Contents
What is sodium metabisulfite
Sodium metabisulfite (Na2S2O5) also known as E 223, sulfur dioxide (E 220), sodium sulfite (E 221), sodium bisulfite (E 222), potassium metabisulfite (E 224), calcium sulfite (E 226), calcium bisulfite (E 227) and potassium bisulfite (E 228) are authorised are allowed as food additives to maintain appearance, increase shelf-life, and prevent the growth of hazardous microorganisms (e.g., fungi, bacteria) in the U.S and the European Union 1, 2, 3. Sulfites also known as sulphites are considered as Generally Recognized as Safe (GRAS) by the U.S. Food and Drug Administration (FDA) since 1959 1, 4. Sulfites are added to foods for a variety of purposes including the control of enzymatic and non-enzymatic browning (e.g. potatoes), the prevention of undesirable bacterial growth (e.g. corn wet milling and wine making), the conditioning of doughs (e.g. some frozen dough products), the prevention of oxidation, and the bleaching of selected products (e.g. maraschino cherries and hominy). Sulfites may be present in baked goods, condiments, shrimp, certain wines (white wine usually more than red wine), beer and fruit juices 5.
Sulfites (SO32–) are naturally found in some foods and are also commonly used food preservatives that reduce spoilage, act as antioxidants, and prevent fruit and vegetable browning 3, 5. Several forms of sulfites exist and are allowed for use in foods. The sulfites used by the food industry are sulfur dioxide (E 220), sodium sulfite (E 221), sodium bisulfite (E 222), sodium metabisulfite (E 223), potassium metabisulfite (E 224), calcium sulfite (E 226), calcium bisulfite (E 227) and potassium bisulfite (E 228) 3. All of these food additives serve similar functions in foods because all of them have similar chemistries in foods dependent upon pH.
Sulfites can also occur naturally in foods, especially dried fruits and fermented foods, as the result of sulfite formation by yeast. The residual levels of sulfites in foods range from less than 10 parts per million (ppm) in many food products to greater than 2000 parts per million (ppm) in certain dried fruits 6. Naturally occurring levels of sulfites are typically quite low, less than 10 ppm with a few exceptions 6.
Whilst the apparent safety of the sulfite additives lead to their widespread use, reports began to emerge during the 1970s that sulfite exposure was associated with adverse reactions 7, 8. These included the triggering of anaphylactic reactions, as well as the elicitation of a wide range of symptoms, including dermatitis, urticaria, flushing, hypotension, abdominal pain and diarrhea, although the vast majority of reports described the triggering of bronchconstriction in asthmatic patients 9, 10. Sulfite-induced asthmatic symptoms range from mild in some individuals, to very severe in others, and in some individuals these reactions can be life threatening 11.
In 1986, following the identification of numerous cases of sulfite-induced asthma occurring on eating of green or fruit salads treated with sulfites, the FDA banned the use of sulfites on most fresh fruits, vegetables, and in salad bars intended to be served raw or presented fresh to the public 12, 13, 14, 4. The only exception is sulfite use on minimally processed potatoes sliced or shredded for frying where sulfite use is still permitted, although FDA has a long-standing, though never finalized, proposal to ban that use also 13. Sulfite use as a fungicide during the shipment of fresh table grapes is regulated by the U.S. Environmental Protection Agency (EPA), but the concentration of sulfur dioxide (SO2) residues on the table grapes as consumed must be less than 10 ppm total sulfur dioxide (SO2) 15, 13.
Asthma exacerbations, anaphylaxis, and urticaria have all been reported from reactions to sulfites, and the FDA requires labeling on any food or beverage with greater than 10 parts per million of these preservatives 5. Some common names include sulfur dioxide, potassium bisulfite, potassium metabisulfite, sodium bisulfite, sodium metabisulfite, or sodium sulfite. There is currently no reliable blood or allergy skin test to test for sulfite reactions 5.
Note that sulfa drugs, antibiotics and other medicines that contain a sulfonamide molecule, are not the same thing as sulfites 5.
Sulfates are salts of sulfuric acid and are present in many medicines, supplements, and personal care products and they are not the same thing as sulfites or sulfa drugs 5.
Sulfur (sulphur) is an element that is essential for life, and is found in sulfites, sulfates, and sulfonamides, but by itself is not responsible for the reactions people have to these other molecules 5. Elemental sulfur (sulphur) can rarely cause problems if inhaled.
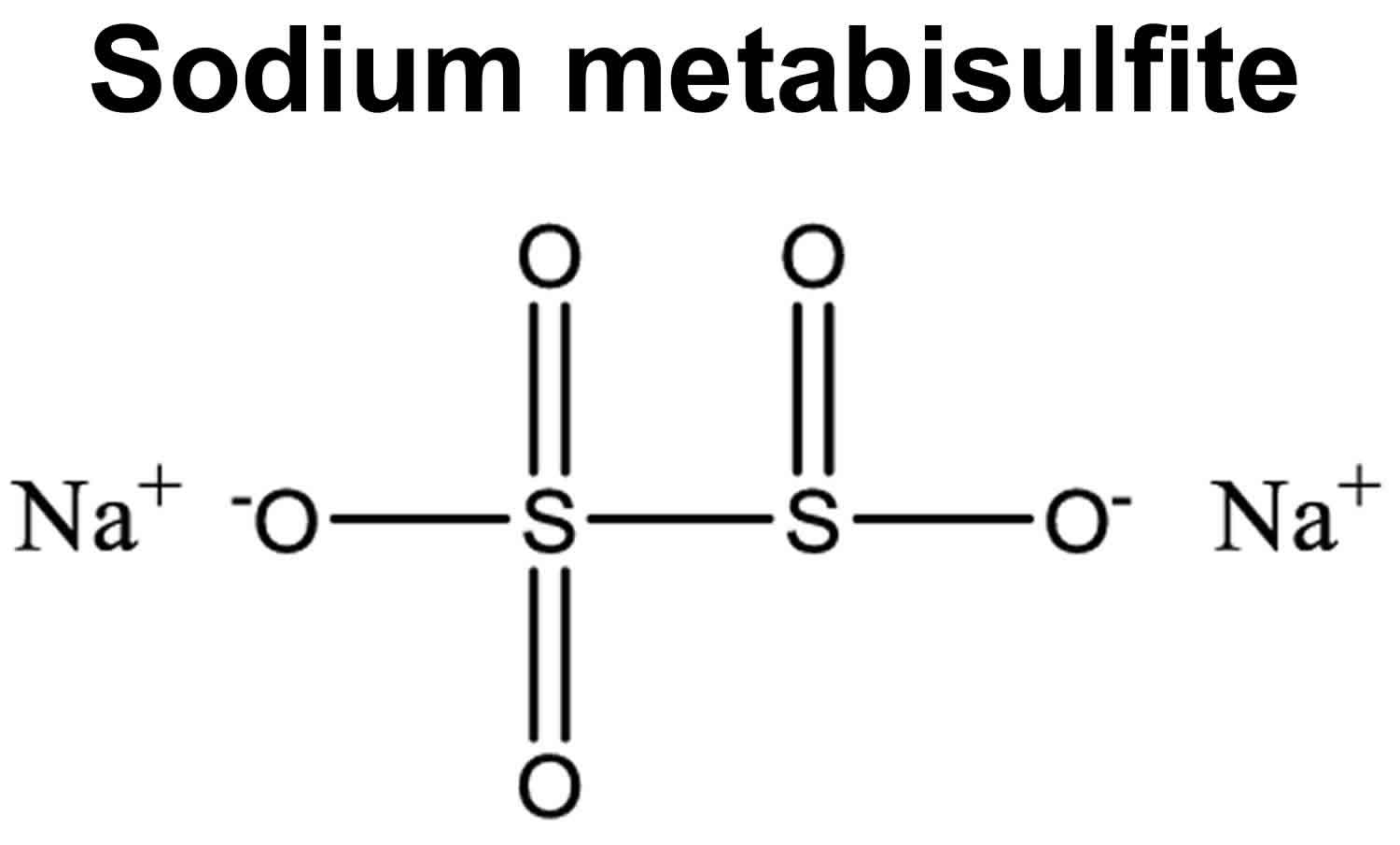
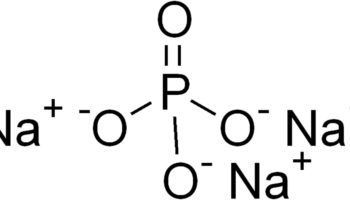
Figure 1. Sodium metabisulfite chemical structure
[Source 2 ]Table 1. Sulfites
| Food additive | Number | Chemical formula | Molecular weight g/mol | CAS | EINECS |
|---|---|---|---|---|---|
| Sulfur dioxide | E 220 | SO2 | 64.06 | 7446‐09‐5 | 231‐195‐2 |
| Sodium sulfite | E 221 | Na2SO3 for anhydrous and | 126.04 | 7757‐83‐7 | 231‐821‐4 |
| Na2SO3·7H2O for heptahydrate | 252.16 | 10102‐15‐5 | – (a) | ||
| Sodium bisulfite | E 222 | NaHSO3 | 104.06 | 7631‐90‐5 | 231‐548‐0 (b) |
| Sodium metabisulfite | E 223 | Na2S2O5 | 190.11 | 7681‐57‐4 | 231‐673‐0 |
| Potassium metabisulfite | E 224 | K2S2O5 | 222.33 | 16731‐55‐8 | 240‐795‐3 |
| Calcium sulfite | E 226 | CaSO3 | 120.14 | 10257‐55‐3 | 233‐596‐8 (c) |
| Calcium bisulfite | E 227 | Ca(HSO3)2 | 202.22 | 13780‐03‐5 | 237‐423‐7 |
| Potassium bisulfite | E 228 | KHSO3 | 120.17 | 7773‐03‐7 | 231‐870‐1 |
Footnotes:
(a) Not registered in the European Commission Inventory.
