Contents
What is marjoram
Marjoram (Origanum majorana L.) commonly known as sweet marjoram from the family Lamiaceae, is a perennial herb that is native to Mediterranean region and cultivated in many countries of Asia, North Africa, and Europe, for example, Spain, Hungary, Portugal, Germany, Egypt, Poland, and France 1. Marjoram (Origanum majorana L.) grows up to 30 to 60 cm. Marjoram is a perennial bushy plant. It has oblique rhizome, hairy shrub like stalks, opposite dark green oval leaves and white or red flowers in clustered bracts. The leaves are whole, larger ones being fragmented, oblate to broadly elliptical 2. In some Middle Eastern countries, marjoram is synonymous with oregano, and there the names sweet marjoram and knotted marjoram are used to distinguish it from other plants of the genus Origanum. Marjoram plant is widely used as a garnish and is used for different medicinal purposes in traditional and folklore medicine of different countries. Sweet marjoram has been used for variety of diseases in traditional and folklore medicines, including ocular disorder, nasopharyngeal disorders, asthma, cold, coughs, cramps, depression, dizziness, gastrointestinal disorders, hay fever, headache, toothache, and sinus congestion and as a diuretic and to promote menstruation 3 and for cardiac, rheumatologic, and neurological disorders 1.
Various compounds have been identified in sweet marjoram. Also, different pharmacological activities have been attributed to this plant. Essential oil containing monoterpene hydrocarbons and oxygenated monoterpenes as well as phenolic compounds are chemical constituents isolated and detected in marjoram. Wide range of pharmacological activities including antioxidant, hepatoprotective, cardioprotective, anti-platelet, gastroprotective, antibacterial and antifungal, antiprotozoal, antiatherosclerosis, anti-inflammatory, antimetastatic, antitumor, antiulcer, and anticholinesterase inhibitory activities have been reported from this plant in modern medicine 1.
Figure 1. Marjoram (sweet marjoram)
Figure 2. Dried marjoram (marjoram herb)
 Marjoram uses
Marjoram uses
Traditional medicine uses
Ethnomedicinal uses of sweet marjoram in different countries are shown in Table 1. The parts of sweet marjoram that are used in folklore medicine are dried leaves, leaves extract, and essential oil. Marjoram leaves have been claimed to have antimicrobial and emmenagogue (a substance that stimulates or increases menstrual blood flow) properties and be useful for treatment of respiratory and gastrointestinal problems 2. Marjoram has been used in Morocco as an antihypertensive plant 4. The marjoram essential oil has been used for pains, gastrointestinal problems, and respiratory tract disorders 4.
Table 1. Traditional medicine uses of marjoram
| Region | Plant Part Used | Traditional Uses |
|---|---|---|
| Iran 5 | Leaves | Antimicrobial, antiseptic, antidote, carminative, antitussive and used for gastrointestinal disorder, head cool, sniffle, vision performance, otitis, melancholia accompanied by flatulence, unilateral facial paralysis, headache, epilepsy, cataract, weakness of sight, ear pain, dyspnea, cardiac pain, dysrhythmia, cramp, obstruction of large intestine, emmenagogue, strangury, dropsy, spondilolysthesis, groin pain, back pain, fatigue, freckle, migraine |
| Azerbaijan 6 | Essential oil | Flatulence, nervousness, diuretic, sedative |
| England 2 | Leaves | Cold, bronchial coughs, asthmatic whooping |
| Egypt 2 | Leaves | Cold, chill |
| India 7 | Essential oil | Toothache, soothe joints, muscular pain |
| Austria 8 | Leaves | Gastrointestinal tract diseases, infections |
| Turkey 9 | Essential oil | Asthma, indigestion, headache, rheumatism |
| Morocco 10 | Leaves | Hypertension |
Phytochemical constituents of marjoram
Table 2. Structure and phytochemical category of compounds isolated from different parts of sweet marjoram.
| Compound | Chemical Category | Part/Extract |
|---|---|---|
| α-Pinene | Monoterpene hydrocarbon | Essential oil11 |
| β-Pinene | Monoterpene hydrocarbon | Essential oil11 |
| ρ-Cymene | Monoterpene hydrocarbon | Essential oil12 |
| Camphene | Monoterpene hydrocarbon | Essential oil11 |
| α-Phellandrene | Monoterpene hydrocarbon | Essential oil 12 |
| β-Phellandrene | Monoterpene hydrocarbon | Essential oil 12 |
| γ-Terpinene | Monoterpene hydrocarbon | Essential oil13 |
| d-Limonene | Monoterpene hydrocarbon | Essential oil 14 |
| α-Terpinene | Monoterpene hydrocarbon | Essential oil 13 |
| Terpinolene | Monoterpene hydrocarbon | Essential oil 12 |
| β-Myrcene | Monoterpene hydrocarbon | Essential oil 12 |
| 2-Carene | Monoterpene hydrocarbon | Essential oil 15 |
| β-Ocimene | Monoterpene hydrocarbon | Essential oil 15 |
| Sabinene | Monoterpene hydrocarbon | Essential oil 2 |
| α-Thujene | Monoterpene hydrocarbon | Essential oil 12 |
| Carvone | Monoterpene hydrocarbon | Essential oil 12 |
| Citronellol | Monoterpene hydrocarbon | Essential oil 14 |
| Terpinen-4-ol | Oxygenated monoterpene | Essential oil 13 / Leaf 16 |
| cis-Sabinene hydrate | Oxygenated monoterpene | Essential oil 13 |
| trans-Sabinene hydrate | Oxygenated monoterpene | Essential oil 13 |
| Linalool | Oxygenated monoterpene | Leaf 16 / Essential oil 14 |
| Thymol | Oxygenated monoterpene | Essential oil 17 |
| α-Terpineol | Oxygenated monoterpene | Essential oil 13 |
| Linalyl acetate | Oxygenated monoterpene | Essential oil 12 |
| Carvacrol | Oxygenated monoterpene | Essential oil 14 |
| 1,8-Cineol | Oxygenated monoterpene | Essential oil 15 |
| Fenchyl alcohol | Oxygenated monoterpene | Essential oil 15 |
