Contents
What is acute radiation syndrome
Acute Radiation Syndrome sometimes known as radiation toxicity or radiation sickness, is an acute illness caused by irradiation >1 Gy (more than 1 Gray or 100 rads) total dose, delivered acutely at a relatively high-dose rate 1 to the entire body (or most of the body ~ 60%) by a high dose of penetrating radiation in a very short period of time (usually a matter of minutes). The major cause of this syndrome is depletion of immature parenchymal stem cells in specific tissues. Examples of people who suffered from Acute Radiation Syndrome are the survivors of the Hiroshima and Nagasaki atomic bombs, the firefighters that first responded after the Chernobyl Nuclear Power Plant event in 1986, and some unintentional exposures to sterilization irradiators. The amount of radiation that a person’s body absorbs is called the radiation dose.
The absorbed dose of radiation is measured in a unit called a gray (Gy). Diagnostic tests that use radiation, such as an X-ray, result in a small dose of radiation — typically well below 0.1 Gy — focused on a few organs or small amount of tissue.
One gray (1 Gy) is the absorption of one joule of energy, in the form of ionizing radiation, per kilogram of matter. To put that into perspective, a 1 Gy exposure is equivalent to having 200,000 dental X-rays or 4.347 million hours of average background radiation (496 years worth of background radiation).
Signs and symptoms of radiation sickness usually appear when the entire body receives an absorbed dose of at least 1 Gy. Doses greater than 10 Gy to the whole body are generally not treatable and usually lead to death within two days to two weeks, depending on the dose and duration of the exposure.
People are constantly exposed to low levels of naturally occurring radiation called background radiation. Background radiation comes from cosmic radiation and from radioactive elements in the air, water, and ground. Cosmic radiation is concentrated at the poles by the earth’s magnetic field and is attenuated by the atmosphere. Thus, exposure is greater for people living at high latitudes, at high altitudes, or both and during airplane flights. Terrestrial sources of external radiation exposure are primarily due to the presence of radioactive elements with half-lives comparable to the age of the earth (~4.5 billion years). In particular, uranium (238U) and thorium (232Th) along with several dozen of their radioactive progeny and a radioactive isotope of potassium (40K) are present in many rocks and minerals. Small quantities of these radionuclides are in the food, water, and air and thus contribute to internal exposure as these radionuclides are invariably incorporated into the body. The majority of the dose from internally incorporated radionuclides is from radioisotopes of carbon (14C) and potassium (40K), and because these and other elements (stable and radioactive forms) are constantly replenished in the body by ingestion and inhalation, there are approximately 7000 atoms undergoing radioactive decay each second.
Internal exposure from the inhalation of radioactive isotopes of the noble gas radon (222Rn and 220Rn), which are also formed from the Uranium (238U) decay series, accounts for the largest portion (73%) of the US population’s average per capita naturally occurring radiation dose. Cosmic radiation accounts for 11%, radioactive elements in the body for 9%, and external terrestrial radiation for 7%. In the US, people receive an average effective dose of about 3 millisieverts (mSv)/yr from natural sources (range ~0.5 to 20 mSv/yr). However, in some parts of the world, people receive > 50 mSv/yr. The doses from natural background radiation are far too low to cause radiation injuries; they may result in a small increase in the risk of cancer, although some experts think there may be no increased risk.
Types of radiation
Radiation includes:
- High-energy electromagnetic waves (x-rays, gamma rays)
- Particles (alpha particles, beta particles, neutrons)
Alpha particles are energetic helium nuclei emitted by some radionuclides with high atomic numbers (eg, plutonium, radium, uranium); they cannot penetrate skin beyond a shallow depth (< 0.1 mm).
Beta particles are high-energy electrons that are emitted from the nuclei of unstable atoms (eg, cesium-137, iodine-131). These particles can penetrate more deeply into skin (1 to 2 cm) and cause both epithelial and subepithelial damage.
Neutrons are electrically neutral particles emitted by a few radionuclides (eg, californium-252) and produced in nuclear fission reactions (eg, in nuclear reactors); their depth of tissue penetration varies from a few millimeters to several tens of centimeters, depending on their energy. They collide with the nuclei of stable atoms, resulting in emission of energetic protons, alpha and beta particles, and gamma radiation.
Gamma radiation and x-rays are electromagnetic radiation (ie, photons) of very short wavelength that can penetrate deeply into tissue (many centimeters). While some photons deposit all their energy in the body, other photons of the same energy may only deposit a fraction of their energy and others may pass completely through the body without interacting.
Because of these characteristics, alpha and beta particles cause the most damage when the radioactive atoms that emit them are within the body (internal contamination) or, in the case of beta-emitters, directly on the body; only tissue in close proximity to the radionuclide is affected. Gamma rays and x-rays can cause damage distant from their source and are typically responsible for acute radiation syndromes. Acute Radiation Syndrome can be caused by a sufficient dose of some internally deposited radionuclides that are widely distributed in tissues and organs and have a high specific activity. For example, polonium-210 (Po-210) has a specific activity of 166 terabecquerels per gm (TBq/g) and 1 mcg (size of a grain of salt) of Po-210 delivers a whole body dose of 50 Sv (~20 times the median lethal dose).
Measurement of radiation
Conventional units of measurement include the roentgen, rad, and rem. The roentgen (R) is a unit of exposure measuring the ionizing ability of x-rays or gamma radiation in air. The radiation absorbed dose (rad) is the amount of that radiation energy absorbed per unit of mass. Because biologic damage per rad varies with radiation type (eg, it is higher for neutrons than for x-rays or gamma radiation), the dose in rad is corrected by a quality factor; the resulting equivalent dose unit is the roentgen equivalent in man (rem). Outside the US and in the scientific literature, SI (International System) units are used, in which the rad is replaced by the gray (Gy) and the rem by the sievert (Sv); 1 Gy = 100 rad and 1 Sv = 100 rem. The rad and rem (and hence Gy and Sv) are essentially equal (ie, the quality factor equals 1) when describing x-rays or gamma or beta radiation.