(b) The EINECS 231‐921‐4 included in the Commission Regulation (EU) No 231/2012 for E 222 is not registered in the European Commission Inventory.
(c) The EINECS included in the Commission Regulation (EU) No 231/2012 for E 226 is of another substance (calcium dibenzoate 218‐235‐4).
Abbreviations: CAS = Chemical Abstract Service; EINECS = European Inventory of Existing Commercial chemical Substances
[Source 3 ]Sodium metabisulfite in food
Sulfite preservatives are commonly known as sodium metabisulfite (E 223), sulfur dioxide (E 220), sodium sulfite (E 221), sodium bisulfite (E 222), potassium metabisulfite (E 224), calcium sulfite (E 226), calcium bisulfite (E 227) and potassium bisulfite (E 228) 1, 2, 3. Sulfites can be used to preserve flavor and color within food, inhibit bacterial growth, stop fresh food from spoiling, and help preserve medication and drinks. Sulfites are most often found in wine, dried fruit, and dried vegetables. Sometimes they are used in sausages and salads. They can also occur naturally in low concentrations. In many countries it is illegal to add sulfites to foods like fresh salads or fruit salads, or to meats like minced meat or sausage meat. The addition of sulfites to beer and wine is permitted in most countries.
Foods containing sulfites include dried fruits, dried vegetables, pickled onions and bottled soft drinks and cordials 16, 10. The addition of sulfite additives to beer and wine is permitted in most countries. The use of sulfites in fresh salads, fruit salads, mince meat or sausage meat, is illegal in many countries, although it may be added illegally.
Total sulfur dioxide (SO2) levels in foods and beverages should be determined by the optimized Monier-Williams distillation-titration procedure, a method approved by the Association of Official Analytical Chemists (AOAC). Alternate methods do exist but FDA and other federal agencies will typically use the Association of Official Analytical Chemists (AOAC) method so any alternative method must provide similar results.
Food products that contain undeclared sulfites at levels above 10 ppm total sulfur dioxide (SO2) will be subject to potential recall actions. In the U.S., three recall categories are used 17:
- Class 1 recalls are the most serious and involve situations where there is a that consumer reasonably could have ingested ≥10 mg of undeclared sulfites on a single occasion that could potentially cause a serious adverse reaction in a susceptible person or deaths. FDA is aware of deaths occurring among sulfite-sensitive asthmatics.
- Class 2 recalls include situations where exposure to sulfites may cause temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is rare.
- Class 3 recalls includes situations where exposure to sulfites is not likely to cause adverse health consequences.
For sulfites, recalls can fall into any of the 3 recall classifications. Using data from controlled oral clinical challenges of sulfite-sensitive asthmatics, the FDA has established dose levels that are associated with Class 1, Class 2 or Class 3 recalls. Analysis is done for total sulfur dioxide (SO2) based upon the Association of Official Analytical Chemists (AOAC) procedure. The analytical result is given in terms of a concentration (ppm). FDA determines a worst case dose (mg SO2 equivalents) by using the 95th percentile level of consumption of the food in question. Dose estimates are based upon single occasion exposures. The estimated 95th percentile sulfite ingestion dose (in mg) for a single occasion is then obtained by multiplying the sulfite concentration of the food product (in ppm or mg per kg of food) by the 95th percentile level of food consumption (in kg) for the specific food.
Table 2. Major types of food that may contain sulfite additives
| Common sources | |
|---|---|
| Drinks | Bottled soft drinks and fruit juices, cordials, cider, beer, wine (including sparkling wine), instant tea. |
| Other liquids | Commercial preparations of lemon and lime juice, vinegar, grape juice |
| Commercial foods | Dried potatoes, gravies, sauces, fruit toppings, maraschino cherries, pickled onions, sauerkraut, pickles, maple syrup, jams, jellies, biscuits, bread, pies and pizza dough. |
| Fruit | Dried apricots, fruit bars and sometimes grapes will be transported with sachets of the sulfite containing preservative. Dried sultanas do not normally contain sulfites. |
| Salads | Restaurant may add sulfites to preserve the colour of salads and fruit salads. |
| Crustaceans | Sulfur powder may be added on top of crustaceans to stop them discolouring. |
| Meat | Sulfites are sometimes added illegally to mincemeat or sausage meat. |
| Other foods | Gelatin*, coconut. |
Footnote: * Gelatin is used in some medications, and trace sulfite residues from gelatin in medications may cause adverse reactions. Allergic reactions to gelatin can also occur in people with mammalian meat allergy
[Source 18 ]Table 3. Food products recall categories
| Class | Est. 95th Percentile Dose |
|---|---|
| Class 1 | ≥10 mg sulfur dioxide (SO2) |
| Class 2 | 3.7 – 9.9 mg sulfur dioxide (SO2) |
| Class 3 | < 3.7 mg sulfur dioxide (SO2) |
How can the presence of sulfites be recognized on labelled foods?
The presence of sulfites must be indicated on the label by code numbers E220 to E228, or the word sulfite, as shown in the following table.
Table 4. Sulfites on labelled foods
| Food additive | Number |
|---|---|
| Sulfur dioxide | E 220 |
| Sodium sulfite | E 221 |
| Sodium bisulfite | E 222 |
| Sodium metabisulfite | E 223 |
| Potassium metabisulfite | E 224 |
| Calcium sulfite | E 226 |
| Calcium bisulfite | E 227 |
| Potassium bisulfite | E 228 |
Are low or no sulfite wines and beers available?
Some wine makers and brewers produce wines and beers which state that they do not add sulfites. However, there are many technical reasons related to wine making and brewing, which may mean that very low levels of sulfites are still present, even when not deliberately added.
Sulfites are generally found at higher levels in cask wine than bottled wine, and are at much higher concentrations in white wine than red wine, which is preserved by natural tannins.
Sodium metabisulfite uses
Sulfites also known as sulphites are widely used as preservative and antioxidant additives in the food and pharmaceutical industries 19. Sulfites have been used since Roman times to preserve food flavor and color, inhibit bacterial growth, reduce food spoilage, and help preserve medications. Burning sulphur was used by the ancient Greeks to fumigate houses, and by the ancient Romans to sanitize wine vessels 20. The sulphite additives are now used widely in the food industry – predominantly as anti-browning agents, antioxidants and preservatives 21, 22. Sulfites (sulphites) are also used extensively in the pharmaceutical industry and have a number of industrial uses including in the photographic and textile industries, and consequently, occupational exposures to these additives may also occur 23, 24, 25.
