Contents
Chronic abdominal wall pain
Chronic abdominal wall pain also called chronic abdominal wall pain syndrome or chronic non-visceral abdominal pain, is a discomfort occurring between the diaphragm and the upper plane of the pelvic cavity that originates from the abdominal wall itself (known as somatic pain) rather than from a viscera underneath the wall such as internal organs of the body (known as visceral pain) 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15. Chronic abdominal wall pain is frequently misinterpreted as visceral or functional abdominal pain 5. The most common cause of chronic abdominal wall pain is anterior cutaneous nerve entrapment syndrome (ACNES). The cutaneous branches of sensory nerves (T7 to T12) go through the posterior rectus sheath, passing through a fibrous ring (see Figure 1). Normally, fat in the neurovascular bundle permits the nerve to slide unimpeded within the fibrous ring. Entrapment of the cutaneous branches of sensory nerve can be caused by intra or extra abdominal pressure or localized scarring. Patients usually localize the pain near the lateral border of the rectus abdominis muscle. The pain is localized to the right upper quadrant in 40% of cases, while pain superior to the umbilicus represents the second most common location 16. The pain is increased by contraction of the abdominal wall muscles as in a “sit-up,” and the site is very tender.
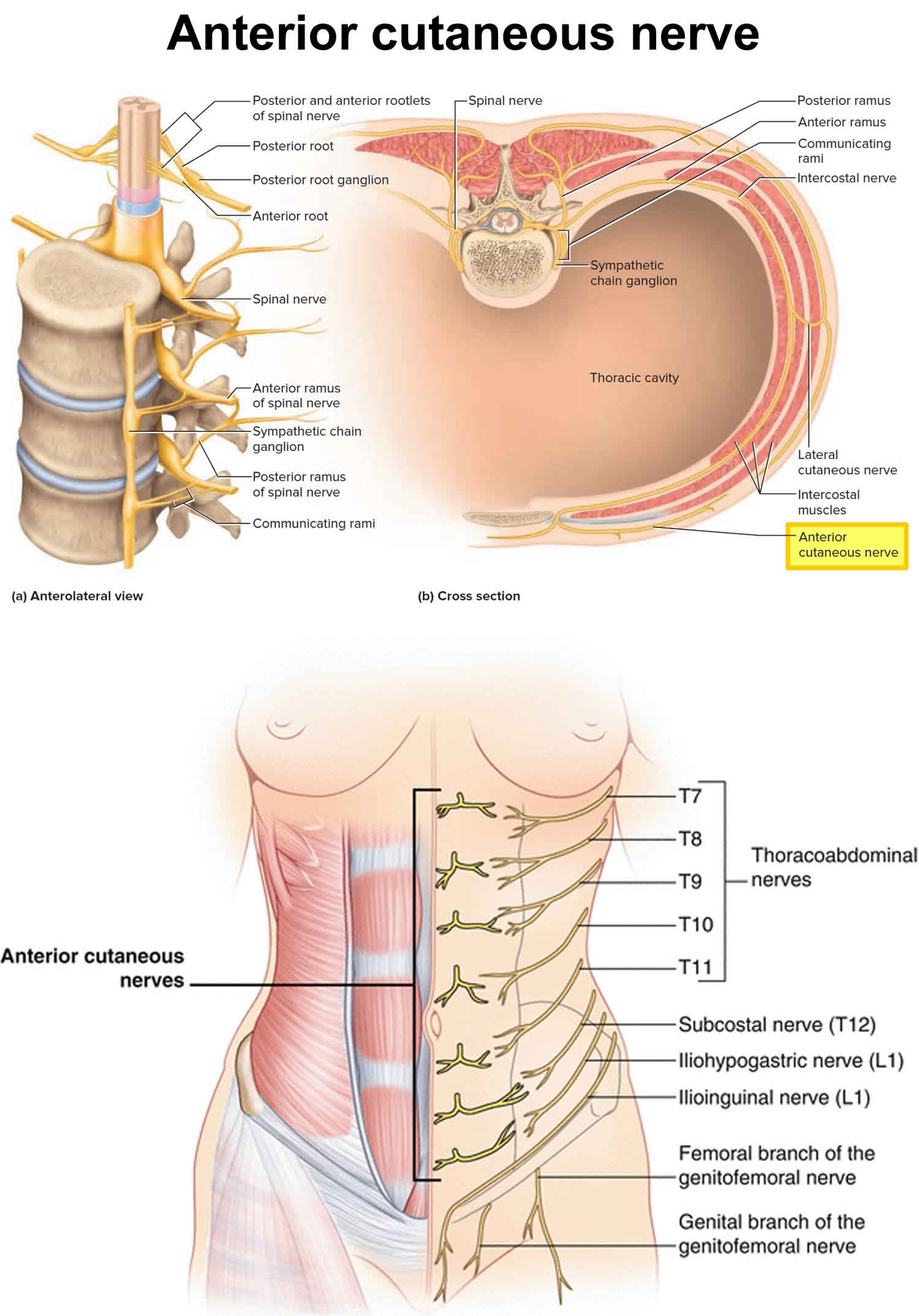
Chronic abdominal wall pain is caused by the entrapment of the anterior cutaneous branches of sensory nerves that supply the abdominal wall 17, 18, 19. The abdominal wall is supplied by nerve roots T7 to T12 (Figure 1). The intercostal nerves (T7 to T12) first run along the ribs, then pass onward to innervate the abdominal wall 20. The critical site for pain is located at the lateral edge of the rectus abdominis muscle. Anatomically, the lower intercostal nerves (T8 to T12) run between the internal oblique and transversus abdominis muscles, up to the point where they reach the rectus abdominis; the cutaneous nerves make a right angle at this point in order to travel from the inner to the outer part of the abdominal wall; they then make another right angle to continue along the abdominal wall (Figure 1) 21, 22. Some authors have suggested that the entrapment is usually caused by this sudden turn in their course into the rectus abdominis channel, leading to anterior cutaneous nerve entrapment syndrome (ACNES) 23, 24, 2, 25, 20. The muscle contraction at this location may cause additional nerve compression, and therefore mechanical or ischemic irritation, which results in severe pain 23, 3, 26. Although the precise mode of damage is not known, postulated mechanisms include intra-abdominal or extra-abdominal pressure, ischemia, and compression of the nerve by herniation of the fat pad that normally protects it into the fibrous canal surrounding the nerve. Applegate 25 coined the term “abdominal cutaneous nerve syndrome” (ACNES), which is widely used in English-speaking countries.
