Contents
- What are Genetically Modified Organisms (GMOs)?
- What is Genetically Modified Food?
- Can GMOs Be Used in Organic Products?
- Benefits from genetically GMO crops
- How different is food from GM crops as compared to food from non-GM crops?
- Genetically Modified Food Pros and Cons
- Are GM foods safe to eat?
- What is glyphosate?
- Is glyphosate safe?
- Table 1. Humans exposed to glyphosate Cancer Outcomes for Solid Tumors
- Table 2. Humans exposed to glyphosate Noncancer Outcomes
- Table 3. Toxicological profile of glyphosate metabolites found as residues in livestock and/or crops
- Animal Health Effects
- Glyphosate and cancer
- Glyphosate and death
- Glyphosate and respiratory effects
- Glyphosate and cardiovascular effects
- Glyphosate and gastrointestinal effects
- Glyphosate and hematological effects
- Glyphosate and liver effects
- Glyphosate and kidney effects
- Glyphosate and rheumatoid arthritis
- Glyphosate and skin effects
- Glyphosate and eye effects
- Glyphosate and endocrine effects
- Glyphosate and reproductive effects
- Glyphosate and neurological effects
- Glyphosate and developmental effects
- Genetically modified food and potential to cause allergy
- Genetically Modified Foods on Human Health
- Summary
What are Genetically Modified Organisms (GMOs)?
Genetically modified organisms (GMOs) are plants, animals, or microbes that have had their DNA changed using genetic engineering techniques 1. Genetic engineering allows scientists to select one specific gene to implant. This avoids introducing other genes with undesirable traits. Genetic engineering also helps speed up the process of creating new foods with desired traits. The process to create GMOs is different than selective breeding. This involves selecting plants or animals with desired traits and breeding them. Over time, this results in offspring with those desired traits. One of the problems with selective breeding is that it can also result in traits that are not desired. According to a new United Nations Department of Economic and Social Affairs report – The World Population Prospects: The 2017 Revision – published on 21 June 2017, the current world population of 7.6 billion is expected to reach 8.6 billion in 2030, 9.8 billion in 2050 and 11.2 billion in 2100 2. By the time someone born today in the US reaches voting age, there are likely to be another one billion people. Most of those people will live in cities. How will we produce enough nutritious food to support this burgeoning population and ensure access to food resources, particularly as climate change stresses the environment is an important question and how do we do that keeping an eye on safety, sustainability, affordability and equity?
Food insecurity is a significant issue in both developed and developing countries. In developed countries, food insecurity is often associated with inequality and inadequate social protections, leading to a reliance on food banks and food waste diversion efforts 3. On the other hand, developing countries face challenges such as low food production, postharvest losses, and acute hunger 4. Genetically modified food (GMF) has been a recent advancement of food-based technologies to address the growing concern for global food security and the challenge to feed 9 billion people of this planet by 2050 5.
Genes change every day by natural mutation and recombination, creating new biological variations 6. Humans have been exploiting this for centuries, shuffling genes in increasingly systematic ways and using extensive crossing and artificial selection to create many combinations that would never otherwise have occurred. Just about everything we eat is derived from livestock, crops, and micro-organisms bred specifically to provide food. Humans have also redistributed genes geographically: the soybean is native to Asia but is now grown throughout the Americas, and the potato, native to the American continent, is grown throughout the temperate world. DNA has never been “static,” neither naturally nor at the hand of people.
In 1946, scientists first discovered that DNA can be transferred between organisms 7. Genetic modification is an extension of this. Combining genes from different organisms is known as recombinant DNA technology and the resulting organism is said to be ‘Genetically modified (GM)’, ‘Genetically engineered’ or ‘Transgenic’ 8. Genetically modified organisms (GMOs) are plants, animals or microorganisms that have had their genetic material altered in the lab to create new varieties of plants, animals, and organisms with desired characteristics 9. GM products which are currently in the pipeline include medicines and vaccines, foods and food ingredients, feeds and fibres 8.
It is now known that there are several mechanisms for DNA transfer and that these occur in nature on a large scale, for example, it is a major mechanism for antibiotic resistance in pathogenic bacteria. However, unlike conventional breeding, in which new assortments of genes are created more or less at random, genetic modification allows specific genes to be identified, isolated, copied, and introduced into other organisms in much more direct and controlled ways. The principal transgenic crops grown commercially in field are herbicide and insecticide resistant soybeans, corn, cotton and canola. Other crops grown commercially and/or field-tested are sweet potato resistant to a virus that could destroy most of the African harvest, rice with increased iron and vitamins that may alleviate chronic malnutrition in Asian countries and a variety of plants that are able to survive weather extremes. There are bananas that produce human vaccines against infectious diseases such as hepatitis B, fish that mature more quickly, fruit and nut trees that yield years earlier and plants that produce new plastics with unique properties 8.
The possible benefits of genetic engineering include:
- A wide range of crops resistant to pests, diseases, and herbicides.
- More nutritious and tastier food.
- Increased supply of food with reduced cost and longer shelf life.
- Faster growing plants and animals.
- Food with more desirable traits, such as potatoes that produce less of a cancer-causing substance when fried.
- Food materials with improved shelf life and processing qualities (such as fruit much less susceptible to mold spoilage) and reduced or eliminated natural toxicants (such as glycoalkaloids in potatoes) or allergens (such as allergenic proteins in nuts).
- Better understanding of responses of crops to environmental stress and development of varieties that can grow in areas currently too inhospitable.
- Disease- and drought-resistant plants that require fewer environmental resources such as water, fertilizer and less use of pesticides.
- Production of high value drugs such as vaccines in high volume agricultural crops such as oilseed rape or livestock such as in milk of dairy cattle.
- Development of renewable and sustainable sources of new materials (such as plastics based on starch or vegetable oil) in designer agricultural crops such as oilseed rape, potato, and maize.
The most obvious difference from conventional breeding is that genetic modification (GM) allows us to transfer genes between species. For example, the gene for bovine chymosin has been transferred to industrial micro-organisms—Kluyveromyces lactis (a yeast), Aspergillus niger var awamori (a fungus), and Escherichia coli K12 (a bacterium). These microbes are grown in fermenters to produce chymosin (rennet) on a commercial scale; this rennet, which replaces the conventional form obtained from slaughtered animals, is now widely used in cheese production 10.
The first genetically modified (GM) plant was produced in 1983, using an antibiotic-resistant tobacco plant 8. China was the first country to commercialize a transgenic crop in the early 1990s with the introduction of virus resistant tobacco.
The potential advantages associated with GM crops include improvements in agricultural productivity, particularly through the development of crops resistant to insects and drought, thereby mitigating global hunger 11. Rodríguez et al 12 asserted that genetically modified food (GMF) offers significant economic, environmental, food security, and human health benefits. Thangarajan et al 13 suggested that genetically modified food (GMF) and crops may alleviate the challenge of insufficient food production to support the growing population. However, in recent decades, the discourse surrounding the concepts of genetically modified organisms (GMOs) and genetically modified foods (GMF) has become highly contentious 14.
Some people have expressed concerns about GM foods, such as:
- Creation of foods that can cause an allergic or toxic reaction
- Unexpected or harmful genetic changes
- Inadvertent transfer of genes from one GM plant or animal to another plant or animal not intended for genetic modification
- Foods that are less nutritious
What is Genetically Modified Food?
Genetically Modified Food (GMF) means any food containing or derived from a genetically engineered organism 15. The majority of the biotech-crops available on the global market have been genetically manipulated to express one of these basic traits: resistance to insects or viruses, tolerance to certain herbicides and nutritionally enhanced quality. There has been a 60-fold rise in the application of Agri-biotechnology since 1996, when the first biotech-crop was commercially produced 16. Advocates say that they have increased agricultural production by more than US$98 billion and saved an estimated 473 million kilograms of pesticides from being sprayed. But critics question their environmental, social and economic impacts 17. Major producers of GM crops include USA, Argentina, Canada, and China 18. In Canada Genetically Engineered (GE) ingredients are used in more than 70% of the processed food products 19. The current rate of biotech crop adoption is remarkably higher in developing versus industrialized countries (21% vs. 9%) 16. Developing countries are rapidly accepting the technology with the hope of alleviating hunger and poverty. These countries account for 40% of the global farmlands used for GM crop cultivation 16. It is predicted that, by 2015, more than 200 million hectares of lands will be planted by biotech crops in about 40 countries 16.
Most processed foods in the U.S. contain genetically engineered (GE) ingredients, since over 90 percent of the corn and 95 percent of the soybeans grown in this country in 2017 were genetically engineered 20. Even though you may not realize it, you are probably eating GMOs right now.
Genetically Modified Food (GMF) found in most of processed foods 21:
- Corn derived ingredients
- Oil, starch, corn syrup, alcohol
- Soybean derived ingredients
- Oil, soy flour, soy proteins, lecithin
- Canola oil
- Sugar from sugar beets
- Papaya, squash, sweet corn
Indirect GMO foods 21:
Animals fed GMO grain
- Meat – beef, chicken, pork
- Milk, yogurt, cheese, butter
- Eggs
Foods made with any of the above
- Prepared meals
- Baked goods
- Desserts
Timeline of GMO being introduced into US Foods 21
- 1992 – FDA policy
- 1994 – Tomato and squash
- 1996 – Corn, soybeans, canola
- 1998 – Papaya
- 2008 – Sugar beets
- 2011 – High-oleic soybeans
The United States Department of Agriculture (USDA) supports all methods of agriculture production, including organic, conventional, and biotechnology. Consumers purchase organic products expecting that they maintain their organic integrity from farm to market, and USDA is committed to meeting these expectations. No matter where it was grown, if a product has the USDA Organic label on it, it wasn’t produced with GMOs. To meet the USDA’s organic regulations, farmers and processors must show they aren’t using GMOs and that they are protecting their products from contact with prohibited substances from farm to table 22.
GMO crops on the US market in 2015 according to Whole Foods 23 :
- 94% of soybeans
- 93% of corn
- 96% of cotton
Cotton, corn, and soybeans are the main GMO crops grown in the United States. Most of these are used to make ingredients for other foods, such as:
- Corn syrup used as a sweetener in many foods and drinks
- Corn starch used in soups and sauces
- Soybean, corn, and canola oils used in snack foods, breads, salad dressings, and mayonnaise
- Sugar from sugar beets
- Livestock feed
Other GMO crops include one or more varieties of the following:
- Apples
- Alfalfa
- Canola
- Eggplant (not available in the United States)
- Papayas
- Pineapples
- Potatoes
- Salmon
- Squash
- Sugar beets
- Sugarcane
- Sweet corn
- Yellow squash
- Zucchini
GMO crops on the US market in 2017 according to the United States Department of Agriculture 24
Genetically engineered (GE) seed varieties were commercially introduced in 1996. Adoption rates for these crops increased rapidly in the years that followed. Currently, over 90 percent of U.S. corn, upland cotton, soybeans, canola, and sugarbeets are produced using genetically engineered varieties 25.
Note:
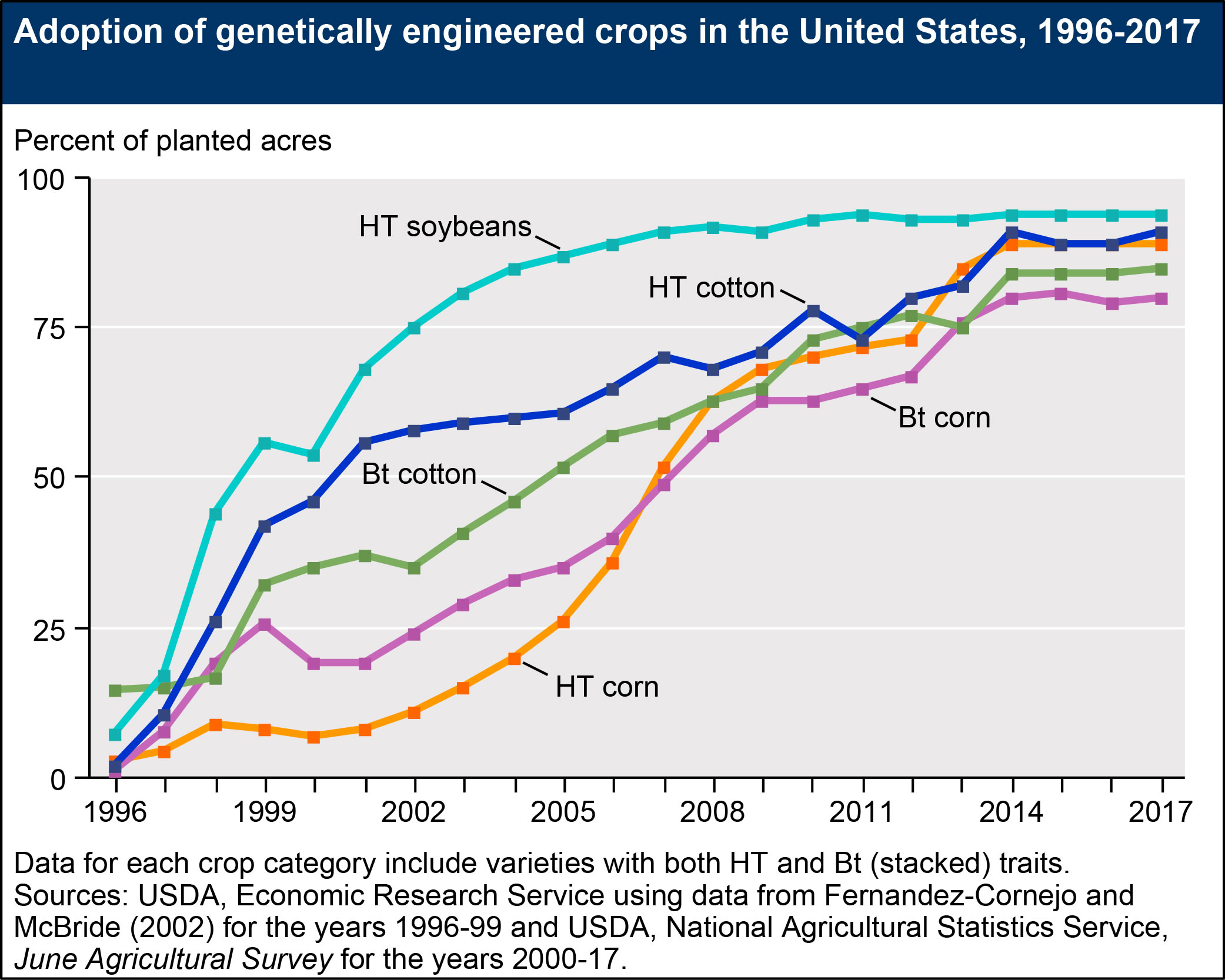
- Herbicide-tolerant (HT) crops, which tolerate potent herbicides (such as glyphosate or glufosinate), provide farmers with a broad variety of options for effective weed control. Based on USDA survey data, the percent of domestic soybean acres planted with HT seeds rose from 17 percent in 1997 to 68 percent in 2001, before plateauing at 94 percent in 2014. HT cotton acreage expanded from approximately 10 percent in 1997 to 56 percent in 2001, before reaching a high of 91 percent in 2014. Adoption rates for HT corn grew relatively slowly immediately following the commercialization of GE seeds. However, adoption rates increased following the turn of the century. Currently, approximately 89 percent of domestic corn acres are produced with HT seeds.
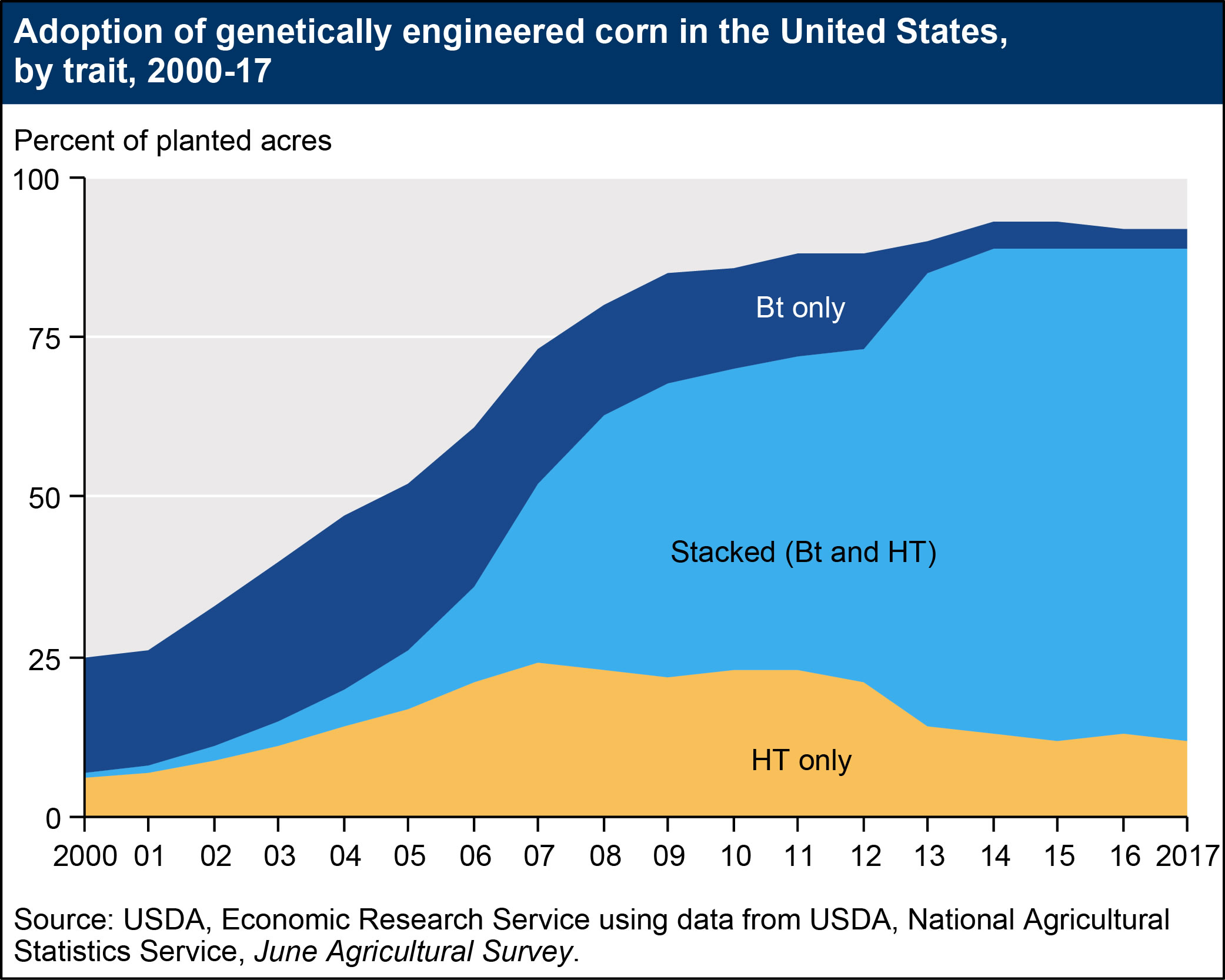
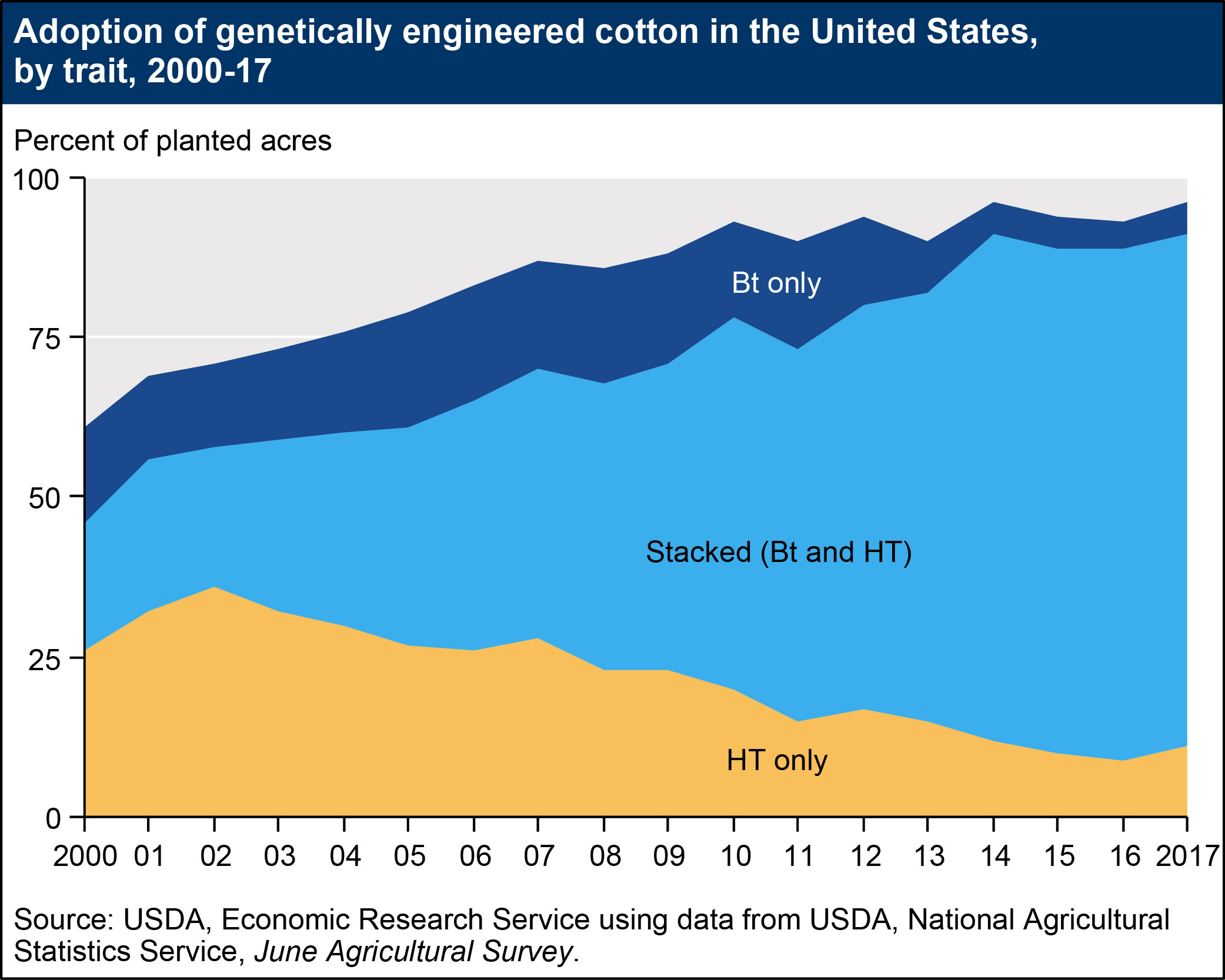
- Insect-resistant crops, which contain genes from the soil bacterium Bt (Bacillus thuringiensis) and produce insecticidal proteins, have been available for corn and cotton since 1996. Domestic Bt corn acreage grew from approximately 8 percent in 1997 to 19 percent in 2000, before climbing to 81 percent in 2015. Bt cotton acreage also expanded rapidly, from 15 percent of U.S. cotton acreage in 1997 to 37 percent in 2001. Currently, 85 percent of U.S. cotton acres are planted with genetically engineered, insect-resistant seeds. Increases in adoption rates for Bt corn may be due to the commercial introduction of new varieties resistant to the corn rootworm and the corn earworm (prior to 2003, Bt corn varieties only targeted the European corn borer). Adoption rates for Bt corn may fluctuate over time, depending on the severity of European corn borer and corn rootworm infestations. Similarly, adoption rates for Bt cotton may depend on the severity of tobacco budworm, bollworm, and pink bollworm infestations. As of 2017, insect-resistant varieties have not been commercialized for soybeans.
The figures below illustrate increases in adoption rates for “stacked” varieties, which have both (in some cases, multiple) HT and Bt traits. Adoption of stacked varieties has accelerated in recent years. Approximately 80 percent of cotton acres and 77 percent of corn acres were planted with stacked seeds in 2017.
[Source 24 ] [Source 24 ]Plants with favourable characteristics have been produced for thousands of years by conventional breeding methods. Desirable traits are selected, combined and propagated by repeated sexual crossings over numerous generations. Farmers have long sought to breed better plants. Traditionally this has involved repeatedly cross-pollinating plants to create an offspring with the desired combination of traits. This process introduces a number of genes into the plant, including some that may have unwanted traits. This is a long process, taking up to 15 years to produce new varieties.
The purpose of genetically altering, also called genetically engineering (GE), plants is to improve their durability, such as through drought tolerance or disease resistance, or their nutritional value. These changes are aimed at increasing yield and lowering costs. GMOs already play a role in feeding extra mouths, and if we let it, that role may grow. Yet they are also still a source of controversy, and there are both valid concerns and misconceptions.
In the developing world, 840 million people are chronically undernourished, surviving on fewer than 8000 kJ/day (2000 Kcal/day). Approximately 1.3 billion people are living on less than US$1/day and do not have secure access to food. Many of these are also rural farmers in developing countries, depending entirely on small-scale agriculture for their own subsistence and to make their living. They generally cannot afford to irrigate their crops or purchase herbicides or pesticides, leading to a vicious circle of poor crop growth, falling yields and pest susceptibility. At the same time, the Intergovernmental Panel on Climate Change predicts that changing weather patterns will almost certainly have a negative impact on crop yields.
In addition, earth’s population has more than doubled since 1960 and the world’s population is predicted to double again over the next 40 years, the UN estimates it will reach 9.8 billion by 2050, with over 95% of individuals being born in developing countries.
It is estimated that to meet these increased demands, crop demand is predicted to increase by 100 to 110 percent of 2005 levels over the same period with food production must increase by at least 40% in the face of decreasing fertile lands and water resources.
GM plant technologies are one of a number of different approaches that are being developed to combat these problems. Specifically, studies are under way to genetically modify plants to increase crop yields, or to directly improve nutritional content.
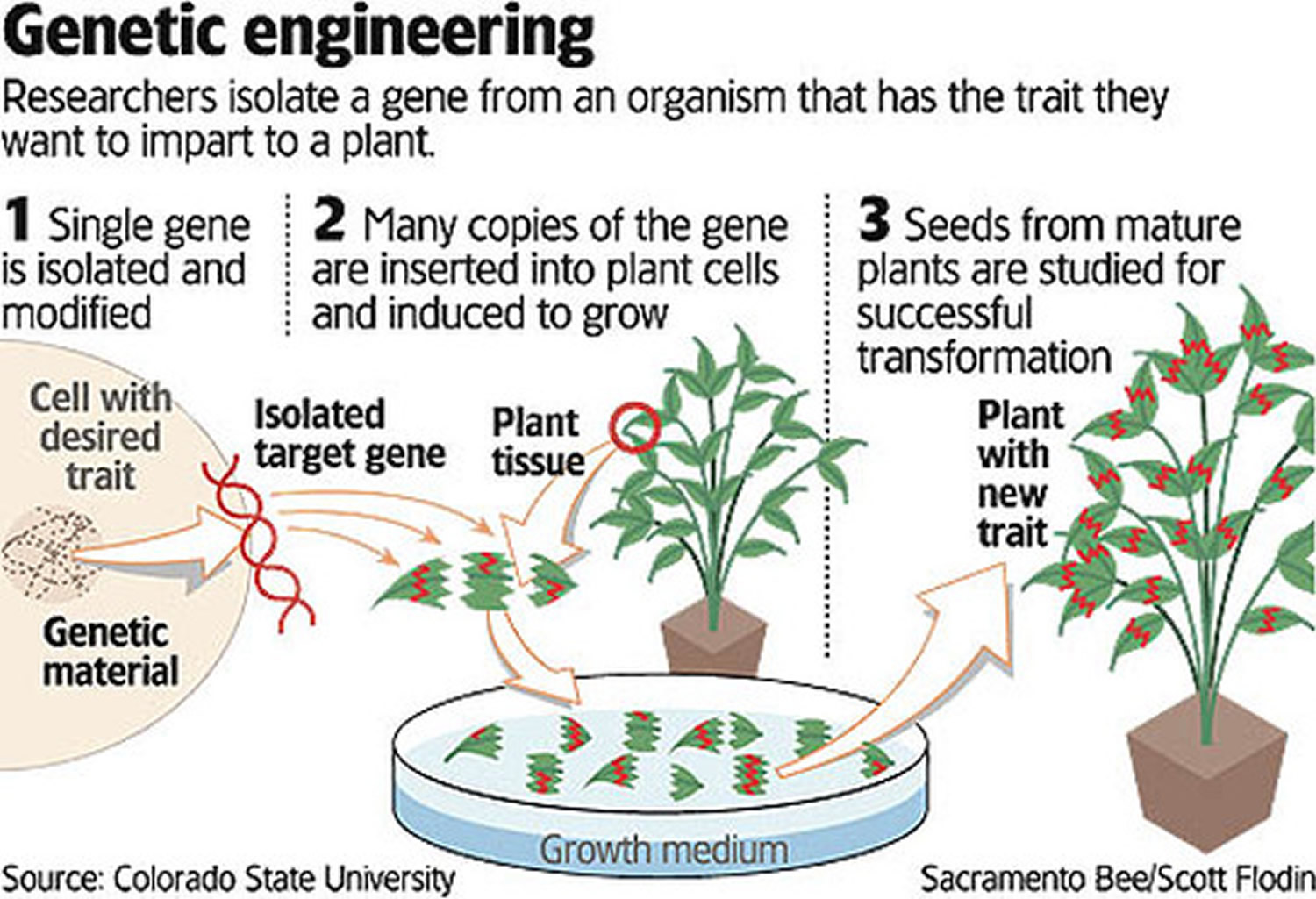
Genetic engineering isolates the gene for the desired trait, adds it to a single plant cell in a lab, and generates a new plant from that cell. By introducing only one gene, scientists hope to avoid transferring unwanted traits from other genes.
Furthermore, genetic engineering not only allows this process to be dramatically accelerated in a highly targeted manner by introducing a small number of genes, it can also overcome the barrier of sexual incompatibility between plant species and vastly increase the size of the available gene pool.
Mixing plant species is how we’ve gotten papayas free of viruses, corn plants that survive drought, soybeans that stand up to weed killer (e.g. Roundup), potatoes that don’t bruise, and crops that yield more and cost less. That’s good news for our food supply and the business of farming.
Now the emergence of the gene-editing tool CRISPR/Cas9 is making it possible to precisely edit the native DNA of organisms with the potential to dramatically increase crop yields. Unlike previous approaches to GM crops that introduce foreign DNA into an organism, genome editing achieves much the same outcome as selective breeding—but in a much faster and more selective way and without having to rely on natural genetic variation.
