Contents
What is mannitol
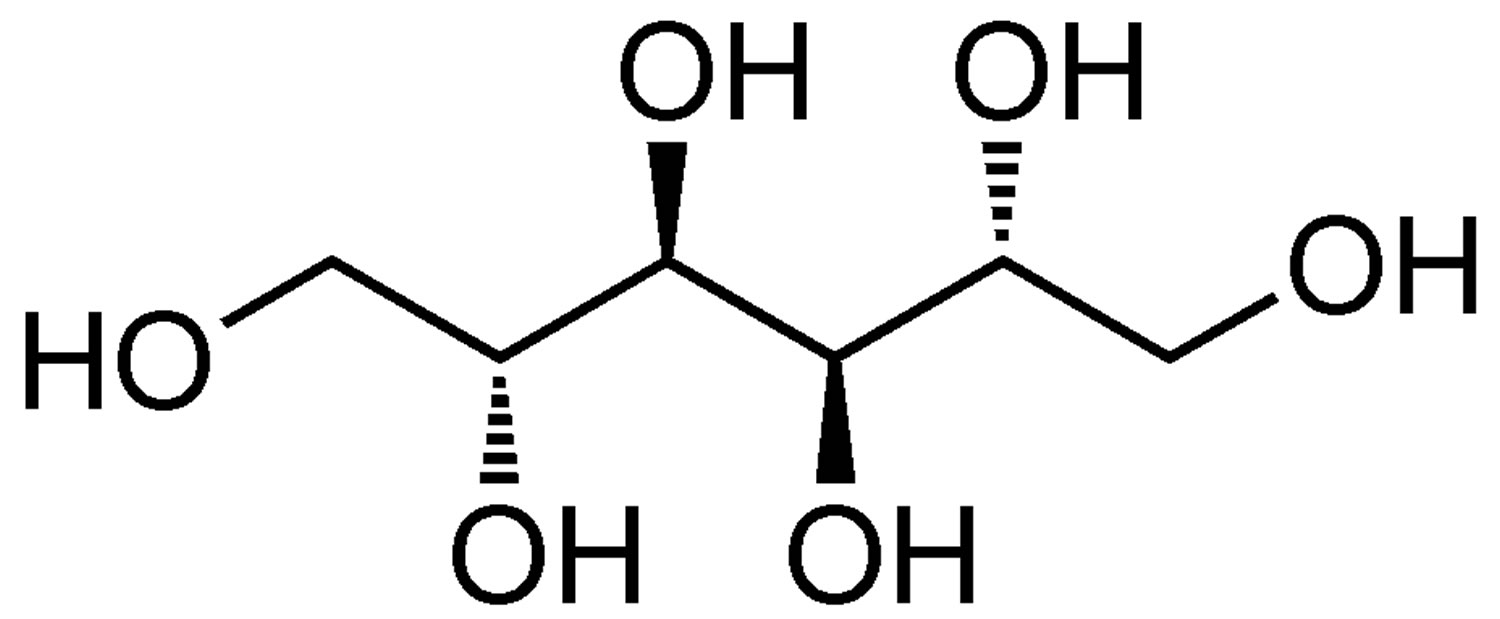
Mannitol is a naturally occurring alcohol found in fruits and vegetables and mannitol is used as an osmotic diuretic and a renal diagnostic aid. Mannitol therapy is widely used in the clinical setting for acute and subacute reduction in brain edema, to decrease muscle damage in compartment syndrome, and to improve renal perfusion 1. Mannitol is freely filtered by the kidney’s glomerulus and mannitol is poorly reabsorbed from the renal tubule, thereby causing an increase in osmolarity of the glomerular filtrate. An increase in osmolarity limits tubular reabsorption of water and inhibits the renal tubular reabsorption of sodium, chloride, and other solutes, thereby promoting diuresis. In addition, mannitol elevates blood plasma osmolarity, resulting in enhanced flow of water from tissues into interstitial fluid and plasma.
Osmotic diuresis is increased urination due to the presence of mannitol in the fluid filtered by the kidneys. This fluid eventually becomes urine. Mannitol causes additional water to come into the urine, increasing its amount.
