Contents
What is Immunoglobulin
Immunoglobulin also called Ig, antibody, immune serum globulin, immune globulin or gamma globulin that helps your body fight infection and are essential in protecting your body against bacteria, viruses, and fungi. Immunoglobulins (Ig) or antibodies are glycoproteins that are produced by specific immune cells called plasma cells (fully differentiated B-lymphocytes) in the bone marrow and other lymphoid tissue in response to bacteria, viruses, fungi, toxins, parasites and other microorganisms as well as exposures to other substances that are recognized by the body as “non-self” harmful foreign substances (antigens) 1, 2. B cells (B-lymphocytes) are instructed by specific immunogens, for, example, bacterial proteins, to differentiate into plasma cells, which are protein-making cells that participate in humoral immune responses against bacteria, viruses, fungi, parasites, cellular antigens, chemicals, and synthetic substances 3. Some immunoglobulins may be found in higher than normal amounts in individuals with certain conditions or certain types of cancer, including multiple myeloma and Waldenstrom macroglobulinemia. Measuring the amount of specific immunoglobulins in the blood and urine may help diagnose cancer or find out how well treatment is working or if cancer has come back. Some immunoglobulins may be used as tumor markers.
The immunogen or antigen reacts with a B-cell receptor on the cell surface of B lymphocytes, and a signal is produced that directs the activation of transcription factors to stimulate the synthesis of antibodies, which are highly specific for the immunogen that stimulated the B cell. Furthermore, one clone of B cell makes an immunoglobulin (specificity). Besides, the immune system remembers the antigens that caused a previous reaction (memory) due to the development of memory B cells. These are intermediate, differentiated B cells with the capability to quickly become plasma cells. Circulating antibodies recognize antigen in tissue fluids and serum.
The first time a person is infected or otherwise exposed to a foreign substance (antigen), their immune system recognizes the microorganism or substance as “non-self” and stimulates plasma cells to produce specific immunoglobulin(s), also called antibodies, that can bind to and neutralize the threat. With subsequent exposures, the immune system “remembers” the antigen that was encountered, which allows for the rapid production of more antibodies and, in the case of microorganisms, helps prevent re-infection.
A solution made from human blood plasma that contains concentrated antibodies (immunoglobulins) that protect against specific diseases, such as short-term protection against certain infections and Rh sensitization during pregnancy; it is made from human blood plasma that has been pooled, processed from donated blood, and purified.
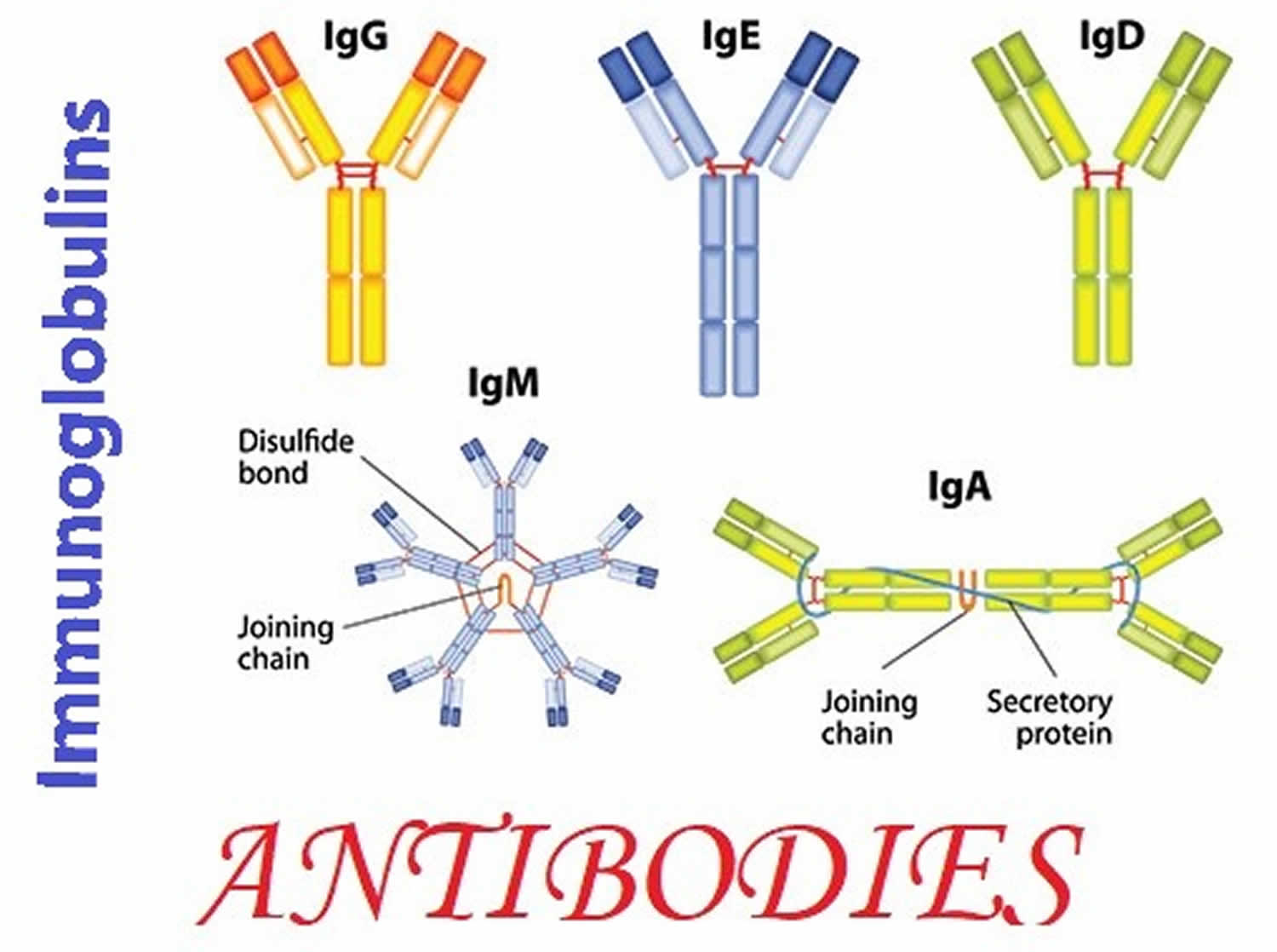
There are five classes of immunoglobulins and several subclasses. Each class represents a group of antibodies and has a slightly different role. Classes of immunoglobulins include:
- Immunoglobulin M (IgM) – Immunoglobulin M or IgM antibodies are produced as a body’s first response to a new infection or to a new “non-self” antigen, providing short-term protection. IgM antibodies are the first immunoglobulins your body makes after you’re exposed to germs or to a new “non-self” antigen. Immunoglobulin M or IgM antibodies are in your blood and lymph fluid (a watery fluid that carries the cells that fight infections and diseases to all parts of your body). Immunoglobulin M (IgM) increase for several weeks and then decline as IgG production begins.
