Contents
What is a bone graft
A bone graft is surgery to place new bone or bone substitutes into spaces around a broken bone or bone defects. Surgeons use bone grafts to repair and rebuild diseased bones in your hips, knees, spine, and sometimes other bones and joints. Bone graft can also repair bone loss caused by some types of fractures or cancers. Once your body accepts the bone graft, it provides a framework for growth of new, living bone.
A bone graft can be taken from the person’s own healthy bone (this is called an autograft). Autograft bone often comes from your ribs, hips or a leg. Currently autografts are the “gold standard” in bone substitution 1.
Or bone graft can be taken from frozen, donated bone (allograft) from another person. Most allograft bone comes from donors who have died. Tissue banks screen these donors and disinfect and test the donated bone to make sure it is safe to use.
In some cases, a manmade (synthetic) bone substitute is used.
Bone loss persists to be an important challenge in surgery, and many alternatives are available. Despite the improvement of research, human bone grafts persist to be the most effective bone substitutes to replace bone loss.
What is the risk of disease transmission with bone allografts?
An allograft is a graft between genetically dissimilar members of the same species. An allograft may be obtained from living donors who are having bone removed during surgery or cadaveric donors. Allograft material has been used for more than 30 years in periodontal therapy (bone graft in mouth).
There are several types of allografts:
- Fresh-frozen bone allograft
- Freeze-dried bone allograft
- Demineralized freeze-dried bone allograft
Both freeze-dried bone allograft and demineralized freeze-dried bone allograft materials are widely used in periodontal therapy and there are no reports of disease transmission during the 30-year history of using freeze-dried bone allografts 2. However, there have been four cases of human immunodeficiency virus (HIV) infection following procedures using fresh-frozen bone allografts. These cases involved surgeries of the spine, hip, and knee. It is important to note, however, that fresh-frozen and fresh bone allografts are not typically used in periodontal therapy. Also, when using freeze-dried bone allograft and demineralized freeze-dried bone allograft, the delay in processing ensures adequate time for testing for potential pathogens.
Most bone banks adhere to the guidelines of the American Association of Tissue Banks (AATB) with respect to procurement, processing, and sterilization of bone grafts. The AATB advocates excluding collection of bone under the following circumstances:
- Donors from high-risk groups, as determined by medical testing and behavioral risk assessments.
- Donors test positive for HIV antibody by ELISA.
- Autopsy of donor reveals occult disease.
- Donor bone tests positive for bacterial contamination.
- Donor and bone test positive for hepatitis B surface antigen (HbsAG) or hepatitis C virus (HCV).
- Donor tests positive for syphilis.
Using donor screening recommendations, it has been calculated that the chance of obtaining a bone graft from an HIV-infected donor (e.g., one who failed to be excluded by one of the exclusionary techniques) is one in 1.67 million 3. Furthermore, the probability that demineralized freeze-dried bone allograft might contain HIV has been calculated to be one in 2.8 billion 4. Therefore, the established exclusionary criteria combined with recommended processing procedures (harvesting in a sterile manner, repeated washings, immersion in ethanol, freezing in liquid nitrogen, freeze-drying, demineralization, and vacuum sealing) render demineralized freeze-dried bone allograft and freeze-dried bone allograft grafts safe for human implantation.
Who needs bone grafting?
Your surgeon may want to use bone grafting for a number of reasons. Possible reasons include situations where healing may be difficult due to the use of nicotine (which has been shown in medical studies to limit healing of the spine) or the presence of diseases such as diabetes or autoimmune deficiencies. Other possible reasons include a large amount of bone or disc material that is removed during surgery, or spinal procedures that span many levels of vertebrae.
One of the most common uses of bone grafts in spine surgery is during spinal fusion. The use of autogenous bone grafts for spinal fusion has been a standard in the spine community for many years and is considered to be the standard by which other bone grafts are measured. Spinal fusions are performed to relieve pain and provide stability to spines in people who have experienced a vertebral fracture or motion between the vertebrae that causes pain, have a spinal deformity, or who have some types of disc herniations. In certain types of spinal fusion, bone grafts or bone graft alternatives are used to replace the cushioning disc material that lies between the vertebrae. When the bone graft is placed between the vertebrae it creates a framework and support that eventually aids in joining the two bones together. Once the bone “bridge” between the vertebrae is in place, the spine is stabilized and movement and pressure on nerve roots is relieved, thus easing pain. Bone grafts can also be used in surgical procedures to stabilize the spine after a fracture or to correct deformity.
Bone graft material
Autograft
An autograft bone is bone taken from your own healthy bone. Autograft bone often comes from your ribs, hips or a leg. This type of bone graft is called an autograft. Autograft bone is one of the safest to use due to the low risk of disease transmission. Autograft bone also offers a better chance of acceptance and effectiveness in the transplant site, since it contains the greatest amount of your own bone growing cells and proteins. Autograft bone provides a strong framework for the new bone to grow into. The downsides of autograft bone are the facts that it adds another surgical site to an existing procedure (and therefore another location to feel postoperative pain and discomfort) and it can increase the cost of the surgical procedure.
The surgeon usually takes the bone from an area of the body where its removal will not cause a problem. In spine surgery, the bone is typically harvested from the iliac crest, which is the rim of the pelvic bone. The surgeon uses sharp tools to “scoop” the bone out of the iliac crest.
Taking bone from the iliac crest has been used with good results; however, there are some disadvantages:
- In some cases, it hurts to remove the bone; this pain may last for some time after the surgery.
- There is a limited amount of bone in the iliac crest, especially as we get older and our bone thins and weakens.