| Piperitol | Oxygenated monoterpene | Essential oil 15 |
| trans-Carveol | Oxygenated monoterpene | Essential oil 15 |
| cis-Carveol | Oxygenated monoterpene | Essential oil 15 |
| Anethole | Oxygenated monoterpene | Essential oil 15 |
| Geraniol | Oxygenated monoterpene | Essential oil 14 |
| α-Terpinyl acetate | Oxygenated monoterpene | Essential oil 12 |
| Geranyl acetate | Oxygenated monoterpene | Essential oil 15 |
| α-Cubebene | Sesquiterpene hydrocarbon | Essential oil 15 |
| Longicyclene | Sesquiterpene hydrocarbon | Essential oil 15 |
| Copaene | Sesquiterpene hydrocarbon | Essential oil 15 |
| β-Longipinene | Sesquiterpene hydrocarbon | Essential oil 15 |
| β-Caryophyllene | Sesquiterpene hydrocarbon | Essential oil 15 |
| Aromadendrene | Sesquiterpene hydrocarbon | Essential oil 15 |
| α-Humulene | Sesquiterpene hydrocarbon | Essential oil 15 |
| β-Farnesene | Sesquiterpene hydrocarbon | Essential oil 15 |
| Alloaromadendrene | Sesquiterpene hydrocarbon | Essential oil 15 |
| α-Selinene | Sesquiterpene hydrocarbon | Essential oil 15 |
| ar-Curcumene | Sesquiterpene hydrocarbon | Essential oil 15 |
| Germacrene D | Sesquiterpene hydrocarbon | Essential oil 15 |
| Valencene | Sesquiterpene hydrocarbon | Essential oil 15 |
| α-Muurolene | Sesquiterpene hydrocarbon | Essential oil 15 |
| α-Farnesene | Sesquiterpene hydrocarbon | Essential oil 15 |
| Spathulenol | Sesquiterpene alcohol | Essential oil 12 |
| Caryophyllene oxide | Oxygenated sesquiterpene | Essential oil12 |
| Carnosic acid | Diterpenoid | Water extract 18 |
| Carnosol | Diterpenoid | Water extract 18 |
| Ursolic acid | Triterpenoid | Water extract 18 |
| Sinapic acid | Phenolic acid | Essential oil 2 |
| Vanillic acid | Phenolic acid | Hydroalcoholic extract 19 / Essential oil 2 |
| Ferulic acid | Phenolic acid | Hydroalcoholic extract 19 / Essential oil 2 |
| Caffeic acid | Phenolic acid | Hydroalcoholic extract 19 / Essential oil 2 |
| Syringic acid | Phenolic acid | Hydroalcoholic extract 19 / Essential oil 2 |
| ρ-Hydroxybenzoic acid | Phenolic acid | Hydroalcoholic extract 19 / Essential oil 2 |
| m-Hydroxybenzoic acid | Phenolic acid | Hydroalcoholic extract 19 |
| Coumarinic acid | Phenolic acid | Essential oil 2 |
| Gallic acid | Phenolic acid | Hydroalcoholic extract 19 |
| Neochlorogenic acid | Phenolic acid | Hydroalcoholic extract 19 |
| Protocatechuic acid | Phenolic acid | Hydroalcoholic extract 19 |
| Caftaric acid | Phenolic acid | Hydroalcoholic extract 19 |
| Rosmarinic acid | Phenolic acid | Ethyl acetate extract 9 / Essential oil 20 |
| Chlorogenic acid | Phenolic acid | Hydroalcoholic extract 19 |
| Cryptochlorogenic acid | Phenolic acid | Hydroalcoholic extract 19 |
| Coumaric acid | Phenolic acid | Hydroalcoholic extract 19 |
| Lithospermic acid | Phenolic acid | Water extract 21 |
| Methyl rosmarinate | Phenolic compound | Hydrophilic extract 22 |
| Hydroquinone | Phenolic compound | Ethyl acetate extract 9 / Essential oil 17 |
| Arbutin | Phenolic glycosides | Ethyl acetate extract 9 / Essential oil 17 |
| Methyl arbutin | Phenolic glycoside | Essential oil 17 |
| Vitexin | Phenolic glycoside | Essential oil 17 |
| Orientinthymonin | Phenolic glycoside | Essential oil 17 |
| Hesperetin | Flavonoid | Ethyl acetate extract 9 |
| Catechin | Flavonoid | Hydroalcoholic extract 19 |
| Quercetin | Flavonoid | Hydroalcoholic extract 19 |
| Kaempferol | Flavonoid | Hydroalcoholic extract 19 |
| Naringenine | Flavonoid | Hydroalcoholic extract 19 |
| Eriodictyol | Flavonoid | Hydroalcoholic extract 19 |
| Diosmetin | Flavonoid | Essential oil 17 |
| Luteolin | Flavonoid | Essential oil 17 |
| Apigenin | Flavonoid | Essential oil 17 |
| 5,6,3′-Trihydroxy-7,8,4′-trimethoxyflavone | Flavonoid | Ethyl acetate extract 9 |
| Kaempferol-3-O-glucoside | Flavonoid glycoside | Hydroalcoholic extract 19 |
| Quercetin-3-O-glucoside | Flavonoid glycoside | Hydroalcoholic extract 19 |
| Narigenin-O-hexoside | Flavonoid glycoside | Hydroalcoholic extract 19 |
| Apigenin-glucuronide | Flavonoid glycoside | Water extract 23 |
| Rutin | Flavonoid glycoside | Hydroalcoholic extract 24 |
| Luteolin-7-O-β-glucuronide | Flavonoid glycoside | Hydrophilic extract 22 |
| Eugenol | Phenyl propene | Essential oil 14 |
| Ethyl cinnamate | Ester | Essential oil 14 |
| Sitosterol | Phytosterol | Essential oil 17 |
| Oleanolic acid | Fatty acid | Essential oil 17 |
| Vitamin A | Vitamin | Essential oil 2 |
| Vitamin C | Vitamin | Essential oil 2 |
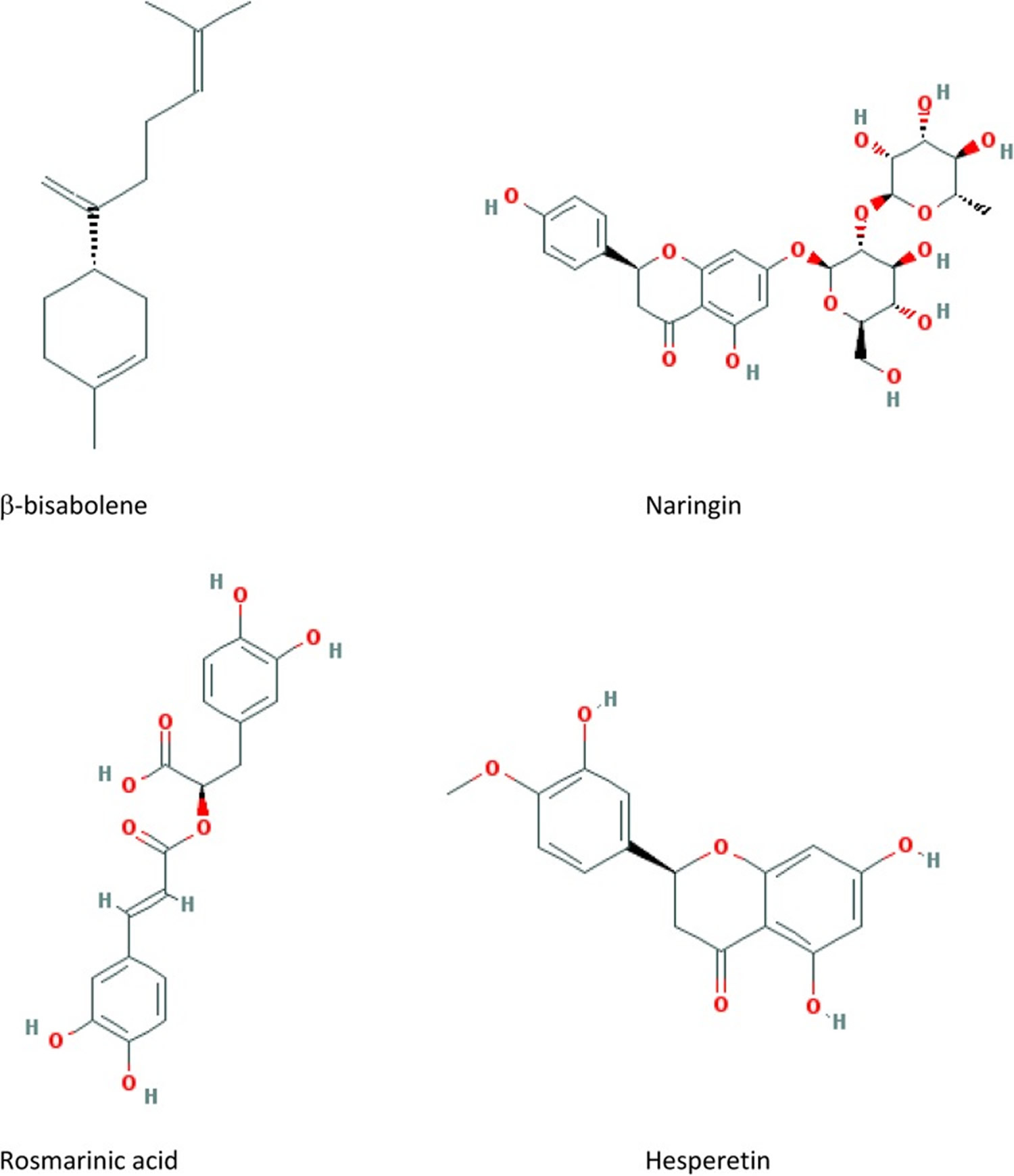
Figure 3. Marjoram active compounds
[Source 1]Marjoram phenolic compounds
Vanillic acid, gallic acid, ferulic acid, caffeic acid, syringic acid, p- and m-Hydroxybenzoic acid, coumaric acid, neochlorogenic acid, protocatechuic acid, chlorogenic acid, cryptochlorogenic acid, caftaric acid are phenolic acids that have been detected in hydroalcoholic extract of leaves of sweet marjoram 25. Rosmarinic acid, sinapic acid, vanillic acid, ferulic acid, caffeic acid, syringic acid, p- and m-hydroxybenzoic acid, and coumarinic acid have been identified in essential oil of sweet marjoram 2. Arbutin, methyl arbutin, vitexin, and orientinthymonin have been reported to be the most predominant phenolic glycosides in essential oil of sweet marjoram.10 Hesperetin, catechin, quercetin, kaempferol, naringenine, eriodictyol, diosmetin, luteolin, and apigenin are the most abundant flavonoids detected in sweet marjoram10,21 and kaempferol-3-O-glucoside, quercetin-3-O-glucoside, narigenin-O-hexoside, and rutin are flavonoid glycosides identified in sweet marjoram 25.
