Contents
What are polyphenols
Polyphenols are naturally occurring compounds such as phenolic acids, flavonoids, stilbenes, and lignans found largely in the fruits, vegetables, cereals and beverages 1, 2, 3, 4, 5, 6. Fruits like grapes, apple, pear, cherries and berries contains up to 200–300 mg polyphenols per 100 grams fresh weight 7. The products manufactured from these fruits, also contain polyphenols in significant amounts. Cereals, dry legumes, chocolate and beverages, such as tea, coffee, or wine also contribute to the polyphenolic intake 8, 9. Typically a glass of red wine or a cup of tea or coffee contains about 100 mg polyphenols.
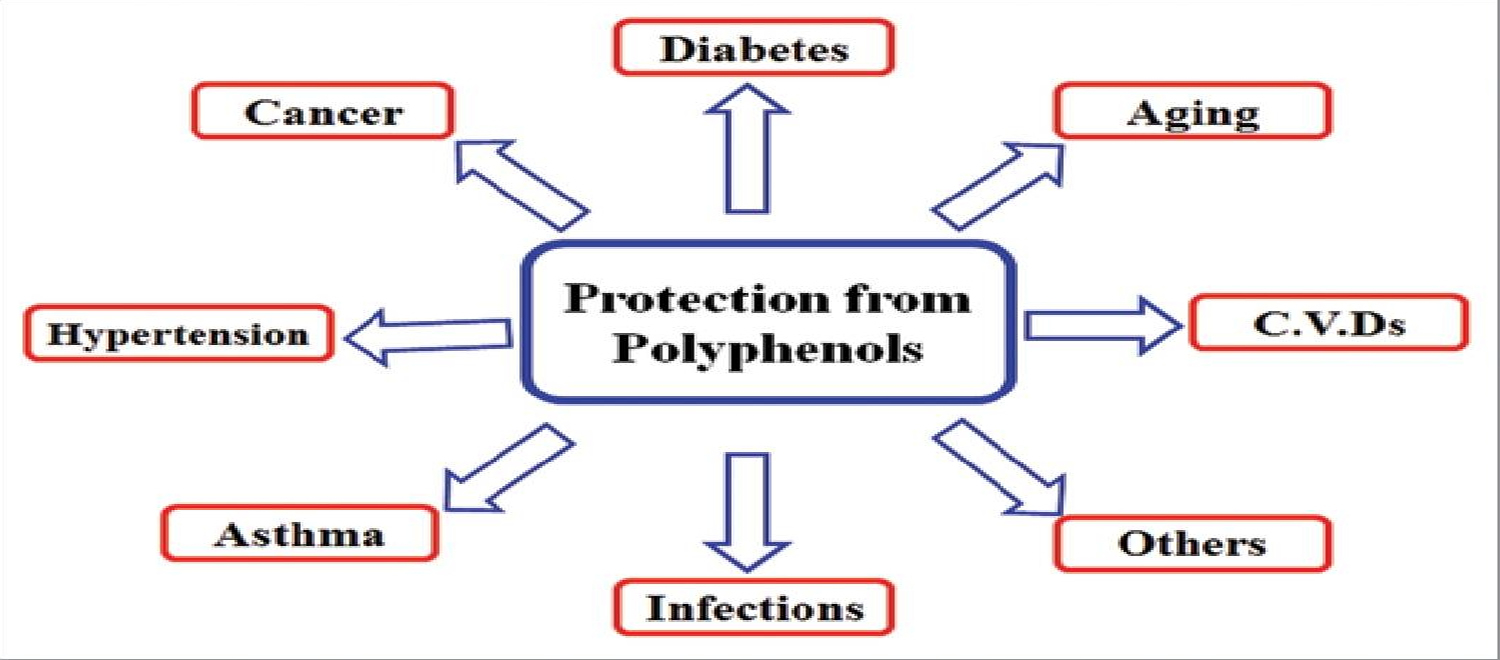
Polyphenols are secondary metabolites of plants and are generally involved in defense against ultraviolet radiation or aggression by pathogens and may also contribute to the bitterness, astringency of the food. Researchers have explored that these molecules are very good antioxidants and may neutralize the destructive reactivity of undesired reactive oxygen/nitrogen species produced as byproduct during metabolic processes in the body. Epidemiological studies have revealed that polyphenols provide a significant protection against development of several chronic diseases such as cardiovascular diseases (CVDs), cancer, diabetes, infections, aging, asthma, inflammation, antimicrobial, cytotoxic activities and prevent neurodegenerative conditions 3, 10, 11, 12, 13, 7, 14, 15, 16, 17, 18, 19, 20, 21.
Towards the end of 20th century, epidemiological studies and associated meta-analyses strongly suggested that long term consumption of diets rich in plant polyphenols offered some protection against development of cancers, cardiovascular diseases, diabetes, osteoporosis and neurodegenerative diseases 22, 23. Whether acting in the gastrointestinal tract or in the liver, the potent antioxidant effects of polyphenols are widely accepted as health promoting 24, 25. Antivira, antibacterial, anti-inflammatory, neuroprotective, and anticarcinogenic effects have also been attributed to polyphenols 26, 27, 28, 29. Polyphenols and other food phenolics are the subject of increasing scientific interest because of their possible beneficial effects on human health.
Polyphenols such as resveratrol, epigallocatechin gallate (EGCG), and curcumin have been acknowledged for having beneficial effects on cardiovascular health, while some have also been shown to be protective in aging.
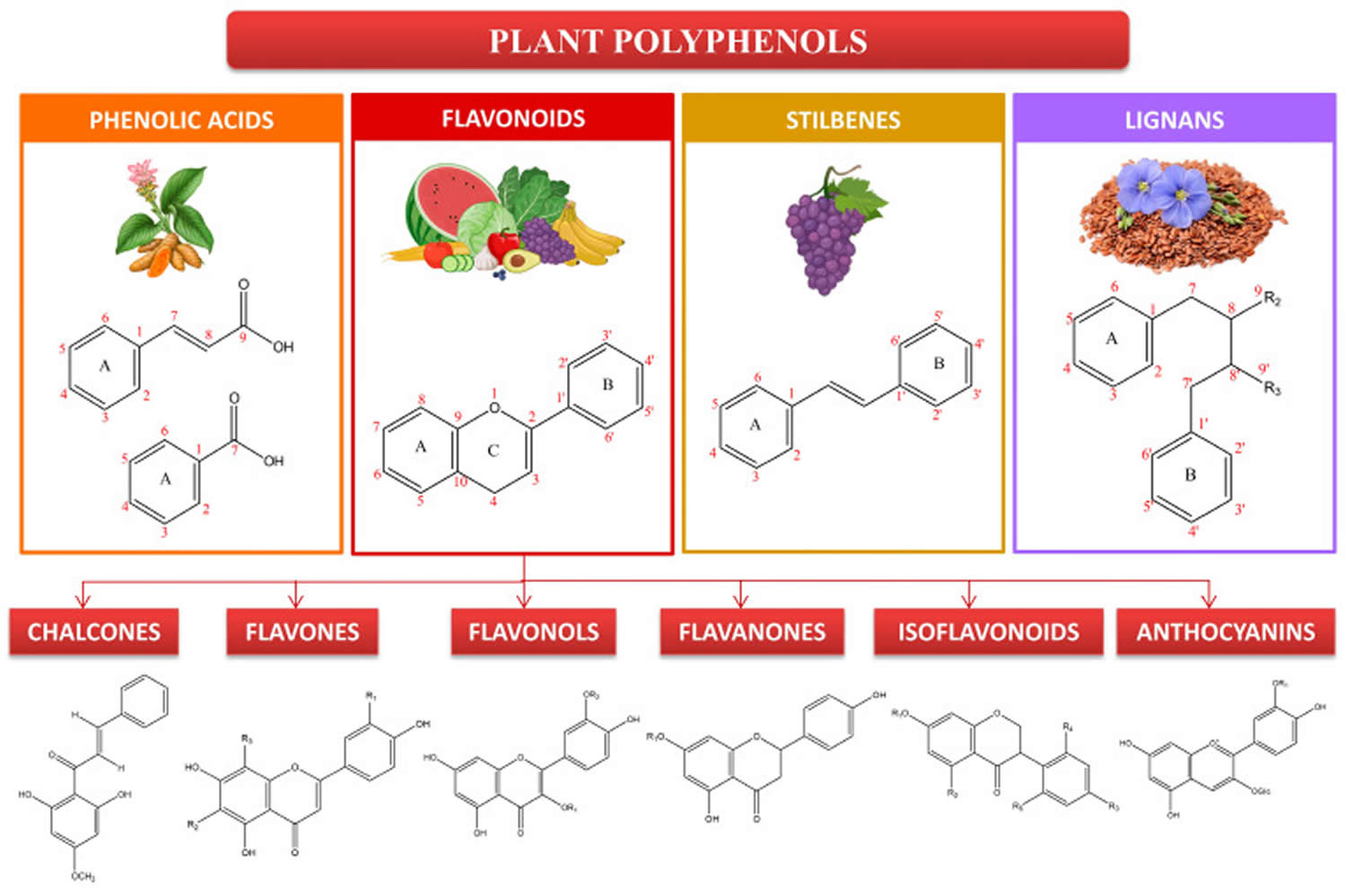
Figure 1. Polyphenols
Footnote: Main categories of polyphenols. Polyphenols are classified as phenolic acids, flavonoids, stilbenes, and lignans based on their structure and the numbering of carbons.
[Source 2 ]Figure 2. Polyphenols Health Benefits
Structure and Classes of Polyphenols
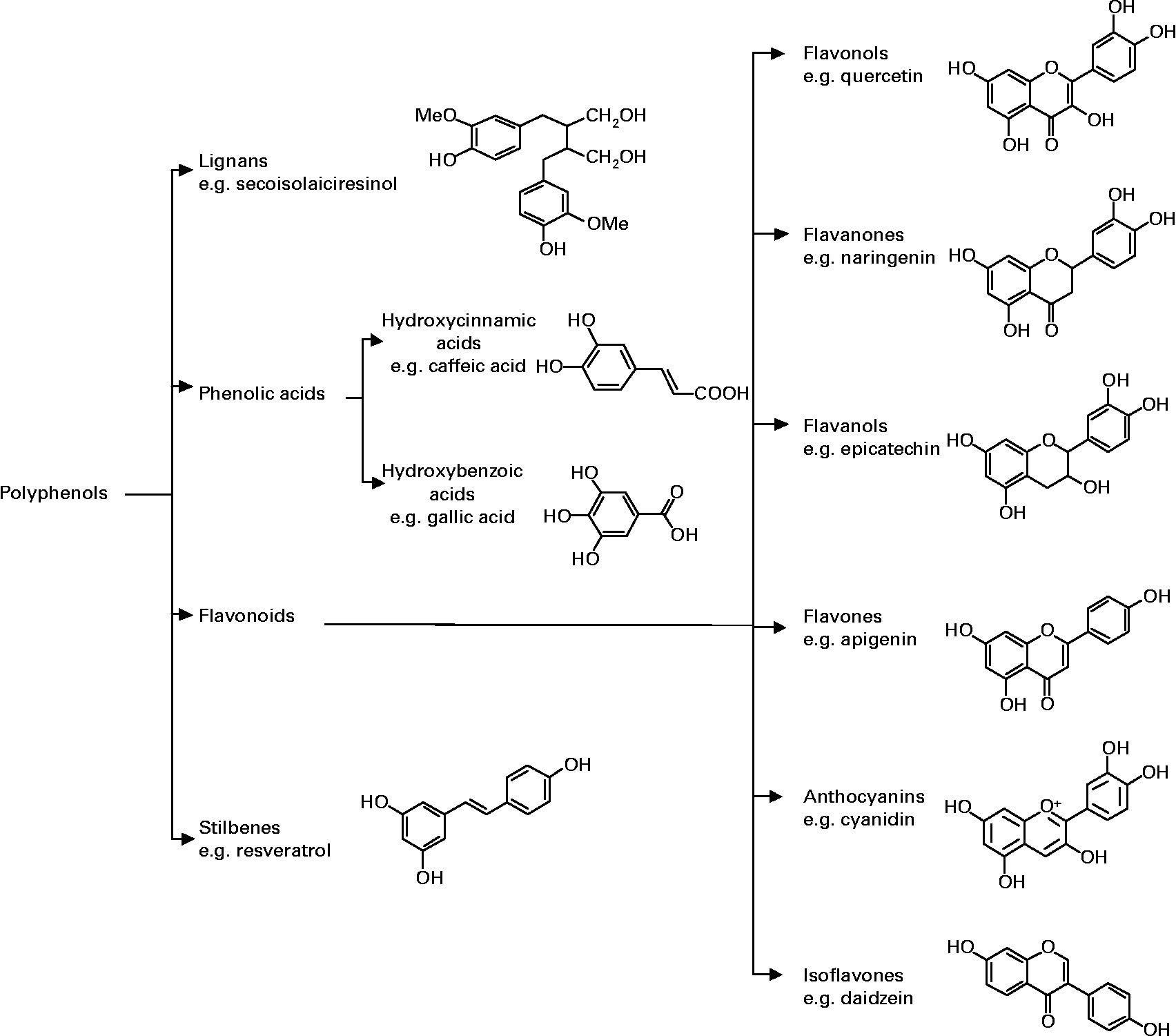
More than 8,000 polyphenolic compounds have been identified in various plant species. All plant phenolic compounds arise from a common intermediate, phenylalanine, or a close precursor, shikimic acid. Primarily they occur in conjugated forms, with one or more sugar residues linked to hydroxyl groups, although direct linkages of the sugar (polysaccharide or monosaccharide) to an aromatic carbon also exist. Association with other compounds, like carboxylic and organic acids, amines, lipids and linkage with other phenol is also common 30.
Chemical structures of the different classes of polyphenols. Polyphenols are classified on the basis of the number of phenol rings that they contain and of the structural elements that bind these rings to one another 7. They are broadly dived in four classes; Phenolic acids, flavonoids, stilbenes and lignans 9. Phenolic acids are further divided into hydroxyl benzoic and hydroxyl cinnamic acids. Phenolic acids account for about a third of the polyphenolic compounds in our diet and are found in all plant material, but are particularly abundant in acidic-tasting fruits. Caffeic acid, gallic acid, ferulic acid are some common phenolic acids. Flavonoids are most abundant polyphenols in human diet and share a common basic structure consist of two aromatic rings, which are bound together by three carbon atoms that form an oxygenated heterocycle 7. Biogenetically, one ring usually arises from a molecule of resorcinol, and other ring is derived from the shikimate pathway. Stilbenes contain two phenyl moieties connected by a twocarbon methylene bridge. Most stilbenes in plants act as antifungal phytoalexins, compounds that are synthesized only in response to infection or injury. The most extensively studied stilbene is resveratrol. Lignans are diphenolic compounds that contain a 2,3-dibenzylbutane structure that is formed by the dimerization of two cinnamic acid residues. Figure 2 illustrates the different groups of polyphenols and their chemical structures.
Figure 3. Polyphenols Chemical Structure
Flavonoids
Flavonoids are the most researched and known class of polyphenols. Flavonoids in foods are divided into 6 different classes which are named as flavanols, flavonols, isoflavones, flavanones, flavones, and anthocyanidins 32, 33. This class of polyphenols can be classified into a number of subgroups depending on the oxidation state of the central carbon. More than 4,000 varieties of flavonoids have been identified, many of which are responsible for the attractive colors, taste, and aroma of the flowers, fruits and leaves 32. Flavonoids, which play a complementary role in plant metabolism, facilitate growth, and also function in protective mechanisms 34, 35. Moreover, flavonoids has an important function in reducing radical oxygen species (ROS) formation in plants under stress, as they can regulate the activity of ROS. Threfore, flavonoids contribute to a variety of potential bioactive activities that improve health in humans, including antioxidant, anti-inflammatory, and protective effects against cardiovascular diseases, and neuroprotective, anticancer, and anti-aging roles 36, 37, 38, 39, 40 .
Flavonoids are generally present at relatively low concentrations of ≈15–30 mg/kg fresh weight. The richest sources are onions (up to 1.2 g/kg fresh weight), curly kale, leeks, broccoli, and blueberries (Table 2). Red wine and tea also contain up to 45 mg flavonols/L. These compounds are present in glycosylated forms. The associated sugar moiety is very often glucose or rhamnose, but other sugars may also be involved (eg, galactose, arabinose, xylose, glucuronic acid). Fruit often contains between 5 and 10 different flavonol glycosides. These flavonols accumulate in the outer and aerial tissues (skin and leaves) because their biosynthesis is stimulated by light. Marked differences in concentration exist between pieces of fruit on the same tree and even between different sides of a single piece of fruit, depending on exposure to sunlight 41. Similarly, in leafy vegetables such as lettuce and cabbage, the glycoside concentration is ≥10 times as high in the green outer leaves as in the inner light-colored leaves 42. This phenomenon also accounts for the higher flavonol content of cherry tomatoes than of standard tomatoes, because they have different proportions of skin to whole fruit.
Flavones are much less common than flavonols in fruit and vegetables. Flavones consist chiefly of glycosides of luteolin and apigenin. The only important edible sources of flavones identified to date are parsley and celery (Table 2). Cereals such as millet and wheat contain C-glycosides of flavones. The skin of citrus fruit contains large quantities of polymethoxylated flavones: tangeretin, nobiletin, and sinensetin (up to 6.5 g/L of essential oil of mandarin). These polymethoxylated flavones are the most hydrophobic flavonoids.
In human foods, flavanones are found in tomatoes and certain aromatic plants such as mint, but they are present in high concentrations only in citrus fruit. The main aglycones are naringenin in grapefruit, hesperetin in oranges, and eriodictyol in lemons. Flavanones are generally glycosylated by a disaccharide at position 7: either a neohesperidose, which imparts a bitter taste (such as to naringin in grapefruit), or a rutinose, which is flavorless. Orange juice contains between 200 and 600 mg hesperidin/L and 15–85 mg narirutin/L, and a single glass of orange juice may contain between 40 and 140 mg flavanone glycosides. Because the solid parts of citrus fruit, particularly the albedo (the white spongy portion) and the membranes separating the segments, have a very high flavanone content, the whole fruit may contain up to 5 times as much as a glass of orange juice.
Isoflavones are flavonoids with structural similarities to estrogens. Although they are not steroids, they have hydroxyl groups in positions 7 and 4 in a configuration analogous to that of the hydroxyls in the estradiol molecule. This confers pseudohormonal properties on them, including the ability to bind to estrogen receptors, and they are consequently classified as phytoestrogens. Isoflavones are found almost exclusively in leguminous plants. Soya and its processed products are the main source of isoflavones in the human diet. They contain 3 main molecules: genistein, daidzein, and glycitein, generally in a concentration ratio of 1:1:0.2. They are sensitive to heat and are often hydrolyzed to glycosides during the course of industrial processing, as in the production of soya milk (36). The fermentation carried out during the manufacturing of certain foods, such as miso and tempeh, results in the hydrolysis of glycosides to aglycones. The aglycones are highly resistant to heat. The isoflavone content of soya and its manufactured products varies greatly as a function of geographic zone, growing conditions, and processing. Soybeans contain between 580 and 3800 mg isoflavones/kg fresh wt, and soymilk contains between 30 and 175 mg/L.
Flavanols exist in both the monomer form (catechins) and the polymer form (proanthocyanidins). Catechins are found in many types of fruit (apricots, which contain 250 mg/kg fresh wt, are the richest source; Table 2). They are also present in red wine (up to 300 mg/L), but green tea and chocolate are by far the richest sources. An infusion of green tea contains up to 200 mg catechins. Black tea contains fewer monomer flavanols, which are oxidized during “fermentation” (heating) of tea leaves to more complex condensed polyphenols known as theaflavins (dimers) and thearubigins (polymers). Catechin and epicatechin are the main flavanols in fruit, whereas gallocatechin, epigallocatechin, and epigallocatechin gallate are found in certain seeds of leguminous plants, in grapes, and more importantly in tea. In contrast to other classes of flavonoids, flavanols are not glycosylated in foods. The tea epicatechins are remarkably stable when exposed to heat as long as the pH is acidic: only ≈15% of these substances are degraded after 7 h in boiling water at pH 5.
Proanthocyanidins, which are also known as condensed tannins, are dimers, oligomers, and polymers of catechins that are bound together.Through the formation of complexes with salivary proteins, condensed tannins are responsible for the astringent character of fruit (grapes, peaches, kakis, apples, pears, berries, etc) and beverages (wine, cider, tea, beer, etc) and for the bitterness of chocolate. This astringency changes over the course of maturation and often disappears when the fruit reaches ripeness; this change has been well explained in the kaki fruit by polymerization reactions with acetaldehyde. Such polymerization of tannins probably accounts for the apparent reduction in tannin content that is commonly seen during the ripening of many types of fruit. It is difficult to estimate the proanthocyanidin content of foods because proanthocyanidins have a wide range of structures and molecular weights. The only available data concern dimers and trimers, which are as abundant as the catechins themselves.
Anthocyanins are pigments dissolved in the vacuolar sap of the epidermal tissues of flowers and fruit, to which they impart a pink, red, blue, or purple color. They exist in different chemical forms, both colored and uncolored, according to pH. Although they are highly unstable in the aglycone form (anthocyanidins), while they are in plants, they are resistant to light, pH, and oxidation conditions that are likely to degrade them. Degradation is prevented by glycosylation, generally with a glucose at position 3, and esterification with various organic acids (citric and malic acids) and phenolic acids. In addition, anthocyanins are stabilized by the formation of complexes with other flavonoids (copigmentation). In the human diet, anthocyanins are found in red wine, certain varieties of cereals, and certain leafy and root vegetables (aubergines, cabbage, beans, onions, radishes), but they are most abundant in fruit. Cyanidin is the most common anthocyanidin in foods. Food contents are generally proportional to color intensity and reach values up to 2–4 g/kg fresh wt in blackcurrants or blackberries (Table 2). These values increase as the fruit ripens. Anthocyanins are found mainly in the skin, except for certain types of red fruit, in which they also occur in the flesh (cherries and strawberries). Wine contains ≈200–350 mg anthocyanins/L, and these anthocyanins are transformed into various complex structures as the wine ages.
Phenolic Acids
Phenolic acids are found abundantly in foods and divided into two classes: derivatives of benzoic acid and derivatives of cinnamic acid. The hydroxybenzoic acid content of edible plants is generally low, with the exception of certain red fruits, black radish and onions, which can have concentrations of several tens of milligrams per kilogram fresh weight 43. The hydroxycinnamic acids are more common than hydroxybenzoic acids and consist chiefly of p-coumaric, caffeic, ferulic and sinapic acids.
Caffeic acid, both free and esterified, is generally the most abundant phenolic acid and represents between 75% and 100% of the total hydroxycinnamic acid content of most fruit. Hydroxycinnamic acids are found in all parts of fruit, although the highest concentrations are seen in the outer parts of ripe fruit. Concentrations generally decrease during the course of ripening, but total quantities increase as the fruit increases in size.
Ferulic acid is the most abundant phenolic acid found in cereal grains, which constitute its main dietary source. The ferulic acid content of wheat grain is ≈0.8–2 g/kg dry wt, which may represent up to 90% of total polyphenols. Ferulic acid is found chiefly in the outer parts of the grain. The aleurone layer and the pericarp of wheat grain contain 98% of the total ferulic acid. The ferulic acid content of different wheat flours is thus directly related to levels of sieving, and bran is the main source of polyphenols 44. Rice and oat flours contain approximately the same quantity of phenolic acids as wheat flour (63 mg/kg), although the content in maize flour is about 3 times as high. Ferulic acid is found chiefly in the trans form, which is esterified to arabinoxylans and hemicelluloses in the aleurone and pericarp. Only 10% of ferulic acid is found in soluble free form in wheat bran. Several dimers of ferulic acid are also found in cereals and form bridge structures between chains of hemicellulose.
Tea is an important source of gallic acid: tea leaves may contain up to 4.5 g/kg fresh wt 45. Furthermore, hydroxybenzoic acids are components of complex structures such as hydrolyzable tannins (gallotannins in mangoes and ellagitannins in red fruit such as strawberries, raspberries, and blackberries) 46. Because these hydroxybenzoic acids, both free and esterified, are found in only a few plants eaten by humans, they have not been extensively studied and are not currently considered to be of great nutritional interest.
