Contents
What is asplenia
Asplenia means the absence of a spleen 1. Asplenia can occur in a variety of clinical settings, and it can refer to an anatomic absence of the spleen or functional asplenia secondary to a variety of disease states. The spleen is one of the primary extramedullary lymphoid organs. Spleen, organ of the lymphatic system located in the left side of the abdominal cavity under the diaphragm, the muscular partition between the abdomen and the chest. In humans it is about the size of a fist and is well supplied with blood. As the lymph nodes are filters for the lymphatic circulation, the spleen is the primary filtering element for the blood. The organ also plays an important role in storing and releasing certain types of immune cells that mediate tissue inflammation. Normally, the spleen is not palpable on physical exam and measures on average 10.65 by 5.16 cm. Normal spleen size can vary by gender and race. For example, men on average have larger spleens than females, and Caucasians, on average, tend to have larger spleens compared to African American individuals 2. When palpable or notably enlarged on imaging, the clinician must consider a variety of underlying disease states (whether acquired or inherited) contributing to the enlargement.
Asplenia can be caused by damage to the white pulp, the red pulp or both. The spleen is a direct and indirect site of potential toxicity that can secondarily arise from underlying disease processes or infection 3. One must understand and appreciate the various etiologies and potential complications of asplenia, such as being a great risk factor for encapsulated microorganisms. Bacterial infections with Neisseria meningitides or Streptococcal pneumoniae, among many others, can become life-threatening if untreated in patients with anatomic or functional asplenia 4. Patients without a spleen have a 200-fold greater risk of death from septicemia compared to those with an adequate functioning spleen. There are many case reports in the medical literature reporting of asplenic patients, unfortunately, succumbing to infectious disease leading to death. One notable example reported by Heubner ML et al. 5 described the rare case of a patient who had required splenectomy due to refractory immune thrombocytopenia (ITP). He subsequently developed a pneumococcal meningitis infection and died despite best supportive efforts. The rate of invasive bacterial infection and mortality in asplenic patients is significantly higher compared to normal controls – 69% in meningitis, 64% in septicemia, and 7% in Purpura fulminans 4.
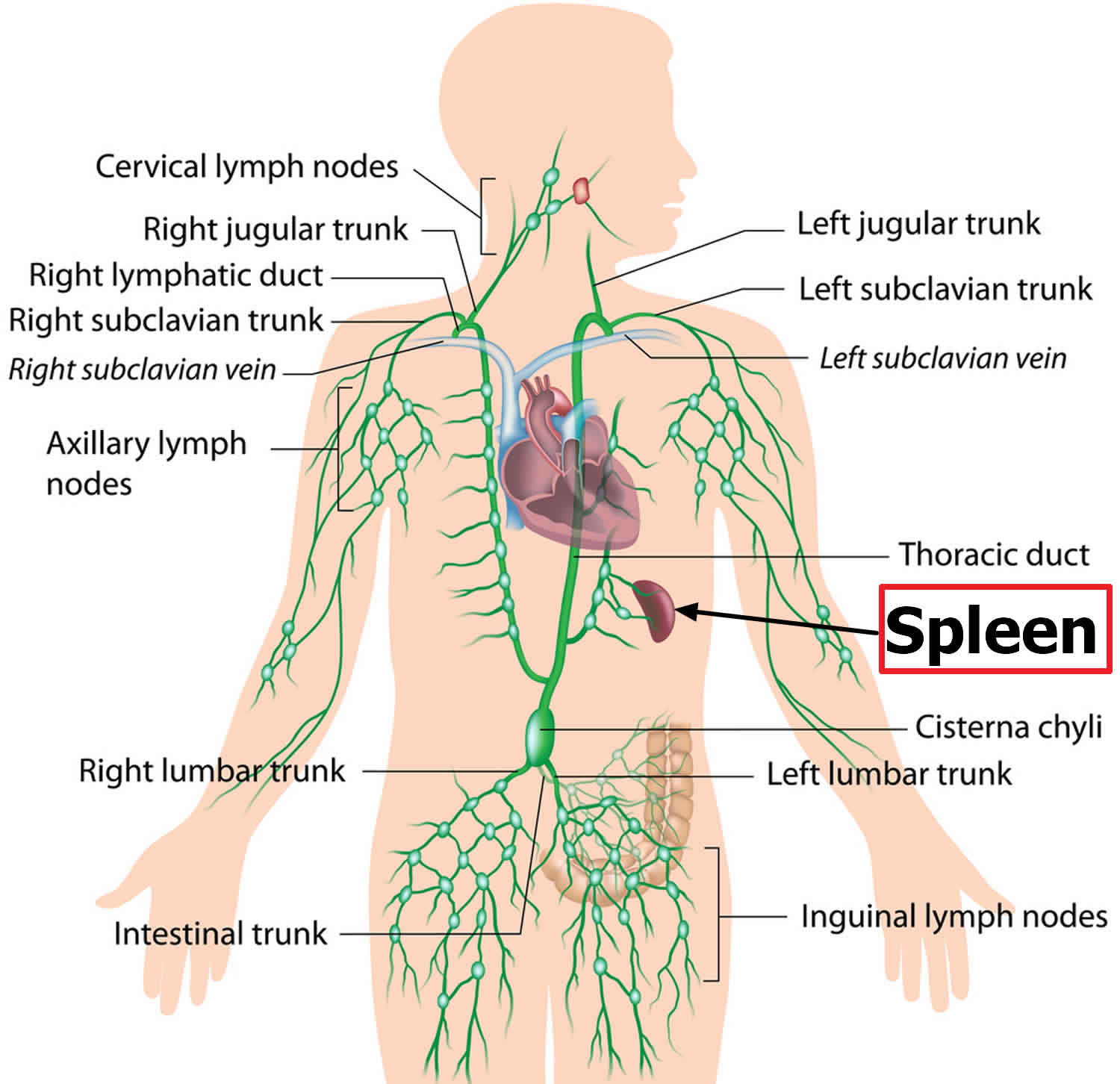