Mannitol has little significant energy value as it is largely eliminated from the body before any metabolism can take place. Mannitol can be used to treat oliguria associated with kidney failure or other manifestations of inadequate renal function and has been used for determination of glomerular filtration rate (GFR). Mannitol is also commonly used as a research tool in cell biological studies, usually to control osmolarity.
Mannitol is removed by hemodialysis and peritoneal dialysis. These may be employed in the treatment of mannitol overdose. Eight patients with severe mannitol intoxication were treated. These patients had CNS (central nervous system) involvement out of proportion to uremia, severe hyponatremia, a large osmolality gap (high measured minus calculated serum osmolality), and fluid overload. Six patients were treated with hemodialysis and one patient received peritoneal dialysis 2. One patient died before any treatment could be started. Mannitol has a half-life of approximately 36 hours during the intervals without treatment. The ideal treatment is hemodialysis that rapidly removes mannitol (half-life, six hours) and replaces it with sodium; peritoneal dialysis removed mannitol slowly (half-life, 21 hours) 2.
Mannitol side effects
Get emergency medical help if you have any of these signs of an allergic reaction: hives; difficult breathing; swelling of your face, lips, tongue, or throat.
Tell your doctor right away if you have:
- swelling in your hands or feet;
- anxiety, sweating, severe shortness of breath, cough with foamy mucus, chest pain;
- painful or difficult urination;
- a light-headed feeling, like you might pass out;
- pain, burning, irritation, or skin changes where the injection was given;
- dehydration symptoms–feeling very thirsty or hot, being unable to urinate, heavy sweating, or hot and dry skin;
- signs of an electrolyte imbalance–dry mouth, increased thirst, confusion, fast heart rate, increased urination, muscle pain or weakness, feeling light-headed, fainting, or seizure (convulsions); or
- signs of a kidney problem–little or no urinating; painful or difficult urination; swelling in your feet or ankles; feeling tired or short of breath.
Common side effects may include:
- increased urination;
- nausea, vomiting;
- fever, chills, headache, runny nose;
- swelling, rapid weight gain;
- chest pain;
- skin rash; or
- dizziness, blurred vision.
Figure 1. Mannitol
Mannitol mechanism of action
The mechanism of action of mannitol is as an osmotic agent 3. The physiologic effect of mannitol is by means of increased diuresis.
Mannitol, when administered intravenously, exerts its osmotic effect as a solute of relatively small molecular size being largely confined to the extracellular space. Only relatively small amounts of the dose administered is metabolized. Mannitol is readily diffused through the glomerulus of the kidney over a wide range of normal and impaired kidney function. In this fashion, approximately 80% of a 100 gram dose of mannitol will appear in the urine in three hours with lesser amounts thereafter. Even at peak concentrations, mannitol will exhibit less than 10% of tubular reabsorption and is not secreted by tubular cells. Mannitol will hinder tubular reabsorption of water and enhance excretion of sodium and chloride by elevating the osmolarity of the glomerular filtrate.
This increase in extracellular osmolarity effected by the intravenous administration of mannitol will induce the movement of intracellular water to the extracellular and vascular spaces. This action underlies the role of mannitol in reducing intracranial pressure, intracranial edema, and elevated intraocular pressure.
Mannitol also acts as a bronchoconstrictor and may cause severe bronchospasm. Mannitol inhalation is used in patients 6 years of age and older to help diagnose asthma. It is used in a procedure called bronchial challenge test to help your doctor measure the effect of this medicine on your lungs and check if you have difficulty with breathing.
Mannitol precautions
General precautions
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during parenteral therapy with a mannitol solution.
Mannitol solution should be used with care in patients with hypervolemia, renal insufficiency, urinary tract obstruction, or impending or frank cardiac decompensation.
The cardiovascular status of the patient should be carefully evaluated before rapidly administering mannitol since sudden expansion of the extracellular fluid may lead to fulminating congestive heart failure.
Shifting of sodium-free intracellular fluid into the extracellular compartment following mannitol infusion may lower serum sodium concentration and aggravate preexisting hyponatremia.
Mannitol administration may obscure and intensify inadequate hydration or hypovolemia by sustaining diuresis.