The amount (quantity) of radioactivity is expressed in terms of the number of nuclear disintegrations (transformations) per second. The becquerel (Bq) is the SI unit of radioactivity; one Bq is 1 disintegration per second (dps). In the US system, one curie is 37 billion Bq.
Sources of high-dose radiation
Possible sources of high-dose radiation include the following:
- An accident at a nuclear industrial facility
- An attack on a nuclear industrial facility
- Detonation of a small radioactive device
- Detonation of a conventional explosive device that disperses radioactive material (dirty bomb)
- Detonation of a standard nuclear weapon
Radiation sickness occurs when high-energy radiation damages or destroys certain cells in your body. Regions of the body most vulnerable to high-energy radiation are cells in the lining of your intestinal tract, including your stomach, and the blood cell-producing cells of bone marrow.
People exposed to radiation will get Acute Radiation Syndrome only if:
- The radiation dose must be large (i.e., greater than 0.7 Gray (Gy) or 70 rads) 2.
- Mild symptoms may be observed with doses as low as 0.3 Gy or 30 rads.
- The dose usually must be external (i.e., the source of radiation is outside of the patient’s body).
- Radioactive materials deposited inside the body have produced some acute radiation syndrome effects only in extremely rare cases.
- The radiation must be penetrating (i.e., able to reach the internal organs).
- High energy X-rays, gamma rays, and neutrons are penetrating radiations.
- The entire body (or a significant portion of it) must have received the dose 3.
- Most radiation injuries are local, frequently involving the hands, and these local injuries seldom cause classical signs of acute radiation syndrome.
- The dose must have been delivered in a short time (usually a matter of minutes).
- Fractionated doses are often used in radiation therapy. These large total doses are delivered in small daily amounts over a period of time. Fractionated doses are less effective at inducing acute radiation syndrome than a single dose of the same magnitude.
Such irradiation injury initially affects all organs to some extent, but the timing and extent of the injury manifestations depend upon the type, rate, and dose of radiation received 4. The percentage of the body that is injured, the dose homogeneity, and the intrinsic radiosensitivity of the exposed individual also influence manifestations. Different ranges of whole-body doses produce different manifestations of injury. The three main ranges that produce the most characteristic manifestations are referred to as the hematological, gastrointestinal, and neurovascular syndromes. These syndromes are, as a rule, produced only with whole-body or near whole-body irradiation by photon or mixed photon/neutron radiation. High-dose injuries to smaller percentages of the body produce local injury effects, but may not cause Acute Radiation Syndrome 5.
The three classic Acute Radiation Syndromes
- Bone marrow syndrome (sometimes referred to as hematopoietic syndrome): the full syndrome will usually occur with a dose greater than approximately 0.7 Gy (70 rads) although mild symptoms may occur as low as 0.3 Gy or 30 rads 6. The survival rate of patients with this syndrome decreases with increasing dose. The primary cause of death is the destruction of the bone marrow, resulting in infection and hemorrhage.
- Gastrointestinal (GI) syndrome: the full syndrome will usually occur with a dose greater than approximately 10 Gy (1000 rads) although some symptoms may occur as low as 6 Gy or 600 rads. Survival is extremely unlikely with this syndrome. Destructive and irreparable changes in the GI tract and bone marrow usually cause infection, dehydration, and electrolyte imbalance. Death usually occurs within 2 weeks.
- Cardiovascular (CV)/ Central Nervous System (CNS) syndrome: the full syndrome will usually occur with a dose greater than approximately 50 Gy (5000 rads) although some symptoms may occur as low as 20 Gy or 2000 rads. Death occurs within 3 days. Death likely is due to collapse of the circulatory system as well as increased pressure in the confining cranial vault as the result of increased fluid content caused by edema, vasculitis, and meningitis.
The four stages of Acute Radiation Syndrome
- Prodromal stage (N-V-D stage): The classic symptoms for this stage are nausea, vomiting, as well as anorexia and possibly diarrhea (depending on dose), which occur from minutes to days following exposure. The symptoms may last (episodically) for minutes up to several days.
- Latent stage: In this stage, the patient looks and feels generally healthy for a few hours or even up to a few weeks.
- Manifest illness stage: In this stage, the symptoms depend on the specific syndrome (see Table 1) and last from hours up to several months.
- Recovery or death: Most patients who do not recover will die within several months of exposure. The recovery process lasts from several weeks up to two years.
These stages are described in more detail in Table 1.
Table 1. Acute radiation syndromes stages
| Syndrome | Signs and symptoms | |||
|---|---|---|---|---|
| Prodromal phase first 48 hoursb | Latent phase lasts up to a month | Manifest illness phase | Final outcome: survival or death | |
| Gastrointestinal Occurs at doses between 5 Gy and 12 Gy | Nausea, vomiting, diarrhea, anorexia, hemorrhage, weakness through denuded areas Loss of absorptive capacity Increased intensity 4–8 hours | Tiredness and anorexia | Vomiting and fever; progression of bloody diarrhea to shock and death or treatment | Radiation 8–30 Gy dose range cause death from gastrointestinal syndrome |
| Hematologic High dose between 2–3 Gy and 8 Gy. Low dose (<2 Gy) radiation | Often asymptomatic Some fatigue, fever, and bacteremia | Lymphopenia Granulocytopenia Thrombocytopenia | Neutropenia (ANC < 0.5)c Fever, sepsis, hemorrhage, purpura, electrolyte disturbances, and epilation | Agranulocytosis irresponsive to GM-CSF after first cell cycle |
| Central nervous system | No specific signs and symptoms Unspecific fatigue, malaise, anorexia, and drowsiness Not consistently correlated to exposed dose | Latency up to a month Asymptomatic phase except for tiredness and weakness | Headache Impaired cognition, disorientation, seizure, tremor, ataxia Grand mal seizures | Irreversible brain damage secondary to continuous cramps |
| Pulmonary dysfunction | Acute radiation pneumonitis Cough, shortness of breath ALId with inflammatory coagulation activation | Pulmonary edema Pneumonitis | ARDSe Intubation and mechanical ventilation Severe pneumonia Lung fibrosis after 14–30 days from first exposure | Absolute respiratory insufficiency Severely reduced oxygen transport capacity |
Abbreviations: ALI, acute lung insufficiency; ANC, absolute neutrophil count; ARDS, acute respiratory distress syndrome; FiO2, fraction of inspired oxygen in a gas mixture; GM-CSF, granulocyte-macrophage colony-stimulating factor; PaO2, partial pressure of oxygen in the blood.