Functionally, the spleen’s primary physiologic role is the filtration and processing of senescent blood cells (predominantly red blood cells) and immunologically helps protect against encapsulated microorganisms and response to infectious pathogens. The spleen contains both hematopoietic and lymphopoietic elements, providing a basis for extramedullary hematopoiesis when necessary 6. The spleen has two functionally and histologically distinct tissues where these processes take place: the white pulp and the red pulp. The white pulp has a large mass of lymphoid tissue that produces antibodies against recognized antigens, whereas the red pulp has a tight network of sinusoids called the cord of Billroth which helps in blood filtration 7. As the body’s largest filter of blood, it helps to remove old red blood cells from the circulation aiding in the removal of bloodborne microorganisms 8. Beyond the spleen, the lymphatic system of the body consists of a complex myriad of lymphatic vessels, lymph nodes, and other specific extramedullary lymphoid organs including the thymus, tonsils, and appendix, which collectively play a very important role in immune defenses.
Figure 1. Spleen and lymphatic system
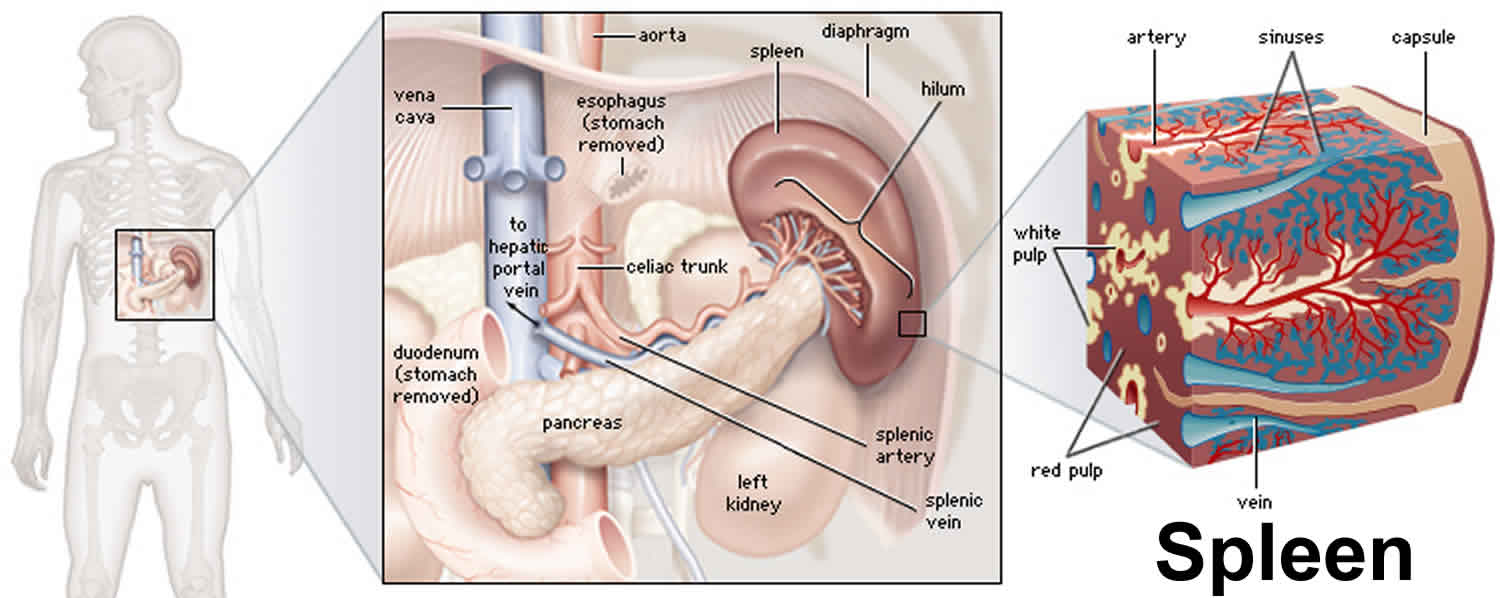
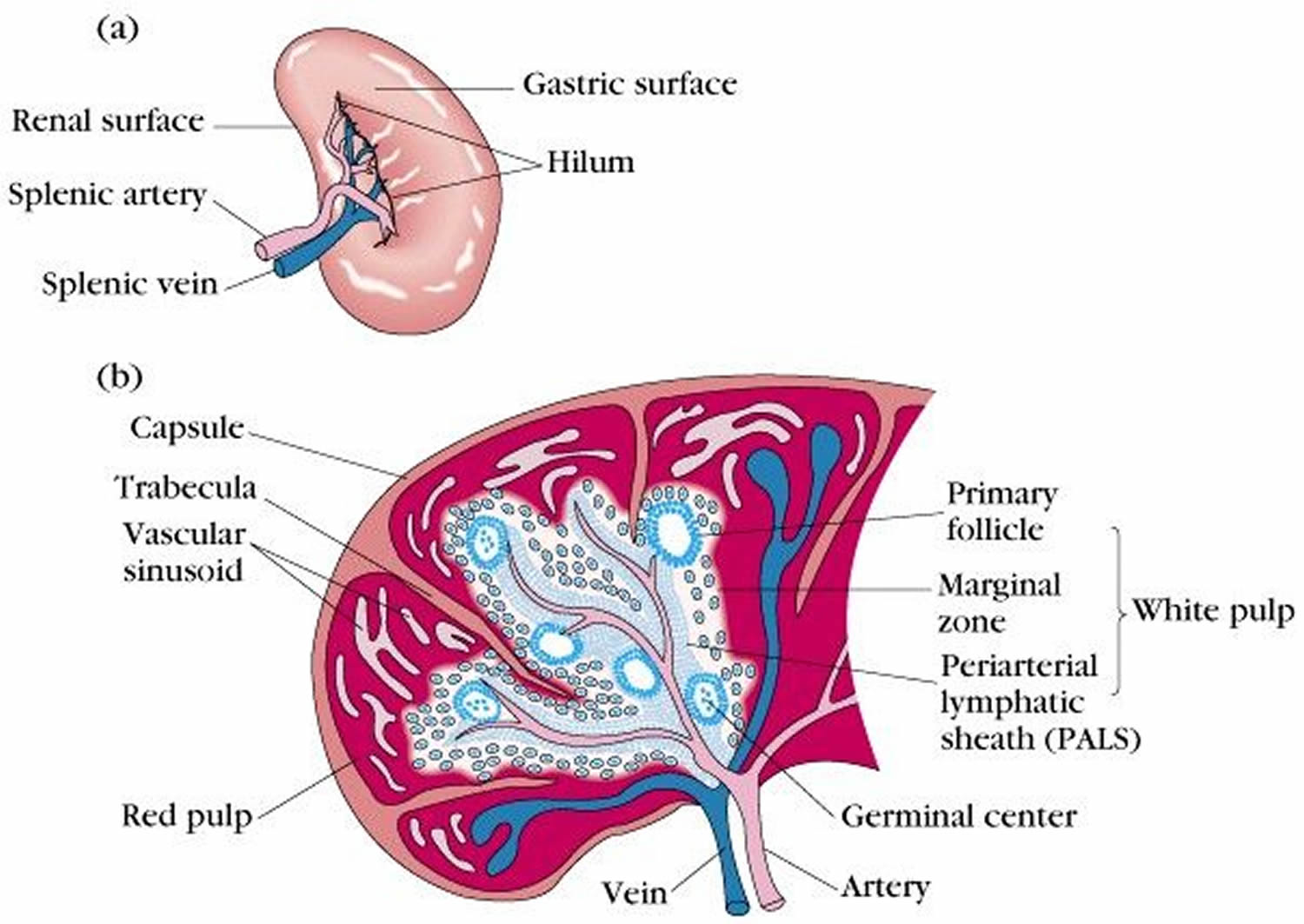
Figure 2. Spleen anatomy
What is the function of spleen?
