Contents
Peliosis
Peliosis is rare benign blood vessel condition characterized by multiple, randomly distributed, blood-filled and cyst-like cavities throughout the tissue of solid organs. Peliosis is a term derived from the Greek pelios, which means “dusky” or “purple,” referring to the color of the liver tissue with peliosis 1. Similar blood-filled spaces may be seen in the spleen (splenic peliosis), lymph nodes, and other organs including the bone marrow, lungs, pleura, kidneys, adrenal glands, stomach, and ileum 2, 3.
Peliosis was first described in the liver (peliosis hepatis) by Wagner in 1861, and was later named by Schoenland in 1916 4, 5. Although the pathogenesis of peliosis is still unknown, several hypotheses have been studied. Some researchers reported that toxic or viral substances induce hepatocellular necrosis with a sinusoidal dilatation, while others proposed a congenital vascular malformation or an active vessel proliferation 6.
Peliosis is generally understood to develop within organs of the mononuclear phagocytic system such as the liver, spleen, marrow, and lymph nodes of the abdomen 2.
Preliminary evidence links the development of peliosis to angiogenesis (formation of new blood vessels) due to elevated levels of vascular endothelial growth factor (VEGF) 7. VEGF is a fundamental regulator of physiologic angiogenesis. Elevated VEGF levels are known to be associated with tumor progression and poor outcomes in a number of cancers. A condition resembling peliosis has been demonstrated in mice with elevated VEGF levels, that were found to have massively dilated hepatic sinusoids and endothelial cell proliferation and apoptosis 7. The experimental link to elevated VEGF levels, warrants investigation into the treatment of peliosis with anti-angiogenic factors 2.
Peliosis hepatis
Peliosis hepatis also called “peliosis hepatitis” is rare benign blood vessel condition characterized by multiple, randomly distributed, blood-filled and cyst-like cavities throughout the liver 8, 9, 10, 11, 4. These blood-filled cavities communicate with the hepatic sinusoids and are associated with rupture of the reticulin framework 10. The size of the cystic lesions may vary from 1 millimeter to several centimeters 1. The cystic lesions are often irregularly shaped and in many cases are incompletely lined with endothelium 12. Peliosis hepatis has been described as typically involving the entire liver 1.
Peliosis mostly exists without symptoms (asymptomatic) being incidentally found on imaging or during an autopsy 13. However, with the progress of modern medical imaging technologies such as ultrasound, magnetic resonance imaging (MRI), FDG-PET (fluorodeoxyglucose positron emission tomography) or computed tomography (CT) scan, peliosis hepatis is now more frequently encountered in clinical practice 14. Despite these various medical imaging modalities, imaging may be inadequate for establishing a clear diagnosis of peliosis 15, 16.
Peliosis hepatis can occur at any age. Although a fetal form exists, peliosis hepatis usually develops in adults without regard to sex 1.
The cause of peliosis hepatis can be related to drugs including anabolic steroids, oral contraceptives, corticosteroids, tamoxifen, diethylstilbestrol, azathioprine, 6-thioguanine, 6-mercaptopurine, and methotrexate; toxins such as polyvinyl chloride, arsenic, and thorium oxide; chronic wasting diseases (e.g., tuberculosis, leprosy, and various cancers, particularly hepatocellular carcinoma); liver and kidney transplantation; and Bartonella henselae and Bartonella quintana infection in AIDS so-called bacillary peliosis 3, 17, 18, 19, 1. In addition, several other conditions are described as associated with peliosis hepatis, including sprue (a disorder in which the intestines are unable to absorb nutrients from food), diabetes mellitus, necrotizing vasculitis, and blood disorders including multiple myeloma, aplastic anemia, Hodgkin disease, and myelofibrosis 1. Moreover, peliosis hepatis may develop after kidney or heart transplantation. In 20 to 50% of patients, no associated condition is identified 1.
Peliosis hepatis is very difficult to diagnose. The diagnosis of peliosis hepatis is often missed or delayed because it is usually asymptomatic, and its radiologic appearance closely resembles a benign and malignant/pre-malignant lesions or multiple abscesses 20, 6. Radiological imaging shows a broad spectrum of appearances because of its dependence on the blood supply to the lesions. Laparoscopy is a useful examination to sporadically detect dark-blue or dark-red patchy patterns on the liver surface, which is a characteristic finding of peliosis hepatis 21. Pathological findings are considered the gold standard for the diagnosis of peliosis hepatis, being histologically characterized by blood-filled cavities. However, a case has been reported in which repeated biopsies were required to diagnose peliosis hepatis 22.
The correct diagnosis of peliosis hepatis is important because withdrawal of the offending drug or toxin can resolve the disease and prevent serious complications such as hepatic failure or death related to intraabdominal hemorrhage 1. Because of peliosis hepatis potential complications, surgical resection of the involved liver parenchyma should always be considered. In HIV-related peliosis hepatis caused by Bartonella henselae, clinical improvement has been documented with the use of antibiotics (i.e., erythromycin).
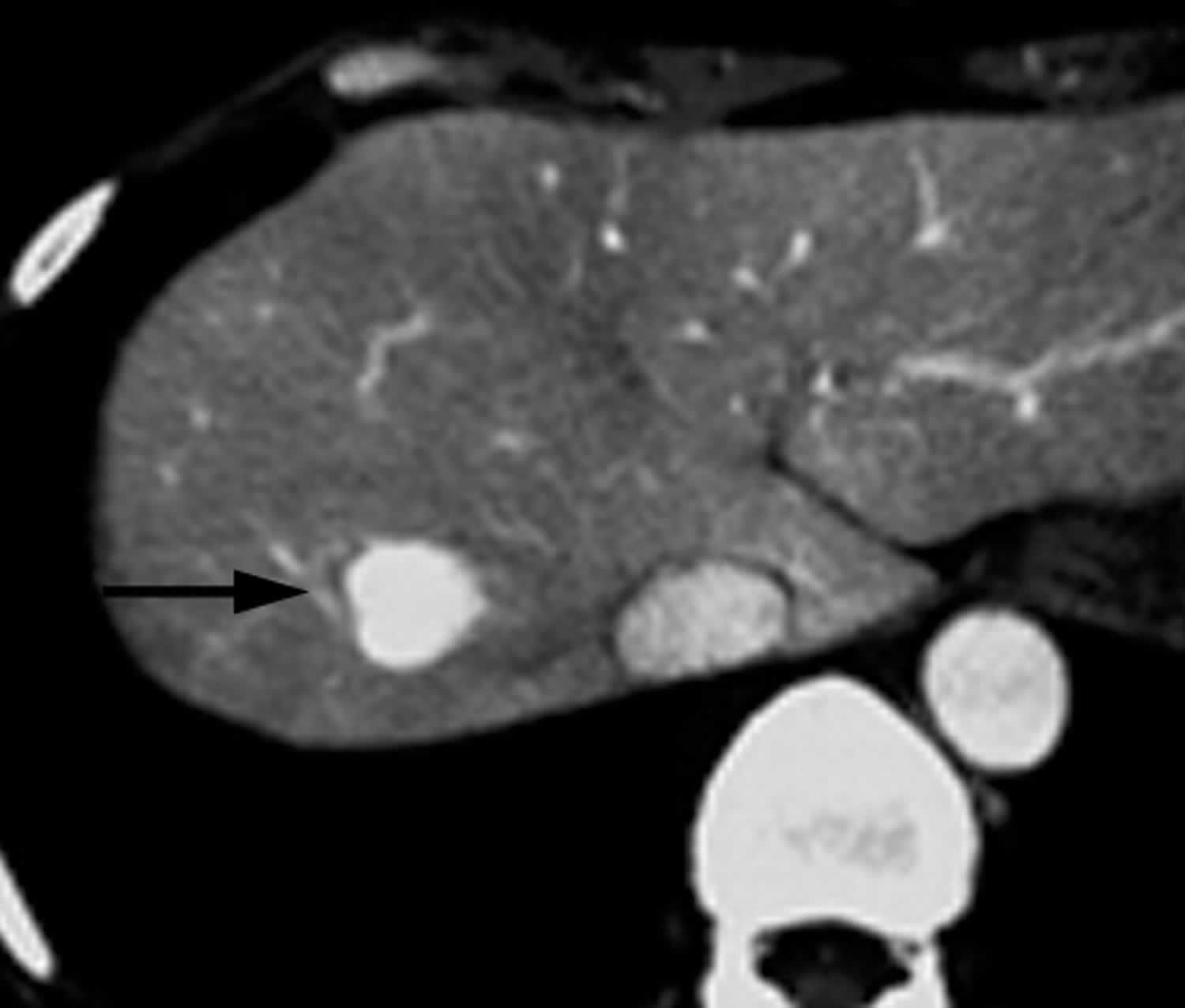
Figure 1. Peliosis hepatis
Footnotes: 51-year-old woman with history of benign ovarian tumor and incidentally discovered hepatic mass. On contrast-enhanced CT during hepatic arterial phase, lesion (arrow) shows marked homogeneous contrast enhancement.
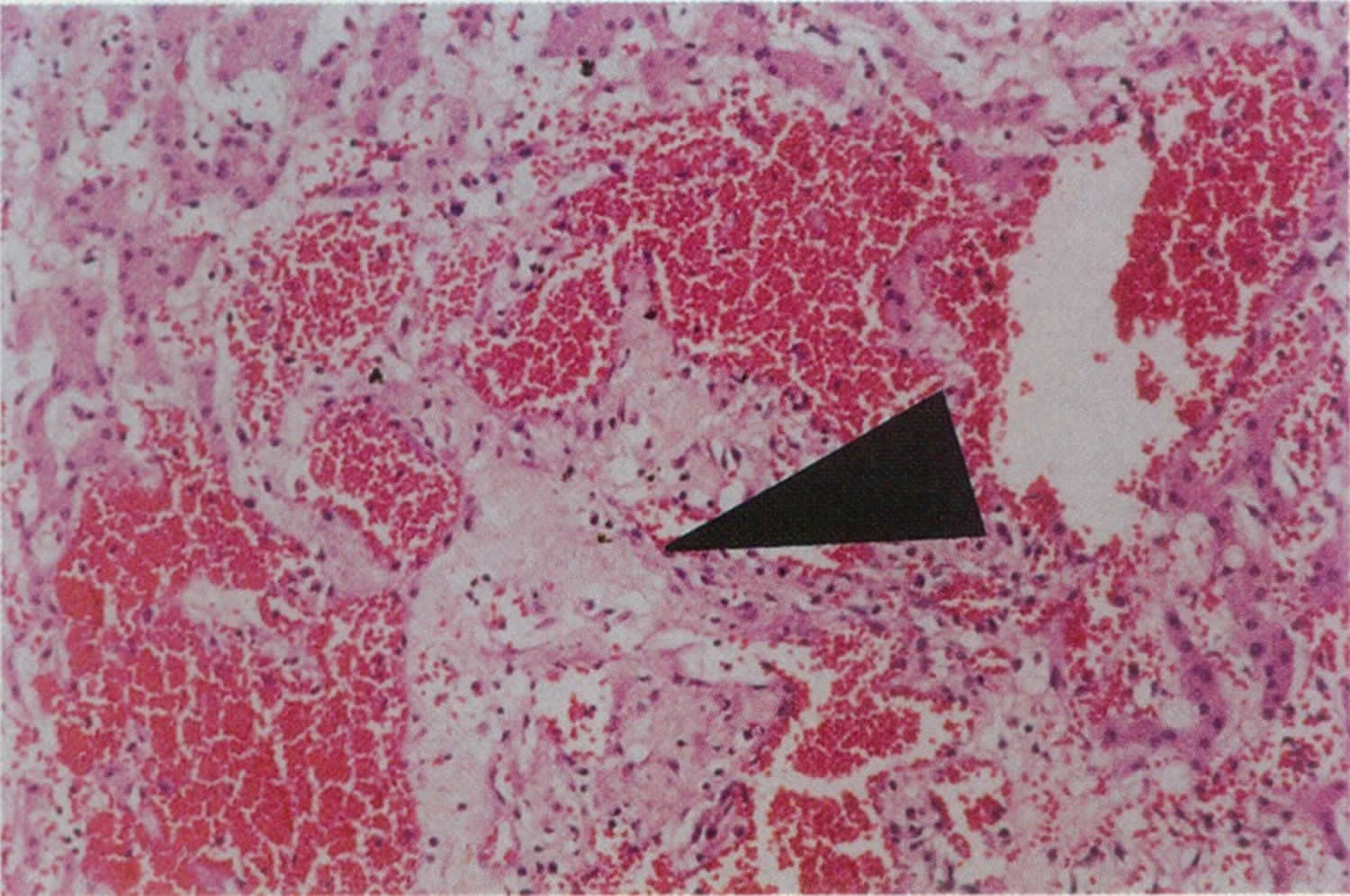
[Source 1 ]Figure 2. Peliosis hepatis liver biopsy specimen
Footnote: Liver biopsy specimen with hematoxylin and eosin staining at 45x magnification showing dilated vascular spaces (peliotic spaces) filled with red blood cells are surrounded by cords of hepatocytes. Note central area of pale myxoid stroma (arrow)
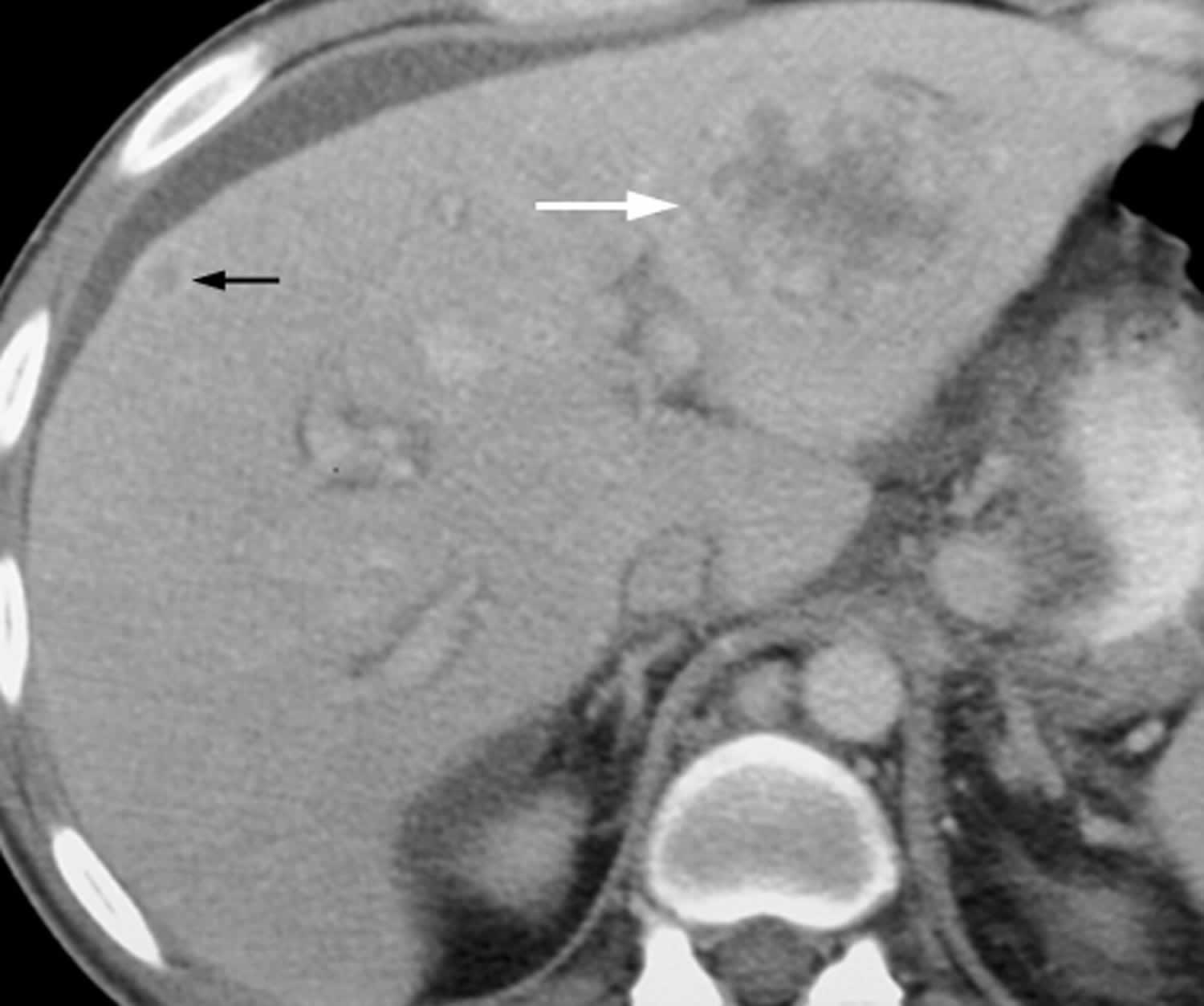
[Source 23 ]Figure 3. Bacillary peliosis
Footnotes: 44-year-old man with AIDS and bacillary peliosis. Transverse contrast-enhanced CT image shows large ill-defined, hypoattenuating lesion (white arrow) with heterogeneous peripheral enhancement within left liver lobe. Smaller subcapsular hypoattenuating lesion (black arrow) with ring enhancement can also be seen in right liver lobe.
