Contents
What is paracentesis
Paracentesis is also called abdominal tap, is a medical procedure in which a needle or catheter is inserted into the peritoneal cavity (the area between the belly wall and the spine) to obtain ascitic fluid for diagnostic or therapeutic purposes 1. Paracentesis procedure may be done in a health care provider’s office, treatment room, or hospital. Ascitic fluid may be used to help determine the cause of ascites, as well as to evaluate for infection or presence of cancer. Ascites is the build-up of fluid in the space between the lining of the abdomen and abdominal organs.
Normally, the abdominal cavity contains only a small amount of fluid. In certain conditions, large amounts of fluid can build up in this space.
Paracentesis can help diagnose the cause of fluid buildup (ascites) or the presence of an infection. Paracentesis may also be done to remove a large amount of fluid to reduce belly pain.
There are 2 kinds of paracentesis:
- Diagnostic paracentesis — A small amount of fluid is taken and sent to the laboratory for testing.
- Large volume paracentesis — Several liters may be removed to relieve abdominal pain and fluid buildup.
Paracentesis should be performed to:
- Rule out spontaneous bacterial peritonitis in patients with known ascites presenting with concerning symptoms such as abdominal pain, fever, gastrointestinal bleed, worsening encephalopathy, new or worsening renal or liver failure, hypotension, or other symptoms of infection or sepsis
- Identify the cause of new-onset ascites
- Alleviate abdominal discomfort or respiratory distress in hemodynamically stable patients with tense ascites or ascites that are refractory to diuretics (large volume therapeutic paracentesis)
Causes of ascites
Ascites results from high pressure in the blood vessels of the liver (portal hypertension) and low levels of a protein called albumin.
Diseases that can cause severe liver damage can lead to ascites. These include long-term hepatitis C or B infection and alcohol abuse over many years, and more and more frequently, fatty liver disease (non-alcoholic steatohepatitis).
People with certain cancers in the abdomen may develop ascites. These include cancer of the appendix, colon, ovaries, uterus, pancreas, and liver.
Other conditions that can cause this ascites include:
- Clots in the veins of the liver (portal vein thrombosis)
- Congestive heart failure
- Pancreatitis
- Thickening and scarring of the sac-like covering of the heart
Kidney dialysis may also be linked to ascites.
Causes of transudative ascites include the following:
- Hepatic cirrhosis
- Alcoholic hepatitis
- Heart failure
- Fulminant hepatic failure
- Portal vein thrombosis
Causes of exudative ascites include the following:
- Peritoneal carcinomatosis
- Inflammation of the pancreas or biliary system
- Nephrotic syndrome
- Peritonitis
- Ischemic or obstructed bowel
Abdominal paracentesis
Diagnostic paracentesis is used for the following 2:
- New-onset ascites – Fluid evaluation helps to determine etiology, differentiate transudate versus exudate, detect the presence of cancerous cells, or address other considerations
- Suspected spontaneous or secondary bacterial peritonitis
- Refractory ascites
Therapeutic paracentesis is used for the following:
- Respiratory compromise secondary to ascites
- Abdominal pain or pressure secondary to ascites (including abdominal compartment syndrome)
A report by Huang et al 3 found that abdominal paracentesis drainage brought about clinical improvement in patients who had non-hypertriglyceridemia-induced severe acute pancreatitis with triglyceride elevation and pancreatitis-associated ascitic fluid.
Large-volume paracentesis is often required in patients with refractory ascites. A report by Bureau et al 4 described the use of a low-flow pump system that moves the fluid from the abdominal cavity into the bladder, from which it is removed via micturition. This was shown to improve patients’ quality of life and reduced the need for repeated large-volume paracentesis.
Dietary sodium restriction and diuretics do not often provide symptomatic relief of refractory ascites in patients in advanced stages of cancer. Although paracentesis does effectively drain ascitic fluid, the condition invariably recurs, and repeated procedures are necessary. A 2008 study reported that a permanent peritoneal catheter to drain abdominal fluid greatly reduced the symptoms of ascites in these patients and avoided the costs and complications of frequent paracentesis procedures 5.
A meta-analysis suggests that the use of albumin in cirrhotic patients undergoing paracentesis reduces paracentesis-induced circulatory dysfunction and reduces death and renal impairment 6.
Paracentesis contraindications
There are few absolute contraindications for paracentesis. Coagulopathy and thrombocytopenia (both very common in cirrhotic patients) are themselves not absolute contraindications as the incidence of bleeding complications from the paracentesis procedure has been shown to be very low.
Severe thrombocytopenia (platelet count less than 20 × 10³/μL) and coagulopathy (international normalized ratio [INR] >2.0) are relative contraindications.
Patients with an international normalized ratio [INR] greater than 2.0 should receive fresh frozen plasma (FFP) prior to the procedure. One strategy is to infuse one unit of fresh frozen plasma before the paracentesis procedure and then perform the procedure while the second unit is infusing.
Patients with a platelet count lower than 20 × 10³/μL should receive an infusion of platelets before the procedure.
In patients without clinical evidence of active bleeding, routine laboratory tests such as prothrombin time (PT), activated partial thromboplastin time (aPTT), and platelet counts may not be needed before the procedure 7. In these patients, pretreatment with fresh frozen plasma (FFP), platelets, or both before paracentesis is also probably not needed.
A study of 608 patients (72% with alcohol-related liver disease) found a low overall rate of complications that required transfusions (0.2%) and a higher incidence of significant hemoglobin drop among those with severe renal failure (creatinine > 6 mg/dL) 7.
A prospective study of 171 patients undergoing paracentesis found that “major” complications occurred in 1.6% of procedures and included five episodes of bleeding and three infections, resulting in death in two cases. Major complications were associated with therapeutic paracentesis but not diagnostic paracentesis procedures and tended to be more prevalent in patients with low platelet counts (9 × 10³/μL), patients who were Child-Pugh stage C, and patients with alcoholic cirrhosis 8.