Electrolyte-free Mannitol Injection should not be given conjointly with blood. If it is essential that blood be given simultaneously, at least 20 mEq of sodium chloride should be added to each liter of mannitol solution to avoid pseudoagglutination. In no other instance should additions be made to 20% Mannitol Injection USP. The addition of sodium chloride to 20% mannitol solution may result in precipitation of mannitol. The final infusate should therefore be inspected for cloudiness or precipitation immediately after mixing, prior to administration, and periodically during administration.
Solutions of mannitol may crystallize when exposed to low temperatures. Concentrations greater than 15% have a greater tendency to crystallization. Inspect for crystals prior to administration. If crystals are observed, the container should be warmed by appropriate means to not greater than 60°C, shaken, then cooled to body temperature before administering. If all crystals cannot be completely redissolved, the container must be rejected. Administer intravenously using sterile, filter-type administration set.
Do not use plastic containers in series connection.
If administration is controlled by a pumping device, care must be taken to discontinue pumping action before the container runs dry or air embolism may result. If administration is not controlled by a pumping device, refrain from applying excessive pressure (>300mmHg) causing distortion to the container such as wringing or twisting. Such handling could result in breakage of the container.
This solution is intended for intravenous administration using sterile equipment. It is recommended that intravenous administration apparatus be replaced at least once every 24 hours.
Use only if solution is clear and container and seals are intact.
Laboratory Tests
Although blood levels of mannitol can be measured, there is little if any clinical virtue in doing so. The appropriate monitoring of blood levels of sodium and potassium; degree of hemoconcentration or hemodilution, if any; indices of renal, cardiac and pulmonary function are paramount in avoiding excessive fluid and electrolyte shifts. The routine features of physical examination and clinical chemistries suffice in achieving an adequate degree of appropriate patient monitoring.
Carcinogenesis, mutagenesis and impairment of fertility
Long term studies in animals to evaluate the carcinogenic and mutagenic potential or the effect on fertility of 20% Mannitol Injection USP have not been conducted.
Pregnancy Teratogenic Effects
Pregnancy Category C. Animal reproduction studies have not been conducted with 20% Mannitol Injection USP. It is also not known whether 20% Mannitol Injection USP can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. 20% Mannitol Injection USP should be given to a pregnant woman only if clearly needed.
Breastfeeding
It is not known whether mannitol is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when 20% Mannitol Injection USP is administered to a nursing woman.
Pediatric Use
Safety and effectiveness in children below the age of 12 years have not been established.
Usage in Children
Dosage requirements for patients 12 years of age and under have not been established.
Geriatric Use
Clinical studies of 20% Mannitol Injection USP did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
Mannitol is known to be substantially excreted by the kidney, and the risk of toxic reactions to mannitol may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
What is mannitol used for?
Mannitol is used as a diuretic. Mannitol is used to force urine production in people with acute (sudden) kidney failure. As a diuretic it can be used to treat patients with intractable edema states, to increase urine flow and flush out debris from the renal tubules in patients with acute tubular necrosis, and to increase toxin excretion in patients with barbiturate, salicylate or bromide intoxication 4. Increased urine production helps to keep the kidneys from shutting down, and also speeds up elimination of certain toxic substances in the body.
Mannitol is also used to reduce swelling and pressure inside the eye or around the brain.
Mannitol may be useful clinically both as a diuretic and as an obligate extracellular solute. As an obligate extracellular solute it may be useful to ameliorate symptoms of the dialysis disequilibrium syndrome, to decrease cerebral edema following trauma or cerebrovascular accident, and to prevent cell swelling related to renal ischemia following cross-clamping of the aorta. Largely unexplored uses for mannitol include its use as an osmotic agent in place of dextrose in peritoneal dialysis solutions, its use to maintain urine output in patients newly begun on hemodialysis, and its use to limit infarct size following acute myocardial infarction.
