What is gingivostomatitis
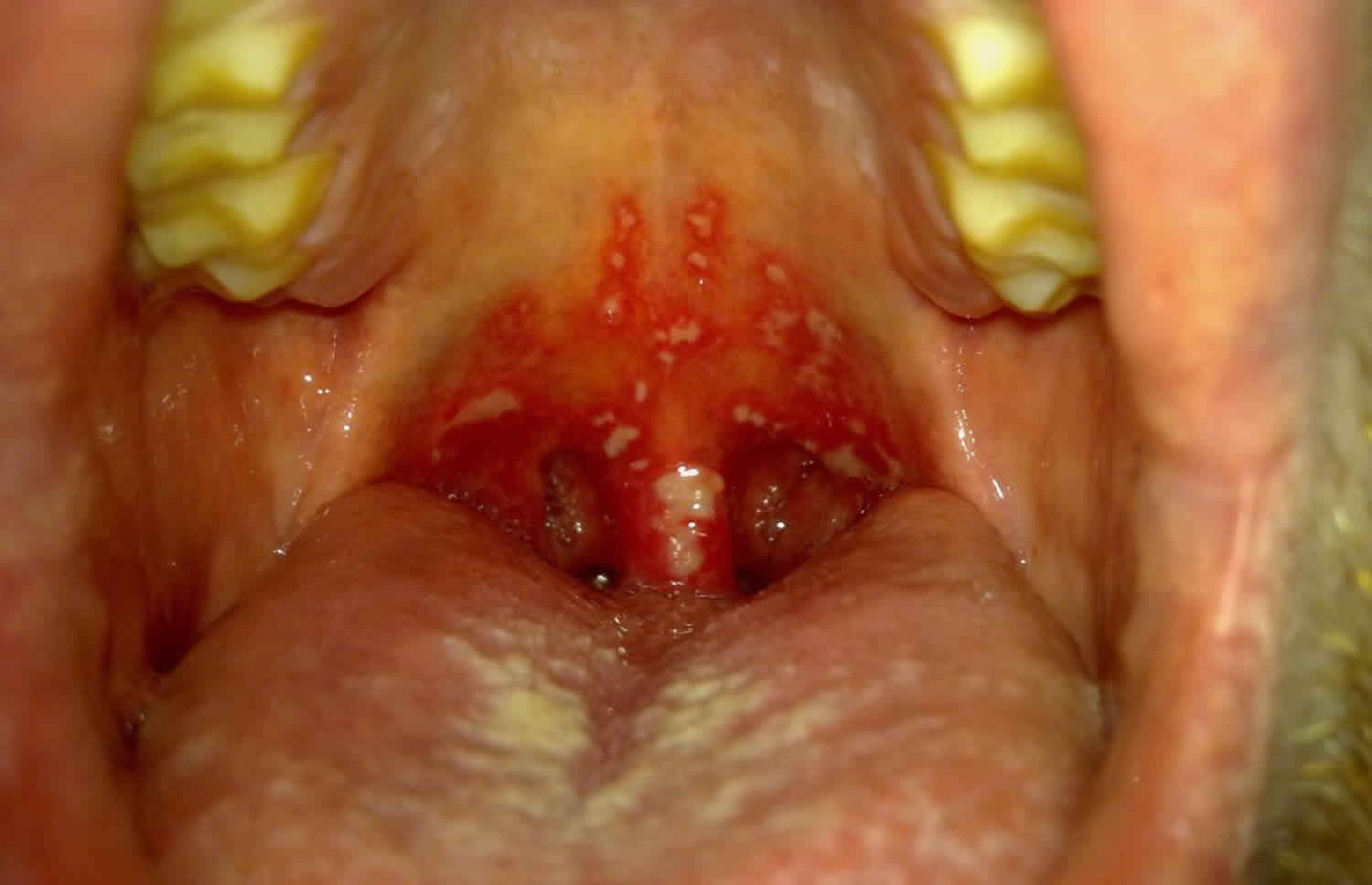
Gingivostomatitis is a highly contagious infection of your mouth and gums that leads to swelling and sores. These sores can develop on the tongue, under the tongue, and on the cheeks inside the mouth, as well as on the lips and gums. Gingivostomatitis main symptoms include pain, swollen gums, blisters, and sores.
Gingivostomatitis may be due to a virus or bacteria.
Gingivostomatitis may disguise other, more serious mouth ulcers.