Antioxidant properties of marjoram
It has been suggested that phenolic compounds from marjoram, such as flavonoids and phenolic acids, might exert anti-inflammatory properties (Table 3) 26. In this regard, Mueller et al. 27 evaluated the anti-inflammatory activity of marjoram hydrophilic extracts on lipopolysaccharide (LPS)-stimulated RAW 264.7 macrophage cells. Pre-treatment of the cells with with the extracts from marjoram at 500 μg/mL and 200 μg/mL the levels of IL-6 were reduced 20% and 17%, respectively, while the iNOS expression was diminished 66%. Even though Mueller et al. 27 did not identify the compounds in the marjoram extracts, they mention that diosmetin, apigenin, luteolin and rosmarinic acid are the compounds to most likely be present in the hydrophilic extracts, so these molecules might be responsible for the activity of marjoram extracts.
Table 3. Summary of the antioxidant capacity of flavonoids and phenolic acids of Marjoram
| Extract | Compounds | Plant Part | Antioxidant Assay | Reference |
| Methanol microwave-assisted | Rosmarinic and caffeic acid, apigenin, rutin | Aerial parts | TPC, DPPH, CUPRAC | 28 |
| Aqueous, methanol | Rosmarinic and caffeic acids | Leaves | TPC, DPPH, β-carotene bleaching | 29 |
| Methanol | Rosmarinic acid, eriodictyol, naringenin, hispidulin, cirsimaritin | Leaves, commercial herbs | TPC, ORAC | 30 |
| Methanol | Rosmarinic acid, epigallocatechin, quercetin, apigenin | Not specified | DPPH, FRAP | 31 |
| Ethanol | Chlorogenic, ferulic, p-coumaric, p-hydroxybenzoic, protocatechuic, rosmarinic and syringic acids, quercetin | Not specified | TPC, DPPH, ABTS | 32 |
Pharmacological Activities of Marjoram
Table 4. Pharmacological properties of marjoram in detail
| harmacological Activity | Plant part / Extract | Method | Result | Active Constituent |
|---|---|---|---|---|
| Antioxidant 18 | Ethanol, n-hexane, supercritical CO2 and water extract of herb | DPPH method and chemiluminometric method | Antioxidant activities of all extracts | Ursolic acid, carnosic acid, carnosol |
| Antioxidant 34 | Essential oil | DPPH reduction test | Low antioxidant activity with EC50 values >250μg/mL | — |
| Antioxidant 35 | Essential oil | (1) DPPH assay (2) Percent inhibition in linoleic acid system (3) Bleaching of β-carotene | 1)IC50 of 89.2 µg/ml 2) 72.8% inhibition of linoleic acid oxidation 3)showed slow rate of color depletion | — |
| Antioxidant 9 | Ethyl acetate extract and isolated compounds | DPPH | Significant antioxidant activities from extract and isolated compounds with IC50 of 2.77 and 1.92 µg/mL, respectively | Hydroquinone |
| Antioxidant 36 | Essential oil / Water extract | ABTS + reducing power were examined for their effect against lipid oxidation in comparison to a tea water extract by measurement of the oil stability index | Remarkable capacity in retarding lipid oxidation with oil stability index 13.9 hours | Bound forms of phenolic compounds such as hydroxycinnamic acid and flavonoids |
| Antioxidant 19 | Hydroalcoholic extract | ABTS + radical decolorization and DPPH assay | Significant antioxidant capacity with 0.84 and 0.33 mmol TE/g DW, respectively | Polyphenolic compounds |
| Antioxidant 37 | Essential oil | Glutathione level and lipid peroxidation content as malondialdehyde in the testis, liver and brain in ethanol treatment male albino rat (ethanol induced reproductive disturbances and oxidative damage in different organs and lipid peroxidation due to the formation of free radicals) | Co-administration of the extract resulted in minimizing the hazard effects of ethanol toxicity on male fertility, liver and brain tissues | — |
| Antioxidant 38 | Essential oil | DPPH, .OH, H2O2, reducing power and lipid peroxidation | IC50 values of 58.67, 67.11, 91.25, 78.67, and 68.75 µg/mL, respectively | — |
| Antioxidant 39 | Water extract | DPPH | High antioxidant capacity | Phenolic compounds |
| Antioxidant 40 | Isolated metabolite | Amyloid β–induced oxidative injury in PC12 nerve cells by MTT, LDH, and trypan blue assays | ↓ Amyloid β–induced neurotoxic effect | Ursolic acid |
| Antioxidant 41 | Plant extract | DPPH and ferric ion reducing antioxidant power assays | A direct, positive, and linear relationship between antioxidant activity and total phenolic content of extract | Rosmarinic acid |
| Antimicrobial 42 | Dried whole plant/oil/leaves aqueous extract | MIC | Better antimicrobial activity of essential oil rather than water extract; inhibition of yeast and lactic acid bacteria by essential oil at a concentration of 5 ppm | — |
| Antimicrobial 43 | Essential oil | ND | The most susceptible organisms were Beneckea natriegens, Erwinia carotovora, and Moraxella sp. and Aspergillus niger | — |
| Antimicrobial 44 | n-Hexane extract, aqueous ethanol, ethanolic ammonia extract | Disk-diffusion method for bacteria and serial dilution method for protozoa | n-Hexane extract showed the highest antibacterial activity and the ethanolic ammonia extract reduced the number of viable Pentatrichomonas hominis trophozoites by 50% at 160 µg/ml | — |
| Antimicrobial 45 | Methanol extract | Filter paper disk diffusion method | Considerable activity against Aspergillus niger, Fusarium solani, and Bacillus subtilis with zone of inhibition 40, 28 and 42 mm, respectively | — |
| Antimicrobial 35 | Essential oil | (1) Disk diffusion (2) Resazurin microtitre-plate | (1) Large zone of inhibition (16.5-27.0 mm) (2) Small MIC against Staphylococcus aureus, Bacillus cereus, B subtilis, Pseudomonas aeruginosa, Salmonella poona, Escherichia coli (40.9-1250.3 μg/mL) | — |
| Antimicrobial 13 | Essential oil | Agar diffusion method | Active against Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, and Klebsiella pneumoniae with inhibition zone of 16, 12, 15, and 13 mm, respectively | cis-Sabinene hydrate |
| Antimicrobial 34 | Essential oil | Microdilution | Inhibitory activity against Staphylococcus aureus and Streptococcus pyogenes with MICs of 125 and 250 μg/mL, respectively | — |
| Antimicrobial 34 | Essential oil | Diffusion assay | Growth inhibitory activity against dermatophytes | — |
| Antimicrobial 46 | Methanol extract of leaves | Zone of inhibition | Inhibitory activity against Escherichia coli with 16 mm diameter zone of inhibition | — |
| Anti-inflammatory 47 | Essential oil | THP-1 human macrophage cells activated by LPS or human ox-LDL, and the cytokine secretion and gene expression, in vitro | Suppression of production of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6 and IL-10) and COX-2 and NFκB gene expression | Sabinene hydrate, terpineol |
| Anticancer 35 | Essential oil | MTT assay | Cytotoxic effect against different cancer cell type, such as MCF-7, LNCaP, NIH-3T3 with IC50 s of 70.0, 85.3, 300.5 µg/ml respectively | — |
| Anticancer 48 | Ethanol, methanol and water extract | MTT assay, trypan blue dye exclusion, AO/EB staining and fluorescence microscopical analysis and DNA fragmentation analysis | Significant cytotoxic activity of ethanolic extract on fibrosarcoma cancer cell line HT-1080 and least toxicity on normal human lymphocytes | — |
| Anticancer 49 | Plant extract | Nonradioactive cytotoxicity assay on human lymphoblastic leukemia cell line Jurkat | ↓ Viability of cells with increase of concentration of plant extract. Induction of apoptosis through upregulation of p53 protein levels and downregulation of Bcl-2α. Strong radical scavenging activity | — |
