Contents
- Vitamin C
- What Does Vitamin C Do?
- How much vitamin C do you need?
- What foods provide vitamin C?
- Vitamin C Supplements
- Liposomal Vitamin C
- Vitamin C Health Benefits
- Vitamin C and Cancer Prevention
- Vitamin C and Cancer Treatment
- High-Dose Intravenous Vitamin C for Cancer Treatment
- Human Clinical Studies of High Dose Intravenous Vitamin C in Cancer Treatment
- Adverse Effects of High Dose Intravenous Vitamin C
- High Dose Intravenous Vitamin C Drug Interactions
- Cardiovascular disease
- Age-related macular degeneration (AMD) and cataracts
- Common cold
- Asthma
- Vitamin C and Skin Health
- Lead toxicity
- Type 2 diabetes mellitus
- Gout
- Adverse pregnancy outcomes
- Alzheimer’s disease
- Sepsis
- Vitamin C Deficiency
- Vitamin C Overdose – Effects of too much Vitamin C
Vitamin C
Vitamin C also known as ascorbic acid, L-ascorbic acid or ascorbate, is an essential, water-soluble vitamin that is naturally present in some foods, added to others and available as a dietary supplement. Vitamin C is synthesized from D-glucose or D-galactose by many plants and animals. However, humans lack the enzyme L-gulonolactone oxidase required for ascorbic acid synthesis and must obtain vitamin C through food or supplements 1, 2. Vitamin C is found in many fruits and vegetables, including citrus fruits, tomatoes, potatoes, red and green peppers, kiwifruit, broccoli, strawberries, brussels sprouts, and cantaloupe. In the body, vitamin C acts as an antioxidant, helping to protect cells from the damage caused by free radicals. Free radicals are compounds formed when our bodies convert the food you eat into energy. People are also exposed to free radicals in the environment from cigarette smoke, air pollution, and ultraviolet light from the sun.
The Recommended Dietary Allowance (RDA; average daily level of intake sufficient to meet the nutrient requirement of 97–98% healthy individuals) for vitamin C ranges from 15 to 115 mg for infants and children (depending on age) and from 75 to 120 mg for nonsmoking adults; people who smoke need 35 mg more per day 3. The intestinal absorption of vitamin C is regulated by at least one specific dose-dependent, active transporter 4. Cells accumulate vitamin C via a second specific transport protein. In vitro studies have found that oxidized vitamin C, or dehydroascorbic acid, enters cells via some facilitated glucose transporters and is then reduced internally to ascorbic acid. The physiologic importance of dehydroascorbic acid uptake and its contribution to overall vitamin C economy is unknown 5.
Fruits and vegetables are the best sources of vitamin C. You can get the recommended amounts of vitamin C by eating a variety of foods including the following:
- Citrus fruits (such as oranges and grapefruit) and their juices, as well as red and green pepper and kiwifruit, which have a lot of vitamin C.
- Other fruits and vegetables—such as broccoli, strawberries, cantaloupe, baked potatoes, and tomatoes—which also have vitamin C.
- Some foods and beverages that are fortified with vitamin C. To find out if vitamin C has been added to a food product, check the product labels.
The vitamin C content of food may be reduced by prolonged storage and by cooking because ascorbic acid is water soluble and is destroyed by heat 3, 6. Steaming or microwaving may lessen cooking losses. Fortunately, many of the best food sources of vitamin C, such as fruits and vegetables, are usually consumed raw. Consuming five varied servings of fruits and vegetables a day can provide more than 200 mg of vitamin C.
Vitamin C plays a role in collagen, carnitine, hormone, and amino acid formation. It is essential for wound healing and facilitates recovery from burns. Vitamin C is also an antioxidant, supports immune function, and facilitates the absorption of iron 7. Vitamin C also plays an important role in both innate and adaptive immunity, probably because of its antioxidant effects, antimicrobial and antiviral actions, and effects on immune system modulators 8. Vitamin C helps maintain epithelial integrity, enhance the differentiation and proliferation of B cells and T cells, enhance phagocytosis, normalize cytokine production, and decrease histamine levels 9. Vitamin C might also inhibit viral replication 10.
Vitamin C deficiency impairs immune function and increases susceptibility to infections 9. Some research suggests that supplemental vitamin C enhances immune function 11, but its effects might vary depending on an individual’s vitamin C status 12.
Vitamin C deficiency is uncommon in the United States, affecting only about 7% of individuals aged 6 years and older 13. People who smoke and those whose diets include a limited variety of foods (such as some older adults and people with alcohol or drug use disorders) are more likely than others to obtain insufficient amounts of vitamin C 11.
Vitamin C is required for the biosynthesis of collagen, L-carnitine, and certain neurotransmitters; vitamin C is also involved in protein metabolism 14. Collagen is an essential component of connective tissue, which plays a vital role in wound healing. Vitamin C is also an important physiological antioxidant 15 and has been shown to regenerate other antioxidants within the body, including vitamin E (alpha-tocopherol) 16. Ongoing research is examining whether vitamin C, by limiting the damaging effects of free radicals through its antioxidant activity, might help prevent or delay the development of certain cancers, cardiovascular disease, and other diseases in which oxidative stress plays a causal role. In addition to its biosynthetic and antioxidant functions, vitamin C plays an important role in immune function 16 and improves the absorption of nonheme iron 17, the form of iron present in plant-based foods. Insufficient vitamin C intake causes scurvy, which is characterized by fatigue or lassitude, widespread connective tissue weakness, and capillary fragility 6.
The intestinal absorption of vitamin C is regulated by at least one specific dose-dependent, active transporter 18. Cells accumulate vitamin C via a second specific transport protein. In vitro studies have found that oxidized vitamin C, or dehydroascorbic acid, enters cells via some facilitated glucose transporters and is then reduced internally to ascorbic acid. The physiologic importance of dehydroascorbic acid uptake and its contribution to overall vitamin C economy is unknown.
Oral vitamin C produces tissue and plasma concentrations that the body tightly controls. Approximately 70%–90% of vitamin C is absorbed at moderate intakes of 30–180 mg/day. However, at doses above 1 g/day, absorption falls to less than 50% and absorbed, unmetabolized ascorbic acid is excreted in the urine 16. Results from pharmacokinetic studies indicate that oral doses of 1.25 g/day ascorbic acid produce mean peak plasma vitamin C concentrations of 135 micromol/L, which are about two times higher than those produced by consuming 200–300 mg/day ascorbic acid from vitamin C-rich foods 19. Pharmacokinetic modeling predicts that even doses as high as 3 g ascorbic acid taken every 4 hours would produce peak plasma concentrations of only 220 micromol/L 19.
The total body content of vitamin C ranges from 300 mg (at near scurvy) to about 2 g 16. High levels of vitamin C (millimolar concentrations) are maintained in cells and tissues, and are highest in leukocytes (white blood cells), eyes, adrenal glands, pituitary gland, and brain. Relatively low levels of vitamin C (micromolar concentrations) are found in extracellular fluids, such as plasma, red blood cells, and saliva 16.
Even before the discovery of vitamin C in 1932, nutrition experts recognized that something in citrus fruits could prevent scurvy, a disease that killed as many as two million sailors between 1500 and 1800 20. Scurvy is a disease caused by a deficiency of vitamin C, characterized by swollen bleeding gums, malaise, lethargy, easy bruising, and spontaneous bleeding and the opening of previously healed wounds 21, which particularly affected poorly nourished sailors until the end of the 18th century 20. High-dose vitamin C has been studied as a treatment for patients with cancer since the 1970s. A Scottish surgeon named Ewan Cameron worked with Nobel Prize-winning chemist Linus Pauling to study the possible benefits of vitamin C therapy in clinical trials of cancer patients in the late 1970s and early 1980’s 22. In the 1970s, Linus Pauling promoted daily megadoses of vitamin C (the amount in 12 to 24 oranges) as a way to prevent colds and some chronic diseases 23. In the mid-20th century, a study hypothesized that cancer may be related to changes in connective tissue, which may be a consequence of vitamin C deficiency 24. A review of evidence published in 1974 suggested that high-dose ascorbic acid may increase host resistance and be a potential cancer therapy 25.
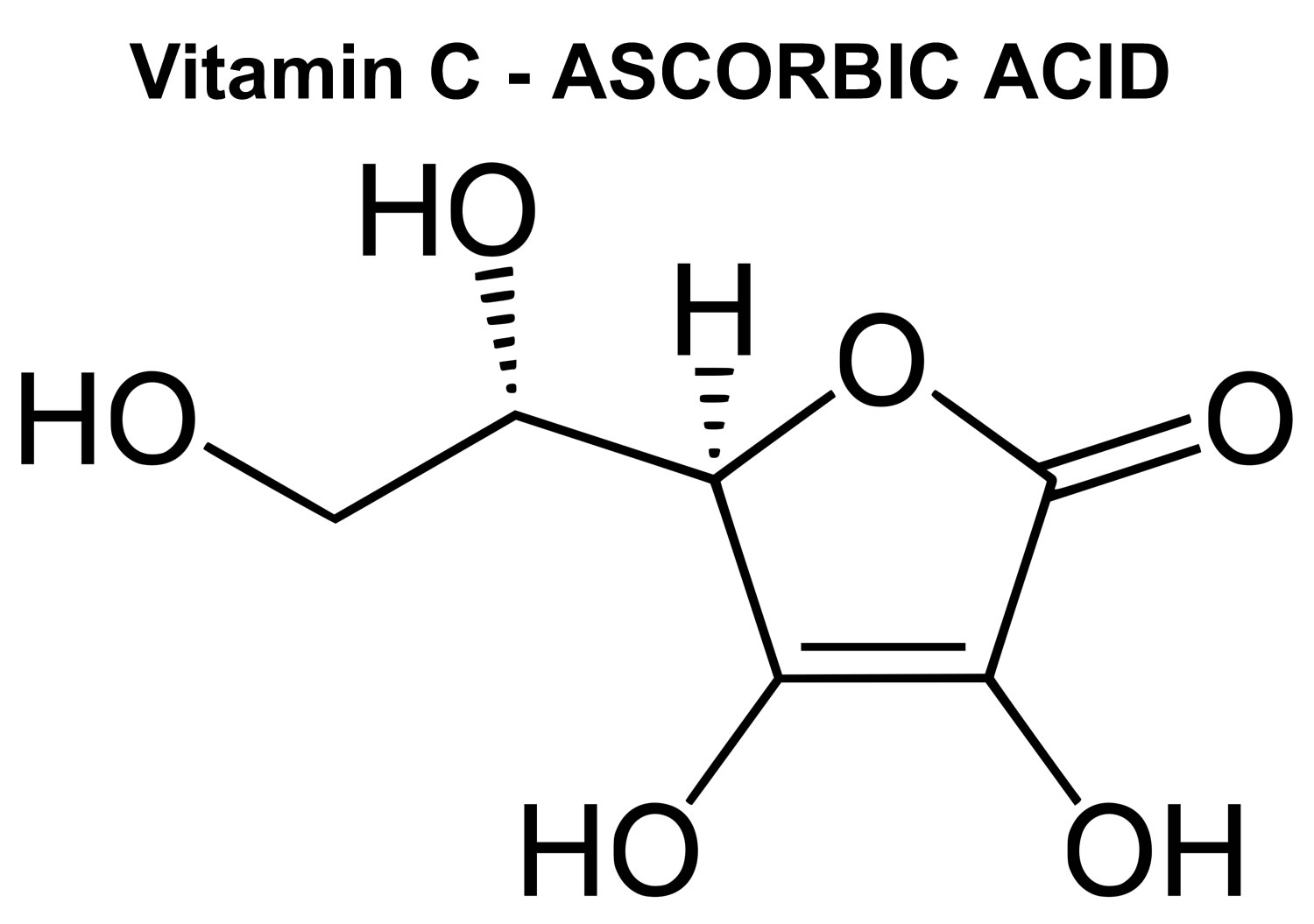
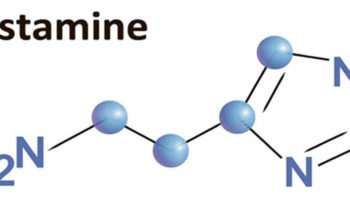
Figure 1. Vitamin C chemical structure
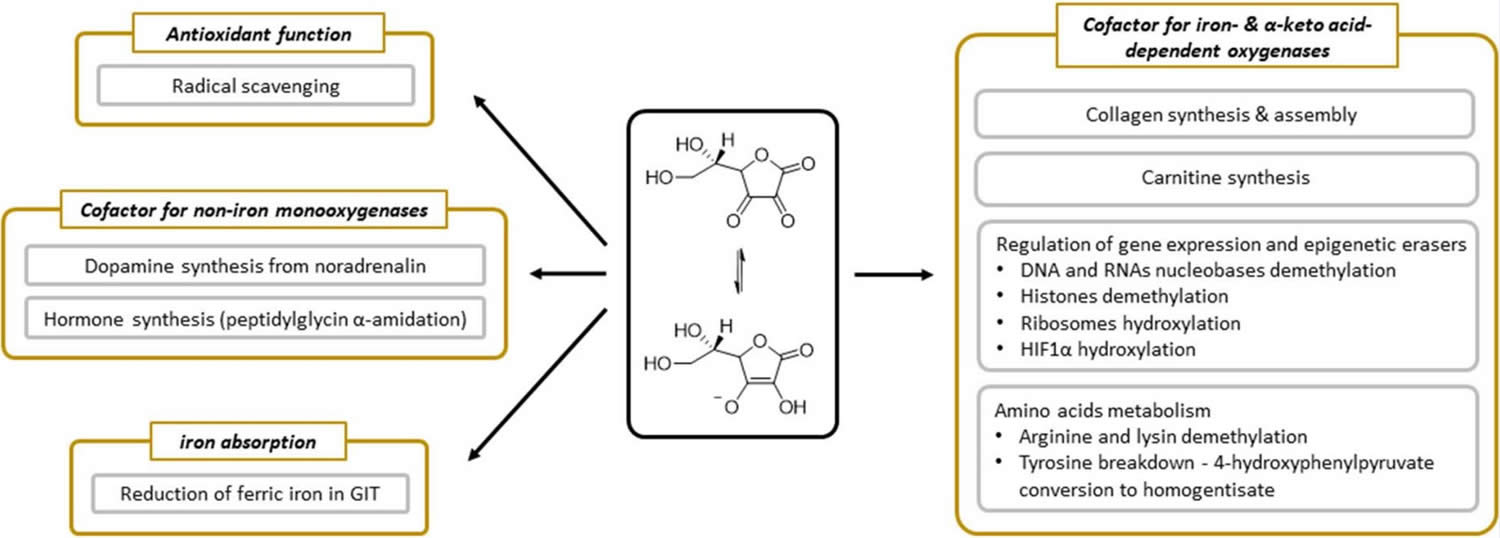
Figure 2. Vitamin C physiological functions
Footnote: Summary of physiological functions of vitamin C.
[Source 26 ]What Does Vitamin C Do?
Vitamin C plays a role in collagen synthesis, synthesis of carnitine, synthesis of hormones (noradrenaline, adrenaline and peptide hormones), gene transcription, and regulation of translation via different mechanisms (hydroxylation of transcription factors, tRNA and ribosomal proteins, demethylation of DNA, and histones), elimination of tyrosine, protection against reactive oxygen species (ROS), and reduction of iron in the gastrointestinal tract 26. Vitamin C is essential for wound healing and facilitates recovery from burns. Vitamin C is also an antioxidant [substance that prevents or reduces damage caused by reactive oxygen species (ROS) or reactive nitrogen species (RNS) by donating electron], supports immune function, and facilitates the absorption of iron 27, 7. Vitamin C is a potent antioxidant (reducing agent), meaning that it readily donates electrons to recipient molecules, which can reduce, and thereby neutralize, reactive oxygen species (ROS) (see Figure 2). Vitamin C also plays an important role in both innate and adaptive immunity, probably because of its antioxidant effects, antimicrobial and antiviral actions, and effects on immune system modulators 8. Vitamin C helps maintain epithelial integrity, enhance the differentiation and proliferation of B cells and T cells, enhance phagocytosis, normalize cytokine production, and decrease histamine levels 9. Vitamin C might also inhibit viral replication 10.
Vitamin C deficiency impairs immune function and increases susceptibility to infections 9. Some research suggests that supplemental vitamin C enhances immune function 11, but its effects might vary depending on an individual’s vitamin C status 12.
High-Dose vitamin C, when taken by intravenous (IV) infusion, vitamin C can reach much higher levels in the blood than when it is taken by mouth. Studies suggest that these higher levels of vitamin C may cause the death of cancer cells in the laboratory. Surveys of healthcare practitioners at United States complementary and alternative medicine conferences in recent years have shown that high-dose IV vitamin C is frequently given to patients as a treatment for infections, fatigue, and cancers, including breast cancer 28.
Vitamin C is required for the biosynthesis of collagen, L-carnitine, and certain neurotransmitters; vitamin C is also involved in protein metabolism 29, 30. Collagen is an essential component of connective tissue, which plays a vital role in wound healing. Vitamin C is also an important physiological antioxidant 31 and has been shown to regenerate other antioxidants within the body, including alpha-tocopherol (vitamin E) 18. Ongoing research is examining whether vitamin C, by limiting the damaging effects of free radicals through its antioxidant activity, might help prevent or delay the development of certain cancers, cardiovascular disease, and other diseases in which oxidative stress plays a causal role. In addition to its biosynthetic and antioxidant functions, vitamin C plays an important role in immune function 18 and improves the absorption of nonheme iron 32, the form of iron present in plant-based foods. Insufficient vitamin C intake causes scurvy, which is characterized by fatigue or lassitude, widespread connective tissue weakness, and capillary fragility 29, 30, 18, 33, 34, 3, 35.
Vitamin C is the primary water-soluble, non-enzymatic antioxidant in plasma and tissues. Even in small amounts, vitamin C can protect indispensable molecules in the body, such as proteins, lipids (fats), carbohydrates, and nucleic acids (DNA and RNA), from damage by free radicals and reactive oxygen species (ROS) that are generated during normal metabolism, by active immune cells, and through exposure to toxins and pollutants (e.g., certain chemotherapy drugs and cigarette smoke). Vitamin C also participates in redox recycling of other important antioxidants; for example, vitamin C is known to regenerate vitamin E from its oxidized form.
The role of vitamin C as a cofactor is also related to its redox potential (another term for an oxidation-reduction reaction). A redox reaction is any reaction in which electrons are removed from one molecule or atom and transferred to another molecule or atom. In such a reaction one substance is oxidized (loses electrons) while the other is reduced (gains electrons). By maintaining enzyme-bound metals in their reduced forms, vitamin C assists mixed-function oxidases in the synthesis of several critical biomolecules 36. These enzymes are either monooxygenases or dioxygenases. Symptoms of vitamin C deficiency, such as poor wound healing and lethargy, likely result from the impairment of these vitamin C-dependent enzymatic reactions leading to the insufficient synthesis of collagen, carnitine, and catecholamines. Moreover, several dioxygenases involved in the regulation of gene expression and the maintenance of genome integrity require vitamin C as a cofactor. Indeed, research has recently uncovered the crucial role played by enzymes, such as the Ten-eleven translocation (TET) dioxygenases and Jumonji domain-containing histone demethylases, in the fate of cells and tissues. These enzymes contribute to the epigenetic regulation of gene expression by catalyzing reactions involved in the demethylation of DNA and histones. The capacity of vitamin C to influence the methylation status of DNA and histones in mammalian cells supports a role for the vitamin in health and disease beyond what was previously understood, in particular by safeguarding genome integrity 37.
Numerous in vitro studies (test tube studies) demonstrate vitamin C’s ability to prevent oxidative stress in human cell lines, a process which has also been shown to occur in the human body. Cooke and colleagues 38 measured urinary and serum levels of 8-oxo-2′-deoxyguanosine (8-oxodG) to evaluate oxidative stress. They measured serum and urinary 8-oxodG after the supplementation of 500 mg of vitamin C in both experimental and control subjects over the course of 25 weeks. Vitamin C supplementation began 3 weeks after a baseline of 8-oxodG was established. After the vitamin C washout period, where no vitamin C was supplemented, there was a significant increase in the levels of 8-oxodG in DNA, enforcing its antioxidant effects Cooke M.S., Evans M.D., Podmore I.D., Herbert K.E., Mistry N., Mistry P., Hickenbotham P.T., Hussieni A., Griffiths H.R., Lunec J. Novel repair action of vitamin C upon in vivo oxidative DNA damage. FEBS Lett. 1998;439:363–367. doi: 10.1016/S0014-5793(98)01403-3. These results were negatively correlated, but the authors did not report the experimental or control 8-oxodG levels in DNA Cooke M.S., Evans M.D., Podmore I.D., Herbert K.E., Mistry N., Mistry P., Hickenbotham P.T., Hussieni A., Griffiths H.R., Lunec J. Novel repair action of vitamin C upon in vivo oxidative DNA damage. FEBS Lett. 1998;439:363–367. doi: 10.1016/S0014-5793(98)01403-3. This study was performed with only 30 healthy volunteers, making it difficult to generalize; however, other studies have shown similar results. Fraga et al. 39 illustrated that with a decrease in the intake of vitamin C, there were elevated levels of 8-oxo-dG in human sperm. In another study, 14 healthy human volunteers who had taken vitamin C had a decrease in H2O2 damage in isolated white blood cells 40. However, there was no change in endogenous DNA damage. Brennan et al. 40 had their participants take 1000 mg vitamin C daily for 42 days or 800 mg vitamin E for 42 days. Peripheral blood was taken and treated with 200 micromolar H2O2, 10 micromolar H2O2, or used as a control. They analyzed DNA damage using ELISA after a 3-week and 6-week wash out period. Cells that were treated with 200 micromolar H2O2 showed a significantly decreased DNA oxidative damage when supplementing with vitamin C 40. For vitamin C, the DNA decreased from roughly 78% to 45%. The control did not have hydrogen peroxide added nor did it have vitamins added. The DNA damage was consistent between 10 and 20% 40. Another study examined lung cancer prevention, demonstrating that smokers who supplemented their diet with vitamin C had less oxidative DNA damage than prior to supplementation 41. The researchers obtained results comparing 500 mg slow-release and plain release tablets of vitamin C paired with an average dose of vitamin E (91 mg), and assessed how this protocol changed the levels of endonuclease 3 and formamidopyrimidine DNA glycosylase enzymes, which mediate DNA repair after oxidative damage. The result was that the slow-release tablet prolonged the protective effect of oxidative DNA damage after a 4-week trial 42. Bo and colleagues 43 performed a meta-analysis of the existing literature to assess the impact of dietary vitamin C on esophageal cancer risk. Their meta-analysis included 15 studies, encompassing 7063 controls and 3955 cancer cases. Their results demonstrate that higher dietary vitamin C intake is inversely associated with esophageal cancer risk 43. Similar results were shown with bladder cancer 44, breast cancer 44 and prostate cancer 45. However, a number of meta-analyses demonstrate non-significant results. One meta-analysis of 47 studies found no association between dietary vitamin C intake and colorectal cancer risk 46. These results support the notion that vitamin C may have site-specific effects, inhibiting certain cancers with no impact on others. Generalization of the results of these studies may be difficult due to the number of confounders that limit each study.
Additionally, many studies have evaluated the impact of supplemental vitamin C and cancer prevention. The Iowa Women’s Health Study published by Kushi et al. 47, followed 34,387 eligible women ages 55–69 through questionnaires for four years. They assessed the antioxidant vitamins A, C, and E. Women that consumed more than 10,000 IU/day of vitamin A demonstrated a slight decrease in age-adjusted risk of breast cancer 47. Those who took vitamin C supplements between 500 and 1000 mg/day had a relative risk of 0.79, but those who took over 1000 mg had a relative risk of 0.77 which showed insignificant differences between the two. After following the women who supplemented vitamin C, there was no significant decrease in risk of developing breast cancer and no significant protective factors against breast cancer 47. In a case control study with 261 women with cervical cancer and 498 controls, diet was assessed and analyzed to see if there was change in cancer after the addition of different supplements 48. No correlation was found between vitamin C and cervical cancer risk 48. One case control study assessed vitamin supplementation and risk of oral or pharyngeal cancer risk 49. After controlling for other risk factors such as smoking and alcohol, there was a significant decrease in risk when supplementing vitamin C 49. However, when adjusting for other use of supplements, the only vitamin that was still associated with a decreased risk was vitamin E 49. Additionally, the PROTEUS study 50, which was a case-control study including 1916 patients with prostate cancer matched with 1915 controls, failed to demonstrate any relationship between dietary or supplemental vitamin C and cancer prevention. In summary, the current body of evidence surrounding the supplementation of vitamin C for cancer prevention fails to demonstrate any definite conclusions. Furthermore, if there is a benefit to supplemental vitamin C for cancer prevention, the potential mechanism may occur through a broad variety of pathways, which may or may not include its antioxidant properties. Future case-control or prospective cohort studies should be designed and control for the impact of multiple vitamin supplements in carcinogenic risk.
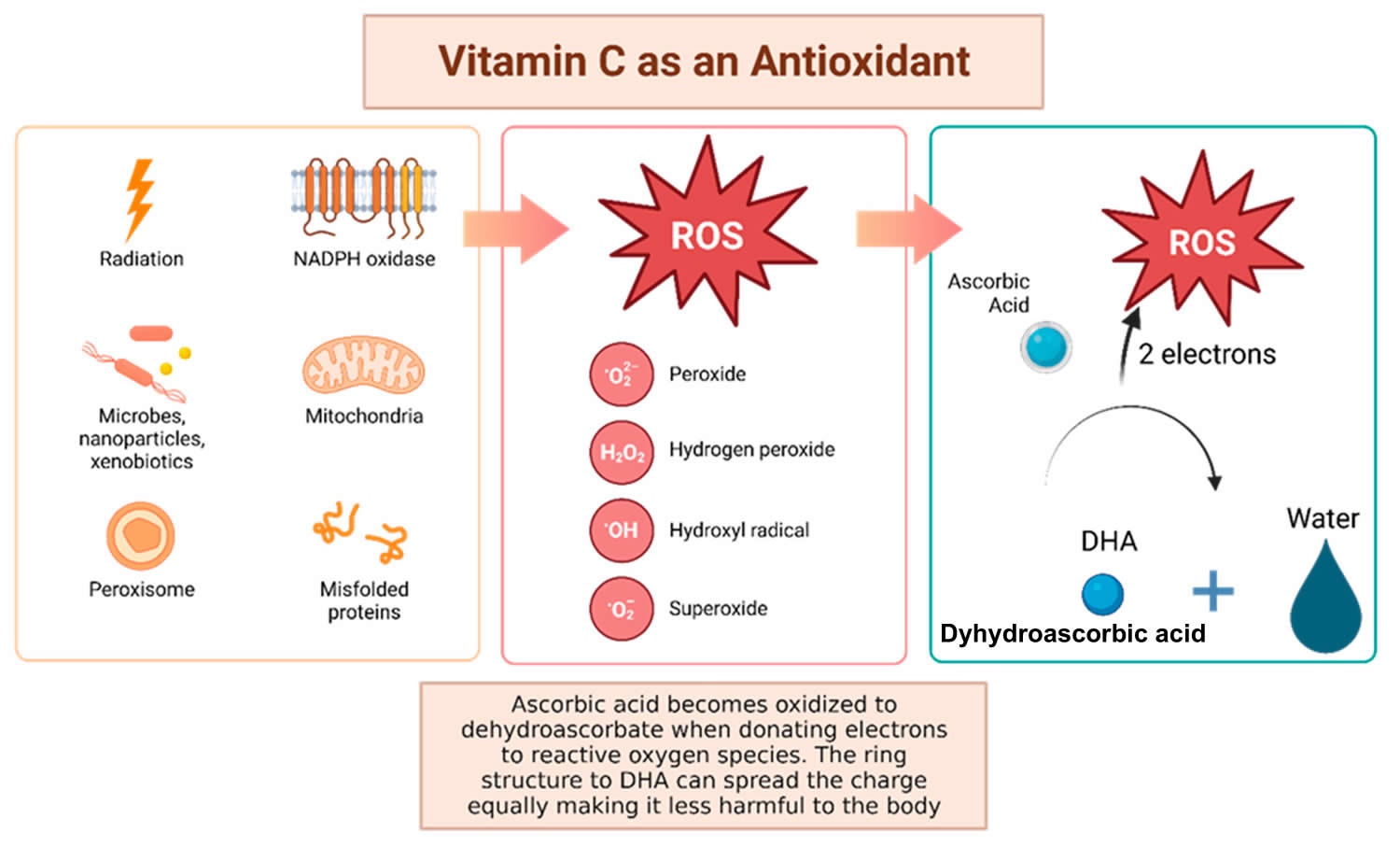
Figure 3. Vitamin C antioxidant properties
Footnotes: Vitamin C has been shown to have antioxidant properties, allowing it to reduce free radicals that may cause harmful damage to DNA. Reactive oxygen species (ROS) may be made by peroxisomes, radiation, the mitochondria, and more biological processes which result in ROS. Vitamin C (ascorbic acid), when ingested, contains electrons that it can give to reactive oxygen species (ROS). These will be reduced to water, and therefore will not be harmful to the body 51. The oxidized version of vitamin C, or dehydroascorbate (DHA), has the ability to even out the positive charge with its ring structure ensuring that it, itself, is not going to damage cells 52.
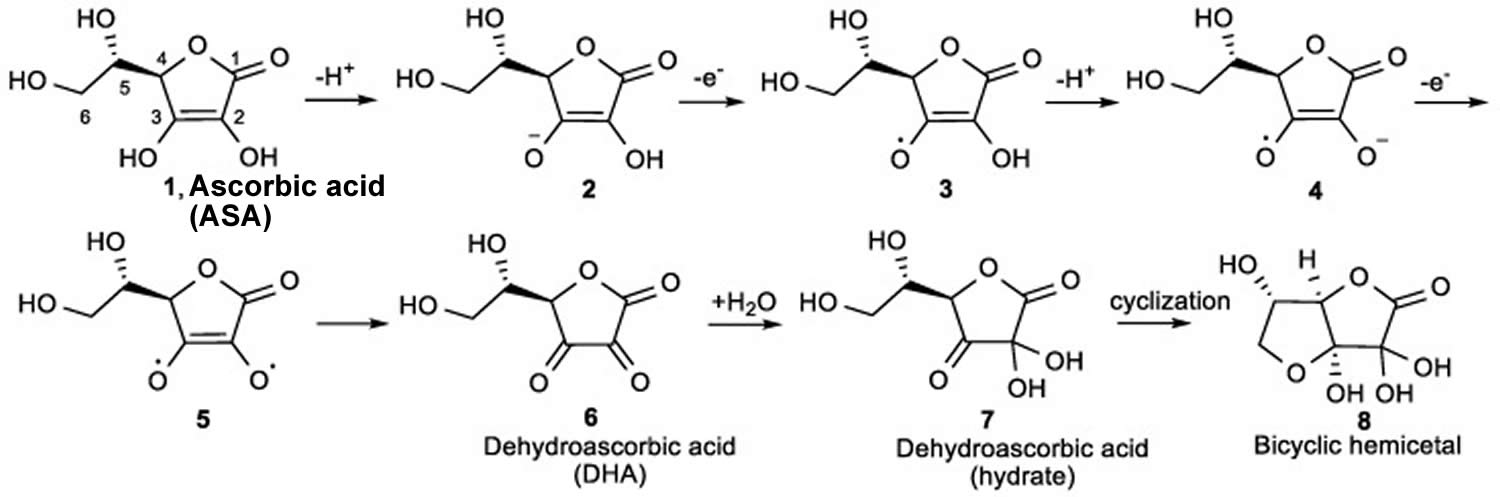
[Source 53 ]Figure 4. Vitamin C Antioxidant Effects
Footnote: The mechanism for the ionization and oxidation of ascorbic acid (ASA) to the inactive dehydroascorbic acid (DHA) and bicyclic hemiacetal.
[Source 54 ]Vitamin C Antioxidant Effects
Vitamin C or ascorbic acid, has hydroxyl groups at a double bond in a lactone ring 53. This allows vitamin C to be a donor of protons and electrons, which is critical in its ability to reduce reactive oxygen species (ROS), including superoxide anions, hydroxyl radicals, and singlet oxygen (Figures 3 and 4) 51. Additionally, vitamin C may prevent cancer by modulating different biological processes. Vitamin C is a critical cofactor for many groups of hydroxylases that are involved in regulating the transcription factor hypoxia-inducible factor 1 (HIF1) 51. Elevated hypoxia-inducible factor (HIF) activity can foster the stem cell phenotype, making the cancer more lethal due to the tumor cell’s ability to rapidly divide and promote poor blood vessel development 53. In order to control HIF and prevent tumor development, HIF hydroxylases must tag the protein for degradation. Vitamin C functions as a cofactor for the HIF hydroxylases; therefore, when cells are deficient in vitamin C acid, HIF hydroxylase activity decreases and HIF transcription activity is increased 51. When HIF levels are high there is increased tumor growth and development, but with the opposing hydroxylases present, HIF can be managed to prevent tumorigenesis 51. Vitamin C is critical for these hydroxylases to function, supporting its possible role in cancer prevention. This has led to the growing research evaluating the addition of vitamin C acid to cancer cells to decrease proliferation 51. While it is possible that the anti-cancer effect of vitamin C may be attributed to its role in modulating hypoxia-inducible factor (HIF) function, there may be multiple pathways by which this effect occurs.
There are a number of studies demonstrating vitamin C’s antioxidant properties. In a study conducted by Lutsenko et al. 55, human kidney 293T cells were treated with 100 micromolar vitamin C and 0.2 micromolar Ci of L-[14C]ascorbic acid for vitamin C uptake or to a mixture of vitamin C and ascorbate oxidase for dehydroascorbic acid uptake. The cells were lysed, and DNA was digested and analyzed for oxidative damage. Cells that were incubated with 100 micromolar of copper and 5 mM H2O2 had significant oxidative damage 55. Cells that were incubated with the copper and H2O2 then with 500 micromolar radiolabeled vitamin C or dehydroascorbic acid showed a decrease in oxidative DNA damage in normal and glutathione depleted cells 55. Overall, when the cells were exposed to the vitamin C, the DNA exhibited less oxidative damage compared to the control 55. This study provides support to vitamin C acting as an antioxidant to prevent oxidative damage, which may reduce carcinogenesis (also called oncogenesis or a pathologic process whereby normal cells are transformed into cancer cells). Leekha et al. 56 tested vitamin C and its anticancer properties with cisplatin chemotherapy on SiHa and HEK293, which are cervical cell lines and control cell lines, respectively. They analyzed the cytotoxicity in cervical cancer cells at varying concentrations of cisplatin and vitamin C separately and combined. Dosing ranged from 5 to 200 micromolar for cisplatin and 25, 50, and 100 μg/mL of vitamin C for 24, 48, and 72 h. The MTT assay used combinations of 100 micromolar cisplatin + 100 μg/mL vitamin C, 50 micromolar cisplatin + 100 μg/mL vitamin C, 5 micromolar cisplatin + 100 μg/mL vitamin C, 1 micromolar cisplatin + 100 μg/mL vitamin C, and 50 micromolar cisplatin + 50 μg/mL vitamin C for time periods 24, 48, and 72 h. There was no significant difference in cytotoxicity across all doses and time periods for the HEK293 cell line, which was the non-tumor control cell line from embryonic kidney stem cells 56. However, there was a significant difference across each time period and varying doses on the SiHa cell lines, which are the cervical cancer cell lines. The combination of vitamin C and cisplatin showed a synergistic amplification in cell death against the cervical cancer cell line SiHa 56. This means that vitamin C is selective for cancer cells and enhanced the killing of tumor cells.