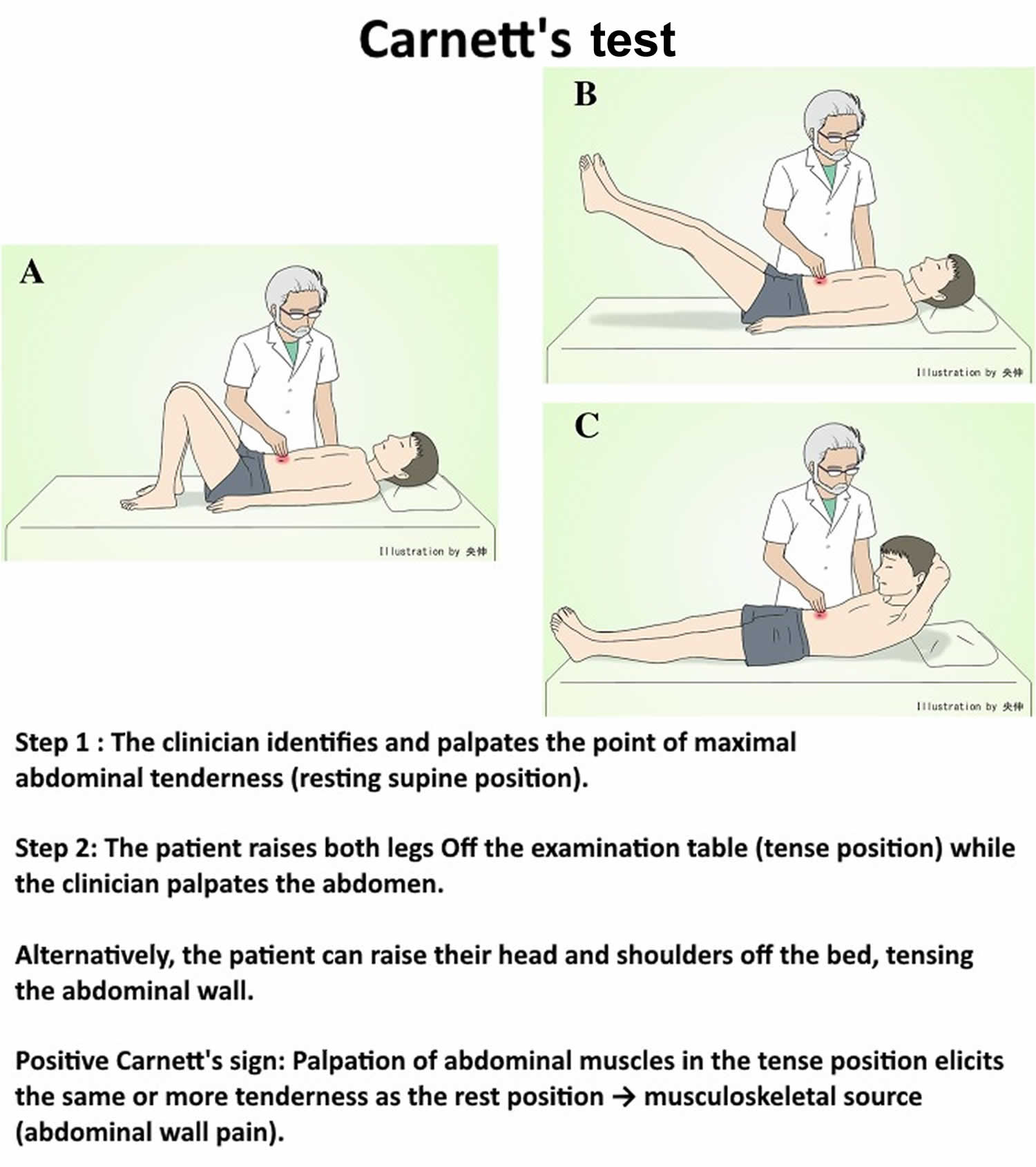
Chronic abdominal wall pain is frequently misdiagnosed as arising from a visceral (internal organs of the body) source, often resulting in inappropriate diagnostic testing and even abdominal operations with negative results, unsatisfactory treatment, and considerable cost. However, with a simple clinical test called Carnett’s test (see Figure 2), over 90% of patients with chronic abdominal wall pain can be recognized, without risk for missing intra-abdominal pathology 27, 4. Carnett’s test is performed by palpating a limited area of tenderness in a supine, relaxed patient (see Figure 2 Carnett’s test A), which subsequently confirms continued tenderness as the patient tenses the abdominal wall while the head and shoulders are elevated or while raising both legs off the table (see Figure 2 Carnett’s test B). Carnett 27 and Greenbaum et al. 7 defined the positive sign as follows: If tensed abdominal muscles have almost as much or more tenderness than the relaxed abdominal muscles, it is positive. Carnett hypothesized that if that pain derives from a visceral source, the tensed abdominal muscles protect the underlying structures, and therefore the tenderness should be reduced, whereas continued tenderness during muscle contraction indicates the abdominal wall as the origin of pain 27. Moreover, Carnett’s test is reported to be useful for diagnosing psychogenic abdominal pain 28. Confirmation of the diagnosis can be made with local anesthetic injections with or without a steroid. The probability of missing visceral disease is small (probably less than 7%) with strict adherence to diagnostic criteria and diligent observation of patients.
The incidence of anterior cutaneous nerve entrapment syndrome (ACNES) is unknown, but chronic abdominal wall pain is estimated to affect approximately 1 in 1800 individuals experiencing abdominal wall pain and occurs in about 30% of all patients presenting with chronic abdominal pain 26, 2, 9. Chronic abdominal wall pain may account for about 10% of patients with chronic idiopathic abdominal pain seen in gastroenterological practices 2.
Chronic abdominal wall pain can affect patients of all ages, but most commonly presents in the fifth and sixth decades of life and is 4 times more prevalent in women than in men 16. Comorbid disorders commonly seen in patients with chronic abdominal wall pain include obesity (38.1%), gastroesophageal reflux disease [GERD] (27.1%), irritable bowel syndrome [IBS] (21.8%), and fibromyalgia (9.9%) 16.
Chronic abdominal wall pain diagnosis is largely based on clinical history and physical examination 29. The patient history should include the duration, onset, description, and intensity of symptoms along with exacerbating and alleviating factors. Patients with chronic abdominal wall pain classically present with chronic (lasting more than 3 months), sharp pain that is localized to a focal part of the abdomen 2. The pain worsens with actions that tense the abdominal muscles, such as standing, sitting, or coughing, and may improve in the supine position. Confirmation of the diagnosis can be made with local anesthetic injections with or without a steroid.
The mainstay of treatment of chronic abdominal wall pain consists of reassurance, activity modification, physical therapy, and pain relief 29. The first and most important step in the management of patients with chronic abdominal wall pain is to provide reassurance that although symptoms can be quite painful and disabling, they are typically nonprogressive and have no long-term health complications 29. Activity modification involves eliminating potential triggers for the abdominal pain, such as vigorous exercises that tense the abdominal muscles or lifting excessive weights 29. An abdominal binder may be helpful in patients in whom applying gentle hand pressure to the abdomen provides relief. Physical therapy can increase the strength, mobility, and flexibility of abdominal muscles and help decrease the intensity of pain 30. For patients with mild symptoms reassurance and activity modification can often help allay patient concerns and enough as the sole treatment 2
For patients with moderate to severe symptoms, pain relief is typically achieved with a trigger point injection using an anesthetic agent with or without a steroid 29. The clinical response is better with combination therapy than with an anesthetic agent alone 3. A commonly used combination consists of 2 mL of 1% lidocaine and 0.5 mL of betamethasone administered to the site of maximal tenderness. Before referring patients for trigger point injection, physicians should review patient allergies to ensure that the patient does not have an allergy to the medications present in the trigger point injection. A comparison between pain relief with a saline injection and that with an anesthetic agent revealed that the latter was superior, arguing against a placebo response 10. Pain relief with a trigger point injection can begin within a few hours because of lidocaine, but may take several days to achieve full effect with the corticosteroid. Patients with partial relief of symptoms or recurrent symptoms after complete remission following 1 trigger point injection could be offered a second injection 1 month after the first. Approximately a quarter to one-third of patients have lasting relief after a single trigger point injection and one-half to three-fourths have sustained, long-term, partial or complete response after multiple trigger point injections 31, 3, 32.
Figure 1. Anterior cutaneous nerve
Footnote: Cross section of thorax showing course of intercostal nerve as it turns sharply from its course between the internal oblique and transversus abdominis muscles anteriorly toward the rectus abdominis. (a) Anterolateral view of the spinal nerves and their subdivisions in relation to the spinal cord and vertebrae. (b) Cross section of the thorax showing innervation of muscles and skin of the chest and back. This section is cut through the intercostal muscles between two ribs.
