Contents
What is hydrops fetalis
Hydrops fetalis is a life-threatening condition in which abnormal amounts of fluid accumulate in two or more body areas of an unborn baby. Hydrops fetalis excessive extravasation of fluid into the third space in a fetus could be due to heart failure, volume overload, decreased oncotic pressure, or increased vascular permeability. Although the fluid buildup may appear anywhere in the baby’s body, it most often occurs in the abdomen, around the heart or lungs, or under the skin. Other symptoms of the condition include higher-than-normal amounts of amniotic fluid (polyhydramnios) and a thickening of the placenta.
The estimated the incidence of hydrops fetalis is at about 1 in 2000 births although this can significantly vary according to different regions.
Hydrops is defined as the accumulation of fluid +/- edema involving at least two fetal components, which may manifest as 1:
- fetal pleural effusion
- fetal pericardial effusion
- fetal ascites
- generalised body edema: fetal anasarca/nuchal edema/cystic hygroma
- placental enlargement
- polyhydramnios
- hepatomegaly
Hydrops fetalis is not a disease, but a symptom of an underlying health problem with the baby. If untreated, the excess fluid can stress the baby’s heart and other vital organs, putting the baby’s life at risk.
Hydrops fetalis is considered a prenatal form of cardiac failure. Hydrops fetalis is traditionally divided into two broad groups:
- Non-immune hydrops fetalis. This type of hydrops fetalis accounts for approximately 80 percent to 90 percent of all cases of the condition. It occurs when an underlying disease, genetic disorder or birth defect interferes with the ability of the baby’s body to manage fluid. There are three main causes for this type hydrops fetalis are heart or lung problems, severe anemia (such as from thalassemia or infections), and genetic or developmental problems, including Turner syndrome.
- Immune hydrops fetalis (< 10% of cases). This type of hydrops fetalis occurs when there is an incompatibility between the red blood cells of the mother and baby. The most common example is when a pregnant woman has Rh-negative blood and her unborn baby has Rh-positive blood (known as Rh incompatibility). The mother’s immune system treats the baby’s Rh-positive red blood cells as “foreign invaders,” and sends out antibodies to fight them. This leads to the destruction of many of the baby’s red blood cells, an outcome that causes anemia. Severe anemia in the baby can lead to heart failure and an abnormal buildup of fluid. Most cases of red blood cell incompatibility create only mild to moderate health problems for the baby. In about one in four cases, however, the situation becomes more serious and leads to the development of hydrops fetalis. Rh incompatibility can be prevented if the mother is given RhoGAM during and after pregnancy.
