Contents
What is dopamine
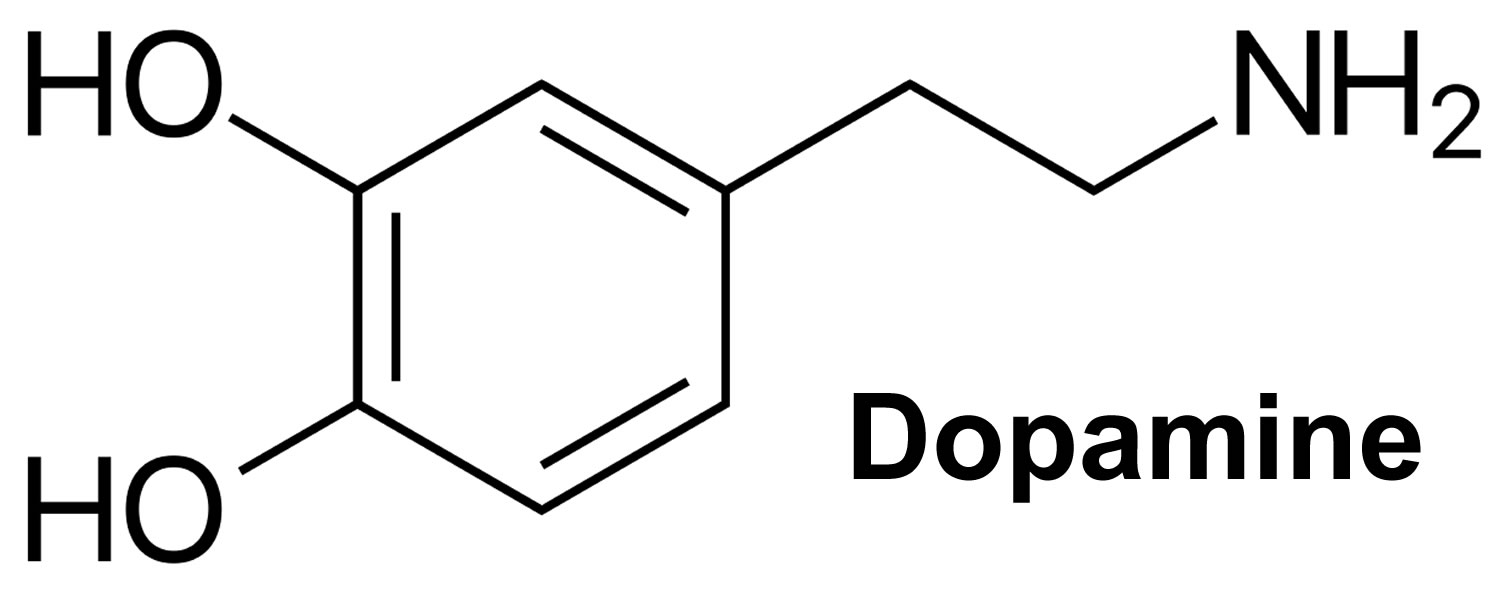
Dopamine (3,4-dihydroxyphenethylamine) is a naturally occurring catecholamine synthesized from the amino acid tyrosine and formed by decarboxylation of L‐dihydroxyphenylanaline (L‐dopa or levodopa) (see Figure 1 below). Catecholamines are a group of similar hormones released into the bloodstream in response to physical or emotional stress. The primary catecholamines are dopamine, epinephrine (adrenaline), and norepinephrine. Dopamine is the precursor to norepinephrine (noradrenaline) and epinephrine (adrenaline). Dopamine is a monoamine compound with positive inotropic activity. Dopamine binds to alpha-1 and beta-1 adrenergic receptors. Mediated through myocardial beta-1 adrenergic receptors, dopamine increase heart rate and force, thereby increasing cardiac output. Alpha-1 adrenergic receptor stimulation on vascular smooth muscle, leads to vasoconstriction and results in an increase in systemic vascular resistance. Stimulation of dopaminergic receptors in renal vasculature, leads to renal blood vessel dilation, and an increase in glomerular filtration rate, renal blood flow, sodium excretion, and urine output. The medication form of dopamine works by improving the pumping strength of your heart and improves blood flow to the kidneys. Dopamine injection (Intropin) is used to treat certain conditions that occur when you are in shock, which may be caused by heart attack, trauma, surgery, heart failure, kidney failure, and other serious medical conditions.
During the past 2 decades, dopamine has become the most frequently used sympathomimetic amine in the treatment of neonatal shock 1. The observations that severe and prolonged hypotension may result in the development of irreversible shock, which is associated with increased mortality and central nervous system morbidity, form the rationale behind the early use of dopamine and other sympathomimetic amines in neonates if volume administration fails. However, although cautious volume administration and the early use of dopamine are effective in improving the cardiovascular status and renal function in most neonates who have hypotension, there is only indirect evidence that the use of dopamine (and the other sympathomimetic amines) in the management of neonatal shock improves mortality and morbidity.
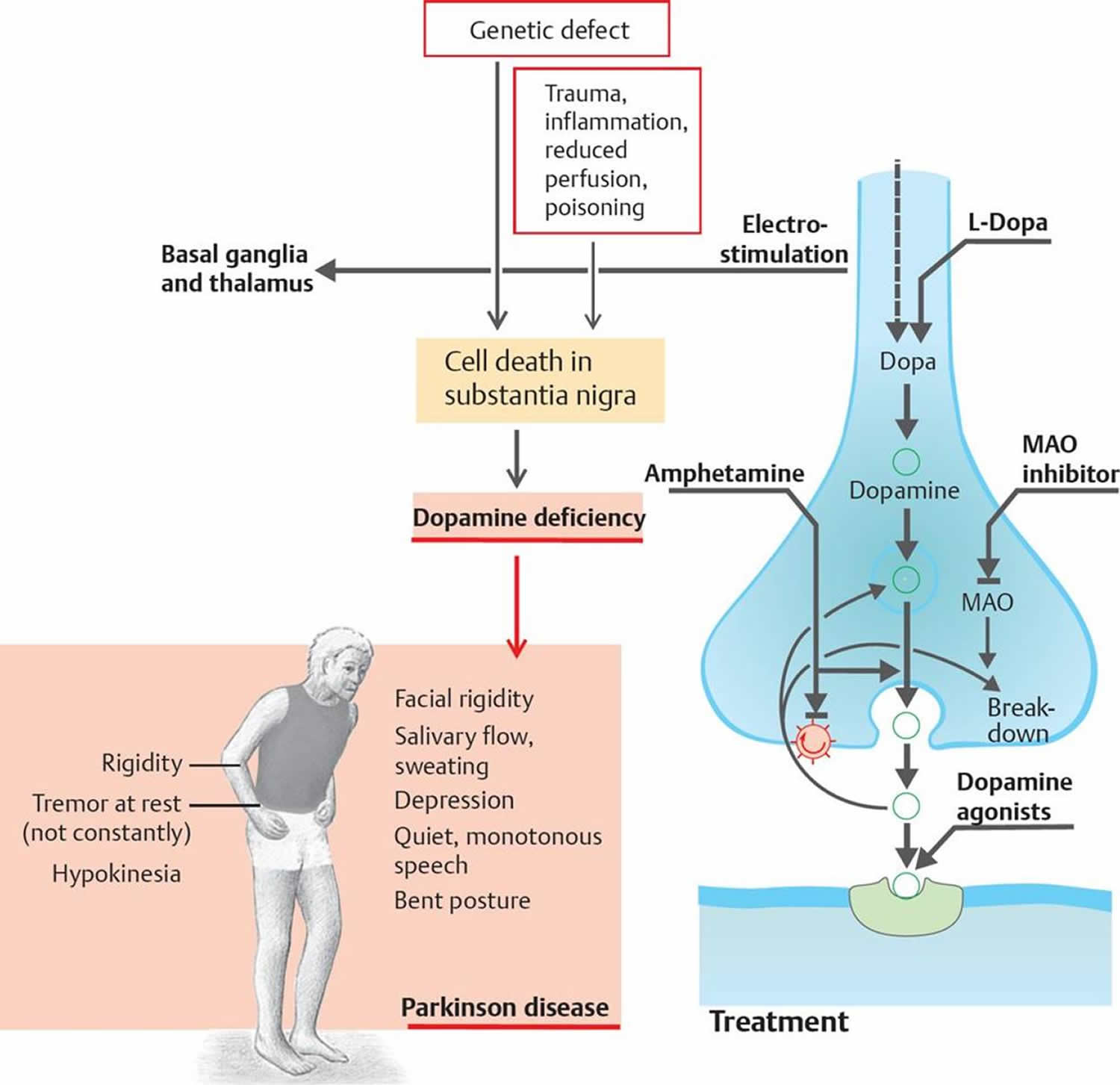
Dopamine is also a major neurotransmitter in the extrapyramidal system of your brain, and important in regulating movement, attention, learning, and the brain’s pleasure and reward system. In 1957, dopamine was shown to occur in the brain, and in 1959 dopamine was found to be enriched in the basal ganglia 2. A reduction in dopamine is responsible for many of the symptoms of Parkinson’s disease. Parkinson’s disease is a brain disorder that leads to shaking, stiffness, and difficulty with walking, balance, and coordination. Parkinson’s disease is caused by a loss of nerve cells in the part of the brain called the substantia nigra. Nerve cells in this part of the brain are responsible for producing a chemical called dopamine. Dopamine acts as a messenger between the parts of the brain and nervous system that help control and co-ordinate body movements. Exactly what causes the loss of nerve cells in Parkinson’s disease is unclear. Parkinson’s disease symptoms usually begin gradually and get worse over time. As the disease progresses, people may have difficulty walking and talking. They may also have mental and behavioral changes, sleep problems, depression, memory difficulties, and fatigue. There’s currently no cure for Parkinson’s disease, but treatments are available to help relieve the symptoms and maintain your quality of life. Unfortunately, taking dopamine as a drug treatment for Parkinson’s disease doesn’t help, because it can’t cross into your brain (the blood brain barrier) where it’s needed. The blood brain barrier (BBB) helps to protect your brain from nasty bacteria and viruses that may have got into your blood. The blood brain barrier (BBB) stops dopamine from passing, which means it cannot get into your brain. Fortunately, levodopa (L-dopa) can cross the blood brain barrier and as it is a ‘precursor’ (chemical building block) to dopamine, your body can turn levodopa into dopamine. When someone takes tablet of levodopa (L-dopa), the idea is that it travels to the brain where cells can turn it into dopamine. Most people with Parkinson’s disease will eventually need a medication called levodopa.
