Contents
What is delirium
Delirium (sudden confusion) is a serious disturbance in mental abilities that results in confused thinking and reduced awareness of your environment that can come and go (fluctuate) over a 24-hour time period 1. A simple test for confusion is to ask the person their name, age and today’s date, and see if they seem unsure or answer incorrectly. The start of delirium is usually rapid within hours or a few days. Delirium makes paying attention or focusing difficult, and sometimes affects your ability to maintain awareness of your surroundings. Some people hallucinate or become paranoid because it becomes difficult to interpret their environment. Other symptoms may include rambling speech and jumbled thoughts. These symptoms tend to come and go during the course of the day. Confusion regarding day-to-day events, daily routines, and the roles of familiar people is common. Changes in personality can occur. Some persons become quiet and withdrawn while others become agitated or hyperactive. Normal patterns of sleeping and eating are often disrupted. Sleep disturbance (with difficulty falling asleep and frequent awakening leading to sleepless nights) is one of the earliest prodromal symptoms of delirium 2. Additional clinical features of delirium include delusions, dysarthria, dysgraphia, emotional lability, and abnormal psychomotor activity (hypo- or hyperactivity).
Delirium can be thought of as “acute brain failure” 3 because of its sudden and dramatic onset. The exact pathogenesis of delirium remains unclear.
Delirium is a clinical diagnosis that is frequently overlooked or misdiagnosed by the healthcare team 4. This may be due to lack of recognition of delirium features, overlap with the clinical features of depression and dementia in particular, the occurrence of fluctuating symptom intensity and transient lucidity, and is compounded by inadequate delirium screening or an absence of regular screening 5.
Delirium may have a single cause or more than one cause, such as a medical condition and medication toxicity. Sometimes no cause can be identified.
Delirium can often be traced to one or more contributing factors, such as:
- an infection,
- a severe or chronic medical illness,
- changes in your metabolic balance (such as low sodium),
- medication,
- surgery,
- constipation,
- dehydration,
- alcohol or drug withdrawal.
People with cognitive impairment, or hearing or vision loss, are also at increased risk of delirium.
People with dementia are six times more likely to get delirium than other people. Because symptoms of delirium and dementia can be similar, input from a family member or caregiver may be important for a doctor to make an accurate diagnosis. Family member or caregiver input about the person’s symptoms, as well as his or her typical thinking and everyday abilities, will be important for a proper diagnosis and for finding the underlying cause.
Delirium often occurs in older people in hospital or in a long-term care facility (nursing home). In the hospital, the prevalence of delirium is 18–35% in general medical inpatients and up to 50% in intensive care patients 6. In a palliative setting, prevalence is reported as 13–42% on admission to inpatient palliative care units, increasing to 88% at the end of life (weeks–hours before death) 7. People who have delirium when they are in hospital are at higher risk of developing further medical complications.
Patients who develop delirium in the intensive care unit (ICU) are at 2–4 fold increased risk of death both in and out of the hospital 8; those who develop delirium on general medical or geriatric wards are at 1.5-fold increased risk for death in the year following hospitalization 9; and patients with delirium in the emergency department have an approximately 70% increased risk of death during the first six months after the visit 10.
Experts have identified three types of delirium 11:
- Hyperactive delirium. Probably the most easily recognized type, this may include restlessness (for example, pacing), agitation, rapid mood changes, hypervigilance and hallucinations.
- Hypoactive delirium. Is the most frequent pattern in older adults. Hypoactive delirium is characterized by inactivity or reduced motor activity, sluggishness, abnormal drowsiness or seeming to be in a daze 12.
- Mixed delirium. This includes both hyperactive and hypoactive symptoms. The person may quickly switch back and forth from hyperactive to hypoactive states.
The more exuberant hyperactive delirium is easier to detect and is often associated with alcohol withdrawal 13. Hypoactive delirium frequently goes undetected or is interpreted as a mood disorder or fatigue, which can delay diagnosis and the implementation of therapeutic measures 14. Although the hypoactive pattern is more commonly indicated in the literature as being associated with a worse prognosis, current evidence to support this claim is conflicting. Of the seven studies identified in a recent systematic review that examined the effects of motor subtype on the clinical course of patients with delirium, only three showed an association between hypoactive delirium and a worse prognosis 15. Of the four other studies, one concluded that hyperactive delirium had worse prognosis 16, and the other three did not find any association between delirium motor subtypes and the examined outcomes 17. A recent study 11 involving 1409 hospital admitted elderly patients with average age of 80 years found that the predominant motor subtype of delirium was the hypoactive form (53%), and that hospitalized older adults with hypoactive delirium, either exclusively or in alternation with the hyperactive pattern, had a substantially lower hospital survival than patients without delirium or with pure hyperactive delirium. In contrast, the authors did not verify statistically significant associations between delirium motor subtypes and 12-month mortality. Other factors demonstrated to have prognostic importance in that study were age, admission from emergency department or ICU, functional dependency, malnutrition, cancer, reduced GFR (kidney function test), hypoalbuminemia, leukocytosis, and elevated C-reactive protein 11.