- The iliac crest may fracture or break.
- An infection or bleeding may occur.
Local Bone Autograft
In some spinal fusion procedures, parts of the spinal bones are removed to relieve pressure on the nerve roots. This removed bone is often saved during the surgery and used as bone graft. The bone is essentially recycled; it is moved from an area where it is no longer needed to the area that the surgeon wants to fuse.
One advantage to using local bone is that it is your own bone and, therefore, cannot be rejected. In addition, it requires no additional incision and does not cause pain in the hip. The disadvantage is that there is only a limited amount of bone that can be harvested from the small spinal bones.
Allograft Bone
Bone graft that comes from a donor is called allograft bone. Many surgeons use bone that is harvested from living donors or cadavers. This type of bone graft—an allograft bone—is typically acquired through a bone bank. Like other organs, bone can be donated upon death.
Allografts have been used for a long time in spinal fusion surgery. Unlike autografts taken from a patient, allografts do not form new bone. Instead, the allograft works as a bridge that allows the natural bone to grow through its surface. Over time, the natural bone replaces the donor bone. However, unlike autograft bone, allograft bone does not always have the same strength properties or the cells and proteins that can influence the growth of new bone.
The advantages of allograft bone are the elimination of the harvesting surgical site, the related postoperative pain and the added expense of a second operative procedure. Disadvantages of allograft bone are the slight chance of disease transmission and a lessened effectiveness since the bone growth cells and proteins are removed during the cleansing and disinfecting process.
Allograft bone comes in different shapes and sizes to fit into the area of the spine where it is needed. In most cases, allografts are good at getting bone to heal. Because the donor bone is dead and is only used as a scaffold for bone growth, the tissue is rarely rejected. The transmission of diseases from the donor bone is also very rare.
Allograft Stem Cells
Allograft stem cells can come from either a donor or from a cadaver. Stem cells are living cells that have the ability to form bone cells and aid in the fusion. They are unlikely to cause tissue rejection because they are basic cells that have not differentiated into specific cell types. Research using this type of bone graft is promising, but long-term studies comparing allograft stem cells to other graft types are not yet available.
Xenografts
Xenograft bone substitutes have their origin from a species other than human, such as bovine bone (or porcine bone) which can be freeze dried or demineralized and deproteinized 5. Bovine bone was first introduced by Maatz and Bauermeister in 1957 6. Xenografts are usually only distributed as a calcified matrix. Madrepore and or millepore type of corals are harvested and treated to become “coral derived granules” and other types of coralline xenografts 7. Coral based xenografts are mainly calcium carbonate (and an important proportion of fluorides, useful in the context of grafting to promote bone development) while natural human bone is made of hydroxyapatite along with calcium phosphate and carbonate. The coral material is thus either transformed industrially into hydroxyapatite through a hydrothermal process, yielding to a non-resorbable xenograft, or simply the process is omitted and the coralline material remains in its calcium carbonate state for better resorption of the graft by the natural bone. The coral xenograft is then saturated with growth enhancing gels and solutions 8. In January 2010 Italian scientists announced a breakthrough in the use of wood as a bone substitute, though this technique is not expected to be used for humans until at the earliest 2015. Various species of wood are pyrolized in an inert atmosphere, the carbonaceous residue is saturated with calcium salts and finally reheated to obtain a highly porous crystallized material of much higher porosity than trabecular titanium or porous hard ceramic bone-substitutes; the inventors claim the wood based material will permit better penetration during bone growth and more flexion than metal or hard ceramic grafts 9. Xenografts have given good results in dentistry, but scarce validation in orthopaedics.
Clinically available coral-based products are Interpore and Pro-osteon (Interpore International, Inc., Irvine, CA) as well as bovine derived products such as Bio-Oss (Geistlich Biomaterials, Geistlich, Switzerland), Osteograf-N (CeraMed Co., Denver, CO), and Endobon (Merck Co., Darmstadt, Germany). Doubts were argumented regard “zoonose” diseases transmitted from animals to humans, like BSE (Bovine spongiform Encephalopathy) or PERV (Porcine Endogenous Retroviruses) 10.
Results are contradictory with some authors reporting favourable data, but in the clinical practice xenografts are scarcely used 11.
Moreover poor results in hip surgery, with 25 % of pseudo infections complications, were recently reported 12.
The advantages are the easy availability, the osteoconductivity, the good mechanical properties and low costs.
Bone Graft Substitutes
Since both allograft and autograft have drawbacks, scientists have long searched for materials that could be used in place of the transplanted bone. Although most of the substitutes available possess some of the positive properties of autograft, none yet have all the benefi ts of one’s own bone. Investigators hope one day to use these substitutes to simultaneously be able to stimulate bone healing and provide a strong and biologically compatible framework for the new bone to grow into.
Some of these bone graft alternatives include:
Demineralized Bone Matrix
Demineralized Bone Matrix (DBM) — is a product of processed allograft bone that is processed in such a way that the calcium in the bone is removed. The benefit of this process is that it makes the proteins trapped in the calcium available to stimulate bone healing. Demineralized Bone Matrix contains collagen, proteins and growth factors that are extracted from the allograft bone. Demineralized Bone Matrix is available in the form of a powder, crushed granules, putty, chips or as a gel that can be injected through a syringe. Demineralized Bone Matrix is extensively processed and therefore has little risk for disease transmission; however, because of the form it takes it does not provide strength to the surgical site. Demineralized Bone Matrixes are usually combined with other types of grafts.