The spleen, a spongy, soft organ encased in a thick connective-tissue capsule about as big as a person’s fist, is located in the upper left part of the abdomen, just under the rib cage. The splenic artery brings blood to the spleen from the heart. Blood leaves the spleen through the splenic vein, which drains into a larger vein (the portal vein) that carries the blood to the liver. The spleen has a covering of fibrous tissue (the splenic capsule) that supports its blood vessels and lymphatic vessels.
The spleen is made up of two basic types of tissue, the red pulp and the white pulp, each with different functions:
- White pulp: The white pulp is lymphoid tissue that usually surrounds splenic blood vessels. The white pulp of the spleen contains typical lymphoid elements, such as plasma cells, lymphocytes, and lymphatic nodules, called follicles in the spleen. The white pulp is part of the infection-fighting system (immune system). It produces white blood cells called lymphocytes, which in turn produce antibodies (specialized proteins that protect against invasion by foreign substances). Germinal centres in the white pulp serve as the sites of lymphocyte production.
- Red pulp: The red pulp is a network of splenic cords (cords of Billroth) and sinusoids (wide vessels) filled with blood, and it is in the red pulp that most of the filtration occurs. The red pulp filters the blood, removing unwanted material. The red pulp contains other white blood cells called phagocytes that ingest microorganisms, such as bacteria, fungi, and viruses. It also monitors red blood cells, destroying those that are abnormal or too old or damaged to function properly. In addition, the red pulp serves as a reservoir for different elements of the blood, especially white blood cells and platelets (cell-like particles involved in clotting). However, releasing these elements is a minor function of the red pulp.
Similar to the lymph nodes, the spleen reacts to microorganisms and other antigens that reach the bloodstream by releasing special phagocytic cells known as macrophages. Splenic macrophages reside in both red and white pulp, and they serve to remove foreign material from the blood and to initiate an immune reaction that results in the production of antibodies.
What causes asplenia?
Asplenia can be acquired, functionally reduced despite its anatomic presence, or in those who congenitally lack a spleen at birth (isolated congenital asplenia) 9. Most commonly, acquired asplenia is due to surgical removal or trauma. According to a survey by the National Hospital Discharge Survey in the United States done in 2005, approximately 22,000 splenectomies were performed 10. The most common indication for splenectomy was due to preceding trauma. According to a retrospective study that reviewed the historic clinical indications for splenectomy, traumatic injury accounted for more than 41.5%; hematologic malignancy accounted for 15.4%, and cytopenias accounted for 15.4% of all splenectomies performed 11.
A variety of benign and malignant hematologic disorders can result in the potential need for splenectomy. Some examples include hereditary spherocytosis, sickle cell disease, thalassemia major or intermedia, refractory immune thrombocytopenia (ITP), myeloproliferative disorders (e.g., severe or symptomatic splenomegaly in myelofibrosis), certain types of autoimmune hemolytic anemias, lymphoproliferative disorders, and rarely in thrombotic thrombocytopenic purpura (TTP) 12.
Congenital asplenia may be isolated or as a clinical sequela of broader (albeit rare) clinical syndromes such as Ivemark syndrome. Ivemark syndrome is seen in 1 in 10,000 to 40,000 of cases 13. This syndrome is classified under heterotaxy syndrome. It is associated with asplenia or hypoplasia of the spleen, malformation of the heart and abnormal arrangements of organs of the chest and abdomen 14.
