Contents
Spleen peliosis
Spleen peliosis also called peliosis of the spleen, splenic peliosis or peliosis lienalis is an extremely rare benign blood vessel condition characterized by multiple, randomly distributed, blood-filled and cyst-like cavities throughout the spleen 1, 2. The size of the lesions may vary from 1 millimetrer to several centimeters 3. Peliosis is a term derived from the Greek pelios, which means “dusky” or “purple,” referring to the color of the liver tissue with peliosis 4, 3. Spleen peliosis has also been reported, either alone 5, 6, 7, 8 or with peliosis hepatis (hepatic peliosis or liver peliosis) 9, 10, 11, 12, 13, 14, 15. Spleen peliosis often presents with spontaneous rupture of the spleen and requires urgent surgical intervention 16), 17, 18, 2, 6, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28. Numerous associations with medications and other conditions have been described in the literature, emphasizing the limited understanding of spleen peliosis 29. However, spleen peliosis may also be recognized as an incidental finding on computed tomography (CT) scans or at autopsy 30, 28, 27, 26, 23, 31, 32, 33.
Spleen peliosis may be idiopathic or associated with infections such as tuberculosis and Bartonella, hematological diseases such as myeloma, myelofibrosis, and Hodgkin’s disease and with the use of androgenic steroids and erythropoetin. Patients with isolated splenic peliosis may be asymptomatic or describe abdominal discomfort. It is often detected as an incidental finding on abdominal imaging or when spontaneous rupture occurs.