Fruits and vegetables
Papaya has been developed by genetic engineering which is ring spot virus resistant and thus enhancing the productivity. This was very much in need as in the early 1990s the Hawaii’s papaya industry was facing disaster because of the deadly papaya ring spot virus. Its single-handed savior was a breed engineered to be resistant to the virus. Without it, the state’s papaya industry would have collapsed. Today 80 % of Hawaiian papaya is genetically engineered, and till now no conventional or organic method is available to control ring spot virus 8.
The NewLeaf™ potato, a GM food developed using naturally-occurring bacteria found in the soil known as Bacillus thuringiensis (Bt), was made to provide in-plant protection from the yield-robbing Colorado potato beetle 8. This was brought to market by Monsanto in the late 1990s, developed for the fast food market. This was forced to withdraw from the market in 2001as the fast food retailers did not pick it up and thereby the food processors ran into export problems. Reports say that currently no transgenic potatoes are marketed for the purpose of human consumption. However, BASF, one of the leading suppliers of plant biotechnology solutions for agriculture requested for the approval for cultivation and marketing as a food and feed for its ‘Fortuna potato’. This GM potato was made resistant to late blight by adding two resistance genes, blb1 and blb2, which was originated from the Mexican wild potato Solanum bulbocastanum. As of 2005, about 13 % of the zucchini grown in the USA is genetically modified to resist three viruses; the zucchini is also grown in Canada 26.
Vegetable oil
Cooking oil, margarine and shortening may also be made from several crops. A large percentage of Canola produced in USA is GM and is mainly used to produce vegetable oil. Canola oil is the third most widely consumed vegetable oil in the world. The genetic modifications are made for providing resistance to herbicides viz. glyphosate or glufosinate and also for improving the oil composition. After removing oil from canola seed, which is ∼43 %, the meal has been used as high quality animal feed. Canola oil is a key ingredient in many foods and is sold directly to consumers as margarine or cooking oil. The oil has many non-food uses, which includes making lipsticks.
Maize, also called corn in the USA and cornmeal, which is ground and dried maize constitute a staple food in many regions of the world. Grown since 1997 in the USA and Canada, 86 % of the USA maize crop was genetically modified in 2010 27 and 32 % of the worldwide maize crop was GM in 2011 7. A good amount of the total maize harvested go for livestock feed including the distillers grains. The remaining has been used for ethanol and high fructose corn syrup production, export, and also used for other sweeteners, cornstarch, alcohol, human food or drink. Corn oil is sold directly as cooking oil and to make shortening and margarine, in addition to make vitamin carriers, as a source of lecithin, as an ingredient in prepared foods like mayonnaise, sauces and soups, and also to fry potato chips and French fries. Cottonseed oil is used as a salad and cooking oil, both domestically and industrially. Nearly 93 % of the cotton crop in USA is GM.
Sugar
The USA imports 10 % of its sugar from other countries, while the remaining 90 % is extracted from domestically grown sugar beet and sugarcane. Out of the domestically grown sugar crops, half of the extracted sugar is derived from sugar beet, and the other half is from sugarcane. After deregulation in 2005, glyphosate-resistant sugar beet was extensively adopted in the USA. In USA 95 % of sugar beet acres were planted with glyphosate-resistant seed 7. Sugar beets that are herbicide-tolerant have been approved in Australia, Canada, Colombia, EU, Japan, Korea, Mexico, New Zealand, Philippines, Russian Federation, Singapore and USA. The food products of sugar beets are refined sugar and molasses. Pulp remaining from the refining process is used as animal feed. The sugar produced from GM sugar beets is highly refined and contains no DNA or protein—it is just sucrose, the same as sugar produced from non-GM sugar beets 28.
Can GMOs Be Used in Organic Products?
The use of genetic engineering, or genetically modified organisms (GMOs), is prohibited in organic products. This means an organic farmer can’t plant GMO seeds, an organic cow can’t eat GMO alfalfa or corn, and an organic soup producer can’t use any GMO ingredients 29.
USDA (United States Department of Agriculture) supports all methods of agriculture production, including organic, conventional, and biotechnology. Consumers purchase organic products expecting that they maintain their organic integrity from farm to market, and USDA is committed to meeting these expectations. No matter where it was grown, if a product has the USDA Organic label on it, it wasn’t produced with GMOs. To meet the USDA’s organic regulations, farmers and processors must show they aren’t using GMOs and that they are protecting their products from contact with prohibited substances from farm to table 22.
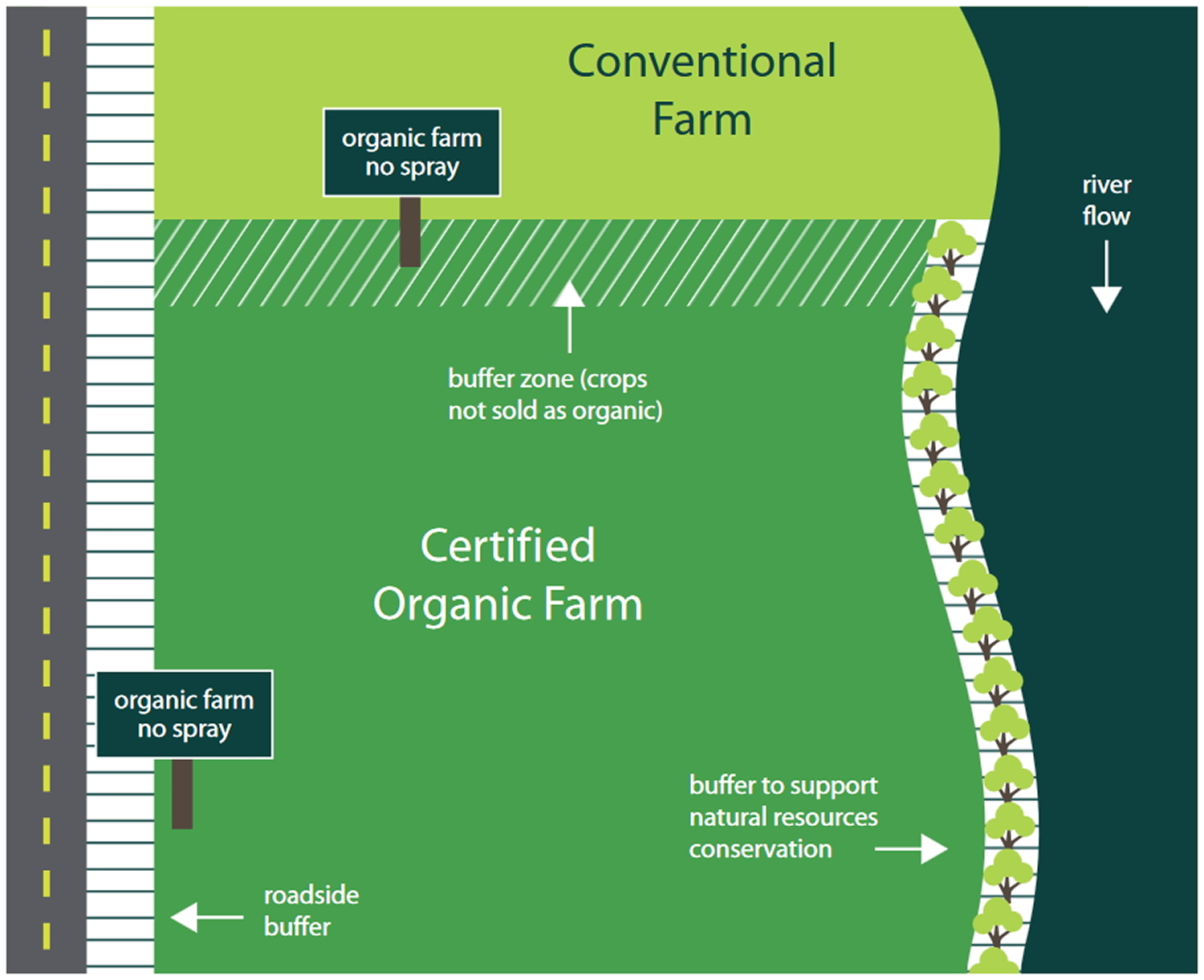
In the sketch below 22, the organic farmer has set up several buffer zones to protect the integrity of her organic crops from GMOs. Where her farm borders the conventional farm, she has set aside an area which she will farm organically (for example, she won’t apply prohibited pesticides), but she won’t sell that land’s crops as organic. She has also posted “no spray” signs on the borders of her property and has another buffer zone on the left side to protect her farm from unintended substances from the local road. A final buffer zone on the right side of her property includes a row of trees to reduce erosion and protect runoff into the bordering river.
Figure 1. Farm Layout between Certified Organic Farm and Conventional Farm per United States Department of Agriculture
[Source 22 ]Benefits from genetically GMO crops
GMO crops can confer social and environmental benefits 30:
- Herbicide resistant crops need less tillage, which enhances the content of organic matter in the soil, reduces erosion by wind and water and improves soil structure. Less herbicide is used resulting in less runoff to watercourses.
- Plants producing toxins selective against caterpillar larvae reduce the number of sprays of persistent chemicals by up to 60%. This spares useful insects and reduces the pollution of soil and waterways with chemicals.
- GMO crops can be made that produce vitamins lacking in some staple crops e.g. genes for vitamin A production introduced into rice 31.
- Research is in progress to increase salt or drought tolerance – an important consideration as the world population rises and, currently, arable land is at a premium.
How different is food from GM crops as compared to food from non-GM crops?
Changes to genes aren’t necessarily a bad thing. They happen in nature. In fact, no matter what’s on the menu, it isn’t exactly the same as what grew hundreds or thousands of years ago.
Bits of DNA, called genes, are responsible for all sorts of characteristics and traits in every living thing, from height to how certain cells work. Useful traits (genes) help the plants and animals with those genes survive or thrive better than ones without them, so they get passed along and eventually become common.
Humans have been “genetically modifying” plants and animals for thousands of years. Five hundred years ago, say a farmer noticed some corn was a little sweeter. To replicate that flavor, the farmer might select those seeds for the next crop. That new trait came about by random genetic mutation, and establishing a noticeably sweeter flavor using selective breeding would take years, if not decades.
Our ancestors sped up the process when they saved seeds of cream-of-the-crop plants to grow the next time, and the next, and the next. That’s what turned small bunches of tiny kernels on tall grass 10,000 years ago into the big ears of juicy corn on the cob we have today. With animals, picks of the litter were paired to breed “new-and-improved” babies.
Genetic engineering does much the same thing, discovering and introducing genes that yield desired traits, but in a faster and more accurate way than selective breeding.
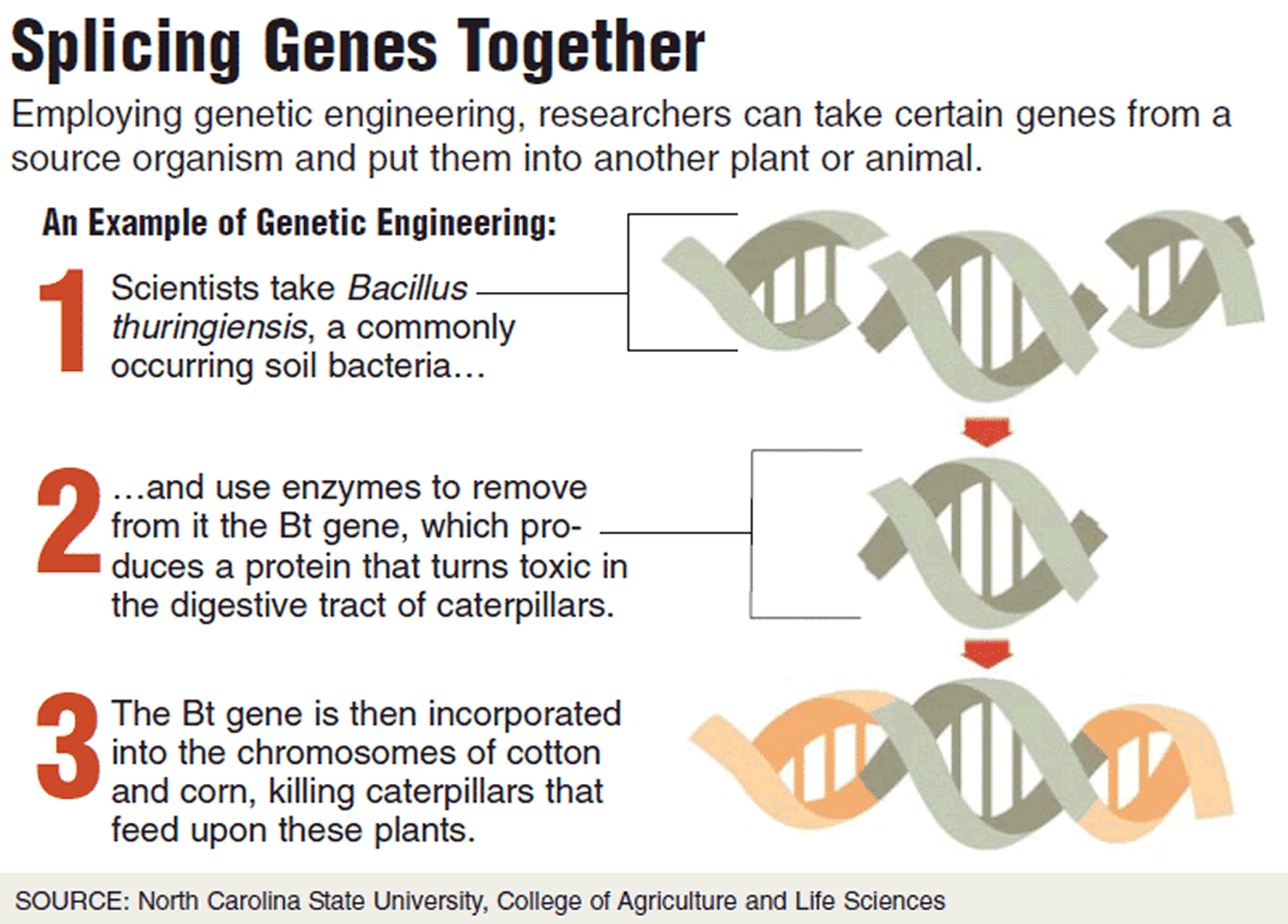
Some GM foods, like BT (Bacillus thuringiensis) crops, are engineered to contain a form of pesticide, which means they don’t need to be sprayed with chemical pesticides. Bacillus thuringiensis (Bt) is a common bacteria that has played a very uncommon role in agriculture and the development of genetically modified foods. The natural insecticidal abilities of these bacteria have made them an important pest control tool for nearly a century. While their use as a natural bio-pesticide is widely accepted and approved for organic applications, the engineering of Bt genes into major crops has been more controversial. Eating food that produces a pesticide sounds scary, but as the video above notes, pesticide doesn’t always mean it is inedible or harmful to humans. Many substances harm insects or animals, but not humans—coffee is one example.
And there are examples of pesticide-resistant GMOs having a tangible positive impact on people. When eggplant farmers in Bangladesh began to get sick from using too much chemical pesticide, for example, they implemented BT and were able to reduce pesticide use by 80 percent.
Some GMOs are specially made to be packed with extra vitamins, minerals, and other health benefits. For example, Swiss researchers created a strain of “golden” rice with a lot of beta-carotene, an antioxidant good for your eyes and skin. Soybeans whose fats have been changed so they’re more like olive oil can be turned into a heart-healthy replacement for oils with trans fats that’s more heat-tolerant and better for cooking. And those bruise-free potatoes are supposed to cut down on cancer-causing chemicals created when spuds become french fries.
Much of the backlash against GMOs is less about genetic engineering and more about the business practices of the corporations that control our food supply. GMO crops have been a money-maker for herbicide companies and as crops have been modified to be herbicide-resistant, herbicide use increases. For companies making GMO seeds and associated herbicides, that’s a lot of power over something as critical as how we feed ourselves.
As the population grows, it’s going to get harder to feed everyone. The Food and Agriculture Organization of the United Nations (FAO) estimates food production will need to double in some parts of the world by 2050. GMOs are one way to make enough nutritious food available with limited land, water, and other resources.
And perhaps we need to be particularly careful when it comes to genetically modified anything, to thoroughly vet it for harm to humans and ecosystems. Once the genie’s out of the bottle, many worry we might not be able to get it back in again.
Genetically Modified Food Pros and Cons
Currently with over 20 years of exposure to genetically modified foods, there is no substantiated allergenicity (that causes an allergy) in humans to genetically modified food or food ingredient 21.
Livestock data 21:
- 100 billion animals fed GMO grain between 2000-2011
- No difference in health outcomes comparing before after GMO introduction
- No difference in nutrient or food composition
Safety studies in animals 21:
- Native plant DNA-fragments may be absorbed with very low frequency
- Assume same for recombinant DNA
- No transfer of recombinant DNA from feed to animal tissues (eggs, meat or milk)
Gut microbiota 21:
- Horizontal gene flow
- DNA rapidly degraded by digestion
- Limited amount may escape to colon
- Native and recombinant DNA similar
- Transformation of gut microbiota highly unlikely
According to this study paper 32 that focuses on (1) the potential of transgenes (genetic engineering) to alter genes stability and (2) the potential risks to food and feed safety associated with genome instability (refers to a high frequency of mutations within the genome of a cellular lineage. These mutations can include changes in nucleic acid sequences, chromosomal rearrangements or aneuploidy). The authors found neither changes in gene expression nor mutations in amino acid sequences are likely to alter the safety of a protein or lead to the production of novel metabolites. Thus far, there is no evidence that a random genomic change in a crop has ever resulted in a novel safety issue, even when new alleles or genes were created. Therefore, other than changes due to the transgene products, the risks of introducing new food hazards are no different from the risks associated with traditional breeding 32.
The weight of the evidence leads to the conclusion that enhanced genetic instability from a transgene or from common sequences in two or more transgenes is unlikely 32. Even then, the probability that any genetic instability will lead to an altered protein or metabolic product that creates a biosafety issue is exceedingly small; the production of a GE stack does not measurably increase this probability. There is no readily identifiable biological reason why genomic changes occurring in the breeding of a GE stack would be different in nature, scale, or frequency from those taking place in non-genetically engineered crops or in genetically engineered crops with a single event.
The likelihood that any one mutation would create a biosafety issue is improbably small and would occur in a single plant in a field containing hundreds to millions of other plants. Thus, any negative consequences from that one mutation would be limited to seeds produced by that one plant, with dilution upon harvest minimizing the likelihood of any deleterious effects resulting from consumption. This large dilution factor helps explain why such changes, which may in principle lead to a negative effect, remain undetected and why breeding is generally considered a safe process 32.
Food applications for GM plants
Genetically modified crops have the potential to eliminate hunger and starvation in millions of people, especially in developing countries because the genetic modification can produce large amounts of foods that are more nutritious. Large quantities are produced because genetically modified crops are more resistant to pests and drought. They also contain greater amounts of nutrients, such as proteins and vitamins 33.
However, there are concerns about the safety of genetically modified crops. The concerns are that they may contain allergenic substances due to introduction of new genes into crops. Another concern is that genetic engineering often involves the use of antibiotic-resistance genes as “selectable markers” and this could lead to production of antibiotic-resistant bacterial strains that are resistant to available antibiotics. This would create a serious public health problem. The genetically modified crops might contain other toxic substances (such as enhanced amounts of heavy metals) and the crops might not be “substantially equivalent” in genome, proteome, and metabolome compared with unmodified crops. Another concern is that genetically modified crops may be less nutritious; for example, they might contain lower amounts of phytoestrogens, which protect against heart disease and cancer. The review of available literature 33 indicates that the genetically modified crops available in the market that are intended for human consumption are generally safe; their consumption is not associated with serious health problems.
To many, an organic diet has never been more in style than it is right now, with millions of consumers willing to shell out extra dollars for organic foods. Most of us have a vague idea that organic is better because it’s more natural and free of genetically modified organisms (GMOs) and pesticides. But have shoppers ever stop to wonder whether organic foods have as much of an impact on their health as they do on my wallet ?
Health experts and consumers have long debated whether organic foods are more nutritious and safer than conventional foods.
A study released in the Annals of Internal Medicine finds that organics do have some safety advantages over conventional foods, nutritionally speaking they have little extra to offer. The Conclusion: “The published literature lacks strong evidence that organic foods are significantly more nutritious than conventional foods. Consumption of organic foods may reduce exposure to pesticide residues and antibiotic-resistant bacteria.”
But what does “natural” even mean ? The line is harder to draw than we may think.
Genetically Modified Foods Role Increasing Food Production
In the developing world, 840 million people are chronically undernourished, surviving on fewer than 8000 kJ/day (2000 Kcal/day) 34, 35. Approximately 1.3 billion people are living on less than US$1/day 36, 36 and do not have secure access to food. Many of these are also rural farmers in developing countries, depending entirely on small-scale agriculture for their own subsistence and to make their living 37. They generally cannot afford to irrigate their crops or purchase herbicides or pesticides, leading to a vicious circle of poor crop growth, falling yields and pest susceptibility 37. In addition, the world’s population is predicted to double over the next 40 years, with over 95% of individuals being born in developing countries 38. It is estimated that to meet these increased demands, food production must increase by at least 40% in the face of decreasing fertile lands and water resources 39, 40. GM plant technologies are one of a number of different approaches that are being developed to combat these problems. Specifically, studies are under way to genetically modify plants to increase crop yields, or to directly improve nutritional content.
Crop yields worldwide are significantly reduced by the action of pathogens, parasites and herbivorous insects 41. Two examples of commercial GM crop growth in this area are the insect-resistant crops expressing the bt gene (from the bacterium Bacillus thuringiensis) and virus-resistant GM papaya 42. The first of these has been particularly successful; in the USA, for example, insect resistant GM maize is grown over an area of 10.6 million hectares and comprises 35% of all maize (GM and non-GM) grown in the country 43. At the laboratory level, resistance has also been engineered to bacterial and fungal plant pathogens.
A primary cause of plant loss worldwide is abiotic stress, particularly salinity, drought, and temperature extremes 44. In the future, these losses will increase as water resources decline and desertification intensifies. Drought and salinity are expected to cause serious salinization of all arable lands by 2050 45, requiring the implementation of new technologies to ensure crop survival. Although a number of promising targets have been identified in the production of abiotic stress tolerant GM plants, research remains at the laboratory-based level. An example is the study by Shou et al. 46 demonstrating that expression of an enzyme in GM maize activates an oxidative signal cascade that confers cold, heat and salinity tolerance.
Genetically Modified Foods Role in Increasing Nutritional Content
In the developed world the nutritional content of food items is not of major concern, as individuals have access to a wide variety of foods that will meet all of their nutritional needs. In the developing world, however, this is often not the case, with people often relying on a single staple food crop for their energy intake 47. GM technology offers a way to alleviate some of these problems by engineering plants to express additional products that can combat malnutrition. An important example of the potential of this technology is the ‘Golden Rice Project’. Vitamin A deficiency is widespread in the developing world and is estimated to account for the deaths of approximately 2 million children per year 47. In surviving children it has been identified as the leading cause of blindness 48. Humans can synthesize vitamin A from its precursor β-carotene, which is commonly found in many plants but not in cereal grains. The strategy of the Golden Rice Project was to introduce the correct metabolic steps into rice endosperm to allow β-carotene synthesis. In 2000, Ye et al. 49 engineered rice that contained moderate levels of β-carotene and since then researchers have produced the much higher yielding ‘Golden Rice 2’ 50. It is estimated that 72 g of dry Golden Rice 2 will provide 50% of the RDA of vitamin A for a 1–3-year-old child 50.
Golden Rice was developed for farmers in the poorest countries, and from the beginning, the aim of the scientists was to provide the technology free of charge, which required the negotiation of more than 100 intellectual and technical property licenses 51. Golden Rice will be given to subsistence farmers with no additional conditions and is an impressive example of a health solution that can be offered by plant biotechnology.
Are GM foods safe to eat?
GM crops are tightly regulated by several government bodies. The European Food Safety Authority and each individual member state have detailed the requirements for a full risk assessment of GM plants and derived food and feed 52. In the USA, the Food and Drug Agency, the Environmental Protection Agency and the US Department of Agriculture, Animal and Plant Health Inspection Service are all involved in the regulatory process for GM crop approval 53. Consequently, GM plants undergo extensive safety testing prior to commercialization (for an example see https://www.efsa.europa.eu/en/topics/genetically-modified-organisms).
Foods derived from GM crops have been consumed by hundreds of millions of people across the world for more than 15 years, with no reported ill effects (or legal cases related to human health), despite many of the consumers coming from that most litigious of countries, the USA.
There is little documented evidence that GM crops are potentially toxic.
This question should be better re-worded to not about the safety of eating the genetically modified foods, instead it should be – Is it safe to eat GMOs that have some herbicide and pesticide residue, even though based on theoretical considerations and in general, pesticide (specifically glyphosate) and herbicide levels in both organic and non-organic foods were within allowable safety limits.
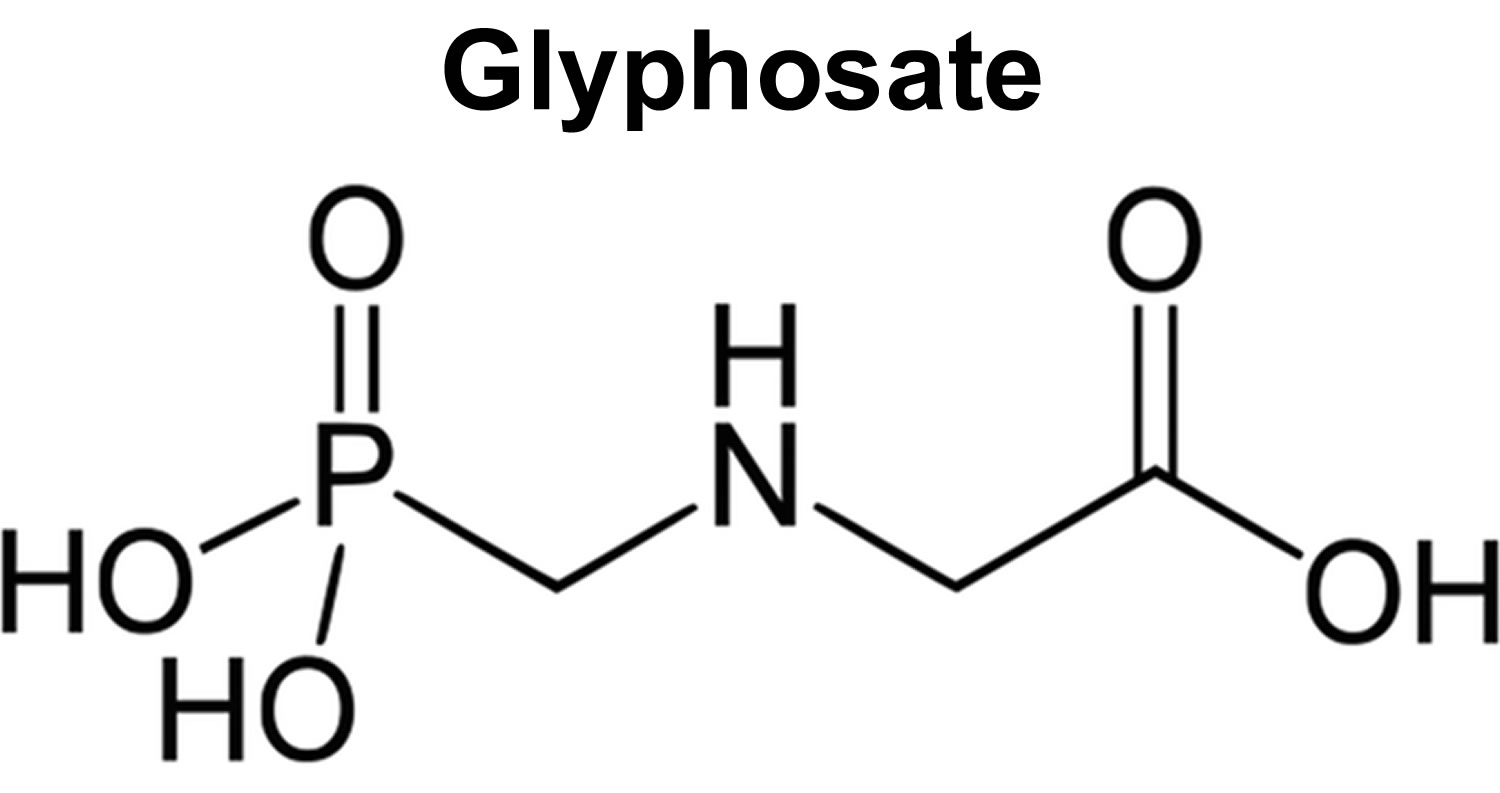
What is glyphosate?
Glyphosate also known as N-phosphonomethyl glycine is a widely used non-selective herbicide that kills weeds, grasses and most plants by blocking a plant enzyme called 5-enolpyruvyl shikimate-3 phosphate (EPSP) synthase disturbing the shikimate pathway that’s essential for plant growth 54, 55, 56, 57, 58, 59. The 5-enolpyruvyl shikimate-3 phosphate (EPSP) synthase enzyme is not found in humans or animals 60. Glyphosate molecule was first synthesized by Henri Martin from Cilag, a small Swiss pharmaceutical company, but was only tested as a herbicide in 1970 by John E. Franz of Monsanto Co. 61. In 1974, after being patented for herbicide use, glyphosate (as an isopropylamine salt of glyphosate) reached the market as a non-selective herbicide 62 and has been used for total control of vegetation since it is a broad-spectrum, non-selective systemic herbicide 63.
Glyphosate comes in many forms, including an acid and several salts. These can be either solids or an amber-colored liquid. There are over 750 products containing glyphosate for sale in the United States. Glyphosate is often used in agriculture, forestry, lawn and garden care to control broad-leafed plants, weeds and grasses that compete with crops 64, 65. Glyphosate can be applied to plants by spraying using different spray equipment, depending on the situation. People can be exposed to glyphosate by getting it on their skin, in their eyes, or breathing it in while using it. People can also swallow it if they eat or smoke after applying it without washing their hands. In humans, glyphosate does not easily pass through the skin. In addition, certain trace amounts of glyphosate chemical residues may remain in or on some crops after they’re harvested. Glyphosate that is absorbed or ingested will pass through the body relatively quickly. The vast majority of glyphosate leaves the body in urine and feces without being changed into another chemical.
When glyphosate is applied to a plant it spreads to all parts of the plant. Glyphosate stops the plant’s cells from making some of the amino acids needed for plant growth. Humans and animals make these amino acids using a different process than plants.
Glyphosate has been detected in the air during spraying, in water, and in food. The general population is exposed primarily through residence near sprayed areas, home use, and diet, and the level that has been observed is generally low.
