Contents
What does d aspartic acid do
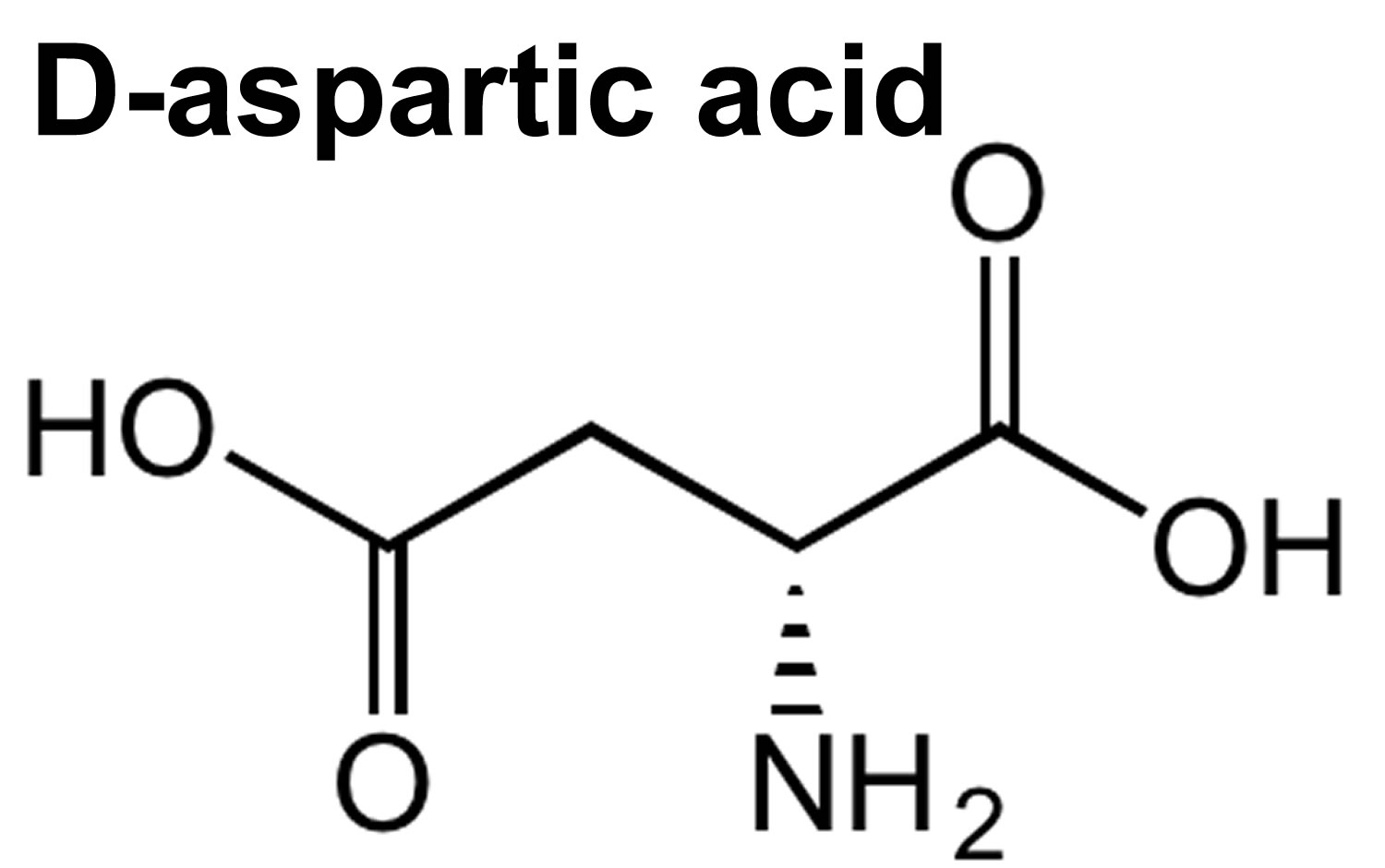
D-Aspartic acid is an non-essential amino acid that plays an important role in tuning testosterone production in the gonads of male vertebrates 1. D-Aspartic acid promotes either the synthesis or the release of testosterone, whereas nitric oxide inhibits it 1. In the adult rat testis D-Aspartic acid operates on Leydig cell steroidogenic pathway by favoring testosterone synthesis 2, whereas nitric oxide (NO) appears to inhibit it 3. These findings agree with a recent study showing that in boar testis testosterone production in the laboratory test tube study is stimulated by D-Aspartic acid and inhibited by an nitric oxide (NO) generating system 4.