Spleen peliosis discovered on imaging should immediately prompt further evaluation. A differential diagnosis will often include malignant, infiltrative, or infectious conditions, as well as post-traumatic splenic cysts. The cysts of spleen peliosis are blood-filled and innumerable in quantity, distinguishing them from post-traumatic fluid-filled cysts of the spleen. Isolated peliosis of the spleen is extremely rare, and is therefore infrequently considered 2, 30, 28, 27. Imaging modalities for further assessing the spleen vary given the reason for testing and the differential diagnosis; they often include ultrasound, CT, positron emission tomography with CT (PET-CT), and magnetic resonance imaging 26, 24, 34. Despite these various imaging modalities, imaging may be inadequate for establishing a clear diagnosis 26, 24. While spleen peliosis often exists without symptoms, atraumatic spleen rupture, requiring urgent surgical intervention is a well-established presentation of spleen peliosis 16), 6, 17, 35, 36, 37, 38, 39, 40, 41, 34, 42.
As there are no clear published guidelines on the management of spleen peliosis, the definitive management of splenic rupture due to peliosis is emergency splenectomy 43, 44, 45, 46. However, prophylactic splenectomy is still debatable if accidental radiological findings were established upon regular check-ups or unrelated complaints 47.
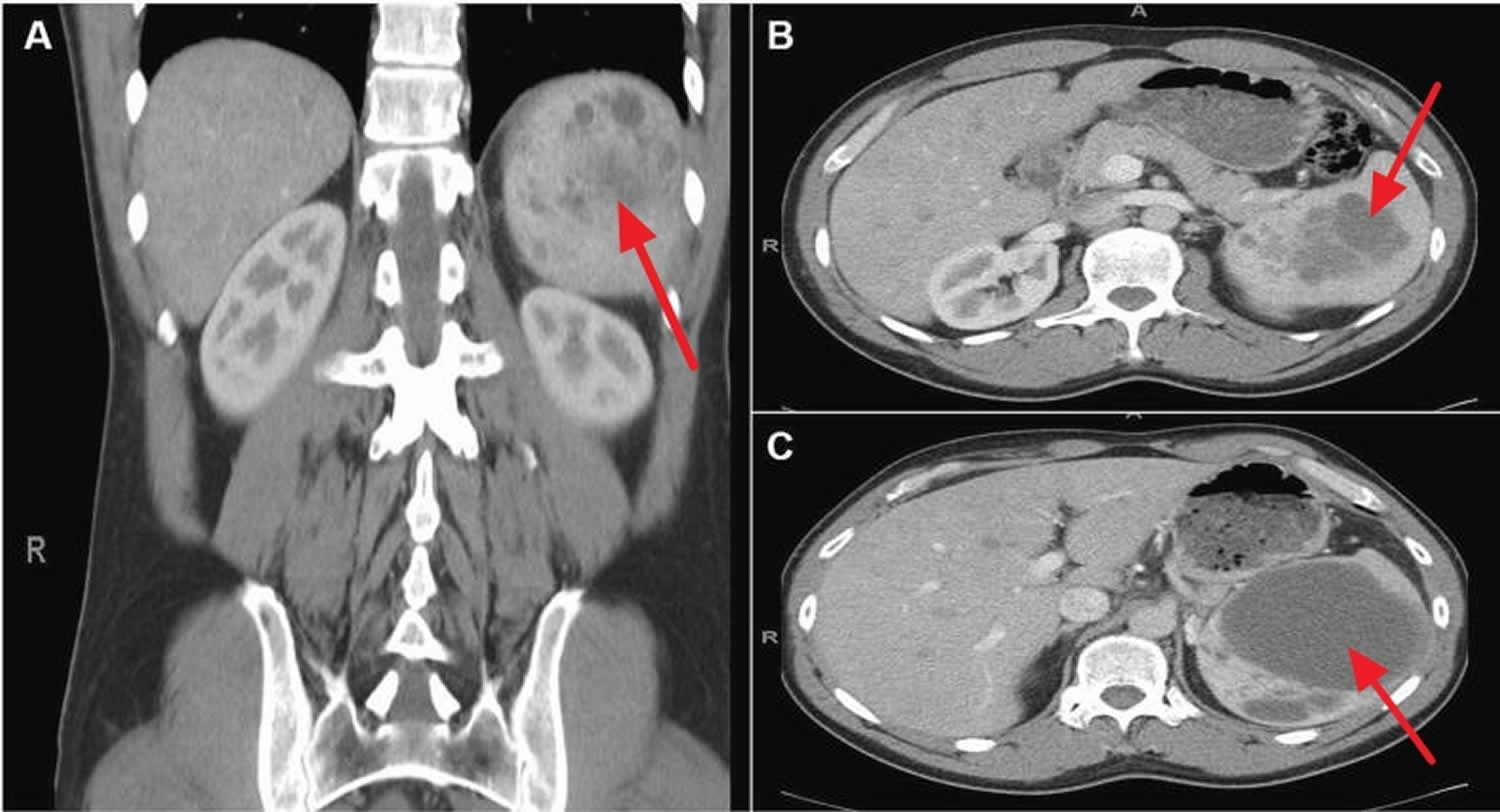
Figure 1. Spleen peliosis
Footnotes: Contrast-enhanced CT scan from a 44-year-old otherwise healthy man presented with left-upper quadrant and left-shoulder pain. The patient denied trauma. A CT scan revealed splenomegaly with multiple splenic cysts and free intraperitoneal fluid consistent with blood. (A) Coronal view, (B) Axial view at the level of L1, (C) Axial view at the level of T12. Significant splenomegaly (14 × 11 × 9 cm). Multiple, loculated, cystic lesions are noted throughout the spleen. The largest cystic lesion measures 9.3 cm in diameter. A minor hemoperitoneum (not visible) was present on the scan.
