Contents
- Hypothyroidism
- Thyroid gland
- Causes of hypothyroidism
- Hypothyroidism signs and symptoms
- Hypothyroidism complications
- Hypothyroidism diagnosis
- Hypothyroidism treatment
- Hashimoto’s thyroiditis
Hypothyroidism
Hypothyroidism is a medical term for underactive thyroid, where your thyroid gland does not make enough thyroid hormone for normal body function 1, 2, 3, 4. Your thyroid gland is a butterfly-shaped gland with 2 lobes (the right lobe and the left lobe — joined by a narrow piece of the thyroid gland called the isthmus) that is located in front of your neck near the base of your throat, beneath the larynx (voice box or Adam’s apple) (Figure 1). In most people, the thyroid gland cannot be seen or felt. Your thyroid gland produces thyroid hormones, tri-iodothyronine (T3) and thyroxine (T4) (the main hormones that your thyroid gland makes) and calcitonin. The thyroid hormones, T3 (tri-iodothyronine) and T4 (thyroxine) influence important body processes such as body temperature, energy levels, growth, your digestion, muscles and heart. Thyroid hormones are important for how your body uses energy, your metabolism, so thyroid hormones affect nearly every organ in your body even the way your heart beats. You might put on weight and feel very tired and lacking in energy if your thyroid gland doesn’t make enough T3 (tri-iodothyronine) and T4 (thyroxine). Although hypothyroidism may be temporary in some cases, it usually is a permanent condition. Of the nearly 30 million people estimated to be suffering from thyroid dysfunction, most have hypothyroidism.
About 4.6 percent of the U.S. population ages 12 and older has hypothyroidism, although most cases are mild or have few obvious symptoms 5, 2. That’s almost 5 people out of 100 6.
Hypothyroidism is more common in women, people with other thyroid problems, and those over 60 years old 7, 8, 9. Hashimoto’s disease (Hashimoto’s thyroiditis) an autoimmune disorder, is the most common cause of hypothyroidism in the United States 10. Other causes of hypothyroidism include thyroid nodules, thyroiditis, congenital hypothyroidism, surgical removal of part or all of the thyroid, radiation treatment of the thyroid, and some medicines. Hypothyroidism is also more common in people with autoimmune diseases, such as type 1 diabetes, autoimmune gastric atrophy, and celiac disease, and can occur as part of multiple autoimmune endocrinopathies. Individuals with Down syndrome or Turner’s syndrome have an increased risk of hypothyroidism 11, 12, 2.
Blood tests are used to diagnose hypothyroidism. The first blood test typically done to diagnose hypothyroidism measures the level of thyroid-stimulating hormone (TSH) in the blood. If the TSH (thyroid-stimulating hormone) is high, the test is done again, along with a blood test for the thyroid hormone T4 (thyroxine). If the results show that TSH is high and T4 (thyroxine) is low, then the diagnosis is hypothyroidism. In some cases, the thyroid hormone T3 (tri-iodothyronine) may be measured as well. If the second test shows high TSH but T4 and T3 are in the standard range, then the diagnosis is a condition called subclinical hypothyroidism. Subclinical hypothyroidism usually doesn’t cause any noticeable symptoms.
TSH tests also play an important role in managing hypothyroidism over time. They help your health care provider find and maintain the right dosage of medication for you.
The results of these blood tests can be affected by some medicines or supplements. This includes biotin, a vitamin taken as a stand-alone supplement or as part of a multivitamin. Before you have blood tests done, tell your health care provider about any medicines or supplements you take.
There is no cure for hypothyroidism, and most patients have it for life 13. However, there are exceptions with many patients with viral thyroiditis have their thyroid function return to normal as do some patients with thyroiditis after pregnancy.
If you are diagnosed with hypothyroidism (underactive thyroid), you will most likely be treated with a thyroid hormone medicine to replace the thyroid hormone. This medicine is a synthetic (artificial) version of the hormone thyroxine called levothyroxine (Levoxyl, Synthroid, others). In most cases, you will need to take it for the rest of your life.
The synthetic thyroid hormone works like the T4 hormone naturally produced by the thyroid. Your hypothyroidism can be well-controlled with thyroid hormone medicine, as long as you take the medicine as instructed by your doctor and have regular follow-up blood tests. It can take some time to find the right dose of medicine for you. Your symptoms should disappear and the serious effects of low thyroid hormone should improve. If you keep your hypothyroidism well-controlled, it will not shorten your life span.
If you have mild hypothyroidism, you may not need to have treatment but get regular thyroid stimulating hormone (TSH) tests to monitor thyroid hormone levels.
Who is more likely to develop hypothyroidism?
Women are much more likely than men to develop hypothyroidism. Hypothyroidism is also more common among people older than age 60.
You are more likely to have hypothyroidism if you 14:
- have had a thyroid problem before, such as a goiter
- have had surgery to correct a thyroid problem
- have received radiation treatment to the thyroid, neck, or chest
- have a family history of thyroid disease
- were pregnant in the past 6 months
- have Turner syndrome, a genetic disorder that affects females
- have other health problems, including:
- Sjögren’s syndrome, a disease that causes dry eyes and mouth
- Pernicious anemia, a condition caused by a vitamin B12 deficiency
- Type 1 diabetes
- Rheumatoid arthritis, an autoimmune disease that affects the joints
- Lupus, a chronic inflammatory condition
How much iodine do I need?
The amount of iodine you need each day depends on your age. Average daily recommended amounts are listed below in micrograms (mcg).
Table 1 lists the current Recommended Dietary Allowances (RDA – the average daily level of intake sufficient to meet the nutrient requirements of nearly all [97%–98%] healthy individuals; often used to plan nutritionally adequate diets for individuals) for iodine 15. For infants from birth to 12 months, the Food and Nutrition Board at the Institute of Medicine of the National Academies established an Adequate Intake (AI) for iodine that is equivalent to the mean intake of iodine in healthy, breastfed infants in the United States.
Women need more iodine when they are pregnant because the baby gets iodine from the mother’s diet. If you are pregnant, talk with your health care provider about how much iodine you need. The World Health Organization (WHO), United Nations Children’s Fund (UNICEF), and the International Council for the Control of Iodine Deficiency Disorders (ICCIDD) recommend a slightly higher iodine intake for pregnant women of 250 mcg per day 16, 17.
Table 1. Recommended Dietary Allowances (RDAs) for Iodine
| Age | Male | Female | Pregnancy | Lactation |
| Birth to 6 months | 110 mcg* | 110 mcg* | ||
| 7–12 months | 130 mcg* | 130 mcg* | ||
| 1–3 years | 90 mcg | 90 mcg | ||
| 4–8 years | 90 mcg | 90 mcg | ||
| 9–13 years | 120 mcg | 120 mcg | ||
| 14–18 years | 150 mcg | 150 mcg | 220 mcg | 290 mcg |
| 19+ years | 150 mcg | 150 mcg | 220 mcg | 290 mcg |
Footnote: * Adequate Intake (AI)
What foods are good source for iodine?
Iodine is found naturally in some foods and is also added to salt that is labeled as “iodized”. You can get recommended amounts of iodine by eating a variety of foods, including the following:
- Fish (such as cod and tuna), seaweed, shrimp, and other seafood, which are generally rich in iodine.
- Kelp, nori, kombu, and wakame is one of the best food sources of iodine
- Iodized salt, more than 70 countries, including the United States and Canada, have salt iodization programs. As a result, approximately 70% of households worldwide use iodized salt, ranging from almost 90% of households in North and South America to less than 50% in Europe and the Eastern Mediterranean regions 18. Processed foods, however, such as canned soups, almost never contain iodized salt.
Seaweed (such as kelp, nori, kombu, and wakame) is one of the best food sources of iodine 19. Other good sources include fish and other seafood, as well as eggs (see Table 2). Iodine is also present in human breast milk 15 and infant formulas 20. The U.S. Department of Agriculture (USDA) lists the iodine content of numerous foods and beverages 20.
Dairy products contain iodine. However, the amount of iodine in dairy products varies by whether the cows received iodine feed supplements and whether iodophor sanitizing agents were used to clean the cows and milk-processing equipment 21. For example, an analysis of 44 samples of nonfat milk found a range of 38 to 159 mcg per cup (with an average of 85 mcg/cup used for Table 2) 20. Plant-based beverages used as milk substitutes, such as soy and almond beverages, contain relatively small amounts of iodine.
Most commercially prepared bread contains very little iodine unless the manufacturer has used potassium iodate or calcium iodate as a dough conditioner 22. Manufacturers list dough conditioners as an ingredient on product labels but are not required to include iodine on the Nutrition Facts label 23, even though these conditioners provide a substantial amount of iodine. According to 2019 data from the USDA Branded Food Products Database, approximately 20% of ingredient labels for white bread, whole-wheat bread, hamburger buns, and hot dog buns listed iodate. Pasta is not a source of iodine unless it is prepared in water containing iodized salt because it absorbs some of the iodine 24.
Most fruits and vegetables are poor sources of iodine, and the amounts they contain are affected by the iodine content of the soil, fertilizer use, and irrigation practices 22. This variability affects the iodine content of meat and animal products because of its impact on the iodine content of foods that the animals consume 25. The iodine amounts in different seaweed species also vary greatly. For example, commercially available seaweeds in whole or sheet form have iodine concentrations ranging from 16 mcg/g to 2,984 mcg/g 26. For these reasons, the values for the foods listed in Table 2 are approximate but can be used as a guide for estimating iodine intakes.
Table 2. Iodine Content of Selected Foods
| Food | Micrograms (mcg) per serving | Percent DV* |
|---|---|---|
| Seaweed, nori, dried, 10 g | 232 | 155 |
| Bread, whole-wheat, made with iodate dough conditioner, 1 slice | 198 | 132 |
| Bread, white, enriched, made with iodate dough conditioner, 1 slice | 185 | 123 |
| Cod, baked, 3 ounces | 158 | 106 |
| Yogurt, Greek, plain, nonfat, 1 cup | 116 | 77 |
| Oysters, cooked, 3 ounces | 93 | 62 |
| Milk, nonfat, 1 cup | 85 | 57 |
| Iodized table salt, 1.5 g (approx. ¼ teaspoon) | 76 | 51 |
| Fish sticks, cooked, 3 ounces | 58 | 39 |
| Pasta, enriched, boiled in water with iodized salt, 1 cup | 36 | 24 |
| Egg, hard boiled, 1 large | 26 | 17 |
| Ice cream, chocolate, ½ cup | 21 | 14 |
| Liver, beef, cooked, 3 ounces | 14 | 9 |
| Cheese, cheddar, 1 ounce | 14 | 9 |
| Shrimp, cooked, 3 ounces | 13 | 9 |
| Tuna, canned in water, drained, 3 ounces | 7 | 5 |
| Soy beverage, 1 cup | 7 | 5 |
| Fruit cocktail in light syrup, canned, ½ cup | 6 | 4 |
| Beef, chuck, roasted, 3 ounces | 3 | 2 |
| Chicken breast, roasted, 3 ounces | 2 | 1 |
| Almond beverage, 1 cup | 2 | 1 |
| Apple juice, 1 cup | 1 | 1 |
| Bread, whole-wheat, made without iodate dough conditioner, 1 slice | 1 | 1 |
| Bread, white, enriched, made without iodate dough conditioner, 1 slice | 1 | 1 |
| Raisin bran cereal, 1 cup | 1 | 1 |
| Rice, brown, cooked, ½ cup | 1 | 1 |
| Corn, canned, ½ cup | 1 | 1 |
| Sea salt, non-iodized, 1.5 g (approx. ¼ teaspoon) | <1 | <1 |
| Broccoli, boiled, ½ cup | 0 | 0 |
| Banana, 1 medium | 0 | 0 |
| Lima beans, mature, boiled, ½ cup | 0 | 0 |
| Green peas, frozen, boiled, ½ cup | 0 | 0 |
| Pasta, enriched, boiled in water without iodized salt, 1 cup | 0 | 0 |
Footnotes: *DV = Daily Value. DVs were developed by the U.S. Food and Drug Administration (FDA) to help consumers compare the nutrient contents of products within the context of a total diet. The DV for iodine is 150 mcg for adults and children aged 4 and older. However, the FDA does not require food labels to list iodine content unless a food has been fortified with this nutrient. Foods providing 20% or more of the DV are considered to be high sources of a nutrient, but foods providing lower percentages of the DV also contribute to a healthful diet.