In addition to food, exposure to sulfites can occur through the use of cosmetics and medicines (Table 5 and 6). Cosmetics containing sulfites include hair colors and bleaches, creams, and perfumes 26. Medicines containing sulfites include eye drops, topical medications, and parenteral medications such as adrenaline, phenylephrine, corticosteroids and local anaesthetics 25, 27.
For the majority of people, exposure to sulfites occurs as a result of consumption of foods and drinks to which sulfites have been added, primarily for the purpose of preservation (see Tables 1 and 2). In addition to their preservative activity, sulphites are used to prevent the browning of foods, as bleaching agents, as dough conditioning agents, to prevent excess alkalinity of foods, as food processing aids, color stabilizers and antioxidants 21, 22. Therefore, in addition to being cheap and convenient, sulfites are extremely versatile, and their addition to many foods serves more than one purpose.
Asthma exacerbations, anaphylaxis, and urticaria have all been reported from reactions to sulfites, and the U.S. Food and Drug Administration (FDA) requires that the presence of sulfites be declared on food labels when used as an ingredient in the food and also when used as a processing aid or when present in an ingredient used in the food (e.g. dried fruit pieces). Sulfites must be declared in these cases when the concentration in the food is greater than 10 parts per million total sulfur dioxide (SO2) 28:
- (a)(4) “For the purposes of paragraph (a)(3) of this section, any sulfiting agent (sulfur dioxide, sodium sulfite, sodium bisulfite, potassium bisulfite, sodium metabisulfite, and potassium metabisulfite) that has been added to any food or to any ingredient in any food and that has no technical effect in that food will be considered to be present in an insignificant amount only if no detectable amount of the agent is present in the finished food. A detectable amount of sulfiting agent is 10 parts per million or more of the sulfite in the finished food.” If naturally occurring sulfites also exists in foods, it would contribute to the analytical result.
What this means is if the food contains ≥ 10 ppm total sulfur dioxide (SO2), then sulfite must be declared on the label. This will most typically occur when sulfite is deliberately added to the food. The specific name of the additive (e.g. sodium bisulfite) must be declared in the case of use as an intentional ingredient.
Sulfites are also prohibited from certain uses in the U.S. 13. Sulfites may not be used in products such as meats that serve as a good source of vitamin B1 because sulfites can scavenge that vitamin from foods. In 1986, following the identification of numerous cases of sulfite-induced asthma occurring on ingestion of green or fruit salads treated with sulfites, FDA prohibited the use of sulfites on fruits and vegetables intended to be served raw or presented fresh to the public 12. The only exception is sulfite use on minimally processed potatoes sliced or shredded for frying where sulfite use is still permitted, although FDA has a long-standing, though never finalized, proposal to ban that use also 13. Sulfite use as a fungicide during the shipment of fresh table grapes is regulated by the U.S. Environmental Protection Agency (EPA), but the concentration of sulfur dioxide (SO2) residues on the table grapes as consumed must be less than 10 ppm total sulfur dioxide (SO2) 13.
The labeling of the presence of sulfites on alcoholic beverages is under the jurisdiction of the Tax and Trade Bureau (TTB) of the U.S. Dept. of Treasury. While most ingredients in alcoholic beverages are not declared on the container, the presence of sulfites must be declared. Sulfites are commonly used in wine fermentation to control undesirable growth of acid-producing bacteria while allowing alcohol-producing yeast to proliferate.
Table 5. Medical and cosmetic uses of sulfites
| Cosmetics | Medications |
|---|---|
| Hair colors and bleaches, home permanent solutions, skin fading/lighteners, false tan lotions, anti-ageing creams and moisturisers, facial cleansers, around-eye creams, body washes/cleansers, hair sprays, perfumes, blush, bronzers/highlighters | Topical anti-fungal and corticosteroid creams and ointments (e.g. Trimovate®, Timodine®, Aureocort®, Aureomycin®, Nizoral®, Nystatin®, Lustra®, Psoradrate®), adrenaline, isoprenaline, isoproterenol, isoetharine, phenylephrine, dexamethasone and injectable corticosteroids, dopamine, local anaesthetics, propofol, aminoglycoside antibiotics, metoclopramide, doxycycline and vitamin B complex |
What types of medications contain sulfites?
Sulfites are used as preservatives in some medications, as shown in the following table.
Table 6. Sulfites in medications
| Administration method | Medications |
|---|---|
| Topical medications | Topical anti-fungal and corticosteroid creams and ointments (e.g. Trimovate®, Timodine®, Aureocort®, Aureomycin®, Nizoral®, Nystatin®, Lustra®, Psoradrate®), adrenaline, isoprenaline, isoproterenol, isoetharine, phenylephrine, dexamethasone and injectable corticosteroids, dopamine, local anaesthetics, propofol, aminoglycoside antibiotics, metoclopramide, doxycycline and vitamin B complex |
| Oral medications | Some oral medications contain gelatin, and trace sulfite residues from gelatin in medications may cause adverse reactions. Allergic reactions to gelatin can also occur in people with mammalian meat allergy. |
| Injected medications | The most common sources of sulfites from injected medications are:
In people with sulfite sensitivity, the benefit of adrenaline to treat anaphylaxis is considered to outweigh any theoretical risk from sulfites in an emergency. |
Sodium metabisulfite allergy
Despite sulfites widespread use in the food industry and as medicinal ingredients, exposure to sulfites has been shown to induce bronchoconstriction in asthmatic patients and a wide range of adverse reactions in sensitive individuals 29, 30, 31. Asthmatic symptoms were shown to be induced by exposure to sulfite in orange drinks in a patient-based study 32. Also, exposure to sulfite through various routes has been linked to a range of adverse clinical effects in sensitive individuals, ranging from dermatitis to life-threatening anaphylactic and asthmatic reactions 30. Recent work by a group of scientists suggested that adverse reactions of (bi)sulfite could be driven by a peroxidase-catalyzed radical pathway 33. Myeloperoxidase (MPO), which is predominantly expressed by neutrophils (a type of white blood cell), can potentially accelerate a reaction by acting as a catalyst the (bi)sulfite-derived formation of protein radicals 14.
For most patients, sulfite sensitivity is reported to be a non-IgE mediated sensitivity, resulting in mast cell degranulation 34, 35. Boxer, et al 36 reported evidence of IgE antibody by passive skin transfer, but no evidence of IgE antibody by ELISA. The authors concluded that the IgE antibody was not specifically related to the presence of a metabisulfite determinant 36.
Yang, et al 37 performed skin prick and intradermal skin testing with potassium metabisulfite (K₂S₂O₅, 1 mg/ml) on 18 subjects, followed single-blind oral provocative challenge tests with placebo (lactose) and with 1, 5, 10, 25, and 50 mg of potassium metabisulfite (K₂S₂O₅). Of 18 patients, five patients had positive skin tests. Single-blind oral provocative challenge testing was positive in three. Passive transfer was attempted in two subjects with positive skin tests by use of unheated serum. The test was positive in both (one subject with asthma and one subject with anaphylaxis). The immediate onset of these reactions, the positive skin tests, the positive provocative oral challenge tests, and positive passive transfer tests suggests that at least some of these reactions are IgE mediated 37.
Currently, there are no published standardized or validated blood or skin allergy test for sulfite reactions 38. The diagnosis of sulfite sensitivity can be confirmed by a “challenge” in which sulfite is administered in solutions or capsules of increasing concentration with 20 to 30 minutes between steps 38. The protocol most useful is published in Up-to-Date, by Simon 39. This protocol does provide an option for skin testing prior to oral challenge.
It is important to note that a negative skin test does not rule out, nor does it confirm sulfite sensitivity. Controlled oral challenge is the only reliable way to diagnose sulfite allergy 38.
How is suspected sulfite sensitivity diagnosed?
There is currently no reliable blood or skin allergy test for sulfite reactions 18. A food challenge under supervision of a clinical immunology or allergy specialist may confirm or exclude sensitivity 18.