In addition to the more common causes of asplenia, many other etiologies have been described in the literature affecting various body systems as listed below.
- Gastrointestinal (GI) disorders such as Celiac disease, Whipple disease, and inflammatory bowel diseases (IBD) can be associated with hypofunction of the spleen 15.
- Infectious disease states and acquired immune deficiency syndromes including HIV 15.
- Hepatic disorders such as alcoholic liver disease, cirrhosis, portal hypertension, and hepatorenal syndrome 15.
- Rheumatologic conditions such as systemic lupus erythematosus (SLE) have been associated with reduced spleen function 7.
Nearly 100% of sickle cell anemia patients (i.e., those with hemoglobin SS disease) will develop asplenia, and they are prone to overwhelming post-splenectomy infection 15. Many other patients with similar hemoglobinopathies are at risk of splenomegaly and a possible need for splenectomy or may develop functional asplenia with the course of their disease (e.g., hemoglobin SC disease).
Other diseases associated with reduced function of the spleen include celiac disease, with an estimated prevalence of 33% to 76%, Whipple disease is 47%, and alcoholic liver disease is 37% to 100% 15.
Asplenia pathophysiology
The pathophysiology of asplenia also varies depending on the underlying etiology and comorbid conditions. In many benign hematologic disorders, for example, functional asplenia is caused by persistent and recurrent hyper sequestration of red blood cells leading to red blood cell entrapment. This entrapment leads to splenic enlargement and then later atrophy. Profound atrophy of the spleen is also known as autosplenectomy. Autosplenectomy is usually seen in sickle cell anemia patients, especially those with HgbSS disease 15. In hematologic malignancies, by contrast, asplenia may result from direct infiltration of the malignant cells into the parenchyma of the spleen.
Many gastrointestinal disorders can lead to functional asplenia. For example, in cases of hepatic dysfunction or failure, asplenia can develop due to the disruption of the normal hepatic circulation, such as in portal hypertension. Alcohol consumption has been known to cause a direct toxic effect on splenic tissue 15. In other disorders like Celiac disease, asplenia is caused by excessive loss of lymphocytes through inflamed enteric mucosa, leading to reticuloendothelial atrophy 15.
Asplenia symptoms
Patients with asplenia can be asymptomatic, while some can present with malaise, fatigue, fever, and/or encephalopathy. On physical examination, a normal spleen is not palpable. When enlarged, it can typically be palpated below the costophrenic border on deep inspiration. In patients with various degrees of functional asplenia (autosplenectomy), the spleen will usually not be palpable due to chronic atrophy.
Patients may also present with signs or symptoms indicative of underlying infection. Severe infection due to Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria meningitides are the most common bacterial infections recognized in these patients. As such, it is important to assess for nuchal rigidity in these patients if they present with fever. In particular, pneumococcal meningitis can be up to four times more fatal in asplenic patients compared to other forms of bacterial meningitis, and over six times more likely to result in poor health outcomes 5.
Asplenia complications
The most common medical complication in asplenic patients is an overwhelming post-splenectomy infection 1. As noted, asplenic patients are at higher risk of infection with encapsulated microorganisms (e.g., streptococcal pneumonia). According to Kirkineska and colleagues 16, overwhelming post-splenectomy infection results in massive bacteremia caused by encapsulated microorganisms. The primary source of infection is not commonly identified, there is a short prodromal phase, and septic shock occasionally associated with disseminated intravascular coagulopathy (DIC) can occur. Up to 60% to 70% mortality rate has been observed in patients with overwhelming post-splenectomy infection if not appropriately managed 16.
In a normal functioning spleen, the red pulp filters blood and removes old red blood cells; it causes phagocytosis of infected erythrocytes which helps to form a defense against intraerythrocytic parasites 17. Parasitic infections such as babesiosis and malaria are common in patients with asplenia due to loss of phagocytosis.
The white pulp of the spleen has both T-cell and B-cell compartments which help with adaptive immunity and antibody production. The spleen provides the largest B cells which help in humoral immune response 17. The lack of memory B cells in the asplenic patient causes severe illness compared to the general population.
Because the spleen functions as a reservoir for non-circulation and undifferentiated monocytes, splenectomy causes lack of this reserve. According to Swirski et al. 18, these undifferentiated monocytes stored in clusters in the red pulp, respond to myocardial injury and help in wound healing.
