Contents
What is mucormycosis
Mucormycosis previously called zygomycosis, is the unifying term used to describe rare opportunistic life-threatening infections of the sinuses, brain, or lungs caused by fungi or molds called mucormycetes belonging to the order Mucorales 1. These molds live throughout the environment. Mucormycosis fungal infection occurs in some people with a weakened immune system or immunosuppressed individuals. Rhizopus, Mucor, and Rhizomucor species account for up to 75% of mucormycosis cases encountered in hematologic malignancy patients 2. Mucormycosis mainly affects people with weakened immune systems and can occur in nearly any part of the body. Mucormycosis most commonly affects the sinuses or the lungs after inhaling fungal spores from the air, or the skin after the fungus enters the skin through a cut, scrape, burn, or other type of skin trauma. A classic clinical sign of mucormycosis is the rapid onset of tissue necrosis with or without fever. Necrosis is the result of invasion of blood vessels and subsequent thrombosis.
Mucorales typically cause acute, aggressive, and frequently angioinvasive infections, especially in immunosuppressed hosts with hematologic malignancy and/or stem cell transplantation 3.
People get mucormycosis by coming in contact with the fungal spores in the environment. For example, the lung or sinus forms of the infection can occur after someone inhales the spores from the air. These forms of mucormycosis usually occur in people who have weakened immune systems. Mucormycosis can also develop on the skin after the fungus enters the skin through a cut, scrape, burn, or other type of skin trauma.
Mucormycosis can affect nearly any part of the body, but it most commonly affects the sinuses or the lungs in people who have weakened immune systems 4:
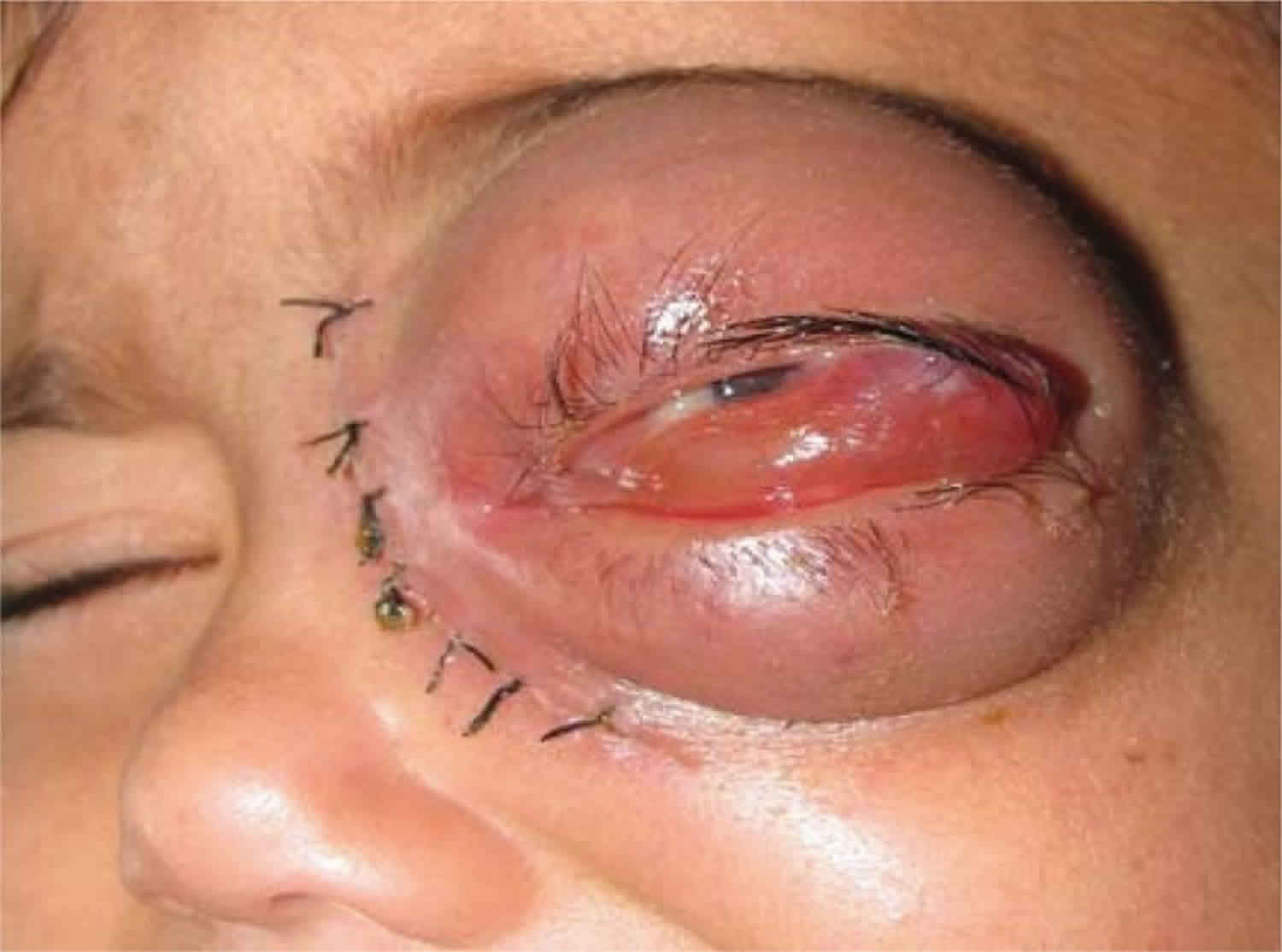
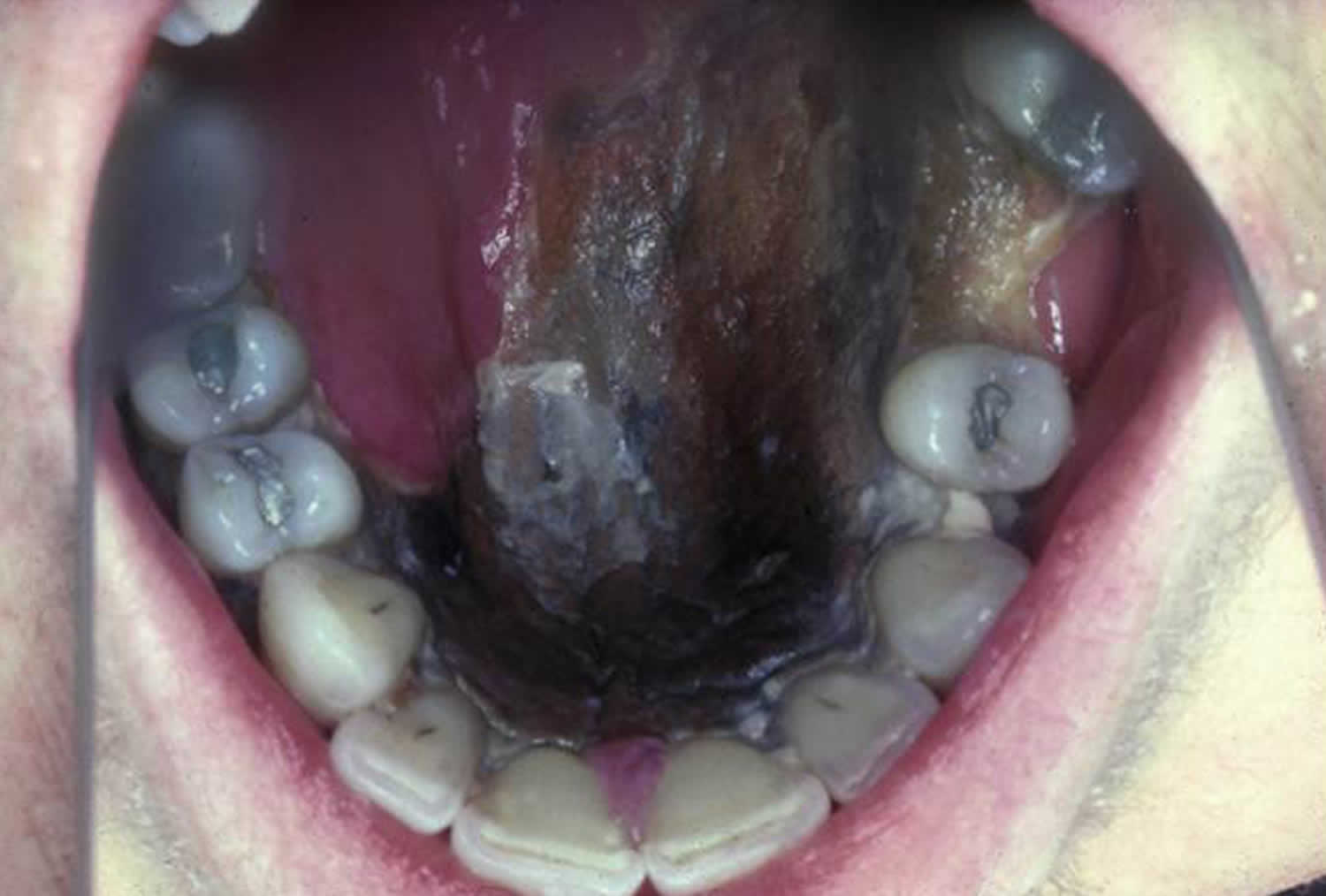
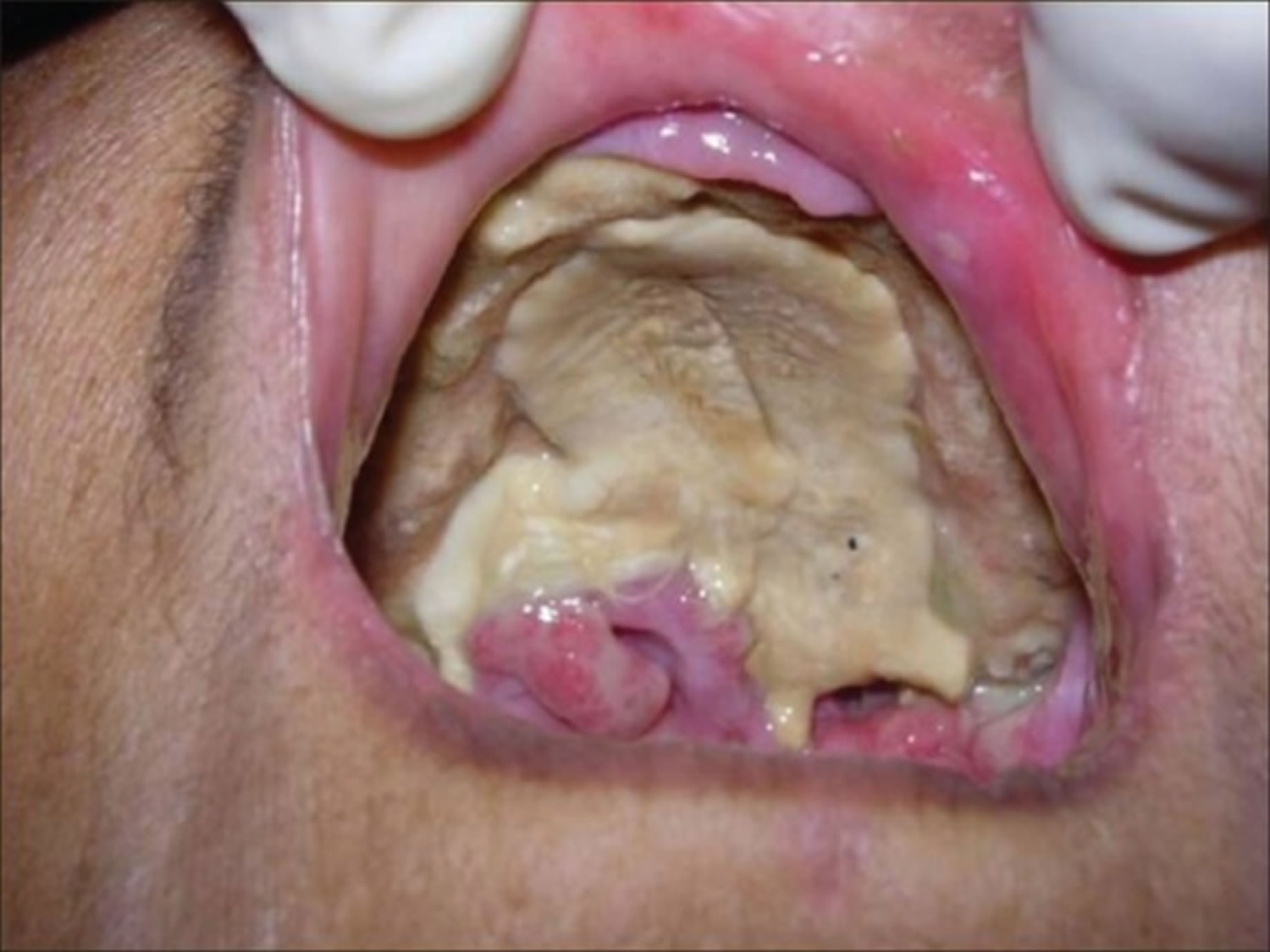
- A sinus and brain infection called rhinocerebral infection: It may start as a sinus infection, and then lead to the swelling of the nerves that stem from the brain. It may also cause blood clots that block vessels to the brain. Rhinocerebral mucormycosis is associated with immunocompromised patient