Since the 1960s, the incidence of immune hydrops has fallen significantly due to the introduction of a medicine called RhoGAM. The drug, which is given as an injection to women whose pregnancies are at risk of Rh incompatibility, keeps the mother’s body from making antibodies against her baby’s red blood cells. There are other, much rarer, blood group incompatibilities that can also cause immune hydrops fetalis but RhoGAM does not help with these.
Treatment depends on the cause. During pregnancy, treatment may include:
- Medicine to cause early labor and delivery of the baby
- Early cesarean delivery if condition gets worse
- Giving blood to the baby while still in the womb (intrauterine fetal blood transfusion)
Treatment for a newborn may include:
- For immune hydrops, direct transfusion of red blood cells that match the infant’s blood type. An exchange transfusion to rid the baby’s body of the substances that are destroying the red blood cells is also done.
- Removing extra fluid from around the lungs and abdominal organs with a needle.
- Medicines to control heart failure and help the kidneys remove extra fluids.
- Methods to help the baby breathe, such as a breathing machine (ventilator).
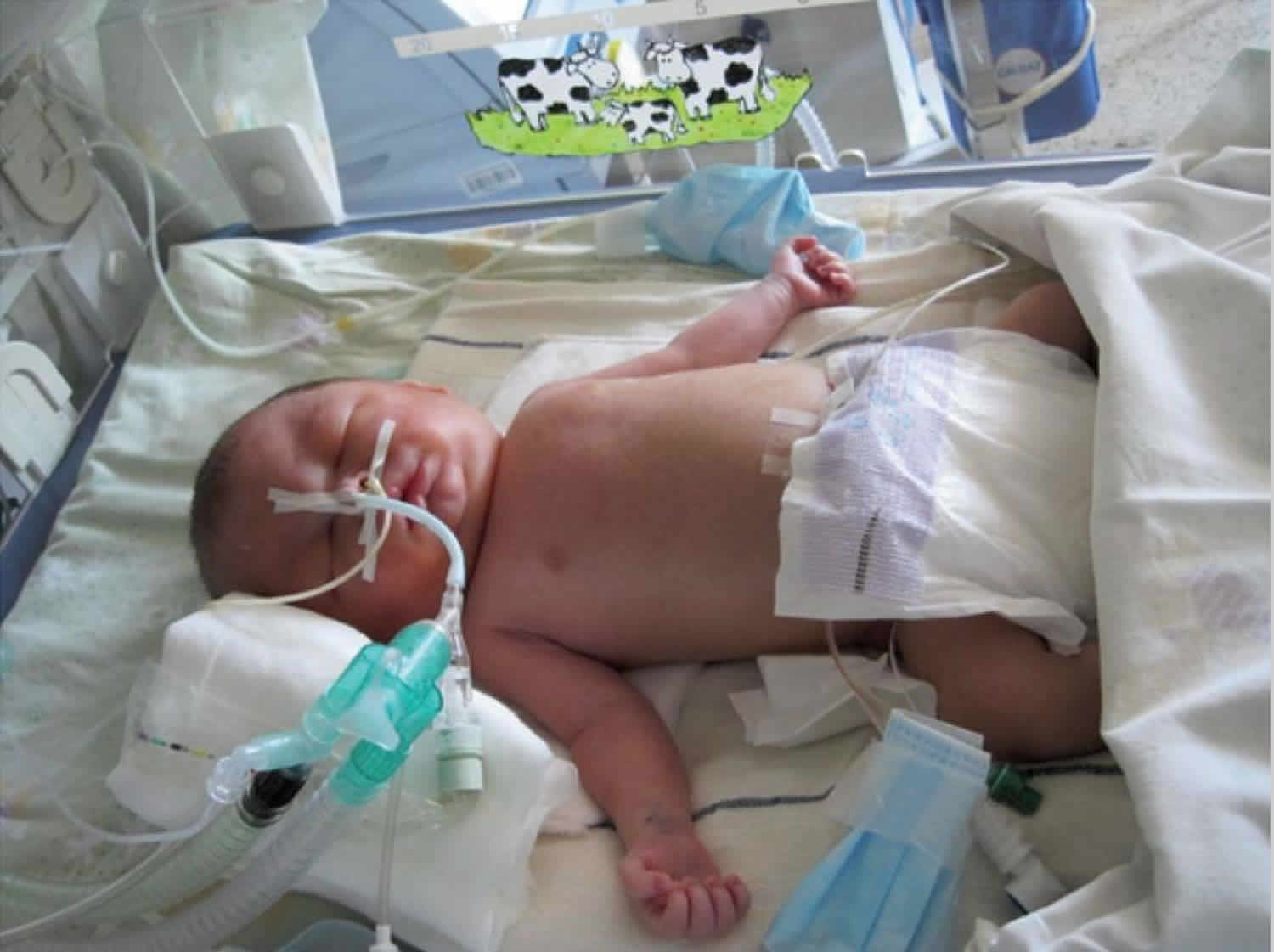
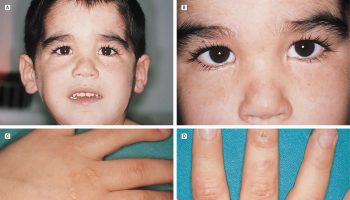
Figure 1. Hydrops fetalis baby
Footnote: A 31-year-old gravida 2 with twins, para 1 presented with spontaneous membrane rupture at 32 weeks gestation after an uncomplicated pregnancy. After admission ultrasound revealed polyhydramnion and severe non-immune hydrops fetalis in baby A. Due to life threatening condition also for healthy baby B an urgent caesarean section was performed. Baby A with non-immune hydrops fetalis was a boy with Apgar score 3/5 and a birth weight of 3,200 g. He was intubated immediately after birth because of severe non-immune hydrops fetalis and respiratory distress. Ultrasound and X-ray revealed a tumor mass in bis right hemithorax. Tumor resection was performed at the age of 7 days. Histology examination revealed an encapsulated immature teratoma. The postoperative course was complicated with respiratory insufficiency which turned into chronic at the age of eight months. At 1 year of age, the boy was discharged from hospital in a stable clinical respiratory condition with tracheostomy and Legendair ventilator. At the age of 2 years, bronchoscopy revealed massive granulation in trachea just below the tracheostoma. After removal of the granulation the tracheostoma was closed and he is breathing on his own.
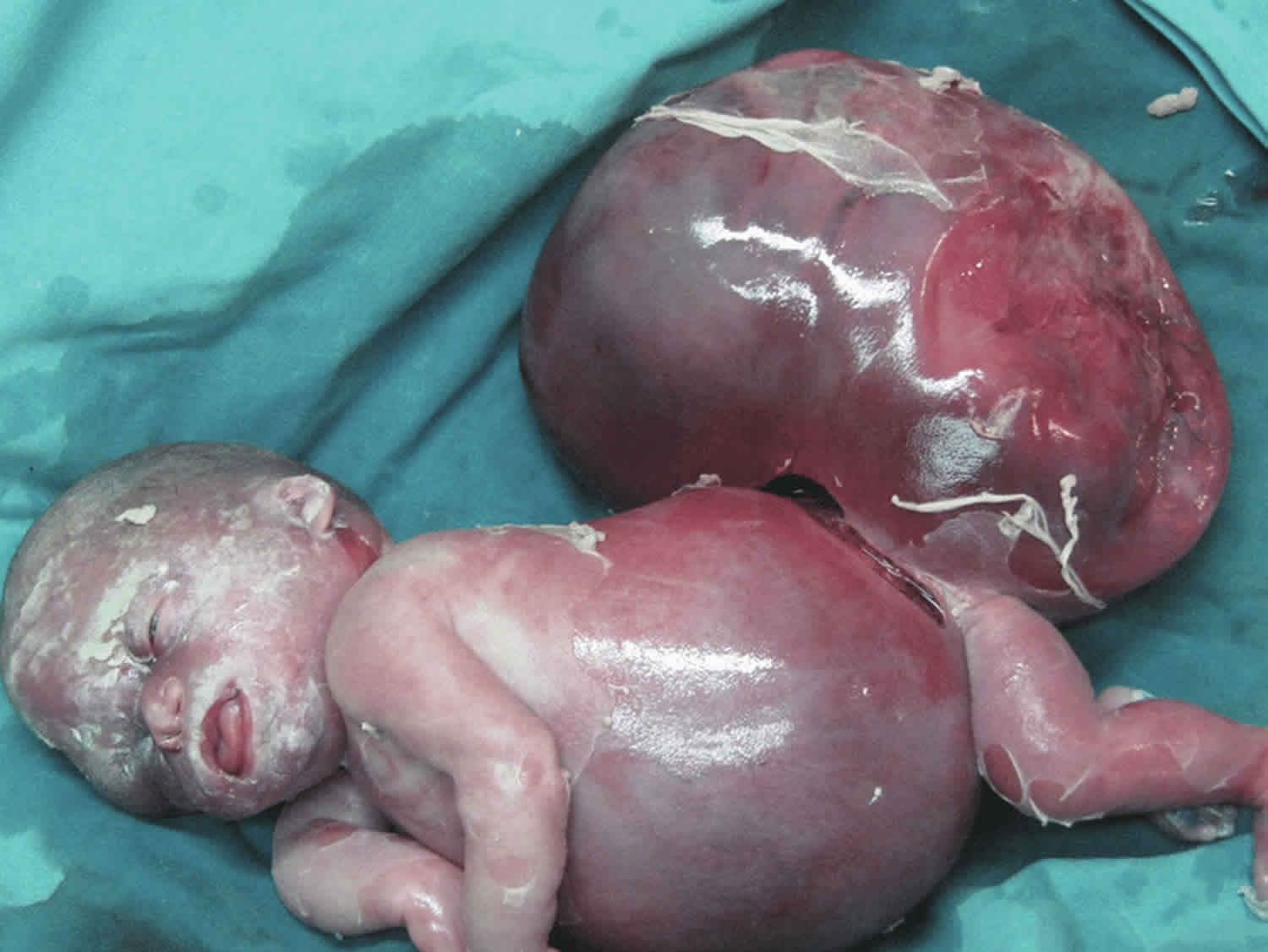
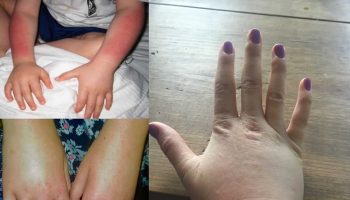
[Source 2 ]Figure 2. Hydrops fetalis baby with sacrococcygeal teratoma
Footnote: An 860 g female infant was delivered at 28 and 4/7 weeks by cesarean delivery with Apgar scores of zero at 1th and 5th minute after delivery. The placenta weighed 980 g. The infant was noted as having marked pitting skin edema, significant pleural effusions, abdominal ascites, and an sacrococcygeal teratoma approximately equal in volume to the remainder of her body.
[Source 3 ]When is hydrops fetalis detected?