The association of hypoactive and mixed delirium subtypes with a worse prognosis was demonstrated even after adjusting the analysis for demographic, clinical and laboratory characteristics. In one of the earliest studies on delirium motor subtypes, Liptzin and Levkoff 18 suggested that, in most cases, the expression of characteristics associated with hyperactive delirium would only happen in individuals who were sufficiently fit to manifest agitation. This theory is consistent with the fact that hypoactive delirium is more common in older, frailer, more dependent patients, and therefore with the perception that it might be a marker of clinical vulnerability. The higher incidence of hypoactive delirium in long term care facilities and geriatric wards, is also consistent with this understanding 19.
Delirium prognosis
Prognosis depends on both successful treatment of the causative disease and the underlying state of the brain. A proportion of patients may not survive their causative illness. With successful treatment of the underlying condition, however, recovery should be expected 4-7 days after resolution of this condition.
Delirium complications
Delirium may last only a few hours or as long as several weeks or months. If issues contributing to delirium are addressed, the recovery time is often shorter.
The degree of recovery depends to some extent on the health and mental status before the onset of delirium. People with dementia, for example, may experience a significant overall decline in memory and thinking skills. People in better health are more likely to fully recover.
People with other serious, chronic or terminal illnesses may not regain the levels of thinking skills or functioning that they had before the onset of delirium. Delirium in seriously ill people is also more likely to lead to:
- General decline in health
- Poor recovery from surgery
- Need for institutional care
- Increased risk of death, especially with hypoactive delirium
Delirium causes
Delirium occurs when the normal sending and receiving of signals in the brain become impaired. This impairment is most likely caused by a combination of factors that make the brain vulnerable and trigger a malfunction in brain activity.
Delirium may have a single cause or more than one cause, such as a medical condition and medication toxicity. Sometimes no cause can be identified.
Possible causes include:
- Certain medications or drug toxicity
- Alcohol or drug abuse or withdrawal
- A medical condition
- Metabolic imbalances, such as low sodium or low calcium
- A lack of oxygen in the blood (hypoxia) – the cause could be anything from a severe asthma attack to a problem with the lungs or heart
- Severe, chronic or terminal illness
- Fever and acute infection, particularly in children
- Exposure to a toxin
- Malnutrition or dehydration
- Sleep deprivation or severe emotional distress
- Pain
- Surgery or other medical procedures that include anesthesia
- A stroke or TIA (“mini stroke”)
- A low blood sugar level (hypoglycemia)
- Diabetic ketoacidosis, a serious complication of diabetes caused by a lack of insulin in the body
- Certain medications, including digoxin, diuretics, steroids, and opiates
- Alcohol poisoning or alcohol withdrawal
- Drug misuse
Several medications or combinations of drugs can trigger delirium, including some types of:
- Pain drugs
- Sleep medications
- Medications for mood disorders, such as anxiety and depression
- Allergy medications (antihistamines)
- Parkinson’s disease drugs
- Drugs for treating spasms or convulsions
- Asthma medications
Less common causes of delirium are:
- an infection of the brain or its lining (encephalitis or meningitis)
- an imbalance of salts and minerals in the blood
- a severely underactive thyroid gland (hypothyroidism)
- thiamine (vitamin B1) deficiency
- a brain tumor
- hypoparathyroidism or hyperparathyroidism (rare hormone disorders)
- Cushing’s disease (a tumor of the pituitary gland)
- an epileptic seizure
- carbon monoxide poisoning
Risk factors for delirium
Any condition that results in a hospital stay, especially in intensive care or after surgery, increases the risk of delirium, as does being a resident in a nursing home.
Examples of other conditions that increase the risk of delirium include:
- Brain disorders such as dementia, stroke, Alzheimer’s disease or Parkinson’s disease
- Older age
- Previous delirium episodes
- Having multiple medical problems
- Cognitive impairment
- Functional impairment
- Vision impairment
- Hearing impairment
- Comorbidity/severity of illness
- Depression
- History of transient ischemia/stroke
- Alcohol abuse
Precipitating Factors
- Medications
- Multiple medications added
- Psychoactive medication use
- Sedative-hypnotics
- Use of physical restraints
- Use of bladder catheter
- Physiologic
- Elevated serum urea
- Elevated BUN/creatinine ratio
- Abnormal serum albumin
- Abnormal sodium, glucose, or potassium
- Metabolic acidosis
- Infection
- Any iatrogenic event
- Surgery
- Aortic aneurysm
- Non-cardiac thoracic
- Neurosurgery
- Trauma admission
- Urgent admission
- Coma
Delirium prevention
The most successful approach to preventing delirium is to target risk factors that might trigger an episode. Hospital environments present a special challenge — frequent room changes, invasive procedures, loud noises, poor lighting and lack of natural light can worsen confusion.
Evidence indicates that these strategies — promoting good sleep habits, helping the person remain calm and well-oriented, and helping prevent medical problems or other complications — can help prevent or reduce the severity of delirium.
Delirium vs Dementia
Dementia and delirium may be particularly difficult to distinguish, and a person may have both. In fact, frequently delirium occurs in people with dementia.