| Anticancer 50 | Ethanol extract | (1) Matrigel invasion assays (2) Gelatin zymography assay (3) Chick embryo tumor growth assay | (1) Significant inhibition of migration and invasion of the MDA-MB-231 cells. Induction of homotypic aggregation of cells associated with an up regulation of E-cadherin protein and decrease the adhesion of cells to HUVECs and inhibition of transendothelial migration of cells through TNF-α-activated HUVECs (2) Suppression of activities of MMP-2 and MMP-9 (3) Inhibition of tumor growth and metastasis | — |
| Anticancer 9 | Ethyl acetate extract and isolated compounds | BrdU cell proliferation enzyme-linked immunosorbent assay and xCELLigence assay against C6 and HeLa cell lines | Strong antiproliferative activities against C6 and HeLa cells | Hesperetin, Hydroquinone |
| Antiplatelet 51 | Methanol extract of leaves | Adhesion, aggregation and protein secretion of the activated platelet to laminin-coated plates | 40% inhibition of platelet adhesion to laminin-coated wells by ethanol extract at concentration of 200 µg/mL | — |
| Antiplatelet 52 | Methanol extract | Platelet aggregation induced by collagen; ADP, arachidonic acid and thrombin | Strong inhibition of platelet aggregation induced by ADP, arachidonic acid and thrombin | Arbutin |
| Antiulcer 53 | Ethanol extract | Hypothermic restraint stress-, indomethacin-, and necrotizing agents–induced ulcers and pylorus ligated Shay rat-model | ↓ Incidence of ulcers, basal gastric secretion and acid output. replenishment of the depleted gastric wall mucus and nonprotein sulfhydryls contents and ↓ malondialdehyde | — |
| Gastric secretory activity 54 | Plant extract | Acid and pepsin secretions in normal Wistar rats | ↑ Basal acid and pepsin secretions | — |
| Cardioprotective activity 55 | Leaves powder and aqueous extract | Isoproterenol-induced myocardial infarction in rats | Alleviation of erythrocytosis, granulocytosis, thrombocytosis, ↓ clotting time, ↑ relative heart weight, ↓ myocardial oxidative stress and the leakage of heart enzymes. inhibition of NO production and lipid peroxidation in heart tissues | — |
| Hepatoprotective activity 17 | Essential oil | Pralletrin-induced oxidative stress in rats (prallethrin caused a significant decrease in the activity of SOD, CAT, and GST in liver of rats) | Depletion of serum marker enzymes and replenishment of antioxidative status | — |
| Antiacetylcholinesterase activities 56 | Essential oil | ND | IC50 value was 36.40 µg/mL | — |
| Anticholinesterase activity 57 | Ethanol extract | In vitro | The Ki value was 6 pM, and IC50 value was 7.5 nM | Ursolic acid |
| Hormonal activity and regulation of menstrual cycle 58 | Water extract | 25 patients were received marjoram tea or a placebo tea twice daily for 1 month. Hormonal and metabolic parameters measured, including FSH, LH, progesterone, oestradiol, total testosterone, DHEA-S, fasting insulin and glucose | ↓ DHEA-S and fasting insulin levels | — |
Abbreviations: ABTS: 2, 2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid); ADP, adenosine diphosphate; CAT, catalase; COX, cyclooxygenase; DHEA-S, dehydroepiandrosterone-sulfate; DPPH, 1,1-diphenyl-2-picryl-hydrazyl; DW, dry weight; EC, effective concentration; FSH, follicle-stimulating hormone; GSH, glutathione S-transferase; IC, inhibitory concentration; IL, interleukin; LDH, lactate dehydrogenase; LH, luteinizing hormone; MIC, minimum inhibitory concentration; MMP, matrix metalloproteinase; MTT, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; ND, not determined; NO, nitric oxide; PCOS, polycystic ovary syndrome; SOD, superoxide dismutase; TE, trolox equivalent; TNF, tumor necrosis factor.
[Source 1]Antimicrobial Activity
Dried whole marjoram plant and its essential oil and water extract of leaves have demonstrated antimicrobial effect and essential oil was more active against lactic acid bacteria and yeasts than water extract 16. Essential oil showed inhibitory activity against various pathogenic bacteria and fungi, including Beneckea natriegens, Erwinia carotovera, Moraxella, Aspergillus, Staphylococcus aureus, Streptococcus pyogenes, Bacillus cereus, B subtilis, Pseudomonas aeruginosa, Salmonella poona, Escherichia coli, and dermatophytes 59. Methanol extract of sweet marjoram exhibited antimicrobial activity against E, Aspergillus niger, Fusarium solani, and Bacillus subtilis 45. The ethanolic ammonia extract reduced the number of viable Pentatrichomonas hominis trophozoites 44. cis-Sabinene hydrate in essential oil of sweet marjoram have been claimed to be responsible for antibacterial effect 13.
Anti-inflammatory Activity
Sabinene hydrate and terpineol in essential oil of sweet marjoram suppressed the production of Tumor necrosis factor-α (TNFα), interleukin 1β (IL-1β), IL-6, and IL-10 inhibited cyclooxygenase 2 (COX2) and NFκB gene expression 60.
Anti-cancer properties of marjoram
Several factors are involved in the onset of cancer such as age, alcohol, cancer-causing substances, diet, hormones, obesity, radiation, tobacco, etc.; and they may play a direct or indirect role in the development and progressions of different types of cancers. The National Cancer Institute 61 states that in test tube and in animal studies have shown that the increased presence of antioxidants prevents free radical damage that has been associated with cancer development. Plant foods are the most significance source of natural antioxidants; from which, flavonoids and phenolic acids have attracted the most attention as potential therapeutic agents against cancer. Shukla and Gupta 62 summarized that the potential anticancer properties of flavonoid and phenolic acid as demonstrated by laboratory studies are due to different mechanisms of action, including antioxidation, induction of detoxification enzymes and inhibition of bioactivation enzymes, estrogenic and anti-estrogenic activity, antiproliferation, cell cycle arrest and apoptosis, promotion of differentiation, regulation of host immune function and inhibition of angiogenesis and metastasis. 5,6,3′-Trihydroxy-7,8,4′-trimethoxyflavone, hesperetin, hydroquinone, arbutin and rosmarinic acid were isolated from the water-soluble ethyl acetate extract of aerial parts of marjoram 63. Hesperetin isolated from Origanum majorana has shown better antiproliferative activity than 5-fluoroacil against Rattus norvegicus brain glioma (C6) and and cervical epithelial carcinoma (HeLa) cell proliferation 63. Ethanol extract of plant have shown significant cytotoxicity against fibrosarcoma cancer cell line, promoting cell cycle arrest and apoptosis of the metastatic breast cell and inhibited the migration and invasion of the MDA-MB-231 cells 64. Hesperetin and hydroquinone isolated from sweet marjoram extract have revealed strong antiproliferative activity 9. The results showed that the marjoram extract and isolated compounds exhibited significant antioxidant activities. Hence marjoram plant has the potential to be a natural antioxidant in the food industry and an anticancer drug 63.
Antiplatelet Activity
Methanol extract of sweet marjoram leaves inhibit adhesion of platelet to laminin-coated plate 65 and strongly inhibited platelet aggregation induced by adenosine diphosphate (ADP), arachidonic acid, and thrombin. Arbutin is responsible for this activity 52.
Antiulcerogenetic Effect
Ethanol extract of sweet marjoram significantly decreased the incidence of ulcers, basal gastric secretion, and acid output and replenished the depleted gastric wall mucus 53.