state, hemochromatosis, desferrioxamine therapy, malignancy, diabetes mellitus with or without ketoacidosis, organ transplantation, severe burns, trauma and prolonged corticosteroid therapy 5. Rhinocerebral mucormycosis is the most common form in patients with diabetes. It also occurs in neutropenic cancer patients and hematopoietic stem cell transplant or solid organ transplant recipients. Symptoms may include unilateral facial swelling, headaches, nasal or sinus congestion or pain, serosanguinous nasal discharge, and fever. As the infection spreads, ptosis, proptosis, loss of extraocular muscle function, and vision disturbance may occur. Necrotic black lesions on the hard palate or nasal turbinate and drainage of black pus from eyes are useful diagnostic signs. Because rhinocerebral mucormycosis occurs infrequently it may pose a diagnostic and therapeutic problems for those who are not familiar with its clinical presentation. Early clinical recognition of this potentially fatal disease followed by aggressive debridement, systemic antifungal therapy, and control of underlying co-morbid factors is the mainstay of therapy 6.
- A lung infection called pulmonary mucormycosis: pneumonia gets worse quickly and may spread to the chest cavity, heart, and brain. Pulmonary (lung) mucormycosis is the most common type of mucormycosis in people with hematologic cancer and in people who have had an organ transplant, profound neutropenia or a stem cell transplant. The symptoms are non-specific and include fever, cough, chest pain, and dyspnea. Angioinvasion results in tissue necrosis, which may ultimately lead to cavitation and/or hemoptysis.
- Gastrointestinal mucormycosis can result from ingestion of the fungal spores. This type of mucormycosis is less common among adults and is more common among young children, especially infants <1 month of age. The stomach, colon, and ileum are most commonly affected. Non-specific abdominal pain and distension, nausea, and vomiting are the most common symptoms, and gastrointestinal bleeding can occur. It is the most common form of mucormycosis among neonates and is challenging to diagnose partly because of its clinical resemblance to necrotizing enterocolitis, a far more common disease 7.