See your doctor if:
- You have mouth sores and fever or other signs of illness
- Mouth sores get worse or do not respond to treatment within 3 weeks
Gingivostomatitis causes
Gingivostomatitis is common among children. Gingivostomatitis may occur after infection with the herpes simplex virus type 1 (HSV-1) where it is called herpetic stomatitis or herpetic gingivostomatitis. The herpes simplex virus type 1 (HSV-1) also causes cold sores.
Gingivostomatitis may also occur after infection with a coxsackie virus.
Gingivostomatitis may occur in people with poor oral hygiene.
It is important to note that many different factors can cause the irritation and sores in the mouth that are characteristic of gingivostomatitis.
The general medical term for inflammation of the mouth and lips is stomatitis. Factors that can cause stomatitis include:
- Herpes viruses
- Enteroviruses, such as the coxsackie virus
- Bacteria
- Allergies
- Exposure to irritating chemicals or other substances
- Dry mouth / xerostomia
- Candida albicans infection
- Trauma including surgery
- Smoking tobacco
- Toxicity of chemotherapy drugs – including methotrexate used for psoriasis and other skin disorders
- Therapeutic radiation e.g. for oral cancer
Some of the causes of gingivostomatitis are listed in the table below.
Table 1. Causes of gingivostomatitis
| Bacterial infection |
|
| Fungal infection |
|
| Viral infection |
|
| Systemic disorder |
|
| Drugs |
|
| Physical irritation |
|
| Contact stomatitis |
|
| Immunobullous disesase |
|
| Other |
|
Gingivostomatitis symptoms
The symptoms can be mild or severe and may include:
- Bad breath
- Fever
- General discomfort, uneasiness, or ill feeling (malaise)
- Sores or ulcers on the inside of your cheeks or gums
- Very sore mouth with no desire to eat or drink
- Drooling, especially in children
- Bad breath
- Red patches
- Blisters
- Peeling
- Swelling
- Oral dysesthesia (numbness)
- Burning mouth syndrome – soreness despite normal appearance
Some cases of gingivostomatitis may be subclinical, which means that the symptoms are not severe, or easy to identify and diagnose.
In other cases, some individuals may go through a period of feeling feverish and having general malaise before the sores develop.
Swollen gums and sores in the mouth make eating and drinking uncomfortable. This can cause children to refuse food and drinks.
One study found that 89 percent of children with gingivostomatitis drank less than usual. To prevent dehydration and poor nutrition, adults should monitor children’s consumption and ensure they are getting enough fluids.
Adopting a diet consisting of soft foods and avoiding citrus or carbonated beverages can help. In some cases, a person can apply numbing medication to provide relief at mealtimes.
Gingivostomatitis diagnosis
Your health care provider will check your mouth for small ulcers. These sores are similar to mouth ulcers caused by other conditions. Cough, fever, or muscle aches may indicate other conditions.
Relevant investigations depend on the likely cause of gingivostomatitis and whether it is accompanied by other symptoms internally or skin rashes.
Investigations may include:
- Bacterial swabs
- Viral swabs
- Tissue scrapings for mycology
- Biopsy for histology and direct immunofluorescence
- Blood tests
- Patch tests to identify contact allergy
Most of the time, no special tests are needed to diagnose gingivostomatitis. However, your doctor may take a small piece of tissue from the sore to check for a viral or bacterial infection. This is called a culture. A biopsy may be done to rule out other types of mouth ulcers.
Gingivostomatitis treatment
The goal of gingivostomatitis treatment is to reduce symptoms.
Treatment for gingivostomatitis depends on the cause. If gingivostomatitis is due to allergy to a medication, the medication must be promptly stopped. However, it may be necessary to continue a causative medication when stomatitis arises as an expected adverse reaction to chemotherapy.
Infections may require specific treatment such as antibiotics for streptococcal pharyngitis, topical antifungal or oral antifungal agent for candida infection.
Nutritional deficiencies should be identified and corrected, for example, folic acid can reduce methotrexate-induced stomatitis.
Immunobullous diseases may be treated with systemic corticosteroids or other immunosuppressive treatments.
Symptomatic treatment may include:
- Antiseptic mouthwash
- Protective pastes
- Local anaesthetic mouthwash or spray
- Oral analgesics (pain killers)
- Topical corticosteroids.
Gingivostomatitis home treatment:
- Practice good oral hygiene. Brush your gums well to reduce the risk of getting another infection.
- Use mouth rinses that reduce pain if your provider recommends them.
- Rinse your mouth with salt water (one-half teaspoon or 3 grams of salt in 1 cup or 240 milliliters of water) or mouthwashes with hydrogen peroxide or Xylocaine to ease discomfort.
- Eat a healthy diet. Soft, bland (non-spicy) foods may reduce discomfort during eating.
You may need to take antibiotics.