Figure 2. Carnett’s test
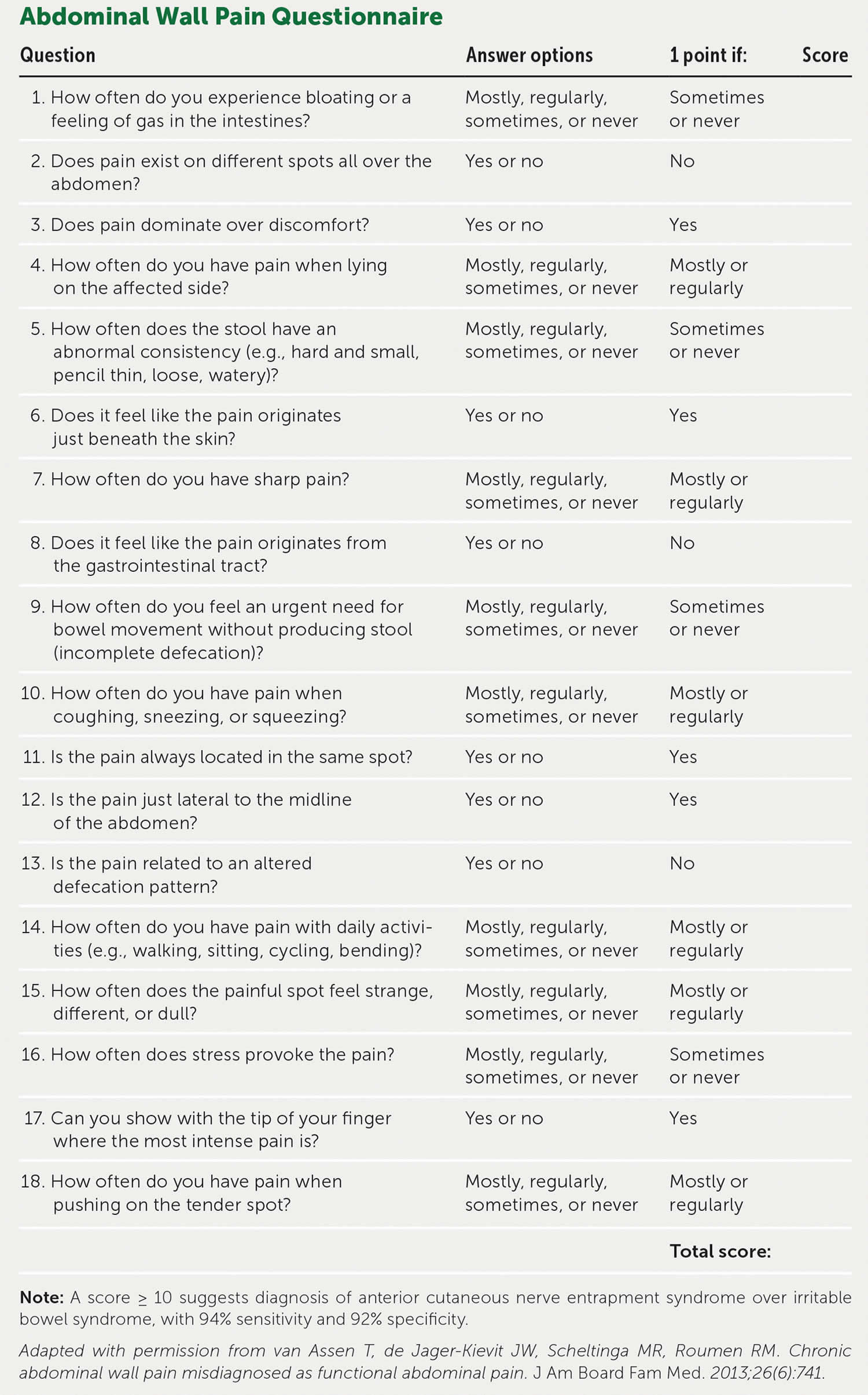
[Source 33 ]Figure 3. Chronic abdominal wall pain questionnaire
Footnote: A score ≥ 10 suggests diagnosis of anterior cutaneous nerve entrapment syndrome over irritable bowel syndrome, with 94% sensitivity and 92% specificity.
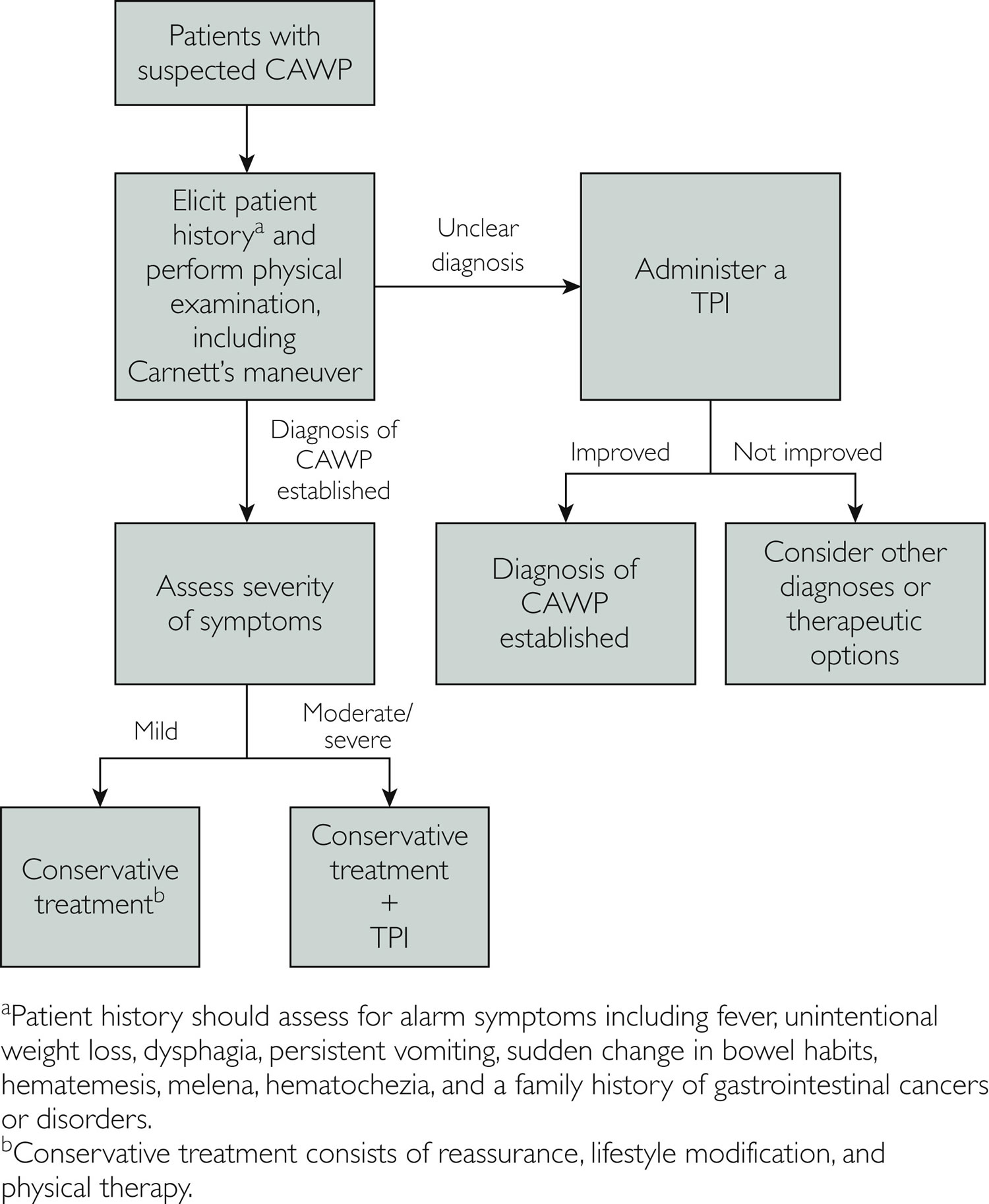
[Source 34 ]Figure 4. Chronic abdominal wall pain diagnostic and treatment algorithm
Footnote: Diagnostic and treatment algorithm for patients with chronic abdominal wall pain (CAWP).
Abbreviation: CAWP = chronic abdominal wall pain; TPI = trigger point injection.
[Source 29 ]Figure 5. Chronic abdominal wall pain treatment
Footnote: Chronic abdominal wall pain (anterior cutaneous nerve entrapment syndrome) treatment algorithm.
[Source 34 ]Chronic abdominal wall pain cause
Chronic abdominal wall pain is caused by the entrapment of the anterior cutaneous branches of sensory nerves that supply the abdominal wall 17, 18, 19. The pain is often localized in nature, as it is mediated by A delta nociceptors; in contrast, visceral pain is diffuse and involves C-type nociceptors 35. Cutaneous branches of the T7 through T12 nerve roots extend anteriorly from the spine toward the rectus sheath through a fibrous ring in the posterior sheath of the rectus abdominus 17. These cutaneous nerves travel in a neurovascular bundle surrounded by a collection of fat that serves to reduce friction and allow seamless sliding of the nerve 17. However, pain can occur if this fat is disrupted, herniated, or compressed. Mechanical abnormalities such as tissue edema, fibrosis, or scarring can result in entrapment of the cutaneous nerves as they pass through the fibrous ring 2. Patients with a history of abdominal surgery are at higher risk of developing chronic abdominal wall pain because of mechanical scarring and compression. Furthermore, pregnancy and oral contraceptives may be risk factors for chronic abdominal wall pain because of hormone-induced fluid retention or redistribution in the abdominal wall that may lead to nerve entrapment at sites of previous abdominal operations 36, 37.
