Contents
What is nephroptosis
Nephroptosis (nephro = kidney; ptosis = falling) is also called floating kidney or wandering kidney, is an inferior displacement or dropping of the kidney. Nephroptosis occurs when the kidney slips from its normal position by more than 2 vertebral bodies (or >5 cm) during a position change from supine to upright because it is not securely held in place by adjacent organs or its covering of fat 1. Nephroptosis develops most often in very thin people whose adipose capsule or renal fascia is deficient. Nephroptosis is dangerous because the ureter may kink and block urine flow. The resulting backup of urine puts pressure on the kidney, which damages the tissue. Twisting of the ureter also causes pain. Nephroptosis is very common; about one in four people has some degree of weakening of the fibrous bands that hold the kidney in place. It is 10 times more common in females than males.
Nephroptosis is a fairly rare condition, and the number of radiological diagnoses exceeds the number of patients with symptoms attributable to the condition. Many studies have estimated that nearly 20% of women have nephroptosis revealed by routine intravenous urography (IVU), but far fewer (10%-20%) actually present with symptoms attributable to nephroptosis 2. The exact incidence of nephroptosis in the general population is unknown but intravenous urography (IVU) in thin females (where an upright image is included) will show renal mobility amounting to nephroptosis in up to 20% of cases 3. Symptomatic nephroptosis is more common in females, with male‐to‐female ratio of radiologically detected nephroptosis is ≈3:100, occurring most frequently in young, slim women. The right side is affected in ≈70% of cases, the left kidney in 10% and 20% have bilateral renal descent 4.
Of interest, nearly 64% of patients with fibromuscular dysplasia of the renal artery also have ipsilateral nephroptosis 5.
Surgical management is considered when symptoms such as obstruction of the collecting system or of renal blood flow are encountered 6. Nephropexy, either performed via laparoscopic or open surgery, is used to affix the affected kidney to retroperitoneal tissues 7. Long-term outcome studies have reported an improvement in the quality of life and reduction in pain after this surgery.
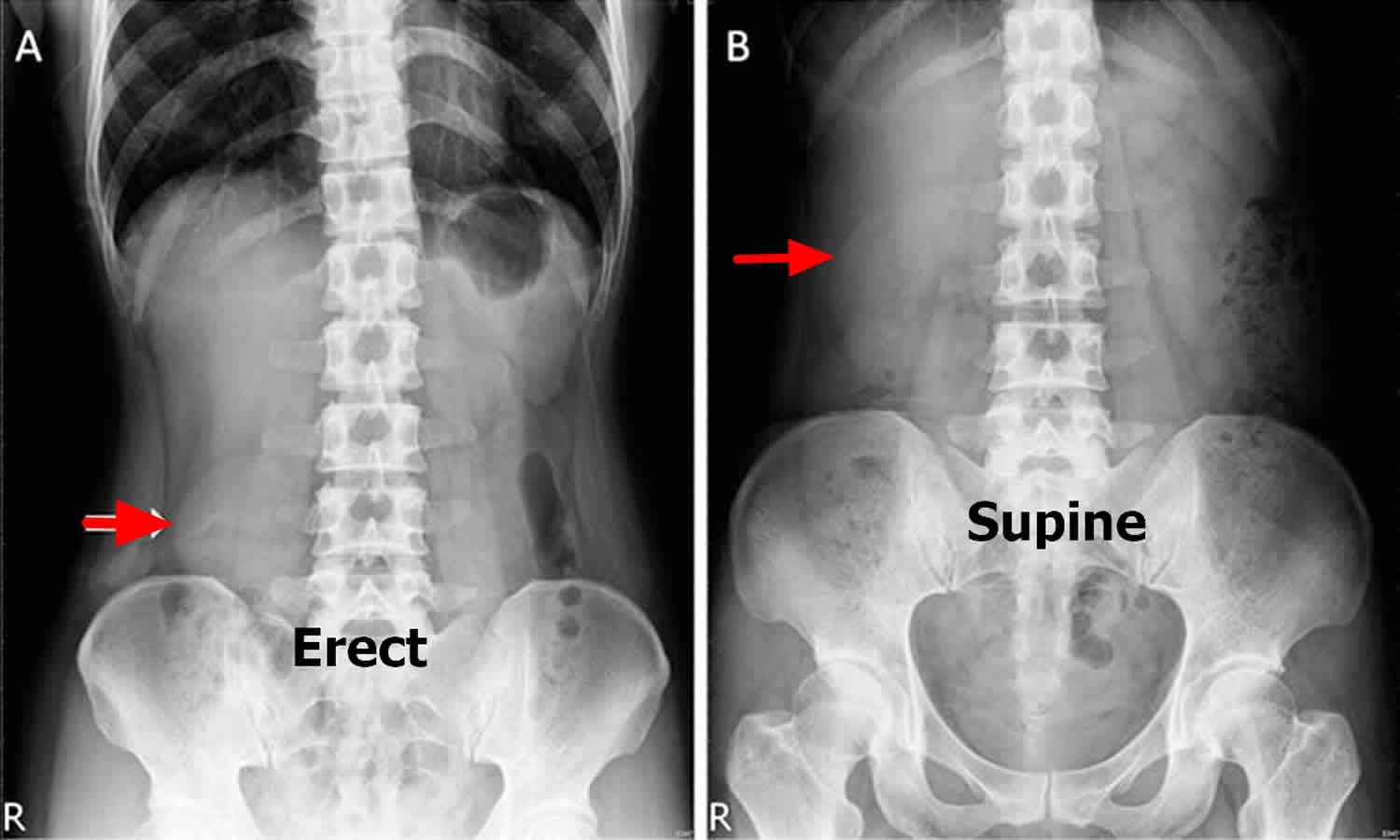
Figure 1. Nephroptosis