Dementia is the progressive decline of memory and other thinking skills due to the gradual dysfunction and loss of brain cells. The most common cause of dementia is Alzheimer’s disease.
Some differences between the symptoms of delirium and dementia include:
- Onset. The onset of delirium occurs within a short time, while dementia usually begins with relatively minor symptoms that gradually worsen over time.
- Attention. The ability to stay focused or maintain attention is significantly impaired with delirium. A person in the early stages of dementia remains generally alert.
- Fluctuation. The appearance of delirium symptoms can fluctuate significantly and frequently throughout the day. While people with dementia have better and worse times of day, their memory and thinking skills stay at a fairly constant level during the course of a day.
Does delirium lead to dementia?
A major area of controversy is whether delirium is simply a marker of vulnerability to dementia, or whether delirium itself leads to dementia. Ultimately, it is likely that both hypotheses are true 3. There is little doubt that occurrence of an episode of delirium can signal vulnerability of the brain with decreased cognitive reserve and increased risk for future dementia. In some cases, delirium may bring previously unrecognized cognitive impairment to medical attention. Delirium and dementia commonly coexist, with dementia being a leading risk factor for delirium, i.e., increasing delirium risk by 2–5 fold on hospital admission. Moreover, the evidence for delirium leading to permanent cognitive impairment and dementia is increasing, ranging from epidemiologic evidence to tissue culture and animal models. A recent meta-analysis 20 involving two studies with 241 total patients demonstrated that delirium was associated with an increased rate of incident dementia. In a sample of 225 cardiac surgery patients, delirium resulted in a severe punctuated decline in cognitive functioning, followed by recovery over 6–12 months in most patients; however, a substantial proportion, particularly those with prolonged delirium, never return to baseline 21. In 263 patients with Alzheimer’s disease, delirium resulted in a fundamental alteration in the trajectory of cognitive decline with a 2-fold acceleration in rate of decline over the year following hospitalization, and accelerated decline persisting over the 5-year follow-up period 22.
Additional evidence supports a more direct role for delirium in dementia. An important study with neuropathological confirmation 23 demonstrated that in 553 individuals who were 85 years and older at baseline, delirium increased the risk of incident dementia. In patients without delirium, Alzheimer’s disease pathology was significantly associated with dementia, whereas no such relationship was seen in those with delirium, raising the possibility of alternative pathologic mechanisms for dementia following delirium. This study was limited, however, by a high rate of missing follow-up observations. Previous studies in animal models and human neuronal cell culture have demonstrated that exposure to inhalational anesthetics may induce neurotoxicity, including apoptosis, caspase activation, A-beta oligomerization and accumulation, neuroinflammation, and mitochondrial dysfunction 24. Preliminary results in humans 25 suggest some inhalational anesthetic agents (e.g., isoflurane) may be more neurotoxic than others. Important recent work involving animal models of delirium have demonstrated that in vulnerable animals, systemic inflammatory insults can cause punctuated cognitive decline typical of delirium, followed by acceleration in disease progression typical of dementia 26. Furthermore, a single dose of lipopolysaccharide, inducing an inflammatory insult comparable to a moderate infection in humans, has been shown to induce neuronal death, microglial activation, decreased regional blood flow, and loss of cholinergic activation 27. This accumulating evidence, therefore, lends strong support for the impact of delirium itself contributing to and/or being a mediator of permanent cognitive impairment. Future human studies with careful baseline characterization of cognitive function, control for confounding factors, and long-term follow-up, including neuropsychological testing and neuroimaging, will be helpful to address this important area.
Delirium symptoms
Signs and symptoms of delirium usually begin over a few hours or a few days. They often fluctuate throughout the day, and there may be periods of no symptoms. Symptoms tend to be worse during the night when it’s dark and things look less familiar. Primary signs and symptoms include those below.
Reduced awareness of the environment
This may result in:
- An inability to stay focused on a topic or to switch topics
- Getting stuck on an idea rather than responding to questions or conversation
- Being easily distracted by unimportant things
- Being withdrawn, with little or no activity or little response to the environment
Poor thinking skills (cognitive impairment)
This may appear as:
- Poor memory, particularly of recent events
- Disorientation, for example, not knowing where you are or who you are
- Difficulty speaking or recalling words
- Rambling or nonsense speech
- Trouble understanding speech
- Difficulty reading or writing
Behavior changes
This may include:
- Seeing things that don’t exist (hallucinations)
- Restlessness, agitation or combative behavior
- Calling out, moaning or making other sounds
- Being quiet and withdrawn — especially in older adults
- Slowed movement or lethargy
- Disturbed sleep habits
- Reversal of night-day sleep-wake cycle
Emotional disturbances
This may appear as:
- Anxiety, fear or paranoia
- Depression
- Irritability or anger
- A sense of feeling elated (euphoria)
- Apathy
- Rapid and unpredictable mood shifts
- Personality changes
Delirium diagnosis
A doctor will diagnose delirium based on medical history, tests to assess mental status and the identification of possible contributing factors. An examination may include:
- Mental status assessment. A doctor starts by assessing awareness, attention and thinking. This may be done informally through conversation, or with tests or screenings that assess mental state, confusion, perception and memory.