D-aspartic acid is an amino acid that exists in central nervous and reproductive tissues 5. Animal and human research suggest that D-aspartic acid functions in the development of the nervous system as well as hormonal regulation 6. Data in mammalian studies indicate that D-aspartic acid supplementation can influence the hypothalamic-pituitary-gonadal axis at the level of the hypothalamus 7, anterior pituitary 8 and the testes 9. Accumulation of D-aspartic acid at these sites is associated with an upregulation of testosterone production in these animals, as well as upstream effectors of the hypothalamic-pituitary-gonadal axis (i.e. luteinizing hormone).
Early human research demonstrated that three grams per day of D-aspartic acid supplementation increased total testosterone in untrained men by ~42% 10. Subsequently the popularity of D-aspartic acid supplementation in recreational resistance training has increased, owing to the positive relationship between testosterone levels and mechanisms of muscular hypertrophy, such as increased protein synthesis 11 and satellite cell proliferation 12. However in resistance-trained men, three grams of D-aspartic acid per day resulted in no meaningful change in testosterone levels, or training outcomes after 14 13 or 30 days of supplementation 14. Indeed, Melville and colleagues 13 reported that a larger daily dose of D-aspartic acid (6 g.d-1) actually decreased basal testosterone by ~12.5% after 14 days of supplementation, suggesting a deleterious effect on negative feedback mechanisms of the hypothalamic-pituitary-gonadal axis. This reduction is a particularly concerning finding considering the supposed importance of basal testosterone, with respect to hypertrophic training outcomes. It is unclear if the observed decline in basal testosterone after 14 days will continue if supplementation is continued for a longer timeframe (i.e. three months). Moreover, the relationship to training outcomes if testosterone continues to decline, or otherwise is maintained at a new basal level, is unknown.
The potential adverse effects of D-aspartic acid on testosterone, in addition to affecting hypertrophic outcomes, may also have an effect on the neural mechanisms associated with increased muscular strength. Strength appears to be a function of both the size 15 and composition of muscle 16, in addition to the rate and magnitude of output from the nervous system 17. Resistance training increases the voluntary activation of the nervous system 18, in addition to increasing the input-output response of cells in the spinal cord 19. There is a growing body of research that suggests D-aspartic acid fills the criteria of a neurotransmitter 20, which potentially could provide strength improvements via increased neurotransmitter availability. Neural plasticity, which is observed in untrained individuals is a key explanatory mechanism of strength development 21, and can involve cells at the spinal and supraspinal level. Measures of neural plasticity that examine spinal and supraspinal output, such as the H-reflex and V-wave respectively, have not been measured following a period of resistance exercise in trained individuals and as such, it is not clear if trained populations have the same neural adaptation response to resistance training as untrained individuals. The link between testosterone and changes in the corticospinal pathway has been explored in humans with artificially induced testosterone levels 22. These increases in testosterone were concurrent with reduced threshold of the evoked potentials from transcranial magnetic stimulation (TMS), indicating that testosterone can positively increase the output from a given input, within the corticospinal pathway 22. It is plausible that artificially changing testosterone levels, via supplementation may affect strength or power, by altering the efficiency of the corticospinal pathway. The effects of D-aspartic acid on neural plasticity has yet to be researched in humans.
D-Aspartic acid results
This comprehensive systematic review 23 showed that exogenous D-aspartic acid enhances testosterone levels in male animal studies, whereas studies in human yielded inconsistent results. The evidence for this association in man is still sparse, mostly because of the lack of good quality studies. Short-term supplementation (14 days) has been shown to reduce testosterone levels in resistance-trained men 13. The results of this randomized, double-blind, placebo controlled trial 5 in healthy resistance-trained men, aged 18–36, who had been performing regular resistance training exercise for at least 3 days per week for the previous 2 years, indicated that d-aspartic acid supplementation is ineffective at changing testosterone levels, or positively affecting training outcomes. Randomized participants were 22 men (d-aspartic acid n = 11; placebo n = 11) (age, 23.8±4.9 year, training age, 3.2±1.5 year). Reductions in estradiol and the blunting of peripheral excitability appear unrelated to improvements from resistance training.