[Source 20, 27, 28, 29 ]Thyroid gland
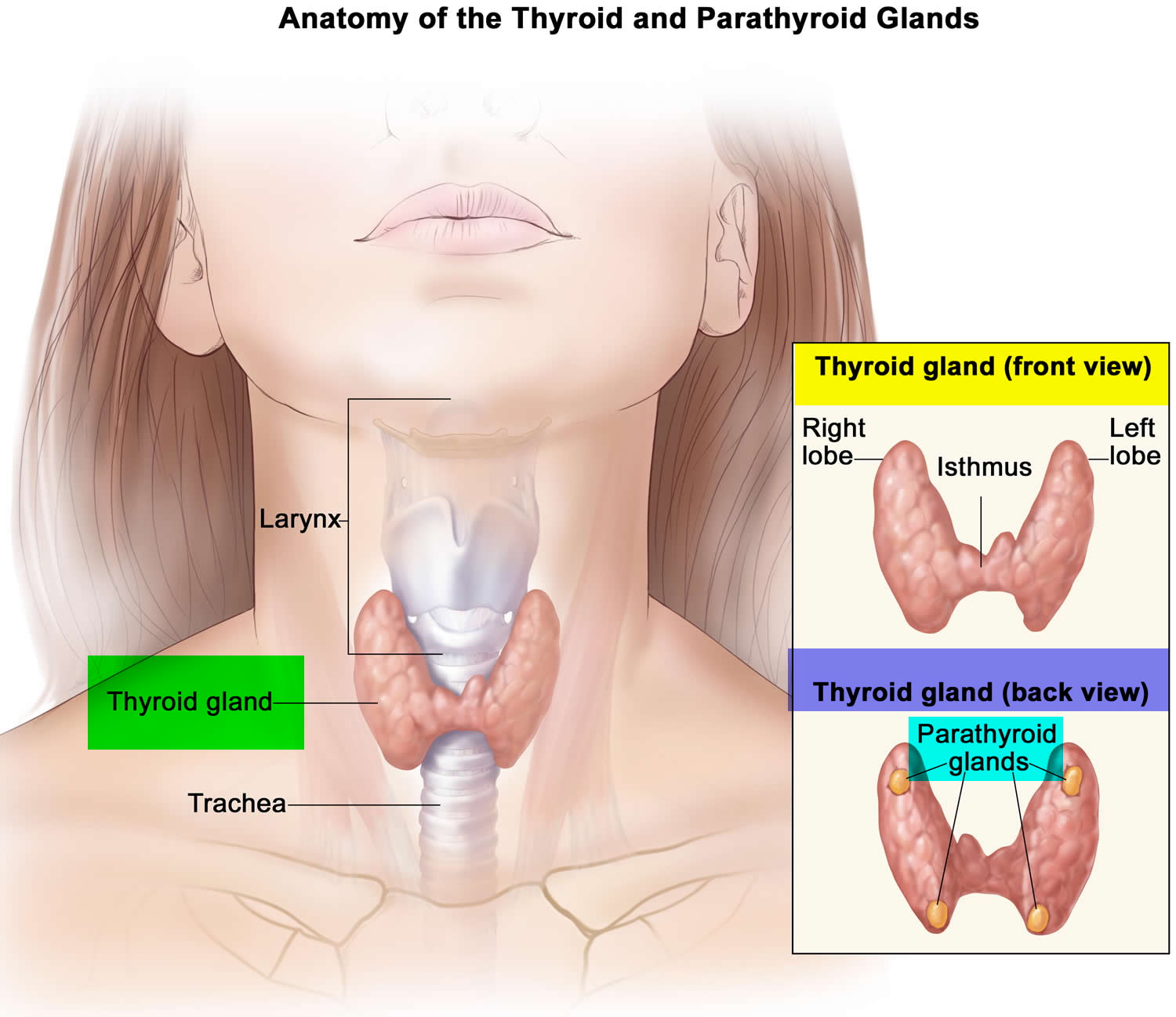
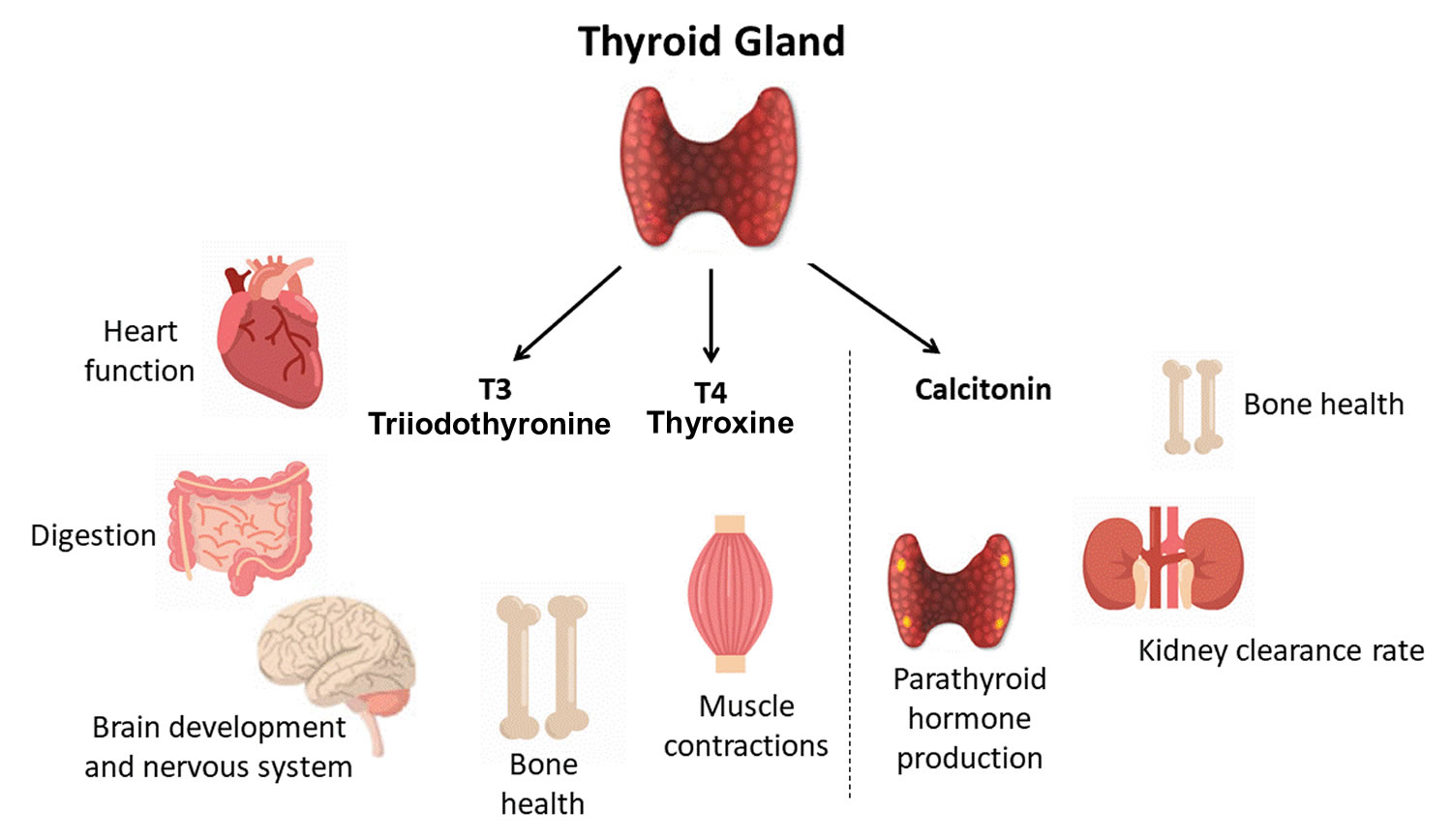
The thyroid gland is the largest adult gland to have a purely endocrine function, weighing about 25-30 g. The thyroid gland is a small butterfly shaped gland with 2 lobes, the right lobe and the left lobe joined by a narrow piece of the thyroid gland called the isthmus, that is located in front of your neck near the base of your throat, beneath the larynx (voice box or Adam’s apple). About 50% of thyroid glands have a small third lobe, called the pyramidal lobe. It extends superiorly from the isthmus. The thyroid gland makes and releases hormones. You can’t usually feel a thyroid gland that is normal.
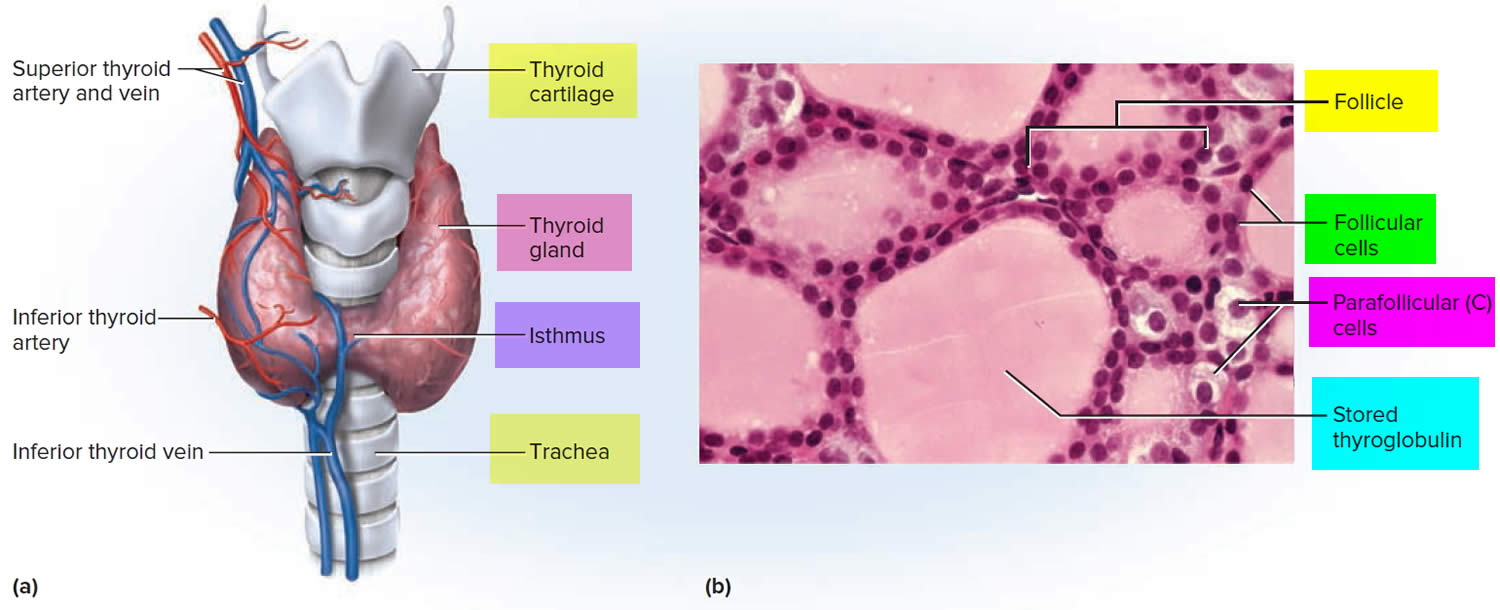
The thyroid gland has 2 main types of cells:
- Follicular cells use iodine from the blood to make thyroid hormones, which help regulate a person’s metabolism. Having too much thyroid hormone (hyperthyroidism) can cause a fast or irregular heartbeat, trouble sleeping, nervousness, hunger, weight loss, and a feeling of being too warm. Having too little thyroid hormone (hypothyroidism) causes a person to slow down, feel tired, and gain weight. The amount of thyroid hormone released by the thyroid gland is regulated by the pituitary gland at the base of the brain, which makes a substance called thyroid-stimulating hormone (TSH) (see Figure 5).
- C cells also called parafollicular cells at the periphery of the follicles that make calcitonin, a hormone that helps control how your body uses calcium. The parafollicular cells (C cells) respond to rising levels of blood calcium by secreting the hormone calcitonin. Calcitonin antagonizes (blocks) parathyroid hormone (PTH) and stimulates osteoblast activity, thus promoting calcium deposition and bone formation. It is important mainly in children, having relatively little effect in adults. Parathyroid hormone is made by parathyroid glands. These sit behind and are attached to the thyroid gland (see Figure 1).
Other, less common cells in the thyroid gland include immune system cells (lymphocytes) and supportive (stromal) cells.
Thyroid hormone is secreted or inhibited in response to fluctuations in metabolic rate. The brain monitors the body’s metabolic rate and stimulates thyroid hormone secretion through the action of thyrotropin-releasing hormone (TRH) and thyroid stimulating hormone (TSH) as depicted in figure 5.
The primary effect of thyroid hormone (TH) is to increase one’s metabolic rate. As a result, it raises oxygen consumption and has a calorigenic effect—it increases heat production. To ensure an adequate blood and oxygen supply to meet this increased metabolic demand, thyroid hormone also raises the breathing (respiratory) rate, heart rate, and strength of the heartbeat. It stimulates the appetite and accelerates the breakdown of carbohydrates, fats, and protein for fuel. Thyroid hormone also promotes alertness and quicker reflexes; growth hormone secretion; growth of the bones, skin, hair, nails, and teeth; and development of the fetal nervous system.
Figure 1. Thyroid gland location and parathyroid gland
Footnotes: Anatomy of the thyroid and parathyroid glands. The thyroid gland lies at the base of the throat near the trachea. It is shaped like a butterfly, with the right lobe and left lobe connected by a thin piece of tissue called the isthmus. The parathyroid glands are four pea-sized organs found in the neck near the thyroid. The thyroid and parathyroid glands make hormones.
Figure 2. Thyroid gland location
Figure 3. Thyroid gland anatomy
Footnote: (a) Gross anatomy, anterior view. (b) Histology, showing the saccular thyroid follicles (the source of thyroid hormone) and nests of C cells (the source of calcitonin).
What does the thyroid gland do?
Formation, storage, and release of thyroid hormones
The thyroid gland is the only endocrine gland that stores its secretory product in large quantities—normally about a 100-day supply. Synthesis and secretion of triiodothyronine (T3) and thyroxine or tetraiodothyronine (T4) occurs as follows:
- Iodide trapping. Thyroid follicular cells trap iodide ions (I −) by actively transporting them from the blood into the cytosol. As a result, the thyroid gland normally contains most of the iodide in the body.
- Synthesis of thyroglobulin. While the follicular cells are trapping I −, they are also synthesizing thyroglobulin (TGB), a large glycoprotein that is produced in the rough endoplasmic reticulum, modified in the Golgi complex, and packaged into secretory vesicles. The vesicles then undergo exocytosis, which releases thyroglobulin into the lumen of the follicle.
- Oxidation of iodide. Some of the amino acids in thyroglobulin are tyrosines that will become iodinated. However, negatively charged iodide (I −) ions cannot bind to tyrosine until they undergo oxidation (removal of electrons) to iodine: I −→ I. As the iodide ions are being oxidized, they pass through the membrane into the lumen of the follicle.
- Iodination of tyrosine. As iodine atoms (I) form, they react with tyrosines that are part of thyroglobulin molecules. Binding of one iodine atom yields monoiodotyrosine (T1), and a second iodination produces diiodotyrosine (T2). The thyroglobulin with attached iodine atoms, a sticky material that accumulates and is stored in the lumen of the thyroid follicle, is termed colloid.
- Coupling of monoiodotyrosine (T1) and diiodotyrosine (T2). During the last step in the synthesis of thyroid hormone, two diiodotyrosine (T2) molecules join to form thyroxine (T4) or one T1 and one T2 join to form triiodothyronine (T3).
- Pinocytosis and digestion of colloid. Droplets of colloid reenter follicular cells by pinocytosis and merge with lysosomes. Digestive enzymes in the lysosomes break down thyroglobulin, cleaving off molecules of triiodothyronine (T3) and thyroxine (T4).
- Secretion of thyroid hormones. Because T3 and T4 are lipid soluble, they diffuse through the plasma membrane into interstitial fluid and then into the blood. T4 normally is secreted in greater quantity than T3, but T3 is several times more potent. Moreover, after T4 enters a body cell, most of it is converted to T3 by removal of one iodine.
- Transport thyroid hormones in the blood. More than 99% of both the T3 and the T4 combine with transport proteins in the blood, mainly thyroxine binding globulin (TBG).
Figure 4. Thyroid hormones
Actions of thyroid hormones
Because most body cells have receptors for thyroid hormones, triiodothyronine (T3) and thyroxine (T4) affect tissues throughout the body. Thyroid hormones act on their target cells mainly by inducing gene transcription and protein synthesis. The newly formed proteins in turn carry out the cellular response.
Functions of thyroid hormones include the following:
- Increase basal metabolic rate. Thyroid hormones raise the basal metabolic rate (BMR), the rate of energy expenditure under standard or basal conditions (awake, at rest, and fasting). When basal metabolic rate increases, cellular metabolism of carbohydrates, lipids, and proteins increases. Thyroid hormones increase BMR in several ways: (1) They stimulate synthesis of additional Na+/K+ ATPases, which use large amounts of ATP to continually eject sodium ions (Na+) from cytosol into extracellular fluid and potassium ions (K+) from extracellular fluid into cytosol; (2) they increase the concentrations of enzymes involved in cellular respiration, which increases the breakdown of organic fuels and ATP production; and (3) they increase the number and activity of mitochondria in cells, which also increases ATP production. As cells produce and use more ATP, basal metabolic rate increases, more heat is given off and body temperature rises, a phenomenon called the calorigenic effect. In this way, thyroid hormones play an important role in the maintenance of normal body temperature. Normal mammals can survive in freezing temperatures, but those whose thyroid glands have been removed cannot.
- Enhance actions of catechlolamines. Thyroid hormones have permissive effects on the catecholamines (epinephrine and norepinephrine) because they up-regulate β-adrenergic receptors. Catecholamines bind to β-adrenergic receptors, promoting sympathetic responses. Therefore, symptoms of excess levels of thyroid hormone include increased heart rate, more forceful heartbeats, and increased blood pressure.
- Regulate development and growth of nervous tissue and bones. Thyroid hormones are necessary for the development of the nervous system: They promote synapse formation, myelin production, and growth of dendrites. Thyroid hormones are also required for growth of the skeletal system: They promote formation of ossification centers in developing bones, synthesis of many bone proteins, and secretion of growth hormone (GH) and insulin-like growth factors (IGFs). Deficiency of thyroid hormones during fetal development, infancy, or childhood causes severe mental retardation and stunted bone growth.
