Contents
Acute pancreatitis
Acute pancreatitis is inflammation of the pancreas. Acute pancreatitis happens when digestive enzymes start digesting the pancreas itself. Acute pancreatitis occurs suddenly and is a short-term condition, meaning it appears suddenly and lasts for days 1, 2. Most people with acute pancreatitis get better, and it goes away in several days with treatment. But some people can have a more severe form of acute pancreatitis and go on to develop serious complications, which requires a lengthy hospital stay. Acute pancreatitis is different to chronic pancreatitis, where the pancreas has become permanently damaged from inflammation over many years. Acute pancreatitis affects men more often than women. Acute pancreatitis is becoming more common, for reasons that are not clear 3. Acute pancreatitis is the cause of up to 275,000 hospitalizations in the United States per year, incurring nearly $2.6 billion in hospitalization costs 4, 5, 6. The incidence of acute pancreatitis in children is at 10–15 cases per 100,000 children 7. While mild acute pancreatitis carries a mortality of <1%, mortality rates for severe pancreatitis can reach as high as 30% 8. Drugs are responsible for 0.1%-2% of acute pancreatitis incidents. The majority of drug-induced pancreatitis cases are mild to moderate in severity; however, severe and even fatal cases can occur. Although pancreatitis is rare in children, the number of children with acute pancreatitis has grown.
Repeat episodes of acute pancreatitis may lead to chronic pancreatitis. Other complications of acute pancreatitis include:
- dehydration
- bleeding
- infection
Most cases of pancreatitis are caused by overuse of alcohol 9, 10. Alcohol use is responsible for up to 70% of cases of acute pancreatitis in the United States. About 5 to 8 drinks per day for 5 or more years can damage the pancreas 11. Gallstones are the next most common cause of acute pancreatitis 12, 13. When the gallstones travel out of the gallbladder into the bile ducts, they block the opening that drains bile and enzymes. The bile and enzymes “back up” into the pancreas and cause swelling. Besides overuse of alcohol and gallstones, other causes of pancreatitis include:
- Heredity — Hereditary pancreatitis is a rare genetic disorder that predisposes a person to develop the disease, usually before age 20 14.
- Genetic causes — Mutations of the cystic fibrosis gene is the most widely recognized genetic cause.
- Blockage of the duct that drains digestive enzymes from the pancreas — If the enzymes don’t drain properly, they can back up and damage the pancreas. Blockage can be caused by gallstones, scarring from prior surgery, tumors, pancreatic cancer or abnormalities of the pancreas or of the shape or location of the pancreatic duct. If the blockage is found early, surgery or a procedure called endoscopic retrograde cholangiopancreatography (ERCP) to relieve the blockage may help to prevent damage to the pancreas. Endoscopic retrograde cholangiopancreatography (ERCP), a procedure used to treat gallstones, also can lead to pancreatitis.
- Autoimmune pancreatitis — For unexplained reasons, some people develop antibodies that attack their own pancreas.
- Very high blood triglyceride levels (hypertriglyceridemia) – most often above 1,000 mg/dL 15, 16, 17, 18.
- Damage to the ducts or pancreas during surgery
- After certain procedures used to diagnose gallbladder and pancreas problems (endoscopic retrograde cholangiopancreatography, ERCP) or ultrasound guided biopsy 19, 20
- Injury to the pancreas from an accident
- Overactive parathyroid gland (hyperparathyroidism)
- Reye syndrome
- Use of certain medicines (especially estrogens, corticosteroids, sulfonamides, thiazides, and azathioprine) 21, 22
- Certain infections, such as viral infections (e.g. mumps, cytomegalovirus, coxsackie B virus) that involve the pancreas
- Smoking. Although heavy smokers tended to be heavy drinkers, smoking itself was a significant risk factor for acute pancreatitis, recurrent acute pancreatitis and chronic pancreatitis 23, 24
Sometimes, a cause for pancreatitis is never found. This is known as idiopathic pancreatitis.
The most common symptoms of acute pancreatitis include 25:
- suddenly getting severe pain in the center of your abdomen (belly) that radiates to your back
- feeling or being sick (nausea and vomiting)
- a high temperature of 38°C (100.4°F) or more (fever)
Acute pancreatitis is usually diagnosed in hospital, where you’ll receive treatment and be monitored for any complications. A doctor will ask you about your symptoms, family history and may feel your abdomen – it will be very tender if you have acute pancreatitis. They’ll also do a blood test, and sometimes a CT scan, to help confirm the diagnosis.
At first, it can be difficult to tell whether your acute pancreatitis is mild or severe. You’ll be monitored closely for signs of serious problems, such as organ failure.
You may have further tests to help determine the severity of your condition and assess your risk of developing more serious complications. These may include:
- a CT scan of your abdomen – where a series of X-rays are taken to build up a more detailed image of your pancreas
- a MRI scan of your abdomen – where strong magnetic fields and radio waves are used to produce a detailed image of the inside of your body
- an ultrasound scan of your abdomen – where sound waves are used to create a picture of your gallbladder to detect gallstones, and a picture of your pancreas
A diagnosis of acute pancreatitis requires 2 out of 3 criteria 26, 27:
- Abdominal pain consistent with pancreatitis,
- A serum amylase or lipase three or more times the upper limit of normal, and
- Findings consistent with pancreatitis on cross-sectional abdominal imaging [in adults: computed tomography (CT) or magnetic resonance imaging (MRI); in children CT, MRI or in some cases transabdominal ultrasound (TUS)].
Acute pancreatitis is treated in hospital, where you’ll be closely monitored for signs of serious problems and given supportive treatment, such as fluids, painkillers (analgesics), oxygen and nutrition 28. Treatment for acute pancreatitis aims to help control the condition and manage any symptoms you may have. You may be given fluids directly into a vein (intravenous fluids), pain relief, liquid food through a tube in your abdomen and oxygen through tubes in your nose. A nil-by-mouth regimen to rest the gut, routine use of prophylactic antibiotics, and avoidance of early opiate analgesia have been invalidated in randomised trials, and do not feature in international guidelines 29, 30, 27.
Most people with mild acute pancreatitis get better within a week and are well enough to leave hospital after a few days.
Recovery can take longer in severe cases, as some people can develop complications.
Those with severe acute pancreatitis can develop complications that require further treatment and may need to be admitted to a high-dependency unit or intensive care unit (ICU). Recovery may take much longer from severe acute pancreatitis, and there’s a risk it could be fatal 31.
See your doctor right away for the following symptoms of severe pancreatitis:
- pain or tenderness in the abdomen that is severe or becomes worse
- nausea and vomiting
- fever or chills
- fast heartbeat
- shortness of breath
- yellowish color of the skin or whites of the eyes, called jaundice
These symptoms may be a sign of:
- serious infection
- inflammation
- blockage of the pancreas, gallbladder, or a bile and pancreatic duct
Left untreated, these problems can be fatal.
Pancreas anatomy
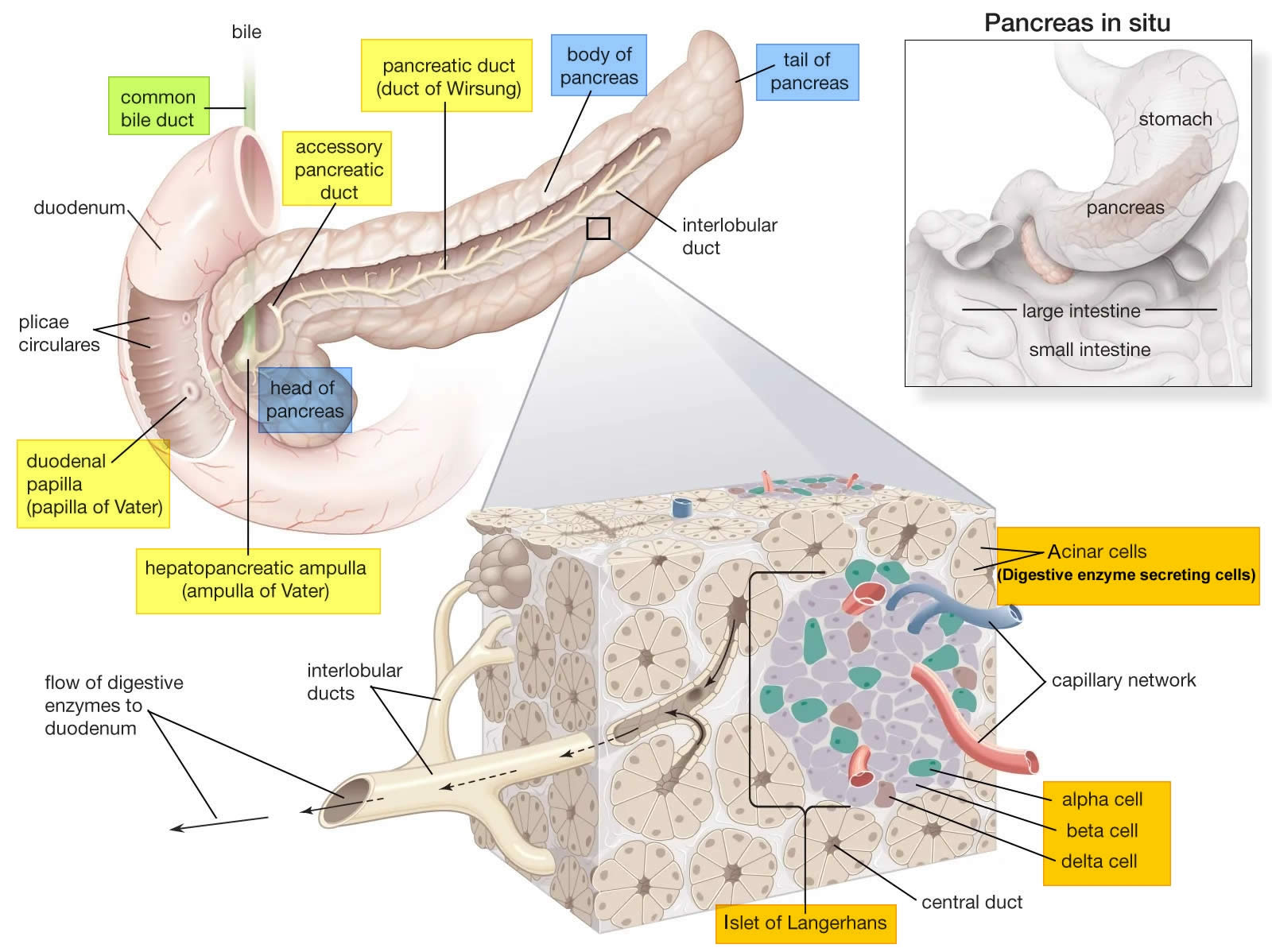
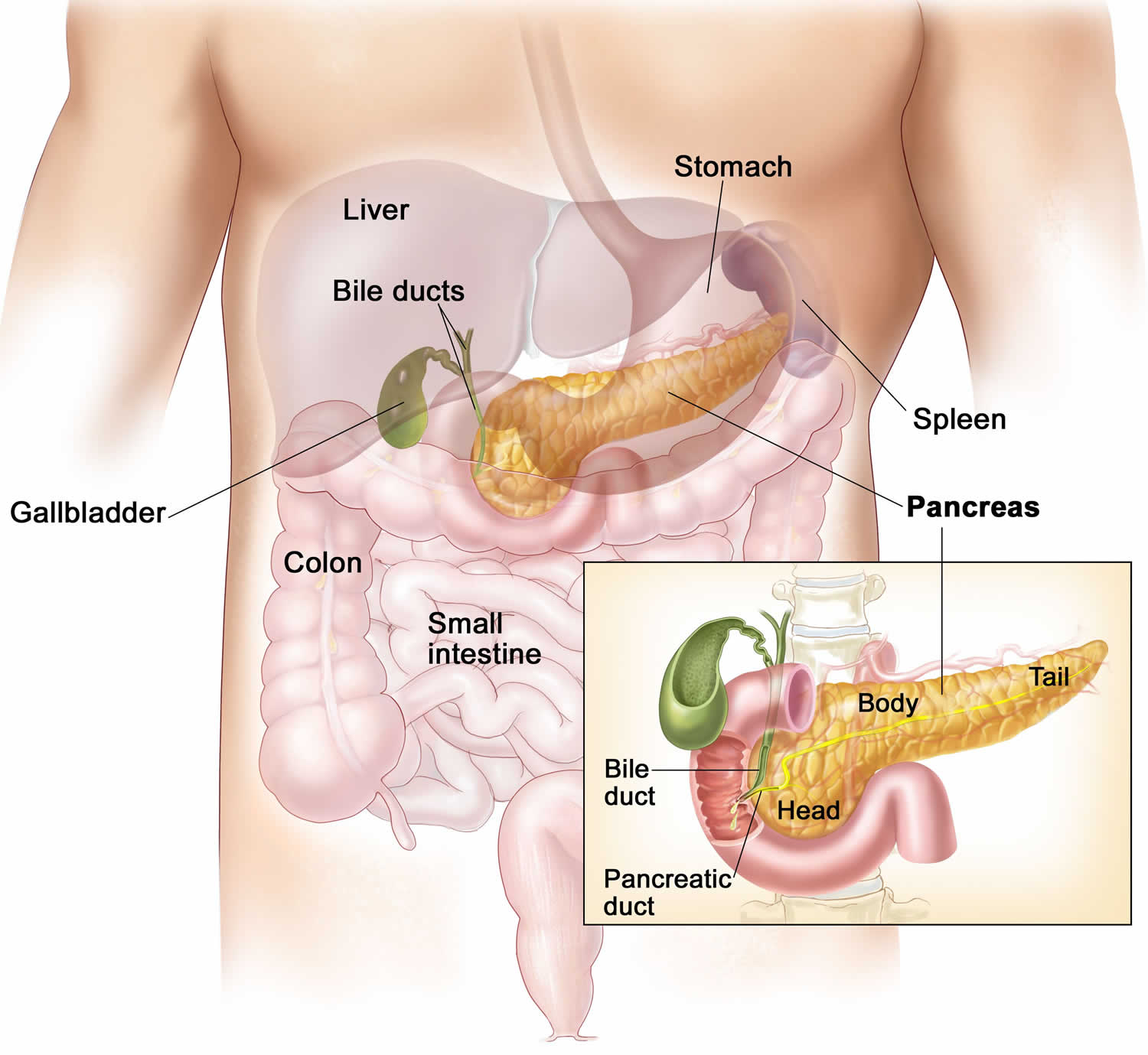
The pancreas is a large gland that sits behind the greater curvature of the stomach and close to the first part of the small intestine (the duodenum). The pancreas is shaped a bit like a fish with a wide head, a tapering body, and a narrow, pointed tail. In adults it’s about 12–15 cm (5–6 inches) long and 2.5 cm (1 in.) thick but less than 2 inches (5 centimeters) wide. The pancreas is both an endocrine and exocrine gland (see Figures 1 and 2).