In another randomized, double-blind interventional study by Willoughby et al, 24 twenty apparent healthy and heavy resistance-trained men were enrolled (49). The participants trained 4 times/week while orally ingesting either 3 grams of placebo or D-aspartic acid supplementat in the morning upon waking. After 28 days D-aspartic acid supplementation had no effect on muscle strength, body mass and serum hormones (total testosterone, free testosterone, LH, GnRH and estradiol). Unlike the previous study, participant demographics had been well described such as age, weight and height. All participants passed a mandatory medical screening.
Another clinical trial study, by Bloomer et al 25 investigated the effect of D-aspartic acid/sodium nitrate/ vitamin D3 supplement on blood total and free testosterone and nitrate/nitrite before and after 14 and 28 days in 10 overweight or obese men with the average age of 42 years. Although in this study basal testosterone levels of subjects were low (approximately 3 ng/mL), after 14 and 28 days of supplementation, testosterone levels were not statistically significant and that is similar to the results obtained by Topo et al 26 and Willoughby et al 24. This is contrary with the hypothesis that D-aspartic acid supplementation is effective with lower testosterone levels.
D-aspartic acid supplementation in bodybuilding
Three months of D-aspartic acid supplementation did not change basal testosterone levels in resistance-trained men 5. The lack of change observed in that study 5 suggests that the previously observed reduction in testosterone after two weeks supplementation may be transitory 13. Previous research in resistance-trained men has observed increases in d-aspartate oxidase 14, which oxidises D-aspartic acid. It is plausible that d-aspartate oxidase production is linked with total testosterone levels, and regulates abnormal levels of D-aspartic acid in the bloodstream. This could explain why increased testosterone from D-aspartic acid supplementation has been observed when levels of testosterone are low-normal 27, in comparison to the lack of change observed when testosterone levels are normal-high 14. The results of this study clearly demonstrate that D-aspartic acid is an ineffective supplement for improving basal testosterone levels in resistance-trained men.
The supplementation of D-aspartic acid caused a marked reduction in estradiol levels. Mechanistically, estradiol was potentially reduced via disruption of the testosterone-estradiol aromatase pathway 28. In the animal model, both positive 29 and negative 30 relationships have been observed between gonadal D-aspartic acid and estradiol. Additionally, in vitro research has observed both increased 31 and decreased 30 estradiol levels with D-aspartic acid supplementation. With this conflicting data, the effects of D-aspartic acid on estradiol in the animal model is unclear. In resistance-trained men estradiol remains unchanged over two 5 and four weeks 14 from a resistance training and supplement protocol. Despite an observed reduction of estradiol in the present study, the training outcomes clearly demonstrate similar improvements in hypertrophy and strength. This is further evidence that basal hormonal fluctuations within normal ranges are not key explanatory mechanisms of training outcomes, rather it is likely intrinsic mechanisms that explain training improvements and that hormones play a permissive role in training adaptions. Intrinsic mechanisms pertaining to hypertrophy include the activation of various signalling pathways (Akt/mTOR, PA/mTOR, mechanoreceptor), phosphorylation of intramuscular signalling proteins, regulation of messenger RNA (translation initiation), increases in AR content, satellite cell activity and expression of muscle-specific microRNA 32. For strength it appears that neural adaptation mechanisms such as earlier recruitment of type II motor units, as demonstrated by a reduction in recruitment threshold distributions; increased observation of ‘true’ doublet spikes; or increased maximal firing frequency of motor units during voluntary ballistic contractions shown by a reduction of inter-spike intervals; increase in efferent neural drive to the muscle: and an increase in the firing frequency of the motor units 33 are driving improvements.
Neural adaptation
This is the first study 5 to examine changes in the H-reflex pathway following a period of resistance exercise in trained men. These results demonstrate for the first time improvements in responsiveness at the level of the alpha motoneuron, similar to that which is observed in novice populations 33. Mechanistically, this could be a result of improved excitability of the alpha motoneuron, improved excitability of the Ia afferent loop, or a decrease in presynaptic inhibition at the level of the interneuron 34.