Mannitol inhalation is used in patients 6 years of age and older to help diagnose asthma. Mannitol is used in a procedure called bronchial challenge test to help your doctor measure the effect of this medicine on your lungs and check if you have difficulty with breathing 5. Aridol is a test kit containing one single patient use inhaler and 3 blister packs containing 19 capsules of mannitol for inhalation in marked doses to perform one bronchial challenge test. It is given by a doctor or other trained health professional who will be with you during the test. After you have completed the test, your doctor will know the result right away (positive or negative for asthma). You should not receive this medicine if you have had an allergic reaction to mannitol or gelatin. You should not receive this medicine if you have other conditions that may cause bronchospasm (lung spasms with breathing problems) such as a heart or blood vessel problem, high blood pressure that is not controlled, or a recent heart attack or stroke.
As a sugar, mannitol is often used as a sweetener in diabetic food, as it is poorly absorbed from the intestines 6. Mannitol increases blood glucose to a lesser extent than sucrose (thus having a relatively low glycemic index) so is used as a sweetener for people with diabetes, and in chewing gums 7.
Mannitol Injection
20% Mannitol Injection USP (United States Pharmacopeia) treats early kidney failure by increasing urination. This helps your body get rid of extra fluids. 20% Mannitol Injection USP treats brain swelling and increased pressure in the eye. Also treats poisoning by increasing urination to remove toxins from the body.
- Promotion of diuresis, in the prevention and/or treatment of the oliguric phase of acute renal failure before irreversible renal failure becomes established.
- Reduction of intracranial pressure and treatment of cerebral edema by reducing brain mass.
- Reduction of elevated intraocular pressure when the pressure cannot be lowered by other means.
- Promotion of urinary excretion of toxins.
You should not receive Mannitol Injection if you have severe dehydration (fluid loss). You should not receive Mannitol Injection if you have kidney failure and you have completely stopped passing urine. You should not use Mannitol Injection if you have bleeding problems in your brain. You should stop Mannitol Injection if it has not made your kidney failure better. You should stop Mannitol Injection if it has caused you to have heart failure or fluid in the lungs.
In patients with severe impairment of renal function, a test dose should be utilized. A second test dose may be tried if there is an inadequate response, but no more than two test doses should be attempted.
The obligatory diuretic response following rapid infusion of 20% Mannitol Injection USP may further aggravate preexisting hemoconcentration. Excessive loss of water and electrolytes may lead to serious imbalances. Serum sodium and potassium should be carefully monitored during mannitol administration.
If urine output continues to decline during mannitol infusion, the patient’s clinical status should be closely reviewed and mannitol infusion suspended if necessary. Accumulation of mannitol may result in overexpansion of the extracellular fluid which may intensify existing or latent congestive heart failure.
Excessive loss of water and electrolytes may lead to serious imbalances. With rapid or prolonged administration of mannitol, loss of water in excess of electrolytes can cause hypernatremia. Electrolyte measurements including sodium and potassium are therefore of vital importance in monitoring the infusion of mannitol.
Osmotic nephrosis, a reversible vacuolization of the tubules of no known clinical significance, may proceed to severe irreversible nephrosis, requiring close monitoring during mannitol infusion.
Mannitol contraindications
Mannitol is considered contraindicated in patients with well established anuria (no urine output) due to acute renal failure, severe pulmonary congestion or frank pulmonary edema, active intracranial bleeding (except during craniotomy), and severe dehydration.
20% Mannitol Injection USP (United States Pharmacopeia) is contraindicated in patients with:
- Well-established anuria due to severe renal disease.
- Severe pulmonary congestion or frank pulmonary edema.
- Active intracranial bleeding except during craniotomy.
- Severe dehydration.
- Progressive renal damage or dysfunction after institution of mannitol therapy, including increasing oliguria and azotemia.
- Progressive heart failure or pulmonary congestion after institution of mannitol therapy.
Mannitol therapy should be discontinued if progression in renal damage or dysfunction, heart failure, or pulmonary congestion occurs.
A test dose should be administered in patients with severe renal impairment.
Diuresis caused by mannitol administration may exacerbate electrolyte imbalances. Electrolytes should be closely monitored, especially sodium and potassium, during mannitol administration.
Urine output should be monitored during mannitol infusion. If output declines, consideration should be given to possible discontinuation of mannitol therapy.
Neurosurgical patients receiving intravenous mannitol should be monitored for increased cerebral blood flow.