The pancreas has 3 parts, the head, body, and tail.
- the wide end is called the head. The head of the pancreas is on the right side of the abdomen (belly), behind where the stomach meets the duodenum (the first part of the small intestine).
- the bit in the middle is called the body. The body of the pancreas is behind the stomach.
- the thin end is called the tail. The tail of the pancreas is on the left side of the abdomen next to the spleen.
About 99% of the pancreas is exocrine tissue made up of small clusters of glandular epithelial cells called acinar cells (acini), which secretes 1,200 to 1,500 mL of pancreatic juice per day – that are released into the small intestines to help you digest foods (especially fats). The digestive enzymes are first released into tiny tubes called central ducts. These merge to form larger ducts, which empty into the pancreatic duct (duct of Wirsung). The pancreatic duct merges with the common bile duct (the duct that carries bile from the liver), and empties into the duodenum (the first part of the small intestine) at the ampulla of Vater (also known as the hepatopancreatic ampulla). The ampulla of Vater (hepatopancreatic ampulla) is where the pancreatic duct and bile duct join together to drain into the duodenum, which is the first part of the small intestine. The passage of pancreatic juice and bile through the hepatopancreatic ampulla (ampulla of Vater) into the duodenum of the small intestine is regulated by a mass of smooth muscle surrounding the ampulla known as the sphincter of the hepatopancreatic ampulla, or sphincter of Oddi. The other major duct of the pancreas, the accessory duct (duct of Santorini), that branches from the main pancreatic duct and opens independently into the duodenum about 2.5 cm (1 in.) superior to the hepatopancreatic ampulla (ampulla of Vater) at the minor duodenal papilla. The accessory duct (duct of Santorini) bypasses the sphincter and allows pancreatic juice to be released into the duodenum even when bile is held back.
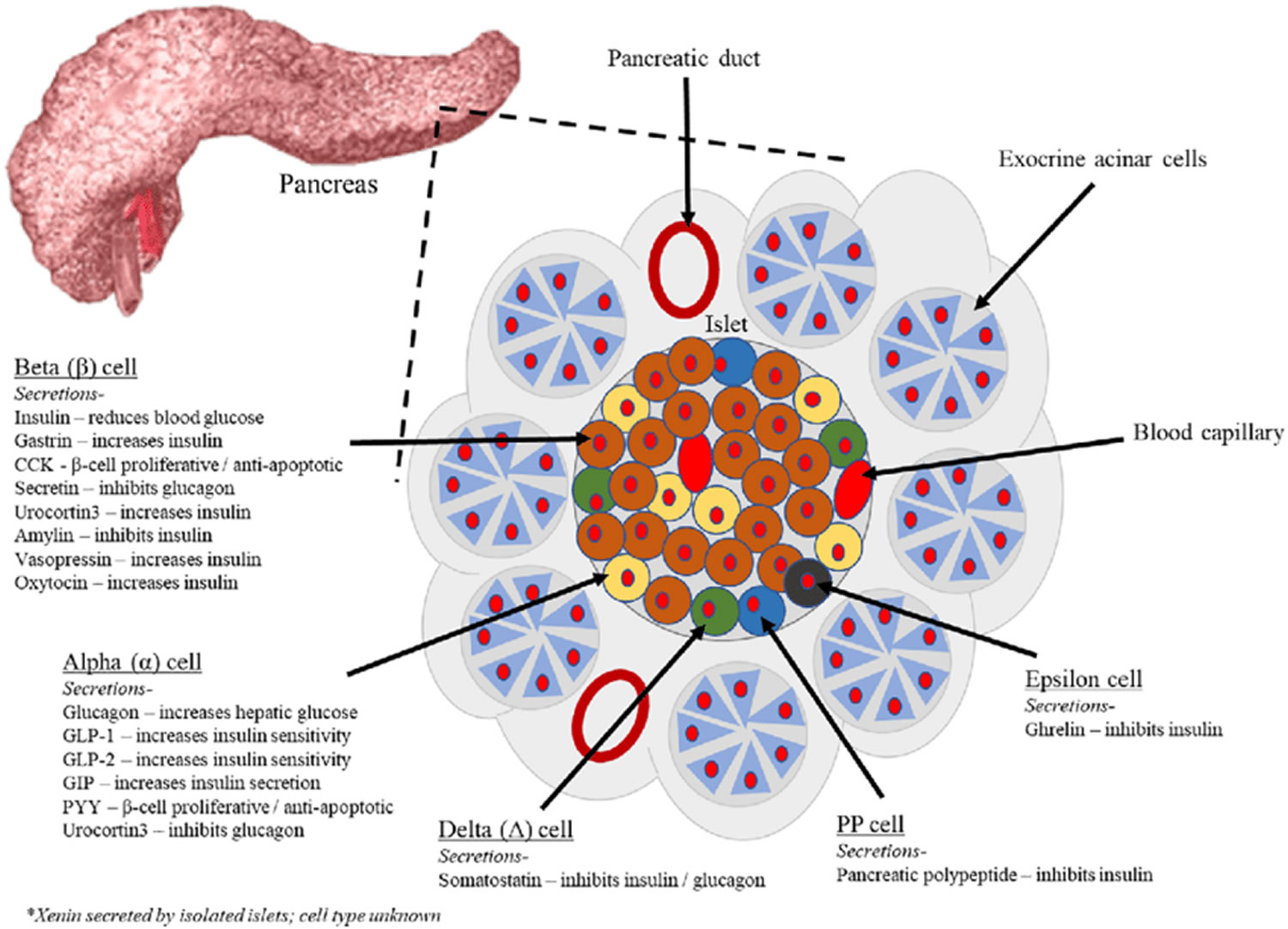
The endocrine part of the pancreas consists of groups of cells that are closely associated with blood vessels. These remaining 1% of the cell clusters form “islands” of cells called pancreatic islets (Islets of Langerhans). The Islets of Langerhans cells secrete the hormones glucagon, insulin, somatostatin, and pancreatic polypeptide (PP). Islets of Langerhans main cell types are alpha cells (20%), beta cells (70%), and delta cells (5%). The pancreatic islets alpha cells secrete the hormone glucagon, and beta cells secrete the hormone insulin (Figure 2). Both insulin and glucagon are important hormones which help control blood sugar levels and are released directly into the bloodstream.
The Islets of Langerhans Delta (δ) cells, or D cells, secrete somatostatin (growth hormone–inhibiting hormone) concurrently with the release of insulin by the beta cells. Somatostatin is a peptide hormone that inhibits the secretion of glucagon and insulin by the nearby alpha and beta cells. Somatostatin also work with amylin to limit the secretion of stomach acid.
Other, minor types of pancreatic cells, about 5% of the total, are called pancreatic polypeptide (PP) and G cells. Pancreatic polypeptide (PP) cells secrete pancreatic polypeptide, a hormone that may inhibit the exocrine activity of the pancreas.
Pancreatic islets (Islets of Langerhans) are relatively concentrated in the tail of the pancreas, whereas the head is more exocrine. Over 90% of pancreatic cancers arise from the ducts of the exocrine portion (ductal carcinomas), so cancer is most common in the head of the pancreas.
Figure 1. The pancreas
Figure 2. Pancreas cell types
Footnotes: Exocrine pancreatic acinar cells constitute most of the pancreatic tissue, these cells produce digestive enzymes which are transported via the pancreatic ducts. The endocrine pancreas is illustrated with all cell types; alpha, beta, delta, pancreatic polypeptide (PP) and epsilon. The endocrine pancreas cells are arranged in compact Islets of Langerhans and secrete a number of classical and ‘nonclassical’ peptides, as depicted.
Figure 3. Pancreas location
Figure 4. Relationship of the pancreas to the liver, gallbladder, and duodenum
Pancreas function
The pancreas has two main functions—to make insulin and to make digestive juices, or enzymes, to help you digest food in the intestine. About 99% of the pancreas is exocrine tissue made up of small clusters of glandular epithelial cells called acinar cells (acini), which secretes 1,200 to 1,500 mL of pancreatic juice per day – that are released into the small intestines to help you digest foods (especially fats). The cells of the secretory acini exhibit a high density of rough ER (endoplasmic reticulum) and secretory vesicles (zymogen granules). The acini open into a system of branched ducts that eventually converge on the main pancreatic duct. This duct runs lengthwise through the middle of the gland and joins the bile duct at the hepatopancreatic ampulla (ampulla of Vater). The hepatopancreatic sphincter (sphincter of Oddi) thus controls the release of both bile and pancreatic juice into the duodenum. Usually, however, there is a smaller accessory pancreatic duct (duct of Santorini) that branches from the main pancreatic duct and opens independently into the duodenum at the minor duodenal papilla, proximal to the major papilla. The accessory duct (duct of Santorini) bypasses the hepatopancreatic sphincter (sphincter of Oddi) and allows pancreatic juice to be released into the duodenum even when bile is held back.
Pancreatic juice is an alkaline mixture of water, enzymes, zymogens, sodium bicarbonate, and other electrolytes. The acini secrete the enzymes and zymogens, whereas the ducts secrete the sodium bicarbonate. The bicarbonate buffers HCl (hydrochloric acid) arriving from the stomach.
Sodium bicarbonate buffers the hydrochloric acid arriving from the stomach, with the reaction:
- HCl + NaHCO3 ⟶ NaCl + H2CO3 (carbonic acid).
The carbonic acid then breaks down to carbon dioxide (CO2) and water. CO2 is absorbed into the blood and ultimately exhaled. What is left in the small intestine, therefore, is salt water—sodium chloride (NaCl) and H2O. Sodium bicarbonate is therefore important in protecting the intestinal mucosa from hydrochloric acid (HCl) as well as raising the intestinal pH to the level needed for activity of the pancreatic and intestinal digestive enzymes.
The pancreatic zymogens are trypsinogen, chymotrypsinogen and procarboxypeptidase. When trypsinogen is secreted into the intestinal lumen, it is converted to trypsin by enteropeptidase, an enzyme on the brush border of the duodenum. Trypsin is autocatalytic—it converts trypsinogen into still more trypsin. Trypsin also converts the other two zymogens into chymotrypsin and carboxypeptidase, in addition to its primary role of digesting dietary protein.
Pancreatic acinar cells also secrete a protein called trypsin inhibitor that combines with any trypsin formed accidentally in the pancreas or in pancreatic juice and blocks its enzymatic activity.
Other pancreatic enzymes include pancreatic amylase, which digests starch; pancreatic lipase, which digests fat; and ribonuclease and deoxyribonuclease, which digest RNA and DNA, respectively. Unlike the zymogens, these enzymes are not altered after secretion. They become fully active, however, only upon exposure to bile or ions in the intestinal lumen.
Regulation of Pancreatic Secretion
Three stimuli are chiefly responsible for the release of pancreatic juice and bile.
- Acetylcholine (ACh), coming from the vagus nerves and enteric neurons. ACh stimulates the pancreatic acini to secrete their enzymes even during the cephalic phase of gastric control, before food is swallowed. The enzymes remain stored in the pancreatic acini and ducts, however, in preparation for release later when chyme enters the duodenum.
- Cholecystokinin (CCK), secreted by the mucosa of the duodenum and proximal jejunum (the next segment of the small intestine), primarily in response to fats in the small intestine. CCK also stimulates the pancreatic acini to secrete enzymes, but it is named for its strongly stimulatory effect on the gallbladder. It induces contractions of the gallbladder and relaxation of the hepatopancreatic sphincter, discharging bile into the duodenum.
- Secretin, produced by the same regions of the small intestine, mainly in response to the acidity of chyme from the stomach. Secretin stimulates the ducts of both the liver and pancreas to secrete an abundant sodium bicarbonate solution. In the pancreas, this flushes the enzymes into the duodenum.
Hormones of the Pancreatic Islets
The pancreas is primarily an exocrine digestive gland. Scattered throughout the exocrine tissue, are 1 to 2 million endocrine groups of cells that are closely associated with blood vessels called pancreatic islets (islets of Langerhans). Although they are less than 2% of the pancreatic tissue, the islets of Langerhans secrete the hormone glucagon and the hormone insulin of vital importance, especially in the regulation of glycemia, the blood glucose concentration. The pancreatic islets of Langerhans include two distinct types of cells—alpha cells, which secrete the hormone glucagon, and beta cells, which secrete insulin hormone. A typical islet measures about 75 × 175 μm and contains from a few to 3,000 cells. Islets of Langerhans main cell types are alpha cells (20%), beta cells (70%), and delta cells (5%). Islets of Langerhans respond directly to blood nutrient levels associated with the cycle of eating and fasting. Their functions are as follows:
- Alpha (α) cells, or A cells, secrete glucagon between meals when the blood glucose concentration falls below 100 mg/dL (5.6 mmol/L). Glucagon exerts two primary actions on the liver: (1) glycogenolysis, the breakdown of glycogen into glucose; and (2) gluconeogenesis, the synthesis of glucose from fats and proteins. These effects lead to the release of glucose into circulation, thus raising the blood glucose level. In adipose tissue, glucagon stimulates fat catabolism and the release of free fatty acids. Glucagon is also secreted in response to rising amino acid levels in the blood after a high-protein meal. It promotes amino acid absorption and thereby provides cells with the raw material for gluconeogenesis.