- Cutaneous (skin) mucormycosis: occurs after the fungi enter the body through a break in the skin (for example, after surgery, a burn, or other type of skin trauma). This is the most common form of mucormycosis among people who do not have weakened immune systems. Cutaneous mucormycosis may be primary or secondary. Primary infection is usually caused by direct inoculation of the fungus into disrupted skin and is most often seen in patients with burns or other forms of local skin trauma. Primary infection produces an acute inflammatory response with pus, abscess formation, tissue swelling, and necrosis. The lesions may appear red and indurated and often progress to black eschars. Secondary cutaneous infection is generally seen when the pathogen spreads hematogenously; lesions typically begin as an erythematous, indurated, and painful cellulitis and then progress to an ulcer covered with a black eschar.
- Disseminated mucormycosis occurs when the infection spreads through the bloodstream to affect another part of the body. The brain is the most commonly affected part of the body, but other organs such as the spleen, heart, and skin can also be affected. Disseminated mucormycosis may follow any of the forms of mucormycosis described above but is usually seen in neutropenic patients with a pulmonary infection.
The predisposing factors for mucormycosis in patients with hematologic cancer are similar to ones encountered in other opportunistic mold infections, such as aspergillosis (Aspergillus species), including profound and protracted neutropenia and monocytopenia, chronic high-dose corticosteroids, reactivation of opportunistic herpes viruses (especially cytomegalovirus), severe graft-versus-host disease and its treatment, active hematologic malignancy, and its associated functional neutropenia, as well as and high-risk stem cell transplantation 8. In addition, uncontrolled hyperglycemia, frank diabetes mellitus and/or diabetic ketoacidosis, and iron overload (associated with multiple transfusions of blood products for thalassemias or leukemic states) appear important contributing factors to increase mucormycosis risk 9, although the relative contribution of each condition to mycormycosis risk is unknown.
As mucormycosis cases are typically seen in high-risk leukemia and allogeneic stem cell transplantation recipients 10, it remains unclear what effects prolonged voriconazole exposure may have in patients to reduce the threshold of breakthrough mucormycosis 1. Given the multiple, interrelated risk factors for invasive fungal infections present in most patients with mucormycosis, it is often impossible to ascribe a single factor that increases the risk and or worsens the prognosis of this devastating infection 11.
Figure 1. Rhinocerebral mucormycosis (necrotic lesion of the hard palate in patient with rhinocerebral mucormycosis)
Figure 2. Rhinocerebral mucormycosis
People with weakened immune systems and immune disorders (including diabetes) should seek medical attention if they develop:
- Fever
- Headache
- Sinus pain
- Eye swelling
- Any of the other symptoms listed below
Who gets mucormycosis?
Overall, mucormycosis is rare, but it’s more common among people with weakened immune systems than among people who are otherwise healthy. Certain groups of people are at higher risk for developing mucormycosis, including people with 12:
- Diabetes, especially with diabetic ketoacidosis
- Cancer
- Organ transplant
- Stem cell transplant
- Neutropenia (low number of white blood cells)
- Long-term corticosteroid use
- Illicit injection drug use
- Too much iron in the body
- Skin trauma (due to surgery, burns, or other skin injuries)
- Prematurity and low birthweight (for gastrointestinal mucormycosis)
How does someone get mucormycosis?
People get mucormycosis by coming in contact with fungal spores in the environment. For example, the lung or sinus forms of the infection can occur after someone inhales the spores from the air. Mucormycosis can develop on the skin after the fungus enters the skin through a cut, scrape, burn, or other type of skin trauma.
Although most cases are sporadic, healthcare-associated outbreaks have been linked to adhesive bandages, wooden tongue depressors, hospital linens, and building construction 13. Community-onset outbreaks have been associated with trauma sustained during natural disasters 14.
Is mucormycosis contagious?
No. Mucormycosis can’t spread between people or between people and animals.
How can I lower the risk of mucormycosis?
It’s difficult to avoid breathing in fungal spores because the fungi that cause mucormycosis are common in the environment. There is no vaccine to prevent mucormycosis. For people who have weakened immune systems, there may be some ways to lower the chances of developing mucormycosis.
- Protect yourself from the environment 15. It’s important to note that although these actions are recommended, they haven’t been proven to prevent mucormycosis.