Control of thyroid hormone secretion
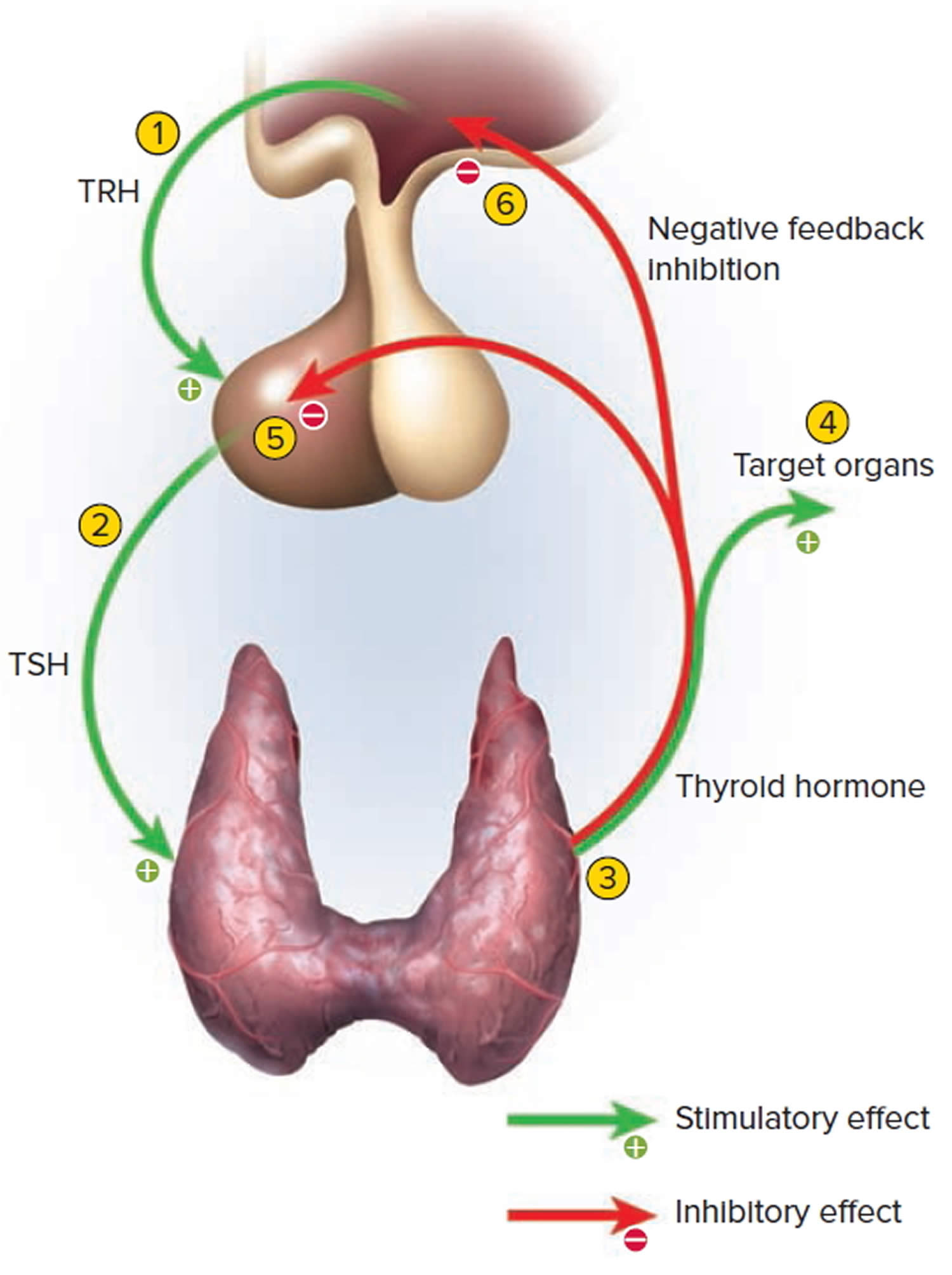
Thyrotropin-releasing hormone (TRH) from the hypothalamus and thyroid-stimulating hormone (TSH) from the anterior pituitary stimulate secretion of thyroid hormones, as shown in Figure 5:
- Low blood levels of T3 and T4 or low metabolic rate stimulate the hypothalamus to secrete thyrotropin-releasing hormone (TRH).
- Thyrotropin-releasing hormone (TRH) enters the hypothalamic–hypophyseal portal system and flows to the anterior pituitary, where it stimulates thyrotrophs to secrete thyroid stimulating hormone (TSH).
- Thyroid stimulating hormone (TSH) stimulates virtually all aspects of thyroid follicular cell activity, including iodide trapping, hormone synthesis and secretion, and growth of the follicular cells.
- The thyroid follicular cells release T3 and T4 into the blood until the metabolic rate returns to normal.
- An elevated level of T3 inhibits release of TRH and TSH (negative feedback inhibition).
Conditions that increase ATP demand—a cold environment, hypoglycemia, high altitude, and pregnancy—increase the secretion of the thyroid hormones.
Figure 5. Control of thyroid hormone secretion
Footnote: Negative Feedback Inhibition of the Anterior Pituitary Gland by the Thyroid Gland
Control of calcium balance
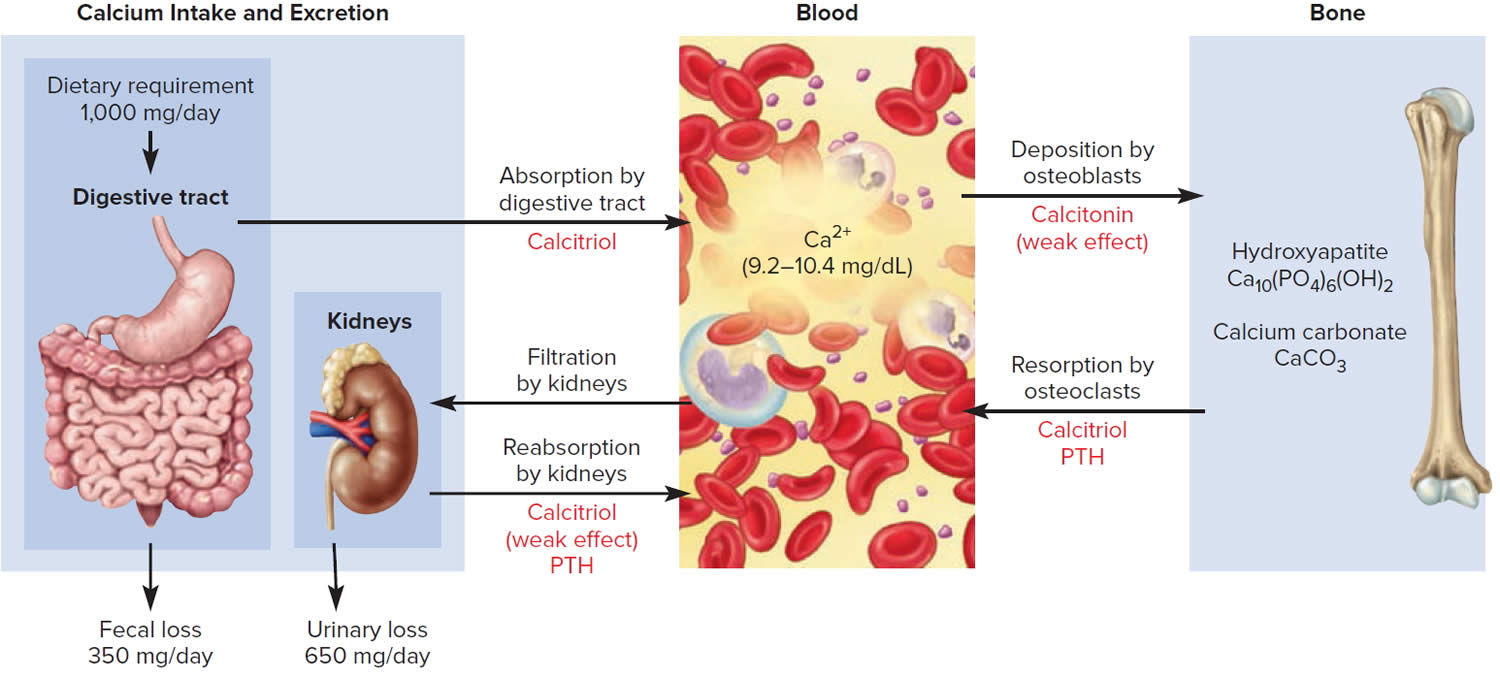
The hormone produced by the parafollicular cells of the thyroid gland is calcitonin. Calcitonin can decrease the level of calcium in the blood by inhibiting the action of osteoclasts, the cells that break down bone extracellular matrix. The secretion of calcitonin is controlled by a negative feedback system (see Figure 7).
Calcitonin is produced by C cells (clear cells) of the thyroid gland. It is secreted when the blood calcium concentration rises too high, and it lowers the concentration by two principal mechanisms:
- Osteoclast inhibition. Within 15 minutes after it is secreted, calcitonin reduces osteoclast activity by as much as 70%, so osteoclasts liberate less calcium from the skeleton.
- Osteoblast stimulation. Within an hour, calcitonin increases the number and activity of osteoblasts, which deposit calcium into the skeleton.
Calcitonin plays an important role in children but has only a weak effect in most adults. The osteoclasts of children are highly active in skeletal remodeling and release 5 g or more of calcium into the blood each day. By inhibiting this activity, calcitonin can significantly lower the blood calcium level in children. In adults, however, the osteoclasts release only about 0.8 g of calcium per day. Calcitonin cannot change adult blood calcium very much by suppressing this lesser contribution. Calcitonin deficiency is not known to cause any adult disease. Calcitonin may, however, inhibit bone loss in pregnant and lactating women. Miacalcin, a calcitonin extract derived from salmon that is 10 times more potent than human calcitonin, is prescribed to treat osteoporosis.
Figure 6. Hormonal control of calcium balance
Footnote: The central panel represents the blood reservoir of calcium and shows its normal (safe) range. Calcitriol and Parathyroid Hormone (PTH) regulate calcium exchanges between the blood and the small intestine and kidneys (left). Calcitonin, calcitriol, and Parathyroid Hormone (PTH) regulate calcium exchanges between blood and bone (right).
Causes of hypothyroidism
There can be many reasons why the cells in the thyroid gland can’t make enough thyroid hormone. Here are the major causes, from the most to the least common.
Autoimmune thyroiditis: In some people’s bodies, the immune system that protects the body from invading infections can mistaken thyroid gland cells and their enzymes for invaders and can attack them. The body’s immune system may produce a reaction in the thyroid gland that results in hypothyroidism and, most often, a goiter (enlargement of the thyroid). This is more common in women than men. Autoimmune thyroiditis can begin suddenly or it can develop slowly over years. The most common forms are Hashimoto’s thyroiditis and atrophic thyroiditis. Other autoimmune diseases may be associated with this disorder, and additional family members may also be affected.
Central or pituitary hypothyroidism: Thyroid-stimulating hormone (TSH) is produced by the pituitary gland, which is located behind the nose at the base of the brain. The pituitary, the “master gland,” tells the thyroid how much hormone to make. When the pituitary is damaged by a tumor, radiation, or surgery, it may no longer be able to give the thyroid instructions, and the thyroid may stop making enough hormone. Any destructive disease of the pituitary gland or hypothalamus, which sits just above the pituitary gland, may cause damage to the cells that secrete TSH 30, 31. Other causes of central hypothyroidism include pituitary or hypothalamic dysfunction due to head trauma, pituitary apoplexy, Sheehan’s syndrome, surgery, radiotherapy, genetic, and infiltrative disease. Several drugs are known to affect the hypothalamic–pituitary–thyroid axis 32, 33. Central or pituitary hypothyroidism is a very rare cause of hypothyroidism.
Congenital hypothyroidism: An infant may be born with an inadequate amount of thyroid tissue or an enzyme defect that does not allow normal thyroid hormone production. In most cases, the reason the thyroid gland didn’t develop properly is not clear. But some children have an inherited form of a thyroid disorder. Often, infants born with hypothyroidism don’t have noticeable symptoms at first. That’s one reason why most states require newborn thyroid screening. If this condition is not treated promptly, physical stunting and/or mental damage (cretinism) may develop.
Medications: A number of medicines may lead to hypothyroidism. Lithium (a bipolar disorder medicine) 34. In a large population-based cohort study,38 6% of patients needed levothyroxine therapy within 18 months of starting lithium treatment 35. High doses of iodine and amiodarone (an iodine-containing drug) — an antiarrhythmic agent used for various types of cardiac arrhythmias can also cause hypothyroidism. About 14% of patients treated with amiodarone develop hypothyroidism 36. Interferon alpha (a cancer medicine) and interleukin-2 (a kidney cancer medicine)– can prevent the thyroid gland from being able to make thyroid hormone normally. These drugs are most likely to trigger hypothyroidism in patients who have a genetic tendency to autoimmune thyroid disease. If you’re taking medicine, ask your doctor about its effect on the thyroid gland.
Postpartum thyroiditis: Five percent to 10 percent of women develop mild to moderate hyperthyroidism within several months of giving birth. Hyperthyroidism in this condition usually lasts for approximately one to two months. It is often followed by several months of hypothyroidism, but most women will eventually recover normal thyroid function. In some cases, however, the thyroid gland does not heal, so the hypothyroidism becomes permanent and requires lifelong thyroid hormone replacement. This condition may occur again with subsequent pregnancies.
Pregnancy. Some people develop hypothyroidism during or after pregnancy. If hypothyroidism happens during pregnancy and isn’t treated, it raises the risk of pregnancy loss, premature delivery and preeclampsia. Preeclampsia causes a significant rise in blood pressure during the last three months of pregnancy. Hypothyroidism also can seriously affect the developing fetus.
Radioactive iodine treatment: Hypothyroidism frequently develops as a desired therapeutic goal after the use of radioactive iodine treatment for hyperthyroidism 37, 38. Some people with Graves’ disease, nodular goiter, or thyroid cancer are treated with radioactive iodine (I-131) for the purpose of destroying their thyroid gland. Patients with Hodgkin’s disease, lymphoma, or cancers of the head or neck are treated with radiation. All these patients can lose part or all of their thyroid function.
Silent Thyroiditis: Transient (temporary) hyperthyroidism can be caused by silent thyroiditis, a condition which appears to be the same as postpartum thyroiditis but not related to pregnancy. It is not accompanied by a painful thyroid gland.
Subacute thyroiditis: This condition may follow a viral infection and is characterized by painful thyroid gland enlargement and inflammation, which results in the release of large amounts of thyroid hormone into the blood. Fortunately, this condition usually resolves spontaneously. The thyroid usually heals itself over several months, but often not before a temporary period of hypothyroidism occurs.
Thyroid surgery: Hypothyroidism may be related to surgery on the thyroid gland, especially if most of the thyroid has been removed. Some people with thyroid nodules, thyroid cancer, or Graves’ disease need to have part or all of their thyroid removed. If the whole thyroid is removed, people will definitely become hypothyroid. If part of the gland is left, it may be able to make enough thyroid hormone to keep blood levels normal.
Too much or too little iodine. The thyroid gland must have iodine to make thyroid hormone. Iodine comes into the body in food and travels through the blood to the thyroid. Iodine is found mainly in seafood, seaweed, plants grown in iodine-rich soil and iodized salt. Keeping thyroid hormone production in balance requires the right amount of iodine. Too little iodine can lead to hypothyroidism. Too much iodine can make hypothyroidism worse in people who already have the condition. In some parts of the world, it’s common for people not to get enough iodine in their diets. The addition of iodine to table salt has almost eliminated this problem in the United States.
Rare disorders that infiltrate the thyroid. In a few people, diseases deposit abnormal substances in the thyroid and impair its ability to function. For example, amyloidosis can deposit amyloid protein, sarcoidosis can deposit granulomas, and hemochromatosis can deposit iron.
Consumptive hypothyroidism. Consumptive hypothyroidism is caused by aberrant expression of the deiodinase 3 enzyme (which inactivates thyroid hormone) in tumor tissues 2. Although very rare, overexpression of deiodinase 3 enzyme can induce severe hypothyroidism. Elevated concentration of deiodinase 3 was first described in a newborn baby with infantile hepatic hemangiomatosis, but can also occur in patients with vascular and fibrotic tumors and gastrointestinal stromal tumors 39. Patients with rare genetic syndromes that lead to a reduced sensitivity to thyroid hormone usually have normal TSH concentrations, but can also present with tissue-specific hypothyroidism 40.
Risk factors for developing hypothyroidism
Although anyone can develop hypothyroidism, you’re at an increased risk if you:
- Are a woman.
- Have a family history of thyroid disease.
- Have an autoimmune disease, such as type 1 diabetes or celiac disease.
- Have received treatment for hyperthyroidism.
- Received radiation to your neck or upper chest.
- Have had thyroid surgery.
Hypothyroidism signs and symptoms
In its earliest stage, hypothyroidism may cause few symptoms, since the body has the ability to partially compensate for a failing thyroid gland by increasing the stimulation to it, much like pressing down on the accelerator when climbing a hill to keep the car going the same speed. As thyroid hormone production decreases and the body’s metabolism slows, a variety of features may result.
Hypothyroidism symptoms may include:
- Pervasive fatigue or tiredness
- More sensitivity to cold
- Drowsiness
- Forgetfulness or memory problems
- Difficulty with learning
- Dry, thinning hair and brittle nails
- Coarse hair and skin
- Dry, itchy skin
- Puffy face
- Constipation
- Muscle aches, tenderness and stiffness
- Muscle weakness
- Weight gain and fluid retention
- Heavy and/or irregular menstrual periods and fertility problems
- Increased frequency of miscarriages
- Increased sensitivity to many medications
- Weight gain
- A puffy face
- Hoarse voice
- Joint and muscle pain
- Decreased sweating
- Depression
- Slowed heart rate (bradycardia).