Levodopa (L-dopa) is absorbed by the nerve cells in your brain and turned into the chemical dopamine, which is used to transmit messages between the parts of the brain and nerves that control movement.
Increasing the levels of dopamine using levodopa usually improves movement problems.
But, when a tablet of levodopa is swallowed, the drug doesn’t make its way directly to the brain where it is needed. It is absorbed in the small intestine and makes its way into the blood stream. From there, the blood takes the drug all around your body. And it is here that the early levodopa tablets had their unwanted effects.
Outside the brain, your body contain proteins that break down levodopa. This means much of the drug (around 60–80%) is deactivated before it even has the chance to get into your brain. The first of these proteins, called DOPA decarboxylase, is the protein that turns levodopa into dopamine. Inside the brain this protein vital for the drug to work, but when levodopa is turned into dopamine outside the brain it causes people to feel nauseous.
To counteract this, today’s medications for Parkinson’s combine levodopa with other drugs — such as carbidopa, which blocks DOPA decarboxylase outside the brain— that stop this break down, allowing more of the levodopa to get into the brain and reducing the side effects from having dopamine in our bodies outside the brain.
Levodopa was first combined with other drugs in the 1970s, and the first levodopa-carbidopa medications (trade name Sinemet) were made commercially available. Later attention was turned to Catechol-O-methyltransferase (COMT), another protein that breaks down levodopa outside the brain, and drugs like entacapone (trade name Comtess) and tocapone (trade name Tasmar) were developed to block Catechol-O-methyltransferase (COMT) and allow more levodopa to get into the brain.
If you’re prescribed levodopa, the initial dose is usually very small and will be gradually increased until it takes effect.
At first, levodopa can cause a dramatic improvement in the symptoms. However, its effects can be less long-lasting over the following years – as more nerve cells in the brain are lost, there are fewer of them to absorb the medicine. This means the dose may need to be increased from time to time.
Long-term use of levodopa is also linked to problems such as uncontrollable, jerky muscle movements (dyskinesias) and “on-off” effects, where the person rapidly switches between being able to move (on) and being immobile (off).
Making dopamine stick around
Once levodopa is in the brain it can be converted to dopamine. Dopamine plays a vital function allowing brain cells in the substantia nigra to communicate, to do this the brains cells release this ‘neurotransmitter’ into a space between the cells called the synapse. Another trick to treating Parkinson’s is to make sure the dopamine sticks around as long as possible in this space where it can continue to work. To do this researchers have developed drugs that stop dopamine that has been released into the synapses being recycled.
The protein Catechol-O-methyltransferase (COMT), as well as being outside the brain stopping levodopa getting in, is also found in the brain. Here is has an additional function to recycle dopamine. As tocapone can cross the blood brain barrier, this drug can also block Catechol-O-methyltransferase (COMT) in the brain and allow the dopamine to stick around longer.
The second type of protein that recycles dopamine is MAO-B (monoamine oxidase-B inhibitor). While drugs that act to block MAO (monoamine oxidase) date back to the 1950s, it wasn’t until the 1970s that researchers in Vienna worked out that a drug called selegiline, could be used in Parkinson’s. Selegiline specifically blocked one of the two types of MAO (the B variety, MAO-B). It was 1993, when this drug (trade name Eldepryl) became the first MAO-B inhibitor (monoamine oxidase-B inhibitor) drug to be made available in the UK for the treatment of Parkinson’s, 3 years before it was made available in the US.
Dopamine agonists
Dopamine agonists act as a substitute for dopamine in the brain and have a similar but milder effect compared with levodopa. They can often be given less frequently than levodopa.
Some dopamine agonists are now available as one-a-day tablets, which can be a convenient option for people.
Pramipexole
- Mirapexin (tablets)
- Mirapexin prolonged release (prolonged release tablets)
- Pipexus (modified release tablets)
Ropinirole
- Unbranded (tablets)
- Adartrel (tablets)
- Ralnea XL (prolonged release tablets)
- Requip (tablets)
- Requip XL (prolonged release tablets)
- Spiroco XL (prolonged release tablets)
- Ipinnia XL (prolonged release tablets)
- Raponer XL (prolonged release tablets)
- Ropilynz XL (prolonged release tablets)
Rotigotine
- Neupro (skin patch)
Apomorphine
- APO-go PEN (pre-filled pen for intermittent injection)
- APO-go PFS (pre-filled syringe for infusion. Can be used with a continuous infusion pump)
Bromocriptine
- Unbranded (tablets)
- Parlodel (tablets, capsules)
Cabergoline
- Unbranded (tablets)
- Cabaser (tablets)
- Dostinex (tablets)
Pergolide
- Unbranded (tablets)
They are often taken as a tablet, but are also available as a skin patch (rotigotine ‘Neupro’).
A skin patch (rotigotine ‘Neupro’) may be useful if you have trouble swallowing tablets. The patch should be held in place for 30 seconds, but left on for 24 hours. It may cause a skin reaction, such as reddening or itching, but this is usually mild or moderate. It should only affect the area of skin where the patch has been and will normally disappear a few hours when you remove the patch. Moving the patch to a different part of your body every day will help to avoid irritation. Some, but not all, patches may need to be stored in a refrigerator – check the instructions included with your medication or speak to your pharmacist.
Sometimes dopamine agonists are taken at the same time as levodopa, as this allows lower doses of levodopa to be used.
Possible side effects of dopamine agonists include:
- nausea or vomiting
- tiredness and sleepiness
- dizziness
Dopamine agonists can also cause hallucinations and increased confusion, so they need to be used with caution, particularly in elderly patients, who are more susceptible.
For some people, dopamine agonists have been linked to the development of compulsive behaviors, especially at high doses, including addictive gambling and an excessively increased libido.
Talk to your healthcare specialist if you think you may be experiencing these problems.
As the person themselves may not realize the problem, it’s key that carers and family members also note any abnormal behavior and discuss it with an appropriate professional at the earliest opportunity.
If you’re prescribed a course of dopamine agonists, the initial dose is usually very small to prevent nausea and other side effects.
The dosage is gradually increased over a few weeks. If nausea becomes a problem, your doctor may prescribe anti-sickness medication.
A potentially serious, but uncommon, complication of dopamine agonist therapy is sudden onset of sleep.
This generally happens as the dose is being increased and tends to settle once the dose is stable.
People are usually advised to avoid driving while the dose is being increased in case this complication occurs.
Benefits of dopamine agonists
Delaying levodopa treatment
Dopamine agonists may be an effective treatment for several years when used alone. However, this will not be the case for everyone.
Lowers levodopa needs
Taking dopamine agonists may mean you can take lower doses of levodopa as your condition progresses. This can reduce the risk of experiencing involuntary movements (dyskinesia) or reduce how severe they are.
Fewer movement problems
Your symptoms may be controlled for longer than is usually the case with levodopa. You may also be less prone to long-term side effects such as dyskinesia.
Helping levodopa work better
Dopamine agonists can also be taken with levodopa, at all stages of Parkinson’s. They can help when the effect of levodopa wears off or doesn’t work as well.
Dopamine agonists may help to smooth out the ‘on/off’ effect that you may have with levodopa.
Fewer tablets to take (in some cases)
There are now some once-daily preparations that could make a big difference by reducing the number of tablets you need to take. But this option may not suit everyone.
Positive effects on non-movement symptoms
Some dopamine agonists have recently been shown to have a good effect on the symptoms of Parkinson’s not related to movement, including sleep problems, pain and mood.
When are dopamine agonists used?
Dopamine agonists are used at all stages of Parkinson’s.
You might take them on their own or alongside levodopa to help the levodopa work better.
Treatment with dopamine agonists has to be started carefully. The dose is gradually increased until you and your specialist or Parkinson’s nurse are happy that your symptoms are under control.
Risks and side effects of dopamine agonists
Heart problems
Some of the older dopamine agonists increase the risk of heart problems. As a group, these are known as the ‘ergot’ types and they include bromocriptine, pergolide and cabergoline. Because of this risk, these Parkinson’s drugs are not commonly used.
All newer dopamine agonists are non-ergot in type. These are apomorphine, pramipexole, ropinirole and rotigotine. They have not been associated with an increased risk of heart damage. Doctors prefer to use these if possible.
Sleepiness and fainting
Dopamine agonist drugs can make you feel very sleepy, faint or dizzy. This is most likely to happen when you start taking the drugs. Once a stable dose is reached, this effect often wears off.
Sudden onset of sleep, without any warning, has been reported. If this happens, it’s important that you tell your specialist or Parkinson’s nurse.