Footnote: A standing abdominal x-ray (A) showing a significant downward displacement (>5 cm) of the kidney (arrow). The supine abdominal x-ray (B) showing right kidney in the normal position. A 43-year-old woman presented with a 2-year history of an intermittent focal protruding abdominal mass. She could palpate the mass around the epigastrium when she lies down supine but not in an erect position, and it would disappear when she pushed it. She did not report abdominal pain, flank pain or haematuria. No abdominal mass was palpable during physical examination either in the supine or erect position. A standing abdominal radiograph showed a small and round-shaped right kidney descent (>5 cm) near the pelvis known as the ‘tennis ball kidney’ (Figure 1A, arrow). Nephroptosis was confirmed after a supine radiograph showed that the right kidney moved back into a normal anatomical site (Figure 1B arrow) and excluded an ectopic kidney. Her renal function was normal and urinalysis was unremarkable. A renal ultrasonography showed no hydronephrosis. Because she was relatively asymptomatic, only observation with follow-up was recommended.
[Source 8 ]Nephroptosis causes
The cause of nephroptosis is relatively unknown, but there are similarities among most symptomatic patients. Typical cases of nephroptosis usually involve white females who are thin, and it is theorized that a lack of perirenal fat and fascial support can lead to the downward translocation of the kidney. In addition, patients with nephroptosis often present with a longer-than-normal renal vascular pedicle, which allows for renal displacement in the sagittal and frontal axes 9. Rarely, nephroptosis may occur in a kidney transplant recipient 10.
Each kidney with its fibrous capsule rests in a layer of perinephric fat, encompassed by the thick Gerota’s fascia. Gerota’s fascia is separated from the muscles of the posterior abdominal wall by another layer of adipose tissue (paranephric fat). Although the aetiology of nephroptosis is not completely understood, the excessive mobility of the kidney is probably related to deficient support from these perinephric structures, permitting excessive renal mobility. Therefore malrotation or maldescent of the kidney can result in either stretching or torsion of the hilar vessels and/or kinking of the pelvic ureteric junction or the proximal ureter.
Studies in children suggested that nephroptosis is a constitutional rather than an acquired phenomenon, and it is likely that the condition precedes the onset of symptoms, usually in adult life, by several decades 11.
The current theory attributes the pain in symptomatic nephroptosis to (1) acute hydronephrosis caused by kinking of the proximal ureter caused by the sudden descent of the kidney, (2) lumen narrowing and transient renal ischemia due to elongation of the renal vessels, (3) visceral nerve stimulation due to traction on the renal hilum 5 and/or (4) symptoms due to secondary pathology (e.g. pyelonephritis, renal calculi) 12.