- Goiter (enlarged thyroid).
Hypothyroidism can contribute to high cholesterol, so people with high cholesterol should be tested for hypothyroidism. Rarely, severe, untreated hypothyroidism may lead to myxedema coma, an extreme form of hypothyroidism in which the body’s functions slow to the point that it becomes life threatening. Myxedema coma requires immediate medical treatment.
Hypothyroidism in infants
Anyone can get hypothyroidism, including infants. Most babies born without a thyroid gland or with a gland that doesn’t work correctly don’t have symptoms right away. But if hypothyroidism isn’t diagnosed and treated, symptoms start to appear. They may include:
- Feeding problems.
- Poor growth.
- Poor weight gain.
- Yellowing of the skin and the whites of the eyes, a condition called jaundice.
- Constipation.
- Poor muscle tone.
- Dry skin.
- Hoarse crying.
- Enlarged tongue.
- A soft swelling or bulge near the belly button, a condition called umbilical hernia.
When hypothyroidism in infants isn’t treated, even mild cases can lead to severe physical and mental development problems.
Hypothyroidism in children and teens
In general, children and teens with hypothyroidism have symptoms similar to those in adults. But they also may have:
- Poor growth that leads to short stature.
- Delayed development of permanent teeth.
- Delayed puberty.
- Poor mental development.
Hypothyroidism during pregnancy
Hypothyroidism that isn’t treated can affect both the mother and the baby. However, thyroid medicines can help prevent problems and are safe to take during pregnancy.
Hypothyroidism in pregnancy is usually caused by Hashimoto’s disease and occurs in three to five out of every 1,000 pregnancies. Hashimoto’s disease is a form of chronic inflammation of the thyroid gland.
Like Graves’ disease, Hashimoto’s disease is an autoimmune disorder. In Hashimoto’s disease, the immune system attacks the thyroid, causing inflammation and interfering with its ability to produce thyroid hormones.
Hypothyroidism in pregnancy can also result from existing hypothyroidism that is inadequately treated or from prior destruction or removal of the thyroid as a treatment for hyperthyroidism.
How does hypothyroidism affect the mother and baby?
Some of the same problems caused by hyperthyroidism can occur with hypothyroidism.
Uncontrolled hypothyroidism during pregnancy can lead to:
- preeclampsia
- anemia—too few red blood cells in the body, which prevents the body from getting enough oxygen
- miscarriage
- low birth weight
- stillbirth
- congestive heart failure, rarely
Because thyroid hormones are crucial to fetal brain and nervous system development, uncontrolled hypothyroidism—especially during the first trimester—can affect the baby’s growth and brain development.
Dietary Supplements for Pregnant Women with Hypothyroidism
Because the thyroid uses iodine to make thyroid hormone, iodine is an important mineral for a mother during pregnancy. During pregnancy, the baby gets iodine from the mother’s diet. Women need more iodine when they are pregnant—about 250 micrograms a day. In the United States, about 7 percent of pregnant women may not get enough iodine in their diet or through prenatal vitamins 41. Choosing iodized salt—salt supplemented with iodine—over plain salt and prenatal vitamins containing iodine will ensure this need is met.
However, people with autoimmune thyroid disease may be sensitive to harmful side effects from iodine. Taking iodine drops or eating foods containing large amounts of iodine—such as seaweed, dulse, or kelp—may cause or worsen hyperthyroidism and hypothyroidism.
To help ensure coordinated and safe care, pregnant women should discuss their use of dietary supplements with their health care provider.
Hypothyroidism complications
Thyroid hormones are essential for the healthy function of many body systems. Hypothyroidism that isn’t treated can lead to other health problems, including:
- Goiter. Hypothyroidism may cause the thyroid gland to become larger. This condition is called a goiter. A large goiter may cause problems with swallowing or breathing.
- Heart problems. Hypothyroidism can lead to a higher risk of heart disease, irregular heartbeats and heart failure. That’s mainly because people with an underactive thyroid tend to develop high levels of low-density lipoprotein (LDL) cholesterol — the “bad” cholesterol.
- Peripheral neuropathy. Hypothyroidism that goes without treatment for a long time can damage the peripheral nerves. These are the nerves that carry information from the brain and spinal cord to the rest of the body. Peripheral neuropathy may cause pain, numbness and tingling in the arms and legs.
- Infertility. Low levels of thyroid hormone can interfere with ovulation, which can limit fertility. Some of the causes of hypothyroidism, such as autoimmune disorders, also can harm fertility.
- Mental health issues. Depression or other mental health disorders may occur early in Hashimoto’s disease and may become more severe over time.
- Sexual and reproductive dysfunction. In women, hypothyroidism can result in a reduced sexual desire (libido), an inability to ovulate, and irregular and excessive menstrual bleeding. Men with hypothyroidism may have a reduced libido, erectile dysfunction and a lowered sperm count.
- Poor pregnancy outcomes. Hypothyroidism during pregnancy may increase the risk of a miscarriage or preterm birth. Babies born to women with untreated hypothyroidism are at risk for decreased intellectual abilities, autism, speech delays and other developmental disorders.
- Birth defects. Babies born to people with untreated thyroid disease may have a higher risk of birth defects compared with babies born to mothers who do not have thyroid disease. Infants with hypothyroidism present at birth that goes untreated are at risk of serious physical and mental development problems. But if the condition is diagnosed within the first few months of life, the chances of typical development are excellent.
- Myxedema coma. This rare, life-threatening condition can happen when hypothyroidism goes without treatment for a long time 42. A myxedema coma may be triggered by sedatives, infection or other stress on the body. Its symptoms include intense cold intolerance and drowsiness, followed by an extreme lack of energy and then unconsciousness. Myxedema coma requires emergency medical treatment.
Hypothyroidism diagnosis
The symptoms of hypothyroidism can be different from person to person. And they often look like symptoms of other health problems. Because of that, a diagnosis of hypothyroidism doesn’t rely on symptoms alone. It’s usually based on the results of blood tests. A primary care physician may make the diagnosis of hypothyroidism, but assistance is often needed from an endocrinologist, a physician who is a specialist in thyroid diseases.
The first blood test typically done to diagnose hypothyroidism measures the level of thyroid-stimulating hormone (TSH) in the blood. If the thyroid-stimulating hormone (TSH) is high, the test is done again, along with a blood test for the thyroid hormone thyroxine (T4). If the results show that TSH (thyroid-stimulating hormone) is high and T4 (thyroxine) is low, then the diagnosis is hypothyroidism. In some cases, the thyroid hormone tri-iodothyronine (T3) may be measured as well.
If the second test shows high thyroid-stimulating hormone (TSH) but thyroxine (T4) and tri-iodothyronine (T3) are in the standard range, then the diagnosis is a condition called subclinical hypothyroidism. It usually doesn’t cause any noticeable symptoms.
TSH (thyroid-stimulating hormone) tests also play an important role in managing hypothyroidism over time. They help your health care provider find and maintain the right dosage of medication for you.
The results of these blood tests can be affected by some medicines or supplements. This includes biotin, a vitamin taken as a stand-alone supplement or as part of a multivitamin. Before you have blood tests done, tell your health care provider about any medicines or supplements you take.
The correct diagnosis of hypothyroidism depends on the following:
Medical and family history. You should tell your doctor:
- about changes in your health that suggest that your body is slowing down;
- if you’ve ever had thyroid surgery;
- if you’ve ever had radiation to your neck to treat cancer;
- if you’re taking any of the medicines that can cause hypothyroidism— amiodarone, lithium, interferon alpha, interleukin-2, and maybe thalidomide;
- whether any of your family members have thyroid disease..
Physical exam. The doctor will check your thyroid gland and look for changes such as dry skin, swelling, slower reflexes, and a slower heart rate.
Blood tests. There are two blood tests that are used in the diagnosis of hypothyroidism.
Thyroid blood tests
Thyroid stimulating hormone (TSH) or thyrotropin. An increased thyroid stimulating hormone (TSH) level in the blood is the most accurate indicator of primary (non-pituitary) hypothyroidism. Production of this pituitary hormone is increased when the thyroid gland even slightly under produces thyroid hormone.
Estimates of free thyroxine (fT4). The active thyroid hormone in the blood. It is important to note that there is a range of free thyroxine levels in the blood of normal people, similar to the range for height, and that a value of free thyroxine that is “within normal limits” for the general population may not be appropriate for a particular individual.
Thyroxine (T4) tests. Most of the thyroxine (T4) in the blood is attached to a protein called thyroxine-binding globulin (TBG). The “bound” T4 can’t get into body cells. Only about 1%–2% of T4 in the blood is unattached (“free”) and can get into cells. The free T4 and the free T4 index are both simple blood tests that measure how much unattached T4 is in the blood and available to get into cells.
Thyroid antibodies. There are different types of thyroid antibodies. Some antibodies destroy thyroid tissue. Others cause the thyroid to make too much of certain thyroid hormones. A thyroid antibodies test usually measures one or more of the following types of antibodies:
- Thyroid peroxidase antibodies (TPO). These antibodies can be a sign of:
- Hashimoto disease, also known as Hashimoto thyroiditis. This is an autoimmune disease and the most common cause of hypothyroidism. Hypothyroidism is a condition in which the thyroid doesn’t make enough thyroid hormones.
- Graves’ disease. This is also an autoimmune disease and the most common cause of hyperthyroidism. Hyperthyroidism is a condition in which the thyroid makes too much of certain thyroid hormones.
- Thyroglobulin antibodies (Tg). These antibodies can also be a sign of Hashimoto disease. Most people with Hashimoto disease have high levels of both anti-thyroglobulin (anti-Tg) and anti-TPO (anti-TPO) antibodies.
- Thyroid-stimulating hormone (TSH) receptor. These antibodies can be a sign of Grave’s disease.
Circulating antibody to thyroid peroxidase (anti-TPO) are found in about 90% of Hashimoto’s disease patients. Anti-thyroglobulin antibodies (anti-Tg) are less sensitive (positive in about 60–80% of patients) and less specific than antibody to thyroid peroxidase (anti-TPO) 43, 44, 45.
You probably won’t need other tests to confirm you have Hashimoto’s disease. However, if your doctor suspects Hashimoto’s disease but you don’t have antithyroid antibodies in your blood, you may have an ultrasound of your thyroid. The ultrasound images can show the size of your thyroid and other features of Hashimoto’s disease. The ultrasound also can rule out other causes of an enlarged thyroid, such as thyroid nodules—small lumps in the thyroid gland.
Hypothyroidism treatment
Treatment for hypothyroidism usually includes taking the synthetic thyroid hormone medicine levothyroxine (Levo-T, Synthroid, others) every day. This medicine is taken by mouth. It returns hormone levels to a healthy range, eliminating symptoms of hypothyroidism. You’ll likely start to feel better one or two weeks after you begin treatment. Treatment with levothyroxine likely will be lifelong. Because the dosage you need may change, your health care provider may check your TSH level every year.
An experienced physician can prescribe the correct form and dosage to return the thyroid balance to normal. Older patients who may have underlying heart disease are usually started at a low dose and gradually increased while younger healthy patients can be started on full replacement doses at once. Thyroid hormone acts very slowly in some parts of the body, so it may take several months after treatment for some features to improve.
Levothyroxine tablets come in 12 different strengths, and it is essential to take them in a consistent manner every day. A dose of thyroid hormone that is too low may fail to prevent enlargement of the thyroid gland, allow symptoms of hypothyroidism to persist, and be associated with increased serum cholesterol levels, which may increase the risk for atherosclerosis and heart disease. A dose that is too high can cause symptoms of hyperthyroidism, create excessive strain on the heart, and lead to an increased risk of developing osteoporosis.
Levothyroxine is absorbed in your small intestine and intake is advised in the morning 30–60 minutes before breakfast. Intake before bedtime (2–3 hours after last meal) might improve absorption and can be considered to increase compliance 46. Gastrointestinal conditions that reduce levothyroxine absorption include Helicobacter pylori gastritis, celiac disease, and autoimmune atrophic gastritis. Results from some studies 47, 48 suggest that liquid and soft gel formulations of levothyroxine do not depend on gastric pH for absorption, and could provide a solution for patients with difficulties ingesting levothyroxine 30–60 minutes before breakfast. In a double-blind randomised crossover trial of liquid thyroxine in 77 treatment-naive patients with hypothyroidism, no significant differences in thyroid function tests were seen when the liquid preparation was ingested at breakfast or 30 minute before breakfast 49. However, no studies have compared liquid gel formulations of levothyroxine with solid formulations in relation to clinical outcomes.
It is extremely important that women planning to become pregnant are kept well adjusted, since hypothyroidism can affect the development of the baby. During pregnancy, thyroid hormone replacement requirements often change, so more frequent monitoring is necessary. Various medications and supplements (particularly iron) may affect the absorption of thyroid hormone; therefore, the levels may need more frequent monitoring during illness or change in medication and supplements.
Thyroid hormone is critical for normal brain development in babies. Infants requiring thyroid hormone therapy should NOT be treated with purchased liquid suspensions, since the active hormone may deteriorate once dissolved and the baby could receive less thyroid hormone than necessary. Instead, infants with hypothyroidism should receive their thyroid hormone by crushing a single tablet daily of the correct dose and suspending it in one teaspoon of liquid and administering it properly.
Since most cases of hypothyroidism are permanent and often progressive, it is usually necessary to treat this condition throughout one’s lifetime. Periodic monitoring of TSH levels and clinical status are necessary to ensure that the proper dose is being given, since medication doses may have to be adjusted from time to time. Optimal adjustment of thyroid hormone dosage is critical, since the body is very sensitive to even small changes in thyroid hormone levels.
Appropriate management of hypothyroidism requires continued care by a physician experienced in the treatment of this condition.
Table 3. Reasons for failure to reach levothyroxine treatment goals and recommendations
| Recommendations | |
|---|---|
| Elevated TSH with or without (persistent) symptoms | |
| Inadequate dose | Consider higher doses, especially in patients with no remaining functional thyroid capacity (eg, after total thyroidectomy or radioablation therapy for Graves’ disease) |
| Simultaneous intake of levothyroxine with food can impair absorption | Intake 30–60 min before breakfast or at bedtime (2–3 h after evening meal); discuss patient preference |
| Some drugs can affect levothyroxine absorption—eg, calcium carbonate*, ferrous sulfate*, proton pump inhibitors, aluminium containing antacid, sucralfate, and orlistat | Separate intake of levothyroxine from interfering medications and supplements (eg,4 h) |
| Some drugs can affect levothyroxine availability and requirement— eg, oestrogens, androgens, sertraline, phenobartbital†, carbamazepine, phenytoin, and rifampicin | Monitor TSH at initiation and adjust levothyroxine dose if required |
| Malabsorption due to gastrointestinal disease and conditions | Helicobacter pylori gastritis, coeliac disease, autoimmune atrophic gastritis, and diabetic gastropathy should be considered and treated if possible |
| Non-adherence | Common cause, but should be suspected after other causes have been excluded; consider thyroxine absorption test |
| Normal TSH and (persistent) symptoms | |
| Concurrent (autoimmune) disease or causes | Autoimmune atrophic gastritis with pernicious anaemia, Addison’s disease, diabetes, and rheumatoid arthritis could be considered |
| Inadequate thyroid hormone concentrations at the tissue level | Acknowledgment of the patient’s symptoms; check if the patient feels better at a different TSH concentration in the normal range (ie, an individual set-point); a trial of levothyroxine–liothyronine combination therapy can be considered in adherent patients with long-lasting steady state of TSH in serum |
| Low TSH with or without (persistent) symptoms | |
| Overtreatment due to high doses | Consider lower doses in elderly individuals and patients with subclinical hypothyroidism; ask if the patient takes any over-the-counter preparations that might contain thyroid hormone |
| Medical conditions—certain drugs (eg, metformin) and weight loss can decrease TSH concentrations | Consider lower doses |
Footnotes:
* Evidence from prospective trials.
† Antiepileptic drugs accelerate thyroxine and tri-iodothyronine conjugation but serum concentrations of TSH do not necessarily increase.