Once in soil, glyphosate may be adsorbed onto soil particles, degraded by microbes, or transferred to deeper soil horizons, migrating via soil pores or root canals. However, some agricultural practices, such as phosphorous amendment, may re-solubilize glyphosate in soils, making it available for leaching 66 and to the rhizosphere (the zone of soil surrounding a plant root where the biology and chemistry of the soil are influenced by the root) of non-target plants.
Recently, a number of studies have focused on the environmental presence of the major glyphosate metabolite aminomethylphosphonic acid (AMPA). Glyphosate and its metabolite, aminomethylphosphonic acid (AMPA), have been detected in water 67. The half-life of glyphosate in water can range from days to months, complicating efforts to fully understand its environmental persistence 68. Glyphosate is quickly degraded to aminomethylphosphonic acid (AMPA) in soils by microorganisms 61, 69. A similar mechanism of glyphosate degradation has been proposed in plants 70; therefore the co-occurrence of glyphosate and AMPA is expected in plant tissues due to glyphosate degradation and/or AMPA uptake from environmental matrices. Upon penetrating the plant tissues, glyphosate will reach active metabolic sites, such as root and shoot meristems, after being translocated through vascular tissues 71, following the same pathway as photoassimilates 72. Similarly, aminomethylphosphonic acid (AMPA) can also be translocated to diverse plant tissues 70. Therefore, plant organs such as nodules, root tips, and shoot apices, which show high rates of metabolism and growth, represent important sinks for glyphosate and aminomethylphosphonic acid (AMPA) 73, 74.
Figure 2. Glyphosate
[Source 75 ]What happens to glyphosate in the environment?
Glyphosate binds tightly to soil 64. It can persist in soil for up to 6 months depending on the climate and the type of soil it is in 64. Glyphosate is broken down by bacteria in the soil.
Glyphosate is not likely to get into groundwater because it binds tightly to soil. In one study, half the glyphosate in dead leaves broke down in 8 or 9 days. Another study found that some glyphosate was taken up by carrots and lettuce after the soil was treated with it.
Pure glyphosate is low in toxicity to fish and wildlife, but some products containing glyphosate may be toxic because of the other ingredients in them. Glyphosate may affect fish and wildlife indirectly because killing the plants alters the animals’ habitat.
Glyphosate mechanism of action
The herbicidal effects of glyphosate are due to the inhibition of the 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS), an enzyme from the shikimate pathway, which leads to prevention of the biosynthesis of the amino acids phenylalanine, tyrosine, and tryptophan 76. The shikimic acid pathway is necessary for plants and some microorganisms.
Glyphosate is usually applied to plants by spraying. Glyphosate is then absorbed through plant leaves. It is then carried by the sap stream into the plant roots, where it prevents them from absorbing nutrients from the soil – thereby killing the plant.
Glyphosate also has several secondary or indirect effects on plant physiology which may also explain its herbicidal effects 77. The toxicity of glyphosate could be related to its effects on other physiological processes such as mineral nutrition and photosynthesis, and to the plant’s hormone and oxidative status 77. The alteration of these cellular processes could be directly linked to the deleterious effects of glyphosate observed on plant growth and production. As a metal chelator, glyphosate could deprive plants from important nutrients which have important roles as enzymatic co-factors, biomolecular constituents, and anti-oxidative systems 77. Oxidative stress, more specifically lipid peroxidation induced by glyphosate, is known to severely damage the cell integrity which may lead to cell death 77. Moreover, increased reactive oxygen species (ROS) production can negatively interfere with photosynthetic processes, for example by decreasing the chlorophyll content, photochemical efficiency, and C metabolism, leading to reduction in plant growth 77.
Annual weeds, including grasses and most broad-leafed plants, are easily controlled using glyphosate. This is because they have soft tissue and when growing actively they quickly absorb
enough chemical to destroy the plant. However, weeds with bulbs and perennial weeds with woody stems are much harder to control. They will only die if sufficient glyphosate reaches each plant’s root system. In all cases, but particularly with bulbs and woody weeds, timing of the spray application is critical.
Glyphosate uses
Glyphosate is often used in agriculture, forestry, lawn and garden care to control broadleaf plants, weeds and grasses that compete with crops 64, 78, 79, 80. In agriculture, glyphosate is used in no-till cropping to kill weeds before planting the next crop. Glyphosate is also used to control invasive weeds to help with conservation of natural habitats. Glyphosate can be applied to plants by spraying using different spray equipment, depending on the situation.
Glyphosate products are supplied as concentrates for professional users and ready to use sprays for home garden users. Always put 80 per cent of the required water into your clean sprayer before adding glyphosate, then slowly add the other 20 per cent of the water. This avoids frothing of the spray mix.
According to data from the Pesticide Action Network (PAN) Pesticide Database, there are 102 products containing glyphosate (CASRN 1071-83-6) as the active ingredient, 94 of which have active registrations in the United States. There are 848 products containing glyphosate isopropylamine salt (CASRN 38641-94-0) as the active ingredient, of which 739 have active registrations in the United States 81, 82.
Dealing with Problem Weeds
The following guidelines are for particular problem weeds will help ensure that glyphosate works well for you without over using or respraying unnecessarily.
Soursobs (Bermuda buttercup or Oxalis corniculata)
For soursobs (Bermuda buttercup or Oxalis corniculata) the best results will be achieved by spraying glyphosate when the plants are about one third in flower. At this growth stage glyphosate will be effectively carried down to the bulbs of the plants.
Dealing with Perennial Plants
To kill perennial plants such as couch grass, glyphosate needs to be applied to fresh, actively growing green vegetation with sufficient leaf area to absorb enough glyphosate to kill the whole plant. Slashing or mowing and allowing ample regrowth (good lush green growth 10 to 15 cm long in the case of kikuyu or couch grass) before spraying can improve the chances of maximum control.
Glyphosate health effects
Ingesting glyphosate can cause mucous membrane irritation, abdominal pain, vomiting, diarrhea, and other effects. In fatal cases, it can cause hypovolemic shock, cardiac arrhythmias, metabolic acidosis, and pulmonary edema.
Is glyphosate safe?
Pure glyphosate is low in toxicity by the oral, dermal and inhalation exposure routes, but products usually contain other ingredients that help the glyphosate get into the plants 83. The other ingredients in the product can make glyphosate product more toxic. Products containing glyphosate may cause eye or skin irritation 84. People who breathed in spray mist from products containing glyphosate felt irritation in their nose and throat. Swallowing products with glyphosate can cause increased saliva, burns in the mouth and throat, nausea, vomiting, and diarrhea. Fatalities have been reported in cases of intentional ingestion. Pets may be at risk if they touch or eat plants that are still wet with spray from products containing glyphosate. Animals exposed to products with glyphosate may drool, vomit, have diarrhea, lose their appetite, ataxia, hunched posture, piloerection, convulsions, reduced activity, or seem sleepy. Clinical signs of glyphosate toxicity were observed in rats and mice following acute oral exposure to > 2,000 mg/kg body weight 85. Long-term feeding studies in animals were assessed by the U.S. Environmental Protection Agency (EPA) and other regulatory authorities. Based on these evaluations, they found there is no evidence glyphosate is toxic to the nervous or immune systems. They also found it is not a developmental or reproductive toxin 85.
in 2015, a committee of scientists working for the International Agency for Research on Cancer (IARC) of the World Health Organization (WHO) classified glyphosate as “probably carcinogenic to humans” (Group 2A) 86, 87. According to the International Agency for Research on Cancer (IARC) there was “limited evidence of carcinogenicity in humans for non-Hodgkin lymphoma” 86. The evidence in humans is from studies of exposures, mostly agricultural, in the USA, Canada, and Sweden published since 2001 88, 89, 90, 91. In addition, there is convincing evidence that glyphosate also can cause cancer in laboratory animals as well as strong evidence that exposure to glyphosate is genotoxic and can induce oxidative stress in experimental animals and in humans in vitro 86. “Limited evidence” means that a positive association has been observed between exposure to glyphosate and cancer but that other explanations for the observations called chance, bias, or confounding could not be ruled out. Group 2A category is also used when there is limited evidence of carcinogenicity in humans and strong data on how the agent causes cancer 86.
Glyphosate also caused DNA and chromosomal damage in human cells, although it gave negative results in tests using bacteria. One study in community residents reported increases in blood markers of chromosomal damage (micronuclei) after glyphosate formulations were sprayed nearby 86.
In 2005, an evaluation of glyphosate and cancer risk was conducted in the Agricultural Health Study 92. This evaluation considered glyphosate use reported at enrollment (1993–1997), and included 2088 cancers diagnosed between enrollment and 2001. No statistically significant associations were found for any cancer sites, including non-Hodgkin lymphoma, but there was an increased risk, though not statistically significant, of multiple myeloma in the highest exposure category based on a small number of cases 92.
In this large 2017 prospective cohort study 93, no association was apparent between glyphosate and any solid tumors or lymphoid malignancies overall, including non-Hodgkin lymphoma and its subtypes. There was some evidence of increased risk of acute myeloid leukemia (AML) among the highest glyphosate exposed group that requires confirmation 93.
Following the 2023 assessment by the European Food Safety Authority (EFSA), “based on the available evidence, glyphosate does not meet the criteria for endocrine disruption” 85.
Table 1. Humans exposed to glyphosate Cancer Outcomes for Solid Tumors
| Reference and study overview | Methods and outcomes | Results (with 95% confidence interval) | Study author conclusions and limitations |
|---|---|---|---|
| Andreotti et al 93 Prospective cohort study of 54,251 licensed pesticide applicators (97% white, 97% male) recruited between 1993 and 1997 in Iowa and North Carolina from the Agricultural Health Study to evaluate agricultural exposure to 50 pesticides (including glyphosate) and cancer incidence cases. 44,932 participants reported ever use of glyphosate, including 5,779 participants with incident cancer cases. | Exposure: Self-reported ever/never use of any glyphosate pesticides, lifetime days of glyphosate use (days per year × number of years), and intensity-weighted lifetime days (lifetime days × intensity score) at enrollment (1993–1997) or follow-up (1999–2005). Intensity-weighted lifetime days of glyphosate use was categorized into quartiles, tertiles, or the median, such that there were at least five exposed cases in each category. Outcome: Incident cancer diagnoses ascertained via linkage to cancer registries in Iowa (enrollment through 2013) and North Carolina (enrollment through 2012). Data analysis: Poisson regression Adjustments: Age, cigarette smoking status, alcohol drinks per month, family history of any cancer, state of recruitment, and the five pesticides (atrazine, alachlor, metolachlor, trifluralin, and 2,4- dichlorophenoxyacetic acid (2,4-D)). Confounders considered included body mass index (body mass index) and pack-years of cigarettes smoked. | Oral cavity: Q4: relative risk 0.84 (0.48–1.46) Colon: Q4: relative risk 1.01 (0.74–1.38) Rectum: Q4: relative risk 0.84 (0.52–1.34) Pancreas: Q4: relative risk 1.06 (0.57–1.97) Lung: Q4: relative risk 1.00 (0.76–1.33) Melanoma: Q4: relative risk 1.17 (0.78–1.74) Prostate: Q4: relative risk 0.99 (0.86–1.13) Testicular: T3: relative risk 0.57 (0.20–1.67) Bladder: Q4: relative risk 1.26 (0.87–1.82) Kidney: Q4: relative risk 1.03 (0.66–1.61) | Conclusions: The authors observed no associations between glyphosate use and overall cancer risk or risk of cancer of the oral cavity, colon, rectum, pancreas, lung, skin, prostate, testes, bladder or kidney. Risk estimates were similar in magnitude between the unlagged and lagged (5 or 20 years) exposure analyses for all sites evaluated. Limitations: Some misclassification of exposure undoubtedly occurred; because many cancer sites were evaluated, there is the possibility that results were observed by chance, and should be interpreted with caution. However, 37% of the participants did not respond to follow-up, which may have resulted in an underestimation of glyphosate exposure though imputation procedures were used in an attempt to mitigate this issue. |
| De Roos et al. 92 Prospective cohort study of 54,315 certified pesticide applicators (97% male, 97% Caucasian) in Iowa and North Carolina (Agricultural Health Study) to evaluate agricultural exposure to glyphosate and cancer incidence. Number cases (exposed percent) for different cancer sites:
| Exposure: Self-reported never/ever use of glyphosate. Cumulative exposure days (cumulative exposure days): 1–20 (reference), 21–56, and 57–2,678 days. Intensity weighted exposure days (intensity weighted exposure days) of 0.1–79.5 (reference), 79.6–337.1, and 337.2–18,241 units. Outcomes/endpoints: Cancer registry files in Iowa and North Carolina for case identification. Incident cases were identified from enrollment to 2001 (median follow-up time: 6.7 years). Data analysis: Poisson regression analyses for all cancers combined and 12 specific cancer sites (with at least 30 cases). Adjustments: Age at enrollment, education, pack-years of cigarette smoking, alcohol consumption, family history of cancer, state of residency, and co-exposure to 10 other pesticides (2,4- dichlorophenoxyacetic acid (2,4-D), alachlor, atrazine, metolachlor, trifluralin, benomyl, maneb, paraquat, carbaryl, and diazinon). | All cancers: Ever used: relative risk 1.0 (0.9–1.2) Lung: Ever used: relative risk 0.9 (0.6–1.3) Oral cavity: Ever used: relative risk 1.0 (0.5–1.8) Colon: Ever used: relative risk 1.4 (0.8–2.2) Rectum: Ever used: relative risk 1.3 (0.7–2.3) Pancreas: Ever used: relative risk 0.7 (0.3–2.0) Kidney: Ever used: relative risk 1.6 (0.7–3.8) Bladder: Ever used: relative risk 1.5 (0.7–3.2) Prostate: Ever used: relative risk 1.1 (0.9–1.3) Melanoma: Ever used: relative risk 1.6 (0.8–3.0) | Conclusions: No association between glyphosate exposure and all cancer incidence or most of the specific cancer subtypes, including non-Hodgkin’s lymphoma. A small number of cases suggested a positive association between multiple myeloma and glyphosate exposure. Limitations: Self-reported exposure information, few cases for many of the cancer subtypes, most applicators were male, there is no information on timing of pesticide use in relation to disease. |
| Engel et al. 94 Prospective cohort study of 30,454 wives (98% Caucasian) of private pesticide applicators (largely farmers) in Iowa and North Carolina (Agricultural Health Study) to evaluate breast cancer risk in relation to use of individual pesticides by the women themselves or by their husbands. Glyphosate analysis for wife’s pesticide use among all wives in the cohort included 82 exposed and 227 unexposed cases (n= 309) and 10,016 exposed and 20,129 (n= 30,145) unexposed controls. Further analysis of husband’s pesticide use among wives who reported never having used pesticides themselves included 109 “exposed” (husband used pesticide) and 43 “unexposed” cases and 9,304 “exposed” and 3,993 “unexposed” controls. | Exposure: Self-reported ever/never use of any glyphosate products at enrollment (1993–1997). Husband’s information was used as a measure of possible indirect pesticide exposure for their wives. Outcomes/endpoints: Breast cancer incident cases identified through state cancer registries from enrollment to 2000 (mean follow-up period: 4.8 years). Data analysis: Poisson regression Adjustments: Age, race, and state of residence. Confounders considered included body mass index, age at menarche, parity, age at first birth, menopausal status, age at menopause, family history of breast cancer, physical activity, smoking, alcohol consumption, fruit and vegetable consumption, and education. | Breast cancer: Wife’s pesticide use among all wives in cohort: relative risk 0.9 (0.7–1.1) Husband’s pesticide use among wives who never used pesticides: relative risk 1.3 (0.8–1.9) | Conclusions: No specific conclusion was given on glyphosate exposure and breast cancer. Limitations: Some associations may have occurred by chance, data on pesticide-specific exposure-response relations were only available for the husband, lack of information on how long each woman had been married to her current partner, limited power to assess associations for less commonly used pesticides, pesticide use was based on self-reporting. |
| Flower et al 95 Prospective and retrospective cohort study of 17,280 children (52% male, 96% Caucasian) of pesticide applicators in Iowa (Agricultural Health Study) to evaluate parental exposure to 50 pesticides (including glyphosate) and childhood cancer risk. Glyphosate analysis included 6,075 children (13 cases) with maternal use and 3,231 children (6 cases) with paternal use of glyphosate. | Exposure: Self-reported parental ever/never use of any glyphosate product by both applicators and spouses at enrollment (1993–1997). Outcomes/endpoints: Childhood cancer cases were both retrospectively and prospectively identified after parental enrollment through Iowa Cancer registries from 1975 to 1998. Data analysis: Multiple logistic regression. Adjustments: Child’s age at parent’s enrollment. Confounders considered included parental age at child’s birth, child’s sex, child’s birth weight, history of parental smoking, paternal history of cancer, and maternal history of miscarriage. | Childhood cancers: Maternal use (ever): odds ratio 0.61 (0.32–1.16) | Conclusions: No significant associations were observed between maternal (or paternal) pesticide including glyphosate application, including increased frequency of application, and risk of childhood cancer risk. Limitations: Small number of cases limits statistical power, maternal use is limited by lack of data on timing of exposure in relation to child’s birth, paternal prenatal use constitutes a broad window of exposure and not necessarily just prenatal. |
| Koutros et al. 96 Prospective cohort study of 54,412 certified pesticide applicators in Iowa and North Carolina (Agricultural Health Study) to evaluate agricultural exposure to 50 pesticides (including glyphosate) and prostate cancer risk. There were 1,962 incident prostate cancer cases, 919 of whom had aggressive prostate cancer. Glyphosate analysis included 1,464 exposed and 498 unexposed cases (n=1,962) and 42,420 exposed and 10,015 unexposed controls (n=52,435). | Exposure: Self-reported ever/never glyphosate use, lifetime days of glyphosate use (years of use × days/year used), intensity-weighted lifetime days of glyphosate use (lifetime days × exposure intensity) at enrollment (1993–1997). Exposure was categorized into non-exposed and quartiles exposure on the basis of the distribution of exposed cases. Outcomes/endpoints: Prostate cancer incidences determined through state cancer registries from enrollment to 2007. Data analysis: Poisson regression. Adjustments: Age at enrollment, race, state, family history of prostate cancer, smoking, fruit servings, and leisure-time physical activity in the winter. Separate glyphosate analyses were conducted by disease aggressiveness and family history of prostate cancer (yes, no). | Cumulative lifetime exposure based on intensity-weighted days: Total prostate cancer: Q4: relative risk 0.99 (0.86–1.15) Aggressive prostate cancer: Q4: relative risk 0.94 (0.75–1.18) Total prostate cancer, no family history: Q4: relative risk 1.02 (0.86–1.21) Total prostate cancer, with family history: Q4: relative risk 0.95 (0.64–1.40) | Conclusions: No significant association was found between any specific pesticide including glyphosate and risk of total prostate cancer. Limitations: Information on Gleason score of severity was missing for some and not standardized, which most likely led to an underestimation of advanced cases; use of take-home questionnaire could introduce selection bias and exposure misclassification; large number of pesticides investigated so cannot rule out the possibility that some findings may be due to chance. |
| Koutros et al 97 Prospective cohort study of 54,344 male pesticide applicators in Iowa and North Carolina (Agricultural Health Study) to evaluate agricultural exposure to 65 pesticides (including glyphosate) and bladder cancer risk (n=321 incident cases identified). Glyphosate analysis included 248 exposed and 73 unexposed cases (n=321) and 54,023 controls. | Exposure: Self-reported ever/never glyphosate use, lifetime days of glyphosate use (years of use × days/year used), intensity-weighted lifetime days of glyphosate use (lifetime days × exposure intensity) at enrollment (1993–1997). Outcomes/endpoints: Bladder cancer incidences determined through state-based cancer registries from enrollment through 2010 in North Carolina and 2011 in Iowa. Data analysis: Poisson regression. Adjustments: Age, race, state, cigarette smoking, and pipe smoking. | Bladder cancer: Ever use: relative risk 1.17 (0.78–1.77) Cumulative lifetime exposure based on intensity-weighted days: Overall Stratification by smoking status Never smoker: Former smoker: Current smoker: | Conclusions: No specific conclusion given on glyphosate exposure and bladder cancer. Never smokers who were heavy users of the glyphosate had increased risk of bladder cancer. Limitations: Potential for exposure misclassification, findings may be due to chance, due to small number of cases. |
| Lee et al 98 Prospective cohort study of 56,813 certified pesticide applicators (97% male, 97% Caucasian) in Iowa and North Carolina (Agricultural Health Study) to evaluate agricultural exposure to 50 pesticides (including glyphosate) and colorectal cancer risk. Glyphosate analysis included 225 exposed and 67 unexposed for colorectal cancer cases (n=305), 151 exposed and 49 unexposed for colon cancer cases (n=212), and 74 exposed and 18 unexposed for rectal cancers (n=93). | Exposure: Self-reported ever use of any glyphosate pesticides at enrollment (1993–1997). Outcomes/endpoints: Colorectal cancer incidences determined through cancer registries from enrollment to 2002 (mean follow-up period: 7.3 years). Data analysis: Unconditional multivariate logistic regressions. Adjustments: Age, state of residence, smoking history, total pesticide application days to any pesticide. Confounders considered included body mass index, race, license type, education level, aspirin intake, family history of colorectal cancer, physical activity, smoking, and intakes of meat, fruits, vegetables, and alcohol. | Colorectal cancer: odds ratio 1.2 (0.9–1.6) Colon cancer: odds ratio 1.0 (0.7–1.5) Rectal cancer: odds ratio 1.6 (0.9–2.9) | Conclusions: No specific conclusion was given on glyphosate exposure and colorectal cancers. Limitations: Since the study examined risks for 50 pesticides, it is possible that some significant findings might occur by chance alone due to the multiple comparisons. Potential recall bias and thus exposure misclassification associated with subjects recalling pesticide use from many years ago. |
| Andreotti et al 99 Nested case-control study of 93 cases of pancreatic cancer (64 applicators and 29 spouses) and 82,503 controls (52,721 applicators and 29,782 spouses) from the Agricultural Health Study, conducted in Iowa and North Carolina, to evaluate the association of pancreatic cancer and use of 24 pesticides (including glyphosate). Glyphosate analysis included 55 exposed and 35 unexposed cases (n= 90) and 48,461 exposed and 31,282 unexposed controls (n= 79,743). | Exposure: Self-reported ever/never use of any glyphosate product for applicators and spouses and intensity-weighted lifetime exposure days for applicators at enrollment (1993–1997). Outcomes/endpoints: Pancreatic cancer incidences identified through state cancer registries from enrollment to 2004 (over 9 years of follow-up time). Data analysis: Unconditional logistic regression. Adjustments: Age, cigarette smoking, diabetes, and subject type for ever/never pesticide exposure (applicator versus spouse). | Pancreatic cancer: Ever/never among applicators and spouses: odds ratio 1.1 (0.6–1.7) Intensity weighted pesticide exposure among applicators: Never: 1.0 (reference) | Conclusions: No specific conclusion given on glyphosate exposure and pancreatic cancer. Limitations: There was a limited number of exposed cases and limited in generalizability due to predominantly white male study population. |
| Band et al 100 Case-control study on male cancer patients (96.8% Caucasian) in British Columbia, Canada, to evaluate exposure to 139 specific active compounds in pesticides (including glyphosate) and prostate cancer risk. Glyphosate analysis included 25 exposed and 1,128 unexposed cases (n=1,153) and 60 exposed and 3,939 age-matched internal controls (patients with cancer of other primary site) controls (n=3,999). | Exposure: Self-reported ever/never use of glyphosate pesticides from questionnaire. Agricultural job exposure matrix (job exposure matrix) was developed for farm workers in British Columbia for the period of 1950–1998. Outcomes/endpoints: Prostate cancer cases identified through British Columbia Cancer Registry for 1983–1990 and histologically confirmed. Data analysis: Conditional logistic regression on age-matched sets of cases and controls. Adjustments: Alcohol consumption, cigarette years, education level, p-years, and respondent. Confounders considered included marital status, smoking (age started smoking, average number of cigarettes, pipe or cigars smoked per day, total years smoked), and ethnicity. | Prostate cancer: odds ratio 1.36 (0.83–2.25) | Conclusions: No specific conclusion given on glyphosate exposure and prostate cancer. job exposure matrix likely to result in non-differential misclassification and may underestimate the true association; thus, negative findings should be regarded as inconclusive. Limitations: Lack of information on familial history, potential for misclassification of exposure due to use of job exposure matrix, use of cancer controls may result in selection bias, statistically significant associations could have occurred by chance as a result of multiple comparisons since 142 active chemicals were examined. |
| Lee et al 101 Case control study of white men and women (ages ≥21 years) diagnosed with stomach adenocarcinoma (n=170) or esophagus adenocarcinoma (n=137) and 502 controls in eastern Nebraska to evaluate the risk of the stomach and esophageal adenocarcinomas associated with farming and agricultural use of 16 insecticides and 14 herbicides (including glyphosate). Glyphosate analysis included 12 cases of stomach cancer and 12 cases of esophageal cancer among farmers, and 46 controls compared to non-farmers (59 stomach cancer, 62 esophageal cancer cases and 184 controls). Controls were randomly selected from a group of controls interviewed in 1986–1987 for a previous population-based case-control study. Controls were frequency-matched by sex and age to the combined distribution of the stomach and esophagus cases. | Exposure: Self- or proxy-reported ever use of glyphosate pesticide at enrollment (1992–1994). Outcomes: Stomach and esophageal cancer cases were identified from the Nebraska Cancer Registry (1988–1990) or by review of discharge diagnosis and pathology records at 14 hospitals (1991–1993). Data analysis: Unconditional logistic regression. Adjustments: Age, sex. Confounders considered included body mass index, smoking, alcohol consumption, educational level, family history of stomach or esophageal cancer, respondent type, dietary intake of vitamin A and C, b-cryptoxanthin, riboflavin, folate, zinc, dietary fiber, protein, and carbohydrate. | Stomach cancer: odds ratio 0.8 (0.4–1.5) Esophageal cancer: odds ratio 0.7 (0.3–1.4) | Conclusions: “No significant associations were found between specific agricultural pesticide exposures (including glyphosate) and the risk of stomach or esophageal adenocarcinomas among Nebraska farmers.” Limitations: Possible misclassification of pesticide exposure and generally small number of farmers exposed to some of the individual pesticides. |
| Lee et al 102 Case control study of 251 white men and women (ages ≥21 years) diagnosed with gliomas and 498 controls in eastern Nebraska (Nebraska Health Study II) to evaluate adult glioma associated with farming and agricultural use of 20 insecticides and 17 herbicides (including glyphosate). Glyphosate analysis (only conducted among male farmers) included 17 cases and 32 controls among farmers compared to non-farmers (49 cases and 112 controls). Among these, self-reported respondents included 4 cases/17 controls for glyphosate users and 20 cases/40 controls for reference non-farmers; proxy-reported respondents included 13 cases/15 controls for glyphosate users and 29 cases/72 controls for reference non-farmers. Controls were randomly selected from a group of controls interviewed in 1986–1987 for a previous population-based case-control study. Controls were frequency-matched by sex, age, and vital status to the combined distribution of the cases. | Exposure: Self- or proxy-reported ever use of glyphosate pesticide at enrollment (1992–1994). Outcomes: Incident primary adult glioma cases diagnosed between 1988 and 1993 were identified from the Nebraska Cancer Registry or from 11 hospitals. Data analysis: Unconditional logistic regression. Separate analyses by sex and respondent type (self- versus proxy-reported) were also conducted. Adjustments: Age, sex, and respondent type. Confounders considered included history of head injury, marital status, education level, alcohol consumption, medical history of diabetes mellitus, dietary intake of a- and b-carotene, and dietary fiber. | Glioma among male farmers: odds ratio 1.5 (0.7–3.1), all reported glyphosate use odds ratio 0.4 (0.1–1.6), self-reported glyphosate use odds ratio 3.1 (1.2–8.2), proxy-reported glyphosate use | Conclusions: “Glioma risk was also significantly increased among men who used specific pesticides (including glyphosate) and pesticide chemical classes; however, the positive results were mostly limited to proxy respondents.” Limitations: The major limitation was the large proportion of proxy respondents. Most of the associations observed were limited to proxy respondents. |
| Pahwa et al 103 Case control study of 357 soft tissue sarcoma cases and 1,506 controls in Canada (all males, ≥19 years of age) to investigate the putative associations of pesticides (including glyphosate) with soft-tissue sarcoma (soft tissue sarcoma). Glyphosate analysis included 36 exposed and 321 unexposed cases and 147 exposed and 1,359 unexposed controls. | Exposure: Self-reported ever use of glyphosate herbicides collected through self-administered postal questionnaire and telephone interviews. Outcomes: soft tissue sarcoma cases (first diagnosed in 1991–1994) ascertained from provincial cancer registries, except in Quebec, where hospital ascertainment was used. Data analysis: Conditional logistic regression. Adjustments: Age, province of residence, medical history. | Soft tissue sarcoma: odds ratio 0.93 (0.60–1.42), stratified by age group and province of residence odds ratio 0.90 (0.58–1.40), adjusted for medical history and with strata for age group and province of residence | Conclusions: “No association between herbicides (individual compound or major chemical class) (including glyphosate) and soft tissue sarcoma.” Limitations: Limitations common to epidemiological case-control studies. |
| Yiin et al 104 Case control study of 798 cases of glioma and 1,175 controls (98% white, aged 18–80 years) in Iowa, Michigan, Minnesota, and Wisconsin (Upper Midwest Health Study) to investigate association between exposure to pesticides (including glyphosate) and risk of glioma in male and female participants. House and garden pesticide use: Glyphosate analysis included 51 exposed and 747 unexposed cases and 76 exposed and 1,099 unexposed controls. Analysis included 28 exposed and 410 unexposed cases and 75 exposed and 1,066 unexposed controls excluding proxy respondents. Randomly-selected, population-based controls were frequency-matched within a state. | Exposure: Self- or proxy-reported ever/never use of glyphosate pesticide through 1992. Outcomes: Cases with a histologically confirmed primary intracranial glioma were identified through medical facilities, oncologists, neurosurgeons, and cancer registries (1995–1997). Data analysis: Unconditional logistic regression. Analyses were separately conducted with or without proxy respondents. Adjustments: Age, sex, education. | Glioma Non-farm job use: odds ratio 0.83 (0.39–1.73) including proxy respondents; odds ratio 0.79 (0.33–1.86) excluding proxy respondents. House and garden use: odds ratio 0.98 (0.67–1.43) including proxy respondents; odds ratio 0.84 (0.52–1.33) excluding proxy respondents | Conclusions: “No individual pesticides (including glyphosate) or broader category of pesticides, with or without proxy respondent, was associated with a statistically significant decrease or elevation in glioma risk.” Limitations: A limitation of this study is the high proportion (45%) of proxy interviews for case participants compared to 2.9% control interviews that were with proxies. The accuracy and completeness of information given by proxy respondents varies by many factors. Another concern is the validity and reliability of the pesticide exposure assessment. |
Footnotes: Relative risk (RR) is a ratio of the probability of an event occurring in the exposed group versus the probability of the event occurring in the non-exposed group. A relative risk (RR) of 1 implies there is no difference of the event if the exposure has or has not occurred. If the relative risk (RR) > 1, then the event is more likely to occur if there was exposure. If the relative risk (RR) < 1, then the event is less likely to occur if there was exposure. For example, when the relative risk (RR) is 2 the chance of a bad outcome is twice as likely to occur with the treatment as without it; whereas an relative risk (RR) of 0.5 means that the chance of a bad outcome is twice as likely to occur without the intervention. When the relative risk (RR) is exactly 1, the risk is unchanged.