Ceramics
Synthetic bone grafts are made from calcium materials (a mix of hydroxyapatite [HA] and Tricalcium Phosphate [TCP]) and are often called “ceramics.” Ceramics are also used as a substitute for bone grafts. Hydroxyapatite [Ca10(PO4)6(OH)2] (HA) is the cristalline form of tricalcium phosaphate (TCP) and is the primary mineral component of teeth and bone. For the past 30 years, it has been popular in orthopaedic, craniofacial and orthognathic surgery, filling bony defects and smoothing contour irregularities. HA (hydroxyapatite) is a relatively inert substance that is retained “in vivo” for prolonged periods of time, whereas the more porous TCP (tricalcium phosphate) typically undergoes biodegradation within 6 weeks of its introduction into the area of bone formation. Hydroxyapatite [HA] achieve very high mechanical strenght, while TCP (tricalcium phosphate) has poor mechanical qualities. Generally the base is a biphasic calcium phosphate, which combine 40–60 % TCP (tricalcium phosphate) with 60–40 % hydroxyapatite [HA], that may yield a more physiological balance between mechanical support and bone resorption 13.
Ceramics are similar in shape and consistency to autograft bone. Ceramics have the advantage of being made without cadaver bone and are available in large amounts. Ceramics offer no possibility for disease transmission, although they may be associated with inflammation in some patients. They are available in many forms such as blocks, granules, and injectable kits and mesh. Although ceramics may provide a framework for bone growth, they contain none of the natural proteins that influence bone growth.
Coral
Bone implants made from coral have shown to be useful in the treatment of bone defects due to trauma, tumors and cysts. Coral is also used for spinal surgery as either a graft additive, or extender, or as an implant to provide a framework for bone to grow into. The use of these substitutes, under certain conditions, has had promising outcomes.
Graft Composites
A newer area of bone graft substitutes, graft composites use combinations of other bone grafting materials and/or bone growth factors to gain the benefits of a variety of substances. Among the combinations in use are a collagen/ceramic composite, which closely reproduces the composition of natural bone; Demineralized Bone Matrix (DBM) combined with bone marrow cells, which aid in the growth of new bone; and a collagen/ceramic/autograft composite.
Bone Morphogenetic Proteins
Bone morphogenetic proteins (BMPs) are produced in your body and regulate bone formation and healing. Scientists have discovered that these bone morphogenetic proteins (BMPs) can speed up healing as well as limit the negative reaction some people have to donor bone and the nonbone substitutes. Scientists have also discovered how to extract these substances from human or cow bones and even produce them in the laboratory.
Bone Morphogenic Protein has very powerful bone-forming capabilities so care must be taken with its use. While some studies of bone morphogenic protein in the spine show that too much bone may be formed or that bone morphogenic protein may increase the patient’s risk for cancer, other studies have shown that the use of bone morphogenic protein is very safe. Your surgeon will talk with you about the risks and benefits of using this powerful protein in your case.
Bone Marrow Aspirate
Bone marrow is located in the centers of long bones, such as the bones in your arms, thighs, and legs. The marrow is the area of the bone that contains stem cells. Stem cells are special cells in your body that can turn into other types of cells. They have some ability to make new bone.
Stem cells can be aspirated or drawn out of the bone marrow with a needle, much like the way blood is drawn from your arm for tests. The cells are then combined with another type of graft—such as local bone autograft, allograft, or synthetic bone—to speed the healing process.
Currently, research is underway to find new ways of harvesting stem cells.
Which type of bone graft is right for me?
The determination if bone graft is to be used, and the type to be used, is best made by your surgeon. Different surgical situations may call for different types of bone grafting and unique bone graft materials. If your surgeon says that he or she would like to use a bone graft on your spine, you should discuss this decision with him or her to determine which bone graft material they feel is best suited for your situation. As with any medical procedure, you should ask your surgeon about any questions or concerns you may have and make sure all of your questions are answered.
Dental bone graft
Uses for bone grafts in dentistry. Although they are often used, bone grafts are not always necessary for dental implants. Your oral surgeon will probably talk with you about getting a bone graft if they see that your jawbone is too thin, soft, or insufficient to be a foundation for the implant. If there’s not enough bone, the implant could fail over time.
Bone grafts are used in dentistry to accomplish the following treatment goals:
- Saving Teeth — When severe periodontal disease causes bone loss, teeth can become loose and at risk of being lost. In order to save them, the bone around them can be regenerated through grafting; this increases bone support and helps keep them in place.
- Tooth Extractions — These days, it is very common to deposit bone grafting material into a tooth socket after a tooth has been removed. That way, should you want to replace your tooth with a dental implant later on, that option will be available.
- Dental Implants — In this optimal tooth-replacement system, a small titanium post embedded in the jawbone is attached to a highly realistic dental crown, permanently replacing the missing tooth. Implants require good bone volume and density to achieve their excellent functionality and high success rates. If you have already experienced bone loss, a graft can help regenerate enough bone to place the implant successfully.
Dental implant surgery is a procedure that replaces tooth roots with metal, screw-like posts and replaces damaged or missing teeth with artificial teeth that look and function much like real ones. Dental bone grafts are often need because you have lost bone in the area where a dental implant is optimum for the success of your procedure. When teeth are lost due to disease or trauma, the surrounding bone will start to “resorb” without any root to stimulate the bone. This bone loss can make restoring missing teeth with dental implants or implant supported dentures very difficult or not possible. So dental bone graft will help maximize the outcome of your procedure. Bone grafting can be a complex procedure. For vast cases, large changes must be made to the size and shape of the area for the dental implant. Surgery must be undertaken before the implant can be placed. However, sometimes there is enough bone present to put the implant, but not quite enough to cover up the sides of the implant.