Sore skin
If you are having apomorphine injections, soreness or nodules can develop at the place where the needle enters your skin.
If this happens, do not stop the treatment, but make sure you get advice from your specialist or Parkinson’s nurse.
It is important to change the injection site each time to give your skin a chance to heal. Simple massage, silicone gel patches or ultrasound can help to reduce any nodules that form.
Other side effects
The patient information leaflet that comes with your medication will tell you the full range of side effects that you may experience.
Some of the possible side effects include:
- nausea – Apomorphine can cause severe short-term nausea, so you may also be given an anti-sickness drug called domperidone (Motilium). It needs to be started at least two days before your apomorphine treatment begins, and may be gradually reduced over time
- constipation
- impulsive and compulsive behaviour
- low blood pressure (hypotension)
- headaches
- psychological problems
- hallucinations
- movement problems
How do injections and infusion pumps (Apomorphine) work?
Apomorphine is a strong dopamine agonist that is given by injection under the skin (subcutaneously) either by:
- a single injection, when required
- a continuous infusion using a small infusion pump carried around on your belt, under your clothing, or in a bag
It is saved for people with more advanced Parkinson’s who do not respond as well to oral drug treatments, or when most oral drug treatments become less effective or stop working. If you continue to have unpredictable changes in your symptoms that aren’t controlled by other Parkinson’s medications, apomorphine may help.
Apomorphine injections are taken in a similar way to insulin for diabetes. There is a ready-to-use injection pen that works within 5-10 minutes and is often used as a ‘rescue’ measure. This is very useful if you have a sudden ‘off’ period.
If you need more than 7-10 injections a day, you may be changed to a syringe driver. This is a small, battery-driven pump that delivers a continuous dose of medication from a syringe through a needle under the skin.
Ideally, apomorphine will be started in hospital, under the guidance of a specialist or Parkinson’s nurse. Once you are settled on this treatment, you and your carer (if you have one) can be trained in how to use it. If you or a carer are unable to do this, district nurses will be able to help you with this treatment.
APO-go nurse advisors can also start you on treatment. They are registered general nurses with specialist experience in APO-go injections and infusions, as well as the general management of Parkinson’s.
Monoamine oxidase-B inhibitors
Monoamine oxidase-B (MAO-B) inhibitors, including selegiline and rasagiline, are another alternative to levodopa for treating early Parkinson’s disease.
Monoamine oxidase-B (MAO-B) inhibitors block the effects of an enzyme or brain substance that breaks down dopamine (monoamine oxidase-B), increasing dopamine levels.
Types of MAO–B inhibitors:
Rasagiline
- Azilect (tablets)
Selegiline
- Unbranded (tablets)
- Eldepryl (tablets)
- Zelapar (tablets that dissolve on the tongue)
Both selegiline and rasagiline can improve the symptoms of Parkinson’s disease, although their effects are small compared with levodopa. They can be used alongside levodopa or dopamine agonists.
Monoamine oxidase-B (MAO-B) inhibitors inhibitors are generally very well tolerated, but can occasionally cause side effects, including:
- nausea
- headache
- abdominal pain
- high blood pressure
Benefits of MAO–B inhibitors
Improving Parkinson’s disease symptoms
On its own, an MAO–B inhibitor can help with some Parkinson’s symptoms, but the effects are modest and it may become less effective over time.
You don’t have to wait to get the maximum dose
Many drug treatments for Parkinson’s have to be started gradually, with the dose slowly being increased over time. This is not the case for MAO-B inhibitors.
Delaying levodopa treatment
By taking an MAO–B inhibitor at an early stage of Parkinson’s, you may be able to delay taking levodopa until your symptoms become more difficult to manage.
Helping levodopa work better
When you have been taking levodopa for a while, you may find that its effects wear off too quickly. An MAO–B inhibitor can help with this.
Taking an MAO–B inhibitor may also reduce the dose of levodopa you need and lengthen the time between your doses of levodopa.
When are MAO–B inhibitors used?
An MAO–B inhibitor can be used on its own in early Parkinson’s disease, or in combination with other drugs at all stages of Parkinson’s disease.
These drugs are mainly available as tablets.
Risks and side effects of MAO–B inhibitors
Combination with antidepressants
If you’re taking some types of antidepressant, you might not be able to take MAO–B inhibitors, as these drugs can interact with each other to raise blood pressure to a dangerous level.
If you are taking antidepressants, your specialist should be able to advise you on how to take these alongside your Parkinson’s medication.
Combination with decongestants
Decongestants or cold remedies can affect some types of MAO–B inhibitors. If you need to use one of these products, check with your pharmacist to find out which one is safest to use.
Worse levodopa side effects
Because MAO–B inhibitors strengthen levodopa, the side effects of levodopa, including involuntary movements and sickness, may get worse too.
If this happens, your specialist or Parkinson’s nurse can reduce your dose of levodopa.
Other side effects
The patient information leaflet that comes with your medication will tell you the full range of side effects that you may experience.
Some of the possible side effects include:
- headaches
- aching joints
- indigestion
- flu-like symptoms
- depression
Catechol-O-methyltransferase inhibitors
Catechol-O-methyltransferase (COMT) inhibitors are prescribed for people in later stages of Parkinson’s disease. They prevent levodopa from being broken down by the enzyme COMT (Catechol-O-methyltransferase).
Types of COMT inhibitor:
Entacapone
- Comtess (tablets)
Co-careldopa plus entacapone
- Stalevo (tablets)
- Sastravi (tablets)
Tolcapone
- Tasmar (tablets)
Opicapone
- Ongentys (tablets)
Side effects of COMT inhibitors include:
- nausea or vomiting
- diarrhea
- abdominal pain
Benefits of COMT inhibitors
Helping levodopa work more smoothly
COMT inhibitors can help when levodopa is not working for long enough and starts to wear off between doses.
They can help to reduce your ‘off’ time, when you have more trouble with your Parkinson’s symptoms, and increase the amount of ‘on’ time, when your symptoms are better controlled.
Taking less levodopa
In many cases, levodopa can be taken less often and at a lower dose.
Improving Parkinson’s symptoms
Sometimes, adding a COMT inhibitor to your medication regime may help your Parkinson’s symptoms.
When are COMT inhibitors used?
These drugs are used alongside levodopa, to help it work more smoothly. They can be tried if your dose of levodopa is not working for long enough.
COMT inhibitors do not help to manage the symptoms of Parkinson’s on their own – they have to be used with levodopa.
COMT inhibitors come as tablets. If you take entacapone, you should take it at exactly the same time as your levodopa medication to get the best results.
There is also a combined tablet that includes levodopa, carbidopa (one of the ‘helper’ drugs that are always given alongside levodopa) and the COMT inhibitor entacapone.
Risks and side effects of COMT inhibitors
Liver damage
With tolcapone, there is a risk of liver damage that can be fatal. It is rare, but for this reason, it is only used if you can’t take entacapone, where the risk of liver damage has not been seen.
If you take tolcapone, you will need regular blood tests to check the health of your liver.
Worse levodopa side effects
Because COMT inhibitors strengthen levodopa, the side effects of levodopa, including involuntary movements and sickness, can get worse too.
If this happens, your specialist or Parkinson’s nurse can reduce your dose of levodopa.
Other side effects
The patient information leaflet that comes with your medication will tell you the full range of side effects that you may experience.
Some of the possible side effects include:
- sleeping problems
- loss of appetite
- diarrhea
- dizziness
- fainting
- falls
- hallucinations
- headaches
- confusion
- dry mouth
- chest pain
- sleepiness
These drugs can also discolor your urine.
Dopamine synthesis and storage
Dopaminergic neurons are involved in motor activity, in which dopamine is synthesized, stored and released to intersynaptic space. Dopamine is synthesized from the amino acid tyrosine in two steps that occur in the cytosol:
- Hydroxylation of tyrosine to L‐dihydroxyphenylanaline (L‐dopa or levodopa), a reaction catalyzed by tyrosine hydroxylase (TH) that requires oxygen and
- Decarboxylation of L‐dihydroxyphenylanaline (L‐dopa or levodopa) to dopamine, a reaction catalyzed by aromatic amino acid decarboxylase (AADC) that generates CO2.
Interestingly, dopamine synthesis does not result in dopamine accumulation in the cytosol as a consequence that tyrosine hydroxylase (TH) and aromatic amino acid decarboxylase (AADC) are associated with the vesicular monoaminergic transporter‐2 (VMAT‐2) generating a complex where tyrosine is converted to l‐dopa that immediately decarboxylated to dopamine. The latter is taken up into the monoaminergic synaptic vesicles by vesicular monoaminergic transporter‐2 (VMAT‐2) 3, a dopamine transporter localized in the membranes of these vesicles 4. This complex (TH‐AADC‐VMAT‐2) seems to play an important role in the prevention of dopamine oxidation as the protons of the hydroxyl groups are dissociated when dopamine is found in the cytosol at pH 7.4 5. Therefore, dopamine uptake into monoaminergic vesicles prevents both the accumulation of free dopamine in the cytosol and the oxidation of dopamine to o‐quinone because the pH inside monoaminergic synaptic vesicles is 2.0–2.4 pH units lower than the pH in the cytosol 6. At this low pH, the protons of dopamine hydroxyl groups are strongly bound to the oxygen of hydroxyl groups. The low pH of monoaminergic vesicles is because of a vesicular monoaminergic transporter‐2 (VMAT‐2)‐coupled vesicular ATPase, which hydrolyzes ATP to ADP, inorganic phosphate and one proton (H+), creating a proton gradient (Figure 1).