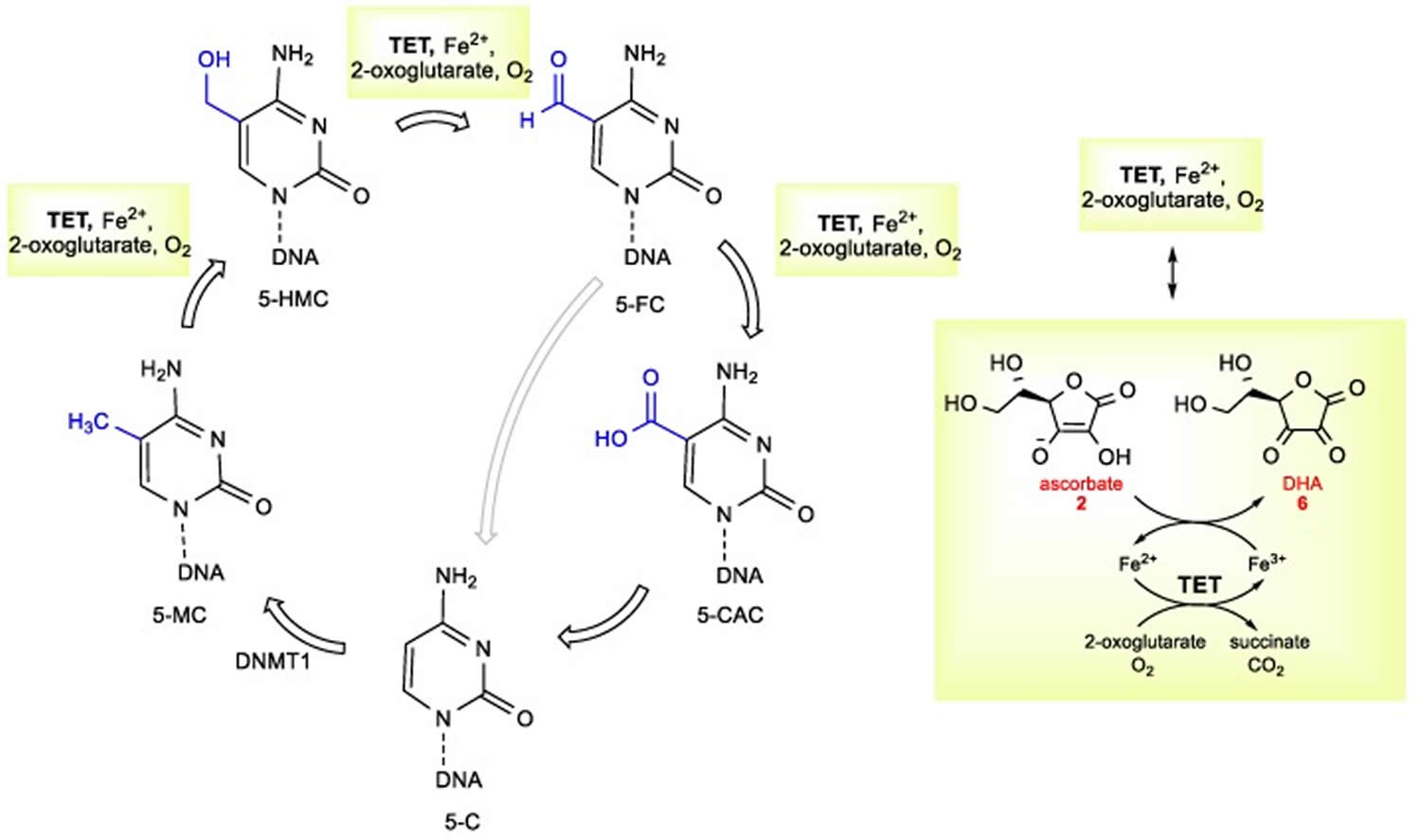
Additionally, vitamin C may indirectly decrease tumorigenesis via its actions as a cofactor for enzymatic reactions. Peng and colleagues 57 evaluated the role of vitamin C in the transition of 5-hydroxymethlcytosine (5hmC) to 5-methylcytosine (5mC), a methylated form of the DNA base cytosine. Loss of 5hmC, which corresponds with increasing DNA methylation, is considered to be an important marker of carcinogenesis 58. Vitamin C acts as a cofactor for Fe-2-oxoglutarate dioxygenases, which include ten-eleven translocation (TET) enzymes 59. TETs reduce DNA methylation by converting 5mC back to 5hMC. Their results demonstrated that vitamin C can increase the content of 5hMC of bladder cancer both in vitro and in vivo, decreasing the malignant phenotype and therefore cancer risk 60. Additionally, ascorbate has been shown to accumulate intracellularly and promote TET activity in hematopoietic stem cells, decreasing leukemogenesis 61. Similar results have been demonstrated in melanoma cells [103]. Further, vitamin C may inhibit tumorigenesis via mitochondrial dysregulation 62. In pancreatic adenocarcinoma cell lines, vitamin C supplementation resulted in decreased cell growth via the inhibition of glucose metabolism without altering the levels of ROS 63. The mechanism by which this occurs is largely unknown but is believed to be related to mitochondrial dysregulation because the addition of pyruvate to the medium rescued cancer cells from death. This suggests that vitamin C supplementation may decrease pyruvate concentrations, suppressing cellular respiration.
Figure 5. Vitamin C in DNA demethylation
Footnote: The mechanistic overview of vitamin C ascorbate in DNA demethylation.
Abbreviations: 5-HMC = 5-hydroxymethylcytosine; 5-MC = 5-methylcytosine; DNMT1 = DNA methyltransferase 1; 5-C = 5-cytosine; 5-CAC = 5-carboxylcytosine; 5-FC = 5-formylcytosine; TET = ten-eleven translocation dioxygenases.
[Source 54 ]Vitamin C Pro-Oxidant and Gene Regulator Properties
Vitamin C may have pro-oxidant functions that lead to cellular damage in vitro 53. The pro-oxidant features of vitamin C are emphasized when it interacts with metals, such as iron and copper. Here, vitamin C will act as a reducing agent and then form oxygen free radicals 52. Interestingly, one mechanism by which vitamin C reduces cancer cell formation may be related to these pro-oxidant capacities 53. Chen and colleagues 64 evaluated whether pharmacologic doses of vitamin C would reduce tumor growth in mice with aggressive glioblastoma, pancreatic, and ovarian tumor xenografts. They discovered that vitamin C supplementation led to an increase in vitamin C radical and hydrogen peroxide formation and a decrease in tumor size across all tumor types by 41–53% 64. This occurred in the interstitial fluid of tumors and not in the blood, suggesting a targeted effect with potentially minimal side effects. Another study evaluated the cytotoxicity of ascorbate, with similar results 65. Ascorbate was shown to induce apoptosis due to the extracellular generation of hydrogen peroxide 65. Given the targeted impact of ascorbate on cancer cells, there is some rationale that this pro-apoptotic effect may occur in newly initiated cancer cells, preventing their proliferation and tumorigenesis.
Vitamin C role in Immunity
Vitamin C affects several components of the human immune system in vitro; for example, vitamin C has been shown to stimulate both the production and function of leukocytes (white blood cells), especially neutrophils, lymphocytes, and phagocytes 66, 67, 68, 69, 70, 71, 72. Specific measures of functions stimulated by vitamin C include cellular motility 73, chemotaxis 66 and phagocytosis 66. Neutrophils, mononuclear phagocytes, and lymphocytes accumulate vitamin C to high concentrations, which can protect these cell types from oxidative damage 74. In response to invading microorganisms, phagocytic leukocytes release non-specific toxins, such as superoxide radicals, hypochlorous acid (“bleach”), and peroxynitrite; these reactive oxygen species kill pathogens and, in the process, can damage the leukocytes themselves 75. Vitamin C, through its antioxidant functions, has been shown to protect leukocytes from self-inflicted oxidative damage 74. Phagocytic leukocytes also produce and release cytokines, including interferons, which have antiviral activity 76. Vitamin C has been shown to increase interferon production in vitro 77. Additional studies have reported that vitamin C enhances the chemotactic and microbial killing capacities of neutrophils and stimulates the proliferation and differentiation of B- and T-lymphocytes 9.
It is widely thought by the general public that vitamin C boosts immune function, yet human studies published to date are conflicting. Disparate results are likely due to study design issues, often linked to a lack of understanding of vitamin C pharmacokinetics and requirements 78.
Finally, vitamin C increases the bioavailability of iron from foods by enhancing intestinal absorption of non-heme iron 79.
Vitamin C and Iron Absorption
The last known function of vitamin C is associated with its potential to reduce ferric ions into ferrous ones 26. In this way, vitamin C increases iron absorption even with a low amount of vitamin C corresponding to its content in a normal diet 80, 81.
Vitamin C as an Enzymatic Cofactor
The enzymatic roles of vitamin C are linked with either dioxygenases (synthesis of collagen and carnitine, involvement in gene transcription, and regulation of translation via different mechanisms and elimination of tyrosine) or monooxygenases (synthesis of hormones) 26. All these vitamin C-dependent oxygenases have a metal, iron, or copper in their active site 26. The involvement of vitamin C in these enzymatic reactions is well documented. The precise mechanism is, however, not fully elucidated, but appears to be related to reduction or maintenance of these metals in the reduced state. In some cases, vitamin C is reduced stoichiometrically within the reaction, suggesting direct involvement 26. However, in others, the stoichiometry is more complicated, implicating rather that vitamin C can recover the enzymatic function if the central metal atom is oxidized. It seems vitamin C to be an ideal cofactor for these enzymes 26. It can be potentially, but not always, replaced by other reductants, which are, however, apparently less active. Individual enzymes and their groups will now be briefly discussed.
The largest group of enzymes using vitamin C as one of their cofactors is the iron-dependent and 2-oxoglutarate-dependent dioxygenases superfamily (2OGO, α-ketoglutarate-dependent hydroxylases). In humans, it represents about 80 enzymes responsible for the modification of important biological substances and processes 82, 83, 84.
The 2-oxoglutarate-dependent dioxygenases superfamily (2OGO, α-ketoglutarate-dependent hydroxylases) were initially identified in the 1970s when studying collagen biosynthesis—a formation of hydroxyproline by collagen prolyl-4-hydroxylase enabling collagen cross-linking and, hence, correct formation of connective tissue 26. Vitamin C is essential to avoid the symptoms of scurvy related to impaired formation of connective tissue. The role(s) of vitamin C in this reaction cycle is (are) not fully identified, but likely the most important role is to maintain the iron atom in the reduced form 85, 86. In any case, vitamin C seems to be irreplaceable for the physiological function of these enzymes. Vvitamin C seems to be oxidized during the process, but the oxidation is not stoichiometric in relation to the reaction. This can support the theory of maintaining the central atom in its ferrous form and/or cause its reduction in the enzymatic process, in particular when the enzymatic reaction becomes uncoupled 87, 82, 86, 88, 89, 90. A simple, non-selective reduction is rather improbable since glutathione, L-cysteine, or dithiothreitol are inactive. Moreover, the intracellular concentration of vitamin C should be apparently relatively high for the optimal function of these enzymes since Km for vitamin C ranges from 140 to 300 µM 89. There are also some reports claiming that vitamin C is not needed, but they were questioned since ferrous ions could, at least, briefly substitute the lack of vitamin C 88. It is possible that there are differences between individual 2-oxoglutarate-dependent dioxygenases superfamily (2OGO) as carnitine synthesis, which does not require vitamin C, according to a complex animal study, and the authors suggested that glutathione can replace it in this case 91.
A relatively new discovery is the finding that vitamin C is the specific cofactor of 2-oxoglutarate-dependent dioxygenases superfamily (2OGO) involved in cellular stress-signalling and epigenetics 83, 86, 92, 93, 94, 95. It comprises hydroxylases involved in the regulation of the classical cellular sensor hypoxia-inducible factor 1α (HIF1 α), e.g., prolyl-hydroxylase domain-containing proteins (PHDs) and factor inhibiting HIF (FIH) 96, 97 and various enzymes involved in epigenetic machinery in humans as epigenetic modification erasers. One of the most known enzyme groups are Jumonji-C (JmjC) domain-containing proteins responsible for hydroxylation of specific histone lysines leading to histone demethylation (JmjC demethylases, e.g., JHDMs and KDMs families) 93, 98, 99, 100. Another 2OGO involved in epigenetic modifications are ten-eleven translocases (TETs) responsible for hydroxylation of 5-methylcytosine to 5-hydroxymethylcytosine primary in DNA 89, 100, 101, 102, 103, 104 and RNA and DNA demethylases from the AlkB family. Generally, 2OGO catalyze specific hydroxylation of substrates, which, in turn, leads to demethylations via other catalytic cycles or downstream pathway involving thymine-DNA-glycosylase, which catalyzed base excision with DNA base excision repair 86, 104.
The last known vitamin C-dependent dioxygenase is 4-hydroxyphenylpyruvate dioxygenase. It is classified with 2OGO into α-keto acid-dependent oxygenases. It has a ferrous ion in the active site and needs oxygen. It catalyzes an uncommon reaction in humans, which involves decarboxylation, substituent migration, and aromatic oxygenation in a single catalytic cycle. The 4-hydroxyphenylpyruvate is converted to homogentisate (2,5-dihydroxyphenylacetate) as a part of the tyrosine elimination pathway. Furthermore, here, the superiority of vitamin C over other reducing agents was shown 105, 106.
How much vitamin C do you need?
The amount of vitamin C you need each day depends on your age. Average daily recommended amounts for different ages are listed below in milligrams (mg). Intake recommendations for vitamin C and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Institute of Medicine 107. DRI is the general term for a set of reference values used for planning and assessing nutrient intakes of healthy people. These values, which vary by age and gender, include:
- Recommended Dietary Allowance (RDA): average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals.
- Adequate Intake (AI): established when evidence is insufficient to develop an RDA and is set at a level assumed to ensure nutritional adequacy.
- Estimated Average Requirement (EAR): Average daily level of intake estimated to meet the requirements of 50% of healthy individuals; usually used to assess the nutrient intakes of groups of people and to plan nutritionally adequate diets for them; can also be used to assess the nutrient intakes of individuals.
- Tolerable Upper Intake Level (UL): maximum daily intake unlikely to cause adverse health effects.
Table 1 lists the current RDAs for vitamin C 107. For infants from birth to 12 months, the Institute of Medicine Food and Nutritional Board established an AI for vitamin C that is equivalent to the mean intake of vitamin C in healthy, breastfed infants.
The amount of vitamin C you need each day depends on your age. Average daily recommended amounts for different ages are listed below in milligrams (mg).
If you smoke, add 35 mg to the above values to calculate your total daily recommended amount.
Vitamin C status is typically assessed by measuring plasma vitamin C levels. Other measures, such as leukocyte vitamin C concentration, could be more accurate indicators of tissue vitamin C levels, but they are more difficult to assess and the results are not always reliable.
Table 1. Vitamin C requirement by age group
| Life Stage | Recommended Amount |
|---|---|
| Birth to 6 months* | 40 mg |
| Infants 7–12 months* | 50 mg |
| Children 1–3 years | 15 mg |
| Children 4–8 years | 25 mg |
| Children 9–13 years | 45 mg |
| Teens 14–18 years (boys) | 75 mg |
| Teens 14–18 years (girls) | 65 mg |
| Adults (men) | 90 mg |
| Adults (women) | 75 mg |
| Pregnant teens | 80 mg |
| Pregnant women | 85 mg |
| Breastfeeding teens | 115 mg |
| Breastfeeding women | 120 mg |
Footnote: If you smoke, add 35 mg to the above values to calculate your total daily recommended amount.
* Adequate Intake (intake at this level is assumed to ensure nutritional adequacy; established when evidence is insufficient to develop an RDA)
[Source 108 ]What foods provide vitamin C?
Fruits and vegetables are the best sources of vitamin C (see Table 2) 5. You can get the recommended amounts of vitamin C by eating a variety of foods including the following:
- Citrus fruits (such as oranges and grapefruit) and their juices, as well as red and green pepper and kiwifruit, which have a lot of vitamin C.
- Other fruits and vegetables—such as broccoli, strawberries, cantaloupe, baked potatoes, and tomatoes—which also have vitamin C.
- Some foods and beverages that are fortified with vitamin C. To find out if vitamin C has been added to a food product, check the product labels.
Citrus fruits, tomatoes and tomato juice, and potatoes are major contributors of vitamin C to the American diet 3. Other good food sources include red and green peppers, kiwifruit, broccoli, strawberries, Brussels sprouts, and cantaloupe (see Table 2) 3. Although vitamin C is not naturally present in grains, it is added to some fortified breakfast cereals.
The vitamin C content of food may be reduced by prolonged storage and by cooking because ascorbic acid is water soluble and is destroyed by heat 3, 6. Steaming or microwaving may lessen cooking losses. Fortunately, many of the best food sources of vitamin C, such as fruits and vegetables, are usually consumed raw. Consuming five varied servings of fruits and vegetables a day can provide more than 200 mg of vitamin C.
The U.S. Department of Agriculture’s (USDA’s) FoodData Central website (https://fdc.nal.usda.gov) lists the nutrient content of many foods and provides a comprehensive list of foods containing vitamin C arranged by nutrient content (https://ods.od.nih.gov/pubs/usdandb/VitaminC-Content.pdf) and by food name (https://ods.od.nih.gov/pubs/usdandb/VitaminC-Food.pdf).
Table 2. Vitamin C content of selected foods
| Food | Milligrams (mg) per serving | Percent (%) DV* |
|---|---|---|
| Red pepper, sweet, raw, ½ cup | 95 | 158 |
| Orange juice, ¾ cup | 93 | 155 |
| Orange, 1 medium | 70 | 117 |
| Grapefruit juice, ¾ cup | 70 | 117 |
| Kiwifruit, 1 medium | 64 | 107 |
| Green pepper, sweet, raw, ½ cup | 60 | 100 |
| Broccoli, cooked, ½ cup | 51 | 85 |
| Strawberries, fresh, sliced, ½ cup | 49 | 82 |
| Brussels sprouts, cooked, ½ cup | 48 | 80 |
| Grapefruit, ½ medium | 39 | 65 |
| Broccoli, raw, ½ cup | 39 | 65 |
| Tomato juice, ¾ cup | 33 | 55 |
| Cantaloupe, ½ cup | 29 | 48 |
| Cabbage, cooked, ½ cup | 28 | 47 |
| Cauliflower, raw, ½ cup | 26 | 43 |
| Potato, baked, 1 medium | 17 | 28 |
| Tomato, raw, 1 medium | 17 | 28 |
| Spinach, cooked, ½ cup | 9 | 15 |
| Green peas, frozen, cooked, ½ cup | 8 | 13 |
Footnote: *DV = Daily Value. The DV (Daily Value) for vitamin C is 90 mg for adults and children age 4 years and older. Foods providing 20% or more of the DV (Daily Value) are considered to be high sources of a nutrient, but foods providing lower percentages of the DV (Daily Value) also contribute to a healthful diet.
[Source 5 ]Are you getting enough vitamin C?
Most people in the United States get enough vitamin C from foods and beverages. However, certain groups of people are more likely than others to have trouble getting enough vitamin C:
- People who smoke and those who are exposed to secondhand smoke, in part because smoke increases the amount of vitamin C that the body needs to repair damage caused by free radicals. People who smoke need 35 mg more vitamin C per day than nonsmokers.
- Infants who are fed evaporated or boiled cow’s milk, because cow’s milk has very little vitamin C and heat can destroy vitamin C. Cow’s milk is not recommended for infants under 1 year of age. Breast milk and infant formula have adequate amounts of vitamin C.
- People who eat a very limited variety of food.
- People with certain medical conditions such as severe malabsorption, some types of cancer, and kidney disease requiring hemodialysis.

Vitamin C Supplements
Supplements typically contain vitamin C in the form of ascorbic acid, which has equivalent bioavailability to that of naturally occurring ascorbic acid in foods, such as orange juice and broccoli 109, 110, 111. Other forms of vitamin C supplements include sodium ascorbate; calcium ascorbate; other mineral ascorbates; ascorbic acid with bioflavonoids; and combination products, such as Ester-C®, which contains calcium ascorbate, dehydroascorbate, calcium threonate, xylonate and lyxonate 112.
A few studies in humans have examined whether bioavailability differs among the various forms of vitamin C. In one study, Ester-C® and ascorbic acid produced the same vitamin C plasma concentrations, but Ester-C® produced significantly higher vitamin C concentrations in leukocytes 24 hours after ingestion 113. Another study found no differences in plasma vitamin C levels or urinary excretion of vitamin C among three different vitamin C sources: ascorbic acid, Ester-C®, and ascorbic acid with bioflavonoids. These findings, coupled with the relatively low cost of ascorbic acid, led the authors to conclude that simple ascorbic acid is the preferred source of supplemental vitamin C 112.
Natural vs. Synthetic vitamin C
Natural and synthetic vitamin C or L-ascorbic acid are chemically identical and there are no known differences in their biological activity. The possibility that the bioavailability of L-ascorbic acid from natural sources might differ from that of synthetic ascorbic acid was investigated in at least two human studies, and no clinically significant differences were observed. A study of 12 males (6 smokers and 6 nonsmokers) found the bioavailability of synthetic ascorbic acid (powder administered in water) to be slightly superior to that of orange juice, based on blood levels of ascorbic acid, and not different based on ascorbic acid in leukocytes (white blood cells) 114. A study in 68 male nonsmokers found that ascorbic acid consumed in cooked broccoli, orange juice, orange slices, and as synthetic ascorbic acid tablets are equally bioavailable, as measured by plasma ascorbic acid levels 115.
Different forms of vitamin C
The gastrointestinal absorption of vitamin C (ascorbic acid) occurs through an active transport process, as well as through passive diffusion. At low gastrointestinal concentrations of ascorbic acid active transport predominates, while at high gastrointestinal concentrations active transport becomes saturated, leaving only passive diffusion. In theory, slowing down the rate of stomach emptying (e.g., by taking vitamin C with food or taking a slow-release form of ascorbic acid) should increase its absorption. While the bioavailability of vitamin C appears equivalent whether it is in the form of powder, chewable tablets, or non-chewable tablets, the bioavailability of ascorbic acid from slow-release preparations is less certain.
A study of three men and one woman found 1 gram of vitamin C (ascorbic acid) to be equally well absorbed from solution, tablets, and chewable tablets, but the absorption from a timed-release capsule was 50% lower. Absorption was assessed by measuring urinary excretion of vitamin C (ascorbic acid) after an intravenous dose of vitamin C (ascorbic acid) and then comparing it to urinary excretion after the oral dosage forms 116.
A more recent study examined the plasma levels of vitamin C (ascorbic acid) in 59 male smokers supplemented for two months with either 500 mg/day of slow-release vitamin C (ascorbic acid), 500 mg/day of plain vitamin C (ascorbic acid), or a placebo. After two months of supplementation no significant differences in plasma vitamin C (ascorbic acid) levels between the slow-release and plain ascorbic acid groups were found 117. A second placebo-controlled trial also evaluated plain vitamin C (ascorbic acid) versus slow-release vitamin C (ascorbic acid) in 48 male smokers 118. Participants were supplemented with either 250 mg plain vitamin C (ascorbic acid), 250 mg slow-release vitamin C (ascorbic acid), or placebo twice daily for four weeks. No differences were observed in the change in plasma ascorbate concentration or area under the curve following ingestion of either formulation.
Mineral ascorbates
Mineral salts of ascorbic acid (mineral ascorbates) are less acidic, and therefore, considered “buffered.” Thus, mineral ascorbates are often recommended to people who experience gastrointestinal problems (upset stomach or diarrhea) with plain vitamin C (ascorbic acid). There appears to be little scientific research to support or refute the claim that mineral ascorbates are less irritating to the gastrointestinal tract. When mineral salts of vitamin C (ascorbic acid) are taken, both the ascorbic acid and the mineral appear to be well absorbed, so it is important to consider the dose of the mineral accompanying the ascorbic acid when taking large doses of mineral ascorbates. For the following discussion, it should be noted that 1 gram (g)= 1,000 milligrams (mg) and 1 milligram (mg) = 1,000 micrograms (μg). Mineral ascorbates are available in the following forms:
- Sodium ascorbate: 1,000 mg of sodium ascorbate generally contains 111 mg of sodium. Individuals following low-sodium diets (e.g., for high blood pressure) are generally advised to keep their total dietary sodium intake to less than 1,500 mg/day. Thus, megadoses of vitamin C in the form of sodium ascorbate could significantly increase sodium intake.
- Calcium ascorbate: Calcium ascorbate generally provides 90-110 mg of calcium (890-910 mg of ascorbic acid) per 1,000 mg of calcium ascorbate. Calcium in this form appears to be reasonably well absorbed. The recommended dietary calcium intake for adults is 1,000 to 1,200 mg/day. Total calcium intake should not exceed the upper level (UL), which is 2,500 mg/day for adults aged 19-50 years and 2,000 mg/day for adults older than 50 years.
The following mineral ascorbates are more likely to be found in combination with other mineral ascorbates, as well as other minerals. It’s a good idea to check the labels of dietary supplements for the ascorbic acid dose as well as the dose of each mineral. Recommended dietary intakes and maximum upper levels of intake (when available) are listed after the individual mineral ascorbates below:
- Potassium ascorbate: The minimal requirement for potassium is thought to be between 1.6 and 2.0 g/day. Fruit and vegetables are rich sources of potassium, and a diet rich in fruit and vegetables may provide as much as 8 to 11 g/day. Acute and potentially fatal potassium toxicity (hyperkalemia) is thought to occur at a daily intake of about 18 g/day of potassium in adults. Individuals taking potassium-sparing diuretics and those with renal insufficiency (kidney failure) should avoid significant intake of potassium ascorbate. The purest form of commercially available potassium ascorbate contains 0.175 grams (175 mg) of potassium per gram of ascorbate.
- Magnesium ascorbate: The recommended dietary allowance (RDA) for magnesium is 400-420 mg/day for adult men and 310-320 mg/day for adult women. The upper level (UL) of intake for magnesium from supplements should not exceed 350 mg/day.
- Zinc ascorbate: The recommended dietary allowance (RDA) for zinc is 11 mg/day for adult men and 8 mg/day for adult women. The upper level (UL) of zinc intake for adults should not exceed 40 mg/day.
Molybdenum ascorbate: The recommended dietary allowance (RDA) for molybdenum is 45 micrograms (μg)/day for adult men and women. The upper level (UL) of molybdenum intake for adults should not exceed 2,000 μg (2 mg)/day. - Chromium ascorbate: The recommended dietary intake (AI) for chromium is 30-35 μg/day for adult men and 20-25 μg/day for adult women. A maximum upper level (UL) of intake has not been determined by the US Food and Nutrition Board.
- Manganese ascorbate: The recommended dietary intake (AI) for manganese is 2.3 mg/day for adult men and 1.8 mg/day for adult women. The upper level (UL) of intake for manganese for adults should not exceed 11 mg/day. Manganese ascorbate is found in some preparations of glucosamine and chondroitin sulfate, and following the recommended dose on the label of such supplements could result in a daily intake exceeding the upper level for manganese.
Vitamin C with bioflavonoids
Bioflavonoids or flavonoids are polyphenolic compounds found in plants. Vitamin C-rich fruit and vegetables, especially citrus fruit, are often rich sources of flavonoids as well. The effect of bioflavonoids on the bioavailability of ascorbic acid has been recently reviewed 119.
Results from the 10 clinical studies comparing the absorption of vitamin C alone or vitamin C in flavonoid-containing foods showed no appreciable differences in bioavailability of ascorbic acid. Only one study, which included five men and three women, found that a 500-mg supplement of synthetic ascorbic acid, given in a natural citrus extract containing bioflavonoids, proteins, and carbohydrates, was more slowly absorbed and 35% more bioavailable than synthetic ascorbic acid alone, when based on plasma levels of ascorbic acid 120. The remaining studies showed either no change or slightly lower plasma ascorbate levels in subjects who consumed vitamin C with flavonoids compared to flavonoids alone 121.
Another assessment of vitamin C bioavailability is measuring urinary ascorbate levels to approximate rates of vitamin C excretion. One study in six young Japanese males (22-26 years old) showed a significant reduction in urinary excretion of ascorbic acid in the presence of acerola juice, a natural source of both vitamin C and flavonoids 122. However, three separate studies showed that urinary levels of vitamin C were increased after consumption of kiwifruit (10), blackcurrant juice (11), or orange juice (1). Overall, the impact of flavonoids on the bioavailability of vitamin C seems to be negligible; however, there is a need for carefully controlled studies using specific flavonoid extracts 121.
Ascorbate and vitamin C metabolites
Ester-C® contains mainly calcium ascorbate, but also contains small amounts of the vitamin C metabolites, dehydroascorbic acid (oxidized ascorbic acid), calcium threonate, and trace levels of xylonate and lyxonate. In their literature, the manufacturers state that the metabolites, especially threonate, increase the bioavailability of the vitamin C in this product, and they indicate that they have performed a study in humans that demonstrates the increased bioavailability of vitamin C in Ester-C®. This study has not been published in a peer-reviewed journal. A small published study of vitamin C bioavailability in eight women and one man found no difference between Ester-C® and commercially available ascorbic acid tablets with respect to the absorption and urinary excretion of vitamin C 123. Ester-C® should not be confused with ascorbyl palmitate, which is also marketed as “vitamin C ester”.
Ascorbyl palmitate
Ascorbyl palmitate is a fat-soluble antioxidant used to increase the shelf life of vegetable oils and potato chips 124. It is an amphipathic molecule, meaning one end is water-soluble and the other end is fat-soluble. This dual solubility allows it to be incorporated into cell membranes. When incorporated into the cell membranes of human red blood cells, ascorbyl palmitate has been found to protect them from oxidative damage and to protect α-tocopherol (a fat-soluble antioxidant) from oxidation by free radicals 125. However, the protective effects of ascorbyl palmitate on cell membranes have only been demonstrated in the test tube. Taking ascorbyl palmitate orally probably doesn’t result in any significant incorporation into cell membranes because most of it appears to be hydrolyzed (broken apart into palmitate and ascorbic acid) in the human digestive tract before it is absorbed. The ascorbic acid released by the hydrolysis of ascorbyl palmitate appears to be as bioavailable as ascorbic acid alone 126. The presence of ascorbyl palmitate in oral supplements contributes to the ascorbic acid content of the supplement and probably helps protect fat-soluble antioxidants in the supplement. The roles of vitamin C in promoting collagen synthesis and as an antioxidant have generated interest in its use on the skin (see the article, Vitamin C and Skin Health). Ascorbyl palmitate is frequently used in topical preparations because it is more stable than some aqueous (water-soluble) forms of vitamin C 127. Ascorbyl palmitate is also marketed as vitamin C ester, which should not be confused with Ester-C®.
D-Isoascorbic acid (Erythorbic acid)
Erythorbic acid is an isomer of ascorbic acid. Isomers are compounds that have the same kinds and numbers of atoms, but different molecular arrangements. The difference in molecular arrangement among isomers may result in different chemical properties. Erythorbic acid is used in the US as an antioxidant food additive and is generally recognized as safe. It has been estimated that more than 200 mg erythorbic acid per capita is introduced daily into the US food system. Unlike ascorbic acid, erythorbic acid does not appear to exert vitamin C activity, for example, it did not prevent scurvy in guinea pigs (one of the few animal species other than humans that does not synthesize ascorbic acid). However, guinea pig studies also indicated that increased erythorbic acid intake reduced the bioavailability of ascorbic acid by up to 50%. In contrast, a series of studies in young women found that up to 1,000 mg/day of erythorbic acid for as long as 40 days was rapidly cleared from the body and had little effect on the bioavailability of ascorbic acid, indicating that erythorbic acid does not diminish the bioavailability of ascorbic acid in humans at nutritionally relevant levels of intake 128.
Other formulations of vitamin C
PureWay-C® is composed of vitamin C and lipid metabolites. Two cell culture studies using PureWay-C® have been published by the same investigators 129, but in vivo data are currently lacking. A small study in healthy adults found that serum levels of vitamin C did not differ when a single oral dose (1 gram) of either PureWay-C® or ascorbic acid was administered 130.
Another formulation of vitamin C, liposomal-encapsulated vitamin C (e.g., Lypo-spheric™ vitamin C) is now commercially available. One report suggested that liposomal-encapsulated vitamin C may be better absorbed than the vitamin in a non-encapsulated form 131.
Large-scale, pharmacokinetic studies are needed to determine how the bioavailability of these vitamin C formulations compares to that of ascorbic acid.
Vitamin C supplement interactions with medications
Vitamin C supplements have the potential to interact with several types of medications. A few examples are provided below. Individuals taking these medications on a regular basis should discuss their vitamin C intakes with their healthcare providers.
Chemotherapy and radiation
The safety and efficacy of the use of vitamin C and other antioxidants during cancer treatment is controversial 132, 133. Some data indicate that antioxidants might protect tumor cells from the action of radiation therapy and chemotherapeutic agents, such as cyclophosphamide, chlorambucil, carmustine, busulfan, thiotepa, and doxorubicin 134, 135. At least some of these data have been criticized because of poor study design 136. Other data suggest that antioxidants might protect normal tissues from chemotherapy- and radiation-induced damage 134 and/or enhance the effectiveness of conventional cancer treatment 137. However, due to the physiologically tight control of vitamin C, it is unclear whether oral vitamin C supplements could alter vitamin C concentrations enough to produce the suggested effects. Individuals undergoing chemotherapy or radiation should consult with their oncologist prior to taking vitamin C or other antioxidant supplements, especially in high doses.
3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (statins)
Vitamin C, in combination with other antioxidants, may attenuate the increase in high-density lipoprotein levels resulting from combination niacin–simvastatin (Zocor®) therapy 138, 139. It is not known whether this interaction occurs with other lipid-altering regimens. Healthcare providers should monitor lipid levels in individuals taking both statins and antioxidant supplements.
Liposomal Vitamin C
Intravenous administration of vitamin C has been shown to decrease oxidative stress and, in some instances, improve physiological function in adult humans. Compared with vitamin C infusion, oral vitamin C administration is typically less effective, in part due to inferior vitamin C bioavailability 140, 141.
Liposomes are manufactured microscopic, hollow spherical vesicles composed of a lipid bilayer. When loaded with pharmaceuticals and/or dietary supplements, liposomes are a very effective method of drug/supplement delivery 142. The majority of those clinically approved have diameters of 50-300 nm. At present, about 600 clinical trials involve lipid particle (liposomes) drug delivery systems. Greater understanding of pharmacokinetics, biodistribution, and disposition of liposomes facilitated particle surface hydration technology (with polyethylene glycol) to reduce rapid clearance and provide sufficient blood circulation time for drug to reach target tissues and cells. Surface hydration enabled the liposome-encapsulated cancer drug doxorubicin (Doxil) to gain clinical approval in 1995. When ingested, the pharmacokinetic properties of liposome intestinal absorption override the usual absorption pattern of the encapsulated drug. That is, the delivery of a drug/supplement with a typically slow or regulated pattern of absorption, such as vitamin C, may be accelerated when encapsulated within a liposome 142, 143.
In a very small study involving 11 adults between the ages of 45–70 years who are overweight or obese as defined by body mass index (>25 kg/m²), to compare the effectiveness between 4 g of unencapsulated vitamin C administered orally with 4 g of liposomal vitamin C (vitamin C encapsulated in liposomes) or intravenous delivery. The findings of this investigation were that the oral delivery of vitamin C encapsulated in liposomes produced circulating concentrations of vitamin C greater than those elicited by oral delivery of unencapsulated vitamin C but less than those following intravenous vitamin C administration 144.
In another study using anticancer drug, epirubicin encapsulated in a liposome with vitamin C. Enhanced epirubicin antitumor activity was a result of the synergistic antineoplastic activity of anthracyclines together with ascorbic acid. The coencapsulation increases its anticancer activity through a possibly synergistic effect previously reported by other groups for a free nonencapsulated drug/vitamin C cocktail 145.
Liposomes have also been intensively investigated as carriers for different applications in dermatology and cosmetics. This research showed ascorbate phosphatidylcholine liposomes formulation could penetrate through the epidermis and allow nonstable hydrophilic active ingredients reach epidermis and dermis preventing skin photodamage 146.
Vitamin C Health Benefits
Due to its function as an antioxidant and its role in immune function, vitamin C has been promoted as a means to help prevent and/or treat numerous health conditions. Scientists are studying vitamin C to understand how it affects health. Here are several examples of what this research has shown.
Vitamin C and Cancer Prevention
Epidemiologic evidence suggests that people with high intakes of vitamin C from fruits and vegetables might have a lower risk of getting many types of cancer, such as lung, breast, and colon cancer, perhaps, in part, due to their high vitamin C content 14, 147, 148, 149, 150. Vitamin C can limit the formation of carcinogens, such as nitrosamines 147, 151, in vivo; modulate immune response 147, 16; and through its antioxidant function, possibly reduce oxidative damage that can lead to cancer 14.
Most case-control studies have found an inverse association between dietary vitamin C intake and cancers of the lung, breast, colon or rectum, stomach, oral cavity, larynx or pharynx, and esophagus 147, 16. Plasma concentrations of vitamin C are also lower in people with cancer than controls 147.
However, randomized, double-blind, placebo-controlled trials that have tested the effect of vitamin C supplementation, alone or in combination with other antioxidant nutrients, on cancer incidence or mortality have shown no effect 152. Furthermore, evidence from prospective cohort studies is inconsistent, possibly due to varying intakes of vitamin C among studies. In a cohort of 82,234 women aged 33–60 years from the Nurses’ Health Study, consumption of an average of 205 mg/day of vitamin C from food (highest quintile of intake) compared with an average of 70 mg/day (lowest quintile of intake) was associated with a 63% lower risk of breast cancer among premenopausal women with a family history of breast cancer 153. Conversely, Kushi and colleagues 154 did not observe a significantly lower risk of breast cancer among postmenopausal women consuming at least 198 mg/day (highest quintile of intake) of vitamin C from food compared with those consuming less than 87 mg/day (lowest quintile of intake). A review by Carr and Frei 147 concluded that in the majority of prospective cohort studies not reporting a significantly lower cancer risk, most participants had relatively high vitamin C intakes, with intakes higher than 86 mg/day in the lowest quintiles. Studies reporting significantly lower cancer risk found these associations in individuals with vitamin C intakes of at least 80–110 mg/day, a range associated with close to vitamin C tissue saturation 147, 155, 156.
Evidence from most randomized clinical trials suggests that vitamin C supplementation, usually in combination with other micronutrients, does not affect cancer risk. In the Supplémentation en Vitamines et Minéraux Antioxydants (SU.VI.MAX) study, a randomized, double-blind, placebo-controlled clinical trial, 13,017 healthy French adults received antioxidant supplementation with 120 mg ascorbic acid, 30 mg vitamin E, 6 mg beta-carotene, 100 mcg selenium, and 20 mg zinc, or placebo 157. After a median follow-up time of 7.5 years, antioxidant supplementation lowered total cancer incidence in men, but not in women. In addition, baseline antioxidant status was related to cancer risk in men, but not in women 158. Supplements of 500 mg/day vitamin C plus 400 IU vitamin E every other day for a mean follow-up period of 8 years failed to reduce the risk of prostate or total cancer compared with placebo in middle-aged and older men participating in the Physicians’ Health Study II 159. Similar findings were reported in women participating in the Women’s Antioxidant Cardiovascular Study 160. Compared with placebo, supplementation with vitamin C 500 mg/day for an average of 9.4 years had no significant effect on total cancer incidence or cancer mortality. In a large intervention trial conducted in Linxian, China, daily supplements of vitamin C (120 mg) plus molybdenum (30 mcg) for 5–6 years did not significantly affect the risk of developing esophageal or gastric cancer 161. Moreover, during 10 years of follow-up, this supplementation regimen failed to significantly affect total morbidity or mortality from esophageal, gastric, or other cancers 162. A 2008 review of vitamin C and other antioxidant supplements for the prevention of gastrointestinal cancers found no convincing evidence that vitamin C (or beta-carotene, vitamin A, or vitamin E) prevents gastrointestinal cancers 163. A similar review by Coulter and colleagues 164 found that vitamin C supplementation, in combination with vitamin E, had no significant effect on death risk due to cancer in healthy individuals.
At this time, the evidence is inconsistent on whether dietary vitamin C intake affects cancer risk. Results from most clinical trials suggest that modest vitamin C supplementation alone or with other nutrients offers no benefit in the prevention of cancer.
