Contents
- Infant respiratory distress syndrome
- What is surfactant?
- What does surfactant do?
- Who is affected by infant respiratory distress syndrome?
- How long does infant respiratory distress syndrome last?
- How can I tell if my baby is getting better?
- What factors determine how infant respiratory distress syndrome progresses?
- Are there long term effects from infant respiratory distress syndrome?
- Infant respiratory distress syndrome causes
- Infant respiratory distress syndrome prevention
- Infant respiratory distress syndrome symptoms
- Infant respiratory distress syndrome complications
- Infant respiratory distress syndrome diagnosis
- Infant respiratory distress syndrome differential diagnosis
- Infant respiratory distress syndrome treatment
- Infant respiratory distress syndrome prognosis
Infant respiratory distress syndrome
Infant respiratory distress syndrome (IRDS) also known as hyaline membrane disease (HMD), respiratory distress syndrome (RDS), neonatal respiratory distress syndrome, or surfactant deficiency lung disease, is a serious lung condition that sometimes affects premature babies who are born six weeks or more before their due dates, caused by a lack of surfactant, a substance that coats the inside of baby’s lungs and keeps them open so that the baby can breathe in air once he or she is born 1, 2, 3. Infant respiratory distress syndrome is uncommon after 36 weeks gestation due to the development of surfactant production around 35 weeks 4. Less often, infant respiratory distress syndrome can affect full-term newborns in the setting of maternal diabetes (mother with diabetes while pregnant) because of high insulin levels, which interfere with surfactant production maturation. Other risk factors for infant respiratory distress syndrome (IRDS) include multiple fetuses (such as twins, triplets, or quadruplets), cesarean section delivery, and perinatal asphyxia 5, 6, 7, 8. Historically, respiratory distress syndrome or infant respiratory distress syndrome (IRDS) was known as hyaline membrane disease (HMD) due to the appearance of hyaline membranes lining the air sacs in the lungs of affected newborns 3.
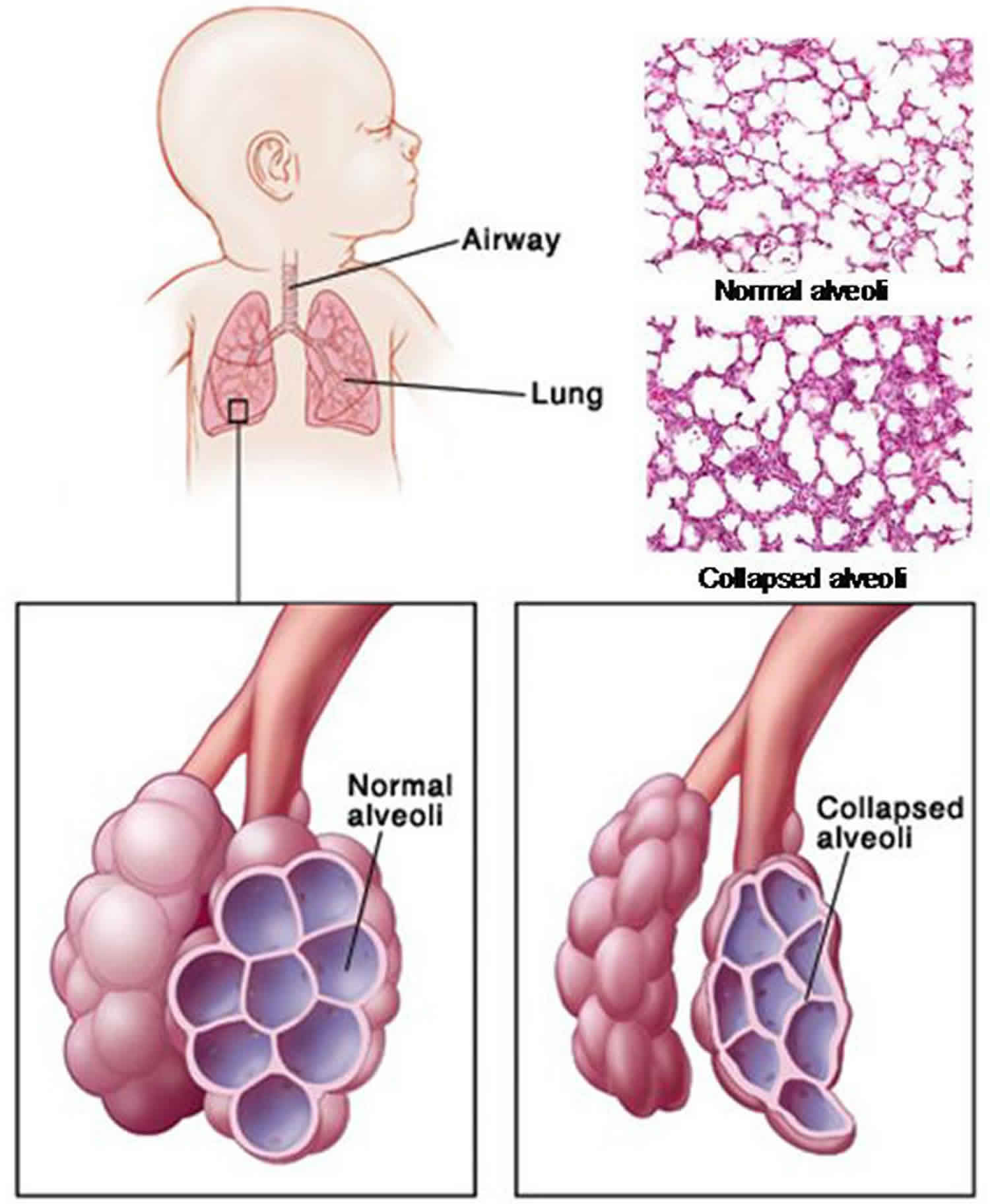
After birth, the baby’s lungs may take up to 5 days to produce surfactant. Without surfactant in infant respiratory distress syndrome (IRDS), the alveoli, or air sacs collapsed, thickened and become inflamed, making it hard for your baby to breathe. This reduces the amount of oxygen-rich blood from reaching your baby’s brain and other tissues in the body. Major symptoms of infant respiratory distress syndrome (IRDS) include difficulty in breathing and collapsed lungs, potentially requiring mechanical ventilation or positive end-expiratory pressure (PEEP). Fortunately, with proper treatment most babies who have infant respiratory distress syndrome (IRDS) survive and these babies may need extra medical care after going home. However, some babies have complications from infant respiratory distress syndrome (IRDS) or its treatment, including the development of another serious lung condition called bronchopulmonary dysplasia (BPD) also known as chronic lung disease of infancy.
Infant respiratory distress syndrome (IRDS) is different from bronchopulmonary dysplasia (BPD), another serious lung condition that also affects premature babies that need oxygen therapy or mechanical ventilation, leading to lung damage and breathing difficulties. While infant respiratory distress syndrome (IRDS) usually develops in the first 24 hours after birth, bronchopulmonary dysplasia (BPD) usually develops within the next week or two. Babies are not born with bronchopulmonary dysplasia (BPD). Instead, they develop bronchopulmonary dysplasia (BPD) as a complication of another breathing condition. For example, most babies who develop bronchopulmonary dysplasia (BPD) are born with respiratory distress syndrome (RDS). However, other types of newborn breathing conditions, infections, and lung injuries from treatments like mechanical ventilation can also lead to bronchopulmonary dysplasia (BPD). Newborns are diagnosed with bronchopulmonary dysplasia (BPD) if they still need breathing support 28 days after birth, or around the time they would have reached their original due date.
Infant respiratory distress syndrome (IRDS) or respiratory distress syndrome (RDS) is caused by surfactant deficiency, especially in the context of immature lungs. Infant respiratory distress syndrome (IRDS) or respiratory distress syndrome (RDS) is most common in premature infants who are 30 weeks gestation or younger. Because a baby’s lungs are not needed for breathing until after birth, they are one of the last organs to fully develop. The more premature the birth, the less able your baby’s lungs are to function well. Infant respiratory distress syndrome (IRDS) incidence is estimated at 6 in 1000 births and about 24,000 infants born in the United States annually 3, 6. The incidence of infant respiratory distress syndrome increases with decreasing gestational age at birth. In one study of babies born between 2003 and 2007 at various National Institute of Child Health and Human Development (NICHD) Neonatal Research Network centers, 98% of babies born at 24 weeks had infant respiratory distress syndrome, while at 34 weeks the incidence was 5%, and at 37 weeks was less than 1% 9. Approximately 50% of the neonates born at 26-28 weeks’ gestation develop respiratory distress syndrome, whereas less than 30% of premature neonates born at 30-31 weeks’ gestation develop the condition 10. In one report, the incidence rate of respiratory distress syndrome was 42% in infants weighing 501-1500g, with 71% reported in infants weighing 501-750g, 54% reported in infants weighing 751-1000g, 36% reported in infants weighing 1001-1250g, and 22% reported in infants weighing 1251-1500g, among the 12 university hospitals participating in the National Institute of Child Health and Human Development (NICHD) Neonatal Research Network 11.
Infant respiratory distress syndrome diagnosis is typically based on clinical signs, oxygen levels in the blood, and chest X-ray results. The chest X-ray findings of untreated surfactant deficiency lung disease depend on the severity of disease and may range from fine granular pulmonary opacities to complete white-out of the lungs 4. Low lung volumes are classically described in untreated surfactant deficiency lung disease and occur as a result of impaired alveolar expansion 4.
Treatment involves supplemental oxygen, continuous positive airway pressure (CPAP) to keep the air sacs open, and in some cases, mechanical ventilation and artificial surfactant. The current preferred treatment strategy for neonatal respiratory distress syndrome is the early initiation of continuous positive airway pressure (CPAP) with selective surfactant administration 12. In most institutions, continuous positive airway pressure (CPAP) is preferred over invasive ventilation as they decrease the risk of mortality, and bronchopulmonary dysplasia (BPD) compared to invasive ventilation with or without surfactant 13, 14.