The dopamine stored in monoaminergic vesicles is released to intersynaptic space to interact with dopamine receptors in postsynaptic neurons. The clearance of dopamine from the synaptic clefts is mediated by a dopamine transporter that is localized in the plasma membrane of the dopaminergic neurons. Knockout animals for dopamine transporter present a dopamine deficiency and decreased vesicular storage of dopamine 7. The reuptake of dopamine from the synaptic clefts mediated by dopamine transporter is also a source of free cytosolic dopamine that can be stored in monoaminergic vesicles by the action of vesicular monoaminergic transporter‐2 (VMAT‐2) (Figure 1).
Figure 1. Dopamine synthesis
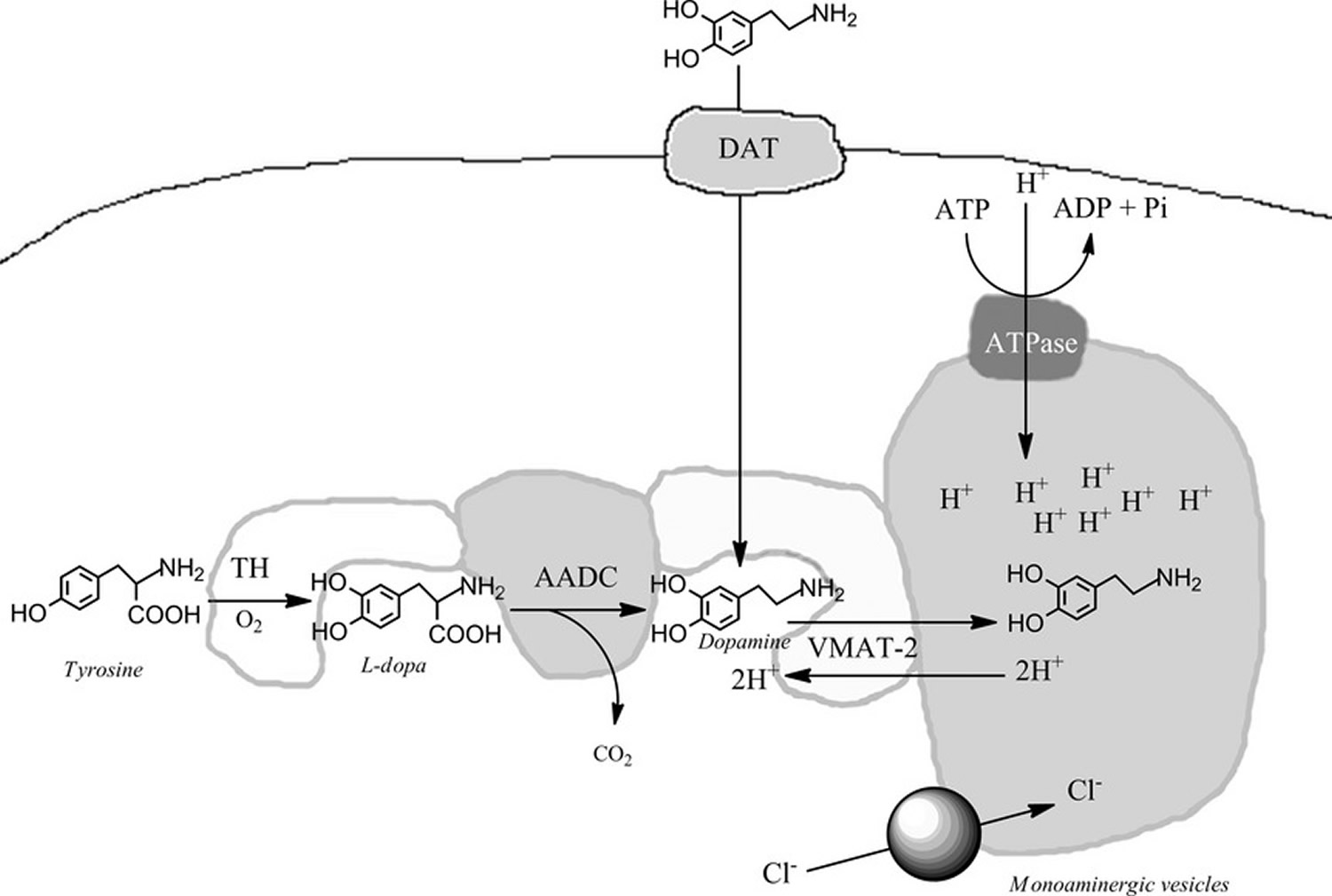 Footnotes: In the presence of oxygen tyrosine hydroxylase (TH) catalyzes the conversion of the amino acid tyrosine to l‐dopa that it is substrate for aromatic amino acid decarboxylase (AADC) that catalyzes the formation of dopamine and CO2. TH and AADC enzymes form a kind of complex with the vesicular monoaminergic transporter‐2 (VMAT‐2) that is localized in the membrane of monoaminergic vesicles. The complex of TH‐AADC‐VMAT‐2 prevents the release of dopamine into the cytosol since the formed dopamine is directly transported into monoaminergic vesicles. The monoaminergic vesicles have an ATPase that pump up protons into the vesicles using ATP, generating a pH decrease and proton gradient that is coupled to dopamine uptake mediated by VMAT‐2 because for one dopamine molecule uptake into the vesicles one proton is released to the cytosol. The low pH inside the monoaminergic vesicles is essential to store high concentration of protonated dopamine hydroxyl groups. [Source 8]
Footnotes: In the presence of oxygen tyrosine hydroxylase (TH) catalyzes the conversion of the amino acid tyrosine to l‐dopa that it is substrate for aromatic amino acid decarboxylase (AADC) that catalyzes the formation of dopamine and CO2. TH and AADC enzymes form a kind of complex with the vesicular monoaminergic transporter‐2 (VMAT‐2) that is localized in the membrane of monoaminergic vesicles. The complex of TH‐AADC‐VMAT‐2 prevents the release of dopamine into the cytosol since the formed dopamine is directly transported into monoaminergic vesicles. The monoaminergic vesicles have an ATPase that pump up protons into the vesicles using ATP, generating a pH decrease and proton gradient that is coupled to dopamine uptake mediated by VMAT‐2 because for one dopamine molecule uptake into the vesicles one proton is released to the cytosol. The low pH inside the monoaminergic vesicles is essential to store high concentration of protonated dopamine hydroxyl groups. [Source 8]Dopamine degradation by monoamine oxidase (MAO)
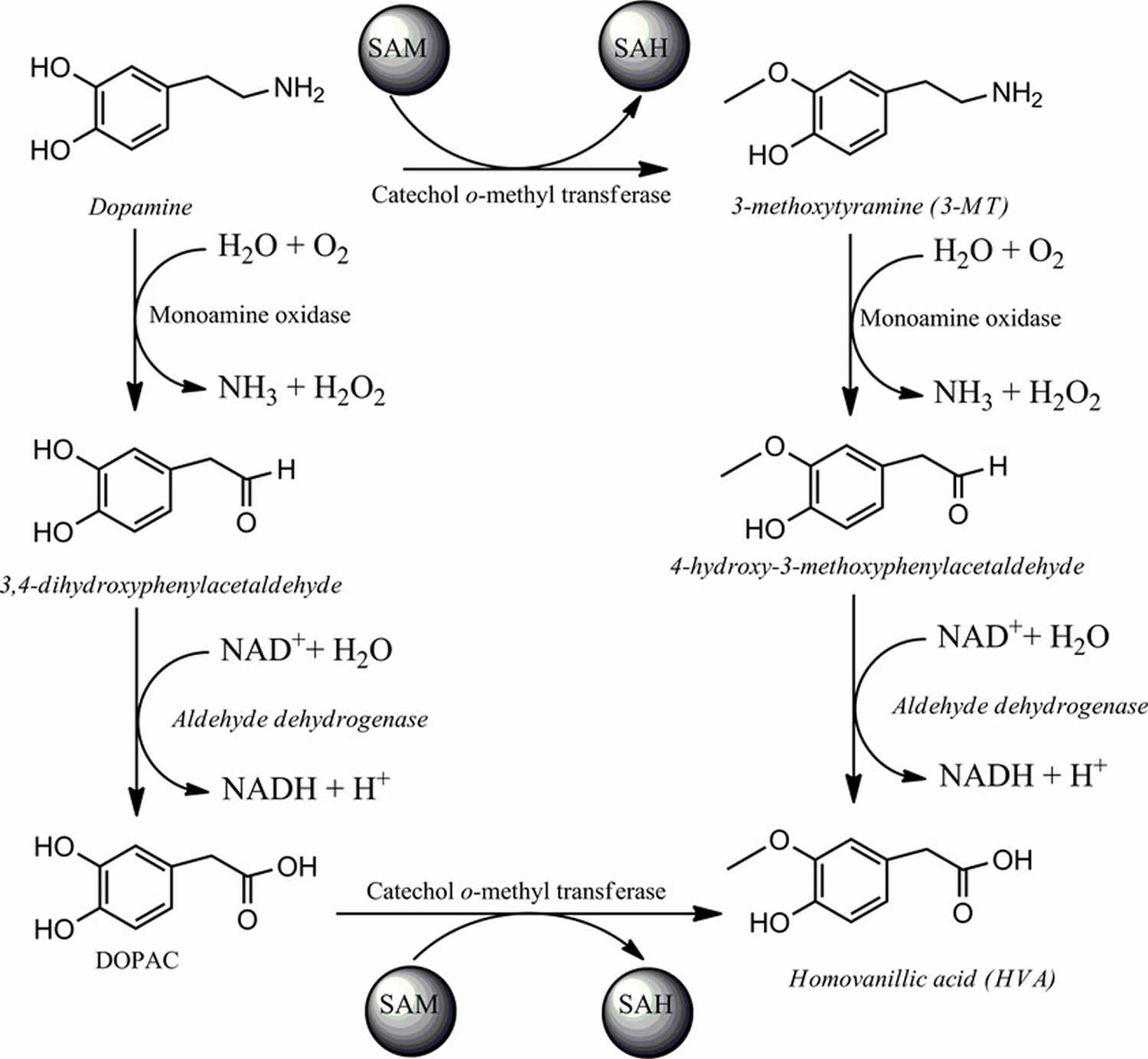
Dopamine excess in the cytosol is degraded by the action of the enzyme monoamine oxidase (MAO) which catalyzes the oxidative deamination of the dopamine amino group to 3,4‐dihydroxyphenylacetaldehyde with concomitant formation of ammonia and hydrogen peroxide. The product of this reaction is metabolized by aldehyde dehydrogenase to 3,4‐dihydroxyphenylacetic acid using NAD as electron donator (Figure 2). The MAO enzymes are flavoenzymes containing FAD 9 where the isozymes labeled as A and B share 70% of primary structure identity. MAO enzymes are found at the outer membranes of mitochondria in neurons and glia cells 10. Monoamine oxidase type B (MAO-B) is expressed in serotonergic and histaminergic neurons as well as in astrocytes 11 whereas monoamine oxidase type A (MAO‐A) is primarily expressed in catecholaminergic neurons.