The most sensitive cells to acute radiation effect are in bone marrow. However, an overlooked fact is that there are other important replicative cells, namely the fixed tissue macrophages in tissue and vital organs. Depending on the absorbed radioactive dose, symptoms appear within hours to weeks, following a predictable clinical course. The prodromal phase of Acute Radiation Syndrome usually occurs in the first 48 hours, but may develop up to 6 days after exposure 7. The latent phase is a short period characterized by improvement of symptoms as the person appears to have recovered. Unfortunately, this effect is transient, lasting for several days to a month.
Symptoms of manifest illness then appear and may last for weeks. This stage is characterized by intense immunosuppression and is the most difficult to manage. If a person survives this stage, recovery is likely. Individuals exposed to a supralethal dose of radiation deteriorate over a period of hours, resulting in early death 5.
Symptoms of acute, high-dose radiation are dependent on the absorbed dose. They may appear within hours to days and follow a somewhat predictable course 8. Early symptoms resulting from an acute whole-body exposure constitute the prodromal radiation syndrome. Virtually all individuals receiving a dose of 10–20 Gy develop prodromal signs and symptoms within 1–72 hours after exposure 7. The initial clinical picture is most often dominated by gastrointestinal signs and symptoms (Table 1) primarily resulting from central nervous system manifestations due to the location of the control center of anorexia, nausea, and vomiting in the brain. Central nervous system dysfunction may be evident early on by changes in the electroencephalography, even at much lower doses. In later phases, the symptoms gradually merge into loss of consciousness, hypotension, and death (components of the cerebrovascular syndrome that is characterized by neurologic failure and cardiovascular collapse) before toxicity to other organ systems (such as the gastrointestinal and hematologic systems) can develop.
Death occurs within a few days after exposure to 10–20 Gy, in absence of treatment 4. A rapid, severe prodromal response is the harbinger of a poor clinical outcome that is complicated by severe leukoneutropenia, thrombocytopenia, and anemia with reticulocytopenia, accompanied by hemorrhage, infection, and death. At lower doses (2–10 Gy), it is difficult to establish a prognosis based on the prodromal phase. The prodromal phase is followed by a phase of manifest illness where syndromes specific to various organ systems emerge. Four major organ subsystems are known to be of critical significance in the development of Acute Radiation Syndrome: the gastrointestinal system, neurovascular system, hematologic system, and pulmonary system. Evaluation of system-specific signs and symptoms is required for triage of victims, selection of therapy, and determination of prognosis 9.
Cutaneous Radiation Syndrome
The concept of cutaneous radiation syndrome was introduced in recent years to describe the complex pathological syndrome that results from acute radiation exposure to the skin.
Acute Radiation Syndrome usually will be accompanied by some skin damage. It is also possible to receive a damaging dose to the skin without symptoms of acute radiation syndrome, especially with acute exposures to beta radiation or X-rays. Sometimes this occurs when radioactive materials contaminate a patient’s skin or clothes.
When the basal cell layer of the skin is damaged by radiation, inflammation, erythema, and dry or moist desquamation can occur. Also, hair follicles may be damaged, causing epilation. Within a few hours after irradiation, a transient and inconsistent erythema (associated with itching) can occur. Then, a latent phase may occur and last from a few days up to several weeks, when intense reddening, blistering, and ulceration of the irradiated site are visible.
In most cases, healing occurs by regenerative means; however, very large skin doses can cause permanent hair loss, damaged sebaceous and sweat glands, atrophy, fibrosis, decreased or increased skin pigmentation, and ulceration or necrosis of the exposed tissue.
Radiation poisoning symptoms
The initial signs and symptoms of treatable radiation sickness are usually nausea and vomiting. The amount of time between exposure and when these symptoms develop is an indicator of how much radiation a person has absorbed.
After the first round of signs and symptoms, a person with radiation sickness may have a brief period with no apparent illness, followed by the onset of new, more-serious symptoms.
In general, the greater your radiation exposure, the more rapid and more severe your symptoms will be.
Symptoms of acute radiation syndrome may include nausea, vomiting, headache, and diarrhea.
- These symptoms start within minutes to days after the exposure, can last for minutes up to several days, and may come and go.
- If you have these symptoms after a radiation emergency, seek medical attention as soon as emergency officials determine it is safe to do so.
After the initial symptoms, a person usually looks and feels healthy for a period of time, after which he or she will become sick again with variable symptoms and severity that vary depending on the radiation dose that he or she received.
- These symptoms include loss of appetite, fatigue, fever, nausea, vomiting, diarrhea, and possibly even seizures and coma.
- This seriously ill stage may last from a few hours up to several months.
- People who receive a high radiation dose also can have skin damage. This damage can start to show within a few hours after exposure or it may be delayed for several days. It can include swelling, itching, and redness of the skin (like a bad sunburn) or may be more severe and include blisters or ulcers.
- The skin may heal for a short time, followed by the return of swelling, itching, and redness days or weeks later.
- Complete healing of the skin may take from several weeks up to a few years.
- The time for skin to heal depends on the radiation dose the person’s skin received.
- People who receive a high radiation dose to all or part of the body also may experience temporary hair loss. It may take several weeks for the hair to grow back.