The American Academy of Pediatrics 2014 guidelines recommend immediate management of all preterm infants with nasal continuous positive airway pressure (nCPAP) 15. Subsequently, surfactant administration may be considered an alternative to intubation with prophylactic or early surfactant administration in selected patients. The first dose is given within two hours of birth, preferably within the first hour. In an intubated infant that requires 30-40% oxygen, a repeat dose is indicated within the next 4 to 12 hours. Surfactant replacement therapy is administered by trained personnel in a clinical setting where equipment for intubation and resuscitation are readily available.
Treatment for newborn respiratory distress syndrome may include:
- Placing an endotracheal tube (breathing tube, also called an ET) into your baby’s windpipe
- Mechanical breathing machine to do the work of breathing for your baby.
- Supplemental oxygen (extra amounts of oxygen)
- Continuous positive airway pressure (CPAP) – a mechanical breathing machine that pushes a continuous flow of air or oxygen to the airways to help keep tiny air passages in the lungs open
- Surfactant replacement with artificial surfactant – this treatment has been shown to reduce the severity of newborn respiratory distress syndrome, and is most effective if started in the first six hours of birth. It may be given as preventive treatment for babies at very high risk for newborn respiratory distress syndrome, or used as a “rescue” method. The drug comes as a powder that is mixed with sterile water and given through the endotracheal tube tube. This treatment is usually administered in several doses.
- Medications to help sedate and ease your baby’s pain during treatment.
Infant respiratory distress syndrome key points:
- Newborn respiratory distress syndrome is one of the most common problems seen in premature babies.
- The more premature the baby, the higher the risk and the more severe the newborn respiratory distress syndrome.
- Newborn respiratory distress syndrome typically worsens over the first 48 to 72 hours and then improves with treatment.
- More than 90 percent of babies with newborn respiratory distress syndrome survive.
- The best way of preventing newborn respiratory distress syndrome is by preventing a preterm birth. When a preterm birth cannot be prevented, giving the mother medications called corticosteroids before delivery has been shown to dramatically lower the risk and severity of newborn respiratory distress syndrome in the baby. These steroids are often given to women between 24 and 34 weeks gestation who are at risk of early delivery.
Figure 1. Newborn respiratory system
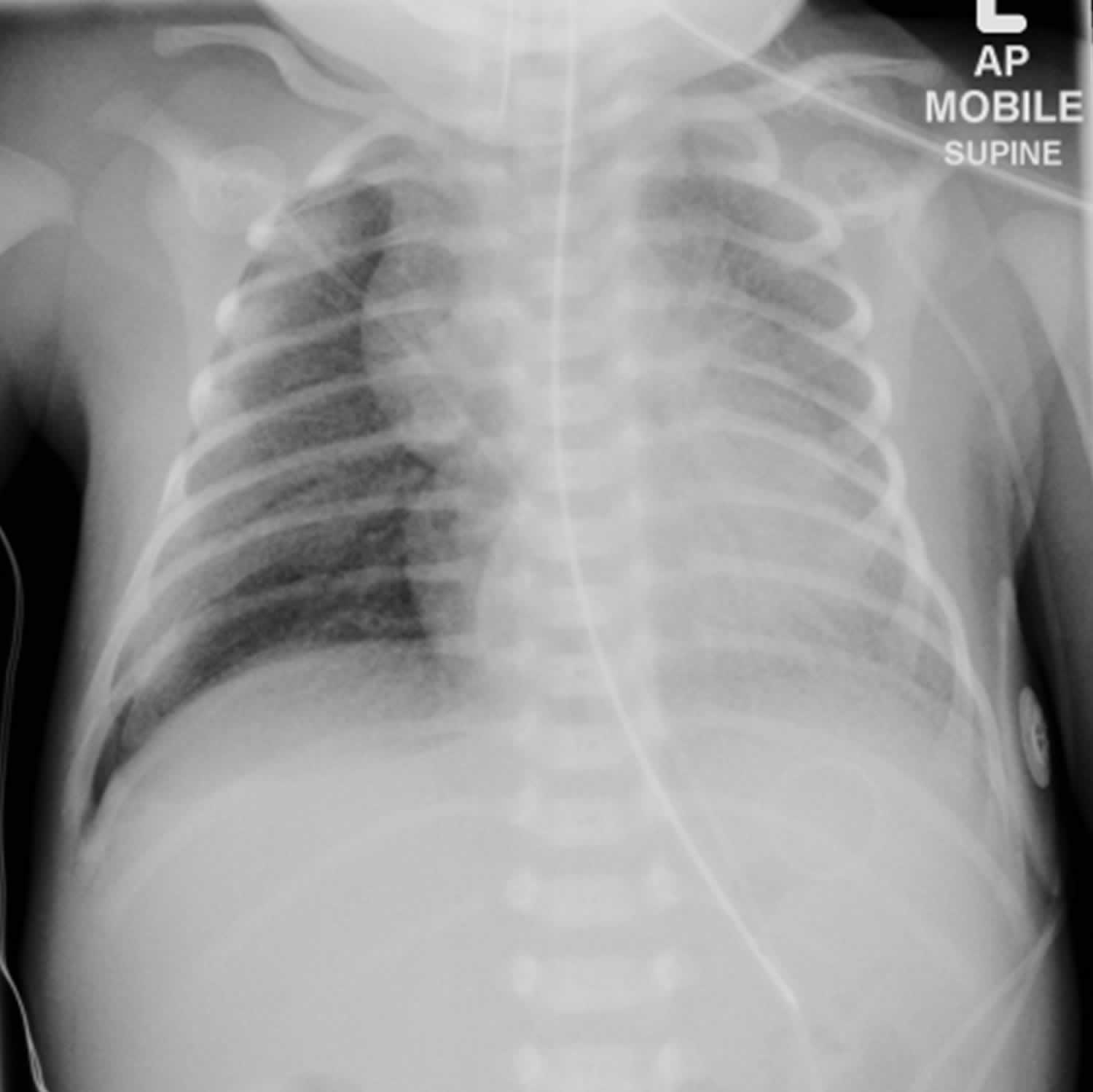
[Source 16 ]Figure 2. Infant respiratory distress syndrome chest X-ray
Footnotes: Intubated newborn day 1 with respiratory distress. Chest X-ray showing bilateral ground glass density throughout both lungs with some air-bronchograms. Relative lucency of the right hemithorax with pleural edge seen laterally and in the right costophrenic region consistent with pneumothorax or collapsed lung. Normal cardiothymic contour. Humeral head ossification centers are not seen suggesting prematurity. Findings are consistent with infant respiratory distress syndrome in a premature neonate complicated by pneumothorax from barotrauma (physical damage to body tissues caused by a difference in pressure between a gas space inside, or in contact with, the body and the surrounding gas or liquid). The lungs are well expanded as the newborn is intubated. In a non-intubated patient with infant respiratory distress syndrome the lungs would demonstrate reduced volume.
[Source 17 ]What is surfactant?
Surfactant is a lipoprotein (phospholipids and protein) complex produced by type 2 pneumocytes (alveolar type 2 cells) that lines the alveoli and decreases the surface tension of alveoli and allows normal expansion of the lungs during respiration and to prevent collapsed lung 18. Surfactant begins to be produced in the fetus at about 24 to 28 weeks of pregnancy, and is found in amniotic fluid between 28 and 32 weeks. By about 35 weeks gestation, most babies have developed adequate amounts of surfactant. Surfactant helps to keep the alveoli from collapsing once they are inflated. In the lungs, the exchange of oxygen and carbon dioxide takes place in the alveoli (small air sacs).
Surfactant is predominantly lipid-dense, comprising of around 70% to 80% phospholipids, 10% protein, and 10% neutral lipids 19, 18. The surfactant consists of four surfactant-specific proteins (SP); SP-A, SP-B, SP-C, and SP-D. SP-A and SP-D are involved in regulating inflammatory processes in the lung 20, 21, 22. SP-B is required for the formation of normal lamellar bodies in the type 2 cells and is also involved in the processing of SP-C. SP-C is a protein that might work with SP-B to improve surfactant deposition and function within the alveoli by lowering surface tension 23. Inside the alveolar type 2 cells, surfactant synthesis starts with phospholipids in the endoplasmic reticulum. The phospholipids transfer through the Golgi apparatus into the lamellar bodies. Surfactant lipoprotein complex (SP-A, SP-B, SP-C, and phospholipids) forms inside lamellar bodies at the apical surface of type 2 cells, which are subsequently released into alveoli by exocytosis.
In surfactant deficiency lung disease, surfactant production is deficient and alveoli are unable to adequately expand. Decreased lung compliance leads to alveolar injury, resulting in accumulation of fibrin and cellular debris called hyaline membranes within the alveoli. This debris, as well as collapsed lung (lung atelectasis), leads to poor oxygen exchange leading to difficulty breathing and potentially requiring medical attention.
Prematurity is the most common risk factor for surfactant deficiency lung disease, with a frequency of 60–80% in children born before 28 weeks and 15–30% in children born between 32 and 36 weeks 5, 6, 7, 8.
What does surfactant do?
In healthy lungs, surfactant is released into the lung tissues where it helps lower surface tension in the airways, which helps keep the lung alveoli (air sacs) open 24, 18. Newborn respiratory distress syndrome occurs when there is not enough of a substance in the lungs called surfactant. When there is not enough surfactant, the tiny alveoli collapse with each breath. As the alveoli collapse, damaged cells collect in the airways, which makes it even harder to breath. These cells are called hyaline membranes. Your baby works harder and harder at breathing, trying to re-inflate the collapsed airways.