A substantial limitation in interpreting many of these studies is that investigators did not measure vitamin C concentrations before or after supplementation. Plasma and tissue concentrations of vitamin C are tightly controlled in humans. At daily intakes of 100 mg or higher, cells appear to be saturated and at intakes of at least 200 mg, plasma concentrations increase only marginally 147, 165, 155, 154, 161. If subjects’ vitamin C levels were already close to saturation at study entry, supplementation would be expected to have made little or no difference on measured outcomes 155, 166, 167, 168.
Vitamin C and Breast cancer prevention
Two large prospective studies found dietary vitamin C intake to be inversely associated with breast cancer incidence in certain subgroups. In the Nurses’ Health Study, premenopausal women with a family history of breast cancer who consumed an average of 205 mg/day of vitamin C from food had a 63% lower risk of breast cancer than those who consumed an average of 70 mg/day 153. In the Swedish Mammography Cohort, overweight women who consumed an average of 110 mg/day of vitamin C had a 39% lower risk of breast cancer compared to overweight women who consumed an average of 31 mg/day 169. More recent prospective cohort studies have reported no association between dietary and/or supplemental vitamin C intake and breast cancer 170, 171, 172.
Vitamin C and Stomach cancer prevention
A number of observational studies have found increased dietary vitamin C intake to be associated with decreased risk of gastric (stomach) cancer, and laboratory experiments indicate that vitamin C inhibits the formation of carcinogenic N-nitroso compounds in the stomach 173, 174, 175. A nested case-control study in the EPIC study found a 45% lower risk of gastric cancer incidence in individuals in the highest (≥51 μmol/L) versus lowest (<29 μmol/L) quartile of plasma vitamin C concentration; no association was observed between dietary vitamin C intake and gastric cancer 176.
Infection with the bacteria, Helicobacter pylori (H. pylori), is known to increase the risk of stomach cancer and is associated with lower vitamin C content of stomach secretions 177, 178. Although two intervention studies failed to show a reduction in stomach cancer incidence with vitamin C supplementation 179, some research suggests that vitamin C supplementation may be a useful addition to standard H. pylori eradication therapy in reducing the risk of gastric cancer 180. Because vitamin C can inactivate urease (an enzyme that facilitates H. pylori survival and colonization of the gastric mucosa at low pH) in vitro, vitamin C may be most effective as a preventive agent in those without achlorhydria (a condition in which the stomach does not produce hydrochloric acid) 181, 182.
Vitamin C and Colon cancer prevention
By pooling data from 13 prospective cohort studies comprising 676,141 participants, it was determined that dietary intake of vitamin C was not associated with colon cancer, while total intake of vitamin C (i.e., from food and supplements) was associated with a 19% reduced risk of colon cancer 183. Each of the cohort studies used self-administered food frequency questionnaires at baseline to assess vitamin C intake. Although the analysis adjusted for several lifestyle and known risk factors, the authors noted that other healthy behaviors and/or folate intake may have confounded the association.
Vitamin C and Non-Hodgkin lymphoma prevention
A population-based, prospective study, the Iowa Women’s Health Study, collected baseline data on diet and supplement use in 35,159 women (aged 55-69 years) and evaluated the risk of developing non-Hodgkin lymphoma (NHL) over 19 years of follow-up 184. Overall, an inverse association between fruit and vegetable intake and risk of non-Hodgkin lymphoma (NHL) was observed. Additionally, dietary, but not supplemental, intake of vitamin C and other antioxidant nutrients (carotenoids, proanthocyanidins, and manganese) was inversely associated with non-Hodgkin lymphoma (NHL) risk. Another large, multi-center, prospective study — the Women’s Health Initiative — that followed 154,363 postmenopausal women for 11 years found that dietary and supplemental vitamin C intake at baseline was inversely associated with diffuse B-cell lymphoma, a subtype of non-Hodgkin lymphoma (NHL) 185.
Vitamin C and other site-specific cancer types prevention
The Physicians’ Health Study 2 was a randomized, placebo-controlled trial that examined the effect of vitamin E (400 IU/day), vitamin C (500 mg/day), and a multivitamin supplement on the risk of cancer in 14,641 middle-aged male physicians over 10.3 years (7.6 years of active treatment plus 2.8 years post-treatment follow-up) 186. Supplementation with vitamin C had no effect on the overall risk of cancer or on the risk of prostate, bladder, or pancreatic cancer; there was a marginal reduction in colorectal cancer incidence with vitamin C compared to placebo 186.
Vitamin C and Cancer Treatment
More than fifty years ago, a study suggested that cancer was a disease of changes in connective tissue caused by a lack of vitamin C. In the 1970’s, it was proposed that high-dose ascorbic acid could help build resistance to disease or infection and possibly treat cancer 187. Several studies of IV vitamin C given alone or in combination with other drugs in people with cancer include the following 188:
- Studies of IV vitamin C alone
- One study found that people with cancer who received IV vitamin C had better quality of life and fewer cancer-related side effects than those who did not receive it.
- In a single-arm pilot study of people with castration-resistant prostate cancer, IV vitamin C did not lower prostate-specific antigen levels or stop tumors from growing.
- In a study of healthy volunteers and people with cancer, vitamin C was shown to be safe at doses up to 1.5 g/kg in people who do not have kidney stones, other kidney diseases, or G6PD deficiency. Studies have also shown that vitamin C levels in the blood are higher when given by IV than when taken by mouth, and last for more than 4 hours.
- Studies of IV vitamin C combined with other drugs have shown mixed results
- In a small study of 14 people with advanced pancreatic cancer, IV vitamin C was given along with chemotherapy and targeted therapy (erlotinib). Five study participants did not complete the vitamin C treatment because the tumor continued to grow during treatment. The nine participants who completed the treatment had stable disease as shown by imaging studies. Very few side effects were reported from the vitamin C treatment.
- In a study of people newly diagnosed with pancreatic cancer, IV vitamin C did not interfere with gemcitabine.
- In another small study, 9 people with advanced pancreatic cancer were given chemotherapy once a week for 3 weeks along with IV vitamin C twice a week for 4 weeks during each treatment cycle. The cancer did not progress over an average of 6 months in these patients. No serious side effects were reported with the combined treatment.
- In a study of 27 people with advanced ovarian cancer, chemotherapy alone was compared with chemotherapy and IV vitamin C. IV vitamin C was given during chemotherapy and for 6 months after chemotherapy ended. Those who received IV vitamin C had fewer side effects from the chemotherapy.
- People with refractory metastatic colorectal cancer or metastatic melanoma were treated with IV vitamin C given along with arsenic trioxide and other drugs. The treatment had no anticancer effect, the tumor continued to grow during treatment, and patients had serious side effects. These studies did not have a comparison group, so it is unclear how much the IV vitamin C affected the side effects.
- In two pilot trials, people with non-small cell lung cancer or glioblastoma multiforme were given standard therapy plus IV vitamin C. Those who received standard therapy plus IV vitamin C had better overall survival and fewer side effects than the control groups.
More studies of combining IV vitamin C with other drugs are being done. These include a number of clinical trials combining IV vitamin C with arsenic trioxide, showing mixed results.
IV vitamin C has caused very few side effects in clinical trials. However, IV vitamin C may be harmful in people with certain risk factors.
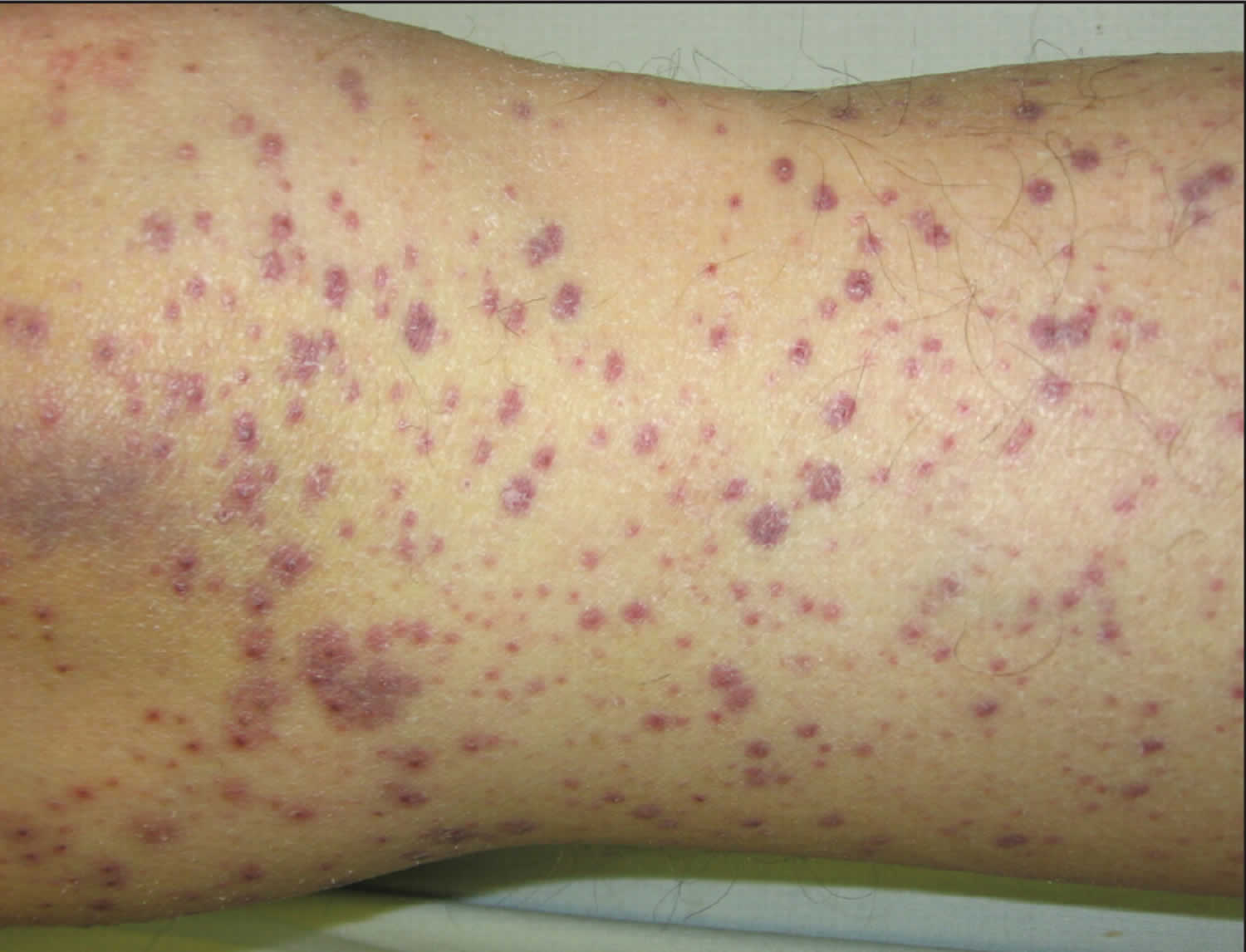
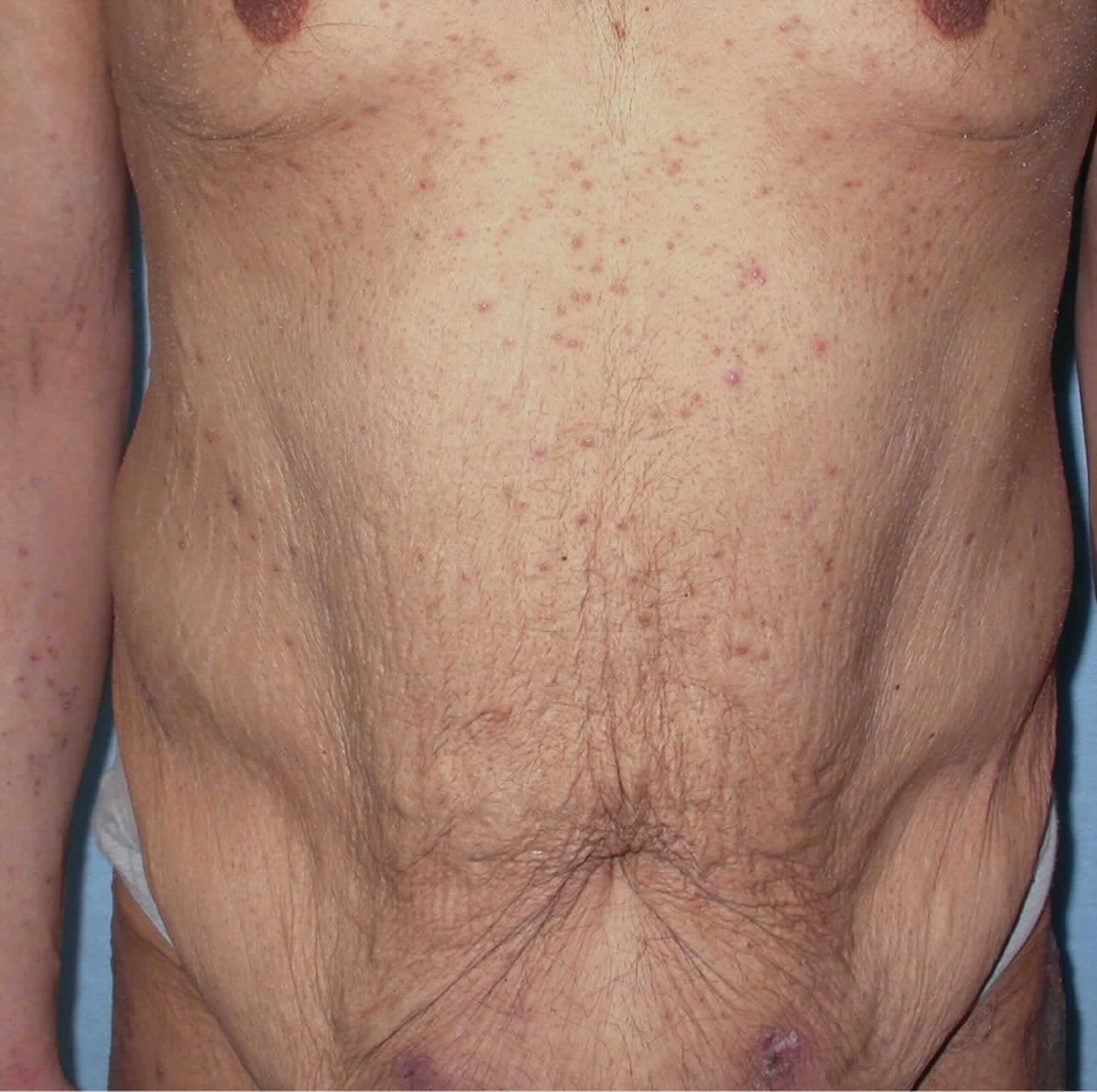
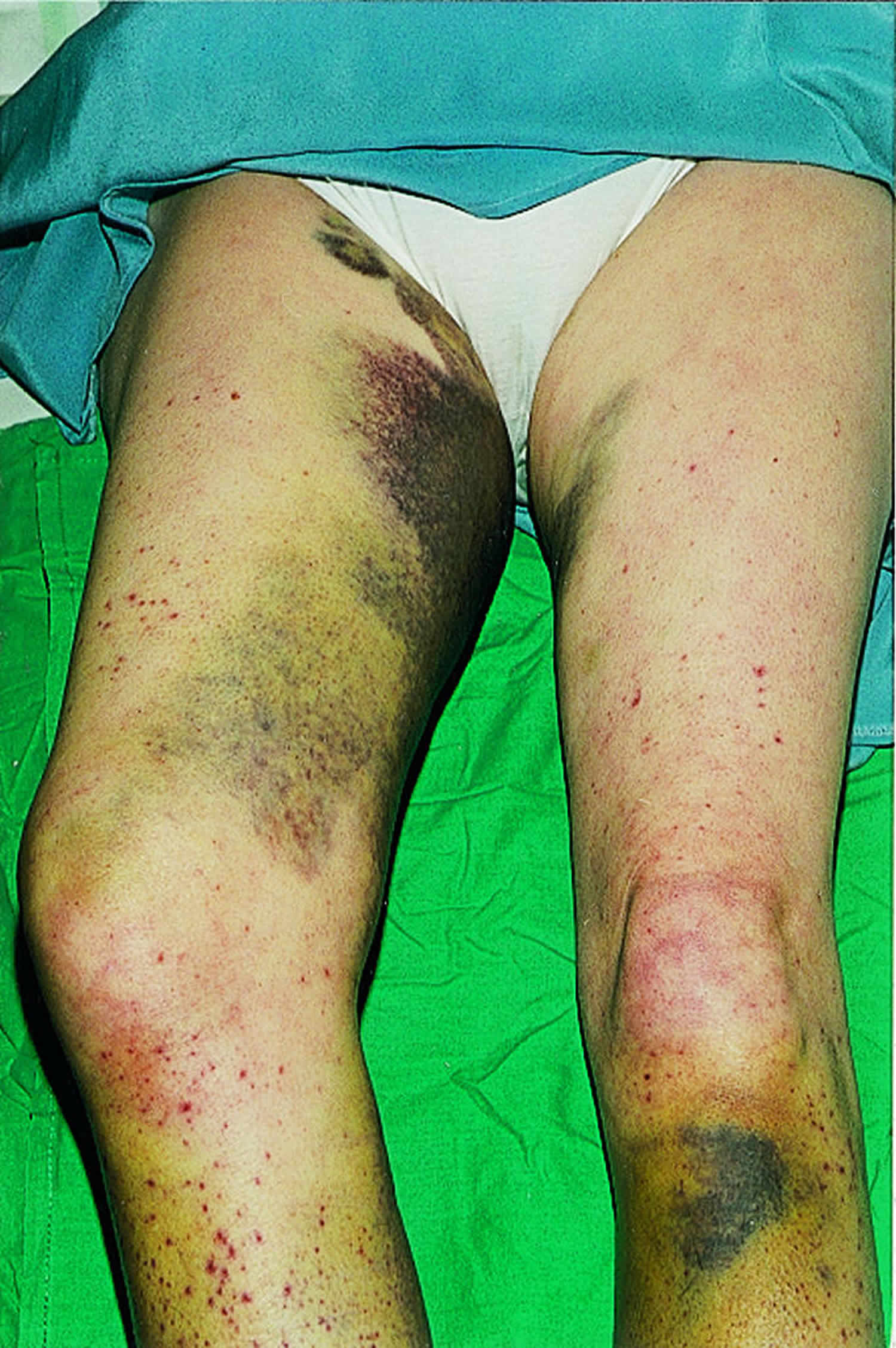
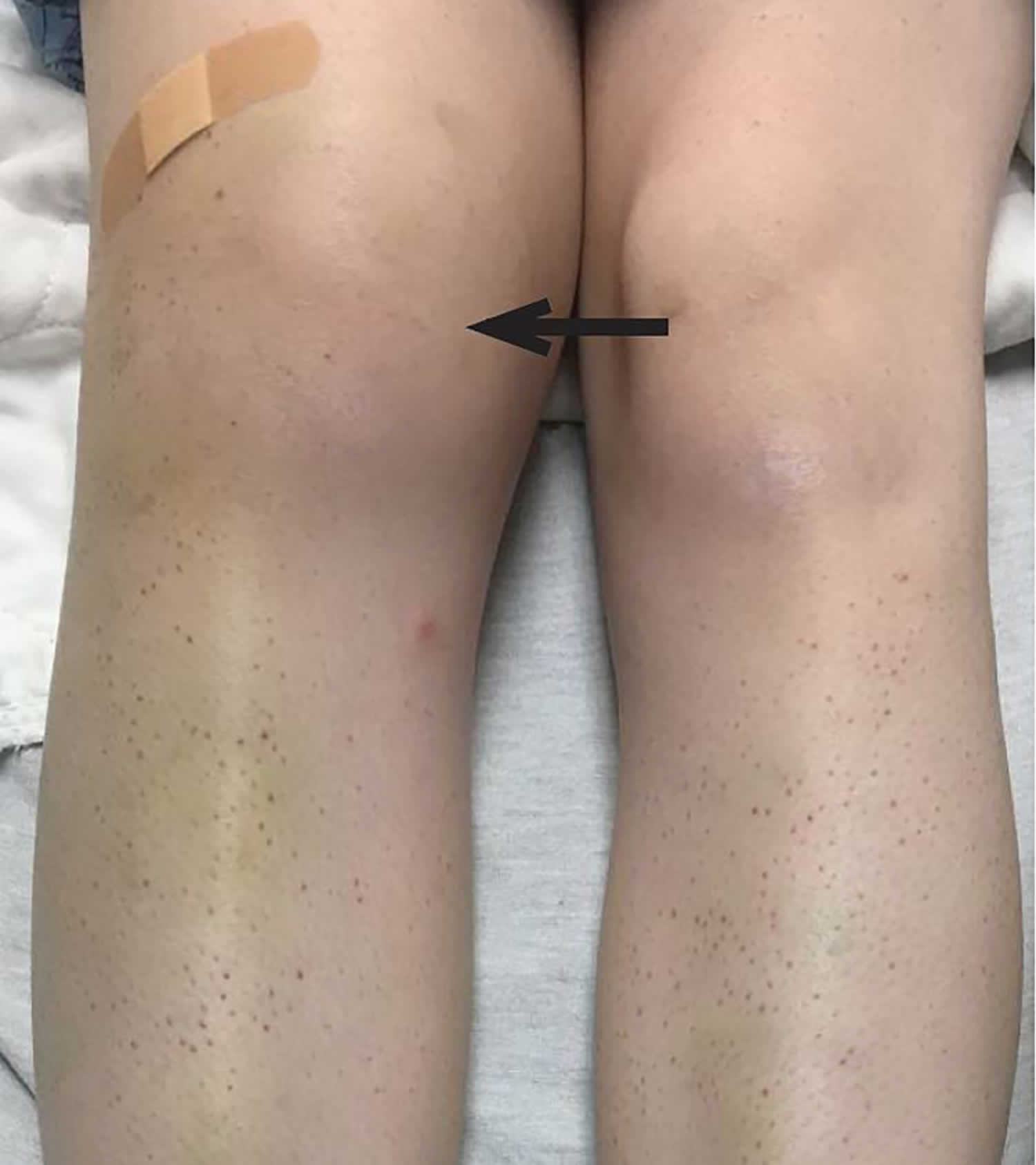
- In people with a history of kidney disease, kidney failure has been reported after treatment with IV vitamin C. People who are likely to develop kidney stones should not be treated with IV vitamin C.
- One study reported too much fluid in the body (fluid overload) related to IV vitamin C. This may have been caused by the IV delivery method and not the vitamin C.
- Case reports have shown that people with an inherited disorder called G6PD deficiency should not be given high doses of vitamin C because it may cause hemolysis.
- Because vitamin C may make iron more easily absorbed and used by the body, high doses of vitamin C are not recommended for people with hemochromatosis (a condition in which the body takes up and stores more iron than it needs).
- A drug interaction is a change in the way a drug acts in the body when taken with certain other drugs. When IV vitamin C is combined with certain anticancer drugs, the anticancer drugs may not work as well. So far, these effects have been seen only in some laboratory and animal studies.
Up to the present time the U.S. Food and Drug Administration (FDA) has not approved the use of IV vitamin C as a treatment for cancer. FDA does not approve dietary supplements as safe or effective. The company that makes the dietary supplements is responsible for making sure they are safe and that the claims on the label are true and do not mislead the consumer. The way that supplements are made is not regulated by FDA, so all batches and brands of IV vitamin C may not be the same.
High-Dose Intravenous Vitamin C for Cancer Treatment
This cancer information summary provides an overview of the use of high-dose vitamin C (also known as ascorbate or L-ascorbic acid) as a treatment for people with cancer 189. This summary includes a brief history of early clinical trials of high-dose vitamin C; reviews of laboratory, animal, and human studies; and current clinical trials.
Summary:
- Vitamin C is an essential nutrient with redox functions at normal physiologic concentrations.
- High-dose vitamin C has been studied as a treatment for cancer patients since the 1970s.
- Laboratory studies have reported that high-dose vitamin C has redox properties and decreased cell proliferation in prostate, pancreatic, hepatocellular, colon, mesothelioma, and neuroblastoma cell lines.
- Two studies of high-dose vitamin C in cancer patients reported improved quality of life and decreases in cancer-related side effects.
- Studies of vitamin C combined with other drugs in animal models have shown mixed results.
- Intravenous vitamin C has been generally well tolerated in clinical trials.
History
The earliest experience of using high-dose vitamin C (intravenous [IV] and oral) for cancer treatment was by a Scottish surgeon, Ewan Cameron, and his colleague, Allan Campbell, in the 1970s 190. This work led to a collaboration between Cameron and the Nobel Prize–winning chemist Linus Pauling, further promoting the potential of vitamin C therapy in cancer management 191, 192. As a result, two clinical trials of oral vitamin C were conducted in the late 1970s and early 1980s 193, 194.
Pharmacokinetic studies later revealed substantial differences in the maximum achieved blood concentrations of vitamin C based on the route of administration. When vitamin C is taken orally, plasma concentrations of the vitamin are tightly controlled, with a peak achievable concentration less than 300 µM. However, this tight control is bypassed with IV administration of the vitamin, resulting in very high levels of vitamin C plasma concentration (i.e., levels up to 20 mM) 165, 195. Further research suggests that pharmacologic concentrations of ascorbate, such as those achieved with IV administration, may result in cell death in many cancer cell lines 196.
Human Clinical Studies of High Dose Intravenous Vitamin C in Cancer Treatment
In the early 1970s, a consecutive case series was conducted in which 50 advanced-cancer patients were treated with large doses of ascorbic acid 190. These patients began ascorbic acid treatment after conventional therapies were deemed unlikely to be effective. Patients received intravenous (IV) ascorbic acid (10 g /day for 10 consecutive days; some patients received higher doses), oral ascorbic acid (10 g/day), or both. The subjects exhibited a wide variety of responses to treatment, including no or minimal response, tumor regression, and tumor hemorrhage. However, the authors noted that lack of controls prevented definitive assignment of any beneficial responses to the ascorbic acid treatment. A case report published in 1975 detailed one of the patients who had experienced tumor regression 197. Diagnosed with reticulum cell sarcoma, the patient exhibited improvement in well-being and resolution of lung masses after being treated with ascorbic acid. When the patient’s daily dose of ascorbic acid was reduced, some of signs of the disease returned; however, remission was achieved again after the patient reverted to the higher initial dose.
A larger case series of terminal cancer patients treated with ascorbate was reported in 1976 190. In this study, 100 terminal cancer patients (50 of whom were reported on previously) 190 were treated with ascorbate (10 g/day for 10 days IV, then orally) and compared with 1,000 matched controls from the same hospital. The mean survival time for ascorbate-treated patients was 300 days longer than that of the matched controls 191, 192.
Two studies tried to reproduce earlier results 193, 194. These studies were randomized, placebo-controlled trials in which cancer patients received either 10 g oral vitamin C or placebo daily until signs of cancer progression. At the end of each study, no significant differences were noted between the two ascorbate-treated and placebo-treated groups for symptoms, performance status, or survival 193, 194.
Recent Case Series Studies and Clinical Trials With Ascorbate Only
One study reported three case reports of cancer patients who received IV vitamin C as their main therapy. During vitamin C therapy, the patients used additional treatments, including vitamins, minerals, and botanicals. According to the authors, the cases were reviewed in accordance with the National Cancer Institute’s Best Case Series guidelines. Histopathologic examination suggested poor prognoses for these patients, but they had long survival times after being treated with IV vitamin C 198. Vitamin C was given at doses ranging from 15 g to 65 g, initially once or twice a week for several months; two patients then received it less frequently for 1 to 4 years.
One study demonstrated that IV vitamin C treatment resulted in improved quality of life and a decrease in cancer-related side effects in cancer patients 199.
A single-arm pilot study of weekly infusions of 60 g of ascorbic acid for 9 weeks in castration-resistant prostate cancer patients failed to observe a reduction in serum prostate-specific antigen (PSA) or tumor regression 200.
Studies have shown that vitamin C can be safely administered to healthy volunteers or cancer patients at doses up to 1.5 g/kg and with screening to eliminate treating individuals with risk factors for toxicity (e.g., glucose-6-phosphate dehydrogenase deficiency [G6PD], renal diseases, or urolithiasis). These studies have also found that plasma concentrations of vitamin C are higher with IV administration than with oral administration and are maintained for more than 4 hours 165, 195.
Ascorbate-Combination Trials
A phase 1 study published in 2012 examined the safety and efficacy of combining IV ascorbate with gemcitabine and erlotinib in patients with stage 4 pancreatic cancer 201. Fourteen patients entered the study and planned to receive IV gemcitabine (1,000 mg/m² over 30 minutes, once a week for 7 weeks), oral erlotinib (100 mg daily for 8 weeks), and IV ascorbate (50 g/infusion, 75 g/infusion, or 100 g/infusion 3 times per week for 8 weeks). Minimal adverse effects were reported for ascorbic acid treatment 201. Five patients received fewer than 18 of the planned 24 ascorbate infusions and thus did not have follow-up imaging to assess response. Three of those patients had clinically determined progressive disease. All of the other nine patients had repeat imaging to assess tumor size, and each met the criteria for having stable disease 201.
A 2013 phase 1 clinical study evaluated the safety of combining pharmacological ascorbate with gemcitabine in treating patients with stage 4 pancreatic cancer 202. During each 4-week cycle, patients received gemcitabine weekly for 3 weeks (1,000 mg/m² over 30 minutes) and twice-weekly ascorbate infusions for 4 weeks (15 g over 30 minutes during the first week, followed by weekly escalations in dose until plasma levels reached at least 350 mg/dL [20 mM]). Among nine patients, mean progression-free survival was 26 weeks and overall survival was 12 months. The combination treatment was well tolerated, and no significant adverse events were reported 203.
In 2014, a phase 1/2A clinical trial evaluated the toxicities of combining IV ascorbate with carboplatin and paclitaxel in stage 3/4 ovarian cancer 204. Twenty-seven patients were randomly assigned to receive either chemotherapy alone or chemotherapy and IV vitamin C concurrently. Chemotherapy was given for 6 months, and IV vitamin C was given for 12 months. The addition of IV vitamin C was associated with reduced chemotherapy-related toxicities 204.
A 2015 phase 1/2 clinical trial of high-dose IV vitamin C (approximately 1.5 g/kg body weight) combined with various chemotherapies, depending on the specific cancer diagnosis, was conducted to do the following 205:
- Observe the associated adverse events.
- Assess the pharmacokinetic profiles of vitamin C and oxalic acid levels prechemotherapy- and postchemotherapy.
- Assess clinical responses.
- Assess changes in mood.
- Assess changes in quality of life.
High-dose IV vitamin C was analyzed in 14 patients and was generally well tolerated and safe 205. Minor temporary adverse effects included increased urinary flow, thirst, nausea, vomiting, and chills, some of which could be prevented. Chemotherapy administration did not affect the plasma concentration of vitamin C. Although a few patients experienced temporary stable disease, functional improvement, and increased energy, the sample size is so small that the generalizability of these results is uncertain 205.
In 2017, a phase 1/2A study reported using IV vitamin C with standard-of-care gemcitabine chemotherapy in patients with newly diagnosed pancreatic cancer 206. Seven participants were initially enrolled. When safety was confirmed, an additional seven participants were enrolled. Twelve of the 14 enrolled participants completed the phase 1 pharmacokinetic evaluation 206. The evaluation consisted of IV vitamin C and gemcitabine pharmacokinetic measurements, each as single drugs, and then followed by the pharmacokinetic measurement of IV vitamin C combined with gemcitabine. IV vitamin C administration did not interfere with gemcitabine.
In May 2019, a phase 1 study was published that examined the safety, pharmacokinetics, and efficacy of high-dose IV vitamin C combined with the combination chemotherapy regimens mFOLFOX6 (oxaliplatin + leucovorin + fluorouracil) or FOLFIRI (leucovorin + fluorouracil + irinotecan hydrochloride) 207. This study consisted of 36 patients with metastatic colorectal cancer or gastric cancer. The main goal was to determine the maximum-tolerated dose and the recommended phase 1 dose of ascorbic acid with coadministration of either mFOLFOX6 (oxaliplatin + leucovorin + fluorouracil) or FOLFIRI (leucovorin + fluorouracil + irinotecan hydrochloride) 207. Patients received chemotherapy treatment on a 14-day cycle with vitamin C infusions occurring for 3 consecutive days for 3 hours at a time. For the dose-escalation portion of the study, ascorbic acid doses ranged from 0.2 g/kg to 1.5 g/kg. To determine the optimal administration rate of ascorbic acid, patient cohorts received infusion rates set at 0.6 g/min, 0.8 g/min, or 1 g/min. The study showed no dose-limiting toxicity for all doses and dosing rates; thus a maximum-tolerated dose was not reached, leading to a recommended phase II dose of 1.5 g/kg for ascorbic acid. Overall, no severe adverse reactions occurred, and the treatments were deemed safe and tolerable. A randomized phase 3 trial is being conducted to determine the clinical efficacy of ascorbic acid with mFOLFOX6 with or without bevacizumab in patients with metastatic colorectal cancer 208.
Trials of high-dose IV vitamin C combined with other drugs are ongoing 204, 201. A number of studies have included small doses of IV ascorbic acid treatment (1,000 mg) with arsenic trioxide regimens, with mixed results.
Various trials of high-dose IV vitamin C with other drugs are ongoing. There are currently five trials being conducted by researchers at the University of Iowa; four phase 2 studies and one phase 1B/2 study. The four phase 2 clinical trials are investigating the efficacy of high-dose ascorbate combined with standard anticancer regimens. The studies are exploring the combination of high-dose ascorbate with the following:
- Standard non-small cell lung cancer therapy, including radiation therapy, carboplatin, and paclitaxel (NCT02905591).
- Standard therapy for metastatic pancreatic adenocarcinoma, including gemcitabine and nab-paclitaxel (NCT02905578).
- Standard therapy for localized pancreatic adenocarcinoma with gemcitabine and radiation therapy (NCT03541486).
- Standard therapy for glioblastoma multiforme, including temozolomide and radiation therapy (NCT02344355).
Another phase 1B/2 trial (NCT03508726) is studying the safety and efficacy of high-dose ascorbate with preoperative radiation therapy in locally advanced soft tissue sarcoma patients.
Several studies have included IV ascorbic acid treatment at a fixed dose of 1,000 mg with arsenic trioxide regimens, with mixed results. Researchers using this approach suggested that the pro-oxidant properties of IV ascorbic acid may have helped to increase the effects of arsenic trioxide by sensitization of malignant cells to arsenic’s cytotoxic effects 209. The combination therapies were well tolerated and suggested beneficial effects in multiple myeloma patients, although the specific contribution of vitamin C could not be determined 210, 211, 212, 213. However, similar combination regimens resulted in severe side effects, disease progression, and no anticancer effect in patients with refractory metastatic colorectal cancer 214 and metastatic melanoma 215. Because these were not placebo-controlled trials, the extent that ascorbate contributed to the toxicity or efficacy demonstrated in these studies is unclear.
Adverse Effects of High Dose Intravenous Vitamin C
Intravenous (IV) high-dose ascorbic acid has been generally well tolerated in clinical trials 216, 217, 218, 219, 220, 221, 222, 223. Renal failure following ascorbic acid treatment has been reported in patients with preexisting renal disorders 224.
Case reports have indicated that patients with glucose-6-phosphate dehydrogenase (G-6-PD) deficiency should not receive high doses of vitamin C because of the risk of developing hemolysis 225, 226, 227.
Vitamin C may increase bioavailability of iron, and high doses of the vitamin are not recommended for patients with hemochromatosis (a hereditary disorder in which iron salts are deposited in the tissues, leading to liver damage, diabetes mellitus, and bronze discoloration of the skin) 228.
High Dose Intravenous Vitamin C Drug Interactions
When administered in high doses, vitamin C may result in adverse interactions with some anticancer agents. These interactions have primarily been detected in preclinical studies. A 2013 phase I clinical study evaluated the safety of combining high-dose IV ascorbate with gemcitabine in stage IV pancreatic cancer patients. The combination therapy was well tolerated by patients, and no significant adverse events were reported 229.
In vitro and in vivo animal studies have suggested that combining oral vitamin C with bortezomib interferes with the drug’s ability to act as a proteasome inhibitor and blocks bortezomib-initiated apoptosis 230, 231, 232. This interference occurred even with the oral administration of vitamin C (40 mg /kg /day) to animals. Studies in cell culture and performed by adding blood plasma from healthy volunteers given oral vitamin C (1 g /day) also showed a significant decrease in bortezomib’s growth inhibitory effect on multiple myeloma cells. Another study found similar results. Plasma from healthy volunteers who took 1 g of oral vitamin C per day was shown to decrease bortezomib growth inhibition in multiple myeloma cells and to block its inhibitory effect on 20S proteasome activity 232. However, a study that utilized mice harboring human prostate cancer cell xenografts failed to find any significant effect of oral vitamin C (40 mg/kg/day or 500 mg/kg/day) on the tumor growth inhibitory action of bortezomib 233.
Several studies have been performed to assess the potential synergistic or inhibitory action of vitamin C on certain chemotherapy drugs, with variable results. A series of studies in cell culture and in animals bearing tumors has shown that when given at high concentrations or dosages, dehydroascorbic acid (an oxidized form of vitamin C) can interfere with the cytotoxic effects of several chemotherapy drugs 234. However, dehydroascorbic acid is generally present only at low concentrations in dietary supplements and fresh foods.
Cardiovascular disease
Evidence from many epidemiological studies suggests that high intakes of fruits and vegetables are associated with a reduced risk of cardiovascular disease 235, 236. This association might be partly attributable to the antioxidant content of these foods because oxidative damage, including oxidative modification of low-density lipoproteins, is a major cause of cardiovascular disease 236. In addition to its antioxidant properties, vitamin C has been shown to reduce monocyte adherence to the endothelium, improve endothelium-dependent nitric oxide production and vasodilation, and reduce vascular smooth-muscle-cell apoptosis, which prevents plaque instability in atherosclerosis 237.