People with acute radiation syndrome typically also have some skin damage. This damage can start to show within a few hours after exposure and can include swelling, itching, and redness of the skin (like a bad sunburn).
There also can be hair loss. As with the other symptoms, the skin may heal for a short time, followed by the return of swelling, itching, and redness days or weeks later. Complete healing of the skin may take from several weeks up to a few years radiation syndrome depending on the radiation dose the person’s skin received.
| Mild exposure (1-2 Gy) | Moderate exposure (2-6 Gy) | Severe exposure (6-9 Gy) | Very severe exposure (10 Gy or higher) | |
|---|---|---|---|---|
| Based on Radiation exposure and contamination. Merck Manual Professional Edition 10. | ||||
| Nausea and vomiting | Within 6 hours | Within 2 hours | Within 1 hour | Within 10 minutes |
| Diarrhea | — | Within 8 hours | Within 3 hours | Within 1 hour |
| Headache | — | Within 24 hours | Within 4 hours | Within 2 hours |
| Fever | — | Within 3 hours | Within 1 hour | Within 1 hour |
| Dizziness and disorientation | — | — | Within 1 week | Immediate |
| Weakness, fatigue | Within 4 weeks | Within 1-4 weeks | Within 1 week | Immediate |
| Hair loss, bloody vomit and stools, infections, poor wound healing, low blood pressure | — | Within 1-4 weeks | Within 1 week | Immediate |
Radiation poisoning prevention
In the event of a radiation emergency, stay tuned to your radio or television to hear what protective actions local, state and federal authorities recommend. Recommended actions will depend on the situation, but you will be told to either stay in place or evacuate your area.
Shelter in place
If you’re advised to stay where you are, whether you’re at home or work or elsewhere, do the following:
- Close and lock all doors and windows.
- Turn off fans, air conditioners and heating units that bring air in from outside.
- Close fireplace dampers.
- Bring pets indoors.
- Move to an inner room or basement.
- Stay tuned to your emergency response network or local news.
Evacuate
If you’re advised to evacuate, follow the instructions provided by your local authorities. Try to stay calm and move quickly and in an orderly manner. In addition, travel lightly, but take supplies, including:
- Flashlight
- Portable radio
- Batteries
- First-aid kit
- Necessary medicines
- Sealed food, such as canned foods, and bottled water
- Manual can opener
- Cash and credit cards
- Extra clothes
Be aware that most emergency vehicles and shelters won’t accept pets. Take them only if you’re driving your own vehicle and going someplace other than a shelter.
Acute Radiation Syndrome Diagnosis
The diagnosis of Acute Radiation Syndrome can be difficult to make because acute radiation syndrome causes no unique disease. Also, depending on the dose, the prodromal stage may not occur for hours or days after exposure, or the patient may already be in the latent stage by the time they receive treatment, in which case the patient may appear and feel well when first assessed.
If a patient received more than 0.05 Gy (5 rads) and three or four complete blood counts (CBCs) are taken within 8 to 12 hours of the exposure, a quick estimate of the dose can be made. If these initial blood counts are not taken, the dose can still be estimated by using complete blood count (CBC) results over the first few days. It would be best to have radiation dosimetrists conduct the dose assessment, if possible.
If a patient is known to have been or suspected of having been exposed to a large radiation dose, draw blood for complete blood count (CBC) analysis with special attention to the lymphocyte count, every 2 to 3 hours during the first 8 hours after exposure (and every 4 to 6 hours for the next 2 days). Observe the patient during this time for symptoms and consult with radiation experts before ruling out acute radiation syndrome.
If no radiation exposure is initially suspected, you may consider acute radiation syndrome in the differential diagnosis if a history exists of nausea and vomiting that is unexplained by other causes. Other indications are bleeding, epilation, or white blood count (WBC) and platelet counts abnormally low a few days or weeks after unexplained nausea and vomiting. Again, consider complete blood count (CBC) and chromosome analysis and consultation with radiation experts to confirm diagnosis.
Radiation poisoning treatment
Treatment of acute radiation syndrome
- Treatment of acute radiation syndrome focuses on reducing and treating infections, maintaining hydration, and treating injuries and burns. Some patients may benefit from treatments that help the bone marrow recover its function.
- The lower the radiation dose, the more likely it is that the person will recover from acute radiation syndrome.
- The cause of death in most cases is the destruction of the person’s bone marrow, which results in infections and internal bleeding.
- For survivors of acute radiation syndrome, the recovery process may last from several weeks up to 2 years.
- Cutaneous Radiation Injury happens when exposure to a large dose of radiation causes injury to the skin. A doctor will suspect the presence of a Cutaneous Radiation Injury when a skin burn develops in a person who was not exposed to a source of heat, electrical current, or chemicals.
Initial Treatment and Diagnostic Evaluation
Treat vomiting 11 and repeat complete blood count (CBC) analysis with special attention to the lymphocyte count every 2 to 3 hours for the first 8 to 12 hours after exposure (and every 4 to 6 hours for the following 2 or 3 days). Sequential changes in absolute lymphocyte counts over time are demonstrated below in the Andrews Lymphocyte Nomogram (see Figure 1). Precisely record all clinical symptoms, particularly nausea, vomiting, diarrhea, and itching, reddening or blistering of the skin. Be sure to include time of onset.