You may need to have the infected tissue removed by the dentist (called debridement).
Gingivostomatitis prognosis
Gingivostomatitis infections range from mild to severe and painful. The sores often get better in 2 or 3 weeks with or without treatment. Treatment may reduce discomfort and speed healing.
Herpetic gingivostomatitis
Herpetic gingivostomatitis is a herpes simplex virus or oral herpes infection of the mouth that causes sores and ulcers. These mouth ulcers are not the same as canker sores, which are not caused by a virus. Young children commonly get primary herpetic gingivostomatitis, in 90% of cases caused by herpes simplex virus type 1 (HSV-1) when they are first exposed to herpes simplex virus (HSV) 1. The first outbreak also called primary herpetic gingivostomatitis, is usually the most severe and usually seen before 6 years of age 2. Epidemiologically, there are two peaks with respect to the age at which primary herpetic gingivostomatitis occurs. The first peak occurs in children aged between 6 months and 5 years, and the second peak occurs in young adults in their early 20s 3. In rare cases, primary herpetic gingivostomatitis can occur in neonates, in adults, and even in the elderly 4. Geographical location and socio-economic status also influence the incidence of herpes simplex virus type 1 (HSV-1) infections. Hence, individuals in developing countries with a lower socio-economic status become seropositive for HSV-1 at an earlier age than their counterparts in developed countries 5.
Herpes simplex virus (HSV) can easily be spread from one child to another. Prodromal symptoms, such as fever, anorexia, irritability, malaise and headache, may occur in advance of primary herpetic gingivostomatitis 2. The primary herpetic gingivostomatitis presents as numerous pin-head vesicles, which rupture rapidly to form painful irregular ulcerations covered by yellow–grey membranes. Sub-mandibular lymphadenitis, halitosis and refusal to drink are usual associated findings 2. Following resolution of the lesions, the herpes simplex virus travels through the nerve endings to the nerve cells serving the affected area, whereupon it enters a latent state. When the host becomes stressed, the herpes simplex virus replicates and migrates in skin, mucosae and, in rare instances, the central nervous system. A range of morbidities, or even mortality, may then occur, i.e., recurrent HSV infections, which are directly or indirectly associated with primary herpetic gingivostomatitis. These pathological entities range from the innocuous herpes labialis to life-threatening meningoencephalitis 2.
If you or another adult in the family has a cold sore, it could have spread to your child and caused herpetic gingivostomatitis. More likely, you won’t know how your child became infected.
While most children will be asymptomatic, diagnosis of children with symptoms is made based on clinical presentation of red gums, mucosal hemorrhages, and clusters of small erupted vesicles throughout the mouth.
Herpetic gingivostomatitis is highly contagious and complications range from indolent cold sores to dehydration 6 and even life-threatening encephalitis 7. Among 61 children 1 to 6 years of age from a study in Israel, 89% drank less than normal, and 2 of 36 patients were unable to drink 6.
Symptomatic relief primarily involves pain management and oral fluids to prevent dehydration until the viral infection subsides. In a chart review in a US children’s hospital, 48 patients 8 months to 12 years of age were treated with fluids and analgesics; 35 of them were also given a mixture of antacid and diphenhydramine and 7 were treated with viscous lidocaine 8. Outcomes were not reported.
Acyclovir is a well established antiviral drug used effectively for the treatment of herpes simplex infections, chickenpox (shortened fever time) 9 and shingles 10. Acyclovir is also used frequently for children with immunodeficiency.
See your doctor if your child develops a fever followed by a sore mouth, and your child stops eating and drinking. Your child can quickly become dehydrated.
If the herpes infection spreads to the eye, it is an emergency and can lead to blindness. Call your doctor right away.
Herpetic gingivostomatitis prevention
About 90% of the population carries herpes simplex virus (HSV). There’s little you can do to prevent your child from picking up the virus sometime during childhood.
Your child should avoid all close contact with people who have cold sores. So if you get a cold sore, explain why you can’t kiss your child until the sore is gone. Your child should also avoid other children with herpetic stomatitis.
If your child has herpetic stomatitis, avoid spreading the virus to other children.
While your child has symptoms:
- Have your child wash his/her hands often.
- Keep toys clean and don’t share them with other children.
- Don’t allow children to share dishes, cups, or eating utensils.
- Don’t let your child kiss other children.
