Contents
What is citric acid
Citric acid (2-hydroxy-1,2,3-propane-tricarboxylic acid) is a weak organic acid found in the greatest amounts in citrus fruits, such as lemon, lime, grapefruit, tangerine, and orange 1. Lemon and lime juices are rich sources of citric acid, containing 1.44 and 1.38 g/oz, respectively, comprising as much as 8% of the dry fruit weight 2. The citric acid content of commercially available lemonade and other juice products varies widely, ranging from 0.03 to 0.22 g/oz. Citric acid is frequently used as a food additive as a natural preservative and also to add an acidic (sour) taste to foods and beverages 3. Citric acid is also a key intermediate in cellular metabolism, being a component of the tricarboxylic acid or Krebs cycle, citric acid is found in all human and animal tissues as an intermediary substance in oxidative metabolism. A major source of citric acid in your body results from endogenous metabolism in the mitochondria via the production of ATP in the citric acid cycle (see Citric acid cycle below).
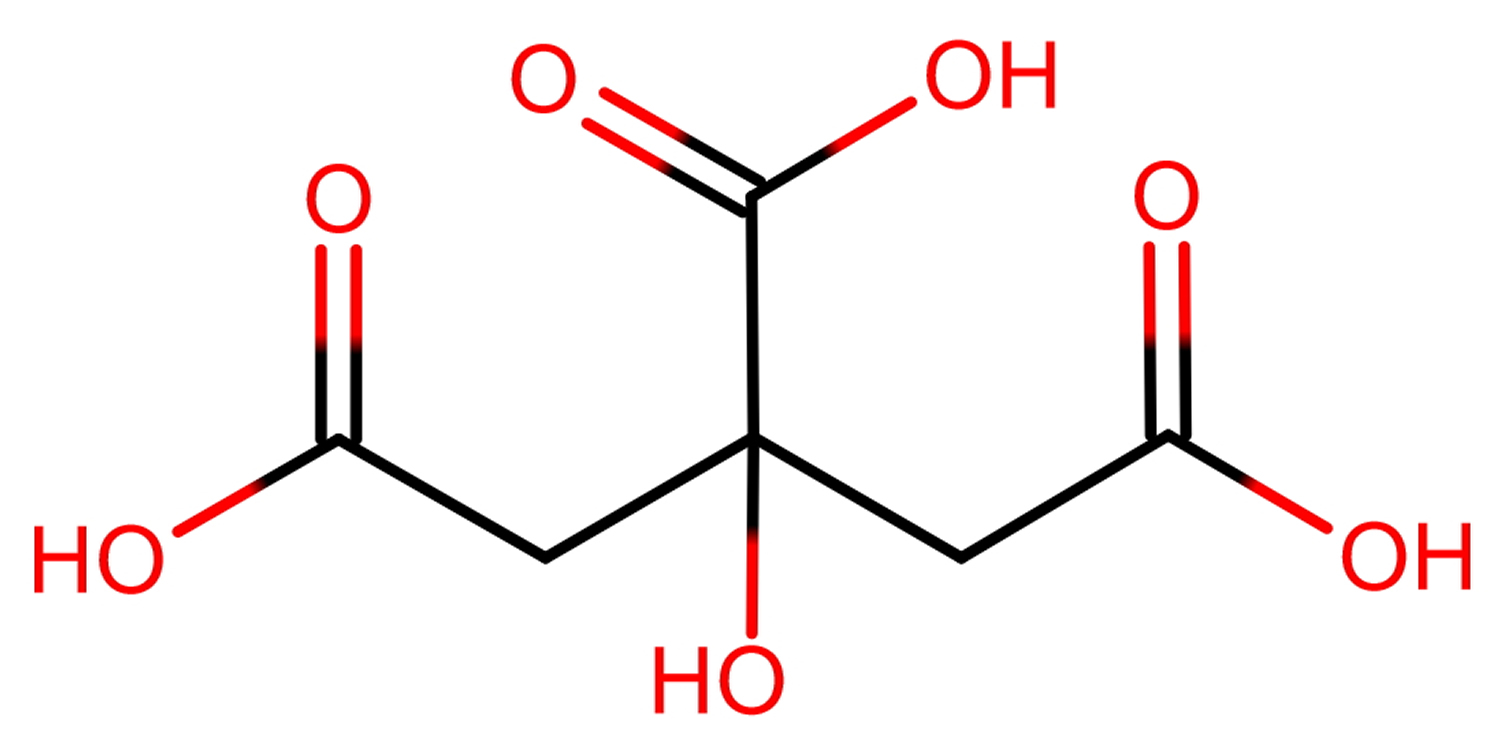
Citric acid molar mass is 192.027 g/mol (anhydrous) and 210.14 g/mol (monohydrate) 4.
Figure 1. Citric acid formula (C6H8O7)
[Source 5]Is citric acid bad for you?
No. Citric acid occurs in all living organisms as an intermediate substance in oxidative metabolism in the tricarboxylic acid or Krebs cycle. A major source of citric acid in your body results from endogenous metabolism in the mitochondria via the production of ATP in the citric acid cycle (see Citric acid cycle below). Citric acid is a natural component of fruit, vegetables and plants (roots and leaves). Citric acid is found in significant quantities in lemon and lime juices (41 and 39 mg/kg for ready-to-consume juice and 31 and 30 mg/kg for concentrates, respectively). Levels of citric acid in commercial lemon juice-baseddrinks, such as lemonade, fall in the range of 0.62 to 0.96 mg/kg. Other dietary sources of citric acid include grapefruits and oranges (significant levels), berries and beans.