Hydrops fetalis is often diagnosed during a routine prenatal ultrasound exam. In some cases, however, your doctor may order an ultrasound to specifically check for the condition. This may happen if your unborn baby’s movements decrease significantly, if the uterus measures large for the baby’s gestational age (the number of weeks the baby has been in the uterus) or if the mother develops early onset preeclampsia (high blood pressure during pregnancy). If there is a known risk due to maternal red cell antibodies (red blood cell incompatibility), routine ultrasound exams will be recommended to screen for fetal anemia and hydrops.
What causes hydrops fetalis
Non immune hydrops fetalis
Non immune hydrops fetalis accounts for a majority of cases (up to 90% of cases of hydrops fetalis) and can arise from many possible causes, including the following:
- Chromosomal anomalies
- Turner syndrome
- Trisomies
- trisomy 13 (Patau syndrome)
- trisomy 18 (Edwards syndrome)
- trisomy 21 (Down syndrome)
- Cardiac causes
- abnormalities in fetal heart rate: e.g. fetal tachyarrhythmias
- congenital cardiac anomalies
- fetal cardiac tumors: cardiac rhabdomyoma
- Twin pregnancy-related complications
- twin to twin transfusion syndrome: in the recipient twin
- twin reversed arterial perfusion sequence: in pump twin
- In utero infection(s)
- congenital viral infection(s)
- TORCH group
- fetal parvovirus B19 infection: considered the most common infectious cause of hydrops; the mechanism is that of anemia
- coxsackie viral infection
- congenital viral infection(s)
- Fetal tumors capable of producing significant AV shunts 4
- sacrococcygeal teratoma
- hepatic hemangioendothelioma
- placental chorioangioma
- Inborn errors of metabolism
- Gaucher disease
- Niemann-Pick disease
- Fetal hypoproteinemic states
- congenital nephrotic syndrome
- Congenital/fetal anemia(s)
- hemoglobin Bart’s (a.k.a. Hb Bart’s hydrops fetalis syndrome)
- 5
- cause of alpha thalassemia
- relatively common cause in Southeast Asia
- Fetal skeletal dysplasia(s) 6
- Fetal lymphovascular anomalies
- cystic hygroma 5
- High output flow states
- vein of Galen aneurysmal malformation
- umbilical vein thrombosis in cases of umbilical vein varix
- Thoracic/pulmonary abnormalities: thought to be from a fetal venous obstruction
- Birth defects that affect how the heart functions, such as a heart defect or an arrhythmia (an abnormal heart beat or beating pattern), or a tumor that compress the heart or the blood flow to the heart
- A hemoglobin abnormality or other genetic disorder that causes the baby’s blood cells to be destroyed
- Chromosomal disorders, such as Down’s syndrome (trisomy 21) or Turner syndrome
- Congenital infections (ones passed from the mother to the baby before birth), including parvovirus B19 (fifth disease) and cytomegalovirus (CMV)
- Liver disease
- Tumors with a large amount of blood flow
- A monochorionic pregnancy (in which two or more genetically identical babies share the same placenta)
- Fetal anemia due to non-immune causes, including several of the conditions listed above, as well as bleeding in the mother’s abdomen, sometimes caused by trauma
In some cases, however, there is no identifiable reason for non-immune hydrops to appear.