Abbreviations: BMI = body mass index; CED = cumulative exposure day; CI = confidence interval; IWED = intensity weighted exposure day; JEM = job exposure matrix; NHL = non-Hodgkin’s lymphoma; OR = odds ratio; RR = relative risk; Q = quartile; STS = soft tissue sarcoma; T = tertile
[Source 105 ]Table 2. Humans exposed to glyphosate Noncancer Outcomes
| Reference and study population | Exposure | Outcomes |
|---|---|---|
| Death | ||
| Cho et al 106 Retrospective cohort study of 150 adults who presented with glyphosate poisoning at a Korean hospital between January 2006 and April 2017. | Exposure: ingestion of glyphosate-based herbicides Multivariate logistic regression: age, amount of glyphosate-based herbicide ingested, elapsed time since ingestion, qSOFA score | 14/150 patients died (9.3% mortality), and age was determined to be a statistically significant factor (p<0.001). Higher qSOFA scores were associated with greater odds of death (odds ratio (OR):2.73, 95% confidence interval: 1.41–5.76) and life threatening complications (odds ratio (OR): 17.19, 95% confidence interval: 6.25–72.65). |
| Respiratory | ||
| Camacho and Mejia 107 Cross-sectional study examining individual health records from the general public over a five-year period (2003 to 2007) merged with data of aerial spraying events. The study examined the data under several specifications:
| Exposure: aerial spraying of glyphosate on coca crops and the general population living in the spray areas within study period 2003 to 2007 Regression Adjustments: age, age square, health regime, municipal tax income, population, area in square km, rurality index, average monthly rainfall, municipal spending on education and health, subsidized regime coverage, year and month dummy | Increased number of respiratory illnesses consistent across all specifications analyzed (only statistically significant p values were presented). |
| Hoppin et al 108 Cohort study of 20,468 participants in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use and application frequency categories Logistic regression adjustments: age, state, smoking history, asthma-atopy status | Wheeze, self-reported odds ratio (OR) 1.05 (0.95–1.17), p=0.04 for trend of increasing exposure days |
| Hoppin et al 109 Prospective cohort study of 20,175 participants in the Agricultural Health Study in Iowa and North Carolina (17,920 farmers and 2,255 commercial pesticide applicators) | Exposure: glyphosate ever use in the year prior to enrollment Logistic regression adjustments: age, state, smoking history, body mass index (BMI) | Wheeze, self-reported
|
| Hoppin et al 110 Cohort study of 2,255 commercial pesticide applicators participating in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use in the year prior to enrollment Logistic regression adjustments: age, smoking status, asthma and atopy history, body mass index (BMI) | Wheeze, self-reported odds ratio (OR) 1.38 (1.03–1.86) odds ratio (OR) 1.14 (0.83–1.57), with adjustment for use of chlorimuron-ethyl pesticide |
| Hoppin et al 111 Prospective cohort study of 20,908 participants in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Logistic regression adjustments: age, state, sex, smoking (pack-years) | Chronic bronchitis odds ratio (OR) 0.99 (0.82–1.19) |
| Hoppin et al 112 Prospective cohort study of 25,814 farm women participating in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Logistic regression adjustments: age, state, smoking status, “grew up on farm” | Atopic asthma odds ratio (OR) 1.31 (1.02–1.67) Nonatopic asthma odds ratio (OR) 1.13 (0.92–1.39) |
| Hoppin et al 113 Prospective cohort study of 19,704 male farmers participating in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Logistic regression adjustments: age, state, smoking status, body mass index (BMI) | Allergic asthma odds ratio (OR) 1.37 (0.86–2.17) Nonallergic asthma odds ratio (OR) 1.15 (0.87–1.51) |
| Hoppin et al 114 Prospective cohort study of 22,134 male farmers participating in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Logistic regression adjustments: age, body mass index (BMI), state, smoking status, current asthma, days applying pesticides, days driving diesel tractors | Allergic wheeze odds ratio (OR) 1.56 (1.19–2.03), higher prevalence with increasing use days per year Nonallergic wheeze odds ratio (OR) 1.24 (1.07–1.44) |
| Slager et al 115 Prospective cohort study of 2,245 commercial applicators participating in the Agricultural Health Study in Iowa | Exposure: any glyphosate use and application frequency categories during the past year Logistic regression adjustments: age, education, “growing up on farm” | Current rhinitis odds ratio (OR) 1.32 (1.08–1.61), p=0.735 for trend for increasing use days per year |
| Slager et al 116 Prospective cohort study of 19,565 farmers participating in the Agricultural Health Study in Iowa and North Carolina | Exposure: any glyphosate use and application frequency categories during the past year Logistic regression adjustments: age; race; education; state; body mass index (BMI); currently working on farm; years mixing pesticides, repairing engines or pesticide equipment, welding, painting, handling stored grain or hay, working in swine areas, working with hogs or other farm animals, butchering animals, and growing cabbage, Christmas trees, field corn, sweet corn, and hay | Current rhinitis odds ratio (OR) 1.09 (1.05–1.13) |
| Cardiovascular Effects | ||
| Dayton et al 117 Case control study of 168 cases of nonfatal myocardial infarction and 22,257 controls in women in Iowa and North Carolina participating in the Agricultural Health Study | Exposure: glyphosate ever use Logistic regression adjustments: age, body mass index (BMI), smoking, state | Nonfatal heart attack odds ratio (OR) 0.8 (0.6–1.2) |
| Mills et al 118 Prospective study of male participants in the Agricultural Health Study in Iowa and North Carolina (n=54,069 for fatal myocardial infarction and 32,024 for nonfatal incidence) | Exposure: glyphosate ever use Cox proportional regression adjustments: age, state, smoking, body mass index (BMI) (nonfatal analysis only) | Fatal heart attack hazard ratio (HR) 0.99 (0.80–1.23) Nonfatal heart attack hazard ratio (HR) 1.10 (0.93–1.31) |
| Musculoskeletal Effects | ||
| De Roos et al 119 Nested case control study of 135 cases of physician-confirmed rheumatoid arthritis and 675 controls participating in the Agricultural Health Study in Iowa and North Carolina (female participants only) | Exposure: glyphosate ever use Logistic regression adjustments: birth date, state | Rheumatoid arthritis odds ratio (OR) 1.2 (0.8–1.8) |
| Parks et al 120 Nested case-control study of cases of physician-confirmed rheumatoid arthritis or self-reported use of disease modifying antirheumatic drugs and noncases participating in the Agricultural Health Study in Iowa and North Carolina (female spouses of licensed pesticide applicators only); enrolled between 1993 and 1997 and followed through 2010 | Exposure: glyphosate ever use Logistic regression adjustments: age, state, pack-years smoking | Rheumatoid arthritis odds ratio (OR) 1.2 (0.95–1.6); based on 100 prevalent cases odds ratio (OR) 1.4 (1.0–2.0); based on 54 incident cases |
| Liver Effects | ||
| Mills et al 121 Case-control study of 97 participants, 63 with nonalcoholic steatohepatitis (NASH) and 34 without NASH | Exposure: glyphosate and glyphosate residue measured from fasting urine; glyphosate residue (calculated) which estimates dietary intake and exposure to residues Multivariable linear adjustments: age, sex, body mass index | Glyphosate residue significantly higher in patients with nonalcoholic steatohepatitis (NASH) compared with patents without nonalcoholic steatohepatitis (NASH) (p=0.008) Significant dose-dependent increase of glyphosate exposure with increase in fibrosis stage when comparing patients without advanced fibrosis to those with Stage 2,3 or 4 fibrosis (glyphosate: 0.230 mg/L, SD: 0.19 vs. 0.351 mg/L, SD: 0.45; glyphosate residue: 0.525 mg/L, SD 0.38 vs. 0.938 mg/L, SD: 0.372,p <0.001). Nonalcoholic steatohepatitis (NASH) cases confirmed through liver biopsy. |
| Skin Effects | ||
| Camacho and Mejia 122 Cross-sectional study examining individual health records from the general public over a five-year period (2003 to 2007) merged with data of aerial spraying events. The study examined the data under several specifications:
| Exposure: aerial spraying of glyphosate on coca crops and the general population living in the spray areas within study period 2003 to 2007 Regression Adjustments: age, age square, health regime, municipal tax income, population, area in square km, rurality index, average monthly rainfall, municipal spending on education and health, subsidized regime coverage, year and month dummy | Increased number of skin conditions consistent across all specifications analyzed (only statistically significant p values were presented). |
| Maibach 123 Experimental study of 24 males and females | Exposure: 0.1 mL applied to intact and Draize-type abraded skin; patch removed after 24 hours | No skin irritation 24 or 48 hours after application to intact skin Irritancy scores 24 hours after application to abraded skin were negative in 10 subjects, equivocal in 4 subjects and erythema was noted in 10 subjects; at 48 hours, the scores were negative in 10 subjects, equivocal in 6 subjects, and erythema was noted in 8 subjects |
| Maibach 123 Experimental study of 23 males and females | Exposure: 0.1 mL applied 5 days/week for 21 days | The average score was 1.4 where a score of 1 indicates redness and 2 indicates redness and swelling; none of the subjects reported burning, stinging, or itching from the test compound |
| Maibach 123 Experimental study of 204 males and females | Exposure: 0.2 mL applied 3 days/week for 3 weeks with patches remaining in place for 48–72 hours; a challenge patch was applied after a 2-week rest period | No skin irritation was observed |
| Maibach 123 Experimental study of 15 males and females | Exposure: Full-strength glyphosate was applied to skin stripped of the stratum corneum; the test site received irradiation with ultraviolet A and ultraviolet B light | No positive results for photoirritation or photosensitization were found |
| Eye Effects | ||
| Kirrane et al 124 Prospective study of 31,173 female spouses of commercial pesticide applicators participating in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Hierarchical regression adjustments: age, state | Retinal degeneration odds ratio (OR) 1.1 (0.8–1.5) |
| Endocrine Effects | ||
| Goldner et al 125 Prospective study of 16,529 participants (female spouses only) in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Polytomous logistic regression adjustments: age, education, smoking status, hormone replacement therapy, body mass index (BMI) | Hyperthyroid disease (overactive thyroid) odds ratio (OR) 0.98 (0.78–1.2) Hypothyroid disease (underactive thyroid) odds ratio (OR) 1.0 (0.91–1.2) Other thyroid disease odds ratio (OR) 0.97 (0.81–1.2) |
| Kongtip et al 126 Cross sectional study of 195 conventional farmers and 222 organic farmers in Thailand. Sera were collected and analyzed for the following thyroid levels: TSH, T3, T4, free T3 and free T4 | Exposure: glyphosate use, amount recorded in a diary Generalized linear regression adjustments: sex, current smoking, current alcohol use, insecticide use at home in the past year, triglyceride levels, and any stress symptoms in the past 2–4 weeks | Log(e) estimates of thyroid hormone levels predicted by models of glyphosate sprayed in past year Expβ (95%confidence interval):
Increased use of glyphosate sprayed (measured in moles) was found to increase T4 levels. |
| Shrestha et al 127 Prospective study of 35,150 male and female pesticide applicators in the Agricultural Health Study in Iowa and North Carolina followed over 20 years | Exposure: glyphosate ever use Logistic regression adjustments: sex, education, state of residence, smoking | Hypothyroid disease (underactive thyroid disease)
|
| Neurological Effects | ||
| Kamel et al 128 Case control study of cases of self-reported Parkinson’s disease (n=83 prevalent cases and 78 incident cases) and controls (n=79,557 prevalent controls and 55,931 incident controls) participating in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Logistic regression adjustments: age, state, type of participant | Parkinson’s disease
Prevalent disease defined as reporting Parkinson’s disease at enrollment and incident disease defined as Parkinson’s disease reported at the study follow-up |
| Zhang et al 129 A cross-sectional study of 218 farmers in China. Abnormalities of peripheral nerve conduction including nerve conduction velocity, distal motor latency, and amplitude were measured with a conventional nerve conduction assessment. | Exposure: glyphosate agriculture use Logistic regression adjustments: age, gender, smoking habit, alcohol consumption, whether adopting personal protective equipment, and whether suffering from diabetes mellitus | Abnormalities in nerve conduction velocity
Abnormalities in nerve conduction distal motor latency odds ratio (OR) 1.05 (0.81-1.37) Abnormalities in nerve conduction amplitude odds ratio (OR) 1.21 (0.75-1.97) |
| Reproductive Effects | ||
| Camacho and Mejia 122 Cross-sectional study examining individual health records from the general public over a five-year period (2003 to 2007) merged with data of aerial spraying events. The study examined the data under several specifications:
| Exposure: aerial spraying of glyphosate on coca crops and the general population living in the spray areas within study period 2003 to 2007 Regression Adjustments: age, age square, health regime, municipal tax income, population, area in square km, rurality index, average monthly rainfall, municipal spending on education and health, subsidized regime coverage, year and month dummy | Increased number of miscarriages consistent across all specifications analyzed (only statistically significant p values were presented). |
| Curtis et al 130 Retrospective cohort study of 2,012 planned pregnancies among participants in the Canadian Ontario Farm Family Health Study | Exposure: glyphosate use on the farm Cox proportional hazard adjustments: age when beginning to try to conceive, recent oral contraceptive use, men’s and women’s smoking, and use of other pesticides | Fecundability (the likelihood of becoming pregnant during a menstrual cycle or within a month)
|
| Developmental Effects | ||
| Arbuckle et al 131 Retrospective cohort study of 2,110 female participants in the Canadian Ontario Farm Family Health Study | Exposure: glyphosate use during gestation Logistic regression adjustments: none | Spontaneous abortion, preconception exposure
Spontaneous abortion, postconception exposure
|
| Garcia et al 132 Case control study of 261 cases of congenital malformations and 261 matched controls in Spain | Exposure: paternal glyphosate use Conditional logistic regression adjustments: paternal age and paternal job and maternal history of spontaneous abortion, twins, drug consumption, heavy smoking, education, occupation | Congenital malformations odds ratio (OR) 0.94 (0.37–2.34) for the acute risk period (during 3 months preceding conception or during the first trimester of pregnancy or both for the father and during 1 month preceding conception or during the first trimester of pregnancy or both for the mother) |
| Garry et al 133 Cross sectional study of 695 families and 1,532 children in Minnesota | Exposure: glyphosate ever use Regression adjustments: maternal age, smoking status, alcohol use, season of conception | Attention deficit disorder/attention deficit hyperactivity disorder (ADD/ADHD), parent reported odds ratio (OR) 3.6 (1.35–9.65) |
| Parvez et al 134 Birth-cohort study of 71 women with singleton pregnancies in Central Indiana | Exposure: maternal glyphosate levels in urine specimens Nonparametric Spearman correlation adjustments: maternal age, pre-pregnancy body mass index (BMI), tobacco use, alcohol use, trimester of pregnancy | Shortened gestational length (r = −0.30, p = 0.01) Reduced birth weight (r = −0.14, p = 0.27) Head circumference (r = −0.06, p = 0.64) |
| Rull et al 135 Case control study of 731 cases of neural tube defects and 940 controls in California | Exposure: maternal residential proximity to glyphosate application (within 1,000 m) Logistic regression adjustments: maternal ethnicity, education, periconceptional smoking, vitamin use | Neural tube defects
|
| Sathyanarayana et al 136 Prospective study of 2,246 women whose most recent singleton birth occurred within 5 years of enrollment in the Agricultural Health Study in Iowa and North Carolina | Exposure: maternal glyphosate ever use (n=700) Linear regression adjustments: maternal body mass index (BMI) and height, parity, preterm status, state, maternal smoking during pregnancy | Multiple regression estimates of change in birth weight (g) in relation to maternal self-reported glyphosate use (coefficient = 4 g; 95% confidence interval −40 to +48 g) indicate no significant association between birth weight and maternal use of glyphosate |
| Savitz et al 137 Retrospective cohort study of 1,898 couples participating in the Canadian Ontario Farm Family Health Study | Exposure: any paternal glyphosate use from 3 months prior to conception through the month of conception Logistic regression adjustments: maternal age, parity, maternal and paternal education, income, maternal and paternal off farm job, maternal smoking and alcohol use during pregnancy, conception to interview interval | Miscarriage odds ratio (OR) 1.5 (0.8–2.7) Preterm delivery odds ratio (OR) 2.4 (0.8–7.9) Small for gestational age odds ratio (OR) 0.8 (0.2–2.3) |
| Other Noncancer Effects | ||
| Montgomery et al 138 Prospective study of 33,457 participants (white males only) in the Agricultural Health Study in Iowa and North Carolina | Exposure: glyphosate ever use Logistic regression adjustments: age, state, body mass index (BMI) | Diabetes incidence odds ratio (OR) 0.85 (0.74–0.98) |
| Saldana et al 139 Prospective study of 11,273 participants in the Agricultural Health Study in Iowa and North Carolina | Exposure: any agricultural glyphosate exposure during the first trimester Logistic regression adjustments: body mass index (BMI) at enrollment, mother’s age at pregnancy, parity, race, state, commonly used pesticides by women | Gestational diabetes mellitus odds ratio (OR) 0.7 (0.2–1.75) |
Footnotes:
- Odds ratio (OR) is a measure of association between an exposure and an outcome. Odds ratio (OR) represents the odds that an outcome will occur given a particular exposure, compared to the odds of the outcome occurring in the absence of that exposure. Odds ratio (OR) > 1 means the event is more likely to occur with exposure; while an Odds ratio (OR) < 1 means the event is less likely to occur with exposure.
- Hazard ratio (HR) is a statistical measure that compares the rate of an event occurring in one group to the rate of the same event occurring in another group over time. Hazard ratios (HRs) are often used interchangeably with relative risk (RR) ratios, but there are important differences between the two. Relative risks (RR) are cumulative over an entire study, while hazard ratios (HRs) define instantaneous risk over a study period. Hazard ratio (HR) of 1 means there’s no difference in survival between the groups, while a hazard ratio (HR) greater than or less than 1 means one group had better survival.
Abbreviations: ADD/ADHD = attention deficit disorder/attention deficit hyperactivity disorder; BMI = body mass index; CFR = conditional fecundability ratio; CI = confidence interval; HR = hazard ratio; OR = odds ratio
[Source 105 ]Table 3. Toxicological profile of glyphosate metabolites found as residues in livestock and/or crops
| Metabolite | Genotoxicity | General toxicity Toxicological reference values (TRVs) | Additional source of human exposure (e.g. groundwater) |
|---|---|---|---|
| AMPA | Unlikely to be genotoxic | TRVs of glyphosate apply | No |
| N-acetyl AMPA | Unlikely to be genotoxic | TRVs of glyphosate apply | No |
| N-acetyl glyphosate | Negative for both mutagenicity and clastogenicity; aneugenicity not sufficiently investigated (data gap) | TRVs of glyphosate apply | No |
| N-methyl AMPA | Unlikely to be genotoxic | No data, not needed for consumer risk assessment | No |
| N-glyceryl AMPA | Negative for mutagenicity. Clastogenicity and aneugenicity not sufficiently investigated(data gap) | No data, not needed for consumer risk assessment | No |
| N-malonyl AMPA | Negative for mutagenicity. Clastogenicity and aneugenicity not sufficiently investigated(data gap) | No data, not needed for consumer risk assessment | No |
Abbreviation: AMPA = aminomethylphosphonic acid; TRVs = toxicological reference values
[Source 85 ]Animal Health Effects
Results from available animal studies identify the following glyphosate toxicity 105:
- Developmental effects: Histopathologic testicular lesions, decreased sperm production, and increased incidence of fetal skeletal malformations were reported in response to oral dosing of rat weanlings or pregnant rats with selected glyphosate formulations in the range of 5–500 mg/kg/day.
- Endocrine effects: Decreased serum testosterone was noted in male rat weanlings administered a glyphosate formulation orally at 5 mg/kg/day.
- Body weight effects: Seriously depressed body weight gain was observed in mice administered a glyphosate formulation orally at 50 mg/kg/day.
- Kidney effects: Histopathologic kidney lesions were noted in male rats gavaged once with a glyphosate formulation at 250 mg/kg.
- Liver effects: Increased serum liver enzyme activity and histopathologic liver lesions were reported in male rats repeatedly gavaged with a glyphosate formulation at 487 mg/kg/day.
- Hematological effects: Decreases in red blood cells, hematocrit, and hemoglobin, and increases in mean corpuscular volume and neutrophils were reported in mice administered a glyphosate formulation orally at 500 mg/kg/day.
- Reproductive effects: Increased percentage of morphologically abnormal sperm was reported among rats receiving a glyphosate formulation from the drinking water for 8 days at 640 mg/kg/day.
Results from the oral animal studies identify glyphosate toxicity at relatively high dose levels 105:
- Gastrointestinal effects: Clinical signs and/or pathological evidence of glyphosate-induced irritation were observed in several animal studies; the lowest dose level resulting in gastrointestinal effects was 175 mg/kg/day for diarrhea and few feces in pregnant rabbits administered glyphosate acid by gavage. Gastrointestinal disturbances are signs and/or symptoms following ingestion of large amounts of glyphosate-containing products.
- Developmental effects: Glyphosate treatment-related developmental effects were noted in a few studies at dose levels (≥1,234 mg/kg/day) resulting in maternal toxicity as well.
- Body weight effects: Depressed body weight and/or depressed body weight gain resulted from repeated dosing of glyphosate technical at dose levels ≥1,183 mg/kg/day.
- Liver effects: Increases in liver weight and serum ALT activity were observed in one repeated-dose study at a dose level of 1,678 mg/kg/day.
- Eye effects: Lens abnormalities were observed in one repeated-dose study at a dose level of 940 mg/kg/day.
- Kidney effects: Indicators of renal toxicity were noted in rats and mice administered glyphosate technical in the diet for 2 years at high doses (940 and 6,069 mg/kg/day, respectively).
- Other effects: Neurological, hematological, immunological, and reproductive endpoints have been evaluated, but do not appear to be particular targets of glyphosate toxicity.
- Cancer: Upon evaluation of available carcinogenicity studies in laboratory rodents, multiple agencies or organizations have concluded that glyphosate technical does not appear to be an animal carcinogen. In contrast, International Agency for Research on Cancer (IARC) considered the animal data to provide “sufficient evidence” of glyphosate carcinogenicity.
Body weight
Evidence of treatment-related effects on body weight among laboratory animals receiving lower oral doses of glyphosate or glyphosate-based herbicides varied by study. Studies using doses of glyphosate at ≤1,000 mg/kg/day during acute-, intermediate-, or chronic-duration exposure found no effects on body weight 140, 141, 142, 143, 144, 145. In contrast, body weight changes were sometimes observed in lower dose oral studies using glyphosate formulations 146, 147, 148, 149, 150, 151.
Several studies evaluated effects of oral exposure to glyphosate formulations on body weight. Limited results indicate that mice may be more sensitive than rats to body weight effects from repeated oral exposure to glyphosate formulations. In intermediate studies on male mice, significantly reduced body weight gain (>70%) occurred at 250 mg/kg/day after 6-12 week exposure 146 and in offspring exposed in utero, during lactation and then orally from post-natal day 21 to 60 (14% less than controls) 148. However, in another intermediate oral study, pregnant C57B1/6 mice showed a 17% decrease in body weight and an approximate 25% decrease in body weight gain, while their male offspring exposed in utero and during lactation did not have significantly different body weight or body weight gain when compared with controls 151. Seriously-depressed mean body weight gain (60–66% less than controls) was reported for albino Swiss mice gavaged with Roundup Original® at 50 mg/kg/day for 15 days and approximately 10% body weight loss for mice dosed at 500 mg/kg/day 152. When compared to controls, male rats orally exposed to a range of concentrations of Roundup® for two weeks showed an estimated 37% decrease in body weight when exposed to 100 mg/kg/day and an estimated 33% decrease in body weight when exposed to 250 mg/kg/day. Pregnant rats fed 500 mg/kg/day Roundup via gavage for 7 days had 10% less body weight gain compared to controls, when exposed simultaneously with paraquat 147. In rats, 8-10% less body weight gain was seen after exposure to 126 mg/kg/day in feed for 60 days 149.
Oral exposure of rats to glyphosate at relatively high doses resulted in significant effects on body weight and/or body weight gain. Pregnant rats gavaged at 3,500 mg/kg/day during gestational days 6–19 exhibited as much as 28.5% lower mean body weight gain than controls 153. Body weight gain was 12–18% less than that of controls in two generations of parental male and female rats exposed via the diet for 14–19 weeks at 2,219 or 3,134 mg/kg/day, respectively 154. No treatment-related effects on body weight were seen among young female mice treated for 28 days at estimated doses up to 1,447.5 mg/kg/day 143. In 13-week oral studies, body weight and/or body weight gain among rats and mice at oral doses in the range of 2,273–11,977 mg/kg/day were 10–18% less than controls 155. In a 2-year study, female rats dosed at 1,183 mg/kg/day exhibited 13% lower mean body weight than controls at treatment week 81 156.
However, other studies found no effects of oral glyphosate exposure on body weight. No significant effects on body weight were observed among Wistar rats gavaged with Roundup at 56 or 560 mg/kg/day for up to 13 weeks 157, male mice orally exposed to Roundup for 35 days 158. Pregnant Wistar rats gavaged with Roundup at 1,000 mg/kg/day during gestational days 6–15 159, or maternal Wistar rats gavaged with Roundup at 50–450 mg/kg/day during gestation and lactation 160. No effects on body weight were observed among male Wistar rats gavaged with Roundup Transorb® at 250 mg/kg/day during postnatal days 23–53 161. After exposure to 1.75 mg/kg/day of glyphosate or Roundup Bioflow® during pregnancy and lactation, no weight changes were observed in Sprague-Dawley F0 females or their male offspring 145. At higher exposure levels to glyphosate formulations (3.69 mg/kg/day and 352.2 mg/kg/day) during pregnancy, F0 and F1 female Wistar rats showed no signs of glyphosate-associated weight changes 162.
Non-oral exposure to glyphosate and glyphosate formulations was not associated with changes in body weight 105. No significant body weight effects occurred in a 4-week inhalation study of rats intermittently exposed to Roundup at exposure levels as high as 360 mg/m3 (approximately 36 mg Roundup/m³) 163. A study on male albino rats intraperitoneally exposed to 269.9 mg/kg/day of Roundup for up to two weeks also found no changes in body weight 141. No significant treatment-related effects on body weight were observed among rabbits administered repeated skin applications of glyphosate at doses in the range of 100–5,000 mg/kg/application for 21 days 164. When mice were acutely exposed subcutaneously to 2 mg/kg body weight, no effects on body weight were seen 165, 166, 167. No effects on body weight were seen in male mice after intranasal exposure to 50 mg/kg/day for 3 times a week for 4 weeks 140.
Respiratory effects
Available data regarding respiratory effects in laboratory animals exposed to glyphosate are limited. In mice, a 50% decrease in relative lung weight was observed following exposure to 250 mg/kg/day for 12 weeks 146. No other observations were made in the lungs. Kumar et al. 168 reported an inflammatory respiratory response (evidenced by increased eosinophil and neutrophil counts, mast cell degranulation, and production of IL-33, TSLP, IL-13, and IL-5) in anesthetized mice exposed intranasally to glyphosate. Adam et al. 169 designed a study to evaluate the effects of glyphosate (200 mg/kg), glyphosate + polyethoxylated tallow amine (POEA) 200 and 100 mg/kg, respectively. Polyethoxylated tallow amine (POEA) alone (100 mg/kg), and Roundup in rats evaluated for 24 hours following intratracheal instillation 169. Control rats received normal saline. Obvious clinical signs of adverse lung effects and mortalities occurred in each group except the saline controls. The study authors stated that the pulmonary effects were more severe and lasted longer in rats treated with polyethoxylated tallow amine (POEA) alone or in combination with glyphosate compared to responses in glyphosate only-treated rats. These results suggest polyethoxylated tallow amine (POEA) was more acutely toxic than glyphosate to the lungs. No respiratory effects occurred in a 120-day study where rats were exposed to 250 mg/kg/day 170. No respiratory effects occurred in a 4-week study of rats intermittently exposed to Roundup at exposure levels as high as 360 mg/m3 (approximately 36 mg Roundup/m³) 163.
Gastrointestinal effects
Limited information was located regarding gastrointestinal effects in laboratory animals following oral exposure to glyphosate 105. Rats exposed daily for 6-12 weeks to glyphosate 250 mg/kg/day exhibited a decreased in total bacterial count in the gut 171. Lozano et al. 172 reported significant differences in microbiome genomic diversity, characterized by an increase in the Bacteroidetes family S24-7 and a decrease in Lactobacillaceae, between treated female rats exposed to 5,000 ppm of Roundup for 673 days when compared to control males, control females, and treated males. The study found that Roundup had a direct selective bactericidal action on isolated gastrointestinal strains 172. In a study designed to evaluate the effects of glyphosate technical (2,000 mg/kg), rats were administered glyphosate + polyethoxylated tallow amine (POEA) 2,000 and 1,000 mg/kg, respectively, polyethoxylated tallow amine (POEA) alone (1,000 mg/kg), or Roundup by gavage, followed by 24 hours of posttreatment observation 169. Control rats received normal saline. Two rats in the polyethoxylated tallow amine (POEA)-only treatment group died. Diarrhea was noted in all groups except the control group. The study authors stated that the groups given polyethoxylated tallow amine (POEA) or mixtures that included polyethoxylated tallow amine (POEA) experienced more rapid and severe diarrhea than those given glyphosate alone. These results suggest that polyethoxylated tallow amine (POEA) was more acutely toxic than glyphosate to the gastrointestinal system. Mao et al. 173 reported that Roundup added to the drinking water of rat dams from gestational day 6 through lactation and to F1 offspring up to PND 125 at a concentration designed to deliver a daily dose of 1.75 mg glyphosate/kg/day (the U.S. glyphosate ADI) resulted in modifications to the gut microbiota in early development, particularly among prepubertal rats.