As your baby’s lung function decreases, less oxygen is taken in and more carbon dioxide builds up in the blood. This can lead to acidosis (increased acid in the blood), a condition that can affect other body organs. Without treatment, your baby becomes exhausted trying to breathe and eventually gives up. A mechanical ventilator (breathing machine) must do the work of breathing instead.
Exogenous surfactant administration or surfactant replacement therapy (SRT) is considered in preterm infants with radiographic (small volume lungs, haziness/ ground-glass appearance, air bronchograms, and loss of cardiac borders on chest radiographs) and clinical evidence of infant respiratory distress syndrome (IRDS) or neonatal respiratory distress syndrome (NRDS) 15. Early (within 1 to 2 hours of birth) surfactant administration in preterm infants with infant respiratory distress syndrome (IRDS) reduces morbidity and mortality 25. There is a reduced risk of air leak and chronic lung disease 25. In neonatal respiratory distress syndrome (NRDS), the earlier surfactant is used, the better the outcomes. Also, the outcome is reportedly better if early surfactant administration is followed by extubation of preterm infants with respiratory distress syndrome (RDS) compared to prolonged ventilation after surfactant administration 26, 27, 28, 29.
Therapeutically, surfactant administration is also indicated in infants with meconium aspiration syndrome (MAS) 18, 30, 31, 28, 25, 32. Surfactant use, in this case, will recover oxygenation and reduce the need for extracorporeal membrane oxygenation (ECMO). Also, infants who undergo intubation for any reason, irrespective of their gestational age or infants with respiratory failure requiring mechanical ventilation, can receive surfactant therapy.
Prophylactic use of surfactant is indicated in neonates who are predisposed to the development of infant respiratory distress syndrome (IRDS) or neonatal respiratory distress syndrome (NRDS) 18. These include infants born before 32 weeks gestation and those with low birth weights (less than 1300 gm) 18. In premature infants at risk for developing infant respiratory distress syndrome (IRDS), the surfactant administration with brief lung-protective ventilation followed by extubation to nasal CPAP has been found to lower the incidence of mechanical ventilation, air leak syndromes and result in a lower incidence of pneumothorax, pulmonary interstitial emphysema and improved survival without bronchopulmonary dysplasia. Prophylactic surfactant is also indicated in infants with laboratory evidence of surfactant deficiency (lecithin/sphingomyelin ratio of less than 2 to 1, evidence of lung immaturity on bubble stability test, or absence of phosphatidylglycerol). Surfactant replacement therapy may also be considered in other situations like worsening pulmonary hemorrhage 33.
The use of surfactant may also be beneficial in severe respiratory syncytial virus (RSV) induced respiratory failure 33. In this case, there may be an improvement in gas exchange and respiration, contributing to shortened hospital stay and duration of mechanical ventilation.
Who is affected by infant respiratory distress syndrome?
Infant respiratory distress syndrome or newborn respiratory distress syndrome occurs in about 60 to 80 percent of babies born before 28 weeks gestation, but only in 15 to 30 percent of those born between 32 and 36 weeks. About 25 percent of babies born at 30 weeks develop newborn respiratory distress syndrome severe enough to need a mechanical ventilator (breathing machine).
Although most babies with infant respiratory distress syndrome are premature, other factors can influence the chances of developing the disease. These include the following:
- Caucasian or male babies
- Previous birth of baby with newborn respiratory distress syndrome
- Cesarean delivery
- Perinatal asphyxia (lack of air immediately before, during or after birth)
- Cold stress (a condition that suppresses surfactant production)
- Perinatal infection
- Multiple births (multiple birth babies are often premature)
- Infants of diabetic mothers (too much insulin in a baby’s system due to maternal diabetes can delay surfactant production)
- Babies with patent ductus arteriosus (PDA)
How long does infant respiratory distress syndrome last?
The recovery period is different for all babies because there are many reasons why some babies recover faster than others. Infant respiratory distress syndrome or newborn respiratory distress syndrome is most severe in the first 2 to 3 days after birth. Then the baby gradually needs less added oxygen. If a baby has mild disease and has not needed a breathing machine, he may be off oxygen in 5-7 days. If a baby has more severe disease, the condition may last 2 to 3 weeks. The speed of recovery depends on the following:
- The size and prematurity of your baby
- The severity of the condition
- Presence of any infection
- Presence of any heart condition
Infants who are on ventilators for a long time may develop changes in their lungs. This may increase the time they need ventilator support. Other factors such as infection can also affect recovery. Each baby responds and recovers in a unique way.
How can I tell if my baby is getting better?
You can tell if your baby is getting better if:
- Your baby is breathing easier
- Your baby is needing less oxygen.
- Your baby is put on lower settings for CPAP or the respirator and will finally be taken off of these machines.
What factors determine how infant respiratory distress syndrome progresses?
The course of illness with infant respiratory distress syndrome or newborn respiratory distress syndrome depends on the size and gestational age of your baby, the severity of the disease, the presence of infection, whether or not your baby has a patent ductus arteriosus (a heart condition) and whether or not she needs mechanical help to breathe.
Are there long term effects from infant respiratory distress syndrome?
Long term effects are more likely if the infant respiratory distress syndrome has been severe or if there have been complications.
Possible long term problems may include:
- Increased severity of colds or other respiratory infections, especially for the first two years.
- Increased sensitivity to lung irritants such as smoke and pollution.
- Greater likelihood of wheezing or other asthma-like problems in childhood.
- Greater likelihood of hospitalization in the first two years of life due to respiratory syncytial virus (RSV).
- If the infant respiratory distress syndrome was severe, the baby may have injury and scaring of the lung called bronchopulmonary dysplasia (BPD) also known as chronic lung disease.
Infant respiratory distress syndrome causes
Infant respiratory distress syndrome or newborn respiratory distress syndrome occurs in infants whose lungs have not yet fully developed. The lung condition is mainly caused by a lack of a slippery substance in the lungs called surfactant. This substance helps the lungs fill with air and keeps the air sacs from deflating. Surfactant is present when the lungs are fully developed.
Historically, neonatal respiratory distress syndrome was known as hyaline membrane disease, owing to an eosinophilic membrane that lines the distal airspaces, usually terminal bronchioles or alveolar ducts, in autopsies of neonates with respiratory distress syndrome 34, 3. Macroscopically, lung tissue from infants with hyaline membrane disease appears similar to liver tissue with a ruddy appearance. The hyaline membrane mentioned above is composed of fibrin, cellular debris from lung epithelium, red blood cells, and leukocytes 3. Microscopic histological examination may also reveal pulmonary tissue with few dilated alveoli among diffuse areas of atelectasis 34.
Infant respiratory distress syndrome or neonatal respiratory distress syndrome can also be due to genetic problems with lung development. Monozygotic twins have a higher incidence of newborn respiratory distress syndrome compared to dizygotic twins, and an increased incidence of newborn respiratory distress syndrome has also been reported in families, thus supporting an underlying genetic predisposition 35. Infants with genetic causes of surfactant protein deficiency can also present with varying degrees of infant respiratory distress syndrome or neonatal respiratory distress syndrome. Rare recessive mutations of the SP-B gene causing SP-B deficiency can present in the neonatal period with severe respiratory distress syndrome and progress to severe respiratory failure 36. On the other hand, SP-C gene mutations are seen in about 0.1% of the population and present with interstitial lung disease, usually beyond the first month of life. Neonatal respiratory distress syndrome is also associated with deletions in ATP binding cassette sub-family A, member 3 (ABCA-3). Although about 4% of the population reportedly carries this deletion, the exact incidence of neonatal respiratory distress syndrome in this population is unknown 37, 38.
The most important risk factors for newborn respiratory distress syndrome are prematurity and low birth weight. Most cases of newborn respiratory distress syndrome occur in babies born before 37 to 39 weeks. The more premature the baby is, the higher the chance of newborn respiratory distress syndrome after birth. The problem is uncommon in babies born full-term (after 39 weeks).
Other risk factors that can increase the risk of newborn respiratory distress syndrome include 39:
- A brother or sister who had newborn respiratory distress syndrome
- Diabetes in the mother (maternal diabetes)
- Cesarean delivery or induction of labor before the baby is full-term
- Problems with delivery that reduce blood flow to the baby (perinatal hypoxia and ischemia)
- Multiple pregnancy (twins or more)
- Rapid labor
- Late preterm delivery
- Delivery in the absence of labor
- White race, male gender.
Infant respiratory distress syndrome prevention
Taking steps to prevent premature birth can help prevent neonatal respiratory distress syndrome. Good prenatal care and regular checkups beginning as soon as a woman discovers she is pregnant can help avoid premature birth.
The risk of newborn respiratory distress syndrome can also be lessened by the proper timing of delivery. An induced delivery or cesarean may be needed. A lab test can be done before delivery to check the readiness of the baby’s lungs. Unless medically necessary, induced or cesarean deliveries should be delayed until at least 39 weeks or until tests show that the baby’s lungs have matured.
Medicines called corticosteroids can help speed up lung development before a baby is born. They are often given to pregnant women between 24 and 34 weeks of pregnancy who seem likely to deliver in the next week. More research is needed to determine if corticosteroids may also benefit babies who are younger than 24 or older than 34 weeks.
At times, it may be possible to give other medicines to delay labor and delivery until the steroid medicine has time to work. This treatment may reduce the severity of newborn respiratory distress syndrome. It may also help prevent other complications of prematurity. However, it will not totally remove the risks.
Infant respiratory distress syndrome symptoms
The infant with infant respiratory distress syndrome is often born premature and presents with signs of respiratory distress usually immediately after delivery, or within minutes of birth. Most of the time, infant respiratory distress syndrome symptoms appear within minutes of birth. However, they may not be seen for several hours.