Figure 1. Andrews Lymphocyte Nomogram
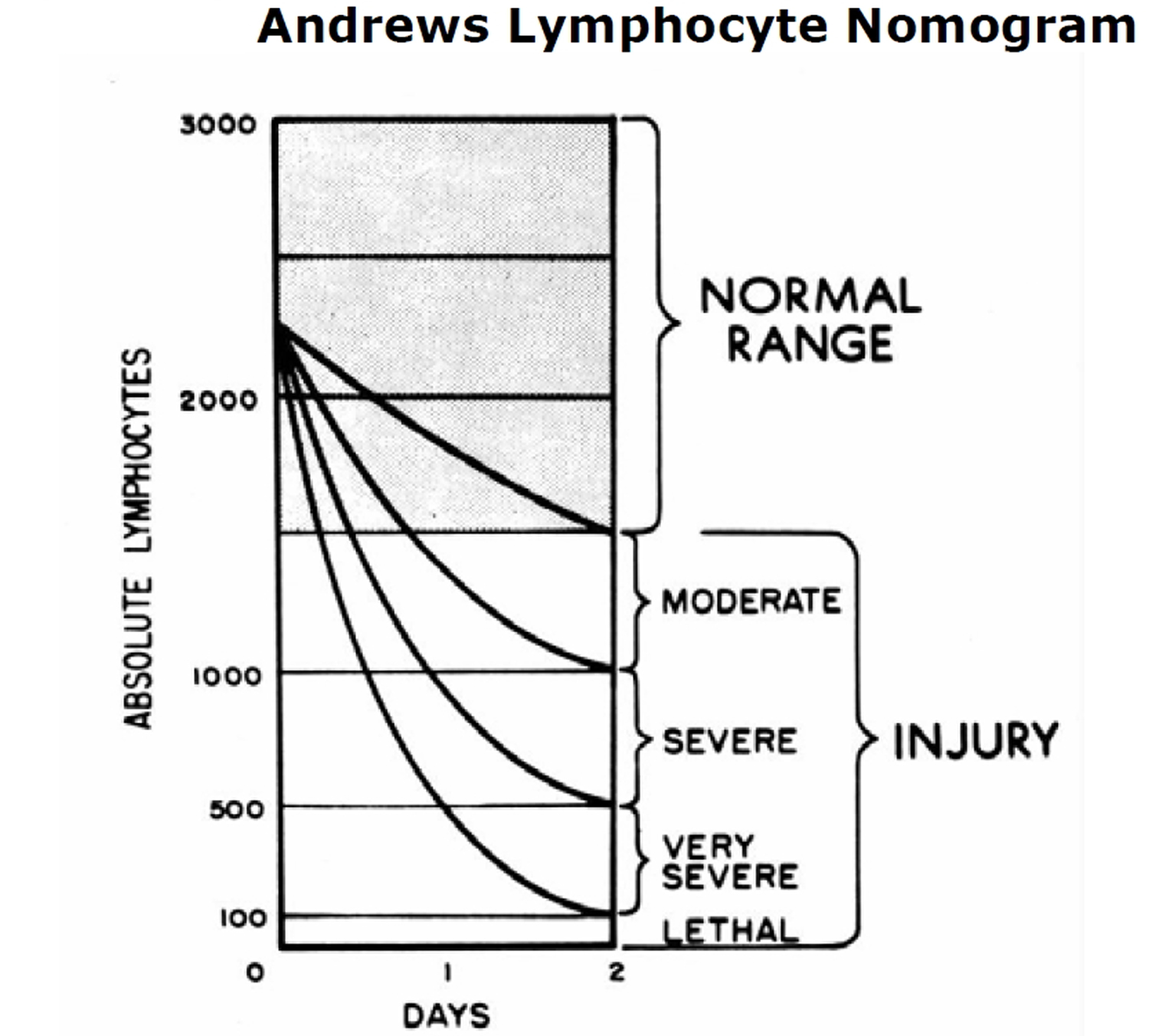 Note and record areas of erythema. If possible, take color photographs of suspected radiation skin damage. Consider tissue, blood typing, and initiating viral prophylaxis.
Note and record areas of erythema. If possible, take color photographs of suspected radiation skin damage. Consider tissue, blood typing, and initiating viral prophylaxis.
After consultation, begin the following treatment (as indicated):
- supportive care in a clean environment (if available, the use of a burn unit may be quite effective)
- prevention and treatment of infections
- stimulation of hematopoiesis by use of growth factors
- stem cell transfusions or platelet transfusions (if platelet count is too low)
- psychological support
- careful observation for erythema (document locations), hair loss, skin injury, mucositis, parotitis, weight loss, or fever
- confirmation of initial dose estimate using chromosome aberration cytogenetic bioassay when possible. Although resource intensive, this is the best method of dose assessment following acute exposures.
- consultation with experts in radiation accident management
Management of the hematologic syndrome, as a component of acute radiation sickness, requires understanding of its manifestations and implementation of clinical biodosimetry to provide appropriate therapeutic support. Hematopoietic growth factors may be of value if administered early as a component of supportive care. Planning for urgent Hematopoietic Stem Cell Transplantation for those with intermediate- to high-dose radiation (4–10 Gy) may be required (Table 2), although the use of Hematopoietic Stem Cell Transplantation is controversial as outcomes after radiation accidents have been poor 12. Establishing contingency plans for triage, assessment, supportive care, and treatment with defined eligibilities, treatment plans, and incorporated data collection to assess results and plan further improvements in care is imperative for the effective management of large scale radiation accidents 9. The hematology/oncology community is most suited to participate in such contingency planning 13.
Table 2. Candidates for bone marrow transplantation
| First step | |
| Prompt growth factor intervention | In the case of aplasia in relation to acute radiation syndrome, emergency HSCT is not necessary. G-CSF/GM-CSF promotes hematological reconstruction and should be evaluated after prompt administration and after 14 days of high dose GM-CSF administration |
| Second step | |
| Final evaluation for candidates for HSCT | HSCT should only be implemented after residual hematopoiesis and only considered if severe aplasia persists after long G-CSF/GM-CSF high-dose treatment |
Abbreviations: G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; HSCT, hematopoietic stem cell transplantation.
Survival requires hematologic recovery
Management of patients with Acute Radiation Syndrome includes early use of hematopoietic cytokines, antimicrobials, and transfusion support. Recommendations based on radiation dose and physiologic response is made for treatment of the hematologic syndrome, and therapy includes treatment with hematopoietic cytokines, blood transfusion, and, in selected cases, Hematopoietic Stem Cell Transplantation 14.