Immune hydrops fetalis
Immune hydrops fetalis is caused by an incompatibility of red blood cells between the mother and her unborn baby. That incompatibility leads to the destruction of a large number of the baby’s red blood cells (also known as hemolytic disease). As the baby’s body tries to make new red blood cells and the heart works harder, excess fluid can form and fill spaces around the baby’s heart, lungs and other organs.
Immune hydrops fetalis accounts for a minority (~10%) of cases in present-day pregnancies 9, although historically accounted for a larger proportion of cases in the early to mid 20th century due to the fetomaternal blood group incompatibility (including rhesus incompatibility): erythroblastosis fetalis or hemolytic disease of the fetus and newborn.
Hydrops fetalis pathophysiology
Several hypotheses regarding the pathophysiologic events that lead to hydrops fetalis have been suggested. The basic mechanism for the formation of hydrops fetalis is an imbalance of interstitial fluid production and the lymphatic return 10. Fluid accumulation in the fetus can result from congestive heart failure, obstructed lymphatic flow, or decreased plasma osmotic pressure. The fetus is particularly susceptible to interstitial fluid accumulation because of its greater capillary permeability, compliant interstitial compartments, and vulnerability to venous pressure on lymphatic return.
Compensatory mechanisms for maintaining homeostasis during hypoxia that results from underlying disease include increased efficiency of oxygen extraction; redistribution of blood flow to the brain, heart, and adrenals, thus causing renal tubular damage; volume augmentation to enhance cardiac output; and marked activation of the renin-angiotensin system. Unfortunately, these mechanisms also increase venous pressure and ultimately produce interstitial fluid accumulation and characteristic hydropic changes in the fetus. Increased venous pressure contributes to edema and effusion by increasing the capillary hydrostatic pressure and decreasing the lymphatic return. Impaired renal function causes oliguria or anuria and, subsequently, hydrops 11.
Furthermore, the hepatic synthesis of albumin may be impaired owing to decreased hepatic perfusion and increased extramedullary hematopoiesis. Because albumin acts as the predominant oncotically active plasma protein, hypoalbuminemia increases transcapillary fluid movement at times of circulatory compromise.
Hydrops has been produced in the ovine fetus by anemia, tachyarrhythmia, occlusion of lymphatic drainage, and obstruction of cardiac venous return. Hypoproteinemia and hypoalbuminemia are common in human hydrops, and reduced intravascular oncotic pressure has been speculated to be a primary cause for the disorder. However, in the sheep model, a 41% reduction in total serum protein accompanied by a 44% decline in colloid osmotic pressure failed to produce fetal hydrops 12. Furthermore, a study in humans showed that, despite a significant negative correlation between the fetal serum albumin level and the degree of fetal hydrops, most fetuses with hydrops had albumin levels within the reference range 13. These results suggest that hypoalbuminemia is unlikely the sole cause for the primary onset of hydrops fetalis.
A closer look at the animal studies provides the clues necessary to understand the pathophysiology of hydrops. In one study, profound anemia was induced in fetal sheep; the hydrops that resulted was unrelated to hematocrit levels, blood gas levels, acid-base balance, plasma proteins, colloid oncotic pressure, or aortic pressure 14. The investigators found the difference was in the central venous pressure (CVP), which was much higher in persons with hydrops. The hematocrit level was reduced by 45% in a study of particular note; however, the central venous pressure was maintained unchanged, and no fetus developed hydrops under these conditions.
Induced fetal tachyarrhythmia has led to fetal hydrops in several studies. Key to the development of fetal hydrops in these studies was an elevation in central venous pressure; the anemia was only of indirect importance. The central venous pressure was markedly elevated, with a range of 25-31 mm Hg in one study. In other reports, hydrops induced by sustained fetal tachycardia was unrelated to levels of blood gases, plasma protein, or albumin turnover; however, a 75-100% increase in central venous pressure was observed in the fetuses that developed hydrops.
Excision of major lymphatic ducts produced fetal hydrops in sheep models. A related study demonstrated an exquisite, linear, inverse relationship between the lymphatic outflow pressure and the central venous pressure; a rise in the central venous pressure of 1 mm Hg reduced lymph flow 13%, and flow stopped at a central venous pressure of 12 mm Hg 15. These results were confirmed by other observations of linear decline in lymph flow when the central venous pressure exceeds 5 mm Hg and a cessation of flow at central venous pressures greater than 18 mm Hg.
Also of note is a computer simulation model in which cardiovascular and fluid electrolyte disturbances (e.g., severe anemia, lymphatic obstruction, excess fluid and electrolyte loads, elevation in angiotensin levels) and compensating homeostatic mechanisms have been examined. This model demonstrated that “…fetal cardiac failure constituted the strongest stimulus for the formation of fetal edema…” 16, thus further substantiating the pivotal role of central venous pressure in the development of fetal hydrops.