In affected newborns, the lungs are stiff and the air sacs tend to collapse completely, emptying the lungs of air. In some very premature newborns, the lungs may be so stiff that the newborns are unable to begin breathing at birth. More commonly, newborns try to breathe, but because the lungs are so stiff, severe trouble breathing (respiratory distress) occurs.
While each baby may experience symptoms differently, some of the most common symptoms of newborn respiratory distress syndrome include:
- Visibly labored and rapid breathing
- Breathing too fast or too slow
- Shallow breathing
- Flaring of the nostrils during breathing in
- Grunting while breathing out
- Unusual breathing movement (such as drawing back of the chest muscles with breathing) or retractions (chest skin pulls in with breaths)
- Chest may collapse with each breath
- Grunting when breathing out
- Skin color may be pale, bluish (cyanosis) or gray
- Brief stop in breathing (apnea)
- Decreased urine output
- Shortness of breath and grunting sounds while breathing.
In untreated newborn respiratory distress syndrome, the symptoms will progressively worsen over 48 to 72 hours towards respiratory failure, and the infant may become lethargic and apneic 40. The infant may also develop peripheral extremity edema and show signs of decreased urine output (oliguria).
Symptoms of newborn respiratory distress syndrome usually peak by the third day and may resolve quickly when your baby begins to diurese (excrete excess water in urine) and needs less oxygen and mechanical help to breathe.
Because much of the lung is airless in infant respiratory distress syndrome, newborns with respiratory distress syndrome have low levels of oxygen in the blood, which causes a bluish or gray discoloration to the skin and/or lips (cyanosis). In newborns of color, the skin may change to colors such as yellow-gray, gray, or white. These changes may be more easily seen in the mucous membranes lining the inside of the mouth, nose, and eyelids.
Over a period of hours, the respiratory distress tends to become more severe as the muscles used for breathing tire, the small amount of surfactant in the lungs is used up, and increasing numbers of air sacs collapse. If the low oxygen levels are not treated, newborns may have damage to their brain and other organs and may die.
Infant respiratory distress syndrome complications
Your baby may develop complications of the infant respiratory distress syndrome or problems as side effects of treatment. As with any disease, more severe cases often have greater risks for complications.
Complications of neonatal respiratory distress syndrome are related mainly to the clinical course of respiratory distress syndrome in neonates and the long term outcomes of the neonates. While surfactant therapy has decreased the morbidity associated with respiratory distress syndrome, many patients continue to have complications during and after the acute course of respiratory distress syndrome.
Some complications associated with newborn respiratory distress syndrome include the following:
Acute complications of respiratory distress syndrome include the following 41:
- Air leaks into the space between the chest wall and the outer tissues of the lungs (pneumothorax) secondary to mechanical ventilation
- Air leaks into the mediastinum (the space between the two pleural sacs containing the lungs) (pneumomediastinum) secondary to mechanical ventilation
- Air leaks into the area between the heart and the thin sac that surrounds the heart (pneumopericardium) secondary to mechanical ventilation
- Air leaks and becomes trapped between the alveoli, the tiny air sacs of the lungs (pulmonary interstitial emphysema) secondary to mechanical ventilation
- Infection. Infections may complicate the management of respiratory distress syndrome and may manifest in various ways, including failure to improve, sudden deterioration, or a change in white blood cell (WBC) count or thrombocytopenia. Also, invasive procedures (eg, venipuncture, catheter insertion, use of respiratory equipment) and use of postnatal steroids provide access for organisms that may invade the immunologically compromised host.
- With the advent of surfactant therapy, small and ill infants are surviving, with an increased incidence of septicemia occurring in them secondary to staphylococcal epidermidis and/or candidal infection. When septicemia is suspected, obtain blood cultures from two sites and start appropriate antibiotics and/or antifungal therapy until culture results are obtained. Some neonatal ICUs use prophylactic fluconazole in the extremely premature infants, achieving a decrease in the incidence of candidal septicemia 42.
- Bleeding into the brain (intraventricular hemorrhage of the newborn) – Bleeding into the brain is quite common in premature babies, but most bleeds are mild and do not cause long-term problems.
- Intraventricular hemorrhage is observed in 20-40% of premature infants, with greater frequency in infants with respiratory distress syndrome who require mechanical ventilation. Cranial ultrasonography is performed in the first week in premature neonates younger than 32 weeks’ gestation and at 36 weeks or at the time of discharge, or as indicated (eg, suspected seizures).
- Use of antenatal steroids has decreased the frequency of intracranial hemorrhage in these patients with respiratory distress syndrome. Although a few studies have shown that prophylactic indomethacin therapy may decrease intraventricular hemorrhage in premature infants, its routine use is discouraged because of the risk of intestinal perforation.
- Periventricular leukomalacia (PVL) is a brain injury, most common in premature babies, where the white matter around the brain’s ventricles is damaged, potentially leading to motor, cognitive, and developmental problems, including cerebral palsy. Hypocarbia (abnormally low carbon dioxide levels in the blood typically below 35 mmHg, often resulting from hyperventilation) and chorioamnionitis are associated with an increase in periventricular leukomalacia.
- Persistent patent ductus arteriosus (PDA) with increasing left-to-right shunt due to reduced oxygen stimulus. Although helpful in the diagnosis of patent ductus arteriosus (PDA), heart murmur and wide pulse pressure are not always apparent in critically ill infants. Infants requiring low fraction of inspired oxygen (FIO2) or who are clinically stable do not require treatment, as the patent ductus arteriosus (PDA) may close spontaneously. Ductal-dependent cardiac anomalies should be excluded prior to initiating therapy. Treat patent ductus arteriosus (PDA) with ibuprofen or indomethacin, which can be repeated during the first 2 weeks if the patent ductus arteriosus (PDA) reopens 43. In refractory incidents of respiratory distress syndrome or in infants in whom medical therapy is contraindicated, surgically close the patent ductus arteriosus (PDA).
- Bleeding into the lung (pulmonary hemorrhage; sometimes associated with surfactant use) – Bleeding into the lungs is treated with increase positive end-expiratory pressure (PEEP) from a ventilator to stop the bleeding, intratracheal epinephrine and a blood transfusion. In some patients, pulmonary hemorrhage may be associated with patent ductus arteriosus (PDA); promptly treat pulmonary hemorrhage in such individuals.
- Necrotizing enterocolitis (NEC) and/or gastrointestinal (GI) perforation. X-ray of the abdomen can assists in confirming their diagnosis. Spontaneous perforation (not necessarily as part of NEC) occasionally occurs in critically ill premature infants and has been associated with the use of steroids and/or indomethacin.
- Apnea of prematurity. Apnea of prematurity is common in immature infants, and its incidence has increased with surfactant therapy, possibly because of early extubation. Manage apnea of prematurity with methylxanthines (caffeine) and/or bubble or continuous flow nasal continuous positive airway pressure (CPAP), nasal intermittent ventilation, or with assisted ventilation in refractory incidents. Exclude septicemia, seizures, gastroesophageal reflux, and metabolic and other causes in infants with apnea of prematurity.
- Oxygen toxicity from treatment
- Subglottic stenosis (secondary to intubation).
Long term problems associated with newborn respiratory distress syndrome or extreme prematurity may include:
- Problems with lung development and growth (bronchopulmonary dysplasia)
- Sometimes ventilation (begun within 24 hours of birth) or the surfactant used to treat newborn respiratory distress syndrome causes scarring to the baby’s lungs, which affects their development. This lung scarring is called bronchopulmonary dysplasia. Symptoms of bronchopulmonary dysplasia include rapid, shallow breathing and shortness of breath. Babies with severe bronchopulmonary dysplasia usually need additional oxygen from tubes into their nose to help with their breathing. This is usually stopped after a few months, when the lungs have healed. But children with bronchopulmonary dysplasia may need regular medication, such as bronchodilators, to help widen their airways and make breathing easier.
- Postnatal use of surfactant therapy, gentler ventilation, vitamin A, low-dose steroids, and inhaled nitric oxide may reduce the severity of bronchopulmonary dysplasia 44.
- Delayed development or intellectual disability associated with brain damage or bleeding. If the baby’s brain is damaged during newborn respiratory distress syndrome, either because of bleeding or a lack of oxygen, it can lead to long-term developmental disabilities, such as learning difficulties, movement problems, impaired hearing and impaired vision. But these developmental problems are not usually severe. For example, 1 survey estimated that 3 out of 4 children with developmental problems only have a mild disability, which should not stop them leading a normal adult life.
- The Mental Developmental Index (MDI) is a standardized test used to assess cognitive development in infants and toddlers, specifically focusing on skills like memory, imitation, social communication, problem-solving, and receptive and expressive language 45, 46. A mean Mental Developmental Index (MDI) score of 100 at the 50th percentile signifies mid-average functioning. Mental Developmental Index (MDI) below 85 (1 SD below the mean) indicate mild impairment or being “at risk” of developmental delay, while Mental Developmental Index (MDI) below 70 (2 SD below the mean) indicate moderate to severe impairment. In a study that assessed the outcomes of 288 very preterm Chinese infants with severe respiratory distress syndrome on mechanical ventilation through 18 months of corrected age, the incidence of cerebral palsy and mental developmental index (MDI) less than 70 were highest among infants born at younger than 28 weeks’ gestation compared to those born at 28-30 and 30-32 weeks’ gestation 47. Factors associated with cerebral palsy and an mental developmental index (MDI) below 70 were the administration of antenatal corticosteroids, decreased weight gain, and the presence of preeclampsia, fetal distress, and early/late bacteremia; factors that increased the risk of cerebral palsy and an mental developmental index (MDI) below 70 were increased length of mechanical ventilator support and blood transfusions 47.
- Problems with eye development (retinopathy of prematurity) and blindness.