Citric acid (E330, anhydrous and monohydrate) is approved as a food additive 6 for general use in foodstuffs following the quantum satis principle (limits set for some food products, i.e. juices, infantfoods). It has a long history of use as an additive in food 7, cosmetics 8, pharmaceuticals (human and veterinary), plant protection products, biocides 9 and household cleaning products.
Citric acid has been assessed by the Joint FAO/WHO (Food and Agriculture Organization/World Health Organization) Experts Committee on Food Additives 8. The Committee allocated an acceptable daily intake (ADI) of ‘not limited’ for citric acid and its calcium, potassium and sodium salts. This position was retained by the Scientific Committee on Food (SCF) 10.
Furthermore, both the European Food Safety Authority (EFSA) 11, 12, 13 and U.S. Food and Drug Administration (FDA) has granted citric acid GRAS status (Generally Recognized as Safe) as an antioxidant, emulsifier, stabilizer and thickener in infant formula 14 and as an emulsifier in combination with lauramide ethyl ester in food in general, including meat and poultry 15.
Citric acid in food
Table 1. Citric Acid Content, in Descending Order, of Various Fruit Juices and Commercially-Available Juice Formulations (Grams per Liter)
| Product | Type of product | Total citric acid Mean |
| Lemon juice | fresh, from fruit | 48 |
| Lime juice | fresh, from fruit | 45.8 |
| Lemon juice, Concord Foods | juice concentrate | 39.2 |
| Lime juice, ReaLime 100% | juice concentrate | 35.4 |
| Lemon juice, ReaLemon 100% | juice concentrate | 34.1 |
| Grapefruit juice, Florida’s Ruby Red | ready-to-consume | 25 |
| Orange juice, Tropicana Pure Premium | ready-to-consume | 16.9 |
| Orange juice, Tropicana Light ‘n Healthy | ready-to-consume | 16.7 |
| Orange juice | fresh, from fruit | 9.1 |
| Limeade/limonada, Minute Maid | ready-to-consume | 7.3 |
| Lemonade, Newman’s Own | ready-to-consume | 6.7 |
| Lemonade, Florida’s Natural | ready-to-consume | 6.2 |
| Lemonade, Minute Maid Light | ready-to-consume | 5.2 |
| Raspberry lemonade, Minute Maid | ready-to-consume | 5 |
| Lemonade, Tropicana | ready-to-consume | 4.83 |
| Pink lemonade, Minute Maid | ready-to-consume | 4.8 |
| Lemonade, Tropicana Sugar-Free | ready-to-consume | 4.6 |
| Lemonade, Minute Maid | ready-to-consume | 4.4 |
| Lemonade mix, Crystal Light | drink mix | 4.2 |
| Pink lemonade mix, Crystal Light | drink mix | 3.4 |
| Raspberry lemonade mix, Crystal Light | drink mix | 3.1 |
| Lemonade mix, Kool-Aid Sugarfree | drink mix | 2.1 |
| Lemonade mix, Country Time | drink mix | 1.6 |
| Crystallized lemon, True Lemon | dry mix | 0.92 |
Citric acid uses
Functional uses of citric acid as synergist for antioxidants, sequestrant, acidity regulator and flavoring agent 17. Citric acid is a preservative that more effective than acetic and lactic acids at inhibiting the growth of thermophilic bacteria 18. Citric acid is widely used in carbonated drinks and as an acidifier (reduce pH) of foods. It is less effective at controlling the growth of yeasts and mold than the other acids.
Citric acid (anhydrous and monohydrate) is approved as a food additive for use as a preservative in a wide range of commonly consumed foods and is authorized as a preservative in feed for all animal species without restrictions 19. “Citric acid is a normal constituent of the diet of humans and animals and, when ingested, is rapidly and completely metabolised to carbon dioxide and water; therefore, the use of citric acid in animal nutrition would not pose a risk to the environment”19.
Citric acid is used as an excipient in pharmaceutical preparations due to its antioxidant properties. Citrate salts of various metals are used to deliver minerals in biologically-available forms; examples include dietary supplements and medications. It maintains stability of active ingredients and is used as a preservative. Citric acid is also used as an acidulant to control pH.
Gastrointestinal absorption of citric acid from dietary sources has been associated with a modest increase in urinary citrate excretion 20. The salts of citric acid (citrates) or anhydrous citric acid can be used as anticoagulants due to their calcium chelating ability in blood and as calcium kidney stones dissolution agent. The mechanism of action of anhydrous citric acid in calcium kidney stones dissolution agent is its acidifying activity and calcium chelating activity. Furthermore, knowledge of the citric acid content of beverages may be useful in nutrition therapy for calcium urolithiasis (formation of calcium kidney stones), especially among patients with hypocitraturia 16. Citrate is a naturally-occurring inhibitor of urinary crystallization; achieving therapeutic urinary citrate concentration is one clinical target in the medical management of calcium urolithiasis. When provided as fluids, beverages containing citric acid add to the total volume of urine, reducing its saturation of calcium and other crystals, and may enhance urinary citrate excretion.