How is suspected sulfite sensitivity treated?
- Avoidance: Reduce sulfite exposure where possible.
- Optimize control of asthma: People with asthma, including those with sulfite sensitivity should:
- Use asthma medications as advised by their doctor.
- Be referred to a clinical immunology or allergy specialist if they have a severe reaction.
Is sensitivity to sulfites the same as sulfonamide antibiotic allergy?
No. Allergic reactions to sulfonamide antibiotics are very different to sulfite sensitivity. Sulfa drugs, antibiotics and other medicines that contain a sulfonamide molecule, are not the same thing as sulfites 5. A person that is allergic to one sulfonamide antibiotic, is at risk of reacting to other sulfonamide antibiotics but not sulfites. There is no relationship between sulfite sensitivity and sulfonamide antibiotic allergy.
Sulfonamide antibiotics that are available on prescription include 40:
- Sulfamethoxazole that is used in combination with trimethoprim.
- Less commonly used sulfonamide antibiotics such as sulfadiazine (tablets, injections or creams), sulfadoxine (for malaria), and sulfacetamide antibiotic eye drops.
- Sulfasalazine (Salazopyrin, Pyralin), that is used in inflammatory bowel disease or arthritis, and is a combination sulfapyridine (a sulfonamide antibiotic) and a salicylate.
If a person has had an allergic reaction to Bactrim, Resprim or Septrin, there is no way of knowing whether the allergy was to sulfamethoxazole or to trimethoprim. Therefore trimethoprim (Alprim, Triprim) and sulfonamide antibiotics should both be avoided.
Sometimes people who have had an allergic reaction to a sulfonamide antibiotic are labelled as “sulfur allergic” or allergic to sulfur, sulphur or sulfa. This wording should not be used since it is unclear and can cause confusion 40. Some people wrongly assume that they will be allergic to non-antibiotic sulfonamides or to other sulfur containing medicines or sulfite preservatives.
Sulfur is an element which occurs throughout the human body as a building block of life, and it is not possible to be allergic to sulfur itself 40. Allergic reactions to sulfonamide antibiotics do not increase the likelihood of allergy to sulfur powder, sulfite preservatives, sulfates (in medicines, or soaps and shampoos) or non-antibiotic sulfonamide medicines like some pain killers or fluid tablets 40.
There is no blood test available for allergy to sulfonamide antibiotics, and skin testing has not been validated. Skin testing has been used to check for trimethoprim allergy (to distinguish from sulfamethoxazole allergy in people who have reacted to Bactrim) but results need to be interpreted with caution. Challenge testing may be carried out under supervision of a clinical immunology and allergy specialist.
People who have had an allergic reaction to one sulfonamide antibiotic are usually advised to avoid all sulfonamide antibiotics. As these antibiotics are not normally used in an emergency, wearing medical identification is not routinely recommended although it may be advisable in people who have had anaphylaxis.
Sulfonamide antibiotic allergy is not always lifelong and cross-reaction does not always occur. Desensitisation to switch off allergy temporarily, is available if a sulfonamide antibiotic is the only suitable drug to use.
Do people who react to sulfites need to avoid sulfates or sulfur?
No. Sulfates are found in some injectable drugs as sulfate compounds, such as heparin sulfate, dextran sulfate, morphine sulfate. The sulfates in soaps and shampoos such as sodium lauryl sulfate are strong detergents and can irritate the skin or eyes. However sulfate are not usually allergenic and do not cause reactions in sulfite-sensitive people. It is usually safe to use a sulfate when a person has a sulfonamide allergy or a sulfite intolerance. Elemental sulfur powder which is used in gardening may cause difficulty breathing if inhaled but is not usually a specific problem for sulfite-sensitive people 40.
Sodium metabisulfite side effects
Sulfites can cause adverse reactions which are like allergy but do not involve the immune system and are therefore called intolerances. Many individuals are sensitive to sulfite additives and may experience a range of symptoms, including dermatological, gastrointestinal and respiratory symptoms. The most common sulfite sensitivity reactions are asthma symptoms (in people with underlying asthma) and rhinitis (hay fever-like) reactions. Occasionally urticaria (hives) may occur, and very rarely, anaphylaxis (severe allergic reaction). Topical, oral or parenteral exposure to sulfites has been reported to induce a range of adverse clinical effects in sensitive individuals, ranging from dermatitis, urticaria, flushing, hypotension, abdominal pain and diarrhea to life-threatening anaphylactic and asthmatic reactions 30. It is important to note that a number of individuals experience an array of symptoms following exposure to sulfites; thus, skin, intestinal and respiratory reactions may occur simultaneously, and in various combinations and severities.
The true prevalence of asthmatic responses to the sulfites remains uncertain, although it is generally agreed that between 3 and 10% of adult asthmatics may exhibit adverse reactions to the sulfite additives, with a number of these individuals experiencing life-threatening reactions 19.
In addition to triggering episodic and acute symptoms, sulfite additives play a role in the chronic symptoms experienced by some individuals. Sensitive individuals who regularly use cosmetics or topical medications containing sulfites have been reported to exhibit chronic skin symptoms, especially on the hands, perineum and face 19. Similarly, occupational exposures to the sulfites have been reported to cause persistent skin symptoms 19. Although the possibility that exposure to sulfites may contribute to chronic asthma has not been widely explored, it is possible that unrecognized regular exposure to the sulfite additives may contribute to the chronic asthma symptoms experienced by some sensitive individuals 19.
Asthma symptoms are the most common adverse reactions caused by sulfites 18:
- Wheezing, chest tightness and coughing are estimated to affect 5-10% of people with asthma.
- Symptoms are more likely when asthma is poorly controlled.
- Adverse reactions to sulfites can occasionally occur when there is no preceding history of asthma.
Sulfites can also cause allergy like reactions (intolerances), with symptoms such as wheezing in people with asthma, allergic rhinitis (hay fever), and urticaria (hives).
In very rare cases it is possible that sulfites may have caused anaphylaxis, the most severe type of allergic reaction. Symptoms include flushing, fast heartbeat, wheezing, hives, dizziness, stomach upset and diarrhoea, collapse, tingling or difficulty swallowing.
Many of these reactions when fully assessed have been found not to be anaphylaxis, or caused by triggers other than sulfites.
In the United States more than 250 cases of sulfite-related adverse reactions, including anaphylactic shock, asthmatic attacks, urticaria and angioedema, nausea, abdominal pain and diarrhea, seizures and death, have been reported, including 6 deaths allegedly associated with restaurant food containing sulfites 4. In Canada 10 sulfite-related adverse reactions have been documented, and 1 death suspected to be sulfite-related has occurred 4. The exact mechanism of sulfite-induced reactions is unknown 4.
Exposure to the sulfites arises mainly from the consumption of foods and drinks that contain these additives; however, exposure may also occur through the use of pharmaceutical products, as well as in occupational settings. While contact sensitivity to sulfites additives in topical medications is increasingly being recognized, skin reactions also occur after ingestion of or parenteral exposure to sulfites. Most studies report a 3-10% prevalence of sulfite sensitivity among asthmatic subjects following ingestion of these additives. However, the severity of these reactions varies, and steroid-dependent asthmatics, those with marked airway hyperresponsiveness, and children with chronic asthma, appear to be at greater risk. In addition to episodic and acute symptoms, sulfites may also contribute to chronic skin and respiratory symptoms.
In summary, sensitivity to the sulfite additives is a very real problem that significantly affects the health of many individuals, particularly asthmatics. The possibility of sulfite sensitivity should be considered when individuals demonstrate adverse reactions to a range of exposures, with no obvious pattern, particularly when these individuals experience a worsening of asthma symptoms following the consumption of foods such as dried fruits and wines, or adverse skin reactions following the use of cosmetics or medicated creams.
What are the mechanisms for adverse reactions to sulfites?
Mechanisms for sulfite adverse reactions can vary, and include:
- Reflex contraction of the airways caused by inhaling sulfur dioxide. This mechanism may explain the rapid onset of symptoms when drinking liquids like beer or wine, when sulfur dioxide is inhaled during the swallowing process.