Herpetic gingivostomatitis symptoms
Symptoms may include:
- Blisters in the mouth, often on the tongue, cheeks, roof of the mouth, gums, and on the border between the inside of the lip and the skin next to it
- After blisters pop, they form ulcers in the mouth, often on the tongue or cheeks
- Difficulty swallowing
- Drooling
- Fever, often as high as 104 °F (40 °C), which may occur 1 to 2 days before blisters and ulcers appear
- Irritability
- Mouth pain
- Swollen gums
Symptoms may be so uncomfortable that your child doesn’t want to eat or drink.
Herpetic gingivostomatitis diagnosis
Your child’s health care provider can most often diagnose this condition by looking at your child’s mouth sores.
Sometimes, special laboratory tests can help confirm the diagnosis.
Herpetic gingivostomatitis treatment
Your child’s doctor may prescribe:
- Acyclovir, a medicine your child takes that fights the virus causing the infection
- Numbing medicine (viscous lidocaine), which you can apply to your child’s mouth to ease severe pain
Use lidocaine with care, because it can numb all feeling in your child’s mouth. This can make it hard for your child to swallow, and may lead to burns in the mouth or throat from eating hot foods, or cause choking.
There are several things you can do at home to help your child feel better:
- Give your child cool, noncarbonated, nonacidic drinks, such as water, milk shakes, or diluted apple juice. Dehydration can occur quickly in children, so make sure your child is getting enough fluids.
- Offer cool, bland, easy-to-swallow foods such as frozen pops, ice cream, mashed potatoes, gelatin, or applesauce.
- Give your child acetaminophen or ibuprofen for pain. (Never give aspirin to a child under age 2. It can cause Reye’s syndrome, a rare, but serious illness.)
- Bad breath and a coated tongue are common side effects. Gently brush your child’s teeth every day.
- Make sure your child gets plenty of sleep and rests as much as possible.
Herpetic gingivostomatitis prognosis
Your child should recover completely within 10 days without medical treatment. Acyclovir may speed up your child’s recovery.
Your child will have the herpes virus for life. In most people, the virus stays inactive in their body. If the virus wakes up again, it most often causes a cold sore on the mouth. Sometimes, it can affect the inside of the mouth, but it won’t be as severe as the first episode.
Herpetic gingivostomatitis possible complications
The complications of primary herpetic gingivostomatitis can range from cold sores to life-threatening encephalitis 2. The most common cause of morbidity following primary herpetic gingivostomatitis is dehydration. Amir et al. 11 found that 89% of patients drank less than normal, and that two of 36 patients were unable to drink. In severe cases, hospitalization and parenteral fluid intake are recommended. Bacteremia caused by the Gram-negative bacillus Kingella kingae has been observed 12, but this complication subsided uneventfully after the administration of antibiotics.
An innocuous morbidity of primary herpetic gingivostomatitis is herpes labialis (also known as ‘cold sores’ or ‘fever blister’). This represents the most common manifestation of HSV reactivation in trigeminal ganglia, with an incidence among adults of 20–40% 13. Two or three recurrences annually are normal, but as many as 12 may occur. herpes simplex virus type 1 (HSV-1) infections recur more often than herpes simplex virus type 2 (HSV-2) infections. The vermilion border and adjacent skin of the lips are the sites affected most frequently, although the skin of the nose, chin or cheek may also be involved. Pain, tingling, burning sensation, itching, fever (fever blisters) or upper respiratory tract infection (cold sores) usually precede the onset of the disease 14. Herpes labialis is characterized by multiple small, erythematous papules that develop and form clusters of fluid-filled vesicles that rupture within 2 days. The total area involved is usually less than 100 mm2, and the lesions progress to being pustular or ulcerative, with crusting within 3–4 days 15. Pain is intense at the outset, but resolves over 4–5 days. Shedding of virus from lesions continues, with progressive healing over 2–3 days. Healing is rapid and is complete within 10 days. Remission of herpes labialis can occur; inciting factors are fever, stress and exposure to UV light 15.
‘Herpetic geometric glossitis’, characterized by linear fissures on the dorsum of the tongue, with branching dendritic lesions, represents an HSV-1 infection in immunocompromised patients 16, whereas recurrent herpetic stomatitis presents in immunocompetent individuals. The latter differs from primary herpetic gingivostomatitis in that it has a more confined enanthema. Tabaee et al. 17 described an oral recurrent HSV infection presenting as an immense tongue mass in a patient who underwent cardiac transplantation. Paradoxically, this lesion was first encountered 10 months after transplantation, although most recurrent oral HSV infections occur within the first month post-transplant 18. The lesion was initially considered to be a squamous carcinoma, so a 0.5-cm free margin excision was performed. Later, following immunostaining, the final diagnosis of HSV infection was established 17.