[Source 1 ]Peliosis hepatis cause
The cause of peliosis hepatis can be related to drugs including anabolic steroids, oral contraceptives, corticosteroids, tamoxifen, diethylstilbestrol, azathioprine, 6-thioguanine, 6-mercaptopurine, and methotrexate; toxins such as polyvinyl chloride, arsenic, and thorium oxide; chronic wasting diseases (e.g., tuberculosis, leprosy); cancers, particularly hepatocellular carcinoma; liver and kidney transplantation; and Bartonella henselae and Bartonella quintana infection in AIDS so-called bacillary peliosis 3, 17, 18, 19, 1. In addition, several other conditions are described as associated with peliosis hepatis, including sprue (a disorder in which the intestines are unable to absorb nutrients from food), diabetes mellitus, alcoholic liver disease, necrotizing vasculitis, and blood disorders including multiple myeloma, aplastic anemia, Hodgkin disease, and myelofibrosis 24, 25, 18, 1. Moreover, peliosis hepatis may develop after kidney or heart transplantation. In 20 to 50% of patients, no associated condition is identified 1.
Peliosis hepatis causes 26:
- Idiopathic: 20 to 50% of cases
- Toxins
- arsenic
- polyvinyl chloride (PVC)
- thorium oxide
- Drugs
- anabolic steroids
- azathioprine
- corticosteroids
- diethylstilbestrol (DES)
- immunoglobulin therapy
- methotrexate
- oral contraceptives
- tamoxifen
- 6-thioguanine (6-TG)
- 6-mercaptopurine (6-MP)
- Chronic illness
- malignancy, particularly hepatocellular carcinoma
- tuberculosis (TB)
- leprosy
- celiac sprue
- diabetes mellitus
- necrotizing vasculitis
- hematologic disorders: Hodgkin disease, multiple myeloma
- Infection in AIDS
- bacillary peliosis caused by Bartonella henselae, Bartonella quintana and Rochalimaea henselae
- Kidney or heart transplantation
Peliosis hepatis pathophysiology
Peliosis hepatis pathogenesis remains poorly understood, with various investigators proposing that the primary event could be obstruction of hepatic outflow at the sinusoidal level, direct breakdown of sinusoidal borders, dilatation of the central vein of the hepatic lobule, or hepatocellular necrosis leading to cavity formation 11. Lastly, post-sinusoidal obstruction from several conditions such as malignant invasion, autoimmune inflammation, and drug injury is also hypothesized to be an alternate mechanism in the formation of peliosis hepatis 27.
The anatomy of the sinusoidal capillaries is different than other capillary beds within the body. The fenestrations which are present within the liver allow for passes and filtration of larger molecules such as albumin 27. Epithelial damage can result from smoking, which can disrupt the architecture through the formation of free radicals. This damage, in turn, impairs vasodilation by inhibiting nitric oxide formation, which allows for normal physiologic relaxation of the arteries 27. Additionally, inflammation, whether it be autoimmune, direct trauma through surgical instrumentation, medication-induced, or invasion by malignancy, can result in the recruitment of inflammatory cytokines and cells, sometimes resulting in further epithelial damage 27. An alternate mechanism may be the depletion of glutathione, a critical regulatory molecule that assists in many biochemical detoxification reactions, which may disrupt the integrity of the epithelial wall and allow toxic metabolites to build up. However, through signal proteins such as vascular endothelial growth factor and others, re-endothelization can occur; thus, that is why using the absence of epithelial lining on imaging studies is not adequate to diagnose patients with peliosis hepatis, though it can raise the concern for it 28, 29.
Histologically, hepatic peliosis is characterized by multiple mottled blood-filled cyst-like spaces within the liver with associated sinusoidal dilatation 30, 31. These vary in size from <1 mm to several centimeters in diameter.
Macroscopically, the liver appears dark or even purple, and usually, the entire liver is involved to a greater or lesser degree. Focal lesions may demonstrate central areas of hemorrhage.
Peliosis hepatis symptoms
Peliosis hepatis varies from minimal asymptomatic lesions that is diagnosed incidentally on imaging or during an autopsy to massive lesions that may present with cholestasis, hepatic failure, or spontaneous rupture requiring liver transplantation 20, 32. In some instances, enlarged liver (hepatomegaly), ascites (accumulation of fluid in the peritoneal cavity), portal hypertension, cholestasis, and hepatic failure may be present 1. Severe abdominal pain may result from rupture and intraperitoneal hemorrhage 1. In the case of bacillary peliosis, enlarged lymph node (lymphadenopathy) with Bartonella henselae and neurologic symptoms with Bartonella quintana are typical findings.
Peliosis hepatis complications
Peliosis hepatis complications may include:
- hepatic failure
- portal hypertension
- rupture and hemorrhage
There is no current consensus on whether patients with asymptomatic peliosis hepatis warrant routine imaging studies to monitor disease burden or progression. However, some experts believe annual or more frequent evaluation with ultrasound is adequate 33.
Although the incidence of intraperitoneal and intrahepatic hemorrhage do not seem to be common complications, the risk does exist 27. Thus surgical resection appears to assist in the avoidance of such complications, especially if imaging studies have demonstrated worsening of the cavitary lesions 34.
Peliosis hepatis diagnosis
Peliosis hepatis is very difficult to diagnose. The diagnosis of peliosis hepatis is often missed or delayed because it is usually asymptomatic, and its radiologic appearance closely resembles a benign and malignant/pre-malignant lesions or multiple abscesses 20, 6. Radiological imaging shows a broad spectrum of appearances because of its dependence on the blood supply to the lesions. In reality, the imaging findings of peliosis hepatis vary depending on the pathologic patterns of disease, various stages of the blood component of the lesions, and concomitant hepatic steatosis 35.
Laparoscopy is a useful examination to sporadically detect dark-blue or dark-red patchy patterns on the liver surface, which is a characteristic finding of peliosis hepatis 21. Pathological findings are considered the gold standard for the diagnosis of peliosis hepatis, being histologically characterized by blood-filled cavities. However, a case has been reported in which repeated biopsies were required to diagnose peliosis hepatis 22.
Ultrasound findings
Conventional gray-scale sonography appearances are non-specific, usually demonstrating an irregular hypoechoic region/mass 31
Doppler studies can show evidence of both perinodular and intranodular vascularity 30. Recently, the use of a sonographic contrast agent (Levovist [Schering], a galactose and palmitic acid compound) has been shown to provide a “fast surge” central-echo enhancement in peliotic lesions 36.
CT findings
Peliosis lesions appearance on pre-contrast CT (unenhanced CT) is variable, depending on liver density, but is usually of multiple hypoattenuating lesions of variable size 32. Central hemorrhage may lead to areas of hyperattenuation and even dystrophic calcification 30. CT findings vary with the size of lesions, presence or absence of thrombus within the cavities, and presence of hemorrhage. In particular, peliosis lesions may be spontaneously hyperattenuating to liver parenchyma in certain patients (probably related to intralesional hemorrhage). In addition, if peliotic cavities are smaller than 1 cm in diameter, CT findings may be normal 3. Calcifications within peliosis lesions have also been described.
On contrast-enhanced CT, peliosis lesions typically show early globular enhancement and multiple small, central accumulations of contrast material during the arterial phase, with a centrifugal progression of enhancement during the portal venous phase 30. In addition, some lesions can also show areas of increased attenuation. Notably, larger cavities communicating with sinusoids display the same attenuation of blood vessels, whereas thrombosed cavities have the same appearance as nonenhancing nodules 3. More often, during the arterial phase of contrast enhancement, peliosis lesions typically show early globular enhancement (vessel-like enhancement) and multiple small accumulations of contrast material in the center of the lesions the so-called target sign 32. During the portal venous phase, a centrifugal progression of enhancement without a mass effect on hepatic vessels is usually observed 32; however, a centripetal progression of enhancement can also be seen 37. On the delayed phase, late diffuse homogeneous hyperattenuation can also be seen in the phlebectatic type of peliosis hepatis because of the lack of hemorrhagic parenchymal necrosis 32. This accumulation of contrast material in the delayed phase can be useful in the differential diagnosis with other focal hepatic lesions that do not show blood pooling. In some instances, small (< 2 cm) peliotic lesions may also show hyperattenuation on both arterial and portal venous phase images.
MRI findings
On MRI, the signal intensities of peliosis lesions largely depend on the stage and the status of the blood components 30. On T1-weighted images, lesions are hypointense or heterogeneously hypointense if complicated by hemorrhage. On T2-weighted images, they are usually hyperintense compared to the liver parenchyma. Hemorrhagic parenchymal necrosis and thrombosed cavities manifest as non-enhancing areas. The differential diagnosis of peliosis hepatis may differ according to the actual imaging findings. On T2-weighted images, high signal together with early lesion enhancement can mimic hepatocellular carcinoma or hypervascular metastasis 37, while a bright T2-weighted signal and persistent delayed-phase CT or MRI may help distinguish peliosis hepatis.
Angiographic findings
On angiography, peliosis lesions appear as multiple vascular nodules (i.e., accumulations of contrast material) during the late arterial phase. The enhancement of peliotic lesions typically is more distinct during the parenchymal phase and persists during the portal venous phase 38.
Biopsy findings
A percutaneous needle biopsy can be used to obtain a specimen to further elucidate imaging findings and confirm the diagnosis. However, a percutaneous liver biopsy carries a risk of bleeding and hepatic hemangioma formation 8. Some authors claim that, due to the high risk of bleeding, an open biopsy is essential in order to realize the differential diagnosis under intraoperative ultrasound supervision 39, 40, 41, 42, 43, 44.
The gross pathologic appearance of peliosis hepatis is that of multiple, irregularly shaped blood-filled hepatic cavities 1. The lesions typically involve the entire liver, but focal peliosis hepatis has been described. At microscopic examination, cystic dilated sinusoids filled with red blood cells and bound by cords of liver cells can be seen (see Figure 1).
Macroscopic examinations show that peliotic lesions in the cutting section present hemorrhagic cyst cavities of various sizes (from < 1 mm to several centimeters) with features of “Swiss cheese”45, 46. From the histopathologic point of view, the differential diagnosis is used in order to diversify the sinusoids’ dilatation from Budd-Chiari syndrome, which determines a venous congestion of the liver due to a vascular occlusion 45, 6, 47.
Pathologists originally classified peliotic lesions by the presence or absence of endothelium (inner cellular lining of the blood vessels) 11. However, even though the blood-filled cavities do not always have endothelial lining , it has been observed that reendothelialization occurs rapidly 48, 49, 1. Therefore, the continuity or rupture of the endothelial lining of hepatic sinusoids is not a reliable criterion to define peliosis hepatis 3. To distinguish peliosis hepatis from hepatic sinusoidal dilatation, lesions should show evidence of rupture of the reticulin fibers that support the hepatocytes and sinusoids 11. This rupture may follow the intrinsic weakness of the fibers of the endothelial wall (peliosis hepatis of the phlebectatic type) or may be associated with focal hepatocyte necrosis (peliosis hepatis of the parenchymal type) 11.
Yanoff et al 50 described microscopically two different types of peliosis hepatis:
- “parenchymal peliosis,” which consists of irregular cavities that are surrounded neither by the sinusoidal cells nor by fibrous tissue, and
- “peliosis flebectasica” which is characterized by spherical regular cavities coated by endothelium and/or fibrosis 46, 48, 51
Peliotic lesions in bacillary peliosis contain clumps of organisms (i.e., Bartonella henselae and Bartonella quintana) that stain with the Warthin-Starry technique 11. Patients with bacillary peliosis often have peliosis of the spleen and lymph nodes and cutaneous angiomatous lesions 1.
Peliosis hepatis differential diagnosis
- Hepatic adenoma: Similar to peliosis, hepatic adenoma might also be associated with the long-term use of estrogens. In the case of diffuse peliosis hepatis, the differential diagnosis is relatively easy. In addition, the presence of fat in some adenomas is a useful sign to make a differential diagnosis. In certain instances, however, focal peliosis can be difficult to differentiate from adenomas. In these patients, biopsy is often required to reach a definitive diagnosis.
- Cavernous hemangioma: The typical enhancement pattern of hemangiomas (i.e., globular discontinuous contrast enhancement tends to be centripetal (periphery first) rather than centrifugal (center first)) is opposite of peliosis hepatis, and therefore differential diagnosis can be achieved in most patients. In addition, hemangiomas may be rather large lesions with a mass effect on the hepatic vessels, whereas peliotic lesions usually show no mass effect on hepatic vessels.