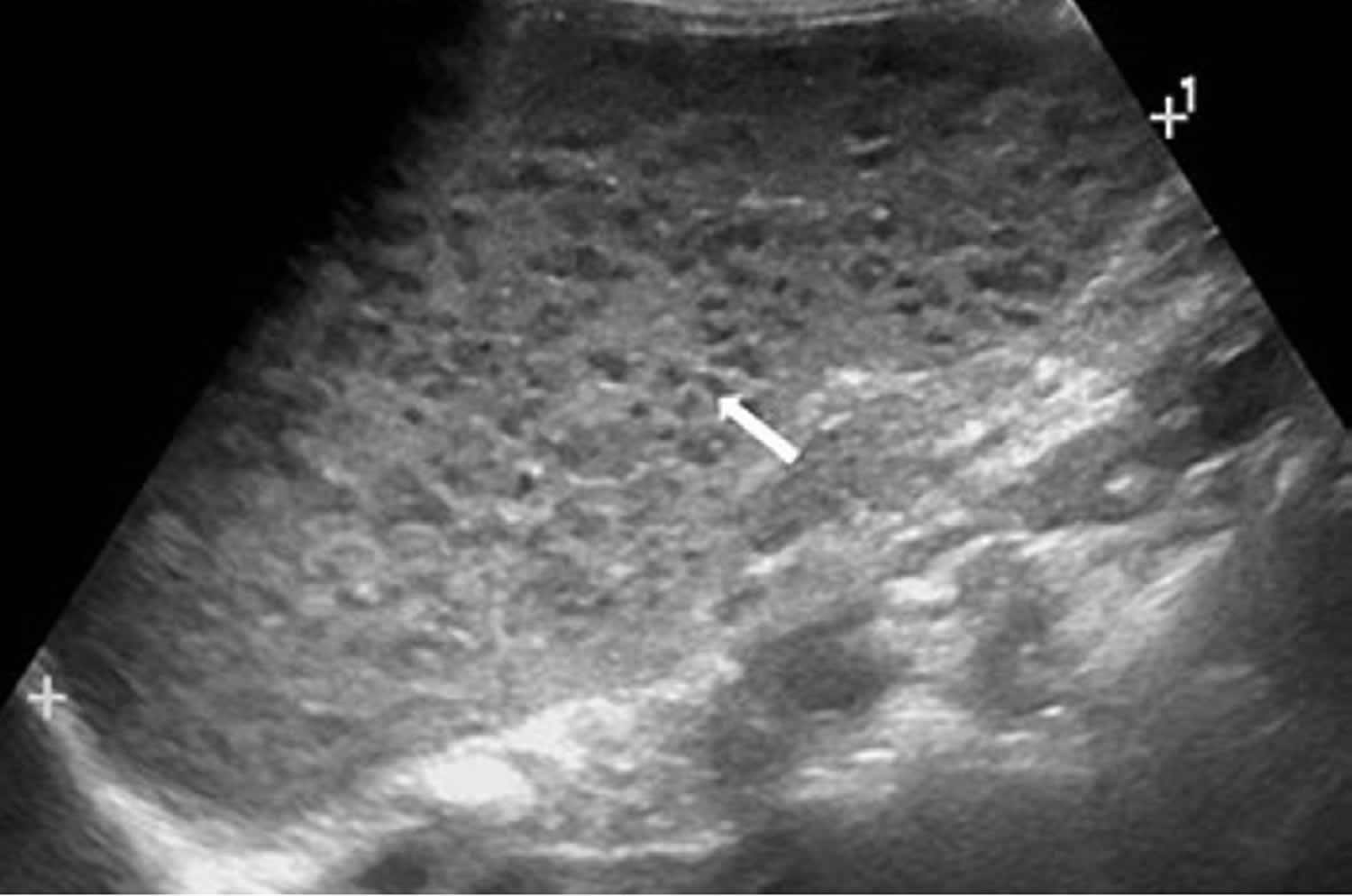
[Source 29 ]Figure 2. Spleen peliosis ultrasound
Footnote: A 3-year-old girl with splenic peliosis. Longitudinal ultrasonography of the spleen shows splenomegaly and multiple poorly defined hypoechoic nodules (arrow).
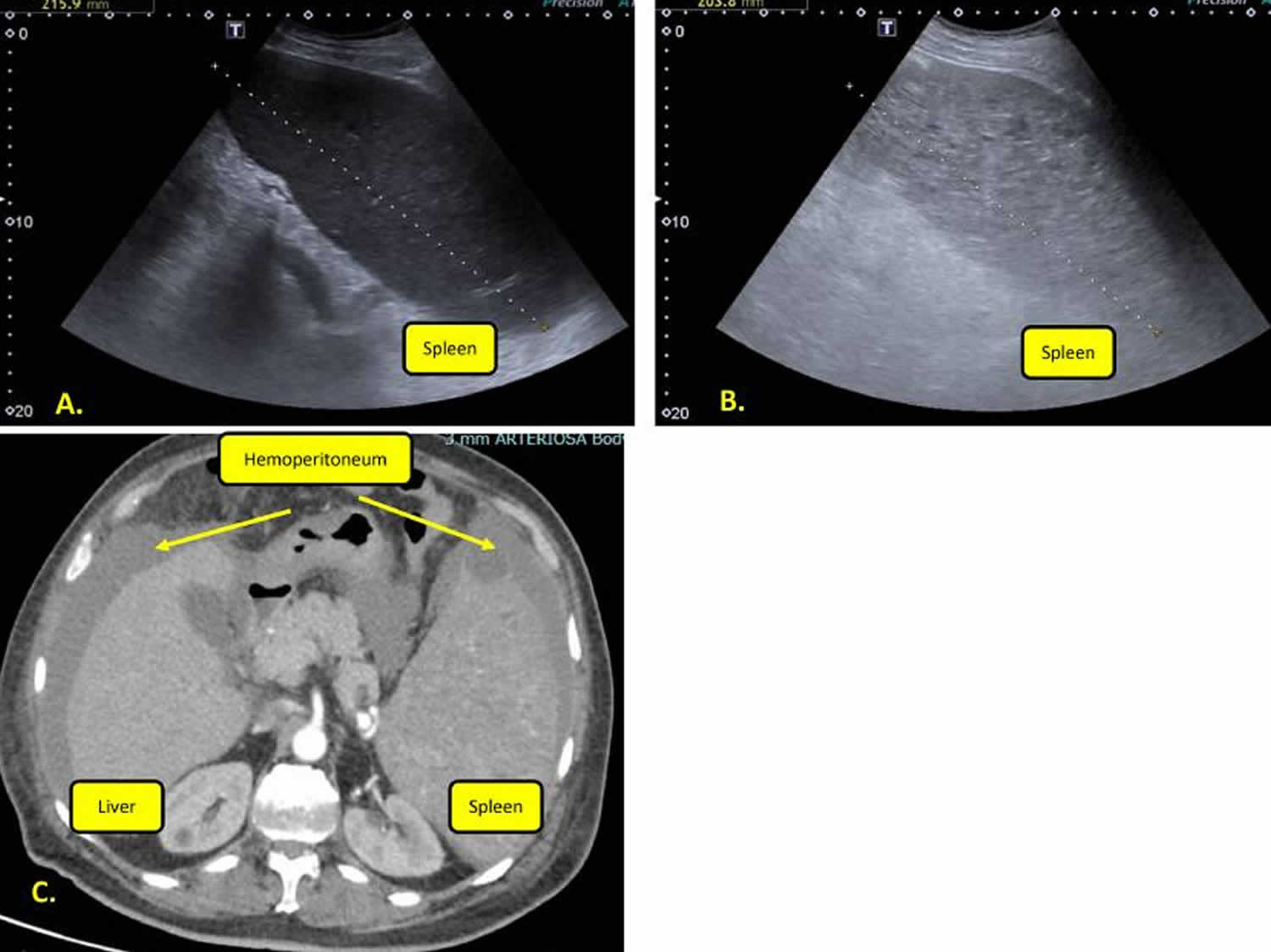
[Source 48 ]Figure 3. Spleen peliosis rupture
Footnotes: (A) Spleen ultrasound appearance at diagnosis. (B) Ultrasound spleen appearance in peliosis rupture. Ultrasonographic imaging showed inhomogeneous splenic parenchyma and increased size. (C) Abdominal computer tomography (CT) frame. CT showed hemoperitoneum and marked splenomegaly (with a pole-to-pole diameter of about 200 mm) with many hyperdense areas.
[Source 49 ]Spleen peliosis cause
The cause of spleen peliosis remains unclear 29. Several causative factors were suggested to be associated with peliosis in the spleen, including the following: intravenous drug abuse, the usage of anabolic steroids, danazol, chronic alcohol use, granulocyte colony-stimulating factor (GSCF) use, tamoxifen, azathioprine, androgens, chemotherapy, liver cirrhosis, tuberculosis, oral contraceptive pills, human immunodeficiency virus (HIV), Hodgkin’s disease, aplastic anemia, and myeloma 45, 46, 50, 27, 26, 25, 24, 23, 22, 21, 20, 51, 52, 53, 36, 37, 38, 39, 40, 41, 34, 54, 55, 56. A number of reported patients were also noted to have blood disorders including multiple myeloma, aplastic anemia, Hodgkin disease, and myelofibrosis 51, 52, 53, 36, 37, 38, 39, 40, 41, 34.