An even more unusual clinical entity is a recrudescent HSV infection identical to primary herpetic gingivostomatitis. Use of laboratory screening tests is mandatory in such cases in order to make the differential diagnosis 19.
Finally, there is a single report 20 postulating that recurrent HSV infection affecting the oral cavity may even manifest as a dry socket, since HSV was isolated from the socket. However, there are reservations concerning the acceptance of this pathology.
A less common presentation of HSV is herpetic whitlow (herpetic paronychia). This may occur as a result of auto-inoculation in children with orofacial herpes (i.e., children sucking their hands), and in adults in association with genital herpes 21. In the past, before the use of protective gloves, dental personnel could come into contact with HSV during the treatment of patients suffering from HSV infection. Whitlow is a noxious pathology characterized by prodromal symptoms, namely pain and burning, during the 2–20-day incubation period. Herpetic whitlow usually manifests with local swelling, erythema, and one or more small, tender vesicles. Fever and malaise may be present, especially in infants. The lesions typically contain clear fluid in the initial stages, but the fluid can become cloudy after a week by virtue of the presence of white blood cells. In contrast, bacterial paronychia presents with suppuration from the onset of the disease. The site affected most commonly is the digital pulp space, but infections of the nail folds or lateral aspects of the fingers can also be encountered. The disease subsides completely within 3 weeks 22.
Another dermatosis caused by HSV is herpes gladiatorum or ‘scrumpox’. This usually affects individuals participating in contact sports, e.g., wrestling or rugby football, or parents who have kissed areas of dermatological injury in children 13.
Eczema herpeticum (also known as Kaposi’s varicelliform eruption) is a skin infection affecting children and adults, in which HSV is implicated. Habif 23 considers that eczema herpeticum is an association of atopic dermatitis with HSV infection. The disease is most common in areas of active or recently healed atopic dermatitis, particularly on the face, but it can also develop in areas with pre-existing dermatoses, such as are seen in atopic dermatitis, Darier’s disease, mycosis fungoides, pemphigus folliaceous and Sezary’s syndrome, and even on normal skin. In one-third of paediatric patients with eczema herpeticum, there is a history of herpes labialis in a parent. The disease is characterised by the eruption of numerous vesicles, which gradually become pustular, and finally are rendered umbilicated. New groups of vesicles may appear during the following weeks. High fever and adenopathy become apparent 2–3 days after the eruption of vesicles. The fever resolves in 4–5 days in uncomplicated cases, and the lesions evolve in the typical manner. The prognosis is usually good, but the patient may succumb when viraemia occurs along with visceral involvement 23.
Erythema multiform is another dermatosis in which HSV is implicated. Indeed, HSV may precede erythema multiform, and post-herpetic erythema multiform is thought to be the sequel of perivascular immune complex deposition of immunoglobulin, herpes virus antigen and complement 24.
An even more severe complication of primary herpetic gingivostomatitis is ocular involvement. This can be caused by auto-inoculation from primary herpetic gingivostomatitis or herpes labialis, or via the nerve route 25. HSV infection of the eye is the most frequent cause of corneal blindness in the USA 26. Herpetic keratoconjunctivitis is associated with acute onset of pain, blurring of vision, characteristic dendritic lesions of the cornea, injection, chemosis, photophobia, lacrimation and eyelid oedema. Less commonly, advanced disease can result in ‘geographical’ or ‘amoeboid’ ulcer of the cornea. The visual acuity declines in the presence of the ulcers after several bouts of infection and, with progressive stromal involvement, opacification of the cornea may occur. Repeated individual attacks may take place for several weeks or even months. Progressive disease can result in visual loss or even rupture of the globe 25.
Herpetic stromal keratitis caused by HSV appears to be related to T-cell-dependent destruction of deep corneal tissue. The UL6 viral protein mimics the corneal antigens, so an autoreaction with corneal antigens targeting T-cells has been postulated to be a factor in this infection 27. Recently, Lundberg et al. 28 proposed a model for herpetic stromal keratitis pathogenesis in which the unmethylated CpG dinucleotides, released by degenerate stromal cells infected previously by HSV, bind to Toll-like receptor 9 of macrophages, which in turn orchestrate the Th1 response with release of cytokines and recruitment of autoreactive T-cells 28.
Herpetic oesophagitis and herpetic infection of the lower respiratory tract may result from direct extension of oropharyngeal infection, including primary herpetic gingivostomatitis 29. The former may also represent the consequence of de-novo reactivation of HSV and propagation to the oesophageal mucosa via the vagus nerve. HSV esophagitis may manifest even in immunocompetent patients, but it is more likely to occur in patients who have undergone kidney or liver transplantation, or in patients suffering from AIDS. The predominant symptoms of HSV oesophagitis are odynophagia, dysphagia, substernal pain and weight loss, while bleeding or even stricture formation may complicate the disease 30. Clinically, there are multiple oval ulcerations on an erythematous base, which may or may not have a patchy white pseudomembrane. The distal oesophagus is involved most commonly, although disseminated disease may occur when the immune system is deranged 31.