Cardioprotective and Hepatoprotective Activity
Leave powder and extract significantly alleviated erythrocytosis, granulocytosis, thrombocytosis, increase heart weight, and myocardial infarction oxidative stress in isoproterenol treated albino rats 55. Essential oil of sweet marjoram depleted serum marker enzymes and replenished antioxidant status in hepatic of rat 17.
Anticholinesterase inhibitory activity
Essential oil and ethanol extract of sweet marjoram have exhibited acetylcholinesterase (AChE) inhibitor activity 38. Ursolic acid (3 beta-Hydroxyurs-12-en-28-oic acid) is responsible for this effect 66. Acetylcholinesterase (AChE) inhibitors, which enhance cholinergic transmission by reducing the enzymatic degradation of acetylcholine, are the only source of compound currently approved for the treatment of Alzheimer’s Disease 66. This study 66 demonstrated that the ursolic acid of marjoram appeared to be a potent acetylcholinesterase (AChE) inhibitor in Alzheimer’s Disease.
Regulation of menstrual cycle
Sweet marjoram tea significantly reduced dehydroepiandrosterone-sulphate (DHEA-S) and was useful in treatment of polycystic ovary syndrome 67. Twenty-five patients were assigned to receive marjoram tea or a placebo tea twice daily for 1 month (intervention group: n = 14; placebo group: n = 11) 67. The hormonal and metabolic parameters measured at baseline, as well as after the intervention, were: follicle-stimulating hormone, luteinising hormone, progesterone, oestradiol, total testosterone, dehydroepiandrosterone-sulphate (DHEA-S), fasting insulin and glucose, homeostasis model assessment for insulin resistance and glucose to insulin ratio. Marjoram tea significantly reduced dehydroepiandrosterone-sulphate (DHEA-S) and fasting insulin levels by a mean of 1.4 (0.5) μmol/L and 1.9 (0.8) μU/mL, respectively. In comparison to the placebo group, the change was only significant for DHEA-S but not for insulin. Homeostasis model assessment for insulin resistance was not reduced significantly in the intervention group, although the change was significant compared to the placebo group. The results obtained in the that study show the beneficial effects of marjoram tea on the hormonal profile of polycystic ovary syndrome (PCOS) women because it was found to improve insulin sensitivity and reduce the levels of adrenal androgens. Further research is needed to confirm these results and to investigate the active components and mechanisms contributing to such potential beneficial effects of marjoram herb.
Marjoram essential oil
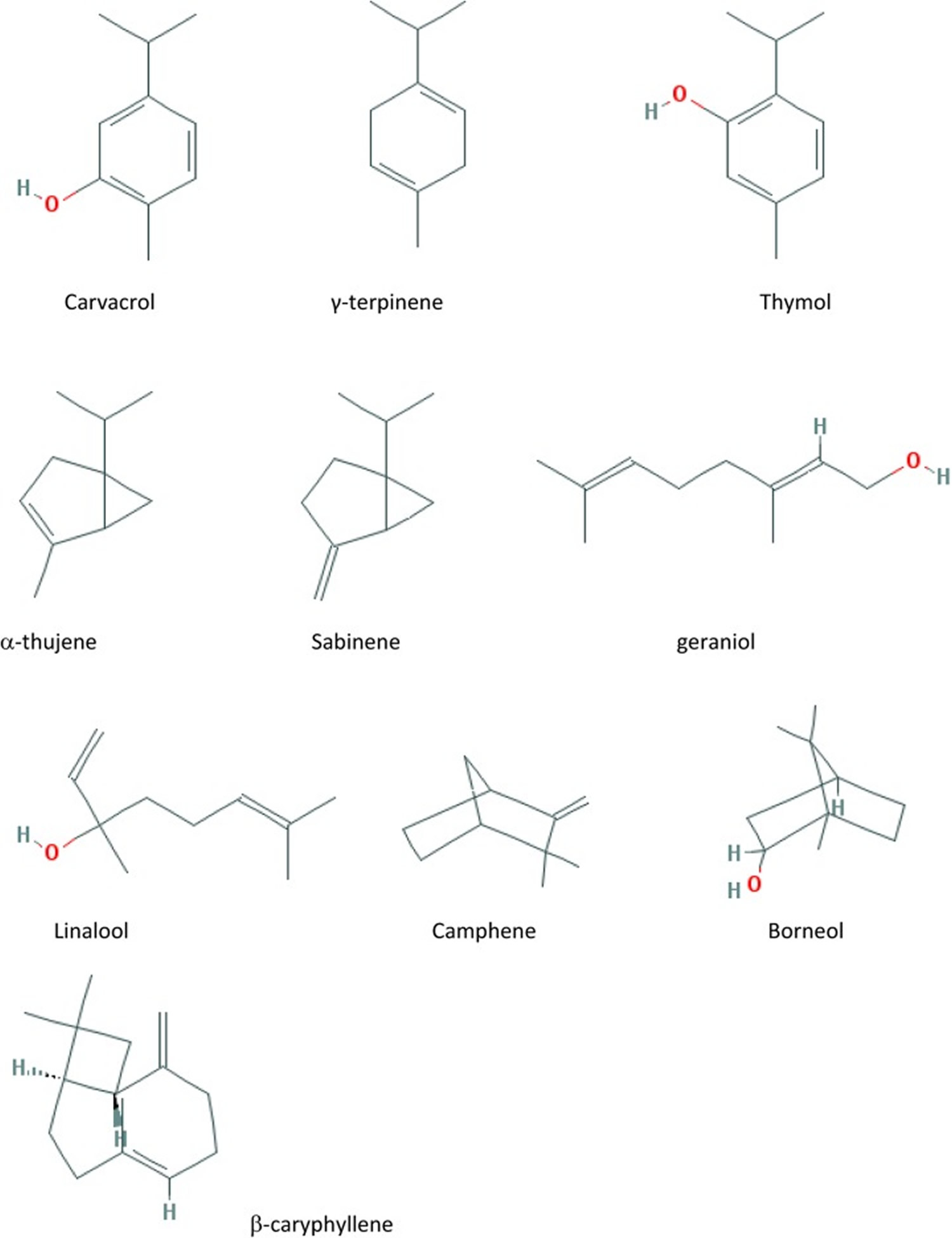
Monoterpene hydrocarbons, including α and β-pinene, camphene, sabinene, α- and β- phellandrene, ρ-cymene, limonene, β-ocimene, γ-terpinene, terpinolene, α-terpinene, carvone, and citronellol have been detected in marjoram essential oil 14. Terpinene 4-ol and cis-sabinene hydrate are 2 main oxygenated monoterpenes isolated from marjoram 13. Linalool, linalyl acetate, α-terpineol, trans- and cis-carveol, thymol, anethole, geraniol, and carvacrol are other oxygenated compounds identified in essential oil 42 and leaves of marjorama 11.
The analysis of the chemical composition of marjoram essential oil samples obtained from different geographical locations indicates that the biological activity is directly related to the concentration of the marjoram essential oil components, which may vary according to the region 68, 69. Moreover, season, climate, stage of plant development at harvest, and the technique of extraction of the product may influence the quantity of the plant compounds 70.
Fifteen compounds were identified in the marjoram essential oil (Table 5). The most abundant compounds were γ-terpinene (25.73%), α-terpinene (17.35%), terpinen-4-ol (17.24%), and sabinene (10.8%). This chemical profile is in accordance with what is reported in the literature, with some quantitative variations. Rodrigues et al. 71 and Vági et al. 72 also reported the presence of terpenes as the major components of the marjoram essential oil. Usually, terpinen-4-ol and γ-terpinene are described as the most abundant compounds in marjoram essential oil and sabinene and α-terpinene are also observed 73.
Table 5. Chemical composition of Marjoram essential oil
| Compound | RIa | % |
|---|---|---|
| α-Thujene | 921 | 3.96 |
| α-Pinene | 927 | 1.24 |
| Sabinene | 966 | 10.80 |
| Myrcene | 986 | 2.08 |
| α-Phellandrene | 1000 | 1.70 |
| α-Terpinene | 1012 | 17.35 |
| o-Cymene | 1019 | 2.24 |
| β-Phellandrene | 1023 | 7.05 |
| γ-Terpinene | 1053 | 25.73 |
| Terpinolene | 1082 | 3.76 |
| N.I.b | 1097 | 1.06 |
| Terpinen-4-ol | 1174 | 17.24 |
| Trans-sabinene hydrate acetate | 1248 | 0.13 |
| Linalool acetate | 1251 | 1.38 |
| Terpinenen-4-ol acetate | 1293 | 0.88 |
| (Z)-caryophyllene | 1404 | 2.72 |
| N.I.b | 1480 | 0.67 |
| Total identified | 98.26 |
Note: aRelative retention index experimentally determined against n-alkanes on Durabond-DB5 column. bCompound not identified.
