Contents
What is a sauna
The sauna (or Finnish bath) has dry air and a high temperature, unlike the Turkish bath 1. Traditional saunas use either wood stoves or 220-V heaters to heat the air to approximately 85°C, which then heats the occupant, mainly via convection 2. Whilst the basic modern sauna is an unpainted, wood-panelled room with wooden platforms and a rock-filled electric heater. The walls are made of spruce or pine, and the benches are made of obeche, spruce or aspen because these types of wood are less hot to sit on. The recommended temperature is 80°C to 100°C at the level of the bather’s face and 30°C at floor level 3. The air should have a relative humidity of 10% to 20% 4. A good sauna has efficient ventilation; the air should change three to eight times per hour 3.
In a sauna, the skin temperature increases rapidly to approximately 40°C 5, whereas the increase in rectal temperature depends on heat exposure 6. Sweating begins quickly and reaches its maximum at approximately 15 min, with an average total secretion of 0.5 kg 7. Skin blood flow increases from 5% to 10%, becoming 50% to 70% of the cardiac output, while blood flow to internal organs decreases 8. Cardiac output increases by 60% to 70% in relation to the increase in heart rate 8, while cardiac stroke volume does not change 9.
Very few sudden deaths take place during or after sauna bathing. Of all sudden deaths in Finland within one year, only 102 (1.7%) occurred within 24 h of the sauna bath 10. One-third of these were accidental, due to consumption of alcohol or drowning; the majority of the non-accidental deaths were due to acute myocardial infarction (heart attack) in which alcohol intake was an important contributing factor 10. An epidemiological study by Romo 11 showed that very few acute heart attacks occur during sauna bathing. Of 1631 acute heart attacks or sudden coronary deaths that occurred during a 16-month period in Helsinki, Finland, ischemic symptoms began or death occurred within 3 h of sauna bathing in only 29 patients (1.8%) 11.
In a large prospective study of 12,310 Finnish men and women, 30 to 59 years of age, there were 77 sudden coronary deaths during the six-year follow-up period but only two of these occurred in saunas 12.
Generally, sauna bathing has been contraindicated for patients with chronic heart failure 13. However, it has been well tolerated and improved hemodynamics has been shown in patients with chronic heart failure after a single exposure and after a four-week period of sauna bathing (five days per week). Left ventricular ejection fraction increased from 24±7% to 31±9% and left ventricular end-diastolic dimension decreased from 66±6 mm to 62±5 mm after four weeks 14.
Sauna bathing has the potential to be an effective therapeutic option for patients with hypertension or heart failure, as well as for patients with known coronary artery disease risk factors. The common mechanism of action is improvement in vascular endothelial function, which reduces cardiac preload and after-load. It would also be interesting to know – provided the improvement is permanent – whether some of the damage on the cardiovascular system can be reversed by sauna therapy. Sauna bathing can be risky in patients receiving alcohol, beta blockade and nitrates 15.
Severe aortic stenosis, unstable angina pectoris and recent heart attack are contraindications to sauna bathing 16. Decompensated heart failure and cardiac arrhythmia are relative contraindications 17. Elderly persons prone to orthostatic hypotension should be cautious in the sauna because a decrease in blood pressure may cause syncope, usually just after sauna bathing 17.
Sauna in healthy volunteers and in patients with heart failure
The physiological effects of exposure to Finnish saunas (80°C to 90°C with 30% to 40% relative humidity) were investigated in 60 healthy volunteers (33 men and 27 women, aged 18 to 63 years). Marked physiological changes appeared in the first few minutes in the sauna without any prodromal warning. After 20 min the mean heart rate was 143±25 beats/min, mean rectal temperature was 38.6±0.6°C and skin temperature was 40.4±1°C. In addition, mean systolic blood pressure was 130.5±26.6 mmHg and mean diastolic blood pressure was 66.6±15.9 mmHg. Three subjects experienced syncope, and one developed an anginal attack with electrocardiogram changes suggestive of an acute coronary event 18.
Recently, the temperature of sauna baths worldwide was decreased to 60°C, and a study 19 in Japan evaluated the safety and efficacy of repeated 60°C sauna bathing in patients with chronic systolic heart failure. This study included 15 heart failure patients with New York Heart Association (NYHA) class III, all in stable condition. Sauna bathing was performed once per day for four weeks. No adverse effect was observed. Symptoms improved in 13 of 15 patients after four weeks. Sauna bathing decreased systolic blood pressure without affecting heart rate, resulting in a significant decrease in the rate-pressure product. Left ventricular ejection fraction, by echocardiography, was significantly increased from 30±11% to 34±11%. Sauna bathing significantly improved exercise tolerance manifested by prolonged 6 min walking distance, increased peak respiratory oxygen uptake and enhanced anaerobic threshold. It also significantly reduced plasma adrenaline and noradrenaline levels, and reduced the number of hospital admissions for heart failure.
Sauna health benefits
The acute hemodynamic effects of thermal vasodilation were studied in 34 patients with chronic heart failure (most of them in NYHA class III and IV with a mean age of 58 years and a mean ejection fraction of 25±9%) 14. All patients had a sauna bath for 15 min at 60°C. There was a mild increase in oxygen consumption, a 1.2°C increase in pulmonary arterial blood temperature and the heart rate increased by 20 to 25 beats/min at the end of the treatment. Systolic blood pressure showed no significant change; however, the diastolic blood pressure decreased significantly during the sauna treatment. Cardiac and stroke indexes increased and systemic vascular resistances decreased significantly during and after the treatment. This study 14 has demonstrated that sauna baths improve the hemodynamics in patients with heart failure.