Paracentesis should be AVOIDED in patients with:
- Disseminated intravascular coagulation (DIC) is a serious disorder in which the proteins that control blood clotting become overactive (consider first administering platelets or fresh frozen plasma)
- An acute abdomen that requires surgery is an absolute contraindication.
It should be performed with caution in:
- Pregnant patients
- Patients with organomegaly, ileus, bowel obstruction or a distended bladder
- Distended urinary bladder
- Abdominal wall cellulitis
- Distended bowel
- Intra-abdominal adhesions
Avoid passing the needle/catheter through sites of skin infection, surgical scars, visibly engorged abdominal wall vessels or abdominal wall hematomas.
Paracentesis procedure
How to prepare for paracentesis
Let your healthcare provider know if you:
- Have any allergies to medicines or numbing medicine
- Are taking any medicines (including herbal remedies)
- Have any bleeding problems
- Might be pregnant
Paracentesis equipment includes the following:
- Antiseptic swab sticks
- Fenestrated drape
- Lidocaine 1%, 5-mL ampule
- Syringe, 10 mL
- Injection needles, 22-gauge (two)
- Injection needle, 25-gauge
- Scalpel, No. 11 blade
- Catheter, 8 French, over 18-gauge × 7.5-in. needle with three-way stopcock, self-sealing valve, and a 5-mL Luer-Lok syringe
- Syringe, 60 mL
- Introducer needle, 20-gauge
- Tubing set with roller clamp
- Drainage bag or vacuum container
- Specimen vials or collection bottles (three)
- Gauze, 4 × 4 in.
- Adhesive dressing
You’ll be instructed to empty your bladder before starting the paracentesis procedure, either through voluntary emptying on the part of the patient or through the use of a Foley catheter.
Paracentesis position
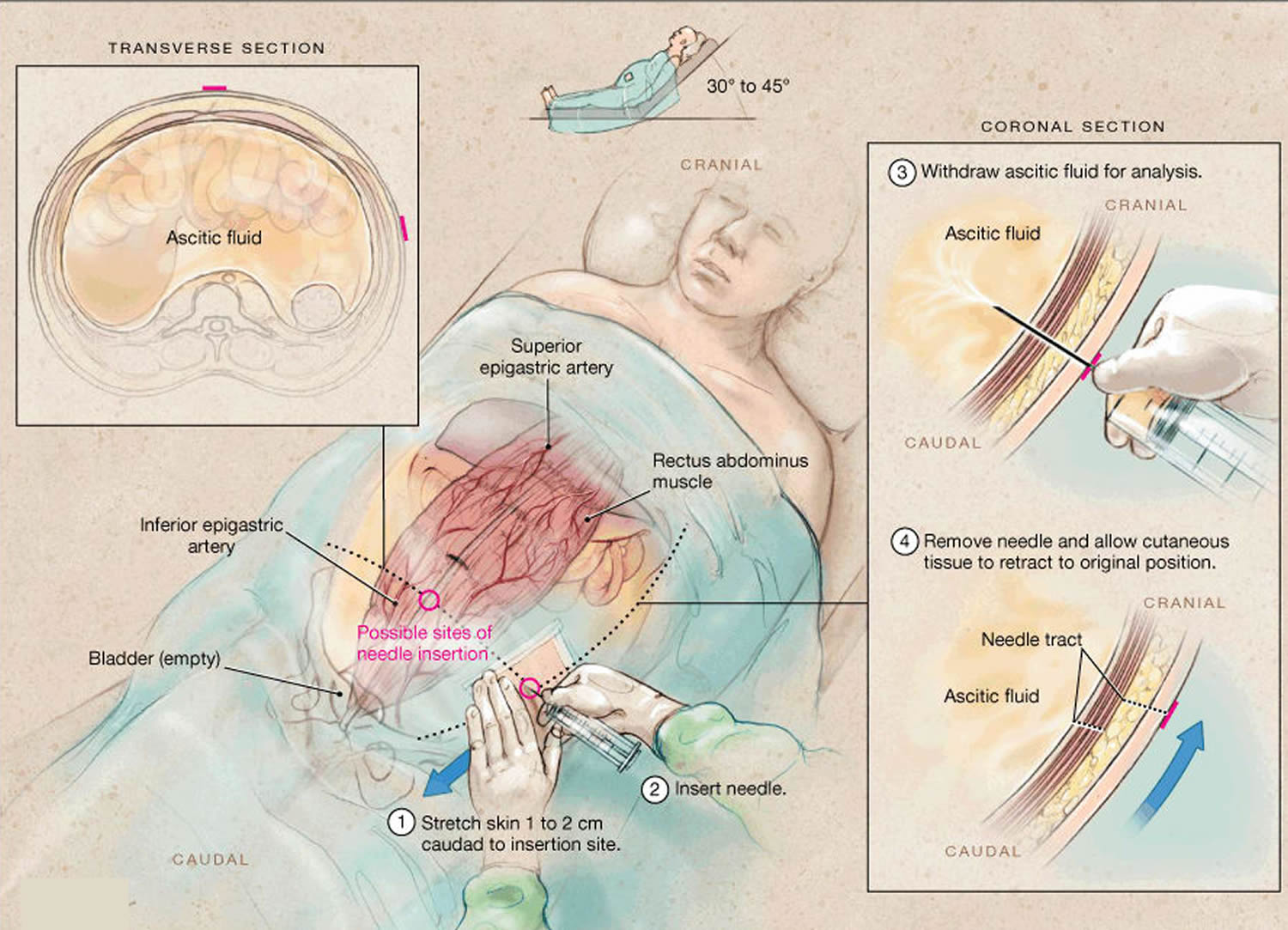
The preferred site for the procedure is in either lower quadrant of the abdomen lateral to the rectus sheath. Paracentesis is done in a lateral decubitus or supine position.