Glyphosate oral exposure in laboratory animals found the most common effect was clinical signs of gastrointestinal disturbances. Such clinical signs are commonly observed in studies of laboratory animals receiving bolus gavage doses of test substances, in which cases the clinical signs may be at least partially the result of the method of gavage dosing. Diarrhea was observed among rats gavaged once with glyphosate technical at 2,000 mg/kg 174. Gastrointestinal disturbances (e.g., soft stool, diarrhea, few feces) were reported among pregnant rats gavaged at 3,500 mg/kg/day during gestational days 6–19 153 and pregnant rabbits gavaged at 350 mg/kg/day during gestational days 6–27 175 or 175 mg/kg/day during gestational days 8–20 144. A slight increase in observations of soft stool and/or diarrhea was noted in the rabbits dosed at 175 mg/kg/day 175. Soft stools were observed in rats exposed via the diet for 2 generations at concentrations resulting in estimated doses in the range of 2,219–2,633 and 3,035–3,134 mg/kg/day for parental males and females, respectively 154. Mao et al. 173 reported that glyphosate added to the drinking water of rat dams from gestational day 6 through lactation and to F1 offspring up to PND 125 at a concentration resulting in a daily dose of 1.75 mg/kg/day (the U.S. acceptable daily intake [ADI]) resulted in modifications to the gut microbiota in early development, particularly among prepubertal rats.
In a 2-year study of rats exposed via the diet 156, 176, inflammation of gastric squamous mucosa was observed in females at an estimated dose level of 457 mg/kg/day; there were no signs of gastrointestinal effects in males at estimated doses as high as 940 mg/kg/day. In another chronic-duration oral rat study 177, there were no signs of treatment-related gastrointestinal effects at the highest estimated dose level (31.45–34.02 mg/kg/day). No clinical signs or histopathological evidence of treatment-related gastrointestinal effects were seen among male or female mice exposed via the diet for 24 months at estimated doses as high as 4,945 and 6,069 mg/kg/day, respectively 178, 179. Increased incidence of exocrine hyperplasia in the pancreas was reported for male rats receiving glyphosate technical from the diet for up to 2 years at an estimated dose of 1,214 mg/kg/day 180. Increased severity of cytoplasmic changes in salivary gland cells (basophilia and hypertrophy of acinar cells in parotid and submandibular salivary glands) was reported for male and female rats receiving glyphosate from the diet for 13 weeks at 410 and 421 mg/kg/day, respectively 155 and other rats similarly treated at 300 mg/kg/day for up to 2 years 180. Similar effects on salivary glands were observed in male and female mice treated for 13 weeks at much higher doses (1,065 and 2,707 mg/kg/day, respectively; not observed at 507 and 753 mg/kg/day, respectively) 155. Although salivary gland cytoplasmic changes were noted in rats at doses <300 mg/kg/day as well, the changes were reported to be only of minimal or mild severity; therefore, they are not considered adverse effects. The toxicological significance of the glyphosate treatment-related effects on salivary glands is uncertain.
Liver effects
The potential for glyphosate to cause liver toxicity was evaluated in studies of rats and mice 105. In a 7-day study of pregnant rats, the liver had a 19-23% increase in thiobarbituric acid reactive substances (TBARS) levels in liver tissue compared to controls, following exposure to 500 mg/kg/day by oral gavage 147. In rats orally administered glyphosate for 28-days up to 10 mg/kg body weight per day, no treatment related findings were reported after gross necropsy. Furthermore, no significant differences in liver weights were reported between glyphosate treated groups and the control 181. However, reactive oxygen species (ROS) levels in the liver were significantly increased at 10 mg/kg body weight per day, while thiobarbituric acid reactive substances (TBARS) concentrations decreased at 0.5, 1.75 and 10 mg/kg body weight per day compared to controls. GHS levels in liver decreased by 22.7% and 27% at 1.75 and 10 mg/kg body weight per day concentrations, respectively 181. Similarly, GHS peroxidase (GSH-Px) activity in the liver was noticeably higher among rats exposed to 0.5, 1.75 and 10 mg/kg body weight per day glyphosate compared to controls. Elevated levels of GSH-Px is reflective of glyphosate inducing the antioxidant defense system in the liver 181.
In a 13-week rat dietary study of glyphosate, increases in liver weight and serum alanine aminotransferase (ALT) were observed in males at 1,678 mg/kg/day; increased liver weight and increased serum alkaline phosphatase (ALP), alanine aminotransferase (ALT) and bile acids were noted in females at 3,393 mg/kg/day 142. There were no indications of treatment-related liver effects among male and female rats treated via the diet for 2 generations at estimated doses as high as 1,234–1,273 mg/kg/day 142 or other rats treated for 2 years to doses as high as 940–1,183 mg/kg/day 156, 176. Male mice exposed via the diet for 13 weeks at doses ≥2,273 mg/kg/day exhibited increased mean relative liver weight (4–9% greater than controls) in the absence of histopathologic liver lesions; there were no effects on liver weight in similarly-treated female mice at doses up to and including 11,977 mg/kg/day 155. Male mice exposed via the diet for 2 years at an estimated dose of 4,945 mg/kg/day exhibited increased incidence of histopathologic central lobular hepatocyte necrosis; there was no evidence of treatment-related liver effects in similarly-treated female mice at an estimated dose of 6,069 mg/kg/day 179.
Following oral exposure to glyphosate (1.0 g/L in drinking water) for 72 weeks, wild type and multiple myeloma model mice (Vk*MYC strain) showed liver fibrosis and blood abnormalities including decreases in hemoglobin levels (i.e. anemia); lower levels of platelet counts and hematocrit were observed in model multiple myeloma mice 182. This study was part of a larger effort to understand the effect of glyphosate on multiple myeloma development 182.
Available information regarding liver endpoints in animals exposed to glyphosate formulations is limited. No liver effects occurred in a 4-week study of rats intermittently exposed to Roundup at exposure levels as high as 360 mg/m³ (approximately 36 mg Roundup/m³) 163. Increased serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activity and histopathologic liver lesions (increased Kupffer cells in hepatic sinusoids and deposition of reticulin fibers) were seen in male rats treated with Glyphosate-Biocarb® by gavage for 75 days (one dose every 2 days) at 487 mg/kg/dosing 183. In an acute oral exposure study, a significant increase in liver index, liver weight, and cytokine expression (IL-1β and TNF-α) was observed in male rats exposed to 100 or 250 mg/kg/day Roundup. Lower doses of Roundup, including exposure to 25 or 50 mg/kg/day, resulted in increased liver weight, higher numbers of liver macrophages, and changes in glycogen storage. However, these results were less consistent and did not adhere to a dose-response relationship 150. Following a 120-day exposure to 500 mg/kg/day Roundup, rats exhibited increased mean liver malondialdehyde levels, decreased superoxide dismutase concentration, catalase activity, increased alanine aminotransferase, aspartate amino transferase, lactate dehydrogenase and enzymatic activity of GSH-Px, moderate hepatocytic degeneration and necrosis 170. Following 6-12 weeks of daily exposure to ≥250 mg/kg/day of Roundup ®, mice showed a 44% decrease in relative liver weight, no other liver observations were made 146.
Rabbits administered repeated skin applications of glyphosate at doses in the range of 100–5,000 mg/kg/application for 21 days exhibited no evidence of treatment-related liver effects 164. However, male albino rats intraperitoneally exposed to 134.95 mg/kg/day glyphosate for up to two weeks showed significantly increased serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP), decreased serum GSH, increased hepatic nitric oxide, and increased hepatic lipid peroxidation, which the author attributed to impairment of liver enzymes and hepatic metabolism 141.
Two-week intraperitoneal exposure of albino rats to 269.9 mg/kg/day Roundup® demonstrated the same liver effects as intraperitoneal exposure to 134.95 mg/kg/day glyphosate technical, including increased serum AST, ALT, and ALP, decreased serum GSH, increased hepatic nitric oxide, and increased hepatic lipid peroxidation. Most of these observed effects were similar in both Roundup®-exposed and glyphosate-exposed rats. However, compared to controls, rats exposed to glyphosate technical showed a larger increase in hepatic nitric oxide than rats exposed to Roundup® (72% increase and 21% increase respectively). Conversely, the increase in hepatic lipid peroxidation compared to controls was much more pronounced in Roundup ®-exposed rats than in glyphosate-exposed rats (630% increase and 432% increase respectively) 141.
In vivo metabolome and proteome profiling of liver obtained from rats chronically exposed to long-term exposure at low levels of Roundup (4 ng/kg bw/day) for two years indicate effects to the liver including metabolite alterations associated with non-alcoholic fatty liver disease and steatohepatosis 184. Metabolome profiling, or the analysis of metabolites characterizing the range of chemical processes, analogous to chemical fingerprinting, revealed a lipotoxic condition, oxidative stress, and markers of hepatotoxicity in the liver 184. Results from the proteome analysis, which characterizes the expression of protein products and their interaction, reported rats exposed to Roundup ® had alterations reflective of peroxisomal proliferation, steatosis, and necrosis 184.
Kidney effects
Several studies evaluated possible kidney toxicity in laboratory animals treated with glyphosate 105. In a 2-generation reproductive toxicity study 142, slightly increased absolute and relative kidney weights (7–11% greater than controls) were reported among female rats dosed at 1,273 mg/kg/day; there was no evidence of histopathologic kidney lesions 105. Therefore, the slightly increased kidney weight was not considered to represent an adverse effect 105. During 2 years of glyphosate dietary treatment of rats, urinalysis revealed increased specific gravity of urine and decreased urinary pH among males treated at an estimated dose of 940 mg/kg/day (no-observed-adverse-effect level (NOAEL) =362 mg/kg/day); there were no signs of treatment-related renal effects in urinalysis results from females treated at an estimated dose as high as 1,183 mg/kg/day 156, 176. Papillary necrosis (males and females) and decreased pH of urine (males only) were observed in a study of rats administered glyphosate in the diet for up to 2 years at estimated doses of 1,214 mg/kg/day (males) and 1,498 mg/kg/day (females); respective no-observed-adverse-effect levels (NOAELs) were 361 and 437 mg/kg/day 180. Another 2-year rat study reported decreased pH of urine among males treated at 1,000 mg/kg/day (NOAEL=300 mg/kg/day); no kidney effects were observed in females at doses as high as 1,000 mg/kg/day 180. Female mice treated for 2 years at an estimated dose of 6,069 mg/kg/day exhibited significantly increased incidence of renal proximal tubule epithelial basophilia and hypertrophy (NOAEL=968 mg/kg/day); there was no evidence of renal effects in similarly-treated male mice at doses as high as 4,945 mg/kg/day 179.
Shorter-term studies on rodents exposed to glyphosate found signs of potential kidney damage. Myeloma model mice orally exposed to glyphosate (1.0 g/L in drinking water) for 72 weeks showed kidney damage, including tubular obstruction by casts 182. This study was part of a larger effort to understand the effect of glyphosate on myeloma development 182. In a two-week study albino rats exposed intraperitoneally to 134.95 mg/kg/day glyphosate showed serum changes indicative of potential kidney effects 141. Compared to control rats, glyphosate-exposed rats had 33.8% decreased serum creatinine, 110% increased serum urea, and 146% increased uric acid. Similar results were seen in the male rats exposed to 269.9 mg/kg/day Roundup 141.
A multigenerational study on reproductive effects measured dam kidney weight and found no difference between controls and dams exposed to 420 mg/kg/day of Roundup 151. In mice, decreased relative kidney weight (50% less than controls) was reported after daily exposure to ≥250 mg/kg/day for 12 weeks 146. No other kidney observations were made. No kidney effects occurred in a 4-week study of rats intermittently exposed to Roundup at exposure levels as high as 360 mg/m³ (approximately 36 mg Roundup/m³) 163. Histopathologic kidney lesions (necrotic and apoptotic cells, localized primarily in tubular epithelium of the proximal straight tubule and thick ascending limb of the loop of Henle) were reported in male rats gavaged once with Concentrate Roundup Weedkiller at dose levels ranging from 250 to 2,500 mg/kg 185. There is some uncertainty regarding the role of glyphosate in the reported effects.
Hematological effects
Available information regarding hematological effects related to glyphosate formulations is limited. No hematological effects occurred in a 4-week study of rats intermittently exposed to Roundup at exposure levels as high as 360 mg/m3 (approximately 36 mg Roundup/m³) 163. Decreases in red blood cell count, hematocrit, and hemoglobin, and increases in corpuscular volume and neutrophil count were reported in mice gavaged with Roundup Original for 15 days at 500 mg/kg/day 152.
There were no apparent treatment-related hematological effects in chronic-duration oral studies of rats, mice, or dogs administered glyphosate at oral doses as high as 940–1,183 mg/kg/day for rats 156, 176, 177, 4,945–6,069 mg/kg/day for mice 179 and 500 mg/kg/day for dogs 186, 187. Rabbits administered repeated dermal applications of glyphosate technical at doses in the range of 100–5,000 mg/kg/application for 21 days exhibited no evidence of treatment-related hematological effects 164. Small changes in hematological parameters were seen in both male and female rats following dietary exposure to glyphosate technical in the 13-week National Toxicology Program study 155. These were considered to be unremarkable and most likely due to mild dehydration 105.
Skin effects
Minor skin irritation was reported in response to topically-applied glyphosate 105. At the application site, very slight redness and swelling were observed in rabbits during 21 days of repeated dermal application of glyphosate at 5,000 mg/kg/application; no skin effects were seen at doses ≤1,000 mg/kg/application 164.
Eye effects
Two chronic-duration oral studies included ophthalmoscopic examinations of laboratory animals exposed to glyphosate. The Environmental Protection Agency 156, 176 reported significantly increased incidence of lens abnormalities in male rats treated via the diet for 2 years at an estimated dose of 940 mg/kg/day; there were no indications of a treatment-related ocular effect in female rats at the highest estimated dose level (1,183 mg/kg/day). No signs of treatment-related ocular effects were seen among dogs treated via capsule for 1 year at estimated doses as high as 500 mg/kg/day 186.
According to Food and Agriculture Organization of the United Nations and World Health Organization 188, glyphosate was moderate to severely irritating to the rabbit eye. There were no signs of exposure-related effects in ophthalmologic examinations of rats intermittently exposed to Roundup for 4 weeks at exposure levels as high as 360 mg/m³ (approximately 36 mg Roundup/m³) 163.
Endocrine effects
Limited information was located regarding the potential for glyphosate to affect the endocrine system in animal studies. While one study reported degeneration of pancreatic acinar cells and islets of Langerhans after male Wistar rats were exposed to 14.4, 375, or 750 mg/kg/day of the herbicide Bushfire via drinking water 189, most of the observed endocrine effects in glyphosate animal studies involved changes in hormone levels. Romano et al. 161 reported dose-related 30–50% decreased serum testosterone in young male rats gavaged with Roundup Transorb® at 5–250 mg/kg/day during postpartum days 23–53. Romano et al. 190 implicated disruption of gonadotropin expression as a mechanism of action for glyphosate-induced effects on male rat sexual development. Pregnant C57B1/6 mice exposed to 420 mg/kg/day Roundup in their drinking water showed no difference in pancreas weight compared to controls, but their male F1 offspring had 195% higher intratesticular testosterone, an estimated 60% increase in plasma LH, and a 11% increase in β-LH pituitary protein compared with male F1 controls 151.
A multi-generational study of F0 Sprague-Dawley rats exposed to either 1.75 mg/kg/day glyphosate or Roundup Bioflow® found no evidence of endocrine organ lesions in F1 offspring for either exposure 145. However, exposure to Roundup Bioflow® was associated with changes in hormone ratios in F1 offspring when compared to controls. Specifically, after 13 weeks of exposure to Roundup Bioflow® in addition to exposure during gestation and lactation, female F1 rats had increased serum total testosterone (TT), decreased 5α-dihydrotestosterone (DHT)/TT ratios, and increased 17β-estradiol (E2)/TT ratios, while male F1 rats had decreased serum DHT and increased plasma thyroid stimulating hormone (TSH) 145. A 12-week study on male albino rats orally exposed to Roundup® also found changes in hormone levels (13% decrease in testosterone, 17% decrease in follicle stimulating hormone, approximately 14% decrease in luteinizing hormone, and approximately 31% increase in prolactin) at doses as low as 3.6 mg/kg/day 191.
However, in an exposure study that exposed male Sprague-Dawley rats to either glyphosate (2.5 mg/kg/day or 25 mg/kg/day) or the glyphosate formulation Glyfonova (25 mg/kg/day) via their drinking water, no changes in intratesticular testosterone levels were observed 192. While the rats exposed to Glyfonova showed a statistically significant upregulation in P450 encoding genes in the testes, the authors concluded this was not indicative of endocrine effects 192. Nevertheless, other studies reported changes in gene and protein expression attributed to glyphosate exposures 190, 151, 193.
Immunological effects
There was no evidence of treatment-related effects on spleen or thymus of mice administered glyphosate in their diet for 28 days at estimated doses as high as 1,447.5 mg/kg/day and no evidence of treatment-related effects on splenic anti-sheep red blood cell (anti-SRBC) anti-body forming cell (AFC) responses to sheep red blood cell (SRBC) 143. The Environmental Protection Agency 177 reported significantly increased incidences of lymphocytic hyperplasia in the thymus from female rats administered glyphosate technical in the diet for up to 26 months at doses of 3.37, 11.22, and 34.02 mg/kg/day (13/32, 18/37, and 17/34, respectively, versus 5/25 controls). However, the Environmental Protection Agency 177 did not consider the lesion to be compound-related because the lesion occurs spontaneously in older rats and is quite variable in the thymus, there was no apparent effect on lymphocytes in the spleen (a much less variable indicator for lymphocytic hyperplasia), and the severity of the lesion was similar among controls and glyphosate-treated groups. Kumar et al. 168 reported an inflammatory respiratory response (evidenced by increased eosinophil and neutrophil counts, mast cell degranulation, and production of interleukin-33, thymic stromal lymphopoietin, interleukin-13, and interleukin-5) in anesthetized mice exposed intranasally to glyphosate.
Neurological effects
Several animal studies in rats and mice have evaluated the neurological effects of glyphosate. Mice exposed once to 250 mg/kg/day Roundup via gavage showed a decrease in aversive memory performance 194. However, no neurological effects were seen in mice given ≤500 mg/kg/day once by oral gavage 146, 171. At 6-12 weeks of daily exposure, mice showed behavioral changes in locomotor activity, increase of anxiety and depression-like behavior levels, decreased memory performance and decreased grooming time with 250 mg/kg/day RoundUp exposure 146, 171, 194. The observed neurobehavioral changes were attributed to neurodevelopmental impairment as evidenced by a reduction in serotonin (5-HT) immunoreactivity following 6 or 12 weeks of oral exposure to 250 or 500 mg/kg/day RoundUp and a reduction in tyrosine hydroxylase (TH) immunoreactivity following 12 weeks of exposure to 250 or 500 mg/kg/day RoundUp 146.
Ait Bali et al. 194, measured acetylcholinesterase activity in rat whole brain, prefrontal cortex and hippocampus, which was inhibited at ≥250 mg/kg/day by >25%, >35%, >25%, respectively. Inhibition in all measured parts was dose-dependent194. In pregnant rats, glutathione was decreased by 16-26% following exposure to 500 mg/kg/day RoundUp on days 1 to 7 of pregnancy 147. In male offspring exposed in utero, during lactation then from post-natal day 21 to 60 to 70 mg/kg/day RoundUp, there was a 23% inhibition of hippocampus cholinesterase after post-natal day 60 148. Oxidative damage, astrocyte dysfunction, glutamate excitotoxicity, and misregulation of cholinergic transmission were also seen 195.
In rats treated via the diet for 13 weeks at doses as high as 1,547–1,631 mg/kg/day and in rats administered glyphosate technical once by gavage at up to 2,000 mg/kg, there was no evidence of treatment-related neurotoxicity seen by functional observational battery, motor activity testing, and gross and histopathologic examination of brain and peripheral nervous tissue 174. However, clinical signs included decreased activity, subdued behavior, and hunched posture.
Glyphosatewas observed to affect monoaminergic neurotransmitters in the central nervous system. In rats administered glyphosate orally up to 800 mg/kg bw/day for 6 days, serotonin neurotransmitter levels were significantly decreased in a dose dependent manner at 75, 150 and 800 mg/kg body weight in various regions of the brain including the striatum, hippocampus, prefrontal cortex, hypothalamus and midbrain region 196. Similarly, dopamine and norepinephrine levels were reported to decrease with increasing concentrations of glyphosate starting at 75 mg/kg body weight per day. Turnover rates of the neurotransmitters were measured, and their metabolites were altered; there was a significant increase in turnover between serotonin and dopamine, and a decrease in turnover with norepinephrine 196.
Rats orally exposed to 5 mg/kg/day of glyphosate or glyphosate-based formulation perinatally from day 9 gestation to day 22 post-natal were found to have increased expression of synaptophysin a marker of synaptic terminals in the hippocampus of both groups 197. Maternal behavior was also observed; at day 1 post-natal, dams were observed to spend less time licking and grooming offspring whereas between day 2 and 6 post-natal, more time was spent with offspring.
Test tube studies have also examined glyphosate and glyphosate-based herbicides for neurotoxicity. Glyphosate and an herbicide containing glyphosate isopropylamine as its active ingredient were tested in vitro at concentrations of 0.005% to 0.0005%. Although no effect was observed for pure glyphosate, glyphosate-based herbicides were reported to interfere with myelination and also as a demyelinating agent in a dose-dependent manner 198. However, after testing for demyelination using glyphosate and isopropylamine additively (rather than as formulated), the authors note that this effect may be due to undisclosed additives 198. Neither glyphosate (pure) nor glyphosate-based herbicide were found to impair neurite development 198.
Glyphosate and cancer
Animal and human studies were evaluated by regulatory agencies in the USA, Canada, Japan, Australia, and the European Union, as well as the Joint Meeting on Pesticide Residues of the United Nations and World Health Organization (WHO). These agencies looked at cancer rates in humans and studies where laboratory animals were fed high doses of glyphosate. Based on these studies, they determined that glyphosate is not likely to be carcinogenic 199, 200, 201, 202, 188, 203, 204, 205, 206. However, in 2015, a committee of scientists working for the International Agency for Research on Cancer (IARC) of the World Health Organization (WHO) classified glyphosate as “probably carcinogenic to humans” (Group 2A) 86, 87. According to the International Agency for Research on Cancer (IARC) there was “limited evidence of carcinogenicity in humans for non-Hodgkin lymphoma” 86. The evidence in humans is from studies of exposures, mostly agricultural, in the USA, Canada, and Sweden published since 2001 88, 89, 90, 91. In addition, there is convincing evidence that glyphosate also can cause cancer in laboratory animals as well as strong evidence that exposure to glyphosate is genotoxic and can induce oxidative stress in experimental animals and in humans in vitro 86. “Limited evidence” means that a positive association has been observed between exposure to glyphosate and cancer but that other explanations for the observations called chance, bias, or confounding could not be ruled out. Group 2A category is also used when there is limited evidence of carcinogenicity in humans and strong data on how the agent causes cancer 86.
Glyphosate also caused DNA and chromosomal damage in human cells, although it gave negative results in tests using bacteria. One study in community residents reported increases in blood markers of chromosomal damage (micronuclei) after glyphosate formulations were sprayed nearby 86. The International Agency for Research on Cancer (IARC) also concluded that there is strong evidence for glyphosate-induced oxidative stress based on results from studies of animal models in vivo and human cells in vitro 207.
In 2005, an evaluation of glyphosate and cancer risk was conducted in the Agricultural Health Study 92. This evaluation considered glyphosate use reported at enrollment (1993–1997), and included 2088 cancers diagnosed between enrollment and 2001. No statistically significant associations were found for any cancer sites, including non-Hodgkin lymphoma, but there was an increased risk, though not statistically significant, of multiple myeloma in the highest exposure category based on a small number of cases 92.
In this large 2017 prospective cohort study 93, no association was apparent between glyphosate and any solid tumors or lymphoid malignancies overall, including non-Hodgkin lymphoma and its subtypes. There was some evidence of increased risk of acute myeloid leukemia (AML) among the highest glyphosate exposed group that requires confirmation 93.
Glyphosate and death
Several case report have reported deaths in individuals intentionally ingesting glyphosate products 208, 209, 210, 211, 212, 213, 214. The main cause of death from glyphosate ingestion was shock (hypovolemic or cardiogenic), low blood pressure (hypotension) and respiratory failure often due to aspiration 208, 209, 210, 213. Among 107 people who ingested glyphosate isopropylamine salt, eleven fatalities were reported (10.3% rate) while none were observed in the glyphosate ammonium salt group, for which there were 40 patients 210. A retrospective cohort study reported that 14 of 150 patients with glyphosate surfactant herbicide poisoning died and that non-surviving patients were statistically older compared to surviving patients 215.
LD50 also called “median lethal dose” and refers to the amount of a substance that is lethal to half of a group of test animals. LD50 is a way to measure how toxic a material is in the short term, or its acute toxicity. An acute oral glyphosate LD50 value of 4,320 mg/kg/day was reported following single oral dosing of rats with glyphosate 216. In a developmental toxicity study, 6/25 pregnant rats died during oral dosing of glyphosate at 3,500 mg/kg/day; there were no deaths during treatment at 1,000 mg/kg/day 153. No adequate sources were located regarding death in laboratory animals exposed to glyphosate by inhalation or dermal routes 105.
In a study of female Wistar rats exposed to 126 or 315 mg/kg/day of a glyphosate for 60 days, 6/12 rats died 149. In a study that employed oral dosing of pregnant rats with Roundup, 8/15 dams died during the first 8 days of treatment at 1,000 mg/kg/day glyphosate 159. No deaths occurred in a 4-week study of rats intermittently exposed to Roundup at exposure levels as high as 360 mg/m3 (approximately 36 mg Roundup/m³) 163. No adequate sources were located regarding death in laboratory animals exposed to glyphosate formulations by the skin route.
Glyphosate and respiratory effects
The Agricultural Health Study participants have examined the possible associations between use of glyphosate-containing products and increased risk of rhinitis, wheezing, atopic asthma, allergic asthma, or chronic bronchitis 108, 110, 109, 217, 112, 113, 115, 116. No associations were found for diagnosed chronic bronchitis 217 or for wheezing after adjusting for confounding exposure to other pesticides 108, 110, 109. Current rhinitis was associated with glyphosate use among commercial applicators 115 and farmers 116, but no relationship between risk and the number of days of use per year was found among the commercial applicators 115. The relationship seen in commercial applicators was limited to applicators that also reported using 2,4- dichlorophenoxyacetic acid (2,4-D) herbicide. Applicators using 2,4- dichlorophenoxyacetic acid (2,4-D) herbicide or glyphosate alone did not show an increased risk of rhinitis 115. An association between glyphosate use and the risk of atopic asthma was found among farm women, but there was no association with nonatopic asthma 112. No associations were found between glyphosate use by male farmers and risk of allergic or nonallergic asthma 113. An association between glyphosate use and the risk of both allergic and nonallergic wheeze was found among male farmers, with an increase in risk for allergic wheeze with increasing days of use per year 114. It is noted that many of these studies did not account for use of other pesticides 105.
A study by Camacho and Mejia 122 investigated the association between aerial applications of glyphosate in Colombia and health effects of individuals living in the sprayed areas. The association was assessed using individual medical record data and information about Colombia’s aerial spraying program. Several health outcomes including respiratory illnesses was examined and a positive association was observed. For each additional square kilometer increase in area sprayed with glyphosate there was an increase in the proportion of respiratory diagnoses 7 to 15 days following exposure 122.
Respiratory failure or distress was reported in about 10–25% of the cases of intentional ingestion of glyphosate products 218, 219, 220, 214. Cho et al. 215 found that the most common complication occurring in patients who had ingested glyphosate surfactant herbicide was acute respiratory distress syndrome (ARDS).
Glyphosate and cardiovascular effects
Two studies of Agricultural Health Study participants did not find associations between the use of glyphosate-containing products and the risk of myocardial infarctions (Dayton et al. 2010; Mills et al. 2009); see Table 2-5 for details. An association was found between using a glyphosate-based herbicide and vasculitic neuropathy in a 70 year old man who sprayed approximately 2,000 mL of the herbicide for several hours without using protective gear 4 months before presenting with symptoms (Kawagashira et al. 2017). In case series reports, abnormal electrocardiogram (EKG) readings have been found in patients ingesting large doses of glyphosate-containing products (Kim et al. 2014; Lee et al. 2000, 2008; Moon and Chun 2010; Moon et al. 2018; Talbot et al. 1991). The most commonly reported alterations included prolonged QTc interval and sinus tachycardia. In the most severe poisoning cases, hypotension and shock have been reported (Picetti et al. 2018; Roberts et al. 2010; Sawada et al. 1988; Tominack et al. 1991). Additionally, adverse cardiovascular events (myocardial injury, shock, ventricular dysrhythmia, or cardiac arrest) have been reported among patients who ingested glyphosate (Moon et al. 2018).
No data were available regarding evaluation of cardiovascular endpoints in laboratory animals exposed to glyphosate technical or glyphosate formulations by any exposure route.
Glyphosate and gastrointestinal effects
Gastrointestinal symptoms are commonly reported in case series reports of patients who ingested glyphosate products 105. In numerous reports, over 40% of the patients reported nausea/vomiting 221, 222, 219, 211, 223. Other gastrointestinal effects reported included abdominal pain 218, 211, 212, 220, 213, sore throat 220, 214, poor appetite 222 and damage to mucosal tissue in the mouth and esophagus 219, 212, 224, 213, 214. One case study reported gastric ulcer and a large pyloric antrum ulcer 222. In a woman who accidentally ingested glyphosate-surfactant herbicide, a CT scan showed aspiration pneumonitis and ileus of the intestine 225.
Glyphosate and hematological effects
After the accidental ingestion of 100 mL of glyphosate-surfactant herbicide a woman exhibited hypoxemia, hyperkalemia, hypotension, and hemoconcentration 225.
Glyphosate and liver effects
Human studies evaluating the relationship between glyphosate and liver endpoints is limited 105. One retrospective cohort study reported acute liver failure as a complication associated with organ injury 215. In a case-control study evaluating the association between glyphosate and patients with nonalcoholic steatohepatitis (NASH), Mills et al. 121 reported significantly higher levels of glyphosate residue in nonalcoholic steatohepatitis (NASH) patients compared to non-NASH patients. Furthermore, a dose-dependent increase of glyphosate exposure was observed with advanced stages of fibrosis (stage 2, 3 or 4). No other information was located regarding liver effects in humans exposed to glyphosate-containing products 105.