Sauna treatment has increased cardiac output and peripheral perfusion 20, and improved hemodynamic variables and clinical symptoms 14 in patients with heart failure.
Experimental studies 21, 22 have demonstrated that heart failure impairs endothelial-dependent vasodilation. One of the proposed mechanisms by which this occurs is through decreased peripheral vascular production of endothelium-derived nitric oxide in patients with heart failure 23. Shear stress is an important stimulus for nitric oxide production 24 and the expression of eNOS [endothelial nitric oxide synthase (eNOS)] 25. Several studies 26, 27 have shown that endothelial function in patients with heart failure was improved by treatment with L-arginine, angiotensin-converting enzyme inhibitors 28, physical training 29, dobutamine 30 or oral vitamin C 31.
In another study 32, 20 heart failure patients (30 to 75 years of age) with NYHA class II and III were studied. Mean left ventricular ejection fraction was 38±14%. Ten patients served as the control group. Thermal therapy with a far infrared dry sauna was performed. Patients were placed in a supine position on a bed in a 60°C sauna for 15 min, and once removed, kept on bedrest for another 30 min with a blanket to keep them warm. In the control group, patients were placed in a supine position on a bed in a temperature-controlled (24°C) room for 45 min. Two weeks of sauna therapy improved endothelial function (measured by the noninvasive brachial artery method) and decreased plasma brain natriuretic peptide (BNP) concentrations in patients with heart failure. A correlation was found between the degree of improvement in the per cent flow-mediated dilation (%FMD) and plasma brain natriuretic peptide concentrations. Repeated 60°C sauna therapy improved peripheral vascular endothelial function, resulting in an improvement in cardiac function in patients with heart failure.
This study 32 demonstrated that endothelial function in the brachial artery significantly improved after two weeks of sauna therapy. It was also found that systemic vascular resistance significantly decreased after two weeks of sauna therapy, suggesting an improvement in endothelial function in resistance vessels. Improved endothelial function leads to dilation of vessels by an increase in NO (nitric oxide) production. The fact that two weeks of sauna therapy significantly decreased systolic blood pressure in this study may reflect the improvement in endothelial function. This results in decreased afterload and, thus, increased cardiac output. These changes improved peripheral circulation, which was probably responsible for the improvement in clinical symptoms. A significant improvement in the per cent flow-mediated dilation (%FMD) was observed in patients whose clinical symptoms improved, whereas the per cent flow-mediated dilation (%FMD) did not improve in patients whose clinical symptoms did not change 32. The improvement in endothelial function after long-term repeated sauna therapy was most probably due to improved NO production by eNOS [endothelial nitric oxide synthase (eNOS)] upregulation in patients with heart failure. Endothelial nitric oxide synthase (eNOS) upregulation was due to a prolonged increase in shear stress; endothelial nitric oxide synthase (eNOS) upregulation in the coronary artery may have directly improved cardiac function due to an increase in coronary perfusion 32.
Another important issue in heart failure is ventricular arrhythmias. In a study by Giannetti et al 33, 30 patients (59±3 years of age) with NYHA class II or III and at least 200 premature ventricular complexes in 24 h were studied. They were randomly assigned into sauna-treated (20 patients) or nontreated (10 patients) groups. The sauna-treated group underwent a two-week program of a daily 60°C far infrared dry sauna for 15 min, followed by 30 min bedrest with blankets, five days a week. Patients in the nontreated group had bedrest in a temperature-controlled room (24°C) for 45 min. The total number of premature ventricular complexes per 24 h in the sauna-treated group decreased compared with that of the non-treated group (848 versus 3097). Heart rate variability increased and plasma brain natriuretic peptide (BNP) concentrations decreased in the sauna-treated group compared with those in the nontreated group. This study 33 has demonstrated that sauna treatment improved ventricular arrhythmias in patients with heart failure.
Fifteen patients with heart failure (mean age 64 years with NYHA class II or III) were assigned to six weeks of hydrotherapy or six weeks’ restriction in a crossover intervention trial 34. Patients with hydrotherapy had a significant improvement in mood, physical capacity and enjoyment, and a significant reduction in heart failure-related symptoms compared with those of the restricted patients. Patients’ heart rates at rest and at 50 W workload were significantly reduced by hydrotherapy. In conclusion, the use of a home-based hydrotherapy sauna was found to improve quality of life, heart failure-related symptoms and heart rate response to exercise in patients with mild chronic heart failure 34.
Another study 35 evaluated the effects of thermal therapy on endothelial function in patients with coronary risk factors. Twenty-five men with at least one coronary risk factor and 10 healthy men without coronary risk factors were enrolled. Patients in the risk group were treated with a 60°C far infrared dry sauna for 15 min and then kept in a bed covered with blankets for 30 min, once a day for two weeks. To assess endothelial function, brachial artery diameter was measured at rest, during reactive hyperemia (%FMD), again at rest and after sublingual nitroglycerin administration (per cent flow independent dilation [%FID]) using high-resolution ultrasound. At the start of the study, the %FMD was significantly impaired in the risk group compared with the level in the control group, while the %FID was similar in both groups. Two weeks of sauna therapy significantly improved the %FMD in the risk group; however, the %FID did not change. Repeated sauna treatment improved impaired vascular endothelial function in the setting of coronary risk factors, suggesting a therapeutic role for sauna treatment in patients with risk factors for atherosclerosis 35.
