugs that are prescription-only, illegal, or untested for human use. This can lead to serious health risks, including stroke, testicular cancer, severe liver injury, and even death 14, 15, 16, 17.
The rate of liver failure caused by dietary supplements has risen 185% in the past decade 18. In the United States, 16% of cases of serious drug-induced liver injury are attributed to dietary supplements. The vast majority are those sold for weight loss and muscle building 19.
Drugs found in supplements for weight loss and muscle building have included:
- The active ingredient in Meridia, a prescription weight-loss drug pulled from the market in 2010 20,
- Drugs that have been designed to be chemically similar to methamphetamine (aka speed) 21,
- Antidepressants such as fluoxetine, the active ingredient in Prozac 22,
- Anabolic steroids and precursor chemicals, which can cause young athletes to fail doping tests and have been associated with testicular cancer 23,
Many supplements whose labels say they contain “pro-hormones,” “natural steroids,” or “testosterone boosters” actually contain designer anabolic steroids–with dangerous consequences. Use of these compounds through dietary supplements is widespread. In younger men, these supplements may be a common cause of liver injury, problems with bile secretion, testicular disorders, growth of breast tissue, and infertility. Authors advise against dietary supplement use, especially those known or suspected to contain designer steroids 24.
Recalls are not enough to protect people from supplements containing unlisted, unapproved ingredients. In November 2009, the U.S. Food and Drug Administration banned Pai You Gou, a dietary supplement sold for weight loss, after detecting at least two banned pharmaceutical compounds, sibutramine and phenolphthalein, in this product. Sibutramine and phenolphthalein have serious and well-documented side effects. But almost a year after the recall, nearly one in four Brazilian-born Massachusetts women were still using this product 25.
Dietary supplements marketed for weight loss and muscle building have dangerous side effects, including hepatitis and liver failure. The U.S. Centers for Disease Control and Prevention documented a series of severe acute hepatitis (liver injury) and liver failure of unknown cause. Of the 29 confirmed cases of acute hepatitis and liver failure, 24 patients reported using OxyELITE Pro, a dietary supplement sold for weight loss and muscle gain, within 60 days before illness onset 26. Dangerous stimulants are often found in widely available supplements for weight loss and muscle building. When one stimulant is banned, supplement makers often continue using the banned substances, or replace them with a related chemical. Many of these chemicals have never been tested for safety in humans.
Researchers found N,α-DEPEA, in a widely available muscle-building supplement. N,α-DEPEA is a banned and potentially dangerous designer stimulant closely related to methamphetamine 27.
Half of supplements marketed as containing the herbal ingredient acacia rigidula were found to contain β-methylphenylethylamine (BMPEA). BMPEA is a stimulant that has not been assessed for safety or efficacy in humans. It is closely related to the banned compound DMAA, which has been associated with several deaths 28.
Another DMAA substitute, 1,3-dimethylbutylamine (DMBA), has also been banned by the FDA. Despite the ban, researchers found at least a dozen supplements to contain DMBA in dosages ranging from 13 to 120 mg per serving 29.
Muscle-building supplements may lead to increased risk of testicular germ cancer in men. A study of nearly 1000 men found that men who developed testicular germ cancer had used more muscle-building supplements than similar men who did not develop testicular germ cancer. The association was particularly strong among early users, long-term users, and use of two or more types of muscle-building supplements 30.
Diet pills may block the function of other drugs, including cancer treatments. Diet pills may also be associated with organ toxicity. Research suggests that the diet drug orlistat limits the function of a kind of protein, CES2, needed to activate a common cancer drug, PPD. Orlistat has also been associated with gastrointestinal issues, as well as liver toxicity 31.
Diet pills may decrease users’ vitamin D levels. Researchers found that adolescent participants’ vitamin D levels decreased after one month of taking orlistat, despite participants also taking a multivitamin daily. Vitamin D is important for bone health, cell growth, and immune function 32.
Diet pills could be abused by people with eating disorders. In anticipation of the release of alli (the brand of orlistat now available over the counter), researchers expressed concerns regarding the potential for alli to be abused by people with eating disorders. Their concerns are based on documentation of adult and adolescent eating disorder patients frequently abusing supplements sold for weight loss, as well as the possibility that alli’s FDA approval would make it more appealing than other products sold for weight loss 33.
What is healthy weight loss ?
It’s natural for anyone trying to lose weight to want to lose it very quickly. But evidence shows that people who lose weight gradually and steadily (about 1 to 2 pounds per week) are more successful at keeping weight off. Healthy weight loss isn’t just about a “diet” or “program”. It’s about an ongoing lifestyle that includes long-term changes in daily eating and exercise habits. For a free copy of Maintaining a Healthy Weight on the Go go here 34.
- To lose weight, you must use up more calories than you take in. Since one pound equals 3,500 calories, you need to reduce your caloric intake by 500—1000 calories per day to lose about 1 to 2 pounds per week 35.
- Once you’ve achieved a healthy weight, by relying on healthful eating and physical activity most days of the week (about 60—90 minutes, moderate intensity), you are more likely to be successful at keeping the weight off over the long term.
Losing weight is not easy, and it takes commitment 34.
The good news is that no matter what your weight loss goal is, even a modest weight loss, such as 5 to 10 percent of your total body weight, is likely to produce health benefits, such as improvements in blood pressure, blood cholesterol, and blood sugars 36.
For example, if you weigh 200 pounds, a 5 percent weight loss equals 10 pounds, bringing your weight down to 190 pounds. While this weight may still be in the “overweight” or “obese” range, this modest weight loss can decrease your risk factors for chronic diseases related to obesity.
So even if the overall goal seems large, see it as a journey rather than just a final destination. You’ll learn new eating and physical activity habits that will help you live a healthier lifestyle. These habits may help you maintain your weight loss over time.
Health care providers use your Body Mass Index (BMI), to measure of your weight in relation to your height, to define overweight and obesity and is universally expressed in units of kg/m2, resulting from mass in kilograms and height in meters. People who have a BMI between 25 and 30 kg/m2 are considered overweight. Obesity is defined as having a BMI of 30 kg/m2 or greater. BMI is considered an important measure for understanding population trends. For individuals, it is one of many factors that should be considered in evaluating healthy weight, along with waist size, body fat composition, waist circumference, blood pressure, cholesterol level and blood sugar.
To calculate your body mass index, you divide your body weight in kilograms by your height in meter squared (commonly expressed as kg/m2), see the body mass index formula below.
To find out about your body mass index (BMI), you can use a FREE online BMI calculators from the Centers for Disease Control and Prevention (CDC) :
- Adults (BMI Calculator Adults. https://www.cdc.gov/healthyweight/assessing/bmi/adult_BMI/english_bmi_calculator/bmi_calculator.html)
For children and adolescents (younger than 20 years of age), overweight and obesity are based on the Centers for Disease Control and Prevention’s (CDC’s) BMI-for-age growth charts, which are available at Centers for Disease Control and Prevention (CDC Clinical Growth Charts https://www.cdc.gov/growthcharts/clinical_charts.htm).
The CDC has a BMI percentile calculator for children and teens at 37.
- Children (BMI Calculator Children. https://www.cdc.gov/healthyweight/bmi/calculator.html)
Body Mass Index for Men and Women Adults
The body mass index is an attempt to quantify the amount of tissue mass (muscle, fat, and bone) in an individual, and then categorize that person as underweight, normal weight, overweight, or obese based on that value. Commonly accepted body mass index ranges are:
A) Underweight: under 18.5 kg/m2,
B) Normal weight: 18.5 to 25 kg/m2,
C) Overweight: 25 to 30 kg/m2,
D) Obese: over 30 to 39.9 kg/m2.