- Beta (β) cells, or B cells, secrete two hormones, insulin and amylin. Insulin, “the hormone of nutrient abundance,” is secreted during and immediately following a meal when blood nutrient levels are rising. Osteocalcin, a hormone from the osteoblasts of bone, also stimulates multiplication of beta cells, insulin secretion, and insulin sensitivity of other body tissues. The principal targets of insulin are the liver, skeletal muscles, and adipose tissue. In times of plenty, insulin stimulates cells to absorb glucose, fatty acids, and amino acids and to store or metabolize them; therefore, it lowers the level of blood glucose and other nutrients. It promotes the synthesis of glycogen, fat, and protein, thereby promoting the storage of excess nutrients for later use and enhancing cellular growth and differentiation. It also antagonizes glucagon, thus suppressing the use of already-stored fuels. The brain, liver, kidneys, and red blood cells absorb and use glucose without need of insulin, but insulin does promote glycogen synthesis in the liver. Insulin insufficiency or inaction is well known as the cause of diabetes. The beta cells also secrete another hormone, amylin, simultaneously with insulin. Amylin helps to reduce spikes in blood glucose by slowing the emptying of the stomach; modulating the secretion of gastric enzymes, acid, and bile; inhibiting glucagon secretion; and stimulating the sense of satiety (having had enough to eat).
- Delta (δ) cells, or D cells, secrete somatostatin (growth hormone–inhibiting hormone) concurrently with the release of insulin by the beta cells. Somatostatin is a peptide hormone that inhibits the secretion of glucagon and insulin by the nearby alpha and beta cells. Somatostatin also work with amylin to limit the secretion of stomach acid.
- Other, minor types of pancreatic cells, about 5% of the total, are called pancreatic polypeptide (PP) and G cells. Pancreatic polypeptide (PP) cells secrete pancreatic polypeptide, a hormone that may inhibit the exocrine activity of the pancreas.
Any hormone that raises blood glucose concentration is called a hyperglycemic hormone. You may have noticed that glucagon is not the only hormone that does so; so do growth hormone, epinephrine, norepinephrine, cortisol, and corticosterone. Insulin is called a hypoglycemic hormone because it lowers blood glucose levels.
Glucagon raises the blood sugar concentration by stimulating the liver to break down glycogen and convert certain noncarbohydrates, such as amino acids, into glucose. These actions raise the blood glucose concentration. Glucagon much more effectively elevates blood glucose than does epinephrine (adrenaline).
A negative feedback system regulates glucagon secretion. A low blood glucose concentration stimulates alpha cells to release glucagon. When the blood glucose concentration rises, glucagon secretion falls. This control prevents hypoglycemia when the blood glucose concentration is relatively low, such as between meals, or when glucose is used rapidly, such as during exercise.
The main effect of insulin is to lower the blood glucose level, exactly opposite that of glucagon. Insulin does this in part by promoting facilitated diffusion of glucose into cells that have insulin receptors, for use in cellular respiration. Such cells include those of adipose tissue, liver, and skeletal muscle. (Glucose uptake by active skeletal muscle does not require insulin.) Insulin also stimulates the liver to form glycogen from glucose and inhibits conversion of noncarbohydrates into glucose. In addition, insulin promotes transport of amino acids into cells, increases the rate of protein synthesis, and stimulates adipose cells to synthesize and store fat.
A negative feedback system sensitive to the blood glucose concentration regulates insulin secretion. When the blood glucose concentration is high, such as after a meal, beta cells release insulin. Insulin helps prevent too high a blood glucose concentration by promoting glycogen formation in the liver and entrance of glucose into adipose and muscle cells.
When glucose concentration falls, such as between meals or during the night, insulin secretion decreases. As insulin secretion decreases, less glucose enters adipose and resting muscle cells. Cells that lack insulin receptors and are therefore not dependent on insulin, such as nerve cells, can still take up glucose from the blood. At the same time that insulin is decreasing, glucagon secretion is increasing. Nerve cells, including those of the brain, obtain glucose by a facilitated diffusion mechanism that does not require insulin, but rather depends only on the blood glucose concentration. For this reason, nerve cells are particularly sensitive to changes in blood glucose concentration. Conditions that cause such changes—for example, oversecretion of insulin leading to decreased blood glucose—are likely to affect brain functions.
Insulin and glucagon are coordinated to maintain a relatively stable blood glucose concentration, despite great variation in the amount of carbohydrates a person eats. About 85% to 90% of people with diabetes mellitus have type 2 diabetes, in which the beta cells produce insulin but body cells lose the ability to recognize it. On the other hand, type 1 diabetes mellitus usually appears before age twenty and it is an autoimmune disease: the immune system destroys the beta cells of the pancreas.
Acute pancreatitis causes
The most common cause of acute pancreatitis is having gallstones. Gallstones cause inflammation of your pancreas as stones pass through and get stuck in a bile or pancreatic duct. This condition is called gallstone pancreatitis. However, in approximately 30 percent of cases, a cause cannot be identified. Sometimes injury to the abdomen — such as a bicycle or playground accident or sports injury — can cause acute pancreatitis, or common medications and conditions, including 32:
- Anti-seizure medications
- Certain antibiotics
- Specific types of chemotherapy
- Heredity — Hereditary pancreatitis is a rare genetic disorder that predisposes a person to develop the disease, usually before age 20 14.
- Genetic causes — Mutations of the cystic fibrosis gene is the most widely recognized genetic cause.
- Blockage of the duct that drains digestive enzymes from the pancreas — If the enzymes don’t drain properly, they can back up and damage the pancreas. Blockage can be caused by gallstones, scarring from prior surgery, tumors, pancreatic cancer or abnormalities of the pancreas or of the shape or location of the pancreatic duct. If the blockage is found early, surgery or a procedure called endoscopic retrograde cholangiopancreatography (ERCP) to relieve the blockage may help to prevent damage to the pancreas. Endoscopic retrograde cholangiopancreatography (ERCP), a procedure used to treat gallstones, also can lead to pancreatitis.
- Autoimmune pancreatitis — For unexplained reasons, some people develop antibodies that attack their own pancreas. Autoimmune pancreatitis is a rare, distinct form of chronic pancreatitis that may present acutely 1. Autoimmune pancreatitis was initially described as chronic pancreatitis with hypergammaglobulinaemia 33, prior to introduction of the term ‘autoimmune pancreatitis’ 34. It was then variously described as chronic pancreatitis with autoimmune features, non-alcoholic duct-destructive chronic pancreatitis, lymphoplasmocytic sclerosing pancreatitis with cholangitis, chronic sclerosing pancreatitis, pseudotumorous pancreatitis and duct-narrowing chronic pancreatitis. The current definition of two types is based on histopathology: type 1 for lymphoplasmacytic sclerosing pancreatitis and type 2 for idiopathic duct centric chronic pancreatitis or autoimmune pancreatitis with granulocytic epithelial lesions. These lesions have also been reported in children with autoimmune sclerosing cholangitis but no pancreatic disease involvement 35, 36. Type 1 autoimmune pancreatitis is a feature of IgG4-related systemic disease 37; others include sclerosing cholangitis, sclerosing sialadenitis, retroperitoneal fibrosis, interstitial nephritis, chronic thyroiditis, interstitial pneumonia and lymphadenopathy. IgG4-related systemic disease is typified by tumor-like mass formation in affected organs that may feature high serum IgG4 concentrations or increased numbers of IgG4 plasma cells in tissues. Type 2 autoimmune pancreatitis is more often associated with inflammatory bowel disease, which confers a less favorable prognosis 38.
- Very high blood triglyceride levels (hypertriglyceridemia) – most often above 1,000 mg/dL 15, 16, 17, 18. Nevertheless, as there is an increased but lower risk of acute pancreatitis at lower levels, also proportional to the increase in triglycerides, 500 mg/dL appears preferable for this definition 39, 40. It is most important that levels are taken on admission to identify hypertriglyceridaemia 27, either as sole cause or a co-factor conferring a worse prognosis, since hypertriglyceridaemia-associated acute pancreatitis is severe more frequently than from other causes 39.
- Damage to the ducts or pancreas during surgery
- After certain procedures used to diagnose gallbladder and pancreas problems (endoscopic retrograde cholangiopancreatography, ERCP) or ultrasound guided biopsy 19, 20
- Injury to the pancreas from an accident
- Overactive parathyroid gland (hyperparathyroidism)
- Reye syndrome
- Use of certain medicines (especially estrogens, corticosteroids, sulfonamides, thiazides, and azathioprine) 21, 22
- Certain infections, such as viral infections (e.g. mumps, cytomegalovirus, coxsackie B virus) that involve the pancreas
- Smoking. Although heavy smokers tended to be heavy drinkers, smoking itself was a significant risk factor for acute pancreatitis, recurrent acute pancreatitis and chronic pancreatitis 23, 24
- Anatomical variants (pancreas divisum, pancreatobiliary ductal malunion)
- Cardiac bypass surgery
- Scorpion bites (notably from Tityus trinitatis)
- Organophosphate poisoning
- Problems when your immune system attacking your body
Other chronic conditions may cause pancreatitis such as inflammatory bowel disease, cystic fibrosis or celiac disease.
Table 1. Drugs associated with acute pancreatitis
| Angiotensin-converting enzyme (ACE) inhibitors | Estrogens | Pentamidine |
| Acetaminophen | Ethacrynic acids | Pergolide |
| Adrenocorticotrophic hormones | Exenatide | Phenolphthalein |
| Ezetimibe | Pilocarpine | |
| Alendronate | Fibrates | Prazosin |
| All-trans-retinoic acid | Finasteride | Procainamide |
| Alpha-methyldopa | Fluoroquinolones | Propofol |
| Aminosalicylates | 5-Fluorouracil | Propoxyphene |
| Amiodarone | Furosemide | Proton pump inhibitors |
| Amlodipine | Gabapentin | Quinupristin/dalfopristin |
| Ampicillin | Gold | Ranitidine |
| Antivirals | HAART agents | Repaglinide |
| Aspirin | HMG-CoA reductase inhibitors | Rifampin |
| Atypical antipsychotics | Rifapentine | |
| Azathioprine | Ifosfamide | Rivastigmine |
| Bupropion | Indomethacin | Ropinirole |
| Calcitriol | Interferon/ribavirin | Saw palmetto |
| Cannabis | Interleukin-2 | Selective serotonin receptor antagonists |
| Capecitabine | Irbesartan | |
| Carbamazepine | Isoniazid | Sirolimus |
| Ceftriaxone | Isotretinoin | Sodium stibogluconate |
| Cimetidine | Lamotrigine | Somatropin |
| Cisplatin | L-asparaginase | Sulfamethoxazole |
| Clomiphene | Macrolides | Sulfasalazine |
| Codeine | Mefenamic acid | Sumatriptan |
| Colchicine | 6-Mercaptopurine | Tacrolimus |
| Corticosteroids | Mesalamine | Tamoxifen |
| COX-2 inhibitors | Metformin | Tetracyclines |
| Cyclophosphamide | Methimazole | Thiazide diuretics |
| Cyclosporine | Methyldopa | Thrombolytic agents |
| Cyproheptadine | Metronidazole | TNF-alpha inhibitors |
| Cytosine | Mirtazapine | Topiramate |
| Danazol | Montelukast | Trimethoprim-sulfamethizole |
| Dapsone | Mycophenolate | |
| Diazoxide | Nitrofurantoin | Valproic acid |
| Diphenoxylate | NSAIDs | Venlafaxine |
| Dipyridamole | Octreotide | Vincristine |
| Doxercalciferol | Paclitaxel | Voriconazole |
| Doxorubicin | Pegaspargase | Zolmitriptan |
| Ertapenem | Penicillin |
Acute pancreatitis pathophysiology
The pancreas normally synthesizes and secretes several pancreatic enzymes involved in digestion. These enzymes are produced in pancreatic acinar cells as inactive zymogens and secreted into the duodenum via the pancreatic duct and sphincter of Oddi where they are activated. The activation process begins as enterokinase, an enzyme produced in the duodenal crypts of Lieberkühn, encounters the pancreatic zymogen trypsinogen. Enterokinase binds to trypsinogen and cleaves an acidic propeptide, leaving active trypsin to initiate a cascade of proteolytic reactions. The reactions lead to the activation of other pancreatic zymogens such as chymotrypsinogen, proelastase, prophospholipase, and procarboxypeptidases that are necessary for digestion 42, 43.
If, however, the zymogens are activated prematurely before they exit the pancreas, autodigestion of the peripancreatic tissue and pancreatic parenchyma can occur. To guard against premature activation, the pancreas has several defense mechanisms. The first is an enzyme, the pancreatic secretory trypsin inhibitor, that can bind to and inactivate 20% of trypsin activity. A second defense mechanism is autolysis of prematurely activated trypsin, and a third defense mechanism involves the action of nonspecific proteases such as alpha-1 antitrypsin 44, 45. For pancreatitis to occur, an initial event must overwhelm these defense mechanisms. Several well-known causes exist, with gallstone obstruction and alcohol abuse the two most prevalent causes. Gallstone obstruction of the ampulla of Vater, which is responsible for 35%-40% of acute pancreatitis cases in the United States, is thought to induce pancreatitis via stasis and reflux of bile into the pancreatic duct 46. Prevailing theories posit that the blockage initiates the pancreatic zymogen cascade that then damages surrounding tissue. Cholecystectomy and bile duct clearance resolve the symptoms, confirming the cause-and-effect relationship 47.