Marjoram essential oil uses
Marjoram essential oil have shown significant results in inhibiting the growth of bacteria and fungi and the synthesis of microbial metabolites 75. Because of its antioxidant effects 76, marjoram essential oil or marjoram extract can be used in the prevention of central nervous system disorders 77. Marjoram essential oil was also able to partially prevent the ethanol-induced decline in sperm quality, testosterone levels, and the weight of reproductive organs in male rats 78. Previous studies have reported the potential use of marjoram ethanolic extract as anticancer agent 79, whereas the marjoram tea extract has been shown to have immunostimulant, antigenotoxic and antimutagenic properties 80. These activities are attributed to the chemical composition, which is characterized as rich in flavonoids and terpenoids – see Tables 2, 3 and 4 above 72.
Marjoram Toxicity
Acute toxicity test has demonstrated a large margin of safety of marjoram extract in mice. Emmenagogue (a substance that stimulates or increases menstrual blood flow) properties of sweet marjoram should be of concerned during pregnancy. Marjoram essential oil must not be used by lactating and pregnant women 81.
Summary
Sweet marjoram is a medicinal plant with various proven pharmacological properties, including antioxidant, antibacterial, hepatoprotective, cardioprotective, antiulcer, anticoagulant, anti-inflammatory, antiproliferative, and antifungal activities. The flowering stems are the medicinal parts. Their constituents include 1% to 2% of an essential oil with a containing terpinenes and terpinols, plus tannins, bitter compounds, carotenes, and vitamin C. These substances give sweet marjoram stomachic, carminative, antispasmodic, and weak sedative properties. Monoterpene hydrocarbons (such as α-pinene, β-pinene, camphene, and γ-terpinene), oxygenated monoterpenes particularly terpinene-4-ol, cis-sabinene hydrate and terpineol, phenolic compounds particularly flavonoids (such as apigenin, hesperetin, quercetin, kaempferol), and phenolic glycosides (such as arbutin) are the active components isolated and detected in marjoram. Figure 3 shows the structure of some main active compounds. Various bioactive compounds have been isolated and identified in O majorana, whereas many active compounds for the traditional medicine uses have not been completely evaluated in clinical trials.
Due to marjoram’s emmenagogue properties, marjoram essential oil must not be used by lactating and pregnant women 81.
- Bina F, Rahimi R. Sweet Marjoram: A Review of Ethnopharmacology, Phytochemistry, and Biological Activities. Journal of Evidence-based Complementary & Alternative Medicine. 2017;22(1):175-185. doi:10.1177/2156587216650793. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5871212/[↩][↩][↩][↩][↩][↩][↩]
- Charles DJ. Marjoram sweet. In: Antioxidant Properties of Spices, Herbs and other Sources. New York, Springer; 2013:393–399.[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Effects of marjoram volatile oil and grape seed extract on ethanol toxicity in male rats. El-Ashmawy IM, Saleh A, Salama OM. Basic Clin Pharmacol Toxicol. 2007 Nov; 101(5):320-7. https://www.ncbi.nlm.nih.gov/pubmed/17910615/[↩]
- Erenler R, Sen O, Aksit H, et al. Isolation and identification of chemical constituents from Origanum majorana and investigation of antiproliferative and antioxidant activities. J Sci Food Agric. 2016;96:822–836 https://www.ncbi.nlm.nih.gov/pubmed/25721137[↩][↩]
- Aghili MH. In: Rahimi R, Shams Ardekani MR, Farjadmand F, editors. , eds. Makhzan-al-Advia. Tehran, Iran: Tehran University of Medical Sciences; 2009:735.[↩]
- Alakbarov F. Aromatic herbal baths of the ancients. HerbalGram American Botanical Council. 2003;57:40–49.[↩]
- http://www.alwaysayurveda.com/origanum%20majorana/[↩]
- Vogl S, Picker P, Mihaly-Bison J, et al. Ethnopharmacological in vitro studies on Austria’s folk medicine—an unexplored lore in vitro anti-inflammatory activities of 71 Austrian traditional herbal drugs. J Ethnopharmacol. 2013;149:750–771[↩]
- Erenler R, Sen O, Aksit H, et al. Isolation and identification of chemical constituents from Origanum majorana and investigation of antiproliferative and antioxidant activities. J Sci Food Agric. 2016;96:822–836[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Tahraoui A, El-Hilaly J, Israili ZH, Lyoussi B. Ethnopharmacological survey of plants used in the traditional treatment of hypertension and diabetes in south-eastern Morocco (Errachidia province). J Ethnopharmacol. 2007;110:105–117.[↩]
- El-Moursi A, Talaat IM, Balbaa LK. Physiological effect of some antioxidant polyphenols on sweet marjoram (Majorana hortensis) plants. Nusantara Biosci. 2012;4:11–15[↩][↩][↩][↩]
- Lis A, Piter S, Gora J. A comparative study on the content and chemical composition of essential oils in commercial aromatic seasonings. Herba Polonica. 2007;53:21–26.[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Ramos S, Rojas LB, Lucena ME, et al. Chemical composition and antibacterial activity of Origanum majorana L. essential oil from the Venezuelan Andes. J Essential Oil Res. 2011;23(5):45–49[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- El-Moursi A, Talaat IM, Balbaa LK. Physiological effect of some antioxidant polyphenols on sweet marjoram (Majorana hortensis) plants. Nusantara Biosci. 2012;4:11–15.[↩][↩][↩][↩][↩][↩][↩][↩]
- Hussain AI, Anwar F, Rasheed Sh, et al. Composition, antioxidant and chemotherapeutic properties of the essential oils from two Origanum species growing in Pakistan. Rev Bras Farmacogn. 2011;21(6). doi:10.1590/S0102-695X2011005000165.[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Charai M, Mosaddak M, Faid M. Chemical composition and antimicrobial activities of two aromatic plants: Origanum majorana L. and O. compactum Benth. J Essential Oil Res. 1996;8:657–664[↩][↩][↩]
- Mossa AT, Refaie AA, Ramadan A, Bouajila J. Amelioration of prallethrin-induced oxidative stress and hepatotoxicity in rat by the administration of Origanum majorana essential oil. Biomed Res Int. 2013;2013:859085 doi:10.1155/2013/859085[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Vagi E, Rapavi E, Hadolin M, et al. Phenolic and triterpenoid antioxidants from Origanum majorana L. herb and extracts obtained with different solvents J Agric Food Chem. 2005;53:17–21[↩][↩][↩][↩]
- Vallverdu Queralt A, Regueiro J, Rinaldi Alvarenga JF, et al. Characterization of the phenolic and antioxidant profiles of selected culinary herbs and spices: caraway, turmeric, dill, marjoram and nutmeg. Food Sci Technol (Campinas). 2015;35(1).doi:10.1590/1678-457X.6580.[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Triantaphyllou K, Blekas G, Boskoua D. Antioxidative properties of water extracts obtained from herbs of the species Lamiaceae. Int J Food Sci Nutr. 2001;52:313–317.[↩]
- Kaiser A, Carle R, Kammerer DR. Effects of blanching on polyphenol stability of innovative paste-like parsley (Petroselinum crispum (Mill.) Nym ex A. W. Hill) and marjoram (Origanum majorana L.) products. Food Chem. 2013;138:1648–1656.[↩]