- Immunoglobulin G (IgG) – About 70-80% of the immunoglobulins in your blood are immunoglobulins G or IgG. Specific IgG antibodies are produced during an initial infection or other antigen exposure, rising a few weeks after it begins, then decreasing and stabilizing. Your body retains a catalogue or “blueprint” of all the IgG antibodies you have made. That way, if you’re exposed to the same germs or antigen again, your immune system can quickly make more IgG antibodies. IgG antibodies form the basis of long-term protection against microorganisms. In those with a normal immune system, sufficient IgG is produced to prevent re-infection. Vaccinations use this process to prevent initial infections and add to the catalog of IgG antibodies, by exposing a person to a weakened, live microorganism or to an antigen that stimulates recognition of the microorganism. Immunoglobulins G or IgG is the only immunoglobulin that can pass through the placenta. The mother’s IgG antibodies provide protection to the fetus during pregnancy and to the baby during its first few months of life. There are four subclasses of IgG: IgG1, IgG2, IgG3, and IgG4.
- Immunoglobulin A (IgA) – Immunoglobulin A or IgA comprises about 15% of the total immunoglobulins in your blood but is also found in saliva, tears, respiratory and gastric secretions, and breast milk. Immunoglobulin A or IgA provides protection against infection in mucosal areas of the body such as the respiratory tract (sinus and lungs) and the gastrointestinal tract (stomach and intestines). When passed from mother to baby during breast-feeding, IgA helps protect the infant’s gastrointestinal tract. Significant amounts of IgA are not produced by a baby until after 6 months of age so any IgA present in a baby’s blood before then is from the mother’s milk. There are two IgA subclasses: IgA1 and IgA2.
- Immunoglobulin E (IgE) – IgE is associated with allergies, allergic diseases, and with parasitic infections. It is almost always measured as part of an allergy testing blood panel but typically is not included as part of a quantitative immunoglobulins test.
- Immunoglobulin D (IgD) – the role of IgD is not completely understood and IgD is not routinely measured.
Figure 1. Immunoglobulins
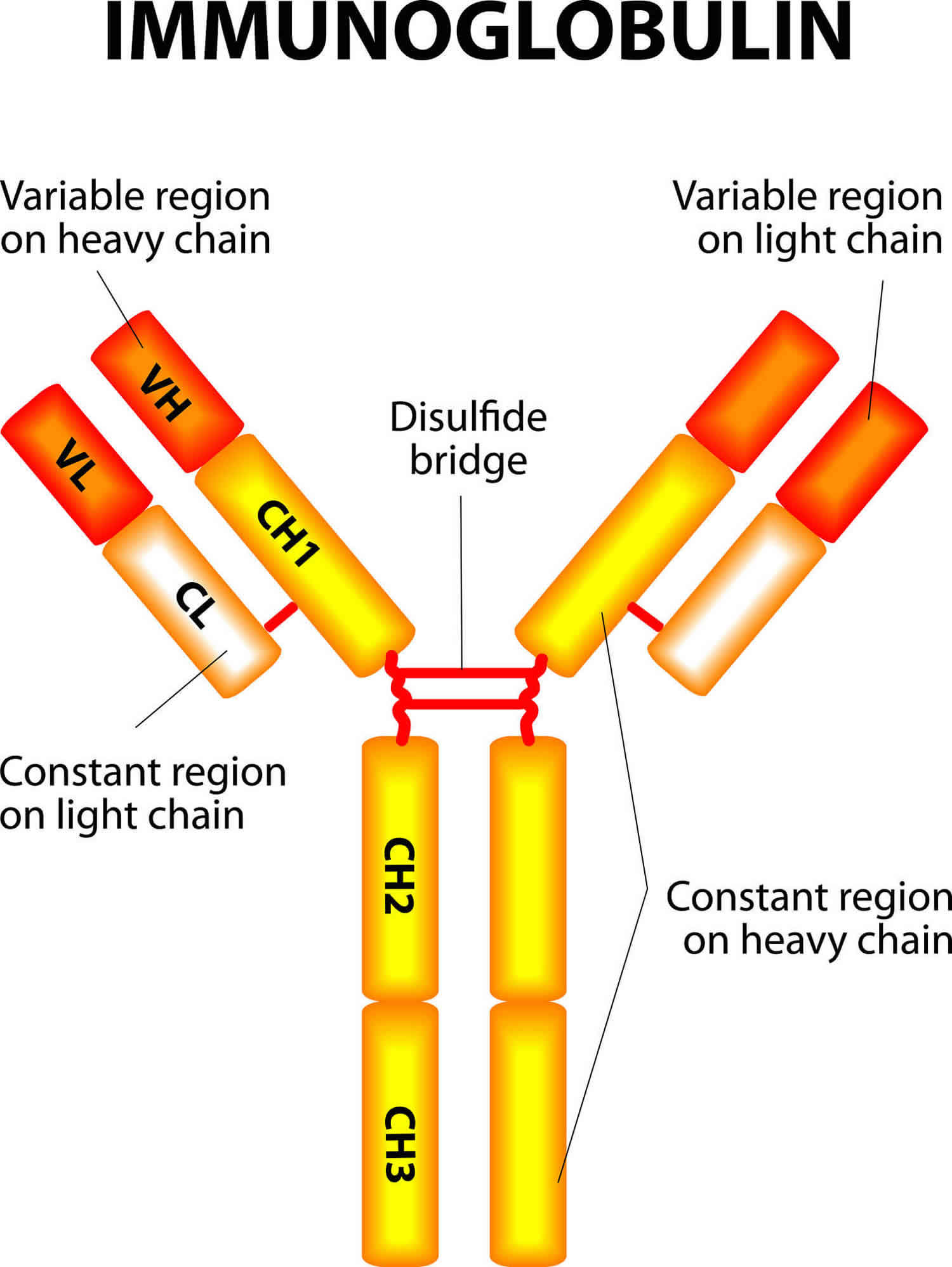
Figure 2. Immunoglobulin structure
Immunoglobulin function
Immunoglobulins or antibodies are proteins made by your immune system to fight bacteria, viruses, fungi and toxins. Immunoglobulins or antibodies have two light chains and two heavy chains in a light-heavy-heavy-light structure arrangement (see Figure 2). The heavy chains differ among classes. They have one Fc region that mediates biological functions (e.g., the binding capacity to cellular receptors) and a Fab region, where resides the antigen-binding sites. The chains are folded into regions called domains. There are 4 or 5 domains in the heavy chain, depending on their class, and two domains in the light chain. In the hypervariable regions (HRR) contain the antigen-binding sites. There are three hypervariable regions in the V domains of each light and heavy chain. These fold into regions that produce 2 antigen-binding sites at the tip of each monomer. All antibodies exhibit one or more functions (bifunctional) including activation of the complement system, opsonization of microbes to be easily phagocytosed, prevention of attachment of the microbes to mucosal surfaces, and neutralization of toxins and viruses 4.
Receptors for Immunoglobulins
For immunoglobulins to fulfill various biological functions, they should interact with receptors that are mainly expressed on mononuclear cells, mast cells, neutrophils, natural killer cells, and eosinophils. Binding to these receptors is essential for immunoglobulin functions 1. It promotes several activities including phagocytosis of bacteria (opsonization); mast cell degranulation (as seen in type 1 hypersensitivity or allergic response); killing of tumors; and activation of antigen-presenting cells including macrophages and dendritic cells, which present antigens to T lymphocytes for the generation of cellular and humoral immune responses 5.