Ethanol abuse is the second most common cause of pancreatitis in the United States, responsible for approximately 30% of cases 48. The pathogenic details of ethanol-induced pancreatitis are yet to be confirmed, but several mechanisms have been proposed. The first involves an oversensitization of pancreatic acinar cells to cholecystokinin and premature zymogen activation. The second suggests that ethanol induces acinar cells to overproduce enzymes that are activated prematurely because of buildup and stasis within the pancreas 49.
Other causes involve smoking, scorpion venom, hypertriglyceridemia, endoscopic retrograde cholangiopancreatography (ERCP), hypercalcemia, steroids, malignancy, infection, trauma, and drugs 50, 51, 52, 53, 54, 55.
Regardless of the mechanism underlying an episode of pancreatitis, once activated, the enzymes will begin to digest the cell membranes of the pancreas, thereby activating an inflammatory response. This response increases the vascular permeability of the pancreas 41. Bleeding, edema, ischemia, and necrosis can ensue 41. The severity of acute pancreatitis can vary as it progresses to systemic inflammatory response syndrome, sepsis, and multiple organ failure 56. Approximately 3%-13% of acute pancreatitis cases develop into chronic pancreatitis 57.
Acute pancreatitis prevention
You can’t prevent pancreatitis, but you can take steps to help you stay healthy.
Maintain a healthy weight or lose weight safely
Maintaining a healthy lifestyle and a healthy weight or losing weight if you’re overweight or obese can help to:
- make your pancreas work better
- lower your chance of getting gallstones, a leading cause of pancreatitis
- prevent obesity, a risk factor for pancreatitis
- prevent diabetes, a risk factor for pancreatitis
Avoid alcohol use
Alcohol use can cause acute and chronic pancreatitis. Talk with your doctor if you need help to stop drinking alcohol.
Avoid smoking
Smoking is a common risk factor for pancreatitis and the chances of getting pancreatitis are even higher in people who smoke and drink alcohol. Talk with your doctor if you need help to stop smoking.
Acute pancreatitis signs and symptoms
People with acute pancreatitis may feel pain in their upper abdomen that may spread to their back.
Acute pancreatitis usually starts with pain that:
- begins slowly or suddenly in your upper abdomen
- sometimes spreads to your back
- can be mild or severe
- may last for several days
Other symptoms may include:
- fever
- nausea and vomiting
- fast heartbeat
- swollen or tender abdomen
People with acute pancreatitis usually look and feel seriously ill and need to see a doctor right away.
A patient with mild acute pancreatitis may experience only minimal tenderness to abdominal palpation. Nevertheless, the pain is constant, usually located in the epigastrium, and generally described as knifelike and radiating to the midcentral back. Patients are restless and may bend forward, bringing their knees to their chest in an effort to alleviate the pain 58, 59.
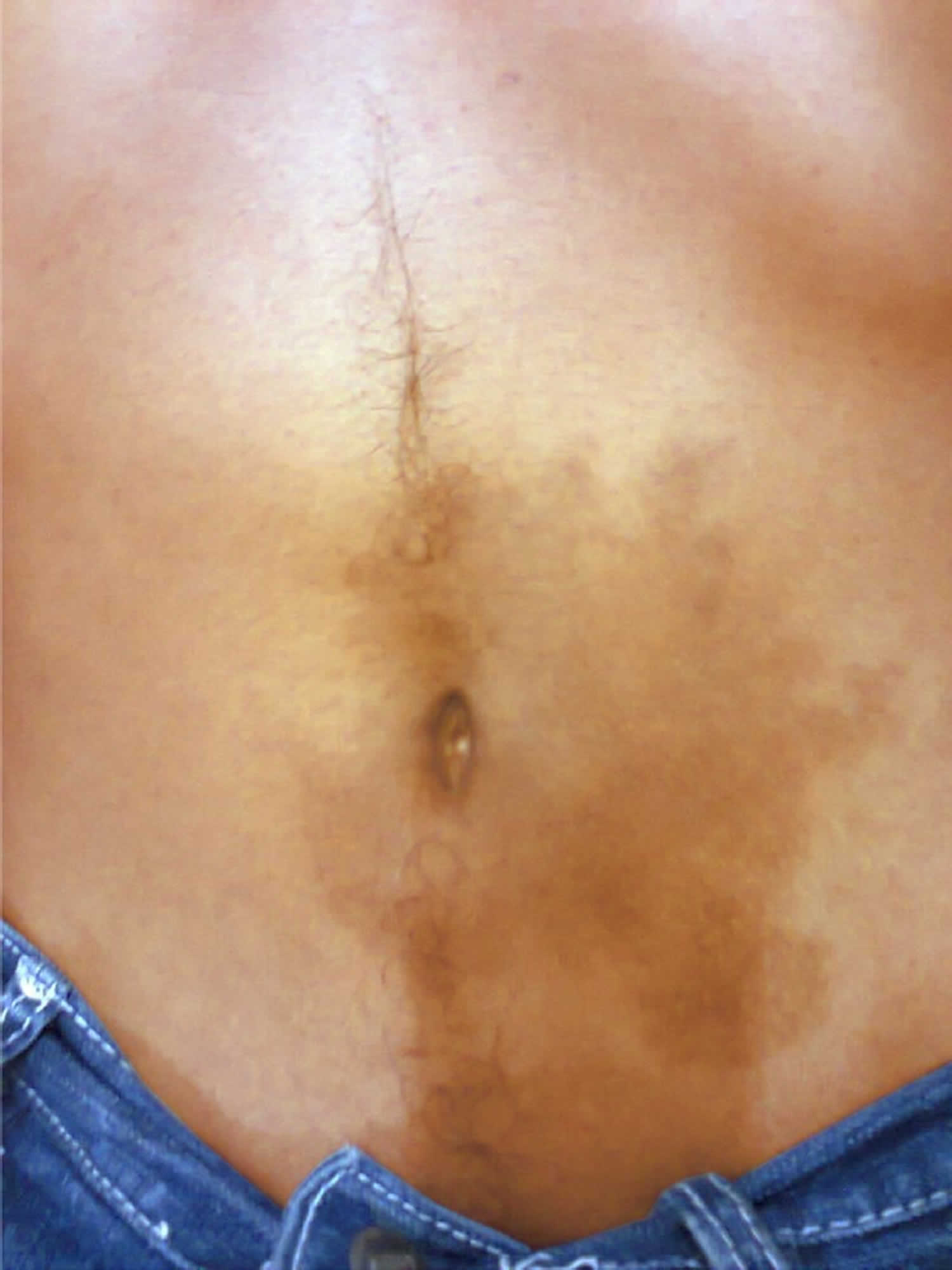
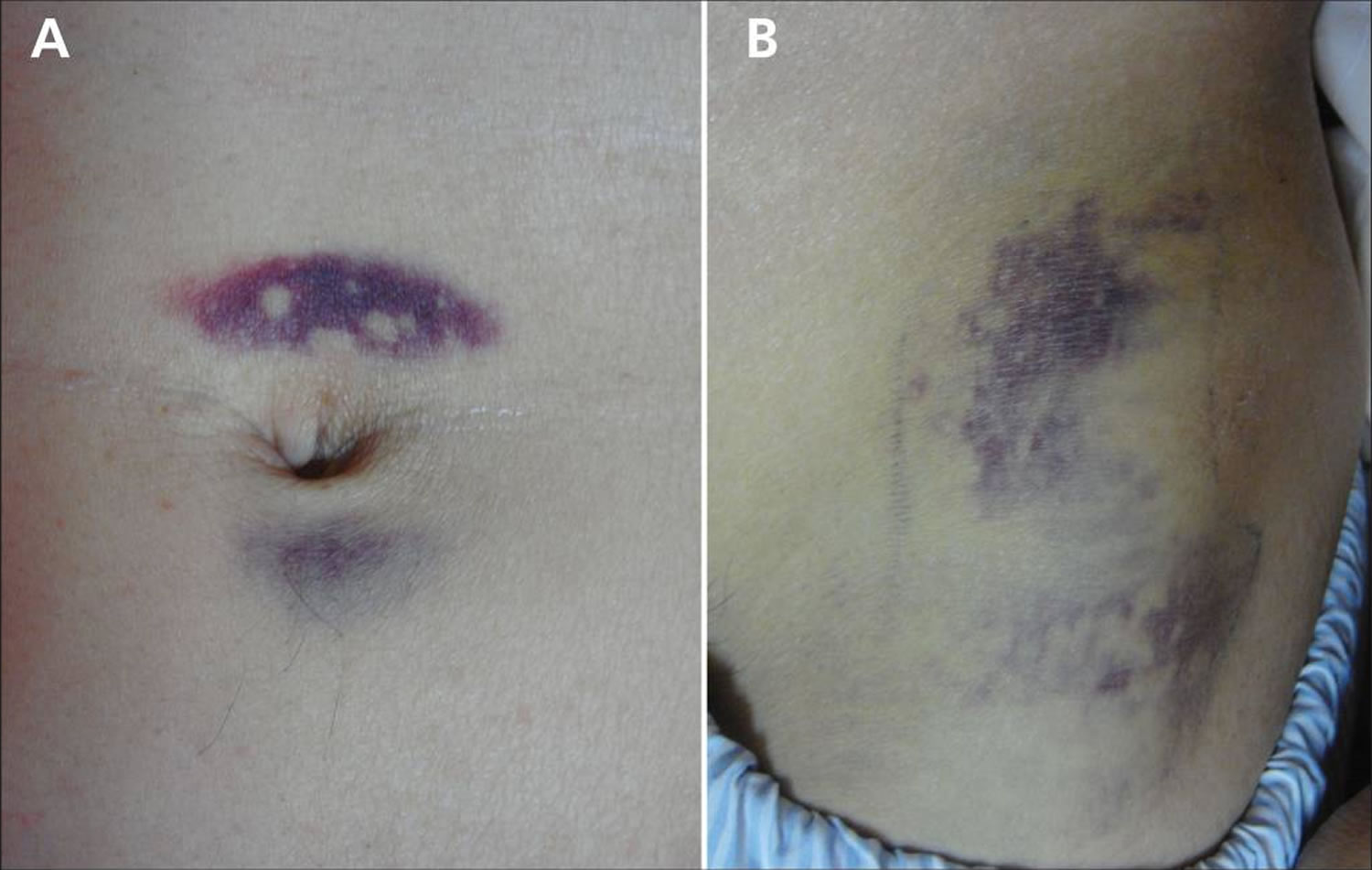
Jaundice (a yellow color of the skin, mucus membranes, or eyes) is a common finding. In 3% of patients with severe acute pancreatitis, flank ecchymosis (Grey Turner sign) or periumbilical ecchymosis (Cullen sign) develops and is suggestive of retroperitoneal hemorrhage. Patients with severe acute pancreatitis can also develop fever, rapid breathing, hypoxemia, and hypotension 60, 61.
Some patients display alterations in mental status. This symptom is more common in drug-induced acute pancreatitis and reflects exposure to drugs or to alcohol but may also result from hypotension, hypoxemia, or the massive release of toxic agents from the inflamed pancreas 53, 62.
Figure 5. Cullen sign
[Source 63 ]Figure 6. Cullen and Grey Turner sign
Footnote: (A) Periumbilical ecchymosis (Cullen sign) and (B) connecting patches of ecchymosis on the left flank (Grey Turner sign) in a 47-year-old man with sudden onset of left-flank pain.
[Source 64 ]Pancreatitis complications
Both acute and chronic pancreatitis can lead to serious complications that include:
- Kidney failure. Acute pancreatitis may cause kidney failure, which can be treated with dialysis if the kidney failure is severe and persistent.
- Breathing problems. Acute pancreatitis can cause chemical changes in your body that affect your lung function, causing the level of oxygen in your blood to fall to dangerously low levels (hypoxemia).
- Infection. Acute pancreatitis can make your pancreas vulnerable to bacteria and infection. Pancreatic infections are serious and require intensive treatment, such as surgery to remove the infected tissue.
- Narrowing or blockage in a bile or pancreatic duct
- Leakage from the pancreatic duct
- Pancreatic pseudocyst. Acute pancreatitis can cause fluid and debris to collect in cystlike pockets in your pancreas. A large pseudocyst that ruptures can cause complications such as internal bleeding and infection.
- Malnutrition. Both acute and chronic pancreatitis can cause your pancreas to produce fewer of the enzymes that are needed to break down and process nutrients from the food you eat. This can lead to malnutrition, diarrhea and weight loss, even though you may be eating the same foods or the same amount of food.
- Diabetes. Damage to insulin-producing cells in your pancreas from chronic pancreatitis can lead to diabetes, a disease that affects the way your body uses blood sugar.
- Pancreatic cancer. Long-standing inflammation in your pancreas caused by chronic pancreatitis is a risk factor for developing pancreatic cancer 2.
- Death.
Pancreatitis complications may arise that include local and systemic consequences. Local complications include fluid accumulation, pancreatic pseudocyst, necrotic collection, and walled-off necrosis. The fluids and necrotic tissue can become secondarily infected, leading to systemic inflammatory response syndrome and sepsis. Systemic complications include splanchnic vein thrombosis, abdominal compartment syndrome, pseudoaneurysm, acute respiratory distress syndrome, and exacerbation of underlying comorbidities such as coronary artery disease and chronic lung disease 65, 66, 67.