- Hepatocellular carcinoma (liver cancer): Hepatocellular carcinoma is usually hyperattenuating in the arterial phase with rapid washout in the portal venous phase and iso- or hypoattenuation in the delayed phase. Although rare, the possibility that peliosis hepatis may mimic the presence of hypervascular hepatocellular carcinoma has been reported in the literature. In these patients, biopsy is often necessary to reach a definitive diagnosis.
- Hepatic abscess (liver abscess): The differential diagnosis between peliosis hepatis and hepatic abscess is extremely important to avoid the percutaneous drainage of peliotic lesions, which can be dangerous and even fatal 52. With regard to imaging criteria, a hepatic abscess usually presents as a mass with a multiseptated or cluster-of-grapes appearance with nonenhancing contents.
- Focal nodular hyperplasia (benign tumor that forms in the liver): Focal nodular hyperplasias are typically homogeneously hyperattenuating masses on the arterial phase and isoattenuating on the portal venous and delayed phases. These lesions often have a central scar with low attenuation on the arterial and portal venous phases and enhancement on the delayed phase images. When such typical imaging characteristics of focal nodular hyperplasia are present, the differential diagnosis with peliosis hepatis can be achieved easily. Atypical forms of focal nodular hyperplasia may not show the characteristic enhancement patterns and the central scar just described, however, and thus pose some problems in the differential diagnosis with peliosis hepatis.
- Hepatic adenoma (an uncommon solid, benign liver lesion that develops in an otherwise normal-appearing liver) may contain fat
- Hypervascular metastases: Although some hypervascular metastases with fibrotic change can show mild hyperattenuation in the delayed phase, hypervascular metastases are usually totally hypoattenuating or isoattenuating in the delayed phase of contrast enhancement because of the rapid washout of contrast material. Thus, in general, peliotic lesions are rarely confused with hypervascular metastases.
- Hepatic sinusoidal dilation: usually the enhancement pattern is different on CT/MRI 53
Peliosis hepatis treatment
Peliosis hepatis treatment depends on the cause. When a causative drug or toxin is suspected, withdrawal of that agent may result in resolution. If peliosis hepatis seen in the setting of HIV/AIDS, antibiotic treatment may be effective in eradicating B. henselae. If focal and hemorrhagic, surgical resection may also be beneficial 30.
It is important not to drain peliosis, having mistaken it for a hepatic abscess, as bleeding can be life threatening 52.
Peliosis hepatis prognosis
The progression of peliosis hepatis is still largely uncertain 27. Researchers have not observed any apparent patterns of peliosis hepatis disease progression. The natural course of peliosis hepatis is regression after drug withdrawal, cessation of steroid therapy, or resolution of an associated infectious disease 1. However, peliosis hepatis can become worse asymptomatically and present as an accidental finding that occurs during investigations of other diseases or on the autopsy table 39, 51. A pseudotumoral and hemorrhagic evolution has also been described 51, 54.
Some authors claim that peliosis hepatis could be associated to liver failure with clinical features of hepatomegaly, portal hypertension, cholestasis, liver rupture leading to intraperitoneal hemorrhage (internal bleeding in which blood gathers in your peritoneal cavity) or shock and, more rarely, in cirrhotic patients positive for hepatitis C virus 51, 40, 55 or in cases of rupture of peliosis hepatis lesions with hemoperitoneum and hemodynamic decompensation with lethargy and abdominal pain 39, 51, 46, 40. In general, if untreated, ruptured peliosis hepatis may be rapidly fatal 55.
In other cases, especially when peliosis hepatis occurs at very young age, the disease shows important effects of compression with stenosis of the vena cava 56.
In still other cases, after the interruption of the steroid therapy or the resolution of the subsequent infections, a total regression was noticed 48, 40, 51. Peliosis hepatis regression can also occur without any connection with the past medical history, especially in those cases (20% to 50%) that do not allow association of peliosis hepatis to any kind of etiology 31.
Even though some authors support possible transplantation in acute liver failure cases or to use hepatectomy for the diagnosis and treatment of peliosis hepatis, surgery is more and more often used in urgent cases as treatment for the bleeding that may occur. At the same time, for the same issue, some authors proposed embolization that may be performed by an interventional radiologist or during the operation or the laparoscopic biopsy 57, 58.
Spleen peliosis
Spleen peliosis also called peliosis of the spleen, splenic peliosis or peliosis lienalis is an extremely rare benign blood vessel condition characterized by multiple, randomly distributed, blood-filled and cyst-like cavities throughout the spleen 59, 60. The size of the lesions may vary from 1 millimetrer to several centimeters 1. Peliosis is a term derived from the Greek pelios, which means “dusky” or “purple,” referring to the color of the liver tissue with peliosis 61, 1. Spleen peliosis has also been reported, either alone 62, 63, 64, 65 or with peliosis hepatis (hepatic peliosis or liver peliosis) 66, 67, 68, 69, 70, 71, 72. Spleen peliosis often presents with spontaneous rupture of the spleen and requires urgent surgical intervention 73), 74, 75, 60, 63, 76, 7, 77, 78, 79, 16, 80, 15, 81, 82. Numerous associations with medications and other conditions have been described in the literature, emphasizing the limited understanding of spleen peliosis 2. However, spleen peliosis may also be recognized as an incidental finding on computed tomography (CT) scans or at autopsy 83, 82, 81, 15, 79, 84, 85, 86.
Spleen peliosis may be idiopathic or associated with infections such as tuberculosis and Bartonella, hematological diseases such as myeloma, myelofibrosis, and Hodgkin’s disease and with the use of androgenic steroids and erythropoetin. Patients with isolated splenic peliosis may be asymptomatic or describe abdominal discomfort. It is often detected as an incidental finding on abdominal imaging or when spontaneous rupture occurs.
Spleen peliosis discovered on imaging should immediately prompt further evaluation. A differential diagnosis will often include malignant, infiltrative, or infectious conditions, as well as post-traumatic splenic cysts. The cysts of spleen peliosis are blood-filled and innumerable in quantity, distinguishing them from post-traumatic fluid-filled cysts of the spleen. Isolated peliosis of the spleen is extremely rare, and is therefore infrequently considered 60, 83, 82, 81. Imaging modalities for further assessing the spleen vary given the reason for testing and the differential diagnosis; they often include ultrasound, CT, positron emission tomography with CT (PET-CT), and magnetic resonance imaging 15, 16, 87. Despite these various imaging modalities, imaging may be inadequate for establishing a clear diagnosis 15, 16. While spleen peliosis often exists without symptoms, atraumatic spleen rupture, requiring urgent surgical intervention is a well-established presentation of spleen peliosis 73), 63, 74, 88, 89, 90, 91, 92, 93, 94, 87, 95.
As there are no clear published guidelines on the management of spleen peliosis, the definitive management of splenic rupture due to peliosis is emergency splenectomy 96, 97, 98, 99. However, prophylactic splenectomy is still debatable if accidental radiological findings were established upon regular check-ups or unrelated complaints 100.
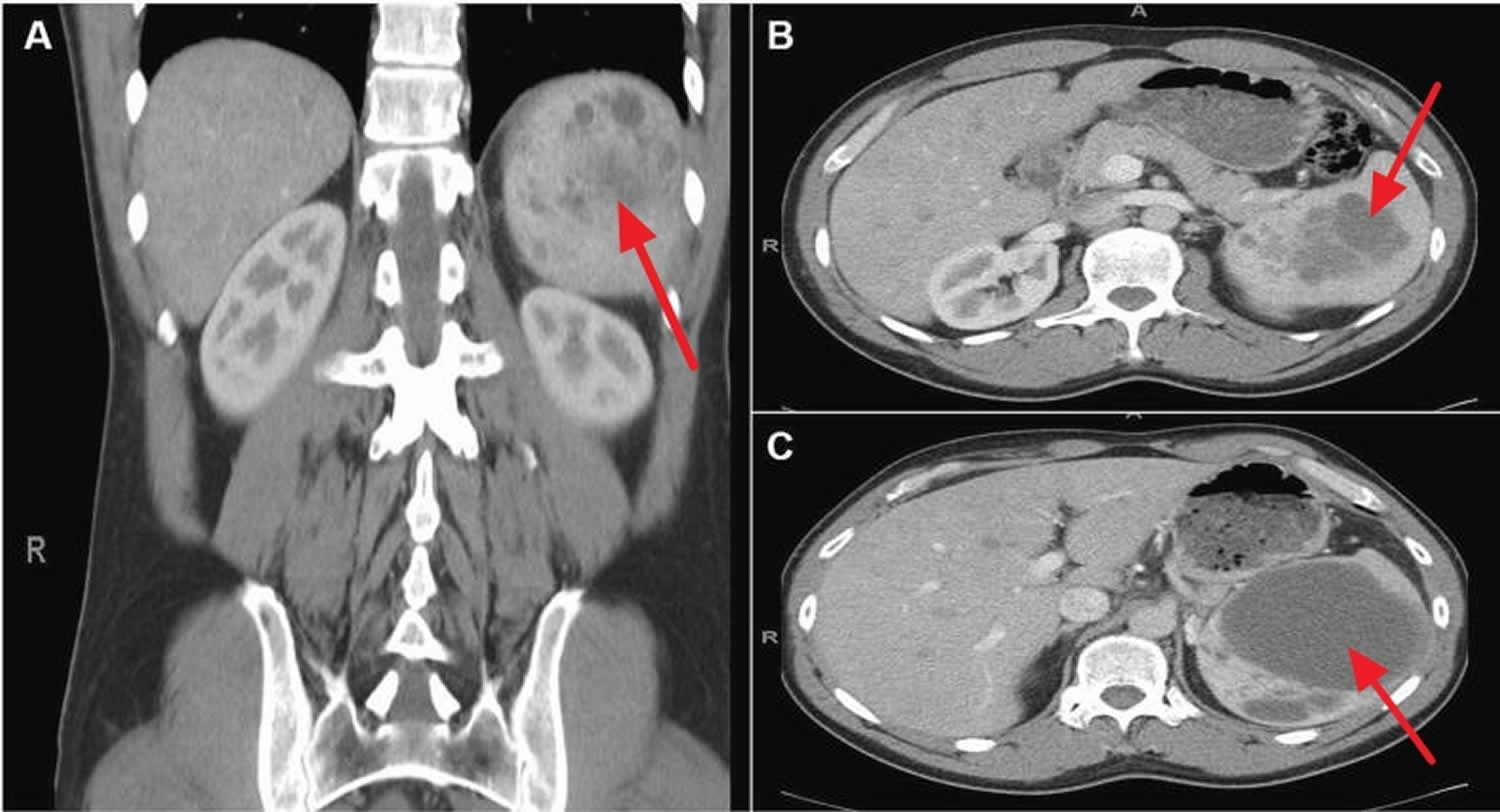
Figure 4. Spleen peliosis
Footnotes: Contrast-enhanced CT scan from a 44-year-old otherwise healthy man presented with left-upper quadrant and left-shoulder pain. The patient denied trauma. A CT scan revealed splenomegaly with multiple splenic cysts and free intraperitoneal fluid consistent with blood. (A) Coronal view, (B) Axial view at the level of L1, (C) Axial view at the level of T12. Significant splenomegaly (14 × 11 × 9 cm). Multiple, loculated, cystic lesions are noted throughout the spleen. The largest cystic lesion measures 9.3 cm in diameter. A minor hemoperitoneum (not visible) was present on the scan.
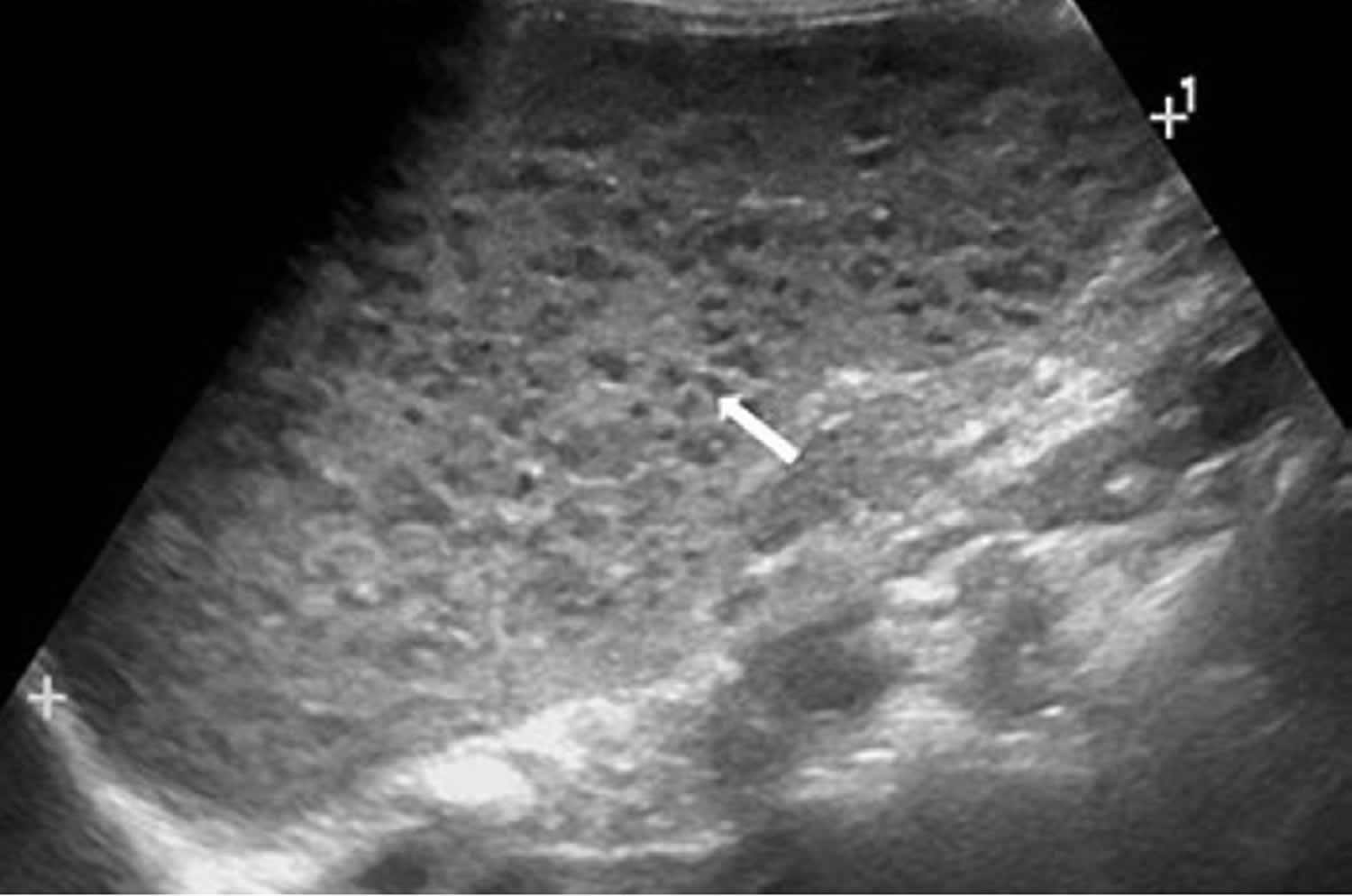
[Source 2 ]Figure 5. Spleen peliosis ultrasound
Footnote: A 3-year-old girl with splenic peliosis. Longitudinal ultrasonography of the spleen shows splenomegaly and multiple poorly defined hypoechoic nodules (arrow).
