Contents
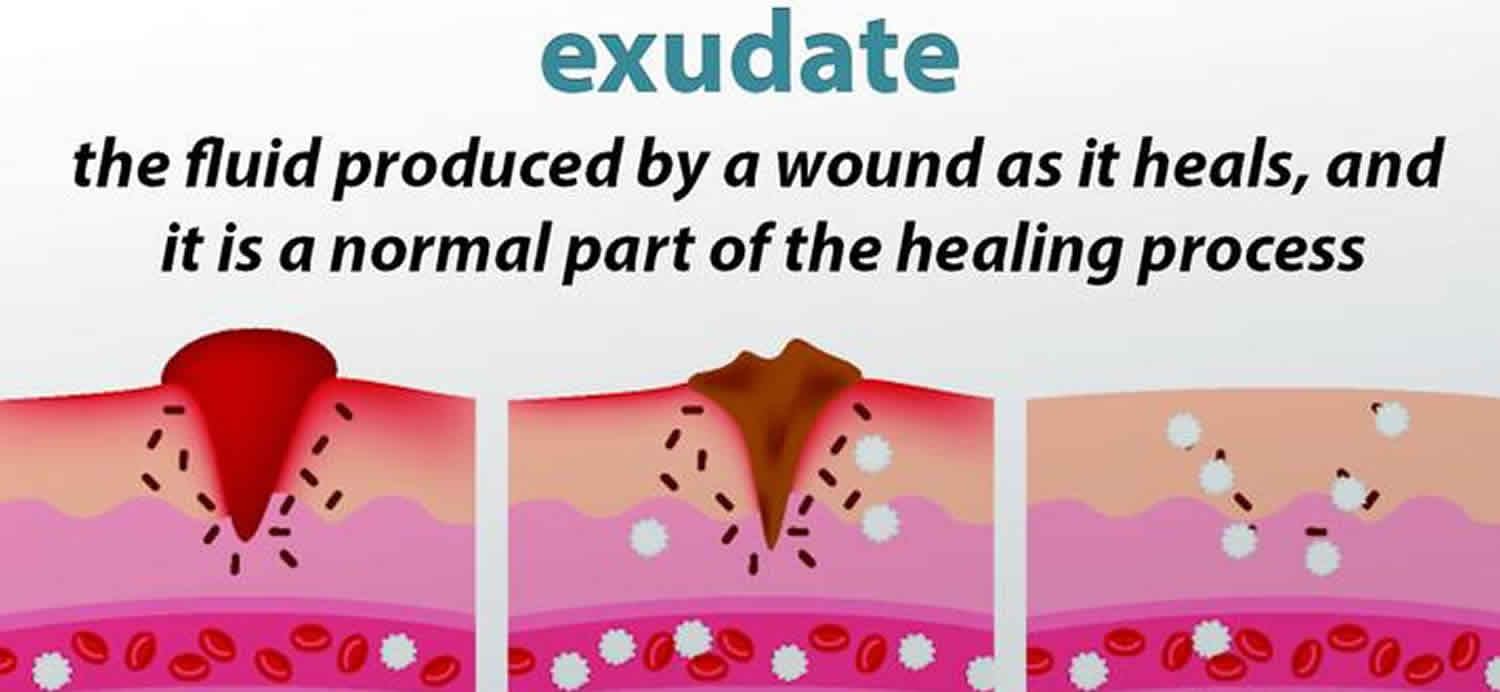
What is exudate
Exudate is fluid that leaks out of blood vessels into a body cavity or nearby tissues as a result of injury or inflammation. Exudate is made of cells, proteins, and solid materials. Exudate may ooze from cuts or from areas of infection or inflammation. An alteration of the local inflammatory factors that precipitate a fluid accumulation represents an exudative effusion. Exudate has a higher than normal protein content and may be cloudy due to increased numbers of white blood cells and is also called pus.
Transudate vs Exudate
Transudate is fluid that leaks out of blood vessels into a body cavity or nearby tissues, due to an imbalance between the pressure within blood vessels (which drives fluid out) and the amount of protein in blood (which keeps fluid in). Transudate is a clear fluid with low protein concentration and a limited number of white blood cells. An imbalance between the hydrostatic and oncotic pressure within the capillaries causes a transudate effusion.
Light’s criteria 1 can be used to determine if the fluid is an exudate versus transudate. To be an exudate, the effusion must have at least one of the following 2:
- Pleural fluid/serum lactate dehydrogenase (LDH) ratio > 0.6, or lactate dehydrogenase (LDH) > 2/3 the upper limit of normal, or
- Pleural fluid/serum total protein ratio > 0.5.
There is no ideal biochemical marker that allows complete discrimination between transudates and exudates 3. Light’s criteria have high sensitivity but lower specificity and therefore do not have a high diagnostic efficiency 4.
Key facts
- The combination of fluid lactate dehydrogenase (LDH) measurements and fluid to serum total protein ratios is useful in differentiating exudates from transudates 3
- Using this combination could help in patient management and avoid unnecessary testing
Transudate: approximately 90% are from congestive heart failure, and the remaining 10% are caused by hepato-hydrothorax, nephrotic syndrome, peritoneal dialysis, myxedema, atelectasis, constrictive pericarditis, SVC syndrome, and hypoalbuminemia.