HSV pneumonitis is uncommon unless an individual is immunocompromised, and may result from extension of herpetic tracheobronchitis into lung parenchyma, whereupon focal necrotising pneumonitis usually ensues. The mortality rate in immunosuppressed patients is high (>80%) 29. Bogger-Goren 32 reported a case of acute epiglottitis in a child aged 16 months that was caused by HSV, and involvement of the peripheral nervous system or the spinal cord may also be complications resulting from primary herpetic gingivostomatitis. Nasatzky and Katz 33 reported a case of Bell’s palsy associated with HSV gingivostomatitis. It has been postulated that Bell’s palsy results from the replication of HSV in geniculate ganglia, and that axonal spread and multiplication of the reactivated virus leads to inflammation, demyelination and palsy, which usually resolve within 6–8 weeks, although sequelae may occur in some patients. It has also been suggested that the anatomical structure of the Fallopian canal may be responsible for nerve compression secondary to oedema, which can lead finally to nerve degeneration and severe facial paralysis.
Galanakis et al. 34 reported a case of transverse myelitis associated with primary herpetic gingivostomatitis in which the patient exhibited the typical signs and symptoms of transverse myelitis, such as acute weakness, acute pain in the lower extremities, lower back and abdomen, weak leg muscles and bladder dysfunction, but retained deep sensation. The patient’s history revealed that he had suffered from herpetic gingivostomatitis, and lumbar puncture, magnetic resonance imaging and serology were all consistent with transverse myelitis caused by HSV-1.
Herpetic meningoencephalitis is the most fatal complication of HSV infection. In general, herpetic encephalitis (HSE) is attributed mainly to HSV-1, whereas meningitis is ascribed to HSV-2 35. Herpetic encephalitis is considered to be the most common cause of sporadic, fatal encephalitis in the USA, and probably worldwide, as it accounts for 10–20% of all cases of viral encephalitis [97, 98], either primary or secondary. There are three patterns of viral propagation to brain parenchyma: (1) exogenously acquired virus entering the CNS via the olfactory epithelium and olfactory bulb; (2) reactivation of latent HSV in ganglia, followed by spread via trigeminal and autonomous nerve roots; and (3) blood-borne spread, especially in neonates or immunocompromised individuals. However, only Ito et al. 36 have reported herpetic encephalitis associated directly with primary herpetic gingivostomatitis. The clinical signs and symptoms of herpetic encephalitis depend on the location of the lesion. Typically, herpetic encephalitis produces focal hemorrhagic necrosis of temporal and frontal lobe structures, including limbic mesocortices, amygdala and hippocampus, although the involvement of other sites cannot be excluded 37. The clinical hallmark of herpetic encephalitis is the triad of fever, headache and altered consciousness 38. Concomitant signs and symptoms include confusion, lethargy, seizures and mental aberrations, but other symptoms may also become apparent, depending on the location of the lesion 39. The presence of neurological symptoms, e.g., altered sensorium or other focal neurological findings, distinguish herpetic encephalitis from HSV meningitis 38. Mortality rates reach 60–70% and, even after adequate medication, permanent neurological deficits will remain, with only 2.5% of surviving patients recovering normal neurological function 37.
In terms of pathogenesis, it should be emphasised that, although herpetic encephalitis has been associated with necrotic cell death resulting from virus replication and inflammatory changes/cerebral oedema secondary to the virus-induced immune response, the role of virus-induced nerve cell apoptosis should not be overlooked 37. Indeed, Perkins et al. 37 demonstrated HSV-1-induced apoptosis mediated by c-jun N-terminal kinase. Likewise, Anglen et al. 40 showed that CD8+ T-cells, which are probably the most efficient cell population active against HSV, may enhance or eliminate brain injury when they infiltrate brain tissues before or after HSV infection, respectively.
In contrast, meningitis is ascribed generally to HSV-2. Headache, asthenia, high fever, neck stiffness, nausea/vomiting, photophobia, muscle pain and radiculalgia are some of the symptoms indicative of meningitis 41. Mommeja-Marin et al. 41 reported no cases of visceral involvement, and considered that meningitis did not result from blood-borne spread ofviraemia, but probably from spread through the nerve tract, starting from sacral ganglia. Cerebrospinal fluid analysis is suggestive of viral meningitis if high levels of white blood cells, especially lymphocytes (lymphocytic pleocytosis), are detected, the protein level is normal or slightly elevated, and the glucose level is normal. PCR is necessary to establish the final diagnosis.