Preliminary evidence links the development of peliosis to angiogenesis (formation of new blood vessels) due to elevated levels of vascular endothelial growth factor (VEGF) 20. VEGF is a fundamental regulator of physiologic angiogenesis. Elevated VEGF levels are known to be associated with tumor progression and poor outcomes in a number of cancers. A condition resembling peliosis has been demonstrated in mice with elevated VEGF levels, that were found to have massively dilated hepatic sinusoids and endothelial cell proliferation and apoptosis 20. The experimental link to elevated VEGF levels, warrants investigation into the treatment of peliosis with anti-angiogenic factors 29.
Spleen peliosis risk factors include:
- use of anabolic steroids
- hematological conditions such as aplastic anemia
- wasting diseases such as
- tuberculosis
- AIDS
- concurrent malignancy
Spleen peliosis symptoms
Patients with spleen peliosis are commonly asymptomatic, and splenic peliosis may present as an incidental finding during imaging studies for other diseases, at autopsy, or with spontaneous rupture of the spleen after minimal or no trauma 29. Although uncommon, some reports suggest that males, ages > 20 years, presence of splenomegaly, and use of chemotherapy might be associated with increased risk of spontaneous splenic rupture in hematology 57. One literature review has shown that all the reported spleen peliosis cases collected from 1980 till 2016 had either presented with abdominal pain or circulatory shock 58. Most cases of spleen peliosis are associated with peliosis hepatis (peliosis of the liver) 20.