The majority of severe pancreatitis complications occur within 48 hours of onset. Ranson’s Criteria (Table 2) was published in 1974 as one of the first prognostic tools established to help clinicians identify severity and mortality of patients with acute pancreatitis 68. A Ranson score of 2 or less is associated with a mortality rate of 0-3% 6.
Table 2. Ranson criteria for acute pancreatitis based on initial lab values
| Ranson criteria at admission | |
|---|---|
| White blood cell count > 16,000 cells/mm³ | 1 point |
| Age > 55 years of age | 1 point |
| Blood glucose > 200 mg/dL | 1 point |
| Aspartate transaminase (AST) > 250 IU/L | 1 point |
| Lactate dehydrogease (LDH) > 350 IU/L | 1 point |
| Ranson criteria within 48 hours | |
| Serum calcium <8.0 mg/dL | 1 point |
| Hematocrit drop >10% | 1 point |
| PaO2 <60 mmHg | 1 point |
| Blood urea nitrogen (BUN) > 5 mg/dL | 1 point |
| Base deficit > 4.0 mEq/L | 1 point |
| Fluid sequestration > 6L | 1 point |
Footnote: Ranson Criteria for acute pancreatitis total point indicates a prognosis for the patient as follows:
- 0 to 2 points: Severe pancreatitis is unlikely; Mortality 0% to 3%
- 3 to 4 points: Severe pancreatitis is likely; Mortality 15%
- 5 to 6 points: Severe pancreatitis is likely; Mortality 40%
- 7 to 11 points: Severe pancreatitis is likely; Mortality nearly 100%
Alternatively, the Bedside Index for Severity in Acute Pancreatitis (BISAP) was developed in 2008 and has been shown to be a more accurate prognostic tool that predicts the mortality risk in acute pancreatitis based on the data within the first 24 hour (Table 3) 70. The Bedside Index for Severity in Acute Pancreatitis (BISAP) scoring index is to be used in the first 24 hour of presentation and the variables included in Bedside Index of Severity in Acute Pancreatitis (BISAP) score include 70:
- Blood urea nitrogen (BUN) > 25 mg/dL
- Abnormal mental status (Glasgow coma score <15)
- Evidence of systemic inflammatory response syndrome (SIRS)
- Greater than or equal to 60 years of age
- Presence of pleural effusion
To calculate the BISAP, sum the number of positive variables (0–5). Multiple studies have validated the performance of the BISAP and pooled estimates demonstrate its accuracy in predicting acute pancreatitis severity 71, 72.
Table 3. Bedside Index for Severity in Acute Pancreatitis (BISAP) score
| Bedside Index for Severity in Acute Pancreatitis (BISAP) score for pancreatitis mortality | |
|---|---|
| Blood urea nitrogen > 25 mg/dL | 1 point |
| Impaired mental status (disorientation, lethargy, somnolence, coma, stupor) | 1 point |
≥ 2 Systemic Inflammatory Response Syndrome (SIRS) Criteria
| 1 point |
| Age ≥ 60 years old | 1 point |
| Pleural effusion present on imaging | 1 point |
Footnotes: Bedside Index for Severity in Acute Pancreatitis (BISAP) score of 0 has < 1% and scores ≤ 2 have 1.9% mortality risk, respectively.
[Source 73 ]Acute pancreatitis diagnosis
To diagnose pancreatitis and find its causes, doctors use:
- Your medical history. Your doctor will ask:
- about your symptoms
- if you have a history of health conditions or concerns that make you more likely to get pancreatitis—including medicines you are taking
- if you have a personal or family medical history of pancreatitis or gallstones
- A physical exam. During a physical exam, your doctor will:
- examine your body
- check your abdomen for pain, swelling, or tenderness
- Lab and imaging tests
Tests and procedures used to diagnose pancreatitis include:
- Blood tests to look for elevated levels of pancreatic enzymes, along with white blood cells, kidney function and liver enzymes
- Abdominal ultrasound to look for gallstones and pancreas inflammation
- Computerized tomography (CT) scan of your abdomen to look for gallstones and assess the extent of pancreas inflammation
- Magnetic resonance imaging (MRI) of your abdomen to look for abnormalities in the gallbladder, pancreas and ducts
- Endoscopic ultrasound to look for inflammation and blockages in the pancreatic duct or bile duct
- Stool tests in chronic pancreatitis to measure levels of fat that could suggest your digestive system isn’t absorbing nutrients adequately
Your doctor may recommend other tests, depending on your particular situation.
Lab tests
Lab tests to help diagnose pancreatitis include the following:
- Blood tests. Your physician may take a blood sample from you and send the sample to a lab to test for:
- high amylase and lipase levels—digestive enzymes made in your pancreas
- high blood glucose, also called blood sugar
- high levels of blood fats, called lipids
- signs of infection or inflammation of the bile ducts, pancreas, gallbladder, or liver
- pancreatic cancer
- Stool tests. Your doctor may test a stool sample to find out if a person has fat malabsorption.
Imaging tests
Your doctor also use imaging tests to diagnose pancreatitis. A technician performs most tests in an outpatient center, a hospital, or a doctor’s office. You don’t need anesthesia, a medicine to keep you calm, for most of these tests.
- Ultrasound. Ultrasound uses a device called a transducer, which bounces safe, painless sound waves off your organs to create a picture of their structure. Ultrasound can find gallstones.
- Computed tomography (CT) scan. CT scans create pictures of your pancreas, gallbladder, and bile ducts. CT scans can show pancreatitis or pancreatic cancer.
- Magnetic resonance cholangiopancreatography (MRCP). MRCP uses a magnetic resonance imaging (MRI) machine, which creates pictures of your organs and soft tissues without x-rays. Your doctor or a specialist may use MRCP to look at your pancreas, gallbladder, and bile ducts for causes of pancreatitis.
- Endoscopic ultrasound (EUS). Your doctor inserts an endoscope—a thin, flexible tube—down your throat, through your stomach, and into your small intestine. The doctor turns on an ultrasound attachment to create pictures of your pancreas and bile ducts. Your doctor may send you to a gastroenterologist to perform this test.
- Pancreatic Function Test (PFT). Your doctor may use this test to measure how your pancreas responds to secretin, a hormone made by the small intestine. This test is done only at some centers in the United States.
Acute pancreatitis treatment
Mild acute pancreatitis usually goes away in a few days with rest and treatment.
If your pancreatitis is more severe, your treatments for acute pancreatitis in the hospital may include:
- Early eating. Old data suggested to stop eating for a couple of days in the hospital in order to give your pancreas a chance to recover. This is no longer practiced. Newer data have suggested that eating as soon as you tolerate food helps heal the pancreas. As the inflammation in your pancreas improves and pain symptoms improve, you should begin drinking clear liquids and eating bland foods. With time, you can go back to your normal diet. If your pancreatitis symptoms persist and you still experience pain when eating, your doctor may recommend a feeding tube to help you get nutrition.
- Pain medications. Pancreatitis can cause severe pain. Your health care team will give you medications to help control the pain.
- Intravenous (IV) fluids. As your body devotes energy and fluids to repairing your pancreas, you may become dehydrated. For this reason, you’ll receive extra fluids through a vein in your arm during your hospital stay.
Pancreatitis-related pain in some patients has been shown to respond to pancreatic enzyme replacement 74 and possibly with supplementation of antioxidants such as S-Adenosyl Methionine (SAMe) (800 mg per day), Vitamin C (180 mg per day), Vitamin E (30 mg per day), Vitamin A (2,400 microg per day), and selenium (75 microg per day) 75. The central nervous system and pain processing system have been implicated as a mechanism of pancreatitis-related pain, so the use of tricyclic antidepressants or gabapentin may be helpful 76. Additionally, while nonsteroidal anti-inflammatory drugs (NSAIDs) are preferred over opioid analgesics for pain relief, they may be contraindicated due to the patient’s comorbidities or not tolerated due to side effects. If opioid analgesics are to be prescribed, long-acting formulations are preferred over short or intermediate acting forms 77.
Once your pancreatitis is under control, your doctor will evaluate and treat the underlying cause of your pancreatitis.
Depending on the cause of your acute pancreatitis, treatment may include:
- Procedures to remove bile duct obstructions. Pancreatitis caused by a narrowed or blocked bile duct may require procedures to open or widen the bile duct. A procedure called endoscopic retrograde cholangiopancreatography (ERCP) uses a long tube with a camera on the end to examine your pancreas and bile ducts. The tube is passed down your throat, and the camera sends pictures of your digestive system to a monitor. Anesthesia is used for this procedure. Your gastroenterologist may use ERCP to diagnose problems in the bile duct and pancreatic duct and in removing obstructions, such as gallstones. In some people, however, ERCP can also lead to acute pancreatitis.
- Gallbladder surgery. If gallstones caused your pancreatitis, your doctor will recommend surgery to remove your gallbladder called cholecystectomy. Having surgery to remove the gallbladder within a few days after you are admitted to the hospital lowers the chance of complications. If you have severe pancreatitis, your doctor may advise delaying surgery to first treat complications.
- Pancreas procedures. Endoscopic procedures may be necessary to drain fluid in your abdomen if you have pancreatic necrosis, fluid collections, an abscess or infected pseudocyst, or a large pseudocyst causing pain or bleeding 78, 79, 80. Your doctor may remove damaged tissue from your pancreas. Local complications are almost always diffuse in the early days of acute pancreatitis, so intervention is better delayed until these are walled off and more suitable for drainage, preferably over 4 weeks, with appropriate patient counseling 1. The presence of infection, as may be evident from the presence of gas on contrast-enhanced CT scans, and critical illness are relative indications to expedite drainage and/or necrosectomy, but randomized trial evidence favors delayed intervention 81. While endoscopic necrosectomy often requires several repeated procedures, it has the advantage of internal drainage without external irrigation, allowing patients to be discharged at an earlier date for repeat outpatient procedures, reducing health service usage and costs 82, 83. In contrast, minimally invasive or open necrosectomy requires continued external irrigation to flush away build-up of septic material, prolonging hospital stays, and imposing additional burdens on patients and hospital staff.
- Pancreatic Enzyme Replacement. Pancreatic exocrine insufficiency can be identified in > 50% of patients during their inpatient stay with acute pancreatitis, although this frequency falls during follow-up, persisting in a minority, including > 50% of those with pancreatic necrosis 84. The normal adult human pancreas secretes between one and two million units of lipase daily, with many proteases, carbohydrate hydrolases, lipid hydrolases and nucleases 85, whereas acute pancreatitis inhibits secretion 84. Pancreatic enzyme replacement therapy is likely to be beneficial in moderately severe and severe acute pancreatitis with oral/enteral feeding until fecal elastase-1 testing is repeatedly normal (≥ 200 μg/g); this therapy is recommended routinely for such patients, and long term for patients with > 50% necrotizing pancreatitis. The presence of exocrine pancreatic insufficiency suggested by steatorrhea warrants pancreatic enzyme replacement therapy after acute pancreatitis of any severity, and indefinitely should fecal elastase-1 remain < 100 μg/g (this therapy does not have to be stopped for fecal elastase-1 testing) 84. A minimum standard dose for adult patients of one of the licensed preparations (Creon, Nutrizyme, Pancrease HL and Pancrex V in the UK) is 50,000 (lipase) units with a meal and half that with a snack, increased if steatorrhoea is insufficiently ameliorated. The total daily dose should be tailored to oral/enteral intake and in children should be adjusted based on a combination of body weight and intake, not exceeding a maximum daily dose of 10,000 IU/kg to achieve satisfactory growth and normal fat-soluble vitamin levels.
- Treatment of infection. Randomized clinical trials have not shown sufficient advantage for prophylactic antibiotics during hospital admission for acute pancreatitis 27, yet recent surveys in 22 countries indicate global overuse 86. Patients who develop moderately severe and severe acute pancreatitis, however, are at increased risk of infective complications, in association with either persistent organ failure or local complications. Bacterial translocation across the injured gut and a defective gastrointestinal permeability barrier is a consequence of the systemic inflammatory response (SIR). Despite enteral nutrition, bacterial translocation may still occur, an important although not the only route of infective complications, notably infecting necrosis; early infection is associated with increased mortality 87. Prompt recognition, for example gas bubbles in necrosis on contrast enhanced CT scan, source control may necessitate percutaneous drainage, appropriate antibiotics, physiological stabilisation, and optimal further interventional approaches are fundamental 88. Nevertheless, during the first 4 weeks of acute pancreatitis inflammation, fluid collections and necrosis are diffuse, such that major interventions are less likely to be effective and can increase patient instability 1. The mainstay of treatment may need to be antibiotics appropriate to Gram-negative intra-abdominal infection, for example piperacillin-tazobactam or tigecycline or a third-generation cephalosporin with metronidazole 88; the choice depends in part on the availability of samples for culture and patient response, best determined in consultation with clinical microbiologists. In the most severe disease, fungal infection is characteristic, may be brought on by extensive antibacterial treatments, and requires early and prolonged treatment with antifungals 89.
- Treatment for alcohol dependence. Drinking several drinks a day over many years can cause pancreatitis. If this is the cause of your pancreatitis, your doctor may recommend you enter a treatment program for alcohol addiction. Continuing to drink alcohol would worsen your pancreatitis and lead to serious complications.
- Medication changes. If a medication is deemed to be a cause of acute pancreatitis, your doctor may stop the medication and work with you to find alternative options.