Additional medical management based on the evolution of clinical signs and symptoms includes the use of antimicrobial agents (quinolones, antiviral therapy, and antifungal agents), antiemetic agents, and analgesic agents. Because of the strong psychological impact of possible radiation exposure, psychosocial support is required for those exposed, regardless of the dose.
The prevention and management of infection is the mainstay of therapy. There is a quantitative relationship between the degree of neutropenia and the increased risk of infectious complications 14. Antibiotic prophylaxis should only be considered in afebrile patients at the highest risk for infection. These patients have profound duration of more than 7 days neutropenia (eg, measured in whole blood as absolute neutrophil count <500/mL). Although the degree of neutropenia is the greatest risk factor for developing infection, other factors influence treatment choice and outcome. Such factors include duration of neutropenia, bactericidal functionality of surviving neutrophils, alteration of physical defense barriers, the patient’s endogenous microflora, and organisms endemic to the hospital and community.19 As the duration of neutropenia increases, the risk of secondary infections such as invasive mycoses also increases 16.
It is for these reasons that adjuvant therapy such as the cytokines sargramostim (GM-CSF [Leukine®; Immunex, Seattle, WA]) and filgrastim (G-CSF [Neupogen®; Amgen, Inc, Thousand Oaks, CA]) will prove invaluable in the treatment of the severely irradiated person, although only sparse data is available 14. The treatment recommendations on irradiation-associated aplasia are based on the known effect of CSF’s (colony stimulating factor) beneficial effect on recovery of neutrophils in oncology and hematology patients 17 and in recipients of bone marrow transplantation 18, on their seemingly positive role on hematological recovery in a small number of radiation accident victims 19 and, most importantly, on the improved survival and positive effects on neutrophils in a number of well-conducted studies of animals exposed to radiation 20.
Treatment with growth factors
Cytokines include GM-CSF, macrophage CSF, G-CSF, stem cell factor, and interleukin series (interleukin-1 to interleukin-16). GM-CSF and G-CSF have been available since 1997 for the treatment of radiation myelosuppression. G-CSF, also known as filgrastim, is administered in a dose of 100–200 mcg/m2/day and GM-CSF, also known as sargramostim, is administered in a dose of 200–400 mcg/m2/day. Both are given intravenously or subcutaneously. Both drugs should be initiated promptly upon diagnosis of significant bone marrow damage and continued until recovery of neutrophil counts is sustained above 800/mm3 15.
G-CSF and GM-CSF are currently in widespread clinical use for the treatment of acute neutropenic conditions, and in turn are used in the management of infections following radiochemotherapy of cancer patients. Both of these agents are potent but selective stimulators of granulopoietic arm of the hematologic system, and serve not only to increase blood neutrophil counts but also to enhance the maturation and function of these vital cells. Both agents have high therapeutic ratios, minimal nonperformance side adverse effects, can be administered and monitored with relative ease, and can effectively serve to minimize the risk of infection resulting from a radiation-compromised lymphohematopoietic system.
G-CSF and GM-CSF are potent stimulators of hematopoiesis and effective in reducing duration and degree of neutropenia. An additional benefit of CSFs is their ability to increase functional capacity of neutrophil and thereby contribute to the prevention of infection in an active role as cellular host. They constitute a remarkable advance in the treatment of neutropenia and are, thus, powerful tools for oncologists in the clinical management of cancer patients. The drugs act upon uncommitted stem cell populations within bone marrow to increase mitotic rate, accelerate repopulation, differentiate daughter cells to become committed stem cells, speed the maturation process, and improve the function of existing granulocytes. In this way, they improve the immune function of existing cells while speeding the recovery of stem cell populations, thus reducing the extent and duration of the white cell nadir and total compromise of the immune system.
Studies of cancer patients suffering from neutropenia consistently show that use of these cytokines reduces infection rates, admissions, and days hospitalized 21. GM-CSF and G-CSF have both been used for treatment of ARS because their effect on granulocytes and myeloid stem cell lines do not come at the expense of other marrow cell lines. In order to achieve maximum clinical response, G-CSF or GM-CSF should be started as soon as possible after exposure 5. This provides the opportunity for maximum recovery. Cytokine administration should continue, with daily consecutive injections, to reach the desired effect of an absolute neutrophil count of 1000/μL after the absolute neutrophil count nadir.
Table 3. Comparison of granulocyte-macrophage colony-stimulating factor (GM-CSF) and granulocyte colony-stimulating factor (G-CSF)
| GM-CSF | G-CSF | |
|---|---|---|
| Upregulatinga | Effect on monocytes, tissue macrophages, and granulocytes | Only effect on granulocytes |
| Adverse effects | Fever, nausea, fatigue, headache, bone pain, myalgia | Medullary bone pain observed shortly after initiation of G-CSF treatment |
| Dosing | 200–400 mg/m2/day | 5 μg/kg body weight |
| Initiation | Promptly when significant radiation dose is suspected | Either promptly when significant radiation dose is suspected or when neutrocytes <0.5 × 109 |
| Stopping criteria | Neutrocytes increasing, eg, >1.0 × 109 ANCb | Neutrocytes increasing, eg, >1.0 × 109 ANC |
| Route of administration | Subcutaneous/Infusion/Inhalation | Subcutaneous/Infusion |
Abbreviation: ANC, absolute neutrophil count.