Summary
Repeated sauna therapy (60°C for 15 min) improved hemodynamic parameters, clinical symptoms, cardiac function and vascular endothelial function in patients with heart failure. In patients with heart failure, clinical symptoms such as fatigue, heaviness in the limbs, edema, appetite loss and constipation are often observed due to increased peripheral vascular resistance and reduced peripheral perfusion. Sauna therapy improved the cardiac index, mean pulmonary wedge pressure, systemic and pulmonary vascular resistance, and cardiac function. Sauna treatment is considered safe for heart failure patients with NYHA classes I, II and III. It seems that sauna treatment may help improve clinical symptoms and hemodynamic parameters secondary to an improvement in the endothelial function of patients with heart failure whose endothelial function is impaired 13.
Infrared sauna benefits
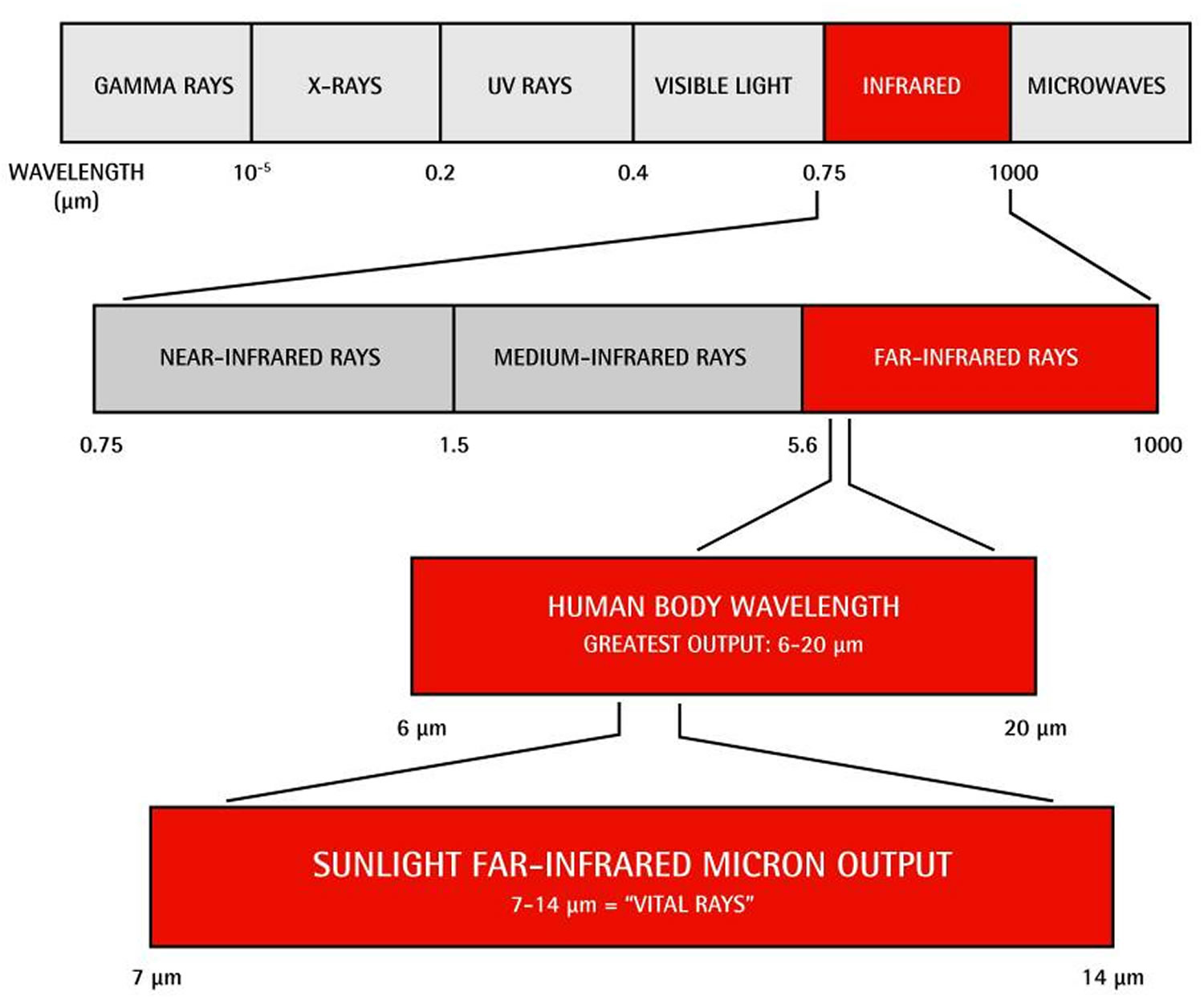
Traditional saunas use either wood stoves or 220-V heaters to heat the air to approximately 85°C, which then heats the occupant, mainly via convection. Some people find the traditional sauna (Finnish sauna) heat uncomfortable. In contrast, far-infrared saunas heat to approximately 60°C, providing a more comfortable and relaxing experience 2. Far-infrared saunas utilize 120-V infrared elements, similar to the infrared warmers on neonatal resuscitation beds, to radiate heat with a wavelength of around 10 μm (see Figure 1). As infrared heat penetrates more deeply than warmed air, users develop a more vigorous sweat at a lower temperature than they would in traditional saunas. The cardiovascular demand imparted by thermoregulatory homeostasis (sweating, vasodilation, decreased afterload, increased heart rate, increased cardiac output) is similar to that achieved by walking at a moderate pace. As such, far-infrared saunas might be of particular benefit to those who are sedentary due to various medical conditions, such as osteoarthritis or cardiovascular or respiratory problems 2.