The hydroxycinnamic acids are more common than are the hydroxybenzoic acids and consist chiefly of p-coumaric, caffeic, ferulic, and sinapic acids. These acids are rarely found in the free form, except in processed food that has undergone freezing, sterilization, or fermentation. The bound forms are glycosylated derivatives or esters of quinic acid, shikimic acid, and tartaric acid. Caffeic and quinic acid combine to form chlorogenic acid, which is found in many types of fruit and in high concentrations in coffee: a single cup may contain 70–350 mg chlorogenic acid 47. The types of fruit having the highest content (blueberries, kiwis, plums, cherries, apples) contain 0.5–2 g hydroxycinnamic acids/kg fresh wt (Table 2) 48.
Stilbenes
Stilbenes are found in only low quantities in the human diet 49. One of these, resveratrol, for which anticarcinogenic effects have been shown during screening of medicinal plants and which has been extensively studied, is found in low quantities in wine (0.3–7 mg aglycones/L and 15 mg glycosides/L in red wine) and naturally present in grapes and peanuts 33, 50, 32. However, because resveratrol is found in such small quantities in the diet, any protective effect of this molecule is unlikely at normal nutritional intakes 51. Most stilbenes in plants act as antifungal phytoalexins, compounds that are synthesized only in response to infection or injury. One of the best studied, naturally occurring polyphenol stilbene is resveratrol (3,4′,5-trihydroxystilbene), found largely in grapes. A product of grapes, red wine also contains significant amount of resveratrol 52, 53.
Lignans
Furans, furofurans, arylnaphthalenes, aryltetralins, dibenzylbutanes, dibenzylbutyrolactones, dibenzocyclooctadienes, and dibenzybutyrolactols are the subgroups of lignans 32. The richest dietary source of lignans is linseed (flaxseed), which contains secoisolariciresinol (up to 3.7 g/kg dry weight) and low quantities of matairesinol 54, 33, 55. Other cereals, grains, fruit, legumes, and certain vegetables also contain traces of these same lignans, but concentrations in linseed are ≈1000 times as high as concentrations in these other food sources 56. Lignans are metabolized to enterodiol and enterolactone by the intestinal microflora. The low quantities of secoisolariciresinol and matairesinol that are ingested as part of normal diet do not account for the concentrations of the metabolites enterodiol and enterolactone that are classically measured in plasma and urine. Therefore, there are undoubtedly other lignans of plant origin that are precursors of enterodiol and enterolactone and that have not yet been identified. Thompson et al 57 confirmed that oleaginous seeds (linseed/flaxseed) are the richest source and identified algae, leguminous plants (lentils), cereals (triticale and wheat), vegetables (garlic, asparagus, carrots), and fruit (pears, prunes) as minor sources. Lignans-rich diet may be beneficial to human health 56. An especially favorable effect of lignans is anticancer activity 58. Lignans may control or prevent cancer by many different mechanisms and there are test tube studies about the anticarcinogenic effect of pure lignans 59, 60, 61, 62, 63, 64, 65. Additionally and apart from that, lignans possess anti-inflammatory, antioxidant, anti-menopausal (protective role form cardiovascular, bone, and other psychological disorders), and antimicrobial properties 66, 67, 68.
Foods high in Polyphenols
In general, the total intake of polyphenols is approximated at 1 g/individual/day and polyphenols are considered by many to be the major source of antioxidants in our diet 69, 70, 71. However, this estimate varies depending on the type of diet. For example, total polyphenol intake in the Finnish diet is 817–919 mg/individual/day 72. In the Vietnamese diet, it is 595 mg/individual/day 73 and in the Mediterranean diet, polyphenol intake ranges between 1800 and 3000 mg/individual/day 74. Still, and due to their low absorption, it has been suggested that their major sites of antioxidant activity are the stomach 75 and the intestine 76.
Fruit and beverages such as tea and red wine constitute the main sources of polyphenols. Certain polyphenols such as quercetin are found in all plant products (fruit, vegetables, cereals, leguminous plants, fruit juices, tea, wine, infusions, etc), whereas others are specific to particular foods (flavanones in citrus fruit, isoflavones in soya, phloridzin in apples). In most cases, foods contain complex mixtures of polyphenols, which are often poorly characterized. Apples, for example, contain flavanol monomers (epicatechin mainly) or oligomers (procyanidin B2 mainly), chlorogenic acid and small quantities of other hydroxycinnamic acids, 2 glycosides of phloretin, several quercetin glycosides, and anthocyanins such as cyanidin 3-galactoside in the skin of certain red varieties. Apples are one of the rare types of food for which fairly precise data on polyphenol composition are available. Differences in polyphenol composition between varieties of apples have notably been studied. The polyphenol profiles of all varieties of apples are practically identical, but concentrations may range from 0.1 to 5 g total polyphenols/kg fresh wt and may be as high as 10 g/kg in certain varieties of cider apples 77, 78.
Distribution of phenolics in plants at the tissue, cellular and sub cellular levels is not uniform. Insoluble phenolics are found in cell walls, while soluble phenolics are present within the plant cell vacuoles 79. Certain polyphenols like quercetin are found in all plant products; fruit, vegetables, cereals, fruit juices, tea, wine, infusions etc., whereas flavanones and isoflavones are specific to particular foods. In most cases, foods contain complex mixtures of polyphenols. The outer layers of plants contain higher levels of phenolics than those located in their inner parts 80.
Numerous factors affect the polyphenol content of plants, these include degree of ripeness at the time of harvest, environmental factors, processing and storage. Polyphenolic content of the foods are greatly affected by environmental factors as well as edaphic factors like soil type, sun exposure, rainfall etc. The degree of ripeness considerably affects the concentrations and proportions of various polyphenols 81. In general, it has been observed that phenolic acid content decreases during ripening, whereas anthocyanin concentrations increase. Many polyphenols, especially phenolic acids, are directly involved in the response of plants to different types of stress: they contribute to healing by lignifications of damaged areas possess antimicrobial properties, and their concentrations may increase after infection 82.
Table 1. Main factors affecting the bioavailability of dietary polyphenols in humans
| External factors | Environmental factors (i.e., sun exposure, degree of ripeness); food availability |
|---|---|
| Food processing related factors | Thermal treatments; homogenization; liophylization; cooking and methods of culinary preparation; storage |
| Food related factors | Food matrix; presence of positive or negative effectors of absorption (i.e., fat, fiber) |
| Interaction with other compounds | Bonds with proteins (i.e., albumin) or with polyphenols with similar mechanism of absorption |
| Polyphenols related factors | Chemical structure; concentration in food; amount introduced |
| Host related factors | Intestinal factors (i.e., enzyme activity; intestinal transit time; colonic microflora). Systemic factors (i.e., gender and age; disorders and/or pathologies; genetics; physiological condition) |
Methods of culinary preparation also have a marked effect on the polyphenol content of foods. For example, simple peeling of fruit and vegetables can eliminate a significant portion of polyphenols because these substances are often present in higher concentrations in the outer parts than in the inner parts. Cooking may also have a major effect. Onions and tomatoes lose between 75% and 80% of their initial quercetin content after boiling for 15 min, 65% after cooking in a microwave oven, and 30% after frying 84. Steam cooking of vegetables, which avoids leaching, is preferable. Potatoes contain up to 190 mg chlorogenic acid/kg, mainly in the skin 85. Extensive loss occurs during cooking, and no remaining phenolic acids were found in French fries or freeze-dried mashed potatoes 86.
Industrial food processing also affects polyphenol content. As with fruit peeling, dehulling of legume seeds and decortication and bolting of cereals can result in a loss of some polyphenols. Grinding of plant tissues may lead to oxidative degradation of polyphenols as a result of cellular decompartmentation and contact between cytoplasmic polyphenol oxidase and phenolic substrates present in the vacuoles. Polyphenols are then transformed into brown pigments that are polymerized to different degrees. This unwanted process can occur, for example, during the process of making jam or compote from fruit. Production of fruit juice often involves clarification or stabilization steps specifically aimed at removing certain flavonoids responsible for discoloration and haze formation. Manufactured fruit juices thus have low flavonoid content. The pectinolytic enzymes used during such processing also hydrolyze the esters of hydroxycinnamic acid 87. Conversely, maceration operations facilitate diffusion of polyphenols in juice, as occurs during vinification of red wine. This maceration accounts for the fact that the polyphenol content of red wines is 10 times as high as that of white wines 88 and is also higher than that of grape juice 89.
Another factor that directly affects the polyphenol content of the foods is storage. Studies have proved that polyphenolic content of the foods change on storage, the reason is easy oxidation of these polyphenols 81. Oxidation reactions result in the formation of more or less polymerized substances, which lead to changes in the quality of foods, particularly in color and organoleptic characteristics. Such changes may be beneficial, as is the case with black tea or harmful as in browning of fruit. Storage of wheat flour results in marked loss of phenolic acids 90. After six months of storage, flour contained the same phenolic acids in qualitative terms, but their concentrations were 70% lower compared with fresh. Cold storage, in contrast, has slight effect on the content of polyphenols in apples, pears or onions 91. Cooking also has a major effect on concentration of polyphenols. Onions and tomatoes lose between 75% and 80% of their initial quercetin content after boiling for 15 min, 65% after cooking in a microwave oven, and 30% after frying 92.
Because of the wide range of existing polyphenols and the considerable number of factors that can modify their concentration in foods, no reference food-composition tables (as they exist for other micronutrients such as vitamins) have yet been drawn up. Only partial data for certain polyphenols, such as flavonols and flavones, catechins, and isoflavones, have been published on the basis of direct food analysis or bibliographic compilations.
Since March 2003, a database in which the flavonoid contents of 225 selected foods were compiled from 97 bibliographic sources has been available on the US Department of Agriculture website at 93 https://www.ars.usda.gov/ARSUserFiles/80400525/Articles/JAFC54_9966-9977Flavonoid.pdf and here at https://search.usa.gov/search?query=flavonoid&op=Search&affiliate=fnic
Table 2. Polyphenols Rich Foods
| Source (serving size) | Polyphenol content | ||
|---|---|---|---|
| By weight or volume | By serving | ||
| mg/kg fresh weight (or mg/L) | mg/serving | ||
| Hydroxybenzoic acids | Blackberry (100 g) | 80–270 | 8–27 |
| Protocatechuic acid | Raspberry (100 g) | 60–100 | 6–10 |
| Gallic acid | Black currant (100 g) | 40–130 | 4–13 |
| p-Hydroxybenzoic acid | Strawberry (200 g) | 20–90 | 4–18 |
| Hydroxycinnamic acids | Blueberry (100 g) | 2000–2200 | 200–220 |
| Caffeic acid | Kiwi (100 g) | 600–1000 | 60–100 |
| Chlorogenic acid | Cherry (200 g) | 180–1150 | 36–230 |
| Coumaric acid | Plum (200 g) | 140–1150 | 28–230 |
| Ferulic acid | Aubergine (200 g) | 600–660 | 120–132 |
| Sinapic acid | Apple (200 g) | 50–600 | 10–120 |
| Pear (200 g) | 15–600 | 3–120 | |
| Chicory (200 g) | 200–500 | 40–100 | |
| Artichoke (100 g) | 450 | 45 | |
| Potato (200 g) | 100–190 | 20–38 | |
| Corn flour (75 g) | 310 | 23 | |
| Flour: wheat, rice, oat (75 g) | 70–90 | 5–7 | |
| Cider (200 mL) | 10–500 | 2–100 | |
| Coffee (200 mL) | 350–1750 | 70–350 | |
| Anthocyanins | Aubergine (200 g) | 7500 | 1500 |
| Cyanidin | Blackberry (100 g) | 1000–4000 | 100–400 |
| Pelargonidin | Black currant (100 g) | 1300–4000 | 130–400 |
| Peonidin | Blueberry (100 g) | 250–5000 | 25–500 |
| Delphinidin | Black grape (200 g) | 300–7500 | 60–1500 |
| Malvidin | Cherry (200 g) | 350–4500 | 70–900 |
| Rhubarb (100 g) | 2000 | 200 | |
| Strawberry (200 g) | 150–750 | 30–150 | |
| Red wine (100 mL) | 200–350 | 20–35 | |
| Plum (200 g) | 20–250 | 4–50 | |
| Red cabbage (200 g) | 250 | 50 | |
| Flavonols | Yellow onion (100 g) | 350–1200 | 35–120 |
| Quercetin | Curly kale (200 g) | 300–600 | 60–120 |
| Kaempferol | Leek (200 g) | 30–225 | 6–45 |
| Myricetin | Cherry tomato (200 g) | 15–200 | 3–40 |
| Broccoli (200 g) | 40–100 | 8–20 | |
| Blueberry (100 g) | 30–160 | 3–16 | |
| Black currant (100 g) | 30–70 | 3–7 | |
| Apricot (200 g) | 25–50 | 5–10 | |
| Apple (200 g) | 20–40 | 4–8 | |
| Beans, green or white (200 g) | 10–50 | 2–10 | |
| Black grape (200 g) | 15–40 | 3–8 | |
| Tomato (200 g) | 2–15 | 0.4–3.0 | |
| Black tea infusion (200 mL) | 30–45 | 6–9 | |
| Green tea infusion (200 mL) | 20–35 | 4–7 | |
| Red wine (100 mL) | 2–30 | 0.2–3 | |
| Flavones | Parsley (5 g) | 240–1850 | 1.2–9.2 |
| Apigenin | Celery (200 g) | 20–140 | 4–28 |
| Luteolin | Capsicum pepper (100 g) | 5–10 | 0.5–1 |
| Flavanones | Orange juice (200 mL) | 215–685 | 40–140 |
| Hesperetin | Grapefruit juice (200 mL) | 100–650 | 20–130 |
| Naringenin | Lemon juice (200 mL) | 50–300 | 10–60 |
| Eriodictyol | |||
| Isoflavones | Soy flour (75 g) | 800–1800 | 60–135 |
| Daidzein | Soybeans, boiled (200 g) | 200–900 | 40–180 |
| Genistein | Miso (100 g) | 250–900 | 25–90 |
| Glycitein | Tofu (100 g) | 80–700 | 8–70 |
| Tempeh (100 g) | 430–530 | 43–53 | |
| Soy milk (200 mL) | 30–175 | 6–35 | |
| Monomeric flavanols | Chocolate (50 g) | 460–610 | 23–30 |
| Catechin | Beans (200 g) | 350–550 | 70–110 |
| Epicatechin | Apricot (200 g) | 100–250 | 20–50 |
| Cherry (200 g) | 50–220 | 10–44 | |
| Grape (200 g) | 30–175 | 6–35 | |
| Peach (200 g) | 50–140 | 10–28 | |
| Blackberry (100 g) | 130 | 13 | |
| Apple (200 g) | 20–120 | 4–24 | |
| Green tea (200 mL) | 100–800 | 20–160 | |
| Black tea (200 mL) | 60–500 | 12–100 | |
| Red wine (100 mL) | 80–300 | 8–30 | |
| Cider (200 mL) | 40 | 8 | |
Table 3. Plant, fruit and vegetable-based Polyphenols separated according to differences in chemical structure
| Phenolic Family | Dietary Sources | Agri-Food Residue or Non-Edible Sources |
|---|---|---|
| Phenolic acid | ||
| Hydroxycinnamic acid | Almonds, cereals, cherries, citrus juices and fruits, coffee, corn flour, peaches, plums, potato, rice flour, spinach, tomatoes, and wheat flour. | Apple pomace, artichoke wastewaters, banana peel, citrus peels, olive mill wastewaters, and spent coffee grounds. |
| Hydroxybenzoic acid | Black currant, blackberry, cereals, coffee, cowpea, oilseeds, raspberry, and wheat flour. | Citrus peels, grape pomace, residual brewing yeast, squash shells and seeds. |
| Flavonoids | ||
| Flavonols | Apples, apricot, arugula, beans, capers, cloves, leeks, lettuce, onions, saffron, and tomatoes. | Apples peels, banana peels, grape pomace and seeds, guava peels and seeds, onion peels, olive leaves and pomace. |
| Flavones | Artichoke, black olive, celery, citrus fruits, oregano, peanut, parsley, pepper, spinach. | Artichokes steams, camu-camu peels and peanut skin and shell |
| Isoflavones | Red clover, soybeans, soymilk and soy flour. | Soy processing waste and peanut skins and shells. |
| Flavanols | Grapes, apples, tomatoes, leeks, lettuces, curly kale, berries, onions, red grapes, beans, green and black, cider, tea, red wine. | Appel peel, grape seeds, peels and pomace and tea by-products. |
| Anthocyanins | Eggplant, grape, plums, pomegranate, raspberries, red and back-currants, red cabbage, red wine, strawberries, and radish. | Grape skins and seeds, grape pomace and floral tepals (saffron). |
| Flavanones | Citrus juices, citrus fruits, peppermint, fennel and rosemary. | Banana peels, citrus seeds, peels and pomace and residual brewing yeast. |
| Stilbenes | Almonds, grapes, red wine and berries. | Avocado peels, grape skins, seeds, pomace and stems. |
| Lignans | Broccoli, flaxseed, kale, lentil, sesame seeds, tea, wine and wheat. | Coffee, soybeans and wine residues. |
| Tannins | ||
| Hydrolizable tannins | Pomegranates, raspberries and tea | Pomegranate peels and seeds and tea by-products. |
| Condense tannins | Apples, chestnut, grapes, pears, peaches, and hazelnuts. | Grape seeds |
| Phlorotannins | – | Brown seaweed |
| Complex tannins | – | Cork by-products (e.g., tree bark) |

Bioavailability of Polyphenols
Bioavailability is the proportion of the nutrient that is digested, absorbed and metabolized through normal pathways. Bioavailability of each and every polyphenol differs however there is no relation between the quantity of polyphenols in food and their bioavailability in human body. Generally, aglycones can be absorbed from the small intestine; however most polyphenols are present in food in the form of esters, glycosides or polymers that cannot be absorbed in native form 95. Before absorption, these compounds must be hydrolyzed by intestinal enzymes or by colonic microflora. During the course of the absorption, polyphenols undergo extensive modification; in fact they are conjugated in the intestinal cells and later in the liver by methylation, sulfation and/or glucuronidation.18 As a consequence, the forms reaching the blood and tissues are different from those present in food and it is very difficult to identify all the metabolites and to evaluate their biological activity. Importantly it is the chemical structure of polyphenols and not its concentration that determines the rate and extent of absorption and the nature of the metabolites circulating in the plasma. The most common polyphenols in our diet are not necessarily those showing highest concentration of active metabolites in target tissues; consequently the biological properties of polyphenols greatly differ from one polyphenol to another. Evidence, although indirect, of their absorption through the gut barrier is given by the increase in the antioxidant capacity of the plasma after the consumption of polyphenols-rich foods.
Polyphenols also differs in their site of absorption in humans. Some of the polyphenols are well absorbed in the gastro-intestinal tract while others in intestine or other part of the digestive tract. In foods, all flavonoids except flavanols exist in glycosylated forms. The fate of glycosides in the stomach is not clear yet. Most of the glycosides probably resist acid hydrolysis in the stomach and thus arrive intact in the intestine where only aglycones and few glucosides can be absorbed. Experimental studies carried out in rats23 showed that the absorption at gastric level is possible for some flavonoids, such as quercetin, but not for their glycosides. Moreover it has been recently shown that, in rats and mice, anthocyanins are absorbed from the stomach.
Accumulation of polyphenols in the tissues is the most important phase of polyphenol metabolism because this is the concentration which is biologically active for exerting the effects of polyphenols. Studies have shown that the polyphenols are able to penetrate tissues, particularly those in which they are metabolized such as intestine and liver. Excretion of polyphenols with their derivatives occurs through urine and bile. It has been observed that the extensively conjugated metabolites are more likely to be eliminated in bile, whereas small conjugates, such as monosulfates, are preferentially excreted in urine. Amount of metabolites excreted in urine is roughly correlated with maximum plasma concentrations. Urinary excretion percentage is quite high for flavanones from citrus fruit and decreases from isoflavones to flavonols. Thus the health beneficial effects of the polyphenols depend upon both the intake and bioavailability 96.
Polyphenols Health Benefits
Epidemiological studies have repeatedly shown an inverse association between the risk of chronic human diseases and the consumption of polyphenolic rich diet 97, 98. It is well established that polyphenol-rich foods and beverages may increase plasma antioxidant capacity 99. This increase in the antioxidative capacity of plasma following the consumption of polyphenol-rich food may be explained either by the presence of reducing polyphenols and their metabolites in plasma, by their effects upon concentrations of other reducing agents (sparing effects of polyphenols on other endogenous antioxidants), or by their effect on the absorption of pro-oxidative food components, such as iron 97. Consumption of antioxidants has been associated with reduced levels of oxidative damage to lymphocytic DNA. Similar observations have been made with plyphenol-rich food and beverages indicating the protective effects of polyphenols 100. There are increasing evidences that as antioxidants, polyphenols may protect cell constituents against oxidative damage and, therefore, limit the risk of various degenerative diseases associated with oxidative stress 101, 102, 103.
Dietary polyphenols exert preventive effects in treatment of asthma 104, 105. In asthma the airways react by narrowing or obstructing when they become irritated. This makes it difficult for the air to move in and out. This narrowing or obstruction can cause one or a combination of symptoms such as wheezing, coughing, shortness of breath and chest tightness. Epidemiological evidence that polyphenols might protect against obstructive lung disease come from studies that have reported negative associations of apple intake with prevalence and incidence of asthma, and a positive association with lung function 106, 104. Increased consumption of the soy isoflavone, genistein, was associated with better lung function in asthmatic patients 105.
Intake of polyphenols is also reported as beneficial in osteoporosis. Supplementation of diet with genistein, daidzein or their glycosides for several weeks prevents the loss of bone mineral density and trabecular volume caused by the ovariectomy 107. Polyphenols also protect skin damages induced from sunlight. Study on animals provide evidence that polyphenols present in the tea, when applied orally or topically, ameliorate adverse skin reactions following UV exposure, including skin damage, erythema and lipid peroxidation 108.
Black tea polyphenols are reported to be helpful in mineral absorption in intestine as well as to possess antiviral activity. Theaflavins present in black tea were found to have anti HIV-1 activity. These polyphenols inhibited the entry of HIV-1 cells into the target cells. HIV-1 entry into the target cell involves fusion of glycoprotein (GP) and envelope of the virus with the cell membrane of the host cells. Haptad repeat units present at N and C terminals of GP41 (membrane protein) on the viral envelope, fuse to form the fusion active GP41 core, which is a six-helical bundle. Theaflavins were found to block the formation of this six-helix bundle required for entry of the virus into the host 109. Theaflavin 3 3′ digallate, and theaflavin 3′ gallate were found to inhibit Severe Acute Respiratory Syndrome (SARS) corona virus. This antiviral activity was due to inhibition of the chymotrypsin like protease (3CL Pro) which is involved in the proteolytic processing during viral multiplication.
