Contents
What is serum osmolality
Osmolality is a measure of the number of dissolved particles in a fluid. A test for osmolality measures the amount of dissolved substances such as sodium, potassium, chloride, glucose, and urea in a sample of blood and sometimes in urine. Alternatively, it can be estimated from the major solutes expected to be in the blood or urine.
Osmolality is dynamic and will fluctuate as the body responds to and corrects temporary water imbalances. Serum and urine osmolality tests must be evaluated in the context of the person’s signs and symptoms and along with the findings of other tests, such as sodium, glucose, and blood urea nitrogen (BUN). Osmolality results are not diagnostic; they suggest that a person has an imbalance, but they do not pinpoint the cause.
Water balance in the body is a dynamic process that is regulated by controlling the amount of water eliminated in the urine by the kidneys and by increasing or decreasing water drinking by regulating “thirst.” In a healthy person, the body perceives and reacts to changes in the amount of water and particles in the blood.
When blood osmolality increases with a decrease in the amount of water in the blood or an increase in the number of particles such as sodium, chloride, and glucose, a gland called the hypothalamus releases antidiuretic hormone (ADH). The kidneys respond to antidiuretic hormone (ADH) by conserving water and producing urine that is more concentrated. The retained water dilutes the blood and lowers blood osmolality back to normal. This also increases blood volume and blood pressure. If this is not sufficient to restore the water balance, then thirst is also stimulated so that the affected person will drink more water.
When blood osmolality decreases, the release of antidiuretic hormone (ADH) is suppressed, the kidneys release more dilute urine, the amount of water in the body decreases, thirst is diminished, and blood osmolality increases back toward normal.
A blood (serum) osmolality test is primarily a measure of sodium dissolved in the serum (the liquid portion of blood). Sodium is the major electrolyte in the blood and urine. It works with potassium, chloride, and CO2 (in the form of bicarbonate) to maintain electrical neutrality in the body and acid-base balance. Sodium comes into the body in the diet and is normally conserved or eliminated in the urine by the kidneys to maintain its concentration in the blood within a healthy range.
In addition to electrolytes, glucose and urea contribute to osmolality. Normally their contributions are small, but when someone has high blood glucose (hyperglycemia, as found in untreated diabetes) or high blood urea (seen in diseases such as kidney failure), their influence can be significant.
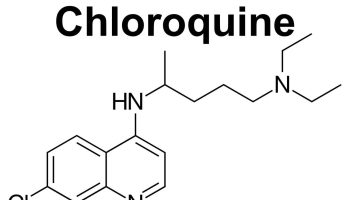
Serum osmolality is often used in cases of suspected poisoning or overdose. Toxins such as methanol, isopropyl alcohol, ethylene glycol, propylene glycol, and acetone, and drugs such as salicylates (aspirin) can also affect osmolality when ingested in sufficiently large amounts.
The osmolality of serum increases with dehydration and decreases with overhydration. The patient receiving intravenous fluids should have a normal osmolality. If the osmolality rises, the fluids contain relatively more electrolytes than water. If the osmolality falls, relatively more water than electrolytes is being administered.
A urine osmolality test primarily measures the waste products urea and creatinine. Urea and creatinine are produced and removed by the body at a relatively constant rate.
A serum osmolal gap (osmotic gap) may also be calculated. It is the difference between measured and calculated (estimated) osmolality results. In order to calculate the osmolal gap, tests for blood sodium, blood urea nitrogen (BUN), and glucose must be performed to calculate the expected osmolality. Some versions of the expected osmolality calculation also include the measurement of ethanol. An increase in the osmolal gap (greater than 10) indicates the presence of substances such as toxins, aspirin (salicylates), or mannitol.
How is the osmolality test used?
The blood osmolality test is primarily used to help determine whether a person has ingested a toxin such as methanol or ethylene glycol (antifreeze). Sometimes it may be used to investigate low blood sodium and your body’s water balance. Osmolality may be measured directly or estimated using a calculation.
Serum osmolality testing may be ordered when a person has an unexplained low blood sodium or signs and symptoms that a healthcare practitioner suspects may be due to low blood sodium such as:
- Excessive thirst
- Confusion
- Nausea
- Headache
- Lethargy
- In severe cases, seizures or coma
In addition to osmolality, the osmolal gap (osmotic gap) may be calculated and used to detect and/or measure toxins in the blood, such as methanol, ethylene glycol, isopropyl alcohol, and propylene glycol.
Urine osmolality may be used along with serum osmolality to help evaluate the body’s water balance and to investigate increased and decreased urination. Urine sodium and creatinine are often ordered along with urine osmolality. Sometimes a urine osmolal gap is calculated and used to help evaluate the kidney’s ability to eliminate acid and reabsorb bicarbonate, to detect the presence of osmotically active molecules, and to compare with the serum osmolal gap.
Osmolality vs osmolarity
Osmolality measures the concentration of solutes in a fluid by looking at the number of particles per weight (kilogram) of fluid (Osm/kg). Osmolarity evaluates the number of particles per volume (liter) of fluid (Osm/L). With dilute fluids, they are essentially the same, but differences will be more noticeable at higher concentrations and care must be taken when comparing them as the units for osmolality and osmolarity are not the same.
The conversion of osmolarity into osmolality by multiplying with the mass density of solvent in solution (kg solvent/kg solution).
