Contents
What is Asherman syndrome
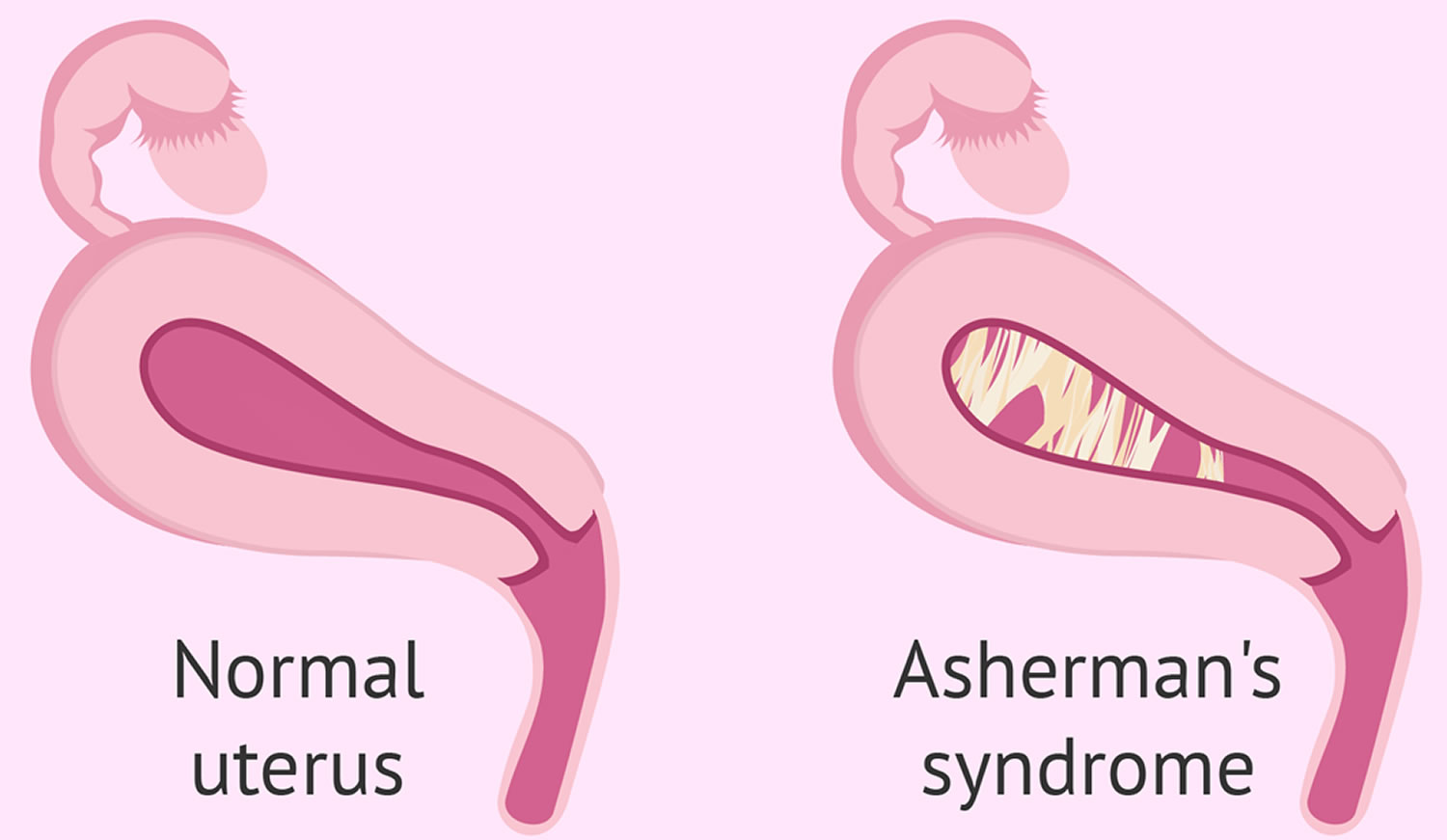
Asherman syndrome also known as intrauterine adhesions, is the formation of scar tissue inside the uterine cavity and/or cervix, leading to infertility or changes to your menstruation (e.g., amenorrhea, dysmenorrhea and oligomenorrhea) 1. In many cases the front and back walls of the uterus stick to one another. In other cases, adhesions only occur in a small portion of the uterus. The extent of the adhesions defines whether the case is mild, moderate, or severe. The adhesions can be thin or thick, spotty in location, or confluent. They are usually not vascular, which is an important attribute that helps in treatment.