- Try to avoid areas with a lot of dust like construction or excavation sites. If you can’t avoid these areas, wear an N95 respirator (a type of face mask) while you’re there. Go here for more information about respirators (https://www.cdc.gov/niosh/topics/respirators/).
- Avoid activities that involve close contact to soil or dust, such as yard work or gardening. If this isn’t possible,
- Wear shoes, long pants, and a long-sleeved shirt when doing outdoor activities such as gardening, yard work, or visiting wooded areas.
- Wear gloves when handling materials such as soil, moss, or manure.
- To reduce the chances of developing a skin infection, clean skin injuries well with soap and water, especially if they have been exposed to soil or dust.
- Antifungal medication. If you are at high risk for developing mucormycosis (for example, if you’ve had an organ transplant or a stem cell transplant), your healthcare provider may prescribe medication to prevent mucormycosis and other mold infections 16. Doctors and scientists are still learning about which transplant patients are at highest risk and how to best prevent fungal infections.
Mucormycosis causes
Mucormycosis is caused by fungi, members of the Mucoraceae family including the Rhizopus, Mucor, Rhizomucor, Cunninghamella bertholletiae, Saksenaea, Apophysomyces species, and Lichtheimia (formerly Absidia) species, that are often found in decaying organic matter 17. These include decaying food such as spoiled bread, fruit, and vegetables, as well as soil, other organic matter, such as animal excreta and compost piles, leaves or rotten wood 18. Mucormycosis fungi have the ability to rapidly grow and release large numbers of spores that become airborne and gain entrance to the human body through inhalation or ingestion 19. Most people come in contact with the fungus at some time. However, it rarely causes an infection in one with an intact immune system which can phagocytize the spores 20.
However, people with a weakened immune system are more likely to develop mucormycosis. These include people with any of the following conditions:
- AIDS
- Burns
- Diabetes (usually poorly controlled)
- Leukemia and lymphoma
- Long-term steroid use
- Metabolic acidosis
- Poor nutrition (malnutrition)
- Use of some medicines
Risk factors for mucormycosis
Predisposing risk factors for mucormycosis in patients with hematologic malignancies and/or stem cell transplantation 1:
- Prolonged (> 3 weeks) and severe (absolute neutrophil count. < 200) neutropenia
- Monocytopenia (< 100 mm³)
- Prolonged (> 3 weekk) high-dose systemic corticosteroids (e.g, prednisone or equivalent of > 1 mg/kg/day)
- Iron overload (assessed by high iron indices, high iron storage by MRI, or high iron staining in bone marrow biopsy)
- High-risk stem cell transplantation (e.g., matched-unrelated donor stem cell transplantation, haploidentical donor stem cell transplantation, cord blood stem cell transplantation, T cell-depleted stem cell transplantation)
- Severe graft-versus-host-disease and its treatment (especially corticosteroids)
- Prolonged hyperglycemia (fasting serum glucose > 200 mg/dL), corticosteroid-associated hyperglycemia, diabetes mellitus
- Colonization by mucormycetes or heavy environmental exposure?
- Previous exposure to Aspergillus-active antifungal agents, especially voriconazole? 21
- Relapsed leukemia
Mucormycosis symptoms
The symptoms of mucormycosis depend on where in the body the fungus is growing 22. Mucormycosis is rare and typically affects people with weakened immune systems. Contact your healthcare provider if you have symptoms that you think are related to mucormycosis.
Symptoms of rhinocerebral mucormycosis include:
- One-sided facial swelling
- Eyes that swell and stick out (protrude)
- Dark scabbing in nasal cavities
- Black lesions on upper inside of mouth
- Fever
- Headache
- Mental status changes
- Redness of skin above sinuses
- Sinus pain or sinus congestion
Symptoms of lung (pulmonary) mucormycosis include:
- Cough
- Chest pain
- Coughing blood (occasionally)
- Fever
- Shortness of breath
Symptoms of gastrointestinal mucormycosis include:
- Abdominal pain
- Blood in the stools
- Diarrhea
- Vomiting blood
Symptoms of kidney (renal) mucormycosis include:
- Fever
- Pain in the upper abdomen or back
Symptoms of skin (cutaneous) mucormycosis can look like blisters or ulcers, and the infected area may turn black. Other symptoms include pain, warmth, excessive redness, or swelling around a wound.