The following are immunoglobulin receptors:
- Fc gamma RI (CD64) binds to monomeric IgG is expressed on phagocytes and is involved in the phagocytosis of immune complexes.
- Fc gamma RII (CD32) attaches to B-cells, monocyte/macrophages (phagocytes) and granulocytes. On B cells regulates cell activation in the presence of a high titer of antibodies.
- Fc gamma RIII (CD16) has 2 types. Fc gamma RIIIa is expressed on macrophages, NK cells, and some T cells. Fc gamma RIIIb is expressed on granulocytes and has low affinity for IgG.
- Fc epsilon RI is a high-affinity receptor for IgE that is shown on mast cells and basophils. It involves an allergic response.
- Fc epsilon RII is expressed on leukocytes and lymphocytes and has homology with mannose-binding lectin.
Genetics of Immunoglobulins
The immune system can respond to many antigens by generating a vast diversity in immunoglobulins produced by plasma cells. V and J gene segments encode immunoglobulin light chains. The above genes, in addition to D gene segments, encode the heavy chains. The mechanisms that contribute to this great diversity of immunoglobulin specificities include somatic mutation (immunoglobulin heavy and light chain genes undergo structural modifications after antigen stimulation) and the presence of multiple V-region genes in the germline (antibody diversity also arises when numerous V genes are recombining with J and D segments). Gene conversion, recombinational inaccuracies, nucleotide addition, and assorted heavy and light chains also contribute to the diversity of immunoglobulin molecules 6, 7, 8.
Immunoglobulin types
There are five classes of immunoglobulins and several subclasses. Each class represents a group of antibodies and has a slightly different role.
Immunoglobulin A
Immunoglobulin A or IgA is found in high concentrations in your body’s mucous membranes, particularly of the respiratory passages (the organs you use to breathe) and gastrointestinal tract (the organs you use to eat and digest food), as well as in saliva and tears 1. Immunoglobulin A or IgA is the major antibody in secretions found in saliva, tears, colostrum, intestinal, genital tract, and respiratory secretions. Immunoglobulin A (IgA) also plays a role in allergic reactions. IgA levels also may be high in autoimmune conditions, disorders in which the body mistakenly makes antibodies against healthy tissues.
Immunoglobulin A (IgA) appears in 2 different molecular structures: monomeric (serum) and dimeric structure (secretory). The serum immunoglobulins A (IgA) has a molecular weight of 160 Kd and a serum concentration of 3 mg/mL 1. Secretory IgA (sIgA) has a molecular weight of 385 Kd and a mean serum concentration of 0.05 mg/mL 1. It appears in mucosa membranes as a dimer (with J chain when secreted) and protects the epithelial surfaces of the respiratory, digestive, and genitourinary system. Immunoglobulins A (IgA) possesses a secretory component that prevents its enzymatic digestion. It activates the alternative pathway of activation of the complement system 9.
Immunoglobulin D
Immunoglobulin D or IgD is a monomer with a molecular weight of 184 Kd and is present in small amounts in serum (0.03 mg/mL). Immunoglobulin D or IgD may play an essential role in antigen-triggered lymphocyte differentiation 10. Immunoglobulin D (IgD) is regarded as B-cell receptor (BCR) on the cell surface of B lymphocytes 11. Immunoglobulin D (IgD) may play an essential role in antigen-triggered lymphocyte differentiation 12, 13.
