Contents
What is babesiosis
Babesiosis is an infectious disease caused by intraerythrocytic, tick-borne protozoa of the Babesia species that infect red blood cells and are spread by certain ticks 1. In the United States, tickborne transmission is most common in particular regions and seasons: it mainly occurs in parts of the Northeast and upper Midwest and usually peaks during the warm months. Babesiosis can range in severity from asymptomatic to life threatening. Although many people who are infected with Babesia do not have symptoms, for those who do effective treatment is available. Babesiosis is preventable, if simple steps are taken to reduce exposure to ticks.
Most cases of babesiosis occur in the Northeast and upper Midwest, particularly in parts of New England, New York state, New Jersey, Wisconsin, and Minnesota. In the Northeast, babesiosis occurs in both inland and coastal areas, including off-shore islands, such as Nantucket and Martha’s Vineyard (Massachusetts); Block Island (Rhode Island); and Shelter Island, Fire Island, and eastern Long Island (New York state).
Babesia species have been subdivided into four categories, known as clades 2. Babesia microti is the most prevalent and well-described species and is a classified as a Clade 1 organism. Babesia microti is transmitted by the bite of infected Ixodes scapularis ticks (also called deer tick or blacklegged tick)—typically, by the nymph stage of the tick, which is about the size of a poppy seed. Babesiosis is typically acquired by bites from ticks carrying the protozoa. As the parasite infects erythrocytes (red blood cells), the infection can be acquired through blood transfusion and therefore can occur in persons who have not traveled to endemic areas. Transplacental transmission has also been reported 3. Occasional (sporadic) cases of babesiosis caused by other Babesia species have been detected.
People can get infected with Babesia parasites in several ways:
- The main way is through the bite of an infected tick—during outdoor activities in areas where babesiosis is found (see below).
- A less common way is by getting a transfusion from a blood donor who has a Babesia infection but does not have any symptoms. (No tests have been licensed yet for screening blood donors for Babesia.)
- Rare cases of congenital transmission—from an infected mother to her baby (during pregnancy or delivery)—have been reported.
Babesia parasites are not transmitted from person-to-person like the flu or the common cold.
Babesia microti is transmitted in nature by Ixodes scapularis ticks (also called blacklegged ticks or deer ticks).