Chronic abdominal wall pain signs and symptoms
Chronic abdominal wall pain has several characteristic clinical features that help differentiate it from other causes of of chronic abdominal pain. Two key clinical features of chronic abdominal wall pain are that it is typically well-localized and positional in nature.
Chronic abdominal wall pain diagnosis
Chronic abdominal wall pain diagnosis is largely based on clinical history and physical examination 29. The patient history should include the duration, onset, description, and intensity of symptoms along with exacerbating and alleviating factors. Patients with chronic abdominal wall pain classically present with chronic (lasting more than 3 months), sharp pain that is localized to a focal part of the abdomen 2. The pain worsens with actions that tense the abdominal muscles, such as standing, sitting, or coughing, and may improve in the supine position.
Patients should be asked about their medical history, as patients with chronic abdominal wall pain often tend to have a history of abdominal surgeries, and the pain may be localized near a scar. The social history should focus on patient work description and recreational activities to identify any potentially reversible lifestyle triggers. An exhaustive review of systems should be performed to identify any alarm symptoms, such as fever, unintentional weight loss, difficulty swallowing (dysphagia), persistent vomiting, sudden change in bowel habits, vomiting blood (hematemesis), melena (black, tarry stool that comes from bleeding in your upper gastrointestinal tract), hematochezia (the passage of fresh blood through the anus, usually in or with stools), and a family history of gastrointestinal cancers or diseases such as inflammatory bowel disease (IBD) or celiac disease. Patients with alarm features are more likely to have an organic disorder of the digestive tract rather than chronic abdominal wall pain.
The next step in the evaluation of such patients is to perform a thorough physical examination, including a detailed abdominal examination. A rectal examination may also be necessary, depending on the patient history. Patients with chronic abdominal wall pain are typically able to point to the area of maximal tenderness with 1 finger. The presence of a positive Carnett’s sign can be assessed using a 2-step examination technique and should be done in all patients suspected of having chronic abdominal wall pain (Figure 2). The clinician first identifies and palpates the area of maximal tenderness while the patient is in a resting supine position (step 1). The patient is then asked to raise both legs off the examination table or raise their head and shoulders off the bed so as to tense the abdominal muscles while the clinician palpates the abdomen (step 2). The Carnett’s maneuver is considered positive when palpation of the abdomen in the tense position elicits the same or more tenderness as the resting position. The patient is then allowed to relax the abdominal musculature, and tenderness typically lessens.
It may be reasonable to perform basic laboratory tests in patients presenting with nonspecific chronic abdominal pain, including complete blood count, electrolyte panel, creatinine level, calcium level, liver biochemistries, lipase level, C-reactive protein level, and thyroid-stimulating hormone level. However, any laboratory and/or imaging tests should be guided by patient history and physical examination. Patients with alarm symptoms should undergo further testing to rule out organic causes of their symptoms. In patients in whom the diagnosis remains unclear but chronic abdominal wall pain seems likely, a trigger point injection can help confirm the diagnosis of chronic abdominal wall pain, as some patients have immediate relief of their symptoms after injection with an anesthetic agent 38. For that reason, a trigger point injection has both diagnostic and therapeutic benefits. Therefore, the diagnosis of chronic abdominal wall pain can be made when a patient presents with well-localized pain over a small part of the abdomen, exhibits a positive Carnett’s sign, and has a good response to a trigger point injection 29.
Carnett sign
The Carnett sign describes an examination finding used to distinguish pain arising from the abdominal wall from pain arising from within the abdomen itself.
Testing for Carnett sign is performed as a two-stage procedure:
- The examiner locates the point of maximal tenderness through palpation of the abdomen with the patient’s abdominal muscles relaxed
- The patient then tenses their abdominal muscles (for example, by lifting their head from the pillow or raising their legs with the knees straightened) and the examiner then applies pressure to the same point of tenderness
Carnett test is postive if the pain remains the same or worsens with tension of the rectus muscles. A positive test is suggestive that the source of the pain is within the abdominal wall as opposed to the abdominal cavity. Differentials of a postive test include anterior cutaneous nerve entrapment and rectus sheath hematoma 39.
Chronic abdominal wall pain differential diagnosis
Other conditions that can present similarly to chronic abdominal wall pain include abdominal wall hernias, abdominal wall endometriosis, slipping rib syndrome, thoracic radiculopathy, postherpetic neuralgia, and iliocostal impingement syndrome 40. Slipping rib syndrome occurs because of hypermobility of ribs 8 through 12 and should be suspected in patients with upper abdominal pain that has a positional component and a positive hooking maneuver on examination 41. The hooking maneuver consists of the clinician placing his fingers in the subcostal area and pulling anteriorly with pain or clicking indicating a positive test result 41.
Patients with thoracic spinal canal or foraminal disease are at risk of thoracic radiculopathy that can result in referred abdominal pain if the T7 through T12 nerve roots are involved 42, 43.
Patients with diabetes can also develop spontaneous thoracic radiculopathy. Iliocostal impingement syndrome, also known as iliocostal friction syndrome, classically occurs in patients with kyphosis and osteoporosis when there is contact between the inferior ribs and the ilium, which can lead to abdominal, flank, or back pain 44.
Those with postherpetic neuralgia will often recall an active case of shingles, but even if that is not the case, tenderness tends to follow dermatomal segments and is superficial, with notable allodynia.
Chronic abdominal wall pain treatment
The mainstay of treatment of chronic abdominal wall pain consists of reassurance, activity modification, physical therapy, and pain relief 29. The first and most important step in the management of patients with chronic abdominal wall pain is to provide reassurance that although symptoms can be quite painful and disabling, they are typically nonprogressive and have no long-term health complications 29. Activity modification involves eliminating potential triggers for the abdominal pain, such as vigorous exercises that tense the abdominal muscles or lifting excessive weights 29. An abdominal binder may be helpful in patients in whom applying gentle hand pressure to the abdomen provides relief. Physical therapy can increase the strength, mobility, and flexibility of abdominal muscles and help decrease the intensity of pain 30. For patients with mild symptoms reassurance and activity modification can often help allay patient concerns and enough as the sole treatment 2
For patients with moderate to severe symptoms, pain relief is typically achieved with a trigger point injection using an anesthetic agent with or without a steroid 29. The clinical response is better with combination therapy than with an anesthetic agent alone 3. A commonly used combination consists of 2 mL of 1% lidocaine and 0.5 mL of betamethasone administered to the site of maximal tenderness. Before referring patients for trigger point injection, physicians should review patient allergies to ensure that the patient does not have an allergy to the medications present in the trigger point injection. A comparison between pain relief with a saline injection and that with an anesthetic agent revealed that the latter was superior, arguing against a placebo response 10. Pain relief with a trigger point injection can begin within a few hours because of lidocaine, but may take several days to achieve full effect with the corticosteroid. Patients with partial relief of symptoms or recurrent symptoms after complete remission following 1 trigger point injection could be offered a second injection 1 month after the first. Approximately a quarter to one-third of patients have lasting relief after a single trigger point injection and one-half to three-fourths have sustained, long-term, partial or complete response after multiple trigger point injections 31, 3, 32.
Ultrasound-guided trigger point injection can be more costly than blind trigger point injection, but may offer improved accuracy of injection and decreased risk of injecting into the peritoneal cavity. A trigger point injection may be difficult to perform in obese patients, and a longer trigger point injection needle may be required in these cases 45, 46. In addition, the use of ultrasound guidance in obese patients may help ensure that the depth of needle insertion is adequate. Although trigger point injections are generally considered a low-risk procedure and antiplatelet agents, including aspirin and clopidogrel, do not need to be discontinued for the procedure, careful consideration should be given if the patient is receiving dual antiplatelet therapy 46. Shared assessment and risk stratification should be performed regarding anticoagulation management with warfarin or direct oral anticoagulants before the procedure 46. The depth of needle insertion may affect the risk of bleeding and should also be taken into consideration.