[Source 1]Prophylactic intervention
A number of papers document that a prompt initiation of treatment with growth factors implies an optimal outcome after radiation exposure 14, as the tissue macrophages will be transformed into fully matured immunocompetent dendritic cells in 7–10 days. This implies that preemptive intervention, or intervention initiated as early as possible after the radiation exposure, is the preferred intervention after an exposure of a high radiation dose, eg, whole-body exposure to a radiation dose of 2–3 Gy, at which time the number and function of the peripheral monocytes and neutrocytes are not yet affected. Treatment of Acute Radiation Syndrome with GM-CSF ensures both quantitative and qualitative effect on all effector cells, ie, monocytes, tissue-bound macrophages, and neutrocytes (Figure 1), as G-CSF does not stimulate local maturation of resting and immunoincompetent macrophages. In organs and tissues, these cells will mediate their front defense bastion in relation to the immune defense. The activated macrophages orchestrate the overall actions and recruitment of systemic host cells like the lymphocytic cells and neutrophils, ensuring and maintaining a normal host barrier function in respect to endogenous and external biological agents in the hypoplastic or aplastic ARS patient. Macrophages are found in all human tissues, not only in bones, skin, and mucosa (eg, of the gut, eye, peritoneum, and meninges), but also in all organs including lungs, kidneys, heart, central nervous system, pericardium, pleura, and liver. Only GM-CSF, and not C-CSF, activates macrophages by production from location-specific tissue cells. Therefore, in the sealed-off compartment of the lungs, activation of the macrophages can only be achieved by stimulation of GM-CSF, either by local production or by inhalation.
Local pulmonary host defense and Acute Radiation Syndrome
There has recently been a discussion whether recombinant proteins should be inhaled or administered systemically in order to achieve a pulmonary effect by reaching the alveolar receptors 22. The question is then, which is the preferred route when intervening with GM-CSF?
In bone marrow, stem cells, and all organs, macrophage activity is enhanced by the systemic administration of GM-CSF dosing. In a radiation disaster, the lungs receive a comparable dose of radiation as the rest of the body, but as the lungs depend solely on local endogenous GM-CSF expression by alveolar macrophages, the endogenous GMCSF produced from systemic tissues does not penetrate across the alveolar capillary barrier.
It has been documented that inhaled GM-CSF increases the number and function of phagocytic cells from bronchoalveolar lavage, but only a sparse and transient increase in the number of myeloid cells in circulation.34 When administered intravenously, however, there is only a limited response in alveolar cellularity. It follows that pulmonary innate host defense is separated from the systemic defense system in respect to GM- CSF, and that biologicsa does not penetrate from systemic circulation to the alveolar space 23. It has been documented that when administered intravenously, only 2% of smaller molecules such as recombinant antitrypsin reach the alveolar space 24. In larger molecules, such as recombinant activated protein C, there is no effect when administered intravenously,50 however when inhaled, activated protein C achieves the expected effect in the alveolus with no adverse effects 25.
An important point is whether the inhaled drug reaches the GM-CSF receptors of alveolar macrophages in the peripheral airways. By using a micropump nebulizer, sufficiently small respirable aerosol particles with a size of <2.5 μm are produced which means that a high degree of peripheral lung deposition is obtained 26.
It has been thoroughly documented that there are no known adverse effects in relation to administering inhaled GM-CSF, even when administered in very high doses 27.
Inhaled GM-CSF in antiradiation intervention in Acute Radiation Syndrome
Inhaled GM-CSF in antiradiation intervention maintains lung host defense and prevents severe pneumonia with endogenous microbiological agents such as viruses, bacteria, and fungi. Inhaled GM-CSF should be administered promptly and concomitantly with systemic intervention in the antiradiation therapy regime. The initial dose should be <300 μg/day up to 300 μg/m2 daily depending on the response 15. The inhaled drug is effective and has no adverse effect even in the very high dose range, making it highly recommendable in Acute Radiation Syndrome 19.
Treatment for internal contamination
Some treatments may reduce damage to internal organs caused by radioactive particles. Medical personnel would use these treatments only if you’ve been exposed to a specific type of radiation. These treatments include the following:
- Potassium iodide (Thyroshield, Iosat). This is a nonradioactive form of iodine. Because iodine is essential for proper thyroid function, the thyroid becomes a “destination” for iodine in the body. If you have internal contamination with radioactive iodine (radioiodine), your thyroid will absorb radioiodine just as it would other forms of iodine. Treatment with potassium iodide may fill “vacancies” in the thyroid and prevent absorption of radioiodine. The radioiodine is eventually cleared from the body in urine. Potassium iodide isn’t a cure-all and is most effective if taken within a day of exposure.
- Prussian blue (Radiogardase). This type of dye binds to particles of radioactive elements known as cesium and thallium. The radioactive particles are then excreted in feces. This treatment speeds up the elimination of the radioactive particles and reduces the amount of radiation cells may absorb.
- Diethylenetriamine pentaacetic acid (DTPA). This substance binds to metals. DTPA binds to particles of the radioactive elements plutonium, americium and curium. The radioactive particles pass out of the body in urine, thereby reducing the amount of radiation absorbed.