- Tickborne transmission primarily occurs in the Northeast and upper Midwest, especially in parts of New England, New York state, New Jersey, Wisconsin, and Minnesota.
- The parasite typically is spread by the young nymph stage of the tick, which is most apt to be found (seeking or “questing” for a blood meal) during warm months (spring and summer), in areas with woods, brush, or grass.
- Infected people might not recall a tick bite because Ixodes scapularis nymphs are very small (about the size of a poppy seed).
Figure 1. Ixodes scapularis tick (adult female)
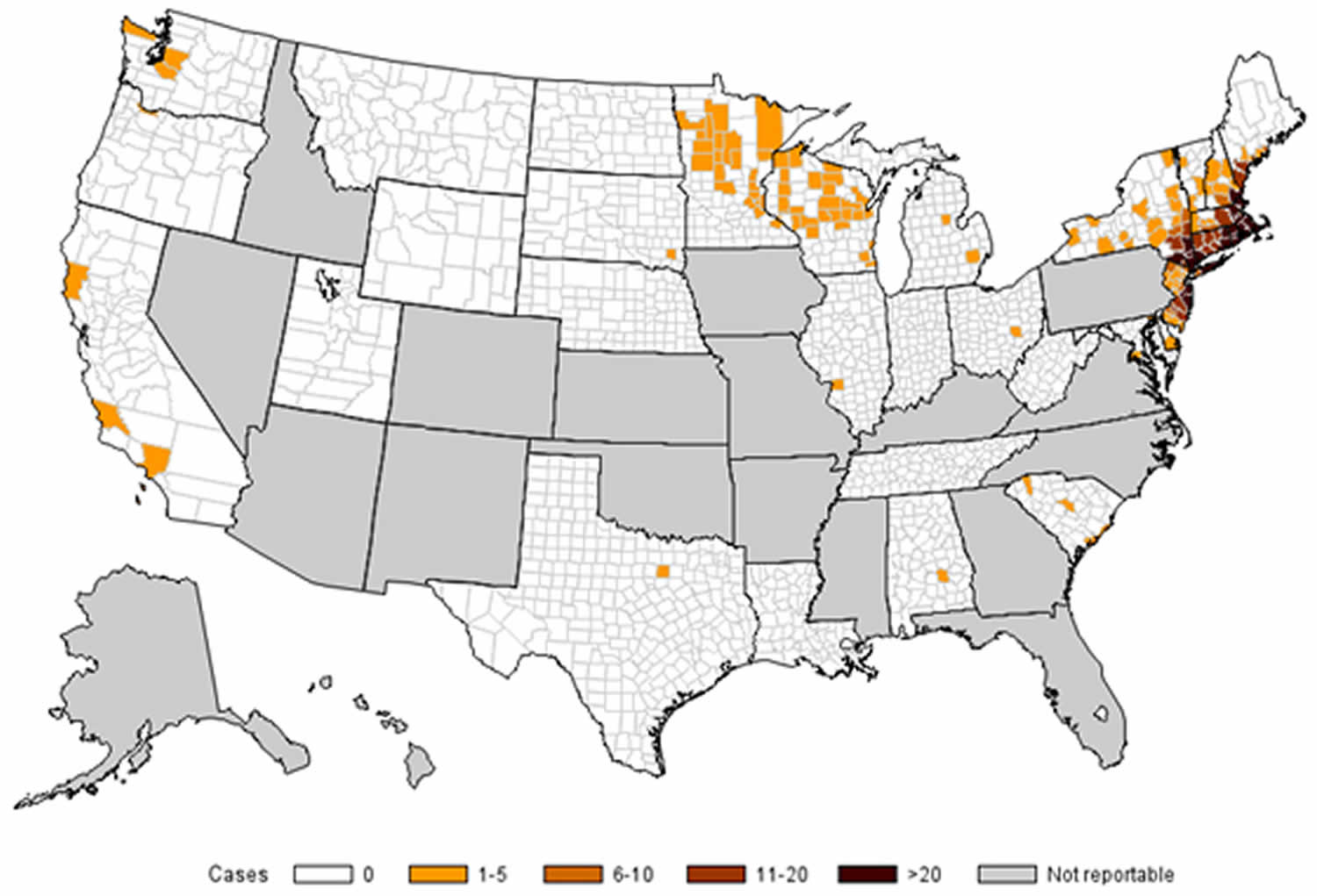
Figure 2. Babesiosis map (number of reported cases of babesiosis, by county of residence — 31 states, 2014)
Footnote:
* Number of reported cases = 1,731; county of residence was known for all but 13 (1%) of the 1,744 total case-patients.
- Cases are mapped to the patients’ county of residence, which was not necessarily where they became infected.
- Surveillance for babesiosis was conducted by 31 states in 2014.
- Babesiosis was not a reportable disease in the gray states, and health departments in those states did not notify CDC of cases.
How do people get infected with babesiosis?
The main way people become infected with Babesia is through the bite of an infected tick.
- Babesia microti is spread by Ixodes scapularis ticks, which are commonly called blacklegged ticks or deer ticks. (Although white-tailed deer are the most important food source for the adult stage of the tick, deer are not infected with B. microti.)
- The parasite typically is spread by the young nymph stage of the tick. Nymphs are mostly found during warm months (spring and summer) in areas with woods, brush, or grass.
- Infected people might not recall a tick bite because I. scapularis nymphs are very small (about the size of a poppy seed).
Other possible ways of becoming infected with Babesia include:
- Receipt of a contaminated blood transfusion (no tests have been licensed yet for donor screening); or
- Transmission from an infected mother to her baby during pregnancy or delivery.
Can babesiosis be treated?
Yes. Effective treatments are available. People who do not have symptoms or signs of babesiosis usually do not need to be treated. Health care providers may talk with Centers for Disease Control and Prevention (CDC) staff about whether an infected person should be treated, what type of therapy to use, and how long to treat. The first step is to make sure the diagnosis is correct.
Can babesiosis be prevented?