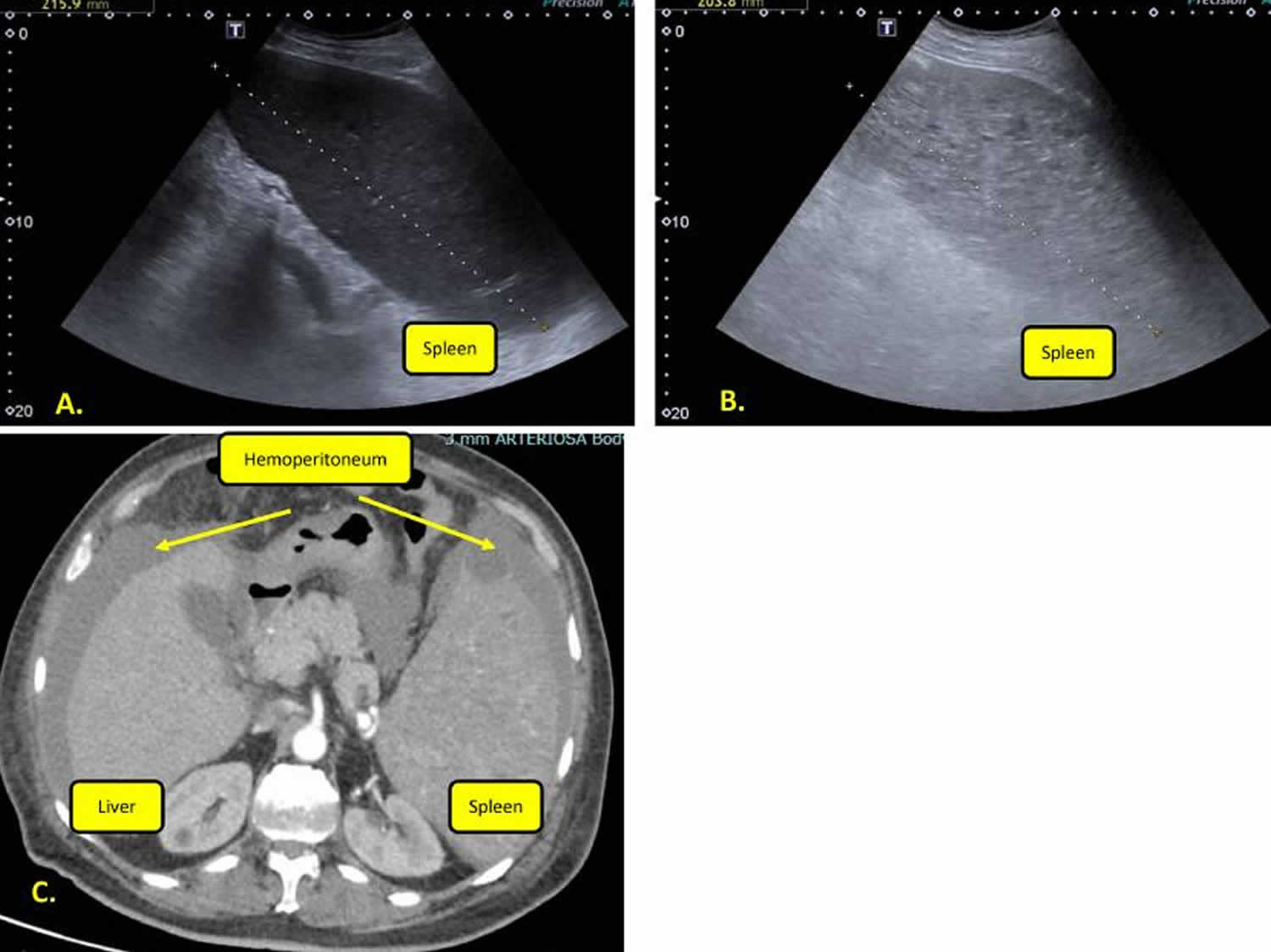
[Source 101 ]Figure 6. Spleen peliosis rupture
Footnotes: (A) Spleen ultrasound appearance at diagnosis. (B) Ultrasound spleen appearance in peliosis rupture. Ultrasonographic imaging showed inhomogeneous splenic parenchyma and increased size. (C) Abdominal computer tomography (CT) frame. CT showed hemoperitoneum and marked splenomegaly (with a pole-to-pole diameter of about 200 mm) with many hyperdense areas.
[Source 102 ]Spleen peliosis cause
The cause of spleen peliosis remains unclear 2. Several causative factors were suggested to be associated with peliosis in the spleen, including the following: intravenous drug abuse, the usage of anabolic steroids, danazol, chronic alcohol use, granulocyte colony-stimulating factor (GSCF) use, tamoxifen, azathioprine, androgens, chemotherapy, liver cirrhosis, tuberculosis, oral contraceptive pills, human immunodeficiency virus (HIV), Hodgkin’s disease, aplastic anemia, and myeloma 98, 99, 103, 81, 15, 80, 16, 79, 78, 77, 7, 104, 105, 106, 89, 90, 91, 92, 93, 94, 87, 107, 108, 109. A number of reported patients were also noted to have blood disorders including multiple myeloma, aplastic anemia, Hodgkin disease, and myelofibrosis 104, 105, 106, 89, 90, 91, 92, 93, 94, 87.
Preliminary evidence links the development of peliosis to angiogenesis (formation of new blood vessels) due to elevated levels of vascular endothelial growth factor (VEGF) 7. VEGF is a fundamental regulator of physiologic angiogenesis. Elevated VEGF levels are known to be associated with tumor progression and poor outcomes in a number of cancers. A condition resembling peliosis has been demonstrated in mice with elevated VEGF levels, that were found to have massively dilated hepatic sinusoids and endothelial cell proliferation and apoptosis 7. The experimental link to elevated VEGF levels, warrants investigation into the treatment of peliosis with anti-angiogenic factors 2.
Spleen peliosis risk factors include:
- use of anabolic steroids
- hematological conditions such as aplastic anemia
- wasting diseases such as
- tuberculosis
- AIDS
- concurrent malignancy
Spleen peliosis symptoms
Patients with spleen peliosis are commonly asymptomatic, and splenic peliosis may present as an incidental finding during imaging studies for other diseases, at autopsy, or with spontaneous rupture of the spleen after minimal or no trauma 2. Although uncommon, some reports suggest that males, ages > 20 years, presence of splenomegaly, and use of chemotherapy might be associated with increased risk of spontaneous splenic rupture in hematology 110. One literature review has shown that all the reported spleen peliosis cases collected from 1980 till 2016 had either presented with abdominal pain or circulatory shock 111. Most cases of spleen peliosis are associated with peliosis hepatis (peliosis of the liver) 7.
Spleen peliosis diagnosis
Spleen peliosis hepatis is very difficult to diagnose because it is usually asymptomatic, and extreme rarity. Peliosis diagnosis is usually obtained after histopathological examination of the resected organ or at autopsy. Radiological imaging shows a broad spectrum of appearances because of its dependence on the blood supply to the lesions. Ultrasound shows multiple poorly defined hypoechoic lesions (Figure 4) and hyperechoic thrombosis 112. The finding of cyst-like hypodense lesions which do not cause mass effect on vessels on contrast-enhanced CT should alert the radiologist to the diagnosis of spleen peliosis. This is of importance in recognizing the risk of spontaneous splenic rupture and in avoiding interventional procedures, such as liver biopsy or percutaneous transhepatic cholangiography, which may precipitate life-threatening iatrogenic organ rupture.
Peliosis histological appearance is characterized by multiple blood-filled cystic cavities primarily involving the mononuclear phagocytic system (liver, spleen, bone marrow, and lymph nodes).
Once the presence of peliosis has been established it is proposed that all necessary investigations be pursued to detect its presence in other organs and to establish a possible cause 113. The implications for this are clear. Spontaneous rupture of the spleen remains an ever-present threat. Physicians should be mindful of this in the event that anticoagulation or thrombolysis is being considered. In addition, the presence of peliosis may prompt the investigation and subsequent discovery of equally serious underlying conditions, for example immunodeficiency or malignancy.
Spleen peliosis treatment
Hemorrhage associated with splenic peliosis is quite amenable to surgical therapy if recognised and treated promptly 114. The existing literature does not however explore the surgical management of patients in whom an incidental diagnosis of splenic peliosis has been made. The introduction of laparoscopic splenectomy has reduced significant morbidity when compared with open surgery 115. Prophylactic laparoscopic splenectomy has been advocated to prevent organ rupture in conditions such as splenic sarcoidosis 116. The role of elective splenectomy in splenic peliosis is not known.
From the patient’s perspective, lifestyle changes may be necessary. Younger patients should be encouraged to avoid high-risk activities, for example, contact sports 113. It would seem wise to avoid or discontinue the oral contraceptive pill, and seek alternative forms of contraception. In addition any attending physician should be aware when other medications thought to cause peliosis are being considered 113.
Bacillary peliosis
Bacillary peliosis also called bacillary peliosis hepatis or Bartonella peliosis hepatis is a blood vessel condition characterized by multiple, randomly distributed, blood-filled and cyst-like cavities throughout the liver that is secondary to Gram-negative Bartonella henselae bacteria infection in immunocompromised patients, first described in human immunodeficiency virus (HIV) infected or AIDS individuals 23, 117, 118, 119. Most patients with bacillary peliosis develop symptoms such as anorexia, weight loss, enlarged tender lymph nodes (lymphadenopathy), chills, fever, headache and malaise in addition to gastrointestinal symptoms or abdominal pain 117, 120, 121, 122, 123, 124. Approximately 1% to 2% of patients with Bartonella infection develop severe systemic disease with involvement of the liver, spleen, bone, central nervous system (brain and spinal cord) or lung 122. Approximately 25% of patients have lymphadenopathy (enlarged lymph nodes), but often the classic skin papule of cat-scratch disease is absent 122, 123, 124.
Bacillary peliosis hepatis patients have enlarged liver (hepatomegaly), and some also have enlarged spleen (splenomegaly). One-quarter of the patients also had skin bacillary angiomatosis (cutaneous bacillary angiomatosis) lesions. Abdominal CT of the peliotic liver usually reveals numerous hypodense lesions, but this appearance is not specific for bacillary peliosis; therfore the diagnosis of Bartonella infection must be confirmed by histopathological evaluation. Additionally, some HIV-infected patients with hepatic Bartonella infection develop inflammatory nodules in the liver without the vascular proliferative characteristics of peliosis hepatis. Patients with splenic bacillary peliosis can have thrombocytopenia or pancytopenia and abdominal ascites.
HIV-associated bacillary peliosis hepatis is an unusual, treatable opportunistic infection that responds to antibiotics. Inadequately treated, HIV-associated bacillary peliosis can be progressive and may be fatal 125, 23.
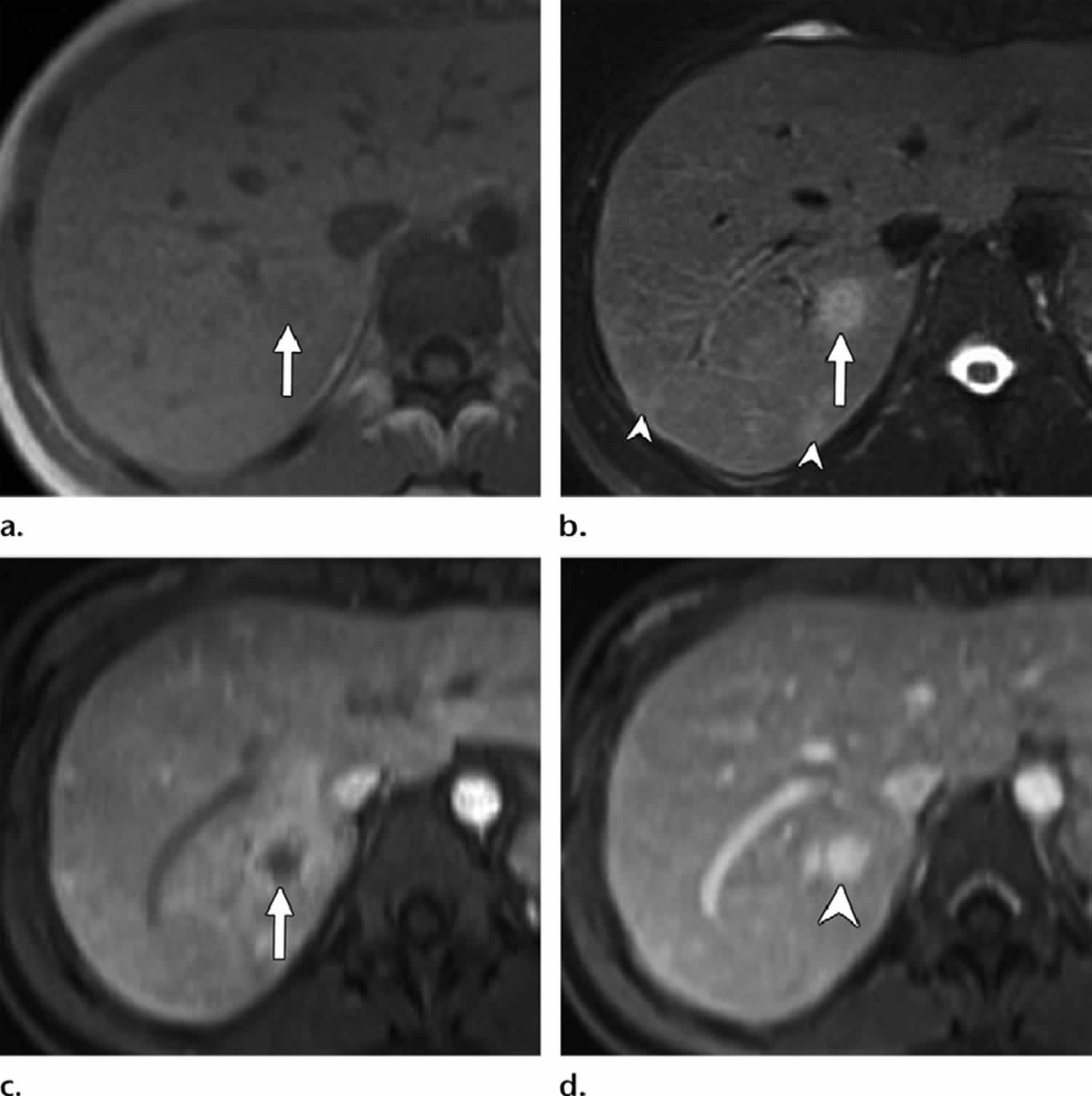
Figure 7. Bacillary peliosis
Footnotes: Bacillary peliosis in a 32-year-old man with HIV/AIDS who presented with fever. (a) Axial in-phase T1-weighted MR image shows an ill-defined iso-to mildly hypointense nodule in the posterior segment of the right hepatic lobe (arrow). (b) Axial fat-saturated T2-weighted MR image shows moderate to high signal intensity of the nodule (arrow). Smaller similar subcapsular nodules are seen posteriorly (arrowheads). (c) Axial contrast-enhanced early phase fatsuppressed T1-weighted MR image shows the hypointense right hepatic nodule with irregular continuous peripheral enhancement (arrow). (d) Axial contrast-enhanced delayed phase T1-weighted MR image shows delayed enhancement within the nodule (arrowhead), which reaches a signal intensity similar to that of the surrounding hepatic venous structures because of contrast agent retention within dilated sinusoids in the lesion.