E) Severely Obese: over 40 kg/m2.
Prescription Weight Loss Pills
Weight-loss medicines, weight loss pills or prescription diet pills approved by the Food and Drug Administration (FDA) might be an option for some people.
If you’re not successful at losing 1 pound a week after 6 months of using lifestyle changes, medicines may help. You should only use medicines as part of a program that includes diet, physical activity, and behavioral changes.
Weight-loss medicines might be suitable for adults who are obese (a BMI of 30 or greater). People who have BMIs of 27 or greater, and who are at risk for heart disease and other health conditions like type 2 diabetes or high blood pressure, also may benefit from weight-loss medicines. You’ll still need to focus on diet and exercise while taking these drugs, and they’re not for everyone.
Only orlistat, lorcaserin (Belviq) and phentermine/topiramate (Qsymia) are FDA-approved for long-term use; the others are approved only for short-term use (usually considered ≤12 weeks).
Table 1. Drugs with an FDA-approved indication for obesity
| Generic Name | Trade Name(s) | Mechanism of action | Dosage | Whole-sale price per mo.* | Mean weight change relative to placebo at 1y, kg** | Interactions | Contraindications& | Common Adverse Events& | Cautions and Warnings& |
|---|---|---|---|---|---|---|---|---|---|
| Phenterminea | Adipex-P, Fastin, Oby-Cap, lonamin, Others | Noradrenergic causing appetite suppression | 15– 37.5mg/d | $6– $45 | Not Available | Guanethidine, CNS stimulants, alcohol, tricyclic antidepressants; requirements for insulin or oral hypoglycemic medications may be altered | Pregnancy or nursing, advanced cardiovascular disease, uncontrolled hypertension, hyperthyroidism, glaucoma, agitated states, history of drug abuse, MAOIs | Insomnia, elevation in heart rate, dry mouth, taste alterations, dizziness, tremors, headache, diarrhea, constipation, vomiting, gastro- intestinal distress, anxiety, and restlessness. | Do not increase beyond recommended dose if tolerance to the anorexiant effect develops. Caution prescribing to patients with even mild hypertension. Caution for patients using alcohol or other CNS active drugs or engaging in hazardous activity. |
| Diethylpropiona | Tenuate, Tenuate Dospan, Tepanil | Noradrenergic causing appetite suppression | 25mg 3 times/d or 75mg sustained- release/d | $47– $120 | Not Available | Same as phentermine | Same as phentermine | Same as phentermine | Same as phentermine |
| Phendimetrazineb | Bontril | Noradrenergic | 17.5–70mg 2–3 times/d or 105mg sustained- release/d | $6– $20 | Not Available | Same as phentermine | Same as phentermine | Same as phentermine | Same as phentermine |
| Benzphetamineb | Didrex | Noradrenergic causing appetite suppression | 25–50mg 1–3 times/d | $20– $50 | Not Available | Same as phentermine | Same as phentermine | Same as phentermine | Same as phentermine |
| Orlistatc | Xenical, Alli | Lipase inhibitor causing excretion of ~30% of ingested triglycerides in stool | 60 or 120mg 3 times/d within 1 hr of a fat- containing meals, plus a daily multi- vitamin | For 60mg TID: $45 For 120mg TID: $207 | For 60mg TID:−2.5 kg (−1.5 to −3.5) For 120mg TID: −3.4 kg (−3.2 to −3.6) | Decreased drug concentrations of cyclosporine and levothyroxine. Doses should be temporally separated from orlistat. Fat soluble vitamin absorption is decreased by orlistat | Pregnancy, chronic malabsorption syndromes, cholestasis | Oily Spotting, Flatus with Discharge, Fecal Urgency, Fatty/Oily Stool, Increased Defecation, Fecal Incontinence | Use with caution in those at risk for renal insufficiency, since treatment may increase urinary oxalate. Cholelithiasis and, rarely, severe liver injury including hepatocellular necrosis and acute hepatic failure leading to death, have been reported |
| Lorcaserina | Belviq | Highly selective serotonergic 5-HT2C receptor agonist causing appetite suppression | 10mg two times/d | $240 | −3.2 kg (−2.7 to −3.8) | Triptans, MAOIs including linezolid, SSRIs, SNRIs, dextro- methorphan, tricyclic antidepressants, bupropion, lithium, tramadol, tryptophan, and St. John’s Wort | Pregnancy | Headache, dizziness, fatigue, nausea, dry mouth, cough, and constipation, and back pain, cough and hypoglycemia in patients with type 2 diabetes. | Risk for Serotonin Syndrome or Neuroleptic Malignant Syndrome-like Reactions. Evaluate patients for signs or symptoms of valvular heart disease. Euphoria, hallucination, and dissociation have been seen with supra- therapeutic doses. Use with caution in men at risk for priapism |
| Phentermine / Topiramate-ERa | Qsymia | Noradrenergic + GABA- receptor activator, kainite/AMPA glutamate receptor inhibitor causing appetite suppression | 3.75/23mg /d for 2 weeks, then 7.5/46mg/ d, escalating to a maximum of 15/92mg/d | $140 – $195 | For 7.5/46mgd: −6.7 kg (−5.9 to −7.5) For 15/92mg/d −8.9 kg (−8.3 to −9.4) | Oral contraceptives, alcohol and other CNS depressants, non-potassium- sparing diuretics | Pregnancy, Glaucoma, Hyperthyroidism, MAOIs |
Abbreviations: MAOI = monoamine oxidase inhibitor, CNS = central nervous system, SSRI = selective serotonin-reuptake inhibitors, SNRI = selective serotonin-norepinephrine reuptake inhibitors.
*Reference prices found on March 8, 2013.
**Weight change relative to placebo (95 percentile confidence interval) using intent-to-treat analyses for each medication at 1 year. No studies for older noradrenergic agents (phentermine, diethylpropion, phendimetrazine, and benzphetamine) met inclusion criteria for length of treatment, sample size, and attrition.
a Medications listed on Drug Enforcement Administration Schedule IV are associated with a lower risk of abuse than
b Medications on Schedule III;
c Orlistat is a non-Drug Enforcement Administration scheduled drug.
& Common adverse events for noradrenergic agents include those listed as common in the NIDDK Weight-control Information Network Fact Sheet “Prescription Medications for the Treatment of Obesity”100 as adverse event frequency is not available in the drug package inserts for these agents. For orlistat, lorcaserin, and phentermine/topiramate ER, common adverse events are those listed in the drug package inserts that are reported to occur more frequently than placebo and with more than 5% prevalence.
1) Orlistat (Xenical)
How it works: Blocks your body from absorbing about a third of the fat you eat. Orlistat is a gastrointestinal lipase inhibitor which, when taken three times a day during or up to 1 hour after meals, leads to the excretion of approximately 30% of ingested fat.
Orlistat is available both in prescription (120mg) and over-the-counter (60mg) strength. Orlistat 120mg is FDA-approved for use in adults and adolescents age 12–16years.
When a doctor prescribes orlistat, it’s called Xenical (Orlistat 120mg). If you get it without a prescription, it’s called Alli, which has half of Xenical’s dose. Alli is the reduced-strength, 60-milligram version of orlistat (Xenical) a 120-milligram prescription drug.