- Placing the patient in the lateral decubitus position can aid in identifying fluid pockets in patients with mild ascites (lower fluid volumes), with the skin entry site near the gurney. The lateral decubitus position is advantageous because air-filled loops of bowel tend to float in a distended abdominal cavity.
- Patients with severe ascites can be positioned supine.
The two recommended areas of abdominal wall entry for paracentesis are as follows (see the Figures 1 and 2 below):
- 2 cm below the umbilicus in the midline (through the linea alba)
- 2 cm to 5 cm superior and medial to the anterior superior iliac spines on either side, lateral to rectus abdominis muscle
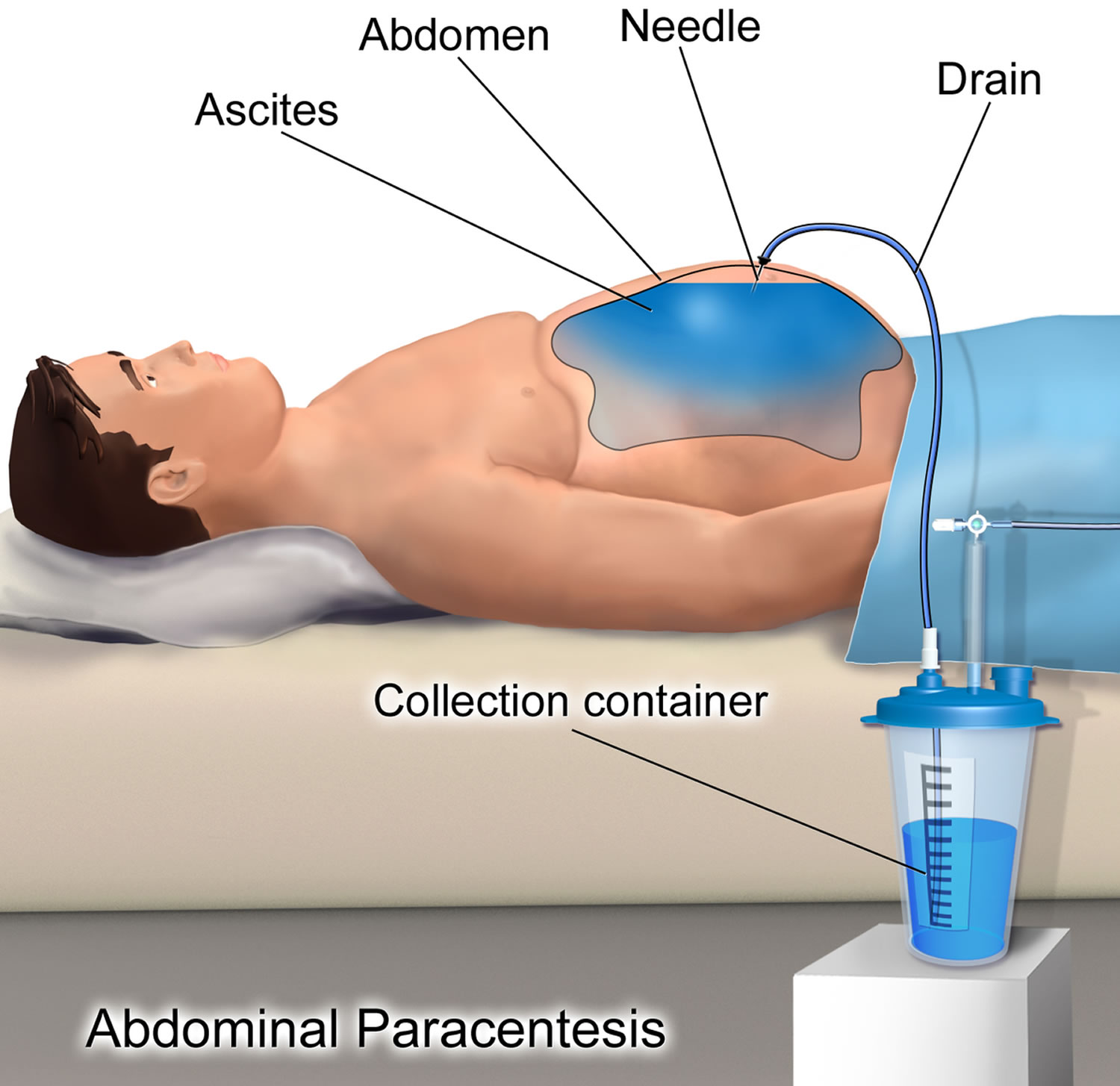
The puncture site will be cleaned and shaved, if necessary. You then receive a local numbing medicine. The ascites fluid level is percussed, and a needle is inserted either in the midline or lateral lower quadrant (2 cm to 5 cm superomedial to anterior superior iliac spine). The paracentesis needle is inserted 1 to 2 inches (2.5 to 5 cm) into the abdomen. Sometimes, a small cut is made to help insert the needle. The fluid is pulled out into a syringe. The needle is removed. A dressing is placed on the puncture site. If a cut was made, one or two stitches may be used to close it.
- This positioning avoids puncture of the inferior epigastric arteries
- Avoid visible superficial veins and surgical scars.
- The needle is inserted at a 45-degree angle or with a z-tracking technique to reduce the risk of developing an ascites fluid leak.
- After proper antiseptic preparation and local anesthesia, a diagnostic paracentesis tap can be performed with a 10- to 20-mL syringe and an 18-gauge needle.
- After proper antiseptic preparation and local anesthesia, a therapeutic paracentesis tap can be performed with an intravenous (IV) catheter over the needle connected to drainage tubing.
The authors 9 recommend the routine use of ultrasonography to verify the presence of a fluid pocket under the selected entry site in order to increase the rate of success (see Figures 3 and 4 below).