Other adjunctive treatment alternatives to trigger point injections may include the use of heating pads and lidocaine patches. Systemic therapy may include nonopioid analgesic agents (acetaminophen and nonsteroidal anti-inflammatory drugs), antiepileptic agents (gabapentin and pregabalin), and low-dose tricyclic antidepressants (amitriptyline), although these confer limited benefit given the underlying disease pathogenesis 47.
If patients fail to respond after the above interventions, other causes of chronic abdominal wall pain should be considered and evaluated. Some patients may exhibit a positive Carnett’s sign, but may not respond to treatment with a trigger point injection 32. Other patients with chronic abdominal wall pain may have symptoms that are responsive, but recur despite treatment with multiple trigger point injections. In patients with refractory symptoms despite treatment with 3 trigger point injections administered to the same site in 1 year, chemical neurolysis with aqueous phenol or surgical treatment with neurectomy can be considered 48, 49.
Trigger point injections
A specific trigger point on the abdominal wall represents the area of nerve entrapment in most cases, and an injection of local anesthetic at this point could be used for confirmation of anterior cutaneous nerve entrapment syndrome and for therapeutic reasons 23, 3, 2, 10. The blind trigger point injection technique, both in adults and children, is always diagnostic and sometimes therapeutic, but might also fail due to various reasons. The main reasons for this failure, as described by Koop et al 3, include selecting a wrong site for the injection in the blind technique (especially in obese patients or those with some kind of altered anatomy, such as previous surgery, scars, and pregnancy) or owing to a different cause of pain with an incorrect diagnosis. Therefore, from the current literature, it can be concluded that ultrasound-guided techniques using local anesthetics initially and then corticosteroids in refractory states can help in accurate administration of drugs injected and avoid complications. However, no study compares ultrasound-guided and freehand techniques. The ideal ultrasound-guided block for anterior cutaneous nerve entrapment syndrome (trigger point injections around the site of entrapment, rectus sheath block, or transversus abdominis plane block) as well as the ideal type and volume of the drug used (local anesthetic alone or combined with a corticosteroid) have not yet defined. Therefore, future research needs to be directed in this way, as the ultrasound-guided techniques facilitate accurate injections with minimization of side effects.
Chemical neurolysis
The use of chemical neurolysis, using absolute alcohol or phenol at the exact point of the nerve entrapment, has also been described in the literature as an option for treating anterior cutaneous nerve entrapment syndrome 23. However, there are no reports about the long-term effects of this neurodestructive modality, and in the literature, no study carried out in the last decade is available.
In a study by Mehta and Ranger 50, a large series of patients with chronic abdominal pain is described. Some of these patients had the characteristics of anterior cutaneous nerve entrapment syndrome, although the syndrome was not described in detail at the time of the study (1971). All patients had a specific tender point of severe pain over the abdominal wall. The authors managed pain with lidocaine injections, along with a corticosteroid (hydrocortisone), and then by neurolysis of the nerve endings using dilute aqueous phenol (5%) and a neurostimulator to identify the relevant nerve’s exit point. Among the 103 patients with chronic abdominal pain, complete pain relief was achieved in 60 patients, partial relief in 33, and there was no response in 10 patients. At a long-term follow-up of 3–4 years, relief was sustained in 58 patients. In this study, no information was given regarding the side effects of the technique or about reoccurrence of pain after a longer period 50.
McGrady and Marks 48 in another study, followed a protocol of using a nerve stimulator for identifying the entrapped nerve in 76 patients with anterior cutaneous nerve entrapment syndrome, and they employed chemical neurolysis using aqueous 6% phenol for treatment. Among the 44 patients with anterior cutaneous nerve entrapment syndrome, 42 exhibited significant improvement in their pain after the treatment. On the other hand, among the 35 patients with possible anterior cutaneous nerve entrapment syndrome, only 16 achieved pain relief after the treatment. The mechanism of action of phenol in both the studies could not be explained, since very dilute mixtures were used. According to the authors, a possible mechanism may be the formation of scar tissue around the entrapped nerve, thus preventing its herniation. The pinprick sensation was maintained after the injection. However, no further studies on this technique could be identified in the literature, thus limiting the clinical applicability of this technique 47.
Radiofrequency ablation
Although there is only one report about the application of radiofrequency ablation for patients with anterior cutaneous nerve entrapment syndrome 51, the published studies regarding the other causes of intercostal neuralgia such as blunt trauma, surgery, and postherpetic neuralgia reported good results after thermal radiofrequency ablation of the nerve itself or of the relevant dorsal root ganglion 52, 53, 54, 55, 56, 57.
Tellez Villajos et al 51 published a case of anterior cutaneous nerve entrapment syndrome management with radiofrequency ablation of the dorsal root ganglia at the level of T11–T12 with good results. Stolker et al 52 and van Kleef and Spaans 53 used pulsed radiofrequency in other cases of intercostal neuralgia and observed a significant reduction in pain, while another study reported better results with the pulsed radiofrequency of the dorsal root ganglia 56. The classic thermal radiofrequency ablation (performed at 80°C for 90 seconds under fluoroscopic guidance and after sensory and motor testing) was also effective in six patients suffering from traumatic intercostal neuralgia from the fourth to the twelfth thoracic nerve level 54. In both techniques, postprocedural pain and pneumothorax were the most frequent complications 54. There is also another case report presented by Birthi et al 57 reporting a patient with chronic abdominal pain who was treated with bilateral pulsed radiofrequency at the level of T10–T11. However, this technique has not been tested in a systematic manner or compared with other treatment options, and its long-term results are unknown.
Van Kleef et al 58 in their evidence-based guidelines for interventional pain management, rated both the pulsed and the thermal radiofrequency approaches of the dorsal root ganglion in the cases of intercostal neuralgia as 2C+ (effectiveness only demonstrated in observational studies). However, anterior cutaneous nerve entrapment syndrome has a different cause; the mechanical irritation of the nerve in its peripheral course is the main cause of injury, but it is still an intercostal neuralgia, and the efficacy of radiofrequency ablation in the refractory cases, when other interventional techniques or surgery fails to provide pain relief, is yet to be defined.
Neuromodulation techniques
The application of neuromodulation techniques, especially peripheral or spinal nerve stimulation has not yet been tried in the therapy of anterior cutaneous nerve entrapment syndrome. However, it might be effective in the management of the disease after the failure of all interventional and surgical therapies, as it has been found to be effective in other forms of neuropathic pain of different origin and etiology. The exact role of nerve stimulation in anterior cutaneous nerve entrapment syndrome is yet to be unraveled, as there is no current evidence to support its use 58, 59.
Botulinum toxin injection
As an alternative technique, Weum and de Weerd 60 tried an ultrasound-guided injection of botulinum toxin at the site of maximal tenderness in 15 patients with anterior cutaneous nerve entrapment syndrome. The authors used the Doppler ultrasound and injected 40 IU/mL of botulinum toxin without local anesthetic in the proximity of the nerve 60. The number of injections for each patient ranged from 2 to 35 and depended on the duration of the action of botulinum toxin in an individual. The advantages of this technique compared to other ultrasound-guided injections are the localization of the perforator complex at the abdominal wall, the administration of the drug in the proximity to the nerve using the perforator as a guide, and most importantly the drug used 60. However, in this study, there were no detailed outcome measures, so it needs to be further evaluated in a more systematic manner.
Surgical anterior neurectomy
Surgical anterior neurectomy has been described as a therapeutic option for anterior cutaneous nerve entrapment syndrome in the patients not responding to conservative treatments. Boelens et al 61 published the largest database of patients with anterior cutaneous nerve entrapment syndrome who were diagnostically confirmed with the trigger point injection of 1% lidocaine and corticosteroid at the site of maximal tenderness. In the case of refractory pain, the patients underwent an open anterior neurectomy. Among these patients, 139 were followed by determining the pain score (Visual Analogue Scale [VAS] 0–100 and Verbal Rating Scale [VRS] 1–5) as an outcome measure for long-term efficacy of the technique 61. The majority of patients (94 patients; 81%) had a significant reduction in the pain (of at least 50% compared to the initial value) after the first local anesthetic injection, and 44 of them achieved permanent relief 61. In the end, 69 patients underwent anterior neurectomy, which was successful in 71% of them (Verbal Rating Scale [VRS] ranging between 1 and 2), whereas 9% of patients showed moderate cure for at least 1 year 61.