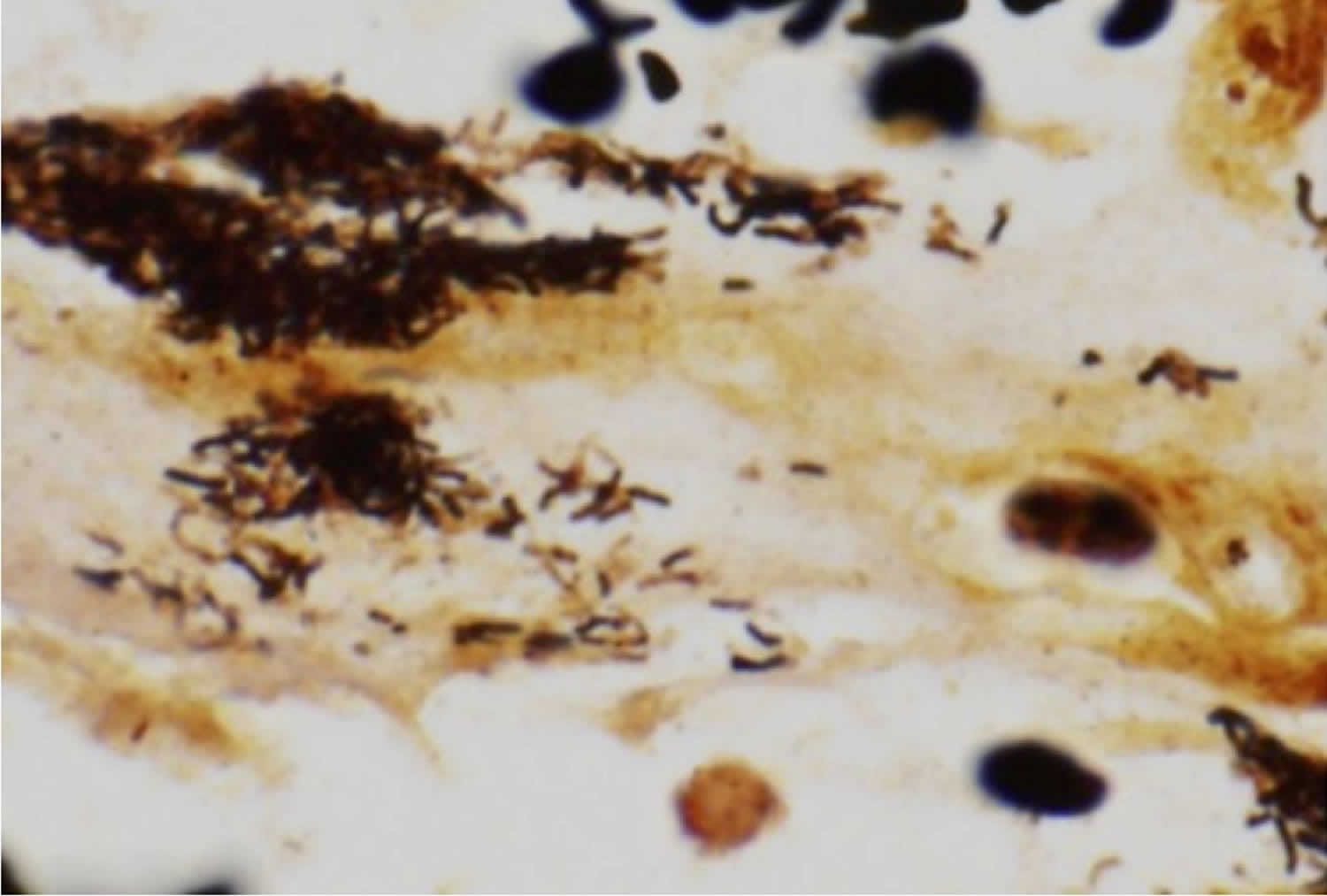
[Source 126 ]Figure 8. Bartonella bacteria
Footnote: Warthin-Starry stain demonstrating the characteristic pleomorphic bacilli of Bartonella bacteria in bone marrow core biopsy
[Source 117 ]Bacillary peliosis cause
Bartonella henselae the bacteria that cause bacillary peliosis commonly cause cat scratch disease or cat scratch fever 124, 123, 127, 128, 129, 130,131, 132. People become infected with Bartonella henselae from the scratch of domestic or feral cats, particularly kittens 129, 133, 134, 124. Cats can have fleas that carry Bartonella henselae bacteria. These bacteria can be transmitted from a cat to a person during a scratch that is contaminated with flea stool. Infected cats that lick a person’s open wound or bite can also spread Bartonella henselae bacteria. Some evidence suggests that these bacteria may spread directly to people by the bite of infected cat fleas, but this has not been proven.
Bartonella henselae infection or cat scratch disease occurs most often in children under the age of 15 (children 5 to 10 years of age) 124, 123, 127. Though more common in the southeast, cat scratch disease occurs throughout the United States. Stray cats are more likely than pets to be infected with B. henselae. In the United States, most cases of cat scratch disease occur in the fall and winter.
Bacillary peliosis symptoms
Most patients with bacillary peliosis develop symptoms such as anorexia, weight loss, enlarged tender lymph nodes, chills, headache, malaise and fever in addition to gastrointestinal symptoms or abdominal pain 117, 125, 120, 121, 122, 123, 124. Approximately 1% to 2% of patients with Bartonella infection develop severe systemic disease with involvement of the liver, spleen, bone, central nervous system (brain and spinal cord) or lung 122. Approximately 25% of patients have lymphadenopathy (enlarged lymph nodes), but often the classic skin papule of cat-scratch disease is absent 122, 123, 124.
. Bacillary peliosis can cause splenic peliosis and peliosis hepatis. Rupture with intraabdominal hemorrhage has previously been reported in both peliosis hepatis 18 and peliosis of the spleen 135, 62 and is another possible mode of presentation, underscoring the need for rapid diagnosis and therapy in these cases.
Bacillary peliosis diagnosis
Bacillary peliosis is very difficult to diagnose. Laboratory findings typically included a mild-to-moderate elevation of the serum aminotransferase level (alanine aminotransferase (ALT) and aspartate aminotransferase (AST)) and a moderate-to-severe elevation of the alkaline phosphatase (ALP) level in association with a normal or only mildly elevated bilirubin level 117. Progressive reduction in the number of blood cells (pancytopenia) occurred in some patients. Imaging studies showed enlarged abdominal lymph glands and a heterogeneous hepatic or splenic parenchyma in some cases 117.
Liver lesions are typically multiple and associated with abdominal lymphadenopathy and, in some cases, splenic lesions; many patients come to biopsy to exclude tumors 122, 127, 136. On laparotomy, the liver may be found to be studded with hard nodules of varying sizes 136.
Biopsy findings
Liver biopsy showed the presence of multiple blood-filled cystic spaces, foci of necrosis, fibromyxoid stroma, and clumps of granular purple material that correspond to organisms on Warthin-Starry stain and electron microscopy 137.
Bartonella infection may cause vascular proliferative lesions in the liver. Bacillary epithelioid angiomatosis is a vasoproliferative tissue reaction to Bartonella henselae or Bartonella quintana that usually occurs in immunocompromised hosts. Hepatic involvement shows sharply demarcated periportal areas in which the normal parenchyma is replaced by vascular tissue with extravasated erythrocytes, delicate spindle cells, neutrophils, and karyorrhexic debris, mimicking Kaposi sarcoma 138. Similar lesions have been designated as bacillary peliosis hepatis; these are characterized by the presence of multiple blood-filled cystic spaces, foci of necrosis, fibromyxoid stroma, and clumps of granular purple material that correspond to organisms on Warthin-Starry stain and electron microscopy 138. Bacillary peliosis hepatis can be mistaken for nonbacillary peliosis hepatis.
The hallmark lesion in hepatic cat-scratch disease is an irregular, stellate microabscess surrounded by a layer of palisading histiocytes, lymphocytes, and a rim of fibrous tissue 122, 136, 139. Younger lesions may show more necrosis with less organization of the inflammatory granulomatous response, whereas older lesions may show confluent granulomas with scarring and scant residual necrosis 122. These hepatic lesions are similar to the ones seen in lymph nodes in patients with cat-scratch disease, but they have also been noted in infections with Y. enterocolitica, F. tularensis, lymphogranuloma venereum, mycobacterial species, Candida, and Actinomyces 122. Other lesions may appear as small, rounded granulomas with giant cells and small foci of central necrosis, similar to caseating granulomas in mycobacterial or fungal infections. The background liver parenchyma shows sinusoidal dilatation, portal mixed but predominantly lymphocytic inflammatory infiltrates, portal fibrosis, periductal concentric fibrosis, and focal bile ductular proliferation 122. These changes are attributed to mass effect and are also seen in other infections that result in space-occupying lesions, such as pyogenic abscesses 122.
Warthin-Starry stains identify Bartonella bacteria in some cases (see Figure 5) and the organisms often cluster around vessels or along collagen fibers; the stain is not specific for the organism 123. Culture is difficult. The diagnosis can be confirmed by polymerase chain reaction (PCR) and Southern blot for Bartonella DNA on tissue, skin testing, or serology 124, 122, 139, 140.
Bacillary peliosis treatment
Bacillary peliosis is treated with antibiotic. Azithromyin, doxycycline, and gentamicin are all considered effective against Bartonella but the regimen of azithromycin and/or doxycycline is most commonly used in solid organ recipients 117. Macrolides (e.g., clarithromycin, azithromycin, erythromycin, clarithromycin, roxithromycin) also have immunomodulatory effects, which include reducing vascular endothelial growth factor (VEGF) activity, which may have additional benefits for Bartonella vascular lesions 141. The duration of therapy is not well-defined though given the high risk for morbidity in the immunosuppressed population and the propensity for relapse of Bartonella infections, a prolonged duration of antimicrobial therapy is typically favored 142.
Antibiotic treatment of bacillary angiomatosis and bacillary peliosis hepatis has never been studied systematically. Two criteria must be met to achieve successful eradication of Bartonella infections in the immunocompromised patient: first, the specific strain of Bartonella henselae and Bartonella quintana infecting the patient must have excellent in vivo susceptibility to the prescribed antibiotic, and second, the treatment must be of sufficient duration to prevent relapse 142. The first patient with bacillary angiomatosis to be described was treated empirically with erythromycin, and the lesions resolved completely143. Subsequently, erythromycin has become the drug of first choice and has successfully been used to treat many patients with bacillary angiomatosis 144, 145. Treatment of bacillary angiomatosis and bacillary peliosis hepatis with oral doxycycline (100 mg twice daily) has also been consistently successful 144. Lesions resolved in several patients treated with ceftriaxone or fluoroquinolone compounds 146, 147, but the progression of bacillary angiomatosis lesions in patients has been observed during treatment with ciprofloxacin 148. Additionally, a Bartonella species has been isolated from the blood or bacillary angiomatosis lesions of patients being treated with narrow-spectrum cephalosporins 149, nafcillin, gentamicin, and trimethoprim-sulfamethoxazole (but never from patients being treated with a macrolide, rifamycin, or a tetracycline) 150. Experts therefore do not recommend fluoroquinolones, trimethoprim-sulfamethoxazole, or narrow-spectrum cephalosporins for the treatment of bacillary angiomatosis or bacillary peliosis hepatis 144. Treatment failures have been reported with many different antibiotics, and these are usually attributable to a lack of susceptibility of Bartonella in vivo and/or an insufficient duration of therapy 144, 151.
The drug of choice for the treatment of bacillary angiomatosis is erythromycin given p.o. (500 mg p.o. four times daily) for 3 months, but intravenous (IV) administration should be used in patients with severe disease 144. Patients intolerant of erythromycin can be treated with doxycycline (100 mg p.o. or i.v. twice daily) 144, 152, 153. The response to treatment appears to be equivalent whether erythromycin or doxycycline is prescribed 154. Combination therapy, with the addition of rifampin (300 mg p.o. twice daily) to either erythromycin or doxycycline, is recommended for immunocompromised patients with acute, life-threatening Bartonella infection. The intravenous route is especially important in cases of gastrointestinal intolerance or poor absorption. The combination of doxycycline and rifampin is preferred for the treatment of patients with CNS Bartonella infection because of the superior CNS penetration of doxycycline compared with those of the other first-line antibiotics.
The response to treatment can be dramatic in immunocompromised patients. In one patient who received a single 250-mg oral dose of erythromycin, blood cultures became sterile and a palpable subcutaneous lesion disappeared within hours (but recurred months later). More chronic lesions resolve more slowly, but after approximately 4 to 7 days of therapy, cutaneous bacillary angiomatosis lesions usually improve and resolve completely after 1 month of treatment 155. Bacillary bacillary peliosis hepatis responds more slowly than cutaneous bacillary angiomatosis, but hepatic lesions should improve after several months of treatment.
Relapses of bacillary peliosis hepatis and bacillary angiomatosis lesions in bone and skin have been reported frequently 156, 149, 157, 158. Relapses occur when antibiotics are given for a shorter duration (<3 months), especially in severely immunocompromised HIV-infected patients 144, 147. For this reason and from extensive experience treating patients with bacillary angiomatosis and bacillary peliosis hepatis, experts recommend that treatment be given for at least 3 months for bacillary angiomatosis and 4 months for bacillary peliosis hepatis 154, 159. All immunocompromised patients with a Bartonella infection should receive antibiotic therapy (erythromycin 500 mg p.o. four times daily or doxycycline 100 mg p.o. twice daily); patients who have relapses after the recommended treatment should then receive secondary prophylactic antibiotic treatment with erythromycin (500 mg p.o. four times daily) or doxycycline (100 mg p.o. twice daily) as long as they are immunocompromised 159. Of note, AIDS patients receiving prophylaxis with a macrolide or rifamycin antibiotic for Mycobacterium avium complex infection appear to be protected from developing infections with Bartonella species 150. Some immunocompromised patients develop a potentially life-threatening Jarisch-Herxheimer-like reaction within hours after institution of antibiotic therapy 149. Physicians should advise patients of this possible treatment complication, and patients with severe respiratory and/or cardiovascular compromise should be monitored carefully following institution of antimicrobial therapy 154.