Urinary citrate is a potent, naturally-occurring inhibitor of urinary crystallization 16. Citrate is freely filtered in the proximal tubule of the kidney. Approximately 10% to 35% of urinary citrate is excreted; the remainder is absorbed in various ways, depending on urine pH and other intra-renal factors. Citrate is the most abundant organic ion found in urine. Hypocitraturia, defined as <320 mg (1.67 mmol) urinary citrate/day 21, is a major risk factor for calcium urolithiasis. The activity of citrate is thought to be related to its concentration in urine, where it exhibits a dual action, opposing crystal formation by both thermodynamic and kinetic mechanisms. Citrate retards stone formation by inhibiting the calcium oxalate nucleation process and the growth of both calcium oxalate and calcium phosphate stones, largely by its ability to bind with urinary calcium and reduce the free calcium concentration, thereby reducing the supersaturation of urine. Citrate binds to the calcium oxalate crystal surface, inhibiting crystal growth and aggregation 22. There is also evidence that citrate blocks the adhesion of calcium oxalate monohydrate crystals to renal epithelial cells 23. Medical interventions to increase urinary citrate are a primary focus in the medical management of urolithiasis 24. The amount of diet-derived citrate that may escape in body conversion to bicarbonate is reportedly minor 25. Nonetheless, a prior study reported increased urinary citrate after 1 week on 4 ounces lemon juice per day, diluted in 2 L water, in stone formers with hypocitraturia 20. Two retrospective studies showed an effect in calcium stone formers of lemon juice and/or lemonade consumption on urinary citrate 26, but a recent clinical trial showed no influence of lemonade on urinary citrate 27.
Studies indicated that citrate decreases lipid peroxidation and downregulates inflammation by reducing polymorphonuclear cell degranulation and attenuating the release of myeloperoxidase, elastase, interleukin (IL)-1β, and platelet factor 4 28. In test tube study, citrate improved endothelial function by reducing the inflammatory markers