Exudate: Low glucose (glucose < 60 or ratio fluid/serum < 0.5), complicated parapneumonic effusion versus empyema, tuberculosis (TB), malignancy, vasculitis (i.e., lupus), esophageal rupture (Tip: rheumatoid or empyema glucose often < 10).
Low pH (pH < 7.2): Complicated parapneumonic effusion versus empyema, tuberculosis, malignancy, vasculitis (i.e., lupus), esophageal rupture, hemothorax, or parasitic (Tip: esophageal rupture often < 6).
Bloody (RBC > 100K): Due to trauma, malignancy, pulmonary embolism, infarction, tuberculosis (TB), or post-operative.
Cell Count high Lymphocytes: As in lymphoma, malignancy not otherwise specified, tuberculosis (TB), fungal, or post-operative.
Types of exudate
Fibrinous exudate – cloudy and thin with strands of fibrin
Serous exudate – a clear, amber, thin and watery plasma. It’s normal during the inflammatory stage of wound healing, and smaller amounts are considered normal.
Sanguineous exudate – a fresh bleeding, seen in deep partial- and full-thickness wounds. A small amount is normal during the inflammatory stage.
Serosanguineous exudate – thin, watery and pale red to pink in color.
Seropurulent exudate– thin, watery, cloudy and yellow to tan in color.
Purulent exudate – a thick and opaque exudate that is tan, yellow, green or brown in color. It’s never normal in a wound bed, and is often associated with infection or high bacteria levels.
Hemopurulent exudate – reddish, viscous and milky
Choice of wound dressing
The most suitable dressing depends on the type of wound.
- Necrotic wounds have a dry black eschar composed of dead epidermis
- Sloughy wounds contain yellow viscous adherent slough
- Granulating wounds contain deep red vascularised granulation tissue
- Epithelialising wounds have a pink margin to the wound or isolated pink islands on the surface
- Infected wounds.
It also depends on the location and size of the wound. Some dressings are easier to use and remove than others. Modern dressings are relatively hypoallergenic and non-adherent but sensitisation may occur to iodine, antibiotics, rubber, adhesives and preservatives. Tape cannot be applied if the skin is treated with emollient or topical steroid creams. Cost and availability must also be considered.
Necrotic wounds
The aim is to rehydrate the dry scab so that it will separate off. Options are: Wet dressings using saline or hypochlorite (Eusol). Hydrogel covered by perforated plastic film absorbent dressing (Melolin or Telfa) or vapour permeable film. Hydrocolloid dressing.
Sloughy wounds
These need debriding to remove the abnormal matrix of fibrin, exudate, inflammatory cells and bacteria. This can be done by surgical debridement or by an agent that soaks up debris and forms a moist gel. Options are:
- Polysaccharide dressing as beads or paste.
- Hydrocolloid dressing if wound less exudative.
- Alginate dressing.
- Enzymes.
Granulating wounds
Granulation tissue is a highly vascular matrix collagen and proteoglycans.
- Cavity wounds are packed with alginate fibre ribbon, silicone foam dressing or foam chips
- Shallow but heavily exuding ulcers are dressed with alginate dressings or hydrophilic foam product
- Less exudative ulcers are dressed with hydrocolloid or thin foam dressing
Epithelializing wounds
Superficial wounds that exude fluid (burns and donor sites):
- Paraffin gauze covered with gauze and cotton tissue (Gamgee)
- Alginate
- Hydrocolloid.
Dry superficial wounds
- Hydrocolloid
- Film dressing
- Perforated plastic film dressing
- Knitted viscose non-adherent dressings.
Infected wounds
Infected wounds need to be covered because they may have an unpleasant odour, and to prevent the spread of the organisms, particularly if they are resistant to standard antibiotics. Several dressings include antibacterial agents. Their use is controversial.
- Framycetin
- Fusidic acid
- Chlorhexidine
- Povidone iodine
Bioengineered skin substitutes
Skin autografts are commonly used to cover acute surgical wounds and chronic ulcers. However, harvesting skin grafts creates another wound that must heal, and suitable skin is unavailable in some cases such as extensive thermal burns. Sterilised cadaver allografts provide temporary wound dressings but eventually slough off. There has been intensive research and development in recent years to provide a satisfactory substitute for healthy skin. Replacement of dermal matrix and epidermis is required.
Cultured keratinocyte autografts can provide permanent coverage of large area from a skin biopsy. However, 3 weeks are needed for graft cultivation.
Keratinocyte allografts cultured from neonatal foreskins are available immediately. They can be cryopreserved and banked, but are not currently commercially available.
Applying a substitute dermal matrix has been shown to improve the likelihood that cultured epidermal cells (or an autologous split skin graft) will take. Several immunologically inert systems are now under investigation for management of refractory venous and diabetic ulcers.
Dressings for acute wounds
Sterile dressings should be applied to acute wounds, including those due to skin surgery, for the following reasons.
- Keep the wound clean and prevent contamination by foreign bodies.