Spleen peliosis diagnosis
Spleen peliosis hepatis is very difficult to diagnose because it is usually asymptomatic, and extreme rarity. Peliosis diagnosis is usually obtained after histopathological examination of the resected organ or at autopsy. Radiological imaging shows a broad spectrum of appearances because of its dependence on the blood supply to the lesions. Ultrasound shows multiple poorly defined hypoechoic lesions (Figure 2) and hyperechoic thrombosis 59. The finding of cyst-like hypodense lesions which do not cause mass effect on vessels on contrast-enhanced CT should alert the radiologist to the diagnosis of spleen peliosis. This is of importance in recognizing the risk of spontaneous splenic rupture and in avoiding interventional procedures, such as liver biopsy or percutaneous transhepatic cholangiography, which may precipitate life-threatening iatrogenic organ rupture.
Peliosis histological appearance is characterized by multiple blood-filled cystic cavities primarily involving the mononuclear phagocytic system (liver, spleen, bone marrow, and lymph nodes).
Once the presence of peliosis has been established it is proposed that all necessary investigations be pursued to detect its presence in other organs and to establish a possible cause 60. The implications for this are clear. Spontaneous rupture of the spleen remains an ever-present threat. Physicians should be mindful of this in the event that anticoagulation or thrombolysis is being considered. In addition, the presence of peliosis may prompt the investigation and subsequent discovery of equally serious underlying conditions, for example immunodeficiency or malignancy.
Spleen peliosis treatment
Hemorrhage associated with splenic peliosis is quite amenable to surgical therapy if recognised and treated promptly 61. The existing literature does not however explore the surgical management of patients in whom an incidental diagnosis of splenic peliosis has been made. The introduction of laparoscopic splenectomy has reduced significant morbidity when compared with open surgery 62. Prophylactic laparoscopic splenectomy has been advocated to prevent organ rupture in conditions such as splenic sarcoidosis 63. The role of elective splenectomy in splenic peliosis is not known.
From the patient’s perspective, lifestyle changes may be necessary. Younger patients should be encouraged to avoid high-risk activities, for example, contact sports 60. It would seem wise to avoid or discontinue the oral contraceptive pill, and seek alternative forms of contraception. In addition any attending physician should be aware when other medications thought to cause peliosis are being considered 60.
- Yasmin, A., McKay, P. & Jackson, B. Splenic peliosis: an unusual cause of splenic lesions. Int J Hematol 87, 107 (2008). https://doi.org/10.1007/s12185-008-0024-4[↩]
- Lashbrook DJ, James RW, Phillips AJ, Holbrook AG, Agombar AC. Splenic peliosis with spontaneous splenic rupture: report of two cases. BMC Surg. 2006 Jun 26;6:9. doi: 10.1186/1471-2482-6-9[↩][↩][↩]
- Iannaccone R, Federle MP, Brancatelli G, Matsui O, Fishman EK, Narra VR, Grazioli L, McCarthy SM, Piacentini F, Maruzzelli L, Passariello R, Vilgrain V. Peliosis hepatis: spectrum of imaging findings. AJR Am J Roentgenol. 2006 Jul;187(1):W43-52. https://doi.org/10.2214/AJR.05.0167[↩][↩]
- Wagner E. Ein fall von Blutcysten in der Leber. Arc Heilkunde. 1861;2:369–370.[↩]
- Garcia RL, Khan MK, Berlin RB. Peliosis of the spleen with rupture. Hum Pathol. 1982 Feb;13(2):177-9. doi: 10.1016/s0046-8177(82)80123-8[↩]