and decreasing neutrophil diapedesis in hyperglycemia 29. Moreover, citric acid has been shown to reduce hepatocellular injury evoked in rats by carbon tetrachloride 30. Citric acid might thus prove of value in decreasing oxidative stress. Another animal study in mice 1 suggests an antioxidant and anti-inflammatory effect for orally given citric acid at 1–2 g/kg in brain tissue, but this protective effect is lost when the dose is increased to 4 g/kg. Citric acid also demonstrated a beneficial hepatic protective effect at this dose range. Given that both increased brain oxidative stress and chronic inflammation have been linked to the development of neurodegenerative diseases, citric acid might thus prove of clinical benefit in such conditions.
Citric acid cycle
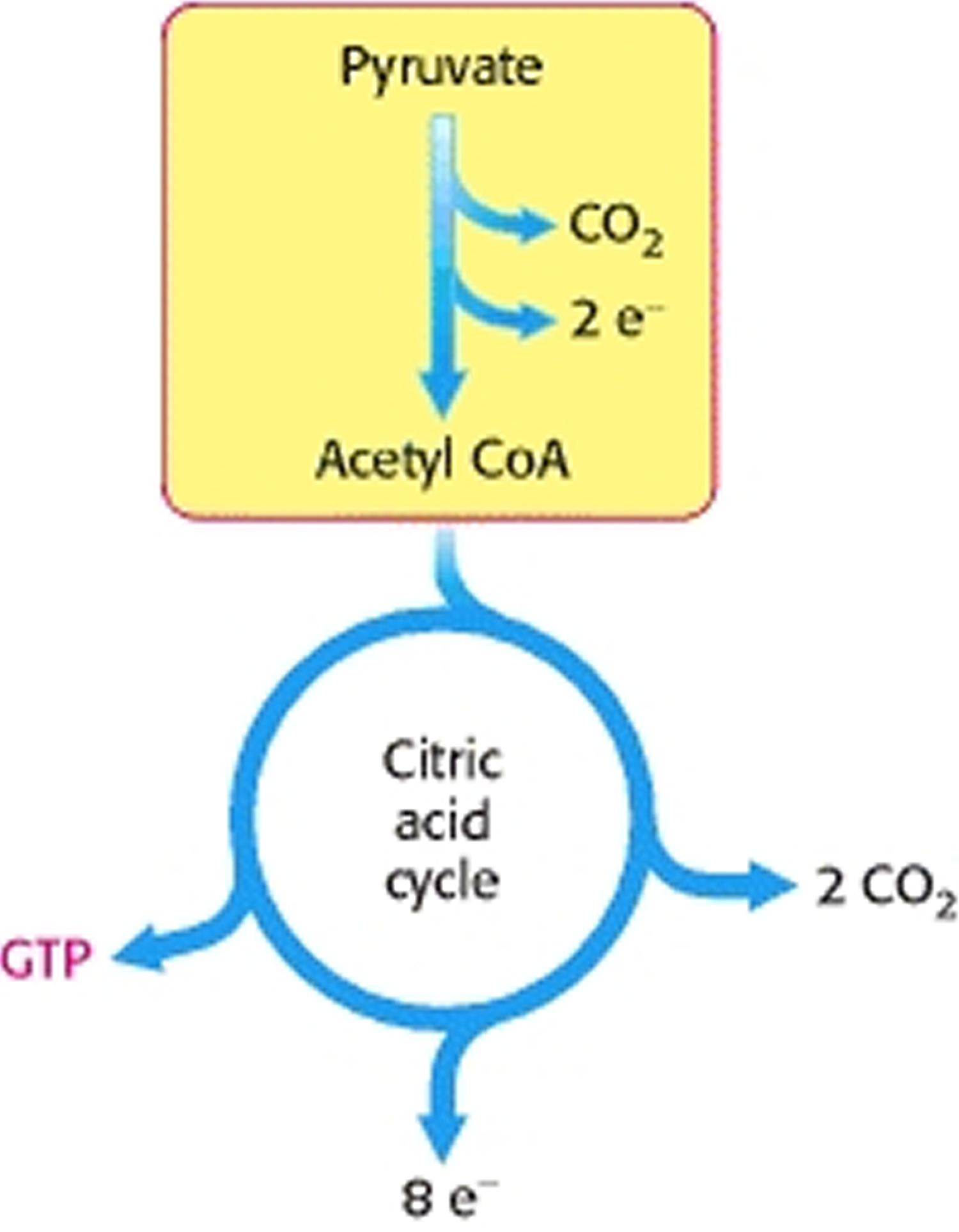
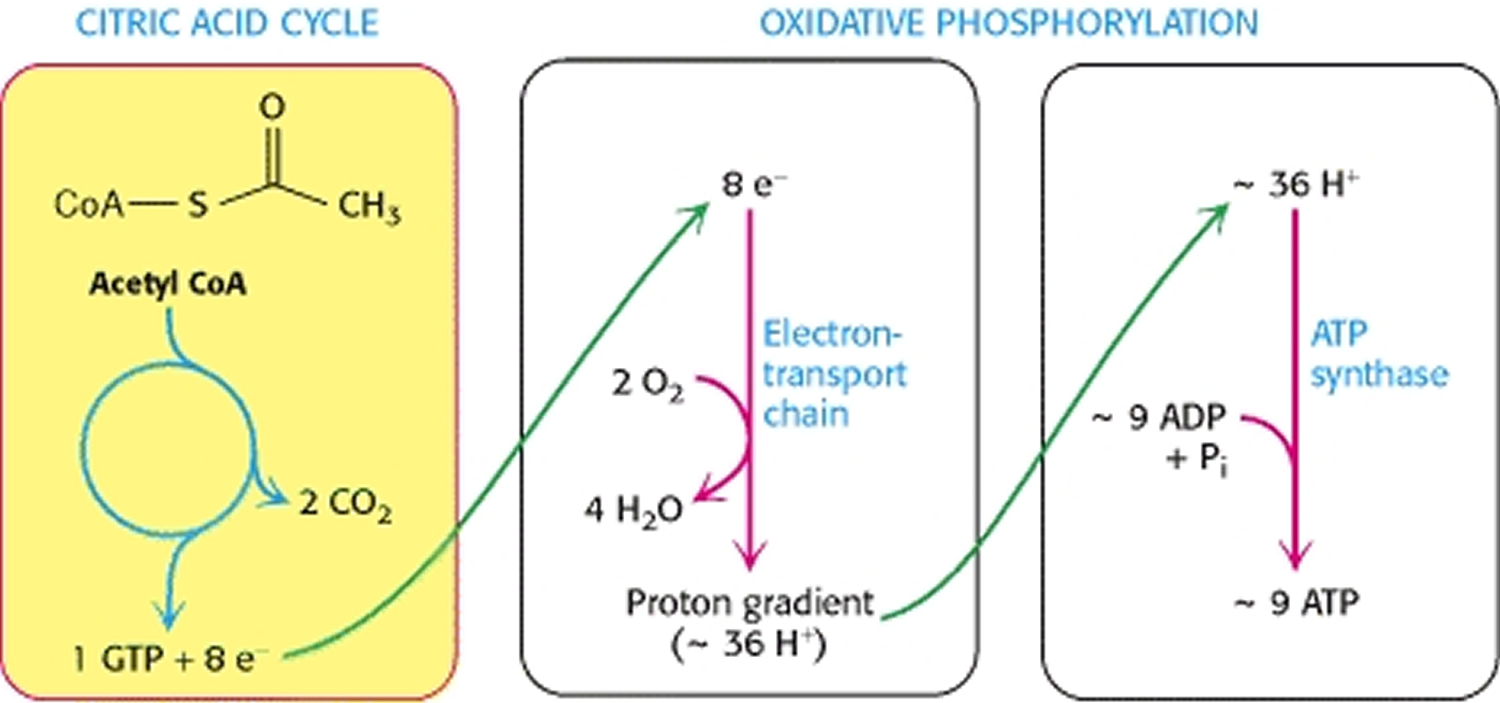
The citric acid cycle is the central metabolic hub of the cell, oxidizing carbon fuels, usually in the form of acetyl coenzyme A (acetyl CoA), as well as serving as a source of precursors for biosynthesis 31. The main function of the citric acid cycle is to transform fuel molecules into ATP. The citric acid cycle, in conjunction with oxidative phosphorylation, provides the vast majority of energy used by aerobic cells—in human beings, greater than 95%. The function of the citric acid cycle is the harvesting of high-energy electrons from carbon fuels. Note that the citric acid cycle itself neither generates a large amount of ATP nor includes oxygen as a reactant (Figure 5). Instead, the citric acid cycle removes electrons from acetyl CoA and uses these electrons to form NADH and FADH2. In oxidative phosphorylation, electrons released in the reoxidation of NADH and FADH2 flow through a series of membrane proteins (referred to as the electron-transport chain) to generate a proton gradient across the membrane. These protons then flow through ATP synthase to generate ATP from ADP and inorganic phosphate. Oxygen is required for the citric acid cycle indirectly inasmuch as it is the electron acceptor at the end of the electron-transport chain, necessary to regenerate NAD+ and FAD.