The mean weight reduction attributable to orlistat 120mg three times daily at 12 months is modest: among adults participating in behavioral weight control programs and prescribed a lower fat diet (~30% of calories from fat), orlistat-treated patients lost on average 3.4 kg (~3.1% of initial weight) more than placebo-treated participants. The percentage of orlistat 120mg-treated participants who achieved clinically-meaningful (≥5%) weight loss at 1 year varied from 35–73% and the proportion losing ≥10% varied from 14–41%, with both ≥5% and ≥10% weight loss at 1 year significantly greater for orlistat-treated than for placebo-treated participants. At the end of a second year of treatment when a weight-maintenance diet was prescribed, orlistat 120mg-treated participants had lost approximately 3.3 kg (~3.3% of initial weight) more and orlistat 60mg-treated participants had lost approximately 2.5 kg (~2.5% of initial weight) more than those given placebo.
Because of its weight-loss related and weight-loss independent 39 actions, orlistat 120mg treatment is associated with significant improvements in cardiovascular risk factors including decreases in total- and LDL- cholesterol, fasting glucose, and systolic and diastolic blood pressures after 1 year of treatment 40.
Data from the XENDOS trial 41 of 3,305 patients treated for up to 4 years (attrition at 4 years: 48% for orlistat-treated and 66% for placebo-treated) found, in an intention-to-treat approach, that orlistat use decreased body weight over 4 years by 2.7 kg (approximately 2.4% of initial body weight) more than placebo and significantly decreased risk for developing type 2 diabetes from 9.0% with placebo to 6.2% with orlistat. Because orlistat leads to obligate increases in undigested stool triglycerides, it may cause considerable gastrointestinal adverse effects that may be decreased by co-administration of fiber-containing supplements 42.
Side effects include abdominal cramping, passing gas, leaking oily stool, having more bowel movements, and not being able to control bowel movements.
These side effects are generally mild and temporary. But they may get worse if you eat high-fat foods.
These adverse effects may cause patients who do not reduce their fat intake to discontinue therapy. Indeed, despite being FDA-approved in 1999 for indefinite treatment of obesity, among those prescribed orlistat 120mg clinically, fewer than 10% take it for at least 1y and <2% of patients use the medication for 2 years 43.
Rare cases of severe liver injury have been reported in people taking orlistat, but it’s not certain that the drug caused those problems.
What else you should know: You should be on a low-fat diet (less than 30% of your daily calories from fat) before taking orlistat.
Also, take a multivitamin at least 2 hours before or after taking orlistat, because the drug temporarily makes it harder for your body to absorb vitamins A, vitamin D, vitamin E, and vitamin K.
Orlistat is the only drug of its kind that’s approved in the U.S. All other prescription weight loss drugs curb your appetite, including the following.
2) Belviq (Lorcaserin Hydrochloride)
How it works: Curbs your appetite. Lorcaserin is a selective serotonin 2C (5HT2c) receptor agonist that was anticipated to recapitulate the weight loss effects of fenfluramine without its adverse cardiac effects 44. Lorcaserin 10mg twice daily was FDA-approved in 2012 on the basis of two large randomized, placebo-controlled trials in nondiabetic patients (BLOOM n=3182, 50% attrition) 45; BLOSSOM n=4004, 45% attrition) 46, along with a third, smaller trial in adults with type 2 diabetes (BLOOM-DM n=603, 34% attrition) 47. In these trials, participants received low-intensity nutritional and exercise counseling. Lorcaserin decreased body weight modestly, by about 3.2 kg (~3.2% of initial body weight) more than placebo 48. However, significantly more patients treated with lorcaserin 10mg twice daily than placebo lost ≥5% (BLOOM: 47 vs. 20%, BLOSSOM: 47 vs. 25%, BLOOM-DM: 37 vs. 16%) or ≥10% (BLOOM: 23 vs. 8%, BLOSSOM: 23 vs. 10%, BLOOM-DM: 16 vs. 4%) of their initial weight. Reduction in body weight below baseline in the one study 45 with data from participants who took lorcaserin for 2 years had average weight loss of 5.6 kg, versus 2.4 kg among placebo-treated participants. Blood pressure, total cholesterol, LDL”bad” cholesterol, and triglycerides also decreased significantly more in lorcaserin-treated participants 49. Among patients with diabetes, lorcaserin treatment led to lower body weight and improved glycated hemoglobin concentrations.30 Adverse effects (Table 1) include headache, nausea, fatigue, and dizziness 49. Although neither incidence of valvulopathy nor hypertension was statistically greater during lorcaserin than placebo treatment, both were numerically somewhat more prevalent and the FDA has requested that a post-approval trial to assess the long-term cardiovascular effects of lorcaserin be conducted 50.
Side effects: The most common side effects in people who don’t have diabetes are headache, dizziness, nausea, fatigue, dry mouth, and constipation.
The most common side effects in those who have diabetes are low blood sugar (hypoglycemia), headache, back pain, cough, and fatigue.
People taking some depression medications with Belviq need to be monitored very closely for a rare but serious reaction that includes fever and confusion.
Women who are pregnant or planning to get pregnant shouldn’t take Belviq.
What else you should know: If you don’t lose 5% of your weight after 12 weeks of taking Belviq, you should stop taking it, because it’s unlikely to work for you, the FDA says.
3) Contrave
How it works: Contrave is a combination of two FDA-approved drugs, naltrexone and bupropion, in an extended-release formula. Naltrexone is approved to treat alcohol and opioid dependence. Bupropion is approved to treat depression, seasonal affective disorder, and help people stop smoking.
Side effects: The most common side effects include nausea, constipation, headache, vomiting, dizziness, insomnia, dry mouth, and diarrhea. Contrave has a boxed warning about the increased risk of suicidal thoughts and behaviors associated with bupropion. The warning also notes that serious neuropsychiatric issues linked to bupropion have been reported. Contrave can cause seizures and must not be used in patients who have seizure disorders. The drug can also increase blood pressure and heart rate.
What else you should know: If you don’t lose 5% of your weight after 12 weeks of taking Contrave, you should stop taking it, because it’s unlikely to work for you, the FDA says.
4) Saxenda
How it works: Saxenda is a higher dose of the type 2 diabetes drug Victoza. It mimics an intestinal hormone that tells the brain your stomach is full.
Side effects: Nausea, vomiting, diarrhea, constipation, low blood pressure, and increased appetite. Serious side effects can include raised heart rate, pancreatitis, gallbladder disease, kidney problems, and suicidal thoughts.
What else you should know: If you don’t lose 4% of your weight after 16 weeks of taking Saxenda, you should stop taking it, because it’s unlikely to work for you, the FDA says.
5) Phentermine
Your doctor may prescribe this under the names Adipex or Suprenza.
How it works: Curbs your appetite.
Phentermine is by far the most widely prescribed obesity medication in the US, with 25.3 million prescriptions dispensed to an estimated 6.2 million users between 2008–2011 43. Phentermine originally approved indication was obesity; and the drug was used on-label until 1977 when it, along with all other drugs approved for treating obesity, were approved a second time after an amendment to the Food Drug and Cosmetic Act required that the FDA approve new drugs based on efficacy as well as safety. There was protracted opposition to re-approval from those who maintained the sympathomimetic obesity drugs that had dangerous addiction potential. No evidence of addiction had appeared during 18 years of increasingly frequent use, but the FDA re-approved them all, having silenced the opposition by announcing the drugs would be approved for short-term use only 51. The FDA has jurisdiction over pharmaceutical companies but cannot regulate medical practice, jurisdiction over which resides in the individual US states. US physicians treating obesity, well aware of these statuary boundaries, continued to use phentermine and the other sympathomimetic amine anorectic drugs off-label long-term. Surveys of prescribing practices among physicians treating obesity have confirmed that a majority of these physicians continue to prescribe the sympathomimetics off-label in this manner 52.