Bone graft for dental implant
If your jawbone isn’t thick enough or is too soft, you may need bone grafting before you can have dental implant surgery. That’s because the powerful chewing action of your mouth exerts great pressure on your bone, and if it can’t support the implant, the surgery likely would fail. A bone graft can create a more solid base for the implant.
Types of dental implant bone grafts
The main five places a dental surgeon can get a bone graft are:
- From your body
- From another person
- From an animal
- Synthetic materials
- From a cadaver
Where the dentist decides to get the bone graft depends on what they believe will work best for each patient. There are also different sizes of bone grafts, depending on what’s needed.
In bone grafting, a piece of bone is removed from another part of your jaw or your body — your hip, for example — and transplanted to your jawbone. Another option is to use artificial bone (bone commercially available) to place in these areas. It may take several months for the transplanted bone to grow enough new bone to support a dental implant.
In some cases, you may need only minor bone grafting, which can be done at the same time as the implant surgery. The condition of your jawbone determines how you proceed. The implant survival rate in anterior iliac bone autografts has been reported to be 92.4% after a follow-up period of 5 years 14.
There are four main types of bone grafts: little, medium, big, and sinus lift. Which one you get depends on how much bone is needed for a procedure.
Little Bone Graft
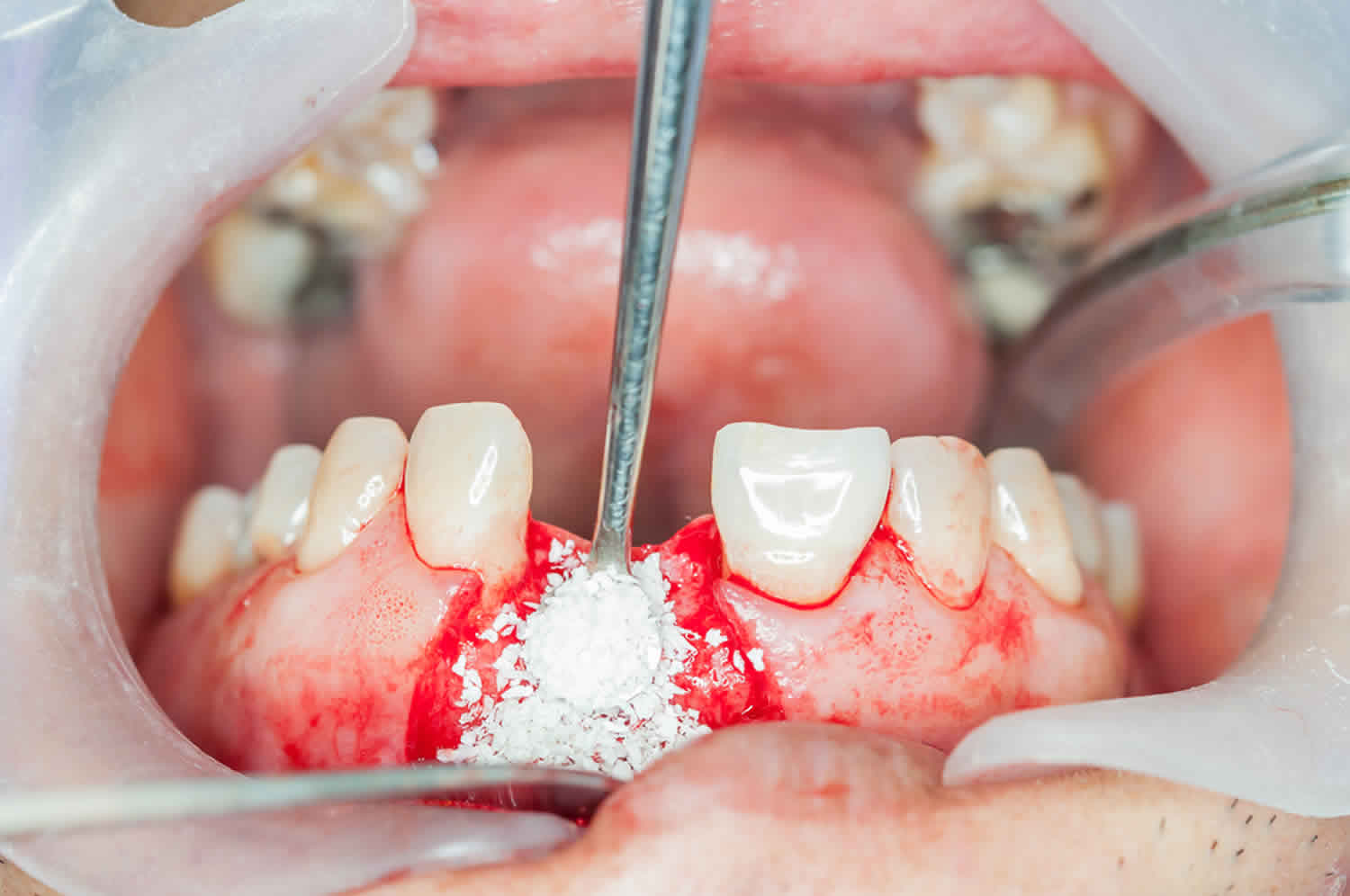
After your dentist has removed any damaged teeth, they may use a little bone graft. This is when human bone in the form of granules (looks like coarse sand) are placed into the empty tooth socket. Then they cover the socket with a protective collagen membrane using stitches. And fortunately, it doesn’t usually had to your recovery time.