Abbreviation: TSH= thyroid-stimulating hormone
[Source 2 ]Finding the right Levothyroxine dosage
The optimal daily dose of levothyroxine in overt hypothyroidism is 1·5—1·8 μg per kg of bodyweight 1, 50, 51. In patients with coronary artery disease, the starting levothyroxine dose is generally 12·5—25·0 μg per day and should be gradually increased on the basis of symptoms and TSH concentrations 1. This regimen is often preferred in the elderly, especially in patients with many co-morbidities 1, (Pearce SH, Brabant G, Duntas LH, Monzani F, Peeters RP, Razvi S, Wemeau JL. 2013 ETA Guideline: Management of Subclinical Hypothyroidism. Eur Thyroid J. 2013 Dec;2(4):215-28. doi: 10.1159/000356507)). In younger patients without comorbidities, the full dose can usually be given from the start with adequate monitoring to avoid overtreatment 2.
To find the right dosage of levothyroxine for you, your health care provider checks your level of TSH about 6 to 8 weeks after you start taking the medicine. You may need another blood test to check TSH again six months later.
Too much levothyroxine can cause side effects, such as:
- Tiredness.
- Increased appetite.
- Sleep problems.
- Shakiness.
- Pounding of the heart, sometimes called heart palpitations.
Levothyroxine typically causes no side effects when used in the correct dose. If you change brands of the medicine, tell your health care provider, as the dosage may need to change.
If you have coronary artery disease or severe hypothyroidism, your health care provider may start treatment with a smaller amount of medicine and then slowly increase the dosage. This allows your heart to adjust to the rise in your body’s metabolism.
Women of childbearing age
Because of physiological changes during pregnancy, an increase in levothyroxine dose is required to maintain normal thyroid function 52. Therefore, women of childbearing age with levothyroxine-treated hypothyroidism should be informed to increase their dose by 30% once pregnant and directly contact their physician for further guidance 53.
Taking levothyroxine correctly
Levothyroxine is best taken on an empty stomach at the same time every day. Ideally, you take the hormone in the morning, and then wait 30 to 60 minutes before you eat or take other medicine. If you take the medicine at bedtime, wait to take it until at least four hours after your last meal or snack.
Don’t skip doses or stop taking the medicine because you feel better. If you do, it’s likely that the symptoms of hypothyroidism will slowly return. If you miss a dose of levothyroxine, take two pills the next day.
Some medicines, supplements and even some foods may affect your body’s ability to absorb levothyroxine. Talk to your health care provider if you eat large amounts of soy products, or if you typically eat a high-fiber diet. Also, tell your doctor if you take other medicines, especially:
- Iron supplements or multivitamins that contain iron.
- Aluminum hydroxide, which is found in some antacids.
- Calcium supplements.
Side effects and complications of thyroxine treatment
The only dangers of thyroxine are caused by taking too little or too much. If you take too little, your hypothyroidism will continue. If you take too much, you’ll develop the symptoms of hyperthyroidism—an overactive thyroid gland. The most common symptoms of too much thyroid hormone are fatigue but inability to sleep, greater appetite, nervousness, shakiness, feeling hot when other people are cold, and trouble exercising because of weak muscles, shortness of breath , and a racing, skipping heart. Patients who have hyperthyroid symptoms at any time during thyroxine replacement therapy should have their TSH tested. If it is low, indicating too much thyroid hormone, their dose needs to be lowered.
On-going care and treatment for hypothyroidism
You’ll need to have your TSH checked 6 to 10 weeks after a thyroxine dose change. You may need tests more often if you’re pregnant or you’re taking a medicine that interferes with your body’s ability to use thyroxine. The goal of treatment is to get and keep your TSH in the normal range. Babies with hypothyroidism must get all their daily treatments and have their TSH levels checked as they grow, to prevent mental retardation and stunted growth. Once you’ve settled into a thyroxine dose, you can return for TSH tests about once a year.
You need to return sooner if any of the following apply to you:
- Your symptoms return or get worse.
- You want to change your thyroxine dose or brand, or change taking your pills with or without food.
- You gain or lose a lot of weight (as little as a 10-pound difference for those who weren’t overweight to begin with).
- You start or stop taking a drug that can interfere with absorbing thyroxine (such as certain antacids, calcium supplements and iron tablets), or you change your dose of such a drug. Medications containing estrogen also impact thyroxine doses, so any change in such a medication should prompt a re-evaluation of your thyroxine dose.
- You start or stop taking certain medicines to control seizures such as phenytoin or tegretol, as such medicines increase the rate at which thyroxine is metabolized in your body, and your dose of thyroxine may need to be adjusted.
- You’re not taking all your thyroxine pills. Tell your doctor honestly how many pills you’ve missed.
- You want to try stopping thyroxine treatment. If ever you think you’re doing well enough not to need thyroxine treatment any longer, try it only under your doctor’s close supervision. Rather than stopping your pills completely, you might ask your doctor to try lowering your dose. If your TSH goes up, you’ll know that you need to continue treatment.
Treatment targets
Treatment targets include normalisation of TSH concentrations and resolution of physical and mental complaints, while avoiding undertreatment or overtreatment 1. Nevertheless, an estimated 35—60% of patients treated with levothyroxine do not reach the target range of TSH (either overtreated or undertreated) 54, 55. Results from a retrospective cohort study in the UK 54 showed that, after 5 years of levothyroxine therapy, almost 6% of patients have TSH concentrations below 0–1 mIU/L and more than 10% have TSH concentrations above 10–0 mIU/L. Overtreatment (ie, subclinical or overt hyperthyroidism) can have deleterious health effects, such as atrial fibrillation and osteoporosis, and should always be avoided, especially in the elderly and postmenopausal women. Undertreatment (ie, persistent thyroid hormone deficiency) can result in an increased risk of cardiovascular disease and persistent signs and symptoms. Treatment targets for central hypothyroidism are different from primary hypothyroidism because clinicians cannot rely on the so- called reflex TSH strategy 30.
Levothyroxine (T4) and triiodothyronine (T3) combination therapy
Several trials using combined levothyroxine (T4)+ triiodothyronine or liothyronine (T3) therapy have been done in the past 15 years 56. Although some studies show a beneficial effect 57, 58, 59, 60, such as patient preference for combination therapy or an improved metabolic profile, patients on combination therapy generally do not have improved outcomes compared with those on levothyroxine monotherapy 61. Possible explanations include inadequate levothyroxine (T4) and liothyronine (T3) doses or frequency of administration 62. Liothyronine (T3) has a short half-life and no previous studies used a slow-release tri-iodothyronine. Additionally, most trials were of short duration (<4 months) and used questionnaires to record patients’ symptoms, which might not have been targeted or sensitive enough.
Alternatively, trials might have failed to identify the appropriate subgroups that would benefit from combination therapy 2. Most trials did not specifically recruit patients who feel unwell on levothyroxine or those with particularly low serum tri-iodothyronine concentrations. Individuals with genetic variations in thyroid hormone metabolism have not been specifically targeted 62. A subgroup that could be targeted is individuals with common genetic variations in DIO2, which encodes the deiodinase 2 enzyme that converts thyroxine to tri-iodothyronine locally in several tissues, including the brain 63. The Thr92Ala polymorphism in DIO2 gives rise to deiodinase 2 with a longer half-life than the wild-type enzyme and ectopic localisation in the Golgi apparatus. It was shown to alter expression profiles in the cerebral cortex in a similar pattern as seen in neurodegenerative disease, without evidence of altered thyroid hormone signalling 64. In a study of 552 people 65, the Thr92Ala polymorphism in DIO2 was associated with lower baseline psychological wellbeing in patients on levothyroxine replacement therapy and with better response to combination therapy, compared with patients without the polymorphism on levothyroxine replacement therapy. However, after appropriate multiple testing correction, the results were not significant. Results from a population-based cohort study 66 showed no effect of the Thr92Ala polymorphism on quality of life or cognitive function measures. Sufficiently powered prospective randomised controlled trials are therefore needed before conclusions can be drawn.
Although the American Thyroid Association 1 and European Thyroid Association guidelines 67 generally recommend against the routine use of combination therapy in patients with hypothyroidism, the recommendations concerning trials in patients with persistent symptoms differ slightly. The European Thyroid Association states that a 3 month trial of levothyroxine–liothyronine combination might be considered experimentally in adherent, biochemically well controlled patients who have persistent complaints despite levothyroxine treatment and provides methods for calculating levothyroxine and liothyronine doses 67. However, treatment should be initiated only by accredited doctors of internal medicine or endocrinologists, closely monitored, and discontinued if no improvement is seen. By contrast, the American Thyroid Association recommends against any routine use of such trials outside of formal research and clinical trials, mainly because of uncertainty regarding benefit and long-term safety 1. Both the European Thyroid Association and American Thyroid Association agree on the need for long-term randomised controlled trials to assess risk-benefit ratios. Such trials would need to incorporate investigation of the ideal thyroid parameters to monitor during combination therapy, and whether tri-iodothyronine concentrations would be an important parameter. The timing of phlebotomy is also important, particularly if liothyronine is being administered more than once daily.
Little evidence exists to support other therapies for hypothyroidism. The use of thyroid extracts or liothyronine monotherapy is generally not recommended because of potential safety concerns associated with the presence of supraphysiological serum tri-iodothyronine concentrations and a paucity of long-term safety outcome data. The use of compounded thyroid hormones, dietary supplements, and any over-the-counter drug for the treatment of hypothyroidism is discouraged.
Subclinical hypothyroidism
If you are diagnosed with subclinical hypothyroidism, talk about treatment with your endocrinologist, a physician who is a specialist in thyroid diseases. For a mild rise in TSH, thyroid hormone medicine may not be useful. If your TSH level is higher, but still in the subclinical range, thyroid hormones may improve some symptoms.
Hashimoto’s thyroiditis
Hashimoto’s thyroiditis also known as Hashimoto’s disease, chronic lymphocytic thyroiditis or autoimmune thyroiditis, is an autoimmune disease where your body’s immune system attacks your thyroid gland, preventing it from producing enough thyroid hormones (tri-iodothyronine [T3] and thyroxine [T4]). Low thyroid hormone levels may cause hypothyroidism with a range of symptoms, such as tiredness or fatigue, weight gain, intolerance to cold temperatures, dry skin or dry thinning hair, slowed heart rate, heavy or irregular menstrual periods or fertility problems. Rarely, early in the course of the disease, thyroid gland damage may lead to the release of too much thyroid hormone into your blood, causing symptoms of hyperthyroidism 68. Too much thyroid hormone (hyperthyroidism) can cause weight loss, despite an increased appetite. You might also feel anxious and find it difficult to relax.
Your thyroid gland is a butterfly-shaped gland with 2 lobes (the right lobe and the left lobe — joined by a narrow piece of the thyroid gland called the isthmus) that is located in front of your neck near the base of your throat, beneath the larynx (voice box or Adam’s apple) (Figure 1). In most people, the thyroid gland cannot be seen or felt. Your thyroid gland produces thyroid hormones, tri-iodothyronine (T3) and thyroxine (T4) (the main hormones that your thyroid gland makes) and calcitonin. The thyroid hormones, T3 (tri-iodothyronine) and T4 (thyroxine) influence important body processes such as body temperature, energy levels, growth, your digestion, muscles and heart. Thyroid hormones are important for how your body uses energy, your metabolism, so thyroid hormones affect nearly every organ in your body even the way your heart beats. You might put on weight and feel very tired and lacking in energy if your thyroid gland doesn’t make enough T3 (tri-iodothyronine) and T4 (thyroxine).
Calcitonin is another hormone produced by the thyroid gland. Calcitonin helps to control the amount of calcium circulating in your blood. Calcitonin works with a hormone called parathyroid hormone (PTH) to do this. Parathyroid hormone is made by parathyroid glands. These sit behind and are attached to the thyroid gland (see Figure 1).
In people with Hashimoto’s disease:
- the immune system makes antibodies that attack the thyroid gland (autoimmune disorder). Usually in Hashimoto’s disease, the immune system produces an antibody to thyroid peroxidase (TPO), a protein that plays an important part in thyroid hormone production. Most people with Hashimoto’s disease will have TPO (thyroid peroxidase) antibodies in their blood. Lab tests for other antibodies associated with Hashimoto’s disease may need to be done.
- large numbers of white blood cells, which are part of the immune system, build up in the thyroid gland
- the thyroid gland becomes damaged and can’t make enough thyroid hormones
Hashimoto’s disease is an autoimmune disorder affecting the thyroid gland. In Hashimoto’s thyroiditis, the immune-system cells lead to the death of the thyroid’s hormone-producing cells. Hashimoto’s disease usually results in a decline in thyroid hormones production (hypothyroidism). The symptoms of hypothyroidism might be mild, or they might be severe. They include:
- fatigue
- being unable to stand the cold
- weight gain
- constipation
- muscle pain
- dry skin, thin hair and / or brittle nails
- low sex drive (libido)
Hashimoto’s disease can also cause cognitive symptoms including:
- depression or low mood
- an inability to concentrate
- poor memory
In some cases, your thyroid gland may become noticeably larger (called a goiter) or it may shrink. Lumps or nodules may also develop in your thyroid gland.
Although Hashimoto’s disease can affect people of all ages, it’s most common in women in their 30s and 40s 69. The female-to-male ratio is at least 10:1 68. If someone in your family has had thyroid disease, you may have an increased risk for Hashimoto’s disease. No one is sure why people get Hashimoto’s disease.
If you have symptoms of hypothyroidism, see your doctor. Your doctor will examine you and may run blood tests, including testing your thyroid hormone levels.
If left untreated, hypothyroidism can lead to problems including goiter (an increase in the size of the thyroid gland), heart problems or mental health problems. Occasionally, it can lead to a potentially life-threatening disorder called myxedema coma.
While there is no cure for Hashimoto’s disease, hypothyroidism can be treated. The primary treatment of Hashimoto’s disease is thyroid hormone replacement. Most people with Hashimoto’s disease take a synthetic thyroid hormone medication called levothyroxine (Levoxyl, Synthroid, others) to treat hypothyroidism. The synthetic thyroid hormone works like the T4 hormone naturally produced by the thyroid. Your hypothyroidism can be well-controlled with thyroid hormone medicine, as long as you take the medicine as instructed by your doctor and have regular follow-up blood tests.
If you have mild hypothyroidism, you may not need to have treatment but get regular thyroid stimulating hormone (TSH) tests to monitor thyroid hormone levels.
How common is Hashimoto’s disease?
The number of people who have Hashimoto’s disease in the United States is unknown. However, Hashimoto’s disease is the most common cause of hypothyroidism the United States, which affects about 5 in 100 Americans 70.
Hashimoto is also the most common cause of hypothyroidism in those areas of the world where iodine intake is adequate. The incidence is estimated to be 0.8 per 1000 per year in men and 3.5 per 1000 per year in women 68. The prevalence of thyroid disease, in general, increases with age.
Hashimoto’s thyroiditis causes
Hashimoto’s disease is an autoimmune disorder. The immune system creates antibodies that attack thyroid cells as if they were bacteria, viruses or some other foreign body. The immune system wrongly enlists disease-fighting agents that damage cells and lead to cell death. What causes the immune system to attack thyroid cells is not clear. Multiple factors from the external environment and the genetic background contribute to the pathogenesis of Hashimoto’s disease 71. These genetic, environmental, and existential factors provoke the immune system to produce antibodies to thyroid antigens 44, 72, 73, 74, 75, 76, 77, 78. The most important factors associated with Hashimoto’s thyroiditis are summarized in Table 3 below.
The onset of Hashimoto’s disease may be related to 79, 80, 2, 81:
- Genetic factors. Twin studies have shown an increased concordance of autoimmune thyroiditis in monozygotic twins as compared with dizygotic twins. Danish studies have demonstrated concordance rates of 55% in monozygotic twins, compared with only 3% in dizygotic twins 82. This data suggests that 79% of predisposition is due to genetic factors, allotting 21% for environmental and sex hormone influences.
- Environmental triggers, such as infection, stress or radiation exposure
- Interactions between environmental and genetic factors.