Improved excitability of the H-reflex pathway in the placebo group was blunted by D-aspartic acid supplementation. It is plausible since D-aspartic acid appears to fit the role of a neurotransmitter 35, that D-aspartic acid is inhibiting the capacity of the neurotransmitter system to adapt over time to the resistance exercise stimulus. D-aspartic acid can enter the neuron via L-glutamate transporters 36. However, the transport of D-aspartic acid could be saturating the L-glutamate transporters. This, in turn, might inhibit a more potent neurotransmitter, such as glutamate 37 and ultimately result in blunted neural adaption. As both groups equally improved plantar flexor isometric and dynamic strength (10RM values), the contribution of between-group differences observed in excitability of the spinal pathway appears negligible. The H-reflex utilises the same pathway as the myotatic reflex 38, which is important reflex in the context of power development because it activates alpha motoneuron activity in a rapidly stretched muscle 34. A potential limitation of this study is the lack of testing for dynamic power (e.g. plyometric testing), as this would help clarify the relevance of the improved H-reflex excitability from the perspective of functional power.
The present study 5 showed that V/Mmax ratio in the plantar flexors did not improve over the course of the training study, suggesting that neither D-aspartic acid supplementation nor training affected supraspinal drive to the plantar flexors in this population. Research in novice populations has demonstrated increases in V/Mmax ratio with resistance training 39, however resistance-trained individuals (e.g. weightlifters) demonstrate significantly larger V-wave responses in comparison to inexperienced trainers 40. Data on endurance training has failed to show any improvements in V-wave measures, suggesting that the improvement of neural drive is an adaptation exclusive to resistance training 21. The results of the current study 5 present novel evidence, demonstrating that resistance-trained individuals do not improve supraspinal drive to the plantar flexors with three months of resistance training. This could suggest that there is a ceiling for adaptation of supraspinal drive and that further neural improvements are exclusive to changes of excitability within the spinal cord. The data from the present study exhibited high variability between subject’s V-waves, which might suggest an issue with the sensitivity of the method that was used. Additionally, the reliance of V-waves to measure a change in supraspinal drive is a limitation of this study. The inclusion of additional measures of corticospinal function from the use of transcranial stimulation techniques may have provided clearer insight into potential supraspinal changes.
Summary
D-aspartic acid is currently purported as a testosterone boosting supplement. The results of this study 5 suggests it does not, the study also adds to the body of evidence that changes in basal hormonal levels within the normal physiological range play a minor role with respect to training outcomes. The long-term effects of D-aspartic acid did not provide any benefit in relation to strength or hypertrophy in a resistance-trained population 5. Additionally, D-aspartic acid appears to be blunting neural adaptation that was evident in the placebo group. As such, the results of this study strongly suggest that D-aspartic acid is not an ideal supplement for resistance-trained men and cannot be recommended for long-term use with resistance training.
The effects of D-aspartic acid have yet to be explored in either a female population, where mechanisms of D-aspartic acid and testosterone manipulation could differ. Additionally, an elderly population, with decreased testosterone levels, may benefit from D-aspartic acid supplementation combined with resistance training, as some research has demonstrated that D-aspartic acid can improve testosterone levels in untrained men. The mechanisms driving power adaptations in untrained individuals are well researched, however, in a resistance-trained population the data is lacking. The link between neural plasticity and changes in functional power from resistance training, from a resistance-trained context could also be explored. Additionally, future studies could further test the theory that resistance-trained individuals experience a ceiling effect with supraspinal neural adaptations, or if they have the ability to continue to improve supraspinal drive.
D-aspartic acid side effects
D-aspartic acid side effects reported were feelings of irritability, nervousness, rapid heart rate, and headache 23.