Women with Asherman syndrome often experience reduced menstrual flow, increased cramping and abdominal pain, eventual cessation of menstrual cycles (amenorrhea), and, in many instances, infertility. Most often these symptoms are the result of severe inflammation of the lining of the uterus (endometriosis) that is caused by the development of bands of scar tissue that join parts of the walls of the uterus to one another, thus reducing the volume of the uterine cavity (intrauterine adhesions and synechiae). Endometrial scarring and intrauterine adhesions may occur as a result of surgical scraping or cleaning of tissue from the uterine wall (dilatation and curettage [D and C]), infections of the endometrium (e.g., tuberculosis), or other factors. Such changes may have an effect on implantation and may lead to infertility since the hypotrophic endometrium becomes unreceptive to an embryo resulting in recurrent pregnancy loss 2. Obstetrical complications and recurrent miscarriages have been reported with Asherman syndrome 3.
Asherman syndrome is also known as uterine atresia, amenorrhea traumatica, endometrial sclerosis, and intrauterine adhesions or synechiae 4. In most cases, Asherman syndrome occurs in women who have had several dilatation and curettage (D&C) procedures. A severe pelvic infection unrelated to surgery may also lead to Asherman syndrome. Intrauterine adhesions can also form after infection with tuberculosis or schistosomiasis 5. These infections are rare in the United States. Uterine complications related to these infections are even less common. The true incidence of Asherman syndrome remains unknown since the majority of the patients are asymptomatic 6.
Asherman syndrome is thought to be under-diagnosed because it is usually undetectable by routine diagnostic procedures such as an ultrasound scan. The condition is estimated to affect 1.5% of women undergoing a hysterosalpingogram (HSG) 7, between 5 and 39% of women with recurrent miscarriage 8, and up to 40% of patients who have undergone D&C for retained products of conception following childbirth or incomplete abortion 9.
Intrauterine adhesions can lead to partial or complete dysfunction of the endometrium with impairment of fertility and menstrual pattern (amenorrhea and hypomenorrhea) 10. When the adhesions are exclusively located in the lower uterine tract and functioning endometrium persists, Asherman syndrome can also cause severe pelvic pain and retrograde menstruation.
Pregnant or early pregnant uterus seems to be more susceptible to develop uterine scarring after curettage 10. Nonetheless any uterine insult or trauma following even less invasive surgical procedure can lead to intrauterine adhesions development.
The impact of the Asherman syndrome on pregnancy is well documented with a high rate of infertility, miscarriage, poor implantation following in vitro fertilization and abnormal placentation 11.
Although the first case of intrauterine adhesion was published in 1894 by Heinrich Fritsch 12, it was only after 54 years that a full description of Asherman syndrome was carried out by Israeli gynaecologist Joseph Asherman 10. Specifically, he identified this pathology in 29 women who showed amenorrhea with stenosis of internal cervical ostium 13. The author speculated that such a manifestation could be a consequence of endometrium trauma. Two years later, he published another case series of intrauterine adhesions, this time involving the uterine cavity and characterized by evident filling defects during hysterography 14.
The cause of Asherman syndrome is not clear 6; however, an event that causes damage to the endometrium can lead to the development of adhesions. Asherman syndrome most frequently occur after repeated dilatation and curettage (D&C), postpartum hemorrhage (PPH), and elective abortion 6. Additionally, Asherman syndrome may occur after a simple operation on the uterus like a cesarean section and myomectomy 15. Presently, Asherman syndrome is known to be Asherman syndromesociated with nontraumatic factors, for example, puerperal sepsis 16, infections such Asherman syndrome tuberculous endometritis, and even after a normal delivery 17.
Direct visualization of the uterus via hysteroscopy is the most reliable method for diagnosis. Hysteroscopic adhesiolysis is the treatment of choice for the management of intrauterine adhesions 18. Hysteroscopic adhesiolysis can be an effective treatment of Asherman syndrome, with an overall pregnancy rate of 40% following surgery 19.
Grades of Ashermans Syndrome Severity
Society for Hysteroscopy, 1989 20:
- I – Thin or filmy adhesions easily ruptured by hysteroscope sheath alone, cornual areas normal;
- II – Singular firm adhesions connecting separate parts of the uterine cavity, visualization of both tubal ostia possible, cannot be ruptured by hysteroscope sheath alone;
- IIa – Occluding adhesions only in the region of the internal cervical OS. Upper uterine cavity normal;
- III – Multiple firm adhesions connecting separate parts of the uterine cavity, unilateral obliteration of ostial areas of the tubes;
- IIIa – Extensive scarring of the uterine cavity wall with amenorrhea or hypomenorrhea;
- IIIb – Combination of III and IIIa;
- IV – Extensive firm adhesions with agglutination of the uterine walls. Both tubal ostial areas occluded
Valle and Sciarra’s 1988 classification
- Mild- Filmy adhesions composed of basal endometrium producing partial or complete uterine cavity occlusion;
- Moderate – Fibromuscular adhesions that are characteristically thick, still covered by endometrium that may bleed on division, partially or totally occluding the uterine cavity;
- Severe – Composed of connective tissue with no endometrial lining and likely to bleed upon division, partially or totally occluding the uterine cavity.