Monoamine oxidase type A (MAO‐A) has higher affinity for serotonin, dopamine, norepinephrine and epinephrine, whereas the substrates of MAO‐B with high affinity include tyramine, phenylethylamine and MPTP 12. However, monoamine oxidase type B (MAO-B) also uses as substrates dopamine, serotonin and norepinephrine 13. MAO‐A plays a role in maintenance of low concentrations of dopamine in the cytosol and it has been suggested that it plays a role in oxidative stress because the enzyme generates hydrogen peroxide and that it is a precursor of hydroxyl radicals. Rasagiline has been suggested to protect the brain in Parkinson’s disease model systems by preventing the formation of reactive oxygen species catalyzed by MAO‐B 14.
Figure 2. Dopamine degradation catalyzed by monoamine oxidase (MAO) and catechol methyl ortho‐transferase (COMT)
Dopamine degradation by catechol ortho‐methyltransferase (COMT)
Catechol ortho‐methyltransferase (COMT) also participates in dopamine degradation by catalyzing the methylation of dopamine to 3‐methoxytyramine. Interestingly, monoamine oxidase (MAO) also catalyzes the oxidative deamination of 3‐methoxytyramine to 3‐methoxy‐4‐hydroxyphenylacetaldehyde. Another enzyme participating in dopamine degradation is aldehyde dehydrogenase that catalyzes the oxidation of 3‐methoxy‐4‐hydroxyphenylacetaldehyde to homovanillic acid with concomitant formation of NADH. However, COMT also catalyzes the formation of homovanillic acid by methylating the metabolite formed during MAO‐dependent dopamine degradation (see Figure 2).
Both soluble (S‐COMT) and membrane‐bound (MB‐COMT) isoforms have been reported in microglial and astroglial cells. COMT is also found in pyramidal neurons, cerebellar Purkinje, granular cells and striatal spiny neurons 15. Membrane‐bound COMT (MB‐COMT) is localized to the cell body, axons and dendrites of rat cortical neurons.
The catalytic domain of COMT is localized in the C‐terminal that is found in the extracellular space 16. Inhibitors of COMT such as entacapone have been used in combination with levodopa to prolong its half‐life 17.
Dopamine injection
Dopamine injection (Intropin) is used to treat certain conditions that occur when you are in shock, which may be caused by heart attack, trauma, surgery, heart failure, kidney failure, and other serious medical conditions.
Dopamine is injected into a vein through an IV. A healthcare provider will give you this injection.
Since the half-life of dopamine in plasma is very short—approximately one minute in adults, two minutes in newborn infants and up to five minutes in preterm infants—it is usually given in a continuous intravenous drip rather than a single injection 18.
Before having dopamine injection
You should not be treated with dopamine if you have pheochromocytoma (tumor of the adrenal gland).
Tell your doctor about all the prescription and over-the-counter medications you use, especially if you have used an MAO inhibitor such as furazolidone (Furoxone), isocarboxazid (Marplan), phenelzine (Nardil), rasagiline (Azilect), selegiline (Eldepryl, Emsam, Zelapar), or tranylcypromine (Parnate) in the last 21 days.
In an emergency situation it may not be possible to tell your caregivers about your health conditions. Make sure any doctor caring for you afterward knows you have received this medicine.
If possible before you receive dopamine, tell your doctor if you have ever had:
- a heart rhythm disorder;
- coronary artery disease (clogged arteries);
- a blood clot;
- an allergy to any foods or drugs;
- asthma or sulfite allergy;
- metabolic acidosis;
- diabetes;
- circulation problems such as Raynaud’s syndrome;
- frostbite;
- Buerger’s disease; or
if you have used an MAO inhibitor in the past 21 days (such as isocarboxazid, linezolid, methylene blue injection, phenelzine, rasagiline, selegiline, tranylcypromine, and others).
In an emergency situation it may not be possible to tell your caregivers if you are pregnant or breast-feeding. Make sure any doctor caring for your pregnancy or your baby knows you have received this medicine.
Dopamine injection side effects
Get emergency medical help if you have signs of an allergic reaction to dopamine: hives; difficult breathing; swelling of your face, lips, tongue, or throat.
Tell your caregivers at once if you have:
- a light-headed feeling, like you might pass out (even while lying down);
- chest pain;
- fast, slow, or pounding heartbeats;
- shortness of breath;
- cold feeling, numbness, or blue-colored appearance in your hands or feet; or
- darkening or skin changes in your hands or feet.
Common dopamine side effects may include:
- headache;
- feeling anxious;
- nausea, vomiting; or
- chills, goose bumps.
Cardiovascular
Gangrene of the feet occurred in patients with preexisting vascular disorders and/or at doses of at least 10 to 14 mcg/kg/min given for prolonged periods of time.
Ventricular arrhythmias occurred at very high doses.
- Common (1% to 10%): Ectopic beats, tachycardia, anginal pain, palpitation, hypotension, vasoconstriction
- Uncommon (0.1% to 1%): Aberrant ventricular conduction/ventricular arrhythmia, bradycardia, widened QRS complex, hypertension
- Frequency not reported: Gangrene of the feet/extremities, peripheral cyanosis, fatal ventricular arrhythmias, atrial fibrillation, cardiac conduction abnormalities
Gastrointestinal
- Common (1% to 10%): Nausea, vomiting
Nervous system
- Common (1% to 10%): Headache
Respiratory
- Common (1% to 10%): Dyspnea
Dermatologic
- Very rare (less than 0.01%): Gangrene
- Frequency not reported: Piloerection
Local
- Necrosis and tissue sloughing may be caused by extravasation of this drug.
- Frequency not reported: Necrosis, tissue sloughing, local ischemia and vasoconstriction
Psychiatric
- Frequency not reported: Anxiety
Renal
- Frequency not reported: Azotemia
Ocular
- Frequency not reported: Mydriasis
Metabolic
- Frequency not reported: Hypovolemia
This is not a complete list of side effects and others may occur.
Serotonin vs Dopamine
Serotonin or 5-HT (5-Hydroxytryptamine) is a neurotransmitter (a messenger chemical that carries signals between nerve cells in the brain) synthesized from the essential amino acid L-Tryptophan. Serotonin (5-Hydroxytryptamine) in the nervous system acts as a local transmitter at synapses, and as a paracrine or hormonal modulator of circuits upon diffusion, allowing a wide variety of “state-dependent” behavioral responses to different stimuli. Serotonin is widely distributed in the nervous system of vertebrates and some of its behavioral effects have been preserved along evolution. Such is the case of aggressive behavior and rhythmic motor patterns , including those responsible for feeding. In vertebrates, serotonin also modulates sleep, the arousal state , sexual behavior, and others, and deficiencies of the serotonergic system causes disorders such as depression, obsessive-compulsive disorder, phobias, posttraumatic stress disorder, epilepsy, and generalized anxiety disorder. Serotonin has three different modes of action in the nervous system: as transmitter, acting locally at synaptic boutons; upon diffusion at a distance from its release sites, producing paracrine (also called volume) effects, and by circulating in the blood stream, producing hormonal effects. The three modes can affect a single neuronal circuit 19.