Many other physiologic disturbances are associated with human hydrops fetalis. Elevated levels of aldosterone, renin, norepinephrine, and angiotensin-I are likely to be secondary consequences. Although infusion of angiotensin-I led to fetal hydrops in nephrectomized sheep, the four-fold rise in the central venous pressure was probably the primary cause of the hydrops. The meaning of increased levels of coenzyme Q10, placental vascular endothelial growth factor, and endothelin and decreased cytokine interleukin-3 levels is unclear at this time.
However, of particular interest is the three- to five-fold increase in atrial natriuretic peptide (ANP) that accompanies both human fetal hydrops (with cardiac anomaly or isoimmunization) 17 and ovine hydrops (induced by obstruction of venous return, sustained tachycardia, or induced anemia) 18. A return of ANP levels to normal parallels the resolution of hydrops. These observations and the observations that vascular permeation of albumin is enhanced and cardiovascular and renal homeostatic adaptations are influenced by this peptide suggest an important role for atrial natriuretic peptide in hydrops fetalis.
Evidence of low fetal plasma levels of cyclic guanosine monophosphate suggests that reduced nitric oxide production due to injury of fetal vascular endothelial cells may be involved in the development of hydrops fetalis 18. This isolated observation requires confirmation and further study.
Despite numerous case reports, decades of clinical experience, and several research studies regarding the cause and pathogenesis of hydrops fetalis, many questions still remain. Currently, which fetal neural and hormonal mechanisms induce and maintain the redistribution of blood flow, and which mechanisms allow metabolic disorders to cause hydrops fetalis, are almost completely unknown 11.
Hydrops fetalis symptoms
Symptoms depend on the severity of the condition. Mild forms may cause:
- Liver swelling
- Change in skin color (pallor)
More severe forms may cause:
- Breathing problems
- Bruising or purplish bruise-like spots on the skin
- Heart failure
- Severe anemia
- Severe jaundice
- Total body swelling
Hydrops fetalis diagnosis
Hydrops fetalis is often diagnosed during a routine prenatal ultrasound exam. In some cases, however, your doctor may order an ultrasound to specifically check for the condition. This may happen if your unborn baby’s movements decrease significantly, if the uterus measures large for the baby’s gestational age (the number of weeks the baby has been in the uterus) or if the mother develops early onset preeclampsia (high blood pressure during pregnancy). If there is a known risk due to maternal red cell antibodies (red blood cell incompatibility), routine ultrasound exams will be recommended to screen for fetal anemia and hydrops.
The diagnosis of hydrops fetalis will be made if the ultrasound reveals abnormal amounts of fluid in two or more areas of your baby’s body. The ultrasound may also reveal other signs associated with hydrops fetalis, including a large amount of amniotic fluid, a thickened placenta, and an enlarged heart, liver or spleen.
To distinguish non-immune from immune hydrops, the mother’s blood will be tested for the presence of antibodies associated with red blood cell incompatibility.
Hydrops fetalis ultrasound
Antenatal ultrasound
Sonographic features can be similar for both immune and non-immune hydrops and include:
- increased amniotic fluid volumes
- increased nuchal translucency (especially 1st trimester) 19
- larger placental size (placentomegaly) / increased placental thickness (placental edema)
- presence of a fetal pleural or pericardial effusion
- generalised fetal body swelling: fetal anasarca and skin thickening
- umbilical venous dilatation
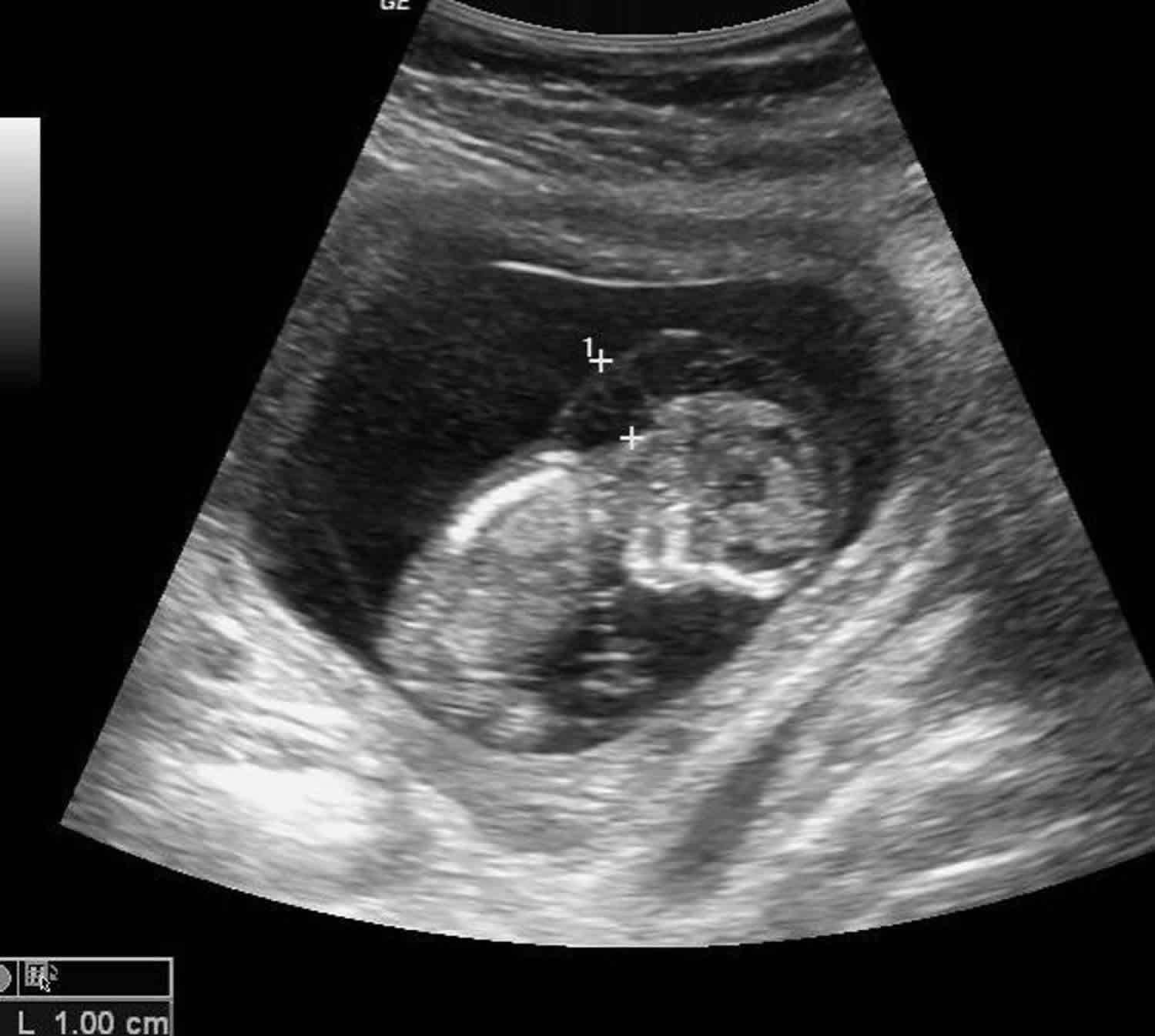
Figure 3. Hydrops fetalis ultrasound
Footnote: Generalized subcutaneous odema involving scalp, chest, abdomen. Pleural effusion on one side only. TORCH test – Positive.
[Source 20 ]Fetal echocardiography