The use of ultrasonography also helps the practitioner avoid a distended urinary bladder or small bowel adhesions below the selected entry point. To minimize complications, avoid areas of prominent veins (caput medusae), infected skin, or scar tissue.
Paracentesis Technique
Prep and drape the patient in a sterile fashion. Cleanse the skin with an antiseptic solution. Administer local anesthesia to the skin and soft tissue (down to peritoneum) at the planned site of needle or catheter insertion.
Bedside ultrasound is sometimes used to identify an appropriate location for the paracentesis procedure. Ultrasound can confirm the presence of fluid and identify an area with a sufficient amount of fluid for aspiration, thereby decreasing the incidence of both unsuccessful aspiration and complications. Ultrasound increases the success rate of paracentesis and helps to prevent an unnecessary invasive procedure in some patients. The procedure can be performed either after marking the site of insertion or in real time by advancing the needle under direct ultrasound guidance.
Insert the traditional needle or IV catheter attached to a syringe or the prepackaged catheter directly perpendicular to the skin or using the z-track method which is thought to decrease the chance of fluid leakage after the procedure. This method entails puncturing the skin then pulling it caudally before advancing the needle through the soft tissue and peritoneum. If using a catheter kit, it may be helpful to make a small nick in the skin using an 11-blade scalpel to be able to advance the catheter through the skin and soft tissue smoothly. Apply negative pressure to the syringe during needle or catheter insertion until a loss of resistance is felt and a steady flow of ascitic fluid is obtained. This is paramount to detect unwanted entry into a vessel or other structure rapidly. Advance the catheter over the needle into the peritoneal cavity. In general, avoid advancing the needle deeper than the safety mark present on most commercially available catheters or deeper than 1 cm beyond the depth at which ascitic fluid was noticed in the lidocaine syringe.