Hypothyroidism can also be caused by:
- some medicines used to treat bipolar disorder or other mental health problems
- iodine-containing medicines used to treat abnormal heart rhythm
- exposure to toxins, such as nuclear radiation
- viruses, such as hepatitis C
Several genes have been involved in Hashimoto’s disease pathogenesis, including genes of the immune response (coded in the Human Leukocyte Antigen (HLA) complex) and thyroid function 71. Other immunoregulatory genes are involved in the development of Hashimoto’s disease, including the single nucleotide polymorphisms (SNPs) in cytotoxic T lymphocyte-associated antigen 4 (CTLA-4), protein tyrosine phosphatase non-receptor type 22 (PTPN22), and CD40 83, 44, 76, 84.
Among the environmental factors are inadequate or excessive iodine intake, infections, or the intake of certain medications 44, 76, 75, 77, 85. Several of the currently used anticancer drugs, such as interferon-alpha, may cause autoimmune thyroid dysfunction 86, 78. The role of smoking and alcohol consumption in the etiopathogenesis of Hashimoto’s disease is still not clear 71. The data suggest that moderate alcohol consumption may protect against Hashimoto’s disease and the development of overt hypothyroidism 78, 87, 88. Furthermore, some studies indicate that smoking decreases the levels of thyroid autoantibodies and the risk of hypothyroidism. However, the mechanism for these protective effects of smoking and drinking remains unclear and must be clarified with future studies 78, 87, 88. In recent years, the influence of stress on the development and course of Hashimoto’s disease has also been investigated. Some studies suggest that stress is involved in the pathogenesis of Hashimoto’s disease, while other evidence indicates that it has no effect 78, 89. A randomized controlled trial by Markomanolaki et al. 90 showed that managing stress is also important in treating Hashimoto’s disease patients. After eight weeks of stress management intervention, patients demonstrated a reduction in antithyroglobulin (anti-Tg) titers, decreased levels of stress, depression, anxiety and improved lifestyle 90. Additionally, the adequate levels of vitamin D and selenium may help prevent or delay the onset of Hashimoto’s disease 75, 77, 91, 92. Moreover, the risk of Hashimoto’s disease is increased in other autoimmune diseases 93, 78.
Most Hashimoto’s disease patients develop antibodies to a variety of thyroid antigens, the most common of which is anti-thyroid peroxidase (anti-TPO). Many also form antithyroglobulin (anti-Tg) and TSH receptor-blocking antibodies (TBII) 68. These antibodies attack the thyroid tissue, eventually leading to inadequate production of thyroid hormone. There is a small subset of the population, no more than 10-15% with the clinically evident disease, that are serum antibody-negative 68.
Table 3. Genetic, environmental and existential factors associated with Hashimoto’s thyroiditis
| Genetic Factors | Environmental Factors | Existential Factors |
|---|---|---|
| Histocompatibility genes (HLA class I and II) | Iodine | Sex |
| Immunoregulatory genes (SNPs in HLA, CTLA-4, PTPN22, CD40 genes) | Medications (e.g., interferon-α, lithium, amiodarone) | Associated diseases (e.g., type 1 diabetes mellitus, pernicious anaemia, coeliac disease, myasthenia gravis) |
| Thyroid-specific genes | Infections (e.g., hepatitis C virus) | Age |
| Genes associated with thyroid peroxidase antibody synthesis | Smoking | Pregnancy |
| Selenium | Down’s syndrome | |
| Vitamin D | Microbiome composition | |
| Alcohol | Familial aggregation | |
| Radiation Exposure |
Risk factors for Hashimoto’s thyroiditis
The following factors are associated with an increased risk of Hashimoto’s disease 94:
- Sex. Women are much more likely to get Hashimoto’s disease.
- Age. Hashimoto’s disease can occur at any age but more commonly occurs during middle age.
- Other autoimmune disease. Having another autoimmune disease — such as rheumatoid arthritis, type 1 diabetes or lupus — increases your risk of developing Hashimoto’s disease.
- Genetics and family history. You’re at higher risk for Hashimoto’s disease if others in your family have thyroid disorders or other autoimmune diseases.
- Pregnancy. Typical changes in immune function during pregnancy may be a factor in Hashimoto’s disease that begins after pregnancy.
- Excessive iodine intake. Too much iodine in the diet may function as a trigger among people already at risk for Hashimoto’s disease.
- Radiation exposure. People exposed to excessive levels of environmental radiation are more prone to Hashimoto’s disease.
Signs and symptoms of Hashimoto’s thyroiditis
Signs and symptoms of Hashimoto’s thyroiditis vary widely and are not specific to the disorder. Hashimoto’s disease progresses slowly over the years. Many people with Hashimoto’s thyroiditis may not notice signs or symptoms of the disease at first. An ordinary blood test may just show a thyroid hormone imbalance. Because the thyroid gland may grow and get larger, you may have a feeling of fullness or tightness in your throat, though it is usually not painful. You may have trouble swallowing food or liquids. You might have a swelling (a bump) in the front of your neck, the enlarged thyroid is called a goiter. After many years, or even decades, damage to the thyroid may cause the gland to shrink and the goiter to disappear.
Some people with Hashimoto’s thyroiditis have symptoms such as tiredness, forgetfulness, depression, coarse dry skin, slow heartbeat, weight gain, constipation and intolerance to cold. A blood test can tell if your thyroid gland is underactive. Other blood tests can be done to look for Hashimoto’s disease.
Eventually, the decline in thyroid hormone production can result in hypothyroidism with any of the following:
- Fatigue and sluggishness
- Increased sensitivity to cold
- Increased sleepiness
- Dry skin
- Constipation
- Muscle weakness
- Muscle aches, tenderness and stiffness
- Joint pain and stiffness
- Irregular or excessive menstrual bleeding
- Depression
- Problems with memory or concentration
- Swelling of the thyroid (goiter)
- A puffy face
- Brittle nails
- Hair loss
- Enlargement of the tongue
Because these symptoms could result from any number of disorders, it’s important to see your doctor as soon as possible for a timely and accurate diagnosis.
How is Hashimoto’s thyroiditis diagnosed?
A number of conditions may lead to the signs and symptoms of Hashimoto’s thyroiditis. If you’re experiencing any of these symptoms, your health care provider will conduct a thorough physical exam, review your medical history and ask questions about your symptoms.
Testing thyroid function
To determine if hypothyroidism is the cause of your symptoms, your doctor will order blood tests that may include the following:
- Thyroid stimulating hormone (TSH) test. Thyroid stimulating hormone (TSH) is produced by the pituitary gland. When the pituitary detects low thyroid hormones in the blood, it sends TSH to the thyroid to prompt an increase in thyroid hormone production. High TSH levels in the blood indicates hypothyroidism.
- Thyroxine (T4) tests. The main thyroid hormone is thyroxine (T4). A low blood level of T4 confirms the findings of a TSH (thyroid stimulating hormone) test and indicates the problem is within the thyroid itself.
Antibody tests
More than one disease process can lead to hypothyroidism. To determine if Hashimoto’s thyroiditis is the cause of hypothyroidism, your doctor will order an antibody test.
The intended purpose of an antibody is to flag disease-causing foreign agents that need to be destroyed by other actors in the immune system. In an autoimmune disorder, the immune system produces rogue antibodies that target healthy cells or proteins in the body.
Usually in Hashimoto’s thyroiditis, the immune system produces an antibody to thyroid peroxidase (anti-TPO), a protein that plays an important part in thyroid hormone production. Most people with Hashimoto’s disease will have TPO antibodies (anti-TPO) in their blood. Lab tests for other antibodies associated with Hashimoto’s disease may also need to be done.
Thyroglobulin antibodies (Tg) can also be a sign of Hashimoto’s thyroiditis. Most people with Hashimoto disease have high levels of both thyroglobulin antibodies (anti-Tg) and TPO antibodies (anti-TPO).
Circulating antibody to thyroid peroxidase (anti-TPO) are found in about 90% of Hashimoto’s disease patients. Anti-thyroglobulin antibodies (anti-Tg) are less sensitive (positive in about 60–80% of patients) and less specific than antibody to thyroid peroxidase (anti-TPO) 43, 44, 45.
You probably won’t need other tests to confirm you have Hashimoto’s disease. However, if your doctor suspects Hashimoto’s disease but you don’t have antithyroid antibodies in your blood, you may have an ultrasound of your thyroid. The ultrasound images can show the size of your thyroid and other features of Hashimoto’s disease. The ultrasound also can rule out other causes of an enlarged thyroid, such as thyroid nodules—small lumps in the thyroid gland.
How is Hashimoto’s thyroiditis treated?
How your doctors treat Hashimoto’s disease usually depends on whether your thyroid is damaged enough to cause hypothyroidism. If you don’t have hypothyroidism or you have mild hypothyroidism, your doctor may choose to simply check your symptoms and do regular thyroid stimulating hormone (TSH) tests to monitor your thyroid hormone levels.
Most people with Hashimoto’s disease need take a synthetic thyroid hormone medication called levothyroxine (Levoxyl, Synthroid, others) to treat hypothyroidism. The synthetic thyroid hormone works like the thyroxine (T4) hormone naturally produced by your thyroid. Prescribed in pill form for many years, this medicine is now also available as a liquid and in a soft gel capsule 94. These newer formulas may be helpful to people with digestive problems that affect how the thyroid hormone pill is absorbed.
Some foods and supplements can affect how well your body absorbs levothyroxine. Examples include grapefruit juice, espresso coffee, soy, and multivitamins that contain iron or calcium 70, 95. Taking levothyroxine on an empty stomach can prevent this from happening. Your doctor may ask you to take the levothyroxine in the morning, 30 to 60 minutes before you eat your first meal.
Your doctor will give you a blood test about 6 to 8 weeks after you begin taking levothyroxine and adjust your dose if needed. Each time you change your dose, you’ll have another blood test. Once you’ve reached a dose that’s working for you, your doctor will likely repeat the blood test in 6 months and then once a year.
Never stop taking your levothyroxine or take a higher dose without talking with your doctor first. Taking too much thyroid hormone medicine can cause serious problems, such as atrial fibrillation or osteoporosis 2.
Thyroxine (T4) hormone replacement therapy
Hypothyroidism associated with Hashimoto’s disease is treated with a synthetic hormone called levothyroxine (Levoxyl, Synthroid, others). The recommended dose of levothyroxine is 1.6 to 1.8 mcg/kg/day 68. The synthetic hormone works like the thyroxine (T4) hormone naturally produced by the thyroid. The treatment goal is to restore and maintain adequate thyroxine (T4) hormone levels and improve symptoms of hypothyroidism. You will need this treatment for the rest of your life.
Monitoring the dosage
Your doctor will determine a dosage of levothyroxine that’s appropriate for your age, weight, current thyroid production, other medical conditions and other factors. Your doctor will retest your TSH (thyroid stimulating hormone) levels about 6 to 10 weeks later and adjust the dosage as necessary.
Once the best dosage is determined, you will continue to take the medication once a day. You’ll need follow-up tests once a year to monitor TSH (thyroid stimulating hormone) levels or any time after your doctor changes your dosage.
A levothyroxine pill is usually taken in the morning before you eat. Talk to your doctor if you have any questions about when or how to take the pill. Also, ask what to do if you accidentally skip a dose. If your health insurance requires you to switch to a generic drug or a different brand, talk to your doctor.
Precautions
Because levothyroxine acts like natural thyroxine (T4) in your body, there are generally no side effects as long as the treatment is resulting in “natural” levels of thyroxine (T4) for your body.
Too much thyroid hormone can worsen bone loss that causes weak, brittle bones (osteoporosis) or cause irregular heartbeats (arrhythmias) the most common being atrial fibrillation.
Effects of other substances
Certain medications, supplements and foods may affect your ability to absorb levothyroxine. It may be necessary to take levothyroxine at least four hours before these substances. Talk to your doctor about any of the following:
- Soy products
- High-fiber foods
- Iron supplements, including multivitamins that contain iron
- Cholestyramine (Prevalite), a medication used to lower blood cholesterol levels
- Aluminum hydroxide, which is found in some antacids
- Sucralfate, an ulcer medication
- Calcium supplements
Triiodothyronine (T3) hormone replacement therapy
Naturally produced thyroxine (T4) is converted into another thyroid hormone called triiodothyronine (T3). The thyroxine (T4) replacement hormone is also converted into triiodothyronine (T3), and for most people the thyroxine (T4) replacement therapy results in an adequate supply of triiodothyronine (T3) for the body.
For people who need better symptom control, a doctor also may prescribe a synthetic triiodothyronine (T3) (Cytomel) or a synthetic T4 and T3 combination. Side effects of triiodothyronine (T3) hormone replacement include rapid heartbeat, insomnia and anxiety. These treatments may be tested with a trial period of 3 to 6 months.
Alternative medicine
Products with triiodothyronine (T3) and thyroxine (T4) hormones derived from pigs or other animals are available as prescriptions or as dietary supplements, such as Armour Thyroid, in the United States. Concerns about these products include the following:
- The balance of thyroxine (T4) and triiodothyronine (T3) in animals isn’t the same as in humans.
- The exact amount of thyroxine (T4) and triiodothyronine (T3) in each batch of a natural extract product can vary, leading to unpredictable levels of these hormones in your blood.
Anti-inflammatory diet
An anti-inflammatory diet rich in vitamins, minerals and polyphenols is recommended as diet therapy for Hashimoto’s disease 96, 85, 97. The theory behind the inflammation has to do with the leaky gut syndrome, where there is an insult to the gut mucosa, which allows the penetrance of proteins that do not typically enter the bloodstream via transporters in the gut mucosa. It is theorized that a response similar to molecular mimicry occurs, and antibodies are produced against the antigens. Unfortunately, the antigen may be very structurally similar to thyroid peroxidase, leading to antibody formation against this enzyme. The concept of an autoimmune diet is based on healing the gut and decreasing the severity of the autoimmune response.
Natural antioxidants like vitamin A, vitamin C and vitamin E are found in products of plant origin, including a wide variety of vegetables and fruits. Sources of vitamin C include broccoli, peppers, black currant, strawberries, lemons, spinach, kiwifruit, oranges, grapefruit, limes, tomatoes, raspberries, asparagus, pineapples, fennel and parsley. The best source of vitamin E is avocado, nuts, seeds, egg, milk and whole grains. In addition, vitamin A is present in foods such as liver, carrot, broccoli, butter, pumpkin, cheese, egg, mango and milk 98. According to the current findings, the Mediterranean diet may show the most benefits for Hashimoto’s disease patients with its antioxidant properties 99.
One study by Ostrowska et al. 100 assessed the effectiveness of two “reducing diets” and their effect on thyroid parameters in female obese patients with Hashimoto’s disease. All women who received levothyroxine, selenium and zinc were randomly assigned to the study group following individually balanced elimination/reducing diets, in accordance with the previously performer food sensitivity tests, and the control group following reducing diets with the same caloric content, but without product elimination. The anthropometric and thyroid parameters have changed in both groups during the nutritional intervention. This research showed that weight reduction may improve thyroid function in patients suffering from obesity and Hashimoto’s disease 100. Moreover, an individually selected elimination reducing diet was more effective than classic reducing diets with the same energy intake and macronutrient content and can lead to better therapeutic outcomes, which may cause an anti-inflammatory effect 100.
One case report 101 showed a novel approach that led to the improvement of symptoms and a reduction of thyroid antibodies in a 23-year-old woman with Hashimoto’s disease. The woman presented with symptoms of fatigue, hair loss, energy and mood disturbance, problems with insomnia and daytime napping. The thyroid antibodies were strongly positive, with a normal TSH level. Integrative treatment was started, which involved nutritional changes and micronutrient supplementation 101. This supplementation supported the methylation cycle, anti-oxidant capacity and stress management, and included vitamin C, vitamin B1, vitamin B2, vitamin B5, vitamin B6, Pyridoxal-5 Phosphate, zinc picolonate, L-5 methyltetrahydrofolate, magnesium glycinate, selenomethionine, N- Acetyl Cysteine and methylcobalamin (vitamin B12). The patient followed a paleo-style diet without grains and dairy products and increased consumption of bone broth and fermented foods as well as organic animal protein as tolerated. In addition, daily meditation and mindfulness techniques were recommended, and gentle exercise three times a week was added. After 15 months of treatment, there was a reduction in antithyroid antibodies and a significant relief of symptoms. This case demonstrated the potential benefits of an integrative approach to autoimmunity and oxidative stress in Hashimoto’s disease 101.