Yes. People can take steps to prevent babesiosis and other tickborne infections. The use of prevention measures is particularly important for people at increased risk for severe babesiosis (for example, people who do not have a spleen). If possible, areas infested with ticks should be avoided, especially during warm months. If such areas cannot be avoided, use protective measures during outdoor activities. See the babesiosis prevention section below for tips, such as checking yourself daily for ticks. The tiny I. scapularis ticks that spread B. microti usually must stay attached to a person for more than 36–48 hours to be able to transmit the parasite. Daily tick checks can prevent transmission of the parasite.
No vaccine is available to protect people against babesiosis.
Babesiosis life cycle
Babesiosis is caused by apicomplexan parasites of the genus Babesia. Although more than 100 Babesia species have been reported, relatively few have caused documented cases of human infection; these include (but are not limited to) Babesia microti, Babesia divergens, Babesia duncani, and a currently unnamed agent designated MO1.
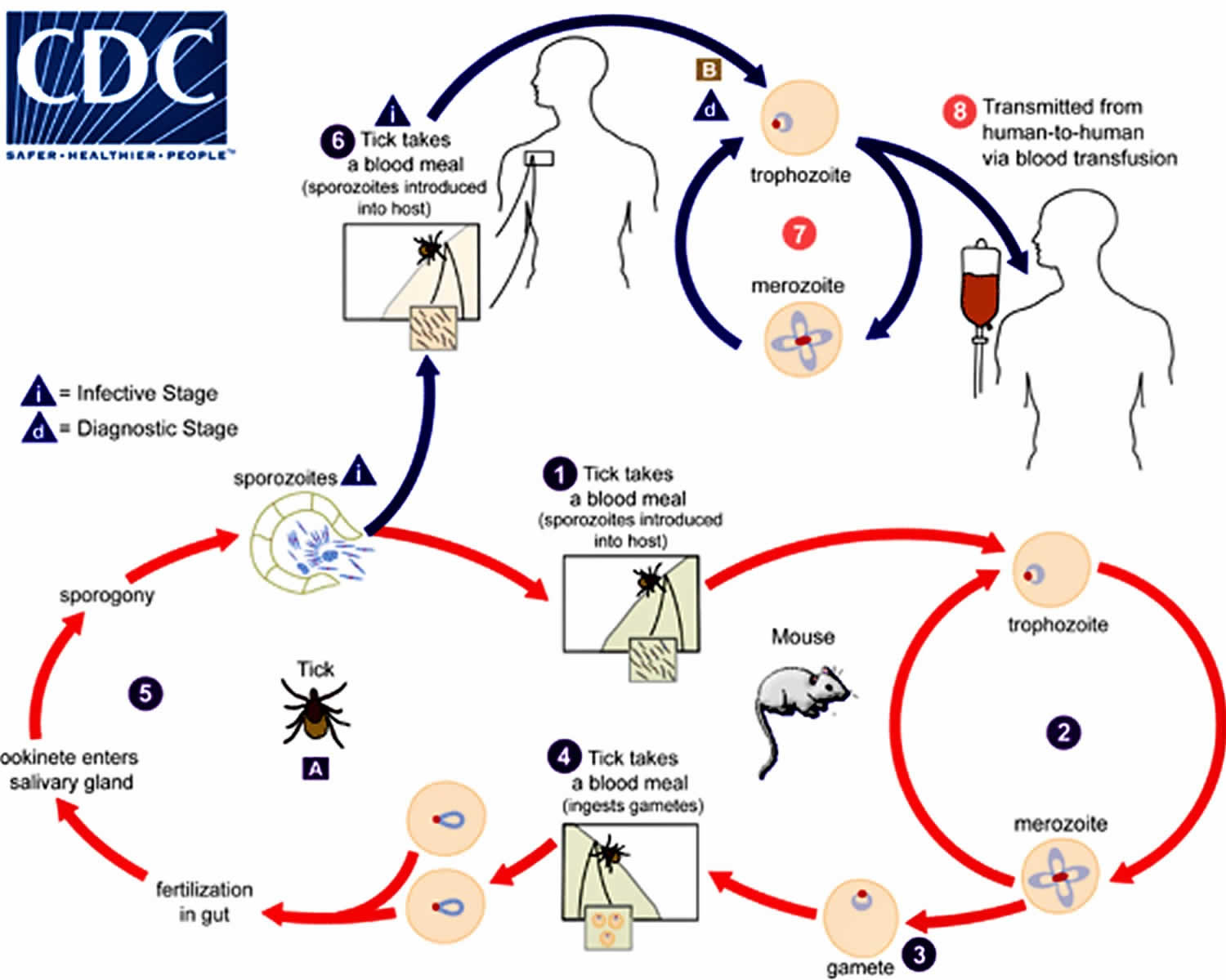
The Babesia microti life cycle involves two hosts (see Figure 3 below), which include a rodent, primarily the white-footed mouse, Peromyscus leucopus, and a tick in the genus Ixodes. During a blood meal, a Babesia-infected tick introduces sporozoites into the mouse host (#1). Sporozoites enter erythrocytes and undergo asexual reproduction (budding) (#2). In the blood, some parasites differentiate into male and female gametes, although these cannot be distinguished by light microscopy (#3). The definitive host is the tick. Once ingested by an appropriate tick (#4), gametes unite and undergo a sporogonic cycle resulting in sporozoites (#5). Transovarial transmission (also known as vertical, or hereditary, transmission) has been documented for “large” Babesia species but not for the “small” Babesia, such as Babesia microti (letter A).