- Warfel KA, Ellis GH. Peliosis of the spleen. Report of a case and review of the literature. Arch Pathol Lab Med. 1982 Feb;106(2):99-100.[↩][↩][↩]
- Lacson A, Berman LD, Neiman RS. Peliosis of the spleen. Am J Clin Pathol. 1979 May;71(5):586-90. doi: 10.1093/ajcp/71.5.586[↩]
- Tada T, Wakabayashi T, Kishimoto H. . Peliosis of the spleen . Am J Clin Pathol 1983; 79:708–13.[↩]
- Young JM. Peliosis hepatis; report of two cases. Am Rev Tuberc. 1953 Mar;67(3):385-90. doi: 10.1164/art.1953.67.3.385[↩]
- Kent G, Thompson JR. . Peliosis hepatis: involvement of the reticuloendothelial system . Arch Pathol 1961; 72:658–64.[↩]
- Hamilton FT, Lubitz JM. . Peliosis hepatis: report of three cases, with discussion of pathogenesis . Arch Pathol 1952; 54:564–72.[↩]
- Karasawa T, Shikata T, Smith RD. Peliosis hepatis. Report of nine cases. Acta Pathol Jpn. 1979 May;29(3):457-69. doi: 10.1111/j.1440-1827.1979.tb00201.x[↩]
- Asano S, Wakasa H, Kaise S, Nishimaki T, Kasukawa R. Peliosis hepatis. Report of two autopsy cases with a review of literature. Acta Pathol Jpn. 1982 Sep;32(5):861-77. doi: 10.1111/j.1440-1827.1982.tb03201.x[↩]
- Tada T, Wakabayashi T, Kishimoto H. Peliosis of the spleen. Am J Clin Pathol. 1983 Jun;79(6):708-13. doi: 10.1093/ajcp/79.6.708[↩]
- Ichijima K, Kobashi Y, Yamabe H, Fujii Y, Inoue Y. Peliosis hepatis. An unusual case involving multiple organs. Acta Pathol Jpn. 1980 Jan;30(1):109-20. doi: 10.1111/j.1440-1827.1980.tb01308.x[↩]
- Kumar Anup Anup-Kumar-Trikannad Ashwini, et al. Splenic Peliosis a unique case of spontaneous rupture. Medical and Research Publications. 2021;4(1[↩][↩]
- Podduturi V, Blessing MM. Fatal Hemoperitoneum Due to Isolated Splenic Peliosis. Am J Forensic Med Pathol. 2021 Mar 1;42(1):85-87. doi: 10.1097/PAF.0000000000000596[↩][↩]
- Agha RA, Sohrabi C, Mathew G, Franchi T, Kerwan A, O’Neill N; PROCESS Group. The PROCESS 2020 Guideline: Updating Consensus Preferred Reporting Of CasESeries in Surgery (PROCESS) Guidelines. Int J Surg. 2020 Dec;84:231-235. doi: 10.1016/j.ijsu.2020.11.005[↩]
- Celebrezze JP Jr, Cottrell DJ, Williams GB. Spontaneous splenic rupture due to isolated splenic peliosis. South Med J. 1998 Aug;91(8):763-4. doi: 10.1097/00007611-199808000-00014[↩]
- Joseph F, Younis N, Haydon G, Adams DH, Wynne S, Gillet MB, Maurice YM, Lipton ME, Berstock D, Jones IR. Peliosis of the spleen with massive recurrent haemorrhagic ascites, despite splenectomy, and associated with elevated levels of vascular endothelial growth factor. Eur J Gastroenterol Hepatol. 2004 Nov;16(12):1401-6. doi: 10.1097/00042737-200412000-00027[↩][↩][↩][↩][↩]
- Etzion Y, Benharroch D, Saidel M, Riesenberg K, Gilad J, Schlaeffer F. Atraumatic rupture of the spleen associated with hemophagocytic syndrome and isolated splenic peliosis. Case report. APMIS. 2005 Jul-Aug;113(7-8):555-7. doi: 10.1111/j.1600-0463.2005.apm_165.x[↩][↩]
- Yasmin A, MccKay P, Jackson B. Splenic peliosis: an unusual cause of splenic lesions. Int J Hematol. 2008 Mar;87(2):107. doi: 10.1007/s12185-008-0024-4[↩][↩]
- Engel P, Tjalve E, Horn T. Peliosis of the spleen associated with a paraganglioma. Case report. Acta Radiol. 1993 Mar;34(2):148-9.[↩][↩][↩]
- Tsuda K, Nakamura H, Murakami T, Kozuka T, Yoshii M, Isozaki K, Tsukahara Y, Takami M, Hanada M. Peliosis of the spleen with intraperitoneal hemorrhage. Abdom Imaging. 1993;18(3):283-5. doi: 10.1007/BF00198124[↩][↩][↩][↩]
- Cochrane LB, Freson M. Peliosis of the spleen. Gastrointest Radiol. 1991 Winter;16(1):83-4. doi: 10.1007/BF01887312[↩][↩]
- Maves CK, Caron KH, Bisset GS 3rd, Agarwal R. Splenic and hepatic peliosis: MR findings. AJR Am J Roentgenol. 1992 Jan;158(1):75-6. doi: 10.2214/ajr.158.1.1727362[↩][↩][↩][↩][↩]