A meta-analysis of 6 studies ranging from 2 to 24 weeks 53 found that patients using 15–30mg/day phentermine had a mean additional weight loss relative to placebo of 3.6 kg, with mean total weight loss of 6.3 kg. The longest published placebo-controlled trial of phentermine 54 lasted 36 weeks in 108 obese women treated with phentermine 30mg/day either continuously or intermittently (alternating months) and found similar weight loss in the continuous (12.2 kg) and intermittent (13.0 kg) arms vs. 4.8 kg with placebo. However, attrition was 41%, and data were presented only for completers, which is likely to overstate efficacy 54. Among completers, transient symptoms of central nervous system stimulation such as insomnia, irritability, and anxiety did not differ between those receiving continuous (24%) vs. intermittent (27%) therapy, compared with 8% for those taking placebo. Several short-term placebo-controlled studies of phentermine have shown elevations in pulse or smaller decreases in pulse and/or blood pressure than would be expected given the degree of weight loss 55.
Approved for long-term use ? No. It’s approved for short-term use (a few weeks) only.
Side effects can be serious, such as raising your blood pressure or causing heart palpitations, restlessness, dizziness, tremor, insomnia, shortness of breath, chest pain, and trouble doing activities you’ve been able to do.
Phentermine may make you drowsy, hampering your ability to drive or operate machinery. As with some other appetite suppressants, there’s a risk of becoming dependent upon the drug.
Less serious side effects include dry mouth, unpleasant taste, diarrhea, constipation, and vomiting.
Don’t take it late in the evening, as it may cause insomnia.
If you take insulin for diabetes, let your doctor know before you take phentermine, as you may need to adjust your insulin dose.
You should not take phentermine if you have a history of heart disease, stroke, congestive heart failure, or uncontrolled high blood pressure. You also shouldn’t take it if you have glaucoma, hyperthyroidism, or a history of drug abuse, or if you are pregnant or nursing.
What else you should know: Phentermine is an amphetamine. Because of the risk of addiction or abuse, such stimulant drugs are “controlled substances,” which means they need a special type of prescription.
Although there are no published data on the frequency of use, phentermine is occasionally prescribed for patients with label contraindications. Coronary artery disease, stroke, arrhythmias, congestive heart failure, and uncontrolled hypertension are listed specifically. In the absence of controlled data supporting these contraindications, there is no unambiguous evidence that suggests these conditions are absolute contraindications. However, US obesity medicine specialists, based on the known mechanism of action of phentermine, and the pathophysiology of the illness, would consider congestive heart failure, uncontrolled hypertension, untreated clinically significant arrhythmias, and severe advanced coronary artery disease to be absolute contraindications 56. However, in cases of less severe coronary artery disease, medically treated arrhythmias and patients who have past history of stroke, some US obesity medicine specialists would weigh the benefits of weight loss in obese, overweight and overfat subjects after a thorough assessment, that would likely include consultation with the patient’s cardiologist and/or medical specialist 56. This viewpoint is supported by reduced mortality observed in the Sibutramine Cardiovascular Outcomes (SCOUT) trial for patients with cardiovascular disease who had moderate weight loss 57. The expectation is that the same will be discovered with cardiovascular outcome trials that the FDA has mandated for the newer obesity drugs since weight loss in overweight and obese patients induces improvement in cardiac dysfunction common in such patients 58.
Other contraindications include hyperthyroidism, glaucoma and history of drug abuse 56. Most physicians would likely agree that phentermine not be used until hyperthyroidism has been treated but most would also agree a history of successful treatment of hyperthyroidism is not a contraindication. As with other medications that have anticholinergic side effects, phentermine is contraindicated in patients with narrow-angle glaucoma. However, it is not contraindicated in patients with open-angle glaucoma.
Phentermine use in clinical practice has not been associated with phentermine cravings, withdrawal, or excessive use leading to psychological or physical impairment. Although there is a widespread presumption that phentermine abuse is common, actual phentermine abuse is not common and appears limited to the use of the drug as a stimulant among people trying to stay awake and those trying to boost their energy level. Phentermine is longer acting than caffeine and does not have the adverse gastrointestinal effects of high doses of caffeine, so some students studying for exams and some long-haul truck drivers use it to stay awake and alert. Evidently, some patients with stimulant use disorder who use cocaine, methamphetamine or other strong stimulants add phentermine to a drug cocktail in an attempt to heighten the stimulant effects, but no data have been published on the frequency of this practice. A telephone survey of 50 addiction treatment centers in the USA found only 2 instances of patients using such cocktails among several thousand admissions, suggesting the addition of phentermine to such cocktails is uncommon 59. Stimulant use disorder due to phentermine alone as the favored drug has not been described, and no such entity is included in the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) 60. Clinically significant psychiatric distress similar to that described in the DSM-5 in discussing stimulant use disorders has never been observed in overweight or obese patients treated with phentermine. A phentermine withdrawal syndrome has never been observed or described. Patients with either overt attention deficit or some of its symptoms typically “like” phentermine because they function better when taking it. Patients who have difficulty staying in control of eating also “like” phentermine because they have less cravings and enjoy better control of their eating behaviors. Patients with symptoms of attention deficit are quite common among the overweight and obese. For these reasons, “liking” phentermine should not be interpreted as an indication phentermine has high abuse potential. Phentermine treatment enhances control of impulsive behavior and control of other harmful eating behaviors; these are goals of behavioral treatment in obese patients.
It may be reasonable to prescribe phentermine for subjects who have a history of recovery from drug abuse. The author has found this safe in carefully selected patients provided their recovery is genuine, recovery has endured for at least several years and phentermine does not induce phentermine cravings or desire for their former drugs of abuse.
The label also lists contraindications that are absolute and seldom ignored, and these include recent monoamine oxidase inhibitor use, agitated states, pregnancy, nursing mothers and known hypersensitivity or idiosyncratic reactions to sympathetic amines.
Phentermine label warnings
Some consider prescribing phentermine despite specific label warnings to be an off-label use. It is recommended that the label warnings be included in an informed consent, and that any concern of the patient be thoroughly discussed and the discussion documented. The rationale for decisions to prescribe phentermine contrary to a warning would best be explicitly documented in the patient record.
Co-administration with other weight-loss drugs
US physicians view this warning as an anachronism since the FDA has approved the two combination drugs phentermine/topiramate and bupropion/naltrexone 56. The warning against combining weight-loss medicines appeared in the phentermine label in 2000 after 1997 reports of valvulopathy induced by the combination of phentermine and fenfluramine, and both dexfenfluramine and fenfluramine were taken off-market. Surveys of US physicians treating obesity have revealed these physicians frequently combined the various antiobesity drugs with other drugs approved for obesity and with a variety of other drugs that have an effect on weight loss. For example, these physicians combined phentermine and topiramate long before the FDA approved a fixed dose combination (Qsymia) in 2012 56. Other combinations in use are discussed.
Primary pulmonary hypertension
Primary pulmonary hypertension has not been associated with phentermine monotherapy. As discussed in a previous communication, isolated reports of primary pulmonary hypertension occurring in patients who have taken phentermine have relied on theoretical but unproven adverse effects and ignored the underlying incidence of idiopathic pulmonary hypertension 61.