Nephroptosis symptoms
Nephroptosis, also known as floating kidney and renal ptosis, is a condition in which the kidney descends more than 2 vertebral bodies (or >5 cm) during a position change from supine to upright. In the upright position, this translocation can lead to symptoms of vomiting and acute abdominal pain due to acute obstruction or ischemia of the kidney.
The typical clinical presentation of symptomatic nephroptosis involves a young (aged 20-40 years) thin woman presenting with costovertebral, flank, or lower-quadrant abdominal pain that occurs in the upright position and that is relieved by lying down and exacerbated by long periods of standing or physical activity 13. Many patients with the condition may also seek care upon palpating a lower-abdominal mass while in the upright position 5.
The most frequently associated symptoms are:
- pain, the principal complaint (90% of patients), usually a ‘dragging’ pain in the flank or abdomen, typically when upright, and relieved by recumbency. Acute episodes might present as renal colic, with no detectable ureteric calculi;
- nausea and vomiting are frequently present, probably due to visceral autonomic nerves stimulation;
- an abdominal mass might be noted when upright in thin patients, and manipulation of the kidney ‘mass’ into its anatomical position might result in resolution of the symptoms. Occasionally, a slim patient might be able themselves to manipulate their kidney into a lower position.
- transient hematuria, both frank and microscopic;
- a history of repeated UTI (urinary tract infection) (up to a third of patients), renal calculi, and hypertension (≈10%).
Risk factors for developing symptomatic nephroptosis include excessive weight loss or frequent extreme physical activity.
The most severe manifestation of symptomatic nephroptosis is Dietl crisis. Dietl’s crisis is a syndrome attributed to acute hydronephrosis due to kinking or obstruction of the ureter of a ‘floating kidney’, and in its classical form is characterized by violent paroxysms of colicky flank pain, nausea, chills, tachycardia, oliguria, transient haematuria or proteinuria, and a palpable enlarged tender kidney 14. The immediate resolution of Dietl’s crisis was based on gentle manual reduction of the kidney into the renal fossa while the patient was supine and with the head down and feet elevated or the patient adopting a supine knee‐chest position 5.
Pertinent questions in the medical history include the following 9:
- Is pain exacerbated by standing up from a supine position?
- Does severe physical activity exacerbate the pain?
- Is the pain alleviated by lying in a supine position?
- Have you had significant weight loss in your lifetime?
- Have you recently had hematuria, recurrent urinary tract infections, renal calculi, or hypertension?
- Can you feel any masses in your lower abdomen on the same side as your pain?
Upon physical examination, at times, the ptotic kidney can be palpated in the ipsilateral lower abdomen when the patient assumes an upright position. The anterior abdominal wall may also show some minor indenting 9.
Renal ptosis and renal ectopia can be included on the same differential, as both are inferior renal displacements of greater than 2 vertebral bodies from L2 (right kidney) and 1 cm higher than L2 (left kidney). However, renal ectopia is a permanent congenital displacement of the kidney to this position, and patients with this condition have shorter ureters and an ectopic renal arterial blood supply. Ptotic kidneys have normal-length ureters and renal arteries with a normal origin from the abdominal aorta, whereas an ectopic kidney has a shorter ureter given its fixed interior position 15.
Other differential diagnoses include the following 9:
- Renal colic with associated urolithiasis
- Cholecystitis (right side)
- Intermittent bowel obstruction
- Spastic bowel disease
- Hematuria
- Pyelonephritis
- Ovarian vein syndrome
- “Nutcracker” syndrome (compression of the left renal vein between the superior mesenteric artery and the abdominal aorta)
- Ovarian cystic disease and possible ruptured ovarian cyst
- Chronic appendicitis or diverticulitis
Nephroptosis diagnosis
The initial assessment must include a detailed history, enquiring specifically about the symptoms mentioned above; full general and abdominal examination; blood pressure measurement, heart rate and temperature. A high index of suspicion is vital in arriving at a prompt and correct diagnosis. Nephtoptosis is a ‘diagnosis of exclusion’ and is usually arrived at after excluding other commoner conditions such as renal/ureteric calculi, pelvic ureteric junction obstruction and recurrent pyelonephritis.