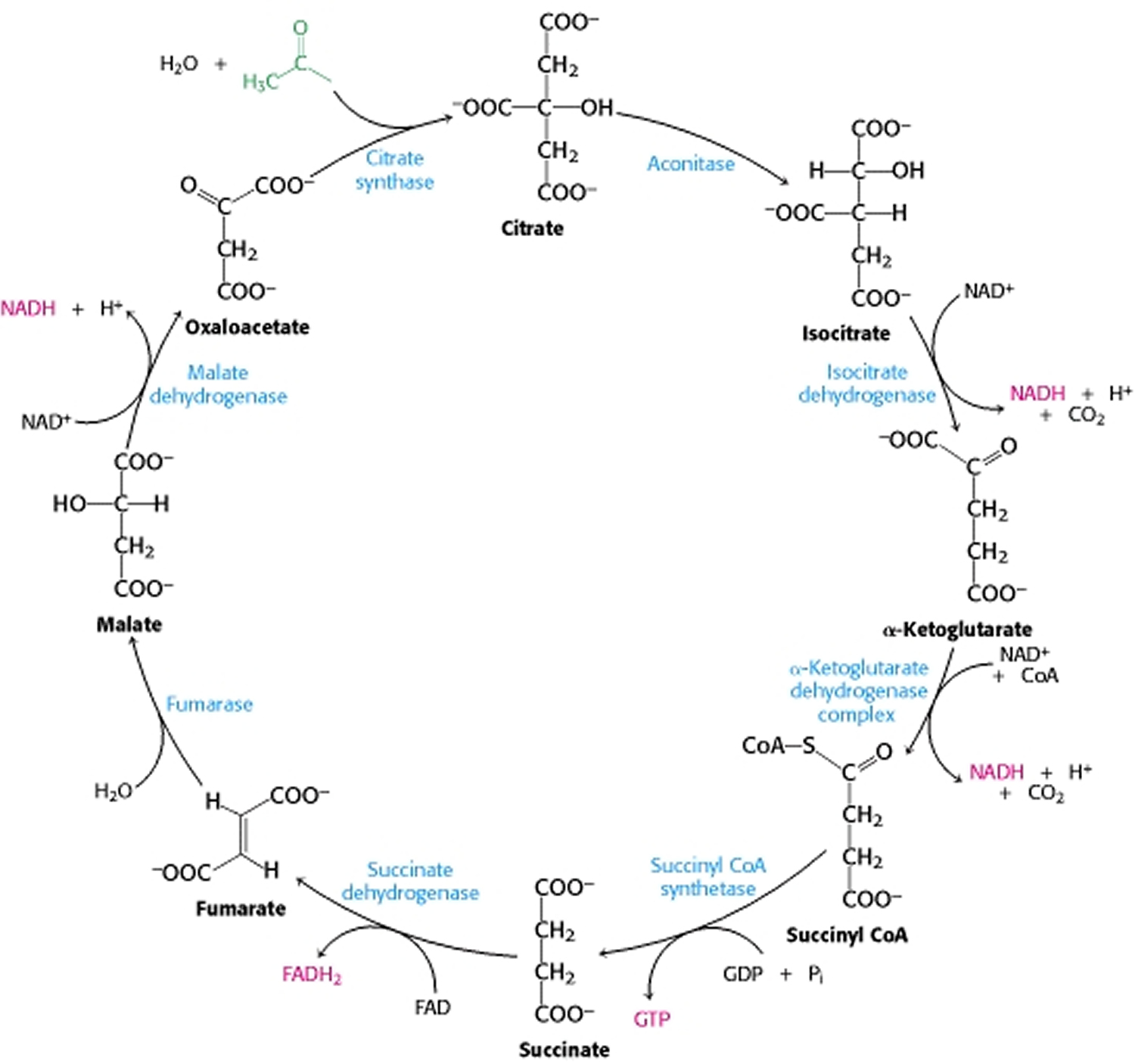
Citric acid cycle is highly efficient because a limited number of molecules can generate large amounts of NADH and FADH2. Note in Figure 4 that the four-carbon molecule, oxaloacetate, that initiates the first step in the citric acid cycle is regenerated at the end of one passage through the cycle. The oxaloacetate acts catalytically: it participates in the oxidation of the acetyl group but is itself regenerated. Thus, one molecule of oxaloacetate is capable of participating in the oxidation of many acetyl molecules.
The overall pattern of the citric acid cycle is shown in Figure 4. A four- carbon compound (oxaloacetate) condenses with a two-carbon acetyl unit to yield a six-carbon tricarboxylic acid (citrate). An isomer of citrate is then oxidatively decarboxylated. The resulting five-carbon compound (α-ketoglutarate) also is oxidatively decarboxylated to yield a four-carbon compound (succinate). Oxaloacetate is then regenerated from succinate. Two carbon atoms enter the cycle as an acetyl unit and two carbon atoms leave the cycle in the form of two molecules of carbon dioxide. Three hydride ions (hence, six electrons) are transferred to three molecules of nicotinamide adenine dinucleotide (NAD+), whereas one pair of hydrogen atoms (hence, two electrons) is transferred to one molecule of flavin adenine dinucleotide (FAD).