Fetal echocardiography (“echo” for short) is performed at by a pediatric cardiologist (a physician who specializes in fetal heart abnormalities). This non-invasive, high-resolution ultrasound procedure looks specifically at how your baby’s heart is structured and how well it is functioning within the womb.
Amniocentesis
An amniocentesis is done to sample the amniotic fluid around your baby. This fluid will contain some of your baby’s cells, which will provide a full set of your baby’s chromosomes for us to analyze. Your doctor can then assess if the cause of the hydrops fetalis is related to a genetic or chromosomal abnormality. The amniotic fluid may also be assessed for infection or liver/metabolic disease — if there is suspicion that these are the possible causes. The amniocentesis procedure is straightforward and can be done in our clinic. It requires placing a small needle through the mother’s abdomen and into the amniotic sac to obtain the fluid sample. Getting final results from the test from our laboratory usually takes several days. The information will be very important for creating your care plan and helping the neonatologist care for your infant after birth.
Hydrops fetalis treatment
Hydrops fetalis treatment before birth
The prenatal management of babies with hydrops fetalis starts with acquiring as much information about the condition’s severity and underlying cause as early as possible. A thorough history of the mother’s health will be obtained to screen for infections or other possible factors related to the mother’s health that can lead to fetal anemia and hydrops fetalis. Your doctor will also gather information through the use of several different technologies, including high-resolution fetal ultrasonography, fetal echocardiography and amniocentesis.
High-resolution fetal ultrasonography is a non-invasive test performed by one of our ultrasound specialists. The test uses reflected sound waves to create images of your baby within the womb. We will use ultrasonography to identify possible birth defects related to hydrops fetalis, as well as to follow the development of your baby’s internal organs and overall growth throughout the pregnancy. The test will also allow us to keep track of the amount of amniotic fluid surrounding your baby, as well as how well blood is flowing through the umbilical cord and within your baby’s other blood vessels. Blood flow within the middle cerebral artery (MCA) in the brain will help us to determine if there is evidence of fetal anemia.
After your doctor(s) have gathered all the anatomic and diagnostic information from the tests, your healthcare team will meet with you to discuss the results and any treatment possibilities. Treatment will depend on several factors, including the cause of your baby’s hydrops fetalis, the extent of the condition and your baby’s gestational age.
If the suspected cause of the condition is a loss of red blood cells (fetal anemia), your doctor may recommend percutaneous umbilical cord blood sampling, also called cordocentesis. A very thin needle will be used to draw a sample of your baby’s red blood cells from the umbilical cord. If the red blood cell count is low, an intrauterine fetal blood transfusion may be recommended. This procedure, which is similar to amniocentesis, injects red blood cells from a compatible donor into the baby’s umbilical cord.
In rare cases of tumor-caused hydrops fetalis, the possibility of prenatal surgery to remove the tumor may be discussed.
Prenatal surgery may also be discussed in cases of complicated monochorionic twin pregnancies.
If your baby has a cardiac arrhythmia, your doctor may recommend a medication to help regulate the baby’s heartbeat. The medication will be given to the mother and then passed through the placenta to the baby.
Excess fluid within the baby can sometimes be removed prenatally during a single procedure with a needle, or a shunt can be placed to continuously drain the fluid. For example, if there is fluid collecting around the baby’s lungs, a small curled tube can be inserted into the chest to drain the fluid into the amniotic cavity. This treatment resolves the pressure against the baby’s heart and lungs.