Apart from its role in immunity, the spleen also has a role in vascular and thrombotic complications. Vascular complications were also observed mostly in patients with hematologic diseases like beta-thalassemia. In a survey done by Taher et al., they found that in patients with asplenia due to beta thalassemia, the prevalence of thrombotic event was 1.65% 19. There have also been increased incidences of stroke, myocardial infarction, and coronary artery disease in these patients 20. These vascular complications could be due to chronic inflammation and dysfunction of the endothelium. The destruction of the endothelial lining could lead to platelet activation and thrombosis 17.
Due to the risk of thrombosis in asplenic patient, another complication reported is pulmonary hypertension. According to Huebner et al 5., the incidence of pulmonary hypertension in asplenia patient is 8% to 11.5%, and numbers are even higher in patients with hemoglobinopathies 17. In patients with sickle cell or beta-thalassemia, the incidence could be as high 30% 21.
Patients also can develop adrenal hemorrhage also known as Waterhouse-Friedrichsen syndrome and Purpura fulminans 22. These two conditions require a high index of suspicion by the treating clinician. Early recognition and management are critical to avoid morbidity and mortality.
Asplenia diagnosis
As noted, analysis of the peripheral blood smear may demonstrate the presence of Howell-Jolly bodies providing the clinician with a clue of underlying splenic dysfunction (e.g., congenital or functional asplenia). To confirm physical exam findings and establish the anatomic presence (or absence) and measurement of the spleen various imaging modalities can be utilized including abdominal ultrasound (US), CT, MRI, or spleen scintigraphy 4. Using ultrasonography, CT or MRI of the abdomen may also help in better characterization of possible congenital or vascular abnormalities. Spleen scintigraphy is a specialized imaging modality using a 99-Tc labeled radio colloid that is specific for splenic tissue; a lack of uptake signifies asplenia.
Asplenia treatment
As noted, patients with asplenia are at a significantly increased risk of severe bacterial infection, especially from encapsulated microorganisms. As a result, efforts have been made over the last decade to improve the prevention, early detection, and management of infections encountered in asplenic patients. The most common bacterial pathogen leading to infection in patients with asplenia is Streptococcus pneumoniae. According to one study by Waghorn et al. 23, up to 87% of asplenic patients were found to have been infected with Streptococcus pneumoniae. To avoid morbidity, regardless of the cause of their asplenia, all patients should be counseled to receive vaccinations against these encapsulated bacteria. Vaccinations are recommended before splenectomy and after the surgical removal. For those with functional asplenia or autosplenectomy, it is also advised to continue aggressive vaccination schedules. Before planned surgery for splenectomy, it is recommended that patients receive the pneumococcal conjugate vaccine (PCV-13) 8 weeks in advance, as well as the pneumococcal polysaccharide vaccine (PPSV-23), Haemophilus influenzae type B vaccine (Hib), and the quadrivalent meningococcal conjugate vaccine 14 days before surgery 5.
Vaccination must continue in the post-operative period, and 5 years after the surgery it is recommended that patients receive the PPSV-23 and then again at age 65. The meningococcal conjugate vaccine should be repeated every 5 years 24. For those with acquired immunodeficiency, it may be advisable to check for titers to determine immune status. Apart from all these vaccines, patients should be encouraged to receive annual vaccination against the common strains of influenza. It has been recommended that prophylactic penicillin be considered in children with asplenia (e.g., patients with sickle cell disease) until the age of 5 17, although this prophylaxis is not recommended in adults. Asplenic adults, however, should have antibiotics readily available via their health care provider for use if they develop signs of infection as any delay in antibiotic access may be detrimental 5.
Overwhelming post-splenectomy infection is common especially in children younger than 16 years old due to their immune immaturity 16. Patients with asplenia who present with signs of infection should be immediately started on broad-spectrum antibiotics until further testing can isolate the causative microorganism. According to the Surviving Sepsis Campaign guidelines, patients suspected of sepsis should be started on antibiotics within 1 hour to avoid poor outcome 17. Aggressive intravenous (IV) hydration should also be promptly initiated as per standard sepsis guidelines and as part of supportive care. Since asplenic patients are prone to developing septic shock, they may also require vasopressors to maintain their blood pressure. In certain circumstances, mechanical ventilation may be necessary if patients develop respiratory failure.