Use one hand to firmly anchor the needle and syringe securely in place to prevent the needle from entering further into the peritoneal cavity. Use the other hand to hold the stopcock and catheter and advance the catheter over the needle and into the peritoneal cavity all the way to the skin. If any resistance is noticed, the catheter was probably misplaced into the subcutaneous tissue. If this is the case, withdraw the device completely and reattempt insertion. When withdrawing the device, always remove the needle and catheter together as a unit in order to prevent the bevel from cutting the catheter.
While holding the stopcock, pull the needle out. The self-sealing valve prevents fluid leak. Attach the 60-mL syringe to the three-way stopcock and aspirate to obtain ascitic fluid, and distribute it to the specimen vials. Use the three-way valve as needed to control fluid flow and prevent leakage when no syringe or tubing is attached. Connect one end of the fluid collection tubing to the stopcock and the other end to a vacuum bottle or a drainage bag.
After you collect sufficient fluid in the syringe for fluid analysis, either remove the traditional needle (if performing a diagnostic paracentesis tap) or connect the collecting tubing to it or the catheter’s stopcock to drain larger volumes of fluid into a vacuum container, plastic canister or a drainage bag. After you have drained the desired amount of fluid, remove the catheter and hold pressure to stop any bleeding from insertion site.
A study by Kelil et al 10 demonstrated that the use of wall suction and plastic canisters to drain and collect fluid during image-guided therapeutic paracenteses was a safe alternative to the use of evacuated glass bottles and reduced per-procedure costs.
The catheter can become occluded by a loop of bowel or omentum. If the flow stops, kink or clasp the tubing to avert loss of suction, then break the seal and manipulate the catheter slightly, and finally reconnect and see if flow resumes. Rotating the catheter about the long axis can sometimes reinstitute flow in models with side ports.
After the desired amount of ascitic fluid has been drained, remove the catheter. Apply firm pressure to stop bleeding, if present. Place a bandage over the skin puncture site.