Boelens et al 11 were the first to conduct a randomized controlled trial for the treatment of anterior cutaneous nerve entrapment syndrome with surgical neurectomy employing a sham (placebo) group. They studied patients (aged >18 years) diagnosed with anterior cutaneous nerve entrapment syndrome according to specific criteria 61. All patients had a diagnostic injection at the relevant trigger point with 1% lidocaine (positive when >50% reduction of pain), but later the relief subsided. The patients were randomly allocated into two groups, with 22 patients in each group. Surgical neurectomy was performed by removal of the neurovascular bundle and all of its branches within a 5-cm radius from the site of fascia opening, while in the sham group only a transverse incision of 5–7 cm, exposing the anterior sheath of the rectus abdominis, was made. The main successful outcome was determined as >50% of pain reduction in Visual Analogue Scale [VAS] score (0–100) or >2 points in Verbal Rating Scale [VRS] (0–4). Based on this outcome measure, it can be gleaned that the neurectomy patients had significantly more pain relief (16/22 neurectomy versus 4/22 sham procedure) after 6 weeks of follow-up. The 18 patients in the sham group, who had no positive outcome were then reoperated after deblinding, and 11 of them showed a positive response to the treatment. The authors pointed out the necessity of using specific diagnostic criteria for anterior cutaneous nerve entrapment syndrome and recommended a series of injections before the procedure 11. However, the surgery was not successful in all patients, which might be due to the anatomic abnormalities leading to the entrapment of the nerve at more dorsal or lateral sites of the rectus abdominis and not at the suspected area, that was the site of the surgery 11, 23, 24. In these cases, the diagnostic injection was mostly positive, but surgery was not. The authors suggested a surgical exploration of a 5×5 cm area around the pain site to identify the possible nerve branches and reduce surgical failure 61.
Van Assen et al 62 described the possible options after a failed anterior neurectomy in patients suffering from anterior cutaneous nerve entrapment syndrome. The failed neurectomy was defined as “continued or recurrent pain after the initial procedure”. Forty-one patients were reoperated, and a posterior neurectomy and/or re-exploration of the operated site (secondary anterior neurectomy) was performed. Among the 41 patients (26 with failed neurectomy and 15 with a recurrence of pain), 66% (27 patients) were significantly improved after the reoperation as followed up to 25 months. Especially the patients who experienced recurrent pain after an initially successful operation had a better outcome compared to the patients in whom the operation was unsuccessful from the beginning. The authors pointed out that approximately one-third of the patients who are operated for anterior cutaneous nerve entrapment syndrome do not respond adequately to this treatment, and the therapeutic options afterward are limited, including mainly systemic pharmacotherapy. Further exploration of the operated area and performing a reoperation may help identify and remove the remaining or persistent nerve endings that cause the pain (secondary anterior neurectomy) or remove the bundle at another level, at the posterior abdominal fascia of the rectus abdominis (posterior neurectomy), leading possibly to a better result 62. Data from this study could also be helpful in identifying the success rate of anterior cutaneous nerve entrapment syndrome treatment including various options. The authors presented their data from 181 procedures (154 patients) 49 and described the treatment efficacy of their stepwise approach: beginning with injections followed by anterior neurectomy in nonresponding cases. Pain scores were recorded using the numerical rating scale (NRS) (0–10) as well as the 6-point verbal category rating scale. In all cases, “success” was defined as a ≥50% reduction in numerical rating scale (NRS) or ≥2 points in category scale. The efficacy of trigger point injections alone was 33% for long-term pain relief. The success score for neurectomy was 70% for short-term efficacy (1–3 months), while the overall long-term efficacy was 61% (after a mean time frame of 32 months). This large database of anterior cutaneous nerve entrapment syndrome patients can be useful in designing a treatment strategy for anterior cutaneous nerve entrapment syndrome. The authors suggested that the initial treatment should be local injections (step 1), followed by anterior neurectomy (step 2), and then reoperation in failed cases (step 3). This approach may achieve up to 90% success rate, as reported by the study authors 62, 49.
Intraperitoneal onlay mesh reinforcement
Since surgery represents a major option of treatment in refractory cases of anterior cutaneous nerve entrapment syndrome, new methods are emerging to provide more long-lasting pain relief. Stirler et al 63 suggested that intraperitoneal onlay mesh reinforcement may prevent the entrapment of anterior cutaneous nerves of the neurovascular bundle in patients with refractory anterior cutaneous nerve entrapment syndrome. All 40 patients who participated in the study had a prior injection of a local anesthetic combined with a corticosteroid (>2 consecutive injections at the relevant trigger point), but there was no permanent relief in 30 of them 63. This study presents the results of the technique in those 30 patients who were scheduled for a laparoscopic insertion of polytetrafluoroethylene mesh at the painful area, aiming to overlap the site of pain, covering at least 4 cm area in all directions. The retrospective analysis of these patients showed excellent short- and long-term success rates of 90% and 71%, respectively. The patients were evaluated for their satisfaction after the treatment based on a verbal rating on a scale of 1–5 (with one being the best satisfaction and five the worst; only scores of 1 and 2 were classified as successful treatment). One patient developed pain 1 month after the operation and underwent another operation using the same technique with successful results. The novelty of this technique relies on the fact that neurovascular bundles of the anterior cutaneous nerves emerging from the lower intercostal nerves normally move freely through a fibrous ring in the abdominal wall. In the patients suffering from anterior cutaneous nerve entrapment syndrome, this bundle is compressed and entrapped on this ring. The application of the mesh may decrease the intra-abdominal pressure on the fibrous ring, therefore preventing the entrapment of the nerve and occurrence of pain. This technique needs further assessment through more studies, preferably randomized controlled trials, in order to provide accurate data about its efficacy and long-term outcome.