Table 4. Polyphenols Health Benefits
| Health Benefits | Polyphenols From | Type of Polyphenols | Outcome | References |
|---|---|---|---|---|
| Antioxidant activity | Rhododendron tomentosum | Rosmarinic acid Caffeic acid Chlorogenic acid Rutin Quercetin | -Exhibit DPPH radical scavenging activity | 110 |
| Rye Bread | * | -Exhibit an improvement in functional and nutritional value | 111 | |
| Red cabbage | * | -Exhibit scavenge free radicals activity | 112 | |
| Herbal tea | Gallic acid Catechin Caffeic acid Ferulic acid Epicatechin Gallate Quercetin Kaempferol | -Brewing time affects the extractability of polyphenols and antioxidant activity in tea | 113 | |
| Fabacea | * | -Exhibit hydrogen peroxide and nitric oxide scavenging activities | 114 | |
| Rosa roxburghii | Gallic acid Ellagic acid Gallocatechin Epigallocatechin Catechin Epicatechin | -Exhibit scavenge free radicals activity | 115 | |
| * | 3,4-dihydroxyphenylacetic acid Homovanillic acid Vanillic acid Caffeic acid Gallic acid Phloroglucinol Pelargonidi Ellagic acid | -Exhibit anti- or pro-oxidants activity -Exhibit hydroxyl radicals scavenging activities | 17 | |
| De-oiled rice bran | Vanillin Ferulic acid Sinapic acid Chlorogenic acid | -Exhibit an enhancement in vitro digestion -Exhibit anti-inflammatory activity | 116 | |
| Corn bran | 4-hydroxybenzaldehyde p-coumaric Sinapic acid Ferulic acid | |||
| Amaranthus lividus | * | -Exhibit scavenge free radicals activity | 117 | |
| Banana | 3-Hydroxyphenylpropionic acid Ferulic acid Caffeic acid Anthocyanins Cyanidin 2′-Hydroxyformononetin Quercetin Neoeriocitrin Scopoletin 2′-Hydroxyformononetin | -Exhibit capturing free radicals and antioxidant activity | 118 | |
| Zhourat | Gallic acid | -Exhibit higher antioxidant capacity | 119 | |
| Leptospermum scoparium | * | -Exhibit scavenge free radicals activity | 120 | |
| Sambucus ebulus | Chlorogenic acid Caffeic acid glucoside 3-p-coumaroylquinic acid 3-p-Feruloylquinic acid Catechin Epicatechin Procyanidin Kaempferol Quercetin Piceid | -Exhibit DPPH radical scavenging activity | 121 | |
| Rubus spp. | Gallic acid Neochlorogenic acid Procyanidin Catechin Vanillic acid Caffeic acid Epicatechin p-coumaric acid Quercetin Ferulic acid Kaempferol | -Exhibit bioavailability and bioactivity | 122 | |
| Thymus serpyllum L. | Rosmarinic acid Luteolin Salvianolic acid | -Exhibit antibacterial activity | 10 | |
| Euphorbia antisyphilitica | * | -Exhibit an inhibition of lipid oxidation | 123 | |
| Herbal tea and green tea | * | -Exhibit scavenge free radicals activity | 124 | |
| Eugenia uniflora leaves Eucalyptus microcorys leaves Myrciaria cauliflora seeds | Ellagic acid Kaempferol Quercetin Myricetin 2,3-Di-O-galloyl-glucose 2,3,6-Tri-O-galloyl-glucose 1,2,3,4,6-Penta-O-galloyl-glucose 4,6-O-HHDP-glucose Gemin Oenothein Isocoriariin Tellimagrandin Pedunculagin Tellimagrandin Eugeniflorin Camptothin Oenothein | -Exhibit antioxidant capacity | 125 | |
| Satureja hortensis L. | Rutin Rosmarinic acid | -Exhibit radical scavenging activity -Exhibit an improvement the lipid peroxidation process | 126 | |
| Chamerion angustifolium | Oenothein Quercetin Myricetin Luteolin Kaempferol Gallic acid Chlorogenic acid p-coumaric acid Ellagic acid Benzoic acid etc. | -Exhibit antioxidant capacity | 127 | |
| Sargassum wightii | Gallic acid Quercetin Ferulic acid Vanillin | -Exhibit higher antioxidant capacity | 18 | |
| Ulva rigida | ||||
| Gracilaria edulis | ||||
| Pistacia lentiscus L. | Feruloylquinic acid p-coumaroylquinic acid 5-O-caffeoylquinic acid Monogalloyl glucose Gallic acid 5-O-galloylquinic acid Chlorogenic acid Digalloylquinic acid Procyanidin Epicatechin Catechin Epigallocatechin gallate Trigalloylquinic acid p-coumaric acid Myricetin Quercetin Kaempferol Luteolin Apigenin | -Exhibit higher antioxidant capacity | 128 | |
| Amaranthus dubius | 2-O- Caffeoylglucaric acid Ferulic acid 4-Hydroxycinnamic acid Kaempferol Caffeoylquinic acid Myricetin Quercetin | -Exhibit higher antioxidant capacity -Exhibit anti-inflammatory activity | 129 | |
| Amaranthus spinosus | Dihydromyricetin Ferulic acid 4-Hydroxycinnamic acid Feruloylquinic acid Kaempferol Caffeoylquinic acid Myricetin Quercetin | |||
| Amaranthus tricolor | 2-O-Caffeoylglucaric acid Ferulic acid 4-Hydroxycinnamic acid Kaempferol Caffeoylquinic acid Myricetin Quercetin | |||
| Amaranthus viridis | Ferulic acid 4-Hydroxycinnamic acid Myricetin Quercetin Quercetin | |||
| Carrot | Gallic acid Protocatechuic acid Vanillic acid 4-hydroxybenzaldehyde | -Exhibit scavenge reactive oxygen species activity -Exhibit antioxidant capacity | 11 | |
| Echinacea Purpurea | Caftaric Chicoric acids Catechins | -Exhibit antioxidant capacity | 12 | |
| Malus domestica borkh | Chlorogenic acid p-coumaric acid Quercetin -3-O-galactoside -3-O-arabinoside Phloretin-2′-O-glucoside Catechin Epicatechin Procyanidin | -Exhibit higher antioxidant capacity | 130 | |
| Eucalypts leaf | * | -Exhibit antioxidant capacity -Exhibit scavenge free radicals activity -Modulate gut microbiota | 131 | |
| Nigella sativa L. | Gallic acid Hydroquinone Apigenin Naringenin Quercetin Kaempferol Rutin | -Exhibit antioxidant capacity | 132 | |
| Ipomoea batatas | Cyanidin Peonidin Pelargonidin | -Exhibit antioxidant capacity | 21 | |
| Vitis vinifera L. | Flavan-3-ol Proanthocyanidin Anthocyanins | -Exhibit antioxidant capacity | 133 | |
| Coffee silverskin | Caffeoylquinic Feruloylquinic acids | -Exhibit an improvement bioaccessibility | 134 | |
| Coffee | * | -Exhibit antioxidant capacity | 135 | |
| Polyscias fruticosa roots | * | -Exhibit antioxidant capacity -Exhibit scavenge free radicals activity | 136 | |
| Chroogomphus rutilus | Protocatechuic acid | -Exhibit higher antioxidant capacity -Exhibit anti-inflammatory activity -Exhibit cytotoxic effect | 13 | |
| Anti-inflammatory activity | Tetraclinis articulata | * | -Exhibit antioxidant capacity -Exhibit scavenge free radicals activity | 137 |
| Pleurotus ostreatus | Cathechin Sinapic acid Resveratrol etc. | -Exhibit antioxidant activity | 138 | |
| Green tea and red wine | * | -Exhibit antioxidant activity | 139 | |
| Punica granatum L. | Luteolin Rosmarinic acid Quercetin Eriodictyol etc. | -Exhibit antioxidant capacity | 140 | |
| Thymus vulgaris | Rosmarinic acid Luteolin etc. | |||
| Rosmarinus officinalis L. | Chlorogenic acid Caffeic acid etc. | |||
| Echinacea purpurea L. | Ellagic acid Gallagic acid etc. | |||
| Maclura tricuspidate Pyrus Montana Naka | Gallic acid Protocatechuic acid Chlorogenic acid p-hydroxybenzoic acid Vanillic acid Caffeic acid Rutin ρ-coumaric acid Ferulic acid Rosmarinic acid Salicylic acid Quercetin Cinnamic acid Taxifolin | -Exhibit antioxidant activity -Modulate iNOS, COX-2, and TNF-α protein expression | 141 | |
| Olive Oil | Oleacein Oleocanthal | -Exhibit scavenge free radicals activity -Modulate the synthesis of pro-inflammatory lipid mediators | 142 | |
| Finger millet | Protocatechuic acid Catechin Chlorogenic acid Naringin | -Exhibit anti-obesity effect -Modulate production of cytokines -Exhibit antioxidant activity | 143 | |
| Kodo millet | Catechin Naringin p-coumaric acid Taxifolin Ferulic acid Sinapic acid Methyl vanillate | |||
| Rhamnus prinoides L’Herit | Caffeic acid Protocatechuic acid Kaempferol Gallocatechin Proanthocyanidin Luteolin Quercetin Apigenin Rutin etc. | -Exhibit antioxidant activity | 144 | |
| Petroselinum crispum Apium graveolens Coriandrum sativum | * | -Exhibit scavenge free radicals activity -Exhibit membrane stabilizing effect | 145 | |
| Huangjiu | Protocatechuic acid Catechin Chlorogenic acid Vanillic acid Caffeic acid Syringic acid p-coumaric acid Ferulic acid Sinapic acid Rutin Quercetin | -Modulate production of cytokines | 146 | |
| Arabidopsis thaliana | Caffeic acid Quercetin Kaempferol Synapic acid Luteolin | -Modulate production of cytokines | 147 | |
| Ilex latifolia | Quinic acid Caffeoylquinic acid Shikimic acid Rutin Hyperoside etc. | -Exhibit scavenge free radicals activity -Exhibit antioxidant activity -Modulate production of cytokines | 148 | |
| Cynara scolymus L. | Hydroxytyrosol Verbascoside Apigetrin Oleuropein Quercetin Pinoresinol Apigenin | -Exhibit antioxidant activity | 149 | |
| Acalypha hispida | Gallic acid Quercetin Ellagic acid p-coumaric acid etc. | -Exhibit antioxidant activity -Modulate inflammatory pathways | 150 | |
| Lonicera caerulea L. | Chlorogenic acid Caffeic acid Catechin Epicatechin Cyanidin etc. | -Modulate production of cytokines | 151 | |
| Prunus domestica L. | Chlorogenic acid p-coumaric acid Rutin etc. | -Exhibit antioxidant activity -Exhibit induction of lipid peroxidation | 152 | |
| Gaultheria procumbens L. | Protocatechuic acid Caffeoylquinic acid p-hydroxybenzoic acid Vanillic acid Catechin Epicatechin p-coumaric acid Procyanidin Quercetin Kaempferol etc. | -Exhibit antioxidant activity | 153 | |
| Baccaurea ramiflora Lour | Rosmarinic acid | -Exhibit antioxidant activity -Modulate production of cytokines | 154 | |
| Libidibia ferrea Parapiptadenia rigida Psidium guajava | Catechin Gallic acid | -Exhibit inhibition character on bacterial zone | 155 | |
| Phaseolus vulgaris bean | Sinapic acid Ferulic acid Naringenin Catechin Quercetin etc. | -Exhibit antioxidant activity -Modulate production of cytokines | 156 | |
| Verbascum phlomoides | Gallic acid Rosmarinic acid Caffeic acid Ferulic acid Quercetin etc. | -Exhibit antioxidant activity | 157 | |
| Rubus coreanus Miquel | * | -Exhibit anti-super bacterial activity -Modulate production of cytokines | 158 | |
| Antimicrobial activity | Guizotia abyssinica L. leaf and flower extracts | Tannins Glycosides Flavanoids Phenols | -Exhibit antioxidant activity | 159 |
| Retama monosperma | Flavonoids Tannins Quinones Anthocyanins | -Exhibit antioxidant activity -Exhibit antimicrobial activity against Staphylococcus aureus, and Bacillus cereus. | 6 | |
| Filipendula ulmaria | Quercetin Rutin | -Exhibit higher antioxidant activity -Exhibit antimicrobial activity against Listeria monocytogenes | 160 | |
| Salvia officinalis | Quercetin Apigenin Naringenin Rutin | |||
| Rosmarinus officinalis | Luteolin Eriodictyol | |||
| Sideritis scardica | Quercetin Rutin Epicatechin | |||
| Geranium purpureum | Quercetin Rutin Catechin Epicatechin Hydroxytyrosol | |||
| Banana peels | * | -Exhibit higher antioxidant activity -Exhibit antimicrobial activity against Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, and Saccharomyces cerevisiae | 161 | |
| Artemisia aucheri | * | -Exhibit an improvement in health effect in mice against Campylobacter jejuni | 162 | |
| Grape pomace | Anthocyanins Phenolic acid Flavonoids Stilbenes | -Modulate antioxidant activity -Exhibit an increase in Lactiplantibacillus plantarum growth -Exhibit antimicrobial activity against Escherichia coli, Bacillus megaterium, and Listeria monocytogenes | 163 | |
| Alcea rosea | Gallic acid Salicylic acid Pyrogallol Cinnamic acid Catechin Naringin Ferulic acid | -Exhibit antimicrobial activity against Escherichia coli | 164 | |
| Achillea millefolium | Salicylic Succinic acids Folic acid Caffeic acid Kaempferol Luteolin Apigenin and other phenolic and flavonoid compounds | -Improve the average daily weight gain, food intake, liver function, and antioxidant status -Decrease the ileum population of C. jejuni in the mice challenged by C. jejuni infection | 165 | |
| Rheum ribes | Gallic acid Salicylic acid Caffeic acid Cinnamic acid Catechin Ellagic acid Ferulic acid | -Exhibit antimicrobial activity against Escherichia coli and improve health parameters of mice | 166 | |
| Lycium chinense Mill. | Quercetin Kaempferol Catechin Flavan-3-ols Coumaric acid Chlorogenic acid Procyanidin | -Exhibit antibacterial activity against Bacillus subtilis and Proteus vulgaris | 167 | |
| Propolis | p-coumaric acid Ferulic acid Chrysin | -Exhibit scavenge free radicals activity -Exhibit antimicrobial activity against Escherichia coli, Bacillus subtilis spizizenii nakamura, and Candida albicans | 168 | |
| * | Combined polyphenols | -Exhibit protective effect against influenza A and SARS-CoV-2 | 169 | |
| Punica granatum L. | Ursolic acid Corosolic acid Arjunolic acid | -Exhibit antimicrobial activity against Staphylococcus aureus | 170 | |
| Hibiscus sabdariffa L. | Kaempferol Cyanidin Quercetin | -Exhibit antimicrobial activity against Candida albicans, Staphylococcus aureus, and Listeria monocytogenes | 171 | |
| Spirulina | * | -Exhibit antimicrobial activity against drug resistant food pathogens. | 172 | |
| Zhourat | Gallic acid etc. | -Exhibit inhibition character on bacterial zone | 119 | |
| Lantana camara L. | Tetramethylhexadec-2-en-1-ol Linolenic acid 2,6-Dimethoxyphenol 9,12-Octadecadienoic acid | -Exhibit scavenge free radicals activity -Exhibit antimicrobial activity against Xanthomonas axonopodis pv. glycines and Xanthomonas oryzae pv. oryzae | 173 | |
| Picea abies L. Larix decidua Mill Pinus sylvestris L. Pseudotsuga menziesii Juniperus communis L. | Gallic acid p-coumaric acid 2,5-dihydroxybenzoic acid 4-hydroxybenzoic acid Chlorogenic acid Caffeic acid Syringic acid Vanillic acid Sinapic acid Ferulic acid Salicylic acid Cinnamic acid Vitexin Apigenin Kaempferol Luteolin Quercetin Naringenin Rutin | -Exhibit antioxidant and antiradical activity -Exhibit antimicrobial activity | 174 | |
| Natural polyphenols | TF3 TF2b TF1 TF2a Hesperidin EGCG Myricetin Quercetagetin | -Exhibit inhibition effect on the RNA-dependent RNA polymerase of SARS-CoV-2 | 175 | |
| * | Apigenin Catechin Luteolin Morin Myricetin Naringin Quercetin Rutin | -Exhibit inhibition effect on bacterial and fungal growth | 176 | |
| Olive oil | * | -Exhibit antibacterial activity against Listeria monocytogenes -Exhibit a reduction in intracellular ATP concentrations | 177 | |
| Prunus dulcis | Epicatechin Catechin | -Exhibit the inhibition character against bacterial growth and reducing expression of viral proteins | 178 | |
| Vitis vinifera L. | Gallic acid Coumaric acid Vanillic acid Chlorogenic acid Cyanidin Catechin Caffeic acid Peonidin 3-O-glucoside Epicatechin Luteolin Resveratrol Ferulic acid | -Exhibit antioxidant activity -Exhibit inhibition character on bacterial zone | 179 | |
| Moringa oleifera | Coumaric acid Myricetin Quercetin Kaempferol Resveratrol Naringenin Biochanin A Naringin Catechin | -Exhibit antioxidant activity -Exhibit inhibition character on bacterial zone | 180 | |
| Olive oil | * | -Exhibit antimicrobial activity against Cronobacter sakazakii -Exhibit a reduction in intracellular ATP concentrations -Exhibit an increase in cell membrane permeability | 181 | |
| Achillea pachycephala Achillea millefolium Achillea nobilis Achillea filipendulina Achillea santolina Achillea aucheri | Chlorogenic acid Caffeic acid Quercetin Luteolin Rutin Ferulic acid | -Exhibit antioxidant activity -Exhibit antibacterial activity against Staphylococcus aureus, Bacillus cereus, Escherichia coli, Staphylococcus epidermidis, and Salmonella typhimurium | 182 | |
| * | Stilbenes Cinnamic Benzoic Flavonoids Coumarins Naphtoquinones | -Exhibit antimicrobial activity against Staphylococcus aureus, Bacillus subtilis, Listeria monocytogenes, Escherichia coli, Pseudomonas aeruginosa, and Salmonella Enteritidis | 183 | |
| Antidiabetic activity | Solanum anguivi | * | -Exhibit antioxidant activity -Exhibit scavenge free radicals activity | 184 |
| Syzygium zeylanicum L. | Gallic acid Catechin Epicatechin Caffeine Quercetin Apigenin Ethyl gallate Rutin Ellagic acid Chlorogenic acid Quercitrin | -Exhibit antidiabetic activity by modulation of gene expressions of lipid and glucose metabolism | 185 | |
| Cucumis dipsaceus | Rutin Gallic acid | -Increase phenolic and flavonoid compounds -Exhibit scavenge free radicals activity | 186 | |
| Phaseolus vulgaris L. | * | -Exhibit antidiabetic activity, improve the T2DM outcomes | 187 | |
| Gracilaria bursa-pastoris | Gallic acid Catechin 4-hydroxy benzoïc acid Chlorogenic acid Caffeic acid Syringic acid Vanilline p-coumaric acid Sinapic acid Quercetin 7,3′,4′-flavon-3-ol Naringin Rutin Salicylic acid Quercetin Cinnamic acid Luteolin Apigenin Kaempferol Flavone Flavanone | -Exhibit antioxidant activity | 188 | |
| Carica papaya | * | -Exhibit fat-lowering effects and stimulate glucose activity in liver cells | 189 | |
| Curcuma longa | Curcumin | -Exhibit pharmacokinetic activity | 190 | |
| Cocos nucifera | Gallic acid Ferulic acid 4-Hydroxycinnamic acid p-coumaric acid Quercetin | -Exhibit DPPH radical scavenging activity -Exhibit α-amylase and α-glucosidase inhibitor activities | 191 | |
| Borassus flabellifer | Gallic acid Ferulic acid 4-Hydroxycinnamic acid Quercetin Myricetin-3-O-glucoside | |||
| Vinegar extract | 4-Hydroxybenzoic acid Ferulic acid Salicylic acid Vanillic acid Protocatechuic acid Catechin Ellagic acid Gallic acid Gallocatechin 3-O-gallate Rutin etc. | -Exhibit an improvement in glucose tolerance and insulin tolerance -Exhibit a reduction in inflammation in the liver | 192 | |
| Vigna radiata L. | Gallic acid Vitexin | -Modulate gut microbiota -Exhibit antioxidant activity -Exhibit anti-inflammatory activity | 193 | |
| Quercus suber Quercus ilex Quercus coccifera Quercus canariensis | Chlorogenic acid | -Exhibit α-amylase inhibitory activity | 194 | |
| Red wine | Gallic acid Caftaric acid Coutaric acid Malvidin 3-O-glucoside Petunidin 3-O-glucoside | -Exhibit anti-radical effect -Exhibit antioxidant activity | 195 | |
| Aerva lanata L. Juss | Gallic acid Protocatechuic acid Caffeic acid Syringic acid 4-hydroxybenzoic acid Vanillic acid Gentisic acid Sinapic acid p-coumaric acid Ferulic acid Rosmarinic acid Isoferulic acid Salicylic acid | -Exhibit antioxidant activity | 196 | |
| Linum usitatissimum | * | -Exhibit reduction in blood glucose levels, weight loss, also possess recovery role for pancreas and liver | 197 | |
| Vigna unguiculata | Gentisic acid Coumaric acid Ferulic acid Quercetin | -Exhibit antioxidant capacity -Exhibit an inhibition of α-glucosidase and α-amylase activities | 198 | |
| Lonicera caerulea L. | Cyanidin Quercetin Chlorogenic acid Flavan-3-ol Catechin Epicatechin | -Exhibit antioxidant activity -Exhibit an inhibition of α-glucosidase and α-amylase activities | 199 | |
| Propolis | Protocatechuic acid Catechin Caffeic acid Syringic Acid Epicatechin p-coumaric acid Ferulic acid Luteolin | -Exhibit antioxidant capacity -Exhibit an inhibition of α-glucosidase and α-amylase activities | 200 | |
| Rosmarinus officinalis L. | * | -Exhibit antioxidant activity -Exhibit anti-aging activity | 201 | |
| Lagerstroemia speciosa | Caffeic acid Ellagitannins Flavonoids Quercetin | -Exhibit a decrease in fasting blood glucose, body weight, levels of serum biomarkers, tissue weight, and body fat | 202 | |
| Peanut shell | Luteolin Pyrogallol Catechol Phloroglucinol Quercetin | -Exhibit protective effects against diabetes -Exhibit a reduction in fasting blood glucose levels | 203 | |
| Skin and hair effects | Caralluma europaea | Luteolin Gallic acid Hesperetin Quercetin Myricetin Ferulic acid Salicylic acid Naringenin | -Exhibited the improving role for wound healing and by a reduction in hepatocellular carcinoma perform anticancer activity | 204 |
| Vitis vinifera seed | * | -Exhibited anti-aging, brightening, and hydrating effects. Also, this effect could be increased by encapsulation | 205 | |
| Rhus coriaria | Anthocyanins Flavonoids Phenols Hydrolyzable tannins Gallic acid Quercetin | -Exhibited that plant extract possesses antibacterial and wound healing properties | 206 | |
| Penthorum chinense Prush | * | -Exhibited anti-aging activity by protection from UVB ray, reduction in free radicals, and increase skin moisture | 207 | |
| * | Naringenin Curcumin | -Exhibited antioxidant effect by decreasing dermal toxicity and also appreciable skin retention effect observed | 208 | |
| Neuroprotective activity | Propolis | * | -Exhibit antioxidative role on PD model | 209 |
| * | Mix of polyphenols | -Exhibit reducing effect against the detrimental process of PD and release of pro-inflammatory cytokines, inhibits apoptosis mechanisms, and decreases oxidative stress | 210 | |
| Phyllanthus emblica L. | Gallic acid Epicatechin Ethly gallate Chebulagic acid Ellagic acid Quercetin | -Exhibit anti-Alzheimer’s effect and antioxidant capacity in transgenic Alzheimer model | 211 | |
| * | Curcumin | -Exhibit improving the median survival life of HD flies and also acts as an antioxidant agent by suppressing oxidative stress | 212 | |
| Olive | * | -Exhibit improving lifespan of PD models and beneficially affect locomotion ability | 213 | |
| * | Resveratrol | -Exhibit enhanced memory function, glutathione, and antioxidative enzymes by stimulating Sirt1 gene expression | 214 | |
| * | Curcumin and fatty acid | -Exhibit reduced effect on gene expression of pro-inflammatory cytokines, thus relaxing symptoms | 215 | |
| * | Curcumin | -Exhibit possess suppressing role on polyQ mediated photo neuron degeneration and reduce locomoter dysfunction | 216 | |
| Grape leaves | * | -Exhibit antioxidative, anti-inflammatory, and neurotrophic effect to support improving brain function | 217 | |
| Tea | * | -Exhibit protection role for dopamine levels by its antioxidative and anti-inflammatory role | 218 | |
| * | Curcumin and fatty acids | -Exhibit decreasing role on interleukin-6 serum levels and decrease in symptoms | 219 | |
| Anti-tumor/Anticancer | Cuminum cyminum | * | -Exhibit inhibitory activity against colon, lung, and breast cancer cell lines -Exhibit antioxidant capacity | 53 |
| Cerasus humilis | * | -Exhibit inhibitory activity against liver, colon, and stomach cancer cells | 220 | |
| Caralluma europaea | Kaempferol Luteolin Trans-ferulic acid Syringic acid | -Exhibit anti-tumoral activity against the human leukemic and liver cancer cell lines | 204 | |
| * | Isoeugenol | -Exhibit anti-proliferative, anti-apoptotic, and anti-migrative against breast cancer cells | 221 | |
| Camellia sinensis | Epigallocatechin-3-gallate | -Exhibit antithrombotic, antitumor, and antiangiogenic activities | 222 | |
| * | Quercetin Fisetin | -Exhibit inhibition of cell proliferation -Exhibit induction of reactive oxygen species formation | 223 | |
| Viscum album | Epicatechin Quercetin | -Exhibit an apoptotic-like effect | 224 | |
| Apple | Cyanidin-3-O-arabinoside | -Exhibit anti-tumoral activity against the human colon cancer cell line -Exhibit an inhibitory effect on proliferation -Exhibit induction of cell apoptosis | 225 | |
| Hippophae rhamnoides | Sinapinic acid Ferulic acid Coumaric acid 7-Hydroxycoumarine Kaempferol 5,7-Dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4-oxo-4H-chromen-3-yl-6-O-(6-deoxy-α-L-mannopyranosyl) hexopyranoside | -Exhibit anti-tumoral activity against the human colon cancer cell line -Modulate miRNA expression profiles | 226 | |
| Artemisia argyi leaf | Neochlorogenic acid Chlorogenic acid Cryptochlorogenic acid Isochlorogenic acid | -Exhibit inhibitory effects on cervical and colon cancer cell lines -Exhibit antioxidant activity | 227 | |
| Ziziphus jujuba | * | -Exhibit inhibitory effects on colon cancer cell lines -Exhibit an inhibitory effect on proliferation | 228 | |
| Coriandrum sativum L. | Flavonoids Catechins Rutin | -Exhibit cytotoxicity against the leukemic cell lines | 229 | |
| Empetrum nigrum | * | -Exhibit strong antioxidant activity -Exhibit high antibacterial potential -Exhibit an inhibition of cell proliferation and induction of apoptosis | 230 | |
| Pinus koraiensis bark | Penta-hydroxy flavone | -Exhibit antioxidant capacity -Exhibit antiproliferative effects -Exhibit scavenge free radicals activity | 231 | |
| Sabal yapa leaves | Tricin Luteolin Apigenin | -Exhibit antioxidant capacity -Exhibit potent anticancer effects against Ehrlich ascites carcinoma cells | 232 | |
| Sugarcane | * | -Exhibits an inhibition of cell proliferation and induction of apoptosis | 233 | |
| Varthemia candicans Peganum harmala Suaeda vermiculata Conyza dioscoridis | * | -Exhibit cytotoxicity against the human hepatocellular carcinoma cells | 234 | |
| Euphorbia lathyris | Esculetin Euphorbetin Gaultherin Kaempferol | -Exhibit inhibitory effects on colon cancer cell lines -Exhibit antiangiogenic capacity | 235 | |
| Ipomoea batatas | Caffeic acid | -Exhibit chemo-sensitizing effects | 236 | |
| Vaccinium spp. | Pelargonidin-3-O-galactoside Delphinidin-3-glucoside Chlorogenic acid isomers Epicatechin gallate Malvidin-3-O-glucose Kaempferol-3-rhamnoside Hexose ferulic acid esters Myricetin-3-O-hexose | -Exhibit antioxidant capacity -Exhibit antiproliferative effects -Exhibit inhibitory effects on breast cancer | 237 | |
| Thalassia testudinum | * | -Exhibit inhibitory effects on colon cancer cell lines | 238 | |
| Eugenia involucrata | Gallic acid Catechin p-coumaric acid Rutin Myricetin Quercetin | -Exhibit antioxidant capacity -Exhibit anti-tumoral activity in a pancreatic cancer cell line | 239 | |
| Agrimonia pilosa | Agrimoniin | -Modulate activation of mitochondria-dependent apoptosis -Exhibit cytotoxicity against the cervical cancer cell line | 240 | |
| Peanut skin | Proanthocyanidin-B2 | -Exhibit antiproliferative effects | 241 | |
| Extra-virgin olive oil | Oleacein | -Exhibit anticancer activity against the cutaneous melanoma -Modulate miRNA expression profiles | 242 | |
| Cinnamomum cassia | * | -Exhibit anticancer activity against the colon cancer cell lines | 243 | |
| Vaccinium macrocarpon | Cyanidin Peonidin | -Exhibit anticancer activity against the colon cancer cell lines | 244 | |
| Camellia sinensis | Epigallocatechin | -Exhibit inhibitory effects on breast cancer | 245 | |
| Green tea | Epigallocatechin | -Exhibit inhibitory effects on human lung cancer cells | 246 | |
| Foxtail millet Bran | Vanillic acid Glucosyringic acid Ferulic acid 4-hydroxybenzoic acid Vanillic acid Syringic acid p-coumaric acid Vitexin Ferulic acid Isoferulic acid Biferulic acid 4,4′-dihydroxy-3,5′-dimethoxy,3′-bicinnamic acid | -Exhibit inhibitory effects on colon cancer cell lines -Modulate gut microbiota | 247 | |
| * | Tannic acid | -Exhibit cytotoxicity against the glioblastoma cells -Exhibit antiglioma activity | 248 | |
| * | Resveratrol Pterostilbene | -Exhibit an inhibition of tumor growth | 249 | |
| Olive oil | Oleacein | -Modulate cell cycle arrest and apoptosis | 250 | |
| Caesalpinia spinosa | * | -Exhibit anti-tumor effects against breast and melanoma tumor -Modulate cell cycle arrest and apoptosis | 251 | |
| Other effects | Green tea Dehydrated red delicious apple Dark chocolate | * | -Exhibit antihypertensive -Exhibit inhibition of cholesterol absorption -Exhibit inhibition of endothelial lipoprotein lipase -Exhibit a reduction in C-reactive protein plasma levels | 252 |
| Fucus vesiculosus | * | -Exhibit anti-radical activity -Exhibit scavenge free radicals activity | 253 |
Footnote: * Not identified.