- A partial deficiency of the enzyme sulfite oxidase (which helps to break down sulfur dioxide), in some people with asthma who react to sulfites.
- Other mechanisms yet to be fully understood.
Given the wide variations in symptoms, in the severity of reactions, and in the sensitivities of individuals to different forms of sulfite, it is unlikely that any single mechanism can explain all reactions to the sulfite additives.
A number of potential mechanisms that might explain asthmatic reactions to the sulfites have been postulated, although the mode of exposure is a confounding factor 9, 10. Nebulized bisulfite solutions, acidified metabisulfite solutions, encapsulated metabisulfite and sulfite containing food or drinks may or may not provoke reactions in the same individual, and the types of reactions and concentrations of sulfite that provoke reactions may vary widely with different forms of exposure. Inhalation of sulphur dioxide (SO2), generated from ingested sulfites in the warm acidic environments of the mouth and stomach, may cause respiratory symptoms. Although nebulized metabisulfite was also thought to cause bronchoconstriction through generation of sulphur dioxide (SO2) in the airways 41, airway responsiveness to acidic metabisulfite solutions and sulphur dioxide (SO2) were not significantly related 42.
Some studies have suggested that sulfites may stimulate the parasympathetic system, with bronchoconstriction being mediated by a cholinergic pathway 9. The enzyme sulfite oxidase oxidizes sulfite to sulphate, and it was suggested that inadequate activity of this enzyme may result in excessive accumulation of sulfite, resulting in cholinergic mediated bronchoconstriction in some individuals 43. The release of histamine and other mediators as a consequence of mast cell degranulation through IgE or non-IgE mediated mechanisms has also been suggested as a possible mechanism in some individuals 44. There is some evidence supporting a role for prostaglandins in sulfite induced asthma 45, and the inhibition of bronchoconstriction by leukotriene receptor antagonists, in asthmatic subjects exposed to sulphur dioxide (SO2), suggests a possible role for leukotrienes 46, 47.
Is sodium metabisulfite safe?
Sulfite food additives are commonly known as sodium metabisulfite (E 223), sulfur dioxide (E 220), sodium sulfite (E 221), sodium bisulfite (E 222), potassium metabisulfite (E 224), calcium sulfite (E 226), calcium bisulfite (E 227) and potassium bisulfite (E 228) are considered as Generally Recognized as Safe (GRAS) by the U.S. Food and Drug Administration (FDA) since 1959 1, 4, 2, 3.
Sulphur dioxide (SO2), as well as being one of the family of sulfite additives that is ‘ingested’ in foods, is also an air pollutant. One of the first recorded adverse reactions triggered by sulfites dates back to the eruption of Mt Vesuvius in 79 AD, when Pliny the Elder, whose airways were “constitutionally weak and narrow and often inflamed”, collapsed and died after inhaling the sulphurous gases emanating from the volcano 32. Whilst most non-asthmatic individuals can tolerate up to 5 ppm sulphur dioxide (SO2), there is evidence that a large number of asthmatics are hypersensitive to this gas 48, 49. It is not entirely clear why this may be the case, but it may be that in these individuals sulphur dioxide (SO2) irritates airways that are already ‘twitchy’. In contrast to hyperresponsiveness to sulphur dioxide (SO2) gas, sulfite sensitivity predominantly refers to the triggering of adverse symptoms following ingestion, or parenteral or topical exposure to these additives. While sensitivity to the sulfites can present in a number of ways, it is the triggering of adverse respiratory symptoms (predominantly amongst asthmatics) that seems to occur most frequently. It has been estimated that 3–10% of asthmatics experience such symptoms 9, 50, 51.
One of the earliest reports suggesting that ingestion of sulfites could cause irritation of the respiratory tract was published in 1973 7. Since then numerous case reports and reviews have been published on the phenomenon of respiratory hypersensitivity to ingested sulfites. The first case of anaphylaxis following ingestion of sodium metabisulfite in a restaurant salad was reported in 1976 8 and the following year sulphur dioxide (SO2) in orange drinks was reported to induce asthma 32. In the early 1980s there were numerous reports suggesting that ingestion of sulfites by susceptible individuals was the cause of severe adverse reactions. Although many of these were asthmatic responses 51, 52, 53, urticaria and angioedema 54, abdominal pain and diarrhea 55, as well as anaphylaxis 56, 57 were reported. In 1985, Yang and Purchase 4 reported that there had been more than 250 cases of sulfite-related adverse reactions, including six deaths, in the United States, while in Canada, 10 sulfite-related adverse reactions and one death, thought to be sulfite related, had been reported.
As a consequence of these reported adverse reactions, the US Food and Drug Administration (FDA) acted in 1986 to prohibit the use of sulfites on fruits and vegetables that were to be served raw or presented as fresh to the public 12. For foods and drinks in which the use of sulfite was permitted, sulfite concentrations >10 ppm had to be declared on the label 15. Despite the introduction of these regulations, there continued to be sporadic reports of serious adverse effects following unintended ingestion of sulfites. The potentially severe nature of sulfite sensitivities is highlighted by a number of reports of life-threatening reactions to sulfite additives 58, 59, 60.
In the early 1980s there were also a number of reports of asthma exacerbations and/or generalized skin reactions among asthmatic patients treated with bronchodilator medications containing sulfite 57, 61, 62, 63. One report highlighted the case of a patient who was hypersensitive to metabisulfite and developed anaphylaxis following ingestion of metabisulfite-treated food 64. This patient had a prolonged clinical course, requiring two visits to the emergency department and three weeks of corticosteroid therapy, suggesting that the relapse and delayed recovery may have been related to continued exposure to sulfites during treatment. Some older, rarely used bronchodilator solutions such as isoproterenol and isoetharine contain sulfites at concentrations sufficient to cause bronchoconstriction in most asthmatic patients, even in the absence of a history of sulfite sensitivity 65. With the availability of selective beta2-agonists such as albuterol that do not contain sulfites, these older bronchodilator solutions need not be used to treat asthmatic patients.
The presence of sulfites in some other pharmaceutical products is also reason for concern. There are published reports of anaphylactic or asthmatic reactions associated with the use of sulfite-containing local anaesthetics, as well as gentamicin, metoclopramide, doxycycline and vitamin B complex 27. The generic form of the anaesthetic agent, propofol, contains sodium metabisulfite and has the potential to cause adverse effects, particularly in the children 66. Treatment of anaphylaxis in patients who are sensitive to sulfite also poses a conundrum in that administration of adrenaline is regarded as the primary treatment for anaphylaxis, and yet all commercially available preparations of adrenaline contain metabisulfite 67. However, even in patients with serious sulfite sensitivity, the benefit from adrenaline is considered to outweigh the risk of sulfite exposure associated with use of adrenaline in an emergency 18.
Asthmatic responses have also been reported following exposure to sulfites in occupational settings. Valero et al. 68 reported the case of a patient who experienced episodes of bronchospasm that required hospitalization after handling sodium bisulfite at work. Metabisulfite-induced occupational asthma has also been reported in a photographic technician 24 and a radiographer 69. Occupational asthma has been reported in a worker who sprinkled dry metabisulfite powder onto potatoes 70 and three cases of occupational asthma related to metabisulfite exposure were reported in France 71. The use of sodium metabisulfite in the fish and prawn-processing industry, with associated exposures to high concentrations of sulphur dioxide (SO2), has been identified as an under-recognized cause of occupational airways disease 72. An increased incidence of asthma and increased asthma-related mortality have also been reported in sulfite pulp mill workers, probably as a consequence of repeated exposures to peak concentrations of sulphur dioxide (SO2) 73, 74.