- Infants with respiratory distress syndrome who have a partial pressure of oxygen (PaO2) value of over 100mm Hg are at increased risk for retinopathy of prematurity. Hence, closely monitor PaO2 and maintain it at 50-70mm Hg. Although pulse oximetry is used in all premature infants, it is not helpful in preventing retinopathy of prematurity in tiny infants because of the flat portion of the oxygen-hemoglobin dissociation curve. An ophthalmologist examines the eyes of all premature infants at 34 weeks’ gestation and thereafter as indicated. If retinopathy of prematurity progresses, laser therapy or cryotherapy is used to prevent retinal detachment and blindness. Closely monitor infants with retinopathy of prematurity for refractive errors. Intraocular bevacizumab, a monoclonal antibody targeting the vascular endothelial growth factor, has been used successfully to treat retinopathy of prematurity. Although it is a promising therapy for retinopathy of prematurity, further studies are needed before it can be recommended for routine use 48.
Acute complications due to positive pressure ventilation or invasive mechanical ventilation include air-leak syndromes such as pneumothorax, pneumomediastinum, and pulmonary interstitial emphysema. There is also an increase in the incidence of intracranial hemorrhage and patent ductus arteriosus in very low birth weight infants with respiratory distress syndrome, although independently linked to prematurity itself.
Bronchopulmonary dysplasia is a chronic complication of respiratory distress syndrome. The pathophysiology of bronchopulmonary dysplasia involves both arrested lung development as well as lung injury and inflammation. Besides a surfactant deficiency, the immature lung of the premature infant has decreased compliance, decreased fluid clearance, and immature vascular development, which predisposes the lung to injury and inflammation, further disrupting the normal development of alveoli and pulmonary vasculature. Also, oxidative stress from hyperoxia secondary to mechanical ventilation, and decreased anti-oxidant capabilities of the premature lung, both lead to further damage to the lung through the increased production on TGF-β1 and other pro-inflammatory cytokines 49.
Neurodevelopmental delay is another complication of respiratory distress syndrome, especially with infants who received mechanical ventilation long-term 47. The incidence of cerebral palsy also was increased in infants with respiratory distress syndrome, with decreasing incidence as gestational age increased. The length of time on mechanical ventilation correlates with increased rates of both cerebral palsy and neurodevelopmental delay 47.
Infant respiratory distress syndrome diagnosis
The following tests are used to detect respiratory distress in babies:
- Blood gas analysis – shows low oxygen and excess acid in the body fluids.
- Chest X-ray – shows a “ground glass” appearance to the lungs that is typical of the disease. This often develops 6 to 12 hours after birth.
- Echocardiography (EKG or ECG) – may be used to rule out heart problems that could cause symptoms similar to newborn respiratory distress syndrome. An electrocardiogram is a test that records the electrical activity of the heart, shows arrhythmias (abnormal rhythms) and detects damage to the heart muscle.
- Lab tests – help to rule out infection as a cause of breathing problems.
The diagnosis of infant respiratory distress syndrome or respiratory distress syndrome is based on signs of respiratory distress, levels of oxygen in the blood, and abnormal chest x-ray results.
Infant respiratory distress syndrome can sometimes accompany a disorder, such as infection in the blood (sepsis) or transient tachypnea of the newborn (TTN). Therefore, doctors may do other tests to rule out these disorders. Cultures of blood and sometimes cerebrospinal fluid may be done to look for certain kinds of infections.
Infant respiratory distress syndrome differential diagnosis
There are numerous causes of neonatal respiratory distress syndrome, including transient tachypnea of the newborn (TTN), pulmonary air leak disorders (pneumothorax, pneumomediastinum), neonatal pneumonia, meconium aspiration, persistent pulmonary hypertension of the newborn, and the broad categories of cyanotic congenital heart disease and interstitial lung disease 40.
- Transient tachypnea of the newborn (TTN), also known as retained fetal fluid or wet lung disease, have impaired resorption of the fetal lung fluid and presents in the neonate as tachypnea for the first few hours of life, lasting up to one day. The tachypnea usually resolves within 48 hours. Chest radiograph shows perihilar streaking, representing perihilar interstitial edema, without the diffuse reticulo-granular ground glass appearance of infant respiratory distress syndrome.
- Pulmonary air leak syndromes such as pneumothorax and pneumomediastinum may also present as respiratory distress, but the onset of symptoms may be more acute. Other clinical clues include chest rise asymmetry, and diminished breath sounds on one side of the chest. Hyperlucent areas on chest radiography can be appreciated if the air leak is significant. Pulmonary interstitial emphysema affects infants who are mechanically ventilated; symptoms of respiratory distress often occur later than expected with respiratory distress syndrome, and the trapped air within the perivascular tissues has a characteristic appearance of cystic lucencies on chest radiography.
- Bacterial pneumonia, especially related to Group B Streptococcus in a newborn is often clinically and radiographically indistinguishable from respiratory distress syndrome. The preferred treatment includes empirical antibiotics in addition to respiratory management.
- Infants with cyanotic congenital heart disease may have similar symptoms clinically, but will not have the diffuse reticulo-granular ground glass appearance on chest radiography. The radiological findings depend on the underlying anatomic abnormality.
Infant respiratory distress syndrome treatment
Treatment before birth
If you’re thought to be at risk of giving birth before week 34 of pregnancy, treatment for newborn respiratory distress syndrome can begin before birth.
Before birth, doctors are able to test the maturity of the fetus’s lungs by measuring the level of surfactant in the amniotic fluid. Amniotic fluid is collected from the sac surrounding the fetus during a procedure called amniocentesis or is collected from the mother’s vagina if the membranes have ruptured. The surfactant level helps doctors determine the best time to deliver the fetus. The risk of respiratory distress syndrome is greatly reduced if delivery can be safely delayed until the fetus’s lungs have produced sufficient surfactant.
When premature birth cannot be avoided, obstetricians may give the mother injections of a corticosteroid (betamethasone). The corticosteroid goes into the fetus through the placenta and accelerates the production of surfactant. Within 48 hours after the steroid injections are started, the fetus’s lungs may mature to the point that respiratory distress syndrome is less likely to develop after delivery or, if it does develop, is likely to be milder. A second dose is usually given 24 hours after the first. It’s estimated that the treatment helps prevent newborn respiratory distress syndrome in a third of premature births.
You may also be offered magnesium sulphate to reduce the risk of developmental problems linked to being born early.
If you take magnesium sulphate for more than 5 to 7 days or several times during your pregnancy, your newborn baby may be offered extra checks. This is because prolonged use of magnesium sulphate in pregnancy has in rare cases been linked to bone problems in newborn babies.
Treatment after the birth
Babies who are premature or have other conditions that make them at high risk for the infant respiratory distress syndrome need to be treated at birth by a medical team that specializes in newborn breathing problems. Your baby may be transferred to a ward that provides specialist care for premature babies (a neonatal unit).
Treatment will depend on how ill the infant is.
- If the symptoms are mild, they may only need extra oxygen. It’s usually given through an incubator or tubes into their nose called a nasal cannula.
- In moderate cases, your baby may need Continuous Positive Airway Pressure (CPAP). CPAP is oxygen delivered under a small amount of pressure usually through little tubes that fit into your baby’s nostrils. Delivering oxygen under pressure helps keep the baby’s air sacs or alveoli open.
- In moderate or severe cases, your baby may need breathing machine (ventilator) to either support or take over their breathing. This is done by inserting a tiny tube into your baby’s wind pipe. This process is called intubation. Once intubated, your baby may be placed on a breathing machine (respirator) to administer breaths to the baby.
- Extra oxygen may be needed. A ventilator (breathing machine) may also be needed to deliver oxygen and help remove carbon dioxide from your baby’s lungs. However, this treatment needs to be monitored carefully to avoid side effects from too much oxygen.
- Once the baby is on the breathing machine (ventilator), surfactant (a medication that replaces the substance that is lacking in your baby’s lungs) may be administered through the airway tube in his wind pipe. This helps the alveoli expand more easily.
- Your baby may also have an Umbilical Catheter placed. This is done by placing a very small, soft catheter (tube) into one or two of the blood vessels in the baby’s umbilical cord. This is not a painful procedure for your baby. These catheters are used for:
- Giving medications like antibiotics
- Giving extra nutrients since he will not be eating normally
- Obtaining frequent blood samples from your baby without using a needle. These blood samples are necessary to make sure your baby is receiving the correct amount of oxygen and to
adjust the settings on the respirator when necessary.
Assisted ventilation with a ventilator (breathing machine) can be lifesaving for some babies. However, use of a breathing machine can damage the lung tissue, so this treatment should be avoided if possible. Babies may need this treatment if they have:
- High level of carbon dioxide in the blood
- Low blood oxygen
- Low blood pH (acidity)
- Repeated pauses in breathing
A treatment called continuous positive airway pressure (CPAP) may prevent the need for assisted ventilation or surfactant in many babies. CPAP sends air into the nose to help keep the airways open. It can be given by a ventilator (while the baby is breathing independently) or with a separate CPAP device.
As babies get better, they are weaned from the oxygen or ventilator over several days. Some infants may continue to need extra oxygen. Most infants are soon breathing on their own and doing well.
A heart monitor will be used, and blood tests and chest X-rays will be done.
Artificial surfactant may be given to newborns who are at high risk of developing respiratory distress syndrome to expand the lungs. At-risk newborns are those who were delivered before 30 weeks of gestational age, especially those whose mother did not receive corticosteroids. Giving extra surfactant to a sick infant has been shown to be helpful. The surfactant preparation can be lifesaving and reduces the risk of some complications, such as collapse of the lung (pneumothorax). The surfactant preparation acts in the same way that natural surfactant does. Surfactant therapy is given to newborns through a tube placed in the mouth that leads to the windpipe called endotracheal intubation and may be given immediately after birth in the delivery room to attempt to prevent respiratory distress syndrome before symptoms develop.