Immunoglobulin E
Immunoglobulin E or IgE is present primarily in the skin and mucous membranes and is believed to function in response to environmental antigens. Immunoglobulin E or IgE protects against parasites and binds to high-affinity receptors on mast cells and basophils, causing allergic reactions 14, 15, 16. IgE is considered the most important host defense against different parasitic infections, including Strongyloides stercoralis, Trichinella spiralis, Ascaris lumbricoides, and hookworms Necator americanus and Ancylostoma duodenale.
Immunoglobulin E (IgE) is a monomer. Immunoglobulins E (IgE) has a molecular weight of 188 Kd and a serum concentration of 0.00005 mg/mL.
Immunoglobulin G
Immunoglobulin G or IgG is widespread in the body and it provides long-lasting defense against most bacterial invasions and other antigens. Immunoglobulin G or IgG is the only immunoglobulin that crosses the placenta as its Fc portion binds to the receptors present on the surface of the placenta, protecting the neonate from infectious diseases 17. IgG is therfore the most abundant antibody present in newborns.
Immunoglobulin G (IgG) is a monomer with an approximate molecular weight of 146 Kd and a serum concentration of 9.0 mg/mL. Immunoglobulin G or IgG is said to be divalent, i.e., it has two identical antigen-binding sites that comprise 2 L chains and 2 H chains joined by disulfide bonds. Immunoglobulins G (IgG) is synthesized mostly in the secondary immune response to pathogens. Immunoglobulin G (IgG) can activate the classical pathway of the complement system, and it also is highly protective. The four subclasses of immunoglobulin G (IgG) include IgG1, IgG2, IgG3, and IgG4. IgG1 is around 65% of the total IgG. IgG2 forms an important host defense against bacteria that are encapsulated.
Immunoglobulin M
Immunoglobulin M or IgM is a large molecule, immunoglobulin M (IgM) is found in blood and is involved in combating blood infections. Immunoglobulin M (IgM) is the first or primary immunoglobulin produced following exposure to an antigen. Immunoglobulin M (IgM) is mainly produced in the primary immune response to infectious agents or antigens. Immunoglobulin M (IgM) is regarded as a potent agglutinin (e.g., anti-A and anti-B isoagglutinin present in type B and type A blood respectively) and a monomer of immunoglobulin M (IgM) is used as a B cell receptor 18.
Immunoglobulin M (IgM) has a molecular weight of 970 Kd and average serum concentration of 1.5 mg/ml. Immunoglobulin M (IgM) is a pentamer and activates the classical pathway of the complement system. A monomer of IgM is used as a B cell receptor (BCR) 19.
Immunoglobulin test
Immunoglobulins testing measures the total amount of each primary immunoglobulin class, IgA, IgM, and IgG in the blood and, in certain circumstances, in cerebrospinal fluid (CSF) or saliva, without distinguishing between subclasses. Separate testing can be performed to measure immunoglobulin subclasses and/or to detect and measure specific antibodies.
A variety of conditions can cause an increase (hypergammaglobulinemia) or decrease (hypogammaglobulinemia) in the production of immunoglobulins. Some cause an excess or deficiency of all classes of immunoglobulins while others affect only one class. Some of the conditions are passed from one generation to the next (inherited) and others are acquired.
How is immunoglobulins test used?
A test for immunoglobulins (Igs) is used to detect an excess or deficiency in the three major classes of immunoglobulins (IgG, IgA, and IgM). It gives important information about the health of an individual’s immune system and is used to help diagnose various conditions and diseases that affect the levels of one or more of these immunoglobulins classes.
In general, immunoglobulin disorders can be classified as:
- Immunoglobulin excess
- Polyclonal—an immunoglobulin excess in any or all immunoglobulin classes from many different immune (plasma) cells
- Monoclonal—the excess immunoglobulins are from the clones of one plasma cell
- Immunoglobulin deficiency
- Secondary (acquired) immunoglobulin deficiency—the most common Ig deficiencies are caused by an underlying condition or contributing factor
- Primary (inherited) immunoglobulin deficiency—these are rare disorders in which the body is not able to produce one or more classes of immunoglobulins
Immunoglobulin test may be ordered along with others, such as a serum and/or urine protein electrophoresis, to help diagnose and monitor conditions associated with abnormal or excessive immunoglobulin production. When this is the case, a urine sample may be collected in addition to blood.
If an excessive amount of one of the immunoglobulin types is present, further testing by immunofixation can be done to determine if the immunoglobulin comes from clones of an abnormal plasma cell (monoclonal gammopathy). Monoclonal gammopathies are seen with multiple myeloma, a malignancy of plasma cells. Serum free light chain testing may also be performed.
In addition to the blood sample for this test, I was instructed to collect a urine sample. Why?
You may have been directed to give a urine sample to check for the presence of protein in your urine and/or to determine the amount and type of protein present in your urine. Your healthcare practitioner may suspect that, based on your medical history, signs and symptoms and/or other test results, you have a condition associated with abnormal or excessive immunoglobulin production. In such cases, a quantitative immunoglobulin test is often performed along with tests such as serum and urine protein electrophoresis to help establish a diagnosis.
When is immunoglobulin test ordered?
Immunoglobulin test is ordered when a person has symptoms of an immunoglobulin deficiency such as recurrent infections, especially of the respiratory tract (sinus, lungs) or digestive system (stomach, intestines), and/or chronic diarrhea.
Immunoglobulins testing may also be ordered when a person has signs of chronic inflammation or chronic infection and when a healthcare practitioner suspects excess or abnormal immunoglobulin production. The test may be ordered periodically to monitor the course of a person’s condition.
This test may also be performed on cerebrospinal fluid (CSF) whenever a healthcare practitioner suspects that a condition affecting the central nervous system may be associated with excess immunoglobulin production.
My newborn had an immunoglobulin test. Why?