Over the past three decades a number of challenge studies have been performed in an attempt to confirm sulfite sensitivity and estimate its prevalence in subjects with suggestive histories. The interpretation of these studies is difficult, as the criteria for the selection of subjects have varied and may have been biased towards those with a history of sensitivity or more severe asthma. In addition, the dose and physical form of sulfite used in challenge protocols has varied widely, as have the criteria considered indicative of a positive response 20, 9, 75, 76, 41. As a consequence there is some uncertainty as to the true prevalence of sulfite sensitivity amongst asthmatic patients, although the literature consistently reports a prevalence of between 3 and 10% 20, 9, 51, 76, 77. Steroid-dependent asthmatics and those with marked airway hyperresponsiveness appear to be at greater risk of adverse reactions to sulfite-containing foods 10. Although there was an early suggestion that as many as 30% of reported cases of sulfite sensitivity occur in individuals with no known history of asthma 78, later reviews of the literature suggested that adverse reactions to sulfites were extremely rare in non-asthmatic subjects 20, 10. There are some indications that respiratory sensitivity to sulfites may be more common amongst women 9, 79 and children 80, 81, 82.
Although the literature regarding the prevalence of skin reactions to the sulfites is somewhat limited, studies suggest that somewhere between 1 and 5% of those patch tested may demonstrate skin sensitivities to these additives 83, 84, 85.
Reports in the literature describe adverse dermatological responses following exposure to cosmetics, such as facial cosmetic creams 86, hair dyes 87 and false tanning lotion 85.
In addition, topical medications, such as antifungal 83 and hemorrhoid creams 88 and eye drops 89 have been associated with the elicitation of skin symptoms. Similarly, a wide range of occupational exposures have also been linked with adverse skin reactions to the sulfites 90, 23, 24, 91, 92, 93, 94.
- Food Additive Status List. https://www.fda.gov/food/food-additives-petitions/food-additive-status-list[↩][↩][↩][↩]
- EFSA ANS Panel (EFSA Panel on Food Additives and Nutrient Sources Added to Food), 2016. Scientific Opinion on the re-evaluation sulfur dioxide (E 220), sodium sulfite (E 221), sodium bisulfite (E 222), sodium metabisulfite (E 223), potassium metabisulfite (E 224), calcium sulfite (E 226), calcium bisulfite (E 227) and potassium bisulfite (E 228) as food additives. EFSA Journal 2016;14(4):4438 151 pp. doi:10.2903/j.efsa.2016.4438[↩][↩][↩][↩]
- EFSA Panel on Food Additives and Flavourings (FAF); Younes M, Aquilina G, Castle L, Engel KH, Fowler PJ, Frutos Fernandez MJ, Fürst P, Gundert-Remy U, Gürtler R, Husøy T, Manco M, Mennes W, Moldeus P, Passamonti S, Shah R, Waalkens-Berendsen I, Boon P, Cheyns K, Crebelli R, FitzGerald R, Lambré C, Mirat M, Ulbrich B, Vleminckx C, Mech A, Rincon AM, Tard A, Horvath Z, Wright M. Follow-up of the re-evaluation of sulfur dioxide (E 220), sodium sulfite (E 221), sodium bisulfite (E 222), sodium metabisulfite (E 223), potassium metabisulfite (E 224), calcium sulfite (E 226), calcium bisulfite (E 227) and potassium bisulfite (E 228). EFSA J. 2022 Nov 24;20(11):e07594. doi: 10.2903/j.efsa.2022.7594[↩][↩][↩][↩][↩][↩][↩]
- Yang WH, Purchase EC. Adverse reactions to sulfites. CMAJ. 1985 Nov 1;133(9):865-7, 880. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1346296/pdf/canmedaj00272-0034.pdf[↩][↩][↩][↩][↩][↩][↩]
- Sulfite and Sulfa Drugs. https://www.aaaai.org/tools-for-the-public/allergy,-asthma-immunology-glossary/sulfite-and-sulfa-drugs[↩][↩][↩][↩][↩][↩][↩][↩]
- Idiosyncratic illnesses. https://farrp.unl.edu/resources/gi-fas/idiosyncratic-illnesses[↩][↩]
- Kochen J. Sulfur dioxide, a respiratory tract irritant, even if ingested. Pediatrics. 1973 Jul;52(1):145-6.[↩][↩]
- Prenner BM, Stevens JJ. Anaphylaxis after ingestion of sodium bisulfite. Ann Allergy. 1976 Sep;37(3):180-2.[↩][↩]
- Gunnison AF, Jacobsen DW. Sulfite hypersensitivity. A critical review. CRC Crit Rev Toxicol. 1987;17(3):185-214. doi: 10.3109/10408448709071208[↩][↩][↩][↩][↩][↩][↩]
- Lester MR. Sulfite sensitivity: significance in human health. J Am Coll Nutr. 1995 Jun;14(3):229-32. doi: 10.1080/07315724.1995.10718500[↩][↩][↩][↩][↩]
- Jamieson DM, Guill MF, Wray BB, May JR. Metabisulfite sensitivity: case report and literature review. Ann Allergy. 1985 Feb;54(2):115-21.[↩]
- https://archives.federalregister.gov/issue_slice/1986/7/9/24971-25026.pdf#page=51[↩][↩][↩]
- Sulfites – USA. https://farrp.unl.edu/sulfites-usa[↩][↩][↩][↩][↩][↩]
- Kumar A, Triquigneaux M, Madenspacher J, Ranguelova K, Bang JJ, Fessler MB, Mason RP. Sulfite-induced protein radical formation in LPS aerosol-challenged mice: Implications for sulfite sensitivity in human lung disease. Redox Biol. 2018 May;15:327-334. doi: 10.1016/j.redox.2017.12.014[↩][↩]
- New sulfite regulations. FDA Drug Bull. 1986 Nov;16(2):17-8.[↩][↩]
- Grotheer G, Marshall M, Simonne A. Sulfites: Separating Fact from Fiction: Institute of Food and Agricultural Sciences. University of Florida; 2005.[↩]
- Timbo B, Koehler KM, Wolyniak C, Klontz KC. Sulfites–a food and drug administration review of recalls and reported adverse events. J Food Prot. 2004 Aug;67(8):1806-11. https://doi.org/10.4315/0362-028X-67.8.1806[↩][↩]
- Sulfite Sensitivity Frequently Asked Questions (FAQ). https://www.allergy.org.au/patients/other-allergy/sulfite-sensitivity-faq[↩][↩][↩][↩][↩][↩]
- Vally H, Misso NL. Adverse reactions to the sulphite additives. Gastroenterol Hepatol Bed Bench. 2012 Winter;5(1):16-23. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4017440[↩][↩][↩][↩][↩]
- Bush RK, Taylor SL, Busse W. A critical evaluation of clinical trials in reactions to sulfites. J Allergy Clin Immunol. 1986 Jul;78(1 Pt 2):191-202. doi: 10.1016/0091-6749(86)90012-6[↩][↩][↩][↩]
- Roberts A, McWeeny D. The use of sulfur dioxide in the food industry. A review. J Fd Technol. 1972;7:221–38.[↩][↩]
- Taylor SL, Higley NA, Bush RK. Sulfites in foods: uses, analytical methods, residues, fate, exposure assessment, metabolism, toxicity, and hypersensitivity. Adv Food Res. 1986;30:1-76. doi: 10.1016/s0065-2628(08)60347-x[↩][↩]
- Apetato M, Marques MS. Contact dermatitis caused by sodium metabisulphite. Contact Dermatitis. 1986 Mar;14(3):194. doi: 10.1111/j.1600-0536.1986.tb01217.x[↩][↩]