- Apply pressure to encourage hemostasis and prevent hematoma
- Immobilize the wound
- Reduce the risk of bacterial infection.
- Absorb drainage of blood or wound exudate
- Occlusion provides thermal insulation.
- Allow for oxygen entry and water vapor escape
- To cover the wound for cosmetic reasons.
These goals can be accomplished with a layered dressing comprised of a bottom, nonadherent contact layer, a middle layer that can absorb wound exudate and exert pressure, and an outer covering of tape or a rolled bandage. Sterile dressings come in various sizes from simple gauze pads to adhesive bandages.
If a wound is discharging fluid (serum), the dressing should be changed often to minimize bacterial growth. After drainage has stopped, there may be no need for a dressing.
Ointments such as petroleum jelly may be applied to limit surface bacterial growth and prevent the dressing from sticking to the wound. Topical antibiotics containing single or multiple antibiotics such as bacitracin, neomycin, or polymyxin B may be useful in a contaminated wound but should be avoided in clean wounds because there is an increasing rate of bacterial resistance to these agents.
Wounds may be cleansed with water, normal saline or with hydrogen peroxide several times daily before the reapplication of ointment.
Use of topical silicone gel sheeting or polyurethane self-adhesive patches after routine dermatologic surgery minimizes the risk of hypertrophic scars and keloids in patients at risk for those lesions.
Dressings for chronic wounds
The principles outlined for acute wounds remain true for chronic wounds including leg ulcers or surgical wounds healing by secondary intention.
In a full-thickness wound, the dermis must be recreated before re-epithelialization can begin. These wounds heal from the base as well as from the edges so the development of some fibrinous exudate in the wound bed is a positive sign.
Occlusion of chronic wounds:
- Allows macrophages and fibroblasts to enter the wound
- Promotes autolysis mediated by enzymes released from leukocytes.
- Favors cell proliferation because of low pH and hypoxia.
- Enhances growth factors and cytokines within wound fluid.
The occlusive dressings may be divided into five categories:
- Hydrogels
- Hydrocolloid
- Foams
- Films
- Alginates.
Hydrogels are composed primarily of water, fixed in a cross-linked polymer (sodium carboxymethylcellullose, or starch). They may be hydrated or in a dehydrated state requiring moistening with water or saline. They are used to rehydrate dried-out necrotic eschar. Hydrogels are also useful for exudative wounds because they have high absorptive capacity and are nonadherent. They cool the wound and can provide excellent pain relief. They are also useful for partial thickness wounds from resurfacing procedures and skin graft donor sites.
The hydrogel should be covered by an absorbent layer, made of gauze or cotton, and an outer layer of tape, netting or roll bandage.
Hydrocolloid dressings are a mixture of a hydrophilic base and adhesive, often with an outer covering of polyurethane. They adhere directly to the wound and do not usually require a secondary dressing to keep them in place. In addition, they absorb mild to moderate wound exudate so they can be worn for three to seven days without changing.
Hydrophilic foam dressings are permeable to oxygen and water vapour. They usually have a hydrophobic backing that provides occlusion and some have an adhesive surface, which makes application easier. They can absorb only limited amounts of wound exudate so may need to be changed every two to three days or even more frequently during early wound healing when exudation is greatest.
Foam dressings are ideally suited for superficial and dry wounds eg after ablative resurfacing procedures and chronic ulcers since they provide padding that can relieve pressure over bony prominences.
Thin transparent film dressings are not very absorptive, so they are not useful for wounds with significant exudate. They can be used to keep other dressings in place, including as top layer of an acute surgical wound dressing. They are often used to cover sites of IV insertion, superficial abrasions and as temporary dressings e.g. over local anaesthetic cream prior to venepuncture.
Alginates are highly absorbent and are indicated when a wound is very exudative. They release calcium ions, which help haemostasis so are useful applied to a surgical wound in a patient with excessive bleeding. In chronic wounds, the exudate combines with the alginate gel to form green or yellowish goo. The alginates are nonadherent unless the wound dries out. They can be soaked off to avoid unnecessarily debriding the wound.
Honey
Honey has been used as a traditional remedy for burns and wounds, and more recently several studies have demonstrated that it has antibacterial activity. Honey can clear infection from cutaneous wounds and improve healing.
Proposed mechanisms include:
- Physicochemical properties (e.g., osmotic effects and pH)
- Antiinflammatory activity stimulates immune responses
- Hydrogen peroxide concentration.
- Light RW, Macgreggor MI, Luchsinger PC, et al. Pleural effusions: the diagnostic separation of transudates and exudates. Ann Intern Med 1972;77:508–13.[↩]
- Wiederhold BD, O’Rourke MC. Thoracentesis. [Updated 2018 Oct 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441866[↩]
- Paramothayan NS, Barron J. New criteria for the differentiation between transudates and exudates. J Clin Pathol. 2002;55(1):69-71. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1769575/[↩][↩]
- Romero S, Martinez A, Hernandez L, et al. Light’s criteria revisited: consistency and comparison with new proposed alternative criteria for separating pleural transudates from exudates. Respiration 2000;67:18–23.[↩]