Far-infrared saunas are approved by the Canadian Standards Association and are sold to the public as recreational saunas. Manufacturers advertise numerous therapeutic effects, including weight loss, improved cardiovascular health, normalized blood pressure (BP), lowered cholesterol, and reduced pain, stress, and fatigue 36.
Figure 1. Light spectrum used in far-infrared saunas
[Source 2]
Table 1. Studies supporting use of Far-infrared saunas
| STUDY | N | DURATION | LEVEL OF EVIDENCE | INDICATION | PRIMARY OUTCOMES | COMMENTS |
|---|---|---|---|---|---|---|
| Kihara et al, 2004 37 | 30 | 14 d | I | Congestive heart failure | Fewer PVCs and lowered BNP levels | Infrared sauna treatment improves ventricular arrhythmias in patients with CHF |
| Kihara et al, 2002 38 | 30 | 14 d | I | Congestive heart failure | Clinical symptoms improved and BNP levels decreased; endothelial function improved | Clinical improvement in CHF seen after FIRS therapy might be owing to improved endothelial function |
| Miyamoto et al, 2005 39 | 15 | 28 d | II | Congestive heart failure | CHF symptoms, ejection fraction, and walking distance improved; catecholamine levels decreased | Infrared sauna therapy is a safe and effective treatment of CHF |
| Sugahara et al, 2003 40 | 12 | 28 d | II | Pediatric CHF due to VSD | Clinical signs and symptoms of CHF decreased, urine nitrate levels decreased, and 75% of patients avoided surgical repair | Infrared sauna therapy is safe and might be helpful in treating pediatric CHF; VSDs have a high incidence of spontaneous improvement |
| Masuda et al, 2004 41 | 28 | 14 d | I | Prevention of atherosclerosis by decreasing oxidative stress | Systolic BP decreased, as did serum PGF2α | Infrared sauna therapy might be cardioprotective |
| Imamura et al, 2001 42 | 35 | 14 d | II | Impaired endothelial function in those with coronary risk factors | Endothelium-dependent vasodilatation improved; BP and fasting glucose levels decreased | Infrared sauna therapy is safe and improves vascular endothelial function |
| Biro et al, 2003 43 | 10 | 14 d | II | Obesity | Despite weight loss, plasma ghrelin and leptin levels did not change | Infrared sauna therapy might blunt neurohormonal effects of weight loss due to calorie restriction |
| Masuda et al, 2005 44 | 46 | 28 d | I | Chronic pain | Increased likelihood of return to work; trend toward decreased pain and anger | Infrared sauna therapy might be promising for the treatment of chronic pain |
| Masuda et al, 2005 45 | 2 | 35 d | II* | Chronic fatigue syndrome | Fatigue, pain, sleep, and low- grade fever improved | Infrared sauna treatment might be helpful for chronic fatigue syndrome |
BNP—brain natriuretic peptide, BP—blood pressure, CHF—congestive heart failure, FIRS—far-infrared sauna, PGF2α—prostaglandin F2α, PVC—premature ventricular contraction, VSD— ventricular septal defect.
Levels of evidence
Level I: At least one properly conducted randomized controlled trial, systematic review, or meta-analysis
Level II: Other comparison trials, non-randomized, cohort, case-control, or epidemiologic studies, and preferably more than one study
Level III: Expert opinion or consensus statements
Congestive heart failure (CHF)
Four papers support the use of infrared sauna therapy for those with CHF (congestive heart failure) 37, 38, 39, 40.
In one randomized controlled trial 37 (level I evidence), 30 subjects with New York Heart Association (NYHA) class II or III heart failure and more than 200 premature ventricular contractions (PVCs) per 24 hours were randomized into treatment and nontreatment groups. Treatment consisted of 10, 15-minute infrared sauna sessions over a 2-week period. After 2 weeks the sauna group had significantly fewer premature ventricular contractions (mean = 848 vs 3097 per 24 hours) and lower brain natriuretic peptide (BNP) levels (mean = 229 pg/mL vs 419 pg/mL) compared with the untreated group 37. Secondary findings included significant improvements to NYHA heart failure class (class II/III = 5/15 before treatment vs 15/5 after treatment), weight (mean = 57 kg before vs 56 kg after), cardiothoracic ratio (mean = 59% before vs 56% after), and left ventricular ejection fraction (LVEF) (mean = 29% before vs 33% after); although BP levels appeared to decrease, results did not reach statistical significance. The authors concluded that repeated far-infrared saunas treatment improved ventricular arrhythmias in patients with NYHA class II and III heart failure 37. Strengths of this study included a control group that was well matched; although the study was not blinded, the control group relaxed in an environment similar to that of the far-infrared saunas (but at 24°C) for the same amount of time. All measurements taken after the intervention were performed on the day after the last treatment and can therefore be considered reliable, as this allowed adequate time for rehydration and fluid compartment redistribution. Patients in this study population, however, had asymptomatic premature ventricular contractions; as such, we cannot generalize these findings to those with symptomatic premature ventricular contractions, although the improvement in NYHA class is clinically relevant.