Results from prospective studies examining associations between vitamin C intake and cardiovascular disease risk are conflicting 236. In the Nurses’ Health Study, a 16-year prospective study involving 85,118 female nurses, total intake of vitamin C from both dietary and supplemental sources was inversely associated with coronary heart disease risk 238. However, intake of vitamin C from diet alone showed no significant associations, suggesting that vitamin C supplement users might be at lower risk of coronary heart disease. A much smaller study indicated that postmenopausal women with diabetes who took at least 300 mg/day vitamin C supplements had increased cardiovascular disease mortality 239.
A prospective study in 20,649 British adults found that those in the top quartile of baseline plasma vitamin C concentrations had a 42% lower risk of stroke than those in the bottom quartile 240. In male physicians participating in the Physicians’ Health Study, use of vitamin C supplements for a mean of 5.5 years was not associated with a significant decrease in total cardiovascular disease mortality or coronary heart disease mortality 241. A pooled analysis of nine prospective studies that included 293,172 subjects free of coronary heart disease at baseline found that people who took ≥700 mg/day of supplemental vitamin C had a 25% lower risk of coronary heart disease incidence than those who took no supplemental vitamin C 242. The authors of a 2008 meta-analysis of prospective cohort studies, including 14 studies reporting on vitamin C for a median follow-up of 10 years, concluded that dietary, but not supplemental, intake of vitamin C is inversely associated with coronary heart disease risk 235.
Results from most clinical intervention trials have failed to show a beneficial effect of vitamin C supplementation on the primary or secondary prevention of cardiovascular disease 243, 244. In the Women’s Antioxidant Cardiovascular Study, a secondary prevention trial involving 8,171 women aged 40 years or older with a history of cardiovascular disease, supplementation with 500 mg/day vitamin C for a mean of 9.4 years showed no overall effect on cardiovascular events 243. Similarly, vitamin C supplementation (500 mg/day) for a mean follow-up of 8 years had no effect on major cardiovascular events in male physicians enrolled in the Physicians’ Health Study II 244.
Other clinical trials have generally examined the effects on cardiovascular disease of supplements combining vitamin C with other antioxidants, such as vitamin E and beta-carotene, making it more difficult to isolate the potential contribution of vitamin C. The SU.VI.MAX study 157 examined the effects of a combination of vitamin C (120 mg/day), vitamin E (30 mg/day), beta-carotene (6 mg/day), selenium (100 mcg/day), and zinc (20 mg/day) in 13,017 French adults from the general population. After a median follow-up time of 7.5 years, the combined supplements had no effect on ischemic cardiovascular disease in either men or women 157. In the Women’s Angiographic Vitamin and Estrogen study 245, involving 423 postmenopausal women with at least one coronary stenosis of 15%–75%, supplements of 500 mg vitamin C plus 400 IU vitamin E twice per day not only provided no cardiovascular benefit, but significantly increased all-cause mortality compared with placebo 245.
The authors of a 2006 meta-analysis of randomized controlled trials concluded that antioxidant supplements (vitamins C and E and beta-carotene or selenium) do not affect the progression of atherosclerosis 246. Similarly, a systematic review of vitamin C’s effects on the prevention and treatment of cardiovascular disease found that vitamin C did not have favorable effects on cardiovascular disease prevention 247. Since then, researchers have published follow-up data from the Linxian trial, a population nutrition intervention trial conducted in China 162. In this trial, daily vitamin C supplements (120 mg) plus molybdenum (30 mcg) for 5–6 years significantly reduced the risk of cerebrovascular deaths by 8% during 10 years of follow-up after the end of the active intervention 162.
Although the Linxian trial 162 data suggest a possible benefit, overall, the findings from most intervention trials do not provide convincing evidence that vitamin C supplements provide protection against cardiovascular disease or reduce its morbidity or mortality. However, as discussed in the cancer prevention section above, clinical trial data for vitamin C are limited by the fact that plasma and tissue concentrations of vitamin C are tightly controlled in humans. If subjects’ vitamin C levels were already close to saturation at study entry, supplementation would be expected to have made little or no difference on measured outcomes 168.
Age-related macular degeneration (AMD) and cataracts are two of the leading causes of vision loss in older individuals. Oxidative stress might contribute to the cause of both conditions. Thus, researchers have hypothesized that vitamin C and other antioxidants play a role in the development and/or treatment of these diseases.
A population-based cohort study in the Netherlands found that adults aged 55 years or older who had high dietary intakes of vitamin C as well as beta-carotene, zinc, and vitamin E had a reduced risk of AMD 248. However, most prospective studies do not support these findings 249. The authors of a 2007 systematic review and meta-analysis of prospective cohort studies and randomized clinical trials concluded that the current evidence does not support a role for vitamin C and other antioxidants, including antioxidant supplements, in the primary prevention of early age-related macular degeneration (AMD) 250.
Although research has not shown that antioxidants play a role in age-related macular degeneration (AMD) development, some evidence suggests that they might help slow AMD progression 251. The Age-Related Eye Disease Study (AREDS), a large, randomized, placebo-controlled clinical trial, evaluated the effect of high doses of selected antioxidants (500 mg vitamin C, 400 IU vitamin E, 15 mg beta-carotene, 80 mg zinc, and 2 mg copper) on the development of advanced AMD in 3,597 older individuals with varying degrees of AMD 252. After an average follow-up period of 6.3 years, participants at high risk of developing advanced AMD (i.e., those with intermediate AMD or those with advanced AMD in one eye) who received the antioxidant supplements had a 28% lower risk of progression to advanced AMD than participants who received a placebo. A follow-up AREDS2 study confirmed the value of this and similar supplement formulations in reducing the progression of AMD over a median follow-up period of 5 years 253.
High dietary intakes of vitamin C and higher plasma ascorbate concentrations have been associated with a lower risk of cataract formation in some studies 147. In a 5-year prospective cohort study conducted in Japan, higher dietary vitamin C intake was associated with a reduced risk of developing cataracts in a cohort of more than 30,000 adults aged 45–64 years 254. Results from two case-control studies indicate that vitamin C intakes greater than 300 mg/day reduce the risk of cataract formation by 70%–75% 16. Use of vitamin C supplements, on the other hand, was associated with a 25% higher risk of age-related cataract extraction among a cohort of 24,593 Swedish women aged 49–83 years 255. These findings applied to study participants who took relatively high-dose vitamin C supplements (approximately 1,000 mg/day) and not to those who took multivitamins containing substantially less vitamin C (approximately 60 mg/day).
Data from clinical trials are limited. In one study, Chinese adults who took daily supplements of 120 mg vitamin C plus 30 mcg molybdenum for 5 years did not have a significantly lower cataract risk 256. However, adults aged 65–74 years who received 180 mg vitamin C plus 30 mcg molybdenum combined with other nutrients in a multivitamin/mineral supplement had a 43% significantly lower risk of developing nuclear cataracts than those who received a placebo 256. In the AREDS study 257, older individuals who received supplements of 500 mg vitamin C, 400 IU vitamin E, and 15 mg beta-carotene for an average of 6.3 years did not have a significantly lower risk of developing cataracts or of cataract progression than those who received a placebo. The AREDS2 study 258, which also tested formulations containing 500 mg vitamin C, confirmed these findings.
Overall, the currently available evidence does not indicate that vitamin C, taken alone or with other antioxidants, affects the risk of developing AMD, although some evidence indicates that the AREDS formulations might slow AMD progression in people at high risk of developing advanced AMD.
Common cold
In the 1970s Linus Pauling suggested that vitamin C could successfully treat and/or prevent the common cold 259. Results of subsequent controlled studies have been inconsistent, resulting in confusion and controversy, although public interest in the subject remains high 260.
A 2007 Cochrane review 260 examined placebo-controlled trials involving the use of at least 200 mg/day vitamin C taken either continuously as a prophylactic treatment or after the onset of cold symptoms. Prophylactic use of vitamin C did not significantly reduce the risk of developing a cold in the general population. However, in trials involving marathon runners, skiers, and soldiers exposed to extreme physical exercise and/or cold environments, prophylactic use of vitamin C in doses ranging from 250 mg/day to 1 g/day reduced cold incidence by 50%. In the general population, use of prophylactic vitamin C modestly reduced cold duration by 8% in adults and 14% in children. When taken after the onset of cold symptoms, vitamin C did not affect cold duration or symptom severity.
In addition, a 2013 Cochrane review by the same investigators identified only two small randomized, double-blind, placebo-controlled trials that examined the effect of vitamin C on the incidence of respiratory infection-induced asthma 261. One trial found that vitamin C supplementation (1 g/day) for 14 weeks reduced the risk of asthma attacks precipitated by respiratory infection. The other trial randomized subjects diagnosed with infection-related asthma to receive 5 g/day of vitamin C or a placebo for one week; a lower proportion of participants was found to present with bronchial hypersensitivity to histamine — which characterizes chronic asthma — in the vitamin C group compared to the control group 261. These observations need to be confirmed in larger, well-designed trials.
Overall, the evidence to date suggests that regular intakes of vitamin C at doses of at least 200 mg/day do not reduce the incidence of the common cold in the general population, but such intakes might be helpful in people exposed to extreme physical exercise or cold environments and those with marginal vitamin C status, such as the elderly and chronic smokers 262. The use of vitamin C supplements might shorten the duration of the common cold and ameliorate symptom severity in the general population 262, possibly due to the anti-histamine effect of high-dose vitamin C 263. However, taking vitamin C after the onset of cold symptoms does not appear to be beneficial 264.
Asthma
A 2013 systematic review identified 11 randomized controlled studies that evaluated the effect of vitamin C on asthma (eight trials) or exercise-induced bronchoconstriction (three trials) 265. Exercise-induced bronchoconstriction is a transient narrowing of the airways that occurs after exercise and is indicated by a ≥10% decline in Forced Expiratory Volume in 1 second (FEV1). In the three trials that included a total of 40 participants with exercise-induced bronchoconstriction, vitamin C administration before exercise (a 0.5-g dose on two subsequent days in one trial, a single dose of 2 g in the second trial, and 1.5 g daily for two weeks in the third trial) significantly reduced the exercise-induced decline in FEV1. Among the five out of eight trials in asthmatic subjects that reported on FEV1 outcomes, none found a difference between vitamin C supplementation and placebo 265.
Vitamin C and Skin Health
Vitamin C (ascorbic acid) is a normal skin constituent that is found at high levels in both the dermis and epidermis 266, 267. The vitamin C content of the epidermis is higher than the dermis, although the vitamin C concentrations in both layers are approximately equal to that of other water-soluble antioxidants, including uric acid and glutathione 267, 268, 269. Aging, however, causes a decline in vitamin C content in both the epidermis and dermis 267. Excessive exposures to ultraviolet (UV) light or pollutants (e.g., cigarette smoke and ozone) may also lower vitamin C content, primarily in the epidermis 269, 270, 271.
The antioxidant properties of vitamin C (ascorbic acid) and its role in collagen synthesis make vitamin C a vital molecule for skin health 272. Dietary and topical vitamin C (ascorbic acid) have beneficial effects on skin cells, and some studies have shown that vitamin C may help prevent and treat ultraviolet (UV)-induced photoaging (photodamage) and is needed for adequate wound healing 272. Oral supplementation with vitamin C may help prevent UV-induced damage, especially in combination with supplemental vitamin E. Dietary vitamin C may also provide photoprotection, but the extent of the protective effects will require additional research. Topical application of vitamin C (ascorbic acid) appears to be an effective route for delivering vitamin C (ascorbic acid) to the skin because ascorbic acid appears to be taken up readily at an acidic pH. Despite inconsistencies in vitamin C preparations and study design, the data suggest that vitamin C is most effective in protecting against damage induced by UV light and also has utility in the treatment of photodamage and/or skin wrinkling. Although vitamin C appears to benefit dry skin and may support wound healing, further research is needed to determine the effect of vitamin C on both. Lastly, the greatest effects of vitamin C supplementation are seen when it is combined with other micronutrients, such as vitamin E and zinc.
Vitamin C in the skin is normally transported from the bloodstream. Transport proteins specific for vitamin C (ascorbic acid) are found on cells in all layers of the skin 273. Keratinocytes (skin cells) have a high capacity for vitamin C transport, possibly to compensate for limited vascularization of the epidermis 273, 274. Oral supplementation with vitamin C effectively increases vitamin C levels in the skin 275, 276. However, when plasma vitamin C levels are saturated, skin vitamin C concentrations no longer increase. Optimum skin concentrations of the vitamin are not yet known 272.
Symptoms of vitamin C deficiency is known as scurvy appears once plasma concentrations of vitamin C (ascorbic acid) drop below 10 micromolar (μM), a level that can be prevented by consuming as little as 10 mg of ascorbic acid daily 277. Skin signs and symptoms of vitamin C deficiency or scurvy result from declines in collagen synthesis, leading to disruption of connective tissue and fragility of blood vessels. Early symptoms in the skin include a thickening of the stratum corneum and spots of small subcutaneous bleeding 278, 279. As scurvy progresses, wound healing is impaired due to the loss of mature collagen, which allows wounds to remain open 280, 281. Skin lesions caused by vitamin C deficiency are treated by an adequate intake of vitamin C.
Ultraviolet protection
Vitamin C limits the damage induced by ultraviolet (UV) light exposure. Vitamin C is not a “sunscreen” because it does not absorb light in the UVA or UVB spectrum. Rather, the antioxidant activity of vitamin C protects against UV-induced damage caused by free radicals 282. Vitamin C transport proteins are increased in keratinocytes in response to UV light, suggesting an increased need for vitamin C uptake for adequate protection 273, 274.
UV light decreases vitamin C content of skin, an effect that is dependent on the intensity and duration of UV exposure 283, 269, 271. In cultured keratinocytes, the addition of vitamin C reduces UV-related DNA damage and lipid peroxidation, limits the release of pro-inflammatory cytokines, and protects against apoptosis 284, 285. Vitamin C also modulates redox-sensitive cell signaling in cultured skin cells and consequently increases cell survival following UV exposure 286, 287.
In two rodent studies, addition of ascorbic acid to the diet reduced the size and number of dermal neoplasms and skin tumors induced by chronic UV exposure 288, 289. To date, no other studies with UV exposure and oral ascorbic acid supplementation in animal models have been published.
In two human studies, oral vitamin C supplementation alone did not significantly increase Minimal Erythemal Dose (MED), a measure of photoprotection from UV light in skin 275, 276. Overall, limited data suggest that vitamin C consumption alone provides insufficient antioxidant protection against UV irradiation. However, multiple studies have found that oral supplementation with a combination of vitamin C and vitamin E effectively increases MED and decreases erythema-induced blood flow to damaged areas of skin 290, 291. Therefore, interactions between the two antioxidant vitamins may be necessary to achieve UV protection by dietary means.
Topical application of vitamin C, alone or in combination with other compounds, may result in greater photoprotection than oral supplementation because of the more direct route of administration. In one mouse study, topical application of ascorbic acid delayed the effects of chronic high-dose UVB exposure on the skin, including a reduction in skin wrinkling and the development of skin tumors 292. In pig models, topical application of vitamin C reduced the number of sunburned cells, decreased redness response, and reduced DNA damage induced by UVA exposure 293, 282. Topically applied combinations of vitamin C and vitamin E are more effective in preventing photodamage than either vitamin alone. In particular, this combination of antioxidant vitamins decreased the immunosuppressive effects of UV exposure 294, increased Minimal Erythemal Dose (MED), and decreased cell damage 293, 295, 296.
Limited human studies are available on photoprotection by topical application of vitamin C. Although topical ascorbic acid reduces radicals in UV-exposed human skin 297, only one study examined its effect on UV-induced erythemal response; this study reported no significant benefit of topical vitamin C 298. Like animal research, human studies using combinations of vitamin C and vitamin E have documented UV protective effects 298, 299, 300.
Photodamage and Wrinkling
The accumulation of oxidative damage to proteins is a distinguishing feature of both photoaging (photodamage) and intrinsic aging; such oxidative damage can lead to changes in skin structure. In addition to its antioxidant functions, vitamin C regulates the synthesis of the structural protein collagen. The role of vitamin C in the hydroxylation of collagen molecules is well characterized 301. Hydroxylation of collagen is necessary for its extracellular stability and support of the epidermis.
In cell culture models, vitamin C supplementation has many beneficial effects in combating photodamage. Specifically, vitamin C has been shown to stabilize collagen mRNA, thus increasing collagen protein synthesis for repair of the damaged skin 302. This occurs concurrently with a decrease in elastin production; the elastin protein is often overproduced in response to photodamage 303. Vitamin C also increases the proliferation rate of fibroblasts, a capacity that is decreased with age 304. Furthermore, vitamin C stimulates DNA repair in cultured fibroblasts 305.
Human studies often assess skin health by changes in depth or number of wrinkles and by the individual’s perception of skin health. Two observational studies found that higher intakes of vitamin C from the diet were associated with better skin appearance, with notable decreases in skin wrinkling 306, 307. The use of vitamin C (3 to 10%) in topical applications for at least 12 weeks has been shown to decrease wrinkling 308, 309, 310, 311, reduce protein fiber damage 310, decrease apparent roughness of skin 308, and increase production of collagen 311, 312. Topical vitamin C has also been shown to reverse some of the age-related structural changes in the interface between the dermis and the epidermis 313. However, the effects of topical vitamin C are not apparent in all individuals, and interestingly, one study found that individuals with high dietary intakes of vitamin C showed no or little effect of a topical administration 312.
Wound healing
One of the distinctive features of scurvy is poor wound healing 314. Vitamin C levels decrease rapidly at a wound site 315, 316. Although inflammatory responses often increase free radicals at the site of injury and the presence of vitamin C may limit free radical damage, free radicals may play a complex role in the healing response that is not yet understood 316, 317. However, the increased demand for dermal collagen synthesis may increase utilization of vitamin C. Vitamin C may have additional roles in wound healing, for example, by promoting keratinocyte differentiation 305, 318, stimulating the formation of the epidermal barrier 319 and re-establishing the stratum corneum 320.
Studies on the effect of vitamin C supplementation on wound healing have reported somewhat mixed results. Data from laboratory animals and humans show that vitamin C deficiency results in poor wound healing, and vitamin C supplementation in deficient individuals shows significant benefits 321, 322, 323. Although vitamin C levels appear to increase collagen synthesis and decrease inflammatory responses at the site of the wound, neither vitamin C supplementation 324, 325, 326 or increased plasma vitamin C status 317 increases wound closure time in otherwise healthy individuals. This suggests that vitamin C may only affect specific facets of the wound healing response. Topical ascorbic acid has not been properly evaluated prior to or during wound healing in humans.
Vitamin C is included in oral therapies for pressure ulcers (bed sores) and burns, along with vitamin E, zinc, and other nutritive factors 327, 328, 329.
Dry skin
Higher intakes of dietary vitamin C have been correlated with a decreased risk of dry skin 306, suggesting that vitamin C (ascorbic acid) may have effects on trans-epidermal water loss. In cell culture models, addition of vitamin C promotes the synthesis of barrier lipids, which would establish a functioning stratum corneum with low water permeability 320, 330. One study using topical vitamin C in human subjects correlated its use with decreased skin roughness 308. However, another study suggested vitamin C increased trans-epidermal water loss when applied to the skin 331. Therefore, the effects of vitamin C on skin dryness are not clear.
Other skin functions
Environmental pollutants, such as ozone, can decrease vitamin C levels in the skin and lead to free radical damage 270. Smoking also leads to increased wrinkling and decreased collagen synthesis, which corresponds to a decline in plasma vitamin C levels 317; however, it is unclear if this correlates to lower vitamin C levels in skin. Topical vitamin C (ascorbic acid) has not been evaluated against pollution-related damage.
Topical vitamin C (ascorbic acid) may be useful against acne to reduce inflammatory lesions. However, studies have primarily focused on the use of sodium ascorbyl phosphate, a synthetic derivative that may be poorly absorbed by the skin 332, 333. Topical vitamin C may also have mild skin lightening effects that are caused by reductions in melanin production and melanin oxidation 334.
Lead toxicity
Although the use of lead paint and leaded gasoline has been discontinued in the US, lead toxicity continues to be a significant health problem, especially in children living in urban areas. Abnormal growth and development have been observed in infants of women exposed to lead during pregnancy, while children who are chronically exposed to lead are more likely to develop learning disabilities, behavioral problems, and to have a low IQ. In adults, lead toxicity may result in kidney damage, high blood pressure, and anemia.
Several cross-sectional studies have reported an inverse association between vitamin C status and blood lead concentration 335. For instance, in a study of 747 older men, blood lead concentration was significantly higher in those who reported total dietary vitamin C intakes averaging less than 109 mg/day compared to those with higher vitamin C intakes 335. A much larger study of 19,578 people, including 4,214 children from 6 to 16 years of age, found higher serum vitamin C concentrations to be associated with significantly lower blood lead concentrations 336. A US national survey of more than 10,000 adults found that blood lead concentrations were inversely related to serum vitamin C concentrations 337.
Cigarette smoking or second-hand exposure to cigarette smoke contributes to increased blood lead concentration and a state of chronic low-level lead exposure. An intervention trial in 75 adult male smokers found that supplementation with 1,000 mg/day of vitamin C resulted in significantly lower blood lead concentration over a four-week treatment period compared to placebo 338. A lower dose of 200 mg/day did not significantly affect blood lead concentration, although serum vitamin C concentrations were not different from those in the group who took 1,000 mg/day.
The mechanism(s) by which vitamin C reduces blood lead concentration is not known, yet it has been proposed that vitamin C could inhibit intestinal absorption 338 or enhance urinary excretion of lead 339.
Type 2 diabetes mellitus
In the National Institutes of Health (NIH)-American Association of Retired Persons (AARP) Diet and Health study that included 232,007 participants, the use of vitamin C supplements for at least seven times a week was associated with a 9% lower risk of developing type 2 diabetes mellitus compared to non-supplement use 340. In a cohort of 21,831 adults followed for 12 years in the EPIC-Norfolk study, high plasma vitamin C was found to be strongly associated with a reduced risk of diabetes 341. Additionally, several cross-sectional studies reported inverse associations between circulating vitamin C concentrations and markers of insulin resistance or glucose intolerance, such as glycated hemoglobin (HbA1c) concentration 342, 343, 344. Yet, short-term randomized controlled studies have found no effect of vitamin C supplementation on fasting glucose, fasting insulin, and HbA1c concentrations in healthy individuals 345. It is not known whether supplemental vitamin C could improve markers of glycemic control in subjects at risk of diabetes.
Gout
Gout, a condition that afflicts more than 4% of US adults 346, is characterized by abnormally high blood concentrations of uric acid (urate) 347. Urate crystals may form in joints, resulting in inflammation and pain, as well as in the kidneys and urinary tract, resulting in kidney stones. The tendency to exhibit elevated blood uric acid concentrations and develop gout is often inherited; however, dietary and lifestyle modification may be helpful in both the prevention and treatment of gout 348. In an observational study that included 1,387 men, higher intakes of vitamin C were associated with lower serum concentrations of uric acid 349. In a cross-sectional study conducted in 4,576 African Americans, the odds of having hyperuricemia was associated with dietary intakes high in fructose, low in vitamin C, or with high fructose-to-vitamin C ratios 350. A prospective study that followed a cohort of 46,994 men for 20 years found that total daily vitamin C intake was inversely associated with incidence of gout, with higher intakes being associated with greater risk reductions 351. The results of this study also indicated that supplemental vitamin C may be helpful in the prevention of gout 351.
A 2011 meta-analysis of 13 randomized controlled trials in healthy individuals with elevated serum uric acid revealed that vitamin C supplementation (a median dose of 500 mg/day for a median duration of 30 days) modestly reduced serum uric acid concentrations by 0.35 mg/dL compared to placebo 352. Such a reduction falls within the range of assay variability and is unlikely to be clinically significant 353. An eight-week, open-label, controlled trial randomized 40 subjects with gout to receive either allopurinol (standard-of-care), vitamin C, or both treatments 354. The effect of vitamin C, alone or with allopurinol, decreasing serum uric acid was modest and much less than that of allopurinol alone. The trial did not examine the effect of vitamin C on other outcomes associated with gout 354.
Although observational studies suggested that supplemental vitamin C may be helpful to prevent incident and recurrent gout, this has not been demonstrated by intervention studies undertaken thus far. In addition, there is currently little evidence to support a role for vitamin C in the management of patients with gout 355.
Adverse pregnancy outcomes
A 2015 meta-analysis of 29 randomized controlled trials 356 found that administration of vitamin C during pregnancy, alone or in combination with a few other supplements, failed to reduce the risks of stillbirth, perinatal death, intrauterine growth restriction, preterm birth, premature rupture of membranes, and preeclampsia. Nonetheless, vitamin C supplementation led to a 36% lower risk of placental abruption and to a significant increase in gestational age at birth 356. Another meta-analysis of 40 randomized controlled trials in 276,820 women found no effect of vitamin C, alone or combined with vitamin E or multivitamins, when supplemented during pregnancy (starting prior to 20 weeks’ gestation), on the risks of overall fetal loss, miscarriage, stillbirth, and congenital malformation 357.
Cigarette smoking during pregnancy causes intrauterine growth restriction and preterm birth, among other pregnancy complications 358, 359 and is the primary cause of childhood respiratory illness 360. For some still unclear reasons, smoking has been associated with a lower risk of preeclampsia during pregnancy 361. A secondary analysis of a multicenter, randomized, double-blind, placebo-controlled trial in nearly 10,000 pregnant women found no reduction in the risk of preeclampsia with supplemental vitamin C (1,000 mg/day) and vitamin E (400 IU/day), regardless of women’s smoking status during pregnancy 362. However, antioxidant supplementation resulted in reduced risks of placental abruption and preterm birth in women who smoked during pregnancy but not in non-smokers 362. Another pilot multicenter trial found better lung function during the first week of life and lower risk of wheezing through one year of age in infants whose smoking mothers were randomized to receive vitamin C (500 mg/day) rather than a placebo during pregnancy 363. The Vitamin C to Decrease the Effects of Smoking in Pregnancy on Infant Lung Function (VCSIP) study is an ongoing trial designed to confirm these preliminary observations using more accurate measurements of pulmonary function in a larger sample of women randomized to receive supplemental vitamin C or placebo 364.
Alzheimer’s disease
In the US, Alzheimer’s disease is the most common form of dementia, affecting 5.5 million individuals 65 years and over 365. Oxidative stress, neuroinflammation, beta-amyloid plaque deposition, Tau protein-forming tangles, and neuronal cell death in the brain of subjects affected by Alzheimer’s disease have been associated with cognitive decline and memory loss. Lower vitamin C concentrations in the cerebrospinal fluid (CSF) and brain extracellular matrix of a mouse model of Alzheimer’s disease were found to increase oxidative stress and accelerate amyloid deposition and disease progression 366. In another Alzheimer’s disease mouse model that was lacking the ability to synthesize vitamin C, supplementation with a high versus low dose of vitamin C reduced amyloid deposition in the cortex and hippocampus and limited blood-brain barrier impairments and mitochondrial dysfunction 367.
The majority of large, population-based studies examining the relationship of vitamin C intake or supplementation with Alzheimer’s disease incidence have reported null results 368. In contrast, observational studies reported lower plasma vitamin C concentrations in Alzheimer’s disease patients compared to cognitively healthy subjects 369 and found better cognitive function or lower risk of cognitive impairment with higher plasma vitamin C 368.
Few studies have measured vitamin C concentration in the cerebrospinal fluid (CSF), which more closely reflects the vitamin C status of the brain. Vitamin C is concentrated in the brain through a combination of active transport into brain tissue and retention via the blood-brain barrier 368. Although cerebrospinal fluid (CSF) vitamin C is maintained at concentrations several-fold higher than plasma vitamin C, the precise function of vitamin C in cognitive function and Alzheimer’s disease cause is not yet fully understood 370. In a small, longitudinal biomarker study in 32 individuals with probable Alzheimer’s disease, a higher CSF-to-plasma vitamin C ratio at baseline was associated with a slower rate of cognitive decline at one year of follow-up 371. Impaired blood-brain barrier integrity may affect the brain’s ability to retain vitamin C and thus to maintain a high CSF-to-plasma vitamin C ratio. The significance of the CSF-to-plasma vitamin C ratio in Alzheimer’s disease progression requires further study.
The effect of vitamin C supplementation, in combination with other antioxidants, on cerebrospinal fluid (CSF) biomarkers and cognitive function has been examined in only a few trials involving Alzheimer’s disease patients. In a small (n=23), open-label trial 372, combined supplementation with vitamin C (1,000 mg/day) and vitamin E (400 IU/day) to Alzheimer’s disease patients taking a cholinesterase inhibitor significantly increased antioxidant levels and decreased lipoprotein oxidation in CSF after one year, but had no effect on the clinical course of Alzheimer’s disease compared to controls. A similar finding was obtained in a double-blind, randomized controlled trial in which combined supplementation with vitamin C (500 mg/day), vitamin E (800 IU/day), and α-lipoic acid (900 mg/day) for 16 weeks reduced lipoprotein oxidation in CSF but elicited no clinical benefit in individuals with mild-to-moderate Alzheimer’s disease (n=78) 373. In this latter trial, a greater decline in the Mini Mental State Examination (MMSE) score was observed in the supplemented group, however, the significance of this observation remains unclear. A third placebo-controlled trial in mildly cognitively impaired older adults (ages, 60-75 years) found that one-year supplementation with vitamin C (400 mg/day) and vitamin E (300 mg/day) improved antioxidant blood capacity but had no effect on MMSE scores 374.
At this time, avoidance of vitamin C deficiency or insufficiency, rather than supplementation in replete individuals, seems prudent for the promotion of healthy brain aging 369.
Sepsis
Sepsis and septic shock — defined as persistent sepsis-induced low blood pressure — are associated with elevated mortality rates in critically ill patients 375, 376. Because systemic inflammatory responses involve excessive oxidative stress, it has been suggested that providing antioxidant nutrients like vitamin C may improve the outcome of critically ill patients in intensive care units. In addition, vitamin C deficiency is common in critically ill patients, especially in those with septic shock, and persists despite enteral/parenteral nutritional therapy providing recommended amounts of vitamin C 377. Vitamin C requirements are likely to be increased in this population due to the hypermetabolic response driven by the systemic inflammatory reaction 378, 379. Intravenous administration of 50 mg or 200 mg of vitamin C per kg per day for 96 hours to patients with sepsis admitted in intensive care unit was found to correct vitamin C deficiency 378, 379. Vitamin C also prevented the rise of Sequential Organ Failure Assessment (SOFA) and Acute Physiologic Assessment and Chronic Health Evaluation (APACHE) 2 scores — used to assess severity of illness and risk of mortality — observed in placebo-treated patients 378, 379. Vitamin C infusion also lowered the concentration of markers of inflammation and endothelial injury in patients compared to placebo 378, 379. In another randomized, double-blind, controlled trial in 28 critically ill patients with septic shock, infusion of 25 mg of vitamin C per kg every six hours for 72 hours significantly limited the requirement to vasopressor norepinephrine — decreasing both the dose and duration of treatment — and dramatically improved the 28-day survival rate 380. Similar results have been reported in septic patients given intravenous vitamin C (1.5 g/6 h), hydrocortisone (50 mg/6 h), and thiamin (200 mg/12 h) until hospital discharge. Compared to standard-of-care, this intervention cocktail more than halved the mean duration of vasopressor use (18.3 h versus 54.9 h) and reduced the odds of mortality by nearly 90% 381. Although intravenous vitamin C administration appears to be safe and well tolerated, there is a non-negligible risk of oxalate nephropathy (a rare cause of kidney failure) in these critically ill patients 381.
Vitamin C Deficiency
Vitamin C deficiency causes Scurvy a disease that occurs when you have a severe and prolonged lack of vitamin C (ascorbic acid) in your diet 382, 383, 384, 385, 386, 387, 388, 389. Scurvy develops in approximately four weeks in those who consume less than 10 mg/day of Vitamin C 5. Early features of the scurvy include general weakness, fatigue, lethargy, lassitude, irritability and aching limbs. If left untreated, more serious problems can develop such as muscle weakness, swollen and bleeding gums, loosening or loss of teeth, corkscrew hairs, petechial hemorrhaging, spontaneous bruises (ecchymoses), anemia, poor wound healing, hyperkeratosis, weakness, myalgia, arthralgia, and weight loss (there can also be a paradoxical weight increase due to swelling). Shortness of breath (dyspnea), generalized edema, severe jaundice, hemolysis, acute spontaneous bleeding, neuropathy, fever and convulsions can be observed as well. In children, bone growth is impaired with vitamin c deficiency and can be associated with bleeding into the periosteum and sub-periosteum 390. Scurvy is fatal if it is not treated and sudden death has occurred as a consequence of a cerebral/myocardial hemorrhage or pneumonia 26, 387.
Vitamin C deficiency or scurvy symptoms generally develop after at least 3 months of severe or total vitamin C deficiency. Scurvy can be cured with vitamin C supplements taken by mouth. Once recovery is complete, dietary modifications to ensure the “recommended daily intake” of vitamin C is reached will prevent relapse. Except in the case of severe dental disease, permanent damage from scurvy does not usually occur 391. Smokers have greater vitamin C requirements than non-smokers, which predisposes them to scurvy 392, 393. The recommended daily allowance (RDA) of vitamin C for men is 90 mg/day and women is 75 mg/day 394, 5. If you smoke, add 35 mg/day to the above values to calculate your total daily recommended amount 5. Daily requirements increase for patients who are pregnant (80 to 85 mg/day) and during lactation (115 to 120 mg/day), or patients who smoke, are on hemodialysis, or have trauma/infection 395, 5. Consuming five varied servings of fruits and vegetables a day can provide more than 200 mg of vitamin C 5.
Scurvy is relatively rare in the United States and Canada, however, scurvy continues to be a problem in malnourished populations around the world such as impoverished, underdeveloped third world countries. In developing countries such as northern India, the incidence of vitamin C deficiency can be as high as 73.9% due to limited access to Vitamin C-rich fruit and vegetables. People who get little or no vitamin C (below about 10 mg per day) for many weeks can get scurvy 6, 387, 155, 396. The prevalence of vitamin C deficiency in the United States is 7.1% 397. Studies estimate that among people with low incomes, there is a 40% prevalence of vitamin C deficiency 398. Populations at risk include low incomes, food insecurity, poor nutrition, alcohol use disorder (alcoholic patients), isolated elderly patients, gastrointestinal disorders and malabsorption syndromes, and eating disorders or people who voluntarily restrict their type of food intake 399. In addition to poor nutritional intake, alcoholic patients develop vitamin C deficiency secondary to increased excretion of vitamin C in the urine 400. In a retrospective chart review conducted at the Mayo Clinic in Rochester, NY, and Scottsdale, AZ, from 1976 to 2002, 10 of 12 scurvy cases were related to alcohol abuse, illicit drug use, and psychiatric disorders 385. Other risk factors include fad diets, routine diets, and severe allergies to food products 401. Anorexia nervosa, Crohn disease, celiac disease, and hemodialysis patients have also been documented as having increased susceptibility to ascorbic acid deficiency (as well as other common nutritional deficiencies).
In a small study of 9 healthy middle-aged men, alcohol consumption (0.58 g/kg) produced a 47% increase in urinary ascorbic acid excretion 4 hours after ingestion 400. A small comparative study of 13 healthy men pretreated with high doses of vitamin C (2 g/d for 2 weeks) before alcohol consumption (0.8 g/kg) demonstrated a significant increase in plasma alcohol clearance when compared with a non-pretreated group 402. The literature suggests a correlation between alcohol metabolism and ascorbic acid excretion; however, the mechanism remains unknown 403. These poorly understood interactions might leave individuals who abuse alcohol at higher risk of developing scurvy.
The timeline for the development of scurvy varies, depending on vitamin C body stores, but signs can appear within 1 month of little or no vitamin C intake (below 10 mg/day) 387. Scurvy symptoms are not seen unless the total vitamin C content in the body falls below 300–400 mg 404, 405, 27, 406, 407. Initial symptoms can include fatigue (probably the result of impaired carnitine biosynthesis), malaise, and inflammation of the gums 388. As vitamin C deficiency progresses, collagen synthesis becomes impaired and connective tissues become weakened, causing petechiae, ecchymoses, purpura, joint pain, poor wound healing, hyperkeratosis, and corkscrew hairs 14. Additional signs of scurvy include depression as well as swollen, bleeding gums and loosening or loss of teeth due to tissue and capillary fragility 387. Iron deficiency anemia can also occur due to increased bleeding and decreased nonheme iron absorption secondary to low vitamin C intake 388. In children, bone disease can be present 6.
The human body lacks the ability to synthesize and make vitamin C (ascorbic acid) and therefore depends on exogenous dietary sources to meet vitamin C needs. Your body’s pool of vitamin C can be depleted in 1-3 months. Ascorbic acid (vitamin C) is prone to oxidation in your body, and your body stores are affected by environmental and lifestyle factors (e.g., smoking), biological conditions (e.g., inflammation, iron excess), and pathologic conditions (e.g., malabsorption) that may alter its oxidation. Consumption of fruits and vegetables or diets fortified with vitamin C is essential to avoid ascorbic acid deficiency 408.
Scurvy develops 1 to 3 months after initiating a vitamin C-deficient diet 409. Individuals may complain of lethargy, fatigue, malaise, emotional lability, arthralgias (joint pain), weight loss, anorexia, and diarrhea. They also may experience easy bleeding, bruising, and poor wound healing.
Your body needs vitamin C to work properly. Without enough vitamin C in your body, you can start to feel very ill. The symptoms of scurvy include:
- feeling tired and weak
- aching legs and arms
- swollen and bleeding gums
- red or blue spots on the skin, usually on the shins
- bruising easily
- wounds taking a long time to heal
Sources of vitamin C
Vitamin C is found in many different fruits and vegetables, including:
- blackcurrants
- citrus fruits – oranges, limes and lemons
- berries
- kiwifruit
- tomatoes
- broccoli
- sprouts
- red, yellow and green capsicum
Cutting and heating foods changes vitamin C and makes it less effective. So it helps to eat fruits and vegetables raw, or lightly cooked, and don’t cut them too long before eating them.
You should be able to get all the vitamin C you need from your diet.