- Di Fiore MM, Lamanna C, Assisi L, Botte V. Opposing effects of D-aspartic acid and nitric oxide on tuning of testosterone production in mallard testis during the reproductive cycle. Reprod Biol Endocrinol. 2008;6:28. Published 2008 Jul 4. doi:10.1186/1477-7827-6-28 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2474629[↩][↩]
- Furuchi T, Homma H. Free D-aspartate in mammals. Biol Pharm Bull. 2005;28:1566–1570.[↩]
- Weissman BA, Niu E, Ge R, Sottas CM, Holmes M, Hutson JC, Hardy MP. Paracrine modulation of androgen synthesis in rat leydig cells by nitric oxide. J Androl. 2005;26:369–378[↩]
- Lamanna C, Assisi L, Vittoria A, Botte V, Di Fiore MM. d-Aspartic acid and nitric oxide as regulators of androgen production in boar testis. Theriogenology. 2007;67:249–254[↩]
- Melville GW, Siegler JC, Marshall PWM. The effects of d-aspartic acid supplementation in resistance-trained men over a three month training period: A randomised controlled trial. PLoS One. 2017;12(8):e0182630. Published 2017 Aug 25. doi:10.1371/journal.pone.0182630 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5571970/[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Neidle A, Dunlop DS. Developmental changes in free D-aspartic acid in the chicken embryo and in the neonatal rat. Life Sciences. 1990;46(21):1517–22[↩]
- D’Aniello A, Di Fiore MM, Fisher GH, Milone A, Seleni A, D’Aniello S, et al. Occurrence of D-aspartic acid and N-methyl-D-aspartic acid in rat neuroendocrine tissues and their role in the modulation of luteinizing hormone and growth hormone release. FASEB Journal. 2000;14(5):699–714[↩]
- D’Aniello A, Di Cosmo A, Di Cristo C, Annunziato L, Petrucelli L, Fisher G. Involvement of D-aspartic acid in the synthesis of testosterone in rat testes. Life Sciences. 1996;59(2):97–104.[↩]
- Nagata Y, Homma H, Lee JA, Imai K. D-Aspartate stimulation of testosterone synthesis in rat Leydig cells. FEBS Letters. 1999;444(2–3):160–4.[↩]
- D’Aniello G, Ronsini S, Notari T, Grieco N, Infante V, D’Angel N, et al. d-Aspartate, a key element for the improvement of sperm quality. Advances in Sexual Medicine. 2012;2(4):45–53.[↩]
- Ferrando AA, Tipton KD, Doyle D, Phillips SM, Cortiella J, Wolfe RR. Testosterone injection stimulates net protein synthesis but not tissue amino acid transport. American Journal of Physiology—Endocrinology and Metabolism. 1998;275(5 38–5):E864–E71.[↩]
- Herbst KL, Bhasin S. Testosterone action on skeletal muscle. Current Opinion in Clinical Nutrition and Metabolic Care. 2004;7(3):271–7.[↩]
- Melville GW, Siegler JC, Marshall PWM. Three and six grams supplementation of d-aspartic acid in resistance trained men. Journal of the International Society of Sports Nutrition. 2015;12(1):1–6.[↩][↩][↩][↩]
- Willoughby DS, Leutholtz B. D-Aspartic acid supplementation combined with 28 days of heavy resistance training has no effect on body composition, muscle strength, and serum hormones associated with the hypothalamo-pituitary-gonadal axis in resistance-trained men. Nutrition Research. 2013;33(10):803–10. doi: 10.1016/j.nutres.2013.07.010[↩][↩][↩][↩]
- Maughan R, Watson JS, Weir J. Strength and cross-sectional area of human skeletal muscle. The Journal of physiology. 1983;338:37[↩]
- Liu Y, Schlumberger A, Wirth K, Schmidtbleicher D, Steinacker JM. Different effects on human skeletal myosin heavy chain isoform expression: strength vs. combination training. J Appl Physiol (1985). 2003;94(6):2282–8.[↩]
- Aagaard P, Simonsen EB, Andersen JL, Magnusson P, Dyhre-Poulsen P. Neural adaptation to resistance training: Changes in evoked V-wave and H-reflex responses. Journal of Applied Physiology. 2002;92(6):2309–18. doi: 10.1152/japplphysiol.01185.2001[↩]
- del Olmo MF, Reimunde P, Viana O, Acero RM, Cudeiro J. Chronic neural adaptation induced by long-term resistance training in humans. Eur J Appl Physiol. 2006;96(6):722–8. doi: 10.1007/s00421-006-0153-5[↩]
- Carroll TJ, Barton J, Hsu M, Lee M. The effect of strength training on the force of twitches evoked by corticospinal stimulation in humans. Acta Physiologica. 2009;197(2):161–73. doi: 10.1111/j.1748-1716.2009.01992.x[↩]
