What is the cecum
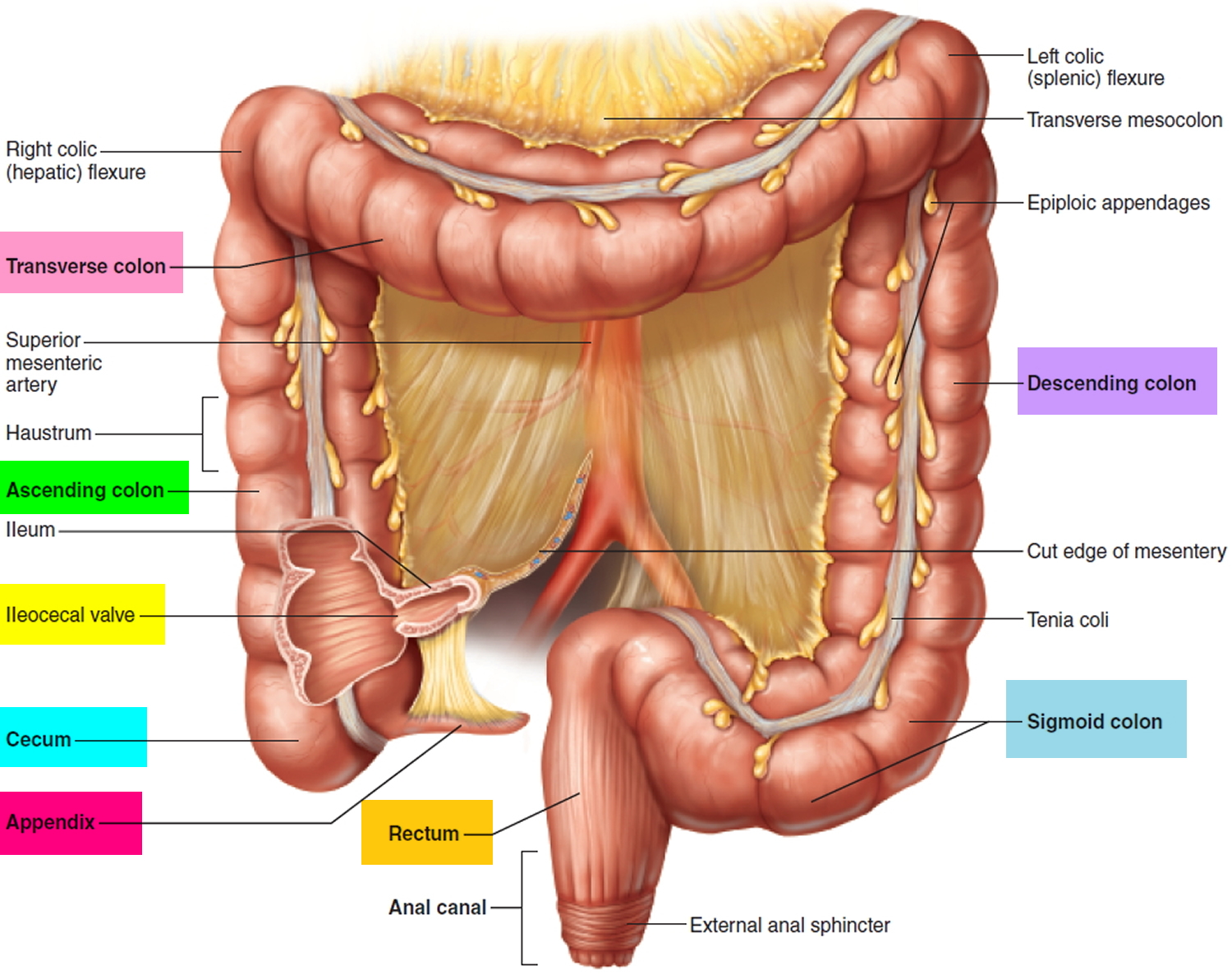
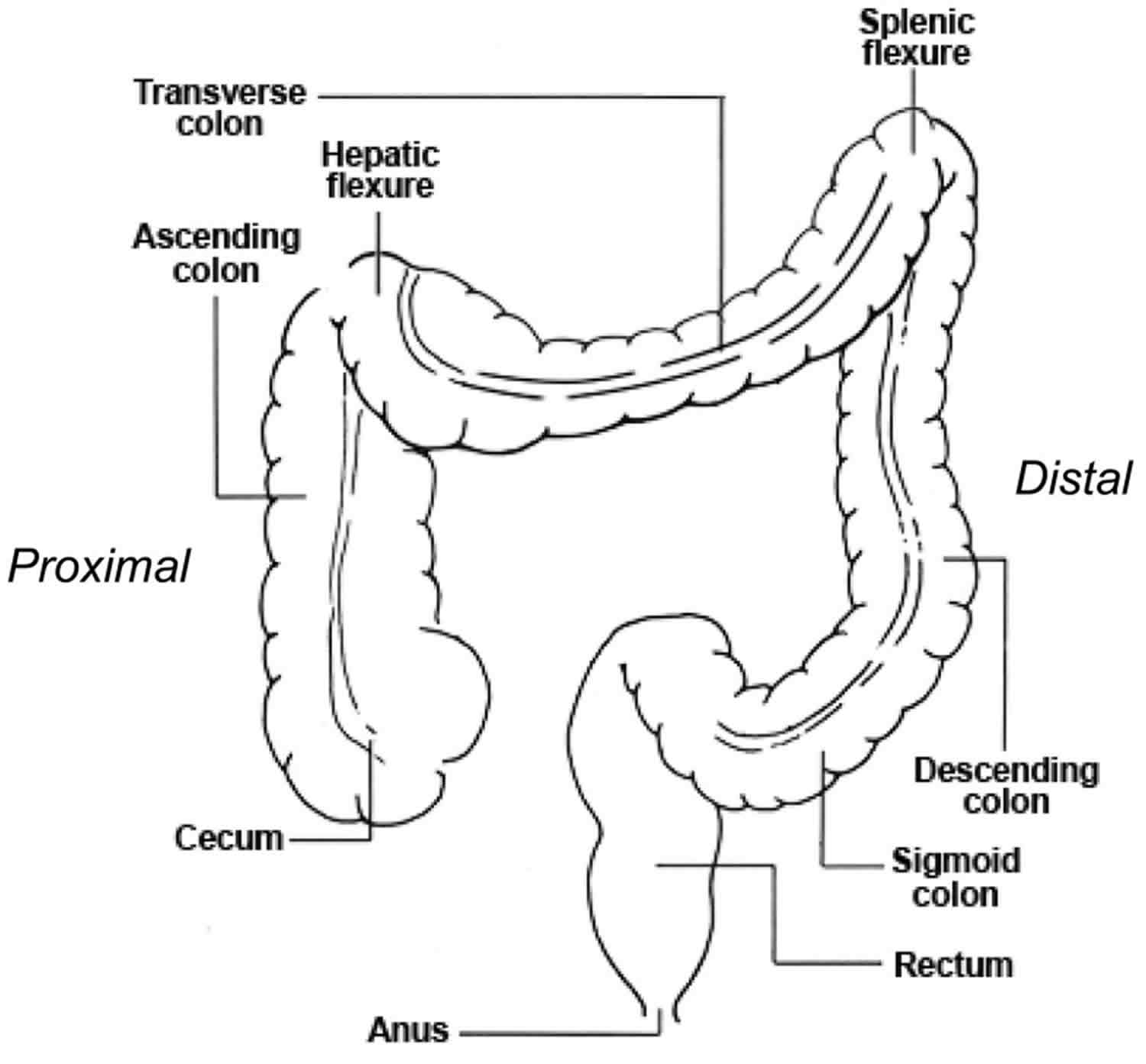
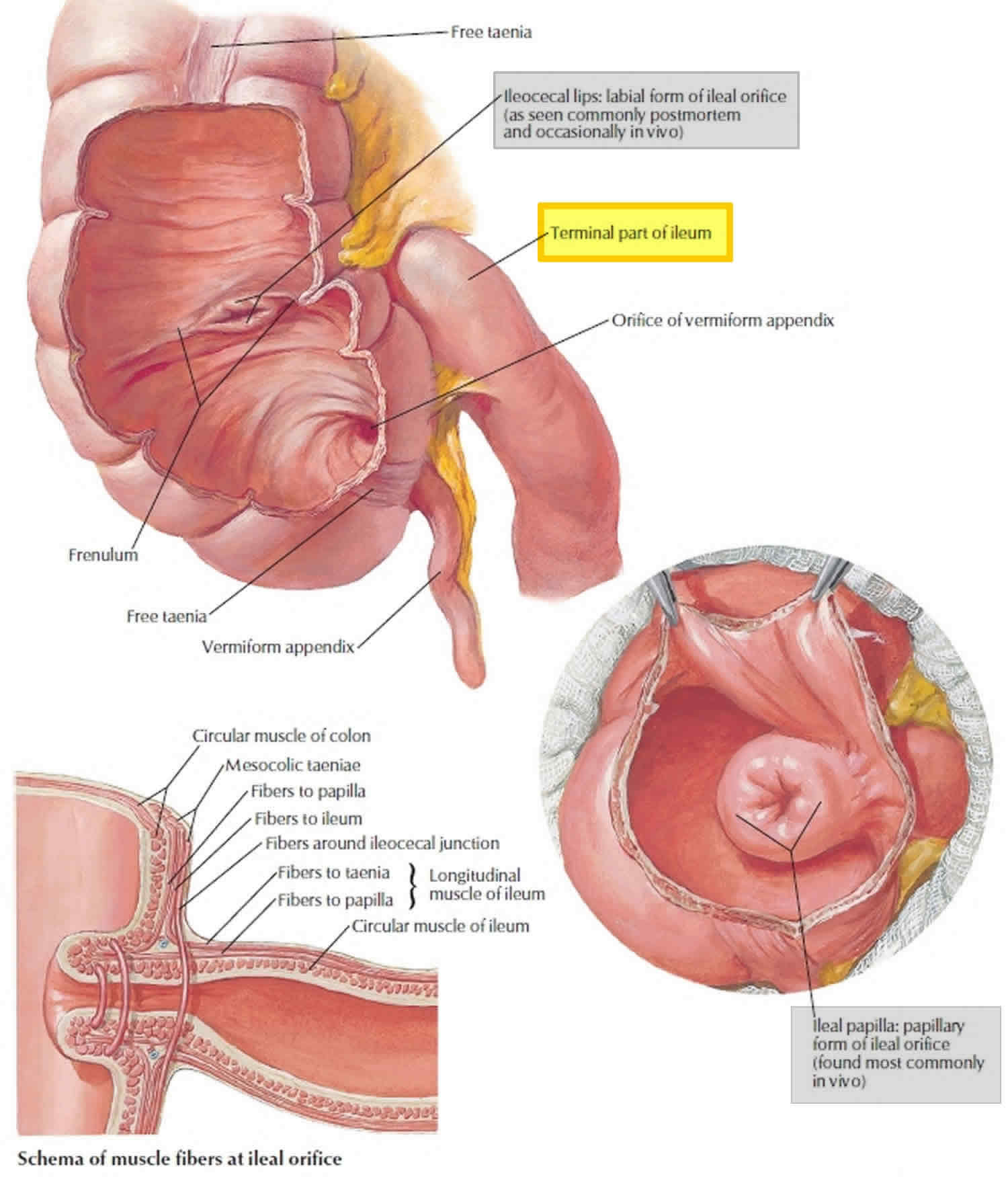
The cecum is a small blind pouch about 6 cm (2.4 inch) long at the beginning of the large intestine (the ascending colon). Attached to the cecum is a twisted, coiled tube called the appendix or vermiform appendix, measuring about 8 cm (3 in.) in length. The base of the appendix lies on the posteromedial wall of the cecum about 1 to 2 centimeters below the ileocecal junction. The tip of the appendix frequently floats in the peritoneal cavity and can be located in a retrocecal position. It has a short triangular mesentery called the mesoappendix. The end of the small intestine is the ileocecal junction, where the terminal ileum joins the cecum of the large intestine. The muscularis of the ileum is thickened at this point to form a sphincter, the ileocecal valve, which protrudes into the cecum. The ileocecal valve regulates the passage of food residue into the large intestine and prevents feces from backing up into the ileum. As the cecum fills with residue, the pressure pinches the ileocecal valve shut and prevents the reflux of cecal contents into the ileum. Chemical digestion and nutrient absorption are essentially finished by the time food residue leaves the small intestine and enters the cecum.