Serotonin (5-HT) is thought to have a good influence on mood, emotion and sleep. In the central nervous system, serotonin (5-HT) pathways modulate behaviors, eating, and sleep, whereas, in the gut, they are involved in the regulation of gastrointestinal motility 20. Serotonin is a chemical derived from the amino acid tryptophan. It is produced as needed by the nervous system, mainly the brain, but also by special cells in the bronchial tubes (lungs) and gastrointestinal tract. More than 90% of serotonin in the blood is found in the platelets. Serotonin helps transmit nerve impulses and constrict blood vessels, is a participant in the wake-sleep cycle, and affects mood. Serotonin is metabolized by the liver and its metabolites, primarily 5-HIAA (5-hydroxyindoleacetic acid), are eliminated in the urine. Fruits, vegetables, and seeds are major sources of 5-HT (serotonin) (Figure 3). In recent years, the number of studies on the content of serotonin (5-HT) in plants has increased, greatly encouraged by the discovery of melatonin, which stimulates the late vegetative growth of different tissue sections 21. Serotonin (5-HT) appeared to be prevalent in the green fruit of the Musa genus (that is, prata banana, and other species), containing about 7100–21,000 ng/g of fresh weight, followed by a significant decrease during ripening 22. Higher concentrations were found in banana peels compared to the pulp 23. The accumulation of 5-HT was also detected in Capsicum annuum L. (that is, pepper) 24, and paprika 25. 5-HT was identified in Corylus avellana L. (that is, hazelnut) 26, fruits of tomato and cherry tomato 25, Ananas comosus L. (that is, pineapple) 27, Prunus domestica L. (that is, plum) 23, Passiflora edulis S. (that is, passion fruit), Carica papaya L. (that is, pawpaw) 28, and in fruits of the Actinidia genus (that is, kiwi) 29. Similar to dopamine, 5-HT was found in the Mucuna pruriens or velvet bean 30. Ly et al. detected about 34,400 ng/g of dry weight in spinach 25. Brassica rapa L. (that is, Chinese cabbage) 25, potato leaves 31, rice plant, and seeds of Oryza sativa L. (that is, wild rice) 32, were also considered sources of 5-HT. This neurotransmitter was found in green coffee beans and, because of its high resistance to roasting, even in coffee powders 33. Traces were found in Punica granatum L. (that is, pomegranate), fruits of the Fragaria genus (that is, strawberry) 34, Cichorium intybus L. (that is, chicory), Allium ascalonicum L. (that is, green onion), and Lactuca sativa L. (that is, lettuce) 25. Some plants, such as nettle 35 and Griffonia simplicifolia DC were found to contain serotonin. Griffonia was marketed for its presumptive anxiolytic effects that were later associated with the content of 5-hydroxy-l-tryptophan, a direct precursor in the synthesis of serotonin 36.
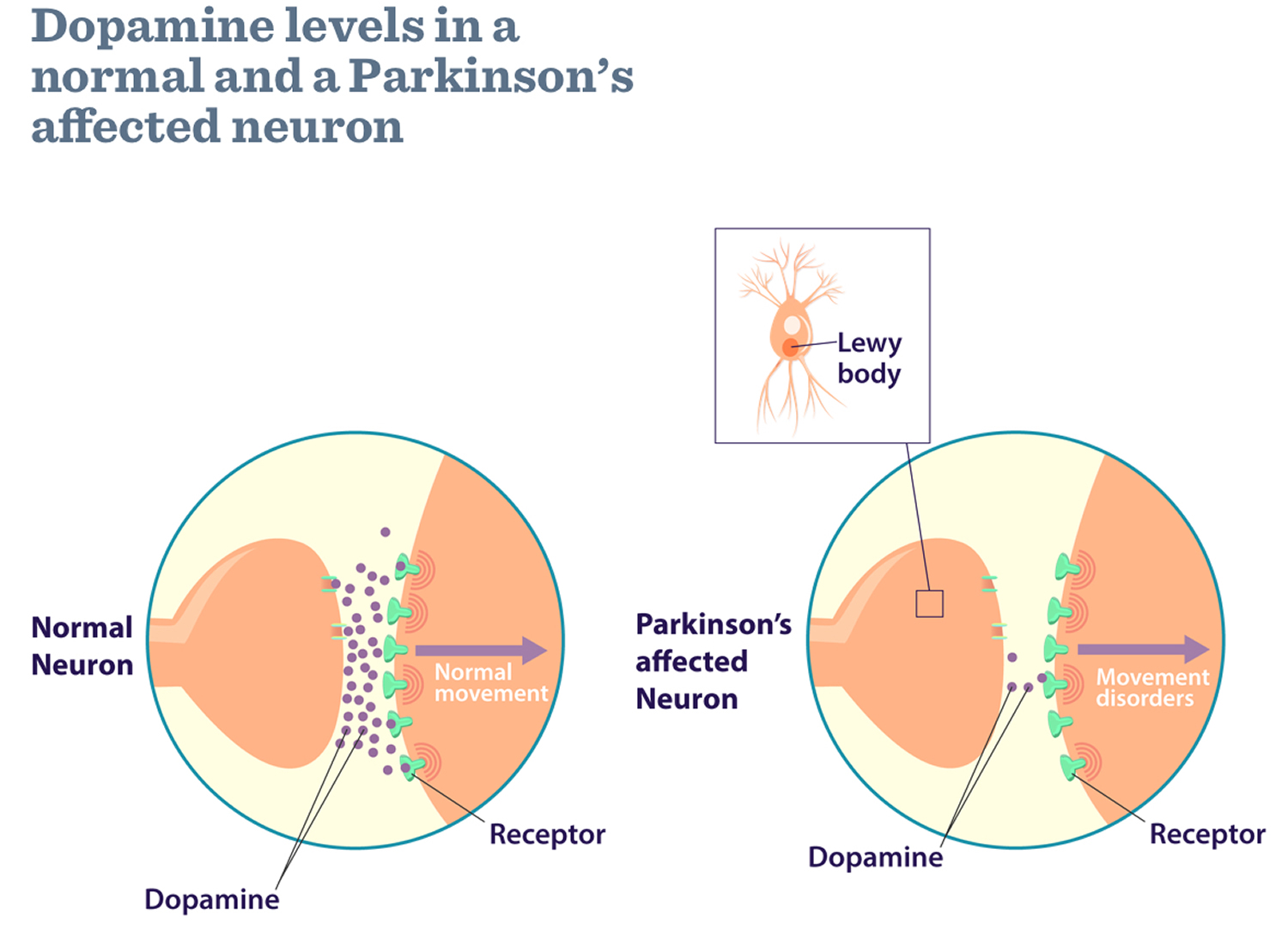
Dopamine deficiency
A brain dopamine deficiency is the defining biochemical feature of Parkinson’s disease. Parkinson’s disease occurs when nerve cells, or neurons, in an area of the brain that controls movement called the substantia nigra become impaired and/or die. Normally, these neurons in substantia nigra produce an important brain chemical known as dopamine. When the neurons die or become impaired, they produce less dopamine in the brain, which causes the movement problems of Parkinson’s disease (movements become slow and abnormal). The loss of nerve cells is a slow process. The symptoms of Parkinson’s disease usually only start to develop when around 80% of the nerve cells in the substantia nigra have been lost. Scientists still do not know what causes cells that produce dopamine to die. Most experts think that a combination of genetic and environmental factors is responsible. Both men and women can have Parkinson’s disease. However, the disease affects about 50 percent more men than women.
People with Parkinson’s also lose the nerve endings that produce norepinephrine (noradrenaline), the main chemical messenger of the sympathetic nervous system, which controls many automatic functions of the body, such as heart rate and blood pressure. The loss of norepinephrine might help explain some of the non-movement features of Parkinson’s, such as fatigue, irregular blood pressure, decreased movement of food through the digestive tract, and sudden drop in blood pressure when a person stands up from a sitting or lying-down position.
Many brain cells of people with Parkinson’s disease contain Lewy bodies, unusual clumps of the protein alpha-synuclein. Scientists are trying to better understand the normal and abnormal functions of alpha-synuclein and its relationship to genetic mutations that impact Parkinson’s disease and Lewy body dementia.
Although some cases of Parkinson’s disease appear to be hereditary, and a few can be traced to specific genetic mutations, in most cases the disease occurs randomly and does not seem to run in families. Many researchers now believe that Parkinson’s disease results from a combination of genetic factors and environmental factors such as exposure to toxins. One clear risk factor for Parkinson’s disease is age. Although most people with Parkinson’s disease first develop the disease at about age 60, about 5 to 10 percent of people with Parkinson’s have “early-onset” disease, which begins before the age of 50. Early-onset forms of Parkinson’s disease are often, but not always, inherited, and some forms have been linked to specific gene mutations.
What causes the loss of nerve cells in Parkinson’s disease?
It’s not known why the loss of nerve cells associated with Parkinson’s disease occurs, although research is ongoing to identify potential causes.
Currently, it’s believed a combination of genetic changes and environmental factors may be responsible for the condition.
Genetics
A number of genetic factors have been shown to increase a person’s risk of developing Parkinson’s disease, although exactly how these make some people more susceptible to the condition is unclear.
The discovery of genes associated with Parkinson’s disease (such as alpha synuclein (SNCA), E3 ubiquitin protein ligase (parkin), DJ‐1 (PARK7), ubiquitin carboxyl‐terminal hydrolase isozyme L1 (UCHL‐1), serine/threonine‐protein kinase (PINK‐1), leucine‐rich repeat kinase 2 (LRRK2), cation‐transporting ATPase 13A1 (ATP13A), etc.) contributed enormously to basic research towards understanding the role of these proteins in the sporadic form of Parkinson’s disease 8. However, it is generally accepted by the scientific community that mitochondria dysfunction, alpha synuclein aggregation, dysfunction of protein degradation, oxidative stress and neuroinflammation are involved in neurodegeneration 8.
Parkinson’s disease can also run in families as a result of faulty genes being passed to a child by their parents. However, it’s rare for Parkinson’s disease to be inherited this way.
Environmental factors
Some researchers also feel environmental factors may increase a person’s risk of developing Parkinson’s disease.
It’s been suggested that pesticides and herbicides used in farming and traffic or industrial pollution may contribute to the condition.
However, the evidence linking environmental factors to Parkinson’s disease is inconclusive.