Often, however, hydrops fetalis can’t be treated during pregnancy. In such cases, we will monitor both mother and baby carefully throughout the pregnancy, looking for any developments that might necessitate an early delivery. Depending on the gestational age, your doctor may recommend biophysical profile studies (BPPs) to evaluate the baby’s wellbeing. During this test, a heart-rate monitor is placed on the mother’s abdomen to observe the baby’s heart rate pattern in addition to an ultrasound exam evaluating the baby’s movements. Your doctor will use the test, which can last for up to 30 minutes, to determine if your baby is receiving enough oxygen from the placenta. The test also helps to assess the amount of amniotic fluid surrounding the baby.
Your healthcare team goal is to get your baby as close to the due date as possible, since early delivery can be life-threatening for your baby. The parents’ wishes will be considered when discussing delivery timing and intervention. Regardless of any potential treatment, hydrops fetalis is a serious condition and there is a chance the baby will not survive the pregnancy or after delivery.
Immediate delivery of a baby with hydrops fetalis is sometimes needed to protect the health of the mother. Women whose babies have hydrops fetalis are at increased risk of developing a potentially life-threatening form of pregnancy-related high blood pressure known as “mirror syndrome.” It’s called that because the mother’s symptoms mirror the fluid buildup in the baby. The only treatment for mirror syndrome is to deliver the baby.
Hydrops fetalis treatment after birth
Your healthcare team goal will be to have your baby’s birth occur as near to your due date as possible, but infants with hydrops fetalis must often be delivered early. Delivery is frequently by cesarean section because there is an increased risk the baby will not tolerate labor and delivery. Your baby will be born at one of only a few centers nationwide with a birth center located within the hospital complex. This means that your baby will be born just a few feet down the hall from our newborn intensive care unit (NICU). Also, many of the doctors you have already met will be present during or immediately after your baby’s birth to help care for your baby right away.
Your baby will need specialized medical care after birth and will therefore be taken to our newborn intensive care unit (NICU). A mechanical breathing machine (ventilator) may be used to help your baby breathe, and a needle may be used to remove fluid from the spaces around the heart, lungs and abdomen. Your baby will also be placed on special intravenous (IV) lines for the delivery of needed nutrients and medications. The medicines are likely to include ones to keep the heart functioning and to help the kidneys remove excess fluid from the baby’s body. If your baby has immune hydrops, he or she may receive an exchange transfusion of red blood cells.
Our goal will be to make a definitive diagnosis of the underlying cause of the hydrops fetalis as quickly as possible. To make that diagnosis, further tests will be done soon after birth.
Will my baby need an operation?
Your baby may need an operation to treat the underlying cause of the hydrops fetalis. Your comprehensive care team at the Midwest Fetal Care Center will explain all treatment options, including surgery, for your baby.
How long will my baby be in the hospital?
Your baby will be discharged from the hospital after the underlying disease that caused the hydrops fetalis has stabilized, the baby has healed from all surgical procedures, and he or she is taking food by mouth without difficulty.
Hydrops fetalis prognosis
The prognosis for babies with hydrops fetalis depends on the underlying cause of the condition. Overall, however, the survival rate is low. More than half of all babies with the condition die before birth or soon after delivery. The risk is highest for those who are diagnosed with hydrops fetalis early (less than 24 weeks into pregnancy) and for those who have a structural abnormality, such as a heart defect.
Hydrops fetalis long term effects
Because of the potential health issues associated with hydrops fetalis, your baby may require long-term follow-up care. Your healthcare team will have developed a detailed care plan for babies born with hydrops fetalis. Your baby’s plan will be implemented by a comprehensive team of specialists, including a pediatrician (who will coordinate your baby’s overall care), a pediatric cardiologist, a developmental specialist and any other caregivers your baby may require.
Brain damage called kernicterus may occur in the case of Rh incompatibility. Developmental delays have been seen in babies who received intrauterine transfusions.
Hydrops fetalis survival rate
Hydrops fetalis often results in death of the infant shortly before or after delivery. The risk is highest for babies who are born very early or who are ill at birth. Babies who have a structural defect, and those with no identified cause for the hydrops fetalis are also at higher risk.
Estimates of mortality vary widely, from nearly zero to virtually 100%. Most case series report a 60%-90% mortality, although some improvements are notable in more recent reports. Many reasons for these variations are recognized, not least of which include the sophistication of diagnostic methods used and the complexity and costs of treatment.
However, the most important single factor is the cause of the hydrops fetalis. A significant proportion of these cases are caused or accompanied by multiple and complex congenital malformations of genetic and/or chromosomal origin, which by themselves are fatal at an early age. Many other causes are accompanied by masses or fluid accumulations, which compress the developing fetal lung and preclude its normal development. Thus, the presence or absence and potential prevention of pulmonary hypoplasia are of crucial importance.
One study showed that mortality rate was highest among neonates with congenital anomalies (57.7%) and lowest among neonates with congenital chylothorax (5.9%) 21. Infants who died were more likely to be more premature, were sicker after birth, with lower 5-minute Apgar scores, and needed higher levels of support during the first day after birth.
Another highly important factor is the very premature delivery of most babies with hydrops consequent to conditions that distend the uterus and provoke early labor, or to therapeutic interventions (e.g., fetal thoracentesis, paracentesis, complex fetal surgical procedures).
A 2017 report of data from the California Office of Statewide Health Planning and Development (2005-2012) regarding 1037 live-born infants with nonimmune hydrops fetalis revealed a 35.1% neonatal mortality and a 43.2% overall mortality at age 1 year 22. Poor prognostic factors were prematurity, polyhydramnios, and large for gestational age.