Laboratory studies
Urinalysis may reveal microhematuria in patients with nephroptosis. However, the workup for microhematuria should be completed in a standard fashion before nephroptosis is implicated as its cause.
Urine culture results are typically negative.
BUN, creatinine, and electrolyte levels are typically normal.
Blood lactate dehydrogenase levels may be acutely elevated, supporting the theory of transient renal ischemia 16.
Imaging Studies
Intravenous urography is the primary diagnostic tool in nephroptosis. In patients with nephroptosis, intravenous urography shows a renal descent of two or more vertebral bodies (i.e, >5 cm) when images taken in the supine to upright positions are compared. Delayed imaging may also show hydronephrosis 7.
In patients with nephroptosis, diuretic (furosemide) renography may show hydronephrosis and/or decreased split renal function when scans taken in the supine position are compared with those taken in the sitting position 17.
Retrograde pyelography can demonstrate obstructive changes in the ureter, pelvis, or calyces (e.g., kinking) when the renal position is changed by tilting the operative table 5.
CT scan findings are usually normal when the patient is in the supine position 7.
Supine to upright ultrasonography and Doppler ultrasonography scans can show renal ptosis, while the Doppler study can show diminished blood flow in the ptotic kidney when the patients is upright. This method may yield sensitivity superior to that of isotope renography (renal scanning) in detecting symptomatic nephroptosis 18.
When scanning is performed in both a supine and sitting position, it may show decreased blood flow and reduced clearance 19.
Nephroptosis treatment
Nephroptosis should be treated only in the rare patients who present with a full array of symptoms and positive diagnosis via one of the imaging studies described above.
Owing to hypochondriac behaviors that often accompany symptomatic nephroptosis, nonsurgical treatments are recommended as a first-line intervention. Some such treatments include weight gain, therapy with gastrointestinal medications, abdominal wall–strengthening exercises, abdominal wall binders (eg, corsets), and frequent rests in the supine position. Extracorporeal support of the ptotic kidney appears to be the most effective nonsurgical treatment, and abdominal binders and corsets were in common use throughout the 1900s 20. Similar to a truss for treating a hernia, these approaches are all temporizing maneuvers.
Nephropexy is indicated in a very small percentage of nephroptosis cases. It is reserved only for symptomatic patients with flank pain (often >1 year in duration) in whom studies confirm renal descent upon transition from a supine to an erect position and in whom intravenous urography, renal ultrasonography, or nuclear renal scintigraphy shows delayed excretion and hydronephrosis 21.
Surgical therapy
All surgical therapies for nephroptosis are based on the principle that the ptotic kidney must be irreversibly fixed into its normal position by securing the kidney or perinephric tissue (renal capsule, perirenal fat, Gerota fascia) to the body wall high in the retroperitoneum via either suture or subsequent adhesion formation. A few of the prominent surgical therapies are discussed below.
Surgical intervention can be divided into open nephropexy (ON), percutaneous nephropexy (PCN) and LN. The principles of an optimal nephropexy procedure, irrespective of whether the approach is laparoscopic or open, are:
- complete nephrolysis (complete exposure of kidney within Gerota’s fascia) and release of existing attachments to the peritoneum;
- immobilization of the kidney in a more cephalad retroperitoneal position;
- relief of any associated urinary obstruction;
- fixation of the renal axis with no tension.
Nephropexy
This procedure is used to affix the kidney to the retroperitoneal tissues via open or laparoscopic surgery. The renal capsule is attached to the psoas or quadratus lumborum muscle with nonabsorbable sutures, fascial or muscle bands, and/or polyglactin mesh 7. This procedure was pioneered by Bassini and is currently the most commonly used open surgical nephropexy technique. Using the 12th rib as a sling or skewer has generally fallen out of favor.
Endourological procedures
In the last 20 years, the endourological procedures laparoscopic nephropexy and circle (U) nephrostomy tube insertion have gained favor as surgical treatments for nephroptosis 22.