[Source 1 ]Polyphenols Cardio-Protective Effect
Number of studies has demonstrated that consumption of polyphenols limits the incidence of coronary heart diseases.39–41 Atherosclerosis is a chronic inflammatory disease that develops in lesion-prone regions of medium-sized arteries. Atherosclerotic lesions may be present and clinically silent for decades before becoming active and producing pathological conditions such as acute myocardial infarction, unstable angina or sudden cardiac death.42 Polyphenols are potent inhibitors of LDL oxidation and this type of oxidation is considered to be a key mechanism in development of atherosclerosis.43 Other mechanisms by which polyphenols may be protective against cardiovascular diseases are antioxidant, anti-platelet, anti-inflammatory effects as well as increasing HDL, and improving endothelial function.44 Polyphenols may also contribute to stabilization of the atheroma plaque.
Quercetin, the abundant polyphenol in onion has been shown to be inversely associated with mortality from coronary heart disease by inhibiting the expression of metalloproteinase 1 (MMP1), and the disruption of atherosclerotic plaques.44 Tea catechins have been shown to inhibit the invasion and proliferation of the smooth muscle cells in the arterial wall, a mechanism that may contribute to slow down the formation of the atheromatous lesion.45 Polyphenols may also exert antithrombotic effects by means of inhibiting platelet aggregation. Consumption of red wine or non-alcoholic wine reduces bleeding time and platelet aggregation. Thrombosis induced by stenosis of coronary artery is inhibited when red wine or grape juice is administrated.46 Polyphenols can improve endothelial dysfunction associated with different risk factors for atherosclerosis before the formation of plaque; its use as a prognostic tool for coronary heart diseases has also been proposed.47 It has been observed that consumption of black tea about 450 ml increases artery dilation 2 hours after intake and consumption of 240 mL red wine for 30 days countered the endothelial dysfunction induced by a high fat diet.48 Long term regular intake of black tea was found to lower blood pressure in a cross-sectional study of 218 women above 70 years of age. Excretion of 4-O-methylgallic acid (4OMGA, a biomarker for tea polyphenols in body) was monitored. A higher consumption of tea and therefore higher excretion of 4OMGA were associated with lower blood pressure (BP). Tea polyphenols may be the components responsible for the lowering of BP. The effect may be due to antioxidant activity as well as improvement of endothelial function or estrogen like activity 254.
Resveratrol, the wine polyphenol prevents the platelet aggregation via preferential inhibition of cyclooxygenase 1 (COX 1) activity, which synthesizes thromboxane A2, an inducer of the platelet aggregation and vasoconstrictor 255. In addition to this, resveratrol is capable of relaxing the isolated arteries and rat aortic rings. The ability to stimulate Ca++-activated K+ channels and to enhance nitric oxide signaling in the endothelium are other pathways by which resveratrol exerts vasorelaxant activity 255, 256. Direct relation between cardiovascular diseases (CVDs) and oxidation of LDL is now well established. Oxidation of LDL particles is strongly associated with the risk of coronary heart diseases and myocardial infarctions. Studies have shown that resveratrol potentially inhibits the oxidation of the LDL particles via chelating copper or by direct scavenging of the free radicals. Resveratrol is the active compound in red wine which is attributed for “French Paradox”, the low incidence of CVD despite the intake of high-fat diet and smoking among French 257, 258. Association between polyphenol intake or the consumption of polyphenol-rich foods and incident of cardiovascular diseases were also examined in several epidemiological studies and it was found that consumption of polyphenol rich diet have been associated to a lower risk of myocardial infarction in both case-control and cohort studies 259.
Polyphenols Anti-Cancer Effect
Effect of polyphenols on human cancer cell lines, is most often protective and induce a reduction of the number of tumors or of their growth 260. These effects have been observed at various sites, including mouth, stomach, duodenum, colon, liver, lung, mammary gland or skin. Many polyphenols, such as quercetin, catechins, isoflavones, lignans, flavanones, ellagic acid, red wine polyphenols, resveratrol and curcumin have been tested; all of them showed protective effects in some models although their mechanisms of action were found to be different 261.
Development of cancer or carcinogenesis is a multistage and microevolutionary process. Into the three major stages of carcinogenesis: initiation, promotion and progression. Initiation is a heritable aberration of a cell. Cells so initiated can undergo transformation to malignancy if promotion and progression follow. Promotion, on the other hand, is affected by factors that do not alter DNA sequences and involves the selection and clonal expansion of initiated cells.
Several mechanisms of action have been identified for chemoprevention effect of polyphenols, these include estrogenic/antiestrogenic activity, antiproliferation, induction of cell cycle arrest or apoptosis, prevention of oxidation, induction of detoxification enzymes, regulation of the host immune system, anti-inflammatory activity and changes in cellular signaling 254.
Polyphenols influence the metabolism of pro-carcinogens by modulating the expression of cytochrome P450 enzymes involved in their activation to carcinogens. They may also facilitate their excretion by increasing the expression of phase II conjugating enzymes. This induction of phase II enzymes may have its origin in the toxicity of polyphenols. Polyphenols can form potentially toxic quinones in the body that are, themselves, substrates of these enzymes. The intake of polyphenols could then activate these enzymes for their own detoxication and, thus, induce a general boosting of our defenses against toxic xenobiotics 262. It has been demonstrated that tea catechins in the form of capsules when given to men with high-grade prostate intraepithelial neoplasia (PIN) demonstrated cancer preventive activity by inhibiting the conversion of high grade PIN lesions to cancer 263.
Theaflavins and thearubigins, the abundant polyphenols in black tea have also been shown to possess strong anticancer property. Black tea polyphenols were found to inhibit proliferation and increase apoptosis in Du 145 prostate carcinoma cells. Higher level of insulin like growth factor-1 (IGF-1) was found to be associated with a higher risk of development of prostate cancer. IGF-1 binding to its receptor is a part of signal transduction pathway which causes cell proliferation 109. Black tea polyphenol addition was found to block IGF-1 induced progression of cells into S phase of cell cycle at a dose of 40 mg/ml in prostate carcinoma cells 109.
Quercetin has also been reported to possess anticancer property against benzo(a)pyrene induced lung carcinogenesis in mice, an effect attrtibuted to its free radical scavenging activity. Resveratrol prevents all stages of development of cancer and has been found to be effective in most types of cancer including lung, skin, breast, prostate, gastric and colorectal cancer. It has also been shown to suppress angiogenesis and metastasis. Extensive data in human cell cultures indicate that resveratrol can modulate multiple pathways involved in cell growth, apoptosis and inflammation. The anti-carcinogenic effects of resveratrol appears to be closely associated with its antioxidant activity, and it has been shown to inhibit cyclooxygenase, hydroperoxidase, protein kinase C, Bcl-2 phosphorylation, Akt, focal adhesion kinase, NFκB, matrix metalloprotease-9 and cell cycle regulators. These and other in vitro and in vivo studies provide a rationale in support of the use of dietary polyphenols in human cancer chemoprevention, in a combinatorial approach with either chemotherapeutic drugs or cytotoxic factors for efficient treatment of drug refractory tumor cells.
Polyphenols Anti-Diabetic Effect
Impairment in glucose metabolism leads to physiological imbalance with the onset of the hyperglycemia and subsequently diabetes mellitus. There are two main categories of diabetes; type-1 and type-2. Studies have shown that several physiological parameters of the body get altered in the diabetic conditions 264, 265. Long term effects of diabetes include progressive development of specific complements such as retinopathy, which affects eyes and lead to blindness; nephropathy in which the renal functions are altered or disturbed and neuropathy which is associated with the risks of amputations, foot ulcers and features of autonomic disturbance including sexual dysfunctions. Numerous studies report the antidiabetic effects of polyphenols. Tea catechins have been investigated for their anti-diabetic potential 266, 267. Polyphenols may affect glycemia through different mechanisms, including the inhibition of glucose absorption in the gut or of its uptake by peripheral tissues. The hypoglycemic effects of diacetylated anthocyanins at a 10 mg/kg diet dosage were observed with maltose as a glucose source, but not with sucrose or glucose 268. This suggests that these effects are due to an inhibition of α-glucosidase in the gut mucosa. Inhibition of α-amylase and sucrase in rats by catechin at a dose of about 50 mg/kg diet or higher was also observed.
The inhibition of intestinal glycosidases and glucose transporter by polyphenols has been studied. Individual polyphenols, such as (+)catechin, (−)epicatechin, (−)epigallocatechin, epicatechin gallate, isoflavones from soyabeans, tannic acid, glycyrrhizin from licorice root, chlorogenic acid and saponins also decrease S-Glut-1 mediated intestinal transport of glucose. Saponins additionally delay the transfer of glucose from stomach to the small intestine. Resveratrol has also been reported to act as an anti-diabetic agent. Many mechanisms have been proposed to explain the anti-diabetic action of this stilbene, modulation of SIRT1 is one of them which improves whole-body glucose homeostasis and insulin sensitivity in diabetic rats. It is reported that in cultured LLC-PK1 cells, high glucose induced cytotoxicity and oxidative stress was inhibited by grape seed polyphenols. Resveratrol inhibits diabetes-induced changes in the kidney (diabetic nephropathy) and significantly ameliorates renal dysfunction and oxidative stress in diabetic rats. Treatment with resveratrol also decreased insulin secretion and delayed the onset of insulin resistance. A possible mechanism was thought to be related to the inhibition of K + ATP and K + V channel in beta cells.
Onion polyphenols, especially quercetin is known to possess strong anti diabetic activity. A recent study shows that quercetin has ability to protect the alterations in diabetic patients during oxidative stress. Quercetin significantly protected the lipid peroxidation and inhibition antioxidant system in diabetics. Hibiscus sabdariffa extract contains polyphenolic acids, flavonoids, protocatechuic acid and anthocyanins. A study performed by Lee et al. 269 showed that polyphenols present in the extracts from Hibiscus sabdariffa attenuate diabetic nephropathy including pathology, serum lipid profile and oxidative markers in kidney. Ferulic acid (FA) is another polyphenol very abundant in vegetables and maize bran. Several lines of evidence have shown that ferulic acid acts as a potent anti-diabetic agent by acting at many levels. It was demonstrated that FA lowered blood glucose followed by a significantly increased plasma insulin and a negative correlation between blood glucose and plasma insulin 270, 271.
Polyphenols Anti-Aging Effect
Aging is the accumulation process of diverse detrimental changes in the cells and tissues with advancing age, resulting in an increase in the risks of disease and death. Among many theories purposed for the explaining the mechanism of aging, free radical/oxidative stress theory is one of the most accepted one 272. A certain amount of oxidative damage takes place even under normal conditions; however, the rate of this damage increases during the aging process as the efficiency of antioxidative and repair mechanisms decrease. Antioxidant capacity of the plasma is related to dietary intake of antioxidants; it has been found that the intake of antioxidant rich diet is effective in reducing the deleterious effects of aging and behavior. Several researches suggest that the combination of antioxidant/anti-inflammatory polyphenolic compounds found in fruits and vegetables may show efficacy as anti-aging compounds 273, 274. Subset of the flavonoids known as anthocyanins, are particularly abundant in brightly colored fruits such as berry fruits and concord grapes and grape seeds. Anthocyanins are responsible for the colors in fruits, and they have been shown to have potent antioxidant/anti-inflammatory activities, as well as to inhibit lipid peroxidation and the inflammatory mediators cyclo-oxygenase (COX)-1 and -2 275.
Fruit and vegetable extracts that have high levels of flavonoids also display high total antioxidant activity such as spinach, strawberries and blueberries. It is reported that the dietary supplementations (for 8 weeks) with spinach, strawberry or blueberry extracts in a control diet were also effective in reversing age-related deficits in brain and behavioral function in aged rats 276. A recent study demonstrates that the tea catechins carry strong anti-aging activity and consuming green-tea rich in these catechins, may delay the onset of aging 277.
Polyphenols are also beneficial in ameliorating the adverse effects of the aging on nervous system or brain. Paramount importance for the relevance of food polyphenols in the protection of the aging brain is the ability of these compounds to cross the blood-brain barrier (BBB), which tightly controls the influx in the brain of metabolites and nutrients as well as of drugs. Resveratrol has been found to consistently prolong the life span; its action is linked to an event called caloric restriction or partial food deprivation 256.
Grape polyphenol, resveratrol is very recent entry as an antiaging agent. It has been shown that the early target of the resveratrol is the sirtuin class of nicotinamide adenine dinucleotide (NAD)-dependent deacetylases. Seven sirtuins have been identified in mammals, of which SIRT-1 is believed to mediate the beneficial effects on health and longevity of both caloric restriction and resveratro 278l. Resveratrol increased insulin sensitivity, decreased the expression of IGF-1 and increased AMP-activated protein kinase (AMPK) and peroxisome proliferator-activated receptor-c coactivator 1a (PGC-1a) activity. When examined for the mechanism, it activated forkhead box O (FOXO), which regulates the expression of genes that contribute both to longevity and resistance to various stresses and insulin-like growth factorbinding protein 1 (IGFBP-1) 279. There are experimental evidences that resveratrol can extend lifespan in the yeast Saccharomyces cerevisiae, the fruit fly Drosophila melanogaster, the nematode worm C. elegans, and seasonal fish Nothobranchius furzeri. Recently quercetin has also been reported to exert preventive effect against aging 280.
Polyphenols Neuro-Protective Effects
Oxidative stress and damage to brain macromolecules is an important process in neurodegenerative diseases. Alzheimer’s disease is one of the most common occurring neurodisorder affecting up to 18 million people worldwide. Because polyphenols are highly antioxidative in nature, their consumption may provide protection in neurological diseases 281. It was observed that the people drinking three to four glasses of wine per day had 80% decreased incidence of dementia and Alzheimer’s disease compared to those who drank less or did not drink at all 282.
Resveratrol, abundantly present in wine scavenges O2− and OH· in vitro, as well as lipid hydroperoxyl free radicals, this efficient antioxidant activity is probably involved in the beneficial effect of the moderate consume of red wine against dementia in the elderly. Resveratrol inhibits nuclear factor κB signaling and thus gives protection against microglia-dependent β-amyloid toxicity in a model of Alzheimer’s disease and this activity is related with the activation of the SIRT-1. It was found that the consumption of fruit and vegetable juices containing high concentrations of polyphenols, at least three times per week, may play an important role in delaying the onset of Alzheimer’s disease 283. Polyphenols from fruits and vegetables seem to be invaluable potential agents in neuroprotection by virtue of their ability to influence and modulate several cellular processes such as signaling, proliferation, apoptosis, redox balance and differentiation 284.
Recently Aquilano et al. 285 reported that administration of polyphenols provide protective effects against Parkinson’s disease, a neurological disorder characterized by degeneration of dopaminergic neurons in the substantia nigra zona compacta. Nutritional studies have linked the consumption of green tea to the reduced risk of developing Parkinson’s disease. In animal models epigallocatechin gallate (EGCG) has been shown to exert a protective role against the neurotoxin MPTP (N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine), an inducer of a Parkinson’s-like disease, either by competitively inhibiting the uptake of the drug, due to molecular similarity or by scavenging MPTP-mediated radical formation. EGCG may also protect neurons by activating several signaling pathways, involving MAP kinases which are fundamental for cell survival. The therapeutic role of catechins in Parkinson’s disease is also due to their ability to chelate iron. This property contributes to their antioxidant activity by preventing redox-active transition metal from catalyzing free radicals formation. Moreover, the antioxidant function is also related to the induction of the expression of antioxidant and detoxifying enzymes particularly in the brain, which is not sufficiently endowed of a well-organized antioxidant defense system.89 Maize bran polyphenol, ferulic acid is also reported to be beneficial in Alzheimer’s disease. This effect is due to its antioxidant and anti-inflammatory properties.
Polyphenols Summary
Polyphenols are a group of powerful plant-derived antioxidants that include a wide range of compounds, such as phenolic acids, flavonoids (flavonols, flavanones, flavan-3-ols, flavones, anthocyanins, and isoflavones), stilbenes, lignans, tannins and coumarins 286, 287, 288. Fruits like grapes, apple, pear, cherries and berries contains up to 200–300 mg polyphenols per 100 grams fresh weight and beverages such as tea and red wine represent the main sources of polyphenols, but vegetables, legumes, and cereals are also important sources. Other food sources of polyphenols are seasonings and dried herbs (e.g., cloves, peppermint, anise, oregano, and rosemary), green tea, cocoa, dark-colored berries, some seeds and nuts (e.g., flaxseed, soybean, chestnut, and hazelnut), and some vegetables (e.g., black olives, globe artichoke heads, and red chicory) 289:S112–S120. doi: 10.1038/ejcn.2010.221)), 290. Furthermore, various fresh, frozen, and dried fruits should also be taken into account (e.g., pomegranate and tropical fruits) 291, 292, 293:3–23. doi: 10.1007/s40279-018-0998-x)). Many people take polyphenols in the form of dietary supplements that contain either purified polyphenols (e.g., quercetin and resveratrol) or extracts from raw materials which are rich in these ingredients (such as extracts from tea, red wine, grapes, blueberries, pomegranate, etc.) 294, 295, 293:3–23. doi: 10.1007/s40279-018-0998-x)), 296, 297, 298. Polyphenols or polyphenol rich diets due to their antioxidant, anti-platelet, antimicrobial and anti-inflammatory properties provide significant protection against the development and progression of many chronic pathological conditions including cancer, metabolic syndrome (which includes type 2 diabetes mellitus, central and abdominal obesity, systemic hypertension, and atherogenic dyslipidaemia), cardiovascular diseases (e.g., atherosclerosis, myocardial infarction, heart failure, and stroke) and aging 4, 2, 299, 300, 301, 302, 303, 304, 305, 306, 307. Although several biological effects based on epidemiological studies can be scientifically explained, the mechanism of action of some effects of polyphenols is not fully understood. However based on our current scientific understanding, polyphenols offer great hope for the prevention of chronic human diseases.