Valvular heart disease
This warning first appeared in the label in 2000 after valvulopathy was discovered in patients taking phentermine and fenfluramine in 1997 and before publication of a report suggesting that fenfluramine, but not phentermine, activated cardiac 5HT2B serotonin receptors that then induced valvulopathy 62. Although the label states “… there have been rare cases of valvular heart disease in patients who reportedly have taken phentermine alone.”
Tolerance
Tolerance refers to a reduced response to a drug after repeated use. Tolerance is a normal physiologic process that occurs with substances of abuse but also with some medicines (e.g., diphenhydramine). Tachyphylaxis refers to very rapid development of tolerance.
Hazardous task ability
Labels for most drugs that work in the central nervous system include this warning. There is no specific evidence that phentermine degrades mental or physical performance. Generally, stimulants will enhance rather than degrade mental and physical performance until extremely high doses are used. The Federal Aviation Administration (FAA) has not accepted phentermine for pilots 61. The only antiobesity drug acceptable to the FAA is orlistat 61.
Risk of abuse and addiction
The Diagnostic and Statistical Manual of Mental Disorders: DSM-5 60 does not have criteria for “addiction” but instead sets forth criteria for diagnosing stimulant-related disorders and discusses “stimulant use disorder” and “stimulant withdrawal” with amphetamine and cocaine as prototypical stimulants. Phentermine is not specifically mentioned in the DSM-5. Stimulant use disorder is defined as a pattern of repeated use leading to clinically significant impairment or distress as manifested by two or more of a list of 11 symptoms, including intense cravings for the substance leading to self-destructive social and job or profession-related behaviors. In phentermine post-marketing studies, we found that long-term phentermine use, even at doses higher than 37.5 mg/day, did not induce phentermine cravings, and that abrupt cessation of long-term phentermine did not induce a stimulant withdrawal syndrome 63.
Assessment of drug “liking” is currently used as a measure of addiction potential 64. Phentermine has been used in studies as an example of a liked drug 65. However, patients taking phentermine often “like” the drug for a variety of valid reasons but do not have other signs or symptoms of addiction or physical dependence. Some like phentermine because they have lost weight taking it and enjoy a better quality of life. Many patients in medical weight management programs have adult attention deficit or at least some symptoms of attention deficit, and many are not taking specific attention medicines. These patients typically are less scattered, can focus better and are more productive when taking phentermine and, realizing this, “like” taking the drug. Discussions of drug liking of phentermine as a sign of addiction typically fail to consider the prevalence of attention deficit in the obese population as a reason for drug liking. Nor do such discussions differentiate between hedonic drug liking versus drug liking because of medical benefits.
Use with alcohol
There is an extensive literature on the effects of ethanol and a wide variety of stimulants in animals and humans. However, there are no specific reports on the effects of combining phentermine and ethanol in humans.
Hypertension
Phentermine-induced increases in blood pressure are often mentioned in both the medical and general literature, but few instances have been recorded 66. US obesity medicine practitioners typically do not prescribe phentermine with poorly controlled hypertension until it is under control, but they do prescribe it in the presence of controlled hypertension, then monitor the patient’s blood pressure closely, discontinuing phentermine if blood pressure rises 61.
6) Diethylpropion
Diethylpropion has a similar adverse-effect and weight loss profile to phentermine, but is much less frequently prescribed, with approximately 1 million prescriptions dispensed between 2008–2011 43. A meta-analysis of 9 small studies ranging from 6–52 weeks 67 found that patients using diethylpropion 75mg/day had a mean additional weight loss relative to placebo of 3.0 kg, with a mean total weight loss of 6.5 kg.
7) Phendimetrazine
Phendimetrazine, despite the paucity of randomized controlled trials 67 is prescribed three times more frequently than diethylpropion for obesity treatment, with more than 3 million phendimetrazine prescriptions estimated to have been filled between 2008–2011 43. In the completer’s analyses from two small 12-week trials 68, phendimetrazine appears to have similar weight loss to other noradrenergic drugs.
8) Benzphetamine
Benzphetamine is less commonly prescribed for obesity treatment than the other noradrenergic drugs 67 and there are few data from controlled trials evaluating its safety or efficacy 67.
Common adverse effects of noradrenergic drugs are shown in Table 1. Because these medications were approved prior to the requirements for long-term trials with adequate power to ascertain clinical endpoints, an adverse effect of noradrenergic obesity drugs on cardiovascular disease events cannot be excluded, and is of concern given their known effect on heart rate and blood pressure.
9) Qsymia
How it works: Curbs your appetite. Qsymia combines phentermine with the seizure/migraine drug topiramate. Topiramate causes weight loss in several ways, including helping you feel full, making foods taste less appealing, and burning more calories.
Phentermine/topiramate-Extended Release (ER) is the first FDA-approved combination drug for obesity, combining low-dose phentermine with a non-standard dose of the antiepileptic medication topiramate-Extended Release (ER).
Phentermine/topiramate-ER, is administered as a once-daily capsule in 4 fixed-dose combinations: 3.75mg phentermine/23mg topiramate (starting dose); 7.5mg phentermine/46mg topiramate (recommended dose); 11.25mg phentermine/69mg topiramate (titration dose); and 15mg phentermine/92mg topiramate (top dose). Dosage is increased over 14 days to 7.5mg phentermine/46mg topiramate, with additional titration to the top dose if weight loss is inadequate 69.
Phentermine/topiramate-ER was recommended for approval based largely on 2 one-year Phase 3 clinical trials (EQUIP, n=1267) 70; CONQUER, n=2487) 71. All groups received a low-intensity lifestyle program. All underwent dose titration over 4 weeks to assigned dose followed by 52 weeks on drug or placebo. EQUIP 70; CONQUER, n=2487) 72 randomized a higher-risk sample of adults with BMI 27–45 kg/m² and ≥2 obesity-associated comorbid conditions, to placebo or phentermine/topiramate-ER. 31% of participants withdrew. One year weight loss was 8.1 kg (7.8%) with the recommended dose and 10.2 kg (9.8%) with the top dose, vs. 1.4 kg (1.2%) with placebo. In addition, 62% (recommended dose) and 70% (top dose) lost ≥5% of initial weight vs. 21% for placebo, with 37%, 48%, and 7% respectively losing ≥10% of initial weight. Many cardiovascular disease risk factors improved with active drug treatment at recommended- or top-dose 73. SEQUEL 74 an extension to CONQUER, followed 78% of CONQUER participants at sites selected for high enrollment and retention and who had completed the initial 56-week trial for a total of 108 weeks. 84% completed their second year of treatment with sustained weight loss of 9.3% and 10.5% at the recommended and top doses, respectively, vs. 1.8% for placebo, and continued differences in many cardiovascular disease risk factors. In addition, there was a significantly lower incidence of progression to type 2 diabetes in the top-dose group (0.9%) vs. placebo (3.7%).
An area of considerable concern, given that most users of obesity medications are women of reproductive age, is the potential for oral clefts in the offspring of women who become pregnant while taking topiramate 75. A risk evaluation and mitigation strategy was developed to minimize the likelihood of pregnancy in women with reproductive potential that includes provider training, dispensing only via certified pharmacies, and supplying patient information regarding risks and the necessity of using effective contraception 76. Women with childbearing potential should have a negative pregnancy test prior to starting phentermine/topiramate-ER and monthly thereafter 76. A small increase in resting heart rate has been observed in the clinical trials of phentermine/topiramate-ER at higher doses, with more patients on top-dose (56.1%) than placebo (42.1%) having increases of more than 10 beats per minute, leading to some concerns regarding its potential long-term effect on cardiovascular disease events. Phentermine/topiramate-ER was approved with a requirement for a post-marketing trial of to assess long-term cardiovascular safety 50. The labeling recommends against prescription in patients with recent or unstable cardiac or cerebrovascular disease, and suggests regular monitoring of resting heart rate 69.