Sometimes an immunoglobulins M (IgM) test is used to determine whether a newborn acquired an infection before birth (congenital). IgM may be produced by a developing baby (fetus) in response to infection. Due to the size of IgM antibodies, they cannot pass through the placenta from mother to baby during pregnancy. Thus, any IgM antibodies present in a newborn’s blood are not from the mother but were produced by the baby. This indicates that an infection began during pregnancy.
What does the immunoglobulin test result mean?
The results of the immunoglobulin tests for immunoglobulins G (IgG), immunoglobulins A (IgA), and immunoglobulins M (IgM) levels are usually evaluated together. Abnormal test results typically indicate that there is something affecting the immune system and suggest the need for further testing. Immunoglobulins testing is not diagnostic but can be a strong indicator of a disease or condition. There are a number of conditions that are associated with increased and decreased immunoglobulins.
High immunoglobulin levels
Increased polyclonal immunoglobulins may be seen with a variety of conditions.
Monoclonal immunoglobulins are seen in blood cell tumors that involve lymphocytes or plasma cells. In these disorders, there is typically a marked increase in one class of immunoglobulin and a decrease in the other two classes. Although affected people may have an increase in total immunoglobulins, they are actually immunocompromised because most of the immunoglobulins produced are abnormal and do not contribute to the immune response.
The following table lists some examples of conditions that may cause increased immunoglobulins.
Table 1. High immunoglobulin levels causes
| Immunoglobulin Result | Associated Conditions |
|---|---|
| Polyclonal increase in any or all of the three classes (IgG, IgA and/or IgM) |
|
| Monoclonal increase in one class with or without decrease in other two classes |
|
Low immunoglobulin levels
The most common causes of decreased immunoglobulins are acquired underlying (secondary) conditions that either affect the body’s ability to produce immunoglobulins or that increase the loss of protein from the body. Deficiencies may also be due to drugs such as immunosuppressants, corticosteroids, phenytoin, and carbamazepine or due to toxins.
Inherited immune deficiencies are rare and are often referred to as primary immunodeficiencies. They may affect the production of all immunoglobulins, a single class, or one or more subclasses. Some of these disorders include agammaglobulinemia, common variable immunodeficiency (CVID), x-linked agammaglobulinemia, ataxia telangiectasia, Wiskott-Aldrich syndrome, hyper-IgM syndrome, and severe combined immunodeficiency (SCID).
In CSF, immunoglobulins normally are present in very low concentrations. Increases may be seen, for example, with central nervous system infections (meningitis, encephalitis), inflammatory conditions, and multiple sclerosis.
Decreases in salivary IgA may be seen in those with recurrent respiratory infections.
People with conditions that cause decreased immunoglobulin levels often do not have a strong immune response to vaccinations; they may not produce a sufficient level of antibody to ensure protection and may not be able to receive live vaccines, such as those for polio or measles.
Many laboratory tests measure antibodies in the blood. Those with immunoglobulin deficiencies may have false-negative results on these types of tests. For example, one test for celiac disease detects the IgA class of anti-tissue transglutaminase antibody (anti-tTG). If a person has a deficiency in IgA, then results of this test may be negative when the person, in fact, has celiac disease. If this is suspected to be the case, then a quantitative test for IgA may be performed.
If IgG or IgA concentrations are decreased, or a deficiency in one of their subclasses is suspected, then subclass testing may be performed to detect and further define the deficiency. Subclass deficiencies can be present even when an immunoglobulin class concentration, such as IgG, is normal.
Some people with IgA deficiencies may develop anti-IgA antibodies. When those with anti-IgA are given blood component transfusions that contain IgA (such as plasma or immunoglobulin treatments), they may experience a severe anaphylactic transfusion reaction.
The table below lists some of the common causes of low levels.
Table 2. Low immunoglobulin levels causes
| Conditions/factors that affect immunoglobulin production |
|
| Conditions that cause an abnormal loss of protein |
|
What is transient hypogammaglobulinemia of infancy?
Infants with otherwise normal immune systems may have temporarily decreased IgG levels when production is delayed. Protection from infections is lost as concentrations of the mother’s IgG in the baby’s blood decrease over several months. The level of IgG remains at low concentrations until the baby’s IgM and IgG production ramps up. This creates a period of time during which the baby is at an increased risk for recurrent infections.
However, infants who are breastfed acquire IgA from breast milk. The IgA in breast milk can be protective against infections, particularly in the time between the decrease of mother’s antibodies and the production of the baby’s own antibodies.
Are there symptoms associated with decreased immunoglobulins?
Not specific ones. Unexplained recurrent infections, multiple infections, or opportunistic infections, with or without chronic diarrhea, may indicate a need to check a person’s immune status. A positive family history of an immunodeficiency may also require follow up. A thorough physical examination and a careful medical history can be critical to a diagnosis.
Is there something I can do to alter my immunoglobulin levels?
In most cases, immunoglobulins do not respond to lifestyle changes. If you are taking a drug that is decreasing one or more of your immunoglobulins, then you and your healthcare practitioner may decide to alter your medications. It is very important, however, NOT to discontinue or change your medication dosage without consulting with your healthcare provider.
Infants gradually lose protection from infections as the levels of IgG they receive through the placenta from their mothers decrease after birth. Greater protection can be provided for babies through breastfeeding since breast milk contains IgA, which protects against infections.
Immunoglobulin deficiency
Immunoglobulins or antibodies play an essential role in the protection against bacteria, viruses, and fungi. When there is a deficiency of these glycoproteins, recurrent infectious diseases occur as seen in the following antibody deficiency disorders 20:
- X-linked agammaglobulinemia
- Transient hypogammaglobulinemia of infancy
- IgA deficiency
- IgG subclass deficiency
- Immunodeficiency with increased IgM
- Common variable immunodeficiency
The most common immunodeficiency is Selective IgA deficiency, characterized by recurrent infections that affect the respiratory, digestive, and genitourinary systems. Recurrent pneumonia, Giardia lamblia infestation, and urinary sepsis are prevalent. The majority of patients can, however, be asymptomatic. They are at higher risks for autoimmune diseases, atopy, and anaphylaxis to IgA-containing products 21.