- Bolat E, Sarıtaş S, Duman H, Eker F, Akdaşçi E, Karav S, Witkowska AM. Polyphenols: Secondary Metabolites with a Biological Impression. Nutrients. 2024 Aug 3;16(15):2550. doi: 10.3390/nu16152550[↩][↩]
- Farhan M. The Promising Role of Polyphenols in Skin Disorders. Molecules. 2024 Feb 15;29(4):865. doi: 10.3390/molecules29040865[↩][↩][↩]
- Vicente-Zurdo D, Gómez-Mejía E, Rosales-Conrado N, León-González ME. A Comprehensive Analytical Review of Polyphenols: Evaluating Neuroprotection in Alzheimer’s Disease. Int J Mol Sci. 2024 May 28;25(11):5906. doi: 10.3390/ijms25115906[↩][↩][↩]
- Duda-Chodak A, Tarko T. Possible Side Effects of Polyphenols and Their Interactions with Medicines. Molecules. 2023 Mar 10;28(6):2536. doi: 10.3390/molecules28062536[↩][↩]
- Grabska-Kobyłecka I, Szpakowski P, Król A, Książek-Winiarek D, Kobyłecki A, Głąbiński A, Nowak D. Polyphenols and Their Impact on the Prevention of Neurodegenerative Diseases and Development. Nutrients. 2023 Aug 4;15(15):3454. doi: 10.3390/nu15153454[↩]
- Benkhouili F.Z., Moutawalli A., Ouchari L., El Fahime E., Benzeid H., Doukkali A., Zahidi A. Evaluation of the Content of Polyphenols, Flavonoids and Tannins, the Antioxidant Capacity, and the Antimicrobial Activity of Different Organic and Aqueous Fractions of Stems of Retama Monosperma. Plant Sci. Today. 2024;11 doi: 10.14719/pst.2944[↩][↩]
- Pandey KB, Rizvi SI. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Medicine and Cellular Longevity. 2009;2(5):270-278. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2835915/[↩][↩][↩][↩][↩]
- Dietary polyphenols and the prevention of diseases. Scalbert A, Manach C, Morand C, Rémésy C, Jiménez L. Crit Rev Food Sci Nutr. 2005; 45(4):287-306. https://www.ncbi.nlm.nih.gov/pubmed/16047496/[↩]
- Biomarkers of the intake of dietary polyphenols: strengths, limitations and application in nutrition research. Spencer JP, Abd El Mohsen MM, Minihane AM, Mathers JC. Br J Nutr. 2008 Jan; 99(1):12-22. https://www.ncbi.nlm.nih.gov/pubmed/17666146/[↩][↩]
- Jovanović A.A., Djordjević V.B., Petrović P.M., Pljevljakušić D.S., Zdunić G.M., Šavikin K.P., Bugarski B.M. The Influence of Different Extraction Conditions on Polyphenol Content, Antioxidant and Antimicrobial Activities of Wild Thyme. J. Appl. Res. Med. Aromat. Plants. 2021;25:100328. doi: 10.1016/j.jarmap.2021.100328[↩][↩]
- Dong R., Yu Q., Liao W., Liu S., He Z., Hu X., Chen Y., Xie J., Nie S., Xie M. Composition of Bound Polyphenols from Carrot Dietary Fiber and Its in Vivo and in Vitro Antioxidant Activity. Food Chem. 2021;339:127879. doi: 10.1016/j.foodchem.2020.127879[↩][↩]
- Banica F., Bungau S., Tit D.M., Behl T., Otrisal P., Nechifor A.C., Gitea D., Pavel F.-M., Nemeth S. Determination of the Total Polyphenols Content and Antioxidant Activity of Echinacea Purpurea Extracts Using Newly Manufactured Glassy Carbon Electrodes Modified with Carbon Nanotubes. Processes. 2020;8:833. doi: 10.3390/pr8070833[↩][↩]
- Zhang Y., Lan M., Lü J., Li J., Zhang K., Zhi H., Zhang H., Sun J. Antioxidant, Anti-inflammatory and Cytotoxic Activities of Polyphenols Extracted from Chroogomphus Rutilus. Chem. Biodivers. 2020;17:e1900479. doi: 10.1002/cbdv.201900479[↩][↩]
- PM S., Tungare K., Sunariwal S., Sonawane S. Extraction and Characterization of Polyphenols from Artocarpus Heterophyllus and Its Effect on Oxidative Stability of Peanut Oil. Int. J. Fruit Sci. 2020;20:S1134–S1155. doi: 10.1080/15538362.2020.1775162[↩]
- Zunino S.J., Storms D.H., Stephensen C.B. Diets Rich in Polyphenols and Vitamin A Inhibit the Development of Type I Autoimmune Diabetes in Nonobese Diabetic Mice. J. Nutr. 2007;137:1216–1221. doi: 10.1093/jn/137.5.1216[↩]
- antos S.C., Fortes G.A.C., Camargo L.T.F.M., Camargo A.J., Ferri P.H. Antioxidant Effects of Polyphenolic Compounds and Structure-Activity Relationship Predicted by Multivariate Regression Tree. LWT. 2021;137:110366. doi: 10.1016/j.lwt.2020.110366[↩]
- Nowak M., Tryniszewski W., Sarniak A., Wlodarczyk A., Nowak P.J., Nowak D. Concentration Dependence of Anti- and Pro-Oxidant Activity of Polyphenols as Evaluated with a Light-Emitting Fe2+-Egta-H2O2 System. Molecules. 2022;27:3453. doi: 10.3390/molecules27113453[↩][↩]
- Kumar Y., Singhal S., Tarafdar A., Pharande A., Ganesan M., Badgujar P.C. Ultrasound Assisted Extraction of Selected Edible Macroalgae: Effect on Antioxidant Activity and Quantitative Assessment of Polyphenols by Liquid Chromatography with Tandem Mass Spectrometry (LC-MS/MS) Algal Res. 2020;52:102114. doi: 10.1016/j.algal.2020.102114[↩][↩]
- Sarıtaş S., Duman H., Pekdemir B., Rocha J.M., Oz F., Karav S. Functional Chocolate: Exploring Advances in Production and Health Benefits. Int. J. Food Sci. Technol. 2024;59:5303–5325. doi: 10.1111/ijfs.17312[↩]
- Ozen I.T., Karav S., Eksi A. Variability of Sorbitol/Xylitol Content in Pomegranate (Punica granatum) Juice As Affected by Processing Conditions. Int. J. Food Nutr. Sci. 2014;3:4–7.[↩]
- Im Y.R., Kim I., Lee J. Phenolic Composition and Antioxidant Activity of Purple Sweet Potato (Ipomoea batatas (L.) Lam.): Varietal Comparisons and Physical Distribution. Antioxidants. 2021;10:462. doi: 10.3390/antiox10030462[↩][↩]
- Flavonols, flavones, flavanones, and human health: epidemiological evidence. Graf BA, Milbury PE, Blumberg JB. J Med Food. 2005 Fall; 8(3):281-90. https://www.ncbi.nlm.nih.gov/pubmed/16176136/[↩]
- Polyphenols and disease risk in epidemiologic studies. Arts IC, Hollman PC. Am J Clin Nutr. 2005 Jan; 81(1 Suppl):317S-325S. https://www.ncbi.nlm.nih.gov/pubmed/15640497/[↩]
- Dufresne C. J., Farnworth E. R. A review of latest research findings on the health promotion properties of tea. Journal of Nutritional Biochemistry. 2001;12(7):404–421. doi: 10.1016/S0955-2863(01)00155-3. https://www.ncbi.nlm.nih.gov/pubmed/11448616[↩]
- Khan N., Mukhtar H. Cancer and metastasis: prevention and treatment by green tea. Cancer and Metastasis Reviews. 2010;29(3):435–445. doi: 10.1007/s10555-010-9236-1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3142888/[↩]
- Aggarwal B. B., Shishodia S. Molecular targets of dietary agents for prevention and therapy of cancer. Biochemical Pharmacology. 2006;71(10):1397–1421. doi: 10.1016/j.bcp.2006.02.009. https://www.ncbi.nlm.nih.gov/pubmed/16563357[↩]
- Garrido G., González D., Lemus Y., et al. In vivo and in vitro anti-inflammatory activity of Mangifera indica L. extract (VIMANG) Pharmacological Research. 2004;50(2):143–149. doi: 10.1016/j.phrs.2003.12.003. https://www.ncbi.nlm.nih.gov/pubmed/15177302[↩]
- Nichenametla S. N., Taruscio T. G., Barney D. L., Exon J. H. A review of the effects and mechanisms of polyphenolics in cancer. Critical Reviews in Food Science and Nutrition. 2006;46(2):161–183. doi: 10.1080/10408390591000541. https://www.ncbi.nlm.nih.gov/pubmed/16431408[↩]
- Zaveri N. T. Green tea and its polyphenolic catechins: medicinal uses in cancer and noncancer applications. Life Sciences. 2006;78(18):2073–2080. doi: 10.1016/j.lfs.2005.12.006. https://www.ncbi.nlm.nih.gov/pubmed/16445946[↩]
- Kondratyuk TP, Pezzuto JM. Natural Product Polyphenols of Relevance to Human Health. Pharm Biol. 2004;42:46–63.[↩]
- British Journal of Nutrition, Volume 99, Issue 1, January 2008 , pp. 12-22. Biomarkers of the intake of dietary polyphenols: strengths, limitations and application in nutrition research. https://www.cambridge.org/core/journals/british-journal-of-nutrition/article/biomarkers-of-the-intake-of-dietary-polyphenols-strengths-limitations-and-application-in-nutrition-research/BBAB7EA49BCD0434EFB38098BDAA387C[↩]
- Charis M. Galanakis. In: Galanakis C.M., editor. Food Bioactives and Health. Springer International Publishing; Cham, Switzerland: 2021.[↩][↩][↩][↩]
- Verma C. Science and Engineering of Polyphenols: Fundamentals and Industrial Scale Applications. Wiley Online Library; Hoboken, NJ, USA: 2024.[↩][↩][↩]
- Di Ferdinando M., Brunetti C., Fini A., Tattini M. Abiotic Stress Responses in Plants. Springer; New York, NY, USA: 2012. Flavonoids as Antioxidants in Plants Under Abiotic Stresses; pp. 159–179.[↩]
- Dias M.C., Pinto D.C.G.A., Silva A.M.S. Plant Flavonoids: Chemical Characteristics and Biological Activity. Molecules. 2021;26:5377. doi: 10.3390/molecules26175377[↩]
- Bondonno C.P., Bondonno N.P., Dalgaard F., Murray K., Gardener S.L., Martins R.N., Rainey-Smith S.R., Cassidy A., Lewis J.R., Croft K.D., et al. Flavonoid Intake and Incident Dementia in the Danish Diet, Cancer, and Health Cohort. Alzheimers Dement. Transl. Res. Clin. Interv. 2021;7:e12175. doi: 10.1002/trc2.12175[↩]
- Rees A., Dodd G., Spencer J. The Effects of Flavonoids on Cardiovascular Health: A Review of Human Intervention Trials and Implications for Cerebrovascular Function. Nutrients. 2018;10:1852. doi: 10.3390/nu10121852[↩]
- Bhosale P.B., Ha S.E., Vetrivel P., Kim H.H., Kim S.M., Kim G.S. Functions of Polyphenols and Its Anticancer Properties in Biomedical Research: A Narrative Review. Transl. Cancer Res. 2020;9:7619–7631. doi: 10.21037/tcr-20-2359[↩]
- Luo J., Si H., Jia Z., Liu D. Dietary Anti-Aging Polyphenols and Potential Mechanisms. Antioxidants. 2021;10:283. doi: 10.3390/antiox10020283[↩]
- Hussain T., Tan B., Yin Y., Blachier F., Tossou M.C.B., Rahu N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid. Med. Cell. Longev. 2016;2016:7432797. doi: 10.1155/2016/7432797[↩]
- Price SF, Breen PJ, Valladao M, Watson BT. Cluster sun exposure and quercetin in Pinot noir grapes and wine. Am J Enol Vitic 1995;46:187–94. http://www.ajevonline.org/content/46/2/187?ijkey=eb1b4a5d9d46a5a9a1e49a17a1b5cdad9edcb61c&keytype2=tf_ipsecsha [↩]
- Herrmann K. Flavonols and flavones in food plants: a review. J Food Technol 1976;11:433–48.[↩]
- Shahidi F, Naczk M. Food phenolics, sources, chemistry, effects, applications. Lancaster, PA: Technomic Publishing Co Inc; 1995.[↩]
- Hatcher DW, Kruger JE. Simple phenolic acids in flours prepared from Canadian wheat: relationship to ash content, color, and polyphenol oxidase activity. Cereal Chem 1997;74:337–43.[↩]
- Tomas-Barberan FA, Clifford MN. Dietary hydroxybenzoic acid derivatives and their possible role in health protection. J Sci Food Agric 2000;80:1024–32.[↩]
- Clifford MN, Scalbert A. Ellagitannins—occurrence in food, bioavailability and cancer prevention. J Food Sci Agric 2000;80:1118–25.[↩]
- Clifford MN. Chlorogenic acids and other cinnamates—nature, occurence and dietary burden. J Sci Food Agric 1999;79:362–72.[↩]
- Macheix J-J, Fleuriet A, Billot J. Fruit phenolics. Boca Raton, FL: CRC Press, 1990.[↩]
- Rivière C., Pawlus A.D., Mérillon J.-M. Natural Stilbenoids: Distribution in the Plant Kingdom and Chemotaxonomic Interest in Vitaceae. Nat. Prod. Rep. 2012;29:1317. doi: 10.1039/c2np20049j[↩]
- Chong J., Poutaraud A., Hugueney P. Metabolism and Roles of Stilbenes in Plants. Plant Sci. 2009;177:143–155. doi: 10.1016/j.plantsci.2009.05.012[↩]
- Wahab A., Gao K., Jia C., Zhang F., Tian G., Murtaza G., Chen J. Significance of Resveratrol in Clinical Management of Chronic Diseases. Molecules. 2017;22:1329. doi: 10.3390/molecules22081329[↩]
- Raposo R., Chinnici F., Ruiz-Moreno M.J., Puertas B., Cuevas F.J., Carbú M., Guerrero R.F., Ortíz-Somovilla V., Moreno-Rojas J.M., Cantos-Villar E. Sulfur Free Red Wines through the Use of Grapevine Shoots: Impact on the Wine Quality. Food Chem. 2018;243:453–460. doi: 10.1016/j.foodchem.2017.09.111[↩]
- El Tannir H., Houhou D., Debs E., Koubaa M., Jammoul A., Azakir B., Khalil M.I., El Darra N., Louka N. Optimization of Aqueous Extraction of Polyphenols from Cuminum Cyminum Seeds Using Response Surface Methodology and Assessment of Biological Activity. BioTech. 2024;13:7. doi: 10.3390/biotech13010007[↩][↩]
- Phyto-oestrogens and Western diseases. Adlercreutz H, Mazur W. Ann Med. 1997 Apr; 29(2):95-120. https://www.ncbi.nlm.nih.gov/pubmed/9187225/[↩]
- Li W., Chen H., Xu B., Wang Y., Zhang C., Cao Y., Xing X. Research Progress on Classification, Sources and Functions of Dietary Polyphenols for Prevention and Treatment of Chronic Diseases. J. Futur. Foods. 2023;3:289–305. doi: 10.1016/j.jfutfo.2023.03.001[↩]
- Critical Reviews in Clinical Laboratory Sciences. Crit. Rev. Clin. Lab. Sci. 2010;47:1–4. doi: 10.3109/10408360903507283[↩][↩]
- Thompson LU, Robb P, Serraino M, Cheung F. Mammalian lignan production from various foods. Nutr Cancer 1991;16:43–52. https://www.ncbi.nlm.nih.gov/pubmed/1656395?dopt=Abstract[↩]
- Adlercreutz H. Lignans and Human Health. Crit. Rev. Clin. Lab. Sci. 2007;44:483–525. doi: 10.1080/10408360701612942[↩]
- Chen J., Thompson L.U. Lignans and Tamoxifen, Alone or in Combination, Reduce Human Breast Cancer Cell Adhesion, Invasion and Migration in Vitro. Breast Cancer Res. Treat. 2003;80:163–170. doi: 10.1023/A:1024513815374[↩]
- Rickard S.E., Yuan Y.V., Chen J., Thompson L.U. Dose Effects of Flaxseed and Its Lignan on N-Methyl-N-Nitrosourea-Induced Mammary Tumorigenesis in Rats. Nutr. Cancer. 1999;35:50–57. doi: 10.1207/S1532791450-57[↩]
- Dabrosin C., Chen J., Wang L., Thompson L.U. Flaxseed Inhibits Metastasis and Decreases Extracellular Vascular Endothelial Growth Factor in Human Breast Cancer Xenografts. Cancer Lett. 2002;185:31–37. doi: 10.1016/S0304-3835(02)00239-2[↩]
- Bylund A., Saarinen N., Zhang J., Bergh A., Widmark A., Johansson A., Lundin E., Adlercreutz H., Hallmans G., Stattin P., et al. Anticancer Effects of a Plant Lignan 7-Hydroxymatairesinol on a Prostate Cancer Model In Vivo. Exp. Biol. Med. 2005;230:217–223. doi: 10.1177/153537020523000308[↩]
- Teodor E.D., Moroeanu V., Radu G.L. Lignans from Medicinal Plants and Their Anticancer Effect. Mini-Reviews Med. Chem. 2020;20:1083–1090. doi: 10.2174/1389557520666200212110513[↩]
- Wang C.Y., Qin F., Wang C.-G., Kim D., Li J.-J., Chen X.-L., Wang H.-S., Lee S.K. Novel Lignans from Zanthoxylum Nitidum and Antiproliferation Activity of Sesaminone in Osimertinib-Resistant Non-Small Cell Lung Cancer Cells. Bioorganic Chem. 2023;134:106445. doi: 10.1016/j.bioorg.2023.106445[↩]
- Bayar İ., Çağlar Yavuz S., Akkoç S. In Vitro and In Silico Studies on Lignan SecoisolariciresinolL Diglycoside. Ank. Univ. Eczaci. Fak. Derg. 2023;48:127–137. doi: 10.33483/jfpau.1368474[↩]
- Jang W.Y., Kim M.-Y., Cho J.Y. Antioxidant, Anti-Inflammatory, Anti-Menopausal, and Anti-Cancer Effects of Lignans and Their Metabolites. Int. J. Mol. Sci. 2022;23:15482. doi: 10.3390/ijms232415482[↩]
- Tago R., Yamauchi S., Maruyama M., AKIYAMA K., SUGAHARA T., KISHIDA T., KOBA Y. Structure-Antibacterial Activity Relationship for 9-O,9′-O-Demethyl (+)-Virgatusin. Biosci. Biotechnol. Biochem. 2008;72:1032–1037. doi: 10.1271/bbb.70783[↩]
- Chen D.-F., Zhang S.-X., Chen K., Zhou B.-N., Wang P., Cosentino L.M., Lee K.-H. Two New Lignans, Interiotherins A and B, as Anti-HIV Principles from Kadsura Interior. J. Nat. Prod. 1996;59:1066–1068. doi: 10.1021/np9601667[↩]
- Scalbert A., Johnson I. T., Saltmarsh M. Polyphenols: antioxidants and beyond. The American Journal of Clinical Nutrition. 2005;81(1):215S–217S. https://www.ncbi.nlm.nih.gov/pubmed/15640483[↩]
- Chabert P., Auger C., Pincemail J., Schini-Kerth V. B. Systems Biology of Free Radicals and Antioxidants. Berlin, Germany: Springer; 2014. Overview of plant-derived antioxidants; pp. 4005–4022.[↩]
- Pérez-Jiménez J., Díaz-Rubio M. E., Saura-Calixto F. Non-extractable polyphenols, a major dietary antioxidant: occurrence, metabolic fate and health effects. Nutrition Research Reviews. 2013;26(2):118–129. doi: 10.1017/s0954422413000097. https://www.ncbi.nlm.nih.gov/pubmed/23930641[↩]
- Ovaskainen M.-L., Törrönen R., Koponen J. M., et al. Dietary intake and major food sources of polyphenols in Finnish adults. Journal of Nutrition. 2008;138(3):562–566. https://www.ncbi.nlm.nih.gov/pubmed/18287367 [↩]
- Thu N. N., Sakurai C., Uto H., et al. The polyphenol content and antioxidant activities of the main edible vegetables in Northern Vietnam. Journal of Nutritional Science and Vitaminology. 2004;50(3):203–210. doi: 10.3177/jnsv.50.203. https://www.ncbi.nlm.nih.gov/pubmed/15386933[↩]
- Saura-Calixto F., Serrano J., Goñi I. Intake and bioaccessibility of total polyphenols in a whole diet. Food Chemistry. 2007;101(2):492–501. doi: 10.1016/j.foodchem.2006.02.006.[↩]
- Kanner J., Lapidot T. The stomach as a bioreactor: dietary lipid peroxidation in the gastric fluid and the effects of plant-derived antioxidants. Free Radical Biology & Medicine. 2001;31(11):1388–1395. doi: 10.1016/s0891-5849(01)00718-3. https://www.ncbi.nlm.nih.gov/pubmed/11728810[↩]
- Kerem Z., Chetrit D., Shoseyov O., Regev-Shoshani G. Protection of lipids from oxidation by epicatechin, trans-resveratrol, and gallic and caffeic acids in intestinal model systems. Journal of Agricultural and Food Chemistry. 2006;54(26):10288–10293. doi: 10.1021/jf0621828. https://www.ncbi.nlm.nih.gov/pubmed/17177572[↩]
- Guyot S, Marnet N, Laraba D, Sanoner P, Drilleau J-F. Reversed-phase HPLC following thiolysis for quantitative estimation and characterization of the four main classes of phenolic compounds in different tissue zones of a French cider apple variety (Malus domestica Var. Kermerrien). J Agric Food Chem 1998;46:1698–705.[↩]
- Sanoner P, Guyot S, Marnet N, Molle D, Drilleau J-F. Polyphenol profiles of French cider apple varieties (Malus domestica sp. ). J Agric Food Chem 1999;47:4847–53. https://www.ncbi.nlm.nih.gov/pubmed/10606541?dopt=Abstract[↩]
- Wink M. Compartmentation of secondary metabolites and xenobiotics in plant vacuoles. Adv Bot Res. 1997;25:141–169.[↩]
- Simon BF, Perez-Ilzarbe J, Hernandez T, Gomez-Cordoves C, Estrella I. Importance of phenolic compounds for the characterization of fruit juices. J Agric Food Sci. 1992;40:1531–1535.[↩]
- Polyphenols: food sources and bioavailability. Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L. Am J Clin Nutr. 2004 May; 79(5):727-47. http://ajcn.nutrition.org/content/79/5/727.long[↩][↩]
- Parr AJ, Bolwell GP. Phenols in the plant and in man. The potential for possible nutritional enhancement of the diet by modifying the phenol content or profile. J Agric Food Chem. 2000;80:985–1012.[↩]
- D’Archivio M, Filesi C, Varì R, Scazzocchio B, Masella R. Bioavailability of the Polyphenols: Status and Controversies. International Journal of Molecular Sciences. 2010;11(4):1321-1342. doi:10.3390/ijms11041321. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2871118/[↩]
- Crozier A, Lean MEJ, McDonald MS, Black C. Quantitative analysis of the flavonoid content of commercial tomatoes, onions, lettuce, and celery. J Agric Food Chem 1997;45:590–5. [↩]
- Friedman M. Chemistry, biochemistry, and dietary role of potato polyphenols. A review. J Agric Food Chem 1997;45:1523–40.[↩]
- Clifford MN. Chlorogenic acids and other cinnamates—nature, occurence, dietary burden, absorption and metabolism. J Sci Food Agric 2000;80:1033–43.[↩]