The citric acid cycle is also an important source of precursors, not only for the storage forms of fuels, but also for the building blocks of many other molecules such as amino acids, nucleotide bases, cholesterol, and porphyrin (the organic component of heme). Recall that fuel molecules are carbon compounds that are capable of being oxidized—of losing electrons. The citric acid cycle includes a series of oxidation-reduction reactions that result in the oxidation of an acetyl group to two molecules of carbon dioxide. Citric acid cycle is the gateway to the aerobic metabolism of any molecule that can be transformed into an acetyl group or dicarboxylic acid.
The Link between Glycolysis and the Citric Acid Cycle
In human body cells, glycolysis (the breakdown of glucose) takes place in the cytoplasm of a cell. This pathway can be thought of as comprising three stages (Figure 2). Stage 1, which is the conversion of glucose into fructose 1,6-bisphosphate, consists of three steps: a phosphorylation, an isomerization, and a second phosphorylation reaction. The strategy of these initial steps in glycolysis is to trap the glucose in the cell and form a compound that can be readily cleaved into phosphorylated three-carbon units. Stage 2 is the cleavage of the fructose 1,6-bisphosphate into two three-carbon fragments. These resulting three-carbon units are readily interconvertible. In stage 3, ATP is harvested when the three-carbon fragments are oxidized to pyruvate.
From Figure 2, we know that glucose can be metabolized to pyruvate anaerobically to synthesize ATP through the glycolytic pathway. Glycolysis, however, harvests but a fraction of the ATP available from glucose. We now begin an exploration of the aerobic processing of glucose, which is the source of most of the ATP generated in metabolism.
The aerobic processing of glucose starts with the complete oxidation of glucose derivatives to carbon dioxide. This oxidation takes place in the citric acid cycle, a series of reactions also known as the tricarboxylic acid cycle or the Krebs cycle. The citric acid cycle is the final common pathway for the oxidation of fuel molecules—amino acids, fatty acids, and carbohydrates. Most fuel molecules enter the cycle as acetyl coenzyme A (acetyl CoA).
Under aerobic conditions, the pyruvate generated from glucose is oxidatively decarboxylated to form acetyl coenzyme A (acetyl CoA). In human cells, the reactions of the citric acid cycle take place inside mitochondria.
Figure 2. Glycolysis stages
Note: The glycolytic pathway can be divided into three stages: (1) glucose is trapped and destabilized; (2) two interconvertible three-carbon molecules are generated by cleavage of six-carbon fructose; and (3) ATP is generated.
[Source 32]The Formation of Acetyl Coenzyme A from Pyruvate
Pyruvate produced by glycolysis is converted into acetyl coenzyme A (acetyl CoA), the fuel of the citric acid cycle. Acetyl Coenzyme A (Acetyl CoA) is the fuel for the citric acid cycle. This important molecule is formed from the breakdown of glycogen (the storage form of glucose), fats, and many amino acids. The formation of acetyl coenzyme A (acetyl CoA) from carbohydrates is less direct than from fat. Carbohydrates, most notably glucose, are processed by glycolysis into pyruvate. Under anaerobic conditions, the pyruvate is converted into lactic acid or ethanol, depending on the organism. Under aerobic conditions, the pyruvate is transported into mitochondria in exchange for OH- by the pyruvate carrier, an antiporter. In the mitochondrial matrix, pyruvate is oxidatively decarboxylated by the pyruvate dehydrogenase complex to form acetyl coenzyme A (acetyl CoA).
This irreversible reaction is the link between glycolysis and the citric acid cycle (Figure 3). Note that, in the preparation of the glucose derivative pyruvate for the citric acid cycle, an oxidative decarboxylation takes place and high-transfer-potential electrons in the form of NADH are captured. Thus, the pyruvate dehydrogenase reaction has many of the key features of the reactions of the citric acid cycle itself.
Figure 3. The link between glycolysis and the citric acid cycle
Figure 4. Citric Acid Cycle
The citric acid cycle oxidizes two-carbon units, producing two molecules of CO2, one molecule of Guanosine-5′-triphosphate (GTP), and high-energy electrons in the form of NADH and FADH2.