- Physical and neurological exams. The doctor performs a physical exam, checking for signs of health problems or underlying disease. A neurological exam — checking vision, balance, coordination and reflexes — can help determine if a stroke or another neurological disease is causing the delirium.
- Other possible tests. The doctor may order blood, urine and other diagnostic tests. Brain-imaging tests may be used when a diagnosis can’t be made with other available information.
Delirium is a clinical diagnosis, which is often unrecognized and easily overlooked. Recognition requires a brief cognitive screening and astute clinical observation. Key diagnostic features include an acute onset and fluctuating course of symptoms, inattention, impaired level of consciousness, and disturbance of cognition (e.g., disorientation, memory impairment, alteration in language) 28.
Supportive features include:
- disturbance in sleep-wake cycle,
- perceptual disturbances (hallucinations or illusions),
- delusions, psychomotor disturbance (hypo- or hyper-activity),
- inappropriate behavior, and
- emotional lability.
The current reference standard diagnostic criteria are the Diagnostic and Statistical Manual of the American Psychiatric Association (DSM-IV TR) 29 and the International Classification of Diseases (ICD-10) from the World Health Organization 30. Over 24 delirium instruments have been used in published studies. The most widely used instrument for identification of delirium is the Confusion Assessment Method (CAM) 31 validated in high quality studies including over 1000 patients with sensitivity of 94%, specificity of 89%, and high inter-rater reliability. Cognitive testing and training are recommended for optimal use of the Confusion Assessment Method (CAM). The Confusion Assessment Method (CAM), which has been used in over 4,000 published studies to date and translated into at least 12 languages, has been adapted for use in the ICU 32, emergency department 33, and nursing home, where it is now included as part of the Minimum Data Set 34, a standardized comprehensive assessment of all residents in U.S. long-term care facilities. Behavioral checklists for delirium symptoms, such as Delirium Observation Screening 35, Nursing Delirium Screening Scale (Nu-DESC) 36 and NEECHAM 37, are used particularly in nursing-based studies. For measuring delirium severity, the most widely used tools include the Delirium Rating Scale (DRS and DRS-98) 38 and Memorial Delirium Assessment Scale (MDAS) 39. Summation of Confusion Assessment Method (CAM) items has been used as a severity indicator 40. A validated chart review method for identification of delirium has been developed for retrospective identification, but its sensitivity is more limited. The Family Confusion Assessment Method (FAM-CAM) 41 has been developed to identify delirium symptoms from reports of family and informal caregivers, which holds promise to assist with early recognition of delirium.
The Confusion Assessment Method (CAM) Diagnostic Algorithm 3
- Feature 1. Acute onset and fluctuating course
This feature is usually obtained from a reliable reporter, such as a family member, caregiver, or nurse and is shown by positive responses to the questions: Is there evidence of an acute change in mental status from the patient’s baseline? Did the (abnormal) behavior fluctuate during the day, that is, tend to come and go, or did it increase or decrease in severity? - Feature 2. Inattention
This feature is shown by a positive response to this question: Did the patient have difficulty focusing attention, for example, being easily distractible, or have difficulty keeping track of what was being said? - Feature 3. Disorganized thinking
This feature is shown by a positive response to this question: Was the patient’s thinking disorganized or incoherent, such as rambling or irrelevant conversation, unclear or illogical flow of ideas, or unpredictable switching from subject to subject? - Feature 4. Altered level of consciousness
This feature is shown by any answer other than “alert” to this question: Overall, how would you rate this patient’s level of consciousness (alert [normal], vigilant [hyperalert], lethargic [drowsy, easily aroused], stupor [difficult to arouse], or coma [unarousable])?
The diagnosis of delirium by Confusion Assessment Method (CAM) requires the presence of features 1 and 2 and of either 3 or 4. The ratings for the CAM should be completed following brief cognitive assessment of the patient, for example, with the Short Portable Mental Status Questionnaire 42, the Mini-Cog 43 or the Montreal Cognitive Asessment 44.
Delirium treatment
The first goal of treatment for delirium is to address any underlying causes or triggers — for example, by stopping use of a particular medication or treating an infection. Treatment then focuses on creating the best environment for healing the body and calming the brain.