Donnez and Nisolle 1994 classification
- I – Central adhesions
- a) thin filmy adhesions (endometrial adhesions)
- b) myofibrous (connective adhesions)
- II – Marginal adhesions (always myofibrous or connective)
- a) wedge like projection
- b) obliteration of one horn
- III – Uterine cavity absent on hysterosalpingogram
- a) occlusion of the internal os (upper cavity normal)
- b) extensive agglutination of uterine walls (absence of uterine cavity – true Asherman’s)
Asherman syndrome symptoms
Most patients with Asherman’s syndrome present with scanty or absent menstrual blood flows (amenorrhea). In a few instances, the menstrual cycle may be normal. In some instances, the affected individual may experience an interrupted menstrual blood flow with substantial pain. Some patients have no periods but feel pain at the time that their period would normally arrive each month. This pain may indicate that menstruation is occurring but the blood cannot exit the uterus because the cervix is blocked by adhesions. Recurrent miscarriage and infertility could also be symptoms 21.
This may occur as a result of blockage of the cervix (the neck of the uterus) by adhesions. Recurrent miscarriages and/or infertility may also be signs of Asherman’s syndrome.
Menstrual cycle of a woman with Asherman syndrome
The hormonal influences remain the same; however, scar tissue inside the uterus will generally prevent the full scope of uterine changes. One theory suggests that the lack of endometrial growth is due to a complex biochemical feedback loop in which the uterus restricts the proliferation of endometrium. Others include significant damage to the basal layer of the endometrium (critical for the growth of the functional layer) or reduced blood flow to the endometrium either from fibrosis of the muscular layer of the uterus or because the scars which seal the uterus closed (partially or completely) reduce blood flow to the endometrium.
Hysteroscopy with lysis of the scar tissue can in many cases help restore normal uterine function in terms of endometrial response to hormonal stimulation. Unfortunately sometimes even after the scar tissue is resected the prior damage to the endometrium will lead to a chronically thin endometrium (i.e., it will undergo the correct proliferative and secretory changes, however full endometrial growth potential will not be recognized). This may be due to a complex chemical feedback mechanism not yet fully understood by medical science.
Thin endometrium is counteracted by a short course of sequential exogenous estrogen to encourage endometrial re-epithilialization (regrowth) and progesterone. In many cases once the uterus is mostly free of scar tissue this course of action can help restore more normal uterine function, and decrease the chance of rescarring.
In a few cases however the endometrial basal layer has been so badly denuded (scarred) that estrogen therapy will not help. This result is more common after fibroid removal surgery.
What causes Asherman syndrome
Asherman’s syndrome occurs when trauma to the endometrial lining triggers the normal wound-healing process, which causes the damaged areas to fuse together. Most commonly, intrauterine adhesions occur after a dilation and curettage (D&C) that was performed because of a missed or incomplete miscarriage, retained placenta with or without hemorrhage after a delivery, or elective abortion. Pregnancy-related D&Cs have been shown to account for 90% of Asherman’s Syndrome cases 22. Sometimes adhesions also occur following other pelvic surgeries such as Cesarean section, surgery to remove fibroids or polyps, or in the developing world, as a result of infections such as genital tuberculosis 23 and schistosomiasis 24.
There is a 25% risk of developing Asherman’s Syndrome from a D&C that is performed 2 to 4 weeks after delivery 25. Dilation and Curettages (D&C) may also lead to Asherman’s Syndrome in 30.9% of procedures for missed miscarriages 26 and 6.4% of procedures for incomplete miscarriages 22. The risk of Asherman syndrome increases with the number of D&Cs performed; after a single termination the risk is 16%, however, after 3 or more D&Cs, the risk increases to 32% 27. Each case of Asherman’s Syndrome is different, and the cause must be determined on a case-by-case basis. In some cases, Asherman’s Syndrome may have been caused by an “overly aggressive” D&C. However, this is not often considered to be the case. The placenta may have attached very deeply in the endometrium or fibrotic activity of retained products of conception could have occurred, both of which make it difficult to remove retained tissue.
There is a variant of Asherman syndrome called “Unstuck Asherman” or endometrial sclerosis that is more difficult to treat. In this condition, which may coexist with the presence of adhesions, the uterine walls are not stuck together. Instead, the endometrium has been denuded. Although curettage can cause this condition, it is more likely after uterine surgery, such as myomectomy. In these cases the endometrium, or at least its basal layer, has been removed or destroyed.
Sporadic inflammation of the mucous membrane lining the uterus (endometriosis), endometritis or endometriosis caused by a tuberculosis infection or certain other infectious diseases may also be causes of Asherman’s syndrome.