In the following weeks, the natural bone will form inside the socket, keeping the original bone height.
Medium Bone Graft
A medium bone graft is when more significant bone loss has happened and both height and width need to be restored. The dentist will make a small incision in the affected bone then insert bone granules. This will help build up the area, giving it width and height as it heals.
Big Bone Graft
When a person has been missing multiple teeth for a long time, often they will have experienced severe bone loss. This is when their dentist may suggest a big bone graft.
Rather than using mainly bone granules, the dentist will opt to use a block of bone from elsewhere in your body. They will attach the bone using screws and plates, providing a foundation for a dental implant. Then they use some bone granules to fill in the spots they need to. Then they put a membrane over the whole area using stitches — this takes months to heal and before the dentist can place any implant.
Sinus Lift
Another type of bone graft is called a sinus lift. If a person has extreme bone loss on their upper jaw, they may need a bone graft as the sinuses are right above the upper teeth and the dentist can’t drill into the sinuses. Obviously, dental implants can’t be secured using a hollow cavity (sinus), so the dentist will need to raise the floor of the sinus to give the area more bone surface to use.
They will make an incision in the gum tissue, creating a little window into the sinus cavity. The dentist will then carefully detach and lift the sinus membrane from the sinus walls. Then bone is placed between the membrane and the sinus floor. Once there’s enough bone in the area, the dentist will place a collagen membrane to protect the inserted bone. Then the little window is closed up with stitches.
A sinus lift requires at least four months to full heal before a dental implant can be placed.
What is the cost a dental bone grafting?
The cost of a dental bone graft can vary depending on the source and the amount of bone needed. Typically, for a bone graft from another human, a cadaver, a cow, or from synthetic bone, the total cost ends up being between $200 and $1,200.
As for getting the bone graft from the patient’s own body, the total cost may be between $2,000 and $3,000. Keep in mind this involves two surgery sites, hospitalization, and an anesthesiologist.
In both cases, when the price of a bone graft is quoted to you, it includes the dental implant. Dental insurance usually won’t cover these costs as many insurance companies view dental implants as optional procedures mainly for cosmetic reasons. However, if the dental implant is necessary after an injury or damage to a tooth, your general health insurance may cover the costs.
Bone graft in mouth healing stages
If you do end up getting a bone graft, it will affect the length of the overall treatment. Because getting a dental implant requires multiple steps and procedures, adding an additional surgery will make the whole process take longer. But for some people, a bone graft is a needed step.
Bone regeneration can be accomplished through three different mechanisms: osteogenesis, osteoinduction, and osteoconduction 15. Osteogenesis is the formation and development of bone, even in the absence of local undifferentiated mesenchymal stem cells. Osteoinduction is the transformation of undifferentiated mesenchymal stem cells into osteoblasts or chondroblasts through growth factors that exist only in living bone. Osteoconduction is the process that provides a bio-inert scaffold, or physical matrix, suitable for the deposition of new bone from the surrounding bone or encourage differentiated mesenchymal cells to grow along the graft surface 16.
All grafting materials have one or more of these three mechanisms of action. The mechanisms by which the grafts act are normally determined by their origin and composition. Autograft bone harvested from the patient forms new bone by osteogenesis, osteoinduction, and osteoconduction 15. Allografts harvested from cadavers have osteoconductive and possibly osteoinductive properties, but they are not osteogenic 15. Xenografts/alloplasts are typically only osteoconductive 15. Xenografts are tissue grafts obtained from animal bones 15. The representative xenograft materials are natural hydroxyapatite (HA) and deorganified bovine bone (anorganic bone matrix). These graft materials are inert osteoconductive filler material, which serves as a scaffold for new bone formation. Natural hydroxyapatite extracted from animal bones has the three-dimensional microstructure of natural bone and is highly biocompatible to adjacent hard and soft tissues. Anorganic bone matrix is an inorganic bone of bovine origin. It is a carbonate containing apatite with crystalline architecture and a calcium/phosphate ratio similar to that of natural bone mineral in humans. With time, anorganic bone matrix graft material becomes integrated into the human bone and is slowly replaced by newly formed bone. However, the remodeling process takes a long time and reports have shown the bovine graft present even after 18 months 17. Human biopsies after sinus augmentation confirm that particles of bovine-derived bone substitutes can still be found up to 10 years postoperatively 18. Disadvantages of xenografts are the increased risk of a host-immune response, brittleness and easy migration 19. Xenografts appear to incorporate into natural bone, but their low resorption rate may negatively impact the healing of the grafted site and compromise the mechanical and biological properties of the regenerated bone 15.
Alloplasts are an inert synthetic graft material 15. The most commonly used alloplast materials are calcium carbonate, calcium sulfate, bioactive glass polymers and ceramic materials, including synthetic hydroxyapatite and tricalcium phosphate 15. The mechanism of action of these materials is strictly osteoconduction. They provide a scaffold for enhanced bone tissue repair and growth.
The use of autografts, allografts, xenografts, or alloplasts, alone or in combination, should be based on the individual’s systemic healing capacity, the osteogenic potential of the recipient site, and the time available for graft maturation. Due to the absence of definitive conclusions as to the relative efficacy of xenografts and alloplasts in the management of periodontal defects, they are recommended to be combined with allografts for small defects in healthy patients. Autograft bone should be added for progressively larger defects, especially for defects and/or patients with lower osteogenic potential. Additionally a barrier membrane should be utilized for better results 20.