Another common problem is the transient hypogammaglobulinemia of infancy. During the first 3 to 5 months the child is healthy, but he becomes sick because of a physiological deficit of immunoglobulins. This disease is characterized by recurrent bacterial infections including pneumonia, meningitis, otitis, arthritis, osteomyelitis, among others. This problem heals once the child starts producing immunoglobulins 22.
X-linked agammaglobulinemia is also called Bruton agammaglobulinemia. It occurs due to a defect in Bruton Tyrosine Kinase (BTK) gene that prevents B-cell maturation. This condition is X-linked recessive and seen mostly in males. They present with recurrent bacterial and enteroviral infections after 6 months, once the maternal IgG is low. No B cells are seen in peripheral blood and immunoglobulins of all classes are absent. Patients also have absent or scanty lymph nodes and tonsils. Live vaccines are contraindicated 23..
In common variable immunodeficiency (CVID), individuals acquire the immunodeficiency in the second or third decade of life or later. Both males and females can develop this problem. Common variable immunodeficiency (CVID) may follow a viral infection, such as infectious mononucleosis. Giardia lamblia infestation and recurrent pyogenic infections characterize common variable immunodeficiency (CVID). It may be due to a defect in B-cell differentiation. The patients have a risk of autoimmune disease, bronchiectasis, lymphoma, and sinopulmonary infections.
Medications and treatments for immune deficiencies include:
- Antimicrobial therapy to fight and prevent infections
- Immune globulin replacement therapy
- Vaccinations
- Specialized immune globulins
- Hematopoietic cell transplant
- Gene therapy
- Enzyme replacement therapy
- Biologics
Immunoglobulin Replacement Therapy
People who are unable to produce adequate amounts or functional immunoglobulins or antibodies may benefit from replacement therapy with gamma globulins. There are a number of immunoglobulins: IgG, IgA, IgM and IgE. Antibody replacement with gamma globulin replaces IgG in the blood. This can be given both intravenously (IVIG) or under the skin, called subcutaneously (SCIG.)
Gamma globulin is made of pooled antibodies from the blood of healthy people. Antibodies from at least 1,000 donors in each treatment provide protection against a wide variety of foreign invaders. The blood is carefully tested, and discarded if there is evidence of contagious diseases such as hepatitis or HIV. Talk with your doctor if you have any questions or concerns.
Because supplemental IgG therapy (IVIG or SCIG) does not contain IgA or IgM, their protective functions are not replaced. People with immune deficiencies may continue to have trouble with certain types of infections despite supplemental IgG therapy. However, treatment with supplemental IgG and early treatment of infections help many people with immune deficiencies lead active and full lives.
In cases of slightly low immunoglobulin (IgG) where antibody production is intact, watchful waiting is encouraged. Infants with transient hypogammaglobulinemia often have resolution of this finding without intervention. Some individuals will have low immunoglobulin without disruption of ability to produce antibody, and require no intervention.
Replacement therapy with immunoglobulin G (IgG), administered intravenously (IVIG) or subcutaneously (SCIG), is the treatment of choice for most primary immunodeficiency syndromes where very low immunoglobulin is a feature, including the following:
- X-linked agammaglobulinemia (Bruton disease; XLA)
- Common variable immune deficiency (CVID)
- Severe combined immunodeficiency (SCID) prior to stem cell or bone marrow transplantation
- Hyper-IgM
- ADA deficiency
- Wiskott-Aldrich syndrome (WAS)
- Syndromes associated with low immunoglobulin or poor antibody production
- Sometimes specific antibody deficiency
If poor T-cell function is also a part of the immune deficiency (ie, severe combined immune deficiency or combined immune deficiency), stem cell transplant or bone marrow transplant may be the definitive treatment, and may replace B cell function so that IgG replacement is no longer necessary 24.
Treatment of secondary hypogammaglobulinemia is directed at the underlying cause, as follows:
- Intravenous immunoglobulin (IVIg) is not indicated for lymphoproliferative disorders unless immunoglobulin levels are low in association with recurrent infections or if IVIG is being used for autoimmune conditions that may accompany these disorders
- If IgG is being lost through the gut or kidney, replacement of IgG will not be effective
Specific Immune Globulins
Pathogen-specific immune globulins may be used in some immunocompromised people for prophylaxis against potentially life-threatening infection from the offending pathogen. Monoclonal antibody treatment against respiratory syncytial virus (RSV) is used to prevent RSV infection in high-risk populations. Specific immune globulins are also used to prevent or treat varicella infections and cytomegalovirus infections.
Antimicrobial therapy
Antimicrobials, such as antibiotics and antifungals, are medications that fight bacterial or fungal infections. They are used when signs of infection are present. It is often difficult to determine whether a pulmonary or sinus infection is caused by bacteria, as viral infections can present with similar signs and symptoms. In these cases, your doctor may perform additional testing, such as imaging (X-ray or CT scan), or obtain cultures of secretions from the infected area to determine the nature of the infectious agent. Cultures can also provide sensitivities of that particular organism to determine the most effective antimicrobial agent.
Antimicrobial therapy may also be used to prevent infections, and may be added as a routine medication that is taken as prophylaxis. Talk with your doctor about the potential side effects of antimicrobials.
Other Treatments
Other treatments are available for some types of immune deficiencies. Bone marrow transplants, hematopoietic cell transplantation, thymus transplantation, gene therapy, enzyme replacement and biologic cytokine inhibitors are examples of other treatments. Researchers in immunology are experimenting with gene therapy and other treatments that will be available in the future.