- Jacobs MC, Rycroft RJ. Contact dermatitis and asthma from sodium metabisulfite in a photographic technician. Contact Dermatitis. 1995 Jul;33(1):65-6. doi: 10.1111/j.1600-0536.1995.tb00463.x[↩][↩][↩]
- Challen RG. Sulphite content of Australian pharmaceutical products. Med J Aust. 1990 Feb 19;152(4):196-8. doi: 10.5694/j.1326-5377.1990.tb125149.x[↩][↩]
- Environmental Working Group. Skin Deep Cosmetic Safety Database. https://www.ewg.org/skindeep[↩]
- Smolinske SC. Review of parenteral sulfite reactions. J Toxicol Clin Toxicol. 1992;30(4):597-606. doi: 10.3109/15563659209017945[↩][↩]
- 21 CFR 101.100. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-101/subpart-G/section-101.100[↩]
- Rall DP. Review of the health effects of sulfur oxides. Environ Health Perspect. 1974 Aug;8:97-121. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1474939/pdf/envhper00498-0103.pdf[↩]
- Vally, H., Misso, N.L.A. and Madan, V. (2009), Clinical effects of sulphite additives. Clinical & Experimental Allergy, 39: 1643-1651. https://doi.org/10.1111/j.1365-2222.2009.03362.x[↩][↩][↩]
- Song A, Lin F, Li J, Liao Q, Liu E, Jiang X, Deng L. Bisulfite and sulfite as derivatives of sulfur dioxide alters biomechanical behaviors of airway smooth muscle cells in culture. Inhal Toxicol. 2014 Feb;26(3):166-74. doi: 10.3109/08958378.2013.872211[↩]
- Freedman BJ. Asthma induced by sulphur dioxide, benzoate and tartrazine contained in orange drinks. Clin Allergy. 1977 Sep;7(5):407-15. doi: 10.1111/j.1365-2222.1977.tb01471.x[↩][↩][↩]
- Ranguelova K, Rice AB, Lardinois OM, Triquigneaux M, Steinckwich N, Deterding LJ, Garantziotis S, Mason RP. Sulfite-mediated oxidation of myeloperoxidase to a free radical: immuno-spin trapping detection in human neutrophils. Free Radic Biol Med. 2013 Jul;60:98-106. doi: 10.1016/j.freeradbiomed.2013.01.022[↩]
- Bold J. Considerations for the diagnosis and management of sulphite sensitivity. Gastroenterol Hepatol Bed Bench. 2012 Winter;5(1):3-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4017445[↩]
- Collaco CR, Hochman DJ, Goldblum RM, Brooks EG. Effect of sodium sulfite on mast cell degranulation and oxidant stress. Ann Allergy Asthma Immunol. 2006 Apr;96(4):550-6. doi: 10.1016/S1081-1206(10)63549-1[↩]
- Boxer MB, Bush RK, Harris KE, Patterson R, Pruzansky JJ, Yang WH. The laboratory evaluation of IgE antibody to metabisulfites in patients skin test positive to metabisulfites. J Allergy Clin Immunol. 1988 Oct;82(4):622-6. doi: 10.1016/0091-6749(88)90974-8[↩][↩]
- Yang WH, Purchase EC, Rivington RN. Positive skin tests and Prausnitz-Küstner reactions in metabisulfite-sensitive subjects. J Allergy Clin Immunol. 1986 Sep;78(3 Pt 1):443-9. doi: 10.1016/0091-6749(86)90031-x. Erratum in: J Allergy Clin Immunol 1987 Jan;79(1):15.[↩][↩]
- Sulfite allergy evaluation. https://www.aaaai.org/allergist-resources/ask-the-expert/answers/2022/sulfite[↩][↩][↩]
- Testing and challenge procedures to evaluate allergic and asthmatic reactions to food additives. https://www.uptodate.com/contents/testing-and-challenge-procedures-to-evaluate-allergic-and-asthmatic-reactions-to-food-additives[↩]
- Sulfonamide Antibiotic Allergy. https://www.allergy.org.au/patients/drug-allergy/sulfonamide-antibiotic-allergy[↩][↩][↩][↩][↩]
- Wright W, Zhang YG, Salome CM, Woolcock AJ. Effect of inhaled preservatives on asthmatic subjects. I. Sodium metabisulfite. Am Rev Respir Dis. 1990 Jun;141(6):1400-4. doi: 10.1164/ajrccm/141.6.1400[↩][↩]
- Field PI, McClean M, Simmul R, Berend N. Comparison of sulphur dioxide and metabisulphite airway reactivity in subjects with asthma. Thorax. 1994 Mar;49(3):250-6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1021155/pdf/thorax00295-0070.pdf[↩]
- Añíbarro B, Caballero T, García-Ara C, Díaz-Pena JM, Ojeda JA. Asthma with sulfite intolerance in children: a blocking study with cyanocobalamin. J Allergy Clin Immunol. 1992 Jul;90(1):103-9. doi: 10.1016/s0091-6749(06)80016-3[↩]
- Dixon CM, Ind PW. Inhaled sodium metabisulphite induced bronchoconstriction: inhibition by nedocromil sodium and sodium cromoglycate. Br J Clin Pharmacol. 1990 Sep;30(3):371-6. doi: 10.1111/j.1365-2125.1990.tb03786.x[↩]
- Wang M, Wisniewski A, Pavord I, Knox A, Tattersfield A. Comparison of three inhaled non-steroidal anti-inflammatory drugs on the airway response to sodium metabisulphite and adenosine 5′-monophosphate challenge in asthma. Thorax. 1996 Aug;51(8):799-804. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC472543/pdf/thorax00327-0039.pdf[↩]
- Lazarus SC, Wong HH, Watts MJ, Boushey HA, Lavins BJ, Minkwitz MC. The leukotriene receptor antagonist zafirlukast inhibits sulfur dioxide-induced bronchoconstriction in patients with asthma. Am J Respir Crit Care Med. 1997 Dec;156(6):1725-30. doi: 10.1164/ajrccm.156.6.9608006[↩]
- Gong H Jr, Linn WS, Terrell SL, Anderson KR, Clark KW. Anti-inflammatory and lung function effects of montelukast in asthmatic volunteers exposed to sulfur dioxide. Chest. 2001 Feb;119(2):402-8. doi: 10.1378/chest.119.2.402[↩]
- Boushey HA. Bronchial hyperreactivity to sulfur dioxide: physiologic and political implications. J Allergy Clin Immunol. 1982 Apr;69(4):335-8. doi: 10.1016/0091-6749(82)90142-7[↩]
- Nowak D, Jörres R, Berger J, Claussen M, Magnussen H. Airway responsiveness to sulfur dioxide in an adult population sample. Am J Respir Crit Care Med. 1997 Oct;156(4 Pt 1):1151-6. doi: 10.1164/ajrccm.156.4.9607025[↩]
- Bush RK, Zoratti E, Taylor SL. Diagnosis of sulfite and aspirin sensitivity. Clin Rev Allergy. 1990 Summer-Fall;8(2-3):159-78. doi: 10.1007/BF02914443[↩]
- Stevenson DD, Simon RA. Sulfites and asthma. J Allergy Clin Immunol. 1984 Oct;74(4 Pt 1):469-72. doi: 10.1016/0091-6749(84)90380-4[↩][↩][↩]
- Schwartz HJ, Chester EH. Bronchospastic responses to aerosolized metabisulfite in asthmatic subjects: potential mechanisms and clinical implications. J Allergy Clin Immunol. 1984 Oct;74(4 Pt 1):511-3. doi: 10.1016/0091-6749(84)90387-7[↩]
- Koepke JW, Christopher KL, Chai H, Selner JC. Dose-Dependent Bronchospasm From Sulfites in Isoetharine. JAMA. 1984;251(22):2982–2983. doi:10.1001/jama.1984.03340460060024[↩]
- Habenicht HA, Preuss L, Lovell RG. Sensitivity to ingested metabisulfites: cause of bronchospasm and urticaria. Immunol Allergy Practice. 1983;5:243.[↩]
- Huang AS, Fraser WM. Are sulfite additives really safe? N Engl J Med. 1984 Aug 23;311(8):542. doi: 10.1056/NEJM198408233110826[↩]