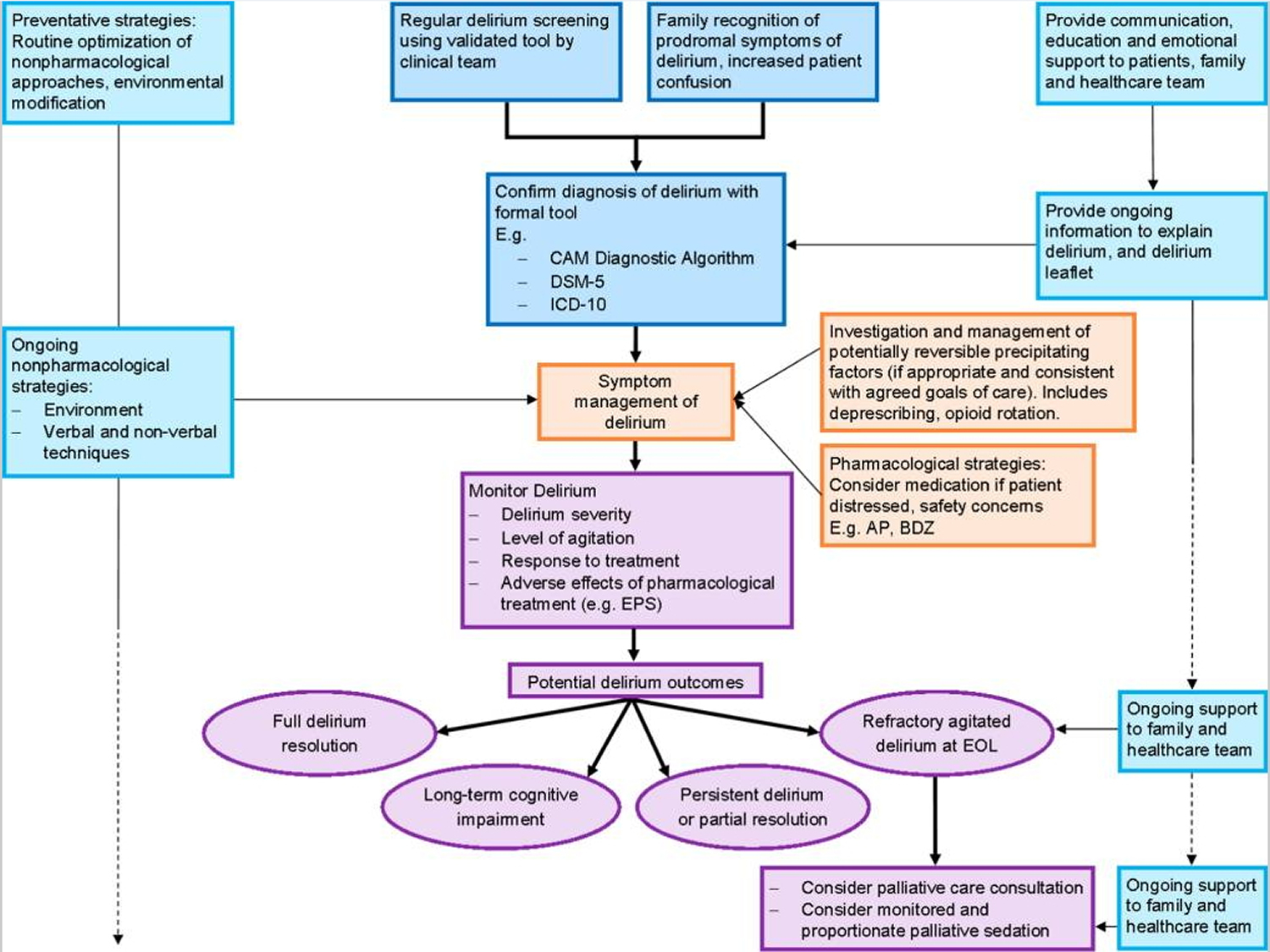
Figure 1. Algorithm for the assessment and management of delirium in patients.
[Source 45]Table 1. Evaluation and Management of Suspected Delirium
| Evaluation of Delirium | |
| History |
|
| Vital signs |
|
| Physical and neurological examination |
|
| Targeted laboratory evaluation (selected tests based on clues from history and physical) | Based on history and physical examination, consider:
|
| Targeted neuroimaging (selected patients) |
|
| Electroencephalography (selected patients) |
|
| Management of Delirium | |
| Medication adjustments |
|
| Address acute medical issues |
|
| Reorientation strategies |
|
| Maintain safe mobility |
|
| Normalize sleep-wake cycle |
|
| Pharmacologic management (severe agitation or psychosis only) |
|
Note: *BID=twice daily; CBC=complete blood count; IM=intramuscular; mgs=milligrams; po=by mouth; PRN=as needed medication.
[Source 3]Supportive care
Supportive care aims to prevent complications by:
- Protecting the airway
- Providing fluids and nutrition
- Assisting with movement
- Treating pain
- Addressing incontinence
- Avoiding use of physical restraints and bladder tubes
- Avoiding changes in surroundings and caregivers when possible
- Encouraging the involvement of family members or familiar people
If you’re a relative or caregiver of someone at risk of or recovering from delirium, you can take steps to help improve the person’s health, prevent a recurrence and manage responsibilities.
Promote good sleep habits
To promote good sleep habits:
- Provide a calm, quiet environment
- Keep inside lighting appropriate for the time of day
- Plan for uninterrupted periods of sleep at night
- Help the person keep a regular daytime schedule
- Encourage self-care and activity during the day
Promote calmness and orientation
To help the person remain calm and well-oriented:
- Provide a clock and calendar and refer to them regularly throughout the day
- Communicate simply about any change in activity, such as time for lunch or time for bed
- Keep familiar and favorite objects and pictures around, but avoid a cluttered environment
- Approach the person calmly
- Identify yourself or other people regularly
- Avoid arguments
- Use comfort measures, such as reassuring touch, when appropriate
- Keep noise levels and other distractions to a minimum
- Provide and maintain eyeglasses and hearing aids
Prevent complicating problems
Help prevent medical problems by:
- Giving the person the proper medication on a regular schedule
- Providing plenty of fluids and a healthy diet
- Encouraging regular physical activity
- Getting treatment for potential problems, such as infection or metabolic imbalances, early
Caring for the caregiver
Providing regular care for a person with delirium can be scary and exhausting. Take care of yourself, too.