Humans enter the cycle when bitten by infected ticks. During a blood meal, a Babesia-infected tick introduces sporozoites into the human host (#6). Sporozoites enter erythrocytes (letter B) and undergo asexual replication (budding) (#7). Multiplication of the blood-stage parasites is responsible for the clinical manifestations of the disease. Humans usually are dead-end hosts. However, human-to-human transmission is well recognized to occur via contaminated blood transfusions (#8).
The nymphal stage of the Ixodes is the primary vector and typically requires attachment to a host for at least 36 to 72 hours to complete a blood meal. If the tick is carrying the protozoa, the second or third day of attachment is typically when the babesia infection occurs with the transmission of sporozoite forms. These sporozoites attach and enter erythrocytes where they mature and divide via binary fission to form merozoites. These merozoites then leave the host erythrocyte, rupturing the cell, and go on to infect other erythrocytes, repeating the cycle above. The spleen is essential in the host’s ability to control this infection. Erythrocytes infected with Babesia are recognized as abnormal as they pass through the spleen and are targeted for destruction by macrophages. People with a history of splenectomy are at high risk for severe infection with high-level parasitemia. Other high-risk populations include those with HIV, older than 50 years, neonates, and immunosuppressed patients (particularly TNF inhibitors or CD20 antibody).
Figure 3. Babesiosis life cycle
Babesiosis prevention
Steps can be taken to reduce the risk for babesiosis and other tickborne infections. The use of prevention measures is especially important for people at increased risk for severe babesiosis (for example, people who do not have a spleen). Avoiding exposure to tick habitats is the best defense.
Babesia microti is spread by Ixodes scapularis ticks, which are mostly found in wooded, brushy, or grassy areas, in certain regions and seasons. No vaccine is available to protect people against babesiosis. However, people who live, work, or travel in tick-infested areas can take simple steps to help protect themselves against tick bites and tickborne infections.
During outdoor activities in tick habitats, take precautions to keep ticks off the skin.
- Walk on cleared trails and stay in the center of the trail, to minimize contact with leaf litter, brush, and overgrown grasses, where ticks are most likely to be found.
- Minimize the amount of exposed skin, by wearing socks, long pants, and a long-sleeved shirt. Tuck the pant legs into the socks, so ticks cannot crawl up the inside of the pants. Wear light-colored clothing, to make it easier to see and remove ticks before they attach to skin.
- Apply repellents to skin and clothing. Follow the instructions on the product label.
- Products that contain DEET (N,N-diethylmetatoluamide) can be directly applied to exposed skin and to clothing, to help keep ticks away (by repelling them). The product label includes details about how and where to apply the repellent, how often to reapply it, and how to use it safely on children.
- Permethrin products can be applied to clothing/boots (not to skin), actually kill ticks that come in contact with the treated clothing, and usually stay effective through several washings.