Intrauterine adhesions are also seen as a consequence of congenital uterine abnormalities, the use of IUD (intrauterine device) birth control devices and genetic predisposition 28. It is well-known that the endometrium is more susceptible to trauma in a pregnant uterus and the incidence of intrauterine adhesions following curettage for retained placental tissue is reported up to 40% 3 months after curettage 29. In patients who develop Asherman syndrome, curettage of a pregnant uterus has been shown as the cause in 64% of the cases.
Asherman syndrome prevention
Incidence of intrauterine adhesions might be lower following medical evacuation (e.g. misoprostol) of the uterus, thus avoiding any intrauterine instrumentation.
Also, the use of systematic antibiotics after D&C to avoid sub-clinical infections, likely a cofactor in the genesis of scarring, might be beneficial.
There is no evidence to suggest suction D&C is less likely to result in adhesions than sharp D&C. Cases of Asherman syndrome have been reported even following manual evacuation, and the rate of Asherman syndrome has not dropped since the introduction of suction D&C.
Asherman syndrome diagnosis
Unless the physician is careful, the diagnosis of Asherman’s syndrome may be needlessly overlooked. A simple X-ray of the uterus with a small tube placed in the cervix is usually diagnostic. However, many physicians will, in order to save time, use a small balloon catheter placed in the uterus. The latter technique will overlook a number of cases of Asherman syndrome. The gold standard for diagnosis uses a hysteroscope that pictures the interior of the uterus directly.
- Blood tests: Estrogen, luteinizing hormone (LH), progesterone, follicle-stimulating hormone (FSH), thyroid, prolactin to rule out hormonal problems affecting ovulation. Some women may not want to jump into hysteroscopy or hysterosalpingogram (HSG), which are not without some risks.
- Gold standard for diagnosis: diagnostic hysteroscopy
- Imaging tests:
- Ultrasound to measure endometrial thickness at ovulation and to check normal follicle development
- Sonohysterography (SHG) or hysterosalpingogram (HSG) these procedures are not as accurate as hysteroscopy, but will confirm patency of tubes if infertility is a problem
Asherman syndrome chances of pregnancy
Pregnancy success following Asherman syndrome treatment is around 90% for stage I, 60% for stage II and 30% for stage III with pregnancy after stage IV uncommon 30.
Pregnancy risks after treatment of Asherman syndrome include: small for dates, slightly higher incidence of miscarriage and preterm delivery, and 5% risk of major haemorrhagic complication at delivery due to placenta accreta.
Pregnancy with Untreated Asherman syndrome and the risks
For women who become pregnant BEFORE having had the chance to correct their Asherman syndrome
- If a woman gets pregnant with adhesions still in her uterus, the risks are different. Getting pregnant with adhesions remaining in the uterus is not recommended because the chances of miscarriage, problems with fetal growth and/or other serious complications of pregnancy are high.
- If you become pregnant before your Asherman’s has been treated, it is imperative that you speak to your doctor right away and that you be closely monitored. Even then, your risk of complications is significantly higher than if you wait and become pregnant after you are cleared by your doctor.
RECOMMENDED MONITORING FOR PREGNANCIES WITH ADHESIONS PRESENT IN UTERUS:
- Weeks 4 to 7: Regular human chorionic gonadotropin (hCG) and progesterone blood tests
- Weeks 6 to 10: Regular ultrasounds, the first at approximately around 6 weeks. This ultrasound will verify that the baby is in the right place (not in a fallopian tube) and is growing normally. Subsequent ultrasounds could be performed weekly in order to identify a normal heartbeat and to verify good interval growth of the baby and that all is going well. If a miscarriage happens, the doctor can get onto it straight away. There should be no delay as this can cause further damage to your uterus.
- Weeks 18 to 20: Regular bi-weekly scans in order to evaluate for an incompetent cervix which is often a risk for those with post-Asherman syndrome pregnancies because of multiple prior cervical dilatations. It involves performing ultrasounds in order to measure cervical length and to detect “funneling” of the fetal membranes. These ultrasound examinations need to be performed by someone with the skill needed to detect these changes which are often subtle. If placental abnormalities or inadequate fetal growth are not detected, the patient can be monitored by a routine schedule.
- Third trimester: An evaluation, often by MRI, in order to detect placenta accreta, increta or percreta. Continued assessment to identify IUGR (intrauterine growth restriction of the fetus) as well as placenta previa. If any of these complications are identified, you should be monitored more closely.
Pregnancy AFTER Asherman syndrome diagnosis
In some cases pregnancy may occur spontaneously and without complication depending on the severity of Asherman syndrome. In some cases where the endometrium has been badly denuded down to the basal layer a condition called “Unstuck Asherman’s” can occur and the endometrium will fail to respond to hormonal influence, even in the presence of exogenous estrogen. In this case surrogacy is the only viable option for a biological child.