The cecum also is located intraperitoneally, but it lacks a mesentery. The cecum is considered to be an intraperitoneal organ, as it is covered on all sides by peritoneum.
The cecum is supplied by the ileocolic artery, which is a terminal branch of the superior mesenteric artery. The ileocolic artery gives rise to the appendicular artery to supply the appendix.
Lymph from both the appendix and cecum drain into the ileocolic lymph nodes. However, while drainage from the cecum is via several intermediate mesenteric lymph nodes, the appendix drains through a single intermediate node. From the ileocolic lymph nodes, drainage proceeds to the superior mesenteric nodes.
Figure 1. Large intestine
Figure 2. Cecum
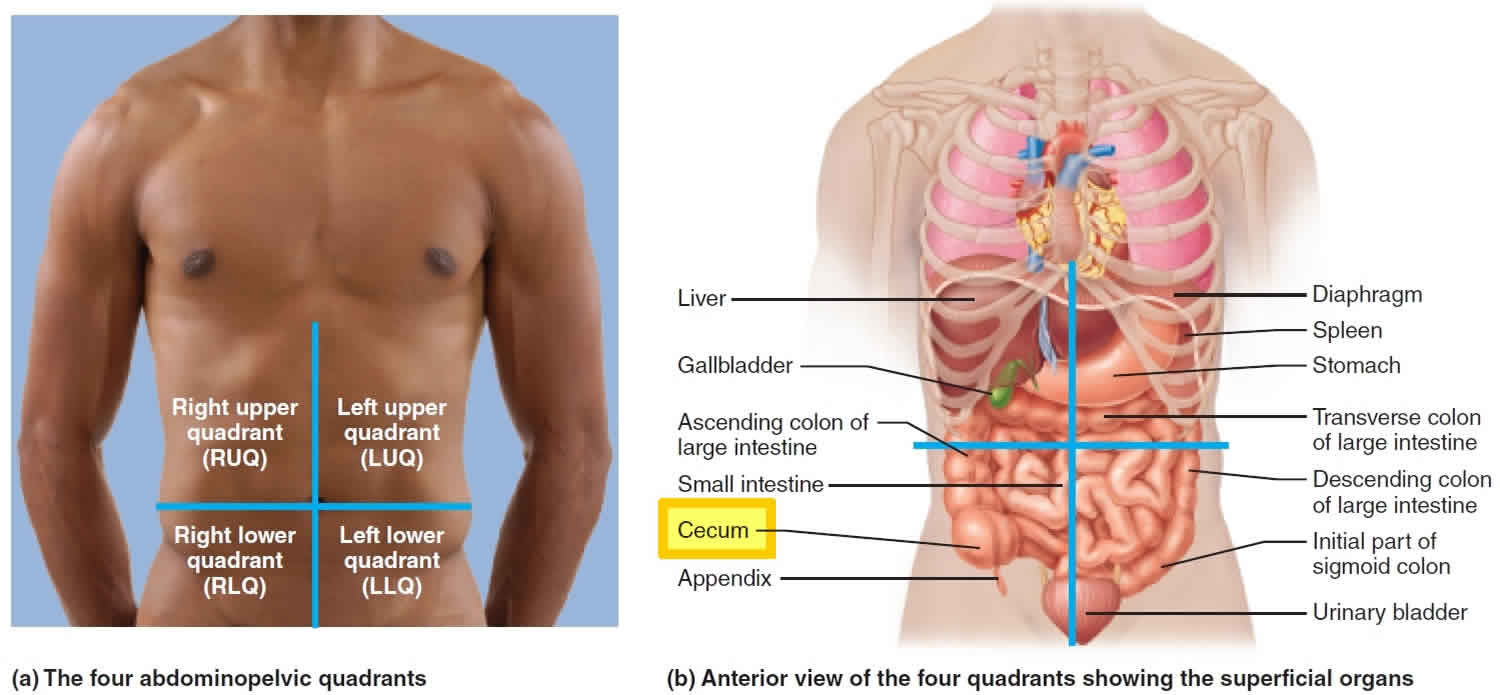
Where is the cecum located
The cecum is a blind pouch in the lower right abdominal quadrant inferior to the ileocecal valve.