How the paracentesis procedure will feel
You may feel a slight sting from the numbing medicine, or pressure as the needle is inserted.
If a large amount of fluid is taken out, you may feel dizzy or lightheaded. Tell the provider if you feel dizzy or lightheaded.
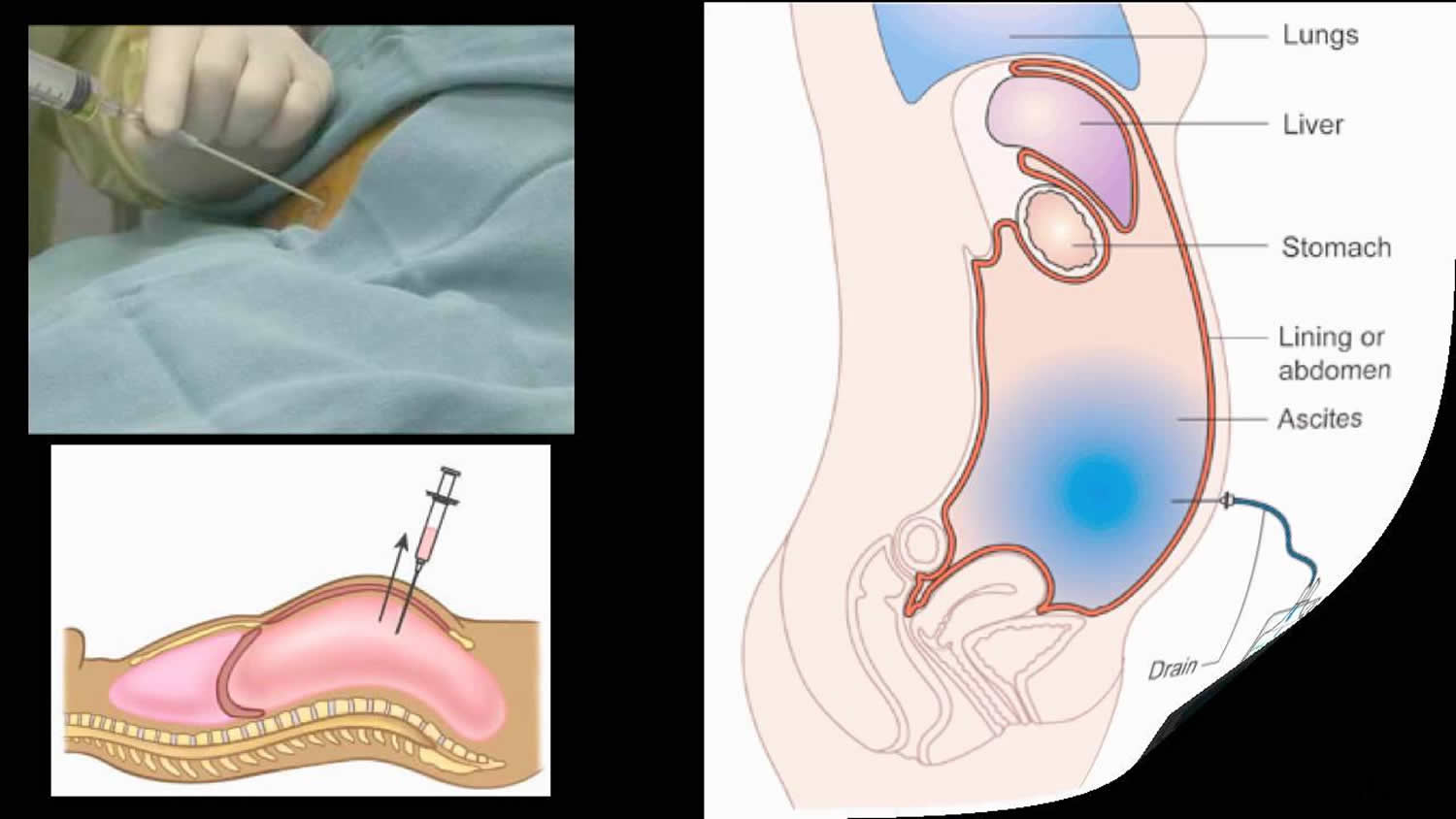
Figure 1. Paracentesis procedure
Figure 2. Paracentesis standard puncture sites
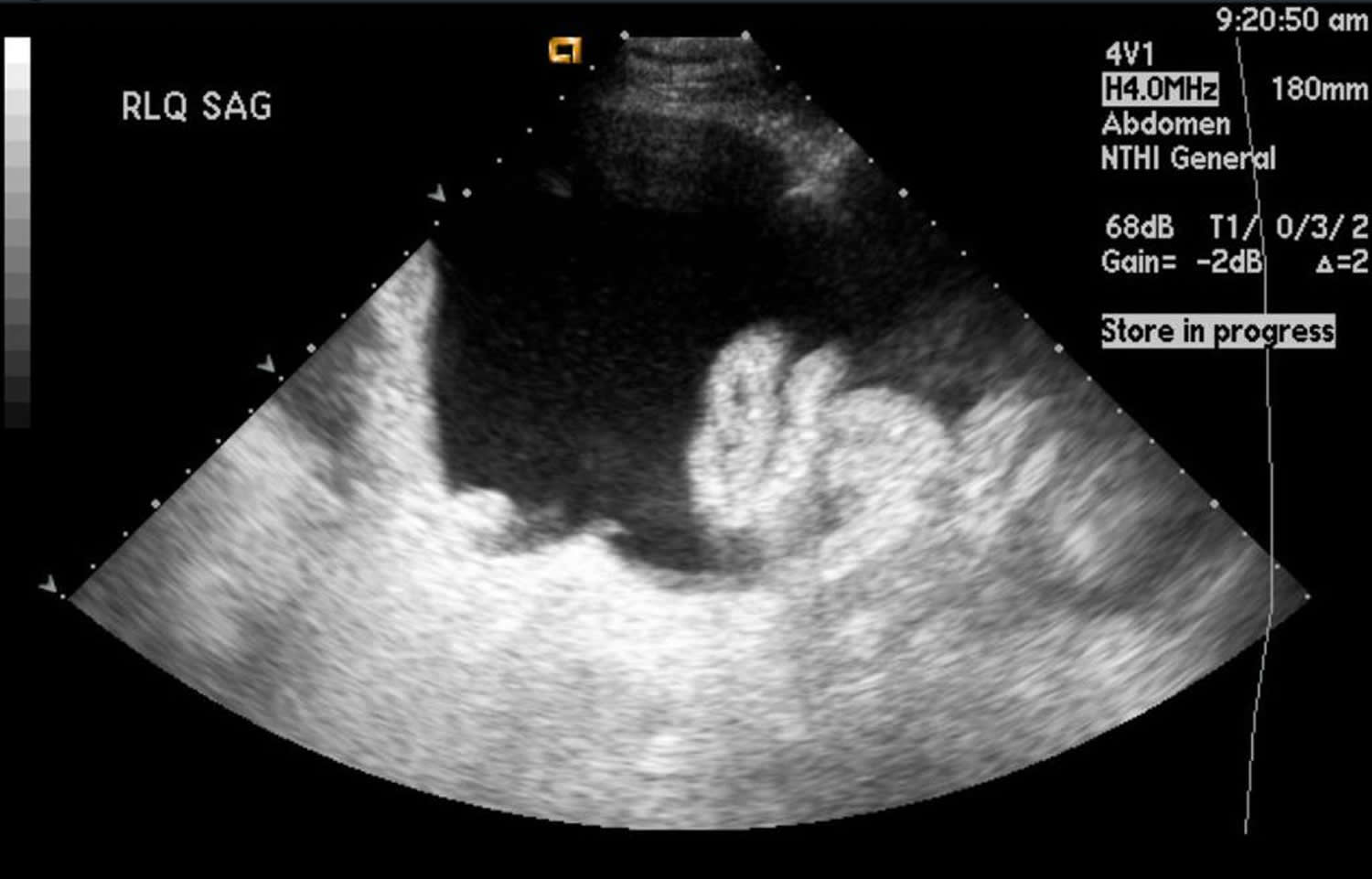
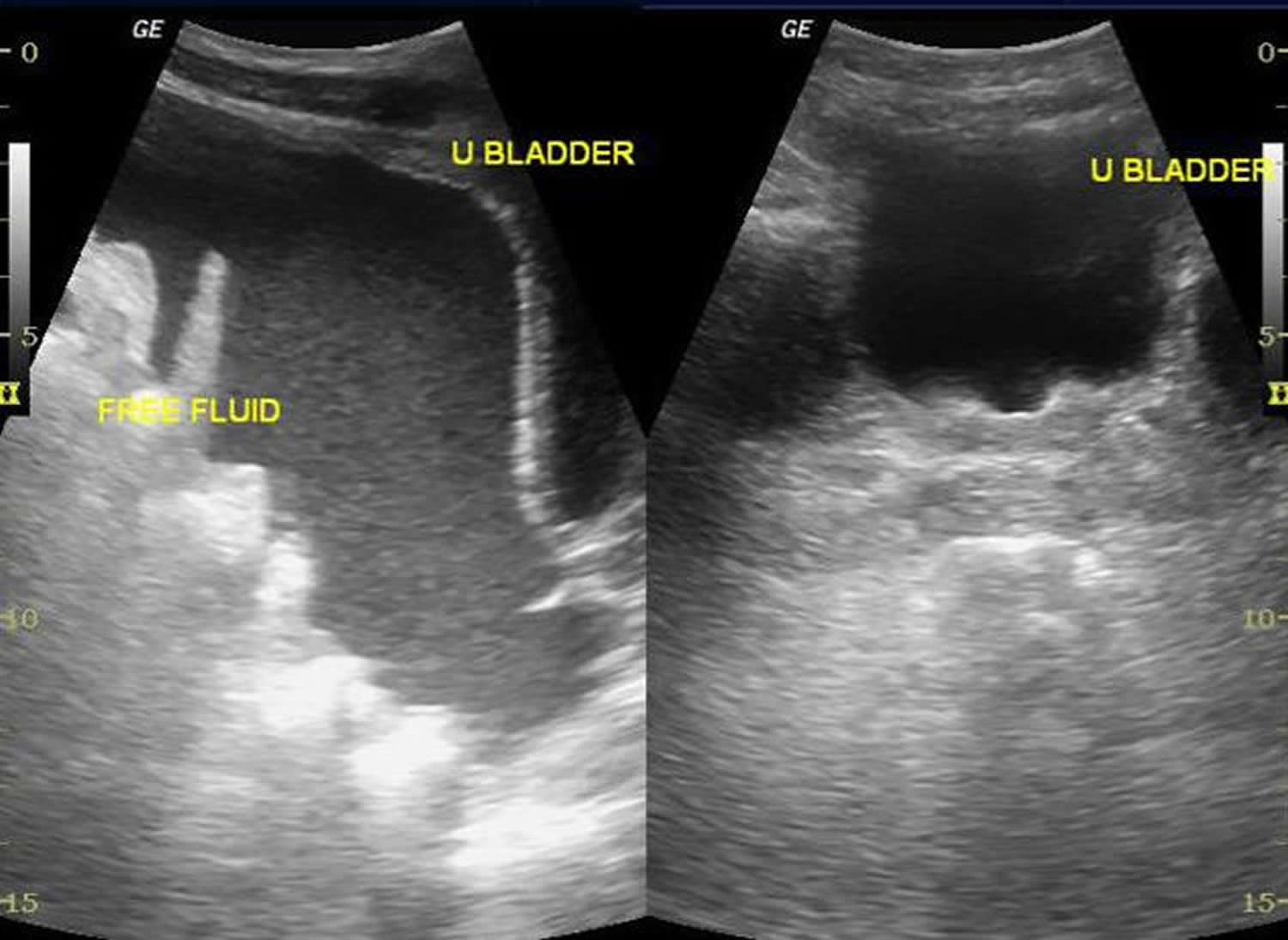
Figure 3. Ultrasonogram showing ascites
Figure 4. Ultrasonogram showing ascites – Gross amount of free fluid is noted in hepatorenal, splenorenal, perihepatic, perisplenic, both paracolic gutters, both iliac fossa and pelvic. Tiny echoes are present in ascites (compare with urinary bladder without echoes).
Figure 5. Abdominal paracentesis
Figure 6. Therapeutic Paracentesis
Paracentesis fluid analysis
Send ascitic fluid for laboratory testing including cell count with differential, Gram stain, and fluid culture. Placing some fluid into bacterial culture bottles can help to increase culture sensitivity. Spontaneous bacterial peritonitis is diagnosed when the absolute neutrophil count is 250 cells/mm³ or more. This is calculated by multiplying the number of white cells by the percentage of neutrophils reported in the differential. Empiric antibiotics, typically a third generation cephalosporin or a fluoroquinolone, should be started in patients with ascites and a high suspicion for spontaneous bacterial peritonitis regardless of the absolute neutrophil count or in patients with an absolute neutrophil count above the cut-off range. Additional tests that can aid in inpatient management include lactate dehydrogenase (LDH), albumin, glucose, protein, cytology and tumor markers. Albumin, in particular, can be used to calculate the serum-ascites albumin gradient (SAAG) which can assist in determining the etiology of ascites by classifying it as either exudative or transudative.
Figure 6. Paracentesis fluid
With regard to differentiation of transudate from exudate, the preferred means for characterizing ascites is the serum-ascitic albumin gradient 11.