Side effects: The most common side effects are tingling hands and feet, dizziness, altered sense of taste, insomnia, constipation, and dry mouth.
Serious side effects include certain birth defects (cleft lip and cleft palate), faster heart rate, suicidal thoughts or actions, and eye problems that could lead to permanent vision loss if not treated.
Women who might become pregnant should get a pregnancy test before taking Qsymia, and should use birth control and get monthly pregnancy tests while on the drug.
You also shouldn’t take Qsymia if you have glaucoma, hyperthyroidism, heart disease, or stroke. Get regular checks of your heart when starting the drug or increasing the dose.
What else you should know: If you don’t lose 3% of your weight after 12 weeks on Qsymia, the FDA recommends that you stop taking it or that your doctor increase your dose for the next 12 weeks — and if that doesn’t work, you should gradually stop taking it.
Bottom Line
- Most dietary supplements marketed for rapid weight loss, such as acai and hoodia, don’t work for keeping weight off in the long term, and some are dangerous. For example, ephedra, which was used in weight loss supplements, was banned because of unreasonable risk of injury or illness.
- Researchers have studied the weight loss potential of a variety of dietary supplements, including omega-3s and fish oil; chitosan, a dietary fiber from shellfish; green tea extracts; Chinese herbs; and bitter orange (Citrus aurantium) extract. None have been shown to be effective for weight loss, and each of these has side effects.
- There’s some emerging evidence suggesting that some mind and body approaches, such as yoga and meditation, particularly mindful eating, may be useful as complements to other weight-loss interventions.
Safety
- The U.S. Food and Drug Administration (FDA) banned the sale of dietary supplements containing ephedra, which was marketed for weight loss, because of serious health risks, such as cardiovascular complications and even risk of death. Ephedra is also called ma huang.
- Many ephedra-free supplements are now being sold, but side effects of some of their ingredients are similar to the banned products. Some ephedra-free supplements also have a lot of caffeine or herbs, such as guarana, that contain caffeine. The products can cause increased heart rate and abnormal heart rhythms.
- Many dietary supplements marketed for weight-loss (including ones sold as “fat burners” or appetite suppressants) have not been tested for safety.
- What’s on the label may not be what’s in the bottle. Analyses of dietary supplements, including herbal supplements, sometimes find differences between labeled and actual ingredients. Also, the FDA has found weight-loss products tainted with prescription drug ingredients.
- Dietary supplements for weight loss are sometimes misused by people with eating disorders, such as anorexia nervosa or bulimia nervosa, to lose weight or induce vomiting.
- If you’re considering a dietary supplement for weight loss, remember that “natural” does not necessarily mean “safe.”
- Mind and body practices, such as meditation and yoga, are generally considered safe for healthy people when practiced appropriately under the guidance of a well-trained instructor. If you have any underlying health conditions, talk to your health care provider about any complementary approach you may be interested in using.
- Use of nonprescription weight loss products: results from a multistate survey. Blanck HM, Khan LK, Serdula MK. JAMA. 2001 Aug 22-29; 286(8):930-5. https://www.ncbi.nlm.nih.gov/pubmed/11509057/[↩]
- Slimming on the Internet. Miles J, Petrie C, Steel M. J R Soc Med. 2000 May; 93(5):254-7. https://www.ncbi.nlm.nih.gov/pubmed/10884770/[↩]
- Blanck HM, Serdula MK, Gillespie C, et al. Use of Nonprescription Dietary Supplements for Weight Loss is Common among Americans. Journal of the American Dietetic Association. 2007;107(3):441-447. doi:10.1016/j.jada.2006.12.009.[↩]
- Wilson KM, Klein, JD, Sesselberg TS, et al. Use of Complementary Medicine and Dietary Supplements among U.S. Adolescents. Journal of Adolescent Health. 2006;38(4):385-394. doi:10.1016/j.jadohealth.2005.01.010.[↩]
- Pomeranz JL, Barbosa G, Killian C, Austin SB. The Dangerous Mix of Adolescents and Dietary Supplements for Weight Loss and Muscle Building. Journal of Public Health Management and Practice. 2015;21(5):496-503. doi:10.1097/phh.0000000000000142.[↩]
- World J Gastroenterol. 2008 Dec 7; 14(45): 6999–7004. Published online 2008 Dec 7. doi: 10.3748/wjg.14.6999. Hydroxycut hepatotoxicity: A case series and review of liver toxicity from herbal weight loss supplements. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2773866/[↩]
- N Engl J Med 2014; 370:1277-1280. DOI: 10.1056/NEJMp1315559. Hazards of Hindsight — Monitoring the Safety of Nutritional Supplements. http://www.nejm.org/doi/full/10.1056/NEJMp1315559[↩][↩][↩]
- Tainted supplements CDER. Silver Spring, MD: Food and Drug Administration. https://www.accessdata.fda.gov/scripts/sda/sdNavigation.cfm?filter=&sortColumn=1d&sd=tainted_supplements_cder&page=1[↩]
- Cohen PA, Travis JC, Venhuis BJ. A methamphetamine analog (N,α-diethyl-phenylethylamine) identified in a mainstream dietary supplement. Drug Test Anal 2013 October 14 (Epub ahead of print).[↩]
- Pawar RS, Grundel E, Fardin-Kia AR, Rader JI. Determination of selected biogenic amines in Acacia rigidula plant materials and dietary supplements using LC-MS/MS methods. J Pharm Biomed Anal 2014;88:457-466. https://www.ncbi.nlm.nih.gov/pubmed/24176750?dopt=Abstract[↩]
- Acute hepatitis and liver failure following the use of a dietary supplement intended for weight loss or muscle building — May-October 2013. MMWR Morb Mortal Wkly Rep 2013;62:817-819. http://cel.webofknowledge.com/InboundService.do?mode=FullRecord&customersID=atyponcel&IsProductCode=Yes&product=CEL&Init=Yes&Func=Frame&action=retrieve&SrcApp=literatum&SrcAuth=atyponcel&SID=X2BSQGDs8Ua8NrRvdHR&UT=WOS%3A000326450800001[↩]
- Wilson KM, Klein, JD, Sesselberg TS, et al. Use of Complementary Medicine and Dietary Supplements among U.S. Adolescents. Journal of Adolescent Health. 2006;38(4):385-394.[↩]
- Golden NH, Schneider M, Wood C. Preventing Obesity and Eating Disorders in Adolescents. Pediatrics. 2016:138(3). doi:10.1542/peds.2016-1649.[↩]
- Abdel-Rahman A, Anyangwe N, Carlacci L, et al. The Safety and Regulation of Natural Products Used as Foods and Food Ingredients. Toxicological Sciences. 2011;123(2):333-348. doi: 10.1093/toxsci/kfr198.[↩]
- Fong TL, Klontz KC, Canas-Coto A, et al. Hepatotoxicity Due to Hydroxycut: A Case Series. American Journal of Gastroenterology. 2009;105(7):1561-1566. doi: 10.1038/ajg.2010.5.[↩]