- Management of diabetes. Elevated blood sugar is indicative of more severe acute pancreatitis. Development of impaired glucose tolerance can be as high as 60% 5 years following a first attack of acute pancreatitis 90; the greatest risk is in those who develop necrotizing pancreatitis. Those requiring necrosectomy are among those who lose the most pancreatic tissue and therefore the most islets of Langerhans. Development of clinical diabetes has been estimated around 15% and 40% following mild or severe acute pancreatitis, respectively 91. Due to the loss of pancreatic tissue, insulin production is reduced, multiple daily insulin injections may be appropriate. For those with recurrent hypoglycaemia despite optimized regimens, continuous subcutaneous insulin pump therapy with or without continuous glucose monitoring is an alternative, guided by specialists in diabetes care 92.
- Treatment for autoimmune pancreatitis. Recommended first-line treatment for autoimmune pancreatitis is oral prednisolone (2 mg/kg, max 60 mg daily) tapered slowly by 5–10 mg, aiming to keep the patient on a maintenance dose (5–7.5 mg/day) for some 6 months and then close follow up 1. During treatment patients should be monitored closely for biochemical, clinical or radiological progress and side effects such as hypertension and diabetes 93. Escalating immunomodulatory treatment with azathioprine, 6-mercaptopurine or rituximab are alternatives for steroid-resistant or relapsing disease 94.
Acute pancreatitis treatment at home
Once you have recovered from your acute pancreatitis and left the hospital, you can take steps to continue your recovery from pancreatitis, such as:
- Stop drinking alcohol. Even if alcohol was not deemed to be the cause of acute pancreatitis, it is prudent to stop drinking alcohol while recovering. If you’re unable to stop drinking alcohol on your own, ask your doctor for help. Your doctor can refer you to local programs to help you stop drinking. Continuing to drink alcohol when you have acute pancreatitis can lead to:
- more episodes of acute pancreatitis
- chronic pancreatitis
- When people with chronic pancreatitis caused by alcohol use continue to drink alcohol, the condition is more likely to lead to severe complications and even death.
- Stop smoking. If you smoke, quit. If you don’t smoke, don’t start. If you can’t quit on your own, ask your doctor for help. Medications and counseling can help you quit smoking. Smoking with acute pancreatitis, especially if it’s caused by alcohol use, greatly raises the chances that your pancreatitis will become chronic. Smoking with pancreatitis also may raise your risk of pancreatic cancer.
- Choose a low-fat diet. Choose a diet that limits fat and emphasizes fresh fruits and vegetables, whole grains, and lean protein.
- Drink more fluids. Pancreatitis can cause dehydration, so drink more fluids throughout the day and limit caffeine. It may help to keep a water bottle or glass of water with you. Health care professionals strongly advise people with pancreatitis not to drink any alcohol, even if your pancreatitis is mild.
Alternative medicine
Alternative therapies can’t treat pancreatitis, but some alternative therapies may help you cope with the pain associated with pancreatitis.
People with chronic pancreatitis may experience constant pain that isn’t easily controlled with medications. Using complementary and alternative medicine therapies along with medications prescribed by your doctor may help you feel more in control of your pain.
Examples of alternative therapies that may help you cope with pain include:
- Meditation
- Relaxation exercises
- Yoga
- Acupuncture
Acute pancreatitis prognosis
Acute pancreatitis is a severe disorder that still carries a mortality of 5 to 15%, depending on the cause, patient age, and comorbidity 95, 96. Patients with gallstone pancreatitis generally have higher mortality than those with alcoholic pancreatitis 95, 96. In addition, the presence of type 2 diabetes significantly increases the risk of complications and death 97. In patients with multiorgan involvement, the mortality can be as high as 20% 95. Most deaths are due to multiorgan failure and hypotensive shock. In patients with pancreatic necrosis without organ failure, the mortality approaches zero 96.
Various classifications have been developed to assess the prognosis of patients with acute pancreatitis, but most are cumbersome for practical use 98, 99, 100.
In the first week of illness, most deaths result from multiorgan system failure 96. In subsequent weeks, infection plays a more significant role, but organ failure still constitutes a major cause of mortality. Acute respiratory distress syndrome (ARDS), acute renal failure, cardiac depression, hemorrhage, and hypotensive shock all may be systemic manifestations of acute pancreatitis in its most severe form.
Identifying patients in the greatest need of aggressive medical treatment by differentiating their disease severity as mild or severe is recommended. In mild disease, the pancreas exhibits interstitial edema, an inflammatory infiltrate without hemorrhage or necrosis, and, usually, minimal or no organ dysfunction. In severe disease, the inflammatory infiltrate is severe, associated with necrosis of the parenchyma, often accompanied by evidence of severe gland dysfunction, and it may be associated with multiorgan system failure.
The International Association of Pancreatology and American Pancreatic Association guidelines states that developing a systemic inflammatory response syndrome (SIRS) at admission and persistent for 48 hours predict severe acute pancreatitis 27. A persistent SIRS is associated with a mortality of 25% when compared with 8% for a transient SIRS 101. However, the sensitivity and specificity for predicting mortality with persistent SIRS are 77-89% and 79-86%, respectively 102. SIRS on day 1 predicted severe disease with high sensitivity (85%-100%). The absence of SIRS on day 1 was associated with a high negative predictive value (98%-100%). Patients with a higher number of systemic inflammatory response (SIR) criteria on day 1 and persistent SIRS had an increased risk for severe disease 103.
Different strategies have been used to assess the severity of acute pancreatitis and predict outcome. Several clinical scoring systems (eg, Ranson criteria, BISAP, Glasgow, Imrie) are available. The APACHE II scoring system, though cumbersome, appears to be the best validated 96 (https://reference.medscape.com/calculator/12/apache-ii). Biological markers have also been used for this purpose. Genetic markers are being studied and have not yet come into clinical use.
Bedside Index for Severity in Acute Pancreatitis (BISAP) is a relatively recent addition (see Table 3 above). The Bedside Index for Severity in Acute Pancreatitis (BISAP) index has good predictive performance for both severe acute pancreatitis and mortality and has been validated prospectively, and it is simple and easy to calculate from initial presentation data. Compared with other scoring systems like Ranson criteria (see Table 2 above) and APACHE II score (https://reference.medscape.com/calculator/12/apache-ii), the BISAP score outperforms in specificity but has a suboptimal sensitivity for mortality 104.
Suppiah et al 105 examined the prognostic value of the neutrophil-lymphocyte ratio (NLR) in 146 consecutive patients with acute pancreatitis. They found that elevation of the neutrophil-lymphocyte ratio during the first 48 hours of hospital admission was significantly associated with severe acute pancreatitis and was an independent negative prognostic indicator 105. The neutrophil-lymphocyte ratio is calculated from the white cell differential and provides an indication of inflammation.
Peritoneal lavage has a high specificity (93%); however, it has a low sensitivity (54%). Dynamic CT scanning of the abdomen is widely available and useful in predicting the outcome of acute pancreatitis. When the Balthazar criteria are used, sensitivity is 87% and specificity is 88%. The modified CT Severity Index (see Table 4) is a sum of scores retrieved from the Balthazar score and those from the evaluation of pancreatic necrosis. It can aid in predicting mortality and detecting any necrosis on CT imaging as a predictor of high mortality 106. Total points are out of ten which helps determine the grade of pancreatitis and aid in treatment; 0 to 2 is mild, 4 to 6 is moderate, and 8 to 10 is severe.
Table 4. CT Severity Index for acute pancreatitis
| Prognostic indicator | Points |
|---|---|
| Pancreatic inflammation | |
| 1. Normal pancreas | 0 |
| 2. Intrinsic pancreatic abnormalities with or without inflammatory changes in peripancreatic fat | 2 |
| 3. Pancreatic or peripancreatic fluid collection and peripancreatic fat necrosis | 4 |
| Pancreatic necrosis | |
| 1. None | 0 |
| 2. Less than or equal to 30% | 2 |
| 3. Greater than 30% | 4 |
| Extrapancreatic complications ( one or more of the following complications, i.e., pleural effusions, abdominal ascites, vascular complications, parenchymal or gastrointestinal tract involvement) | 2 |
Khan et al 107 examined the prognostic value of Modified Early Warning Score (MEWS) in identifying severe acute pancreatitis in 200 patients admitted to a single institution (Table 5). The investigators tracked the highest and mean daily scores. They found that patients with a high Modified Early Warning Score (MEWS) value > 2 on day one or mean value > 1.2 on day two was most accurate in predicting severe acute pancreatitis 107. The investigators concluded that MEWS is a reliable, safe, and inexpensive score that can be used easily at all levels of health care for prognosticating patients with acute pancreatitis.
Table 5. Modified Early Warning Score for acute pancreatitis
[Source 107 ]In a retrospective study of data from 822 patients hospitalized with acute pancreatitis, Mikolasevic et al 108 found that patients who had nonalcoholic fatty liver at admission (n = 198; 24.1%) had a statistically higher incidence of moderately severe (35.4% vs 14.6%) and severe acute pancreatitis (20.7% vs 9.6%) than those without nonalcoholic fatty liver. Moreover, these patients had higher (1) C-reactive protein levels not only on the day of admission but also at day 3, (2) APACHE II scores at admission, (3) CT scan severity index, and (4) occurrence of organ failure and local complications. Although mortality was also higher in the nonalcoholic fatty liver group compared to the group without this disease, the difference was not statistically significant 108.
- Szatmary P, Grammatikopoulos T, Cai W, Huang W, Mukherjee R, Halloran C, Beyer G, Sutton R. Acute Pancreatitis: Diagnosis and Treatment. Drugs. 2022 Aug;82(12):1251-1276. doi: 10.1007/s40265-022-01766-4[↩][↩][↩][↩][↩]
- Yadav D, Lowenfels AB. The epidemiology of pancreatitis and pancreatic cancer. Gastroenterology. 2013 Jun;144(6):1252-61. doi: 10.1053/j.gastro.2013.01.068[↩][↩]
- Definition & Facts for Pancreatitis. https://www.niddk.nih.gov/health-information/digestive-diseases/pancreatitis/definition-facts[↩]
- Peery AF, Crockett SD, Barritt AS, Dellon ES, Eluri S, Gangarosa LM, Jensen ET, Lund JL, Pasricha S, Runge T, Schmidt M, Shaheen NJ, Sandler RS. Burden of Gastrointestinal, Liver, and Pancreatic Diseases in the United States. Gastroenterology. 2015 Dec;149(7):1731-1741.e3. doi: 10.1053/j.gastro.2015.08.045[↩]
- Jones MR, Hall OM, Kaye AM, Kaye AD. Drug-induced acute pancreatitis: a review. Ochsner J. 2015 Spring;15(1):45-51. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4365846[↩]
- Forsmark CE, Vege SS, Wilcox CM. Acute Pancreatitis. N Engl J Med. 2016 Nov 17;375(20):1972-1981. doi: 10.1056/NEJMra1505202[↩][↩]