- Macheix JJ, Fleuriet A. Phenolic acids in fruits. In: Rice-Evans C, Packer L, eds. Flavonoids in health and disease. New York: Marcel Dekker, Inc, 1998:35–59.[↩]
- Vinson JA, Hontz BA. Phenol antioxidant index: comparative antioxidant effectiveness of red and white wines. J Agric Food Chem 1995;43:401–3.[↩]
- Abu-Amsha Caccetta AR, Croft KD, Puddey IB, Proudfoot JM, Beilin LJ. Phenolic content of various beverages determines the extent of inhibition of human serum and low-density lipoprotein oxidation in vitro: identification and mechanism of action of some cinnamic acid derivatives from red wine. Clin Sci 1996;91:449–58. https://www.ncbi.nlm.nih.gov/pubmed/8983870?dopt=Abstract [↩]
- Sosulski FW, Krygier K, Hogge L. Importance of phenolic compounds for the characterization of fruit juices. J Agric Food Chem. 1982;30:337–340.[↩]
- Price KR, Bacon JR, Rhodes MJC. Effect of storage and domestic processing on the content and composition of flavonol glucosides in onion (Allium cepa) J Agric Food Chem. 1997;45:938–942.[↩]
- Crozier A, Lean MEJ, McDonald MS, Black C. Quantitative analysis of the flavonoid content of commercial tomatoes, onions, lettuce, and celery. J Agric Food Chem. 1997;45:590–595.[↩]
- US Department of Agriculture. USDA database for the flavonoid content of selected foods. https://www.ars.usda.gov/ARSUserFiles/80400525/Articles/JAFC54_9966-9977Flavonoid.pdf[↩]
- Polyphenols: food sources and bioavailability. Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L. Am J Clin Nutr. 2004 May; 79(5):727-47. https://ajcn.nutrition.org/article/S0002-9165(22)03914-4/fulltext[↩]
- Polyphenols, dietary sources and bioavailability. D’Archivio M, Filesi C, Di Benedetto R, Gargiulo R, Giovannini C, Masella R. Ann Ist Super Sanita. 2007; 43(4):348-61. https://www.ncbi.nlm.nih.gov/pubmed/18209268/[↩]
- Polyphenols, dietary sources and bioavailability. D’Archivio M, Filesi C, Di Benedetto R, Gargiulo R, Giovannini C, Masella R. Ann Ist Super Sanita. 2007; 43(4):348-61. http://www.iss.it/publ/anna/2007/4/434348.pdf[↩]
- Scalbert A, Manach C, Morand C, Remesy C. Dietary polyphenols and the prevention of diseases. Crit Rev Food Sci Nutr. 2005;45:287–306. https://www.ncbi.nlm.nih.gov/pubmed/16047496[↩][↩]
- Arts ICW, Hollman PCH. Polyphenols and disease risk in epidemiologic studies. Am J Clin Nutr. 2005;81:317–325. https://www.ncbi.nlm.nih.gov/pubmed/15640497[↩]
- Clifford MN. Chlorogenic acids and other cinnamates. Nature, occurence, dietary burden, absorption and metabolism. J Sci Food Agric. 2000;80:1033–1043.[↩]
- Vitrac X, Moni JP, Vercauteren J, Deffieux G, Mérillon JM. Direct liquid chromatography analysis of resveratrol derivatives and flavanonols in wines with absorbance and fluorescence detection. Anal Chim Acta. 2002;458:103–110.[↩]
- Luqman S, Rizvi SI. Protection of lipid peroxidation and carbonyl formation in proteins by capsaicin in human erythrocytes subjected to oxidative stress. Phytother Res. 2006;20:303–306. https://www.ncbi.nlm.nih.gov/pubmed/16557614[↩]
- Pandey KB, Mishra N, Rizvi SI. Protective role of myricetin on markers of oxidative stress in human erythrocytes subjected to oxidative stress. Nat Prod Commun. 2009;4:221–226. https://www.ncbi.nlm.nih.gov/pubmed/19370927[↩]
- Pandey KB, Rizvi SI. Protective effect of resveratrol on markers of oxidative stress in human erythrocytes subjected to in vitro oxidative insult. Phytother Res. 2010 Jan;24 Suppl 1:S11-4. doi: 10.1002/ptr.2853. https://www.ncbi.nlm.nih.gov/pubmed/19441064[↩]
- Woods RK, Raven JM, Wolfe R, Ireland PD, Thien FCK, Abramson MJ. Food and nutrient intakes and asthma risk in young adults. Am J Clin Nutr. 2003;78:414–421. http://ajcn.nutrition.org/content/78/3/414.long[↩][↩]
- Smith LJ, Holbrook JT, Wise R, Blumenthal M, Dozor AJ, Mastronarde J, et al. Dietary intake of soy genistein is associated with lung function in patients with asthma. J Asthma. 2004;41:833–843. https://www.ncbi.nlm.nih.gov/pubmed/15641633[↩][↩]
- Tabak C, Arts ICW, Smit HA, Heederik D, Kromhout D. Chronic obstructive pulmonary disease and intake of catechins, flavonols, and flavones. The MORGEN study. Am J Respir Crit Care Med. 2001;164:61–64. http://www.atsjournals.org/doi/full/10.1164/ajrccm.164.1.2010025[↩]
- Nakajima D, Kim CS, Oh TW, Yang CY, Naka T, Igawa S, Ohta F. Suppressive effects of genistein dosage and resistance exercise on bone loss in ovariectomized rats. J Physiol Anthropol Appl Human Sci. 2001;20:285–291. https://www.jstage.jst.go.jp/article/jpa/20/5/20_5_285/_pdf[↩]
- Kim J, Hwang JS, Cho YK, Han Y, Jeon YJ, Yang KH. Protective effects of (-)-epigallocatechin-3-gallate on UVAand UVB-induced skin damage. Skin Pharmacol Appl Skin Physiol. 2001;14:11–19. https://www.ncbi.nlm.nih.gov/pubmed/11174086[↩]
- Sharma V, Rao LJ. A thought on the biological activities of black tea. Crit Rev Food Sci Nutr. 2009;49:379–404. https://www.ncbi.nlm.nih.gov/pubmed/19399668[↩][↩][↩]
- Kukhtenko H., Bevz N., Konechnyi Y., Kukhtenko O., Jasicka-Misiak I. Spectrophotometric and Chromatographic Assessment of Total Polyphenol and Flavonoid Content in Rhododendron Tomentosum Extracts and Their Antioxidant and Antimicrobial Activity. Molecules. 2024;29:1095. doi: 10.3390/molecules29051095[↩]
- Wojciechowicz-Budzisz A., Pejcz E., Spychaj R., Harasym J. Mixed Psyllium Fiber Improves the Quality, Nutritional Value, Polyphenols and Antioxidant Activity of Rye Bread. Foods. 2023;12:3534. doi: 10.3390/foods12193534[↩]
- Tan S., Lan X., Chen S., Zhong X., Li W. Physical Character, Total Polyphenols, Anthocyanin Profile and Antioxidant Activity of Red Cabbage as Affected by Five Processing Methods. Food Res. Int. 2023;169:112929. doi: 10.1016/j.foodres.2023.112929[↩]
- Vuong Q.V., Pham H.N.T., Negus C. From Herbal Teabag to Infusion—Impact of Brewing on Polyphenols and Antioxidant Capacity. Beverages. 2022;8:81. doi: 10.3390/beverages8040081[↩]
- Janarny G., Ranaweera K.K.D.S., Gunathilake K.D.P.P. Digestive Recovery of Polyphenols, Antioxidant Activity, and Anti-inflammatory Activity of Selected Edible Flowers from the Family Fabaceae. J. Food Biochem. 2022;46:e14052. doi: 10.1111/jfbc.14052[↩]
- Huang D., Li C., Chen Q., Xie X., Fu X., Chen C., Huang Q., Huang Z., Dong H. Identification of Polyphenols from Rosa Roxburghii Tratt Pomace and Evaluation of in Vitro and in Vivo Antioxidant Activity. Food Chem. 2022;377:131922. doi: 10.1016/j.foodchem.2021.131922[↩]
- Mehta D., Yadav K., Chaturvedi K., Shivhare U.S., Yadav S.K. Impact of Cold Plasma on Extraction of Polyphenol From De-Oiled Rice and Corn Bran: Improvement in Extraction Efficiency, In Vitro Digestibility, Antioxidant Activity, Cytotoxicity and Anti-Inflammatory Responses. Food Bioprocess Technol. 2022;15:1142–1156. doi: 10.1007/s11947-022-02801-8[↩]
- Hossain M.N., Sarker U., Raihan M.S., Al-Huqail A.A., Siddiqui M.H., Oba S. Influence of Salinity Stress on Color Parameters, Leaf Pigmentation, Polyphenol and Flavonoid Contents, and Antioxidant Activity of Amaranthus Lividus Leafy Vegetables. Molecules. 2022;27:1821. doi: 10.3390/molecules27061821[↩]
- Bashmil Y.M., Ali A., BK A., Dunshea F.R., Suleria H.A.R. Screening and Characterization of Phenolic Compounds from Australian Grown Bananas and Their Antioxidant Capacity. Antioxidants. 2021;10:1521. doi: 10.3390/antiox10101521[↩]
- Menhem C., Mattar J., Carrillo C., Serhan M. Determination of Polyphenols, Antioxidant Activity, and Antimicrobial Properties of Zhourat Using Different Extraction Conditions. Appl. Food Res. 2021;1:100021. doi: 10.1016/j.afres.2021.100021[↩][↩]
- Alsaud N., Shahbaz K., Farid M. Application of Deep Eutectic Solvents in the Extraction of Polyphenolic Antioxidants from New Zealand Manuka Leaves (Leptospermum scoparium): Optimization and Antioxidant Activity. J. Mol. Liq. 2021;337:116385. doi: 10.1016/j.molliq.2021.116385[↩]
- Kiselova-Kaneva Y., Galunska B., Nikolova M., Dincheva I., Badjakov I. High Resolution LC-MS/MS Characterization of Polyphenolic Composition and Evaluation of Antioxidant Activity of Sambucus Ebulus Fruit Tea Traditionally Used in Bulgaria as a Functional Food. Food Chem. 2022;367:130759. doi: 10.1016/j.foodchem.2021.130759[↩]
- Sánchez-Velázquez O.A., Mulero M., Cuevas-Rodríguez E.O., Mondor M., Arcand Y., Hernández-Álvarez A.J. In Vitro Gastrointestinal Digestion Impact on Stability, Bioaccessibility and Antioxidant Activity of Polyphenols from Wild and Commercial Blackberries (Rubus Spp.) Food Funct. 2021;12:7358–7378. doi: 10.1039/D1FO00986A[↩]
- Bautista-Hernández I., Aranda-Ledesma N.E., Rojas R., Tafolla-Arellano J.C., Martínez-Ávila G.C.G. Antioxidant Activity of Polyphenolic Compounds Obtained from Euphorbia Antisyphilitica By-Products. Heliyon. 2021;7:e06734. doi: 10.1016/j.heliyon.2021.e06734[↩]
- Abdullah S.S.S., Mazlan A.N. Quantification of Polyphenols and Antioxidant Activity in Several Herbal and Green Tea Products in Malaysia. Mater. Today Proc. 2020;31:A106–A113. doi: 10.1016/j.matpr.2020.12.1083[↩]
- Santos S.C., Fortes G.A.C., Camargo L.T.F.M., Camargo A.J., Ferri P.H. Antioxidant Effects of Polyphenolic Compounds and Structure-Activity Relationship Predicted by Multivariate Regression Tree. LWT. 2021;137:110366. doi: 10.1016/j.lwt.2020.110366[↩]
- Bimbiraitė-Survilienė K., Stankevičius M., Šuštauskaitė S., Gęgotek A., Maruška A., Skrzydlewska E., Barsteigienė Z., Akuņeca I., Ragažinskienė O., Lukošius A. Evaluation of Chemical Composition, Radical Scavenging and Antitumor Activities of Satureja hortensis L. Herb Extracts. Antioxidants. 2021;10:53. doi: 10.3390/antiox10010053[↩]
- Jariene E., Lasinskas M., Danilcenko H., Vaitkeviciene N., Slepetiene A., Najman K., Hallmann E. Polyphenols, Antioxidant Activity and Volatile Compounds in Fermented Leaves of Medicinal Plant Rosebay Willowherb (Chamerion angustifolium (L.) Holub) Plants. 2020;9:1683. doi: 10.3390/plants9121683[↩]
- Elez Garofulić I., Kruk V., Martić A., Martić I., Zorić Z., Pedisić S., Dragović S., Dragović-Uzelac V. Evaluation of Polyphenolic Profile and Antioxidant Activity of Pistacia lentiscus L. Leaves and Fruit Extract Obtained by Optimized Microwave-Assisted Extraction. Foods. 2020;9:1556. doi: 10.3390/foods9111556[↩]
- House N.C., Puthenparampil D., Malayil D., Narayanankutty A. Variation in the Polyphenol Composition, Antioxidant, and Anticancer Activity among Different Amaranthus Species. South Afr. J. Bot. 2020;135:408–412. doi: 10.1016/j.sajb.2020.09.026[↩]
- Wojdyło A., Oszmiański J. Antioxidant Activity Modulated by Polyphenol Contents in Apple and Leaves during Fruit Development and Ripening. Antioxidants. 2020;9:567. doi: 10.3390/antiox9070567[↩]
- Li W., Zhang X., He Z., Chen Y., Li Z., Meng T., Li Y., Cao Y. In Vitro and in Vivo Antioxidant Activity of Eucalyptus Leaf Polyphenols Extract and Its Effect on Chicken Meat Quality and Cecum Microbiota. Food Res. Int. 2020;136:109302. doi: 10.1016/j.foodres.2020.109302[↩]
- Tiji S., Benayad O., Berrabah M., El Mounsi I., Mimouni M. Phytochemical Profile and Antioxidant Activity of Nigella Sativa L Growing in Morocco. Sci. World J. 2021;2021:6623609. doi: 10.1155/2021/6623609[↩]
- Ghidossi R., Yammine S., Delsart C., Vitrac X., Mietton Peuchot M. Characterisation of Polyphenols and Antioxidant Potential of Red and White Pomace By-Product Extracts Using Subcritical Water Extraction. OENO One. 2020;54:263–278. doi: 10.20870/oeno-one.2020.54..2346[↩]
- Castaldo L., Narváez A., Izzo L., Graziani G., Ritieni A. In Vitro Bioaccessibility and Antioxidant Activity of Coffee Silverskin Polyphenolic Extract and Characterization of Bioactive Compounds Using UHPLC-Q-Orbitrap HRMS. Molecules. 2020;25:2132. doi: 10.3390/molecules25092132[↩]
- Bobková A., Hudáček M., Jakabová S., Belej Ľ., Capcarová M., Čurlej J., Bobko M., Árvay J., Jakab I., Čapla J., et al. The Effect of Roasting on the Total Polyphenols and Antioxidant Activity of Coffee. J. Environ. Sci. Health Part B. 2020;55:495–500. doi: 10.1080/03601234.2020.1724660[↩]
- Nguyen N.Q., Nguyen M.T., Nguyen V.T., Le V.M., Trieu L.H., Le X.T., Khang T.V., Giang N.T.L., Thach N.Q., Hung T.T. The Effects of Different Extraction Conditions on the Polyphenol, Flavonoids Components and Antioxidant Activity of Polyscias Fruticosa Roots. IOP Conf. Ser. Mater. Sci. Eng. 2020;736:022067. doi: 10.1088/1757-899X/736/2/022067[↩]
- Labhar A., Ahidar N., El-Mernissi Y., Benamari O., Ait Alla K., Jahjah S., Salhi A., Ahari M., Abdellah E., Amhamdi H. Phytochemical, Antioxidant, and Anti-Inflammatory Properties of Tetraclinis Articulata (Vahl) Masters Extracts from Al Hoceima Province. E3S Web Conf. 2024;527:01016. doi: 10.1051/e3sconf/202452701016[↩]
- Bassi S., Benvenuti M., Mirata S., Di Piazza S., Salis A., Damonte G., Zotti M., Scarfì S. Enhanced Antioxidant and Anti-Inflammatory Activity of the Extracts of Pleurotus Ostreatus Edible Mushroom Grown on Lavandula Angustifolia Residues. Food Biosci. 2024;60:104382. doi: 10.1016/j.fbio.2024.104382[↩]
- Gulua L., Shakulashvili N., Tskokilauri A., Kotorashvili T., Pazouki N., Rodrigues K., Aslani M., Ghorbani M., Mgbedo N.E. Saba Rayyani Polyphenol Content, Antioxidant and Anti-Inflammatory Activities of Georgian Wine and Green Tea. World J. Biol. Pharm. Health Sci. 2024;18:246–250. doi: 10.30574/wjbphs.2024.18.2.0276[↩]
- Ozturk T., Ávila-Gálvez M.Á., Mercier S., Vallejo F., Bred A., Fraisse D., Morand C., Pelvan E., Monfoulet L.-E., González-Sarrías A. Impact of Lactic Acid Bacteria Fermentation on (Poly)Phenolic Profile and In Vitro Antioxidant and Anti-Inflammatory Properties of Herbal Infusions. Antioxidants. 2024;13:562. doi: 10.3390/antiox13050562[↩]
- Sim S.-H., Choi H.K., Lee D.E., Na S.C., Hwang D.I., Bin Oh H., Lim Y.T., Kim T.-Y., Kim D.-W. Changes in Physiologically Active Ingredients and Anti-Inflammatory Properties of Underutilized Wild Vegetables by Complex Fermentation Using Beneficial Microorganisms. Food Sci. Preserv. 2024;31:287–297. doi: 10.11002/fsp.2024.31.2.287[↩]
- Costa V., Costa M., Videira R.A., Andrade P.B., Paiva-Martins F. Anti-Inflammatory Activity of Olive Oil Polyphenols—The Role of Oleacein and Its Metabolites. Biomedicines. 2022;10:2990. doi: 10.3390/biomedicines10112990[↩]
- Khare P., Maurya R., Bhatia R., Mangal P., Singh J., Podili K., Bishnoi M., Kondepudi K.K. Polyphenol Rich Extracts of Finger Millet and Kodo Millet Ameliorate High Fat Diet-Induced Metabolic Alterations. Food Funct. 2020;11:9833–9847. doi: 10.1039/D0FO01643H[↩]
- Chen G.-L., Munyao Mutie F., Xu Y.-B., Saleri F.D., Hu G.-W., Guo M.-Q. Antioxidant, Anti-Inflammatory Activities and Polyphenol Profile of Rhamnus Prinoides. Pharmaceuticals. 2020;13:55. doi: 10.3390/ph13040055[↩]
- Derouich M., Bouhlali E.D.T., Hmidani A., Bammou M., Bourkhis B., Sellam K., Alem C. Assessment of Total Polyphenols, Flavonoids and Anti-Inflammatory Potential of Three Apiaceae Species Grown in the Southeast of Morocco. Sci. Afr. 2020;9:e00507. doi: 10.1016/j.sciaf.2020.e00507[↩]
- Peng L., Ai-lati A., Ji Z., Chen S., Mao J. Polyphenols Extracted from Huangjiu Have Anti-Inflammatory Activity in Lipopolysaccharide Stimulated RAW264.7 Cells. RSC Adv. 2019;9:5295–5301. doi: 10.1039/C8RA09671F[↩]
- Mattioli R., Francioso A., D’Erme M., Trovato M., Mancini P., Piacentini L., Casale A., Wessjohann L., Gazzino R., Costantino P., et al. Anti-Inflammatory Activity of A Polyphenolic Extract from Arabidopsis Thaliana in In Vitro and In Vivo Models of Alzheimer’s Disease. Int. J. Mol. Sci. 2019;20:708. doi: 10.3390/ijms20030708[↩]
- Zhang T.-T., Hu T., Jiang J.-G., Zhao J.-W., Zhu W. Antioxidant and Anti-Inflammatory Effects of Polyphenols Extracted from Ilex Latifolia Thunb. RSC Adv. 2018;8:7134–7141. doi: 10.1039/C7RA13569F[↩]
- Ben Salem M., Affes H., Athmouni K., Ksouda K., Dhouibi R., Sahnoun Z., Hammami S., Zeghal K.M. Chemicals Compositions, Antioxidant and Anti-Inflammatory Activity of Cynara Scolymus Leaves Extracts, and Analysis of Major Bioactive Polyphenols by HPLC. Evid.-Based Complement. Altern. Med. 2017;2017:4951937. doi: 10.1155/2017/4951937[↩]
- Siraj M.A., Shilpi J.A., Hossain M.G., Uddin S.J., Islam M.K., Jahan I.A., Hossain H. Anti-Inflammatory and Antioxidant Activity of Acalypha Hispida Leaf and Analysis of Its Major Bioactive Polyphenols by HPLC. Adv. Pharm. Bull. 2016;6:275–283. doi: 10.15171/apb.2016.039[↩]
- Rupasinghe H., Boehm M., Sekhon-Loodu S., Parmar I., Bors B., Jamieson A. Anti-Inflammatory Activity of Haskap Cultivars Is Polyphenols-Dependent. Biomolecules. 2015;5:1079–1098. doi: 10.3390/biom5021079[↩]
- Hooshmand S., Kumar A., Zhang J.Y., Johnson S.A., Chai S.C., Arjmandi B.H. Evidence for Anti-Inflammatory and Antioxidative Properties of Dried Plum Polyphenols in Macrophage RAW 264.7 Cells. Food Funct. 2015;6:1719–1725. doi: 10.1039/C5FO00173K[↩]
- Michel P., Dobrowolska A., Kicel A., Owczarek A., Bazylko A., Granica S., Piwowarski J., Olszewska M. Polyphenolic Profile, Antioxidant and Anti-Inflammatory Activity of Eastern Teaberry (Gaultheria procumbens L.) Leaf Extracts. Molecules. 2014;19:20498–20520. doi: 10.3390/molecules191220498[↩]
- Usha T., Middha S., Bhattacharya M., Lokesh P., Goyal A. Rosmarinic Acid, a New Polyphenol from Baccaurea Ramiflora Lour. Leaf: A Probable Compound for Its Anti-Inflammatory Activity. Antioxidants. 2014;3:830–842. doi: 10.3390/antiox3040830[↩]
- de Araújo A.A., Soares L.A.L., Assunção Ferreira M.R., de Souza Neto M.A., da Silva G.R., de Araújo R.F., Guerra G.C.B., de Melo M.C.N. Quantification of Polyphenols and Evaluation of Antimicrobial, Analgesic and Anti-Inflammatory Activities of Aqueous and Acetone–Water Extracts of Libidibia Ferrea, Parapiptadenia Rigida and Psidium Guajava. J. Ethnopharmacol. 2014;156:88–96. doi: 10.1016/j.jep.2014.07.031[↩]
- García-Lafuente A., Moro C., Manchón N., Gonzalo-Ruiz A., Villares A., Guillamón E., Rostagno M., Mateo-Vivaracho L. In Vitro Anti-Inflammatory Activity of Phenolic Rich Extracts from White and Red Common Beans. Food Chem. 2014;161:216–223. doi: 10.1016/j.foodchem.2014.04.004[↩]
- Grigore A., Colceru-Mihul S., Litescu S., Panteli M., Rasit I. Correlation between Polyphenol Content and Anti-Inflammatory Activity of Verbascum Phlomoides (Mullein) Pharm. Biol. 2013;51:925–929. doi: 10.3109/13880209.2013.767361[↩]
- Kim S.K., Kim H., Kim S.A., Park H.K., Kim W. Anti-Inflammatory and Anti-Superbacterial Activity of Polyphenols Isolated from Black Raspberry. Korean J. Physiol. Pharmacol. 2013;17:73. doi: 10.4196/kjpp.2013.17.1.73[↩]
- Prasad P., Mansoori A., Prajapati N., Tripathi J., Sharma K., Kumar A., Narayan Das S. Phytochemical Screening and Antimicrobial Activities of Guizotia abyssinica L. Leaf and Flower Extracts. J. Nat. Pestic. Res. 2024;9:100083. doi: 10.1016/j.napere.2024.100083[↩]