- Goldman RD. Acyclovir for herpetic gingivostomatitis in children. Can Fam Physician. 2016;62(5):403-4. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4865337[↩]
- Herpes simplex virus infection, with particular reference to the progression and complications of primary herpetic gingivostomatitis. Kolokotronis A, Doumas S. Clin Microbiol Infect. 2006 Mar; 12(3):202-11. https://www.clinicalmicrobiologyandinfection.com/article/S1198-743X(14)63911-2/fulltext[↩][↩][↩][↩][↩]
- Ajar, AH and Chauvin, PJ. Acute herpetic gingivostomatitis in adults: a review of 13 cases, including diagnosis and management. J Can Dent Assoc. 2002; 68: 247–251[↩]
- Gandara‐Rey, JM, Pacheco‐Martins, JL, Gandara‐Vila, P et al. Primary herpetic gingivostomatitis in a 65‐year‐old patient. Med Oral. 2002; 7: 180–183[↩]
- Yeung‐Yue, KA, Brentjens, MH, Lee, PC et al. Herpes simplex viruses 1 and 2. Dermatol Clin. 2002; 20: 249–266[↩]
- Amir J, Harel L, Smetana Z, Varsano I. The natural history of primary herpes simplex type 1 gingivostomatitis in children. Pediatr Dermatol. 1999;16(4):259–63[↩][↩]
- Sarioglu B, Kose SS, Saritas S, Kose E, Kanik A, Helvaci M. Severe acute disseminated encephalomyelitis with clinical findings of transverse myelitis after herpes simplex virus infection. J Child Neurol. 2014;29(11):1519–23[↩]
- Faden H. Management of primary herpetic gingivostomatitis in young children. Pediatr Emerg Care. 2006;22(4):268–9.[↩]
- Klassen TP, Hartling L, Wiebe N, Belseck EM. Acyclovir for treating varicella in otherwise healthy children and adolescents. Cochrane Database Syst Rev. 2005;(4):CD002980[↩]
- Wutzler P. Antiviral therapy of herpes simplex and varicella-zoster virus infections. Intervirology. 1997;40(5–6):343–56.[↩]
- Amir, J, Harel, L, Smetana, Z et al. The natural history of primary herpes simplex type 1 gingivostomatitis in children. Pediatr Dermatol. 1999; 16: 259–263[↩]
- Yagupsky, P, Dagan, R, Howard, CB et al. Kingella kingae: an emerging course of invasive infections in young children. Clin Infect Dis. 1997; 24: 860–866[↩]
- Simmons, A. Clinical manifestations and treatment considerations of herpes simplex virus infection. J Infect Dis. 2002; 186: S1–S7[↩][↩]
- Neville, BW, Damm, DD, and Allen, CM. Herpes simplex virus. in: BW Neville (Ed.) Oral and maxillofacial pathology. 2nd edn. Saunders, Philadelphia, PA; 2003: 213–220[↩]
- Whitley, RJ. Herpes simplex virus infections. in: R Glaser, JF Jones (Eds.) Herpesvirus infections. 1st edn. Marcel Decker, New York; 1996: 1–57[↩][↩]
- Scully, C. Herpes simplex infections. in: C Scully (Ed.) Handbook of oral diseases. Diagnosis and management. 1st edn. Dunitz, London; 1999: 139–145[↩]
- Tabaee, A, Saltman, B, Shutter, J et al. Recurrent oral herpes simplex virus infection presenting as a tongue mass. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004; 97: 376–380[↩][↩]
- Simon, DM and Levin, S. Infectious complications of solid organ transplantations. in: BE Cunha (Ed.) Infectious disease clinics in North America. Infections in the compromised host. 1st edn. Saunders, Philadelphia, PA; 2001: 521–550[↩]
- Christie, SN, McCaughey, C, Marey, JJ et al. Recrudescent herpes simplex infection mimicking primary herpetic gingivostomatitis. J Oral Pathol Med. 1998; 27: 8–10[↩]
- Hedner, E, Vahlne, A, Kahnberg, KE et al. Reactivated herpes simplex virus infection as a possible cause of dry socket after tooth extraction. J Oral Maxillofac Surg. 1993; 51: 370–376[↩]