In some cases pregnancy can occur with the aid of fertility drugs and/or treatments such as IVF (in vitro fertilization). Pregnancy after treatment for Asherman syndrome will greatly increase the chance of placental implantation issues such as placenta previa (low lying placenta growing over the cervix which mandates a cesarean section and which can be caused by previous scarring of the fundus of the uterus), placenta accreta (placenta attaches too deeply causing problems with placental removal and hemorrhage at the time of delivery), cervical incompetence secondary to multiple dilations of the cervix from multiple curettages and/or multiple hysteroscopies to treat the Asherman’s and abnormal fetal lie (the fetus lies is and grows in an abnormal position presumably due to abnormal and restricted uterine shape). These conditions can be detected during routine ultrasound.
A recent study by Roy et al. 19 reported pregnancy rates of 58% in mild Asherman, 30% in moderate Asherman, and 33.3% in severe cases of Asherman syndrome following hysteroscopic adhesiolysis. Furthermore, the live birth rate reported was 86.1%, the miscarriage rate 11.1% and the cumulative pregnancy rate showed that 97.2% of the patients conceived within 24 months postoperatively 19.
Studies evaluating obstetric complications in pregnant women with previous Asherman syndrome are sparse. A number of cases have been reported but large observational studies are lacking. In 1982, Schenker and Margalioth 31 found an incidence of placenta accreta in 13–14% of patients with previous Asherman syndrome. Roy et al. 19 reported an incidence of postpartum hemorrhage due to adherent placenta in 12.5% of the 89 women who had undergone hysteroscopic adhesiolysis due to Asherman syndrome. These numbers present a remarkably low incidence of abnormal placentation in a group of women, who all had known trauma of the endometrium (i.e., 87.5% did not have an adherent placenta and subsequent postpartum hemorrhage).
Conventional 2 dimensional ultrasonography is currently the best screening tool for detecting placenta accreta with a sensitivity of 77–90.7%, a specificity of 96–98%, a positive predictive value of 65–93%, and a negative predictive value of 98% 32. Another diagnostic tool for detection of abnormal placentation is Magnetic Resonance Imaging (MRI). MRI has a sensitivity of 80–85% and a specificity of 65–100% 33. MRI can be used in conjunction with conventional ultrasonography 34, and can be helpful in some cases, especially if the placenta is located on the posterior uterine wall. Though it is important to notice, that for cases of placenta accreta, MRI is not a good prognostic tool for changing of surgical management 35. Retrospective studies have shown that women with an antenatal diagnosis of placenta accreta have less blood loss and requirement for blood transfusion than women in whom the abnormal placentation was diagnosed during cesarean section 36.
Any patient with a previous history of intrauterine surgery or Asherman syndrome should be thoroughly examined by a skilled sonographer for possible abnormal placentation. In case of any suspicion of abnormal placentation, the patient should be scheduled for planned cesarean section with a set-up of skilled clinicians due to risk of severe postpartum hemorrhage.
In conclusion, it is important to be aware of the risk of a placenta accreta in patients with a previous history of Asherman syndrome and uterine scarring. Antenatal diagnosis is important to counsel the pregnant woman and to plan mode of delivery.
Pregnancy Risks in a Post-Ashermans Pregnancy
You should ask your doctor about the risks involved in a post-Asherman syndrome pregnancy. Also, note that the information provided here applies only to women who have had their adhesions removed. If a woman gets pregnant with adhesions still in her uterus, the risks are different. Getting pregnant with adhesions remaining in the uterus is usually not recommended.
What are the risks involved in a post-Asherman’s pregnancy?
The risks involved in a post-Asherman syndrome pregnancy vary, depending on how severe the Asherman’s was. If the Asherman syndrome was very mild, then after the adhesions are removed, pregnancy is statistically no riskier than for a woman who never had Asherman syndrome. Obviously this can vary in individual cases, but the statistics show no increased risk.
If the Asherman syndrome was moderate or severe, there is some increased risk in subsequent pregnancies.
The risks are:
- First-trimester miscarriage. This is a significant risk in any pregnancy, but it’s higher after Asherman syndrome .