Laparoscopic nephropexy
This is a laparoscopic procedure that closely duplicates the open nephropexy described above but that results in significantly lower morbidity. Laparoscopic nephropexy confers a significant and durable improvement in quality of life in approximately 70%-90% of patients who undergo the procedure 23.The technique is practiced at many centers with extensive laparoscopic experience.
In 1997, Fornara and colleagues 24 compared open to laparoscopic nephropexy. The mean operative time was 49 minutes and 61 minutes, respectively. Postoperative analgesic control was measured to be an average of 15 mg morphine equivalents in the laparoscopic group and 38 mg in the open group. In the laparoscopic group, the average hospital stay was 3.7 days, versus 16 days in the open surgery group. In addition, split renal function improved from 38% to 47% postoperatively in the laparoscopic group.
Multiple laparoscopic procedural techniques exist, including the use of absorbable mesh, fixation staples, and/or sutures placed through Gerota fascia and sutures through the renal capsule. Many studies have shown that tissue adhesives (eg, butyl cyanoacrylate) and medical devices (eg, tension-free vaginal tape) can also be used to secure the kidney high in the retroperitoneum 25.
Multiple long-term outcome studies have proven the efficacy of laparoscopic nephropexy 26. These reports, with follow-up times ranging from 3.3-8.2 years, showed that, postprocedure, 71% of patients reported an improvement in quality of life and showed an 80%-91% reduction in pain.
The original laparoscopic procedure, known as the Washington University technique (transperitoneal laparoscopic retroperitoneal nephropexy), is described in details. In this procedure, first described by Szekely and colleagues in 1997, a 14F-16F nephrostomy tube is placed through two points in the middle and upper calices, with the tube entering above the 12th rib and exiting below the 12th rib, forming a sling used to suspend the kidney to the 12th rib. The tube is then sutured to the skin with moderate tension and left in place for 2-3 weeks to allow sufficient scar formation for kidney fixation 27.
In a long-term outcome study of 90 patients over 12 years, Szekely and colleagues were able to show a 79% long-term success rate with nephrostomy tube insertion based on pain intensity. They reported a mean operative time of 18 minutes and an average hospital stay of 3 days for the procedure. This method, although lacking in widespread application, is considered a low-morbidity, easy-to-perform, cost- and time-effective alternative to laparoscopic nephropexy by those adept in its use 28. However, because this technique is not widespread in application, corroborating data from other institutions as to its success are absent.
- Nephroptosis. https://emedicine.medscape.com/article/1458935-overview[↩]
- Plas E, Daha K, Riedl CR, Hubner WA, Pfluger H. Long-term followup after laparoscopic nephropexy for symptomatic nephroptosis. J Urol. 2001 Aug. 166(2):449-52 [↩]
- Narath PA. Nephroptosis. Urol Int 1961; 12: 164–71[↩]
- Plas E, Daha K, Riedl CR, Hunber WA, Pfluger H. Long‐term follow‐up after laparoscopic nephropexy for symptomatic nepnroptosis. J Urol 2001; 166: 449– 5[↩]
- Hoenig DM, Hemal AK, Shalhav AL, Clayman RV. Nephroptosis: a “disparaged” condition revisited. Urology. 1999 Oct. 54(4):590-6[↩][↩][↩][↩][↩]
- Chan VSH, Lam TPW, Lam WWM. Nephroptosis: The wandering kidney. Kidney Res Clin Pract. 2018;37(3):306–307. doi:10.23876/j.krcp.2018.37.3.306 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6147184[↩]