Figure 5. Cellular Respiration
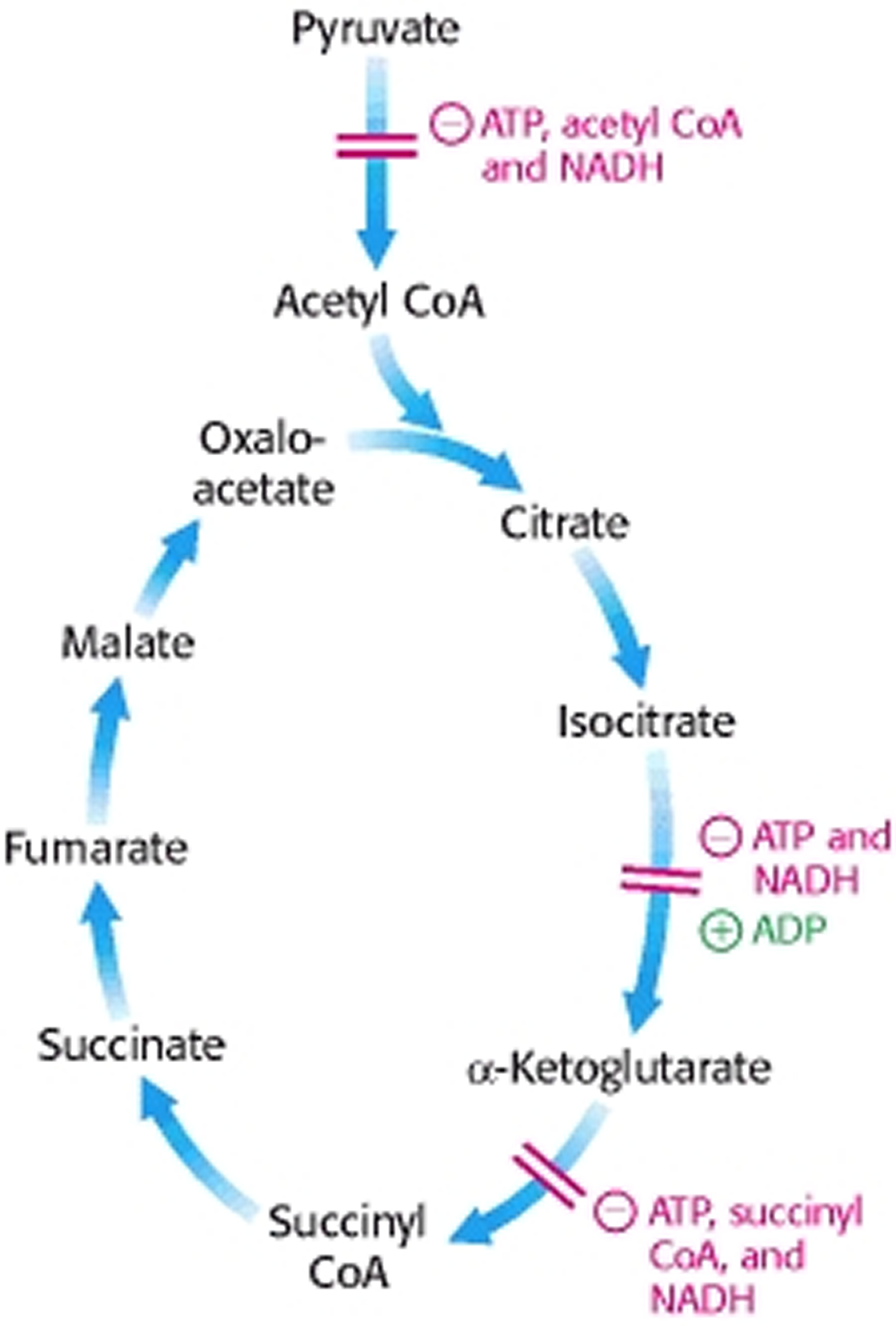
Control of the Citric Acid Cycle
The citric acid cycle is regulated primarily by the concentration of ATP and NADH. The key control points are the enzymes isocitrate dehydrogenase and α-ketoglutarate dehydrogenase.
Isocitrate dehydrogenase is allosterically stimulated by ADP, which enhances the enzyme’s affinity for substrates. The binding of isocitrate, NAD+, Mg2+, and ADP is mutually cooperative. In contrast, NADH inhibits iso-citrate dehydrogenase by directly displacing NAD+. ATP, too, is inhibitory. It is important to note that several steps in the cycle require NAD+ or FAD, which are abundant only when the energy charge is low.
A second control site in the citric acid cycle is α-ketoglutarate dehydrogenase. Some aspects of this enzyme’s control are like those of the pyruvate dehydrogenase complex, as might be expected from the homology of the two enzymes. α-Ketoglutarate dehydrogenase is inhibited by succinyl CoA and NADH, the products of the reaction that it catalyzes. In addition, α-ketoglutarate dehydrogenase is inhibited by a high energy charge. Thus, the rate of the cycle is reduced when the cell has a high level of ATP.