- Placenta accreta. One study of 137 post-Asherman syndrome pregnancies found a 9% risk of placenta accreta. Placenta accreta means the placenta grows into the wall of the uterus. This isn’t harmful to the baby, but after the baby is born, when it’s time for the placenta to come out, if you have placenta accreta the placenta will be “stuck” and not come out. The doctor might have to do a D&C to remove it. In the worst cases of placenta accreta, in which they cannot get the placenta out without causing the mother to hemorrhage uncontrollably, they sometimes have to do a hysterectomy. Several members of the Asherman syndrome message board have had placenta accreta but most just needed a D&C, not a hysterectomy. Note that when they do the hysterectomy, they can usually just remove the uterus but leave the ovaries in place, which means you’ll continue to ovulate each month, and you won’t go through menopause until you reach the normal age for menopause. (Of course you won’t get a period, but that’s not the same as menopause.) Besides placenta accreta, placenta increta and placenta percreta are also risks, although they are extremely rare. means the placenta grows even more deeply and firmly into the uterine wall than placenta accreta. means the placenta grows all the way through the wall of the uterus and sometimes extends to nearby organs.
- Placenta previa. This means low-lying placenta. Normally the placenta should implant high up in your uterus but if the uterine walls aren’t in good condition (for example due to you having had Asherman syndrome ), sometimes the placenta implants lower, sometimes even covering the cervix. Placenta previa can increase your risk for bleeding during pregnancy, increase the risk of preterm delivery, and increase the risk of harm to mother and baby due to blood loss during delivery. However, if discovered early, more than 99% of mothers with placenta previa are just fine, and most of the babies come out of it just fine too. You could end up on bed rest for a substantial part of the pregnancy though. Fortunately the risk of placenta previa seems to be much lower than the risk of placenta accreta. Some of the A-list doctors think placenta previa is not a significant risk at all, if you’ve had all your adhesions removed. We have seen it occasionally on the Asherman’s message board though.
- Incompetent cervix (this is where the cervix dilates way too early in the pregnancy, resulting in loss of the baby or resulting in preterm delivery, depending on what week of pregnancy you’re in). Asherman’s doesn’t increase your risk of incompetent cervix, but having multiple D&Cs does increase the risk. Most doctors think having a lot of hysteroscopies doesn’t increase the risk, but a few doctors think it does. So it’s wise to be monitored for incompetent cervix if you get pregnant, just in case.
- Some perinatologists say that Asherman syndrome theoretically increases the risk of intrauterine growth retardation (IUGR), which is where the fetus doesn’t grow as well as it should, possibly due to placental insufficiency, but we have never seen a case of post- Asherman’s IUGR on the Asherman’s message board.
- Vasa Previa. This is a rare (1:3000), heartbreaking condition which occurs when one or more of the baby’s placental or umbilical blood vessels cross the entrance to the birth canal beneath the baby. When the cervix dilates or the membranes rupture, the unprotected vessels can tear, causing rapid fetal hemorrhage. When the baby drops in to the pelvis, the vessels can be compressed, compromising the baby’s blood supply and causing oxygen deprivation.
Non Reproductive Consequences of Asherman syndrome
The reproductive consequences of Asherman syndrome, including infertility, recurrent miscarriage, intrauterine growth restriction, placenta accreta and others, are well known. However, for all women with intrauterine scarring and amenorrhea, including those who may have completed childbearing, there are other concerns. Although the lack of menstrual periods could be secondary to hormonal abnormalities, it is more likely caused by either complete destruction of the uterine lining or by obstruction of the cervix or lower portion of the uterus; thus, menses are either retained in the uterus (leading to pelvic pain and a condition called hematometra) or flow into the abdominal cavity leading to endometriosis. Women with Asherman’s syndrome may develop uterine cancer, either before or after menopause. This risk is NOT increased and may be lower than in the general population. However, the usual warning sign of uterine cancer is excessive uterine bleeding: those with obstructed menstrual flow cannot have that symptom even if they harbor a uterine growth. Therefore, pelvic ultrasound should be a routine part of their annual gynecologic visit.
Asherman syndrome treatment
First-line treatment of Asherman syndrome is to remove the scar tissue and promote the growth of the endometrium to reduce the formation of new scar tissue. Hysteroscopic lysis of adhesions is the main method of treatment.
Hormone support therapy is used in almost all cases, except in stage I. Use of a stent to keep the uterine walls separated during the healing phase, is recommended in stages III or IV. Repeat intervention is common in stage III and IV. Increasingly early reintervention either with standard hysteroscopy, or most recently with flexible hysteroscopy, has been advocated.
Many physicians argue against the use of lasers or energy sources inside the uterus (this means removing scars with scissors rather than with energy-generating instruments such as resectoscopes or lasers, although not all surgeons agree with this) to remove the adhesions. These doctors claim that the use of small cutting devices is less likely to irritate the lining of the uterus or to cause infection. Adhesions have a tendency to reform, especially in more severe cases. There are different methods to prevent re-scarring after surgery for Asherman syndrome. Many surgeons prescribe estrogen supplementation to stimulate uterine healing and place a splint or balloon to prevent apposition of the walls during the immediate post-operative healing phase. Other surgeons recommend weekly in-office hysteroscopy after the main surgery to cut away any newly formed adhesions. As of yet, studies have not confirmed the method of treatment that is most likely to have a successful outcome, which would be one where the uterus/cervix remains scar-free and fertility is restored.