The serum-ascitic albumin gradient is calculated by subtracting the albumin concentration of the ascitic fluid from the albumin concentration of a serum specimen obtained on the same day. The serum-ascitic albumin gradient correlates directly with portal pressure. Transudative ascites occurs when a patient’s serum-ascitic albumin gradient level is greater than or equal to 1.1 g/dL (portal hypertension). Exudative ascites occurs when patients have serum-ascitic albumin gradient levels lower than 1.1 g/dL.
An alternative way of differentiating ascites due to portal hypertension from that due to other causes is to measure ascitic fluid viscosity with a cutoff of 1.65 12. Ascitic fluid viscosity has also been demonstrated to predict renal impairment in hepatic patients at a cutoff of 1.35 and a long intensive care unit (ICU) stay at a cutoff of 1.995.
A newer noninvasive method of differentiating exudative from transudative ascites by using B-mode gray-scale ultrasound histogram analysis has been described and appears to be effective 13. In this method, the ascites-to-rectus abdominis muscle echogenicity ratio is measured. A value higher than 0.002 is regarded as exudative ascites, whereas a value lower than 0.002 is regarded as transudative ascites.
Infection of ascitic fluid without intra-abdominal infection usually occurs in patients with chronic liver disease due to translocation of enteric bacteria. Common pathogens include Escherichia coli, Klebsiella pneumoniae, enterococcal species, and Streptococcus pneumoniae 14. Patients with renal failure who use abdominal peritoneal dialysis are also at increased risk, as are children with nephrosis or systemic lupus erythematosus. Anaerobic bacteria are not associated with spontaneous bacterial peritonitis.
An ascitic fluid polymorphonuclear leukocyte count higher than 250 cells/mm³ (neutrocytic ascites), with the percentage of polymorphonuclear leukocytes in the fluid usually greater than 50%, is presumptive evidence of spontaneous bacterial peritonitis. Patients whose ascitic fluid meets these criteria should be treated empirically, regardless of symptoms. Secondary bacterial peritonitis is defined as infected ascitic fluid associated with an intra-abdominal infection.
A report by Lutz et al 15 demonstrated that the relative polymorphonuclear leukocyte count, as compared with the absolute polymorphonuclear leukocytes count, is a less expensive marker associated with bacterascites and can be used to predict future episodes of spontaneous bacterial peritonitis. This may be useful for the purposes of risk stratification.
Best practices
Depending on the clinical situation, ascitic fluid may be sent for the following laboratory tests:
- Gram stain – In a retrospective review of 796 peritoneal fluid samples, the evaluation of Gram stain results rarely provided clinically useful information for the detection of spontaneous bacterial peritonitis 16
- Cell count (elevated counts may suggest infection)
- Bacterial culture
- Total protein level
- Triglyceride levels (elevated in chylous ascites)
- Bilirubin level (may be elevated in bowel perforation)
- Glucose level
- Albumin level, used in conjunction with serum albumin levels obtained the same day used to calculate serum-ascites albumin gradient (SAAG)
- Amylase level (elevation suggests pancreatic source)
- Lactate dehydrogenase (LDH) level
- Cytology
In patients who are afebrile, alert, and have no other signs of bacterial peritonitis, ascitic fluid labs are often not necessary to rule out spontaneous bacterial peritonitis) 17.
To minimize the risk of persistent leak from the puncture site, use a small-gauge needle or take a “Z” track during insertion of the needle. (During removal of the needle, the subcutaneous tissue seals on itself.)
In a retrospective review of 796 peritoneal fluid samples, the evaluation of Gram stain results rarely provided clinically useful information for the detection of spontaneous bacterial peritonitis 16.
What abnormal results mean
An exam of abdominal fluid (ascitic fluid) may show:
- Cancer that has spread to the abdominal cavity (most often cancer of the ovaries)
- Cirrhosis of the liver
- Damaged bowel
- Heart disease
- Infection
- Kidney disease
- Pancreatic disease
Paracentesis complications
Paracentesis is a safe procedure, however there is a slight chance that the needle could puncture the bowel, bladder, or a blood vessel in the abdomen. If a large quantity of fluid is removed, there is a slight risk of lowered blood pressure and kidney failure. There is also a slight chance of infection.
A prospective study of 171 patients undergoing paracentesis found that “major” complications occurred in 1.6% of procedures and included five episodes of bleeding and three infections, resulting in death in two cases. Major complications were associated with therapeutic but not diagnostic procedures and tended to be more prevalent in patients with low platelet counts, patients who were Child-Pugh stage C, and patients with alcoholic cirrhosis 8.
Possible complications of paracentesis procedure include:
- Persistent leakage of ascitic fluid at the needle insertion site. This can often be addressed with a single skin suture.
- Abdominal wall hematoma
- Wound infection
- Perforation of surrounding vessels or viscera (extremely rare)
- Hypotension after large volume fluid removal (more than 5 liters to 6 liters). Albumin is often administered after removal of more than 5 L of fluid to prevent this complication.
- Failed attempt to collect peritoneal fluid
- Spontaneous hemoperitoneum – This rare complication is due to mesenteric variceal bleeding after removal of a large amount of ascitic fluid (>4 L).
- Hollow viscus perforation (small or large bowel, stomach, bladder)
- Catheter laceration and loss in abdominal cavity
- Laceration of major blood vessel (aorta, mesenteric artery, iliac artery)
- Postparacentesis hypotension
- Dilutional hyponatremia
- Hepatorenal syndrome
Complication prevention
In cases with a persistent leak, a single skin suture might solve the problem. The application of an ostomy bag around the puncture site keeps the leak contained until it is eventually sealed off.
Postparacentesis hypotension is a delayed complication that may occur more than 12 hours after a procedure in which large volumes are taken off. Patients can be pretreated with a colloid solution, such as albumin, to decrease the frequency of this complication, though no difference in survival has been noted relative to other plasma expanders 18.