- Winfield H. Nephroptosis. The 5-Minute Urology Consult. Philadelphia: Lippincott Williams and Wilkins; 2000. Vol 1: 368-9.[↩][↩][↩][↩]
- Lim KH, Chang YH. Tennis ball kidney of nephroptosis. BMJ Case Rep. 2011;2011:bcr0720103174. Published 2011 Feb 18. doi:10.1136/bcr.07.2010.3174 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3062823[↩]
- Winfield H. Nephroptosis. The 5-Minute Urology Consult. Philadelphia: Lippincott Williams and Wilkins; 2000. Vol 1: 368-9[↩][↩][↩][↩]
- Dosch AR, Pahl M, Reddy U, Foster CE 3rd, Ichii H. Post-transplantation nephroptosis causing recurrent episodes of acute renal failure and hypertension secondary to intermittent vascular torsion of intraperitoneal renal allograft. J Surg Case Rep. 2017 May. 2017 (5):rjx033[↩]
- Prandota J, Ostrowska‐Skora J. Normal limits for renal mobility in children. Int J Pediatr Nephrol 1984; 5: 171–4[↩]
- Srirangam, S. J., Pollard, A. J., Adeyoju, A. A. and O’Reilly, P. H. (2009), Nephroptosis: seriously misunderstood?. BJU International, 103: 296-300. doi:10.1111/j.1464-410X.2008.08082.x https://onlinelibrary.wiley.com/doi/pdf/10.1111/j.1464-410X.2008.08082.x[↩]
- Bishoff JT, Kavoussi LR. Nephropexy. Campbell-Walsh Urology. Philadelphia: Saunders Elsevier; 2007. 9(2): 1776-8; Ch 51.[↩]
- Dietl J. Wanderende Nieren und deren Einkleemmung. WienMed Wschr 1864; 14: 563, 579, 593[↩]
- Friedenberg RM, Harris RD. Techniques and applications of urological imaging. Clinical Urography. Philadelphia: Saunders; 2000. 2(1): 200; Ch 6.[↩]
- Nakada SY, McDougall EM, Clayman RV. Laparoscopic Nephropexy. Smith’s Textbook of Endourology. St. Louis: Quality Medical; 1996. 1(2): 945-7; Ch 58.[↩]
- Tartaglione G, D’Addessi A, De Waure C, Pagan M, Raccioppi M, Sacco E, et al. (99m)Tc-MAG3 diuretic renography in diagnosis of obstructive nephropathy in adults: a comparison between F-15 and a new procedure F+10(sp) in seated position. Clin Nucl Med. 2013 Jun. 38(6):432-6[↩]
- Strohmeyer DM, Peschel R, Effert P, et al. Changes of renal blood flow in nephroptosis: assessment by color Doppler imaging, isotope renography and correlation with clinical outcome after laparoscopic nephropexy. Eur Urol. 2004 Jun. 45(6):790-3[↩]
- Landman J, McDougall EM, Gill IS, Clayman RV. Retroperitoneum: Nephropexy. Adult and Pediatric Urology. Philadelphia: Lippincott Williams and Wilkins; 2002. 4(1): 707-9; Ch 18.[↩]
- Hoenig DM, Hemal AK, Shalhav AL, Clayman RV. Nephroptosis: a “disparaged” condition revisited. Urology. 1999 Oct. 54(4):590-6.[↩]
- Murari SB, Gadepalli T, Rao VP, Ram R. Renal scintigraphy in diagnosis and management of nephroptosis. Indian J Nucl Med. 2012 Jan. 27(1):52-4[↩]
- Hoenig DM, Hemal AK, Shalhav AL, Clayman RV. Nephroptosis: a “disparaged” condition revisited. Urology. 1999 Oct. 54(4):590-6 [↩]
- Bansal D, Defoor WR Jr, Noh PH. Pediatric robotic assisted laparoscopic nephropexy: case study. Springerplus. 2013. 2:321[↩]
- Fornara P, Doehn C, Jocham D. Laparoscopic nephropexy: 3-year experience. J Urol. 1997 Nov. 158(5):1679-83[↩]
- Hübner WA, Schlarp O, Riedl C, Plas E, Reiter WJ. Laparoscopic nephropexy using tension-free vaginal tape for symptomatic nephroptosis. Urology. 2004 Aug. 64(2):372-4[↩]
- Vodopija N, Korsic L, Zupancic M, Kramer F, Krstanoski Z, Parac I. Is laparoscopic nephropexy improving the quality of life. Coll Antropol. 2007 Sep. 31(3):689-92[↩]
- Szekely JG. Re: Laparoscopic nephropexy: Washington University experience. J Urol. 1997 Jan. 157(1):266[↩]
- Szekely J, Bagheri F, Villa nyi K, Pusztai C, et al. Percutaneous nephropexy with U-tube nephrostomy: long-term follow-up of an alternative technique for treatment of symptomatic nephroptosis. Urology. 2006. 68 (Supplement 5A):227[↩]