Another randomized controlled trial 38 (level I evidence) studied 30 patients with NYHA class II or III heart failure: 10 control subjects matched to 20 intervention subjects. The intervention group had 10, 15-minute far-infrared saunas treatments over a 2-week period. There were no adverse events and there were no changes in liver or renal function, electrolyte levels, or hematocrit levels. Clinical symptoms improved in 17 out of 20 patients in the intervention group and were unchanged in the remaining 3 patients 38. Patients’ NYHA classes improved (NYHA class I/II/III = 0/10/10 before treatment vs 1/14/5 after treatment), as did their systolic BP (mean = 107 mm Hg before vs 97 mm Hg after). Diastolic BP, body weight, and left ventricular ejection fraction (LVEF) did not improve significantly. Brain natriuretic peptide levels decreased significantly (mean = 441 pg/mL before vs 293 pg/mL after). In contrast, the control group did not improve either clinically or in terms of brain natriuretic peptide levels. Endothelial function, as assessed by high-resolution ultrasound to determine endothelium-dependent flow-mediated dilation, improved with treatment (mean = 4.4% vs 5.7%, n = 20). The authors 38 concluded that far-infrared saunas treatment improved cardiac function and clinical symptoms in those with heart failure and that this improvement was the result of improved vascular endothelial function. Strengths of the study were similar to those in the study by Kihara et al 37 . Furthermore, before study entry all subjects were in stable clinical condition for at least 1 month, medications had not been changed for at least 2 weeks, and adverse events were defined (dyspnea, angina pectoris, palpitations, hypotension, or dehydration). Although clinical symptoms were assessed using an unvalidated tool, it is reassuring to note the correlation between flow-mediated dilation improvement and both clinical and brain natriuretic peptide improvement.
In a sequential, longitudinal, interrupted time series trial 39 (level II evidence), 15 hospitalized patients with NYHA class II or III heart failure underwent daily 15-minute far-infrared saunas treatment for 4 weeks. Sauna treatment was safely completed without any adverse effects. Symptoms improved in 13 of 15 patients (87%); LVEF increased (mean = 30% vs 34%); every patient’s 6-minute walking distance increased (mean = 388 m vs 448 m); and plasma epinephrine and norepinephrine concentrations both decreased (mean = 40 pg/mL vs 21 pg/mL, 633 pg/mL vs 443 pg/mL, respectively). Further, systolic BP and cardiothoracic ratios improved (mean = 101 mm Hg vs 98 mm Hg, 59% vs 58%, respectively). Changes in weight and brain natriuretic peptide levels did not reach statistical significance, nor did changes in NYHA class (class II/III = 3/12 vs 6/9). The authors 39 concluded that far-infrared saunas treatment was a safe and effective adjunct therapy for heart failure. This is the longest study of far-infrared saunas therapy for heart failure; it is also the only study to document the effect of far-infrared saunas therapy on exercise tolerance. It was, however, not randomized and had a small sample size. Further, adverse events were not defined, clinical symptoms were not evaluated using validated questionnaires, and measurements were taken “immediately after” participants’ last sauna sessions.
Although an additional study supports the use of far-infrared saunas therapy for neonates with heart failure40 , review of that study is beyond the scope of this paper, as the cause of neonatal heart failure is fundamentally different from that of adult heart failure. A brief overview is provided in Table 1 above.
Regular saunas can harm sperm quality
Researchers wanted to look at the effect of long-term sauna use on sperm quality, as previous research has indicated that higher scrotal temperatures in men can lead to poorer sperm health. The researchers 46 recruited 10 healthy male volunteers (average age 33.2 years) to take part in a Finnish sauna programme consisting of two sauna sessions per week for three months at 80-90°C, each lasting 15 minutes. Very few details about the men were reported, so their nationality, demographics and lifestyle behaviors are unknown. Men who had used a sauna in the previous year were not included in the study, so the 10 men didn’t have a history of regular sauna exposure.
Sex hormones, sperm parameters, sperm DNA structure, sperm death (‘apoptosis’, a process where the sperm self-destructs in response to external stimuli) and the expression of genes involved in sperm response to heat and lack of oxygen were all evaluated at the start (baseline). Further measurements were then taken:
- at the end of each sauna exposure
- at the end of the sauna programme (three months after the baseline)
- six months after the baseline (three months after the men had stopped using saunas)
Scrotal temperature was measured with an infrared thermometer before and immediately after each sauna session.
The statistical analysis was basic and compared sperm measures at baseline with the different time points so the effect of the sauna visits on sperm health could be measured.
What were the basic results ?
The average scrotal temperature before the sauna sessions was 34.5°C, which increased to 37.5°C after the session, a statistically significant increase of 3°.
The study found a statistically significant impairment of sperm count and sperm motility both at the end of each sauna session and again at three months 46. No significant difference in sex hormones was found.