Figure 6. Control of the Citric Acid Cycle
- Abdel-Salam OME, Youness ER, Mohammed NA, Morsy SMY, Omara EA, Sleem AA. Citric Acid Effects on Brain and Liver Oxidative Stress in Lipopolysaccharide-Treated Mice. Journal of Medicinal Food. 2014;17(5):588-598. doi:10.1089/jmf.2013.0065. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4026104/[↩][↩]
- Penniston KL, Nakada SY, Holmes RP, et al. : Quantitative assessment of citric acid in lemon juice, lime juice, and commercially-available fruit juice products. J Endourol 2008;22:567–570 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2637791/[↩]
- Grigor JMV, Johnson WS, Salminen S: Food additives for special dietary purposes. In: Food Additives, 2nd edition. (Branen AL, editor; , Davidson PM, editor; , Salminen S, editor; , Thorngate JH 3rd, editor. , eds.) Marcel Dekker, Inc., Basel, New York, 2002, pp. 341[↩]
- Citric acid.https://pubchem.ncbi.nlm.nih.gov/compound/citric_acid[↩]
- https://chem.nlm.nih.gov/chemidplus/structure/viewer/77-92-9[↩]
- Hill T, Bateman H, Aldrich J, Quigley J and Schlotterbeck R, 2013. Evaluation of ad libitum acidified milk replacer programs for dairy calves. Journal of Dairy Science, 96, 3153–3162[↩]
- House W A and Van Campen D, 1971. Magnesium metabolism of sheep fed different levels of potassium and citric acid. Journal of Nutrition, 101, 1483–1492.[↩]
- JECFA (Joint FAO/WHO Expert Committee on Food Additives), 1974. Toxicological evaluation of some food additives including anticaking agents, antimicrobials, antioxidants, emulsifiers, and thickening agents. WHO Food Additives Series, 5[↩][↩]
- Kil D, Kwon W and Kim B, 2011, Dietary acidifiers in weanling pig diets: a review. Revista Colombiana de Ciencias Pecuarias, 24, 231–247[↩]
- EC (European Commission), 1991. Food science and techniques. First series of food additives of various technological functions. Reports of the Scientific Committee for Food (SCF), 25th series https://ec.europa.eu/info/departments/health-and-food-safety_en[↩]
- http://www.efsa.europa.eu/en/search/site/citric%20acid[↩]
- https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2015.4009[↩]
- https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2015.4010[↩]
- https://www.accessdata.fda.gov/scripts/fdcc/?set=GRASNotices&id=511&sort=GRN_No&order=DESC&startrow=1&type=basic&search=citric%20acid[↩]
- https://www.accessdata.fda.gov/scripts/fdcc/?set=GRASNotices&id=222&sort=GRN_No&order=DESC&startrow=1&type=basic&search=citric%20acid[↩]
- PENNISTON KL, NAKADA SY, HOLMES RP, ASSIMOS DG. Quantitative Assessment of Citric Acid in Lemon Juice, Lime Juice, and Commercially-Available Fruit Juice Products. Journal of endourology / Endourological Society. 2008;22(3):567-570. doi:10.1089/end.2007.0304. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2637791/[↩][↩][↩]
- Citric acid. http://www.fao.org/docrep/w6355e/w6355e0c.htm[↩]
- Preservatives. http://www.fao.org/3/a-au117e.pdf[↩]
- Scientific Opinion on the safety and efficacy of citric acid when used as a technological additive (acidity regulator) for all animal species. EFSA Journal 2015;13(2):4010 https://efsa.onlinelibrary.wiley.com/doi/pdf/10.2903/j.efsa.2015.4010[↩][↩]
- Seltzer MA, Low RK, McDonald M, et al. Dietary manipulation with lemonade to treat hypocitraturic calcium nephrolithiasis. J Urol. 1996;156:907. https://www.ncbi.nlm.nih.gov/pubmed/8709360[↩][↩]
- Pak CYC. Citrate and renal calculi: An update. Miner Electrolyte Metab. 1994;20:371 https://www.ncbi.nlm.nih.gov/pubmed/7783699[↩]
- Ryall RL. Urinary inhibitors of calcium oxalate crystallization and their potential role in stone formation. World J Urol. 1997;15:155 https://www.ncbi.nlm.nih.gov/pubmed/9228722[↩]
- Sheng X, Jung T, Wesson JA, et al. Adhesion at calcium oxalate crystal surfaces and the effect of urinary constituents. Proc Natl Acad Sci USA. 2005;102:267. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC544292/[↩]
- Heilberg IP. Update on dietary recommendations and medical treatment of renal stone disease. Nephrol Dial Transplant. 2000;15:117 https://www.ncbi.nlm.nih.gov/pubmed/10607782[↩]
- Meschi T, Maggiore U, Fiaccadori E, et al. The effect of fruits and vegetables on urinary stone risk factors. Kidney Int. 2004;66:2402. https://www.ncbi.nlm.nih.gov/pubmed/15569332[↩]
- Kang D, Haleblian GE, Sur RL, et al. Long-term lemonade based dietary manipulation in patients with hypocitraturic nephrolithia-sis. J Urol. 2007;177:1358 https://www.ncbi.nlm.nih.gov/pubmed/17382731[↩]
- Odvina CV. Comparative value of orange juice versus lemonade in reducing stone-forming risk. Clin J Am Soc Nephrol. 2006;1:1269 http://cjasn.asnjournals.org/content/1/6/1269.long[↩]
- Tiranathanagul K, Jearnsujitwimol O, Susantitaphong P, et al. : Regional citrate anticoagulation reduces polymorphonuclear cell degranulation in critically ill patients treated with continuous venovenous hemofiltration. Ther Apher Dial 2011;15:556–564 https://www.ncbi.nlm.nih.gov/pubmed/22107692[↩]
- Bryland A, Wieslander A, Carlsson O, et al. : Citrate treatment reduces endothelial death and inflammation under hyperglycaemic conditions. Diab Vasc Dis Res 2012;9:42–51 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3267553/[↩]
- Abdel Salam OME, Sleem AA, Shaffie NM: Hepatoprotective effects of citric acid and aspartame on carbon tetrachloride-induced hepatic damage in rats. EXCLI J 2009;8:41–49[↩]
- Berg JM, Tymoczko JL, Stryer L. Biochemistry. 5th edition. New York: W H Freeman; 2002. Chapter 17, The Citric Acid Cycle. Available from: https://www.ncbi.nlm.nih.gov/books/NBK21163/[↩]
- Berg JM, Tymoczko JL, Stryer L. Biochemistry. 5th edition. New York: W H Freeman; 2002. Section 16.1, Glycolysis Is an Energy-Conversion Pathway in Many Organisms. Available from: https://www.ncbi.nlm.nih.gov/books/NBK22593/[↩]