- Abo D, Terae S, Fukasawa Y, Seki H, Hasegawa Y, Sakuhara Y, Shimizu T, Shirato H. Splenic peliosis mimicking a solid tumor: findings in multimodalities. J Comput Assist Tomogr. 2008 Nov-Dec;32(6):890-2. doi: 10.1097/RCT.0b013e31815b3ef5[↩][↩][↩][↩]
- Giri N, Pitel PA, Green D, Alter BP. Splenic peliosis and rupture in patients with dyskeratosis congenita on androgens and granulocyte colony-stimulating factor. Br J Haematol. 2007 Sep;138(6):815-7. doi: 10.1111/j.1365-2141.2007.06718.x[↩][↩][↩]
- Chandra A, Navarro SM, Killeen TF, Nemanich G, Harmon JV. Spontaneous splenic rupture due to Peliosis and the association with malignancy: A case series and literature review. Int J Surg Case Rep. 2023 Sep;110:108676. doi: 10.1016/j.ijscr.2023.108676[↩][↩][↩][↩][↩]
- Körner M, Gebbers JO. Peliosis of the spleen and haemolytic anaemia. Histopathology. 2002 Aug;41(2):179-80. doi: 10.1046/j.1365-2559.2002.01424_8.x[↩][↩]
- Adachi K, Ui M, Nojima H, Takada Y, Enatsu K. Isolated splenic peliosis presenting with giant splenomegaly and severe coagulopathy. Am J Surg. 2011 Aug;202(2):e17-9. doi: 10.1016/j.amjsurg.2010.10.002[↩]
- Lal A, Singhal M, Sharma N, Bhalla A, Khandelwal N. Isolated splenic peliosis with spontaneous rupture after a viperine bite. Am J Emerg Med. 2014 Feb;32(2):194.e3-4. doi: 10.1016/j.ajem.2013.09.021[↩]
- Qureshi S, Choong AM, Tadrous PJ, Bhutiani RP. ‘Not just another appendicitis!’ – a case report of acute abdominal pain caused by splenic rupture secondary to isolated splenic peliosis. Ann R Coll Surg Engl. 2009 Nov;91(8):W1-4. doi: 10.1308/147870809X450610[↩]
- Shimono T, Yamaoka T, Nishimura K, Naya M, Hojo M, Yamamoto E, Mukaihara S, Hayakawa K. Peliosis of the spleen: splenic rupture with intraperitoneal hemorrhage. Abdom Imaging. 1998 Mar-Apr;23(2):201-2. doi: 10.1007/s002619900323[↩][↩][↩][↩]
- Rhu J, Cho J. Ruptured splenic peliosis in a patient with no comorbidity: A case report. World J Clin Cases. 2020 Feb 6;8(3):535-539. doi: 10.12998/wjcc.v8.i3.535[↩]
- Stojanović V, Mitić I, Jokić R, Vucković N, Doronjski A, Vijatov G, Milosević B, Djapić M. Splenic peliosis in the course of IgA nephropathy. Pediatr Nephrol. 2007 Dec;22(12):2137-40. doi: 10.1007/s00467-007-0598-8[↩][↩][↩]
- Tsokos M, Püschel K. Isolated peliosis of the spleen: report of 2 autopsy cases. Am J Forensic Med Pathol. 2004 Sep;25(3):251-4. doi: 10.1097/01.paf.0000127401.89952.65[↩][↩][↩]
- Ortega Deballon P, Ferńandez Lobato R, Ortega Muñoz P, Artuñedo Pé P, Ferńandez Luengas D, Moreno Azcoita M. Splenic peliosis: a cause of spontaneous splenic rupture. Surgery. 1999 Sep;126(3):585-6.[↩][↩][↩]
- Raghavan R, Alley S, Tawfik O, Webb P, Forster J, Uhl M. Splenic peliosis: a rare complication following liver transplantation. Dig Dis Sci. 1999 Jun;44(6):1128-31. doi: 10.1023/a:1026663821099[↩][↩][↩]
- Rege JD, Kavishwar VS, Mopkar PS. Peliosis of spleen presenting as splenic rupture with haemoperitoneum–a case report. Indian J Pathol Microbiol. 1998 Oct;41(4):465-7.[↩][↩][↩]
- Gugger M, Gebbers JO. Peliosis of the spleen: an immune-complex disease? Histopathology. 1998 Oct;33(4):387-9. doi: 10.1046/j.1365-2559.1998.00493.x[↩][↩][↩]
- Makdisi WJ, Cherian R, Vanveldhuizen PJ, Talley RL, Stark SP, Dixon AY. Fatal peliosis of the liver and spleen in a patient with agnogenic myeloid metaplasia treated with danazol. Am J Gastroenterol. 1995 Feb;90(2):317-8.[↩]
- Rhu J, Cho J. Ruptured splenic peliosis in a patient with no comorbidity: a case report. World J Clin Cases. 2020;8(3):535–539. doi: 10.12998/wjcc.v8.i3.535[↩]