- Ota N, Shi T, Sweedler JV. D-Aspartate acts as a signaling molecule in nervous and neuroendocrine systems. Amino Acids. 2012;43(5):1873–86. doi: 10.1007/s00726-012-1364-1[↩]
- Vila-Cha C, Falla D, Correia MV, Farina D. Changes in H reflex and V wave following short-term endurance and strength training. Journal of Applied Physiology. 2012;112(1):54–63. doi: 10.1152/japplphysiol.00802.2011[↩][↩]
- Bonifazi M, Ginanneschi F, Della Volpe R, Rossi A. Effects of gonadal steroids on the input-output relationship of the corticospinal pathway in humans. Brain Research. 2004;1011(2):187–94. doi: 10.1016/j.brainres.2004.03.022[↩][↩]
- Roshanzamir F, Safavi SM. The putative effects of D-Aspartic acid on blood testosterone levels: A systematic review. Int J Reprod Biomed (Yazd). 2017;15(1):1-10. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5340133[↩][↩]
- Willoughby DS, Leutholtz B. D-Aspartic acid supplementation combined with 28 days of heavy resistance training has no effect on body composition, muscle strength, and serum hormones associated with the hypothalamo-pituitary-gonadal axis in resistance-trained men. Nutr Res. 2013;33:803–810[↩][↩]
- Bloomer RJ, Gunnels TA, Moran RG, Schriefer JM. Influence of a D-aspartic Acid/Sodium Nitrate/Vitamin D3 Dietary Supplement on Physiological Parameters in Middle-aged Men: A Pilot Study. Open Nutraceut J. 2015;8:43–48[↩]
- Topo E, Soricelli A, D’Aniello A, Ronsini S, D’Aniello G. The role and molecular mechanism of D-aspartic acid in the release and synthesis of LH and testosterone in humans and rats. Reprod Biol Endocrinol. 2009;7:1482–1488[↩]
- Topo E, Soricelli A, D’Aniello A, Ronsini S, D’Aniello G. The role and molecular mechanism of D-aspartic acid in the release and synthesis of LH and testosterone in humans and rats. Reproductive Biology and Endocrinology. 2009;7(120):1482–8[↩]
- Simpson ER, Clyne C, Rubin G, Boon WC, Robertson K, Britt K, et al. Aromatase—A brief overview. Annual Review of Physiology. 2002;64(1):93–127.[↩]
- Assisi L, Botte V, D’Aniello A, Di Fiore MM. Enhancement of aromatase activity by D-aspartic acid in the ovary of the lizard Podarcis s. sicula. Reproduction. 2001;121(5):803–8.[↩]
- Raucci F, D’Aniello S, Di Fiore MM. Endocrine roles of D-aspartic acid in the testis of lizard Podarcis s. sicula. J Endocrinol. 2005;187(3):347–59. doi: 10.1677/joe.1.06115[↩][↩]
- Lamanna C, Assisi L, Botte V, Di Fiore MM. Involvement of D-Asp in P450 aromatase activity and estrogen receptors in boar testis. Amino Acids. 2007;32(1):45–51[↩]
- Gonzalez AM, Hoffman JR, Stout JR, Fukuda DH, Willoughby DS. Intramuscular Anabolic Signaling and Endocrine Response Following Resistance Exercise: Implications for Muscle Hypertrophy. Sports Medicine. 2016;46(5):671–85. doi: 10.1007/s40279-015-0450-4[↩]
- Dragert K, Zehr EP. Bilateral neuromuscular plasticity from unilateral training of the ankle dorsiflexors. Experimental Brain Research. 2011;208(2):217–27. doi: 10.1007/s00221-010-2472-3[↩][↩]
- Zehr EP. Considerations for use of the Hoffmann reflex in exercise studies. European Journal of Applied Physiology. 2002;86(6):455–68. doi: 10.1007/s00421-002-0577-5[↩][↩]
- D’Aniello A. d-Aspartic acid: An endogenous amino acid with an important neuroendocrine role. Brain Research Reviews. 2007;53(2):215–34. doi: 10.1016/j.brainresrev.2006.08.005[↩]
- Koyama H, Sekine M, Furuchi T, Katane M, Nimura N, Shimamoto K, et al. A novel L-glutamate transporter inhibitor reveals endogenous D-aspartate homeostasis in rat pheochromocytoma MPT1 cells. Life Sciences. 2005;76(25):2933–44. doi: 10.1016/j.lfs.2004.10.057[↩]
- Engelsen B. Neurotransmitter glutamate: Its clinical importance. Acta Neurologica Scandinavica. 1986;74(5):337–55.[↩]
- Schieppati M. The Hoffmann reflex: A means of assessing spinal reflex excitability and its descending control in man. Progress in Neurobiology. 1987;28(4):345–76.[↩]
- Nordlund MM. Improvements in dynamic plantar flexor strength after resistance training are associated with increased voluntary activation and V-to-M ratio. Journal of Applied Physiology. 2010;109(1):19–26. doi: 10.1152/japplphysiol.01307.2009[↩]
- Sale DG, Upton ARM, McComas AJ, MacDougall JD. Neuromuscular function in weight-trainers. Experimental Neurology. 1983;82(3):521–31[↩]