Glyphosate and kidney effects
Available human data regarding the possible association between exposure to glyphosate and risk of kidney effects is limited to a few case-control studies, two case reports and one retrospective cohort study 105. One case-control study of patients with chronic kidney disease (CKD) found an increased risk of chronic kidney disease among glyphosate applicators 226. However, uncertainty regarding an association between exposure to glyphosate and risk of chronic kidney disease includes the finding that the glyphosate applicators were also exposed to high levels of calcium, magnesium, barium, strontium, iron, titanium, and vanadium by drinking water from abandoned wells 105. In the case of a 55 year old man who ingested 200 mL of a glyphosate mixture, acute renal failure occurred 219. Acute kidney injury and metabolic acidosis occurred in a woman who accidentally ingested glyphosate-surfactant herbicide 225. Acute kidney injury associated with glyphosate-based herbicide ingestion was also reported in a retrospective cohort study as a complication associated with organ injury 215.
Glyphosate and rheumatoid arthritis
De Roos et al. 119 did not find an association between glyphosate use and the risk of rheumatoid arthritis among participants of the Agricultural Health Study. In a subsequent study of female spouses of licensed pesticide applicators, Parks et al. 120 reported a weakly positive association between spousal use of glyphosate and risk of rheumatoid arthritis (based on incident cases).
Glyphosate and skin effects
One woman developed skin lesions after accidental dermal exposure to glyphosate in an herbicide and was diagnosed with toxic hand dermatitis 227. After treatment did not fully resolve the lesions, she was diagnosed with koebnerization of psoriasis caused by acute irritant contact dermatitis. According to the Environmental Protection Agency 228, glyphosate is considered a slight skin irritant following acute skin application.
In another study, Camacho and Mejia 122 evaluated the association between aerial applications of glyphosate in Colombia and health effects of individuals living in the sprayed areas. The association was assessed using individual medical record data and information about Colombia’s aerial spraying program. Several health outcomes including dermatological effects. Their results observed that for each additional square kilometer increase in area sprayed with glyphosate there was an increase in the proportion of skin disorders 7 to 15 days following exposure 122.
One study evaluated the potential skin toxicity of glyphosate in humans. In an experimental study 123, a single application of Roundup to intact skin for 24 hours did not result in irritation. When applied to abraded skin, redness was noted in 42% of the subjects after 24 hours. Mild skin irritation was observed in a repeated glyphosate exposure test study 123. No skin irritation was observed in a Draize skin sensitization test or in a photosensitivity/photoirritation test 123.
Glyphosate and eye effects
In a study of wives of commercial pesticide applicators, no association was found between glyphosate use among the wives and retinal degeneration 124. In a case series report of 1,513 eye exposures to glyphosate, minor symptoms (primarily transient irritation) were observed in 70% of the cases; most (99%) complained of eye pain 229. Moderate effects, such as persistent irritation or low-grade corneal burns or abrasions, were observed in about 2% of the cases 229. Among the cases with moderate effects, 93% reported eye pain, 20% reported lacrimation, and 27% reported blurred vision 229. According to the Environmental Protection Agency 228, glyphosate is considered mildly irritating to the eye following ocular instillation. The European Food Safety Authority 201 stated that glyphosate acid was a severe eye irritant, but that salts of glyphosate do not require classification as eye irritants.
Glyphosate and endocrine effects
Available human information regarding possible associations between exposure to glyphosate or glyphosate-containing products and risk of endocrinological effects is limited to 3 studies with conflicting results 105. Goldner et al. 125 reported no associations between any glyphosate exposure and the risks of thyroid diseases. Shrestha et al. 127 found a significant association between ever-use of glyphosate and hypothyroidism (underactive thyroid) (hazard ratio = 1.28). Kongtip et al. 126 reported a positive association between increasing use of glyphosate and increased T4 thyroid hormone levels.
To evaluate the potential for glyphosate to affect the endocrine system, the Environmental Protection Agency 230 subjected glyphosate to the Endocrine Disruptor Screening Program Tier 1 (a battery of in vitro assays designed assist in evaluation of the potential for a substance to interact with estrogen, androgen, or thyroid signaling pathways). Environmental Protection Agency evaluated results from the battery of in vitro assays and relevant laboratory mammalian and wildlife studies. Using this approach, EPA determined that there is no convincing evidence of potential interaction between glyphosate and estrogen, androgen, or thyroid pathways in mammals or wildlife 230. Included in the evaluation of the estrogen pathway were estrogen receptor (ER) binding assays, an ER transactivation assay, aromatase and steroidogenesis assays, a fish short-term reproduction assay, and mammalian and wildlife studies that assessed female reproductive parameters. Included in the evaluation of the androgen pathway were androgen receptor (AR) binding and steroidogenesis assays, a fish short-term reproduction assay, Hershberger and male pubertal assays, an androgen receptor (AR) transactivation assay, and mammalian and wildlife studies that assessed male reproductive parameters. Included in the evaluation of the thyroid pathway were male and female pubertal assays, an amphibian metamorphosis assay, and mammalian and wildlife studies that assessed thyroid parameters 230, 231.
Glyphosate and reproductive effects
No association between glyphosate use and fecundability (the likelihood of becoming pregnant during a menstrual cycle or within a month) was found among women living at farms in which pesticides were used and were involved in pesticide activities 232. This study also reported an association with improved fecundability when the women were not involved in pesticide activities 232. Sanin et al. 233 examined the fertility of women living in regions of Columbia with varying agricultural usage of glyphosate. Although time to pregnancy varied widely by region, no significant associations were found with level of glyphosate usage.
In another study, Camacho and Mejia 122 evaluated the association between aerial applications of glyphosate in Colombia and miscarriages by women living in the sprayed areas. Their results observed a positive association. For each additional square kilometer increase in area sprayed with glyphosate there was an increase in the number of miscarriage diagnoses. However, the authors note the way miscarriage was defined in the study may overestimate the number of actual miscarriages 122. In the study, miscarriage was defined when a mother was observed to have attended a prenatal care visit, but a corresponding birth registry was not located after 10 months 122.
Glyphosate and neurological effects
Available information regarding possible associations between exposure to glyphosate-containing products and risk of neurological effects in humans is limited. One case-control study did not find an association between glyphosate exposure and Parkinson’s disease 128. In a cross-sectional study conducted on farmers in China, Zhang et al. 129 reported glyphosate use was not associated with increased odds or incidence rates of abnormalities of peripheral nerve conduction.
One case study found that a man who ingested 200 ml of glyphosate developed Parkinson’s symptoms 4 years after exposure 221. A spatial analysis of exposure to pesticides in Washington State found an association between glyphosate exposure and increased odds of premature mortality attributable to Parkinson’s disease (odds ratio = 1.33) 234.
Test tube studies have also examined glyphosate and glyphosate-based herbicides for neurotoxicity. Glyphosate and an herbicide containing glyphosate isopropylamine as its active ingredient were tested in test tube at concentrations of 0.005% to 0.0005%. Although no effect was observed for pure glyphosate, glyphosate-based herbicides were reported to interfere with myelination and also as a demyelinating agent in a dose-dependent manner 198. However, after testing for demyelination using glyphosate and isopropylamine additively (rather than as formulated), the authors note that this effect may be due to undisclosed additives 198. Neither glyphosate (pure) nor glyphosate-based herbicide were found to impair neurite development 198.
Glyphosate and developmental effects
Several epidemiology studies have examined possible associations between glyphosate use and developmental toxicity. Given that only one study examined each endpoint and the lack of quantification of glyphosate exposure across studies, these results were not considered sufficient for drawing conclusions on the risk of developmental toxicity associated with glyphosate exposure in humans 105. Arbuckle et al. 131 reported a positive association between maternal preconception exposure to glyphosate and increased risk of spontaneous abortion (miscarriage). Garry et al. 133 reported a positive association between phosphonamino- herbicides (glyphosate, Roundup) exposure and parent-reported attention deficit disorder/attention deficit hyperactivity disorder (ADD/ADHD). No associations were found between paternal exposure and risk of miscarriages, preterm delivery, small for gestational age risk 137, or congenital malformations 132. Similarly, no associations were found between maternal glyphosate exposure and birth weight 136, 134, neural tube defects 135 or head circumference 134.
Genetically modified food and potential to cause allergy
Potential allergenicity (potential to cause allergy) to the novel gene product is another commonly expressed concern. Allergies to non-GM foods such as soft fleshed fruit, potatoes and soy are widespread. Clearly, new varieties of crops produced by either GM techniques or conventional breeding both have the potential to be allergenic 235. Concern surrounding this topic relates to two factors; the possibility that genes from known allergens may be inserted into crops not typically associated with allergenicity and the possibility of creating new, unknown allergens by either inserting novel genes into crops or changing the expression of endogenous proteins. Assessment of the allergenic potential of compounds is problematic and a number of different bodies have produced guidelines and decision trees to experimentally evaluate allergenic potential 236, 237, 238. These are effective at assessing compounds which may prove to be hazardous through a hierarchical approach which includes determining whether the source of the introduced gene is from an allergenic plant, whether GM foods react with antibodies in the sera of patients with known allergies and whether the product encoded by the new gene has similar properties to known allergens. In addition, animal models are used to screen GM foods 239. Tests are not performed to formally assess any risk posed by inhalation of pollens and dusts; however, this is not assessed for conventionally grown foods and feeds either, and no allergies have been attributed to commercially grown GM pollen to date.
Two examples are frequently quoted regarding GM crop allergenicity:
- A project to develop genetically modified peas by adding a protein from beans that conferred resistance to weevils was abandoned after it was shown that the GM peas caused a lung allergy in mice 240
- Soya bean engineered to express a Brazil nut protein was withdrawn from production after it was also found to be allergenic in tests 241
Opponents of GM technology often cite these examples as proof that it is inherently unpredictable and dangerous, although another interpretation would be to say that safety testing of GM plants was effective in both cases, having identified allergenic potential before either product was released to market. Moreover, GM technology might also be used to decrease the levels of allergens present in plants by reducing expression levels of the relevant genes. For example, research was recently undertaken to identify an allergen in soybeans and remove it using GM technology 242.
Genetically Modified Food and Gene transfer studies in human volunteers
There has only been one human feeding study conducted on the effects of GM foods. The study involved seven human volunteers who previously had their large intestines removed for medical reasons. These volunteers were provided with GM soy to eat to see if the DNA of the GM soy transferred to the bacteria that naturally lives in the human gut. Researchers identified that three of the seven volunteers had transgenes from GM soya transferred into the bacteria living in their gut before the start of the feeding experiment. As this low-frequency transfer did not increase after the consumption of GM soy, the researchers concluded that gene transfer did not occur during the experiment. In volunteers with complete digestive tracts, the transgene did not survive passage through intact gastrointestinal tract 243. Two studies on the possible effects of giving GM feed to animals found that there were no significant differences in the safety and nutritional value of feed stuffs containing material derived from GM plants 244, 245. Specifically, the studies noted that no residues of recombinant DNA or novel proteins have been found in any organ or tissue samples obtained from animals fed with GM plants 246, 247.
Genetically Modified Foods on Human Health
Genetically modified foods consumption began 20 years ago:
- No documented evidence for health risks
- Exposure rate is low
- Processing of grains
- Extraction of ingredients
- Heat treatment
- Low relative component of food
Looking at the cancer rates for both males and females in the United Stattes in 1999 and 2014, you can see cancer rates have actually fallen 248.
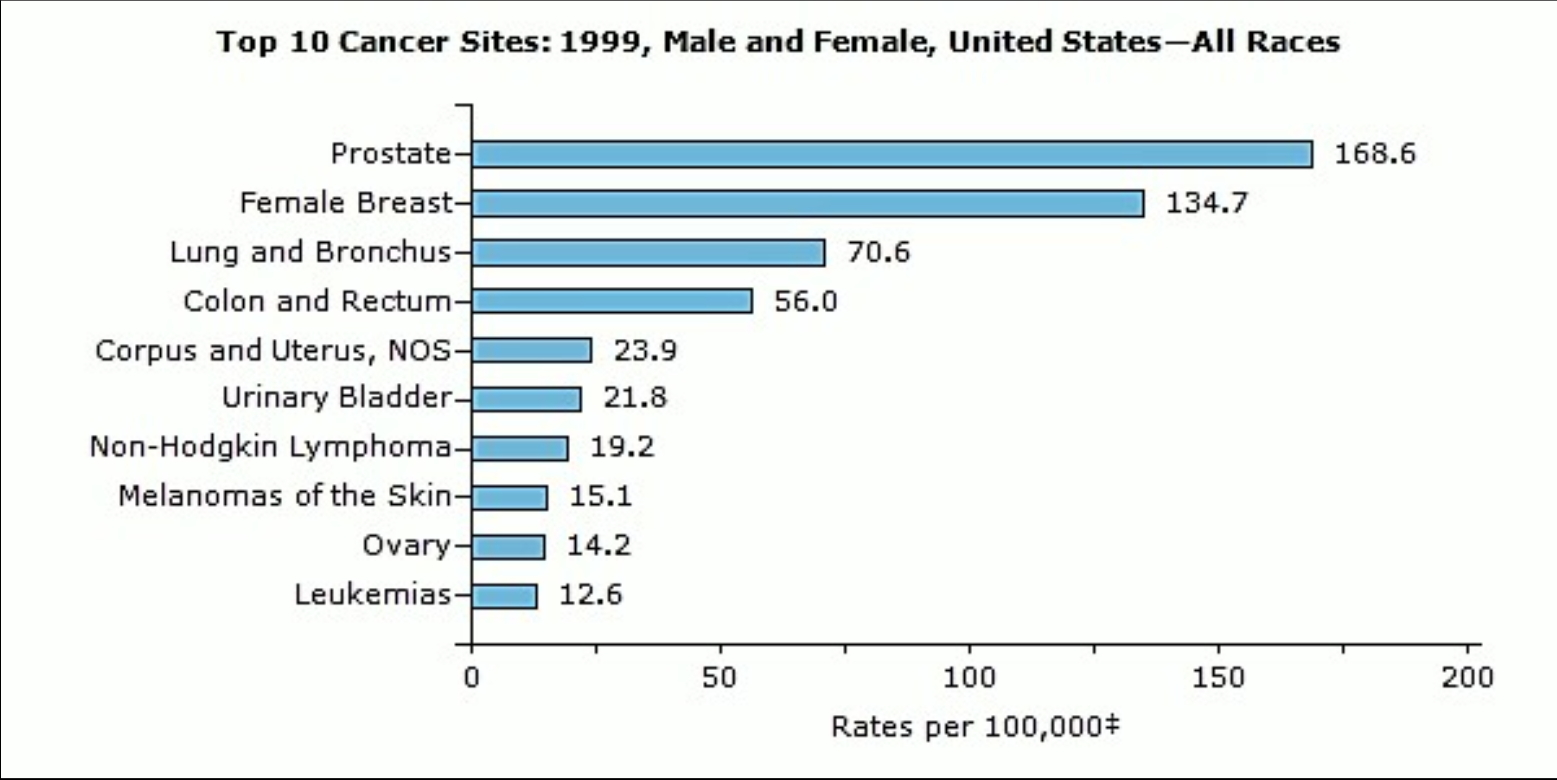
(Source U.S. Cancer Statistics Working Group. United States Cancer Statistics: 1999-2014 Incidence and Mortality Web-based Report. 248).
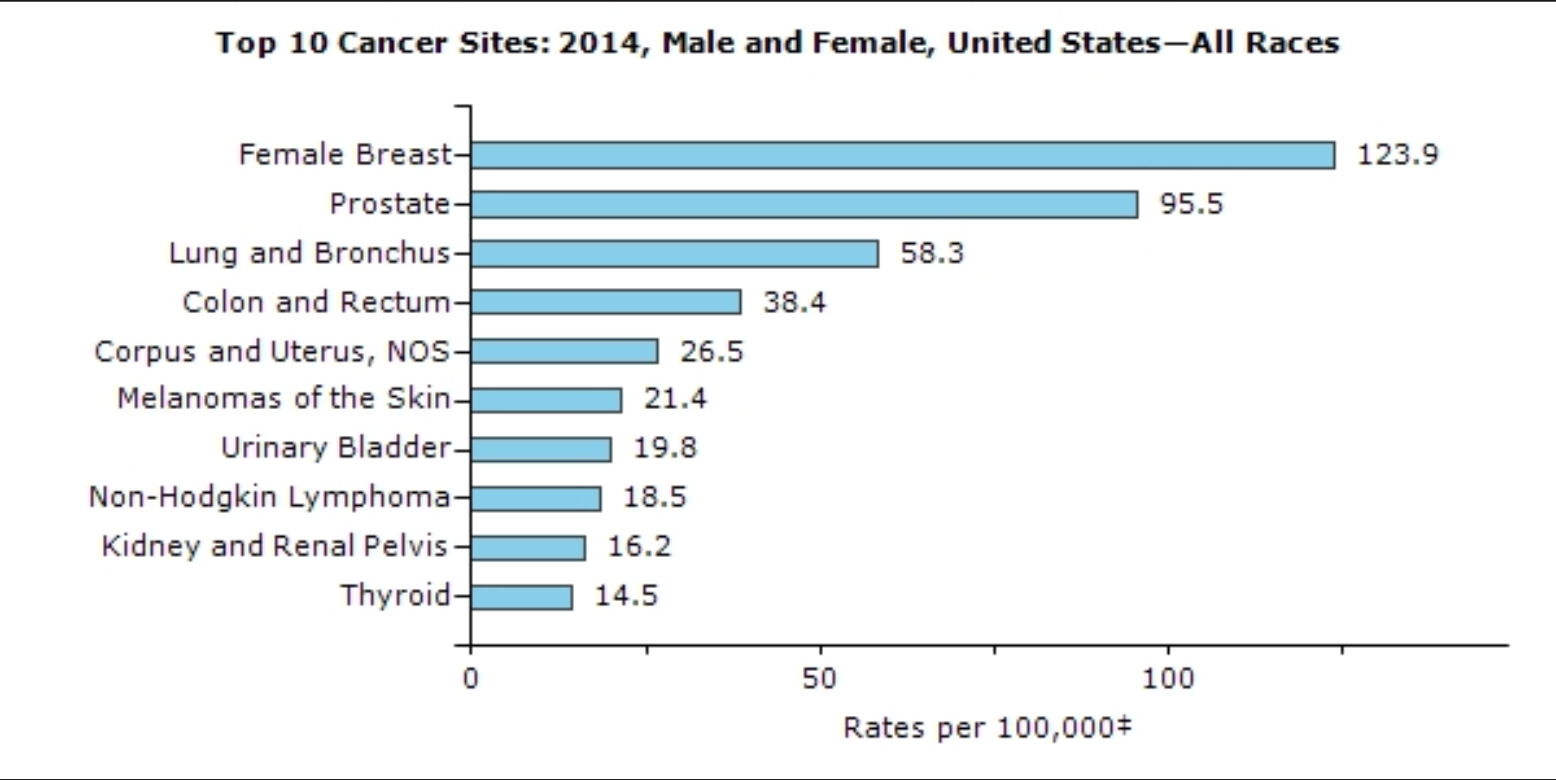
(Source U.S. Cancer Statistics Working Group. United States Cancer Statistics: 1999-2014 Incidence and Mortality Web-based Report. 248).
Snell et al. 249 examined 12 long-term studies (of more than 90 days, up to 2 years in duration) and 12 multigenerational studies (from 2 to 5 generations) on the effects of diets containing GM maize, potato, soybean, rice, or triticale on animal health. They referenced the 90-day studies on GM feed for which long-term or multigenerational study data were available. Many parameters have been examined using biochemical analyses, histological examination of specific organs, hematology and the detection of transgenic DNA. Results from all the 24 studies do not suggest any health hazards and, in general, there were no statistically significant differences within parameters observed.
Summary
The GM foods have the potential to solve many of the world’s hunger and malnutrition problems, and to help protect and preserve the environment by increasing yield and reducing reliance upon synthetic pesticides and herbicides. The debate on the safety of genetically modified organisms (GMOs) used for food and feed is still very lively throughout the world, more than 15 years after their first commercial release. There are controversies around GM food on several levels, including whether food produced with it is safe, whether it should be labelled and if so how, whether agricultural biotechnology and it is needed to address world hunger now or in the future, and more specifically with respect to intellectual property and market dynamics, environmental effects of GM crops and GM crops’ role in industrial agricultural more generally.
Review of literature found no plausible mechanism by which glyphosate would induce adverse developmental or reproductive outcome.
Almost all GMOs disseminated in the environment are plants, namely soy, maize, cotton, and oilseed rape (1995-2010) and more recently in November 2015, the U.S. Food and Drug Administration (FDA) after several years of study approved genetically modified (GM) faster-growing salmon as safe for human consumption 250. This is the first genetically modified animal to gain FDA approval and will probably be the first animal GMO in the supermarket 251. The faster growing salmon is a product of AquaBounty Technologies and was created by inserting a growth hormone gene from Chinook salmon and a promoter gene from Ocean Pout into the Atlantic salmon’s genome. Plainly stated, the promoter gene of the Ocean Pout turns on the growth gene from the Chinook salmon, allowing the fish to grow to market size in half the time as its non-GM counterpart (18 months vs. 3 years) 252. According to the FDA, since GM salmon was determined to be safe to eat and nutritionally equivalent to its non-GM counterpart, it does not require additional labeling identifying it as genetically modified 250, 252.
Apart from the faster-growing salmon, the plants, their genetic and phenotypic modifications are only herbicide tolerance and / or insecticide production (modified Bt toxins) in more than 99% cases. As a matter of fact, 97% of edible GMOs among cultivated GMOs (soy, corn and oilseed rape or canola, excluding cotton) are grown in South and North America – all these plants have been modified to tolerate and/or produce one or more pesticides and contain therefore such residues at various levels. Most are Roundup residues (it is a major herbicide used worldwide and tolerated by about 80% of GMOs). Other residues are from modified Bt insecticide toxins, which are directly synthesized by the GM plants from transgenes.
Current epidemiological evidence, albeit limited to a few studies using non-quantitative and indirect estimates and dichotomous analysis of exposures, does not lend support to public concerns that glyphosate-based pesticides might pose developmental risks to the unborn child 253. Nonetheless, owing to methodological limitations of existing analytical observational studies, and particularly to a lack of a direct measurement (urine and/or blood levels), or an indirect estimation of exposure that has proven valid, these negative findings cannot be taken as definitive evidence that glyphosate, at current levels of occupational and environmental exposures, brings no risk for human development and reproduction 253. Furthermore, because of potential for exposure of a large segment of human population to genetically modified foods, more research is needed to ensure that the genetically modified foods are safe for human consumption.