- Grundlingh J, Dargan PI, El-Zanfaly M, Wood DM. 2,4-Dinitrophenol (DNP): A Weight Loss Agent with Significant Acute Toxicity and Risk of Death. Journal of Medical Toxicology. 2011;7(3):205-212. doi:10.1007/s13181-011-0162-6.[↩]
- Guyda HJ. Use of Dietary Supplements and Hormones in Adolescents: A Cautionary Tale. Pediatric Child Health. 2005;10(10):587-590.[↩]
- Navarro VJ, Barnhart HX, Bonkovsky HL, et al. The Rising Burden of Herbal and Dietary Supplement Induced Hepatotoxicity in the U.S.A. Program and Abstracts of the 64th Annual Meeting of the American Association for the Study of Liver Diseases; November 1-5, 2013; Washington, DC. Abstract 113.[↩]
- Navarro V, Barnhart H, Bonkovsky H, et al. Herbal and dietary supplement induced hepatotoxicity in the U.S. Gastroenterology. 2012; 142(5 Supp 1): S-41.[↩]
- Cohen, P. A., Benner, C., & McCormick, D. (2012). Use of a pharmaceutically adulterated dietary supplement, Pai You Guo, among Brazilian-born women in the United States. Journal of general internal medicine, 27(1), 51-56.[↩]
- Cohen P, Travis J, Venhuis B. A methamphetamine analog (N,α -diethyl-phenylethylamine) identified in a mainstream dietary supplement: N,α -diethyl-phenylethylamine identified in workout supplement. Drug Testing and Analysis. 2014;6(7-8):805–807. [↩]
- De Carvalho, L. M., Martini, M., Moreira, A. P. L., De Lima, A. P. S., Correia, D., Falcão, T., … & Bohrer, D. (2011). Presence of synthetic pharmaceuticals as adulterants in slimming phytotherapeutic formulations and their analytical determination. Forensic Science International, 204(1), 6-12.[↩]
- Li, N., Hauser, R., Holford, T., Zhu, Y., Zhang, Y., Bassig, B. A., … & Schwartz, S. M. (2015). Muscle-building supplement use and increased risk of testicular germ cell cancer in men from Connecticut and Massachusetts. British journal of cancer, 112(7), 1247-1250.[↩]
- Rahnema C, Crosnoe L, Kim E. Designer steroids – over-the-counter supplements and their androgenic component: Review of an increasing problem. 2015;3(2):150-155.[↩]
- Cohen P, Benner C, McCormick D. Use of a pharmaceutically adulterated dietary supplement, Pai You Guo, among Brazilian-born women in the United States. Journal of General Internal Medicine. 2012;27(1):51–56.[↩]
- Park S, Viray M, Johnston D, et al. Acute hepatitis and liver failure following the use of a dietary supplement intended for weight loss or muscle building – May-October 2013. Morbidity and Mortality Weekly Report. 2013;62(40):817-819.[↩]
- Cohen P, Travis J, Venhuis B. A methamphetamine analog (N,α -diethyl-phenylethylamine) identified in a mainstream dietary supplement: N,α -diethyl-phenylethylamine identified in workout supplement. Drug Testing and Analysis. 2014;6(7-8):805–807.[↩]
- Cohen P, Bloszies C, Yee C, Gerona R. An amphetamine isomer whose efficacy and safety in humans has never been studied, β-methylphenylethylamine (BMPEA), is found in multiple dietary supplements. Drug Testing and Analysis. 2015;8(3-4), 328-333.[↩]
- Cohen P, Travis J, Venhuis B. A synthetic stimulant never tested in humans, 1,3-dimethylbutylamine (DMBA), is identified in multiple dietary supplements: 1,3-dimethylbutylamine (DMBA) in dietary supplements. Drug Testing and Analysis. 2015;7(1): 83–87.[↩]
- Li N, Hauser R, Holford T, et al. Muscle-building supplement use and increased risk of testicular germ cell cancer in men from Connecticut and Massachusetts. British Journal of Cancer. 2015;112:1247-1250.[↩]
- Xiao D, Shi D, Yang D, Barthel B, Koch TH, Yan B. Carboxylesterase-2 is a highly sensitive target of the antiobesity agent orlistat with profound implications in the activation of anticancer prodrugs. Biochemical Pharmacology. 2013;85:439–447.[↩]
- McDuffie JR, Calis KA, Booth SL, Uwaifo GI, Yanovski, JA. Effects of orlistat on fat-soluble vitamins in obese adolescents. 2002;22(7):814-822.[↩]
- Cumella EJ, Hahn J, Woods BK. Weighing Alli’s Impact: Eating disorder patients might be tempted to abuse the first FDA-approved nonprescription diet pill. Behavioral Healthcare. 2007;27:32–34.[↩]
- Maintaining a Healthy Weight On the Go. A Pocket Guide. Available online: https://www.nhlbi.nih.gov/files/docs/public/heart/AIM_Pocket_Guide_tagged.pdf[↩][↩]
- Blackburn G. (1995). Effect of degree of weight loss on health benefits. Obesity Research 3: 211S-216S.[↩]
- NIH, NHLBI Obesity Education Initiative. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. https://www.nhlbi.nih.gov/files/docs/guidelines/ob_gdlns.pdf [↩]
- Centers for Disease Control and Prevention. BMI Percentile Calculator for Child and Teen. https://www.cdc.gov/healthyweight/bmi/calculator.html[↩]
- Yanovski SZ, Yanovski JA. Long-term Drug Treatment for Obesity: A Systematic and Clinical Review. JAMA : the journal of the American Medical Association. 2014;311(1):74-86. doi:10.1001/jama.2013.281361.[↩]
- Cholesterol lowering effect of dietary weight loss and orlistat treatment–efficacy and limitations. Erdmann J, Lippl F, Klose G, Schusdziarra V. Aliment Pharmacol Ther. 2004 Jun 1; 19(11):1173-9. https://www.ncbi.nlm.nih.gov/pubmed/15153170/[↩]
- Long-term changes in blood pressure following orlistat and sibutramine treatment: a meta-analysis. Johansson K, Sundström J, Neovius K, Rössner S, Neovius M. Obes Rev. 2010 Nov; 11(11):777-91. https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0030322/[↩]
- Torgerson JS, Hauptman J, Boldrin MN, Sjostrom L. XENical in the prevention of diabetes in obese subjects (XENDOS) study: a randomized study of orlistat as an adjunct to lifestyle changes for the prevention of type 2 diabetes in obese patients. Diabetes Care. 2004;27(1):155–161 http://care.diabetesjournals.org/content/27/1/155.long[↩]
- Cavaliere H, Floriano I, Medeiros-Neto G. Gastrointestinal side effects of orlistat may be prevented by concomitant prescription of natural fibers (psyllium mucilloid) Int J Obes Relat Metab Disord. 2001;25(7):1095–1099.[↩]
- Use of prescription antiobesity drugs in the United States. Hampp C, Kang EM, Borders-Hemphill V. Pharmacotherapy. 2013 Dec; 33(12):1299-307. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4740913/[↩][↩][↩][↩]
- Connolly HM, Crary JL, McGoon MD, et al. Valvular heart disease associated with fenfluramine-phentermine. N Engl J Med. 1997;337(9):581–588.[↩]
- Smith SR, Weissman NJ, Anderson CM, et al. Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med. 2010;363(3):245–256.[↩][↩]
- Fidler MC, Sanchez M, Raether B, et al. A One-Year Randomized Trial of Lorcaserin for Weight Loss in Obese and Overweight Adults: The BLOSSOM Trial. J Clin Endocrinol Metab. 2011[↩]