Living with immunoglobulin deficiency
People living with immune deficiency can lead active and full lives. The goal of treating an immune deficiency is to help you regain or maintain control of your life. Sometimes a chronic illness can be hard on the whole family. If your immune deficiency is causing family problems, be sure to talk with someone. Your doctor can help you find an expert to talk with. There also may be support groups where you can meet and talk to other people with an immune deficiency. Having an immune deficiency may require treatment with medications that require approval from your insurance company. If you are having problems getting proper coverage for these medications, it is important to tell your doctor. Your doctor may be able to talk with your insurance company and fix these insurance problems. Your doctor is your partner in taking care of your immune deficiency.
For many people this may include:
- Participating in work, school, family and social activities
- Decreasing the number and severity of infections
- Having few, if any, side effects from medications and other treatments
- Feeling good about yourself and your treatment program
Your doctor may prescribe medications or other treatments to make you feel better and to protect you from foreign invaders. Medications and treatments must be chosen for your individual needs and may be adjusted as your needs change.
Preventing Infections
There are things you can do to prevent infection. They include:
- Stay away from people who have a cold or other infection.
- Wash your hands with soap before eating, after outings and after using the bathroom.
- Although hand-washing seems simple, make sure it is effective. Use liquid soap and scrub your hands using plenty of lather for 10-15 seconds. Rinse your hands and dry them completely.
- If a sink is not available, use alcohol-based hand sanitizer over the entire surface of your hands until dry.
- Clean cuts and scrapes right away with warm, soapy water. Apply antibacterial cream or ointment and cover the cut or scrape with a fresh bandage at least daily. Tell your doctor if any redness or drainage develops.
- Brush your teeth after meals, and floss at least daily. Have a dental exam every six months. Tooth decay and gum disease are types of infection.
- There are vaccines available for some common viruses (like the flu) and bacteria (like certain types of pneumonia) that you may want to consider. Getting vaccinated may lessen the severity of symptoms or prevent these infections altogether. Talk to your doctor about whether you should get a flu, pneumonia or other vaccine.
If you notice any of the following, you may have an infection:
- Fever
- Chills
- Nasal congestion or discharge
- Cough
- Diarrhea
- Vomiting
- Tenderness or pain
- Unusual discharge
- Swelling or redness on the skin
- If you suspect you have an infection of any kind, call your doctor right away.
Healthy Diet
A healthy diet boosts the immune system’s ability to fight infection. Actions you can take to help strengthen the immune system include:
- Maintain a healthy weight.
- Eat a variety of foods daily, including grains, fruits, vegetables, lean meats and dairy products.
- Take a multivitamin supplement containing 100 percent of the Recommended Daily Allowance (RDA). RDAs are the minimum amounts of nutrients that a healthy person requires each day.
- Your doctor or registered dietitian can help recommend a healthy diet for you.
Enough Rest
Adequate sleep and rest give your body and your immune system time to maintain and repair themselves. Quiet activities like reading or crafts are helpful during times of rest or illness.
Regular Exercise
Regular exercise impacts your overall fitness by improving heart and muscle function. Most people who exercise regularly also feel an increased sense of well-being. Walking, swimming and biking are only some of the activities people with immune deficiencies can do. Children should be encouraged to participate in safe, age-appropriate activities. Talk with your doctor about recommending an exercise program for you.
Immunoglobulin therapy
Immunoglobulins or antibodies can be used as a form of immunotherapy. Like drugs, immunoglobulins are prepared from a pool of blood donated at blood collection centers and processed through fractionation to separate the protein fraction from the cellular component. The purified immunoglobulin to some various degrees of purity can be used in the treatment of many immunological problems, including antibody deficiencies, severe combined immunodeficiency disorders (SCID), multiple sclerosis, myasthenia gravis, Kawasaki disease, systemic lupus erythematosus (SLE), organ transplantations, and many others 25. Some people have frequent, severe infections because their bodies don’t make enough immunoglobulins (antibodies). Antibodies are proteins that the body produces to fight off harmful substances. For people who don’t produce enough immunoglobulins (antibodies), immunoglobulin replacement therapy can be a lifesaver. But other people get the treatment even though they don’t need it. Here’s what you need to know.
Immunoglobulin treatment can help people with primary immunodeficiency disease. Primary immunodeficiency disease (PIDD) usually appears in childhood, but can also appear in adulthood. It increases the risk of infections, like pneumonia and sinusitis. At least one person in 1,200 in the U.S. has primary immunodeficiency disease. People with primary immunodeficiency disease lack antibodies, especially immunoglobulins G (IgG).
Immunoglobulins G (IgG) antibodies are the body’s main defense against bacterial infection. Immunoglobulins G (IgG) treatment replaces these antibodies. It can stop or prevent most of their infections, but it doesn’t help most people with frequent infections.
Immunoglobulins G (IgG) only helps people who lack the IgG antibody.
- It won’t help you if your immune system already makes enough antibodies.
- It isn’t helpful for every form of primary immunodeficiency disease. It won’t help people who lack IgA, a different antibody.
- It’s usually not needed if a person just has low IgG blood levels. Often, they still have enough antibodies to prevent infections or control them with the help of antibiotics.
Immunoglobulins G (IgG) therapy has risks
Immunoglobulins G (IgG) antibodies are injected into a vein or under the skin. Both methods can cause side effects, although they are more common with vein injections. Side effects can include serious allergic reactions, kidney failure, and headache and flu-like symptoms. In rare cases, patients with severe headaches can have irritation of the lining around the brain. There can also be swelling, bruising, or infection at the injection site. Finally, the medicine is made from human blood, so it has a very small risk of carrying viruses.
Immunoglobulins G (IgG) treatment can be costly
The cost depends on your dose and body weight, and the injection method. Treatment can cost more than $30,000 a year. It must be repeated regularly, usually for life.
When is Immunoglobulins G (IgG) treatment a good idea?