- Papastavropoulou K., Oz E., Oz F., Proestos C. Polyphenols from Plants: Phytochemical Characterization, Antioxidant Capacity, and Antimicrobial Activity of Some Plants from Different Sites of Greece. Separations. 2022;9:186. doi: 10.3390/separations9080186[↩]
- Chaudhry F., Ahmad M.L., Hayat Z., Ranjha M.M.A.N., Chaudhry K., Elboughdiri N., Asmari M., Uddin J. Extraction and Evaluation of the Antimicrobial Activity of Polyphenols from Banana Peels Employing Different Extraction Techniques. Separations. 2022;9:165. doi: 10.3390/separations9070165[↩]
- Mehdizadeh A., Karimi E., Oskoueian E. Nano-liposomal Encapsulation of Artemisia Aucheri Phenolics as a Potential Phytobiotic against Campylobacter Jejuni Infection in Mice. Food Sci. Nutr. 2022;10:3314–3322. doi: 10.1002/fsn3.2921[↩]
- Caponio G., Noviello M., Calabrese F., Gambacorta G., Giannelli G., De Angelis M. Effects of Grape Pomace Polyphenols and In Vitro Gastrointestinal Digestion on Antimicrobial Activity: Recovery of Bioactive Compounds. Antioxidants. 2022;11:567. doi: 10.3390/antiox11030567[↩]
- Hassirian N., Karimi E., Oskoueian E. Nanoliposome-Encapsulated Phenolic-Rich Fraction from Alcea Rosea as a Dietary Phytobiotic in Mice Challenged by Escherichia Coli. Ann. Microbiol. 2022;72:6. doi: 10.1186/s13213-022-01665-9[↩]
- Nateghi N., Karimi E., Oskoueian E. Nanoliposome-Encapsulated and Non-Encapsulated Phenolics From Achillea Millefolium and Their Biological Function in Mice Challenged by Campylobacter Jejuni: A Comparative Study. Front. Mol. Biosci. 2022;8:832022. doi: 10.3389/fmolb.2021.832022[↩]
- Shamansoori M.T., Karimi E., Oskoueian E. Rheum Ribes Extract-loaded Nanoliposome as a Novel Phytogenic Antibiotic Alternative in Mice Challenged by Escherichia Coli (O157:H7) Biotechnol. Appl. Biochem. 2022;69:2540–2549. doi: 10.1002/bab.2303[↩]
- Kruczek A., Krupa-Małkiewicz M., Lachowicz S., Oszmiański J., Ochmian I. Health-Promoting Capacities of In Vitro and Cultivated Goji (Lycium chinense Mill.) Fruit and Leaves; Polyphenols, Antimicrobial Activity, Macro- and Microelements and Heavy Metals. Molecules. 2020;25:5314. doi: 10.3390/molecules25225314[↩]
- Nichitoi M.M., Josceanu A.M., Isopescu R.D., Isopencu G.O., Geana E.-I., Ciucure C.T., Lavric V. Polyphenolics Profile Effects upon the Antioxidant and Antimicrobial Activity of Propolis Extracts. Sci. Rep. 2021;11:20113. doi: 10.1038/s41598-021-97130-9[↩]
- De Angelis M., Della-Morte D., Buttinelli G., Di Martino A., Pacifici F., Checconi P., Ambrosio L., Stefanelli P., Palamara A.T., Garaci E., et al. Protective Role of Combined Polyphenols and Micronutrients against Influenza A Virus and SARS-CoV-2 Infection In Vitro. Biomedicines. 2021;9:1721. doi: 10.3390/biomedicines9111721[↩]
- Sun S., Huang S., Shi Y., Shao Y., Qiu J., Sedjoah R.-C.A.-A., Yan Z., Ding L., Zou D., Xin Z. Extraction, Isolation, Characterization and Antimicrobial Activities of Non-Extractable Polyphenols from Pomegranate Peel. Food Chem. 2021;351:129232. doi: 10.1016/j.foodchem.2021.129232[↩]
- El Majdoub Y.O., Ginestra G., Mandalari G., Dugo P., Mondello L., Cacciola F. The Digestibility of Hibiscus sabdariffa L. Polyphenols Using an In Vitro Human Digestion Model and Evaluation of Their Antimicrobial Activity. Nutrients. 2021;13:2360. doi: 10.3390/nu13072360[↩]
- Alshuniaber M.A., Krishnamoorthy R., AlQhtani W.H. Antimicrobial Activity of Polyphenolic Compounds from Spirulina against Food-Borne Bacterial Pathogens. Saudi J. Biol. Sci. 2021;28:459–464. doi: 10.1016/j.sjbs.2020.10.029[↩]
- Mansoori A., Singh N., Dubey S.K., Thakur T.K., Alkan N., Das S.N., Kumar A. Phytochemical Characterization and Assessment of Crude Extracts from Lantana camara L. for Antioxidant and Antimicrobial Activity. Front. Agron. 2020;2:582268. doi: 10.3389/fagro.2020.582268[↩]
- Dziedzinski M., Kobus-Cisowska J., Szymanowska D., Stuper-Szablewska K., Baranowska M. Identification of Polyphenols from Coniferous Shoots as Natural Antioxidants and Antimicrobial Compounds. Molecules. 2020;25:3527. doi: 10.3390/molecules25153527[↩]
- Singh S., Sk M.F., Sonawane A., Kar P., Sadhukhan S. Plant-Derived Natural Polyphenols as Potential Antiviral Drugs against SARS-CoV-2 via RNA-dependent RNA Polymerase (RdRp) Inhibition: An in-Silico Analysis. J. Biomol. Struct. Dyn. 2021;39:6249–6264. doi: 10.1080/07391102.2020.1796810[↩]
- Gutiérrez-Venegas G., Gómez-Mora J.A., Meraz-Rodríguez M.A., Flores-Sánchez M.A., Ortiz-Miranda L.F. Effect of Flavonoids on Antimicrobial Activity of Microorganisms Present in Dental Plaque. Heliyon. 2019;5:e03013. doi: 10.1016/j.heliyon.2019.e03013[↩]
- Guo L., Sun Q., Gong S., Bi X., Jiang W., Xue W., Fei P. Antimicrobial Activity and Action Approach of the Olive Oil Polyphenol Extract Against Listeria Monocytogenes. Front. Microbiol. 2019;10:1586. doi: 10.3389/fmicb.2019.01586[↩]
- Musarra-Pizzo M., Ginestra G., Smeriglio A., Pennisi R., Sciortino M.T., Mandalari G. The Antimicrobial and Antiviral Activity of Polyphenols from Almond (Prunus dulcis L.) Skin. Nutrients. 2019;11:2355. doi: 10.3390/nu11102355[↩]
- Silva V., Igrejas G., Falco V., Santos T.P., Torres C., Oliveira A.M.P., Pereira J.E., Amaral J.S., Poeta P. Chemical Composition, Antioxidant and Antimicrobial Activity of Phenolic Compounds Extracted from Wine Industry by-Products. Food Control. 2018;92:516–522. doi: 10.1016/j.foodcont.2018.05.031[↩]
- Prabakaran M., Kim S.-H., Sasireka A., Chandrasekaran M., Chung I.-M. Polyphenol Composition and Antimicrobial Activity of Various Solvent Extracts from Different Plant Parts of Moringa Oleifera. Food Biosci. 2018;26:23–29. doi: 10.1016/j.fbio.2018.09.003[↩]
- Fei P., Ali M.A., Gong S., Sun Q., Bi X., Liu S., Guo L. Antimicrobial Activity and Mechanism of Action of Olive Oil Polyphenols Extract against Cronobacter Sakazakii. Food Control. 2018;94:289–294. doi: 10.1016/j.foodcont.2018.07.022[↩]
- Afshari M., Rahimmalek M., Miroliaei M. Variation in Polyphenolic Profiles, Antioxidant and Antimicrobial Activity of Different Achillea Species as Natural Sources of Antiglycative Compounds. Chem. Biodivers. 2018;15:e1800075. doi: 10.1002/cbdv.201800075[↩]
- Bouarab-Chibane L., Forquet V., Lantéri P., Clément Y., Léonard-Akkari L., Oulahal N., Degraeve P., Bordes C. Antibacterial Properties of Polyphenols: Characterization and QSAR (Quantitative Structure–Activity Relationship) Models. Front. Microbiol. 2019;10:829. doi: 10.3389/fmicb.2019.00829[↩]
- Ojo A.B., Adanlawo I.G. Antioxidant, Antidiabetic, and Anti-Inflammatory Activities of Flavonoid-Rich Fractions of Solanum Anguivi Lam. Fruit: In Vitro and Ex Vivo Studies. Heliyon. 2024;10:e31895. doi: 10.1016/j.heliyon.2024.e31895[↩]
- Nguyen M.-T., Bui T.-B.-H., Pham V.-H., Tran M.-D., Nguyen Q.-V. Syzygium zeylanicum (L.) DC. Polyphenols Exhibit Anti-Diabetic Activity by Modulation of ACC1, SGLT1, and GLP-1 Genes and Restoration of Gut Microbiota in Overfeeding and High Glucose Exposure-Induced Diabetic Zebrafish. J. Funct. Foods. 2024;112:105921. doi: 10.1016/j.jff.2023.105921[↩]
- Paul B.M., Jagadeesan G., Kannan G., Jegan Raj F., Annadurai Y., Piramanayagam S., Thangaraj P. Exploring the Hypoglycaemic Efficacy of Bio-Accessed Antioxidative Polyphenolics in Thermally Processed Cucumis Dipsaceus Fruits—An in Vitro and in Silico Study. Food Chem. 2024;435:137577. doi: 10.1016/j.foodchem.2023.137577[↩]
- Zuo Z., Pang W., Sun W., Lu B., Zou L., Zhang D., Wang Y. Metallothionein–Kidney Bean Polyphenol Complexes Showed Antidiabetic Activity in Type 2 Diabetic Rats by Improving Insulin Resistance and Regulating Gut Microbiota. Foods. 2023;12:3139. doi: 10.3390/foods12163139[↩]
- Ouahabi S., Loukili E.H., Daoudi N.E., Chebaibi M., Ramdani M., Rahhou I., Bnouham M., Fauconnier M.-L., Hammouti B., Rhazi L., et al. Study of the Phytochemical Composition, Antioxidant Properties, and In Vitro Anti-Diabetic Efficacy of Gracilaria Bursa-Pastoris Extracts. Mar. Drugs. 2023;21:372. doi: 10.3390/md21070372[↩]
- Li H., Beg O.U., Rafie A.R., Kanwal S., Ovalle-Cisneros A., Faison M.O., Siddiqui R.A. Characterization of Green and Yellow Papaya (Carica papaya) for Anti-Diabetic Activity in Liver and Myoblast Cells and Wound-Healing Activity in Fibroblast Cells. Nutrients. 2023;15:1929. doi: 10.3390/nu15081929[↩]
- Vaithiyalingam M., Sumathi D.L., Sabarathinam S. Isolation and In Silico Study of Curcumin from Curcuma Longa and Its Anti-Diabetic Activity. Appl. Biochem. Biotechnol. 2023;195:947–957. doi: 10.1007/s12010-022-04173-3[↩]
- Narayanankutty A., Job J.T., Moothakoottil Kuttithodi A., Sasidharan A., Benil P.B., Ramesh V., Farouk Elsadek M., Rizwana H., Essam El-Din M.M. Proximate Composition, Antioxidant, Anti-Inflammatory and Anti-Diabetic Properties of the Haustorium from Coconut (Cocos nucifera L.) and Palmyra Palm (Borassus flabellifer L.) J. King Saud Univ.-Sci. 2023;35:102404. doi: 10.1016/j.jksus.2022.102404[↩]
- Xia T., Zhang Z., Zhao Y., Kang C., Zhang X., Tian Y., Yu J., Cao H., Wang M. The Anti-Diabetic Activity of Polyphenols-Rich Vinegar Extract in Mice via Regulating Gut Microbiota and Liver Inflammation. Food Chem. 2022;393:133443. doi: 10.1016/j.foodchem.2022.133443[↩]
- Charoensiddhi S., Chanput W.P., Sae-tan S. Gut Microbiota Modulation, Anti-Diabetic and Anti-Inflammatory Properties of Polyphenol Extract from Mung Bean Seed Coat (Vigna radiata L.) Nutrients. 2022;14:2275. doi: 10.3390/nu14112275[↩]
- Mezni F., Stiti B., Fkiri S., Ayari F., Ben Slimane L., Ksouri R., Khaldi A. Phenolic Profile and in Vitro Anti-Diabetic Activity of Acorn from Four African Quercus Species (Q. Suber, Q. Canariensis, Q. Coccifera and Q. Ilex) South Afr. J. Bot. 2022;146:771–775. doi: 10.1016/j.sajb.2021.12.023[↩]
- Sabadashka M., Hertsyk D., Strugała-Danak P., Dudek A., Kanyuka O., Kucharska A.Z., Kaprelyants L., Sybirna N. Anti-Diabetic and Antioxidant Activities of Red Wine Concentrate Enriched with Polyphenol Compounds under Experimental Diabetes in Rats. Antioxidants. 2021;10:1399. doi: 10.3390/antiox10091399[↩]
- Pieczykolan A., Pietrzak W., Gawlik-Dziki U., Nowak R. Antioxidant, Anti-Inflammatory, and Anti-Diabetic Activity of Phenolic Acids Fractions Obtained from Aerva lanata (L.) Juss. Molecules. 2021;26:3486. doi: 10.3390/molecules26123486[↩]
- Draganescu D., Andritoiu C., Hritcu D., Dodi G., Popa M.I. Flaxseed Lignans and Polyphenols Enhanced Activity in Streptozotocin-Induced Diabetic Rats. Biology. 2021;10:43. doi: 10.3390/biology10010043[↩]
- Moloto M.R., Phan A.D.T., Shai J.L., Sultanbawa Y., Sivakumar D. Comparison of Phenolic Compounds, Carotenoids, Amino Acid Composition, In Vitro Antioxidant and Anti-Diabetic Activities in the Leaves of Seven Cowpea (Vigna unguiculata) Cultivars. Foods. 2020;9:1285. doi: 10.3390/foods9091285[↩]
- De Silva A.B.K.H., Rupasinghe H.P.V. Polyphenols Composition and Anti-Diabetic Properties in Vitro of Haskap (Lonicera caerulea L.) Berries in Relation to Cultivar and Harvesting Date. J. Food Compos. Anal. 2020;88:103402. doi: 10.1016/j.jfca.2019.103402[↩]
- El Adaouia Taleb R., Djebli N., Chenini H., Sahin H., Kolayli S. In Vivo and in Vitro Anti-diabetic Activity of Ethanolic Propolis Extract. J. Food Biochem. 2020;44:e13267. doi: 10.1111/jfbc.13267[↩]
- Ali A., Oon C.C., Chua B.L., Figiel A., Chong C.H., Wojdylo A., Turkiewicz I.P., Szumny A., Łyczko J. Volatile and Polyphenol Composition, Anti-Oxidant, Anti-Diabetic and Anti-Aging Properties, and Drying Kinetics as Affected by Convective and Hybrid Vacuum Microwave Drying of Rosmarinus officinalis L. Ind. Crop. Prod. 2020;151:112463. doi: 10.1016/j.indcrop.2020.112463[↩]
- Guo S., Ren X., He K., Chen X., Zhang S., Roller M., Zheng B., Zheng Q., Ho C.-T., Bai N. The Anti-Diabetic Effect of Eight Lagerstroemia Speciosa Leaf Extracts Based on the Contents of Ellagitannins and Ellagic Acid Derivatives. Food Funct. 2020;11:1560–1571. doi: 10.1039/C9FO03091C[↩]
- Sun X., Ye H., Liu J., Wu L., Lin D., Yu Y., Gao F. Assessment of Anti-Diabetic Activity of Peanut Shell Polyphenol Extracts. J. Zhejiang Univ. B. 2018;19:764–775. doi: 10.1631/jzus.B1700401[↩]
- Amrati F.E.-Z., Chebaibi M., Galvão de Azevedo R., Conte R., Slighoua M., Mssillou I., Kiokias S., de Freitas Gomes A., Soares Pontes G., Bousta D. Phenolic Composition, Wound Healing, Antinociceptive, and Anticancer Effects of Caralluma Europaea Extracts. Molecules. 2023;28:1780. doi: 10.3390/molecules28041780[↩][↩]
- Tao K., Guo L., Hu X., Fitzgerald C., Rouzard K., Healy J., Tamura M., Stock J.B., Stock M., Pérez E., et al. Encapsulated Activated Grape Seed Extract: A Novel Formulation with Anti-Aging, Skin-Brightening, and Hydration Properties. Cosmetics. 2021;9:4. doi: 10.3390/cosmetics9010004[↩]
- Gabr S.A., Alghadir A.H. Evaluation of the Biological Effects of Lyophilized Hydrophilic Extract of Rhus Coriaria on Myeloperoxidase (MPO) Activity, Wound Healing, and Microbial Infections of Skin Wound Tissues. Evid.-Based Complement. Altern. Med. 2019;2019:5861537. doi: 10.1155/2019/5861537[↩]
- Jeong D., Lee J., Park S.H., Kim Y.A., Park B.J., Oh J., Sung G.-H., Aravinthan A., Kim J.-H., Kang H., et al. Antiphotoaging and Antimelanogenic Effects of Penthorum Chinense Pursh Ethanol Extract Due to Antioxidant- and Autophagy-Inducing Properties. Oxidative Med. Cell. Longev. 2019;2019:9679731. doi: 10.1155/2019/9679731[↩]
- Joshi H., Hegde A.R., Shetty P.K., Gollavilli H., Managuli R.S., Kalthur G., Mutalik S. Sunscreen Creams Containing Naringenin Nanoparticles: Formulation Development and in Vitro and in Vivo Evaluations. Photodermatol. Photoimmunol. Photomed. 2018;34:69–81. doi: 10.1111/phpp.12335[↩]
- Mamashli F., Meratan A.A., Ghasemi A., Obeidi N., Salmani B., Atarod D., Pirhaghi M., Moosavi-Movahedi F., Mohammad-Zaheri M., Shahsavani M.B., et al. Neuroprotective Effect of Propolis Polyphenol-Based Nanosheets in Cellular and Animal Models of Rotenone-Induced Parkinson’s Disease. ACS Chem. Neurosci. 2023;14:851–863. doi: 10.1021/acschemneuro.2c00605[↩]
- Pacifici F., Salimei C., Pastore D., Malatesta G., Ricordi C., Donadel G., Bellia A., Rovella V., Tafani M., Garaci E., et al. The Protective Effect of a Unique Mix of Polyphenols and Micronutrients against Neurodegeneration Induced by an In Vitro Model of Parkinson’s Disease. Int. J. Mol. Sci. 2022;23:3110. doi: 10.3390/ijms23063110[↩]
- Gan J., Zhang X., Ma C., Sun L., Feng Y., He Z., Zhang H. Purification of Polyphenols from Phyllanthus emblica L. Pomace Using Macroporous Resins: Antioxidant Activity and Potential Anti-Alzheimer’s Effects. J. Food Sci. 2022;87:1244–1256. doi: 10.1111/1750-3841.16028[↩]
- Aditi K., Singh A., Shakarad M.N., Agrawal N. Management of Altered Metabolic Activity in Drosophila Model of Huntington’s Disease by Curcumin. Exp. Biol. Med. 2022;247:152–164. doi: 10.1177/15353702211046927[↩]
- Di Rosa G., Brunetti G., Scuto M., Trovato Salinaro A., Calabrese E.J., Crea R., Schmitz-Linneweber C., Calabrese V., Saul N. Healthspan Enhancement by Olive Polyphenols in C. Elegans Wild Type and Parkinson’s Models. Int. J. Mol. Sci. 2020;21:3893. doi: 10.3390/ijms21113893[↩]
- Ma X., Sun Z., Han X., Li S., Jiang X., Chen S., Zhang J., Lu H. Neuroprotective Effect of Resveratrol via Activation of Sirt1 Signaling in a Rat Model of Combined Diabetes and Alzheimer’s Disease. Front. Neurosci. 2020;13:1400. doi: 10.3389/fnins.2019.01400[↩]
- Abdolahi M., Jafarieh A., Sarraf P., Sedighiyan M., Yousefi A., Tafakhori A., Abdollahi H., Salehinia F., Djalali M. The Neuromodulatory Effects of ω-3 Fatty Acids and Nano-Curcumin on the COX-2/INOS Network in Migraines: A Clinical Trial Study from Gene Expression to Clinical Symptoms. Endocrine, Metab. Immune Disord.-Drug Targets. 2019;19:874–884. doi: 10.2174/1871530319666190212170140[↩]
- Elifani F., Amico E., Pepe G., Capocci L., Castaldo S., Rosa P., Montano E., Pollice A., Madonna M., Filosa S., et al. Curcumin Dietary Supplementation Ameliorates Disease Phenotype in an Animal Model of Huntington’s Disease. Hum. Mol. Genet. 2019;28:4012–4021. doi: 10.1093/hmg/ddz247[↩]
- Qi Y., Shang L., Liao Z., Su H., Jing H., Wu B., Bi K., Jia Y. Intracerebroventricular Injection of Resveratrol Ameliorated Aβ-Induced Learning and Cognitive Decline in Mice. Metab. Brain Dis. 2019;34:257–266. doi: 10.1007/s11011-018-0348-6[↩]
- Zhou Z.D., Xie S.P., Saw W.T., Ho P.G.H., Wang H.Y., Zhou L., Zhao Y., Tan E.K. The Therapeutic Implications of Tea Polyphenols Against Dopamine (DA) Neuron Degeneration in Parkinson’s Disease (PD) Cells. 2019;8:911. doi: 10.3390/cells8080911[↩]
- Abdolahi M., Sarraf P., Javanbakht M.H., Honarvar N.M., Hatami M., Soveyd N., Tafakhori A., Sedighiyan M., Djalali M., Jafarieh A., et al. A Novel Combination of ω-3 Fatty Acids and Nano-Curcumin Modulates Interleukin-6 Gene Expression and High Sensitivity C-Reactive Protein Serum Levels in Patients with Migraine: A Randomized Clinical Trial Study. CNS Neurol. Disord.-Drug Targets. 2018;17:430–438. doi: 10.2174/1871527317666180625101643[↩]
- Zhang Y., Gu J., Wang Y., Zhao Z., Wang Z., Li W. The Anti-Tumor Activity and Critical Active Compounds of Polyphenols from Chinese Dwarf Cherry (Cerasus humilis) J. Berry Res. 2023;13:211–225. doi: 10.3233/JBR-230005[↩]
- Erden Tayhan S., Bilgin S., Yıldırım A., Koç E. Biological Screening of Polyphenol Derivatives for Anti-Proliferative, Anti-Apoptotic and Anti-Migrative Activities in Human Breast Cancer Cell Lines MCF-7. Chem. Biodivers. 2023;20:e202200872. doi: 10.1002/cbdv.202200872[↩]
- da Luz J.R.D., López J.A., Ferreira M.P., de Sousa R.M., e Silva S.V., das Graças Almeida M., Araujo-Silva G. In Vitro Antithrombotic, Antitumor and Antiangiogenic Activities of Green Tea Polyphenols and Its Main Constituent Epigallocatechin-3-Gallate. Processes. 2022;11:76. doi: 10.3390/pr11010076[↩]
- Wang B., Wang J., Zhao X.-H. Bioactivity of Two Polyphenols Quercetin and Fisetin against Human Gastric Adenocarcinoma AGS Cells as Affected by Two Coexisting Proteins. Molecules. 2022;27:2877. doi: 10.3390/molecules27092877[↩]
- PALFI SALAVAT M.-C. Polyphenols Content and In Vitro Antitumour Activity of Hydroal Coholic Ectract of Viscum Album In Two Pigmented and Unpigmented Skin Cancer Cell Lines. Farmacia. 2022;70:807–815. doi: 10.31925/farmacia.2022.5.5[↩]
- Zhang D., Yu Z., Zhao W., Liu J. Assessment of the Anti-Tumor Activity of Cyanidin-3-O-Arabinoside from Apple against APN, JAK, and EZH2 Target Proteins. Food Biosci. 2022;48:101788. doi: 10.1016/j.fbio.2022.101788[↩]
- Wu H., Li C., Cui M., Guo H., Chen S., Du J., Li H., Li Z. Polyphenols from Hippophae Rhamnoides Suppressed Colon Cancer Growth by Regulating MiRNA-Mediated Cell Cycle Arrest and Apoptosis in Vitro and in Vivo. J. Funct. Foods. 2021;87:104780. doi: 10.1016/j.jff.2021.104780[↩]
- Wu W., Xiang F., He F. Polyphenols from Artemisia Argyi Leaves: Environmentally Friendly Extraction under High Hydrostatic Pressure and Biological Activities. Ind. Crop. Prod. 2021;171:113951. doi: 10.1016/j.indcrop.2021.113951[↩]