- Consider joining a support group for caregivers.
- Learn more about the condition.
- Ask for educational materials or other resources from a health care provider, nonprofit organizations, community health services or government agencies.
- Share caregiving with family and friends who are familiar the person so you get a break.
Examples of organizations that may provide helpful information include the National Family Caregivers Association and the National Institute on Aging.
Medications
Talk with the doctor about avoiding or minimizing the use of drugs that may trigger delirium. Certain medications may be needed to control pain that’s causing delirium.
Other types of drugs may help calm a person who misinterprets the environment in a way that leads to severe paranoia, fear or hallucinations, and when severe agitation or confusion occurs. These drugs may be needed when certain behaviors:
- Prevent the performance of a medical exam or treatment
- Endanger the person or threaten the safety of others
- Don’t lessen with nondrug treatments
These medications are usually reduced in dose or discontinued when the delirium resolves.
Delirium tremens treatment
Delirium tremens is a severe form of alcohol withdrawal. It involves sudden and severe mental or nervous system changes.
Delirium tremens can occur when you stop drinking alcohol after a period of heavy drinking, especially if you do not eat enough food.
Delirium tremens may also be caused by head injury, infection, or illness in people with a history of heavy alcohol use.
It occurs most often in people who have a history of alcohol withdrawal. It is especially common in those who drink 4 to 5 pints (1.8 to 2.4 liters) of wine, 7 to 8 pints (3.3 to 3.8 liters) of beer, or 1 pint (1/2 liter) of “hard” alcohol every day for several months. Delirium tremens also commonly affects people who have used alcohol for more than 10 years.
Symptoms most often occur within 48 to 96 hours after the last drink. But, they can occur 7 to 10 days after the last drink.
Symptoms may get worse quickly, and can include:
- Delirium, which is sudden severe confusion
- Body tremors
- Changes in mental function
- Agitation, irritability
- Deep sleep that lasts for a day or longer
- Excitement or fear
- Hallucinations (seeing or feeling things that are not really there)
- Bursts of energy
- Quick mood changes
- Restlessness, excitement
- Sensitivity to light, sound, touch
- Stupor, sleepiness, fatigue
Seizures (may occur without other symptoms of delirium tremens):
- Most common in the first 12 to 48 hours after the last drink
- Most common in people with past complications from alcohol withdrawal
- Usually generalized tonic-clonic seizures
Symptoms of alcohol withdrawal, including:
- Anxiety, depression
- Fatigue
- Headache
- Insomnia (difficulty falling and staying asleep)
- Irritability or excitability
- Loss of appetite
- Nausea, vomiting
- Nervousness, jumpiness, shakiness, palpitations (sensation of feeling the heart beat)
- Pale skin
- Rapid emotional changes
- Sweating, especially on the palms of the hands or the face
Other symptoms that may occur:
- Chest pain
- Fever
- Stomach pain
Delirium tremens treatment
The goals of treatment are to:
- Save the person’s life
- Relieve symptoms
- Prevent complications
A hospital stay is needed. The health care team will regularly check:
- Blood chemistry results, such as electrolyte levels
- Body fluid levels
- Vital signs (temperature, pulse, breathing rate, blood pressure)
While in the hospital, the person will receive medicines to:
- Stay calm and relaxed (sedated) until the delirium tremens are finished
- Treat seizures, anxiety, or tremors
- Treat mental disorders, if any
Long-term preventive treatment should begin after the person recovers from delirium tremens symptoms. This may involve:
- A “drying out” period, in which no alcohol is allowed
- Total and lifelong avoidance of alcohol (abstinence)
- Counseling
- Going to support groups (such as Alcoholics Anonymous)
Treatment may be needed for other medical problems that can occur with alcohol use, including:
- Alcoholic cardiomyopathy
- Alcoholic liver disease
- Alcoholic neuropathy
- Wernicke-Korsakoff syndrome
- American Psychiatric Association . Diagnostic and statistical manual of mental disorders, fifth edition (DSM-5) Arlington: American Psychiatric Association; 2013.[↩]
- Kerr CW, Donnelly J, Wright S, Luczkiewicz D, McKenzie K, Hang P, et al. Progression of delirium in advanced illness: a multivariate model of caregiver and clinician perspectives. J Palliat Med. 2013;16(7):768–773. https://www.ncbi.nlm.nih.gov/pubmed/23718872[↩]