Other causes of parkinsonism
“Parkinsonism” is the umbrella term used to describe the symptoms of tremors, muscle rigidity and slowness of movement.
Parkinson’s disease is the most common type of parkinsonism, but there are also some rarer types where a specific cause can be identified.
These include parkinsonism caused by:
- medication (drug-induced parkinsonism) – where symptoms develop after taking certain medications, such as some types of antipsychotic medication, and usually improve once the medication is stopped
- other progressive brain conditions – such as progressive supranuclear palsy, multiple systems atrophy, and corticobasal degeneration
- cerebrovascular disease – where a series of small strokes cause several parts of the brain to die
Symptoms of Parkinson’s Disease
Parkinson’s disease has four main symptoms 37:
- Tremor (trembling) in hands, arms, legs, jaw, or head – shaking, which usually begins in the hand or arm and is more likely to occur when the limb is relaxed and resting
- Stiffness and inflexible muscles of the limbs and trunk – stiffness and tension in the muscles, which can make it difficult to move around and make facial expressions, and can result in painful muscle cramps (dystonia)
- Slowness of movement (bradykinesia) – where physical movements are much slower than normal, which can make everyday tasks difficult and can result in a distinctive slow, shuffling walk with very small steps
- Impaired balance and coordination, sometimes leading to falls
These main symptoms are sometimes referred to by doctors as parkinsonism as there can be causes other than Parkinson’s disease.
Parkinson’s disease can also cause a range of other physical and mental symptoms.
Other symptoms may include depression and other emotional changes; difficulty swallowing, chewing, and speaking; urinary problems or constipation; skin problems; and sleep disruptions.
Physical symptoms
- balance problems – these can make someone with the condition more likely to have a fall and injure themselves
- loss of sense of smell (anosmia) – sometimes occurs several years before other symptoms develop
- nerve pain – can cause unpleasant sensations, such as burning, coldness or numbness
- problems with urination – such as having to get up frequently during the night to urinate or unintentionally passing urine (urinary incontinence)
- constipation
- an inability to obtain or sustain an erection (erectile dysfunction) in men
- difficulty becoming sexually aroused and achieving an orgasm (sexual dysfunction) in women
- dizziness, blurred vision or fainting when moving from a sitting or lying position to a standing one – caused by a sudden drop in blood pressure
- excessive sweating (hyperhidrosis)
- swallowing difficulties (dysphagia) – this can lead to malnutrition and dehydration
- excessive production of saliva (drooling)
- problems sleeping (insomnia) – this can result in excessive sleepiness during the day
Cognitive and psychiatric symptoms
- depression and anxiety
- mild cognitive impairment – slight memory problems and problems with activities that require planning and organization
- dementia – a group of symptoms, including more severe memory problems, personality changes, seeing things that aren’t there (visual hallucinations) and believing things that aren’t true (delusions)
Symptoms of Parkinson’s disease and the rate of progression differ among individuals. Sometimes people dismiss early symptoms of Parkinson’s disease as the effects of normal aging. In most cases, there are no medical tests to definitively detect Parkinson’s disease, so it can be difficult to diagnose accurately.
Early symptoms of Parkinson’s disease are subtle and occur gradually. For example, affected people may feel mild tremors or have difficulty getting out of a chair. They may notice that they speak too softly, or that their handwriting is slow and looks cramped or small. Friends or family members may be the first to notice changes in someone with early Parkinson’s disease. They may see that the person’s face lacks expression and animation, or that the person does not move an arm or leg normally.
People with Parkinson’s disease often develop a parkinsonian gait that includes a tendency to lean forward, small quick steps as if hurrying forward, and reduced swinging of the arms. They also may have trouble initiating or continuing movement.
Symptoms often begin on one side of the body or even in one limb on one side of the body. As Parkinson’s disease progresses, it eventually affects both sides. However, the symptoms may still be more severe on one side than on the other.
Many people with Parkinson’s disease note that prior to experiencing stiffness and tremor, they had sleep problems, constipation, decreased ability to smell, and restless legs.
Diagnosis of Parkinson’s Disease
There are currently no blood or laboratory tests to diagnose nongenetic cases of Parkinson’s disease. Diagnosis is based on a person’s medical history and a neurological examination. Improvement after initiating medication is another important hallmark of Parkinson’s disease.
A diagnosis of Parkinson’s disease is likely if you have at least two of the three following symptoms:
- shaking or tremor in a part of your body that usually only occurs at rest
- slowness of movement (bradykinesia)
- muscle stiffness (rigidity)
If your symptoms improve after taking a medication called levodopa (L-dopa), it’s more likely you have Parkinson’s disease.
Special brain scans, such as a single photon emission computed tomography (SPECT) scan, may also be carried out in some cases to try to rule out other causes of your symptoms.
A number of disorders can cause symptoms similar to those of Parkinson’s disease. People with Parkinson’s-like symptoms that result from other causes are sometimes said to have parkinsonism. While these disorders initially may be misdiagnosed as Parkinson’s disease, certain medical tests, as well as response to drug treatment, may help to distinguish them from Parkinson’s disease. Since many other diseases have similar features but require different treatments, it is important to make an exact diagnosis as soon as possible.
Treatment of Parkinson’s Disease
Although there is no cure for Parkinson’s disease, medicines, surgical treatment, and other therapies can often relieve some symptoms.
Medicines for Parkinson’s Disease
Medicines prescribed for Parkinson’s include:
- Drugs that increase the level of dopamine in the brain
- Drugs that affect other brain chemicals in the body
- Drugs that help control nonmotor symptoms
Unfortunately, taking dopamine as a drug treatment for Parkinson’s disease doesn’t help, because it can’t cross into your brain where it’s needed. However, you can boost the supply in your brain by taking levodopa, a drug that acts as a chemical building block to help your body produce more dopamine.
The main therapy for Parkinson’s disease is levodopa, also called L-dopa. Nerve cells use levodopa to make dopamine to replenish the brain’s dwindling supply. Usually, people take levodopa along with another medication called carbidopa. Carbidopa is an inhibitor of dopa decarboxylase, preventing the conversion of levodopa to dopamine in your body so more levodopa can reach your brain. Carbidopa prevents or reduces some of the side effects of levodopa therapy—such as nausea, vomiting, low blood pressure, and restlessness—and reduces the amount of levodopa needed to improve symptoms.
Levodopa (L-dopa) is usually taken as a tablet or liquid, and is often combined with other medication, such as benserazide or carbidopa. These medications stop the levodopa being broken down in the bloodstream before it has a chance to get to the brain. They also reduce the side effects of levodopa, which include:
- feeling sick (nausea) or vomiting
- tiredness
- dizziness
People with Parkinson’s disease should never stop taking levodopa without telling their doctor. Suddenly stopping levodopa (L-dopa) may have serious side effects, such as being unable to move or having difficulty breathing.
Other medicines used to treat Parkinson’s disease symptoms include:
- Dopamine agonists to mimic the role of dopamine in the brain
- Monoamine oxidase-B inhibitors (MAO-B) inhibitors to slow down an enzyme that breaks down dopamine in the brain
- Catechol-O-methyltransferase (COMT) inhibitors to help break down dopamine
- Amantadine, an old antiviral drug, to reduce involuntary movements
- Anticholinergic drugs to reduce tremors and muscle rigidity
Randomized control trials showed that levodopa-carbidopa tablets taken with entacapone or tocapone can help to reduce motor symptom fluctuations, although they weren’t without side effects, and these individual drugs have been on the market since the late 1990s. Entacapone has since been made into combined medications where this drug is in the same tablet as levodopa and carbidopa (e.g. Stalevo or Sastravi). However, none of these medications can slow the progression of Parkinson’s.
Non-oral therapies
When Parkinson’s symptoms become difficult to control with tablets alone, a number of other treatments can be considered.
Apomorphine
A dopamine agonist called apomorphine can be injected under the skin (subcutaneously) either by:
- a single injection, when required
- a continuous infusion using a small pump carried around on your belt, under your clothing, or in a bag
Duodopa
If you have severe on-off fluctuations, a type of levodopa called duodopa may be used.
This medication comes as a gel that’s continuously pumped into your gut through a tube inserted through your abdominal wall.
There’s an external pump attached to the end of the tube, which you carry around with you.
This treatment is only available if you have very severe on-off fluctuations or involuntary movements.
Deep Brain Stimulation
For people with Parkinson’s disease who do not respond well to medications, deep brain stimulation, or DBS, may be appropriate. Deep Brain Stimulation is a surgical procedure that surgically implants electrodes into part of the brain and connects them to a small electrical device implanted in the chest. The device and electrodes painlessly stimulate the brain in a way that helps stop many of the movement-related symptoms of Parkinson’s, such as tremor, slowness of movement, and rigidity.
Other Therapies
Other therapies may be used to help with Parkinson’s disease symptoms. They include physical, occupational, and speech therapies, which help with gait and voice disorders, tremors and rigidity, and decline in mental functions. Other supportive therapies include a healthy diet and exercises to strengthen muscles and improve balance.
Parkinson’s disease prognosis
Parkinson’s disease is both chronic, meaning it persists over a long period of time, and progressive, meaning its symptoms grow worse over time. Although some people become severely disabled, others experience only minor motor disruptions. Tremor is the major symptom for some individuals, while for others tremor is only a minor complaint and other symptoms are more troublesome. It is currently not possible to predict which symptoms will affect an individual, and the intensity of the symptoms also varies from person to person.
As the condition progresses, the symptoms of Parkinson’s disease can get worse and it can become increasingly difficult to carry out everyday activities without assistance.