- Applegate WV. Abdominal cutaneous nerve entrapment syndrome (ACNES): a commonly overlooked cause of abdominal pain. Perm J 2002;6:20–27.[↩]
- Srinivasan R, Greenbaum DS. Chronic abdominal wall pain: a frequently overlooked problem. Practical approach to diagnosis and management. Am J Gastroenterol. 2002 Apr;97(4):824-30. doi: 10.1111/j.1572-0241.2002.05662.x[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Koop H, Koprdova S, Schürmann C. Chronic Abdominal Wall Pain. Dtsch Arztebl Int. 2016 Jan 29;113(4):51-7. doi: 10.3238/arztebl.2016.0051[↩][↩][↩][↩][↩][↩][↩][↩]
- Hidalgo, Diego F. MD1; Phemister, Jennifer MD1; Ordoñez, Andrea C. Ortiz MD2; James, Swenson MD3; Young, Mark MD1. Carnett’s Sign: An Easy Tool That Saves Unnecessary Expenses in the Evaluation of Chronic Abdominal Pain: 1402. American Journal of Gastroenterology 112():p S760-S761, October 2017. https://journals.lww.com/ajg/fulltext/2017/10001/carnett_s_sign__an_easy_tool_that_saves.1403.aspx[↩][↩]
- Lindsetmo RO, Stulberg J. Chronic abdominal wall pain–a diagnostic challenge for the surgeon. Am J Surg. 2009 Jul;198(1):129-34. doi: 10.1016/j.amjsurg.2008.10.027[↩][↩]
- Sun XX, Liu H, Qin XZ, Li MR, Yan QH, Zhang GJ. The Diagnostic Value of Carnett’s Test with Chronic Abdominal Pain: A Narrative Review. Curr Pain Headache Rep. 2024 Apr;28(4):251-257. doi: 10.1007/s11916-024-01223-9[↩]
- Greenbaum DS, Greenbaum RB, Joseph JG, Natale JE. Chronic abdominal wall pain. Dig Dis Sci. 1994;39:1935–1941. doi: 10.1007/BF02088128[↩][↩]
- Sharpstone D, Colin-Jones DG. Chronic, non-visceral abdominal pain. Gut. 1994 Jun;35(6):833-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1374889/pdf/gut00540-0117.pdf[↩]
- van Assen T, de Jager-Kievit JW, Scheltinga MR, Roumen RM. Chronic abdominal wall pain misdiagnosed as functional abdominal pain. J Am Board Fam Med. 2013 Nov-Dec;26(6):738-44. doi: 10.3122/jabfm.2013.06.130115[↩][↩]
- Boelens OB, Scheltinga MR, Houterman S, Roumen RM. Randomized clinical trial of trigger point infiltration with lidocaine to diagnose anterior cutaneous nerve entrapment syndrome. Br J Surg. 2013 Jan;100(2):217-21. doi: 10.1002/bjs.8958[↩][↩][↩][↩]
- Boelens OB, van Assen T, Houterman S, Scheltinga MR, Roumen RM. A double-blind, randomized, controlled trial on surgery for chronic abdominal pain due to anterior cutaneous nerve entrapment syndrome. Ann Surg. 2013 May;257(5):845-9. doi: 10.1097/SLA.0b013e318285f930[↩][↩][↩][↩]
- Hershfield NB. The abdominal wall. A frequently overlooked source of abdominal pain. J Clin Gastroenterol. 1992;14:199–202. doi: 10.1097/00004836-199204000-00004[↩]
- Loos MJ, Scheltinga MR, Roumen RM. Tailored neurectomy for treatment of postherniorrhaphy inguinal neuralgia. Surgery. 2010 Feb;147(2):275-81. doi: 10.1016/j.surg.2009.08.008[↩]
- Choi YK, Chou S. Rectus syndrome. Another cause of upper abdominal pain. Reg Anesth. 1995 Jul-Aug;20(4):347-51.[↩]
- Haynsworth RF Jr, Noe CE. An unusual presentation of intercostal neuralgia. Anesthesiology. 1990 Oct;73(4):779-80. doi: 10.1097/00000542-199010000-00027[↩]
- Costanza CD, Longstreth GF, Liu AL. Chronic abdominal wall pain: clinical features, health care costs, and long-term outcome. Clin Gastroenterol Hepatol. 2004 May;2(5):395-9. doi: 10.1016/s1542-3565(04)00124-7[↩][↩][↩]
- Abdominal cutaneous nerve entrapment syndrome. Contin Educ Anaesth Crit Care Pain. 2015; 15: 60-63. https://www.bjaed.org/article/S1743-1816(17)30002-1/fulltext[↩][↩][↩][↩]
- Kanakarajan S, Dharmavaram S, Tadros A, Pushparaj H, Rose A. Abdominal cutaneous nerve entrapment syndrome: A cross sectional survey of treatment outcomes. Br J Pain. 2022 Oct;16(5):538-545. doi: 10.1177/20494637221101719[↩][↩]
- Oor JE, Ünlü Ç, Hazebroek EJ. A systematic review of the treatment for abdominal cutaneous nerve entrapment syndrome. Am J Surg. 2016 Jul;212(1):165-74. doi: 10.1016/j.amjsurg.2015.12.013[↩][↩]
- Koop H, Schürmann C. Chronischer Bauchwandschmerz. Gastroenterol up2date. 2014;10:129–140.[↩][↩]
- Kanakarajan S, High K, Nagaraja R. Chronic abdominal wall pain and ultrasound-guided abdominal cutaneous nerve infiltration: a case series. Pain Med. 2011 Mar;12(3):382-6. doi: 10.1111/j.1526-4637.2011.01056.x[↩]
- Siawash M, Maatman R, Tjon A Ten W, van Heurn E, Roumen R, Scheltinga M. Anterior neurectomy in children with a recalcitrant anterior cutaneous nerve entrapment syndrome is safe and successful. J Pediatr Surg. 2017 Mar;52(3):478-480. doi: 10.1016/j.jpedsurg.2016.08.020[↩]
- Applegate WV. Abdominal cutaneous nerve entrapment syndrome (ACNES): a commonly overlooked cause of abdominal pain. Perm J. 2002;6:20–27.[↩][↩][↩][↩][↩]
- Applegate WV, Buckwalter NR. Microanatomy of the structures contributing to abdominal cutaneous nerve entrapment syndrome. J Am Board Fam Pract. 1997 Sep-Oct;10(5):329-32.[↩][↩]
- Applegate WV. Abdominal cutaneous nerve entrapment syndrome. Surgery. 1972 Jan;71(1):118-24.[↩][↩]
- van Assen T, Brouns JA, Scheltinga MR, Roumen RM. Incidence of abdominal pain due to the anterior cutaneous nerve entrapment syndrome in an emergency department. Scand J Trauma Resusc Emerg Med. 2015 Feb 8;23:19. doi: 10.1186/s13049-015-0096-0[↩][↩]
- Carnett JB. Intercostal neuralgia as a cause of abdominal pain and tenderness. Surg Gynecol Obstet. 1926;42:625–632.[↩][↩][↩]
- Takada T, Ikusaka M, Ohira Y, Noda K, Tsukamoto T. Diagnostic usefulness of Carnett’s test in psychogenic abdominal pain. Intern Med. 2011;50:213–217. doi: 10.2169/internalmedicine.50.4179[↩]
- Kamboj AK, Hoversten P, Oxentenko AS. Chronic Abdominal Wall Pain: A Common Yet Overlooked Etiology of Chronic Abdominal Pain. Mayo Clin Proc. 2019 Jan;94(1):139-144. https://www.mayoclinicproceedings.org/article/S0025-6196(18)30852-8/fulltext[↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Rodeghero J.R., Denninger T.R., Ross M.D. Abdominal pain in physical therapy practice: 3 patient cases. J Orthop Sport Phys Ther. 2013; 43 ([published correction appears in J Orthop Sports Phys Ther. 2013;43(2):196]): 44-53[↩][↩]
- Nazareno J., Ponich T., Gregor J. Long-term follow-up of trigger point injections for abdominal wall pain. Can J Gastroenterol. 2005; 19: 561-565[↩][↩]
- Alnahhas MF, Oxentenko SC, Locke GR 3rd, Hansel S, Schleck CD, Zinsmeister AR, Farrugia G, Grover M. Outcomes of Ultrasound-Guided Trigger Point Injection for Abdominal Wall Pain. Dig Dis Sci. 2016 Feb;61(2):572-7. doi: 10.1007/s10620-015-3857-8[↩][↩][↩]
- Kijima T, Hyakudomi R, Hashimoto T, Kusaka A, Nakatani T, Ishibashi Y. Adhesion-induced chronic abdominal pain: a case report on the diagnostic value of Carnett’s test. J Med Case Rep. 2019 Apr 18;13(1):93. doi: 10.1186/s13256-019-2026-7[↩]
- Shian B, Larson ST. Abdominal Wall Pain: Clinical Evaluation, Differential Diagnosis, and Treatment. Am Fam Physician. 2018 Oct 1;98(7):429-436. https://www.aafp.org/pubs/afp/issues/2018/1001/p429.html[↩][↩]
- Dubin AE, Patapoutian A. Nociceptors: the sensors of the pain pathway. J Clin Invest. 2010 Nov;120(11):3760-72. doi: 10.1172/JCI42843[↩]
- Peleg R, Gohar J, Koretz M, Peleg A. Abdominal wall pain in pregnant women caused by thoracic lateral cutaneous nerve entrapment. Eur J Obstet Gynecol Reprod Biol. 1997 Aug;74(2):169-71. doi: 10.1016/s0301-2115(97)00114-0[↩]
- Peleg R. Abdominal wall pain caused by cutaneous nerve entrapment in an adolescent girl taking oral contraceptive pills. J Adolesc Health. 1999 Jan;24(1):45-7. doi: 10.1016/s1054-139x(98)00034-2[↩]
- Greenbaum DS, Greenbaum RB, Joseph JG, Natale JE. Chronic abdominal wall pain. Diagnostic validity and costs. Dig Dis Sci. 1994 Sep;39(9):1935-41. doi: 10.1007/BF02088128[↩]
- Yale SH, Tekiner H, Yale ES. Fothergill and Carnett signs and rectus sheath hematoma. J Rural Med. 2020 Jul;15(3):130-131. doi: 10.2185/jrm.2019-019[↩]
- Kamboj AK, Hoversten P, Oxentenko AS. Chronic Abdominal Wall Pain: A Common Yet Overlooked Etiology of Chronic Abdominal Pain. Mayo Clin Proc. 2019 Jan;94(1):139-144. https://www.mayoclinicproceedings.org/article/S0025-6196(18)30852-8/fulltext).