- Inouye SK, Westendorp RGJ, Saczynski JS. Delirium in elderly people. Lancet. 2014;383(9920):911-922. doi:10.1016/S0140-6736(13)60688-1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4120864/[↩][↩][↩][↩]
- De La Cruz M, Fan J, Yennu S, Tanco K, Shin S, Wu J, et al. The frequency of missed delirium in patients referred to palliative care in a comprehensive cancer center. Support Care Cancer. 2015;23(8):2427–2433. https://www.ncbi.nlm.nih.gov/pubmed/25617070[↩]
- Lawlor PG, Bush SH. Delirium in patients with cancer: assessment, impact, mechanisms and management. Nat Rev Clin Oncol. 2015;12(2):77–92. https://www.ncbi.nlm.nih.gov/pubmed/25178632[↩]
- Inouye SK, Westendorp RG, Saczynski JS. Delirium in elderly people. Lancet. 2014;383(9920):911–922. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4120864/[↩]
- Hosie A, Davidson PM, Agar M, Sanderson CR, Phillips J. Delirium prevalence, incidence, and implications for screening in specialist palliative care inpatient settings: a systematic review. Palliat Med. 2013;27(6):486–498. https://www.ncbi.nlm.nih.gov/pubmed/22988044[↩]
- van den Boogaard M, Schoonhoven L, van der Hoeven JG, van Achterberg T, Pickkers P. Incidence and short-term consequences of delirium in critically ill patients: A prospective observational cohort study. Int J Nurs Stud. 2012 Jul;49(7):775–83. https://www.ncbi.nlm.nih.gov/pubmed/22197051[↩]
- Buurman BM, Hoogerduijn JG, de Haan RJ, Abu-Hanna A, Lagaay AM, Verhaar HJ, et al. Geriatric conditions in acutely hospitalized older patients: prevalence and one-year survival and functional decline. PLoS One. 2011;6(11):e26951. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3215703/[↩]
- Han JH, Shintani A, Eden S, Morandi A, Solberg LM, Schnelle J, et al. Delirium in the emergency department: an independent predictor of death within 6 months. Ann Emerg Med. 2010 Sep;56(3):244–52. e1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3768121/[↩]
- Avelino-Silva TJ, Campora F, Curiati JAE, Jacob-Filho W (2018) Prognostic effects of delirium motor subtypes in hospitalized older adults: A prospective cohort study. PLoS ONE 13(1): e0191092. https://doi.org/10.1371/journal.pone.0191092[↩][↩][↩]
- Morandi A, Di Santo SG, Cherubini A, Mossello E, Meagher D, Mazzone A, et al. Clinical Features Associated with Delirium Motor Subtypes in Older Inpatients: Results of a Multicenter Study. Am Journal Geriatr Psychiatry. 2017. pmid:28579352.[↩]
- Inouye SK, Westendorp RG, Saczynski JS. Delirium in elderly people. Lancet. 2014;383(9920):911–22. Epub 2013/09/03.[↩]
- Eubank KJ, Covinsky KE. Delirium severity in the hospitalized patient: time to pay attention. Ann Intern Med. 2014;160(8):574–5. Epub 2014/04/16.[↩]
- Jackson TA, Wilson D, Richardson S, Lord JM. Predicting outcome in older hospital patients with delirium: a systematic literature review. Int J Geriatr Psychiatry. 2015[↩]
- Marcantonio E, Ta T, Duthie E, Resnick NM. Delirium severity and psychomotor types: their relationship with outcomes after hip fracture repair. J Am Geriatr Soc. 2002;50(5):850–7. Epub 2002/05/25.[↩]
- Slor CJ, Adamis D, Jansen RW, Meagher DJ, Witlox J, Houdijk AP, et al. Delirium motor subtypes in elderly hip fracture patients: risk factors, outcomes and longitudinal stability. J Psychosom Res. 2013;74(5):444–9.[↩]
- Liptzin B, Levkoff SE. An empirical study of delirium subtypes. Br J Psychiatry. 1992;161:843–5. Epub 1992/12/01.[↩]
- DeCrane SK, Culp KR, Wakefield B. Twelve-month mortality among delirium subtypes. Clinical Nurs Res. 2011;20(4):404–21.[↩]
- Witlox J, Eurelings LS, de Jonghe JF, Kalisvaart KJ, Eikelenboom P, van Gool WA. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010 Jul 28;304(4):443–51. https://www.ncbi.nlm.nih.gov/pubmed/20664045[↩]
- Saczynski JS, Marcantonio ER, Quach L, Fong TG, Gross A, Inouye SK, et al. Cognitive trajectories after postoperative delirium. N Engl J Med. 2012 Jul 5;367(1):30–9. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3433229/[↩]
- Gross AL, Jones RN, Habtemariam DA, Fong TG, Tommet D, Quach L, et al. Delirium and Long-term Cognitive Trajectory Among Persons With Dementia. Archives of internal medicine. 2012;172(17):1–8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3740440/[↩]
- Davis DH, Muniz Terrera G, Keage H, Rahkonen T, Oinas M, Matthews FE, et al. Delirium is a strong risk factor for dementia in the oldest-old: a population-based cohort study. Brain. 2012 Sep;135(Pt 9):2809–16. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3437024/[↩]
- Zhang Y, Xu Z, Wang H, Dong Y, Shi HN, Culley DJ, et al. Anesthetics isoflurane and desflurane differently affect mitochondrial function, learning, and memory. Ann Neurol. 2012 May;71(5):687–98. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3942786/[↩]