See a doctor if you’re at risk of scurvy and you:
- feel very tired and weak all the time
- feel irritable and sad all the time
- have severe joint or leg pain
- have swollen, bleeding gums – sometimes teeth can fall out
- develop red or blue spots on the skin, usually on your shins
- have skin that bruises easily
These might be symptoms of scurvy.
Scurvy is easily treated by adding some vitamin C to your diet, such as fresh fruit and vegetables.
Your doctor may also recommend taking vitamin C supplements until you feel better. The treatment for scurvy is vitamin C supplementation. Recommendations are that 1 to 2 grams of vitamin C be administered daily for the first 2 to 3 days followed by 500 mg per day for the next week 403. Afterward, a daily intake of 100 mg of vitamin C should be given for 1 to 3 months 403.
Your doctor might arrange a blood test to confirm you have scurvy if they’re not sure.
Most people treated for scurvy feel better within 48 hours and make a full recovery within 2 weeks.
Your doctor may refer you to a specialist for treatment, support or advice. This depends on what’s causing your scurvy.
Figure 5. Corkscrew hair
Figure 6. Inflamed marginal gingiva in scurvy
Figure 7. Gingival hyperplasia due to scurvy
Footnote: Gingival hyperplasia, most prominent in the patient’s left upper gum line (arrow), is a typical sign of scurvy.
[Source 394 ]Figure 8. Perifollicular hemorrhagic papules
[Source 410 ]Figure 9. Hyperkeratotic hair follicles
[Source 411 ]Figure 10. Extensive bruising
Footnote: A 59 year old woman presented to our department with a month’s history of extensive bruising of the legs and a more recent haemarthrosis of the right knee. Full blood count, blood film, and clotting screen were normal, but she had longstanding oesophageal reflux, a very restricted diet, and undetectable serum concentration of vitamin C. Her doctor diagnosed scurvy and treated her with vitamin C. Hemorrhagic symptoms settled within two weeks, as did her gastrointestinal symptoms. She now has a more normal diet.
[Source 412 ]Figure 10. Hemarthrosis (bleeding into the joint cavity)
[Source 394 ]Vitamin C deficiency causes
You can develop scurvy if you don’t have enough vitamin C in your diet for at least 3 months, which reduces total body vitamin C stores from 1500mg to less than 300mg and the plasma vitamin C level is less than 0.2 mg/dL (less than 11 umol/L) 413. You can’t store vitamin C in your body for long, so you need to take it in regularly. Vitamin C, which is also known as ascorbic acid, is found mainly in fruit and vegetables. Unlike nearly all other animals, humans are unable to synthesize Vitamin C which must be obtained from the diet.
Good sources of vitamin C include oranges, lemons, limes, guavas and kiwi fruit. Vegetables like broccoli, cauliflower, cabbage, Asian greens and tomatoes also have a lot of vitamin C. You can find vitamin C in many other fruits and vegetables and in fresh fruit and vegetable juices. The richest source is the bush food known as the Kakadu plum, salty plum or gubinge.
You are more at risk of getting scurvy if you:
- eat a diet without many fruit and vegetables
- cook your vegetables for long periods, since this destroys vitamin C
- have a health condition that makes it difficult to digest food, like Crohn’s disease or ulcerative colitis
- are on a very restrictive diet
- have an eating disorder like anorexia nervosa
- smoke, drink a lot of alcohol, or take drugs
You can also develop scurvy if you have a bad diet and you are pregnant or breastfeeding. Young children can get scurvy too.
You can even develop scurvy if you are overweight. You might consume a lot of calories, but if you don’t eat enough fresh fruit and vegetables you still might not be getting enough vitamin C.
Signs and symptoms of vitamin C deficiency or scurvy usually develop after 1 to 3 months of insufficient vitamin C 414. Vitamin C plays an integral role in several biochemical pathways, such as collagen biosynthesis and iron absorption 403. Mature collagen is composed of three polypeptide molecules that form a triple helix. Vitamin C is needed as a cofactor in the hydroxylation of lysine and proline residues on the polypeptides to allow for the formation of the triple helix structure 415. If this reaction does not occur, the polypeptides are unstable and unable to form rigid, triple helices. This collagen abnormality leads to blood vessel fragility and poor wound healing 403. Defective collagen synthesis and poor iron absorption are responsible for scurvy’s many clinical signs and symptoms, such as easy bruising, petechiae, bleeding gums, myalgia, anemia, hemarthrosis, perifollicular hemorrhages, and corkscrew hairs 416, 417. Extracutaneous hemorrhage may also occur in muscles, bones, eyes, heart, and the nervous system 418, 419, 420, 421, resulting in hematomas, subperiosteal bleeding, fractures due to osteopenia, loosening and subsequent loss of teeth, conjunctival varicosities, retrobulbar hemorrhages, hemopericardium, cardiac tamponade, and neuropathy due to hemorrhage into nerve sheaths 419, 395, 422, 390.
Who is at risk of vitamin C deficiency?
Vitamin C deficiency is rare. Vitamin C deficiency usually arises in the setting of decreased intake or increased requirements or losses. Persons at risk for inadequate intake of vitamin include patients in the following groups:
- Find it difficult to maintain a healthy diet of fresh fruit and vegetables (e.g. elderly people, low-income households, people with an eating disorder)
- Those who smoke heavily or are dependent on alcohol or drugs
- Have a health condition that makes it difficult to digest food, such as celiac disease, ulcerative colitis or Crohn’s disease
- Those with alcoholism, anorexia, or cancer
- Eat very little food at all – possible reasons include treatments that make you feel very sick all the time (such as chemotherapy) or an eating disorder such as anorexia
- Practicing food fads – with very few or no sources of vitamin C
- Those with presumed food allergies
- Receiving unsupplemented parenteral nutrition
- Those on restricted diets secondary to inflammatory bowel disease, gastrointestinal reflux or Whipple disease
- Taking medications such as aspirin, indomethacin, oral contraceptives, tetracyclines, and corticosteroids.
- Those who have renal failure due to filtration of water-soluble vitamin C during dialysis
- Those with a complication of interleukin-2 treatment of metastatic renal cell carcinoma
- Receiving liver transplants
- Have a poor diet and are pregnant or breastfeeding – your body needs more vitamin C at these times
- Babies and young children who aren’t getting the recommended amount of vitamins
Groups at Risk of Vitamin C Deficiency
Acute vitamin C deficiency leads to scurvy 387. The timeline for the development of scurvy varies, depending on vitamin C body stores, but signs can appear within 1 month of little or no vitamin C intake (below 10 mg/day) 166. Initial symptoms can include fatigue (probably the result of impaired carnitine biosynthesis), malaise, and inflammation of the gums 388. As vitamin C deficiency progresses, collagen synthesis becomes impaired and connective tissues become weakened, causing petechiae, ecchymoses, purpura, joint pain, poor wound healing, hyperkeratosis, and corkscrew hairs 14. Additional signs of scurvy include depression as well as swollen, bleeding gums and loosening or loss of teeth due to tissue and capillary fragility 423. Iron deficiency anemia can also occur due to increased bleeding and decreased nonheme iron absorption secondary to low vitamin C intake 6. In children, bone disease can be present 6. Left untreated, scurvy is fatal 6.
Today, vitamin C deficiency and scurvy are rare in developed countries 107. Overt deficiency symptoms occur only if vitamin C intake falls below approximately 10 mg/day for many weeks 387. Vitamin C deficiency is uncommon in developed countries but can still occur in people with limited food variety.
In developed countries vitamin C deficiency affects up to 10% of women and 14% of men. Groups affected include:
- Children with very restricted diets such as may be seen with autism spectrum disorder, eating disorders, or food-fads; sickle cell disease or thalassaemia requiring blood transfusions resulting in iron overload; haemodialysis; bowel disease such as coeliac disease or Crohn disease
- Adults associated with:
- Pregnancy (due to an increased requirement)
- Psychiatric and behavioral disorders including depression, anorexia nervosa, alcoholism
- Malabsorption due to bowel disease, iron overload, or alcohol excess
- Cancer patients due to anorexia related to the malignancy, treatment, or depression.
- Elderly and others living alone, on a low income, homeless, or with poor dentition who are not eating a well-balanced diet
Smokers and passive smokers
Studies consistently show that smokers have lower plasma and leukocyte vitamin C levels than nonsmokers, due in part to increased oxidative stress 107. For this reason, the Institute of Medicine (IOM) of the National Academies concluded that smokers need 35 mg more vitamin C per day than nonsmokers 107. Exposure to secondhand smoke also decreases vitamin C levels. Although the Institute of Medicine (IOM) of the National Academies was unable to establish a specific vitamin C requirement for nonsmokers who are regularly exposed to secondhand smoke, these individuals should ensure that they meet the RDA for vitamin C 18, 107.
Infants fed evaporated or boiled milk
Most infants in developed countries are fed breastmilk and/or infant formula, both of which supply adequate amounts of vitamin C 107, 424. For many reasons, feeding infants evaporated or boiled cow’s milk is not recommended. This practice can cause vitamin C deficiency because cow’s milk naturally has very little vitamin C and heat can destroy vitamin C 33, 425.
Individuals with limited food variety
Although fruits and vegetables are the best sources of vitamin C, many other foods have small amounts of this nutrient 107. Thus, through a varied diet, most people should be able to meet the vitamin C RDA or at least obtain enough to prevent scurvy. People who have limited food variety—including some elderly, indigent individuals who prepare their own food; people who abuse alcohol or drugs; food faddists; people with mental illness; and, occasionally, children—might not obtain sufficient vitamin C 107.
People with malabsorption and certain chronic diseases
Some medical conditions can reduce the absorption of vitamin C and/or increase the amount needed by the body. People with severe intestinal malabsorption or cachexia and some cancer patients might be at increased risk of vitamin C inadequacy 426. Low vitamin C concentrations can also occur in patients with end-stage renal disease on chronic hemodialysis 427.
Vitamin C deficiency prevention
Eating a healthy, balanced diet, with plenty of fruit and vegetables, is the best way to prevent scurvy.
It’s best to steam vegetables rather than boil them. Or, you could eat vegetables raw or in a soup or stew. If you prefer to boil them, do it lightly.
Your doctor might refer you to a nutritionist, or another specialist, to help you improve your diet.
The amount of vitamin C you need each day depends on your age. Average daily recommended amounts for different ages are listed below in milligrams (mg). Intake recommendations for vitamin C and other nutrients are provided in the Dietary Reference Intakes (DRIs) developed by the Institute of Medicine 107. DRI is the general term for a set of reference values used for planning and assessing nutrient intakes of healthy people. These values, which vary by age and gender, include:
- Recommended Dietary Allowance (RDA): average daily level of intake sufficient to meet the nutrient requirements of nearly all (97%–98%) healthy individuals.
- Adequate Intake (AI): established when evidence is insufficient to develop an RDA and is set at a level assumed to ensure nutritional adequacy.
- Estimated Average Requirement (EAR): Average daily level of intake estimated to meet the requirements of 50% of healthy individuals; usually used to assess the nutrient intakes of groups of people and to plan nutritionally adequate diets for them; can also be used to assess the nutrient intakes of individuals.
- Tolerable Upper Intake Level (UL): maximum daily intake unlikely to cause adverse health effects.
Table 1 lists the current RDAs for vitamin C 107. For infants from birth to 12 months, the Institute of Medicine Food and Nutritional Board established an AI for vitamin C that is equivalent to the mean intake of vitamin C in healthy, breastfed infants.
The amount of vitamin C you need each day depends on your age. Average daily recommended amounts for different ages are listed below in milligrams (mg).
If you smoke, add 35 mg to the above values to calculate your total daily recommended amount.
Vitamin C status is typically assessed by measuring plasma vitamin C levels. Other measures, such as leukocyte vitamin C concentration, could be more accurate indicators of tissue vitamin C levels, but they are more difficult to assess and the results are not always reliable.
Table 1. Vitamin C requirement by age group
| Life Stage | Recommended Amount |
|---|---|
| Birth to 6 months* | 40 mg |
| Infants 7–12 months* | 50 mg |
| Children 1–3 years | 15 mg |
| Children 4–8 years | 25 mg |
| Children 9–13 years | 45 mg |
| Teens 14–18 years (boys) | 75 mg |
| Teens 14–18 years (girls) | 65 mg |
| Adults (men) | 90 mg |
| Adults (women) | 75 mg |
| Pregnant teens | 80 mg |
| Pregnant women | 85 mg |
| Breastfeeding teens | 115 mg |
| Breastfeeding women | 120 mg |
Footnote: If you smoke, add 35 mg to the above values to calculate your total daily recommended amount.
* Adequate Intake (intake at this level is assumed to ensure nutritional adequacy; established when evidence is insufficient to develop an RDA)
[Source 108 ]Vitamin C deficiency signs and symptoms
Other nutritional deficiencies are commonly associated with vitamin C deficiency or scurvy and clinical features may therefore be mixed and therefore confusing. Given that vitamin C and folate are often found in the same foods and that vitamin C also promotes iron absorption, patients who are deficient in vitamin C are often iron and folate-deficient 428.
Vitamin C deficiency manifests symptomatically after 8 to 12 weeks of inadequate intake and patients initially complain of weakness, fatigue, listlessness and aching limbs, especially in the legs. If left untreated, scurvy can progress to the following more severe problems. After these initial symptoms, dermatologic findings include poor wound healing, gingival swelling with loss of teeth, mucocutaneous petechiae, ecchymosis, and hyperkeratosis. Because of the disruption of disulfide bond formation both corkscrew and swan-neck hairs occur. Perifollicular hemorrhages often are localized to the lower extremities, as capillary fragility is unable to withstand the gravity-dependent hydrostatic pressure. This can result in “woody edema.” Nail findings include koilonychia and splinter hemorrhages. Beyond mucocutaneous manifestations, multiple other organ systems also are involved. Rheumatologic problems occur, including painful hemarthrosis and subperiosteal hemorrhage. This bleeding results from vascular fragility from impaired collagen formation. Osseous pathology also occurs and presents with fractures in brittle bones from disrupted endochondral bone formation. A “scorbutic rosary” at the costochondral junction and sternal depression may occur. Ocular manifestations of hemorrhage include flame hemorrhages, cotton-wool spots, and retrobulbar bleeding into optic nerves, resulting in atrophy and papilledema. The late disease may be life-threatening with anasarca, hemolysis, jaundice, and convulsions.
- Skin problems – one of the first signs of scurvy is the development of perifollicular hyperkeratotic papules (phrynoderma), often on the shins. These appear as reddish/bluish bruise-like spots surrounding hair follicles. The central hairs are twisted like corkscrews that may break easily. The papules may join together to form large areas of palpable purpura or ecchymoses (bruises).
- Oral problems – gums may swell and become red, soft and spongy. Any slight friction may cause the gums to bleed. Often this results in poor oral hygiene and dental diseases.
- Musculoskeletal problems – bleeding in the joints causes extreme discomfort and pain. Joints may be swollen and tender and the pain can be so severe that patients cannot walk.
- Eye problems – patients may complain of dryness, irritation, light intolerance, transient visual blurring and stickiness. Haemorrhaging (bleeding) beneath the conjunctiva and within the optic nerve sheath may also occur.
- Anemia – this develops in 75% of patients as a result of blood loss into tissue, altered absorptions and metabolism of iron and folate, gastrointestinal bleeding and intravascular hemolysis 429, 390.
- Heart and lung problems – shortness of breath, low blood pressure, and chest pain leading to shock and death.
The cutaneous manifestations of scurvy include phrynoderma, corkscrew hairs, perifollicular hemorrhage and purpura, edema of the lower extremities, and splinter hemorrhages. Phrynoderma, or enlarged hyperkeratotic hair follicles, initially present on the posterolateral arms. This subsequently generalizes to involve the buttocks, posterior thighs, calves, shins, and back. Corkscrew hairs represent fractured and coiled hairs due to impaired keratin cross-links by disulfide bonds. With time, significant vascular congestion occurs, particularly in the lower extremities, leading to perifollicular hemorrhage and edema. This purpura is occasionally palpable, mimicking a cutaneous vasculitis. Blood vessel wall fragility also results in splinter hemorrhages of the nail bed. Oral disease is prominent among those with pre-existing poor dentition. Individuals may develop a hemorrhagic gingivitis, where the gingiva is initially red, swollen, and shiny and later becomes purple, necrotic, and prone to bleeding. Additionally, poorly formed soft teeth are prone to infection. Musculoskeletal disease is frequently seen in children. Hemorrhage can be intramuscular, intra-articular, or subperiosteal, leading to pain and pseudoparalysis. Bowing of the long bones, depression of the sternum, and swelling of the costochondral junctions are noted on physical examination. Radiographic findings include a transverse metaphyseal radiolucent band (scurvy line or Trummerfeld zone), widening at the zone of calcification (white line of Frankel), a ring of increased density around the epiphysis (Wimberger ring) and metaphyseal spurs with marginal fractures (Pelkan spurs). Conjunctival, intraocular, intracerebral, and gastrointestinal bleeding have been reported.
Mucocutaneous features of scurvy
- Follicular hyperkeratotic papules appear first on the upper arms, often spreading to the legs and buttocks
- Perifollicular haemorrhages, purpura (which can be palpable), and ecchymoses on the legs
- Hairs are often twisted and fragile (corkscrew hairs, swan-neck hairs)
- Poor wound healing and re-opening of old healed scars
- Splinter hemorrhages in the nails
- Red, swollen gums in patients with teeth (particularly around the upper incisors) which may later become purple or black
- Bleeding from the gums
- Loosening and loss of teeth
Ocular effects of scurvy
- Eye dryness and irritation
- Subconjunctival, periorbital, or orbital hemorrhage
Musculoskeletal effects of scurvy
- Painful hemarthosis (bleeding into the joint cavity)
- Subperiosteal haemorrhage, particularly femur and proximal tibia
- Costochondral junction beading – ‘scorbutic rosary’
- Intramuscular bleeding leading to woody oedema
- Severe pain of arms and legs (the major feature of scurvy in infants) and reluctance to walk.
Vitamin C deficiency complications
The predominant morbidity associated with vitamin C deficiency or scurvy is a result of bleeding into various tissues and depends on the site of involvement 430. Subperiosteal hemorrhages cause pain and tenderness, resulting in pseudoparalysis. Loss of function at the site of the hemorrhage and anemia are typical sequelae of the hemorrhages observed in scurvy. Subperiosteal hemorrhage in the tibia and femur causes excruciating pain.
Laboratory data suggest that the neonatal brain is particularly susceptible to vitamin C deficiency and that this condition may adversely affect early brain development 431.
Until minimal daily requirements of vitamin C were supplied, scurvy plagued prolonged naval voyages and military campaigns as personnel succumbed to its devastating effects. Lethargy, fatigue, and hemorrhagic manifestations of impaired collagen synthesis affecting oral, ophthalmic, musculoskeletal, cardiac, and gastrointestinal structures and functions incapacitated or killed more people than enemy action in many cases.
Vitamin C deficiency diagnosis
The diagnosis of vitamin C deficiency or scurvy is primarily a clinical one, based on a dietary history of inadequate vitamin C intake and the signs and symptoms described above. To diagnose scurvy, your doctor will examine you and ask questions about your diet. They might also arrange a blood test to test for vitamin C. Adults vitamin C concentrations in serum should be 4-15 mg/L 432. Measurement of serum level of ascorbic acid before and after treatment, although seldom done, can confirm the diagnosis when symptoms improve or resolve within weeks 419. However, serum vitamin C measurements may not correlate well with vitamin C levels in tissue 433. Furthermore, the diagnosis of scurvy is frequently delayed or overlooked because of its rarity and can lead to unnecessary exhaustive workups 434. The differential diagnosis can be broad, encompassing other causes of hemorrhage, purpura, and joint effusion. This includes coagulation disorders, vasculitis, idiopathic thrombocytopenic purpura, rheumatoid arthritis, and septic arthritis 429.
Vitamin C deficiency or scurvy diagnosis begins with the evaluation of risk factors and a physical examination. Dermoscopy can be used to aid in diagnosis, confirming follicular purpura and corkscrew hairs with a 4 mm punch biopsy of affected areas showing similar findings by histopathology. Biopsy specimens of skin lesions often demonstrate follicular hyperkeratosis, perifollicular hemorrhage, a proliferation of blood vessels, and coiled hair follicles 419. Serum testing for low plasma vitamin C (less than 0.2 mg/dL or less than 10 μmol/L) is usually consistent with scurvy 435; however, as stated above, recent intake or supplementation may elevate plasma levels and not be reflective of a prior prolonged deficit. The level of vitamin C in leukocytes is more accurate when assessing the sparse vitamin C stores as they are less affected by acute dietary changes. A leukocyte vitamin C level of 0 mg/dL is indicative of latent scurvy. Zero to 7 mg/dL is consistent with vitamin C deficiency, and greater than 15 mg/dl is adequate.
In addition to assessing vitamin C levels, screening for concomitant other vitamin deficiencies should be undertaken. As deficiency is primarily related to poor intake, those affected also may have poor intake of other essential vitamins and minerals. Vitamin B12, folate, calcium, zinc, and iron have been notably low in this patient population. Additionally, vitamin C’s role in iron absorption cause those with scurvy to be more prone to bleeding and iron deficiency, in particular, should be assessed.
Vitamin C deficiency treatment
Direct replacement of vitamin C is standard, with up to 300 mg daily for children and 500 mg to 1000 mg daily for adults. Vitamin C may be given by intravenous (IV) infusion or taken by mouth. When given by IV infusion, vitamin C can reach much higher levels in the blood than when it is taken by mouth. The endpoint of replacement is one month or upon resolution of clinical signs and symptoms. Alternative treatment regimens for adults include 1 g to 2 g for up to 3 days followed by 500 mg daily for a week followed by 100 mg daily for up to 3 months. In addition to immediate supplementation, educate the patient on lifestyle modifications to ensure adequate intake, and recommend cessation of alcohol, and tobacco use.
In children, The American Academy of Paediatrics recommends children receive 100 mg 3 times daily for at least 1 week, followed by 100 mg daily until symptoms have resolved.
Transfusion is sometimes required for severe anemia, especially if acute related to hemorrhage.
Children with bone disease may require surgery if symptoms do not resolve with vitamin C supplements.
Identifying and treating comorbid nutritional deficiencies (eg, iron deficiency anemia, folate deficiency, other vitamin deficiencies) are integral parts of management.
As patients with scurvy are often deficient in other nutrients, close attention is needed to prevent the development of refeeding syndrome, which is a result of profound hypophosphatemia and is common in patients after prolonged starvation 436. Refeeding syndrome can produce rhabdomyolysis, hypotension, arrhythmias, seizures, and may result in multiorgan failure and death in 0.43% to 34% of these patients if untreated 437, 438. Therefore electrolytes, especially serum phosphate levels, need to be monitored at least three times a week during hospital treatment 439.
Vitamin C deficiency prognosis
Improvement of symptoms of fatigue, lethargy, pain, anorexia, and confusion usually within 24 hours of vitamin C supplementation 403, 440. Bruising, perifollicular hemorrhages, gingival bleeding, and weakness usually improve within 1 to 2 weeks 403, 440. Corkscrew hairs take up to 4 weeks to resolve, and complete resolution is usually seen after approximately 3 months of regular vitamin C supplementation 441. Bone abnormalities may require surgical intervention 440.
Vitamin C Overdose – Effects of too much Vitamin C
Vitamin C is water-soluble (dissolves in water) and so even if you eat lots of foods that are rich in vitamin C, your body will only absorb what it needs 442. Any extra vitamin C you consume in food or drink will pass naturally. Taking too much vitamin C, which only occurs if you take high-dose vitamin C supplements, can cause diarrhea, nausea, stomach cramps, headaches, fatigue and kidney stones 16, 442. However, studies evaluating the effects on urinary oxalate excretion of vitamin C intakes ranging from 30 mg to 10 g/day have had conflicting results, so it is not clear whether vitamin C actually plays a role in the development of kidney stones 443, 444, 445. The best evidence that vitamin C contributes to kidney stone formation is in patients with pre-existing hyperoxaluria 166.
In people with a condition called hemochromatosis, which causes the body to store too much iron, high doses of vitamin C might cause excess iron absorption which could worsen iron overload and damage body tissues 442, 16.
If you have diabetes, take special care not to overdose on vitamin C supplements, as too much of vitamin C may interfere with your medical tests.
In healthy individuals, high vitamin C intake does not appear to be a concern. Taking less than 1,000mg of vitamin C supplements a day is unlikely to cause any harm 442.
Taking large amounts (more than 1,000mg per day) of vitamin C can cause:
- stomach pain
- diarrhea
- flatulence
These symptoms should disappear once you stop taking vitamin C supplements.
Other reported effects of high intakes of vitamin C include reduced vitamin B12 and copper levels, accelerated metabolism or excretion of ascorbic acid, erosion of dental enamel, and allergic responses 442. However, at least some of these conclusions were a consequence of assay artifact, and additional studies have not confirmed these observations 442.
Under certain conditions, vitamin C can act as a pro-oxidant, potentially contributing to oxidative damage 442. A few studies in test tubes (in vitro) have suggested that by acting as a pro-oxidant, supplemental oral vitamin C could cause chromosomal and/or DNA damage and possibly contribute to the development of cancer 446, 447. However, other studies have not shown increased oxidative damage or increased cancer risk with high intakes of vitamin C 448.
The Food and Nutrition Board at the Institute of Medicine has established the Tolerable Upper Intake Levels (ULs) for vitamin C that apply to both food and supplement intakes (Table 3) 442. Long-term intakes of vitamin C above the Tolerable Upper Intake Level (UL) may increase the risk of adverse health effects. The Tolerable Upper Intake Levels (ULs) do not apply to individuals receiving vitamin C for medical treatment, but such individuals should be under the care of a physician 442.
Table 3. Tolerable Upper Intake Levels (ULs) for vitamin C
| Age | Male | Female | Pregnancy | Lactation |
|---|---|---|---|---|
| 0–12 months | Not possible to establish* | Not possible to establish* | ||
| 1–3 years | 400 mg | 400 mg | ||
| 4–8 years | 650 mg | 650 mg | ||
| 9–13 years | 1,200 mg | 1,200 mg | ||
| 14–18 years | 1,800 mg | 1,800 mg | 1,800 mg | 1,800 mg |
| 19+ years | 2,000 mg | 2,000 mg | 2,000 mg | 2,000 mg |
Footnotes: *Formula and food should be the only sources of vitamin C for infants.
[Source 442 ]Can vitamin C be harmful?
Taking too much vitamin C can cause diarrhea, nausea, and stomach cramps. In people with a condition called hemochromatosis, which causes the body to store too much iron, high doses of vitamin C could worsen iron overload and damage body tissues.
- Naidu KA: Vitamin C in human health and disease is still a mystery? An overview. Nutr J 2: 7, 2003. https://www.ncbi.nlm.nih.gov/pubmed/14498993[↩]
- Li Y, Schellhorn HE. New developments and novel therapeutic perspectives for vitamin C. J Nutr 2007;137:2171-84. https://www.ncbi.nlm.nih.gov/pubmed/17884994[↩]
- Institute of Medicine (US) Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington (DC): National Academies Press (US); 2000. 5, Vitamin C. Available from: https://www.ncbi.nlm.nih.gov/books/NBK225480[↩][↩][↩][↩][↩][↩]
- Jacob RA, Sotoudeh G. Vitamin C function and status in chronic disease. Nutr Clin Care 2002;5:66-74. https://www.ncbi.nlm.nih.gov/pubmed/12134712[↩]
- Vitamin C. https://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional[↩][↩][↩][↩][↩][↩][↩][↩]
- Weinstein M, Babyn P, Zlotkin S. An orange a day keeps the doctor away: scurvy in the year 2000. Pediatrics. 2001 Sep;108(3):E55. doi: 10.1542/peds.108.3.e55[↩][↩][↩][↩][↩][↩][↩][↩]
- Merck Sharp & Dohme Corp., Merck Manual. Vitamin C (Ascorbic Acid). https://www.merckmanuals.com/professional/nutritional-disorders/vitamin-deficiency,-dependency,-and-toxicity/vitamin-c[↩][↩]
- Holford P, Carr AC, Jovic TH, Ali SR, Whitaker IS, Marik PE, Smith AD. Vitamin C-An Adjunctive Therapy for Respiratory Infection, Sepsis and COVID-19. Nutrients. 2020 Dec 7;12(12):3760. doi: 10.3390/nu12123760[↩][↩]
- Carr AC, Maggini S. Vitamin C and Immune Function. Nutrients. 2017 Nov 3;9(11):1211. doi: 10.3390/nu9111211[↩][↩][↩][↩][↩]
- Hemilä H, Chalker E. Vitamin C for preventing and treating the common cold. Cochrane Database Syst Rev. 2013 Jan 31;2013(1):CD000980. doi: 10.1002/14651858.CD000980.pub4[↩][↩]
- Johnston CS. Vitamin C. In: Marriott BP, Birt DF, Stallings VA, Yates AA, eds. Present Knowledge in Nutrition 11th ed. Cambridge, MA: Elsevier; 2020:155-69.[↩][↩][↩]
- Hemilä H. Vitamin C and Infections. Nutrients. 2017 Mar 29;9(4):339. doi: 10.3390/nu9040339[↩][↩]
- Schleicher RL, Carroll MD, Ford ES, Lacher DA. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003-2004 National Health and Nutrition Examination Survey (NHANES). Am J Clin Nutr. 2009 Nov;90(5):1252-63. doi: 10.3945/ajcn.2008.27016[↩]
- Li Y, Schellhorn HE. New developments and novel therapeutic perspectives for vitamin C. J Nutr. 2007 Oct;137(10):2171-84. doi: 10.1093/jn/137.10.2171[↩][↩][↩][↩][↩]
- Frei B, England L, Ames BN. Ascorbate is an outstanding antioxidant in human blood plasma. Proc Natl Acad Sci U S A. 1989 Aug;86(16):6377-81. doi: 10.1073/pnas.86.16.6377[↩]
- Jacob RA, Sotoudeh G. Vitamin C function and status in chronic disease. Nutr Clin Care. 2002 Mar-Apr;5(2):66-74. doi: 10.1046/j.1523-5408.2002.00005.x[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Gershoff SN. Vitamin C (ascorbic acid): new roles, new requirements? Nutr Rev. 1993 Nov;51(11):313-26. doi: 10.1111/j.1753-4887.1993.tb03757.x[↩]
- Jacob RA, Sotoudeh G. Vitamin C function and status in chronic disease. Nutr Clin Care 2002;5:66-74. https://www.ncbi.nlm.nih.gov/pubmed/12134712?dopt=Abstract[↩][↩][↩][↩][↩]
- Padayatty SJ, Sun H, Wang Y, Riordan HD, Hewitt SM, Katz A, Wesley RA, Levine M. Vitamin C pharmacokinetics: implications for oral and intravenous use. Ann Intern Med 2004;140:533-7. https://www.ncbi.nlm.nih.gov/pubmed/15068981[↩][↩]
- Carpenter KJ. The history of scurvy and vitamin C. Cambridge: Cambridge University Press, 1986.[↩][↩]
- Padayatty S, Espey MG, Levine M: Vitamin C. In: Coates PM, Betz JM, Blackman MR, et al., eds.: Encyclopedia of Dietary Supplements. 2nd ed. New York, NY: Informa Healthcare, 2010, pp 821-31.[↩]
- National Cancer Institute. High-Dose Vitamin C. https://www.cancer.gov/about-cancer/treatment/cam/patient/vitamin-c-pdq[↩]
- Pauling L. The significance of the evidence about ascorbic acid and the common cold. Proc Natl Acad Sci U S A 1971;68:2678-81. https://www.ncbi.nlm.nih.gov/pubmed/4941984?dopt=Abstract[↩]
- McCormick WJ: Cancer: a collagen disease, secondary to a nutritional deficiency. Arch Pediatr 76 (4): 166-71, 1959. https://www.ncbi.nlm.nih.gov/pubmed/13638066?dopt=Abstract[↩]
- Cameron E, Pauling L: The orthomolecular treatment of cancer. I. The role of ascorbic acid in host resistance. Chem Biol Interact 9 (4): 273-83, 1974. https://www.ncbi.nlm.nih.gov/pubmed/4609626?dopt=Abstract[↩]
- Doseděl M, Jirkovský E, Macáková K, Krčmová LK, Javorská L, Pourová J, Mercolini L, Remião F, Nováková L, Mladěnka P, On Behalf Of The Oemonom. Vitamin C-Sources, Physiological Role, Kinetics, Deficiency, Use, Toxicity, and Determination. Nutrients. 2021 Feb 13;13(2):615. doi: 10.3390/nu13020615[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Padayatty S.J., Levine M. Vitamin C: The known and the unknown and Goldilocks. Oral. Dis. 2016;22:463–493. doi: 10.1111/odi.12446[↩][↩]
- National Cancer Institute. High-Dose Vitamin C–Patient Version. https://www.cancer.gov/about-cancer/treatment/cam/patient/vitamin-c-pdq#link/_5[↩]
- Li Y, Schellhorn HE. New developments and novel therapeutic perspectives for vitamin C. J Nutr 2007;137:2171-84. https://www.ncbi.nlm.nih.gov/pubmed/17884994?dopt=Abstract[↩][↩]
- Carr AC, Frei B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am J Clin Nutr 1999;69:1086-107. https://www.ncbi.nlm.nih.gov/pubmed/10357726?dopt=Abstract[↩][↩]
- Proc Natl Acad Sci U S A. 1989 Aug;86(16):6377-81. Ascorbate is an outstanding antioxidant in human blood plasma. https://www.ncbi.nlm.nih.gov/pubmed/2762330%20?dopt=Abstract[↩]
- Gershoff SN. Vitamin C (ascorbic acid): new roles, new requirements? Nutr Rev 1993;51:313-26. https://www.ncbi.nlm.nih.gov/pubmed/8108031?dopt=Abstract[↩]
- Weinstein M, Babyn P, Zlotkin S. An orange a day keeps the doctor away: scurvy in the year 2000. Pediatrics 2001;108:E55. https://www.ncbi.nlm.nih.gov/pubmed/11533373?dopt=Abstract[↩][↩]
- Wang AH, Still C. Old world meets modern: a case report of scurvy. Nutr Clin Pract 2007;22:445-8. https://www.ncbi.nlm.nih.gov/pubmed/17644699?dopt=Abstract[↩]
- Stephen R, Utecht T. Scurvy identified in the emergency department: a case report. J Emerg Med 2001;21:235-7. https://www.ncbi.nlm.nih.gov/pubmed/11604276?dopt=Abstract[↩]
- Levine M, Padayatty SJ. Vitamin C. In: Ross AC, Caballero B, Cousins RJ, Tucker KL, Ziegler TR, eds. Modern Nutrition in Health and Disease, 11th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2014:416-426.[↩]
- Camarena V, Wang G. The epigenetic role of vitamin C in health and disease. Cell Mol Life Sci. 2016 Apr;73(8):1645-58. doi: 10.1007/s00018-016-2145-x[↩]
- Cooke M.S., Evans M.D., Podmore I.D., Herbert K.E., Mistry N., Mistry P., Hickenbotham P.T., Hussieni A., Griffiths H.R., Lunec J. Novel repair action of vitamin C upon in vivo oxidative DNA damage. FEBS Lett. 1998;439:363–367. doi: 10.1016/S0014-5793(98)01403-3[↩]
- Fraga C.G., Motchnik P.A., Shigenaga M.K., Helbock H.J., Jacob R.A., Ames B.N. Ascorbic acid protects against endogenous oxidative DNA damage in human sperm. Proc. Natl. Acad. Sci. USA. 1991;88:11003–11006. doi: 10.1073/pnas.88.24.11003[↩]
- Brennan L.A., Morris G.M., Wasson G.R., Hannigan B.M., Barnett Y.A. The effect of vitamin C or vitamin E supplementation on basal and H2O2-induced DNA damage in human lymphocytes. Br. J. Nutr. 2000;84:195–202. doi: 10.1017/S0007114500001422[↩][↩][↩][↩]
- Moller P., Viscovich M., Lykkesfeldt J., Loft S., Jensen A., Poulsen H.E. Vitamin C supplementation decreases oxidative DNA damage in mononuclear blood cells of smokers. Eur. J. Nutr. 2004;43:267–274. doi: 10.1007/s00394-004-0470-6[↩]
- Moller P., Viscovich M., Lykkesfeldt J., Loft S., Jensen A., Poulsen H.E. Vitamin C supplementation decreases oxidative DNA damage in mononuclear blood cells of smokers. Eur. J. Nutr. 2004;43:267–274. doi: 10.1007/s00394-004-0470-6). In summary, vitamin C may be a promising supplement for individuals who have a predisposition for DNA damage. These studies demonstrate that the antioxidant role of vitamin C is not limited to in vitro (test tube studies) contexts, but occurs within the human body as well, providing some rationale for an anti-cancer effect. However, the evidence of this anti-cancer effect is unclear.