In a pilot study by Abbott et al. 102, women participated in a 10-week online health coaching program focused on implementing an “autoimmune protocol diet”. They applied a modified paleolithic diet. In the referred study, there were no significant changes in thyroid function markers, as well as serum antithyroid antibody concentrations, although the number of immune cells and an inflammatory processes marker (high sensitivity CRP) were decreased. These results suggest that an “autoimmune protocol” may decrease inflammation and modulate the immune system. Moreover, the therapy improves health-related quality of life (measured by 36-Item Short-Form Health Survey) and reduces symptoms of the diseases (measured by the Medical Symptoms Questionnaire) 102. A case study with a 49-year-old obese Hashimoto’s disease woman indicated that a modified autoimmune paleo low-calorie diet might improve TSH, anti-TPO antibody, body composition and lipid profile 103.
- Jonklaas J, Bianco AC, Bauer AJ, Burman KD, Cappola AR, Celi FS, Cooper DS, Kim BW, Peeters RP, Rosenthal MS, Sawka AM; American Thyroid Association Task Force on Thyroid Hormone Replacement. Guidelines for the treatment of hypothyroidism: prepared by the american thyroid association task force on thyroid hormone replacement. Thyroid. 2014 Dec;24(12):1670-751. doi: 10.1089/thy.2014.0028[↩][↩][↩][↩][↩][↩][↩]
- Chaker L, Bianco AC, Jonklaas J, Peeters RP. Hypothyroidism. Lancet. 2017 Sep 23;390(10101):1550-1562. doi: 10.1016/S0140-6736(17)30703-1[↩][↩][↩][↩][↩][↩][↩][↩][↩]
- Evered DC, Ormston BJ, Smith PA, Hall R, Bird T. Grades of hypothyroidism. Br Med J. 1973 Mar 17;1(5854):657-62. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1588654/pdf/brmedj01547-0041.pdf[↩]
- Hypothyroidism, Medline Plus. https://medlineplus.gov/hypothyroidism.html[↩]
- Patil N, Rehman A, Jialal I. Hypothyroidism. [Updated 2022 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK519536[↩]
- Garber JR, Cobin RH, Garib H, et al. Clinical Practice Guidelines for Hypothyroidism in Adults: Cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocrine Practice. 2012;18(6):988–1028.[↩]
- Aoki Y, Belin RM, Clickner R, Jeffries R, Phillips L, Mahaffey KR. Serum TSH and total T4 in the United States population and their association with participant characteristics: National Health and Nutrition Examination Survey (NHANES 1999-2002). Thyroid. 2007 Dec;17(12):1211-23. doi: 10.1089/thy.2006.0235[↩]
- McLeod DS, Caturegli P, Cooper DS, Matos PG, Hutfless S. Variation in rates of autoimmune thyroid disease by race/ethnicity in US military personnel. JAMA. 2014 Apr 16;311(15):1563-5. doi: 10.1001/jama.2013.285606[↩]
- Sichieri, R., Baima, J., Marante, T., De Vasconcellos, M.T.L., Moura, A.S. and Vaisman, M. (2007), Low prevalence of hypothyroidism among black and Mulatto people in a population-based study of Brazilian women. Clinical Endocrinology, 66: 803-807. https://doi.org/10.1111/j.1365-2265.2007.02816.x[↩]
- Taylor PN, Albrecht D, Scholz A, Gutierrez-Buey G, Lazarus JH, Dayan CM, Okosieme OE. Global epidemiology of hyperthyroidism and hypothyroidism. Nat Rev Endocrinol. 2018 May;14(5):301-316. doi: 10.1038/nrendo.2018.18[↩]
- Carlé A, Pedersen IB, Knudsen N, Perrild H, Ovesen L, Rasmussen LB, Jørgensen T, Laurberg P. Moderate alcohol consumption may protect against overt autoimmune hypothyroidism: a population-based case-control study. Eur J Endocrinol. 2012 Oct;167(4):483-90. doi: 10.1530/EJE-12-0356[↩]
- Asvold BO, Bjøro T, Nilsen TI, Vatten LJ. Tobacco smoking and thyroid function: a population-based study. Arch Intern Med. 2007 Jul 9;167(13):1428-32. doi: 10.1001/archinte.167.13.1428[↩]
- Hypothyroidism. American Thyroid Association. https://www.thyroid.org/hypothyroidism/[↩]
- Hypothyroidism (Underactive Thyroid). The National Institute of Diabetes and Digestive and Kidney Diseases. https://www.niddk.nih.gov/health-information/endocrine-diseases/hypothyroidism[↩]
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. Washington, DC: National Academy Press, 2001. https://www.nap.edu/read/10026/chapter/2[↩][↩]
- World Health Organization. United Nations Children’s Fund & International Council for the Control of Iodine Deficiency Disorders. Assessment of iodine deficiency disorders and monitoring their elimination. 3rd ed. Geneva, Switzerland: WHO, 2007. http://apps.who.int/iris/bitstream/handle/10665/43781/9789241595827_eng.pdf;jsessionid=2E9F56538AEFD33C83934FB34BD4E8C4[↩]
- WHO Secretariat, Andersson M, de Benoist B, Delange F, Zupan J. Prevention and control of iodine deficiency in pregnant and lactating women and in children less than 2-years-old: conclusions and recommendations of the Technical Consultation. Public Health Nutr. 2007 Dec;10(12A):1606-11. doi: 10.1017/S1368980007361004. Erratum in: Public Health Nutr. 2008 Mar;11(3):327.[↩]
- World Health Organization. United Nations Children’s Fund & International Council for the Control of Iodine Deficiency Disorders. Assessment of iodine deficiency disorders and monitoring their elimination. 3rd ed. Geneva, Switzerland: WHO, 2007.[↩]
- Zimmermann MB. Iodine deficiency. Endocr Rev. 2009 Jun;30(4):376-408. doi: 10.1210/er.2009-0011[↩]
- USDA, FDA, and ODS-NIH Database for the Iodine Content of Common Foods Release 1.0. 2020. https://www.ars.usda.gov/ARSUSERFILES/80400535/DATA/IODINE/IODINE_DATABASE_PDFVersion_2020.PDF[↩][↩][↩][↩]
- Pennington JA, Young B. Iron, zinc, copper, manganese, selenium, and iodine in foods from the United States Total Diet Studyexternal link disclaimer. J Food Compost Anal. 1990 June;3(2):166-184. https://www.sciencedirect.com/science/article/abs/pii/088915759090022E[↩]
- Ershow AG, Skeaff SA, Merkel JM, Pehrsson PR. Development of Databases on Iodine in Foods and Dietary Supplements. Nutrients. 2018 Jan 17;10(1):100. doi: 10.3390/nu10010100[↩][↩]
- Food Labeling: Revision of the Nutrition and Supplement Facts Labels. https://www.federalregister.gov/documents/2016/05/27/2016-11867/food-labeling-revision-of-the-nutrition-and-supplement-facts-labels[↩]
- Patterson KY, Spungen JH, Roseland JM, Pehrsson PR, Ershow AG, Gahche JJ. USDA-FDA-ODS database for the iodine content of common foods (release one). Iodine database PDF. Methods and Application of Food Composition Laboratory, Beltsville Human Nutrition Research Center, Agricultural Research Service, U.S. Department of Agriculture, Beltsville MD. July 2020. https://www.ars.usda.gov/ARSUSERFILES/80400535/DATA/IODINE/IODINE_DATABASE.PDF[↩]
- Pennington JAT, Schoen SA, Salmon GD, Young B, Johnson RD, Marts RW. Composition of Core Foods of the U.S. Food Supply, 1982-1991. III. Copper, Manganese, Selenium, and Iodine. J Food Comp Anal. 1995;8(2):171-217. https://www.sciencedirect.com/science/article/abs/pii/S0889157585710149[↩]
- Teas J, Pino S, Critchley A, Braverman LE. Variability of iodine content in common commercially available edible seaweeds. Thyroid. 2004 Oct;14(10):836-41. doi: 10.1089/thy.2004.14.836[↩]
- Pennington JAT, Schoen SA, Salmon GD, Young B, Johnson RD, Marts RW. Composition of Core Foods of the U.S. Food Supply, 1982-1991. III. Copper, Manganese, Selenium, and Iodine. J Food Comp Anal. 1995;8(2):171-217.[↩]
- Teas J, Pino S, Critchley A, Braverman LE. Variability of iodine content in common commercially available edible seaweeds. Thyroid. 2004 Oct;14(10):836-841. https://www.ncbi.nlm.nih.gov/pubmed/15588380?dopt=Abstract[↩]
- Dasgupta PK, Liu Y, Dyke JV. Iodine nutrition: iodine content of iodized salt in the United States. Environ Sci Technol. 2008 Feb 15;42(4):1315-1323. https://www.ncbi.nlm.nih.gov/pubmed/18351111?dopt=Abstract[↩]
- Persani L. Clinical review: Central hypothyroidism: pathogenic, diagnostic, and therapeutic challenges. J Clin Endocrinol Metab. 2012 Sep;97(9):3068-78. doi: 10.1210/jc.2012-1616[↩][↩]
- Persani L, Ferretti E, Borgato S, Faglia G, Beck-Peccoz P. Circulating thyrotropin bioactivity in sporadic central hypothyroidism. J Clin Endocrinol Metab. 2000 Oct;85(10):3631-5. doi: 10.1210/jcem.85.10.6895[↩]
- Graeppi-Dulac J, Vlaeminck-Guillem V, Perier-Muzet M, Dalle S, Orgiazzi J. Endocrine side-effects of anti-cancer drugs: the impact of retinoids on the thyroid axis. Eur J Endocrinol. 2014 Jun;170(6):R253-62. doi: 10.1530/EJE-13-0920[↩]
- Grunenwald S, Caron P. Central hypothyroidism in adults: better understanding for better care. Pituitary. 2015 Feb;18(1):169-75. doi: 10.1007/s11102-014-0559-8[↩]
- Shine B, McKnight RF, Leaver L, Geddes JR. Long-term effects of lithium on renal, thyroid, and parathyroid function: a retrospective analysis of laboratory data. Lancet. 2015 Aug 1;386(9992):461-8. doi: 10.1016/S0140-6736(14)61842-0[↩]
- Shulman KI, Sykora K, Gill SS, Mamdani M, Anderson G, Marras C, Wodchis WP, Lee PE, Rochon P. New thyroxine treatment in older adults beginning lithium therapy: implications for clinical practice. Am J Geriatr Psychiatry. 2005 Apr;13(4):299-304. doi: 10.1176/appi.ajgp.13.4.299[↩]
- Zhong B, Wang Y, Zhang G, Wang Z. Environmental Iodine Content, Female Sex and Age Are Associated with New-Onset Amiodarone-Induced Hypothyroidism: A Systematic Review and Meta-Analysis of Adverse Reactions of Amiodarone on the Thyroid. Cardiology. 2016;134(3):366-71. doi: 10.1159/000444578[↩]
- Krohn T, Hänscheid H, Müller B, Behrendt FF, Heinzel A, Mottaghy FM, Verburg FA. Maximum dose rate is a determinant of hypothyroidism after 131I therapy of Graves’ disease but the total thyroid absorbed dose is not. J Clin Endocrinol Metab. 2014 Nov;99(11):4109-15. doi: 10.1210/jc.2014-1347[↩]
- Lee V, Chan SY, Choi CW, Kwong D, Lam KO, Tong CC, Sze CK, Ng S, Leung TW, Lee A. Dosimetric Predictors of Hypothyroidism After Radical Intensity-modulated Radiation Therapy for Non-metastatic Nasopharyngeal Carcinoma. Clin Oncol (R Coll Radiol). 2016 Aug;28(8):e52-60. doi: 10.1016/j.clon.2016.05.004[↩]
- Huang SA, Tu HM, Harney JW, Venihaki M, Butte AJ, Kozakewich HP, Fishman SJ, Larsen PR. Severe hypothyroidism caused by type 3 iodothyronine deiodinase in infantile hemangiomas. N Engl J Med. 2000 Jul 20;343(3):185-9. doi: 10.1056/NEJM200007203430305[↩]
- Dumitrescu AM, Refetoff S. The syndromes of reduced sensitivity to thyroid hormone. Biochim Biophys Acta. 2013 Jul;1830(7):3987-4003. doi: 10.1016/j.bbagen.2012.08.005[↩]
- Zimmerman MB. Iodine deficiency in pregnancy and the effects of maternal iodine supplementation on the offspring: a review. American Journal of Clinical Nutrition. 2009;89(2):668S–672S.[↩]
- Beynon J, Akhtar S, Kearney T. Predictors of outcome in myxoedema coma. Crit Care. 2008;12(1):111. doi: 10.1186/cc6218[↩]
- Caturegli P., De Remigis A., Rose N.R. Hashimoto Thyroiditis: Clinical and Diagnostic Criteria. Autoimmun. Rev. 2014;13:391–397. doi: 10.1016/j.autrev.2014.01.007[↩][↩]
- Ragusa F., Fallahi P., Elia G., Gonnella D., Paparo S.R., Giusti C., Churilov L.P., Ferrari S.M., Antonelli A. Hashimotos’ Thyroiditis: Epidemiology, Pathogenesis, Clinic and Therapy. Best Pract. Res. Clin. Endocrinol. Metab. 2019;33:101367. doi: 10.1016/j.beem.2019.101367[↩][↩][↩][↩][↩]
- Iddah M.A., Macharia B.N. Autoimmune Thyroid Disorders. ISRN Endocrinol. 2013;2013:e509764. doi: 10.1155/2013/509764[↩][↩]
- Bolk N, Visser TJ, Nijman J, Jongste IJ, Tijssen JG, Berghout A. Effects of evening vs morning levothyroxine intake: a randomized double-blind crossover trial. Arch Intern Med. 2010 Dec 13;170(22):1996-2003. doi: 10.1001/archinternmed.2010.436[↩]
- Vita R, Saraceno G, Trimarchi F, Benvenga S. Switching levothyroxine from the tablet to the oral solution formulation corrects the impaired absorption of levothyroxine induced by proton-pump inhibitors. J Clin Endocrinol Metab. 2014 Dec;99(12):4481-6. doi: 10.1210/jc.2014-2684[↩]
- Seng Yue C, Benvenga S, Scarsi C, Loprete L, Ducharme MP. When Bioequivalence in Healthy Volunteers May not Translate to Bioequivalence in Patients: Differential Effects of Increased Gastric pH on the Pharmacokinetics of Levothyroxine Capsules and Tablets. J Pharm Pharm Sci. 2015;18(5):844-55. doi: 10.18433/j36p5m[↩]
- Cappelli C, Pirola I, Daffini L, Formenti A, Iacobello C, Cristiano A, Gandossi E, Agabiti Rosei E, Castellano M. A Double-Blind Placebo-Controlled Trial of Liquid Thyroxine Ingested at Breakfast: Results of the TICO Study. Thyroid. 2016 Feb;26(2):197-202. https://core.ac.uk/reader/80138967[↩]
- Pearce SH, Brabant G, Duntas LH, Monzani F, Peeters RP, Razvi S, Wemeau JL. 2013 ETA Guideline: Management of Subclinical Hypothyroidism. Eur Thyroid J. 2013 Dec;2(4):215-28. doi: 10.1159/000356507[↩]
- Roos A, Linn-Rasker SP, van Domburg RT, Tijssen JP, Berghout A. The starting dose of levothyroxine in primary hypothyroidism treatment: a prospective, randomized, double-blind trial. Arch Intern Med. 2005 Aug 8-22;165(15):1714-20. doi: 10.1001/archinte.165.15.1714. Erratum in: Arch Intern Med. 2005 Oct 24;165(19):2227.[↩]