- Genetically modified organisms – GMOs. https://medlineplus.gov/ency/article/002432.htm[↩]
- United Nations Department of Economic and Social Affairs 21 June 2017. World population projected to reach 9.8 billion in 2050, and 11.2 billion in 2100. https://www.un.org/development/desa/en/news/population/world-population-prospects-2017.html[↩]
- Pollard C.M., S., Food Insecurity and Hunger in Rich Countries—It Is Time for Action against Inequality, Int. J. Environ. Res. Public Health 2019, Vol. 16, Page 1804 16 (2019) 1804, 10.3390/IJERPH16101804[↩]
- Joardder M.U.H., Masud M.H., Correction to: Food Preservation in Developing Countries: Challenges and solutions, Food Preserv. Dev. Ctries. Chall. Solut. (2019) C1–C1, 10.1007/978-3-030-11530-2_11[↩]
- Adeyeye S.A.O., Idowu-Adebayo F. Genetically modified and biofortified crops and food security in developing countries. Nutr. Food Sci. 2019;49:978–986. doi: 10.1108/NFS-12-2018-0335[↩]
- Jones L. Genetically modified foods. BMJ : British Medical Journal. 1999;318(7183):581-584. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1115027/[↩]
- Clive J. Global status of commercialized Biotech/GM crops. ISAAA Briefs 43. Ithaca: International Service for the Acquisition of Agri-biotech Applications; 2011.[↩][↩][↩]
- Bawa AS, Anilakumar KR. Genetically modified foods: safety, risks and public concerns—a review. Journal of Food Science and Technology. 2013;50(6):1035-1046. doi:10.1007/s13197-012-0899-1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3791249/[↩][↩][↩][↩][↩][↩]
- University of Minnesota. Considering the Mandatory Labeling of Genetically-Engineered (GE) Foods in the U.S. https://www.foodpolicy.umn.edu/policy-summaries-and-analyses/considering-mandatory-labeling-genetically-engineered-ge-foods-us [↩]
- Border P, Norton M. Genetically modified foods—benefits and risks, regulation and public acceptance. London: Parliamentary Office of Science and Technology; 1998.[↩]
- Linnhoff S., Volovich E., Martin H.M., Smith L.M., An examination of millennials’ attitudes toward genetically modified organism (GMO) foods: is it Franken-food or super-food?, Int. J. Agric. Resour. Gov. Ecol. 13 (2017) 371, 10.1504/IJARGE.2017.088403[↩]
- Vega Rodríguez A., Rodríguez-Oramas C., Sanjuán Velázquez E., Hardisson de la Torre A., Rubio Armendáriz C., Carrascosa Iruzubieta C., Myths and Realities about Genetically Modified Food: A Risk-Benefit Analysis, Appl. Sci. 12 (2022) 2861, 10.3390/app12062861[↩]
- Thangarajan H., Remesh S.D., Kumar P.S., Chandran A.R.R., Rajaendran S., Sandrasaigaran P., Genetically Modified Foods for Sustainable Food Security: Debunking the Myths, Malays. J. Sci. Adv. Technol. 1 (2021) 129–135, 10.56532/MJSAT.V1I4.28[↩]
- Dong W., Anwar M.A., Nawaz M.A., Rongting Z., Asmi F., Sajjad A., et al. Knowledge mapping of GMO/GMF research in social sphere, Cogent. Food Agric. 5 (2019) 1602103, 10.1080/23311932.2019.1602103[↩]
- Genetically modified crops: methodology, benefits, regulation and public concerns. Halford NG, Shewry PR. Br Med Bull. 2000; 56(1):62-73. https://www.ncbi.nlm.nih.gov/pubmed/10885105/[↩]
- Global Biotech Area Surges Past 100 Million Hectares on 13 Percent Growth: International Service for the Acquisition of Agri. Biotech Applications ISAAA (US) 2007. http://www.bionity.com/en/news/61027/global-biotech-area-surges-past-100-million-hectares-on-13-percent-growth.html[↩][↩][↩][↩]
- Nature 01 May 2013. Case studies: A hard look at GM crops. https://www.nature.com/news/case-studies-a-hard-look-at-gm-crops-1.12907[↩]
- Brookes G, Barfoot P. GM crops The First Ten Years-Global Socio-economic and Environmental Impacts; UK: PG Economics Ltd; 2006. The International Service for the Acquisition of Agri-biotech Applications[↩]
- Improving the Regulation of Genetically Modified Foods and Other Novel Foods in Canada. Canadian Biotechnology Advisory Committee, Report to the Government of Canada Biotechnology Ministerial Coordinating Committee. 2002 Aug; http://publications.gc.ca/collections/Collection/C2-589-2001-1E.pdf[↩]
- United States Department of Agriculture, Economic Research Service. Recent Trends in GE Adoption. https://www.ers.usda.gov/data-products/adoption-of-genetically-engineered-crops-in-the-us/recent-trends-in-ge-adoption/[↩]
- Iowa State University. Food Science and Human Nutrition. Safety of Genetically Modified Foods and Food Ingredients. https://www.aphis.usda.gov/stakeholders/downloads/2015/coexistence/Ruth-MacDonald.pdf[↩][↩][↩][↩][↩][↩][↩]
- United States Department of Agriculture, Agricultural Marketing Service. Can GMOs Be Used in Organic Products ? https://www.ams.usda.gov/sites/default/files/media/Can%20GMOs%20be%20Used.pdf[↩][↩][↩][↩]
- Whole Foods Market. Organic and Non GMO Market Growth 2015. https://www.aphis.usda.gov/stakeholders/downloads/2015/coexistence/Errol-Schweizer.pdf[↩]
- United States Department of Agriculture, Economic Research Service. Recent Trends in GE Adoption. https://www.ers.usda.gov/data-products/adoption-of-genetically-engineered-crops-in-the-us/recent-trends-in-ge-adoption/[↩][↩][↩][↩]
- United States Department of Agriculture, Economic Research Service. Biotechnology Overview. https://www.ers.usda.gov/topics/farm-practices-management/biotechnology/[↩]
- Johnson SR. Quantification of the impacts on US Agriculture of Biotechnology-Derived Crops Planted in 2006. Washington DC: National Centre for Food and Agricultural Policy; 2008.[↩]
- Hamer H, Scuse T (2010) National Agricultural Statistics Service (NASS), Agricultural Statistics Board, US Department of Agriculture. Acreage report, NY.[↩]
- Joana C, Isabel M, Joana SA, Oliveira MBPP. Monitoring genetically modified soybean along the industrial soybean oil extraction and refining processes by polymerase chain reaction techniques. Food Res Int. 2010;43:301–306. doi: 10.1016/j.foodres.2009.10.003.[↩]
- United States Department of Agriculture, Agricultural Marketing Service. Can GMOs Be Used in Organic Products ? https://www.ams.usda.gov/publications/content/can-gmos-be-used-organic-products[↩]
- Australian Government Department of the Environment and Energy. Genetically modified organisms. http://www.environment.gov.au/topics/science-and-research/state-environment-reporting/soe-2006-gmos[↩]
- MacLean GD, Waterhouse PM, Evans G and Gibbs MJ (eds) 1997, Commercialisation of Transgenic Crops, Bureau of Resource Sciences, Australian Government Publishing Service.[↩]
- Plant Physiology Vol. 160, Issue 4, Dec 2012. https://doi.org/10.1104/pp.112.204271. Crop Genome Plasticity and Its Relevance to Food and Feed Safety of Genetically Engineered Breeding Stacks. https://www.ncbi.nlm.nih.gov/pubmed/23060369[↩][↩][↩][↩]
- Anita Bakshi. Journal of Toxicology and Environmental Health, Part B Vol. 6 , Iss. 3,2003. Potential Adverse Health Effects of Genetically Modified Crops. https://www.ncbi.nlm.nih.gov/pubmed/12746139[↩][↩]
- Pinstrup-Anderson P, Pandra-Lorch R, Rosegrant MW. World food prospects: critical issues for the early twenty-first century. 1999 Food policy report. Washington DC: International Food Policy Research Institute; 1999.[↩]
- Food and Agriculture Organisation. The State of Food Insecurity in the World. Rome: FAO; 2001.[↩]
- Smith LC, El Obeid AE, Jensen HH. The geography and causes of food insecurity in developing countries. Agriculture Economics. 2000;22:199–215.[↩][↩]
- Christou P, Twyman RM. The potential of genetically enhanced plants to address food insecurity. Nut Research Rev. 2004;17:23–42. https://www.ncbi.nlm.nih.gov/pubmed/19079913[↩][↩]
- Byrnes BH, Bumb BL. Population growth, food production and nutrient requirements. J Crop Prod. 1998;1:1–27.[↩]
- Byerlee D, Helsey P, Pingali PL. Realising yield gains for food staples in developing countries in the early 21st century: prospects and challenges. In: Chang BM, Colombo M, Soronolo M, editors. Food Needs of the Developing World in the 21st Century. Vatican City: Political Academy of Sciences; 2000. pp. 207–50.[↩]
- Rosegrant MW, Paisner MS, Mejer S, Witcover J. 2020 Global Food Outlook Trends, Alternatives and Choices. A 2020 Vision for Food Agriculture and the Environment Initiative. Washington DC: IFPRI; 2001.[↩]
- Emerging infectious diseases of plants: pathogen pollution, climate change and agrotechnology drivers. Anderson PK, Cunningham AA, Patel NG, Morales FJ, Epstein PR, Daszak P. Trends Ecol Evol. 2004 Oct; 19(10):535-44. https://www.ncbi.nlm.nih.gov/pubmed/16701319/[↩]
- Engineering plants with increased disease resistance: what are we going to express? Gurr SJ, Rushton PJ. Trends Biotechnol. 2005 Jun; 23(6):275-82. https://www.ncbi.nlm.nih.gov/pubmed/15922079/[↩]
- Brookes G, Barfoot P. ISAAA briefs. ISAAA; 2006. http://www.isaaa.org/[↩]
- Recent advances in engineering plant tolerance to abiotic stress: achievements and limitations. Vinocur B, Altman A. Curr Opin Biotechnol. 2005 Apr; 16(2):123-32. https://www.ncbi.nlm.nih.gov/pubmed/15831376/[↩]
- Ashraf M. Breeding for salinity tolerance in plants. Crit Rev Plant Sci. 1994;13:17–42.[↩]
- Expression of an active tobacco mitogen-activated protein kinase kinase kinase enhances freezing tolerance in transgenic maize. Shou H, Bordallo P, Fan JB, Yeakley JM, Bibikova M, Sheen J, Wang K. Proc Natl Acad Sci U S A. 2004 Mar 2; 101(9):3298-303. https://www.ncbi.nlm.nih.gov/pubmed/14960727/[↩]
- The potential of genetically enhanced plants to address food insecurity. Christou P, Twyman RM. Nutr Res Rev. 2004 Jun; 17(1):23-42. https://www.ncbi.nlm.nih.gov/pubmed/19079913/[↩][↩]
- World Health Organisation. Combating vitamin A deficiency. Geneva: WHO; 2017. http://www.who.int/mediacentre/factsheets/malnutrition/en/[↩]
- Engineering the provitamin A (beta-carotene) biosynthetic pathway into (carotenoid-free) rice endosperm. Ye X, Al-Babili S, Klöti A, Zhang J, Lucca P, Beyer P, Potrykus I. Science. 2000 Jan 14; 287(5451):303-5. https://www.ncbi.nlm.nih.gov/pubmed/10634784/[↩]
- Improving the nutritional value of Golden Rice through increased pro-vitamin A content. Paine JA, Shipton CA, Chaggar S, Howells RM, Kennedy MJ, Vernon G, Wright SY, Hinchliffe E, Adams JL, Silverstone AL, Drake R. Nat Biotechnol. 2005 Apr; 23(4):482-7. https://www.ncbi.nlm.nih.gov/pubmed/15793573/[↩][↩]
- Golden rice and beyond. Potrykus I. Plant Physiol. 2001 Mar; 125(3):1157-61. https://www.ncbi.nlm.nih.gov/pubmed/11244094/[↩]
- The European Food Safety Authority. Genetically Modified Organisms. http://www.efsa.europa.eu/en/topics/topic/genetically-modified-organisms[↩]
- McKeon TA. Genetically modified crops for industrial products and processes and their effects on human health. Trends Food Sci Tech. 2003;14:229–41.[↩]
- What is glyphosate? https://www.apvma.gov.au/resources/chemicals-news/glyphosate/glyphosate-australia[↩]
- Richmond, M. E. Glyphosate: A review of its global use, environmental impact, and potential health effects on humans and other species. J. Environ. Stud. Sci.8, 416–434 (2018).[↩]
- Clapp, J. Explaining growing glyphosate use: The political economy of herbicide-dependent agriculture. Glob. Environ. Change67, 102239 (2021).[↩]
- Araujo, L. G., Zordan, D. F., Celzard, A. & Fierro, V. Glyphosate uses, adverse effects and alternatives: focus on the current scenario in Brazil. Environ. Geochem. Health45, 9559–9582 (2023).[↩]
- Tauhata, S. B. F. et al. The glyphosate controversy: An update. Arq. Inst. Biol. (Sao. Paulo)87, 1–8 (2020).[↩]
- Kraus EC, Stout MJ. Direct and Indirect Effects of Herbicides on Insect Herbivores in Rice, Oryza sativa. Sci Rep. 2019 May 6;9(1):6998. doi: 10.1038/s41598-019-43361-w[↩]
- Gomes MP, Smedbol E, Chalifour A, Hénault-Ethier L, Labrecque M, Lepage L, Lucotte M, Juneau P. Alteration of plant physiology by glyphosate and its by-product aminomethylphosphonic acid: an overview. J Exp Bot. 2014 Sep;65(17):4691-703. doi: 10.1093/jxb/eru269.[↩]
- Franz J Mao M Sikorski J . 1997. Glyphosate: a unique and global herbicide. American Chemical Society Monograph 189: Washington DC.[↩][↩]
- Duke SO, Powles SB. Glyphosate: a once-in-a-century herbicide. Pest Manag Sci. 2008 Apr;64(4):319-25. doi: 10.1002/ps.1518[↩]
- Cobb A . 1992. Herbicides and plant physiology. London: Chapman and Hall.[↩]
- Glyphosate. http://npic.orst.edu/factsheets/glyphogen.html[↩][↩][↩][↩]
- Xu J, et al. Glyphosate contamination in grains and foods: an overview. Food Control; 2019. p. 106710.[↩]
- Borggaard OK Gimsing AL . 2008. Fate of glyphosate in soil and the possibility of leaching to ground and surface waters: a review. Pest Management Science456, 441–456.[↩]
- Pompermaier A, Alves C, Chagas FB, Tamagno WA, Bridi C, Ferreira GF, Hartmann PA, Hartmann M. Effects of glyphosate based herbicide exposure in early developmental stages of Physalaemus gracilis. Sci Rep. 2024 Oct 27;14(1):25652. doi: 10.1038/s41598-024-76338-5[↩]
- Grunewald, K., Schmidt, W., Unger, C. & Hanschmann, G. Behavior of glyphosate and aminomethylphosphonic acid (AMPA) in soils and water of reservoir Radeburg II catchment (Saxony/Germany). J. plant Nutr. soil Sci.164, 65–70 (2001).[↩]
- Van Eerd LL Hoagland RE Zablotowicz RM Hall JC . 2003. Pesticide metabolism in plants and microorganisms. Weed Science51, 472–495.[↩]
- Reddy KN Rimando AM Duke SO . 2004. Aminomethylphosphonic acid, a metabolite of glyphosate, causes injury in glyphosate-treated, glyphosate-resistant soybean. Journal of Agricultural and Food Chemistry52, 5139–5143.[↩][↩]
- Satchivi NM Wax LM Stoller EW Briskin DP . 2000. Absorption and translocation of glyphosate isopropylamine and trimethylsulfonium salts in Abutilon theophrasti and Setaria faberi. Weed Science48, 675–679.[↩]
- Monquero PA Christoffoleti PJ Osuna MD De Prado RA . 2004. Absorção, translocação e metabolismo do glyphosate por plantas tolerantes e suscetíveis a este herbicida. Planta Daninha22, 445–451.[↩]
- Hetherington PR Reynolds TL Marshall G Kirkwood RC . 1999. The absorption, translocation and distribution of the herbicide glyphosate in maize expressing the CP-4 transgene. Journal of Experimental Botany50, 1567–1576.[↩]
- Feng PCC Chiu T Douglas Sammons R . 2003. Glyphosate efficacy is contributed by its tissue concentration and sensitivity in velvetleaf (Abutilon theophrasti). Pesticide Biochemistry and Physiology77, 83–91.[↩]
- Viirlaid E, Ilisson M, Kopanchuk S, Mäeorg U, Rinken A, Rinken T. Immunoassay for rapid on-site detection of glyphosate herbicide. Environ Monit Assess. 2019 Jul 24;191(8):507. doi: 10.1007/s10661-019-7657-z[↩]
- Siehl D . 1997. Inhibitors of EPSPS synthase, glutamine synthetase and histidine synthesis. In: Roe R Burton J Kuhr R, eds. Herbicide activity: toxicology, biochemistry and molecular biology. Amsterdam: IOS Press, 37–67.[↩]
- Marcelo P. Gomes, Elise Smedbol, Annie Chalifour, Louise Hénault-Ethier, Michel Labrecque, Laurent Lepage, Marc Lucotte, Philippe Juneau, Alteration of plant physiology by glyphosate and its by-product aminomethylphosphonic acid: an overview, Journal of Experimental Botany, Volume 65, Issue 17, September 2014, Pages 4691–4703, https://doi.org/10.1093/jxb/eru269[↩][↩][↩][↩][↩]
- Muller F, Applebyke AP. 2010. Weed control, 2. Individual herbicides. In: Ullmann’s encyclopedia of industrial chemistry. Wiley VCH, Verlag GmbH & Co. KGaA. 10.1002/14356007.o28_o01[↩]
- Plimmer JR, Bradow JM, Dionigi CP, et al. 2004. Herbicides. In: Kirk-Othmer encyclopedia of chemical technology. John Wiley & Sons, Inc. 10.1002/0471238961.0805180202180104.a01.pub2[↩]
- Dow. 2017. Rodeo herbicide. Specimen label revised 01-05-17. Dow Chemical Company, Dow Agrosciences.[↩]
- PAN. 2016a. PAN pesticides database-California pesticide use. Oakland, CA: Pesticide Action Network, North America. http://www.pesticideinfo.org/Detail_ChemUse.jsp?Rec_Id=PC33138[↩]
- PAN. 2016b. PAN pesticides database-Pesticide products. Oakland, CA: Pesticide Action Network, North America. http://www.pesticideinfo.org/List_Products.jsp?Rec_Id=PC33138&Chem_Name=Glyphosate&PC_Code=417300,%20471300[↩]
- Kováčik J, Novotný V, Bujdoš M, Dresler S, Hladký J, Babula P. Glyphosate does not show higher phytotoxicity than cadmium: Cross talk and metabolic changes in common herb. J Hazard Mater. 2020 Feb 5;383:121250. doi: 10.1016/j.jhazmat.2019.121250[↩]
- ECHA (European Chemicals Agency), 2022. Committee for Risk Assessment (RAC) Opinion proposing harmonised classification and labelling at EU level of glyphosate (ISO); N-(phosphonomethyl)glycine. CLH-O-0000007122-85-01/F[↩]
- Álvarez, F., Arena, M., Auteri, D., Binaglia, M., Castoldi, A. F., Chiusolo, A., Crivellente, F., Egsmose, M., Fait, G., Ferilli, F., Gouliarmou, V., Nogareda, L. H., Ippolito, A., Istace, F., Jarrah, S., Kardassi, D., Kienzler, A., Lanzoni, A., … Villamar-Bouza, L. (2023). Peer review of the pesticide risk assessment of the active substance glyphosate. EFSA Journal, 21(7), 1–52. http://doi.org.10.2903/j.efsa.2023.8164[↩][↩][↩][↩]
- IARC Monographs Volume 112: evaluation of five organophosphate insecticides and herbicides. https://www.iarc.who.int/wp-content/uploads/2018/07/MonographVolume112-1.pdf[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- IARC Monograph on Glyphosate. https://www.iarc.who.int/featured-news/media-centre-iarc-news-glyphosate/[↩][↩]
- De Roos AJ, Zahm SH, Cantor KP, Weisenburger DD, Holmes FF, Burmeister LF, Blair A. Integrative assessment of multiple pesticides as risk factors for non-Hodgkin’s lymphoma among men. Occup Environ Med. 2003 Sep;60(9):E11. https://pmc.ncbi.nlm.nih.gov/articles/instance/1740618/pdf/v060p00e11.pdf[↩][↩]
- Hardell L, Eriksson M. A case-control study of non-Hodgkin lymphoma and exposure to pesticides. Cancer. 1999 Mar 15;85(6):1353-60. doi: 10.1002/(sici)1097-0142(19990315)85:6<1353::aid-cncr19>3.0.co;2-1[↩][↩]
- Hardell L, Eriksson M, Nordstrom M. Exposure to pesticides as risk factor for non-Hodgkin’s lymphoma and hairy cell leukemia: pooled analysis of two Swedish case-control studies. Leuk Lymphoma. 2002 May;43(5):1043-9. doi: 10.1080/10428190290021560[↩][↩]
- McDuffie HH, Pahwa P, McLaughlin JR, Spinelli JJ, Fincham S, Dosman JA, Robson D, Skinnider LF, Choi NW. Non-Hodgkin’s lymphoma and specific pesticide exposures in men: cross-Canada study of pesticides and health. Cancer Epidemiol Biomarkers Prev. 2001 Nov;10(11):1155-63.[↩][↩]
- De Roos AJ, Blair A, Rusiecki JA, Hoppin JA, Svec M, Dosemeci M, Sandler DP, Alavanja MC. Cancer incidence among glyphosate-exposed pesticide applicators in the Agricultural Health Study. Environ Health Perspect. 2005 Jan;113(1):49-54. doi: 10.1289/ehp.7340[↩][↩][↩][↩][↩]
- Andreotti G, Koutros S, Hofmann JN, Sandler DP, Lubin JH, Lynch CF, Lerro CC, De Roos AJ, Parks CG, Alavanja MC, Silverman DT, Beane Freeman LE. Glyphosate Use and Cancer Incidence in the Agricultural Health Study. J Natl Cancer Inst. 2018 May 1;110(5):509-516. doi: 10.1093/jnci/djx233[↩][↩][↩][↩][↩]
- Engel LS, Hill DA, Hoppin JA, Lubin JH, Lynch CF, Pierce J, Samanic C, Sandler DP, Blair A, Alavanja MC. Pesticide use and breast cancer risk among farmers’ wives in the agricultural health study. Am J Epidemiol. 2005 Jan 15;161(2):121-35. doi: 10.1093/aje/kwi022[↩]
- Flower KB, Hoppin JA, Lynch CF, Blair A, Knott C, Shore DL, Sandler DP. Cancer risk and parental pesticide application in children of Agricultural Health Study participants. Environ Health Perspect. 2004 Apr;112(5):631-5. doi: 10.1289/ehp.6586[↩]
- Koutros S, Beane Freeman LE, Lubin JH, Heltshe SL, Andreotti G, Barry KH, DellaValle CT, Hoppin JA, Sandler DP, Lynch CF, Blair A, Alavanja MC. Risk of total and aggressive prostate cancer and pesticide use in the Agricultural Health Study. Am J Epidemiol. 2013 Jan 1;177(1):59-74. doi: 10.1093/aje/kws225[↩]
- Koutros S, Silverman DT, Alavanja MC, Andreotti G, Lerro CC, Heltshe S, Lynch CF, Sandler DP, Blair A, Beane Freeman LE. Occupational exposure to pesticides and bladder cancer risk. Int J Epidemiol. 2016 Jun;45(3):792-805. doi: 10.1093/ije/dyv195[↩]
- Lee WJ, Sandler DP, Blair A, Samanic C, Cross AJ, Alavanja MC. Pesticide use and colorectal cancer risk in the Agricultural Health Study. Int J Cancer. 2007 Jul 15;121(2):339-46. doi: 10.1002/ijc.22635[↩]
- Andreotti G, Freeman LE, Hou L, Coble J, Rusiecki J, Hoppin JA, Silverman DT, Alavanja MC. Agricultural pesticide use and pancreatic cancer risk in the Agricultural Health Study Cohort. Int J Cancer. 2009 May 15;124(10):2495-500. doi: 10.1002/ijc.24185[↩]
- Band PR, Abanto Z, Bert J, Lang B, Fang R, Gallagher RP, Le ND. Prostate cancer risk and exposure to pesticides in British Columbia farmers. Prostate. 2011 Feb 1;71(2):168-83. doi: 10.1002/pros.21232[↩]
- Lee WJ, Lijinsky W, Heineman EF, Markin RS, Weisenburger DD, Ward MH. Agricultural pesticide use and adenocarcinomas of the stomach and oesophagus. Occup Environ Med. 2004 Sep;61(9):743-9. doi: 10.1136/oem.2003.011858[↩]
- Lee WJ, Colt JS, Heineman EF, McComb R, Weisenburger DD, Lijinsky W, Ward MH. Agricultural pesticide use and risk of glioma in Nebraska, United States. Occup Environ Med. 2005 Nov;62(11):786-92. doi: 10.1136/oem.2005.020230[↩]
- Pahwa P, Karunanayake CP, Dosman JA, Spinelli JJ, McLaughlin JR; Cross-Canada Group. Soft-tissue sarcoma and pesticides exposure in men: results of a Canadian case-control study. J Occup Environ Med. 2011 Nov;53(11):1279-86. doi: 10.1097/JOM.0b013e3182307845[↩]
- Yiin JH, Ruder AM, Stewart PA, Waters MA, Carreón T, Butler MA, Calvert GM, Davis-King KE, Schulte PA, Mandel JS, Morton RF, Reding DJ, Rosenman KD; Brain Cancer Collaborative Study Group. The Upper Midwest Health Study: a case-control study of pesticide applicators and risk of glioma. Environ Health. 2012 Jun 12;11:39. doi: 10.1186/1476-069X-11-39[↩]
- Toxicological Profile for Glyphosate. Atlanta (GA): Agency for Toxic Substances and Disease Registry (US); 2020 Aug. CHAPTER 2, HEALTH EFFECTS. Available from: https://www.ncbi.nlm.nih.gov/books/NBK591692[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Cho YS, Moon JM, Chun BJ, Lee BK. Use of qSOFA Score in Predicting the Outcomes of Patients With Glyphosate Surfactant Herbicide Poisoning Immediately Upon Arrival at the Emergency Department. Shock. 2019 Apr;51(4):447-452. doi: 10.1097/SHK.0000000000001201[↩]
- Camacho A, Mejía D. The health consequences of aerial spraying illicit crops: The case of Colombia. J Health Econ. 2017 Jul;54:147-160. doi: 10.1016/j.jhealeco.2017.04.005[↩]
- Hoppin JA, Umbach DM, London SJ, et al. 2002. Chemical predictors of wheeze among farmer pesticide applicators in the Agricultural Health Study. Am J Respir Crit Care Med 165(5):683–689. 10.1164/ajrccm.165.5.2106074[↩][↩][↩]
- Hoppin JA, Umbach DM, London SJ, Lynch CF, Alavanja MC, Sandler DP. Pesticides and adult respiratory outcomes in the agricultural health study. Ann N Y Acad Sci. 2006 Sep;1076:343-54. doi: 10.1196/annals.1371.044[↩][↩][↩]
- Hoppin JA, Umbach DM, London SJ, et al. 2006b. Pesticides associated with wheeze among commercial pesticide applicators in the Agricultural Health Study. Am J Epidemiol 163(12):1129–1137. 10.1093/aje/kwj138[↩][↩][↩]
- Hoppin JA, Valcin M, Henneberger PK, Kullman GJ, Umbach DM, London SJ, Alavanja MC, Sandler DP. Pesticide use and chronic bronchitis among farmers in the Agricultural Health Study. Am J Ind Med. 2007 Dec;50(12):969-79. doi: 10.1002/ajim.20523[↩]
- Hoppin JA, Umbach DM, London SJ, et al. 2008. Pesticides and atopic and nonatopic asthma among farm women in the Agricultural Health Study. Am J Respir Crit Care Med 177(1):11–18. 10.1164/rccm.200706-821OC[↩][↩][↩]
- Hoppin JA, Umbach DM, London SJ, et al. 2009. Pesticide use and adult-onset asthma among male farmers in the Agricultural Health Study. Eur Respir J 34(6):1296–1303. 10.1183/09031936.00005509[↩][↩][↩]
- Hoppin JA, Umbach DM, Long S, et al. 2017. Pesticides are associated with allergic and non-allergic wheeze among male farmers. Environ Health Perspect 125(4):535–543. 10.1289/EHP315[↩][↩]
- Slager RE, Poole JA, LeVan TD, et al. 2009. Rhinitis associated with pesticide exposure among commercial pesticide applicators in the Agricultural Health Study. Occup Environ Med 66(11):718–724. 10.1136/oem.2008.041798[↩][↩][↩][↩][↩]
- Slager RE, Simpson SL, Levan TD, et al. 2010. Rhinitis associated with pesticide use among private pesticide applicators in the Agricultural Health Study. J Toxicol Environ Health A 73(20):1382–1393. 10.1080/15287394.2010.497443[↩][↩][↩]
- Dayton SB, Sandler DP, Blair A, et al. 2010. Pesticide use and myocardial infarction incidence among farm women in the Agricultural Health Study. J Occup Environ Med 52(7):693–697. 10.1097/JOM.0b013e3181e66d25[↩]
- Mills KT, Blair A, Freeman LE, et al. 2009. Pesticides and myocardial infarction incidence and mortality among male pesticide applicators in the Agricultural Health Study. Am J Epidemiol 170(7):892–900. 10.1093/aje/kwp214[↩]
- De Roos AJ, Cooper GS, Alavanja MC, et al. 2005b. Rheumatoid arthritis among women in the Agricultural Health Study: Risk associated with farming activities and exposures. Ann Epidemiol 15(10):762–770. 10.1016/j.annepidem.2005.08.001[↩][↩]
- Parks CG, Hoppin JA, De Roos AJ, Costenbader KH, Alavanja MC, Sandler DP. Rheumatoid Arthritis in Agricultural Health Study Spouses: Associations with Pesticides and Other Farm Exposures. Environ Health Perspect. 2016 Nov;124(11):1728-1734. doi: 10.1289/EHP129[↩][↩]
- Mills PJ, Caussy C, Loomba R. 2019. Glyphosate excretion is associated with steatohepatitis and advanced liver fibrosis in patients with fatty liver disease. Clin Gastroenterol Hepatol. 10.1016/j.cgh.2019.03.045[↩][↩]
- Camacho A, Mejia D. 2017. The health consequences of aerial spraying illicit crops: The case of Colombia. J Health Econ 54147–160. 10.1016/j.jhealeco.2017.04.005[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Maibach HI. Irritation, sensitization, photoirritation and photosensitization assays with a glyphosate herbicide. Contact Dermatitis. 1986 Sep;15(3):152-6. doi: 10.1111/j.1600-0536.1986.tb01316.x[↩][↩][↩][↩][↩][↩][↩]
- Kirrane EF, Hoppin JA, Kamel F, et al. 2005. Retinal degeneration and other eye disorders in wives of farmer pesticide applicators enrolled in the Agricultural Health Study. Am J Epidemiol 161(11):1020–1029. 10.1093/aje/kwi140[↩][↩]
- Goldner WS, Sandler DP, Yu F, et al. 2010. Pesticide use and thyroid disease among women in the Agricultural Health Study. Am J Epidemiol 171(4):455–464. 10.1093/aje/kwp404[↩][↩]
- Kongtip P, Nankongnab N, Kallayanatham N, et al. 2019. Thyroid hormones in conventional and organic farmers in Thailand. Int J Environ Res Public Health 16(15). 10.3390/ijerph16152704[↩][↩]
- Shrestha S, Parks CG, Goldner WS, et al. 2018. Pesticide use and incident hypothyroidism in pesticide applicators in the agricultural health study. Environ Health Perspect 126(9):097008. 10.1289/EHP3194[↩][↩]
- Kamel F, Tanner C, Umbach D, et al. 2007. Pesticide exposure and self-reported Parkinson’s disease in the Agricultural Health Study. Am J Epidemiol 165(4):364–374. 10.1093/aje/kwk024[↩][↩]
- Zhang C, Sun Y, Hu R, et al. 2018. A comparison of the effects of agricultural pesticide uses on peripheral nerve conduction in China. Sci Rep 8(1):9621. 10.1038/s41598-018-27713-6[↩][↩]
- Curtis KM, Savitz DA, Weinberg CR, Arbuckle TE. The effect of pesticide exposure on time to pregnancy. Epidemiology. 1999 Mar;10(2):112-7. Erratum in: Epidemiology 1999 Jul;10(4):470[↩]
- Arbuckle TE, Lin Z, Mery LS. An exploratory analysis of the effect of pesticide exposure on the risk of spontaneous abortion in an Ontario farm population. Environ Health Perspect. 2001 Aug;109(8):851-7. doi: 10.1289/ehp.01109851[↩][↩]
- García AM, Benavides FG, Fletcher T, Orts E. Paternal exposure to pesticides and congenital malformations. Scand J Work Environ Health. 1998 Dec;24(6):473-80. doi: 10.5271/sjweh.371[↩][↩]
- Garry VF, Harkins ME, Erickson LL, Long-Simpson LK, Holland SE, Burroughs BL. Birth defects, season of conception, and sex of children born to pesticide applicators living in the Red River Valley of Minnesota, USA. Environ Health Perspect. 2002 Jun;110 Suppl 3(Suppl 3):441-9. doi: 10.1289/ehp.02110s3441[↩][↩]
- Parvez S, Gerona RR, Proctor C, et al. 2018. Glyphosate exposure in pregnancy and shortened gestational length: a prospective Indiana birth cohort study. Environ Health 17(1):23. 10.1186/s12940-018-0367-0[↩][↩][↩]
- Rull RP, Ritz B, Shaw GM. 2006. Neural tube defects and maternal residential proximity to agricultural pesticide applications. Am J Epidemiol 163(8): 743–753. 10.1093/aje/kwj101[↩][↩]
- Sathyanarayana S, Basso O, Karr CJ, et al. 2010. Maternal pesticide use and birth weight in the Agricultural Health Study. J Agromedicine 15(2):127–136. 10.1080/10599241003622699[↩][↩]
- Savitz DA, Arbuckle T, Kaczor D, Curtis KM. Male pesticide exposure and pregnancy outcome. Am J Epidemiol. 1997 Dec 15;146(12):1025-36. doi: 10.1093/oxfordjournals.aje.a009231[↩][↩]
- Montgomery MP, Kamel F, Saldana TM, et al. 2008. Incident diabetes and pesticide exposure among licensed pesticide applicators: Agricultural Health Study, 1993-2003. Am J Epidemiol 167(10):1235–1246. 10.1093/aje/kwn028[↩]