Many people respond well to treatment and only experience mild to moderate disability, whereas the minority may not respond as well and can, in time, become more severely disabled.
Parkinson’s disease doesn’t directly cause people to die, but the condition can place great strain on the body, and can make some people more vulnerable to serious and life-threatening infections.
However, with advances in treatment, most people with Parkinson’s disease now have a normal or near-normal life expectancy.
- Pharmacology Review. NeoReviews Oct 2003, 4 (10) e283-e288; DOI: 10.1542/neo.4-10-e283[↩]
- A brief history of levodopa. J Neurol (2010) 257(Suppl 2): 249. https://doi.org/10.1007/s00415-010-5741-y[↩]
- Cartier E. A., Parra L. A., Baust T. B., Quiroz M., Salazar G., Faundez V., Egaña L. and Torres G. E. (2010) A biochemical and functional protein complex involving dopamine synthesis and transport into synaptic vesicles. J. Biol. Chem. 151, 957–966.[↩]
- Miller G. W., Erickson J. D., Perez J. T., Penland S. N., Mash D. C., Rye D. B. and Levey A. I. (1999) Immunochemical analysis of vesicular monoamine transporter (VMAT2) protein in Parkinson’s disease. Exp. Neurol. 156, 138–148.[↩]
- Linert W., Herlinger E., Jameson R. F., Kienzl E., Jellinger K. and Youdim M. B. (1996) Dopamine, 6‐hydroxydopamine, iron, and dioxygen–their mutual interactions and possible implication in the development of Parkinson’s disease. Biochim. Biophys. Acta 1316, 160–168.[↩]
- Guillot T. S. and Miller G. W. (2009) Protective actions of the vesicular monoamine transporter 2 (VMAT2) in monoaminergic neurons. Mol. Neurobiol. 39, 149–170.[↩]
- Eriksen J., Jørgensen T. N. and Gether U. (2010) Regulation of dopamine transporter function by protein‐protein interactions: new discoveries and methodological challenges. J. Neurochem. 113, 27–41.[↩]
- Protective and toxic roles of dopamine in Parkinson’s disease. Journal of Neurochemistry 18 Feb 2014. https://onlinelibrary.wiley.com/doi/full/10.1111/jnc.12686[↩][↩][↩][↩]
- Bach A. W. J., Lan N. C., Johnson D. L., Abell C. W., Bemkenek M. E., Kwan S. W., Seeburg P. H. and Shih J. C. (1988) cDNA cloning of human liver monoamine oxidase A and B: molecular basis of differences in enzymatic properties. Proc. Natl Acad. Sci. USA 85, 4934–4938.[↩]
- Shih J. C., Grimsby J. and Chen K. (1997) Molecular biology of monoamine oxidase A and B: their role in the degradation of serotonin, in Handbook of Experimental Pharmacology. Serotoninergic Neurons and 5‐HT Receptors in the CNS (Baumgarten H. G. and Gothert M., eds), Vol. 129, pp. 655–670. Springer‐Verlag, Berlin.[↩]
- Saura J., Luque J. M., Cesura A. M., Da Prada M., Chan‐Palay V., Huber G., Loffler J. and Richards J. G. (1994) Increased monoamine oxidase B activity in plaque‐associated astrocytes of Alzheimer brains revealed by quantitative enzyme radioautography. Neuroscience 62, 15–30.[↩]
- Cases O., Seif I., Grimsby J. et al. (1995) Aggressive behavior and altered amounts of brain serotonin and norepinephrine in mice lacking MAOA. Science 268, 1763–1766.[↩]
- Geha R. M., Chen K., Wouters J., Ooms F. and Shih J. C. (2002) Analysis of conserved active site residues in monoamine oxidase A and B and their three‐dimensional molecular modeling. J. Biol. Chem. 277, 17209–17216.[↩]
- Weinreb O., Amit T., Riederer P., Youdim M. B. and Mandel S. A. (2011) Neuroprotective profile of the multitarget drug rasagiline in Parkinson’s disease. Int. Rev. Neurobiol. 100, 127–149.[↩]
- Myöhänen T. T., Schendzielorz N. and Männistö P. T. (2010) Distribution of catechol‐O‐methyltransferase (COMT) proteins and enzymatic activities in wild‐type and soluble COMT deficient mice. J. Neurochem. 113, 1632–1643.[↩]
- Chen J., Song J., Yuan P., Tian Q., Ji Y., Ren‐Patterson R., Liu G., Sei Y. and Weinberger D. R. (2011) Orientation and cellular distribution of membrane‐bound catechol‐O‐methyltransferase in cortical neurons: implications for drug development. J. Biol. Chem. 286, 34752–34760.[↩]
- Marin C. and Obeso J. A. (2010) Catechol‐O‐methyltransferase inhibitors in preclinical models as adjuncts of L‐dopa treatment. Int. Rev. Neurobiol. 95, 191–205.[↩]
- Dopamine and dobutamine in pediatric therapy. Pharmacotherapy. 1989;9(5):303-14. https://www.ncbi.nlm.nih.gov/pubmed/2682552[↩]
- Synaptic and extrasynaptic secretion of serotonin. Cell Mol Neurobiol. 2005 Mar;25(2):297-312. https://www.ncbi.nlm.nih.gov/pubmed/16047543[↩]
- Briguglio M, Dell’Osso B, Panzica G, et al. Dietary Neurotransmitters: A Narrative Review on Current Knowledge. Nutrients. 2018;10(5):591. doi:10.3390/nu10050591 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5986471/[↩]
- Huang X., Mazza G. Application of LC and LC-MS to the analysis of melatonin and serotonin in edible plants. Crit. Rev. Food Sci. Nutr. 2011;51:269–284. doi: 10.1080/10408398.2010.529193[↩]
- Adão R.C., Glória M.B.A. Bioactive amines and carbohydrate changes during ripening of ‘Prata’ banana (Musa acuminata × M. balbisiana) Food Chem. 2005;90:705–711. doi: 10.1016/j.foodchem.2004.05.020[↩]
- Udenfriend S., Lovenberg W., Sjoerdsma A. Physiologically active amines in common fruits and vegetables. Arch. Biochem. Biophys. 1959;85:487–490. doi: 10.1016/0003-9861(59)90516-8[↩][↩]
- Kang S., Back K. Enriched production of N-hydroxycinnamic acid amides and biogenic amines in pepper (Capsicum annuum) flowers. Sci. Hortic. 2006;108:337–341. doi: 10.1016/j.scienta.2006.01.037[↩]
- Ly D., Kang K., Choi J.Y., Ishihara A., Back K., Lee S.G. HPLC analysis of serotonin, tryptamine, tyramine, and the hydroxycinnamic acid amides of serotonin and tyramine in food vegetables. J. Med. Food. 2008;11:385–389. doi: 10.1089/jmf.2007.514[↩][↩][↩][↩][↩]
- Lavizzari T., Teresa Veciana-Nogues M., Bover-Cid S., Marine-Font A., Carmen Vidal-Carou M. Improved method for the determination of biogenic amines and polyamines in vegetable products by ion-pair high-performance liquid chromatography. J. Chromatogr. A. 2006;1129:67–72. doi: 10.1016/j.chroma.2006.06.090[↩]
- Foy J.M., Parratt J.R. 5-Hydroxytryptamine in pineapples. J. Pharm. Pharmacol. 1961;13:382–383. doi: 10.1111/j.2042-7158.1961.tb11840.x[↩]
- Council N.R. Toxicants Occurring Naturally in Foods. National Academy of Sciences; Washington, DC, USA: 1973.[↩]
- Feldman J.M., Lee E.M. Serotonin content of foods: Effect on urinary excretion of 5-hydroxyindoleacetic acid. Am. J. Clin. Nutr. 1985;42:639–643. doi: 10.1093/ajcn/42.4.639[↩]
- Bowden K., Brown B.G., Batty J.E. 5-Hydroxytryptamine: Its occurrence in cowhage. Nature. 1954;174:925–926. doi: 10.1038/174925a0[↩]
- Engstrom K., Lundgren L., Samuelsson G. Bioassay-guided isolation of serotonin from fruits of Solanum tuberosum L. Acta Pharm. Nord. 1992;4:91–92[↩]
- Kang S., Kang K., Lee K., Back K. Characterization of tryptamine 5-hydroxylase and serotonin synthesis in rice plants. Plant Cell Rep. 2007;26:2009–2015. doi: 10.1007/s00299-007-0405-9[↩]
- Ramakrishna A., Giridhar P., Sankar K.U., Ravishankar G.A. Melatonin and serotonin profiles in beans of Coffea species. J. Pineal Res. 2012;52:470–476. doi: 10.1111/j.1600-079X.2011.00964.x[↩]
- Badria F. Melatonin, serotonin, and tryptamine in some egyptian food and medicinal plants. J. Med. Food. 2002;5:153–157. doi: 10.1089/10966200260398189[↩]
- Collier H.O., Chesher G.B. Identification of 5-hydroxytryptamine in the sting of the nettle (urtica dioica) Br. J. Pharmacol. Chemother. 1956;11:186–189. doi: 10.1111/j.1476-5381.1956.tb01051.x[↩]
- Carnevale G., Di Viesti V., Zavatti M., Zanoli P. Anxiolytic-like effect of Griffonia simplicifolia Baill. seed extract in rats. Phytomedicine. 2011;18:848–851. doi: 10.1016/j.phymed.2011.01.016[↩]
- Parkinson’s Disease. https://www.nia.nih.gov/health/parkinsons-disease[↩]