After outdoor activities, conduct daily tick checks and promptly remove any ticks that are found. Thorough, daily tick checks are very important. The I. scapularis nymphs that typically spread B. microti are so small (about the size of a poppy seed) that they are easily overlooked. But they usually must stay attached to a person for more than 36-48 hours to be able to transmit the parasite.
- Remove ticks from clothing and pets before going indoors.
- Conduct a full-body exam for ticks. Use a hand-held or full-length mirror to view all parts of the body. Be sure to check behind the knees, between the legs (groin/thighs), between the toes, under the arms (armpits), around the waist, inside the belly button, the back of the neck, behind and in the ears, as well as in and around the scalp, hairline, and hair. Remember to check children and pets, too.
Remove ticks that are attached to the skin as soon as possible, preferably by using pointed (fine-tipped) tweezers. Grab the tick’s mouth parts close to the skin, and slowly pull the tick straight out (with steady outward pressure), until the tick lets go.
How to remove a tick
If you find a tick attached to your skin, there’s no need to panic—the key is to remove the tick as soon as possible. There are several tick removal devices on the market, but a plain set of fine-tipped tweezers work very well.
To remove a tick, follow these steps:
- Using a pair of fine-tipped or pointy tweezers, grasp the tick by the head or mouth-parts right where they enter the skin. DO NOT grasp the tick by the body.
- Without jerking, pull firmly and steadily directly outward. DO NOT twist the tick out or apply petroleum jelly, a hot match, alcohol, nail polish or any other irritant to the tick in an attempt to get it to back out. Your goal is to remove the tick as quickly as possible–not waiting for it to detach. If the mouth-parts to break off and remain in the skin, remove the mouth-parts with tweezers. If you are unable to remove the mouth easily with clean tweezers, leave it alone and let the skin heal.
- Never crush a tick with your fingers. Dispose of a live tick by putting it in alcohol, placing it in a sealed bag/container, wrapping it tightly in tape, or flushing it down the toilet.
- Clean the bite wound with with rubbing alcohol or soap and water. Keep in mind that certain types of fine-pointed tweezers, especially those that are etched, or rasped, at the tips, may not be effective in removing nymphal deer ticks. Choose unrasped fine-pointed tweezers whose tips align tightly when pressed firmly together.
Then, monitor the site of the bite for the appearance of a rash beginning 3 to 30 days after the bite. At the same time, learn about the other early symptoms of Lyme disease and watch to see if they appear in about the same timeframe. If a rash or other early symptoms develop, see a physician immediately. Be sure to tell the doctor about your recent tick bite, when the bite occurred, and where you most likely acquired the tick.
Figure 4. How to remove a tick

Prevention of transfusion-associated babesiosis
Before giving blood, potential blood donors are asked if they have ever been diagnosed with babesiosis. If the answer is “yes,” they are indefinitely deferred from donating blood. If you have a patient who has (or had) laboratory evidence of Babesia infection, advise the patient to refrain indefinitely from donating blood. If the patient recently donated blood, alert the appropriate blood collection agencies and public health authorities (i.e., local or state health department). In March 2018, FDA approved the first B. microti screening tests for U.S. blood donors, who can feel fine despite being infected.
Babesiosis symptoms
Many people who are infected with Babesia microti feel fine and do not have any symptoms. The asymptomatic infection has been reported in up to 20% of adults and 50% of children 2.
The incubation period has been reported to be typically between one to six weeks.
Symptomatic illness in patients without immunodeficiencies usually consists of a febrile, flu-like illness often with a chill, sweats, malaise, fatigue, body aches, and headache. Other less common symptoms include a cough, arthralgia, sore throat, abdominal pain, nausea, loss of appetite, emotional lability, and depression.
On exam, apart from fever, patients may have hepatosplenomegaly, jaundice, retinopathy, or pharyngeal erythema. A rash is not a common symptom and may indicate co-infection with Lyme disease.
Because Babesia parasites infect and destroy red blood cells, babesiosis can cause a special type of anemia called hemolytic anemia. This type of anemia can lead to jaundice (yellowing of the skin) and dark urine.
The severity of babesiosis is often dependent on the immunocompetence of the host. Severe disease typically occurs only in high-risk populations, particularly in those with a history of asplenia. These patients may have multi-organ dysfunction, including respiratory distress, congestive heart failure, renal failure, splenic rupture, disseminated intravascular coagulation (DIC), hepatitis, or coma.