- Iannaccone R, Federle MP, Brancatelli G, Matsui O, Fishman EK, Narra VR, Grazioli L, McCarthy SM, Piacentini F, Maruzzelli L, Passariello R, Vilgrain V. Peliosis hepatis: spectrum of imaging findings. AJR Am J Roentgenol. 2006 Jul;187(1):W43-52. https://doi.org/10.2214/AJR.05.0167[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Chandra A, Navarro SM, Killeen TF, Nemanich G, Harmon JV. Spontaneous splenic rupture due to Peliosis and the association with malignancy: A case series and literature review. Int J Surg Case Rep. 2023 Sep;110:108676. doi: 10.1016/j.ijscr.2023.108676[↩][↩][↩][↩][↩][↩][↩][↩]
- Radin DR, Kanel GC. Peliosis hepatis in a patient with human immunodeficiency virus infection. AJR Am J Roentgenol. 1991 Jan;156(1):91-2. doi: 10.2214/ajr.156.1.1845436[↩][↩][↩][↩][↩][↩]
- Zak FG. Peliosis hepatis. Am J Pathol. 1950 Jan;26(1):1-15, incl 2 pl. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1942842/pdf/amjpathol00591-0007.pdf[↩][↩]
- Gushiken FC. Peliosis hepatis after treatment with 2-chloro-3′-deoxyadenosine. South Med J. 2000 Jun;93(6):625-6.[↩]
- Tsokos M, Erbersdobler A. Pathology of peliosis. Forensic Sci Int. 2005 Apr 20;149(1):25-33. doi: 10.1016/j.forsciint.2004.05.010[↩][↩][↩][↩]
- Joseph F, Younis N, Haydon G, Adams DH, Wynne S, Gillet MB, Maurice YM, Lipton ME, Berstock D, Jones IR. Peliosis of the spleen with massive recurrent haemorrhagic ascites, despite splenectomy, and associated with elevated levels of vascular endothelial growth factor. Eur J Gastroenterol Hepatol. 2004 Nov;16(12):1401-6. doi: 10.1097/00042737-200412000-00027[↩][↩][↩][↩][↩][↩][↩]
- Crocetti D, Palmieri A, Pedullà G, Pasta V, D’Orazi V, Grazi GL. Peliosis hepatis: Personal experience and literature review. World J Gastroenterol. 2015 Dec 14;21(46):13188-94. doi: 10.3748/wjg.v21.i46.13188[↩][↩]
- Dong Y, Wang WP, Lim A, Lee WJ, Clevert DA, Höpfner M, Tannapfel A, Dietrich CF. Ultrasound findings in peliosis hepatis. Ultrasonography. 2021 Oct;40(4):546-554. doi: 10.14366/usg.20162[↩]
- Iwata T, Adachi K, Takahashi M. Peliosis Hepatis Mimicking Malignant Hypervascular Tumors. J Gastrointest Surg. 2017 Jun;21(6):1095-1098. doi: 10.1007/s11605-016-3298-5[↩][↩]
- Wanless IR. Vascular disorders. In: MacSween RNM, Burt AD, Portmann BC, Ishak KG, Scheuer PJ, Anthony PP, eds. Pathology of the liver, 4th ed. Glasgow, UK: Churchill Living stone,2002 : 553-555[↩][↩][↩][↩][↩][↩]
- Yanoff M, Rawson AJ. Peliosis hepatis. an anatomic study with demonstration of two varieties. Arch Pathol 1964;77:159-165.[↩]
- Torabi M, Hosseinzadeh K, Federle MP. CT of nonneoplastic hepatic vascular and perfusion disorders. Radiographics. 2008 Nov-Dec;28(7):1967-82. doi: 10.1148/rg.287085067[↩]
- Levin D, Hod N, Anconina R, Ezroh Kazap D, Shaco-Levy R, Lantsberg S. Peliosis Hepatis Simulating Metastatic Liver Disease on FDG PET/CT. Clin Nucl Med. 2018 Jul;43(7):e234-e236. doi: 10.1097/RLU.0000000000002114[↩]
- Maves CK, Caron KH, Bisset GS 3rd, Agarwal R. Splenic and hepatic peliosis: MR findings. AJR Am J Roentgenol. 1992 Jan;158(1):75-6. doi: 10.2214/ajr.158.1.1727362[↩][↩][↩][↩][↩][↩]
- Tsuda K, Nakamura H, Murakami T, Kozuka T, Yoshii M, Isozaki K, Tsukahara Y, Takami M, Hanada M. Peliosis of the spleen with intraperitoneal hemorrhage. Abdom Imaging. 1993;18(3):283-5. doi: 10.1007/BF00198124[↩][↩][↩][↩][↩]
- Ahsan N, Holman MJ, Riley TR, Abendroth CS, Langhoff EG, Yang HC. Peloisis hepatis due to Bartonella henselae in transplantation: a hemato-hepato-renal syndrome. Transplantation. 1998 Apr 15;65(7):1000-3. doi: 10.1097/00007890-199804150-00024[↩][↩]
- Bagheri SA, Boyer JL. Peliosis hepatis associated with androgenic-anabolic steroid therapy. A severe form of hepatic injury. Ann Intern Med. 1974 Nov;81(5):610-8. doi: 10.7326/0003-4819-81-5-610[↩][↩][↩][↩]
- van Erpecum KJ, Janssens AR, Kreuning J, Ruiter DJ, Kroon HM, Grond AJ. Generalized peliosis hepatis and cirrhosis after long-term use of oral contraceptives. Am J Gastroenterol. 1988 May;83(5):572-5.[↩][↩]
- Clin Mol Hepatol. 2015;21 (4): 387-392. Publication Date (Web): 2015 December 24 (Case Report) https://doi.org/10.3350/cmh.2015.21.4.387[↩][↩][↩]
- Dai YN, Ren ZZ, Song WY, Huang HJ, Yang DH, Wang MS, Huang YC, Chen MJ, Zhang JJ, Tong YX, Pan HY. Peliosis hepatis: 2 case reports of a rare liver disorder and its differential diagnosis. Medicine (Baltimore). 2017 Mar;96(13):e6471. doi: 10.1097/MD.0000000000006471[↩][↩]
- Grønlykke L, Tarp B, Dutoit SH, Wilkens R. Peliosis hepatis: a complicating finding in a case of biliary colic. BMJ Case Rep. 2013 Sep 26;2013:bcr2013200539. doi: 10.1136/bcr-2013-200539[↩][↩]
- Perkocha LA, Geaghan SM, Yen TS, Nishimura SL, Chan SP, Garcia-Kennedy R, Honda G, Stoloff AC, Klein HZ, Goldman RL, et al. Clinical and pathological features of bacillary peliosis hepatis in association with human immunodeficiency virus infection. N Engl J Med. 1990 Dec 6;323(23):1581-6. doi: 10.1056/NEJM199012063232302[↩][↩][↩]
- Elsing C, Placke J, Herrmann T. Alcohol binging causes peliosis hepatis during azathioprine therapy in Crohn’s disease. World J Gastroenterol. 2007 Sep 14;13(34):4646-8. doi: 10.3748/wjg.v13.i34.4646[↩]
- Sanz-Canalejas L, Gómez-Mampaso E, Cantón-Moreno R, Varona-Crespo C, Fortún J, Dronda F. Peliosis hepatis due to disseminated tuberculosis in a patient with AIDS. Infection. 2014 Feb;42(1):185-9. doi: 10.1007/s15010-013-0490-3[↩]
- Hepatic peliosis. https://radiopaedia.org/articles/hepatic-peliosis?lang=us[↩]
- Siddiqi I, Gupta N. Peliosis Hepatis. [Updated 2023 Jun 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK554470[↩][↩][↩][↩][↩][↩]
- Kaplowitz N. The importance and regulation of hepatic glutathione. Yale J Biol Med. 1981 Nov-Dec;54(6):497-502. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2596047/pdf/yjbm00117-0089.pdf[↩]
- Bedair TM, ElNaggar MA, Joung YK, Han DK. Recent advances to accelerate re-endothelialization for vascular stents. J Tissue Eng. 2017 Sep 28;8:2041731417731546. doi: 10.1177/2041731417731546[↩]
- Iannaccone R, Federle MP, Brancatelli G, Matsui O, Fishman EK, Narra VR, Grazioli L, McCarthy SM, Piacentini F, Maruzzelli L, Passariello R, Vilgrain V. Peliosis hepatis: spectrum of imaging findings. AJR Am J Roentgenol. 2006 Jul;187(1):W43-52. doi: 10.2214/AJR.05.0167[↩][↩][↩][↩][↩][↩]
- Savastano S, San Bortolo O, Velo E, Rettore C, Altavilla G. Pseudotumoral appearance of peliosis hepatis. AJR Am J Roentgenol. 2005 Aug;185(2):558-9. doi: 10.2214/ajr.185.2.01850558[↩][↩][↩]
- Gouya H, Vignaux O, Legmann P, de Pigneux G, Bonnin A. Peliosis hepatis: triphasic helical CT and dynamic MRI findings. Abdom Imaging. 2001 Sep-Oct;26(5):507-9. doi: 10.1007/s00261-001-0023-x[↩][↩][↩][↩][↩]
- Orandi M, Pirozynski WJ. Peliosis hepatis. Can Med Assoc J. 1967 Apr 29;96(17):1219-20. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1922866/pdf/canmedaj01213-0036.pdf[↩]
- Charatcharoenwitthaya P, Tanwandee T. Education and Imaging: hepatobiliary and pancreatic: spontaneous intrahepatic hemorrhage from peliosis hepatis-an uncommon complication of a rare liver disorder. J Gastroenterol Hepatol. 2014 Oct;29(10):1754. doi: 10.1111/jgh.12636[↩]
- Kim SH, Lee JM, Kim WH, Han JK, Lee JY, Choi BI. Focal peliosis hepatis as a mimicker of hepatic tumors: radiological-pathological correlation. J Comput Assist Tomogr. 2007 Jan-Feb;31(1):79-85. doi: 10.1097/01.rct.0000232919.22287.20[↩]
- Kleinig P, Davies RP, Maddern G, Kew J. Peliosis hepatis: central “fast surge” ultrasound enhancement and multislice CT appearances. Clin Radiol. 2003 Dec;58(12):995-8. doi: 10.1016/s0009-9260(03)00141-7[↩]
- Steinke K, Terraciano L, Wiesner W. Unusual cross-sectional imaging findings in hepatic peliosis. Eur Radiol. 2003 Aug;13(8):1916-9. doi: 10.1007/s00330-002-1675-9[↩][↩]
- Tsukamoto Y, Nakata H, Kimoto T, Noda T, Kuroda Y, Haratake J. CT and angiography of peliosis hepatis. AJR Am J Roentgenol. 1984 Mar;142(3):539-40. doi: 10.2214/ajr.142.3.539[↩]
- Choi SK, Jin JS, Cho SG, Choi SJ, Kim CS, Choe YM, Lee KY. Spontaneous liver rupture in a patient with peliosis hepatis: a case report. World J Gastroenterol. 2009 Nov 21;15(43):5493-7. doi: 10.3748/wjg.15.5493[↩][↩][↩]
- Samyn M, Hadzic N, Davenport M, Verma A, Karani J, Portmann B, Mieli-Vergani G. Peliosis hepatis in childhood: case report and review of the literature. J Pediatr Gastroenterol Nutr. 2004 Oct;39(4):431-4. doi: 10.1097/00005176-200410000-00024[↩][↩][↩][↩]
- Kleger A, Bommer M, Kunze M, Klaus J, Leithaeuser F, Wegener M, Adler G, Dikopoulos N. First reported case of disease: peliosis hepatis as cardinal symptom of Hodgkin’s lymphoma. Oncologist. 2009 Nov;14(11):1088-94. doi: 10.1634/theoncologist.2009-0215[↩]
- Tallón García M, Cobelas Cobelas MC, Fernández Sanmartín M, Bao Corral A, Granja Martínez MC. Peliosis hepática secundaria a tratamiento hormonal [Peliosis hepatitis secondary to hormone treatment]. An Pediatr (Barc). 2011 Oct;75(4):286-8. Spanish. doi: 10.1016/j.anpedi.2011.05.012[↩]
- Dai W, Zhong D. Peliosis hepatis mimicking cancer: A case report. Oncol Lett. 2013 Oct;6(4):960-962. doi: 10.3892/ol.2013.1479[↩]
- Battal B, Kocaoglu M, Atay AA, Bulakbasi N. Multifocal peliosis hepatis: MR and diffusion-weighted MR-imaging findings of an atypical case. Ups J Med Sci. 2010 May;115(2):153-6. doi: 10.1080/03009730903262118[↩]
- Sommacale D, Palladino E, Tamby EL, Diebold MD, Kianmanesh AR. Spontaneous hepatic rupture in a patient with peliosis hepatis: A report of one case. Int J Surg Case Rep. 2013;4(5):508-10. doi: 10.1016/j.ijscr.2013.01.030[↩][↩]
- Caremani M, Tacconi D, Lapini L. Acute nontraumatic liver lesions. J Ultrasound. 2013 Nov 26;16(4):179-86. doi: 10.1007/s40477-013-0049-2[↩][↩][↩]
- Picardi N, Pasta V, Monti M. Possibilità di compenso emodinamico del sistema del Retzius e delle vene paravertebrali nella sindrome di Budd-Chiari [Possibility of hemodynamic compensation of the Retzius system and of the paravertebral veins in Budd-Chiari syndrome]. Ann Ital Chir. 1975-1976;49(1-6):163-77. Italian.[↩]
- Wang SY, Ruggles S, Vade A, Newman BM, Borge MA. Hepatic rupture caused by peliosis hepatis. J Pediatr Surg. 2001 Sep;36(9):1456-9. doi: 10.1053/jpsu.2001.26397[↩][↩][↩]
- Buelow B, Otjen J, Sabath AP, Harruff RC. Peliosis hepatis presenting as liver rupture in a vulnerable adult: a case report. Am J Forensic Med Pathol. 2012 Dec;33(4):307-10. doi: 10.1097/PAF.0b013e31823a8b38[↩]
- Yanoff M, Rawson AJ. Peliosis hepatis. An anatomic study with demonstration of two varieties. Arch Pathol. 1964;77:159–165.[↩]
- Ferrozzi F, Tognini G, Zuccoli G, Cademartiri F, Pavone P. Peliosis hepatis with pseudotumoral and hemorrhagic evolution: CT and MR findings. Abdom Imaging. 2001 Mar-Apr;26(2):197-9. doi: 10.1007/s002610000131[↩][↩][↩][↩][↩][↩]
- Cohen GS, Ball DS, Boyd-Kranis R, Gembala RB, Wurzel J. Peliosis hepatis mimicking hepatic abscess: fatal outcome following percutaneous drainage. J Vasc Interv Radiol. 1994 Jul-Aug;5(4):643-5. doi: 10.1016/s1051-0443(94)71572-4[↩][↩]