Figure 4. Cecum location
Cecum function
Anatomically connecting the ileum and ascending colon via the ileocecal valve, the cecum receives digested chyme with intestinal fluid and begins to absorb water and other nutrition physiologically 1. Therefore, it is anatomically and physiologically a transition site for digestive function. Additionally, located at the initial part of cecum, the appendix is a blind tube of the cecum and is considered part of the immune system 2.
The passage of chyme (partially digested food) from the ileum into the cecum is regulated by the action of the ileocecal sphincter. Normally, the valve remains partially closed so that the passage of chyme into the cecum usually occurs slowly. Immediately after a meal, a gastroileal reflex intensifies peristalsis in the ileum and forces any chyme into the cecum. The hormone gastrin also relaxes the sphincter. Whenever the cecum is distended, the degree of contraction of the ileocecal sphincter intensifies. Movements of the colon begin when substances pass the ileocecal sphincter. Because chyme moves through the small intestine at a fairly constant rate, the time required for a meal to pass into the colon is determined by gastric emptying time. As food passes through the ileocecal sphincter, it fills the cecum and accumulates in the ascending colon.
Cecum cancer
Colorectal cancer (colon cancer) is one of the most commonly diagnosed malignant tumors in the digestive system, with an age-standardized incidence rate of 36.3 per 100,000 individuals in more developed countries and 13.7 per 100,000 individuals in less developed countries 3. According to the estimated data from the National Cancer Institute, colorectal cancer accounts for approximately 8% of all patients suffering from any type of tumor in 2017, regardless of sex 4. The proximal segments of the colon, cecum and ascending colon are considered similar to the right-sided colon, and the morbidity and mortality of carcinomas of these segments are included within right-sided colon carcinomas 5.
Adenocarcinomas of the cecum and ascending colon are both considered right-sided colon carcinomas, which exhibit exophytic pathological behavior and a poorer overall survival than left-sided colon carcinomas 6. Consistent with the prognostic differences between RCCs and LCCs, it has been found that tumor location is a significant prognostic factor in patients with adenocarcinomas of the cecum or ascending colon 7. Developing rapidly from the cecal diverticulum in the 5-week-old embryo, the cecum differs from the ascending colon, which is instead developed from the caudal limb of the midgut loop. Although the cecum and ascending colon are both considered to originate from the midgut, there may be differences between adenocarcinomas of the cecum and ascending colon due to their different locations of origin and developmental processes. The cecum, a junctional structure separated from the ileum by the ileocecal valve, receives chyme from the ileum and connects to the ascending colon.
In this study 5, compared to adenocarcinomas of the ascending colon, patients with adenocarcinomas of the cecum had a worse cancer-specific survival, indicating the heterogeneity within different subtypes of right-sided colon carcinoma. This variability may be attributed to the different tumor biological behavior or potential gene expressive differences between these two subtypes. Notably, in this study 5, significant differences were observed in the depth of tumor invasion and regional lymph node metastasis between adenocarcinomas of the cecum and ascending colon, which indicated that intraoperative local lymph node examination and regional lymphadenectomy are essential in surgical therapy for adenocarcinomas of the cecum and ascending colon. The results of the same study 5 also revealed the potential prognostic differences between adenocarcinomas of the cecum and ascending colon, which may be caused by the differential expression of the SLCO1B3 gene. Including the expression level of SLCO1B3 in intraoperatively examined lymph nodes, eight factors could predict the prognosis for patients with cancers of the cecum and ascending colon. Regarding surgical therapeutic strategies, resection of more than 15 local lymph nodes is appropriate for improving prognosis 5.