- Zhang B, Tian M, Zhen Y, Yue Y, Sherman J, Zheng H, et al. The effects of isoflurane and desflurane on cognitive function in humans. Anesth Analg. 2012 Feb;114(2):410–5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3972614/[↩]
- Cunningham C, Campion S, Lunnon K, Murray CL, Woods JF, Deacon RM, et al. Systemic inflammation induces acute behavioral and cognitive changes and accelerates neurodegenerative disease. Biol Psychiatry. 2009 Feb 15;65(4):304–12. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2633437/[↩]
- Cunningham C. Systemic inflammation and delirium: important co-factors in the progression of dementia. Biochem Soc Trans. 2011 Aug;39(4):945–53. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4157218/[↩]
- American Psychiatric Association. Task force on DSM-IV, diagnostic and statistical manual of mental disorders: DSM-IV. 4. Washington, DC: The Association; 1994.[↩]
- American Psychiatric Association. Task force on DSM-IV, diagnostic and statistical manual of mental disorders: DSM-IV (text revision) 4. Washington, DC: The Association; 2000.[↩]
- World Health Organization. The ICD-10 classification of mental and behavioural disorders: diagnostic criteria for research. World Health Organization; 1993.[↩]
- O’Mahony R, Murthy L, Akunne A, Young J. Synopsis of the National Institute for Health and Clinical Excellence guideline for prevention of delirium. Ann Intern Med. 2011 Jun 7;154(11):746–51. https://www.ncbi.nlm.nih.gov/pubmed/21646557[↩]
- Ely EW, Margolin R, Francis J, May L, Truman B, Dittus R, et al. Evaluation of delirium in critically ill patients: validation of the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) Crit Care Med. 2001 Jul;29(7):1370–9. https://www.ncbi.nlm.nih.gov/pubmed/11445689[↩]
- Han JH, Zimmerman EE, Cutler N, Schnelle J, Morandi A, Dittus RS, et al. Delirium in older emergency department patients: recognition, risk factors, and psychomotor subtypes. Acad Emerg Med. 2009 Mar;16(3):193–200. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5015887/[↩]
- Center for Medicare & Medicaid Services (CMS) Minimum Data Set, Version 3.0. Centers for Medicare & Medicaid Services; 2010.[↩]
- Schuurmans MJ, Shortridge-Baggett LM, Duursma SA. The Delirium Observation Screening Scale: a screening instrument for delirium. Res Theory Nurs Pract. 2003 Spring;17(1):31–50. https://www.ncbi.nlm.nih.gov/pubmed/12751884[↩]
- Gaudreau JD, Gagnon P, Harel F, Tremblay A, Roy MA. Fast, systematic, and continuous delirium assessment in hospitalized patients: the nursing delirium screening scale. J Pain Symptom Manage. 2005 Apr;29(4):368–75. https://www.ncbi.nlm.nih.gov/pubmed/15857740[↩]
- Neelon VJ, Champagne MT, Carlson JR, Funk SG. The NEECHAM Confusion Scale: construction, validation, and clinical testing. Nurs Res. 1996 Nov-Dec;45(6):324–30. https://www.ncbi.nlm.nih.gov/pubmed/8941300[↩]
- Trzepacz PT, Mittal D, Torres R, Kanary K, Norton J, Jimerson N. Validation of the Delirium Rating Scale-revised-98: comparison with the delirium rating scale and the cognitive test for delirium. J Neuropsychiatry Clin Neurosci. 2001 Spring;13(2):229–42. https://www.ncbi.nlm.nih.gov/pubmed/11449030[↩]
- Breitbart W, Rosenfeld B, Roth A, Smith MJ, Cohen K, Passik S. The Memorial Delirium Assessment Scale. J Pain Symptom Manage. 1997 Mar;13(3):128–37. https://www.ncbi.nlm.nih.gov/pubmed/9114631[↩]
- Cole MG, Dendukuri N, McCusker J, Han L. An empirical study of different diagnostic criteria for delirium among elderly medical inpatients. J Neuropsychiatry Clin Neurosci. 2003 Spring;15(2):200–7. https://www.ncbi.nlm.nih.gov/pubmed/12724462[↩]
- Steis MR, Evans L, Hirschman KB, Hanlon A, Fick DM, Flanagan N, et al. Screening for delirium using family caregivers: convergent validity of the family confusion assessment method and interviewer-rated confusion assessment method. J Am Geriatr Soc. 2012 Nov;60(11):2121–6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3498543/[↩]
- Pfeiffer E. A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients. J Am Geriatr Soc. 1975 Oct;23(10):433-41.[↩]
- Borson S, Scanlan J, Brush M, Vitaliano P, Dokmak A. The mini-cog: a cognitive ‘vital signs’ measure for dementia screening in multi-lingual elderly. Int J Geriatr Psychiatry. 2000 Nov;15(11):1021-7.[↩]
- Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005 Apr;53(4):695-9.[↩]
- Bush SH, Tierney S, Lawlor PG. Clinical Assessment and Management of Delirium in the Palliative Care Setting. Drugs. 2017;77(15):1623-1643. doi:10.1007/s40265-017-0804-3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5613058/[↩]