Mannitol should not be administered until renal function and urinary output are determined to be adequate.
Cardiovascular status should be established prior to rapid administration of mannitol.
Do not administer electrolyte-free mannitol solutions with blood.
Bronchial challenge testing with mannitol inhalation should not be performed in children less than 6 years of age due to their inability to provide reliable spirometric measurements.
Mannitol powder by inhlation
You should not receive this medicine if you have had an allergic reaction to mannitol or gelatin. You should not receive this medicine if you have other conditions that may cause bronchospasm (lung spasms with breathing problems) such as a heart or blood vessel problem, high blood pressure that is not controlled, or a recent heart attack or stroke.
Mannitol dose
Mannitol by injection
Mannitol is injected into a vein through an IV. A healthcare provider will give you this injection.
Mannitol must be given slowly through an IV infusion, and you may receive the medication around the clock.
To be sure mannitol is helping your condition and not causing harmful effects, your blood will need to be tested often. This will help your doctor determine how long to treat you with mannitol. Your heart function will also need to be tested.
Adult dose for oliguria
A nurse or other health provider will give you this medicine. Your doctor will prescribe your dose and schedule. This medicine is given through a needle placed in a vein.
Renal function test dose prior to initiation of treatment: 0.2 g/kg IV over 3 to 5 min resulting in a urine flow of at least 30 to 50 mL/hr. A second test dose may be administered if the urine flow does not increase. If no response is seen following the second test dose, the patient should be reevaluated.
Treatment: 300 to 400 mg/kg (21 to 28 g for a 70 kg patient) or up to 100 g of 15% to 20% solution IV once. Treatment should not be repeated in patients with persistent oliguria.
Prevention (for use during cardiovascular and other types of surgery): 50 to 100 g IV. usually a 5 , 10, or 20% solution is used depending on the fluid requirements of the patient.
Adult dose for cerebral edema
0.25 to 2 g/kg as a 15 to 20% solution IV over at least 30 min administered not more frequently than every 6 to 8 hrs.
To yield a satisfactory reduction in intracranial pressure, the osmotic gradient between the blood and cerebrospinal fluid (CSF) should remain approximately 20 mOsmol.
In small and/or debilitated patients 500 mg/kg may be sufficient.
Renal dose adjustments
Do not initiate treatment until renal function and urinary output have been established as adequate. Mannitol is considered contraindicated in patients with well established anuria due to acute renal failure.
A test dose should be administered in patients with severe renal impairment.
Urine output should be monitored during mannitol infusion. If output declines, consideration should be given to possible discontinuation of mannitol therapy.
Liver dose adjustments
Data not available
How to use mannitol inhalation capsule
Mannitol inhalation is used in patients 6 years of age and older to help diagnose asthma. It is used in a procedure called bronchial challenge test to help your doctor measure the effect of this medicine on your lungs and check if you have difficulty with breathing.
Mannitol inhalation is to be given only by or under the immediate supervision of your doctor.
Mannitol acts as a bronchoconstrictor and may cause severe bronchospasm. Mannitol bronchial challenge testing is for diagnostic purposes only and should be conducted only by trained professionals under a physician familiar with the test and management of acute bronchospasm (ie, testing area equipped with appropriate medications and equipment). Immediately administer a short-acting inhaled beta-agonist in the event of severe bronchospasm. Do not perform bronchial challenge testing with mannitol in a patient with asthma or very low baseline pulmonary function tests (e.g, FEV1 less than 1 to 1.5 L or less than 70% of the predicted values) .
This medicine is used with a special inhaler which will measure the effect of Aridol™ on your lungs. It is given by a doctor or other trained health professional who will be with you during the test. After you have completed the test, your doctor will know the result right away (positive or negative for asthma).
Aridol™ is a test kit containing one single patient use inhaler and 3 blister packs containing 19 capsules of mannitol for inhalation in marked doses to perform one bronchial challenge test.
Do NOT put the capsules in your mouth or swallow them.
To perform the test:
- Before doing the bronchial challenge test, your doctor may ask you to perform a breathing or lung test (such as spirometry test).
- A nose clip will then be put on your nose so you will only able to breathe in and out of your mouth.