- Della Corte C, Faraci S, Majo F, Lucidi V, Fishman DS, Nobili V. Pancreatic disorders in children: new clues on the horizon. Dig Liver Dis. 2018;50(9):886–893. doi: 10.1016/j.dld.2018.06.016[↩]
- Vege SS, Yadav D, Chari ST. Pancreatitis. In: Talley NJ, GR Locke, Saito YA, editors. GI Epidemiology. Malden, MA: Blackwell Publishing;; 2007.[↩]
- Yadav D, Lowenfels AB. The epidemiology of pancreatitis and pancreatic cancer. Gastroenterology. 2013;144(6):1252–1261. doi: 10.1053/j.gastro.2013.01.068[↩]
- Roberts SE, Morrison-Rees S, John A, Williams JG, Brown TH, Samuel DG. The incidence and aetiology of acute pancreatitis across Europe. Pancreatology. 2017;17(2):155–165. doi: 10.1016/j.pan.2017.01.005[↩]
- Nielsen JK, Olafsson S, Bergmann OM, Runarsdottir V, Hansdottir I, Sigurdardottir R, et al. Lifetime drinking history in patients with alcoholic liver disease and patients with alcohol use disorder without liver disease. Scand J Gastroenterol. 2017;52(6–7):762–767. doi: 10.1080/00365521.2017.1295466[↩]
- Lankisch PG, Assmus C, Lehnick D, Maisonneuve P, Lowenfels AB. Acute pancreatitis: does gender matter? Dig Dis Sci. 2001;46(11):2470–2474. doi: 10.1023/A:1012332121574[↩]
- Matta B, Gougol A, Gao X, Reddy N, Talukdar R, Kochhar R, Goenka MK, Gulla A, Gonzalez JA, Singh VK, Ferreira M, Stevens T, Barbu ST, Nawaz H, Gutierrez SC, Zarnescu NO, Capurso G, Easler J, Triantafyllou K, Pelaez-Luna M, Thakkar S, Ocampo C, de-Madaria E, Cote GA, Wu BU, Paragomi P, Pothoulakis I, Tang G, Papachristou GI. Worldwide Variations in Demographics, Management, and Outcomes of Acute Pancreatitis. Clin Gastroenterol Hepatol. 2020 Jun;18(7):1567-1575.e2. doi: 10.1016/j.cgh.2019.11.017[↩]
- Whitcomb DC, LaRusch J, Krasinskas AM, Klei L, Smith JP, Brand RE, et al. Common genetic variants in the CLDN2 and PRSS1-PRSS2 loci alter risk for alcohol-related and sporadic pancreatitis. Nat Genet. 2012;44(12):1349–1354. doi: 10.1038/ng.2466[↩][↩]
- Pownall HJ, Ballantyne CM, Kimball KT, Simpson SL, Yeshurun D, Gotto AM., Jr Effect of moderate alcohol consumption on hypertriglyceridemia: a study in the fasting state. Arch Intern Med. 1999;159(9):981–987. doi: 10.1001/archinte.159.9.981[↩][↩]
- Carr RA, Rejowski BJ, Cote GA, Pitt HA, Zyromski NJ. Systematic review of hypertriglyceridemia-induced acute pancreatitis: a more virulent etiology? Pancreatology. 2016;16(4):469–476. doi: 10.1016/j.pan.2016.02.011[↩][↩]
- Murphy MJ, Sheng X, MacDonald TM, Wei L. Hypertriglyceridemia and acute pancreatitis. JAMA Intern Med. 2013;173(2):162–164. doi: 10.1001/2013.jamainternmed.477[↩][↩]
- Kiss L, Fur G, Matrai P, Hegyi P, Ivany E, Cazacu IM, et al. The effect of serum triglyceride concentration on the outcome of acute pancreatitis: systematic review and meta-analysis. Sci Rep. 2018;8(1):14096. doi: 10.1038/s41598-018-32337-x[↩][↩]
- Kochar B, Akshintala VS, Afghani E, Elmunzer BJ, Kim KJ, Lennon AM, et al. Incidence, severity, and mortality of post-ERCP pancreatitis: a systematic review by using randomized, controlled trials. Gastrointest Endosc. 2015;81(1):143 e9–149 e9. doi: 10.1016/j.gie.2014.06.045[↩][↩]
- Njei B, McCarty TR, Muniraj T, Sharma P, Jamidar PA, Aslanian HR, et al. Comparative effectiveness of pharmacologic and endoscopic interventions for prevention of post-ERCP pancreatitis: a network meta-analysis. Endosc Int Open. 2020;8(1):E29–e40. doi: 10.1055/a-1005-6366[↩][↩]
- Vinklerová I, Procházka M, Procházka V, Urbánek K. Incidence, severity, and etiology of drug-induced acute pancreatitis. Dig Dis Sci. 2010;55(10):2977–2981. doi: 10.1007/s10620-010-1277-3[↩][↩]
- Spanier BW, Tuynman HA, van der Hulst RW, Dijkgraaf MG, Bruno MJ. Acute pancreatitis and concomitant use of pancreatitis-associated drugs. Am J Gastroenterol. 2011;106(12):2183–2188. doi: 10.1038/ajg.2011.303[↩][↩]
- Yadav D, Hawes RH, Brand RE, Anderson MA, Money ME, Banks PA, et al. Alcohol consumption, cigarette smoking, and the risk of recurrent acute and chronic pancreatitis. Arch Intern Med. 2009;169(11):1035–1045. doi: 10.1001/archinternmed.2009.125[↩][↩]
- Setiawan VW, Pandol SJ, Porcel J, Wilkens LR, Le Marchand L, Pike MC, et al. Prospective study of alcohol drinking, smoking, and pancreatitis: the multiethnic cohort. Pancreas. 2016;45(6):819–825. doi: 10.1097/MPA.0000000000000657[↩][↩]
- Kiriyama S, Gabata T, Takada T, Hirata K, Yoshida M, Mayumi T, et al. New diagnostic criteria of acute pancreatitis. J Hepatobiliary Pancreat Sci. 2010;17(1):24–36. doi: 10.1007/s00534-009-0214-3[↩]
- Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, et al. Classification of acute pancreatitis—2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62(1):102–111. doi: 10.1136/gutjnl-2012-302779[↩]
- Working Group IAP/APA Acute Pancreatitis Guidelines. IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013 Jul-Aug;13(4 Suppl 2):e1-15. doi: 10.1016/j.pan.2013.07.063[↩][↩][↩][↩][↩]
- Vege SS, DiMagno MJ, Forsmark CE, Martel M, Barkun AN. Initial medical treatment of acute pancreatitis: American Gastroenterological Association Institute technical review. Gastroenterology. 2018;154(4):1103–1139. doi: 10.1053/j.gastro.2018.01.031[↩]
- Pancreatitis. https://www.nice.org.uk/guidance/ng104/chapter/Recommendations[↩]
- Leppäniemi A, Tolonen M, Tarasconi A, Segovia-Lohse H, Gamberini E, Kirkpatrick AW, Ball CG, Parry N, Sartelli M, Wolbrink D, van Goor H, Baiocchi G, Ansaloni L, Biffl W, Coccolini F, Di Saverio S, Kluger Y, Moore E, Catena F. 2019 WSES guidelines for the management of severe acute pancreatitis. World J Emerg Surg. 2019 Jun 13;14:27. doi: 10.1186/s13017-019-0247-0[↩]
- Petrov MS, Yadav D. Global epidemiology and holistic prevention of pancreatitis. Nat Rev Gastroenterol Hepatol. 2019;16(3):175–184. doi: 10.1038/s41575-018-0087-5[↩]
- Gardner TB. Acute pancreatitis. Ann Intern Med. 2021;174(2):itc17–itc32. doi: 10.7326/AITC202102160[↩]
- Sarles H, Sarles JC, Muratore R, Guien C. Chronic inflammatory sclerosis of the pancreas—an autonomous pancreatic disease? Am J Dig Dis. 1961;6:688–698. doi: 10.1007/BF02232341[↩]
- Yoshida K, Toki F, Takeuchi T, Watanabe S, Shiratori K, Hayashi N. Chronic pancreatitis caused by an autoimmune abnormality. Proposal of the concept of autoimmune pancreatitis. Dig Dis Sci. 1995;40(7):1561–1568. doi: 10.1007/BF02285209[↩]
- Zen Y, Grammatikopoulos T, Hadzic N. Autoimmune pancreatitis in children: insights into the diagnostic challenge. J Pediatr Gastroenterol Nutr. 2014;59(5):e42–e45. doi: 10.1097/MPG.0b013e3182994559[↩]
- Zen Y, Grammatikopoulos T, Heneghan MA, Vergani D, Mieli-Vergani G, Portmann BC. Sclerosing cholangitis with granulocytic epithelial lesion: a benign form of sclerosing cholangiopathy. Am J Surg Pathol. 2012;36(10):1555–1561. doi: 10.1097/PAS.0b013e31825faae0[↩]
- Perugino CA, Stone JH. IgG4-related disease: an update on pathophysiology and implications for clinical care. Nat Rev Rheumatol. 2020;16(12):702–714. doi: 10.1038/s41584-020-0500-7[↩]
- Lorenzo D, Maire F, Stefanescu C, Gornet JM, Seksik P, Serrero M, et al. Features of autoimmune pancreatitis associated with inflammatory bowel diseases. Clin Gastroenterol Hepatol. 2018;16(1):59–67. doi: 10.1016/j.cgh.2017.07.033[↩]
- Zhang R, Deng L, Jin T, Zhu P, Shi N, Jiang K, et al. Hypertriglyceridaemia-associated acute pancreatitis: diagnosis and impact on severity. HPB (Oxford) 2019;21(9):1240–1249. doi: 10.1016/j.hpb.2019.01.015[↩][↩]
- Wang Q, Wang G, Qiu Z, He X, Liu C. Elevated serum triglycerides in the prognostic assessment of acute pancreatitis: a systematic review and meta-analysis of observational studies. J Clin Gastroenterol. 2017;51(7):586–593. doi: 10.1097/MCG.0000000000000846[↩]
- Kaurich T. Drug-induced acute pancreatitis. Proc (Bayl Univ Med Cent). 2008 Jan;21(1):77-81. doi: 10.1080/08998280.2008.11928366[↩][↩][↩]
- Kitamoto Y, Yuan X, Wu Q, McCourt DW, Sadler JE. Enterokinase, the initiator of intestinal digestion, is a mosaic protease composed of a distinctive assortment of domains. Proc Natl Acad Sci U S A. 1994 Aug 2;91(16):7588-92. doi: 10.1073/pnas.91.16.7588[↩]
- Yamashina I. The action of enterokinase on trypsinogen. Biochim Biophys Acta. 1956 May;20(2):433–434. doi: 10.1016/0006-3002(56)90329-8[↩]
- Aoun E, Chang CC, Greer JB, Papachristou GI, Barmada MM, Whitcomb DC. Pathways to injury in chronic pancreatitis: decoding the role of the high-risk SPINK1 N34S haplotype using meta-analysis. PLoS One. 2008 Apr 16;3(4):e2003. doi: 10.1371/journal.pone.0002003[↩]
- Rowntree, R.K. and Harris, A. (2003), The Phenotypic Consequences of CFTR Mutations. Annals of Human Genetics, 67: 471-485. https://doi.org/10.1046/j.1469-1809.2003.00028.x[↩]
- Forsmark CE, Baillie J; AGA Institute Clinical Practice and Economics Committee; AGA Institute Governing Board. AGA Institute technical review on acute pancreatitis. Gastroenterology. 2007 May;132(5):2022-44. doi: 10.1053/j.gastro.2007.03.065[↩]
- Moreau JA, Zinsmeister AR, Melton LJ 3rd, DiMagno EP. Gallstone pancreatitis and the effect of cholecystectomy: a population-based cohort study. Mayo Clin Proc. 1988 May;63(5):466-73. doi: 10.1016/s0025-6196(12)65644-4[↩]
- Yang AL, Vadhavkar S, Singh G, Omary MB. Epidemiology of alcohol-related liver and pancreatic disease in the United States. Arch Intern Med. 2008 Mar 24;168(6):649-56. doi: 10.1001/archinte.168.6.649[↩]
- Apte MV, Wilson JS, McCaughan GW, Korsten MA, Haber PS, Norton ID, Pirola RC. Ethanol-induced alterations in messenger RNA levels correlate with glandular content of pancreatic enzymes. J Lab Clin Med. 1995 May;125(5):634-40.[↩]
- Toskes PP. Hyperlipidemic pancreatitis. Gastroenterol Clin North Am. 1990 Dec;19(4):783-91.[↩]
- Mithöfer K, Fernández-del Castillo C, Frick TW, Lewandrowski KB, Rattner DW, Warshaw AL. Acute hypercalcemia causes acute pancreatitis and ectopic trypsinogen activation in the rat. Gastroenterology. 1995 Jul;109(1):239-46. doi: 10.1016/0016-5085(95)90290-2[↩]
- Wilmink T, Frick TW. Drug-induced pancreatitis. Drug Saf. 1996 Jun;14(6):406-23. doi: 10.2165/00002018-199614060-00006[↩]
- Badalov N, Baradarian R, Iswara K, Li J, Steinberg W, Tenner S. Drug-induced acute pancreatitis: an evidence-based review. Clin Gastroenterol Hepatol. 2007 Jun;5(6):648-61; quiz 644. doi: 10.1016/j.cgh.2006.11.023[↩][↩]
- Sadr-Azodi O, Mattsson F, Bexlius TS, Lindblad M, Lagergren J, Ljung R. Association of oral glucocorticoid use with an increased risk of acute pancreatitis: a population-based nested case-control study. JAMA Intern Med. 2013 Mar 25;173(6):444-9. doi: 10.1001/jamainternmed.2013.2737[↩]
- Aliperti G. Complications related to diagnostic and therapeutic endoscopic retrograde cholangiopancreatography. Gastrointest Endosc Clin N Am. 1996 Apr;6(2):379-407.[↩]
- Neoptolemos JP, Raraty M, Finch M, Sutton R. Acute pancreatitis: the substantial human and financial costs. Gut. 1998 Jun;42(6):886-91. doi: 10.1136/gut.42.6.886[↩]
- Sekimoto M, Takada T, Kawarada Y, Hirata K, Mayumi T, Yoshida M, Hirota M, Kimura Y, Takeda K, Isaji S, Koizumi M, Otsuki M, Matsuno S; JPN. JPN Guidelines for the management of acute pancreatitis: epidemiology, etiology, natural history, and outcome predictors in acute pancreatitis. J Hepatobiliary Pancreat Surg. 2006;13(1):10-24. doi: 10.1007/s00534-005-1047-3[↩]
- Silen W. Acute pancreatitis. In: Silen W, Cope Z, editors. Cope’s Early Diagnosis of the Acute Abdomen. 18th ed. New York, NY: Oxford University Press; 1999. pp. 123–131.[↩]
- Mitchell RM, Byrne MF, Baillie J. Pancreatitis. Lancet. 2003 Apr 26;361(9367):1447-55. doi: 10.1016/s0140-6736(03)13139-x[↩]
- Swaroop VS, Chari ST, Clain JE. Severe acute pancreatitis. JAMA. 2004 Jun 16;291(23):2865-8. doi: 10.1001/jama.291.23.2865[↩]