- Fecka I, Turek S. Determination of polyphenolic compounds in commercial herbal drugs and spices from Lamiaceae: thyme, wild thyme and sweet marjoram by chromatographic techniques. Food Chem. 2008;108:1039–1053[↩][↩]
- Kaiser A, Carle R, Kammerer DR. Effects of blanching on polyphenol stability of innovative paste-like parsley (Petroselinum crispum (Mill.) Nym ex A. W. Hill) and marjoram (Origanum majorana L.) products. Food Chem. 2013;138:1648–1656[↩]
- Kozlowska M, Laudy AE, Starosciak BJ. Antimicrobial and antiprotozoal effect of sweet marjoram (Origanum majorana L.). Acta Sci Pol Hortorum Cultus. 2010;9:133–141[↩]
- Vallverdu Queralt A, Regueiro J, Rinaldi Alvarenga JF, et al. Characterization of the phenolic and antioxidant profiles of selected culinary herbs and spices: caraway, turmeric, dill, marjoram and nutmeg. Food Sci Technol (Campinas). 2015;35(1).doi:10.1590/1678-457X.6580[↩][↩]
- Chen Y.-S., Yu H.-M., Shie J.-J., Cheng T.-J.R., Wu C.-Y., Fang J.-M., Wong C.-H. Chemical constituents of Plectranthus amboinicus and the synthetic analogs possessing anti-inflammatory activity. Bioorgan. Med. Chem. 2014;22:1766–1772. doi: 10.1016/j.bmc.2014.01.009[↩]
- Mueller M., Hobiger S., Jungbauer A. Anti-inflammatory activity of extracts from fruits, herbs and spices. Food Chem. 2010;122:987–996. doi: 10.1016/j.foodchem.2010.03.041[↩][↩]
- Çelik S.E., Tufan A.N., Bekdeşer B., Özyürek M., Güçlü K., Apak R. Identification and determination of phenolics in Lamiaceae species by UPLC-DAD-ESI-MS/MS. J. Chromatogr. Sci. 2017;55:291–300. doi: 10.1093/chromsci/bmw184[↩]
- Elansary H.O., Mahmoud E.A. Egyptian herbal tea infusions’ antioxidants and their antiproliferative and cytotoxic activities against cancer cells. Nat. Prod. Res. 2015;29:474–479. doi: 10.1080/14786419.2014.951354[↩]
- Bower A.M., Hernandez L.M.R., Berhow M.A., de Mejia E.G. Bioactive compounds from culinary herbs inhibit a molecular target for type 2 diabetes management, dipeptidyl peptidase iv. J. Agric. Food Chem. 2014;62:6147–6158. doi: 10.1021/jf500639f[↩]
- Hossain M.B., Camphuis G., Aguiló-Aguayo I., Gangopadhyay N., Rai D.K. Antioxidant activity guided separation of major polyphenols of marjoram (Origanum majorana L.) using flash chromatography and their identification by liquid chromatography coupled with electrospray ionization tandem mass spectrometry. J. Sep. Sci. 2014;37:3205–3213. doi: 10.1002/jssc.201400597[↩]
- Vallverdú-Queralt A., Regueiro J., Alvarenga J.F.R., Martinez-Huelamo M., Leal L.N., Lamuela-Raventos R.M. Characterization of the phenolic and antioxidant profiles of selected culinary herbs and spices: Caraway, turmeric, dill, marjoram and nutmeg. Food Sc. Technol. (Campinas) 2015;35:189–195. doi: 10.1590/1678-457X.6580[↩]
- Gutiérrez-Grijalva EP, Picos-Salas MA, Leyva-López N, Criollo-Mendoza MS, Vazquez-Olivo G, Heredia JB. Flavonoids and Phenolic Acids from Oregano: Occurrence, Biological Activity and Health Benefits. Plants. 2018;7(1):2. doi:10.3390/plants7010002. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5874591/[↩]
- Guerra-Boone L, Alvarez-Roman R, Salazar-Aranda R, et al. Antimicrobial and antioxidant activities and chemical characterization of essential oils of Thymus vulgaris, Rosmarinus officinalis, and Origanum majorana from northeastern Mexico. Pak J Pharm Sci. 2015;28:363S–369S[↩][↩][↩]
- Hussain AI, Anwar F, Rasheed Sh, et al. Composition, antioxidant and chemotherapeutic properties of the essential oils from two Origanum species growing in Pakistan. Rev Bras Farmacogn. 2011;21(6). doi:10.1590/S0102-695X2011005000165[↩][↩][↩]
- Triantaphyllou K, Blekas G, Boskoua D. Antioxidative properties of water extracts obtained from herbs of the species Lamiaceae. Int J Food Sci Nutr. 2001;52:313–317[↩]
- El-Ashmawy IM, Saleh A, Osama M. Effects of marjoram volatile oil and grape seed extract on ethanol toxicity in male rats. Basic Clin Pharmacol Toxicol. 2007;101:320–327[↩]
- Mossa AT, Nawwar GA. Free radical scavenging and antiacetylcholinesterase activities of Origanum majorana L. essential oil. Hum Exp Toxicol. 2011;30:1501–1513[↩][↩]
- Chrpova D, Kourrimska L, Gordon MH, et al. Antioxidant activity of selected phenols and herbs used in diets for medical conditions. Czech J Food Sci. 2010;28:317–325[↩]
- Heo HJ, Cho HY, Hong B, et al. Ursolic acid of Origanum majorana L. reduces Abeta-induced oxidative injury. Mol Cells. 2002;13:5–11[↩]
- Hossain MB, Camphuis G, Aguilo-Aguayo I, et al. Antioxidant activity guided separation of major polyphenols of marjoram (Origanum majorana L.) using flash chromatography and their identification by liquid chromatography coupled with electrospray ionization tandem mass spectrometry. J Separation Sci. 2014;37:3205–3213[↩]
- Charai M, Mosaddak M, Faid M. Chemical composition and antimicrobial activities of two aromatic plants: Origanum majorana L. and O. compactum Benth. J Essential Oil Res. 1996;8:657–664.[↩][↩]
- Deans SG, Svoboda KP. The antimicrobial properties of marjoram (Origanum majorana L.) volatile oil. Flavour Frag J. 1990;5:187–190[↩]
- Kozlowska M, Laudy AE, Starosciak BJ. Antimicrobial and antiprotozoal effect of sweet marjoram (Origanum majorana L.). Acta Sci Pol Hortorum Cultus. 2010;9:133–141.[↩][↩]
- Leeja L, Thoppil JE. Antimicrobial activity of methanol extract of Origanum majorana L. (sweet marjoram). J Environ Biol. 2007;28:145–146[↩][↩]
- Bonjar GHS. Screening for antibacterial properties of some Iranian plants against Escherichia coli. Asian J Plant Sci. 2004;3:310–314.[↩]
- Arranz E, Jaime L, Lopez MC, et al. Supercritical fluid extraction as an alternative process to obtain essential oils with anti-inflammatory properties from marjoram and sweet basil. Ind Crop Prod. 2015;67:121–129.[↩]
- Rao Sh, Timsina B, Nadumane VK. Evaluation of the anticancer potentials of Origanum marjorana on fibrosarcoma (HT-1080) cell line. Asian Pac J Trop Dis. 2014;4:S389–S394.[↩]
- Abdel-Massih RM, Fares R, Bazzi S, et al. The apoptotic and anti-proliferative activity of Origanum majorana extracts on human leukemic cell line. Leuk Res. 2010;34:1052–1056[↩]
- Al Dhaheri Y, Attoub S, Arafat K, et al. Anti-metastatic and anti-tumor growth effects of Origanum majorana on highly metastatic human breast cancer cells: inhibition of NFκB signaling and reduction of nitric oxide production. PLoS One. 2013;8(7). doi:10.1371/journal.pone.0068808[↩]
- Yazdanparast R, Shahriyary L. Comparative effects of Artemisia dracunculus, Satureja hortensis and Origanum majorana on inhibition of blood platelet adhesion, aggregation and secretion. Vascul Pharmacol. 2008;48:32–37[↩]
- Okazaki K, Nakayama Sh, Kawazoe K. Antiaggregant effects on human platelets of culinary herbs. Phytother Res. 1998;12:603–605[↩][↩]
- Al-Howiriny T, Alsheikh A, Alqasoumi S, et al. Protective effect of Origanum majorana L. ‘Marjoram’ on various models of gastric mucosal injury in rats. Am J Chin Med. 2009;37:531–545[↩][↩]