A number of observational studies have assessed dietary vitamin C intake and cancer risk, with mixed results ((Chen Z., Huang Y., Cao D., Qiu S., Chen B., Li J., Bao Y., Wei Q., Han P., Liu L. Vitamin C Intake and Cancers: An Umbrella Review. Front. Nutr. 2021;8:812394. doi: 10.3389/fnut.2021.812394[↩]
- Bo Y., Lu Y., Zhao Y., Zhao E., Yuan L., Lu W., Cui L., Lu Q. Association between dietary vitamin C intake and risk of esophageal cancer: A dose-response meta-analysis. Int. J. Cancer. 2016;138:1843–1850. doi: 10.1002/ijc.29838[↩][↩]
- Zhang D., Xu P., Li Y., Wei B., Yang S., Zheng Y., Lyu L., Deng Y., Zhai Z., Li N., et al. Association of vitamin C intake with breast cancer risk and mortality: A meta-analysis of observational studies. Aging. 2020;12:18415–18435. doi: 10.18632/aging.103769[↩][↩]
- Bai X.Y., Qu X., Jiang X., Xu Z., Yang Y., Su Q., Wang M., Wu H. Association between Dietary Vitamin C Intake and Risk of Prostate Cancer: A Meta-analysis Involving 103,658 Subjects. J. Cancer. 2015;6:913–921. doi: 10.7150/jca.12162[↩]
- Liu Y., Yu Q., Zhu Z., Zhang J., Chen M., Tang P., Li K. Vitamin and multiple-vitamin supplement intake and incidence of colorectal cancer: A meta-analysis of cohort studies. Med. Oncol. 2015;32:434. doi: 10.1007/s12032-014-0434-5[↩]
- Kushi L.H., Fee R.M., Sellers T.A., Zheng W., Folsom A.R. Intake of vitamins A, C, and E and postmenopausal breast cancer. The Iowa Women’s Health Study. Am. J. Epidemiol. 1996;144:165–174. doi: 10.1093/oxfordjournals.aje.a008904[↩][↩][↩]
- Ziegler R.G., Brinton L.A., Hamman R.F., Lehman H.F., Levine R.S., Mallin K., Norman S.A., Rosenthal J.F., Trumble A.C., Hoover R.N. Diet and the risk of invasive cervical cancer among white women in the United States. Am. J. Epidemiol. 1990;132:432–445. doi: 10.1093/oxfordjournals.aje.a115678[↩][↩]
- Gridley G., McLaughlin J.K., Block G., Blot W.J., Gluch M., Fraumeni J.F., Jr. Vitamin supplement use and reduced risk of oral and pharyngeal cancer. Am. J. Epidemiol. 1992;135:1083–1092. doi: 10.1093/oxfordjournals.aje.a116208[↩][↩][↩]
- Parent M.E., Richard H., Rousseau M.C., Trudeau K. Vitamin C Intake and Risk of Prostate Cancer: The Montreal PROtEuS Study. Front. Physiol. 2018;9:1218. doi: 10.3389/fphys.2018.01218[↩]
- Vissers M.C.M., Das A.B. Potential Mechanisms of Action for Vitamin C in Cancer: Reviewing the Evidence. Front. Physiol. 2018;9:809. doi: 10.3389/fphys.2018.00809[↩][↩][↩][↩][↩][↩]
- Kazmierczak-Baranska J., Boguszewska K., Adamus-Grabicka A., Karwowski B.T. Two Faces of Vitamin C-Antioxidative and Pro-Oxidative Agent. Nutrients. 2020;12:1501. doi: 10.3390/nu12051501[↩][↩]
- Didier AJ, Stiene J, Fang L, Watkins D, Dworkin LD, Creeden JF. Antioxidant and Anti-Tumor Effects of Dietary Vitamins A, C, and E. Antioxidants (Basel). 2023 Mar 3;12(3):632. doi: 10.3390/antiox12030632[↩][↩][↩][↩][↩]
- Meščić Macan A, Gazivoda Kraljević T, Raić-Malić S. Therapeutic Perspective of Vitamin C and Its Derivatives. Antioxidants (Basel). 2019 Jul 26;8(8):247. doi: 10.3390/antiox8080247[↩][↩]
- Lutsenko E.A., Carcamo J.M., Golde D.W. Vitamin C prevents DNA mutation induced by oxidative stress. J. Biol. Chem. 2002;277:16895–16899. doi: 10.1074/jbc.M201151200[↩][↩][↩][↩]
- Leekha A., Gurjar B.S., Tyagi A., Rizvi M.A., Verma A.K. Vitamin C in synergism with cisplatin induces cell death in cervical cancer cells through altered redox cycling and p53 upregulation. J. Cancer Res. Clin. Oncol. 2016;142:2503–2514. doi: 10.1007/s00432-016-2235-z[↩][↩][↩]
- Peng D., Ge G., Gong Y., Zhan Y., He S., Guan B., Li Y., Xu Z., Hao H., He Z., et al. Vitamin C increases 5-hydroxymethylcytosine level and inhibits the growth of bladder cancer. Clin. Epigenet. 2018;10:94. doi: 10.1186/s13148-018-0527-7[↩]
- Lian C.G., Xu Y., Ceol C., Wu F., Larson A., Dresser K., Xu W., Tan L., Hu Y., Zhan Q., et al. Loss of 5-hydroxymethylcytosine is an epigenetic hallmark of melanoma. Cell. 2012;150:1135–1146. doi: 10.1016/j.cell.2012.07.033[↩]
- Updated Cervical Cancer Screening Guidelines. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2021/04/updated-cervical-cancer-screening-guidelines[↩]
- Cadet J., Wagner J.R. TET enzymatic oxidation of 5-methylcytosine, 5-hydroxymethylcytosine and 5-formylcytosine. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2014;764–765:18–35. doi: 10.1016/j.mrgentox.2013.09.001[↩]
- Agathocleous M., Meacham C.E., Burgess R.J., Piskounova E., Zhao Z., Crane G.M., Cowin B.L., Bruner E., Murphy M.M., Chen W., et al. Ascorbate regulates haematopoietic stem cell function and leukaemogenesis. Nature. 2017;549:476–481. doi: 10.1038/nature23876[↩]
- Gustafson C.B., Yang C., Dickson K.M., Shao H., Van Booven D., Harbour J.W., Liu Z.J., Wang G. Epigenetic reprogramming of melanoma cells by vitamin C treatment. Clin. Epigenet. 2015;7:51. doi: 10.1186/s13148-015-0087-z[↩]
- Kim J.H., Hwang S., Lee J.H., Im S.S., Son J. Vitamin C Suppresses Pancreatic Carcinogenesis through the Inhibition of Both Glucose Metabolism and Wnt Signaling. Int. J. Mol. Sci. 2022;23:12249. doi: 10.3390/ijms232012249[↩]
- Chen Q., Espey M.G., Sun A.Y., Pooput C., Kirk K.L., Krishna M.C., Khosh D.B., Drisko J., Levine M. Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc. Natl. Acad. Sci. USA. 2008;105:11105–11109. doi: 10.1073/pnas.0804226105[↩][↩]
- Fromberg A., Gutsch D., Schulze D., Vollbracht C., Weiss G., Czubayko F., Aigner A. Ascorbate exerts anti-proliferative effects through cell cycle inhibition and sensitizes tumor cells towards cytostatic drugs. Cancer Chemother. Pharmacol. 2011;67:1157–1166. doi: 10.1007/s00280-010-1418-6[↩][↩]
- Levy R, Shriker O, Porath A, Riesenberg K, Schlaeffer F. Vitamin C for the treatment of recurrent furunculosis in patients with impaired neutrophil functions. J Infect Dis. 1996;173(6):1502-1505.[↩][↩][↩]
- Jariwalla RJ, Harakeh S. Antiviral and immunomodulatory activities of ascorbic acid. In: Harris JR, ed. Subcellular Biochemistry. Vol. 25. Ascorbic Acid: Biochemistry and Biomedical Cell Biology. New York: Plenum Press; 1996:215-231.[↩]
- Anderson R, Oosthuizen R, Maritz R, Theron A, Van Rensburg AJ. The effects of increasing weekly doses of ascorbate on certain cellular and humoral immune functions in normal volunteers. Am J Clin Nutr. 1980 Jan;33(1):71-6. doi: 10.1093/ajcn/33.1.71[↩]
- Kennes B, Dumont I, Brohee D, Hubert C, Neve P. Effect of vitamin C supplements on cell-mediated immunity in old people. Gerontology. 1983;29(5):305-10. doi: 10.1159/000213131[↩]
- Panush RS, Delafuente JC, Katz P, Johnson J. Modulation of certain immunologic responses by vitamin C. III. Potentiation of in vitro and in vivo lymphocyte responses. Int J Vitam Nutr Res Suppl. 1982;23:35-47.[↩]
- Prinz W, Bortz R, Bregin B, Hersch M. The effect of ascorbic acid supplementation on some parameters of the human immunological defence system. Int J Vitam Nutr Res. 1977;47(3):248-57.[↩]
- Vallance S. Relationships between ascorbic acid and serum proteins of the immune system. Br Med J. 1977 Aug 13;2(6084):437-8. doi: 10.1136/bmj.2.6084.437[↩]
- Anderson R, Oosthuizen R, Maritz R, Theron A, Van Rensburg AJ. The effects of increasing weekly doses of ascorbate on certain cellular and humoral immune functions in normal volunteers. Am J Clin Nutr. 1980;33(1):71-76.[↩]
- Jariwalla RJ, Harakeh S. Mechanisms underlying the action of vitamin C in viral and immunodeficiency disease. In: Packer L, Fuchs J, eds. Vitamin C in Health and Disease. New York: Macel Dekker, Inc.; 1997:309-322.[↩][↩]
- Alberts B, Bray D, Lewis J, Raff M. Differentiated cells and the maintenance of tissues. Molecular Biology of the Cell. 3rd ed. New York: Garland Publishing, Inc.; 1994:1139-1193.[↩]
- Pauling L. The immune system. How to Live Longer and Feel Better. 20th Anniversary ed. Corvallis: Oregon State University Press; 2006:105-111.[↩]
- Dahl H, Degre M. The effect of ascorbic acid on production of human interferon and the antiviral activity in vitro. Acta Pathol Microbiol Scand B. 1976;84B(5):280-284.[↩]
- Michels AJ, Frei B. Myths, artifacts, and fatal flaws: identifying limitations and opportunities in vitamin C research. Nutrients. 2013;5(12):5161-5192.[↩]
- Johnston CS. Vitamin C. In: Erdman JWJ, Macdonald IA, Zeisel SH, eds. Present Knowledge in Nutrition. 10th ed. Ames, Iowa: Wiley-Blackwell; 2012:248-260.[↩]
- Levine M., Rumsey S., Daruwala R., Park J., Wang Y. Criteria and recommendations for vitamin C intake. JAMA. 1999;281:1415–1423. doi: 10.1001/jama.281.15.1415[↩]
- Padayatty S.J., Katz A., Wang Y., Eck P., Kwon O., Lee J.H., Chen S., Corpe C., Dutta A., Dutta S.K., et al. Vitamin C as an antioxidant: Evaluation of its role in disease prevention. J. Am. Coll. Nutr. 2003;22:18–35. doi: 10.1080/07315724.2003.10719272[↩]
- McDonough M., Loenarz C., Chowdhury R., Clifton I., Schofield C. Structural studies on human 2-oxoglutarate dependent oxygenases. Curr. Opin. Struct. Biol. 2010;20:659–672. doi: 10.1016/j.sbi.2010.08.006[↩][↩]
- Kuiper C., Vissers M.C. Ascorbate as a co-factor for fe- and 2-oxoglutarate dependent dioxygenases: Physiological activity in tumor growth and progression. Front. Oncol. 2014;4:359. doi: 10.3389/fonc.2014.00359[↩][↩]
- Loenarz C., Schofield C.J. Physiological and biochemical aspects of hydroxylations and demethylations catalyzed by human 2-oxoglutarate oxygenases. Trends Biochem. Sci. 2011;36:7–18. doi: 10.1016/j.tibs.2010.07.002[↩]
- Myllyla R., Kuutti-Savolainen E.R., Kivirikko K.I. The role of ascorbate in the prolyl hydroxylase reaction. Biochem. Biophys. Res. Commun. 1978;83:441–448. doi: 10.1016/0006-291X(78)91010-0[↩]
- Islam M.S., Leissing T., Chowdhury R., Hopkinson R., Schofield C. 2-Oxoglutarate-dependent oxygenases. Annu. Rev. Biochem. 2018;87 doi: 10.1146/annurev-biochem-061516-044724[↩][↩][↩][↩]
- May J.M., Harrison F.E. Role of vitamin C in the function of the vascular endothelium. Antioxid. Redox Sign. 2013;19:2068–2083. doi: 10.1089/ars.2013.5205[↩]
- Young J.I., Zuchner S., Wang G. Regulation of the epigenome by vitamin C. Annu. Rev. Nutr. 2015;35:545–564. doi: 10.1146/annurev-nutr-071714-034228[↩][↩]
- Cimmino L., Neel B.G., Aifantis I. Vitamin C in stem cell reprogramming and cancer. Trends Cell Biol. 2018;28:698–708. doi: 10.1016/j.tcb.2018.04.001[↩][↩][↩]
- Amer J., Zelig O., Fibach E. Oxidative status of red blood cells, neutrophils, and platelets in paroxysmal nocturnal hemoglobinuria. Exp. Hematol. 2008;36:369–377. doi: 10.1016/j.exphem.2007.12.003[↩]
- Furusawa H., Sato Y., Tanaka Y., Inai Y., Amano A., Iwama M., Kondo Y., Handa S., Murata A., Nishikimi M., et al. Vitamin C is not essential for carnitine biosynthesis in vivo: Verification in vitamin C-depleted senescence marker protein-30/gluconolactonase knockout mice. Biol. Pharm. Bull. 2008;31:1673–1679. doi: 10.1248/bpb.31.1673[↩]
- oung J.I., Zuchner S., Wang G. Regulation of the epigenome by vitamin C. Annu. Rev. Nutr. 2015;35:545–564. doi: 10.1146/annurev-nutr-071714-034228[↩]
- Monfort A., Wutz A. Breathing-in epigenetic change with vitamin C. EMBO Rep. 2013;14:337–346. doi: 10.1038/embor.2013.29[↩][↩]
- Das A.B., Smith-Diaz C.C., Vissers M.C.M. Emerging epigenetic therapeutics for myeloid leukemia: Modulating demethylase activity with ascorbate. Haematologica. 2020;106 doi: 10.3324/haematol.2020.259283[↩]
- Lee Chong T., Ahearn E.L., Cimmino L. Reprogramming the epigenome with vitamin C. Front. Cell Dev. Biol. 2019;7:128. doi: 10.3389/fcell.2019.00128[↩]
- Ozer A., Bruick R.K. Non-heme dioxygenases: Cellular sensors and regulators jelly rolled into one? Nat. Chem. Biol. 2007;3:144–153. doi: 10.1038/nchembio863[↩]
- Kuiper C., Dachs G.U., Currie M.J., Vissers M.C. Intracellular ascorbate enhances hypoxia-inducible factor (HIF)-hydroxylase activity and preferentially suppresses the HIF-1 transcriptional response. Free Radic. Biol. Med. 2014;69:308–317. doi: 10.1016/j.freeradbiomed.2014.01.033[↩]
- Wang T., Chen K., Zeng X., Yang J., Wu Y., Shi X., Qin B., Zeng L., Esteban M.A., Pan G., et al. The histone demethylases Jhdm1a/1b enhance somatic cell reprogramming in a vitamin-C-dependent manner. Cell Stem. Cell. 2011;9:575–587. doi: 10.1016/j.stem.2011.10.005[↩]
- Zhang T., Huang K., Zhu Y., Wang T., Shan Y., Long B., Li Y., Chen Q., Wang P., Zhao S., et al. Vitamin C-dependent lysine demethylase 6 (KDM6)-mediated demethylation promotes a chromatin state that supports the endothelial-to-hematopoietic transition. J. Biol. Chem. 2019;294:13657–13670. doi: 10.1074/jbc.RA119.009757[↩]
- D’Oto A., Tian Q.W., Davidoff A.M., Yang J. Histone demethylases and their roles in cancer epigenetics. J. Med. Oncol. Ther. 2016;1:34–40. doi: 10.35841/medical-oncology.1.2.34-40[↩][↩]
- Blaschke K., Ebata K.T., Karimi M.M., Zepeda-Martinez J.A., Goyal P., Mahapatra S., Tam A., Laird D.J., Hirst M., Rao A., et al. Vitamin C induces Tet-dependent DNA demethylation and a blastocyst-like state in ES cells. Nature. 2013;500:222–226. doi: 10.1038/nature12362[↩]
- Chen J., Guo L., Zhang L., Wu H., Yang J., Liu H., Wang X., Hu X., Gu T., Zhou Z., et al. Vitamin C modulates TET1 function during somatic cell reprogramming. Nat. Genet. 2013;45:1504–1509. doi: 10.1038/ng.2807[↩]
- Minor E.A., Court B.L., Young J.I., Wang G. Ascorbate induces ten-eleven translocation (Tet) methylcytosine dioxygenase-mediated generation of 5-hydroxymethylcytosine. J. Biol Chem. 2013;288:13669–13674. doi: 10.1074/jbc.C113.464800[↩]
- Rasmussen K.D., Helin K. Role of TET enzymes in DNA methylation, development, and cancer. Genes Dev. 2016;30:733–750. doi: 10.1101/gad.276568.115[↩][↩]
- Englard S., Seifter S. The biochemical functions of ascorbic acid. Annu. Rev. Nutr. 1986;6:365–406. doi: 10.1146/annurev.nu.06.070186.002053[↩]
- Moran G.R. 4-Hydroxyphenylpyruvate dioxygenase. Arch. Biochem. Biophys. 2005;433:117–128. doi: 10.1016/j.abb.2004.08.015[↩]
- Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: National Academy Press, 2000. https://www.nap.edu/catalog/9810/dietary-reference-intakes-for-vitamin-c-vitamin-e-selenium-and-carotenoids[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Vitamin C. https://ods.od.nih.gov/factsheets/VitaminC-Consumer/[↩][↩]
- Bates CJ. Bioavailability of vitamin C. Eur J Clin Nutr. 1997 Jan;51 Suppl 1:S28-33.[↩]
- Mangels AR, Block G, Frey CM, Patterson BH, Taylor PR, Norkus EP, Levander OA. The bioavailability to humans of ascorbic acid from oranges, orange juice and cooked broccoli is similar to that of synthetic ascorbic acid. J Nutr. 1993 Jun;123(6):1054-61. doi: 10.1093/jn/123.6.1054[↩]
- Gregory JF 3rd. Ascorbic acid bioavailability in foods and supplements. Nutr Rev. 1993 Oct;51(10):301-3. doi: 10.1111/j.1753-4887.1993.tb03059.x[↩]
- Johnston CS, Luo B. Comparison of the absorption and excretion of three commercially available sources of vitamin C. J Am Diet Assoc. 1994 Jul;94(7):779-81. doi: 10.1016/0002-8223(94)91950-x[↩][↩]
- Moyad MA, Combs MA, Vrablic AS, Velasquez J, Turner B, Bernal S. Vitamin C metabolites, independent of smoking status, significantly enhance leukocyte, but not plasma ascorbate concentrations. Adv Ther. 2008 Oct;25(10):995-1009. doi: 10.1007/s12325-008-0106-y[↩]
- Pelletier, O. & Keith, M.O. Bioavailability of synthetic and natural ascorbic acid. Journal of the American Dietetic Association. 1974; 64: 271-275.[↩]
- Mangels, A.R. et al. The bioavailability to humans of ascorbic acid from oranges, orange juice, and cooked broccoli is similar to that of synthetic ascorbic acid. Journal of Nutrition. 1993; volume 123: pages 1054-1061.[↩]
- Yung S, Mayersohn M, Robinson JB. Ascorbic acid absorption in humans: a comparison among several dosage forms. J Pharm Sci. 1982 Mar;71(3):282-5. doi: 10.1002/jps.2600710304[↩]
- Nyyssonen, K. et al. Effect of supplementation of smoking men with plain or slow release ascorbic acid on lipoprotein oxidation. European Journal of Clinical Nutrition. 1997; volume 51: pages 154-163.[↩]
- Viscovich M, Lykkesfeldt J, Poulsen HE. Vitamin C pharmacokinetics of plain and slow release formulations in smokers. Clinical nutrition. 2004;23(5):1043-1050.[↩]
- Carr AC, Vissers MC. Synthetic or food-derived vitamin C–are they equally bioavailable? Nutrients. 2013 Oct 28;5(11):4284-304. doi: 10.3390/nu5114284[↩]
- Vinson, J.A. & Bose, P. Comparative bioavailability to humans of ascorbic acid alone or in a citrus extract. American Journal of Clinical Nutrition. 1988; volume 48: pages 501-604.[↩]
- Carr AC, Vissers MC. Synthetic or food-derived vitamin C-are they equally bioavailable? Nutrients. 2013;5(11):4284-4304.[↩][↩]
- Uchida E, Kondo Y, Amano A, et al. Absorption and excretion of ascorbic acid alone and in acerola (Malpighia emarginata) juice: comparison in healthy Japanese subjects. Biol Pharm Bull. 2011;34(11):1744-1747.[↩]
- Johnston, C.S. & Luo, B. Comparison of the absorption and excretion of three commercially available sources of vitamin C. Journal of the American Dietetic Association. 1994; volume 94: pages 779-781.[↩]
- Cort, W.M. Antioxidant activity of tocopherols, ascorbyl palmitate, and ascorbic acid and their mode of action. Journal of the American Oil Chemists’ Society. 1974; volume 51: pages 321-325.[↩]
- Ross, D. et al. Ascorbate 6-palmitate protects human erythrocytes from oxidative damage. Free Radical Biology and Medicine. 1999; volume 26: pages 81-89.[↩]
- DeRitter, E. et al. Physiologic availability of dehydro-L-ascorbic acid and palmitoyl-L-ascorbic acid. Science. 1951; volume 113: pages 628-631.[↩]
- Austria R. et al. Stability of vitamin C derivatives in solution and in topical formulations. Journal of Pharmacology and Biomedical Analysis. 1997; volume 15: pages 795-801.[↩]
- Sauberlich, H.E. et al. Effects of erythorbic acid on vitamin C metabolism in young women. American Journal of Clinical Nutrition. 1996; volume 64: pages 336-346.[↩]
- Weeks BS, Perez PP. Absorption rates and free radical scavenging values of vitamin C-lipid metabolites in human lymphoblastic cells. Med Sci Monit. 2007;13(10):BR205-210.[↩]
- Pancorbo D, Vazquez C, Fletcher MA. Vitamin C-lipid metabolites: uptake and retention and effect on plasma C-reactive protein and oxidized LDL levels in healthy volunteers. Med Sci Monit. 2008;14(11):CR547-551.[↩]
- Davis JL, Paris HL, Beals JW, et al. Liposomal-encapsulated ascorbic scid: influence on vitamin C bioavailability and capacity to protect against ischemia-reperfusion injury. Nutr Metab Insights. 2016;9:25-30.[↩]
- Seifried HE, Anderson DE, Sorkin BC, Costello RB. Free radicals: the pros and cons of antioxidants. Executive summary report. J Nutr. 2004 Nov;134(11):3143S-3163S. doi: 10.1093/jn/134.11.3143S[↩]
- Block KI, Koch AC, Mead MN, Tothy PK, Newman RA, Gyllenhaal C. Impact of antioxidant supplementation on chemotherapeutic efficacy: a systematic review of the evidence from randomized controlled trials. Cancer Treat Rev. 2007 Aug;33(5):407-18. doi: 10.1016/j.ctrv.2007.01.005[↩]
- Lawenda BD, Kelly KM, Ladas EJ, Sagar SM, Vickers A, Blumberg JB. Should supplemental antioxidant administration be avoided during chemotherapy and radiation therapy? J Natl Cancer Inst. 2008 Jun 4;100(11):773-83. doi: 10.1093/jnci/djn148[↩][↩]
- Heaney ML, Gardner JR, Karasavvas N, Golde DW, Scheinberg DA, Smith EA, O’Connor OA. Vitamin C antagonizes the cytotoxic effects of antineoplastic drugs. Cancer Res. 2008 Oct 1;68(19):8031-8. doi: 10.1158/0008-5472.CAN-08-1490[↩]
- Levine M, Espey MG, Chen Q. Losing and finding a way at C: new promise for pharmacologic ascorbate in cancer treatment. Free Radic Biol Med. 2009 Jul 1;47(1):27-9. doi: 10.1016/j.freeradbiomed.2009.04.001[↩]
- Prasad KN. Rationale for using high-dose multiple dietary antioxidants as an adjunct to radiation therapy and chemotherapy. J Nutr. 2004 Nov;134(11):3182S-3S. doi: 10.1093/jn/134.11.3182S[↩]
- Brown BG, Zhao XQ, Chait A, Fisher LD, Cheung MC, Morse JS, Dowdy AA, Marino EK, Bolson EL, Alaupovic P, Frohlich J, Albers JJ. Simvastatin and niacin, antioxidant vitamins, or the combination for the prevention of coronary disease. N Engl J Med. 2001 Nov 29;345(22):1583-92. doi: 10.1056/NEJMoa011090[↩]
- Cheung MC, Zhao XQ, Chait A, Albers JJ, Brown BG. Antioxidant supplements block the response of HDL to simvastatin-niacin therapy in patients with coronary artery disease and low HDL. Arterioscler Thromb Vasc Biol. 2001 Aug;21(8):1320-6. doi: 10.1161/hq0801.095151[↩]
- Vitamin C pharmacokinetics: implications for oral and intravenous use. Padayatty SJ, Sun H, Wang Y, Riordan HD, Hewitt SM, Katz A, Wesley RA, Levine M. Ann Intern Med. 2004 Apr 6; 140(7):533-7. https://www.ncbi.nlm.nih.gov/pubmed/15068981/[↩]
- Vitamin C: a concentration-function approach yields pharmacology and therapeutic discoveries. Levine M, Padayatty SJ, Espey MG. Adv Nutr. 2011 Mar; 2(2):78-88. https://www.ncbi.nlm.nih.gov/pubmed/22332036/[↩]
- Emerging research and clinical development trends of liposome and lipid nanoparticle drug delivery systems. Kraft JC, Freeling JP, Wang Z, Ho RJ. J Pharm Sci. 2014 Jan; 103(1):29-52. https://www.ncbi.nlm.nih.gov/pubmed/24338748/[↩][↩]
- The potential of liposomes in oral drug delivery. Rogers JA, Anderson KE. Crit Rev Ther Drug Carrier Syst. 1998; 15(5):421-80. https://www.ncbi.nlm.nih.gov/pubmed/9822867/[↩]
- Nutr Metab Insights. 2016; 9: 25–30. Published online 2016 Jun 20. doi: 10.4137/NMI.S39764. Liposomal-encapsulated Ascorbic Acid: Influence on Vitamin C Bioavailability and Capacity to Protect Against Ischemia–Reperfusion Injury. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4915787/[↩]
- Int J Nanomedicine. 2013; 8: 3573–3585. Published online 2013 Sep 23. doi: 10.2147/IJN.S47745. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3790897/[↩]
- Clin Cosmet Investig Dermatol. 2015; 8: 591–599. Published online 2015 Dec 17. doi: 10.2147/CCID.S90781. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4687614/[↩]
- Carr AC, Frei B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am J Clin Nutr. 1999 Jun;69(6):1086-107. doi: 10.1093/ajcn/69.6.1086[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Bertoia M, Albanes D, Mayne ST, Männistö S, Virtamo J, Wright ME. No association between fruit, vegetables, antioxidant nutrients and risk of renal cell carcinoma. Int J Cancer. 2010 Mar 15;126(6):1504-12. doi: 10.1002/ijc.24829[↩]
- Heinen MM, Verhage BA, Goldbohm RA, van den Brandt PA. Intake of vegetables, fruits, carotenoids and vitamins C and E and pancreatic cancer risk in The Netherlands Cohort Study. Int J Cancer. 2012 Jan 1;130(1):147-58. doi: 10.1002/ijc.25989[↩]
- Roswall N, Olsen A, Christensen J, Dragsted LO, Overvad K, Tjønneland A. Micronutrient intake and risk of urothelial carcinoma in a prospective Danish cohort. Eur Urol. 2009 Nov;56(5):764-70. doi: 10.1016/j.eururo.2009.06.030[↩]
- Hecht SS. Approaches to cancer prevention based on an understanding of N-nitrosamine carcinogenesis. Proc Soc Exp Biol Med. 1997 Nov;216(2):181-91. doi: 10.3181/00379727-216-44168[↩]
- Goodman M, Bostick RM, Kucuk O, Jones DP. Clinical trials of antioxidants as cancer prevention agents: past, present, and future. Free Radic Biol Med. 2011 Sep 1;51(5):1068-84. doi: 10.1016/j.freeradbiomed.2011.05.018[↩]
- Zhang S, Hunter DJ, Forman MR, Rosner BA, Speizer FE, Colditz GA, Manson JE, Hankinson SE, Willett WC. Dietary carotenoids and vitamins A, C, and E and risk of breast cancer. J Natl Cancer Inst. 1999 Mar 17;91(6):547-56. doi: 10.1093/jnci/91.6.547[↩][↩]
- Kushi LH, Fee RM, Sellers TA, Zheng W, Folsom AR. Intake of vitamins A, C, and E and postmenopausal breast cancer. The Iowa Women’s Health Study. Am J Epidemiol. 1996 Jul 15;144(2):165-74. doi: 10.1093/oxfordjournals.aje.a008904[↩][↩]
- Levine M, Conry-Cantilena C, Wang Y, Welch RW, Washko PW, Dhariwal KR, Park JB, Lazarev A, Graumlich JF, King J, Cantilena LR. Vitamin C pharmacokinetics in healthy volunteers: evidence for a recommended dietary allowance. Proc Natl Acad Sci U S A. 1996 Apr 16;93(8):3704-9. doi: 10.1073/pnas.93.8.3704[↩][↩][↩][↩]
- Levine M, Wang Y, Padayatty SJ, Morrow J. A new recommended dietary allowance of vitamin C for healthy young women. Proc Natl Acad Sci U S A. 2001 Aug 14;98(17):9842-6. doi: 10.1073/pnas.171318198[↩]
- Hercberg S, Galan P, Preziosi P, Bertrais S, Mennen L, Malvy D, Roussel AM, Favier A, Briançon S. The SU.VI.MAX Study: a randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch Intern Med. 2004 Nov 22;164(21):2335-42. doi: 10.1001/archinte.164.21.2335. Erratum in: Arch Intern Med. 2005 Feb 14;165(3):286.[↩][↩][↩]
- Galan P, Briançon S, Favier A, Bertrais S, Preziosi P, Faure H, Arnaud J, Arnault N, Czernichow S, Mennen L, Hercberg S. Antioxidant status and risk of cancer in the SU.VI.MAX study: is the effect of supplementation dependent on baseline levels? Br J Nutr. 2005 Jul;94(1):125-32. doi: 10.1079/bjn20051462[↩]
- Gaziano JM, Glynn RJ, Christen WG, Kurth T, Belanger C, MacFadyen J, Bubes V, Manson JE, Sesso HD, Buring JE. Vitamins E and C in the prevention of prostate and total cancer in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2009 Jan 7;301(1):52-62. doi: 10.1001/jama.2008.862[↩]
- Lin J, Cook NR, Albert C, Zaharris E, Gaziano JM, Van Denburgh M, Buring JE, Manson JE. Vitamins C and E and beta carotene supplementation and cancer risk: a randomized controlled trial. J Natl Cancer Inst. 2009 Jan 7;101(1):14-23. doi: 10.1093/jnci/djn438[↩]
- Taylor PR, Li B, Dawsey SM, Li JY, Yang CS, Guo W, Blot WJ. Prevention of esophageal cancer: the nutrition intervention trials in Linxian, China. Linxian Nutrition Intervention Trials Study Group. Cancer Res. 1994 Apr 1;54(7 Suppl):2029s-2031s.[↩][↩]
- Qiao YL, Dawsey SM, Kamangar F, Fan JH, Abnet CC, Sun XD, Johnson LL, Gail MH, Dong ZW, Yu B, Mark SD, Taylor PR. Total and cancer mortality after supplementation with vitamins and minerals: follow-up of the Linxian General Population Nutrition Intervention Trial. J Natl Cancer Inst. 2009 Apr 1;101(7):507-18. doi: 10.1093/jnci/djp037. Epub 2009 Mar 24. Erratum in: J Natl Cancer Inst. 2010 Jan 20;102(2):140.[↩][↩][↩][↩]
- Bjelakovic G, Nikolova D, Simonetti RG, Gluud C. Antioxidant supplements for preventing gastrointestinal cancers. Cochrane Database Syst Rev. 2004 Oct 18;(4):CD004183. doi: 10.1002/14651858.CD004183.pub2. Update in: Cochrane Database Syst Rev. 2008;(3):CD004183.[↩]
- Shekelle P, Hardy ML, Coulter I, et al. Effect of the Supplemental Use of Antioxidants Vitamin C, Vitamin E, and Coenzyme Q10 for the Prevention and Treatment of Cancer: Summary. 2003 Oct. In: AHRQ Evidence Report Summaries. Rockville (MD): Agency for Healthcare Research and Quality (US); 1998-2005. 75. Available from: https://www.ncbi.nlm.nih.gov/books/NBK11931[↩]
- Padayatty SJ, Sun H, Wang Y, Riordan HD, Hewitt SM, Katz A, Wesley RA, Levine M. Vitamin C pharmacokinetics: implications for oral and intravenous use. Ann Intern Med. 2004 Apr 6;140(7):533-7. doi: 10.7326/0003-4819-140-7-200404060-00010[↩][↩][↩]
- Levine M, Rumsey SC, Daruwala R, Park JB, Wang Y. Criteria and recommendations for vitamin C intake. JAMA. 1999 Apr 21;281(15):1415-23. doi: 10.1001/jama.281.15.1415[↩][↩][↩]
- Padayatty SJ, Levine M. Vitamins C and E and the prevention of preeclampsia. N Engl J Med. 2006 Sep 7;355(10):1065; author reply 1066. doi: 10.1056/NEJMc061414[↩]
- Padayatty SJ, Levine M. Antioxidant supplements and cardiovascular disease in men. JAMA. 2009 Apr 1;301(13):1336; author reply 1336-7. doi: 10.1001/jama.2009.316[↩][↩]
- Michels, K.B., Holmberg, L., Bergkvist, L., Ljung, H., Bruce, Å. and Wolk, A. (2001), Dietary antioxidant vitamins, retinol, and breast cancer incidence in a cohort of Swedish women. Int. J. Cancer, 91: 563-567. https://doi.org/10.1002/1097-0215(200002)9999:9999<::AID-IJC1079>3.0.CO;2-9[↩]
- Hutchinson J, Lentjes MA, Greenwood DC, Burley VJ, Cade JE, Cleghorn CL, Threapleton DE, Key TJ, Cairns BJ, Keogh RH, Dahm CC, Brunner EJ, Shipley MJ, Kuh D, Mishra G, Stephen AM, Bhaniani A, Borgulya G, Khaw KT. Vitamin C intake from diary recordings and risk of breast cancer in the UK Dietary Cohort Consortium. Eur J Clin Nutr. 2012 May;66(5):561-8. doi: 10.1038/ejcn.2011.197[↩]
- Nagel G, Linseisen J, van Gils CH, et al. Dietary beta-carotene, vitamin C and E intake and breast cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC). Breast Cancer Res Treat. 2010 Feb;119(3):753-65. doi: 10.1007/s10549-009-0444-8[↩]
- Roswall N, Olsen A, Christensen J, Dragsted LO, Overvad K, Tjønneland A. Micronutrient intake and breast cancer characteristics among postmenopausal women. Eur J Cancer Prev. 2010 Sep;19(5):360-5. doi: 10.1097/cej.0b013e32833ade68[↩]
- Liu C, Russell RM. Nutrition and gastric cancer risk: an update. Nutr Rev. 2008 May;66(5):237-49. doi: 10.1111/j.1753-4887.2008.00029.x[↩]
- Mirvish SS, Wallcave L, Eagen M, Shubik P. Ascorbate-nitrite reaction: possible means of blocking the formation of carcinogenic N-nitroso compounds. Science. 1972 Jul 7;177(4043):65-8. doi: 10.1126/science.177.4043.65[↩]
- Tsugane S, Sasazuki S. Diet and the risk of gastric cancer: review of epidemiological evidence. Gastric Cancer. 2007;10(2):75-83. doi: 10.1007/s10120-007-0420-0[↩]
- Jenab M, Riboli E, Ferrari P, et al. Plasma and dietary vitamin C levels and risk of gastric cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC-EURGAST). Carcinogenesis. 2006 Nov;27(11):2250-7. doi: 10.1093/carcin/bgl096[↩]
- Banerjee S, Hawksby C, Miller S, Dahill S, Beattie AD, McColl KE. Effect of Helicobacter pylori and its eradication on gastric juice ascorbic acid. Gut. 1994 Mar;35(3):317-22. doi: 10.1136/gut.35.3.317[↩]
- Zhang ZW, Patchett SE, Perrett D, Katelaris PH, Domizio P, Farthing MJ. The relation between gastric vitamin C concentrations, mucosal histology, and CagA seropositivity in the human stomach. Gut. 1998 Sep;43(3):322-6. doi: 10.1136/gut.43.3.322[↩]
- Food and Nutrition Board, Institute of Medicine. Vitamin C. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, D.C.: National Academy Press; 2000:95-185. https://nap.nationalacademies.org/read/9810/chapter/7[↩]
- Chuang CH, Sheu BS, Kao AW, Cheng HC, Huang AH, Yang HB, Wu JJ. Adjuvant effect of vitamin C on omeprazole-amoxicillin-clarithromycin triple therapy for Helicobacter pylori eradication. Hepatogastroenterology. 2007 Jan-Feb;54(73):320-4.[↩]
- Krajewska B, Brindell M. Urease activity and L-ascorbic acid. J Enzyme Inhib Med Chem. 2011 Jun;26(3):309-18. doi: 10.3109/14756366.2010.504675[↩]