Angiogenesis and ample blood supply are mandatory for bone development and maintenance. Formation of new blood vessels usually proceeds from existing blood vessels. For an intact dentate alveolar ridge, blood supply includes the complex of supraperiosteal arterioles, the subepithelial capillary network of the gingiva and the periodontal ligament, and the arterioles penetrating the interdental alveolar bone. However, when a tooth is lost, the blood supply from the periodontal ligament disappears, and the blood supply is only from the soft tissue and the supraperosteal blood vessels of the bone.
The cortical bone surface is usually perforated with a small round bur prior to placing a bone graft to open the marrow cavity and to stimulate bleeding into the defect area. This is called decortication or bone marrow penetration [28]. The rationale may include: (1) to enhance the healing process by promoting bleeding and blood clot formation; (2) to allow progenitor cells and blood vessels to reach the bone graft site 21 which facilitate angiogenesis; and (3) to improve the physical interlocking of grafted bone and a recipient site 22. However, bone marrow penetration may also have some negative effects; additional blood loss, potentially greater postoperative pain, increased bone loss, and increased operative time 23.
Conflicting information has been reported with regard to the ability of bone marrow penetration to accelerate or increase bone regeneration in the experimental animal studies 24. Delloye et al. 25 found that perforating a cortical bone graft substantially improved the amount of new bone formation by the host compared to using a non-perforated cortical bone graft. In a controlled clinical trial using a rat model, tibial or femoral grafts were placed on tibial bones with or without cortical perforation. It was noted that after 20 weeks of healing, there was migration of marrow components through the perforated area with an increased level of lamellar bone apposition compared to the non-decorticated grafts 26. Decortications were also studied in a rat/rabbit spinal fusion model and found that decortications of vertebrae bone resulted in a statistically significant larger percentage of bone formation during spinal fusion and better graft integration after 9-10 weeks compared to sites that were not decorticated 27. Similar results were reported using a dog spinal fusion model for the first three months. However, no such benefits from decortications were identified at 6 months. Mixed results also exist with animal mandibular onlay bone grafting model. de Carvalho et al. 28 studied the healing of autogenous monocortical bone grafts placed on the mandible in six dogs and demonstrated a better healing with integrated bone at cortical perforation sites as opposed to non-perforated sites after 90 days. In contrast, other studies found decortication did not enhance the incorporation of onlay mandibular bone grafts 24. There was no appreciable histological difference in healing with or without prior bone marrow penetration 29.
Similarly, conflicting results have also been reported about skeletal or extra-skeletal guided bone regeneration (using barrier membranes) with or without decortications. Guided bone regeneration is a surgical procedure that uses barrier membranes with or without particulate bone grafts or/and bone substitutes. There are two approaches of guided bone regeneration in implant therapy: guided bone regeneration at implant placement (simultaneous approach) and guided bone regeneration before implant placement to increase the alveolar ridge or improve ridge morphology (staged approach). Angiogenesis and ample blood supply play a critical role in promoting bone regeneration.
Using a rabbit calvaria titanium dome model, there were more osteoblast-like cells at sites under the titanium dome that underwent decortications compared to controls after 2-3 months and the percentage of bone regeneration was significantly higher 30. A similar result was found using a calvaria rat model after 4 months 31. However, several animal studies with negative results were also reported and claimed that cortical perforation did not enhance the amount of bone augmentation in rabbits 32. Other studies showed guided bone regeneration procedures could be performed successfully to different degrees without decortications 33.
Regarding the effect of different sizes of cortical perforation, the data available is minimal. Nishimura et al. 34 found that initially (week 2-6), the larger cortical openings (3 x 15 mm) were associated with faster and more new bone formation compared to smaller perforation (1 x 15 mm). However, no significant difference was found regarding to the amount of bone regeneration after 12 weeks.
Dental bone graft complications
After hearing about the details of the different types of bone grafts, you may be wondering: is bone grafting for dental implants safe?
Simply put, yes, they are safe when performed by a licensed professional. Remember, almost every dental implant procedure is successful, and many of them involve bone grafts.
However, as with any surgery, bone grafts are not without their possible complications and risks. Even though they’re rare, complications can still occur. Potential problems after bone grafting include wound infection, fracture of the jaw bone, swelling, bleeding, pain, loss of sensation, sinusitis, headaches and difficulty eating or speaking related to the procedure.
Here are some potential risks you should be aware of:
- Infection
- Damage to natural teeth, blood vessels, or nerves
- Numbness, tingling in gums, lips, and cheek
- Sinus problems (with upper jaw implants)
- Swelling, pain, bruising.
Dental bone graft recovery
Many factors come into play here. For instance, is it in the upper or lower jaw? What bone material or membrane is going to be used? How much height or width of the bone graft is trying to be achieved? Healing time following bone grafting depends on the amount of bone loss and the location of the graft area. The success of bone grafting is also highly dependant on good oral hygiene. With that said, most bone grafts heal in around 4 months, but some take as long as 9 months or over a year if you need an additional procedure. Your dentist should be able to give you a precise timeline based on your care.
Most dental implant procedures are successful — about 95% of them. But it’s still important to do everything you can to help the process at each stage.
For example, your doctor may ask you to eat only soft foods so as to not put pressure on the surgical sites. Plus, you can keep up top-notch oral hygiene to help the healing process.
Here are some things you can do to take care of your dental implant as well as your natural teeth:
- Brush, floss, and rinse with non-alcoholic mouthwash 2-3 times a day. You can also get special dental implant tools that can reach where a toothbrush or floss cannot.
- See your dentist regularly, preferably every six months. This will help ensure your implant is healing properly and will continue to perform well.