The American Academy of Pediatrics 2014 guidelines recommend immediate management of all preterm infants with nasal continuous positive airway pressure 15, 50. Subsequently, in selected patients, surfactant administration may be considered as an alternative to intubation with prophylactic or early surfactant administration. The surfactant replacement therapy is administered by trained personnel, in a clinical setting where equipment for intubation and resuscitation are readily available.
Endotracheal installation of surfactant is the most widely accepted technique. Surfactant is administered in liquid form via an endotracheal tube in a single bolus dose as quickly as the neonate tolerates. Some studies recommend the administration of all the surfactant at once while others advocate dividing the bolus into smaller aliquots.
Another technique of administration known as the INSURE technique is approached with the neonate not already intubated. The INSURE technique uses an in-out intubation procedure to administer the surfactant. This process includes intubation, followed by administration of the drug, and then extubation. Newer methods to limit the invasiveness of older approaches are in practice and targets of research today 51. One such technique is known as the Minimally Invasive Surfactant Therapy or MIST 52. The MIST method connects the patient to non-invasive respiratory support, and through that, the surfactant administration is with the work of spontaneous breathing. This method is increasingly being used to reduce intubation rates and their associated pathologies. Another method is the less invasive surfactant administration or LISA technique 53. This method utilizes a thin catheter for surfactant delivery, which serves to reduce the possibility of lung injury as is possible with intubation. At present, five surfactants (Lucinactant, colfosceril palmitate, beractant, calefacient, and protectant alfa) have FDA approval. Colfosceril palmitate is no longer commercially available. Lucinactant is the first U.S. FDA-approved protein-containing synthetic surfactant.
The usage of surfactant can lead to bradycardia and hypotension in the neonate. There are also some reports of instances of oxygen desaturation upon administration of the drug. Also, because conventional methods of surfactant administration require an endotracheal tube, certain risks are associated with tubal administration of any drug. Hygiene is of vital importance, especially in neonates as their immunities are underdeveloped. Infections and sepsis can occur if measures are not taken to prevent them. Unless the neonate is already intubated, intubation must take place first, which presents with the risk of injury and air leaks. Intubation also poses the risk of airway obstruction, mechanical damage to the airway, and it may require frequent suctioning. There is a risk of alterations in cerebral flow and hypoxemia. Concomitant administration of oxygen can damage lung tissue from oxygen pressure. Excess oxygenation and subsequent cessation of it leads to diseases like bronchopulmonary dysplasia and retrolental fibroplasia in infants as well 54.
Since surfactant extraction is from animal sources, a risk of immune activation exists. Rarely the drug can lead to pneumothorax and/or pulmonary hemorrhage. Other side effects may include intraventricular hemorrhage, patent ductus arteriosus, retinopathy, necrotizing enterocolitis, and/or blockage of the endotracheal tube can cause complications as well.
Babies with newborn respiratory distress syndrome need close care. This includes:
- Having a calm setting
- Gentle handling
- Staying at an ideal body temperature
- Carefully managing fluids and nutrition
- Treating infections right away.
Continuous Positive Airway Pressure (CPAP)
Nasal CPAP (NCPAP) is an initial intervention in preterm infants with infant respiratory distress syndrome or risk of respiratory distress syndrome without respiratory failure. Multiple modalities are available for CPAP delivery, including ventilator derived CPAP as well as a less expensive bubble CPAP device. Infants who received CPAP fared as well as infants who received prophylactic surfactant therapy along with mechanical ventilation in the SUPPORT trial (Surfactant Positive Airway Pressure and Pulse Oximetry Randomized Trial) 55 and those who received early CPAP had a reduced need for surfactant therapy. Also, the incidence of bronchopulmonary dysplasia decreased with the use of CPAP 56. The goals of treatment include keeping SpO2 (peripheral oxygen saturation or blood oxygen saturation) between 90-95%, and PaCO2 (partial pressure of carbon dioxide in arterial blood) between 45-65 mmHg.
High Flow Nasal Canula
Heated humidified high-flow nasal cannulas (HFNC) are also used in some centers as an alternative to CPAP to provide positive distending pressure ventilation to neonates with respiratory distress syndrome. A clinical trial by Roberts et al. 57, high-flow nasal cannulas was found to be inferior to CPAP.
Mechanical Ventilation
Infants who do not respond to CPAP, develop respiratory acidosis (PH < 7.2 and PaCo2 > 60-65 mm of Hg), hypoxemia (PaO2 < 50 mm of Hg or Fio2 > 0.40 on CPAP), or severe apnea are managed with endotracheal intubation and mechanical ventilation 3. The goals of mechanical ventilation include providing adequate respiratory support while balancing the risks of barotrauma, volutrauma, and oxygen toxicity. Time-cycled pressure limited ventilation is the preferred initial mode of ventilation in preterm infants with respiratory distress syndrome. High-frequency oscillatory ventilation (HFOV) and high-frequency jet ventilation (HFJV) are often used as rescue modalities when requiring high conventional ventilator support or concerns for pulmonary air leaks 3. Other strategies include the use of high-frequency ventilation empirically in extremely preterm infants to minimize lung injury 3.
Surfactant Therapy
The targeted treatment for surfactant deficiency is intratracheal surfactant replacement therapy via an endotracheal tube. Surfactant administered within 30 to 60 minutes of the birth of a premature neonate is found to be beneficial 3. Surfactant hastens recovery and decreases the risk of pneumothorax, interstitial emphysema, intraventricular hemorrhage, bronchopulmonary dysplasia, and neonatal mortality in the hospital and at one year 3. However, neonates who receive surfactant for established infant respiratory distress syndrome, have an increased risk of apnea of prematurity. According to European census guidelines, the surfactant is administered to immature babies with FiO2 (fraction of inspired oxygen or the percentage of oxygen a person inhales, ranging from 21% in room air to 100% with pure oxygen) > 30%, and mature babies with FiO2 > 40%. Currently, there are no clinically significant advantages of using one type over another when used in similar doses 58:
- Beractant: This is a modified natural surfactant prepared from minced bovine lungs with the additives
- Poractant alfa: This is a modified natural surfactant derived from minced porcine lung extract
- Calfactant: This is a natural surfactant obtained from lavaging calf lung alveoli and contains 80% phosphatidylcholine with only 1% protein
- Synthetic surfactant: Clinical trials are ongoing
Surfactant is administered either by standard endotracheal intubation, which needs experienced practitioner or through less invasive surfactant administration technique like aerosolized nebulized surfactant preparations, laryngeal mask, pharyngeal instillation, and thin intratracheal catheters 59, 60, 61, 62, 63. The standard technique of surfactant administration by endotracheal intubation and mechanical ventilation may result in transient airway obstruction, pulmonary injury, pulmonary air leak, and airway injury 64, 65. Emerging evidence shows that the less invasive surfactant administration technique is associated with a lower rate of bronchopulmonary dysplasia, death, and need for mechanical ventilation compared to surfactant administration through endotracheal intubation 66. Still, further investigations are required to prefer the less invasive surfactant administration technique as the standard technique of surfactant administration in place of endotracheal intubation. If the neonates maintain adequate respiratory drive with FiO2 < 30%, it should be planned to stop surfactant and switch to CPAP. Oxygen saturation (>90%), thermoregulation (36.5 to 37.5 °C), and fluid and nutrition status should be monitored.
Supportive Care
Preterm infants with apnea of prematurity may require caffeine therapy. Caffeine can also be administered to preterm infants < 28 weeks with extremely low birth weight (birth weight <1000 g) to increase respiratory drive and enhance the use of CPAP. There was a low incidence of bronchopulmonary dysplasia and earlier extubation in preterm infants who received caffeine compared to placebo 67.
Optimal fluid and electrolyte management is critical in the initial course of infant respiratory distress syndrome. Some neonates may require volume resuscitation using crystalloids as well as vasopressors for hypotension. Furthermore, the overall care of a preterm infant also includes optimizing thermoregulation, nutritional support, blood transfusions for anemia, treatment for hemodynamically significant patent ductus arteriosus (PDA), and antibiotic therapy as necessary.
Infant respiratory distress syndrome prognosis
Infant respiratory distress syndrome often gets worse for 2 to 4 days after birth and improves slowly after that. Prognosis of infants managed with antenatal steroids, respiratory support, and exogenous surfactant therapy is excellent. Natural production of surfactant increases after birth. With continued production of surfactant and sometimes with breathing support and surfactant therapy along with the onset of diuresis, respiratory distress syndrome usually resolves within 4 or 5 days. But the more premature the baby, the higher the risk and the more severe the infant respiratory distress syndrome. Some infants with severe infant respiratory distress syndrome will die. This most often occurs between days 2 and 7. Mortality is less than 10%, with some studies showing survival rates of up to 98% with advanced care 3. Increased survival in developed countries is in stark comparison to babies who received no intervention in low-income countries, where the mortality rate for premature infants with respiratory distress syndrome is significantly higher, at times close to 100% 68.
Without treatment that increases blood oxygen levels, newborns may develop heart failure and have damage to the brain or other organs and may die.
Long-term complications may develop due to:
- Too much oxygen.
- High pressure delivered to the lungs.
- More severe disease or immaturity. newborn respiratory distress syndrome can be associated with inflammation that causes lung or brain damage.
- Periods when the brain or other organs did not get enough oxygen.
Some infants who need treatment for a long time develop bronchopulmonary dysplasia.