Disseminated mucormycosis typically occurs in people who are already sick from other medical conditions, so it can be difficult to know which symptoms are related to mucormycosis. Patients with disseminated infection in the brain can develop mental status changes or coma.
Mucormycosis possible complications
These complications may occur:
- Blindness (if the optic nerve is involved)
- Clotting or blockage of brain or lung blood vessels
- Death
- Nerve damage
Mucormycosis diagnosis
Healthcare providers consider your medical history, symptoms, physical examinations, and laboratory tests when diagnosing mucormycosis. See an ear-nose-throat (ENT) doctor if you are having sinus problems. If your healthcare provider suspects that you have mucormycosis in your lungs or sinuses, he or she might collect a sample of fluid from your respiratory system to send to a laboratory. Your healthcare provider may perform a tissue biopsy, in which a small sample of affected tissue is analyzed in a laboratory for evidence of mucormycosis under a microscope or in a fungal culture. You may also need imaging tests such as a CT scan of your lungs, sinuses, or other parts of your body depending on the location of the suspected infection.
Testing depends on your symptoms, but may include these imaging tests:
- CT scans
- MRI scans
Mucormycosis treatment
Mucormycosis is a serious infection and needs to be treated with prescription antifungal medication, usually amphotericin B, posaconazole or isavuconazole. These medications are given through a vein (amphotericin B, posaconazole, isavuconazole) or by mouth (posaconazole, isavuconazole). Often, mucormycosis requires surgery right away to remove all dead and infected tissue, particularly for rhinocerebral, cutaneous, and gastrointestinal infections 7. Surgery can lead to disfiguration because it may involve removal of the palate, parts of the nose, or parts of the eye. But, without such aggressive surgery, chances of survival are greatly decreased.
Amphotericin B, posaconazole, and isavuconazole are active against most mucormycetes. Lipid formulations of amphotericin B are often used as first-line treatment 4. In addition, control of the underlying immunocompromising condition should be attempted when possible 4. The efficacy of other treatments such as hyperbaric oxygen therapy is uncertain but have been useful in certain situations 23.
Mucormycosis prognosis
Early recognition, diagnosis, and prompt administration of appropriate antifungal treatment are important for improving outcomes for patients with mucormycosis 4. Mucormycosis has a very high extremely high death rate, even when aggressive surgery is done. Risk of death depends on the area of the body involved and your overall health.
The overall prognosis depends on several factors, including the rapidity of diagnosis and treatment, the site of infection, and the patient’s underlying conditions and degree of immunosuppression. The overall mortality rate is approximately 50% 17, although early identification and treatment can lead to better outcomes 12.