- Dickson AP, Imrie CW. The incidence and prognosis of body wall ecchymosis in acute pancreatitis. Surg Gynecol Obstet. 1984 Oct;159(4):343-7.[↩]
- McARTHUR, K.E. (1996), Review article: Drug-induced pancreatitis. Alimentary Pharmacology & Therapeutics, 10: 23-38. https://doi.org/10.1111/j.1365-2036.1996.tb00174.x[↩]
- Parikh RP, Upadhyay KJ. Cullen’s sign for acute haemorrhagic pancreatitis. Indian J Med Res. 2013;137(6):1210. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3734730[↩]
- Chung KM, Chuang SS. Cullen and Grey Turner signs in idiopathic perirenal hemorrhage. CMAJ. 2011;183(16):E1221. doi:10.1503/cmaj.101548 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3216450[↩]
- Nadkarni NA, Khanna S, Vege SS. Splanchnic venous thrombosis and pancreatitis. Pancreas. 2013 Aug;42(6):924-31. doi: 10.1097/MPA.0b013e318287cd3d[↩]
- Morgan DE, Baron TH, Smith JK, Robbin ML, Kenney PJ. Pancreatic fluid collections prior to intervention: evaluation with MR imaging compared with CT and US. Radiology. 1997 Jun;203(3):773-8. doi: 10.1148/radiology.203.3.9169703[↩]
- Ranson JH, Turner JW, Roses DF, Rifkind KM, Spencer FC. Respiratory complications in acute pancreatitis. Ann Surg. 1974 May;179(5):557-66. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1356020/pdf/annsurg00243-0045.pdf[↩]
- Ranson JH, Rifkind KM, Roses DF, Fink SD, Eng K, Spencer FC. Prognostic signs and the role of operative management in acute pancreatitis. Surg Gynecol Obstet. 1974 Jul;139(1):69-81.[↩]
- Ducarme G, Maire F, Chatel P, Luton D, Hammel P. Acute pancreatitis during pregnancy: a review. J Perinatol. 2014 Feb;34(2):87-94. doi: 10.1038/jp.2013.161[↩]
- Papachristou GI, Muddana V, Yadav D, O’Connell M, Sanders MK, Slivka A, Whitcomb DC. Comparison of BISAP, Ranson’s, APACHE-II, and CTSI scores in predicting organ failure, complications, and mortality in acute pancreatitis. Am J Gastroenterol. 2010 Feb;105(2):435-41; quiz 442. doi: 10.1038/ajg.2009.622[↩][↩]
- Chandra S, Murali A, Bansal R, Agarwal D, Holm A. The Bedside Index for Severity in Acute Pancreatitis: a systematic review of prospective studies to determine predictive performance. J Community Hosp Intern Med Perspect. 2017 Sep 19;7(4):208-213. doi: 10.1080/20009666.2017.1361292[↩]
- Pérez Campos A, Bravo Paredes E, Prochazka Zarate R, Bussalleu A, Pinto Valdivia J, Valenzuela Granados V. BISAP-O y APACHE-O: utilidad en la predicción de severidad en la pancreatitis aguda según la clasificación modificada de Atlanta [BISAP-O y APACHE-O: utility in predicting severity in acute pancreatitis in modified Atlanta classification]. Rev Gastroenterol Peru. 2015 Jan;35(1):15-24. Spanish.[↩]
- Kothari S, Kalinowski M, Kobeszko M, Almouradi T. Computed tomography scan imaging in diagnosing acute uncomplicated pancreatitis: Usefulness vs cost. World J Gastroenterol. 2019 Mar 7;25(9):1080-1087. doi: 10.3748/wjg.v25.i9.1080[↩]
- Halgreen H, Pedersen NT, Worning H. Symptomatic effect of pancreatic enzyme therapy in patients with chronic pancreatitis. Scand J Gastroenterol. 1986 Jan;21(1):104-8. doi: 10.3109/00365528609034631[↩]
- Uomo G, Talamini G, Rabitti PG. Antioxidant treatment in hereditary pancreatitis. A pilot study on three young patients. Dig Liver Dis. 2001 Jan-Feb;33(1):58-62. doi: 10.1016/s1590-8658(01)80136-5[↩]
- Frøkjær JB, Olesen SS, Gram M, Yavarian Y, Bouwense SA, Wilder-Smith OH, Drewes AM. Altered brain microstructure assessed by diffusion tensor imaging in patients with chronic pancreatitis. Gut. 2011 Nov;60(11):1554-62. doi: 10.1136/gut.2010.236620[↩]
- Patel MR, Eppolito AL, Willingham FF. Hereditary pancreatitis for the endoscopist. Therap Adv Gastroenterol. 2013 Mar;6(2):169-79. doi: 10.1177/1756283X12467565[↩]
- Baron TH, DiMaio CJ, Wang AY, Morgan KA. American Gastroenterological Association clinical practice update: management of pancreatic necrosis. Gastroenterology. 2020;158(1):67–75.e1. doi: 10.1053/j.gastro.2019.07.064[↩]
- Hollemans RA, Bakker OJ, Boermeester MA, Bollen TL, Bosscha K, Bruno MJ, Buskens E, Dejong CH, van Duijvendijk P, van Eijck CH, Fockens P, van Goor H, van Grevenstein WM, van der Harst E, Heisterkamp J, Hesselink EJ, Hofker S, Houdijk AP, Karsten T, Kruyt PM, van Laarhoven CJ, Laméris JS, van Leeuwen MS, Manusama ER, Molenaar IQ, Nieuwenhuijs VB, van Ramshorst B, Roos D, Rosman C, Schaapherder AF, van der Schelling GP, Timmer R, Verdonk RC, de Wit RJ, Gooszen HG, Besselink MG, van Santvoort HC; Dutch Pancreatitis Study Group. Superiority of Step-up Approach vs Open Necrosectomy in Long-term Follow-up of Patients With Necrotizing Pancreatitis. Gastroenterology. 2019 Mar;156(4):1016-1026. doi: 10.1053/j.gastro.2018.10.045[↩]
- Saunders R, Neoptolemos JP, Hughes F, Ghaneh P, Halloran CM. Single port retroperitoneal pancreatic necrosectomy for the treatment of extra-pancreatic walled off necrotic collections. Ann Surg Open. 2021 Mar;2(1):e019. doi: 10.1097/AS9.0000000000000019[↩]
- Boxhoorn L, van Dijk SM, van Grinsven J, Verdonk RC, Boermeester MA, Bollen TL, et al. Immediate versus postponed intervention for infected necrotizing pancreatitis. N Engl J Med. 2021;385(15):1372–1381. doi: 10.1056/NEJMoa2100826[↩]
- Bang JY, Arnoletti JP, Holt BA, Sutton B, Hasan MK, Navaneethan U, et al. An endoscopic transluminal approach, compared with minimally invasive surgery, reduces complications and costs for patients with necrotizing pancreatitis. Gastroenterology. 2019;156(4):1027–40.e3. doi: 10.1053/j.gastro.2018.11.031[↩]
- Saunders R, Hughes FE, Evans JC, Smart HL, Ghaneh P, Ramesh J, Sutton R, Halloran CM. Cost analysis and outcomes of endoscopic, minimal access and open pancreatic necrosectomy. Ann Surg Open. 2021 Jun 1;2(2):e068. doi: 10.1097/AS9.0000000000000068[↩]
- Huang W, de la Iglesia-Garcia D, Baston-Rey I, Calvino-Suarez C, Larino-Noia J, Iglesias-Garcia J, et al. Exocrine pancreatic insufficiency following acute pancreatitis: systematic review and meta-analysis. Dig Dis Sci. 2019;64(7):1985–2005. doi: 10.1007/s10620-019-05568-9[↩][↩][↩]
- Keller J, Layer P. Human pancreatic exocrine response to nutrients in health and disease. Gut. 2005;54(Suppl 6):vi1–vi28. doi: 10.1136/gut.2005.065946[↩]
- Parniczky A, Lantos T, Toth EM, Szakacs Z, Godi S, Hagendorn R, et al. Antibiotic therapy in acute pancreatitis: from global overuse to evidence based recommendations. Pancreatology. 2019;19(4):488–499. doi: 10.1016/j.pan.2019.04.003[↩]
- Moran RA, Halloran C, Guo Q, Umapathy C, Jalaly NY, Jain S, et al. Early infection is an independent risk factor for increased mortality in patients with culture-confirmed infected pancreatic necrosis. Pancreatology. 2022;22(1):67–73. doi: 10.1016/j.pan.2021.11.003[↩]
- Fugazzola P, Ceresoli M, Coccolini F, Gabrielli F, Puzziello A, Monzani F, et al. The WSES/SICG/ACOI/SICUT/AcEMC/SIFIPAC guidelines for diagnosis and treatment of acute left colonic diverticulitis in the elderly. World J Emerg Surg. 2022;17(1):5. doi: 10.1186/s13017-022-00408-0[↩][↩]
- Singh RR, Mitchell W, David Y, Cheesman A, Dixon RE, Nagula S, et al. Pancreatic fungal infection in patients with necrotizing pancreatitis: a systematic review and meta-analysis. J Clin Gastroenterol. 2021;55(3):218–226. doi: 10.1097/MCG.0000000000001467[↩]
- Tu J, Yang Y, Zhang J, Yang Q, Lu G, Li B, et al. Effect of the disease severity on the risk of developing new-onset diabetes after acute pancreatitis. Medicine (Baltimore) 2018;97(22):e10713. doi: 10.1097/MD.0000000000010713[↩]
- Zhi M, Zhu X, Lugea A, Waldron RT, Pandol SJ, Li L. Incidence of new onset diabetes mellitus secondary to acute pancreatitis: a systematic review and meta-analysis. Front Physiol. 2019;10:637. doi: 10.3389/fphys.2019.00637[↩]
- Dovc K, Battelino T. Evolution of diabetes technology. Endocrinol Metab Clin N Am. 2020;49(1):1–18. doi: 10.1016/j.ecl.2019.10.009[↩]
- Lee HM, Deheragoda M, Harrison P, Devlin J, Sellars M, Hadzic N, et al. Autoimmune pancreatitis in children: a single centre experience in diagnosis, management and long term follow up. Pancreatology. 2019;19(1):169–176. doi: 10.1016/j.pan.2018.11.004[↩]
- Matsubayashi H, Ishiwatari H, Imai K, Kishida Y, Ito S, Hotta K, et al. Steroid therapy and steroid response in autoimmune pancreatitis. Int J Mol Sci. 2019;21(1):257. doi: 10.3390/ijms21010257[↩]
- Gapp J, Tariq A, Chandra S. Acute Pancreatitis. [Updated 2022 Nov 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482468[↩][↩][↩]
- Acute Pancreatitis. https://emedicine.medscape.com/article/181364-overview#a6[↩][↩][↩][↩][↩]
- Huh JH, Jeon H, Park SM, Choi E, Lee GS, Kim JW, Lee KJ. Diabetes Mellitus is Associated With Mortality in Acute Pancreatitis. J Clin Gastroenterol. 2018 Feb;52(2):178-183. doi: 10.1097/MCG.0000000000000783[↩]
- Jin T, Jiang K, Deng L, Guo J, Wu Y, Wang Z, Shi N, Zhang X, Lin Z, Asrani V, Jones P, Mittal A, Phillips A, Sutton R, Huang W, Yang X, Xia Q, Windsor JA. Response and outcome from fluid resuscitation in acute pancreatitis: a prospective cohort study. HPB (Oxford). 2018 Nov;20(11):1082-1091. doi: 10.1016/j.hpb.2018.05.018[↩]
- Amas Gómez L, Zubia Olaskoaga F. Results of the modification of an acute pancreatitis management protocol in Intensive Care medicine. Med Intensiva (Engl Ed). 2019 Dec;43(9):546-555. English, Spanish. doi: 10.1016/j.medin.2018.05.004[↩]
- Garret C, Péron M, Reignier J, Le Thuaut A, Lascarrou JB, Douane F, Lerhun M, Archambeaud I, Brulé N, Bretonnière C, Zambon O, Nicolet L, Regenet N, Guitton C, Coron E. Risk factors and outcomes of infected pancreatic necrosis: Retrospective cohort of 148 patients admitted to the ICU for acute pancreatitis. United European Gastroenterol J. 2018 Jul;6(6):910-918. doi: 10.1177/2050640618764049[↩]
- R Mofidi, M D Duff, S J Wigmore, K K Madhavan, O J Garden, R W Parks, Association between early systemic inflammatory response, severity of multiorgan dysfunction and death in acute pancreatitis, British Journal of Surgery, Volume 93, Issue 6, June 2006, Pages 738–744, https://doi.org/10.1002/bjs.5290[↩]
- A Buter, C W Imrie, C R Carter, S Evans, C J McKay, Dynamic nature of early organ dysfunction determines outcome in acute pancreatitis, British Journal of Surgery, Volume 89, Issue 3, March 2002, Pages 298–302, https://doi.org/10.1046/j.0007-1323.2001.02025.x[↩]
- Singh VK, Wu BU, Bollen TL, Repas K, Maurer R, Mortele KJ, Banks PA. Early systemic inflammatory response syndrome is associated with severe acute pancreatitis. Clin Gastroenterol Hepatol. 2009 Nov;7(11):1247-51. doi: 10.1016/j.cgh.2009.08.012[↩]
- Gao W, Yang HX, Ma CE. The Value of BISAP Score for Predicting Mortality and Severity in Acute Pancreatitis: A Systematic Review and Meta-Analysis. PLoS One. 2015 Jun 19;10(6):e0130412. doi: 10.1371/journal.pone.0130412. Erratum in: PLoS One. 2015;10(10):e0142025[↩]
- Suppiah A, Malde D, Arab T, Hamed M, Allgar V, Smith AM, Morris-Stiff G. The prognostic value of the neutrophil-lymphocyte ratio (NLR) in acute pancreatitis: identification of an optimal NLR. J Gastrointest Surg. 2013 Apr;17(4):675-81. doi: 10.1007/s11605-012-2121-1[↩][↩]
- Mortele KJ, Wiesner W, Intriere L, Shankar S, Zou KH, Kalantari BN, Perez A, vanSonnenberg E, Ros PR, Banks PA, Silverman SG. A modified CT severity index for evaluating acute pancreatitis: improved correlation with patient outcome. AJR Am J Roentgenol. 2004 Nov;183(5):1261-5. doi: 10.2214/ajr.183.5.1831261[↩][↩]
- Khan A, Sarma D, Gowda C, Rodrigues G. The Role of Modified Early Warning Score (MEWS) in the Prognosis of Acute Pancreatitis. Oman Med J. 2021 May 31;36(3):e272. doi: 10.5001/omj.2021.72[↩][↩][↩]
- Mikolasevic I, Orlic L, Poropat G, Jakopcic I, Stimac D, Klanac A, Carovic F, Milic S. Nonalcoholic fatty liver and the severity of acute pancreatitis. Eur J Intern Med. 2017 Mar;38:73-78. doi: 10.1016/j.ejim.2016.10.019[↩][↩]