- Yang DM, Jung DH, Park CH, Kim JE, Choi SJ. Imaging findings of hepatic sinusoidal dilatation. AJR Am J Roentgenol. 2004 Oct;183(4):1075-7. doi: 10.2214/ajr.183.4.1831075[↩]
- Verswijvel G, Janssens F, Colla P, Mampaey S, Verhelst H, Van Eycken P, Erven W. Peliosis hepatis presenting as a multifocal hepatic pseudotumor: MR findings in two cases. Eur Radiol. 2003 Dec;13 Suppl 4:L40-4. doi: 10.1007/s00330-003-1852-5[↩]
- Hyodo M, Mogensen AM, Larsen PN, Wettergren A, Rasmussen A, Kirkegaard P, Yasuda Y, Nagai H. Idiopathic extensive peliosis hepatis treated with liver transplantation. J Hepatobiliary Pancreat Surg. 2004;11(5):371-4. doi: 10.1007/s00534-004-0908-5[↩][↩]
- Hiorns MP, Rossi UG, Roebuck DJ. Peliosis hepatis causing inferior vena cava compression in a 3-year-old child. Pediatr Radiol. 2005 Feb;35(2):209-11. doi: 10.1007/s00247-004-1311-8[↩]
- Suzuki S, Suzuki H, Mochida Y, Hirai H, Yoshida T, Ide M, Tani M, Shimura T, Morinaga N, Ishizaki M, Kuwano H. Liver hemorrhage due to idiopathic peliosis hepatis successfully treated with hepatic artery embolization. Int Surg. 2011 Oct-Dec;96(4):310-5. doi: 10.9738/cc43.1[↩]
- Omori H, Asahi H, Irinoda T, Takahashi M, Kato K, Saito K. Peliosis hepatis during postpartum period: successful embolization of hepatic artery. J Gastroenterol. 2004;39(2):168-71. doi: 10.1007/s00535-003-1268-7[↩]
- Yasmin, A., McKay, P. & Jackson, B. Splenic peliosis: an unusual cause of splenic lesions. Int J Hematol 87, 107 (2008). https://doi.org/10.1007/s12185-008-0024-4[↩]
- Lashbrook DJ, James RW, Phillips AJ, Holbrook AG, Agombar AC. Splenic peliosis with spontaneous splenic rupture: report of two cases. BMC Surg. 2006 Jun 26;6:9. doi: 10.1186/1471-2482-6-9[↩][↩][↩]
- Wagner E. Ein fall von Blutcysten in der Leber. Arc Heilkunde. 1861;2:369–370.[↩]
- Garcia RL, Khan MK, Berlin RB. Peliosis of the spleen with rupture. Hum Pathol. 1982 Feb;13(2):177-9. doi: 10.1016/s0046-8177(82)80123-8[↩][↩]
- Warfel KA, Ellis GH. Peliosis of the spleen. Report of a case and review of the literature. Arch Pathol Lab Med. 1982 Feb;106(2):99-100.[↩][↩][↩]
- Lacson A, Berman LD, Neiman RS. Peliosis of the spleen. Am J Clin Pathol. 1979 May;71(5):586-90. doi: 10.1093/ajcp/71.5.586[↩]
- Tada T, Wakabayashi T, Kishimoto H. . Peliosis of the spleen . Am J Clin Pathol 1983; 79:708–13.[↩]
- Young JM. Peliosis hepatis; report of two cases. Am Rev Tuberc. 1953 Mar;67(3):385-90. doi: 10.1164/art.1953.67.3.385[↩]
- Kent G, Thompson JR. . Peliosis hepatis: involvement of the reticuloendothelial system . Arch Pathol 1961; 72:658–64.[↩]
- Hamilton FT, Lubitz JM. . Peliosis hepatis: report of three cases, with discussion of pathogenesis . Arch Pathol 1952; 54:564–72.[↩]
- Karasawa T, Shikata T, Smith RD. Peliosis hepatis. Report of nine cases. Acta Pathol Jpn. 1979 May;29(3):457-69. doi: 10.1111/j.1440-1827.1979.tb00201.x[↩]
- Asano S, Wakasa H, Kaise S, Nishimaki T, Kasukawa R. Peliosis hepatis. Report of two autopsy cases with a review of literature. Acta Pathol Jpn. 1982 Sep;32(5):861-77. doi: 10.1111/j.1440-1827.1982.tb03201.x[↩]
- Tada T, Wakabayashi T, Kishimoto H. Peliosis of the spleen. Am J Clin Pathol. 1983 Jun;79(6):708-13. doi: 10.1093/ajcp/79.6.708[↩]
- Ichijima K, Kobashi Y, Yamabe H, Fujii Y, Inoue Y. Peliosis hepatis. An unusual case involving multiple organs. Acta Pathol Jpn. 1980 Jan;30(1):109-20. doi: 10.1111/j.1440-1827.1980.tb01308.x[↩]
- Kumar Anup Anup-Kumar-Trikannad Ashwini, et al. Splenic Peliosis a unique case of spontaneous rupture. Medical and Research Publications. 2021;4(1[↩][↩]
- Podduturi V, Blessing MM. Fatal Hemoperitoneum Due to Isolated Splenic Peliosis. Am J Forensic Med Pathol. 2021 Mar 1;42(1):85-87. doi: 10.1097/PAF.0000000000000596[↩][↩]
- Agha RA, Sohrabi C, Mathew G, Franchi T, Kerwan A, O’Neill N; PROCESS Group. The PROCESS 2020 Guideline: Updating Consensus Preferred Reporting Of CasESeries in Surgery (PROCESS) Guidelines. Int J Surg. 2020 Dec;84:231-235. doi: 10.1016/j.ijsu.2020.11.005[↩]
- Celebrezze JP Jr, Cottrell DJ, Williams GB. Spontaneous splenic rupture due to isolated splenic peliosis. South Med J. 1998 Aug;91(8):763-4. doi: 10.1097/00007611-199808000-00014[↩]
- Etzion Y, Benharroch D, Saidel M, Riesenberg K, Gilad J, Schlaeffer F. Atraumatic rupture of the spleen associated with hemophagocytic syndrome and isolated splenic peliosis. Case report. APMIS. 2005 Jul-Aug;113(7-8):555-7. doi: 10.1111/j.1600-0463.2005.apm_165.x[↩][↩]
- Yasmin A, MccKay P, Jackson B. Splenic peliosis: an unusual cause of splenic lesions. Int J Hematol. 2008 Mar;87(2):107. doi: 10.1007/s12185-008-0024-4[↩][↩]
- Engel P, Tjalve E, Horn T. Peliosis of the spleen associated with a paraganglioma. Case report. Acta Radiol. 1993 Mar;34(2):148-9.[↩][↩][↩]
- Cochrane LB, Freson M. Peliosis of the spleen. Gastrointest Radiol. 1991 Winter;16(1):83-4. doi: 10.1007/BF01887312[↩][↩]
- Abo D, Terae S, Fukasawa Y, Seki H, Hasegawa Y, Sakuhara Y, Shimizu T, Shirato H. Splenic peliosis mimicking a solid tumor: findings in multimodalities. J Comput Assist Tomogr. 2008 Nov-Dec;32(6):890-2. doi: 10.1097/RCT.0b013e31815b3ef5[↩][↩][↩][↩]
- Giri N, Pitel PA, Green D, Alter BP. Splenic peliosis and rupture in patients with dyskeratosis congenita on androgens and granulocyte colony-stimulating factor. Br J Haematol. 2007 Sep;138(6):815-7. doi: 10.1111/j.1365-2141.2007.06718.x[↩][↩][↩]
- Körner M, Gebbers JO. Peliosis of the spleen and haemolytic anaemia. Histopathology. 2002 Aug;41(2):179-80. doi: 10.1046/j.1365-2559.2002.01424_8.x[↩][↩]
- Adachi K, Ui M, Nojima H, Takada Y, Enatsu K. Isolated splenic peliosis presenting with giant splenomegaly and severe coagulopathy. Am J Surg. 2011 Aug;202(2):e17-9. doi: 10.1016/j.amjsurg.2010.10.002[↩]
- Lal A, Singhal M, Sharma N, Bhalla A, Khandelwal N. Isolated splenic peliosis with spontaneous rupture after a viperine bite. Am J Emerg Med. 2014 Feb;32(2):194.e3-4. doi: 10.1016/j.ajem.2013.09.021[↩]
- Qureshi S, Choong AM, Tadrous PJ, Bhutiani RP. ‘Not just another appendicitis!’ – a case report of acute abdominal pain caused by splenic rupture secondary to isolated splenic peliosis. Ann R Coll Surg Engl. 2009 Nov;91(8):W1-4. doi: 10.1308/147870809X450610[↩]
- Shimono T, Yamaoka T, Nishimura K, Naya M, Hojo M, Yamamoto E, Mukaihara S, Hayakawa K. Peliosis of the spleen: splenic rupture with intraperitoneal hemorrhage. Abdom Imaging. 1998 Mar-Apr;23(2):201-2. doi: 10.1007/s002619900323[↩][↩][↩][↩]
- Rhu J, Cho J. Ruptured splenic peliosis in a patient with no comorbidity: A case report. World J Clin Cases. 2020 Feb 6;8(3):535-539. doi: 10.12998/wjcc.v8.i3.535[↩]
- Stojanović V, Mitić I, Jokić R, Vucković N, Doronjski A, Vijatov G, Milosević B, Djapić M. Splenic peliosis in the course of IgA nephropathy. Pediatr Nephrol. 2007 Dec;22(12):2137-40. doi: 10.1007/s00467-007-0598-8[↩][↩][↩]
- Tsokos M, Püschel K. Isolated peliosis of the spleen: report of 2 autopsy cases. Am J Forensic Med Pathol. 2004 Sep;25(3):251-4. doi: 10.1097/01.paf.0000127401.89952.65[↩][↩][↩]
- Ortega Deballon P, Ferńandez Lobato R, Ortega Muñoz P, Artuñedo Pé P, Ferńandez Luengas D, Moreno Azcoita M. Splenic peliosis: a cause of spontaneous splenic rupture. Surgery. 1999 Sep;126(3):585-6.[↩][↩][↩]
- Raghavan R, Alley S, Tawfik O, Webb P, Forster J, Uhl M. Splenic peliosis: a rare complication following liver transplantation. Dig Dis Sci. 1999 Jun;44(6):1128-31. doi: 10.1023/a:1026663821099[↩][↩][↩]
- Rege JD, Kavishwar VS, Mopkar PS. Peliosis of spleen presenting as splenic rupture with haemoperitoneum–a case report. Indian J Pathol Microbiol. 1998 Oct;41(4):465-7.[↩][↩][↩]
- Gugger M, Gebbers JO. Peliosis of the spleen: an immune-complex disease? Histopathology. 1998 Oct;33(4):387-9. doi: 10.1046/j.1365-2559.1998.00493.x[↩][↩][↩]
- Makdisi WJ, Cherian R, Vanveldhuizen PJ, Talley RL, Stark SP, Dixon AY. Fatal peliosis of the liver and spleen in a patient with agnogenic myeloid metaplasia treated with danazol. Am J Gastroenterol. 1995 Feb;90(2):317-8.[↩]
- Rhu J, Cho J. Ruptured splenic peliosis in a patient with no comorbidity: a case report. World J Clin Cases. 2020;8(3):535–539. doi: 10.12998/wjcc.v8.i3.535[↩]
- Lashbrook DJ, James RW, Phillips AJ, Holbrook AG, Agombar AC. Splenic peliosis with spontaneous splenic rupture: report of two cases. BMC Surg. 2006;6(1):1–7. doi: 10.1186/1471-2482-6-9[↩]
- Davidson J, Tung K. Splenic peliosis: an unusual entity. Br J Radiol. 2010;83(990):e126–e128. doi: 10.1259/bjr/71300465[↩][↩]
- Sharija S. Isolated peliosis lienis—a case report and literature review. J Foren Path. 2017;2:105.[↩][↩]
- Nassr B, Hassan WA, Nassr H, Allouzi A, Al-Shebly M. Isolated splenic peliosis: a case report. J Med Case Rep. 2023 Jun 30;17(1):298. doi: 10.1186/s13256-023-03929[↩]
- Choi G, Kim KA, Lee J, Park YS, Lee J, Choi JW, Lee CH. Ultrasonographic atlas of splenic lesions. Ultrasonography. 2022 Apr;41(2):416-429. doi: 10.14366/usg.21189[↩]
- Tavarozzi R, Borra T, Catania G, Depaoli L, Corsetti MT, Gaidano V, Limberti G, Ravazzoni F, Mariani N, Zallio F, Nozza P, Ladetto M. Spontaneous splenic rupture during induction therapy in acute myeloid leukemia: An unusual case. Radiol Case Rep. 2021 Sep 1;16(11):3406-3409. doi: 10.1016/j.radcr.2021.07.087[↩]
- Tsokos M, Erbersdobler A. Pathology of peliosis. Forensic Sci Int. 2005;149(1):25–33. doi: 10.1016/j.forsciint.2004.05.010[↩]
- Börcek P, Özdemir BH, Yılmaz Akçay E, Haberal M. Splenic Peliosis Resulting in Spontaneous Splenic Rupture in a Concomitant Hepatic and Renal Allograft Recipient. Exp Clin Transplant. 2016 Nov;14(Suppl 3):114-115.[↩][↩]
- Hakoda S, Shinya H, Kiuchi S. Spontaneous splenic rupture caused by splenic peliosis of a hemodialysis patient with chronic renal failure receiving erythropoietin. Am J Emerg Med. 2008 Jan;26(1):109.e1-2. doi: 10.1016/j.ajem.2007.03.025[↩][↩]
- Arai S, Asai T, Uozaki H, Hangaishi A, Kanda Y, Motokura T, Chiba S, Kurokawa M. Splenic peliosis in a patient with aplastic anemia during danazol therapy. Int J Hematol. 2007 Nov;86(4):329-32. doi: 10.1532/IJH97.07078[↩][↩]
- Townsley DM, et al. Danazol treatment for telomere diseases. N. Engl. J. Med. 2016;374:1922–1931. doi: 10.1056/NEJMoa1515319[↩]
- Khincha PP, Wentzensen IM, Giri N, Alter BP, Savage SA. Response to androgen therapy in patients with dyskeratosis congenita. Br. J. Haematol. 2014;165:349–357. doi: 10.1111/bjh.12748[↩]
- Khincha PP, et al. Similar telomere attrition rates in androgen-treated and untreated patients with dyskeratosis congenita. Blood Adv. 2018;2:1243–1249. doi: 10.1182/bloodadvances.2018016964[↩]
- Han JS, Oh SY, Kim SH, Kwon HC, Hong SH, Han JY, Park KJ, Kim HJ. A case of pathologic splenic rupture as the initial manifestation of acute myeloid leukemia M2. Yonsei Med J. 2010 Jan;51(1):138-40. doi: 10.3349/ymj.2010.51.1.138[↩]