- Pal J, Sanal MG, Gopal GJ. Vitamin-C as anti-Helicobacter pylori agent: More prophylactic than curative- Critical review. Indian J Pharmacol. 2011 Nov;43(6):624-7. doi: 10.4103/0253-7613.89814[↩]
- Park Y, Spiegelman D, Hunter DJ, et al. Intakes of vitamins A, C, and E and use of multiple vitamin supplements and risk of colon cancer: a pooled analysis of prospective cohort studies. Cancer Causes Control. 2010 Nov;21(11):1745-57. doi: 10.1007/s10552-010-9549-y[↩]
- Thompson CA, Cerhan JR. Fruit and vegetable intake and survival from non-Hodgkin lymphoma: does an apple a day keep the doctor away? Leuk Lymphoma. 2010 Jun;51(6):963-4. doi: 10.3109/10428194.2010.483305[↩]
- Kabat GC, Kim MY, Wactawski-Wende J, Shikany JM, Vitolins MZ, Rohan TE. Intake of antioxidant nutrients and risk of non-Hodgkin’s Lymphoma in the Women’s Health Initiative. Nutr Cancer. 2012;64(2):245-54. doi: 10.1080/01635581.2012.642454[↩]
- Wang L, Sesso HD, Glynn RJ, Christen WG, Bubes V, Manson JE, Buring JE, Gaziano JM. Vitamin E and C supplementation and risk of cancer in men: posttrial follow-up in the Physicians’ Health Study II randomized trial. Am J Clin Nutr. 2014 Sep;100(3):915-23. doi: 10.3945/ajcn.114.085480[↩][↩]
- National Cancer Institute. High-Dose Vitamin C – Health Professional Version. https://www.cancer.gov/about-cancer/treatment/cam/hp/vitamin-c-pdq#section/_14[↩]
- Intravenous Vitamin C (PDQ®)–Patient Version. https://www.cancer.gov/about-cancer/treatment/cam/patient/vitamin-c-pdq[↩]
- National Cancer Institute. High-Dose Vitamin C –Health Professional Version. https://www.cancer.gov/about-cancer/treatment/cam/hp/vitamin-c-pdq[↩]
- Cameron E, Campbell A. The orthomolecular treatment of cancer. II. Clinical trial of high-dose ascorbic acid supplements in advanced human cancer. Chem Biol Interact. 1974 Oct;9(4):285-315. doi: 10.1016/0009-2797(74)90019-2[↩][↩][↩][↩]
- Cameron E, Pauling L. Supplemental ascorbate in the supportive treatment of cancer: Prolongation of survival times in terminal human cancer. Proc Natl Acad Sci U S A. 1976 Oct;73(10):3685-9. doi: 10.1073/pnas.73.10.3685[↩][↩]
- Cameron E, Pauling L. Supplemental ascorbate in the supportive treatment of cancer: reevaluation of prolongation of survival times in terminal human cancer. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4538-42. doi: 10.1073/pnas.75.9.4538[↩][↩]
- Creagan ET, Moertel CG, O’Fallon JR, Schutt AJ, O’Connell MJ, Rubin J, Frytak S. Failure of high-dose vitamin C (ascorbic acid) therapy to benefit patients with advanced cancer. A controlled trial. N Engl J Med. 1979 Sep 27;301(13):687-90. doi: 10.1056/NEJM197909273011303[↩][↩][↩]
- Moertel CG, Fleming TR, Creagan ET, Rubin J, O’Connell MJ, Ames MM. High-dose vitamin C versus placebo in the treatment of patients with advanced cancer who have had no prior chemotherapy. A randomized double-blind comparison. N Engl J Med. 1985 Jan 17;312(3):137-41. doi: 10.1056/NEJM198501173120301[↩][↩][↩]
- Hoffer LJ, Levine M, Assouline S, Melnychuk D, Padayatty SJ, Rosadiuk K, Rousseau C, Robitaille L, Miller WH Jr. Phase I clinical trial of i.v. ascorbic acid in advanced malignancy. Ann Oncol. 2008 Nov;19(11):1969-74. doi: 10.1093/annonc/mdn377. Epub 2008 Jun 9. Erratum in: Ann Oncol. 2008 Dec;19(12):2095.[↩][↩]
- Verrax J, Calderon PB. Pharmacologic concentrations of ascorbate are achieved by parenteral administration and exhibit antitumoral effects. Free Radic Biol Med. 2009 Jul 1;47(1):32-40. doi: 10.1016/j.freeradbiomed.2009.02.016[↩]
- Cameron E, Campbell A, Jack T. The orthomolecular treatment of cancer. III. Reticulum cell sarcoma: double complete regression induced by high-dose ascorbic acid therapy. Chem Biol Interact. 1975 Nov;11(5):387-93. doi: 10.1016/0009-2797(75)90007-1[↩]
- Padayatty SJ, Riordan HD, Hewitt SM, Katz A, Hoffer LJ, Levine M. Intravenously administered vitamin C as cancer therapy: three cases. CMAJ. 2006 Mar 28;174(7):937-42. doi: 10.1503/cmaj.050346[↩]
- Yeom CH, Jung GC, Song KJ. Changes of terminal cancer patients’ health-related quality of life after high dose vitamin C administration. J Korean Med Sci. 2007 Feb;22(1):7-11. doi: 10.3346/jkms.2007.22.1.7[↩]
- Nielsen TK, Højgaard M, Andersen JT, Jørgensen NR, Zerahn B, Kristensen B, Henriksen T, Lykkesfeldt J, Mikines KJ, Poulsen HE. Weekly ascorbic acid infusion in castration-resistant prostate cancer patients: a single-arm phase II trial. Transl Androl Urol. 2017 Jun;6(3):517-528. doi: 10.21037/tau.2017.04.42[↩]
- Monti DA, Mitchell E, Bazzan AJ, Littman S, Zabrecky G, Yeo CJ, Pillai MV, Newberg AB, Deshmukh S, Levine M. Phase I evaluation of intravenous ascorbic acid in combination with gemcitabine and erlotinib in patients with metastatic pancreatic cancer. PLoS One. 2012;7(1):e29794. doi: 10.1371/journal.pone.0029794[↩][↩][↩][↩]
- A Research Trial of High Dose Vitamin C and Chemotherapy for Metastatic Pancreatic Cancer. https://clinicaltrials.gov/study/NCT01049880[↩]
- Welsh JL, Wagner BA, van’t Erve TJ, Zehr PS, Berg DJ, Halfdanarson TR, Yee NS, Bodeker KL, Du J, Roberts LJ 2nd, Drisko J, Levine M, Buettner GR, Cullen JJ. Pharmacological ascorbate with gemcitabine for the control of metastatic and node-positive pancreatic cancer (PACMAN): results from a phase I clinical trial. Cancer Chemother Pharmacol. 2013 Mar;71(3):765-75. doi: 10.1007/s00280-013-2070-8[↩]
- Ma Y, Chapman J, Levine M, Polireddy K, Drisko J, Chen Q. High-dose parenteral ascorbate enhanced chemosensitivity of ovarian cancer and reduced toxicity of chemotherapy. Sci Transl Med. 2014 Feb 5;6(222):222ra18. doi: 10.1126/scitranslmed.3007154[↩][↩][↩]
- Hoffer LJ, Robitaille L, Zakarian R, Melnychuk D, Kavan P, Agulnik J, Cohen V, Small D, Miller WH Jr. High-dose intravenous vitamin C combined with cytotoxic chemotherapy in patients with advanced cancer: a phase I-II clinical trial. PLoS One. 2015 Apr 7;10(4):e0120228. doi: 10.1371/journal.pone.0120228[↩][↩][↩]
- Polireddy K, Dong R, Reed G, Yu J, Chen P, Williamson S, Violet PC, Pessetto Z, Godwin AK, Fan F, Levine M, Drisko JA, Chen Q. High Dose Parenteral Ascorbate Inhibited Pancreatic Cancer Growth and Metastasis: Mechanisms and a Phase I/IIa study. Sci Rep. 2017 Dec 7;7(1):17188. doi: 10.1038/s41598-017-17568-8[↩][↩]
- Wang F, He MM, Wang ZX, Li S, Jin Y, Ren C, Shi SM, Bi BT, Chen SZ, Lv ZD, Hu JJ, Wang ZQ, Wang FH, Wang DS, Li YH, Xu RH. Phase I study of high-dose ascorbic acid with mFOLFOX6 or FOLFIRI in patients with metastatic colorectal cancer or gastric cancer. BMC Cancer. 2019 May 16;19(1):460. doi: 10.1186/s12885-019-5696-z[↩][↩]
- Vitamin C Intravenously With Chemotherapy in Advanced Colorectal Cancer (Vitality). https://clinicaltrials.gov/study/NCT02969681[↩]
- Campbell RA, Sanchez E, Steinberg JA, Baritaki S, Gordon M, Wang C, Shalitin D, Chen H, Pang S, Bonavida B, Said J, Berenson JR. Antimyeloma effects of arsenic trioxide are enhanced by melphalan, bortezomib and ascorbic acid. Br J Haematol. 2007 Aug;138(4):467-78. doi: 10.1111/j.1365-2141.2007.06675.x[↩]
- Qazilbash MH, Saliba RM, Nieto Y, Parikh G, Pelosini M, Khan FB, Jones RB, Hosing C, Mendoza F, Weber DM, Wang M, Popat U, Alousi A, Anderlini P, Champlin RE, Giralt S. Arsenic trioxide with ascorbic acid and high-dose melphalan: results of a phase II randomized trial. Biol Blood Marrow Transplant. 2008 Dec;14(12):1401-7. doi: 10.1016/j.bbmt.2008.09.019[↩]
- Abou-Jawde RM, Reed J, Kelly M, Walker E, Andresen S, Baz R, Karam MA, Hussein M. Efficacy and safety results with the combination therapy of arsenic trioxide, dexamethasone, and ascorbic acid in multiple myeloma patients: a phase 2 trial. Med Oncol. 2006;23(2):263-72. doi: 10.1385/MO:23:2:263[↩]
- Berenson JR, Matous J, Swift RA, Mapes R, Morrison B, Yeh HS. A phase I/II study of arsenic trioxide/bortezomib/ascorbic acid combination therapy for the treatment of relapsed or refractory multiple myeloma. Clin Cancer Res. 2007 Mar 15;13(6):1762-8. doi: 10.1158/1078-0432.CCR-06-1812[↩]
- Berenson JR, Boccia R, Siegel D, Bozdech M, Bessudo A, Stadtmauer E, Talisman Pomeroy J, Steis R, Flam M, Lutzky J, Jilani S, Volk J, Wong SF, Moss R, Patel R, Ferretti D, Russell K, Louie R, Yeh HS, Swift RA. Efficacy and safety of melphalan, arsenic trioxide and ascorbic acid combination therapy in patients with relapsed or refractory multiple myeloma: a prospective, multicentre, phase II, single-arm study. Br J Haematol. 2006 Oct;135(2):174-83. doi: 10.1111/j.1365-2141.2006.06280.x. Erratum in: Br J Haematol. 2007 Jan;136(1):173-4.[↩]
- Subbarayan PR, Lima M, Ardalan B. Arsenic trioxide/ascorbic acid therapy in patients with refractory metastatic colorectal carcinoma: a clinical experience. Acta Oncol. 2007;46(4):557-61. doi: 10.1080/02841860601042456[↩]
- Bael TE, Peterson BL, Gollob JA. Phase II trial of arsenic trioxide and ascorbic acid with temozolomide in patients with metastatic melanoma with or without central nervous system metastases. Melanoma Res. 2008 Apr;18(2):147-51. doi: 10.1097/CMR.0b013e3282f2a7ae[↩]
- Padayatty SJ, Sun H, Wang Y, et al.: Vitamin C pharmacokinetics: implications for oral and intravenous use. Ann Intern Med 140 (7): 533-7, 2004. https://www.ncbi.nlm.nih.gov/pubmed/15068981?dopt=Abstract[↩]
- Hoffer LJ, Levine M, Assouline S, et al.: Phase I clinical trial of i.v. ascorbic acid in advanced malignancy. Ann Oncol 19 (11): 1969-74, 2008. https://www.ncbi.nlm.nih.gov/pubmed/18544557?dopt=Abstract[↩]
- Chen Q, Espey MG, Sun AY, et al.: Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc Natl Acad Sci U S A 105 (32): 11105-9, 2008. https://www.ncbi.nlm.nih.gov/pubmed/18678913?dopt=Abstract[↩]
- Monti DA, Mitchell E, Bazzan AJ, et al.: Phase I evaluation of intravenous ascorbic acid in combination with gemcitabine and erlotinib in patients with metastatic pancreatic cancer. PLoS One 7 (1): e29794, 2012. https://www.ncbi.nlm.nih.gov/pubmed/22272248?dopt=Abstract[↩]
- Abou-Jawde RM, Reed J, Kelly M, et al.: Efficacy and safety results with the combination therapy of arsenic trioxide, dexamethasone, and ascorbic acid in multiple myeloma patients: a phase 2 trial. Med Oncol 23 (2): 263-72, 2006. https://www.ncbi.nlm.nih.gov/pubmed/16720927?dopt=Abstract[↩]
- Berenson JR, Matous J, Swift RA, et al.: A phase I/II study of arsenic trioxide/bortezomib/ascorbic acid combination therapy for the treatment of relapsed or refractory multiple myeloma. Clin Cancer Res 13 (6): 1762-8, 2007. https://www.ncbi.nlm.nih.gov/pubmed/17363530?dopt=Abstract[↩]
- Qazilbash MH, Saliba RM, Nieto Y, et al.: Arsenic trioxide with ascorbic acid and high-dose melphalan: results of a phase II randomized trial. Biol Blood Marrow Transplant 14 (12): 1401-7, 2008. https://www.ncbi.nlm.nih.gov/pubmed/19041063?dopt=Abstract[↩]
- Ma Y, Chapman J, Levine M, et al.: High-dose parenteral ascorbate enhanced chemosensitivity of ovarian cancer and reduced toxicity of chemotherapy. Sci Transl Med 6 (222): 222ra18, 2014. https://www.ncbi.nlm.nih.gov/pubmed/24500406?dopt=Abstract[↩]
- Padayatty SJ, Sun AY, Chen Q, et al.: Vitamin C: intravenous use by complementary and alternative medicine practitioners and adverse effects. PLoS One 5 (7): e11414, 2010. https://www.ncbi.nlm.nih.gov/pubmed/20628650?dopt=Abstract[↩]
- Campbell GD Jr, Steinberg MH, Bower JD: Letter: Ascorbic acid-induced hemolysis in G-6-PD deficiency. Ann Intern Med 82 (6): 810, 1975. https://www.ncbi.nlm.nih.gov/pubmed/1138591?dopt=Abstract[↩]
- Mehta JB, Singhal SB, Mehta BC: Ascorbic-acid-induced haemolysis in G-6-PD deficiency. Lancet 336 (8720): 944, 1990. https://www.ncbi.nlm.nih.gov/pubmed/1976956?dopt=Abstract[↩]
- Rees DC, Kelsey H, Richards JD: Acute haemolysis induced by high dose ascorbic acid in glucose-6-phosphate dehydrogenase deficiency. BMJ 306 (6881): 841-2, 1993. https://www.ncbi.nlm.nih.gov/pubmed/8490379?dopt=Abstract[↩]
- Barton JC, McDonnell SM, Adams PC, et al.: Management of hemochromatosis. Hemochromatosis Management Working Group. Ann Intern Med 129 (11): 932-9, 1998. https://www.ncbi.nlm.nih.gov/pubmed/9867745?dopt=Abstract[↩]
- Welsh JL, Wagner BA, van’t Erve TJ, et al.: Pharmacological ascorbate with gemcitabine for the control of metastatic and node-positive pancreatic cancer (PACMAN): results from a phase I clinical trial. Cancer Chemother Pharmacol 71 (3): 765-75, 2013. https://www.ncbi.nlm.nih.gov/pubmed/23381814?dopt=Abstract[↩]
- Zou W, Yue P, Lin N, et al.: Vitamin C inactivates the proteasome inhibitor PS-341 in human cancer cells. Clin Cancer Res 12 (1): 273-80, 2006. https://www.ncbi.nlm.nih.gov/pubmed/16397052?dopt=Abstract[↩]
- Llobet D, Eritja N, Encinas M, et al.: Antioxidants block proteasome inhibitor function in endometrial carcinoma cells. Anticancer Drugs 19 (2): 115-24, 2008. https://www.ncbi.nlm.nih.gov/pubmed/18176107?dopt=Abstract[↩]
- Perrone G, Hideshima T, Ikeda H, et al.: Ascorbic acid inhibits antitumor activity of bortezomib in vivo. Leukemia 23 (9): 1679-86, 2009. https://www.ncbi.nlm.nih.gov/pubmed/19369963?dopt=Abstract[↩][↩]
- Bannerman B, Xu L, Jones M, et al.: Preclinical evaluation of the antitumor activity of bortezomib in combination with vitamin C or with epigallocatechin gallate, a component of green tea. Cancer Chemother Pharmacol 68 (5): 1145-54, 2011. https://www.ncbi.nlm.nih.gov/pubmed/21400028?dopt=Abstract[↩]
- Heaney ML, Gardner JR, Karasavvas N, et al.: Vitamin C antagonizes the cytotoxic effects of antineoplastic drugs. Cancer Res 68 (19): 8031-8, 2008. https://www.ncbi.nlm.nih.gov/pubmed/18829561?dopt=Abstract[↩]
- Ye Z, Song H. Antioxidant vitamins intake and the risk of coronary heart disease: meta-analysis of cohort studies. Eur J Cardiovasc Prev Rehabil. 2008 Feb;15(1):26-34. doi: 10.1097/HJR.0b013e3282f11f95[↩][↩]
- Willcox BJ, Curb JD, Rodriguez BL. Antioxidants in cardiovascular health and disease: key lessons from epidemiologic studies. Am J Cardiol. 2008 May 22;101(10A):75D-86D. doi: 10.1016/j.amjcard.2008.02.012[↩][↩][↩]
- Honarbakhsh S, Schachter M. Vitamins and cardiovascular disease. Br J Nutr. 2009 Apr;101(8):1113-31. doi: 10.1017/S000711450809123X[↩]
- Osganian SK, Stampfer MJ, Rimm E, Spiegelman D, Hu FB, Manson JE, Willett WC. Vitamin C and risk of coronary heart disease in women. J Am Coll Cardiol. 2003 Jul 16;42(2):246-52. doi: 10.1016/s0735-1097(03)00575-8[↩]
- Lee DH, Folsom AR, Harnack L, Halliwell B, Jacobs DR Jr. Does supplemental vitamin C increase cardiovascular disease risk in women with diabetes? Am J Clin Nutr. 2004 Nov;80(5):1194-200. doi: 10.1093/ajcn/80.5.1194[↩]
- Myint PK, Luben RN, Welch AA, Bingham SA, Wareham NJ, Khaw KT. Plasma vitamin C concentrations predict risk of incident stroke over 10 y in 20 649 participants of the European Prospective Investigation into Cancer Norfolk prospective population study. Am J Clin Nutr. 2008 Jan;87(1):64-9. doi: 10.1093/ajcn/87.1.64[↩]
- Muntwyler J, Hennekens CH, Manson JE, Buring JE, Gaziano JM. Vitamin supplement use in a low-risk population of US male physicians and subsequent cardiovascular mortality. Arch Intern Med. 2002 Jul 8;162(13):1472-6. doi: 10.1001/archinte.162.13.1472[↩]
- Knekt P, Ritz J, Pereira MA, O’Reilly EJ, Augustsson K, Fraser GE, Goldbourt U, Heitmann BL, Hallmans G, Liu S, Pietinen P, Spiegelman D, Stevens J, Virtamo J, Willett WC, Rimm EB, Ascherio A. Antioxidant vitamins and coronary heart disease risk: a pooled analysis of 9 cohorts. Am J Clin Nutr. 2004 Dec;80(6):1508-20. doi: 10.1093/ajcn/80.6.1508[↩]
- Cook NR, Albert CM, Gaziano JM, Zaharris E, MacFadyen J, Danielson E, Buring JE, Manson JE. A randomized factorial trial of vitamins C and E and beta carotene in the secondary prevention of cardiovascular events in women: results from the Women’s Antioxidant Cardiovascular Study. Arch Intern Med. 2007 Aug 13-27;167(15):1610-8. doi: 10.1001/archinte.167.15.1610[↩][↩]
- Sesso HD, Buring JE, Christen WG, Kurth T, Belanger C, MacFadyen J, Bubes V, Manson JE, Glynn RJ, Gaziano JM. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians’ Health Study II randomized controlled trial. JAMA. 2008 Nov 12;300(18):2123-33. doi: 10.1001/jama.2008.600[↩][↩]
- Waters DD, Alderman EL, Hsia J, Howard BV, Cobb FR, Rogers WJ, Ouyang P, Thompson P, Tardif JC, Higginson L, Bittner V, Steffes M, Gordon DJ, Proschan M, Younes N, Verter JI. Effects of hormone replacement therapy and antioxidant vitamin supplements on coronary atherosclerosis in postmenopausal women: a randomized controlled trial. JAMA. 2002 Nov 20;288(19):2432-40. doi: 10.1001/jama.288.19.2432[↩][↩]
- Bleys J, Miller ER 3rd, Pastor-Barriuso R, Appel LJ, Guallar E. Vitamin-mineral supplementation and the progression of atherosclerosis: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2006 Oct;84(4):880-7; quiz 954-5. doi: 10.1093/ajcn/84.4.880[↩]
- Shekelle P, Morton SC, Hardy M. Effect of Supplemental Antioxidants Vitamin C, Vitamin E, and Coenzyme Q10 for the Prevention and Treatment of Cardiovascular Disease. Rockville (MD): Agency for Healthcare Research and Quality (US); 2003 Jul. (Evidence Reports/Technology Assessments, No. 83.) Available from: https://www.ncbi.nlm.nih.gov/books/NBK37059[↩]
- van Leeuwen R, Boekhoorn S, Vingerling JR, Witteman JC, Klaver CC, Hofman A, de Jong PT. Dietary intake of antioxidants and risk of age-related macular degeneration. JAMA. 2005 Dec 28;294(24):3101-7. doi: 10.1001/jama.294.24.3101[↩]
- Evans J. Primary prevention of age related macular degeneration. BMJ. 2007 Oct 13;335(7623):729. doi: 10.1136/bmj.39351.478924.BE[↩]
- Chong EW, Wong TY, Kreis AJ, Simpson JA, Guymer RH. Dietary antioxidants and primary prevention of age related macular degeneration: systematic review and meta-analysis. BMJ. 2007 Oct 13;335(7623):755. doi: 10.1136/bmj.39350.500428.47[↩]
- Evans JR. Antioxidant vitamin and mineral supplements for slowing the progression of age-related macular degeneration. Cochrane Database Syst Rev. 2006 Apr 19;(2):CD000254. doi: 10.1002/14651858.CD000254.pub2. Update in: Cochrane Database Syst Rev. 2012;11:CD000254.[↩]
- Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001 Oct;119(10):1417-36. doi: 10.1001/archopht.119.10.1417. Erratum in: Arch Ophthalmol. 2008 Sep;126(9):1251.[↩]
- Age-Related Eye Disease Study 2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: the Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA. 2013 May 15;309(19):2005-15. doi: 10.1001/jama.2013.4997. Erratum in: JAMA. 2013 Jul 10;310(2):208.[↩]
- Yoshida M, Takashima Y, Inoue M, Iwasaki M, Otani T, Sasaki S, Tsugane S; JPHC Study Group. Prospective study showing that dietary vitamin C reduced the risk of age-related cataracts in a middle-aged Japanese population. Eur J Nutr. 2007 Mar;46(2):118-24. doi: 10.1007/s00394-006-0641-8[↩]
- Rautiainen S, Lindblad BE, Morgenstern R, Wolk A. Vitamin C supplements and the risk of age-related cataract: a population-based prospective cohort study in women. Am J Clin Nutr. 2010 Feb;91(2):487-93. doi: 10.3945/ajcn.2009.28528[↩]
- Sperduto RD, Hu TS, Milton RC, Zhao JL, Everett DF, Cheng QF, Blot WJ, Bing L, Taylor PR, Li JY, et al. The Linxian cataract studies. Two nutrition intervention trials. Arch Ophthalmol. 1993 Sep;111(9):1246-53. doi: 10.1001/archopht.1993.01090090098027[↩][↩]
- Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E and beta carotene for age-related cataract and vision loss: AREDS report no. 9. Arch Ophthalmol. 2001 Oct;119(10):1439-52. doi: 10.1001/archopht.119.10.1439. Erratum in: Arch Ophthalmol. 2008 Sep;126(9):1251.[↩]
- Age-Related Eye Disease Study 2 (AREDS2) Research Group, Chew EY, SanGiovanni JP, Ferris FL, Wong WT, Agron E, Clemons TE, Sperduto R, Danis R, Chandra SR, Blodi BA, Domalpally A, Elman MJ, Antoszyk AN, Ruby AJ, Orth D, Bressler SB, Fish GE, Hubbard GB, Klein ML, Friberg TR, Rosenfeld PJ, Toth CA, Bernstein P. Lutein/zeaxanthin for the treatment of age-related cataract: AREDS2 randomized trial report no. 4. JAMA Ophthalmol. 2013 Jul;131(7):843-50. doi: 10.1001/jamaophthalmol.2013.4412[↩]
- Pauling L. The significance of the evidence about ascorbic acid and the common cold. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2678-81. doi: 10.1073/pnas.68.11.2678[↩]
- Douglas RM, Hemilä H, Chalker E, Treacy B. Vitamin C for preventing and treating the common cold. Cochrane Database Syst Rev. 2007 Jul 18;(3):CD000980. doi: 10.1002/14651858.CD000980.pub3. Update in: Cochrane Database Syst Rev. 2013;1:CD000980.[↩][↩]
- Hemilä H. Vitamin C and common cold-induced asthma: a systematic review and statistical analysis. Allergy Asthma Clin Immunol. 2013 Nov 26;9(1):46. doi: 10.1186/1710-1492-9-46[↩][↩]
- The Role of Vitamin C in the Treatment of the Common Cold. Am Fam Physician. 2007 Oct 15;76(8):1111-1115. https://www.aafp.org/afp/2007/1015/p1111a.html[↩][↩]
- Johnston CS. The antihistamine action of ascorbic acid. Subcell Biochem. 1996;25:189-213. doi: 10.1007/978-1-4613-0325-1_10[↩]
- Douglas RM, Hemilä H, Chalker E, Treacy B. Vitamin C for preventing and treating the common cold. Cochrane Database Syst Rev. 2007 Jul 18;(3):CD000980. doi: 10.1002/14651858.CD000980.pub3. Update in: Cochrane Database Syst Rev. 2013;1:CD000980[↩]
- Milan SJ, Hart A, Wilkinson M. Vitamin C for asthma and exercise-induced bronchoconstriction. Cochrane Database Syst Rev. 2013 Oct 23;2013(10):CD010391. doi: 10.1002/14651858.CD010391.pub2[↩][↩]
- Shindo Y, Witt E, Han D, Epstein W, Packer L. Enzymic and non-enzymic antioxidants in epidermis and dermis of human skin. J Invest Dermatol. 1994 Jan;102(1):122-4. doi: 10.1111/1523-1747.ep12371744[↩]
- Rhie G, Shin MH, Seo JY, Choi WW, Cho KH, Kim KH, Park KC, Eun HC, Chung JH. Aging- and photoaging-dependent changes of enzymic and nonenzymic antioxidants in the epidermis and dermis of human skin in vivo. J Invest Dermatol. 2001 Nov;117(5):1212-7. doi: 10.1046/j.0022-202x.2001.01469.x. Erratum in: J Invest Dermatol 2002 Apr;118(4):741.[↩][↩][↩]
- Lopez-Torres M, Shindo Y, Packer L. Effect of age on antioxidants and molecular markers of oxidative damage in murine epidermis and dermis. J Invest Dermatol. 1994 Apr;102(4):476-80. doi: 10.1111/1523-1747.ep12373032[↩]
- Shindo Y, Witt E, Packer L. Antioxidant defense mechanisms in murine epidermis and dermis and their responses to ultraviolet light. J Invest Dermatol. 1993 Mar;100(3):260-5. doi: 10.1111/1523-1747.ep12469048[↩][↩][↩]
- Thiele JJ, Traber MG, Tsang K, Cross CE, Packer L. In vivo exposure to ozone depletes vitamins C and E and induces lipid peroxidation in epidermal layers of murine skin. Free Radic Biol Med. 1997;23(3):385-91. doi: 10.1016/s0891-5849(96)00617-x[↩][↩]
- Podda M, Traber MG, Weber C, Yan LJ, Packer L. UV-irradiation depletes antioxidants and causes oxidative damage in a model of human skin. Free Radic Biol Med. 1998 Jan 1;24(1):55-65. doi: 10.1016/s0891-5849(97)00142-1[↩][↩]
- Vitamin C and Skin Health. https://lpi.oregonstate.edu/mic/health-disease/skin-health/vitamin-C[↩][↩][↩]
- Steiling H, Longet K, Moodycliffe A, Mansourian R, Bertschy E, Smola H, Mauch C, Williamson G. Sodium-dependent vitamin C transporter isoforms in skin: Distribution, kinetics, and effect of UVB-induced oxidative stress. Free Radic Biol Med. 2007 Sep 1;43(5):752-62. doi: 10.1016/j.freeradbiomed.2007.05.001[↩][↩][↩]
- Kang JS, Kim HN, Jung DJ, Kim JE, Mun GH, Kim YS, Cho D, Shin DH, Hwang YI, Lee WJ. Regulation of UVB-induced IL-8 and MCP-1 production in skin keratinocytes by increasing vitamin C uptake via the redistribution of SVCT-1 from the cytosol to the membrane. J Invest Dermatol. 2007 Mar;127(3):698-706. doi: 10.1038/sj.jid.5700572[↩][↩]
- McArdle F, Rhodes LE, Parslew R, Jack CI, Friedmann PS, Jackson MJ. UVR-induced oxidative stress in human skin in vivo: effects of oral vitamin C supplementation. Free Radic Biol Med. 2002 Nov 15;33(10):1355-62. doi: 10.1016/s0891-5849(02)01042-0[↩][↩]
- Fuchs J, Kern H. Modulation of UV-light-induced skin inflammation by D-alpha-tocopherol and L-ascorbic acid: a clinical study using solar simulated radiation. Free Radic Biol Med. 1998 Dec;25(9):1006-12. doi: 10.1016/s0891-5849(98)00132-4[↩][↩]
- Padayatty SJ, Katz A, Wang Y, Eck P, Kwon O, Lee JH, Chen S, Corpe C, Dutta A, Dutta SK, Levine M. Vitamin C as an antioxidant: evaluation of its role in disease prevention. J Am Coll Nutr. 2003 Feb;22(1):18-35. doi: 10.1080/07315724.2003.10719272[↩]
- Hodges RE, Hood J, Canham JE, Sauberlich HE, Baker EM. Clinical manifestations of ascorbic acid deficiency in man. Am J Clin Nutr. 1971 Apr;24(4):432-43. doi: 10.1093/ajcn/24.4.432[↩]
- Hodges RE, Baker EM, Hood J, Sauberlich HE, March SC. Experimental scurvy in man. Am J Clin Nutr. 1969 May;22(5):535-48. doi: 10.1093/ajcn/22.5.535[↩]
- Pehlivan, F.E. (2017). Vitamin C: An Antioxidant Agent. https://pdfs.semanticscholar.org/07c9/9d103859bae18c6a69dd4a5ecf7b812713c5.pdf[↩]
- ROSS R, BENDITT EP. Wound healing and collagen formation. II. Fine structure in experimental scurvy. J Cell Biol. 1962 Mar;12(3):533-51. doi: 10.1083/jcb.12.3.533[↩]
- Darr D, Combs S, Dunston S, Manning T, Pinnell S. Topical vitamin C protects porcine skin from ultraviolet radiation-induced damage. Br J Dermatol. 1992 Sep;127(3):247-53. doi: 10.1111/j.1365-2133.1992.tb00122.x[↩][↩]
- Shindo Y, Witt E, Han D, Packer L. Dose-response effects of acute ultraviolet irradiation on antioxidants and molecular markers of oxidation in murine epidermis and dermis. J Invest Dermatol. 1994 Apr;102(4):470-5. doi: 10.1111/1523-1747.ep12373027[↩]
- Tebbe B, Wu S, Geilen CC, Eberle J, Kodelja V, Orfanos CE. L-ascorbic acid inhibits UVA-induced lipid peroxidation and secretion of IL-1alpha and IL-6 in cultured human keratinocytes in vitro. J Invest Dermatol. 1997 Mar;108(3):302-6. doi: 10.1111/1523-1747.ep12286468[↩]
- Stewart MS, Cameron GS, Pence BC. Antioxidant nutrients protect against UVB-induced oxidative damage to DNA of mouse keratinocytes in culture. J Invest Dermatol. 1996 May;106(5):1086-9. doi: 10.1111/1523-1747.ep12339344[↩]
- Nakamura T, Pinnell SR, Darr D, Kurimoto I, Itami S, Yoshikawa K, Streilein JW. Vitamin C abrogates the deleterious effects of UVB radiation on cutaneous immunity by a mechanism that does not depend on TNF-alpha. J Invest Dermatol. 1997 Jul;109(1):20-4. doi: 10.1111/1523-1747.ep12276349[↩]
- Savini I, D’Angelo I, Ranalli M, Melino G, Avigliano L. Ascorbic acid maintenance in HaCaT cells prevents radical formation and apoptosis by UV-B. Free Radic Biol Med. 1999 May;26(9-10):1172-80. doi: 10.1016/s0891-5849(98)00311-6[↩]
- Dunham WB, Zuckerkandl E, Reynolds R, Willoughby R, Marcuson R, Barth R, Pauling L. Effects of intake of L-ascorbic acid on the incidence of dermal neoplasms induced in mice by ultraviolet light. Proc Natl Acad Sci U S A. 1982 Dec;79(23):7532-6. doi: 10.1073/pnas.79.23.7532[↩]
- Pauling L, Willoughby R, Reynolds R, Blaisdell BE, Lawson S. Incidence of squamous cell carcinoma in hairless mice irradiated with ultraviolet light in relation to intake of ascorbic acid (vitamin C) and of D, L-alpha-tocopheryl acetate (vitamin E). Int J Vitam Nutr Res Suppl. 1982;23:53-82.[↩]
- Placzek M, Gaube S, Kerkmann U, Gilbertz KP, Herzinger T, Haen E, Przybilla B. Ultraviolet B-induced DNA damage in human epidermis is modified by the antioxidants ascorbic acid and D-alpha-tocopherol. J Invest Dermatol. 2005 Feb;124(2):304-7. doi: 10.1111/j.0022-202X.2004.23560.x[↩]
- Eberlein-König B, Placzek M, Przybilla B. Protective effect against sunburn of combined systemic ascorbic acid (vitamin C) and d-alpha-tocopherol (vitamin E). J Am Acad Dermatol. 1998 Jan;38(1):45-8. doi: 10.1016/s0190-9622(98)70537-7[↩]
- Bissett DL, Chatterjee R, Hannon DP. Photoprotective effect of superoxide-scavenging antioxidants against ultraviolet radiation-induced chronic skin damage in the hairless mouse. Photodermatol Photoimmunol Photomed. 1990 Apr;7(2):56-62.[↩]
- Lin JY, Selim MA, Shea CR, Grichnik JM, Omar MM, Monteiro-Riviere NA, Pinnell SR. UV photoprotection by combination topical antioxidants vitamin C and vitamin E. J Am Acad Dermatol. 2003 Jun;48(6):866-74. doi: 10.1067/mjd.2003.425[↩][↩]
- Quevedo WC Jr, Holstein TJ, Dyckman J, McDonald CJ, Isaacson EL. Inhibition of UVR-induced tanning and immunosuppression by topical applications of vitamins C and E to the skin of hairless (hr/hr) mice. Pigment Cell Res. 2000 Apr;13(2):89-98. doi: 10.1034/j.1600-0749.2000.130207.x[↩]
- Darr D, Dunston S, Faust H, Pinnell S. Effectiveness of antioxidants (vitamin C and E) with and without sunscreens as topical photoprotectants. Acta Derm Venereol. 1996 Jul;76(4):264-8. doi: 10.2340/0001555576264268[↩]
- Lin FH, Lin JY, Gupta RD, Tournas JA, Burch JA, Selim MA, Monteiro-Riviere NA, Grichnik JM, Zielinski J, Pinnell SR. Ferulic acid stabilizes a solution of vitamins C and E and doubles its photoprotection of skin. J Invest Dermatol. 2005 Oct;125(4):826-32. doi: 10.1111/j.0022-202X.2005.23768.x[↩]
- Ou-Yang H, Stamatas G, Saliou C, Kollias N. A chemiluminescence study of UVA-induced oxidative stress in human skin in vivo. J Invest Dermatol. 2004 Apr;122(4):1020-9. doi: 10.1111/j.0022-202X.2004.22405.x[↩]
- Dreher F, Gabard B, Schwindt DA, Maibach HI. Topical melatonin in combination with vitamins E and C protects skin from ultraviolet-induced erythema: a human study in vivo. Br J Dermatol. 1998 Aug;139(2):332-9. doi: 10.1046/j.1365-2133.1998.02447.x[↩][↩]
- Murray JC, Burch JA, Streilein RD, Iannacchione MA, Hall RP, Pinnell SR. A topical antioxidant solution containing vitamins C and E stabilized by ferulic acid provides protection for human skin against damage caused by ultraviolet irradiation. J Am Acad Dermatol. 2008 Sep;59(3):418-25. doi: 10.1016/j.jaad.2008.05.004[↩]
- Oresajo C, Stephens T, Hino PD, Law RM, Yatskayer M, Foltis P, Pillai S, Pinnell SR. Protective effects of a topical antioxidant mixture containing vitamin C, ferulic acid, and phloretin against ultraviolet-induced photodamage in human skin. J Cosmet Dermatol. 2008 Dec;7(4):290-7. doi: 10.1111/j.1473-2165.2008.00408.x[↩]
- Peterkofsky B. Ascorbate requirement for hydroxylation and secretion of procollagen: relationship to inhibition of collagen synthesis in scurvy. Am J Clin Nutr. 1991 Dec;54(6 Suppl):1135S-1140S. doi: 10.1093/ajcn/54.6.1135s[↩]
- Geesin JC, Darr D, Kaufman R, Murad S, Pinnell SR. Ascorbic acid specifically increases type I and type III procollagen messenger RNA levels in human skin fibroblast. J Invest Dermatol. 1988 Apr;90(4):420-4. doi: 10.1111/1523-1747.ep12460849[↩]