Decreases in the percentage of sperm with normal DNA structure and other internal biological sperm structures were also observed after each sauna session and three months after sauna exposure 46.
This corresponded with an increase in the activity of sperm genes associated with coping with heat stress and lack of oxygen, which the researchers believe was brought on by the sauna experience.
The major effects of the sauna were to reduce sperm count and motility 46. Semen volume, sperm structure, and how often the sperm self-destructed did not change throughout the study.
Crucially, the researchers reported that all the effects were reported to be reversed at the six-month time point 46. This suggests the adverse effects of sauna exposure appear to be temporary, at least in men with healthy sperm 46.
How did the researchers interpret the results ?
The researchers concluded that in men with normal sperm, “sauna exposure induces a significant but reversible impairment of spermatogenesis [making sperm], including alteration of sperm parameters, mitochondrial function and sperm DNA packaging” 46.
They went on to suggest that, “the large use of Finnish sauna in Nordic countries and its growing use in other parts of the world makes it important to consider the impact of this lifestyle choice on men’s fertility” 46.
Conclusion
Past research has shown that higher scrotal temperatures in men can lead to poorer sperm health. This proof of concept study tells us that this may also apply to men who use saunas regularly.
The study found that in men with initially healthy sperm, repeated sauna exposure over a three-month period lowered sperm count and reduced sperm motility. Many other aspects of sperm health were unchanged.
Crucially, it also showed that these negative effects were completely reversed at the six-month time point – that is, following a further period of three months without using a sauna.
On its own, this research provides very weak evidence due its tiny sample size – just 10 men were recruited. A study that plucks 10 men from a general population of millions is prone to chance findings and significant bias that may not be applicable to the vast majority of men. For these findings to have more weight, more men would need to be recruited to participate in a larger study.
Other areas that need further investigation include the effects of sauna exposure in men who already have abnormal sperm. These findings may be different (for example, the negative effects may not be as reversible) and this issue was not addressed by this study.
It is also worth pointing out that the majority of British men may not use saunas twice a week for months at a time, so the results are only applicable to the minority that do. Even then, larger studies need to confirm this theory before it can be believed with any certainty.
Overall, this research is consistent with existing fertility advice for men hoping to conceive. Men are advised to keep their testicles cool at around 34.5°C, a few degrees colder than the rest of the body. This helps your body produce the best quality sperm.
Is it safe to use a sauna or jacuzzi if I’m pregnant ?
There’s little research on using saunas, jacuzzis, hot tubs and steam rooms during pregnancy. However, it’s advisable to avoid them because of the risks of overheating, dehydration and fainting.
You’re likely to feel warmer than normal during pregnancy. This is caused by hormonal changes and an increase in blood supply to the skin. These hormonal changes can also make pregnant women feel faint.
Overheating
When you use a sauna, jacuzzi, hot tub or steam room, your body is unable to lose heat effectively by sweating. Your body’s core temperature therefore rises.
It’s possible that a significant rise in your core temperature may affect your unborn baby’s development, particularly in the first 12 weeks of pregnancy.
Feeling faint
If you overheat, more blood flows close to your skin to help cool your body by sweating. This means there’s less blood flow to internal organs, such as your brain. If you don’t get enough blood and oxygen to your brain, it can make you feel faint.
When you’re pregnant, the hormonal changes in your body can make you feel faint more often. You therefore may want to avoid situations where you could get too hot, such as sitting in a jacuzzi or steam room.
Water temperature
The Royal College of Obstetricians and Gynaecologists recommends that if you’re exercising in water – for example, at an antenatal class – the temperature of the water shouldn’t be above 32°C (89.6°F).
If you’re using a hydrotherapy pool, the temperature shouldn’t be above 35°C (95°F). Some hot tubs can be as hot as 40°C (104°F), so it’s best to avoid them.