- Cleveland RH, Rhein L. Newborn chest. In: Cleveland RH, ed. Imaging in pediatric pulmonology. New York, NY: Springer, 2012:81–97[↩]
- Rodriguez R, Martin R, Fanaroff A. Respiratory distress syndrome and its management. In: Fanaroff A, Martin R, eds. Fanaroff and Martin’s neonatal-perinatal medicine: diseases of the fetus and infant. St. Louis, MO: Mosby, 2002:1001–1011[↩]
- Yadav S, Lee B. Neonatal Respiratory Distress Syndrome. [Updated 2023 Jul 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560779[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Liszewski MC, Lee EY. Neonatal Lung Disorders: Pattern Recognition Approach to Diagnosis. AJR Am J Roentgenol. 2018 May;210(5):964-975. doi: 10.2214/AJR.17.19231[↩][↩][↩]
- Ueda T, Ikegami M, Jobe AH. Developmental changes of sheep surfactant: in vivo function and in vitro subtype conversion. J Appl Physiol (1985). 1994 Jun;76(6):2701-6. doi: 10.1152/jappl.1994.76.6.2701[↩][↩]
- Agrons GA, Courtney SE, Stocker JT, Markowitz RI. From the archives of the AFIP: Lung disease in premature neonates: radiologic-pathologic correlation. Radiographics. 2005 Jul-Aug;25(4):1047-73. doi: 10.1148/rg.254055019[↩][↩][↩]
- Cleveland RH. A radiologic update on medical diseases of the newborn chest. Pediatr Radiol. 1995;25(8):631-7. doi: 10.1007/BF02011835[↩][↩]
- Arthur R. The neonatal chest X-ray. Paediatr Respir Rev. 2001 Dec;2(4):311-23. doi: 10.1053/prrv.2001.0169[↩][↩]
- Smith PB, Ambalavanan N, Li L, Cotten CM, Laughon M, Walsh MC, Das A, Bell EF, Carlo WA, Stoll BJ, Shankaran S, Laptook AR, Higgins RD, Goldberg RN; Generic Database Subcommittee; Eunice Kennedy Shriver National Institute of Child Health Human Development Neonatal Research Network. Approach to infants born at 22 to 24 weeks’ gestation: relationship to outcomes of more-mature infants. Pediatrics. 2012 Jun;129(6):e1508-16. doi: 10.1542/peds.2011-2216[↩]
- Respiratory Distress Syndrome. https://emedicine.medscape.com/article/976034-overview#a6[↩]
- Hintz SR, Van Meurs KP, Perritt R, et al. NICHD Neonatal Research Network. Neurodevelopmental outcomes of premature infants with severe respiratory failure enrolled in a randomized controlled trial of inhaled nitric oxide. J Pediatr. 2007 Jul;151(1):16-22, 22.e1-3. doi: 10.1016/j.jpeds.2007.03.017[↩]
- American Academy of Pediatrics Steering Committee on Quality Improvement and Management. Classifying recommendations for clinical practice guidelines. Pediatrics. 2004 Sep;114(3):874-7. doi: 10.1542/peds.2004-1260[↩]
- Verder H, Albertsen P, Ebbesen F, Greisen G, Robertson B, Bertelsen A, Agertoft L, Djernes B, Nathan E, Reinholdt J. Nasal continuous positive airway pressure and early surfactant therapy for respiratory distress syndrome in newborns of less than 30 weeks’ gestation. Pediatrics. 1999 Feb;103(2):E24. doi: 10.1542/peds.103.2.e24[↩]
- Ho JJ, Subramaniam P, Davis PG. Continuous positive airway pressure (CPAP) for respiratory distress in preterm infants. Cochrane Database Syst Rev. 2020 Oct 15;10(10):CD002271. doi: 10.1002/14651858.CD002271.pub3[↩]
- Polin RA, Carlo WA; Committee on Fetus and Newborn; American Academy of Pediatrics. Surfactant replacement therapy for preterm and term neonates with respiratory distress. Pediatrics. 2014 Jan;133(1):156-63. doi: 10.1542/peds.2013-3443[↩][↩][↩]
- Panetti B, Bucci I, Di Ludovico A, Pellegrino GM, Di Filippo P, Di Pillo S, Chiarelli F, Attanasi M, Sferrazza Papa GF. Acute Respiratory Failure in Children: A Clinical Update on Diagnosis. Children (Basel). 2024 Oct 12;11(10):1232. doi: 10.3390/children11101232[↩]
- Hyaline membrane disease. https://radiopaedia.org/cases/hyaline-membrane-disease?lang=us[↩]
- Khawar H, Marwaha K. Surfactant. [Updated 2023 Jun 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK546600[↩][↩][↩][↩][↩][↩]
- Agassandian M, Mallampalli RK. Surfactant phospholipid metabolism. Biochim Biophys Acta. 2013 Mar;1831(3):612-25. doi: 10.1016/j.bbalip.2012.09.010[↩]
- Lawson PR, Reid KB. The roles of surfactant proteins A and D in innate immunity. Immunol Rev. 2000 Feb;173:66-78. doi: 10.1034/j.1600-065x.2000.917308.x[↩]
- Whitsett JA, Alenghat T. Respiratory epithelial cells orchestrate pulmonary innate immunity. Nat Immunol. 2015 Jan;16(1):27-35. doi: 10.1038/ni.3045[↩]
- Ariki S, Nishitani C, Kuroki Y. Diverse functions of pulmonary collectins in host defense of the lung. J Biomed Biotechnol. 2012;2012:532071. doi: 10.1155/2012/532071[↩]
- Whitsett JA, Weaver TE. Hydrophobic surfactant proteins in lung function and disease. N Engl J Med. 2002 Dec 26;347(26):2141-8. doi: 10.1056/NEJMra022387[↩]
- Griese M. Pulmonary surfactant in health and human lung diseases: state of the art. Eur Respir J. 1999 Jun;13(6):1455-76. doi: 10.1183/09031936.99.13614779[↩]
- Bahadue FL, Soll R. Early versus delayed selective surfactant treatment for neonatal respiratory distress syndrome. Cochrane Database Syst Rev. 2012 Nov 14;11(11):CD001456. doi: 10.1002/14651858.CD001456.pub2[↩][↩][↩]
- Stevens TP, Harrington EW, Blennow M, Soll RF. Early surfactant administration with brief ventilation vs. selective surfactant and continued mechanical ventilation for preterm infants with or at risk for respiratory distress syndrome. Cochrane Database Syst Rev. 2007 Oct 17;2007(4):CD003063. doi: 10.1002/14651858.CD003063.pub3[↩]
- Dunn MS, Kaempf J, de Klerk A, de Klerk R, Reilly M, Howard D, Ferrelli K, O’Conor J, Soll RF; Vermont Oxford Network DRM Study Group. Randomized trial comparing 3 approaches to the initial respiratory management of preterm neonates. Pediatrics. 2011 Nov;128(5):e1069-76. doi: 10.1542/peds.2010-3848[↩]
- Rojas-Reyes MX, Morley CJ, Soll R. Prophylactic versus selective use of surfactant in preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev. 2012 Mar 14;(3):CD000510. doi: 10.1002/14651858.CD000510.pub2[↩][↩]
- Escobedo MB, Gunkel JH, Kennedy KA, Shattuck KE, Sánchez PJ, Seidner S, Hensley G, Cochran CK, Moya F, Morris B, Denson S, Stribley R, Naqvi M, Lasky RE; Texas Neonatal Research Group. Early surfactant for neonates with mild to moderate respiratory distress syndrome: a multicenter, randomized trial. J Pediatr. 2004 Jun;144(6):804-8. doi: 10.1016/j.jpeds.2004.03.024[↩]
- Soll RF. Synthetic surfactant for respiratory distress syndrome in preterm infants. Cochrane Database Syst Rev. 2000;1998(2):CD001149. doi: 10.1002/14651858.CD001149[↩]
- Seger N, Soll R. Animal derived surfactant extract for treatment of respiratory distress syndrome. Cochrane Database Syst Rev. 2009 Apr 15;(2):CD007836. doi: 10.1002/14651858.CD007836[↩]
- Soll R, Ozek E. Prophylactic protein free synthetic surfactant for preventing morbidity and mortality in preterm infants. Cochrane Database Syst Rev. 2010 Jan 20;2010(1):CD001079. doi: 10.1002/14651858.CD001079.pub2[↩]
- Walsh BK, Daigle B, DiBlasi RM, Restrepo RD; American Association for Respiratory Care. AARC Clinical Practice Guideline. Surfactant replacement therapy: 2013. Respir Care. 2013 Feb;58(2):367-75. doi: 10.4187/respcare.02189[↩][↩]
- deMello DE, Chi EY, Doo E, Lagunoff D. Absence of tubular myelin in lungs of infants dying with hyaline membrane disease. Am J Pathol. 1987 Apr;127(1):131-9. https://pmc.ncbi.nlm.nih.gov/articles/instance/1899586/pdf/amjpathol00145-0136.pdf[↩][↩]
- Levit O, Jiang Y, Bizzarro MJ, Hussain N, Buhimschi CS, Gruen JR, Zhang H, Bhandari V. The genetic susceptibility to respiratory distress syndrome. Pediatr Res. 2009 Dec;66(6):693-7. doi: 10.1203/PDR.0b013e3181bbce86[↩]
- Cole FS, Hamvas A, Nogee LM. Genetic disorders of neonatal respiratory function. Pediatr Res. 2001 Aug;50(2):157-62. doi: 10.1203/00006450-200108000-00001[↩]
- Shulenin S, Nogee LM, Annilo T, Wert SE, Whitsett JA, Dean M. ABCA3 gene mutations in newborns with fatal surfactant deficiency. N Engl J Med. 2004 Mar 25;350(13):1296-303. doi: 10.1056/NEJMoa032178[↩]