- Place 0 mg capsule into the inhaler. Press the side buttons of the inhaler once to puncture the capsule.
- To inhale this medicine, breathe out fully, trying to get as much air out of the lungs as possible. Put the inhaler just in front of your mouth.
- Open your mouth and breathe in slowly and deeply (like yawning).
- Hold your breath for about 5 seconds, then breathe out slowly before removal of the nose clip.
- You will be asked to repeat the above steps up to 8 times (total of 9 increasing doses of Aridol™). This is to measure the effect of Aridol™ in your lungs.
- Once you have finished the test, you will be given a short-acting inhaler to help you breathe (for patients who have a positive result).
- Throw away the inhaler after using.
Mannitol side effects
Applies to mannitol powder inhalation capsule
Along with its needed effects, mannitol may cause some unwanted effects. Although not all of these side effects may occur, if they do occur they may need medical attention.
Check with your doctor or nurse immediately if any of the following side effects occur while taking mannitol 8:
Less Common
- chest discomfort
- cough
- difficult or labored breathing
- dry heaves
- runny nose
- shortness of breath
- sore throat
- tightness in the chest
- troubled breathing
- vomiting
- wheezing
Incidence not known
- Gagging
Some side effects of mannitol may occur that usually do not need medical attention. These side effects may go away during treatment as your body adjusts to the medicine. Also, your health care professional may be able to tell you about ways to prevent or reduce some of these side effects. Check with your health care professional if any of the following side effects continue or are bothersome or if you have any questions about them:
More Common
- Headache
Less Common
- dizziness
- nausea
- sore throat
Applies to mannitol compounding powder, inhalation kit, intravenous solution and irrigation solution
Call your doctor right away if you notice any of these side effects:
- Allergic reaction: Itching or hives, swelling in your face or hands, swelling or tingling in your mouth or throat, chest tightness, trouble breathing
- Chest pain, arm pain, or a heartbeat that is fast or uneven.
- Confusion, muscle weakness, or muscle cramps.
- Constipation (hard dry stools that are less often than usual) or belly pain.
- Coughing or shortness of breath.
- Fever or chills.
- Loss of feeling or tingling anywhere in your body.
- No increase in passing urine, trouble passing urine, or a decrease in how much or how often you pass urine.
- Seizures.
- Skin rash.
- Swelling in your hands, ankles, or feet.
- Swelling, redness, or pain where the needle was placed.
If you notice these less serious side effects, talk with your doctor:
- Blurred vision or problems with your eyes.
- Dizziness, headache, or fainting.
- Dry mouth or increased thirst.
- Nausea or vomiting.
- Runny nose.
If you notice other side effects that you think are caused by this medicine, tell your doctor.
Call your doctor for medical advice about side effects.
Cardiovascular
Cardiovascular side effects have included hypotension and tachycardia. Venous thrombosis or phlebitis extending from the injection site and hypervolemia have occurred rarely and are generally associated with the solution or technique used in administration.
Respiratory
Respiratory side effects have included pulmonary congestion and rhinitis. Respiratory side effects associated with mannitol inhalation have included cough, gagging, wheeze, and decreased forced expiratory volume.
Metabolic
Metabolic side effects have included fluid and electrolyte imbalance, acidosis, and electrolyte loss.
Nervous system
Nervous system side effects have included headache, convulsions, and dizziness.
Hematologic
Hematologic side effects have included thrombophlebitis.
Other
Eight cases of mannitol IV overdose in patients with preexisting renal failure were reviewed 2. Symptoms presented in the reviewed cases were CNS involvement out of proportion to uremia, severe hyponatremia, large osmolality gap, and fluid overload. Six patients were treated with hemodialysis, one patient with peritoneal dialysis, and one patient died before initiation of treatment. All patients received large doses over 1 to 3 days with a mean dose of 310 +/- 182.8 g. CNS involvement consisted of CNS depression, confusion, lethargy, stupor, and coma 2. Two patients recovered cerebral function and continued on lifetime dialysis, 3 patients recovered renal and cerebral function, and 1 patient recovered renal function but had severe cerebral dysfunction 2.
Other side effects have included dryness of mouth, thirst, edema, arm pain, chills, dehydration, fever, mannitol intoxication, and angina-like pain.