- Avoid damaging the implant by not eating hard foods, like ice or hard candy. These types of things can break the crown. Also avoid tobacco and excessive caffeine as these can stain your natural and artificial teeth.
Signs of dental bone graft failure
Current 20-year follow-up studies suggest that 99% of implants remain osseointegrated after 1 year, but this drops to 93% after 5 years 35. Implant failure can be arbitrarily classified into early (failure to achieve osseointegration) and late (failure to maintain established osseointegration during functional loading) 36. The cause of early failure is assumed to include excessive surgical trauma, impaired healing, bacterial contamination and premature overloading, and that for late failure to include excessive occlusal stress and bacterial-induced peri-implant bone loss 37. The current series of failed dental implants, irrespective of their occurrence at an early or late stage, shared a clinical picture that pointed to the manifestation of an infectious/inflammatory process, characterized by signs of swelling, pain, fistula and purulent exudate. The radiological features were essentially similar, and included radiolucent or mixed radiolucent–radiopaque lesions with poorly defined borders. Some of them were limited to the area of the failed implant, while others were extensive and involved a large part of the mandible. The histopathological findings ranged from acute osteomyelitis and chronic osteomyelitis with features of a fibro-osseous-like lesion and occasional rimming of atypical osteoblasts, to osteogenic sarcoma that was admixed with a component of osteomyelitis 38.
Clinical manifestation of early and late implant failure can be either symptomatic or asymptomatic (i.e., only affecting mobility) with or without radiological changes. The radiological change that is usually seen surrounding an implant failure is that of a peri-implant radiolucency, frequently with irregular contours 37. Untreated bacteria-induced peri-implantitis may eventually lead to deep bone invasion of the bacteria and cause true osteomyelitis, which is a rare complication relative to the large number of dental implants that are placed nowadays 39. Acute and subsequent secondary chronic osteomyelitis in the context of implant failures has been reported in a small number of patients 40. Radiological features that are mainly associated with the chronic form of osteomyelitis consist of poorly-defined, moth-eaten, radiolucent bone loss admixed with radio-opaque areas, representing the formation of a bony sequestrum 39.
- Autograft versus BMPs for the treatment of non-unions: what is the evidence? Blokhuis TJ, Calori GM, Schmidmaier G. Injury. 2013 Jan; 44 Suppl 1():S40-2.[↩]
- Frequently Asked Questions – Bone Allografts. https://www.cdc.gov/oralhealth/infectioncontrol/faq/allografts.htm[↩]
- Buck BE, Malinin T, Brown MD. Bone transplantation and human immunodeficiency virus. Clin Orthop 1994;303:8–17.[↩]
- Russo R, Scarborough N. Inactivation of viruses in demineralized bone matrix. FDA workshop on tissue transplantation and reproductive tissue, June 20–21, 1995, Bethesda, MD. Simmonds RJ, Holmberg SD, Hurwitz RL, et al. Transmission of human immunodeficiency virus type 1 from a seronegative organ and tissue donor. N Engl J Med 1992;326:726–732.[↩]
- Campana V, Milano G, Pagano E, et al. Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med. 2014;25(10):2445-61. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4169585[↩]
- Bauermeister A, Maatz R. A method of bone maceration results of animal experiments. J Bone Joint Surg Am. 1957;39(1):153–166.[↩]
- Jensen SS, Terheyden H. Bone augmentation procedures in localized defects in the alveolar ridge: clinical results with different bone grafts and bone-substitute materials. Int J Oral Maxillofac Implants. 2009;24(Suppl):218–236.[↩]
- Dumitrescu AL. Bone grafts and bone graft substitutes in periodontal therapy. In: Chemicals in surgical periodontal therapy. Berlin: Springer; 2011. p. 92.[↩]
- Tampieri A, Sprio S, Ruffini A, Celotti G, Lesci IG, Roveri N. From wood to bone: multi-step process to convert wood hierarchical structures into biomimetic hydroxyapatite scaffolds for bone tissue engineering. J Mater Chem. 2009;19:4973–4980.[↩]
- Laurencin CT, El-Amin SF. Xenotransplantation in orthopaedic surgery. J Am Acad Orthop Surg. 2008;16(1):4–8.[↩]
- Lofgren H, Johannsson V, Olsson T, Ryd L, Levander B. Rigid fusion after cloward operation for cervical disc disease using autograft, allograft, or xenograft: a randomized study with radiostereometric and clinical follow-up assessment. Spine. 2000;25(15):1908–1916.[↩]
- Charalambides C, Beer M, Cobb AG. Poor results after augmenting autograft with xenograft (Surgibone) in hip revision surgery: a report of 27 cases. Acta Orthop. 2005;76(4):544–549.[↩]
- Boyan BD, McMillan J, Lohmann CH, Ranly DM, Schwartz Z. Basic information for successful clinical use with special focus on synthetic graft substitutes. In: Laurencin CT, editor. Bone graft substitutes. Philadelphia: ASTM Int; 2002. pp. 231–259.[↩]