- Heslet L, Bay C, Nepper-Christensen S. Acute radiation syndrome (ARS) – treatment of the reduced host defense. International Journal of General Medicine. 2012;5:105-115. doi:10.2147/IJGM.S22177. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3273373/[↩][↩][↩]
- Berger ME, O’Hare FM Jr, Ricks RC, editors. The Medical Basis for Radiation Accident Preparedness: The Clinical Care of Victims. REAC/TS Conference on the Medical Basis for Radiation Accident Preparedness. New York: Parthenon Publishing; 2002.[↩]
- Jarrett DG. Medical Management of Radiological Casualties Handbook, 1st ed. Bethesda, Maryland: Armed Forces Radiobiology Research Institute (AFRRI); 1999.[↩]
- Anno GH, Baum SJ, Withers HR, Young RW. Symptomatology of acute radiation effects in humans after exposure to doses of 0.5–30 Gy. Health Phys. 1989;56(6):821–838. https://www.ncbi.nlm.nih.gov/pubmed/2722506[↩][↩]
- Mettler FA, Jr, Gus’kova AK, Gusev I. Health effects in those with acute radiation sickness from the Chernobyl accident. Health Phys. 2007;93(5):462–469. https://www.ncbi.nlm.nih.gov/pubmed/18049222[↩][↩][↩]
- LaTorre TE. Primer of Medical Radiobiology, 2nd ed. Chicago: Year Book Medical Publishers, Inc.; 1989.[↩]
- Hall EJ. Acute effects of total-body irradiation. In: Hall EJ, editor. Radiobiology for the Radiologist. 5th ed. Philadelphia, PA: Lippincott Williams and Wilkins; 2000. pp. 117–128.[↩][↩]
- Schull WJ. Effects of Atomic Radiation. New York, NY: Wiley-Liss; 1995.[↩]
- Weisdorf D, Chao N, Waselenko JK, et al. Acute radiation injury: contingency planning for triage, supportive care, and transplantation. Biol Blood Marrow Transplant. 2006;12(6):672–682. https://www.ncbi.nlm.nih.gov/pubmed/16737941[↩][↩]
- Radiation Exposure and Contamination. https://www.merckmanuals.com/professional/injuries-poisoning/radiation-exposure-and-contamination/radiation-exposure-and-contamination[↩]
- National Council on Radiation Protection and Measurements (NCRP). Management of Terrorist Events Involving Radioactive Material, NCRP Report No. 138. Bethesda, Maryland: NCRP; 2001.[↩]
- Baranov A, Gale RP, Guskova A, et al. Bone marrow transplantation after the Chernobyl nuclear accident. N Engl J Med. 1989;321(4):205–212. https://www.ncbi.nlm.nih.gov/pubmed/2664512[↩]
- Fliedner TM. Nuclear terrorism: the role of hematology in coping with its health consequences. Curr Opin Hematol. 2006;13(6):436–444. https://www.ncbi.nlm.nih.gov/pubmed/17053455[↩]
- Gourmelon P, Benderitter M, Bertho JM, Huet C, Gorin NC, De Revel P. European consensus on the medical management of acute radiation syndrome and analysis of the radiation accidents in Belgium and Senegal. Health Phys. 2010;98(6):825–832. https://www.ncbi.nlm.nih.gov/pubmed/20445389[↩][↩][↩][↩][↩]
- Waselenko JK, MacVittie TJ, Blakely WF, et al. Medical management of the acute radiation syndrome: recommendations of the Strategic National Stockpile Radiation Working Group. Ann Intern Med. 2004;140(12):1037–1051. https://www.ncbi.nlm.nih.gov/pubmed/15197022[↩][↩][↩]
- Fliedner TM, Andrews GA, Cronkite EP, Bond VP. Early and late cytologic effects of whole body irradiation on human marrow. Blood. 1964;23:471–487. http://www.bloodjournal.org/content/23/4/471.long[↩]
- Gurion R, Gafter-Gvili A, Paul M, et al. Hematopoietic growth factors in aplastic anemia patients treated with immunosuppressive therapy-systematic review and meta-analysis. Haematologica. 2009;94(5):712–719. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2675684/[↩]
- Nemunaitis J, Rabinowe SN, Singer JW, et al. Recombinant granulocyte-macrophage colony-stimulating factor after autologous bone marrow transplantation for lymphoid cancer. N Engl J Med. 1991;324(25):1773–1778. http://www.nejm.org/doi/full/10.1056/NEJM199106203242504[↩]
- Liu Q, Jiang B, Jiang LP, et al. Clinical report of three cases of acute radiation sickness from a (60)Co radiation accident in Henan Province in China. J Radiat Res (Tokyo) 2008;49(1):63–69. https://www.ncbi.nlm.nih.gov/pubmed/18187937[↩][↩]
- Mayer P, Schütze E, Lam C, Kricek F, Liehl E. Recombinant murine granulocyte-macrophage colony-stimulating factor augments neutrophil recovery and enhances resistance to infections in myelosuppressed mice. J Infect Dis. 1991;163(3):584–590. https://www.ncbi.nlm.nih.gov/pubmed/1995731[↩]
- Anderson PM, Markovic SN, Sloan JA, et al. Aerosol granulocyte macrophage-colony stimulating factor: a low toxicity, lung-specific biological therapy in patients with lung metastases. Clin Cancer Res. 1999;5(9):2316–2323. https://www.ncbi.nlm.nih.gov/pubmed/10499599[↩]
- Heslet L. Look on the “air side” in pneumonia. Crit Care Med. 2009;37(2):774–775. https://www.ncbi.nlm.nih.gov/pubmed/19325384[↩]
- Rose RM, Kobzik L, Dushay K, et al. The effect of aerosolized recombinant human granulocyte macrophage colony-stimulating factor on lung leukocytes in nonhuman primates. Am Rev Respir Dis. 1992;146(5 Pt 1):1279–1286. https://www.ncbi.nlm.nih.gov/pubmed/1443885[↩]
- Brand P, Beckmann H, Maas Enriquez M, et al. Peripheral deposition of alpha1-protease inhibitor using commercial inhalation devices. Eur Respir J. 2003;22(2):263–267. https://www.ncbi.nlm.nih.gov/pubmed/12952258[↩]
- Waerhaug K, Kuzkov VV, Kuklin VN, et al. Inhaled aerosolised recombinant human activated protein C ameliorates endotoxin-induced lung injury in anaesthetised sheep. Crit Care. 2009;13(2):R51. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2689497/[↩]
- Luisetti M, Kroneberg P, Suzuki T, et al. Physical properties, lung deposition modeling, and bioactivity of recombinant GM-CSF aerosolised with a highly efficient nebulizer. Pulm Pharmacol Ther. 2011;24(1):123–127. https://www.ncbi.nlm.nih.gov/pubmed/20728558[↩]
- Anderson PM, Markovic SN, Sloan JA, et al. Aerosol granulocyte macrophage-colony stimulating factor: a low toxicity, lung-specific biological therapy in patients with lung metastases. Clin Cancer Res. 1999;5(9):2316–2323. http://clincancerres.aacrjournals.org/content/5/9/2316.long[↩]