Kidney
Renal side effects have included acute renal failure 9.
Local adverse reactions
Local side effects have included extravasation. This effect is generally attributed to the solution or technique used in administration.
Skin
Dermatologic side effects have included urticaria and skin necrosis.
Eyes
Ocular side effects have included blurred vision.
Immunologic
Immunologic side effects have included infection at the injection site and febrile response. These effects are usually attributed to solution or technique used in administration.
Gastrointestinal
Gastrointestinal side effects have included nausea and vomiting.
Genitourinary
Genitourinary side effects have included marked diuresis and urinary retention.
Psychiatric
Psychiatric side effects have rarely included mania (1 case report).
A 75-year-old woman with severe major depression experienced a manic episode 30 minutes after initiation of a 20% mannitol intravenous infusion for the treatment of acute angle closure glaucoma 10. The patient had been started on nortriptyline 50 mg per day for the treatment of depression ten days earlier. She received oral acetazolamide, topical pilocarpine, topical timolol, and topical dexamethasone concomitantly for the treatment of glaucoma. The mania resolved within approximately 1 hour following discontinuation of the mannitol infusion, and the patient returned to a severe depressive state. An extensive lab evaluation, toxicology screening, and medical examination failed to show additional secondary causes for mania in this patient.
Interactions with medicines
Although certain medicines should not be used together at all, in other cases two different medicines may be used together even if an interaction might occur. In these cases, your doctor may want to change the dose, or other precautions may be necessary. When you are receiving this diagnostic test, it is especially important that your healthcare professional know if you are taking any of the medicines listed below. The following interactions have been selected on the basis of their potential significance and are not necessarily all-inclusive.
Receiving this diagnostic test with any of the following medicines is usually not recommended, but may be required in some cases. If both medicines are prescribed together, your doctor may change the dose or how often you use one or both of the medicines.
- Arsenic Trioxide
- Droperidol
- Levomethadyl
- Sotalol
- Tobramycin
Receiving this diagnostic test with any of the following medicines may cause an increased risk of certain side effects, but using both drugs may be the best treatment for you. If both medicines are prescribed together, your doctor may change the dose or how often you use one or both of the medicines.
- Licorice
Other Interactions
Certain medicines should not be used at or around the time of eating food or eating certain types of food since interactions may occur. Using alcohol or tobacco with certain medicines may also cause interactions to occur. Discuss with your healthcare professional the use of your medicine with food, alcohol, or tobacco.
- Mannitol at clinical concentrations activates multiple signaling pathways and induces apoptosis in endothelial cells. Stroke. 1998 Dec;29(12):2631-40. https://www.ncbi.nlm.nih.gov/pubmed/9836777/[↩]
- Borges HF, Hocks J, Kjellstrand CM. Mannitol Intoxication in Patients With Renal Failure. Arch Intern Med. 1982;142(1):63–66. doi:10.1001/archinte.1982.00340140065013[↩][↩][↩][↩][↩]
- Le TN, Blakley BW. Mannitol and the blood-labyrinth barrier. Journal of Otolaryngology – Head & Neck Surgery. 2017;46:66. doi:10.1186/s40463-017-0245-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5725891/[↩]
- Nissenson AR, Weston RE, Kleeman CR. Mannitol. Western Journal of Medicine. 1979;131(4):277-284. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1271822/pdf/westjmed00242-0017.pdf[↩]
- Mannitol (By breathing). https://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0011032[↩]
- Sweeteners: Nutritional Aspects, Applications, and Production Technology. CRC Press. pp. 59–60. ISBN 9781439876732[↩]
- Grenby, T.H (2011) Advances in Sweeteners. Springer. ISBN 1461285224. p. 66[↩]
- Mannitol (Inhalation route). https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0045434/#DDIC603407.side_effects_section[↩]
- Doi K, Ogawa N, Suzuki E, Noiri E, Fujita T “Mannitol-induced acute renal failure.” Am J Med 115 (2003): 593-4[↩]
- Navarro V, Vieta E, Gasto C “Mannitol-induced acute manic state.” J Clin Psychiatry 62 (2001): 126[↩]