Osmolarity = osmolality x (ρsol − ca)
where:
- ρsol is the density of the solution in g/ml, which is 1.025 g/ml for blood plasma
- ca is the (anhydrous) solute concentration in g/ml – not to be confused with the density of dried plasma
Serum osmolality equation
Serum Osmolality (mOsm/kg H2O)= 2 x [Na+] + (Glucose/18) + (BUN/2.8) + (Ethanol/3.8)
Note: Ethanol not always included. Glucose, BUN, and ethanol may be reported in mg/dL (milligrams per deciliter) or mmol/L (millimole per liter). The numbers shown in the equation above are used to convert from mg/dL to mmol/L. For mmol/L, the equation would be: 2 x [Na+] + (Glucose) + (BUN) + (Ethanol)
Serum osmolal gap calculation
Serum osmolal gap = serum osmolality (measured) – serum osmolality (calculated)
Normal serum osmolality
Normal plasma osmolality values range from 275 to 295 mOsm/kg (275 to 295 mmol/kg).
Normal value ranges may vary slightly among different laboratories. Some labs use different measurements or test different samples. Talk to your provider about the meaning of your specific test results.
High serum osmolality
In general, increased serum osmolality may be due to either decreased water in the blood or increased solutes. Examples of conditions in which blood (serum) osmolality may be increased include:
- Toxic ingestion of ethanol, methanol, ethylene glycol, isopropyl alcohol or aspirin (salicylates), for example
- Water loss (dehydration)
- Diabetes
- Increased nitrogen waste products in the blood (uremia)
- Stroke or head trauma that leads to decreased antidiuretic hormone (ADH) secretion
- Kidney damage and disease
- Mannitol therapy–used in the treatment of brain swelling (cerebral edema)
- Shock
- Diabetes insipidus
- High blood sugar level (hyperglycemia)
- High level of nitrogen waste products in the blood (uremia)
- High blood sodium level (hypernatremia)
Low serum osmolality
In general, a decreased serum osmolality may be due to increased fluids. Examples of conditions causing decreased serum osmolality include:
- Excess hydration (drinking excessive amounts of water, water retention or decreased ability of the kidneys to produce urine)
- Decreased blood sodium
- Antidiuretic hormone (ADH) oversecretion
- Adrenal gland not working normally
- Conditions linked to lung cancer
- Drinking too much water or fluid
- Low sodium level (hyponatremia)
- Underactive thyroid gland (hypothyroidism)
Urine osmolality
Urine osmolality is a measure of the concentration of osmotically active particles, principally sodium, chloride, potassium, and urea; glucose can contribute significantly to the osmolality when present in substantial amounts in urine. Urinary osmolality corresponds to urine specific gravity in nondisease states.
The ability of the kidney to maintain both tonicity and water balance of the extracellular fluid can be evaluated by measuring the osmolality of the urine either routinely or under artificial conditions. More information concerning the state of renal water handling or abnormalities of urine dilution or concentration can be obtained if urinary osmolality is compared to serum osmolality and if urine electrolyte studies are performed. Normally, the ratio of urine osmolality to serum osmolality is 1.0 to 3.0, reflecting a wide range of urine osmolality.
Urine osmolality is often evaluated in the context of how much urine the person is producing. Increased amounts of urine may be due to increased fluid intake, lack of appropriate amounts of ADH, or diabetes, with increased glucose levels leading to increased urine output. Decreased amounts of urine may be due to a variety of causes, including decreased blood flow to the kidneys, an appropriate response to dehydration, or damage to tubular cells in the kidneys.
- When a person produces increased amounts of urine and urine osmolality is low, then the person either is ridding their body of excess fluids or is unable to concentrate urine appropriately.
- Increased amounts of urine and a high osmolality may be seen when there is a substance being flushed from the body, such as excess glucose with diabetes.
- If a person produces decreased amounts of urine and has high urine osmolality, then the person may be dehydrated
- If someone produces decreased amounts of urine and has low or normal urine osmolality, the person may have kidney damage.
A urine osmolality test may be ordered along with serum osmolality testing when a health practitioner wants to compare urine results with the serum osmolality and/or when the person being tested is producing increased or decreased amounts of urine.
Examples of conditions that can cause increased urine osmolality include:
- Dehydration
- Congestive heart failure
- Increased sodium
- Inappropriate ADH secretion
- Adrenal insufficiency/Addison’s disease
- Liver damage
- Shock
- Glucose in the urine (from uncontrolled diabetes)
Examples of conditions that can cause decreased urine osmolality include:
- Aldosteronism
- Diabetes insipidus
- Drinking excess fluids
- Increased blood calcium
- Decreased blood potassium
- Kidney disease or kidney failure e.g., renal tubular necrosis or severe pyelonephritis
Normal urine osmolality
- Age 0-11 months: 50-750 mOsm/kg
- Age > or =12 months: 150-1,150 mOsm/kg
With normal fluid intake and normal diet, a patient will produce a urine of about 500 to 850 mosmol/kg water. Above age of 20 years there is an age dependent decline in the upper reference range of approximately 5 mOsm/kg/year.
The normal kidney can concentrate a urine to 800 to 1,400 mosmol/kg and with excess fluid intake, a minimal osmolality of 40 to 80 mosmol/kg can be obtained.
With dehydration, the urine osmolality should be 3 to 4 times the plasma osmolality.
What is the difference between urine osmolality and specific gravity?
Specific gravity is a common part of a urinalysis. It evaluates the weight of solids in water. Osmolality and specific gravity usually change in parallel to each other. When large and heavy molecules (such as glucose and protein) are present in the urine, however, the results will diverge. Specific gravity will be increased more, due to the weight of the molecules, while urine osmolality will be increased less, reflecting the number of molecules. Generally, osmolality is considered a more exact measurement of urine concentration than specific gravity.