- Alexander EK, Marqusee E, Lawrence J, Jarolim P, Fischer GA, Larsen PR. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med. 2004 Jul 15;351(3):241-9. doi: 10.1056/NEJMoa040079[↩]
- Stagnaro-Green A, Abalovich M, Alexander E, Azizi F, Mestman J, Negro R, Nixon A, Pearce EN, Soldin OP, Sullivan S, Wiersinga W; American Thyroid Association Taskforce on Thyroid Disease During Pregnancy and Postpartum. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid. 2011 Oct;21(10):1081-125. doi: 10.1089/thy.2011.0087[↩]
- Taylor PN, Iqbal A, Minassian C, Sayers A, Draman MS, Greenwood R, Hamilton W, Okosieme O, Panicker V, Thomas SL, Dayan C. Falling threshold for treatment of borderline elevated thyrotropin levels-balancing benefits and risks: evidence from a large community-based study. JAMA Intern Med. 2014 Jan;174(1):32-9. doi: 10.1001/jamainternmed.2013.11312[↩][↩]
- Somwaru LL, Arnold AM, Joshi N, Fried LP, Cappola AR. High frequency of and factors associated with thyroid hormone over-replacement and under-replacement in men and women aged 65 and over. J Clin Endocrinol Metab. 2009 Apr;94(4):1342-5. doi: 10.1210/jc.2008-1696[↩]
- Bunevicius R, Kazanavicius G, Zalinkevicius R, Prange AJ Jr. Effects of thyroxine as compared with thyroxine plus triiodothyronine in patients with hypothyroidism. N Engl J Med. 1999 Feb 11;340(6):424-9. doi: 10.1056/NEJM199902113400603[↩]
- Nygaard B, Jensen EW, Kvetny J, Jarløv A, Faber J. Effect of combination therapy with thyroxine (T4) and 3,5,3′-triiodothyronine versus T4 monotherapy in patients with hypothyroidism, a double-blind, randomised cross-over study. Eur J Endocrinol. 2009 Dec;161(6):895-902. doi: 10.1530/EJE-09-0542[↩]
- Walsh JP, Shiels L, Lim EM, Bhagat CI, Ward LC, Stuckey BG, Dhaliwal SS, Chew GT, Bhagat MC, Cussons AJ. Combined thyroxine/liothyronine treatment does not improve well-being, quality of life, or cognitive function compared to thyroxine alone: a randomized controlled trial in patients with primary hypothyroidism. J Clin Endocrinol Metab. 2003 Oct;88(10):4543-50. doi: 10.1210/jc.2003-030249[↩]
- Appelhof BC, Fliers E, Wekking EM, Schene AH, Huyser J, Tijssen JG, Endert E, van Weert HC, Wiersinga WM. Combined therapy with levothyroxine and liothyronine in two ratios, compared with levothyroxine monotherapy in primary hypothyroidism: a double-blind, randomized, controlled clinical trial. J Clin Endocrinol Metab. 2005 May;90(5):2666-74. doi: 10.1210/jc.2004-2111[↩]
- Escobar-Morreale HF, Botella-Carretero JI, Gómez-Bueno M, Galán JM, Barrios V, Sancho J. Thyroid hormone replacement therapy in primary hypothyroidism: a randomized trial comparing L-thyroxine plus liothyronine with L-thyroxine alone. Ann Intern Med. 2005 Mar 15;142(6):412-24. doi: 10.7326/0003-4819-142-6-200503150-00007[↩]
- Grozinsky-Glasberg S, Fraser A, Nahshoni E, Weizman A, Leibovici L. Thyroxine-triiodothyronine combination therapy versus thyroxine monotherapy for clinical hypothyroidism: meta-analysis of randomized controlled trials. J Clin Endocrinol Metab. 2006 Jul;91(7):2592-9. doi: 10.1210/jc.2006-0448[↩]
- Wiersinga WM. Paradigm shifts in thyroid hormone replacement therapies for hypothyroidism. Nat Rev Endocrinol. 2014 Mar;10(3):164-74. doi: 10.1038/nrendo.2013.258[↩][↩]
- McAninch EA, Bianco AC. New insights into the variable effectiveness of levothyroxine monotherapy for hypothyroidism. Lancet Diabetes Endocrinol. 2015 Oct;3(10):756-8. doi: 10.1016/S2213-8587(15)00325-3[↩]
- McAninch EA, Jo S, Preite NZ, Farkas E, Mohácsik P, Fekete C, Egri P, Gereben B, Li Y, Deng Y, Patti ME, Zevenbergen C, Peeters RP, Mash DC, Bianco AC. Prevalent polymorphism in thyroid hormone-activating enzyme leaves a genetic fingerprint that underlies associated clinical syndromes. J Clin Endocrinol Metab. 2015 Mar;100(3):920-33. doi: 10.1210/jc.2014-4092[↩]
- Panicker V, Saravanan P, Vaidya B, Evans J, Hattersley AT, Frayling TM, Dayan CM. Common variation in the DIO2 gene predicts baseline psychological well-being and response to combination thyroxine plus triiodothyronine therapy in hypothyroid patients. J Clin Endocrinol Metab. 2009 May;94(5):1623-9. doi: 10.1210/jc.2008-1301[↩]
- Wouters HJ, van Loon HC, van der Klauw MM, et al. No effect of the Thr92Ala polymorphism of deiodinase-2 on thyroid hormone parameters, health-related quality of life, and cognitive functioning in a large population-based cohort study. Thyroid 2016; published online Oct 27 DOI: 10.1089/thy.2016.0199[↩]
- Wiersinga WM, Duntas L, Fadeyev V, Nygaard B, Vanderpump MP. 2012 ETA Guidelines: The Use of L-T4 + L-T3 in the Treatment of Hypothyroidism. Eur Thyroid J. 2012 Jul;1(2):55-71. doi: 10.1159/000339444[↩][↩]
- Mincer DL, Jialal I. Hashimoto Thyroiditis. [Updated 2022 Jun 21]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459262[↩][↩][↩][↩][↩][↩]
- Hashimoto’s Disease: What It Is and How It’s Treated. https://www.aafp.org/pubs/afp/issues/2000/0215/p1054.html[↩]
- Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, Mechanick JI, Pessah-Pollack R, Singer PA, Woeber KA; American Association of Clinical Endocrinologists and American Thyroid Association Taskforce on Hypothyroidism in Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012 Nov-Dec;18(6):988-1028. doi: 10.4158/EP12280.GL. Erratum in: Endocr Pract. 2013 Jan-Feb;19(1):175.[↩][↩]
- Mikulska AA, Karaźniewicz-Łada M, Filipowicz D, Ruchała M, Główka FK. Metabolic Characteristics of Hashimoto’s Thyroiditis Patients and the Role of Microelements and Diet in the Disease Management-An Overview. Int J Mol Sci. 2022 Jun 13;23(12):6580. doi: 10.3390/ijms23126580[↩][↩][↩][↩]
- Shukla S.K., Singh G., Ahmad S., Pant P. Infections, Genetic and Environmental Factors in Pathogenesis of Autoimmune Thyroid Diseases. Microb. Pathog. 2018;116:279–288. doi: 10.1016/j.micpath.2018.01.004[↩]
- Weetman A.P. An Update on the Pathogenesis of Hashimoto’s Thyroiditis. J. Endocrinol. Invest. 2021;44:883–890. doi: 10.1007/s40618-020-01477-1[↩]
- Ferrari S.M., Fallahi P., Antonelli A., Benvenga S. Environmental Issues in Thyroid Diseases. Front. Endocrinol. 2017;8:50. doi: 10.3389/fendo.2017.00050[↩]
- Wiersinga W.M. Clinical Relevance of Environmental Factors in the Pathogenesis of Autoimmune Thyroid Disease. Endocrinol. Metab. 2016;31:213–222. doi: 10.3803/EnM.2016.31.2.213[↩][↩][↩]
- Ralli M., Angeletti D., Fiore M., D’Aguanno V., Lambiase A., Artico M., de Vincentiis M., Greco A. Hashimoto’s Thyroiditis: An Update on Pathogenic Mechanisms, Diagnostic Protocols, Therapeutic Strategies, and Potential Malignant Transformation. Autoimmun. Rev. 2020;19:102649. doi: 10.1016/j.autrev.2020.102649[↩][↩][↩]
- Effraimidis G., Wiersinga W.M. Mechanisms in Endocrinology: Autoimmune Thyroid Disease: Old and New Players. Eur. J. Endocrinol. 2014;170:R241–R252. doi: 10.1530/EJE-14-0047[↩][↩][↩]
- Ajjan R.A., Weetman A.P. The Pathogenesis of Hashimoto’s Thyroiditis: Further Developments in Our Understanding. Horm. Metab. Res. 2015;47:702–710. doi: 10.1055/s-0035-1548832[↩][↩][↩][↩][↩][↩]
- Leung AKC, Leung AAC. Evaluation and management of the child with hypothyroidism. World J Pediatr. 2019 Apr;15(2):124-134. doi: 10.1007/s12519-019-00230-w[↩]
- Yuan J, Sun C, Jiang S, Lu Y, Zhang Y, Gao XH, Wu Y, Chen HD. The Prevalence of Thyroid Disorders in Patients With Vitiligo: A Systematic Review and Meta-Analysis. Front Endocrinol (Lausanne). 2019 Jan 15;9:803. doi: 10.3389/fendo.2018.00803[↩]
- Ott J, Promberger R, Kober F, Neuhold N, Tea M, Huber JC, Hermann M. Hashimoto’s thyroiditis affects symptom load and quality of life unrelated to hypothyroidism: a prospective case-control study in women undergoing thyroidectomy for benign goiter. Thyroid. 2011 Feb;21(2):161-7. doi: 10.1089/thy.2010.0191. Epub 2010 Dec 27. Erratum in: Thyroid. 2011 Apr;21(4):467.[↩]
- Brix TH, Hegedüs L, Gardas A, Banga JP, Nielsen CH. Monozygotic twin pairs discordant for Hashimoto’s thyroiditis share a high proportion of thyroid peroxidase autoantibodies to the immunodominant region A. Further evidence for genetic transmission of epitopic “fingerprints”. Autoimmunity. 2011 May;44(3):188-94. doi: 10.3109/08916934.2010.518575[↩]
- Klubo-Gwiezdzinska J., Wartofsky L. Hashimoto Thyroiditis: An Evidence-Based Guide to Etiology, Diagnosis and Treatment. Pol. Arch. Intern. Med. 2022;132:16222. doi: 10.20452/pamw.16222[↩]
- Kust D., Matesa N. The Impact of Familial Predisposition on the Development of Hashimoto’s Thyroiditis. Acta Clin. Belg. 2020;75:104–108. doi: 10.1080/17843286.2018.1555115[↩]
- Ihnatowicz P., Drywień M., Wątor P., Wojsiat J. The Importance of Nutritional Factors and Dietary Management of Hashimoto’s Thyroiditis. Ann. Agric. Environ. Med. 2020;27:184–193. doi: 10.26444/aaem/112331[↩][↩]
- Torino F., Barnabei A., Paragliola R., Baldelli R., Appetecchia M., Corsello S.M. Thyroid Dysfunction as an Unintended Side Effect of Anticancer Drugs. Thyroid. 2013;23:1345–1366. doi: 10.1089/thy.2013.0241[↩]
- Carlé A., Pedersen I.B., Knudsen N., Perrild H., Ovesen L., Rasmussen L.B., Jørgensen T., Laurberg P. Moderate Alcohol Consumption May Protect against Overt Autoimmune Hypothyroidism: A Population-Based Case-Control Study. Eur. J. Endocrinol. 2012;167:483–490. doi: 10.1530/EJE-12-0356[↩][↩]
- Effraimidis G., Tijssen J.G.P., Wiersinga W.M. Alcohol Consumption as a Risk Factor for Autoimmune Thyroid Disease: A Prospective Study. Eur. Thyroid J. 2012;1:99–104. doi: 10.1159/000338920[↩][↩]
- Effraimidis G., Tijssen J.G.P., Brosschot J.F., Wiersinga W.M. Involvement of Stress in the Pathogenesis of Autoimmune Thyroid Disease: A Prospective Study. Psychoneuroendocrinology. 2012;37:1191–1198. doi: 10.1016/j.psyneuen.2011.12.009[↩]
- Markomanolaki ZS, Tigani X, Siamatras T, Bacopoulou F, Tsartsalis A, Artemiadis A, Megalooikonomou V, Vlachakis D, Chrousos GP, Darviri C. Stress Management in Women with Hashimoto’s thyroiditis: A Randomized Controlled Trial. J Mol Biochem. 2019;8(1):3-12. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6688766[↩][↩]
- Rostami R., Nourooz-Zadeh S., Mohammadi A., Khalkhali H.R., Ferns G., Nourooz-Zadeh J. Serum Selenium Status and Its Interrelationship with Serum Biomarkers of Thyroid Function and Antioxidant Defense in Hashimoto’s Thyroiditis. Antioxidants. 2020;9:1070. doi: 10.3390/antiox9111070[↩]
- Mazokopakis EE, Papadomanolaki MG, Tsekouras KC, Evangelopoulos AD, Kotsiris DA, Tzortzinis AA. Is vitamin D related to pathogenesis and treatment of Hashimoto’s thyroiditis? Hell J Nucl Med. 2015 Sep-Dec;18(3):222-7.[↩]
- Wiebolt J., Achterbergh R., den Boer A., van der Leij S., Marsch E., Suelmann B., de Vries R., van Haeften T.W. Clustering of Additional Autoimmunity Behaves Differently in Hashimoto’s Patients Compared with Graves’ Patients. Eur. J. Endocrinol. 2011;164:789–794. doi: 10.1530/EJE-10-1172[↩]
- Ragusa F, Fallahi P, Elia G, Gonnella D, Paparo SR, Giusti C, Churilov LP, Ferrari SM, Antonelli A. Hashimotos’ thyroiditis: Epidemiology, pathogenesis, clinic and therapy. Best Pract Res Clin Endocrinol Metab. 2019 Dec;33(6):101367. doi: 10.1016/j.beem.2019.101367[↩][↩]
- Burch HB. Drug Effects on the Thyroid. N Engl J Med. 2019 Aug 22;381(8):749-761. doi: 10.1056/NEJMra1901214[↩]
- Kawicka A., Regulska-Ilow B., Regulska-Ilow B. Metabolic Disorders and Nutritional Status in Autoimmune Thyroid Diseases. Postepy Hig. Med. Doswiadczalnej Online. 2015;69:80–90. doi: 10.5604/17322693.1136383[↩]
- Szczuko M., Syrenicz A., Szymkowiak K., Przybylska A., Szczuko U., Pobłocki J., Kulpa D. Doubtful Justification of the Gluten-Free Diet in the Course of Hashimoto’s Disease. Nutrients. 2022;14:1727. doi: 10.3390/nu14091727[↩]
- Landete J.M. Dietary Intake of Natural Antioxidants: Vitamins and Polyphenols. Crit. Rev. Food Sci. Nutr. 2013;53:706–721. doi: 10.1080/10408398.2011.555018[↩]
- Ruggeri R.M., Giovinazzo S., Barbalace M.C., Cristani M., Alibrandi A., Vicchio T.M., Giuffrida G., Aguennouz M.H., Malaguti M., Angeloni C., et al. Influence of Dietary Habits on Oxidative Stress Markers in Hashimoto’s Thyroiditis. Thyroid. 2021;31:96–105. doi: 10.1089/thy.2020.0299[↩]
- Ostrowska L., Gier D., Zyśk B. The Influence of Reducing Diets on Changes in Thyroid Parameters in Women Suffering from Obesity and Hashimoto’s Disease. Nutrients. 2021;13:862. doi: 10.3390/nu13030862[↩][↩][↩]
- Avard N., Grant S. A Case Report of a Novel, Integrative Approach to Hashimoto’s Thyroiditis with Unexpected Results. Adv. Integr. Med. 2018;5:75–79. doi: 10.1016/j.aimed.2018.03.003[↩][↩][↩]
- Abbott R.D., Sadowski A., Alt A.G. Efficacy of the Autoimmune Protocol Diet as Part of a Multi-Disciplinary, Supported Lifestyle Intervention for Hashimoto’s Thyroiditis. Cureus. 2019;11:e4556. doi: 10.7759/cureus.4556[↩][↩]
- Al-Bayyari N.S. Successful Dietary Intervention Plan for Hashimoto’s Thyroiditis: A Case Study. Rom. J. Diabetes Nutr. Metab. Dis. 2020;27:381–385.[↩]