- Schwartz HJ. Sensitivity to ingested metabisulfite: variations in clinical presentation. J Allergy Clin Immunol. 1983 May;71(5):487-9. doi: 10.1016/0091-6749(83)90466-9[↩]
- Twarog FJ, Leung DYM. Anaphylaxis to a Component of Isoetharine (Sodium Bisulfite). JAMA. 1982;248(16):2030–2031. doi:10.1001/jama.1982.03330160078030[↩][↩]
- Wüthrich B, Huwyler T. Das Disulfit-Asthma [Asthma due to disulfites]. Schweiz Med Wochenschr. 1989 Sep 2;119(35):1177-84. German.[↩]
- Tsevat J, Gross GN, Dowling GP. Fatal asthma after ingestion of sulfite-containing wine. Ann Intern Med. 1987 Aug;107(2):263. doi: 10.7326/0003-4819-107-2-263_2[↩]
- Nagy SM, Teuber SS, Loscutoff SM, Murphy PJ. Clustered outbreak of adverse reactions to a salsa containing high levels of sulfites. J Food Prot. 1995;58:95–97.[↩]
- Baker GJ, Collett P, Allen DH. Bronchospasm induced by metabisulphite-containing foods and drugs. Med J Aust. 1981 Nov 28;2(11):614-7. doi: 10.5694/j.1326-5377.1981.tb113018.x[↩]
- Koepke JW, Selner JC, Dunhill AL. Presence of sulfur dioxide in commonly used bronchodilator solutions. J Allergy Clin Immunol. 1983 Nov;72(5 Pt 1):504-8. doi: 10.1016/0091-6749(83)90588-2[↩]
- Sher TH, Schwartz HJ. Bisulfite sensitivity manifesting as an allergic reaction to aerosol therapy. Ann Allergy. 1985 Mar;54(3):224-6.[↩]
- Riggs BS, Harchelroad FP Jr, Poole C. Allergic reaction to sulfiting agents. Ann Emerg Med. 1986 Jan;15(1):77-9. doi: 10.1016/s0196-0644(86)80492-9[↩]
- Asmus MJ, Sherman J, Hendeles L. Bronchoconstrictor additives in bronchodilator solutions. J Allergy Clin Immunol. 1999 Aug;104(2 Pt 2):S53-60. doi: 10.1016/s0091-6749(99)70274-5[↩]
- Langevin PB. Propofol containing sulfite-potential for injury. Chest. 1999 Oct;116(4):1140-1. doi: 10.1378/chest.116.4.1140-a[↩]
- Roth JV, Shields A. A dilemma: How does one treat anaphylaxis in the sulfite allergic patient since epinephrine contains sodium metabisulfite? Anesth Analg. 2004 May;98(5):1499; author reply 1500. doi: 10.1213/01.ane.0000120092.39021.f2[↩]
- Valero AL, Bescos M, Amat P, Malet A. Asma bronquial por exposición laboral a sulfitos [Bronchial asthma caused by occupational sulfite exposure]. Allergol Immunopathol (Madr). 1993 Nov-Dec;21(6):221-4. Spanish.[↩]
- Merget R, Korn M. Metabisulphite-induced occupational asthma in a radiographer. Eur Respir J. 2005 Feb;25(2):386-8. doi: 10.1183/09031936.05.00024304[↩]
- Malo JL, Cartier A, Desjardins A. Occupational asthma caused by dry metabisulphite. Thorax. 1995 May;50(5):585-6; discussion 589. doi: 10.1136/thx.50.5.585[↩]
- Agard C, Nicolet-Akhavan F, Bouillard J, Sandron D. Asthme professionnel aux métabisulfites. Trois observations [Occupational asthma to metabisulfites . Three cases]. Rev Mal Respir. 1998 Sep;15(4):537-40. French.[↩]
- Atkinson DA, Sim TC, Grant JA. Sodium metabisulfite and SO2 release: an under-recognized hazard among shrimp fishermen. Ann Allergy. 1993 Dec;71(6):563-6.[↩]
- Andersson E, Nilsson T, Persson B, Wingren G, Torén K. Mortality from asthma and cancer among sulfite mill workers. Scand J Work Environ Health. 1998 Feb;24(1):12-7. doi: 10.5271/sjweh.273[↩]
- Andersson E, Knutsson A, Hagberg S, Nilsson T, Karlsson B, Alfredsson L, Torén K. Incidence of asthma among workers exposed to sulphur dioxide and other irritant gases. Eur Respir J. 2006 Apr;27(4):720-5. doi: 10.1183/09031936.06.00034305[↩]
- Stevenson DD, Simon RA. Sensitivity to ingested metabisulfites in asthmatic subjects. J Allergy Clin Immunol. 1981 Jul;68(1):26-32. doi: 10.1016/0091-6749(81)90119-6[↩]
- McClellan MD, Wanger JS, Cherniack RM. Attenuation of the metabisulfite-induced bronchoconstrictive response by pretreatment with cromolyn. Chest. 1990 Apr;97(4):826-30. doi: 10.1378/chest.97.4.826[↩][↩]
- Prieto L, Juyol M, Paricio A, Martínez MA, Palop J, Castro J. Oral challenge test with sodium metabisulfite in steroid-dependent asthmatic patients. Allergol Immunopathol (Madr). 1988 Nov-Dec;16(6):393-6.[↩]
- Nolan AL. The sulfite controversy. Food Eng. 1983;84-85:89–90.[↩]
- Simon RA. Sulfite challenge for the diagnosis of sensitivity. Allergy Proc. 1989 Sep-Oct;10(5):357-62. doi: 10.2500/108854189778959858[↩]
- Towns SJ, Mellis CM. Role of acetyl salicylic acid and sodium metabisulfite in chronic childhood asthma. Pediatrics. 1984 May;73(5):631-7.[↩]
- Steinman HA, Le Roux M, Potter PC. Sulphur dioxide sensitivity in South African asthmatic children. S Afr Med J. 1993 Jun;83(6):387-90.[↩]
- Sanz J, Martorell A, Torro I, Carlos Cerda J, Alvarez V. Intolerance to sodium metabisulfite in children with steroid-dependent asthma. J Investig Allergol Clin Immunol. 1992 Jan-Feb;2(1):36-8.[↩]
- Petersen CS, Menné T. Consecutive patch testing with sodium sulfite in eczema patients. Contact Dermatitis. 1992 Nov;27(5):344-5. doi: 10.1111/j.1600-0536.1992.tb03306.x[↩][↩]
- Vena GA, Foti C, Angelini G. Sulfite contact allergy. Contact Dermatitis. 1994 Sep;31(3):172-5. doi: 10.1111/j.1600-0536.1994.tb01959.x[↩]
- Madan V, Walker SL, Beck MH. Sodium metabisulfite allergy is common but is it relevant? Contact Dermatitis. 2007 Sep;57(3):173-6. doi: 10.1111/j.1600-0536.2007.01188.x[↩][↩]
- Malik MM, Hegarty MA, Bourke JF. Sodium metabisulfite–a marker for cosmetic allergy? Contact Dermatitis. 2007 Apr;56(4):241-2. doi: 10.1111/j.1600-0536.2007.00972.x[↩]
- Schorr WF. Multiple injuries from permanents; Presented at Cosmetic Symposium; Chicago, IL: American Academy of Dermatology; 1983. Dec 3[↩]
- Sánchez-Pérez J, Abajo P, Córdoba S, García-Díez A. Allergic contact dermatitis from sodium metabisulfite in an antihemorrhoidal cream. Contact Dermatitis. 2000 Mar;42(3):176-7.[↩]
- Nagayama H, Hatamochi A, Shinkai H. A case of contact dermatitis due to sodium bisulfite in an ophthalmic solution. J Dermatol. 1997 Oct;24(10):675-7. doi: 10.1111/j.1346-8138.1997.tb02315.x[↩]
- Epstein E. Sodium bisulfite. Contact Dematitis Newsletter. 1970;7:155.[↩]
- Nater JP. Allergic contact dermatitis caused by potassium metabisulfite. Dermatologica. 1968;136(6):477-8. doi: 10.1159/000254143[↩]
- Camarasa JG, Barnadas M. Occupational dermatosis by vitamin K3 sodium bisulphite. Contact Dermatitis. 1982 Jul;8(4):268. doi: 10.1111/j.1600-0536.1982.tb04214.x[↩]
- Lee A, Nixon R. Contact dermatitis from sodium metabisulfite in a baker. Contact Dermatitis. 2001 Feb;44(2):127-8. doi: 10.1034/j.1600-0536.2001.44020924.x[↩]
- Dooms-Goossens A, de Alam AG, Degreef H, Kochuyt A. Local anesthetic intolerance due to metabisulfite. Contact Dermatitis. 1989 Feb;20(2):124-6. doi: 10.1111/j.1600-0536.1989.tb03120.x[↩]