- Wu Z., Gao R., Li H., Wang Y., Luo Y., Zou J., Zhao B., Chen S. New Insight into the Joint Significance of Dietary Jujube Polysaccharides and 6-Gingerol in Antioxidant and Antitumor Activities. RSC Adv. 2021;11:33219–33234. doi: 10.1039/D1RA03640H[↩]
- Mechchate H., Costa de Oliveira R., Es-safi I., Vasconcelos Mourão E.M., Bouhrim M., Kyrylchuk A., Soares Pontes G., Bousta D., Grafov A. Antileukemic Activity and Molecular Docking Study of a Polyphenolic Extract from Coriander Seeds. Pharmaceuticals. 2021;14:770. doi: 10.3390/ph14080770[↩]
- Gao Y., Wang S., Dang S., Han S., Yun C., Wang W., Wang H. Optimized Ultrasound-Assisted Extraction of Total Polyphenols from Empetrum Nigrum and Its Bioactivities. J. Chromatogr. B. 2021;1173:122699. doi: 10.1016/j.jchromb.2021.122699[↩]
- Huang Y., Zhu X., Zhu Y., Wang Z. Pinus Koraiensis Polyphenols: Structural Identification, in Vitro Antioxidant Activity, Immune Function and Inhibition of Cancer Cell Proliferation. Food Funct. 2021;12:4176–4198. doi: 10.1039/D0FO03347B[↩]
- Fahmi A.A., El Raey M.A., Ibrahim A.Y., Abdelfattah M.A.O., Abdelmageed A.M., Sobeh M. A Sulfated Polyphenols-Rich Extract from Sabal Yapa Exhibits Antitumor Activities in Ehrlich Ascites Carcinoma. Saudi J. Biol. Sci. 2021;28:3117–3125. doi: 10.1016/j.sjbs.2021.02.056[↩]
- Prakash M.D., Stojanovska L., Feehan J., Nurgali K., Donald E.L., Plebanski M., Flavel M., Kitchen B., Apostolopoulos V. Anti-Cancer Effects of Polyphenol-Rich Sugarcane Extract. PLoS ONE. 2021;16:e0247492. doi: 10.1371/journal.pone.0247492[↩]
- Diab T.A., Donia T., Saad-Allah K.M. Characterization, Antioxidant, and Cytotoxic Effects of Some Egyptian Wild Plant Extracts. Beni-Suef Univ. J. Basic Appl. Sci. 2021;10:13. doi: 10.1186/s43088-021-00103-0[↩]
- Mesas C., Martínez R., Ortíz R., Galisteo M., López-Jurado M., Cabeza L., Perazzoli G., Melguizo C., Porres J.M., Prados J. Antitumor Effect of the Ethanolic Extract from Seeds of Euphorbia lathyris in Colorectal Cancer. Nutrients. 2021;13:566. doi: 10.3390/nu13020566[↩]
- Kato K., Nagane M., Aihara N., Kamiie J., Miyanabe M., Hiraki S., Luo X., Nakanishi I., Shoji Y., Matsumoto K., et al. Lipid-Soluble Polyphenols from Sweet Potato Exert Antitumor Activity and Enhance Chemosensitivity in Breast Cancer. J. Clin. Biochem. Nutr. 2021;68:193–200. doi: 10.3164/jcbn.20-73[↩]
- Yang S., Wang C., Li X., Wu C., Liu C., Xue Z., Kou X. Investigation on the Biological Activity of Anthocyanins and Polyphenols in Blueberry. J. Food Sci. 2021;86:614–627. doi: 10.1111/1750-3841.15598[↩]
- Delgado-Roche L., González K., Mesta F., Couder B., Tavarez Z., Zavala R., Hernandez I., Garrido G., Rodeiro I., Vanden Berghe W. Polyphenolic Fraction Obtained From Thalassia Testudinum Marine Plant and Thalassiolin B Exert Cytotoxic Effects in Colorectal Cancer Cells and Arrest Tumor Progression in a Xenograft Mouse Model. Front. Pharmacol. 2020;11:592985. doi: 10.3389/fphar.2020.592985[↩]
- Girardelo J.R., Munari E.L., Dallorsoleta J.C.S., Cechinel G., Goetten A.L.F., Sales L.R., Reginatto F.H., Chaves V.C., Smaniotto F.A., Somacal S., et al. Bioactive Compounds, Antioxidant Capacity and Antitumoral Activity of Ethanolic Extracts from Fruits and Seeds of Eugenia involucrata DC. Food Res. Int. 2020;137:109615. doi: 10.1016/j.foodres.2020.109615[↩]
- Fedotcheva T.A., Sheichenko O.P., Fedotcheva N.I. New Properties and Mitochondrial Targets of Polyphenol Agrimoniin as a Natural Anticancer and Preventive Agent. Pharmaceutics. 2021;13:2089. doi: 10.3390/pharmaceutics13122089[↩]
- Liu G., Shi A., Wang N., Li M., He X., Yin C., Tu Q., Shen X., Tao Y., Wang Q., et al. Polyphenolic Proanthocyanidin-B2 Suppresses Proliferation of Liver Cancer Cells and Hepatocellular Carcinogenesis through Directly Binding and Inhibiting AKT Activity. Redox Biol. 2020;37:101701. doi: 10.1016/j.redox.2020.101701[↩]
- Carpi S., Polini B., Manera C., Digiacomo M., Salsano J.E., Macchia M., Scoditti E., Nieri P. MiRNA Modulation and Antitumor Activity by the Extra-Virgin Olive Oil Polyphenol Oleacein in Human Melanoma Cells. Front. Pharmacol. 2020;11:574317. doi: 10.3389/fphar.2020.574317[↩]
- Gao L., Gou N., Yuan E., Ren J. Bioactivity-Oriented Purification of Polyphenols from Cinnamomum Cassia Presl. with Anti-Proliferation Effects on Colorectal Cancer Cells. Plant Foods Hum. Nutr. 2020;75:561–568. doi: 10.1007/s11130-020-00846-8[↩]
- Wu X., Xue L., Tata A., Song M., Neto C.C., Xiao H. Bioactive Components of Polyphenol-Rich and Non-Polyphenol-Rich Cranberry Fruit Extracts and Their Chemopreventive Effects on Colitis-Associated Colon Cancer. J. Agric. Food Chem. 2020;68:6845–6853. doi: 10.1021/acs.jafc.0c02604[↩]
- Xu P., Yan F., Zhao Y., Chen X., Sun S., Wang Y., Ying L. Green Tea Polyphenol EGCG Attenuates MDSCs-Mediated Immunosuppression through Canonical and Non-Canonical Pathways in a 4T1 Murine Breast Cancer Model. Nutrients. 2020;12:1042. doi: 10.3390/nu12041042[↩]
- Chen B.-H., Hsieh C.-H., Tsai S.-Y., Wang C.-Y., Wang C.-C. Anticancer Effects of Epigallocatechin-3-Gallate Nanoemulsion on Lung Cancer Cells through the Activation of AMP-Activated Protein Kinase Signaling Pathway. Sci. Rep. 2020;10:5163. doi: 10.1038/s41598-020-62136-2[↩]
- Yang R., Shan S., Zhang C., Shi J., Li H., Li Z. Inhibitory Effects of Bound Polyphenol from Foxtail Millet Bran on Colitis-Associated Carcinogenesis by the Restoration of Gut Microbiota in a Mice Model. J. Agric. Food Chem. 2020;68:3506–3517. doi: 10.1021/acs.jafc.0c00370[↩]
- Bona N.P., Pedra N.S., Azambuja J.H., Soares M.S.P., Spohr L., Gelsleichter N.E., de M. Meine B., Sekine F.G., Mendonça L.T., de Oliveira F.H., et al. Tannic Acid Elicits Selective Antitumoral Activity in Vitro and Inhibits Cancer Cell Growth in a Preclinical Model of Glioblastoma Multiforme. Metab. Brain Dis. 2020;35:283–293. doi: 10.1007/s11011-019-00519-9[↩]
- Chatterjee K., Mukherjee S., Vanmanen J., Banerjee P., Fata J.E. Dietary Polyphenols, Resveratrol and Pterostilbene Exhibit Antitumor Activity on an HPV E6-Positive Cervical Cancer Model: An in Vitro and in Vivo Analysis. Front. Oncol. 2019;9:352. doi: 10.3389/fonc.2019.00352[↩]
- Juli G., Oliverio M., Bellizzi D., Gallo Cantafio M.E., Grillone K., Passarino G., Colica C., Nardi M., Rossi M., Procopio A., et al. Anti-Tumor Activity and Epigenetic Impact of the Polyphenol Oleacein in Multiple Myeloma. Cancers. 2019;11:990. doi: 10.3390/cancers11070990[↩]
- Prieto K., Cao Y., Mohamed E., Trillo-Tinoco J., Sierra R.A., Urueña C., Sandoval T.A., Fiorentino S., Rodriguez P.C., Barreto A. Polyphenol-Rich Extract Induces Apoptosis with Immunogenic Markers in Melanoma Cells through the ER Stress-Associated Kinase PERK. Cell Death Discov. 2019;5:134. doi: 10.1038/s41420-019-0214-2[↩]
- de Jesús Romero-Prado M.M., Curiel-Beltrán J.A., Miramontes-Espino M.V., Cardona-Muñoz E.G., Rios-Arellano A., Balam-Salazar L. Dietary Flavonoids Added to Pharmacological Antihypertensive Therapy Are Effective in Improving Blood Pressure. Basic Clin. Pharmacol. Toxicol. 2015;117:57–64. doi: 10.1111/bcpt.12360[↩]
- Bogolitsyn K., Dobrodeeva L., Samodova A., Parshina A. In Vitro Immunostimulant Activity of the Polyphenolic Extract from the Arctic Brown Algae Fucus Vesiculosus. Plant Foods Hum. Nutr. 2024;79:511–517. doi: 10.1007/s11130-024-01174-x[↩]
- García-Lafuente A, Guillamón E, Villares A, Rostagno MA, Martínez JA. Flavonoids as antiinflammatory agents: implications in cancer and cardiovascular disease. Inflamm Res. 2009;58:537–552. https://www.ncbi.nlm.nih.gov/pubmed/19381780[↩][↩]
- Pirola L, Frojdo S. Resveratrol: One Molecule, Many Targets. IUBMB Life. 2008;60:323–332. https://www.ncbi.nlm.nih.gov/pubmed/18421779[↩][↩]
- Harikumar KB, Aggarwal BB. Resveratrol: a multitargeted agent for age-associated chronic diseases. Cell Cycle. 2008;7:1020–1035. https://www.ncbi.nlm.nih.gov/pubmed/18414053[↩][↩]
- Cucciolla V, Borriello A, Oliva A, Galletti P, Zappia V, Della Ragione F. Resveratrol: from basic science to the clinic. Cell Cycle. 2007;6:2495–2510. https://www.ncbi.nlm.nih.gov/pubmed/17726376[↩]
- Shakibaei M, Harikumar KB, Aggarwal BB. Resveratrol addiction: to die or not to die. Mol Nutr Food Res. 2009;53:115–128. https://www.ncbi.nlm.nih.gov/pubmed/19072742[↩]
- Peters U, Poole C, Arab L. Does tea affect cardiovascular disease? A meta-analysis. Am J Epidemiol. 2001;154:495–503. https://www.ncbi.nlm.nih.gov/pubmed/11549554[↩]
- Yang CS, Landau JM, Huang MT, Newmark HL. Inhibition of carcinogenesis by dietary polyphenolic compounds. Ann Rev Nutr. 2001;21:381–406. http://www.annualreviews.org/doi/full/10.1146/annurev.nutr.21.1.381[↩]
- Johnson IT, Williamson G, Musk SRR. Anticarcinogenic factors in plant foods: A new class of nutrients? Nutr Res Rev. 1994;7:175–204. https://www.cambridge.org/core/journals/nutrition-research-reviews/article/anticarcinogenic-factors-in-plant-foods-a-new-class-of-nutrients/227E1AF9647A3665125BF88846D94611[↩]
- Talalay P, De Long MJ, Prochaska HJ. Identification of a common chemical signal regulating the induction of enzymes that protect against chemical carcinogenesis. Proc Natl Acad Sci USA. 1988;85:8261–8265. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC282409/[↩]
- Khan N, Mukhtar H. Multitargeted therapy of cancer by green tea polyphenols. Cancer Lett. 2008;269:269–280. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2996277/[↩]
- Rizvi SI, Zaid MA. Intracellular reduced glutathione content in normal and type 2 diabetic erythrocytes: effect of Insulin and (-)epicatechin. J Physiol Pharmacol. 2001;52:483–488. https://www.ncbi.nlm.nih.gov/pubmed/11596865[↩]
- Rizvi SI, Zaid MA. Impairment of sodium pump and Na/H exchanger in erythrocytes from non-insulin dependent diabetes mellitus patients: effect of tea catechins. Clin Chim Acta. 2005;354:59–67. https://www.ncbi.nlm.nih.gov/pubmed/15748600[↩]
- Rizvi SI, Zaid MA, Anis R, Mishra N. Protective role of tea catechins against oxidation-induced damage of type 2 diabetic erythrocytes. Clin Exp Pharmacol Physiol. 2005;32:70–75. https://www.ncbi.nlm.nih.gov/pubmed/15730438[↩]
- Rizvi S I, Zaid M A. Insulin like effect of epicatechin on membrane acetylcholinesterase activity in type 2 diabetes mellitus. Clin Exp Pharmacol Physiol. 2001;28:776–778. https://www.ncbi.nlm.nih.gov/pubmed/11553037[↩]
- Matsui T, Ebuchi S, Kobayashi M, Fukui K, Sugita K, Terahara N, Matsumoto K. Anti-hyperglycemic effect of diacylated anthocyanin derived from Ipomoea batatas cultivar Ayamurasaki can be achieved through the alpha-glucosidase inhibitory action. J Agric Food Chem. 2002;50:7244–7248. https://www.ncbi.nlm.nih.gov/pubmed/12452639[↩]
- Lee WC, Wang CJ, Chen YH, Hsu JD, Cheng SY, Chen HC, et al. Polyphenol extracts from Hibiscus sabdariffa Linnaeus attenuate nephropathy in experimental type 1 diabetes. J Agric Food Chem. 2009;57:2206–2210. https://www.ncbi.nlm.nih.gov/pubmed/19219995[↩]
- Barone E, Calabrese V, Mancuso C. Ferulic acid and its therapeutic potential as a hormetin for age-related diseases. Biogerontology. 2009;10:97–108. https://www.ncbi.nlm.nih.gov/pubmed/18651237[↩]
- Jung EH, Kim SR, Hwang IK, Ha TY. Hypoglycemic effects of a phenolic acid fraction of rice bran and ferulic acid in C57BL/KsJ-db/db mice. J Agric Food Chem. 2007;55:9800–9804. https://www.ncbi.nlm.nih.gov/pubmed/17973443[↩]
- Harman D. Free radical theory of aging: an update. Ann N Y Acad Sci. 2006;1067:1–12. https://www.ncbi.nlm.nih.gov/pubmed/16803965[↩]
- Cao G, Booth S L, Sadowsky J A, Prior R L. Increases in human plasma antioxidant capacity after consumption of controlled diets high in fruit and vegetables. Am J Clin Nutr. 1998;68:1081–1087. https://www.ncbi.nlm.nih.gov/pubmed/9808226[↩]
- Joseph JA, Shukitt-Hale B, Casadesus G. Reversing the deleterious effects of aging on neuronal communication and behavior: beneficial properties of fruit polyphenolic compounds. Am J Clin Nutr. 2005;81:313–316. https://www.ncbi.nlm.nih.gov/pubmed/15640496[↩]
- Seeram NP, Cichewicz R H, Chandra A, Nair MG. Cyclooxygenase inhibitory and antioxidant compounds from crabapple fruits. J Agric Food Chem. 2003;51:1948–1951. https://www.ncbi.nlm.nih.gov/pubmed/12643656[↩]
- Shukitt-Hale B, Lau FC, Joseph JA. Berry Fruit Supplementation and the Aging Brain. J Agric Food Chem. 2008;56:636–641. https://www.ncbi.nlm.nih.gov/pubmed/18211020[↩]
- Maurya PK, Rizvi SI. Protective role of tea catechins on erythrocytes subjected to oxidative stress during human aging. Nat Prod Res. 2008:1–8. https://www.ncbi.nlm.nih.gov/pubmed/18846469[↩]
- Markus MA, Morris BJ. Resveratrol in prevention and treatment of common clinical conditions of aging. Clin Interv Aging. 2008;3:331–339. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2546476/[↩]
- Barger JL, Kayo T, Vann JM, Arias EB, Wang J, Hacker TA, et al. A low dose of dietary resveratrol partially mimics caloric restriction and retards aging parameters in mice. PLoS One. 2008;3:e2264. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2386967/[↩]
- Belinha I, Amorim MA, Rodrigues P, de Freitas V, Moradas-Ferreira P, Mateus N, et al. Quercetin increases oxidative stress resistance and longevity in Saccharomyces cerevisiae. J Agric Food Chem. 2007;55:2446–2451. https://www.ncbi.nlm.nih.gov/pubmed/17323973[↩]
- Letenneur L, Proust-Lima C, Le Gouge A, Dartigues J, Barberger-Gateau P. Flavonoid intake and cognitive decline over a 10-year period. Am J Epidemiol. 2007;165:1364–1371. https://www.ncbi.nlm.nih.gov/pubmed/17369607[↩]
- Scarmeas N, Luchsinger J A, Mayeux R, Stern Y. Mediterranean diet and Alzheimer disease mortality. Neurology. 2007;69:1084–1093. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2673956/[↩]
- Dai Q, Borenstein AR, Wu Y, Jackson JC, Larson EB. Fruit and vegetable juices and Alzheimer’s disease: the Kame Project. Am J Med 2. 2006;119:751–759. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2266591/[↩]
- Singh M, Arseneault M, Sanderson T, Murthy V, Ramassamy C. Challenges for research on polyphenols from foods in Alzheimer’s disease: Bioavailability, metabolism, and cellular and molecular mechanisms. J Agric Food Chem. 2008;56:4855–4873. https://www.ncbi.nlm.nih.gov/pubmed/18557624[↩]
- Aquilano K, Baldelli S, Rotilio G, Ciriolo MR. Role of nitric oxide synthases in Parkinson’s disease: a review on the antioxidant and anti-inflammatory activity of polyphenols. Neurochem Res. 2008;33:2416–2426. https://www.ncbi.nlm.nih.gov/pubmed/18415676[↩]
- Tsao R. Chemistry and Biochemistry of Dietary Polyphenols. Nutrients. 2010;2:1231–1246. doi: 10.3390/nu2121231[↩]
- Pandey K.B., Rizvi S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell Longev. 2009;2:270–278. doi: 10.4161/oxim.2.5.9498[↩]
- Makarewicz M., Drożdż I., Tarko T., Duda-Chodak A. The interactions between polyphenols and microorganisms, especially gut microbiota. Antioxidants. 2021;10:188. doi: 10.3390/antiox10020188[↩]
- Pérez-Jiménez J., Neveu V., Vos F., Scalbert A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010;64((Suppl. 3[↩]
- Bongiorno D., Di Stefano V., Indelicato S., Avellone G., Ceraulo L. Bio-phenols determination in olive oils: Recent mass spectrometry approaches. Mass Spectrom Rev. 2021:e21744. doi: 10.1002/mas.21744[↩]
- Indelicato S., Houmanat K., Bongiorno D., Ejjilani A., Hssaini L., Razouk R., Charafi J., Ennahli S., Hanine H. Freeze dried pomegranate juices of Moroccan fruits: Main representative phenolic compounds. J. Sci. Food Agric. 2023;103:1355–1365. doi: 10.1002/jsfa.12229[↩]
- Lyu X., Agar O.T., Barrow C.J., Dunshea F.R., Suleria H.A.R. Phenolic compounds profiling and their antioxidant capacity in the peel, pulp, and seed of Australian grown avocado. Antioxidants. 2023;12:185. doi: 10.3390/antiox12010185[↩]
- Bowtell J., Kelly V. Fruit-derived polyphenol supplementation for athlete recovery and performance. Sports Med. 2019;49((Suppl. S1[↩][↩]
- Ramírez-Garza S.L., Laveriano-Santos E.P., Marhuenda-Muñoz M., Storniolo C.E., Tresserra-Rimbau A., Vallverdú-Queralt A., Lamuela-Raventós R.M. Health Effects of Resveratrol: Results from Human Intervention Trials. Nutrients. 2018;10:1892. doi: 10.3390/nu10121892[↩]
- Askari G, Ghiasvand R, Feizi A, Ghanadian SM, Karimian J. The effect of quercetin supplementation on selected markers of inflammation and oxidative stress. J Res Med Sci. 2012 Jul;17(7):637-41. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3685779[↩]
- Petrisor G., Motelica L., Ficai D., Trusca R.D., Surdu V.-A., Voicu G., Oprea O.C., Ficai A., Andronescu E. New mesoporous silica materials loaded with polyphenols: Caffeic acid, ferulic acid and p-coumaric acid as dietary supplements for oral administration. Materials. 2022;15:7982. doi: 10.3390/ma15227982[↩]
- Myburgh K.H. Polyphenol supplementation: Benefits for exercise performance or oxidative stress? Sports Med. 2014;44 Suppl. S1:S57–S70. doi: 10.1007/s40279-014-0151-4[↩]
- Martin K., Appel C.L. Polyphenols as dietary supplements: A double-edged sword. Nutr. Diet. Suppl. 2010;2:1–12. doi: 10.2147/NDS.S6422[↩]
- Bunte K., Hensel A., Beikler T. Polyphenols in the prevention and treatment of periodontal disease: A systematic review of in vivo, ex vivo and in vitro studies. Fitoterapia. 2019;132:30–39. doi: 10.1016/j.fitote.2018.11.012[↩]
- Petti S., Scully C. Polyphenols, oral health and disease: A review. J. Dent. 2009;37:413–423. doi: 10.1016/j.jdent.2009.02.003[↩]
- Ding Y., Yao H., Yao Y., Fai L.Y., Zhang Z. Protection of dietary polyphenols against oral cancer. Nutrients. 2013;5:2173–2191. doi: 10.3390/nu5062173[↩]
- Álvarez-Martínez F.J., Barrajón-Catalán E., Encinar J.A., Rodríguez-Díaz J.C., Micol V. Antimicrobial capacity of plant polyphenols against gram-positive bacteria: A comprehensive review. Curr. Med. Chem. 2020;27:2576–2606. doi: 10.2174/0929867325666181008115650[↩]
- Bors W., Michel C. Chemistry of the antioxidant effect of polyphenols. Ann. N. Y. Acad. Sci. 2002;957:57–69. doi: 10.1111/j.1749-6632.2002.tb02905.x[↩]
- Manach C., Mazur A., Scalbert A. Polyphenols and prevention of cardiovascular diseases. Curr. Opin. Lipidol. 2005;16:77–84. doi: 10.1097/00041433-200502000-00013[↩]
- Zhou Y., Zheng J., Li Y., Xu D.P., Li S., Chen Y.M., Li H.B. Natural polyphenols for prevention and treatment of cancer. Nutrients. 2016;8:515. doi: 10.3390/nu8080515[↩]
- González R., Ballester I., López-Posadas R., Suárez M.D., Zarzuelo A., Martínez-Augustin O., Sánchez de Medina F. Effects of flavonoids and other polyphenols on inflammation. Crit. Rev. Food Sci. Nutr. 2011;51:331–362. doi: 10.1080/10408390903584094[↩]
- Cheng Y.C., Sheen J.M., Hu W.L., Hung Y.C. Polyphenols and oxidative stress in atherosclerosis-related ischemic heart disease and stroke. Oxidative Med. Cell. Longev. 2017;2017:8526438. doi: 10.1155/2017/8526438[↩]