If you have severe, frequent, unusual, or persistent infections, talk to your doctor. Your doctor can order a series of tests to see if immunoglobulins G (IgG) therapy might help you. First, you should have a blood test to check your antibody levels. Then, you will get one or more vaccines and another antibody test. If your body doesn’t make antibodies in response to vaccination, immunoglobulins G (IgG) treatment might be a good idea. It will help your doctor decide if IgG treatment may help reduce the frequency or severity of infections.
Immunoglobulins G (IgG) treatment may also be helpful if:
- You have very low blood levels of IgG.
- You are diagnosed with certain immune disorders.
- Justiz Vaillant AA, Jamal Z, Patel P, et al. Immunoglobulin. [Updated 2023 May 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513460[↩][↩][↩][↩][↩]
- Cooper MD. The early history of B cells. Nat Rev Immunol. 2015 Mar;15(3):191-7. doi: 10.1038/nri3801[↩]
- Burton DR. Antibody: the flexible adaptor molecule. Trends Biochem. Sci. 1990 Feb;15(2):64-9.[↩]
- Burton DR. Antibody: the flexible adaptor molecule. Trends Biochem Sci. 1990 Feb;15(2):64-9. doi: 10.1016/0968-0004(90)90178-e[↩]
- Engels N, Wienands J. Memory control by the B cell antigen receptor. Immunol Rev. 2018 May;283(1):150-160. doi: 10.1111/imr.12651[↩]
- Wienands J, Engels N. Control of memory B cell responses by extrinsic and intrinsic mechanisms. Immunol Lett. 2016 Oct;178:27-30. doi: 10.1016/j.imlet.2016.05.010[↩]
- Lewis SM. Evolution of immunoglobulin and T-cell receptor gene assembly. Ann N Y Acad Sci. 1999 May 18;870:58-67. doi: 10.1111/j.1749-6632.1999.tb08865.x[↩]
- Lewis SM, Wu GE. The origins of V(D)J recombination. Cell. 1997 Jan 24;88(2):159-62. doi: 10.1016/s0092-8674(00)81833-4[↩]
- Daha MR, van Kooten C. Role of complement in IgA nephropathy. J Nephrol. 2016 Feb;29(1):1-4. doi: 10.1007/s40620-015-0245-6[↩]
- Goding JW. Allotypes of IgM and IgD receptors in the mouse: a probe for lymphocyte differentiation. Contemp Top Immunobiol. 1978;8:203-43. doi: 10.1007/978-1-4684-0922-2_7[↩]
- Gutzeit C, Chen K, Cerutti A. The enigmatic function of IgD: some answers at last. Eur J Immunol. 2018 Jul;48(7):1101-1113. doi: 10.1002/eji.201646547[↩]
- Chen K, Cerutti A. New insights into the enigma of immunoglobulin D. Immunol Rev. 2010 Sep;237(1):160-79. doi: 10.1111/j.1600-065X.2010.00929.x[↩]
- Blattner FR, Tucker PW. The molecular biology of immunoglobulin D. Nature. 1984 Feb 2-8;307(5950):417-22. doi: 10.1038/307417a0[↩]
- Maurer M, Altrichter S, Schmetzer O, Scheffel J, Church MK, Metz M. Immunoglobulin E-Mediated Autoimmunity. Front Immunol. 2018 Apr 9;9:689. doi: 10.3389/fimmu.2018.00689[↩]
- Mukai K, Tsai M, Starkl P, Marichal T, Galli SJ. IgE and mast cells in host defense against parasites and venoms. Semin Immunopathol. 2016 Sep;38(5):581-603. doi: 10.1007/s00281-016-0565-1[↩]
- Schwartz C, Eberle JU, Voehringer D. Basophils in inflammation. Eur J Pharmacol. 2016 May 5;778:90-5. doi: 10.1016/j.ejphar.2015.04.049[↩]
- Palmeira P, Quinello C, Silveira-Lessa AL, Zago CA, Carneiro-Sampaio M. IgG placental transfer in healthy and pathological pregnancies. Clin Dev Immunol. 2012;2012:985646. doi: 10.1155/2012/985646[↩]
- Vasilev N, Smales CM, Schillberg S, Fischer R, Schiermeyer A. Developments in the production of mucosal antibodies in plants. Biotechnol. Adv. 2016 Mar-Apr;34(2):77-87[↩]
- Vasilev N, Smales CM, Schillberg S, Fischer R, Schiermeyer A. Developments in the production of mucosal antibodies in plants. Biotechnol Adv. 2016 Mar-Apr;34(2):77-87. doi: 10.1016/j.biotechadv.2015.11.002[↩]
- Justiz Vaillant AA, Ramphul K. StatPearls [Internet]. StatPearls Publishing; Treasure Island (FL): Feb 22, 2019. Antibody Deficiency Disorder[↩]
- Stonebraker JS, Farrugia A, Gathmann B, ESID Registry Working Party. Orange JS. Modeling primary immunodeficiency disease epidemiology and its treatment to estimate latent therapeutic demand for immunoglobulin. J. Clin. Immunol. 2014 Feb;34(2):233-44[↩]
- Justiz Vaillant AA, Qurie A. StatPearls [Internet]. StatPearls Publishing; Treasure Island (FL): Feb 22, 2019. Immunodeficiency[↩]
- Taneja A, Chhabra A. StatPearls [Internet]. StatPearls Publishing; Treasure Island (FL): Jan 1, 2019. Bruton Agammaglobulinemia.[↩]
- Ochs HD, Smith CIE, Puck JM. Primary Immunodeficiency Diseases: A Molecular & Cellular Approach. 2nd ed. USA: Oxford University Press; 2006.[↩]
- Vince N, Mouillot G, Malphettes M, Limou S, Boutboul D, Guignet A, Bertrand V, Pellet P, Gourraud PA, Debré P, Oksenhendler E, Théodorou I, Fieschi C., DEFI Study Group. Genetic screening of male patients with primary hypogammaglobulinemia can guide diagnosis and clinical management. Hum. Immunol. 2018 Jul;79(7):571-577[↩]