- Kontoyiannis DP, Lewis RE. How I treat mucormycosis. Blood. 2011;118(5):1216-24. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3292433/[↩][↩][↩]
- Neofytos D, Horn D, Anaissie E, et al. Epidemiology and outcome of invasive fungal infection in adult hematopoietic stem cell transplant recipients: analysis of Multicenter Prospective Antifungal Therapy (PATH) Alliance registry. Clin Infect Dis. 2009;48(3):265–273[↩]
- Chamilos G, Lewis RE, Kontoyiannis DP. Delaying amphotericin B-based frontline therapy significantly increases mortality among patients with hematologic malignancy who have zygomycosis. Clin Infect Dis. 2008;47(4):503–509[↩]
- Lewis RE, Kontoyiannis DP. Epidemiology and treatment of mucormycosis. Future microbiology. 2013 Sep;8(9):1163-75.[↩][↩][↩][↩]
- Prabhu Rm, Patel R. Mucormycosis and entomophthoramycosis: A review of the clinical manifestations, diagnosis and treatment. Clin Microbiol Infect. 2004;10:31–47.[↩]
- Onyango JF, Kayima JK, Owen WO. Rhinocerebral mucormycosis: Case report. East Afr Med J. 2002;79:390–3[↩]
- Vallabhaneni S, Mody RK. Gastrointestinal Mucormycosis in Neonates: a Review. Current Fungal Infection Reports. 2015.[↩][↩]
- Kontoyiannis DP, Lewis RE. Invasive zygomycosis: update on pathogenesis, clinical manifestations, and management. Infect Dis Clin North Am. 2006;20(3):581–607[↩]
- Sujobert P, Boissel N, Bergeron A, et al. Breakthrough zygomycosis following empirical caspofungin treatment: report of two patients with leukemia and literature review. Open J Hematol. 2010:1–3.[↩]
- Kontoyiannis DP, Lionakis MS, Lewis RE, et al. Zygomycosis in a tertiary-care cancer center in the era of Aspergillus-active antifungal therapy: a case-control observational study of 27 recent cases. J Infect Dis. 2005;191(8):1350–1360[↩]
- Pongas GN, Lewis RE, Samonis G, Kontoyiannis DP. Voriconazole-associated zygomycosis: a significant consequence of evolving antifungal prophylaxis and immunosuppression practices? Clin Microbiol Infect. 2009;15(50):93–97[↩]
- Walsh TJ, Gamaletsou MN, McGinnis MR, Hayden RT, Kontoyiannis DP. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis. 2012 Feb;54 Suppl 1:S55-60.[↩][↩]
- Duffy J, Harris J, Gade L, Sehulster L, Newhouse E, O’Connell H, et al. Mucormycosis outbreak associated with hospital linens. Pediatr Infect Dis J. 2014 May;33(5):472-6.[↩]
- Neblett Fanfair R, Benedict K, Bos J, Bennett SD, Lo YC, Adebanjo T, et al. Necrotizing cutaneous mucormycosis after a tornado in Joplin, Missouri, in 2011. New Engl J Med. 2012 Dec 6;367(23):2214-25.[↩]
- Avery RK, Michaels MG. Strategies for safe living after solid organ transplantation. Am J Transplant. 2013 Mar;13 Suppl 4:304-10.[↩]
- Brizendine KD, Vishin S, Baddley JW. Antifungal prophylaxis in solid organ transplant recipients. Expert Rev Anti Infect Ther. 2011 May;9(5):571-81.[↩]
- Roden MM, Zaoutis TE, Buchanan WL, Knudsen TA, Sarkisova TA, Schaufele RL, et al. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis. 2005 Sep 1;41(5):634-53.[↩][↩]
- Kwon-Chung KJ. Taxonomy of Fungi Causing Mucormycosis and Entomophthoramycosis (Zygomycosis) and Nomenclature of the Disease: Molecular Mycologic Perspectives. Clin Infect Dis. 2012;54:S8–15[↩]
- Mohammadi R, Nazeri M, Sayedayn SM, Ehteram H. A successful treatment of rhinocerebral mucormycosis due to Rhizopus oryzae. J Res Med Sci. 2014;19(1):72-4. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3963327/[↩]
- Casqueiro J, Casqueiro J, Alves C. Infections in patients with diabetes mellitus: A review of pathogenesis. Indian J Endocrinol Metab. 2012;16:S27–36.[↩]
- Trifilio S, Singhal S, Williams S, et al. Breakthrough fungal infections after allogeneic hematopoietic stem cell transplantation in patients on prophylactic voriconazole. Bone Marrow Transplant. 2007;40(5):451–456[↩]
- Petrikkos G, Skiada A, Lortholary O, Roilides E, Walsh TJ, Kontoyiannis DP. Epidemiology and clinical manifestations of mucormycosis. Clin Infect Dis. 2012 Feb;54 Suppl 1:S23-34.[↩]
- John BV, Chamilos G, Kontoyiannis DP. Hyperbaric oxygen as an adjunctive treatment for zygomycosis. Clin Microbiol Infect. 2005 Jul;11(7):515-7.[↩]