- O’Neil PM, Smith SR, Weissman NJ, et al. Randomized Placebo-Controlled Clinical Trial of Lorcaserin for Weight Loss in Type 2 Diabetes Mellitus: The BLOOM-DM Study. Obesity (Silver Spring) 2012;20(7):1426–1436.[↩]
- Chan EW, He Y, Chui CS, Wong AY, Lau WC, Wong IC. Efficacy and safety of lorcaserin in obese adults: a meta-analysis of 1-year randomized controlled trials (RCTs) and narrative review on short-term RCTs. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2013;14(5):383–392.[↩]
- BELVIQ (lorcaserin hydrochloride) tablets, for oral use. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/022529lbl.pdf[↩][↩]
- Colman E, Golden J, Roberts M, Egan A, Weaver J, Rosebraugh C. The FDA’s assessment of two drugs for chronic weight management. The New England journal of medicine. 2012;367(17):1577–1579.[↩][↩]
- Colman E. Anorectics on trial: a half century of federal regulation of prescription appetite suppressants. Ann Intern Med. 2005;143(5):380–385[↩]
- Schmidt SL, Bryman D, Greenway FL, Hendricks EJ. How physician obesity medicine specialists treated obesity before 2012 new drug approvals. Obes Surg. 2015;25(1):186–190. https://www.ncbi.nlm.nih.gov/pubmed/25344465[↩]
- Haddock CK, Poston WS, Dill PL, Foreyt JP, Ericsson M. Pharmacotherapy for obesity: a quantitative analysis of four decades of published randomized clinical trials. Int J Obes Relat Metab Disord. 2002;26(2):262–273[↩]
- Munro JF, MacCuish AC, Wilson EM, Duncan LJ. Comparison of continuous and intermittent anorectic therapy in obesity. Br Med J. 1968;1(5588):352–354. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1984840/pdf/brmedj02070-0040.pdf[↩][↩]
- Ryan DH, Bray GA. Pharmacologic treatment options for obesity: what is old is new again. Curr Hypertens Rep. 2013;15(3):182–189.[↩]
- Hendricks EJ. Off-label drugs for weight management. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 2017;10:223-234. doi:10.2147/DMSO.S95299. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5473499/[↩][↩][↩][↩][↩]
- Caterson ID, Finer N, Coutinho W, et al. Maintained intentional weight loss reduces cardiovascular outcomes: results from the Sibutramine Cardiovascular OUTcomes (SCOUT) trial. Diabetes Obes Metab. 2012;14(6):523–530.[↩]
- de las Fuentes L, Waggoner AD, Mohammed BS, et al. Effect of moderate diet-induced weight loss and weight regain on cardiovascular structure and function. J Am Coll Cardiol. 2009;54(25):2376–2381.[↩]
- Bryman D, Hendricks EJ, Schmidt SL. Incidence of addiction and abuse due to phentermine, diethylpropion, and phendimetrazine in the United States. Telephone Survey of Emergency Rooms and Addiction Treatment Centers ed. Unpublished manuscript. 2013[↩]
- American Psychiatric Association, DSM-5 Task Force . Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th ed. American Psychiatric Association: Arlington, VA; 2013.[↩][↩]
- Hendricks EJ, Rothman RB. RE: pulmonary hypertension associated with use of phentermine? Yonsei Med J. 2011;52(5):869–870 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3159930/[↩][↩][↩][↩]
- Rothman RB, Baumann MH, Savage JE, et al. Evidence for possible involvement of 5-HT(2B) receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications. Circulation. 2000;102(23):2836–2841.[↩]
- Hendricks EJ, Greenway FL. A study of abrupt phentermine cessation in patients in a weight management program. Am J Ther. 2011;18(4):292–299.[↩]
- Berridge KC, Robinson TE. Liking, wanting, and the incentive-sensitization theory of addiction. Am Psychol. 2016;71(8):670–679.[↩]
- Schoedel KA, Addy C, Chakraborty B, et al. Human abuse potential and cognitive effects of taranabant, a cannabinoid 1 receptor inverse agonist: a randomized, double-blind, placebo- and active-controlled, crossover study in recreational polydrug users. J Clin Psychopharmacol. 2012;32(4):492–502.[↩]
- Hendricks EJ, Rothman RB. Phentermine therapy for obesity does not elevate blood pressure. Diabetes Obes Metab. 2011;13(10):963–964. https://www.ncbi.nlm.nih.gov/pubmed/21896124[↩]
- Pharmacotherapy for obesity: a quantitative analysis of four decades of published randomized clinical trials. Haddock CK, Poston WS, Dill PL, Foreyt JP, Ericsson M. Int J Obes Relat Metab Disord. 2002 Feb; 26(2):262-73. https://www.ncbi.nlm.nih.gov/pubmed/11850760/[↩][↩][↩][↩]
- Sustained-action phendimetrazine in obesity. Hadler AJ. J Clin Pharmacol J New Drugs. 1968 Mar-Apr; 8(2):113-7. https://www.ncbi.nlm.nih.gov/pubmed/4871210/[↩]
- Qsymia (phentermine and topiramate extended-release) capsules, for oral use. https://www.accessdata.fda.gov/drugsatfda_docs/label/2013/022580s004lbl.pdf[↩][↩]
- Allison DB, Gadde KM, Garvey WT, et al. Controlled-release phentermine/topiramate in severely obese adults: a randomized controlled trial (EQUIP) Obesity (Silver Spring) 2012;20(2):330–342. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3270297/[↩][↩]
- Gadde KM, Allison DB, Ryan DH, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet. 2011;377(9774):1341–1352. https://www.ncbi.nlm.nih.gov/pubmed/21481449[↩]
- Gadde KM, Allison DB, Ryan DH, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight a randomized adults without diabetes and with BMI ≥35 kg/m² to placebo, phentermine/topiramate-ER 3.75/23mg (starting dose), or 15/92mg (top dose). 40% of participants withdrew. At the top dose, mean 1 year weight loss was 10.9% vs. 1.6% of initial weight for placebo. 67% of patients given the top dose lost ≥5% of initial weight and 47% lost ≥10% of initial weight, compared with 17% and 7%, respectively for placebo. CONQUER ((Gadde KM, Allison DB, Ryan DH, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet. 2011;377(9774):1341–1352. https://www.ncbi.nlm.nih.gov/pubmed/21481449[↩]
- Davidson MH, Tonstad S, Oparil S, Schwiers M, Day WW, Bowden CH. Changes in cardiovascular risk associated with phentermine and topiramate extended-release in participants with comorbidities and a body mass index >/=27 kg/m(2) The American journal of cardiology. 2013;111(8):1131–1138.[↩]
- Garvey WT, Ryan DH, Look M, et al. Two-year sustained weight loss and metabolic benefits with controlled-release phentermine/topiramate in obese and overweight adults (SEQUEL): a randomized, placebo-controlled, phase 3 extension study. Am J Clin Nutr. 2012;95(2):297–308. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3260065/[↩]
- Margulis AV, Mitchell AA, Gilboa SM, et al. Use of topiramate in pregnancy and risk of oral clefts. American journal of obstetrics and gynecology. 2012;207(5):405, e401–407. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3484193/[↩]
- QSYMIA (phentermine and topiramate extended-release) Capsules. Risk evaluation and mitigation strategy (REMS). https://www.fda.gov/downloads/forindustry/userfees/prescriptiondruguserfee/ucm361075.pdf[↩][↩]