- Rafsanjani FN, Shahrani M, Ardakani ZV, Ardakani MV. Marjoram increases basal gastric acid and pepsin secretions in rats. Phytother Res. 2007;21:1036–1038[↩]
- Ramadan G, El-Beih NM, Arafa NM, Zahra MM. Preventive effects of Egyptian sweet marjoram (Origanum majorana L.) leaves on hematological changes and cardiotoxicity in isoproterenol-treated albino rats. Cardiovasc Toxicol. 2013;13:100–109[↩][↩]
- Mossa AT, Nawwar GA. Free radical scavenging and antiacetylcholinesterase activities of Origanum majorana L. essential oil. Hum Exp Toxicol. 2011;30:1501–1513 https://www.ncbi.nlm.nih.gov/pubmed/21239482[↩]
- Chung YK, Heo HJ, Kim EK, et al. Inhibitory effect of ursolic acid purified from Origanum majorana L on the acetyl cholinesterase. Mol Cells. 2001;11:137–143.[↩]
- Haj-Husein I, Tukan S, Alkazaleh F. The effect of marjoram (Origanum majorana) tea on the hormonal profile of women with polycystic ovary syndrome: a randomized controlled pilot study. J Hum Nutr Diet. 2016;29:105–111. doi:10.1111/jhn.12290[↩]
- Ramos S, Rojas LB, Lucena ME, et al. Chemical composition and antibacterial activity of Origanum majorana L. essential oil from the Venezuelan Andes. J Essential Oil Res. 2011;23(5):45–49.[↩]
- Arranz E, Jaime L, Lopez MC, et al. Supercritical fluid extraction as an alternative process to obtain essential oils with anti-inflammatory properties from marjoram and sweet basil. Ind Crop Prod. 2015;67:121–129[↩]
- National Cancer Institute Antioxidants and Cancer Prevention. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/antioxidants-fact-sheet[↩]
- Shukla S., Gupta S. Apigenin and cancer chemoprevention. In: Watson R.R., Preedy V.R., editors. Bioactive Foods in Promoting Health: Fruits and Vegetables. Elsevier Academic Press Inc.; San Diego, CA, USA: 2010. pp. 663–689.[↩]
- Isolation and identification of chemical constituents from Origanum majorana and investigation of antiproliferative and antioxidant activities. Isolation and identification of chemical constituents from Origanum majorana and investigation of antiproliferative and antioxidant activities. https://onlinelibrary.wiley.com/doi/pdf/10.1002/jsfa.7155[↩][↩][↩]
- Rao Sh, Timsina B, Nadumane VK. Evaluation of the anticancer potentials of Origanum marjorana on fibrosarcoma (HT-1080) cell line. Asian Pac J Trop Dis. 2014;4:S389–S394[↩]
- Yazdanparast R, Shahriyary L. Comparative effects of Artemisia dracunculus, Satureja hortensis and Origanum majorana on inhibition of blood platelet adhesion, aggregation and secretion. Vascul Pharmacol. 2008;48:32–37.[↩]
- Chung YK, Heo HJ, Kim EK, et al. Inhibitory effect of ursolic acid purified from Origanum majorana L on the acetyl cholinesterase. Mol Cells. 2001;11:137–143. https://www.ncbi.nlm.nih.gov/pubmed/11355692[↩][↩][↩]
- Haj-Husein I, Tukan S, Alkazaleh F. The effect of marjoram (Origanum majorana) tea on the hormonal profile of women with polycystic ovary syndrome: a randomized controlled pilot study. J Hum Nutr Diet. 2016;29:105–111. doi:10.1111/jhn.12290 https://www.ncbi.nlm.nih.gov/pubmed/25662759[↩][↩]
- Ramos S., Rojas L. B., Lucena M. E., Meccia G., Usubillaga A. Chemical composition and antibacterial activity of Origanum majorana L. essential oil from the Venezuelan Andes. Journal of Essential Oil Research. 2011;23(5):45–49. doi: 10.1080/10412905.2011.9700481[↩]
- Busatta C., Vidal R. S., Popiolski A. S., et al. Application of Origanum majorana L. essential oil as an antimicrobial agent in sausage. Food Microbiology. 2008;25(1):207–211. doi: 10.1016/j.fm.2007.07.003 https://www.ncbi.nlm.nih.gov/pubmed/17993397[↩]
- Ncube B., Finnie J. F., Van Staden J. Quality from the field: the impact of environmental factors as quality determinants in medicinal plants. South African Journal of Botany. 2012;82:11–20. doi: 10.1016/j.sajb.2012.05.009[↩]
- Rodrigues M. R. A., Caramão E. B., Arce L., Ríos A., Valcárcel M. Determination of monoterpene hydrocarbons and alcohols in Majorana hortensis Moench by micellar electrokinetic capillary chromatographic. Journal of Agricultural and Food Chemistry. 2002;50(15):4215–4220. doi: 10.1021/jf011667n https://www.ncbi.nlm.nih.gov/pubmed/12105948[↩]
- Vági E., Simándi B., Suhajda Á., Héthelyi É. Essential oil composition and antimicrobial activity of Origanum majorana L. extracts obtained with ethyl alcohol and supercritical carbon dioxide. Food Research International. 2005;38(1):51–57. doi: 10.1016/j.foodres.2004.07.006[↩][↩]
- Jelali N., Dhifi W., Chahed T., Marzouk B. Salinity effects on growth, essential oil yield and composition and phenolic compounds content of marjoram (origanum majorana l.) leaves. Journal of Food Biochemistry. 2011;35(5):1443–1450. doi: 10.1111/j.1745-4514.2010.00465.x[↩]
- Dantas A dos S, Klein-Júnior LC, Machado MS, et al. Origanum majorana Essential Oil Lacks Mutagenic Activity in the Salmonella/Microsome and Micronucleus Assays. The Scientific World Journal. 2016;2016:3694901. doi:10.1155/2016/3694901. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5116495/[↩]
- Abdel-Massih R. M., Abraham A. Extracts of Rosmarinus officinalis, Rheum rhaponticum, and Origanum majorana exhibit significant anti-staphylococcal activity. International Journal of Pharmaceutical Sciences and Research. 2014;5:819–828.[↩]
- Roby M. H. H., Sarhan M. A., Selim K. A.-H., Khalel K. I. Evaluation of antioxidant activity, total phenols and phenolic compounds in thyme (Thymus vulgaris L.), sage (Salvia officinalis L.), and marjoram (Origanum majorana L.) extracts. Industrial Crops and Products. 2013;43(1):827–831. doi: 10.1016/j.indcrop.2012.08.029.[↩]
- Rezaie A., Jafari B., Mousavi G., et al. Comparative study of sedative, pre-anesthetic and anti-anxiety effect of Origanum majorana extract with diazepam on rats. Research Journal of Biological Sciences. 2011;6:611–614. doi: 10.3923/rjbsci.2011.611.614[↩]
- El-Ashmawy I. M., Saleh A., Salama O. M. Effects of marjoram volatile oil and grape seed extract on ethanol toxicity in male rats. Basic and Clinical Pharmacology and Toxicology. 2007;101(5):320–327. doi: 10.1111/j.1742-7835.2007.00125.x. https://www.ncbi.nlm.nih.gov/pubmed/17910615[↩]
- Al Dhaheri Y., Attoub S., Arafat K., et al. Anti-metastatic and anti-tumor growth effects of Origanum majorana on highly metastatic human breast cancer cells: inhibition of NFκB signaling and reduction of nitric oxide production. PLoS ONE. 2013;8(7) doi: 10.1371/journal.pone.0068808.e68808 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3707896/[↩]
- Khan J. A., Jalal J. A., Ioanndes C., Moselhy S. S. Impact of aqueous doash extract on urinary mutagenicity in rats exposed to heterocyclic amines. Toxicology and Industrial Health. 2013;29(2):142–148. doi: 10.1177/0748233711427053 https://www.ncbi.nlm.nih.gov/pubmed/22173956[↩]
- Ernst E. Herbal medicinal products during pregnancy: are they safe? BJOG. 2002;109:227–235. https://obgyn.onlinelibrary.wiley.com/doi/pdf/10.1111/j.1471-0528.2002.t01-1-01009.x[↩][↩]