Babesiosis can be a severe, life-threatening disease, particularly in people who:
- Do not have a spleen;
- Have a weak immune system for other reasons (such as cancer, lymphoma, or AIDS);
- Have other serious health conditions (such as liver or kidney disease); or
- Are elderly.
Complications of babesiosis can include:
- A low and unstable blood pressure;
- Severe hemolytic anemia (hemolysis);
- A very low platelet count (thrombocytopenia);
- Disseminated intravascular coagulation (also known as “DIC” or consumptive coagulopathy), which can lead to blood clots and bleeding;
- Malfunction of vital organs (such as the kidneys, lungs, and liver); or
- Death.
Babesiosis diagnosis
In symptomatic people, babesiosis usually is diagnosed by examining blood specimens under a microscope and seeing Babesia parasites inside red blood cells.
To be sure the diagnosis is correct, your health care provider might have specimens of your blood tested by a specialized reference laboratory (such as at Centers for Disease Control and Prevention (CDC) or a health department).
Babesiosis blood smear
If the diagnosis of babesiosis is being considered, manual (non-automated) review of blood smears should be requested explicitly. The diagnosis of babesiosis is typically made by identifying the organism on a thin smear of peripheral blood, using Giemsa or Wright staining, and the severity of parasitemia can be assessed 2. In early infection, it is recommended that multiple thin smears be examined as parasite burden may be low initially. Ring forms are most commonly seen and can have multiple rings per cell. Tetrad formations, also known as Maltese cross, are occasionally seen.
In symptomatic patients with acute infection, Babesia parasites typically can be detected by light-microscopic examination of blood smears, although multiple smears may need to be examined. Sometimes it can be difficult to distinguish between Babesia and Plasmodium (especially P. falciparum) parasites and even between parasites and artifacts (such as stain or platelet debris). Consider having a reference laboratory confirm the diagnosis—by blood-smear examination and, if indicated, by other means, such as molecular and/or serologic methods tailored to the setting/species.
Babesiosis test
PCR testing is also available at reference laboratories and is more sensitive than peripheral smears. Serology is performed via indirect immunofluorescent antibody testing and can be useful for confirming the diagnosis. A single positive serology cannot distinguish between acute and previous infection, but a four-fold rise in acute and convalescent titers confirms a recent infection. Lab abnormalities that may be seen in babesiosis include anemia, elevated LDH, thrombocytopenia, transaminitis, proteinuria and elevated BUN and creatinine 5.
Babesiosis treatment
Most asymptomatic persons do not require treatment. Health care providers may consult CDC staff about whether to treat someone who has babesiosis, what type(s) of therapy to use, how to monitor the status of the infection, and how long to treat. Treatment decisions should be individualized, especially for patients who have (or are at risk for) severe or relapsing infection.
For ill patients, babesiosis usually is treated for at least 7-10 days with a combination of two prescription medications — typically either:
- Atovaquone PLUS azithromycin; OR
- Clindamycin PLUS quinine (this combination is the standard of care for severely ill patients).
The typical daily doses for adults are provided in the table below.
| Drug | Adult dosage (usually treat for at least 7–10 days) |
|---|---|
| Atovaquone | 750 mg orally twice a day |
| along with | |
| Azithromycin | On the first day, give a total dose in the range of 500–1000 mg orally; on subsequent days, give a total daily dose in the range of 250–1000 mg |
| or | |
| Clindamycin | 600 mg orally 3 times a day or 300–600 mg intravenously 4 times a day |
| along with | |
| Quinine | 650 mg orally 3 times a day |
Some patients—including those with severe illness—might require or benefit from supportive care, such as:
- Antipyretics;
- Vasopressors (if the blood pressure is low and unstable);
- Blood transfusions;
- Exchange transfusions (in which portions of a patient’s blood or blood cells are replaced with transfused blood components);
- Mechanical ventilation; or
- Dialysis
Partial or complete red blood cell (RBC) exchange transfusion is indicated in patients presenting with parasitemia of at least 10% and anemia with hemoglobin of <10 g/dL. Consideration for exchange transfusion should be strongly given in those with infection due to B. divergens pulmonary, renal or hepatic dysfunction, regardless of parasitemia level 6.