- Dohvoma, CN. Primary herpetic gingivostomatitis with multiple herpetic whitlows. Br Dent J. 1994; 177: 251–252[↩]
- Szinnai, G, Schaad, UB, and Heininger, U. Multiple herpetic whitlow lesions in a 4‐year‐old girl: case report and review of the literature. Eur J Pediatr. 2001; 160: 528–533[↩]
- Habif, TP. Herpes simplex. in: TP Habif (Ed.) Clinical dermatology. 3rd edn. Mosby, St Louis; 1996: 337–344[↩][↩]
- Stoopler, ET and Greenberg, MS. Update on herpesvirus infections. Dent Clin North Am. 2003; 47: 517–532[↩]
- Spalton, DJ, Hitchings, RA, and Hunter, PA. Herpes simplex. in: DJ Spalton, RA Hitchings, PA Hunter (Eds.) Atlas of clinical ophthalmology. 2nd edn. Wolfe, London; 1994: 4.4–4.8[↩][↩]
- Roizman, B and Knipe, DM. Herpes simplex viruses and their replication. in: DM Knipe, PM Howley (Eds.) Fields virology. 4th edn. Lippincott Williams & Wilkins, Philadelphia, PA; 2001: 2399–2459[↩]
- Albert, LJ, Zhao, Y‐X, and Inman, RD. Molecular mimicry. in: RG Lahita, N Chiorazzi, WH Reeves (Eds.) Textbook of the autoimmune diseases. 1st edn. Lippincott William & Wilkins, Philadelphia, PA; 2000: 153–174[↩]
- Lundberg, P, Welander, P, Han, X et al. Herpes simplex virus type 1 DNA is immunostimulatory in vitro and in vivo. J Virol. 2003; 77: 11158–11169[↩][↩]
- Corey, L. Herpes simplex viruses. in: E Braunwald, AS Fauci, DL Kasper (Eds.) Harrison’s principles of internal medicine. vol. 1. 15th edn. McGraw‐Hill, New York; 2001: 1100–1105[↩][↩]
- Yamada, DH, Laine, L, Owyang, Ch et al. Infectious esophagitis. in: DH Yamada, L Laine, Ch Owyang (Eds.) Textbook of gastroenterology. vol. 1. 3rd edn. Lippincott Williams & Wilkins, Philadelphia, PA; 1999: 1265–1266[↩]
- Walker, WA, Durie, PR, and Hamilton, RJ. Viral esophagitis. in: WA Walker, PR Durie, RJ Hamilton (Eds.) Pediatric gastrointestinal disease. vol. 1. 2nd edn. Mosby, St Louis; 1996: 456–457[↩]
- Bogger‐Goren, S. Acute epiglottitis caused by herpes simplex virus. Pediatr Infect Dis J. 1987; 6: 1133–1134[↩]
- Nasatzky, E and Katz, J. Bell’s palsy associated with herpes simplex gingivostomatitis. A case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998; 86: 293–296[↩]
- Galanakis, E, Bikouvarakis, S, Mamoulakis, D et al. Transverse myelitis associated with herpes simplex virus infection. J Child Neurol. 2001; 16: 866–867[↩]
- O’Sullivan, CE, Aksamit, AJ, and Harrington, JR. Clinical spectrum and laboratory characteristics associated with detection of herpes simplex virus DNA in cerebrospinal fluid. Mayo Clin Proc. 2003; 78: 1347–1352[↩]
- Ito, T, Watanabe, A, and Akanaba, J. Acute disseminated encephalomyelitis developed after acute herpetic gingivostomatitis. Tohoku J Exp Med (Japan). 2000; 192: 151–155[↩]
- Perkins, D, Gyure, KA, Pereira, EFR et al. Herpes simplex virus 1‐induced encephalitis has an apoptotic component associated with activation of c‐Jun N‐terminal kinase. J Neurovirol. 2003; 9: 101–111[↩][↩][↩][↩]
- Whitley, RJ and Gnann, JW. Viral encephalitis: familial infections and emerging pathogens. Lancet. 2002; 359: 507–514[↩][↩]
- Crumpacker, CS, Gonzalez, RG, and Makar, RS. Case 26‐2003: a 50‐year‐old Colombian man with fever and seizures. N Engl J Med. 2003; 349: 789–796[↩]
- Anglen, CS, Truckenmiller, ME, Schell, TD et al. The dual role of CD8+ T lymphocytes in the development of stress‐induced herpes simplex encephalitis. J Neuroimmunol. 2003; 140: 13–27[↩]
- Mommeja‐Marin, H, Lafaurie, M, Scieux, C et al. Herpes simplex virus type 2 as a cause of severe meningitis in immunocompromised adults. Clin Infect Dis. 2003; 37: 1527–1533[↩][↩]