Timing of paracentesis
At this time and in the absence of prospective, randomized, controlled data, the authors 19, 20 recommend early diagnostic or therapeutic paracentesis and early empiric antibiotic administration in patient suspected of having spontaneous bacterial peritonitis. When it appears that large-volume paracentesis is likely to be required, the authors suggest consideration of early small-volume paracentesis (ultrasound, syringe, and needle technique) followed by delayed and planned large-volume paracentesis (during hospitalization) under appropriate monitoring and hemodynamic support to minimize the risk of circulatory dysfunction induced by large-volume paracentesis.
- Wong CL, Holroyd-Leduc J, Thorpe KE, Straus SE. Does this patient have bacterial peritonitis or portal hypertension? How do I perform a paracentesis and analyze the results?. JAMA. 2008 Mar 12. 299(10):1166-78.[↩]
- Aponte EM, O’Rourke MC. Paracentesis. [Updated 2017 May 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK435998/[↩]
- Huang Z, Yu SH, Liang HY, Zhou J, Yan HT, Chen T, et al. Outcome benefit of abdominal paracentesis drainage for severe acute pancreatitis patients with serum triglyceride elevation by decreasing serum lipid metabolites. Lipids Health Dis. 2016 Jun 24. 15:110.[↩]
- Bureau C, Adebayo D, de Rieu MC, Elkrief L, Valla D, Peck-Radosavljevic M, et al. alfapump System vs. Large Volume Paracentesis for Refractory Ascites: A Multicenter Randomized Controlled Study. J Hepatol. 2017 Jun 20.[↩]
- Mercadante S, Intravaia G, Ferrera P, Villari P, David F. Peritoneal catheter for continuous drainage of ascites in advanced cancer patients. Support Care Cancer. 2008 Aug. 16(8):975-8.[↩]
- Kwok CS, Krupa L, Mahtani A, et al. Albumin Reduces Paracentesis-Induced Circulatory Dysfunction and Reduces Death and Renal Impairment among Patients with Cirrhosis and Infection: A Systematic Review and Meta-Analysis. Biomed Res Int. 2013. 2013:295153.[↩]
- McVay PA, Toy PT. Lack of increased bleeding after paracentesis and thoracentesis in patients with mild coagulation abnormalities. Transfusion. 1991 Feb. 31(2):164-71.[↩][↩]
- De Gottardi A, Thévenot T, Spahr L, Morard I, Bresson-Hadni S, Torres F, et al. Risk of complications after abdominal paracentesis in cirrhotic patients: a prospective study. Clin Gastroenterol Hepatol. 2009 Aug. 7(8):906-9.[↩][↩]
- Nazeer SR, Dewbre H, Miller AH. Ultrasound-assisted paracentesis performed by emergency physicians vs the traditional technique: a prospective, randomized study. Am J Emerg Med. 2005 May. 23(3):363-7.[↩]
- Kelil T, Shyn PB, Wu LE, Levesque VM, Kacher D, Khorasani R, et al. Wall suction-assisted image-guided therapeutic paracentesis: a safe and less expensive alternative to evacuated bottles. Abdom Radiol (NY). 2016 Jul. 41 (7):1333-7.[↩]
- McGibbon A, Chen GI, Peltekian KM, van Zanten SV. An evidence-based manual for abdominal paracentesis. Dig Dis Sci. 2007 Dec. 52(12):3307-15.[↩]
- Hanafy AS. The role of ascitic fluid viscosity in differentiating the nature of ascites and in the prediction of renal impairment and duration of ICU stay. Eur J Gastroenterol Hepatol. 2016 May 23.[↩]
- Çekiç B, Toslak IE, Şahintürk Y, Cekin AH, Koksel YK, Koroglu M, et al. Differentiating Transudative From Exudative Ascites Using Quantitative B-Mode Gray-Scale Ultrasound Histogram. AJR Am J Roentgenol. 2017 Jun 1. 1-7.[↩]
- Kuiper JJ, van Buuren HR, de Man RA. Ascites in cirrhosis: a review of management and complications. Neth J Med. 2007 Sep. 65(8):283-8.[↩]
- Lutz P, Goeser F, Kaczmarek DJ, Schlabe S, Nischalke HD, Nattermann J, et al. Relative Ascites Polymorphonuclear Cell Count Indicates Bacterascites and Risk of Spontaneous Bacterial Peritonitis. Dig Dis Sci. 2017 Jun 9[↩]
- Chinnock B, Fox C, Hendey GW. Gram’s Stain of Peritoneal Fluid Is Rarely Helpful in the Evaluation of the Ascites Patient. Ann Emerg Med. 2009 Feb 5.[↩][↩]
- Cadranel JF, Nousbaum JB, Bessaguet C, et al. Low incidence of spontaneous bacterial peritonitis in asymptomatic cirrhotic outpatients. World J Hepatol. 2013 Mar 27. 5(3):104-8.[↩]
- Gines A, Fernandez-Esparrach G, Monescillo A, Vila C, Domenech E, Abecasis R. Randomized trial comparing albumin, dextran 70, and polygeline in cirrhotic patients with ascites treated by paracentesis. Gastroenterology. 1996 Oct. 111(4):1002-10.[↩]
- Kim JJ, Tsukamoto MM, Mathur AK, Ghomri YM, Hou LA, Sheibani S, et al. Delayed paracentesis is associated with increased in-hospital mortality in patients with spontaneous bacterial peritonitis. Am J Gastroenterol. 2014 Sep. 109(9):1436-42.[↩]
- Orman ES, Hayashi PH, Bataller R, Barritt AS 4th. Paracentesis is associated with reduced mortality in patients hospitalized with cirrhosis and ascites. Clin Gastroenterol Hepatol. 2014 Mar. 12(3):496-503.e1.[↩]