In a separate 2017 report that analyzed data from an international registry of 69 survivors with hemoglobin Bart hydrops fetalis, investigators found that more than half survived beyond age 5 years (n = 39; 56.5%), of whom half (n = 18; 26.1%) were older than 10 years 23. Although intrauterine therapy appeared to be beneficial during the perinatal and neonatal periods, the investigators suggested that further benefits might not extend to long-term growth and neurodevelopment outcomes. Indeed, over time, about 40% of these patients had severe weight retardation and 50% had height retardation, and 20% had a neurodevelopment delay of 6 months or longer 23.
- Hydrops fetalis. https://radiopaedia.org/articles/hydrops-fetalis?lang=us[↩]
- Simoncic M, Kopriva S, Zupancic Z, et al. Mediastinal teratoma with hydrops fetalis in a newborn and development of chronic respiratory insufficiency. Radiol Oncol. 2014;48(4):397-402. Published 2014 Nov 5. doi:10.2478/raon-2013-0080 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4230561/[↩]
- Kafali H, Onaran YA, Keskin E, Sari U, Kirbas I. Ovarian vein thrombosis and mirror syndrome in association with sacrococcygeal teratoma. Clinics (Sao Paulo). 2010;65(4):452-5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2862666/[↩]
- Woodward PJ, Sohaey R, Kennedy A et-al. From the archives of the AFIP: a comprehensive review of fetal tumors with pathologic correlation. Radiographics. 25 (1): 215-42. doi:10.1148/rg.251045156[↩]
- Ismail KM, Martin WL, Ghosh S et-al. Etiology and outcome of hydrops fetalis. J Matern Fetal Med. 2001;10 (3): 175-81. https://www.ncbi.nlm.nih.gov/pubmed/11444786[↩][↩]
- Jauniaux E, Van maldergem L, De munter C et-al. Nonimmune hydrops fetalis associated with genetic abnormalities. Obstet Gynecol. 1990;75 (3 Pt 2): 568-72.[↩]
- Merz E, Bahlmann F. Ultrasound in obstetrics and gynecology. Thieme Medical Publishers. (2005) ISBN:1588901475.[↩][↩]
- da Silva OP, Ramanan R, Romano W et-al. Nonimmune hydrops fetalis, pulmonary sequestration, and favorable neonatal outcome. Obstet Gynecol. 1996;88 (4 Pt 2): 681-3.[↩]
- Etiology and Outcome of Hydrops Fetalis: Report of 62 Cases. Pediatrics and Neonatology, Volume 55, Number 2, 2014, pp. 108-113(6) https://doi.org/10.1016/j.pedneo.2013.07.008[↩]
- Pediatric Hydrops fetalis. https://emedicine.medscape.com/article/974571-overview[↩]
- Bellini C, Hennekam RC. Non-immune hydrops fetalis: a short review of etiology and pathophysiology. Am J Med Genet A. 2012 Mar. 158A(3):597-605.[↩][↩]
- Moise AA, Gest AL, Weickmann PH, McMicken HW. Reduction in plasma protein does not affect body water content in fetal sheep. Pediatr Res. 1991 Jun. 29(6):623-6.[↩]
- Pasman SA, Meerman RH, Vandenbussche FP, Oepkes D. Hypoalbuminemia: a cause of fetal hydrops?. Am J Obstet Gynecol. 2006 Apr. 194(4):972-5.[↩]
- Blair DK, Vander Straten MC, Gest AL. Hydrops in fetal sheep from rapid induction of anemia. Pediatr Res. 1994 May. 35(5):560-4.[↩]
- Brace RA. Effects of outflow pressure on fetal lymph flow. Am J Obstet Gynecol. 1989 Feb. 160(2):494-7.[↩]
- Shinbane JS, Wood MA, Jensen DN, et al. Tachycardia-induced cardiomyopathy: a review of animal models and clinical studies. J Am Coll Cardiol. 1997 Mar 15. 29(4):709-15.[↩]
- Nimrod C, Keane P, Harder J, et al. Atrial natriuretic peptide production in association with nonimmune fetal hydrops. Am J Obstet Gynecol. 1988 Sep. 159(3):625-8.[↩]
- Silberbach M, Woods LL, Hohimer AR, et al. Role of endogenous atrial natriuretic peptide in chronic anemia in the ovine fetus: effects of a non-peptide antagonist for atrial natriuretic peptide receptor. Pediatr Res. 1995 Nov. 38(5):722-8.[↩][↩]
- Has R, Recep H. Non-immune hydrops fetalis in the first trimester: a review of 30 cases. Clin Exp Obstet Gynecol. 2001;28 (3): 187-90.[↩]
- Hydrops fetalis. https://radiopaedia.org/cases/hydrops-fetalis-5[↩]
- Abrams ME, Meredith KS, Kinnard P, Clark RH. Hydrops fetalis: a retrospective review of cases reported to a large national database and identification of risk factors associated with death. Pediatrics. 2007 Jul. 120(1):84-9.[↩]
- Steurer MA, Peyvandi S, Baer RJ, et al. Epidemiology of live born infants with nonimmune hydrops fetalis-insights from a population-based dataset. J Pediatr. 2017 May 19.[↩]
- Songdej D, Babbs C, Higgs DR, for the BHFS International Consortium. An international registry of survivors with Hb Bart’s hydrops fetalis syndrome. Blood. 2017 Mar 9. 129 (10):1251-9.[↩][↩]