- Karjanoja M, Peltonen J, Peltonen K. Sauna – Made in Finland. Tampere, Finland: Tammi; 1997.[↩]
- Beever R. Far-infrared saunas for treatment of cardiovascular risk factors: Summary of published evidence. Canadian Family Physician. 2009;55(7):691-696. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2718593/[↩][↩][↩][↩][↩]
- The secret of good ‘löyly’. Helamaa E, Aikäs E. Ann Clin Res. 1988; 20(4):224-9. https://www.ncbi.nlm.nih.gov/pubmed/3218892/[↩][↩]
- Human thermoregulation in sauna. Leppäluoto J. Ann Clin Res. 1988; 20(4):240-3. https://www.ncbi.nlm.nih.gov/pubmed/3218894/[↩]
- Kauppinen K. Sauna, shower, and ice water immersion. Physiological responses to brief exposures to heat, cool, and cold. Part III. Body temperatures. Arctic Med Res. 1989;48:75–86. https://www.ncbi.nlm.nih.gov/pubmed/2736003[↩]
- Leppaluoto J, Arjamaa O, Vuolteenaho O, Ruskoaho H. Passive heat exposure leads to delayed increase in plasma levels of atrial natriuretic peptide in humans. J Appl Physiol. 1991;71:716–20. https://www.ncbi.nlm.nih.gov/pubmed/1834624[↩]
- Kauppinen K. Sauna, shower, and ice water immersion. Physiological responses to brief exposures to heat, cool, and cold. Part I. Body fluid balance. Arctic Med Res. 1989;48:55–63. https://www.ncbi.nlm.nih.gov/pubmed/2736001[↩]
- Vuori I. Sauna bather’s circulation. Ann Clin Res. 1988;20:249–56. https://www.ncbi.nlm.nih.gov/pubmed/3218896[↩][↩]
- Eisalo A, Katila M, Kihlberg J, Takkunen J. Determination of cardiac output from blood pressure measurements and by the dye dilution method. Ann Med Intern Fenn. 1995;45:195–201. https://www.ncbi.nlm.nih.gov/pubmed/13444884[↩]
- Luurila OJ. Cardiac arrhythmias, sudden death and the Finnish sauna bath. Adv Cardiol. 1978;25:73–81. https://www.karger.com/Article/Abstract/402007[↩][↩]
- Romo M. Heart-attacks and the sauna. Lancet. 1976;2:809. https://www.ncbi.nlm.nih.gov/pubmed/61485[↩][↩]
- Suhonen O. Sudden coronary death in middle age and characteristics of its victims in Finland. A prospective population study. Acta Med Scand. 1983;214:207–14. https://www.ncbi.nlm.nih.gov/pubmed/6660027[↩]
- Blum N, Blum A. Beneficial effects of sauna bathing for heart failure patients. Experimental & Clinical Cardiology. 2007;12(1):29-32. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2359619/[↩][↩]
- Tei C, Horikiri Y, Park JC, et al. Acute hemodynamic improvement by thermal vasodilation in congestive heart failure. Circulation. 1995;91:2582–90. http://circ.ahajournals.org/content/91/10/2582.long[↩][↩][↩][↩]
- Nguyen Y, Naseer N, Frishman WH. Sauna as a therapeutic option for cardiovascular disease. Cardiol Rev. 2004;12:321–4. https://www.ncbi.nlm.nih.gov/pubmed/15476570[↩]
- Tei C, Tanaka N. Thermal vasodilation as a treatment of congestive heart failure: A novel approach. J Cardiol. 1996;27:29–30. https://www.ncbi.nlm.nih.gov/pubmed/8683432[↩]
- Eisalo A, Luurila OJ. The Finnish sauna and cardiovascular diseases. Ann Clin Res. 1988;20:267–70. https://www.ncbi.nlm.nih.gov/pubmed/3218899[↩][↩]
- Luurila OJ. The sauna and the heart. J Intern Med. 1992;231:319–20. https://www.ncbi.nlm.nih.gov/pubmed/1588253[↩]
- Sohar E, Shoenfeld Y, Shapiro Y, Ohry A, Cabili S. Effects of exposure to Finnish sauna. Isr J Med Sci. 1976;12:1275–82. https://www.ncbi.nlm.nih.gov/pubmed/1017928[↩]
- Ikeda Y, Biro S, Kamogawa Y, et al. Repeated thermal therapy upregulates arterial nitric oxide synthase expression in Syrian golden hamsters. Jpn Circ J. 2001;65:434–8. https://www.jstage.jst.go.jp/article/jcj/65/5/65_5_434/_pdf/-char/en[↩]
- Kirsch KA, Rocker L, von Ameln H, Hrynyschyn K. The cardiac filling pressure following exercise and thermal stress. Yale J Biol Med. 1986;59:257–65. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2590149/[↩]
- Drexler H, Hayoz D, Munzel T, et al. Endothelial function in chronic congestive heart failure. Am J Cardiol. 1992;69:1596–601. https://www.ncbi.nlm.nih.gov/pubmed/1598876[↩]
- Katz SD, Khan T, Zeballos GA, et al. Decreased activity of the L-arginine-nitric oxide metabolic pathway in patients with congestive heart failure. Circulation. 1999;99:2113–7. http://circ.ahajournals.org/content/99/16/2113.long[↩]
- Zhao G, Shen W, Zhang X, Smith CJ, Hintze TH. Loss of nitric oxide production in the coronary circulation after the development of dilated cardiomyopathy: A specific defect in the neural regulation of coronary blood flow. Clin Exp Pharmacol Physiol. 1996;23:715–21. https://www.ncbi.nlm.nih.gov/pubmed/8886496[↩]
- Noris M, Morigi M, Donadelli R, et al. Nitric oxide synthesis by cultured endothelial cells is modulated by flow conditions. Circ Res. 1995;76:536–43. http://circres.ahajournals.org/content/76/4/536.long[↩]