Asplenia vaccines
If you do not have a spleen or your spleen does not work well, talk with your doctor about:
- Influenza vaccine each year to protect against seasonal flu
- DTaP or Tdap vaccine to protect against whooping cough and tetanus. Children younger than 7 years old get DTaP, while older children, teens, and adults get Tdap.
- Hib vaccine to protect against Haemophilus influenzae type b (Hib) if you were not previously vaccinated with the vaccine
- Pneumococcal vaccines (PCV13 and PPSV23) to protect against pneumonia and other pneumococcal disease. The pneumococcal conjugate vaccine (PCV13 or Prevnar 13®) protects against 13 types of pneumococcal bacteria. The Centers for Disease Control and Prevention (CDC) recommends PCV13 for use in infants and young children and adults 65 years or older. Older children and adults younger than 65 years old who are at increased risk for getting pneumococcal disease may also need a dose of PCV13. The pneumococcal polysaccharide vaccine (PPSV23 or Pneumovax®) protects against 23 types of pneumococcal bacteria. CDC recommends it for all adults 65 years or older and for those 2 years or older at increased risk for disease.
- Meningococcal vaccines (both types) to protect against meningitis and other meningococcal disease. There are two types of meningococcal vaccines licensed in the United States: Meningococcal conjugate vaccines (MenACWY) and Serogroup B meningococcal vaccines (MenB). These vaccines help prevent the most common causes of meningococcal disease in the United States. All 11 to 12 year olds should get a single dose of a MenACWY vaccine. CDC recommends a booster dose at age 16. The booster dose gives teens continued protection during the ages when they are at highest risk. If your teenager missed getting a dose, ask their clinician about getting it now. Teens and young adults (16 through 23 year olds) may also get a MenB vaccine, preferably at 16 through 18 years old. People need multiple doses of a MenB vaccine for best protection. People must get the same brand for all doses. Talk with your teen’s clinician if you are interested in MenB vaccination.
- Zoster vaccine to protect against shingles if you are 60 years and older. Two vaccines are licensed and recommended to prevent shingles in the U.S.. Zoster vaccine live (ZVL, Zostavax) has been in use since 2006. Recombinant zoster vaccine (RZV, Shingrix), has been in use since 2017 and is recommended by Advisory Committee on Immunization Practices as the preferred shingles vaccine.
- HPV vaccine series to protect against human papillomavirus if you are a man up to age 21 or woman up to age 26. Two doses of the HPV vaccine are recommended for all boys and girls at ages 11-12; the vaccine can be given as early as age 9. If you wait until they’re older, they may need three doses instead of two. Children who start the vaccine series on or after their 15th birthday need three shots given over 6 months. If your teen hasn’t gotten the vaccine yet, talk to his/her doctor about getting it as soon as possible. HPV vaccination is recommended through age 26 for women, and through age 21 for men, if they did not get vaccinated when they were younger.
- MMR vaccine to protect against measles, mumps, and rubella if you were born in 1957 or after and have not gotten this vaccine or do not have immunity to these diseases
- Varicella vaccine to protect against chickenpox if you were born in 1980 or after and have not gotten two doses of this vaccine or do not have immunity to this disease
Asplenia prognosis
The prognosis of asplenia is poor if left unrecognized, as patients will be at higher risk of infection without proper vaccination. Antibiotic prophylaxis and vaccinations are the recommended management in many asplenic patients and those with acquired immunodeficiency and asplenia 16. As noted, patients who do not receive immunizations are at high risk of severe bacterial infection, sepsis, septic shock, and death. One case report by Huebner and colleagues provides evidence of the poor prognosis in asplenic patients who succumb to infection despite receiving standard medical care. This case described a 45-year-old man with a history of splenectomy during childhood who presented with high fevers. He was found to have pneumococcal meningitis and was appropriately started on antibiotics and fluid resuscitation, though unfortunately his clinical status deteriorated quickly and was pronounced dead from sepsis 5. Many such illustrative cases and case series are reported in the medical literature.