- Begum S, Khan MR. Splenic peliosis and rupture—a surgical emergency: case report and review of the available literature. J Appl Hematol. 2016;7:143–147. doi: 10.4103/1658-5127.198508[↩]
- Abbott RM, Levy AD, Aguilera NS, Gorospe L, Thompson WM. From the archives of the AFIP: primary vascular neoplasms of the spleen: radiologic-pathologic correlation. Radiographics. 2004 Jul-Aug;24(4):1137-63. doi: 10.1148/rg.244045006[↩]
- Lashbrook, D.J., James, R.W., Phillips, A.J. et al. Splenic peliosis with spontaneous splenic rupture: report of two cases. BMC Surg 6, 9 (2006). https://doi.org/10.1186/1471-2482-6-9[↩][↩][↩]
- Kohr RM, Haendiges M, Taube RR. Peliosis of the spleen: a rare cause of spontaneous splenic rupture with surgical implications. Am Surg. 1993 Mar;59(3):197-9.[↩]
- Katkhouda N, Mavor E. Laparoscopic Splenectomy. Surg Clin North Am. 2000;80:1285–97. doi: 10.1016/S0039-6109(05)70225-5[↩]
- Zia H, Zemon H, Brody F. Laparoscopic splenectomy for isolated sarcoidosis of the spleen. J Laparoendosc Adv Surg Tech A. 2005;15:160–2. doi: 10.1089/lap.2005.15.160[↩]
- Steed D, Collins J, Farris AB, Guarner J, Yarar D, Friedman-Moraco R, Doane T, Pouch S, Marshall Lyon G 3rd, Woodworth MH. Haemophagocytic lymphohistiocytosis associated with bartonella peliosis hepatis following kidney transplantation in a patient with HIV. Lancet Infect Dis. 2022 Oct;22(10):e303-e309. doi: 10.1016/S1473-3099(22)00276-6[↩][↩][↩][↩][↩][↩][↩]
- Maurin M, Birtles R, Raoult D. Current knowledge of Bartonella species. Eur J Clin Microbiol Infect Dis. 1997 Jul;16(7):487-506. doi: 10.1007/BF01708232[↩]
- Angelakis E, Raoult D. Pathogenicity and treatment of Bartonella infections. Int J Antimicrob Agents. 2014 Jul;44(1):16-25. doi: 10.1016/j.ijantimicag.2014.04.006[↩]
- Mohle-Boetani JC, Koehler JE, Berger TG, LeBoit PE, Kemper CA, Reingold AL, Plikaytis BD, Wenger JD, Tappero JW. Bacillary angiomatosis and bacillary peliosis in patients infected with human immunodeficiency virus: clinical characteristics in a case-control study. Clin Infect Dis. 1996 May;22(5):794-800. doi: 10.1093/clinids/22.5.794[↩][↩]
- Mosepele M, Mazo D, Cohn J. Bartonella infection in immunocompromised hosts: immunology of vascular infection and vasoproliferation. Clin Dev Immunol. 2012;2012:612809. doi: 10.1155/2012/612809[↩][↩]
- Lamps LW, Gray GF, Scott MA. The histologic spectrum of hepatic cat scratch disease. A series of six cases with confirmed Bartonella henselae infection. Am J Surg Pathol. 1996 Oct;20(10):1253-9. doi: 10.1097/00000478-199610000-00011[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Lenoir AA, Storch GA, DeSchryver-Kecskemeti K, Shackelford GD, Rothbaum RJ, Wear DJ, Rosenblum JL. Granulomatous hepatitis associated with cat scratch disease. Lancet. 1988 May 21;1(8595):1132-6. doi: 10.1016/s0140-6736(88)91952-6[↩][↩][↩][↩][↩][↩][↩]
- Arisoy ES, Correa AG, Wagner ML, Kaplan SL. Hepatosplenic cat-scratch disease in children: selected clinical features and treatment. Clin Infect Dis. 1999 Apr;28(4):778-84. doi: 10.1086/515197[↩][↩][↩][↩][↩][↩][↩][↩]
- Bass JW, Vincent JM, Person DA. The expanding spectrum of Bartonella infections: II. Cat-scratch disease. Pediatr Infect Dis J. 1997 Feb;16(2):163-79. doi: 10.1097/00006454-199702000-00002[↩][↩]
- Bächler P, Baladron MJ, Menias C, Beddings I, Loch R, Zalaquett E, Vargas M, Connolly S, Bhalla S, Huete Á. Multimodality Imaging of Liver Infections: Differential Diagnosis and Potential Pitfalls. Radiographics. 2016 Jul-Aug;36(4):1001-23. doi: 10.1148/rg.2016150196[↩]
- Ventura A, Massei F, Not T, Massimetti M, Bussani R, Maggiore G. Systemic Bartonella henselae infection with hepatosplenic involvement. J Pediatr Gastroenterol Nutr. 1999 Jul;29(1):52-6. doi: 10.1097/00005176-199907000-00014[↩][↩][↩]
- Carithers HA. Cat-scratch disease. An overview based on a study of 1,200 patients. Am J Dis Child. 1985 Nov;139(11):1124-33. doi: 10.1001/archpedi.1985.02140130062031[↩]
- Margileth AM. Cat scratch disease. Adv Pediatr Infect Dis. 1993;8:1-21.[↩][↩]
- Chomel BB, Boulouis HJ, Breitschwerdt EB. Cat scratch disease and other zoonotic Bartonella infections. J Am Vet Med Assoc. 2004 Apr 15;224(8):1270-9. doi: 10.2460/javma.2004.224.1270[↩]
- Chomel BB, Kasten RW, Floyd-Hawkins K, Chi B, Yamamoto K, Roberts-Wilson J, Gurfield AN, Abbott RC, Pedersen NC, Koehler JE. Experimental transmission of Bartonella henselae by the cat flea. J Clin Microbiol. 1996 Aug;34(8):1952-6. doi: 10.1128/jcm.34.8.1952-1956.1996[↩]
- Chomel BB, Abbott RC, Kasten RW, Floyd-Hawkins KA, Kass PH, Glaser CA, Pedersen NC, Koehler JE. Bartonella henselae prevalence in domestic cats in California: risk factors and association between bacteremia and antibody titers. J Clin Microbiol. 1995 Sep;33(9):2445-50. doi: 10.1128/jcm.33.9.2445-2450.1995[↩]
- Scott MA, McCurley TL, Vnencak-Jones CL, Hager C, McCoy JA, Anderson B, Collins RD, Edwards KM. Cat scratch disease: detection of Bartonella henselae DNA in archival biopsies from patients with clinically, serologically, and histologically defined disease. Am J Pathol. 1996 Dec;149(6):2161-7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1865337/pdf/amjpathol00036-0364.pdf[↩]
- Hajjaji N, Hocqueloux L, Kerdraon R, Bret L. Bone infection in cat-scratch disease: a review of the literature. J Infect. 2007 May;54(5):417-21. doi: 10.1016/j.jinf.2006.10.045[↩]
- Taxy JB. Peliosis: a morphologic curiosity becomes an iatrogenic problem. Hum Pathol. 1978 May;9(3):331-40. doi: 10.1016/s0046-8177(78)80090-2[↩]
- Malatack JJ, Jaffe R. Granulomatous hepatitis in three children due to cat-scratch disease without peripheral adenopathy. An unrecognized cause of fever of unknown origin. Am J Dis Child. 1993 Sep;147(9):949-53. doi: 10.1001/archpedi.1993.02160330039014[↩][↩][↩]
- Theresa A. Steeper, Hanan Rosenstein, John Weiser, Subbarao Inampudi, Dale C. Snover, Bacillary Epithelioid Angiomatosis Involving the Liver, Spleen, and Skin in an AIDS Patient with Concurrent Kaposi’s Sarcoma, American Journal of Clinical Pathology, Volume 97, Issue 5, 1 May 1992, Pages 713–718, https://doi.org/10.1093/ajcp/97.5.713[↩]
- Steeper TA, Rosenstein H, Weiser J, Inampudi S, Snover DC. Bacillary epithelioid angiomatosis involving the liver, spleen, and skin in an AIDS patient with concurrent Kaposi’s sarcoma. Am J Clin Pathol. 1992 May;97(5):713-8. doi: 10.1093/ajcp/97.5.713[↩][↩]
- VanderHeyden TR, Yong SL, Breitschwerdt EB, Maggi RG, Mihalik AR, Parada JP, Fimmel CJ. Granulomatous hepatitis due to Bartonella henselae infection in an immunocompetent patient. BMC Infect Dis. 2012 Jan 23;12:17. doi: 10.1186/1471-2334-12-17[↩][↩]
- Liston TE, Koehler JE. Granulomatous hepatitis and necrotizing splenitis due to Bartonella henselae in a patient with cancer: case report and review of hepatosplenic manifestations of bartonella infection. Clin Infect Dis. 1996 Jun;22(6):951-7. doi: 10.1093/clinids/22.6.951[↩]
- Zimmermann P, Ziesenitz VC, Curtis N, Ritz N. The Immunomodulatory Effects of Macrolides-A Systematic Review of the Underlying Mechanisms. Front Immunol. 2018 Mar 13;9:302. doi: 10.3389/fimmu.2018.00302[↩]
- Rolain JM, Brouqui P, Koehler JE, Maguina C, Dolan MJ, Raoult D. Recommendations for treatment of human infections caused by Bartonella species. Antimicrob Agents Chemother. 2004 Jun;48(6):1921-33. doi: 10.1128/AAC.48.6.1921-1933.2004[↩][↩]
- Stoler MH, Bonfiglio TA, Steigbigel RT, Pereira M. An atypical subcutaneous infection associated with acquired immune deficiency syndrome. Am J Clin Pathol. 1983 Nov;80(5):714-8. doi: 10.1093/ajcp/80.5.714[↩]
- Koehler JE, Tappero JW. Bacillary angiomatosis and bacillary peliosis in patients infected with human immunodeficiency virus. Clin Infect Dis. 1993 Oct;17(4):612-24. doi: 10.1093/clinids/17.4.612[↩][↩][↩][↩][↩][↩][↩]
- Tappero JW, Koehler JE, Berger TG, Cockerell CJ, Lee TH, Busch MP, Stites DP, Mohle-Boetani J, Reingold AL, LeBoit PE. Bacillary angiomatosis and bacillary splenitis in immunocompetent adults. Ann Intern Med. 1993 Mar 1;118(5):363-5. doi: 10.7326/0003-4819-118-5-199303010-00007[↩]
- Lucey D, Dolan MJ, Moss CW, Garcia M, Hollis DG, Wegner S, Morgan G, Almeida R, Leong D, Greisen KS, et al. Relapsing illness due to Rochalimaea henselae in immunocompetent hosts: implication for therapy and new epidemiological associations. Clin Infect Dis. 1992 Mar;14(3):683-8. doi: 10.1093/clinids/14.3.683[↩]
- Slater LN, Welch DF, Hensel D, Coody DW. A newly recognized fastidious gram-negative pathogen as a cause of fever and bacteremia. N Engl J Med. 1990 Dec 6;323(23):1587-93. doi: 10.1056/NEJM199012063232303[↩][↩]
- Tappero JW, Koehler JE. Cat-scratch Disease and Bacillary Angiomatosis. JAMA. 1991;266(14):1938–1939. doi:10.1001/jama.1991.03470140050020[↩]
- Koehler JE, Quinn FD, Berger TG, LeBoit PE, Tappero JW. Isolation of Rochalimaea species from cutaneous and osseous lesions of bacillary angiomatosis. N Engl J Med. 1992 Dec 3;327(23):1625-31. doi: 10.1056/NEJM199212033272303[↩][↩][↩]
- Koehler JE, Sanchez MA, Garrido CS, Whitfeld MJ, Chen FM, Berger TG, Rodriguez-Barradas MC, LeBoit PE, Tappero JW. Molecular epidemiology of bartonella infections in patients with bacillary angiomatosis-peliosis. N Engl J Med. 1997 Dec 25;337(26):1876-83. doi: 10.1056/NEJM199712253372603[↩][↩]
- Maurin M, Raoult D. Antimicrobial susceptibility of Rochalimaea quintana, Rochalimaea vinsonii, and the newly recognized Rochalimaea henselae. J Antimicrob Chemother. 1993 Oct;32(4):587-94. doi: 10.1093/jac/32.4.587[↩]
- Koehler JE. Bartonella-associated infections in HIV-infected patients. AIDS Clin Care. 1995 Dec;7(12):97-102.[↩]
- Ramírez Ramírez CR, Saavedra S, Ramírez Ronda CH. Bacillary angiomatosis: microbiology, histopathology, clinical presentation, diagnosis and management. Bol Asoc Med P R. 1996 Apr-Jun;88(4-6):46-51.[↩]
- Koehler, J. E., and D. A. Relman. 2002. Bartonella species, p. 925-931. In V. L. Yu, R. Weber, and D. Raoult (ed.), Antimicrobial therapy and vaccines. Apple Trees Production, LLC, New York, N.Y.[↩][↩][↩]
- Berger TG, Perkocha LA. Bacillary angiomatosis. AIDS Clin Rev. 1991:81-95.[↩]
- Gazineo JL, Trope BM, Maceira JP, May SB, Coelho JM, Lambert JS, Nogueira SA. Bacillary angiomatosis: description of 13 cases reported in five reference centers for AIDS treatment in Rio de Janeiro, Brazil. Rev Inst Med Trop Sao Paulo. 2001 Jan-Feb;43(1):1-6. doi: 10.1590/s0036-46652001000100001[↩]
- Krekorian TD, Radner AB, Alcorn JM, Haghighi P, Fang FC. Biliary obstruction caused by epithelioid angiomatosis in a patient with AIDS. Am J Med. 1990 Dec;89(6):820-2. doi: 10.1016/0002-9343(90)90231-2[↩]
- Szaniawski WK, Don PC, Bitterman SR, Schachner JR. Epithelioid angiomatosis in patients with AIDS. Report of seven cases and review of the literature. J Am Acad Dermatol. 1990 Jul;23(1):41-8. doi: 10.1016/0190-9622(90)70183-i[↩]
- Cotell SL, Noskin GA. Bacillary angiomatosis. Clinical and histologic features, diagnosis, and treatment. Arch Intern Med. 1994 Mar 14;154(5):524-8. doi: 10.1001/archinte.154.5.524[↩][↩]