- You will need to have Hysteroscopic and possibly Laparascopic surgery to remove your adhesions. This is the most important phase of your treatment. Only a highly skilled surgeon with experience in AS should do this. Protecting your uterine lining is very important.
- After your surgery you will most likely have a balloon catheter inserted into your uterus, this is used to keep your uterine walls from adhering together during the healing process. Your doctor may want this to stay inside for 5-14 days. You will also take an antibiotic to prevent infection. Note, not all Dr.’s use a balloon.
- Once the balloon is removed you will be prescribed a regimen of estrogen and progesterone. The dose and length of this regimen will vary depending on your doctor.
- 2-3 months after your surgery you should have an HSG, SHG or diagnostic hysteroscopy to view the inside of your uterus and your fallopian tubes for remaining scar tissue.
- Subsequent surgery may be necessary.
Hormonal therapy is also used to encourage menstruation.
Treatment with estrogen has shown good results in preventing reformation of adhesions following hysteroscopic surgery for Asherman syndrome 37. Use of peroral estrogen gives better fertility and menstrual outcome when given in combination with ancillary treatment (IUD, balloon or hyaluronic acid) 32.
If you have healed from your surgery and your uterus is free of scar tissue your doctor may give you the “green light” to try and conceive. It is very important that this not be rushed and that your uterus is at least 90% free of scar tissue before getting pregnant. Some of the risks that you now face with carrying a child are: Placenta Previa, Placenta Accreta, Premature rupture of membranes and possibly incompetent cervix.
During your continued treatment your doctor may want to track your ovulation and measure your endometrial lining and follicles during ovulation. Your doctor may also suggest that you purchase a fertility monitor to pinpoint your ovulation day and schedule intercourse appropriately. Your doctor may also consider fertility medication. This is usually prescribed when you have a blocked tube or when blood tests indicate a hormonal imbalance. Fertility medication is not necessary for every woman with Asherman syndrome. If your doctor prescribes this for you, ask why and which type would be the best for you.
- Intrauterine adhesions. March CM. Obstet Gynecol Clin North Am. 1995 Sep; 22(3):491-505.[↩]
- March CM. Asherman’s syndrome. Semin. Reprod. Med. 2011;29:83–94.[↩]
- Posttraumatic intrauterine synechiae and pregnancy. Forssman L. Obstet Gynecol. 1965 Nov; 26(5):710-3.[↩]
- Management of Asherman’s syndrome. March CM. Reprod Biomed Online. 2011 Jul; 23(1):63-76. https://www.ncbi.nlm.nih.gov/pubmed/21549641/[↩]
- Asherman syndrome. https://medlineplus.gov/ency/article/001483.htm[↩]
- Baradwan S, Baradwan A, Al-Jaroudi D. The association between menstrual cycle pattern and hysteroscopic march classification with endometrial thickness among infertile women with Asherman syndrome. Chatzistamatiou. K, ed. Medicine. 2018;97(27):e11314. doi:10.1097/MD.0000000000011314. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6076072/[↩][↩][↩]
- Dmowski WP, Greenblatt RB. Asherman?s syndrome and risk of placenta accreta. Obstet Gynecol 1969; 34:288-299.[↩]
- Ventolini G, Zhang M, Gruber J. Hysteroscopy in the evaluation of patients with recurrent pregnancy loss: a cohort study in a primary care population. Surg Endosc 2004; 18:1782-1784.[↩]
- Westendorp ICD, Ankum WM, Mol BWJ, Vonk J. Prevalence of Asherman?s syndrome after secondary removal of placental remnants or a repeat curettage for incomplete abortion. Hum Reprod 1998; 13:3347-3350.[↩]
- Conforti A, Alviggi C, Mollo A, De Placido G, Magos A. The management of Asherman syndrome: a review of literature. Reproductive Biology and Endocrinology : RB&E. 2013;11:118. doi:10.1186/1477-7827-11-118. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3880005/[↩][↩][↩]
- Deans R, Abbott J. Review of intrauterine adhesions. J Minim Invasive Gynecol. 2010;11:555–569. doi: 10.1016/j.jmig.2010.04.016[↩]