- Ashorobi D, Fernandez R. Asplenia. [Updated 2019 Apr 21]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538171[↩][↩]
- Hosey RG, Mattacola CG, Kriss V, Armsey T, Quarles JD, Jagger J. Ultrasound assessment of spleen size in collegiate athletes. Br J Sports Med. 2006 Mar;40(3):251-4; discussion 251-4.[↩]
- Suttie AW. Histopathology of the spleen. Toxicol Pathol. 2006;34(5):466-503.[↩]
- Erdem SB, Genel F, Erdur B, Ozbek E, Gulez N, Mese T. Asplenia in children with congenital heart disease as a cause of poor outcome. Cent Eur J Immunol. 2015;40(2):266-9.[↩][↩][↩]
- Huebner ML, Milota KA. Asplenia and fever. Proc (Bayl Univ Med Cent). 2015 Jul;28(3):340-1.[↩][↩][↩][↩][↩][↩]
- Suttie AW. Histopathology of the spleen. Toxicol Pathol. 2006;34(5):466-503[↩]
- Crary SE, Buchanan GR. Vascular complications after splenectomy for hematologic disorders. Blood. 2009 Oct 01;114(14):2861-8[↩][↩]
- Mebius RE, Kraal G. Structure and function of the spleen. Nat. Rev. Immunol. 2005 Aug;5(8):606-16.[↩]
- Isolated congenital asplenia. https://ghr.nlm.nih.gov/condition/isolated-congenital-asplenia[↩]
- DeFrances CJ, Cullen KA, Kozak LJ. National Hospital Discharge Survey: 2005 annual summary with detailed diagnosis and procedure data. Vital Health Stat 13. 2007 Dec;(165):1-209.[↩]
- Rose AT, Newman MI, Debelak J, Pinson CW, Morris JA, Harley DD, Chapman WC. The incidence of splenectomy is decreasing: lessons learned from trauma experience. Am Surg. 2000 May;66(5):481-6.[↩]
- Crary SE, Buchanan GR. Vascular complications after splenectomy for hematologic disorders. Blood. 2009 Oct 01;114(14):2861-8.[↩]
- Erdem SB, Genel F, Erdur B, Ozbek E, Gulez N, Mese T. Asplenia in children with congenital heart disease as a cause of poor outcome. Cent Eur J Immunol. 2015;40(2):266-9[↩]
- Masiwal P, Chenthil KS, Priyadarsini B, Gnanaprakasam J, Srihari I. Ivemark Syndrome. J Assoc Physicians India. 2016 May;64(5):73-75.[↩]
- Kirkineska L, Perifanis V, Vasiliadis T. Functional hyposplenism. Hippokratia. 2014 Jan;18(1):7-11[↩][↩][↩][↩][↩][↩][↩][↩]
- Kirkineska L, Perifanis V, Vasiliadis T. Functional hyposplenism. Hippokratia. 2014 Jan;18(1):7-11.[↩][↩][↩][↩]
- O’Neal HR, Niven AS, Karam GH. Critical Illness in Patients With Asplenia. Chest. 2016 Dec;150(6):1394-1402.[↩][↩][↩][↩][↩][↩]
- Swirski FK, Nahrendorf M, Etzrodt M, Wildgruber M, Cortez-Retamozo V, Panizzi P, Figueiredo JL, Kohler RH, Chudnovskiy A, Waterman P, Aikawa E, Mempel TR, Libby P, Weissleder R, Pittet MJ. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science. 2009 Jul 31;325(5940):612-6.[↩]
- Taher A, Isma’eel H, Mehio G, Bignamini D, Kattamis A, Rachmilewitz EA, Cappellini MD. Prevalence of thromboembolic events among 8,860 patients with thalassaemia major and intermedia in the Mediterranean area and Iran. Thromb. Haemost. 2006 Oct;96(4):488-91.[↩]
- Schilling RF. Spherocytosis, splenectomy, strokes, and heat attacks. Lancet. 1997 Dec 06;350(9092):1677-8.[↩]
- Phrommintikul A, Sukonthasarn A, Kanjanavanit R, Nawarawong W. Splenectomy: a strong risk factor for pulmonary hypertension in patients with thalassaemia. Heart. 2006 Oct;92(10):1467-72.[↩]
- Hale AJ, LaSalvia M, Kirby JE, Kimball A, Baden R. Fatal purpura fulminans and Waterhouse-Friderichsen syndrome from fulminant Streptococcus pneumoniae sepsis in an asplenic young adult. IDCases. 2016;6:1-4.[↩]
- Waghorn DJ. Overwhelming infection in asplenic patients: current best practice preventive measures are not being followed. J. Clin. Pathol. 2001 Mar;54(3):214-8[↩]
- Rubin LG, Schaffner W. Clinical practice. Care of the asplenic patient. N. Engl. J. Med. 2014 Jul 24;371(4):349-56.[↩]