- Onlay augmentation versus sinuslift procedure in the treatment of the severely resorbed maxilla: a 5-year comparative longitudinal study. Wiltfang J, Schultze-Mosgau S, Nkenke E, Thorwarth M, Neukam FW, Schlegel KA. Int J Oral Maxillofac Surg. 2005 Dec; 34(8):885-9.[↩]
- Liu J, Kerns DG. Mechanisms of guided bone regeneration: a review. Open Dent J. 2014;8:56-65. Published 2014 May 16. doi:10.2174/1874210601408010056 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4040931/[↩][↩][↩][↩][↩][↩][↩][↩]
- Misch CE, Dietsh F. Bone-grafting materials in implant dentistry. Implant Dent. 1993;2:158–67.[↩]
- van Steenberghe D, Callens A, Geers L , et al. The clinical use of deproteinized bovine bone mineral on bone regeneration in conjunction with immediate implant installation. Clin Oral Implants Res. 2000;11:210–6.[↩]
- Piattelli M, Favero GA, Scarano A , et al. Bone reactions to anorganic bovine bone (Bio-Oss) used in sinus augmentation procedures: a histologic long-term report of 20 cases in humans. Int J Oral Maxillofac Implants. 1999;14:835–40.[↩]
- Lane JM. Bone graft substitutes. West J Med. 1995;163:565–6.[↩]
- Isaksson S, Alberius P, Klinge B. Influence of three alloplastic materials on calvarial bone healing: an experimental evaluation of HTR-polymer. lactomer bads.and a carrier gel. Int J Oral Maxillofac Surg. 1993; 22:375–81.[↩]
- Buser D, Bragger U, Lang NP , et al. Regeneration and enlargement of jaw bone using guided tissue regeneration. Clinl Oral Implants Res. 1990;1:22–32.[↩]
- Alberius P, Gordh M, Lindberg L , et al. Onlay bone graft behaviour after marrow exposure of the recipient rat skull bone. Scand J Plast Reconstr Surg Hand Surg. 1996;30:257–66.[↩]
- Greenstein G, Greenstein B, Cavallaro J , et al. The role of bone decortication in enhancing the results of guided bone regeneration: a literature review. J Periodontol. 2009;80:175–89.[↩]
- Adeyemo WL, Reuther T, Bloch W , et al. Influence of host periosteum and recipient bed perforation on the healing of onlay mandibular bone graft: an experimental pilot study in the sheep. Oral Maxillofac Surg. 2008;12:19–28.[↩][↩]
- Delloye C, Simon P, Nyssen-Behets C , et al. Perforations of cortical bone allografts improve their incorporation. Clin Orthop Relat Res. 2002;396:240–7.[↩]
- Gordh M, Alberius P, Lindberg L , et al. Bone graft incorporation after cortical perforations of the host bed. Otolaryngol Head Neck Surg. 1997;117:664–70.[↩]
- Canto FR, Garcia SB, Issa JP , et al. Influence of decortication of the recipient graft bed on graft integration and tissue neoformation in the graft-recipient bed interface. Eur Spine J. 2008;17:706–14.[↩]
- de Carvalho PS, Vasconcellos LW, Pi J. Influence of bed preparation on the incorporation of autogenous bone grafts: a study in dogs. Int J Oral Maxillofac Implants. 2000;15:565–70.[↩]
- Huebsch RF, Wellington JS. Osseous healing in dog mandibles with and without decortication. Oral Surg Oral Med Oral Pathol. 1967;23:236–40.[↩]
- Min S, Sato S, Murai M , et al. Effects of marrow penetration on bone augmentation within a titanium cap in rabbit calvarium. J Periodontol. 2007;78:1978–84.[↩]
- Rompen EH, Biewer R, Vanheusden A , et al. The influence of cortical perforations and of space filling with peripheral blood on the kinetics of guided bone generation: a comparative histometric study in the rat. Clin Oral Implants Res. 1999;10:85–94.[↩]
- Barbosa DZ, de Assis WF, Shirato FB , et al. Autogenous bone graft with or without perforation of the receptor bed: histologic study in rabbit calvaria. Int J Oral Maxillofac Implants. 2009;24:463–8.[↩]
- Dongieux JW, Block MS, Morris G , et al. The effect of different membranes on onlay bone graft success in the dog mandible. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;86:145–51.[↩]
- Nishimura I, Shimizu Y, Ooya K. Effects of cortical bone perforation on experimental guided bone regeneration. Clin Oral Implants Res. 2004;15:293–300.[↩]
- Implant treatment in the edentulous mandible: a prospective study on Brånemark system implants over more than 20 years. Ekelund JA, Lindquist LW, Carlsson GE, Jemt T. Int J Prosthodont. 2003 Nov-Dec; 16(6):602-8.[↩]
- Wiskott HW, Dubrez B, Scherrer SS, Belser UC. Reversible and irreversible peri-implant lesions: report and etiopathogenetic analysis of 7 cases. J Oral Implantol. 2004;30:255–266. doi: 10.1563/1548-1336(2004)30<255:RAIPLR>2.0.CO;2[↩]
- Esposito M, Thompsen P, Ericson LE, Sennerby L, Lekholm U. Histopathologic observations on late implant failures. Clin Implant Dent Relat Res. 2000;2:18–32. doi: 10.1111/j.1708-8208.2000.tb00103.x[↩][↩]
- Shnaiderman-Shapiro A, Dayan D, Buchner A, Schwartz I, Yahalom R, Vered M. Histopathological spectrum of bone lesions associated with dental implant failure: osteomyelitis and beyond. Head Neck Pathol. 2014;9(1):140-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4382485/[↩]
- Zimmerli W. Osteomyelitis therapy–antibiotic therapy. In: Baltensperger MM, Eyrich GK, editors. Osteomyelitis of the jaws. Berlin: Springer; 2009. pp. 179–190.[↩][↩]
- Rokadiya S, Malden NJ. An implant periapical lesion leading to acute osteomyelitis with isolation of Staphylococcus aureus. Br Dent J. 2008;205:489–491. doi: 10.1038/sj.bdj.2008.935.[↩]