- Wambach JA, Wegner DJ, Depass K, Heins H, Druley TE, Mitra RD, An P, Zhang Q, Nogee LM, Cole FS, Hamvas A. Single ABCA3 mutations increase risk for neonatal respiratory distress syndrome. Pediatrics. 2012 Dec;130(6):e1575-82. doi: 10.1542/peds.2012-0918[↩]
- Li Y, Wang W, Zhang D. Maternal diabetes mellitus and risk of neonatal respiratory distress syndrome: a meta-analysis. Acta Diabetol. 2019 Jul;56(7):729-740. doi: 10.1007/s00592-019-01327-4[↩]
- Reuter S, Moser C, Baack M. Respiratory distress in the newborn. Pediatr Rev. 2014 Oct;35(10):417-28; quiz 429. doi: 10.1542/pir.35-10-417[↩][↩]
- Fanaroff AA, Stoll BJ, Wright LL, Carlo WA, Ehrenkranz RA, Stark AR, Bauer CR, Donovan EF, Korones SB, Laptook AR, Lemons JA, Oh W, Papile LA, Shankaran S, Stevenson DK, Tyson JE, Poole WK; NICHD Neonatal Research Network. Trends in neonatal morbidity and mortality for very low birthweight infants. Am J Obstet Gynecol. 2007 Feb;196(2):147.e1-8. doi: 10.1016/j.ajog.2006.09.014[↩]
- Kaufman D, Boyle R, Hazen KC, Patrie JT, Robinson M, Donowitz LG. Fluconazole prophylaxis against fungal colonization and infection in preterm infants. N Engl J Med. 2001 Dec 6;345(23):1660-6. doi: 10.1056/NEJMoa010494[↩]
- Ohlsson A, Walia R, Shah SS. Ibuprofen for the treatment of patent ductus arteriosus in preterm or low birth weight (or both) infants. Cochrane Database Syst Rev. 2020 Feb 11;2(2):CD003481. doi: 10.1002/14651858.CD003481.pub8[↩]
- Su PH, Chen JY. Inhaled nitric oxide in the management of preterm infants with severe respiratory failure. J Perinatol. 2008 Feb;28(2):112-6. doi: 10.1038/sj.jp.7211881[↩]
- Lowe JR, Erickson SJ, Schrader R, Duncan AF. Comparison of the Bayley II Mental Developmental Index and the Bayley III Cognitive Scale: are we measuring the same thing? Acta Paediatr. 2012 Feb;101(2):e55-8. doi: 10.1111/j.1651-2227.2011.02517.x[↩]
- Principles of Biostatistics. Published in Marcello Pagano, Kimberlee Gauvreau, Heather Mattie, Principles of Biostatistics, 2022. https://doi.org/10.1201/9780429340512[↩]
- Sun H, Zhou Y, Xiong H, Kang W, Xu B, Liu D, Zhang X, Li H, Zhou C, Zhang Y, Zhou M, Meng Q. Prognosis of very preterm infants with severe respiratory distress syndrome receiving mechanical ventilation. Lung. 2015 Apr;193(2):249-54. doi: 10.1007/s00408-014-9683-5[↩][↩][↩][↩]
- Darlow BA, Ells AL, Gilbert CE, Gole GA, Quinn GE. Are we there yet? Bevacizumab therapy for retinopathy of prematurity. Arch Dis Child Fetal Neonatal Ed. 2013 Mar;98(2):F170-4. doi: 10.1136/archdischild-2011-301148[↩]
- Pasha AB, Chen XQ, Zhou GP. Bronchopulmonary dysplasia: Pathogenesis and treatment. Exp Ther Med. 2018 Dec;16(6):4315-4321. doi: 10.3892/etm.2018.6780[↩]
- Khawar H, Marwaha K. Surfactant. [Updated 2019 Sep 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK546600[↩]
- Canals Candela FJ, Vizcaíno Díaz C, Ferrández Berenguer MJ, Serrano Robles MI, Vázquez Gomis C, Quiles Durá JL. [Surfactant replacement therapy with a minimally invasive technique: Experience in a tertiary hospital]. An Pediatr (Barc). 2016 Feb;84(2):79-84.[↩]
- Kribs A. Minimally Invasive Surfactant Therapy and Noninvasive Respiratory Support. Clin Perinatol. 2016 Dec;43(4):755-771.[↩]
- Aldana-Aguirre JC, Pinto M, Featherstone RM, Kumar M. Less invasive surfactant administration versus intubation for surfactant delivery in preterm infants with respiratory distress syndrome: a systematic review and meta-analysis. Arch. Dis. Child. Fetal Neonatal Ed. 2017 Jan;102(1):F17-F23.[↩]
- Foglia EE, Jensen EA, Kirpalani H. Delivery room interventions to prevent bronchopulmonary dysplasia in extremely preterm infants. J Perinatol. 2017 Nov;37(11):1171-1179.[↩]
- SUPPORT Study Group of the Eunice Kennedy Shriver NICHD Neonatal Research Network; Finer NN, Carlo WA, Walsh MC, Rich W, Gantz MG, Laptook AR, Yoder BA, Faix RG, Das A, Poole WK, Donovan EF, Newman NS, Ambalavanan N, Frantz ID 3rd, Buchter S, Sánchez PJ, Kennedy KA, Laroia N, Poindexter BB, Cotten CM, Van Meurs KP, Duara S, Narendran V, Sood BG, O’Shea TM, Bell EF, Bhandari V, Watterberg KL, Higgins RD. Early CPAP versus surfactant in extremely preterm infants. N Engl J Med. 2010 May 27;362(21):1970-9. doi: 10.1056/NEJMoa0911783. Erratum in: N Engl J Med. 2010 Jun 10;362(23):2235.[↩]
- Committee on Fetus and Newborn; American Academy of Pediatrics. Respiratory support in preterm infants at birth. Pediatrics. 2014 Jan;133(1):171-4. doi: 10.1542/peds.2013-3442[↩]
- Roberts CT, Owen LS, Manley BJ, Frøisland DH, Donath SM, Dalziel KM, Pritchard MA, Cartwright DW, Collins CL, Malhotra A, Davis PG; HIPSTER Trial Investigators. Nasal High-Flow Therapy for Primary Respiratory Support in Preterm Infants. N Engl J Med. 2016 Sep 22;375(12):1142-51. doi: 10.1056/NEJMoa1603694[↩]
- Singh N, Halliday HL, Stevens TP, Suresh G, Soll R, Rojas-Reyes MX. Comparison of animal-derived surfactants for the prevention and treatment of respiratory distress syndrome in preterm infants. Cochrane Database Syst Rev. 2015 Dec 21;2015(12):CD010249. doi: 10.1002/14651858.CD010249.pub2[↩]
- Dargaville PA, Aiyappan A, Cornelius A, Williams C, De Paoli AG. Preliminary evaluation of a new technique of minimally invasive surfactant therapy. Arch Dis Child Fetal Neonatal Ed. 2011 Jul;96(4):F243-8. doi: 10.1136/adc.2010.192518[↩]
- Abdel-Latif ME, Osborn DA. Laryngeal mask airway surfactant administration for prevention of morbidity and mortality in preterm infants with or at risk of respiratory distress syndrome. Cochrane Database Syst Rev. 2011 Jul 6;(7):CD008309. doi: 10.1002/14651858.CD008309.pub2. Update in: Cochrane Database Syst Rev. 2024 Jan 25;1:CD008309. doi: 10.1002/14651858.CD008309.pub3[↩]
- Göpel W, Kribs A, Ziegler A, Laux R, Hoehn T, Wieg C, Siegel J, Avenarius S, von der Wense A, Vochem M, Groneck P, Weller U, Möller J, Härtel C, Haller S, Roth B, Herting E; German Neonatal Network. Avoidance of mechanical ventilation by surfactant treatment of spontaneously breathing preterm infants (AMV): an open-label, randomised, controlled trial. Lancet. 2011 Nov 5;378(9803):1627-34. doi: 10.1016/S0140-6736(11)60986-0[↩]
- Pinheiro JM, Santana-Rivas Q, Pezzano C. Randomized trial of laryngeal mask airway versus endotracheal intubation for surfactant delivery. J Perinatol. 2016 Mar;36(3):196-201. doi: 10.1038/jp.2015.177[↩]
- Minocchieri S, Berry CA, Pillow JJ; CureNeb Study Team. Nebulised surfactant to reduce severity of respiratory distress: a blinded, parallel, randomised controlled trial. Arch Dis Child Fetal Neonatal Ed. 2019 May;104(3):F313-F319. doi: 10.1136/archdischild-2018-315051. Epub 2018 Jul 26. Erratum in: Arch Dis Child Fetal Neonatal Ed. 2020 Mar;105(2):e1. doi: 10.1136/archdischild-2018-315051corr1[↩]
- Hatch LD, Grubb PH, Lea AS, Walsh WF, Markham MH, Whitney GM, Slaughter JC, Stark AR, Ely EW. Endotracheal Intubation in Neonates: A Prospective Study of Adverse Safety Events in 162 Infants. J Pediatr. 2016 Jan;168:62-66.e6. doi: 10.1016/j.jpeds.2015.09.077[↩]
- Tarawneh A, Kaczmarek J, Bottino MN, Sant’anna GM. Severe airway obstruction during surfactant administration using a standardized protocol: a prospective, observational study. J Perinatol. 2012 Apr;32(4):270-5. doi: 10.1038/jp.2011.89[↩]
- Aldana-Aguirre JC, Pinto M, Featherstone RM, Kumar M. Less invasive surfactant administration versus intubation for surfactant delivery in preterm infants with respiratory distress syndrome: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2017 Jan;102(1):F17-F23. doi: 10.1136/archdischild-2015-310299[↩]
- Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, Ohlsson A, Solimano A, Tin W; Caffeine for Apnea of Prematurity Trial Group. Caffeine therapy for apnea of prematurity. N Engl J Med. 2006 May 18;354(20):2112-21. doi: 10.1056/NEJMoa054065[↩]
- Kamath BD, Macguire ER, McClure EM, Goldenberg RL, Jobe AH. Neonatal mortality from respiratory distress syndrome: lessons for low-resource countries. Pediatrics. 2011 Jun;127(6):1139-46. doi: 10.1542/peds.2010-3212[↩]