Cecum inflammation
Inflammatory bowel disease (IBD) encompasses two types of idiopathic intestinal disease that are differentiated by their location and depth of involvement in the bowel wall 8. Ulcerative colitis involves diffuse inflammation of the colonic mucosa. Most often ulcerative colitis affects the rectum (proctitis), but it may extend into the sigmoid (proctosigmoiditis), beyond the sigmoid (distal ulcerative colitis), or include the entire colon into the cecum (pancolitis). Crohn disease (Crohn’s disease) results in transmural ulceration of any portion of the gastrointestinal tract (GI) most often affecting the terminal ileum and colon 9. Both diseases are classified by extent (mild, moderate, or severe) and location. Crohn disease also is classified by phenotype- inflammatory, structuring, or penetrating 10.
Although the exact etiology of inflammatory bowel disease (IBD) is not known, there is substantial evidence to suggest that the disease is resulting from an inappropriate immune response in the bowel to situations from environmental factors such as drugs, toxins, infections or intestinal microbes in a genetically susceptible host. More than a hundred genes associated with inflammatory bowel disease have been identified. In Crohn’s disease particularly, there appears to be a genetic association with phenotypes. Specifically, NOD2/CARD15 mutations were found to be associated with a phenotype of Crohn’s disease which was associated in those diagnosed at a younger age, with ileal involvement, increased severity of ileal disease requiring surgical intervention/reoperation 11. In the future, this genotyping could potentially provide prognostic information on the severity of the disease. Furthermore, it could predict which patient’s should be considered for surgical management vs. medical management based on a more detailed understanding of genetic analysis.
The North American incidence of inflammatory bowel disease ranges from 2.2 to 19.2 cases per 100,000 person-years for ulcerative colitis and 3.1 to 20.2 cases per 200,000 person-years for Crohn’s disease. Prevalence in the United States of adult ulcerative colitis was 238 per 100,000 population and 201 per 100,000 population from data in a large study based on insurance claims. inflammatory bowel disease is much more prevalent in North America and Europe than Asia or Africa. Although most inflammatory bowel disease occurs in people age 15 to 30 years, up to 25% of patients will develop inflammatory bowel disease by adolescence. There appears to be a bimodal distribution with a second peak of 10% to 15% developing inflammatory bowel disease after age 60 12.
Prognosis for both ulcerative colitis and Crohn’s disease depends on the extent of disease and treatment response. The stool markers lactoferrin and calprotectin are useful in determining postoperative recurrence of Crohn’s disease. Some evidence exists that these can also be used to predict future flares.
Continued surveillance for dysplasia is critical for long-standing ulcerative colitis patients. Cumulative risk of colorectal cancer is estimated to be as high as 30% for those with the disease of 30 years or more. The extraintestinal manifestation of primary sclerosing cholangitis leads to liver failure 13.
The mortality of patients with inflammatory bowel disease is about 1.5 -5 times higher compared to the general population. Patients with Crohn disease suffer the highest morbidity and mortality. The major cause of death include infections, the progression of disease, surgical complications, and multiorgan involvement. More important, patients with inflammatory bowel disease also have a high rate of colorectal cancer. Patients who develop pancolitis have the highest risk of colon cancer within two decades. Hence screening colonoscopy is recommended at 1-2 year intervals 14.
In addition to the disease, these patients are also managed with potent medications like steroids and biological agents, which have a host of adverse effects. Thus, the importance of the pharmacist who should be alert for any adverse reaction. Patients with inflammatory bowel disease are also at risk for asthma or chronic obstructive pulmonary disease (COPD).
Finally, inflammatory bowel disease has enormous mental morbidity. Many patients develop depression, suicidal tendencies, and anxiety. thus, at every visit, the nurse should monitor the patient’s mental status and make appropriate referrals 15. The quality of life for most patients is poor to okay.