Atovaquone
Atovaquone in pregnancy
- Atovaquone is in pregnancy category C. Data on the use of atovaquone in pregnant women are limited, and the risk to the embryo-fetus is unknown. Because data are available about safe administration of quinine plus clindamycin during pregnancy, this drug combination, rather than atovaquone (plus azithromycin), generally is recommended for treatment of symptomatic babesiosis during pregnancy, unless the preferred medications are not available or tolerated.
- Pregnancy category C: Animal reproduction studies have shown an adverse effect on the fetus and there are no adequate and well-controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks.
Atovaquone in breastfeeding
- It is not known whether atovaquone is excreted in breast milk. Atovaquone should be used with caution in women breastfeeding infants who weigh <5 kg.
Atovaquone for children
- Atovaquone has been used safely in children who weigh > 5 kg.
Azithromycin
Azithromycin in pregnancy
- Azithromycin is in pregnancy category B. Data on the use of azithromycin in pregnant women are limited. Azithromycin may be used during pregnancy in those patients who will clearly benefit from the drug.
- Pregnancy Category B: Animal reproduction studies have failed to demonstrate a risk to the fetus and there are no adequate and well-controlled studies in pregnant women.
Azithromycin in breastfeeding
- According to a case report, azithromycin was excreted in breast milk and the nursing infant did not have adverse effects. Azithromycin should be used with caution in breastfeeding women, although the risk to the exposed infant probably is low.
Azithromycin for children
- In controlled clinical trials for various bacterial infections, oral azithromycin has been safely administered to pediatric patients aged 6 months to 16 years. Anecdotal cases of babesiosis in children, including infants, have been safely treated with azithromycin plus atovaquone.
Clindamycin
Clindamycin in pregnancy
- Clindamycin is in pregnancy category B. Data on the use of clindamycin in pregnant women are limited, although no congenital anomalies have been reported. Clindamycin may be used during pregnancy in those patients who will clearly benefit from the drug.
- Pregnancy Category B: Either animal-reproduction studies have not demonstrated a fetal risk but there are no controlled studies in pregnant women or animal-reproduction studies have shown an adverse effect (other than a decrease in fertility) that was not confirmed in controlled studies in women in the first trimester (and there is no evidence of a risk in later trimesters).
Clindamycin in breastfeeding
- Clindamycin is excreted in breast milk. The American Academy of Pediatrics classifies clindamycin as usually compatible with breastfeeding.
Clindamycin in children
- The parenteral form of clindamycin contains benzyl alcohol, which has been associated with a fatal “gasping syndrome” in premature infants.
- Patel KM, Johnson JE, Reece R, Mermel LA. Babesiosis-Associated Splenic Rupture: Case Series from a Hyperendemic Region. Clin. Infect. Dis. 2018 Dec 12[↩]
- Zimmer AJ, Simonsen KA. Babesiosis. [Updated 2018 Dec 16]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430715[↩][↩][↩]
- Abraham A, Brasov I, Thekkiniath J, Kilian N, Lawres L, Gao R, DeBus K, He L, Yu X, Zhu G, Graham MM, Liu X, Molestina R, Ben Mamoun C. Establishment of a continuous in vitro culture of Babesia duncani in human erythrocytes reveals unusually high tolerance to recommended therapies. J. Biol. Chem. 2018 Dec 28;293(52):19974-19981[↩]
- Babesiosis Maps. https://www.cdc.gov/parasites/babesiosis/data-statistics/maps/maps.html[↩]
- Hoversten K, Bartlett MA. Diagnosis of a tick-borne coinfection in a patient with persistent symptoms following treatment for Lyme disease. BMJ Case Rep. 2018 Sep 27;2018[↩]
- Lempereur L, Beck R, Fonseca I, Marques C, Duarte A, Santos M, Zúquete S, Gomes J, Walder G, Domingos A, Antunes S, Baneth G, Silaghi C, Holman P, Zintl A. Guidelines for the Detection of Babesia and Theileria Parasites. Vector Borne Zoonotic Dis. 2017 Jan;17(1):51-65[↩]