Hernias and endometriosis of the abdominal wall can also present with localized pain and tenderness to palpation. However, these disorders can be differentiated from chronic abdominal wall pain that is secondary to anterior cutaneous nerve entrapment, as patients with hernias or endometriosis of the abdominal wall typically exhibit a palpable mass or fullness on abdominal examination ((Ecker AM, Donnellan NM, Shepherd JP, Lee TT. Abdominal wall endometriosis: 12 years of experience at a large academic institution. Am J Obstet Gynecol. 2014 Oct;211(4):363.e1-5. doi: 10.1016/j.ajog.2014.04.011[↩]
- Khan NAJ, Waseem S, Ullah S, Mehmood H. Slipping Rib Syndrome in a Female Adult with Longstanding Intractable Upper Abdominal Pain. Case Rep Med. 2018 Jul 2;2018:7484560. doi: 10.1155/2018/7484560[↩][↩]
- Ecker AM, Donnellan NM, Shepherd JP, Lee TT. Abdominal wall endometriosis: 12 years of experience at a large academic institution. Am J Obstet Gynecol. 2014 Oct;211(4):363.e1-5. doi: 10.1016/j.ajog.2014.04.011[↩]
- Whitcomb DC, Martin SP, Schoen RE, Jho HD. Chronic abdominal pain caused by thoracic disc herniation. Am J Gastroenterol. 1995 May;90(5):835-7.[↩]
- Brubaker ML, Sinaki M. Successful management of iliocostal impingement syndrome: A case series. Prosthet Orthot Int. 2016 Jun;40(3):384-7. doi: 10.1177/0309364615605394[↩]
- Suleiman S, Johnston DE. The abdominal wall: an overlooked source of pain. Am Fam Physician. 2001 Aug 1;64(3):431-8. https://www.aafp.org/pubs/afp/issues/2001/0801/p431.html[↩]
- Narouze S, Benzon HT, Provenzano DA, Buvanendran A, De Andres J, Deer TR, Rauck R, Huntoon MA. Interventional spine and pain procedures in patients on antiplatelet and anticoagulant medications: guidelines from the American Society of Regional Anesthesia and Pain Medicine, the European Society of Regional Anaesthesia and Pain Therapy, the American Academy of Pain Medicine, the International Neuromodulation Society, the North American Neuromodulation Society, and the World Institute of Pain. Reg Anesth Pain Med. 2015 May-Jun;40(3):182-212. doi: 10.1097/AAP.0000000000000223[↩][↩][↩]
- Chrona E, Kostopanagiotou G, Damigos D, Batistaki C. Anterior cutaneous nerve entrapment syndrome: management challenges. J Pain Res. 2017 Jan 13;10:145-156. doi: 10.2147/JPR.S99337[↩][↩]
- McGrady EM, Marks RL. Treatment of abdominal nerve entrapment syndrome using a nerve stimulator. Ann R Coll Surg Engl. 1988 May;70(3):120-2. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2498727/pdf/annrcse01550-0008.pdf[↩][↩]
- van Assen T, Boelens OB, van Eerten PV, Perquin C, Scheltinga MR, Roumen RM. Long-term success rates after an anterior neurectomy in patients with an abdominal cutaneous nerve entrapment syndrome. Surgery. 2015 Jan;157(1):137-43. doi: 10.1016/j.surg.2014.05.022[↩][↩][↩]
- Mehta M, Ranger I. Persistent abdominal pain. Treatment by nerve block. Anaesthesia. 1971 Jul;26(3):330-3. doi: 10.1111/j.1365-2044.1971.tb04794.x[↩][↩]
- Téllez Villajos L, Hinojal Olmedillo B, Moreira Vicente V, de la Calle Reviriego JL, Senosiain Lalastra C, Foruny Olcina JR. Radiofrecuencia pulsada en el tratamiento del síndrome de atrapamiento del nervio cutáneo abdominal [Pulsed radiofrequency in the treatment of abdominal cutaneous nerve entrapament syndrome]. Gastroenterol Hepatol. 2015 Jan;38(1):14-6. Spanish. doi: 10.1016/j.gastrohep.2014.03.010[↩][↩]
- Stolker RJ, Vervest AC, Groen GJ. The treatment of chronic thoracic segmental pain by radiofrequency percutaneous partial rhizotomy. J Neurosurg. 1994 Jun;80(6):986-92. doi: 10.3171/jns.1994.80.6.0986[↩][↩]
- van Kleef M, Barendse GA, Dingemans WA, Wingen C, Lousberg R, de Lange S, Sluijter ME. Effects of producing a radiofrequency lesion adjacent to the dorsal root ganglion in patients with thoracic segmental pain. Clin J Pain. 1995 Dec;11(4):325-32. doi: 10.1097/00002508-199512000-00010[↩][↩]
- Engel AJ. Utility of intercostal nerve conventional thermal radiofrequency ablations in the injured worker after blunt trauma. Pain Physician. 2012 Sep-Oct;15(5):E711-8.[↩][↩][↩]
- Akkaya T, Ozkan D. Ultrasound-guided pulsed radiofrequency treatment of the intercostal nerve: three cases. J Anesth. 2013 Dec;27(6):968-9. doi: 10.1007/s00540-013-1649-x[↩]
- Cohen SP, Sireci A, Wu CL, Larkin TM, Williams KA, Hurley RW. Pulsed radiofrequency of the dorsal root ganglia is superior to pharmacotherapy or pulsed radiofrequency of the intercostal nerves in the treatment of chronic postsurgical thoracic pain. Pain Physician. 2006 Jul;9(3):227-35.[↩][↩]
- Birthi P, Calhoun D, Grider JS. Pulsed radiofrequency for chronic abdominal pain. Pain Physician. 2013 Jul-Aug;16(4):E443-5.[↩][↩]
- Van Kleef M, Stolker R, Lataster A, Geurts J, Benzon H, Mekhail N. Thoracic pain. In: Van Zundert J, editor. Evidence-Based Interventional Pain Management: According to Clinical Diagnosis. Chichester, UK: Wiley-Blackwell; 2012. pp. 62–70.[↩][↩]
- Rosati C, Stain S. Abdominal pain. In: Smith H, editor. Current Therapy in Pain. Philadelphia PA: Saunders; 2009. pp. 201–204.[↩]
- Weum S, de Weerd L. Perforator-Guided Drug Injection in the Treatment of Abdominal Wall Pain. Pain Med. 2016 Jul;17(7):1229-32. doi: 10.1093/pm/pnv011[↩][↩][↩]
- Boelens OB, Scheltinga MR, Houterman S, Roumen RM. Management of anterior cutaneous nerve entrapment syndrome in a cohort of 139 patients. Ann Surg. 2011 Dec;254(6):1054-8. doi: 10.1097/SLA.0b013e31822d78b8[↩][↩][↩][↩][↩][↩]
- van Assen T, Boelens OB, van Eerten PV, Scheltinga MR, Roumen RM. Surgical options after a failed neurectomy in anterior cutaneous nerve entrapment syndrome. World J Surg. 2014 Dec;38(12):3105-11. doi: 10.1007/s00268-014-2737-2[↩][↩][↩]
- Stirler VM, Raymakers JT, Rakic S. Intraperitoneal onlay mesh reinforcement of the abdominal wall: a new surgical option for treatment of anterior cutaneous nerve entrapment syndrome-a retrospective cohort analysis of 30 consecutive patients. Surg Endosc. 2016 Jul;30(7):2711-5. doi: 10.1007/s00464-015-4533-y[↩][↩]