- Guarner F, Malagelada JR. Gut flora in health and disease. Lancet. 2003;361:512–519. doi: 10.1016/S0140-6736(03)12489-0[↩]
- Ruoff C, Hanna L, Zhi W, Shahzad G, Gotlieb V, Saif MW. Cancers of the appendix: Review of the literatures. ISRN Oncol. 2011;2011:728579[↩]
- Global cancer statistics, 2012. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. CA Cancer J Clin. 2015 Mar; 65(2):87-108.[↩]
- Cancer Statistics, 2017. Siegel RL, Miller KD, Jemal A. CA Cancer J Clin. 2017 Jan; 67(1):7-30.[↩]
- Xie X, Zhou Z, Song Y, Wang W, Dang C, Zhang H. Differences between carcinoma of the cecum and ascending colon: Evidence based on clinical and embryological data. Int J Oncol. 2018;53(1):87-98. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5958713/[↩][↩][↩][↩][↩]
- The Worse Prognosis of Right-Sided Compared with Left-Sided Colon Cancers: a Systematic Review and Meta-analysis. Yahagi M, Okabayashi K, Hasegawa H, Tsuruta M, Kitagawa Y. J Gastrointest Surg. 2016 Mar; 20(3):648-55.[↩]
- He WZ, Liao FX, Jiang C, Kong PF, Yin CX, Yang Q, Qiu HJ, Zhang B, Xia LP. Primary tumor location as a predictive factor for first-line bevacizumab effectiveness in metastatic colorectal cancer patients. J Cancer. 2017;8:388–394. doi: 10.7150/jca.16804[↩]
- McDowell C, Bhimji SS. Bowel, Inflammatory Disease (IBD) [Updated 2018 Aug 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470312[↩]
- Ranasinghe IR, Hsu R. Crohn Disease. [Updated 2017 Jun 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK436021[↩]
- Christian Maaser, Andreas Sturm, Stephan R Vavricka, Torsten Kucharzik, Gionata Fiorino, Vito Annese, Emma Calabrese, Daniel C Baumgart, Dominik Bettenworth, Paula Borralho Nunes, Johan Burisch, Fabiana Castiglione, Rami Eliakim, Pierre Ellul, Yago González-Lama, Hannah Gordon, Steve Halligan, Konstantinos Katsanos, Uri Kopylov, Paulo G Kotze, Eduards Krustiņš, Andrea Laghi, Jimmy K Limdi, Florian Rieder, Jordi Rimola, Stuart A Taylor, Damian Tolan, Patrick van Rheenen, Bram Verstockt, Jaap Stoker, European Crohn’s and Colitis Organisation [ECCO] and the European Society of Gastrointestinal and Abdominal Radiology [ESGAR]; ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications, Journal of Crohn’s and Colitis, , jjy113, https://doi.org/10.1093/ecco-jcc/jjy113[↩]
- Ranasinghe IR, Hsu R. Crohn Disease. [Updated 2017 Jun 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK436021/[↩]
- Su HJ, Chiu YT, Chiu CT, Lin YC, Wang CY, Hsieh JY, Wei SC. Inflammatory bowel disease and its treatment in 2018: Global and Taiwanese status updates. J. Formos. Med. Assoc. 2018 Jul 24[↩]
- Kobayashi T, Hisamatsu T, Suzuki Y, Ogata H, Andoh A, Araki T, Hokari R, Iijima H, Ikeuchi H, Ishiguro Y, Kato S, Kunisaki R, Matsumoto T, Motoya S, Nagahori M, Nakamura S, Nakase H, Tsujikawa T, Sasaki M, Yokoyama K, Yoshimura N, Watanabe K, Katafuchi M, Watanabe M, Hibi T. Predicting outcomes to optimize disease management in inflammatory bowel disease in Japan: their differences and similarities to Western countries. Intest Res. 2018 Apr;16(2):168-177[↩]
- Chung AE, Vu MB, Myers K, Burris J, Kappelman MD. Crohn’s and Colitis Foundation of America Partners Patient-Powered Research Network: Patient Perspectives on Facilitators and Barriers to Building an Impactful Patient-Powered Research Network. Med Care. 2018 Oct;56 Suppl 10 Suppl 1:S33-S40[↩]
- Viganò CA, Beltrami MM, Bosi MF, Zanello R, Valtorta M, Maconi G. Alexithymia and Psychopathology in Patients Suffering From Inflammatory Bowel Disease: Arising Differences and Correlations to Tailoring Therapeutic Strategies. Front Psychiatry. 2018;9:324[↩]