- Amenorrhoea traumatica (atretica). ASHERMAN JG. J Obstet Gynaecol Br Emp. 1948 Feb; 55(1):23-30.[↩]
- Asherman JG. Amenorrhoea traumatica (atretica) J Obstet Gynaecol Br Emp. 1948;11:23–30. doi: 10.1111/j.1471-0528.1948.tb07045.x[↩]
- Asherman JG. Traumatic intrauterine adhesions. J Obstet Gynaecol Br Emp. 1950;11:892–896. doi: 10.1111/j.1471-0528.1950.tb06053.x.[↩]
- Orhue AA, Aziken ME, Igbefoh JO. A comparison of two adjunctive treatments for intrauterine adhesions following lysis. Int J Gynaecol Obstet 2003;82:49–56.[↩]
- Polishuk WZ, Anteby SO, Weinstein D. Puerperal endometritis and intrauterine adhesions. Int Surg 1975;60:418–20.[↩]
- Zondek R, Rozin S. Filling defect in the hysterogram simulating intrauterine synechae which disappear after denervation. Am J Obstet Gynecol 1964;88:123–7.[↩]
- March C, Israel R. Intrauterine adhesions secondary to elective abortion. Hysteroscopic diagnosis and management. Obstet Gynecol 1976;48:422–4.[↩]
- Roy KK, Baruah J, Sharma JB, Kumar S, Kachawa G. Singh N. Reproductive outcome following hysteroscopic adhesiolysis in patients with infertility due to Asherman’s syndrome. Arch. Gynecol. Obstet. 2010;281:355–361.[↩][↩][↩][↩]
- Grades of Ashermans Syndrome Severity. http://www.ashermans.org/information/ashermans-grades[↩]
- Valle RF, and Sciarra JJ. Intrauterine adhesions: Hystreoscopic diagnosis, classification, treatment and reproductive outcome. Am J Obstet 1988; 158:1459-1470.[↩]
- Schenker JG, Margalioth EJ. Intra-uterine adhesions: an updated appraisal. Fertility Sterility 1982; 37:593-610.[↩][↩]
- Netter AP, Musset R, Lambert A et al. Traumatic intrauterine synechiae: a common cause of menstrual insufficiency, sterility and abortion. Am J Obstet Gynecol 1956; 71:368.[↩]
- Krolikowski A, Janowski K, Larsen JV. Asherman syndrome caused by schistosomiasis.Obstet Gynecol. 1995; 85(5 Pt 2):898-9.[↩]
- Fedele L, Bianchi S, Frontino G. Septums and synechiae: approaches to surgical correction. Clin Obstet Gynecol 2006; 49:767-788.[↩]
- Adoni, A, Palti, Z, Milwidsky, A, and Dolberg, M. The incidence of intrauterine adhesions following spontaneous abortion. Int J Fertil 1982;27(2):117-8.[↩]
- Friedler S, Margalioth EJ, Karfka I, Yaffe H. Incidence of post-abortion intrauterine adhesions evaluated by hysteroscopy-a prospective study. Hum Reprod 1993; 8:442-4.[↩]
- March CM. Management of Asherman’s syndrome. Reprod. Biomed. Online. 2011;23:63–76.[↩]
- Westendorp IC, Ankum WM, Mol BW. Vonk J. Prevalence of Asherman’s syndrome after secondary removal of placental remnants or a repeat curettage for incomplete abortion. Hum. Reprod. 1998;13:3347–3350.[↩]
- Asherman’s syndrome: A rare infertility disorder – Medical Observer 12 April 2013. https://jeanhailes.org.au/contents/documents/Resources/Medical__health_articles/Medical_Observer/2013/Ashermans_12_April_2013.pdf[↩]
- Schenker JG. Margalioth EJ. Intrauterine adhesions: an updated appraisal. Fertil. Steril. 1982;37:593–610.[↩]
- D’Antonio F, Iacovella C. Bhide A. Prenatal identification of invasive placentation using ultrasound: systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2013;42:509–517.[↩][↩]
- Gielchinsky Y, Mankuta D, Rojansky N, Laufer N, Gielchinsky I. Ezra Y. Perinatal outcome of pregnancies complicated by placenta accreta. Obstet. Gynecol. 2004;104:527–530.[↩]
- Levine D, Hulka CA, Ludmir J, Li W. Edelman RR. Placenta accreta: evaluation with color Doppler US, power Doppler US, and MR imaging. Radiology. 1997;205:773–776.[↩]
- McLean LA, Heilbrun ME, Eller AG, Kennedy AM. Woodward PJ. Assessing the role of magnetic resonance imaging in the management of gravid patients at risk for placenta accreta. Acad. Radiol. 2011;18:1175–1180.[↩]
- Tikkanen M, Paavonen J, Loukovaara M. Stefanovic V. Antenatal diagnosis of placenta accreta leads to reduced blood loss. Acta Obstet. Gynecol. Scand. 2011;90:1140–6.[↩]
- Engelbrechtsen L, Langhoff-Roos J, Kjer JJ, Istre O. Placenta accreta: adherent placenta due to Asherman syndrome. Clinical Case Reports. 2015;3(3):175-178. doi:10.1002/ccr3.194. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4377250/[↩]