- Nadaud S, Philippe M, Arnal JF, Michel JB, Soubrier F. Sustained increase in aortic endothelial nitric oxide synthase expression in vivo in a model of chronic high blood flow. Circ Res. 1996;79:857–63. http://circres.ahajournals.org/content/79/4/857.long[↩]
- Hirooka Y, Imaizumi T, Tagawa T, et al. Effects of L-arginine on impaired acetylcholine-induced and ischemic vasodilation of the forearm in patients with heart failure. Circulation. 1994;90:658–68. https://www.ncbi.nlm.nih.gov/pubmed/8044935[↩]
- Jeserich M, Pape L, Just H, et al. Effect of long-term angiotensin-converting enzyme inhibition on vascular function in patients with chronic congestive heart failure. Am J Cardiol. 1995;76:1079–82.[↩]
- Nakamura M, Funakoshi N, Arakawa N, Yoshida H, Makita S, Hiramori K. Effect of angiotensin-converting enzyme inhibitors on endothelium-dependent peripheral vasodilation in patients with chronic heart failure. J Am Coll Cardiol. 1994;24:1321–7.[↩]
- Hornig B, Maier V, Drexler H. Physical training improves endothelial function in patients with chronic heart failure. Circulation. 1996;93:210–4.[↩]
- Patel MB, Kaplan IV, Patni RN, et al. Sustained improvement in flow-mediated vasodilation after short-term administration of dobutamine in patients with severe congestive heart failure. Circulation. 1999;99:60–4.[↩]
- Hornig N, Arakawa N, Kohler C, Drexler H. Vitamin C improves endothelial function of conduit arteries in patients with chronic heart failure. Circulation. 1998;97:363–8.[↩][↩][↩][↩]
- Giannetti N, Juneau M, Arsenault A, et al. Sauna-induced myocardial ischemia in patients with coronary artery disease. Am J Med. 1999;107:228–33. https://www.ncbi.nlm.nih.gov/pubmed/10492315[↩][↩]
- Kihara T, Biro S, Ikeda Y, et al. Effects of repeated sauna treatment on ventricular arrhythmias in patients with chronic heart failure. Circ J. 2004;68:1146–51. https://www.jstage.jst.go.jp/article/circj/68/12/68_12_1146/_pdf/-char/en[↩][↩]
- Michalsen A, Ludtke R, Buhring M, Spahn G, Langhorst J, Dobos GJ. Thermal hydrotherapy improves quality of life and hemodynamic function in patients with chronic heart failure. Am Heart J. 2003;146:728–33. https://www.ncbi.nlm.nih.gov/pubmed/14564334[↩][↩]
- Soleil Saunas [advertisement] Just for Canadian Doctors. 2006 Winter;:2.[↩]
- Kihara T, Biro S, Ikeda Y, Fukudone T, Shinsato T, Masuda A, et al. Effects of repeated sauna treatment on ventricular arrhythmias in patents with chronic heart failure. Circ J. 2004;68(12):1146–51. https://www.jstage.jst.go.jp/article/circj/68/12/68_12_1146/_pdf/-char/en[↩][↩][↩][↩][↩][↩]
- Kihara T, Biro S, Imamura M, Yoshifuku S, Takasaki K, Ikeda Y, et al. Repeated sauna treatment improves vascular endothelial and cardiac function in patients with chronic heart failure. J Am Coll Cardiol. 2002;39(5):754–9. http://www.sciencedirect.com/science/article/pii/S0735109701018241[↩][↩][↩][↩][↩]
- Miyamoto H, Kai H, Nakaura H, Osada K, Mizuta Y, Matsumoto A, et al. Safety and efficacy of repeated sauna bathing in patients with chronic systolic heart failure: a preliminary report. J Card Fail. 2005;11(6):432–6. https://www.ncbi.nlm.nih.gov/pubmed/16105634[↩][↩][↩][↩]
- Sugahara Y, Ishii M, Muta H, Egami K, Akagi T, Matsuishi T. Efficacy and safety of thermal vasodilation therapy by sauna in infants with severe congestive heart failure secondary to ventricular septal defect. Am J Cardiol. 2003;92(1):109–13. https://www.ncbi.nlm.nih.gov/pubmed/12842264[↩][↩][↩]
- Masuda A, Miyata M, Kihara T, Minagoe S, Tei C. Repeated sauna therapy reduces urinary 8-epi-prostaglandin F(2alpha) Jpn Heart J. 2004;45(2):297–303. https://www.jstage.jst.go.jp/article/jhj/45/2/45_2_297/_pdf/-char/en[↩]
- Imamura M, Biro S, Kihara T, Yoshifuku S, Takasaki K, Otsuji Y, et al. Repeated thermal therapy improves impaired vascular endothelial function in patients with coronary risk factors. J Am Coll Cardiol. 2001;38(4):1083–8. http://www.sciencedirect.com/science/article/pii/S073510970101467X[↩]
- Biro S, Masuda A, Kihara T, Tei C. Clinical implications of thermal therapy in lifestyle-related disease. Exp Biol Med (Maywood) 2003;228(10):1245–9. https://www.ncbi.nlm.nih.gov/pubmed/14610268[↩]
- Masuda A, Koga Y, Hattanmura M, Minagoe S, Tei C. The effects of repeated thermal therapy for patients with chronic pain. Psychother Psychosom. 2005;74(5):288–94. https://www.karger.com/Article/Abstract/86319[↩]
- Masuda A, Kihara T, Fukudome T, Shinsato T, Minagoe S, Tei C. The effects of repeated thermal therapy for two patients with chronic fatigue syndrome. J Psychosom Res. 2005;58(4):383–7. https://www.ncbi.nlm.nih.gov/pubmed/15992574[↩]
- Garolla A, Torino M, Sartini B, et al. Seminal and molecular evidence that sauna exposure affects human spermatogenesis. Human Reproduction, Volume 28, Issue 4, 1 April 2013, Pages 877–885, https://doi.org/10.1093/humrep/det020 https://academic.oup.com/humrep/article/28/4/877/653255[↩][↩][↩][↩][↩][↩][↩][↩]