- Lashbrook DJ, James RW, Phillips AJ, Holbrook AG, Agombar AC. Splenic peliosis with spontaneous splenic rupture: report of two cases. BMC Surg. 2006;6(1):1–7. doi: 10.1186/1471-2482-6-9[↩]
- Davidson J, Tung K. Splenic peliosis: an unusual entity. Br J Radiol. 2010;83(990):e126–e128. doi: 10.1259/bjr/71300465[↩][↩]
- Sharija S. Isolated peliosis lienis—a case report and literature review. J Foren Path. 2017;2:105.[↩][↩]
- Nassr B, Hassan WA, Nassr H, Allouzi A, Al-Shebly M. Isolated splenic peliosis: a case report. J Med Case Rep. 2023 Jun 30;17(1):298. doi: 10.1186/s13256-023-03929[↩]
- Choi G, Kim KA, Lee J, Park YS, Lee J, Choi JW, Lee CH. Ultrasonographic atlas of splenic lesions. Ultrasonography. 2022 Apr;41(2):416-429. doi: 10.14366/usg.21189[↩]
- Tavarozzi R, Borra T, Catania G, Depaoli L, Corsetti MT, Gaidano V, Limberti G, Ravazzoni F, Mariani N, Zallio F, Nozza P, Ladetto M. Spontaneous splenic rupture during induction therapy in acute myeloid leukemia: An unusual case. Radiol Case Rep. 2021 Sep 1;16(11):3406-3409. doi: 10.1016/j.radcr.2021.07.087[↩]
- Tsokos M, Erbersdobler A. Pathology of peliosis. Forensic Sci Int. 2005;149(1):25–33. doi: 10.1016/j.forsciint.2004.05.010[↩]
- Börcek P, Özdemir BH, Yılmaz Akçay E, Haberal M. Splenic Peliosis Resulting in Spontaneous Splenic Rupture in a Concomitant Hepatic and Renal Allograft Recipient. Exp Clin Transplant. 2016 Nov;14(Suppl 3):114-115.[↩][↩]
- Hakoda S, Shinya H, Kiuchi S. Spontaneous splenic rupture caused by splenic peliosis of a hemodialysis patient with chronic renal failure receiving erythropoietin. Am J Emerg Med. 2008 Jan;26(1):109.e1-2. doi: 10.1016/j.ajem.2007.03.025[↩][↩]
- Arai S, Asai T, Uozaki H, Hangaishi A, Kanda Y, Motokura T, Chiba S, Kurokawa M. Splenic peliosis in a patient with aplastic anemia during danazol therapy. Int J Hematol. 2007 Nov;86(4):329-32. doi: 10.1532/IJH97.07078[↩][↩]
- Townsley DM, et al. Danazol treatment for telomere diseases. N. Engl. J. Med. 2016;374:1922–1931. doi: 10.1056/NEJMoa1515319[↩]
- Khincha PP, Wentzensen IM, Giri N, Alter BP, Savage SA. Response to androgen therapy in patients with dyskeratosis congenita. Br. J. Haematol. 2014;165:349–357. doi: 10.1111/bjh.12748[↩]
- Khincha PP, et al. Similar telomere attrition rates in androgen-treated and untreated patients with dyskeratosis congenita. Blood Adv. 2018;2:1243–1249. doi: 10.1182/bloodadvances.2018016964[↩]
- Han JS, Oh SY, Kim SH, Kwon HC, Hong SH, Han JY, Park KJ, Kim HJ. A case of pathologic splenic rupture as the initial manifestation of acute myeloid leukemia M2. Yonsei Med J. 2010 Jan;51(1):138-40. doi: 10.3349/ymj.2010.51.1.138[↩]
- Begum S, Khan MR. Splenic peliosis and rupture—a surgical emergency: case report and review of the available literature. J Appl Hematol. 2016;7:143–147. doi: 10.4103/1658-5127.198508[↩]
- Abbott RM, Levy AD, Aguilera NS, Gorospe L, Thompson WM. From the archives of the AFIP: primary vascular neoplasms of the spleen: radiologic-pathologic correlation. Radiographics. 2004 Jul-Aug;24(4):1137-63. doi: 10.1148/rg.244045006[↩]
- Lashbrook, D.J., James, R.W., Phillips, A.J. et al. Splenic peliosis with spontaneous splenic rupture: report of two cases. BMC Surg 6, 9 (2006). https://doi.org/10.1186/1471-2482-6-9[↩][↩][↩]
- Kohr RM, Haendiges M, Taube RR. Peliosis of the spleen: a rare cause of spontaneous splenic rupture with surgical implications. Am Surg. 1993 Mar;59(3):197-9.[↩]
- Katkhouda N, Mavor E. Laparoscopic Splenectomy. Surg Clin North Am. 2000;80:1285–97. doi: 10.1016/S0039-6109(05)70225-5[↩]
- Zia H, Zemon H, Brody F. Laparoscopic splenectomy for isolated sarcoidosis of the spleen. J Laparoendosc Adv Surg Tech A. 2005;15:160–2. doi: 10.1089/lap.2005.15.160[↩]