- Saldana TM, Basso O, Hoppin JA, et al. 2007. Pesticide exposure and self-reported gestational diabetes mellitus in the Agricultural Health Study. Diabetes Care 30(3):529–534. 10.2337/dc06-1832[↩]
- Baier CJ, Gallegos CE, Raisman-Vozari R, et al. 2017. Behavioral impairments following repeated intranasal glyphosate-based herbicide administration in mice. Neurotoxicol Teratol 64:63–72. 10.1016/j.ntt.2017.10.004[↩][↩]
- El-Shenawy NS. 2009. Oxidative stress responses of rats exposed to Roundup and its active ingredient glyphosate. Environ Toxicol Pharmacol 28(3):379–385. 10.1016/j.etap.2009.06.001[↩][↩][↩][↩][↩][↩]
- EPA. 2013a. Memorandum. 14-Mar-2013. Memorandum: Glyphosate. Review and generation of data evaluation records. Reproduction and fertility effects study-rat OCSPP870.3800; OECD 416. MRID numbers 48865101, 48865102, 48865103, 48865104, 48865105 [Received via FOIA request]. U.S. Environmental Protection Agency.[↩][↩][↩][↩]
- EPA. 2013b. Memorandum. February 27, 2013. Memorandum: Glyphosate. Immunotoxicity study in mice. MRID 48934207 [Received via FOIA request]. U.S. Environmental Protection Agency.[↩][↩][↩]
- EPA. 2017b. Memorandum. December 13, 2017. Glyphosate: Preparation of data evaluation records for developmental rat and rabbit toxicity studies. MRID numbers 43320615, 43320616. Washington, DC: U.S. Environmental Protection Agency, Office of Chemical Safety and Pollution Prevention.[↩][↩]
- Manservisi F, Lesseur C, Panzacchi S, et al. 2019. The Ramazzini Institute 13-week pilot study glyphosate-based herbicides administered at human-equivalent dose to Sprague Dawley rats: effects on development and endocrine system. Environ Health 18(1):15. 10.1186/s12940-019-0453-y[↩][↩][↩][↩]
- Ait Bali Y, Ba-Mhamed S, Bennis M. 2017. Behavioral and immunohistochemical study of the effects of subchronic and chronic exposure to glyphosate in mice. Front Behav Neurosci 11:146. 10.3389/fnbeh.2017.00146[↩][↩][↩][↩][↩][↩][↩][↩]
- Almeida LL, Teixeira AAC, Soares AF, et al. 2017. Effects of melatonin in rats in the initial third stage of pregnancy exposed to sub-lethal doses of herbicides. Acta Histochem 119(3):220–227. 10.1016/j.acthis.2017.01.003[↩][↩][↩][↩]
- Cattani D, Cesconetto PA, Tavares MK, et al. 2017. Developmental exposure to glyphosate-based herbicide and depressive-like behavior in adult offspring: Implication of glutamate excitotoxicity and oxidative stress. Toxicology 38767–80. 10.1016/j.tox.2017.06.001[↩][↩][↩]
- Hamdaoui LN, M.; Rahmouni, F.; Harrabi, B.; Ayadi, F.; Sahnoun, Z.; Rebai, T. 2018. Subchronic exposure to kalach 360 SL-induced endocrine disruption and ovary damage in female rats. Arch Physiol Biochem 124(1):27–34. 10.1080/13813455.2017.1352606[↩][↩][↩]
- Pandey A, Dhabade P, Kumarasamy A. 2019. Inflammatory effects of subacute exposure of roundup in rat liver and adipose tissue. Dose Response 17(2):1559325819843380. 10.1177/1559325819843380[↩][↩]
- Teleken JL, Gomes ECZ, Marmentini C, et al. 2019. Glyphosate-based herbicide exposure during pregnancy and lactation malprograms the male reproductive morphofunction in F1 offspring. J Dev Orig Health Dis: 1–8. 10.1017/S2040174419000382[↩][↩][↩][↩][↩]
- Jasper R, Locatelli GO, Pilati C, et al. 2012. Evaluation of biochemical, hematological and oxidative parameters in mice exposed to the herbicide glyphosate-Roundup. Interdiscip Toxicol 5(3):133–140. I 10.2478/v10102-012-0022-5[↩][↩]
- EPA. 1992e. Data evaluation report. Test material: Glyphosate, technical; 98.7% purity; lot XHJ-64; white powder. Teratology study in rats. MRID 00098460. In: July 22, 1992. Memorandum. Glyphosate-List A chemical for reregistration-rereview of toxicology studies for acceptability. The attached 7-22-92 memorandum contains the Agency reviews for the following glyphosate studies: MRID numbers 0046362, 00046363, 00046364, 00067039, 00078619, 00078620, 00093879, 00098460, 00105995, 00132681, 00132683, 00132685 and 00132686 [Received via FOIA request]. HED pages 41–49. U.S. Environmental Protection Agency.[↩][↩][↩]
- EPA. 1992a. July 29, 1992. Memorandum. 48 Page(s). William Dykstra. Toxicology Branch I. Glyphosate (Roundup); review of 2-generation rat reproduction study; PP #0F03865, 2H05635 – Glyphosate in/on wheat. MRID 416215-01. Pages 9-10, 21-48 removed, registrant data. Tox review 009634. U.S. Environmental Protection Agency. https://archive.epa.gov/pesticides/chemicalsearch/chemical/foia/web/pdf/103601/103601-273.pdf[↩][↩]
- NTP. 1992. NTP technical report on the toxicity studies of glyphosate (CAS No. 1071-83-6) administered in dosed feed To F344/N rats and B6C3F1 mice. Toxicity Report Series, No. 16. National Toxicology Program, U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health.[↩][↩][↩][↩][↩]
- EPA. 1991a. June 03, 1991. Memorandum. 40 Page(s). William Dykstra. Toxicology Branch. Glyphosate; 2-Year combined chronic toxicity/carcinogenicity study in Sprague-Dawley rats – List A Pesticide for Reregistration Pages 29-40 removed-registrant data. MRID 416438-01. Tox review 008390. U.S. Environmental Protection Agency. https://archive.epa.gov/pesticides/chemicalsearch/chemical/foia/web/pdf/103601/103601-263.pdf[↩][↩][↩][↩][↩][↩]
- Caglar S, Kolankaya D. 2008. The effect of sub-acute and sub-chronic exposure of rats to the glyphosate-based herbicide Roundup. Environ Toxicol Pharmacol 25(1):57–62. 10.1016/j.etap.2007.08.011[↩]
- Jiang X, Zhang N, Yin L, et al. 2018. A commercial Roundup(R) formulation induced male germ cell apoptosis by promoting the expression of XAF1 in adult mice. Toxicol Lett 296:163–172. 10.1016/j.toxlet.2018.06.1067[↩]
- Dallegrave E, Mantese FD, Coelho RS, Pereira JD, Dalsenter PR, Langeloh A. The teratogenic potential of the herbicide glyphosate-Roundup in Wistar rats. Toxicol Lett. 2003 Apr 30;142(1-2):45-52. doi: 10.1016/s0378-4274(02)00483-6[↩][↩]
- Dallegrave E, Mantese FD, Oliveira RT, et al. 2007. Pre- and postnatal toxicity of the commercial glyphosate formulation in Wistar rats. Arch Toxicol 81(9):665–673. 10.1007/s00204-006-0170-5[↩]
- Romano RM, Romano MA, Bernardi MM, et al. 2010. Prepubertal exposure to commercial formulation of the herbicide glyphosate alters testosterone levels and testicular morphology. Arch Toxicol 84(4):309–317. 10.1007/s00204-009-0494-z[↩][↩]
- Milesi MM, Lorenz V, Pacini G, et al. 2018. Perinatal exposure to a glyphosate-based herbicide impairs female reproductive outcomes and induces second-generation adverse effects in Wistar rats. Arch Toxicol 92(8):2629–2643. 10.1007/s00204-018-2236-6[↩]
- EPA. 1985c. March 27, 1985. Memorandum. 5 Page(s). Stephen Saunders. Toxicology Branch. 4-week subchronic inhalation in rats with Roundup 33 1/3% use-dilution. EPA Reg. No. 524-308; Accession No. 252621. U.S. Environmental Protection Agency. https://archive.epa.gov/pesticides/chemicalsearch/chemical/foia/web/pdf/103601/103601-178.pdf[↩][↩][↩][↩][↩][↩][↩]
- EPA. 1992c. Data evaluation report. Test material: Glyphosate technical, white powder. 21-Day dermal toxicity study in rats, MRID 00098460. In: July 22, 1992. Memorandum. Glyphosate-List A chemical for reregistration-rereview of toxicology studies for acceptability. The attached 7-22-92 memorandum contains the Agency reviews for the following glyphosate studies: MRID numbers 0046362, 00046363, 00046364, 00067039, 00078619, 00078620, 00093879, 00098460, 00105995, 00132681, 00132683, 00132685 and 00132686 [Received via FOIA request]. HED pages 9–16. U.S. Environmental Protection Agency.[↩][↩][↩][↩]
- Altamirano GA, Delconte MB, Gomez AL, et al. 2018. Postnatal exposure to a glyphosate-based herbicide modifies mammary gland growth and development in Wistar male rats. Food Chem Toxicol 118:111–118. 10.1016/j.fct.2018.05.011[↩]
- Guerrero Schimpf M, Milesi MM, Ingaramo PI, et al. 2017. Neonatal exposure to a glyphosate based herbicide alters the development of the rat uterus. Toxicology 376:2–14. 10.1016/j.tox.2016.06.004[↩]
- Guerrero Schimpf M, Milesi MM, Luque EH, et al. 2018. Glyphosate-based herbicide enhances the uterine sensitivity to estradiol in rats. J Endocrinol. 10.1530/JOE-18-0207[↩]
- Kumar S, Khodoun M, Kettleson EM, et al. 2014. Glyphosate-rich air samples induce (L-33, TSLP and generate IL-13 dependent airway inflammation. Toxicology 325:42–51. 10.1016/j.tox.2014.08.008[↩][↩]
- Adam A, Marzuki A, Abdul Rahman H, Abdul Aziz M. The oral and intratracheal toxicities of ROUNDUP and its components to rats. Vet Hum Toxicol. 1997 Jun;39(3):147-51.[↩][↩][↩]
- Dar MA, Sultana M, Mir AH, et al. 2019. Effect of Repeated Oral Administration of Roundup and Ammonium Nitrate on Liver of Wistar Rats. Proc Natl Acad Sci India Sect B 89(2):505–510. 10.1007/s40011-017-0961-x[↩][↩]
- Aitbali Y, Ba-M’hamed S, Elhidar N, et al. 2018. Glyphosate based-herbicide exposure affects gut microbiota, anxiety and depression-like behaviors in mice. Neurotoxicol Teratol 67:44–49. 10.1016/j.ntt.2018.04.002[↩][↩][↩]
- Lozano VL, Defarge N, Rocque L-M, et al. 2018. Sex-dependent impact of Roundup on the rat gut microbiome. Toxicol Rep 5:96–107. 10.1016/j.toxrep.2017.12.005[↩][↩]
- Mao Q, Manservisi F, Panzacchi S, et al. 2018. The Ramazzini Institute 13-week pilot study of glyphosate and Roundup administered at human-equivalent dose to Sprague Dawley rats: Effects on the microbiome. Environ Health 17(1):50. 10.1186/s12940-018-0394-x[↩][↩]
- EPA. 2013c. Memorandum. 04-Jun-2013. Memorandum: Glyphosate. Review and generation of data evaluation records. MRID numbers 44320610, 44320612. [Received via FOIA request]. U.S. Environmental Protection Agency.[↩][↩]
- EPA. 1992f. Data evaluation report. Test material: Glyphosate, technical; 98.7% purity; lot XHJ-64; white powder. Teratology study in rabbits. MRID 00046363. In: July 22, 1992. Memorandum. Glyphosate-List A chemical for reregistration-rereview of toxicology studies for acceptability. The attached 7-22-92 memorandum contains the Agency reviews for the following glyphosate studies: MRID numbers 0046362, 00046363, 00046364, 00067039, 00078619, 00078620, 00093879, 00098460, 00105995, 00132681, 00132683, 00132685 and 00132686 [Received via FOIA request]. HED pages 50–58. U.S. Environmental Protection Agency.[↩][↩]
- EPA. 1991b. December 13, 1991. Memorandum. 38 Page(s). William Dykstra. Toxicology Branch I. Glyphosate – EPA Registration No. 524-308 – 2-Year chronic feeding/oncogenicity study in rats with technical glyphosate. MRID 416438-01. Tox review 008897. U.S. Environmental Protection Agency. https://archive.epa.gov/pesticides/chemicalsearch/chemical/foia/web/pdf/103601/103601-268.pdf[↩][↩][↩][↩][↩]
- EPA. 1992d. Data evaluation report. Test material: Glyphosate, technical; 98.7% purity; lot XHJ-64; white powder. A lifetime feeding study of glyphosate in rats. MRID 00098460. In: July 22, 1992. Memorandum. Glyphosate-List A chemical for reregistration-rereview of toxicology studies for acceptability. The attached 7-22-92 memorandum contains the agency reviews for the following glyphosate studies: MRID numbers 0046362, 00046363, 00046364, 00067039, 00078619, 00078620, 00093879, 00098460, 00105995, 00132681, 00132683, 00132685 and 00132686 [Received via FOIA request]. HED pages 17–40. U.S. Environmental Protection Agency.[↩][↩][↩][↩]
- EPA. 1985a. April 03, 1985. Memorandum. 4 Page(s). William Dykstra. Toxicology Branch. Glyphosate; EPA Reg. # 524-308; mouse oncogenicity study Accession No. 251007-014. Tox review 004370. U.S. Environmental Protection Agency. https://archive.epa.gov/pesticides/chemicalsearch/chemical/foia/web/pdf/103601/103601-183.pdf[↩]
- EPA. 2015a. Memorandum. November 17, 2015. Glyphosate, updated data evaluation record for a mouse carcinogenicity study. MRID 00130406 [Received via FOIA request]. U.S. Environmental Protection Agency.[↩][↩][↩][↩]
- EPA. 2015c. Memorandum. November 17, 2015. Glyphosate. Review and generation of data evaluation records for three rodent carcinogenicity studies. MRID numbers 49707601, 49631701, 49631702. U.S. Environmental Protection Agency.[↩][↩][↩][↩]
- Milic M, Zunec S, Micek V, et al. 2018. Oxidative stress, cholinesterase activity, and DNA damage in the liver, whole blood, and plasma of Wistar rats following a 28-day exposure to glyphosate. Arh Hig Rada Toksikol 69(2):154–168. 10.2478/aiht-2018-69-3114[↩][↩][↩]
- Wang L, Deng Q, Hu H, et al. 2019. Glyphosate induces benign monoclonal gammopathy and promotes multiple myeloma progression in mice. J Hematol Oncol 12(1):70. 10.1186/s13045-019-0767-9[↩][↩][↩][↩]
- Benedetti AL, de Lourdes C, Trentin AG, et al. 2004. The effects of sub-chronic exposure of Wistar rats to the herbicide Glyphosate-Biocarb. Toxicol Lett 153(2):227–232. 10.1016/j.toxlet.2004.04.008[↩]
- Mesnage R, Antoniou MN, Renney G, et al. 2017. Multiomics reveal non-alcoholic fatty liver disease in rats following chronic exposure to an ultra-low dose of Roundup herbicide. Sci Rep 7:39328. 10.1038/srep39328[↩][↩][↩]
- Wunnapuk K, Gobe G, Endre Z, et al. 2014. Use of a glyphosate-based herbicide-induced nephrotoxicity model to investigate a panel of kidney injury biomarkers. Toxicol Lett 225(1):192–200. 10.1016/j.toxlet.2013.12.009[↩]
- EPA. 1986a. March 12, 1986. Memorandum. 4 Page(s). William Dykstra. Toxicology Branch. EPA Reg. No. 524-308; Glyphosate; miscellaneous data; one-year dog study. Accession No. 260021 Tox review 004975. U.S. Environmental Protection Agency. https://archive.epa.gov/pesticides/chemicalsearch/chemical/foia/web/pdf/103601/103601-212.pdf[↩][↩]
- EPA. 1987. January 12, 1987. Memorandum. 5 Page(s). William Dykstra. Toxicology Branch. Glyphosate; Roundup; EPA Reg. No. 524-308; Addendum to one year dog study with glyphosate; PP# 6F3380/6H5502; Glyphosate in/on soybeans; revised Section F; and amended label text. Acc 264334. Tox review 005651 (excerpt). U.S. Environmental Protection Agency. https://archive.epa.gov/pesticides/chemicalsearch/chemical/foia/web/pdf/103601/103601-229.pdf[↩]
- FAO and WHO. 2016. Pesticides residues in food 2016. Special session of the Joint FAO/WHO meeting on pesticide residues. FAO plant production and protection paper. Food and Agriculture Organization of the United Nations, World Health Organization. http://www.fao.org/3/a-i5693e.pdf[↩][↩]
- Tizhe E, Ibrahim N, Fatihu M, et al. 2018. Pancreatic function and histoarchitecture in Wistar rats following chronic exposure to Bushfire(R): the mitigating role of zinc. J Int Med Res 46(8):3296–3305. 10.1177/0300060518778640[↩]
- Romano MA, Romano RM, Santos LD, et al. 2012. Glyphosate impairs male offspring reproductive development by disrupting gonadotropin expression. Arch Toxicol 86(4):663–673. 10.1007/s00204-011-0788-9[↩][↩]
- Owagboriaye FO, Dedeke GA, Ademolu KO, et al. 2017. Reproductive toxicity of Roundup herbicide exposure in male albino rat. Exp Toxicol Pathol 69(7):461–468. 10.1016/j.etp.2017.04.007[↩]
- Johansson HKL, Schwartz CL, Nielsen LN, et al. 2018. Exposure to a glyphosate-based herbicide formulation, but not glyphosate alone, has only minor effects on adult rat testis. Reprod Toxicol 82:25–31. 10.1016/j.reprotox.2018.09.008[↩][↩]
- Varayoud J, Durando M, Ramos JG, et al. 2017. Effects of a glyphosate-based herbicide on the uterus of adult ovariectomized rats. Environ Toxicol 32(4):1191–1201. 10.1002/tox.22316[↩]
- Ait Bali Y, Kaikai N, Ba-M’hamed S, et al. 2019. Learning and memory impairments associated to acetylcholinesterase inhibition and oxidative stress following glyphosate based-herbicide exposure in mice. Toxicology 415:18–525. 10.1016/j.tox.2019.01.010[↩][↩][↩][↩]
- (Cattani D, Cesconetto PA, Tavares MK, et al. 2017. Developmental exposure to glyphosate-based herbicide and depressive-like behavior in adult offspring: Implication of glutamate excitotoxicity and oxidative stress. Toxicology 38767–80. 10.1016/j.tox.2017.06.001[↩]
- Martinez MA, Ares I, Rodriguez JL, et al. 2018. Neurotransmitter changes in rat brain regions following glyphosate exposure. Environ Res 161:212–219. 10.1016/j.envres.2017.10.051[↩][↩]
- Dechartres J, Pawluski JL, Gueguen MM, Jablaoui A, Maguin E, Rhimi M, Charlier TD. Glyphosate and glyphosate-based herbicide exposure during the peripartum period affects maternal brain plasticity, maternal behaviour and microbiome. J Neuroendocrinol. 2019 Sep;31(9):e12731. doi: 10.1111/jne.12731[↩]
- Szepanowski F, Szepanowski LP, Mausberg AK, et al. 2018. Differential impact of pure glyphosate and glyphosate-based herbicide in a model of peripheral nervous system myelination. Acta Neuropathol 136(6):979–982. 10.1007/s00401-018-1938-4[↩][↩][↩][↩][↩][↩]
- APVMA. 2017. Final regulatory position: Consideration of the evidence for a formal reconsideration of glyphosate. Australian Pesticides and Veterinary Medicines Authority. http://apvma.gov.au/sites/default/files/publication/26561-glyphosate-final-regulatory-position-report-final_0.pdf[↩]
- Brusick D, Aardema M, Kier L, et al. 2016. Genotoxicity Expert Panel review: Weight of evidence evaluation of the genotoxicity of glyphosate, glyphosate-based formulations, and aminomethylphosphonic acid. Crit Rev Toxicol 46(Supp1):56–74. 10.1080/10408444.2016.1214680[↩]
- EFSA. 2015. Conclusion on the peer review of the pesticide risk assessment of the active substance glyphosate. EFSA (Journal European Food Safety Authority) 13(11):4302. http://onlinelibrary.wiley.com/doi/10.2903/j.efsa.2015.4302/epdf[↩][↩]
- EPA. 2017c. Revised glyphosate issue paper. Evaluation of carcinogenic potential. December 12, 2017. U.S. Environmental Protection Agency, Office of Pesticide Programs.[↩]
- Health Canada. 2017. Re-evaluation decision. Glyphosate. Ottawa, Ontario: Pest Management Regulatory Agency, Health Canada. http://publications.gc.ca/collections/collection_2017/sc-hc/H113-28/H113-28-2017-1-eng.pdf[↩]
- Kier LD, Kirkland DJ. 2013. Review of genotoxicity studies of glyphosate and glyphosate-based formulations. Crit Rev Toxicol 43(4):283–315. 10.3109/10408444.2013.770820[↩]
- NZ EPA. 2016. Review of the evidence relating to glyphosate and carcinogenicity. New Zealand Environmental Protection Agency. http://www.epa.govt.nz/Publications/EPA_glyphosate_review.pdf[↩]
- Williams GM, Aardema M, Acquavella J, Berry SC, Brusick D, Burns MM, de Camargo JLV, Garabrant D, Greim HA, Kier LD, Kirkland DJ, Marsh G, Solomon KR, Sorahan T, Roberts A, Weed DL. A review of the carcinogenic potential of glyphosate by four independent expert panels and comparison to the IARC assessment. Crit Rev Toxicol. 2016 Sep;46(sup1):3-20. doi: 10.1080/10408444.2016.1214677. Erratum in: Crit Rev Toxicol. 2018 Nov;48(10):907-908. doi: 10.1080/10408444.2018.1522175[↩]
- IARC. 2017. Glyphosate. Some organophosphate insecticides and herbicides. In: IARC monographs on the evaluation of carcinogenic risks to humans. Volume 112. International Agency for Research on Cancer. http://monographs.iarc.fr/ENG/Monographs/vol112/mono112.pdf[↩]
- Chen YJ, Wu ML, Deng JF, et al. 2009. The epidemiology of glyphosate-surfactant herbicide poisoning in Taiwan, 1986-2007: A poison center study. Clin Toxicol (Phila) 47(7):670–677. 10.1080/15563650903140399[↩][↩]
- Kim YH, Lee JH, Hong CK, et al. 2014. Heart rate-corrected QT interval predicts mortality in glyphosate-surfactant herbicide-poisoned patients. Am J Emerg Med 32(3):203–207. 10.1016/j.ajem.2013.09.025[↩][↩]
- Moon JM, Chun BJ, Cho YS, et al. 2018. Cardiovascular effects and fatality may differ according to the formulation of glyphosate salt herbicide. Cardiovasc Toxicol 18(1):99–107. 10.1007/s12012-017-9418-y[↩][↩][↩]
- Roberts DM, Buckley NA, Mohamed F, et al. 2010. A prospective observational study of the clinical toxicology of glyphosate-containing herbicides in adults with acute self-poisoning. Clin Toxicol (Phila) 48(2):129–136. 10.3109/15563650903476491[↩][↩][↩]
- Sawada Y, Nagai Y, Ueyama M, Yamamoto I. Probable toxicity of surface-active agent in commercial herbicide containing glyphosate. Lancet. 1988 Feb 6;1(8580):299. doi: 10.1016/s0140-6736(88)90379-0[↩][↩][↩]
- Talbot AR, Shiaw MH, Huang JS, Yang SF, Goo TS, Wang SH, Chen CL, Sanford TR. Acute poisoning with a glyphosate-surfactant herbicide (‘Roundup’): a review of 93 cases. Hum Exp Toxicol. 1991 Jan;10(1):1-8. doi: 10.1177/096032719101000101[↩][↩][↩][↩]
- Tominack RL, Yang GY, Tsai WJ, Chung HM, Deng JF. Taiwan National Poison Center survey of glyphosate–surfactant herbicide ingestions. J Toxicol Clin Toxicol. 1991;29(1):91-109. doi: 10.3109/15563659109038601[↩][↩][↩][↩]
- Cho YS, Moon JM, Chun BJ, et al. 2019. Use of qSOFA score in predicting the outcomes of patients with glyphosate surfactant herbicide poisoning immediately upon arrival at the emergency department. Shock 51(4):447–452. 10.1097/SHK.0000000000001201[↩][↩][↩][↩]
- EPA. 1992b. Data evaluation report. Test material: Glyphosate, technical; sample No. 96. Toxicological investigation of: CP67573-3, MRID 00067039. In: July 22, 1992. Memorandum. Glyphosate-List A chemical for reregistration-rereview of toxicology studies for acceptability. The attached 7-22-92 memorandum contains the agency reviews for the following glyphosate studies: MRID numbers 0046362, 00046363, 00046364, 00067039, 00078619, 00078620, 00093879, 00098460, 00105995, 00132681, 00132683, 00132685 and 00132686 [Received via FOIA request]. HED pages 3–5. U.S. Environmental Protection Agency.[↩]
- Hoppin JA, Valcin M, Henneberger PK, et al. 2007. Pesticide use and chronic bronchitis among farmers in the Agricultural Health Study. Am J Ind Med 50(12):969–979. 10.1002/ajim.20523[↩][↩]
- Moon JM, Chun BJ. 2010. Predicting acute complicated glyphosate intoxication in the emergency department. Clin Toxicol (Phila) 48(7):718–724. 10.3109/15563650.2010.488640[↩][↩]
- Picetti E, Generali M, Mensi F, et al. 2018. Glyphosate ingestion causing multiple organ failure: A near-fatal case report. Acta Biomed 88(4):533–537. 10.23750/abm.v88i4.6322[↩][↩][↩][↩]
- Lee HL, Chen KW, Chi CH, Huang JJ, Tsai LM. Clinical presentations and prognostic factors of a glyphosate-surfactant herbicide intoxication: a review of 131 cases. Acad Emerg Med. 2000 Aug;7(8):906-10. doi: 10.1111/j.1553-2712.2000.tb02069.x[↩][↩][↩]
- Eriguchi M, Iida K, Ikeda S, et al. 2019. Parkinsonism relating to intoxication with glyphosate. Intern Med 58(13):1935–1938. 10.2169/internalmedicine.2028-18[↩][↩]
- Luo W, Lu T, Li F, et al. 2019. Surgical treatment of pyloric stenosis caused by glyphosate poisoning: A case report. Medicine (Baltimore) 98(30):e16590. 10.1097/MD.0000000000016590[↩][↩][↩]
- Lee CH, Shih CP, Hsu KH, et al. 2008. The early prognostic factors of glyphosate-surfactant intoxication. Am J Emerg Med 26(3):275–281. 10.1016/j.ajem.2007.05.011[↩]
- Chang CY, Peng YC, Hung DZ, Hu WH, Yang DY, Lin TJ. Clinical impact of upper gastrointestinal tract injuries in glyphosate-surfactant oral intoxication. Hum Exp Toxicol. 1999 Aug;18(8):475-8. doi: 10.1191/096032799678847078[↩]
- Ozaki T, Sofue T, Kuroda Y. 2017. Severe glyphosate-surfactant intoxication successfully treated with continuous hemodiafiltration and direct hemoperfusion: Case report. Ther Apher Dial 21(3):296–297. 10.1111/1744-9987.12565[↩][↩][↩]
- Jayasumana C, Paranagama P, Agampodi S, et al. 2015. Drinking well water and occupational exposure to herbicides is associated with chronic kidney disease in Padavi-Sripura, Sri Lanka. Environ Health 14(1):6. 10.1186/1476-069x-14-6[↩]
- Elsner P, Darr-Foit S, Schliemann S. 2018. Occupational koebnerization of psoriasis caused by glyphosate. J Dtsch Dermatol Ges 16(1):70–71. 10.1111/ddg.13393[↩]
- EPA. 1993. Reregistration Eligibility Decision (RED): Glyphosate. U.S. Environmental Protection Agency. EPA738R93014. https://www3.epa.gov/pesticides/chem_search/reg_actions/reregistration/red_PC-417300_1-Sep-93.pdf[↩][↩]
- Acquavella JF, Weber JA, Cullen MR, Cruz OA, Martens MA, Holden LR, Riordan S, Thompson M, Farmer D. Human ocular effects from self-reported exposures to Roundup herbicides. Hum Exp Toxicol. 1999 Aug;18(8):479-86. doi: 10.1191/096032799678847087[↩][↩][↩]
- EPA. 2015b. Memorandum. June 29, 2015. EDSP weight of evidence conclusions on the tier 1 screening assays for the list 1 chemicals. EDSP: Weight of evidence analysis of potential Minteraction with the estrogen, androgen or thyroid pathways. U.S. Environmental Protection Agency. https://www.regulations.gov/contentStreamer?documentId=EPA-HQ-OPP-2009-0361-0047&disposition=attachment&contentType=pdf[↩][↩][↩]
- EPA. 2015d. Memorandum. June 29, 2015. Glyphosate. Data Evaluation Records (DERs) for EDSP Tier 1 Assays. U.S. Environmental Protection Agency. https://www.regulations.gov/contentStreamer?documentId=EPA-HQ-OPP-2009-0361-0047&disposition=attachment&contentType=pdf[↩]
- Curtis KM, Savitz DA, Weinberg CR, Arbuckle TE. The effect of pesticide exposure on time to pregnancy. Epidemiology. 1999 Mar;10(2):112-7. Erratum in: Epidemiology 1999 Jul;10(4):470.[↩][↩]
- Sanin LH, Carrasquilla G, Solomon KR, et al. 2009. Regional differences in time to pregnancy among fertile women from five Colombian regions with different use of glyphosate. J Toxicol Environ Health A 72(15–16):949–960. 10.1080/15287390902929691[↩]
- Caballero M, Amiri S, Denney JT, et al. 2018. Estimated residential exposure to agricultural chemicals and premature mortality by Parkinson’s disease in Washington State. Int J Environ Res Public Health 15(12). 10.3390/ijerph15122885[↩]
- Key S, Ma JK-C, Drake PM. Genetically modified plants and human health. Journal of the Royal Society of Medicine. 2008;101(6):290-298. doi:10.1258/jrsm.2008.070372. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2408621/[↩]
- WHO/FAO. Strategies for assessing the safety of foods produced by biotechnology. Report of the Joint WHO/FAO Consultation. Geneva: FAO/WHO; 1991.[↩]
- FDA Statement of policy, foods derived from new plant varieties. Fed Reg. 1992;57[↩]
- OECD. Food Safety Evaluation. Paris: OECD Documents; 1996.[↩]
- Malarkey T. Human Health concerns with GM crops. Mut Res. 2003;544:217–22. https://www.ncbi.nlm.nih.gov/pubmed/14644323[↩]
- Prescott VE, Campbell PM, Moore A, et al. Transgenic expression of bean α-amylase inhibitor in peas results in altered structure and immunogenicity. J Agric Food Chem. 2005;53:9023–30. https://www.ncbi.nlm.nih.gov/pubmed/16277398[↩]
- Nordlee JA, Taylor SL, Townsend JA, Thomas LA, Bush RK. Identification of a brazil-nut allergen in GM soybeans. NEJM. 1996;334:688–92. https://www.ncbi.nlm.nih.gov/pubmed/8594427[↩]
- Herman E. Soybean allergenicity and suppression of the immunodominant allergen. Crop Sci. 2005;45:462–7.[↩]
- Assessing the survival of transgenic plant DNA in the human gastrointestinal tract. Netherwood T, Martín-Orúe SM, O’Donnell AG, Gockling S, Graham J, Mathers JC, Gilbert HJ. Nat Biotechnol. 2004 Feb; 22(2):204-9. https://www.ncbi.nlm.nih.gov/pubmed/14730317/[↩]
- Animal nutrition with feeds from genetically modified plants. Flachowsky G, Chesson A, Aulrich K. Arch Anim Nutr. 2005 Feb; 59(1):1-40. https://www.ncbi.nlm.nih.gov/pubmed/15889650/[↩]
- The digestive fate of Escherichia coli glutamate dehydrogenase deoxyribonucleic acid from transgenic corn in diets fed to weanling pigs. Beagle JM, Apgar GA, Jones KL, Griswold KE, Radcliffe JS, Qiu X, Lightfoot DA, Iqbal MJ. J Anim Sci. 2006 Mar; 84(3):597-607. https://www.ncbi.nlm.nih.gov/pubmed/16478951/[↩]
- Identification of a Brazil-nut allergen in transgenic soybeans. Nordlee JA, Taylor SL, Townsend JA, Thomas LA, Bush RK. N Engl J Med. 1996 Mar 14; 334(11):688-92. https://www.ncbi.nlm.nih.gov/pubmed/8594427/[↩]
- Streit L. Association of the Brazil nut protein gene and Kunitz trypsin inhibitor alleles with soybean protease inhibitor activity and agronomic traits. Crop Sci. 2001;41:1757–1760. doi: 10.2135/cropsci2001.1757.[↩]
- U.S. Cancer Statistics Working Group. United States Cancer Statistics: 1999-2014 Incidence and Mortality Web-based Report. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention and National Cancer Institute; 2017. https://nccd.cdc.gov/uscs/toptencancers.aspx[↩][↩][↩]
- Assessment of the health impact of GM plant diets in long-term and multigenerational animal feeding trials: a literature review. Snell C, Bernheim A, Bergé JB, Kuntz M, Pascal G, Paris A, Ricroch AE. Food Chem Toxicol. 2012 Mar; 50(3-4):1134-48. https://www.ncbi.nlm.nih.gov/pubmed/22155268/[↩]
- U.S. Food and Drug Administration. FDA Has Determined That the AquAdvantage Salmon is as Safe to Eat as Non-GE Salmon. https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm472487.htm[↩][↩]
- U.S. Food and Drug Administration. FDA takes several actions involving genetically engineered plants and animals for food. https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm473249.htm[↩]
- U.S. Food and Drug Administration. Questions and Answers on FDA’s Approval of AquAdvantage Salmon. https://www.fda.gov/AnimalVeterinary/DevelopmentApprovalProcess/GeneticEngineering/GeneticallyEngineeredAnimals/ucm473237.htm[↩][↩]
- de Araujo JS, Delgado IF, Paumgartten FJ. Glyphosate and adverse pregnancy outcomes, a systematic review of observational studies. BMC Public Health 2016;16:472. https://www.ncbi.nlm.nih.gov/pubmed/27267204[↩][↩]