- Davidson JM, LuValle PA, Zoia O, Quaglino D Jr, Giro M. Ascorbate differentially regulates elastin and collagen biosynthesis in vascular smooth muscle cells and skin fibroblasts by pretranslational mechanisms. J Biol Chem. 1997 Jan 3;272(1):345-52. doi: 10.1074/jbc.272.1.345[↩]
- Phillips CL, Combs SB, Pinnell SR. Effects of ascorbic acid on proliferation and collagen synthesis in relation to the donor age of human dermal fibroblasts. J Invest Dermatol. 1994 Aug;103(2):228-32. doi: 10.1111/1523-1747.ep12393187[↩]
- Duarte TL, Cooke MS, Jones GD. Gene expression profiling reveals new protective roles for vitamin C in human skin cells. Free Radic Biol Med. 2009 Jan 1;46(1):78-87. doi: 10.1016/j.freeradbiomed.2008.09.028[↩][↩]
- Cosgrove MC, Franco OH, Granger SP, Murray PG, Mayes AE. Dietary nutrient intakes and skin-aging appearance among middle-aged American women. Am J Clin Nutr. 2007 Oct;86(4):1225-31. doi: 10.1093/ajcn/86.4.1225. Erratum in: Am J Clin Nutr. 2008 Aug;88(2):480.[↩][↩]
- Purba MB, Kouris-Blazos A, Wattanapenpaiboon N, Lukito W, Rothenberg EM, Steen BC, Wahlqvist ML. Skin wrinkling: can food make a difference? J Am Coll Nutr. 2001 Feb;20(1):71-80. doi: 10.1080/07315724.2001.10719017[↩]
- Traikovich SS. Use of topical ascorbic acid and its effects on photodamaged skin topography. Arch Otolaryngol Head Neck Surg. 1999 Oct;125(10):1091-8. doi: 10.1001/archotol.125.10.1091[↩][↩][↩]
- Raschke T, Koop U, Düsing HJ, Filbry A, Sauermann K, Jaspers S, Wenck H, Wittern KP. Topical activity of ascorbic acid: from in vitro optimization to in vivo efficacy. Skin Pharmacol Physiol. 2004 Jul-Aug;17(4):200-6. doi: 10.1159/000078824[↩]
- Humbert PG, Haftek M, Creidi P, Lapière C, Nusgens B, Richard A, Schmitt D, Rougier A, Zahouani H. Topical ascorbic acid on photoaged skin. Clinical, topographical and ultrastructural evaluation: double-blind study vs. placebo. Exp Dermatol. 2003 Jun;12(3):237-44. doi: 10.1034/j.1600-0625.2003.00008.x[↩][↩]
- Fitzpatrick RE, Rostan EF. Double-blind, half-face study comparing topical vitamin C and vehicle for rejuvenation of photodamage. Dermatol Surg. 2002 Mar;28(3):231-6. doi: 10.1046/j.1524-4725.2002.01129.x[↩][↩]
- Nusgens BV, Humbert P, Rougier A, Colige AC, Haftek M, Lambert CA, Richard A, Creidi P, Lapière CM. Topically applied vitamin C enhances the mRNA level of collagens I and III, their processing enzymes and tissue inhibitor of matrix metalloproteinase 1 in the human dermis. J Invest Dermatol. 2001 Jun;116(6):853-9. doi: 10.1046/j.0022-202x.2001.01362.x[↩][↩]
- Sauermann K, Jaspers S, Koop U, Wenck H. Topically applied vitamin C increases the density of dermal papillae in aged human skin. BMC Dermatol. 2004 Sep 29;4(1):13. doi: 10.1186/1471-5945-4-13[↩]
- Lind J. A Treatise on the Scurvy. In. London: A. Millar; 1753.[↩]
- Kim M, Otsuka M, Yu R, Kurata T, Arakawa N. The distribution of ascorbic acid and dehydroascorbic acid during tissue regeneration in wounded dorsal skin of guinea pigs. Int J Vitam Nutr Res. 1994;64(1):56-9.[↩]
- Shukla A, Rasik AM, Patnaik GK. Depletion of reduced glutathione, ascorbic acid, vitamin E and antioxidant defence enzymes in a healing cutaneous wound. Free Radic Res. 1997 Feb;26(2):93-101. doi: 10.3109/10715769709097788[↩][↩]
- Sørensen LT, Toft BG, Rygaard J, Ladelund S, Paddon M, James T, Taylor R, Gottrup F. Effect of smoking, smoking cessation, and nicotine patch on wound dimension, vitamin C, and systemic markers of collagen metabolism. Surgery. 2010 Nov;148(5):982-90. doi: 10.1016/j.surg.2010.02.005[↩][↩][↩]
- Savini I, Catani MV, Rossi A, Duranti G, Melino G, Avigliano L. Characterization of keratinocyte differentiation induced by ascorbic acid: protein kinase C involvement and vitamin C homeostasis. J Invest Dermatol. 2002 Feb;118(2):372-9. doi: 10.1046/j.0022-202x.2001.01624.x[↩]
- Boyce ST, Supp AP, Swope VB, Warden GD. Vitamin C regulates keratinocyte viability, epidermal barrier, and basement membrane in vitro, and reduces wound contraction after grafting of cultured skin substitutes. J Invest Dermatol. 2002 Apr;118(4):565-72. doi: 10.1046/j.1523-1747.2002.01717.x[↩]
- Ponec M, Weerheim A, Kempenaar J, Mulder A, Gooris GS, Bouwstra J, Mommaas AM. The formation of competent barrier lipids in reconstructed human epidermis requires the presence of vitamin C. J Invest Dermatol. 1997 Sep;109(3):348-55. doi: 10.1111/1523-1747.ep12336024[↩][↩]
- Krámer GM, Fillios LC, Bowler EC. Ascorbic acid treatment on early collagen production and wound healing in the guinea pig. J Periodontol. 1979 Apr;50(4):189-92. doi: 10.1902/jop.1979.50.4.189[↩]
- Young ME. Malnutrition and wound healing. Heart Lung. 1988 Jan;17(1):60-7.[↩]
- DUNPHY JE, EDWARDS LC, UDUPA KN. Wound healing; a new perspective with particular reference to ascorbic acid deficiency. Ann Surg. 1956 Sep;144(3):304-17. doi: 10.1097/00000658-195609000-00002[↩]
- Silverstein RJ, Landsman AS. The effects of a moderate and high dose of vitamin C on wound healing in a controlled guinea pig model. J Foot Ankle Surg. 1999 Sep-Oct;38(5):333-8. doi: 10.1016/s1067-2516(99)80004-0[↩]
- Thompson C, Fuhrman MP. Nutrients and wound healing: still searching for the magic bullet. Nutr Clin Pract. 2005 Jun;20(3):331-47. doi: 10.1177/0115426505020003331[↩]
- Vaxman F, Olender S, Lambert A, Nisand G, Aprahamian M, Bruch JF, Didier E, Volkmar P, Grenier JF. Effect of pantothenic acid and ascorbic acid supplementation on human skin wound healing process. A double-blind, prospective and randomized trial. Eur Surg Res. 1995;27(3):158-66. doi: 10.1159/000129395[↩]
- Desneves KJ, Todorovic BE, Cassar A, Crowe TC. Treatment with supplementary arginine, vitamin C and zinc in patients with pressure ulcers: a randomised controlled trial. Clin Nutr. 2005 Dec;24(6):979-87. doi: 10.1016/j.clnu.2005.06.011[↩]
- Ellinger S, Stehle P. Efficacy of vitamin supplementation in situations with wound healing disorders: results from clinical intervention studies. Curr Opin Clin Nutr Metab Care. 2009 Nov;12(6):588-95. doi: 10.1097/MCO.0b013e328331a5b5[↩]
- Barbosa E, Faintuch J, Machado Moreira EA, Gonçalves da Silva VR, Lopes Pereima MJ, Martins Fagundes RL, Filho DW. Supplementation of vitamin E, vitamin C, and zinc attenuates oxidative stress in burned children: a randomized, double-blind, placebo-controlled pilot study. J Burn Care Res. 2009 Sep-Oct;30(5):859-66. doi: 10.1097/BCR.0b013e3181b487a8[↩]
- Pasonen-Seppänen S, Suhonen TM, Kirjavainen M, Suihko E, Urtti A, Miettinen M, Hyttinen M, Tammi M, Tammi R. Vitamin C enhances differentiation of a continuous keratinocyte cell line (REK) into epidermis with normal stratum corneum ultrastructure and functional permeability barrier. Histochem Cell Biol. 2001 Oct;116(4):287-97. doi: 10.1007/s004180100312[↩]
- Campos PM, Gonçalves GM, Gaspar LR. In vitro antioxidant activity and in vivo efficacy of topical formulations containing vitamin C and its derivatives studied by non-invasive methods. Skin Res Technol. 2008 Aug;14(3):376-80. doi: 10.1111/j.1600-0846.2008.00288.x[↩]
- Klock J, Ikeno H, Ohmori K, Nishikawa T, Vollhardt J, Schehlmann V. Sodium ascorbyl phosphate shows in vitro and in vivo efficacy in the prevention and treatment of acne vulgaris. Int J Cosmet Sci. 2005 Jun;27(3):171-6. doi: 10.1111/j.1467-2494.2005.00263.x[↩]
- Woolery-Lloyd H, Baumann L, Ikeno H. Sodium L-ascorbyl-2-phosphate 5% lotion for the treatment of acne vulgaris: a randomized, double-blind, controlled trial. J Cosmet Dermatol. 2010 Mar;9(1):22-7. doi: 10.1111/j.1473-2165.2010.00480.x[↩]
- Kameyama K, Sakai C, Kondoh S, Yonemoto K, Nishiyama S, Tagawa M, Murata T, Ohnuma T, Quigley J, Dorsky A, Bucks D, Blanock K. Inhibitory effect of magnesium L-ascorbyl-2-phosphate (VC-PMG) on melanogenesis in vitro and in vivo. J Am Acad Dermatol. 1996 Jan;34(1):29-33. doi: 10.1016/s0190-9622(96)90830-0[↩]
- Cheng Y, Willett WC, Schwartz J, Sparrow D, Weiss S, Hu H. Relation of nutrition to bone lead and blood lead levels in middle-aged to elderly men. The Normative Aging Study. Am J Epidemiol. 1998 Jun 15;147(12):1162-74. doi: 10.1093/oxfordjournals.aje.a009415[↩][↩]
- Simon JA, Hudes ES. Relationship of ascorbic acid to blood lead levels. JAMA. 1999 Jun 23-30;281(24):2289-93. doi: 10.1001/jama.281.24.2289[↩]
- Lee DH, Lim JS, Song K, Boo Y, Jacobs DR Jr. Graded associations of blood lead and urinary cadmium concentrations with oxidative-stress-related markers in the U.S. population: results from the third National Health and Nutrition Examination Survey. Environ Health Perspect. 2006 Mar;114(3):350-4. doi: 10.1289/ehp.8518[↩]
- Dawson EB, Evans DR, Harris WA, Teter MC, McGanity WJ. The effect of ascorbic acid supplementation on the blood lead levels of smokers. J Am Coll Nutr. 1999 Apr;18(2):166-70. doi: 10.1080/07315724.1999.10718845[↩][↩]
- Abam E, Okediran BS, Odukoya OO, Adamson I, Ademuyiwa O. Reversal of ionoregulatory disruptions in occupational lead exposure by vitamin C. Environ Toxicol Pharmacol. 2008 Nov;26(3):297-304. doi: 10.1016/j.etap.2008.05.008[↩]
- Song Y, Xu Q, Park Y, Hollenbeck A, Schatzkin A, Chen H. Multivitamins, individual vitamin and mineral supplements, and risk of diabetes among older U.S. adults. Diabetes Care. 2011 Jan;34(1):108-14. doi: 10.2337/dc10-1260[↩]
- Harding AH, Wareham NJ, Bingham SA, Khaw K, Luben R, Welch A, Forouhi NG. Plasma vitamin C level, fruit and vegetable consumption, and the risk of new-onset type 2 diabetes mellitus: the European prospective investigation of cancer–Norfolk prospective study. Arch Intern Med. 2008 Jul 28;168(14):1493-9. doi: 10.1001/archinte.168.14.1493[↩]
- Donin AS, Dent JE, Nightingale CM, Sattar N, Owen CG, Rudnicka AR, Perkin MR, Stephen AM, Jebb SA, Cook DG, Whincup PH. Fruit, vegetable and vitamin C intakes and plasma vitamin C: cross-sectional associations with insulin resistance and glycaemia in 9-10 year-old children. Diabet Med. 2016 Mar;33(3):307-15. doi: 10.1111/dme.13006[↩]
- Kositsawat J, Freeman VL. Vitamin C and A1c relationship in the National Health and Nutrition Examination Survey (NHANES) 2003-2006. J Am Coll Nutr. 2011 Dec;30(6):477-83. doi: 10.1080/07315724.2011.10719993[↩]
- Carter P, Gray LJ, Troughton J, Khunti K, Davies MJ. Fruit and vegetable intake and incidence of type 2 diabetes mellitus: systematic review and meta-analysis. BMJ. 2010 Aug 18;341:c4229. doi: 10.1136/bmj.c4229[↩]
- Ashor AW, Werner AD, Lara J, Willis ND, Mathers JC, Siervo M. Effects of vitamin C supplementation on glycaemic control: a systematic review and meta-analysis of randomised controlled trials. Eur J Clin Nutr. 2017 Dec;71(12):1371-1380. doi: 10.1038/ejcn.2017.24[↩]
- Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007-2008. Arthritis Rheum. 2011 Oct;63(10):3136-41. doi: 10.1002/art.30520[↩]
- Saag KG, Choi H. Epidemiology, risk factors, and lifestyle modifications for gout. Arthritis Res Ther. 2006;8 Suppl 1(Suppl 1):S2. doi: 10.1186/ar1907[↩]
- Choi HK, Curhan G. Gout: epidemiology and lifestyle choices. Curr Opin Rheumatol. 2005 May;17(3):341-5.[↩]
- Gao X, Curhan G, Forman JP, Ascherio A, Choi HK. Vitamin C intake and serum uric acid concentration in men. J Rheumatol. 2008 Sep;35(9):1853-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2853937[↩]
- Zheng Z, Harman JL, Coresh J, Köttgen A, McAdams-DeMarco MA, Correa A, Young BA, Katz R, Rebholz CM. The Dietary Fructose:Vitamin C Intake Ratio Is Associated with Hyperuricemia in African-American Adults. J Nutr. 2018 Mar 1;148(3):419-426. doi: 10.1093/jn/nxx054[↩]
- Choi HK, Gao X, Curhan G. Vitamin C intake and the risk of gout in men: a prospective study. Arch Intern Med. 2009 Mar 9;169(5):502-7. doi: 10.1001/archinternmed.2008.606[↩][↩]
- Juraschek SP, Miller ER 3rd, Gelber AC. Effect of oral vitamin C supplementation on serum uric acid: a meta-analysis of randomized controlled trials. Arthritis Care Res (Hoboken). 2011 Sep;63(9):1295-306. doi: 10.1002/acr.20519[↩]
- Stamp LK, Zhu X, Dalbeth N, Jordan S, Edwards NL, Taylor W. Serum urate as a soluble biomarker in chronic gout-evidence that serum urate fulfills the OMERACT validation criteria for soluble biomarkers. Semin Arthritis Rheum. 2011 Jun;40(6):483-500. doi: 10.1016/j.semarthrit.2010.09.003[↩]
- Stamp LK, O’Donnell JL, Frampton C, Drake JM, Zhang M, Chapman PT. Clinically insignificant effect of supplemental vitamin C on serum urate in patients with gout: a pilot randomized controlled trial. Arthritis Rheum. 2013 Jun;65(6):1636-42. doi: 10.1002/art.37925[↩][↩]
- Andrés M, Sivera F, Falzon L, Buchbinder R, Carmona L. Dietary supplements for chronic gout. Cochrane Database Syst Rev. 2014 Oct 7;(10):CD010156. doi: 10.1002/14651858.CD010156.pub2. Update in: Cochrane Database Syst Rev. 2021 Nov 12;11:CD010156.[↩]
- Rumbold A, Ota E, Nagata C, Shahrook S, Crowther CA. Vitamin C supplementation in pregnancy. Cochrane Database Syst Rev. 2015 Sep 29;2015(9):CD004072. doi: 10.1002/14651858.CD004072.pub3[↩][↩]
- Balogun OO, da Silva Lopes K, Ota E, Takemoto Y, Rumbold A, Takegata M, Mori R. Vitamin supplementation for preventing miscarriage. Cochrane Database Syst Rev. 2016 May 6;2016(5):CD004073. doi: 10.1002/14651858.CD004073.pub4[↩]
- Hammoud AO, Bujold E, Sorokin Y, Schild C, Krapp M, Baumann P. Smoking in pregnancy revisited: findings from a large population-based study. Am J Obstet Gynecol. 2005 Jun;192(6):1856-62; discussion 1862-3. doi: 10.1016/j.ajog.2004.12.057[↩]
- Kitsantas P, Christopher KE. Smoking and respiratory conditions in pregnancy: associations with adverse pregnancy outcomes. South Med J. 2013 May;106(5):310-5. doi: 10.1097/SMJ.0b013e318290c6e8[↩]
- Milner AD, Rao H, Greenough A. The effects of antenatal smoking on lung function and respiratory symptoms in infants and children. Early Hum Dev. 2007 Nov;83(11):707-11. doi: 10.1016/j.earlhumdev.2007.07.014[↩]
- Conde-Agudelo A, Althabe F, Belizán JM, Kafury-Goeta AC. Cigarette smoking during pregnancy and risk of preeclampsia: a systematic review. Am J Obstet Gynecol. 1999 Oct;181(4):1026-35. doi: 10.1016/s0002-9378(99)70341-8[↩]
- Abramovici A, Gandley RE, Clifton RG, et al. Eunice Kennedy Shriver National Institute of Child Health Human Development Maternal-Fetal Medicine Units Network. Prenatal vitamin C and E supplementation in smokers is associated with reduced placental abruption and preterm birth: a secondary analysis. BJOG. 2015 Dec;122(13):1740-7. doi: 10.1111/1471-0528.13201[↩][↩]
- McEvoy CT, Schilling D, Clay N, Jackson K, Go MD, Spitale P, Bunten C, Leiva M, Gonzales D, Hollister-Smith J, Durand M, Frei B, Buist AS, Peters D, Morris CD, Spindel ER. Vitamin C supplementation for pregnant smoking women and pulmonary function in their newborn infants: a randomized clinical trial. JAMA. 2014 May;311(20):2074-82. doi: 10.1001/jama.2014.5217[↩]
- McEvoy CT, Milner KF, Scherman AJ, Schilling DG, Tiller CJ, Vuylsteke B, Shorey-Kendrick LE, Spindel ER, Schuff R, Mitchell J, Peters D, Metz J, Haas D, Jackson K, Tepper RS, Morris CD. Vitamin C to Decrease the Effects of Smoking in Pregnancy on Infant Lung Function (VCSIP): Rationale, design, and methods of a randomized, controlled trial of vitamin C supplementation in pregnancy for the primary prevention of effects of in utero tobacco smoke exposure on infant lung function and respiratory health. Contemp Clin Trials. 2017 Jul;58:66-77. doi: 10.1016/j.cct.2017.05.008[↩]
- Alzheimer’s Disease Facts and Figures. https://www.alz.org/alzheimers-dementia/facts-figures[↩]
- Hu X, Yuan L, Wang H, Li C, Cai J, Hu Y, Ma C. Efficacy and safety of vitamin C for atrial fibrillation after cardiac surgery: A meta-analysis with trial sequential analysis of randomized controlled trials. Int J Surg. 2017 Jan;37:58-64. doi: 10.1016/j.ijsu.2016.12.009[↩]
- Kook SY, Lee KM, Kim Y, Cha MY, Kang S, Baik SH, Lee H, Park R, Mook-Jung I. High-dose of vitamin C supplementation reduces amyloid plaque burden and ameliorates pathological changes in the brain of 5XFAD mice. Cell Death Dis. 2014 Feb 27;5(2):e1083. doi: 10.1038/cddis.2014.26[↩]
- Bowman GL. Ascorbic acid, cognitive function, and Alzheimer’s disease: a current review and future direction. Biofactors. 2012 Mar-Apr;38(2):114-22. doi: 10.1002/biof.1002[↩][↩][↩]
- Harrison J, Rentz DM, McLaughlin T, Niecko T, Gregg KM, Black RS, Buchanan J, Liu E, Grundman M; ELN-AIP-901 Study Investigator Group. Cognition in MCI and Alzheimer’s disease: baseline data from a longitudinal study of the NTB. Clin Neuropsychol. 2014;28(2):252-68. doi: 10.1080/13854046.2013.875595[↩][↩]
- Hansen SN, Tveden-Nyborg P, Lykkesfeldt J. Does vitamin C deficiency affect cognitive development and function? Nutrients. 2014 Sep 19;6(9):3818-46. doi: 10.3390/nu6093818[↩]
- Bowman GL, Dodge H, Frei B, Calabrese C, Oken BS, Kaye JA, Quinn JF. Ascorbic acid and rates of cognitive decline in Alzheimer’s disease. J Alzheimers Dis. 2009;16(1):93-8. doi: 10.3233/JAD-2009-0923[↩]
- Arlt S, Müller-Thomsen T, Beisiegel U, Kontush A. Effect of one-year vitamin C- and E-supplementation on cerebrospinal fluid oxidation parameters and clinical course in Alzheimer’s disease. Neurochem Res. 2012 Dec;37(12):2706-14. doi: 10.1007/s11064-012-0860-8[↩]
- Galasko DR, Peskind E, Clark CM, Quinn JF, Ringman JM, Jicha GA, Cotman C, Cottrell B, Montine TJ, Thomas RG, Aisen P; Alzheimer’s Disease Cooperative Study. Antioxidants for Alzheimer disease: a randomized clinical trial with cerebrospinal fluid biomarker measures. Arch Neurol. 2012 Jul;69(7):836-41. doi: 10.1001/archneurol.2012.85[↩]
- Naeini AM, Elmadfa I, Djazayery A, Barekatain M, Ghazvini MR, Djalali M, Feizi A. The effect of antioxidant vitamins E and C on cognitive performance of the elderly with mild cognitive impairment in Isfahan, Iran: a double-blind, randomized, placebo-controlled trial. Eur J Nutr. 2014 Aug;53(5):1255-62. doi: 10.1007/s00394-013-0628-1[↩]
- Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, Cohen J, Opal SM, Vincent JL, Ramsay G; SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003 Apr;31(4):1250-6. doi: 10.1097/01.CCM.0000050454.01978.3B[↩]
- Mann EA, Baun MM, Meininger JC, Wade CE. Comparison of mortality associated with sepsis in the burn, trauma, and general intensive care unit patient: a systematic review of the literature. Shock. 2012 Jan;37(1):4-16. doi: 10.1097/SHK.0b013e318237d6bf[↩]
- Carr AC, Rosengrave PC, Bayer S, Chambers S, Mehrtens J, Shaw GM. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit Care. 2017 Dec 11;21(1):300. doi: 10.1186/s13054-017-1891-y[↩]
- Pravda J. Metabolic theory of septic shock. World J Crit Care Med. 2014 May 4;3(2):45-54. doi: 10.5492/wjccm.v3.i2.45[↩][↩][↩][↩]
- Fowler AA 3rd, Syed AA, Knowlson S, Sculthorpe R, Farthing D, DeWilde C, Farthing CA, Larus TL, Martin E, Brophy DF, Gupta S; Medical Respiratory Intensive Care Unit Nursing; Fisher BJ, Natarajan R. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J Transl Med. 2014 Jan 31;12:32. doi: 10.1186/1479-5876-12-32[↩][↩][↩][↩]
- Zabet MH, Mohammadi M, Ramezani M, Khalili H. Effect of high-dose Ascorbic acid on vasopressor’s requirement in septic shock. J Res Pharm Pract. 2016 Apr-Jun;5(2):94-100. doi: 10.4103/2279-042X.179569[↩]
- Marik PE, Khangoora V, Rivera R, Hooper MH, Catravas J. Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Before-After Study. Chest. 2017 Jun;151(6):1229-1238. doi: 10.1016/j.chest.2016.11.036[↩][↩]
- Magiorkinis E, Beloukas A, Diamantis A: Scurvy: past, present and future. Eur J Intern Med. 2011, 22:147-52. https://doi.org/10.1016/j.ejim.2010.10.006[↩]
- Kluesner NH, Miller DG. Scurvy: malnourishment in the land of plenty. J Emerg Med. 2014 Apr;46(4):530-2. doi: 10.1016/j.jemermed.2013.09.027[↩]
- Al-Dabagh A, Milliron BJ, Strowd L, Feldman SR. A disease of the present: scurvy in “well-nourished” patients. J Am Acad Dermatol. 2013 Nov;69(5):e246-e247. doi: 10.1016/j.jaad.2013.04.051[↩]
- Olmedo JM, Yiannias JA, Windgassen EB, Gornet MK. Scurvy: a disease almost forgotten. Int J Dermatol. 2006 Aug;45(8):909-13. doi: 10.1111/j.1365-4632.2006.02844.x[↩][↩]
- Popovich D, McAlhany A, Adewumi AO, Barnes MM. Scurvy: forgotten but definitely not gone. J Pediatr Health Care. 2009 Nov-Dec;23(6):405-15. doi: 10.1016/j.pedhc.2008.10.008[↩]
- Wang AH, Still C. Old world meets modern: a case report of scurvy. Nutr Clin Pract. 2007 Aug;22(4):445-8. doi: 10.1177/0115426507022004445[↩][↩][↩][↩][↩][↩][↩]
- Francescone MA, Levitt J. Scurvy masquerading as leukocytoclastic vasculitis: a case report and review of the literature. Cutis. 2005 Oct;76(4):261-6.[↩][↩][↩][↩]
- Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. National Academies of Sciences, Engineering, and Medicine. 2000. Washington, DC: The National Academies Press. https://nap.nationalacademies.org/read/9810/chapter/1[↩]
- Hafez D, Saint S, Griauzde J, Mody R, Meddings J. CLINICAL PROBLEM-SOLVING. A Deficient Diagnosis. N Engl J Med. 2016 Apr 7;374(14):1369-74. doi: 10.1056/NEJMcps1407520[↩][↩][↩]
- Scurvy. https://emedicine.medscape.com/article/125350-overview[↩]
- Schectman G. Estimating ascorbic acid requirements for cigarette smokers. Ann N Y Acad Sci. 1993 May 28;686:335-45; discussion 345-6. doi: 10.1111/j.1749-6632.1993.tb39197.x[↩]
- Weber P, Bendich A, Schalch W. Vitamin C and human health—a review of recent data relevant to human requirements. Int J Vitam Nutr Res 1996;66:19-30.[↩]
- Perry ME, Page N, Manthey DE, Zavitz JM. Scurvy: Dietary Discretion in a Developed Country. Clin Pract Cases Emerg Med. 2018 Mar 14;2(2):147-150. doi: 10.5811/cpcem.2018.1.36860[↩][↩][↩]
- Pimentel L. Scurvy: historical review and current diagnostic approach. Am J Emerg Med. 2003 Jul;21(4):328-32. doi: 10.1016/s0735-6757(03)00083-4[↩][↩]
- Levine M, Rumsey SC, Daruwala R, Park JB, Wang Y. Criteria and recommendations for vitamin C intake. JAMA. 1999 Apr 21;281(15):1415-23. doi: 10.1001/jama.281.15.141[↩]
- Schleicher RL, Carroll MD, Ford ES, Lacher DA: Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003-2004 National Health and Nutrition Examination Survey (NHANES). Am J Clin Nutr. 2009, 90:1252-63. 10.3945/ajcn.2008.27016[↩]
- Mosdøl A, Erens B, Brunner EJ: Estimated prevalence and predictors of vitamin C deficiency within UK’s low-income population. J Public Health (Oxf). 2008, 30:456-60. 10.1093/pubmed/fdn076[↩]
- Khalife R, Grieco A, Khamisa K, Tinmouh A, McCudden C, Saidenberg E: Scurvy, an old story in a new time: the hematologist’s experience. Blood Cells Mol Dis. 2019, 76:40-4. 10.1016/j.bcmd.2019.01.004[↩]
- Faizallah R, Morris AI, Krasner N, Walker RJ. Alcohol enhances vitamin C excretion in the urine. Alcohol Alcohol. 1986;21(1):81-4.[↩][↩]
- Des Roches A, Paradis L, Paradis J, Singer S. Food allergy as a new risk factor for scurvy. Allergy. 2006 Dec;61(12):1487-8. doi: 10.1111/j.1398-9995.2006.01200.x[↩]
- Chen MF, Boyce HW Jr, Hsu JM. Effect of ascorbic acid on plasma alcohol clearance. J Am Coll Nutr. 1990 Jun;9(3):185-9. doi: 10.1080/07315724.1990.10720368[↩]
- Léger D. Scurvy: reemergence of nutritional deficiencies. Can Fam Physician. 2008 Oct;54(10):1403-6.[↩][↩][↩][↩][↩][↩][↩]
- Granger M., Eck P. Dietary vitamin C in human health. Adv. Food Nutr. Res. 2018;83:281–310. doi: 10.1016/bs.afnr.2017.11.006[↩]
- World Health Organization . Scurvy and its Prevention and Control in Major Emergencies/Prepared by Zita Weise Prinzo. World Health Organization; Geneva, Switzerland: 1999.[↩]
- Carr A.C., Maggini S. Vitamin C and immune function. Nutrients. 2017;9:1211. doi: 10.3390/nu9111211[↩]
- Hodges R.E., Hood J., Canham J.E., Sauberlich H.E., Baker E.M. Clinical manifestations of ascorbic acid deficiency in man. Am. J. Clin. Nutr. 1971;24:432–443. doi: 10.1093/ajcn/24.4.432[↩]
- Valerio E, Meneghel A, Masiero S, Zangardi T, Zanconato S. Scurvy: just think about it. J Pediatr. 2013 Dec. 163 (6):1786-7.[↩]
- Abdullah M, Attia FN. Vitamin C (Ascorbic Acid) [Updated 2018 Oct 27]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK499877[↩]
- Scurvy in a man with schizophrenia. Mark Dubé. CMAJ Aug 2011, 183 (11) E760; DOI: 10.1503/cmaj.080505 http://www.cmaj.ca/content/183/11/E760[↩]
- Phrynoderma: a cutaneous sign of an inadequate diet. Alessandro Di Stefani, Augusto Orlandi, Sergio Chimenti, Luca Bianchi. CMAJ Oct 2007, 177 (8) 855-856; DOI: 10.1503/cmaj.070086 http://www.cmaj.ca/content/177/8/855[↩]
- BMJ. 2001;322(7280):246. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1119499/[↩]
- Moser MA, Chun OK. Vitamin C and Heart Health: A Review Based on Findings from Epidemiologic Studies. Int J Mol Sci. 2016 Aug 12;17(8):1328. doi: 10.3390/ijms17081328[↩]
- Tembunde Y, Ge S, Turney K, Driscoll M. Scurvy: A Diagnosis Not to Be Missed. Cureus. 2022 Dec 28;14(12):e33050. doi: 10.7759/cureus.33050[↩]
- Hall SL, Greendale GA. The relation of dietary vitamin C intake to bone mineral density: results from the PEPI study. Calcif Tissue Int. 1998 Sep;63(3):183-9. doi: 10.1007/s002239900512[↩]
- Smith A, Di Primio G, Humphrey-Murto S. Scurvy in the developed world. CMAJ. 2011 Aug 9;183(11):E752-5. doi: 10.1503/cmaj.091938[↩]
- Velandia B, Centor RM, McConnell V, Shah M. Scurvy is still present in developed countries. J Gen Intern Med. 2008 Aug;23(8):1281-4. doi: 10.1007/s11606-008-0577-1[↩]
- Pangan AL, Robinson D. Hemarthrosis as initial presentation of scurvy. J Rheumatol. 2001 Aug;28(8):1923-5.[↩]
- Hirschmann JV, Raugi GJ. Adult scurvy. J Am Acad Dermatol. 1999 Dec;41(6):895-906; quiz 907-10. doi: 10.1016/s0190-9622(99)70244-6[↩][↩][↩][↩]
- Leggett J, Convery R. Images in clinical medicine. Scurvy. N Engl J Med. 2001 Dec 20;345(25):1818. doi: 10.1056/NEJMicm010202[↩]
- Blanchard MS, Romero JM, Hoang MP. Case records of the Massachusetts General Hospital. Case 1-2014. A 32-year-old man with loss of vision and a rash. N Engl J Med. 2014 Jan 9;370(2):159-66. doi: 10.1056/NEJMcpc1214217[↩]
- Chang CY, Rosenthal DI, Mitchell DM, Handa A, Kattapuram SV, Huang AJ. Imaging Findings of Metabolic Bone Disease. Radiographics. 2016 Oct;36(6):1871-1887. doi: 10.1148/rg.2016160004[↩]
- Stephen R, Utecht T. Scurvy identified in the emergency department: a case report. J Emerg Med. 2001 Oct;21(3):235-7. doi: 10.1016/s0736-4679(01)00377-8[↩]
- Bates CJ. Bioavailability of vitamin C. Eur J Clin Nutr 1997;51 (Suppl 1):S28-33. https://www.ncbi.nlm.nih.gov/pubmed/9023477?dopt=Abstract[↩]
- U.S. Department of Agriculture, Agricultural Research Service. 2011. USDA National Nutrient Database for Standard Reference, Release 24. Nutrient Data Laboratory Home Page, https://www.ars.usda.gov/northeast-area/beltsville-md/beltsville-human-nutrition-research-center/nutrient-data-laboratory/[↩]
- Hoffman FA. Micronutrient requirements of cancer patients. Cancer. 1985;55 (1 Suppl):295-300. https://www.ncbi.nlm.nih.gov/pubmed/3917362?dopt=Abstract[↩]
- Deicher R, Hörl WH. Vitamin C in chronic kidney disease and hemodialysis patients. Kidney Blood Press Res 2003;26:100-6. https://www.ncbi.nlm.nih.gov/pubmed/12771534%20?dopt=Abstract[↩]
- Reuler JB, Broudy VC, Cooney TG. Adult scurvy. JAMA. 1985 Feb 8;253(6):805-7.[↩]
- Montalto M, Porceddu E, Pero E, Lupascu A, Gallo A, De Simone C, Nucera E, Aruanno A, Giarretta I, Pola R, Landolfi R. Scurvy: A Disease not to be Forgotten. Nutr Clin Pract. 2021 Oct;36(5):1063-1067. doi: 10.1002/ncp.10616[↩][↩]
- Scurvy. https://emedicine.medscape.com/article/125350-overview#a7[↩]
- Tveden-Nyborg P, Lykkesfeldt J. Does vitamin C deficiency result in impaired brain development in infants? Redox Rep. 2009;14(1):2-6. doi: 10.1179/135100009X392412[↩]
- An HIV-Infected Man with Odynophagia and Rash, Clinical Infectious Diseases, Volume 41, Issue 5, 1 September 2005, Pages 686–688, https://doi.org/10.1086/432575[↩]
- Emadi-Konjin P, Verjee Z, Levin AV, Adeli K. Measurement of intracellular vitamin C levels in human lymphocytes by reverse phase high performance liquid chromatography (HPLC). Clin Biochem. 2005 May;38(5):450-6. doi: 10.1016/j.clinbiochem.2005.01.018[↩]
- Khalife R, Grieco A, Khamisa K, Tinmouh A, McCudden C, Saidenberg E. Scurvy, an old story in a new time: The hematologist’s experience. Blood Cells Mol Dis. 2019 May;76:40-44. doi: 10.1016/j.bcmd.2019.01.004[↩]
- Levavasseur M, Becquart C, Pape E, Pigeyre M, Rousseaux J, Staumont-Sallé D, Delaporte E. Severe scurvy: an underestimated disease. Eur J Clin Nutr. 2015 Sep;69(9):1076-7. doi: 10.1038/ejcn.2015.99[↩]
- Choh CT, Rai S, Abdelhamid M, Lester W, Vohra RK. Unrecognised scurvy. BMJ. 2009 Sep 17;339:b3580. doi: 10.1136/bmj.b3580[↩]
- Hearing SD. Refeeding syndrome. BMJ. 2004 Apr 17;328(7445):908-9. doi: 10.1136/bmj.328.7445.908[↩]
- Mehanna HM, Moledina J, Travis J. Refeeding syndrome: what it is, and how to prevent and treat it. BMJ. 2008 Jun 28;336(7659):1495-8. doi: 10.1136/bmj.a301[↩]
- Nutrition support in adults. Quality standard [QS24]. https://www.nice.org.uk/guidance/qs24[↩]
- Maxfield L, Crane JS. Vitamin C Deficiency. [Updated 2022 Oct 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK493187[↩][↩][↩]
- De Luna RH, Colley BJ 3rd, Smith K, Divers SG, Rinehart J, Marques MB. Scurvy: an often forgotten cause of bleeding. Am J Hematol. 2003 Sep;74(1):85-7. doi: 10.1002/ajh.10354[↩]
- Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: National Academy Press, 2000. https://nap.nationalacademies.org/catalog/9810/dietary-reference-intakes-for-vitamin-c-vitamin-e-selenium-and-carotenoids[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Curhan GC, Willett WC, Rimm EB, Stampfer MJ. A prospective study of the intake of vitamins C and B6, and the risk of kidney stones in men. J Urol. 1996 Jun;155(6):1847-51.[↩]
- Curhan GC, Willett WC, Speizer FE, Stampfer MJ. Intake of vitamins B6 and C and the risk of kidney stones in women. J Am Soc Nephrol. 1999 Apr;10(4):840-5. doi: 10.1681/ASN.V104840[↩]
- Taylor EN, Stampfer MJ, Curhan GC. Dietary factors and the risk of incident kidney stones in men: new insights after 14 years of follow-up. J Am Soc Nephrol. 2004 Dec;15(12):3225-32. doi: 10.1097/01.ASN.0000146012.44570.20[↩]
- Lee SH, Oe T, Blair IA. Vitamin C-induced decomposition of lipid hydroperoxides to endogenous genotoxins. Science. 2001 Jun 15;292(5524):2083-6. doi: 10.1126/science.1059501[↩]
- Podmore ID, Griffiths HR, Herbert KE, Mistry N, Mistry P, Lunec J. Vitamin C exhibits pro-oxidant properties. Nature. 1998 Apr 9;392(6676):559. doi: 10.1038/33308[↩]
- Carr, A. and Frei, B. (1999), Does vitamin C act as a pro-oxidant under physiological conditions?. The FASEB Journal, 13: 1007-1024. https://doi.org/10.1096/fasebj.13.9.1007[↩]